User login
Targeting a New Safe Zone: A Step in the Development of Patient-Specific Component Positioning for Total Hip Arthroplasty
Postoperative dislocation remains a common complication of primary total hip arthroplasties (THAs), affecting less than 1% to more than 10% in reported series.1,2 In large datasets for modern implants, the incidence of dislocation is 2% to 4%.3,4 Given that more than 200,000 THAs are performed in the United States each year,5 these low percentages represent a large number of patients. The multiplex patient variables that affect THA stability include age, sex, body mass index (BMI), and comorbid conditions.6-8 Surgical approach, restoration of leg length and femoral offset, femoral head size, and component positioning are also important surgical factors that can increase or decrease the incidence of dislocation.3,8,9 In particular, appropriate acetabular component orientation is crucial; surgeons can control this factor and thereby limit the occurrence of dislocation.10 Furthermore, acetabular malpositioning can increase the risk of liner fractures and accelerate bearing-surface wear.11-14
To minimize the risk of postoperative dislocation, surgeons traditionally have targeted the Lewinnek safe zone, with its mean (SD) inclination of 40° (10°) and mean (SD) anteversion of 15° (10°), for acetabular component orientation.15 However, the applicability of this target zone to preventing hip instability using modern implant designs, components, and surgical techniques remains unknown. Achieving acetabular orientation based on maximizing range of motion (ROM) before impingement may be optimal, with anteversion from 20° to 30° and inclination from 40° to 45°.16,17 Furthermore, mean (SD) native acetabular anteversion ranges from 21.3° (6.2°) for men to 24.6° (6.6°) for women.18 Placing THA acetabular components near the native range for anteversion may best provide impingement-free ROM and thus optimize THA stability,16,19 but this has not been proved in a clinical study.
Early dislocation is typically classified as occurring within 6 months after surgery,9 with almost 80% of dislocations occurring within 3 months after surgery.10 Surgeon-specific factors, such as acetabular component positioning, are thought to have a predominant effect on dislocations in the early postoperative period.10 Computer-assisted surgery (CAS), such as imageless navigation, is more accurate than conventional methods for acetabular component placement,20-23 but the clinical relevance of improving accuracy for acetabular component placement has not been shown with respect to altering patient outcomes.23
We conducted a study in a large single-surgeon patient cohort to determine the incidence of early postoperative dislocation with target anteversion increased to 25°, approximating mean native acetabular anteversion.16,19 In addition, we sought to determine the accuracy of imageless navigation in achieving target acetabular component placement.
Materials and Methods
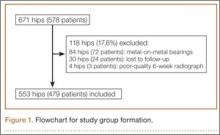
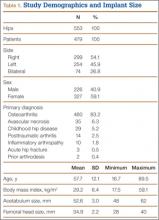
After obtaining institutional review board approval for this retrospective clinical study, we reviewed 671 consecutive cases of primary THA performed by a single surgeon using an imageless CAS system (AchieveCAS; Smith & Nephew, Memphis, Tennessee) between July 2006 and October 2012. THAs were excluded if a metal-on-metal bearing surface was used, if an adequate 6-week postoperative supine anteroposterior (AP) pelvis radiograph was unavailable, or if 6-month clinical follow-up findings were not available (Figure 1). The quality of AP radiographs was deemed poor if they were not centered on the symphysis pubis and if the sacrococcygeal joint was not centered over the symphysis pubis. After exclusion criteria were applied, 553 arthroplasties (479 patients) with a mean (SD) follow-up of 2.4 (1.4) years remained. Perioperative demographic data and component sizes are listed in Table 1.
During surgery, the anterior pelvic plane, defined by the anterior-superior iliac spines and pubic tubercle, was registered with the CAS system with the patient in the supine position. THA was performed with the patient in the lateral decubitus position using a posterolateral technique. For all patients, the surgeon used a hemispherical acetabular component (R3 Acetabular System; Smith & Nephew); bearings that were either metal on highly cross-linked polyethylene (XLPE) or Oxinium (Smith & Nephew) on XLPE; and neutral XLPE acetabular inserts. The goals for acetabular inclination and anteversion were 40° and 25°, respectively, with ±10° each for the target zone. The CAS system was used to adjust target anteversion for sagittal pelvic tilt.24 Uncemented femoral components were used for all patients, and the goal for femoral component anteversion was 15°. Transosseous repair of the posterior capsule and short external rotators was performed after component implantation.25
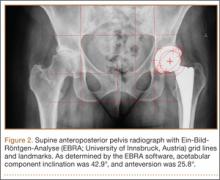
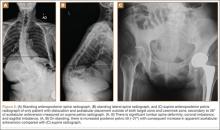
On each 6-week postoperative radiograph, acetabular orientation was measured with Ein-Bild-Röntgen-Analyse (EBRA; University of Innsbruck, Austria) software, which provides a validated method for measuring acetabular inclination and anteversion on supine AP pelvis radiographs.10,26 Pelvic boundaries were delineated with grid lines defining pelvic position. Reference points around the projections of the prosthetic femoral head, the hemispherical cup, and the rim of the cup were marked (Figure 2). EBRA calculated radiographic inclination and anteversion of the acetabular component based on the spatial position of the cup center in relation to the plane of the radiograph and the pelvic position.26
Charts were reviewed to identify patients with early postoperative dislocations, as well as dislocation timing, recurrence, and other characteristics. We defined early dislocation as instability occurring within 6 months after surgery. Revision surgery for instability was also identified.
For the statistical analysis, orientation error was defined as the absolute value of the difference between target orientation (40° inclination, 25° anteversion) and radiographic measurements. Repeated-measures multiple regression with the generalized estimating equations approach was used to identify baseline patient characteristics (age, sex, BMI, primary diagnosis, laterality) associated with component positioning outside of our targeted ranges for inclination and anteversion. Fisher exact tests were used to examine the relationship between dislocation and component placement in either the Lewinnek safe zone or our targeted zone. All tests were 2-sided with a significance level of .05. All analyses were performed with SAS for Windows 9.3 (SAS Institute, Cary, North Carolina).
Results
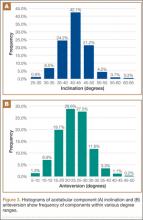
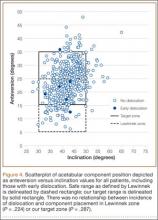
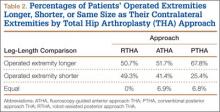
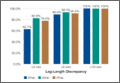
Mean (SD) acetabular inclination was 42.2° (4.9°) (range, 27.6°-65.0°), with a mean (SD) orientation error of 4.2° (3.4°) (Figure 3A). Mean (SD) anteversion was 23.9° (6.5°) (range, 6.2°-48.0°), with a mean (SD) orientation error of 5.2° (4.1°) (Figure 3B). Components were placed outside the Lewinnek safe zone for inclination or anteversion in 46.5% of cases and outside the target zone in 17.7% of cases (Figure 4). Variation in acetabular anteversion alone accounted for 67.3% of target zone outliers (Table 2). Only 0.9% of components were placed outside the target ranges for both inclination and anteversion.
Regression analysis was performed separately for inclination and anteversion to determine the risk factors for placing the acetabular component outside the target orientation ranges. Only higher BMI was associated with malposition with respect to inclination (hazard ratio [HR], 1.059; 95% confidence interval [CI], 1.011-1.111; P = .017). Of obese patients with inclination outside the target range, 90.9% had an inclination angle of more than 50°. Associations between inclination outside the target range and age (P = .769), sex (P = .217), preoperative diagnosis (P > .99), and laterality (P = .106) were statistically insignificant. Only female sex was associated with position of the acetabular component outside the target range for anteversion (HR, 1.871; 95% CI, 1.061-3.299; P = .030). Of female patients with anteversion outside the target range, 70.0% had anteversion of less than 15°. Associations between anteversion outside the target range and age (P = .762), BMI (P = .583), preoperative diagnosis (P > .99), and laterality (P = .235) were statistically insignificant.
Six THAs (1.1%) in 6 patients experienced dislocation within 6 months after surgery (Table 3); mean (SD) time of dislocation was 58.3 (13.8) days after surgery. There was no relationship between dislocation incidence and component placement in the Lewinnek zone (P = .224) or our target zone (P = .287). Of the dislocation cases, 50% involved female patients, and 50% involved right hips. Mean (SD) age of these patients was 53.3 (7.6) years. Mean (SD) BMI was 25.4 (0.9) kg/m2. Osteoarthritis was the primary diagnosis for all patients with early dislocation; 32- or 36-mm femoral heads were used in these cases. Two patients had acetabular components placed outside of our target zone. One patient, who had abnormal pelvic obliquity and sagittal tilt from scoliosis (Figures 5A, 5B), had an acetabular component placed outside both the target zone and the Lewinnek safe zone. Mean (SD) acetabular inclination was 39.8° (3.6°), and mean (SD) anteversion was 21.8° (7.3°) (Figure 5C). Two dislocations resulted from trauma, 1 dislocation was related to hyperlaxity, 1 patient had cerebral palsy, and 1 patient had no evident predisposing risk factors. Three patients (0.54%) had multiple episodes of instability requiring revision during the follow-up period.
Discussion
To our knowledge, this study represents the largest cohort of primary THAs performed with an imageless navigation system. Our results showed that increasing targeted acetabular anteversion to 25° using a posterolateral surgical approach and modern implants resulted in a 1.1% incidence of early dislocation and a 0.54% incidence of recurrent instability requiring reoperation. Of the patients with a dislocation, only 1 did not experience trauma and did not have a risk factor for dislocation. Only 1 patient with a dislocation had acetabular components positioned outside both the target zone and the Lewinnek safe zone. The acetabular component was placed within the target zone in 82.3% of cases in which the imageless navigation system was used. In our cohort, BMI was the only risk factor for placement of the acetabular component outside our target range for inclination, and sex was associated with components outside the target range for anteversion.
Early dislocation after THA is often related to improper implant orientation, inadequate restoration of offset and myofascial tension, and decreased femoral head–neck ratio.8 Although dislocation rates in the literature vary widely,1,2 Medicare data suggest that the rate for the first 6 months after surgery can be as high as 4.21%.3,4 Although use of femoral heads with a diameter of 32 mm or larger may decrease this rate to 2.14%,3 accurate acetabular component orientation helps prevent postoperative dislocation.10 Using an imageless navigation system to target 25° of anteversion and 40° of inclination resulted in an early-dislocation rate about 49% less than the rate in a Medicare population treated with similar, modern implants.3
Callanan and colleagues11 found that freehand techniques were inaccurate for acetabular positioning in up to 50% of cases, and several studies have demonstrated that imageless navigation systems were more accurate than conventional guides.20,21,27-29 Higher BMI has been implicated as a risk factor for acetabular malpositioning in several studies of the accuracy of freehand techniques11 and imageless navigation techniques.23,30 Soft-tissue impediment to the component insertion handle poses a risk of increased inclination and inadequate anteversion, regardless of method used (conventional, CAS). When the acetabular component is placed freehand in obese patients, it is difficult to judge the position of the pelvis on the operating room table. For imageless navigation, a larger amount of adipose tissue over bony landmarks may limit the accuracy of anterior pelvic plane registration.30 Sex typically is not cited as a risk factor for inaccurate acetabular component positioning. We speculate that omitted-variable bias may explain the observed association between female sex and anteversion. For example, changes in postoperative pelvic tilt alter apparent anteversion on plain radiographs,31-34 but preoperative and postoperative sagittal pelvic tilt was not recorded in this study.
The proper position of the acetabular component has been debated.15,16,35,36 Although it is generally agreed that inclination of 40° ± 10° balances ROM, stability, and bearing-surface wear,12,13,15,16 proposed targets for anteversion vary widely, from 0° to 40°.35,36 Patel and colleagues16 formulated computer models based on cadaveric specimens to determine that THA impingement was minimized when the acetabular component was placed to match the native anteversion of the acetabulum.In their study model, 20° of anteversion paralleled native acetabular orientation. Tohtz and colleagues18 reviewed computed tomography scans of 144 female hips and 192 male hips and found that mean (SD) anteversion was 24.6° (6.6°) for women and 21.3° (6.2°) for men. Whether native anatomy is a valid reference for acetabular anteversion is controversial,19 and definitive recommendations for target anteversion cannot be made, as the effect of acetabular anteversion on the wear of various bearing materials is unknown.14 Yet, as with inclination, ideal anteversion is likely a compromise between maximizing impingement-free ROM and minimizing wear.
The present study had several limitations. A single-surgeon patient series was reviewed retrospectively, and there was no control group. We determined the incidence only of early dislocation, and 5.3% of THAs that were not metal-on-metal were either lost to follow-up or had inadequate radiographs. However, of the patients excluded for inadequate radiographs, none had an early dislocation. The effects of our surgical techniques on long-term outcomes, bearing wear, and dislocation are unknown. We were not able to comment on the direction of dislocation for any of the 6 patients with early dislocation, as all dislocations were reduced at facilities other than our hospital. Therefore, we cannot determine whether increasing acetabular anteversion resulted in a larger number of anterior versus posterior dislocations.15
We did not use CAS to place any of the femoral components. Therefore, we could not accurately target combined anteversion, defined as the sum of acetabular and femoral version, which may be an important determinant of THA stability.28 Although restoration of femoral offset and leg length is important in preventing THA dislocation,8 the CAS techniques used did not influence these parameters, and they were not measured.
As an imageless navigation system was used, there were no preoperative axial images, which could have been used to assess native acetabular orientation. This limited our assessment with respect to matching each patient’s natural anteversion. Imageless navigation, which references only the anterior pelvic plane, may not be reliable in patients with excessive sagittal pelvic tilt.37 Furthermore, changes in the functional position of the pelvis from supine to sitting to standing were not accounted for, and changes in sagittal tilt between these positions can be significant.38 Changes in sagittal pelvic tilt affect measurement of acetabular anteversion on plain radiographs, with anterior tilt reducing apparent anteversion and posterior tilt increasing it.32,34 Although postoperative computed tomography is the gold standard for assessing acetabular component orientation, EBRA significantly reduces errors of measurement on plain radiographs.10 Some variability in measured anteversion may be explained by our surgical technique. In particular, if the cup was uncovered anteriorly, additional anteversion was usually accepted during surgery to minimize anterior impingement and limit the risk of iliopsoas tendonitis.16,39
Our study results suggested that increasing target acetabular anteversion to 25° may reduce the incidence of early postoperative instability relative to rates reported in the literature. Despite the higher accuracy of component placement with an imageless navigation system, dislocations occurred in patients with acetabular components positioned in our target zone and in the historical safe zone. These dislocations support the notion that there likely is no absolute safe range for acetabular component positioning, as THA stability depends on many factors. Ideal targets for implant orientation for acetabulum and femur may be patient-specific.16,19 Investigators should prospectively evaluate patient-specific THA component positioning and determine its effect on postoperative dislocation and bearing-surface wear. As specific implant targets are further defined, tools that are more precise and accurate than conventional techniques will be needed to achieve goal component positioning. Our study results confirmed that imageless navigation is an accurate method for achieving acetabular orientation targets.
1. Kwon MS, Kuskowski M, Mulhall KJ, Macaulay W, Brown TE, Saleh KJ. Does surgical approach affect total hip arthroplasty dislocation rates? Clin Orthop. 2006;(447):34-38.
2. Sierra RJ, Raposo JM, Trousdale RT, Cabanela ME. Dislocation of primary THA done through a posterolateral approach in the elderly. Clin Orthop. 2005;(441):262-267.
3. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
4. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
5. Nho SJ, Kymes SM, Callaghan JJ, Felson DT. The burden of hip osteoarthritis in the United States: epidemiologic and economic considerations. J Am Acad Orthop Surg. 2013;21(suppl 1):S1-S6.
6. Sadr Azodi O, Adami J, Lindstrom D, Eriksson KO, Wladis A, Bellocco R. High body mass index is associated with increased risk of implant dislocation following primary total hip replacement: 2,106 patients followed for up to 8 years. Acta Orthop. 2008;79(1):141-147.
7. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty. J Arthroplasty. 2008;23(6):867-872.
8. Morrey BF. Difficult complications after hip joint replacement. Dislocation. Clin Orthop. 1997;(344):179-187.
9. Ho KW, Whitwell GS, Young SK. Reducing the rate of early primary hip dislocation by combining a change in surgical technique and an increase in femoral head diameter to 36 mm. Arch Orthop Trauma Surg. 2012;132(7):1031-1036.
10. Biedermann R, Tonin A, Krismer M, Rachbauer F, Eibl G, Stockl B. Reducing the risk of dislocation after total hip arthroplasty: the effect of orientation of the acetabular component. J Bone Joint Surg Br. 2005;87(6):762-769.
11. Callanan MC, Jarrett B, Bragdon CR, et al. The John Charnley Award: risk factors for cup malpositioning: quality improvement through a joint registry at a tertiary hospital. Clin Orthop. 2011;469(2):319-329.
12. Gallo J, Havranek V, Zapletalova J. Risk factors for accelerated polyethylene wear and osteolysis in ABG I total hip arthroplasty. Int Orthop. 2010;34(1):19-26.
13. Leslie IJ, Williams S, Isaac G, Ingham E, Fisher J. High cup angle and microseparation increase the wear of hip surface replacements. Clin Orthop. 2009;467(9):2259-2265.
14. Esposito CI, Walter WL, Roques A, et al. Wear in alumina-on-alumina ceramic total hip replacements: a retrieval analysis of edge loading. J Bone Joint Surg Br. 2012;94(7):901-907.
15. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
16. Patel AB, Wagle RR, Usrey MM, Thompson MT, Incavo SJ, Noble PC. Guidelines for implant placement to minimize impingement during activities of daily living after total hip arthroplasty. J Arthroplasty. 2010;25(8):1275-1281.e1.
17. Widmer KH, Zurfluh B. Compliant positioning of total hip components for optimal range of motion. J Orthop Res. 2004;22(4):815-821.
18. Tohtz SW, Sassy D, Matziolis G, Preininger B, Perka C, Hasart O. CT evaluation of native acetabular orientation and localization: sex-specific data comparison on 336 hip joints. Technol Health Care. 2010;18(2):129-136.
19. Merle C, Grammatopoulos G, Waldstein W, et al. Comparison of native anatomy with recommended safe component orientation in total hip arthroplasty for primary osteoarthritis. J Bone Joint Surg Am. 2013;95(22):e172.
20. Nogler M, Kessler O, Prassl A, et al. Reduced variability of acetabular cup positioning with use of an imageless navigation system. Clin Orthop. 2004;(426):159-163.
21. Wixson RL, MacDonald MA. Total hip arthroplasty through a minimal posterior approach using imageless computer-assisted hip navigation. J Arthroplasty. 2005;20(7 suppl 3):51-56.
22. Jolles BM, Genoud P, Hoffmeyer P. Computer-assisted cup placement techniques in total hip arthroplasty improve accuracy of placement. Clin Orthop. 2004;(426):174-179.
23. Lass R, Kubista B, Olischar B, Frantal S, Windhager R, Giurea A. Total hip arthroplasty using imageless computer-assisted hip navigation: a prospective randomized study. J Arthroplasty. 2014;29(4):786-791.
24. Babisch JW, Layher F, Amiot LP. The rationale for tilt-adjusted acetabular cup navigation. J Bone Joint Surg Am. 2008;90(2):357-365.
25. Pellicci PM, Bostrom M, Poss R. Posterior approach to total hip replacement using enhanced posterior soft tissue repair. Clin Orthop. 1998;(355):224-228.
26. Krismer M, Bauer R, Tschupik J, Mayrhofer P. EBRA: a method to measure migration of acetabular components. J Biomech. 1995;28(10):1225-1236.
27. Parratte S, Argenson JN. Validation and usefulness of a computer-assisted cup-positioning system in total hip arthroplasty. A prospective, randomized, controlled study. J Bone Joint Surg Am. 2007;89(3):494-499.
28. Dorr LD, Malik A, Wan Z, Long WT, Harris M. Precision and bias of imageless computer navigation and surgeon estimates for acetabular component position. Clin Orthop. 2007;(465):92-99.
29. Najarian BC, Kilgore JE, Markel DC. Evaluation of component positioning in primary total hip arthroplasty using an imageless navigation device compared with traditional methods. J Arthroplasty. 2009;24(1):15-21.
30. Hohmann E, Bryant A, Tetsworth K. Anterior pelvic soft tissue thickness influences acetabular cup positioning with imageless navigation. J Arthroplasty. 2012;27(6):945-952.
31. Nguyen AD, Shultz SJ. Sex differences in clinical measures of lower extremity alignment. J Orthop Sports Phys Ther. 2007;37(7):389-398.
32. Malik A, Wan Z, Jaramaz B, Bowman G, Dorr LD. A validation model for measurement of acetabular component position. J Arthroplasty. 2010;25(5):812-819.
33. Tannast M, Murphy SB, Langlotz F, Anderson SE, Siebenrock KA. Estimation of pelvic tilt on anteroposterior X-rays—a comparison of six parameters. Skeletal Radiol. 2006;35(3):149-155.
34. Parratte S, Pagnano MW, Coleman-Wood K, Kaufman KR, Berry DJ. The 2008 Frank Stinchfield Award: variation in postoperative pelvic tilt may confound the accuracy of hip navigation systems. Clin Orthop. 2009;467(1):43-49.
35. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
36. Kummer FJ, Shah S, Iyer S, DiCesare PE. The effect of acetabular cup orientations on limiting hip rotation. J Arthroplasty. 1999;14(4):509-513.
37. Lin F, Lim D, Wixson RL, Milos S, Hendrix RW, Makhsous M. Limitations of imageless computer-assisted navigation for total hip arthroplasty. J Arthroplasty. 2011;26(4):596-605.
38. Lazennec JY, Riwan A, Gravez F, et al. Hip spine relationships: application to total hip arthroplasty. Hip Int. 2007;17(suppl 5):S91-S104.
39. Trousdale RT, Cabanela ME, Berry DJ. Anterior iliopsoas impingement after total hip arthroplasty. J Arthroplasty. 1995;10(4):546-549.
Postoperative dislocation remains a common complication of primary total hip arthroplasties (THAs), affecting less than 1% to more than 10% in reported series.1,2 In large datasets for modern implants, the incidence of dislocation is 2% to 4%.3,4 Given that more than 200,000 THAs are performed in the United States each year,5 these low percentages represent a large number of patients. The multiplex patient variables that affect THA stability include age, sex, body mass index (BMI), and comorbid conditions.6-8 Surgical approach, restoration of leg length and femoral offset, femoral head size, and component positioning are also important surgical factors that can increase or decrease the incidence of dislocation.3,8,9 In particular, appropriate acetabular component orientation is crucial; surgeons can control this factor and thereby limit the occurrence of dislocation.10 Furthermore, acetabular malpositioning can increase the risk of liner fractures and accelerate bearing-surface wear.11-14
To minimize the risk of postoperative dislocation, surgeons traditionally have targeted the Lewinnek safe zone, with its mean (SD) inclination of 40° (10°) and mean (SD) anteversion of 15° (10°), for acetabular component orientation.15 However, the applicability of this target zone to preventing hip instability using modern implant designs, components, and surgical techniques remains unknown. Achieving acetabular orientation based on maximizing range of motion (ROM) before impingement may be optimal, with anteversion from 20° to 30° and inclination from 40° to 45°.16,17 Furthermore, mean (SD) native acetabular anteversion ranges from 21.3° (6.2°) for men to 24.6° (6.6°) for women.18 Placing THA acetabular components near the native range for anteversion may best provide impingement-free ROM and thus optimize THA stability,16,19 but this has not been proved in a clinical study.
Early dislocation is typically classified as occurring within 6 months after surgery,9 with almost 80% of dislocations occurring within 3 months after surgery.10 Surgeon-specific factors, such as acetabular component positioning, are thought to have a predominant effect on dislocations in the early postoperative period.10 Computer-assisted surgery (CAS), such as imageless navigation, is more accurate than conventional methods for acetabular component placement,20-23 but the clinical relevance of improving accuracy for acetabular component placement has not been shown with respect to altering patient outcomes.23
We conducted a study in a large single-surgeon patient cohort to determine the incidence of early postoperative dislocation with target anteversion increased to 25°, approximating mean native acetabular anteversion.16,19 In addition, we sought to determine the accuracy of imageless navigation in achieving target acetabular component placement.
Materials and Methods
After obtaining institutional review board approval for this retrospective clinical study, we reviewed 671 consecutive cases of primary THA performed by a single surgeon using an imageless CAS system (AchieveCAS; Smith & Nephew, Memphis, Tennessee) between July 2006 and October 2012. THAs were excluded if a metal-on-metal bearing surface was used, if an adequate 6-week postoperative supine anteroposterior (AP) pelvis radiograph was unavailable, or if 6-month clinical follow-up findings were not available (Figure 1). The quality of AP radiographs was deemed poor if they were not centered on the symphysis pubis and if the sacrococcygeal joint was not centered over the symphysis pubis. After exclusion criteria were applied, 553 arthroplasties (479 patients) with a mean (SD) follow-up of 2.4 (1.4) years remained. Perioperative demographic data and component sizes are listed in Table 1.
During surgery, the anterior pelvic plane, defined by the anterior-superior iliac spines and pubic tubercle, was registered with the CAS system with the patient in the supine position. THA was performed with the patient in the lateral decubitus position using a posterolateral technique. For all patients, the surgeon used a hemispherical acetabular component (R3 Acetabular System; Smith & Nephew); bearings that were either metal on highly cross-linked polyethylene (XLPE) or Oxinium (Smith & Nephew) on XLPE; and neutral XLPE acetabular inserts. The goals for acetabular inclination and anteversion were 40° and 25°, respectively, with ±10° each for the target zone. The CAS system was used to adjust target anteversion for sagittal pelvic tilt.24 Uncemented femoral components were used for all patients, and the goal for femoral component anteversion was 15°. Transosseous repair of the posterior capsule and short external rotators was performed after component implantation.25
On each 6-week postoperative radiograph, acetabular orientation was measured with Ein-Bild-Röntgen-Analyse (EBRA; University of Innsbruck, Austria) software, which provides a validated method for measuring acetabular inclination and anteversion on supine AP pelvis radiographs.10,26 Pelvic boundaries were delineated with grid lines defining pelvic position. Reference points around the projections of the prosthetic femoral head, the hemispherical cup, and the rim of the cup were marked (Figure 2). EBRA calculated radiographic inclination and anteversion of the acetabular component based on the spatial position of the cup center in relation to the plane of the radiograph and the pelvic position.26
Charts were reviewed to identify patients with early postoperative dislocations, as well as dislocation timing, recurrence, and other characteristics. We defined early dislocation as instability occurring within 6 months after surgery. Revision surgery for instability was also identified.
For the statistical analysis, orientation error was defined as the absolute value of the difference between target orientation (40° inclination, 25° anteversion) and radiographic measurements. Repeated-measures multiple regression with the generalized estimating equations approach was used to identify baseline patient characteristics (age, sex, BMI, primary diagnosis, laterality) associated with component positioning outside of our targeted ranges for inclination and anteversion. Fisher exact tests were used to examine the relationship between dislocation and component placement in either the Lewinnek safe zone or our targeted zone. All tests were 2-sided with a significance level of .05. All analyses were performed with SAS for Windows 9.3 (SAS Institute, Cary, North Carolina).
Results
Mean (SD) acetabular inclination was 42.2° (4.9°) (range, 27.6°-65.0°), with a mean (SD) orientation error of 4.2° (3.4°) (Figure 3A). Mean (SD) anteversion was 23.9° (6.5°) (range, 6.2°-48.0°), with a mean (SD) orientation error of 5.2° (4.1°) (Figure 3B). Components were placed outside the Lewinnek safe zone for inclination or anteversion in 46.5% of cases and outside the target zone in 17.7% of cases (Figure 4). Variation in acetabular anteversion alone accounted for 67.3% of target zone outliers (Table 2). Only 0.9% of components were placed outside the target ranges for both inclination and anteversion.
Regression analysis was performed separately for inclination and anteversion to determine the risk factors for placing the acetabular component outside the target orientation ranges. Only higher BMI was associated with malposition with respect to inclination (hazard ratio [HR], 1.059; 95% confidence interval [CI], 1.011-1.111; P = .017). Of obese patients with inclination outside the target range, 90.9% had an inclination angle of more than 50°. Associations between inclination outside the target range and age (P = .769), sex (P = .217), preoperative diagnosis (P > .99), and laterality (P = .106) were statistically insignificant. Only female sex was associated with position of the acetabular component outside the target range for anteversion (HR, 1.871; 95% CI, 1.061-3.299; P = .030). Of female patients with anteversion outside the target range, 70.0% had anteversion of less than 15°. Associations between anteversion outside the target range and age (P = .762), BMI (P = .583), preoperative diagnosis (P > .99), and laterality (P = .235) were statistically insignificant.
Six THAs (1.1%) in 6 patients experienced dislocation within 6 months after surgery (Table 3); mean (SD) time of dislocation was 58.3 (13.8) days after surgery. There was no relationship between dislocation incidence and component placement in the Lewinnek zone (P = .224) or our target zone (P = .287). Of the dislocation cases, 50% involved female patients, and 50% involved right hips. Mean (SD) age of these patients was 53.3 (7.6) years. Mean (SD) BMI was 25.4 (0.9) kg/m2. Osteoarthritis was the primary diagnosis for all patients with early dislocation; 32- or 36-mm femoral heads were used in these cases. Two patients had acetabular components placed outside of our target zone. One patient, who had abnormal pelvic obliquity and sagittal tilt from scoliosis (Figures 5A, 5B), had an acetabular component placed outside both the target zone and the Lewinnek safe zone. Mean (SD) acetabular inclination was 39.8° (3.6°), and mean (SD) anteversion was 21.8° (7.3°) (Figure 5C). Two dislocations resulted from trauma, 1 dislocation was related to hyperlaxity, 1 patient had cerebral palsy, and 1 patient had no evident predisposing risk factors. Three patients (0.54%) had multiple episodes of instability requiring revision during the follow-up period.
Discussion
To our knowledge, this study represents the largest cohort of primary THAs performed with an imageless navigation system. Our results showed that increasing targeted acetabular anteversion to 25° using a posterolateral surgical approach and modern implants resulted in a 1.1% incidence of early dislocation and a 0.54% incidence of recurrent instability requiring reoperation. Of the patients with a dislocation, only 1 did not experience trauma and did not have a risk factor for dislocation. Only 1 patient with a dislocation had acetabular components positioned outside both the target zone and the Lewinnek safe zone. The acetabular component was placed within the target zone in 82.3% of cases in which the imageless navigation system was used. In our cohort, BMI was the only risk factor for placement of the acetabular component outside our target range for inclination, and sex was associated with components outside the target range for anteversion.
Early dislocation after THA is often related to improper implant orientation, inadequate restoration of offset and myofascial tension, and decreased femoral head–neck ratio.8 Although dislocation rates in the literature vary widely,1,2 Medicare data suggest that the rate for the first 6 months after surgery can be as high as 4.21%.3,4 Although use of femoral heads with a diameter of 32 mm or larger may decrease this rate to 2.14%,3 accurate acetabular component orientation helps prevent postoperative dislocation.10 Using an imageless navigation system to target 25° of anteversion and 40° of inclination resulted in an early-dislocation rate about 49% less than the rate in a Medicare population treated with similar, modern implants.3
Callanan and colleagues11 found that freehand techniques were inaccurate for acetabular positioning in up to 50% of cases, and several studies have demonstrated that imageless navigation systems were more accurate than conventional guides.20,21,27-29 Higher BMI has been implicated as a risk factor for acetabular malpositioning in several studies of the accuracy of freehand techniques11 and imageless navigation techniques.23,30 Soft-tissue impediment to the component insertion handle poses a risk of increased inclination and inadequate anteversion, regardless of method used (conventional, CAS). When the acetabular component is placed freehand in obese patients, it is difficult to judge the position of the pelvis on the operating room table. For imageless navigation, a larger amount of adipose tissue over bony landmarks may limit the accuracy of anterior pelvic plane registration.30 Sex typically is not cited as a risk factor for inaccurate acetabular component positioning. We speculate that omitted-variable bias may explain the observed association between female sex and anteversion. For example, changes in postoperative pelvic tilt alter apparent anteversion on plain radiographs,31-34 but preoperative and postoperative sagittal pelvic tilt was not recorded in this study.
The proper position of the acetabular component has been debated.15,16,35,36 Although it is generally agreed that inclination of 40° ± 10° balances ROM, stability, and bearing-surface wear,12,13,15,16 proposed targets for anteversion vary widely, from 0° to 40°.35,36 Patel and colleagues16 formulated computer models based on cadaveric specimens to determine that THA impingement was minimized when the acetabular component was placed to match the native anteversion of the acetabulum.In their study model, 20° of anteversion paralleled native acetabular orientation. Tohtz and colleagues18 reviewed computed tomography scans of 144 female hips and 192 male hips and found that mean (SD) anteversion was 24.6° (6.6°) for women and 21.3° (6.2°) for men. Whether native anatomy is a valid reference for acetabular anteversion is controversial,19 and definitive recommendations for target anteversion cannot be made, as the effect of acetabular anteversion on the wear of various bearing materials is unknown.14 Yet, as with inclination, ideal anteversion is likely a compromise between maximizing impingement-free ROM and minimizing wear.
The present study had several limitations. A single-surgeon patient series was reviewed retrospectively, and there was no control group. We determined the incidence only of early dislocation, and 5.3% of THAs that were not metal-on-metal were either lost to follow-up or had inadequate radiographs. However, of the patients excluded for inadequate radiographs, none had an early dislocation. The effects of our surgical techniques on long-term outcomes, bearing wear, and dislocation are unknown. We were not able to comment on the direction of dislocation for any of the 6 patients with early dislocation, as all dislocations were reduced at facilities other than our hospital. Therefore, we cannot determine whether increasing acetabular anteversion resulted in a larger number of anterior versus posterior dislocations.15
We did not use CAS to place any of the femoral components. Therefore, we could not accurately target combined anteversion, defined as the sum of acetabular and femoral version, which may be an important determinant of THA stability.28 Although restoration of femoral offset and leg length is important in preventing THA dislocation,8 the CAS techniques used did not influence these parameters, and they were not measured.
As an imageless navigation system was used, there were no preoperative axial images, which could have been used to assess native acetabular orientation. This limited our assessment with respect to matching each patient’s natural anteversion. Imageless navigation, which references only the anterior pelvic plane, may not be reliable in patients with excessive sagittal pelvic tilt.37 Furthermore, changes in the functional position of the pelvis from supine to sitting to standing were not accounted for, and changes in sagittal tilt between these positions can be significant.38 Changes in sagittal pelvic tilt affect measurement of acetabular anteversion on plain radiographs, with anterior tilt reducing apparent anteversion and posterior tilt increasing it.32,34 Although postoperative computed tomography is the gold standard for assessing acetabular component orientation, EBRA significantly reduces errors of measurement on plain radiographs.10 Some variability in measured anteversion may be explained by our surgical technique. In particular, if the cup was uncovered anteriorly, additional anteversion was usually accepted during surgery to minimize anterior impingement and limit the risk of iliopsoas tendonitis.16,39
Our study results suggested that increasing target acetabular anteversion to 25° may reduce the incidence of early postoperative instability relative to rates reported in the literature. Despite the higher accuracy of component placement with an imageless navigation system, dislocations occurred in patients with acetabular components positioned in our target zone and in the historical safe zone. These dislocations support the notion that there likely is no absolute safe range for acetabular component positioning, as THA stability depends on many factors. Ideal targets for implant orientation for acetabulum and femur may be patient-specific.16,19 Investigators should prospectively evaluate patient-specific THA component positioning and determine its effect on postoperative dislocation and bearing-surface wear. As specific implant targets are further defined, tools that are more precise and accurate than conventional techniques will be needed to achieve goal component positioning. Our study results confirmed that imageless navigation is an accurate method for achieving acetabular orientation targets.
Postoperative dislocation remains a common complication of primary total hip arthroplasties (THAs), affecting less than 1% to more than 10% in reported series.1,2 In large datasets for modern implants, the incidence of dislocation is 2% to 4%.3,4 Given that more than 200,000 THAs are performed in the United States each year,5 these low percentages represent a large number of patients. The multiplex patient variables that affect THA stability include age, sex, body mass index (BMI), and comorbid conditions.6-8 Surgical approach, restoration of leg length and femoral offset, femoral head size, and component positioning are also important surgical factors that can increase or decrease the incidence of dislocation.3,8,9 In particular, appropriate acetabular component orientation is crucial; surgeons can control this factor and thereby limit the occurrence of dislocation.10 Furthermore, acetabular malpositioning can increase the risk of liner fractures and accelerate bearing-surface wear.11-14
To minimize the risk of postoperative dislocation, surgeons traditionally have targeted the Lewinnek safe zone, with its mean (SD) inclination of 40° (10°) and mean (SD) anteversion of 15° (10°), for acetabular component orientation.15 However, the applicability of this target zone to preventing hip instability using modern implant designs, components, and surgical techniques remains unknown. Achieving acetabular orientation based on maximizing range of motion (ROM) before impingement may be optimal, with anteversion from 20° to 30° and inclination from 40° to 45°.16,17 Furthermore, mean (SD) native acetabular anteversion ranges from 21.3° (6.2°) for men to 24.6° (6.6°) for women.18 Placing THA acetabular components near the native range for anteversion may best provide impingement-free ROM and thus optimize THA stability,16,19 but this has not been proved in a clinical study.
Early dislocation is typically classified as occurring within 6 months after surgery,9 with almost 80% of dislocations occurring within 3 months after surgery.10 Surgeon-specific factors, such as acetabular component positioning, are thought to have a predominant effect on dislocations in the early postoperative period.10 Computer-assisted surgery (CAS), such as imageless navigation, is more accurate than conventional methods for acetabular component placement,20-23 but the clinical relevance of improving accuracy for acetabular component placement has not been shown with respect to altering patient outcomes.23
We conducted a study in a large single-surgeon patient cohort to determine the incidence of early postoperative dislocation with target anteversion increased to 25°, approximating mean native acetabular anteversion.16,19 In addition, we sought to determine the accuracy of imageless navigation in achieving target acetabular component placement.
Materials and Methods
After obtaining institutional review board approval for this retrospective clinical study, we reviewed 671 consecutive cases of primary THA performed by a single surgeon using an imageless CAS system (AchieveCAS; Smith & Nephew, Memphis, Tennessee) between July 2006 and October 2012. THAs were excluded if a metal-on-metal bearing surface was used, if an adequate 6-week postoperative supine anteroposterior (AP) pelvis radiograph was unavailable, or if 6-month clinical follow-up findings were not available (Figure 1). The quality of AP radiographs was deemed poor if they were not centered on the symphysis pubis and if the sacrococcygeal joint was not centered over the symphysis pubis. After exclusion criteria were applied, 553 arthroplasties (479 patients) with a mean (SD) follow-up of 2.4 (1.4) years remained. Perioperative demographic data and component sizes are listed in Table 1.
During surgery, the anterior pelvic plane, defined by the anterior-superior iliac spines and pubic tubercle, was registered with the CAS system with the patient in the supine position. THA was performed with the patient in the lateral decubitus position using a posterolateral technique. For all patients, the surgeon used a hemispherical acetabular component (R3 Acetabular System; Smith & Nephew); bearings that were either metal on highly cross-linked polyethylene (XLPE) or Oxinium (Smith & Nephew) on XLPE; and neutral XLPE acetabular inserts. The goals for acetabular inclination and anteversion were 40° and 25°, respectively, with ±10° each for the target zone. The CAS system was used to adjust target anteversion for sagittal pelvic tilt.24 Uncemented femoral components were used for all patients, and the goal for femoral component anteversion was 15°. Transosseous repair of the posterior capsule and short external rotators was performed after component implantation.25
On each 6-week postoperative radiograph, acetabular orientation was measured with Ein-Bild-Röntgen-Analyse (EBRA; University of Innsbruck, Austria) software, which provides a validated method for measuring acetabular inclination and anteversion on supine AP pelvis radiographs.10,26 Pelvic boundaries were delineated with grid lines defining pelvic position. Reference points around the projections of the prosthetic femoral head, the hemispherical cup, and the rim of the cup were marked (Figure 2). EBRA calculated radiographic inclination and anteversion of the acetabular component based on the spatial position of the cup center in relation to the plane of the radiograph and the pelvic position.26
Charts were reviewed to identify patients with early postoperative dislocations, as well as dislocation timing, recurrence, and other characteristics. We defined early dislocation as instability occurring within 6 months after surgery. Revision surgery for instability was also identified.
For the statistical analysis, orientation error was defined as the absolute value of the difference between target orientation (40° inclination, 25° anteversion) and radiographic measurements. Repeated-measures multiple regression with the generalized estimating equations approach was used to identify baseline patient characteristics (age, sex, BMI, primary diagnosis, laterality) associated with component positioning outside of our targeted ranges for inclination and anteversion. Fisher exact tests were used to examine the relationship between dislocation and component placement in either the Lewinnek safe zone or our targeted zone. All tests were 2-sided with a significance level of .05. All analyses were performed with SAS for Windows 9.3 (SAS Institute, Cary, North Carolina).
Results
Mean (SD) acetabular inclination was 42.2° (4.9°) (range, 27.6°-65.0°), with a mean (SD) orientation error of 4.2° (3.4°) (Figure 3A). Mean (SD) anteversion was 23.9° (6.5°) (range, 6.2°-48.0°), with a mean (SD) orientation error of 5.2° (4.1°) (Figure 3B). Components were placed outside the Lewinnek safe zone for inclination or anteversion in 46.5% of cases and outside the target zone in 17.7% of cases (Figure 4). Variation in acetabular anteversion alone accounted for 67.3% of target zone outliers (Table 2). Only 0.9% of components were placed outside the target ranges for both inclination and anteversion.
Regression analysis was performed separately for inclination and anteversion to determine the risk factors for placing the acetabular component outside the target orientation ranges. Only higher BMI was associated with malposition with respect to inclination (hazard ratio [HR], 1.059; 95% confidence interval [CI], 1.011-1.111; P = .017). Of obese patients with inclination outside the target range, 90.9% had an inclination angle of more than 50°. Associations between inclination outside the target range and age (P = .769), sex (P = .217), preoperative diagnosis (P > .99), and laterality (P = .106) were statistically insignificant. Only female sex was associated with position of the acetabular component outside the target range for anteversion (HR, 1.871; 95% CI, 1.061-3.299; P = .030). Of female patients with anteversion outside the target range, 70.0% had anteversion of less than 15°. Associations between anteversion outside the target range and age (P = .762), BMI (P = .583), preoperative diagnosis (P > .99), and laterality (P = .235) were statistically insignificant.
Six THAs (1.1%) in 6 patients experienced dislocation within 6 months after surgery (Table 3); mean (SD) time of dislocation was 58.3 (13.8) days after surgery. There was no relationship between dislocation incidence and component placement in the Lewinnek zone (P = .224) or our target zone (P = .287). Of the dislocation cases, 50% involved female patients, and 50% involved right hips. Mean (SD) age of these patients was 53.3 (7.6) years. Mean (SD) BMI was 25.4 (0.9) kg/m2. Osteoarthritis was the primary diagnosis for all patients with early dislocation; 32- or 36-mm femoral heads were used in these cases. Two patients had acetabular components placed outside of our target zone. One patient, who had abnormal pelvic obliquity and sagittal tilt from scoliosis (Figures 5A, 5B), had an acetabular component placed outside both the target zone and the Lewinnek safe zone. Mean (SD) acetabular inclination was 39.8° (3.6°), and mean (SD) anteversion was 21.8° (7.3°) (Figure 5C). Two dislocations resulted from trauma, 1 dislocation was related to hyperlaxity, 1 patient had cerebral palsy, and 1 patient had no evident predisposing risk factors. Three patients (0.54%) had multiple episodes of instability requiring revision during the follow-up period.
Discussion
To our knowledge, this study represents the largest cohort of primary THAs performed with an imageless navigation system. Our results showed that increasing targeted acetabular anteversion to 25° using a posterolateral surgical approach and modern implants resulted in a 1.1% incidence of early dislocation and a 0.54% incidence of recurrent instability requiring reoperation. Of the patients with a dislocation, only 1 did not experience trauma and did not have a risk factor for dislocation. Only 1 patient with a dislocation had acetabular components positioned outside both the target zone and the Lewinnek safe zone. The acetabular component was placed within the target zone in 82.3% of cases in which the imageless navigation system was used. In our cohort, BMI was the only risk factor for placement of the acetabular component outside our target range for inclination, and sex was associated with components outside the target range for anteversion.
Early dislocation after THA is often related to improper implant orientation, inadequate restoration of offset and myofascial tension, and decreased femoral head–neck ratio.8 Although dislocation rates in the literature vary widely,1,2 Medicare data suggest that the rate for the first 6 months after surgery can be as high as 4.21%.3,4 Although use of femoral heads with a diameter of 32 mm or larger may decrease this rate to 2.14%,3 accurate acetabular component orientation helps prevent postoperative dislocation.10 Using an imageless navigation system to target 25° of anteversion and 40° of inclination resulted in an early-dislocation rate about 49% less than the rate in a Medicare population treated with similar, modern implants.3
Callanan and colleagues11 found that freehand techniques were inaccurate for acetabular positioning in up to 50% of cases, and several studies have demonstrated that imageless navigation systems were more accurate than conventional guides.20,21,27-29 Higher BMI has been implicated as a risk factor for acetabular malpositioning in several studies of the accuracy of freehand techniques11 and imageless navigation techniques.23,30 Soft-tissue impediment to the component insertion handle poses a risk of increased inclination and inadequate anteversion, regardless of method used (conventional, CAS). When the acetabular component is placed freehand in obese patients, it is difficult to judge the position of the pelvis on the operating room table. For imageless navigation, a larger amount of adipose tissue over bony landmarks may limit the accuracy of anterior pelvic plane registration.30 Sex typically is not cited as a risk factor for inaccurate acetabular component positioning. We speculate that omitted-variable bias may explain the observed association between female sex and anteversion. For example, changes in postoperative pelvic tilt alter apparent anteversion on plain radiographs,31-34 but preoperative and postoperative sagittal pelvic tilt was not recorded in this study.
The proper position of the acetabular component has been debated.15,16,35,36 Although it is generally agreed that inclination of 40° ± 10° balances ROM, stability, and bearing-surface wear,12,13,15,16 proposed targets for anteversion vary widely, from 0° to 40°.35,36 Patel and colleagues16 formulated computer models based on cadaveric specimens to determine that THA impingement was minimized when the acetabular component was placed to match the native anteversion of the acetabulum.In their study model, 20° of anteversion paralleled native acetabular orientation. Tohtz and colleagues18 reviewed computed tomography scans of 144 female hips and 192 male hips and found that mean (SD) anteversion was 24.6° (6.6°) for women and 21.3° (6.2°) for men. Whether native anatomy is a valid reference for acetabular anteversion is controversial,19 and definitive recommendations for target anteversion cannot be made, as the effect of acetabular anteversion on the wear of various bearing materials is unknown.14 Yet, as with inclination, ideal anteversion is likely a compromise between maximizing impingement-free ROM and minimizing wear.
The present study had several limitations. A single-surgeon patient series was reviewed retrospectively, and there was no control group. We determined the incidence only of early dislocation, and 5.3% of THAs that were not metal-on-metal were either lost to follow-up or had inadequate radiographs. However, of the patients excluded for inadequate radiographs, none had an early dislocation. The effects of our surgical techniques on long-term outcomes, bearing wear, and dislocation are unknown. We were not able to comment on the direction of dislocation for any of the 6 patients with early dislocation, as all dislocations were reduced at facilities other than our hospital. Therefore, we cannot determine whether increasing acetabular anteversion resulted in a larger number of anterior versus posterior dislocations.15
We did not use CAS to place any of the femoral components. Therefore, we could not accurately target combined anteversion, defined as the sum of acetabular and femoral version, which may be an important determinant of THA stability.28 Although restoration of femoral offset and leg length is important in preventing THA dislocation,8 the CAS techniques used did not influence these parameters, and they were not measured.
As an imageless navigation system was used, there were no preoperative axial images, which could have been used to assess native acetabular orientation. This limited our assessment with respect to matching each patient’s natural anteversion. Imageless navigation, which references only the anterior pelvic plane, may not be reliable in patients with excessive sagittal pelvic tilt.37 Furthermore, changes in the functional position of the pelvis from supine to sitting to standing were not accounted for, and changes in sagittal tilt between these positions can be significant.38 Changes in sagittal pelvic tilt affect measurement of acetabular anteversion on plain radiographs, with anterior tilt reducing apparent anteversion and posterior tilt increasing it.32,34 Although postoperative computed tomography is the gold standard for assessing acetabular component orientation, EBRA significantly reduces errors of measurement on plain radiographs.10 Some variability in measured anteversion may be explained by our surgical technique. In particular, if the cup was uncovered anteriorly, additional anteversion was usually accepted during surgery to minimize anterior impingement and limit the risk of iliopsoas tendonitis.16,39
Our study results suggested that increasing target acetabular anteversion to 25° may reduce the incidence of early postoperative instability relative to rates reported in the literature. Despite the higher accuracy of component placement with an imageless navigation system, dislocations occurred in patients with acetabular components positioned in our target zone and in the historical safe zone. These dislocations support the notion that there likely is no absolute safe range for acetabular component positioning, as THA stability depends on many factors. Ideal targets for implant orientation for acetabulum and femur may be patient-specific.16,19 Investigators should prospectively evaluate patient-specific THA component positioning and determine its effect on postoperative dislocation and bearing-surface wear. As specific implant targets are further defined, tools that are more precise and accurate than conventional techniques will be needed to achieve goal component positioning. Our study results confirmed that imageless navigation is an accurate method for achieving acetabular orientation targets.
1. Kwon MS, Kuskowski M, Mulhall KJ, Macaulay W, Brown TE, Saleh KJ. Does surgical approach affect total hip arthroplasty dislocation rates? Clin Orthop. 2006;(447):34-38.
2. Sierra RJ, Raposo JM, Trousdale RT, Cabanela ME. Dislocation of primary THA done through a posterolateral approach in the elderly. Clin Orthop. 2005;(441):262-267.
3. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
4. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
5. Nho SJ, Kymes SM, Callaghan JJ, Felson DT. The burden of hip osteoarthritis in the United States: epidemiologic and economic considerations. J Am Acad Orthop Surg. 2013;21(suppl 1):S1-S6.
6. Sadr Azodi O, Adami J, Lindstrom D, Eriksson KO, Wladis A, Bellocco R. High body mass index is associated with increased risk of implant dislocation following primary total hip replacement: 2,106 patients followed for up to 8 years. Acta Orthop. 2008;79(1):141-147.
7. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty. J Arthroplasty. 2008;23(6):867-872.
8. Morrey BF. Difficult complications after hip joint replacement. Dislocation. Clin Orthop. 1997;(344):179-187.
9. Ho KW, Whitwell GS, Young SK. Reducing the rate of early primary hip dislocation by combining a change in surgical technique and an increase in femoral head diameter to 36 mm. Arch Orthop Trauma Surg. 2012;132(7):1031-1036.
10. Biedermann R, Tonin A, Krismer M, Rachbauer F, Eibl G, Stockl B. Reducing the risk of dislocation after total hip arthroplasty: the effect of orientation of the acetabular component. J Bone Joint Surg Br. 2005;87(6):762-769.
11. Callanan MC, Jarrett B, Bragdon CR, et al. The John Charnley Award: risk factors for cup malpositioning: quality improvement through a joint registry at a tertiary hospital. Clin Orthop. 2011;469(2):319-329.
12. Gallo J, Havranek V, Zapletalova J. Risk factors for accelerated polyethylene wear and osteolysis in ABG I total hip arthroplasty. Int Orthop. 2010;34(1):19-26.
13. Leslie IJ, Williams S, Isaac G, Ingham E, Fisher J. High cup angle and microseparation increase the wear of hip surface replacements. Clin Orthop. 2009;467(9):2259-2265.
14. Esposito CI, Walter WL, Roques A, et al. Wear in alumina-on-alumina ceramic total hip replacements: a retrieval analysis of edge loading. J Bone Joint Surg Br. 2012;94(7):901-907.
15. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
16. Patel AB, Wagle RR, Usrey MM, Thompson MT, Incavo SJ, Noble PC. Guidelines for implant placement to minimize impingement during activities of daily living after total hip arthroplasty. J Arthroplasty. 2010;25(8):1275-1281.e1.
17. Widmer KH, Zurfluh B. Compliant positioning of total hip components for optimal range of motion. J Orthop Res. 2004;22(4):815-821.
18. Tohtz SW, Sassy D, Matziolis G, Preininger B, Perka C, Hasart O. CT evaluation of native acetabular orientation and localization: sex-specific data comparison on 336 hip joints. Technol Health Care. 2010;18(2):129-136.
19. Merle C, Grammatopoulos G, Waldstein W, et al. Comparison of native anatomy with recommended safe component orientation in total hip arthroplasty for primary osteoarthritis. J Bone Joint Surg Am. 2013;95(22):e172.
20. Nogler M, Kessler O, Prassl A, et al. Reduced variability of acetabular cup positioning with use of an imageless navigation system. Clin Orthop. 2004;(426):159-163.
21. Wixson RL, MacDonald MA. Total hip arthroplasty through a minimal posterior approach using imageless computer-assisted hip navigation. J Arthroplasty. 2005;20(7 suppl 3):51-56.
22. Jolles BM, Genoud P, Hoffmeyer P. Computer-assisted cup placement techniques in total hip arthroplasty improve accuracy of placement. Clin Orthop. 2004;(426):174-179.
23. Lass R, Kubista B, Olischar B, Frantal S, Windhager R, Giurea A. Total hip arthroplasty using imageless computer-assisted hip navigation: a prospective randomized study. J Arthroplasty. 2014;29(4):786-791.
24. Babisch JW, Layher F, Amiot LP. The rationale for tilt-adjusted acetabular cup navigation. J Bone Joint Surg Am. 2008;90(2):357-365.
25. Pellicci PM, Bostrom M, Poss R. Posterior approach to total hip replacement using enhanced posterior soft tissue repair. Clin Orthop. 1998;(355):224-228.
26. Krismer M, Bauer R, Tschupik J, Mayrhofer P. EBRA: a method to measure migration of acetabular components. J Biomech. 1995;28(10):1225-1236.
27. Parratte S, Argenson JN. Validation and usefulness of a computer-assisted cup-positioning system in total hip arthroplasty. A prospective, randomized, controlled study. J Bone Joint Surg Am. 2007;89(3):494-499.
28. Dorr LD, Malik A, Wan Z, Long WT, Harris M. Precision and bias of imageless computer navigation and surgeon estimates for acetabular component position. Clin Orthop. 2007;(465):92-99.
29. Najarian BC, Kilgore JE, Markel DC. Evaluation of component positioning in primary total hip arthroplasty using an imageless navigation device compared with traditional methods. J Arthroplasty. 2009;24(1):15-21.
30. Hohmann E, Bryant A, Tetsworth K. Anterior pelvic soft tissue thickness influences acetabular cup positioning with imageless navigation. J Arthroplasty. 2012;27(6):945-952.
31. Nguyen AD, Shultz SJ. Sex differences in clinical measures of lower extremity alignment. J Orthop Sports Phys Ther. 2007;37(7):389-398.
32. Malik A, Wan Z, Jaramaz B, Bowman G, Dorr LD. A validation model for measurement of acetabular component position. J Arthroplasty. 2010;25(5):812-819.
33. Tannast M, Murphy SB, Langlotz F, Anderson SE, Siebenrock KA. Estimation of pelvic tilt on anteroposterior X-rays—a comparison of six parameters. Skeletal Radiol. 2006;35(3):149-155.
34. Parratte S, Pagnano MW, Coleman-Wood K, Kaufman KR, Berry DJ. The 2008 Frank Stinchfield Award: variation in postoperative pelvic tilt may confound the accuracy of hip navigation systems. Clin Orthop. 2009;467(1):43-49.
35. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
36. Kummer FJ, Shah S, Iyer S, DiCesare PE. The effect of acetabular cup orientations on limiting hip rotation. J Arthroplasty. 1999;14(4):509-513.
37. Lin F, Lim D, Wixson RL, Milos S, Hendrix RW, Makhsous M. Limitations of imageless computer-assisted navigation for total hip arthroplasty. J Arthroplasty. 2011;26(4):596-605.
38. Lazennec JY, Riwan A, Gravez F, et al. Hip spine relationships: application to total hip arthroplasty. Hip Int. 2007;17(suppl 5):S91-S104.
39. Trousdale RT, Cabanela ME, Berry DJ. Anterior iliopsoas impingement after total hip arthroplasty. J Arthroplasty. 1995;10(4):546-549.
1. Kwon MS, Kuskowski M, Mulhall KJ, Macaulay W, Brown TE, Saleh KJ. Does surgical approach affect total hip arthroplasty dislocation rates? Clin Orthop. 2006;(447):34-38.
2. Sierra RJ, Raposo JM, Trousdale RT, Cabanela ME. Dislocation of primary THA done through a posterolateral approach in the elderly. Clin Orthop. 2005;(441):262-267.
3. Malkani AL, Ong KL, Lau E, Kurtz SM, Justice BJ, Manley MT. Early- and late-term dislocation risk after primary hip arthroplasty in the Medicare population. J Arthroplasty. 2010;25(6 suppl):21-25.
4. Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456-2463.
5. Nho SJ, Kymes SM, Callaghan JJ, Felson DT. The burden of hip osteoarthritis in the United States: epidemiologic and economic considerations. J Am Acad Orthop Surg. 2013;21(suppl 1):S1-S6.
6. Sadr Azodi O, Adami J, Lindstrom D, Eriksson KO, Wladis A, Bellocco R. High body mass index is associated with increased risk of implant dislocation following primary total hip replacement: 2,106 patients followed for up to 8 years. Acta Orthop. 2008;79(1):141-147.
7. Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty. J Arthroplasty. 2008;23(6):867-872.
8. Morrey BF. Difficult complications after hip joint replacement. Dislocation. Clin Orthop. 1997;(344):179-187.
9. Ho KW, Whitwell GS, Young SK. Reducing the rate of early primary hip dislocation by combining a change in surgical technique and an increase in femoral head diameter to 36 mm. Arch Orthop Trauma Surg. 2012;132(7):1031-1036.
10. Biedermann R, Tonin A, Krismer M, Rachbauer F, Eibl G, Stockl B. Reducing the risk of dislocation after total hip arthroplasty: the effect of orientation of the acetabular component. J Bone Joint Surg Br. 2005;87(6):762-769.
11. Callanan MC, Jarrett B, Bragdon CR, et al. The John Charnley Award: risk factors for cup malpositioning: quality improvement through a joint registry at a tertiary hospital. Clin Orthop. 2011;469(2):319-329.
12. Gallo J, Havranek V, Zapletalova J. Risk factors for accelerated polyethylene wear and osteolysis in ABG I total hip arthroplasty. Int Orthop. 2010;34(1):19-26.
13. Leslie IJ, Williams S, Isaac G, Ingham E, Fisher J. High cup angle and microseparation increase the wear of hip surface replacements. Clin Orthop. 2009;467(9):2259-2265.
14. Esposito CI, Walter WL, Roques A, et al. Wear in alumina-on-alumina ceramic total hip replacements: a retrieval analysis of edge loading. J Bone Joint Surg Br. 2012;94(7):901-907.
15. Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60(2):217-220.
16. Patel AB, Wagle RR, Usrey MM, Thompson MT, Incavo SJ, Noble PC. Guidelines for implant placement to minimize impingement during activities of daily living after total hip arthroplasty. J Arthroplasty. 2010;25(8):1275-1281.e1.
17. Widmer KH, Zurfluh B. Compliant positioning of total hip components for optimal range of motion. J Orthop Res. 2004;22(4):815-821.
18. Tohtz SW, Sassy D, Matziolis G, Preininger B, Perka C, Hasart O. CT evaluation of native acetabular orientation and localization: sex-specific data comparison on 336 hip joints. Technol Health Care. 2010;18(2):129-136.
19. Merle C, Grammatopoulos G, Waldstein W, et al. Comparison of native anatomy with recommended safe component orientation in total hip arthroplasty for primary osteoarthritis. J Bone Joint Surg Am. 2013;95(22):e172.
20. Nogler M, Kessler O, Prassl A, et al. Reduced variability of acetabular cup positioning with use of an imageless navigation system. Clin Orthop. 2004;(426):159-163.
21. Wixson RL, MacDonald MA. Total hip arthroplasty through a minimal posterior approach using imageless computer-assisted hip navigation. J Arthroplasty. 2005;20(7 suppl 3):51-56.
22. Jolles BM, Genoud P, Hoffmeyer P. Computer-assisted cup placement techniques in total hip arthroplasty improve accuracy of placement. Clin Orthop. 2004;(426):174-179.
23. Lass R, Kubista B, Olischar B, Frantal S, Windhager R, Giurea A. Total hip arthroplasty using imageless computer-assisted hip navigation: a prospective randomized study. J Arthroplasty. 2014;29(4):786-791.
24. Babisch JW, Layher F, Amiot LP. The rationale for tilt-adjusted acetabular cup navigation. J Bone Joint Surg Am. 2008;90(2):357-365.
25. Pellicci PM, Bostrom M, Poss R. Posterior approach to total hip replacement using enhanced posterior soft tissue repair. Clin Orthop. 1998;(355):224-228.
26. Krismer M, Bauer R, Tschupik J, Mayrhofer P. EBRA: a method to measure migration of acetabular components. J Biomech. 1995;28(10):1225-1236.
27. Parratte S, Argenson JN. Validation and usefulness of a computer-assisted cup-positioning system in total hip arthroplasty. A prospective, randomized, controlled study. J Bone Joint Surg Am. 2007;89(3):494-499.
28. Dorr LD, Malik A, Wan Z, Long WT, Harris M. Precision and bias of imageless computer navigation and surgeon estimates for acetabular component position. Clin Orthop. 2007;(465):92-99.
29. Najarian BC, Kilgore JE, Markel DC. Evaluation of component positioning in primary total hip arthroplasty using an imageless navigation device compared with traditional methods. J Arthroplasty. 2009;24(1):15-21.
30. Hohmann E, Bryant A, Tetsworth K. Anterior pelvic soft tissue thickness influences acetabular cup positioning with imageless navigation. J Arthroplasty. 2012;27(6):945-952.
31. Nguyen AD, Shultz SJ. Sex differences in clinical measures of lower extremity alignment. J Orthop Sports Phys Ther. 2007;37(7):389-398.
32. Malik A, Wan Z, Jaramaz B, Bowman G, Dorr LD. A validation model for measurement of acetabular component position. J Arthroplasty. 2010;25(5):812-819.
33. Tannast M, Murphy SB, Langlotz F, Anderson SE, Siebenrock KA. Estimation of pelvic tilt on anteroposterior X-rays—a comparison of six parameters. Skeletal Radiol. 2006;35(3):149-155.
34. Parratte S, Pagnano MW, Coleman-Wood K, Kaufman KR, Berry DJ. The 2008 Frank Stinchfield Award: variation in postoperative pelvic tilt may confound the accuracy of hip navigation systems. Clin Orthop. 2009;467(1):43-49.
35. McCollum DE, Gray WJ. Dislocation after total hip arthroplasty. Causes and prevention. Clin Orthop. 1990;(261):159-170.
36. Kummer FJ, Shah S, Iyer S, DiCesare PE. The effect of acetabular cup orientations on limiting hip rotation. J Arthroplasty. 1999;14(4):509-513.
37. Lin F, Lim D, Wixson RL, Milos S, Hendrix RW, Makhsous M. Limitations of imageless computer-assisted navigation for total hip arthroplasty. J Arthroplasty. 2011;26(4):596-605.
38. Lazennec JY, Riwan A, Gravez F, et al. Hip spine relationships: application to total hip arthroplasty. Hip Int. 2007;17(suppl 5):S91-S104.
39. Trousdale RT, Cabanela ME, Berry DJ. Anterior iliopsoas impingement after total hip arthroplasty. J Arthroplasty. 1995;10(4):546-549.
Recognizing autophonia in patients with anorexia nervosa
Anorexia nervosa can affect a number of systems of the body, including the otolaryngologic presentation of autophonia1,2—a rare hyperperception of an abnormally intense hearing of one’s own voice and respiratory sounds.2 The most common cause of autophonia in patients with anorexia is a patulous (patent) eustachian tube, which can be caused by extreme weight loss.2,3
Significant reduction in the quantity of fat tissue at the location of the eustachian tube can cause patency.3 This creates an abnormal connection between the nasopharynx and tympanic membrane, in which sounds are transmitted directly from the oral cavity to the middle ear, causing autophonia, tinnitus, or sound distortion.4
What are the symptoms?Patients often report hearing their own voice more loudly in the affected ear. This can be distressing, and they might become preoccupied with the sound of their voice—thus affecting quality of life.2,4
The intensity of symptoms varies: from a mild sensation of a clogged ear to extremely bothersome discomfort much like a middle-ear infection.2,4 Autophonia, however, cannot be relieved by conventional therapies for those conditions.2,3
A patulous eustachian tube is difficult to detect and can be misdiagnosed as another condition. Pregnancy, stress, fatigue, radiation therapy, hormonal therapy, and dramatic weight loss also can cause a patulous eustachian tube.2
How is the diagnosis made?The diagnosis of autophonia is clinical and begins with a detailed history. Symptoms often appear within the time frame of rapid weight loss and without evidence of infection or other illness.2,3 The clinical examination is otherwise unremarkable.2,4
Is there treatment?To improve the patient’s comfort and quality of life, intervention is required, best provided by an integrated team of medical specialists. Weight gain, of course, is the treatment goal in anorexia, but this is a complex process often marked by relapse; a detailed discussion of treatment strategies is beyond the scope of this “Pearl.” Symptoms usually diminish as fatty tissue is restored upon successful treatment of anorexia, which closes the abnormal eustachian tube opening.2,3
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Olthoff A, Laskawi R, Kruse E. Successful treatment of autophonia with botulinum toxin: case report. Ann Otol Rhinol Laryngol. 2007;116(8):594-598.
2. Godbole M, Key A. Autophonia in anorexia nervosa. Int J Eat Disord. 2010;43(5):480-482.
3. Karwautz A, Hafferl A, Ungar D, et al. Patulous eustachian tube in a case of adolescent anorexia nervosa. Int J Eat Disord. 1999;25(3):353-355.
4. Dornhoffer JL, Leuwer R, Schwager K, et al. A practical guide to the eustachian tube. New York, NY: Springer; 2014:23-41.
Anorexia nervosa can affect a number of systems of the body, including the otolaryngologic presentation of autophonia1,2—a rare hyperperception of an abnormally intense hearing of one’s own voice and respiratory sounds.2 The most common cause of autophonia in patients with anorexia is a patulous (patent) eustachian tube, which can be caused by extreme weight loss.2,3
Significant reduction in the quantity of fat tissue at the location of the eustachian tube can cause patency.3 This creates an abnormal connection between the nasopharynx and tympanic membrane, in which sounds are transmitted directly from the oral cavity to the middle ear, causing autophonia, tinnitus, or sound distortion.4
What are the symptoms?Patients often report hearing their own voice more loudly in the affected ear. This can be distressing, and they might become preoccupied with the sound of their voice—thus affecting quality of life.2,4
The intensity of symptoms varies: from a mild sensation of a clogged ear to extremely bothersome discomfort much like a middle-ear infection.2,4 Autophonia, however, cannot be relieved by conventional therapies for those conditions.2,3
A patulous eustachian tube is difficult to detect and can be misdiagnosed as another condition. Pregnancy, stress, fatigue, radiation therapy, hormonal therapy, and dramatic weight loss also can cause a patulous eustachian tube.2
How is the diagnosis made?The diagnosis of autophonia is clinical and begins with a detailed history. Symptoms often appear within the time frame of rapid weight loss and without evidence of infection or other illness.2,3 The clinical examination is otherwise unremarkable.2,4
Is there treatment?To improve the patient’s comfort and quality of life, intervention is required, best provided by an integrated team of medical specialists. Weight gain, of course, is the treatment goal in anorexia, but this is a complex process often marked by relapse; a detailed discussion of treatment strategies is beyond the scope of this “Pearl.” Symptoms usually diminish as fatty tissue is restored upon successful treatment of anorexia, which closes the abnormal eustachian tube opening.2,3
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Anorexia nervosa can affect a number of systems of the body, including the otolaryngologic presentation of autophonia1,2—a rare hyperperception of an abnormally intense hearing of one’s own voice and respiratory sounds.2 The most common cause of autophonia in patients with anorexia is a patulous (patent) eustachian tube, which can be caused by extreme weight loss.2,3
Significant reduction in the quantity of fat tissue at the location of the eustachian tube can cause patency.3 This creates an abnormal connection between the nasopharynx and tympanic membrane, in which sounds are transmitted directly from the oral cavity to the middle ear, causing autophonia, tinnitus, or sound distortion.4
What are the symptoms?Patients often report hearing their own voice more loudly in the affected ear. This can be distressing, and they might become preoccupied with the sound of their voice—thus affecting quality of life.2,4
The intensity of symptoms varies: from a mild sensation of a clogged ear to extremely bothersome discomfort much like a middle-ear infection.2,4 Autophonia, however, cannot be relieved by conventional therapies for those conditions.2,3
A patulous eustachian tube is difficult to detect and can be misdiagnosed as another condition. Pregnancy, stress, fatigue, radiation therapy, hormonal therapy, and dramatic weight loss also can cause a patulous eustachian tube.2
How is the diagnosis made?The diagnosis of autophonia is clinical and begins with a detailed history. Symptoms often appear within the time frame of rapid weight loss and without evidence of infection or other illness.2,3 The clinical examination is otherwise unremarkable.2,4
Is there treatment?To improve the patient’s comfort and quality of life, intervention is required, best provided by an integrated team of medical specialists. Weight gain, of course, is the treatment goal in anorexia, but this is a complex process often marked by relapse; a detailed discussion of treatment strategies is beyond the scope of this “Pearl.” Symptoms usually diminish as fatty tissue is restored upon successful treatment of anorexia, which closes the abnormal eustachian tube opening.2,3
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Olthoff A, Laskawi R, Kruse E. Successful treatment of autophonia with botulinum toxin: case report. Ann Otol Rhinol Laryngol. 2007;116(8):594-598.
2. Godbole M, Key A. Autophonia in anorexia nervosa. Int J Eat Disord. 2010;43(5):480-482.
3. Karwautz A, Hafferl A, Ungar D, et al. Patulous eustachian tube in a case of adolescent anorexia nervosa. Int J Eat Disord. 1999;25(3):353-355.
4. Dornhoffer JL, Leuwer R, Schwager K, et al. A practical guide to the eustachian tube. New York, NY: Springer; 2014:23-41.
1. Olthoff A, Laskawi R, Kruse E. Successful treatment of autophonia with botulinum toxin: case report. Ann Otol Rhinol Laryngol. 2007;116(8):594-598.
2. Godbole M, Key A. Autophonia in anorexia nervosa. Int J Eat Disord. 2010;43(5):480-482.
3. Karwautz A, Hafferl A, Ungar D, et al. Patulous eustachian tube in a case of adolescent anorexia nervosa. Int J Eat Disord. 1999;25(3):353-355.
4. Dornhoffer JL, Leuwer R, Schwager K, et al. A practical guide to the eustachian tube. New York, NY: Springer; 2014:23-41.
Leg-Length Discrepancy After Total Hip Arthroplasty: Comparison of Robot-Assisted Posterior, Fluoroscopy-Guided Anterior, and Conventional Posterior Approaches
Total hip arthroplasty (THA) effectively provides adequate pain relief and favorable outcomes in patients with hip osteoarthritis (OA). However, leg-length discrepancy (LLD) is still a significant cause of morbidity,1 including nerve damage,2,3 low back pain,2,4,5 and abnormal gait.2,6,7 Although most of the LLD values reported in the literature fall under the acceptable threshold of 10 mm,8 some patients report dissatisfaction,9 leading to litigation against orthopedic surgeons.2 However, lower extremity lengthening is sometimes needed to achieve adequate hip joint stability and prevent dislocations.2,10
Several methods have been developed to help surgeons estimate the change in leg length during surgery in an attempt to improve clinical outcomes. Use of guide pins as a reference on the pelvis decreased LLD and improved outcomes in some published studies.11,12 Preoperative templating of implant size, cup position, and level of femoral neck cut is very important in helping minimize clinically significant LLD after THA.2,13,14 Computer-assisted THA has also been introduced to try to improve component positioning, restoration of hip center of rotation, and minimizing of LLD.15-17 However, cost and increased operative time have prevented widespread adoption of computer-assisted surgery in THA.
Proponents of different surgical approaches have argued about the superiority of one approach over another. The posterior approach is the gold standard in THA because it is safe, easy to perform, and, if needed, extensile.11 However, exact determination of the intraoperative 3-dimensional (3-D) orientation of the pelvis, and subsequently of LLD, is challenging when the patient lies in the lateral position. The anterior approach has gained in popularity because of its advantages in accelerating postoperative rehabilitation and decreasing hospital length of stay.18 Placing the patient supine is advantageous because it allows leveling of the pelvis and estimation of LLD (by comparing the positions of the lower extremities).19 The anterior approach also allows for radiographic measurements on the operating table.19,20 However, this approach has a high learning curve21 and is not extensile.21 To date, no study has shown superiority of the anterior approach over either the conventional posterior approach or the robot-assisted posterior approach in minimizing LLD after THA.
We conducted a study to compare LLD in patients who underwent THA performed with a robot-assisted posterior approach (RTHA), a fluoroscopy-guided anterior approach (ATHA), or a conventional posterior approach (PTHA). We hypothesized that, compared with PTHA, both RTHA and ATHA would result in reduced LLD.
Materials and Methods
We reviewed all RTHAs, ATHAs, and PTHAs performed by Dr. Domb between September 2008 and December 2012. Study inclusion criteria were a diagnosis of hip OA and the availability of postoperative supine anteroposterior pelvis radiographs. Exclusion criteria were a diagnosis other than hip OA, missing or improper postoperative radiographs (radiographs with rotated or tilted pelvis),22 and radiographs on which at least one of the lesser trochanters was difficult to define. Of the 155 cases included in the study, 67 were RTHAs, 29 were ATHAs, and 59 were PTHAs.
All patients scheduled for THA underwent preoperative planning; plain radiographs were used to determine component size and position, level of neck cut, and amount of leg lengthening or shortening needed. In all RTHA cases, computed tomography of the involved hip was performed before surgery. The MAKO system (MAKO Surgical Corporation, Davie, Florida) was used to develop a patient-specific 3-D model of the pelvis and proximal femur, and this model was used to guide THA execution. The system was then used to detect patient-specific landmarks during surgery, to register the femur and the acetabulum, and to help determine the position of the pelvis and proximal femur during surgery. This system, which uses a haptic robotic arm that guides acetabular reaming and cup placement, provides feedback regarding cup placement, stem version, leg length, and global offset. Pelvic tilt and rotation were accounted for by the MAKO software, and all provided measurements were made on the coronal (functional) plane of the body, as described by Murray.23 ATHA was performed with the patient in the supine position on a Hana table (Mizuho OSI, Union City, California) with fluoroscopic guidance. PTHA was performed in the conventional way, with the patient in the lateral position.
Radiographic measurements of LLD were made with TraumaCad software (Build 2.2.535.0; Voyant Health, Petah-Tikva, Israel). The accuracy of this software has been studied and reported in the literature.24-26 Radiographs were calibrated using the known size of each femoral head as a marker. The reference on the pelvis was the interobturator line (line tangent to inferior border of obturator foramina), and the reference on the femurs was the most superior and medial aspect of each lesser trochanter. Two lines were drawn, each perpendicular to the interobturator line, starting from the previously defined reference point on each lesser trochanter. The difference in length between these 2 lines was recorded as the LLD. Values were recorded relative to the operative extremity. For example, if the operative extremity was longer than the nonoperative extremity, the LLD was given a positive value.
To eliminate bias and increase measurement accuracy, the study had each of 2 observers collect the LLD data twice, 2 months apart. These observers were blinded to each other’s results and to the type of surgery performed. (Neither observer was Dr. Domb, the senior surgeon.) IBM SPSS Statistics software (Version 20; IBM, Armonk, New York) was used for statistical analysis. Each patient’s 4 measurements were averaged into a single number for LLD, and the absolute LLD values were used in all statistical analyses. Means, standard deviations (SDs), and 95% confidence intervals (CIs) were calculated for LLD in each of the 3 groups. Pearson correlation coefficient was used to determine interobserver and intraobserver reliability. One-way analysis of variance (ANOVA) was used to compare group means for age, body mass index (BMI), and LLD. In each group, number of outliers was determined with outliers set at LLDs of more than 3 mm and more than 5 mm. Fischer exact test was used to compare number of outliers in each group. P < .05 was considered statistically significant.
Results
Table 1 lists the demographic data, including age, sex, and BMI, and compares the means. There were strong interobserver and intraobserver correlations for all LLD measurements (r > 0.9; P < .001). Mean (SD) LLD was 2.7 (1.8) mm (95% CI, 2.3-3.2) in the RTHA group, 1.8 (1.6) mm (95% CI, 1.2-2.4) in the ATHA group, and 1.9 (1.6) mm (95% CI, 1.5-2.4) in the PTHA group (P = .01). When LLD of more than 3 mm was set as an outlier, percentage of outliers was 37.3% (RTHA), 17.2% (ATHA), and 22% (PTHA) (P = .06-.78). When LLD of more than 5 mm was set as an outlier, percentage of outliers was 10.4% (RTHA), 6.9% (ATHA), and 8.5% (PTHA) (P = .72 to >.99). No patient in any group had LLD of 10 mm or more (Figure). Table 2 lists percentages of patients’ operated extremities that were longer, shorter, or the same size as their contralateral extremities. Six (9.0%) of the 67 RTHA patients, 4 (13.8%) of the 29 ATHA patients, and 3 (5.1%) of the 59 PTHA patients had a contralateral THA.
Discussion
Our study results showed that RTHA, ATHA, and PTHA were equally effective in minimizing LLD. There was a statistically significant difference in mean LLD among the 3 groups studied. The RTHA group had the largest mean (SD) LLD: 2.7 (1.8) mm. However, statistically significant differences do not always indicate clinical significance.27 Therefore, comparison of the 3 groups’ means is not enough for drawing significant conclusions. The more important point to consider is the number of cases of LLD of 10 mm or more—a discrepancy that would be perceptible to patients and thus become a source of dissatisfaction with painless THA.28 Patients perceive LLD when shortening exceeds 10 mm and lengthening exceeds 6 mm,29 or when LLD is more than 10 mm.16,19,20 Despite significant differences in means, all our cases came in under the 10-mm threshold. When the threshold was decreased to 5 mm (and to 3 mm), there was no statistically significant difference among the groups in the number of cases above the threshold.
LLD remains a source of significant post-THA comorbidity and patient dissatisfaction.1-7,19 Despite surgeons’ efforts to minimize LLD, some patients can detect even a subtle LLD after surgery.1,8,29 Most LLD values reported in the literature fall under the 10-mm threshold.16,19,20 In some cases, however, postoperative LLD is more than 1 cm, enough to prompt litigation against orthopedic surgeons.2 Surgeons have tried to improve LLD with use of multiple techniques, including use of intraoperative measuring devices,30 patient positioning during surgery,20 use of computer-assisted surgery,19 and use of intraoperative fluoroscopy.20
Proponents of computer-assisted THA have argued that this technique improves accuracy in placing the acetabular cup in the safe zone,31 minimizes LLD, and restores femoral offset.32,33 Manzotti and colleagues16 reported on 48 cases of computer-assisted THA matched to 48 cases of conventional THA using the posterior approach. Mean (SD) LLD was 5.06 (2.99) mm in the computer-assisted group and 7.64 (4.36) mm in the conventional group; there was a statistically significant difference in favor of the computer-assisted group (P = .04). However, 5 patients in the computer-assisted group and 13 in the conventional group had LLD of more than 10 mm, and the difference was statistically significant.16 Moreover, the study population was heterogeneous, with 12 patients in both groups having developmental dysplasia as a primary diagnosis.16 All the cases in our study had a diagnosis of OA, and no case had LLD of 10 mm or more.
Several advantages have been proposed for the anterior approach. The supine position (with direct comparison of leg lengths) and the use of fluoroscopy have been described as advantageous in minimizing LLD.20,21 In their study of 494 primary THAs performed with the anterior approach, Matta and colleagues20 reported mean (SD) postoperative LLD of 3 (2) mm (range, 0-26 mm) and concluded that the anterior approach was effective in restoring leg lengths and ensuring proper cup placement while not increasing the dislocation rate. However, they did not compare this approach with others or with computer-assisted THA with respect to LLD.
In another study, Nam and colleagues19 compared LLD after THA performed with 3 different approaches (anterior, conventional posterior, posterior-navigated) and found no statistically significant difference in LLD among the groups. However, LLD was more than 10 mm in 2.2% of anterior cases, 4.4% of conventional posterior cases, and 4.4% of posterior-navigated cases. When 5 mm was used as a cutoff, percentage of patients who were outliers was 31.1% (anterior), 20% (conventional posterior), and 23.3% (navigated-posterior). Our data showed superior results in using 5 mm as a cutoff, with percentage of outliers of 6.9% with ATHA, 8.5% with PTHA, and 10.4% with RTHA. However, Nam and colleagues19 used a larger patient cohort and different techniques for measuring LLD on anteroposterior pelvis radiographs.
The most likely reason that the groups in our study were comparable in terms of LLD accuracy and lack of outliers over the 10-mm cutoff was Dr. Domb’s high accuracy in minimizing LLD using each of the 3 techniques. For ATHA, mean (SD) LLD was 1.8 (1.6) mm (no LLD of ≥10 mm), better than the 3 (2) mm (0.9% with LLD of >10 mm) reported by Matta and colleagues20 and the 3.8 (3.9) mm (2.2% with LLD of >10 mm) reported by Nam and colleagues.19 For PTHA, mean (SD) LLD was 1.9 (1.6) mm (no LLD of ≥10 mm), comparable to some of the best results reported in the literature—for example, the 1 mm (3% with LLD of >10 mm) reported by Woolson and colleagues.34 For RTHA, mean (SD) LLD was 2.7 (1.8) mm (no LLD of ≥10 mm), superior to the 3.9 (2.7) mm (4.4% with LLD of >10 mm) reported by Nam and colleagues19 for posterior-navigated THA and the 5.06 (2.99) mm (10.4% with LLD of >10 mm) reported by Manzotti and colleagues16 for computer-assisted THA.
This study had several notable strengths. All patients had a diagnosis of hip OA and were operated on by a single surgeon. Radiographs were calibrated using the size of the implanted femoral head. Radiographic data were measured using the same technique in all cases and were collected twice by 2 observers (not the senior surgeon) to decrease bias and determine interobserver and intraobserver reliability. In addition, surgeon experience might have played an important role in minimizing LLD regardless of technique and approach used for THA.
Study limitations were different number of cases in each group, lack of matching, lack of clinical follow-up, and lack of long-term assessment of clinical outcomes and complications.
Conclusion
As performed by an experienced surgeon, RTHA, ATHA, and PTHA did not differ in obtaining minimal LLD. All 3 groups had a low frequency of outliers, using thresholds of 3 mm and 5 mm, and no patient in any group had LLD of 10 mm or more. All 3 techniques are effective in achieving accuracy in LLD.
1. Maloney WJ, Keeney JA. Leg length discrepancy after total hip arthroplasty. J Arthroplasty. 2004;19(4 suppl 1):108-110.
2. Clark CR, Huddleston HD, Schoch EP 3rd, Thomas BJ. Leg-length discrepancy after total hip arthroplasty. J Am Acad Orthop Surg. 2006;14(1):38-45.
3. Edwards BN, Tullos HS, Noble PC. Contributory factors and etiology of sciatic nerve palsy in total hip arthroplasty. Clin Orthop. 1987;(218):136-141.
4. Giles LG, Taylor JR. Low-back pain associated with leg length inequality. Spine. 1981;6(5):510-521.
5. Parvizi J, Sharkey PF, Bissett GA, Rothman RH, Hozack WJ. Surgical treatment of limb-length discrepancy following total hip arthroplasty. J Bone Joint Surg Am. 2003;85(12):2310-2317.
6. Edeen J, Sharkey PF, Alexander AH. Clinical significance of leg-length inequality after total hip arthroplasty. Am J Orthop. 1995;24(4):347-351.
7. Gurney B, Mermier C, Robergs R, Gibson A, Rivero D. Effects of limb-length discrepancy on gait economy and lower-extremity muscle activity in older adults. J Bone Joint Surg Am. 2001;83(6):907-915.
8. O’Brien S, Kernohan G, Fitzpatrick C, Hill J, Beverland D. Perception of imposed leg length inequality in normal subjects. Hip Int. 2010;20(4):505-511.
9. Hofmann AA, Skrzynski MC. Leg-length inequality and nerve palsy in total hip arthroplasty: a lawyer awaits! Orthopedics. 2000;23(9):943-944.
10. Miyamoto RG, Kaplan KM, Levine BR, Egol KA, Zuckerman JD. Surgical management of hip fractures: an evidence-based review of the literature. I: femoral neck fractures. J Am Acad Orthop Surg. 2008;16(10):596-607.
11. Ranawat CS, Rao RR, Rodriguez JA, Bhende HS. Correction of limb-length inequality during total hip arthroplasty. J Arthroplasty. 2001;16(6):715-720.
12. McGee HM, Scott JH. A simple method of obtaining equal leg length in total hip arthroplasty. Clin Orthop. 1985;(194):269-270.
13. Della Valle AG, Padgett DE, Salvati EA. Preoperative planning for primary total hip arthroplasty. J Am Acad Orthop Surg. 2005;13(7):455-462.
14. Gonzalez Della Valle A, Slullitel G, Piccaluga F, Salvati EA. The precision and usefulness of preoperative planning for cemented and hybrid primary total hip arthroplasty. J Arthroplasty. 2005;20(1):51-58.
15. Confalonieri N, Manzotti A, Montironi F, Pullen C. Leg length discrepancy, dislocation rate, and offset in total hip replacement using a short modular stem: navigation vs conventional freehand. Orthopedics. 2008;31(10 suppl 1).
16. Manzotti A, Cerveri P, De Momi E, Pullen C, Confalonieri N. Does computer-assisted surgery benefit leg length restoration in total hip replacement? Navigation versus conventional freehand. Int Orthop. 2011;35(1):19-24.
17. Nishio S, Fukunishi S, Fukui T, Fujihara Y, Yoshiya S. Adjustment of leg length using imageless navigation THA software without a femoral tracker. J Orthop Sci. 2011;16(2):171-176.
18. Martin CT, Pugely AJ, Gao Y, Clark CR. A comparison of hospital length of stay and short-term morbidity between the anterior and the posterior approaches to total hip arthroplasty. J Arthroplasty. 2013;28(5):849-854.
19. Nam D, Sculco PK, Abdel MP, Alexiades MM, Figgie MP, Mayman DJ. Leg-length inequalities following THA based on surgical technique. Orthopedics. 2013;36(4):e395-e400.
20. Matta JM, Shahrdar C, Ferguson T. Single-incision anterior approach for total hip arthroplasty on an orthopaedic table. Clin Orthop. 2005;(441):115-124.
21. Yi C, Agudelo JF, Dayton MR, Morgan SJ. Early complications of anterior supine intermuscular total hip arthroplasty. Orthopedics. 2013;36(3):e276-e281.
22. Siebenrock KA, Kalbermatten DF, Ganz R. Effect of pelvic tilt on acetabular retroversion: a study of pelves from cadavers. Clin Orthop. 2003;(407):241-248.
23. Murray DW. The definition and measurement of acetabular orientation. J Bone Joint Surg Br. 1993;75(2):228-232.
24. Kumar PG, Kirmani SJ, Humberg H, Kavarthapu V, Li P. Reproducibility and accuracy of templating uncemented THA with digital radiographic and digital TraumaCad templating software. Orthopedics. 2009;32(11):815.
25. Steinberg EL, Shasha N, Menahem A, Dekel S. Preoperative planning of total hip replacement using the TraumaCad system. Arch Orthop Trauma Surg. 2010;130(12):1429-1432.
26. Westacott DJ, McArthur J, King RJ, Foguet P. Assessment of cup orientation in hip resurfacing: a comparison of TraumaCad and computed tomography. J Orthop Surg Res. 2013;8:8.
27. Copay AG, Subach BR, Glassman SD, Polly DW Jr, Schuler TC. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 2007;7(5):541-546.
28. Abraham WD, Dimon JH 3rd. Leg length discrepancy in total hip arthroplasty. Orthop Clin North Am. 1992;23(2):201-209.
29. Konyves A, Bannister GC. The importance of leg length discrepancy after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(2):155-157.
30. Matsuda K, Nakamura S, Matsushita T. A simple method to minimize limb-length discrepancy after hip arthroplasty. Acta Orthop. 2006;77(3):375-379.
31. Haaker RG, Tiedjen K, Ottersbach A, Rubenthaler F, Stockheim M, Stiehl JB. Comparison of conventional versus computer-navigated acetabular component insertion. J Arthroplasty. 2007;22(2):151-159.
32. Renkawitz T, Schuster T, Herold T, et al. Measuring leg length and offset with an imageless navigation system during total hip arthroplasty: is it really accurate? Int J Med Robot. 2009;5(2):192-197.
33. Nakamura N, Sugano N, Nishii T, Kakimoto A, Miki H. A comparison between robotic-assisted and manual implantation of cementless total hip arthroplasty. Clin Orthop. 2010;468(4):1072-1081.
34. Woolson ST, Hartford JM, Sawyer A. Results of a method of leg-length equalization for patients undergoing primary total hip replacement. J Arthroplasty. 1999;14(2):159-164.
Total hip arthroplasty (THA) effectively provides adequate pain relief and favorable outcomes in patients with hip osteoarthritis (OA). However, leg-length discrepancy (LLD) is still a significant cause of morbidity,1 including nerve damage,2,3 low back pain,2,4,5 and abnormal gait.2,6,7 Although most of the LLD values reported in the literature fall under the acceptable threshold of 10 mm,8 some patients report dissatisfaction,9 leading to litigation against orthopedic surgeons.2 However, lower extremity lengthening is sometimes needed to achieve adequate hip joint stability and prevent dislocations.2,10
Several methods have been developed to help surgeons estimate the change in leg length during surgery in an attempt to improve clinical outcomes. Use of guide pins as a reference on the pelvis decreased LLD and improved outcomes in some published studies.11,12 Preoperative templating of implant size, cup position, and level of femoral neck cut is very important in helping minimize clinically significant LLD after THA.2,13,14 Computer-assisted THA has also been introduced to try to improve component positioning, restoration of hip center of rotation, and minimizing of LLD.15-17 However, cost and increased operative time have prevented widespread adoption of computer-assisted surgery in THA.
Proponents of different surgical approaches have argued about the superiority of one approach over another. The posterior approach is the gold standard in THA because it is safe, easy to perform, and, if needed, extensile.11 However, exact determination of the intraoperative 3-dimensional (3-D) orientation of the pelvis, and subsequently of LLD, is challenging when the patient lies in the lateral position. The anterior approach has gained in popularity because of its advantages in accelerating postoperative rehabilitation and decreasing hospital length of stay.18 Placing the patient supine is advantageous because it allows leveling of the pelvis and estimation of LLD (by comparing the positions of the lower extremities).19 The anterior approach also allows for radiographic measurements on the operating table.19,20 However, this approach has a high learning curve21 and is not extensile.21 To date, no study has shown superiority of the anterior approach over either the conventional posterior approach or the robot-assisted posterior approach in minimizing LLD after THA.
We conducted a study to compare LLD in patients who underwent THA performed with a robot-assisted posterior approach (RTHA), a fluoroscopy-guided anterior approach (ATHA), or a conventional posterior approach (PTHA). We hypothesized that, compared with PTHA, both RTHA and ATHA would result in reduced LLD.
Materials and Methods
We reviewed all RTHAs, ATHAs, and PTHAs performed by Dr. Domb between September 2008 and December 2012. Study inclusion criteria were a diagnosis of hip OA and the availability of postoperative supine anteroposterior pelvis radiographs. Exclusion criteria were a diagnosis other than hip OA, missing or improper postoperative radiographs (radiographs with rotated or tilted pelvis),22 and radiographs on which at least one of the lesser trochanters was difficult to define. Of the 155 cases included in the study, 67 were RTHAs, 29 were ATHAs, and 59 were PTHAs.
All patients scheduled for THA underwent preoperative planning; plain radiographs were used to determine component size and position, level of neck cut, and amount of leg lengthening or shortening needed. In all RTHA cases, computed tomography of the involved hip was performed before surgery. The MAKO system (MAKO Surgical Corporation, Davie, Florida) was used to develop a patient-specific 3-D model of the pelvis and proximal femur, and this model was used to guide THA execution. The system was then used to detect patient-specific landmarks during surgery, to register the femur and the acetabulum, and to help determine the position of the pelvis and proximal femur during surgery. This system, which uses a haptic robotic arm that guides acetabular reaming and cup placement, provides feedback regarding cup placement, stem version, leg length, and global offset. Pelvic tilt and rotation were accounted for by the MAKO software, and all provided measurements were made on the coronal (functional) plane of the body, as described by Murray.23 ATHA was performed with the patient in the supine position on a Hana table (Mizuho OSI, Union City, California) with fluoroscopic guidance. PTHA was performed in the conventional way, with the patient in the lateral position.
Radiographic measurements of LLD were made with TraumaCad software (Build 2.2.535.0; Voyant Health, Petah-Tikva, Israel). The accuracy of this software has been studied and reported in the literature.24-26 Radiographs were calibrated using the known size of each femoral head as a marker. The reference on the pelvis was the interobturator line (line tangent to inferior border of obturator foramina), and the reference on the femurs was the most superior and medial aspect of each lesser trochanter. Two lines were drawn, each perpendicular to the interobturator line, starting from the previously defined reference point on each lesser trochanter. The difference in length between these 2 lines was recorded as the LLD. Values were recorded relative to the operative extremity. For example, if the operative extremity was longer than the nonoperative extremity, the LLD was given a positive value.
To eliminate bias and increase measurement accuracy, the study had each of 2 observers collect the LLD data twice, 2 months apart. These observers were blinded to each other’s results and to the type of surgery performed. (Neither observer was Dr. Domb, the senior surgeon.) IBM SPSS Statistics software (Version 20; IBM, Armonk, New York) was used for statistical analysis. Each patient’s 4 measurements were averaged into a single number for LLD, and the absolute LLD values were used in all statistical analyses. Means, standard deviations (SDs), and 95% confidence intervals (CIs) were calculated for LLD in each of the 3 groups. Pearson correlation coefficient was used to determine interobserver and intraobserver reliability. One-way analysis of variance (ANOVA) was used to compare group means for age, body mass index (BMI), and LLD. In each group, number of outliers was determined with outliers set at LLDs of more than 3 mm and more than 5 mm. Fischer exact test was used to compare number of outliers in each group. P < .05 was considered statistically significant.
Results
Table 1 lists the demographic data, including age, sex, and BMI, and compares the means. There were strong interobserver and intraobserver correlations for all LLD measurements (r > 0.9; P < .001). Mean (SD) LLD was 2.7 (1.8) mm (95% CI, 2.3-3.2) in the RTHA group, 1.8 (1.6) mm (95% CI, 1.2-2.4) in the ATHA group, and 1.9 (1.6) mm (95% CI, 1.5-2.4) in the PTHA group (P = .01). When LLD of more than 3 mm was set as an outlier, percentage of outliers was 37.3% (RTHA), 17.2% (ATHA), and 22% (PTHA) (P = .06-.78). When LLD of more than 5 mm was set as an outlier, percentage of outliers was 10.4% (RTHA), 6.9% (ATHA), and 8.5% (PTHA) (P = .72 to >.99). No patient in any group had LLD of 10 mm or more (Figure). Table 2 lists percentages of patients’ operated extremities that were longer, shorter, or the same size as their contralateral extremities. Six (9.0%) of the 67 RTHA patients, 4 (13.8%) of the 29 ATHA patients, and 3 (5.1%) of the 59 PTHA patients had a contralateral THA.
Discussion
Our study results showed that RTHA, ATHA, and PTHA were equally effective in minimizing LLD. There was a statistically significant difference in mean LLD among the 3 groups studied. The RTHA group had the largest mean (SD) LLD: 2.7 (1.8) mm. However, statistically significant differences do not always indicate clinical significance.27 Therefore, comparison of the 3 groups’ means is not enough for drawing significant conclusions. The more important point to consider is the number of cases of LLD of 10 mm or more—a discrepancy that would be perceptible to patients and thus become a source of dissatisfaction with painless THA.28 Patients perceive LLD when shortening exceeds 10 mm and lengthening exceeds 6 mm,29 or when LLD is more than 10 mm.16,19,20 Despite significant differences in means, all our cases came in under the 10-mm threshold. When the threshold was decreased to 5 mm (and to 3 mm), there was no statistically significant difference among the groups in the number of cases above the threshold.
LLD remains a source of significant post-THA comorbidity and patient dissatisfaction.1-7,19 Despite surgeons’ efforts to minimize LLD, some patients can detect even a subtle LLD after surgery.1,8,29 Most LLD values reported in the literature fall under the 10-mm threshold.16,19,20 In some cases, however, postoperative LLD is more than 1 cm, enough to prompt litigation against orthopedic surgeons.2 Surgeons have tried to improve LLD with use of multiple techniques, including use of intraoperative measuring devices,30 patient positioning during surgery,20 use of computer-assisted surgery,19 and use of intraoperative fluoroscopy.20
Proponents of computer-assisted THA have argued that this technique improves accuracy in placing the acetabular cup in the safe zone,31 minimizes LLD, and restores femoral offset.32,33 Manzotti and colleagues16 reported on 48 cases of computer-assisted THA matched to 48 cases of conventional THA using the posterior approach. Mean (SD) LLD was 5.06 (2.99) mm in the computer-assisted group and 7.64 (4.36) mm in the conventional group; there was a statistically significant difference in favor of the computer-assisted group (P = .04). However, 5 patients in the computer-assisted group and 13 in the conventional group had LLD of more than 10 mm, and the difference was statistically significant.16 Moreover, the study population was heterogeneous, with 12 patients in both groups having developmental dysplasia as a primary diagnosis.16 All the cases in our study had a diagnosis of OA, and no case had LLD of 10 mm or more.
Several advantages have been proposed for the anterior approach. The supine position (with direct comparison of leg lengths) and the use of fluoroscopy have been described as advantageous in minimizing LLD.20,21 In their study of 494 primary THAs performed with the anterior approach, Matta and colleagues20 reported mean (SD) postoperative LLD of 3 (2) mm (range, 0-26 mm) and concluded that the anterior approach was effective in restoring leg lengths and ensuring proper cup placement while not increasing the dislocation rate. However, they did not compare this approach with others or with computer-assisted THA with respect to LLD.
In another study, Nam and colleagues19 compared LLD after THA performed with 3 different approaches (anterior, conventional posterior, posterior-navigated) and found no statistically significant difference in LLD among the groups. However, LLD was more than 10 mm in 2.2% of anterior cases, 4.4% of conventional posterior cases, and 4.4% of posterior-navigated cases. When 5 mm was used as a cutoff, percentage of patients who were outliers was 31.1% (anterior), 20% (conventional posterior), and 23.3% (navigated-posterior). Our data showed superior results in using 5 mm as a cutoff, with percentage of outliers of 6.9% with ATHA, 8.5% with PTHA, and 10.4% with RTHA. However, Nam and colleagues19 used a larger patient cohort and different techniques for measuring LLD on anteroposterior pelvis radiographs.
The most likely reason that the groups in our study were comparable in terms of LLD accuracy and lack of outliers over the 10-mm cutoff was Dr. Domb’s high accuracy in minimizing LLD using each of the 3 techniques. For ATHA, mean (SD) LLD was 1.8 (1.6) mm (no LLD of ≥10 mm), better than the 3 (2) mm (0.9% with LLD of >10 mm) reported by Matta and colleagues20 and the 3.8 (3.9) mm (2.2% with LLD of >10 mm) reported by Nam and colleagues.19 For PTHA, mean (SD) LLD was 1.9 (1.6) mm (no LLD of ≥10 mm), comparable to some of the best results reported in the literature—for example, the 1 mm (3% with LLD of >10 mm) reported by Woolson and colleagues.34 For RTHA, mean (SD) LLD was 2.7 (1.8) mm (no LLD of ≥10 mm), superior to the 3.9 (2.7) mm (4.4% with LLD of >10 mm) reported by Nam and colleagues19 for posterior-navigated THA and the 5.06 (2.99) mm (10.4% with LLD of >10 mm) reported by Manzotti and colleagues16 for computer-assisted THA.
This study had several notable strengths. All patients had a diagnosis of hip OA and were operated on by a single surgeon. Radiographs were calibrated using the size of the implanted femoral head. Radiographic data were measured using the same technique in all cases and were collected twice by 2 observers (not the senior surgeon) to decrease bias and determine interobserver and intraobserver reliability. In addition, surgeon experience might have played an important role in minimizing LLD regardless of technique and approach used for THA.
Study limitations were different number of cases in each group, lack of matching, lack of clinical follow-up, and lack of long-term assessment of clinical outcomes and complications.
Conclusion
As performed by an experienced surgeon, RTHA, ATHA, and PTHA did not differ in obtaining minimal LLD. All 3 groups had a low frequency of outliers, using thresholds of 3 mm and 5 mm, and no patient in any group had LLD of 10 mm or more. All 3 techniques are effective in achieving accuracy in LLD.
Total hip arthroplasty (THA) effectively provides adequate pain relief and favorable outcomes in patients with hip osteoarthritis (OA). However, leg-length discrepancy (LLD) is still a significant cause of morbidity,1 including nerve damage,2,3 low back pain,2,4,5 and abnormal gait.2,6,7 Although most of the LLD values reported in the literature fall under the acceptable threshold of 10 mm,8 some patients report dissatisfaction,9 leading to litigation against orthopedic surgeons.2 However, lower extremity lengthening is sometimes needed to achieve adequate hip joint stability and prevent dislocations.2,10
Several methods have been developed to help surgeons estimate the change in leg length during surgery in an attempt to improve clinical outcomes. Use of guide pins as a reference on the pelvis decreased LLD and improved outcomes in some published studies.11,12 Preoperative templating of implant size, cup position, and level of femoral neck cut is very important in helping minimize clinically significant LLD after THA.2,13,14 Computer-assisted THA has also been introduced to try to improve component positioning, restoration of hip center of rotation, and minimizing of LLD.15-17 However, cost and increased operative time have prevented widespread adoption of computer-assisted surgery in THA.
Proponents of different surgical approaches have argued about the superiority of one approach over another. The posterior approach is the gold standard in THA because it is safe, easy to perform, and, if needed, extensile.11 However, exact determination of the intraoperative 3-dimensional (3-D) orientation of the pelvis, and subsequently of LLD, is challenging when the patient lies in the lateral position. The anterior approach has gained in popularity because of its advantages in accelerating postoperative rehabilitation and decreasing hospital length of stay.18 Placing the patient supine is advantageous because it allows leveling of the pelvis and estimation of LLD (by comparing the positions of the lower extremities).19 The anterior approach also allows for radiographic measurements on the operating table.19,20 However, this approach has a high learning curve21 and is not extensile.21 To date, no study has shown superiority of the anterior approach over either the conventional posterior approach or the robot-assisted posterior approach in minimizing LLD after THA.
We conducted a study to compare LLD in patients who underwent THA performed with a robot-assisted posterior approach (RTHA), a fluoroscopy-guided anterior approach (ATHA), or a conventional posterior approach (PTHA). We hypothesized that, compared with PTHA, both RTHA and ATHA would result in reduced LLD.
Materials and Methods
We reviewed all RTHAs, ATHAs, and PTHAs performed by Dr. Domb between September 2008 and December 2012. Study inclusion criteria were a diagnosis of hip OA and the availability of postoperative supine anteroposterior pelvis radiographs. Exclusion criteria were a diagnosis other than hip OA, missing or improper postoperative radiographs (radiographs with rotated or tilted pelvis),22 and radiographs on which at least one of the lesser trochanters was difficult to define. Of the 155 cases included in the study, 67 were RTHAs, 29 were ATHAs, and 59 were PTHAs.
All patients scheduled for THA underwent preoperative planning; plain radiographs were used to determine component size and position, level of neck cut, and amount of leg lengthening or shortening needed. In all RTHA cases, computed tomography of the involved hip was performed before surgery. The MAKO system (MAKO Surgical Corporation, Davie, Florida) was used to develop a patient-specific 3-D model of the pelvis and proximal femur, and this model was used to guide THA execution. The system was then used to detect patient-specific landmarks during surgery, to register the femur and the acetabulum, and to help determine the position of the pelvis and proximal femur during surgery. This system, which uses a haptic robotic arm that guides acetabular reaming and cup placement, provides feedback regarding cup placement, stem version, leg length, and global offset. Pelvic tilt and rotation were accounted for by the MAKO software, and all provided measurements were made on the coronal (functional) plane of the body, as described by Murray.23 ATHA was performed with the patient in the supine position on a Hana table (Mizuho OSI, Union City, California) with fluoroscopic guidance. PTHA was performed in the conventional way, with the patient in the lateral position.
Radiographic measurements of LLD were made with TraumaCad software (Build 2.2.535.0; Voyant Health, Petah-Tikva, Israel). The accuracy of this software has been studied and reported in the literature.24-26 Radiographs were calibrated using the known size of each femoral head as a marker. The reference on the pelvis was the interobturator line (line tangent to inferior border of obturator foramina), and the reference on the femurs was the most superior and medial aspect of each lesser trochanter. Two lines were drawn, each perpendicular to the interobturator line, starting from the previously defined reference point on each lesser trochanter. The difference in length between these 2 lines was recorded as the LLD. Values were recorded relative to the operative extremity. For example, if the operative extremity was longer than the nonoperative extremity, the LLD was given a positive value.
To eliminate bias and increase measurement accuracy, the study had each of 2 observers collect the LLD data twice, 2 months apart. These observers were blinded to each other’s results and to the type of surgery performed. (Neither observer was Dr. Domb, the senior surgeon.) IBM SPSS Statistics software (Version 20; IBM, Armonk, New York) was used for statistical analysis. Each patient’s 4 measurements were averaged into a single number for LLD, and the absolute LLD values were used in all statistical analyses. Means, standard deviations (SDs), and 95% confidence intervals (CIs) were calculated for LLD in each of the 3 groups. Pearson correlation coefficient was used to determine interobserver and intraobserver reliability. One-way analysis of variance (ANOVA) was used to compare group means for age, body mass index (BMI), and LLD. In each group, number of outliers was determined with outliers set at LLDs of more than 3 mm and more than 5 mm. Fischer exact test was used to compare number of outliers in each group. P < .05 was considered statistically significant.
Results
Table 1 lists the demographic data, including age, sex, and BMI, and compares the means. There were strong interobserver and intraobserver correlations for all LLD measurements (r > 0.9; P < .001). Mean (SD) LLD was 2.7 (1.8) mm (95% CI, 2.3-3.2) in the RTHA group, 1.8 (1.6) mm (95% CI, 1.2-2.4) in the ATHA group, and 1.9 (1.6) mm (95% CI, 1.5-2.4) in the PTHA group (P = .01). When LLD of more than 3 mm was set as an outlier, percentage of outliers was 37.3% (RTHA), 17.2% (ATHA), and 22% (PTHA) (P = .06-.78). When LLD of more than 5 mm was set as an outlier, percentage of outliers was 10.4% (RTHA), 6.9% (ATHA), and 8.5% (PTHA) (P = .72 to >.99). No patient in any group had LLD of 10 mm or more (Figure). Table 2 lists percentages of patients’ operated extremities that were longer, shorter, or the same size as their contralateral extremities. Six (9.0%) of the 67 RTHA patients, 4 (13.8%) of the 29 ATHA patients, and 3 (5.1%) of the 59 PTHA patients had a contralateral THA.
Discussion
Our study results showed that RTHA, ATHA, and PTHA were equally effective in minimizing LLD. There was a statistically significant difference in mean LLD among the 3 groups studied. The RTHA group had the largest mean (SD) LLD: 2.7 (1.8) mm. However, statistically significant differences do not always indicate clinical significance.27 Therefore, comparison of the 3 groups’ means is not enough for drawing significant conclusions. The more important point to consider is the number of cases of LLD of 10 mm or more—a discrepancy that would be perceptible to patients and thus become a source of dissatisfaction with painless THA.28 Patients perceive LLD when shortening exceeds 10 mm and lengthening exceeds 6 mm,29 or when LLD is more than 10 mm.16,19,20 Despite significant differences in means, all our cases came in under the 10-mm threshold. When the threshold was decreased to 5 mm (and to 3 mm), there was no statistically significant difference among the groups in the number of cases above the threshold.
LLD remains a source of significant post-THA comorbidity and patient dissatisfaction.1-7,19 Despite surgeons’ efforts to minimize LLD, some patients can detect even a subtle LLD after surgery.1,8,29 Most LLD values reported in the literature fall under the 10-mm threshold.16,19,20 In some cases, however, postoperative LLD is more than 1 cm, enough to prompt litigation against orthopedic surgeons.2 Surgeons have tried to improve LLD with use of multiple techniques, including use of intraoperative measuring devices,30 patient positioning during surgery,20 use of computer-assisted surgery,19 and use of intraoperative fluoroscopy.20
Proponents of computer-assisted THA have argued that this technique improves accuracy in placing the acetabular cup in the safe zone,31 minimizes LLD, and restores femoral offset.32,33 Manzotti and colleagues16 reported on 48 cases of computer-assisted THA matched to 48 cases of conventional THA using the posterior approach. Mean (SD) LLD was 5.06 (2.99) mm in the computer-assisted group and 7.64 (4.36) mm in the conventional group; there was a statistically significant difference in favor of the computer-assisted group (P = .04). However, 5 patients in the computer-assisted group and 13 in the conventional group had LLD of more than 10 mm, and the difference was statistically significant.16 Moreover, the study population was heterogeneous, with 12 patients in both groups having developmental dysplasia as a primary diagnosis.16 All the cases in our study had a diagnosis of OA, and no case had LLD of 10 mm or more.
Several advantages have been proposed for the anterior approach. The supine position (with direct comparison of leg lengths) and the use of fluoroscopy have been described as advantageous in minimizing LLD.20,21 In their study of 494 primary THAs performed with the anterior approach, Matta and colleagues20 reported mean (SD) postoperative LLD of 3 (2) mm (range, 0-26 mm) and concluded that the anterior approach was effective in restoring leg lengths and ensuring proper cup placement while not increasing the dislocation rate. However, they did not compare this approach with others or with computer-assisted THA with respect to LLD.
In another study, Nam and colleagues19 compared LLD after THA performed with 3 different approaches (anterior, conventional posterior, posterior-navigated) and found no statistically significant difference in LLD among the groups. However, LLD was more than 10 mm in 2.2% of anterior cases, 4.4% of conventional posterior cases, and 4.4% of posterior-navigated cases. When 5 mm was used as a cutoff, percentage of patients who were outliers was 31.1% (anterior), 20% (conventional posterior), and 23.3% (navigated-posterior). Our data showed superior results in using 5 mm as a cutoff, with percentage of outliers of 6.9% with ATHA, 8.5% with PTHA, and 10.4% with RTHA. However, Nam and colleagues19 used a larger patient cohort and different techniques for measuring LLD on anteroposterior pelvis radiographs.
The most likely reason that the groups in our study were comparable in terms of LLD accuracy and lack of outliers over the 10-mm cutoff was Dr. Domb’s high accuracy in minimizing LLD using each of the 3 techniques. For ATHA, mean (SD) LLD was 1.8 (1.6) mm (no LLD of ≥10 mm), better than the 3 (2) mm (0.9% with LLD of >10 mm) reported by Matta and colleagues20 and the 3.8 (3.9) mm (2.2% with LLD of >10 mm) reported by Nam and colleagues.19 For PTHA, mean (SD) LLD was 1.9 (1.6) mm (no LLD of ≥10 mm), comparable to some of the best results reported in the literature—for example, the 1 mm (3% with LLD of >10 mm) reported by Woolson and colleagues.34 For RTHA, mean (SD) LLD was 2.7 (1.8) mm (no LLD of ≥10 mm), superior to the 3.9 (2.7) mm (4.4% with LLD of >10 mm) reported by Nam and colleagues19 for posterior-navigated THA and the 5.06 (2.99) mm (10.4% with LLD of >10 mm) reported by Manzotti and colleagues16 for computer-assisted THA.
This study had several notable strengths. All patients had a diagnosis of hip OA and were operated on by a single surgeon. Radiographs were calibrated using the size of the implanted femoral head. Radiographic data were measured using the same technique in all cases and were collected twice by 2 observers (not the senior surgeon) to decrease bias and determine interobserver and intraobserver reliability. In addition, surgeon experience might have played an important role in minimizing LLD regardless of technique and approach used for THA.
Study limitations were different number of cases in each group, lack of matching, lack of clinical follow-up, and lack of long-term assessment of clinical outcomes and complications.
Conclusion
As performed by an experienced surgeon, RTHA, ATHA, and PTHA did not differ in obtaining minimal LLD. All 3 groups had a low frequency of outliers, using thresholds of 3 mm and 5 mm, and no patient in any group had LLD of 10 mm or more. All 3 techniques are effective in achieving accuracy in LLD.
1. Maloney WJ, Keeney JA. Leg length discrepancy after total hip arthroplasty. J Arthroplasty. 2004;19(4 suppl 1):108-110.
2. Clark CR, Huddleston HD, Schoch EP 3rd, Thomas BJ. Leg-length discrepancy after total hip arthroplasty. J Am Acad Orthop Surg. 2006;14(1):38-45.
3. Edwards BN, Tullos HS, Noble PC. Contributory factors and etiology of sciatic nerve palsy in total hip arthroplasty. Clin Orthop. 1987;(218):136-141.
4. Giles LG, Taylor JR. Low-back pain associated with leg length inequality. Spine. 1981;6(5):510-521.
5. Parvizi J, Sharkey PF, Bissett GA, Rothman RH, Hozack WJ. Surgical treatment of limb-length discrepancy following total hip arthroplasty. J Bone Joint Surg Am. 2003;85(12):2310-2317.
6. Edeen J, Sharkey PF, Alexander AH. Clinical significance of leg-length inequality after total hip arthroplasty. Am J Orthop. 1995;24(4):347-351.
7. Gurney B, Mermier C, Robergs R, Gibson A, Rivero D. Effects of limb-length discrepancy on gait economy and lower-extremity muscle activity in older adults. J Bone Joint Surg Am. 2001;83(6):907-915.
8. O’Brien S, Kernohan G, Fitzpatrick C, Hill J, Beverland D. Perception of imposed leg length inequality in normal subjects. Hip Int. 2010;20(4):505-511.
9. Hofmann AA, Skrzynski MC. Leg-length inequality and nerve palsy in total hip arthroplasty: a lawyer awaits! Orthopedics. 2000;23(9):943-944.
10. Miyamoto RG, Kaplan KM, Levine BR, Egol KA, Zuckerman JD. Surgical management of hip fractures: an evidence-based review of the literature. I: femoral neck fractures. J Am Acad Orthop Surg. 2008;16(10):596-607.
11. Ranawat CS, Rao RR, Rodriguez JA, Bhende HS. Correction of limb-length inequality during total hip arthroplasty. J Arthroplasty. 2001;16(6):715-720.
12. McGee HM, Scott JH. A simple method of obtaining equal leg length in total hip arthroplasty. Clin Orthop. 1985;(194):269-270.
13. Della Valle AG, Padgett DE, Salvati EA. Preoperative planning for primary total hip arthroplasty. J Am Acad Orthop Surg. 2005;13(7):455-462.
14. Gonzalez Della Valle A, Slullitel G, Piccaluga F, Salvati EA. The precision and usefulness of preoperative planning for cemented and hybrid primary total hip arthroplasty. J Arthroplasty. 2005;20(1):51-58.
15. Confalonieri N, Manzotti A, Montironi F, Pullen C. Leg length discrepancy, dislocation rate, and offset in total hip replacement using a short modular stem: navigation vs conventional freehand. Orthopedics. 2008;31(10 suppl 1).
16. Manzotti A, Cerveri P, De Momi E, Pullen C, Confalonieri N. Does computer-assisted surgery benefit leg length restoration in total hip replacement? Navigation versus conventional freehand. Int Orthop. 2011;35(1):19-24.
17. Nishio S, Fukunishi S, Fukui T, Fujihara Y, Yoshiya S. Adjustment of leg length using imageless navigation THA software without a femoral tracker. J Orthop Sci. 2011;16(2):171-176.
18. Martin CT, Pugely AJ, Gao Y, Clark CR. A comparison of hospital length of stay and short-term morbidity between the anterior and the posterior approaches to total hip arthroplasty. J Arthroplasty. 2013;28(5):849-854.
19. Nam D, Sculco PK, Abdel MP, Alexiades MM, Figgie MP, Mayman DJ. Leg-length inequalities following THA based on surgical technique. Orthopedics. 2013;36(4):e395-e400.
20. Matta JM, Shahrdar C, Ferguson T. Single-incision anterior approach for total hip arthroplasty on an orthopaedic table. Clin Orthop. 2005;(441):115-124.
21. Yi C, Agudelo JF, Dayton MR, Morgan SJ. Early complications of anterior supine intermuscular total hip arthroplasty. Orthopedics. 2013;36(3):e276-e281.
22. Siebenrock KA, Kalbermatten DF, Ganz R. Effect of pelvic tilt on acetabular retroversion: a study of pelves from cadavers. Clin Orthop. 2003;(407):241-248.
23. Murray DW. The definition and measurement of acetabular orientation. J Bone Joint Surg Br. 1993;75(2):228-232.
24. Kumar PG, Kirmani SJ, Humberg H, Kavarthapu V, Li P. Reproducibility and accuracy of templating uncemented THA with digital radiographic and digital TraumaCad templating software. Orthopedics. 2009;32(11):815.
25. Steinberg EL, Shasha N, Menahem A, Dekel S. Preoperative planning of total hip replacement using the TraumaCad system. Arch Orthop Trauma Surg. 2010;130(12):1429-1432.
26. Westacott DJ, McArthur J, King RJ, Foguet P. Assessment of cup orientation in hip resurfacing: a comparison of TraumaCad and computed tomography. J Orthop Surg Res. 2013;8:8.
27. Copay AG, Subach BR, Glassman SD, Polly DW Jr, Schuler TC. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 2007;7(5):541-546.
28. Abraham WD, Dimon JH 3rd. Leg length discrepancy in total hip arthroplasty. Orthop Clin North Am. 1992;23(2):201-209.
29. Konyves A, Bannister GC. The importance of leg length discrepancy after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(2):155-157.
30. Matsuda K, Nakamura S, Matsushita T. A simple method to minimize limb-length discrepancy after hip arthroplasty. Acta Orthop. 2006;77(3):375-379.
31. Haaker RG, Tiedjen K, Ottersbach A, Rubenthaler F, Stockheim M, Stiehl JB. Comparison of conventional versus computer-navigated acetabular component insertion. J Arthroplasty. 2007;22(2):151-159.
32. Renkawitz T, Schuster T, Herold T, et al. Measuring leg length and offset with an imageless navigation system during total hip arthroplasty: is it really accurate? Int J Med Robot. 2009;5(2):192-197.
33. Nakamura N, Sugano N, Nishii T, Kakimoto A, Miki H. A comparison between robotic-assisted and manual implantation of cementless total hip arthroplasty. Clin Orthop. 2010;468(4):1072-1081.
34. Woolson ST, Hartford JM, Sawyer A. Results of a method of leg-length equalization for patients undergoing primary total hip replacement. J Arthroplasty. 1999;14(2):159-164.
1. Maloney WJ, Keeney JA. Leg length discrepancy after total hip arthroplasty. J Arthroplasty. 2004;19(4 suppl 1):108-110.
2. Clark CR, Huddleston HD, Schoch EP 3rd, Thomas BJ. Leg-length discrepancy after total hip arthroplasty. J Am Acad Orthop Surg. 2006;14(1):38-45.
3. Edwards BN, Tullos HS, Noble PC. Contributory factors and etiology of sciatic nerve palsy in total hip arthroplasty. Clin Orthop. 1987;(218):136-141.
4. Giles LG, Taylor JR. Low-back pain associated with leg length inequality. Spine. 1981;6(5):510-521.
5. Parvizi J, Sharkey PF, Bissett GA, Rothman RH, Hozack WJ. Surgical treatment of limb-length discrepancy following total hip arthroplasty. J Bone Joint Surg Am. 2003;85(12):2310-2317.
6. Edeen J, Sharkey PF, Alexander AH. Clinical significance of leg-length inequality after total hip arthroplasty. Am J Orthop. 1995;24(4):347-351.
7. Gurney B, Mermier C, Robergs R, Gibson A, Rivero D. Effects of limb-length discrepancy on gait economy and lower-extremity muscle activity in older adults. J Bone Joint Surg Am. 2001;83(6):907-915.
8. O’Brien S, Kernohan G, Fitzpatrick C, Hill J, Beverland D. Perception of imposed leg length inequality in normal subjects. Hip Int. 2010;20(4):505-511.
9. Hofmann AA, Skrzynski MC. Leg-length inequality and nerve palsy in total hip arthroplasty: a lawyer awaits! Orthopedics. 2000;23(9):943-944.
10. Miyamoto RG, Kaplan KM, Levine BR, Egol KA, Zuckerman JD. Surgical management of hip fractures: an evidence-based review of the literature. I: femoral neck fractures. J Am Acad Orthop Surg. 2008;16(10):596-607.
11. Ranawat CS, Rao RR, Rodriguez JA, Bhende HS. Correction of limb-length inequality during total hip arthroplasty. J Arthroplasty. 2001;16(6):715-720.
12. McGee HM, Scott JH. A simple method of obtaining equal leg length in total hip arthroplasty. Clin Orthop. 1985;(194):269-270.
13. Della Valle AG, Padgett DE, Salvati EA. Preoperative planning for primary total hip arthroplasty. J Am Acad Orthop Surg. 2005;13(7):455-462.
14. Gonzalez Della Valle A, Slullitel G, Piccaluga F, Salvati EA. The precision and usefulness of preoperative planning for cemented and hybrid primary total hip arthroplasty. J Arthroplasty. 2005;20(1):51-58.
15. Confalonieri N, Manzotti A, Montironi F, Pullen C. Leg length discrepancy, dislocation rate, and offset in total hip replacement using a short modular stem: navigation vs conventional freehand. Orthopedics. 2008;31(10 suppl 1).
16. Manzotti A, Cerveri P, De Momi E, Pullen C, Confalonieri N. Does computer-assisted surgery benefit leg length restoration in total hip replacement? Navigation versus conventional freehand. Int Orthop. 2011;35(1):19-24.
17. Nishio S, Fukunishi S, Fukui T, Fujihara Y, Yoshiya S. Adjustment of leg length using imageless navigation THA software without a femoral tracker. J Orthop Sci. 2011;16(2):171-176.
18. Martin CT, Pugely AJ, Gao Y, Clark CR. A comparison of hospital length of stay and short-term morbidity between the anterior and the posterior approaches to total hip arthroplasty. J Arthroplasty. 2013;28(5):849-854.
19. Nam D, Sculco PK, Abdel MP, Alexiades MM, Figgie MP, Mayman DJ. Leg-length inequalities following THA based on surgical technique. Orthopedics. 2013;36(4):e395-e400.
20. Matta JM, Shahrdar C, Ferguson T. Single-incision anterior approach for total hip arthroplasty on an orthopaedic table. Clin Orthop. 2005;(441):115-124.
21. Yi C, Agudelo JF, Dayton MR, Morgan SJ. Early complications of anterior supine intermuscular total hip arthroplasty. Orthopedics. 2013;36(3):e276-e281.
22. Siebenrock KA, Kalbermatten DF, Ganz R. Effect of pelvic tilt on acetabular retroversion: a study of pelves from cadavers. Clin Orthop. 2003;(407):241-248.
23. Murray DW. The definition and measurement of acetabular orientation. J Bone Joint Surg Br. 1993;75(2):228-232.
24. Kumar PG, Kirmani SJ, Humberg H, Kavarthapu V, Li P. Reproducibility and accuracy of templating uncemented THA with digital radiographic and digital TraumaCad templating software. Orthopedics. 2009;32(11):815.
25. Steinberg EL, Shasha N, Menahem A, Dekel S. Preoperative planning of total hip replacement using the TraumaCad system. Arch Orthop Trauma Surg. 2010;130(12):1429-1432.
26. Westacott DJ, McArthur J, King RJ, Foguet P. Assessment of cup orientation in hip resurfacing: a comparison of TraumaCad and computed tomography. J Orthop Surg Res. 2013;8:8.
27. Copay AG, Subach BR, Glassman SD, Polly DW Jr, Schuler TC. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 2007;7(5):541-546.
28. Abraham WD, Dimon JH 3rd. Leg length discrepancy in total hip arthroplasty. Orthop Clin North Am. 1992;23(2):201-209.
29. Konyves A, Bannister GC. The importance of leg length discrepancy after total hip arthroplasty. J Bone Joint Surg Br. 2005;87(2):155-157.
30. Matsuda K, Nakamura S, Matsushita T. A simple method to minimize limb-length discrepancy after hip arthroplasty. Acta Orthop. 2006;77(3):375-379.
31. Haaker RG, Tiedjen K, Ottersbach A, Rubenthaler F, Stockheim M, Stiehl JB. Comparison of conventional versus computer-navigated acetabular component insertion. J Arthroplasty. 2007;22(2):151-159.
32. Renkawitz T, Schuster T, Herold T, et al. Measuring leg length and offset with an imageless navigation system during total hip arthroplasty: is it really accurate? Int J Med Robot. 2009;5(2):192-197.
33. Nakamura N, Sugano N, Nishii T, Kakimoto A, Miki H. A comparison between robotic-assisted and manual implantation of cementless total hip arthroplasty. Clin Orthop. 2010;468(4):1072-1081.
34. Woolson ST, Hartford JM, Sawyer A. Results of a method of leg-length equalization for patients undergoing primary total hip replacement. J Arthroplasty. 1999;14(2):159-164.
Teaching trainees how to discern professional boundaries
Psychiatrists often serve as risk-management consultants for our medical colleagues. As part of this role, psychiatrists working with trainees— including resident physicians, medical students, and physician assistant students— have an opportunity to emphasize the importance of professional boundaries.1 Discussing appropriate professional boundaries and describing what might represent a violation of these boundaries is meaningful because a good understanding of these concepts promotes high-quality treatment and minimizes professional liability.2
Physical boundaries
Psychiatric patients might be agitated or display potentially dangerous behaviors; discussing the importance of body language and contact between physicians and their patients is, therefore, first and foremost, a matter of safety. Students who can recognize the signs and symptoms of agitation and maintain a safe distance between themselves and their patients are less likely to be injured.
Addressing romantic and sexual relationships between patients and their health care providers also is necessary. One study reported that 21% of medical students surveyed might not regard sexual contact with a patient as inappropriate.3 An adequate discussion of this topic is necessary to protect trainees and patients from a catastrophic misstep.
Emotional boundaries
Maintaining appropriate emotional boundaries is necessary in psychiatry. Given the prevalence of mental illness and substance abuse, many trainees have personal experience with psychiatric illness outside of their training. Discussing issues of transference and countertransference with students will prepare them for intense emotional reactions they will experience while working in psychiatry. Students who feel comfortable recognizing their own countertransference feelings and discussing them in supervision with their attending psychiatrist will be more successful in addressing the complex interpersonal challenges that their patients face.
Personal and informational boundaries
Discussing personal and informational boundaries can protect trainees from uncomfortable experiences in their non-clinical lives. Although, in previous decades, we needed to discourage students only from sharing their home address and telephone number with patients, the Internet and social media have made it easier for patients to discover personal information about their treatment team. Addressing issues related to social networks and instructing students on how to appropriately address and decline requests for personal information can prevent unwanted boundary crossings.
Psychiatrists are well suited to discuss these issues with trainees. In doing so, we can help them become knowledgeable health care providers—no matter which medical discipline they specialize in.
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Duckworth KS, Kahn MW, Gutheil TG. Roles, quandaries, and remedies: teaching professional boundaries to medical students. Harv Rev Psychiatry. 1994;1(5):266-270.
2. Gutheil TG, Gabbard GO. The concept of boundaries in clinical practice: theoretical and risk-management dimensions. Am J Psychiatry. 1993;150(2):188-196.
3. White GE. Medical students’ learning needs about setting and maintaining social and sexual boundaries: a report. Med Educ. 2003;37(11):1017-1019.
Psychiatrists often serve as risk-management consultants for our medical colleagues. As part of this role, psychiatrists working with trainees— including resident physicians, medical students, and physician assistant students— have an opportunity to emphasize the importance of professional boundaries.1 Discussing appropriate professional boundaries and describing what might represent a violation of these boundaries is meaningful because a good understanding of these concepts promotes high-quality treatment and minimizes professional liability.2
Physical boundaries
Psychiatric patients might be agitated or display potentially dangerous behaviors; discussing the importance of body language and contact between physicians and their patients is, therefore, first and foremost, a matter of safety. Students who can recognize the signs and symptoms of agitation and maintain a safe distance between themselves and their patients are less likely to be injured.
Addressing romantic and sexual relationships between patients and their health care providers also is necessary. One study reported that 21% of medical students surveyed might not regard sexual contact with a patient as inappropriate.3 An adequate discussion of this topic is necessary to protect trainees and patients from a catastrophic misstep.
Emotional boundaries
Maintaining appropriate emotional boundaries is necessary in psychiatry. Given the prevalence of mental illness and substance abuse, many trainees have personal experience with psychiatric illness outside of their training. Discussing issues of transference and countertransference with students will prepare them for intense emotional reactions they will experience while working in psychiatry. Students who feel comfortable recognizing their own countertransference feelings and discussing them in supervision with their attending psychiatrist will be more successful in addressing the complex interpersonal challenges that their patients face.
Personal and informational boundaries
Discussing personal and informational boundaries can protect trainees from uncomfortable experiences in their non-clinical lives. Although, in previous decades, we needed to discourage students only from sharing their home address and telephone number with patients, the Internet and social media have made it easier for patients to discover personal information about their treatment team. Addressing issues related to social networks and instructing students on how to appropriately address and decline requests for personal information can prevent unwanted boundary crossings.
Psychiatrists are well suited to discuss these issues with trainees. In doing so, we can help them become knowledgeable health care providers—no matter which medical discipline they specialize in.
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Psychiatrists often serve as risk-management consultants for our medical colleagues. As part of this role, psychiatrists working with trainees— including resident physicians, medical students, and physician assistant students— have an opportunity to emphasize the importance of professional boundaries.1 Discussing appropriate professional boundaries and describing what might represent a violation of these boundaries is meaningful because a good understanding of these concepts promotes high-quality treatment and minimizes professional liability.2
Physical boundaries
Psychiatric patients might be agitated or display potentially dangerous behaviors; discussing the importance of body language and contact between physicians and their patients is, therefore, first and foremost, a matter of safety. Students who can recognize the signs and symptoms of agitation and maintain a safe distance between themselves and their patients are less likely to be injured.
Addressing romantic and sexual relationships between patients and their health care providers also is necessary. One study reported that 21% of medical students surveyed might not regard sexual contact with a patient as inappropriate.3 An adequate discussion of this topic is necessary to protect trainees and patients from a catastrophic misstep.
Emotional boundaries
Maintaining appropriate emotional boundaries is necessary in psychiatry. Given the prevalence of mental illness and substance abuse, many trainees have personal experience with psychiatric illness outside of their training. Discussing issues of transference and countertransference with students will prepare them for intense emotional reactions they will experience while working in psychiatry. Students who feel comfortable recognizing their own countertransference feelings and discussing them in supervision with their attending psychiatrist will be more successful in addressing the complex interpersonal challenges that their patients face.
Personal and informational boundaries
Discussing personal and informational boundaries can protect trainees from uncomfortable experiences in their non-clinical lives. Although, in previous decades, we needed to discourage students only from sharing their home address and telephone number with patients, the Internet and social media have made it easier for patients to discover personal information about their treatment team. Addressing issues related to social networks and instructing students on how to appropriately address and decline requests for personal information can prevent unwanted boundary crossings.
Psychiatrists are well suited to discuss these issues with trainees. In doing so, we can help them become knowledgeable health care providers—no matter which medical discipline they specialize in.
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Duckworth KS, Kahn MW, Gutheil TG. Roles, quandaries, and remedies: teaching professional boundaries to medical students. Harv Rev Psychiatry. 1994;1(5):266-270.
2. Gutheil TG, Gabbard GO. The concept of boundaries in clinical practice: theoretical and risk-management dimensions. Am J Psychiatry. 1993;150(2):188-196.
3. White GE. Medical students’ learning needs about setting and maintaining social and sexual boundaries: a report. Med Educ. 2003;37(11):1017-1019.
1. Duckworth KS, Kahn MW, Gutheil TG. Roles, quandaries, and remedies: teaching professional boundaries to medical students. Harv Rev Psychiatry. 1994;1(5):266-270.
2. Gutheil TG, Gabbard GO. The concept of boundaries in clinical practice: theoretical and risk-management dimensions. Am J Psychiatry. 1993;150(2):188-196.
3. White GE. Medical students’ learning needs about setting and maintaining social and sexual boundaries: a report. Med Educ. 2003;37(11):1017-1019.
Computer Navigation and Robotics for Total Knee Arthroplasty
Total knee arthroplasty (TKA) is a good surgical option to relieve pain and improve function in patients with osteoarthritis. The goal of surgery is to achieve a well-aligned prosthesis with well-balanced ligaments in order to minimize wear and improve implant survival. Overall, 82% to 89% of patients are satisfied with their outcomes after TKA, with good 10- to 15-year implant survivorship; however, there is still a subset of patients that are unsatisfied. In many cases, patient dissatisfaction is attributed to improper component alignment.1-3 Over the past decade, computer navigation and robotics have been introduced to control surgical variables so as to gain greater consistency in implant placement and postoperative component alignment.
Computer-assisted navigation tools were introduced not only to improve implant alignment but, more importantly, to optimize clinical outcomes. Most studies have demonstrated that the use of navigation is associated with fewer radiographic outliers after TKA.4 Various studies have compared radiographic results of navigated TKA with results of TKA using standard instrumentation.4-7 While long-term studies are necessary, short-term follow-up has shown that computer-assisted TKA can improve alignment, especially in patients with severe deformity.8-10 Currently, there is no definitive consensus that computer-assisted TKA leads to significantly better component alignment or postoperative outcomes due to the fact that many studies are limited by study design or small cohorts. However, the currently published articles support better component alignment and clinical outcomes with computer-assisted TKA. While some argue that the use of computer-assisted surgery is dependent on the user’s experience, computer-assisted surgery can assist less-experienced surgeons to reliably achieve good midterm outcomes with a low complication rate.8,11 Various studies have looked at computer-assisted TKA at midterm follow-up, with no significant differences in clinical outcome between navigated and traditional techniques. However, long-term studies showing the benefits of computer navigation are beginning to emerge. For example, de Steiger and colleagues12 recently found that computer-assisted TKA reduced the overall revision rate for loosening after TKA in patients less than 65 years of age.
While surgical navigation helps improve implant planning, robotic tools have emerged as a tool to help refine surgical execution. Coupled with surgical navigation tools, robotic control of surgical gestures may further enhance precision in implant placement and/or enable novel implant design features. At present, robotic techniques are increasingly used in unicompartmental knee arthroplasty (UKA) and TKA.13 Studies have demonstrated that the robotic tool is 3 times more accurate with 3 times less variability than conventional techniques in UKA.14 The utility of robotic tools for TKA remains unclear. Robotic-driven automatic cutting guides have been shown to reduce time and improve accuracy compared with navigation guides in femoral TKA cutting procedures in a cadaveric model.15 However, robotic-enabled TKA procedures are poorly described at present, and the clinical implications of their proposed improved precision remain unclear.
Computer navigation and robotic tools in TKA hold the promise of enhanced control of surgical variables that influence clinical outcome. The variables that may be impacted by these advanced tools include implant positioning, lower limb alignment, soft-tissue balance, and, potentially, implant design and fixation. At present, these tools have primarily been shown to improve lower limb alignment in TKA. The clinical impact of the enhanced control of this single surgical variable (lower limb alignment) has been muted in short-term and midterm studies. Future studies should be directed at understanding which surgical variable, or combination of variables, it is most essential to precisely control so as to positively impact clinical outcomes. ◾
1. Bourne RB, Chesworth BM, Davis AM, Mahomed NN, Charron KD. Patient satisfaction after total knee arthroplasty: who is satisfied and who is not? Clin Orthop Relat Res. 2010;468(1):57-63.
2. Sharkey PF, Hozack WJ, Rothman RH, Shastri S, Jacoby SM. Insall Award paper. Why are total knee arthroplasties failing today? Clin Orthop Relat Res. 2002;(404):7-13.
3. Emmerson KP, Morgan CG, Pinder IM. Survivorship analysis of the Kinematic Stabilizer total knee replacement: a 10- to 14-year follow-up. J Bone Joint Surg Br. 1996;78(3):441-445.
4. Liow MH, Xia Z, Wong MK, Tay KJ, Yeo SJ, Chin PL. Robot-assisted total knee arthroplasty accurately restores the joint line and mechanical axis. A prospective randomized study. J Arthroplasty. 2014;29(12):2373-2377.
5. Sparmann M, Wolke B, Czupalla H, Banzer D, Zink A. Positioning of total knee arthroplasty with and without navigation support. A prospective, randomized study. J Bone Joint Surg Br. 2003;85(6):830-835.
6. Hoffart HE, Langenstein E, Vasak N. A prospective study comparing the functional outcome of computer-assisted and conventional total knee replacement. J Bone Joint Surg Br. 2012;94(2):194-199.
7. Cip J, Widemschek M, Luegmair M, Sheinkop MB, Benesch T, Martin A. Conventional versus computer-assisted technique for total knee arthroplasty: a minimum of 5-year follow-up of 200 patients in a prospective randomized comparative trial. J Arthroplasty. 2014;29(9):1795-1802.
8. Huang TW, Peng KT, Huang KC, Lee MS, Hsu RW. Differences in component and limb alignment between computer-assisted and conventional surgery total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):2954-2961.
9. Lee CY, Lin SJ, Kuo LT, et al. The benefits of computer-assisted total knee arthroplasty on coronal alignment with marked femoral bowing in Asian patients. J Orthop Surg Res. 2014;9:122.
10. Hernandez-Vaquero D, Noriega-Fernandez A, Fernandez-Carreira JM, Fernandez-Simon JM, Llorens de los Rios J. Computer-assisted surgery improves rotational positioning of the femoral component but not the tibial component in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):3127-3134.
11. Khakha RS, Chowdhry M, Sivaprakasam M, Kheiran A, Chauhan SK. Radiological and functional outcomes in computer assisted total knee arthroplasty between consultants and trainees - a prospective randomized controlled trial [published online ahead of print March 14, 2015]. J Arthroplasty.
12. de Steiger RN, Liu YL, Graves SE. Computer navigation for total knee arthroplasty reduces revision rate for patients less than sixty-five years of age. J Bone Joint Surg Am. 2015;97(8):635-642.
13. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
14. Citak M, Suero EM, Citak M, et al. Unicompartmental knee arthroplasty: is robotic technology more accurate than conventional technique? Knee. 2013;20(4):268-271.
15. Koulalis D, O’Loughlin PF, Plaskos C, Kendoff D, Cross MB, Pearle AD. Sequential versus automated cutting guides in computer-assisted total knee arthroplasty. Knee. 2011;18(6):436-442.
Total knee arthroplasty (TKA) is a good surgical option to relieve pain and improve function in patients with osteoarthritis. The goal of surgery is to achieve a well-aligned prosthesis with well-balanced ligaments in order to minimize wear and improve implant survival. Overall, 82% to 89% of patients are satisfied with their outcomes after TKA, with good 10- to 15-year implant survivorship; however, there is still a subset of patients that are unsatisfied. In many cases, patient dissatisfaction is attributed to improper component alignment.1-3 Over the past decade, computer navigation and robotics have been introduced to control surgical variables so as to gain greater consistency in implant placement and postoperative component alignment.
Computer-assisted navigation tools were introduced not only to improve implant alignment but, more importantly, to optimize clinical outcomes. Most studies have demonstrated that the use of navigation is associated with fewer radiographic outliers after TKA.4 Various studies have compared radiographic results of navigated TKA with results of TKA using standard instrumentation.4-7 While long-term studies are necessary, short-term follow-up has shown that computer-assisted TKA can improve alignment, especially in patients with severe deformity.8-10 Currently, there is no definitive consensus that computer-assisted TKA leads to significantly better component alignment or postoperative outcomes due to the fact that many studies are limited by study design or small cohorts. However, the currently published articles support better component alignment and clinical outcomes with computer-assisted TKA. While some argue that the use of computer-assisted surgery is dependent on the user’s experience, computer-assisted surgery can assist less-experienced surgeons to reliably achieve good midterm outcomes with a low complication rate.8,11 Various studies have looked at computer-assisted TKA at midterm follow-up, with no significant differences in clinical outcome between navigated and traditional techniques. However, long-term studies showing the benefits of computer navigation are beginning to emerge. For example, de Steiger and colleagues12 recently found that computer-assisted TKA reduced the overall revision rate for loosening after TKA in patients less than 65 years of age.
While surgical navigation helps improve implant planning, robotic tools have emerged as a tool to help refine surgical execution. Coupled with surgical navigation tools, robotic control of surgical gestures may further enhance precision in implant placement and/or enable novel implant design features. At present, robotic techniques are increasingly used in unicompartmental knee arthroplasty (UKA) and TKA.13 Studies have demonstrated that the robotic tool is 3 times more accurate with 3 times less variability than conventional techniques in UKA.14 The utility of robotic tools for TKA remains unclear. Robotic-driven automatic cutting guides have been shown to reduce time and improve accuracy compared with navigation guides in femoral TKA cutting procedures in a cadaveric model.15 However, robotic-enabled TKA procedures are poorly described at present, and the clinical implications of their proposed improved precision remain unclear.
Computer navigation and robotic tools in TKA hold the promise of enhanced control of surgical variables that influence clinical outcome. The variables that may be impacted by these advanced tools include implant positioning, lower limb alignment, soft-tissue balance, and, potentially, implant design and fixation. At present, these tools have primarily been shown to improve lower limb alignment in TKA. The clinical impact of the enhanced control of this single surgical variable (lower limb alignment) has been muted in short-term and midterm studies. Future studies should be directed at understanding which surgical variable, or combination of variables, it is most essential to precisely control so as to positively impact clinical outcomes. ◾
Total knee arthroplasty (TKA) is a good surgical option to relieve pain and improve function in patients with osteoarthritis. The goal of surgery is to achieve a well-aligned prosthesis with well-balanced ligaments in order to minimize wear and improve implant survival. Overall, 82% to 89% of patients are satisfied with their outcomes after TKA, with good 10- to 15-year implant survivorship; however, there is still a subset of patients that are unsatisfied. In many cases, patient dissatisfaction is attributed to improper component alignment.1-3 Over the past decade, computer navigation and robotics have been introduced to control surgical variables so as to gain greater consistency in implant placement and postoperative component alignment.
Computer-assisted navigation tools were introduced not only to improve implant alignment but, more importantly, to optimize clinical outcomes. Most studies have demonstrated that the use of navigation is associated with fewer radiographic outliers after TKA.4 Various studies have compared radiographic results of navigated TKA with results of TKA using standard instrumentation.4-7 While long-term studies are necessary, short-term follow-up has shown that computer-assisted TKA can improve alignment, especially in patients with severe deformity.8-10 Currently, there is no definitive consensus that computer-assisted TKA leads to significantly better component alignment or postoperative outcomes due to the fact that many studies are limited by study design or small cohorts. However, the currently published articles support better component alignment and clinical outcomes with computer-assisted TKA. While some argue that the use of computer-assisted surgery is dependent on the user’s experience, computer-assisted surgery can assist less-experienced surgeons to reliably achieve good midterm outcomes with a low complication rate.8,11 Various studies have looked at computer-assisted TKA at midterm follow-up, with no significant differences in clinical outcome between navigated and traditional techniques. However, long-term studies showing the benefits of computer navigation are beginning to emerge. For example, de Steiger and colleagues12 recently found that computer-assisted TKA reduced the overall revision rate for loosening after TKA in patients less than 65 years of age.
While surgical navigation helps improve implant planning, robotic tools have emerged as a tool to help refine surgical execution. Coupled with surgical navigation tools, robotic control of surgical gestures may further enhance precision in implant placement and/or enable novel implant design features. At present, robotic techniques are increasingly used in unicompartmental knee arthroplasty (UKA) and TKA.13 Studies have demonstrated that the robotic tool is 3 times more accurate with 3 times less variability than conventional techniques in UKA.14 The utility of robotic tools for TKA remains unclear. Robotic-driven automatic cutting guides have been shown to reduce time and improve accuracy compared with navigation guides in femoral TKA cutting procedures in a cadaveric model.15 However, robotic-enabled TKA procedures are poorly described at present, and the clinical implications of their proposed improved precision remain unclear.
Computer navigation and robotic tools in TKA hold the promise of enhanced control of surgical variables that influence clinical outcome. The variables that may be impacted by these advanced tools include implant positioning, lower limb alignment, soft-tissue balance, and, potentially, implant design and fixation. At present, these tools have primarily been shown to improve lower limb alignment in TKA. The clinical impact of the enhanced control of this single surgical variable (lower limb alignment) has been muted in short-term and midterm studies. Future studies should be directed at understanding which surgical variable, or combination of variables, it is most essential to precisely control so as to positively impact clinical outcomes. ◾
1. Bourne RB, Chesworth BM, Davis AM, Mahomed NN, Charron KD. Patient satisfaction after total knee arthroplasty: who is satisfied and who is not? Clin Orthop Relat Res. 2010;468(1):57-63.
2. Sharkey PF, Hozack WJ, Rothman RH, Shastri S, Jacoby SM. Insall Award paper. Why are total knee arthroplasties failing today? Clin Orthop Relat Res. 2002;(404):7-13.
3. Emmerson KP, Morgan CG, Pinder IM. Survivorship analysis of the Kinematic Stabilizer total knee replacement: a 10- to 14-year follow-up. J Bone Joint Surg Br. 1996;78(3):441-445.
4. Liow MH, Xia Z, Wong MK, Tay KJ, Yeo SJ, Chin PL. Robot-assisted total knee arthroplasty accurately restores the joint line and mechanical axis. A prospective randomized study. J Arthroplasty. 2014;29(12):2373-2377.
5. Sparmann M, Wolke B, Czupalla H, Banzer D, Zink A. Positioning of total knee arthroplasty with and without navigation support. A prospective, randomized study. J Bone Joint Surg Br. 2003;85(6):830-835.
6. Hoffart HE, Langenstein E, Vasak N. A prospective study comparing the functional outcome of computer-assisted and conventional total knee replacement. J Bone Joint Surg Br. 2012;94(2):194-199.
7. Cip J, Widemschek M, Luegmair M, Sheinkop MB, Benesch T, Martin A. Conventional versus computer-assisted technique for total knee arthroplasty: a minimum of 5-year follow-up of 200 patients in a prospective randomized comparative trial. J Arthroplasty. 2014;29(9):1795-1802.
8. Huang TW, Peng KT, Huang KC, Lee MS, Hsu RW. Differences in component and limb alignment between computer-assisted and conventional surgery total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):2954-2961.
9. Lee CY, Lin SJ, Kuo LT, et al. The benefits of computer-assisted total knee arthroplasty on coronal alignment with marked femoral bowing in Asian patients. J Orthop Surg Res. 2014;9:122.
10. Hernandez-Vaquero D, Noriega-Fernandez A, Fernandez-Carreira JM, Fernandez-Simon JM, Llorens de los Rios J. Computer-assisted surgery improves rotational positioning of the femoral component but not the tibial component in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):3127-3134.
11. Khakha RS, Chowdhry M, Sivaprakasam M, Kheiran A, Chauhan SK. Radiological and functional outcomes in computer assisted total knee arthroplasty between consultants and trainees - a prospective randomized controlled trial [published online ahead of print March 14, 2015]. J Arthroplasty.
12. de Steiger RN, Liu YL, Graves SE. Computer navigation for total knee arthroplasty reduces revision rate for patients less than sixty-five years of age. J Bone Joint Surg Am. 2015;97(8):635-642.
13. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
14. Citak M, Suero EM, Citak M, et al. Unicompartmental knee arthroplasty: is robotic technology more accurate than conventional technique? Knee. 2013;20(4):268-271.
15. Koulalis D, O’Loughlin PF, Plaskos C, Kendoff D, Cross MB, Pearle AD. Sequential versus automated cutting guides in computer-assisted total knee arthroplasty. Knee. 2011;18(6):436-442.
1. Bourne RB, Chesworth BM, Davis AM, Mahomed NN, Charron KD. Patient satisfaction after total knee arthroplasty: who is satisfied and who is not? Clin Orthop Relat Res. 2010;468(1):57-63.
2. Sharkey PF, Hozack WJ, Rothman RH, Shastri S, Jacoby SM. Insall Award paper. Why are total knee arthroplasties failing today? Clin Orthop Relat Res. 2002;(404):7-13.
3. Emmerson KP, Morgan CG, Pinder IM. Survivorship analysis of the Kinematic Stabilizer total knee replacement: a 10- to 14-year follow-up. J Bone Joint Surg Br. 1996;78(3):441-445.
4. Liow MH, Xia Z, Wong MK, Tay KJ, Yeo SJ, Chin PL. Robot-assisted total knee arthroplasty accurately restores the joint line and mechanical axis. A prospective randomized study. J Arthroplasty. 2014;29(12):2373-2377.
5. Sparmann M, Wolke B, Czupalla H, Banzer D, Zink A. Positioning of total knee arthroplasty with and without navigation support. A prospective, randomized study. J Bone Joint Surg Br. 2003;85(6):830-835.
6. Hoffart HE, Langenstein E, Vasak N. A prospective study comparing the functional outcome of computer-assisted and conventional total knee replacement. J Bone Joint Surg Br. 2012;94(2):194-199.
7. Cip J, Widemschek M, Luegmair M, Sheinkop MB, Benesch T, Martin A. Conventional versus computer-assisted technique for total knee arthroplasty: a minimum of 5-year follow-up of 200 patients in a prospective randomized comparative trial. J Arthroplasty. 2014;29(9):1795-1802.
8. Huang TW, Peng KT, Huang KC, Lee MS, Hsu RW. Differences in component and limb alignment between computer-assisted and conventional surgery total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):2954-2961.
9. Lee CY, Lin SJ, Kuo LT, et al. The benefits of computer-assisted total knee arthroplasty on coronal alignment with marked femoral bowing in Asian patients. J Orthop Surg Res. 2014;9:122.
10. Hernandez-Vaquero D, Noriega-Fernandez A, Fernandez-Carreira JM, Fernandez-Simon JM, Llorens de los Rios J. Computer-assisted surgery improves rotational positioning of the femoral component but not the tibial component in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2014;22(12):3127-3134.
11. Khakha RS, Chowdhry M, Sivaprakasam M, Kheiran A, Chauhan SK. Radiological and functional outcomes in computer assisted total knee arthroplasty between consultants and trainees - a prospective randomized controlled trial [published online ahead of print March 14, 2015]. J Arthroplasty.
12. de Steiger RN, Liu YL, Graves SE. Computer navigation for total knee arthroplasty reduces revision rate for patients less than sixty-five years of age. J Bone Joint Surg Am. 2015;97(8):635-642.
13. Pearle AD, O’Loughlin PF, Kendoff DO. Robot-assisted unicompartmental knee arthroplasty. J Arthroplasty. 2010;25(2):230-237.
14. Citak M, Suero EM, Citak M, et al. Unicompartmental knee arthroplasty: is robotic technology more accurate than conventional technique? Knee. 2013;20(4):268-271.
15. Koulalis D, O’Loughlin PF, Plaskos C, Kendoff D, Cross MB, Pearle AD. Sequential versus automated cutting guides in computer-assisted total knee arthroplasty. Knee. 2011;18(6):436-442.
DDW: Postbleed blood thinners up rebleeding risk, lower death risk
WASHINGTON – Early resumption of antiplatelet agents or anticoagulants after a major gastrointestinal bleeding event is clearly associated with an increased risk of rebleeding, but a decreased risk of death, results from an observational study show.
Furthermore, anticoagulant treatment “is associated with a higher risk of rebleeding and death compared with antiplatelet treatment after a previous GI event,” Dr. Angel Lanas said to an overflow crowd at the annual Digestive Disease Week.
In a separate case-control study, Dr. Lanas and his associates recently reported that the risk of GI bleeding was twofold higher for anticoagulants than for low-dose aspirin in patients hospitalized for GI bleeding (Clin. Gastroenterol. Hepatol. 2015 May;13:906-12.e2. [doi:10.1016/j.cgh.2014.11.007])
The current study examined adverse events in a cohort of 160 patients who developed a major gastrointestinal bleed (GIB) while using anticoagulants and/or antiplatelet therapy between March 2008 and July 2013. Long-term interruption or short-term resumption of these treatments has important clinical implications and differences in the intrinsic risks between antiplatelet or anticoagulant users after drug resumption are not well established, said Dr. Lanas of the University of Zaragoza, Spain.
Drug use information was prospectively collected during the GIB event, with data during the follow-up period obtained from two different Spanish databases.
Treatment during the index bleeding event was continued without interruption in 11 patients and interrupted in 149 patients (93%). Among those whose therapy was interrupted, 21 (14%) never resumed therapy and 128 (86%) resumed therapy (118 patients within 15 days and 10 patients after 15 days). The 86% treatment resumption rate is much higher than the 40%-66% rates reported in previous studies, indicating that Spanish physicians restarted treatment quite early, Dr. Lanas observed.
The mean age at baseline was 76.6 years, 61.3% of patients were men, and half had a Charlson index score > 4. Median follow-up was 21.5 months (range 1-63 months).
Ischemic events did not differ between patients who did or did not restart anticoagulants or antiplatelets (16.4% vs. 14.3%; P value = .806). However, rebleeding occurred in 32% of patients who resumed therapy versus none who did not (P = .002), but deaths were higher in those who did not restart therapy (38.1% vs. 12.5%; P = .003), Dr. Lanas said.
These differences remain significant in Kaplan-Meier survival curves for death (P = .021) and rebleeding (P = .004).
A comparison of early therapy resumption (≤ 15 days) vs. delayed (mean delay 62 days) or no resumption revealed similar results. Early resumption was associated with a higher rate of rebleeding (32.2% vs. 9.7%; P = .012), but a lower rate of death (11% vs. 35.5%; P = .001), with no difference in ischemic events (17% vs. 13%; P = .586), Dr. Lanas said.
Again, the differences remain significant in Kaplan-Meier survival curves for death (P = .011) and rebleeding (P = .013).
When the investigators looked at rebleeding according to drug use, patients receiving anticoagulants vs. antiplatelets had significantly higher rates of rebleeding (34.7% vs. 20.5%; P = .043), death (22.2% vs. 10.2%; P = .038), and any event (68.1% vs. 52.3%; P = .043).
After adjustment for gender, age, Charlson index, diabetes, and arterial hypertension, the risk of rebleeding was more than threefold higher for dual antiplatelet and anticoagulant users than for antiplatelet-alone users (odds ratio, 3.45; P = .025) and was twofold higher for anticoagulant vs. antiplatelet users (OR, 2.07; P = .045), Dr. Lanas said.
Finally, an analysis of the cause of bleeding suggests the cause of rebleeding may be different from the original event and that there is a shift toward the lower GI tract, he added.
The index bleeding event was caused largely by an upper GI peptic ulcer in 48% of all 160 patients, with 43.7% of events due to lower GI diverticulosis, vascular lesions, ischemic, or other lesions. In contrast, peptic ulcers accounted for only 7% of rebleeding events, while lower GI events accounted for 72%. Proton pump inhibition use was evenly distributed in upper and lower GI bleeding, although effective endoscopic treatment may have influenced upper GI bleeds, Dr. Lanas said.
“The importance of this is that we may have very good therapy tools for the upper GI, but still we have problems controlling the bleeding from the lower GI,” he added.
During a discussion of the study, an audience member asked how many days clinicians should wait to restart anticoagulants or antiplatelets.
“In those patients with peptic ulcer bleeding, it’s better to just give the antiplatelet therapy soon after the bleeding event or just to not interrupt the aspirin because the morality at 30 days was higher in those who were interrupted,” Dr. Lanas advised. “...I think for the cutoff point to show differences for patients with a worse outcome versus those with a better outcome, you shouldn’t restart anticoagulant therapy before day 15 after the bleeding event.”
Dr. Lanas received consulting fees, speaking and teaching fees, other financial support, and grant and research support from Bayer.
On Twitter @pwendl
WASHINGTON – Early resumption of antiplatelet agents or anticoagulants after a major gastrointestinal bleeding event is clearly associated with an increased risk of rebleeding, but a decreased risk of death, results from an observational study show.
Furthermore, anticoagulant treatment “is associated with a higher risk of rebleeding and death compared with antiplatelet treatment after a previous GI event,” Dr. Angel Lanas said to an overflow crowd at the annual Digestive Disease Week.
In a separate case-control study, Dr. Lanas and his associates recently reported that the risk of GI bleeding was twofold higher for anticoagulants than for low-dose aspirin in patients hospitalized for GI bleeding (Clin. Gastroenterol. Hepatol. 2015 May;13:906-12.e2. [doi:10.1016/j.cgh.2014.11.007])
The current study examined adverse events in a cohort of 160 patients who developed a major gastrointestinal bleed (GIB) while using anticoagulants and/or antiplatelet therapy between March 2008 and July 2013. Long-term interruption or short-term resumption of these treatments has important clinical implications and differences in the intrinsic risks between antiplatelet or anticoagulant users after drug resumption are not well established, said Dr. Lanas of the University of Zaragoza, Spain.
Drug use information was prospectively collected during the GIB event, with data during the follow-up period obtained from two different Spanish databases.
Treatment during the index bleeding event was continued without interruption in 11 patients and interrupted in 149 patients (93%). Among those whose therapy was interrupted, 21 (14%) never resumed therapy and 128 (86%) resumed therapy (118 patients within 15 days and 10 patients after 15 days). The 86% treatment resumption rate is much higher than the 40%-66% rates reported in previous studies, indicating that Spanish physicians restarted treatment quite early, Dr. Lanas observed.
The mean age at baseline was 76.6 years, 61.3% of patients were men, and half had a Charlson index score > 4. Median follow-up was 21.5 months (range 1-63 months).
Ischemic events did not differ between patients who did or did not restart anticoagulants or antiplatelets (16.4% vs. 14.3%; P value = .806). However, rebleeding occurred in 32% of patients who resumed therapy versus none who did not (P = .002), but deaths were higher in those who did not restart therapy (38.1% vs. 12.5%; P = .003), Dr. Lanas said.
These differences remain significant in Kaplan-Meier survival curves for death (P = .021) and rebleeding (P = .004).
A comparison of early therapy resumption (≤ 15 days) vs. delayed (mean delay 62 days) or no resumption revealed similar results. Early resumption was associated with a higher rate of rebleeding (32.2% vs. 9.7%; P = .012), but a lower rate of death (11% vs. 35.5%; P = .001), with no difference in ischemic events (17% vs. 13%; P = .586), Dr. Lanas said.
Again, the differences remain significant in Kaplan-Meier survival curves for death (P = .011) and rebleeding (P = .013).
When the investigators looked at rebleeding according to drug use, patients receiving anticoagulants vs. antiplatelets had significantly higher rates of rebleeding (34.7% vs. 20.5%; P = .043), death (22.2% vs. 10.2%; P = .038), and any event (68.1% vs. 52.3%; P = .043).
After adjustment for gender, age, Charlson index, diabetes, and arterial hypertension, the risk of rebleeding was more than threefold higher for dual antiplatelet and anticoagulant users than for antiplatelet-alone users (odds ratio, 3.45; P = .025) and was twofold higher for anticoagulant vs. antiplatelet users (OR, 2.07; P = .045), Dr. Lanas said.
Finally, an analysis of the cause of bleeding suggests the cause of rebleeding may be different from the original event and that there is a shift toward the lower GI tract, he added.
The index bleeding event was caused largely by an upper GI peptic ulcer in 48% of all 160 patients, with 43.7% of events due to lower GI diverticulosis, vascular lesions, ischemic, or other lesions. In contrast, peptic ulcers accounted for only 7% of rebleeding events, while lower GI events accounted for 72%. Proton pump inhibition use was evenly distributed in upper and lower GI bleeding, although effective endoscopic treatment may have influenced upper GI bleeds, Dr. Lanas said.
“The importance of this is that we may have very good therapy tools for the upper GI, but still we have problems controlling the bleeding from the lower GI,” he added.
During a discussion of the study, an audience member asked how many days clinicians should wait to restart anticoagulants or antiplatelets.
“In those patients with peptic ulcer bleeding, it’s better to just give the antiplatelet therapy soon after the bleeding event or just to not interrupt the aspirin because the morality at 30 days was higher in those who were interrupted,” Dr. Lanas advised. “...I think for the cutoff point to show differences for patients with a worse outcome versus those with a better outcome, you shouldn’t restart anticoagulant therapy before day 15 after the bleeding event.”
Dr. Lanas received consulting fees, speaking and teaching fees, other financial support, and grant and research support from Bayer.
On Twitter @pwendl
WASHINGTON – Early resumption of antiplatelet agents or anticoagulants after a major gastrointestinal bleeding event is clearly associated with an increased risk of rebleeding, but a decreased risk of death, results from an observational study show.
Furthermore, anticoagulant treatment “is associated with a higher risk of rebleeding and death compared with antiplatelet treatment after a previous GI event,” Dr. Angel Lanas said to an overflow crowd at the annual Digestive Disease Week.
In a separate case-control study, Dr. Lanas and his associates recently reported that the risk of GI bleeding was twofold higher for anticoagulants than for low-dose aspirin in patients hospitalized for GI bleeding (Clin. Gastroenterol. Hepatol. 2015 May;13:906-12.e2. [doi:10.1016/j.cgh.2014.11.007])
The current study examined adverse events in a cohort of 160 patients who developed a major gastrointestinal bleed (GIB) while using anticoagulants and/or antiplatelet therapy between March 2008 and July 2013. Long-term interruption or short-term resumption of these treatments has important clinical implications and differences in the intrinsic risks between antiplatelet or anticoagulant users after drug resumption are not well established, said Dr. Lanas of the University of Zaragoza, Spain.
Drug use information was prospectively collected during the GIB event, with data during the follow-up period obtained from two different Spanish databases.
Treatment during the index bleeding event was continued without interruption in 11 patients and interrupted in 149 patients (93%). Among those whose therapy was interrupted, 21 (14%) never resumed therapy and 128 (86%) resumed therapy (118 patients within 15 days and 10 patients after 15 days). The 86% treatment resumption rate is much higher than the 40%-66% rates reported in previous studies, indicating that Spanish physicians restarted treatment quite early, Dr. Lanas observed.
The mean age at baseline was 76.6 years, 61.3% of patients were men, and half had a Charlson index score > 4. Median follow-up was 21.5 months (range 1-63 months).
Ischemic events did not differ between patients who did or did not restart anticoagulants or antiplatelets (16.4% vs. 14.3%; P value = .806). However, rebleeding occurred in 32% of patients who resumed therapy versus none who did not (P = .002), but deaths were higher in those who did not restart therapy (38.1% vs. 12.5%; P = .003), Dr. Lanas said.
These differences remain significant in Kaplan-Meier survival curves for death (P = .021) and rebleeding (P = .004).
A comparison of early therapy resumption (≤ 15 days) vs. delayed (mean delay 62 days) or no resumption revealed similar results. Early resumption was associated with a higher rate of rebleeding (32.2% vs. 9.7%; P = .012), but a lower rate of death (11% vs. 35.5%; P = .001), with no difference in ischemic events (17% vs. 13%; P = .586), Dr. Lanas said.
Again, the differences remain significant in Kaplan-Meier survival curves for death (P = .011) and rebleeding (P = .013).
When the investigators looked at rebleeding according to drug use, patients receiving anticoagulants vs. antiplatelets had significantly higher rates of rebleeding (34.7% vs. 20.5%; P = .043), death (22.2% vs. 10.2%; P = .038), and any event (68.1% vs. 52.3%; P = .043).
After adjustment for gender, age, Charlson index, diabetes, and arterial hypertension, the risk of rebleeding was more than threefold higher for dual antiplatelet and anticoagulant users than for antiplatelet-alone users (odds ratio, 3.45; P = .025) and was twofold higher for anticoagulant vs. antiplatelet users (OR, 2.07; P = .045), Dr. Lanas said.
Finally, an analysis of the cause of bleeding suggests the cause of rebleeding may be different from the original event and that there is a shift toward the lower GI tract, he added.
The index bleeding event was caused largely by an upper GI peptic ulcer in 48% of all 160 patients, with 43.7% of events due to lower GI diverticulosis, vascular lesions, ischemic, or other lesions. In contrast, peptic ulcers accounted for only 7% of rebleeding events, while lower GI events accounted for 72%. Proton pump inhibition use was evenly distributed in upper and lower GI bleeding, although effective endoscopic treatment may have influenced upper GI bleeds, Dr. Lanas said.
“The importance of this is that we may have very good therapy tools for the upper GI, but still we have problems controlling the bleeding from the lower GI,” he added.
During a discussion of the study, an audience member asked how many days clinicians should wait to restart anticoagulants or antiplatelets.
“In those patients with peptic ulcer bleeding, it’s better to just give the antiplatelet therapy soon after the bleeding event or just to not interrupt the aspirin because the morality at 30 days was higher in those who were interrupted,” Dr. Lanas advised. “...I think for the cutoff point to show differences for patients with a worse outcome versus those with a better outcome, you shouldn’t restart anticoagulant therapy before day 15 after the bleeding event.”
Dr. Lanas received consulting fees, speaking and teaching fees, other financial support, and grant and research support from Bayer.
On Twitter @pwendl
EXPERT ANALYSIS FROM DDW 2015
Key clinical point: Early resumption of antiplatelet agents or anticoagulants after a major gastrointestinal bleeding event is clearly associated with an increased risk of rebleeding, but a decreased risk of death.
Major finding: Rebleeding occurred in 32% of patients who resumed therapy versus none who did not (P = .002), but deaths were higher in those who did not restart therapy (38.1% vs. 12.5%; P = .003).
Data source: Retrospective, observational cohort study of 160 patients who developed GI bleeding while on antiplatelet or anticoagulant therapy.
Disclosures: Dr. Lanas received consulting fees, speaking and teaching fees, other financial support, and grant and research support from Bayer.
A physician who feels hopeless and worthless and complains of pain
CASE Feeling hopeless
Dr. D, age 33, a white, male physician, presents with worsening depression, suicidal ideation, and somatic complaints. Dr. D says his personal life has become increasingly unhappy. He describes the pressures of a busy practice and conflict with his wife about his availability to her. He is feeling financial pressure and general disappointment about practicing medicine. Lack of recreational activities and close friends and absent spiritual life has led to feelings of isolation and depression.
Dr. D reports difficulty falling asleep, waking up early, and feeling fatigued. He describes obsessive, negative thoughts about his work and his personal life; he is anxious and tense. Dissatisfied and exhausted, he says he feels hopeless and empty and has become preoccupied with thoughts of death.
Dr. D describes musculoskeletal tension in the neck, shoulders, and face, with pain in the back of the neck. When the depressive symptoms or pain are particularly severe, he admits that his attention to critical information lapses. When interacting with his patients, he has missed important nuances about medication side effects, for example, frustrating his patients and himself.
Dr. D and his wife do not have children. His mother and paternal grandfather had depression, but Dr. D has no family history of suicide or drug or alcohol abuse. He has no significant medical conditions, and is not taking any medications. Dr. D drinks 1 or 2 cups of caffeinated coffee a day. He does not smoke, use recreational drugs, or drink alcohol regularly.
What would be your next step in treating Dr. D?
a) alert the state medical board about his suicidal ideation
b) recommend inpatient treatment
c) refer Dr. D to a clinician who has experience treating physicians
d) formulate a suicide risk assessment
The authors’ observation
Assessment of the suicidal physician is complex. It requires patience and ability to understand the source and the extent of the physician’s desperation and suffering. Not all psychiatrists are well suited to working with patients who also are peers. An experienced clinician, who has confronted the challenges of practice and treated individuals from many professions, could be better equipped than a recent graduate. Physician− patients might not be forthcoming about the extent of their suicidal thinking, because they fear involuntary hospitalization and jeopardizing their career.1
The evaluating clinician must be thorough and clear, and able to facilitate a trusting relationship. The ill physician should be encouraged to express suicidal ideation freely—without judgments, restrictions, or threats—to a trusted psychiatrist. Questions should be clear without possibility of misinterpretation. Ask:
• “Do you have thoughts of death, dying, or wanting to be dead?”
• “Do you think about suicide?”
• “Do you feel you might act on those thoughts?”
• “What keeps you safe?”
Physicians and other health professional have a higher relative risk of suicide (Table 1).2 Hospitalization should be considered and the decision based on the severity of the illness and the associated risk. Dr. D has several risk factors for suicide, including marital discord, pain, professional demands, and access to lethal means (Table 2).1,3,4
HISTORY Pain and disappointment
After medical school, Dr. D completed residency and joined a large clinic with outpatient and inpatient services. His supervisor was pleased with his work and encouraged him to take on more responsibility. However, within the first years of practice, his mood slowly deteriorated; he came to realize that he was deeply sad and, likely, clinically depressed.
Dr. D describes his parents as detached and emotionally unavailable to him. His mother’s depression sometimes was severe enough that she stayed in her bedroom, isolating herself from her son. Dr. D did not feel close to either of his parents; his mother continued to work despite the depression, which meant that both parents were away from home for long hours. Dr. D became interested in service to others and found that those he served responded to him in a positive way. Service to others became a way to feel recognized, appreciated, respected, and even loved.
Dr. D’s depressive symptoms became worse when he discovered his wife was having an affair. The depression became so debilitating that he requested, and was granted, an 8-week medical leave. Once away from the daily pressures of work, his depression improved somewhat, but conflict with his wife intensified and thoughts of suicide became more frequent. Soon afterward, Dr. D and his wife separated and he moved out. His supervisor recommended that Dr. D obtain treatment, but it was only after the separation that Dr. D decided to seek psychiatric care.
What type of psychotherapy is recommended for physicians with suicidal ideation?
a) psychodynamic psychotherapy
b) person-centered therapy
c) cognitive-behavioral therapy (CBT)
d) dialectical behavior therapy (DBT)
The authors’ observation
Reassure your physician−patients that it is safe and reasonable to take personal time off from work to recover from any illness, whether physical or mental. Consider the best treatment approaches to ensure patient’s safety, comfort, and rapid recovery. A critical part of treatment is exploring and identifying changes needed to achieve a life that is compatible with the ideal self, the patient’s view of himself, his beliefs, goals, and life’s meaning.
Physicians are at particular risk of losing the ideal self.5 Loss of the ideal self is common, and can be life threatening. Person-centered psychotherapy, CBT, supportive psychotherapy, DBT, and pharmacotherapy are used to lessen emotional distress and promote adaptive coping strategies, but approaches are different. Short-term counseling can reduce the effects of job stress,6 but a longer-term intervention likely is necessary for a mood disorder with thoughts of self-harm.
CBT emphasizes helping physicians recognize cognitive distortions and finding solutions. The behavioral aspects of CBT promote physical and mental relaxation, which is helpful in easing muscle tension, lowering heart rate, and decreasing the tendency to hyperventilate during stress.7 Mindfulness-based stress reduction programs can provide physical and mental benefits.8 DBT, a type of behavioral therapy, combines mindfulness, acceptance of the current state, skills to regulate emotion, and positive interpersonal relationship strategies.9
Pharmacotherapy should be focused on improving sleep, anxiety, appetite, and mood. Your patient may have other symptoms that need to be addressed: Ask what symptom bothers your patient the most, then work to provide solutions. Some interventions could promote adaptive coping strategies to identify ways to increase perceived control over the work day.10
TREATMENT Self-exploration
The treatment team instructs Dr. D to take a personal inventory of the elements of his ideal self, along the lines suggested in person-centered therapy.11,12 How did Dr. D envision his practice when he was in residency? What other domains of life were important to him? When Dr. D comes back with his list, the need for change is discussed and the process for incorporating these elements into his life begins. He begins to realize that returning to the elements of his ideal self brought opportunities, friendship, love, and faith back into his life.13,14
Maintaining balance between work responsibilities and pleasurable activities is part of achieving the ideal self. Recreation, social support, and exercise decrease the experience of stress and promote wellness.15,16
An important discussion centers on Dr. D’s risk of losing meaning in life after distancing himself from his original motivation to help people though practicing medicine. Dr. D understands that the distance between his expectations and dreams as a student and his current reality contributed to his depression.17 These conversations and changes in behavior brings Dr. D’s actual life closer to this ideal self, reducing self-discrepancy and lessening negative mood.18
The treating psychiatrist is aware of the reporting requirements to the state medical board, which are discussed with Dr. D. No report is deemed necessary.
The authors’ observation
Dr. D’s treatment course was challenging and required a multi-component approach. Establishing trust, while defining the limits of confidentiality, formed the foundation for the therapeutic relationship. The treatment provider asked for names of colleagues or friends to be contacted in case of an emergency. Dr. D chose his physician supervisor and agreed that the psychiatrist could contact the supervisor and vice versa.
Medication was prescribed at the end of the first session to begin to address anxiety and sleep problems. The initial medication was fluvoxamine, 50 mg/d, for anxiety and depression, clonazepam, 0.5 mg/d for anxiety, and zolpidem, 10 mg/d, for sleep. Adjustments were made in the dosage of antidepressant and responses monitored closely until the therapeutic dosage was reached with minimal side effects. Sleep improved, irritability lessened, and Dr. D’s obsessive, negative thinking and depression improved. Deeper, restorative sleep also began to reduce physical tension and pain. Improved sleep and decreased measures of depression are associated with significantly reduced risk of suicide.19
A treating psychiatrist should be aware of the state medical board requirements. In Ohio, where this case unfolded, reporting is required when the physician−patient is deemed unable to practice medicine according to acceptable and prevailing standards of care.20
Relieving tension and somatic complaints
An important part of the treatment plan consisted of managing chronic muscle tension and pain. We decided to front-load treatment, addressing the severe depression, anxiety, and pain simultaneously. Even moderate pain relief would give Dr. D a greater sense of control and improve his mood.
Dr. D understood that a return to normal biorhythms was necessary to form the foundation for the next step of therapy.21 The treatment team introduced mindful breathing, but Dr. D questioned how something so simple could lift severe depression. Focused, mindful breathing was not a cure, but a first step in regaining control over the current disarray of physical and emotional variations. We encouraged daily practice and he agreed to 5 practice sessions per week.
Next, the treatment team introduced progressive relaxation. Again, the simplicity of this process of tensing and relaxing groups of muscles was met with disbelief. Our therapist explained that voluntarily producing muscle tension facilitates the relaxation response of both the mind and the body. The mind first commands the muscles to do what it does easily— “tense”; then is asked to elicit what is more difficult—“relax.” Repetition of the simple commands “tense—relax” in the arms, legs, back, abdomen, shoulders, neck, and face establishes a calming rhythm, again increasing the sense of control.22 We strongly encouraged daily practice of this exercise and Dr. D committed to the mindful breathing and relaxation exercise.
OUTCOME Recovery, maintenance
Dr. D and his psychotherapist address his anger, all-or-nothing thinking, and loneliness. Grief over his failed marriage was identified, giving them an opportunity to explore this loss and past, perceived losses of his parents’ affection in the context of the therapeutic relationship. Supportive therapy promoted ways to fulfill his ideal self.
Treatment lasted 2 years. Dr. D’s prior depressive episode indicates a need for maintenance medication. The antidepressant is continued and, with help from supportive psychotherapy, stress management, 8 weeks away from work, and the life changes mentioned above, our patient has not had a relapse.
Bottom Line
Depression and thoughts of suicide are common among physicians. Grant time off from work and reassure the physician that he (she) is entitled to seek medical treatment without repercussions. Adapt the type of psychotherapy to the physician’s specific concerns. Because physicians are at particular risk for loss of the ideal self, first consider person-centered therapy.
Related Resources
• Vanderbilt Center for Professional Health. www.mc.vanderbilt.edu/cph.
• Federation of State Physician Health Programs, Inc. www.fsphp.org.
Drug Brand Names
Clonazepam • Klonopin Fluvoxamine • Luvox Zolpidem • Ambien
AcknowledgementThe authors wish to acknowledge the contribution of Rachel Sieke, BS, Research Assistant, Department of Psychiatry, University of Toledo Medical Center, Toledo, Ohio.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Bright RP, Krahn L. Depression and suicide among physicians. Current Psychiatry. 2011;10(4):16-17,25-26,30.
2. Burnett C, Maurer J, Dosemecl M. Mortality by occupation, industry, and cause of death: 24 reporting states (1984-1988). Centers for Disease Control and Prevention. http://www. cdc.gov/niosh/docs/97-114. Published June 1997. Accessed October 3, 2014.
3. Silverman MM. Physicians and suicide. In: Goldman LS, Myers M, Dickstein LJ, eds. The handbook of physician health: essential guide to understanding the health care needs of physicians. Chicago, IL: American Medical Association; 2000:95-117.
4. Lindeman S, Laara E, Hakko H, et al. A systematic review on gender-specific suicide mortality in medical doctors. Br J Psychiatry. 1996;168(3):274-279.
5. Baumeister RF. Suicide as escape from self. Psychol Rev. 1990;97(1):90-113.
6. Rø KE, Gude T, Tyssen R, et al. Counselling for burnout in Norwegian doctors: one year cohort study. BMJ. 2008;337:a2004. doi: 10.1136/bmj.a2004.
7. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
8. Irving JA, Dobkin PL, Park J. Cultivating mindfulness in health care professionals: a review of empirical studies of mindfulness-based stress reduction (MBSR). Complement Ther Clin Pract. 2009;15(2):61-66.
9. Robins C, Schmidt H, Linehan MM. Dialectical behavior therapy synthesizing radical acceptance with skillful means. In: Hayes S, Follette V, Linehan M, eds. Mindfulness and acceptance: expanding the cognitive-behavioral tradition. New York, NY: Guilford Press; 2004:30-44.
10. Dunn PM, Arnetz BB, Christensen JF, et al. Meeting the imperative to improve physician well-being: assessment of an innovative program. J Gen Intern Med. 2007;22(11):1544-1552.
11. Nevid JS, Rathus SA, Greene B. Abnormal psychology in a changing world, 7th ed. Upper Saddle River, NJ: Prentice- Hall; 2008:111-112.
12. Rogers CR. Client-centered therapy. Boston, MA: Houghton Mifflin; 1951.
13. Selimbegovic´ L, Chatard A. The mirror effect: self-awareness alone increases suicide thought accessibility. Conscious Cogn. 2013;22(3):756-764.
14. Cornette M. Staff perspective: self-discrepancy and suicidal ideation. Center for Deployment Psychology. http:// www.deploymentpsych.org/blog/staff-perspective-self-discrepancy-and-suicidal-ideation. Published February 19, 2014. Accessed August 7, 2014.
15. Shanafelt TD, Novotny P, Johnson ME, et al. The well-being and personal wellness promotion strategies of medical oncologists in the North Central Cancer Treatment Group. Oncology. 2005;68(1):23-32.
16. Meldrum H. Exemplary physicians’ strategies for avoiding burnout. Health Care Manag (Frederick). 2010;29(4):324-331.
17. Orbach I, Mikulincer M, Stein D, et al. Self-representation of suicidal adolescents. J Abnorm Psychol. 1998;107(3):435-439.
18. Higgins ET. Self-discrepancy: a theory related self and affect. Psychol Rev. 1987;94(3):319-340.
19. Christensen H, Batterham PJ, Mackinnon AJ, et al. Predictors of the risk factors for suicide identified by the interpersonal-psychological theory of suicidal behaviour. Psychiatry Res. 2014;219(2):290-297.
20. Ohio State Medical Board. Section 4731.22 (B), Rule 4731-18- 01. 2014.
21. McGrady A, Moss D. Pathways to illness, pathways to health. New York, NY: Springer; 2013.
22. Davis M, Eshelman ER, McKay M. The relaxation and stress reduction workbook, 6th ed. Oakland, CA: New Harbinger Publications, Inc; 2008.
CASE Feeling hopeless
Dr. D, age 33, a white, male physician, presents with worsening depression, suicidal ideation, and somatic complaints. Dr. D says his personal life has become increasingly unhappy. He describes the pressures of a busy practice and conflict with his wife about his availability to her. He is feeling financial pressure and general disappointment about practicing medicine. Lack of recreational activities and close friends and absent spiritual life has led to feelings of isolation and depression.
Dr. D reports difficulty falling asleep, waking up early, and feeling fatigued. He describes obsessive, negative thoughts about his work and his personal life; he is anxious and tense. Dissatisfied and exhausted, he says he feels hopeless and empty and has become preoccupied with thoughts of death.
Dr. D describes musculoskeletal tension in the neck, shoulders, and face, with pain in the back of the neck. When the depressive symptoms or pain are particularly severe, he admits that his attention to critical information lapses. When interacting with his patients, he has missed important nuances about medication side effects, for example, frustrating his patients and himself.
Dr. D and his wife do not have children. His mother and paternal grandfather had depression, but Dr. D has no family history of suicide or drug or alcohol abuse. He has no significant medical conditions, and is not taking any medications. Dr. D drinks 1 or 2 cups of caffeinated coffee a day. He does not smoke, use recreational drugs, or drink alcohol regularly.
What would be your next step in treating Dr. D?
a) alert the state medical board about his suicidal ideation
b) recommend inpatient treatment
c) refer Dr. D to a clinician who has experience treating physicians
d) formulate a suicide risk assessment
The authors’ observation
Assessment of the suicidal physician is complex. It requires patience and ability to understand the source and the extent of the physician’s desperation and suffering. Not all psychiatrists are well suited to working with patients who also are peers. An experienced clinician, who has confronted the challenges of practice and treated individuals from many professions, could be better equipped than a recent graduate. Physician− patients might not be forthcoming about the extent of their suicidal thinking, because they fear involuntary hospitalization and jeopardizing their career.1
The evaluating clinician must be thorough and clear, and able to facilitate a trusting relationship. The ill physician should be encouraged to express suicidal ideation freely—without judgments, restrictions, or threats—to a trusted psychiatrist. Questions should be clear without possibility of misinterpretation. Ask:
• “Do you have thoughts of death, dying, or wanting to be dead?”
• “Do you think about suicide?”
• “Do you feel you might act on those thoughts?”
• “What keeps you safe?”
Physicians and other health professional have a higher relative risk of suicide (Table 1).2 Hospitalization should be considered and the decision based on the severity of the illness and the associated risk. Dr. D has several risk factors for suicide, including marital discord, pain, professional demands, and access to lethal means (Table 2).1,3,4
HISTORY Pain and disappointment
After medical school, Dr. D completed residency and joined a large clinic with outpatient and inpatient services. His supervisor was pleased with his work and encouraged him to take on more responsibility. However, within the first years of practice, his mood slowly deteriorated; he came to realize that he was deeply sad and, likely, clinically depressed.
Dr. D describes his parents as detached and emotionally unavailable to him. His mother’s depression sometimes was severe enough that she stayed in her bedroom, isolating herself from her son. Dr. D did not feel close to either of his parents; his mother continued to work despite the depression, which meant that both parents were away from home for long hours. Dr. D became interested in service to others and found that those he served responded to him in a positive way. Service to others became a way to feel recognized, appreciated, respected, and even loved.
Dr. D’s depressive symptoms became worse when he discovered his wife was having an affair. The depression became so debilitating that he requested, and was granted, an 8-week medical leave. Once away from the daily pressures of work, his depression improved somewhat, but conflict with his wife intensified and thoughts of suicide became more frequent. Soon afterward, Dr. D and his wife separated and he moved out. His supervisor recommended that Dr. D obtain treatment, but it was only after the separation that Dr. D decided to seek psychiatric care.
What type of psychotherapy is recommended for physicians with suicidal ideation?
a) psychodynamic psychotherapy
b) person-centered therapy
c) cognitive-behavioral therapy (CBT)
d) dialectical behavior therapy (DBT)
The authors’ observation
Reassure your physician−patients that it is safe and reasonable to take personal time off from work to recover from any illness, whether physical or mental. Consider the best treatment approaches to ensure patient’s safety, comfort, and rapid recovery. A critical part of treatment is exploring and identifying changes needed to achieve a life that is compatible with the ideal self, the patient’s view of himself, his beliefs, goals, and life’s meaning.
Physicians are at particular risk of losing the ideal self.5 Loss of the ideal self is common, and can be life threatening. Person-centered psychotherapy, CBT, supportive psychotherapy, DBT, and pharmacotherapy are used to lessen emotional distress and promote adaptive coping strategies, but approaches are different. Short-term counseling can reduce the effects of job stress,6 but a longer-term intervention likely is necessary for a mood disorder with thoughts of self-harm.
CBT emphasizes helping physicians recognize cognitive distortions and finding solutions. The behavioral aspects of CBT promote physical and mental relaxation, which is helpful in easing muscle tension, lowering heart rate, and decreasing the tendency to hyperventilate during stress.7 Mindfulness-based stress reduction programs can provide physical and mental benefits.8 DBT, a type of behavioral therapy, combines mindfulness, acceptance of the current state, skills to regulate emotion, and positive interpersonal relationship strategies.9
Pharmacotherapy should be focused on improving sleep, anxiety, appetite, and mood. Your patient may have other symptoms that need to be addressed: Ask what symptom bothers your patient the most, then work to provide solutions. Some interventions could promote adaptive coping strategies to identify ways to increase perceived control over the work day.10
TREATMENT Self-exploration
The treatment team instructs Dr. D to take a personal inventory of the elements of his ideal self, along the lines suggested in person-centered therapy.11,12 How did Dr. D envision his practice when he was in residency? What other domains of life were important to him? When Dr. D comes back with his list, the need for change is discussed and the process for incorporating these elements into his life begins. He begins to realize that returning to the elements of his ideal self brought opportunities, friendship, love, and faith back into his life.13,14
Maintaining balance between work responsibilities and pleasurable activities is part of achieving the ideal self. Recreation, social support, and exercise decrease the experience of stress and promote wellness.15,16
An important discussion centers on Dr. D’s risk of losing meaning in life after distancing himself from his original motivation to help people though practicing medicine. Dr. D understands that the distance between his expectations and dreams as a student and his current reality contributed to his depression.17 These conversations and changes in behavior brings Dr. D’s actual life closer to this ideal self, reducing self-discrepancy and lessening negative mood.18
The treating psychiatrist is aware of the reporting requirements to the state medical board, which are discussed with Dr. D. No report is deemed necessary.
The authors’ observation
Dr. D’s treatment course was challenging and required a multi-component approach. Establishing trust, while defining the limits of confidentiality, formed the foundation for the therapeutic relationship. The treatment provider asked for names of colleagues or friends to be contacted in case of an emergency. Dr. D chose his physician supervisor and agreed that the psychiatrist could contact the supervisor and vice versa.
Medication was prescribed at the end of the first session to begin to address anxiety and sleep problems. The initial medication was fluvoxamine, 50 mg/d, for anxiety and depression, clonazepam, 0.5 mg/d for anxiety, and zolpidem, 10 mg/d, for sleep. Adjustments were made in the dosage of antidepressant and responses monitored closely until the therapeutic dosage was reached with minimal side effects. Sleep improved, irritability lessened, and Dr. D’s obsessive, negative thinking and depression improved. Deeper, restorative sleep also began to reduce physical tension and pain. Improved sleep and decreased measures of depression are associated with significantly reduced risk of suicide.19
A treating psychiatrist should be aware of the state medical board requirements. In Ohio, where this case unfolded, reporting is required when the physician−patient is deemed unable to practice medicine according to acceptable and prevailing standards of care.20
Relieving tension and somatic complaints
An important part of the treatment plan consisted of managing chronic muscle tension and pain. We decided to front-load treatment, addressing the severe depression, anxiety, and pain simultaneously. Even moderate pain relief would give Dr. D a greater sense of control and improve his mood.
Dr. D understood that a return to normal biorhythms was necessary to form the foundation for the next step of therapy.21 The treatment team introduced mindful breathing, but Dr. D questioned how something so simple could lift severe depression. Focused, mindful breathing was not a cure, but a first step in regaining control over the current disarray of physical and emotional variations. We encouraged daily practice and he agreed to 5 practice sessions per week.
Next, the treatment team introduced progressive relaxation. Again, the simplicity of this process of tensing and relaxing groups of muscles was met with disbelief. Our therapist explained that voluntarily producing muscle tension facilitates the relaxation response of both the mind and the body. The mind first commands the muscles to do what it does easily— “tense”; then is asked to elicit what is more difficult—“relax.” Repetition of the simple commands “tense—relax” in the arms, legs, back, abdomen, shoulders, neck, and face establishes a calming rhythm, again increasing the sense of control.22 We strongly encouraged daily practice of this exercise and Dr. D committed to the mindful breathing and relaxation exercise.
OUTCOME Recovery, maintenance
Dr. D and his psychotherapist address his anger, all-or-nothing thinking, and loneliness. Grief over his failed marriage was identified, giving them an opportunity to explore this loss and past, perceived losses of his parents’ affection in the context of the therapeutic relationship. Supportive therapy promoted ways to fulfill his ideal self.
Treatment lasted 2 years. Dr. D’s prior depressive episode indicates a need for maintenance medication. The antidepressant is continued and, with help from supportive psychotherapy, stress management, 8 weeks away from work, and the life changes mentioned above, our patient has not had a relapse.
Bottom Line
Depression and thoughts of suicide are common among physicians. Grant time off from work and reassure the physician that he (she) is entitled to seek medical treatment without repercussions. Adapt the type of psychotherapy to the physician’s specific concerns. Because physicians are at particular risk for loss of the ideal self, first consider person-centered therapy.
Related Resources
• Vanderbilt Center for Professional Health. www.mc.vanderbilt.edu/cph.
• Federation of State Physician Health Programs, Inc. www.fsphp.org.
Drug Brand Names
Clonazepam • Klonopin Fluvoxamine • Luvox Zolpidem • Ambien
AcknowledgementThe authors wish to acknowledge the contribution of Rachel Sieke, BS, Research Assistant, Department of Psychiatry, University of Toledo Medical Center, Toledo, Ohio.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
CASE Feeling hopeless
Dr. D, age 33, a white, male physician, presents with worsening depression, suicidal ideation, and somatic complaints. Dr. D says his personal life has become increasingly unhappy. He describes the pressures of a busy practice and conflict with his wife about his availability to her. He is feeling financial pressure and general disappointment about practicing medicine. Lack of recreational activities and close friends and absent spiritual life has led to feelings of isolation and depression.
Dr. D reports difficulty falling asleep, waking up early, and feeling fatigued. He describes obsessive, negative thoughts about his work and his personal life; he is anxious and tense. Dissatisfied and exhausted, he says he feels hopeless and empty and has become preoccupied with thoughts of death.
Dr. D describes musculoskeletal tension in the neck, shoulders, and face, with pain in the back of the neck. When the depressive symptoms or pain are particularly severe, he admits that his attention to critical information lapses. When interacting with his patients, he has missed important nuances about medication side effects, for example, frustrating his patients and himself.
Dr. D and his wife do not have children. His mother and paternal grandfather had depression, but Dr. D has no family history of suicide or drug or alcohol abuse. He has no significant medical conditions, and is not taking any medications. Dr. D drinks 1 or 2 cups of caffeinated coffee a day. He does not smoke, use recreational drugs, or drink alcohol regularly.
What would be your next step in treating Dr. D?
a) alert the state medical board about his suicidal ideation
b) recommend inpatient treatment
c) refer Dr. D to a clinician who has experience treating physicians
d) formulate a suicide risk assessment
The authors’ observation
Assessment of the suicidal physician is complex. It requires patience and ability to understand the source and the extent of the physician’s desperation and suffering. Not all psychiatrists are well suited to working with patients who also are peers. An experienced clinician, who has confronted the challenges of practice and treated individuals from many professions, could be better equipped than a recent graduate. Physician− patients might not be forthcoming about the extent of their suicidal thinking, because they fear involuntary hospitalization and jeopardizing their career.1
The evaluating clinician must be thorough and clear, and able to facilitate a trusting relationship. The ill physician should be encouraged to express suicidal ideation freely—without judgments, restrictions, or threats—to a trusted psychiatrist. Questions should be clear without possibility of misinterpretation. Ask:
• “Do you have thoughts of death, dying, or wanting to be dead?”
• “Do you think about suicide?”
• “Do you feel you might act on those thoughts?”
• “What keeps you safe?”
Physicians and other health professional have a higher relative risk of suicide (Table 1).2 Hospitalization should be considered and the decision based on the severity of the illness and the associated risk. Dr. D has several risk factors for suicide, including marital discord, pain, professional demands, and access to lethal means (Table 2).1,3,4
HISTORY Pain and disappointment
After medical school, Dr. D completed residency and joined a large clinic with outpatient and inpatient services. His supervisor was pleased with his work and encouraged him to take on more responsibility. However, within the first years of practice, his mood slowly deteriorated; he came to realize that he was deeply sad and, likely, clinically depressed.
Dr. D describes his parents as detached and emotionally unavailable to him. His mother’s depression sometimes was severe enough that she stayed in her bedroom, isolating herself from her son. Dr. D did not feel close to either of his parents; his mother continued to work despite the depression, which meant that both parents were away from home for long hours. Dr. D became interested in service to others and found that those he served responded to him in a positive way. Service to others became a way to feel recognized, appreciated, respected, and even loved.
Dr. D’s depressive symptoms became worse when he discovered his wife was having an affair. The depression became so debilitating that he requested, and was granted, an 8-week medical leave. Once away from the daily pressures of work, his depression improved somewhat, but conflict with his wife intensified and thoughts of suicide became more frequent. Soon afterward, Dr. D and his wife separated and he moved out. His supervisor recommended that Dr. D obtain treatment, but it was only after the separation that Dr. D decided to seek psychiatric care.
What type of psychotherapy is recommended for physicians with suicidal ideation?
a) psychodynamic psychotherapy
b) person-centered therapy
c) cognitive-behavioral therapy (CBT)
d) dialectical behavior therapy (DBT)
The authors’ observation
Reassure your physician−patients that it is safe and reasonable to take personal time off from work to recover from any illness, whether physical or mental. Consider the best treatment approaches to ensure patient’s safety, comfort, and rapid recovery. A critical part of treatment is exploring and identifying changes needed to achieve a life that is compatible with the ideal self, the patient’s view of himself, his beliefs, goals, and life’s meaning.
Physicians are at particular risk of losing the ideal self.5 Loss of the ideal self is common, and can be life threatening. Person-centered psychotherapy, CBT, supportive psychotherapy, DBT, and pharmacotherapy are used to lessen emotional distress and promote adaptive coping strategies, but approaches are different. Short-term counseling can reduce the effects of job stress,6 but a longer-term intervention likely is necessary for a mood disorder with thoughts of self-harm.
CBT emphasizes helping physicians recognize cognitive distortions and finding solutions. The behavioral aspects of CBT promote physical and mental relaxation, which is helpful in easing muscle tension, lowering heart rate, and decreasing the tendency to hyperventilate during stress.7 Mindfulness-based stress reduction programs can provide physical and mental benefits.8 DBT, a type of behavioral therapy, combines mindfulness, acceptance of the current state, skills to regulate emotion, and positive interpersonal relationship strategies.9
Pharmacotherapy should be focused on improving sleep, anxiety, appetite, and mood. Your patient may have other symptoms that need to be addressed: Ask what symptom bothers your patient the most, then work to provide solutions. Some interventions could promote adaptive coping strategies to identify ways to increase perceived control over the work day.10
TREATMENT Self-exploration
The treatment team instructs Dr. D to take a personal inventory of the elements of his ideal self, along the lines suggested in person-centered therapy.11,12 How did Dr. D envision his practice when he was in residency? What other domains of life were important to him? When Dr. D comes back with his list, the need for change is discussed and the process for incorporating these elements into his life begins. He begins to realize that returning to the elements of his ideal self brought opportunities, friendship, love, and faith back into his life.13,14
Maintaining balance between work responsibilities and pleasurable activities is part of achieving the ideal self. Recreation, social support, and exercise decrease the experience of stress and promote wellness.15,16
An important discussion centers on Dr. D’s risk of losing meaning in life after distancing himself from his original motivation to help people though practicing medicine. Dr. D understands that the distance between his expectations and dreams as a student and his current reality contributed to his depression.17 These conversations and changes in behavior brings Dr. D’s actual life closer to this ideal self, reducing self-discrepancy and lessening negative mood.18
The treating psychiatrist is aware of the reporting requirements to the state medical board, which are discussed with Dr. D. No report is deemed necessary.
The authors’ observation
Dr. D’s treatment course was challenging and required a multi-component approach. Establishing trust, while defining the limits of confidentiality, formed the foundation for the therapeutic relationship. The treatment provider asked for names of colleagues or friends to be contacted in case of an emergency. Dr. D chose his physician supervisor and agreed that the psychiatrist could contact the supervisor and vice versa.
Medication was prescribed at the end of the first session to begin to address anxiety and sleep problems. The initial medication was fluvoxamine, 50 mg/d, for anxiety and depression, clonazepam, 0.5 mg/d for anxiety, and zolpidem, 10 mg/d, for sleep. Adjustments were made in the dosage of antidepressant and responses monitored closely until the therapeutic dosage was reached with minimal side effects. Sleep improved, irritability lessened, and Dr. D’s obsessive, negative thinking and depression improved. Deeper, restorative sleep also began to reduce physical tension and pain. Improved sleep and decreased measures of depression are associated with significantly reduced risk of suicide.19
A treating psychiatrist should be aware of the state medical board requirements. In Ohio, where this case unfolded, reporting is required when the physician−patient is deemed unable to practice medicine according to acceptable and prevailing standards of care.20
Relieving tension and somatic complaints
An important part of the treatment plan consisted of managing chronic muscle tension and pain. We decided to front-load treatment, addressing the severe depression, anxiety, and pain simultaneously. Even moderate pain relief would give Dr. D a greater sense of control and improve his mood.
Dr. D understood that a return to normal biorhythms was necessary to form the foundation for the next step of therapy.21 The treatment team introduced mindful breathing, but Dr. D questioned how something so simple could lift severe depression. Focused, mindful breathing was not a cure, but a first step in regaining control over the current disarray of physical and emotional variations. We encouraged daily practice and he agreed to 5 practice sessions per week.
Next, the treatment team introduced progressive relaxation. Again, the simplicity of this process of tensing and relaxing groups of muscles was met with disbelief. Our therapist explained that voluntarily producing muscle tension facilitates the relaxation response of both the mind and the body. The mind first commands the muscles to do what it does easily— “tense”; then is asked to elicit what is more difficult—“relax.” Repetition of the simple commands “tense—relax” in the arms, legs, back, abdomen, shoulders, neck, and face establishes a calming rhythm, again increasing the sense of control.22 We strongly encouraged daily practice of this exercise and Dr. D committed to the mindful breathing and relaxation exercise.
OUTCOME Recovery, maintenance
Dr. D and his psychotherapist address his anger, all-or-nothing thinking, and loneliness. Grief over his failed marriage was identified, giving them an opportunity to explore this loss and past, perceived losses of his parents’ affection in the context of the therapeutic relationship. Supportive therapy promoted ways to fulfill his ideal self.
Treatment lasted 2 years. Dr. D’s prior depressive episode indicates a need for maintenance medication. The antidepressant is continued and, with help from supportive psychotherapy, stress management, 8 weeks away from work, and the life changes mentioned above, our patient has not had a relapse.
Bottom Line
Depression and thoughts of suicide are common among physicians. Grant time off from work and reassure the physician that he (she) is entitled to seek medical treatment without repercussions. Adapt the type of psychotherapy to the physician’s specific concerns. Because physicians are at particular risk for loss of the ideal self, first consider person-centered therapy.
Related Resources
• Vanderbilt Center for Professional Health. www.mc.vanderbilt.edu/cph.
• Federation of State Physician Health Programs, Inc. www.fsphp.org.
Drug Brand Names
Clonazepam • Klonopin Fluvoxamine • Luvox Zolpidem • Ambien
AcknowledgementThe authors wish to acknowledge the contribution of Rachel Sieke, BS, Research Assistant, Department of Psychiatry, University of Toledo Medical Center, Toledo, Ohio.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Bright RP, Krahn L. Depression and suicide among physicians. Current Psychiatry. 2011;10(4):16-17,25-26,30.
2. Burnett C, Maurer J, Dosemecl M. Mortality by occupation, industry, and cause of death: 24 reporting states (1984-1988). Centers for Disease Control and Prevention. http://www. cdc.gov/niosh/docs/97-114. Published June 1997. Accessed October 3, 2014.
3. Silverman MM. Physicians and suicide. In: Goldman LS, Myers M, Dickstein LJ, eds. The handbook of physician health: essential guide to understanding the health care needs of physicians. Chicago, IL: American Medical Association; 2000:95-117.
4. Lindeman S, Laara E, Hakko H, et al. A systematic review on gender-specific suicide mortality in medical doctors. Br J Psychiatry. 1996;168(3):274-279.
5. Baumeister RF. Suicide as escape from self. Psychol Rev. 1990;97(1):90-113.
6. Rø KE, Gude T, Tyssen R, et al. Counselling for burnout in Norwegian doctors: one year cohort study. BMJ. 2008;337:a2004. doi: 10.1136/bmj.a2004.
7. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
8. Irving JA, Dobkin PL, Park J. Cultivating mindfulness in health care professionals: a review of empirical studies of mindfulness-based stress reduction (MBSR). Complement Ther Clin Pract. 2009;15(2):61-66.
9. Robins C, Schmidt H, Linehan MM. Dialectical behavior therapy synthesizing radical acceptance with skillful means. In: Hayes S, Follette V, Linehan M, eds. Mindfulness and acceptance: expanding the cognitive-behavioral tradition. New York, NY: Guilford Press; 2004:30-44.
10. Dunn PM, Arnetz BB, Christensen JF, et al. Meeting the imperative to improve physician well-being: assessment of an innovative program. J Gen Intern Med. 2007;22(11):1544-1552.
11. Nevid JS, Rathus SA, Greene B. Abnormal psychology in a changing world, 7th ed. Upper Saddle River, NJ: Prentice- Hall; 2008:111-112.
12. Rogers CR. Client-centered therapy. Boston, MA: Houghton Mifflin; 1951.
13. Selimbegovic´ L, Chatard A. The mirror effect: self-awareness alone increases suicide thought accessibility. Conscious Cogn. 2013;22(3):756-764.
14. Cornette M. Staff perspective: self-discrepancy and suicidal ideation. Center for Deployment Psychology. http:// www.deploymentpsych.org/blog/staff-perspective-self-discrepancy-and-suicidal-ideation. Published February 19, 2014. Accessed August 7, 2014.
15. Shanafelt TD, Novotny P, Johnson ME, et al. The well-being and personal wellness promotion strategies of medical oncologists in the North Central Cancer Treatment Group. Oncology. 2005;68(1):23-32.
16. Meldrum H. Exemplary physicians’ strategies for avoiding burnout. Health Care Manag (Frederick). 2010;29(4):324-331.
17. Orbach I, Mikulincer M, Stein D, et al. Self-representation of suicidal adolescents. J Abnorm Psychol. 1998;107(3):435-439.
18. Higgins ET. Self-discrepancy: a theory related self and affect. Psychol Rev. 1987;94(3):319-340.
19. Christensen H, Batterham PJ, Mackinnon AJ, et al. Predictors of the risk factors for suicide identified by the interpersonal-psychological theory of suicidal behaviour. Psychiatry Res. 2014;219(2):290-297.
20. Ohio State Medical Board. Section 4731.22 (B), Rule 4731-18- 01. 2014.
21. McGrady A, Moss D. Pathways to illness, pathways to health. New York, NY: Springer; 2013.
22. Davis M, Eshelman ER, McKay M. The relaxation and stress reduction workbook, 6th ed. Oakland, CA: New Harbinger Publications, Inc; 2008.
1. Bright RP, Krahn L. Depression and suicide among physicians. Current Psychiatry. 2011;10(4):16-17,25-26,30.
2. Burnett C, Maurer J, Dosemecl M. Mortality by occupation, industry, and cause of death: 24 reporting states (1984-1988). Centers for Disease Control and Prevention. http://www. cdc.gov/niosh/docs/97-114. Published June 1997. Accessed October 3, 2014.
3. Silverman MM. Physicians and suicide. In: Goldman LS, Myers M, Dickstein LJ, eds. The handbook of physician health: essential guide to understanding the health care needs of physicians. Chicago, IL: American Medical Association; 2000:95-117.
4. Lindeman S, Laara E, Hakko H, et al. A systematic review on gender-specific suicide mortality in medical doctors. Br J Psychiatry. 1996;168(3):274-279.
5. Baumeister RF. Suicide as escape from self. Psychol Rev. 1990;97(1):90-113.
6. Rø KE, Gude T, Tyssen R, et al. Counselling for burnout in Norwegian doctors: one year cohort study. BMJ. 2008;337:a2004. doi: 10.1136/bmj.a2004.
7. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
8. Irving JA, Dobkin PL, Park J. Cultivating mindfulness in health care professionals: a review of empirical studies of mindfulness-based stress reduction (MBSR). Complement Ther Clin Pract. 2009;15(2):61-66.
9. Robins C, Schmidt H, Linehan MM. Dialectical behavior therapy synthesizing radical acceptance with skillful means. In: Hayes S, Follette V, Linehan M, eds. Mindfulness and acceptance: expanding the cognitive-behavioral tradition. New York, NY: Guilford Press; 2004:30-44.
10. Dunn PM, Arnetz BB, Christensen JF, et al. Meeting the imperative to improve physician well-being: assessment of an innovative program. J Gen Intern Med. 2007;22(11):1544-1552.
11. Nevid JS, Rathus SA, Greene B. Abnormal psychology in a changing world, 7th ed. Upper Saddle River, NJ: Prentice- Hall; 2008:111-112.
12. Rogers CR. Client-centered therapy. Boston, MA: Houghton Mifflin; 1951.
13. Selimbegovic´ L, Chatard A. The mirror effect: self-awareness alone increases suicide thought accessibility. Conscious Cogn. 2013;22(3):756-764.
14. Cornette M. Staff perspective: self-discrepancy and suicidal ideation. Center for Deployment Psychology. http:// www.deploymentpsych.org/blog/staff-perspective-self-discrepancy-and-suicidal-ideation. Published February 19, 2014. Accessed August 7, 2014.
15. Shanafelt TD, Novotny P, Johnson ME, et al. The well-being and personal wellness promotion strategies of medical oncologists in the North Central Cancer Treatment Group. Oncology. 2005;68(1):23-32.
16. Meldrum H. Exemplary physicians’ strategies for avoiding burnout. Health Care Manag (Frederick). 2010;29(4):324-331.
17. Orbach I, Mikulincer M, Stein D, et al. Self-representation of suicidal adolescents. J Abnorm Psychol. 1998;107(3):435-439.
18. Higgins ET. Self-discrepancy: a theory related self and affect. Psychol Rev. 1987;94(3):319-340.
19. Christensen H, Batterham PJ, Mackinnon AJ, et al. Predictors of the risk factors for suicide identified by the interpersonal-psychological theory of suicidal behaviour. Psychiatry Res. 2014;219(2):290-297.
20. Ohio State Medical Board. Section 4731.22 (B), Rule 4731-18- 01. 2014.
21. McGrady A, Moss D. Pathways to illness, pathways to health. New York, NY: Springer; 2013.
22. Davis M, Eshelman ER, McKay M. The relaxation and stress reduction workbook, 6th ed. Oakland, CA: New Harbinger Publications, Inc; 2008.
Newer oral contraceptives pose higher VTE risk
The risk of developing venous thromboembolism is generally greater for women using oral contraceptives with newer types of progestogen hormones than for those taking older, second-generation birth control pills, study results showed.
“Women exposed to drospirenone, gestodene, cyproterone, and desogestrel within the last 28 days had around a four times increased risk of venous thromboembolism,” the investigators found. Women exposed to levonorgestrel, norethisterone, and norgestimate had about a 2.5 times greater risk of venous thromboembolism than did women not exposed in the past year, said Yana Vinogradova and her colleagues at the University of Nottingham (England) (BMJ. 2015 May 26 [doi:10.1136/bmj.h2135]).
The researchers conducted two nested case-control studies using data from 618 primary care practices in the Clinical Practice Research Datalink (CPRD) and 722 practices in the QResearch primary care database. A total of 5,062 cases from CPRD and 5,500 cases from QResearch were matched one to five with 19,638 and 22,396 controls, respectively.
Approximately 29% of CPRD patients and 26% of QResearch patients used oral contraceptives, most commonly levonorgestrel. Overall, any use of combined oral contraceptives resulted in a three times increased risk for venous thromboembolism, compared with no use in the past year.
After accounting for smoking, obesity, a wide range of other health conditions, alcohol consumption, polycystic ovary syndrome and recent infections, surgeries, leg/hip fractures, and hospital admission, the researchers calculated an increased odds ratio for each hormone: desogestrel (4.28), cyproterone (4.27), drospirenone (4.12), gestodene (3.64), levonorgestrel (2.38), norgestimate (2.53), and norethisterone (2.56). The increased VTE risk in patients on these hormones was compared with no exposure to oral contraceptives in the previous year.
In terms of numbers needed to harm, the researchers estimated that use of levonorgestrel and norgestimate resulted in 6 extra cases of VTE each year per 10,000 treated women aged 15-49, and 7 extra cases for women aged 25-49.
Desogestrel and cyproterone each contributed 14 additional cases of VTE each year per 10,000 treated women aged 15-49, and drospirenone, desogestrel, and cyproterone each contributed to an extra 17 cases of VTE each year per 10,000 women aged 25-49.
“We believe this study has the statistical power and sufficient adjustment for relevant confounders to be regarded as an important clarifying study, which has produced the most reliable possible risk estimates using currently available U.K. prescription data,” the researchers wrote.
There was no external funding for the study. Julia Hippisley-Cox is the unpaid director of QResearch, a not-for-profit organization that is a joint partnership between the University of Nottingham and EMIS, a commercial IT supplier. She is also a paid director of ClinRisk, which produces clinical risk algorithm-related software.
The risk of developing venous thromboembolism is generally greater for women using oral contraceptives with newer types of progestogen hormones than for those taking older, second-generation birth control pills, study results showed.
“Women exposed to drospirenone, gestodene, cyproterone, and desogestrel within the last 28 days had around a four times increased risk of venous thromboembolism,” the investigators found. Women exposed to levonorgestrel, norethisterone, and norgestimate had about a 2.5 times greater risk of venous thromboembolism than did women not exposed in the past year, said Yana Vinogradova and her colleagues at the University of Nottingham (England) (BMJ. 2015 May 26 [doi:10.1136/bmj.h2135]).
The researchers conducted two nested case-control studies using data from 618 primary care practices in the Clinical Practice Research Datalink (CPRD) and 722 practices in the QResearch primary care database. A total of 5,062 cases from CPRD and 5,500 cases from QResearch were matched one to five with 19,638 and 22,396 controls, respectively.
Approximately 29% of CPRD patients and 26% of QResearch patients used oral contraceptives, most commonly levonorgestrel. Overall, any use of combined oral contraceptives resulted in a three times increased risk for venous thromboembolism, compared with no use in the past year.
After accounting for smoking, obesity, a wide range of other health conditions, alcohol consumption, polycystic ovary syndrome and recent infections, surgeries, leg/hip fractures, and hospital admission, the researchers calculated an increased odds ratio for each hormone: desogestrel (4.28), cyproterone (4.27), drospirenone (4.12), gestodene (3.64), levonorgestrel (2.38), norgestimate (2.53), and norethisterone (2.56). The increased VTE risk in patients on these hormones was compared with no exposure to oral contraceptives in the previous year.
In terms of numbers needed to harm, the researchers estimated that use of levonorgestrel and norgestimate resulted in 6 extra cases of VTE each year per 10,000 treated women aged 15-49, and 7 extra cases for women aged 25-49.
Desogestrel and cyproterone each contributed 14 additional cases of VTE each year per 10,000 treated women aged 15-49, and drospirenone, desogestrel, and cyproterone each contributed to an extra 17 cases of VTE each year per 10,000 women aged 25-49.
“We believe this study has the statistical power and sufficient adjustment for relevant confounders to be regarded as an important clarifying study, which has produced the most reliable possible risk estimates using currently available U.K. prescription data,” the researchers wrote.
There was no external funding for the study. Julia Hippisley-Cox is the unpaid director of QResearch, a not-for-profit organization that is a joint partnership between the University of Nottingham and EMIS, a commercial IT supplier. She is also a paid director of ClinRisk, which produces clinical risk algorithm-related software.
The risk of developing venous thromboembolism is generally greater for women using oral contraceptives with newer types of progestogen hormones than for those taking older, second-generation birth control pills, study results showed.
“Women exposed to drospirenone, gestodene, cyproterone, and desogestrel within the last 28 days had around a four times increased risk of venous thromboembolism,” the investigators found. Women exposed to levonorgestrel, norethisterone, and norgestimate had about a 2.5 times greater risk of venous thromboembolism than did women not exposed in the past year, said Yana Vinogradova and her colleagues at the University of Nottingham (England) (BMJ. 2015 May 26 [doi:10.1136/bmj.h2135]).
The researchers conducted two nested case-control studies using data from 618 primary care practices in the Clinical Practice Research Datalink (CPRD) and 722 practices in the QResearch primary care database. A total of 5,062 cases from CPRD and 5,500 cases from QResearch were matched one to five with 19,638 and 22,396 controls, respectively.
Approximately 29% of CPRD patients and 26% of QResearch patients used oral contraceptives, most commonly levonorgestrel. Overall, any use of combined oral contraceptives resulted in a three times increased risk for venous thromboembolism, compared with no use in the past year.
After accounting for smoking, obesity, a wide range of other health conditions, alcohol consumption, polycystic ovary syndrome and recent infections, surgeries, leg/hip fractures, and hospital admission, the researchers calculated an increased odds ratio for each hormone: desogestrel (4.28), cyproterone (4.27), drospirenone (4.12), gestodene (3.64), levonorgestrel (2.38), norgestimate (2.53), and norethisterone (2.56). The increased VTE risk in patients on these hormones was compared with no exposure to oral contraceptives in the previous year.
In terms of numbers needed to harm, the researchers estimated that use of levonorgestrel and norgestimate resulted in 6 extra cases of VTE each year per 10,000 treated women aged 15-49, and 7 extra cases for women aged 25-49.
Desogestrel and cyproterone each contributed 14 additional cases of VTE each year per 10,000 treated women aged 15-49, and drospirenone, desogestrel, and cyproterone each contributed to an extra 17 cases of VTE each year per 10,000 women aged 25-49.
“We believe this study has the statistical power and sufficient adjustment for relevant confounders to be regarded as an important clarifying study, which has produced the most reliable possible risk estimates using currently available U.K. prescription data,” the researchers wrote.
There was no external funding for the study. Julia Hippisley-Cox is the unpaid director of QResearch, a not-for-profit organization that is a joint partnership between the University of Nottingham and EMIS, a commercial IT supplier. She is also a paid director of ClinRisk, which produces clinical risk algorithm-related software.
FROM BMJ
Key clinical point: Newer progestogen hormones in oral contraceptives are associated with higher risks of venous thromboembolism than are older progestogen hormones.
Major finding: Compared with no oral contraceptive exposure, desogestrel (adjusted odds ratio, 4.28), gestodene (aOR ,3.64), drospirenone (aOR, 4.12), cyproterone (aOR, 4.27), levonorgestrel (aOR, 2.38), norethisterone (aOR, 2.56) and norgestimate (aOR, 2.53) confer a higher risk for venous thromboembolism.
Data source: Two nested case-control studies involving 10,562 women with a diagnosis of VTE and 42,034 women without VTE.
Disclosures: There was no external funding for the study. Julia Hippisley-Cox is the unpaid director of QResearch, a not-for-profit organization that is a joint partnership between the University of Nottingham and EMIS, a commercial IT supplier. She is also a paid director of ClinRisk, which produces clinical risk algorithm-related software.
Grapefruit juice and psychotropics: How to avoid potential interactions
Ms. H, age 42, was given a diagnosis of bipolar disorder 10 years ago and has been taking carbamazepine, 1,200 mg/d, and olanzapine, 10 mg/d, for the past 2 years. She has not experienced a mood episode while on this regimen, and her carbamazepine level was 9.2 μg/mL 6 months ago. The only adverse effect she experienced was weight gain of approximately 10 lb. Ms. H takes a calcium supplement, but no other medications.
Ms. H reports to her psychiatrist that, for the past few days, she has been feeling nauseated, fatigued, and dizzy, but has continued taking her medications as prescribed. Her carbamazepine level is found to be 13.1 μg/mL. Ms. H states she has not started any new medications or supplements; her serum creatinine and liver function test results are within normal limits.
Upon further questioning, Ms. H says that an upper respiratory infection has been “going around her office,” so she increased her vitamin C intake by drinking 2 glasses of grapefruit juice a day (she doesn’t like orange juice). She has heard grapefruit juice can cause problems with some drugs so she is careful not to drink it at the same time she takes her medications. Her psychiatrist recognizes there may be a drug interaction involved, and recommends Ms. H hold her carbamazepine for 1 day and not consume any more grapefruit juice. A few days later, she reports feeling much better during a follow-up call and she makes an appointment to have her carbamazepine level rechecked in a we
Although grapefruit products are high in vitamins and low in calories, they can be associated with potentially serious drug interactions. The interaction between grapefruit juice and the calcium channel blocker felodipine was discovered inadvertently >20 years ago; since that time, possible interactions with >85 medications have been identified.1 Interactions with grapefruit products are complicated because, although most result in increased drug exposure, reduced exposure of the medication also can occur. Additionally, the degree and clinical significance of the interaction varies among individuals and from one drug to another.
Mechanism of action
Most interactions with grapefruit products are thought to result from the inhibition of intestinal cytochrome P450 3A4 (CYP3A4). CYP3A4 is involved in the metabolism of numerous drugs, and is the most abundant cytochrome P450 enzyme in the liver and epithelial cells lining the intestine.2 Although hepatic CYP3A4 is thought to be minimally affected by grapefruit, inhibition of intestinal CYP3A4 can result in an overall increase in bioavailability of medications that are substrates and raise the risk of potential toxicity.3 Grapefruit contains various chemicals collectively known as furanocoumarins, which are largely responsible for inhibition of intestinal CYP3A4.4 Additionally, Seville oranges and the pomelo (a large, sweet grapefruit-like citrus fruit) also contain furanocoumarins and could have a similar effect, warranting caution with certain medications.5
Inhibition of CYP3A4 by furanocoumarins cannot be reversed, and new enzymes must be synthesized to return to the previous level of function.6 Therefore, drug interactions resulting from CYP3A4 inhibition can last for as long as 72 hours after ingesting grapefruit products.7 Separating consumption of grapefruit products and medication administration will not help manage this interaction.
Grapefruit products also could affect drug disposition through effects on various drug transporters. Decreased systemic exposure to certain medications could occur through grapefruit’s inhibition of organic anion-transporting polypeptides (OATPs). OATPs form a family of drug uptake transporters found in the intestine, liver, kidney, and brain.8 For drugs that are substrates of OATPs, grapefruit’s inhibition of this transporter can result in decreased absorption and a resulting decrease in efficacy. Flavanoids in grapefruit, such as naringin, inhibit OATPs, which is competitive in nature.9 Unlike the irreversible inhibition of CYP3A4 by furanocoumarins, flavanoids effects on OATPs have been shown to decrease within 4 hours.10
No psychotropic medications have been identified as being susceptible to this interaction, but for those medications affected—including fexofenadine and levothyroxine—separating consumption of grapefruit and medication administration by 4 hours could avoid this interaction.11 Additional data indicate that orange juice and apple juice could have similar effects on OATPs.12
Perhaps the most well-known drug transporter, P-glycoprotein is part of the multidrug-resistant subfamily of transporters. It is located throughout the body, including in the intestine, kidneys, liver, and blood-brain barrier. P-glycoprotein acts as an export pump to decrease the cellular concentration of many different drug substrates, and many agents can alter P-glycoprotein’s expression or function.
Small changes in P-glycoprotein’s activity can result in substantial changes in the disposition of substrates, which can include certain antineoplastics and antiretrovirals. Most reports have found grapefruit juice inhibits P-glycoprotein-mediated efflux; however, there also are reports of transporter activation.6 Additionally, P-glycoprotein and CYP3A4 share many substrates, so it can be difficult to isolate the contribution of P-glycoprotein to grapefruit−drug interactions.13 The effect of grapefruit on P-glycoprotein activity has been difficult to fully elucidate; more studies are needed.
Grapefruit consumption and its effect
Drug interactions can occur by consuming commercially produced grapefruit juice and juice from concentrate, as well as freshly squeezed juice and grapefruit segments.14 CYP3A4-inhibiting furanocoumarins also have been isolated in grapefruit peel; it is not known, however, whether items made from peel (marmalade, candied peel) contain concentrations high enough to pose a risk of a drug interaction.14 Contributing to the unpredictability of grapefruit-drug interactions, the amount or concentration of furanocoumarins can vary among grapefruit products and brands.15 This variability can be influenced by the variety or maturity of the fruit and the fruit’s exposure to environmental stress.4
The frequency of consuming a grapefruit product can influence the degree of a drug interaction. In general, consuming one 8-oz glass of grapefruit juice or the segments from a whole grapefruit is enough to alter a susceptible drug’s pharmacokinetics.14 Regular grapefruit product consumption, however, can result in an overall greater effect.16,17
Lilja et al16 conducted a randomized, 4-phase, crossover study to look at the effect of grapefruit juice dose on kinetics of triazolam. Grapefruit juice was found to increase the mean area under the concentration-time curve (AUC) of triazolam compared with water, but no difference was found between single glasses of normal-strength and double-strength grapefruit juice. However, repeated consumption of double-strength grapefruit juice (200 mL, 3 times/d for 3 days) increased triazolam’s mean AUC by 143%, compared with an increase of 49% with just a single 200-mL glass of double-strength juice.16 Recurrent consumption of grapefruit juice (8 oz, 3 times/d for 6 days) also was found to increase the kinetics of the antihypertensive felodipine more than a single glass of grapefruit juice.17
Clinical consequences of an interaction between a drug and grapefruit can be difficult to predict. Drug concentration changes caused by a grapefruit interaction could vary based on interindividual differences. The amount and activity of intestinal CYP3A4 can vary from person to person, and can be influenced by genetic polymorphisms in addition to race, age, and environmental variables.18 Interindividual sensitivity to a change in a drug’s concentration also will differ, and patient-specific factors, such as concomitant drugs or diseases, could influence the likelihood of harm.
Interactions with grapefruit products are not necessarily a “class effect,” and specific drugs within a therapeutic category can be affected (although others might not). Several drug-specific characteristics can help gauge the risk of a clinically relevant interaction with grapefruit, including:
• metabolism through CYP3A4
• low bioavailability
• oral administration
• a narrow therapeutic index.1
For drugs with low bioavailability because of first-pass metabolism, grapefruit’s inhibition of intestinal CYP3A4 can result in a greater relative increase in plasma concentrations compared with a drug with high bioavailability.19
For example, an increase in bioavailability from 5% to 10% will result in a much larger increase in AUC and overall clinical exposure compared with an increase from 85% to 90% even though both represent an absolute increase of 5%. Although a drug does not have to have low oral bioavailability for an interaction to occur, lower bioavailability means that a drug has a higher likelihood of causing a significant interaction because of altered pharmacokinetics. Of note, injectable medications will not interact with grapefruit because metabolism through intestinal CYP3A4 is bypassed and grapefruit does not significantly inhibit hepatic CYP3A4.
Although grapefruit products could alter the pharmacokinetics of susceptible drugs, those changes might not be associated with adverse effects. Therefore, a factor to consider in evaluating a potential interaction with grapefruit is the drug’s therapeutic index and its risk of serious adverse effects. Drugs with a narrow therapeutic index are of particular concern because a significant increase in therapeutic or adverse effects could result from a relatively small increase in the drug’s concentration.7
Which medications are affected?
Among medications identified as interacting with grapefruit, some cardiovascular agents and several of the HMG-CoA reductase inhibitors (statins) have garnered the most attention. However, grapefruit also can affect the metabolism of several psychotropic medications through inhibition of intestinal CYP3A4 (Table).16,20-35 Prescribing information for some drugs warns against consuming grapefruit while using the medication. Among CNS agents, buspirone, carbamazepine, lurasidone, pimozide, triazolam, and oral midazolam all have such warnings in their product labeling.
Buspirone currently is not recommended with “large quantities of grapefruit juice.”20 A randomized, 2-phase crossover study looking at the effects of grapefruit juice on buspirone’s pharmacokinetics found that double-strength grapefruit juice (200 mL, administered 3 times/d for 3 days) resulted in a 9.2-fold increase in mean AUC and a 4.3-fold increase in mean Cmax after a single 10-mg buspirone dose.22 Highlighting the wide interindividual variability seen with drug-grapefruit interactions, the increase found in buspirone’s AUC ranged from 3-fold to 20-fold among study participants.22
Carbamazepine product labeling lists grapefruit juice as a CYP3A4 inhibitor that is expected to or has been found to increase plasma levels of the drug.20 Carbamazepine’s bioavailability is influenced by intestinal CYP3A4 activity; in a randomized, 2-phase crossover study of 10 patients with epilepsy, grapefruit juice was found to increase AUC of carbamazepine by 41% and Cmax by 40%.23,36
Lurasidone and pimozide, although not specifically studied, have product labels that recommend avoiding grapefruit juice because it could inhibit metabolism of these agents by CYP3A4.20 Of particular concern is the potential for elevated levels of pimozide to increase the risk of adverse cardiovascular effects including QT interval prolongation.19
Midazolam. Although grapefruit juice does not affect the disposition of IV midazolam, pretreatment with grapefruit juice was found to increase the AUC and Cmax of oral midazolam by 52% and 56%, respectively.30
Other considerations in drug-grapefruit interactions
Cautionary statements about a possible interaction with grapefruit juice for many other psychotropics can be found in commonly used drug information references or online sources. If you are concerned about a possible interaction and avoiding grapefruit products is not feasible, consider a different medication in the same class.
However, you also should consider the level of evidence supporting any purported interaction. Several psychotropic agents do have studies or case reports supporting an interaction with grapefruit, but cautionary statements could be based on theoretical concerns because of a medication’s bioavailability, metabolic pathway, and concern for increased adverse events related to higher drug concentrations. Adding to the confusion, cautionary statements can be found about medications, such as clozapine, that have not been shown to have an interaction with grapefruit juice when studied.
With many of the drugs that have a reported or theoretical interaction with grapefruit, data are inconsistent as to whether the resulting interaction will be clinically relevant. A number of variables relating to the individual patient, grapefruit product, or particular drug can play a role in the significance of an interaction. Additionally, effects on drug disposition can last for a few days after consuming a grapefruit product.
Keep alert to situations of increased risk
Recall that the case patient, Ms. H, presented with an elevated carbamazepine level and suffered resulting adverse effects because of an interaction between the drug and grapefruit juice. Although Ms. H was careful to separate intake of grapefruit juice from carbamazepine administration, grapefruit’s inhibition of intestinal CYP3A4 still was present, leading to the interaction.
It is important for health care professionals to recognize this potential risk and to advise patients regarding possible interactions between medications and grapefruit products.
Related Resources
• U.S. Food and Drug Administration. Grapefruit juice and medicine may not mix. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm292276.htm.
• Hanley MJ, Cancalon P, Widmer WW, et al. The effect of grapefruit juice on drug disposition. Expert Opin Drug Metab Toxicol. 2011;7(3):267-286.
• Andrade C. Fruit juice, organic anion transporting polypeptides, and drug interactions in psychiatry. J Clin Psychiatry. 2014;75(11):e1323-e1325.
Drug Brand Names
Alprazolam • Xanax Lurasidone • Latuda
Buspirone • BuSpar Midazolam • Versed
Carbamazepine • Tegretol Methadone • Dolophine
Clomipramine • Anafranil Nefazodone • Serzone
Clozapine • Clozaril Olanzapine • Zyprexa
Diazepam • Valium Pimozide • Orap
Felodipine • Plendil Quetiapine • Seroquel
Fexofenadine • Allegra Sertraline • Zoloft
Fluoxetine • Prozac Trazodone • Desyrel
Fluvoxamine • Luvox Triazolam • Halcion
Haloperidol • Haldol Ziprasidone • Geodon
Levothyroxine • Levoxyl, Synthroid
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Bailey DG, Dresser G, Arnold JM. Grapefruit-medication interactions: forbidden fruit or avoidable consequences? CMAJ. 2013;185(4):309-316.
2. Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med. 2005;352(21): 2211-2221.
3. Saito M, Hirata-Koizumi M, Matsumoto M, et al. Undesirable effects of citrus juice on the pharmacokinetics of drugs: focus on recent studies. Drug Saf. 2005;28(8):677- 694.
4. Cancalon PF, Barros SM, Haun C, et al. Effect of maturity, processing, and storage on the furanocoumarin composition of grapefruit and grapefruit juice. J Food Sci. 2011;76(4):C543-C548.
5. Pirmohamed M. Drug-grapefruit juice interactions: two mechanisms are clear but individual responses vary. BMJ. 2013;346:f1. doi: 10.1136/bmj.f1.
6. Dahan A, Altman H. Food-drug interaction: grapefruit juice augments drug bioavailability–mechanism, extent and relevance. Eur J Clin Nutr. 2004;58:1-9.
7. Stump AL, Mayo T, Blum A. Management of grapefruit-drug interactions. Am Fam Physician. 2006;74(4):605-608.
8. Kim RB. Organic anion-transporting polypeptide (OATP) transporter family and drug disposition. Eur J Clin Invest. 2003;33(suppl 2):1-5.
9. Bailey DG, Dressker GK, Leak BF, et al. Naringin is a major and selective clinical inhibitor of organic anion-transporting polypeptide 1A2 (OATP1A2) in grapefruit juice. Clin Pharmacol Ther. 2007;81(4):495-502.
10. Glaeser H, Bailey DG, Dresser GK, et al. Intestinal drug transporter expression and the impact of grapefruit juice in humans. Clin Pharmacol Ther. 2007;81(3):362-370.
11. Bailey DG. Fruit juice inhibition of uptake transport: a new type of food-drug interaction. Br J Clin Pharmacol. 2010;70(5): 645-655.
12. Dresser GK, Bailey DG, Leake BF, et al. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin Pharmacol Ther. 2002;71:11-20.
13. Seden K, Dickinson L, Khoo S, et al. Grapefruit-drug interactions. Drugs. 2010;70(18):2373-2407.
14. Bailey DG, Dresser GK, Kreeft JH, et al. Grapefruit-felodipine interaction: effect of unprocessed fruit and probable active ingredients. Clin Pharmacol Ther. 2000;68(5):468-477.
15. De Castro WV, Mertens-Talcott S, Rubner A, et al. Variation of flavonoids and furanocoumarins in grapefruit juices: a potential source of variability in grapefruit juice-drug interaction studies. J Agric Food Chem. 2006;54(1):249-255.
16. Lilja JJ, Kivistö KT, Backman JT, et al. Effect of grapefruit juice on grapefruit juice-triazolam interaction: repeated consumption prolongs triazolam half-life. Eur J Clin Pharmacol. 2000;56(5):411-415.
17. Lown KS, Bailey DG, Fontana RJ, et al. Grapefruit juice increases felodipine oral availability in humans by decreasing intestinal CYP3A protein expression. J Clin Invest. 1997;99(10):2545-2553.
18. Lin JH, Lu AY. Interindividual variability in inhibition and induction of cytochrome P450 enzymes. Annu Rev Pharmacol Toxicol. 2001;41:535-567.
19. Dresser GK, Spence JD, Bailey DG. Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet. 2000; 38(1):41-57.
20. U.S. Food and Drug Administration. Drugs@FDA. http://www.accessdata.fda.gov/scripts/cder/drugsatfda. Accessed July 14, 2014.
21. Yasui, N, Kondo T, Furukori H, et al. Effects of repeated ingestion of grapefruit juice on the single and multiple oral-dose pharmacokinetics and pharmacodynamics of alprazolam. Psychopharmacology (Berl). 2000;15(2):185-190.
22. Lilja JJ, Kivistö KT, Backman JT, et al. Grapefruit juice substantially increases plasma concentrations of buspirone. Clin Pharmacol Ther. 1998;64(6):655-660.
23. Garg SK, Kumar N, Bhargava VK, et al. Effect of grapefruit juice on carbamazepine bioavailability in patients with epilepsy. Clin Pharmacol Ther. 1998;64(3):286-288.
24. Oesterheld J, Kallepalli BR. Grapefruit juice and clomipramine: shifting metabolic ratios. J Clin Psychopharm. 1997;17(1):62-63.
25. Lane HY, Jann MW, Chang YC, et al. Repeated ingestion of grapefruit juice does not alter clozapine’s steady-state plasma levels, effectiveness, and tolerability. J Clin Psychiatry. 2001;62(10):812-817.
26. Ozdemir M, Aktan Y, Boydag BS, et al. Interaction between grapefruit juice and diazepam in humans. Eur J Drug Metab Pharmacokinet. 1998;23(1):55-59.
27. DeSilva KE, Le Flore DB, Marston BJ, et al. Serotonin syndrome in HIV-infected individuals receiving antiretroviral therapy and fluoxetine. AIDS. 2001;15(10):1281-1285.
28. Hori H, Yoshimura R, Ueda N, et al. Grapefruit juice-fluvoxamine interaction—is it risky or not? J Clin Psychopharmacol. 2003;23(4):422-424.
29. Yasui N, Kondo T, Suzuki A, et al. Lack of significant pharmacokinetic interaction between haloperidol and grapefruit juice. Int Clin Psychopharmacol. 1999;142(2):113-118.
30. Kupferschmidt HH, Ha HR, Ziegler WH, et al. Interaction between grapefruit juice and midazolam in humans. Clin Pharmacol Ther. 1995;58(1):20-28.
31. Benmebarek M, Cevaud C, Gex-Fabry M, et al. Effects of grapefruit juice on the pharmacokinetics of the enantiomers of methadone. Clin Pharmacol Ther. 2004;76(1):55-63.
32. DeVane CL, Nemeroff CB. Clinical pharmacokinetics of quetiapine: an atypical antipsychotic. Clin Pharmacokinet. 2001;40(7):509-522.
33. Ueda N, Yoshimura R, Umene-Nakano W, et al. Grapefruit juice alters plasma sertraline levels after single ingestion of sertraline in healthy volunteers. World J Biol Psychiatry. 2009;10(4 pt 3):832-835.
34. Lee AJ, Chan WK, Harralson AF, et al. The effects of grapefruit juice on sertraline metabolism: an in vitro and in vivo study. Clin Ther. 1999;21(11):1890-1899.
35. Sugimoto K, Araki N, Ohmori M, et al. Interaction between grapefruit juice and hypnotic drugs: comparison of triazolam and quazepam. Eur J Clin Pharmacol. 2006;62(3):209-215.
36. Fagiolino P, Vazquez M, Olano I, et al. Systemic and presystemic conversion of carbamazepine to carbamazepine- 10-11-epoxide during long term treatment. Journal of Epilepsy and Clinical Neurophysiology. 2006;12(1):13-16.
Ms. H, age 42, was given a diagnosis of bipolar disorder 10 years ago and has been taking carbamazepine, 1,200 mg/d, and olanzapine, 10 mg/d, for the past 2 years. She has not experienced a mood episode while on this regimen, and her carbamazepine level was 9.2 μg/mL 6 months ago. The only adverse effect she experienced was weight gain of approximately 10 lb. Ms. H takes a calcium supplement, but no other medications.
Ms. H reports to her psychiatrist that, for the past few days, she has been feeling nauseated, fatigued, and dizzy, but has continued taking her medications as prescribed. Her carbamazepine level is found to be 13.1 μg/mL. Ms. H states she has not started any new medications or supplements; her serum creatinine and liver function test results are within normal limits.
Upon further questioning, Ms. H says that an upper respiratory infection has been “going around her office,” so she increased her vitamin C intake by drinking 2 glasses of grapefruit juice a day (she doesn’t like orange juice). She has heard grapefruit juice can cause problems with some drugs so she is careful not to drink it at the same time she takes her medications. Her psychiatrist recognizes there may be a drug interaction involved, and recommends Ms. H hold her carbamazepine for 1 day and not consume any more grapefruit juice. A few days later, she reports feeling much better during a follow-up call and she makes an appointment to have her carbamazepine level rechecked in a we
Although grapefruit products are high in vitamins and low in calories, they can be associated with potentially serious drug interactions. The interaction between grapefruit juice and the calcium channel blocker felodipine was discovered inadvertently >20 years ago; since that time, possible interactions with >85 medications have been identified.1 Interactions with grapefruit products are complicated because, although most result in increased drug exposure, reduced exposure of the medication also can occur. Additionally, the degree and clinical significance of the interaction varies among individuals and from one drug to another.
Mechanism of action
Most interactions with grapefruit products are thought to result from the inhibition of intestinal cytochrome P450 3A4 (CYP3A4). CYP3A4 is involved in the metabolism of numerous drugs, and is the most abundant cytochrome P450 enzyme in the liver and epithelial cells lining the intestine.2 Although hepatic CYP3A4 is thought to be minimally affected by grapefruit, inhibition of intestinal CYP3A4 can result in an overall increase in bioavailability of medications that are substrates and raise the risk of potential toxicity.3 Grapefruit contains various chemicals collectively known as furanocoumarins, which are largely responsible for inhibition of intestinal CYP3A4.4 Additionally, Seville oranges and the pomelo (a large, sweet grapefruit-like citrus fruit) also contain furanocoumarins and could have a similar effect, warranting caution with certain medications.5
Inhibition of CYP3A4 by furanocoumarins cannot be reversed, and new enzymes must be synthesized to return to the previous level of function.6 Therefore, drug interactions resulting from CYP3A4 inhibition can last for as long as 72 hours after ingesting grapefruit products.7 Separating consumption of grapefruit products and medication administration will not help manage this interaction.
Grapefruit products also could affect drug disposition through effects on various drug transporters. Decreased systemic exposure to certain medications could occur through grapefruit’s inhibition of organic anion-transporting polypeptides (OATPs). OATPs form a family of drug uptake transporters found in the intestine, liver, kidney, and brain.8 For drugs that are substrates of OATPs, grapefruit’s inhibition of this transporter can result in decreased absorption and a resulting decrease in efficacy. Flavanoids in grapefruit, such as naringin, inhibit OATPs, which is competitive in nature.9 Unlike the irreversible inhibition of CYP3A4 by furanocoumarins, flavanoids effects on OATPs have been shown to decrease within 4 hours.10
No psychotropic medications have been identified as being susceptible to this interaction, but for those medications affected—including fexofenadine and levothyroxine—separating consumption of grapefruit and medication administration by 4 hours could avoid this interaction.11 Additional data indicate that orange juice and apple juice could have similar effects on OATPs.12
Perhaps the most well-known drug transporter, P-glycoprotein is part of the multidrug-resistant subfamily of transporters. It is located throughout the body, including in the intestine, kidneys, liver, and blood-brain barrier. P-glycoprotein acts as an export pump to decrease the cellular concentration of many different drug substrates, and many agents can alter P-glycoprotein’s expression or function.
Small changes in P-glycoprotein’s activity can result in substantial changes in the disposition of substrates, which can include certain antineoplastics and antiretrovirals. Most reports have found grapefruit juice inhibits P-glycoprotein-mediated efflux; however, there also are reports of transporter activation.6 Additionally, P-glycoprotein and CYP3A4 share many substrates, so it can be difficult to isolate the contribution of P-glycoprotein to grapefruit−drug interactions.13 The effect of grapefruit on P-glycoprotein activity has been difficult to fully elucidate; more studies are needed.
Grapefruit consumption and its effect
Drug interactions can occur by consuming commercially produced grapefruit juice and juice from concentrate, as well as freshly squeezed juice and grapefruit segments.14 CYP3A4-inhibiting furanocoumarins also have been isolated in grapefruit peel; it is not known, however, whether items made from peel (marmalade, candied peel) contain concentrations high enough to pose a risk of a drug interaction.14 Contributing to the unpredictability of grapefruit-drug interactions, the amount or concentration of furanocoumarins can vary among grapefruit products and brands.15 This variability can be influenced by the variety or maturity of the fruit and the fruit’s exposure to environmental stress.4
The frequency of consuming a grapefruit product can influence the degree of a drug interaction. In general, consuming one 8-oz glass of grapefruit juice or the segments from a whole grapefruit is enough to alter a susceptible drug’s pharmacokinetics.14 Regular grapefruit product consumption, however, can result in an overall greater effect.16,17
Lilja et al16 conducted a randomized, 4-phase, crossover study to look at the effect of grapefruit juice dose on kinetics of triazolam. Grapefruit juice was found to increase the mean area under the concentration-time curve (AUC) of triazolam compared with water, but no difference was found between single glasses of normal-strength and double-strength grapefruit juice. However, repeated consumption of double-strength grapefruit juice (200 mL, 3 times/d for 3 days) increased triazolam’s mean AUC by 143%, compared with an increase of 49% with just a single 200-mL glass of double-strength juice.16 Recurrent consumption of grapefruit juice (8 oz, 3 times/d for 6 days) also was found to increase the kinetics of the antihypertensive felodipine more than a single glass of grapefruit juice.17
Clinical consequences of an interaction between a drug and grapefruit can be difficult to predict. Drug concentration changes caused by a grapefruit interaction could vary based on interindividual differences. The amount and activity of intestinal CYP3A4 can vary from person to person, and can be influenced by genetic polymorphisms in addition to race, age, and environmental variables.18 Interindividual sensitivity to a change in a drug’s concentration also will differ, and patient-specific factors, such as concomitant drugs or diseases, could influence the likelihood of harm.
Interactions with grapefruit products are not necessarily a “class effect,” and specific drugs within a therapeutic category can be affected (although others might not). Several drug-specific characteristics can help gauge the risk of a clinically relevant interaction with grapefruit, including:
• metabolism through CYP3A4
• low bioavailability
• oral administration
• a narrow therapeutic index.1
For drugs with low bioavailability because of first-pass metabolism, grapefruit’s inhibition of intestinal CYP3A4 can result in a greater relative increase in plasma concentrations compared with a drug with high bioavailability.19
For example, an increase in bioavailability from 5% to 10% will result in a much larger increase in AUC and overall clinical exposure compared with an increase from 85% to 90% even though both represent an absolute increase of 5%. Although a drug does not have to have low oral bioavailability for an interaction to occur, lower bioavailability means that a drug has a higher likelihood of causing a significant interaction because of altered pharmacokinetics. Of note, injectable medications will not interact with grapefruit because metabolism through intestinal CYP3A4 is bypassed and grapefruit does not significantly inhibit hepatic CYP3A4.
Although grapefruit products could alter the pharmacokinetics of susceptible drugs, those changes might not be associated with adverse effects. Therefore, a factor to consider in evaluating a potential interaction with grapefruit is the drug’s therapeutic index and its risk of serious adverse effects. Drugs with a narrow therapeutic index are of particular concern because a significant increase in therapeutic or adverse effects could result from a relatively small increase in the drug’s concentration.7
Which medications are affected?
Among medications identified as interacting with grapefruit, some cardiovascular agents and several of the HMG-CoA reductase inhibitors (statins) have garnered the most attention. However, grapefruit also can affect the metabolism of several psychotropic medications through inhibition of intestinal CYP3A4 (Table).16,20-35 Prescribing information for some drugs warns against consuming grapefruit while using the medication. Among CNS agents, buspirone, carbamazepine, lurasidone, pimozide, triazolam, and oral midazolam all have such warnings in their product labeling.
Buspirone currently is not recommended with “large quantities of grapefruit juice.”20 A randomized, 2-phase crossover study looking at the effects of grapefruit juice on buspirone’s pharmacokinetics found that double-strength grapefruit juice (200 mL, administered 3 times/d for 3 days) resulted in a 9.2-fold increase in mean AUC and a 4.3-fold increase in mean Cmax after a single 10-mg buspirone dose.22 Highlighting the wide interindividual variability seen with drug-grapefruit interactions, the increase found in buspirone’s AUC ranged from 3-fold to 20-fold among study participants.22
Carbamazepine product labeling lists grapefruit juice as a CYP3A4 inhibitor that is expected to or has been found to increase plasma levels of the drug.20 Carbamazepine’s bioavailability is influenced by intestinal CYP3A4 activity; in a randomized, 2-phase crossover study of 10 patients with epilepsy, grapefruit juice was found to increase AUC of carbamazepine by 41% and Cmax by 40%.23,36
Lurasidone and pimozide, although not specifically studied, have product labels that recommend avoiding grapefruit juice because it could inhibit metabolism of these agents by CYP3A4.20 Of particular concern is the potential for elevated levels of pimozide to increase the risk of adverse cardiovascular effects including QT interval prolongation.19
Midazolam. Although grapefruit juice does not affect the disposition of IV midazolam, pretreatment with grapefruit juice was found to increase the AUC and Cmax of oral midazolam by 52% and 56%, respectively.30
Other considerations in drug-grapefruit interactions
Cautionary statements about a possible interaction with grapefruit juice for many other psychotropics can be found in commonly used drug information references or online sources. If you are concerned about a possible interaction and avoiding grapefruit products is not feasible, consider a different medication in the same class.
However, you also should consider the level of evidence supporting any purported interaction. Several psychotropic agents do have studies or case reports supporting an interaction with grapefruit, but cautionary statements could be based on theoretical concerns because of a medication’s bioavailability, metabolic pathway, and concern for increased adverse events related to higher drug concentrations. Adding to the confusion, cautionary statements can be found about medications, such as clozapine, that have not been shown to have an interaction with grapefruit juice when studied.
With many of the drugs that have a reported or theoretical interaction with grapefruit, data are inconsistent as to whether the resulting interaction will be clinically relevant. A number of variables relating to the individual patient, grapefruit product, or particular drug can play a role in the significance of an interaction. Additionally, effects on drug disposition can last for a few days after consuming a grapefruit product.
Keep alert to situations of increased risk
Recall that the case patient, Ms. H, presented with an elevated carbamazepine level and suffered resulting adverse effects because of an interaction between the drug and grapefruit juice. Although Ms. H was careful to separate intake of grapefruit juice from carbamazepine administration, grapefruit’s inhibition of intestinal CYP3A4 still was present, leading to the interaction.
It is important for health care professionals to recognize this potential risk and to advise patients regarding possible interactions between medications and grapefruit products.
Related Resources
• U.S. Food and Drug Administration. Grapefruit juice and medicine may not mix. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm292276.htm.
• Hanley MJ, Cancalon P, Widmer WW, et al. The effect of grapefruit juice on drug disposition. Expert Opin Drug Metab Toxicol. 2011;7(3):267-286.
• Andrade C. Fruit juice, organic anion transporting polypeptides, and drug interactions in psychiatry. J Clin Psychiatry. 2014;75(11):e1323-e1325.
Drug Brand Names
Alprazolam • Xanax Lurasidone • Latuda
Buspirone • BuSpar Midazolam • Versed
Carbamazepine • Tegretol Methadone • Dolophine
Clomipramine • Anafranil Nefazodone • Serzone
Clozapine • Clozaril Olanzapine • Zyprexa
Diazepam • Valium Pimozide • Orap
Felodipine • Plendil Quetiapine • Seroquel
Fexofenadine • Allegra Sertraline • Zoloft
Fluoxetine • Prozac Trazodone • Desyrel
Fluvoxamine • Luvox Triazolam • Halcion
Haloperidol • Haldol Ziprasidone • Geodon
Levothyroxine • Levoxyl, Synthroid
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Ms. H, age 42, was given a diagnosis of bipolar disorder 10 years ago and has been taking carbamazepine, 1,200 mg/d, and olanzapine, 10 mg/d, for the past 2 years. She has not experienced a mood episode while on this regimen, and her carbamazepine level was 9.2 μg/mL 6 months ago. The only adverse effect she experienced was weight gain of approximately 10 lb. Ms. H takes a calcium supplement, but no other medications.
Ms. H reports to her psychiatrist that, for the past few days, she has been feeling nauseated, fatigued, and dizzy, but has continued taking her medications as prescribed. Her carbamazepine level is found to be 13.1 μg/mL. Ms. H states she has not started any new medications or supplements; her serum creatinine and liver function test results are within normal limits.
Upon further questioning, Ms. H says that an upper respiratory infection has been “going around her office,” so she increased her vitamin C intake by drinking 2 glasses of grapefruit juice a day (she doesn’t like orange juice). She has heard grapefruit juice can cause problems with some drugs so she is careful not to drink it at the same time she takes her medications. Her psychiatrist recognizes there may be a drug interaction involved, and recommends Ms. H hold her carbamazepine for 1 day and not consume any more grapefruit juice. A few days later, she reports feeling much better during a follow-up call and she makes an appointment to have her carbamazepine level rechecked in a we
Although grapefruit products are high in vitamins and low in calories, they can be associated with potentially serious drug interactions. The interaction between grapefruit juice and the calcium channel blocker felodipine was discovered inadvertently >20 years ago; since that time, possible interactions with >85 medications have been identified.1 Interactions with grapefruit products are complicated because, although most result in increased drug exposure, reduced exposure of the medication also can occur. Additionally, the degree and clinical significance of the interaction varies among individuals and from one drug to another.
Mechanism of action
Most interactions with grapefruit products are thought to result from the inhibition of intestinal cytochrome P450 3A4 (CYP3A4). CYP3A4 is involved in the metabolism of numerous drugs, and is the most abundant cytochrome P450 enzyme in the liver and epithelial cells lining the intestine.2 Although hepatic CYP3A4 is thought to be minimally affected by grapefruit, inhibition of intestinal CYP3A4 can result in an overall increase in bioavailability of medications that are substrates and raise the risk of potential toxicity.3 Grapefruit contains various chemicals collectively known as furanocoumarins, which are largely responsible for inhibition of intestinal CYP3A4.4 Additionally, Seville oranges and the pomelo (a large, sweet grapefruit-like citrus fruit) also contain furanocoumarins and could have a similar effect, warranting caution with certain medications.5
Inhibition of CYP3A4 by furanocoumarins cannot be reversed, and new enzymes must be synthesized to return to the previous level of function.6 Therefore, drug interactions resulting from CYP3A4 inhibition can last for as long as 72 hours after ingesting grapefruit products.7 Separating consumption of grapefruit products and medication administration will not help manage this interaction.
Grapefruit products also could affect drug disposition through effects on various drug transporters. Decreased systemic exposure to certain medications could occur through grapefruit’s inhibition of organic anion-transporting polypeptides (OATPs). OATPs form a family of drug uptake transporters found in the intestine, liver, kidney, and brain.8 For drugs that are substrates of OATPs, grapefruit’s inhibition of this transporter can result in decreased absorption and a resulting decrease in efficacy. Flavanoids in grapefruit, such as naringin, inhibit OATPs, which is competitive in nature.9 Unlike the irreversible inhibition of CYP3A4 by furanocoumarins, flavanoids effects on OATPs have been shown to decrease within 4 hours.10
No psychotropic medications have been identified as being susceptible to this interaction, but for those medications affected—including fexofenadine and levothyroxine—separating consumption of grapefruit and medication administration by 4 hours could avoid this interaction.11 Additional data indicate that orange juice and apple juice could have similar effects on OATPs.12
Perhaps the most well-known drug transporter, P-glycoprotein is part of the multidrug-resistant subfamily of transporters. It is located throughout the body, including in the intestine, kidneys, liver, and blood-brain barrier. P-glycoprotein acts as an export pump to decrease the cellular concentration of many different drug substrates, and many agents can alter P-glycoprotein’s expression or function.
Small changes in P-glycoprotein’s activity can result in substantial changes in the disposition of substrates, which can include certain antineoplastics and antiretrovirals. Most reports have found grapefruit juice inhibits P-glycoprotein-mediated efflux; however, there also are reports of transporter activation.6 Additionally, P-glycoprotein and CYP3A4 share many substrates, so it can be difficult to isolate the contribution of P-glycoprotein to grapefruit−drug interactions.13 The effect of grapefruit on P-glycoprotein activity has been difficult to fully elucidate; more studies are needed.
Grapefruit consumption and its effect
Drug interactions can occur by consuming commercially produced grapefruit juice and juice from concentrate, as well as freshly squeezed juice and grapefruit segments.14 CYP3A4-inhibiting furanocoumarins also have been isolated in grapefruit peel; it is not known, however, whether items made from peel (marmalade, candied peel) contain concentrations high enough to pose a risk of a drug interaction.14 Contributing to the unpredictability of grapefruit-drug interactions, the amount or concentration of furanocoumarins can vary among grapefruit products and brands.15 This variability can be influenced by the variety or maturity of the fruit and the fruit’s exposure to environmental stress.4
The frequency of consuming a grapefruit product can influence the degree of a drug interaction. In general, consuming one 8-oz glass of grapefruit juice or the segments from a whole grapefruit is enough to alter a susceptible drug’s pharmacokinetics.14 Regular grapefruit product consumption, however, can result in an overall greater effect.16,17
Lilja et al16 conducted a randomized, 4-phase, crossover study to look at the effect of grapefruit juice dose on kinetics of triazolam. Grapefruit juice was found to increase the mean area under the concentration-time curve (AUC) of triazolam compared with water, but no difference was found between single glasses of normal-strength and double-strength grapefruit juice. However, repeated consumption of double-strength grapefruit juice (200 mL, 3 times/d for 3 days) increased triazolam’s mean AUC by 143%, compared with an increase of 49% with just a single 200-mL glass of double-strength juice.16 Recurrent consumption of grapefruit juice (8 oz, 3 times/d for 6 days) also was found to increase the kinetics of the antihypertensive felodipine more than a single glass of grapefruit juice.17
Clinical consequences of an interaction between a drug and grapefruit can be difficult to predict. Drug concentration changes caused by a grapefruit interaction could vary based on interindividual differences. The amount and activity of intestinal CYP3A4 can vary from person to person, and can be influenced by genetic polymorphisms in addition to race, age, and environmental variables.18 Interindividual sensitivity to a change in a drug’s concentration also will differ, and patient-specific factors, such as concomitant drugs or diseases, could influence the likelihood of harm.
Interactions with grapefruit products are not necessarily a “class effect,” and specific drugs within a therapeutic category can be affected (although others might not). Several drug-specific characteristics can help gauge the risk of a clinically relevant interaction with grapefruit, including:
• metabolism through CYP3A4
• low bioavailability
• oral administration
• a narrow therapeutic index.1
For drugs with low bioavailability because of first-pass metabolism, grapefruit’s inhibition of intestinal CYP3A4 can result in a greater relative increase in plasma concentrations compared with a drug with high bioavailability.19
For example, an increase in bioavailability from 5% to 10% will result in a much larger increase in AUC and overall clinical exposure compared with an increase from 85% to 90% even though both represent an absolute increase of 5%. Although a drug does not have to have low oral bioavailability for an interaction to occur, lower bioavailability means that a drug has a higher likelihood of causing a significant interaction because of altered pharmacokinetics. Of note, injectable medications will not interact with grapefruit because metabolism through intestinal CYP3A4 is bypassed and grapefruit does not significantly inhibit hepatic CYP3A4.
Although grapefruit products could alter the pharmacokinetics of susceptible drugs, those changes might not be associated with adverse effects. Therefore, a factor to consider in evaluating a potential interaction with grapefruit is the drug’s therapeutic index and its risk of serious adverse effects. Drugs with a narrow therapeutic index are of particular concern because a significant increase in therapeutic or adverse effects could result from a relatively small increase in the drug’s concentration.7
Which medications are affected?
Among medications identified as interacting with grapefruit, some cardiovascular agents and several of the HMG-CoA reductase inhibitors (statins) have garnered the most attention. However, grapefruit also can affect the metabolism of several psychotropic medications through inhibition of intestinal CYP3A4 (Table).16,20-35 Prescribing information for some drugs warns against consuming grapefruit while using the medication. Among CNS agents, buspirone, carbamazepine, lurasidone, pimozide, triazolam, and oral midazolam all have such warnings in their product labeling.
Buspirone currently is not recommended with “large quantities of grapefruit juice.”20 A randomized, 2-phase crossover study looking at the effects of grapefruit juice on buspirone’s pharmacokinetics found that double-strength grapefruit juice (200 mL, administered 3 times/d for 3 days) resulted in a 9.2-fold increase in mean AUC and a 4.3-fold increase in mean Cmax after a single 10-mg buspirone dose.22 Highlighting the wide interindividual variability seen with drug-grapefruit interactions, the increase found in buspirone’s AUC ranged from 3-fold to 20-fold among study participants.22
Carbamazepine product labeling lists grapefruit juice as a CYP3A4 inhibitor that is expected to or has been found to increase plasma levels of the drug.20 Carbamazepine’s bioavailability is influenced by intestinal CYP3A4 activity; in a randomized, 2-phase crossover study of 10 patients with epilepsy, grapefruit juice was found to increase AUC of carbamazepine by 41% and Cmax by 40%.23,36
Lurasidone and pimozide, although not specifically studied, have product labels that recommend avoiding grapefruit juice because it could inhibit metabolism of these agents by CYP3A4.20 Of particular concern is the potential for elevated levels of pimozide to increase the risk of adverse cardiovascular effects including QT interval prolongation.19
Midazolam. Although grapefruit juice does not affect the disposition of IV midazolam, pretreatment with grapefruit juice was found to increase the AUC and Cmax of oral midazolam by 52% and 56%, respectively.30
Other considerations in drug-grapefruit interactions
Cautionary statements about a possible interaction with grapefruit juice for many other psychotropics can be found in commonly used drug information references or online sources. If you are concerned about a possible interaction and avoiding grapefruit products is not feasible, consider a different medication in the same class.
However, you also should consider the level of evidence supporting any purported interaction. Several psychotropic agents do have studies or case reports supporting an interaction with grapefruit, but cautionary statements could be based on theoretical concerns because of a medication’s bioavailability, metabolic pathway, and concern for increased adverse events related to higher drug concentrations. Adding to the confusion, cautionary statements can be found about medications, such as clozapine, that have not been shown to have an interaction with grapefruit juice when studied.
With many of the drugs that have a reported or theoretical interaction with grapefruit, data are inconsistent as to whether the resulting interaction will be clinically relevant. A number of variables relating to the individual patient, grapefruit product, or particular drug can play a role in the significance of an interaction. Additionally, effects on drug disposition can last for a few days after consuming a grapefruit product.
Keep alert to situations of increased risk
Recall that the case patient, Ms. H, presented with an elevated carbamazepine level and suffered resulting adverse effects because of an interaction between the drug and grapefruit juice. Although Ms. H was careful to separate intake of grapefruit juice from carbamazepine administration, grapefruit’s inhibition of intestinal CYP3A4 still was present, leading to the interaction.
It is important for health care professionals to recognize this potential risk and to advise patients regarding possible interactions between medications and grapefruit products.
Related Resources
• U.S. Food and Drug Administration. Grapefruit juice and medicine may not mix. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm292276.htm.
• Hanley MJ, Cancalon P, Widmer WW, et al. The effect of grapefruit juice on drug disposition. Expert Opin Drug Metab Toxicol. 2011;7(3):267-286.
• Andrade C. Fruit juice, organic anion transporting polypeptides, and drug interactions in psychiatry. J Clin Psychiatry. 2014;75(11):e1323-e1325.
Drug Brand Names
Alprazolam • Xanax Lurasidone • Latuda
Buspirone • BuSpar Midazolam • Versed
Carbamazepine • Tegretol Methadone • Dolophine
Clomipramine • Anafranil Nefazodone • Serzone
Clozapine • Clozaril Olanzapine • Zyprexa
Diazepam • Valium Pimozide • Orap
Felodipine • Plendil Quetiapine • Seroquel
Fexofenadine • Allegra Sertraline • Zoloft
Fluoxetine • Prozac Trazodone • Desyrel
Fluvoxamine • Luvox Triazolam • Halcion
Haloperidol • Haldol Ziprasidone • Geodon
Levothyroxine • Levoxyl, Synthroid
Disclosure
The author reports no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Bailey DG, Dresser G, Arnold JM. Grapefruit-medication interactions: forbidden fruit or avoidable consequences? CMAJ. 2013;185(4):309-316.
2. Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med. 2005;352(21): 2211-2221.
3. Saito M, Hirata-Koizumi M, Matsumoto M, et al. Undesirable effects of citrus juice on the pharmacokinetics of drugs: focus on recent studies. Drug Saf. 2005;28(8):677- 694.
4. Cancalon PF, Barros SM, Haun C, et al. Effect of maturity, processing, and storage on the furanocoumarin composition of grapefruit and grapefruit juice. J Food Sci. 2011;76(4):C543-C548.
5. Pirmohamed M. Drug-grapefruit juice interactions: two mechanisms are clear but individual responses vary. BMJ. 2013;346:f1. doi: 10.1136/bmj.f1.
6. Dahan A, Altman H. Food-drug interaction: grapefruit juice augments drug bioavailability–mechanism, extent and relevance. Eur J Clin Nutr. 2004;58:1-9.
7. Stump AL, Mayo T, Blum A. Management of grapefruit-drug interactions. Am Fam Physician. 2006;74(4):605-608.
8. Kim RB. Organic anion-transporting polypeptide (OATP) transporter family and drug disposition. Eur J Clin Invest. 2003;33(suppl 2):1-5.
9. Bailey DG, Dressker GK, Leak BF, et al. Naringin is a major and selective clinical inhibitor of organic anion-transporting polypeptide 1A2 (OATP1A2) in grapefruit juice. Clin Pharmacol Ther. 2007;81(4):495-502.
10. Glaeser H, Bailey DG, Dresser GK, et al. Intestinal drug transporter expression and the impact of grapefruit juice in humans. Clin Pharmacol Ther. 2007;81(3):362-370.
11. Bailey DG. Fruit juice inhibition of uptake transport: a new type of food-drug interaction. Br J Clin Pharmacol. 2010;70(5): 645-655.
12. Dresser GK, Bailey DG, Leake BF, et al. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin Pharmacol Ther. 2002;71:11-20.
13. Seden K, Dickinson L, Khoo S, et al. Grapefruit-drug interactions. Drugs. 2010;70(18):2373-2407.
14. Bailey DG, Dresser GK, Kreeft JH, et al. Grapefruit-felodipine interaction: effect of unprocessed fruit and probable active ingredients. Clin Pharmacol Ther. 2000;68(5):468-477.
15. De Castro WV, Mertens-Talcott S, Rubner A, et al. Variation of flavonoids and furanocoumarins in grapefruit juices: a potential source of variability in grapefruit juice-drug interaction studies. J Agric Food Chem. 2006;54(1):249-255.
16. Lilja JJ, Kivistö KT, Backman JT, et al. Effect of grapefruit juice on grapefruit juice-triazolam interaction: repeated consumption prolongs triazolam half-life. Eur J Clin Pharmacol. 2000;56(5):411-415.
17. Lown KS, Bailey DG, Fontana RJ, et al. Grapefruit juice increases felodipine oral availability in humans by decreasing intestinal CYP3A protein expression. J Clin Invest. 1997;99(10):2545-2553.
18. Lin JH, Lu AY. Interindividual variability in inhibition and induction of cytochrome P450 enzymes. Annu Rev Pharmacol Toxicol. 2001;41:535-567.
19. Dresser GK, Spence JD, Bailey DG. Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet. 2000; 38(1):41-57.
20. U.S. Food and Drug Administration. Drugs@FDA. http://www.accessdata.fda.gov/scripts/cder/drugsatfda. Accessed July 14, 2014.
21. Yasui, N, Kondo T, Furukori H, et al. Effects of repeated ingestion of grapefruit juice on the single and multiple oral-dose pharmacokinetics and pharmacodynamics of alprazolam. Psychopharmacology (Berl). 2000;15(2):185-190.
22. Lilja JJ, Kivistö KT, Backman JT, et al. Grapefruit juice substantially increases plasma concentrations of buspirone. Clin Pharmacol Ther. 1998;64(6):655-660.
23. Garg SK, Kumar N, Bhargava VK, et al. Effect of grapefruit juice on carbamazepine bioavailability in patients with epilepsy. Clin Pharmacol Ther. 1998;64(3):286-288.
24. Oesterheld J, Kallepalli BR. Grapefruit juice and clomipramine: shifting metabolic ratios. J Clin Psychopharm. 1997;17(1):62-63.
25. Lane HY, Jann MW, Chang YC, et al. Repeated ingestion of grapefruit juice does not alter clozapine’s steady-state plasma levels, effectiveness, and tolerability. J Clin Psychiatry. 2001;62(10):812-817.
26. Ozdemir M, Aktan Y, Boydag BS, et al. Interaction between grapefruit juice and diazepam in humans. Eur J Drug Metab Pharmacokinet. 1998;23(1):55-59.
27. DeSilva KE, Le Flore DB, Marston BJ, et al. Serotonin syndrome in HIV-infected individuals receiving antiretroviral therapy and fluoxetine. AIDS. 2001;15(10):1281-1285.
28. Hori H, Yoshimura R, Ueda N, et al. Grapefruit juice-fluvoxamine interaction—is it risky or not? J Clin Psychopharmacol. 2003;23(4):422-424.
29. Yasui N, Kondo T, Suzuki A, et al. Lack of significant pharmacokinetic interaction between haloperidol and grapefruit juice. Int Clin Psychopharmacol. 1999;142(2):113-118.
30. Kupferschmidt HH, Ha HR, Ziegler WH, et al. Interaction between grapefruit juice and midazolam in humans. Clin Pharmacol Ther. 1995;58(1):20-28.
31. Benmebarek M, Cevaud C, Gex-Fabry M, et al. Effects of grapefruit juice on the pharmacokinetics of the enantiomers of methadone. Clin Pharmacol Ther. 2004;76(1):55-63.
32. DeVane CL, Nemeroff CB. Clinical pharmacokinetics of quetiapine: an atypical antipsychotic. Clin Pharmacokinet. 2001;40(7):509-522.
33. Ueda N, Yoshimura R, Umene-Nakano W, et al. Grapefruit juice alters plasma sertraline levels after single ingestion of sertraline in healthy volunteers. World J Biol Psychiatry. 2009;10(4 pt 3):832-835.
34. Lee AJ, Chan WK, Harralson AF, et al. The effects of grapefruit juice on sertraline metabolism: an in vitro and in vivo study. Clin Ther. 1999;21(11):1890-1899.
35. Sugimoto K, Araki N, Ohmori M, et al. Interaction between grapefruit juice and hypnotic drugs: comparison of triazolam and quazepam. Eur J Clin Pharmacol. 2006;62(3):209-215.
36. Fagiolino P, Vazquez M, Olano I, et al. Systemic and presystemic conversion of carbamazepine to carbamazepine- 10-11-epoxide during long term treatment. Journal of Epilepsy and Clinical Neurophysiology. 2006;12(1):13-16.
1. Bailey DG, Dresser G, Arnold JM. Grapefruit-medication interactions: forbidden fruit or avoidable consequences? CMAJ. 2013;185(4):309-316.
2. Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med. 2005;352(21): 2211-2221.
3. Saito M, Hirata-Koizumi M, Matsumoto M, et al. Undesirable effects of citrus juice on the pharmacokinetics of drugs: focus on recent studies. Drug Saf. 2005;28(8):677- 694.
4. Cancalon PF, Barros SM, Haun C, et al. Effect of maturity, processing, and storage on the furanocoumarin composition of grapefruit and grapefruit juice. J Food Sci. 2011;76(4):C543-C548.
5. Pirmohamed M. Drug-grapefruit juice interactions: two mechanisms are clear but individual responses vary. BMJ. 2013;346:f1. doi: 10.1136/bmj.f1.
6. Dahan A, Altman H. Food-drug interaction: grapefruit juice augments drug bioavailability–mechanism, extent and relevance. Eur J Clin Nutr. 2004;58:1-9.
7. Stump AL, Mayo T, Blum A. Management of grapefruit-drug interactions. Am Fam Physician. 2006;74(4):605-608.
8. Kim RB. Organic anion-transporting polypeptide (OATP) transporter family and drug disposition. Eur J Clin Invest. 2003;33(suppl 2):1-5.
9. Bailey DG, Dressker GK, Leak BF, et al. Naringin is a major and selective clinical inhibitor of organic anion-transporting polypeptide 1A2 (OATP1A2) in grapefruit juice. Clin Pharmacol Ther. 2007;81(4):495-502.
10. Glaeser H, Bailey DG, Dresser GK, et al. Intestinal drug transporter expression and the impact of grapefruit juice in humans. Clin Pharmacol Ther. 2007;81(3):362-370.
11. Bailey DG. Fruit juice inhibition of uptake transport: a new type of food-drug interaction. Br J Clin Pharmacol. 2010;70(5): 645-655.
12. Dresser GK, Bailey DG, Leake BF, et al. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin Pharmacol Ther. 2002;71:11-20.
13. Seden K, Dickinson L, Khoo S, et al. Grapefruit-drug interactions. Drugs. 2010;70(18):2373-2407.
14. Bailey DG, Dresser GK, Kreeft JH, et al. Grapefruit-felodipine interaction: effect of unprocessed fruit and probable active ingredients. Clin Pharmacol Ther. 2000;68(5):468-477.
15. De Castro WV, Mertens-Talcott S, Rubner A, et al. Variation of flavonoids and furanocoumarins in grapefruit juices: a potential source of variability in grapefruit juice-drug interaction studies. J Agric Food Chem. 2006;54(1):249-255.
16. Lilja JJ, Kivistö KT, Backman JT, et al. Effect of grapefruit juice on grapefruit juice-triazolam interaction: repeated consumption prolongs triazolam half-life. Eur J Clin Pharmacol. 2000;56(5):411-415.
17. Lown KS, Bailey DG, Fontana RJ, et al. Grapefruit juice increases felodipine oral availability in humans by decreasing intestinal CYP3A protein expression. J Clin Invest. 1997;99(10):2545-2553.
18. Lin JH, Lu AY. Interindividual variability in inhibition and induction of cytochrome P450 enzymes. Annu Rev Pharmacol Toxicol. 2001;41:535-567.
19. Dresser GK, Spence JD, Bailey DG. Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet. 2000; 38(1):41-57.
20. U.S. Food and Drug Administration. Drugs@FDA. http://www.accessdata.fda.gov/scripts/cder/drugsatfda. Accessed July 14, 2014.
21. Yasui, N, Kondo T, Furukori H, et al. Effects of repeated ingestion of grapefruit juice on the single and multiple oral-dose pharmacokinetics and pharmacodynamics of alprazolam. Psychopharmacology (Berl). 2000;15(2):185-190.
22. Lilja JJ, Kivistö KT, Backman JT, et al. Grapefruit juice substantially increases plasma concentrations of buspirone. Clin Pharmacol Ther. 1998;64(6):655-660.
23. Garg SK, Kumar N, Bhargava VK, et al. Effect of grapefruit juice on carbamazepine bioavailability in patients with epilepsy. Clin Pharmacol Ther. 1998;64(3):286-288.
24. Oesterheld J, Kallepalli BR. Grapefruit juice and clomipramine: shifting metabolic ratios. J Clin Psychopharm. 1997;17(1):62-63.
25. Lane HY, Jann MW, Chang YC, et al. Repeated ingestion of grapefruit juice does not alter clozapine’s steady-state plasma levels, effectiveness, and tolerability. J Clin Psychiatry. 2001;62(10):812-817.
26. Ozdemir M, Aktan Y, Boydag BS, et al. Interaction between grapefruit juice and diazepam in humans. Eur J Drug Metab Pharmacokinet. 1998;23(1):55-59.
27. DeSilva KE, Le Flore DB, Marston BJ, et al. Serotonin syndrome in HIV-infected individuals receiving antiretroviral therapy and fluoxetine. AIDS. 2001;15(10):1281-1285.
28. Hori H, Yoshimura R, Ueda N, et al. Grapefruit juice-fluvoxamine interaction—is it risky or not? J Clin Psychopharmacol. 2003;23(4):422-424.
29. Yasui N, Kondo T, Suzuki A, et al. Lack of significant pharmacokinetic interaction between haloperidol and grapefruit juice. Int Clin Psychopharmacol. 1999;142(2):113-118.
30. Kupferschmidt HH, Ha HR, Ziegler WH, et al. Interaction between grapefruit juice and midazolam in humans. Clin Pharmacol Ther. 1995;58(1):20-28.
31. Benmebarek M, Cevaud C, Gex-Fabry M, et al. Effects of grapefruit juice on the pharmacokinetics of the enantiomers of methadone. Clin Pharmacol Ther. 2004;76(1):55-63.
32. DeVane CL, Nemeroff CB. Clinical pharmacokinetics of quetiapine: an atypical antipsychotic. Clin Pharmacokinet. 2001;40(7):509-522.
33. Ueda N, Yoshimura R, Umene-Nakano W, et al. Grapefruit juice alters plasma sertraline levels after single ingestion of sertraline in healthy volunteers. World J Biol Psychiatry. 2009;10(4 pt 3):832-835.
34. Lee AJ, Chan WK, Harralson AF, et al. The effects of grapefruit juice on sertraline metabolism: an in vitro and in vivo study. Clin Ther. 1999;21(11):1890-1899.
35. Sugimoto K, Araki N, Ohmori M, et al. Interaction between grapefruit juice and hypnotic drugs: comparison of triazolam and quazepam. Eur J Clin Pharmacol. 2006;62(3):209-215.
36. Fagiolino P, Vazquez M, Olano I, et al. Systemic and presystemic conversion of carbamazepine to carbamazepine- 10-11-epoxide during long term treatment. Journal of Epilepsy and Clinical Neurophysiology. 2006;12(1):13-16.
PSYCHIATRY UPDATE 2015
Current Psychiatry welcomed more than 650 psychiatric practitioners from across the United States and abroad to this annual conference, which was headed by Meeting Co-chairs Richard Balon, MD, and Donald W. Black, MD, April 16-18, 2015, at the Hilton Chicago in Chicago, Illinois. Attendees earned as many as 18 AMA PRA Category 1 Credits™. We welcome you to join us next year in Chicago, March 10-12, 2016.
THURSDAY, APRIL 16, 2015
MORNING SESSION
Attention-deficit/hyperactivity disorder (ADHD) is a lifespan disorder that is “everywhere,” Anthony L. Rostain, MD, MA, University of Pennsylvania Perelman School of Medicine, began—including in adults and even “seniors.” This means that the disorder “is not a diagnosis of exclusion,” and that “comorbidity is the rule,” including learning difficulties. Among adults, the focus of symptoms and management is on executive dysfunction and its characteristics: difficulty multitasking, problems keeping commitments, and excessive reliance on help from others. Inattention and disorganization are hallmarks of adult ADHD, and become worse as environmental demands (work, home) increase; hyperactivity decreases with age. Dr. Rostain recommends ruling out other causes of a patient’s symptoms when an adult self-reports ADHD, including transient stressors, medical conditions, psychiatric disorders, and malingering.
Donald W. Black, MD, University of Iowa, reviewed DSM-5 criteria for borderline personality disorder (BPD) and offered tips for avoiding misdiagnosis, including obtaining collateral information and using rating scales. Co-occuring disorders, such as depression and substance abuse, are common. Treatment for BPD patients includes psychotherapy (individual or group), medication, and lifestyle changes. Psychotropics treat symptoms of depression, anxiety, hostility, and impulsivity of BPD but not the fundamental nature of the disorder. When establishing a patient’s treatment plan, consider the stage of illness, evaluate for any co-occurring disorders, and ask the patient what he (she) wants from treatment.
Dr. Rostain began by discussing the neurobiological basis of ADHD, which guides pharmacotherapy. He reviewed the response rate of FDA-approved agents for adults with ADHD, including stimulants, atomoxetine, and alpha-adrenergic agonists. Best response is seen with stimulants, but some patients improve with bupropion and tricyclic antidepressants (TCAs). Employ a multimodal treatment approach, Dr. Rostain recommended, which should include psychoeducation and environmental restructuring, because, as he says, “Pills don’t teach skills.” He also reviewed strategies for treating ADHD in patients who have a comorbid disorder, such as bipolar disorder, major depressive disorder, or substance abuse.
Patients with psychotic depression meet criteria for major depressive disorder but also have delusions or hallucinations. Diagnostic issues include increased guilt, cognitive impairment, paranoia, and increased hopelessness. Anthony J. Rothschild, MD, University of Massachusetts Medical School, reviewed methods for differentiating psychotic depression from schizophrenia, posttraumatic stress disorder, obsessive-compulsive disorder, and body dysmorphic disorder. There are no FDA-approved medications for psychotic depression, Dr. Rothschild explained; however, evidence shows that the combination of an antidepressant and an antipsychotic is superior to monotherapy with an agent from either class. In addition, he noted, studies show a high response rate with electroconvulsive therapy (ECT).
AFTERNOON SESSION
Return of symptoms after initial remission— while the patient is still taking an antidepressant—is considered tachyphylaxis, or “poop out.” Residual depressive symptoms, when a patient meets criteria for remission but still has troubling symptoms, is a different phenomenon, although symptoms can overlap. First, Dr. Rothschild advised, ensure that patients are given an adequate trial of an antidepressant. Options are similar when tachyphylaxis or residual symptoms are present: switch drugs or add augmentation therapy, such as lithium, thyroid hormone, or an atypical antipsychotic. Data on the efficacy for bupropion and buspirone are not strong. For treatment-resistant depression when a patient does not respond to 3 adequate antidepressant trials—consider ECT or rTMS, if available, or a monoamine oxidase inhibitor or a TCA.
Dr. Black defines antisocial personality disorder (ASPD) as a disorder of lifelong serial misbehavior, one characterized by impaired relationships, aggressive behavior, non-aggressive delinquent behavior, manipulation, and a disturbing lack of conscience. There is no standard treatment for ASPD, and no FDA-approved medications; however, potential treatments have not been adequately studied, he pointed out. Cognitive-behavioral therapy might be appropriate in mild cases; some patients benefit from specific programs— for example, ones that address drug or alcohol addiction or anger, although evidence is limited. When treating ASPD patients, Dr. Black concluded, be mindful of high attrition, possible misuse of prescribed medications, and drug-drug or drug-alcohol interactions.
Bipolar disorder is associated with the highest risk of suicide and increased lethality among all psychiatric disorders. Lithium has evidence of an anti-suicidality effect and may reduce suicide by decreasing relapse, aggression, and impulsivity. An FDA advisory on increased risk of suicidality with anticonvulsants was based on data about patients with epilepsy, not bipolar disorder. Second-generation antipsychotics, including olanzapine, quetiapine, and lurasidone, have been shown to be effective for bipolar depression. Avoid antidepressants if possible, Philip G. Janicak, MD, Northwestern University Feinberg School of Medicine, advised; if you must prescribe one, reassess the need for the drug often. Several psychotherapy modalities have evidence supporting their use in bipolar disorder.
FRIDAY, APRIL 17, 2015
MORNING SESSION
Henry A. Nasrallah, MD, Saint Louis University School of Medicine, offered enlightening historical touch-points on how psychiatry’s understanding of, and its approach to, schizophrenia have changed in the past 50 years. His goal? To challenge practitioners to rethink ideas about the disorder and how they care for affected patients. From a laundry list of comparative shifts, here are a few of Dr. Nasrallah’s “then” and “now” observations:
• The old paradigm was: Clinical and functional deterioration are inevitable in schizophrenia. The new paradigm is: Complete remission and restoration of function are feasible in many patients when they are fully adherent to the treatment plan.
• The old: Long-acting injectable (LAI) antipsychotics are a last-resort treatment, to be prescribed after a patient is stabilized. The new: Use LAI antipsychotics early in the course.
• Old: Begin treatment when psychosis appears. New: Work to prevent conversion to psychosis.
• Old: The disorder is considered a consequence of neurochemical dysregulation. New: Impaired neuroplasticity is to blame.
• Old: Treatment is a matter of trial and error. New: We can apply pharmaco-genomics to predict a patient’s response to various drugs and thus increase the likelihood of therapeutic success.
In his second presentation, Dr. Nasrallah described the many pathways to psychosis and several psychotic disorders other than schizophrenia, including schizoaffective, delusional disorder, and psychotic disorder caused by a general medical condition. He listed symptom clusters in psychosis beyond positive and negative symptoms, including neuromotor symptoms, mood symptoms, and neurocognitive deficits. Development of schizophrenia is multifactorial and involves risk genes and environmental factors seen before conception, during birth, and in early childhood; good prenatal care is the best way to prevent schizophrenia, Dr. Nasrallah noted. Several general medical conditions can produce schizophrenia-like psychosis, including some CNS disorders, toxins, autoimmune diseases, infectious diseases, and chromosomal abnormalities. The session concluded with a live interview with one of Dr. Nasrallah’s patients, whose schizophrenia is in remission with clozapine.
Drug abuse can mask signs and symptoms of bipolar disorder, which can delay diagnosis. Commonly abused substances are nicotine, alcohol, Cannabis, and cocaine; polysubstance abuse is the rule. Bipolar disorder and substance abuse share common mechanisms: impulsivity, poor modulation of motivation and response to reward, and behavioral sensitization. Treatment approaches should be flexible. Dr. Janicak reviewed the evidence for using anticonvulsants, antipsychotics, and bupropion for alcohol, Cannabis, and cocaine abuse; there are no data on treating opioid abuse. He also discussed the evidence for using naltrexone, acamprosate, disulfiram, and varenicline, as well as psychotherapeutic options, to treat substance abuse. Dr. Janicak encouraged clinicians in the audience to treat substance abuse in bipolar disorder patients themselves, instead of referring them to a subspecialist.
Untreated psychiatric disorders increase obstetrical complications, possibly through decreased self-care or increased stress. For mild or moderate depression, psychotherapy might be sufficient treatment; but for severe cases, medication is the first-line approach. In her presentation on mood disorders during pregnancy, Marlene P. Freeman, MD, Massachusetts General Hospital, advises that clinicians select medications based on known safety information, patient preference, and the previous course of illness. Results of studies that lasted 4 to 5 years do not show major long-term adverse effects of antidepressant exposure on neurodevelopment or neurobehavior. When treating patients for bipolar disorder, valproate is associated with an increased risk of adverse cognitive and neurodevelopmental effects in infants compared with other anticonvulsants; evidence suggests that lamotrigine is a safer option. The research does not show an increased risk of major malformations with second-generation antipsychotics.
AFTERNOON SESSION
Most women have premenstrual symptoms; a minority have a full-blown syndrome, now known as premenstrual dysphoric disorder (PMDD). This is not an existing mood disorder that becomes worse premenstrually. Clinician and patients should track the temporal relationship of symptoms on a calendar for a few months. Selective serotonin reuptake inhibitors (SSRIs) and venlafaxine have been well studied and are effective compared with placebo, but don’t help all patients with PMDD. Consider flexible dosing strategies with SSRIs—perhaps daily use, a higher dosage premenstrually, and as-needed administration. Start with an oral contraceptive or SSRI; if symptoms don’t respond, add the other. Serotonergic antidepressants have been shown helpful for hot flashes and depressive symptoms in perimenopause. Dr. Freeman reviewed the evidence for using complementary and alternative therapies for menopausal symptoms and hot flushes.
Smoking contributes to excess mortality in seriously mentally ill patients as a result of such tobacco-related illnesses as heart disease, lung disease, and cancer. Overall improvement in mental health as well as physical health is seen when a patient stops smoking. All nicotine replacement products are effective, but patients often don’t use them long enough or correctly. Robert M. Anthenelli, MD, University of California, San Diego, said to begin sustained-release bupropion 1 or 2 weeks before quit date; maintain the dosage for 1 to 12 weeks after quit date and consider maintenance therapy for as long as 6 months. Varenicline is superior to placebo and bupropion, but is known to have gastrointestinal (GI) and sleep disturbance adverse effects. Quitting smoking can increase the blood level of some psychotropics, meaning that you might need to reduce their dosage. It is best to begin smoking cessation when patients are mentally stable, when motivated, and stable on their medications.
In discussing trends in substance abuse, Dr. Anthenelli
faddish. Fentanyl and fentanyl analogues are 100 times more powerful than morphine; ingestion of even a minuscule dose can be fatal. Synthetic cannabinoids primarily are a problem among adolescents; they are more dangerous than marijuana and are associated with aggressive and suicidal behaviors. A standard toxicology screen will not detect synthetic cannabinoids.
E-cigarettes are considered by users to be safer than tobacco cigarettes—and probably are—but they still put patients at risk of nicotine addiction. There are no safety data on e-cigarettes; the devices might contain potentially harmful chemicals and potentially toxic nicotine levels. Dr. Anthenelli reported that topiramate is “the best medication I’ve used” for alcohol abuse disorder. The drug is not FDA-approved for this use, but has been used in a number of studies with positive outcomes.
SATURDAY, APRIL 18, 2015
MORNING SESSION
Psychiatrists are well positioned to help patients with mental illness lose weight because of their psychotherapeutic background. Best treatment strategy is diet plus exercise plus behavioral modification. Robert M. McCarron, DO, University of California, Davis, recommends keeping it simple and telling patients to only consider calories of foods, and not to worry about sodium or fat content. Ask patients “How many minutes a day of exercise can you do?” but recommend that patients walk for 30 minutes a day at 4 mph, 5 days per week, which will help patients lose 1% to 3% of body weight. For treatment-refractory obese patients, consider medications such as bupropion, orlistat, lorcaserin, topiramate, or metformin; for those with a BMI ≥40, recommend bariatric surgery.
George T. Grossberg, MD, Saint Louis University School of Medicine, reviewed the evidence for anxiety disorders in older adults, including generalized anxiety disorder, obsessive-compulsive disorder, panic disorder, and posttraumatic stress disorder. Older patients with cardiovascular disease, cancer, Parkinson’s disease, diabetes, GI disorders, or chronic obstructive pulmonary disease are at high risk of anxiety symptoms. In a study of centenarians, predictors of anxiety are worse health perception, financial concerns related to medical expenses, higher number of medical conditions, and loneliness. Secondary anxiety is prevalent in Alzheimer’s disease; the condition can present as fidgeting, pacing, anger, or agitation, and can be prompted by a change in routine. Acute, new-onset anxiety symptoms should trigger a complete medical evaluation, including a review of medications, supplements, and substance use. In geriatric patients, minimize use of benzodiazepines and avoid anticholinergics.
Overall, psychiatry patients do not receive optimal preventive and primary medical care, leading to decreased life expectancy, often as a result of cardiovascular disease. Psychiatric patients have a high rate of dyslipidemia, hypertension, smoking, and obesity. Psychiatrists often don’t treat these conditions, but they need to be aware of changing standard practices in preventive medicine; be able to recognize a potential problem; and make referrals when appropriate. Dr. McCarron reviewed age-based screening recommendations for hypertension, dyslipidemia, and diabetes from the book Preventive Medical Care in Psychiatry, which he co-edited. He recommends using online cardiovascular risk calculators to determine which patients need to be screened.
AFTERNOON SESSION
Some older patients who abuse substances took drugs as young adults and never gave them up; others have rediscovered drugs in later life. Potential indicators of alcohol abuse in older patients are changes in cognition, mood, memory, hygiene, or sleep. Substance abuse in older adults frequently is comorbid with depression or bereavement, anxiety, and adjustment disorders. Dr. Grossberg recommends addressing the topic directly with patients. Although there are few data to guide treatment, prompt detection and appropriate treatment can improve the quality of life of older adults and their family.
SPONSORS AND SUPPORTERS
• American Professional Agency
• American Psychiatric Publishing
• Arbor Pharmaceuticals
• AstraZeneca
• Banner Health
• Bassett Healthcare Network
• Ministry Health Care
• Pine Rest Christian Mental Health Services
• PRMS
• Sinai Health System
• Sunovion
• Takeda Pharmaceuticals
• U.S. Army Healthcare
• Wexford Health Sources
• Wolters Kluwer Health
The meeting organizers acknowledge the support provided by the sponsors. Determination of educational content for this program and the selection of speakers are responsibilities of the program director and co-directors. Sponsors and supporters did not have input in these areas.
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults
Current Psychiatry welcomed more than 650 psychiatric practitioners from across the United States and abroad to this annual conference, which was headed by Meeting Co-chairs Richard Balon, MD, and Donald W. Black, MD, April 16-18, 2015, at the Hilton Chicago in Chicago, Illinois. Attendees earned as many as 18 AMA PRA Category 1 Credits™. We welcome you to join us next year in Chicago, March 10-12, 2016.
THURSDAY, APRIL 16, 2015
MORNING SESSION
Attention-deficit/hyperactivity disorder (ADHD) is a lifespan disorder that is “everywhere,” Anthony L. Rostain, MD, MA, University of Pennsylvania Perelman School of Medicine, began—including in adults and even “seniors.” This means that the disorder “is not a diagnosis of exclusion,” and that “comorbidity is the rule,” including learning difficulties. Among adults, the focus of symptoms and management is on executive dysfunction and its characteristics: difficulty multitasking, problems keeping commitments, and excessive reliance on help from others. Inattention and disorganization are hallmarks of adult ADHD, and become worse as environmental demands (work, home) increase; hyperactivity decreases with age. Dr. Rostain recommends ruling out other causes of a patient’s symptoms when an adult self-reports ADHD, including transient stressors, medical conditions, psychiatric disorders, and malingering.
Donald W. Black, MD, University of Iowa, reviewed DSM-5 criteria for borderline personality disorder (BPD) and offered tips for avoiding misdiagnosis, including obtaining collateral information and using rating scales. Co-occuring disorders, such as depression and substance abuse, are common. Treatment for BPD patients includes psychotherapy (individual or group), medication, and lifestyle changes. Psychotropics treat symptoms of depression, anxiety, hostility, and impulsivity of BPD but not the fundamental nature of the disorder. When establishing a patient’s treatment plan, consider the stage of illness, evaluate for any co-occurring disorders, and ask the patient what he (she) wants from treatment.
Dr. Rostain began by discussing the neurobiological basis of ADHD, which guides pharmacotherapy. He reviewed the response rate of FDA-approved agents for adults with ADHD, including stimulants, atomoxetine, and alpha-adrenergic agonists. Best response is seen with stimulants, but some patients improve with bupropion and tricyclic antidepressants (TCAs). Employ a multimodal treatment approach, Dr. Rostain recommended, which should include psychoeducation and environmental restructuring, because, as he says, “Pills don’t teach skills.” He also reviewed strategies for treating ADHD in patients who have a comorbid disorder, such as bipolar disorder, major depressive disorder, or substance abuse.
Patients with psychotic depression meet criteria for major depressive disorder but also have delusions or hallucinations. Diagnostic issues include increased guilt, cognitive impairment, paranoia, and increased hopelessness. Anthony J. Rothschild, MD, University of Massachusetts Medical School, reviewed methods for differentiating psychotic depression from schizophrenia, posttraumatic stress disorder, obsessive-compulsive disorder, and body dysmorphic disorder. There are no FDA-approved medications for psychotic depression, Dr. Rothschild explained; however, evidence shows that the combination of an antidepressant and an antipsychotic is superior to monotherapy with an agent from either class. In addition, he noted, studies show a high response rate with electroconvulsive therapy (ECT).
AFTERNOON SESSION
Return of symptoms after initial remission— while the patient is still taking an antidepressant—is considered tachyphylaxis, or “poop out.” Residual depressive symptoms, when a patient meets criteria for remission but still has troubling symptoms, is a different phenomenon, although symptoms can overlap. First, Dr. Rothschild advised, ensure that patients are given an adequate trial of an antidepressant. Options are similar when tachyphylaxis or residual symptoms are present: switch drugs or add augmentation therapy, such as lithium, thyroid hormone, or an atypical antipsychotic. Data on the efficacy for bupropion and buspirone are not strong. For treatment-resistant depression when a patient does not respond to 3 adequate antidepressant trials—consider ECT or rTMS, if available, or a monoamine oxidase inhibitor or a TCA.
Dr. Black defines antisocial personality disorder (ASPD) as a disorder of lifelong serial misbehavior, one characterized by impaired relationships, aggressive behavior, non-aggressive delinquent behavior, manipulation, and a disturbing lack of conscience. There is no standard treatment for ASPD, and no FDA-approved medications; however, potential treatments have not been adequately studied, he pointed out. Cognitive-behavioral therapy might be appropriate in mild cases; some patients benefit from specific programs— for example, ones that address drug or alcohol addiction or anger, although evidence is limited. When treating ASPD patients, Dr. Black concluded, be mindful of high attrition, possible misuse of prescribed medications, and drug-drug or drug-alcohol interactions.
Bipolar disorder is associated with the highest risk of suicide and increased lethality among all psychiatric disorders. Lithium has evidence of an anti-suicidality effect and may reduce suicide by decreasing relapse, aggression, and impulsivity. An FDA advisory on increased risk of suicidality with anticonvulsants was based on data about patients with epilepsy, not bipolar disorder. Second-generation antipsychotics, including olanzapine, quetiapine, and lurasidone, have been shown to be effective for bipolar depression. Avoid antidepressants if possible, Philip G. Janicak, MD, Northwestern University Feinberg School of Medicine, advised; if you must prescribe one, reassess the need for the drug often. Several psychotherapy modalities have evidence supporting their use in bipolar disorder.
FRIDAY, APRIL 17, 2015
MORNING SESSION
Henry A. Nasrallah, MD, Saint Louis University School of Medicine, offered enlightening historical touch-points on how psychiatry’s understanding of, and its approach to, schizophrenia have changed in the past 50 years. His goal? To challenge practitioners to rethink ideas about the disorder and how they care for affected patients. From a laundry list of comparative shifts, here are a few of Dr. Nasrallah’s “then” and “now” observations:
• The old paradigm was: Clinical and functional deterioration are inevitable in schizophrenia. The new paradigm is: Complete remission and restoration of function are feasible in many patients when they are fully adherent to the treatment plan.
• The old: Long-acting injectable (LAI) antipsychotics are a last-resort treatment, to be prescribed after a patient is stabilized. The new: Use LAI antipsychotics early in the course.
• Old: Begin treatment when psychosis appears. New: Work to prevent conversion to psychosis.
• Old: The disorder is considered a consequence of neurochemical dysregulation. New: Impaired neuroplasticity is to blame.
• Old: Treatment is a matter of trial and error. New: We can apply pharmaco-genomics to predict a patient’s response to various drugs and thus increase the likelihood of therapeutic success.
In his second presentation, Dr. Nasrallah described the many pathways to psychosis and several psychotic disorders other than schizophrenia, including schizoaffective, delusional disorder, and psychotic disorder caused by a general medical condition. He listed symptom clusters in psychosis beyond positive and negative symptoms, including neuromotor symptoms, mood symptoms, and neurocognitive deficits. Development of schizophrenia is multifactorial and involves risk genes and environmental factors seen before conception, during birth, and in early childhood; good prenatal care is the best way to prevent schizophrenia, Dr. Nasrallah noted. Several general medical conditions can produce schizophrenia-like psychosis, including some CNS disorders, toxins, autoimmune diseases, infectious diseases, and chromosomal abnormalities. The session concluded with a live interview with one of Dr. Nasrallah’s patients, whose schizophrenia is in remission with clozapine.
Drug abuse can mask signs and symptoms of bipolar disorder, which can delay diagnosis. Commonly abused substances are nicotine, alcohol, Cannabis, and cocaine; polysubstance abuse is the rule. Bipolar disorder and substance abuse share common mechanisms: impulsivity, poor modulation of motivation and response to reward, and behavioral sensitization. Treatment approaches should be flexible. Dr. Janicak reviewed the evidence for using anticonvulsants, antipsychotics, and bupropion for alcohol, Cannabis, and cocaine abuse; there are no data on treating opioid abuse. He also discussed the evidence for using naltrexone, acamprosate, disulfiram, and varenicline, as well as psychotherapeutic options, to treat substance abuse. Dr. Janicak encouraged clinicians in the audience to treat substance abuse in bipolar disorder patients themselves, instead of referring them to a subspecialist.
Untreated psychiatric disorders increase obstetrical complications, possibly through decreased self-care or increased stress. For mild or moderate depression, psychotherapy might be sufficient treatment; but for severe cases, medication is the first-line approach. In her presentation on mood disorders during pregnancy, Marlene P. Freeman, MD, Massachusetts General Hospital, advises that clinicians select medications based on known safety information, patient preference, and the previous course of illness. Results of studies that lasted 4 to 5 years do not show major long-term adverse effects of antidepressant exposure on neurodevelopment or neurobehavior. When treating patients for bipolar disorder, valproate is associated with an increased risk of adverse cognitive and neurodevelopmental effects in infants compared with other anticonvulsants; evidence suggests that lamotrigine is a safer option. The research does not show an increased risk of major malformations with second-generation antipsychotics.
AFTERNOON SESSION
Most women have premenstrual symptoms; a minority have a full-blown syndrome, now known as premenstrual dysphoric disorder (PMDD). This is not an existing mood disorder that becomes worse premenstrually. Clinician and patients should track the temporal relationship of symptoms on a calendar for a few months. Selective serotonin reuptake inhibitors (SSRIs) and venlafaxine have been well studied and are effective compared with placebo, but don’t help all patients with PMDD. Consider flexible dosing strategies with SSRIs—perhaps daily use, a higher dosage premenstrually, and as-needed administration. Start with an oral contraceptive or SSRI; if symptoms don’t respond, add the other. Serotonergic antidepressants have been shown helpful for hot flashes and depressive symptoms in perimenopause. Dr. Freeman reviewed the evidence for using complementary and alternative therapies for menopausal symptoms and hot flushes.
Smoking contributes to excess mortality in seriously mentally ill patients as a result of such tobacco-related illnesses as heart disease, lung disease, and cancer. Overall improvement in mental health as well as physical health is seen when a patient stops smoking. All nicotine replacement products are effective, but patients often don’t use them long enough or correctly. Robert M. Anthenelli, MD, University of California, San Diego, said to begin sustained-release bupropion 1 or 2 weeks before quit date; maintain the dosage for 1 to 12 weeks after quit date and consider maintenance therapy for as long as 6 months. Varenicline is superior to placebo and bupropion, but is known to have gastrointestinal (GI) and sleep disturbance adverse effects. Quitting smoking can increase the blood level of some psychotropics, meaning that you might need to reduce their dosage. It is best to begin smoking cessation when patients are mentally stable, when motivated, and stable on their medications.
In discussing trends in substance abuse, Dr. Anthenelli
faddish. Fentanyl and fentanyl analogues are 100 times more powerful than morphine; ingestion of even a minuscule dose can be fatal. Synthetic cannabinoids primarily are a problem among adolescents; they are more dangerous than marijuana and are associated with aggressive and suicidal behaviors. A standard toxicology screen will not detect synthetic cannabinoids.
E-cigarettes are considered by users to be safer than tobacco cigarettes—and probably are—but they still put patients at risk of nicotine addiction. There are no safety data on e-cigarettes; the devices might contain potentially harmful chemicals and potentially toxic nicotine levels. Dr. Anthenelli reported that topiramate is “the best medication I’ve used” for alcohol abuse disorder. The drug is not FDA-approved for this use, but has been used in a number of studies with positive outcomes.
SATURDAY, APRIL 18, 2015
MORNING SESSION
Psychiatrists are well positioned to help patients with mental illness lose weight because of their psychotherapeutic background. Best treatment strategy is diet plus exercise plus behavioral modification. Robert M. McCarron, DO, University of California, Davis, recommends keeping it simple and telling patients to only consider calories of foods, and not to worry about sodium or fat content. Ask patients “How many minutes a day of exercise can you do?” but recommend that patients walk for 30 minutes a day at 4 mph, 5 days per week, which will help patients lose 1% to 3% of body weight. For treatment-refractory obese patients, consider medications such as bupropion, orlistat, lorcaserin, topiramate, or metformin; for those with a BMI ≥40, recommend bariatric surgery.
George T. Grossberg, MD, Saint Louis University School of Medicine, reviewed the evidence for anxiety disorders in older adults, including generalized anxiety disorder, obsessive-compulsive disorder, panic disorder, and posttraumatic stress disorder. Older patients with cardiovascular disease, cancer, Parkinson’s disease, diabetes, GI disorders, or chronic obstructive pulmonary disease are at high risk of anxiety symptoms. In a study of centenarians, predictors of anxiety are worse health perception, financial concerns related to medical expenses, higher number of medical conditions, and loneliness. Secondary anxiety is prevalent in Alzheimer’s disease; the condition can present as fidgeting, pacing, anger, or agitation, and can be prompted by a change in routine. Acute, new-onset anxiety symptoms should trigger a complete medical evaluation, including a review of medications, supplements, and substance use. In geriatric patients, minimize use of benzodiazepines and avoid anticholinergics.
Overall, psychiatry patients do not receive optimal preventive and primary medical care, leading to decreased life expectancy, often as a result of cardiovascular disease. Psychiatric patients have a high rate of dyslipidemia, hypertension, smoking, and obesity. Psychiatrists often don’t treat these conditions, but they need to be aware of changing standard practices in preventive medicine; be able to recognize a potential problem; and make referrals when appropriate. Dr. McCarron reviewed age-based screening recommendations for hypertension, dyslipidemia, and diabetes from the book Preventive Medical Care in Psychiatry, which he co-edited. He recommends using online cardiovascular risk calculators to determine which patients need to be screened.
AFTERNOON SESSION
Some older patients who abuse substances took drugs as young adults and never gave them up; others have rediscovered drugs in later life. Potential indicators of alcohol abuse in older patients are changes in cognition, mood, memory, hygiene, or sleep. Substance abuse in older adults frequently is comorbid with depression or bereavement, anxiety, and adjustment disorders. Dr. Grossberg recommends addressing the topic directly with patients. Although there are few data to guide treatment, prompt detection and appropriate treatment can improve the quality of life of older adults and their family.
SPONSORS AND SUPPORTERS
• American Professional Agency
• American Psychiatric Publishing
• Arbor Pharmaceuticals
• AstraZeneca
• Banner Health
• Bassett Healthcare Network
• Ministry Health Care
• Pine Rest Christian Mental Health Services
• PRMS
• Sinai Health System
• Sunovion
• Takeda Pharmaceuticals
• U.S. Army Healthcare
• Wexford Health Sources
• Wolters Kluwer Health
The meeting organizers acknowledge the support provided by the sponsors. Determination of educational content for this program and the selection of speakers are responsibilities of the program director and co-directors. Sponsors and supporters did not have input in these areas.
Current Psychiatry welcomed more than 650 psychiatric practitioners from across the United States and abroad to this annual conference, which was headed by Meeting Co-chairs Richard Balon, MD, and Donald W. Black, MD, April 16-18, 2015, at the Hilton Chicago in Chicago, Illinois. Attendees earned as many as 18 AMA PRA Category 1 Credits™. We welcome you to join us next year in Chicago, March 10-12, 2016.
THURSDAY, APRIL 16, 2015
MORNING SESSION
Attention-deficit/hyperactivity disorder (ADHD) is a lifespan disorder that is “everywhere,” Anthony L. Rostain, MD, MA, University of Pennsylvania Perelman School of Medicine, began—including in adults and even “seniors.” This means that the disorder “is not a diagnosis of exclusion,” and that “comorbidity is the rule,” including learning difficulties. Among adults, the focus of symptoms and management is on executive dysfunction and its characteristics: difficulty multitasking, problems keeping commitments, and excessive reliance on help from others. Inattention and disorganization are hallmarks of adult ADHD, and become worse as environmental demands (work, home) increase; hyperactivity decreases with age. Dr. Rostain recommends ruling out other causes of a patient’s symptoms when an adult self-reports ADHD, including transient stressors, medical conditions, psychiatric disorders, and malingering.
Donald W. Black, MD, University of Iowa, reviewed DSM-5 criteria for borderline personality disorder (BPD) and offered tips for avoiding misdiagnosis, including obtaining collateral information and using rating scales. Co-occuring disorders, such as depression and substance abuse, are common. Treatment for BPD patients includes psychotherapy (individual or group), medication, and lifestyle changes. Psychotropics treat symptoms of depression, anxiety, hostility, and impulsivity of BPD but not the fundamental nature of the disorder. When establishing a patient’s treatment plan, consider the stage of illness, evaluate for any co-occurring disorders, and ask the patient what he (she) wants from treatment.
Dr. Rostain began by discussing the neurobiological basis of ADHD, which guides pharmacotherapy. He reviewed the response rate of FDA-approved agents for adults with ADHD, including stimulants, atomoxetine, and alpha-adrenergic agonists. Best response is seen with stimulants, but some patients improve with bupropion and tricyclic antidepressants (TCAs). Employ a multimodal treatment approach, Dr. Rostain recommended, which should include psychoeducation and environmental restructuring, because, as he says, “Pills don’t teach skills.” He also reviewed strategies for treating ADHD in patients who have a comorbid disorder, such as bipolar disorder, major depressive disorder, or substance abuse.
Patients with psychotic depression meet criteria for major depressive disorder but also have delusions or hallucinations. Diagnostic issues include increased guilt, cognitive impairment, paranoia, and increased hopelessness. Anthony J. Rothschild, MD, University of Massachusetts Medical School, reviewed methods for differentiating psychotic depression from schizophrenia, posttraumatic stress disorder, obsessive-compulsive disorder, and body dysmorphic disorder. There are no FDA-approved medications for psychotic depression, Dr. Rothschild explained; however, evidence shows that the combination of an antidepressant and an antipsychotic is superior to monotherapy with an agent from either class. In addition, he noted, studies show a high response rate with electroconvulsive therapy (ECT).
AFTERNOON SESSION
Return of symptoms after initial remission— while the patient is still taking an antidepressant—is considered tachyphylaxis, or “poop out.” Residual depressive symptoms, when a patient meets criteria for remission but still has troubling symptoms, is a different phenomenon, although symptoms can overlap. First, Dr. Rothschild advised, ensure that patients are given an adequate trial of an antidepressant. Options are similar when tachyphylaxis or residual symptoms are present: switch drugs or add augmentation therapy, such as lithium, thyroid hormone, or an atypical antipsychotic. Data on the efficacy for bupropion and buspirone are not strong. For treatment-resistant depression when a patient does not respond to 3 adequate antidepressant trials—consider ECT or rTMS, if available, or a monoamine oxidase inhibitor or a TCA.
Dr. Black defines antisocial personality disorder (ASPD) as a disorder of lifelong serial misbehavior, one characterized by impaired relationships, aggressive behavior, non-aggressive delinquent behavior, manipulation, and a disturbing lack of conscience. There is no standard treatment for ASPD, and no FDA-approved medications; however, potential treatments have not been adequately studied, he pointed out. Cognitive-behavioral therapy might be appropriate in mild cases; some patients benefit from specific programs— for example, ones that address drug or alcohol addiction or anger, although evidence is limited. When treating ASPD patients, Dr. Black concluded, be mindful of high attrition, possible misuse of prescribed medications, and drug-drug or drug-alcohol interactions.
Bipolar disorder is associated with the highest risk of suicide and increased lethality among all psychiatric disorders. Lithium has evidence of an anti-suicidality effect and may reduce suicide by decreasing relapse, aggression, and impulsivity. An FDA advisory on increased risk of suicidality with anticonvulsants was based on data about patients with epilepsy, not bipolar disorder. Second-generation antipsychotics, including olanzapine, quetiapine, and lurasidone, have been shown to be effective for bipolar depression. Avoid antidepressants if possible, Philip G. Janicak, MD, Northwestern University Feinberg School of Medicine, advised; if you must prescribe one, reassess the need for the drug often. Several psychotherapy modalities have evidence supporting their use in bipolar disorder.
FRIDAY, APRIL 17, 2015
MORNING SESSION
Henry A. Nasrallah, MD, Saint Louis University School of Medicine, offered enlightening historical touch-points on how psychiatry’s understanding of, and its approach to, schizophrenia have changed in the past 50 years. His goal? To challenge practitioners to rethink ideas about the disorder and how they care for affected patients. From a laundry list of comparative shifts, here are a few of Dr. Nasrallah’s “then” and “now” observations:
• The old paradigm was: Clinical and functional deterioration are inevitable in schizophrenia. The new paradigm is: Complete remission and restoration of function are feasible in many patients when they are fully adherent to the treatment plan.
• The old: Long-acting injectable (LAI) antipsychotics are a last-resort treatment, to be prescribed after a patient is stabilized. The new: Use LAI antipsychotics early in the course.
• Old: Begin treatment when psychosis appears. New: Work to prevent conversion to psychosis.
• Old: The disorder is considered a consequence of neurochemical dysregulation. New: Impaired neuroplasticity is to blame.
• Old: Treatment is a matter of trial and error. New: We can apply pharmaco-genomics to predict a patient’s response to various drugs and thus increase the likelihood of therapeutic success.
In his second presentation, Dr. Nasrallah described the many pathways to psychosis and several psychotic disorders other than schizophrenia, including schizoaffective, delusional disorder, and psychotic disorder caused by a general medical condition. He listed symptom clusters in psychosis beyond positive and negative symptoms, including neuromotor symptoms, mood symptoms, and neurocognitive deficits. Development of schizophrenia is multifactorial and involves risk genes and environmental factors seen before conception, during birth, and in early childhood; good prenatal care is the best way to prevent schizophrenia, Dr. Nasrallah noted. Several general medical conditions can produce schizophrenia-like psychosis, including some CNS disorders, toxins, autoimmune diseases, infectious diseases, and chromosomal abnormalities. The session concluded with a live interview with one of Dr. Nasrallah’s patients, whose schizophrenia is in remission with clozapine.
Drug abuse can mask signs and symptoms of bipolar disorder, which can delay diagnosis. Commonly abused substances are nicotine, alcohol, Cannabis, and cocaine; polysubstance abuse is the rule. Bipolar disorder and substance abuse share common mechanisms: impulsivity, poor modulation of motivation and response to reward, and behavioral sensitization. Treatment approaches should be flexible. Dr. Janicak reviewed the evidence for using anticonvulsants, antipsychotics, and bupropion for alcohol, Cannabis, and cocaine abuse; there are no data on treating opioid abuse. He also discussed the evidence for using naltrexone, acamprosate, disulfiram, and varenicline, as well as psychotherapeutic options, to treat substance abuse. Dr. Janicak encouraged clinicians in the audience to treat substance abuse in bipolar disorder patients themselves, instead of referring them to a subspecialist.
Untreated psychiatric disorders increase obstetrical complications, possibly through decreased self-care or increased stress. For mild or moderate depression, psychotherapy might be sufficient treatment; but for severe cases, medication is the first-line approach. In her presentation on mood disorders during pregnancy, Marlene P. Freeman, MD, Massachusetts General Hospital, advises that clinicians select medications based on known safety information, patient preference, and the previous course of illness. Results of studies that lasted 4 to 5 years do not show major long-term adverse effects of antidepressant exposure on neurodevelopment or neurobehavior. When treating patients for bipolar disorder, valproate is associated with an increased risk of adverse cognitive and neurodevelopmental effects in infants compared with other anticonvulsants; evidence suggests that lamotrigine is a safer option. The research does not show an increased risk of major malformations with second-generation antipsychotics.
AFTERNOON SESSION
Most women have premenstrual symptoms; a minority have a full-blown syndrome, now known as premenstrual dysphoric disorder (PMDD). This is not an existing mood disorder that becomes worse premenstrually. Clinician and patients should track the temporal relationship of symptoms on a calendar for a few months. Selective serotonin reuptake inhibitors (SSRIs) and venlafaxine have been well studied and are effective compared with placebo, but don’t help all patients with PMDD. Consider flexible dosing strategies with SSRIs—perhaps daily use, a higher dosage premenstrually, and as-needed administration. Start with an oral contraceptive or SSRI; if symptoms don’t respond, add the other. Serotonergic antidepressants have been shown helpful for hot flashes and depressive symptoms in perimenopause. Dr. Freeman reviewed the evidence for using complementary and alternative therapies for menopausal symptoms and hot flushes.
Smoking contributes to excess mortality in seriously mentally ill patients as a result of such tobacco-related illnesses as heart disease, lung disease, and cancer. Overall improvement in mental health as well as physical health is seen when a patient stops smoking. All nicotine replacement products are effective, but patients often don’t use them long enough or correctly. Robert M. Anthenelli, MD, University of California, San Diego, said to begin sustained-release bupropion 1 or 2 weeks before quit date; maintain the dosage for 1 to 12 weeks after quit date and consider maintenance therapy for as long as 6 months. Varenicline is superior to placebo and bupropion, but is known to have gastrointestinal (GI) and sleep disturbance adverse effects. Quitting smoking can increase the blood level of some psychotropics, meaning that you might need to reduce their dosage. It is best to begin smoking cessation when patients are mentally stable, when motivated, and stable on their medications.
In discussing trends in substance abuse, Dr. Anthenelli
faddish. Fentanyl and fentanyl analogues are 100 times more powerful than morphine; ingestion of even a minuscule dose can be fatal. Synthetic cannabinoids primarily are a problem among adolescents; they are more dangerous than marijuana and are associated with aggressive and suicidal behaviors. A standard toxicology screen will not detect synthetic cannabinoids.
E-cigarettes are considered by users to be safer than tobacco cigarettes—and probably are—but they still put patients at risk of nicotine addiction. There are no safety data on e-cigarettes; the devices might contain potentially harmful chemicals and potentially toxic nicotine levels. Dr. Anthenelli reported that topiramate is “the best medication I’ve used” for alcohol abuse disorder. The drug is not FDA-approved for this use, but has been used in a number of studies with positive outcomes.
SATURDAY, APRIL 18, 2015
MORNING SESSION
Psychiatrists are well positioned to help patients with mental illness lose weight because of their psychotherapeutic background. Best treatment strategy is diet plus exercise plus behavioral modification. Robert M. McCarron, DO, University of California, Davis, recommends keeping it simple and telling patients to only consider calories of foods, and not to worry about sodium or fat content. Ask patients “How many minutes a day of exercise can you do?” but recommend that patients walk for 30 minutes a day at 4 mph, 5 days per week, which will help patients lose 1% to 3% of body weight. For treatment-refractory obese patients, consider medications such as bupropion, orlistat, lorcaserin, topiramate, or metformin; for those with a BMI ≥40, recommend bariatric surgery.
George T. Grossberg, MD, Saint Louis University School of Medicine, reviewed the evidence for anxiety disorders in older adults, including generalized anxiety disorder, obsessive-compulsive disorder, panic disorder, and posttraumatic stress disorder. Older patients with cardiovascular disease, cancer, Parkinson’s disease, diabetes, GI disorders, or chronic obstructive pulmonary disease are at high risk of anxiety symptoms. In a study of centenarians, predictors of anxiety are worse health perception, financial concerns related to medical expenses, higher number of medical conditions, and loneliness. Secondary anxiety is prevalent in Alzheimer’s disease; the condition can present as fidgeting, pacing, anger, or agitation, and can be prompted by a change in routine. Acute, new-onset anxiety symptoms should trigger a complete medical evaluation, including a review of medications, supplements, and substance use. In geriatric patients, minimize use of benzodiazepines and avoid anticholinergics.
Overall, psychiatry patients do not receive optimal preventive and primary medical care, leading to decreased life expectancy, often as a result of cardiovascular disease. Psychiatric patients have a high rate of dyslipidemia, hypertension, smoking, and obesity. Psychiatrists often don’t treat these conditions, but they need to be aware of changing standard practices in preventive medicine; be able to recognize a potential problem; and make referrals when appropriate. Dr. McCarron reviewed age-based screening recommendations for hypertension, dyslipidemia, and diabetes from the book Preventive Medical Care in Psychiatry, which he co-edited. He recommends using online cardiovascular risk calculators to determine which patients need to be screened.
AFTERNOON SESSION
Some older patients who abuse substances took drugs as young adults and never gave them up; others have rediscovered drugs in later life. Potential indicators of alcohol abuse in older patients are changes in cognition, mood, memory, hygiene, or sleep. Substance abuse in older adults frequently is comorbid with depression or bereavement, anxiety, and adjustment disorders. Dr. Grossberg recommends addressing the topic directly with patients. Although there are few data to guide treatment, prompt detection and appropriate treatment can improve the quality of life of older adults and their family.
SPONSORS AND SUPPORTERS
• American Professional Agency
• American Psychiatric Publishing
• Arbor Pharmaceuticals
• AstraZeneca
• Banner Health
• Bassett Healthcare Network
• Ministry Health Care
• Pine Rest Christian Mental Health Services
• PRMS
• Sinai Health System
• Sunovion
• Takeda Pharmaceuticals
• U.S. Army Healthcare
• Wexford Health Sources
• Wolters Kluwer Health
The meeting organizers acknowledge the support provided by the sponsors. Determination of educational content for this program and the selection of speakers are responsibilities of the program director and co-directors. Sponsors and supporters did not have input in these areas.
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults