User login
Data collection urged on patients treated with talk therapy
SCOTTSDALE, ARIZ. – Most psychiatrists are familiar with many of the basic tenets of psychoanalysis, but they probably aren’t relying on its therapeutic powers in practice as much as they are pharmacotherapy, according to an expert.
“There’s a great deal of discussion about whether psychoanalysis has an adequate evidence base. That’s a popular concept. It’s not so much about whether practitioners want to use it, but whether or not they can defend using it in their dialogs with insurance companies, government agencies, and other sources of support,” Robert Michels, MD, a Walsh McDermott University Professor of Medicine, and professor of psychiatry, at Cornell University, New York, said at the annual meeting of the American College of Psychiatrists. “My summary is that it has less adequate data supporting it [than pharmacologic interventions]. … There is, however, convincing evidence that all of these treatments, ranging from cognitive-behavioral therapy to [dialectical behavior therapy], to dynamic psychoanalysis, to mentalization-based treatment, do have an effect.”
“The SSRI probably did more to change psychotherapy than any other evolutionary change in the theory of psychotherapy,” Dr. Michels said. “In the beginning, [pharmacologic treatments] were not terribly useful to the patients of psychoanalysis, because they were too toxic or too psychologically disturbing to use in people without major psychiatric problems. Now, [there is] intense competition from biologic treatments.”
Whereas the goal of pharmacotherapy is cure, the point of psychoanalysis is to derive meaning from neurosis, and ultimately, from life. A sea change in thought about how psychoanalysis should be conducted was well underway before the advent of fluoxetine, however. With the death in 1939 of Sigmund Freud, MD, the founder of psychoanalysis, a wave of theories began to wash over the field. Most of those theories do not see memories as the enemy, or detachment from the patient as appropriate, according to Dr. Michels.
And while the form might vary across the different approaches, the function is the same. “The goal is no longer to recover the lost memory of the childhood trauma. More important is the journey in attempting to discover it,” Dr. Michels said.
Freud avoided seeing his patients as having personality disorders, while the next generations of psychotherapists largely embraced and conceptualized diagnoses, going beyond symptoms and instead focusing on the person’s predisposition to symptom formation. Psychotherapy today sees pathology not as something to be cured but as a reaction to problems tied to everyday living. It assumes that a more effective reaction to those problems can be learned.
Rather than shun what were once considered obstacles – resistance, enactment, transference, countertransference, and working through interpretations – the psychotherapist uses those to establish a relationship with the patient; emotions stirred by the therapist become important markers of patterns that have persisted in the person’s life that prevent forming smooth relationships, and vice versa. “In the new model, the therapist is an actively engaged participant whose core skill is to stay fully involved and interconnected while also being able to step back and say, ‘What is happening here?’ ” Dr. Michels said. “Transference and countertransference are essential tools to the therapeutic process. Hopefully, we change the patient’s attitude to his own mental life and toward others around him. We have a much more open, broader, wider way of dealing with patients, and we are much more humble about what we know before the treatment starts.”
In an interview, Dr. Michels said this has been especially helpful in treating people with borderline personality disorder, for whom the neutral face of a therapist can often be a trigger. “You end up being a better player through collaboration. If you carry the argument to its extreme, we’d say it doesn’t make any difference if you discover the flaw.”
Also in the interview, Dr. Michels said that, while many therapists are aware that psychotherapy has evolved and that it is effective, it is still not commonly taught in medical schools beyond a basic level because of the “finite nature of the amount of time to learn what students are required to learn in order to be a skilled psychotherapist, regardless of whether they have the raw talent for it.” Outcomes of pharmacologic therapies are easier to measure, and that is another reason psychotherapy is “troublesome,” he said. Most psychotherapists prefer not having to justify its use to third-party payers.
The tension between the therapist and insurers should not automatically be a deterrent to expanding access to talk therapy according to the meeting’s program chair, Scott T. Aaronson, MD, director of clinical research at the Sheppard Pratt Health System, Baltimore. “I don’t think that psychoanalysis has ever been a great friend of insurance. I think we need to educate insurance companies on what psychotherapy means. Instead, we just sort of allow them to make rules. It’s been a one-way street that needs to change,” Dr. Aaronson said in an interview.
He and Dr. Michels said data collection on patient outcomes would help. Collecting the hospitalization rates, morbidity and mortality, and medical care costs of people who have received psychotherapy may be worthwhile and would be fairly straightforward to do, according to Dr. Aaronson. “I would worry [about factoring in] those who have been getting psychiatric care off the grid skewing statistics, but if your patient population is of a moderate-income group, very few of those people could afford private care.”
Even if clinicians do not include newer psychoanalytic techniques in practice, Dr. Aaronson said, they “should keep up to date on the trends, and be aware that for many patients, modalities such as mentalization have been exquisitely helpful.”
Neither Dr. Michels nor Dr. Aaronson had any relevant disclosures.
SCOTTSDALE, ARIZ. – Most psychiatrists are familiar with many of the basic tenets of psychoanalysis, but they probably aren’t relying on its therapeutic powers in practice as much as they are pharmacotherapy, according to an expert.
“There’s a great deal of discussion about whether psychoanalysis has an adequate evidence base. That’s a popular concept. It’s not so much about whether practitioners want to use it, but whether or not they can defend using it in their dialogs with insurance companies, government agencies, and other sources of support,” Robert Michels, MD, a Walsh McDermott University Professor of Medicine, and professor of psychiatry, at Cornell University, New York, said at the annual meeting of the American College of Psychiatrists. “My summary is that it has less adequate data supporting it [than pharmacologic interventions]. … There is, however, convincing evidence that all of these treatments, ranging from cognitive-behavioral therapy to [dialectical behavior therapy], to dynamic psychoanalysis, to mentalization-based treatment, do have an effect.”
“The SSRI probably did more to change psychotherapy than any other evolutionary change in the theory of psychotherapy,” Dr. Michels said. “In the beginning, [pharmacologic treatments] were not terribly useful to the patients of psychoanalysis, because they were too toxic or too psychologically disturbing to use in people without major psychiatric problems. Now, [there is] intense competition from biologic treatments.”
Whereas the goal of pharmacotherapy is cure, the point of psychoanalysis is to derive meaning from neurosis, and ultimately, from life. A sea change in thought about how psychoanalysis should be conducted was well underway before the advent of fluoxetine, however. With the death in 1939 of Sigmund Freud, MD, the founder of psychoanalysis, a wave of theories began to wash over the field. Most of those theories do not see memories as the enemy, or detachment from the patient as appropriate, according to Dr. Michels.
And while the form might vary across the different approaches, the function is the same. “The goal is no longer to recover the lost memory of the childhood trauma. More important is the journey in attempting to discover it,” Dr. Michels said.
Freud avoided seeing his patients as having personality disorders, while the next generations of psychotherapists largely embraced and conceptualized diagnoses, going beyond symptoms and instead focusing on the person’s predisposition to symptom formation. Psychotherapy today sees pathology not as something to be cured but as a reaction to problems tied to everyday living. It assumes that a more effective reaction to those problems can be learned.
Rather than shun what were once considered obstacles – resistance, enactment, transference, countertransference, and working through interpretations – the psychotherapist uses those to establish a relationship with the patient; emotions stirred by the therapist become important markers of patterns that have persisted in the person’s life that prevent forming smooth relationships, and vice versa. “In the new model, the therapist is an actively engaged participant whose core skill is to stay fully involved and interconnected while also being able to step back and say, ‘What is happening here?’ ” Dr. Michels said. “Transference and countertransference are essential tools to the therapeutic process. Hopefully, we change the patient’s attitude to his own mental life and toward others around him. We have a much more open, broader, wider way of dealing with patients, and we are much more humble about what we know before the treatment starts.”
In an interview, Dr. Michels said this has been especially helpful in treating people with borderline personality disorder, for whom the neutral face of a therapist can often be a trigger. “You end up being a better player through collaboration. If you carry the argument to its extreme, we’d say it doesn’t make any difference if you discover the flaw.”
Also in the interview, Dr. Michels said that, while many therapists are aware that psychotherapy has evolved and that it is effective, it is still not commonly taught in medical schools beyond a basic level because of the “finite nature of the amount of time to learn what students are required to learn in order to be a skilled psychotherapist, regardless of whether they have the raw talent for it.” Outcomes of pharmacologic therapies are easier to measure, and that is another reason psychotherapy is “troublesome,” he said. Most psychotherapists prefer not having to justify its use to third-party payers.
The tension between the therapist and insurers should not automatically be a deterrent to expanding access to talk therapy according to the meeting’s program chair, Scott T. Aaronson, MD, director of clinical research at the Sheppard Pratt Health System, Baltimore. “I don’t think that psychoanalysis has ever been a great friend of insurance. I think we need to educate insurance companies on what psychotherapy means. Instead, we just sort of allow them to make rules. It’s been a one-way street that needs to change,” Dr. Aaronson said in an interview.
He and Dr. Michels said data collection on patient outcomes would help. Collecting the hospitalization rates, morbidity and mortality, and medical care costs of people who have received psychotherapy may be worthwhile and would be fairly straightforward to do, according to Dr. Aaronson. “I would worry [about factoring in] those who have been getting psychiatric care off the grid skewing statistics, but if your patient population is of a moderate-income group, very few of those people could afford private care.”
Even if clinicians do not include newer psychoanalytic techniques in practice, Dr. Aaronson said, they “should keep up to date on the trends, and be aware that for many patients, modalities such as mentalization have been exquisitely helpful.”
Neither Dr. Michels nor Dr. Aaronson had any relevant disclosures.
SCOTTSDALE, ARIZ. – Most psychiatrists are familiar with many of the basic tenets of psychoanalysis, but they probably aren’t relying on its therapeutic powers in practice as much as they are pharmacotherapy, according to an expert.
“There’s a great deal of discussion about whether psychoanalysis has an adequate evidence base. That’s a popular concept. It’s not so much about whether practitioners want to use it, but whether or not they can defend using it in their dialogs with insurance companies, government agencies, and other sources of support,” Robert Michels, MD, a Walsh McDermott University Professor of Medicine, and professor of psychiatry, at Cornell University, New York, said at the annual meeting of the American College of Psychiatrists. “My summary is that it has less adequate data supporting it [than pharmacologic interventions]. … There is, however, convincing evidence that all of these treatments, ranging from cognitive-behavioral therapy to [dialectical behavior therapy], to dynamic psychoanalysis, to mentalization-based treatment, do have an effect.”
“The SSRI probably did more to change psychotherapy than any other evolutionary change in the theory of psychotherapy,” Dr. Michels said. “In the beginning, [pharmacologic treatments] were not terribly useful to the patients of psychoanalysis, because they were too toxic or too psychologically disturbing to use in people without major psychiatric problems. Now, [there is] intense competition from biologic treatments.”
Whereas the goal of pharmacotherapy is cure, the point of psychoanalysis is to derive meaning from neurosis, and ultimately, from life. A sea change in thought about how psychoanalysis should be conducted was well underway before the advent of fluoxetine, however. With the death in 1939 of Sigmund Freud, MD, the founder of psychoanalysis, a wave of theories began to wash over the field. Most of those theories do not see memories as the enemy, or detachment from the patient as appropriate, according to Dr. Michels.
And while the form might vary across the different approaches, the function is the same. “The goal is no longer to recover the lost memory of the childhood trauma. More important is the journey in attempting to discover it,” Dr. Michels said.
Freud avoided seeing his patients as having personality disorders, while the next generations of psychotherapists largely embraced and conceptualized diagnoses, going beyond symptoms and instead focusing on the person’s predisposition to symptom formation. Psychotherapy today sees pathology not as something to be cured but as a reaction to problems tied to everyday living. It assumes that a more effective reaction to those problems can be learned.
Rather than shun what were once considered obstacles – resistance, enactment, transference, countertransference, and working through interpretations – the psychotherapist uses those to establish a relationship with the patient; emotions stirred by the therapist become important markers of patterns that have persisted in the person’s life that prevent forming smooth relationships, and vice versa. “In the new model, the therapist is an actively engaged participant whose core skill is to stay fully involved and interconnected while also being able to step back and say, ‘What is happening here?’ ” Dr. Michels said. “Transference and countertransference are essential tools to the therapeutic process. Hopefully, we change the patient’s attitude to his own mental life and toward others around him. We have a much more open, broader, wider way of dealing with patients, and we are much more humble about what we know before the treatment starts.”
In an interview, Dr. Michels said this has been especially helpful in treating people with borderline personality disorder, for whom the neutral face of a therapist can often be a trigger. “You end up being a better player through collaboration. If you carry the argument to its extreme, we’d say it doesn’t make any difference if you discover the flaw.”
Also in the interview, Dr. Michels said that, while many therapists are aware that psychotherapy has evolved and that it is effective, it is still not commonly taught in medical schools beyond a basic level because of the “finite nature of the amount of time to learn what students are required to learn in order to be a skilled psychotherapist, regardless of whether they have the raw talent for it.” Outcomes of pharmacologic therapies are easier to measure, and that is another reason psychotherapy is “troublesome,” he said. Most psychotherapists prefer not having to justify its use to third-party payers.
The tension between the therapist and insurers should not automatically be a deterrent to expanding access to talk therapy according to the meeting’s program chair, Scott T. Aaronson, MD, director of clinical research at the Sheppard Pratt Health System, Baltimore. “I don’t think that psychoanalysis has ever been a great friend of insurance. I think we need to educate insurance companies on what psychotherapy means. Instead, we just sort of allow them to make rules. It’s been a one-way street that needs to change,” Dr. Aaronson said in an interview.
He and Dr. Michels said data collection on patient outcomes would help. Collecting the hospitalization rates, morbidity and mortality, and medical care costs of people who have received psychotherapy may be worthwhile and would be fairly straightforward to do, according to Dr. Aaronson. “I would worry [about factoring in] those who have been getting psychiatric care off the grid skewing statistics, but if your patient population is of a moderate-income group, very few of those people could afford private care.”
Even if clinicians do not include newer psychoanalytic techniques in practice, Dr. Aaronson said, they “should keep up to date on the trends, and be aware that for many patients, modalities such as mentalization have been exquisitely helpful.”
Neither Dr. Michels nor Dr. Aaronson had any relevant disclosures.
EXPERT ANALYSIS AT THE AMERICAN COLLEGE OF PSYCHIATRISTS ANNUAL MEETING
Promising ibrutinib data prompt frontline cGVHD therapy study
ORLANDO – Ibrutinib was associated with clinically meaningful and durable responses in patients with chronic graft-versus-host disease that did not respond to frontline systemic therapy, based on the final results of a phase II study.
Preliminary findings from that study led in 2016 to a Food and Drug Administration Breakthrough Therapy Designation for ibrutinib for chronic graft-versus-host disease (cGVHD) after the failure of one or more lines of systemic therapy, and the responses seen in this pretreated, high-risk population support the study of ibrutinib for frontline treatment of cGVHD, said David Miklos, MD, of Stanford (Calif.) University.
Of 20 patients with multiple organ involvement, 25 (80%) had responses in at least two organs, and of 9 patients with three or more involved organs, 5 (56%) had responses in at least three organs, he reported at the combined annual meetings of the Center for International Blood & Marrow Transplant Research and the American Society for Blood and Marrow Transplantation..
“We think that the responses across different organ involvement as well as multiple organ responses speaks to the underlying impact of ibrutinib on the pathogenic pathway and not to masking symptoms or indirect effect,” he said.
The median steroid dose among responders decreased from 0.29 mg/kg daily at baseline to 0.19 mg/kg daily and 0.12 mg/kg daily at weeks 25 and 49, respectively. Overall, 62% of all patients reached steroid doses less than 0.15 mg/kg daily, and five responders discontinued steroid treatment.
“Patients also had clinically meaningful improvement as assessed by the Lee symptoms scale,” he said, noting that scores improved in 61% of responders and 11% of nonresponders.
Study participants had a median age of 56 years and a median of 7.6 months from allogeneic transplant to diagnosis of cGVHD. All had been treated with up to three prior cGVHD regimens (median, two) and had either a rash that exceeded 25% of their body surface area or a National Institutes of Health consensus mouth score greater than 4. They were treated with ibrutinib at a dose of 420 mg/day until cGVHD progression or unacceptable toxicity. The cGVHD response – the primary endpoint of the study – was measured using 2005 NIH response criteria.
Adverse events occurring in at least 20% of patients included fatigue, diarrhea, muscle spasms, nausea, and bruising. Grade 3 or higher adverse events occurring in at least 10% of patients included pneumonia, fatigue, and diarrhea.
Serious adverse events occurred in 52% of patients. Grade 3 or higher serious adverse events occurred in 40% of patients and included pneumonia, septic shock, and pyrexia. Two fatal events were reported and included one case of multilobular pneumonia and one case of bronchopulmonary aspergillosis.
Twelve patients (29%) remained on ibrutinib at 14 months; Of those who discontinued therapy, 5 discontinued because of progressive cGVHD, and 14 because of adverse events.
Patients who have cGVHD and don’t respond to frontline therapy have previously had no effective options. Ibrutinib showed promise in preclinical models; it reduced the severity of cGVHD through inhibition of Bruton’s tyrosine kinase and interleukin-2–inducible T-cell kinase, Dr. Miklos explained, noting that both B and T cells play a role in the pathophysiology of cGVHD.
The findings from this phase II trial demonstrate that ibrutinib does indeed lead to durable improvement in this patient population, and its safety profile is consistent with that previously reported for B-cell malignancies treated with ibrutinib and for cGVHD patients treated with concomitant steroids, he said.
“We think the efficacy of ibrutinib in this population supports further study in frontline treatment of cGVHD in a randomized, double-blinded study,” he concluded.
A phase III study – the INTEGRATE clinical trial – is now open. The international study will compare ibrutinib and prednisone with placebo and prednisone as a frontline therapy for moderate and severe cGVHD with a primary endpoint of response rate at 24 weeks.
The study was sponsored by Pharmacyclics in collaboration with Janssen Research & Development. Dr. Miklos reported various financial relationships with Pharmacyclics (the maker of ibrutinib [Imbruvica]), Velos, Kite Pharma, Sanofi Oncology, Adaptive Biotechnologies, and Genentech.
sworcester@frontlinemedcom.com
ORLANDO – Ibrutinib was associated with clinically meaningful and durable responses in patients with chronic graft-versus-host disease that did not respond to frontline systemic therapy, based on the final results of a phase II study.
Preliminary findings from that study led in 2016 to a Food and Drug Administration Breakthrough Therapy Designation for ibrutinib for chronic graft-versus-host disease (cGVHD) after the failure of one or more lines of systemic therapy, and the responses seen in this pretreated, high-risk population support the study of ibrutinib for frontline treatment of cGVHD, said David Miklos, MD, of Stanford (Calif.) University.
Of 20 patients with multiple organ involvement, 25 (80%) had responses in at least two organs, and of 9 patients with three or more involved organs, 5 (56%) had responses in at least three organs, he reported at the combined annual meetings of the Center for International Blood & Marrow Transplant Research and the American Society for Blood and Marrow Transplantation..
“We think that the responses across different organ involvement as well as multiple organ responses speaks to the underlying impact of ibrutinib on the pathogenic pathway and not to masking symptoms or indirect effect,” he said.
The median steroid dose among responders decreased from 0.29 mg/kg daily at baseline to 0.19 mg/kg daily and 0.12 mg/kg daily at weeks 25 and 49, respectively. Overall, 62% of all patients reached steroid doses less than 0.15 mg/kg daily, and five responders discontinued steroid treatment.
“Patients also had clinically meaningful improvement as assessed by the Lee symptoms scale,” he said, noting that scores improved in 61% of responders and 11% of nonresponders.
Study participants had a median age of 56 years and a median of 7.6 months from allogeneic transplant to diagnosis of cGVHD. All had been treated with up to three prior cGVHD regimens (median, two) and had either a rash that exceeded 25% of their body surface area or a National Institutes of Health consensus mouth score greater than 4. They were treated with ibrutinib at a dose of 420 mg/day until cGVHD progression or unacceptable toxicity. The cGVHD response – the primary endpoint of the study – was measured using 2005 NIH response criteria.
Adverse events occurring in at least 20% of patients included fatigue, diarrhea, muscle spasms, nausea, and bruising. Grade 3 or higher adverse events occurring in at least 10% of patients included pneumonia, fatigue, and diarrhea.
Serious adverse events occurred in 52% of patients. Grade 3 or higher serious adverse events occurred in 40% of patients and included pneumonia, septic shock, and pyrexia. Two fatal events were reported and included one case of multilobular pneumonia and one case of bronchopulmonary aspergillosis.
Twelve patients (29%) remained on ibrutinib at 14 months; Of those who discontinued therapy, 5 discontinued because of progressive cGVHD, and 14 because of adverse events.
Patients who have cGVHD and don’t respond to frontline therapy have previously had no effective options. Ibrutinib showed promise in preclinical models; it reduced the severity of cGVHD through inhibition of Bruton’s tyrosine kinase and interleukin-2–inducible T-cell kinase, Dr. Miklos explained, noting that both B and T cells play a role in the pathophysiology of cGVHD.
The findings from this phase II trial demonstrate that ibrutinib does indeed lead to durable improvement in this patient population, and its safety profile is consistent with that previously reported for B-cell malignancies treated with ibrutinib and for cGVHD patients treated with concomitant steroids, he said.
“We think the efficacy of ibrutinib in this population supports further study in frontline treatment of cGVHD in a randomized, double-blinded study,” he concluded.
A phase III study – the INTEGRATE clinical trial – is now open. The international study will compare ibrutinib and prednisone with placebo and prednisone as a frontline therapy for moderate and severe cGVHD with a primary endpoint of response rate at 24 weeks.
The study was sponsored by Pharmacyclics in collaboration with Janssen Research & Development. Dr. Miklos reported various financial relationships with Pharmacyclics (the maker of ibrutinib [Imbruvica]), Velos, Kite Pharma, Sanofi Oncology, Adaptive Biotechnologies, and Genentech.
sworcester@frontlinemedcom.com
ORLANDO – Ibrutinib was associated with clinically meaningful and durable responses in patients with chronic graft-versus-host disease that did not respond to frontline systemic therapy, based on the final results of a phase II study.
Preliminary findings from that study led in 2016 to a Food and Drug Administration Breakthrough Therapy Designation for ibrutinib for chronic graft-versus-host disease (cGVHD) after the failure of one or more lines of systemic therapy, and the responses seen in this pretreated, high-risk population support the study of ibrutinib for frontline treatment of cGVHD, said David Miklos, MD, of Stanford (Calif.) University.
Of 20 patients with multiple organ involvement, 25 (80%) had responses in at least two organs, and of 9 patients with three or more involved organs, 5 (56%) had responses in at least three organs, he reported at the combined annual meetings of the Center for International Blood & Marrow Transplant Research and the American Society for Blood and Marrow Transplantation..
“We think that the responses across different organ involvement as well as multiple organ responses speaks to the underlying impact of ibrutinib on the pathogenic pathway and not to masking symptoms or indirect effect,” he said.
The median steroid dose among responders decreased from 0.29 mg/kg daily at baseline to 0.19 mg/kg daily and 0.12 mg/kg daily at weeks 25 and 49, respectively. Overall, 62% of all patients reached steroid doses less than 0.15 mg/kg daily, and five responders discontinued steroid treatment.
“Patients also had clinically meaningful improvement as assessed by the Lee symptoms scale,” he said, noting that scores improved in 61% of responders and 11% of nonresponders.
Study participants had a median age of 56 years and a median of 7.6 months from allogeneic transplant to diagnosis of cGVHD. All had been treated with up to three prior cGVHD regimens (median, two) and had either a rash that exceeded 25% of their body surface area or a National Institutes of Health consensus mouth score greater than 4. They were treated with ibrutinib at a dose of 420 mg/day until cGVHD progression or unacceptable toxicity. The cGVHD response – the primary endpoint of the study – was measured using 2005 NIH response criteria.
Adverse events occurring in at least 20% of patients included fatigue, diarrhea, muscle spasms, nausea, and bruising. Grade 3 or higher adverse events occurring in at least 10% of patients included pneumonia, fatigue, and diarrhea.
Serious adverse events occurred in 52% of patients. Grade 3 or higher serious adverse events occurred in 40% of patients and included pneumonia, septic shock, and pyrexia. Two fatal events were reported and included one case of multilobular pneumonia and one case of bronchopulmonary aspergillosis.
Twelve patients (29%) remained on ibrutinib at 14 months; Of those who discontinued therapy, 5 discontinued because of progressive cGVHD, and 14 because of adverse events.
Patients who have cGVHD and don’t respond to frontline therapy have previously had no effective options. Ibrutinib showed promise in preclinical models; it reduced the severity of cGVHD through inhibition of Bruton’s tyrosine kinase and interleukin-2–inducible T-cell kinase, Dr. Miklos explained, noting that both B and T cells play a role in the pathophysiology of cGVHD.
The findings from this phase II trial demonstrate that ibrutinib does indeed lead to durable improvement in this patient population, and its safety profile is consistent with that previously reported for B-cell malignancies treated with ibrutinib and for cGVHD patients treated with concomitant steroids, he said.
“We think the efficacy of ibrutinib in this population supports further study in frontline treatment of cGVHD in a randomized, double-blinded study,” he concluded.
A phase III study – the INTEGRATE clinical trial – is now open. The international study will compare ibrutinib and prednisone with placebo and prednisone as a frontline therapy for moderate and severe cGVHD with a primary endpoint of response rate at 24 weeks.
The study was sponsored by Pharmacyclics in collaboration with Janssen Research & Development. Dr. Miklos reported various financial relationships with Pharmacyclics (the maker of ibrutinib [Imbruvica]), Velos, Kite Pharma, Sanofi Oncology, Adaptive Biotechnologies, and Genentech.
sworcester@frontlinemedcom.com
AT THE 2017 BMT TANDEM MEETINGS
Key clinical point:
Major finding: At a median follow-up of 14 months, the overall response rate among 42 patients treated with ibrutinib for cGVHD was 67%, with a third of responders achieving a complete response.
Data source: A phase II study of 42 patients.
Disclosures: The study was sponsored by Pharmacyclics in collaboration with Janssen Research & Development. Dr. Miklos reported various financial relationships with Pharmacyclics (the maker of ibrutinib [Imbruvica]), Velos, Kite Pharma, Sanofi Oncology, Adaptive Biotechnologies, and Genentech.
In beta thalassemia major, liver stiffness declines with deferasirox
Patients with beta thalassemia major who were chelated only with oral deferasirox experienced significant improvements in liver stiffness, both from baseline and compared with patients who interrupted deferasirox therapy because of pregnancy, according to a small prospective 5-year study.
Transient elastography showed that continuous therapy with deferasirox (median dose, 35 mg per kg) yielded an 0.85 kPa average improvement in liver stiffness compared with baseline (P = .02), reported Nikolaos Sousos of Aristotle University of Thessaloniki, Greece, and his associates (Br J Haematol. 2017 Jan 20. doi: 10.1111/bjh.14509).
In contrast, interrupting therapy for a median of 16 months because of successful pregnancy led to an average increase in liver stiffness of 1.84 kPa – a significant difference between groups (P = .005) even after the researchers controlled for gender, age, ferritin levels, and T2-weighted magnetic resonance imaging (MRI) measurements of iron deposition in the liver and heart.
Patients with beta thalassemia major often develop liver fibrosis because of excessive intestinal iron absorption, iron overload from transfusions, or hepatitis C virus infection, the investigators noted. The median age of the patients in this study was 32 years (range, 20-47 years), they were HCV negative, and they received regular transfusions to maintain hemoglobin levels above 95 g/L. The seven female participants who temporarily stopped deferasirox because of pregnancy all restarted therapy at least 8 months before their follow-up transient elastography liver stiffness measurement, the investigators reported.
T2-weighted MRI measurements of liver iron concentration also had improved at follow-up, reflecting “better control of iron overload,” although the difference from baseline was not statistically significant, the investigators noted. This result reinforces previous findings (Gastroenterology. 2011;141[4]:1202-11) that long-term deferasirox therapy can significantly improve liver fibrosis in patients with beta thalassemia, regardless of liver iron concentration, the researchers added. Together, those findings suggest that “improvement in liver fibrosis, rather than liver iron concentration should be the primary effect of chelation therapy,” they suggested.
The Research Committee of Aristotle University of Thessaloniki, Novartis Hellas (Novartis AG, Basel, Switzerland), and the Greek Thalassaemia Association funded the study. The authors declared having no competing interests.
Patients with beta thalassemia major who were chelated only with oral deferasirox experienced significant improvements in liver stiffness, both from baseline and compared with patients who interrupted deferasirox therapy because of pregnancy, according to a small prospective 5-year study.
Transient elastography showed that continuous therapy with deferasirox (median dose, 35 mg per kg) yielded an 0.85 kPa average improvement in liver stiffness compared with baseline (P = .02), reported Nikolaos Sousos of Aristotle University of Thessaloniki, Greece, and his associates (Br J Haematol. 2017 Jan 20. doi: 10.1111/bjh.14509).
In contrast, interrupting therapy for a median of 16 months because of successful pregnancy led to an average increase in liver stiffness of 1.84 kPa – a significant difference between groups (P = .005) even after the researchers controlled for gender, age, ferritin levels, and T2-weighted magnetic resonance imaging (MRI) measurements of iron deposition in the liver and heart.
Patients with beta thalassemia major often develop liver fibrosis because of excessive intestinal iron absorption, iron overload from transfusions, or hepatitis C virus infection, the investigators noted. The median age of the patients in this study was 32 years (range, 20-47 years), they were HCV negative, and they received regular transfusions to maintain hemoglobin levels above 95 g/L. The seven female participants who temporarily stopped deferasirox because of pregnancy all restarted therapy at least 8 months before their follow-up transient elastography liver stiffness measurement, the investigators reported.
T2-weighted MRI measurements of liver iron concentration also had improved at follow-up, reflecting “better control of iron overload,” although the difference from baseline was not statistically significant, the investigators noted. This result reinforces previous findings (Gastroenterology. 2011;141[4]:1202-11) that long-term deferasirox therapy can significantly improve liver fibrosis in patients with beta thalassemia, regardless of liver iron concentration, the researchers added. Together, those findings suggest that “improvement in liver fibrosis, rather than liver iron concentration should be the primary effect of chelation therapy,” they suggested.
The Research Committee of Aristotle University of Thessaloniki, Novartis Hellas (Novartis AG, Basel, Switzerland), and the Greek Thalassaemia Association funded the study. The authors declared having no competing interests.
Patients with beta thalassemia major who were chelated only with oral deferasirox experienced significant improvements in liver stiffness, both from baseline and compared with patients who interrupted deferasirox therapy because of pregnancy, according to a small prospective 5-year study.
Transient elastography showed that continuous therapy with deferasirox (median dose, 35 mg per kg) yielded an 0.85 kPa average improvement in liver stiffness compared with baseline (P = .02), reported Nikolaos Sousos of Aristotle University of Thessaloniki, Greece, and his associates (Br J Haematol. 2017 Jan 20. doi: 10.1111/bjh.14509).
In contrast, interrupting therapy for a median of 16 months because of successful pregnancy led to an average increase in liver stiffness of 1.84 kPa – a significant difference between groups (P = .005) even after the researchers controlled for gender, age, ferritin levels, and T2-weighted magnetic resonance imaging (MRI) measurements of iron deposition in the liver and heart.
Patients with beta thalassemia major often develop liver fibrosis because of excessive intestinal iron absorption, iron overload from transfusions, or hepatitis C virus infection, the investigators noted. The median age of the patients in this study was 32 years (range, 20-47 years), they were HCV negative, and they received regular transfusions to maintain hemoglobin levels above 95 g/L. The seven female participants who temporarily stopped deferasirox because of pregnancy all restarted therapy at least 8 months before their follow-up transient elastography liver stiffness measurement, the investigators reported.
T2-weighted MRI measurements of liver iron concentration also had improved at follow-up, reflecting “better control of iron overload,” although the difference from baseline was not statistically significant, the investigators noted. This result reinforces previous findings (Gastroenterology. 2011;141[4]:1202-11) that long-term deferasirox therapy can significantly improve liver fibrosis in patients with beta thalassemia, regardless of liver iron concentration, the researchers added. Together, those findings suggest that “improvement in liver fibrosis, rather than liver iron concentration should be the primary effect of chelation therapy,” they suggested.
The Research Committee of Aristotle University of Thessaloniki, Novartis Hellas (Novartis AG, Basel, Switzerland), and the Greek Thalassaemia Association funded the study. The authors declared having no competing interests.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Key clinical point. For patients with beta thalassemia major, long-term chelation with deferasirox led to significant improvements in liver stiffness.
Major finding: Five years of continuous therapy yielded an 0.85 kPa average improvement in liver stiffness compared with baseline (P = .02).
Data source: A 5-year, single-center prospective study of 22 patients.
Disclosures: The Research Committee of Aristotle University of Thessaloniki, Novartis Hellas (Novartis AG, Basel, Switzerland), and the Greek Thalassaemia Association funded the study. The authors declared having no competing interests.
Chemo gives no boost to ADT for patients with localized prostate cancer
Mitoxantrone plus prednisone (MP) added to androgen deprivation therapy (ADT) does not improve outcomes in patients with clinically localized prostate cancer, according to the results of a large long-term multicenter clinical trial.
At a follow-up time of almost 11 years, outcomes were nearly the same whether patients had received chemotherapy and ADT or just ADT.
Overall survival was 87% in the cohort that received ADT only and 86% in the study arm that received ADT plus MP (HR 1.05 (CI 0.78, 1.42), P = .74). Recurrence-free survival was 84% in both groups (HR 0.98 (CI 0.78, 1.23), P = .83).
Disease free survival was 72% in both arms, and there was no statistical difference. Similarly, death without recurrence were also similar in both arms.
“Survival was greater than anticipated in both arms,” lead author L. Michael Glode, MD, of the University of Colorado, Denver, said in a press briefing held at the 2017 genitourinary cancers symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology. “There is no evidence that MP improves prostate cancer specific survival when added to 2 years of adjuvant ADT.”
There were 85 deaths in the ADT arm compared to 91 in the chemotherapy arm. Prostate cancer accounted for 18% of deaths among the patients receiving ADT only and 22% in the chemotherapy arm, but the incidence of other cancers was twice as common in the chemotherapy arm as in the ADT only arm.
“The predominant cancers were GI and lung,” said Dr. Glode, “And noncancer deaths were variable.”
Of note, MP increased the incidence of leukemia (one case in the ADT group vs. five in the chemotherapy group).
“This trial demonstrates the feasibility of doing adjuvant trials in prostate cancer post radical prostatectomy,” said Dr. Glode. “Survival was greater than anticipated in both arms.”
The assumptions of this trial were that 2 years of adjuvant ADT would improve overall survival and progression-free survival although definitive data were unavailable.
The rationale for doing a study using adjuvant therapy was based on published literature, in which findings had showed that while short-term neoadjuvant ADT prior to prostatectomy reduced positive margins, it had no effect on disease-free survival. In addition, research had shown that longer-term ADT improved outcomes for patients undergoing curative radiation therapy.
For chemotherapy, Dr. Glode pointed to data showing that adjuvant chemotherapy improved progression-free survival in patients who had undergone both prostatectomy and radiation therapy.
“We hypothesized that the addition of modestly active chemotherapy earlier in disease might improve overall survival and progression-free survival,” he said.
The primary objective was overall survival and the secondary endpoint was disease-free survival.
The S9921 trial enrolled 983 patients from October 1999 to January 2007 with clinically localized prostate cancer, before enrollment ceased because of the increased incidence of leukemia in the ADT plus MP arm.
Of this group, 22 patients were ineligible, and the remaining patients were assigned to goserelin acetate 10.8 mg plus bicalutamide 50 mg (n = 481) or the same ADT plus MP (n = 480).
The patients were stratified by stage (≤pT2, ≥pT3, N0 or N+), Gleason score, and intent to receive adjuvant radiation, and the presurgical PSA was 7.6 ng/mL. Radiation therapy was allowed in both arms at physician discretion, and 26% intended to receive radiation therapy.
In the ADT only arm, 402 completed the treatment, and 390 completed treatment in the ADT plus chemotherapy arm.
Grade 3 or higher adverse events were more common in the chemotherapy group (56%/30%, P less than .0001). “As for toxicities, the main difference was the presence of leukopenia in the chemotherapy arm,” said Dr. Glode.
The study was funded by Southwest Oncology Group’s Urologic Cancer Outreach Program, Eastern Cooperative Oncology Group, Cancer and Leukemia Group B, Clinical Trials Support Unit, and National Cancer Institute. None of the authors had disclosures.
Mitoxantrone plus prednisone (MP) added to androgen deprivation therapy (ADT) does not improve outcomes in patients with clinically localized prostate cancer, according to the results of a large long-term multicenter clinical trial.
At a follow-up time of almost 11 years, outcomes were nearly the same whether patients had received chemotherapy and ADT or just ADT.
Overall survival was 87% in the cohort that received ADT only and 86% in the study arm that received ADT plus MP (HR 1.05 (CI 0.78, 1.42), P = .74). Recurrence-free survival was 84% in both groups (HR 0.98 (CI 0.78, 1.23), P = .83).
Disease free survival was 72% in both arms, and there was no statistical difference. Similarly, death without recurrence were also similar in both arms.
“Survival was greater than anticipated in both arms,” lead author L. Michael Glode, MD, of the University of Colorado, Denver, said in a press briefing held at the 2017 genitourinary cancers symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology. “There is no evidence that MP improves prostate cancer specific survival when added to 2 years of adjuvant ADT.”
There were 85 deaths in the ADT arm compared to 91 in the chemotherapy arm. Prostate cancer accounted for 18% of deaths among the patients receiving ADT only and 22% in the chemotherapy arm, but the incidence of other cancers was twice as common in the chemotherapy arm as in the ADT only arm.
“The predominant cancers were GI and lung,” said Dr. Glode, “And noncancer deaths were variable.”
Of note, MP increased the incidence of leukemia (one case in the ADT group vs. five in the chemotherapy group).
“This trial demonstrates the feasibility of doing adjuvant trials in prostate cancer post radical prostatectomy,” said Dr. Glode. “Survival was greater than anticipated in both arms.”
The assumptions of this trial were that 2 years of adjuvant ADT would improve overall survival and progression-free survival although definitive data were unavailable.
The rationale for doing a study using adjuvant therapy was based on published literature, in which findings had showed that while short-term neoadjuvant ADT prior to prostatectomy reduced positive margins, it had no effect on disease-free survival. In addition, research had shown that longer-term ADT improved outcomes for patients undergoing curative radiation therapy.
For chemotherapy, Dr. Glode pointed to data showing that adjuvant chemotherapy improved progression-free survival in patients who had undergone both prostatectomy and radiation therapy.
“We hypothesized that the addition of modestly active chemotherapy earlier in disease might improve overall survival and progression-free survival,” he said.
The primary objective was overall survival and the secondary endpoint was disease-free survival.
The S9921 trial enrolled 983 patients from October 1999 to January 2007 with clinically localized prostate cancer, before enrollment ceased because of the increased incidence of leukemia in the ADT plus MP arm.
Of this group, 22 patients were ineligible, and the remaining patients were assigned to goserelin acetate 10.8 mg plus bicalutamide 50 mg (n = 481) or the same ADT plus MP (n = 480).
The patients were stratified by stage (≤pT2, ≥pT3, N0 or N+), Gleason score, and intent to receive adjuvant radiation, and the presurgical PSA was 7.6 ng/mL. Radiation therapy was allowed in both arms at physician discretion, and 26% intended to receive radiation therapy.
In the ADT only arm, 402 completed the treatment, and 390 completed treatment in the ADT plus chemotherapy arm.
Grade 3 or higher adverse events were more common in the chemotherapy group (56%/30%, P less than .0001). “As for toxicities, the main difference was the presence of leukopenia in the chemotherapy arm,” said Dr. Glode.
The study was funded by Southwest Oncology Group’s Urologic Cancer Outreach Program, Eastern Cooperative Oncology Group, Cancer and Leukemia Group B, Clinical Trials Support Unit, and National Cancer Institute. None of the authors had disclosures.
Mitoxantrone plus prednisone (MP) added to androgen deprivation therapy (ADT) does not improve outcomes in patients with clinically localized prostate cancer, according to the results of a large long-term multicenter clinical trial.
At a follow-up time of almost 11 years, outcomes were nearly the same whether patients had received chemotherapy and ADT or just ADT.
Overall survival was 87% in the cohort that received ADT only and 86% in the study arm that received ADT plus MP (HR 1.05 (CI 0.78, 1.42), P = .74). Recurrence-free survival was 84% in both groups (HR 0.98 (CI 0.78, 1.23), P = .83).
Disease free survival was 72% in both arms, and there was no statistical difference. Similarly, death without recurrence were also similar in both arms.
“Survival was greater than anticipated in both arms,” lead author L. Michael Glode, MD, of the University of Colorado, Denver, said in a press briefing held at the 2017 genitourinary cancers symposium sponsored by the American Society of Clinical Oncology, ASTRO, and the Society of Urologic Oncology. “There is no evidence that MP improves prostate cancer specific survival when added to 2 years of adjuvant ADT.”
There were 85 deaths in the ADT arm compared to 91 in the chemotherapy arm. Prostate cancer accounted for 18% of deaths among the patients receiving ADT only and 22% in the chemotherapy arm, but the incidence of other cancers was twice as common in the chemotherapy arm as in the ADT only arm.
“The predominant cancers were GI and lung,” said Dr. Glode, “And noncancer deaths were variable.”
Of note, MP increased the incidence of leukemia (one case in the ADT group vs. five in the chemotherapy group).
“This trial demonstrates the feasibility of doing adjuvant trials in prostate cancer post radical prostatectomy,” said Dr. Glode. “Survival was greater than anticipated in both arms.”
The assumptions of this trial were that 2 years of adjuvant ADT would improve overall survival and progression-free survival although definitive data were unavailable.
The rationale for doing a study using adjuvant therapy was based on published literature, in which findings had showed that while short-term neoadjuvant ADT prior to prostatectomy reduced positive margins, it had no effect on disease-free survival. In addition, research had shown that longer-term ADT improved outcomes for patients undergoing curative radiation therapy.
For chemotherapy, Dr. Glode pointed to data showing that adjuvant chemotherapy improved progression-free survival in patients who had undergone both prostatectomy and radiation therapy.
“We hypothesized that the addition of modestly active chemotherapy earlier in disease might improve overall survival and progression-free survival,” he said.
The primary objective was overall survival and the secondary endpoint was disease-free survival.
The S9921 trial enrolled 983 patients from October 1999 to January 2007 with clinically localized prostate cancer, before enrollment ceased because of the increased incidence of leukemia in the ADT plus MP arm.
Of this group, 22 patients were ineligible, and the remaining patients were assigned to goserelin acetate 10.8 mg plus bicalutamide 50 mg (n = 481) or the same ADT plus MP (n = 480).
The patients were stratified by stage (≤pT2, ≥pT3, N0 or N+), Gleason score, and intent to receive adjuvant radiation, and the presurgical PSA was 7.6 ng/mL. Radiation therapy was allowed in both arms at physician discretion, and 26% intended to receive radiation therapy.
In the ADT only arm, 402 completed the treatment, and 390 completed treatment in the ADT plus chemotherapy arm.
Grade 3 or higher adverse events were more common in the chemotherapy group (56%/30%, P less than .0001). “As for toxicities, the main difference was the presence of leukopenia in the chemotherapy arm,” said Dr. Glode.
The study was funded by Southwest Oncology Group’s Urologic Cancer Outreach Program, Eastern Cooperative Oncology Group, Cancer and Leukemia Group B, Clinical Trials Support Unit, and National Cancer Institute. None of the authors had disclosures.
FROM THE GENITOURINARY CANCERS SYMPOSIUM
Key clinical point: Chemotherapy added to adjuvant androgen deprivation therapy in patients with clinically localized prostate cancer does not improve outcomes.
Major finding: Overall survival was the same in both arms of the study: 87% in the cohort that received ADT only and 86% for those receiving chemotherapy (1.05 [0.78, 1.42], P = .74).
Data source: A phase III randomized trial of 983 patients to determine the utility of adding chemotherapy to adjuvant ADT.
Disclosures: The study was funded by Southwest Oncology Group’s Urologic Cancer Outreach Program, Eastern Cooperative Oncology Group, Cancer and Leukemia Group B, Clinical Trials Support Unit, and National Cancer Institute. None of the authors had disclosures.
VIDEO: Point-of-care assay caught acetaminophen toxicity
A rapid point-of-care assay for acetaminophen-related liver toxicity had a sensitivity of 100% and a specificity of 86%, compared with etiologic diagnosis, based on the results of a multicenter study published in the April issue of Clinical Gastroenterology and Hepatology.
The test might help guide treatment decisions for these patients in the emergency department and intensive care unit, said Dean W. Roberts, PhD, of the University of Arkansas, Little Rock, and his associates.
About 45% of acute liver failure cases in the United States stem from acetaminophen toxicity, but the diagnosis can be hard to confirm because the drug has a short half-life and patients often cannot or will not report an overdose, which also may consist of multiple exposures, limiting the interpretability of the Rumack nonogram. High-pressure liquid chromatography with electrochemical detection (HPLC-EC) accurately detects acetaminophen-protein adducts (3-[cysteine-S-yl] acetaminophen) released by lysed hepatocytes into the peripheral circulation, but this test requires specialized equipment and skilled personnel, the researchers noted (Clin Gastroenterol Hepatol. 2016 Sep 15. doi: 10.1016/j.cgh.2016.09.007).
The point-of-care assay was positive in all 33 patients diagnosed with acetaminophen toxicity, for a test sensitivity of 100%, the researchers reported. The median band amplitude for cases was 584 (range, 222-1,027), significantly lower than that for patients with nonacetaminophen acute liver failure (3,678; range, 394-8,289; P less than .001) or for controls (8,971; range, 5,151-11,108; P less than .001). Band amplitude correlated inversely with adduct levels because AcetaSTAT is a competitive immunoassay – the presence of adducts decreases reactions at the test band, the investigators reported.
AcetaSTAT results were negative for 25 of 29 patients who were initially diagnosed with nonacetaminophen liver failure, for a test specificity of 86%, a positive predictive value of 89%, and a negative predictive value of 100%. Among the remaining four “false positives,” three tested near or above the toxicity threshold on HPLC-EC and were considered positive after further review, the investigators said. The fourth false-positive case was HPLC-EC–negative autoimmune hepatitis.
AcetaSTAT might not catch cases very early after acetaminophen overdose or that have only mild toxicity, the researchers noted. Nonetheless, it can help guide treatment decisions “at the point of clinical care,” they said. “Because the survival rate of acetaminophen acute liver failure is more favorable than that of other causes of acute live failure, assay results could impact future physician referral patterns and reduce medical costs associated with additional tests to determine the etiology of liver injury.”
The National Institute of Diabetes and Digestive and Kidney Diseases funded the study. Dr. Roberts and two coinvestigators are part owners of Acetaminophen Toxicity Diagnostics and have submitted a patent application for the AcetaSTAT serum assay used in this study. There were no other disclosures.
Source: American Gastroenterological Association
A rapid point-of-care assay for acetaminophen-related liver toxicity had a sensitivity of 100% and a specificity of 86%, compared with etiologic diagnosis, based on the results of a multicenter study published in the April issue of Clinical Gastroenterology and Hepatology.
The test might help guide treatment decisions for these patients in the emergency department and intensive care unit, said Dean W. Roberts, PhD, of the University of Arkansas, Little Rock, and his associates.
About 45% of acute liver failure cases in the United States stem from acetaminophen toxicity, but the diagnosis can be hard to confirm because the drug has a short half-life and patients often cannot or will not report an overdose, which also may consist of multiple exposures, limiting the interpretability of the Rumack nonogram. High-pressure liquid chromatography with electrochemical detection (HPLC-EC) accurately detects acetaminophen-protein adducts (3-[cysteine-S-yl] acetaminophen) released by lysed hepatocytes into the peripheral circulation, but this test requires specialized equipment and skilled personnel, the researchers noted (Clin Gastroenterol Hepatol. 2016 Sep 15. doi: 10.1016/j.cgh.2016.09.007).
The point-of-care assay was positive in all 33 patients diagnosed with acetaminophen toxicity, for a test sensitivity of 100%, the researchers reported. The median band amplitude for cases was 584 (range, 222-1,027), significantly lower than that for patients with nonacetaminophen acute liver failure (3,678; range, 394-8,289; P less than .001) or for controls (8,971; range, 5,151-11,108; P less than .001). Band amplitude correlated inversely with adduct levels because AcetaSTAT is a competitive immunoassay – the presence of adducts decreases reactions at the test band, the investigators reported.
AcetaSTAT results were negative for 25 of 29 patients who were initially diagnosed with nonacetaminophen liver failure, for a test specificity of 86%, a positive predictive value of 89%, and a negative predictive value of 100%. Among the remaining four “false positives,” three tested near or above the toxicity threshold on HPLC-EC and were considered positive after further review, the investigators said. The fourth false-positive case was HPLC-EC–negative autoimmune hepatitis.
AcetaSTAT might not catch cases very early after acetaminophen overdose or that have only mild toxicity, the researchers noted. Nonetheless, it can help guide treatment decisions “at the point of clinical care,” they said. “Because the survival rate of acetaminophen acute liver failure is more favorable than that of other causes of acute live failure, assay results could impact future physician referral patterns and reduce medical costs associated with additional tests to determine the etiology of liver injury.”
The National Institute of Diabetes and Digestive and Kidney Diseases funded the study. Dr. Roberts and two coinvestigators are part owners of Acetaminophen Toxicity Diagnostics and have submitted a patent application for the AcetaSTAT serum assay used in this study. There were no other disclosures.
Source: American Gastroenterological Association
A rapid point-of-care assay for acetaminophen-related liver toxicity had a sensitivity of 100% and a specificity of 86%, compared with etiologic diagnosis, based on the results of a multicenter study published in the April issue of Clinical Gastroenterology and Hepatology.
The test might help guide treatment decisions for these patients in the emergency department and intensive care unit, said Dean W. Roberts, PhD, of the University of Arkansas, Little Rock, and his associates.
About 45% of acute liver failure cases in the United States stem from acetaminophen toxicity, but the diagnosis can be hard to confirm because the drug has a short half-life and patients often cannot or will not report an overdose, which also may consist of multiple exposures, limiting the interpretability of the Rumack nonogram. High-pressure liquid chromatography with electrochemical detection (HPLC-EC) accurately detects acetaminophen-protein adducts (3-[cysteine-S-yl] acetaminophen) released by lysed hepatocytes into the peripheral circulation, but this test requires specialized equipment and skilled personnel, the researchers noted (Clin Gastroenterol Hepatol. 2016 Sep 15. doi: 10.1016/j.cgh.2016.09.007).
The point-of-care assay was positive in all 33 patients diagnosed with acetaminophen toxicity, for a test sensitivity of 100%, the researchers reported. The median band amplitude for cases was 584 (range, 222-1,027), significantly lower than that for patients with nonacetaminophen acute liver failure (3,678; range, 394-8,289; P less than .001) or for controls (8,971; range, 5,151-11,108; P less than .001). Band amplitude correlated inversely with adduct levels because AcetaSTAT is a competitive immunoassay – the presence of adducts decreases reactions at the test band, the investigators reported.
AcetaSTAT results were negative for 25 of 29 patients who were initially diagnosed with nonacetaminophen liver failure, for a test specificity of 86%, a positive predictive value of 89%, and a negative predictive value of 100%. Among the remaining four “false positives,” three tested near or above the toxicity threshold on HPLC-EC and were considered positive after further review, the investigators said. The fourth false-positive case was HPLC-EC–negative autoimmune hepatitis.
AcetaSTAT might not catch cases very early after acetaminophen overdose or that have only mild toxicity, the researchers noted. Nonetheless, it can help guide treatment decisions “at the point of clinical care,” they said. “Because the survival rate of acetaminophen acute liver failure is more favorable than that of other causes of acute live failure, assay results could impact future physician referral patterns and reduce medical costs associated with additional tests to determine the etiology of liver injury.”
The National Institute of Diabetes and Digestive and Kidney Diseases funded the study. Dr. Roberts and two coinvestigators are part owners of Acetaminophen Toxicity Diagnostics and have submitted a patent application for the AcetaSTAT serum assay used in this study. There were no other disclosures.
Source: American Gastroenterological Association
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point:
Major finding: Compared with etiologic diagnosis, its sensitivity was 100%, specificity was 86%, positive predictive value was 89%, and negative predictive value was 100%.
Data source: Competitive immunoassays of serum samples from 19 healthy controls, 29 patients with nonacetaminophen acute liver failure, and 33 patients with acetaminophen-induced acute liver failure.
Disclosures: The National Institute of Diabetes and Digestive and Kidney Diseases funded the study. Dr. Roberts and two coinvestigators are part owners of Acetaminophen Toxicity Diagnostics and have submitted a patent application for the AcetaSTAT serum assay used in this study. There were no other disclosures.
Trump Promises Funding Boost for VA and DoD
In his first address to a joint session of Congress, President Donald Trump promised “heroic veterans will get the care they so desperately need,” that he would eliminate the defense sequester, and called for “one of the largest increases in national defense spending in American history.”
“This looks like an increase in resources for us,” VA Secretary David Shulkin, MD, is reported to have told reporters early in the day at an American Legion meeting. “I'm confident this budget is going to reflect the President’s commitment to his ability to deliver on his promises to make veterans care better and stronger.” According to reports, the White House already has approved 37,000 exemptions from the federal hiring ban to help fill the VA’s 45,000 current job vacancies.
Reports also suggest that the military will receive an additional $54 billion in funding, a 10% increase. Details of where that extra money will go have not been released. The President’s military budget is less than the $640 billion budget proposed by Senator John McCain (R- AZ) and represents a 3% increase over the budget that had been projected by President Obama.
The overall impact on federal health care remains unclear. While President Trump held to his promise to repeal and replace the Affordable Care Act, to “invest in women’s health,” and to “give our state governors the resources and flexibility they need with Medicaid to make sure no one is left out,” there were few details of how those promises would be implemented and where the funding will come from.
In the address, the President outlined 4 necessary elements of an Affordable Care Act replacement:
- Americans with pre-existing conditions would have access to coverage, and a stable transition for health care exchanges enrollees;
- The use of tax credits and savings accounts for purchasing private insurance;
- Flexibility for states to expand Medicaid coverage; and
- Legal reforms that “protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately.”
The FDA also drew the President’s attention. Calling the approval process “slow and burdensome,” the President charged that the FDA “keeps too many advances… from reaching those in need.” The agency is still waiting on a nomination for its commissioner position, and it is unclear how it can speed up approval while under the federal hiring freeze.
Recognizing the devastation of opioid addiction, the President also promised to “stop the drugs from pouring into our country and poisoning our youth, and we will expand treatment for those who have become so badly addicted.” The President did not specify whether treatment resources would be exempt from the hiring freeze, incorporated into the Affordable Care Act, or handled in a different manner.
In his first address to a joint session of Congress, President Donald Trump promised “heroic veterans will get the care they so desperately need,” that he would eliminate the defense sequester, and called for “one of the largest increases in national defense spending in American history.”
“This looks like an increase in resources for us,” VA Secretary David Shulkin, MD, is reported to have told reporters early in the day at an American Legion meeting. “I'm confident this budget is going to reflect the President’s commitment to his ability to deliver on his promises to make veterans care better and stronger.” According to reports, the White House already has approved 37,000 exemptions from the federal hiring ban to help fill the VA’s 45,000 current job vacancies.
Reports also suggest that the military will receive an additional $54 billion in funding, a 10% increase. Details of where that extra money will go have not been released. The President’s military budget is less than the $640 billion budget proposed by Senator John McCain (R- AZ) and represents a 3% increase over the budget that had been projected by President Obama.
The overall impact on federal health care remains unclear. While President Trump held to his promise to repeal and replace the Affordable Care Act, to “invest in women’s health,” and to “give our state governors the resources and flexibility they need with Medicaid to make sure no one is left out,” there were few details of how those promises would be implemented and where the funding will come from.
In the address, the President outlined 4 necessary elements of an Affordable Care Act replacement:
- Americans with pre-existing conditions would have access to coverage, and a stable transition for health care exchanges enrollees;
- The use of tax credits and savings accounts for purchasing private insurance;
- Flexibility for states to expand Medicaid coverage; and
- Legal reforms that “protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately.”
The FDA also drew the President’s attention. Calling the approval process “slow and burdensome,” the President charged that the FDA “keeps too many advances… from reaching those in need.” The agency is still waiting on a nomination for its commissioner position, and it is unclear how it can speed up approval while under the federal hiring freeze.
Recognizing the devastation of opioid addiction, the President also promised to “stop the drugs from pouring into our country and poisoning our youth, and we will expand treatment for those who have become so badly addicted.” The President did not specify whether treatment resources would be exempt from the hiring freeze, incorporated into the Affordable Care Act, or handled in a different manner.
In his first address to a joint session of Congress, President Donald Trump promised “heroic veterans will get the care they so desperately need,” that he would eliminate the defense sequester, and called for “one of the largest increases in national defense spending in American history.”
“This looks like an increase in resources for us,” VA Secretary David Shulkin, MD, is reported to have told reporters early in the day at an American Legion meeting. “I'm confident this budget is going to reflect the President’s commitment to his ability to deliver on his promises to make veterans care better and stronger.” According to reports, the White House already has approved 37,000 exemptions from the federal hiring ban to help fill the VA’s 45,000 current job vacancies.
Reports also suggest that the military will receive an additional $54 billion in funding, a 10% increase. Details of where that extra money will go have not been released. The President’s military budget is less than the $640 billion budget proposed by Senator John McCain (R- AZ) and represents a 3% increase over the budget that had been projected by President Obama.
The overall impact on federal health care remains unclear. While President Trump held to his promise to repeal and replace the Affordable Care Act, to “invest in women’s health,” and to “give our state governors the resources and flexibility they need with Medicaid to make sure no one is left out,” there were few details of how those promises would be implemented and where the funding will come from.
In the address, the President outlined 4 necessary elements of an Affordable Care Act replacement:
- Americans with pre-existing conditions would have access to coverage, and a stable transition for health care exchanges enrollees;
- The use of tax credits and savings accounts for purchasing private insurance;
- Flexibility for states to expand Medicaid coverage; and
- Legal reforms that “protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately.”
The FDA also drew the President’s attention. Calling the approval process “slow and burdensome,” the President charged that the FDA “keeps too many advances… from reaching those in need.” The agency is still waiting on a nomination for its commissioner position, and it is unclear how it can speed up approval while under the federal hiring freeze.
Recognizing the devastation of opioid addiction, the President also promised to “stop the drugs from pouring into our country and poisoning our youth, and we will expand treatment for those who have become so badly addicted.” The President did not specify whether treatment resources would be exempt from the hiring freeze, incorporated into the Affordable Care Act, or handled in a different manner.
Treating tardive dyskinesia
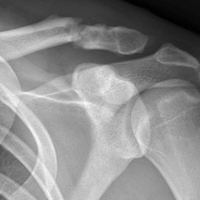
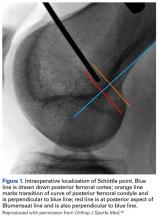
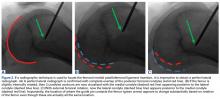
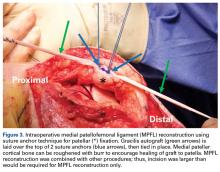
The Patellofemoral Compartment: Making Sense of It
Editor’s Note: One of the goals of the new AJO is to offer solutions to common problems we face as orthopedists. With that in mind, this issue tackles the patellofemoral joint and represents a collaboration between our journal and some of the key leaders of the Patellofemoral Study Group. I’m indebted to my friend and mentor, Jack Farr, for organizing this issue and a continuing patellofemoral series. I know this series will provide an invaluable look into the thought process of true orthopedic legends and find a permanent place on your shelf of orthopedic reference materials.
I’m also pleased to introduce a new feature, our online Lifestyles section. Sometimes, as orthopedists, we spend so much time taking care of others that we forget to look after ourselves and our loved ones. In an effort to make this easier, AJO has collaborated with Inspirato, the premiere luxury destination club. As a member, I’ve enjoyed truly life-changing vacations with my family and now have a way to share that opportunity with our readers. Inspirato is offering a complimentary 6-month Key membership and $250 spending credit to all AJO readers. Simply visit www.inspirato.com/orthopedics to sign up and start booking your vacations like a member. Look for future lifestyle features and special opportunities online in upcoming issues.
—Bryan T. Hanypsiak, MD
The patellofemoral compartment of the knee has been an enigma for many years. Clinicians who enjoy treating patients with knee problems have the choice of either ignoring one-third of the knee or grappling with this unique compartment. In attempting to make sense of this area of the knee, it is necessary to take into account the vast and complex overlay of multiple factors affecting this compartment. These factors span the gamut from psycho-social, to “core to floor” physiologic imbalance, to overuse, to the seemingly more “objective” elements of alignment, stability, morphology, bone, and cartilage.
Fortunately, a small merry band of international experts has made the patellofemoral compartment its “badge of courage” and continues to attempt to make sense of this small mobile sesamoid bone. We have invited a few of these stalwarts to share their experience and wisdom with us in this first of an ongoing patellofemoral series in The American Journal of Orthopedics. I appreciate the honor of assembling the works of these worldly patellofemoral gurus.
How many of us routinely order a “Merchant view”, discuss a “Fulkerson osteotomy”, or tell patients they are out of their Scott Dye “envelope of function” and they need to allow their knee to return to homeostasis through a “core to floor” rehabilitation program? We are lucky to have these living legends offer us insight into their thinking process. I purposely have begun this patellofemoral series with some of my personal mentors to set the tone: think first, understand the problem, design an evidence-based medicine approach and, above all, do no harm. To that point, Dr. Merchant, Dr. Fulkerson, Dr. Dye, and Dr. Post each detail their approach to anterior knee pain, followed by a discussion on nonoperative therapy intervention by Dr. Hiemstra. However, I understand that most readers are surgeons and, therefore I have added two articles to pique your interest: the hot topic of medial patellofemoral ligament (MPFL)—“To repair or not to repair, that is NOT the question.” The question is: “When does repair potentially benefit the patient and when is reconstruction the best approach?” Dr. Duchman and Dr. Bollier address the former, and Dr. Burrus and colleagues discuss optimizing MPFL reconstruction. I hope you enjoy learning from these authors as much as I have while producing this issue.
Am J Orthop. 2017;46(2):64. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Editor’s Note: One of the goals of the new AJO is to offer solutions to common problems we face as orthopedists. With that in mind, this issue tackles the patellofemoral joint and represents a collaboration between our journal and some of the key leaders of the Patellofemoral Study Group. I’m indebted to my friend and mentor, Jack Farr, for organizing this issue and a continuing patellofemoral series. I know this series will provide an invaluable look into the thought process of true orthopedic legends and find a permanent place on your shelf of orthopedic reference materials.
I’m also pleased to introduce a new feature, our online Lifestyles section. Sometimes, as orthopedists, we spend so much time taking care of others that we forget to look after ourselves and our loved ones. In an effort to make this easier, AJO has collaborated with Inspirato, the premiere luxury destination club. As a member, I’ve enjoyed truly life-changing vacations with my family and now have a way to share that opportunity with our readers. Inspirato is offering a complimentary 6-month Key membership and $250 spending credit to all AJO readers. Simply visit www.inspirato.com/orthopedics to sign up and start booking your vacations like a member. Look for future lifestyle features and special opportunities online in upcoming issues.
—Bryan T. Hanypsiak, MD
The patellofemoral compartment of the knee has been an enigma for many years. Clinicians who enjoy treating patients with knee problems have the choice of either ignoring one-third of the knee or grappling with this unique compartment. In attempting to make sense of this area of the knee, it is necessary to take into account the vast and complex overlay of multiple factors affecting this compartment. These factors span the gamut from psycho-social, to “core to floor” physiologic imbalance, to overuse, to the seemingly more “objective” elements of alignment, stability, morphology, bone, and cartilage.
Fortunately, a small merry band of international experts has made the patellofemoral compartment its “badge of courage” and continues to attempt to make sense of this small mobile sesamoid bone. We have invited a few of these stalwarts to share their experience and wisdom with us in this first of an ongoing patellofemoral series in The American Journal of Orthopedics. I appreciate the honor of assembling the works of these worldly patellofemoral gurus.
How many of us routinely order a “Merchant view”, discuss a “Fulkerson osteotomy”, or tell patients they are out of their Scott Dye “envelope of function” and they need to allow their knee to return to homeostasis through a “core to floor” rehabilitation program? We are lucky to have these living legends offer us insight into their thinking process. I purposely have begun this patellofemoral series with some of my personal mentors to set the tone: think first, understand the problem, design an evidence-based medicine approach and, above all, do no harm. To that point, Dr. Merchant, Dr. Fulkerson, Dr. Dye, and Dr. Post each detail their approach to anterior knee pain, followed by a discussion on nonoperative therapy intervention by Dr. Hiemstra. However, I understand that most readers are surgeons and, therefore I have added two articles to pique your interest: the hot topic of medial patellofemoral ligament (MPFL)—“To repair or not to repair, that is NOT the question.” The question is: “When does repair potentially benefit the patient and when is reconstruction the best approach?” Dr. Duchman and Dr. Bollier address the former, and Dr. Burrus and colleagues discuss optimizing MPFL reconstruction. I hope you enjoy learning from these authors as much as I have while producing this issue.
Am J Orthop. 2017;46(2):64. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Editor’s Note: One of the goals of the new AJO is to offer solutions to common problems we face as orthopedists. With that in mind, this issue tackles the patellofemoral joint and represents a collaboration between our journal and some of the key leaders of the Patellofemoral Study Group. I’m indebted to my friend and mentor, Jack Farr, for organizing this issue and a continuing patellofemoral series. I know this series will provide an invaluable look into the thought process of true orthopedic legends and find a permanent place on your shelf of orthopedic reference materials.
I’m also pleased to introduce a new feature, our online Lifestyles section. Sometimes, as orthopedists, we spend so much time taking care of others that we forget to look after ourselves and our loved ones. In an effort to make this easier, AJO has collaborated with Inspirato, the premiere luxury destination club. As a member, I’ve enjoyed truly life-changing vacations with my family and now have a way to share that opportunity with our readers. Inspirato is offering a complimentary 6-month Key membership and $250 spending credit to all AJO readers. Simply visit www.inspirato.com/orthopedics to sign up and start booking your vacations like a member. Look for future lifestyle features and special opportunities online in upcoming issues.
—Bryan T. Hanypsiak, MD
The patellofemoral compartment of the knee has been an enigma for many years. Clinicians who enjoy treating patients with knee problems have the choice of either ignoring one-third of the knee or grappling with this unique compartment. In attempting to make sense of this area of the knee, it is necessary to take into account the vast and complex overlay of multiple factors affecting this compartment. These factors span the gamut from psycho-social, to “core to floor” physiologic imbalance, to overuse, to the seemingly more “objective” elements of alignment, stability, morphology, bone, and cartilage.
Fortunately, a small merry band of international experts has made the patellofemoral compartment its “badge of courage” and continues to attempt to make sense of this small mobile sesamoid bone. We have invited a few of these stalwarts to share their experience and wisdom with us in this first of an ongoing patellofemoral series in The American Journal of Orthopedics. I appreciate the honor of assembling the works of these worldly patellofemoral gurus.
How many of us routinely order a “Merchant view”, discuss a “Fulkerson osteotomy”, or tell patients they are out of their Scott Dye “envelope of function” and they need to allow their knee to return to homeostasis through a “core to floor” rehabilitation program? We are lucky to have these living legends offer us insight into their thinking process. I purposely have begun this patellofemoral series with some of my personal mentors to set the tone: think first, understand the problem, design an evidence-based medicine approach and, above all, do no harm. To that point, Dr. Merchant, Dr. Fulkerson, Dr. Dye, and Dr. Post each detail their approach to anterior knee pain, followed by a discussion on nonoperative therapy intervention by Dr. Hiemstra. However, I understand that most readers are surgeons and, therefore I have added two articles to pique your interest: the hot topic of medial patellofemoral ligament (MPFL)—“To repair or not to repair, that is NOT the question.” The question is: “When does repair potentially benefit the patient and when is reconstruction the best approach?” Dr. Duchman and Dr. Bollier address the former, and Dr. Burrus and colleagues discuss optimizing MPFL reconstruction. I hope you enjoy learning from these authors as much as I have while producing this issue.
Am J Orthop. 2017;46(2):64. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
The Diagnosis and Initial Treatment of Patellofemoral Disorders
Take-Home Points
- Patellofemoral disorders should be classified and diagnosed according to specific diagnostic categories (eg, lateral patellar compression syndrome) based on etiology rather than nondescriptive terminology (eg, internal derangement, patellofemoral pain syndrome).
- Patellofemoral dysplasia defines a spectrum of abnormalities ranging from the mild lateral patellar compression syndrome to the severe recurrent patellar dislocation.
- There is an inverse relationship between patient activity level and underlying patellofemoral dysplasia. This relationship determines threshold levels for each patient becoming symptomatic.
- Patients should be examined for 7 physical abnormalities, and if present, in what severity. These 7 are: vastus medialis obliquus deficiency, medial patellofemoral ligament laxity, lateral retinaculum tightness, increased quadriceps angle, hip abductor weakness, patella alta, and trochlear dysplasia.
- Advanced imaging is rarely, if ever, needed to make a diagnosis or to formulate an initial treatment plan for these common patellofemoral disorders.
To diagnose any disease or disorder implies an understanding of the condition’s cause(s), which should then lead to a logical treatment plan. For all too long, however, the diagnosis and treatment of patellofemoral disorders have been hampered by diagnoses that lack specific definitions based on etiology. A few of these are: internal derangement, chondromalacia patellae, patellar maltracking, and patellofemoral pain syndrome.
To simplify the diagnosis of patellofemoral disorders, we use a clinical classification based on etiology. This system’s defined diagnostic categories are useful in identifying probable cause(s), which can be appropriately evaluated and treated (Table).1 In simple terms, the philosophy of this approach is to try to find out what’s wrong, and try to fix it!
This clinical classification provides a framework for common patellofemoral conditions that are more easily diagnosed, yet is intentionally incomplete omitting rare conditions (eg, tumors, metabolic bone disease, neurologic conditions).
Patellofemoral Dysplasia
Patellofemoral dysplasia (or extensor mechanism malfunction) is a cluster of physical abnormalities relating to the patellofemoral joint that vary from mild to severe and affect the normal function of that joint. As such, patellofemoral dysplasia itself should be considered on a continuum of mild to severe. To simplify the diagnosis, the clinician should systematically identify these factors and their severity. Armed with this information, the clinician can make the diagnosis and formulate a logical treatment plan for each individual patient.
This article focuses on 7 physical abnormalities that are most likely developmental and that can be identified through physical and radiologic examination. When and how each patient with patellofemoral dysplasia becomes symptomatic are determined by 2 key factors: patellofemoral dysplasia severity and activity level (sedentary to strenuous), in an inverse relationship (Figure 1).2
Seven Key Patellofemoral Physical Abnormalities
Of the 7 commonly identified physical abnormalities that affect the normal functioning of the patellofemoral joint, 5 are discovered by physical examination and 2 by radiography; CT and MRI are seldom needed in the initial evaluation. The most accurate and objective method should be used to assess the presence and severity of each abnormality.
The 7 abnormalities are vastus medialis obliquus (VMO) deficiency, medial patellofemoral ligament (MPFL) laxity, lateral retinaculum (LR) tightness, increased quadriceps (Q) angle, hip abductor weakness, patella alta, and trochlear dysplasia. We list these not in order of importance but in the order in which they are usually encountered during initial evaluation. We advocate for examining both knees including axial patellofemoral radiographs because patellofemoral disorders are frequently bilateral. It is helpful to use an abnormality checklist so none are forgotten. Also useful is a simple shorthand for findings: 0 = normal (no abnormality), 1 = mild abnormality, 2 = moderate abnormality, 3 = severe abnormality, with the right knee always recorded first (R/L). For example, severe left MPFL laxity is recorded as 0/3. Numerical values (eg, Q angles) can be directly recorded in this manner: 14°/23°.
1. Vastus Medialis Obliquus Deficiency
VMO deficiency is best seen as the sitting patient actively maintains the unsupported foot and leg at 30° knee flexion. Normally, the VMO inserts into the upper half or third of the medial edge of the patella; a deficient VMO inserts higher into the medial edge of the quadriceps tendon, or it is absent and leaves a characteristic hollow at the medial edge of the patella (Figure 2).4
2. Medial Patellofemoral Ligament Laxity
MPFL laxity is assessed with the lateral glide test. Again, the patient sits, but with quadriceps relaxed and foot and leg supported at 30° knee flexion. With the clinician mentally dividing the patella into vertical quadrants and pushing the patella laterally, the normal patella moves about 1 quadrant or 1 fingerbreadth. Severe MPFL laxity often elicits a positive apprehension response during the test. (Tip: Many patients are unable to relax the quadriceps while sitting; therefore, examine them supine and lift the knee into 30° flexion.) Such laxity usually means the MPFL was torn in a previous dislocation and remains elongated, leaving the patella vulnerable to repeated dislocations. The clinician should be alert to the possibility of hyperelastosis (Ehlers-Danlos syndrome) and a hyper-mobile patella. The opposite limb should be evaluated for asymmetric laxity.
3. Lateral Retinaculum Tightness
LR tightness is assessed with the medial glide test, again with the quadriceps relaxed and the knee supported at 30° flexion. With a normal LR, the patella can be pushed medially about 1 quadrant or 1 fingerbreadth. Some clinicians prefer the lateral tilt-up test, in which the lateral edge of the patella is lifted up, but this method is more difficult to quantify, is affected by the cross-sectional shape of the patella, and lacks consistency.
4. Increased Quadriceps Angle
The Q angle is one of the most important factors in the normal functioning of the patellofemoral joint. For more than a century,8 multiple operations have been used successfully to move the tibial tubercle (TT) and patellar ligament from a lateral position to a medial position thereby decreasing the Q angle. It is only logical to measure this angle at every knee examination to check for an abnormal increase, and the degree. The term quadriceps angle, or Q angle, was first used in 1964 by Brattström,7 who defined it as the “supplemental angle” to the valgus angle formed by the “quadriceps’ resultant” (line of force or vector) “+ patella + ligamentum patellae”. This might be called the dynamic Q angle. With there being no clinical method of measuring the “quadriceps line of force”, or quadriceps vector, clinicians used a line from the anterior superior iliac spine to the center of the patella, yet still called it the Q angle. By convention, this anatomical Q angle has been accepted as the Q angle.
Because the Q angle is the only clinical measurement of TT lateralization at initial evaluation, its measurement should be standardized, accurate, and simple to perform. Placing the patient supine with the lower limb in neutral rotation (patella anterior) and the knee in full extension standardizes the position. In full extension, the tibia reaches its maximum external rotation owing to the terminal “screw home” mechanism. The clinician should center the patella to the trochlear groove (TG) while measuring the Q angle, as it is the relationship of the TT to the trochlea, not to the patella, that is important. If LR tightness prevents the patella from centering, that fact should be recorded during the medial glide test for LR tightness.
Despite the importance of measuring the Q angle, there has been no standard technique. Multiple authors have attempted to define the “normal” Q angle. In 1999, Post9 reviewed 7 articles on the topic and found no agreement. Mean normal Q angles varied widely, from 5° to 23° (SD range, 0.08°-5°). Grelsamer and colleagues,10 using a long-armed goniometer and standard technique, found a mean Q angle of 15.7° for women and 13.3° for men; the small, 2.4° difference between them disappeared when the measurements were corrected for height. Men and women of similar height have similar Q angles. These findings disproved the common misattribution of the differences to the wider female pelvis.
Given this confusion and the lack of accuracy in measuring the Q angle, many, if not most, surgeons turned to special CT and MRI scanning techniques to measure the distance of lateralization from TT to TG (TT-TG distance). This technique, by necessity, enforced a standardization not found in the earlier Q angles studies. Patients were positioned supine with the knee fully extended, and patellar position was ignored in favor of the TG. However, recent articles11-14have called into question the accuracy and usefulness of TT-TG distance as an assessment of TT lateralization. As such, standardized measurement of the Q angle remains a simple, inexpensive, and clinically relevant method of assessing TT lateralization.
The possible causes of an increased Q angle are valgus limb alignment, internal femoral torsion, external tibial torsion, combined internal femoral and external tibial torsion with foot pronation (the “miserable malalignment” of James and colleagues15), and a TT-lateralizing proximal tibial malformation.
5. Hip Abductor Weakness
The step-down test is easily performed in the office by having the patient stand on a short stool or stair and then slowly step down with the opposite limb to just touch the heel and slowly arise again. A positive test is indicated by the Trendelenburg sign, with the pelvis dropping down and away from the symptomatic supporting limb, the flexing knee collapsing into valgus, and the patient tending to wobble and lack stability (Figure 4).16
6. Patella Alta
Patella alta not only allows the patella to escape the confines of the trochlea earlier during active knee extension increasing the risk of patellar dislocation, but also decreases the contact footprint with the trochlea, increasing the patellofemoral joint reaction force and potentially causing patellofemoral pain and even secondary chondrosis. The simplest way to assess patellar height is with a lateral radiograph of the knee. The 3 popular methods (Insall-Salvati, Caton-Deschamps, Blackburn-Peel) all put the normal patellar height ratio at approximately 1:1, ± 20%. Berg and colleagues18 compared radiologic techniques for measuring patellar height ratio and found that Blackburn-Peel was the most accurate, reliable, and reproducible method.
7. Trochlear Dysplasia
Trochlear dysplasia, most simply a flattening of the TG, is perhaps the most important factor effecting normal patellofemoral function. However, it remains the most difficult to correctly address surgically. Senavongse and Amis19 conducted a cadaveric study demonstrating the prime importance of the TG. They found patellar stability was reduced 30% by releasing the VMO, 49% by cutting the MPFL in full knee extension, and 70% by flattening the trochlea. The most common, successful operations for correcting patellar instability depend on changing other factors that guide patellar excursion to compensate for this trochlear flattening.
The simplest way to assess trochlear dysplasia is to measure the sulcus angle on an accurate axial view radiograph of the knee at 45° flexion (Merchant view).20 Dejour and colleagues21 popularized a technique of assessing and classifying trochlear dysplasia from a true lateral radiograph of the knee, which has the advantage of showing the trochlear at its proximal extent. Davies and colleagues22 evaluated the Dejour technique, along with patellar tilt, patellar height, and sulcus angle, to identify a rapid and reproducible radiologic feature that would indicate the need for further analysis by other imaging studies (eg, CT, MRI). They found that, if the sulcus angle was normal, analysis of other radiologic features was unlikely to reveal additional useful information. They also showed a correlation of increasing sulcus angle and severity of those other dysplasia features. Merchant and colleagues20 found a mean normal sulcus angle of 138º (SD, 6º; range, 126º-150º), and Aglietti and colleagues23 confirmed those findings with nearly identical values (mean, 137º; SD, 6º; range, 116º-151º).
Diagnosis and Initial Treatment Plan
Patellofemoral disorders generally are divided into patellofemoral pain and instability, but these 2 diagnostic categories are too broad to be useful. Patellofemoral pain is a symptom. Patellofemoral pain syndrome should never be used as a diagnosis because there is no accepted definition for the cluster of findings that customarily defines a syndrome. At initial evaluation, after the easily diagnosed causes of anterior knee pain (eg, prepatellar bursitis, TT apophysitis, patellar and quadriceps tendinitis) have been ruled out, the clinician should consider types of patellofemoral dysplasia for a presumptive diagnosis, which will then lead to a logical treatment program for each identified disorder. With a presumptive diagnosis established, almost all patients suffering from chronic anterior knee pain without history of injury are treated initially with rest, ice, and nonsteroidal anti-inflammatory drugs to restore joint homeostasis.3
Lateral Patellar Compression Syndrome
In 1975, Ficat and colleagues24 described features of what they called syndrome d’hyperpression externe de la rotule. Two years later, Ficat and Hungerford25 defined the syndrome as one “in which the patella is well centered in the trochlear sulcus and stable, but in which there is a functional lateralization onto a physiologically and often anatomically predominant lateral facet.” Using the tools we have described here, the clinician usually finds the cause(s) of this “functional lateralization.” Four abnormalities—VMO deficiency, LR tightness, increased standardized Q angle, and hip abductor weakness—can cause functional lateralization either alone when severe or in combination when mild or moderate.
For a presumptive diagnosis of LPCS, initial treatment is nonoperative, and successful in about 90% of patients. It should be obvious that most patients with chronic anterior knee pain have quadriceps atrophy. Physical therapy should be specifically focused on quadriceps strengthening, with absolutely no stress placed on the patellofemoral joint in flexion initially, and on hip abductor strengthening. Progressive resistive isometric quadriceps exercises can be performed with a weight-bench technique (Figures 5A-5D).26
Chronic Subluxation of Patella
With the use of axial patellofemoral radiographs (Merchant views),20 the clinician can determine if the “patella is well centered in the trochlear sulcus and stable” (an important part of the definition of LPCS). If the patient has no symptoms of recurrent instability or patellar dislocation, and these radiographs show a laterally subluxed patella (one not well centered in the trochlea), the diagnosis is most likely CSP, a moderate form of patellofemoral dysplasia (section II of the Table). In addition to the 4 abnormalities used in the diagnosis of LPCS (mentioned earlier), trochlear dysplasia also comes into play in the diagnosis of CSP. Just as the other abnormalities can vary from mild to severe, trochlear dysplasia can vary from mild (slightly shallow sulcus angle) to severe (flat or even convex sulcus angle). As the sulcus becomes shallower, the patella slides more laterally, increasing the likelihood of patellar dislocation.
As the patient with CSP gives no history of episodic patellar instability, treatment for CSP is almost identical to that for LPCS, with the primary focus on isometric quadriceps strengthening (DeHaven isometric exercises)27 and hip abductor muscle strengthening. In the presence of CSP radiographically, it is important to use McConnell taping and/or patellar bracing during muscular strengthening. A patient who achieves 20-lb isometric quadriceps strength, demonstrates a normal step-down test, and is assumed to be asymptomatic can be allowed to return to sports activities with use of a patellar brace. The patient should be counseled that there is an increased risk for patellar dislocation because of this chronic subluxation and the shallower sulcus.
As in LPCS, CSP symptoms that persist after dynamic strength is regained may require surgical intervention. The severity of identified abnormal factors (tight LR, increased Q angle, trochlear dysplasia) guides the surgeon in selecting appropriate corrective technique(s).
Recurrent Dislocation of Patella
Admittedly, given the number and subtlety of abnormal factors, the diagnosis of LPCS as a cause of patellofemoral pain can be challenging. However, RDP is at the opposite end of the spectrum. A history of prior patellar dislocation(s) almost always makes the diagnosis of RDP easier. The patient occasionally complains of a recurrent symptom, the knee “going out” or “giving way,” indicating that the diagnosis might be RDP. By carefully asking what the patient was doing and what happened when the knee “went out”, the clinician may be able to determine if the injury stemmed from sudden patellar pain causing reflex inhibition of the quadriceps or was a true dislocation. Both may be described as “going out” or “giving way”.
Assessment for the same 7 abnormalities helps establish the diagnosis, a logical treatment plan, and a guide for indicated surgery. The diagnostic focus is MPFL laxity and trochlear dysplasia. Prior lateral dislocation of the patella almost always requires rupture of the normal MPFL. The infrequent exception is a patient with hyper-elasticity of the skin and multiple joints (Ehlers-Danlos syndrome). Trochlear dysplasia is a significant risk factor for patellar dislocation. If the trochlea is normal and there is no MPFL laxity, the diagnosis of RDP should be questioned.
If surgery is indicated, the surgeon uses a list of the patient’s abnormalities and their severity as a guide in selecting reconstructive techniques. The more abnormalities found and the greater the severity of each, the more techniques are needed to achieve success. Preoperative exercises help speed postoperative recovery by addressing quadriceps and hip abductor weakness. In addition, an active exercise program gives the surgeon insight into the patient’s desire for and commitment to recovery. Other physical abnormalities to be considered in preoperative planning include MPFL laxity, LR tightness, increased Q angle, patella alta, and trochlear dysplasia.
Surgical tips: 1. When releasing the LR, never cut the vastus lateralis tendon, as this has a high likelihood of causing iatrogenic medial patellar subluxation.29 2. When medializing the TT, consider compensating for a shallow trochlea by “over-correcting” the Q angle to 5° to 10° measured with a surgical goniometer intraoperatively.
Summary
Basing clinical classification of disorders on etiology is a simple and effective way to diagnose common patellofemoral conditions. Identifying and rating the severity of patellofemoral dysplasia, using 7 commonly found physical abnormalities, guide the physician to a proper diagnosis and down logical treatment pathways. These principles should be incorporated into the routine evaluation of patellofemoral disorders to optimize diagnosis, formulate a treatment plan, and improve patient outcomes. After all, this is what our patients are asking us to do: Try to find what’s wrong, and then try to fix it!
Am J Orthop. 2017;46(2):68-75. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
1. Merchant AC. Classification of patellofemoral disorders. Arthroscopy. 1988;4(4):235-240.
2. Merchant AC. Patellofemoral disorders: biomechanics, diagnosis, and nonoperative treatment. In: McGinty JB, Caspari RB, Jackson RW, Poehling GG, eds. Operative Arthroscopy. New York, NY: Raven Press; 1991:261-275.
3. Dye SF. The knee as a biologic transmission with an envelope of function: a theory. Clin Orthop Relat Res. 1996;(325):10-18.
4. Merchant AC. A philosophy of the patellofemoral joint: a logical clinical approach. In: Sanchis-Alfonso V, ed. Anterior Knee Pain and Patellar Instability. 2nd ed. London, England: Springer; 2011:519-530.
5. Jan MH, Lin DH, Lin JJ, Lin CH, Cheng CK, Lin YF. Differences in sonographic characteristics of the vastus medialis obliquus between patients with patellofemoral pain syndrome and healthy adults. Am J Sports Med. 2009;37(9):1743-1749.
6. Pattyn E, Verdonk P, Steyaert A, et al. Vastus medialis obliquus atrophy: does it exist in patellofemoral pain syndrome? Am J Sports Med. 2011;39(7):1450-1455.
7. Brattström H. Shape of the intercondylar groove normally and in recurrent dislocation of the patella. A clinical and x-ray anatomical investigation. Acta Orthop Scand Suppl. 1964;68:1-147.
8. Roux D. Luxation habituelle de la rotule: traitement operatoire. Rev Chir Orthop Reparatrice Appar Mot. 1888;8:682-689.
9. Post WR. Clinical evaluation of patients with patellofemoral disorders. Arthroscopy. 1999;15(8):841-851.
10. Grelsamer RP, Dubey A, Weinstein CH. Men and women have similar Q angles: a clinical and trigonometric evaluation. J Bone Joint Surg Br. 2005;87(11):1498-1501.
11. Skelley N, Friedman M, McGinnis M, Smith C, Hillen T, Matava M. Inter- and intraobserver reliability in the MRI measurement of the tibial tubercle-trochlear groove distance and trochlea dysplasia. Am J Sports Med. 2015;43(4):873-878.
12. Tensho K, Akaoka Y, Shimodaira H, et al. What components comprise the measurement of the tibial tuberosity-trochlear groove distance in a patellar dislocation population? J Bone Joint Surg Am. 2015;97(17):1441-1448.
13. Camp CL, Heidenreich MJ, Dahm DL, Stuart MJ, Levy BA, Krych AJ. Individualizing the tibial tubercle-trochlear groove distance: patellar instability ratios that predict recurrent instability. Am J Sports Med. 2016;44(2):393-399.
14. Ridley TJ, Hinckel BB, Kruckeberg BM, Agel J, Arendt EA. Anatomical patella instability risk factors on MRI show sensitivity without specificity in patients with patellofemoral instability: a systematic review. JISAKOS. 2016;1(3):141-152.
15. James SL, Bates BT, Osternig LR. Injuries to runners. Am J Sports Med. 1978;6(2):40-50.
16. Powers CM, Souza RB, Fulkerson JP. Patellofemoral joint. In: Magee DJ, Zachazewski JE, Quillen WS, eds. Pathology and Intervention in Musculoskeletal Rehabilitation. St. Louis, MO: Saunders Elsevier; 2008:601-636.
17. Khayambashi K, Mohammadkhani Z, Ghaznavi K, Lyle MA, Powers CM. The effects of isolated hip abductor and external rotator muscle strengthening on pain, health status, and strength in females with patellofemoral pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2012;42(1):22-29.
18. Berg EE, Mason SL, Lucas MJ. Patellar height ratios. A comparison of four measurement methods. Am J Sports Med. 1996;24(2):218-221.
19. Senavongse W, Amis AA. The effects of articular, retinacular, or muscular deficiencies on patellofemoral joint stability: a biomechanical study in vitro. J Bone Joint Surg Br. 2005;87(4):577-582.
20. Merchant AC, Mercer RL, Jacobsen RH, Cool CR. Roentgenographic analysis of patellofemoral congruence. J Bone Joint Surg Am. 1974;56(7):1391-1396.
21. Dejour H, Neyret P, Walch G. Factors in patellar instability. In: Aichroth PM, Cannon WD Jr, Patel DV, eds. Knee Surgery: Current Practice. London, England: Martin Dunitz; 1992.
22. Davies AP, Costa ML, Shepstone L, Glasgow MM, Donell S. The sulcus angle and malalignment of the extensor mechanism of the knee. J Bone Joint Surg Br. 2000;82(8):1162-1166.
23. Aglietti P, Insall JN, Cerulli G. Patellar pain and incongruence. I: measurements of incongruence. Clin Orthop Relat Res. 1983;(176):217-224.
24. Ficat P, Ficat C, Bailieaux A. External hypertension syndrome of the patella. Its significance in the recognition of arthrosis [in French]. Rev Chir Orthop Reparatrice Appar Mot. 1975;61(1):39-59.
25. Ficat P, Hungerford DS. Disorders of the Patellofemoral Joint. Baltimore, MD: Williams & Wilkins; 1977.
26. Merchant AC. The lateral compression syndrome. In: Fox JM, Del Pizzo W, eds. The Patellofemoral Joint. New York, NY: McGraw-Hill; 1993:157-175.
27. DeHaven KE, Dolan WA, Mayer PJ. Chondromalacia patellae in athletes. Clinical presentation and conservative management. Am J Sports Med. 1979;7(1):5-11.
28. Merchant AC. Patellofemoral joint disorders. In: Chapman MW, ed. Operative Orthopedics. Vol 3. Philadelphia, PA: Lippincott; 1988:2321-2366.
29. Sanchis-Alfonso V, Merchant AC. Iatrogenic medial patellar instability: an avoidable injury. Arthroscopy. 2015;31(8):1628-1632.
Take-Home Points
- Patellofemoral disorders should be classified and diagnosed according to specific diagnostic categories (eg, lateral patellar compression syndrome) based on etiology rather than nondescriptive terminology (eg, internal derangement, patellofemoral pain syndrome).
- Patellofemoral dysplasia defines a spectrum of abnormalities ranging from the mild lateral patellar compression syndrome to the severe recurrent patellar dislocation.
- There is an inverse relationship between patient activity level and underlying patellofemoral dysplasia. This relationship determines threshold levels for each patient becoming symptomatic.
- Patients should be examined for 7 physical abnormalities, and if present, in what severity. These 7 are: vastus medialis obliquus deficiency, medial patellofemoral ligament laxity, lateral retinaculum tightness, increased quadriceps angle, hip abductor weakness, patella alta, and trochlear dysplasia.
- Advanced imaging is rarely, if ever, needed to make a diagnosis or to formulate an initial treatment plan for these common patellofemoral disorders.
To diagnose any disease or disorder implies an understanding of the condition’s cause(s), which should then lead to a logical treatment plan. For all too long, however, the diagnosis and treatment of patellofemoral disorders have been hampered by diagnoses that lack specific definitions based on etiology. A few of these are: internal derangement, chondromalacia patellae, patellar maltracking, and patellofemoral pain syndrome.
To simplify the diagnosis of patellofemoral disorders, we use a clinical classification based on etiology. This system’s defined diagnostic categories are useful in identifying probable cause(s), which can be appropriately evaluated and treated (Table).1 In simple terms, the philosophy of this approach is to try to find out what’s wrong, and try to fix it!
This clinical classification provides a framework for common patellofemoral conditions that are more easily diagnosed, yet is intentionally incomplete omitting rare conditions (eg, tumors, metabolic bone disease, neurologic conditions).
Patellofemoral Dysplasia
Patellofemoral dysplasia (or extensor mechanism malfunction) is a cluster of physical abnormalities relating to the patellofemoral joint that vary from mild to severe and affect the normal function of that joint. As such, patellofemoral dysplasia itself should be considered on a continuum of mild to severe. To simplify the diagnosis, the clinician should systematically identify these factors and their severity. Armed with this information, the clinician can make the diagnosis and formulate a logical treatment plan for each individual patient.
This article focuses on 7 physical abnormalities that are most likely developmental and that can be identified through physical and radiologic examination. When and how each patient with patellofemoral dysplasia becomes symptomatic are determined by 2 key factors: patellofemoral dysplasia severity and activity level (sedentary to strenuous), in an inverse relationship (Figure 1).2
Seven Key Patellofemoral Physical Abnormalities
Of the 7 commonly identified physical abnormalities that affect the normal functioning of the patellofemoral joint, 5 are discovered by physical examination and 2 by radiography; CT and MRI are seldom needed in the initial evaluation. The most accurate and objective method should be used to assess the presence and severity of each abnormality.
The 7 abnormalities are vastus medialis obliquus (VMO) deficiency, medial patellofemoral ligament (MPFL) laxity, lateral retinaculum (LR) tightness, increased quadriceps (Q) angle, hip abductor weakness, patella alta, and trochlear dysplasia. We list these not in order of importance but in the order in which they are usually encountered during initial evaluation. We advocate for examining both knees including axial patellofemoral radiographs because patellofemoral disorders are frequently bilateral. It is helpful to use an abnormality checklist so none are forgotten. Also useful is a simple shorthand for findings: 0 = normal (no abnormality), 1 = mild abnormality, 2 = moderate abnormality, 3 = severe abnormality, with the right knee always recorded first (R/L). For example, severe left MPFL laxity is recorded as 0/3. Numerical values (eg, Q angles) can be directly recorded in this manner: 14°/23°.
1. Vastus Medialis Obliquus Deficiency
VMO deficiency is best seen as the sitting patient actively maintains the unsupported foot and leg at 30° knee flexion. Normally, the VMO inserts into the upper half or third of the medial edge of the patella; a deficient VMO inserts higher into the medial edge of the quadriceps tendon, or it is absent and leaves a characteristic hollow at the medial edge of the patella (Figure 2).4
2. Medial Patellofemoral Ligament Laxity
MPFL laxity is assessed with the lateral glide test. Again, the patient sits, but with quadriceps relaxed and foot and leg supported at 30° knee flexion. With the clinician mentally dividing the patella into vertical quadrants and pushing the patella laterally, the normal patella moves about 1 quadrant or 1 fingerbreadth. Severe MPFL laxity often elicits a positive apprehension response during the test. (Tip: Many patients are unable to relax the quadriceps while sitting; therefore, examine them supine and lift the knee into 30° flexion.) Such laxity usually means the MPFL was torn in a previous dislocation and remains elongated, leaving the patella vulnerable to repeated dislocations. The clinician should be alert to the possibility of hyperelastosis (Ehlers-Danlos syndrome) and a hyper-mobile patella. The opposite limb should be evaluated for asymmetric laxity.
3. Lateral Retinaculum Tightness
LR tightness is assessed with the medial glide test, again with the quadriceps relaxed and the knee supported at 30° flexion. With a normal LR, the patella can be pushed medially about 1 quadrant or 1 fingerbreadth. Some clinicians prefer the lateral tilt-up test, in which the lateral edge of the patella is lifted up, but this method is more difficult to quantify, is affected by the cross-sectional shape of the patella, and lacks consistency.
4. Increased Quadriceps Angle
The Q angle is one of the most important factors in the normal functioning of the patellofemoral joint. For more than a century,8 multiple operations have been used successfully to move the tibial tubercle (TT) and patellar ligament from a lateral position to a medial position thereby decreasing the Q angle. It is only logical to measure this angle at every knee examination to check for an abnormal increase, and the degree. The term quadriceps angle, or Q angle, was first used in 1964 by Brattström,7 who defined it as the “supplemental angle” to the valgus angle formed by the “quadriceps’ resultant” (line of force or vector) “+ patella + ligamentum patellae”. This might be called the dynamic Q angle. With there being no clinical method of measuring the “quadriceps line of force”, or quadriceps vector, clinicians used a line from the anterior superior iliac spine to the center of the patella, yet still called it the Q angle. By convention, this anatomical Q angle has been accepted as the Q angle.
Because the Q angle is the only clinical measurement of TT lateralization at initial evaluation, its measurement should be standardized, accurate, and simple to perform. Placing the patient supine with the lower limb in neutral rotation (patella anterior) and the knee in full extension standardizes the position. In full extension, the tibia reaches its maximum external rotation owing to the terminal “screw home” mechanism. The clinician should center the patella to the trochlear groove (TG) while measuring the Q angle, as it is the relationship of the TT to the trochlea, not to the patella, that is important. If LR tightness prevents the patella from centering, that fact should be recorded during the medial glide test for LR tightness.
Despite the importance of measuring the Q angle, there has been no standard technique. Multiple authors have attempted to define the “normal” Q angle. In 1999, Post9 reviewed 7 articles on the topic and found no agreement. Mean normal Q angles varied widely, from 5° to 23° (SD range, 0.08°-5°). Grelsamer and colleagues,10 using a long-armed goniometer and standard technique, found a mean Q angle of 15.7° for women and 13.3° for men; the small, 2.4° difference between them disappeared when the measurements were corrected for height. Men and women of similar height have similar Q angles. These findings disproved the common misattribution of the differences to the wider female pelvis.
Given this confusion and the lack of accuracy in measuring the Q angle, many, if not most, surgeons turned to special CT and MRI scanning techniques to measure the distance of lateralization from TT to TG (TT-TG distance). This technique, by necessity, enforced a standardization not found in the earlier Q angles studies. Patients were positioned supine with the knee fully extended, and patellar position was ignored in favor of the TG. However, recent articles11-14have called into question the accuracy and usefulness of TT-TG distance as an assessment of TT lateralization. As such, standardized measurement of the Q angle remains a simple, inexpensive, and clinically relevant method of assessing TT lateralization.
The possible causes of an increased Q angle are valgus limb alignment, internal femoral torsion, external tibial torsion, combined internal femoral and external tibial torsion with foot pronation (the “miserable malalignment” of James and colleagues15), and a TT-lateralizing proximal tibial malformation.
5. Hip Abductor Weakness
The step-down test is easily performed in the office by having the patient stand on a short stool or stair and then slowly step down with the opposite limb to just touch the heel and slowly arise again. A positive test is indicated by the Trendelenburg sign, with the pelvis dropping down and away from the symptomatic supporting limb, the flexing knee collapsing into valgus, and the patient tending to wobble and lack stability (Figure 4).16
6. Patella Alta
Patella alta not only allows the patella to escape the confines of the trochlea earlier during active knee extension increasing the risk of patellar dislocation, but also decreases the contact footprint with the trochlea, increasing the patellofemoral joint reaction force and potentially causing patellofemoral pain and even secondary chondrosis. The simplest way to assess patellar height is with a lateral radiograph of the knee. The 3 popular methods (Insall-Salvati, Caton-Deschamps, Blackburn-Peel) all put the normal patellar height ratio at approximately 1:1, ± 20%. Berg and colleagues18 compared radiologic techniques for measuring patellar height ratio and found that Blackburn-Peel was the most accurate, reliable, and reproducible method.
7. Trochlear Dysplasia
Trochlear dysplasia, most simply a flattening of the TG, is perhaps the most important factor effecting normal patellofemoral function. However, it remains the most difficult to correctly address surgically. Senavongse and Amis19 conducted a cadaveric study demonstrating the prime importance of the TG. They found patellar stability was reduced 30% by releasing the VMO, 49% by cutting the MPFL in full knee extension, and 70% by flattening the trochlea. The most common, successful operations for correcting patellar instability depend on changing other factors that guide patellar excursion to compensate for this trochlear flattening.
The simplest way to assess trochlear dysplasia is to measure the sulcus angle on an accurate axial view radiograph of the knee at 45° flexion (Merchant view).20 Dejour and colleagues21 popularized a technique of assessing and classifying trochlear dysplasia from a true lateral radiograph of the knee, which has the advantage of showing the trochlear at its proximal extent. Davies and colleagues22 evaluated the Dejour technique, along with patellar tilt, patellar height, and sulcus angle, to identify a rapid and reproducible radiologic feature that would indicate the need for further analysis by other imaging studies (eg, CT, MRI). They found that, if the sulcus angle was normal, analysis of other radiologic features was unlikely to reveal additional useful information. They also showed a correlation of increasing sulcus angle and severity of those other dysplasia features. Merchant and colleagues20 found a mean normal sulcus angle of 138º (SD, 6º; range, 126º-150º), and Aglietti and colleagues23 confirmed those findings with nearly identical values (mean, 137º; SD, 6º; range, 116º-151º).
Diagnosis and Initial Treatment Plan
Patellofemoral disorders generally are divided into patellofemoral pain and instability, but these 2 diagnostic categories are too broad to be useful. Patellofemoral pain is a symptom. Patellofemoral pain syndrome should never be used as a diagnosis because there is no accepted definition for the cluster of findings that customarily defines a syndrome. At initial evaluation, after the easily diagnosed causes of anterior knee pain (eg, prepatellar bursitis, TT apophysitis, patellar and quadriceps tendinitis) have been ruled out, the clinician should consider types of patellofemoral dysplasia for a presumptive diagnosis, which will then lead to a logical treatment program for each identified disorder. With a presumptive diagnosis established, almost all patients suffering from chronic anterior knee pain without history of injury are treated initially with rest, ice, and nonsteroidal anti-inflammatory drugs to restore joint homeostasis.3
Lateral Patellar Compression Syndrome
In 1975, Ficat and colleagues24 described features of what they called syndrome d’hyperpression externe de la rotule. Two years later, Ficat and Hungerford25 defined the syndrome as one “in which the patella is well centered in the trochlear sulcus and stable, but in which there is a functional lateralization onto a physiologically and often anatomically predominant lateral facet.” Using the tools we have described here, the clinician usually finds the cause(s) of this “functional lateralization.” Four abnormalities—VMO deficiency, LR tightness, increased standardized Q angle, and hip abductor weakness—can cause functional lateralization either alone when severe or in combination when mild or moderate.
For a presumptive diagnosis of LPCS, initial treatment is nonoperative, and successful in about 90% of patients. It should be obvious that most patients with chronic anterior knee pain have quadriceps atrophy. Physical therapy should be specifically focused on quadriceps strengthening, with absolutely no stress placed on the patellofemoral joint in flexion initially, and on hip abductor strengthening. Progressive resistive isometric quadriceps exercises can be performed with a weight-bench technique (Figures 5A-5D).26
Chronic Subluxation of Patella
With the use of axial patellofemoral radiographs (Merchant views),20 the clinician can determine if the “patella is well centered in the trochlear sulcus and stable” (an important part of the definition of LPCS). If the patient has no symptoms of recurrent instability or patellar dislocation, and these radiographs show a laterally subluxed patella (one not well centered in the trochlea), the diagnosis is most likely CSP, a moderate form of patellofemoral dysplasia (section II of the Table). In addition to the 4 abnormalities used in the diagnosis of LPCS (mentioned earlier), trochlear dysplasia also comes into play in the diagnosis of CSP. Just as the other abnormalities can vary from mild to severe, trochlear dysplasia can vary from mild (slightly shallow sulcus angle) to severe (flat or even convex sulcus angle). As the sulcus becomes shallower, the patella slides more laterally, increasing the likelihood of patellar dislocation.
As the patient with CSP gives no history of episodic patellar instability, treatment for CSP is almost identical to that for LPCS, with the primary focus on isometric quadriceps strengthening (DeHaven isometric exercises)27 and hip abductor muscle strengthening. In the presence of CSP radiographically, it is important to use McConnell taping and/or patellar bracing during muscular strengthening. A patient who achieves 20-lb isometric quadriceps strength, demonstrates a normal step-down test, and is assumed to be asymptomatic can be allowed to return to sports activities with use of a patellar brace. The patient should be counseled that there is an increased risk for patellar dislocation because of this chronic subluxation and the shallower sulcus.
As in LPCS, CSP symptoms that persist after dynamic strength is regained may require surgical intervention. The severity of identified abnormal factors (tight LR, increased Q angle, trochlear dysplasia) guides the surgeon in selecting appropriate corrective technique(s).
Recurrent Dislocation of Patella
Admittedly, given the number and subtlety of abnormal factors, the diagnosis of LPCS as a cause of patellofemoral pain can be challenging. However, RDP is at the opposite end of the spectrum. A history of prior patellar dislocation(s) almost always makes the diagnosis of RDP easier. The patient occasionally complains of a recurrent symptom, the knee “going out” or “giving way,” indicating that the diagnosis might be RDP. By carefully asking what the patient was doing and what happened when the knee “went out”, the clinician may be able to determine if the injury stemmed from sudden patellar pain causing reflex inhibition of the quadriceps or was a true dislocation. Both may be described as “going out” or “giving way”.
Assessment for the same 7 abnormalities helps establish the diagnosis, a logical treatment plan, and a guide for indicated surgery. The diagnostic focus is MPFL laxity and trochlear dysplasia. Prior lateral dislocation of the patella almost always requires rupture of the normal MPFL. The infrequent exception is a patient with hyper-elasticity of the skin and multiple joints (Ehlers-Danlos syndrome). Trochlear dysplasia is a significant risk factor for patellar dislocation. If the trochlea is normal and there is no MPFL laxity, the diagnosis of RDP should be questioned.
If surgery is indicated, the surgeon uses a list of the patient’s abnormalities and their severity as a guide in selecting reconstructive techniques. The more abnormalities found and the greater the severity of each, the more techniques are needed to achieve success. Preoperative exercises help speed postoperative recovery by addressing quadriceps and hip abductor weakness. In addition, an active exercise program gives the surgeon insight into the patient’s desire for and commitment to recovery. Other physical abnormalities to be considered in preoperative planning include MPFL laxity, LR tightness, increased Q angle, patella alta, and trochlear dysplasia.
Surgical tips: 1. When releasing the LR, never cut the vastus lateralis tendon, as this has a high likelihood of causing iatrogenic medial patellar subluxation.29 2. When medializing the TT, consider compensating for a shallow trochlea by “over-correcting” the Q angle to 5° to 10° measured with a surgical goniometer intraoperatively.
Summary
Basing clinical classification of disorders on etiology is a simple and effective way to diagnose common patellofemoral conditions. Identifying and rating the severity of patellofemoral dysplasia, using 7 commonly found physical abnormalities, guide the physician to a proper diagnosis and down logical treatment pathways. These principles should be incorporated into the routine evaluation of patellofemoral disorders to optimize diagnosis, formulate a treatment plan, and improve patient outcomes. After all, this is what our patients are asking us to do: Try to find what’s wrong, and then try to fix it!
Am J Orthop. 2017;46(2):68-75. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Take-Home Points
- Patellofemoral disorders should be classified and diagnosed according to specific diagnostic categories (eg, lateral patellar compression syndrome) based on etiology rather than nondescriptive terminology (eg, internal derangement, patellofemoral pain syndrome).
- Patellofemoral dysplasia defines a spectrum of abnormalities ranging from the mild lateral patellar compression syndrome to the severe recurrent patellar dislocation.
- There is an inverse relationship between patient activity level and underlying patellofemoral dysplasia. This relationship determines threshold levels for each patient becoming symptomatic.
- Patients should be examined for 7 physical abnormalities, and if present, in what severity. These 7 are: vastus medialis obliquus deficiency, medial patellofemoral ligament laxity, lateral retinaculum tightness, increased quadriceps angle, hip abductor weakness, patella alta, and trochlear dysplasia.
- Advanced imaging is rarely, if ever, needed to make a diagnosis or to formulate an initial treatment plan for these common patellofemoral disorders.
To diagnose any disease or disorder implies an understanding of the condition’s cause(s), which should then lead to a logical treatment plan. For all too long, however, the diagnosis and treatment of patellofemoral disorders have been hampered by diagnoses that lack specific definitions based on etiology. A few of these are: internal derangement, chondromalacia patellae, patellar maltracking, and patellofemoral pain syndrome.
To simplify the diagnosis of patellofemoral disorders, we use a clinical classification based on etiology. This system’s defined diagnostic categories are useful in identifying probable cause(s), which can be appropriately evaluated and treated (Table).1 In simple terms, the philosophy of this approach is to try to find out what’s wrong, and try to fix it!
This clinical classification provides a framework for common patellofemoral conditions that are more easily diagnosed, yet is intentionally incomplete omitting rare conditions (eg, tumors, metabolic bone disease, neurologic conditions).
Patellofemoral Dysplasia
Patellofemoral dysplasia (or extensor mechanism malfunction) is a cluster of physical abnormalities relating to the patellofemoral joint that vary from mild to severe and affect the normal function of that joint. As such, patellofemoral dysplasia itself should be considered on a continuum of mild to severe. To simplify the diagnosis, the clinician should systematically identify these factors and their severity. Armed with this information, the clinician can make the diagnosis and formulate a logical treatment plan for each individual patient.
This article focuses on 7 physical abnormalities that are most likely developmental and that can be identified through physical and radiologic examination. When and how each patient with patellofemoral dysplasia becomes symptomatic are determined by 2 key factors: patellofemoral dysplasia severity and activity level (sedentary to strenuous), in an inverse relationship (Figure 1).2
Seven Key Patellofemoral Physical Abnormalities
Of the 7 commonly identified physical abnormalities that affect the normal functioning of the patellofemoral joint, 5 are discovered by physical examination and 2 by radiography; CT and MRI are seldom needed in the initial evaluation. The most accurate and objective method should be used to assess the presence and severity of each abnormality.
The 7 abnormalities are vastus medialis obliquus (VMO) deficiency, medial patellofemoral ligament (MPFL) laxity, lateral retinaculum (LR) tightness, increased quadriceps (Q) angle, hip abductor weakness, patella alta, and trochlear dysplasia. We list these not in order of importance but in the order in which they are usually encountered during initial evaluation. We advocate for examining both knees including axial patellofemoral radiographs because patellofemoral disorders are frequently bilateral. It is helpful to use an abnormality checklist so none are forgotten. Also useful is a simple shorthand for findings: 0 = normal (no abnormality), 1 = mild abnormality, 2 = moderate abnormality, 3 = severe abnormality, with the right knee always recorded first (R/L). For example, severe left MPFL laxity is recorded as 0/3. Numerical values (eg, Q angles) can be directly recorded in this manner: 14°/23°.
1. Vastus Medialis Obliquus Deficiency
VMO deficiency is best seen as the sitting patient actively maintains the unsupported foot and leg at 30° knee flexion. Normally, the VMO inserts into the upper half or third of the medial edge of the patella; a deficient VMO inserts higher into the medial edge of the quadriceps tendon, or it is absent and leaves a characteristic hollow at the medial edge of the patella (Figure 2).4
2. Medial Patellofemoral Ligament Laxity
MPFL laxity is assessed with the lateral glide test. Again, the patient sits, but with quadriceps relaxed and foot and leg supported at 30° knee flexion. With the clinician mentally dividing the patella into vertical quadrants and pushing the patella laterally, the normal patella moves about 1 quadrant or 1 fingerbreadth. Severe MPFL laxity often elicits a positive apprehension response during the test. (Tip: Many patients are unable to relax the quadriceps while sitting; therefore, examine them supine and lift the knee into 30° flexion.) Such laxity usually means the MPFL was torn in a previous dislocation and remains elongated, leaving the patella vulnerable to repeated dislocations. The clinician should be alert to the possibility of hyperelastosis (Ehlers-Danlos syndrome) and a hyper-mobile patella. The opposite limb should be evaluated for asymmetric laxity.
3. Lateral Retinaculum Tightness
LR tightness is assessed with the medial glide test, again with the quadriceps relaxed and the knee supported at 30° flexion. With a normal LR, the patella can be pushed medially about 1 quadrant or 1 fingerbreadth. Some clinicians prefer the lateral tilt-up test, in which the lateral edge of the patella is lifted up, but this method is more difficult to quantify, is affected by the cross-sectional shape of the patella, and lacks consistency.
4. Increased Quadriceps Angle
The Q angle is one of the most important factors in the normal functioning of the patellofemoral joint. For more than a century,8 multiple operations have been used successfully to move the tibial tubercle (TT) and patellar ligament from a lateral position to a medial position thereby decreasing the Q angle. It is only logical to measure this angle at every knee examination to check for an abnormal increase, and the degree. The term quadriceps angle, or Q angle, was first used in 1964 by Brattström,7 who defined it as the “supplemental angle” to the valgus angle formed by the “quadriceps’ resultant” (line of force or vector) “+ patella + ligamentum patellae”. This might be called the dynamic Q angle. With there being no clinical method of measuring the “quadriceps line of force”, or quadriceps vector, clinicians used a line from the anterior superior iliac spine to the center of the patella, yet still called it the Q angle. By convention, this anatomical Q angle has been accepted as the Q angle.
Because the Q angle is the only clinical measurement of TT lateralization at initial evaluation, its measurement should be standardized, accurate, and simple to perform. Placing the patient supine with the lower limb in neutral rotation (patella anterior) and the knee in full extension standardizes the position. In full extension, the tibia reaches its maximum external rotation owing to the terminal “screw home” mechanism. The clinician should center the patella to the trochlear groove (TG) while measuring the Q angle, as it is the relationship of the TT to the trochlea, not to the patella, that is important. If LR tightness prevents the patella from centering, that fact should be recorded during the medial glide test for LR tightness.
Despite the importance of measuring the Q angle, there has been no standard technique. Multiple authors have attempted to define the “normal” Q angle. In 1999, Post9 reviewed 7 articles on the topic and found no agreement. Mean normal Q angles varied widely, from 5° to 23° (SD range, 0.08°-5°). Grelsamer and colleagues,10 using a long-armed goniometer and standard technique, found a mean Q angle of 15.7° for women and 13.3° for men; the small, 2.4° difference between them disappeared when the measurements were corrected for height. Men and women of similar height have similar Q angles. These findings disproved the common misattribution of the differences to the wider female pelvis.
Given this confusion and the lack of accuracy in measuring the Q angle, many, if not most, surgeons turned to special CT and MRI scanning techniques to measure the distance of lateralization from TT to TG (TT-TG distance). This technique, by necessity, enforced a standardization not found in the earlier Q angles studies. Patients were positioned supine with the knee fully extended, and patellar position was ignored in favor of the TG. However, recent articles11-14have called into question the accuracy and usefulness of TT-TG distance as an assessment of TT lateralization. As such, standardized measurement of the Q angle remains a simple, inexpensive, and clinically relevant method of assessing TT lateralization.
The possible causes of an increased Q angle are valgus limb alignment, internal femoral torsion, external tibial torsion, combined internal femoral and external tibial torsion with foot pronation (the “miserable malalignment” of James and colleagues15), and a TT-lateralizing proximal tibial malformation.
5. Hip Abductor Weakness
The step-down test is easily performed in the office by having the patient stand on a short stool or stair and then slowly step down with the opposite limb to just touch the heel and slowly arise again. A positive test is indicated by the Trendelenburg sign, with the pelvis dropping down and away from the symptomatic supporting limb, the flexing knee collapsing into valgus, and the patient tending to wobble and lack stability (Figure 4).16
6. Patella Alta
Patella alta not only allows the patella to escape the confines of the trochlea earlier during active knee extension increasing the risk of patellar dislocation, but also decreases the contact footprint with the trochlea, increasing the patellofemoral joint reaction force and potentially causing patellofemoral pain and even secondary chondrosis. The simplest way to assess patellar height is with a lateral radiograph of the knee. The 3 popular methods (Insall-Salvati, Caton-Deschamps, Blackburn-Peel) all put the normal patellar height ratio at approximately 1:1, ± 20%. Berg and colleagues18 compared radiologic techniques for measuring patellar height ratio and found that Blackburn-Peel was the most accurate, reliable, and reproducible method.
7. Trochlear Dysplasia
Trochlear dysplasia, most simply a flattening of the TG, is perhaps the most important factor effecting normal patellofemoral function. However, it remains the most difficult to correctly address surgically. Senavongse and Amis19 conducted a cadaveric study demonstrating the prime importance of the TG. They found patellar stability was reduced 30% by releasing the VMO, 49% by cutting the MPFL in full knee extension, and 70% by flattening the trochlea. The most common, successful operations for correcting patellar instability depend on changing other factors that guide patellar excursion to compensate for this trochlear flattening.
The simplest way to assess trochlear dysplasia is to measure the sulcus angle on an accurate axial view radiograph of the knee at 45° flexion (Merchant view).20 Dejour and colleagues21 popularized a technique of assessing and classifying trochlear dysplasia from a true lateral radiograph of the knee, which has the advantage of showing the trochlear at its proximal extent. Davies and colleagues22 evaluated the Dejour technique, along with patellar tilt, patellar height, and sulcus angle, to identify a rapid and reproducible radiologic feature that would indicate the need for further analysis by other imaging studies (eg, CT, MRI). They found that, if the sulcus angle was normal, analysis of other radiologic features was unlikely to reveal additional useful information. They also showed a correlation of increasing sulcus angle and severity of those other dysplasia features. Merchant and colleagues20 found a mean normal sulcus angle of 138º (SD, 6º; range, 126º-150º), and Aglietti and colleagues23 confirmed those findings with nearly identical values (mean, 137º; SD, 6º; range, 116º-151º).
Diagnosis and Initial Treatment Plan
Patellofemoral disorders generally are divided into patellofemoral pain and instability, but these 2 diagnostic categories are too broad to be useful. Patellofemoral pain is a symptom. Patellofemoral pain syndrome should never be used as a diagnosis because there is no accepted definition for the cluster of findings that customarily defines a syndrome. At initial evaluation, after the easily diagnosed causes of anterior knee pain (eg, prepatellar bursitis, TT apophysitis, patellar and quadriceps tendinitis) have been ruled out, the clinician should consider types of patellofemoral dysplasia for a presumptive diagnosis, which will then lead to a logical treatment program for each identified disorder. With a presumptive diagnosis established, almost all patients suffering from chronic anterior knee pain without history of injury are treated initially with rest, ice, and nonsteroidal anti-inflammatory drugs to restore joint homeostasis.3
Lateral Patellar Compression Syndrome
In 1975, Ficat and colleagues24 described features of what they called syndrome d’hyperpression externe de la rotule. Two years later, Ficat and Hungerford25 defined the syndrome as one “in which the patella is well centered in the trochlear sulcus and stable, but in which there is a functional lateralization onto a physiologically and often anatomically predominant lateral facet.” Using the tools we have described here, the clinician usually finds the cause(s) of this “functional lateralization.” Four abnormalities—VMO deficiency, LR tightness, increased standardized Q angle, and hip abductor weakness—can cause functional lateralization either alone when severe or in combination when mild or moderate.
For a presumptive diagnosis of LPCS, initial treatment is nonoperative, and successful in about 90% of patients. It should be obvious that most patients with chronic anterior knee pain have quadriceps atrophy. Physical therapy should be specifically focused on quadriceps strengthening, with absolutely no stress placed on the patellofemoral joint in flexion initially, and on hip abductor strengthening. Progressive resistive isometric quadriceps exercises can be performed with a weight-bench technique (Figures 5A-5D).26
Chronic Subluxation of Patella
With the use of axial patellofemoral radiographs (Merchant views),20 the clinician can determine if the “patella is well centered in the trochlear sulcus and stable” (an important part of the definition of LPCS). If the patient has no symptoms of recurrent instability or patellar dislocation, and these radiographs show a laterally subluxed patella (one not well centered in the trochlea), the diagnosis is most likely CSP, a moderate form of patellofemoral dysplasia (section II of the Table). In addition to the 4 abnormalities used in the diagnosis of LPCS (mentioned earlier), trochlear dysplasia also comes into play in the diagnosis of CSP. Just as the other abnormalities can vary from mild to severe, trochlear dysplasia can vary from mild (slightly shallow sulcus angle) to severe (flat or even convex sulcus angle). As the sulcus becomes shallower, the patella slides more laterally, increasing the likelihood of patellar dislocation.
As the patient with CSP gives no history of episodic patellar instability, treatment for CSP is almost identical to that for LPCS, with the primary focus on isometric quadriceps strengthening (DeHaven isometric exercises)27 and hip abductor muscle strengthening. In the presence of CSP radiographically, it is important to use McConnell taping and/or patellar bracing during muscular strengthening. A patient who achieves 20-lb isometric quadriceps strength, demonstrates a normal step-down test, and is assumed to be asymptomatic can be allowed to return to sports activities with use of a patellar brace. The patient should be counseled that there is an increased risk for patellar dislocation because of this chronic subluxation and the shallower sulcus.
As in LPCS, CSP symptoms that persist after dynamic strength is regained may require surgical intervention. The severity of identified abnormal factors (tight LR, increased Q angle, trochlear dysplasia) guides the surgeon in selecting appropriate corrective technique(s).
Recurrent Dislocation of Patella
Admittedly, given the number and subtlety of abnormal factors, the diagnosis of LPCS as a cause of patellofemoral pain can be challenging. However, RDP is at the opposite end of the spectrum. A history of prior patellar dislocation(s) almost always makes the diagnosis of RDP easier. The patient occasionally complains of a recurrent symptom, the knee “going out” or “giving way,” indicating that the diagnosis might be RDP. By carefully asking what the patient was doing and what happened when the knee “went out”, the clinician may be able to determine if the injury stemmed from sudden patellar pain causing reflex inhibition of the quadriceps or was a true dislocation. Both may be described as “going out” or “giving way”.
Assessment for the same 7 abnormalities helps establish the diagnosis, a logical treatment plan, and a guide for indicated surgery. The diagnostic focus is MPFL laxity and trochlear dysplasia. Prior lateral dislocation of the patella almost always requires rupture of the normal MPFL. The infrequent exception is a patient with hyper-elasticity of the skin and multiple joints (Ehlers-Danlos syndrome). Trochlear dysplasia is a significant risk factor for patellar dislocation. If the trochlea is normal and there is no MPFL laxity, the diagnosis of RDP should be questioned.
If surgery is indicated, the surgeon uses a list of the patient’s abnormalities and their severity as a guide in selecting reconstructive techniques. The more abnormalities found and the greater the severity of each, the more techniques are needed to achieve success. Preoperative exercises help speed postoperative recovery by addressing quadriceps and hip abductor weakness. In addition, an active exercise program gives the surgeon insight into the patient’s desire for and commitment to recovery. Other physical abnormalities to be considered in preoperative planning include MPFL laxity, LR tightness, increased Q angle, patella alta, and trochlear dysplasia.
Surgical tips: 1. When releasing the LR, never cut the vastus lateralis tendon, as this has a high likelihood of causing iatrogenic medial patellar subluxation.29 2. When medializing the TT, consider compensating for a shallow trochlea by “over-correcting” the Q angle to 5° to 10° measured with a surgical goniometer intraoperatively.
Summary
Basing clinical classification of disorders on etiology is a simple and effective way to diagnose common patellofemoral conditions. Identifying and rating the severity of patellofemoral dysplasia, using 7 commonly found physical abnormalities, guide the physician to a proper diagnosis and down logical treatment pathways. These principles should be incorporated into the routine evaluation of patellofemoral disorders to optimize diagnosis, formulate a treatment plan, and improve patient outcomes. After all, this is what our patients are asking us to do: Try to find what’s wrong, and then try to fix it!
Am J Orthop. 2017;46(2):68-75. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
1. Merchant AC. Classification of patellofemoral disorders. Arthroscopy. 1988;4(4):235-240.
2. Merchant AC. Patellofemoral disorders: biomechanics, diagnosis, and nonoperative treatment. In: McGinty JB, Caspari RB, Jackson RW, Poehling GG, eds. Operative Arthroscopy. New York, NY: Raven Press; 1991:261-275.
3. Dye SF. The knee as a biologic transmission with an envelope of function: a theory. Clin Orthop Relat Res. 1996;(325):10-18.
4. Merchant AC. A philosophy of the patellofemoral joint: a logical clinical approach. In: Sanchis-Alfonso V, ed. Anterior Knee Pain and Patellar Instability. 2nd ed. London, England: Springer; 2011:519-530.
5. Jan MH, Lin DH, Lin JJ, Lin CH, Cheng CK, Lin YF. Differences in sonographic characteristics of the vastus medialis obliquus between patients with patellofemoral pain syndrome and healthy adults. Am J Sports Med. 2009;37(9):1743-1749.
6. Pattyn E, Verdonk P, Steyaert A, et al. Vastus medialis obliquus atrophy: does it exist in patellofemoral pain syndrome? Am J Sports Med. 2011;39(7):1450-1455.
7. Brattström H. Shape of the intercondylar groove normally and in recurrent dislocation of the patella. A clinical and x-ray anatomical investigation. Acta Orthop Scand Suppl. 1964;68:1-147.
8. Roux D. Luxation habituelle de la rotule: traitement operatoire. Rev Chir Orthop Reparatrice Appar Mot. 1888;8:682-689.
9. Post WR. Clinical evaluation of patients with patellofemoral disorders. Arthroscopy. 1999;15(8):841-851.
10. Grelsamer RP, Dubey A, Weinstein CH. Men and women have similar Q angles: a clinical and trigonometric evaluation. J Bone Joint Surg Br. 2005;87(11):1498-1501.
11. Skelley N, Friedman M, McGinnis M, Smith C, Hillen T, Matava M. Inter- and intraobserver reliability in the MRI measurement of the tibial tubercle-trochlear groove distance and trochlea dysplasia. Am J Sports Med. 2015;43(4):873-878.
12. Tensho K, Akaoka Y, Shimodaira H, et al. What components comprise the measurement of the tibial tuberosity-trochlear groove distance in a patellar dislocation population? J Bone Joint Surg Am. 2015;97(17):1441-1448.
13. Camp CL, Heidenreich MJ, Dahm DL, Stuart MJ, Levy BA, Krych AJ. Individualizing the tibial tubercle-trochlear groove distance: patellar instability ratios that predict recurrent instability. Am J Sports Med. 2016;44(2):393-399.
14. Ridley TJ, Hinckel BB, Kruckeberg BM, Agel J, Arendt EA. Anatomical patella instability risk factors on MRI show sensitivity without specificity in patients with patellofemoral instability: a systematic review. JISAKOS. 2016;1(3):141-152.
15. James SL, Bates BT, Osternig LR. Injuries to runners. Am J Sports Med. 1978;6(2):40-50.
16. Powers CM, Souza RB, Fulkerson JP. Patellofemoral joint. In: Magee DJ, Zachazewski JE, Quillen WS, eds. Pathology and Intervention in Musculoskeletal Rehabilitation. St. Louis, MO: Saunders Elsevier; 2008:601-636.
17. Khayambashi K, Mohammadkhani Z, Ghaznavi K, Lyle MA, Powers CM. The effects of isolated hip abductor and external rotator muscle strengthening on pain, health status, and strength in females with patellofemoral pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2012;42(1):22-29.
18. Berg EE, Mason SL, Lucas MJ. Patellar height ratios. A comparison of four measurement methods. Am J Sports Med. 1996;24(2):218-221.
19. Senavongse W, Amis AA. The effects of articular, retinacular, or muscular deficiencies on patellofemoral joint stability: a biomechanical study in vitro. J Bone Joint Surg Br. 2005;87(4):577-582.
20. Merchant AC, Mercer RL, Jacobsen RH, Cool CR. Roentgenographic analysis of patellofemoral congruence. J Bone Joint Surg Am. 1974;56(7):1391-1396.
21. Dejour H, Neyret P, Walch G. Factors in patellar instability. In: Aichroth PM, Cannon WD Jr, Patel DV, eds. Knee Surgery: Current Practice. London, England: Martin Dunitz; 1992.
22. Davies AP, Costa ML, Shepstone L, Glasgow MM, Donell S. The sulcus angle and malalignment of the extensor mechanism of the knee. J Bone Joint Surg Br. 2000;82(8):1162-1166.
23. Aglietti P, Insall JN, Cerulli G. Patellar pain and incongruence. I: measurements of incongruence. Clin Orthop Relat Res. 1983;(176):217-224.
24. Ficat P, Ficat C, Bailieaux A. External hypertension syndrome of the patella. Its significance in the recognition of arthrosis [in French]. Rev Chir Orthop Reparatrice Appar Mot. 1975;61(1):39-59.
25. Ficat P, Hungerford DS. Disorders of the Patellofemoral Joint. Baltimore, MD: Williams & Wilkins; 1977.
26. Merchant AC. The lateral compression syndrome. In: Fox JM, Del Pizzo W, eds. The Patellofemoral Joint. New York, NY: McGraw-Hill; 1993:157-175.
27. DeHaven KE, Dolan WA, Mayer PJ. Chondromalacia patellae in athletes. Clinical presentation and conservative management. Am J Sports Med. 1979;7(1):5-11.
28. Merchant AC. Patellofemoral joint disorders. In: Chapman MW, ed. Operative Orthopedics. Vol 3. Philadelphia, PA: Lippincott; 1988:2321-2366.
29. Sanchis-Alfonso V, Merchant AC. Iatrogenic medial patellar instability: an avoidable injury. Arthroscopy. 2015;31(8):1628-1632.
1. Merchant AC. Classification of patellofemoral disorders. Arthroscopy. 1988;4(4):235-240.
2. Merchant AC. Patellofemoral disorders: biomechanics, diagnosis, and nonoperative treatment. In: McGinty JB, Caspari RB, Jackson RW, Poehling GG, eds. Operative Arthroscopy. New York, NY: Raven Press; 1991:261-275.
3. Dye SF. The knee as a biologic transmission with an envelope of function: a theory. Clin Orthop Relat Res. 1996;(325):10-18.
4. Merchant AC. A philosophy of the patellofemoral joint: a logical clinical approach. In: Sanchis-Alfonso V, ed. Anterior Knee Pain and Patellar Instability. 2nd ed. London, England: Springer; 2011:519-530.
5. Jan MH, Lin DH, Lin JJ, Lin CH, Cheng CK, Lin YF. Differences in sonographic characteristics of the vastus medialis obliquus between patients with patellofemoral pain syndrome and healthy adults. Am J Sports Med. 2009;37(9):1743-1749.
6. Pattyn E, Verdonk P, Steyaert A, et al. Vastus medialis obliquus atrophy: does it exist in patellofemoral pain syndrome? Am J Sports Med. 2011;39(7):1450-1455.
7. Brattström H. Shape of the intercondylar groove normally and in recurrent dislocation of the patella. A clinical and x-ray anatomical investigation. Acta Orthop Scand Suppl. 1964;68:1-147.
8. Roux D. Luxation habituelle de la rotule: traitement operatoire. Rev Chir Orthop Reparatrice Appar Mot. 1888;8:682-689.
9. Post WR. Clinical evaluation of patients with patellofemoral disorders. Arthroscopy. 1999;15(8):841-851.
10. Grelsamer RP, Dubey A, Weinstein CH. Men and women have similar Q angles: a clinical and trigonometric evaluation. J Bone Joint Surg Br. 2005;87(11):1498-1501.
11. Skelley N, Friedman M, McGinnis M, Smith C, Hillen T, Matava M. Inter- and intraobserver reliability in the MRI measurement of the tibial tubercle-trochlear groove distance and trochlea dysplasia. Am J Sports Med. 2015;43(4):873-878.
12. Tensho K, Akaoka Y, Shimodaira H, et al. What components comprise the measurement of the tibial tuberosity-trochlear groove distance in a patellar dislocation population? J Bone Joint Surg Am. 2015;97(17):1441-1448.
13. Camp CL, Heidenreich MJ, Dahm DL, Stuart MJ, Levy BA, Krych AJ. Individualizing the tibial tubercle-trochlear groove distance: patellar instability ratios that predict recurrent instability. Am J Sports Med. 2016;44(2):393-399.
14. Ridley TJ, Hinckel BB, Kruckeberg BM, Agel J, Arendt EA. Anatomical patella instability risk factors on MRI show sensitivity without specificity in patients with patellofemoral instability: a systematic review. JISAKOS. 2016;1(3):141-152.
15. James SL, Bates BT, Osternig LR. Injuries to runners. Am J Sports Med. 1978;6(2):40-50.
16. Powers CM, Souza RB, Fulkerson JP. Patellofemoral joint. In: Magee DJ, Zachazewski JE, Quillen WS, eds. Pathology and Intervention in Musculoskeletal Rehabilitation. St. Louis, MO: Saunders Elsevier; 2008:601-636.
17. Khayambashi K, Mohammadkhani Z, Ghaznavi K, Lyle MA, Powers CM. The effects of isolated hip abductor and external rotator muscle strengthening on pain, health status, and strength in females with patellofemoral pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2012;42(1):22-29.
18. Berg EE, Mason SL, Lucas MJ. Patellar height ratios. A comparison of four measurement methods. Am J Sports Med. 1996;24(2):218-221.
19. Senavongse W, Amis AA. The effects of articular, retinacular, or muscular deficiencies on patellofemoral joint stability: a biomechanical study in vitro. J Bone Joint Surg Br. 2005;87(4):577-582.
20. Merchant AC, Mercer RL, Jacobsen RH, Cool CR. Roentgenographic analysis of patellofemoral congruence. J Bone Joint Surg Am. 1974;56(7):1391-1396.
21. Dejour H, Neyret P, Walch G. Factors in patellar instability. In: Aichroth PM, Cannon WD Jr, Patel DV, eds. Knee Surgery: Current Practice. London, England: Martin Dunitz; 1992.
22. Davies AP, Costa ML, Shepstone L, Glasgow MM, Donell S. The sulcus angle and malalignment of the extensor mechanism of the knee. J Bone Joint Surg Br. 2000;82(8):1162-1166.
23. Aglietti P, Insall JN, Cerulli G. Patellar pain and incongruence. I: measurements of incongruence. Clin Orthop Relat Res. 1983;(176):217-224.
24. Ficat P, Ficat C, Bailieaux A. External hypertension syndrome of the patella. Its significance in the recognition of arthrosis [in French]. Rev Chir Orthop Reparatrice Appar Mot. 1975;61(1):39-59.
25. Ficat P, Hungerford DS. Disorders of the Patellofemoral Joint. Baltimore, MD: Williams & Wilkins; 1977.
26. Merchant AC. The lateral compression syndrome. In: Fox JM, Del Pizzo W, eds. The Patellofemoral Joint. New York, NY: McGraw-Hill; 1993:157-175.
27. DeHaven KE, Dolan WA, Mayer PJ. Chondromalacia patellae in athletes. Clinical presentation and conservative management. Am J Sports Med. 1979;7(1):5-11.
28. Merchant AC. Patellofemoral joint disorders. In: Chapman MW, ed. Operative Orthopedics. Vol 3. Philadelphia, PA: Lippincott; 1988:2321-2366.
29. Sanchis-Alfonso V, Merchant AC. Iatrogenic medial patellar instability: an avoidable injury. Arthroscopy. 2015;31(8):1628-1632.
Correct Positioning of the Medial Patellofemoral Ligament: Troubleshooting in the Operating Room
Take-Home Points
- Use fluoroscopy, isometry, or both to double-check the femoral attachment point. Failure to do so can lead to an overtensioned or undertensioned graft caused by anisometric graft placement.
- To minimize the risk of fracture, avoid drilling transverse tunnels across the patella.
- Do not “pre-tension” the medial patellofemoral ligament graft. There should be little or no tension in the graft when the patella is centered in the groove, regardless of the angle of knee flexion.
- The angle of knee flexion during securing of the graft may be important for inaccurate femoral tunnel placement. Before final fixation of the graft, always range the knee fully to make sure full passive motion will be possible once the graft is secured.
- Understanding the anatomy of the MPFL is key before considering reconstructing: That is, fluoroscopy only suggests a “cloud” to begin assessment of the femoral attachment site and is secondary to anatomic references and check of length changes between the attachment point through range of motion. New studies demonstrate the patellar attachment is broad and extends proximally from the historical patellar attachment site to an equal distance along the distal quadriceps.
The medial patellofemoral ligament (MPFL), which is essential in preventing lateral patellar instability, becomes torn in almost 100% of dislocation events.1 Therefore, in cases of failed nonoperative management, this important constraint should be reconstructed. Reconstruction is technically challenging, precision is needed to avoid postoperative complications, and a thorough understanding of the native MPFL anatomy is paramount.
As a thickening of the medial patellar retinaculum, the MPFL connects the medial patella to the medial femur. The femoral insertion has been described a few ways. In a cadaveric study, LaPrade and colleagues2 noted that it inserts 1.9 mm anterior and 3.2 mm distal to the adductor tubercle. Radiographically, the attachment has been described by Schöttle and colleagues3 and Stephen and colleagues.4 These techniques are discussed in more detail later.
The MPFL is a static restraint to lateral patellar translation—it acts only as a checkrein. It functions mainly in 0° to 30° of knee flexion because once the patella engages the trochlear groove, the bony articulation guides the patella during the rest of knee flexion.5 Most authors agree that the native MPFL is mostly isometric, and the re-created ligament should replicate it.6,7 Using cadaveric specimens, Steensen and colleagues6 found that, from 0° to 90° of knee flexion, the distance from the inferior patellar attachment to the superior femoral attachment changed only 1.1 mm.
Biomechanical studies have shown that a MPFL graft with excessive tension predisposes to postoperative abnormal patellofemoral contact pressures, which cause anterior knee pain, loss of knee flexion, and patellofemoral chondrosis.8-10 Furthermore, an overtensioned graft can cause iatrogenic medial patellar subluxation, and an undertensioned graft may still allow for pathologic lateral patellar translation.
Anatomical Bony Insertions
Femoral Insertion
Precise localization of the proper anatomical femoral attachment of the MPFL is a crucial step in reconstruction.11 Small errors in femoral location have resulted in significant loss of graft isometry, increased patellofemoral contact pressures in cadaveric models,4,7 and increased rates of failure after both MPFL repair12 and reconstruction.13 Several methods for confirming proper femoral location during surgery have been described; these methods help obviate the need for large formal dissection of the medial knee.
In a cadaveric study, Schöttle and colleagues3 described a reproducible radiographic point that precisely identifies the appropriate femoral location for MPFL graft placement. The point is located on a standard true lateral radiograph of the distal femur. First, a line is drawn extending the posterior cortex of the femur distally. Next, 2 lines are drawn perpendicular to the first: one intersecting the posterior point of the Blumensaat line, the other intersecting the transition between the posterior femoral condyle and the posterior femoral cortex3 (Figure 1).
Another radiographic method for intraoperatively identifying the anatomical MPFL femoral attachment was described by Stephen and colleagues.4 They used a cadaveric model to confirm radiographic findings and found that the femoral attachment point, taking the anterior-to-posterior medial femoral condyle distance to be 100%, was identified 40% from the posterior border of the medial femoral condyle, 50% from the distal border, and 60% from the anterior border. This simple “40%–50%–60%” normalizing rule for radiographically defining the femoral attachment point is another helpful intraoperative adjunct for templating the appropriate location for graft placement, but calculation in a sterile operative environment can be difficult.
Both of these techniques depend on a perfect lateral radiograph of the knee, as even minor variations in a radiograph can have a dramatic effect on the appearance of the starting point.
Palpation of bony landmarks is another method for preliminarily identifying the appropriate location for femoral pin placement. If done properly, palpation helps obviate the need for corrections when confirming location using isometry or radiography. The center of the femoral attachment of the MPFL can be located in a groove midway between the medial epicondyle and the adductor tubercle.4 Fujino and colleagues15 conducted a cadaveric study of 31 knees in an effort to relate osseous landmarks with the femoral attachment of the MPFL. In all knees, the adductor tubercle was a reliable osseous landmark. The anatomical MPFL attachment was 10.6 mm distal to the apex of the adductor tubercle and was consistent between knees.
Although all these options offer the best available and most reproducible methods for establishing an anatomical femoral graft insertion site, it is important to note that they are based on cadaveric specimens without recurrent patellar instability. Most knees with chronic patellar instability have associated anatomical abnormalities that are not present in nondysplastic cadaveric specimens, which may alter the relationship of osseous landmarks such as the medial epicondyle and adductor tubercle.16 In a recent study of 30 patients with chronic lateral patellar instability, Sanchis-Alfonso and colleagues16 used 3-dimensional computed tomography with these radiographic landmarks and simulated femoral graft attachment sites. They found that the methods of Schöttle and colleagues3 and Stephen and colleagues4 did not provide precise anatomical femoral placement. Ziegler and colleagues14 correlated the anatomical femoral location of the MPFL with the Schöttle point and found the radiographic site to be 4 mm, on average, off the anatomical location. The location of an appropriate anatomical femoral attachment should be confirmed using multiple methods, including palpation of known osseous landmarks, intraoperative fluoroscopy, and, most important, assessment of graft isometry through full range of motion (ROM).
Patellar Insertion
The patellar attachment of the MPFL has received considerably less attention than the femoral attachment.11 Anatomical studies have shown that the MPFL inserts on the superomedial half to third of the patella, in addition to a portion inserting on the undersurface of the vastus medialis.17
Troubleshooting
It is essential to check graft tension through full knee ROM and observe how the graft behaves in order to prevent iatrogenic complications11 (Figures 6A, 6B).
If the graft is secured in high degrees of knee flexion, and the femoral location is not anatomical, a different phenomenon occurs when the knee is brought back into extension. For proximal femoral tunnels, the graft loosens in knee extension and may lead to continued lateral patellar instability. On the other hand, a distal femoral tunnel may result in iatrogenic medial patellar subluxation as the graft becomes too tight in extension.
Correct Amount of Graft Tension
Overtightening the MPFL during fixation is an easy but avoidable mistake. Unlike the anterior cruciate ligament, the MPFL should not be secured while applying maximum tension. Stephen and colleagues7 and Beck and colleagues8 found that tension of only 2 N (~0.5 lb) is needed to accurately re-create the biomechanics of the native graft.
The amount of tension may inadvertently be increased by an interference screw, which tends to pull the graft into the femoral tunnel during insertion. Attention should be given to watching and palpating the graft as the screw is inserted, especially during the last few turns. Turning the screw half a turn backwards after full insertion can release this increased tension and help avoid overtensioning.
Correct Amount of Knee Flexion
This is probably the least studied aspect of MPFL reconstruction. Recommendations range from 0° to 90° of knee flexion during fixation.7,25-30 Most recommendations are surgeon preference, or are based on a sound rationale that lacks supporting research. Tensioning in full extension has been advocated for assessing for the appropriate amount of lateral patellar translation.27 Authors who endorse deeper knee flexion (60°-90°) think that, because the patella engages a deeper trochlear groove in increased flexion, the bony articulation can be used to establish graft length.30,31
Our cadaveric study showed that lower degrees of knee flexion are safest for minimizing the effect of a malpositioned femoral tunnel.26 If femoral tunnel location is not exactly anatomical, any errors are magnified (with even worse graft mechanics) the deeper in flexion the graft is fixed. Once the patella engages the trochlear groove, at about 30° of knee flexion, this can assist in establishing correct graft length. Therefore, we recommend fixation of the graft in 30° to 45° of knee flexion. Our study results also showed that, if femoral tunnel location is anatomical, the graft will be mostly isometric through knee ROM, and, therefore, amount of initial knee flexion does not affect graft behavior.
Regardless of knee flexion chosen, it is imperative to take the knee through full ROM after fixation to ensure the graft does not excessively loosen or tighten in flexion or extension.
Conclusion
MPFL reconstruction is fraught with errors and technical nuances that may be underappreciated. Accurately locating the femoral insertion is crucial to a biomechanically sound graft, and this location should be scrutinized during surgery with accurate radiographs or bony landmarks and verified with knee ROM. Although there is no clear gold standard for fixation and graft options, the graft should be secured while pulling very little tension (2 N) and with the knee in 30° to 45° of flexion to minimize the effect of any inaccuracies in femoral location. Overall, most patients do well after MPFL reconstruction, and attention to surgical technical detail helps maximize the chances of a satisfactory outcome.
Am J Orthop. 2017;46(2):76-81. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
1. Sallay PI, Poggi J, Speer KP, Garrett WE. Acute dislocation of the patella. A correlative pathoanatomic study. Am J Sports Med. 1996;24(1):52-60.
2. LaPrade RF, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L. The anatomy of the medial part of the knee. J Bone Joint Surg Am. 2007;89(9):2000-2010.
3. Schöttle PB, Schmeling A, Rosenstiel N, Weiler A. Radiographic landmarks for femoral tunnel placement in medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(5):801-804.
4. Stephen JM, Lumpaopong P, Deehan DJ, Kader D, Amis AA. The medial patellofemoral ligament: location of femoral attachment and length change patterns resulting from anatomic and nonanatomic attachments. Am J Sports Med. 2012;40(8):1871-1879.
5. Amis AA, Firer P, Mountney J, Senavongse W, Thomas NP. Anatomy and biomechanics of the medial patellofemoral ligament. Knee. 2003;10(3):215-220.
6. Steensen RN, Dopirak RM, McDonald WG 3rd. The anatomy and isometry of the medial patellofemoral ligament: implications for reconstruction. Am J Sports Med. 2004;32(6):1509-1513.
7. Stephen JM, Kaider D, Lumpaopong P, Deehan DJ, Amis AA. The effect of femoral tunnel position and graft tension on patellar contact mechanics and kinematics after medial patellofemoral ligament reconstruction. Am J Sports Med. 2014;42(2):364-372.
8. Beck P, Brown NA, Greis PE, Burks RT. Patellofemoral contact pressures and lateral patellar translation after medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(9):1557-1563.
9. Bollier M, Fulkerson J, Cosgarea A, Tanaka M. Technical failure of medial patellofemoral ligament reconstruction. Arthroscopy. 2011;27(8):1153-1159.
10. Elias JJ, Cosgarea AJ. Technical errors during medial patellofemoral ligament reconstruction could overload medial patellofemoral cartilage: a computational analysis. Am J Sports Med. 2006;34(9):1478-1485.
11. Sanchis-Alfonso V. Guidelines for medial patellofemoral ligament reconstruction in chronic lateral patellar instability. J Am Acad Orthop Surg. 2014;22(3):175-182.
12. Camp CL, Krych AJ, Dahm DL, Levy BA, Stuart MJ. Medial patellofemoral ligament repair for recurrent patellar dislocation. Am J Sports Med. 2010;38(11):2248-2254.
13. Hopper GP, Leach WJ, Rooney BP, Walker CR, Blyth MJ. Does degree of trochlear dysplasia and position of femoral tunnel influence outcome after medial patellofemoral ligament reconstruction? Am J Sports Med. 2014;42(3):716-722.
14. Ziegler CG, Fulkerson JP, Edgar C. Radiographic reference points are inaccurate with and without a true lateral radiograph: the importance of anatomy in medial patellofemoral ligament reconstruction. Am J Sports Med. 2016;44(1):133-142.
15. Fujino K, Tajima G, Yan J, et al. Morphology of the femoral insertion site of the medial patellofemoral ligament. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):998-1003.
16. Sanchis-Alfonso V, Ramirez-Fuentes C, Montesinos-Berry E, Aparisi-Rodriguez F, Martí-Bonmatí L. Does radiographic location ensure precise anatomic location of the femoral fixation site in medial patellofemoral ligament surgery? Knee Surg Sports Traumatol Arthrosc. 2016;24(9):2838-2844.
17. Smirk C, Morris H. The anatomy and reconstruction of the medial patellofemoral ligament. Knee. 2003;10(3):221-227.
18. Tateishi T, Tsuchiya M, Motosugi N, et al. Graft length change and radiographic assessment of femoral drill hole position for medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2011;19(3):400-407.
19. Mariani PP, Liguori L, Cerullo G, Iannella G, Floris L. Arthroscopic patellar reinsertion of the MPFL in acute patellar dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(4):628-633.
20. Schöttle PB, Hensler D, Imhoff AB. Anatomical double-bundle MPFL reconstruction with an aperture fixation. Knee Surg Sports Traumatol Arthrosc. 2010;18(2):147-151.
21. Siebold R, Chikale S, Sartory N, Hariri N, Feil S, Pässler HH. Hamstring graft fixation in MPFL reconstruction at the patella using a transosseous suture technique. Knee Surg Sports Traumatol Arthrosc. 2010;18(11):1542-1544.
22. Song SY, Kim IS, Chang HG, Shin JH, Kim HJ, Seo YJ. Anatomic medial patellofemoral ligament reconstruction using patellar suture anchor fixation for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2431-2437.
23. Burrus MT, Werner BC, Conte EJ, Diduch DR. Troubleshooting the femoral attachment during medial patellofemoral ligament reconstruction: location, location, location. Orthop J Sports Med. 2015;3(1):2325967115569198.
24. Thaunat M, Erasmus PJ. Management of overtight medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2009;17(5):480-483.
25. Arendt EA, Moeller A, Agel J. Clinical outcomes of medial patellofemoral ligament repair in recurrent (chronic) lateral patella dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(11):1909-1914.
26. Burrus MT, Werner BC, Cancienne JM, Gwathmey FW, Diduch DR. MPFL graft fixation in low degrees of knee flexion minimizes errors made in the femoral location [published online April 16, 2016]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-016-4111-4.
27. Feller JA, Richmond AK, Wasiak J. Medial patellofemoral ligament reconstruction as an isolated or combined procedure for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2470-2476.
28. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
29. Nelitz M, Dreyhaupt J, Reichel H, Woelfle J, Lippacher S. Anatomic reconstruction of the medial patellofemoral ligament in children and adolescents with open growth plates: surgical technique and clinical outcome. Am J Sports Med. 2013;41(1):58-63.
30. Nomura E, Horiuchi Y, Kihara M. A mid-term follow-up of medial patellofemoral ligament reconstruction using an artificial ligament for recurrent patellar dislocation. Knee. 2000;7(4):211-215.
31. Steiner TM, Torga-Spak R, Teitge RA. Medial patellofemoral ligament reconstruction in patients with lateral patellar instability and trochlear dysplasia. Am J Sports Med. 2006;34(8):1254-1261.
Take-Home Points
- Use fluoroscopy, isometry, or both to double-check the femoral attachment point. Failure to do so can lead to an overtensioned or undertensioned graft caused by anisometric graft placement.
- To minimize the risk of fracture, avoid drilling transverse tunnels across the patella.
- Do not “pre-tension” the medial patellofemoral ligament graft. There should be little or no tension in the graft when the patella is centered in the groove, regardless of the angle of knee flexion.
- The angle of knee flexion during securing of the graft may be important for inaccurate femoral tunnel placement. Before final fixation of the graft, always range the knee fully to make sure full passive motion will be possible once the graft is secured.
- Understanding the anatomy of the MPFL is key before considering reconstructing: That is, fluoroscopy only suggests a “cloud” to begin assessment of the femoral attachment site and is secondary to anatomic references and check of length changes between the attachment point through range of motion. New studies demonstrate the patellar attachment is broad and extends proximally from the historical patellar attachment site to an equal distance along the distal quadriceps.
The medial patellofemoral ligament (MPFL), which is essential in preventing lateral patellar instability, becomes torn in almost 100% of dislocation events.1 Therefore, in cases of failed nonoperative management, this important constraint should be reconstructed. Reconstruction is technically challenging, precision is needed to avoid postoperative complications, and a thorough understanding of the native MPFL anatomy is paramount.
As a thickening of the medial patellar retinaculum, the MPFL connects the medial patella to the medial femur. The femoral insertion has been described a few ways. In a cadaveric study, LaPrade and colleagues2 noted that it inserts 1.9 mm anterior and 3.2 mm distal to the adductor tubercle. Radiographically, the attachment has been described by Schöttle and colleagues3 and Stephen and colleagues.4 These techniques are discussed in more detail later.
The MPFL is a static restraint to lateral patellar translation—it acts only as a checkrein. It functions mainly in 0° to 30° of knee flexion because once the patella engages the trochlear groove, the bony articulation guides the patella during the rest of knee flexion.5 Most authors agree that the native MPFL is mostly isometric, and the re-created ligament should replicate it.6,7 Using cadaveric specimens, Steensen and colleagues6 found that, from 0° to 90° of knee flexion, the distance from the inferior patellar attachment to the superior femoral attachment changed only 1.1 mm.
Biomechanical studies have shown that a MPFL graft with excessive tension predisposes to postoperative abnormal patellofemoral contact pressures, which cause anterior knee pain, loss of knee flexion, and patellofemoral chondrosis.8-10 Furthermore, an overtensioned graft can cause iatrogenic medial patellar subluxation, and an undertensioned graft may still allow for pathologic lateral patellar translation.
Anatomical Bony Insertions
Femoral Insertion
Precise localization of the proper anatomical femoral attachment of the MPFL is a crucial step in reconstruction.11 Small errors in femoral location have resulted in significant loss of graft isometry, increased patellofemoral contact pressures in cadaveric models,4,7 and increased rates of failure after both MPFL repair12 and reconstruction.13 Several methods for confirming proper femoral location during surgery have been described; these methods help obviate the need for large formal dissection of the medial knee.
In a cadaveric study, Schöttle and colleagues3 described a reproducible radiographic point that precisely identifies the appropriate femoral location for MPFL graft placement. The point is located on a standard true lateral radiograph of the distal femur. First, a line is drawn extending the posterior cortex of the femur distally. Next, 2 lines are drawn perpendicular to the first: one intersecting the posterior point of the Blumensaat line, the other intersecting the transition between the posterior femoral condyle and the posterior femoral cortex3 (Figure 1).
Another radiographic method for intraoperatively identifying the anatomical MPFL femoral attachment was described by Stephen and colleagues.4 They used a cadaveric model to confirm radiographic findings and found that the femoral attachment point, taking the anterior-to-posterior medial femoral condyle distance to be 100%, was identified 40% from the posterior border of the medial femoral condyle, 50% from the distal border, and 60% from the anterior border. This simple “40%–50%–60%” normalizing rule for radiographically defining the femoral attachment point is another helpful intraoperative adjunct for templating the appropriate location for graft placement, but calculation in a sterile operative environment can be difficult.
Both of these techniques depend on a perfect lateral radiograph of the knee, as even minor variations in a radiograph can have a dramatic effect on the appearance of the starting point.
Palpation of bony landmarks is another method for preliminarily identifying the appropriate location for femoral pin placement. If done properly, palpation helps obviate the need for corrections when confirming location using isometry or radiography. The center of the femoral attachment of the MPFL can be located in a groove midway between the medial epicondyle and the adductor tubercle.4 Fujino and colleagues15 conducted a cadaveric study of 31 knees in an effort to relate osseous landmarks with the femoral attachment of the MPFL. In all knees, the adductor tubercle was a reliable osseous landmark. The anatomical MPFL attachment was 10.6 mm distal to the apex of the adductor tubercle and was consistent between knees.
Although all these options offer the best available and most reproducible methods for establishing an anatomical femoral graft insertion site, it is important to note that they are based on cadaveric specimens without recurrent patellar instability. Most knees with chronic patellar instability have associated anatomical abnormalities that are not present in nondysplastic cadaveric specimens, which may alter the relationship of osseous landmarks such as the medial epicondyle and adductor tubercle.16 In a recent study of 30 patients with chronic lateral patellar instability, Sanchis-Alfonso and colleagues16 used 3-dimensional computed tomography with these radiographic landmarks and simulated femoral graft attachment sites. They found that the methods of Schöttle and colleagues3 and Stephen and colleagues4 did not provide precise anatomical femoral placement. Ziegler and colleagues14 correlated the anatomical femoral location of the MPFL with the Schöttle point and found the radiographic site to be 4 mm, on average, off the anatomical location. The location of an appropriate anatomical femoral attachment should be confirmed using multiple methods, including palpation of known osseous landmarks, intraoperative fluoroscopy, and, most important, assessment of graft isometry through full range of motion (ROM).
Patellar Insertion
The patellar attachment of the MPFL has received considerably less attention than the femoral attachment.11 Anatomical studies have shown that the MPFL inserts on the superomedial half to third of the patella, in addition to a portion inserting on the undersurface of the vastus medialis.17
Troubleshooting
It is essential to check graft tension through full knee ROM and observe how the graft behaves in order to prevent iatrogenic complications11 (Figures 6A, 6B).
If the graft is secured in high degrees of knee flexion, and the femoral location is not anatomical, a different phenomenon occurs when the knee is brought back into extension. For proximal femoral tunnels, the graft loosens in knee extension and may lead to continued lateral patellar instability. On the other hand, a distal femoral tunnel may result in iatrogenic medial patellar subluxation as the graft becomes too tight in extension.
Correct Amount of Graft Tension
Overtightening the MPFL during fixation is an easy but avoidable mistake. Unlike the anterior cruciate ligament, the MPFL should not be secured while applying maximum tension. Stephen and colleagues7 and Beck and colleagues8 found that tension of only 2 N (~0.5 lb) is needed to accurately re-create the biomechanics of the native graft.
The amount of tension may inadvertently be increased by an interference screw, which tends to pull the graft into the femoral tunnel during insertion. Attention should be given to watching and palpating the graft as the screw is inserted, especially during the last few turns. Turning the screw half a turn backwards after full insertion can release this increased tension and help avoid overtensioning.
Correct Amount of Knee Flexion
This is probably the least studied aspect of MPFL reconstruction. Recommendations range from 0° to 90° of knee flexion during fixation.7,25-30 Most recommendations are surgeon preference, or are based on a sound rationale that lacks supporting research. Tensioning in full extension has been advocated for assessing for the appropriate amount of lateral patellar translation.27 Authors who endorse deeper knee flexion (60°-90°) think that, because the patella engages a deeper trochlear groove in increased flexion, the bony articulation can be used to establish graft length.30,31
Our cadaveric study showed that lower degrees of knee flexion are safest for minimizing the effect of a malpositioned femoral tunnel.26 If femoral tunnel location is not exactly anatomical, any errors are magnified (with even worse graft mechanics) the deeper in flexion the graft is fixed. Once the patella engages the trochlear groove, at about 30° of knee flexion, this can assist in establishing correct graft length. Therefore, we recommend fixation of the graft in 30° to 45° of knee flexion. Our study results also showed that, if femoral tunnel location is anatomical, the graft will be mostly isometric through knee ROM, and, therefore, amount of initial knee flexion does not affect graft behavior.
Regardless of knee flexion chosen, it is imperative to take the knee through full ROM after fixation to ensure the graft does not excessively loosen or tighten in flexion or extension.
Conclusion
MPFL reconstruction is fraught with errors and technical nuances that may be underappreciated. Accurately locating the femoral insertion is crucial to a biomechanically sound graft, and this location should be scrutinized during surgery with accurate radiographs or bony landmarks and verified with knee ROM. Although there is no clear gold standard for fixation and graft options, the graft should be secured while pulling very little tension (2 N) and with the knee in 30° to 45° of flexion to minimize the effect of any inaccuracies in femoral location. Overall, most patients do well after MPFL reconstruction, and attention to surgical technical detail helps maximize the chances of a satisfactory outcome.
Am J Orthop. 2017;46(2):76-81. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Take-Home Points
- Use fluoroscopy, isometry, or both to double-check the femoral attachment point. Failure to do so can lead to an overtensioned or undertensioned graft caused by anisometric graft placement.
- To minimize the risk of fracture, avoid drilling transverse tunnels across the patella.
- Do not “pre-tension” the medial patellofemoral ligament graft. There should be little or no tension in the graft when the patella is centered in the groove, regardless of the angle of knee flexion.
- The angle of knee flexion during securing of the graft may be important for inaccurate femoral tunnel placement. Before final fixation of the graft, always range the knee fully to make sure full passive motion will be possible once the graft is secured.
- Understanding the anatomy of the MPFL is key before considering reconstructing: That is, fluoroscopy only suggests a “cloud” to begin assessment of the femoral attachment site and is secondary to anatomic references and check of length changes between the attachment point through range of motion. New studies demonstrate the patellar attachment is broad and extends proximally from the historical patellar attachment site to an equal distance along the distal quadriceps.
The medial patellofemoral ligament (MPFL), which is essential in preventing lateral patellar instability, becomes torn in almost 100% of dislocation events.1 Therefore, in cases of failed nonoperative management, this important constraint should be reconstructed. Reconstruction is technically challenging, precision is needed to avoid postoperative complications, and a thorough understanding of the native MPFL anatomy is paramount.
As a thickening of the medial patellar retinaculum, the MPFL connects the medial patella to the medial femur. The femoral insertion has been described a few ways. In a cadaveric study, LaPrade and colleagues2 noted that it inserts 1.9 mm anterior and 3.2 mm distal to the adductor tubercle. Radiographically, the attachment has been described by Schöttle and colleagues3 and Stephen and colleagues.4 These techniques are discussed in more detail later.
The MPFL is a static restraint to lateral patellar translation—it acts only as a checkrein. It functions mainly in 0° to 30° of knee flexion because once the patella engages the trochlear groove, the bony articulation guides the patella during the rest of knee flexion.5 Most authors agree that the native MPFL is mostly isometric, and the re-created ligament should replicate it.6,7 Using cadaveric specimens, Steensen and colleagues6 found that, from 0° to 90° of knee flexion, the distance from the inferior patellar attachment to the superior femoral attachment changed only 1.1 mm.
Biomechanical studies have shown that a MPFL graft with excessive tension predisposes to postoperative abnormal patellofemoral contact pressures, which cause anterior knee pain, loss of knee flexion, and patellofemoral chondrosis.8-10 Furthermore, an overtensioned graft can cause iatrogenic medial patellar subluxation, and an undertensioned graft may still allow for pathologic lateral patellar translation.
Anatomical Bony Insertions
Femoral Insertion
Precise localization of the proper anatomical femoral attachment of the MPFL is a crucial step in reconstruction.11 Small errors in femoral location have resulted in significant loss of graft isometry, increased patellofemoral contact pressures in cadaveric models,4,7 and increased rates of failure after both MPFL repair12 and reconstruction.13 Several methods for confirming proper femoral location during surgery have been described; these methods help obviate the need for large formal dissection of the medial knee.
In a cadaveric study, Schöttle and colleagues3 described a reproducible radiographic point that precisely identifies the appropriate femoral location for MPFL graft placement. The point is located on a standard true lateral radiograph of the distal femur. First, a line is drawn extending the posterior cortex of the femur distally. Next, 2 lines are drawn perpendicular to the first: one intersecting the posterior point of the Blumensaat line, the other intersecting the transition between the posterior femoral condyle and the posterior femoral cortex3 (Figure 1).
Another radiographic method for intraoperatively identifying the anatomical MPFL femoral attachment was described by Stephen and colleagues.4 They used a cadaveric model to confirm radiographic findings and found that the femoral attachment point, taking the anterior-to-posterior medial femoral condyle distance to be 100%, was identified 40% from the posterior border of the medial femoral condyle, 50% from the distal border, and 60% from the anterior border. This simple “40%–50%–60%” normalizing rule for radiographically defining the femoral attachment point is another helpful intraoperative adjunct for templating the appropriate location for graft placement, but calculation in a sterile operative environment can be difficult.
Both of these techniques depend on a perfect lateral radiograph of the knee, as even minor variations in a radiograph can have a dramatic effect on the appearance of the starting point.
Palpation of bony landmarks is another method for preliminarily identifying the appropriate location for femoral pin placement. If done properly, palpation helps obviate the need for corrections when confirming location using isometry or radiography. The center of the femoral attachment of the MPFL can be located in a groove midway between the medial epicondyle and the adductor tubercle.4 Fujino and colleagues15 conducted a cadaveric study of 31 knees in an effort to relate osseous landmarks with the femoral attachment of the MPFL. In all knees, the adductor tubercle was a reliable osseous landmark. The anatomical MPFL attachment was 10.6 mm distal to the apex of the adductor tubercle and was consistent between knees.
Although all these options offer the best available and most reproducible methods for establishing an anatomical femoral graft insertion site, it is important to note that they are based on cadaveric specimens without recurrent patellar instability. Most knees with chronic patellar instability have associated anatomical abnormalities that are not present in nondysplastic cadaveric specimens, which may alter the relationship of osseous landmarks such as the medial epicondyle and adductor tubercle.16 In a recent study of 30 patients with chronic lateral patellar instability, Sanchis-Alfonso and colleagues16 used 3-dimensional computed tomography with these radiographic landmarks and simulated femoral graft attachment sites. They found that the methods of Schöttle and colleagues3 and Stephen and colleagues4 did not provide precise anatomical femoral placement. Ziegler and colleagues14 correlated the anatomical femoral location of the MPFL with the Schöttle point and found the radiographic site to be 4 mm, on average, off the anatomical location. The location of an appropriate anatomical femoral attachment should be confirmed using multiple methods, including palpation of known osseous landmarks, intraoperative fluoroscopy, and, most important, assessment of graft isometry through full range of motion (ROM).
Patellar Insertion
The patellar attachment of the MPFL has received considerably less attention than the femoral attachment.11 Anatomical studies have shown that the MPFL inserts on the superomedial half to third of the patella, in addition to a portion inserting on the undersurface of the vastus medialis.17
Troubleshooting
It is essential to check graft tension through full knee ROM and observe how the graft behaves in order to prevent iatrogenic complications11 (Figures 6A, 6B).
If the graft is secured in high degrees of knee flexion, and the femoral location is not anatomical, a different phenomenon occurs when the knee is brought back into extension. For proximal femoral tunnels, the graft loosens in knee extension and may lead to continued lateral patellar instability. On the other hand, a distal femoral tunnel may result in iatrogenic medial patellar subluxation as the graft becomes too tight in extension.
Correct Amount of Graft Tension
Overtightening the MPFL during fixation is an easy but avoidable mistake. Unlike the anterior cruciate ligament, the MPFL should not be secured while applying maximum tension. Stephen and colleagues7 and Beck and colleagues8 found that tension of only 2 N (~0.5 lb) is needed to accurately re-create the biomechanics of the native graft.
The amount of tension may inadvertently be increased by an interference screw, which tends to pull the graft into the femoral tunnel during insertion. Attention should be given to watching and palpating the graft as the screw is inserted, especially during the last few turns. Turning the screw half a turn backwards after full insertion can release this increased tension and help avoid overtensioning.
Correct Amount of Knee Flexion
This is probably the least studied aspect of MPFL reconstruction. Recommendations range from 0° to 90° of knee flexion during fixation.7,25-30 Most recommendations are surgeon preference, or are based on a sound rationale that lacks supporting research. Tensioning in full extension has been advocated for assessing for the appropriate amount of lateral patellar translation.27 Authors who endorse deeper knee flexion (60°-90°) think that, because the patella engages a deeper trochlear groove in increased flexion, the bony articulation can be used to establish graft length.30,31
Our cadaveric study showed that lower degrees of knee flexion are safest for minimizing the effect of a malpositioned femoral tunnel.26 If femoral tunnel location is not exactly anatomical, any errors are magnified (with even worse graft mechanics) the deeper in flexion the graft is fixed. Once the patella engages the trochlear groove, at about 30° of knee flexion, this can assist in establishing correct graft length. Therefore, we recommend fixation of the graft in 30° to 45° of knee flexion. Our study results also showed that, if femoral tunnel location is anatomical, the graft will be mostly isometric through knee ROM, and, therefore, amount of initial knee flexion does not affect graft behavior.
Regardless of knee flexion chosen, it is imperative to take the knee through full ROM after fixation to ensure the graft does not excessively loosen or tighten in flexion or extension.
Conclusion
MPFL reconstruction is fraught with errors and technical nuances that may be underappreciated. Accurately locating the femoral insertion is crucial to a biomechanically sound graft, and this location should be scrutinized during surgery with accurate radiographs or bony landmarks and verified with knee ROM. Although there is no clear gold standard for fixation and graft options, the graft should be secured while pulling very little tension (2 N) and with the knee in 30° to 45° of flexion to minimize the effect of any inaccuracies in femoral location. Overall, most patients do well after MPFL reconstruction, and attention to surgical technical detail helps maximize the chances of a satisfactory outcome.
Am J Orthop. 2017;46(2):76-81. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
1. Sallay PI, Poggi J, Speer KP, Garrett WE. Acute dislocation of the patella. A correlative pathoanatomic study. Am J Sports Med. 1996;24(1):52-60.
2. LaPrade RF, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L. The anatomy of the medial part of the knee. J Bone Joint Surg Am. 2007;89(9):2000-2010.
3. Schöttle PB, Schmeling A, Rosenstiel N, Weiler A. Radiographic landmarks for femoral tunnel placement in medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(5):801-804.
4. Stephen JM, Lumpaopong P, Deehan DJ, Kader D, Amis AA. The medial patellofemoral ligament: location of femoral attachment and length change patterns resulting from anatomic and nonanatomic attachments. Am J Sports Med. 2012;40(8):1871-1879.
5. Amis AA, Firer P, Mountney J, Senavongse W, Thomas NP. Anatomy and biomechanics of the medial patellofemoral ligament. Knee. 2003;10(3):215-220.
6. Steensen RN, Dopirak RM, McDonald WG 3rd. The anatomy and isometry of the medial patellofemoral ligament: implications for reconstruction. Am J Sports Med. 2004;32(6):1509-1513.
7. Stephen JM, Kaider D, Lumpaopong P, Deehan DJ, Amis AA. The effect of femoral tunnel position and graft tension on patellar contact mechanics and kinematics after medial patellofemoral ligament reconstruction. Am J Sports Med. 2014;42(2):364-372.
8. Beck P, Brown NA, Greis PE, Burks RT. Patellofemoral contact pressures and lateral patellar translation after medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(9):1557-1563.
9. Bollier M, Fulkerson J, Cosgarea A, Tanaka M. Technical failure of medial patellofemoral ligament reconstruction. Arthroscopy. 2011;27(8):1153-1159.
10. Elias JJ, Cosgarea AJ. Technical errors during medial patellofemoral ligament reconstruction could overload medial patellofemoral cartilage: a computational analysis. Am J Sports Med. 2006;34(9):1478-1485.
11. Sanchis-Alfonso V. Guidelines for medial patellofemoral ligament reconstruction in chronic lateral patellar instability. J Am Acad Orthop Surg. 2014;22(3):175-182.
12. Camp CL, Krych AJ, Dahm DL, Levy BA, Stuart MJ. Medial patellofemoral ligament repair for recurrent patellar dislocation. Am J Sports Med. 2010;38(11):2248-2254.
13. Hopper GP, Leach WJ, Rooney BP, Walker CR, Blyth MJ. Does degree of trochlear dysplasia and position of femoral tunnel influence outcome after medial patellofemoral ligament reconstruction? Am J Sports Med. 2014;42(3):716-722.
14. Ziegler CG, Fulkerson JP, Edgar C. Radiographic reference points are inaccurate with and without a true lateral radiograph: the importance of anatomy in medial patellofemoral ligament reconstruction. Am J Sports Med. 2016;44(1):133-142.
15. Fujino K, Tajima G, Yan J, et al. Morphology of the femoral insertion site of the medial patellofemoral ligament. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):998-1003.
16. Sanchis-Alfonso V, Ramirez-Fuentes C, Montesinos-Berry E, Aparisi-Rodriguez F, Martí-Bonmatí L. Does radiographic location ensure precise anatomic location of the femoral fixation site in medial patellofemoral ligament surgery? Knee Surg Sports Traumatol Arthrosc. 2016;24(9):2838-2844.
17. Smirk C, Morris H. The anatomy and reconstruction of the medial patellofemoral ligament. Knee. 2003;10(3):221-227.
18. Tateishi T, Tsuchiya M, Motosugi N, et al. Graft length change and radiographic assessment of femoral drill hole position for medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2011;19(3):400-407.
19. Mariani PP, Liguori L, Cerullo G, Iannella G, Floris L. Arthroscopic patellar reinsertion of the MPFL in acute patellar dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(4):628-633.
20. Schöttle PB, Hensler D, Imhoff AB. Anatomical double-bundle MPFL reconstruction with an aperture fixation. Knee Surg Sports Traumatol Arthrosc. 2010;18(2):147-151.
21. Siebold R, Chikale S, Sartory N, Hariri N, Feil S, Pässler HH. Hamstring graft fixation in MPFL reconstruction at the patella using a transosseous suture technique. Knee Surg Sports Traumatol Arthrosc. 2010;18(11):1542-1544.
22. Song SY, Kim IS, Chang HG, Shin JH, Kim HJ, Seo YJ. Anatomic medial patellofemoral ligament reconstruction using patellar suture anchor fixation for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2431-2437.
23. Burrus MT, Werner BC, Conte EJ, Diduch DR. Troubleshooting the femoral attachment during medial patellofemoral ligament reconstruction: location, location, location. Orthop J Sports Med. 2015;3(1):2325967115569198.
24. Thaunat M, Erasmus PJ. Management of overtight medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2009;17(5):480-483.
25. Arendt EA, Moeller A, Agel J. Clinical outcomes of medial patellofemoral ligament repair in recurrent (chronic) lateral patella dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(11):1909-1914.
26. Burrus MT, Werner BC, Cancienne JM, Gwathmey FW, Diduch DR. MPFL graft fixation in low degrees of knee flexion minimizes errors made in the femoral location [published online April 16, 2016]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-016-4111-4.
27. Feller JA, Richmond AK, Wasiak J. Medial patellofemoral ligament reconstruction as an isolated or combined procedure for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2470-2476.
28. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
29. Nelitz M, Dreyhaupt J, Reichel H, Woelfle J, Lippacher S. Anatomic reconstruction of the medial patellofemoral ligament in children and adolescents with open growth plates: surgical technique and clinical outcome. Am J Sports Med. 2013;41(1):58-63.
30. Nomura E, Horiuchi Y, Kihara M. A mid-term follow-up of medial patellofemoral ligament reconstruction using an artificial ligament for recurrent patellar dislocation. Knee. 2000;7(4):211-215.
31. Steiner TM, Torga-Spak R, Teitge RA. Medial patellofemoral ligament reconstruction in patients with lateral patellar instability and trochlear dysplasia. Am J Sports Med. 2006;34(8):1254-1261.
1. Sallay PI, Poggi J, Speer KP, Garrett WE. Acute dislocation of the patella. A correlative pathoanatomic study. Am J Sports Med. 1996;24(1):52-60.
2. LaPrade RF, Engebretsen AH, Ly TV, Johansen S, Wentorf FA, Engebretsen L. The anatomy of the medial part of the knee. J Bone Joint Surg Am. 2007;89(9):2000-2010.
3. Schöttle PB, Schmeling A, Rosenstiel N, Weiler A. Radiographic landmarks for femoral tunnel placement in medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(5):801-804.
4. Stephen JM, Lumpaopong P, Deehan DJ, Kader D, Amis AA. The medial patellofemoral ligament: location of femoral attachment and length change patterns resulting from anatomic and nonanatomic attachments. Am J Sports Med. 2012;40(8):1871-1879.
5. Amis AA, Firer P, Mountney J, Senavongse W, Thomas NP. Anatomy and biomechanics of the medial patellofemoral ligament. Knee. 2003;10(3):215-220.
6. Steensen RN, Dopirak RM, McDonald WG 3rd. The anatomy and isometry of the medial patellofemoral ligament: implications for reconstruction. Am J Sports Med. 2004;32(6):1509-1513.
7. Stephen JM, Kaider D, Lumpaopong P, Deehan DJ, Amis AA. The effect of femoral tunnel position and graft tension on patellar contact mechanics and kinematics after medial patellofemoral ligament reconstruction. Am J Sports Med. 2014;42(2):364-372.
8. Beck P, Brown NA, Greis PE, Burks RT. Patellofemoral contact pressures and lateral patellar translation after medial patellofemoral ligament reconstruction. Am J Sports Med. 2007;35(9):1557-1563.
9. Bollier M, Fulkerson J, Cosgarea A, Tanaka M. Technical failure of medial patellofemoral ligament reconstruction. Arthroscopy. 2011;27(8):1153-1159.
10. Elias JJ, Cosgarea AJ. Technical errors during medial patellofemoral ligament reconstruction could overload medial patellofemoral cartilage: a computational analysis. Am J Sports Med. 2006;34(9):1478-1485.
11. Sanchis-Alfonso V. Guidelines for medial patellofemoral ligament reconstruction in chronic lateral patellar instability. J Am Acad Orthop Surg. 2014;22(3):175-182.
12. Camp CL, Krych AJ, Dahm DL, Levy BA, Stuart MJ. Medial patellofemoral ligament repair for recurrent patellar dislocation. Am J Sports Med. 2010;38(11):2248-2254.
13. Hopper GP, Leach WJ, Rooney BP, Walker CR, Blyth MJ. Does degree of trochlear dysplasia and position of femoral tunnel influence outcome after medial patellofemoral ligament reconstruction? Am J Sports Med. 2014;42(3):716-722.
14. Ziegler CG, Fulkerson JP, Edgar C. Radiographic reference points are inaccurate with and without a true lateral radiograph: the importance of anatomy in medial patellofemoral ligament reconstruction. Am J Sports Med. 2016;44(1):133-142.
15. Fujino K, Tajima G, Yan J, et al. Morphology of the femoral insertion site of the medial patellofemoral ligament. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):998-1003.
16. Sanchis-Alfonso V, Ramirez-Fuentes C, Montesinos-Berry E, Aparisi-Rodriguez F, Martí-Bonmatí L. Does radiographic location ensure precise anatomic location of the femoral fixation site in medial patellofemoral ligament surgery? Knee Surg Sports Traumatol Arthrosc. 2016;24(9):2838-2844.
17. Smirk C, Morris H. The anatomy and reconstruction of the medial patellofemoral ligament. Knee. 2003;10(3):221-227.
18. Tateishi T, Tsuchiya M, Motosugi N, et al. Graft length change and radiographic assessment of femoral drill hole position for medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2011;19(3):400-407.
19. Mariani PP, Liguori L, Cerullo G, Iannella G, Floris L. Arthroscopic patellar reinsertion of the MPFL in acute patellar dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(4):628-633.
20. Schöttle PB, Hensler D, Imhoff AB. Anatomical double-bundle MPFL reconstruction with an aperture fixation. Knee Surg Sports Traumatol Arthrosc. 2010;18(2):147-151.
21. Siebold R, Chikale S, Sartory N, Hariri N, Feil S, Pässler HH. Hamstring graft fixation in MPFL reconstruction at the patella using a transosseous suture technique. Knee Surg Sports Traumatol Arthrosc. 2010;18(11):1542-1544.
22. Song SY, Kim IS, Chang HG, Shin JH, Kim HJ, Seo YJ. Anatomic medial patellofemoral ligament reconstruction using patellar suture anchor fixation for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2431-2437.
23. Burrus MT, Werner BC, Conte EJ, Diduch DR. Troubleshooting the femoral attachment during medial patellofemoral ligament reconstruction: location, location, location. Orthop J Sports Med. 2015;3(1):2325967115569198.
24. Thaunat M, Erasmus PJ. Management of overtight medial patellofemoral ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2009;17(5):480-483.
25. Arendt EA, Moeller A, Agel J. Clinical outcomes of medial patellofemoral ligament repair in recurrent (chronic) lateral patella dislocations. Knee Surg Sports Traumatol Arthrosc. 2011;19(11):1909-1914.
26. Burrus MT, Werner BC, Cancienne JM, Gwathmey FW, Diduch DR. MPFL graft fixation in low degrees of knee flexion minimizes errors made in the femoral location [published online April 16, 2016]. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-016-4111-4.
27. Feller JA, Richmond AK, Wasiak J. Medial patellofemoral ligament reconstruction as an isolated or combined procedure for recurrent patellar instability. Knee Surg Sports Traumatol Arthrosc. 2014;22(10):2470-2476.
28. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
29. Nelitz M, Dreyhaupt J, Reichel H, Woelfle J, Lippacher S. Anatomic reconstruction of the medial patellofemoral ligament in children and adolescents with open growth plates: surgical technique and clinical outcome. Am J Sports Med. 2013;41(1):58-63.
30. Nomura E, Horiuchi Y, Kihara M. A mid-term follow-up of medial patellofemoral ligament reconstruction using an artificial ligament for recurrent patellar dislocation. Knee. 2000;7(4):211-215.
31. Steiner TM, Torga-Spak R, Teitge RA. Medial patellofemoral ligament reconstruction in patients with lateral patellar instability and trochlear dysplasia. Am J Sports Med. 2006;34(8):1254-1261.