User login
Managing psychotropic-induced hyperhidrosis
Ms. K, age 32, presents to the psychiatric clinic for a routine follow-up. Her history includes agoraphobia, attention-deficit/hyperactivity disorder, and schizoaffective disorder. Ms. K’s current medications are oral hydroxyzine 50 mg 4 times daily as needed for anxiety and paliperidone palmitate 234 mg IM monthly. Since her last follow-up, she has been switched from oral sertraline 150 mg/d to oral paroxetine 20 mg/d. Ms. K reports having constipation (which improves by taking oral docusate 100 mg twice daily) and generalized hyperhidrosis. She wants to alleviate the hyperhidrosis without changing her paroxetine because that medication improved her symptoms.
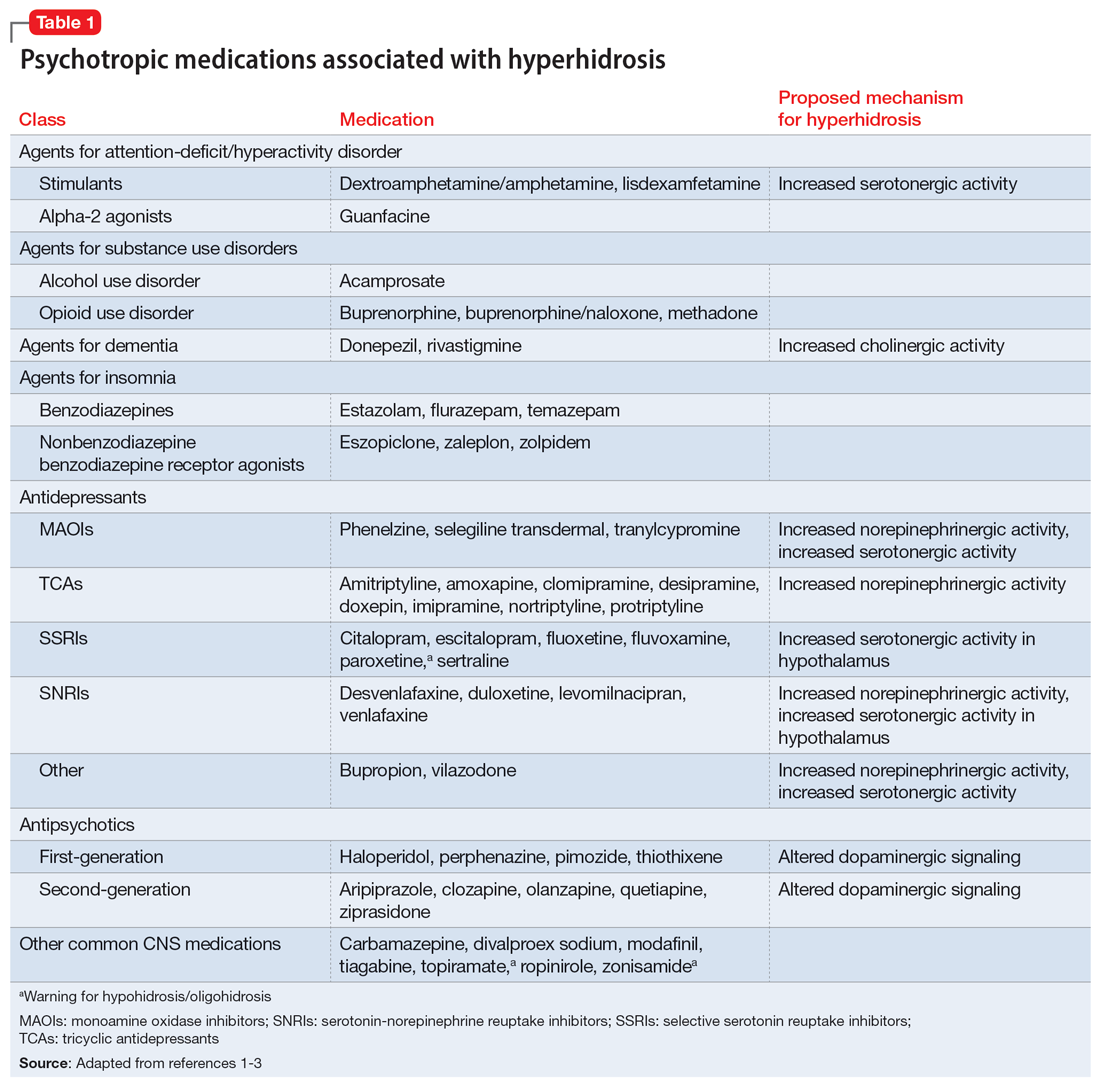
Hyperhidrosis—excessive sweating not needed to maintain a normal body temperature—is an uncommon and uncomfortable adverse effect of many medications, including psychotropics.1 This long-term adverse effect typically is not dose-related and does not remit with continued therapy.2Table 11-3 lists psychotropic medications associated with hyperhidrosis as well as postulated mechanisms.
The incidence of medication-induced hyperhidrosis is unknown,but for psychotropic medications it is estimated to be 5% to 20%.3 Patients may not report hyperhidrosis due to embarrassment; in clinical trials, reporting measures may be inconsistent and, in some cases, misleading. For example, it is possible hyperhidrosis that appears to be associated with buprenorphine is actually a symptom of the withdrawal syndrome rather than a direct effect of the medication. Also, some medications, including certain psychotropics (eg, paroxetine4 and topiramate3) may cause either hyperhidrosis or hypohidrosis (decreased sweating). Few medications carry labeled warnings for hypohidrosis; the condition generally is not of clinical concern unless patients experience heat intolerance or hyperthermia.3
Psychotropic-induced hyperhidrosis is likely an idiopathic effect. There are few known predisposing factors, but some medications carry a greater risk than others. In a meta-analysis, Beyer et al2 found certain selective serotonin reuptake inhibitors (SSRIs), such as sertraline and paroxetine, had a higher risk of causing hyperhidrosis. Fluvoxamine, bupropion, and vortioxetine had the lowest risk. The class risk for SSRIs was comparable to that of serotonin-norepinephrine reuptake inhibitors (SNRIs), which all carried a comparable risk. In this analysis, neither indication nor dose were reliable indicators of risk of causing hyperhidrosis. However, the study found that for both SSRIs and SNRIs, increased affinity for the dopamine transporter was correlated with an increased risk of hyperhidrosis.2
Treatment
Treatment of hyperhidrosis depends on its cause and presentation.5 Hyperhidrosis may be categorized as primary (idiopathic) or secondary (also termed diaphoresis), and either focal or generalized.6 Many treatment recommendations focus on primary or focal hyperhidrosis and prioritize topical therapies.5 Because medication-induced hyperhidrosis most commonly presents as generalized3 and thus affects a large body surface area, the use of topical therapies is precluded. Topical therapy for psychotropic-induced hyperhidrosis should be pursued only if the patient’s sweating is localized.
Treating medication-induced hyperhidrosis becomes more complicated if it is not possible to alter the inciting medication (ie, because the medication is effective or the patient is resistant to change). In such scenarios, discontinuing the medication and initiating an alternative therapy may not be effective or feasible.2 For generalized presentations of medication-induced hyperhidrosis, if the inciting medication cannot be altered, initiating an oral systemic therapy is the preferred treatment.3,5
Oral anticholinergic medications (eg, benztropine, glycopyrrolate, and oxybutynin),4-6 act directly on muscarinic receptors within the eccrine sweat glands to decrease or stop sweating. They are considered first-line for generalized hyperhidrosis but may be inappropriate for psychotropic-induced hyperhidrosis because many psychotropics (eg, tricyclic antidepressants, paroxetine, olanzapine, quetiapine, and clozapine) have anticholinergic properties. Adding an anticholinergic medication to these patients’ regimens may increase the adverse effect burden and worsen cognitive deficits. Additionally, approximately one-third of patients discontinue anticholinergic medications due to tolerability issues (eg, dry mouth).
Continue to: However, anticholinergic medications...
However, anticholinergic medications may still have a role in treating psychotropic-induced hyperhidrosis. Benztropine3,7,8 and cyproheptadine2,3,9 may be effective options, though their role in treating psychotropic-induced hyperhidrosis should be limited and reserved for patients who have another compelling indication for these medications (eg, extrapyramidal symptoms) or when other treatment options are ineffective or intolerable.
Avoiding anticholinergic medications can also be justified based on the proposed mechanism of psychotropic-induced hyperhidrosis as an extension of the medication’s toxic effects. Conceptualizing psychotropic-induced hyperhidrosis as similar to the diaphoresis and hyperthermia observed in neuroleptic malignant syndrome and serotonin syndrome offers a clearer target for treatment. Though the specifics of the mechanisms remain unknown,2 many medications that cause hyperhidrosis do so by increasing sweat gland secretions, either directly by increasing cholinergic activity or indirectly via increased sympathetic transmission.
Considering this pathophysiology, another target for psychotropic-induced hyperhidrosis may be altered and/or excessive catecholamine activity. The use of medications such as clonidine,3-6 propranolol,4-6 or terazosin2,3,10 should be considered given their beneficial effects on the activation of the sympathetic nervous system, although clonidine also possesses anticholinergic activity. The calcium channel blocker diltiazem can improve hyperhidrosis symptoms by interfering with the calcium signaling necessary for normal sweat gland function.4,5 Comorbid cardiovascular diseases and tachycardia, an adverse effect of many psychotropic medications, may also be managed with these treatment options. Some research suggests using benzodiazepines to treat psychotropic-induced hyperhidrosis.4-6 As is the case for anticholinergic medications, the use of benzodiazepines would require another compelling indication for long-term use.
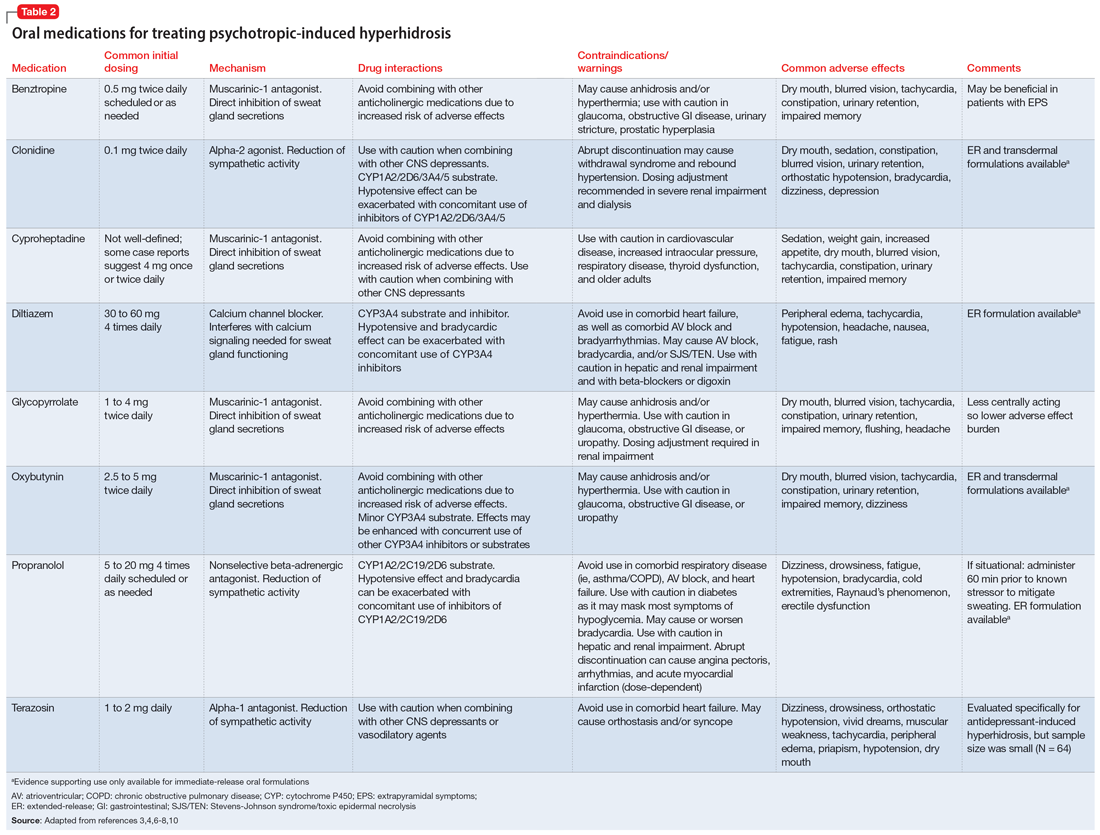
Table 23,4,6-8,10 provides recommended dosing and caveats for the use of these medications and other potentially appropriate medications.
Research of investigational treatments for generalized hyperhidrosis is ongoing. It is possible some of these medications may have a future role in the treatment of psychotropic-induced hyperhidrosis, with improved efficacy and better tolerability.
Continue to: CASE CONTINUED
CASE CONTINUED
Because Ms. K’s medication-induced hyperhidrosis is generalized and therefore ineligible for topical therapies, and because the inciting medication (paroxetine) cannot be switched to an alternative, the treatment team considers adding an oral medication. Treatment with an anticholinergic medication, such as benztropine, is not preferred due to the anticholinergic activity associated with paroxetine and Ms. K’s history of constipation. After discussing other oral treatment options with Ms. K, the team ultimately decides to initiate propranolol at a low dose (5 mg twice daily) to minimize the chances of an interaction with paroxetine, and titrate based on efficacy and tolerability.
Related Resources
- International Hyperhidrosis Society. Hyperhidrosis treatment overview. www.sweathelp.org/hyperhidrosis-treatments/treatment-overview.html
Drug Brand Names
Acamprosate • Campral
Aripiprazole • Abilify
Buprenorphine • Sublocade
Buprenorphine/naloxone • Zubsolv
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Citalopram • Celexa
Clomipramine • Anafranil
Clonidine • Catapres
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextroamphetamine/amphetamine • Adderall
Diltiazem • Cardizem
Divalproex • Depakote
Donepezil • Aricept
Doxepin • Silenor
Duloxetine • Cymbalta
Escitalopram • Lexapro
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Guanfacine • Intuniv
Glycopyrrolate • Cuvposa
Hydroxyzine • Vistaril
Imipramine • Tofranil
Levomilnacipran • Fetzima
Lisdexamfetamine • Vyvanse
Methadone • Dolophine, Methadose
Modafinil • Provigil
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paliperidone palmitate • Invega Sustenna
Paroxetine • Paxil
Phenelzine • Nardil
Pimozide • Orap
Protriptyline • Vivactil
Quetiapine • Seroquel
Rivastigmine • Exelon
Selegiline transdermal • Emsam
Sertraline • Zoloft
Temazepam • Restoril
Thiothixene • Navane
Tiagabine • Gabitril
Topiramate • Topamax
Tranylcypromine • Parnate
Vilazodone • Viibryd
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
Zonisamide • Zonegran
1. International Hyperhidrosis Society. Drugs/medications known to cause hyperhidrosis. Sweathelp.org. 2022. Accessed September 6, 2022. https://www.sweathelp.org/pdf/drugs_2009.pdf
2. Beyer C, Cappetta K, Johnson JA, et al. Meta-analysis: risk of hyperhidrosis with second-generation antidepressants. Depress Anxiety. 2017;34(12):1134-1146. doi:10.1002/da.22680
3. Cheshire WP, Fealey RD. Drug-induced hyperhidrosis and hypohidrosis: incidence, prevention and management. Drug Saf. 2008;31(2):109-126. doi:10.2165/00002018-200831020-00002
4. del Boz J. Systemic treatment of hyperhidrosis. Actas Dermosifiliogr. 2015;106(4):271-277. doi:10.1016/j.ad.2014.11.012
5. Nawrocki S, Cha J. The etiology, diagnosis, and management of hyperhidrosis: a comprehensive review: therapeutic options. J Am Acad Dermatol. 2019;81(3):669-680. doi:10.1016/j.jaad2018.11.066
6. Glaser DA. Oral medications. Dermatol Clin. 2014;32(4):527-532. doi:10.1016/j.det.2014.06.002
7. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177. doi:10.4088/jcp.v58n0407e
8. Kolli V, Ramaswamy S. Improvement of antidepressant-induced sweating with as-required benztropine. Innov Clin Neurosci. 2013;10(11-12):10-11.
9. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):875. doi:10.1176/APPI.AJP.159.5.874-A
10. Ghaleiha A, Shahidi KM, Afzali S, et al. Effect of terazosin on sweating in patients with major depressive disorder receiving sertraline: a randomized controlled trial. Int J Psychiatry Clin Pract. 2013;17(1):44-47. doi:10.3109/13651501.2012.687449
Ms. K, age 32, presents to the psychiatric clinic for a routine follow-up. Her history includes agoraphobia, attention-deficit/hyperactivity disorder, and schizoaffective disorder. Ms. K’s current medications are oral hydroxyzine 50 mg 4 times daily as needed for anxiety and paliperidone palmitate 234 mg IM monthly. Since her last follow-up, she has been switched from oral sertraline 150 mg/d to oral paroxetine 20 mg/d. Ms. K reports having constipation (which improves by taking oral docusate 100 mg twice daily) and generalized hyperhidrosis. She wants to alleviate the hyperhidrosis without changing her paroxetine because that medication improved her symptoms.
Hyperhidrosis—excessive sweating not needed to maintain a normal body temperature—is an uncommon and uncomfortable adverse effect of many medications, including psychotropics.1 This long-term adverse effect typically is not dose-related and does not remit with continued therapy.2Table 11-3 lists psychotropic medications associated with hyperhidrosis as well as postulated mechanisms.

The incidence of medication-induced hyperhidrosis is unknown,but for psychotropic medications it is estimated to be 5% to 20%.3 Patients may not report hyperhidrosis due to embarrassment; in clinical trials, reporting measures may be inconsistent and, in some cases, misleading. For example, it is possible hyperhidrosis that appears to be associated with buprenorphine is actually a symptom of the withdrawal syndrome rather than a direct effect of the medication. Also, some medications, including certain psychotropics (eg, paroxetine4 and topiramate3) may cause either hyperhidrosis or hypohidrosis (decreased sweating). Few medications carry labeled warnings for hypohidrosis; the condition generally is not of clinical concern unless patients experience heat intolerance or hyperthermia.3
Psychotropic-induced hyperhidrosis is likely an idiopathic effect. There are few known predisposing factors, but some medications carry a greater risk than others. In a meta-analysis, Beyer et al2 found certain selective serotonin reuptake inhibitors (SSRIs), such as sertraline and paroxetine, had a higher risk of causing hyperhidrosis. Fluvoxamine, bupropion, and vortioxetine had the lowest risk. The class risk for SSRIs was comparable to that of serotonin-norepinephrine reuptake inhibitors (SNRIs), which all carried a comparable risk. In this analysis, neither indication nor dose were reliable indicators of risk of causing hyperhidrosis. However, the study found that for both SSRIs and SNRIs, increased affinity for the dopamine transporter was correlated with an increased risk of hyperhidrosis.2
Treatment
Treatment of hyperhidrosis depends on its cause and presentation.5 Hyperhidrosis may be categorized as primary (idiopathic) or secondary (also termed diaphoresis), and either focal or generalized.6 Many treatment recommendations focus on primary or focal hyperhidrosis and prioritize topical therapies.5 Because medication-induced hyperhidrosis most commonly presents as generalized3 and thus affects a large body surface area, the use of topical therapies is precluded. Topical therapy for psychotropic-induced hyperhidrosis should be pursued only if the patient’s sweating is localized.
Treating medication-induced hyperhidrosis becomes more complicated if it is not possible to alter the inciting medication (ie, because the medication is effective or the patient is resistant to change). In such scenarios, discontinuing the medication and initiating an alternative therapy may not be effective or feasible.2 For generalized presentations of medication-induced hyperhidrosis, if the inciting medication cannot be altered, initiating an oral systemic therapy is the preferred treatment.3,5
Oral anticholinergic medications (eg, benztropine, glycopyrrolate, and oxybutynin),4-6 act directly on muscarinic receptors within the eccrine sweat glands to decrease or stop sweating. They are considered first-line for generalized hyperhidrosis but may be inappropriate for psychotropic-induced hyperhidrosis because many psychotropics (eg, tricyclic antidepressants, paroxetine, olanzapine, quetiapine, and clozapine) have anticholinergic properties. Adding an anticholinergic medication to these patients’ regimens may increase the adverse effect burden and worsen cognitive deficits. Additionally, approximately one-third of patients discontinue anticholinergic medications due to tolerability issues (eg, dry mouth).
Continue to: However, anticholinergic medications...
However, anticholinergic medications may still have a role in treating psychotropic-induced hyperhidrosis. Benztropine3,7,8 and cyproheptadine2,3,9 may be effective options, though their role in treating psychotropic-induced hyperhidrosis should be limited and reserved for patients who have another compelling indication for these medications (eg, extrapyramidal symptoms) or when other treatment options are ineffective or intolerable.
Avoiding anticholinergic medications can also be justified based on the proposed mechanism of psychotropic-induced hyperhidrosis as an extension of the medication’s toxic effects. Conceptualizing psychotropic-induced hyperhidrosis as similar to the diaphoresis and hyperthermia observed in neuroleptic malignant syndrome and serotonin syndrome offers a clearer target for treatment. Though the specifics of the mechanisms remain unknown,2 many medications that cause hyperhidrosis do so by increasing sweat gland secretions, either directly by increasing cholinergic activity or indirectly via increased sympathetic transmission.
Considering this pathophysiology, another target for psychotropic-induced hyperhidrosis may be altered and/or excessive catecholamine activity. The use of medications such as clonidine,3-6 propranolol,4-6 or terazosin2,3,10 should be considered given their beneficial effects on the activation of the sympathetic nervous system, although clonidine also possesses anticholinergic activity. The calcium channel blocker diltiazem can improve hyperhidrosis symptoms by interfering with the calcium signaling necessary for normal sweat gland function.4,5 Comorbid cardiovascular diseases and tachycardia, an adverse effect of many psychotropic medications, may also be managed with these treatment options. Some research suggests using benzodiazepines to treat psychotropic-induced hyperhidrosis.4-6 As is the case for anticholinergic medications, the use of benzodiazepines would require another compelling indication for long-term use.
Table 23,4,6-8,10 provides recommended dosing and caveats for the use of these medications and other potentially appropriate medications.

Research of investigational treatments for generalized hyperhidrosis is ongoing. It is possible some of these medications may have a future role in the treatment of psychotropic-induced hyperhidrosis, with improved efficacy and better tolerability.
Continue to: CASE CONTINUED
CASE CONTINUED
Because Ms. K’s medication-induced hyperhidrosis is generalized and therefore ineligible for topical therapies, and because the inciting medication (paroxetine) cannot be switched to an alternative, the treatment team considers adding an oral medication. Treatment with an anticholinergic medication, such as benztropine, is not preferred due to the anticholinergic activity associated with paroxetine and Ms. K’s history of constipation. After discussing other oral treatment options with Ms. K, the team ultimately decides to initiate propranolol at a low dose (5 mg twice daily) to minimize the chances of an interaction with paroxetine, and titrate based on efficacy and tolerability.
Related Resources
- International Hyperhidrosis Society. Hyperhidrosis treatment overview. www.sweathelp.org/hyperhidrosis-treatments/treatment-overview.html
Drug Brand Names
Acamprosate • Campral
Aripiprazole • Abilify
Buprenorphine • Sublocade
Buprenorphine/naloxone • Zubsolv
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Citalopram • Celexa
Clomipramine • Anafranil
Clonidine • Catapres
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextroamphetamine/amphetamine • Adderall
Diltiazem • Cardizem
Divalproex • Depakote
Donepezil • Aricept
Doxepin • Silenor
Duloxetine • Cymbalta
Escitalopram • Lexapro
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Guanfacine • Intuniv
Glycopyrrolate • Cuvposa
Hydroxyzine • Vistaril
Imipramine • Tofranil
Levomilnacipran • Fetzima
Lisdexamfetamine • Vyvanse
Methadone • Dolophine, Methadose
Modafinil • Provigil
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paliperidone palmitate • Invega Sustenna
Paroxetine • Paxil
Phenelzine • Nardil
Pimozide • Orap
Protriptyline • Vivactil
Quetiapine • Seroquel
Rivastigmine • Exelon
Selegiline transdermal • Emsam
Sertraline • Zoloft
Temazepam • Restoril
Thiothixene • Navane
Tiagabine • Gabitril
Topiramate • Topamax
Tranylcypromine • Parnate
Vilazodone • Viibryd
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
Zonisamide • Zonegran
Ms. K, age 32, presents to the psychiatric clinic for a routine follow-up. Her history includes agoraphobia, attention-deficit/hyperactivity disorder, and schizoaffective disorder. Ms. K’s current medications are oral hydroxyzine 50 mg 4 times daily as needed for anxiety and paliperidone palmitate 234 mg IM monthly. Since her last follow-up, she has been switched from oral sertraline 150 mg/d to oral paroxetine 20 mg/d. Ms. K reports having constipation (which improves by taking oral docusate 100 mg twice daily) and generalized hyperhidrosis. She wants to alleviate the hyperhidrosis without changing her paroxetine because that medication improved her symptoms.
Hyperhidrosis—excessive sweating not needed to maintain a normal body temperature—is an uncommon and uncomfortable adverse effect of many medications, including psychotropics.1 This long-term adverse effect typically is not dose-related and does not remit with continued therapy.2Table 11-3 lists psychotropic medications associated with hyperhidrosis as well as postulated mechanisms.

The incidence of medication-induced hyperhidrosis is unknown,but for psychotropic medications it is estimated to be 5% to 20%.3 Patients may not report hyperhidrosis due to embarrassment; in clinical trials, reporting measures may be inconsistent and, in some cases, misleading. For example, it is possible hyperhidrosis that appears to be associated with buprenorphine is actually a symptom of the withdrawal syndrome rather than a direct effect of the medication. Also, some medications, including certain psychotropics (eg, paroxetine4 and topiramate3) may cause either hyperhidrosis or hypohidrosis (decreased sweating). Few medications carry labeled warnings for hypohidrosis; the condition generally is not of clinical concern unless patients experience heat intolerance or hyperthermia.3
Psychotropic-induced hyperhidrosis is likely an idiopathic effect. There are few known predisposing factors, but some medications carry a greater risk than others. In a meta-analysis, Beyer et al2 found certain selective serotonin reuptake inhibitors (SSRIs), such as sertraline and paroxetine, had a higher risk of causing hyperhidrosis. Fluvoxamine, bupropion, and vortioxetine had the lowest risk. The class risk for SSRIs was comparable to that of serotonin-norepinephrine reuptake inhibitors (SNRIs), which all carried a comparable risk. In this analysis, neither indication nor dose were reliable indicators of risk of causing hyperhidrosis. However, the study found that for both SSRIs and SNRIs, increased affinity for the dopamine transporter was correlated with an increased risk of hyperhidrosis.2
Treatment
Treatment of hyperhidrosis depends on its cause and presentation.5 Hyperhidrosis may be categorized as primary (idiopathic) or secondary (also termed diaphoresis), and either focal or generalized.6 Many treatment recommendations focus on primary or focal hyperhidrosis and prioritize topical therapies.5 Because medication-induced hyperhidrosis most commonly presents as generalized3 and thus affects a large body surface area, the use of topical therapies is precluded. Topical therapy for psychotropic-induced hyperhidrosis should be pursued only if the patient’s sweating is localized.
Treating medication-induced hyperhidrosis becomes more complicated if it is not possible to alter the inciting medication (ie, because the medication is effective or the patient is resistant to change). In such scenarios, discontinuing the medication and initiating an alternative therapy may not be effective or feasible.2 For generalized presentations of medication-induced hyperhidrosis, if the inciting medication cannot be altered, initiating an oral systemic therapy is the preferred treatment.3,5
Oral anticholinergic medications (eg, benztropine, glycopyrrolate, and oxybutynin),4-6 act directly on muscarinic receptors within the eccrine sweat glands to decrease or stop sweating. They are considered first-line for generalized hyperhidrosis but may be inappropriate for psychotropic-induced hyperhidrosis because many psychotropics (eg, tricyclic antidepressants, paroxetine, olanzapine, quetiapine, and clozapine) have anticholinergic properties. Adding an anticholinergic medication to these patients’ regimens may increase the adverse effect burden and worsen cognitive deficits. Additionally, approximately one-third of patients discontinue anticholinergic medications due to tolerability issues (eg, dry mouth).
Continue to: However, anticholinergic medications...
However, anticholinergic medications may still have a role in treating psychotropic-induced hyperhidrosis. Benztropine3,7,8 and cyproheptadine2,3,9 may be effective options, though their role in treating psychotropic-induced hyperhidrosis should be limited and reserved for patients who have another compelling indication for these medications (eg, extrapyramidal symptoms) or when other treatment options are ineffective or intolerable.
Avoiding anticholinergic medications can also be justified based on the proposed mechanism of psychotropic-induced hyperhidrosis as an extension of the medication’s toxic effects. Conceptualizing psychotropic-induced hyperhidrosis as similar to the diaphoresis and hyperthermia observed in neuroleptic malignant syndrome and serotonin syndrome offers a clearer target for treatment. Though the specifics of the mechanisms remain unknown,2 many medications that cause hyperhidrosis do so by increasing sweat gland secretions, either directly by increasing cholinergic activity or indirectly via increased sympathetic transmission.
Considering this pathophysiology, another target for psychotropic-induced hyperhidrosis may be altered and/or excessive catecholamine activity. The use of medications such as clonidine,3-6 propranolol,4-6 or terazosin2,3,10 should be considered given their beneficial effects on the activation of the sympathetic nervous system, although clonidine also possesses anticholinergic activity. The calcium channel blocker diltiazem can improve hyperhidrosis symptoms by interfering with the calcium signaling necessary for normal sweat gland function.4,5 Comorbid cardiovascular diseases and tachycardia, an adverse effect of many psychotropic medications, may also be managed with these treatment options. Some research suggests using benzodiazepines to treat psychotropic-induced hyperhidrosis.4-6 As is the case for anticholinergic medications, the use of benzodiazepines would require another compelling indication for long-term use.
Table 23,4,6-8,10 provides recommended dosing and caveats for the use of these medications and other potentially appropriate medications.

Research of investigational treatments for generalized hyperhidrosis is ongoing. It is possible some of these medications may have a future role in the treatment of psychotropic-induced hyperhidrosis, with improved efficacy and better tolerability.
Continue to: CASE CONTINUED
CASE CONTINUED
Because Ms. K’s medication-induced hyperhidrosis is generalized and therefore ineligible for topical therapies, and because the inciting medication (paroxetine) cannot be switched to an alternative, the treatment team considers adding an oral medication. Treatment with an anticholinergic medication, such as benztropine, is not preferred due to the anticholinergic activity associated with paroxetine and Ms. K’s history of constipation. After discussing other oral treatment options with Ms. K, the team ultimately decides to initiate propranolol at a low dose (5 mg twice daily) to minimize the chances of an interaction with paroxetine, and titrate based on efficacy and tolerability.
Related Resources
- International Hyperhidrosis Society. Hyperhidrosis treatment overview. www.sweathelp.org/hyperhidrosis-treatments/treatment-overview.html
Drug Brand Names
Acamprosate • Campral
Aripiprazole • Abilify
Buprenorphine • Sublocade
Buprenorphine/naloxone • Zubsolv
Bupropion • Wellbutrin
Carbamazepine • Tegretol
Citalopram • Celexa
Clomipramine • Anafranil
Clonidine • Catapres
Clozapine • Clozaril
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Dextroamphetamine/amphetamine • Adderall
Diltiazem • Cardizem
Divalproex • Depakote
Donepezil • Aricept
Doxepin • Silenor
Duloxetine • Cymbalta
Escitalopram • Lexapro
Eszopiclone • Lunesta
Fluoxetine • Prozac
Fluvoxamine • Luvox
Guanfacine • Intuniv
Glycopyrrolate • Cuvposa
Hydroxyzine • Vistaril
Imipramine • Tofranil
Levomilnacipran • Fetzima
Lisdexamfetamine • Vyvanse
Methadone • Dolophine, Methadose
Modafinil • Provigil
Nortriptyline • Pamelor
Olanzapine • Zyprexa
Paliperidone palmitate • Invega Sustenna
Paroxetine • Paxil
Phenelzine • Nardil
Pimozide • Orap
Protriptyline • Vivactil
Quetiapine • Seroquel
Rivastigmine • Exelon
Selegiline transdermal • Emsam
Sertraline • Zoloft
Temazepam • Restoril
Thiothixene • Navane
Tiagabine • Gabitril
Topiramate • Topamax
Tranylcypromine • Parnate
Vilazodone • Viibryd
Vortioxetine • Trintellix
Zaleplon • Sonata
Ziprasidone • Geodon
Zolpidem • Ambien
Zonisamide • Zonegran
1. International Hyperhidrosis Society. Drugs/medications known to cause hyperhidrosis. Sweathelp.org. 2022. Accessed September 6, 2022. https://www.sweathelp.org/pdf/drugs_2009.pdf
2. Beyer C, Cappetta K, Johnson JA, et al. Meta-analysis: risk of hyperhidrosis with second-generation antidepressants. Depress Anxiety. 2017;34(12):1134-1146. doi:10.1002/da.22680
3. Cheshire WP, Fealey RD. Drug-induced hyperhidrosis and hypohidrosis: incidence, prevention and management. Drug Saf. 2008;31(2):109-126. doi:10.2165/00002018-200831020-00002
4. del Boz J. Systemic treatment of hyperhidrosis. Actas Dermosifiliogr. 2015;106(4):271-277. doi:10.1016/j.ad.2014.11.012
5. Nawrocki S, Cha J. The etiology, diagnosis, and management of hyperhidrosis: a comprehensive review: therapeutic options. J Am Acad Dermatol. 2019;81(3):669-680. doi:10.1016/j.jaad2018.11.066
6. Glaser DA. Oral medications. Dermatol Clin. 2014;32(4):527-532. doi:10.1016/j.det.2014.06.002
7. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177. doi:10.4088/jcp.v58n0407e
8. Kolli V, Ramaswamy S. Improvement of antidepressant-induced sweating with as-required benztropine. Innov Clin Neurosci. 2013;10(11-12):10-11.
9. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):875. doi:10.1176/APPI.AJP.159.5.874-A
10. Ghaleiha A, Shahidi KM, Afzali S, et al. Effect of terazosin on sweating in patients with major depressive disorder receiving sertraline: a randomized controlled trial. Int J Psychiatry Clin Pract. 2013;17(1):44-47. doi:10.3109/13651501.2012.687449
1. International Hyperhidrosis Society. Drugs/medications known to cause hyperhidrosis. Sweathelp.org. 2022. Accessed September 6, 2022. https://www.sweathelp.org/pdf/drugs_2009.pdf
2. Beyer C, Cappetta K, Johnson JA, et al. Meta-analysis: risk of hyperhidrosis with second-generation antidepressants. Depress Anxiety. 2017;34(12):1134-1146. doi:10.1002/da.22680
3. Cheshire WP, Fealey RD. Drug-induced hyperhidrosis and hypohidrosis: incidence, prevention and management. Drug Saf. 2008;31(2):109-126. doi:10.2165/00002018-200831020-00002
4. del Boz J. Systemic treatment of hyperhidrosis. Actas Dermosifiliogr. 2015;106(4):271-277. doi:10.1016/j.ad.2014.11.012
5. Nawrocki S, Cha J. The etiology, diagnosis, and management of hyperhidrosis: a comprehensive review: therapeutic options. J Am Acad Dermatol. 2019;81(3):669-680. doi:10.1016/j.jaad2018.11.066
6. Glaser DA. Oral medications. Dermatol Clin. 2014;32(4):527-532. doi:10.1016/j.det.2014.06.002
7. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177. doi:10.4088/jcp.v58n0407e
8. Kolli V, Ramaswamy S. Improvement of antidepressant-induced sweating with as-required benztropine. Innov Clin Neurosci. 2013;10(11-12):10-11.
9. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):875. doi:10.1176/APPI.AJP.159.5.874-A
10. Ghaleiha A, Shahidi KM, Afzali S, et al. Effect of terazosin on sweating in patients with major depressive disorder receiving sertraline: a randomized controlled trial. Int J Psychiatry Clin Pract. 2013;17(1):44-47. doi:10.3109/13651501.2012.687449
Psychedelic therapy tied to reduced depression, anxiety
TOPLINE:
Perhaps most surprising to investigators, however, was that treatment was also associated with improved cognitive scores in the veterans, many of whom had traumatic brain injuries.
METHODOLOGY:
- Investigators reviewed clinical charts of 86 SOFVs who received psychedelic-assisted treatment at a therapeutic program in Mexico, 86% of whom sustained head injuries during deployment.
- On the first day of the study, participants received a single oral dose (10 mg/kg) of ibogaine hydrochloride in a group setting with two to five other attendees and spent the next day reflecting on their experience with program staff.
- On the third day, participants inhaled 5-MeO-DMT in three incremental doses for a total of 50 mg and were then invited to reflect on their experience both individually and with the group of peers who shared the experience.
- Follow-up surveys at 1, 3, and 6 months posttreatment between September 2019 to March 2021 measured symptoms of posttraumatic stress disorder, cognitive functioning, generalized anxiety disorder, depression, and quality of life.
TAKEAWAY:
- There were significant improvements in self-reported PTSD symptoms, depression, anxiety, insomnia severity, anger, and a large improvement in self-reported satisfaction with life (P < .001 for all).
- Participants reported significant increases in psychological flexibility (P < .001), cognitive functioning (P < .001), and postconcussive symptoms (P < .001).
- Treatment was also associated with a significant reduction in suicidal ideation from pretreatment to 1-month follow-up (P < .01).
IN PRACTICE:
“If consistently replicated, this could have major implications for the landscape of mental health care if people are able to experience significant and sustained healing with 3 days of intensive treatment, relative to our traditionally available interventions that require 8-12 weeks of weekly therapy (for example, gold standard talk therapies such as [prolonged exposure] or [cognitive processing therapy]), or daily use of a pharmacotherapy such as [a selective serotonin reuptake inhibitor] for months to years,” study authors write.
SOURCE:
Alan Kooi Davis, PhD, of the Center for Psychedelic Drug Research and Education at Ohio State University, led the study, which was published online in the American Journal of Drug and Alcohol Abuse.
LIMITATIONS:
Study assessments are based solely on self-report measures. Future research should implement carefully designed batteries that include both self-report and gold-standard clinician-administered measures to better capture symptom improvement and other information. The sample also lacked diversity with regard to race, religion, and socioeconomic status.
DISCLOSURES:
The study was funded by Veterans Exploring Treatment Solutions. Dr. Davis is a board member at Source Resource Foundation and a lead trainer at Fluence. Full disclosures are included in the original article.
A version of this article first appeared on Medscape.com.
TOPLINE:
Perhaps most surprising to investigators, however, was that treatment was also associated with improved cognitive scores in the veterans, many of whom had traumatic brain injuries.
METHODOLOGY:
- Investigators reviewed clinical charts of 86 SOFVs who received psychedelic-assisted treatment at a therapeutic program in Mexico, 86% of whom sustained head injuries during deployment.
- On the first day of the study, participants received a single oral dose (10 mg/kg) of ibogaine hydrochloride in a group setting with two to five other attendees and spent the next day reflecting on their experience with program staff.
- On the third day, participants inhaled 5-MeO-DMT in three incremental doses for a total of 50 mg and were then invited to reflect on their experience both individually and with the group of peers who shared the experience.
- Follow-up surveys at 1, 3, and 6 months posttreatment between September 2019 to March 2021 measured symptoms of posttraumatic stress disorder, cognitive functioning, generalized anxiety disorder, depression, and quality of life.
TAKEAWAY:
- There were significant improvements in self-reported PTSD symptoms, depression, anxiety, insomnia severity, anger, and a large improvement in self-reported satisfaction with life (P < .001 for all).
- Participants reported significant increases in psychological flexibility (P < .001), cognitive functioning (P < .001), and postconcussive symptoms (P < .001).
- Treatment was also associated with a significant reduction in suicidal ideation from pretreatment to 1-month follow-up (P < .01).
IN PRACTICE:
“If consistently replicated, this could have major implications for the landscape of mental health care if people are able to experience significant and sustained healing with 3 days of intensive treatment, relative to our traditionally available interventions that require 8-12 weeks of weekly therapy (for example, gold standard talk therapies such as [prolonged exposure] or [cognitive processing therapy]), or daily use of a pharmacotherapy such as [a selective serotonin reuptake inhibitor] for months to years,” study authors write.
SOURCE:
Alan Kooi Davis, PhD, of the Center for Psychedelic Drug Research and Education at Ohio State University, led the study, which was published online in the American Journal of Drug and Alcohol Abuse.
LIMITATIONS:
Study assessments are based solely on self-report measures. Future research should implement carefully designed batteries that include both self-report and gold-standard clinician-administered measures to better capture symptom improvement and other information. The sample also lacked diversity with regard to race, religion, and socioeconomic status.
DISCLOSURES:
The study was funded by Veterans Exploring Treatment Solutions. Dr. Davis is a board member at Source Resource Foundation and a lead trainer at Fluence. Full disclosures are included in the original article.
A version of this article first appeared on Medscape.com.
TOPLINE:
Perhaps most surprising to investigators, however, was that treatment was also associated with improved cognitive scores in the veterans, many of whom had traumatic brain injuries.
METHODOLOGY:
- Investigators reviewed clinical charts of 86 SOFVs who received psychedelic-assisted treatment at a therapeutic program in Mexico, 86% of whom sustained head injuries during deployment.
- On the first day of the study, participants received a single oral dose (10 mg/kg) of ibogaine hydrochloride in a group setting with two to five other attendees and spent the next day reflecting on their experience with program staff.
- On the third day, participants inhaled 5-MeO-DMT in three incremental doses for a total of 50 mg and were then invited to reflect on their experience both individually and with the group of peers who shared the experience.
- Follow-up surveys at 1, 3, and 6 months posttreatment between September 2019 to March 2021 measured symptoms of posttraumatic stress disorder, cognitive functioning, generalized anxiety disorder, depression, and quality of life.
TAKEAWAY:
- There were significant improvements in self-reported PTSD symptoms, depression, anxiety, insomnia severity, anger, and a large improvement in self-reported satisfaction with life (P < .001 for all).
- Participants reported significant increases in psychological flexibility (P < .001), cognitive functioning (P < .001), and postconcussive symptoms (P < .001).
- Treatment was also associated with a significant reduction in suicidal ideation from pretreatment to 1-month follow-up (P < .01).
IN PRACTICE:
“If consistently replicated, this could have major implications for the landscape of mental health care if people are able to experience significant and sustained healing with 3 days of intensive treatment, relative to our traditionally available interventions that require 8-12 weeks of weekly therapy (for example, gold standard talk therapies such as [prolonged exposure] or [cognitive processing therapy]), or daily use of a pharmacotherapy such as [a selective serotonin reuptake inhibitor] for months to years,” study authors write.
SOURCE:
Alan Kooi Davis, PhD, of the Center for Psychedelic Drug Research and Education at Ohio State University, led the study, which was published online in the American Journal of Drug and Alcohol Abuse.
LIMITATIONS:
Study assessments are based solely on self-report measures. Future research should implement carefully designed batteries that include both self-report and gold-standard clinician-administered measures to better capture symptom improvement and other information. The sample also lacked diversity with regard to race, religion, and socioeconomic status.
DISCLOSURES:
The study was funded by Veterans Exploring Treatment Solutions. Dr. Davis is a board member at Source Resource Foundation and a lead trainer at Fluence. Full disclosures are included in the original article.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF DRUG AND ALCOHOL ABUSE
Pandemic-era telehealth led to fewer therapy disruptions
TOPLINE:
METHODOLOGY:
- Retrospective study using electronic health records and insurance claims data from three large U.S. health systems.
- Sample included 110,089 patients with mental health conditions who attended at least two psychotherapy visits during the 9 months before and 9 months after the onset of COVID-19, defined in this study as March 14, 2020.
- Outcome was disruption in psychotherapy, defined as a gap of more than 45 days between visits.
TAKEAWAY:
- Before the pandemic, 96.9% of psychotherapy visits were in person and 35.4% were followed by a gap of more than 45 days.
- After the onset of the pandemic, more than half of visits (51.8%) were virtual, and only 17.9% were followed by a gap of more than 45 days.
- Prior to the pandemic, the median time between visits was 27 days, and after the pandemic, it dropped to 14 days, suggesting individuals were more likely to return for additional psychotherapy after the widespread shift to virtual care.
- Over the entire study period, individuals with depressive, anxiety, or bipolar disorders were more likely to maintain consistent psychotherapy visits, whereas those with schizophrenia, ADHD, autism, conduct or disruptive disorders, dementia, or personality disorders were more likely to have a disruption in their visits.
IN PRACTICE:
“These findings support continued use of virtual psychotherapy as an option for care when appropriate infrastructure is in place. In addition, these findings support the continuation of policies that provide access to and coverage for virtual psychotherapy,” the authors write.
SOURCE:
The study, led by Brian K. Ahmedani, PhD, with the Center for Health Policy and Health Services Research, Henry Ford Health, Detroit, was published online in Psychiatric Services.
LIMITATIONS:
The study was conducted in three large health systems with virtual care infrastructure already in place. Researchers did not examine use of virtual care for medication management or for types of care other than psychotherapy, which may present different challenges.
DISCLOSURES:
The study was supported by the National Institute of Mental Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Retrospective study using electronic health records and insurance claims data from three large U.S. health systems.
- Sample included 110,089 patients with mental health conditions who attended at least two psychotherapy visits during the 9 months before and 9 months after the onset of COVID-19, defined in this study as March 14, 2020.
- Outcome was disruption in psychotherapy, defined as a gap of more than 45 days between visits.
TAKEAWAY:
- Before the pandemic, 96.9% of psychotherapy visits were in person and 35.4% were followed by a gap of more than 45 days.
- After the onset of the pandemic, more than half of visits (51.8%) were virtual, and only 17.9% were followed by a gap of more than 45 days.
- Prior to the pandemic, the median time between visits was 27 days, and after the pandemic, it dropped to 14 days, suggesting individuals were more likely to return for additional psychotherapy after the widespread shift to virtual care.
- Over the entire study period, individuals with depressive, anxiety, or bipolar disorders were more likely to maintain consistent psychotherapy visits, whereas those with schizophrenia, ADHD, autism, conduct or disruptive disorders, dementia, or personality disorders were more likely to have a disruption in their visits.
IN PRACTICE:
“These findings support continued use of virtual psychotherapy as an option for care when appropriate infrastructure is in place. In addition, these findings support the continuation of policies that provide access to and coverage for virtual psychotherapy,” the authors write.
SOURCE:
The study, led by Brian K. Ahmedani, PhD, with the Center for Health Policy and Health Services Research, Henry Ford Health, Detroit, was published online in Psychiatric Services.
LIMITATIONS:
The study was conducted in three large health systems with virtual care infrastructure already in place. Researchers did not examine use of virtual care for medication management or for types of care other than psychotherapy, which may present different challenges.
DISCLOSURES:
The study was supported by the National Institute of Mental Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Retrospective study using electronic health records and insurance claims data from three large U.S. health systems.
- Sample included 110,089 patients with mental health conditions who attended at least two psychotherapy visits during the 9 months before and 9 months after the onset of COVID-19, defined in this study as March 14, 2020.
- Outcome was disruption in psychotherapy, defined as a gap of more than 45 days between visits.
TAKEAWAY:
- Before the pandemic, 96.9% of psychotherapy visits were in person and 35.4% were followed by a gap of more than 45 days.
- After the onset of the pandemic, more than half of visits (51.8%) were virtual, and only 17.9% were followed by a gap of more than 45 days.
- Prior to the pandemic, the median time between visits was 27 days, and after the pandemic, it dropped to 14 days, suggesting individuals were more likely to return for additional psychotherapy after the widespread shift to virtual care.
- Over the entire study period, individuals with depressive, anxiety, or bipolar disorders were more likely to maintain consistent psychotherapy visits, whereas those with schizophrenia, ADHD, autism, conduct or disruptive disorders, dementia, or personality disorders were more likely to have a disruption in their visits.
IN PRACTICE:
“These findings support continued use of virtual psychotherapy as an option for care when appropriate infrastructure is in place. In addition, these findings support the continuation of policies that provide access to and coverage for virtual psychotherapy,” the authors write.
SOURCE:
The study, led by Brian K. Ahmedani, PhD, with the Center for Health Policy and Health Services Research, Henry Ford Health, Detroit, was published online in Psychiatric Services.
LIMITATIONS:
The study was conducted in three large health systems with virtual care infrastructure already in place. Researchers did not examine use of virtual care for medication management or for types of care other than psychotherapy, which may present different challenges.
DISCLOSURES:
The study was supported by the National Institute of Mental Health. The authors have no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM PSYCHIATRIC SERVICES
Antidepressants ‘don’t blunt’ semaglutide and weight loss
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
in a post hoc analysis of the Semaglutide Treatment Effect in People with Obesity (STEP) program.
Adverse events, including psychiatric events, were slightly more usual in the patients on antidepressants, Robert Kushner, MD, noted, in an oral session at the annual meeting of the Obesity Society.
“It is very common that patients who present for weight management are taking antidepressants for various reasons, including depression, anxiety, insomnia, or chronic pain,”Dr. Kushner, from Northwestern University in Chicago, said in an email. “We wanted to see if these participants responded differently to semaglutide, compared to those not on antidepressants.”
“We found that antidepressants do not blunt the effect of semaglutide for weight loss,” he said. “However, there is a slight increase in reported adverse effects.”
“Semaglutide 2.4 mg provides an effective treatment option for weight management, regardless of antidepressant use at baseline,” Dr. Kushner summarized. “Clinicians should be assured that we can use semaglutide in this population of patients.”
Jack Yanovski, MD, PhD, said this was a “great presentation,” noting that “it’s really important that we understand what goes on in patients with depression.”
“Of course, all these trials still had rules that prevent the folks with the most severe depressive symptoms or past suicidality to participate,” added Dr. Yanovski, chief of the Growth and Obesity Section, Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md. “We need specific trials to know exactly how well we do.”
Dr. Kushner agreed, but also noted that, ever since some earlier antidepressants were associated with risk for suicidal ideation and death, strict guidelines were put in place that exclude certain patients from participating in clinical trials.
Dr. Yanovski suggested that now that the drugs are approved, it would be possible to study this, and the information would be important for clinicians.
Dr. Kushner said he hopes that such studies are forthcoming. In the meantime, “data like this will add some support and understanding,” he suggested.
36,000 Patients with obesity, 500 on antidepressants
Many people living with obesity report taking antidepressants for depression, anxiety, chronic pain, obsessive-compulsive disorder, sleep disturbance, neuropathy, panic disorder, or posttraumatic stress disorder, Dr. Kushner noted.
However, some of these medications can cause weight gain, and little is known about treatment outcomes for people with obesity who are on antidepressants, since most weight-loss studies exclude people with active major depressive disorder.
The researchers analyzed data from 1,961 patients in STEP 1 and 807 patients in STEP 2 as well as 611 patients in STEP 3 and 304 patients in STEP 5 – 3,683 participants in total, of which 539 were on antidepressants at baseline.
The patients were randomly assigned to 2.4 mg semaglutide vs. placebo plus a lifestyle intervention (STEP 1, 2, and 5) or intensive behavioral therapy (STEP 3 only), for 68 weeks, except STEP 5, which was 104 weeks.
Patients were included if they were aged 18 or older with a body mass index ≥30 kg/m2, or ≥27 kg/m2 with more than one weight-related complication (STEP 1, 3, and 5) or BMI ≥27 kg/m2 with type 2 diabetes (STEP 2 only), and at least one self-reported unsuccessful effort to lose weight by diet.
They were excluded if they had active major depressive disorder within 2 years prior to screening (or other severe psychiatric disorders such as schizophrenia or bipolar disorder) or a Patient Health Questionnaire-9 score of 15 or higher (indicating moderately severe or severe depression), or suicide ideation (type 4 or 5 on the Columbia Suicide Severity Rating Scale) or suicide behavior, within 30 days of screening.
From baseline to week 68, patients on semaglutide (with/without baseline antidepressant use) had a significantly greater change in weight vs. patients on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: –15.7% / –14.7% vs. –0.2% / –2.8%
- STEP 2: –10.7% / –9.5% vs. –3.3% / –3.4%
- STEP 3: –16.2% / –15.9% vs. –5.0% / –5.9%
- STEP 5: –19.0% / –14.1% vs. +1.6% / – 4.0%.
The proportion of reported adverse events was generally slightly greater in patients receiving semaglutide (with/without baseline antidepressant use) than those on placebo (with/without baseline antidepressant use), respectively:
- STEP 1: 97.7% vs 88.6% and 92.9% vs. 86%
- STEP 2: 97.6% vs 86.5% and 88.6% vs. 77.2%
- STEP 3: 97.6% vs 95.3% and 100% vs. 95.8%
- STEP 5: 100% vs 94.8% and 95.5% vs. 89.2%.
Gastrointestinal adverse events were more frequently reported in the semaglutide group and in patients on antidepressants at baseline. The proportion of patients with psychiatric adverse events was greater in participants on antidepressants at baseline. There were no differences in suicidal ideation/behavior in patients with/without antidepressant use at baseline.
The STEP trials were funded by Novo Nordisk. Dr. Kushner discloses that he served as a consultant for Novo Nordisk, WeightWatchers, Eli Lilly, and Pfizer, and received a research grant from Epitomee.
A version of this article appeared on Medscape.com.
FROM OBESITYWEEK® 2023
Repetitive primary care screenings may miss depression and anxiety
Routine screening for depression and anxiety at each primary care clinical encounter in order to meet performance metrics could compromise accuracy and clinical care, based on data from more than 380,000 individuals in primary care.
“Prioritizing repetition of intake screening questionnaires at primary care visits may have unintended consequences such as administrative burden, provision of low-value care, and reduced clinical capacity to deliver other, high-value services,” but the accuracy of workflow-based intake screening on subsequent diagnosis has not been explored, wrote Jodi Simon, DrPH, of AllianceChicago, Ill., and colleagues.
In a study published in the Annals of Family Medicine, the researchers reviewed data from screenings performed on 380,057 patients in primary care settings. They examined the accuracy and utility of the Patient Health Questionnaire (PHQ-2) for depression and the Generalized Anxiety Disorder 2 (GAD-2) for anxiety.
The data included 1,883,317 screenings with PHQ-2s and 1,573,107 with GAD-2s. Of these, 92.3% of PHQ-2 screenings and 91.4% of GAD-2 screenings indicated low likelihood of depression or anxiety (defined as cumulative scores of 0 or 1). Mean scores for the PHQ-2 and GAD-2 in the study population were 0.29 and 0.35, respectively.
In the current study, 11% of patients had positive PHQ-2 scores (defined as 2 or higher) vs. 47%-53% seen in previous studies and census data.
In an analysis of new diagnoses of depression and anxiety, the researchers found that 42.3% of patients with a new depression diagnosis were not identified on intake screening; they had scores of 0 or 1 on the PHQ-2 in the past 30 days. Similarly, 42.7% of patients with a new anxiety diagnosis had scores of 0 or 1 on the GAD-2 in the past 30 days.
In other words, “Screening only detected risk in 57.7% of patients subsequently diagnosed with depression and 57.3% of patients subsequently diagnosed with anxiety,” the researchers said. This low positivity rate in patients diagnosed within 30 days merits further research, they added.
More studies are needed, but preliminary interviews with patients, clinicians, and staff indicate that time constraints and variation in the administration of questionnaires are among the factors contributing to inaccurate screening, the researchers noted.
The current study results suggest that screenings for anxiety and depression may occur in a perfunctory or inconsistent manner that might compromise accuracy when they are part of the workflow for each clinical visit in order to meet performance metrics, they said. “Ineffective screening may unintentionally detract from clinical care because care teams and patients have less time and cognitive energy to focus on other priorities during busy clinical encounters,” they added.
Alternatively, , the researchers concluded.
The study was funded by the American Medical Association Transformation Initiative. The researchers had no financial conflicts to disclose.
Routine screening for depression and anxiety at each primary care clinical encounter in order to meet performance metrics could compromise accuracy and clinical care, based on data from more than 380,000 individuals in primary care.
“Prioritizing repetition of intake screening questionnaires at primary care visits may have unintended consequences such as administrative burden, provision of low-value care, and reduced clinical capacity to deliver other, high-value services,” but the accuracy of workflow-based intake screening on subsequent diagnosis has not been explored, wrote Jodi Simon, DrPH, of AllianceChicago, Ill., and colleagues.
In a study published in the Annals of Family Medicine, the researchers reviewed data from screenings performed on 380,057 patients in primary care settings. They examined the accuracy and utility of the Patient Health Questionnaire (PHQ-2) for depression and the Generalized Anxiety Disorder 2 (GAD-2) for anxiety.
The data included 1,883,317 screenings with PHQ-2s and 1,573,107 with GAD-2s. Of these, 92.3% of PHQ-2 screenings and 91.4% of GAD-2 screenings indicated low likelihood of depression or anxiety (defined as cumulative scores of 0 or 1). Mean scores for the PHQ-2 and GAD-2 in the study population were 0.29 and 0.35, respectively.
In the current study, 11% of patients had positive PHQ-2 scores (defined as 2 or higher) vs. 47%-53% seen in previous studies and census data.
In an analysis of new diagnoses of depression and anxiety, the researchers found that 42.3% of patients with a new depression diagnosis were not identified on intake screening; they had scores of 0 or 1 on the PHQ-2 in the past 30 days. Similarly, 42.7% of patients with a new anxiety diagnosis had scores of 0 or 1 on the GAD-2 in the past 30 days.
In other words, “Screening only detected risk in 57.7% of patients subsequently diagnosed with depression and 57.3% of patients subsequently diagnosed with anxiety,” the researchers said. This low positivity rate in patients diagnosed within 30 days merits further research, they added.
More studies are needed, but preliminary interviews with patients, clinicians, and staff indicate that time constraints and variation in the administration of questionnaires are among the factors contributing to inaccurate screening, the researchers noted.
The current study results suggest that screenings for anxiety and depression may occur in a perfunctory or inconsistent manner that might compromise accuracy when they are part of the workflow for each clinical visit in order to meet performance metrics, they said. “Ineffective screening may unintentionally detract from clinical care because care teams and patients have less time and cognitive energy to focus on other priorities during busy clinical encounters,” they added.
Alternatively, , the researchers concluded.
The study was funded by the American Medical Association Transformation Initiative. The researchers had no financial conflicts to disclose.
Routine screening for depression and anxiety at each primary care clinical encounter in order to meet performance metrics could compromise accuracy and clinical care, based on data from more than 380,000 individuals in primary care.
“Prioritizing repetition of intake screening questionnaires at primary care visits may have unintended consequences such as administrative burden, provision of low-value care, and reduced clinical capacity to deliver other, high-value services,” but the accuracy of workflow-based intake screening on subsequent diagnosis has not been explored, wrote Jodi Simon, DrPH, of AllianceChicago, Ill., and colleagues.
In a study published in the Annals of Family Medicine, the researchers reviewed data from screenings performed on 380,057 patients in primary care settings. They examined the accuracy and utility of the Patient Health Questionnaire (PHQ-2) for depression and the Generalized Anxiety Disorder 2 (GAD-2) for anxiety.
The data included 1,883,317 screenings with PHQ-2s and 1,573,107 with GAD-2s. Of these, 92.3% of PHQ-2 screenings and 91.4% of GAD-2 screenings indicated low likelihood of depression or anxiety (defined as cumulative scores of 0 or 1). Mean scores for the PHQ-2 and GAD-2 in the study population were 0.29 and 0.35, respectively.
In the current study, 11% of patients had positive PHQ-2 scores (defined as 2 or higher) vs. 47%-53% seen in previous studies and census data.
In an analysis of new diagnoses of depression and anxiety, the researchers found that 42.3% of patients with a new depression diagnosis were not identified on intake screening; they had scores of 0 or 1 on the PHQ-2 in the past 30 days. Similarly, 42.7% of patients with a new anxiety diagnosis had scores of 0 or 1 on the GAD-2 in the past 30 days.
In other words, “Screening only detected risk in 57.7% of patients subsequently diagnosed with depression and 57.3% of patients subsequently diagnosed with anxiety,” the researchers said. This low positivity rate in patients diagnosed within 30 days merits further research, they added.
More studies are needed, but preliminary interviews with patients, clinicians, and staff indicate that time constraints and variation in the administration of questionnaires are among the factors contributing to inaccurate screening, the researchers noted.
The current study results suggest that screenings for anxiety and depression may occur in a perfunctory or inconsistent manner that might compromise accuracy when they are part of the workflow for each clinical visit in order to meet performance metrics, they said. “Ineffective screening may unintentionally detract from clinical care because care teams and patients have less time and cognitive energy to focus on other priorities during busy clinical encounters,” they added.
Alternatively, , the researchers concluded.
The study was funded by the American Medical Association Transformation Initiative. The researchers had no financial conflicts to disclose.
FROM THE ANNALS OF FAMILY MEDICINE
Confirmed: Intermittent use of benzodiazepines is the safest option
BARCELONA – results of a large-scale study show.
Investigators matched more than 57,000 chronic benzodiazepine users with nearly 114,000 intermittent users and found that, at 1 year, chronic users had an 8% increased risk for emergency department visits and/or hospitalizations for falls.
Chronic users also had a 25% increased risk for hip fracture, a 4% raised risk for ED visits and/or hospitalizations for any reason, and a 23% increased risk for death.
Study investigator Simon J.C. Davies, MD, PhD, MSc, Centre for Addiction & Mental Health, Toronto, said that the research shows that, where possible, patients older than 65 years with anxiety or insomnia who are taking benzodiazepines should not stay on these medications continuously.
However, he acknowledged that, “in practical terms, there will be some who can’t change or do not want to change” their treatment.
The findings were presented at the annual meeting of the European College of Neuropsychopharmacology.
Wide range of adverse outcomes
The authors noted that benzodiazepines are used to treat anxiety and insomnia but are associated with a range of adverse outcomes, including falls, fractures, cognitive impairment, and mortality as well as tolerance and dose escalation.
“These risks are especially relevant in older adults,” they added, noting that some guidelines recommend avoiding the drugs in this population, whereas other suggest short-term benzodiazepine use for a maximum of 4 weeks.
Despite this, “benzodiazepines are widely prescribed in older adults.” One study showed that almost 15% of adults aged 65 years or older received at least one benzodiazepine prescription.
Moreover, chronic use is more common in older versus younger patients.
Benzodiazepine use among older adults “used to be higher,” Dr. Davies said in an interview, at around 20%, but the “numbers have come down,” partly because of the introduction of benzodiazepine-like sleep medications but also because of educational efforts.
“There are certainly campaigns in Ontario to educate physicians,” Dr. Davies said, “but I think more broadly people are aware of the activity of these drugs, and the tolerance and other issues.”
To compare the risk associated with chronic versus intermittent use of benzodiazepines in older adults, the team performed a population-based cohort study using linked health care databases in Ontario.
They focused on adults aged 65 years or older with a first benzodiazepine prescription after at least 1 year without taking the drugs.
Chronic benzodiazepine use was defined as 120 days of prescriptions over the first 180 days after the index prescription. Patients who met these criteria were matched with intermittent users in a 2:1 ratio by age and sex.
Patients were then propensity matched using 24 variables, including health system use in the year prior to the index prescription, clinical diagnoses, prior psychiatric health system use, falls, and income level.
The team identified 57,072 chronic benzodiazepine users and 312,468 intermittent users, of whom, 57,041 and 113,839, respectively, were propensity matched.
As expected, chronic users were prescribed benzodiazepines for more days than were the intermittent users over both the initial 180-day exposure period, at 141 days versus 33 days, and again during a further 180-day follow-up period, at 181 days versus 19 days.
Over the follow-up period, the daily lorazepam dose-equivalents of chronic users four times that of intermittent users.
Hospitalizations and/or ED visits for falls were higher among patients in the chronic benzodiazepine group, at 4.6% versus 3.2% in those who took the drugs intermittently.
After adjusting for benzodiazepine dose, the team found that chronic benzodiazepine use was associated with a significant increase in the risk for falls leading to hospital presentation over the 360-day study period, compared with intermittent use (hazard ratio, 1.08; P = .0124).
Sex differences
In addition, chronic use was linked to a significantly increased risk for hip fracture (HR, 1.25; P = .0095), and long-term care admission (HR, 1.32; P < .0001).
There was also a significant increase in ED visits and/or hospitalizations for any reason with chronic benzodiazepine use versus intermittent use (HR, 1.04; P = .0007), and an increase in the risk for death (HR, 1.23; P < .0001).
A nonsignificant increased risk for wrist fracture was also associated with chronic use of benzodiazepines (HR, 1.02; P = .8683).
Further analysis revealed some sex differences. For instance, men had a marked increase in the risk for hip fracture with chronic use (HR, 1.50; P = .0154), whereas the risk was not significant in women (HR, 1.16; P = .1332). In addition, mortality risk associated with chronic use was higher in men than in women (HR, 1.39; P < .0001 vs. HR, 1.10; P = .2245).
The decision to discontinue chronic benzodiazepine use can be challenging, said Dr. Davies. “If you’re advising people to stop, what happens to the treatment of their anxiety?”
He said that there are many other treatment options for anxiety that don’t come with tolerance or risk for addiction.
“My position would be that intermittent use is perfectly acceptable while you bide your time to explore other treatments. They may be pharmacological; they may, of course, be lifestyle changes, psychotherapies, and so on,” said Dr. Davies.
If, however, patients feel that chronic benzodiazepine use is their only option, this research informs that decision by quantifying the risks.
“We’ve always known that there was a problem, but there haven’t been high-quality epidemiological studies like this that allowed us to say what the numbers are,” said Dr. Davies.
Confirmatory research
In a comment, Christoph U. Correll, MD, professor of psychiatry at Hofstra University, Hempstead, N.Y., noted that the risk associated with benzodiazepine use, especially in older people, has been demonstrated repeatedly.
“In that context, it is not surprising that less continuous exposure to an established risk factor attenuates the risk for these adverse outcomes,” he said.
Dr. Correll, who was not involved in the study pointed out there is nevertheless a “risk of residual confounding by indication.”
In other words, “people with intermittent benzodiazepine use may have less severe underlying illness and better healthy lifestyle behaviors than those requiring chronic benzodiazepine administration.”
Also commenting on the research, Christian Vinkers, MD, PhD, psychiatrist and professor of stress and resilience, Amsterdam University Medical Centre, said that it confirms “once again that long-term benzodiazepine use should not be encouraged.”
“The risk of falls, as well as cognitive side effects and impaired driving skills, with the risk of road accidents, make chronic overuse of benzodiazepines a public health issue. Of course, there is a small group of patients who should have access to long-term use, but it is reasonable to assume that this group is currently too large,” he added.
The study was funded through a grant from the University of Toronto Department of Psychiatry Excellence Funds. No relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
BARCELONA – results of a large-scale study show.
Investigators matched more than 57,000 chronic benzodiazepine users with nearly 114,000 intermittent users and found that, at 1 year, chronic users had an 8% increased risk for emergency department visits and/or hospitalizations for falls.
Chronic users also had a 25% increased risk for hip fracture, a 4% raised risk for ED visits and/or hospitalizations for any reason, and a 23% increased risk for death.
Study investigator Simon J.C. Davies, MD, PhD, MSc, Centre for Addiction & Mental Health, Toronto, said that the research shows that, where possible, patients older than 65 years with anxiety or insomnia who are taking benzodiazepines should not stay on these medications continuously.
However, he acknowledged that, “in practical terms, there will be some who can’t change or do not want to change” their treatment.
The findings were presented at the annual meeting of the European College of Neuropsychopharmacology.
Wide range of adverse outcomes
The authors noted that benzodiazepines are used to treat anxiety and insomnia but are associated with a range of adverse outcomes, including falls, fractures, cognitive impairment, and mortality as well as tolerance and dose escalation.
“These risks are especially relevant in older adults,” they added, noting that some guidelines recommend avoiding the drugs in this population, whereas other suggest short-term benzodiazepine use for a maximum of 4 weeks.
Despite this, “benzodiazepines are widely prescribed in older adults.” One study showed that almost 15% of adults aged 65 years or older received at least one benzodiazepine prescription.
Moreover, chronic use is more common in older versus younger patients.
Benzodiazepine use among older adults “used to be higher,” Dr. Davies said in an interview, at around 20%, but the “numbers have come down,” partly because of the introduction of benzodiazepine-like sleep medications but also because of educational efforts.
“There are certainly campaigns in Ontario to educate physicians,” Dr. Davies said, “but I think more broadly people are aware of the activity of these drugs, and the tolerance and other issues.”
To compare the risk associated with chronic versus intermittent use of benzodiazepines in older adults, the team performed a population-based cohort study using linked health care databases in Ontario.
They focused on adults aged 65 years or older with a first benzodiazepine prescription after at least 1 year without taking the drugs.
Chronic benzodiazepine use was defined as 120 days of prescriptions over the first 180 days after the index prescription. Patients who met these criteria were matched with intermittent users in a 2:1 ratio by age and sex.
Patients were then propensity matched using 24 variables, including health system use in the year prior to the index prescription, clinical diagnoses, prior psychiatric health system use, falls, and income level.
The team identified 57,072 chronic benzodiazepine users and 312,468 intermittent users, of whom, 57,041 and 113,839, respectively, were propensity matched.
As expected, chronic users were prescribed benzodiazepines for more days than were the intermittent users over both the initial 180-day exposure period, at 141 days versus 33 days, and again during a further 180-day follow-up period, at 181 days versus 19 days.
Over the follow-up period, the daily lorazepam dose-equivalents of chronic users four times that of intermittent users.
Hospitalizations and/or ED visits for falls were higher among patients in the chronic benzodiazepine group, at 4.6% versus 3.2% in those who took the drugs intermittently.
After adjusting for benzodiazepine dose, the team found that chronic benzodiazepine use was associated with a significant increase in the risk for falls leading to hospital presentation over the 360-day study period, compared with intermittent use (hazard ratio, 1.08; P = .0124).
Sex differences
In addition, chronic use was linked to a significantly increased risk for hip fracture (HR, 1.25; P = .0095), and long-term care admission (HR, 1.32; P < .0001).
There was also a significant increase in ED visits and/or hospitalizations for any reason with chronic benzodiazepine use versus intermittent use (HR, 1.04; P = .0007), and an increase in the risk for death (HR, 1.23; P < .0001).
A nonsignificant increased risk for wrist fracture was also associated with chronic use of benzodiazepines (HR, 1.02; P = .8683).
Further analysis revealed some sex differences. For instance, men had a marked increase in the risk for hip fracture with chronic use (HR, 1.50; P = .0154), whereas the risk was not significant in women (HR, 1.16; P = .1332). In addition, mortality risk associated with chronic use was higher in men than in women (HR, 1.39; P < .0001 vs. HR, 1.10; P = .2245).
The decision to discontinue chronic benzodiazepine use can be challenging, said Dr. Davies. “If you’re advising people to stop, what happens to the treatment of their anxiety?”
He said that there are many other treatment options for anxiety that don’t come with tolerance or risk for addiction.
“My position would be that intermittent use is perfectly acceptable while you bide your time to explore other treatments. They may be pharmacological; they may, of course, be lifestyle changes, psychotherapies, and so on,” said Dr. Davies.
If, however, patients feel that chronic benzodiazepine use is their only option, this research informs that decision by quantifying the risks.
“We’ve always known that there was a problem, but there haven’t been high-quality epidemiological studies like this that allowed us to say what the numbers are,” said Dr. Davies.
Confirmatory research
In a comment, Christoph U. Correll, MD, professor of psychiatry at Hofstra University, Hempstead, N.Y., noted that the risk associated with benzodiazepine use, especially in older people, has been demonstrated repeatedly.
“In that context, it is not surprising that less continuous exposure to an established risk factor attenuates the risk for these adverse outcomes,” he said.
Dr. Correll, who was not involved in the study pointed out there is nevertheless a “risk of residual confounding by indication.”
In other words, “people with intermittent benzodiazepine use may have less severe underlying illness and better healthy lifestyle behaviors than those requiring chronic benzodiazepine administration.”
Also commenting on the research, Christian Vinkers, MD, PhD, psychiatrist and professor of stress and resilience, Amsterdam University Medical Centre, said that it confirms “once again that long-term benzodiazepine use should not be encouraged.”
“The risk of falls, as well as cognitive side effects and impaired driving skills, with the risk of road accidents, make chronic overuse of benzodiazepines a public health issue. Of course, there is a small group of patients who should have access to long-term use, but it is reasonable to assume that this group is currently too large,” he added.
The study was funded through a grant from the University of Toronto Department of Psychiatry Excellence Funds. No relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
BARCELONA – results of a large-scale study show.
Investigators matched more than 57,000 chronic benzodiazepine users with nearly 114,000 intermittent users and found that, at 1 year, chronic users had an 8% increased risk for emergency department visits and/or hospitalizations for falls.
Chronic users also had a 25% increased risk for hip fracture, a 4% raised risk for ED visits and/or hospitalizations for any reason, and a 23% increased risk for death.
Study investigator Simon J.C. Davies, MD, PhD, MSc, Centre for Addiction & Mental Health, Toronto, said that the research shows that, where possible, patients older than 65 years with anxiety or insomnia who are taking benzodiazepines should not stay on these medications continuously.
However, he acknowledged that, “in practical terms, there will be some who can’t change or do not want to change” their treatment.
The findings were presented at the annual meeting of the European College of Neuropsychopharmacology.
Wide range of adverse outcomes
The authors noted that benzodiazepines are used to treat anxiety and insomnia but are associated with a range of adverse outcomes, including falls, fractures, cognitive impairment, and mortality as well as tolerance and dose escalation.
“These risks are especially relevant in older adults,” they added, noting that some guidelines recommend avoiding the drugs in this population, whereas other suggest short-term benzodiazepine use for a maximum of 4 weeks.
Despite this, “benzodiazepines are widely prescribed in older adults.” One study showed that almost 15% of adults aged 65 years or older received at least one benzodiazepine prescription.
Moreover, chronic use is more common in older versus younger patients.
Benzodiazepine use among older adults “used to be higher,” Dr. Davies said in an interview, at around 20%, but the “numbers have come down,” partly because of the introduction of benzodiazepine-like sleep medications but also because of educational efforts.
“There are certainly campaigns in Ontario to educate physicians,” Dr. Davies said, “but I think more broadly people are aware of the activity of these drugs, and the tolerance and other issues.”
To compare the risk associated with chronic versus intermittent use of benzodiazepines in older adults, the team performed a population-based cohort study using linked health care databases in Ontario.
They focused on adults aged 65 years or older with a first benzodiazepine prescription after at least 1 year without taking the drugs.
Chronic benzodiazepine use was defined as 120 days of prescriptions over the first 180 days after the index prescription. Patients who met these criteria were matched with intermittent users in a 2:1 ratio by age and sex.
Patients were then propensity matched using 24 variables, including health system use in the year prior to the index prescription, clinical diagnoses, prior psychiatric health system use, falls, and income level.
The team identified 57,072 chronic benzodiazepine users and 312,468 intermittent users, of whom, 57,041 and 113,839, respectively, were propensity matched.
As expected, chronic users were prescribed benzodiazepines for more days than were the intermittent users over both the initial 180-day exposure period, at 141 days versus 33 days, and again during a further 180-day follow-up period, at 181 days versus 19 days.
Over the follow-up period, the daily lorazepam dose-equivalents of chronic users four times that of intermittent users.
Hospitalizations and/or ED visits for falls were higher among patients in the chronic benzodiazepine group, at 4.6% versus 3.2% in those who took the drugs intermittently.
After adjusting for benzodiazepine dose, the team found that chronic benzodiazepine use was associated with a significant increase in the risk for falls leading to hospital presentation over the 360-day study period, compared with intermittent use (hazard ratio, 1.08; P = .0124).
Sex differences
In addition, chronic use was linked to a significantly increased risk for hip fracture (HR, 1.25; P = .0095), and long-term care admission (HR, 1.32; P < .0001).
There was also a significant increase in ED visits and/or hospitalizations for any reason with chronic benzodiazepine use versus intermittent use (HR, 1.04; P = .0007), and an increase in the risk for death (HR, 1.23; P < .0001).
A nonsignificant increased risk for wrist fracture was also associated with chronic use of benzodiazepines (HR, 1.02; P = .8683).
Further analysis revealed some sex differences. For instance, men had a marked increase in the risk for hip fracture with chronic use (HR, 1.50; P = .0154), whereas the risk was not significant in women (HR, 1.16; P = .1332). In addition, mortality risk associated with chronic use was higher in men than in women (HR, 1.39; P < .0001 vs. HR, 1.10; P = .2245).
The decision to discontinue chronic benzodiazepine use can be challenging, said Dr. Davies. “If you’re advising people to stop, what happens to the treatment of their anxiety?”
He said that there are many other treatment options for anxiety that don’t come with tolerance or risk for addiction.
“My position would be that intermittent use is perfectly acceptable while you bide your time to explore other treatments. They may be pharmacological; they may, of course, be lifestyle changes, psychotherapies, and so on,” said Dr. Davies.
If, however, patients feel that chronic benzodiazepine use is their only option, this research informs that decision by quantifying the risks.
“We’ve always known that there was a problem, but there haven’t been high-quality epidemiological studies like this that allowed us to say what the numbers are,” said Dr. Davies.
Confirmatory research
In a comment, Christoph U. Correll, MD, professor of psychiatry at Hofstra University, Hempstead, N.Y., noted that the risk associated with benzodiazepine use, especially in older people, has been demonstrated repeatedly.
“In that context, it is not surprising that less continuous exposure to an established risk factor attenuates the risk for these adverse outcomes,” he said.
Dr. Correll, who was not involved in the study pointed out there is nevertheless a “risk of residual confounding by indication.”
In other words, “people with intermittent benzodiazepine use may have less severe underlying illness and better healthy lifestyle behaviors than those requiring chronic benzodiazepine administration.”
Also commenting on the research, Christian Vinkers, MD, PhD, psychiatrist and professor of stress and resilience, Amsterdam University Medical Centre, said that it confirms “once again that long-term benzodiazepine use should not be encouraged.”
“The risk of falls, as well as cognitive side effects and impaired driving skills, with the risk of road accidents, make chronic overuse of benzodiazepines a public health issue. Of course, there is a small group of patients who should have access to long-term use, but it is reasonable to assume that this group is currently too large,” he added.
The study was funded through a grant from the University of Toronto Department of Psychiatry Excellence Funds. No relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
AT ECNP 2023
CBT effectively treats sexual concerns in menopausal women
PHILADELPHIA – . Four CBT sessions specifically focused on sexual concerns resulted in decreased sexual distress and concern, reduced depressive and menopausal symptoms, and increased sexual desire and functioning, as well as improved body image and relationship satisfaction.
An estimated 68%-87% of perimenopausal and postmenopausal women report sexual concerns, Sheryl Green, PhD, CPsych, an associate professor of psychiatry and behavioral neurosciences at McMaster University and a psychologist at St. Joseph’s Healthcare’s Women’s Health Concerns Clinic, both in Hamilton, Ont., told attendees at the meeting.
“Sexual concerns over the menopausal transition are not just physical, but they’re also psychological and emotional,” Dr. Green said. “Three common challenges include decreased sexual desire, a reduction in physical arousal and ability to achieve an orgasm, and sexual pain and discomfort during intercourse.”
The reasons for these concerns are multifactorial, she said. Decreased sexual desire can stem from stress, medical problems, their relationship with their partner, or other causes. A woman’s difficulty with reduced physical arousal or ability to have an orgasm can result from changes in hormone levels and vaginal changes, such as vaginal atrophy, which can also contribute to the sexual pain or discomfort reported by 17%-45% of postmenopausal women.
Two pharmacologic treatments exist for sexual concerns: oral flibanserin (Addyi) and injectable bremelanotide (Vyleesi). But many women may be unable or unwilling to take medication for their concerns. Previous research from Lori Brotto has found cognitive behavioral therapy and mindfulness interventions to effectively improve sexual functioning in women treated for gynecologic cancer and in women without a history of cancer.
“Sexual function needs to be understood from a bio-psychosocial model, looking at the biologic factors, the psychological factors, the sociocultural factors, and the interpersonal factors,” Sheryl Kingsberg, PhD, a professor of psychiatry and reproductive biology at Case Western Reserve University and a psychologist at University Hospitals in Cleveland, said in an interview.
“They can all overlap, and the clinician can ask a few pointed questions that help identify what the source of the problem is,” said Dr. Kingsberg, who was not involved in this study. She noted that the International Society for the Study of Women’s Sexual Health has an algorithm that can help in determining the source of the problems.
“Sometimes it’s going to be a biologic condition for which pharmacologic options are nice, but even if it is primarily pharmacologic, psychotherapy is always useful,” Dr. Kingsberg said. “Once the problem is there, even if it’s biologically based, then you have all the things in terms of the cognitive distortion, anxiety,” and other issues that a cognitive behavioral approach can help address. “And access is now much wider because of telehealth,” she added.
‘Psychology of menopause’
The study led by Dr. Green focused on peri- and postmenopausal women, with an average age of 50, who were experiencing primary sexual concerns based on a score of at least 26 on the Female Sexual Function Index (FSFI). Among the 20 women recruited for the study, 6 had already been prescribed hormone therapy for sexual concerns.
All reported decreased sexual desire, 17 reported decreased sexual arousal, 14 had body image dissatisfaction related to sexual concerns, and 6 reported urogenital problems. Nine of the women were in full remission from major depressive disorder, one had post-traumatic stress syndrome, and one had subclinical generalized anxiety disorder.
After spending 4 weeks on a wait list as self-control group for the study, the 15 women who completed the trial underwent four individual CBT sessions focusing on sexual concerns. The first session focused on psychoeducation and thought monitoring, and the second focused on cognitive distortions, cognitive strategies, and unhelpful beliefs or expectations related to sexual concerns. The third session looked at the role of problematic behaviors and behavioral experiments, and the fourth focused on continuation of strategies, long-term goals, and maintaining gains.
The participants completed eight measures at baseline, after the 4 weeks on the wait list, and after the four CBT sessions to assess the following:
- Sexual satisfaction, distress, and desire, using the FSFI, the Female Sexual Distress Scale-Revised (FSDS-R), and the Female Sexual Desire Questionnaire (FSDQ).
- Menopause symptoms, using the Greene Climacteric Scale (GCS).
- Body image, using the Dresden Body Image Questionnaire (DBIQ).
- Relationship satisfaction, using the Couples Satisfaction Index (CSI).
- Depression, using the Beck Depression Inventory-II (BDI-II).
- Anxiety, using the Hamilton Anxiety Rating Scale (HAM-A).
The women did not experience any significant changes while on the wait list except a slight decrease on the FSDQ concern subscale. Following the CBT sessions, however, the women experienced a significant decrease in sexual distress and concern as well as an increase in sexual dyadic desire and sexual functioning (P = .003 for FSFI, P = .002 for FSDS-R, and P = .003 for FSDQ).
Participants also experienced a decrease in depression (P < .0001) and menopausal symptoms (P = .001) and an increase in body-image satisfaction (P = .018) and relationship satisfaction (P = .0011) after the CBT sessions. The researchers assessed participants’ satisfaction with the Client Satisfaction Questionnaire after the CBT sessions and reported some of the qualitative findings.
“The treatment program was able to assist me with recognizing that some of my sexual concerns were normal, emotional as well as physical and hormonal, and provided me the ability to delve more deeply into the psychology of menopause and how to work through symptoms and concerns in more manageable pieces,” one participant wrote. Another found helpful the “homework exercises of recognizing a thought/feeling/emotion surrounding how I feel about myself/body and working through. More positive thought pattern/restructuring a response the most helpful.”
The main complaint about the program was that it was too short, with women wanting more sessions to help continue their progress.
Not an ‘either-or’ approach
Dr. Kingsberg said ISSWSH has a variety of sexual medicine practitioners, including providers who can provide CBT for sexual concerns, and the American Association of Sexuality Educators, Counselors and Therapists has a referral directory.
“Keeping in mind the bio-psychosocial model, sometimes psychotherapy is going to be a really effective treatment for sexual concerns,” Dr. Kingsberg said. “Sometimes the pharmacologic option is going to be a really effective treatment for some concerns, and sometimes the combination is going to have a really nice treatment effect. So it’s not a one-size-fits-all, and it doesn’t have to be an either-or.”
The sexual concerns of women still do not get adequately addressed in medical schools and residencies, Dr. Kingsberg said, which is distinctly different from how male sexual concerns are addressed in health care.
“Erectile dysfunction is kind of in the norm, and women are still a little hesitant to bring up their sexual concerns,” Dr. Kingsberg said. “They don’t know if it’s appropriate and they’re hoping that their clinician will ask.”
One way clinicians can do that is with a global question for all their patients: “Most of my patients have sexual questions or concerns; what concerns do you have?”
“They don’t have to go through a checklist of 10 things,” Dr. Kingsberg said. If the patient does not bring anything up, providers can then ask a single follow up question: “Do you have any concerns with desire, arousal, orgasm, or pain?” That question, Dr. Kingsberg said, covers the four main areas of concern.
The study was funded by the Canadian Institute of Health Research. Dr. Green reported no disclosures. Dr. Kingsberg has consulted for or served on the advisory board for Alloy, Astellas, Bayer, Dare Bioscience, Freya, Reunion Neuroscience, Materna Medical, Madorra, Palatin, Pfizer, ReJoy, Sprout, Strategic Science Technologies, and MsMedicine.
PHILADELPHIA – . Four CBT sessions specifically focused on sexual concerns resulted in decreased sexual distress and concern, reduced depressive and menopausal symptoms, and increased sexual desire and functioning, as well as improved body image and relationship satisfaction.
An estimated 68%-87% of perimenopausal and postmenopausal women report sexual concerns, Sheryl Green, PhD, CPsych, an associate professor of psychiatry and behavioral neurosciences at McMaster University and a psychologist at St. Joseph’s Healthcare’s Women’s Health Concerns Clinic, both in Hamilton, Ont., told attendees at the meeting.
“Sexual concerns over the menopausal transition are not just physical, but they’re also psychological and emotional,” Dr. Green said. “Three common challenges include decreased sexual desire, a reduction in physical arousal and ability to achieve an orgasm, and sexual pain and discomfort during intercourse.”
The reasons for these concerns are multifactorial, she said. Decreased sexual desire can stem from stress, medical problems, their relationship with their partner, or other causes. A woman’s difficulty with reduced physical arousal or ability to have an orgasm can result from changes in hormone levels and vaginal changes, such as vaginal atrophy, which can also contribute to the sexual pain or discomfort reported by 17%-45% of postmenopausal women.
Two pharmacologic treatments exist for sexual concerns: oral flibanserin (Addyi) and injectable bremelanotide (Vyleesi). But many women may be unable or unwilling to take medication for their concerns. Previous research from Lori Brotto has found cognitive behavioral therapy and mindfulness interventions to effectively improve sexual functioning in women treated for gynecologic cancer and in women without a history of cancer.
“Sexual function needs to be understood from a bio-psychosocial model, looking at the biologic factors, the psychological factors, the sociocultural factors, and the interpersonal factors,” Sheryl Kingsberg, PhD, a professor of psychiatry and reproductive biology at Case Western Reserve University and a psychologist at University Hospitals in Cleveland, said in an interview.
“They can all overlap, and the clinician can ask a few pointed questions that help identify what the source of the problem is,” said Dr. Kingsberg, who was not involved in this study. She noted that the International Society for the Study of Women’s Sexual Health has an algorithm that can help in determining the source of the problems.
“Sometimes it’s going to be a biologic condition for which pharmacologic options are nice, but even if it is primarily pharmacologic, psychotherapy is always useful,” Dr. Kingsberg said. “Once the problem is there, even if it’s biologically based, then you have all the things in terms of the cognitive distortion, anxiety,” and other issues that a cognitive behavioral approach can help address. “And access is now much wider because of telehealth,” she added.
‘Psychology of menopause’
The study led by Dr. Green focused on peri- and postmenopausal women, with an average age of 50, who were experiencing primary sexual concerns based on a score of at least 26 on the Female Sexual Function Index (FSFI). Among the 20 women recruited for the study, 6 had already been prescribed hormone therapy for sexual concerns.
All reported decreased sexual desire, 17 reported decreased sexual arousal, 14 had body image dissatisfaction related to sexual concerns, and 6 reported urogenital problems. Nine of the women were in full remission from major depressive disorder, one had post-traumatic stress syndrome, and one had subclinical generalized anxiety disorder.
After spending 4 weeks on a wait list as self-control group for the study, the 15 women who completed the trial underwent four individual CBT sessions focusing on sexual concerns. The first session focused on psychoeducation and thought monitoring, and the second focused on cognitive distortions, cognitive strategies, and unhelpful beliefs or expectations related to sexual concerns. The third session looked at the role of problematic behaviors and behavioral experiments, and the fourth focused on continuation of strategies, long-term goals, and maintaining gains.
The participants completed eight measures at baseline, after the 4 weeks on the wait list, and after the four CBT sessions to assess the following:
- Sexual satisfaction, distress, and desire, using the FSFI, the Female Sexual Distress Scale-Revised (FSDS-R), and the Female Sexual Desire Questionnaire (FSDQ).
- Menopause symptoms, using the Greene Climacteric Scale (GCS).
- Body image, using the Dresden Body Image Questionnaire (DBIQ).
- Relationship satisfaction, using the Couples Satisfaction Index (CSI).
- Depression, using the Beck Depression Inventory-II (BDI-II).
- Anxiety, using the Hamilton Anxiety Rating Scale (HAM-A).
The women did not experience any significant changes while on the wait list except a slight decrease on the FSDQ concern subscale. Following the CBT sessions, however, the women experienced a significant decrease in sexual distress and concern as well as an increase in sexual dyadic desire and sexual functioning (P = .003 for FSFI, P = .002 for FSDS-R, and P = .003 for FSDQ).
Participants also experienced a decrease in depression (P < .0001) and menopausal symptoms (P = .001) and an increase in body-image satisfaction (P = .018) and relationship satisfaction (P = .0011) after the CBT sessions. The researchers assessed participants’ satisfaction with the Client Satisfaction Questionnaire after the CBT sessions and reported some of the qualitative findings.
“The treatment program was able to assist me with recognizing that some of my sexual concerns were normal, emotional as well as physical and hormonal, and provided me the ability to delve more deeply into the psychology of menopause and how to work through symptoms and concerns in more manageable pieces,” one participant wrote. Another found helpful the “homework exercises of recognizing a thought/feeling/emotion surrounding how I feel about myself/body and working through. More positive thought pattern/restructuring a response the most helpful.”
The main complaint about the program was that it was too short, with women wanting more sessions to help continue their progress.
Not an ‘either-or’ approach
Dr. Kingsberg said ISSWSH has a variety of sexual medicine practitioners, including providers who can provide CBT for sexual concerns, and the American Association of Sexuality Educators, Counselors and Therapists has a referral directory.
“Keeping in mind the bio-psychosocial model, sometimes psychotherapy is going to be a really effective treatment for sexual concerns,” Dr. Kingsberg said. “Sometimes the pharmacologic option is going to be a really effective treatment for some concerns, and sometimes the combination is going to have a really nice treatment effect. So it’s not a one-size-fits-all, and it doesn’t have to be an either-or.”
The sexual concerns of women still do not get adequately addressed in medical schools and residencies, Dr. Kingsberg said, which is distinctly different from how male sexual concerns are addressed in health care.
“Erectile dysfunction is kind of in the norm, and women are still a little hesitant to bring up their sexual concerns,” Dr. Kingsberg said. “They don’t know if it’s appropriate and they’re hoping that their clinician will ask.”
One way clinicians can do that is with a global question for all their patients: “Most of my patients have sexual questions or concerns; what concerns do you have?”
“They don’t have to go through a checklist of 10 things,” Dr. Kingsberg said. If the patient does not bring anything up, providers can then ask a single follow up question: “Do you have any concerns with desire, arousal, orgasm, or pain?” That question, Dr. Kingsberg said, covers the four main areas of concern.
The study was funded by the Canadian Institute of Health Research. Dr. Green reported no disclosures. Dr. Kingsberg has consulted for or served on the advisory board for Alloy, Astellas, Bayer, Dare Bioscience, Freya, Reunion Neuroscience, Materna Medical, Madorra, Palatin, Pfizer, ReJoy, Sprout, Strategic Science Technologies, and MsMedicine.
PHILADELPHIA – . Four CBT sessions specifically focused on sexual concerns resulted in decreased sexual distress and concern, reduced depressive and menopausal symptoms, and increased sexual desire and functioning, as well as improved body image and relationship satisfaction.
An estimated 68%-87% of perimenopausal and postmenopausal women report sexual concerns, Sheryl Green, PhD, CPsych, an associate professor of psychiatry and behavioral neurosciences at McMaster University and a psychologist at St. Joseph’s Healthcare’s Women’s Health Concerns Clinic, both in Hamilton, Ont., told attendees at the meeting.
“Sexual concerns over the menopausal transition are not just physical, but they’re also psychological and emotional,” Dr. Green said. “Three common challenges include decreased sexual desire, a reduction in physical arousal and ability to achieve an orgasm, and sexual pain and discomfort during intercourse.”
The reasons for these concerns are multifactorial, she said. Decreased sexual desire can stem from stress, medical problems, their relationship with their partner, or other causes. A woman’s difficulty with reduced physical arousal or ability to have an orgasm can result from changes in hormone levels and vaginal changes, such as vaginal atrophy, which can also contribute to the sexual pain or discomfort reported by 17%-45% of postmenopausal women.
Two pharmacologic treatments exist for sexual concerns: oral flibanserin (Addyi) and injectable bremelanotide (Vyleesi). But many women may be unable or unwilling to take medication for their concerns. Previous research from Lori Brotto has found cognitive behavioral therapy and mindfulness interventions to effectively improve sexual functioning in women treated for gynecologic cancer and in women without a history of cancer.
“Sexual function needs to be understood from a bio-psychosocial model, looking at the biologic factors, the psychological factors, the sociocultural factors, and the interpersonal factors,” Sheryl Kingsberg, PhD, a professor of psychiatry and reproductive biology at Case Western Reserve University and a psychologist at University Hospitals in Cleveland, said in an interview.
“They can all overlap, and the clinician can ask a few pointed questions that help identify what the source of the problem is,” said Dr. Kingsberg, who was not involved in this study. She noted that the International Society for the Study of Women’s Sexual Health has an algorithm that can help in determining the source of the problems.
“Sometimes it’s going to be a biologic condition for which pharmacologic options are nice, but even if it is primarily pharmacologic, psychotherapy is always useful,” Dr. Kingsberg said. “Once the problem is there, even if it’s biologically based, then you have all the things in terms of the cognitive distortion, anxiety,” and other issues that a cognitive behavioral approach can help address. “And access is now much wider because of telehealth,” she added.
‘Psychology of menopause’
The study led by Dr. Green focused on peri- and postmenopausal women, with an average age of 50, who were experiencing primary sexual concerns based on a score of at least 26 on the Female Sexual Function Index (FSFI). Among the 20 women recruited for the study, 6 had already been prescribed hormone therapy for sexual concerns.
All reported decreased sexual desire, 17 reported decreased sexual arousal, 14 had body image dissatisfaction related to sexual concerns, and 6 reported urogenital problems. Nine of the women were in full remission from major depressive disorder, one had post-traumatic stress syndrome, and one had subclinical generalized anxiety disorder.
After spending 4 weeks on a wait list as self-control group for the study, the 15 women who completed the trial underwent four individual CBT sessions focusing on sexual concerns. The first session focused on psychoeducation and thought monitoring, and the second focused on cognitive distortions, cognitive strategies, and unhelpful beliefs or expectations related to sexual concerns. The third session looked at the role of problematic behaviors and behavioral experiments, and the fourth focused on continuation of strategies, long-term goals, and maintaining gains.
The participants completed eight measures at baseline, after the 4 weeks on the wait list, and after the four CBT sessions to assess the following:
- Sexual satisfaction, distress, and desire, using the FSFI, the Female Sexual Distress Scale-Revised (FSDS-R), and the Female Sexual Desire Questionnaire (FSDQ).
- Menopause symptoms, using the Greene Climacteric Scale (GCS).
- Body image, using the Dresden Body Image Questionnaire (DBIQ).
- Relationship satisfaction, using the Couples Satisfaction Index (CSI).
- Depression, using the Beck Depression Inventory-II (BDI-II).
- Anxiety, using the Hamilton Anxiety Rating Scale (HAM-A).
The women did not experience any significant changes while on the wait list except a slight decrease on the FSDQ concern subscale. Following the CBT sessions, however, the women experienced a significant decrease in sexual distress and concern as well as an increase in sexual dyadic desire and sexual functioning (P = .003 for FSFI, P = .002 for FSDS-R, and P = .003 for FSDQ).
Participants also experienced a decrease in depression (P < .0001) and menopausal symptoms (P = .001) and an increase in body-image satisfaction (P = .018) and relationship satisfaction (P = .0011) after the CBT sessions. The researchers assessed participants’ satisfaction with the Client Satisfaction Questionnaire after the CBT sessions and reported some of the qualitative findings.
“The treatment program was able to assist me with recognizing that some of my sexual concerns were normal, emotional as well as physical and hormonal, and provided me the ability to delve more deeply into the psychology of menopause and how to work through symptoms and concerns in more manageable pieces,” one participant wrote. Another found helpful the “homework exercises of recognizing a thought/feeling/emotion surrounding how I feel about myself/body and working through. More positive thought pattern/restructuring a response the most helpful.”
The main complaint about the program was that it was too short, with women wanting more sessions to help continue their progress.
Not an ‘either-or’ approach
Dr. Kingsberg said ISSWSH has a variety of sexual medicine practitioners, including providers who can provide CBT for sexual concerns, and the American Association of Sexuality Educators, Counselors and Therapists has a referral directory.
“Keeping in mind the bio-psychosocial model, sometimes psychotherapy is going to be a really effective treatment for sexual concerns,” Dr. Kingsberg said. “Sometimes the pharmacologic option is going to be a really effective treatment for some concerns, and sometimes the combination is going to have a really nice treatment effect. So it’s not a one-size-fits-all, and it doesn’t have to be an either-or.”
The sexual concerns of women still do not get adequately addressed in medical schools and residencies, Dr. Kingsberg said, which is distinctly different from how male sexual concerns are addressed in health care.
“Erectile dysfunction is kind of in the norm, and women are still a little hesitant to bring up their sexual concerns,” Dr. Kingsberg said. “They don’t know if it’s appropriate and they’re hoping that their clinician will ask.”
One way clinicians can do that is with a global question for all their patients: “Most of my patients have sexual questions or concerns; what concerns do you have?”
“They don’t have to go through a checklist of 10 things,” Dr. Kingsberg said. If the patient does not bring anything up, providers can then ask a single follow up question: “Do you have any concerns with desire, arousal, orgasm, or pain?” That question, Dr. Kingsberg said, covers the four main areas of concern.
The study was funded by the Canadian Institute of Health Research. Dr. Green reported no disclosures. Dr. Kingsberg has consulted for or served on the advisory board for Alloy, Astellas, Bayer, Dare Bioscience, Freya, Reunion Neuroscience, Materna Medical, Madorra, Palatin, Pfizer, ReJoy, Sprout, Strategic Science Technologies, and MsMedicine.
AT NAMS 2023
Neuropsychiatric aspects of Parkinson’s disease: Practical considerations
Parkinson’s disease (PD) is a neurodegenerative condition diagnosed pathologically by alpha synuclein–containing Lewy bodies and dopaminergic cell loss in the substantia nigra pars compacta of the midbrain. Loss of dopaminergic input to the caudate and putamen disrupts the direct and indirect basal ganglia pathways for motor control and contributes to the motor symptoms of PD.1 According to the Movement Disorder Society criteria, PD is diagnosed clinically by bradykinesia (slowness of movement) plus resting tremor and/or rigidity in the presence of supportive criteria, such as levodopa responsiveness and hyposmia, and in the absence of exclusion criteria and red flags that would suggest atypical parkinsonism or an alternative diagnosis.2
Although the diagnosis and treatment of PD focus heavily on the motor symptoms, nonmotor symptoms can arise decades before the onset of motor symptoms and continue throughout the lifespan. Nonmotor symptoms affect patients from head (ie, cognition and mood) to toe (ie, striatal toe pain) and multiple organ systems in between, including the olfactory, integumentary, cardiovascular, gastrointestinal, genitourinary, and autonomic nervous systems. Thus, it is not surprising that nonmotor symptoms of PD impact health-related quality of life more substantially than motor symptoms.3 A helpful analogy is to consider the motor symptoms of PD as the tip of the iceberg and the nonmotor symptoms as the larger, submerged portions of the iceberg.4
Nonmotor symptoms can negatively impact the treatment of motor symptoms. For example, imagine a patient who is very rigid and dyscoordinated in the arms and legs, which limits their ability to dress and walk. If this patient also suffers from nonmotor symptoms of orthostatic hypotension and psychosis—both of which can be exacerbated by levodopa—dose escalation of levodopa for the rigidity and dyscoordination could be compromised, rendering the patient undertreated and less mobile.
In this review, we focus on identifying and managing nonmotor symptoms of PD that are relevant to psychiatric practice, including mood and motivational disorders, anxiety disorders, psychosis, cognitive disorders, and disorders related to the pharmacologic and surgical treatment of PD (Figure 1).
Mood and motivational disorders
Depression
Depression is a common symptom in PD that can occur in the prodromal period years to decades before the onset of motor symptoms, as well as throughout the disease course.5 The prevalence of depression in PD varies from 3% to 90%, depending on the methods of assessment, clinical setting of assessment, motor symptom severity, and other factors; clinically significant depression likely affects approximately 35% to 38% of patients.5,6 How depression in patients with PD differs from depression in the general population is not entirely understood, but there does seem to be less guilt and suicidal ideation and a substantial component of negative affect, including dysphoria and anxiety.7 Practically speaking, depression is treated similarly in PD and general populations, with a few considerations.
Despite limited randomized controlled trials (RCTs) for efficacy specifically in patients with PD, selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are generally considered first-line treatments. There is also evidence for tricyclic antidepressants (TCAs), but due to potential worsening of orthostatic hypotension and cognition, TCAs may not be a favorable option for certain patients with PD.8,9 All antidepressants have the potential to worsen tremor. Theoretically, SNRIs, with noradrenergic activity, may be less tolerable than SSRIs in patients with PD. However, worsening tremor generally has not been a clinically significant adverse event reported in PD depression clinical trials, although it was seen in 17% of patients receiving paroxetine and 21% of patients receiving venlafaxine compared to 7% of patients receiving placebo.9-11 If tremor worsens, mirtazapine could be considered because it has been reported to cause less tremor than SSRIs or TCAs.12
Among medications for PD, pramipexole, a dopamine agonist, may have a beneficial effect on depression.13 Additionally, some evidence supports rasagiline, a monoamine oxidase type B inhibitor, as an adjunctive medication for depression in PD.14 Nevertheless, antidepressant medications remain the standard pharmacologic treatment for PD depression.
Continue to: In terms of nonpharmacologic options...
In terms of nonpharmacologic options, cognitive-behavioral therapy (CBT) is likely efficacious, exercise (especially yoga) is likely efficacious, and repetitive transcranial magnetic stimulation may be efficacious.15,16 While further high-quality trials are needed, these treatments are low-risk and can be considered, especially for patients who cannot tolerate medications.
Apathy
Apathy—a loss of motivation and goal-directed behavior—can occur in up to 30% of patients during the prodromal period of PD, and in up to 70% of patients throughout the disease course.17 Apathy can coexist with depression, which can make apathy difficult to diagnose.17 Given the time constraints of a clinic visit, a practical approach would be to first screen for depression and cognitive impairment. If there is continued suspicion of apathy, the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale part I question (“In the past week have you felt indifferent to doing activities or being with people?”) can be used to screen for apathy, and more detailed scales, such as the Apathy Scale (AS) or Lille Apathy Rating Scale (LARS), could be used if indicated.18
There are limited high-quality positive trials of apathy-specific treatments in PD. In an RCT of patients with PD who did not have depression or dementia, rivastigmine improved LARS scores compared to placebo.15 Piribedil, a D2/D3 receptor agonist, improved apathy in patients who underwent subthalamic nucleus deep brain stimulation (STN DBS).15 Exercise such as individualized physical therapy programs, dance, and Nordic walking as well as mindfulness interventions were shown to significantly reduce apathy scale scores.19 SSRIs, SNRIs, and rotigotine showed a trend toward reducing AS scores in RCTs.10,20
Larger, high-quality studies are needed to clarify the treatment of apathy in PD. In the meantime, a reasonable approach is to first treat any comorbid psychiatric or cognitive disorders, since apathy can be associated with these conditions, and to optimize antiparkinsonian medications for motor symptoms, motor fluctuations, and nonmotor fluctuations. Then, the investigational apathy treatments described in this section could be considered on an individual basis.
Anxiety disorders
Anxiety is seen throughout the disease course of PD in approximately 30% to 50% of patients.21 It can manifest as generalized anxiety disorder, panic disorder, and other anxiety disorders. There are no high-quality RCTs of pharmacologic treatments of anxiety specifically in patients with PD, except for a negative safety and tolerability study of buspirone in which one-half of patients experienced worsening motor symptoms.15,22 Thus, the treatment of anxiety in patients with PD is similar to treatments in the general population. SSRIs and SNRIs are typically considered first-line, benzodiazepines are sometimes used with caution (although cognitive adverse effects and fall risk need to be considered), and nonpharmacologic treatments such as mindfulness yoga, exercise, CBT, and psychotherapy can be effective.16,21,23
Continue to: Because there is the lack...
Because there is the lack of evidence-based treatments for anxiety in PD, we highlight 2 PD-specific anxiety disorders: internal tremor, and nonmotor “off” anxiety.
Internal tremor
Internal tremor is a sense of vibration in the axial and/or appendicular muscles that cannot be seen externally by the patient or examiner. It is not yet fully understood if this phenomenon is sensory, anxiety-related, related to subclinical tremor, or the result of a combination of these factors (ie, sensory awareness of a subclinical tremor that triggers or is worsened by anxiety). There is some evidence for subclinical tremor on electromyography, but internal tremor does not respond to antiparkinsonian medications in 70% of patients.24 More electrophysiological research is needed to clarify this phenomenon. Internal tremor has been associated with anxiety in 64% of patients and often improves with anxiolytic therapies.24
Although poorly understood, internal tremor is a documented phenomenon in 33% to 44% of patients with PD, and in some cases, it may be an initial symptom that motivates a patient to seek medical attention for the first time.24,25 Internal tremor has also been reported in patients with essential tremor and multiple sclerosis.25 Therefore, physicians should be aware of internal tremor because this symptom could herald an underlying neurological disease.
Nonmotor ‘off’ anxiety
Patients with PD are commonly prescribed carbidopa-levodopa, a dopamine precursor, at least 3 times daily. Initially, this medication controls motor symptoms well from 1 dose to the next. However, as the disease progresses, some patients report motor fluctuations in which an individual dose of carbidopa-levodopa may wear off early, take longer than usual to take effect, or not take effect at all. Patients describe these periods as an “off” state in which they do not feel their medications are working. Such motor fluctuations can lead to anxiety and avoidance behaviors, because patients fear being in public at times when the medication does not adequately control their motor symptoms.
In addition to these motor symptom fluctuations and related anxiety, patients can also experience nonmotor symptom fluctuations. A wide variety of nonmotor symptoms, such as mood, cognitive, and behavioral symptoms, have been reported to fluctuate in parallel with motor symptoms.26,27 One study reported fluctuating restlessness in 39% of patients with PD, excessive worry in 17%, shortness of breath in 13%, excessive sweating and fear in 12%, and palpitations in 10%.27 A patient with fluctuating shortness of breath, sweating, and palpitations (for example) may repeatedly present to the emergency department with a negative cardiac workup and eventually be diagnosed with panic disorder, whereas the patient is truly experiencing nonmotor “off” symptoms. Thus, it is important to be aware of nonmotor fluctuations so this diagnosis can be made and the symptoms appropriately treated. The first step in treating nonmotor fluctuations is to optimize the antiparkinsonian regimen to minimize fluctuations. If “off” anxiety symptoms persist, anxiolytic medications can be prescribed.21
Continue to: Psychosis
Psychosis
Psychosis can occur in prodromal and early PD but is most common in advanced PD.28 One study reported that 60% of patients developed hallucinations or delusions after 12 years of follow-up.29 Disease duration, disease severity, dementia, and rapid eye movement sleep behavior disorder are significant risk factors for psychosis in PD.30 Well-formed visual hallucinations are the most common manifestation of psychosis in patients with PD. Auditory hallucinations and delusions are less common. Delusions are usually seen in patients with dementia and are often paranoid delusions, such as of spousal infidelity.30 Sensory hallucinations can occur, but should not be mistaken with formication, a central pain syndrome in PD that can represent a nonmotor “off” symptom that may respond to dopaminergic medication.31 Other more mild psychotic symptoms include illusions or misinterpretation of stimuli, false sense of presence, and passage hallucinations of fleeting figures in the peripheral vision.30
The pathophysiology of PD psychosis is not entirely understood but differs from psychosis in other disorders. It can occur in the absence of antiparkinsonian medication exposure and is thought to be a consequence of the underlying disease process of PD involving neurodegeneration in certain brain regions and aberrant neurotransmission of not only dopamine but also serotonin, acetylcholine, and glutamate.30
Figure 2 outlines the management of psychosis in PD. After addressing medical and medication-related causes, it is important to determine if the psychotic symptom is sufficiently bothersome to and/or potentially dangerous for the patient to warrant treatment. If treatment is indicated, pimavanserin and clozapine are efficacious for psychosis in PD without worsening motor symptoms, and quetiapine is possibly efficacious with a low risk of worsening motor symptoms.15 Other antipsychotics, such as olanzapine, risperidone, and haloperidol, can substantially worsen motor symptoms.15 Both second-generation antipsychotics and pimavanserin have an FDA black-box warning for a higher risk of all-cause mortality in older patients with dementia; however, because psychosis is associated with early mortality in PD, the risk/benefit ratio should be discussed with the patient and family for shared decision-making.30 If the patient also has dementia, rivastigmine—which is FDA-approved for PD dementia (PDD)—may also improve hallucinations.32
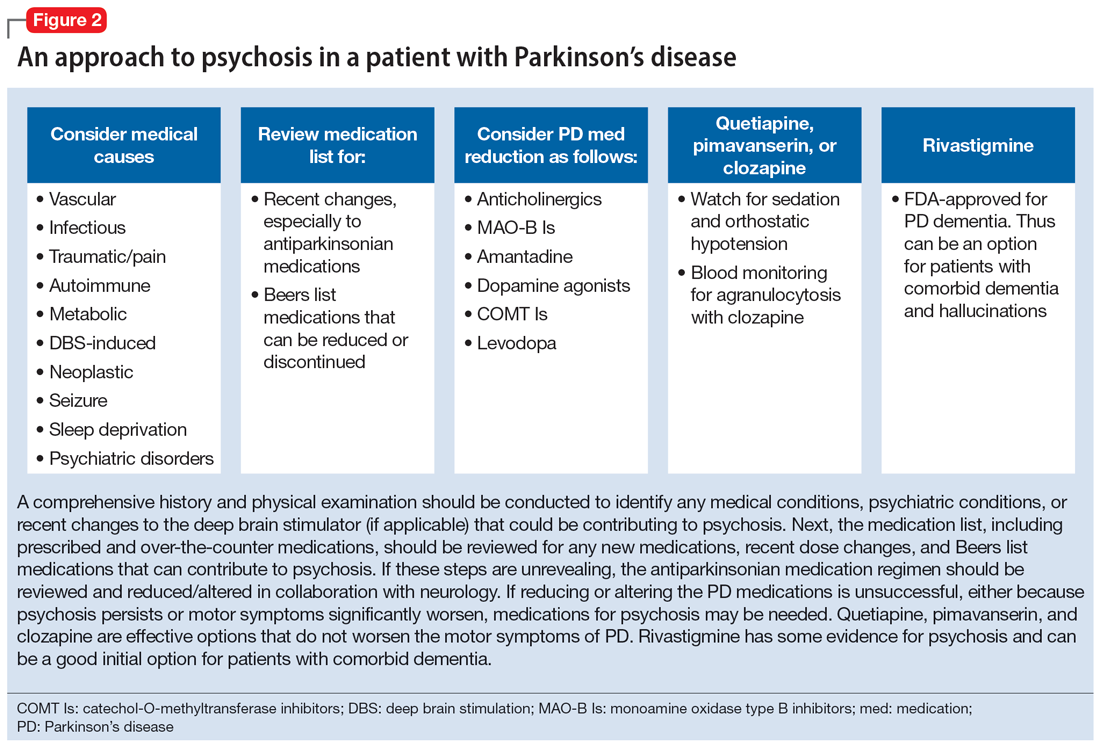
Cognitive disorders
This section focuses on PD mild cognitive impairment (PD-MCI) and PDD. When a patient with PD reports cognitive concerns, the approach outlined in Figure 3 can be used to diagnose the cognitive disorder. A detailed history, medication review, and physical examination can identify any medical or psychiatric conditions that could affect cognition. The American Academy of Neurology recommends screening for depression, obtaining blood levels of vitamin B12 and thyroid-stimulating hormone, and obtaining a CT or MRI of the brain to rule out reversible causes of dementia.33 A validated screening test such as the Montreal Cognitive Assessment, which has higher sensitivity for PD-MCI than the Mini-Mental State Examination, is used to identify and quantify cognitive impairment.34 Neuropsychological testing is the gold standard and can be used to confirm and/or better quantify the degree and domains of cognitive impairment.35 Typically, cognitive deficits in PD affect executive function, attention, and/or visuospatial domains more than memory and language early on, and deficits in visuospatial and language domains have the highest sensitivity for predicting progression to PDD.36
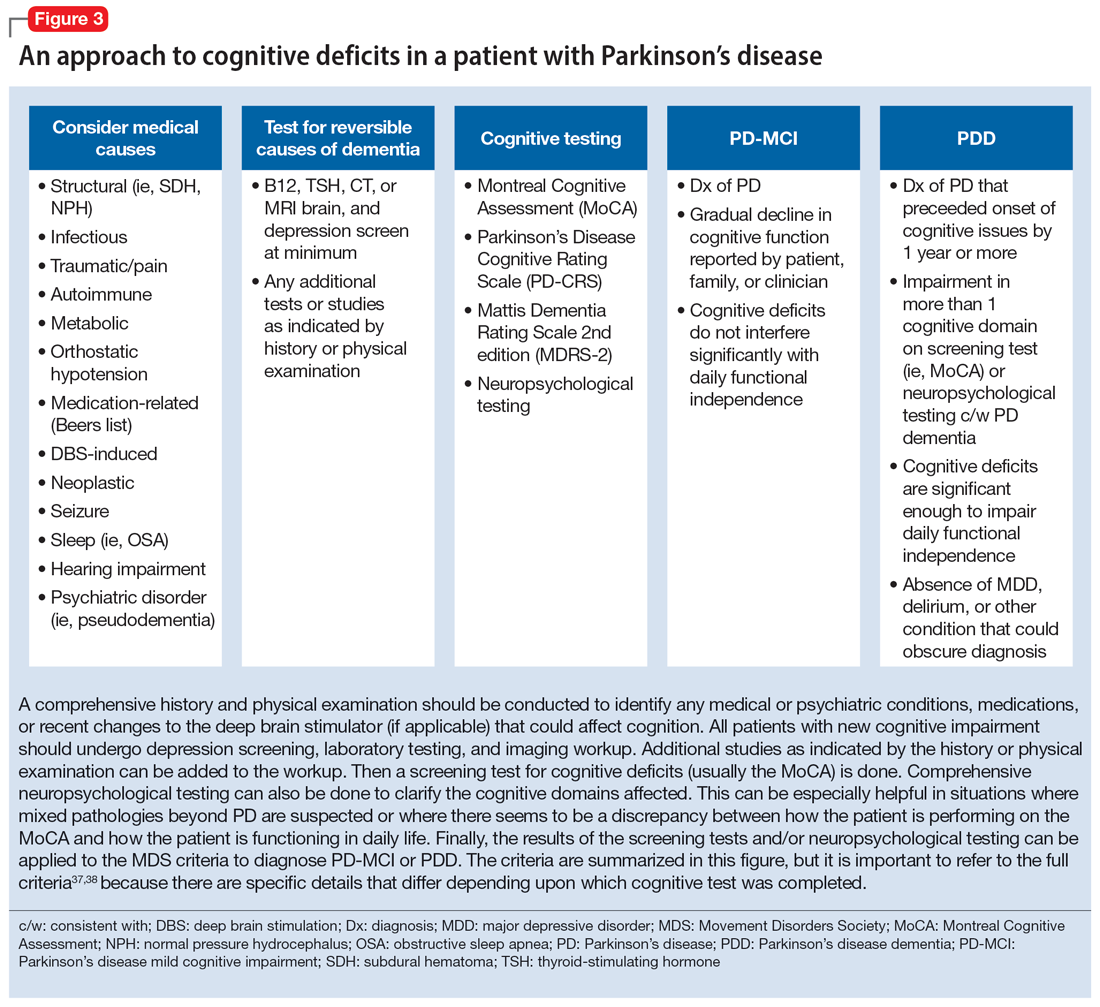
Once reversible causes of dementia are addressed or ruled out and cognitive testing is completed, the Movement Disorder Society (MDS) criteria for PD-MCI and PDD summarized in Figure 3 can be used to diagnose the cognitive disorder.37,38 The MDS criteria for PDD require a diagnosis of PD for ≥1 year prior to the onset of dementia to differentiate PDD from dementia with Lewy bodies (DLB). If the dementia starts within 1 year of the onset of parkinsonism, the diagnosis would be DLB. PDD and DLB are on the spectrum of Lewy body dementia, with the same Lewy body pathology in different temporal and spatial distributions in the brain.38
Continue to: PD-MCI is present in...
PD-MCI is present in approximately 25% of patients.35 PD-MCI does not always progress to dementia but increases the risk of dementia 6-fold. The prevalence of PDD increases with disease duration; it is present in approximately 50% of patients at 10 years and 80% of patients at 20 years of disease.35 Rivastigmine is the only FDA-approved medication to slow progression of PDD. There is insufficient evidence for other acetylcholinesterase inhibitors and memantine.15 Unfortunately, RCTs of pharmacotherapy for PD-MCI have failed to show efficacy. However, exercise, cognitive rehabilitation, and neuromodulation are being studied. In the meantime, addressing modifiable risk factors (such as vascular risk factors and alcohol consumption) and treating comorbid orthostatic hypotension, obstructive sleep apnea, and depression may improve cognition.35,39
Treatment-related disorders
Impulse control disorders
Impulse control disorders (ICDs) are an important medication-related consideration in patients with PD. The ICDs seen in PD include pathological gambling, binge eating, excessive shopping, hypersexual behaviors, and dopamine dysregulation syndrome (Table). These disorders are more common in younger patients with a history of impulsive personality traits and addictive behaviors (eg, history of tobacco or alcohol abuse), and are most strongly associated with dopaminergic therapies, particularly the dopamine agonists.40,41 In the DOMINION study, the odds of ICDs were 2- to 3.5-fold higher in patients taking dopamine agonists.42 This is mainly thought to be due to stimulation of D2/D3 receptors in the mesolimbic system.40 High doses of levodopa, monoamine oxidase inhibitors, and amantadine are also associated with ICDs.40-42
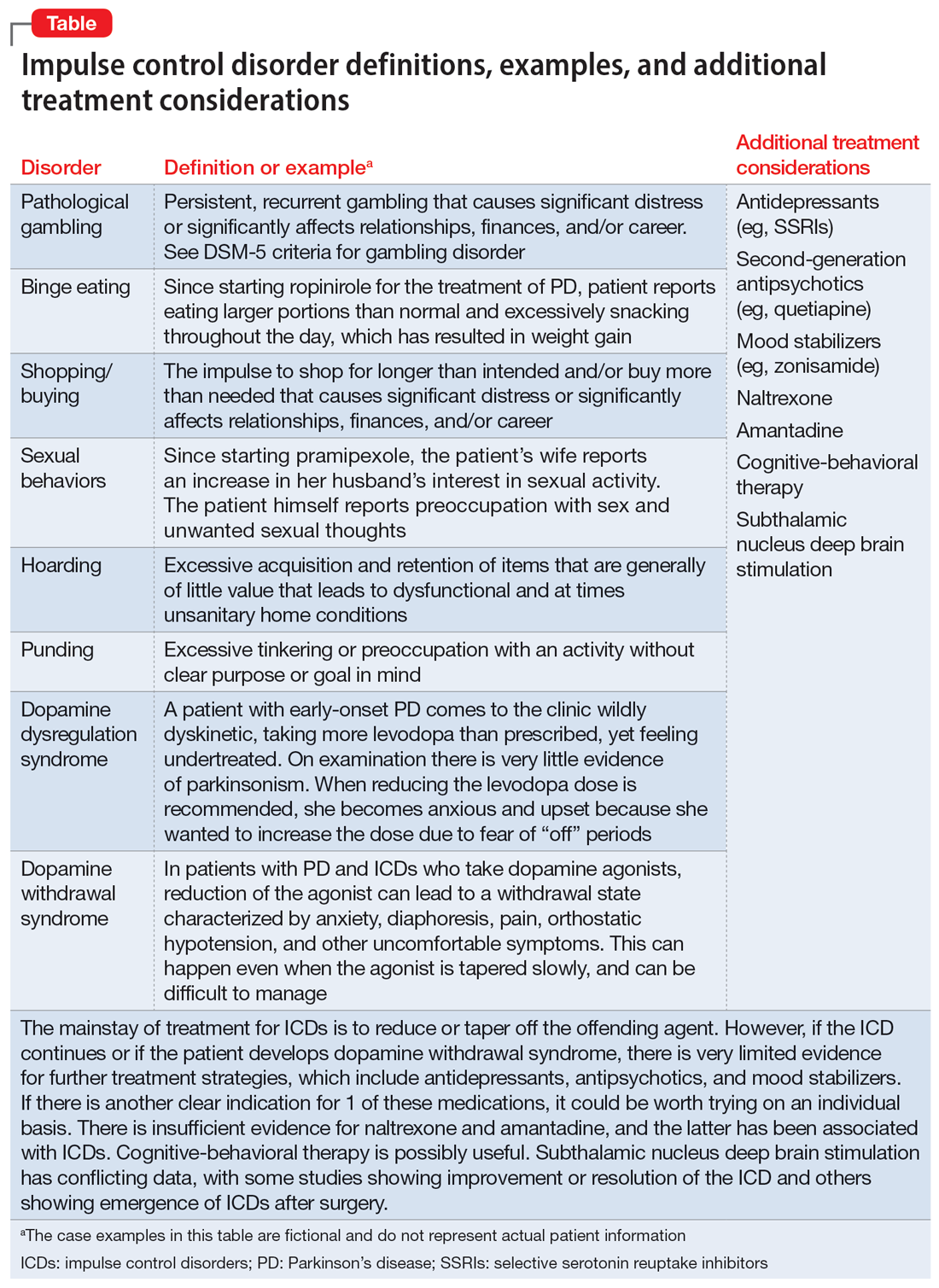
The first step in managing ICDs is diagnosing them, which can be difficult because patients often are not forthcoming about these problems due to embarrassment or failure to recognize that the ICD is related to PD medications. If a family member accompanies the patient at the visit, the patient may not want to disclose the amount of money they spend or the extent to which the behavior is a problem. Thus, a screening questionnaire, such as the Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease (QUIP) can be a helpful way for patients to alert the clinician to the issue.41 Education for the patient and family is crucial before the ICD causes significant financial, health, or relationship problems.
The mainstay of treatment is to reduce or taper off the dopamine agonist or other offending agent while monitoring for worsening motor symptoms and dopamine withdrawal syndrome. If this is unsuccessful, there is very limited evidence for further treatment strategies (Table), including antidepressants, antipsychotics, and mood stabilizers.40,43,44 There is insufficient evidence for naltrexone based on an RCT that failed to meet its primary endpoint, although naltrexone did significantly reduce QUIP scores.15,44 There is also insufficient evidence for amantadine, which showed benefit in some studies but was associated with ICDs in the DOMINION study.15,40,42 In terms of nonpharmacologic treatments, CBT is likely efficacious.15,40 There are mixed results for STN DBS. Some studies showed improvement in the ICD, due at least in part to dopaminergic medication reduction postoperatively, but this treatment has also been reported to increase impulsivity.40,45
Deep brain stimulation–related disorders
For patients with PD, the ideal lead location for STN DBS is the dorsolateral aspect of the STN, as this is the motor region of the nucleus. The STN functions in indirect and hyperdirect pathways to put the brake on certain motor programs so only the desired movement can be executed. Its function is clinically demonstrated by patients with STN stroke who develop excessive ballistic movements. Adjacent to the motor region of the STN is a centrally located associative region and a medially located limbic region. Thus, when stimulating the dorsolateral STN, current can spread to those regions as well, and the STN’s ability to put the brake on behavioral and emotional programs can be affected.46 Stimulation of the STN has been associated with mania, euphoria, new-onset ICDs, decreased verbal fluency, and executive dysfunction. Depression, apathy, and anxiety can also occur, but more commonly result from rapid withdrawal of antiparkinsonian medications after DBS surgery.46,47 Therefore, for PD patients with DBS with new or worsening psychiatric or cognitive symptoms, it is important to inquire about any recent programming sessions with neurology as well as recent self-increases in stimulation by the patient using their controller. Collaboration with neurology is important to troubleshoot whether stimulation could be contributing to the patient’s psychiatric or cognitive symptoms.
Continue to: Bottom Line
Bottom Line
Mood, anxiety, psychotic, and cognitive symptoms and disorders are common psychiatric manifestations associated with Parkinson’s disease (PD). In addition, patients with PD may experience impulsive control disorders and other symptoms related to treatments they receive for PD. Careful assessment and collaboration with neurology is crucial to alleviating the effects of these conditions.
Related Resources
- Weintraub D, Aarsland D, Chaudhuri KR, et al. The neuropsychiatry of Parkinson’s disease: advances and challenges. Lancet Neurology. 2022;21(1):89-102. doi:10.1016/S1474-4422(21)00330-6
- Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurologic Clinics. 2020;38(2):269-292. doi:10.1016/j.ncl.2019.12.003
- Castrioto A, Lhommee E, Moro E et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurology. 2014;13(3):287-305. doi:10.1016/ S1474-4422(13)70294-1
Drug Brand Names
Amantadine • Gocovri
Carbidopa-levodopa • Sinemet
Clozapine • Clozaril
Haloperidol • Haldol
Memantine • Namenda
Mirtazapine • Remeron
Naltrexone • Vivitrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimavanserin • Nuplazid
Piribedil • Pronoran
Pramipexole • Mirapex
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neupro
Venlafaxine • Effexor
Zonisamide • Zonegran
1. Bloem BR, Okun MS, Klein C. Parkinson’s disease. Lancet Neurology. 2021;397(10291):2284-2303.
2. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Movement Disorders. 2015;30(12):1591-1601.
3. Martinez-Martin P, Rodriguez-Blazquez C, Kurtiz MM, et al. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord. 2011;26(3):399-406.
4. Langston WJ. The Parkinson’s complex: parkinsonism is just the tip of the iceberg. Ann Neurol. 2006;59(4):591-596.
5. Cong S, Xiang C, Zhang S, et al. Prevalence and clinical aspects of depression in Parkinson’s disease: a systematic review and meta‑analysis of 129 studies. Neurosci Biobehav Rev. 2022;141:104749. doi:10.1016/j.neubiorev.2022.104749
6. Reijnders JS, Ehrt U, Weber WE, et al. A systematic review of prevalence studies in depression in Parkinson’s disease. Mov Disord. 2008;23(2):183-189.
7. Zahodne LB, Marsiske M, Okun MS, et al. Components of depression in Parkinson disease. J Geriatr Psychiatry Neurol. 2012;25(3):131-137.
8. Skapinakis P, Bakola E, Salanti G, et al. Efficacy and acceptability of selective serotonin reuptake inhibitors for the treatment of depression in Parkinson’s disease: a systematic review and meta-analysis of randomized controlled trials. BMC Neurology. 2010;10:49. doi:10.1186/1471-2377-10-49
9. Richard IH, McDermott MP, Kurlan R, et al; SAD-PD Study Group. A randomized, double-blind placebo-controlled trial of antidepressants in Parkinson’s disease. Neurology. 2012;78(16):1229-1236.
10. Takahashi M, Tabu H, Ozaki A, et al. Antidepressants for depression, apathy, and gait instability in Parkinson’s disease: a multicenter randomized study. Intern Med. 2019;58(3):361-368.
11. Bonuccelli U, Mecco G, Fabrini G, et al. A non-comparative assessment of tolerability and efficacy of duloxetine in the treatment of depressed patients with Parkinson’s disease. Expert Opin Pharmacother. 2012;13(16):2269-2280.
12. Wantanabe N, Omorio IM, Nakagawa A, et al; MANGA (Meta-Analysis of New Generation Antidepressants) Study Group. Safety reporting and adverse-event profile of mirtazapine described in randomized controlled trials in comparison with other classes of antidepressants in the acute-phase treatment of adults with depression. CNS Drugs. 2010;24(1):35-53.
13. Barone P, Scarzella L, Marconi R, et al; Depression/Parkinson Italian Study Group. Pramipexole versus sertraline in the treatment of depression in Parkinson’s disease: a national multicenter parallel-group randomized study. J Neurol. 2006;253(5):601-607.
14. Smith KM, Eyal E, Weintraub D, et al; ADAGIO Investigators. Combined rasagiline and anti-depressant use in Parkinson’s disease in the ADAGIO study: effects on non-motor symptoms and tolerability. JAMA Neurology. 2015;72(1):88-95.
15. Seppi K, Chaudhuri R, Coelho M, et al; the collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine Committee. Update on treatments for nonmotor symptoms of Parkinson’s disease--an evidence-based medicine review. Mov Disord. 2019;34(2):180-198.
16. Kwok JYY, Kwan JCY, Auyeung M, et al. Effects of mindfulness yoga vs stretching and resistance training exercises on anxiety and depression for people with Parkinson disease: a randomized clinical trial. JAMA Neurol. 2019;76(7):755-763.
17. De Waele S, Cras P, Crosiers D. Apathy in Parkinson’s disease: defining the Park apathy subtype. Brain Sci. 2022;12(7):923.
18. Mele B, Van S, Holroyd-Leduc J, et al. Diagnosis, treatment and management of apathy in Parkinson’s disease: a scoping review. BMJ Open. 2020;10(9):037632. doi:10.1136/bmjopen-2020-037632
19. Mele B, Ismail Z, Goodarzi Z, et al. Non-pharmacological interventions to treat apathy in Parkinson’s disease: a realist review. Clin Park Relat Disord. 2021;4:100096. doi:10.1016/j.prdoa.2021.100096
20. Chung SJ, Asgharnejad M, Bauer L, et al. Evaluation of rotigotine transdermal patch for the treatment of depressive symptoms in patients with Parkinson’s disease. Expert Opin Pharmacother. 2016;(17)11:1453-1461.
21. Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurol Clin. 2020;38(2):269-292.
22. Schneider RB, Auinger P, Tarolli CG, et al. A trial of buspirone for anxiety in Parkinson’s disease: safety and tolerability. Parkinsonism Relat Disord. 2020;81:69-74.
23. Moonen AJH, Mulders AEP, Defebvre L, et al. Cognitive behavioral therapy for anxiety in Parkinson’s disease: a randomized controlled trial. Mov Disord. 2021;36(11):2539-2548.
24. Shulman LM, Singer C, Bean JA, et al. Internal tremor in patient with Parkinson’s disease. Mov Disord. 1996;11(1):3-7.
25. Cochrane GD, Rizvi S, Abrantes A, et al. Internal tremor in Parkinson’s disease, multiple sclerosis, and essential tremor. Parkinsonism Relat Disord. 2015;21(10):1145-1147.
26. Del Prete E, Schmitt E, Meoni S, et al. Do neuropsychiatric fluctuations temporally match motor fluctuations in Parkinson’s disease? Neurol Sci. 2022;43(6):3641-3647.
27. Kleiner G, Fernandez HH, Chou KL, et al. Non-motor fluctuations in Parkinson’s disease: validation of the non-motor fluctuation assessment questionnaire. Mov Disord. 2021;36(6):1392-1400.
28. Pachi I, Maraki MI, Giagkou N, et al. Late life psychotic features in prodromal Parkinson’s disease. Parkinsonism Relat Disord. 2021;86:67-73.
29. Forsaa EB, Larsen JP, Wentzel-Larsen T, et al. A 12-year population-based study of psychosis in Parkinson’s disease. Arch Neurol. 2010;67(8):996-1001.
30. Chang A, Fox SH. Psychosis in Parkinson’s disease: epidemiology, pathophysiology, and management. Drugs. 2016;76(11):1093-1118.
31. Kasunich A, Kilbane C, Wiggins R. Movement disorders moment: pain and palliative care in movement disorders. Practical Neurology. 2021;20(4):63-67.
32. Burn D, Emre M, McKeith I, et al. Effects of rivastigmine in patients with and without visual hallucinations in dementia associated with Parkinson’s disease. Mov Disord. 2006;21(11):1899-1907.
33. Tripathi M, Vibha D. Reversible dementias. Indian J Psychiatry. 2009; 51 Suppl 1(Suppl 1): S52-S55.
34. Dalrymple-Alford JC, MacAskill MR, Nakas CT, et al. The MoCA: well-suited screen for cognitive impairment in Parkinson disease. Neurology. 2010;75(19):1717-1725.
35. Goldman J, Sieg, E. Cognitive impairment and dementia in Parkinson disease. Clin Geriatr Med. 2020;36(2):365-377.
36. Gonzalez-Latapi P, Bayram E, Litvan I, et al. Cognitive impairment in Parkinson’s disease: epidemiology, clinical profile, protective and risk factors. Behav Sci (Basel). 2021;11(5):74.
37. Litvan I, Goldman JG, Tröster AI, et al. Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society Task Force Guidelines. Mov Disord. 2012;27(3):349-356.
38. Dubois B, Burn D, Goetz C, et al. Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord. 2007;22(16):2314-2324.
39. Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers. 2021;7(1):47. doi:10.1038/s41572-021-00280-3
40. Weintraub D, Claassen DO. Impulse control and related disorders in Parkinson’s disease. Int Rev Neurobiol. 2017;133:679-717.
41. Vilas D, Pont-Sunyer C, Tolosa E. Impulse control disorders in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18 Suppl 1:S80-S84.
42. Weintraub D, Koester J, Potenza MN, et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch Neurol. 2010;67(5):589-595.
43. Faouzi J, Corvol JC, Mariani LL. Impulse control disorders and related behaviors in Parkinson’s disease: risk factors, clinical and genetic aspects, and management. Curr Opin Neurol. 2021;34(4):547-555.
44. Samuel M, Rodriguez-Oroz M, Antonini A, et al. Impulse control disorders in Parkinson’s disease: management, controversies, and potential approaches. Mov Disord. 2015;30(2):150-159.
45. Frank MJ, Samanta J, Moustafa AA, et al. Hold your horses: impulsivity, deep brain stimulation and medication in Parkinsonism. Science. 2007;318(5854):1309-1312.
46. Jahanshahi M, Obeso I, Baunez C, et al. Parkinson’s disease, the subthalamic nucleus, inhibition, and impulsivity. Mov Disord. 2015;30(2):128-140.
47. Castrioto A, Lhommée E, Moro E, et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurol. 2014;13(3):287-305.
Parkinson’s disease (PD) is a neurodegenerative condition diagnosed pathologically by alpha synuclein–containing Lewy bodies and dopaminergic cell loss in the substantia nigra pars compacta of the midbrain. Loss of dopaminergic input to the caudate and putamen disrupts the direct and indirect basal ganglia pathways for motor control and contributes to the motor symptoms of PD.1 According to the Movement Disorder Society criteria, PD is diagnosed clinically by bradykinesia (slowness of movement) plus resting tremor and/or rigidity in the presence of supportive criteria, such as levodopa responsiveness and hyposmia, and in the absence of exclusion criteria and red flags that would suggest atypical parkinsonism or an alternative diagnosis.2
Although the diagnosis and treatment of PD focus heavily on the motor symptoms, nonmotor symptoms can arise decades before the onset of motor symptoms and continue throughout the lifespan. Nonmotor symptoms affect patients from head (ie, cognition and mood) to toe (ie, striatal toe pain) and multiple organ systems in between, including the olfactory, integumentary, cardiovascular, gastrointestinal, genitourinary, and autonomic nervous systems. Thus, it is not surprising that nonmotor symptoms of PD impact health-related quality of life more substantially than motor symptoms.3 A helpful analogy is to consider the motor symptoms of PD as the tip of the iceberg and the nonmotor symptoms as the larger, submerged portions of the iceberg.4
Nonmotor symptoms can negatively impact the treatment of motor symptoms. For example, imagine a patient who is very rigid and dyscoordinated in the arms and legs, which limits their ability to dress and walk. If this patient also suffers from nonmotor symptoms of orthostatic hypotension and psychosis—both of which can be exacerbated by levodopa—dose escalation of levodopa for the rigidity and dyscoordination could be compromised, rendering the patient undertreated and less mobile.
In this review, we focus on identifying and managing nonmotor symptoms of PD that are relevant to psychiatric practice, including mood and motivational disorders, anxiety disorders, psychosis, cognitive disorders, and disorders related to the pharmacologic and surgical treatment of PD (Figure 1).

Mood and motivational disorders
Depression
Depression is a common symptom in PD that can occur in the prodromal period years to decades before the onset of motor symptoms, as well as throughout the disease course.5 The prevalence of depression in PD varies from 3% to 90%, depending on the methods of assessment, clinical setting of assessment, motor symptom severity, and other factors; clinically significant depression likely affects approximately 35% to 38% of patients.5,6 How depression in patients with PD differs from depression in the general population is not entirely understood, but there does seem to be less guilt and suicidal ideation and a substantial component of negative affect, including dysphoria and anxiety.7 Practically speaking, depression is treated similarly in PD and general populations, with a few considerations.
Despite limited randomized controlled trials (RCTs) for efficacy specifically in patients with PD, selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are generally considered first-line treatments. There is also evidence for tricyclic antidepressants (TCAs), but due to potential worsening of orthostatic hypotension and cognition, TCAs may not be a favorable option for certain patients with PD.8,9 All antidepressants have the potential to worsen tremor. Theoretically, SNRIs, with noradrenergic activity, may be less tolerable than SSRIs in patients with PD. However, worsening tremor generally has not been a clinically significant adverse event reported in PD depression clinical trials, although it was seen in 17% of patients receiving paroxetine and 21% of patients receiving venlafaxine compared to 7% of patients receiving placebo.9-11 If tremor worsens, mirtazapine could be considered because it has been reported to cause less tremor than SSRIs or TCAs.12
Among medications for PD, pramipexole, a dopamine agonist, may have a beneficial effect on depression.13 Additionally, some evidence supports rasagiline, a monoamine oxidase type B inhibitor, as an adjunctive medication for depression in PD.14 Nevertheless, antidepressant medications remain the standard pharmacologic treatment for PD depression.
Continue to: In terms of nonpharmacologic options...
In terms of nonpharmacologic options, cognitive-behavioral therapy (CBT) is likely efficacious, exercise (especially yoga) is likely efficacious, and repetitive transcranial magnetic stimulation may be efficacious.15,16 While further high-quality trials are needed, these treatments are low-risk and can be considered, especially for patients who cannot tolerate medications.
Apathy
Apathy—a loss of motivation and goal-directed behavior—can occur in up to 30% of patients during the prodromal period of PD, and in up to 70% of patients throughout the disease course.17 Apathy can coexist with depression, which can make apathy difficult to diagnose.17 Given the time constraints of a clinic visit, a practical approach would be to first screen for depression and cognitive impairment. If there is continued suspicion of apathy, the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale part I question (“In the past week have you felt indifferent to doing activities or being with people?”) can be used to screen for apathy, and more detailed scales, such as the Apathy Scale (AS) or Lille Apathy Rating Scale (LARS), could be used if indicated.18
There are limited high-quality positive trials of apathy-specific treatments in PD. In an RCT of patients with PD who did not have depression or dementia, rivastigmine improved LARS scores compared to placebo.15 Piribedil, a D2/D3 receptor agonist, improved apathy in patients who underwent subthalamic nucleus deep brain stimulation (STN DBS).15 Exercise such as individualized physical therapy programs, dance, and Nordic walking as well as mindfulness interventions were shown to significantly reduce apathy scale scores.19 SSRIs, SNRIs, and rotigotine showed a trend toward reducing AS scores in RCTs.10,20
Larger, high-quality studies are needed to clarify the treatment of apathy in PD. In the meantime, a reasonable approach is to first treat any comorbid psychiatric or cognitive disorders, since apathy can be associated with these conditions, and to optimize antiparkinsonian medications for motor symptoms, motor fluctuations, and nonmotor fluctuations. Then, the investigational apathy treatments described in this section could be considered on an individual basis.
Anxiety disorders
Anxiety is seen throughout the disease course of PD in approximately 30% to 50% of patients.21 It can manifest as generalized anxiety disorder, panic disorder, and other anxiety disorders. There are no high-quality RCTs of pharmacologic treatments of anxiety specifically in patients with PD, except for a negative safety and tolerability study of buspirone in which one-half of patients experienced worsening motor symptoms.15,22 Thus, the treatment of anxiety in patients with PD is similar to treatments in the general population. SSRIs and SNRIs are typically considered first-line, benzodiazepines are sometimes used with caution (although cognitive adverse effects and fall risk need to be considered), and nonpharmacologic treatments such as mindfulness yoga, exercise, CBT, and psychotherapy can be effective.16,21,23
Continue to: Because there is the lack...
Because there is the lack of evidence-based treatments for anxiety in PD, we highlight 2 PD-specific anxiety disorders: internal tremor, and nonmotor “off” anxiety.
Internal tremor
Internal tremor is a sense of vibration in the axial and/or appendicular muscles that cannot be seen externally by the patient or examiner. It is not yet fully understood if this phenomenon is sensory, anxiety-related, related to subclinical tremor, or the result of a combination of these factors (ie, sensory awareness of a subclinical tremor that triggers or is worsened by anxiety). There is some evidence for subclinical tremor on electromyography, but internal tremor does not respond to antiparkinsonian medications in 70% of patients.24 More electrophysiological research is needed to clarify this phenomenon. Internal tremor has been associated with anxiety in 64% of patients and often improves with anxiolytic therapies.24
Although poorly understood, internal tremor is a documented phenomenon in 33% to 44% of patients with PD, and in some cases, it may be an initial symptom that motivates a patient to seek medical attention for the first time.24,25 Internal tremor has also been reported in patients with essential tremor and multiple sclerosis.25 Therefore, physicians should be aware of internal tremor because this symptom could herald an underlying neurological disease.
Nonmotor ‘off’ anxiety
Patients with PD are commonly prescribed carbidopa-levodopa, a dopamine precursor, at least 3 times daily. Initially, this medication controls motor symptoms well from 1 dose to the next. However, as the disease progresses, some patients report motor fluctuations in which an individual dose of carbidopa-levodopa may wear off early, take longer than usual to take effect, or not take effect at all. Patients describe these periods as an “off” state in which they do not feel their medications are working. Such motor fluctuations can lead to anxiety and avoidance behaviors, because patients fear being in public at times when the medication does not adequately control their motor symptoms.
In addition to these motor symptom fluctuations and related anxiety, patients can also experience nonmotor symptom fluctuations. A wide variety of nonmotor symptoms, such as mood, cognitive, and behavioral symptoms, have been reported to fluctuate in parallel with motor symptoms.26,27 One study reported fluctuating restlessness in 39% of patients with PD, excessive worry in 17%, shortness of breath in 13%, excessive sweating and fear in 12%, and palpitations in 10%.27 A patient with fluctuating shortness of breath, sweating, and palpitations (for example) may repeatedly present to the emergency department with a negative cardiac workup and eventually be diagnosed with panic disorder, whereas the patient is truly experiencing nonmotor “off” symptoms. Thus, it is important to be aware of nonmotor fluctuations so this diagnosis can be made and the symptoms appropriately treated. The first step in treating nonmotor fluctuations is to optimize the antiparkinsonian regimen to minimize fluctuations. If “off” anxiety symptoms persist, anxiolytic medications can be prescribed.21
Continue to: Psychosis
Psychosis
Psychosis can occur in prodromal and early PD but is most common in advanced PD.28 One study reported that 60% of patients developed hallucinations or delusions after 12 years of follow-up.29 Disease duration, disease severity, dementia, and rapid eye movement sleep behavior disorder are significant risk factors for psychosis in PD.30 Well-formed visual hallucinations are the most common manifestation of psychosis in patients with PD. Auditory hallucinations and delusions are less common. Delusions are usually seen in patients with dementia and are often paranoid delusions, such as of spousal infidelity.30 Sensory hallucinations can occur, but should not be mistaken with formication, a central pain syndrome in PD that can represent a nonmotor “off” symptom that may respond to dopaminergic medication.31 Other more mild psychotic symptoms include illusions or misinterpretation of stimuli, false sense of presence, and passage hallucinations of fleeting figures in the peripheral vision.30
The pathophysiology of PD psychosis is not entirely understood but differs from psychosis in other disorders. It can occur in the absence of antiparkinsonian medication exposure and is thought to be a consequence of the underlying disease process of PD involving neurodegeneration in certain brain regions and aberrant neurotransmission of not only dopamine but also serotonin, acetylcholine, and glutamate.30
Figure 2 outlines the management of psychosis in PD. After addressing medical and medication-related causes, it is important to determine if the psychotic symptom is sufficiently bothersome to and/or potentially dangerous for the patient to warrant treatment. If treatment is indicated, pimavanserin and clozapine are efficacious for psychosis in PD without worsening motor symptoms, and quetiapine is possibly efficacious with a low risk of worsening motor symptoms.15 Other antipsychotics, such as olanzapine, risperidone, and haloperidol, can substantially worsen motor symptoms.15 Both second-generation antipsychotics and pimavanserin have an FDA black-box warning for a higher risk of all-cause mortality in older patients with dementia; however, because psychosis is associated with early mortality in PD, the risk/benefit ratio should be discussed with the patient and family for shared decision-making.30 If the patient also has dementia, rivastigmine—which is FDA-approved for PD dementia (PDD)—may also improve hallucinations.32

Cognitive disorders
This section focuses on PD mild cognitive impairment (PD-MCI) and PDD. When a patient with PD reports cognitive concerns, the approach outlined in Figure 3 can be used to diagnose the cognitive disorder. A detailed history, medication review, and physical examination can identify any medical or psychiatric conditions that could affect cognition. The American Academy of Neurology recommends screening for depression, obtaining blood levels of vitamin B12 and thyroid-stimulating hormone, and obtaining a CT or MRI of the brain to rule out reversible causes of dementia.33 A validated screening test such as the Montreal Cognitive Assessment, which has higher sensitivity for PD-MCI than the Mini-Mental State Examination, is used to identify and quantify cognitive impairment.34 Neuropsychological testing is the gold standard and can be used to confirm and/or better quantify the degree and domains of cognitive impairment.35 Typically, cognitive deficits in PD affect executive function, attention, and/or visuospatial domains more than memory and language early on, and deficits in visuospatial and language domains have the highest sensitivity for predicting progression to PDD.36

Once reversible causes of dementia are addressed or ruled out and cognitive testing is completed, the Movement Disorder Society (MDS) criteria for PD-MCI and PDD summarized in Figure 3 can be used to diagnose the cognitive disorder.37,38 The MDS criteria for PDD require a diagnosis of PD for ≥1 year prior to the onset of dementia to differentiate PDD from dementia with Lewy bodies (DLB). If the dementia starts within 1 year of the onset of parkinsonism, the diagnosis would be DLB. PDD and DLB are on the spectrum of Lewy body dementia, with the same Lewy body pathology in different temporal and spatial distributions in the brain.38
Continue to: PD-MCI is present in...
PD-MCI is present in approximately 25% of patients.35 PD-MCI does not always progress to dementia but increases the risk of dementia 6-fold. The prevalence of PDD increases with disease duration; it is present in approximately 50% of patients at 10 years and 80% of patients at 20 years of disease.35 Rivastigmine is the only FDA-approved medication to slow progression of PDD. There is insufficient evidence for other acetylcholinesterase inhibitors and memantine.15 Unfortunately, RCTs of pharmacotherapy for PD-MCI have failed to show efficacy. However, exercise, cognitive rehabilitation, and neuromodulation are being studied. In the meantime, addressing modifiable risk factors (such as vascular risk factors and alcohol consumption) and treating comorbid orthostatic hypotension, obstructive sleep apnea, and depression may improve cognition.35,39
Treatment-related disorders
Impulse control disorders
Impulse control disorders (ICDs) are an important medication-related consideration in patients with PD. The ICDs seen in PD include pathological gambling, binge eating, excessive shopping, hypersexual behaviors, and dopamine dysregulation syndrome (Table). These disorders are more common in younger patients with a history of impulsive personality traits and addictive behaviors (eg, history of tobacco or alcohol abuse), and are most strongly associated with dopaminergic therapies, particularly the dopamine agonists.40,41 In the DOMINION study, the odds of ICDs were 2- to 3.5-fold higher in patients taking dopamine agonists.42 This is mainly thought to be due to stimulation of D2/D3 receptors in the mesolimbic system.40 High doses of levodopa, monoamine oxidase inhibitors, and amantadine are also associated with ICDs.40-42

The first step in managing ICDs is diagnosing them, which can be difficult because patients often are not forthcoming about these problems due to embarrassment or failure to recognize that the ICD is related to PD medications. If a family member accompanies the patient at the visit, the patient may not want to disclose the amount of money they spend or the extent to which the behavior is a problem. Thus, a screening questionnaire, such as the Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease (QUIP) can be a helpful way for patients to alert the clinician to the issue.41 Education for the patient and family is crucial before the ICD causes significant financial, health, or relationship problems.
The mainstay of treatment is to reduce or taper off the dopamine agonist or other offending agent while monitoring for worsening motor symptoms and dopamine withdrawal syndrome. If this is unsuccessful, there is very limited evidence for further treatment strategies (Table), including antidepressants, antipsychotics, and mood stabilizers.40,43,44 There is insufficient evidence for naltrexone based on an RCT that failed to meet its primary endpoint, although naltrexone did significantly reduce QUIP scores.15,44 There is also insufficient evidence for amantadine, which showed benefit in some studies but was associated with ICDs in the DOMINION study.15,40,42 In terms of nonpharmacologic treatments, CBT is likely efficacious.15,40 There are mixed results for STN DBS. Some studies showed improvement in the ICD, due at least in part to dopaminergic medication reduction postoperatively, but this treatment has also been reported to increase impulsivity.40,45
Deep brain stimulation–related disorders
For patients with PD, the ideal lead location for STN DBS is the dorsolateral aspect of the STN, as this is the motor region of the nucleus. The STN functions in indirect and hyperdirect pathways to put the brake on certain motor programs so only the desired movement can be executed. Its function is clinically demonstrated by patients with STN stroke who develop excessive ballistic movements. Adjacent to the motor region of the STN is a centrally located associative region and a medially located limbic region. Thus, when stimulating the dorsolateral STN, current can spread to those regions as well, and the STN’s ability to put the brake on behavioral and emotional programs can be affected.46 Stimulation of the STN has been associated with mania, euphoria, new-onset ICDs, decreased verbal fluency, and executive dysfunction. Depression, apathy, and anxiety can also occur, but more commonly result from rapid withdrawal of antiparkinsonian medications after DBS surgery.46,47 Therefore, for PD patients with DBS with new or worsening psychiatric or cognitive symptoms, it is important to inquire about any recent programming sessions with neurology as well as recent self-increases in stimulation by the patient using their controller. Collaboration with neurology is important to troubleshoot whether stimulation could be contributing to the patient’s psychiatric or cognitive symptoms.
Continue to: Bottom Line
Bottom Line
Mood, anxiety, psychotic, and cognitive symptoms and disorders are common psychiatric manifestations associated with Parkinson’s disease (PD). In addition, patients with PD may experience impulsive control disorders and other symptoms related to treatments they receive for PD. Careful assessment and collaboration with neurology is crucial to alleviating the effects of these conditions.
Related Resources
- Weintraub D, Aarsland D, Chaudhuri KR, et al. The neuropsychiatry of Parkinson’s disease: advances and challenges. Lancet Neurology. 2022;21(1):89-102. doi:10.1016/S1474-4422(21)00330-6
- Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurologic Clinics. 2020;38(2):269-292. doi:10.1016/j.ncl.2019.12.003
- Castrioto A, Lhommee E, Moro E et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurology. 2014;13(3):287-305. doi:10.1016/ S1474-4422(13)70294-1
Drug Brand Names
Amantadine • Gocovri
Carbidopa-levodopa • Sinemet
Clozapine • Clozaril
Haloperidol • Haldol
Memantine • Namenda
Mirtazapine • Remeron
Naltrexone • Vivitrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimavanserin • Nuplazid
Piribedil • Pronoran
Pramipexole • Mirapex
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neupro
Venlafaxine • Effexor
Zonisamide • Zonegran
Parkinson’s disease (PD) is a neurodegenerative condition diagnosed pathologically by alpha synuclein–containing Lewy bodies and dopaminergic cell loss in the substantia nigra pars compacta of the midbrain. Loss of dopaminergic input to the caudate and putamen disrupts the direct and indirect basal ganglia pathways for motor control and contributes to the motor symptoms of PD.1 According to the Movement Disorder Society criteria, PD is diagnosed clinically by bradykinesia (slowness of movement) plus resting tremor and/or rigidity in the presence of supportive criteria, such as levodopa responsiveness and hyposmia, and in the absence of exclusion criteria and red flags that would suggest atypical parkinsonism or an alternative diagnosis.2
Although the diagnosis and treatment of PD focus heavily on the motor symptoms, nonmotor symptoms can arise decades before the onset of motor symptoms and continue throughout the lifespan. Nonmotor symptoms affect patients from head (ie, cognition and mood) to toe (ie, striatal toe pain) and multiple organ systems in between, including the olfactory, integumentary, cardiovascular, gastrointestinal, genitourinary, and autonomic nervous systems. Thus, it is not surprising that nonmotor symptoms of PD impact health-related quality of life more substantially than motor symptoms.3 A helpful analogy is to consider the motor symptoms of PD as the tip of the iceberg and the nonmotor symptoms as the larger, submerged portions of the iceberg.4
Nonmotor symptoms can negatively impact the treatment of motor symptoms. For example, imagine a patient who is very rigid and dyscoordinated in the arms and legs, which limits their ability to dress and walk. If this patient also suffers from nonmotor symptoms of orthostatic hypotension and psychosis—both of which can be exacerbated by levodopa—dose escalation of levodopa for the rigidity and dyscoordination could be compromised, rendering the patient undertreated and less mobile.
In this review, we focus on identifying and managing nonmotor symptoms of PD that are relevant to psychiatric practice, including mood and motivational disorders, anxiety disorders, psychosis, cognitive disorders, and disorders related to the pharmacologic and surgical treatment of PD (Figure 1).

Mood and motivational disorders
Depression
Depression is a common symptom in PD that can occur in the prodromal period years to decades before the onset of motor symptoms, as well as throughout the disease course.5 The prevalence of depression in PD varies from 3% to 90%, depending on the methods of assessment, clinical setting of assessment, motor symptom severity, and other factors; clinically significant depression likely affects approximately 35% to 38% of patients.5,6 How depression in patients with PD differs from depression in the general population is not entirely understood, but there does seem to be less guilt and suicidal ideation and a substantial component of negative affect, including dysphoria and anxiety.7 Practically speaking, depression is treated similarly in PD and general populations, with a few considerations.
Despite limited randomized controlled trials (RCTs) for efficacy specifically in patients with PD, selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are generally considered first-line treatments. There is also evidence for tricyclic antidepressants (TCAs), but due to potential worsening of orthostatic hypotension and cognition, TCAs may not be a favorable option for certain patients with PD.8,9 All antidepressants have the potential to worsen tremor. Theoretically, SNRIs, with noradrenergic activity, may be less tolerable than SSRIs in patients with PD. However, worsening tremor generally has not been a clinically significant adverse event reported in PD depression clinical trials, although it was seen in 17% of patients receiving paroxetine and 21% of patients receiving venlafaxine compared to 7% of patients receiving placebo.9-11 If tremor worsens, mirtazapine could be considered because it has been reported to cause less tremor than SSRIs or TCAs.12
Among medications for PD, pramipexole, a dopamine agonist, may have a beneficial effect on depression.13 Additionally, some evidence supports rasagiline, a monoamine oxidase type B inhibitor, as an adjunctive medication for depression in PD.14 Nevertheless, antidepressant medications remain the standard pharmacologic treatment for PD depression.
Continue to: In terms of nonpharmacologic options...
In terms of nonpharmacologic options, cognitive-behavioral therapy (CBT) is likely efficacious, exercise (especially yoga) is likely efficacious, and repetitive transcranial magnetic stimulation may be efficacious.15,16 While further high-quality trials are needed, these treatments are low-risk and can be considered, especially for patients who cannot tolerate medications.
Apathy
Apathy—a loss of motivation and goal-directed behavior—can occur in up to 30% of patients during the prodromal period of PD, and in up to 70% of patients throughout the disease course.17 Apathy can coexist with depression, which can make apathy difficult to diagnose.17 Given the time constraints of a clinic visit, a practical approach would be to first screen for depression and cognitive impairment. If there is continued suspicion of apathy, the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale part I question (“In the past week have you felt indifferent to doing activities or being with people?”) can be used to screen for apathy, and more detailed scales, such as the Apathy Scale (AS) or Lille Apathy Rating Scale (LARS), could be used if indicated.18
There are limited high-quality positive trials of apathy-specific treatments in PD. In an RCT of patients with PD who did not have depression or dementia, rivastigmine improved LARS scores compared to placebo.15 Piribedil, a D2/D3 receptor agonist, improved apathy in patients who underwent subthalamic nucleus deep brain stimulation (STN DBS).15 Exercise such as individualized physical therapy programs, dance, and Nordic walking as well as mindfulness interventions were shown to significantly reduce apathy scale scores.19 SSRIs, SNRIs, and rotigotine showed a trend toward reducing AS scores in RCTs.10,20
Larger, high-quality studies are needed to clarify the treatment of apathy in PD. In the meantime, a reasonable approach is to first treat any comorbid psychiatric or cognitive disorders, since apathy can be associated with these conditions, and to optimize antiparkinsonian medications for motor symptoms, motor fluctuations, and nonmotor fluctuations. Then, the investigational apathy treatments described in this section could be considered on an individual basis.
Anxiety disorders
Anxiety is seen throughout the disease course of PD in approximately 30% to 50% of patients.21 It can manifest as generalized anxiety disorder, panic disorder, and other anxiety disorders. There are no high-quality RCTs of pharmacologic treatments of anxiety specifically in patients with PD, except for a negative safety and tolerability study of buspirone in which one-half of patients experienced worsening motor symptoms.15,22 Thus, the treatment of anxiety in patients with PD is similar to treatments in the general population. SSRIs and SNRIs are typically considered first-line, benzodiazepines are sometimes used with caution (although cognitive adverse effects and fall risk need to be considered), and nonpharmacologic treatments such as mindfulness yoga, exercise, CBT, and psychotherapy can be effective.16,21,23
Continue to: Because there is the lack...
Because there is the lack of evidence-based treatments for anxiety in PD, we highlight 2 PD-specific anxiety disorders: internal tremor, and nonmotor “off” anxiety.
Internal tremor
Internal tremor is a sense of vibration in the axial and/or appendicular muscles that cannot be seen externally by the patient or examiner. It is not yet fully understood if this phenomenon is sensory, anxiety-related, related to subclinical tremor, or the result of a combination of these factors (ie, sensory awareness of a subclinical tremor that triggers or is worsened by anxiety). There is some evidence for subclinical tremor on electromyography, but internal tremor does not respond to antiparkinsonian medications in 70% of patients.24 More electrophysiological research is needed to clarify this phenomenon. Internal tremor has been associated with anxiety in 64% of patients and often improves with anxiolytic therapies.24
Although poorly understood, internal tremor is a documented phenomenon in 33% to 44% of patients with PD, and in some cases, it may be an initial symptom that motivates a patient to seek medical attention for the first time.24,25 Internal tremor has also been reported in patients with essential tremor and multiple sclerosis.25 Therefore, physicians should be aware of internal tremor because this symptom could herald an underlying neurological disease.
Nonmotor ‘off’ anxiety
Patients with PD are commonly prescribed carbidopa-levodopa, a dopamine precursor, at least 3 times daily. Initially, this medication controls motor symptoms well from 1 dose to the next. However, as the disease progresses, some patients report motor fluctuations in which an individual dose of carbidopa-levodopa may wear off early, take longer than usual to take effect, or not take effect at all. Patients describe these periods as an “off” state in which they do not feel their medications are working. Such motor fluctuations can lead to anxiety and avoidance behaviors, because patients fear being in public at times when the medication does not adequately control their motor symptoms.
In addition to these motor symptom fluctuations and related anxiety, patients can also experience nonmotor symptom fluctuations. A wide variety of nonmotor symptoms, such as mood, cognitive, and behavioral symptoms, have been reported to fluctuate in parallel with motor symptoms.26,27 One study reported fluctuating restlessness in 39% of patients with PD, excessive worry in 17%, shortness of breath in 13%, excessive sweating and fear in 12%, and palpitations in 10%.27 A patient with fluctuating shortness of breath, sweating, and palpitations (for example) may repeatedly present to the emergency department with a negative cardiac workup and eventually be diagnosed with panic disorder, whereas the patient is truly experiencing nonmotor “off” symptoms. Thus, it is important to be aware of nonmotor fluctuations so this diagnosis can be made and the symptoms appropriately treated. The first step in treating nonmotor fluctuations is to optimize the antiparkinsonian regimen to minimize fluctuations. If “off” anxiety symptoms persist, anxiolytic medications can be prescribed.21
Continue to: Psychosis
Psychosis
Psychosis can occur in prodromal and early PD but is most common in advanced PD.28 One study reported that 60% of patients developed hallucinations or delusions after 12 years of follow-up.29 Disease duration, disease severity, dementia, and rapid eye movement sleep behavior disorder are significant risk factors for psychosis in PD.30 Well-formed visual hallucinations are the most common manifestation of psychosis in patients with PD. Auditory hallucinations and delusions are less common. Delusions are usually seen in patients with dementia and are often paranoid delusions, such as of spousal infidelity.30 Sensory hallucinations can occur, but should not be mistaken with formication, a central pain syndrome in PD that can represent a nonmotor “off” symptom that may respond to dopaminergic medication.31 Other more mild psychotic symptoms include illusions or misinterpretation of stimuli, false sense of presence, and passage hallucinations of fleeting figures in the peripheral vision.30
The pathophysiology of PD psychosis is not entirely understood but differs from psychosis in other disorders. It can occur in the absence of antiparkinsonian medication exposure and is thought to be a consequence of the underlying disease process of PD involving neurodegeneration in certain brain regions and aberrant neurotransmission of not only dopamine but also serotonin, acetylcholine, and glutamate.30
Figure 2 outlines the management of psychosis in PD. After addressing medical and medication-related causes, it is important to determine if the psychotic symptom is sufficiently bothersome to and/or potentially dangerous for the patient to warrant treatment. If treatment is indicated, pimavanserin and clozapine are efficacious for psychosis in PD without worsening motor symptoms, and quetiapine is possibly efficacious with a low risk of worsening motor symptoms.15 Other antipsychotics, such as olanzapine, risperidone, and haloperidol, can substantially worsen motor symptoms.15 Both second-generation antipsychotics and pimavanserin have an FDA black-box warning for a higher risk of all-cause mortality in older patients with dementia; however, because psychosis is associated with early mortality in PD, the risk/benefit ratio should be discussed with the patient and family for shared decision-making.30 If the patient also has dementia, rivastigmine—which is FDA-approved for PD dementia (PDD)—may also improve hallucinations.32

Cognitive disorders
This section focuses on PD mild cognitive impairment (PD-MCI) and PDD. When a patient with PD reports cognitive concerns, the approach outlined in Figure 3 can be used to diagnose the cognitive disorder. A detailed history, medication review, and physical examination can identify any medical or psychiatric conditions that could affect cognition. The American Academy of Neurology recommends screening for depression, obtaining blood levels of vitamin B12 and thyroid-stimulating hormone, and obtaining a CT or MRI of the brain to rule out reversible causes of dementia.33 A validated screening test such as the Montreal Cognitive Assessment, which has higher sensitivity for PD-MCI than the Mini-Mental State Examination, is used to identify and quantify cognitive impairment.34 Neuropsychological testing is the gold standard and can be used to confirm and/or better quantify the degree and domains of cognitive impairment.35 Typically, cognitive deficits in PD affect executive function, attention, and/or visuospatial domains more than memory and language early on, and deficits in visuospatial and language domains have the highest sensitivity for predicting progression to PDD.36

Once reversible causes of dementia are addressed or ruled out and cognitive testing is completed, the Movement Disorder Society (MDS) criteria for PD-MCI and PDD summarized in Figure 3 can be used to diagnose the cognitive disorder.37,38 The MDS criteria for PDD require a diagnosis of PD for ≥1 year prior to the onset of dementia to differentiate PDD from dementia with Lewy bodies (DLB). If the dementia starts within 1 year of the onset of parkinsonism, the diagnosis would be DLB. PDD and DLB are on the spectrum of Lewy body dementia, with the same Lewy body pathology in different temporal and spatial distributions in the brain.38
Continue to: PD-MCI is present in...
PD-MCI is present in approximately 25% of patients.35 PD-MCI does not always progress to dementia but increases the risk of dementia 6-fold. The prevalence of PDD increases with disease duration; it is present in approximately 50% of patients at 10 years and 80% of patients at 20 years of disease.35 Rivastigmine is the only FDA-approved medication to slow progression of PDD. There is insufficient evidence for other acetylcholinesterase inhibitors and memantine.15 Unfortunately, RCTs of pharmacotherapy for PD-MCI have failed to show efficacy. However, exercise, cognitive rehabilitation, and neuromodulation are being studied. In the meantime, addressing modifiable risk factors (such as vascular risk factors and alcohol consumption) and treating comorbid orthostatic hypotension, obstructive sleep apnea, and depression may improve cognition.35,39
Treatment-related disorders
Impulse control disorders
Impulse control disorders (ICDs) are an important medication-related consideration in patients with PD. The ICDs seen in PD include pathological gambling, binge eating, excessive shopping, hypersexual behaviors, and dopamine dysregulation syndrome (Table). These disorders are more common in younger patients with a history of impulsive personality traits and addictive behaviors (eg, history of tobacco or alcohol abuse), and are most strongly associated with dopaminergic therapies, particularly the dopamine agonists.40,41 In the DOMINION study, the odds of ICDs were 2- to 3.5-fold higher in patients taking dopamine agonists.42 This is mainly thought to be due to stimulation of D2/D3 receptors in the mesolimbic system.40 High doses of levodopa, monoamine oxidase inhibitors, and amantadine are also associated with ICDs.40-42

The first step in managing ICDs is diagnosing them, which can be difficult because patients often are not forthcoming about these problems due to embarrassment or failure to recognize that the ICD is related to PD medications. If a family member accompanies the patient at the visit, the patient may not want to disclose the amount of money they spend or the extent to which the behavior is a problem. Thus, a screening questionnaire, such as the Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease (QUIP) can be a helpful way for patients to alert the clinician to the issue.41 Education for the patient and family is crucial before the ICD causes significant financial, health, or relationship problems.
The mainstay of treatment is to reduce or taper off the dopamine agonist or other offending agent while monitoring for worsening motor symptoms and dopamine withdrawal syndrome. If this is unsuccessful, there is very limited evidence for further treatment strategies (Table), including antidepressants, antipsychotics, and mood stabilizers.40,43,44 There is insufficient evidence for naltrexone based on an RCT that failed to meet its primary endpoint, although naltrexone did significantly reduce QUIP scores.15,44 There is also insufficient evidence for amantadine, which showed benefit in some studies but was associated with ICDs in the DOMINION study.15,40,42 In terms of nonpharmacologic treatments, CBT is likely efficacious.15,40 There are mixed results for STN DBS. Some studies showed improvement in the ICD, due at least in part to dopaminergic medication reduction postoperatively, but this treatment has also been reported to increase impulsivity.40,45
Deep brain stimulation–related disorders
For patients with PD, the ideal lead location for STN DBS is the dorsolateral aspect of the STN, as this is the motor region of the nucleus. The STN functions in indirect and hyperdirect pathways to put the brake on certain motor programs so only the desired movement can be executed. Its function is clinically demonstrated by patients with STN stroke who develop excessive ballistic movements. Adjacent to the motor region of the STN is a centrally located associative region and a medially located limbic region. Thus, when stimulating the dorsolateral STN, current can spread to those regions as well, and the STN’s ability to put the brake on behavioral and emotional programs can be affected.46 Stimulation of the STN has been associated with mania, euphoria, new-onset ICDs, decreased verbal fluency, and executive dysfunction. Depression, apathy, and anxiety can also occur, but more commonly result from rapid withdrawal of antiparkinsonian medications after DBS surgery.46,47 Therefore, for PD patients with DBS with new or worsening psychiatric or cognitive symptoms, it is important to inquire about any recent programming sessions with neurology as well as recent self-increases in stimulation by the patient using their controller. Collaboration with neurology is important to troubleshoot whether stimulation could be contributing to the patient’s psychiatric or cognitive symptoms.
Continue to: Bottom Line
Bottom Line
Mood, anxiety, psychotic, and cognitive symptoms and disorders are common psychiatric manifestations associated with Parkinson’s disease (PD). In addition, patients with PD may experience impulsive control disorders and other symptoms related to treatments they receive for PD. Careful assessment and collaboration with neurology is crucial to alleviating the effects of these conditions.
Related Resources
- Weintraub D, Aarsland D, Chaudhuri KR, et al. The neuropsychiatry of Parkinson’s disease: advances and challenges. Lancet Neurology. 2022;21(1):89-102. doi:10.1016/S1474-4422(21)00330-6
- Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurologic Clinics. 2020;38(2):269-292. doi:10.1016/j.ncl.2019.12.003
- Castrioto A, Lhommee E, Moro E et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurology. 2014;13(3):287-305. doi:10.1016/ S1474-4422(13)70294-1
Drug Brand Names
Amantadine • Gocovri
Carbidopa-levodopa • Sinemet
Clozapine • Clozaril
Haloperidol • Haldol
Memantine • Namenda
Mirtazapine • Remeron
Naltrexone • Vivitrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Pimavanserin • Nuplazid
Piribedil • Pronoran
Pramipexole • Mirapex
Quetiapine • Seroquel
Rasagiline • Azilect
Risperidone • Risperdal
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neupro
Venlafaxine • Effexor
Zonisamide • Zonegran
1. Bloem BR, Okun MS, Klein C. Parkinson’s disease. Lancet Neurology. 2021;397(10291):2284-2303.
2. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Movement Disorders. 2015;30(12):1591-1601.
3. Martinez-Martin P, Rodriguez-Blazquez C, Kurtiz MM, et al. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord. 2011;26(3):399-406.
4. Langston WJ. The Parkinson’s complex: parkinsonism is just the tip of the iceberg. Ann Neurol. 2006;59(4):591-596.
5. Cong S, Xiang C, Zhang S, et al. Prevalence and clinical aspects of depression in Parkinson’s disease: a systematic review and meta‑analysis of 129 studies. Neurosci Biobehav Rev. 2022;141:104749. doi:10.1016/j.neubiorev.2022.104749
6. Reijnders JS, Ehrt U, Weber WE, et al. A systematic review of prevalence studies in depression in Parkinson’s disease. Mov Disord. 2008;23(2):183-189.
7. Zahodne LB, Marsiske M, Okun MS, et al. Components of depression in Parkinson disease. J Geriatr Psychiatry Neurol. 2012;25(3):131-137.
8. Skapinakis P, Bakola E, Salanti G, et al. Efficacy and acceptability of selective serotonin reuptake inhibitors for the treatment of depression in Parkinson’s disease: a systematic review and meta-analysis of randomized controlled trials. BMC Neurology. 2010;10:49. doi:10.1186/1471-2377-10-49
9. Richard IH, McDermott MP, Kurlan R, et al; SAD-PD Study Group. A randomized, double-blind placebo-controlled trial of antidepressants in Parkinson’s disease. Neurology. 2012;78(16):1229-1236.
10. Takahashi M, Tabu H, Ozaki A, et al. Antidepressants for depression, apathy, and gait instability in Parkinson’s disease: a multicenter randomized study. Intern Med. 2019;58(3):361-368.
11. Bonuccelli U, Mecco G, Fabrini G, et al. A non-comparative assessment of tolerability and efficacy of duloxetine in the treatment of depressed patients with Parkinson’s disease. Expert Opin Pharmacother. 2012;13(16):2269-2280.
12. Wantanabe N, Omorio IM, Nakagawa A, et al; MANGA (Meta-Analysis of New Generation Antidepressants) Study Group. Safety reporting and adverse-event profile of mirtazapine described in randomized controlled trials in comparison with other classes of antidepressants in the acute-phase treatment of adults with depression. CNS Drugs. 2010;24(1):35-53.
13. Barone P, Scarzella L, Marconi R, et al; Depression/Parkinson Italian Study Group. Pramipexole versus sertraline in the treatment of depression in Parkinson’s disease: a national multicenter parallel-group randomized study. J Neurol. 2006;253(5):601-607.
14. Smith KM, Eyal E, Weintraub D, et al; ADAGIO Investigators. Combined rasagiline and anti-depressant use in Parkinson’s disease in the ADAGIO study: effects on non-motor symptoms and tolerability. JAMA Neurology. 2015;72(1):88-95.
15. Seppi K, Chaudhuri R, Coelho M, et al; the collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine Committee. Update on treatments for nonmotor symptoms of Parkinson’s disease--an evidence-based medicine review. Mov Disord. 2019;34(2):180-198.
16. Kwok JYY, Kwan JCY, Auyeung M, et al. Effects of mindfulness yoga vs stretching and resistance training exercises on anxiety and depression for people with Parkinson disease: a randomized clinical trial. JAMA Neurol. 2019;76(7):755-763.
17. De Waele S, Cras P, Crosiers D. Apathy in Parkinson’s disease: defining the Park apathy subtype. Brain Sci. 2022;12(7):923.
18. Mele B, Van S, Holroyd-Leduc J, et al. Diagnosis, treatment and management of apathy in Parkinson’s disease: a scoping review. BMJ Open. 2020;10(9):037632. doi:10.1136/bmjopen-2020-037632
19. Mele B, Ismail Z, Goodarzi Z, et al. Non-pharmacological interventions to treat apathy in Parkinson’s disease: a realist review. Clin Park Relat Disord. 2021;4:100096. doi:10.1016/j.prdoa.2021.100096
20. Chung SJ, Asgharnejad M, Bauer L, et al. Evaluation of rotigotine transdermal patch for the treatment of depressive symptoms in patients with Parkinson’s disease. Expert Opin Pharmacother. 2016;(17)11:1453-1461.
21. Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurol Clin. 2020;38(2):269-292.
22. Schneider RB, Auinger P, Tarolli CG, et al. A trial of buspirone for anxiety in Parkinson’s disease: safety and tolerability. Parkinsonism Relat Disord. 2020;81:69-74.
23. Moonen AJH, Mulders AEP, Defebvre L, et al. Cognitive behavioral therapy for anxiety in Parkinson’s disease: a randomized controlled trial. Mov Disord. 2021;36(11):2539-2548.
24. Shulman LM, Singer C, Bean JA, et al. Internal tremor in patient with Parkinson’s disease. Mov Disord. 1996;11(1):3-7.
25. Cochrane GD, Rizvi S, Abrantes A, et al. Internal tremor in Parkinson’s disease, multiple sclerosis, and essential tremor. Parkinsonism Relat Disord. 2015;21(10):1145-1147.
26. Del Prete E, Schmitt E, Meoni S, et al. Do neuropsychiatric fluctuations temporally match motor fluctuations in Parkinson’s disease? Neurol Sci. 2022;43(6):3641-3647.
27. Kleiner G, Fernandez HH, Chou KL, et al. Non-motor fluctuations in Parkinson’s disease: validation of the non-motor fluctuation assessment questionnaire. Mov Disord. 2021;36(6):1392-1400.
28. Pachi I, Maraki MI, Giagkou N, et al. Late life psychotic features in prodromal Parkinson’s disease. Parkinsonism Relat Disord. 2021;86:67-73.
29. Forsaa EB, Larsen JP, Wentzel-Larsen T, et al. A 12-year population-based study of psychosis in Parkinson’s disease. Arch Neurol. 2010;67(8):996-1001.
30. Chang A, Fox SH. Psychosis in Parkinson’s disease: epidemiology, pathophysiology, and management. Drugs. 2016;76(11):1093-1118.
31. Kasunich A, Kilbane C, Wiggins R. Movement disorders moment: pain and palliative care in movement disorders. Practical Neurology. 2021;20(4):63-67.
32. Burn D, Emre M, McKeith I, et al. Effects of rivastigmine in patients with and without visual hallucinations in dementia associated with Parkinson’s disease. Mov Disord. 2006;21(11):1899-1907.
33. Tripathi M, Vibha D. Reversible dementias. Indian J Psychiatry. 2009; 51 Suppl 1(Suppl 1): S52-S55.
34. Dalrymple-Alford JC, MacAskill MR, Nakas CT, et al. The MoCA: well-suited screen for cognitive impairment in Parkinson disease. Neurology. 2010;75(19):1717-1725.
35. Goldman J, Sieg, E. Cognitive impairment and dementia in Parkinson disease. Clin Geriatr Med. 2020;36(2):365-377.
36. Gonzalez-Latapi P, Bayram E, Litvan I, et al. Cognitive impairment in Parkinson’s disease: epidemiology, clinical profile, protective and risk factors. Behav Sci (Basel). 2021;11(5):74.
37. Litvan I, Goldman JG, Tröster AI, et al. Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society Task Force Guidelines. Mov Disord. 2012;27(3):349-356.
38. Dubois B, Burn D, Goetz C, et al. Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord. 2007;22(16):2314-2324.
39. Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers. 2021;7(1):47. doi:10.1038/s41572-021-00280-3
40. Weintraub D, Claassen DO. Impulse control and related disorders in Parkinson’s disease. Int Rev Neurobiol. 2017;133:679-717.
41. Vilas D, Pont-Sunyer C, Tolosa E. Impulse control disorders in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18 Suppl 1:S80-S84.
42. Weintraub D, Koester J, Potenza MN, et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch Neurol. 2010;67(5):589-595.
43. Faouzi J, Corvol JC, Mariani LL. Impulse control disorders and related behaviors in Parkinson’s disease: risk factors, clinical and genetic aspects, and management. Curr Opin Neurol. 2021;34(4):547-555.
44. Samuel M, Rodriguez-Oroz M, Antonini A, et al. Impulse control disorders in Parkinson’s disease: management, controversies, and potential approaches. Mov Disord. 2015;30(2):150-159.
45. Frank MJ, Samanta J, Moustafa AA, et al. Hold your horses: impulsivity, deep brain stimulation and medication in Parkinsonism. Science. 2007;318(5854):1309-1312.
46. Jahanshahi M, Obeso I, Baunez C, et al. Parkinson’s disease, the subthalamic nucleus, inhibition, and impulsivity. Mov Disord. 2015;30(2):128-140.
47. Castrioto A, Lhommée E, Moro E, et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurol. 2014;13(3):287-305.
1. Bloem BR, Okun MS, Klein C. Parkinson’s disease. Lancet Neurology. 2021;397(10291):2284-2303.
2. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Movement Disorders. 2015;30(12):1591-1601.
3. Martinez-Martin P, Rodriguez-Blazquez C, Kurtiz MM, et al. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord. 2011;26(3):399-406.
4. Langston WJ. The Parkinson’s complex: parkinsonism is just the tip of the iceberg. Ann Neurol. 2006;59(4):591-596.
5. Cong S, Xiang C, Zhang S, et al. Prevalence and clinical aspects of depression in Parkinson’s disease: a systematic review and meta‑analysis of 129 studies. Neurosci Biobehav Rev. 2022;141:104749. doi:10.1016/j.neubiorev.2022.104749
6. Reijnders JS, Ehrt U, Weber WE, et al. A systematic review of prevalence studies in depression in Parkinson’s disease. Mov Disord. 2008;23(2):183-189.
7. Zahodne LB, Marsiske M, Okun MS, et al. Components of depression in Parkinson disease. J Geriatr Psychiatry Neurol. 2012;25(3):131-137.
8. Skapinakis P, Bakola E, Salanti G, et al. Efficacy and acceptability of selective serotonin reuptake inhibitors for the treatment of depression in Parkinson’s disease: a systematic review and meta-analysis of randomized controlled trials. BMC Neurology. 2010;10:49. doi:10.1186/1471-2377-10-49
9. Richard IH, McDermott MP, Kurlan R, et al; SAD-PD Study Group. A randomized, double-blind placebo-controlled trial of antidepressants in Parkinson’s disease. Neurology. 2012;78(16):1229-1236.
10. Takahashi M, Tabu H, Ozaki A, et al. Antidepressants for depression, apathy, and gait instability in Parkinson’s disease: a multicenter randomized study. Intern Med. 2019;58(3):361-368.
11. Bonuccelli U, Mecco G, Fabrini G, et al. A non-comparative assessment of tolerability and efficacy of duloxetine in the treatment of depressed patients with Parkinson’s disease. Expert Opin Pharmacother. 2012;13(16):2269-2280.
12. Wantanabe N, Omorio IM, Nakagawa A, et al; MANGA (Meta-Analysis of New Generation Antidepressants) Study Group. Safety reporting and adverse-event profile of mirtazapine described in randomized controlled trials in comparison with other classes of antidepressants in the acute-phase treatment of adults with depression. CNS Drugs. 2010;24(1):35-53.
13. Barone P, Scarzella L, Marconi R, et al; Depression/Parkinson Italian Study Group. Pramipexole versus sertraline in the treatment of depression in Parkinson’s disease: a national multicenter parallel-group randomized study. J Neurol. 2006;253(5):601-607.
14. Smith KM, Eyal E, Weintraub D, et al; ADAGIO Investigators. Combined rasagiline and anti-depressant use in Parkinson’s disease in the ADAGIO study: effects on non-motor symptoms and tolerability. JAMA Neurology. 2015;72(1):88-95.
15. Seppi K, Chaudhuri R, Coelho M, et al; the collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine Committee. Update on treatments for nonmotor symptoms of Parkinson’s disease--an evidence-based medicine review. Mov Disord. 2019;34(2):180-198.
16. Kwok JYY, Kwan JCY, Auyeung M, et al. Effects of mindfulness yoga vs stretching and resistance training exercises on anxiety and depression for people with Parkinson disease: a randomized clinical trial. JAMA Neurol. 2019;76(7):755-763.
17. De Waele S, Cras P, Crosiers D. Apathy in Parkinson’s disease: defining the Park apathy subtype. Brain Sci. 2022;12(7):923.
18. Mele B, Van S, Holroyd-Leduc J, et al. Diagnosis, treatment and management of apathy in Parkinson’s disease: a scoping review. BMJ Open. 2020;10(9):037632. doi:10.1136/bmjopen-2020-037632
19. Mele B, Ismail Z, Goodarzi Z, et al. Non-pharmacological interventions to treat apathy in Parkinson’s disease: a realist review. Clin Park Relat Disord. 2021;4:100096. doi:10.1016/j.prdoa.2021.100096
20. Chung SJ, Asgharnejad M, Bauer L, et al. Evaluation of rotigotine transdermal patch for the treatment of depressive symptoms in patients with Parkinson’s disease. Expert Opin Pharmacother. 2016;(17)11:1453-1461.
21. Goldman JG, Guerra CM. Treatment of nonmotor symptoms associated with Parkinson disease. Neurol Clin. 2020;38(2):269-292.
22. Schneider RB, Auinger P, Tarolli CG, et al. A trial of buspirone for anxiety in Parkinson’s disease: safety and tolerability. Parkinsonism Relat Disord. 2020;81:69-74.
23. Moonen AJH, Mulders AEP, Defebvre L, et al. Cognitive behavioral therapy for anxiety in Parkinson’s disease: a randomized controlled trial. Mov Disord. 2021;36(11):2539-2548.
24. Shulman LM, Singer C, Bean JA, et al. Internal tremor in patient with Parkinson’s disease. Mov Disord. 1996;11(1):3-7.
25. Cochrane GD, Rizvi S, Abrantes A, et al. Internal tremor in Parkinson’s disease, multiple sclerosis, and essential tremor. Parkinsonism Relat Disord. 2015;21(10):1145-1147.
26. Del Prete E, Schmitt E, Meoni S, et al. Do neuropsychiatric fluctuations temporally match motor fluctuations in Parkinson’s disease? Neurol Sci. 2022;43(6):3641-3647.
27. Kleiner G, Fernandez HH, Chou KL, et al. Non-motor fluctuations in Parkinson’s disease: validation of the non-motor fluctuation assessment questionnaire. Mov Disord. 2021;36(6):1392-1400.
28. Pachi I, Maraki MI, Giagkou N, et al. Late life psychotic features in prodromal Parkinson’s disease. Parkinsonism Relat Disord. 2021;86:67-73.
29. Forsaa EB, Larsen JP, Wentzel-Larsen T, et al. A 12-year population-based study of psychosis in Parkinson’s disease. Arch Neurol. 2010;67(8):996-1001.
30. Chang A, Fox SH. Psychosis in Parkinson’s disease: epidemiology, pathophysiology, and management. Drugs. 2016;76(11):1093-1118.
31. Kasunich A, Kilbane C, Wiggins R. Movement disorders moment: pain and palliative care in movement disorders. Practical Neurology. 2021;20(4):63-67.
32. Burn D, Emre M, McKeith I, et al. Effects of rivastigmine in patients with and without visual hallucinations in dementia associated with Parkinson’s disease. Mov Disord. 2006;21(11):1899-1907.
33. Tripathi M, Vibha D. Reversible dementias. Indian J Psychiatry. 2009; 51 Suppl 1(Suppl 1): S52-S55.
34. Dalrymple-Alford JC, MacAskill MR, Nakas CT, et al. The MoCA: well-suited screen for cognitive impairment in Parkinson disease. Neurology. 2010;75(19):1717-1725.
35. Goldman J, Sieg, E. Cognitive impairment and dementia in Parkinson disease. Clin Geriatr Med. 2020;36(2):365-377.
36. Gonzalez-Latapi P, Bayram E, Litvan I, et al. Cognitive impairment in Parkinson’s disease: epidemiology, clinical profile, protective and risk factors. Behav Sci (Basel). 2021;11(5):74.
37. Litvan I, Goldman JG, Tröster AI, et al. Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society Task Force Guidelines. Mov Disord. 2012;27(3):349-356.
38. Dubois B, Burn D, Goetz C, et al. Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord. 2007;22(16):2314-2324.
39. Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers. 2021;7(1):47. doi:10.1038/s41572-021-00280-3
40. Weintraub D, Claassen DO. Impulse control and related disorders in Parkinson’s disease. Int Rev Neurobiol. 2017;133:679-717.
41. Vilas D, Pont-Sunyer C, Tolosa E. Impulse control disorders in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18 Suppl 1:S80-S84.
42. Weintraub D, Koester J, Potenza MN, et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch Neurol. 2010;67(5):589-595.
43. Faouzi J, Corvol JC, Mariani LL. Impulse control disorders and related behaviors in Parkinson’s disease: risk factors, clinical and genetic aspects, and management. Curr Opin Neurol. 2021;34(4):547-555.
44. Samuel M, Rodriguez-Oroz M, Antonini A, et al. Impulse control disorders in Parkinson’s disease: management, controversies, and potential approaches. Mov Disord. 2015;30(2):150-159.
45. Frank MJ, Samanta J, Moustafa AA, et al. Hold your horses: impulsivity, deep brain stimulation and medication in Parkinsonism. Science. 2007;318(5854):1309-1312.
46. Jahanshahi M, Obeso I, Baunez C, et al. Parkinson’s disease, the subthalamic nucleus, inhibition, and impulsivity. Mov Disord. 2015;30(2):128-140.
47. Castrioto A, Lhommée E, Moro E, et al. Mood and behavioral effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurol. 2014;13(3):287-305.
Online CBT aids remission of anxiety, depression in students
, according to a study published in JAMA Psychiatry . The intervention was developed by researchers from the United States, Mexico, and Colombia and studied in undergraduate university students.
The research included 1,319 students with anxiety and depression. The students were randomly assigned to three groups that received either remote (internet-based) cognitive behavioral therapy guided by a therapist, self-guided cognitive behavioral therapy (without support from a therapist), or standard treatment provided by the health care services within their community (the control condition).
Students who received guided cognitive behavioral therapy had higher combined rates of remission of these disorders (51.8%) than students who received self-guided therapy (37.8%) or conventional therapy (40%). These differences were not significant for remission of anxiety, however.
Guided cognitive behavioral therapy was associated with the highest probability of remission of anxiety and depression in 91.7% of students, the highest probability of remission of anxiety in all students, and the highest probability of remission of depression in 71.5% of participants.
The results of this analysis could be used to improve psychological care by optimizing how different treatment methods are assigned, especially in mental health institutions where available technical and human resources are limited, according to the investigators.
“We started designing this study before COVID-19 with the idea of optimizing care for these mental health problems,” said study author Corina Benjet Miner, PhD, an epidemiological and psychosocial researcher at the Ramón de la Fuente National Institute of Psychiatry, Mexico City. “We wanted to find additional strategies to achieve better care. The pandemic helped us because, even though this has been undergoing research for many years, internet-delivered interventions were not as well accepted. But during the pandemic, there weren’t any other options.”
Given the high prevalence of mental disorders before and after the pandemic, no health care system in the world would be able to provide in-person care to each patient with depression or anxiety, said Dr. Benjet Miner. “So, the idea is to look for other cost-effective strategies that can ramp up our interventions and reach a greater number of people without negatively impacting the quality of care,” she explained.
“I believe that [the precision model] is an excellent proposal that can save financial resources and avoid transfers,” said Juana Olvera Méndez, PhD, research professor working with the cognitive behavioral approach at the Iztacala Faculty of Higher Studies (FESI) of the National Autonomous University of Mexico, Mexico City. “It also makes it possible to provide patients with immediate care, in contrast to when someone has to go in for [in-person] therapy, which will depend a lot on how the therapist approaches the situation.”
Students from seven universities in Colombia and Mexico were included in the study. They were aged 18 years or older and had a score of 10 or greater on the self-administered Generalized Anxiety Disorder scale-7 test, or had depression with scores of 10 or greater on the nine-item Patient Health Questionnaire, which is also self-administered.
The study’s exclusion criteria included a history of bipolar disorder, nonaffective psychosis, or suicidal ideation with suicide attempts. The investigators used 284 prescription predictors to anticipate the differential response to antianxiety and antidepression therapy.
By grouping these predictors into 11 conceptual categories (such as demographic characteristics, COVID-19–linked stressors, or mental disorder comorbidities) and using machine learning algorithms, the investigators were able to predict in an individualized manner the probability of remission for participants in each of the groups.
“For depression, we found that 28.5% of patients could experience better or equivalent effects from the self-guided program (in comparison to the guided program). Once you have this program, it doesn’t cost anything, so there could be a massive number of people who could benefit from a cost-free therapy,” said Dr. Benjet Miner.
While numerous studies in precision medicine have tried to determine the most appropriate treatment for each patient, “they don’t have the high number of predictors that we used in this research, and I feel like this gives us a significant edge,” she added.
She also explained that they found no differences in user satisfaction between the guided and unguided version of the therapy, so now they must discover why the guided version works better. One notable point is that patients accessed (online) the guided program twice as many times as those who used the self-guided version, but the number of times used is not enough to explain the better outcomes.
“We believe that patients develop some sort of connection with the guides, who are not providing therapy but only making recommendations in brief interactions with patients once a week. It has something to do with that connection, in addition to the longer time spent interacting with the platform, which provides better results with the guided version,” stated Dr. Benjet Miner.
One of the main limitations of this study is that, though it compares three treatment methods, the third one (standard care) is not homogeneous, because each of the seven universities from which the students were selected has different resources for this purpose. “Some universities, like the National Autonomous University of Mexico, have very formal services, with teams of psychologists and psychiatrists, while others don’t have this type of service, or they cover additional aspects, like vocational counseling. So, it’s very difficult to determine exactly what kind of care patients are receiving, because it’s not homogeneous,” she said.
As many as nine assessments using psychometric tests are sometimes required before the intervention can be evaluated, said Dr. Méndez. “This study doesn’t go into too much detail in that area, focusing rather on treatment. So, it would be important to know the diagnoses of the users, who may be experiencing different degrees of depression or anxiety. It would be worth asking what happens if a user requires psychiatric treatment or support.”
Dr. Méndez, who provides psychological therapy in person and online at the Student Support and Counselling Center at FESI, pointed out that it would be important to provide close follow-up on these results to see whether they are sustained in the short and long terms. In her opinion, this model could be presented to other users requiring treatment for anxiety or depression, provided that they can use information and communication technologies.
This precision model, which can also be supported on mobile phones or tablets, could be transferred to primary care facilities or vulnerable populations in rural areas, said Dr. Benjet Miner. “The idea is to reach a point where these algorithms become accurate enough and have a really strong predictive power so that clinicians can use them. The goal is always to find the best treatment at the lowest cost, so that it’s sustainable,” she concluded.
This study was funded by grant number R01MH120648 from the National Institute of Mental Health and the Fogarty International Center. Dr. Benjet Miner reports no relevant financial relationships; the declarations of the remaining authors can be found at the publication’s website.
This article was translated from Medscape’s Spanish Edition and a version first appeared on Medscape.com.
, according to a study published in JAMA Psychiatry . The intervention was developed by researchers from the United States, Mexico, and Colombia and studied in undergraduate university students.
The research included 1,319 students with anxiety and depression. The students were randomly assigned to three groups that received either remote (internet-based) cognitive behavioral therapy guided by a therapist, self-guided cognitive behavioral therapy (without support from a therapist), or standard treatment provided by the health care services within their community (the control condition).
Students who received guided cognitive behavioral therapy had higher combined rates of remission of these disorders (51.8%) than students who received self-guided therapy (37.8%) or conventional therapy (40%). These differences were not significant for remission of anxiety, however.
Guided cognitive behavioral therapy was associated with the highest probability of remission of anxiety and depression in 91.7% of students, the highest probability of remission of anxiety in all students, and the highest probability of remission of depression in 71.5% of participants.
The results of this analysis could be used to improve psychological care by optimizing how different treatment methods are assigned, especially in mental health institutions where available technical and human resources are limited, according to the investigators.
“We started designing this study before COVID-19 with the idea of optimizing care for these mental health problems,” said study author Corina Benjet Miner, PhD, an epidemiological and psychosocial researcher at the Ramón de la Fuente National Institute of Psychiatry, Mexico City. “We wanted to find additional strategies to achieve better care. The pandemic helped us because, even though this has been undergoing research for many years, internet-delivered interventions were not as well accepted. But during the pandemic, there weren’t any other options.”
Given the high prevalence of mental disorders before and after the pandemic, no health care system in the world would be able to provide in-person care to each patient with depression or anxiety, said Dr. Benjet Miner. “So, the idea is to look for other cost-effective strategies that can ramp up our interventions and reach a greater number of people without negatively impacting the quality of care,” she explained.
“I believe that [the precision model] is an excellent proposal that can save financial resources and avoid transfers,” said Juana Olvera Méndez, PhD, research professor working with the cognitive behavioral approach at the Iztacala Faculty of Higher Studies (FESI) of the National Autonomous University of Mexico, Mexico City. “It also makes it possible to provide patients with immediate care, in contrast to when someone has to go in for [in-person] therapy, which will depend a lot on how the therapist approaches the situation.”
Students from seven universities in Colombia and Mexico were included in the study. They were aged 18 years or older and had a score of 10 or greater on the self-administered Generalized Anxiety Disorder scale-7 test, or had depression with scores of 10 or greater on the nine-item Patient Health Questionnaire, which is also self-administered.
The study’s exclusion criteria included a history of bipolar disorder, nonaffective psychosis, or suicidal ideation with suicide attempts. The investigators used 284 prescription predictors to anticipate the differential response to antianxiety and antidepression therapy.
By grouping these predictors into 11 conceptual categories (such as demographic characteristics, COVID-19–linked stressors, or mental disorder comorbidities) and using machine learning algorithms, the investigators were able to predict in an individualized manner the probability of remission for participants in each of the groups.
“For depression, we found that 28.5% of patients could experience better or equivalent effects from the self-guided program (in comparison to the guided program). Once you have this program, it doesn’t cost anything, so there could be a massive number of people who could benefit from a cost-free therapy,” said Dr. Benjet Miner.
While numerous studies in precision medicine have tried to determine the most appropriate treatment for each patient, “they don’t have the high number of predictors that we used in this research, and I feel like this gives us a significant edge,” she added.
She also explained that they found no differences in user satisfaction between the guided and unguided version of the therapy, so now they must discover why the guided version works better. One notable point is that patients accessed (online) the guided program twice as many times as those who used the self-guided version, but the number of times used is not enough to explain the better outcomes.
“We believe that patients develop some sort of connection with the guides, who are not providing therapy but only making recommendations in brief interactions with patients once a week. It has something to do with that connection, in addition to the longer time spent interacting with the platform, which provides better results with the guided version,” stated Dr. Benjet Miner.
One of the main limitations of this study is that, though it compares three treatment methods, the third one (standard care) is not homogeneous, because each of the seven universities from which the students were selected has different resources for this purpose. “Some universities, like the National Autonomous University of Mexico, have very formal services, with teams of psychologists and psychiatrists, while others don’t have this type of service, or they cover additional aspects, like vocational counseling. So, it’s very difficult to determine exactly what kind of care patients are receiving, because it’s not homogeneous,” she said.
As many as nine assessments using psychometric tests are sometimes required before the intervention can be evaluated, said Dr. Méndez. “This study doesn’t go into too much detail in that area, focusing rather on treatment. So, it would be important to know the diagnoses of the users, who may be experiencing different degrees of depression or anxiety. It would be worth asking what happens if a user requires psychiatric treatment or support.”
Dr. Méndez, who provides psychological therapy in person and online at the Student Support and Counselling Center at FESI, pointed out that it would be important to provide close follow-up on these results to see whether they are sustained in the short and long terms. In her opinion, this model could be presented to other users requiring treatment for anxiety or depression, provided that they can use information and communication technologies.
This precision model, which can also be supported on mobile phones or tablets, could be transferred to primary care facilities or vulnerable populations in rural areas, said Dr. Benjet Miner. “The idea is to reach a point where these algorithms become accurate enough and have a really strong predictive power so that clinicians can use them. The goal is always to find the best treatment at the lowest cost, so that it’s sustainable,” she concluded.
This study was funded by grant number R01MH120648 from the National Institute of Mental Health and the Fogarty International Center. Dr. Benjet Miner reports no relevant financial relationships; the declarations of the remaining authors can be found at the publication’s website.
This article was translated from Medscape’s Spanish Edition and a version first appeared on Medscape.com.
, according to a study published in JAMA Psychiatry . The intervention was developed by researchers from the United States, Mexico, and Colombia and studied in undergraduate university students.
The research included 1,319 students with anxiety and depression. The students were randomly assigned to three groups that received either remote (internet-based) cognitive behavioral therapy guided by a therapist, self-guided cognitive behavioral therapy (without support from a therapist), or standard treatment provided by the health care services within their community (the control condition).
Students who received guided cognitive behavioral therapy had higher combined rates of remission of these disorders (51.8%) than students who received self-guided therapy (37.8%) or conventional therapy (40%). These differences were not significant for remission of anxiety, however.
Guided cognitive behavioral therapy was associated with the highest probability of remission of anxiety and depression in 91.7% of students, the highest probability of remission of anxiety in all students, and the highest probability of remission of depression in 71.5% of participants.
The results of this analysis could be used to improve psychological care by optimizing how different treatment methods are assigned, especially in mental health institutions where available technical and human resources are limited, according to the investigators.
“We started designing this study before COVID-19 with the idea of optimizing care for these mental health problems,” said study author Corina Benjet Miner, PhD, an epidemiological and psychosocial researcher at the Ramón de la Fuente National Institute of Psychiatry, Mexico City. “We wanted to find additional strategies to achieve better care. The pandemic helped us because, even though this has been undergoing research for many years, internet-delivered interventions were not as well accepted. But during the pandemic, there weren’t any other options.”
Given the high prevalence of mental disorders before and after the pandemic, no health care system in the world would be able to provide in-person care to each patient with depression or anxiety, said Dr. Benjet Miner. “So, the idea is to look for other cost-effective strategies that can ramp up our interventions and reach a greater number of people without negatively impacting the quality of care,” she explained.
“I believe that [the precision model] is an excellent proposal that can save financial resources and avoid transfers,” said Juana Olvera Méndez, PhD, research professor working with the cognitive behavioral approach at the Iztacala Faculty of Higher Studies (FESI) of the National Autonomous University of Mexico, Mexico City. “It also makes it possible to provide patients with immediate care, in contrast to when someone has to go in for [in-person] therapy, which will depend a lot on how the therapist approaches the situation.”
Students from seven universities in Colombia and Mexico were included in the study. They were aged 18 years or older and had a score of 10 or greater on the self-administered Generalized Anxiety Disorder scale-7 test, or had depression with scores of 10 or greater on the nine-item Patient Health Questionnaire, which is also self-administered.
The study’s exclusion criteria included a history of bipolar disorder, nonaffective psychosis, or suicidal ideation with suicide attempts. The investigators used 284 prescription predictors to anticipate the differential response to antianxiety and antidepression therapy.
By grouping these predictors into 11 conceptual categories (such as demographic characteristics, COVID-19–linked stressors, or mental disorder comorbidities) and using machine learning algorithms, the investigators were able to predict in an individualized manner the probability of remission for participants in each of the groups.
“For depression, we found that 28.5% of patients could experience better or equivalent effects from the self-guided program (in comparison to the guided program). Once you have this program, it doesn’t cost anything, so there could be a massive number of people who could benefit from a cost-free therapy,” said Dr. Benjet Miner.
While numerous studies in precision medicine have tried to determine the most appropriate treatment for each patient, “they don’t have the high number of predictors that we used in this research, and I feel like this gives us a significant edge,” she added.
She also explained that they found no differences in user satisfaction between the guided and unguided version of the therapy, so now they must discover why the guided version works better. One notable point is that patients accessed (online) the guided program twice as many times as those who used the self-guided version, but the number of times used is not enough to explain the better outcomes.
“We believe that patients develop some sort of connection with the guides, who are not providing therapy but only making recommendations in brief interactions with patients once a week. It has something to do with that connection, in addition to the longer time spent interacting with the platform, which provides better results with the guided version,” stated Dr. Benjet Miner.
One of the main limitations of this study is that, though it compares three treatment methods, the third one (standard care) is not homogeneous, because each of the seven universities from which the students were selected has different resources for this purpose. “Some universities, like the National Autonomous University of Mexico, have very formal services, with teams of psychologists and psychiatrists, while others don’t have this type of service, or they cover additional aspects, like vocational counseling. So, it’s very difficult to determine exactly what kind of care patients are receiving, because it’s not homogeneous,” she said.
As many as nine assessments using psychometric tests are sometimes required before the intervention can be evaluated, said Dr. Méndez. “This study doesn’t go into too much detail in that area, focusing rather on treatment. So, it would be important to know the diagnoses of the users, who may be experiencing different degrees of depression or anxiety. It would be worth asking what happens if a user requires psychiatric treatment or support.”
Dr. Méndez, who provides psychological therapy in person and online at the Student Support and Counselling Center at FESI, pointed out that it would be important to provide close follow-up on these results to see whether they are sustained in the short and long terms. In her opinion, this model could be presented to other users requiring treatment for anxiety or depression, provided that they can use information and communication technologies.
This precision model, which can also be supported on mobile phones or tablets, could be transferred to primary care facilities or vulnerable populations in rural areas, said Dr. Benjet Miner. “The idea is to reach a point where these algorithms become accurate enough and have a really strong predictive power so that clinicians can use them. The goal is always to find the best treatment at the lowest cost, so that it’s sustainable,” she concluded.
This study was funded by grant number R01MH120648 from the National Institute of Mental Health and the Fogarty International Center. Dr. Benjet Miner reports no relevant financial relationships; the declarations of the remaining authors can be found at the publication’s website.
This article was translated from Medscape’s Spanish Edition and a version first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Universal anxiety screening recommendation is a good start
A very good thing happened this summer for patients with anxiety and the psychiatrists, psychologists, and other mental health professionals who provide treatment for them. The U.S. Preventive Services Task Force recommended anxiety screening for all adults younger than 65.
On the surface, this is a great recommendation for recognition and caring for those who deal with and suffer from an anxiety disorder or multiple anxiety disorders. Although the USPSTF recommendations are independent of the U.S. government and are not an official position of the Department of Health & Human Services, they are a wonderful start at recognizing the importance of mental health care.
After all, anxiety disorders are the most commonly experienced and diagnosed mental disorders, according to the DSM-5.
They range mainly from generalized anxiety disorder (GAD), to panic attacks and panic disorder, separation anxiety, specific type phobias (bridges, tunnels, insects, snakes, and the list goes on), to other phobias, including agoraphobia, social phobia, and of course, anxiety caused by medical conditions. GAD alone occurs in, at least, more than 3% of the population.
Those of us who have been treating anxiety disorders for decades recognize them as an issue affecting both mental and physical well-being, not only because of the emotional causes but the physical distress and illnesses that anxiety may precipitate or worsen.
For example, blood pressure– and heart-related issues, GI disorders, and musculoskeletal issues are just a few examples of how our bodies and organ systems are affected by anxiety. Just the momentary physical symptoms of tachycardia or the “runs” before an exam are fine examples of how anxiety may affect patients physically, and an ongoing, consistent anxiety is potentially more harmful.
In fact, a first panic attack or episode of generalized anxiety may be so serious that an emergency department or physician visit is necessary to rule out a heart attack, asthma, or breathing issues – even a hormone or thyroid emergency, or a cardiac arrhythmia. Panic attacks alone create a high number of ED visits.
Treatments mainly include medication management and a variety of psychotherapy techniques. Currently, the most preferred, first-choice medications are the SSRI antidepressants, which are Food and Drug Administration approved for anxiety as well. These include Zoloft (sertraline), Prozac/Sarafem (fluoxetine), Celexa (citalopram), and Lexapro (escitalopram).
For many years, benzodiazepines (that is, tranquillizers) such as Valium (diazepam), Ativan (lorazepam), and Klonopin/Rivotril (clonazepam) to name a few, were the mainstay of anxiety treatment, but they have proven addictive and may affect cognition and memory. As the current opioid epidemic has shown, when combined with opioids, benzodiazepines are a potentially lethal combination and when used, they need to be for shorter-term care and monitored very judiciously.
It should be noted that after ongoing long-term use of an SSRI for anxiety or depression, it should not be stopped abruptly, as a variety of physical symptoms (for example, flu-like symptoms) may occur.
Benefits of nonmedicinal therapies
There are a variety of talk therapies, from dynamic psychotherapies to cognitive-behavioral therapies (CBT), plus relaxation techniques and guided imagery that have all had a good amount of success in treating generalized anxiety, panic disorder, as well as various types of phobias.
When medications are stopped, the anxiety symptoms may well return. But when using nonmedicinal therapies, clinicians have discovered that when patients develop a new perspective on the anxiety problem or have a new technique to treat anxiety, it may well be long lasting.
For me, using CBT, relaxation techniques, hypnosis, and guided imagery has been very successful in treating anxiety disorders with long-lasting results. Once a person learns to relax, whether it’s from deep breathing exercises, hypnosis (which is not sleep), mindfulness, or meditation, a strategy of guided imagery can be taught, which allows a person to practice as well as control their anxiety as a lifetime process. For example, I like imagining a large movie screen to desensitize and project anxieties.
In many instances, a combination of a medication and a talk therapy approach works best, but there are an equal number of instances in which just medication or just talk therapy is needed. Once again, knowledge, clinical judgment, and the art of care are required to make these assessments.
In other words, recognizing and treating anxiety requires highly specialized training, which is why I thought the USPSTF recommendations raise a few critical questions.
Questions and concerns
One issue, of course, is the exclusion of those patients over age 65 because of a lack of “data.” Why such an exclusion? Does this mean that data are lacking for this age group?
The concept of using solely evidenced-based data in psychiatry is itself an interesting concept because our profession, like many other medical specialties, requires practitioners to use a combination of art and science. And much can be said either way about the clarity of accuracy in the diversity of issues that arise when treating emotional disorders.
When looking at the over-65 population, has anyone thought of clinical knowledge, judgment, experience, observation, and, of course, common sense?
Just consider the worry (a cardinal feature of anxiety) that besets people over 65 when it comes to issues such as retirement, financial security, “empty nesting,” physical health issues, decreased socialization that resulted from the COVID-19 pandemic, and the perpetual loss of relatives and friends.
In addition, as we age, anxiety can come simply from the loss of identity as active lifestyles decrease and the reality of nearing life’s end becomes more of a reality. It would seem that this population would benefit enormously from anxiety screening and possible treatment.
Another major concern is that the screening and potential treatment of patients is aimed at primary care physicians. Putting the sole responsibility of providing mental health care on these overworked PCPs defies common sense unless we’re okay with 1- to 2-minute assessments of mental health issues and no doubt, a pharmacology-only approach.
If this follows the same route as well-intentioned PCPs treating depression, where 5-minute medication management is far too common, the only proper diagnostic course – the in-depth interview necessary to make a proper diagnosis – is often missing.
For example, in depression alone, it takes psychiatric experience and time to differentiate a major depressive disorder from a bipolar depression and to provide the appropriate medication and treatment plan with careful follow-up. In my experience, this usually does not happen in the exceedingly overworked, time-driven day of a PCP.
Anxiety disorders and depression can prove debilitating, and if a PCP wants the responsibility of treatment, a mandated mental health program should be followed – just as here in New York, prescribers are mandated to take a pain control, opioid, and infection control CME course to keep our licenses up to date.
Short of mandating a mental health program for PCPs, it should be part of training and CME courses that Psychiatry is a super specialty, much like orthopedics and ophthalmology, and primary care physicians should never hesitate to make referrals to the specialist.
The big picture for me, and I hope for us all, is that the USPSTF has started things rolling by making it clear that PCPs and other health care clinicians need to screen for anxiety as a disabling disorder that is quite treatable.
This approach will help to advance the destigmatization of mental health disorders. But as result, with more patients diagnosed, there will be a need for more psychiatrists – and psychologists with PhDs or PsyDs – to fill the gaps in mental health care.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.
A very good thing happened this summer for patients with anxiety and the psychiatrists, psychologists, and other mental health professionals who provide treatment for them. The U.S. Preventive Services Task Force recommended anxiety screening for all adults younger than 65.
On the surface, this is a great recommendation for recognition and caring for those who deal with and suffer from an anxiety disorder or multiple anxiety disorders. Although the USPSTF recommendations are independent of the U.S. government and are not an official position of the Department of Health & Human Services, they are a wonderful start at recognizing the importance of mental health care.
After all, anxiety disorders are the most commonly experienced and diagnosed mental disorders, according to the DSM-5.
They range mainly from generalized anxiety disorder (GAD), to panic attacks and panic disorder, separation anxiety, specific type phobias (bridges, tunnels, insects, snakes, and the list goes on), to other phobias, including agoraphobia, social phobia, and of course, anxiety caused by medical conditions. GAD alone occurs in, at least, more than 3% of the population.
Those of us who have been treating anxiety disorders for decades recognize them as an issue affecting both mental and physical well-being, not only because of the emotional causes but the physical distress and illnesses that anxiety may precipitate or worsen.
For example, blood pressure– and heart-related issues, GI disorders, and musculoskeletal issues are just a few examples of how our bodies and organ systems are affected by anxiety. Just the momentary physical symptoms of tachycardia or the “runs” before an exam are fine examples of how anxiety may affect patients physically, and an ongoing, consistent anxiety is potentially more harmful.
In fact, a first panic attack or episode of generalized anxiety may be so serious that an emergency department or physician visit is necessary to rule out a heart attack, asthma, or breathing issues – even a hormone or thyroid emergency, or a cardiac arrhythmia. Panic attacks alone create a high number of ED visits.
Treatments mainly include medication management and a variety of psychotherapy techniques. Currently, the most preferred, first-choice medications are the SSRI antidepressants, which are Food and Drug Administration approved for anxiety as well. These include Zoloft (sertraline), Prozac/Sarafem (fluoxetine), Celexa (citalopram), and Lexapro (escitalopram).
For many years, benzodiazepines (that is, tranquillizers) such as Valium (diazepam), Ativan (lorazepam), and Klonopin/Rivotril (clonazepam) to name a few, were the mainstay of anxiety treatment, but they have proven addictive and may affect cognition and memory. As the current opioid epidemic has shown, when combined with opioids, benzodiazepines are a potentially lethal combination and when used, they need to be for shorter-term care and monitored very judiciously.
It should be noted that after ongoing long-term use of an SSRI for anxiety or depression, it should not be stopped abruptly, as a variety of physical symptoms (for example, flu-like symptoms) may occur.
Benefits of nonmedicinal therapies
There are a variety of talk therapies, from dynamic psychotherapies to cognitive-behavioral therapies (CBT), plus relaxation techniques and guided imagery that have all had a good amount of success in treating generalized anxiety, panic disorder, as well as various types of phobias.
When medications are stopped, the anxiety symptoms may well return. But when using nonmedicinal therapies, clinicians have discovered that when patients develop a new perspective on the anxiety problem or have a new technique to treat anxiety, it may well be long lasting.
For me, using CBT, relaxation techniques, hypnosis, and guided imagery has been very successful in treating anxiety disorders with long-lasting results. Once a person learns to relax, whether it’s from deep breathing exercises, hypnosis (which is not sleep), mindfulness, or meditation, a strategy of guided imagery can be taught, which allows a person to practice as well as control their anxiety as a lifetime process. For example, I like imagining a large movie screen to desensitize and project anxieties.
In many instances, a combination of a medication and a talk therapy approach works best, but there are an equal number of instances in which just medication or just talk therapy is needed. Once again, knowledge, clinical judgment, and the art of care are required to make these assessments.
In other words, recognizing and treating anxiety requires highly specialized training, which is why I thought the USPSTF recommendations raise a few critical questions.
Questions and concerns
One issue, of course, is the exclusion of those patients over age 65 because of a lack of “data.” Why such an exclusion? Does this mean that data are lacking for this age group?
The concept of using solely evidenced-based data in psychiatry is itself an interesting concept because our profession, like many other medical specialties, requires practitioners to use a combination of art and science. And much can be said either way about the clarity of accuracy in the diversity of issues that arise when treating emotional disorders.
When looking at the over-65 population, has anyone thought of clinical knowledge, judgment, experience, observation, and, of course, common sense?
Just consider the worry (a cardinal feature of anxiety) that besets people over 65 when it comes to issues such as retirement, financial security, “empty nesting,” physical health issues, decreased socialization that resulted from the COVID-19 pandemic, and the perpetual loss of relatives and friends.
In addition, as we age, anxiety can come simply from the loss of identity as active lifestyles decrease and the reality of nearing life’s end becomes more of a reality. It would seem that this population would benefit enormously from anxiety screening and possible treatment.
Another major concern is that the screening and potential treatment of patients is aimed at primary care physicians. Putting the sole responsibility of providing mental health care on these overworked PCPs defies common sense unless we’re okay with 1- to 2-minute assessments of mental health issues and no doubt, a pharmacology-only approach.
If this follows the same route as well-intentioned PCPs treating depression, where 5-minute medication management is far too common, the only proper diagnostic course – the in-depth interview necessary to make a proper diagnosis – is often missing.
For example, in depression alone, it takes psychiatric experience and time to differentiate a major depressive disorder from a bipolar depression and to provide the appropriate medication and treatment plan with careful follow-up. In my experience, this usually does not happen in the exceedingly overworked, time-driven day of a PCP.
Anxiety disorders and depression can prove debilitating, and if a PCP wants the responsibility of treatment, a mandated mental health program should be followed – just as here in New York, prescribers are mandated to take a pain control, opioid, and infection control CME course to keep our licenses up to date.
Short of mandating a mental health program for PCPs, it should be part of training and CME courses that Psychiatry is a super specialty, much like orthopedics and ophthalmology, and primary care physicians should never hesitate to make referrals to the specialist.
The big picture for me, and I hope for us all, is that the USPSTF has started things rolling by making it clear that PCPs and other health care clinicians need to screen for anxiety as a disabling disorder that is quite treatable.
This approach will help to advance the destigmatization of mental health disorders. But as result, with more patients diagnosed, there will be a need for more psychiatrists – and psychologists with PhDs or PsyDs – to fill the gaps in mental health care.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.
A very good thing happened this summer for patients with anxiety and the psychiatrists, psychologists, and other mental health professionals who provide treatment for them. The U.S. Preventive Services Task Force recommended anxiety screening for all adults younger than 65.
On the surface, this is a great recommendation for recognition and caring for those who deal with and suffer from an anxiety disorder or multiple anxiety disorders. Although the USPSTF recommendations are independent of the U.S. government and are not an official position of the Department of Health & Human Services, they are a wonderful start at recognizing the importance of mental health care.
After all, anxiety disorders are the most commonly experienced and diagnosed mental disorders, according to the DSM-5.
They range mainly from generalized anxiety disorder (GAD), to panic attacks and panic disorder, separation anxiety, specific type phobias (bridges, tunnels, insects, snakes, and the list goes on), to other phobias, including agoraphobia, social phobia, and of course, anxiety caused by medical conditions. GAD alone occurs in, at least, more than 3% of the population.
Those of us who have been treating anxiety disorders for decades recognize them as an issue affecting both mental and physical well-being, not only because of the emotional causes but the physical distress and illnesses that anxiety may precipitate or worsen.
For example, blood pressure– and heart-related issues, GI disorders, and musculoskeletal issues are just a few examples of how our bodies and organ systems are affected by anxiety. Just the momentary physical symptoms of tachycardia or the “runs” before an exam are fine examples of how anxiety may affect patients physically, and an ongoing, consistent anxiety is potentially more harmful.
In fact, a first panic attack or episode of generalized anxiety may be so serious that an emergency department or physician visit is necessary to rule out a heart attack, asthma, or breathing issues – even a hormone or thyroid emergency, or a cardiac arrhythmia. Panic attacks alone create a high number of ED visits.
Treatments mainly include medication management and a variety of psychotherapy techniques. Currently, the most preferred, first-choice medications are the SSRI antidepressants, which are Food and Drug Administration approved for anxiety as well. These include Zoloft (sertraline), Prozac/Sarafem (fluoxetine), Celexa (citalopram), and Lexapro (escitalopram).
For many years, benzodiazepines (that is, tranquillizers) such as Valium (diazepam), Ativan (lorazepam), and Klonopin/Rivotril (clonazepam) to name a few, were the mainstay of anxiety treatment, but they have proven addictive and may affect cognition and memory. As the current opioid epidemic has shown, when combined with opioids, benzodiazepines are a potentially lethal combination and when used, they need to be for shorter-term care and monitored very judiciously.
It should be noted that after ongoing long-term use of an SSRI for anxiety or depression, it should not be stopped abruptly, as a variety of physical symptoms (for example, flu-like symptoms) may occur.
Benefits of nonmedicinal therapies
There are a variety of talk therapies, from dynamic psychotherapies to cognitive-behavioral therapies (CBT), plus relaxation techniques and guided imagery that have all had a good amount of success in treating generalized anxiety, panic disorder, as well as various types of phobias.
When medications are stopped, the anxiety symptoms may well return. But when using nonmedicinal therapies, clinicians have discovered that when patients develop a new perspective on the anxiety problem or have a new technique to treat anxiety, it may well be long lasting.
For me, using CBT, relaxation techniques, hypnosis, and guided imagery has been very successful in treating anxiety disorders with long-lasting results. Once a person learns to relax, whether it’s from deep breathing exercises, hypnosis (which is not sleep), mindfulness, or meditation, a strategy of guided imagery can be taught, which allows a person to practice as well as control their anxiety as a lifetime process. For example, I like imagining a large movie screen to desensitize and project anxieties.
In many instances, a combination of a medication and a talk therapy approach works best, but there are an equal number of instances in which just medication or just talk therapy is needed. Once again, knowledge, clinical judgment, and the art of care are required to make these assessments.
In other words, recognizing and treating anxiety requires highly specialized training, which is why I thought the USPSTF recommendations raise a few critical questions.
Questions and concerns
One issue, of course, is the exclusion of those patients over age 65 because of a lack of “data.” Why such an exclusion? Does this mean that data are lacking for this age group?
The concept of using solely evidenced-based data in psychiatry is itself an interesting concept because our profession, like many other medical specialties, requires practitioners to use a combination of art and science. And much can be said either way about the clarity of accuracy in the diversity of issues that arise when treating emotional disorders.
When looking at the over-65 population, has anyone thought of clinical knowledge, judgment, experience, observation, and, of course, common sense?
Just consider the worry (a cardinal feature of anxiety) that besets people over 65 when it comes to issues such as retirement, financial security, “empty nesting,” physical health issues, decreased socialization that resulted from the COVID-19 pandemic, and the perpetual loss of relatives and friends.
In addition, as we age, anxiety can come simply from the loss of identity as active lifestyles decrease and the reality of nearing life’s end becomes more of a reality. It would seem that this population would benefit enormously from anxiety screening and possible treatment.
Another major concern is that the screening and potential treatment of patients is aimed at primary care physicians. Putting the sole responsibility of providing mental health care on these overworked PCPs defies common sense unless we’re okay with 1- to 2-minute assessments of mental health issues and no doubt, a pharmacology-only approach.
If this follows the same route as well-intentioned PCPs treating depression, where 5-minute medication management is far too common, the only proper diagnostic course – the in-depth interview necessary to make a proper diagnosis – is often missing.
For example, in depression alone, it takes psychiatric experience and time to differentiate a major depressive disorder from a bipolar depression and to provide the appropriate medication and treatment plan with careful follow-up. In my experience, this usually does not happen in the exceedingly overworked, time-driven day of a PCP.
Anxiety disorders and depression can prove debilitating, and if a PCP wants the responsibility of treatment, a mandated mental health program should be followed – just as here in New York, prescribers are mandated to take a pain control, opioid, and infection control CME course to keep our licenses up to date.
Short of mandating a mental health program for PCPs, it should be part of training and CME courses that Psychiatry is a super specialty, much like orthopedics and ophthalmology, and primary care physicians should never hesitate to make referrals to the specialist.
The big picture for me, and I hope for us all, is that the USPSTF has started things rolling by making it clear that PCPs and other health care clinicians need to screen for anxiety as a disabling disorder that is quite treatable.
This approach will help to advance the destigmatization of mental health disorders. But as result, with more patients diagnosed, there will be a need for more psychiatrists – and psychologists with PhDs or PsyDs – to fill the gaps in mental health care.
Dr. London is a practicing psychiatrist and has been a newspaper columnist for 35 years, specializing in and writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.