User login
Gastrointestinal Symptoms and Lactic Acidosis in a Chronic Marijuana User
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
Role of Speech Pathology in a Multidisciplinary Approach to a Patient With Mild Traumatic Brain Injury
Speech-language pathologists can fill a unique need in the treatment of patients with several conditions that are seen regularly in primary care.
Speech-language pathologists (SLPs) are integral to the comprehensive treatment of mild traumatic brain injury (mTBI), yet the evaluation and treatment options they offer may not be known to all primary care providers (PCPs). As the research on the management and treatment of mTBI continues to evolve, the PCPs role in referring patients with mTBI to the appropriate resources becomes imperative.
mTBI is a common injury in both military and civilian settings, but it can be difficult to diagnose and is not always well understood. Long-term debilitating effects have been associated with mTBI, with literature linking it to an increased risk of developing Alzheimer disease, motor neuron disease, and Parkinson disease.1 In addition, mTBI is a strong predictor for the development of posttraumatic stress disorder (PTSD). Among returning Iraq and Afghanistan service members, the incidence of mTBI associated mental health conditions have been reported to be as high as 22.8%, affecting > 320,000 veterans.2-5
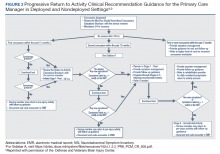
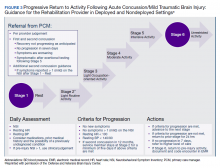
The US Department of Veteran Affairs (VA) health care system offers these returning veterans a comprehensive, multidisciplinary treatment strategy. The care is often coordinated by the veteran’s patient aligned care team (PACT) that consists of a PCP, nurses, and a medical support associate. The US Department of Defense (DoD) and VA also facilitated the development of a clinical practice guideline (CPG) that can be used by the PACT and other health care providers to support evidence based patient-centered care. This CPG is extensive and has recommendations for evaluation and treatment of mTBI and the symptoms associated such as impaired memory and alterations in executive function.6
The following hypothetical case is based on an actual patient. This case illustrates the role of speech pathology in caring for patients with mTBI.
Case Presentation
A 25-year-old male combat veteran presented to his VA PACT team for a new patient visit. As part of the screening of his medical history, mTBI was fully defined for the patient to include “alteration” in consciousness. This reminded the patient of an injury that occurred 1 year prior to presentation during a routine convoy mission. He was riding in the back of a Humvee when it hit a large pothole slamming his head into the side of the vehicle. He reported that he felt “dazed and dizzy” with “ringing” in his ears immediately following the event, without an overt loss of consciousness. He was unable to seek medical attention secondary to the urgency of the convoy mission, so he “shook it off” and kept going. Later that week he noted headache and insomnia. He was seen and evaluated by his health care provider for insomnia, but when questioned he reported no head trauma as he had forgotten the incident. Upon follow-up with his PCP, he reported his headaches were manageable, and his insomnia was somewhat improved with recommended life-style modifications and good sleep hygiene.
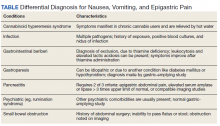
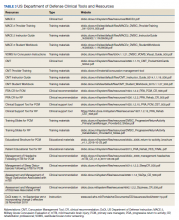
He still had frequent headaches, dizziness, and some insomnia. However, his chief concern was that he was struggling with new schoolwork. He noted that he was a straight-A student prior to his military service. A review of his medical history in his medical chart showed that a previous PCP had treated his associated symptoms of insomnia and headache without improvement. In addition, he had recently been diagnosed with PTSD. As his symptoms had lasted > 90 days, not resolved with initial treatment in primary care, and were causing a significant impact on his activities of daily living, his PCP placed a consult to Speech Pathology for cognitive-linguistic assessment and treatment, if indicated, noting that he may have had a mTBI.6 Although not intended to be comprehensive, Table 1 describes several clinical areas where a speech pathology referral may be appropriate.
The Role of the Speech-Language Pathologist
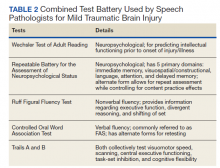
The speech-language pathologist takes an additional history of the patient. This better quantifies specific details of the veteran’s functional concerns pertaining to possible difficulty with attention, memory, executive function, visuospatial awareness, etc. Examples might include difficulty with attention/memory, including not remembering what to get at the store, forgetting to take medications, forgetting appointments, and difficulty in school, among many others. Reports of feeling “stupid” also are common. Assessment varies by clinician, but it is not uncommon for the SLP to administer a battery of evaluations to help identify a range of possible impairments. Choosing testing that is sensitive to even mild impairment is important and should be used in combination with subjective complaints. Mild deficits can sometimes be missed in those with average performance, but whose premorbid intelligence was above average. One combination of test batteries sometimes utilized is the Wechsler Test of Adult Reading (WTAR), the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), the Ruff Figural Fluency Test (RFFT), the Controlled Oral Word Association Test (COWAT), and Trails A and B (Table 2).
The initial testing results are discussed with the veteran. If patient concerns and/or testing reveal impairment that is amenable to treatment and the veteran wishes to proceed, subsequent treatment sessions are scheduled. The first treatment session is spent establishing and prioritizing functional goals specific to that individual and their needs (eg, for daily life, work, school). In a case of subacute or older mTBI, as is often seen in veterans coming to the VA, intervention often targets strategies and techniques that can help the individual compensate for current deficits.
Many patients already own a smartphone, so this device often is used functionally as a cognitive prosthetic as early as the first treatment session. In an effort to immediately start addressing important issues like medication management and attending appointments, the veteran is educated to the benefit of entering important information into the calendar and/or reminder apps on their phone and setting associated alarms that would serve as a reminder for what was entered. Patients are often encouraged by the positive impact of these initial strategies and look forward to future treatment sessions to address compensation for their functional deficits.
If a veteran with TBI has numerous needs, it can be beneficial for the care team to discuss the care plan at an interdisciplinary team meeting. It is not uncommon for veterans like the one discussed above to be referred to neurology (persistent headaches and further neurological evaluation); mental health (PTSD treatment and family support/counseling options); occupational therapy (visuospatial needs); and audiology (vestibular concerns). Social work involvement is often extremely beneficial for coordination of care in more complex cases. If patient is having difficulty making healthy eating choices or with meal preparation, a consult to a dietitian may prove invaluable. Concerns related to trouble with medication adherence (beyond memory-related adherence issues that speech pathology would address) or polypharmacy can be directed to a clinical pharmacy specialist, who could prepare a medication chart, review optimal medication timing, and provide education on adverse effects. A veteran's communication with the team can be facilitated through secure messaging (a method of secure emailing) and encouraging use of the My HealtheVet portal. With this modality, patients could review chart notes and results and share them with non-VA health care providers and/or family members as indicated.
A whole health approach also may appeal to some mTBI patients. This approach focuses on the totality of patient needs for healthy living and on patient-centered goal setting. Services provided may differ at various VA medical centers, but the PACT team can connect the veteran to the services of interest.
Conclusions
A team approach to veterans with mTBI provides a comprehensive way to treat the various problems associated with the condition. Further research into the role of multidisciplinary teams in the management of mTBI was recommended in the 2016 CPG.6 The unique role that the speech-language pathologist plays as part of this team has been highlighted, as it is important that PCP’s be aware of the extent of evaluation and treatment services they offer. Beyond mTBI, speech pathologists evaluate and treat patients with several conditions that are seen regularly in primary care.
1. McKee AC, Robinson ME. Military-related traumatic brain injury and neurodegeneration. Alzheimers Dement. 2014;10(3 suppl):S242-S253. doi:10.1016/j.jalz.2014.04.003
2. Yurgil KA, Barkauskas DA, Vasterling JJ, et al. Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty Marines. JAMA Psychiatry. 2014;71(2):149-157. doi:10.1001/jamapsychiatry.2013.3080
3. Chin DL, Zeber JE. Mental Health Outcomes Among Military Service Members After Severe Injury in Combat and TBI. Mil Med. 2020;185(5-6):e711-e718. doi:10.1093/milmed/usz440
4. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295(9):1023-1032. doi:10.1001/jama.295.9.1023
5. Miles SR, Harik JM, Hundt NE, et al. Delivery of mental health treatment to combat veterans with psychiatric diagnoses and TBI histories. PLoS One. 2017;12(9):e0184265. Published 2017 Sep 8. doi:10.1371/journal.pone.0184265
6. US Department of Defense, US Department of Veterans Affairs; Management of Concussion/mTBI Working Group. VA/DoD clinical practice guideline for management of concussion/mild traumatic brain injury. Version 2.0. Published February 2016. Accessed February 8, 2021. https://www.healthquality.va.gov/guidelines/Rehab/mtbi/mTBICPGFullCPG50821816.pdf
Speech-language pathologists can fill a unique need in the treatment of patients with several conditions that are seen regularly in primary care.
Speech-language pathologists can fill a unique need in the treatment of patients with several conditions that are seen regularly in primary care.
Speech-language pathologists (SLPs) are integral to the comprehensive treatment of mild traumatic brain injury (mTBI), yet the evaluation and treatment options they offer may not be known to all primary care providers (PCPs). As the research on the management and treatment of mTBI continues to evolve, the PCPs role in referring patients with mTBI to the appropriate resources becomes imperative.
mTBI is a common injury in both military and civilian settings, but it can be difficult to diagnose and is not always well understood. Long-term debilitating effects have been associated with mTBI, with literature linking it to an increased risk of developing Alzheimer disease, motor neuron disease, and Parkinson disease.1 In addition, mTBI is a strong predictor for the development of posttraumatic stress disorder (PTSD). Among returning Iraq and Afghanistan service members, the incidence of mTBI associated mental health conditions have been reported to be as high as 22.8%, affecting > 320,000 veterans.2-5
The US Department of Veteran Affairs (VA) health care system offers these returning veterans a comprehensive, multidisciplinary treatment strategy. The care is often coordinated by the veteran’s patient aligned care team (PACT) that consists of a PCP, nurses, and a medical support associate. The US Department of Defense (DoD) and VA also facilitated the development of a clinical practice guideline (CPG) that can be used by the PACT and other health care providers to support evidence based patient-centered care. This CPG is extensive and has recommendations for evaluation and treatment of mTBI and the symptoms associated such as impaired memory and alterations in executive function.6
The following hypothetical case is based on an actual patient. This case illustrates the role of speech pathology in caring for patients with mTBI.
Case Presentation
A 25-year-old male combat veteran presented to his VA PACT team for a new patient visit. As part of the screening of his medical history, mTBI was fully defined for the patient to include “alteration” in consciousness. This reminded the patient of an injury that occurred 1 year prior to presentation during a routine convoy mission. He was riding in the back of a Humvee when it hit a large pothole slamming his head into the side of the vehicle. He reported that he felt “dazed and dizzy” with “ringing” in his ears immediately following the event, without an overt loss of consciousness. He was unable to seek medical attention secondary to the urgency of the convoy mission, so he “shook it off” and kept going. Later that week he noted headache and insomnia. He was seen and evaluated by his health care provider for insomnia, but when questioned he reported no head trauma as he had forgotten the incident. Upon follow-up with his PCP, he reported his headaches were manageable, and his insomnia was somewhat improved with recommended life-style modifications and good sleep hygiene.
He still had frequent headaches, dizziness, and some insomnia. However, his chief concern was that he was struggling with new schoolwork. He noted that he was a straight-A student prior to his military service. A review of his medical history in his medical chart showed that a previous PCP had treated his associated symptoms of insomnia and headache without improvement. In addition, he had recently been diagnosed with PTSD. As his symptoms had lasted > 90 days, not resolved with initial treatment in primary care, and were causing a significant impact on his activities of daily living, his PCP placed a consult to Speech Pathology for cognitive-linguistic assessment and treatment, if indicated, noting that he may have had a mTBI.6 Although not intended to be comprehensive, Table 1 describes several clinical areas where a speech pathology referral may be appropriate.
The Role of the Speech-Language Pathologist
The speech-language pathologist takes an additional history of the patient. This better quantifies specific details of the veteran’s functional concerns pertaining to possible difficulty with attention, memory, executive function, visuospatial awareness, etc. Examples might include difficulty with attention/memory, including not remembering what to get at the store, forgetting to take medications, forgetting appointments, and difficulty in school, among many others. Reports of feeling “stupid” also are common. Assessment varies by clinician, but it is not uncommon for the SLP to administer a battery of evaluations to help identify a range of possible impairments. Choosing testing that is sensitive to even mild impairment is important and should be used in combination with subjective complaints. Mild deficits can sometimes be missed in those with average performance, but whose premorbid intelligence was above average. One combination of test batteries sometimes utilized is the Wechsler Test of Adult Reading (WTAR), the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), the Ruff Figural Fluency Test (RFFT), the Controlled Oral Word Association Test (COWAT), and Trails A and B (Table 2).
The initial testing results are discussed with the veteran. If patient concerns and/or testing reveal impairment that is amenable to treatment and the veteran wishes to proceed, subsequent treatment sessions are scheduled. The first treatment session is spent establishing and prioritizing functional goals specific to that individual and their needs (eg, for daily life, work, school). In a case of subacute or older mTBI, as is often seen in veterans coming to the VA, intervention often targets strategies and techniques that can help the individual compensate for current deficits.
Many patients already own a smartphone, so this device often is used functionally as a cognitive prosthetic as early as the first treatment session. In an effort to immediately start addressing important issues like medication management and attending appointments, the veteran is educated to the benefit of entering important information into the calendar and/or reminder apps on their phone and setting associated alarms that would serve as a reminder for what was entered. Patients are often encouraged by the positive impact of these initial strategies and look forward to future treatment sessions to address compensation for their functional deficits.
If a veteran with TBI has numerous needs, it can be beneficial for the care team to discuss the care plan at an interdisciplinary team meeting. It is not uncommon for veterans like the one discussed above to be referred to neurology (persistent headaches and further neurological evaluation); mental health (PTSD treatment and family support/counseling options); occupational therapy (visuospatial needs); and audiology (vestibular concerns). Social work involvement is often extremely beneficial for coordination of care in more complex cases. If patient is having difficulty making healthy eating choices or with meal preparation, a consult to a dietitian may prove invaluable. Concerns related to trouble with medication adherence (beyond memory-related adherence issues that speech pathology would address) or polypharmacy can be directed to a clinical pharmacy specialist, who could prepare a medication chart, review optimal medication timing, and provide education on adverse effects. A veteran's communication with the team can be facilitated through secure messaging (a method of secure emailing) and encouraging use of the My HealtheVet portal. With this modality, patients could review chart notes and results and share them with non-VA health care providers and/or family members as indicated.
A whole health approach also may appeal to some mTBI patients. This approach focuses on the totality of patient needs for healthy living and on patient-centered goal setting. Services provided may differ at various VA medical centers, but the PACT team can connect the veteran to the services of interest.
Conclusions
A team approach to veterans with mTBI provides a comprehensive way to treat the various problems associated with the condition. Further research into the role of multidisciplinary teams in the management of mTBI was recommended in the 2016 CPG.6 The unique role that the speech-language pathologist plays as part of this team has been highlighted, as it is important that PCP’s be aware of the extent of evaluation and treatment services they offer. Beyond mTBI, speech pathologists evaluate and treat patients with several conditions that are seen regularly in primary care.
Speech-language pathologists (SLPs) are integral to the comprehensive treatment of mild traumatic brain injury (mTBI), yet the evaluation and treatment options they offer may not be known to all primary care providers (PCPs). As the research on the management and treatment of mTBI continues to evolve, the PCPs role in referring patients with mTBI to the appropriate resources becomes imperative.
mTBI is a common injury in both military and civilian settings, but it can be difficult to diagnose and is not always well understood. Long-term debilitating effects have been associated with mTBI, with literature linking it to an increased risk of developing Alzheimer disease, motor neuron disease, and Parkinson disease.1 In addition, mTBI is a strong predictor for the development of posttraumatic stress disorder (PTSD). Among returning Iraq and Afghanistan service members, the incidence of mTBI associated mental health conditions have been reported to be as high as 22.8%, affecting > 320,000 veterans.2-5
The US Department of Veteran Affairs (VA) health care system offers these returning veterans a comprehensive, multidisciplinary treatment strategy. The care is often coordinated by the veteran’s patient aligned care team (PACT) that consists of a PCP, nurses, and a medical support associate. The US Department of Defense (DoD) and VA also facilitated the development of a clinical practice guideline (CPG) that can be used by the PACT and other health care providers to support evidence based patient-centered care. This CPG is extensive and has recommendations for evaluation and treatment of mTBI and the symptoms associated such as impaired memory and alterations in executive function.6
The following hypothetical case is based on an actual patient. This case illustrates the role of speech pathology in caring for patients with mTBI.
Case Presentation
A 25-year-old male combat veteran presented to his VA PACT team for a new patient visit. As part of the screening of his medical history, mTBI was fully defined for the patient to include “alteration” in consciousness. This reminded the patient of an injury that occurred 1 year prior to presentation during a routine convoy mission. He was riding in the back of a Humvee when it hit a large pothole slamming his head into the side of the vehicle. He reported that he felt “dazed and dizzy” with “ringing” in his ears immediately following the event, without an overt loss of consciousness. He was unable to seek medical attention secondary to the urgency of the convoy mission, so he “shook it off” and kept going. Later that week he noted headache and insomnia. He was seen and evaluated by his health care provider for insomnia, but when questioned he reported no head trauma as he had forgotten the incident. Upon follow-up with his PCP, he reported his headaches were manageable, and his insomnia was somewhat improved with recommended life-style modifications and good sleep hygiene.
He still had frequent headaches, dizziness, and some insomnia. However, his chief concern was that he was struggling with new schoolwork. He noted that he was a straight-A student prior to his military service. A review of his medical history in his medical chart showed that a previous PCP had treated his associated symptoms of insomnia and headache without improvement. In addition, he had recently been diagnosed with PTSD. As his symptoms had lasted > 90 days, not resolved with initial treatment in primary care, and were causing a significant impact on his activities of daily living, his PCP placed a consult to Speech Pathology for cognitive-linguistic assessment and treatment, if indicated, noting that he may have had a mTBI.6 Although not intended to be comprehensive, Table 1 describes several clinical areas where a speech pathology referral may be appropriate.
The Role of the Speech-Language Pathologist
The speech-language pathologist takes an additional history of the patient. This better quantifies specific details of the veteran’s functional concerns pertaining to possible difficulty with attention, memory, executive function, visuospatial awareness, etc. Examples might include difficulty with attention/memory, including not remembering what to get at the store, forgetting to take medications, forgetting appointments, and difficulty in school, among many others. Reports of feeling “stupid” also are common. Assessment varies by clinician, but it is not uncommon for the SLP to administer a battery of evaluations to help identify a range of possible impairments. Choosing testing that is sensitive to even mild impairment is important and should be used in combination with subjective complaints. Mild deficits can sometimes be missed in those with average performance, but whose premorbid intelligence was above average. One combination of test batteries sometimes utilized is the Wechsler Test of Adult Reading (WTAR), the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), the Ruff Figural Fluency Test (RFFT), the Controlled Oral Word Association Test (COWAT), and Trails A and B (Table 2).
The initial testing results are discussed with the veteran. If patient concerns and/or testing reveal impairment that is amenable to treatment and the veteran wishes to proceed, subsequent treatment sessions are scheduled. The first treatment session is spent establishing and prioritizing functional goals specific to that individual and their needs (eg, for daily life, work, school). In a case of subacute or older mTBI, as is often seen in veterans coming to the VA, intervention often targets strategies and techniques that can help the individual compensate for current deficits.
Many patients already own a smartphone, so this device often is used functionally as a cognitive prosthetic as early as the first treatment session. In an effort to immediately start addressing important issues like medication management and attending appointments, the veteran is educated to the benefit of entering important information into the calendar and/or reminder apps on their phone and setting associated alarms that would serve as a reminder for what was entered. Patients are often encouraged by the positive impact of these initial strategies and look forward to future treatment sessions to address compensation for their functional deficits.
If a veteran with TBI has numerous needs, it can be beneficial for the care team to discuss the care plan at an interdisciplinary team meeting. It is not uncommon for veterans like the one discussed above to be referred to neurology (persistent headaches and further neurological evaluation); mental health (PTSD treatment and family support/counseling options); occupational therapy (visuospatial needs); and audiology (vestibular concerns). Social work involvement is often extremely beneficial for coordination of care in more complex cases. If patient is having difficulty making healthy eating choices or with meal preparation, a consult to a dietitian may prove invaluable. Concerns related to trouble with medication adherence (beyond memory-related adherence issues that speech pathology would address) or polypharmacy can be directed to a clinical pharmacy specialist, who could prepare a medication chart, review optimal medication timing, and provide education on adverse effects. A veteran's communication with the team can be facilitated through secure messaging (a method of secure emailing) and encouraging use of the My HealtheVet portal. With this modality, patients could review chart notes and results and share them with non-VA health care providers and/or family members as indicated.
A whole health approach also may appeal to some mTBI patients. This approach focuses on the totality of patient needs for healthy living and on patient-centered goal setting. Services provided may differ at various VA medical centers, but the PACT team can connect the veteran to the services of interest.
Conclusions
A team approach to veterans with mTBI provides a comprehensive way to treat the various problems associated with the condition. Further research into the role of multidisciplinary teams in the management of mTBI was recommended in the 2016 CPG.6 The unique role that the speech-language pathologist plays as part of this team has been highlighted, as it is important that PCP’s be aware of the extent of evaluation and treatment services they offer. Beyond mTBI, speech pathologists evaluate and treat patients with several conditions that are seen regularly in primary care.
1. McKee AC, Robinson ME. Military-related traumatic brain injury and neurodegeneration. Alzheimers Dement. 2014;10(3 suppl):S242-S253. doi:10.1016/j.jalz.2014.04.003
2. Yurgil KA, Barkauskas DA, Vasterling JJ, et al. Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty Marines. JAMA Psychiatry. 2014;71(2):149-157. doi:10.1001/jamapsychiatry.2013.3080
3. Chin DL, Zeber JE. Mental Health Outcomes Among Military Service Members After Severe Injury in Combat and TBI. Mil Med. 2020;185(5-6):e711-e718. doi:10.1093/milmed/usz440
4. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295(9):1023-1032. doi:10.1001/jama.295.9.1023
5. Miles SR, Harik JM, Hundt NE, et al. Delivery of mental health treatment to combat veterans with psychiatric diagnoses and TBI histories. PLoS One. 2017;12(9):e0184265. Published 2017 Sep 8. doi:10.1371/journal.pone.0184265
6. US Department of Defense, US Department of Veterans Affairs; Management of Concussion/mTBI Working Group. VA/DoD clinical practice guideline for management of concussion/mild traumatic brain injury. Version 2.0. Published February 2016. Accessed February 8, 2021. https://www.healthquality.va.gov/guidelines/Rehab/mtbi/mTBICPGFullCPG50821816.pdf
1. McKee AC, Robinson ME. Military-related traumatic brain injury and neurodegeneration. Alzheimers Dement. 2014;10(3 suppl):S242-S253. doi:10.1016/j.jalz.2014.04.003
2. Yurgil KA, Barkauskas DA, Vasterling JJ, et al. Association between traumatic brain injury and risk of posttraumatic stress disorder in active-duty Marines. JAMA Psychiatry. 2014;71(2):149-157. doi:10.1001/jamapsychiatry.2013.3080
3. Chin DL, Zeber JE. Mental Health Outcomes Among Military Service Members After Severe Injury in Combat and TBI. Mil Med. 2020;185(5-6):e711-e718. doi:10.1093/milmed/usz440
4. Hoge CW, Auchterlonie JL, Milliken CS. Mental health problems, use of mental health services, and attrition from military service after returning from deployment to Iraq or Afghanistan. JAMA. 2006;295(9):1023-1032. doi:10.1001/jama.295.9.1023
5. Miles SR, Harik JM, Hundt NE, et al. Delivery of mental health treatment to combat veterans with psychiatric diagnoses and TBI histories. PLoS One. 2017;12(9):e0184265. Published 2017 Sep 8. doi:10.1371/journal.pone.0184265
6. US Department of Defense, US Department of Veterans Affairs; Management of Concussion/mTBI Working Group. VA/DoD clinical practice guideline for management of concussion/mild traumatic brain injury. Version 2.0. Published February 2016. Accessed February 8, 2021. https://www.healthquality.va.gov/guidelines/Rehab/mtbi/mTBICPGFullCPG50821816.pdf
Neurologic disorders ubiquitous and rising in the U.S.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
FROM JAMA NEUROLOGY
New data may help intercept head injuries in college football
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
FROM JAMA NEUROLOGY
Bedside EEG test aids prognosis in patients with brain injury
results of a new study suggest. The study showed that the use of a paradigm that measures the strength of responses to speech improved the accuracy of prognosis for these patients, compared with prognoses made solely on the basis of standard clinical characteristics.
“What we found is really compelling evidence” of the usefulness of the test, lead study author Rodika Sokoliuk, PhD, a postdoctoral researcher at the Center for Human Brain Health, University of Birmingham (England), said in an interview.
The passive measure of comprehension, which doesn’t require any other response from the patient, can reduce uncertainty at a critical phase of decision-making in the ICU, said Dr. Sokoliuk.
The study was published online Dec. 23, 2020, in Annals of Neurology.
Useful information at a time of ‘considerable prognostic uncertainty’
Accurate, early prognostication is vital for efficient stratification of patients after a TBI, the authors wrote. This can often be achieved from patient behavior and CT at admission, but some patients continue to fail to obey commands after washout of sedation.
These patients pose a significant challenge for neurologic prognostication, they noted. In these cases, clinicians and families must decide whether to “wait and see” or consider treatment withdrawal.
The authors noted that a lack of command following early in the postsedation period is associated with poor outcome, including vegetative state/unresponsive wakefulness syndrome (VS/UWS). This, they said, represents a “window of opportunity” for cessation of life-sustaining therapy at a time of considerable prognostic uncertainty.
Recent research shows that a significant proportion of unresponsive patients retain a level of cognition, and even consciousness, that isn’t evident from their external behavior – the so-called cognitive-motor dissociation.
The new study included 28 adult patients who had experienced a TBI and were admitted to the ICU of the Queen Elizabeth Hospital in Birmingham, England. The patients had a Glasgow Coma Scale motor score less than 6 (i.e., they were incapable of obeying commands). They had been sedation free for 2-7 days.
For the paradigm, researchers constructed 288 English words using the male voice of the Apple synthesizer. The words required the same amount of time to be generated (320 ms) and were monosyllabic, so the rhythms of the sounds were the same.
The words were presented in a specific order: an adjective, then a noun, then a verb, then a noun. Two words – for example, an adjective and noun – “would build a meaningful phrase,” and four words would build a sentence, said Dr. Sokoliuk.
The researchers built 72 of these four-word sentences. A trial comprised 12 of these sentences, resulting in a total of 864 four-word sentences.
Dr. Sokoliuk likened the paradigm to a rap song with a specific beat that is continually repeated. “Basically, we play 12 of these four-word sentences in a row, without any gaps,” she said.
Each sentence was played to patients, in random order, a minimum of eight and a maximum of nine times per patient throughout the experiment. The patients’ brain activity was recorded on EEG.
Dr. Sokoliuk noted that brain activity in healthy people synchronizes only with the rhythm of phrases and sentences when listeners consciously comprehend the speech. The researchers assessed the level of comprehension in the unresponsive patients by measuring the strength of this synchronicity or brain pattern.
After exclusions, 17 patients were available for outcome assessment 3 months post EEG, and 16 patients were available 6 months post EEG.
The analysis showed that outcome significantly correlated with the strength of patients’ acute cortical tracking of phrases and sentences (r > 0.6; P < .007), quantified by intertrial phase coherence.
Linear regressions revealed that the strength of this comprehension response (beta, 0.603; P = .006) significantly improved the accuracy of prognoses relative to clinical characteristics alone, such as the Glasgow Coma Scale or CT grade.
Previous studies showed that, if there is no understanding of the language used or if the subject is asleep, the brain doesn’t have the “signature” of tracking phrases and sentences, so it doesn’t have the synchronicity or the pattern of individuals with normal cognition, said Dr. Sokoliuk.
“You need a certain level of consciousness, and you need to understand the language, so your brain can actually track sentences or phrases,” she said.
Dr. Sokoliuk explained that the paradigm shows that patients are understanding the sentences and are not just hearing them.
“It’s not showing us that they only hear it, because there are no obvious gaps between the sentences; if there were gaps between sentences, it would probably only show that they hear it. It could be both, that they hear and understand it, but we wouldn’t know.”
A receiver operating characteristics analysis indicated 100% sensitivity and 80% specificity for a distinction between bad outcome (death, VS/UWS) and good outcome at 6 months.
“We could actually define a threshold of the tracking,” said Dr. Sokoliuk. “Patients who had phrases and sentences tracking below this threshold had worse outcome than those whose tracking value was above this threshold.”
The study illustrates that some posttraumatic patients who remain in an unresponsive state despite being sedation free may nevertheless comprehend speech.
The EEG paradigm approach, the authors said, may significantly reduce prognostic uncertainty in a critical phase of medical decision-making. It could also help clinicians make more appropriate decisions about whether or not to continue life-sustaining therapy and ensure more appropriate distribution of limited rehabilitation resources to patients most likely to benefit.
Dr. Sokoliuk stressed that the paradigm could be used at the bedside soon after a brain injury. “The critical thing is, we can actually use it during the acute phase, which is very important for clinical decisions about life-sustaining methods, therapy, and long-term care.”
A prognostic tool
The simple approach promises to be more accessible than fMRI, said Dr. Sokoliuk. “Putting an unresponsive coma patient in a scanner is very difficult and also much more expensive,” she said.
The next step, said Dr. Sokoliuk, is to repeat the study with a larger sample. “The number in the current study was quite small, and we can’t say if the sensitivity of the paradigm is strong enough to use it as a standard prognostic tool.”
To use it in clinical setting, “we really have to have robust measures,” she added.
She aims to conduct a collaborative study involving several institutions and more patients.
The research team plans to eventually build “an open-access toolbox” that would include the auditory streams to be played during EEG recordings and a program to analyze the data, said Dr. Sokoliuk. “Then, in the end, you would get a threshold or a value of tracking for phrases and sentences, and this could then classify a patient to be in a good-outcome or in bad-outcome group.”
She stressed this is a prognostic tool, not a diagnostic tool, and it should not be used in isolation. “It’s important to know that no clinician should only use this paradigm to prognosticate a patient; our paradigm should be part of a bigger battery of tests.”
But it could go a long way toward helping families as well as physicians. “If they know that the patient would be better in 3 months’ time, it’s easier for them to decide what should come next,” she said.
And it’s heartening to know that when families talk to their unresponsive loved one, the patient understands them, she added.
Promising basic research
Commenting on the study in an interview, Christine Blume, PhD, of the Center for Chronobiology, University of Basel (Switzerland), whose research interests include cognitive processing of patients with disorders of consciousness, described it as “very elegant and appealing” and the paradigm it used as “really promising.”
“However, we do, of course, not yet know about the prognostic value on a single-subject level, as the authors performed only group analyses,” said Dr. Blume. “This will require more extensive and perhaps even multicenter studies.”
It would also require developing a “solution” that “allows clinicians with limited time resources and perhaps lacking expert knowledge on the paradigm and the necessary analyses to apply the paradigm at bedside,” said Dr. Blume.
She agreed that a passive paradigm that helps determine whether a patient consciously understands speech, without the need for further processing, “has the potential to really improve the diagnostic process and uncover covert consciousness.”
One should bear in mind, though, that the paradigm “makes one essential assumption: that patients can understand speech,” said Dr. Blume. “For example, an aphasic patient might not understand but still be conscious.”
In this context, she added, “it’s essential to note that while the presence of a response suggests consciousness, the absence of a response does not suggest the absence of consciousness.”
Dr. Blume cautioned that the approach used in the study “is still at the stage of basic research.” Although the paradigm is promising, “I do not think it is ‘around the corner,’ ” she said.
The study was funded by the Medical Research Council. It was further supported by the National Institute for Health Research Surgical Reconstruction and Microbiology Research Center. Dr. Sokoliuk and Dr. Blume have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
results of a new study suggest. The study showed that the use of a paradigm that measures the strength of responses to speech improved the accuracy of prognosis for these patients, compared with prognoses made solely on the basis of standard clinical characteristics.
“What we found is really compelling evidence” of the usefulness of the test, lead study author Rodika Sokoliuk, PhD, a postdoctoral researcher at the Center for Human Brain Health, University of Birmingham (England), said in an interview.
The passive measure of comprehension, which doesn’t require any other response from the patient, can reduce uncertainty at a critical phase of decision-making in the ICU, said Dr. Sokoliuk.
The study was published online Dec. 23, 2020, in Annals of Neurology.
Useful information at a time of ‘considerable prognostic uncertainty’
Accurate, early prognostication is vital for efficient stratification of patients after a TBI, the authors wrote. This can often be achieved from patient behavior and CT at admission, but some patients continue to fail to obey commands after washout of sedation.
These patients pose a significant challenge for neurologic prognostication, they noted. In these cases, clinicians and families must decide whether to “wait and see” or consider treatment withdrawal.
The authors noted that a lack of command following early in the postsedation period is associated with poor outcome, including vegetative state/unresponsive wakefulness syndrome (VS/UWS). This, they said, represents a “window of opportunity” for cessation of life-sustaining therapy at a time of considerable prognostic uncertainty.
Recent research shows that a significant proportion of unresponsive patients retain a level of cognition, and even consciousness, that isn’t evident from their external behavior – the so-called cognitive-motor dissociation.
The new study included 28 adult patients who had experienced a TBI and were admitted to the ICU of the Queen Elizabeth Hospital in Birmingham, England. The patients had a Glasgow Coma Scale motor score less than 6 (i.e., they were incapable of obeying commands). They had been sedation free for 2-7 days.
For the paradigm, researchers constructed 288 English words using the male voice of the Apple synthesizer. The words required the same amount of time to be generated (320 ms) and were monosyllabic, so the rhythms of the sounds were the same.
The words were presented in a specific order: an adjective, then a noun, then a verb, then a noun. Two words – for example, an adjective and noun – “would build a meaningful phrase,” and four words would build a sentence, said Dr. Sokoliuk.
The researchers built 72 of these four-word sentences. A trial comprised 12 of these sentences, resulting in a total of 864 four-word sentences.
Dr. Sokoliuk likened the paradigm to a rap song with a specific beat that is continually repeated. “Basically, we play 12 of these four-word sentences in a row, without any gaps,” she said.
Each sentence was played to patients, in random order, a minimum of eight and a maximum of nine times per patient throughout the experiment. The patients’ brain activity was recorded on EEG.
Dr. Sokoliuk noted that brain activity in healthy people synchronizes only with the rhythm of phrases and sentences when listeners consciously comprehend the speech. The researchers assessed the level of comprehension in the unresponsive patients by measuring the strength of this synchronicity or brain pattern.
After exclusions, 17 patients were available for outcome assessment 3 months post EEG, and 16 patients were available 6 months post EEG.
The analysis showed that outcome significantly correlated with the strength of patients’ acute cortical tracking of phrases and sentences (r > 0.6; P < .007), quantified by intertrial phase coherence.
Linear regressions revealed that the strength of this comprehension response (beta, 0.603; P = .006) significantly improved the accuracy of prognoses relative to clinical characteristics alone, such as the Glasgow Coma Scale or CT grade.
Previous studies showed that, if there is no understanding of the language used or if the subject is asleep, the brain doesn’t have the “signature” of tracking phrases and sentences, so it doesn’t have the synchronicity or the pattern of individuals with normal cognition, said Dr. Sokoliuk.
“You need a certain level of consciousness, and you need to understand the language, so your brain can actually track sentences or phrases,” she said.
Dr. Sokoliuk explained that the paradigm shows that patients are understanding the sentences and are not just hearing them.
“It’s not showing us that they only hear it, because there are no obvious gaps between the sentences; if there were gaps between sentences, it would probably only show that they hear it. It could be both, that they hear and understand it, but we wouldn’t know.”
A receiver operating characteristics analysis indicated 100% sensitivity and 80% specificity for a distinction between bad outcome (death, VS/UWS) and good outcome at 6 months.
“We could actually define a threshold of the tracking,” said Dr. Sokoliuk. “Patients who had phrases and sentences tracking below this threshold had worse outcome than those whose tracking value was above this threshold.”
The study illustrates that some posttraumatic patients who remain in an unresponsive state despite being sedation free may nevertheless comprehend speech.
The EEG paradigm approach, the authors said, may significantly reduce prognostic uncertainty in a critical phase of medical decision-making. It could also help clinicians make more appropriate decisions about whether or not to continue life-sustaining therapy and ensure more appropriate distribution of limited rehabilitation resources to patients most likely to benefit.
Dr. Sokoliuk stressed that the paradigm could be used at the bedside soon after a brain injury. “The critical thing is, we can actually use it during the acute phase, which is very important for clinical decisions about life-sustaining methods, therapy, and long-term care.”
A prognostic tool
The simple approach promises to be more accessible than fMRI, said Dr. Sokoliuk. “Putting an unresponsive coma patient in a scanner is very difficult and also much more expensive,” she said.
The next step, said Dr. Sokoliuk, is to repeat the study with a larger sample. “The number in the current study was quite small, and we can’t say if the sensitivity of the paradigm is strong enough to use it as a standard prognostic tool.”
To use it in clinical setting, “we really have to have robust measures,” she added.
She aims to conduct a collaborative study involving several institutions and more patients.
The research team plans to eventually build “an open-access toolbox” that would include the auditory streams to be played during EEG recordings and a program to analyze the data, said Dr. Sokoliuk. “Then, in the end, you would get a threshold or a value of tracking for phrases and sentences, and this could then classify a patient to be in a good-outcome or in bad-outcome group.”
She stressed this is a prognostic tool, not a diagnostic tool, and it should not be used in isolation. “It’s important to know that no clinician should only use this paradigm to prognosticate a patient; our paradigm should be part of a bigger battery of tests.”
But it could go a long way toward helping families as well as physicians. “If they know that the patient would be better in 3 months’ time, it’s easier for them to decide what should come next,” she said.
And it’s heartening to know that when families talk to their unresponsive loved one, the patient understands them, she added.
Promising basic research
Commenting on the study in an interview, Christine Blume, PhD, of the Center for Chronobiology, University of Basel (Switzerland), whose research interests include cognitive processing of patients with disorders of consciousness, described it as “very elegant and appealing” and the paradigm it used as “really promising.”
“However, we do, of course, not yet know about the prognostic value on a single-subject level, as the authors performed only group analyses,” said Dr. Blume. “This will require more extensive and perhaps even multicenter studies.”
It would also require developing a “solution” that “allows clinicians with limited time resources and perhaps lacking expert knowledge on the paradigm and the necessary analyses to apply the paradigm at bedside,” said Dr. Blume.
She agreed that a passive paradigm that helps determine whether a patient consciously understands speech, without the need for further processing, “has the potential to really improve the diagnostic process and uncover covert consciousness.”
One should bear in mind, though, that the paradigm “makes one essential assumption: that patients can understand speech,” said Dr. Blume. “For example, an aphasic patient might not understand but still be conscious.”
In this context, she added, “it’s essential to note that while the presence of a response suggests consciousness, the absence of a response does not suggest the absence of consciousness.”
Dr. Blume cautioned that the approach used in the study “is still at the stage of basic research.” Although the paradigm is promising, “I do not think it is ‘around the corner,’ ” she said.
The study was funded by the Medical Research Council. It was further supported by the National Institute for Health Research Surgical Reconstruction and Microbiology Research Center. Dr. Sokoliuk and Dr. Blume have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
results of a new study suggest. The study showed that the use of a paradigm that measures the strength of responses to speech improved the accuracy of prognosis for these patients, compared with prognoses made solely on the basis of standard clinical characteristics.
“What we found is really compelling evidence” of the usefulness of the test, lead study author Rodika Sokoliuk, PhD, a postdoctoral researcher at the Center for Human Brain Health, University of Birmingham (England), said in an interview.
The passive measure of comprehension, which doesn’t require any other response from the patient, can reduce uncertainty at a critical phase of decision-making in the ICU, said Dr. Sokoliuk.
The study was published online Dec. 23, 2020, in Annals of Neurology.
Useful information at a time of ‘considerable prognostic uncertainty’
Accurate, early prognostication is vital for efficient stratification of patients after a TBI, the authors wrote. This can often be achieved from patient behavior and CT at admission, but some patients continue to fail to obey commands after washout of sedation.
These patients pose a significant challenge for neurologic prognostication, they noted. In these cases, clinicians and families must decide whether to “wait and see” or consider treatment withdrawal.
The authors noted that a lack of command following early in the postsedation period is associated with poor outcome, including vegetative state/unresponsive wakefulness syndrome (VS/UWS). This, they said, represents a “window of opportunity” for cessation of life-sustaining therapy at a time of considerable prognostic uncertainty.
Recent research shows that a significant proportion of unresponsive patients retain a level of cognition, and even consciousness, that isn’t evident from their external behavior – the so-called cognitive-motor dissociation.
The new study included 28 adult patients who had experienced a TBI and were admitted to the ICU of the Queen Elizabeth Hospital in Birmingham, England. The patients had a Glasgow Coma Scale motor score less than 6 (i.e., they were incapable of obeying commands). They had been sedation free for 2-7 days.
For the paradigm, researchers constructed 288 English words using the male voice of the Apple synthesizer. The words required the same amount of time to be generated (320 ms) and were monosyllabic, so the rhythms of the sounds were the same.
The words were presented in a specific order: an adjective, then a noun, then a verb, then a noun. Two words – for example, an adjective and noun – “would build a meaningful phrase,” and four words would build a sentence, said Dr. Sokoliuk.
The researchers built 72 of these four-word sentences. A trial comprised 12 of these sentences, resulting in a total of 864 four-word sentences.
Dr. Sokoliuk likened the paradigm to a rap song with a specific beat that is continually repeated. “Basically, we play 12 of these four-word sentences in a row, without any gaps,” she said.
Each sentence was played to patients, in random order, a minimum of eight and a maximum of nine times per patient throughout the experiment. The patients’ brain activity was recorded on EEG.
Dr. Sokoliuk noted that brain activity in healthy people synchronizes only with the rhythm of phrases and sentences when listeners consciously comprehend the speech. The researchers assessed the level of comprehension in the unresponsive patients by measuring the strength of this synchronicity or brain pattern.
After exclusions, 17 patients were available for outcome assessment 3 months post EEG, and 16 patients were available 6 months post EEG.
The analysis showed that outcome significantly correlated with the strength of patients’ acute cortical tracking of phrases and sentences (r > 0.6; P < .007), quantified by intertrial phase coherence.
Linear regressions revealed that the strength of this comprehension response (beta, 0.603; P = .006) significantly improved the accuracy of prognoses relative to clinical characteristics alone, such as the Glasgow Coma Scale or CT grade.
Previous studies showed that, if there is no understanding of the language used or if the subject is asleep, the brain doesn’t have the “signature” of tracking phrases and sentences, so it doesn’t have the synchronicity or the pattern of individuals with normal cognition, said Dr. Sokoliuk.
“You need a certain level of consciousness, and you need to understand the language, so your brain can actually track sentences or phrases,” she said.
Dr. Sokoliuk explained that the paradigm shows that patients are understanding the sentences and are not just hearing them.
“It’s not showing us that they only hear it, because there are no obvious gaps between the sentences; if there were gaps between sentences, it would probably only show that they hear it. It could be both, that they hear and understand it, but we wouldn’t know.”
A receiver operating characteristics analysis indicated 100% sensitivity and 80% specificity for a distinction between bad outcome (death, VS/UWS) and good outcome at 6 months.
“We could actually define a threshold of the tracking,” said Dr. Sokoliuk. “Patients who had phrases and sentences tracking below this threshold had worse outcome than those whose tracking value was above this threshold.”
The study illustrates that some posttraumatic patients who remain in an unresponsive state despite being sedation free may nevertheless comprehend speech.
The EEG paradigm approach, the authors said, may significantly reduce prognostic uncertainty in a critical phase of medical decision-making. It could also help clinicians make more appropriate decisions about whether or not to continue life-sustaining therapy and ensure more appropriate distribution of limited rehabilitation resources to patients most likely to benefit.
Dr. Sokoliuk stressed that the paradigm could be used at the bedside soon after a brain injury. “The critical thing is, we can actually use it during the acute phase, which is very important for clinical decisions about life-sustaining methods, therapy, and long-term care.”
A prognostic tool
The simple approach promises to be more accessible than fMRI, said Dr. Sokoliuk. “Putting an unresponsive coma patient in a scanner is very difficult and also much more expensive,” she said.
The next step, said Dr. Sokoliuk, is to repeat the study with a larger sample. “The number in the current study was quite small, and we can’t say if the sensitivity of the paradigm is strong enough to use it as a standard prognostic tool.”
To use it in clinical setting, “we really have to have robust measures,” she added.
She aims to conduct a collaborative study involving several institutions and more patients.
The research team plans to eventually build “an open-access toolbox” that would include the auditory streams to be played during EEG recordings and a program to analyze the data, said Dr. Sokoliuk. “Then, in the end, you would get a threshold or a value of tracking for phrases and sentences, and this could then classify a patient to be in a good-outcome or in bad-outcome group.”
She stressed this is a prognostic tool, not a diagnostic tool, and it should not be used in isolation. “It’s important to know that no clinician should only use this paradigm to prognosticate a patient; our paradigm should be part of a bigger battery of tests.”
But it could go a long way toward helping families as well as physicians. “If they know that the patient would be better in 3 months’ time, it’s easier for them to decide what should come next,” she said.
And it’s heartening to know that when families talk to their unresponsive loved one, the patient understands them, she added.
Promising basic research
Commenting on the study in an interview, Christine Blume, PhD, of the Center for Chronobiology, University of Basel (Switzerland), whose research interests include cognitive processing of patients with disorders of consciousness, described it as “very elegant and appealing” and the paradigm it used as “really promising.”
“However, we do, of course, not yet know about the prognostic value on a single-subject level, as the authors performed only group analyses,” said Dr. Blume. “This will require more extensive and perhaps even multicenter studies.”
It would also require developing a “solution” that “allows clinicians with limited time resources and perhaps lacking expert knowledge on the paradigm and the necessary analyses to apply the paradigm at bedside,” said Dr. Blume.
She agreed that a passive paradigm that helps determine whether a patient consciously understands speech, without the need for further processing, “has the potential to really improve the diagnostic process and uncover covert consciousness.”
One should bear in mind, though, that the paradigm “makes one essential assumption: that patients can understand speech,” said Dr. Blume. “For example, an aphasic patient might not understand but still be conscious.”
In this context, she added, “it’s essential to note that while the presence of a response suggests consciousness, the absence of a response does not suggest the absence of consciousness.”
Dr. Blume cautioned that the approach used in the study “is still at the stage of basic research.” Although the paradigm is promising, “I do not think it is ‘around the corner,’ ” she said.
The study was funded by the Medical Research Council. It was further supported by the National Institute for Health Research Surgical Reconstruction and Microbiology Research Center. Dr. Sokoliuk and Dr. Blume have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF NEUROLOGY
Concussion linked to risk for dementia, Parkinson’s disease, and ADHD
new research suggests. Results from a retrospective, population-based cohort study showed that controlling for socioeconomic status and overall health did not significantly affect this association.
The link between concussion and risk for ADHD and for mood and anxiety disorder was stronger in the women than in the men. In addition, having a history of multiple concussions strengthened the association between concussion and subsequent mood and anxiety disorder, dementia, and Parkinson’s disease compared with experiencing just one concussion.
The findings are similar to those of previous studies, noted lead author Marc P. Morissette, PhD, research assistant at the Pan Am Clinic Foundation in Winnipeg, Manitoba, Canada. “The main methodological differences separating our study from previous studies in this area is a focus on concussion-specific injuries identified from medical records and the potential for study participants to have up to 25 years of follow-up data,” said Dr. Morissette.
The findings were published online July 27 in Family Medicine and Community Health, a BMJ journal.
Almost 190,000 participants
Several studies have shown associations between head injury and increased risk for ADHD, depression, anxiety, Alzheimer’s disease, and Parkinson’s disease. However, many of these studies relied on self-reported medical history, included all forms of traumatic brain injury, and failed to adjust for preexisting health conditions.
An improved understanding of concussion and the risks associated with it could help physicians manage their patients’ long-term needs, the investigators noted.
In the current study, the researchers examined anonymized administrative health data collected between the periods of 1990–1991 and 2014–2015 in the Manitoba Population Research Data Repository at the Manitoba Center for Health Policy.
Eligible patients had been diagnosed with concussion in accordance with standard criteria. Participants were excluded if they had been diagnosed with dementia or Parkinson’s disease before the incident concussion during the study period. The investigators matched three control participants to each included patient on the basis of age, sex, and location.
Study outcome was time from index date (date of first concussion) to diagnosis of ADHD, mood and anxiety disorder, dementia, or Parkinson’s disease. The researchers controlled for socioeconomic status using the Socioeconomic Factor Index, version 2 (SEFI2), and for preexisting medical conditions using the Charlson Comorbidity Index (CCI).
The study included 28,021 men (mean age, 25 years) and 19,462 women (mean age, 30 years) in the concussion group and 81,871 men (mean age, 25 years) and 57,159 women (mean age, 30 years) in the control group. Mean SEFI2 score was approximately −0.05, and mean CCI score was approximately 0.2.
Dose effect?
Results showed that concussion was associated with an increased risk for ADHD (hazard ratio [HR], 1.39), mood and anxiety disorder (HR, 1.72), dementia (HR, 1.72), and Parkinson’s disease (HR, 1.57).
After a concussion, the risk of developing ADHD was 28% higher and the risk of developing mood and anxiety disorder was 7% higher among women than among men. Gender was not associated with risk for dementia or Parkinson’s disease after concussion.
Sustaining a second concussion increased the strength of the association with risk for dementia compared with sustaining a single concussion (HR, 1.62). Similarly, sustaining more than three concussions increased the strength of the association with the risk for mood and anxiety disorders (HR for more than three vs one concussion, 1.22) and Parkinson›s disease (HR, 3.27).
A sensitivity analysis found similar associations between concussion and risk for mood and anxiety disorder among all age groups. Younger participants were at greater risk for ADHD, however, and older participants were at greater risk for dementia and Parkinson’s disease.
Increased awareness of concussion and the outcomes of interest, along with improved diagnostic tools, may have influenced the study’s findings, Dr. Morissette noted. “The sex-based differences may be due to either pathophysiological differences in response to concussive injuries or potentially a difference in willingness to seek medical care or share symptoms, concussion-related or otherwise, with a medical professional,” he said.
“We are hopeful that our findings will encourage practitioners to be cognizant of various conditions that may present in individuals who have previously experienced a concussion,” Dr. Morissette added. “If physicians are aware of the various associations identified following a concussion, it may lead to more thorough clinical examination at initial presentation, along with more dedicated care throughout the patient’s life.”
Association versus causation
Commenting on the research, Steven Erickson, MD, sports medicine specialist at Banner–University Medicine Neuroscience Institute, Phoenix, Ariz., noted that although the study showed an association between concussion and subsequent diagnosis of ADHD, anxiety, and Parkinson’s disease, “this association should not be misconstrued as causation.” He added that the study’s conclusions “are just as likely to be due to labeling theory” or a self-fulfilling prophecy.
“Patients diagnosed with ADHD, anxiety, or Parkinson’s disease may recall concussion and associate the two diagnoses; but patients who have not previously been diagnosed with a concussion cannot draw that conclusion,” said Dr. Erickson, who was not involved with the research.
Citing the apparent gender difference in the strength of the association between concussion and the outcomes of interest, Dr. Erickson noted that women are more likely to report symptoms in general “and therefore are more likely to be diagnosed with ADHD and anxiety disorders” because of differences in reporting rather than incidence of disease.
“Further research needs to be done to definitively determine a causal relationship between concussion and any psychiatric or neurologic diagnosis,” Dr. Erickson concluded.
The study was funded by the Pan Am Clinic Foundation. Dr. Morissette and Dr. Erickson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests. Results from a retrospective, population-based cohort study showed that controlling for socioeconomic status and overall health did not significantly affect this association.
The link between concussion and risk for ADHD and for mood and anxiety disorder was stronger in the women than in the men. In addition, having a history of multiple concussions strengthened the association between concussion and subsequent mood and anxiety disorder, dementia, and Parkinson’s disease compared with experiencing just one concussion.
The findings are similar to those of previous studies, noted lead author Marc P. Morissette, PhD, research assistant at the Pan Am Clinic Foundation in Winnipeg, Manitoba, Canada. “The main methodological differences separating our study from previous studies in this area is a focus on concussion-specific injuries identified from medical records and the potential for study participants to have up to 25 years of follow-up data,” said Dr. Morissette.
The findings were published online July 27 in Family Medicine and Community Health, a BMJ journal.
Almost 190,000 participants
Several studies have shown associations between head injury and increased risk for ADHD, depression, anxiety, Alzheimer’s disease, and Parkinson’s disease. However, many of these studies relied on self-reported medical history, included all forms of traumatic brain injury, and failed to adjust for preexisting health conditions.
An improved understanding of concussion and the risks associated with it could help physicians manage their patients’ long-term needs, the investigators noted.
In the current study, the researchers examined anonymized administrative health data collected between the periods of 1990–1991 and 2014–2015 in the Manitoba Population Research Data Repository at the Manitoba Center for Health Policy.
Eligible patients had been diagnosed with concussion in accordance with standard criteria. Participants were excluded if they had been diagnosed with dementia or Parkinson’s disease before the incident concussion during the study period. The investigators matched three control participants to each included patient on the basis of age, sex, and location.
Study outcome was time from index date (date of first concussion) to diagnosis of ADHD, mood and anxiety disorder, dementia, or Parkinson’s disease. The researchers controlled for socioeconomic status using the Socioeconomic Factor Index, version 2 (SEFI2), and for preexisting medical conditions using the Charlson Comorbidity Index (CCI).
The study included 28,021 men (mean age, 25 years) and 19,462 women (mean age, 30 years) in the concussion group and 81,871 men (mean age, 25 years) and 57,159 women (mean age, 30 years) in the control group. Mean SEFI2 score was approximately −0.05, and mean CCI score was approximately 0.2.
Dose effect?
Results showed that concussion was associated with an increased risk for ADHD (hazard ratio [HR], 1.39), mood and anxiety disorder (HR, 1.72), dementia (HR, 1.72), and Parkinson’s disease (HR, 1.57).
After a concussion, the risk of developing ADHD was 28% higher and the risk of developing mood and anxiety disorder was 7% higher among women than among men. Gender was not associated with risk for dementia or Parkinson’s disease after concussion.
Sustaining a second concussion increased the strength of the association with risk for dementia compared with sustaining a single concussion (HR, 1.62). Similarly, sustaining more than three concussions increased the strength of the association with the risk for mood and anxiety disorders (HR for more than three vs one concussion, 1.22) and Parkinson›s disease (HR, 3.27).
A sensitivity analysis found similar associations between concussion and risk for mood and anxiety disorder among all age groups. Younger participants were at greater risk for ADHD, however, and older participants were at greater risk for dementia and Parkinson’s disease.
Increased awareness of concussion and the outcomes of interest, along with improved diagnostic tools, may have influenced the study’s findings, Dr. Morissette noted. “The sex-based differences may be due to either pathophysiological differences in response to concussive injuries or potentially a difference in willingness to seek medical care or share symptoms, concussion-related or otherwise, with a medical professional,” he said.
“We are hopeful that our findings will encourage practitioners to be cognizant of various conditions that may present in individuals who have previously experienced a concussion,” Dr. Morissette added. “If physicians are aware of the various associations identified following a concussion, it may lead to more thorough clinical examination at initial presentation, along with more dedicated care throughout the patient’s life.”
Association versus causation
Commenting on the research, Steven Erickson, MD, sports medicine specialist at Banner–University Medicine Neuroscience Institute, Phoenix, Ariz., noted that although the study showed an association between concussion and subsequent diagnosis of ADHD, anxiety, and Parkinson’s disease, “this association should not be misconstrued as causation.” He added that the study’s conclusions “are just as likely to be due to labeling theory” or a self-fulfilling prophecy.
“Patients diagnosed with ADHD, anxiety, or Parkinson’s disease may recall concussion and associate the two diagnoses; but patients who have not previously been diagnosed with a concussion cannot draw that conclusion,” said Dr. Erickson, who was not involved with the research.
Citing the apparent gender difference in the strength of the association between concussion and the outcomes of interest, Dr. Erickson noted that women are more likely to report symptoms in general “and therefore are more likely to be diagnosed with ADHD and anxiety disorders” because of differences in reporting rather than incidence of disease.
“Further research needs to be done to definitively determine a causal relationship between concussion and any psychiatric or neurologic diagnosis,” Dr. Erickson concluded.
The study was funded by the Pan Am Clinic Foundation. Dr. Morissette and Dr. Erickson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests. Results from a retrospective, population-based cohort study showed that controlling for socioeconomic status and overall health did not significantly affect this association.
The link between concussion and risk for ADHD and for mood and anxiety disorder was stronger in the women than in the men. In addition, having a history of multiple concussions strengthened the association between concussion and subsequent mood and anxiety disorder, dementia, and Parkinson’s disease compared with experiencing just one concussion.
The findings are similar to those of previous studies, noted lead author Marc P. Morissette, PhD, research assistant at the Pan Am Clinic Foundation in Winnipeg, Manitoba, Canada. “The main methodological differences separating our study from previous studies in this area is a focus on concussion-specific injuries identified from medical records and the potential for study participants to have up to 25 years of follow-up data,” said Dr. Morissette.
The findings were published online July 27 in Family Medicine and Community Health, a BMJ journal.
Almost 190,000 participants
Several studies have shown associations between head injury and increased risk for ADHD, depression, anxiety, Alzheimer’s disease, and Parkinson’s disease. However, many of these studies relied on self-reported medical history, included all forms of traumatic brain injury, and failed to adjust for preexisting health conditions.
An improved understanding of concussion and the risks associated with it could help physicians manage their patients’ long-term needs, the investigators noted.
In the current study, the researchers examined anonymized administrative health data collected between the periods of 1990–1991 and 2014–2015 in the Manitoba Population Research Data Repository at the Manitoba Center for Health Policy.
Eligible patients had been diagnosed with concussion in accordance with standard criteria. Participants were excluded if they had been diagnosed with dementia or Parkinson’s disease before the incident concussion during the study period. The investigators matched three control participants to each included patient on the basis of age, sex, and location.
Study outcome was time from index date (date of first concussion) to diagnosis of ADHD, mood and anxiety disorder, dementia, or Parkinson’s disease. The researchers controlled for socioeconomic status using the Socioeconomic Factor Index, version 2 (SEFI2), and for preexisting medical conditions using the Charlson Comorbidity Index (CCI).
The study included 28,021 men (mean age, 25 years) and 19,462 women (mean age, 30 years) in the concussion group and 81,871 men (mean age, 25 years) and 57,159 women (mean age, 30 years) in the control group. Mean SEFI2 score was approximately −0.05, and mean CCI score was approximately 0.2.
Dose effect?
Results showed that concussion was associated with an increased risk for ADHD (hazard ratio [HR], 1.39), mood and anxiety disorder (HR, 1.72), dementia (HR, 1.72), and Parkinson’s disease (HR, 1.57).
After a concussion, the risk of developing ADHD was 28% higher and the risk of developing mood and anxiety disorder was 7% higher among women than among men. Gender was not associated with risk for dementia or Parkinson’s disease after concussion.
Sustaining a second concussion increased the strength of the association with risk for dementia compared with sustaining a single concussion (HR, 1.62). Similarly, sustaining more than three concussions increased the strength of the association with the risk for mood and anxiety disorders (HR for more than three vs one concussion, 1.22) and Parkinson›s disease (HR, 3.27).
A sensitivity analysis found similar associations between concussion and risk for mood and anxiety disorder among all age groups. Younger participants were at greater risk for ADHD, however, and older participants were at greater risk for dementia and Parkinson’s disease.
Increased awareness of concussion and the outcomes of interest, along with improved diagnostic tools, may have influenced the study’s findings, Dr. Morissette noted. “The sex-based differences may be due to either pathophysiological differences in response to concussive injuries or potentially a difference in willingness to seek medical care or share symptoms, concussion-related or otherwise, with a medical professional,” he said.
“We are hopeful that our findings will encourage practitioners to be cognizant of various conditions that may present in individuals who have previously experienced a concussion,” Dr. Morissette added. “If physicians are aware of the various associations identified following a concussion, it may lead to more thorough clinical examination at initial presentation, along with more dedicated care throughout the patient’s life.”
Association versus causation
Commenting on the research, Steven Erickson, MD, sports medicine specialist at Banner–University Medicine Neuroscience Institute, Phoenix, Ariz., noted that although the study showed an association between concussion and subsequent diagnosis of ADHD, anxiety, and Parkinson’s disease, “this association should not be misconstrued as causation.” He added that the study’s conclusions “are just as likely to be due to labeling theory” or a self-fulfilling prophecy.
“Patients diagnosed with ADHD, anxiety, or Parkinson’s disease may recall concussion and associate the two diagnoses; but patients who have not previously been diagnosed with a concussion cannot draw that conclusion,” said Dr. Erickson, who was not involved with the research.
Citing the apparent gender difference in the strength of the association between concussion and the outcomes of interest, Dr. Erickson noted that women are more likely to report symptoms in general “and therefore are more likely to be diagnosed with ADHD and anxiety disorders” because of differences in reporting rather than incidence of disease.
“Further research needs to be done to definitively determine a causal relationship between concussion and any psychiatric or neurologic diagnosis,” Dr. Erickson concluded.
The study was funded by the Pan Am Clinic Foundation. Dr. Morissette and Dr. Erickson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
From Family Medicine and Community Health
‘Landmark’ study pushed detection of covert consciousness in TBI
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ANA 2020
Cerebral blood flow may predict children’s recovery from persistent postconcussion symptoms
, according to a study presented at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year. Furthermore, cerebral blood flow at 4-6 weeks predicts recovery during the next 4 weeks in 77% of children.
“This is the first study to examine cerebral blood flow changes in children with persistent postconcussion symptoms,” said Karen Barlow, MBChB, associate professor of biomedical sciences at the University of Queensland in St. Lucia, Australia. “Our findings support the link between neurovascular unit dysfunction and persistent postconcussion symptoms in children, potentially because of injury or dysfunction in the GABAergic interneurons.”
Quantifying cerebral tissue perfusion
At least 25% of children with concussion have persistent postconcussion symptoms at 1 month post injury. Understanding the factors that influence the speed of recovery may help clarify the biology of postconcussion symptoms and suggest new treatments. In previous research, Dr. Barlow and colleagues found that children with early recovery (i.e., recovery by 4 weeks post injury) have decreases in cerebral blood flow, when compared with normal children. Children with persistent symptoms, however, have increases in cerebral blood flow. Dr. Barlow and colleagues conducted a new study to examine how cerebral blood flow changes in children with persistent postconcussion symptoms.
The investigators recruited participants through the randomized controlled Play Game trial, which examined melatonin as a treatment for persistent postconcussion symptoms. Among the exclusion criteria were history of assault, drug or alcohol use, significant past medical or psychiatric history, concussion within the previous 3 months, and use of psychoactive medications.
Children entered the study at 4-8 weeks after injury and received treatment for 4 weeks. Participants underwent 3-D pseudo-continuous arterial spin–labeled MRI before and after the treatment period (i.e., at 5 and 10 weeks post injury). This imaging technique provides a quantitative assessment of cerebral tissue perfusion. “You can do it without manipulating the cerebral circulation, making it particularly useful for research and in children,” said Dr. Barlow.
She and her colleagues evaluated recovery using the Post-Concussion Symptom Inventory. They defined good recovery as a total score at or below baseline at 10 weeks post injury. They considered any children who did not meet this criterion to have poor recovery.
Speed of blood-flow change varied
In all, 124 children were eligible for the study, and 76 had MRIs at both time points. Fourteen participants were excluded because of motion artifacts, slice truncation, and normalization failure. The population’s average age was approximately 14 years. About half of participants were males. The first MRI was performed at 37 days post injury, and the second MRI at around 70 days post injury. Twenty-three children had good recovery.
Children with poor recovery at 10 weeks had higher relative cerebral blood flow, compared with children with good recovery. Treatment group, age, and sex did not affect the changes in relative cerebral blood flow over time. Dr. Barlow and colleagues also measured mean total gray matter cerebral blood flow. Children with poor recovery had higher cerebral blood flow at 5 and 10 weeks post injury, compared with children with good recovery. In addition, cerebral blood flow changed more slowly in participants with poor recovery, compared with those with good recovery. Logistic regression analysis indicated that the mean absolute gray matter cerebral blood flow at 4-6 weeks post injury significantly predicted which children would recover by 10 weeks post injury, with an area under the receiver operating characteristic curve of 77%.
Funders for the study included Alberta Children’s Hospital, the Canadian Institutes of Health Research, and the University of Calgary. Dr. Barlow had no disclosures or conflicts of interest.
SOURCE: Barlow K et al. CNS-ICNA 2020. Abstract PL100.
, according to a study presented at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year. Furthermore, cerebral blood flow at 4-6 weeks predicts recovery during the next 4 weeks in 77% of children.
“This is the first study to examine cerebral blood flow changes in children with persistent postconcussion symptoms,” said Karen Barlow, MBChB, associate professor of biomedical sciences at the University of Queensland in St. Lucia, Australia. “Our findings support the link between neurovascular unit dysfunction and persistent postconcussion symptoms in children, potentially because of injury or dysfunction in the GABAergic interneurons.”
Quantifying cerebral tissue perfusion
At least 25% of children with concussion have persistent postconcussion symptoms at 1 month post injury. Understanding the factors that influence the speed of recovery may help clarify the biology of postconcussion symptoms and suggest new treatments. In previous research, Dr. Barlow and colleagues found that children with early recovery (i.e., recovery by 4 weeks post injury) have decreases in cerebral blood flow, when compared with normal children. Children with persistent symptoms, however, have increases in cerebral blood flow. Dr. Barlow and colleagues conducted a new study to examine how cerebral blood flow changes in children with persistent postconcussion symptoms.
The investigators recruited participants through the randomized controlled Play Game trial, which examined melatonin as a treatment for persistent postconcussion symptoms. Among the exclusion criteria were history of assault, drug or alcohol use, significant past medical or psychiatric history, concussion within the previous 3 months, and use of psychoactive medications.
Children entered the study at 4-8 weeks after injury and received treatment for 4 weeks. Participants underwent 3-D pseudo-continuous arterial spin–labeled MRI before and after the treatment period (i.e., at 5 and 10 weeks post injury). This imaging technique provides a quantitative assessment of cerebral tissue perfusion. “You can do it without manipulating the cerebral circulation, making it particularly useful for research and in children,” said Dr. Barlow.
She and her colleagues evaluated recovery using the Post-Concussion Symptom Inventory. They defined good recovery as a total score at or below baseline at 10 weeks post injury. They considered any children who did not meet this criterion to have poor recovery.
Speed of blood-flow change varied
In all, 124 children were eligible for the study, and 76 had MRIs at both time points. Fourteen participants were excluded because of motion artifacts, slice truncation, and normalization failure. The population’s average age was approximately 14 years. About half of participants were males. The first MRI was performed at 37 days post injury, and the second MRI at around 70 days post injury. Twenty-three children had good recovery.
Children with poor recovery at 10 weeks had higher relative cerebral blood flow, compared with children with good recovery. Treatment group, age, and sex did not affect the changes in relative cerebral blood flow over time. Dr. Barlow and colleagues also measured mean total gray matter cerebral blood flow. Children with poor recovery had higher cerebral blood flow at 5 and 10 weeks post injury, compared with children with good recovery. In addition, cerebral blood flow changed more slowly in participants with poor recovery, compared with those with good recovery. Logistic regression analysis indicated that the mean absolute gray matter cerebral blood flow at 4-6 weeks post injury significantly predicted which children would recover by 10 weeks post injury, with an area under the receiver operating characteristic curve of 77%.
Funders for the study included Alberta Children’s Hospital, the Canadian Institutes of Health Research, and the University of Calgary. Dr. Barlow had no disclosures or conflicts of interest.
SOURCE: Barlow K et al. CNS-ICNA 2020. Abstract PL100.
, according to a study presented at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year. Furthermore, cerebral blood flow at 4-6 weeks predicts recovery during the next 4 weeks in 77% of children.
“This is the first study to examine cerebral blood flow changes in children with persistent postconcussion symptoms,” said Karen Barlow, MBChB, associate professor of biomedical sciences at the University of Queensland in St. Lucia, Australia. “Our findings support the link between neurovascular unit dysfunction and persistent postconcussion symptoms in children, potentially because of injury or dysfunction in the GABAergic interneurons.”
Quantifying cerebral tissue perfusion
At least 25% of children with concussion have persistent postconcussion symptoms at 1 month post injury. Understanding the factors that influence the speed of recovery may help clarify the biology of postconcussion symptoms and suggest new treatments. In previous research, Dr. Barlow and colleagues found that children with early recovery (i.e., recovery by 4 weeks post injury) have decreases in cerebral blood flow, when compared with normal children. Children with persistent symptoms, however, have increases in cerebral blood flow. Dr. Barlow and colleagues conducted a new study to examine how cerebral blood flow changes in children with persistent postconcussion symptoms.
The investigators recruited participants through the randomized controlled Play Game trial, which examined melatonin as a treatment for persistent postconcussion symptoms. Among the exclusion criteria were history of assault, drug or alcohol use, significant past medical or psychiatric history, concussion within the previous 3 months, and use of psychoactive medications.
Children entered the study at 4-8 weeks after injury and received treatment for 4 weeks. Participants underwent 3-D pseudo-continuous arterial spin–labeled MRI before and after the treatment period (i.e., at 5 and 10 weeks post injury). This imaging technique provides a quantitative assessment of cerebral tissue perfusion. “You can do it without manipulating the cerebral circulation, making it particularly useful for research and in children,” said Dr. Barlow.
She and her colleagues evaluated recovery using the Post-Concussion Symptom Inventory. They defined good recovery as a total score at or below baseline at 10 weeks post injury. They considered any children who did not meet this criterion to have poor recovery.
Speed of blood-flow change varied
In all, 124 children were eligible for the study, and 76 had MRIs at both time points. Fourteen participants were excluded because of motion artifacts, slice truncation, and normalization failure. The population’s average age was approximately 14 years. About half of participants were males. The first MRI was performed at 37 days post injury, and the second MRI at around 70 days post injury. Twenty-three children had good recovery.
Children with poor recovery at 10 weeks had higher relative cerebral blood flow, compared with children with good recovery. Treatment group, age, and sex did not affect the changes in relative cerebral blood flow over time. Dr. Barlow and colleagues also measured mean total gray matter cerebral blood flow. Children with poor recovery had higher cerebral blood flow at 5 and 10 weeks post injury, compared with children with good recovery. In addition, cerebral blood flow changed more slowly in participants with poor recovery, compared with those with good recovery. Logistic regression analysis indicated that the mean absolute gray matter cerebral blood flow at 4-6 weeks post injury significantly predicted which children would recover by 10 weeks post injury, with an area under the receiver operating characteristic curve of 77%.
Funders for the study included Alberta Children’s Hospital, the Canadian Institutes of Health Research, and the University of Calgary. Dr. Barlow had no disclosures or conflicts of interest.
SOURCE: Barlow K et al. CNS-ICNA 2020. Abstract PL100.
FROM CNS-ICNA 2020
Blood biomarkers could help predict when athletes recover from concussions
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
FROM JAMA NETWORK OPEN
Mild TBI/Concussion Clinical Tools for Providers Used Within the Department of Defense and Defense Health Agency
Traumatic brain injury (TBI) is a major health concern that can cause significant disability as well as economic and social burden. The Centers for Disease Control and Prevention (CDC) reported a 58% increase in the number of TBI-related emergency department visits, hospitalizations, and deaths from 2006 to 2014.1 In the CDC report, falls and motor vehicle accidents accounted for 52.3% and 20.4%, respectively, of all civilian TBI-related hospitalizations. In 2014, 56,800 TBIs in the US resulted in death. A large proportion of severe TBI survivors continue to experience long-term physical, cognitive, and psychologic disorders and require extensive rehabilitation, which may disrupt relationships and prevent return to work.2 About 37% of people with mild TBI (mTBI) cases and 51% of severe cases were unable to return to previous jobs. A study examining psychosocial burden found that people with a history of TBI reported greater feelings of loneliness compared with individuals without TBI.3
Within the US military, the Defense and Veterans Brain Injury Center (DVBIC) indicates that > 417,503 service members (SMs) have been diagnosed with TBI since November 2000.4 Of these, 82.4% were classified as having a mTBI, or concussion (Tables 1 and 2). The nature of combat and military training to which SMs are routinely exposed may increase the risk for sustaining a TBI. Specifically, the increased use of improvised explosives devices by enemy combatants in the recent military conflicts (ie, Operation Enduring Freedom, Operation Iraqi Freedom and Operation New Dawn) resulted in TBI being recognized as the signature injury of these conflicts and brought attention to the prevalence of concussion within the US military.5,6 In the military, the effects of concussion can decrease individual and unit effectiveness, emphasizing the importance of prompt diagnosis and proper management.7
Typically, patients recover from concussion within a few weeks of injury; however, some individuals experience symptoms that persist for months or years. Studies found that early intervention after concussion may aid in expediting recovery, stressing the importance of identifying concussion as promptly as possible.8,9 Active treatment is centered on patient education and symptom management, in addition to a progressive return to activities, as tolerated. Patient education may help validate the symptoms of some patients, as well as help to reattribute the symptoms to benign causes, leading to better outcomes.10 Since TBI is such a relevant health concern within the DoD, it is paramount for practitioners to understand what resources are available in order to identify and initiate treatment expeditiously.
This article focuses on the clinical tools used in evaluating and treating concussion, and best practices treatment guidelines for health care providers (HCPs) who are required to evaluate and treat military populations. While these resources are used for military SMs, they can also be used in veteran and civilian populations. This article showcases 3 DoD clinical tools that assist HCPs in evaluating and treating patients with TBI: (1) the Military Acute Concussion Evaluation 2 (MACE 2); (2) the Progressive Return to Activity (PRA) Clinical Recommendation (CR); and (3) the Concussion Management Tool (CMT). Additional DoD clinical tools and resources are discussed, and resources and links for the practitioner are provided for easy access and reference.
Military Acute Concussion Evaluation 2
Early concussion identification and evaluation are important steps in the treatment process to ensure timely recovery and return to duty for SMs. As such, DVBIC assembled a working group of military and civilian brain injury experts to create an evidence-based clinical practice guideline for the assessment and management of concussion in a military operational setting that could be learned and effectively used by corpsmen and combat medics in the battlefield to screen for a possible concussion.7 This team created the first version of the MACE, a clinical tool that prompted a systematic assessment of concussion related symptoms, neurologic signs, and cognitive deficits. The cognitive assessment portion was based on the standardized assessment of concussion (SAC) that had been reported by McCrea and colleagues in 1998.11 Soon after its creation, field utilization of the MACE for screening of concussion was mandated by the Army through an All Army Action (ALARACT 178/2008) and for all of the Services through the DoD Instruction (DoDI) 6490.11 published in 2014.12
The MACE has been updated several times since the original version. Most recently, the MACE was revised in 2018 to include a vestibular oculomotor assessment section, and red flags that immediately alert the HCP to the need for immediate triage referral and treatment of the patient possibly at a higher echelon of care or with more emergent evaluation.13-15 Additionally, the neurologic examination was expanded to increase clarity and comprehensiveness, including speech and balance testing. Updates made to the tool were intended to provide a more thorough and informative evaluation of the SM with suspected concussion.
This latest version, MACE 2, is designed to be used by any HCP who is treating SMs with a suspected or potential TBI, not just corpsmen and combat medics in theater. The MACE 2 is a comprehensive evaluation within a set of portable pocket cards designed to assist end-users in the proper triage of potentially concussed individuals. The DoD has specified 4 events that require a MACE 2 evaluation: (1) SM was in a vehicle associated with a blast event, collision, or roll over; (2) SM was within 50 meters of a blast; (3) anyone who sustained a direct blow to the head; or (4) when command provides direction (eg, repeated exposures to the events above or in accordance with protocols).12 Sleep deprivation, medications, and pain may affect MACE 2 results, in addition to deployment related stress, chronic stress, high adrenaline sustained over time, and additional comorbidities. This tool is most effective when used as close to the time of injury as possible but also may be used later (after 24 hours of rest) to reevaluate symptoms. The MACE 2 Instructor Guide, a student workbook, HCP training, and Vestibular/Ocular-Motor Screening (VOMS) for Concussion instructions can be found on the DVBIC website (Table 3).
Description
The MACE 2 is a brief multimodal screening tool that assists medics, corpsman, and primary care managers (PCMs) in the assessment and identification of a potential concussion (Figure 1). Embedded in the MACE 2 is the Standardized Assessment of Concussion (SAC), a well-validated sports concussion tool, and the VOMS tool as portions of the 2-part cognitive examination. The entirety of the tool has 5 sections: (1) red flags; (2) acute concussion screening; (3) cognitive examination, part 1; (4) neurologic examination; and (5) cognitive examination, part 2. The end of the MACE 2 includes sections on the scoring, instructions for International Classification of Diseases, Tenth Revision, TBI coding, and next steps following completion of the MACE 2. The latest version of this screening tool impacts TBI care in several noteworthy ways. First, it broadens the scope of users by expanding use to all medically trained personnel, allowing any provider to treat SMs in the field. Second, it combines state-of-the-science advances from the research field and reflects feedback from end-users collected during the development. Last, the MACE 2 is updated as changes in the field occur, and is currently undergoing research to better identify end-user utility and usability.
Screening Tools
• Red Flags. The red flags section aids in identifying potentially serious underlying conditions in patients presenting with Glasgow Coma Scale (GCS) between 13 and 15. A positive red flag prompts the practitioner to stop administering the MACE 2 and immediately consult a higher level of care and consider urgent evacuation. While the red flags are completed first, and advancement to later sections of the MACE 2 is dependent upon the absence of red flags, the red flags should be monitored throughout the completion of the MACE 2. Upon completion of patient demographics and red flags, the remaining sections of the MACE 2 are dedicated to acute concussion screening.
• Acute Concussion Screening. The acute concussion screening portion consists of 4 sections: description of the incident; alteration of consciousness or memory; a “check all that apply” symptom inventory; and a patient history that includes concussions within the past 12 months, headache disorders, and/or behavioral health concerns. The final portion of the acute concussion screening section provides an algorithm to identify a positive or negative concussion screen. When a negative screen is identified, the user is prompted to prescribe a 24-hour rest period and follow up with the SM based on the guidance in the CMT. A positive screen warrants the user to continue administration of the MACE 2.
Neurologic and CognitiveExaminations
• Cognitive Exam Part 1. The initial cognitive examination is designed to assess orientation to time (eg, What is the day of the week, day of the month, the month, the year, and the timeof day?) as well as immediate recall of a short list of concrete words (5 words total, repeated for 3 trials). These tests are based on other neuropsychological measures designed to assess cognitive/mental status and short-term memory.
• The Neurological Exam. The neurological exam section of the MACE 2 includes brief neuropsychologic tests such as speech fluency and word finding. Other sections within the neurological exam assess the
following: grip strength, vestibular function/balance (eg, tandem gait and single leg stance), as well as motor function (eg, pronator drift), autonomic nervous system function (eg, pupil response), and vestibular function (eye-tracking).
• Cognitive Exam Part 2. After completion of the first cognitive examination and the neurologic examination, the second part of the cognitive examination is initiated. Part 2 includes measures of short-term and working memory (eg, digits-reverse tasks, listing the months in reverse order, and a delayed recall task of the short list of concrete words presented in the first part). The final assessment is the administration of the VOMS, a tool developed from the sports concussion field and designed to measure vestibular-ocular function.13 It is critical to note that the VOMS is contraindicated if there is concern of an unstable cervical spine or absence of a trained HCP. An examination summary provides guidance on test scoring and yields a positive or negative indication for concussive injury. A positive test refers users to guidelines listed in the Concussion Management Tool for recommendations. The final page provides coding instructions for entering the results into the patient’s electronic medical record for documentation and future reference.
Progressive Return To Activities Clinical Recommendation
The Progressive Return to Activities Clinical Recommendation (PRA CR) also was developed by DVBIC for the DoD to assist military HCPs in managing SMs with concussion by providing systematic and evidence-based guidance to both prevent extended rest and promote return to full duty as quickly and safely as clinically indicated. The general guidance is to monitor the SM at each of the 6 stages in the process and safely and gradually increase activity to the next stage as tolerated. Daily symptoms are measured using the Neurobehavioral Symptom Inventory (NSI), which SMs self-administer every morning at each stage within the process.
Prior to initiation of the progressive return to activity, SM education using the educational brochure is strongly encouraged, as previous evidence suggests that it is an effective intervention during the acute stages of injury.10,11 Return to activity follows a 6 stage process, from stage 1 (rest) through stage 6 (unrestricted activity) (Table 4). Referral to rehabilitation providers (RPs) or higher care is left to the discretion of the PCM when (1) recovery is not progressing as anticipated; (2) progression is not being made within a 7-day period; or (3) symptoms worsen with time. The guidance outlined in the PRA CR is consistent with current policies and medical literature, and undergoes reviews as updates in the field emerge. The PRA for PCM, PRA for RP, Clinical Support Tool for PCM, Clinical Support Tool for RP, Training Slides for PCM, Training Slides for RP, Educational Brochure for PCM, and Patient Educational Tool for RP can be found on the DVBIC website (dvbic.dcoe.mil).
Description
To improve the clinical utility, 2 separate PRA CRs were developed specifically for PCMs (Figure 2) and RPs (Figure 3). The PRA CR for PCMs provides the initial framework to monitor SMs during recovery and gradually increase physical, cognitive, and vestibular/balance activities as symptoms improve in order to return to preinjury activities. The PRA CR for RPs outlines the approach for treating SMs who meet 1 of the following criteria: recovery is not progressing as anticipated, there is no progression in 7 days, symptoms are worsening, the SM is symptomatic after exertional testing following stage 5, or referral made per PCM judgment. Following the mandatory 24-hour rest period after a diagnosis of a concussion, progression through the PRA algorithm is based on history of concussion within the past 12 months (ie, 1, 2, or ≥ 3 concussions) and symptomatology, with varying treatment pathways depending on the SM’s responses to history and symptomology.
Guidelines
• One Concussion within Past 12 Months. Following the mandatory 24-hour rest period, if the SM is asymptomatic, then exertional testing (eg, activities such as push-ups, sit-ups, running in place, step aerobics, stationary bike, treadmill and/or hand crank) is performed at 65 to 85% of target heart rate for 2 minutes and symptoms are reassessed. If still asymptomatic, the SM may return to preinjury activity; however, if exertional testing provokes symptoms > 1 (mild) on the NSI, the SM should return to stage 1 with an additional 24 hours of rest. A second exertional test can be performed after stage 1, and if symptoms are provoked, progression through the remaining stages 2 to 5 is encouraged. Symptoms are continually monitored throughout each stage to determine whether the SM is recovered sufficiently to proceed to the next stage.
• Two Concussion Within Past 12 Months. Following the mandatory 24-hour rest period, no exertional testing is performed, and SMs move directly into stage 1 and remain at stage 1 or stage 2 for 7 consecutive days with no symptoms > 1 on the NSI before advancing through the remaining stages. Some defining features are longer rest periods (eg, 5 additional days of rest at stage 2) and additional patient education, symptom management, and follow-up.
• Three or more Concussions Within Past 12 Months. Following the 24 hour mandatory rest period, in cases where ≥ 3 concussions have occurred within a 12 month period, the recommendation is to provide guidance for symptom management rest and refer the SM to a higher level of care.
Concussion Management Tool
Beyond the initial assessment and concussion evaluation and the promotion of SMs’ timely return to duty, the DoD developed a tool to help endpoint users manage concussion, to include those with more protracted symptoms (Figure 4). The CMT assists HCPs and the SMs they treat in the management of symptoms before and after they return to duty. Specifically, the CMT is designed to be given in combination with guidelines issued by the DoD in the PRA CR but extends management of concussion to include those symptoms experienced more long-term, or symptoms that are not solely addressed during the timeline of the PRA CR. Together, the MACE 2, PRA CR, and the CMT provide endpoint users with a set of tools to comprehensively evaluate, treat, and manage concussions in SMs.
Description
The CMT provides step-by-step guidance for the initial and comprehensive management of concussion, once a diagnosis is made using assessments in the MACE 2. All types of HCPs, particularly those with limited training, such as Navy Hospital Corpsman and Army Combat Medics, are the intended clinical audience for the CMT. This tool was revised in 2019 to better align with the MACE 2, PRA CR, and other DVBIC CRs, and replaces the 2012 Concussion Management Algorithm and the 2014 Army Concussion Management in Garrison Setting Algorithm. The first 2 sections of the CMT are action cards, which provide management guidelines for acute injuries up to 7 days following injury and for comprehensive management beyond 1 week. Guidelines within the CMT partially overlap with those in the PRA CR; however, the PRA is designed for a more acute timeline, whereas the CMT focuses on symptom management following a more protracted recovery. The CMT clinical tool, provider training, instructor guide, and student workbook all can be found on the DVBIC website (Table 3).
Discussion
It is important for HCPs to have the skills and clinically relevant tools to optimize accurate TBI assessment. Early and accurate assessment and effective symptom management allows SMs to receive timely treatment based on clinical recommendations, and prevent and/or minimize secondary injury and prolonged recovery. Several longitudinal studies emphasize the benefits of early diagnosis and systematic follow-up.16-18 Prompt diagnosis, patient education, and early initiation to treatment may help optimize triage to care, mitigate prolonged symptoms by educating the patient on what to expect, and target specific symptoms early.8,10 Beyond the health outcomes of an individual SM, TBI recovery impacts unit readiness and consequently force readiness. As such, health outcomes and medical readiness are a priority of the Defense Health Agency (DHA).
The DHA priorities are, in part, based on DoD policy guidance for the management of concussion in the deployed setting. According to DoD instruction, “Medically documented mTBI/concussion in service members shall be clinically evaluated, treated, and managed according to the most current DoD clinical practice guidance for the deployed environment found in the Defense and Veterans Brain Injury Center (DVBIC) guidance, ‘Medical Providers: Clinical Tools.’”12 In 2018, the Deputy Secretary of Defense issued a memorandum regarding the comprehensive strategy and action plan for warfighter brain health.12 Therein, the memorandum acknowledges the enduring responsibility of the DoD to promote and protect the health and well-being of members of the nation’s armed forces. Particular emphasis was placed on issuing a response to the effects caused by concussive impacts and exposure to blast waves. This response resulted in a commitment by the DoD to understanding, preventing, diagnosing, and treating TBI in all forms. Taken together, the message from the secretary of defense and instruction from the DoD is clear and makes imperative the use of DoD clinical tools to accomplish this commitment.
Conclusion
This article showcases 3 of the DoD’s TBI clinical tools (MACE 2, PRA CR, and CMT) that assist HCPs in identifying and treating concussion. Over time, these tools undergo revisions according to the state of the science, and are adapted to meet the needs of clinicians and the SMs they treat. Studies are currently ongoing to better understand the effectiveness of these tools as well as to assist clinicians in making return-to-duty and/or medical separation decisions. These tools assist clinicians throughout the recovery process; from initial assessment and treatment (acute phase), as well as with symptom management (acute and protracted symptoms).
Concussion is not a homogenous condition and the experiences of the SM, including events that may cause emotional distress, other injuries and/or other factors, may further complicate the injury. Accordingly, there is no single clinical tool that can conclusively determine return-to-duty status; rather, these tools can help characterize injury, validate, and treat symptoms, which have been suggested to improve outcomes. More research and data are needed confirm the effectiveness of these tools to improve outcomes.
It is beyond the scope of this article to provide a more in-depth discussion on TBI prevention or longer term effects/care. However, there are additional, personalized tools for specific symptoms, deficits, or dysfunctions following concussion. These tools include the Management of Headache Following mTBI for PCM CR, Management of Sleep Disturbances Following mTBI for PCM CR, Assessment and Management of Visual Dysfunction Associated with mTBI CR, and Assessment and Management of Dizziness Associated mTBI CR. These tools enable endpoint users to evaluate and treat SMs as well as know when to elevate to higher levels of care.
The DoD commitment toward treating TBI influenced the development of the clinical tools highlighted in this article. They are the result of collective efforts among military and civilian TBI subject matter experts, data from medical literature and state-of-the-science research, and feedback from endpoint users to create the most effective, evidence-based tools. These tools undergo continuous review and revision to ensure alignment with the most up-to-date science within the field, to meet the needs of SMs and to continue the commitment to DoD concussion care.
Acknowledgments
This work was prepared under Contract (HT0014-19-C-0004) General Dynamics Information Technology and (W81XWH-16-F-0330) Credence Management Solutions, and is defined as U.S. Government work under Title 17 U.S.C.§101. Per Title 17 U.S.C.§105, copyright protection is not available for any work of the U.S. Government. For more information, please contact dha.DVBICinfo@mail.mil.
1. Centers for Disease Control and Prevention. Surveillance report of traumatic brain injury-related emergency department visits, hospitalizations, and deaths. https://www.cdc.gov/traumaticbraininjury/pdf/TBI-Surveillance-Report-FINAL_508.pdf. Published 2014. Accessed August 18, 2020.
2. Stocchetti N, Zanier ER. Chronic impact of traumatic brain injury on outcome and quality of life: a narrative review. Crit Care. 2016;20(1):148. Published 2016 Jun 21. doi:10.1186/s13054-016-1318-1
3. Kumar RG, Ornstein KA, Bollens-Lund E, et al. Lifetime history of traumatic brain injury is associated with increased loneliness in adults: A US nationally representative study. Int J Geriatr Psychiatry. 2020;35(5):553-563. doi:10.1002/gps.5271
4. Defense and Veterans Brain Injury Center. Worldwide DoD numbers for traumatic brain injury. 2020; https://dvbic.dcoe.mil/sites/default/files/tbi-numbers/DVBIC_WorldwideTotal_2000-2019.pdf. Updated March 10, 2020. Accessed August 18, 2020.
5. Kennedy JE, Lu LH, Reid MW, Leal FO, Cooper DB. Correlates of depression in U.S. military service members with a history of mild traumatic brain injury. Mil Med. 2019;184(suppl 1):148-154. doi:10.1093/milmed/usy321
6. Marshall KR, Holland SL, Meyer KS, Martin EM, Wilmore M, Grimes JB. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177(suppl 8):67-75. doi:10.7205/milmed-d-12-00110
7. French L, McCrea M., Baggett M. The Military Acute Concussion Evaluation. J Spec Oper Med. 2008;8(1):68-77. https://www.jsomonline.org/Publications/2008168French.pdf. Accessed August 18, 2020.
8. Kontos AP, Jorgensen-Wagers K, Trbovich AM, et al. Association of time since injury to the first clinic visit with recovery following concussion. JAMA Neurol. 2020;77(4):435-440. doi:10.1001/jamaneurol.2019.4552
9. Ponsford J, Willmott C, Rothwell A, et al. Impact of early intervention on outcome following mild head injury in adults. J Neurol Neurosurg Psychiatry. 2002;73(3):330-332. doi:10.1136/jnnp.73.3.33010.
10. Mittenberg W, Canyock EM, Condit D, Patton C. Treatment of post-concussion syndrome following mild head injury. J Clin Exp Neuropsychol. 2001;23(6):829-836. doi:10.1076/jcen.23.6.829.1022
11. McCrea M, Kelly JP, Randolph C, et al. Standardized assessment of concussion (SAC): on-site mental status evaluation of the athlete. J Head Trauma Rehabil. 1998;13(2):27-35. doi:10.1097/00001199-199804000-00005
12. US Department of Defense. Department of Defense Instruction, Number 6490.11. Policy guidance for management of mild traumatic brain injury/concussion in the deployed setting. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649011p.pdf. Updated November 26, 2019. Accessed August 18, 2020.
13. Mucha A, Collins MW, Elbin RJ, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions: preliminary findings. Am J Sports Med. 2014;42(10):2479-2486. doi:10.1177/0363546514543775
14. Defense and Veterans Brain Injury Center. Military Acute Concussion Evaluation 2 (MACE 2). https://dvbic.dcoe.mil/material/military-acute-concussion-evaluation-2-mace-2. Updated August 18, 2020. Accessed August 18, 2020.
15. US Department of Defense, Defense Health Agency. Defense and Veterans Brain Injury Center releases new concussion screening tool. https://www.health.mil/News/Articles/2019/03/15/Defense-and-Veterans-Brain-Injury-Center-releases-new-concussion-screening-tool. Published March 15, 2019. Accessed August 18, 2020.
16. Schwab K, Terrio HP, Brenner LA, et al. Epidemiology and prognosis of mild traumatic brain injury in returning soldiers: a cohort study. Neurology. 2017;88(16):1571-1579. doi:10.1212/WNL.0000000000003839
17. Mac Donald CL, Johnson AM, Wierzechowski L, et al. Outcome trends after US military concussive traumatic brain injury. J Neurotrauma. 2017;34(14):2206-2219. doi:10.1089/neu.2016.4434
18. Andelic N, Howe EI, Hellstrøm T, et al. Disability and quality of life 20 years after traumatic brain injury. Brain Behav. 2018;8(7):e01018. doi:10.1002/brb3.1018
Traumatic brain injury (TBI) is a major health concern that can cause significant disability as well as economic and social burden. The Centers for Disease Control and Prevention (CDC) reported a 58% increase in the number of TBI-related emergency department visits, hospitalizations, and deaths from 2006 to 2014.1 In the CDC report, falls and motor vehicle accidents accounted for 52.3% and 20.4%, respectively, of all civilian TBI-related hospitalizations. In 2014, 56,800 TBIs in the US resulted in death. A large proportion of severe TBI survivors continue to experience long-term physical, cognitive, and psychologic disorders and require extensive rehabilitation, which may disrupt relationships and prevent return to work.2 About 37% of people with mild TBI (mTBI) cases and 51% of severe cases were unable to return to previous jobs. A study examining psychosocial burden found that people with a history of TBI reported greater feelings of loneliness compared with individuals without TBI.3
Within the US military, the Defense and Veterans Brain Injury Center (DVBIC) indicates that > 417,503 service members (SMs) have been diagnosed with TBI since November 2000.4 Of these, 82.4% were classified as having a mTBI, or concussion (Tables 1 and 2). The nature of combat and military training to which SMs are routinely exposed may increase the risk for sustaining a TBI. Specifically, the increased use of improvised explosives devices by enemy combatants in the recent military conflicts (ie, Operation Enduring Freedom, Operation Iraqi Freedom and Operation New Dawn) resulted in TBI being recognized as the signature injury of these conflicts and brought attention to the prevalence of concussion within the US military.5,6 In the military, the effects of concussion can decrease individual and unit effectiveness, emphasizing the importance of prompt diagnosis and proper management.7
Typically, patients recover from concussion within a few weeks of injury; however, some individuals experience symptoms that persist for months or years. Studies found that early intervention after concussion may aid in expediting recovery, stressing the importance of identifying concussion as promptly as possible.8,9 Active treatment is centered on patient education and symptom management, in addition to a progressive return to activities, as tolerated. Patient education may help validate the symptoms of some patients, as well as help to reattribute the symptoms to benign causes, leading to better outcomes.10 Since TBI is such a relevant health concern within the DoD, it is paramount for practitioners to understand what resources are available in order to identify and initiate treatment expeditiously.
This article focuses on the clinical tools used in evaluating and treating concussion, and best practices treatment guidelines for health care providers (HCPs) who are required to evaluate and treat military populations. While these resources are used for military SMs, they can also be used in veteran and civilian populations. This article showcases 3 DoD clinical tools that assist HCPs in evaluating and treating patients with TBI: (1) the Military Acute Concussion Evaluation 2 (MACE 2); (2) the Progressive Return to Activity (PRA) Clinical Recommendation (CR); and (3) the Concussion Management Tool (CMT). Additional DoD clinical tools and resources are discussed, and resources and links for the practitioner are provided for easy access and reference.
Military Acute Concussion Evaluation 2
Early concussion identification and evaluation are important steps in the treatment process to ensure timely recovery and return to duty for SMs. As such, DVBIC assembled a working group of military and civilian brain injury experts to create an evidence-based clinical practice guideline for the assessment and management of concussion in a military operational setting that could be learned and effectively used by corpsmen and combat medics in the battlefield to screen for a possible concussion.7 This team created the first version of the MACE, a clinical tool that prompted a systematic assessment of concussion related symptoms, neurologic signs, and cognitive deficits. The cognitive assessment portion was based on the standardized assessment of concussion (SAC) that had been reported by McCrea and colleagues in 1998.11 Soon after its creation, field utilization of the MACE for screening of concussion was mandated by the Army through an All Army Action (ALARACT 178/2008) and for all of the Services through the DoD Instruction (DoDI) 6490.11 published in 2014.12
The MACE has been updated several times since the original version. Most recently, the MACE was revised in 2018 to include a vestibular oculomotor assessment section, and red flags that immediately alert the HCP to the need for immediate triage referral and treatment of the patient possibly at a higher echelon of care or with more emergent evaluation.13-15 Additionally, the neurologic examination was expanded to increase clarity and comprehensiveness, including speech and balance testing. Updates made to the tool were intended to provide a more thorough and informative evaluation of the SM with suspected concussion.
This latest version, MACE 2, is designed to be used by any HCP who is treating SMs with a suspected or potential TBI, not just corpsmen and combat medics in theater. The MACE 2 is a comprehensive evaluation within a set of portable pocket cards designed to assist end-users in the proper triage of potentially concussed individuals. The DoD has specified 4 events that require a MACE 2 evaluation: (1) SM was in a vehicle associated with a blast event, collision, or roll over; (2) SM was within 50 meters of a blast; (3) anyone who sustained a direct blow to the head; or (4) when command provides direction (eg, repeated exposures to the events above or in accordance with protocols).12 Sleep deprivation, medications, and pain may affect MACE 2 results, in addition to deployment related stress, chronic stress, high adrenaline sustained over time, and additional comorbidities. This tool is most effective when used as close to the time of injury as possible but also may be used later (after 24 hours of rest) to reevaluate symptoms. The MACE 2 Instructor Guide, a student workbook, HCP training, and Vestibular/Ocular-Motor Screening (VOMS) for Concussion instructions can be found on the DVBIC website (Table 3).
Description
The MACE 2 is a brief multimodal screening tool that assists medics, corpsman, and primary care managers (PCMs) in the assessment and identification of a potential concussion (Figure 1). Embedded in the MACE 2 is the Standardized Assessment of Concussion (SAC), a well-validated sports concussion tool, and the VOMS tool as portions of the 2-part cognitive examination. The entirety of the tool has 5 sections: (1) red flags; (2) acute concussion screening; (3) cognitive examination, part 1; (4) neurologic examination; and (5) cognitive examination, part 2. The end of the MACE 2 includes sections on the scoring, instructions for International Classification of Diseases, Tenth Revision, TBI coding, and next steps following completion of the MACE 2. The latest version of this screening tool impacts TBI care in several noteworthy ways. First, it broadens the scope of users by expanding use to all medically trained personnel, allowing any provider to treat SMs in the field. Second, it combines state-of-the-science advances from the research field and reflects feedback from end-users collected during the development. Last, the MACE 2 is updated as changes in the field occur, and is currently undergoing research to better identify end-user utility and usability.
Screening Tools
• Red Flags. The red flags section aids in identifying potentially serious underlying conditions in patients presenting with Glasgow Coma Scale (GCS) between 13 and 15. A positive red flag prompts the practitioner to stop administering the MACE 2 and immediately consult a higher level of care and consider urgent evacuation. While the red flags are completed first, and advancement to later sections of the MACE 2 is dependent upon the absence of red flags, the red flags should be monitored throughout the completion of the MACE 2. Upon completion of patient demographics and red flags, the remaining sections of the MACE 2 are dedicated to acute concussion screening.
• Acute Concussion Screening. The acute concussion screening portion consists of 4 sections: description of the incident; alteration of consciousness or memory; a “check all that apply” symptom inventory; and a patient history that includes concussions within the past 12 months, headache disorders, and/or behavioral health concerns. The final portion of the acute concussion screening section provides an algorithm to identify a positive or negative concussion screen. When a negative screen is identified, the user is prompted to prescribe a 24-hour rest period and follow up with the SM based on the guidance in the CMT. A positive screen warrants the user to continue administration of the MACE 2.
Neurologic and CognitiveExaminations
• Cognitive Exam Part 1. The initial cognitive examination is designed to assess orientation to time (eg, What is the day of the week, day of the month, the month, the year, and the timeof day?) as well as immediate recall of a short list of concrete words (5 words total, repeated for 3 trials). These tests are based on other neuropsychological measures designed to assess cognitive/mental status and short-term memory.
• The Neurological Exam. The neurological exam section of the MACE 2 includes brief neuropsychologic tests such as speech fluency and word finding. Other sections within the neurological exam assess the
following: grip strength, vestibular function/balance (eg, tandem gait and single leg stance), as well as motor function (eg, pronator drift), autonomic nervous system function (eg, pupil response), and vestibular function (eye-tracking).
• Cognitive Exam Part 2. After completion of the first cognitive examination and the neurologic examination, the second part of the cognitive examination is initiated. Part 2 includes measures of short-term and working memory (eg, digits-reverse tasks, listing the months in reverse order, and a delayed recall task of the short list of concrete words presented in the first part). The final assessment is the administration of the VOMS, a tool developed from the sports concussion field and designed to measure vestibular-ocular function.13 It is critical to note that the VOMS is contraindicated if there is concern of an unstable cervical spine or absence of a trained HCP. An examination summary provides guidance on test scoring and yields a positive or negative indication for concussive injury. A positive test refers users to guidelines listed in the Concussion Management Tool for recommendations. The final page provides coding instructions for entering the results into the patient’s electronic medical record for documentation and future reference.
Progressive Return To Activities Clinical Recommendation
The Progressive Return to Activities Clinical Recommendation (PRA CR) also was developed by DVBIC for the DoD to assist military HCPs in managing SMs with concussion by providing systematic and evidence-based guidance to both prevent extended rest and promote return to full duty as quickly and safely as clinically indicated. The general guidance is to monitor the SM at each of the 6 stages in the process and safely and gradually increase activity to the next stage as tolerated. Daily symptoms are measured using the Neurobehavioral Symptom Inventory (NSI), which SMs self-administer every morning at each stage within the process.
Prior to initiation of the progressive return to activity, SM education using the educational brochure is strongly encouraged, as previous evidence suggests that it is an effective intervention during the acute stages of injury.10,11 Return to activity follows a 6 stage process, from stage 1 (rest) through stage 6 (unrestricted activity) (Table 4). Referral to rehabilitation providers (RPs) or higher care is left to the discretion of the PCM when (1) recovery is not progressing as anticipated; (2) progression is not being made within a 7-day period; or (3) symptoms worsen with time. The guidance outlined in the PRA CR is consistent with current policies and medical literature, and undergoes reviews as updates in the field emerge. The PRA for PCM, PRA for RP, Clinical Support Tool for PCM, Clinical Support Tool for RP, Training Slides for PCM, Training Slides for RP, Educational Brochure for PCM, and Patient Educational Tool for RP can be found on the DVBIC website (dvbic.dcoe.mil).
Description
To improve the clinical utility, 2 separate PRA CRs were developed specifically for PCMs (Figure 2) and RPs (Figure 3). The PRA CR for PCMs provides the initial framework to monitor SMs during recovery and gradually increase physical, cognitive, and vestibular/balance activities as symptoms improve in order to return to preinjury activities. The PRA CR for RPs outlines the approach for treating SMs who meet 1 of the following criteria: recovery is not progressing as anticipated, there is no progression in 7 days, symptoms are worsening, the SM is symptomatic after exertional testing following stage 5, or referral made per PCM judgment. Following the mandatory 24-hour rest period after a diagnosis of a concussion, progression through the PRA algorithm is based on history of concussion within the past 12 months (ie, 1, 2, or ≥ 3 concussions) and symptomatology, with varying treatment pathways depending on the SM’s responses to history and symptomology.
Guidelines
• One Concussion within Past 12 Months. Following the mandatory 24-hour rest period, if the SM is asymptomatic, then exertional testing (eg, activities such as push-ups, sit-ups, running in place, step aerobics, stationary bike, treadmill and/or hand crank) is performed at 65 to 85% of target heart rate for 2 minutes and symptoms are reassessed. If still asymptomatic, the SM may return to preinjury activity; however, if exertional testing provokes symptoms > 1 (mild) on the NSI, the SM should return to stage 1 with an additional 24 hours of rest. A second exertional test can be performed after stage 1, and if symptoms are provoked, progression through the remaining stages 2 to 5 is encouraged. Symptoms are continually monitored throughout each stage to determine whether the SM is recovered sufficiently to proceed to the next stage.
• Two Concussion Within Past 12 Months. Following the mandatory 24-hour rest period, no exertional testing is performed, and SMs move directly into stage 1 and remain at stage 1 or stage 2 for 7 consecutive days with no symptoms > 1 on the NSI before advancing through the remaining stages. Some defining features are longer rest periods (eg, 5 additional days of rest at stage 2) and additional patient education, symptom management, and follow-up.
• Three or more Concussions Within Past 12 Months. Following the 24 hour mandatory rest period, in cases where ≥ 3 concussions have occurred within a 12 month period, the recommendation is to provide guidance for symptom management rest and refer the SM to a higher level of care.
Concussion Management Tool
Beyond the initial assessment and concussion evaluation and the promotion of SMs’ timely return to duty, the DoD developed a tool to help endpoint users manage concussion, to include those with more protracted symptoms (Figure 4). The CMT assists HCPs and the SMs they treat in the management of symptoms before and after they return to duty. Specifically, the CMT is designed to be given in combination with guidelines issued by the DoD in the PRA CR but extends management of concussion to include those symptoms experienced more long-term, or symptoms that are not solely addressed during the timeline of the PRA CR. Together, the MACE 2, PRA CR, and the CMT provide endpoint users with a set of tools to comprehensively evaluate, treat, and manage concussions in SMs.
Description
The CMT provides step-by-step guidance for the initial and comprehensive management of concussion, once a diagnosis is made using assessments in the MACE 2. All types of HCPs, particularly those with limited training, such as Navy Hospital Corpsman and Army Combat Medics, are the intended clinical audience for the CMT. This tool was revised in 2019 to better align with the MACE 2, PRA CR, and other DVBIC CRs, and replaces the 2012 Concussion Management Algorithm and the 2014 Army Concussion Management in Garrison Setting Algorithm. The first 2 sections of the CMT are action cards, which provide management guidelines for acute injuries up to 7 days following injury and for comprehensive management beyond 1 week. Guidelines within the CMT partially overlap with those in the PRA CR; however, the PRA is designed for a more acute timeline, whereas the CMT focuses on symptom management following a more protracted recovery. The CMT clinical tool, provider training, instructor guide, and student workbook all can be found on the DVBIC website (Table 3).
Discussion
It is important for HCPs to have the skills and clinically relevant tools to optimize accurate TBI assessment. Early and accurate assessment and effective symptom management allows SMs to receive timely treatment based on clinical recommendations, and prevent and/or minimize secondary injury and prolonged recovery. Several longitudinal studies emphasize the benefits of early diagnosis and systematic follow-up.16-18 Prompt diagnosis, patient education, and early initiation to treatment may help optimize triage to care, mitigate prolonged symptoms by educating the patient on what to expect, and target specific symptoms early.8,10 Beyond the health outcomes of an individual SM, TBI recovery impacts unit readiness and consequently force readiness. As such, health outcomes and medical readiness are a priority of the Defense Health Agency (DHA).
The DHA priorities are, in part, based on DoD policy guidance for the management of concussion in the deployed setting. According to DoD instruction, “Medically documented mTBI/concussion in service members shall be clinically evaluated, treated, and managed according to the most current DoD clinical practice guidance for the deployed environment found in the Defense and Veterans Brain Injury Center (DVBIC) guidance, ‘Medical Providers: Clinical Tools.’”12 In 2018, the Deputy Secretary of Defense issued a memorandum regarding the comprehensive strategy and action plan for warfighter brain health.12 Therein, the memorandum acknowledges the enduring responsibility of the DoD to promote and protect the health and well-being of members of the nation’s armed forces. Particular emphasis was placed on issuing a response to the effects caused by concussive impacts and exposure to blast waves. This response resulted in a commitment by the DoD to understanding, preventing, diagnosing, and treating TBI in all forms. Taken together, the message from the secretary of defense and instruction from the DoD is clear and makes imperative the use of DoD clinical tools to accomplish this commitment.
Conclusion
This article showcases 3 of the DoD’s TBI clinical tools (MACE 2, PRA CR, and CMT) that assist HCPs in identifying and treating concussion. Over time, these tools undergo revisions according to the state of the science, and are adapted to meet the needs of clinicians and the SMs they treat. Studies are currently ongoing to better understand the effectiveness of these tools as well as to assist clinicians in making return-to-duty and/or medical separation decisions. These tools assist clinicians throughout the recovery process; from initial assessment and treatment (acute phase), as well as with symptom management (acute and protracted symptoms).
Concussion is not a homogenous condition and the experiences of the SM, including events that may cause emotional distress, other injuries and/or other factors, may further complicate the injury. Accordingly, there is no single clinical tool that can conclusively determine return-to-duty status; rather, these tools can help characterize injury, validate, and treat symptoms, which have been suggested to improve outcomes. More research and data are needed confirm the effectiveness of these tools to improve outcomes.
It is beyond the scope of this article to provide a more in-depth discussion on TBI prevention or longer term effects/care. However, there are additional, personalized tools for specific symptoms, deficits, or dysfunctions following concussion. These tools include the Management of Headache Following mTBI for PCM CR, Management of Sleep Disturbances Following mTBI for PCM CR, Assessment and Management of Visual Dysfunction Associated with mTBI CR, and Assessment and Management of Dizziness Associated mTBI CR. These tools enable endpoint users to evaluate and treat SMs as well as know when to elevate to higher levels of care.
The DoD commitment toward treating TBI influenced the development of the clinical tools highlighted in this article. They are the result of collective efforts among military and civilian TBI subject matter experts, data from medical literature and state-of-the-science research, and feedback from endpoint users to create the most effective, evidence-based tools. These tools undergo continuous review and revision to ensure alignment with the most up-to-date science within the field, to meet the needs of SMs and to continue the commitment to DoD concussion care.
Acknowledgments
This work was prepared under Contract (HT0014-19-C-0004) General Dynamics Information Technology and (W81XWH-16-F-0330) Credence Management Solutions, and is defined as U.S. Government work under Title 17 U.S.C.§101. Per Title 17 U.S.C.§105, copyright protection is not available for any work of the U.S. Government. For more information, please contact dha.DVBICinfo@mail.mil.
Traumatic brain injury (TBI) is a major health concern that can cause significant disability as well as economic and social burden. The Centers for Disease Control and Prevention (CDC) reported a 58% increase in the number of TBI-related emergency department visits, hospitalizations, and deaths from 2006 to 2014.1 In the CDC report, falls and motor vehicle accidents accounted for 52.3% and 20.4%, respectively, of all civilian TBI-related hospitalizations. In 2014, 56,800 TBIs in the US resulted in death. A large proportion of severe TBI survivors continue to experience long-term physical, cognitive, and psychologic disorders and require extensive rehabilitation, which may disrupt relationships and prevent return to work.2 About 37% of people with mild TBI (mTBI) cases and 51% of severe cases were unable to return to previous jobs. A study examining psychosocial burden found that people with a history of TBI reported greater feelings of loneliness compared with individuals without TBI.3
Within the US military, the Defense and Veterans Brain Injury Center (DVBIC) indicates that > 417,503 service members (SMs) have been diagnosed with TBI since November 2000.4 Of these, 82.4% were classified as having a mTBI, or concussion (Tables 1 and 2). The nature of combat and military training to which SMs are routinely exposed may increase the risk for sustaining a TBI. Specifically, the increased use of improvised explosives devices by enemy combatants in the recent military conflicts (ie, Operation Enduring Freedom, Operation Iraqi Freedom and Operation New Dawn) resulted in TBI being recognized as the signature injury of these conflicts and brought attention to the prevalence of concussion within the US military.5,6 In the military, the effects of concussion can decrease individual and unit effectiveness, emphasizing the importance of prompt diagnosis and proper management.7
Typically, patients recover from concussion within a few weeks of injury; however, some individuals experience symptoms that persist for months or years. Studies found that early intervention after concussion may aid in expediting recovery, stressing the importance of identifying concussion as promptly as possible.8,9 Active treatment is centered on patient education and symptom management, in addition to a progressive return to activities, as tolerated. Patient education may help validate the symptoms of some patients, as well as help to reattribute the symptoms to benign causes, leading to better outcomes.10 Since TBI is such a relevant health concern within the DoD, it is paramount for practitioners to understand what resources are available in order to identify and initiate treatment expeditiously.
This article focuses on the clinical tools used in evaluating and treating concussion, and best practices treatment guidelines for health care providers (HCPs) who are required to evaluate and treat military populations. While these resources are used for military SMs, they can also be used in veteran and civilian populations. This article showcases 3 DoD clinical tools that assist HCPs in evaluating and treating patients with TBI: (1) the Military Acute Concussion Evaluation 2 (MACE 2); (2) the Progressive Return to Activity (PRA) Clinical Recommendation (CR); and (3) the Concussion Management Tool (CMT). Additional DoD clinical tools and resources are discussed, and resources and links for the practitioner are provided for easy access and reference.
Military Acute Concussion Evaluation 2
Early concussion identification and evaluation are important steps in the treatment process to ensure timely recovery and return to duty for SMs. As such, DVBIC assembled a working group of military and civilian brain injury experts to create an evidence-based clinical practice guideline for the assessment and management of concussion in a military operational setting that could be learned and effectively used by corpsmen and combat medics in the battlefield to screen for a possible concussion.7 This team created the first version of the MACE, a clinical tool that prompted a systematic assessment of concussion related symptoms, neurologic signs, and cognitive deficits. The cognitive assessment portion was based on the standardized assessment of concussion (SAC) that had been reported by McCrea and colleagues in 1998.11 Soon after its creation, field utilization of the MACE for screening of concussion was mandated by the Army through an All Army Action (ALARACT 178/2008) and for all of the Services through the DoD Instruction (DoDI) 6490.11 published in 2014.12
The MACE has been updated several times since the original version. Most recently, the MACE was revised in 2018 to include a vestibular oculomotor assessment section, and red flags that immediately alert the HCP to the need for immediate triage referral and treatment of the patient possibly at a higher echelon of care or with more emergent evaluation.13-15 Additionally, the neurologic examination was expanded to increase clarity and comprehensiveness, including speech and balance testing. Updates made to the tool were intended to provide a more thorough and informative evaluation of the SM with suspected concussion.
This latest version, MACE 2, is designed to be used by any HCP who is treating SMs with a suspected or potential TBI, not just corpsmen and combat medics in theater. The MACE 2 is a comprehensive evaluation within a set of portable pocket cards designed to assist end-users in the proper triage of potentially concussed individuals. The DoD has specified 4 events that require a MACE 2 evaluation: (1) SM was in a vehicle associated with a blast event, collision, or roll over; (2) SM was within 50 meters of a blast; (3) anyone who sustained a direct blow to the head; or (4) when command provides direction (eg, repeated exposures to the events above or in accordance with protocols).12 Sleep deprivation, medications, and pain may affect MACE 2 results, in addition to deployment related stress, chronic stress, high adrenaline sustained over time, and additional comorbidities. This tool is most effective when used as close to the time of injury as possible but also may be used later (after 24 hours of rest) to reevaluate symptoms. The MACE 2 Instructor Guide, a student workbook, HCP training, and Vestibular/Ocular-Motor Screening (VOMS) for Concussion instructions can be found on the DVBIC website (Table 3).
Description
The MACE 2 is a brief multimodal screening tool that assists medics, corpsman, and primary care managers (PCMs) in the assessment and identification of a potential concussion (Figure 1). Embedded in the MACE 2 is the Standardized Assessment of Concussion (SAC), a well-validated sports concussion tool, and the VOMS tool as portions of the 2-part cognitive examination. The entirety of the tool has 5 sections: (1) red flags; (2) acute concussion screening; (3) cognitive examination, part 1; (4) neurologic examination; and (5) cognitive examination, part 2. The end of the MACE 2 includes sections on the scoring, instructions for International Classification of Diseases, Tenth Revision, TBI coding, and next steps following completion of the MACE 2. The latest version of this screening tool impacts TBI care in several noteworthy ways. First, it broadens the scope of users by expanding use to all medically trained personnel, allowing any provider to treat SMs in the field. Second, it combines state-of-the-science advances from the research field and reflects feedback from end-users collected during the development. Last, the MACE 2 is updated as changes in the field occur, and is currently undergoing research to better identify end-user utility and usability.
Screening Tools
• Red Flags. The red flags section aids in identifying potentially serious underlying conditions in patients presenting with Glasgow Coma Scale (GCS) between 13 and 15. A positive red flag prompts the practitioner to stop administering the MACE 2 and immediately consult a higher level of care and consider urgent evacuation. While the red flags are completed first, and advancement to later sections of the MACE 2 is dependent upon the absence of red flags, the red flags should be monitored throughout the completion of the MACE 2. Upon completion of patient demographics and red flags, the remaining sections of the MACE 2 are dedicated to acute concussion screening.
• Acute Concussion Screening. The acute concussion screening portion consists of 4 sections: description of the incident; alteration of consciousness or memory; a “check all that apply” symptom inventory; and a patient history that includes concussions within the past 12 months, headache disorders, and/or behavioral health concerns. The final portion of the acute concussion screening section provides an algorithm to identify a positive or negative concussion screen. When a negative screen is identified, the user is prompted to prescribe a 24-hour rest period and follow up with the SM based on the guidance in the CMT. A positive screen warrants the user to continue administration of the MACE 2.
Neurologic and CognitiveExaminations
• Cognitive Exam Part 1. The initial cognitive examination is designed to assess orientation to time (eg, What is the day of the week, day of the month, the month, the year, and the timeof day?) as well as immediate recall of a short list of concrete words (5 words total, repeated for 3 trials). These tests are based on other neuropsychological measures designed to assess cognitive/mental status and short-term memory.
• The Neurological Exam. The neurological exam section of the MACE 2 includes brief neuropsychologic tests such as speech fluency and word finding. Other sections within the neurological exam assess the
following: grip strength, vestibular function/balance (eg, tandem gait and single leg stance), as well as motor function (eg, pronator drift), autonomic nervous system function (eg, pupil response), and vestibular function (eye-tracking).
• Cognitive Exam Part 2. After completion of the first cognitive examination and the neurologic examination, the second part of the cognitive examination is initiated. Part 2 includes measures of short-term and working memory (eg, digits-reverse tasks, listing the months in reverse order, and a delayed recall task of the short list of concrete words presented in the first part). The final assessment is the administration of the VOMS, a tool developed from the sports concussion field and designed to measure vestibular-ocular function.13 It is critical to note that the VOMS is contraindicated if there is concern of an unstable cervical spine or absence of a trained HCP. An examination summary provides guidance on test scoring and yields a positive or negative indication for concussive injury. A positive test refers users to guidelines listed in the Concussion Management Tool for recommendations. The final page provides coding instructions for entering the results into the patient’s electronic medical record for documentation and future reference.
Progressive Return To Activities Clinical Recommendation
The Progressive Return to Activities Clinical Recommendation (PRA CR) also was developed by DVBIC for the DoD to assist military HCPs in managing SMs with concussion by providing systematic and evidence-based guidance to both prevent extended rest and promote return to full duty as quickly and safely as clinically indicated. The general guidance is to monitor the SM at each of the 6 stages in the process and safely and gradually increase activity to the next stage as tolerated. Daily symptoms are measured using the Neurobehavioral Symptom Inventory (NSI), which SMs self-administer every morning at each stage within the process.
Prior to initiation of the progressive return to activity, SM education using the educational brochure is strongly encouraged, as previous evidence suggests that it is an effective intervention during the acute stages of injury.10,11 Return to activity follows a 6 stage process, from stage 1 (rest) through stage 6 (unrestricted activity) (Table 4). Referral to rehabilitation providers (RPs) or higher care is left to the discretion of the PCM when (1) recovery is not progressing as anticipated; (2) progression is not being made within a 7-day period; or (3) symptoms worsen with time. The guidance outlined in the PRA CR is consistent with current policies and medical literature, and undergoes reviews as updates in the field emerge. The PRA for PCM, PRA for RP, Clinical Support Tool for PCM, Clinical Support Tool for RP, Training Slides for PCM, Training Slides for RP, Educational Brochure for PCM, and Patient Educational Tool for RP can be found on the DVBIC website (dvbic.dcoe.mil).
Description
To improve the clinical utility, 2 separate PRA CRs were developed specifically for PCMs (Figure 2) and RPs (Figure 3). The PRA CR for PCMs provides the initial framework to monitor SMs during recovery and gradually increase physical, cognitive, and vestibular/balance activities as symptoms improve in order to return to preinjury activities. The PRA CR for RPs outlines the approach for treating SMs who meet 1 of the following criteria: recovery is not progressing as anticipated, there is no progression in 7 days, symptoms are worsening, the SM is symptomatic after exertional testing following stage 5, or referral made per PCM judgment. Following the mandatory 24-hour rest period after a diagnosis of a concussion, progression through the PRA algorithm is based on history of concussion within the past 12 months (ie, 1, 2, or ≥ 3 concussions) and symptomatology, with varying treatment pathways depending on the SM’s responses to history and symptomology.
Guidelines
• One Concussion within Past 12 Months. Following the mandatory 24-hour rest period, if the SM is asymptomatic, then exertional testing (eg, activities such as push-ups, sit-ups, running in place, step aerobics, stationary bike, treadmill and/or hand crank) is performed at 65 to 85% of target heart rate for 2 minutes and symptoms are reassessed. If still asymptomatic, the SM may return to preinjury activity; however, if exertional testing provokes symptoms > 1 (mild) on the NSI, the SM should return to stage 1 with an additional 24 hours of rest. A second exertional test can be performed after stage 1, and if symptoms are provoked, progression through the remaining stages 2 to 5 is encouraged. Symptoms are continually monitored throughout each stage to determine whether the SM is recovered sufficiently to proceed to the next stage.
• Two Concussion Within Past 12 Months. Following the mandatory 24-hour rest period, no exertional testing is performed, and SMs move directly into stage 1 and remain at stage 1 or stage 2 for 7 consecutive days with no symptoms > 1 on the NSI before advancing through the remaining stages. Some defining features are longer rest periods (eg, 5 additional days of rest at stage 2) and additional patient education, symptom management, and follow-up.
• Three or more Concussions Within Past 12 Months. Following the 24 hour mandatory rest period, in cases where ≥ 3 concussions have occurred within a 12 month period, the recommendation is to provide guidance for symptom management rest and refer the SM to a higher level of care.
Concussion Management Tool
Beyond the initial assessment and concussion evaluation and the promotion of SMs’ timely return to duty, the DoD developed a tool to help endpoint users manage concussion, to include those with more protracted symptoms (Figure 4). The CMT assists HCPs and the SMs they treat in the management of symptoms before and after they return to duty. Specifically, the CMT is designed to be given in combination with guidelines issued by the DoD in the PRA CR but extends management of concussion to include those symptoms experienced more long-term, or symptoms that are not solely addressed during the timeline of the PRA CR. Together, the MACE 2, PRA CR, and the CMT provide endpoint users with a set of tools to comprehensively evaluate, treat, and manage concussions in SMs.
Description
The CMT provides step-by-step guidance for the initial and comprehensive management of concussion, once a diagnosis is made using assessments in the MACE 2. All types of HCPs, particularly those with limited training, such as Navy Hospital Corpsman and Army Combat Medics, are the intended clinical audience for the CMT. This tool was revised in 2019 to better align with the MACE 2, PRA CR, and other DVBIC CRs, and replaces the 2012 Concussion Management Algorithm and the 2014 Army Concussion Management in Garrison Setting Algorithm. The first 2 sections of the CMT are action cards, which provide management guidelines for acute injuries up to 7 days following injury and for comprehensive management beyond 1 week. Guidelines within the CMT partially overlap with those in the PRA CR; however, the PRA is designed for a more acute timeline, whereas the CMT focuses on symptom management following a more protracted recovery. The CMT clinical tool, provider training, instructor guide, and student workbook all can be found on the DVBIC website (Table 3).
Discussion
It is important for HCPs to have the skills and clinically relevant tools to optimize accurate TBI assessment. Early and accurate assessment and effective symptom management allows SMs to receive timely treatment based on clinical recommendations, and prevent and/or minimize secondary injury and prolonged recovery. Several longitudinal studies emphasize the benefits of early diagnosis and systematic follow-up.16-18 Prompt diagnosis, patient education, and early initiation to treatment may help optimize triage to care, mitigate prolonged symptoms by educating the patient on what to expect, and target specific symptoms early.8,10 Beyond the health outcomes of an individual SM, TBI recovery impacts unit readiness and consequently force readiness. As such, health outcomes and medical readiness are a priority of the Defense Health Agency (DHA).
The DHA priorities are, in part, based on DoD policy guidance for the management of concussion in the deployed setting. According to DoD instruction, “Medically documented mTBI/concussion in service members shall be clinically evaluated, treated, and managed according to the most current DoD clinical practice guidance for the deployed environment found in the Defense and Veterans Brain Injury Center (DVBIC) guidance, ‘Medical Providers: Clinical Tools.’”12 In 2018, the Deputy Secretary of Defense issued a memorandum regarding the comprehensive strategy and action plan for warfighter brain health.12 Therein, the memorandum acknowledges the enduring responsibility of the DoD to promote and protect the health and well-being of members of the nation’s armed forces. Particular emphasis was placed on issuing a response to the effects caused by concussive impacts and exposure to blast waves. This response resulted in a commitment by the DoD to understanding, preventing, diagnosing, and treating TBI in all forms. Taken together, the message from the secretary of defense and instruction from the DoD is clear and makes imperative the use of DoD clinical tools to accomplish this commitment.
Conclusion
This article showcases 3 of the DoD’s TBI clinical tools (MACE 2, PRA CR, and CMT) that assist HCPs in identifying and treating concussion. Over time, these tools undergo revisions according to the state of the science, and are adapted to meet the needs of clinicians and the SMs they treat. Studies are currently ongoing to better understand the effectiveness of these tools as well as to assist clinicians in making return-to-duty and/or medical separation decisions. These tools assist clinicians throughout the recovery process; from initial assessment and treatment (acute phase), as well as with symptom management (acute and protracted symptoms).
Concussion is not a homogenous condition and the experiences of the SM, including events that may cause emotional distress, other injuries and/or other factors, may further complicate the injury. Accordingly, there is no single clinical tool that can conclusively determine return-to-duty status; rather, these tools can help characterize injury, validate, and treat symptoms, which have been suggested to improve outcomes. More research and data are needed confirm the effectiveness of these tools to improve outcomes.
It is beyond the scope of this article to provide a more in-depth discussion on TBI prevention or longer term effects/care. However, there are additional, personalized tools for specific symptoms, deficits, or dysfunctions following concussion. These tools include the Management of Headache Following mTBI for PCM CR, Management of Sleep Disturbances Following mTBI for PCM CR, Assessment and Management of Visual Dysfunction Associated with mTBI CR, and Assessment and Management of Dizziness Associated mTBI CR. These tools enable endpoint users to evaluate and treat SMs as well as know when to elevate to higher levels of care.
The DoD commitment toward treating TBI influenced the development of the clinical tools highlighted in this article. They are the result of collective efforts among military and civilian TBI subject matter experts, data from medical literature and state-of-the-science research, and feedback from endpoint users to create the most effective, evidence-based tools. These tools undergo continuous review and revision to ensure alignment with the most up-to-date science within the field, to meet the needs of SMs and to continue the commitment to DoD concussion care.
Acknowledgments
This work was prepared under Contract (HT0014-19-C-0004) General Dynamics Information Technology and (W81XWH-16-F-0330) Credence Management Solutions, and is defined as U.S. Government work under Title 17 U.S.C.§101. Per Title 17 U.S.C.§105, copyright protection is not available for any work of the U.S. Government. For more information, please contact dha.DVBICinfo@mail.mil.
1. Centers for Disease Control and Prevention. Surveillance report of traumatic brain injury-related emergency department visits, hospitalizations, and deaths. https://www.cdc.gov/traumaticbraininjury/pdf/TBI-Surveillance-Report-FINAL_508.pdf. Published 2014. Accessed August 18, 2020.
2. Stocchetti N, Zanier ER. Chronic impact of traumatic brain injury on outcome and quality of life: a narrative review. Crit Care. 2016;20(1):148. Published 2016 Jun 21. doi:10.1186/s13054-016-1318-1
3. Kumar RG, Ornstein KA, Bollens-Lund E, et al. Lifetime history of traumatic brain injury is associated with increased loneliness in adults: A US nationally representative study. Int J Geriatr Psychiatry. 2020;35(5):553-563. doi:10.1002/gps.5271
4. Defense and Veterans Brain Injury Center. Worldwide DoD numbers for traumatic brain injury. 2020; https://dvbic.dcoe.mil/sites/default/files/tbi-numbers/DVBIC_WorldwideTotal_2000-2019.pdf. Updated March 10, 2020. Accessed August 18, 2020.
5. Kennedy JE, Lu LH, Reid MW, Leal FO, Cooper DB. Correlates of depression in U.S. military service members with a history of mild traumatic brain injury. Mil Med. 2019;184(suppl 1):148-154. doi:10.1093/milmed/usy321
6. Marshall KR, Holland SL, Meyer KS, Martin EM, Wilmore M, Grimes JB. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177(suppl 8):67-75. doi:10.7205/milmed-d-12-00110
7. French L, McCrea M., Baggett M. The Military Acute Concussion Evaluation. J Spec Oper Med. 2008;8(1):68-77. https://www.jsomonline.org/Publications/2008168French.pdf. Accessed August 18, 2020.
8. Kontos AP, Jorgensen-Wagers K, Trbovich AM, et al. Association of time since injury to the first clinic visit with recovery following concussion. JAMA Neurol. 2020;77(4):435-440. doi:10.1001/jamaneurol.2019.4552
9. Ponsford J, Willmott C, Rothwell A, et al. Impact of early intervention on outcome following mild head injury in adults. J Neurol Neurosurg Psychiatry. 2002;73(3):330-332. doi:10.1136/jnnp.73.3.33010.
10. Mittenberg W, Canyock EM, Condit D, Patton C. Treatment of post-concussion syndrome following mild head injury. J Clin Exp Neuropsychol. 2001;23(6):829-836. doi:10.1076/jcen.23.6.829.1022
11. McCrea M, Kelly JP, Randolph C, et al. Standardized assessment of concussion (SAC): on-site mental status evaluation of the athlete. J Head Trauma Rehabil. 1998;13(2):27-35. doi:10.1097/00001199-199804000-00005
12. US Department of Defense. Department of Defense Instruction, Number 6490.11. Policy guidance for management of mild traumatic brain injury/concussion in the deployed setting. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649011p.pdf. Updated November 26, 2019. Accessed August 18, 2020.
13. Mucha A, Collins MW, Elbin RJ, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions: preliminary findings. Am J Sports Med. 2014;42(10):2479-2486. doi:10.1177/0363546514543775
14. Defense and Veterans Brain Injury Center. Military Acute Concussion Evaluation 2 (MACE 2). https://dvbic.dcoe.mil/material/military-acute-concussion-evaluation-2-mace-2. Updated August 18, 2020. Accessed August 18, 2020.
15. US Department of Defense, Defense Health Agency. Defense and Veterans Brain Injury Center releases new concussion screening tool. https://www.health.mil/News/Articles/2019/03/15/Defense-and-Veterans-Brain-Injury-Center-releases-new-concussion-screening-tool. Published March 15, 2019. Accessed August 18, 2020.
16. Schwab K, Terrio HP, Brenner LA, et al. Epidemiology and prognosis of mild traumatic brain injury in returning soldiers: a cohort study. Neurology. 2017;88(16):1571-1579. doi:10.1212/WNL.0000000000003839
17. Mac Donald CL, Johnson AM, Wierzechowski L, et al. Outcome trends after US military concussive traumatic brain injury. J Neurotrauma. 2017;34(14):2206-2219. doi:10.1089/neu.2016.4434
18. Andelic N, Howe EI, Hellstrøm T, et al. Disability and quality of life 20 years after traumatic brain injury. Brain Behav. 2018;8(7):e01018. doi:10.1002/brb3.1018
1. Centers for Disease Control and Prevention. Surveillance report of traumatic brain injury-related emergency department visits, hospitalizations, and deaths. https://www.cdc.gov/traumaticbraininjury/pdf/TBI-Surveillance-Report-FINAL_508.pdf. Published 2014. Accessed August 18, 2020.
2. Stocchetti N, Zanier ER. Chronic impact of traumatic brain injury on outcome and quality of life: a narrative review. Crit Care. 2016;20(1):148. Published 2016 Jun 21. doi:10.1186/s13054-016-1318-1
3. Kumar RG, Ornstein KA, Bollens-Lund E, et al. Lifetime history of traumatic brain injury is associated with increased loneliness in adults: A US nationally representative study. Int J Geriatr Psychiatry. 2020;35(5):553-563. doi:10.1002/gps.5271
4. Defense and Veterans Brain Injury Center. Worldwide DoD numbers for traumatic brain injury. 2020; https://dvbic.dcoe.mil/sites/default/files/tbi-numbers/DVBIC_WorldwideTotal_2000-2019.pdf. Updated March 10, 2020. Accessed August 18, 2020.
5. Kennedy JE, Lu LH, Reid MW, Leal FO, Cooper DB. Correlates of depression in U.S. military service members with a history of mild traumatic brain injury. Mil Med. 2019;184(suppl 1):148-154. doi:10.1093/milmed/usy321
6. Marshall KR, Holland SL, Meyer KS, Martin EM, Wilmore M, Grimes JB. Mild traumatic brain injury screening, diagnosis, and treatment. Mil Med. 2012;177(suppl 8):67-75. doi:10.7205/milmed-d-12-00110
7. French L, McCrea M., Baggett M. The Military Acute Concussion Evaluation. J Spec Oper Med. 2008;8(1):68-77. https://www.jsomonline.org/Publications/2008168French.pdf. Accessed August 18, 2020.
8. Kontos AP, Jorgensen-Wagers K, Trbovich AM, et al. Association of time since injury to the first clinic visit with recovery following concussion. JAMA Neurol. 2020;77(4):435-440. doi:10.1001/jamaneurol.2019.4552
9. Ponsford J, Willmott C, Rothwell A, et al. Impact of early intervention on outcome following mild head injury in adults. J Neurol Neurosurg Psychiatry. 2002;73(3):330-332. doi:10.1136/jnnp.73.3.33010.
10. Mittenberg W, Canyock EM, Condit D, Patton C. Treatment of post-concussion syndrome following mild head injury. J Clin Exp Neuropsychol. 2001;23(6):829-836. doi:10.1076/jcen.23.6.829.1022
11. McCrea M, Kelly JP, Randolph C, et al. Standardized assessment of concussion (SAC): on-site mental status evaluation of the athlete. J Head Trauma Rehabil. 1998;13(2):27-35. doi:10.1097/00001199-199804000-00005
12. US Department of Defense. Department of Defense Instruction, Number 6490.11. Policy guidance for management of mild traumatic brain injury/concussion in the deployed setting. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649011p.pdf. Updated November 26, 2019. Accessed August 18, 2020.
13. Mucha A, Collins MW, Elbin RJ, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions: preliminary findings. Am J Sports Med. 2014;42(10):2479-2486. doi:10.1177/0363546514543775
14. Defense and Veterans Brain Injury Center. Military Acute Concussion Evaluation 2 (MACE 2). https://dvbic.dcoe.mil/material/military-acute-concussion-evaluation-2-mace-2. Updated August 18, 2020. Accessed August 18, 2020.
15. US Department of Defense, Defense Health Agency. Defense and Veterans Brain Injury Center releases new concussion screening tool. https://www.health.mil/News/Articles/2019/03/15/Defense-and-Veterans-Brain-Injury-Center-releases-new-concussion-screening-tool. Published March 15, 2019. Accessed August 18, 2020.
16. Schwab K, Terrio HP, Brenner LA, et al. Epidemiology and prognosis of mild traumatic brain injury in returning soldiers: a cohort study. Neurology. 2017;88(16):1571-1579. doi:10.1212/WNL.0000000000003839
17. Mac Donald CL, Johnson AM, Wierzechowski L, et al. Outcome trends after US military concussive traumatic brain injury. J Neurotrauma. 2017;34(14):2206-2219. doi:10.1089/neu.2016.4434
18. Andelic N, Howe EI, Hellstrøm T, et al. Disability and quality of life 20 years after traumatic brain injury. Brain Behav. 2018;8(7):e01018. doi:10.1002/brb3.1018