User login
Catch-and-Treat Strategy Identifies Undiagnosed Asthma and COPD
SAN DIEGO — You can’t treat patients if you can’t find them. But as investigators in a randomized controlled trial showed, a case-finding method based on spirometry results can identify individuals in the community with undiagnosed chronic obstructive pulmonary disease (COPD) or asthma whose lives could be significantly improved with proper care.
“By diagnosing people early and treating them intensively, you can really improve their quality of life,” said lead investigator Shawn D. Aaron, MD, from the Ottawa Hospital Research Institute and University of Ottawa, Ontario, Canada.
Even those patients in the study who were randomly assigned to receive care from a general practice physician had improvements in lung function and quality of life, although on a smaller scale than patients assigned to a specialty team, Dr. Aaron said at the American Thoracic Society’s international conference.
He reported results of the study in a late-breaking oral abstract session. The study findings were also published online in The New England Journal of Medicine.
Undiagnosed diseases
“The simple problem is that 70% of individuals with asthma or COPD are likely undiagnosed,” Dr. Aaron said.
He noted that the 2007-2012 US National Health and Nutritional Examination Survey found obstructive lung disease in 13% of randomly selected US adults, but 71% of these people had never been diagnosed with asthma or COPD.
“So our questions were in this study: One, can we find adults with undiagnosed asthma or COPD in the community? The second question was: If we find them, are they sick? And the third and most important question was: Can we treat them early and improve their health outcomes?” he said.
Asthma and COPD both present with similar respiratory symptoms, including dyspnea, cough, wheeze, and/or chest tightness, and the two conditions share expiratory airflow obstruction as a common physiologic impairment that can be detected with spirometry.
Study details
To identify participants, the investigators hired a commercial survey firm to contact households asking whether any member aged 18 years or older had respiratory symptoms such as shortness of breath, wheezing, increased mucus or sputum production, or prolonged cough in the past 6 months. Those who responded yes were then contacted by a trial coordinator, and the symptomatic household member was asked to complete the Asthma Screening Questionnaire over the phone. Participants aged 60 years or older and those younger than 60 years with a score of 6 or higher on the asthma screen also completed the COPD Diagnostic Questionnaire.
Those with a score of 6 or higher on the asthma screen or 20 or higher on the COPD screen were invited to undergo spirometry at a trial site.
The investigators ultimately identified 508 adults with undiagnosed asthma or COPD and randomly assigned them on an equal basis to an intervention group (253 patients) or control group (255 patients).
In the intervention group treatment was provided by a study pulmonologist and asthma-COPD educator who started guideline-based care. Patients were prescribed inhalers and were taught how to use them, and many were given action plans that included smoking cessation aids, exercise and weight counseling, and vaccinations against influenza and pneumonia.
Participants assigned to the control group would receive usual care provided by their primary care practitioner.
Improvements abound
During the 12 months of the study, 92% of patients in the intervention group and 60% in the control group were started on new medications for their condition.
Only 13.4% of those in the intervention group received either no respiratory treatments or a short-acting beta 2 agonist only during the entire trial period compared with 49.8% of controls, “so the usual care arm was undertreated relative to the intervention arm, and because of that under-treatment we saw a tremendous difference in the primary outcome,” Dr. Aaron said.
The primary outcome, the annualized rate of patient-initiated healthcare utilization for respiratory illness, was significantly lower in the intervention group, translating into an incidence rate ratio of 0.48 (P < .001).
Secondary outcomes were also better in the intervention group. For example, total scores on the St. George Respiratory Questionnaire (SGRQ) declined by 10.2 points from baseline in intervention group compared with a 6.8-point drop in the usual-care group. The mean difference was 3.5 points (P = .009). Lower scores on the 0-100 SGRQ scale indicate better health status.
Similarly, total scores on the COPD Assessment Test, a scale of 0-40 with lower scores indicating better health, declined by 3.8 points and 2.6 points, respectively, over 12 months, for a mean difference of 1.3 points (P = .03).
In addition, those in the intervention arm had a 119-mL improvement in forced expiratory volume in 1 second over the 12 months of the study compared with only a 22-mL improvement in the usual-care group.
Translatable results?
Dr. Aaron acknowledged that the investigators could have chosen to keep those who were assigned to the control group unaware of their diagnosis during the study but because all patients enrolled were symptomatic, it would have been unethical to do so. All participants were informed of their diagnosis at randomization, and the information was conveyed to each patient’s primary care practitioner as well.
In fact, many patients in the control group decided to seek treatment for either asthma or COPD after learning of their diagnosis, which may have contributed to improved outcomes in the control arm, he said.
“What this means is if you make the diagnosis early in the community, and at least have them see a primary care practitioner, they will improve their quality of life and their health status,” he concluded.
Ravi Kalhan, MD, MS, from the Northwestern University Feinberg School Of Medicine in Chicago, who co-moderated the session but was not involved in the study, said in an interview that the case-finding model used in the trial would be difficult to replicate elsewhere.
“This idea of seeking out undiagnosed people by doing spirometry, so-called ‘case finding’ as they described it, testing highly symptomatic people with spirometry, is really challenging in the US, because symptoms are not collected proactively very much,” he said.
Persons with acute respiratory symptoms in the US typically seek healthcare at urgent-care clinics or have unscheduled visits with their primary care physicians, “and by all accounts those people should have spirometry, but they just don’t in the US, as best as I can tell,” he added.
He agreed that getting patients to a specialist can result in better outcomes but said that implementing a systematic approach such as the one described in the study would be extremely difficult in the fragmented US healthcare system.
Dr. Kalhan’s co-moderator, Nuala J. Meyer, MD, MS, from the Hospital of the University of Pennsylvania, Philadelphia, told Chest Physician that “it was interesting that even those who were not in the intervention group but had these details passed on to their primary care physicians still had improvements,” and that it would be beneficial if primary care practitioners were routinely informed about the results of urgent care visits.
She added, however, that in the US the flow of information between urgent care clinics, primary care offices, and specialty clinics is problematic, suggesting that symptomatic patients may not always receive the additional care that they need.
The study was supported by the Canadian Institutes of Health Research. Dr. Aaron, Dr. Kalhan, and Dr. Meyer all reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
SAN DIEGO — You can’t treat patients if you can’t find them. But as investigators in a randomized controlled trial showed, a case-finding method based on spirometry results can identify individuals in the community with undiagnosed chronic obstructive pulmonary disease (COPD) or asthma whose lives could be significantly improved with proper care.
“By diagnosing people early and treating them intensively, you can really improve their quality of life,” said lead investigator Shawn D. Aaron, MD, from the Ottawa Hospital Research Institute and University of Ottawa, Ontario, Canada.
Even those patients in the study who were randomly assigned to receive care from a general practice physician had improvements in lung function and quality of life, although on a smaller scale than patients assigned to a specialty team, Dr. Aaron said at the American Thoracic Society’s international conference.
He reported results of the study in a late-breaking oral abstract session. The study findings were also published online in The New England Journal of Medicine.
Undiagnosed diseases
“The simple problem is that 70% of individuals with asthma or COPD are likely undiagnosed,” Dr. Aaron said.
He noted that the 2007-2012 US National Health and Nutritional Examination Survey found obstructive lung disease in 13% of randomly selected US adults, but 71% of these people had never been diagnosed with asthma or COPD.
“So our questions were in this study: One, can we find adults with undiagnosed asthma or COPD in the community? The second question was: If we find them, are they sick? And the third and most important question was: Can we treat them early and improve their health outcomes?” he said.
Asthma and COPD both present with similar respiratory symptoms, including dyspnea, cough, wheeze, and/or chest tightness, and the two conditions share expiratory airflow obstruction as a common physiologic impairment that can be detected with spirometry.
Study details
To identify participants, the investigators hired a commercial survey firm to contact households asking whether any member aged 18 years or older had respiratory symptoms such as shortness of breath, wheezing, increased mucus or sputum production, or prolonged cough in the past 6 months. Those who responded yes were then contacted by a trial coordinator, and the symptomatic household member was asked to complete the Asthma Screening Questionnaire over the phone. Participants aged 60 years or older and those younger than 60 years with a score of 6 or higher on the asthma screen also completed the COPD Diagnostic Questionnaire.
Those with a score of 6 or higher on the asthma screen or 20 or higher on the COPD screen were invited to undergo spirometry at a trial site.
The investigators ultimately identified 508 adults with undiagnosed asthma or COPD and randomly assigned them on an equal basis to an intervention group (253 patients) or control group (255 patients).
In the intervention group treatment was provided by a study pulmonologist and asthma-COPD educator who started guideline-based care. Patients were prescribed inhalers and were taught how to use them, and many were given action plans that included smoking cessation aids, exercise and weight counseling, and vaccinations against influenza and pneumonia.
Participants assigned to the control group would receive usual care provided by their primary care practitioner.
Improvements abound
During the 12 months of the study, 92% of patients in the intervention group and 60% in the control group were started on new medications for their condition.
Only 13.4% of those in the intervention group received either no respiratory treatments or a short-acting beta 2 agonist only during the entire trial period compared with 49.8% of controls, “so the usual care arm was undertreated relative to the intervention arm, and because of that under-treatment we saw a tremendous difference in the primary outcome,” Dr. Aaron said.
The primary outcome, the annualized rate of patient-initiated healthcare utilization for respiratory illness, was significantly lower in the intervention group, translating into an incidence rate ratio of 0.48 (P < .001).
Secondary outcomes were also better in the intervention group. For example, total scores on the St. George Respiratory Questionnaire (SGRQ) declined by 10.2 points from baseline in intervention group compared with a 6.8-point drop in the usual-care group. The mean difference was 3.5 points (P = .009). Lower scores on the 0-100 SGRQ scale indicate better health status.
Similarly, total scores on the COPD Assessment Test, a scale of 0-40 with lower scores indicating better health, declined by 3.8 points and 2.6 points, respectively, over 12 months, for a mean difference of 1.3 points (P = .03).
In addition, those in the intervention arm had a 119-mL improvement in forced expiratory volume in 1 second over the 12 months of the study compared with only a 22-mL improvement in the usual-care group.
Translatable results?
Dr. Aaron acknowledged that the investigators could have chosen to keep those who were assigned to the control group unaware of their diagnosis during the study but because all patients enrolled were symptomatic, it would have been unethical to do so. All participants were informed of their diagnosis at randomization, and the information was conveyed to each patient’s primary care practitioner as well.
In fact, many patients in the control group decided to seek treatment for either asthma or COPD after learning of their diagnosis, which may have contributed to improved outcomes in the control arm, he said.
“What this means is if you make the diagnosis early in the community, and at least have them see a primary care practitioner, they will improve their quality of life and their health status,” he concluded.
Ravi Kalhan, MD, MS, from the Northwestern University Feinberg School Of Medicine in Chicago, who co-moderated the session but was not involved in the study, said in an interview that the case-finding model used in the trial would be difficult to replicate elsewhere.
“This idea of seeking out undiagnosed people by doing spirometry, so-called ‘case finding’ as they described it, testing highly symptomatic people with spirometry, is really challenging in the US, because symptoms are not collected proactively very much,” he said.
Persons with acute respiratory symptoms in the US typically seek healthcare at urgent-care clinics or have unscheduled visits with their primary care physicians, “and by all accounts those people should have spirometry, but they just don’t in the US, as best as I can tell,” he added.
He agreed that getting patients to a specialist can result in better outcomes but said that implementing a systematic approach such as the one described in the study would be extremely difficult in the fragmented US healthcare system.
Dr. Kalhan’s co-moderator, Nuala J. Meyer, MD, MS, from the Hospital of the University of Pennsylvania, Philadelphia, told Chest Physician that “it was interesting that even those who were not in the intervention group but had these details passed on to their primary care physicians still had improvements,” and that it would be beneficial if primary care practitioners were routinely informed about the results of urgent care visits.
She added, however, that in the US the flow of information between urgent care clinics, primary care offices, and specialty clinics is problematic, suggesting that symptomatic patients may not always receive the additional care that they need.
The study was supported by the Canadian Institutes of Health Research. Dr. Aaron, Dr. Kalhan, and Dr. Meyer all reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
SAN DIEGO — You can’t treat patients if you can’t find them. But as investigators in a randomized controlled trial showed, a case-finding method based on spirometry results can identify individuals in the community with undiagnosed chronic obstructive pulmonary disease (COPD) or asthma whose lives could be significantly improved with proper care.
“By diagnosing people early and treating them intensively, you can really improve their quality of life,” said lead investigator Shawn D. Aaron, MD, from the Ottawa Hospital Research Institute and University of Ottawa, Ontario, Canada.
Even those patients in the study who were randomly assigned to receive care from a general practice physician had improvements in lung function and quality of life, although on a smaller scale than patients assigned to a specialty team, Dr. Aaron said at the American Thoracic Society’s international conference.
He reported results of the study in a late-breaking oral abstract session. The study findings were also published online in The New England Journal of Medicine.
Undiagnosed diseases
“The simple problem is that 70% of individuals with asthma or COPD are likely undiagnosed,” Dr. Aaron said.
He noted that the 2007-2012 US National Health and Nutritional Examination Survey found obstructive lung disease in 13% of randomly selected US adults, but 71% of these people had never been diagnosed with asthma or COPD.
“So our questions were in this study: One, can we find adults with undiagnosed asthma or COPD in the community? The second question was: If we find them, are they sick? And the third and most important question was: Can we treat them early and improve their health outcomes?” he said.
Asthma and COPD both present with similar respiratory symptoms, including dyspnea, cough, wheeze, and/or chest tightness, and the two conditions share expiratory airflow obstruction as a common physiologic impairment that can be detected with spirometry.
Study details
To identify participants, the investigators hired a commercial survey firm to contact households asking whether any member aged 18 years or older had respiratory symptoms such as shortness of breath, wheezing, increased mucus or sputum production, or prolonged cough in the past 6 months. Those who responded yes were then contacted by a trial coordinator, and the symptomatic household member was asked to complete the Asthma Screening Questionnaire over the phone. Participants aged 60 years or older and those younger than 60 years with a score of 6 or higher on the asthma screen also completed the COPD Diagnostic Questionnaire.
Those with a score of 6 or higher on the asthma screen or 20 or higher on the COPD screen were invited to undergo spirometry at a trial site.
The investigators ultimately identified 508 adults with undiagnosed asthma or COPD and randomly assigned them on an equal basis to an intervention group (253 patients) or control group (255 patients).
In the intervention group treatment was provided by a study pulmonologist and asthma-COPD educator who started guideline-based care. Patients were prescribed inhalers and were taught how to use them, and many were given action plans that included smoking cessation aids, exercise and weight counseling, and vaccinations against influenza and pneumonia.
Participants assigned to the control group would receive usual care provided by their primary care practitioner.
Improvements abound
During the 12 months of the study, 92% of patients in the intervention group and 60% in the control group were started on new medications for their condition.
Only 13.4% of those in the intervention group received either no respiratory treatments or a short-acting beta 2 agonist only during the entire trial period compared with 49.8% of controls, “so the usual care arm was undertreated relative to the intervention arm, and because of that under-treatment we saw a tremendous difference in the primary outcome,” Dr. Aaron said.
The primary outcome, the annualized rate of patient-initiated healthcare utilization for respiratory illness, was significantly lower in the intervention group, translating into an incidence rate ratio of 0.48 (P < .001).
Secondary outcomes were also better in the intervention group. For example, total scores on the St. George Respiratory Questionnaire (SGRQ) declined by 10.2 points from baseline in intervention group compared with a 6.8-point drop in the usual-care group. The mean difference was 3.5 points (P = .009). Lower scores on the 0-100 SGRQ scale indicate better health status.
Similarly, total scores on the COPD Assessment Test, a scale of 0-40 with lower scores indicating better health, declined by 3.8 points and 2.6 points, respectively, over 12 months, for a mean difference of 1.3 points (P = .03).
In addition, those in the intervention arm had a 119-mL improvement in forced expiratory volume in 1 second over the 12 months of the study compared with only a 22-mL improvement in the usual-care group.
Translatable results?
Dr. Aaron acknowledged that the investigators could have chosen to keep those who were assigned to the control group unaware of their diagnosis during the study but because all patients enrolled were symptomatic, it would have been unethical to do so. All participants were informed of their diagnosis at randomization, and the information was conveyed to each patient’s primary care practitioner as well.
In fact, many patients in the control group decided to seek treatment for either asthma or COPD after learning of their diagnosis, which may have contributed to improved outcomes in the control arm, he said.
“What this means is if you make the diagnosis early in the community, and at least have them see a primary care practitioner, they will improve their quality of life and their health status,” he concluded.
Ravi Kalhan, MD, MS, from the Northwestern University Feinberg School Of Medicine in Chicago, who co-moderated the session but was not involved in the study, said in an interview that the case-finding model used in the trial would be difficult to replicate elsewhere.
“This idea of seeking out undiagnosed people by doing spirometry, so-called ‘case finding’ as they described it, testing highly symptomatic people with spirometry, is really challenging in the US, because symptoms are not collected proactively very much,” he said.
Persons with acute respiratory symptoms in the US typically seek healthcare at urgent-care clinics or have unscheduled visits with their primary care physicians, “and by all accounts those people should have spirometry, but they just don’t in the US, as best as I can tell,” he added.
He agreed that getting patients to a specialist can result in better outcomes but said that implementing a systematic approach such as the one described in the study would be extremely difficult in the fragmented US healthcare system.
Dr. Kalhan’s co-moderator, Nuala J. Meyer, MD, MS, from the Hospital of the University of Pennsylvania, Philadelphia, told Chest Physician that “it was interesting that even those who were not in the intervention group but had these details passed on to their primary care physicians still had improvements,” and that it would be beneficial if primary care practitioners were routinely informed about the results of urgent care visits.
She added, however, that in the US the flow of information between urgent care clinics, primary care offices, and specialty clinics is problematic, suggesting that symptomatic patients may not always receive the additional care that they need.
The study was supported by the Canadian Institutes of Health Research. Dr. Aaron, Dr. Kalhan, and Dr. Meyer all reported having no relevant disclosures.
A version of this article appeared on Medscape.com.
Treating High Stage cSCC: Better Results With Mohs Surgery vs Wide Local Excision, Study Finds
PHOENIX — . The benefit was seen across all outcome measures, including rates of recurrence, metastasis, and mortality.
These data support Mohs surgery as being the preferred surgical treatment option for high-stage cSCC, commented lead author David M. Wang, MD, Mohs Micrographic Surgery and Dermatologic Oncology Fellow, at Harvard’s Brigham and Women’s Hospital (BWH)/Dana-Farber Cancer Institute, Boston. “We found that across all outcomes, high-stage cSCC treated with WLE had a roughly twofold greater risk for recurrence, metastasis, or disease-specific death compared to Mohs,” he said at the annual meeting of the American College of Mohs Surgery (ACMS), where he presented the results.
External validation using data from a multicenter cSCC research collaboration from 12 contributing sites from across the United States, as well as international sites, was also conducted. “We performed the external validation by comparing results of the BWH-only cohort, which was the primary study, with the full multicenter data and with the full multicenter data minus the BWH cohort, and the findings were nearly identical in all three analyses,” Dr. Wang said.
Although patients diagnosed with cSCC usually have good outcomes, high-stage disease is associated with a higher risk for recurrence, metastasis, and death. Both Mohs surgery and WLE are used to treat cSCC, but a comparison of outcomes has not been well established in the setting of high-stage cSCC. Comparing the two surgical strategies can be problematic, as both patient and/or tumor characteristics can make it difficult to determine which outcomes can be attributed solely to the treatment type.
Mohs Superior Across the Board
In the retrospective cohort study, Dr. Wang and colleagues aimed to compare the results of Mohs surgery and WLE in patients with high-stage cSCC (BWH Staging System T2b or T3) and used statistical methods to balance baseline patient and tumor characteristics.
“To control for confounding by indication — differences in baseline patient or tumor characteristics — that are associated with both the treatment received and outcomes, we used propensity score weighting so that the baseline characteristics were balanced in the two treatment groups,” Dr. Wang told this news organization. “This statistical method aims to simulate randomization in a randomized controlled trial such that any differences in outcomes after propensity score weighting is attributed solely to the treatment received.”
The study used electronic medical records from a single tertiary care academic institution, and 216 patients with high-stage cSCC who had undergone surgery from January 2000 to December 2019 were included in the analysis. The median follow-up time was 33.1 months.
They found that overall, the risk for all adverse outcomes was lower among patients who had undergone Mohs surgery than among those treated with WLE, with the following results: Rates of local recurrence (5-year CI, 10.8% vs 22.1%, respectively; P = .003), nodal metastasis (11.9% vs 19.3%; P = .04), distant metastasis (4.7% vs 9.0%; P = .09), any recurrence (17.0% vs 34.2%; P < .001), and disease-specific death (8.5% vs 20.3%; P = .001).
“The data supports Mohs surgery as the preferred surgical treatment option for high-stage cSCC in accordance with NCCN [National Comprehensive Cancer Network] guidelines for very high-risk cSCC,” Dr. Wang said. He pointed out that the terminology “very high risk” in NCCN equates to “high stage” in other staging systems (BWH T2b or higher, AJCC T3 or higher).
There is still “a substantial proportion” of patients with high-stage cSCC who are eligible for Mohs but are treated with WLE, he added. “Our hope is that these findings provide additional data to support Mohs as the standard of care for primary surgical treatment of high-stage cSCC.”
Supports Benefits of Mohs
Weighing in on the research, Thomas E. Rohrer, MD, a dermatologic surgeon in Chestnut Hill, Massachusetts, noted that this was an excellent study that demonstrates benefits of Mohs surgery over straight excision on essentially all outcomes investigated and measured.
“The data clearly shows that Mohs should be used whenever possible,” he said. “There are some patients and facilities that do not have access or timely access to Mohs, so they would likely proceed with standard wide local excision. Otherwise, if there is the capability to perform Mohs, it would be preferred,” he added.
“There is no benefit to a standard excision over Mohs,” Dr. Rohrer emphasized. “If a surgeon is not sure if they have attained clear margins, they could and often do take a little more tissue to be certain.”
Also asked to comment on the data, Chad L. Prather, MD, a dermatologist in Baton Rouge, Louisiana, said, “We know that Mohs has been used for cancers that are not highly staged and we know it’s better than WLE, but this study shows that it is beneficial for higher stage cancers.”
However, he cautioned that unlike early-stage cancers, where Mohs is usually a definitive treatment, with higher stage disease it is a starting point. “As a takeaway, Mohs is superior, but it needs to be followed through,” he said. “These patients need to be closely followed as they are at a high risk for recurrence and metastasis and may need to be worked up for lymph node involvement and need additional therapy going forward.”
Dr. Prather also pointed out that there are circumstances when WLE may be a more suitable treatment. “Mohs is not very good if there is bony involvement,” he said. “This most often happens when the lesion is on the scalp and has invaded the skull. WLE may still be the preferred choice.”
Additionally, Mohs is not the best choice if the tumor is broken into multiple segments. “In these cases, WLE may be preferred,” Dr. Prather added. “But overall, Mohs is one of the best tools we have, and it stands to reason that it works well for high-risk tumors, as this study shows.”
The study was independently supported. Dr. Wang reported no relevant financial relationships. Dr. Rohrer and Dr. Prather had no relevant disclosures.
A version of this article appeared on Medscape.com.
PHOENIX — . The benefit was seen across all outcome measures, including rates of recurrence, metastasis, and mortality.
These data support Mohs surgery as being the preferred surgical treatment option for high-stage cSCC, commented lead author David M. Wang, MD, Mohs Micrographic Surgery and Dermatologic Oncology Fellow, at Harvard’s Brigham and Women’s Hospital (BWH)/Dana-Farber Cancer Institute, Boston. “We found that across all outcomes, high-stage cSCC treated with WLE had a roughly twofold greater risk for recurrence, metastasis, or disease-specific death compared to Mohs,” he said at the annual meeting of the American College of Mohs Surgery (ACMS), where he presented the results.
External validation using data from a multicenter cSCC research collaboration from 12 contributing sites from across the United States, as well as international sites, was also conducted. “We performed the external validation by comparing results of the BWH-only cohort, which was the primary study, with the full multicenter data and with the full multicenter data minus the BWH cohort, and the findings were nearly identical in all three analyses,” Dr. Wang said.
Although patients diagnosed with cSCC usually have good outcomes, high-stage disease is associated with a higher risk for recurrence, metastasis, and death. Both Mohs surgery and WLE are used to treat cSCC, but a comparison of outcomes has not been well established in the setting of high-stage cSCC. Comparing the two surgical strategies can be problematic, as both patient and/or tumor characteristics can make it difficult to determine which outcomes can be attributed solely to the treatment type.
Mohs Superior Across the Board
In the retrospective cohort study, Dr. Wang and colleagues aimed to compare the results of Mohs surgery and WLE in patients with high-stage cSCC (BWH Staging System T2b or T3) and used statistical methods to balance baseline patient and tumor characteristics.
“To control for confounding by indication — differences in baseline patient or tumor characteristics — that are associated with both the treatment received and outcomes, we used propensity score weighting so that the baseline characteristics were balanced in the two treatment groups,” Dr. Wang told this news organization. “This statistical method aims to simulate randomization in a randomized controlled trial such that any differences in outcomes after propensity score weighting is attributed solely to the treatment received.”
The study used electronic medical records from a single tertiary care academic institution, and 216 patients with high-stage cSCC who had undergone surgery from January 2000 to December 2019 were included in the analysis. The median follow-up time was 33.1 months.
They found that overall, the risk for all adverse outcomes was lower among patients who had undergone Mohs surgery than among those treated with WLE, with the following results: Rates of local recurrence (5-year CI, 10.8% vs 22.1%, respectively; P = .003), nodal metastasis (11.9% vs 19.3%; P = .04), distant metastasis (4.7% vs 9.0%; P = .09), any recurrence (17.0% vs 34.2%; P < .001), and disease-specific death (8.5% vs 20.3%; P = .001).
“The data supports Mohs surgery as the preferred surgical treatment option for high-stage cSCC in accordance with NCCN [National Comprehensive Cancer Network] guidelines for very high-risk cSCC,” Dr. Wang said. He pointed out that the terminology “very high risk” in NCCN equates to “high stage” in other staging systems (BWH T2b or higher, AJCC T3 or higher).
There is still “a substantial proportion” of patients with high-stage cSCC who are eligible for Mohs but are treated with WLE, he added. “Our hope is that these findings provide additional data to support Mohs as the standard of care for primary surgical treatment of high-stage cSCC.”
Supports Benefits of Mohs
Weighing in on the research, Thomas E. Rohrer, MD, a dermatologic surgeon in Chestnut Hill, Massachusetts, noted that this was an excellent study that demonstrates benefits of Mohs surgery over straight excision on essentially all outcomes investigated and measured.
“The data clearly shows that Mohs should be used whenever possible,” he said. “There are some patients and facilities that do not have access or timely access to Mohs, so they would likely proceed with standard wide local excision. Otherwise, if there is the capability to perform Mohs, it would be preferred,” he added.
“There is no benefit to a standard excision over Mohs,” Dr. Rohrer emphasized. “If a surgeon is not sure if they have attained clear margins, they could and often do take a little more tissue to be certain.”
Also asked to comment on the data, Chad L. Prather, MD, a dermatologist in Baton Rouge, Louisiana, said, “We know that Mohs has been used for cancers that are not highly staged and we know it’s better than WLE, but this study shows that it is beneficial for higher stage cancers.”
However, he cautioned that unlike early-stage cancers, where Mohs is usually a definitive treatment, with higher stage disease it is a starting point. “As a takeaway, Mohs is superior, but it needs to be followed through,” he said. “These patients need to be closely followed as they are at a high risk for recurrence and metastasis and may need to be worked up for lymph node involvement and need additional therapy going forward.”
Dr. Prather also pointed out that there are circumstances when WLE may be a more suitable treatment. “Mohs is not very good if there is bony involvement,” he said. “This most often happens when the lesion is on the scalp and has invaded the skull. WLE may still be the preferred choice.”
Additionally, Mohs is not the best choice if the tumor is broken into multiple segments. “In these cases, WLE may be preferred,” Dr. Prather added. “But overall, Mohs is one of the best tools we have, and it stands to reason that it works well for high-risk tumors, as this study shows.”
The study was independently supported. Dr. Wang reported no relevant financial relationships. Dr. Rohrer and Dr. Prather had no relevant disclosures.
A version of this article appeared on Medscape.com.
PHOENIX — . The benefit was seen across all outcome measures, including rates of recurrence, metastasis, and mortality.
These data support Mohs surgery as being the preferred surgical treatment option for high-stage cSCC, commented lead author David M. Wang, MD, Mohs Micrographic Surgery and Dermatologic Oncology Fellow, at Harvard’s Brigham and Women’s Hospital (BWH)/Dana-Farber Cancer Institute, Boston. “We found that across all outcomes, high-stage cSCC treated with WLE had a roughly twofold greater risk for recurrence, metastasis, or disease-specific death compared to Mohs,” he said at the annual meeting of the American College of Mohs Surgery (ACMS), where he presented the results.
External validation using data from a multicenter cSCC research collaboration from 12 contributing sites from across the United States, as well as international sites, was also conducted. “We performed the external validation by comparing results of the BWH-only cohort, which was the primary study, with the full multicenter data and with the full multicenter data minus the BWH cohort, and the findings were nearly identical in all three analyses,” Dr. Wang said.
Although patients diagnosed with cSCC usually have good outcomes, high-stage disease is associated with a higher risk for recurrence, metastasis, and death. Both Mohs surgery and WLE are used to treat cSCC, but a comparison of outcomes has not been well established in the setting of high-stage cSCC. Comparing the two surgical strategies can be problematic, as both patient and/or tumor characteristics can make it difficult to determine which outcomes can be attributed solely to the treatment type.
Mohs Superior Across the Board
In the retrospective cohort study, Dr. Wang and colleagues aimed to compare the results of Mohs surgery and WLE in patients with high-stage cSCC (BWH Staging System T2b or T3) and used statistical methods to balance baseline patient and tumor characteristics.
“To control for confounding by indication — differences in baseline patient or tumor characteristics — that are associated with both the treatment received and outcomes, we used propensity score weighting so that the baseline characteristics were balanced in the two treatment groups,” Dr. Wang told this news organization. “This statistical method aims to simulate randomization in a randomized controlled trial such that any differences in outcomes after propensity score weighting is attributed solely to the treatment received.”
The study used electronic medical records from a single tertiary care academic institution, and 216 patients with high-stage cSCC who had undergone surgery from January 2000 to December 2019 were included in the analysis. The median follow-up time was 33.1 months.
They found that overall, the risk for all adverse outcomes was lower among patients who had undergone Mohs surgery than among those treated with WLE, with the following results: Rates of local recurrence (5-year CI, 10.8% vs 22.1%, respectively; P = .003), nodal metastasis (11.9% vs 19.3%; P = .04), distant metastasis (4.7% vs 9.0%; P = .09), any recurrence (17.0% vs 34.2%; P < .001), and disease-specific death (8.5% vs 20.3%; P = .001).
“The data supports Mohs surgery as the preferred surgical treatment option for high-stage cSCC in accordance with NCCN [National Comprehensive Cancer Network] guidelines for very high-risk cSCC,” Dr. Wang said. He pointed out that the terminology “very high risk” in NCCN equates to “high stage” in other staging systems (BWH T2b or higher, AJCC T3 or higher).
There is still “a substantial proportion” of patients with high-stage cSCC who are eligible for Mohs but are treated with WLE, he added. “Our hope is that these findings provide additional data to support Mohs as the standard of care for primary surgical treatment of high-stage cSCC.”
Supports Benefits of Mohs
Weighing in on the research, Thomas E. Rohrer, MD, a dermatologic surgeon in Chestnut Hill, Massachusetts, noted that this was an excellent study that demonstrates benefits of Mohs surgery over straight excision on essentially all outcomes investigated and measured.
“The data clearly shows that Mohs should be used whenever possible,” he said. “There are some patients and facilities that do not have access or timely access to Mohs, so they would likely proceed with standard wide local excision. Otherwise, if there is the capability to perform Mohs, it would be preferred,” he added.
“There is no benefit to a standard excision over Mohs,” Dr. Rohrer emphasized. “If a surgeon is not sure if they have attained clear margins, they could and often do take a little more tissue to be certain.”
Also asked to comment on the data, Chad L. Prather, MD, a dermatologist in Baton Rouge, Louisiana, said, “We know that Mohs has been used for cancers that are not highly staged and we know it’s better than WLE, but this study shows that it is beneficial for higher stage cancers.”
However, he cautioned that unlike early-stage cancers, where Mohs is usually a definitive treatment, with higher stage disease it is a starting point. “As a takeaway, Mohs is superior, but it needs to be followed through,” he said. “These patients need to be closely followed as they are at a high risk for recurrence and metastasis and may need to be worked up for lymph node involvement and need additional therapy going forward.”
Dr. Prather also pointed out that there are circumstances when WLE may be a more suitable treatment. “Mohs is not very good if there is bony involvement,” he said. “This most often happens when the lesion is on the scalp and has invaded the skull. WLE may still be the preferred choice.”
Additionally, Mohs is not the best choice if the tumor is broken into multiple segments. “In these cases, WLE may be preferred,” Dr. Prather added. “But overall, Mohs is one of the best tools we have, and it stands to reason that it works well for high-risk tumors, as this study shows.”
The study was independently supported. Dr. Wang reported no relevant financial relationships. Dr. Rohrer and Dr. Prather had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ACMS 2024
Pulmonary telerehabilitation for COPD: Promising, but more data needed
As COVID-19 cedes its pandemic-scale status to the past, its wake is revealing surprises and raising questions, particularly in relation to pulmonary medicine. The need for isolation at COVID’s outset kept many millions at home, creating conditions favorable for the rapid expansion of technologies that were taken up quickly in telehealth applications. The need was overwhelming. But just how effective telehealth actually is at replacing on-site programs for COPD pulmonary rehab has remained a research challenge, although results from early studies show unmistakable value. Creating conditions conducive to research into the strengths and weaknesses of pulmonary rehab, and determining how research can be applied effectively, remain formidable challenges.
Early studies of telehealth pulmonary rehabilitation have not uncovered any glaring erosion of pulmonary rehabilitation’s well-established benefits. But, at the same time, the relatively young field of pulmonary telerehabilitation for chronic obstructive pulmonary disease (COPD) has lacked coordinated efforts to determine its key practices and the instruments for measuring them, both basic elements for pursuing research questions.
A 2021 American Thoracic Society workshop report (AE Holland, https://doi.org/10.1513/AnnalsATS.202102-146ST) identified essential components of a pulmonary rehabilitation model through an online Delphi process involving about 50 international experts. Components ultimately included those with median scores of 2 or higher (strongly agree or agree that the item is essential) and high consensus (interquartile range, 0). Thirteen essential components fit into four categories (Patient Assessment, Program Components, Method of Delivery and Quality Assurance). The Patient Assessment category included seven items: (1) An initial center-based assessment by a health care professional, (2) An exercise test at the time of assessment, (3) A field exercise test, (4) Quality of life measure, (5) Dyspnea assessment, (6) Nutritional status evaluation, and (7) Occupational status evaluation. The Program Components: (8) Endurance training and (9) Resistance training). The Method of Delivery: (10) An exercise program that is individually prescribed, (11) An exercise program that is individually progressed, and (12) Team includes a health care professional with experience in exercise prescription and progression. The single Quality Assurance item: (13) Health care professionals are trained to deliver the components of the model that is deployed.
Cochrane Library review
2021 “Intervention Review” (“Telerehabilitation for chronic respiratory disease,” https://doi.org/10.1002/14651858.CD013040.pub2). Using their own databases (eg, Cochrane Airways Trials Register) and others, the authors included controlled trials published up to November 30, 2020 with at least 50% of the rehabilitation delivered by telerehabilitation. The authors’ analysis of 15 studies (with 32 reports) including 1904 participants (99% with COPD): “There was probably little or no difference between telerehabilitation and in-person pulmonary rehabilitation for exercise capacity measured as 6-Minute Walking Distance (mean difference 0.06 meters (m), 95% confidence interval (CI) -10.82 m to 10.94 m).” They reached the same conclusion for quality of life, and for breathlessness. Completion of rehabilitation programs, however, was more likely with telerehabilitation at 93% versus 70% for in-person rehabilitation. No adverse effects of telerehabilitation were observed over and above those for in-person or no rehabilitation. An obvious limitation of the findings is that the studies all pre-date COVID-19, which would have introduced very significant disincentives for in-person rehabilitation completion.
An older (2016) international randomized controlled study (Zanaboni et al, https://doi.org/10.1186/s12890-016-0288-z) comparing long-term telerehabilitation or unsupervised treadmill training at home with standard care included 120 participants with COPD and had 2-years of follow-up. Telerehabilitation consisted of individualized treadmill training at home. Participants had scheduled exercise sessions supervised by a physiotherapist via videoconferencing following a standardized protocol. Participants in the unsupervised training group were provided with a treadmill only to perform unsupervised exercise at home. They also received an exercise booklet, a paper exercise diary to record their training sessions, and an individualized training program but without regular review or progression of the program. For the primary outcomes of combined hospitalizations and emergency department presentations, incidence rate of hospitalizations and emergency department presentations was lower with telerehabilitation (1.18 events per person-year; 95% confidence interval [CI], 0.94–1.46) and with unsupervised training group (1.14; 95% CI, 0.92–1.41) than in the control group (1.88; 95% CI, 1.58–2.21; P < .001 compared with intervention groups). Both training groups had better health status at 1-year, and achieved and maintained clinically significant improvements in exercise capacity.
Access to pulmonary rehabilitation
Continuing evidence of clear telerehabilitation benefits is good news, especially in the light of impediments to attendance at in-clinic programs. Although the COVID-provoked disincentives have been diminishing, persisting access issues remain for substantial portions of eligible populations, according to a recent (2024) cross-sectional study (PA Kahn, WA Mathis, doi:10.1001/jamanetworkopen.2023.54867) looking at travel time to pulmonary rehabilitation programs as a marker for pulmonary rehabilitation access. The report, based on US Census designations (lower 48 states and Washington, D.C.) found that while 80.3% of the population lives in urban or suburban areas within a 30-minute drive of a pulmonary rehabilitation program, travel time exceeds that in rural and other sparsely populated areas with more than 14 million people residing in areas demanding more than 1-hour for travel. A further analysis showed also that nearly 30% of American Indian and Alaska Native populations live more than 60 minutes from a pulmonary rehabilitation program.
Aside from the obvious restraints for homebound patients or those lacking transportation or who need medical transport, other common impediments inhibit on-site pulmonary rehabilitation attendance, said Corinne Young, MSN, FNP-C, FCCP. Ms. Young is the director of Advance Practice Provider and Clinical Services for Colorado Springs Pulmonary Consultants, president and founder of the Association of Pulmonary Advance Practice Providers, and a member of the CHEST Physician Editorial Board. “I have some patients who say ‘There’s no way I could do onsite pulmonary rehab because of my knee — or back, or shoulder.’ But in their own home environment they may feel more comfortable. They may be willing to try new things at their own pace, whereas for them a program may feel too regimented.” For others, Ms. Young said, aspects of a formal program are a clear plus factor. “They love to hear their progress at the end of — say a 12-week program — where their virtual respiratory therapist records and reports to them their six-minute walk and other test results. Feedback is a great reinforcer.” Quality of life improvements, Ms. Young commented, were one of the very impressive benefits that appeared in the initial studies of pulmonary rehabilitation for COPD patients. “Being patient-centric, you want to improve quality of life for them as much as possible and we see telerehabilitation as a great opportunity for many,” she added.
“I would like to see head-to-head data on outpatient versus at-home pulmonary rehabilitation on hospitalizations, time to exacerbation and, of course, mortality. We have all that for outpatient rehab, but it would be great to be able to compare them. Knowing that would influence what we recommend, especially for patients who could go either way. Also, you have to assess their motivation and discipline to know who might be more appropriate for unsupervised pulmonary rehabilitation.”
The current reality for Ms. Young is that in her Colorado Springs vicinity, where both in-patient programs are only 15 minutes apart, she knows of no telerehabilitation programs being offered. While there are contract telerehabilitation providers, Young said, and her organization (The Association of Pulmonary Advanced Practice Providers) has been approached by one, none are licensed in Colorado, and telerehabilitation is not a billable service.
“As of yet, I’m not aware of any telemedicine pulmonary rehab available at our institution,” said pulmonologist Mary Jo S. Farmer, MD, PhD, FCCP, Associate Professor of Medicine at UMass Chan Medical School – Baystate, Springfield, MA, and a member of the CHEST Physician Editorial Board. A brief internet search identified a telerehabilitation contract provider available only in Arizona.
Reimbursement will also be a foundational concern, Ms. Young commented. While a physician, nurse practitioner, or physician virtual visit for education may be billable, telerehabilitation reimbursement is new territory. “How that all is going to work out is a big unknown piece,” she said.
Minimal components
Effective pulmonary telerehabilitation programs, Ms. Young said, need to provide exercise with an aerobic device, either a treadmill, a stationary bike or even a Cubii-type under desk foot pedal/elliptical machine, and some resistance training (elastic bands, or weights, for example). “But 50% of pulmonary rehabilitation is education about breathing techniques, purse-lip breathing, and pulmonary nutrition.” Also essential: one-on-one discussion with a qualified medical practitioner who checks on oximeter use, inhaler technique, and titrating oxygen therapy. “At our elevation of 6500 feet, most of our patients are on that.” Optimal frequency of encounters between providers and remote patients has to be elucidated by future research, Ms. Young said.
Ms. Young commented further, “With outpatient pulmonary rehabilitation there often isn’t a lot of one-on-one, but rather a big group of people exercising at the same time. I think actually there may be the potential to have more individualization with pulmonary telerehabilitation. But the barriers, the reimbursement/financial part, and the red tape and bureaucracy have to be worked on.”
As COVID-19 cedes its pandemic-scale status to the past, its wake is revealing surprises and raising questions, particularly in relation to pulmonary medicine. The need for isolation at COVID’s outset kept many millions at home, creating conditions favorable for the rapid expansion of technologies that were taken up quickly in telehealth applications. The need was overwhelming. But just how effective telehealth actually is at replacing on-site programs for COPD pulmonary rehab has remained a research challenge, although results from early studies show unmistakable value. Creating conditions conducive to research into the strengths and weaknesses of pulmonary rehab, and determining how research can be applied effectively, remain formidable challenges.
Early studies of telehealth pulmonary rehabilitation have not uncovered any glaring erosion of pulmonary rehabilitation’s well-established benefits. But, at the same time, the relatively young field of pulmonary telerehabilitation for chronic obstructive pulmonary disease (COPD) has lacked coordinated efforts to determine its key practices and the instruments for measuring them, both basic elements for pursuing research questions.
A 2021 American Thoracic Society workshop report (AE Holland, https://doi.org/10.1513/AnnalsATS.202102-146ST) identified essential components of a pulmonary rehabilitation model through an online Delphi process involving about 50 international experts. Components ultimately included those with median scores of 2 or higher (strongly agree or agree that the item is essential) and high consensus (interquartile range, 0). Thirteen essential components fit into four categories (Patient Assessment, Program Components, Method of Delivery and Quality Assurance). The Patient Assessment category included seven items: (1) An initial center-based assessment by a health care professional, (2) An exercise test at the time of assessment, (3) A field exercise test, (4) Quality of life measure, (5) Dyspnea assessment, (6) Nutritional status evaluation, and (7) Occupational status evaluation. The Program Components: (8) Endurance training and (9) Resistance training). The Method of Delivery: (10) An exercise program that is individually prescribed, (11) An exercise program that is individually progressed, and (12) Team includes a health care professional with experience in exercise prescription and progression. The single Quality Assurance item: (13) Health care professionals are trained to deliver the components of the model that is deployed.
Cochrane Library review
2021 “Intervention Review” (“Telerehabilitation for chronic respiratory disease,” https://doi.org/10.1002/14651858.CD013040.pub2). Using their own databases (eg, Cochrane Airways Trials Register) and others, the authors included controlled trials published up to November 30, 2020 with at least 50% of the rehabilitation delivered by telerehabilitation. The authors’ analysis of 15 studies (with 32 reports) including 1904 participants (99% with COPD): “There was probably little or no difference between telerehabilitation and in-person pulmonary rehabilitation for exercise capacity measured as 6-Minute Walking Distance (mean difference 0.06 meters (m), 95% confidence interval (CI) -10.82 m to 10.94 m).” They reached the same conclusion for quality of life, and for breathlessness. Completion of rehabilitation programs, however, was more likely with telerehabilitation at 93% versus 70% for in-person rehabilitation. No adverse effects of telerehabilitation were observed over and above those for in-person or no rehabilitation. An obvious limitation of the findings is that the studies all pre-date COVID-19, which would have introduced very significant disincentives for in-person rehabilitation completion.
An older (2016) international randomized controlled study (Zanaboni et al, https://doi.org/10.1186/s12890-016-0288-z) comparing long-term telerehabilitation or unsupervised treadmill training at home with standard care included 120 participants with COPD and had 2-years of follow-up. Telerehabilitation consisted of individualized treadmill training at home. Participants had scheduled exercise sessions supervised by a physiotherapist via videoconferencing following a standardized protocol. Participants in the unsupervised training group were provided with a treadmill only to perform unsupervised exercise at home. They also received an exercise booklet, a paper exercise diary to record their training sessions, and an individualized training program but without regular review or progression of the program. For the primary outcomes of combined hospitalizations and emergency department presentations, incidence rate of hospitalizations and emergency department presentations was lower with telerehabilitation (1.18 events per person-year; 95% confidence interval [CI], 0.94–1.46) and with unsupervised training group (1.14; 95% CI, 0.92–1.41) than in the control group (1.88; 95% CI, 1.58–2.21; P < .001 compared with intervention groups). Both training groups had better health status at 1-year, and achieved and maintained clinically significant improvements in exercise capacity.
Access to pulmonary rehabilitation
Continuing evidence of clear telerehabilitation benefits is good news, especially in the light of impediments to attendance at in-clinic programs. Although the COVID-provoked disincentives have been diminishing, persisting access issues remain for substantial portions of eligible populations, according to a recent (2024) cross-sectional study (PA Kahn, WA Mathis, doi:10.1001/jamanetworkopen.2023.54867) looking at travel time to pulmonary rehabilitation programs as a marker for pulmonary rehabilitation access. The report, based on US Census designations (lower 48 states and Washington, D.C.) found that while 80.3% of the population lives in urban or suburban areas within a 30-minute drive of a pulmonary rehabilitation program, travel time exceeds that in rural and other sparsely populated areas with more than 14 million people residing in areas demanding more than 1-hour for travel. A further analysis showed also that nearly 30% of American Indian and Alaska Native populations live more than 60 minutes from a pulmonary rehabilitation program.
Aside from the obvious restraints for homebound patients or those lacking transportation or who need medical transport, other common impediments inhibit on-site pulmonary rehabilitation attendance, said Corinne Young, MSN, FNP-C, FCCP. Ms. Young is the director of Advance Practice Provider and Clinical Services for Colorado Springs Pulmonary Consultants, president and founder of the Association of Pulmonary Advance Practice Providers, and a member of the CHEST Physician Editorial Board. “I have some patients who say ‘There’s no way I could do onsite pulmonary rehab because of my knee — or back, or shoulder.’ But in their own home environment they may feel more comfortable. They may be willing to try new things at their own pace, whereas for them a program may feel too regimented.” For others, Ms. Young said, aspects of a formal program are a clear plus factor. “They love to hear their progress at the end of — say a 12-week program — where their virtual respiratory therapist records and reports to them their six-minute walk and other test results. Feedback is a great reinforcer.” Quality of life improvements, Ms. Young commented, were one of the very impressive benefits that appeared in the initial studies of pulmonary rehabilitation for COPD patients. “Being patient-centric, you want to improve quality of life for them as much as possible and we see telerehabilitation as a great opportunity for many,” she added.
“I would like to see head-to-head data on outpatient versus at-home pulmonary rehabilitation on hospitalizations, time to exacerbation and, of course, mortality. We have all that for outpatient rehab, but it would be great to be able to compare them. Knowing that would influence what we recommend, especially for patients who could go either way. Also, you have to assess their motivation and discipline to know who might be more appropriate for unsupervised pulmonary rehabilitation.”
The current reality for Ms. Young is that in her Colorado Springs vicinity, where both in-patient programs are only 15 minutes apart, she knows of no telerehabilitation programs being offered. While there are contract telerehabilitation providers, Young said, and her organization (The Association of Pulmonary Advanced Practice Providers) has been approached by one, none are licensed in Colorado, and telerehabilitation is not a billable service.
“As of yet, I’m not aware of any telemedicine pulmonary rehab available at our institution,” said pulmonologist Mary Jo S. Farmer, MD, PhD, FCCP, Associate Professor of Medicine at UMass Chan Medical School – Baystate, Springfield, MA, and a member of the CHEST Physician Editorial Board. A brief internet search identified a telerehabilitation contract provider available only in Arizona.
Reimbursement will also be a foundational concern, Ms. Young commented. While a physician, nurse practitioner, or physician virtual visit for education may be billable, telerehabilitation reimbursement is new territory. “How that all is going to work out is a big unknown piece,” she said.
Minimal components
Effective pulmonary telerehabilitation programs, Ms. Young said, need to provide exercise with an aerobic device, either a treadmill, a stationary bike or even a Cubii-type under desk foot pedal/elliptical machine, and some resistance training (elastic bands, or weights, for example). “But 50% of pulmonary rehabilitation is education about breathing techniques, purse-lip breathing, and pulmonary nutrition.” Also essential: one-on-one discussion with a qualified medical practitioner who checks on oximeter use, inhaler technique, and titrating oxygen therapy. “At our elevation of 6500 feet, most of our patients are on that.” Optimal frequency of encounters between providers and remote patients has to be elucidated by future research, Ms. Young said.
Ms. Young commented further, “With outpatient pulmonary rehabilitation there often isn’t a lot of one-on-one, but rather a big group of people exercising at the same time. I think actually there may be the potential to have more individualization with pulmonary telerehabilitation. But the barriers, the reimbursement/financial part, and the red tape and bureaucracy have to be worked on.”
As COVID-19 cedes its pandemic-scale status to the past, its wake is revealing surprises and raising questions, particularly in relation to pulmonary medicine. The need for isolation at COVID’s outset kept many millions at home, creating conditions favorable for the rapid expansion of technologies that were taken up quickly in telehealth applications. The need was overwhelming. But just how effective telehealth actually is at replacing on-site programs for COPD pulmonary rehab has remained a research challenge, although results from early studies show unmistakable value. Creating conditions conducive to research into the strengths and weaknesses of pulmonary rehab, and determining how research can be applied effectively, remain formidable challenges.
Early studies of telehealth pulmonary rehabilitation have not uncovered any glaring erosion of pulmonary rehabilitation’s well-established benefits. But, at the same time, the relatively young field of pulmonary telerehabilitation for chronic obstructive pulmonary disease (COPD) has lacked coordinated efforts to determine its key practices and the instruments for measuring them, both basic elements for pursuing research questions.
A 2021 American Thoracic Society workshop report (AE Holland, https://doi.org/10.1513/AnnalsATS.202102-146ST) identified essential components of a pulmonary rehabilitation model through an online Delphi process involving about 50 international experts. Components ultimately included those with median scores of 2 or higher (strongly agree or agree that the item is essential) and high consensus (interquartile range, 0). Thirteen essential components fit into four categories (Patient Assessment, Program Components, Method of Delivery and Quality Assurance). The Patient Assessment category included seven items: (1) An initial center-based assessment by a health care professional, (2) An exercise test at the time of assessment, (3) A field exercise test, (4) Quality of life measure, (5) Dyspnea assessment, (6) Nutritional status evaluation, and (7) Occupational status evaluation. The Program Components: (8) Endurance training and (9) Resistance training). The Method of Delivery: (10) An exercise program that is individually prescribed, (11) An exercise program that is individually progressed, and (12) Team includes a health care professional with experience in exercise prescription and progression. The single Quality Assurance item: (13) Health care professionals are trained to deliver the components of the model that is deployed.
Cochrane Library review
2021 “Intervention Review” (“Telerehabilitation for chronic respiratory disease,” https://doi.org/10.1002/14651858.CD013040.pub2). Using their own databases (eg, Cochrane Airways Trials Register) and others, the authors included controlled trials published up to November 30, 2020 with at least 50% of the rehabilitation delivered by telerehabilitation. The authors’ analysis of 15 studies (with 32 reports) including 1904 participants (99% with COPD): “There was probably little or no difference between telerehabilitation and in-person pulmonary rehabilitation for exercise capacity measured as 6-Minute Walking Distance (mean difference 0.06 meters (m), 95% confidence interval (CI) -10.82 m to 10.94 m).” They reached the same conclusion for quality of life, and for breathlessness. Completion of rehabilitation programs, however, was more likely with telerehabilitation at 93% versus 70% for in-person rehabilitation. No adverse effects of telerehabilitation were observed over and above those for in-person or no rehabilitation. An obvious limitation of the findings is that the studies all pre-date COVID-19, which would have introduced very significant disincentives for in-person rehabilitation completion.
An older (2016) international randomized controlled study (Zanaboni et al, https://doi.org/10.1186/s12890-016-0288-z) comparing long-term telerehabilitation or unsupervised treadmill training at home with standard care included 120 participants with COPD and had 2-years of follow-up. Telerehabilitation consisted of individualized treadmill training at home. Participants had scheduled exercise sessions supervised by a physiotherapist via videoconferencing following a standardized protocol. Participants in the unsupervised training group were provided with a treadmill only to perform unsupervised exercise at home. They also received an exercise booklet, a paper exercise diary to record their training sessions, and an individualized training program but without regular review or progression of the program. For the primary outcomes of combined hospitalizations and emergency department presentations, incidence rate of hospitalizations and emergency department presentations was lower with telerehabilitation (1.18 events per person-year; 95% confidence interval [CI], 0.94–1.46) and with unsupervised training group (1.14; 95% CI, 0.92–1.41) than in the control group (1.88; 95% CI, 1.58–2.21; P < .001 compared with intervention groups). Both training groups had better health status at 1-year, and achieved and maintained clinically significant improvements in exercise capacity.
Access to pulmonary rehabilitation
Continuing evidence of clear telerehabilitation benefits is good news, especially in the light of impediments to attendance at in-clinic programs. Although the COVID-provoked disincentives have been diminishing, persisting access issues remain for substantial portions of eligible populations, according to a recent (2024) cross-sectional study (PA Kahn, WA Mathis, doi:10.1001/jamanetworkopen.2023.54867) looking at travel time to pulmonary rehabilitation programs as a marker for pulmonary rehabilitation access. The report, based on US Census designations (lower 48 states and Washington, D.C.) found that while 80.3% of the population lives in urban or suburban areas within a 30-minute drive of a pulmonary rehabilitation program, travel time exceeds that in rural and other sparsely populated areas with more than 14 million people residing in areas demanding more than 1-hour for travel. A further analysis showed also that nearly 30% of American Indian and Alaska Native populations live more than 60 minutes from a pulmonary rehabilitation program.
Aside from the obvious restraints for homebound patients or those lacking transportation or who need medical transport, other common impediments inhibit on-site pulmonary rehabilitation attendance, said Corinne Young, MSN, FNP-C, FCCP. Ms. Young is the director of Advance Practice Provider and Clinical Services for Colorado Springs Pulmonary Consultants, president and founder of the Association of Pulmonary Advance Practice Providers, and a member of the CHEST Physician Editorial Board. “I have some patients who say ‘There’s no way I could do onsite pulmonary rehab because of my knee — or back, or shoulder.’ But in their own home environment they may feel more comfortable. They may be willing to try new things at their own pace, whereas for them a program may feel too regimented.” For others, Ms. Young said, aspects of a formal program are a clear plus factor. “They love to hear their progress at the end of — say a 12-week program — where their virtual respiratory therapist records and reports to them their six-minute walk and other test results. Feedback is a great reinforcer.” Quality of life improvements, Ms. Young commented, were one of the very impressive benefits that appeared in the initial studies of pulmonary rehabilitation for COPD patients. “Being patient-centric, you want to improve quality of life for them as much as possible and we see telerehabilitation as a great opportunity for many,” she added.
“I would like to see head-to-head data on outpatient versus at-home pulmonary rehabilitation on hospitalizations, time to exacerbation and, of course, mortality. We have all that for outpatient rehab, but it would be great to be able to compare them. Knowing that would influence what we recommend, especially for patients who could go either way. Also, you have to assess their motivation and discipline to know who might be more appropriate for unsupervised pulmonary rehabilitation.”
The current reality for Ms. Young is that in her Colorado Springs vicinity, where both in-patient programs are only 15 minutes apart, she knows of no telerehabilitation programs being offered. While there are contract telerehabilitation providers, Young said, and her organization (The Association of Pulmonary Advanced Practice Providers) has been approached by one, none are licensed in Colorado, and telerehabilitation is not a billable service.
“As of yet, I’m not aware of any telemedicine pulmonary rehab available at our institution,” said pulmonologist Mary Jo S. Farmer, MD, PhD, FCCP, Associate Professor of Medicine at UMass Chan Medical School – Baystate, Springfield, MA, and a member of the CHEST Physician Editorial Board. A brief internet search identified a telerehabilitation contract provider available only in Arizona.
Reimbursement will also be a foundational concern, Ms. Young commented. While a physician, nurse practitioner, or physician virtual visit for education may be billable, telerehabilitation reimbursement is new territory. “How that all is going to work out is a big unknown piece,” she said.
Minimal components
Effective pulmonary telerehabilitation programs, Ms. Young said, need to provide exercise with an aerobic device, either a treadmill, a stationary bike or even a Cubii-type under desk foot pedal/elliptical machine, and some resistance training (elastic bands, or weights, for example). “But 50% of pulmonary rehabilitation is education about breathing techniques, purse-lip breathing, and pulmonary nutrition.” Also essential: one-on-one discussion with a qualified medical practitioner who checks on oximeter use, inhaler technique, and titrating oxygen therapy. “At our elevation of 6500 feet, most of our patients are on that.” Optimal frequency of encounters between providers and remote patients has to be elucidated by future research, Ms. Young said.
Ms. Young commented further, “With outpatient pulmonary rehabilitation there often isn’t a lot of one-on-one, but rather a big group of people exercising at the same time. I think actually there may be the potential to have more individualization with pulmonary telerehabilitation. But the barriers, the reimbursement/financial part, and the red tape and bureaucracy have to be worked on.”
Recalcitrant Folliculitis Decalvans Treatment Outcomes With Biologics and Small Molecule Inhibitors
Folliculitis decalvans (FD) is classified as a rare primary neutrophilic cicatricial alopecia occurring predominantly in middle-aged adults. Although the true etiology is still unknown, the pathogenesis behind the inflammatory follicular lesions stems from possible Staphylococcus aureus infection and an impaired host immune system in response to released superantigens. 1 The clinical severity of this inflammatory scalp disorder can range from mild to severe and debilitating. Multiple treatment regimens have been developed with the goal of maintaining full remission. We provide a summary of tumor necrosis factor (TNF) inhibitors, Janus kinase (JAK) inhibitors, phosphodiesterase 4 (PDE4) inhibitors, and monoclonal antibodies being utilized for patients with therapy-recalcitrant FD.
Methods
We conducted a PubMed, Medline, and Google Scholar search for the terms refractory FD, recalcitrant FD, or therapy-resistant FD to identify articles published in English from 1998 to 2022. Articles that reported recalcitrant cases and subsequent therapy with TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies were included. Articles were excluded if recalcitrant cases were not clearly defined. Remission was defined as no recurrence in lesions or pustules or as a reduction in the inflammatory process with stabilization upon continuation or discontinuation of the therapy regimen. Two reviewers (T.F. and K.U.) independently searched for and screened each report.
Results
Treatment of recalcitrant FD with biologics or small molecule inhibitors was discussed in 9 studies with a combined total of 35 patients.2-10 The treatment regimens included TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies (Table).
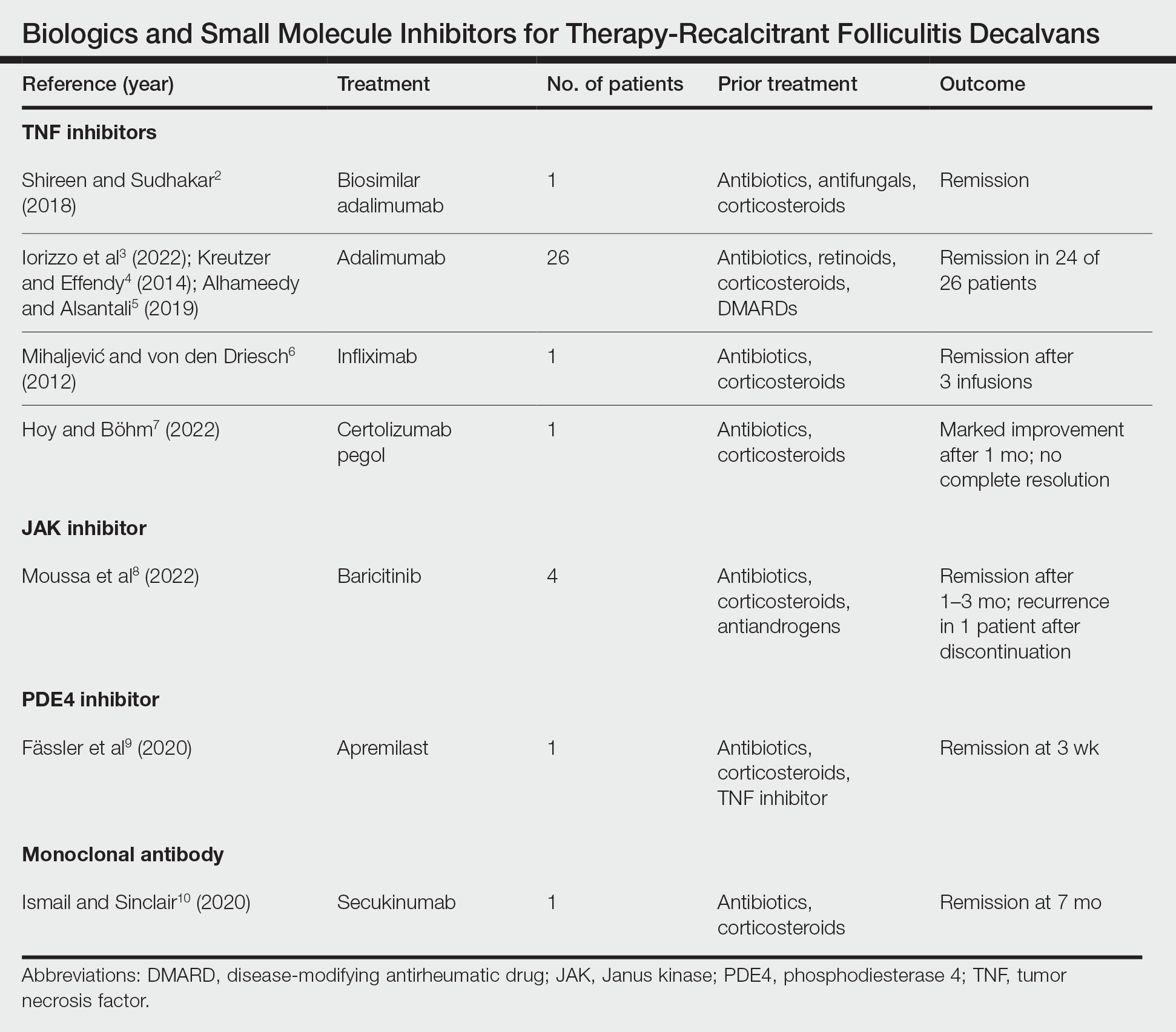
The TNF inhibitors were utilized in 6 reports with a combined total of 29 patients. Treatments included adalimumab or biosimilar adalimumab (27/29 patients), infliximab (1/29 patients), and certolizumab pegol (1/29 patients). Remission was reported in 26 of 29 cases. There were 2 nonresponders to adalimumab and marked improvement with certolizumab pegol without complete resolution. The use of the JAK inhibitor baricitinib in 4 patients resulted in remission. In all 4 patients, baricitinib was used with concurrent treatments, and remission was achieved in an average of 2.25 months. The use of a PDE4 inhibitor, apremilast, was reported in 1 case; remission was achieved in 3 weeks. Secukinumab, a monoclonal antibody that targets IL-17, was utilized in 1 patient. Marked improvement was seen after 2 months, with complete remission in 7 months.
Comment
Traditional treatment regimens for FD most often include a combination of topical and oral antibiotics; isotretinoin; and oral, topical, or intralesional corticosteroids. In the past, interventions typically were suppressive as opposed to curative; however, recent treatment advancements have shown promise in achieving lasting remission.
Most reports targeting treatment-resistant FD involved the use of TNF inhibitors, including adalimumab, biosimilar adalimumab, infliximab, and certolizumab pegol. Adalimumab was the most frequently used TNF inhibitor, with 24 of 26 treated patients achieving remission. Adalimumab may have been used the most in the treatment of FD because TNF is pronounced in other neutrophilic dermatoses that have been successfully treated with TNF inhibitors. It has been reported that adalimumab needs to be continued, as stoppage or interruption led to relapse.3
Although there are few reports of the use of JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies for FD, these treatment modalities show promise, as their use led to marked improvement or lasting remission with ongoing treatment. The use of the PDE4 inhibitor apremilast displayed the most rapid improvement of any of the reviewed treatments, with remission achieved in just 3 weeks.9 The rapid success of apremilast may be attributed to the inhibitory effect on neutrophils.
Miguel-Gómez et al11 provided a therapeutic protocol for FD based on the severity of disease (N=60). The protocol included rifampicin plus clindamycin for the treatment of severe disease, as 90.5% (19/21) of resistant cases showed clinical response, with remission of 5 months’ duration. Although this may be acceptable for some patients, others may require an alternative approach. Tietze et al12 showed that rifampicin and clindamycin had the lowest success rate for long-term remission, with 8 of 10 patients relapsing within 2 to 4 months. In addition, the emergence of antimicrobial resistance remains a major concern in the treatment of FD. Upon the review of the most recent reports of successful treatment of therapy-resistant FD, biologics and small molecule inhibitors have shown remission extending through a 12-month follow-up period. We suggest considering the addition of biologics and small molecule inhibitors to the treatment protocol for severe or resistant disease.
Limitations—In the articles reviewed, the definition of remission was inconsistent among authors—some characterized it as no recurrence in lesions or pustules and some as a reduction in the inflammatory process. True duration of remission was difficult to assess from case reports, as follow-up periods varied prior to publication. The studies included in this review consisted mainly of small sample sizes owing to the rarity of FD, and consequently, strength of evidence is lacking. Inherent to the nature of systematic reviews, publication bias may have occurred. Lastly, several studies were impacted by difficulty in obtaining optimal treatment due to financial hardship, and regimens were adjusted accordingly.
Conclusion
The relapsing nature of FD leads to frustration and poor quality of life for patients. There is a paucity of data to guide treatment when FD remains recalcitrant to traditional therapy. Therapies such as TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies have shown success in the treatment of this often difficult-to-treat disease. Small sample sizes in reports discussing treatment for resistant cases as well as conflicting results make it challenging to draw conclusions about treatment efficacy. Larger studies are needed to understand the long-term outcomes of treatment options. Regardless, disease severity, patient history, patient preferences, and treatment goals can guide the selection of therapeutic options.
- Otberg N, Kang H, Alzolibani AA, et al. Folliculitis decalvans. Dermatol Ther. 2008;21:238-244. doi:10.1111/j.1529-8019.2008.00204.x
- Shireen F, Sudhakar A. A case of isotretinoin therapy-refractory folliculitis decalvans treated successfully with biosimilar adalimumab (Exemptia). Int J Trichology. 2018;10:240-241.
- Iorizzo M, Starace M, Vano-Galvan S, et al. Refractory folliculitis decalvans treated with adalimumab: a case series of 23 patients. J Am Acad Dermatol. 2022;87:666-669. doi:10.1016/j.jaad.2022.02.044
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76. doi:10.1111/ddg.12224
- Alhameedy MM, Alsantali AM. Therapy-recalcitrant folliculitis decalvans controlled successfully with adalimumab. Int J Trichology. 2019;11:241-243. doi:10.4103/ijt.ijt_92_19
- Mihaljevic´ N, von den Driesch P. Successful use of infliximab in a patient with recalcitrant folliculitis decalvans. J Dtsch Dermatol Ges. 2012;10:589-590. doi:10.1111/j.1610-0387.2012.07972.x
- Hoy M, Böhm M. Therapy-refractory folliculitis decalvans treated with certolizumab pegol. Int J Dermatol. 2022;61:e26-e28. doi:10.1111/ijd.15914
- Moussa A, Asfour L, Eisman S, et al. Successful treatment of folliculitis decalvans with baricitinib: a case series. Australas J Dermatol. 2022;63:279-281. doi:10.1111/ajd.13786
- Fässler M, Radonjic-Hoesli S, Feldmeyer L, et al. Successful treatment of refractory folliculitis decalvans with apremilast. JAAD Case Rep. 2020;6:1079-1081. doi:10.1016/j.jdcr.2020.08.019
- Ismail FF, Sinclair R. Successful treatment of refractory folliculitis decalvans with secukinumab. Australas J Dermatol. 2020;61:165-166. doi:10.1111/ajd.13190
- Miguel-Gómez L, Rodrigues-Barata AR, Molina-Ruiz A, et al. Folliculitis decalvans: effectiveness of therapies and prognostic factors in a multicenter series of 60 patients with long-term follow-up. J Am Acad Dermatol. 2018;79:878-883. doi:10.1016/j.jaad.2018.05.1240
- Tietze JK, Heppt MV, von Preußen A, et al. Oral isotretinoin as the most effective treatment in folliculitis decalvans: a retrospective comparison of different treatment regimens in 28 patients. J Eur Acad Dermatol Venereol. 2015;29:1816-1821. doi:10.1111/jdv.13052
Folliculitis decalvans (FD) is classified as a rare primary neutrophilic cicatricial alopecia occurring predominantly in middle-aged adults. Although the true etiology is still unknown, the pathogenesis behind the inflammatory follicular lesions stems from possible Staphylococcus aureus infection and an impaired host immune system in response to released superantigens. 1 The clinical severity of this inflammatory scalp disorder can range from mild to severe and debilitating. Multiple treatment regimens have been developed with the goal of maintaining full remission. We provide a summary of tumor necrosis factor (TNF) inhibitors, Janus kinase (JAK) inhibitors, phosphodiesterase 4 (PDE4) inhibitors, and monoclonal antibodies being utilized for patients with therapy-recalcitrant FD.
Methods
We conducted a PubMed, Medline, and Google Scholar search for the terms refractory FD, recalcitrant FD, or therapy-resistant FD to identify articles published in English from 1998 to 2022. Articles that reported recalcitrant cases and subsequent therapy with TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies were included. Articles were excluded if recalcitrant cases were not clearly defined. Remission was defined as no recurrence in lesions or pustules or as a reduction in the inflammatory process with stabilization upon continuation or discontinuation of the therapy regimen. Two reviewers (T.F. and K.U.) independently searched for and screened each report.
Results
Treatment of recalcitrant FD with biologics or small molecule inhibitors was discussed in 9 studies with a combined total of 35 patients.2-10 The treatment regimens included TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies (Table).

The TNF inhibitors were utilized in 6 reports with a combined total of 29 patients. Treatments included adalimumab or biosimilar adalimumab (27/29 patients), infliximab (1/29 patients), and certolizumab pegol (1/29 patients). Remission was reported in 26 of 29 cases. There were 2 nonresponders to adalimumab and marked improvement with certolizumab pegol without complete resolution. The use of the JAK inhibitor baricitinib in 4 patients resulted in remission. In all 4 patients, baricitinib was used with concurrent treatments, and remission was achieved in an average of 2.25 months. The use of a PDE4 inhibitor, apremilast, was reported in 1 case; remission was achieved in 3 weeks. Secukinumab, a monoclonal antibody that targets IL-17, was utilized in 1 patient. Marked improvement was seen after 2 months, with complete remission in 7 months.
Comment
Traditional treatment regimens for FD most often include a combination of topical and oral antibiotics; isotretinoin; and oral, topical, or intralesional corticosteroids. In the past, interventions typically were suppressive as opposed to curative; however, recent treatment advancements have shown promise in achieving lasting remission.
Most reports targeting treatment-resistant FD involved the use of TNF inhibitors, including adalimumab, biosimilar adalimumab, infliximab, and certolizumab pegol. Adalimumab was the most frequently used TNF inhibitor, with 24 of 26 treated patients achieving remission. Adalimumab may have been used the most in the treatment of FD because TNF is pronounced in other neutrophilic dermatoses that have been successfully treated with TNF inhibitors. It has been reported that adalimumab needs to be continued, as stoppage or interruption led to relapse.3
Although there are few reports of the use of JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies for FD, these treatment modalities show promise, as their use led to marked improvement or lasting remission with ongoing treatment. The use of the PDE4 inhibitor apremilast displayed the most rapid improvement of any of the reviewed treatments, with remission achieved in just 3 weeks.9 The rapid success of apremilast may be attributed to the inhibitory effect on neutrophils.
Miguel-Gómez et al11 provided a therapeutic protocol for FD based on the severity of disease (N=60). The protocol included rifampicin plus clindamycin for the treatment of severe disease, as 90.5% (19/21) of resistant cases showed clinical response, with remission of 5 months’ duration. Although this may be acceptable for some patients, others may require an alternative approach. Tietze et al12 showed that rifampicin and clindamycin had the lowest success rate for long-term remission, with 8 of 10 patients relapsing within 2 to 4 months. In addition, the emergence of antimicrobial resistance remains a major concern in the treatment of FD. Upon the review of the most recent reports of successful treatment of therapy-resistant FD, biologics and small molecule inhibitors have shown remission extending through a 12-month follow-up period. We suggest considering the addition of biologics and small molecule inhibitors to the treatment protocol for severe or resistant disease.
Limitations—In the articles reviewed, the definition of remission was inconsistent among authors—some characterized it as no recurrence in lesions or pustules and some as a reduction in the inflammatory process. True duration of remission was difficult to assess from case reports, as follow-up periods varied prior to publication. The studies included in this review consisted mainly of small sample sizes owing to the rarity of FD, and consequently, strength of evidence is lacking. Inherent to the nature of systematic reviews, publication bias may have occurred. Lastly, several studies were impacted by difficulty in obtaining optimal treatment due to financial hardship, and regimens were adjusted accordingly.
Conclusion
The relapsing nature of FD leads to frustration and poor quality of life for patients. There is a paucity of data to guide treatment when FD remains recalcitrant to traditional therapy. Therapies such as TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies have shown success in the treatment of this often difficult-to-treat disease. Small sample sizes in reports discussing treatment for resistant cases as well as conflicting results make it challenging to draw conclusions about treatment efficacy. Larger studies are needed to understand the long-term outcomes of treatment options. Regardless, disease severity, patient history, patient preferences, and treatment goals can guide the selection of therapeutic options.
Folliculitis decalvans (FD) is classified as a rare primary neutrophilic cicatricial alopecia occurring predominantly in middle-aged adults. Although the true etiology is still unknown, the pathogenesis behind the inflammatory follicular lesions stems from possible Staphylococcus aureus infection and an impaired host immune system in response to released superantigens. 1 The clinical severity of this inflammatory scalp disorder can range from mild to severe and debilitating. Multiple treatment regimens have been developed with the goal of maintaining full remission. We provide a summary of tumor necrosis factor (TNF) inhibitors, Janus kinase (JAK) inhibitors, phosphodiesterase 4 (PDE4) inhibitors, and monoclonal antibodies being utilized for patients with therapy-recalcitrant FD.
Methods
We conducted a PubMed, Medline, and Google Scholar search for the terms refractory FD, recalcitrant FD, or therapy-resistant FD to identify articles published in English from 1998 to 2022. Articles that reported recalcitrant cases and subsequent therapy with TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies were included. Articles were excluded if recalcitrant cases were not clearly defined. Remission was defined as no recurrence in lesions or pustules or as a reduction in the inflammatory process with stabilization upon continuation or discontinuation of the therapy regimen. Two reviewers (T.F. and K.U.) independently searched for and screened each report.
Results
Treatment of recalcitrant FD with biologics or small molecule inhibitors was discussed in 9 studies with a combined total of 35 patients.2-10 The treatment regimens included TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies (Table).

The TNF inhibitors were utilized in 6 reports with a combined total of 29 patients. Treatments included adalimumab or biosimilar adalimumab (27/29 patients), infliximab (1/29 patients), and certolizumab pegol (1/29 patients). Remission was reported in 26 of 29 cases. There were 2 nonresponders to adalimumab and marked improvement with certolizumab pegol without complete resolution. The use of the JAK inhibitor baricitinib in 4 patients resulted in remission. In all 4 patients, baricitinib was used with concurrent treatments, and remission was achieved in an average of 2.25 months. The use of a PDE4 inhibitor, apremilast, was reported in 1 case; remission was achieved in 3 weeks. Secukinumab, a monoclonal antibody that targets IL-17, was utilized in 1 patient. Marked improvement was seen after 2 months, with complete remission in 7 months.
Comment
Traditional treatment regimens for FD most often include a combination of topical and oral antibiotics; isotretinoin; and oral, topical, or intralesional corticosteroids. In the past, interventions typically were suppressive as opposed to curative; however, recent treatment advancements have shown promise in achieving lasting remission.
Most reports targeting treatment-resistant FD involved the use of TNF inhibitors, including adalimumab, biosimilar adalimumab, infliximab, and certolizumab pegol. Adalimumab was the most frequently used TNF inhibitor, with 24 of 26 treated patients achieving remission. Adalimumab may have been used the most in the treatment of FD because TNF is pronounced in other neutrophilic dermatoses that have been successfully treated with TNF inhibitors. It has been reported that adalimumab needs to be continued, as stoppage or interruption led to relapse.3
Although there are few reports of the use of JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies for FD, these treatment modalities show promise, as their use led to marked improvement or lasting remission with ongoing treatment. The use of the PDE4 inhibitor apremilast displayed the most rapid improvement of any of the reviewed treatments, with remission achieved in just 3 weeks.9 The rapid success of apremilast may be attributed to the inhibitory effect on neutrophils.
Miguel-Gómez et al11 provided a therapeutic protocol for FD based on the severity of disease (N=60). The protocol included rifampicin plus clindamycin for the treatment of severe disease, as 90.5% (19/21) of resistant cases showed clinical response, with remission of 5 months’ duration. Although this may be acceptable for some patients, others may require an alternative approach. Tietze et al12 showed that rifampicin and clindamycin had the lowest success rate for long-term remission, with 8 of 10 patients relapsing within 2 to 4 months. In addition, the emergence of antimicrobial resistance remains a major concern in the treatment of FD. Upon the review of the most recent reports of successful treatment of therapy-resistant FD, biologics and small molecule inhibitors have shown remission extending through a 12-month follow-up period. We suggest considering the addition of biologics and small molecule inhibitors to the treatment protocol for severe or resistant disease.
Limitations—In the articles reviewed, the definition of remission was inconsistent among authors—some characterized it as no recurrence in lesions or pustules and some as a reduction in the inflammatory process. True duration of remission was difficult to assess from case reports, as follow-up periods varied prior to publication. The studies included in this review consisted mainly of small sample sizes owing to the rarity of FD, and consequently, strength of evidence is lacking. Inherent to the nature of systematic reviews, publication bias may have occurred. Lastly, several studies were impacted by difficulty in obtaining optimal treatment due to financial hardship, and regimens were adjusted accordingly.
Conclusion
The relapsing nature of FD leads to frustration and poor quality of life for patients. There is a paucity of data to guide treatment when FD remains recalcitrant to traditional therapy. Therapies such as TNF inhibitors, JAK inhibitors, PDE4 inhibitors, and monoclonal antibodies have shown success in the treatment of this often difficult-to-treat disease. Small sample sizes in reports discussing treatment for resistant cases as well as conflicting results make it challenging to draw conclusions about treatment efficacy. Larger studies are needed to understand the long-term outcomes of treatment options. Regardless, disease severity, patient history, patient preferences, and treatment goals can guide the selection of therapeutic options.
- Otberg N, Kang H, Alzolibani AA, et al. Folliculitis decalvans. Dermatol Ther. 2008;21:238-244. doi:10.1111/j.1529-8019.2008.00204.x
- Shireen F, Sudhakar A. A case of isotretinoin therapy-refractory folliculitis decalvans treated successfully with biosimilar adalimumab (Exemptia). Int J Trichology. 2018;10:240-241.
- Iorizzo M, Starace M, Vano-Galvan S, et al. Refractory folliculitis decalvans treated with adalimumab: a case series of 23 patients. J Am Acad Dermatol. 2022;87:666-669. doi:10.1016/j.jaad.2022.02.044
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76. doi:10.1111/ddg.12224
- Alhameedy MM, Alsantali AM. Therapy-recalcitrant folliculitis decalvans controlled successfully with adalimumab. Int J Trichology. 2019;11:241-243. doi:10.4103/ijt.ijt_92_19
- Mihaljevic´ N, von den Driesch P. Successful use of infliximab in a patient with recalcitrant folliculitis decalvans. J Dtsch Dermatol Ges. 2012;10:589-590. doi:10.1111/j.1610-0387.2012.07972.x
- Hoy M, Böhm M. Therapy-refractory folliculitis decalvans treated with certolizumab pegol. Int J Dermatol. 2022;61:e26-e28. doi:10.1111/ijd.15914
- Moussa A, Asfour L, Eisman S, et al. Successful treatment of folliculitis decalvans with baricitinib: a case series. Australas J Dermatol. 2022;63:279-281. doi:10.1111/ajd.13786
- Fässler M, Radonjic-Hoesli S, Feldmeyer L, et al. Successful treatment of refractory folliculitis decalvans with apremilast. JAAD Case Rep. 2020;6:1079-1081. doi:10.1016/j.jdcr.2020.08.019
- Ismail FF, Sinclair R. Successful treatment of refractory folliculitis decalvans with secukinumab. Australas J Dermatol. 2020;61:165-166. doi:10.1111/ajd.13190
- Miguel-Gómez L, Rodrigues-Barata AR, Molina-Ruiz A, et al. Folliculitis decalvans: effectiveness of therapies and prognostic factors in a multicenter series of 60 patients with long-term follow-up. J Am Acad Dermatol. 2018;79:878-883. doi:10.1016/j.jaad.2018.05.1240
- Tietze JK, Heppt MV, von Preußen A, et al. Oral isotretinoin as the most effective treatment in folliculitis decalvans: a retrospective comparison of different treatment regimens in 28 patients. J Eur Acad Dermatol Venereol. 2015;29:1816-1821. doi:10.1111/jdv.13052
- Otberg N, Kang H, Alzolibani AA, et al. Folliculitis decalvans. Dermatol Ther. 2008;21:238-244. doi:10.1111/j.1529-8019.2008.00204.x
- Shireen F, Sudhakar A. A case of isotretinoin therapy-refractory folliculitis decalvans treated successfully with biosimilar adalimumab (Exemptia). Int J Trichology. 2018;10:240-241.
- Iorizzo M, Starace M, Vano-Galvan S, et al. Refractory folliculitis decalvans treated with adalimumab: a case series of 23 patients. J Am Acad Dermatol. 2022;87:666-669. doi:10.1016/j.jaad.2022.02.044
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76. doi:10.1111/ddg.12224
- Alhameedy MM, Alsantali AM. Therapy-recalcitrant folliculitis decalvans controlled successfully with adalimumab. Int J Trichology. 2019;11:241-243. doi:10.4103/ijt.ijt_92_19
- Mihaljevic´ N, von den Driesch P. Successful use of infliximab in a patient with recalcitrant folliculitis decalvans. J Dtsch Dermatol Ges. 2012;10:589-590. doi:10.1111/j.1610-0387.2012.07972.x
- Hoy M, Böhm M. Therapy-refractory folliculitis decalvans treated with certolizumab pegol. Int J Dermatol. 2022;61:e26-e28. doi:10.1111/ijd.15914
- Moussa A, Asfour L, Eisman S, et al. Successful treatment of folliculitis decalvans with baricitinib: a case series. Australas J Dermatol. 2022;63:279-281. doi:10.1111/ajd.13786
- Fässler M, Radonjic-Hoesli S, Feldmeyer L, et al. Successful treatment of refractory folliculitis decalvans with apremilast. JAAD Case Rep. 2020;6:1079-1081. doi:10.1016/j.jdcr.2020.08.019
- Ismail FF, Sinclair R. Successful treatment of refractory folliculitis decalvans with secukinumab. Australas J Dermatol. 2020;61:165-166. doi:10.1111/ajd.13190
- Miguel-Gómez L, Rodrigues-Barata AR, Molina-Ruiz A, et al. Folliculitis decalvans: effectiveness of therapies and prognostic factors in a multicenter series of 60 patients with long-term follow-up. J Am Acad Dermatol. 2018;79:878-883. doi:10.1016/j.jaad.2018.05.1240
- Tietze JK, Heppt MV, von Preußen A, et al. Oral isotretinoin as the most effective treatment in folliculitis decalvans: a retrospective comparison of different treatment regimens in 28 patients. J Eur Acad Dermatol Venereol. 2015;29:1816-1821. doi:10.1111/jdv.13052
Practice Points
- Tumor necrosis factor inhibitors, Janus kinase inhibitors, phosphodiesterase 4 inhibitors, and monoclonal antibodies have shown success in the treatment of folliculitis decalvans resistant to traditional therapies.
- The true etiology of folliculitis decalvans is still unknown, but possible factors include Staphylococcus aureus infection and an impaired host immune system, which may benefit from treatment with biologics and small molecule inhibitors.
Complementing, not competing
As we enter summer, it’s hard to believe that we’re halfway through my presidency. Registration for CHEST 2024 (October 6 to 9) is now open, and October will be here before we know it.
I’m happy to share that we received more than 4,000 abstract and case report submissions from clinicians at all stages of their careers, and, for the first year, we had a dedicated category to solicit submissions from physician assistants (PAs), nurse practitioners (NPs), respiratory therapists, and other members of the broader health care team.
In both my practice and my time as CHEST President, I’ve been reflecting on the benefits of the multidisciplinary team—especially in the ICU. Because this is a setting that relies heavily on a team aspect, every member of the care team is a great asset.
CHEST is working to ensure that all integral members of our professional health care teams have the resources they need to best serve our patients. We encourage advanced practice providers (APPs) to apply to serve on our committees during the current open call, and we recently launched a dedicated APP Intersection column, called APP Intersection, within this publication to elevate diverse perspectives. I anticipate more is on the horizon.
In my experience, I have seen tremendous success in partnering with and complementing each other, rather than competing for space when caring for a patient. Each and every one of us shares the same goal of providing the best patient care, and we each bring our own strengths.
Our future is ripe with opportunities to better serve the whole care team—MDs, PAs, NPs, and more—and it starts with recognizing the needs of everyone within the organization. To help CHEST better serve our members, I encourage you to take a short survey about your professional hurdles.
And please, do not hesitate to contact me (president@chestnet.org) with suggestions or just to introduce yourself.
All the best,
Jack
As we enter summer, it’s hard to believe that we’re halfway through my presidency. Registration for CHEST 2024 (October 6 to 9) is now open, and October will be here before we know it.
I’m happy to share that we received more than 4,000 abstract and case report submissions from clinicians at all stages of their careers, and, for the first year, we had a dedicated category to solicit submissions from physician assistants (PAs), nurse practitioners (NPs), respiratory therapists, and other members of the broader health care team.
In both my practice and my time as CHEST President, I’ve been reflecting on the benefits of the multidisciplinary team—especially in the ICU. Because this is a setting that relies heavily on a team aspect, every member of the care team is a great asset.
CHEST is working to ensure that all integral members of our professional health care teams have the resources they need to best serve our patients. We encourage advanced practice providers (APPs) to apply to serve on our committees during the current open call, and we recently launched a dedicated APP Intersection column, called APP Intersection, within this publication to elevate diverse perspectives. I anticipate more is on the horizon.
In my experience, I have seen tremendous success in partnering with and complementing each other, rather than competing for space when caring for a patient. Each and every one of us shares the same goal of providing the best patient care, and we each bring our own strengths.
Our future is ripe with opportunities to better serve the whole care team—MDs, PAs, NPs, and more—and it starts with recognizing the needs of everyone within the organization. To help CHEST better serve our members, I encourage you to take a short survey about your professional hurdles.
And please, do not hesitate to contact me (president@chestnet.org) with suggestions or just to introduce yourself.
All the best,
Jack
As we enter summer, it’s hard to believe that we’re halfway through my presidency. Registration for CHEST 2024 (October 6 to 9) is now open, and October will be here before we know it.
I’m happy to share that we received more than 4,000 abstract and case report submissions from clinicians at all stages of their careers, and, for the first year, we had a dedicated category to solicit submissions from physician assistants (PAs), nurse practitioners (NPs), respiratory therapists, and other members of the broader health care team.
In both my practice and my time as CHEST President, I’ve been reflecting on the benefits of the multidisciplinary team—especially in the ICU. Because this is a setting that relies heavily on a team aspect, every member of the care team is a great asset.
CHEST is working to ensure that all integral members of our professional health care teams have the resources they need to best serve our patients. We encourage advanced practice providers (APPs) to apply to serve on our committees during the current open call, and we recently launched a dedicated APP Intersection column, called APP Intersection, within this publication to elevate diverse perspectives. I anticipate more is on the horizon.
In my experience, I have seen tremendous success in partnering with and complementing each other, rather than competing for space when caring for a patient. Each and every one of us shares the same goal of providing the best patient care, and we each bring our own strengths.
Our future is ripe with opportunities to better serve the whole care team—MDs, PAs, NPs, and more—and it starts with recognizing the needs of everyone within the organization. To help CHEST better serve our members, I encourage you to take a short survey about your professional hurdles.
And please, do not hesitate to contact me (president@chestnet.org) with suggestions or just to introduce yourself.
All the best,
Jack
Nurse practitioners as advocates for health policy and patient care
APP INTERSECTION
In the intricate tapestry of health care, the roles of advanced practice registered nurses, also known as nurse practitioners (NPs), have evolved beyond the confines of clinical settings.
The journey into the realm of nursing often begins with a passion for healing and caring for others. However, for many, this path frequently leads to overcoming barriers to patient care, restrictions to practice, and lengthy red tape in the face of care teams that do not fully understand the scope and role of the NP.
Delving into health policy intricacies, efforts focus on understanding how legislative decisions directly impact patient care. Involvement in advocacy aims to improve health care access, promote patient-centered policies, and reduce disparities in the workforce and patient care.
Achieving sound primary care for people with multiple comorbid conditions requires the skills and abilities of all members of the health care workforce, including NPs.1 NPs have assumed an increasing role in recent decades as primary care providers for people with chronic diseases, while national trends show few physicians entering and staying in primary care.2,3,4,5 NPs are the nation’s fastest-growing primary care workforce, with nearly 90% of them trained to deliver primary care.6,7 Yet, NPs continue to experience reduced reimbursement for services compared with their physician counterparts.
Barriers to practice reduce the productivity and capacity of these health care professionals. Not permitting NPs to practice to the full extent of their licensure and education decreases the types and amounts of health care services that can be provided for people who need care.8 As noted in the Future of Nursing 2020-2030 report, this restriction also has significant implications for addressing the disparities in access to health care between rural and urban areas. A recent systemic review revealed that full practice authority is associated with higher numbers of NPs in rural areas and in primary care where there is a shortage of physicians. Full practice authority is associated with increased access to care and utilization of health care services, lower cost of care, and no decrease in quality of care.9,10 As stated in the National Council of State Boards of Nursing 2022 Environmental Scan, regulators and nurse leaders are responsible for upholding rules and regulations of nursing practice as well as ensuring that standards of care are met and patients are protected.11
Of equal importance is regulator awareness of the degree to which barriers continue to impact NP practice and limit aspects of care that directly influence care quality and access. Nursing leaders can have a significant impact on removing nonregulatory barriers to practice, such as changing outdated hospital bylaws that restrict NP practice. In turn, regulators can support efforts to remove unnecessary barriers to NP practice.
Fueled by experience in reduced practice, NPs become catalysts for change, transcending the role of health care providers. At the core of the NP role lies a commitment to patient advocacy. Beyond diagnosing and treating illnesses, NPs champion patients’ rights, ensuring their voices in health care decision-making. Advocacy efforts range from expanding access to essential services to promoting preventive care and fighting discrimination. By embracing roles as caregivers and agents of change, NPs can help the health care system emerge equitable, accessible, and patient-centric.
Ms. Kavanaugh is Assistant Dean of Graduate Nursing, University of Mount Saint Vincent, Pulmonary Critical Care Nurse Practitioner, Stamford Hospital.
References
1. McMenamin, A., Turi, E., Schlak, A., & Poghosyan, L. (2023). A Systematic Review of Outcomes Related to Nurse Practitioner-Delivered Primary Care for Multiple Chronic Conditions. Medical care research and review: MCRR, 80(6), 563–581. https://doi.org/10.1177/10775587231186720
2. Fraze T, Briggs A, Whitcomb E, Peck K, & Meara E (2020). Role of nurse practitioners in caring for patients with complex health needs. Medical Care, 58(10), 853–860. 10.1097/mlr.0000000000001364
3. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
4. Buerhaus P (2018). Nurse practitioners: A solution to America’s primary care crisis. American Enterprise Institute. https://www.aei.org/research-products/report/nurse-practitioners-a-solution-to-americas-primary-care-crisis/
5. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
6. American Association of Nurse Practitioners. (2022). Nurse practitioners in primary care. https://www.aanp.org/advocacy/advocacy-resource/position-statements/nurse-practitioners-in-primary-care
7. Auerbach, D. I. (2012). Will the NP workforce grow in the future? New forecasts and implications for healthcare delivery. Medical Care, 50(7), 606-610. doi: http://dx.doi.org/10.1097/MLR.0b013e318249d6e7
8. National Academy of Medicine . The National Academies Press; 2021. The future of nursing 2020-2030: Charting a path to achieve health equity.
9. Yang BK, Trinkoff AM, Zito JM, Burcu M, Safer DJ, Storr CL, Johantgen ME, & Idzik S (2017). Nurse practitioner independent practice authority and mental health service delivery in U.S. Community Health Centers. Psychiatric Services, 68(10), 1032–1038. 10.1176/appi.ps.201600495
10. Xue Y., Kannan V., Greener E., Smith J.A., Brasch J., Johnson B.A., Spetz J. Full scope-of-practice regulation is associated with higher supply of nurse practitioners in rural and primary care health professional shortage counties. Journal of Nursing Regulation. 2018;8(4):5–13. doi: 10.1016/S2155-8256(17)30176-X.
11. National Council of State Boards of Nursing The NCSBN 2022 environmental scan: Resiliency, achievement, and public protection. Journal of Nursing Regulation. 2022;12:S3–S55. doi: 10.1016/S2155-8256(22)00015-1.
APP INTERSECTION
In the intricate tapestry of health care, the roles of advanced practice registered nurses, also known as nurse practitioners (NPs), have evolved beyond the confines of clinical settings.
The journey into the realm of nursing often begins with a passion for healing and caring for others. However, for many, this path frequently leads to overcoming barriers to patient care, restrictions to practice, and lengthy red tape in the face of care teams that do not fully understand the scope and role of the NP.
Delving into health policy intricacies, efforts focus on understanding how legislative decisions directly impact patient care. Involvement in advocacy aims to improve health care access, promote patient-centered policies, and reduce disparities in the workforce and patient care.
Achieving sound primary care for people with multiple comorbid conditions requires the skills and abilities of all members of the health care workforce, including NPs.1 NPs have assumed an increasing role in recent decades as primary care providers for people with chronic diseases, while national trends show few physicians entering and staying in primary care.2,3,4,5 NPs are the nation’s fastest-growing primary care workforce, with nearly 90% of them trained to deliver primary care.6,7 Yet, NPs continue to experience reduced reimbursement for services compared with their physician counterparts.
Barriers to practice reduce the productivity and capacity of these health care professionals. Not permitting NPs to practice to the full extent of their licensure and education decreases the types and amounts of health care services that can be provided for people who need care.8 As noted in the Future of Nursing 2020-2030 report, this restriction also has significant implications for addressing the disparities in access to health care between rural and urban areas. A recent systemic review revealed that full practice authority is associated with higher numbers of NPs in rural areas and in primary care where there is a shortage of physicians. Full practice authority is associated with increased access to care and utilization of health care services, lower cost of care, and no decrease in quality of care.9,10 As stated in the National Council of State Boards of Nursing 2022 Environmental Scan, regulators and nurse leaders are responsible for upholding rules and regulations of nursing practice as well as ensuring that standards of care are met and patients are protected.11
Of equal importance is regulator awareness of the degree to which barriers continue to impact NP practice and limit aspects of care that directly influence care quality and access. Nursing leaders can have a significant impact on removing nonregulatory barriers to practice, such as changing outdated hospital bylaws that restrict NP practice. In turn, regulators can support efforts to remove unnecessary barriers to NP practice.
Fueled by experience in reduced practice, NPs become catalysts for change, transcending the role of health care providers. At the core of the NP role lies a commitment to patient advocacy. Beyond diagnosing and treating illnesses, NPs champion patients’ rights, ensuring their voices in health care decision-making. Advocacy efforts range from expanding access to essential services to promoting preventive care and fighting discrimination. By embracing roles as caregivers and agents of change, NPs can help the health care system emerge equitable, accessible, and patient-centric.
Ms. Kavanaugh is Assistant Dean of Graduate Nursing, University of Mount Saint Vincent, Pulmonary Critical Care Nurse Practitioner, Stamford Hospital.
References
1. McMenamin, A., Turi, E., Schlak, A., & Poghosyan, L. (2023). A Systematic Review of Outcomes Related to Nurse Practitioner-Delivered Primary Care for Multiple Chronic Conditions. Medical care research and review: MCRR, 80(6), 563–581. https://doi.org/10.1177/10775587231186720
2. Fraze T, Briggs A, Whitcomb E, Peck K, & Meara E (2020). Role of nurse practitioners in caring for patients with complex health needs. Medical Care, 58(10), 853–860. 10.1097/mlr.0000000000001364
3. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
4. Buerhaus P (2018). Nurse practitioners: A solution to America’s primary care crisis. American Enterprise Institute. https://www.aei.org/research-products/report/nurse-practitioners-a-solution-to-americas-primary-care-crisis/
5. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
6. American Association of Nurse Practitioners. (2022). Nurse practitioners in primary care. https://www.aanp.org/advocacy/advocacy-resource/position-statements/nurse-practitioners-in-primary-care
7. Auerbach, D. I. (2012). Will the NP workforce grow in the future? New forecasts and implications for healthcare delivery. Medical Care, 50(7), 606-610. doi: http://dx.doi.org/10.1097/MLR.0b013e318249d6e7
8. National Academy of Medicine . The National Academies Press; 2021. The future of nursing 2020-2030: Charting a path to achieve health equity.
9. Yang BK, Trinkoff AM, Zito JM, Burcu M, Safer DJ, Storr CL, Johantgen ME, & Idzik S (2017). Nurse practitioner independent practice authority and mental health service delivery in U.S. Community Health Centers. Psychiatric Services, 68(10), 1032–1038. 10.1176/appi.ps.201600495
10. Xue Y., Kannan V., Greener E., Smith J.A., Brasch J., Johnson B.A., Spetz J. Full scope-of-practice regulation is associated with higher supply of nurse practitioners in rural and primary care health professional shortage counties. Journal of Nursing Regulation. 2018;8(4):5–13. doi: 10.1016/S2155-8256(17)30176-X.
11. National Council of State Boards of Nursing The NCSBN 2022 environmental scan: Resiliency, achievement, and public protection. Journal of Nursing Regulation. 2022;12:S3–S55. doi: 10.1016/S2155-8256(22)00015-1.
APP INTERSECTION
In the intricate tapestry of health care, the roles of advanced practice registered nurses, also known as nurse practitioners (NPs), have evolved beyond the confines of clinical settings.
The journey into the realm of nursing often begins with a passion for healing and caring for others. However, for many, this path frequently leads to overcoming barriers to patient care, restrictions to practice, and lengthy red tape in the face of care teams that do not fully understand the scope and role of the NP.
Delving into health policy intricacies, efforts focus on understanding how legislative decisions directly impact patient care. Involvement in advocacy aims to improve health care access, promote patient-centered policies, and reduce disparities in the workforce and patient care.
Achieving sound primary care for people with multiple comorbid conditions requires the skills and abilities of all members of the health care workforce, including NPs.1 NPs have assumed an increasing role in recent decades as primary care providers for people with chronic diseases, while national trends show few physicians entering and staying in primary care.2,3,4,5 NPs are the nation’s fastest-growing primary care workforce, with nearly 90% of them trained to deliver primary care.6,7 Yet, NPs continue to experience reduced reimbursement for services compared with their physician counterparts.
Barriers to practice reduce the productivity and capacity of these health care professionals. Not permitting NPs to practice to the full extent of their licensure and education decreases the types and amounts of health care services that can be provided for people who need care.8 As noted in the Future of Nursing 2020-2030 report, this restriction also has significant implications for addressing the disparities in access to health care between rural and urban areas. A recent systemic review revealed that full practice authority is associated with higher numbers of NPs in rural areas and in primary care where there is a shortage of physicians. Full practice authority is associated with increased access to care and utilization of health care services, lower cost of care, and no decrease in quality of care.9,10 As stated in the National Council of State Boards of Nursing 2022 Environmental Scan, regulators and nurse leaders are responsible for upholding rules and regulations of nursing practice as well as ensuring that standards of care are met and patients are protected.11
Of equal importance is regulator awareness of the degree to which barriers continue to impact NP practice and limit aspects of care that directly influence care quality and access. Nursing leaders can have a significant impact on removing nonregulatory barriers to practice, such as changing outdated hospital bylaws that restrict NP practice. In turn, regulators can support efforts to remove unnecessary barriers to NP practice.
Fueled by experience in reduced practice, NPs become catalysts for change, transcending the role of health care providers. At the core of the NP role lies a commitment to patient advocacy. Beyond diagnosing and treating illnesses, NPs champion patients’ rights, ensuring their voices in health care decision-making. Advocacy efforts range from expanding access to essential services to promoting preventive care and fighting discrimination. By embracing roles as caregivers and agents of change, NPs can help the health care system emerge equitable, accessible, and patient-centric.
Ms. Kavanaugh is Assistant Dean of Graduate Nursing, University of Mount Saint Vincent, Pulmonary Critical Care Nurse Practitioner, Stamford Hospital.
References
1. McMenamin, A., Turi, E., Schlak, A., & Poghosyan, L. (2023). A Systematic Review of Outcomes Related to Nurse Practitioner-Delivered Primary Care for Multiple Chronic Conditions. Medical care research and review: MCRR, 80(6), 563–581. https://doi.org/10.1177/10775587231186720
2. Fraze T, Briggs A, Whitcomb E, Peck K, & Meara E (2020). Role of nurse practitioners in caring for patients with complex health needs. Medical Care, 58(10), 853–860. 10.1097/mlr.0000000000001364
3. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
4. Buerhaus P (2018). Nurse practitioners: A solution to America’s primary care crisis. American Enterprise Institute. https://www.aei.org/research-products/report/nurse-practitioners-a-solution-to-americas-primary-care-crisis/
5. Xue Y, Goodwin J, Adhikari D, Raji M, & Kuo Y (2017). Trends in primary care provision to medicare beneficiaries by physicians, nurse practitioners, or physician assistants: 2008–2014. Journal of Primary Care & Community Health, 8(4), 256–263. 10.1177/2150131917736634
6. American Association of Nurse Practitioners. (2022). Nurse practitioners in primary care. https://www.aanp.org/advocacy/advocacy-resource/position-statements/nurse-practitioners-in-primary-care
7. Auerbach, D. I. (2012). Will the NP workforce grow in the future? New forecasts and implications for healthcare delivery. Medical Care, 50(7), 606-610. doi: http://dx.doi.org/10.1097/MLR.0b013e318249d6e7
8. National Academy of Medicine . The National Academies Press; 2021. The future of nursing 2020-2030: Charting a path to achieve health equity.
9. Yang BK, Trinkoff AM, Zito JM, Burcu M, Safer DJ, Storr CL, Johantgen ME, & Idzik S (2017). Nurse practitioner independent practice authority and mental health service delivery in U.S. Community Health Centers. Psychiatric Services, 68(10), 1032–1038. 10.1176/appi.ps.201600495
10. Xue Y., Kannan V., Greener E., Smith J.A., Brasch J., Johnson B.A., Spetz J. Full scope-of-practice regulation is associated with higher supply of nurse practitioners in rural and primary care health professional shortage counties. Journal of Nursing Regulation. 2018;8(4):5–13. doi: 10.1016/S2155-8256(17)30176-X.
11. National Council of State Boards of Nursing The NCSBN 2022 environmental scan: Resiliency, achievement, and public protection. Journal of Nursing Regulation. 2022;12:S3–S55. doi: 10.1016/S2155-8256(22)00015-1.
Military burn pits: Their evidence and implications for respiratory health
Military service is a hazard-ridden profession. It’s easy to recognize the direct dangers from warfighting, such as gunfire and explosions, but the risks from environmental, chemical, and other occupational exposures can be harder to see.
Combustion-based waste management systems, otherwise known as “burn pits,” were used in deployed environments by the US military from the 1990s to the early 2010s. These burn pits were commonly used to eliminate plastics, electronics, munitions, metals, wood, chemicals, and even human waste. At the height of the recent conflicts in Afghanistan, Iraq, and other southwest Asia locations, more than 70% of military installations employed at least one, and nearly 4 million service members were exposed to some degree to their emissions.
Reports of burn pits being related to organic disease have garnered widespread media attention. Initially, this came through anecdotal reports of post-deployment respiratory symptoms. Over time, the conditions attributed to burn pits expanded to include newly diagnosed respiratory diseases and malignancies.
Ultimately, Congress passed the 2022 Promise to Address Comprehensive Toxins (PACT) Act, presumptively linking more than 20 diagnoses to burn pits. The PACT Act provides countless veterans access to low-cost or free medical care for their respective conditions.
What do we know about burn pits and deployment-related respiratory disease?
Data from the Millennium Cohort Study noted an approximately 40% increase in respiratory symptoms among individuals returning from deployment but no increase in the frequency of diagnosed respiratory diseases.1 This study and others definitively established a temporal relationship between deployment and respiratory symptoms. Soon after, a retrospective, observational study of service members with post-deployment respiratory symptoms found a high prevalence of constrictive bronchiolitis (CB) identified by lung biopsy.2 Patients in this group reported exposure to burn pits and a sulfur mine fire in the Mosul area while deployed. Most had normal imaging and pulmonary function testing before biopsy, confounding the clinical significance of the CB finding. The publication of this report led to increased investigation of respiratory function during and after deployment.
In a series of prospective studies that included full pulmonary function testing, impulse oscillometry, cardiopulmonary exercise testing, bronchoscopy, and, occasionally, lung biopsy to evaluate post-deployment dyspnea, only a small minority received a diagnosis of clinically significant lung disease.3,4 Additionally, when comparing spirometry and impulse oscillometry results from before and after deployment, no decline in lung function was observed in a population of service members reporting regular burn pit exposure.5 These studies suggest that at the population level, deployment does not lead to abnormalities in the structure and function of the respiratory system.
The National Academies of Sciences published two separate reviews of burn pit exposure and outcomes in 2011 and 2020.6,7 They found insufficient evidence to support a causal relationship between burn pit exposure and pulmonary disease. They highlighted studies on the composition of emissions from the area surrounding the largest military burn pit in Iraq. Levels of particulate matter, volatile organic compounds, and polycyclic aromatic hydrocarbons were elevated when compared with those of a typical American city but were similar to the pollution levels seen in the region at the time. Given these findings, they suggested ambient air pollution may have contributed more to clinically significant disease than burn pit emissions.
How do we interpret this mixed data?
At the population level, we have yet to find conclusive data directly linking burn pit exposure to the development of any respiratory disease. Does this mean that burn pits are not harmful?
Not necessarily. Research on outcomes related to burn pit exposure is challenging given the heterogeneity in exposure volume. Much of the research is retrospective and subject to recall bias. Relationships may be distorted, and the precision of reported symptoms and exposure levels is altered. Given these challenges, it’s unsurprising that evidence of causality has yet to be proven. In addition, some portion of service members has been diagnosed with respiratory disease that could be related to burn pit exposure.
What is now indisputable is that deployment to southwest Asia leads to an increase in respiratory complaints. Whether veteran respiratory symptoms are due to burn pits, ambient pollution, environmental particulate matter, or dust storms is less clinically relevant. These symptoms require attention, investigation, and management.
What does this mean for the future medical care of service members and veterans?
Many veterans with post-deployment respiratory symptoms undergo extensive evaluations without obtaining a definitive diagnosis. A recent consensus statement on deployment-related respiratory symptoms provides a framework for evaluation in such cases.8 In keeping with that statement, we recommend veterans be referred to centers with expertise in this field, such as the Department of Veterans Affairs (VA) or military health centers, when deployment-related respiratory symptoms are reported. When the evaluation does not lead to a treatable diagnosis, these centers can provide multidisciplinary care to address the symptoms of dyspnea, cough, fatigue, and exercise intolerance to improve functional status.
Despite uncertainty in the evidence or challenges in diagnosis, both the Department of Defense (DoD) and VA remain fully committed to addressing the health concerns of service members and veterans. Notably, the VA has already screened more than 5 million veterans for toxic military exposures in accordance with the PACT Act and is providing ongoing screening and care for veterans with post-deployment respiratory symptoms. Furthermore, the DoD and VA have dedicated large portions of their research budgets to investigating the impacts of exposures during military service and optimizing the care of those with respiratory symptoms. With these commitments to patient care and research, our veterans’ respiratory health can now be optimized, and future risks can be mitigated.
Dr. Haynes is Fellow, Pulmonary and Critical Care Medicine, Walter Reed National Military Medical Center, Assistant Professor of Medicine, Uniformed Services University. Dr. Nations is Pulmonary and Critical Care Medicine, Deputy Chief of Staff for Operations, Washington DC VA Medical Center, Associate Professor of Medicine, Uniformed Services University.
References
1. Smith B, Wong CA, Smith TC, Boyko EJ, Gackstetter GD; Margaret A. K. Ryan for the Millennium Cohort Study Team. Newly reported respiratory symptoms and conditions among military personnel deployed to Iraq and Afghanistan: a prospective population-based study. Am J Epidemiol. 2009;170(11):1433-1442. Preprint. Posted online October 22, 2009. PMID: 19850627. doi: 10.1093/aje/kwp287
2. King MS, Eisenberg R, Newman JH, et al. Constrictive bronchiolitis in soldiers returning from Iraq and Afghanistan. N Engl J Med. 2011;365(3):222-230. Erratum in: N Engl J Med. 2011;365(18):1749. PMID: 21774710; PMCID: PMC3296566. doi: 10.1056/NEJMoa1101388
3. Morris MJ, Dodson DW, Lucero PF, et al. Study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE). Am J Respir Crit Care Med. 2014;190(1):77-84. PMID: 24922562. doi: 10.1164/rccm.201402-0372OC
4. Morris MJ, Walter RJ, McCann ET, et al. Clinical evaluation of deployed military personnel with chronic respiratory symptoms: study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE) III. Chest. 2020;157(6):1559-1567. Preprint. Posted online February 1, 2020. PMID: 32017933. doi: 10.1016/j.chest.2020.01.024
5. Morris MJ, Skabelund AJ, Rawlins FA 3rd, Gallup RA, Aden JK, Holley AB. Study of active duty military personnel for environmental deployment exposures: pre- and post-deployment spirometry (STAMPEDE II). Respir Care. 2019;64(5):536-544. Preprint. Posted online January 8, 2019.PMID: 30622173. doi: 10.4187/respcare.06396
6. Institute of Medicine. Long-Term Health Consequences of Exposure to Burn Pits in Iraq and Afghanistan. The National Academies Press; 2011. https://doi.org/10.17226/13209
7. National Academies of Sciences, Engineering, and Medicine. Respiratory Health Effects of Airborne Hazards Exposures in the Southwest Asia Theater of Military Operations. The National Academies Press; 2020. https://doi.org/10.17226/25837
8. Falvo MJ, Sotolongo AM, Osterholzer JJ, et al. Consensus statements on deployment-related respiratory disease, inclusive of constrictive bronchiolitis: a modified Delphi study. Chest. 2023;163(3):599-609. Preprint. Posted November 4, 2022. PMID: 36343686; PMCID: PMC10154857. doi: 10.1016/j.chest.2022.10.031
Military service is a hazard-ridden profession. It’s easy to recognize the direct dangers from warfighting, such as gunfire and explosions, but the risks from environmental, chemical, and other occupational exposures can be harder to see.
Combustion-based waste management systems, otherwise known as “burn pits,” were used in deployed environments by the US military from the 1990s to the early 2010s. These burn pits were commonly used to eliminate plastics, electronics, munitions, metals, wood, chemicals, and even human waste. At the height of the recent conflicts in Afghanistan, Iraq, and other southwest Asia locations, more than 70% of military installations employed at least one, and nearly 4 million service members were exposed to some degree to their emissions.
Reports of burn pits being related to organic disease have garnered widespread media attention. Initially, this came through anecdotal reports of post-deployment respiratory symptoms. Over time, the conditions attributed to burn pits expanded to include newly diagnosed respiratory diseases and malignancies.
Ultimately, Congress passed the 2022 Promise to Address Comprehensive Toxins (PACT) Act, presumptively linking more than 20 diagnoses to burn pits. The PACT Act provides countless veterans access to low-cost or free medical care for their respective conditions.
What do we know about burn pits and deployment-related respiratory disease?
Data from the Millennium Cohort Study noted an approximately 40% increase in respiratory symptoms among individuals returning from deployment but no increase in the frequency of diagnosed respiratory diseases.1 This study and others definitively established a temporal relationship between deployment and respiratory symptoms. Soon after, a retrospective, observational study of service members with post-deployment respiratory symptoms found a high prevalence of constrictive bronchiolitis (CB) identified by lung biopsy.2 Patients in this group reported exposure to burn pits and a sulfur mine fire in the Mosul area while deployed. Most had normal imaging and pulmonary function testing before biopsy, confounding the clinical significance of the CB finding. The publication of this report led to increased investigation of respiratory function during and after deployment.
In a series of prospective studies that included full pulmonary function testing, impulse oscillometry, cardiopulmonary exercise testing, bronchoscopy, and, occasionally, lung biopsy to evaluate post-deployment dyspnea, only a small minority received a diagnosis of clinically significant lung disease.3,4 Additionally, when comparing spirometry and impulse oscillometry results from before and after deployment, no decline in lung function was observed in a population of service members reporting regular burn pit exposure.5 These studies suggest that at the population level, deployment does not lead to abnormalities in the structure and function of the respiratory system.
The National Academies of Sciences published two separate reviews of burn pit exposure and outcomes in 2011 and 2020.6,7 They found insufficient evidence to support a causal relationship between burn pit exposure and pulmonary disease. They highlighted studies on the composition of emissions from the area surrounding the largest military burn pit in Iraq. Levels of particulate matter, volatile organic compounds, and polycyclic aromatic hydrocarbons were elevated when compared with those of a typical American city but were similar to the pollution levels seen in the region at the time. Given these findings, they suggested ambient air pollution may have contributed more to clinically significant disease than burn pit emissions.
How do we interpret this mixed data?
At the population level, we have yet to find conclusive data directly linking burn pit exposure to the development of any respiratory disease. Does this mean that burn pits are not harmful?
Not necessarily. Research on outcomes related to burn pit exposure is challenging given the heterogeneity in exposure volume. Much of the research is retrospective and subject to recall bias. Relationships may be distorted, and the precision of reported symptoms and exposure levels is altered. Given these challenges, it’s unsurprising that evidence of causality has yet to be proven. In addition, some portion of service members has been diagnosed with respiratory disease that could be related to burn pit exposure.
What is now indisputable is that deployment to southwest Asia leads to an increase in respiratory complaints. Whether veteran respiratory symptoms are due to burn pits, ambient pollution, environmental particulate matter, or dust storms is less clinically relevant. These symptoms require attention, investigation, and management.
What does this mean for the future medical care of service members and veterans?
Many veterans with post-deployment respiratory symptoms undergo extensive evaluations without obtaining a definitive diagnosis. A recent consensus statement on deployment-related respiratory symptoms provides a framework for evaluation in such cases.8 In keeping with that statement, we recommend veterans be referred to centers with expertise in this field, such as the Department of Veterans Affairs (VA) or military health centers, when deployment-related respiratory symptoms are reported. When the evaluation does not lead to a treatable diagnosis, these centers can provide multidisciplinary care to address the symptoms of dyspnea, cough, fatigue, and exercise intolerance to improve functional status.
Despite uncertainty in the evidence or challenges in diagnosis, both the Department of Defense (DoD) and VA remain fully committed to addressing the health concerns of service members and veterans. Notably, the VA has already screened more than 5 million veterans for toxic military exposures in accordance with the PACT Act and is providing ongoing screening and care for veterans with post-deployment respiratory symptoms. Furthermore, the DoD and VA have dedicated large portions of their research budgets to investigating the impacts of exposures during military service and optimizing the care of those with respiratory symptoms. With these commitments to patient care and research, our veterans’ respiratory health can now be optimized, and future risks can be mitigated.
Dr. Haynes is Fellow, Pulmonary and Critical Care Medicine, Walter Reed National Military Medical Center, Assistant Professor of Medicine, Uniformed Services University. Dr. Nations is Pulmonary and Critical Care Medicine, Deputy Chief of Staff for Operations, Washington DC VA Medical Center, Associate Professor of Medicine, Uniformed Services University.
References
1. Smith B, Wong CA, Smith TC, Boyko EJ, Gackstetter GD; Margaret A. K. Ryan for the Millennium Cohort Study Team. Newly reported respiratory symptoms and conditions among military personnel deployed to Iraq and Afghanistan: a prospective population-based study. Am J Epidemiol. 2009;170(11):1433-1442. Preprint. Posted online October 22, 2009. PMID: 19850627. doi: 10.1093/aje/kwp287
2. King MS, Eisenberg R, Newman JH, et al. Constrictive bronchiolitis in soldiers returning from Iraq and Afghanistan. N Engl J Med. 2011;365(3):222-230. Erratum in: N Engl J Med. 2011;365(18):1749. PMID: 21774710; PMCID: PMC3296566. doi: 10.1056/NEJMoa1101388
3. Morris MJ, Dodson DW, Lucero PF, et al. Study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE). Am J Respir Crit Care Med. 2014;190(1):77-84. PMID: 24922562. doi: 10.1164/rccm.201402-0372OC
4. Morris MJ, Walter RJ, McCann ET, et al. Clinical evaluation of deployed military personnel with chronic respiratory symptoms: study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE) III. Chest. 2020;157(6):1559-1567. Preprint. Posted online February 1, 2020. PMID: 32017933. doi: 10.1016/j.chest.2020.01.024
5. Morris MJ, Skabelund AJ, Rawlins FA 3rd, Gallup RA, Aden JK, Holley AB. Study of active duty military personnel for environmental deployment exposures: pre- and post-deployment spirometry (STAMPEDE II). Respir Care. 2019;64(5):536-544. Preprint. Posted online January 8, 2019.PMID: 30622173. doi: 10.4187/respcare.06396
6. Institute of Medicine. Long-Term Health Consequences of Exposure to Burn Pits in Iraq and Afghanistan. The National Academies Press; 2011. https://doi.org/10.17226/13209
7. National Academies of Sciences, Engineering, and Medicine. Respiratory Health Effects of Airborne Hazards Exposures in the Southwest Asia Theater of Military Operations. The National Academies Press; 2020. https://doi.org/10.17226/25837
8. Falvo MJ, Sotolongo AM, Osterholzer JJ, et al. Consensus statements on deployment-related respiratory disease, inclusive of constrictive bronchiolitis: a modified Delphi study. Chest. 2023;163(3):599-609. Preprint. Posted November 4, 2022. PMID: 36343686; PMCID: PMC10154857. doi: 10.1016/j.chest.2022.10.031
Military service is a hazard-ridden profession. It’s easy to recognize the direct dangers from warfighting, such as gunfire and explosions, but the risks from environmental, chemical, and other occupational exposures can be harder to see.
Combustion-based waste management systems, otherwise known as “burn pits,” were used in deployed environments by the US military from the 1990s to the early 2010s. These burn pits were commonly used to eliminate plastics, electronics, munitions, metals, wood, chemicals, and even human waste. At the height of the recent conflicts in Afghanistan, Iraq, and other southwest Asia locations, more than 70% of military installations employed at least one, and nearly 4 million service members were exposed to some degree to their emissions.
Reports of burn pits being related to organic disease have garnered widespread media attention. Initially, this came through anecdotal reports of post-deployment respiratory symptoms. Over time, the conditions attributed to burn pits expanded to include newly diagnosed respiratory diseases and malignancies.
Ultimately, Congress passed the 2022 Promise to Address Comprehensive Toxins (PACT) Act, presumptively linking more than 20 diagnoses to burn pits. The PACT Act provides countless veterans access to low-cost or free medical care for their respective conditions.
What do we know about burn pits and deployment-related respiratory disease?
Data from the Millennium Cohort Study noted an approximately 40% increase in respiratory symptoms among individuals returning from deployment but no increase in the frequency of diagnosed respiratory diseases.1 This study and others definitively established a temporal relationship between deployment and respiratory symptoms. Soon after, a retrospective, observational study of service members with post-deployment respiratory symptoms found a high prevalence of constrictive bronchiolitis (CB) identified by lung biopsy.2 Patients in this group reported exposure to burn pits and a sulfur mine fire in the Mosul area while deployed. Most had normal imaging and pulmonary function testing before biopsy, confounding the clinical significance of the CB finding. The publication of this report led to increased investigation of respiratory function during and after deployment.
In a series of prospective studies that included full pulmonary function testing, impulse oscillometry, cardiopulmonary exercise testing, bronchoscopy, and, occasionally, lung biopsy to evaluate post-deployment dyspnea, only a small minority received a diagnosis of clinically significant lung disease.3,4 Additionally, when comparing spirometry and impulse oscillometry results from before and after deployment, no decline in lung function was observed in a population of service members reporting regular burn pit exposure.5 These studies suggest that at the population level, deployment does not lead to abnormalities in the structure and function of the respiratory system.
The National Academies of Sciences published two separate reviews of burn pit exposure and outcomes in 2011 and 2020.6,7 They found insufficient evidence to support a causal relationship between burn pit exposure and pulmonary disease. They highlighted studies on the composition of emissions from the area surrounding the largest military burn pit in Iraq. Levels of particulate matter, volatile organic compounds, and polycyclic aromatic hydrocarbons were elevated when compared with those of a typical American city but were similar to the pollution levels seen in the region at the time. Given these findings, they suggested ambient air pollution may have contributed more to clinically significant disease than burn pit emissions.
How do we interpret this mixed data?
At the population level, we have yet to find conclusive data directly linking burn pit exposure to the development of any respiratory disease. Does this mean that burn pits are not harmful?
Not necessarily. Research on outcomes related to burn pit exposure is challenging given the heterogeneity in exposure volume. Much of the research is retrospective and subject to recall bias. Relationships may be distorted, and the precision of reported symptoms and exposure levels is altered. Given these challenges, it’s unsurprising that evidence of causality has yet to be proven. In addition, some portion of service members has been diagnosed with respiratory disease that could be related to burn pit exposure.
What is now indisputable is that deployment to southwest Asia leads to an increase in respiratory complaints. Whether veteran respiratory symptoms are due to burn pits, ambient pollution, environmental particulate matter, or dust storms is less clinically relevant. These symptoms require attention, investigation, and management.
What does this mean for the future medical care of service members and veterans?
Many veterans with post-deployment respiratory symptoms undergo extensive evaluations without obtaining a definitive diagnosis. A recent consensus statement on deployment-related respiratory symptoms provides a framework for evaluation in such cases.8 In keeping with that statement, we recommend veterans be referred to centers with expertise in this field, such as the Department of Veterans Affairs (VA) or military health centers, when deployment-related respiratory symptoms are reported. When the evaluation does not lead to a treatable diagnosis, these centers can provide multidisciplinary care to address the symptoms of dyspnea, cough, fatigue, and exercise intolerance to improve functional status.
Despite uncertainty in the evidence or challenges in diagnosis, both the Department of Defense (DoD) and VA remain fully committed to addressing the health concerns of service members and veterans. Notably, the VA has already screened more than 5 million veterans for toxic military exposures in accordance with the PACT Act and is providing ongoing screening and care for veterans with post-deployment respiratory symptoms. Furthermore, the DoD and VA have dedicated large portions of their research budgets to investigating the impacts of exposures during military service and optimizing the care of those with respiratory symptoms. With these commitments to patient care and research, our veterans’ respiratory health can now be optimized, and future risks can be mitigated.
Dr. Haynes is Fellow, Pulmonary and Critical Care Medicine, Walter Reed National Military Medical Center, Assistant Professor of Medicine, Uniformed Services University. Dr. Nations is Pulmonary and Critical Care Medicine, Deputy Chief of Staff for Operations, Washington DC VA Medical Center, Associate Professor of Medicine, Uniformed Services University.
References
1. Smith B, Wong CA, Smith TC, Boyko EJ, Gackstetter GD; Margaret A. K. Ryan for the Millennium Cohort Study Team. Newly reported respiratory symptoms and conditions among military personnel deployed to Iraq and Afghanistan: a prospective population-based study. Am J Epidemiol. 2009;170(11):1433-1442. Preprint. Posted online October 22, 2009. PMID: 19850627. doi: 10.1093/aje/kwp287
2. King MS, Eisenberg R, Newman JH, et al. Constrictive bronchiolitis in soldiers returning from Iraq and Afghanistan. N Engl J Med. 2011;365(3):222-230. Erratum in: N Engl J Med. 2011;365(18):1749. PMID: 21774710; PMCID: PMC3296566. doi: 10.1056/NEJMoa1101388
3. Morris MJ, Dodson DW, Lucero PF, et al. Study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE). Am J Respir Crit Care Med. 2014;190(1):77-84. PMID: 24922562. doi: 10.1164/rccm.201402-0372OC
4. Morris MJ, Walter RJ, McCann ET, et al. Clinical evaluation of deployed military personnel with chronic respiratory symptoms: study of active duty military for pulmonary disease related to environmental deployment exposures (STAMPEDE) III. Chest. 2020;157(6):1559-1567. Preprint. Posted online February 1, 2020. PMID: 32017933. doi: 10.1016/j.chest.2020.01.024
5. Morris MJ, Skabelund AJ, Rawlins FA 3rd, Gallup RA, Aden JK, Holley AB. Study of active duty military personnel for environmental deployment exposures: pre- and post-deployment spirometry (STAMPEDE II). Respir Care. 2019;64(5):536-544. Preprint. Posted online January 8, 2019.PMID: 30622173. doi: 10.4187/respcare.06396
6. Institute of Medicine. Long-Term Health Consequences of Exposure to Burn Pits in Iraq and Afghanistan. The National Academies Press; 2011. https://doi.org/10.17226/13209
7. National Academies of Sciences, Engineering, and Medicine. Respiratory Health Effects of Airborne Hazards Exposures in the Southwest Asia Theater of Military Operations. The National Academies Press; 2020. https://doi.org/10.17226/25837
8. Falvo MJ, Sotolongo AM, Osterholzer JJ, et al. Consensus statements on deployment-related respiratory disease, inclusive of constrictive bronchiolitis: a modified Delphi study. Chest. 2023;163(3):599-609. Preprint. Posted November 4, 2022. PMID: 36343686; PMCID: PMC10154857. doi: 10.1016/j.chest.2022.10.031
Hospital-onset sepsis: Why the brouhaha?
A 47-year-old woman with a history of cirrhosis is admitted with an acute kidney injury and altered mental status. On the initial workup, there are no signs of infection, and dehydration is determined to be the cause of the kidney injury. There are signs of improvement in the kidney injury with hydration. On hospital day 3, the patient develops a fever (101.9 o F) with accompanying leukocytosis to 14,000. Concerned for infection, the team starts empiric broad spectrum antibiotics for presumed spontaneous bacterial peritonitis. The next day (hospital day 4), a rapid response evaluation is activated as the patient is demonstrating increasing confusion, hypotension with a systolic blood pressure of 70 mm Hg, and elevated lactic acid. The patient receives 1 L of normal saline and transfers to the ICU. The new critical care fellow, who has just read up on sepsis early management bundles, and specifically the Severe Sepsis and Septic Shock Management Bundle (SEP-1), is reviewing the chart and notices a history of multidrug-resistant organisms in her urine cultures from an admission 2 months ago. They ask of the transferring team, “When was time zero, and was the 3-hour bundle completed?”
1 The excessive cost, both of human life and monetary, has led to the commitment of significant resources to sepsis care. Improved recognition and timely intervention for sepsis have led to noteworthy improvement in mortality. Most of this effort has been directed toward patients with sepsis diagnosed in the emergency department (ED) who are presenting with community-onset sepsis (COS). A new entity, called hospital-onset sepsis (HOS), has been described recently, defined by the Centers for Disease Control and Prevention (CDC) as both infection and organ dysfunction developing more than 48 hours after hospital admission.2
A systematic review of 51 studies found approximately 23.6% of all sepsis cases are HOS. The proportion of HOS is even higher (more than 45%) in patients admitted to the ICU with sepsis.3 The outcome for this group remains comparatively poor. The hospital mortality among patients with HOS is 35%, which increases to 52% with progression to septic shock compared with 25% with COS.3 Even after adjusting for baseline factors that make one prone to developing infection in the hospital, a patient developing HOS has three-times a higher risk of dying compared with a patient who never developed sepsis and two-times a higher risk of dying compared with patients with COS.4 Furthermore, HOS utilizes more resources with significantly longer ICU and hospital stays and has five-times the hospital cost compared with COS.4
The two most crucial factors in improving sepsis outcomes, as identified by the Surviving Sepsis Campaign guidelines, are: 1) prompt identification and treatment within the first few hours of onset and 2) regular reevaluation of the patient’s response to treatment.
Prompt identification
Diagnosing sepsis in the patient who is hospitalized is challenging. Patients admitted to the hospital often have competing comorbidities, have existing organ failure, or are in a postoperative/intervention state that clouds the application and interpretation of vital sign triggers customarily used to identify sepsis. The positive predictive value for all existing sepsis definitions and diagnostic criteria is dismally low. 5 And while automated electronic sepsis alerts may improve processes of care, they still have poor positive predictive value and have not impacted patient-centered outcomes (mortality or length of stay). Furthermore, the causative microorganisms often associated with hospital-acquired infections are complex, are drug-resistant, and can have courses which further delay identification. Finally, cognitive errors, such as anchoring biases or premature diagnosis closure, can contribute to provider-level identification delays that are only further exacerbated by system issues, such as capacity constraints, staffing issues, and differing paces between wards that tend to impede time-sensitive evaluations and interventions. 4,6,7
Management
The SEP-1 core measure uses a framework of early recognition of infection and completion of the sepsis bundles in a timely manner to improve outcomes. Patients with HOS are less likely than those with COS to receive Centers for Medicare & Medicaid Services SEP-1-compliant care, including timely blood culture collection, initial and repeat lactate testing, and fluid resuscitation.8 The Surviving Sepsis Campaign has explored barriers to managing HOS. Among caregivers, these include delay in recognition, poor communication regarding change in patient status, not prioritizing treatment for sepsis, failure to measure lactate, delayed or no antimicrobial administration, and inadequate fluid resuscitation. In one study, the adherence to SEP-1 for HOS was reported at 13% compared with 39.9% in COS. The differences in initial sepsis management included timing of antimicrobials and fluid resuscitation, which accounted for 23% of observed greater mortality risk among patients with HOS compared with COS.6,8 It remains unclear how these recommendations should be applied and whether some of these recommendations confer the same benefits for patients with HOS as for those with COS. For example, administration of fluids conferred no additional benefit to patients with HOS, while rapid antimicrobial administration was shown to be associated with improved mortality in patients with HOS. Although, the optimal timing for treatment initiation and microbial coverage has not been established.
The path forward
Effective HOS management requires both individual and systematic approaches. How clinicians identify a patient with sepsis must be context-dependent. Although standard criteria exist for defining sepsis, the approach to a patient presenting to the ED from home should differ from that of a patient who has been hospitalized for several days, is postoperative, or is in the ICU on multiple forms of life support. Clinical medicine is context-dependent, and the same principles apply to sepsis management. To address the diagnostic uncertainty of the syndrome, providers must remain vigilant and maintain a clinical “iterative urgency” in diagnosing and managing sepsis. While machine learning algorithms have potential, they still rely on human intervention and interaction to navigate the complexities of HOS diagnosis.
At the system level, survival from sepsis is determined by the speed with which complex medical care is delivered and the effectiveness with which resources and personnel are mobilized and coordinated. The Hospital Sepsis Program Core Elements, released by the CDC, serves as an initial playbook to aid hospitals in establishing comprehensive sepsis improvement programs.
A second invaluable resource for hospitals in sepsis management is the rapid response team (RRT). Studies have shown that resolute RRTs can enhance patient outcomes and compliance with sepsis bundles; though, the composition and scope of these teams are crucial to their effectiveness. Responding to in-hospital emergencies and urgencies without conflicting responsibilities is an essential feature of a successful RRT. Often, they are familiar with bundles, protocols, and documentation, and members of these teams can offer clinical and/or technical expertise as well as support active participation and reengagement with bedside staff, which fosters trust and collaboration. This partnership is key, as these interactions instill a common mission and foster a culture of sepsis improvement that is required to achieve sustained success and improved patient outcomes.
Dr. Dugar is Director, Point-of-Care Ultrasound, Department of Critical Care, Respiratory Institute, Assistant Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH. Dr. Jayaprakash is Associate Medical Director, Quality, Emergency Medicine, Physician Lead, Henry Ford Health Sepsis Program. Dr. Reilkoff is Executive Medical Director of Critical Care, M Health Fairview Intensive Care Units, Director of Acting Internship in Critical Care, University of Minnesota Medical School, Associate Professor of Medicine and Surgery, University of Minnesota. Dr. Duggal is Vice-Chair, Department of Critical Care, Respiratory Institute, Director, Critical Care Clinical Research, Associate Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH
References
1. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA. 2016;315(8):801-810.
2. Ginestra JC, Coz Yataco AO, Dugar SP, Dettmer MR. Hospital-onset sepsis warrants expanded investigation and consideration as a unique clinical entity. Chest. 2024;S0012-3692(24):00039-4.
3. Markwart R, Saito H, Harder T, et al. Epidemiology and burden of sepsis acquired in hospitals and intensive care units: a systematic review and meta-analysis. Intensive Care Med. 2020;46(8):1536-1551.
4. Rhee C, Wang R, Zhang Z, et al. Epidemiology of hospital-onset versus community-onset sepsis in U.S. hospitals and association with mortality: a retrospective analysis using electronic clinical data. Crit Care Med. 2019;47(9):1169-1176.
5. Wong A, Otles E, Donnelly JP, et al. External validation of a widely implemented proprietary sepsis prediction model in hospitalized patients. JAMA Intern Med. 2021;181(8):1065-1070.
6. Baghdadi JD, Brook RH, Uslan DZ, et al. Association of a care bundle for early sepsis management with mortality among patients with hospital-onset or community-onset sepsis. JAMA Intern Med. 2020;180(5):707-716.
7. Baghdadi JD, Wong MD, Uslan DZ, et al. Adherence to the SEP-1 sepsis bundle in hospital-onset v. community-onset sepsis: a multicenter retrospective cohort study. J Gen Intern Med. 2020;35(4):1153-1160.
8. Basheer A. Patients with hospital-onset sepsis are less likely to receive sepsis bundle care than those with community-onset sepsis. Evid Based Nurs. 2021;24(3):99.
A 47-year-old woman with a history of cirrhosis is admitted with an acute kidney injury and altered mental status. On the initial workup, there are no signs of infection, and dehydration is determined to be the cause of the kidney injury. There are signs of improvement in the kidney injury with hydration. On hospital day 3, the patient develops a fever (101.9 o F) with accompanying leukocytosis to 14,000. Concerned for infection, the team starts empiric broad spectrum antibiotics for presumed spontaneous bacterial peritonitis. The next day (hospital day 4), a rapid response evaluation is activated as the patient is demonstrating increasing confusion, hypotension with a systolic blood pressure of 70 mm Hg, and elevated lactic acid. The patient receives 1 L of normal saline and transfers to the ICU. The new critical care fellow, who has just read up on sepsis early management bundles, and specifically the Severe Sepsis and Septic Shock Management Bundle (SEP-1), is reviewing the chart and notices a history of multidrug-resistant organisms in her urine cultures from an admission 2 months ago. They ask of the transferring team, “When was time zero, and was the 3-hour bundle completed?”
1 The excessive cost, both of human life and monetary, has led to the commitment of significant resources to sepsis care. Improved recognition and timely intervention for sepsis have led to noteworthy improvement in mortality. Most of this effort has been directed toward patients with sepsis diagnosed in the emergency department (ED) who are presenting with community-onset sepsis (COS). A new entity, called hospital-onset sepsis (HOS), has been described recently, defined by the Centers for Disease Control and Prevention (CDC) as both infection and organ dysfunction developing more than 48 hours after hospital admission.2
A systematic review of 51 studies found approximately 23.6% of all sepsis cases are HOS. The proportion of HOS is even higher (more than 45%) in patients admitted to the ICU with sepsis.3 The outcome for this group remains comparatively poor. The hospital mortality among patients with HOS is 35%, which increases to 52% with progression to septic shock compared with 25% with COS.3 Even after adjusting for baseline factors that make one prone to developing infection in the hospital, a patient developing HOS has three-times a higher risk of dying compared with a patient who never developed sepsis and two-times a higher risk of dying compared with patients with COS.4 Furthermore, HOS utilizes more resources with significantly longer ICU and hospital stays and has five-times the hospital cost compared with COS.4
The two most crucial factors in improving sepsis outcomes, as identified by the Surviving Sepsis Campaign guidelines, are: 1) prompt identification and treatment within the first few hours of onset and 2) regular reevaluation of the patient’s response to treatment.
Prompt identification
Diagnosing sepsis in the patient who is hospitalized is challenging. Patients admitted to the hospital often have competing comorbidities, have existing organ failure, or are in a postoperative/intervention state that clouds the application and interpretation of vital sign triggers customarily used to identify sepsis. The positive predictive value for all existing sepsis definitions and diagnostic criteria is dismally low. 5 And while automated electronic sepsis alerts may improve processes of care, they still have poor positive predictive value and have not impacted patient-centered outcomes (mortality or length of stay). Furthermore, the causative microorganisms often associated with hospital-acquired infections are complex, are drug-resistant, and can have courses which further delay identification. Finally, cognitive errors, such as anchoring biases or premature diagnosis closure, can contribute to provider-level identification delays that are only further exacerbated by system issues, such as capacity constraints, staffing issues, and differing paces between wards that tend to impede time-sensitive evaluations and interventions. 4,6,7
Management
The SEP-1 core measure uses a framework of early recognition of infection and completion of the sepsis bundles in a timely manner to improve outcomes. Patients with HOS are less likely than those with COS to receive Centers for Medicare & Medicaid Services SEP-1-compliant care, including timely blood culture collection, initial and repeat lactate testing, and fluid resuscitation.8 The Surviving Sepsis Campaign has explored barriers to managing HOS. Among caregivers, these include delay in recognition, poor communication regarding change in patient status, not prioritizing treatment for sepsis, failure to measure lactate, delayed or no antimicrobial administration, and inadequate fluid resuscitation. In one study, the adherence to SEP-1 for HOS was reported at 13% compared with 39.9% in COS. The differences in initial sepsis management included timing of antimicrobials and fluid resuscitation, which accounted for 23% of observed greater mortality risk among patients with HOS compared with COS.6,8 It remains unclear how these recommendations should be applied and whether some of these recommendations confer the same benefits for patients with HOS as for those with COS. For example, administration of fluids conferred no additional benefit to patients with HOS, while rapid antimicrobial administration was shown to be associated with improved mortality in patients with HOS. Although, the optimal timing for treatment initiation and microbial coverage has not been established.
The path forward
Effective HOS management requires both individual and systematic approaches. How clinicians identify a patient with sepsis must be context-dependent. Although standard criteria exist for defining sepsis, the approach to a patient presenting to the ED from home should differ from that of a patient who has been hospitalized for several days, is postoperative, or is in the ICU on multiple forms of life support. Clinical medicine is context-dependent, and the same principles apply to sepsis management. To address the diagnostic uncertainty of the syndrome, providers must remain vigilant and maintain a clinical “iterative urgency” in diagnosing and managing sepsis. While machine learning algorithms have potential, they still rely on human intervention and interaction to navigate the complexities of HOS diagnosis.
At the system level, survival from sepsis is determined by the speed with which complex medical care is delivered and the effectiveness with which resources and personnel are mobilized and coordinated. The Hospital Sepsis Program Core Elements, released by the CDC, serves as an initial playbook to aid hospitals in establishing comprehensive sepsis improvement programs.
A second invaluable resource for hospitals in sepsis management is the rapid response team (RRT). Studies have shown that resolute RRTs can enhance patient outcomes and compliance with sepsis bundles; though, the composition and scope of these teams are crucial to their effectiveness. Responding to in-hospital emergencies and urgencies without conflicting responsibilities is an essential feature of a successful RRT. Often, they are familiar with bundles, protocols, and documentation, and members of these teams can offer clinical and/or technical expertise as well as support active participation and reengagement with bedside staff, which fosters trust and collaboration. This partnership is key, as these interactions instill a common mission and foster a culture of sepsis improvement that is required to achieve sustained success and improved patient outcomes.
Dr. Dugar is Director, Point-of-Care Ultrasound, Department of Critical Care, Respiratory Institute, Assistant Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH. Dr. Jayaprakash is Associate Medical Director, Quality, Emergency Medicine, Physician Lead, Henry Ford Health Sepsis Program. Dr. Reilkoff is Executive Medical Director of Critical Care, M Health Fairview Intensive Care Units, Director of Acting Internship in Critical Care, University of Minnesota Medical School, Associate Professor of Medicine and Surgery, University of Minnesota. Dr. Duggal is Vice-Chair, Department of Critical Care, Respiratory Institute, Director, Critical Care Clinical Research, Associate Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH
References
1. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA. 2016;315(8):801-810.
2. Ginestra JC, Coz Yataco AO, Dugar SP, Dettmer MR. Hospital-onset sepsis warrants expanded investigation and consideration as a unique clinical entity. Chest. 2024;S0012-3692(24):00039-4.
3. Markwart R, Saito H, Harder T, et al. Epidemiology and burden of sepsis acquired in hospitals and intensive care units: a systematic review and meta-analysis. Intensive Care Med. 2020;46(8):1536-1551.
4. Rhee C, Wang R, Zhang Z, et al. Epidemiology of hospital-onset versus community-onset sepsis in U.S. hospitals and association with mortality: a retrospective analysis using electronic clinical data. Crit Care Med. 2019;47(9):1169-1176.
5. Wong A, Otles E, Donnelly JP, et al. External validation of a widely implemented proprietary sepsis prediction model in hospitalized patients. JAMA Intern Med. 2021;181(8):1065-1070.
6. Baghdadi JD, Brook RH, Uslan DZ, et al. Association of a care bundle for early sepsis management with mortality among patients with hospital-onset or community-onset sepsis. JAMA Intern Med. 2020;180(5):707-716.
7. Baghdadi JD, Wong MD, Uslan DZ, et al. Adherence to the SEP-1 sepsis bundle in hospital-onset v. community-onset sepsis: a multicenter retrospective cohort study. J Gen Intern Med. 2020;35(4):1153-1160.
8. Basheer A. Patients with hospital-onset sepsis are less likely to receive sepsis bundle care than those with community-onset sepsis. Evid Based Nurs. 2021;24(3):99.
A 47-year-old woman with a history of cirrhosis is admitted with an acute kidney injury and altered mental status. On the initial workup, there are no signs of infection, and dehydration is determined to be the cause of the kidney injury. There are signs of improvement in the kidney injury with hydration. On hospital day 3, the patient develops a fever (101.9 o F) with accompanying leukocytosis to 14,000. Concerned for infection, the team starts empiric broad spectrum antibiotics for presumed spontaneous bacterial peritonitis. The next day (hospital day 4), a rapid response evaluation is activated as the patient is demonstrating increasing confusion, hypotension with a systolic blood pressure of 70 mm Hg, and elevated lactic acid. The patient receives 1 L of normal saline and transfers to the ICU. The new critical care fellow, who has just read up on sepsis early management bundles, and specifically the Severe Sepsis and Septic Shock Management Bundle (SEP-1), is reviewing the chart and notices a history of multidrug-resistant organisms in her urine cultures from an admission 2 months ago. They ask of the transferring team, “When was time zero, and was the 3-hour bundle completed?”
1 The excessive cost, both of human life and monetary, has led to the commitment of significant resources to sepsis care. Improved recognition and timely intervention for sepsis have led to noteworthy improvement in mortality. Most of this effort has been directed toward patients with sepsis diagnosed in the emergency department (ED) who are presenting with community-onset sepsis (COS). A new entity, called hospital-onset sepsis (HOS), has been described recently, defined by the Centers for Disease Control and Prevention (CDC) as both infection and organ dysfunction developing more than 48 hours after hospital admission.2
A systematic review of 51 studies found approximately 23.6% of all sepsis cases are HOS. The proportion of HOS is even higher (more than 45%) in patients admitted to the ICU with sepsis.3 The outcome for this group remains comparatively poor. The hospital mortality among patients with HOS is 35%, which increases to 52% with progression to septic shock compared with 25% with COS.3 Even after adjusting for baseline factors that make one prone to developing infection in the hospital, a patient developing HOS has three-times a higher risk of dying compared with a patient who never developed sepsis and two-times a higher risk of dying compared with patients with COS.4 Furthermore, HOS utilizes more resources with significantly longer ICU and hospital stays and has five-times the hospital cost compared with COS.4
The two most crucial factors in improving sepsis outcomes, as identified by the Surviving Sepsis Campaign guidelines, are: 1) prompt identification and treatment within the first few hours of onset and 2) regular reevaluation of the patient’s response to treatment.
Prompt identification
Diagnosing sepsis in the patient who is hospitalized is challenging. Patients admitted to the hospital often have competing comorbidities, have existing organ failure, or are in a postoperative/intervention state that clouds the application and interpretation of vital sign triggers customarily used to identify sepsis. The positive predictive value for all existing sepsis definitions and diagnostic criteria is dismally low. 5 And while automated electronic sepsis alerts may improve processes of care, they still have poor positive predictive value and have not impacted patient-centered outcomes (mortality or length of stay). Furthermore, the causative microorganisms often associated with hospital-acquired infections are complex, are drug-resistant, and can have courses which further delay identification. Finally, cognitive errors, such as anchoring biases or premature diagnosis closure, can contribute to provider-level identification delays that are only further exacerbated by system issues, such as capacity constraints, staffing issues, and differing paces between wards that tend to impede time-sensitive evaluations and interventions. 4,6,7
Management
The SEP-1 core measure uses a framework of early recognition of infection and completion of the sepsis bundles in a timely manner to improve outcomes. Patients with HOS are less likely than those with COS to receive Centers for Medicare & Medicaid Services SEP-1-compliant care, including timely blood culture collection, initial and repeat lactate testing, and fluid resuscitation.8 The Surviving Sepsis Campaign has explored barriers to managing HOS. Among caregivers, these include delay in recognition, poor communication regarding change in patient status, not prioritizing treatment for sepsis, failure to measure lactate, delayed or no antimicrobial administration, and inadequate fluid resuscitation. In one study, the adherence to SEP-1 for HOS was reported at 13% compared with 39.9% in COS. The differences in initial sepsis management included timing of antimicrobials and fluid resuscitation, which accounted for 23% of observed greater mortality risk among patients with HOS compared with COS.6,8 It remains unclear how these recommendations should be applied and whether some of these recommendations confer the same benefits for patients with HOS as for those with COS. For example, administration of fluids conferred no additional benefit to patients with HOS, while rapid antimicrobial administration was shown to be associated with improved mortality in patients with HOS. Although, the optimal timing for treatment initiation and microbial coverage has not been established.
The path forward
Effective HOS management requires both individual and systematic approaches. How clinicians identify a patient with sepsis must be context-dependent. Although standard criteria exist for defining sepsis, the approach to a patient presenting to the ED from home should differ from that of a patient who has been hospitalized for several days, is postoperative, or is in the ICU on multiple forms of life support. Clinical medicine is context-dependent, and the same principles apply to sepsis management. To address the diagnostic uncertainty of the syndrome, providers must remain vigilant and maintain a clinical “iterative urgency” in diagnosing and managing sepsis. While machine learning algorithms have potential, they still rely on human intervention and interaction to navigate the complexities of HOS diagnosis.
At the system level, survival from sepsis is determined by the speed with which complex medical care is delivered and the effectiveness with which resources and personnel are mobilized and coordinated. The Hospital Sepsis Program Core Elements, released by the CDC, serves as an initial playbook to aid hospitals in establishing comprehensive sepsis improvement programs.
A second invaluable resource for hospitals in sepsis management is the rapid response team (RRT). Studies have shown that resolute RRTs can enhance patient outcomes and compliance with sepsis bundles; though, the composition and scope of these teams are crucial to their effectiveness. Responding to in-hospital emergencies and urgencies without conflicting responsibilities is an essential feature of a successful RRT. Often, they are familiar with bundles, protocols, and documentation, and members of these teams can offer clinical and/or technical expertise as well as support active participation and reengagement with bedside staff, which fosters trust and collaboration. This partnership is key, as these interactions instill a common mission and foster a culture of sepsis improvement that is required to achieve sustained success and improved patient outcomes.
Dr. Dugar is Director, Point-of-Care Ultrasound, Department of Critical Care, Respiratory Institute, Assistant Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH. Dr. Jayaprakash is Associate Medical Director, Quality, Emergency Medicine, Physician Lead, Henry Ford Health Sepsis Program. Dr. Reilkoff is Executive Medical Director of Critical Care, M Health Fairview Intensive Care Units, Director of Acting Internship in Critical Care, University of Minnesota Medical School, Associate Professor of Medicine and Surgery, University of Minnesota. Dr. Duggal is Vice-Chair, Department of Critical Care, Respiratory Institute, Director, Critical Care Clinical Research, Associate Professor, Cleveland Clinic Lerner College of Medicine, Cleveland, OH
References
1. Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA. 2016;315(8):801-810.
2. Ginestra JC, Coz Yataco AO, Dugar SP, Dettmer MR. Hospital-onset sepsis warrants expanded investigation and consideration as a unique clinical entity. Chest. 2024;S0012-3692(24):00039-4.
3. Markwart R, Saito H, Harder T, et al. Epidemiology and burden of sepsis acquired in hospitals and intensive care units: a systematic review and meta-analysis. Intensive Care Med. 2020;46(8):1536-1551.
4. Rhee C, Wang R, Zhang Z, et al. Epidemiology of hospital-onset versus community-onset sepsis in U.S. hospitals and association with mortality: a retrospective analysis using electronic clinical data. Crit Care Med. 2019;47(9):1169-1176.
5. Wong A, Otles E, Donnelly JP, et al. External validation of a widely implemented proprietary sepsis prediction model in hospitalized patients. JAMA Intern Med. 2021;181(8):1065-1070.
6. Baghdadi JD, Brook RH, Uslan DZ, et al. Association of a care bundle for early sepsis management with mortality among patients with hospital-onset or community-onset sepsis. JAMA Intern Med. 2020;180(5):707-716.
7. Baghdadi JD, Wong MD, Uslan DZ, et al. Adherence to the SEP-1 sepsis bundle in hospital-onset v. community-onset sepsis: a multicenter retrospective cohort study. J Gen Intern Med. 2020;35(4):1153-1160.
8. Basheer A. Patients with hospital-onset sepsis are less likely to receive sepsis bundle care than those with community-onset sepsis. Evid Based Nurs. 2021;24(3):99.
AI Tools Could Change Pulmonological Imaging and Prognosis
MADRID — Artificial intelligence (AI) can enhance endobronchial ultrasound (EBUS) image processing and new techniques such as cryoEBUS to achieve significant diagnostic and prognostic breakthroughs in interventional pulmonology and general pulmonology.
Pulmonologists are witnessing a surge in new technologies for endoscopy and pulmonology in general. Some, such as AI, robotic bronchoscopy, radiomics, or improvements in electromagnetic bronchial navigation, are minimally invasive diagnostic techniques that significantly enhance the characterization of lung lesions, said Virginia Pajares, MD, a member of the Catalan Society of Pulmonology and coordinator of the Bronchoscopy Unit at Hospital de Sant Pau in Barcelona, Spain. She spoke at the XLI Pneumological Day of the Catalan Society of Pulmonology in Vilanova i la Geltrú, Spain.
Regarding AI, pulmonologists “already have platforms that enable the calculation of the malignancy risk of lung lesions and mediastinal adenopathies. In addition, some devices that allow for an initial radiological assessment of lung nodules are starting to be used,” said Dr. Pajares.
Radiomics: Histology and Markers
“At an endoscopic level, some studies have confirmed the ability of AI applied to imaging to differentiate between benign and malignant lesions, although currently the studies are limited and in the initial stages,” said Dr. Pajares. “AI in interventional pulmonology will be highly beneficial in image interpretation and patient assessment for those who require more invasive diagnostic techniques or for follow-up.”
Regarding the application of AI in medicine, “we lack knowledge and require specific training, especially concerning the learning curve of different technologies, such as electromagnetic navigation, cryoEBUS, or robotic bronchoscopy, which require significant training efforts,” said Dr. Pajares. “The use of AI without a specific goal, that is, creating a mathematical algorithm and feeding it with clinical patient data without control and validation, can lead to inaccurate conclusions. Additionally, we need to determine how to input patient data into these systems to avoid ethical issues, and, of course, legislation on this matter is essential.”
Electromagnetic Navigation
Bronchial electromagnetic navigation is a bronchoscopic technique that allows access to peripheral lung lesions. “It involves virtual route planning using the patient’s chest CT scan and subsequently performing bronchoscopy with navigation using a dirigible electromagnetic probe that allows access to the lesion,” Dr. Pajares explained. “Currently, we have navigators that can incorporate imaging techniques (fluoroscopy or cone-beam CT) to immediately correct discrepancies observed during navigation.”
These new technologies enable a greater number of precise diagnoses and may bring greater patient safety. Studies like NAVIGATE, which was published in 2022 by Folch and colleagues, confirm the diagnostic possibilities and performance of electromagnetic navigation.
In this prospective study, which followed patients for 24 months, the indications are broad. “Its most common use is as a diagnostic technique for peripheral lung nodules and for marking lung lesions for surgical resection or marking for radiotherapy field fiducial placement,” said Dr. Pajares. “Results are also beginning to be published on the treatment of lung lesions using electromagnetic navigation ablation, demonstrating its safety and efficacy in this area.”
Nonsolid Imaging
The challenges in navigation include “improving the diagnosis of lung lesions that are nonsolid, known as ground glass opacities, and verifying it as an additional treatment option for lung nodules in patients who are not candidates for surgical resection,” said Dr. Pajares.
Tess Kramer, PhD, of Amsterdam University Medical Center, Amsterdam, the Netherlands, advocates for the combined use of different technologies to have a beneficial impact on patients’ clinical outcomes.
Robotic bronchoscopy has been implemented in the United States for several years, enhancing the precision of lung nodule diagnosis. However, “currently, there are no clear differences in the diagnostic performance of robotic bronchoscopy compared with navigation in general. Soon, there will be studies to assess in which type of nodules one technique may be more cost-effective.” No centers in Spain have this technology yet, “although some are already evaluating the acquisition of robotic bronchoscopy; it’s only a matter of time,” said Dr. Pajares.
Improvements in echobronchoscopy technology include high-quality image processors and smaller device calibers with greater angulation to diagnose lesions and hard-to-reach adenopathies. From an imaging perspective, AI, combined with the creation of risk calculators, could enable the prediction of lymph node malignancy.
Moreover, the use of small-caliber cryoprobes (1.1 mm) for obtaining samples of adenopathies (cryoEBUS) has enhanced diagnosis by enabling larger tissue samples. Current studies are being conducted to confirm the utility of cryoEBUS in pathologies requiring extensive molecular and immunohistochemical studies for diagnosing lymphoproliferative syndromes or neoplasms.
In a different context, liquid biopsy, a recent laboratory technology unrelated to bronchoscopy, allows the analysis of blood/pleural fluid samples that were extracted using the aforementioned technologies to locate tumor cells and differentiate between malignancy and benignity.
The Challenge of Pneumonitis
Samantha Aso, MD, a pulmonologist, member of the Catalan Society of Pulmonology, and specialist at the Lung Unit of Bellvitge University Hospital in Barcelona, Spain, discussed the challenge of managing pneumonitis in oncology patients.
Pneumonitis is an inflammation of the lungs that can be secondary to treatments, such as oncological therapy, which is the leading cause in 15%-50% of cases. Most oncological treatments can result in this process, including chemotherapy, chest radiotherapy, targeted therapies, conjugated monoclonal antibodies, and monotherapy.
To date, there is no known idiosyncratic cause of this process, except for autoimmune diseases. Pulmonary fibrosis is believed to be a risk factor. “Patients with interstitial lung disease and pulmonary fibrosis have been found to have a higher mortality risk due to pneumonitis. Consequently, cancer treatment cannot be administered to these patients,” said Dr. Aso.
Pulmonologists face the challenge of managing pneumonitis secondary to monotherapy, which currently is treated with cortisone. Patients respond well to this medication, but after corticosteroid withdrawal, reinflammation may occur. “In pneumonitis patients, oncological treatment (monotherapy) should be suspended while pulmonologists manage the pneumonitis with corticosteroids. However, we are uncertain about how rapidly or slowly to reduce the dosage. We cannot taper these doses as quickly as desired because reinflammation may occur, and to date, there are no alternative treatments apart from corticosteroids,” said Dr. Aso.
She noted that excellent survival results are achieved with monotherapy, but further research is required on the safety of antineoplastic drugs as a secondary endpoint. “Suspending oncological treatment due to pneumonitis means that patients are not receiving adequate cancer treatment, which has a significant psychological impact that also needs to be addressed,” Dr. Aso concluded.
Dr. Pajares and Dr. Aso declared no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
MADRID — Artificial intelligence (AI) can enhance endobronchial ultrasound (EBUS) image processing and new techniques such as cryoEBUS to achieve significant diagnostic and prognostic breakthroughs in interventional pulmonology and general pulmonology.
Pulmonologists are witnessing a surge in new technologies for endoscopy and pulmonology in general. Some, such as AI, robotic bronchoscopy, radiomics, or improvements in electromagnetic bronchial navigation, are minimally invasive diagnostic techniques that significantly enhance the characterization of lung lesions, said Virginia Pajares, MD, a member of the Catalan Society of Pulmonology and coordinator of the Bronchoscopy Unit at Hospital de Sant Pau in Barcelona, Spain. She spoke at the XLI Pneumological Day of the Catalan Society of Pulmonology in Vilanova i la Geltrú, Spain.
Regarding AI, pulmonologists “already have platforms that enable the calculation of the malignancy risk of lung lesions and mediastinal adenopathies. In addition, some devices that allow for an initial radiological assessment of lung nodules are starting to be used,” said Dr. Pajares.
Radiomics: Histology and Markers
“At an endoscopic level, some studies have confirmed the ability of AI applied to imaging to differentiate between benign and malignant lesions, although currently the studies are limited and in the initial stages,” said Dr. Pajares. “AI in interventional pulmonology will be highly beneficial in image interpretation and patient assessment for those who require more invasive diagnostic techniques or for follow-up.”
Regarding the application of AI in medicine, “we lack knowledge and require specific training, especially concerning the learning curve of different technologies, such as electromagnetic navigation, cryoEBUS, or robotic bronchoscopy, which require significant training efforts,” said Dr. Pajares. “The use of AI without a specific goal, that is, creating a mathematical algorithm and feeding it with clinical patient data without control and validation, can lead to inaccurate conclusions. Additionally, we need to determine how to input patient data into these systems to avoid ethical issues, and, of course, legislation on this matter is essential.”
Electromagnetic Navigation
Bronchial electromagnetic navigation is a bronchoscopic technique that allows access to peripheral lung lesions. “It involves virtual route planning using the patient’s chest CT scan and subsequently performing bronchoscopy with navigation using a dirigible electromagnetic probe that allows access to the lesion,” Dr. Pajares explained. “Currently, we have navigators that can incorporate imaging techniques (fluoroscopy or cone-beam CT) to immediately correct discrepancies observed during navigation.”
These new technologies enable a greater number of precise diagnoses and may bring greater patient safety. Studies like NAVIGATE, which was published in 2022 by Folch and colleagues, confirm the diagnostic possibilities and performance of electromagnetic navigation.
In this prospective study, which followed patients for 24 months, the indications are broad. “Its most common use is as a diagnostic technique for peripheral lung nodules and for marking lung lesions for surgical resection or marking for radiotherapy field fiducial placement,” said Dr. Pajares. “Results are also beginning to be published on the treatment of lung lesions using electromagnetic navigation ablation, demonstrating its safety and efficacy in this area.”
Nonsolid Imaging
The challenges in navigation include “improving the diagnosis of lung lesions that are nonsolid, known as ground glass opacities, and verifying it as an additional treatment option for lung nodules in patients who are not candidates for surgical resection,” said Dr. Pajares.
Tess Kramer, PhD, of Amsterdam University Medical Center, Amsterdam, the Netherlands, advocates for the combined use of different technologies to have a beneficial impact on patients’ clinical outcomes.
Robotic bronchoscopy has been implemented in the United States for several years, enhancing the precision of lung nodule diagnosis. However, “currently, there are no clear differences in the diagnostic performance of robotic bronchoscopy compared with navigation in general. Soon, there will be studies to assess in which type of nodules one technique may be more cost-effective.” No centers in Spain have this technology yet, “although some are already evaluating the acquisition of robotic bronchoscopy; it’s only a matter of time,” said Dr. Pajares.
Improvements in echobronchoscopy technology include high-quality image processors and smaller device calibers with greater angulation to diagnose lesions and hard-to-reach adenopathies. From an imaging perspective, AI, combined with the creation of risk calculators, could enable the prediction of lymph node malignancy.
Moreover, the use of small-caliber cryoprobes (1.1 mm) for obtaining samples of adenopathies (cryoEBUS) has enhanced diagnosis by enabling larger tissue samples. Current studies are being conducted to confirm the utility of cryoEBUS in pathologies requiring extensive molecular and immunohistochemical studies for diagnosing lymphoproliferative syndromes or neoplasms.
In a different context, liquid biopsy, a recent laboratory technology unrelated to bronchoscopy, allows the analysis of blood/pleural fluid samples that were extracted using the aforementioned technologies to locate tumor cells and differentiate between malignancy and benignity.
The Challenge of Pneumonitis
Samantha Aso, MD, a pulmonologist, member of the Catalan Society of Pulmonology, and specialist at the Lung Unit of Bellvitge University Hospital in Barcelona, Spain, discussed the challenge of managing pneumonitis in oncology patients.
Pneumonitis is an inflammation of the lungs that can be secondary to treatments, such as oncological therapy, which is the leading cause in 15%-50% of cases. Most oncological treatments can result in this process, including chemotherapy, chest radiotherapy, targeted therapies, conjugated monoclonal antibodies, and monotherapy.
To date, there is no known idiosyncratic cause of this process, except for autoimmune diseases. Pulmonary fibrosis is believed to be a risk factor. “Patients with interstitial lung disease and pulmonary fibrosis have been found to have a higher mortality risk due to pneumonitis. Consequently, cancer treatment cannot be administered to these patients,” said Dr. Aso.
Pulmonologists face the challenge of managing pneumonitis secondary to monotherapy, which currently is treated with cortisone. Patients respond well to this medication, but after corticosteroid withdrawal, reinflammation may occur. “In pneumonitis patients, oncological treatment (monotherapy) should be suspended while pulmonologists manage the pneumonitis with corticosteroids. However, we are uncertain about how rapidly or slowly to reduce the dosage. We cannot taper these doses as quickly as desired because reinflammation may occur, and to date, there are no alternative treatments apart from corticosteroids,” said Dr. Aso.
She noted that excellent survival results are achieved with monotherapy, but further research is required on the safety of antineoplastic drugs as a secondary endpoint. “Suspending oncological treatment due to pneumonitis means that patients are not receiving adequate cancer treatment, which has a significant psychological impact that also needs to be addressed,” Dr. Aso concluded.
Dr. Pajares and Dr. Aso declared no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
MADRID — Artificial intelligence (AI) can enhance endobronchial ultrasound (EBUS) image processing and new techniques such as cryoEBUS to achieve significant diagnostic and prognostic breakthroughs in interventional pulmonology and general pulmonology.
Pulmonologists are witnessing a surge in new technologies for endoscopy and pulmonology in general. Some, such as AI, robotic bronchoscopy, radiomics, or improvements in electromagnetic bronchial navigation, are minimally invasive diagnostic techniques that significantly enhance the characterization of lung lesions, said Virginia Pajares, MD, a member of the Catalan Society of Pulmonology and coordinator of the Bronchoscopy Unit at Hospital de Sant Pau in Barcelona, Spain. She spoke at the XLI Pneumological Day of the Catalan Society of Pulmonology in Vilanova i la Geltrú, Spain.
Regarding AI, pulmonologists “already have platforms that enable the calculation of the malignancy risk of lung lesions and mediastinal adenopathies. In addition, some devices that allow for an initial radiological assessment of lung nodules are starting to be used,” said Dr. Pajares.
Radiomics: Histology and Markers
“At an endoscopic level, some studies have confirmed the ability of AI applied to imaging to differentiate between benign and malignant lesions, although currently the studies are limited and in the initial stages,” said Dr. Pajares. “AI in interventional pulmonology will be highly beneficial in image interpretation and patient assessment for those who require more invasive diagnostic techniques or for follow-up.”
Regarding the application of AI in medicine, “we lack knowledge and require specific training, especially concerning the learning curve of different technologies, such as electromagnetic navigation, cryoEBUS, or robotic bronchoscopy, which require significant training efforts,” said Dr. Pajares. “The use of AI without a specific goal, that is, creating a mathematical algorithm and feeding it with clinical patient data without control and validation, can lead to inaccurate conclusions. Additionally, we need to determine how to input patient data into these systems to avoid ethical issues, and, of course, legislation on this matter is essential.”
Electromagnetic Navigation
Bronchial electromagnetic navigation is a bronchoscopic technique that allows access to peripheral lung lesions. “It involves virtual route planning using the patient’s chest CT scan and subsequently performing bronchoscopy with navigation using a dirigible electromagnetic probe that allows access to the lesion,” Dr. Pajares explained. “Currently, we have navigators that can incorporate imaging techniques (fluoroscopy or cone-beam CT) to immediately correct discrepancies observed during navigation.”
These new technologies enable a greater number of precise diagnoses and may bring greater patient safety. Studies like NAVIGATE, which was published in 2022 by Folch and colleagues, confirm the diagnostic possibilities and performance of electromagnetic navigation.
In this prospective study, which followed patients for 24 months, the indications are broad. “Its most common use is as a diagnostic technique for peripheral lung nodules and for marking lung lesions for surgical resection or marking for radiotherapy field fiducial placement,” said Dr. Pajares. “Results are also beginning to be published on the treatment of lung lesions using electromagnetic navigation ablation, demonstrating its safety and efficacy in this area.”
Nonsolid Imaging
The challenges in navigation include “improving the diagnosis of lung lesions that are nonsolid, known as ground glass opacities, and verifying it as an additional treatment option for lung nodules in patients who are not candidates for surgical resection,” said Dr. Pajares.
Tess Kramer, PhD, of Amsterdam University Medical Center, Amsterdam, the Netherlands, advocates for the combined use of different technologies to have a beneficial impact on patients’ clinical outcomes.
Robotic bronchoscopy has been implemented in the United States for several years, enhancing the precision of lung nodule diagnosis. However, “currently, there are no clear differences in the diagnostic performance of robotic bronchoscopy compared with navigation in general. Soon, there will be studies to assess in which type of nodules one technique may be more cost-effective.” No centers in Spain have this technology yet, “although some are already evaluating the acquisition of robotic bronchoscopy; it’s only a matter of time,” said Dr. Pajares.
Improvements in echobronchoscopy technology include high-quality image processors and smaller device calibers with greater angulation to diagnose lesions and hard-to-reach adenopathies. From an imaging perspective, AI, combined with the creation of risk calculators, could enable the prediction of lymph node malignancy.
Moreover, the use of small-caliber cryoprobes (1.1 mm) for obtaining samples of adenopathies (cryoEBUS) has enhanced diagnosis by enabling larger tissue samples. Current studies are being conducted to confirm the utility of cryoEBUS in pathologies requiring extensive molecular and immunohistochemical studies for diagnosing lymphoproliferative syndromes or neoplasms.
In a different context, liquid biopsy, a recent laboratory technology unrelated to bronchoscopy, allows the analysis of blood/pleural fluid samples that were extracted using the aforementioned technologies to locate tumor cells and differentiate between malignancy and benignity.
The Challenge of Pneumonitis
Samantha Aso, MD, a pulmonologist, member of the Catalan Society of Pulmonology, and specialist at the Lung Unit of Bellvitge University Hospital in Barcelona, Spain, discussed the challenge of managing pneumonitis in oncology patients.
Pneumonitis is an inflammation of the lungs that can be secondary to treatments, such as oncological therapy, which is the leading cause in 15%-50% of cases. Most oncological treatments can result in this process, including chemotherapy, chest radiotherapy, targeted therapies, conjugated monoclonal antibodies, and monotherapy.
To date, there is no known idiosyncratic cause of this process, except for autoimmune diseases. Pulmonary fibrosis is believed to be a risk factor. “Patients with interstitial lung disease and pulmonary fibrosis have been found to have a higher mortality risk due to pneumonitis. Consequently, cancer treatment cannot be administered to these patients,” said Dr. Aso.
Pulmonologists face the challenge of managing pneumonitis secondary to monotherapy, which currently is treated with cortisone. Patients respond well to this medication, but after corticosteroid withdrawal, reinflammation may occur. “In pneumonitis patients, oncological treatment (monotherapy) should be suspended while pulmonologists manage the pneumonitis with corticosteroids. However, we are uncertain about how rapidly or slowly to reduce the dosage. We cannot taper these doses as quickly as desired because reinflammation may occur, and to date, there are no alternative treatments apart from corticosteroids,” said Dr. Aso.
She noted that excellent survival results are achieved with monotherapy, but further research is required on the safety of antineoplastic drugs as a secondary endpoint. “Suspending oncological treatment due to pneumonitis means that patients are not receiving adequate cancer treatment, which has a significant psychological impact that also needs to be addressed,” Dr. Aso concluded.
Dr. Pajares and Dr. Aso declared no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
NIH to Begin Long COVID Trial Focused on Sleep, Exercise
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.














