User login
Expanding medication options for pediatric ADHD
Molly, age 9, is diagnosed with attention-deficit/hyperactivity disorder (ADHD) by her psychiatrist, who prescribes a long-acting methylphenidate formulation at 1 mg/kg. She tolerates the medication without side effects and shows significant improvement in her academic performance and on-task behavior in school. Molly takes methylphenidate before school at 7:00 am; this dose usually wears off at approximately 3:30 pm.
Molly and her parents are pleased with her response to methylphenidate, but report that she has difficulty getting ready for school because of distractibility. In the evenings Molly has trouble staying seated to do homework and often interrupts and argues with family members, but cannot tolerate afternoon dosing of immediate-release methylphenidate because of insomnia.
ADHD, the most common childhood neurobehavioral disorder, is characterized by difficulties with attention, impulse control, and modulating activity level. The pathophysiology of ADHD is thought to involve dysregulation of brain dopamine and norepinephrine systems.1 Managing ADHD includes pharmacotherapeutic and nonpharmacotherapeutic—ie, behavioral and psychoeducational—interventions.2,3
In this article, we provide an overview of the efficacy, side effects, and dosing for the 3 classes of ADHD medication—psychostimulants, atomoxetine, and α2 adrenergic agonists—including guidance on medication choice and combination treatment. We also discuss the effects of psychostimulants on tics, cardiovascular concerns, and substance abuse potential.
Psychostimulants
Methylphenidates and amphetamines are first-line agents for ADHD. Their primary mechanism of action involves blocking dopamine transporters, with additional effects including blockade of norepinephrine transporters, dampening action of monoamine oxidase (which slows dopamine and norepinephrine degradation), and enhanced release of dopamine into the synaptic space.1
Efficacy and response rates are similar for methylphenidate and amphetamine medications, although as many as 25% of patients may respond to only 1 agent.1 More than 90% of patients will have a positive response to one of the psychostimulants.1 The beneficial effects of psychostimulants on inattention, hyperactivity, and impulsivity are well documented.2Improvements in noncompliance, aggression, social interactions, and academic productivity also have been observed.4,5
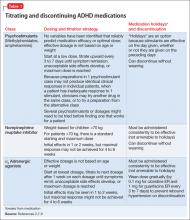
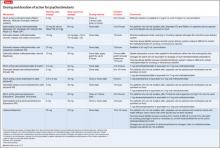
Because of increased recognition of pervasive ADHD-related impairments, which can affect functioning in social, family, and extracurricular settings, practitioners have shifted to long-acting psychostimulants to reduce the need for in-school dosing, improve compliance, and obtain more after-school treatment effects. Long-acting formulations produce a slower rise and fall of psychostimulant levels in the brain, which may decrease side effects and potential for later drug abuse.6 See Table 12,7-9 and Table 22,7,9 for titration, dosing, and duration of action of psychostimulants.
The most common side effects of psychostimulants are appetite loss, abdominal pain, headaches, and sleep disturbances.2 Emotional symptoms—irritability and nervousness—may be observed with psychostimulant use, but these behaviors may improve, rather than become worse, with treatment.5 Methylphenidates and amphetamines share many of the same side effects,2 with many studies indicating no differences between their side-effect profiles.1 Other studies indicate that sleep and emotional side effects may be more prominent with amphetamines than methylphenidates,10 although response varies by individual.
There is little evidence that methylphenidate, low-dose amphetamine, or low-dose dextroamphetamine makes tics worse in most children who have them, although significant tic exacerbation has been observed with higher-dose dextroamphetamine.11,12 In patients with comorbid ADHD and tic disorders, a trial of psychostimulants with monitoring for worsening tics is appropriate.
Changes in heart rate and blood pressure generally are not clinically significant in patients taking psychostimulants (average increases: 1 or 2 beats per minute and 1 to 4 mm Hg for systolic and diastolic blood pressures).12 However, psychostimulants may be associated with more substantial increases in heart rate and blood pressure in a subset of individuals (5% to 15%).12 Large studies of children and adults in the general population have not found an association between psychostimulant use and severe cardiovascular events (sudden cardiac death, myocardial infarction, stroke).12-14 Because of reports of sudden cardiac death in children with underlying heart disease who take a psychostimulant,15 clinicians are advised to screen patients and consider an electrocardiogram or evaluation by a cardiologist before starting a psychostimulant in a patient who has a personal or family history of specific cardiovascular risk factors (see Perrin et al16 and Cortese et al12 for screening questions and conditions).
Modest reductions in height (1 or 2 cm after 3 years of psychostimulant treatment) appear to be dose-dependent, and are similar across the methylphenidate and amphetamine classes. Some studies have shown reversal of growth deficits after treatment is stopped treatment and no adverse effects on final adult height.12,17 More study is needed to clarify the effects of continuous psychostimulant treatment from childhood to adulthood on growth.
Studies have failed to show an increased risk of substance abuse in persons with ADHD who were treated with psychostimulants during childhood. Some studies document a lower rate of later substance abuse in youths who received ADHD medications, although other reports show no effect of psychostimulant treatment on subsequent substance use disorder risk.12 Be aware that psychostimulants can be misused (eg, to get “high,” for performance enhancement, to suppress appetite, etc.). Misuse of psychostimulants is most common with short-acting preparations, and generally more difficult with long-acting preparations because extracting the active ingredients for snorting is difficult.2,12 Monitor refill requests and patient behavior for signs of misuse, and be alert for signs of illegal drug use in the patient’s family.
Psychotic symptoms—including hallucinations, delusions, mania, and extreme agitation—with psychostimulant treatment are rare, occurring at a rate of 1.5%.12
Atomoxetine
Approved by the FDA in 2002 for ADHD, atomoxetine is effective and generally well tolerated, although it is not as effective as psychostimulants.2 Atomoxetine is a potent norepinephrine reuptake inhibitor18 that does not produce euphoria, does not have potential for abuse, and has not been linked to increased tic onset or severity.19 Atomoxetine treatment is associated with a lower rate of sleep initiation difficulty compared with psychostimulants.18 Some studies suggest that atomoxetine may have mild beneficial effects on anxiety disorders,18 making it a reasonable choice for patients with significant anxiety or insomnia during psychostimulant treatment. Table 12,7-9 and Table 32,7,9 include information on dosing and duration of action for atomoxetine.
Common side effects of atomoxetine include sedation and fatigue, upset stomach, nausea and vomiting, reduced appetite, headache, and irritability.18 Inform patients that atomoxetine carries an FDA black-box warning for suicide risk; a review of 14 studies showed suicidal ideation was more common with atomoxetine than placebo, although no suicides occurred in any trials.20
Hepatotoxicity is rare with atomoxetine.21 Although routine liver enzyme testing is not required, discontinue atomoxetine if jaundice develops or elevated levels of liver enzymes are noted. Other rare but potentially serious side effects include changes in heart rate (≥20 beats per min) or blood pressure that occur in 5% to 10% of patients taking atomoxetine.22 The risk of serious cardiovascular events and sudden cardiac death with atomoxetine is extremely low, but patients should be screened for a personal and family history of cardiovascular risk factors and, if any of these are present, evaluated further before starting atomoxetine. Routine heart rate and blood pressure monitoring is recommended for all patients.12-14,16
Last, atomoxetine has been linked to growth delays in the first 1 or 2 years of treatment, with a return to expected measurements after an average 2 or 3 years of treatment; persistent decreases in growth rate were observed in patients who were taller or heavier than average before treatment.23
α2 Adrenergic agonists
Guanfacine ER and clonidine ER, the extended release (ER) formulations of α2 adrenergic agonists, were FDA-approved for treating ADHD in 2009 and 2010, respectively. Short-acting guanfacine and clonidine also are used for treating ADHD.24 Their mechanism of action involves stimulation of the pre-synaptic and post-synapic α2 adrenergic receptors, which control the release of norepinephrine and the rate of cell firing.25 The α2 agonists are considered a second-line treatment for ADHD because their efficacy and response rate for core ADHD symptoms lags behind those of psychostimulants.25 In addition to treating core ADHD symptoms, guanfacine and clonidine are used to treat tics and oppositional/aggressive behavior comorbid with ADHD.24,26 Clonidine, which is more sedating than guanfacine, can be used to treat comorbid ADHD and sleep disorders.24 The α2 agonists do not produce euphoria and do not have drug abuse potential.2Table 12,7-9 and Table 32,7,9 provide guidelines for prescribing guanfacine ER and clonidine ER.
The most common adverse effect is drowsiness; other common side effects include dizziness, irritability, headache, and abdominal pain.24 Short-term studies of α2 agonist treatment of ADHD have shown small, non-clinically significant reductions in heart rate and blood pressure; α2 agonist-associated bradycardia, increased QT interval, and cardiac arrhythmias have been reported,7,24,27 as well as rebound hypertension with abrupt discontinuation.24 Screen patients for a personal and family history of cardiovascular risk factors and, if present, evaluate further before initiating α2 agonists.
Combining ADHD medication classes
Combination therapy with >1 ADHD medications is employed when 1 class does not provide adequate symptom coverage or produces problematic side effects.8,24 Psychostimulants can be combined with low-dose atomoxetine (0.5 to 1.0 mg/kg/d) when atomoxetine does not adequately cover ADHD symptoms in school, or when psychostimulants do not adequately cover evening symptoms or patients experience problems with evening psychostimulant rebound.8 To date, prospective data on the safety and efficacy of combining atomoxetine and psychostimulants are limited, but what evidence is available suggests improved symptom control for some, but not all, patients, and a lack of serious adverse events.28
Psychostimulants have been combined with α2agonists when children have an inadequate response to psychostimulants alone, or in cases of ADHD comorbid with aggression or tics.24 Although early case reports raised concern about the safety of combining psychostimulants and α2 agonists, subsequent studies suggest that clonidine and guanfacine generally are well-tolerated when co-administered with psychostimulants.24,27,29
Case continued
Molly has derived substantial benefit from long-acting methylphenidate during the school day, but continues to have significant ADHD-related impairment in the mornings and evenings. Her physician tried afternoon dosing of immediate-release methylphenidate to address evening difficulties, but Molly experienced insomnia. It would be reasonable to consider adjunctive therapy with a non-stimulant medication. A medication that can provide round-the-clock ADHD symptom coverage—such as atomoxetine, guanfacine ER, or clonidine ER—could be added to her current day-time psychostimulant treatment, potentially improving her functioning at home before school and in the evenings.
Additional considerations
Combining medication and behavior therapy offers greater improvements on academic, conduct, and family satisfaction measures than either treatment alone.2 Clinicians can choose to employ behavior therapy alone, particularly if parents feel uncomfortable with—or children have not tolerated—medication.2,3 Evidence-based behavioral parent training and classroom management strategies (implemented by teachers) have shown the strongest and most consistent effects among nonpharmacotherapeutic interventions for ADHD.2 Most studies comparing behavior therapy to psychostimulants have found a stronger effect on core ADHD symptoms from psychostimulants than from behavior therapy.
When a patient does not respond adequately to FDA-approved ADHD medications alone or in combination, consider bupropion, an antidepressant with indirect dopamine and noradrenergic effects. Off-label bupropion has been shown to be effective for ADHD in controlled trials of both children and adults.30
Clinicians often encounter children who meet criteria for ADHD and an anxiety or mood disorder. Table 48,31 summarizes treatment recommendations for these patients.
Clinical considerations
- Begin treatment with a psychostimulant at a low dosage, and titrate gradually until symptoms are controlled or side effects develop.
- Keep in mind that an effective dosage of a psychostimulant is not closely correlated with age, weight, or severity of symptoms.
- Monitor refill requests and patient behavior for signs of psychostimulant misuse. Be alert for signs of illegal drug use in patient family members.
- Lisdexamfetamine, dermal methylphenidate, and osmotic release oral system methylphenidate are the formulations least likely to be misused because their delivery systems make it difficult to extract the active ingredient for snorting or intravenous injection.
- Psychostimulants have not been shown to exacerbate tics in most children who have comorbid ADHD and a tic disorder. When a stimulant is associated with an exacerbation of tics, switching treatment to atomoxetine or α2 agonists is reasonable.
- For patients whose use of a stimulant is limited by an adverse effect on sleep, consider atomoxetine and α2 adrenergic agonists as alternative or adjunctive treatments.
- All 3 classes of FDA-approved ADHD medications (psychostimulants, atomoxetine, and adrenergic agonists) have been associated with adverse cardiac events in children who have underlying cardiovascular conditions. Before initiating treatment, screen patients for a personal or family history of cardiovascular risk factors, and undertake further evaluation as indicated.
Bottom Line
In general, the evidence supports psychostimulants as initial pharmacotherapy for ADHD, with additional options including atomoxetine and α2 agonists. When one medication class does not provide adequate coverage for ADHD symptoms, combining medication classes can be beneficial.
Related Resources
- National Institute of Mental Health. What is attention deficit hyperactivity disorder (ADHD, ADD)?” www.nimh.nih.gov/health/topics/attention-deficit-hyperactivity-disorder-adhd/index.shtml.
- National Resource Center on AD/HD. Managing medication for children and adolescents with ADHD. www.help4adhd.org/en/treatment/medication/WWK3.
Drug Brand Names
Atomoxetine • Strattera
Lisdexamfetamine • Vyvanse
Bupropion • Wellbutrin, Zyban
Clonidine extended release • Kapvay
Guanfacine extended release • Intuniv
Dexmethylphenidate • Focalin, Focalin XR
Mixed amphetamine salts • Adderall, Adderall XR
Dextroamphetamine • Dexedrine, Dexedrine SR, DextroStat, ProCentra
Methylphenidate • Ritalin, Methylin, Metadate CD, Metadate ER, Methylin ER, Ritalin LA, Ritalin SR, Concerta, Quillivant XR, Daytrana
Disclosures
Dr. Froehlich receives support from the National Institute of Mental Health Grant K23 MH083881. Dr. Delgado has received research support from Pfizer, Inc. Dr. Anixt reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Solanto MV. Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res. 1998; 94(1):127-152.
2. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management; Wolraich M, Brown L, Brown RT, et al. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
3. Pliszka S; AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
4. Zametkin AJ, Ernst M. Problems in the management of attention-deficit-hyperactivity disorder. N Engl J Med. 1999;340(1):40-46.
5. Goldman LS, Genel M, Bezman RJ, et al. Diagnosis and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Council on Scientific Affairs, American Medical Association. JAMA. 1998;279(14):1100-1107.
6. Swanson J, Gupta S, Lam A, et al. Development of a new once-a-day formulation of methylphenidate for the treatment of attention-deficit/hyperactivity disorder: proof-of-concept and proof-of-product studies. Arch Gen Psychiatry. 2003;60(2):204-211.
7. Vaughan B, Kratochvil CJ. Pharmacotherapy of pediatric attention-deficit/hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2012;21(4):941-955.
8. Pliszka SR, Crismon ML, Hughes CW, et al; Texas Consensus Conference Panel on Pharmacotherapy of Childhood Attention Deficit Hyperactivity Disorder. The Texas Children’s Medication Algorithm Project: revision of the algorithm for pharmacotherapy of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2006;45(6):642-657.
9. Antshel KM, Hargrave TM, Simonescu M, et al. Advances in understanding and treating ADHD. BMC Med. 2011;9:72.
10. Efron D, Jarman F, Barker M. Side effects of methylphenidate and dexamphetamine in children with attention deficit hyperactivity disorder: a double-blind, crossover trial. Pediatrics. 1997;100(4):662-666.
11. Pringsheim T, Steeves T. Pharmacological treatment for attention deficit hyperactivity disorder (ADHD) in children with comorbid tic disorders. Cochrane Database Syst Rev. 2011(4):CD007990.
12. Cortese S, Holtmann M, Banaschewski T, et al. Practitioner review: current best practice in the management of adverse events during treatment with ADHD medications in children and adolescents. J Child Psychol Psychiatry. 2013; 54(3):227-246.
13. Cooper WO, Habel LA, Sox CM, et al. ADHD drugs and serious cardiovascular events in children and young adults. N Engl J Med. 2011;365(20):1896-1904.
14. Martinez-Raga J, Knecht C, Szerman N, et al. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs. 2013;27(1):15-30.
15. Vetter VL, Elia J, Erickson C, et al; American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee; American Heart Association Council on Cardiovascular Nursing. Cardiovascular monitoring of children and adolescents with heart disease receiving medications for attention deficit/hyperactivity disorder [corrected]: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee and the Council on Cardiovascular Nursing. Circulation. 2008;117(18):2407-2423.
16. Perrin JM, Friedman RA, Knilans TK; Black Box Working Group; Section on Cardiology and Cardiac Surgery. Cardiovascular monitoring and stimulant drugs for attention-deficit/hyperactivity disorder. Pediatrics. 2008;122(2):451-453.
17. Faraone SV, Biederman J, Morley CP, et al. Effect of stimulants on height and weight: a review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(9):994-1009.
18. Garnock-Jones KP, Keating GM. Atomoxetine: a review of its use in attention-deficit hyperactivity disorder in children and adolescents. Paediatr Drugs. 2009;11(3):203-226.
19. Bymaster FP, Katner JS, Nelson DL, et al. Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology. 2002;27(5):699-711.
20. Bangs ME, Tauscher-Wisniewski S, Polzer J, et al. Meta-analysis of suicide-related behavior events in patients treated with atomoxetine. J Am Acad Child Adolesc Psychiatry. 2008;47(2):209-218.
21. Bangs ME, Jin L, Zhang S, et al. Hepatic events associated with atomoxetine treatment for attention-deficit hyperactivity disorder. Drug Saf. 2008;31(4):345-354.
22. U.S. Food and Drug Administration. Strattera (atomoxetine hydrochloride) capsule. http://www.fda.gov/Safety/MedWatch/SafetyInformation/ucm223889.htm. Published August 2013. Accessed October 31, 2013.
23. Spencer TJ, Kratochvil CJ, Sangal RB, et al. Effects of atomoxetine on growth in children with attention-deficit/hyperactivity disorder following up to five years of treatment. J Child Adolesc Psychopharmacol. 2007;17(5):689-700.
24. Connor DF. Other medications. In: Barkley RA, ed. Attention-deficit/hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:658-677.
25. May DE, Kratochvil CJ. Attention-deficit hyperactivity disorder: recent advances in paediatric pharmacotherapy. Drugs. 2010;70(1):15-40.
26. Connor DF, Findling RL, Kollins SH, et al. Effects of guanfacine extended release on oppositional symptoms in children aged 6-12 years with attention-deficit hyperactivity disorder and oppositional symptoms: a randomized, double-blind, placebo-controlled trial. CNS Drugs. 2010; 24(9):755-768.
27. Croxtall JD. Clonidine extended-release: in attention-deficit hyperactivity disorder. Paediatr Drugs. 2011;13(5):329-336.
28. Treuer T, Gau SS, Mendez L, et al. A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability. J Child Adolesc Psychopharmacol. 2013;23(3):179-193.
29. Sallee FR. The role of alpha2-adrenergic agonists in attention-deficit/hyperactivity disorder. Postgrad Med. 2010;122(5):78-87.
30. Spencer TJ. Antidepressant and specific norepinephrine reuptake inhibitor treatments. In: Barkley RA, ed. Attention-deficit hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:648-657.
31. Singh MK, DelBello MP, Kowatch RA, et al. Co-occurrence of bipolar and attention-deficit hyperactivity disorders in children. Bipolar Disord. 2006;8(6):710-720.
Molly, age 9, is diagnosed with attention-deficit/hyperactivity disorder (ADHD) by her psychiatrist, who prescribes a long-acting methylphenidate formulation at 1 mg/kg. She tolerates the medication without side effects and shows significant improvement in her academic performance and on-task behavior in school. Molly takes methylphenidate before school at 7:00 am; this dose usually wears off at approximately 3:30 pm.
Molly and her parents are pleased with her response to methylphenidate, but report that she has difficulty getting ready for school because of distractibility. In the evenings Molly has trouble staying seated to do homework and often interrupts and argues with family members, but cannot tolerate afternoon dosing of immediate-release methylphenidate because of insomnia.
ADHD, the most common childhood neurobehavioral disorder, is characterized by difficulties with attention, impulse control, and modulating activity level. The pathophysiology of ADHD is thought to involve dysregulation of brain dopamine and norepinephrine systems.1 Managing ADHD includes pharmacotherapeutic and nonpharmacotherapeutic—ie, behavioral and psychoeducational—interventions.2,3
In this article, we provide an overview of the efficacy, side effects, and dosing for the 3 classes of ADHD medication—psychostimulants, atomoxetine, and α2 adrenergic agonists—including guidance on medication choice and combination treatment. We also discuss the effects of psychostimulants on tics, cardiovascular concerns, and substance abuse potential.
Psychostimulants
Methylphenidates and amphetamines are first-line agents for ADHD. Their primary mechanism of action involves blocking dopamine transporters, with additional effects including blockade of norepinephrine transporters, dampening action of monoamine oxidase (which slows dopamine and norepinephrine degradation), and enhanced release of dopamine into the synaptic space.1
Efficacy and response rates are similar for methylphenidate and amphetamine medications, although as many as 25% of patients may respond to only 1 agent.1 More than 90% of patients will have a positive response to one of the psychostimulants.1 The beneficial effects of psychostimulants on inattention, hyperactivity, and impulsivity are well documented.2Improvements in noncompliance, aggression, social interactions, and academic productivity also have been observed.4,5
Because of increased recognition of pervasive ADHD-related impairments, which can affect functioning in social, family, and extracurricular settings, practitioners have shifted to long-acting psychostimulants to reduce the need for in-school dosing, improve compliance, and obtain more after-school treatment effects. Long-acting formulations produce a slower rise and fall of psychostimulant levels in the brain, which may decrease side effects and potential for later drug abuse.6 See Table 12,7-9 and Table 22,7,9 for titration, dosing, and duration of action of psychostimulants.
The most common side effects of psychostimulants are appetite loss, abdominal pain, headaches, and sleep disturbances.2 Emotional symptoms—irritability and nervousness—may be observed with psychostimulant use, but these behaviors may improve, rather than become worse, with treatment.5 Methylphenidates and amphetamines share many of the same side effects,2 with many studies indicating no differences between their side-effect profiles.1 Other studies indicate that sleep and emotional side effects may be more prominent with amphetamines than methylphenidates,10 although response varies by individual.
There is little evidence that methylphenidate, low-dose amphetamine, or low-dose dextroamphetamine makes tics worse in most children who have them, although significant tic exacerbation has been observed with higher-dose dextroamphetamine.11,12 In patients with comorbid ADHD and tic disorders, a trial of psychostimulants with monitoring for worsening tics is appropriate.
Changes in heart rate and blood pressure generally are not clinically significant in patients taking psychostimulants (average increases: 1 or 2 beats per minute and 1 to 4 mm Hg for systolic and diastolic blood pressures).12 However, psychostimulants may be associated with more substantial increases in heart rate and blood pressure in a subset of individuals (5% to 15%).12 Large studies of children and adults in the general population have not found an association between psychostimulant use and severe cardiovascular events (sudden cardiac death, myocardial infarction, stroke).12-14 Because of reports of sudden cardiac death in children with underlying heart disease who take a psychostimulant,15 clinicians are advised to screen patients and consider an electrocardiogram or evaluation by a cardiologist before starting a psychostimulant in a patient who has a personal or family history of specific cardiovascular risk factors (see Perrin et al16 and Cortese et al12 for screening questions and conditions).
Modest reductions in height (1 or 2 cm after 3 years of psychostimulant treatment) appear to be dose-dependent, and are similar across the methylphenidate and amphetamine classes. Some studies have shown reversal of growth deficits after treatment is stopped treatment and no adverse effects on final adult height.12,17 More study is needed to clarify the effects of continuous psychostimulant treatment from childhood to adulthood on growth.
Studies have failed to show an increased risk of substance abuse in persons with ADHD who were treated with psychostimulants during childhood. Some studies document a lower rate of later substance abuse in youths who received ADHD medications, although other reports show no effect of psychostimulant treatment on subsequent substance use disorder risk.12 Be aware that psychostimulants can be misused (eg, to get “high,” for performance enhancement, to suppress appetite, etc.). Misuse of psychostimulants is most common with short-acting preparations, and generally more difficult with long-acting preparations because extracting the active ingredients for snorting is difficult.2,12 Monitor refill requests and patient behavior for signs of misuse, and be alert for signs of illegal drug use in the patient’s family.
Psychotic symptoms—including hallucinations, delusions, mania, and extreme agitation—with psychostimulant treatment are rare, occurring at a rate of 1.5%.12
Atomoxetine
Approved by the FDA in 2002 for ADHD, atomoxetine is effective and generally well tolerated, although it is not as effective as psychostimulants.2 Atomoxetine is a potent norepinephrine reuptake inhibitor18 that does not produce euphoria, does not have potential for abuse, and has not been linked to increased tic onset or severity.19 Atomoxetine treatment is associated with a lower rate of sleep initiation difficulty compared with psychostimulants.18 Some studies suggest that atomoxetine may have mild beneficial effects on anxiety disorders,18 making it a reasonable choice for patients with significant anxiety or insomnia during psychostimulant treatment. Table 12,7-9 and Table 32,7,9 include information on dosing and duration of action for atomoxetine.
Common side effects of atomoxetine include sedation and fatigue, upset stomach, nausea and vomiting, reduced appetite, headache, and irritability.18 Inform patients that atomoxetine carries an FDA black-box warning for suicide risk; a review of 14 studies showed suicidal ideation was more common with atomoxetine than placebo, although no suicides occurred in any trials.20
Hepatotoxicity is rare with atomoxetine.21 Although routine liver enzyme testing is not required, discontinue atomoxetine if jaundice develops or elevated levels of liver enzymes are noted. Other rare but potentially serious side effects include changes in heart rate (≥20 beats per min) or blood pressure that occur in 5% to 10% of patients taking atomoxetine.22 The risk of serious cardiovascular events and sudden cardiac death with atomoxetine is extremely low, but patients should be screened for a personal and family history of cardiovascular risk factors and, if any of these are present, evaluated further before starting atomoxetine. Routine heart rate and blood pressure monitoring is recommended for all patients.12-14,16
Last, atomoxetine has been linked to growth delays in the first 1 or 2 years of treatment, with a return to expected measurements after an average 2 or 3 years of treatment; persistent decreases in growth rate were observed in patients who were taller or heavier than average before treatment.23
α2 Adrenergic agonists
Guanfacine ER and clonidine ER, the extended release (ER) formulations of α2 adrenergic agonists, were FDA-approved for treating ADHD in 2009 and 2010, respectively. Short-acting guanfacine and clonidine also are used for treating ADHD.24 Their mechanism of action involves stimulation of the pre-synaptic and post-synapic α2 adrenergic receptors, which control the release of norepinephrine and the rate of cell firing.25 The α2 agonists are considered a second-line treatment for ADHD because their efficacy and response rate for core ADHD symptoms lags behind those of psychostimulants.25 In addition to treating core ADHD symptoms, guanfacine and clonidine are used to treat tics and oppositional/aggressive behavior comorbid with ADHD.24,26 Clonidine, which is more sedating than guanfacine, can be used to treat comorbid ADHD and sleep disorders.24 The α2 agonists do not produce euphoria and do not have drug abuse potential.2Table 12,7-9 and Table 32,7,9 provide guidelines for prescribing guanfacine ER and clonidine ER.
The most common adverse effect is drowsiness; other common side effects include dizziness, irritability, headache, and abdominal pain.24 Short-term studies of α2 agonist treatment of ADHD have shown small, non-clinically significant reductions in heart rate and blood pressure; α2 agonist-associated bradycardia, increased QT interval, and cardiac arrhythmias have been reported,7,24,27 as well as rebound hypertension with abrupt discontinuation.24 Screen patients for a personal and family history of cardiovascular risk factors and, if present, evaluate further before initiating α2 agonists.
Combining ADHD medication classes
Combination therapy with >1 ADHD medications is employed when 1 class does not provide adequate symptom coverage or produces problematic side effects.8,24 Psychostimulants can be combined with low-dose atomoxetine (0.5 to 1.0 mg/kg/d) when atomoxetine does not adequately cover ADHD symptoms in school, or when psychostimulants do not adequately cover evening symptoms or patients experience problems with evening psychostimulant rebound.8 To date, prospective data on the safety and efficacy of combining atomoxetine and psychostimulants are limited, but what evidence is available suggests improved symptom control for some, but not all, patients, and a lack of serious adverse events.28
Psychostimulants have been combined with α2agonists when children have an inadequate response to psychostimulants alone, or in cases of ADHD comorbid with aggression or tics.24 Although early case reports raised concern about the safety of combining psychostimulants and α2 agonists, subsequent studies suggest that clonidine and guanfacine generally are well-tolerated when co-administered with psychostimulants.24,27,29
Case continued
Molly has derived substantial benefit from long-acting methylphenidate during the school day, but continues to have significant ADHD-related impairment in the mornings and evenings. Her physician tried afternoon dosing of immediate-release methylphenidate to address evening difficulties, but Molly experienced insomnia. It would be reasonable to consider adjunctive therapy with a non-stimulant medication. A medication that can provide round-the-clock ADHD symptom coverage—such as atomoxetine, guanfacine ER, or clonidine ER—could be added to her current day-time psychostimulant treatment, potentially improving her functioning at home before school and in the evenings.
Additional considerations
Combining medication and behavior therapy offers greater improvements on academic, conduct, and family satisfaction measures than either treatment alone.2 Clinicians can choose to employ behavior therapy alone, particularly if parents feel uncomfortable with—or children have not tolerated—medication.2,3 Evidence-based behavioral parent training and classroom management strategies (implemented by teachers) have shown the strongest and most consistent effects among nonpharmacotherapeutic interventions for ADHD.2 Most studies comparing behavior therapy to psychostimulants have found a stronger effect on core ADHD symptoms from psychostimulants than from behavior therapy.
When a patient does not respond adequately to FDA-approved ADHD medications alone or in combination, consider bupropion, an antidepressant with indirect dopamine and noradrenergic effects. Off-label bupropion has been shown to be effective for ADHD in controlled trials of both children and adults.30
Clinicians often encounter children who meet criteria for ADHD and an anxiety or mood disorder. Table 48,31 summarizes treatment recommendations for these patients.
Clinical considerations
- Begin treatment with a psychostimulant at a low dosage, and titrate gradually until symptoms are controlled or side effects develop.
- Keep in mind that an effective dosage of a psychostimulant is not closely correlated with age, weight, or severity of symptoms.
- Monitor refill requests and patient behavior for signs of psychostimulant misuse. Be alert for signs of illegal drug use in patient family members.
- Lisdexamfetamine, dermal methylphenidate, and osmotic release oral system methylphenidate are the formulations least likely to be misused because their delivery systems make it difficult to extract the active ingredient for snorting or intravenous injection.
- Psychostimulants have not been shown to exacerbate tics in most children who have comorbid ADHD and a tic disorder. When a stimulant is associated with an exacerbation of tics, switching treatment to atomoxetine or α2 agonists is reasonable.
- For patients whose use of a stimulant is limited by an adverse effect on sleep, consider atomoxetine and α2 adrenergic agonists as alternative or adjunctive treatments.
- All 3 classes of FDA-approved ADHD medications (psychostimulants, atomoxetine, and adrenergic agonists) have been associated with adverse cardiac events in children who have underlying cardiovascular conditions. Before initiating treatment, screen patients for a personal or family history of cardiovascular risk factors, and undertake further evaluation as indicated.
Bottom Line
In general, the evidence supports psychostimulants as initial pharmacotherapy for ADHD, with additional options including atomoxetine and α2 agonists. When one medication class does not provide adequate coverage for ADHD symptoms, combining medication classes can be beneficial.
Related Resources
- National Institute of Mental Health. What is attention deficit hyperactivity disorder (ADHD, ADD)?” www.nimh.nih.gov/health/topics/attention-deficit-hyperactivity-disorder-adhd/index.shtml.
- National Resource Center on AD/HD. Managing medication for children and adolescents with ADHD. www.help4adhd.org/en/treatment/medication/WWK3.
Drug Brand Names
Atomoxetine • Strattera
Lisdexamfetamine • Vyvanse
Bupropion • Wellbutrin, Zyban
Clonidine extended release • Kapvay
Guanfacine extended release • Intuniv
Dexmethylphenidate • Focalin, Focalin XR
Mixed amphetamine salts • Adderall, Adderall XR
Dextroamphetamine • Dexedrine, Dexedrine SR, DextroStat, ProCentra
Methylphenidate • Ritalin, Methylin, Metadate CD, Metadate ER, Methylin ER, Ritalin LA, Ritalin SR, Concerta, Quillivant XR, Daytrana
Disclosures
Dr. Froehlich receives support from the National Institute of Mental Health Grant K23 MH083881. Dr. Delgado has received research support from Pfizer, Inc. Dr. Anixt reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Molly, age 9, is diagnosed with attention-deficit/hyperactivity disorder (ADHD) by her psychiatrist, who prescribes a long-acting methylphenidate formulation at 1 mg/kg. She tolerates the medication without side effects and shows significant improvement in her academic performance and on-task behavior in school. Molly takes methylphenidate before school at 7:00 am; this dose usually wears off at approximately 3:30 pm.
Molly and her parents are pleased with her response to methylphenidate, but report that she has difficulty getting ready for school because of distractibility. In the evenings Molly has trouble staying seated to do homework and often interrupts and argues with family members, but cannot tolerate afternoon dosing of immediate-release methylphenidate because of insomnia.
ADHD, the most common childhood neurobehavioral disorder, is characterized by difficulties with attention, impulse control, and modulating activity level. The pathophysiology of ADHD is thought to involve dysregulation of brain dopamine and norepinephrine systems.1 Managing ADHD includes pharmacotherapeutic and nonpharmacotherapeutic—ie, behavioral and psychoeducational—interventions.2,3
In this article, we provide an overview of the efficacy, side effects, and dosing for the 3 classes of ADHD medication—psychostimulants, atomoxetine, and α2 adrenergic agonists—including guidance on medication choice and combination treatment. We also discuss the effects of psychostimulants on tics, cardiovascular concerns, and substance abuse potential.
Psychostimulants
Methylphenidates and amphetamines are first-line agents for ADHD. Their primary mechanism of action involves blocking dopamine transporters, with additional effects including blockade of norepinephrine transporters, dampening action of monoamine oxidase (which slows dopamine and norepinephrine degradation), and enhanced release of dopamine into the synaptic space.1
Efficacy and response rates are similar for methylphenidate and amphetamine medications, although as many as 25% of patients may respond to only 1 agent.1 More than 90% of patients will have a positive response to one of the psychostimulants.1 The beneficial effects of psychostimulants on inattention, hyperactivity, and impulsivity are well documented.2Improvements in noncompliance, aggression, social interactions, and academic productivity also have been observed.4,5
Because of increased recognition of pervasive ADHD-related impairments, which can affect functioning in social, family, and extracurricular settings, practitioners have shifted to long-acting psychostimulants to reduce the need for in-school dosing, improve compliance, and obtain more after-school treatment effects. Long-acting formulations produce a slower rise and fall of psychostimulant levels in the brain, which may decrease side effects and potential for later drug abuse.6 See Table 12,7-9 and Table 22,7,9 for titration, dosing, and duration of action of psychostimulants.
The most common side effects of psychostimulants are appetite loss, abdominal pain, headaches, and sleep disturbances.2 Emotional symptoms—irritability and nervousness—may be observed with psychostimulant use, but these behaviors may improve, rather than become worse, with treatment.5 Methylphenidates and amphetamines share many of the same side effects,2 with many studies indicating no differences between their side-effect profiles.1 Other studies indicate that sleep and emotional side effects may be more prominent with amphetamines than methylphenidates,10 although response varies by individual.
There is little evidence that methylphenidate, low-dose amphetamine, or low-dose dextroamphetamine makes tics worse in most children who have them, although significant tic exacerbation has been observed with higher-dose dextroamphetamine.11,12 In patients with comorbid ADHD and tic disorders, a trial of psychostimulants with monitoring for worsening tics is appropriate.
Changes in heart rate and blood pressure generally are not clinically significant in patients taking psychostimulants (average increases: 1 or 2 beats per minute and 1 to 4 mm Hg for systolic and diastolic blood pressures).12 However, psychostimulants may be associated with more substantial increases in heart rate and blood pressure in a subset of individuals (5% to 15%).12 Large studies of children and adults in the general population have not found an association between psychostimulant use and severe cardiovascular events (sudden cardiac death, myocardial infarction, stroke).12-14 Because of reports of sudden cardiac death in children with underlying heart disease who take a psychostimulant,15 clinicians are advised to screen patients and consider an electrocardiogram or evaluation by a cardiologist before starting a psychostimulant in a patient who has a personal or family history of specific cardiovascular risk factors (see Perrin et al16 and Cortese et al12 for screening questions and conditions).
Modest reductions in height (1 or 2 cm after 3 years of psychostimulant treatment) appear to be dose-dependent, and are similar across the methylphenidate and amphetamine classes. Some studies have shown reversal of growth deficits after treatment is stopped treatment and no adverse effects on final adult height.12,17 More study is needed to clarify the effects of continuous psychostimulant treatment from childhood to adulthood on growth.
Studies have failed to show an increased risk of substance abuse in persons with ADHD who were treated with psychostimulants during childhood. Some studies document a lower rate of later substance abuse in youths who received ADHD medications, although other reports show no effect of psychostimulant treatment on subsequent substance use disorder risk.12 Be aware that psychostimulants can be misused (eg, to get “high,” for performance enhancement, to suppress appetite, etc.). Misuse of psychostimulants is most common with short-acting preparations, and generally more difficult with long-acting preparations because extracting the active ingredients for snorting is difficult.2,12 Monitor refill requests and patient behavior for signs of misuse, and be alert for signs of illegal drug use in the patient’s family.
Psychotic symptoms—including hallucinations, delusions, mania, and extreme agitation—with psychostimulant treatment are rare, occurring at a rate of 1.5%.12
Atomoxetine
Approved by the FDA in 2002 for ADHD, atomoxetine is effective and generally well tolerated, although it is not as effective as psychostimulants.2 Atomoxetine is a potent norepinephrine reuptake inhibitor18 that does not produce euphoria, does not have potential for abuse, and has not been linked to increased tic onset or severity.19 Atomoxetine treatment is associated with a lower rate of sleep initiation difficulty compared with psychostimulants.18 Some studies suggest that atomoxetine may have mild beneficial effects on anxiety disorders,18 making it a reasonable choice for patients with significant anxiety or insomnia during psychostimulant treatment. Table 12,7-9 and Table 32,7,9 include information on dosing and duration of action for atomoxetine.
Common side effects of atomoxetine include sedation and fatigue, upset stomach, nausea and vomiting, reduced appetite, headache, and irritability.18 Inform patients that atomoxetine carries an FDA black-box warning for suicide risk; a review of 14 studies showed suicidal ideation was more common with atomoxetine than placebo, although no suicides occurred in any trials.20
Hepatotoxicity is rare with atomoxetine.21 Although routine liver enzyme testing is not required, discontinue atomoxetine if jaundice develops or elevated levels of liver enzymes are noted. Other rare but potentially serious side effects include changes in heart rate (≥20 beats per min) or blood pressure that occur in 5% to 10% of patients taking atomoxetine.22 The risk of serious cardiovascular events and sudden cardiac death with atomoxetine is extremely low, but patients should be screened for a personal and family history of cardiovascular risk factors and, if any of these are present, evaluated further before starting atomoxetine. Routine heart rate and blood pressure monitoring is recommended for all patients.12-14,16
Last, atomoxetine has been linked to growth delays in the first 1 or 2 years of treatment, with a return to expected measurements after an average 2 or 3 years of treatment; persistent decreases in growth rate were observed in patients who were taller or heavier than average before treatment.23
α2 Adrenergic agonists
Guanfacine ER and clonidine ER, the extended release (ER) formulations of α2 adrenergic agonists, were FDA-approved for treating ADHD in 2009 and 2010, respectively. Short-acting guanfacine and clonidine also are used for treating ADHD.24 Their mechanism of action involves stimulation of the pre-synaptic and post-synapic α2 adrenergic receptors, which control the release of norepinephrine and the rate of cell firing.25 The α2 agonists are considered a second-line treatment for ADHD because their efficacy and response rate for core ADHD symptoms lags behind those of psychostimulants.25 In addition to treating core ADHD symptoms, guanfacine and clonidine are used to treat tics and oppositional/aggressive behavior comorbid with ADHD.24,26 Clonidine, which is more sedating than guanfacine, can be used to treat comorbid ADHD and sleep disorders.24 The α2 agonists do not produce euphoria and do not have drug abuse potential.2Table 12,7-9 and Table 32,7,9 provide guidelines for prescribing guanfacine ER and clonidine ER.
The most common adverse effect is drowsiness; other common side effects include dizziness, irritability, headache, and abdominal pain.24 Short-term studies of α2 agonist treatment of ADHD have shown small, non-clinically significant reductions in heart rate and blood pressure; α2 agonist-associated bradycardia, increased QT interval, and cardiac arrhythmias have been reported,7,24,27 as well as rebound hypertension with abrupt discontinuation.24 Screen patients for a personal and family history of cardiovascular risk factors and, if present, evaluate further before initiating α2 agonists.
Combining ADHD medication classes
Combination therapy with >1 ADHD medications is employed when 1 class does not provide adequate symptom coverage or produces problematic side effects.8,24 Psychostimulants can be combined with low-dose atomoxetine (0.5 to 1.0 mg/kg/d) when atomoxetine does not adequately cover ADHD symptoms in school, or when psychostimulants do not adequately cover evening symptoms or patients experience problems with evening psychostimulant rebound.8 To date, prospective data on the safety and efficacy of combining atomoxetine and psychostimulants are limited, but what evidence is available suggests improved symptom control for some, but not all, patients, and a lack of serious adverse events.28
Psychostimulants have been combined with α2agonists when children have an inadequate response to psychostimulants alone, or in cases of ADHD comorbid with aggression or tics.24 Although early case reports raised concern about the safety of combining psychostimulants and α2 agonists, subsequent studies suggest that clonidine and guanfacine generally are well-tolerated when co-administered with psychostimulants.24,27,29
Case continued
Molly has derived substantial benefit from long-acting methylphenidate during the school day, but continues to have significant ADHD-related impairment in the mornings and evenings. Her physician tried afternoon dosing of immediate-release methylphenidate to address evening difficulties, but Molly experienced insomnia. It would be reasonable to consider adjunctive therapy with a non-stimulant medication. A medication that can provide round-the-clock ADHD symptom coverage—such as atomoxetine, guanfacine ER, or clonidine ER—could be added to her current day-time psychostimulant treatment, potentially improving her functioning at home before school and in the evenings.
Additional considerations
Combining medication and behavior therapy offers greater improvements on academic, conduct, and family satisfaction measures than either treatment alone.2 Clinicians can choose to employ behavior therapy alone, particularly if parents feel uncomfortable with—or children have not tolerated—medication.2,3 Evidence-based behavioral parent training and classroom management strategies (implemented by teachers) have shown the strongest and most consistent effects among nonpharmacotherapeutic interventions for ADHD.2 Most studies comparing behavior therapy to psychostimulants have found a stronger effect on core ADHD symptoms from psychostimulants than from behavior therapy.
When a patient does not respond adequately to FDA-approved ADHD medications alone or in combination, consider bupropion, an antidepressant with indirect dopamine and noradrenergic effects. Off-label bupropion has been shown to be effective for ADHD in controlled trials of both children and adults.30
Clinicians often encounter children who meet criteria for ADHD and an anxiety or mood disorder. Table 48,31 summarizes treatment recommendations for these patients.
Clinical considerations
- Begin treatment with a psychostimulant at a low dosage, and titrate gradually until symptoms are controlled or side effects develop.
- Keep in mind that an effective dosage of a psychostimulant is not closely correlated with age, weight, or severity of symptoms.
- Monitor refill requests and patient behavior for signs of psychostimulant misuse. Be alert for signs of illegal drug use in patient family members.
- Lisdexamfetamine, dermal methylphenidate, and osmotic release oral system methylphenidate are the formulations least likely to be misused because their delivery systems make it difficult to extract the active ingredient for snorting or intravenous injection.
- Psychostimulants have not been shown to exacerbate tics in most children who have comorbid ADHD and a tic disorder. When a stimulant is associated with an exacerbation of tics, switching treatment to atomoxetine or α2 agonists is reasonable.
- For patients whose use of a stimulant is limited by an adverse effect on sleep, consider atomoxetine and α2 adrenergic agonists as alternative or adjunctive treatments.
- All 3 classes of FDA-approved ADHD medications (psychostimulants, atomoxetine, and adrenergic agonists) have been associated with adverse cardiac events in children who have underlying cardiovascular conditions. Before initiating treatment, screen patients for a personal or family history of cardiovascular risk factors, and undertake further evaluation as indicated.
Bottom Line
In general, the evidence supports psychostimulants as initial pharmacotherapy for ADHD, with additional options including atomoxetine and α2 agonists. When one medication class does not provide adequate coverage for ADHD symptoms, combining medication classes can be beneficial.
Related Resources
- National Institute of Mental Health. What is attention deficit hyperactivity disorder (ADHD, ADD)?” www.nimh.nih.gov/health/topics/attention-deficit-hyperactivity-disorder-adhd/index.shtml.
- National Resource Center on AD/HD. Managing medication for children and adolescents with ADHD. www.help4adhd.org/en/treatment/medication/WWK3.
Drug Brand Names
Atomoxetine • Strattera
Lisdexamfetamine • Vyvanse
Bupropion • Wellbutrin, Zyban
Clonidine extended release • Kapvay
Guanfacine extended release • Intuniv
Dexmethylphenidate • Focalin, Focalin XR
Mixed amphetamine salts • Adderall, Adderall XR
Dextroamphetamine • Dexedrine, Dexedrine SR, DextroStat, ProCentra
Methylphenidate • Ritalin, Methylin, Metadate CD, Metadate ER, Methylin ER, Ritalin LA, Ritalin SR, Concerta, Quillivant XR, Daytrana
Disclosures
Dr. Froehlich receives support from the National Institute of Mental Health Grant K23 MH083881. Dr. Delgado has received research support from Pfizer, Inc. Dr. Anixt reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Solanto MV. Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res. 1998; 94(1):127-152.
2. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management; Wolraich M, Brown L, Brown RT, et al. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
3. Pliszka S; AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
4. Zametkin AJ, Ernst M. Problems in the management of attention-deficit-hyperactivity disorder. N Engl J Med. 1999;340(1):40-46.
5. Goldman LS, Genel M, Bezman RJ, et al. Diagnosis and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Council on Scientific Affairs, American Medical Association. JAMA. 1998;279(14):1100-1107.
6. Swanson J, Gupta S, Lam A, et al. Development of a new once-a-day formulation of methylphenidate for the treatment of attention-deficit/hyperactivity disorder: proof-of-concept and proof-of-product studies. Arch Gen Psychiatry. 2003;60(2):204-211.
7. Vaughan B, Kratochvil CJ. Pharmacotherapy of pediatric attention-deficit/hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2012;21(4):941-955.
8. Pliszka SR, Crismon ML, Hughes CW, et al; Texas Consensus Conference Panel on Pharmacotherapy of Childhood Attention Deficit Hyperactivity Disorder. The Texas Children’s Medication Algorithm Project: revision of the algorithm for pharmacotherapy of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2006;45(6):642-657.
9. Antshel KM, Hargrave TM, Simonescu M, et al. Advances in understanding and treating ADHD. BMC Med. 2011;9:72.
10. Efron D, Jarman F, Barker M. Side effects of methylphenidate and dexamphetamine in children with attention deficit hyperactivity disorder: a double-blind, crossover trial. Pediatrics. 1997;100(4):662-666.
11. Pringsheim T, Steeves T. Pharmacological treatment for attention deficit hyperactivity disorder (ADHD) in children with comorbid tic disorders. Cochrane Database Syst Rev. 2011(4):CD007990.
12. Cortese S, Holtmann M, Banaschewski T, et al. Practitioner review: current best practice in the management of adverse events during treatment with ADHD medications in children and adolescents. J Child Psychol Psychiatry. 2013; 54(3):227-246.
13. Cooper WO, Habel LA, Sox CM, et al. ADHD drugs and serious cardiovascular events in children and young adults. N Engl J Med. 2011;365(20):1896-1904.
14. Martinez-Raga J, Knecht C, Szerman N, et al. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs. 2013;27(1):15-30.
15. Vetter VL, Elia J, Erickson C, et al; American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee; American Heart Association Council on Cardiovascular Nursing. Cardiovascular monitoring of children and adolescents with heart disease receiving medications for attention deficit/hyperactivity disorder [corrected]: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee and the Council on Cardiovascular Nursing. Circulation. 2008;117(18):2407-2423.
16. Perrin JM, Friedman RA, Knilans TK; Black Box Working Group; Section on Cardiology and Cardiac Surgery. Cardiovascular monitoring and stimulant drugs for attention-deficit/hyperactivity disorder. Pediatrics. 2008;122(2):451-453.
17. Faraone SV, Biederman J, Morley CP, et al. Effect of stimulants on height and weight: a review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(9):994-1009.
18. Garnock-Jones KP, Keating GM. Atomoxetine: a review of its use in attention-deficit hyperactivity disorder in children and adolescents. Paediatr Drugs. 2009;11(3):203-226.
19. Bymaster FP, Katner JS, Nelson DL, et al. Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology. 2002;27(5):699-711.
20. Bangs ME, Tauscher-Wisniewski S, Polzer J, et al. Meta-analysis of suicide-related behavior events in patients treated with atomoxetine. J Am Acad Child Adolesc Psychiatry. 2008;47(2):209-218.
21. Bangs ME, Jin L, Zhang S, et al. Hepatic events associated with atomoxetine treatment for attention-deficit hyperactivity disorder. Drug Saf. 2008;31(4):345-354.
22. U.S. Food and Drug Administration. Strattera (atomoxetine hydrochloride) capsule. http://www.fda.gov/Safety/MedWatch/SafetyInformation/ucm223889.htm. Published August 2013. Accessed October 31, 2013.
23. Spencer TJ, Kratochvil CJ, Sangal RB, et al. Effects of atomoxetine on growth in children with attention-deficit/hyperactivity disorder following up to five years of treatment. J Child Adolesc Psychopharmacol. 2007;17(5):689-700.
24. Connor DF. Other medications. In: Barkley RA, ed. Attention-deficit/hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:658-677.
25. May DE, Kratochvil CJ. Attention-deficit hyperactivity disorder: recent advances in paediatric pharmacotherapy. Drugs. 2010;70(1):15-40.
26. Connor DF, Findling RL, Kollins SH, et al. Effects of guanfacine extended release on oppositional symptoms in children aged 6-12 years with attention-deficit hyperactivity disorder and oppositional symptoms: a randomized, double-blind, placebo-controlled trial. CNS Drugs. 2010; 24(9):755-768.
27. Croxtall JD. Clonidine extended-release: in attention-deficit hyperactivity disorder. Paediatr Drugs. 2011;13(5):329-336.
28. Treuer T, Gau SS, Mendez L, et al. A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability. J Child Adolesc Psychopharmacol. 2013;23(3):179-193.
29. Sallee FR. The role of alpha2-adrenergic agonists in attention-deficit/hyperactivity disorder. Postgrad Med. 2010;122(5):78-87.
30. Spencer TJ. Antidepressant and specific norepinephrine reuptake inhibitor treatments. In: Barkley RA, ed. Attention-deficit hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:648-657.
31. Singh MK, DelBello MP, Kowatch RA, et al. Co-occurrence of bipolar and attention-deficit hyperactivity disorders in children. Bipolar Disord. 2006;8(6):710-720.
1. Solanto MV. Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res. 1998; 94(1):127-152.
2. Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management; Wolraich M, Brown L, Brown RT, et al. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011;128(5):1007-1022.
3. Pliszka S; AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(7):894-921.
4. Zametkin AJ, Ernst M. Problems in the management of attention-deficit-hyperactivity disorder. N Engl J Med. 1999;340(1):40-46.
5. Goldman LS, Genel M, Bezman RJ, et al. Diagnosis and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Council on Scientific Affairs, American Medical Association. JAMA. 1998;279(14):1100-1107.
6. Swanson J, Gupta S, Lam A, et al. Development of a new once-a-day formulation of methylphenidate for the treatment of attention-deficit/hyperactivity disorder: proof-of-concept and proof-of-product studies. Arch Gen Psychiatry. 2003;60(2):204-211.
7. Vaughan B, Kratochvil CJ. Pharmacotherapy of pediatric attention-deficit/hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2012;21(4):941-955.
8. Pliszka SR, Crismon ML, Hughes CW, et al; Texas Consensus Conference Panel on Pharmacotherapy of Childhood Attention Deficit Hyperactivity Disorder. The Texas Children’s Medication Algorithm Project: revision of the algorithm for pharmacotherapy of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2006;45(6):642-657.
9. Antshel KM, Hargrave TM, Simonescu M, et al. Advances in understanding and treating ADHD. BMC Med. 2011;9:72.
10. Efron D, Jarman F, Barker M. Side effects of methylphenidate and dexamphetamine in children with attention deficit hyperactivity disorder: a double-blind, crossover trial. Pediatrics. 1997;100(4):662-666.
11. Pringsheim T, Steeves T. Pharmacological treatment for attention deficit hyperactivity disorder (ADHD) in children with comorbid tic disorders. Cochrane Database Syst Rev. 2011(4):CD007990.
12. Cortese S, Holtmann M, Banaschewski T, et al. Practitioner review: current best practice in the management of adverse events during treatment with ADHD medications in children and adolescents. J Child Psychol Psychiatry. 2013; 54(3):227-246.
13. Cooper WO, Habel LA, Sox CM, et al. ADHD drugs and serious cardiovascular events in children and young adults. N Engl J Med. 2011;365(20):1896-1904.
14. Martinez-Raga J, Knecht C, Szerman N, et al. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs. 2013;27(1):15-30.
15. Vetter VL, Elia J, Erickson C, et al; American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee; American Heart Association Council on Cardiovascular Nursing. Cardiovascular monitoring of children and adolescents with heart disease receiving medications for attention deficit/hyperactivity disorder [corrected]: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young Congenital Cardiac Defects Committee and the Council on Cardiovascular Nursing. Circulation. 2008;117(18):2407-2423.
16. Perrin JM, Friedman RA, Knilans TK; Black Box Working Group; Section on Cardiology and Cardiac Surgery. Cardiovascular monitoring and stimulant drugs for attention-deficit/hyperactivity disorder. Pediatrics. 2008;122(2):451-453.
17. Faraone SV, Biederman J, Morley CP, et al. Effect of stimulants on height and weight: a review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(9):994-1009.
18. Garnock-Jones KP, Keating GM. Atomoxetine: a review of its use in attention-deficit hyperactivity disorder in children and adolescents. Paediatr Drugs. 2009;11(3):203-226.
19. Bymaster FP, Katner JS, Nelson DL, et al. Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology. 2002;27(5):699-711.
20. Bangs ME, Tauscher-Wisniewski S, Polzer J, et al. Meta-analysis of suicide-related behavior events in patients treated with atomoxetine. J Am Acad Child Adolesc Psychiatry. 2008;47(2):209-218.
21. Bangs ME, Jin L, Zhang S, et al. Hepatic events associated with atomoxetine treatment for attention-deficit hyperactivity disorder. Drug Saf. 2008;31(4):345-354.
22. U.S. Food and Drug Administration. Strattera (atomoxetine hydrochloride) capsule. http://www.fda.gov/Safety/MedWatch/SafetyInformation/ucm223889.htm. Published August 2013. Accessed October 31, 2013.
23. Spencer TJ, Kratochvil CJ, Sangal RB, et al. Effects of atomoxetine on growth in children with attention-deficit/hyperactivity disorder following up to five years of treatment. J Child Adolesc Psychopharmacol. 2007;17(5):689-700.
24. Connor DF. Other medications. In: Barkley RA, ed. Attention-deficit/hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:658-677.
25. May DE, Kratochvil CJ. Attention-deficit hyperactivity disorder: recent advances in paediatric pharmacotherapy. Drugs. 2010;70(1):15-40.
26. Connor DF, Findling RL, Kollins SH, et al. Effects of guanfacine extended release on oppositional symptoms in children aged 6-12 years with attention-deficit hyperactivity disorder and oppositional symptoms: a randomized, double-blind, placebo-controlled trial. CNS Drugs. 2010; 24(9):755-768.
27. Croxtall JD. Clonidine extended-release: in attention-deficit hyperactivity disorder. Paediatr Drugs. 2011;13(5):329-336.
28. Treuer T, Gau SS, Mendez L, et al. A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability. J Child Adolesc Psychopharmacol. 2013;23(3):179-193.
29. Sallee FR. The role of alpha2-adrenergic agonists in attention-deficit/hyperactivity disorder. Postgrad Med. 2010;122(5):78-87.
30. Spencer TJ. Antidepressant and specific norepinephrine reuptake inhibitor treatments. In: Barkley RA, ed. Attention-deficit hyperactivity disorder: a handbook for diagnosis and treatment. 3rd ed. New York, NY: The Guilford Press; 2006:648-657.
31. Singh MK, DelBello MP, Kowatch RA, et al. Co-occurrence of bipolar and attention-deficit hyperactivity disorders in children. Bipolar Disord. 2006;8(6):710-720.
Dementia, bizarre creatures, and a white knight to the rescue
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
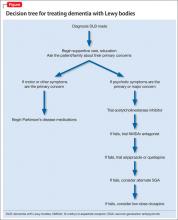
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
CASE Strange creatures
Ms. L, age 78, is admitted to the inpatient unit for treatment of psychosis and behavioral changes. In the months before this admission, she had visited the emergency room several times for recurrent falls. CT scans of the head show no acute changes; brain and spinal MRI reveal evidence of chronic white matter disease and degenerative changes of the spine. Medical workup is unremarkable and includes evaluation for syncope and ambulation impairments related to degenerative disease of the hip joints.
Ms. L and her family are instructed to follow-up with her primary care physician and a neurologist for neuromuscular workup.
She next presents to her primary care physician, describing hallucinations of strangers walking around her house. Over a few weeks, hallucinations expand to include a fixed hallucination of creatures that she describes as having qualities of insects and plants, “piling up” around her. She describes tactile hallucinations of these creatures crawling on her skin, and she tracks their movements around her. She complains of vivid visual hallucinations of these creatures spinning webs across the room and she says she keeps the lights on at night. Ms. L becomes anxious and depressed, and her insomnia becomes worse.
She is referred for outpatient psychiatric evaluation and treatment.
Ms. L’s family notes lapses of short-term memory, disorganization, and difficulty with tasks such as cooking because she has trouble following steps. These deficits come and go, with periods when she is functional and others during which she experiences considerable confusion. The family is uncertain when these signs and symptoms first appeared, but are clear that these deficits are having an impact on her day-to-day life. She can conduct activities of daily living, but with increasing difficulty—and only with help from her husband for tasks that require complex order and movement.
Over several months, Ms. L’s gait stability decreases and she begins to rely on a walker to keep from falling. On the Montreal Cognitive Assessment screening for cognitive dysfunction, she scores 19 out of 30 (normal range >25). This suggests cognitive impairment greater than expected for her age, compared with normal controls, and, when coupled with her functional impairment, raises the possibility of a diagnosis of dementia with Lewy bodies (DLB).
a) donepezil
b) memantine
c) quetiapine
d) low-dose clozapine
The authors’ observations
Limited literature exists of placebo-controlled, large-scale studies on DLB treatment. Cholinesterase inhibitors have shown some symptomatic benefit, including for hallucinations.1-3 Memantine, an N-methyl-d-aspartate receptor blocker, shows mixed results.4 Many studies explore the use of neuroleptics for treating hallucinations in psychosis in Parkinson’s disease and Parkinson’s disease dementia (PDD) but, in DLB, the literature primarily consists of case reports.2 Much of DLB treatment is inferred and intermixed with studies on PDD.5,6
Low-dose clozapine has become a standard treatment for psychosis in Parkinson’s disease based on the findings of several trials.6 Despite its side-effect profile, clozapine has been shown to ameliorate hallucinations in PDD without exacerbating parkinsonian symptoms,7,8 and is the only medication with proven efficacy in PDD.2 The French Clozapine Parkinson Study Group demonstrated relief of psychotic symptoms of Parkinson’s disease with clozapine, 6.25 mg/d.9 The Clozapine Study Group found complete resolution of hallucinations in some patients within 1 day of initiating clozapine. Among patients in this study who did not see immediate benefit, most showed significant improvement of psychotic symptoms in 1 or 2 weeks.10
TREATMENT Few options
Ms. L’s psychiatrist and primary care physician start her on a series of medications. Donepezil is initiated for suspected dementia. We begin a trial of quetiapine to address the hallucinations, but the drug makes her movement symptoms worse. Risperidone also is tried but, again, the drugs make movement symptoms, particularly gait instability, tremor, and rigidity worse without alleviating the hallucinations. Neuroleptics seem to exacerbate confusion. Because of worsening depressive symptoms and our concern over possible pseudodementia, we try several selective serotonin reuptake inhibitors (SSRIs) and mirtazapine. Antidepressants have little effect on her depressive symptoms and do not improve hallucinations or insomnia.
Ms. L’s signs and symptoms become worse over the next few months, with more severe hallucinations, agitation, insomnia, and gait instability. Her agitation over the hallucinations increases and she begins pouring bleach around herself in bed and spraying her house with toxic bug spray. Ms. L’s family brings her to the hospital after they observe her scratching the hallucinatory creatures off of her skin with a razor blade and trying to pry them out of her mouth with a piece of metal.
In the hospital, medical and neurologic workups rule out organic causes for her symptoms and signs. MRI is consistent with imaging from 6 months earlier. Focal neurologic signs are absent. Blood work is within normal limits, failing to reveal any pathology that would suggest a cause for her symptoms and signs, such as syphilis, vitamin deficiency, and Lyme disease.
Ms. L’s symptoms were consistent with consensus guideline criteria for a clinical diagnosis of DLB (Table 1).11-18
She is started on low-dose quetiapine, which she tolerates poorly with worsening confusion, rigidity, tremor, and gait instability. Because other agents failed, Ms. L’s providers and family decide on a trial of clozapine.
Within 24 hours after the first dose of clozapine, 25 mg, sleep improves, the tactile component of hallucinations diminish, and she begins to spend increasing periods of time “observing the creatures” rather than fighting with them.
Over the next few days, Ms. L’s attitude towards the creatures changes. Now, as she sits observing them intently, the hallucinations evolve: rather than tormenting her and causing distress, the plant-creatures burst apart and a miniature knight on horseback charges out. The rest of the creatures then gather into a rank and file and the knight leads them to the nearest exit.
Clozapine is titrated to 50 mg/d, which she tolerates well without exacerbation of cognitive symptoms or movement disorder. The only notable adverse effect at the time of her discharge is sialorrhea.
with an antipsychotic?
a) start low and go slow
b) monitor her heart rate and blood pressure
c) readminister the Montreal Cognitive Assessment
d) all of the above
The authors’ observations
Ideally, in psychosis, antipsychotics eliminate positive symptoms such as hallucinations and delusions. In DLB, the aim is to alleviate the agitation and suffering brought on by the psychotic symptoms without exacerbating other motor and cognitive symptoms. The hallucinations are obstinate, and it is a well-known quality of this disorder that patients are exceptionally susceptible to a range of antipsychotic side effects including cognitive impairment, fatigue, neuroleptic malignant syndrome, and parkinsonism.19
Treatment in DLB requires trial and error, and medications with fewer associated risks should be administered first. Patients with DLB treated with neuroleptics have an increased risk of death compared with those who are not treated.19 Moreover, prescribing information for clozapine includes a black-box warning that the drug:
- is not approved for dementia-related psychosis and
- is associated with an increased risk of death in elderly patients with these conditions, similar to what is seen with other neuroleptics.20
Despite these well-known concerns, it remains difficult for clinicians not to try to treat the distress caused by these symptoms.
We chose clozapine for Ms. L because:
- other neuroleptics failed
- acetylcholinesterase inhibitors did not alleviate Ms. L’s psychosis and associated behavioral disturbance
- there is substantial evidence that the drug can be effective in Parkinson’s disease with psychosis.
There is controversy regarding use of clozapine in DLB. In one case series, clozapine trigger extreme neuroleptic reactions in some patients, similar to what occurs with other second-generation antipsychotics.21 Another case series provides examples of the drug’s efficacy in treating hallucinations and delusions with minimal adverse effects.22
It is important to emphasize that Ms. L’s hallucinations did not go away; rather, they changed to a more benign presentation that she could manage and, occasionally, found pleasant. Ultimately, her agitation—the primary target of treatment—improved markedly with the arrival of the knight in shining armor.
Treatment recommendations
If neuropsychiatric symptoms in DLB are the primary concern of the patient and family, we recommend the following:
- Begin treatment with a cholinesterase inhibitor. The best evidence exists for rivastigmine and donepezil. These drugs have a low risk of side effects, which are primarily gastrointestinal effects with some reports of worsening extrapyramidal symptoms.23-25
- If the patient obtains minimal benefit or develops a significant adverse effect from cholinesterase inhibitors, consider memantine. Its efficacy is under examination and results are mixed; it can be used in combination with cholinesterase inhibitors.26-28
- If psychotic symptoms are upsetting and refractory to other therapies, consider antipsychotics. Avoid first-generation antipsychotics. The American Psychiatric Association recommends aripiprazole or quetiapine initially, although there is little evidence comparing neuroleptics in DLB.29 Because of its risks, reserve clozapine for refractory cases. An exception might be made for patients sensitive to extrapyramidal effects, in whom clozapine could be considered earlier.
There are no formal neuroleptic dosing guidelines beyond a general urging towards minimalism. Mosimann and McKeith30 recommend clozapine, 12.5 mg/d; olanzapine, 2.5 mg/d; risperidone, 0.25 mg/d; or quetiapine, 12.5 mg/d. Such dosages might be effective while producing only minimal side effects.9,31
SSRIs and other antidepressants have not been shown to improve neuropsychiatric symptoms, and often are poorly tolerated.32
One study found efficacy with electroconvulsive therapy and transcranial magnetic stimulation in treatment-resistant patients.33
In addition to these treatments, nonpharmaceutical interventions should be employed from the earliest stages of diagnosis and treatment (Table 2). See the Figure for an algorithm for treating DLB. These include educational and behavioral interventions, social support, psychological interventions, and environmental therapies and modifications.
OUTCOME New friends
The creatures return from time to time, Ms. L reports, but are no longer upsetting because the white knight (a sort of mental deus ex machina) leads the once-terrifying things away. She describes the hallucination as a kind of zoological observation, refers to the creatures that once horrified her as “her friends,” and chuckles as she observes their natural history. This new, far more benign hallucination becomes a mainstay of her symptoms, and she is discharged to the care of her husband and family.
Soon after her discharge, her hallucinations resolved completely, but returned briefly when Ms. L resumed smoking cigarettes because smoking is known to lower clozapine serum levels.34
Bottom Line
Consider a low dosage of a neuroleptic when a patient suffers significant distress and behavioral disturbance related to psychotic symptoms in dementia with Lewy bodies and those problems are not relieved by other agents. Low-dose clozapine is an option for refractory psychotic symptoms or in patients with severe extrapyramidal sensitivity. Start low, and go slow.
Related Resources
- Bishnoi RJ, Grossberg GT, Manepalli J. Differentiating Alzheimer’s disease from dementia with Lewy bodies. Current Psychiatry. 2012;11(11):22-27.
- McKeith I, Emre M. Management of Parkinson’s disease dementia and dementia with Lewy bodies. In: Emre M, ed. Cognitive impairment and dementia in Parkinson’s disease. Oxford, United Kingdom: Oxford University Press; 2010:245-256.
Drug Brand Names
Aripiprazole • Abilify Mirtazapine • Remeron
Clozapine • Clozaril Olanzapine • Zyprexa
Donepezil • Aricept Quetiapine • Seroquel
Haloperidol • Haldol Risperidone • Risperdal
Memantine • Namenda Rivastigmine • Exelon
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.
1. Wesnes KA, McKeith IG, Ferrara R, et al. Effects of rivastigmine on cognitive function in dementia with Lewy bodies: a randomised placebo-controlled international study using the Cognitive Drug Research computerized assessment system. Dement Geriatr Cogn Disord. 2002; 13(3):183-192.
2. Weintraub D, Hurtig HI. Presentation and management of psychosis in Parkinson’s disease and dementia with Lewy bodies. Am J Psychiatry. 2007;164(10):1491-1498.
3. McKeith IG, Wesnes KA, Perry E, et al. Hallucinations predict attentional improvements with rivastigmine in dementia with Lewy bodies. Dement Geriatr Cogn Disord. 2004;18(1):94-100.
4. Emre M, Tsolaki , Bonuccelli U, et al. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(10):969-977.
5. Aarsland D, Ballard C, Walker Z, et al. Clinical trials of dementia with Lewy bodies and Parkinson’s disease dementia. Curr Neurol Neurosci Rep. 2012;12(5):492-501.
6. Drach LM. Drug treatment of dementia with Lewy bodies and Parkinson’s disease dementia--common features and differences [in German]. Med Monatsschr Pharm. 2011; 34(2):47-52.
7. Frieling H, Hillemacher T, Ziegenbein M, et al. Treating dopamimetic psychosis in Parkinson’s disease: Structured review and meta-analysis. Eur Neuropsychopharmacol. 2007;17(3):165-171.
8. Marti MJ, Tolosa E, de la Cerda A. Dementia in Parkinson’s disease. J Neurol. 2007;254(suppl 5):41-48.
9. French Clozapine Parkinson Study Group. Clozapine in drug-induced psychosis in Parkinson’s disease. Lancet. 1999;353(9169):2041-2042.
10. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15(2):201-211.
11. McKeith IG, Galasko D, Kosaka K, et al. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology. 1996;47(5):1113-1124.
12. McKeith IG, Ballard CG, Perry RH et al. Prospective validation of consensus criteria for the diagnosis of dementia with Lewy bodies. Neurology. 2000;54(5):1050-1058.
13. McKeith IG, Dickson DW, Lowe J, et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872.
14. McKeith IG. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis. 2006;9(suppl 3):417-423.
15. Geser F, Wenning GK, Poewe W, et al. How to diagnose dementia with Lewy bodies: state of the art. Mov Disord. 2005;20(suppl 12):S11-20.
16. Latoo J, Jan F. Dementia with Lewy bodies: clinical review. British Journal of Medical Practioners. 2008;1(1):10-14.
17. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6(3):333-341.
18. Litvan I, Bhatia KP, Burn DJ, et al; Movement Disorders Society Scientific Issues Committee. SIC Task Force Appraisal of clinical diagnostic criteria for parkinsonian disorders. Mov Disord. 2003;18(5):467-486.
19. McKeith I, Fairbairn A, Perry R, et al. Neuroleptic sensitivity in patients with senile dementia of Lewy body type. BMJ. 1992;305(6855):673-678.
20. Clozapine Monitoring Guidelines. 2008. http://www.clozapineregistry.com/resuming_treatment_after_interruption.pdf.ashx. Accessed October 31, 2013.
21. Burke WJ, Pfeiffer RF, McComb RD. Neuroleptic sensitivity to clozapine in dementia with Lewy bodies. J Neuropsychiatry Clin Neurosci. 1998;10(2):227-229.
22. Chacko RC, Hurley RA, Jankovic J. Clozapine use in diffuse Lewy body disease. J Neuropsychiatry Clin Neurosci. 1993;5(2):206-208.
23. McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. 2000;356(9247):2031-2036
24. Mori E, Ikeda M, Kosaka K; Donepezil-DLB Study Investiagors. Donepezil for dementia with Lewy bodies: a randomized, placebo‐controlled trial. Ann Neurol. 2012; 72(1):41-52.
25. Ukai K, Aleksic B, Ishihara R, et al. Efficacy of donepezil for the treatment of visual and multiple sensory hallucinations in dementia with Lewy bodies. Clinical Neuropsychopharmacology and Therapeutics. 2011;2:56-58.
26. Aarsland D, Ballard C, Walker Z, et al. Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol. 2009;8(7):613-618.
27. Boeve BF, Silber MH, Ferman TJ. Melatonin for treatment of REM sleep behavior disorder in neurologic disorders: results in 14 patients. Sleep Med. 2003;4(4):281-284.
28. Mathys ML, McCarrell J, Sleeper RB, et al. Visual hallucinations treated with the reinitiation of memantine in a patient with Lewy body dementia. Ann Pharmacother. 2013;47(2):e10.
29. American Psychiatric Association. Practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. 2nd ed. http://psychiatryonline.org/pdfaccess.ashx?ResourceID=243205&PDFSource=6. Accessed November 1, 2013.
30. Mosimann U, McKeith IG. Dementia with lewy bodies—diagnosis and treatment. Swiss Med Wkly. 2003;133:131-142.
31. Baskys A, Davis P Atypical antipsychotic quetiapine in the treatment of the psychosis associated with Lewy body dementia. Neurobiol Aging. 2002;23:S63.
32. Culo S, Mulsant BH, Rosen J, et al. Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled-trial. Alzheimer Dis Assoc Disord. 2010;24(4):306-364.
33. Takahashi S, Mizukami K, Yasuno F, et al. Depression associated with dementia with Lewy bodies (DLB) and the effect of somatotherapy. Psychogeriatrics. 2009;9(2):56-61.
34. van der Weide J, Steijns LS, van Weelden MJ. The effect of smoking and cytochrome P450 CYP1A2 genetic polymorphism on clozapine clearance and dose requirement. Pharmacogenetics. 2003;13(3):169-172.
Are you admitting malpractice if you apologize to a patient?
Dear Dr. Mossman:
Recently, my prescribing error caused a patient to get very sick. I feel terrible. I want to tell my patient I’m sorry, but I’ve heard that a lawyer could use my “confession” to prove I’ve committed malpractice. If I apologize, could my words come back to haunt me if a lawsuit is filed?
Submitted by “Dr. E”
As several faiths have long recognized, apologies are important social acts that express our awareness of and obligations to each other. In recent years, psychologists have established how apologies confer emotional benefits on those who give and receive them.1 Offering a sincere apology can be the right thing to do and a beneficial act for both the apologizing and the injured parties.
Traditionally, physicians have avoided apologizing for errors that harmed patients. Part of the reluctance stemmed from pride or wanting to avoid shame. But as Dr. E’s question suggests, doctors also have feared—and lawyers have advised—that apologizing might compromise a malpractice defense.2
Attitudes have changed in recent years, however. Increasingly, practitioners, medical organizations, and risk management entities are telling physicians they should apologize for errors, and many states have laws that mitigate adverse legal consequences of saying “I’m sorry.”
In response to Dr. E’s questions, I’ll examine:
• ethical and professional obligations following unexpected outcomes
• physicians’ reasons for being reluctant to apologize
• the benefits of apologizing
• legal protections for apologies.
Owning up: Ethical and professional expectations
Current codes of medical ethics say that physicians should tell patients when mistakes and misjudgments have caused harm. “It is a fundamental ethical requirement that a physician should at all times deal honestly and openly with patients,” states the American Medical Association’s Code of Ethics. When a patient suffers because of a medical error, “the physician is ethically required to inform the patient of all the facts necessary to ensure” the patient can “make informed decisions regarding future medical care.”3
The National Patient Safety Foundation,4 the American College of Physicians,5 and the Joint Commission (the agency that provides official accreditation of thousands of healthcare organizations) have voiced similar positions for years. Since 2001, the Joint Commission has required that practitioners and medical facilities tell patients and families “about the outcomes of care, treatment, and services ... including unanticipated outcomes.”6
Reluctance is understandable
Although these recommendations and policies suggest that telling patients about medical errors is an established professional expectation, physicians remain reluctant to apologize to patients for emotional and legal reasons that are easy to understand.
Apologizing is hard. On one hand, research shows that refusing to apologize sometimes increases feelings of empowerment and control, and can boost self-esteem more than apologizing does.7 On the other, apologizing often requires one to acknowledge a failure or betrayal of trust and to experience guilt, shame, embarrassment, or fear that one’s apology will be met with anger or rejection.8
Physicians historically have treated errors as personal failures. Apologizing in a medical context can feel like saying, “I am incompetent.”9,10 The law has reinforced this attitude. As the Mississippi Supreme Court put it, “Medical malpractice is legal fault by a physician or surgeon. It arises from the failure of a physician to provide the quality of care required by law” (emphasis added).11
Some lawyers continue to advise physicians not to make admissions that could be used in a malpractice case. Their reasoning: If a doctor does something that adversely affects a malpractice insurer’s ability to defend the case, the insurer might not provide liability coverage for the adverse event.12
Emotional and legal benefits
Against this no-apology stance is a growing body of theoretical, empirical, and practical arguments favoring apologies for medical errors. Case studies suggest that anger is behind much medical malpractice litigation and that physicians’ apologies—which reduce anger and increase communication—might reduce patients’ motivations to sue.13 Apologies sometimes lead to forgiveness, an emotional state that “can provide victims and offenders with many important benefits, including enhanced psychological well-being ... and greater physiological health.”14 Apologies do this by mitigating the injured party’s negative emotional states and diminishing rumination about the transgression and perceived harm severity.
The practical argument favoring apologizing is that it may defuse feelings that lead to lawsuits and reduce the size of payouts. Experimental studies suggest that apologizing leads to earlier satisfaction and closure, faster settlements, and lower damage payments. When apologies include admissions of fault, injured parties feel greater respect for and less need to punish those who have harmed them, are more willing to forgive, and are more likely to accept settlement offers.15
Hospitals in Pennsylvania, Kentucky, and Michigan have found that sincere apologies and effective error disclosure programs reduce malpractice payouts and lead to faster settlements.16 As some plaintiffs’ lawyers point out, being honest and forthright and fixing the injured parties’ problems can quickly defuse a lawsuit. One attorney explained things this way: “We never sue the nice, contrite doctors. Their patients never call our offices. But the doctors who are poor communicators and abandon their patients get sued all the time. Their patients come to our offices looking for answers.”17
Apology laws: Protection from your own words
The belief that apologies by physicians can help patients emotionally and reduce malpractice litigation has led state legislatures to enact so-called apology laws in many jurisdictions in the United States.18 The general point of these rules and statutes is to prevent later use of doctors’ words in litigation. States differ substantially in the scope and type of protection that their laws offer. Some states prohibit doctors’ apologies for adverse outcomes from being used in litigation to prove negligence, while others only exclude expressions of sympathy or offers to pay for corrective treatment. Selected language from several states’ apology laws appears in the Table.19-23
Do apology laws work? Recent research by economists Ho and Liu indicates that they do. Comparing payouts in states with and without apology laws, they conclude that “apology laws have the greatest reduction in average payment size and settlement time in cases involving more severe patient outcomes,”13 such as obstetrics and anesthesia cases, cases that involve infants, and cases in which physicians improperly manage or fail to properly diagnose an illness.24
The practical impact of apologizing for psychiatric malpractice cases is unclear, but forensic psychiatrists Marilyn Price and Patricia Recupero believe that, following some unexpected outcomes, thoughtful expressions of sympathy, regret, and—if the outcome resulted from an error—apologies may be appropriate. Price and Recupero caution that such conversations should occur as part of broader programs that investigate unanticipated adverse events and provide education and coaching about appropriate ways to make disclosures. Clinicians also should consult with legal counsel, risk management officers, and liability insurance carriers before initiating such disclosures.25
Bottom Line
Apologizing for medical errors may mitigate malpractice liability and can help injured parties and physicians feel better. Whether plaintiffs can use apologies as evidence of malpractice depends on state laws and rules of evidence. Before you apologize for an unanticipated outcome, discuss the situation with your legal counsel, risk management officers, and insurers.
Disclosure
Dr. Mossman reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Featured Audio
Douglas Mossman, MD, talks about who you should consult before apologizing to a patient for a bad outcome. Dr. Mossman is Professor of Clinical Psychiatry and Director, Division of Forensic Psychiatry, University of Cincinnati College of Medicine, Cincinnati, Ohio.
1. McCullough ME, Sandage SJ, Brown SW, et al. Interpersonal forgiving in close relationships: II. Theoretical elaboration and measurement. J Pers Soc Psychol. 1998;75:1586-1603.
2. O’Reilly KB. “I’m sorry”: why is that so hard for doctors to say? http://www.amednews.com/article/20100201/profession/302019937/4. Published February 1, 2010. Accessed September 30, 2013.
3. American Medical Association. AMA Code of Medical Ethics, Opinion 8.12 – Patient information. http://www.ama-assn.org//ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion812.page. Published June 1994. Accessed September 30, 2013.
4. Hickson GB, Pichert JW. Disclosure and apology. http://www.npsf.org/wp-content/uploads/2011/10/RG_SUPS_After_Mod1_Hickson.pdf. Accessed October 4, 2013.
5. Snyder L, American College of Physicians Ethics, Professionalism, and Human Rights Committee. American College of Physicians ethics manual: sixth edition. Ann Intern Med. 2012;156(1, pt 2):73-104.
6. ECRI Institute. Disclosure of unanticipated outcomes. In: Healthcare risk control Supplement A, Risk analysis. Plymouth Meeting, PA: ECRI; 2002.
7. Okimoto TG, Wenzel M, Hedrick K. Refusing to apologize can have psychological benefits (and we issue no mea culpa for this research finding). Eur J Soc Psychol. 2013;43:22-31.
8. Lazare A. On apology. New York, NY: Oxford University Press; 2004.
9. Hilfiker D. Facing our mistakes. N Engl J Med. 1984;310:
118-122.
10. Leape LL. Error in medicine. JAMA. 1994;272:1851-1857.
11. Hall v. Hilbun, 466 So.2d 856 (Miss. 1985).
12. Kern SI. You continue to face exposure if you apologize. http://medicaleconomics.modernmedicine.com/medical-economics/news/modernmedicine/modern-medicine-now/you-continue-face-exposure-if-you-apologiz. Published September 24, 2010. Accessed October 1, 2013.
13. Ho B, Liu E. Does sorry work? The impact of apology laws on medical malpractice. J Risk Uncertain. 2011;43(2):141-167.
14. Fehr R, Gelfand MJ, Nag M. The road to forgiveness: a meta-analytic synthesis of its situational and dispositional correlates. Psychol Bull. 2010;136:894-914.
15. Robbennolt JK. Apologies and settlement. Court Review. 2009;45:90-97.
16. Saitta N, Hodge SD. Efficacy of a physician’s words of empathy: an overview of state apology laws. J Am Osteopath Assoc. 2012;112(5):302-306.
17. Wojcieszak D, Banja J, Houk C. The sorry works! coalition: making the case for full disclosure. Jt Comm J Qual Patient Saf. 2006;32(6):344-350.
18. National Conference of State Legislatures. Medical liability/Medical malpractice laws. http://www.ncsl.org/issues-research/banking/medical-liability-medical-malpractice-laws.aspx. Published August 15, 2011. Accessed October 4, 2013.
19. Conn Gen Stat Ann §52-184d(b).
20. Fla Stat §90.4026(2).
21. Ill Comp Stat §5/8-1901.
22. NC Gen Stat §8C-1, Rule 413.
23. Tex. Civ. Prac. & Rem. Code §18.061.
24. Ho B, Liu E. What’s an apology worth? Decomposing the effect of apologies on medical malpractice payments using state apology laws. J Empir Leg Stud. 2011;8:179-199.
25. Price M, Recupero PR. Risk management. In: Sharfstein SS, Dickerson FB, Oldham JM, eds. Textbook of hospital psychiatry. Arlington, VA: American Psychiatric Publishing, Inc.; 2009:411-412.
Dear Dr. Mossman:
Recently, my prescribing error caused a patient to get very sick. I feel terrible. I want to tell my patient I’m sorry, but I’ve heard that a lawyer could use my “confession” to prove I’ve committed malpractice. If I apologize, could my words come back to haunt me if a lawsuit is filed?
Submitted by “Dr. E”
As several faiths have long recognized, apologies are important social acts that express our awareness of and obligations to each other. In recent years, psychologists have established how apologies confer emotional benefits on those who give and receive them.1 Offering a sincere apology can be the right thing to do and a beneficial act for both the apologizing and the injured parties.
Traditionally, physicians have avoided apologizing for errors that harmed patients. Part of the reluctance stemmed from pride or wanting to avoid shame. But as Dr. E’s question suggests, doctors also have feared—and lawyers have advised—that apologizing might compromise a malpractice defense.2
Attitudes have changed in recent years, however. Increasingly, practitioners, medical organizations, and risk management entities are telling physicians they should apologize for errors, and many states have laws that mitigate adverse legal consequences of saying “I’m sorry.”
In response to Dr. E’s questions, I’ll examine:
• ethical and professional obligations following unexpected outcomes
• physicians’ reasons for being reluctant to apologize
• the benefits of apologizing
• legal protections for apologies.
Owning up: Ethical and professional expectations
Current codes of medical ethics say that physicians should tell patients when mistakes and misjudgments have caused harm. “It is a fundamental ethical requirement that a physician should at all times deal honestly and openly with patients,” states the American Medical Association’s Code of Ethics. When a patient suffers because of a medical error, “the physician is ethically required to inform the patient of all the facts necessary to ensure” the patient can “make informed decisions regarding future medical care.”3
The National Patient Safety Foundation,4 the American College of Physicians,5 and the Joint Commission (the agency that provides official accreditation of thousands of healthcare organizations) have voiced similar positions for years. Since 2001, the Joint Commission has required that practitioners and medical facilities tell patients and families “about the outcomes of care, treatment, and services ... including unanticipated outcomes.”6
Reluctance is understandable
Although these recommendations and policies suggest that telling patients about medical errors is an established professional expectation, physicians remain reluctant to apologize to patients for emotional and legal reasons that are easy to understand.
Apologizing is hard. On one hand, research shows that refusing to apologize sometimes increases feelings of empowerment and control, and can boost self-esteem more than apologizing does.7 On the other, apologizing often requires one to acknowledge a failure or betrayal of trust and to experience guilt, shame, embarrassment, or fear that one’s apology will be met with anger or rejection.8
Physicians historically have treated errors as personal failures. Apologizing in a medical context can feel like saying, “I am incompetent.”9,10 The law has reinforced this attitude. As the Mississippi Supreme Court put it, “Medical malpractice is legal fault by a physician or surgeon. It arises from the failure of a physician to provide the quality of care required by law” (emphasis added).11
Some lawyers continue to advise physicians not to make admissions that could be used in a malpractice case. Their reasoning: If a doctor does something that adversely affects a malpractice insurer’s ability to defend the case, the insurer might not provide liability coverage for the adverse event.12
Emotional and legal benefits
Against this no-apology stance is a growing body of theoretical, empirical, and practical arguments favoring apologies for medical errors. Case studies suggest that anger is behind much medical malpractice litigation and that physicians’ apologies—which reduce anger and increase communication—might reduce patients’ motivations to sue.13 Apologies sometimes lead to forgiveness, an emotional state that “can provide victims and offenders with many important benefits, including enhanced psychological well-being ... and greater physiological health.”14 Apologies do this by mitigating the injured party’s negative emotional states and diminishing rumination about the transgression and perceived harm severity.
The practical argument favoring apologizing is that it may defuse feelings that lead to lawsuits and reduce the size of payouts. Experimental studies suggest that apologizing leads to earlier satisfaction and closure, faster settlements, and lower damage payments. When apologies include admissions of fault, injured parties feel greater respect for and less need to punish those who have harmed them, are more willing to forgive, and are more likely to accept settlement offers.15
Hospitals in Pennsylvania, Kentucky, and Michigan have found that sincere apologies and effective error disclosure programs reduce malpractice payouts and lead to faster settlements.16 As some plaintiffs’ lawyers point out, being honest and forthright and fixing the injured parties’ problems can quickly defuse a lawsuit. One attorney explained things this way: “We never sue the nice, contrite doctors. Their patients never call our offices. But the doctors who are poor communicators and abandon their patients get sued all the time. Their patients come to our offices looking for answers.”17
Apology laws: Protection from your own words
The belief that apologies by physicians can help patients emotionally and reduce malpractice litigation has led state legislatures to enact so-called apology laws in many jurisdictions in the United States.18 The general point of these rules and statutes is to prevent later use of doctors’ words in litigation. States differ substantially in the scope and type of protection that their laws offer. Some states prohibit doctors’ apologies for adverse outcomes from being used in litigation to prove negligence, while others only exclude expressions of sympathy or offers to pay for corrective treatment. Selected language from several states’ apology laws appears in the Table.19-23
Do apology laws work? Recent research by economists Ho and Liu indicates that they do. Comparing payouts in states with and without apology laws, they conclude that “apology laws have the greatest reduction in average payment size and settlement time in cases involving more severe patient outcomes,”13 such as obstetrics and anesthesia cases, cases that involve infants, and cases in which physicians improperly manage or fail to properly diagnose an illness.24
The practical impact of apologizing for psychiatric malpractice cases is unclear, but forensic psychiatrists Marilyn Price and Patricia Recupero believe that, following some unexpected outcomes, thoughtful expressions of sympathy, regret, and—if the outcome resulted from an error—apologies may be appropriate. Price and Recupero caution that such conversations should occur as part of broader programs that investigate unanticipated adverse events and provide education and coaching about appropriate ways to make disclosures. Clinicians also should consult with legal counsel, risk management officers, and liability insurance carriers before initiating such disclosures.25
Bottom Line
Apologizing for medical errors may mitigate malpractice liability and can help injured parties and physicians feel better. Whether plaintiffs can use apologies as evidence of malpractice depends on state laws and rules of evidence. Before you apologize for an unanticipated outcome, discuss the situation with your legal counsel, risk management officers, and insurers.
Disclosure
Dr. Mossman reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Featured Audio
Douglas Mossman, MD, talks about who you should consult before apologizing to a patient for a bad outcome. Dr. Mossman is Professor of Clinical Psychiatry and Director, Division of Forensic Psychiatry, University of Cincinnati College of Medicine, Cincinnati, Ohio.
Dear Dr. Mossman:
Recently, my prescribing error caused a patient to get very sick. I feel terrible. I want to tell my patient I’m sorry, but I’ve heard that a lawyer could use my “confession” to prove I’ve committed malpractice. If I apologize, could my words come back to haunt me if a lawsuit is filed?
Submitted by “Dr. E”
As several faiths have long recognized, apologies are important social acts that express our awareness of and obligations to each other. In recent years, psychologists have established how apologies confer emotional benefits on those who give and receive them.1 Offering a sincere apology can be the right thing to do and a beneficial act for both the apologizing and the injured parties.
Traditionally, physicians have avoided apologizing for errors that harmed patients. Part of the reluctance stemmed from pride or wanting to avoid shame. But as Dr. E’s question suggests, doctors also have feared—and lawyers have advised—that apologizing might compromise a malpractice defense.2
Attitudes have changed in recent years, however. Increasingly, practitioners, medical organizations, and risk management entities are telling physicians they should apologize for errors, and many states have laws that mitigate adverse legal consequences of saying “I’m sorry.”
In response to Dr. E’s questions, I’ll examine:
• ethical and professional obligations following unexpected outcomes
• physicians’ reasons for being reluctant to apologize
• the benefits of apologizing
• legal protections for apologies.
Owning up: Ethical and professional expectations
Current codes of medical ethics say that physicians should tell patients when mistakes and misjudgments have caused harm. “It is a fundamental ethical requirement that a physician should at all times deal honestly and openly with patients,” states the American Medical Association’s Code of Ethics. When a patient suffers because of a medical error, “the physician is ethically required to inform the patient of all the facts necessary to ensure” the patient can “make informed decisions regarding future medical care.”3
The National Patient Safety Foundation,4 the American College of Physicians,5 and the Joint Commission (the agency that provides official accreditation of thousands of healthcare organizations) have voiced similar positions for years. Since 2001, the Joint Commission has required that practitioners and medical facilities tell patients and families “about the outcomes of care, treatment, and services ... including unanticipated outcomes.”6
Reluctance is understandable
Although these recommendations and policies suggest that telling patients about medical errors is an established professional expectation, physicians remain reluctant to apologize to patients for emotional and legal reasons that are easy to understand.
Apologizing is hard. On one hand, research shows that refusing to apologize sometimes increases feelings of empowerment and control, and can boost self-esteem more than apologizing does.7 On the other, apologizing often requires one to acknowledge a failure or betrayal of trust and to experience guilt, shame, embarrassment, or fear that one’s apology will be met with anger or rejection.8
Physicians historically have treated errors as personal failures. Apologizing in a medical context can feel like saying, “I am incompetent.”9,10 The law has reinforced this attitude. As the Mississippi Supreme Court put it, “Medical malpractice is legal fault by a physician or surgeon. It arises from the failure of a physician to provide the quality of care required by law” (emphasis added).11
Some lawyers continue to advise physicians not to make admissions that could be used in a malpractice case. Their reasoning: If a doctor does something that adversely affects a malpractice insurer’s ability to defend the case, the insurer might not provide liability coverage for the adverse event.12
Emotional and legal benefits
Against this no-apology stance is a growing body of theoretical, empirical, and practical arguments favoring apologies for medical errors. Case studies suggest that anger is behind much medical malpractice litigation and that physicians’ apologies—which reduce anger and increase communication—might reduce patients’ motivations to sue.13 Apologies sometimes lead to forgiveness, an emotional state that “can provide victims and offenders with many important benefits, including enhanced psychological well-being ... and greater physiological health.”14 Apologies do this by mitigating the injured party’s negative emotional states and diminishing rumination about the transgression and perceived harm severity.
The practical argument favoring apologizing is that it may defuse feelings that lead to lawsuits and reduce the size of payouts. Experimental studies suggest that apologizing leads to earlier satisfaction and closure, faster settlements, and lower damage payments. When apologies include admissions of fault, injured parties feel greater respect for and less need to punish those who have harmed them, are more willing to forgive, and are more likely to accept settlement offers.15
Hospitals in Pennsylvania, Kentucky, and Michigan have found that sincere apologies and effective error disclosure programs reduce malpractice payouts and lead to faster settlements.16 As some plaintiffs’ lawyers point out, being honest and forthright and fixing the injured parties’ problems can quickly defuse a lawsuit. One attorney explained things this way: “We never sue the nice, contrite doctors. Their patients never call our offices. But the doctors who are poor communicators and abandon their patients get sued all the time. Their patients come to our offices looking for answers.”17
Apology laws: Protection from your own words
The belief that apologies by physicians can help patients emotionally and reduce malpractice litigation has led state legislatures to enact so-called apology laws in many jurisdictions in the United States.18 The general point of these rules and statutes is to prevent later use of doctors’ words in litigation. States differ substantially in the scope and type of protection that their laws offer. Some states prohibit doctors’ apologies for adverse outcomes from being used in litigation to prove negligence, while others only exclude expressions of sympathy or offers to pay for corrective treatment. Selected language from several states’ apology laws appears in the Table.19-23
Do apology laws work? Recent research by economists Ho and Liu indicates that they do. Comparing payouts in states with and without apology laws, they conclude that “apology laws have the greatest reduction in average payment size and settlement time in cases involving more severe patient outcomes,”13 such as obstetrics and anesthesia cases, cases that involve infants, and cases in which physicians improperly manage or fail to properly diagnose an illness.24
The practical impact of apologizing for psychiatric malpractice cases is unclear, but forensic psychiatrists Marilyn Price and Patricia Recupero believe that, following some unexpected outcomes, thoughtful expressions of sympathy, regret, and—if the outcome resulted from an error—apologies may be appropriate. Price and Recupero caution that such conversations should occur as part of broader programs that investigate unanticipated adverse events and provide education and coaching about appropriate ways to make disclosures. Clinicians also should consult with legal counsel, risk management officers, and liability insurance carriers before initiating such disclosures.25
Bottom Line
Apologizing for medical errors may mitigate malpractice liability and can help injured parties and physicians feel better. Whether plaintiffs can use apologies as evidence of malpractice depends on state laws and rules of evidence. Before you apologize for an unanticipated outcome, discuss the situation with your legal counsel, risk management officers, and insurers.
Disclosure
Dr. Mossman reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Featured Audio
Douglas Mossman, MD, talks about who you should consult before apologizing to a patient for a bad outcome. Dr. Mossman is Professor of Clinical Psychiatry and Director, Division of Forensic Psychiatry, University of Cincinnati College of Medicine, Cincinnati, Ohio.
1. McCullough ME, Sandage SJ, Brown SW, et al. Interpersonal forgiving in close relationships: II. Theoretical elaboration and measurement. J Pers Soc Psychol. 1998;75:1586-1603.
2. O’Reilly KB. “I’m sorry”: why is that so hard for doctors to say? http://www.amednews.com/article/20100201/profession/302019937/4. Published February 1, 2010. Accessed September 30, 2013.
3. American Medical Association. AMA Code of Medical Ethics, Opinion 8.12 – Patient information. http://www.ama-assn.org//ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion812.page. Published June 1994. Accessed September 30, 2013.
4. Hickson GB, Pichert JW. Disclosure and apology. http://www.npsf.org/wp-content/uploads/2011/10/RG_SUPS_After_Mod1_Hickson.pdf. Accessed October 4, 2013.
5. Snyder L, American College of Physicians Ethics, Professionalism, and Human Rights Committee. American College of Physicians ethics manual: sixth edition. Ann Intern Med. 2012;156(1, pt 2):73-104.
6. ECRI Institute. Disclosure of unanticipated outcomes. In: Healthcare risk control Supplement A, Risk analysis. Plymouth Meeting, PA: ECRI; 2002.
7. Okimoto TG, Wenzel M, Hedrick K. Refusing to apologize can have psychological benefits (and we issue no mea culpa for this research finding). Eur J Soc Psychol. 2013;43:22-31.
8. Lazare A. On apology. New York, NY: Oxford University Press; 2004.
9. Hilfiker D. Facing our mistakes. N Engl J Med. 1984;310:
118-122.
10. Leape LL. Error in medicine. JAMA. 1994;272:1851-1857.
11. Hall v. Hilbun, 466 So.2d 856 (Miss. 1985).
12. Kern SI. You continue to face exposure if you apologize. http://medicaleconomics.modernmedicine.com/medical-economics/news/modernmedicine/modern-medicine-now/you-continue-face-exposure-if-you-apologiz. Published September 24, 2010. Accessed October 1, 2013.
13. Ho B, Liu E. Does sorry work? The impact of apology laws on medical malpractice. J Risk Uncertain. 2011;43(2):141-167.
14. Fehr R, Gelfand MJ, Nag M. The road to forgiveness: a meta-analytic synthesis of its situational and dispositional correlates. Psychol Bull. 2010;136:894-914.
15. Robbennolt JK. Apologies and settlement. Court Review. 2009;45:90-97.
16. Saitta N, Hodge SD. Efficacy of a physician’s words of empathy: an overview of state apology laws. J Am Osteopath Assoc. 2012;112(5):302-306.
17. Wojcieszak D, Banja J, Houk C. The sorry works! coalition: making the case for full disclosure. Jt Comm J Qual Patient Saf. 2006;32(6):344-350.
18. National Conference of State Legislatures. Medical liability/Medical malpractice laws. http://www.ncsl.org/issues-research/banking/medical-liability-medical-malpractice-laws.aspx. Published August 15, 2011. Accessed October 4, 2013.
19. Conn Gen Stat Ann §52-184d(b).
20. Fla Stat §90.4026(2).
21. Ill Comp Stat §5/8-1901.
22. NC Gen Stat §8C-1, Rule 413.
23. Tex. Civ. Prac. & Rem. Code §18.061.
24. Ho B, Liu E. What’s an apology worth? Decomposing the effect of apologies on medical malpractice payments using state apology laws. J Empir Leg Stud. 2011;8:179-199.
25. Price M, Recupero PR. Risk management. In: Sharfstein SS, Dickerson FB, Oldham JM, eds. Textbook of hospital psychiatry. Arlington, VA: American Psychiatric Publishing, Inc.; 2009:411-412.
1. McCullough ME, Sandage SJ, Brown SW, et al. Interpersonal forgiving in close relationships: II. Theoretical elaboration and measurement. J Pers Soc Psychol. 1998;75:1586-1603.
2. O’Reilly KB. “I’m sorry”: why is that so hard for doctors to say? http://www.amednews.com/article/20100201/profession/302019937/4. Published February 1, 2010. Accessed September 30, 2013.
3. American Medical Association. AMA Code of Medical Ethics, Opinion 8.12 – Patient information. http://www.ama-assn.org//ama/pub/physician-resources/medical-ethics/code-medical-ethics/opinion812.page. Published June 1994. Accessed September 30, 2013.
4. Hickson GB, Pichert JW. Disclosure and apology. http://www.npsf.org/wp-content/uploads/2011/10/RG_SUPS_After_Mod1_Hickson.pdf. Accessed October 4, 2013.
5. Snyder L, American College of Physicians Ethics, Professionalism, and Human Rights Committee. American College of Physicians ethics manual: sixth edition. Ann Intern Med. 2012;156(1, pt 2):73-104.
6. ECRI Institute. Disclosure of unanticipated outcomes. In: Healthcare risk control Supplement A, Risk analysis. Plymouth Meeting, PA: ECRI; 2002.
7. Okimoto TG, Wenzel M, Hedrick K. Refusing to apologize can have psychological benefits (and we issue no mea culpa for this research finding). Eur J Soc Psychol. 2013;43:22-31.
8. Lazare A. On apology. New York, NY: Oxford University Press; 2004.
9. Hilfiker D. Facing our mistakes. N Engl J Med. 1984;310:
118-122.
10. Leape LL. Error in medicine. JAMA. 1994;272:1851-1857.
11. Hall v. Hilbun, 466 So.2d 856 (Miss. 1985).
12. Kern SI. You continue to face exposure if you apologize. http://medicaleconomics.modernmedicine.com/medical-economics/news/modernmedicine/modern-medicine-now/you-continue-face-exposure-if-you-apologiz. Published September 24, 2010. Accessed October 1, 2013.
13. Ho B, Liu E. Does sorry work? The impact of apology laws on medical malpractice. J Risk Uncertain. 2011;43(2):141-167.
14. Fehr R, Gelfand MJ, Nag M. The road to forgiveness: a meta-analytic synthesis of its situational and dispositional correlates. Psychol Bull. 2010;136:894-914.
15. Robbennolt JK. Apologies and settlement. Court Review. 2009;45:90-97.
16. Saitta N, Hodge SD. Efficacy of a physician’s words of empathy: an overview of state apology laws. J Am Osteopath Assoc. 2012;112(5):302-306.
17. Wojcieszak D, Banja J, Houk C. The sorry works! coalition: making the case for full disclosure. Jt Comm J Qual Patient Saf. 2006;32(6):344-350.
18. National Conference of State Legislatures. Medical liability/Medical malpractice laws. http://www.ncsl.org/issues-research/banking/medical-liability-medical-malpractice-laws.aspx. Published August 15, 2011. Accessed October 4, 2013.
19. Conn Gen Stat Ann §52-184d(b).
20. Fla Stat §90.4026(2).
21. Ill Comp Stat §5/8-1901.
22. NC Gen Stat §8C-1, Rule 413.
23. Tex. Civ. Prac. & Rem. Code §18.061.
24. Ho B, Liu E. What’s an apology worth? Decomposing the effect of apologies on medical malpractice payments using state apology laws. J Empir Leg Stud. 2011;8:179-199.
25. Price M, Recupero PR. Risk management. In: Sharfstein SS, Dickerson FB, Oldham JM, eds. Textbook of hospital psychiatry. Arlington, VA: American Psychiatric Publishing, Inc.; 2009:411-412.
Report Cites Wide Variation in Prescription Drug Use by Medicare Patients
A recent Dartmouth Atlas Project report that highlights regional differences for prescription drug use among Medicare patients across the U.S. provides insights into best practices related to effective and high-risk prescription drug therapy.
The report [PDF] found geographic disparity in the total use of prescription medications, variations in effective prescription care, dissimilarities in the use of potentially harmful medications, and differences in total spending on prescription drugs.
Lead author Jeffrey Munson, MD, MSCE, says he expected some geographic variation in the use of discretionary medications, but was surprised by the discrepancy in patients' medication usage. For example, in San Angelo, Texas, 91.4% of heart attack survivors in 2008–2009 filled at least one prescription for beta blockers in the year after their discharge, compared with just 62.5% of the same population of patients in Salem, Ore.
"Clearly, there are regions of the country that have figured out how to best handle certain aspects of medication usage," says Dr. Munson, assistant professor at the Dartmouth Institute for Health Policy & Clinical Practice in Lebanon, N.H. "Instead of just looking at regions that are high performing and saying, 'Wow, that seems to be a really high bar they've set, I'm not sure we can achieve it,' I wonder if maybe it's time we look at those regions and say, 'How are you achieving those very high standards, and what about what you do can I do where I live.'"
The Dartmouth Atlas Project report documents geographic variation in healthcare utilization unrelated to outcome and offers an extensive database for comparison by state, county, region, and facility. Dr. Munson says he understands that healthcare reform is pushing hospitalists and other physicians to focus on many new issues, but that medication usage by patients is among the most pressing issues in healthcare.
"I know that everybody is under increasing time pressures," he adds, "but it’s hard to imagine a larger problem than not getting people the drugs they need to prevent really significant clinical outcomes."
Visit our website for more information on medication issues.
A recent Dartmouth Atlas Project report that highlights regional differences for prescription drug use among Medicare patients across the U.S. provides insights into best practices related to effective and high-risk prescription drug therapy.
The report [PDF] found geographic disparity in the total use of prescription medications, variations in effective prescription care, dissimilarities in the use of potentially harmful medications, and differences in total spending on prescription drugs.
Lead author Jeffrey Munson, MD, MSCE, says he expected some geographic variation in the use of discretionary medications, but was surprised by the discrepancy in patients' medication usage. For example, in San Angelo, Texas, 91.4% of heart attack survivors in 2008–2009 filled at least one prescription for beta blockers in the year after their discharge, compared with just 62.5% of the same population of patients in Salem, Ore.
"Clearly, there are regions of the country that have figured out how to best handle certain aspects of medication usage," says Dr. Munson, assistant professor at the Dartmouth Institute for Health Policy & Clinical Practice in Lebanon, N.H. "Instead of just looking at regions that are high performing and saying, 'Wow, that seems to be a really high bar they've set, I'm not sure we can achieve it,' I wonder if maybe it's time we look at those regions and say, 'How are you achieving those very high standards, and what about what you do can I do where I live.'"
The Dartmouth Atlas Project report documents geographic variation in healthcare utilization unrelated to outcome and offers an extensive database for comparison by state, county, region, and facility. Dr. Munson says he understands that healthcare reform is pushing hospitalists and other physicians to focus on many new issues, but that medication usage by patients is among the most pressing issues in healthcare.
"I know that everybody is under increasing time pressures," he adds, "but it’s hard to imagine a larger problem than not getting people the drugs they need to prevent really significant clinical outcomes."
Visit our website for more information on medication issues.
A recent Dartmouth Atlas Project report that highlights regional differences for prescription drug use among Medicare patients across the U.S. provides insights into best practices related to effective and high-risk prescription drug therapy.
The report [PDF] found geographic disparity in the total use of prescription medications, variations in effective prescription care, dissimilarities in the use of potentially harmful medications, and differences in total spending on prescription drugs.
Lead author Jeffrey Munson, MD, MSCE, says he expected some geographic variation in the use of discretionary medications, but was surprised by the discrepancy in patients' medication usage. For example, in San Angelo, Texas, 91.4% of heart attack survivors in 2008–2009 filled at least one prescription for beta blockers in the year after their discharge, compared with just 62.5% of the same population of patients in Salem, Ore.
"Clearly, there are regions of the country that have figured out how to best handle certain aspects of medication usage," says Dr. Munson, assistant professor at the Dartmouth Institute for Health Policy & Clinical Practice in Lebanon, N.H. "Instead of just looking at regions that are high performing and saying, 'Wow, that seems to be a really high bar they've set, I'm not sure we can achieve it,' I wonder if maybe it's time we look at those regions and say, 'How are you achieving those very high standards, and what about what you do can I do where I live.'"
The Dartmouth Atlas Project report documents geographic variation in healthcare utilization unrelated to outcome and offers an extensive database for comparison by state, county, region, and facility. Dr. Munson says he understands that healthcare reform is pushing hospitalists and other physicians to focus on many new issues, but that medication usage by patients is among the most pressing issues in healthcare.
"I know that everybody is under increasing time pressures," he adds, "but it’s hard to imagine a larger problem than not getting people the drugs they need to prevent really significant clinical outcomes."
Visit our website for more information on medication issues.
Risk of Perioperative Morbidity, Post-Op Mortality Higher for Current Smokers
Clinical question: Is there an association between current and past smoking on outcomes among patients having major surgery?
Background: Smoking is associated with adverse postoperative outcomes, but it is not known whether the associations are dose-dependent or limited to patients with smoking-related diseases. Smoking-related effects on postoperative events among patients having major surgery are also not well established.
Study design: Retrospective cohort study.
Setting: Four hundred forty-eight non-VA hospitals across the U.S., Canada, Lebanon, and the United Arab Emirates.
Synopsis: Data from 607,558 adult patients undergoing major surgery were obtained from the American College of Surgeons (ACS) National Surgical Quality Improvement Program (NSQIP) database. After adjusting for confounders (cardiopulmonary diseases and cancer), the effects of current and past smoking (quit >1 year prior) on 30-day postoperative outcomes were measured.
There were 125,192 (21%) current smokers and 78,763 (13%) past smokers. Increased odds of post-op mortality were noted in current smokers only (odds ratio [OR] 1.17; 95% CI, 1.10–1.24). The adjusted odds ratios were higher for arterial and respiratory events among current smokers compared with past smokers (OR 1.65; 95% CI, 1.51–1.81 vs. OR 1.20; CI, 1.09–1.31 for arterial events, respectively) and (OR, 1.45; CI, 1.40–1.51 vs. OR, 1.13; CI, 1.08–1.18, for respiratory events, respectively). No significant effects on venous events were observed.
There was an increased adjusted odds of mortality for current smokers with <10 pack-years, while the effects on arterial and respiratory events increased incrementally with increased pack-years. Smoking was associated with adverse post-op outcomes regardless of smoking-related diseases. Variability in hospital quality or surgical strategies may have confounded the results.
Bottom line: Among patients undergoing major surgery, current but not past smoking was associated with higher mortality; smoking cessation for at least a year prior to surgery may decrease postoperative adverse events.
Citation: Musallam KM, Rosendaal FR, Zaatari G, et al. Smoking and the risk of mortality and vascular and respiratory events in patients undergoing major surgery. JAMA Surg. 2013;148:755-762.
Clinical question: Is there an association between current and past smoking on outcomes among patients having major surgery?
Background: Smoking is associated with adverse postoperative outcomes, but it is not known whether the associations are dose-dependent or limited to patients with smoking-related diseases. Smoking-related effects on postoperative events among patients having major surgery are also not well established.
Study design: Retrospective cohort study.
Setting: Four hundred forty-eight non-VA hospitals across the U.S., Canada, Lebanon, and the United Arab Emirates.
Synopsis: Data from 607,558 adult patients undergoing major surgery were obtained from the American College of Surgeons (ACS) National Surgical Quality Improvement Program (NSQIP) database. After adjusting for confounders (cardiopulmonary diseases and cancer), the effects of current and past smoking (quit >1 year prior) on 30-day postoperative outcomes were measured.
There were 125,192 (21%) current smokers and 78,763 (13%) past smokers. Increased odds of post-op mortality were noted in current smokers only (odds ratio [OR] 1.17; 95% CI, 1.10–1.24). The adjusted odds ratios were higher for arterial and respiratory events among current smokers compared with past smokers (OR 1.65; 95% CI, 1.51–1.81 vs. OR 1.20; CI, 1.09–1.31 for arterial events, respectively) and (OR, 1.45; CI, 1.40–1.51 vs. OR, 1.13; CI, 1.08–1.18, for respiratory events, respectively). No significant effects on venous events were observed.
There was an increased adjusted odds of mortality for current smokers with <10 pack-years, while the effects on arterial and respiratory events increased incrementally with increased pack-years. Smoking was associated with adverse post-op outcomes regardless of smoking-related diseases. Variability in hospital quality or surgical strategies may have confounded the results.
Bottom line: Among patients undergoing major surgery, current but not past smoking was associated with higher mortality; smoking cessation for at least a year prior to surgery may decrease postoperative adverse events.
Citation: Musallam KM, Rosendaal FR, Zaatari G, et al. Smoking and the risk of mortality and vascular and respiratory events in patients undergoing major surgery. JAMA Surg. 2013;148:755-762.
Clinical question: Is there an association between current and past smoking on outcomes among patients having major surgery?
Background: Smoking is associated with adverse postoperative outcomes, but it is not known whether the associations are dose-dependent or limited to patients with smoking-related diseases. Smoking-related effects on postoperative events among patients having major surgery are also not well established.
Study design: Retrospective cohort study.
Setting: Four hundred forty-eight non-VA hospitals across the U.S., Canada, Lebanon, and the United Arab Emirates.
Synopsis: Data from 607,558 adult patients undergoing major surgery were obtained from the American College of Surgeons (ACS) National Surgical Quality Improvement Program (NSQIP) database. After adjusting for confounders (cardiopulmonary diseases and cancer), the effects of current and past smoking (quit >1 year prior) on 30-day postoperative outcomes were measured.
There were 125,192 (21%) current smokers and 78,763 (13%) past smokers. Increased odds of post-op mortality were noted in current smokers only (odds ratio [OR] 1.17; 95% CI, 1.10–1.24). The adjusted odds ratios were higher for arterial and respiratory events among current smokers compared with past smokers (OR 1.65; 95% CI, 1.51–1.81 vs. OR 1.20; CI, 1.09–1.31 for arterial events, respectively) and (OR, 1.45; CI, 1.40–1.51 vs. OR, 1.13; CI, 1.08–1.18, for respiratory events, respectively). No significant effects on venous events were observed.
There was an increased adjusted odds of mortality for current smokers with <10 pack-years, while the effects on arterial and respiratory events increased incrementally with increased pack-years. Smoking was associated with adverse post-op outcomes regardless of smoking-related diseases. Variability in hospital quality or surgical strategies may have confounded the results.
Bottom line: Among patients undergoing major surgery, current but not past smoking was associated with higher mortality; smoking cessation for at least a year prior to surgery may decrease postoperative adverse events.
Citation: Musallam KM, Rosendaal FR, Zaatari G, et al. Smoking and the risk of mortality and vascular and respiratory events in patients undergoing major surgery. JAMA Surg. 2013;148:755-762.
Medical conferences going digital
The medical conferences of the future made a preview appearance at this year’s Transcatheter Cardiovascular Therapeutics annual meeting in San Francisco. Paperless, electronic, interactive, and definitely high tech it was.
Every paid attendee was offered a new Samsung tablet computer, preloaded with pertinent apps and information, to personalize and keep if they wanted or return at the end of the meeting. If attendees preferred to download the apps to their own devices, that was fine too, and many of them did. (I got a loaner through the press room, and found it easy to use.)
Rather than tack the cost of the tablets onto registration fees, the organizers shifted funds from the no-longer-needed bulky printed programs and other materials to pay for the tablets, according to the Cardiovascular Research Foundation, cosponsor of the TCT meeting with the American College of Cardiology. No funds from industry were solicited for the tablets, no advertising appeared on the home screens, and the tablets were not being used to mine for user data of any kind, but the preloaded apps did contain some advertisements.
Paperless medical conferences are not new – many conferences eschew pulp these days, providing materials on zip drives instead of printed programs that attendees can load onto their computers. And apps for the larger medical conferences now are commonplace, too, for those who have their own smartphones or tablets. But this is the first time I’ve seen a conference give out tablets and include interactive social media features, convenient continuing medical education mechanisms, and more.
Through the apps, attendees could navigate the convention center; view abstracts; download speaker slides and disclosures; watch live cases; take notes; contact some faculty; find shuttle buses, hotels, and restaurants; and access exhibition materials. After attending a session, they could log their hours, write a review, and apply for CME credits through the apps. If they were willing to enable certain settings, they could see who else at the meeting was in their vicinity, and communicate with them.
Each of the major sessions I covered included a "digital moderator" in addition to the regular moderator. Instead of standing in line at microphones to ask questions, members of the audience texted comments and questions that appeared on a screen to the side of the main screen showing the presenter’s slides, so everyone could see them in real time. This feature wasn’t as much used as one might fear – doctors were still paying attention to the speaker, not staring down at their devices, for the most part. From what I could see, the digital moderators provided most of the texted comments and questions, though at one session the live moderator noted that audience texts were asking the speaker to comment about stroke risk, so he raised the question.
Keep in mind, the TCT always has been one of the most high-tech conferences happening in a very high-tech specialty, interventional cardiology. The typical setup in their main forum was similar to that in past meetings, a multitasking-palooza featuring a long dais of speakers and multiple video screens, with individual headsets that let you tune into whichever "channel" interests you most at the moment. Screens with live cases flank either end, with the presenter and his or her slides in the middle and screens promoting upcoming sessions and showing the audience texts in between the other screens.
TCT comes to San Francisco regularly because the city has the infrastructure to support these technologic demands, a spokeswoman in their press room told me. Some other locations haven’t been able to handle their needs.
I wondered if the technology will be so appealing that attendees might prefer virtual attendance rather than having to be there. It’s possible, she said, but unlikely. Like most people, these doctors value their face time.
On Twitter @sherryboschert
The medical conferences of the future made a preview appearance at this year’s Transcatheter Cardiovascular Therapeutics annual meeting in San Francisco. Paperless, electronic, interactive, and definitely high tech it was.
Every paid attendee was offered a new Samsung tablet computer, preloaded with pertinent apps and information, to personalize and keep if they wanted or return at the end of the meeting. If attendees preferred to download the apps to their own devices, that was fine too, and many of them did. (I got a loaner through the press room, and found it easy to use.)
Rather than tack the cost of the tablets onto registration fees, the organizers shifted funds from the no-longer-needed bulky printed programs and other materials to pay for the tablets, according to the Cardiovascular Research Foundation, cosponsor of the TCT meeting with the American College of Cardiology. No funds from industry were solicited for the tablets, no advertising appeared on the home screens, and the tablets were not being used to mine for user data of any kind, but the preloaded apps did contain some advertisements.
Paperless medical conferences are not new – many conferences eschew pulp these days, providing materials on zip drives instead of printed programs that attendees can load onto their computers. And apps for the larger medical conferences now are commonplace, too, for those who have their own smartphones or tablets. But this is the first time I’ve seen a conference give out tablets and include interactive social media features, convenient continuing medical education mechanisms, and more.
Through the apps, attendees could navigate the convention center; view abstracts; download speaker slides and disclosures; watch live cases; take notes; contact some faculty; find shuttle buses, hotels, and restaurants; and access exhibition materials. After attending a session, they could log their hours, write a review, and apply for CME credits through the apps. If they were willing to enable certain settings, they could see who else at the meeting was in their vicinity, and communicate with them.
Each of the major sessions I covered included a "digital moderator" in addition to the regular moderator. Instead of standing in line at microphones to ask questions, members of the audience texted comments and questions that appeared on a screen to the side of the main screen showing the presenter’s slides, so everyone could see them in real time. This feature wasn’t as much used as one might fear – doctors were still paying attention to the speaker, not staring down at their devices, for the most part. From what I could see, the digital moderators provided most of the texted comments and questions, though at one session the live moderator noted that audience texts were asking the speaker to comment about stroke risk, so he raised the question.
Keep in mind, the TCT always has been one of the most high-tech conferences happening in a very high-tech specialty, interventional cardiology. The typical setup in their main forum was similar to that in past meetings, a multitasking-palooza featuring a long dais of speakers and multiple video screens, with individual headsets that let you tune into whichever "channel" interests you most at the moment. Screens with live cases flank either end, with the presenter and his or her slides in the middle and screens promoting upcoming sessions and showing the audience texts in between the other screens.
TCT comes to San Francisco regularly because the city has the infrastructure to support these technologic demands, a spokeswoman in their press room told me. Some other locations haven’t been able to handle their needs.
I wondered if the technology will be so appealing that attendees might prefer virtual attendance rather than having to be there. It’s possible, she said, but unlikely. Like most people, these doctors value their face time.
On Twitter @sherryboschert
The medical conferences of the future made a preview appearance at this year’s Transcatheter Cardiovascular Therapeutics annual meeting in San Francisco. Paperless, electronic, interactive, and definitely high tech it was.
Every paid attendee was offered a new Samsung tablet computer, preloaded with pertinent apps and information, to personalize and keep if they wanted or return at the end of the meeting. If attendees preferred to download the apps to their own devices, that was fine too, and many of them did. (I got a loaner through the press room, and found it easy to use.)
Rather than tack the cost of the tablets onto registration fees, the organizers shifted funds from the no-longer-needed bulky printed programs and other materials to pay for the tablets, according to the Cardiovascular Research Foundation, cosponsor of the TCT meeting with the American College of Cardiology. No funds from industry were solicited for the tablets, no advertising appeared on the home screens, and the tablets were not being used to mine for user data of any kind, but the preloaded apps did contain some advertisements.
Paperless medical conferences are not new – many conferences eschew pulp these days, providing materials on zip drives instead of printed programs that attendees can load onto their computers. And apps for the larger medical conferences now are commonplace, too, for those who have their own smartphones or tablets. But this is the first time I’ve seen a conference give out tablets and include interactive social media features, convenient continuing medical education mechanisms, and more.
Through the apps, attendees could navigate the convention center; view abstracts; download speaker slides and disclosures; watch live cases; take notes; contact some faculty; find shuttle buses, hotels, and restaurants; and access exhibition materials. After attending a session, they could log their hours, write a review, and apply for CME credits through the apps. If they were willing to enable certain settings, they could see who else at the meeting was in their vicinity, and communicate with them.
Each of the major sessions I covered included a "digital moderator" in addition to the regular moderator. Instead of standing in line at microphones to ask questions, members of the audience texted comments and questions that appeared on a screen to the side of the main screen showing the presenter’s slides, so everyone could see them in real time. This feature wasn’t as much used as one might fear – doctors were still paying attention to the speaker, not staring down at their devices, for the most part. From what I could see, the digital moderators provided most of the texted comments and questions, though at one session the live moderator noted that audience texts were asking the speaker to comment about stroke risk, so he raised the question.
Keep in mind, the TCT always has been one of the most high-tech conferences happening in a very high-tech specialty, interventional cardiology. The typical setup in their main forum was similar to that in past meetings, a multitasking-palooza featuring a long dais of speakers and multiple video screens, with individual headsets that let you tune into whichever "channel" interests you most at the moment. Screens with live cases flank either end, with the presenter and his or her slides in the middle and screens promoting upcoming sessions and showing the audience texts in between the other screens.
TCT comes to San Francisco regularly because the city has the infrastructure to support these technologic demands, a spokeswoman in their press room told me. Some other locations haven’t been able to handle their needs.
I wondered if the technology will be so appealing that attendees might prefer virtual attendance rather than having to be there. It’s possible, she said, but unlikely. Like most people, these doctors value their face time.
On Twitter @sherryboschert
A rapid-fire update on lasers in dermatologic surgery
If you have 5 minutes and need an update on the latest advancements in lasers in dermatologic surgery, watch our interview with Dr. Elizabeth Tanzi of Washington Institute of Dermatologic Surgery. Dr. Tanzi talks about the new trends in laser treatments, her two favorite laser machines, and shares her thoughts on the future of lasers.
If you have 5 minutes and need an update on the latest advancements in lasers in dermatologic surgery, watch our interview with Dr. Elizabeth Tanzi of Washington Institute of Dermatologic Surgery. Dr. Tanzi talks about the new trends in laser treatments, her two favorite laser machines, and shares her thoughts on the future of lasers.
If you have 5 minutes and need an update on the latest advancements in lasers in dermatologic surgery, watch our interview with Dr. Elizabeth Tanzi of Washington Institute of Dermatologic Surgery. Dr. Tanzi talks about the new trends in laser treatments, her two favorite laser machines, and shares her thoughts on the future of lasers.
Impact of psoriasis on sexual activity
One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.
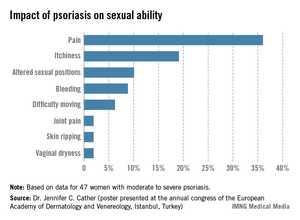
One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.

One third of a group of women with psoriasis reported that the pain associated with their condition interfered with their sexual activity, according to findings from a survey presented by Dr. Jennifer C. Cather.
Based on responses from a survey of 60 women with moderate to severe psoriasis, the specific complaints that were the most common ways in which psoriasis interfered with sexual activity were itchiness (19%), the need to adjust sexual position (10%), and bleeding (9%), Dr. Cather reported at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar. The survey was part of an effort to determine the impact of psoriasis on women’s sexual activity, desires, and relationships.
The data were previously presented in a poster at the annual congress of the European Academy of Dermatology and Venereology (Istanbul.
SDEF and this news organization are owned by Frontline Medical Communications. Dr. Cather disclosed that she is a consultant, speaker, or researcher for AbbVie, Novartis, Leo, Janssen, Amgen, Celgene, Merck, and Pfizer.

EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
New and Noteworthy Information—November 2013
In early December 2013, the FDA will formally recommend to the US Department of Health and Human Services (HHS) that hydrocodone combination products be reclassified as Schedule II drugs. The proposed change would tighten the controls on these products, which now are classified as Schedule III. The recommendation follows the US Drug Enforcement Administration’s (DEA) 2009 request for guidance from HHS regarding hydrocodone combination products such as Vicodin. The FDA’s determination is the result of an analysis of the scientific literature, a review of hundreds of public comments on the issue, and several public meetings, according to a statement by Janet Woodcock, MD, Director of the Center for Drug Evaluation and Research. The recommendation will influence the DEA’s final decision on the appropriate scheduling of these products.
Influenza vaccination may reduce patients’ risk of major adverse cardiovascular events, including stroke, according to a meta-analysis published October 23 in JAMA. Researchers conducted a systematic review of randomized clinical trials listed in MEDLINE, EMBASE, and the Cochrane Library Central Register of Controlled Trials that compared influenza vaccine with placebo or control in patients at high risk of cardiovascular disease. Six trials encompassing 6,735 patients were included. Influenza vaccine was associated with a lower risk of composite cardiovascular events (2.9% vs 4.7%) in published trials. A treatment interaction was detected between patients with and without recent acute coronary syndrome. The greatest treatment effect was observed among the highest-risk patients with more active coronary disease, and a larger trial is warranted to assess these findings, said the researchers.
Short sleep duration and poor sleep quality may be associated with greater β-amyloid burden among community-dwelling older adults, according to research published online ahead of print October 21 in JAMA Neurology. Investigators performed a cross-sectional study of 70 adult participants (mean age, 76) in the neuroimaging substudy of the Baltimore Longitudinal Study of Aging. The study’s main outcome measure was β-amyloid burden, measured by carbon 11-labeled Pittsburgh compound B PET distribution volume ratios. After adjustment for potential confounders, the researchers found an association between reports of shorter sleep duration and greater β-amyloid burden, measured by mean cortical distribution volume ratio and precuneus distribution volume ratio. Reports of lower sleep quality were associated with greater β-amyloid burden, measured by precuneus distribution volume ratio.
High blood glucose levels may adversely affect cognition, even among patients without type 2 diabetes or impaired glucose tolerance, researchers reported online ahead of print October 23 in Neurology. The effect may be mediated by structural changes in learning-relevant brain areas, the authors noted. The group tested memory in 141 individuals using the Rey Auditory Verbal Learning Test and acquired peripheral levels of fasting HbA1c, glucose, and insulin. Clinicians performed 3-T MRI scans to assess hippocampal volume and microstructure. Lower HbA1c and glucose levels were significantly associated with better scores in delayed recall, learning ability, and memory consolidation. In multiple regression models, HbA1c remained strongly associated with memory performance. Mediation analyses indicated that beneficial effects of lower HbA1c on memory are partly mediated by hippocampal volume and microstructure.
Among older adults, arterial stiffness may be associated with b-amyloid plaque deposition in the brain, independent of blood pressure and APOE ε4 allele, according to a study published online ahead of print October 16 in Neurology. Investigators studied 91 dementia-free participants between ages 83 and 96. Participants underwent brain MRI and PET imaging with Pittsburgh compound B. The researchers measured resting blood pressure (BP), mean arterial pressure (MAP), and arterial stiffness by pulse wave velocity (PWV) in the central, peripheral, and mixed vascular beds. A total of 44 subjects were β-amyloid positive on PET scan. The investigators found that β-amyloid deposition was associated with mixed PWV, systolic BP, and MAP. One SD increase in brachial ankle PWV resulted in a twofold increase in the odds of being β-amyloid positive.
The FDA has approved Vizamyl (flutemetamol F 18 injection), a radioactive diagnostic drug, for use with PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia. Vizamyl attaches to β-amyloid and produces a PET image that is used to evaluate the presence of β-amyloid. The drug’s effectiveness was established in two clinical studies of 384 participants with a range of cognitive function. All participants were injected with Vizamyl and scanned. The images were interpreted by five independent readers masked to all clinical information. A portion of scan results was also confirmed by autopsy. Following the approval of Amyvid (Florbetapir F 18 injection) in 2012, Vizamyl, manufactured by Medi-Physics (Arlington Heights, Illinois), becomes the second diagnostic drug available for visualizing β-amyloid on a PET scan of the brain.
Clostridium perfringens type B, an epsilon toxin-secreting bacillus, may trigger multiple sclerosis (MS), according to research published October 16 in PLOS One. After detecting C. perfringens type B in a woman with MS, investigators tested blood and CSF from patients with MS and controls for antibody reactivity to the epsilon toxin. Levels of epsilon toxin antibodies were 10 times higher in patients with MS, compared with controls. After examining stool samples, the study authors found the human commensal C. perfringens type A in approximately 50% of healthy controls, compared with 23% of patients with MS. C. perfringens epsilon toxin fits mechanistically with nascent MS lesion formation because these lesions are characterized by blood–brain barrier permeability and oligodendrocyte cell death in the absence of an adaptive immune infiltrate, said the researchers.
Chronic cerebrospinal venous insufficiency (CCSVI), which has been proposed as a contributor to multiple sclerosis (MS), occurs rarely in patients with MS and in controls, according to a study published online ahead of print October 8 in Lancet. Researchers performed an assessor-blinded, case-control, multicenter study of 79 people with MS, 55 unaffected siblings, and 43 unrelated healthy volunteers. Catheter venography criteria for CCSVI were positive for 2% of people with MS, 2% of siblings, and 3% of unrelated controls. Greater than 50% narrowing of any major vein was present in 74% of people with MS, 66% of siblings, and 70% of unrelated controls. The Zamboni ultrasound criteria are neither sensitive nor specific for narrowing on catheter venography, and the significance of venous narrowing to MS remains unknown, said the investigators.
Measures of α-synuclein deposition in cutaneous autonomic nerves may be a useful biomarker in patients with Parkinson’s disease, according to research published online ahead of print October 2 in Neurology. Investigators examined 20 patients with Parkinson’s disease and 14 age- and sex-matched control subjects. The researchers performed autonomic testing and skin biopsies at the distal leg, distal thigh, and proximal thigh for all participants. Deposition of α-synuclein and the density of intraepidermal, sudomotor, and pilomotor nerve fibers were measured. The investigators normalized α-synuclein deposition to nerve fiber density. Patients with Parkinson’s disease had greater α-synuclein deposition and higher α-synuclein ratios than controls within pilomotor nerves and sudomotor nerves, but not sensory nerves. Higher α-synuclein ratios correlated with Hoehn and Yahr scores, sympathetic adrenergic function, and parasympathetic function.
Depression may be an independent risk factor for Parkinson’s disease, according to research published October 22 in Neurology. Investigators conducted a retrospective study of 4,634 patients with depression and 18,544 matched controls who were selected from a national health insurance database. Patients were observed for a maximum of 10 years to determine the rates of new-onset Parkinson’s disease. Cox regression was used to identify the predictors of the disease. During the follow-up period, 66 patients with depression and 97 controls were diagnosed with Parkinson’s disease. After adjusting for age and sex, the researchers found that patients with depression were 3.24 times more likely to develop Parkinson’s disease, compared with the control patients. The investigators observed that age and difficult-to-treat depression are independent risk factors for Parkinson’s disease in patients with depression.
The levels of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) in red blood cells may have no association with age-associated cognitive decline, researchers reported in the October 22 issue of Neurology. The investigators conducted a retrospective cohort study of 2,157 women with normal cognition who were followed with annual cognitive testing for a median of 5.9 years. End points were composite cognitive function and performance in seven cognitive domains. After adjustment for demographic, clinical, and behavioral characteristics, the investigators found no significant cross-sectional cognitive differences between women in the high and low DHA and EPA tertiles at the first annual cognitive battery. In addition, no significant differences were found between the high and low DHA and EPA tertiles in the rate of cognitive change over time.
Common psychosocial stressors (eg, divorce, widowhood, work problems, and illness in a relative) may have severe and long-standing physiologic and psychologic consequences such as dementia, according to research published September 30 in BMJ Open. In a prospective longitudinal population study, clinicians performed psychiatric examinations for 800 women born in 1914, 1918, 1922, and 1930. Baseline examinations took place in 1968, and follow-up occurred in 1974, 1980, 1992, 2000, and 2005. During follow-up, 153 women developed dementia. The number of psychosocial stressors in 1968 was associated with higher incidence of dementia and Alzheimer’s disease (AD) between 1968 and 2005 in multivariate Cox regressions. The number of psychosocial stressors in 1968 was also associated with distress in 1968, 1974, 1980, 2000, and 2005 in multivariate logistic regressions.
Aggressive medical management may provide more benefit than percutaneous transluminal angioplasty and stenting (PTAS) for high-risk patients with atherosclerotic intracranial arterial stenosis, according to a study published online ahead of print October 26 in Lancet. Investigators randomized 451 patients with recent transient ischemic attack or stroke to aggressive medical management or aggressive medical management plus stenting with the Wingspan stent. The cumulative probability of stroke or death was smaller in the medical group vs the PTAS group. Beyond 30 days, 10% of patients in the medical group and 10% of patients in the stenting group had a primary end point. The absolute differences in the primary end point rates between the two groups were 7.1% at year 1, 6.5% at year 2 and 9.0% at year 3.
—Erik Greb
Senior Associate Editor
In early December 2013, the FDA will formally recommend to the US Department of Health and Human Services (HHS) that hydrocodone combination products be reclassified as Schedule II drugs. The proposed change would tighten the controls on these products, which now are classified as Schedule III. The recommendation follows the US Drug Enforcement Administration’s (DEA) 2009 request for guidance from HHS regarding hydrocodone combination products such as Vicodin. The FDA’s determination is the result of an analysis of the scientific literature, a review of hundreds of public comments on the issue, and several public meetings, according to a statement by Janet Woodcock, MD, Director of the Center for Drug Evaluation and Research. The recommendation will influence the DEA’s final decision on the appropriate scheduling of these products.
Influenza vaccination may reduce patients’ risk of major adverse cardiovascular events, including stroke, according to a meta-analysis published October 23 in JAMA. Researchers conducted a systematic review of randomized clinical trials listed in MEDLINE, EMBASE, and the Cochrane Library Central Register of Controlled Trials that compared influenza vaccine with placebo or control in patients at high risk of cardiovascular disease. Six trials encompassing 6,735 patients were included. Influenza vaccine was associated with a lower risk of composite cardiovascular events (2.9% vs 4.7%) in published trials. A treatment interaction was detected between patients with and without recent acute coronary syndrome. The greatest treatment effect was observed among the highest-risk patients with more active coronary disease, and a larger trial is warranted to assess these findings, said the researchers.
Short sleep duration and poor sleep quality may be associated with greater β-amyloid burden among community-dwelling older adults, according to research published online ahead of print October 21 in JAMA Neurology. Investigators performed a cross-sectional study of 70 adult participants (mean age, 76) in the neuroimaging substudy of the Baltimore Longitudinal Study of Aging. The study’s main outcome measure was β-amyloid burden, measured by carbon 11-labeled Pittsburgh compound B PET distribution volume ratios. After adjustment for potential confounders, the researchers found an association between reports of shorter sleep duration and greater β-amyloid burden, measured by mean cortical distribution volume ratio and precuneus distribution volume ratio. Reports of lower sleep quality were associated with greater β-amyloid burden, measured by precuneus distribution volume ratio.
High blood glucose levels may adversely affect cognition, even among patients without type 2 diabetes or impaired glucose tolerance, researchers reported online ahead of print October 23 in Neurology. The effect may be mediated by structural changes in learning-relevant brain areas, the authors noted. The group tested memory in 141 individuals using the Rey Auditory Verbal Learning Test and acquired peripheral levels of fasting HbA1c, glucose, and insulin. Clinicians performed 3-T MRI scans to assess hippocampal volume and microstructure. Lower HbA1c and glucose levels were significantly associated with better scores in delayed recall, learning ability, and memory consolidation. In multiple regression models, HbA1c remained strongly associated with memory performance. Mediation analyses indicated that beneficial effects of lower HbA1c on memory are partly mediated by hippocampal volume and microstructure.
Among older adults, arterial stiffness may be associated with b-amyloid plaque deposition in the brain, independent of blood pressure and APOE ε4 allele, according to a study published online ahead of print October 16 in Neurology. Investigators studied 91 dementia-free participants between ages 83 and 96. Participants underwent brain MRI and PET imaging with Pittsburgh compound B. The researchers measured resting blood pressure (BP), mean arterial pressure (MAP), and arterial stiffness by pulse wave velocity (PWV) in the central, peripheral, and mixed vascular beds. A total of 44 subjects were β-amyloid positive on PET scan. The investigators found that β-amyloid deposition was associated with mixed PWV, systolic BP, and MAP. One SD increase in brachial ankle PWV resulted in a twofold increase in the odds of being β-amyloid positive.
The FDA has approved Vizamyl (flutemetamol F 18 injection), a radioactive diagnostic drug, for use with PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia. Vizamyl attaches to β-amyloid and produces a PET image that is used to evaluate the presence of β-amyloid. The drug’s effectiveness was established in two clinical studies of 384 participants with a range of cognitive function. All participants were injected with Vizamyl and scanned. The images were interpreted by five independent readers masked to all clinical information. A portion of scan results was also confirmed by autopsy. Following the approval of Amyvid (Florbetapir F 18 injection) in 2012, Vizamyl, manufactured by Medi-Physics (Arlington Heights, Illinois), becomes the second diagnostic drug available for visualizing β-amyloid on a PET scan of the brain.
Clostridium perfringens type B, an epsilon toxin-secreting bacillus, may trigger multiple sclerosis (MS), according to research published October 16 in PLOS One. After detecting C. perfringens type B in a woman with MS, investigators tested blood and CSF from patients with MS and controls for antibody reactivity to the epsilon toxin. Levels of epsilon toxin antibodies were 10 times higher in patients with MS, compared with controls. After examining stool samples, the study authors found the human commensal C. perfringens type A in approximately 50% of healthy controls, compared with 23% of patients with MS. C. perfringens epsilon toxin fits mechanistically with nascent MS lesion formation because these lesions are characterized by blood–brain barrier permeability and oligodendrocyte cell death in the absence of an adaptive immune infiltrate, said the researchers.
Chronic cerebrospinal venous insufficiency (CCSVI), which has been proposed as a contributor to multiple sclerosis (MS), occurs rarely in patients with MS and in controls, according to a study published online ahead of print October 8 in Lancet. Researchers performed an assessor-blinded, case-control, multicenter study of 79 people with MS, 55 unaffected siblings, and 43 unrelated healthy volunteers. Catheter venography criteria for CCSVI were positive for 2% of people with MS, 2% of siblings, and 3% of unrelated controls. Greater than 50% narrowing of any major vein was present in 74% of people with MS, 66% of siblings, and 70% of unrelated controls. The Zamboni ultrasound criteria are neither sensitive nor specific for narrowing on catheter venography, and the significance of venous narrowing to MS remains unknown, said the investigators.
Measures of α-synuclein deposition in cutaneous autonomic nerves may be a useful biomarker in patients with Parkinson’s disease, according to research published online ahead of print October 2 in Neurology. Investigators examined 20 patients with Parkinson’s disease and 14 age- and sex-matched control subjects. The researchers performed autonomic testing and skin biopsies at the distal leg, distal thigh, and proximal thigh for all participants. Deposition of α-synuclein and the density of intraepidermal, sudomotor, and pilomotor nerve fibers were measured. The investigators normalized α-synuclein deposition to nerve fiber density. Patients with Parkinson’s disease had greater α-synuclein deposition and higher α-synuclein ratios than controls within pilomotor nerves and sudomotor nerves, but not sensory nerves. Higher α-synuclein ratios correlated with Hoehn and Yahr scores, sympathetic adrenergic function, and parasympathetic function.
Depression may be an independent risk factor for Parkinson’s disease, according to research published October 22 in Neurology. Investigators conducted a retrospective study of 4,634 patients with depression and 18,544 matched controls who were selected from a national health insurance database. Patients were observed for a maximum of 10 years to determine the rates of new-onset Parkinson’s disease. Cox regression was used to identify the predictors of the disease. During the follow-up period, 66 patients with depression and 97 controls were diagnosed with Parkinson’s disease. After adjusting for age and sex, the researchers found that patients with depression were 3.24 times more likely to develop Parkinson’s disease, compared with the control patients. The investigators observed that age and difficult-to-treat depression are independent risk factors for Parkinson’s disease in patients with depression.
The levels of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) in red blood cells may have no association with age-associated cognitive decline, researchers reported in the October 22 issue of Neurology. The investigators conducted a retrospective cohort study of 2,157 women with normal cognition who were followed with annual cognitive testing for a median of 5.9 years. End points were composite cognitive function and performance in seven cognitive domains. After adjustment for demographic, clinical, and behavioral characteristics, the investigators found no significant cross-sectional cognitive differences between women in the high and low DHA and EPA tertiles at the first annual cognitive battery. In addition, no significant differences were found between the high and low DHA and EPA tertiles in the rate of cognitive change over time.
Common psychosocial stressors (eg, divorce, widowhood, work problems, and illness in a relative) may have severe and long-standing physiologic and psychologic consequences such as dementia, according to research published September 30 in BMJ Open. In a prospective longitudinal population study, clinicians performed psychiatric examinations for 800 women born in 1914, 1918, 1922, and 1930. Baseline examinations took place in 1968, and follow-up occurred in 1974, 1980, 1992, 2000, and 2005. During follow-up, 153 women developed dementia. The number of psychosocial stressors in 1968 was associated with higher incidence of dementia and Alzheimer’s disease (AD) between 1968 and 2005 in multivariate Cox regressions. The number of psychosocial stressors in 1968 was also associated with distress in 1968, 1974, 1980, 2000, and 2005 in multivariate logistic regressions.
Aggressive medical management may provide more benefit than percutaneous transluminal angioplasty and stenting (PTAS) for high-risk patients with atherosclerotic intracranial arterial stenosis, according to a study published online ahead of print October 26 in Lancet. Investigators randomized 451 patients with recent transient ischemic attack or stroke to aggressive medical management or aggressive medical management plus stenting with the Wingspan stent. The cumulative probability of stroke or death was smaller in the medical group vs the PTAS group. Beyond 30 days, 10% of patients in the medical group and 10% of patients in the stenting group had a primary end point. The absolute differences in the primary end point rates between the two groups were 7.1% at year 1, 6.5% at year 2 and 9.0% at year 3.
—Erik Greb
Senior Associate Editor
In early December 2013, the FDA will formally recommend to the US Department of Health and Human Services (HHS) that hydrocodone combination products be reclassified as Schedule II drugs. The proposed change would tighten the controls on these products, which now are classified as Schedule III. The recommendation follows the US Drug Enforcement Administration’s (DEA) 2009 request for guidance from HHS regarding hydrocodone combination products such as Vicodin. The FDA’s determination is the result of an analysis of the scientific literature, a review of hundreds of public comments on the issue, and several public meetings, according to a statement by Janet Woodcock, MD, Director of the Center for Drug Evaluation and Research. The recommendation will influence the DEA’s final decision on the appropriate scheduling of these products.
Influenza vaccination may reduce patients’ risk of major adverse cardiovascular events, including stroke, according to a meta-analysis published October 23 in JAMA. Researchers conducted a systematic review of randomized clinical trials listed in MEDLINE, EMBASE, and the Cochrane Library Central Register of Controlled Trials that compared influenza vaccine with placebo or control in patients at high risk of cardiovascular disease. Six trials encompassing 6,735 patients were included. Influenza vaccine was associated with a lower risk of composite cardiovascular events (2.9% vs 4.7%) in published trials. A treatment interaction was detected between patients with and without recent acute coronary syndrome. The greatest treatment effect was observed among the highest-risk patients with more active coronary disease, and a larger trial is warranted to assess these findings, said the researchers.
Short sleep duration and poor sleep quality may be associated with greater β-amyloid burden among community-dwelling older adults, according to research published online ahead of print October 21 in JAMA Neurology. Investigators performed a cross-sectional study of 70 adult participants (mean age, 76) in the neuroimaging substudy of the Baltimore Longitudinal Study of Aging. The study’s main outcome measure was β-amyloid burden, measured by carbon 11-labeled Pittsburgh compound B PET distribution volume ratios. After adjustment for potential confounders, the researchers found an association between reports of shorter sleep duration and greater β-amyloid burden, measured by mean cortical distribution volume ratio and precuneus distribution volume ratio. Reports of lower sleep quality were associated with greater β-amyloid burden, measured by precuneus distribution volume ratio.
High blood glucose levels may adversely affect cognition, even among patients without type 2 diabetes or impaired glucose tolerance, researchers reported online ahead of print October 23 in Neurology. The effect may be mediated by structural changes in learning-relevant brain areas, the authors noted. The group tested memory in 141 individuals using the Rey Auditory Verbal Learning Test and acquired peripheral levels of fasting HbA1c, glucose, and insulin. Clinicians performed 3-T MRI scans to assess hippocampal volume and microstructure. Lower HbA1c and glucose levels were significantly associated with better scores in delayed recall, learning ability, and memory consolidation. In multiple regression models, HbA1c remained strongly associated with memory performance. Mediation analyses indicated that beneficial effects of lower HbA1c on memory are partly mediated by hippocampal volume and microstructure.
Among older adults, arterial stiffness may be associated with b-amyloid plaque deposition in the brain, independent of blood pressure and APOE ε4 allele, according to a study published online ahead of print October 16 in Neurology. Investigators studied 91 dementia-free participants between ages 83 and 96. Participants underwent brain MRI and PET imaging with Pittsburgh compound B. The researchers measured resting blood pressure (BP), mean arterial pressure (MAP), and arterial stiffness by pulse wave velocity (PWV) in the central, peripheral, and mixed vascular beds. A total of 44 subjects were β-amyloid positive on PET scan. The investigators found that β-amyloid deposition was associated with mixed PWV, systolic BP, and MAP. One SD increase in brachial ankle PWV resulted in a twofold increase in the odds of being β-amyloid positive.
The FDA has approved Vizamyl (flutemetamol F 18 injection), a radioactive diagnostic drug, for use with PET imaging of the brain in adults being evaluated for Alzheimer’s disease and dementia. Vizamyl attaches to β-amyloid and produces a PET image that is used to evaluate the presence of β-amyloid. The drug’s effectiveness was established in two clinical studies of 384 participants with a range of cognitive function. All participants were injected with Vizamyl and scanned. The images were interpreted by five independent readers masked to all clinical information. A portion of scan results was also confirmed by autopsy. Following the approval of Amyvid (Florbetapir F 18 injection) in 2012, Vizamyl, manufactured by Medi-Physics (Arlington Heights, Illinois), becomes the second diagnostic drug available for visualizing β-amyloid on a PET scan of the brain.
Clostridium perfringens type B, an epsilon toxin-secreting bacillus, may trigger multiple sclerosis (MS), according to research published October 16 in PLOS One. After detecting C. perfringens type B in a woman with MS, investigators tested blood and CSF from patients with MS and controls for antibody reactivity to the epsilon toxin. Levels of epsilon toxin antibodies were 10 times higher in patients with MS, compared with controls. After examining stool samples, the study authors found the human commensal C. perfringens type A in approximately 50% of healthy controls, compared with 23% of patients with MS. C. perfringens epsilon toxin fits mechanistically with nascent MS lesion formation because these lesions are characterized by blood–brain barrier permeability and oligodendrocyte cell death in the absence of an adaptive immune infiltrate, said the researchers.
Chronic cerebrospinal venous insufficiency (CCSVI), which has been proposed as a contributor to multiple sclerosis (MS), occurs rarely in patients with MS and in controls, according to a study published online ahead of print October 8 in Lancet. Researchers performed an assessor-blinded, case-control, multicenter study of 79 people with MS, 55 unaffected siblings, and 43 unrelated healthy volunteers. Catheter venography criteria for CCSVI were positive for 2% of people with MS, 2% of siblings, and 3% of unrelated controls. Greater than 50% narrowing of any major vein was present in 74% of people with MS, 66% of siblings, and 70% of unrelated controls. The Zamboni ultrasound criteria are neither sensitive nor specific for narrowing on catheter venography, and the significance of venous narrowing to MS remains unknown, said the investigators.
Measures of α-synuclein deposition in cutaneous autonomic nerves may be a useful biomarker in patients with Parkinson’s disease, according to research published online ahead of print October 2 in Neurology. Investigators examined 20 patients with Parkinson’s disease and 14 age- and sex-matched control subjects. The researchers performed autonomic testing and skin biopsies at the distal leg, distal thigh, and proximal thigh for all participants. Deposition of α-synuclein and the density of intraepidermal, sudomotor, and pilomotor nerve fibers were measured. The investigators normalized α-synuclein deposition to nerve fiber density. Patients with Parkinson’s disease had greater α-synuclein deposition and higher α-synuclein ratios than controls within pilomotor nerves and sudomotor nerves, but not sensory nerves. Higher α-synuclein ratios correlated with Hoehn and Yahr scores, sympathetic adrenergic function, and parasympathetic function.
Depression may be an independent risk factor for Parkinson’s disease, according to research published October 22 in Neurology. Investigators conducted a retrospective study of 4,634 patients with depression and 18,544 matched controls who were selected from a national health insurance database. Patients were observed for a maximum of 10 years to determine the rates of new-onset Parkinson’s disease. Cox regression was used to identify the predictors of the disease. During the follow-up period, 66 patients with depression and 97 controls were diagnosed with Parkinson’s disease. After adjusting for age and sex, the researchers found that patients with depression were 3.24 times more likely to develop Parkinson’s disease, compared with the control patients. The investigators observed that age and difficult-to-treat depression are independent risk factors for Parkinson’s disease in patients with depression.
The levels of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) in red blood cells may have no association with age-associated cognitive decline, researchers reported in the October 22 issue of Neurology. The investigators conducted a retrospective cohort study of 2,157 women with normal cognition who were followed with annual cognitive testing for a median of 5.9 years. End points were composite cognitive function and performance in seven cognitive domains. After adjustment for demographic, clinical, and behavioral characteristics, the investigators found no significant cross-sectional cognitive differences between women in the high and low DHA and EPA tertiles at the first annual cognitive battery. In addition, no significant differences were found between the high and low DHA and EPA tertiles in the rate of cognitive change over time.
Common psychosocial stressors (eg, divorce, widowhood, work problems, and illness in a relative) may have severe and long-standing physiologic and psychologic consequences such as dementia, according to research published September 30 in BMJ Open. In a prospective longitudinal population study, clinicians performed psychiatric examinations for 800 women born in 1914, 1918, 1922, and 1930. Baseline examinations took place in 1968, and follow-up occurred in 1974, 1980, 1992, 2000, and 2005. During follow-up, 153 women developed dementia. The number of psychosocial stressors in 1968 was associated with higher incidence of dementia and Alzheimer’s disease (AD) between 1968 and 2005 in multivariate Cox regressions. The number of psychosocial stressors in 1968 was also associated with distress in 1968, 1974, 1980, 2000, and 2005 in multivariate logistic regressions.
Aggressive medical management may provide more benefit than percutaneous transluminal angioplasty and stenting (PTAS) for high-risk patients with atherosclerotic intracranial arterial stenosis, according to a study published online ahead of print October 26 in Lancet. Investigators randomized 451 patients with recent transient ischemic attack or stroke to aggressive medical management or aggressive medical management plus stenting with the Wingspan stent. The cumulative probability of stroke or death was smaller in the medical group vs the PTAS group. Beyond 30 days, 10% of patients in the medical group and 10% of patients in the stenting group had a primary end point. The absolute differences in the primary end point rates between the two groups were 7.1% at year 1, 6.5% at year 2 and 9.0% at year 3.
—Erik Greb
Senior Associate Editor
Ventricular Assist Device Therapy: A Roundtable Discussion
Topics
- Anticipated Time Horizon For VAD Support
- Incorporating VAD Implantation Earlier In Heart Failure Progression
- Support Structure For VAD Program Success
Faculty/Faculty Disclosures
John B. O’Connell, MD
Thoratec Corporation
Pleasanton, CA
Dr O’Connell discloses that at the time of the roundtable discussion he was a paid consultant for Thoratec Corporation. As of September 30, 2013, he is a full-time employee of Thoratec Corporation. Dr O’Connell also discloses that he is on the Data and Safety Monitoring Committee for Auven Therapeutics.
Walter Dembitsky, MD
Sharp Memorial Hospital
San Diego, CA
Dr Dembitsky discloses that he is a paid consultant and lecturer for, and has received research grants from, Thoratec Corporation.
Ranjit John, MD
University of Minnesota
Minneapolis, MI
Dr John discloses that he is a paid consultant for, and has received research grants from, Thoratec Corporation.
Jaap Lahpor, MD, PhD
University Medical Centre Utrecht
Utrecht, Netherlands
Dr Lahpor discloses that he is a paid consultant and speaker for Thoratec Corporation.
Jonathan D. Rich, MD
Northwestern University
Chicago, IL
Dr Rich discloses that he has received honoraria in the past from Thoratec Corporation.
Welcome to Audiocast #1 of 3. In this audiocast we will discuss the question: When you evaluate a patient for VAD therapy, what time horizon for support do you have in mind?
Welcome to Audiocast #2 of 3. In this audiocast we will answer the question: As patients are supported for longer periods of time, is trying to implant patients earlier in their heart failure progression becoming more important?
Welcome to Audiocast #3 of 3. In this audiocast we will ask the question: As more and more patients are supported for longer periods of time, what types of additional support structure will be required and how are we measuring success?
Topics
- Anticipated Time Horizon For VAD Support
- Incorporating VAD Implantation Earlier In Heart Failure Progression
- Support Structure For VAD Program Success
Faculty/Faculty Disclosures
John B. O’Connell, MD
Thoratec Corporation
Pleasanton, CA
Dr O’Connell discloses that at the time of the roundtable discussion he was a paid consultant for Thoratec Corporation. As of September 30, 2013, he is a full-time employee of Thoratec Corporation. Dr O’Connell also discloses that he is on the Data and Safety Monitoring Committee for Auven Therapeutics.
Walter Dembitsky, MD
Sharp Memorial Hospital
San Diego, CA
Dr Dembitsky discloses that he is a paid consultant and lecturer for, and has received research grants from, Thoratec Corporation.
Ranjit John, MD
University of Minnesota
Minneapolis, MI
Dr John discloses that he is a paid consultant for, and has received research grants from, Thoratec Corporation.
Jaap Lahpor, MD, PhD
University Medical Centre Utrecht
Utrecht, Netherlands
Dr Lahpor discloses that he is a paid consultant and speaker for Thoratec Corporation.
Jonathan D. Rich, MD
Northwestern University
Chicago, IL
Dr Rich discloses that he has received honoraria in the past from Thoratec Corporation.
Welcome to Audiocast #1 of 3. In this audiocast we will discuss the question: When you evaluate a patient for VAD therapy, what time horizon for support do you have in mind?
Welcome to Audiocast #2 of 3. In this audiocast we will answer the question: As patients are supported for longer periods of time, is trying to implant patients earlier in their heart failure progression becoming more important?
Welcome to Audiocast #3 of 3. In this audiocast we will ask the question: As more and more patients are supported for longer periods of time, what types of additional support structure will be required and how are we measuring success?
Topics
- Anticipated Time Horizon For VAD Support
- Incorporating VAD Implantation Earlier In Heart Failure Progression
- Support Structure For VAD Program Success
Faculty/Faculty Disclosures
John B. O’Connell, MD
Thoratec Corporation
Pleasanton, CA
Dr O’Connell discloses that at the time of the roundtable discussion he was a paid consultant for Thoratec Corporation. As of September 30, 2013, he is a full-time employee of Thoratec Corporation. Dr O’Connell also discloses that he is on the Data and Safety Monitoring Committee for Auven Therapeutics.
Walter Dembitsky, MD
Sharp Memorial Hospital
San Diego, CA
Dr Dembitsky discloses that he is a paid consultant and lecturer for, and has received research grants from, Thoratec Corporation.
Ranjit John, MD
University of Minnesota
Minneapolis, MI
Dr John discloses that he is a paid consultant for, and has received research grants from, Thoratec Corporation.
Jaap Lahpor, MD, PhD
University Medical Centre Utrecht
Utrecht, Netherlands
Dr Lahpor discloses that he is a paid consultant and speaker for Thoratec Corporation.
Jonathan D. Rich, MD
Northwestern University
Chicago, IL
Dr Rich discloses that he has received honoraria in the past from Thoratec Corporation.
Welcome to Audiocast #1 of 3. In this audiocast we will discuss the question: When you evaluate a patient for VAD therapy, what time horizon for support do you have in mind?
Welcome to Audiocast #2 of 3. In this audiocast we will answer the question: As patients are supported for longer periods of time, is trying to implant patients earlier in their heart failure progression becoming more important?
Welcome to Audiocast #3 of 3. In this audiocast we will ask the question: As more and more patients are supported for longer periods of time, what types of additional support structure will be required and how are we measuring success?