User login
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).
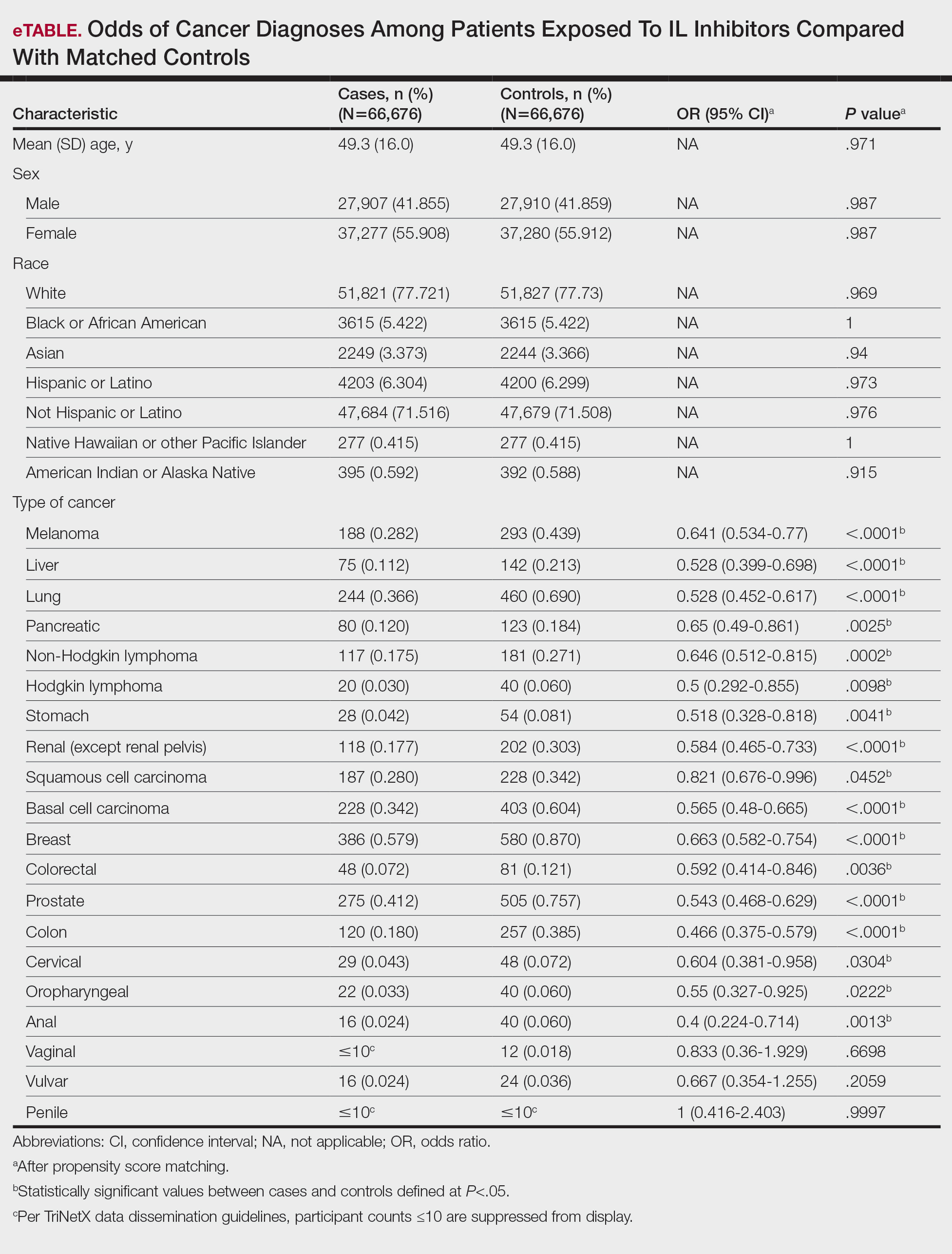
Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Practice Points
- Interleukin (IL) inhibitor therapy for psoriasis was associated with reduced odds of multiple malignancies in a large matched-cohort analysis.
- Potential mechanisms for reduced cancer risk include inhibition of tumor-promoting inflammation and restoration of antitumor immune surveillance, although further mechanistic and longitudinal studies are needed.
- These findings provide real-world evidence supporting the long-term malignancy safety of IL inhibitors, which may reassure clinicians and patients considering these agents for chronic disease management.
The Future of Psoriasis Care
The Future of Psoriasis Care
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
The Future of Psoriasis Care
The Future of Psoriasis Care
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.
Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.
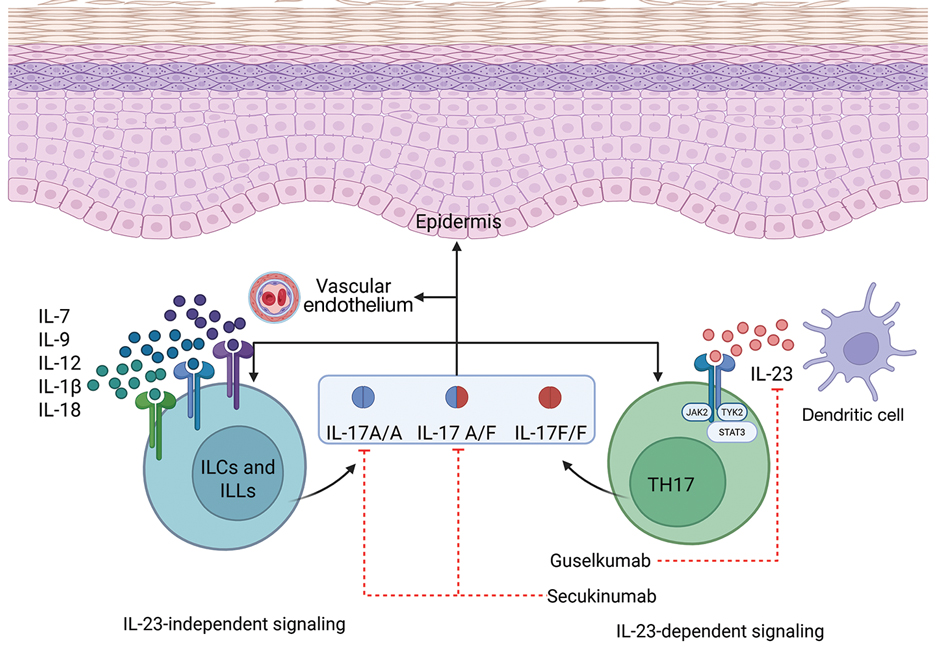
The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Practice Points
- Combination therapy with 2 complementary biologics for psoriasis may have synergistic effects.
- Washout periods likely are unnecessary when switching between guselkumab and secukinumab.
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).
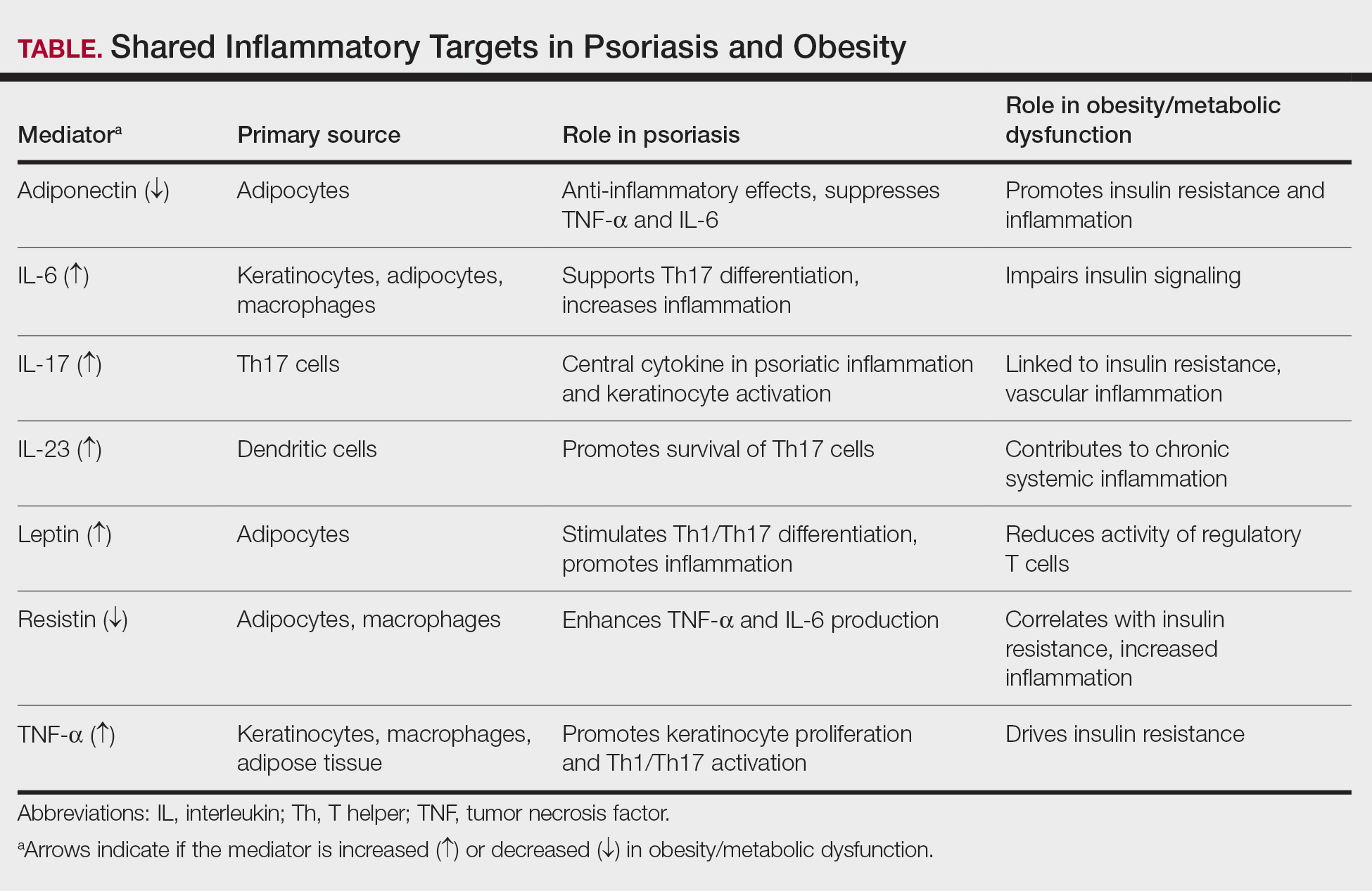
Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Practice Points
- Obesity is an independent risk factor for psoriasis onset and severity. Both conditions share overlapping inflammatory pathways that create a self-perpetuating cycle of metabolic and cutaneous dysfunction.
- Dermatologists play a key role in early detection of comorbidities, and patients with psoriasis and obesity should undergo regular screening for metabolic syndrome, liver disease, and psoriatic arthritis.
- Weight loss is a critical therapeutic intervention that may improve Psoriasis Area and Severity Index scores and restore therapeutic responsiveness.
Safety and Effectiveness of Nonsteroidal Tapinarof Cream 1% Added to Ongoing Biologic Therapy for Treatment of Moderate to Severe Plaque Psoriasis
Safety and Effectiveness of Nonsteroidal Tapinarof Cream 1% Added to Ongoing Biologic Therapy for Treatment of Moderate to Severe Plaque Psoriasis
The estimated prevalence of psoriasis in individuals older than 20 years in the United States has been reported at approximately 3%, or more than 7.5 million people.1 There currently is no cure for psoriasis, and available therapeutics, including phototherapy,2 topical therapies,3 systemic medications,4 and biologic agents,5 are focused only on controlling symptoms. The National Psoriasis Foundation defines an acceptable treatment response for plaque psoriasis as 3% or lower body surface area (BSA) involvement after 3 months of therapy, with a treat-to-target (TTT) goal of 1% or less BSA involvement.6
Cytokines are known to mediate psoriasis pathology, and biologic therapies target the signaling cascade of various cytokines. Biologics approved to treat moderate to severe plaque psoriasis include IgG monoclonal antibodies binding and inhibiting the activity of interleukin (IL)-17 (ixekizumab,7 secukinumab8), IL-23 (guselkumab,9 risankizumab,10 tildrakizumab11), and IL-12/23 (ustekinumab12). Despite targeting these cytokines, biologics may not sufficiently suppress the symptoms of psoriatic disease and their severity in all patients. Adding a topical treatment to biologic therapy can augment clinical response without increasing the incidence of adverse effects13-15 and may reduce the need to switch biologics due to ineffectiveness. Switching biologics likely would increase cost burden to the health care system and/or patient depending on their insurance plan and possibly introduce new safety and/or tolerability issues.16,17
In patients who do not adequately respond to biologics, better responses were reported when topical medications including halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17,18 were administered. In randomized or open-label, real-world studies, patients with psoriasis responded well when topical medications were added to a biologic, such as tildrakizumab combined with halcinonide ointment 0.1%,19 etanercept combined with topical clobetasol propionate foam,20 or adalimumab combined with calcipotriene/betamethasone dipropionate foam.21 No additional safety concerns were observed with the topical add-ons in any of these studies.
Tapinarof is an aryl hydrocarbon receptor agonist approved by the US Food and Drug Administration for topical treatment of plaque psoriasis in adults.22 It is a first-in-class small molecule with a novel mechanism of action that downregulates IL-17A and IL-17F and normalizes the skin barrier through expression of filaggrin, loricrin, and involucrin; it also has antioxidant activity.23 In the phase 3 PSOARING 1 and 2 trials, daily application of tapinarof cream was safe and efficacious in patients with plaque psoriasis,24,25 with a remittive (maintenance) effect of a median of approximately 4 months after discontinuation.25 In these 2 phase 3 studies, tapinarof significantly (P<0.01 at week 12) relieved itch, which was seen rapidly (P<0.05 at week 2),26 improved quality of life,27 and led to high patient satisfaction.27 When tapinarof cream was combined with deucravacitinib in a patient with severe plaque psoriasis, symptoms rapidly cleared, with a 75% decrease in disease severity after 4 weeks.28
The objective of this prospective, open-label, real-world, single-center study was to assess the effectiveness, safety, and remittive (or maintenance) effect of nonsteroidal tapinarof cream 1% added to ongoing biologic therapy in patients with plaque psoriasis who were not adequately responding to a biologic alone.
Methods
Study Design and Participants—This prospective, open-label, real-world, single-center study assessed the safety and effectiveness of
Eligible participants were otherwise healthy males and females aged 18 years and older with moderate to severe plaque psoriasis (BSA involvement ≥3%) who had been treated with a biologic for 24 weeks or more. Patients were recruited from the Psoriasis Treatment Center of New Jersey (East Windsor, New Jersey). Exclusion criteria were recent use of oral systemic therapies (within 4 weeks of baseline) or topical therapies (within 2 weeks) to treat psoriasis, recent use of UVB (within 2 weeks) or psoralen plus UVA (within 4 weeks) phototherapy, or use of any investigational drug within 4 weeks of baseline (or within 5 pharmacokinetic/pharmacodynamic half-lives, whichever was longer). Patients who were pregnant or breastfeeding or who had any known hypersensitivity to the excipients of tapinarof cream also were excluded from the study.
Eligible participants received tapinarof cream 1% once daily plus their ongoing biologic for 12 weeks, after which tapinarof was discontinued and the biologic was continued for an additional 4 weeks. A remittive (maintenance) effect was assessed at week 16.
Study Outcomes—Safety and efficacy were evaluated at baseline and weeks 2, 4, 8, 12, and 16. The primary end point was the proportion of patients who reached the TTT goal of 1% or less BSA involvement at week 12. Secondary end points included the proportion of patients with 1% or less BSA involvement at weeks 2, 4, 8, and 16; and PGA scores, composite PGA multiplied by mean percentage of BSA involvement (PGA×BSA), and PASI scores at baseline and weeks 2, 4, 8, 12, and 16. The patient-reported outcomes of Dermatology Life Quality Index (DLQI) and Worst Itch Numeric Rating Scale (WI-NRS) scores also were evaluated at baseline and weeks 2, 4, 8, 12, and 16. In patients who had disease involvement on the scalp or genital region at baseline, Psoriasis Scalp Severity Index (PSSI) and Static Physician’s Global Assessment of Genitalia scores, respectively, were assessed at baseline and weeks 2, 4, 8, 12, and 16. Safety was determined by the incidence, severity, and relatedness of adverse events (AEs) and serious AEs.
Statistical Analysis—Approximately 30 participants were planned for enrollment and recruited consecutively as they were identified during screening against inclusion and exclusion criteria. Changes from baseline in all outcomes were summarized descriptively. Missing data were not imputed. Given the sample size, no formal statistical analyses were conducted. Safety was summarized by descriptively collating AEs and serious AEs, including their frequency, severity, and treatment relatedness.
Results
Thirty participants were enrolled in the study, and 20 fully completed the study. Nine discontinued treatment before week 12 (6 were lost to follow-up, 2 were terminated early by the investigators, and 1 voluntarily withdrew); 1 additional participant was lost to follow-up after week 12. Patients were predominantly male (20/30 [66.7%]) and White (21/30 [70.0%]); the mean age of all participants was 55.4 years, and the mean (SD) duration of psoriasis was 21.4 (15.0) years (Table 1). The mean baseline percentage of BSA involvement and mean baseline PGA, PASI, and DLQI scores are shown in Table 1. Most (19/30 [63.3%]) patients received biologics that inhibited IL-23 activity (guselkumab, risankizumab, tildrakizumab), approximately one-third (9/30 [30.0%]) received biologics that inhibited IL-17 activity (ixekizumab, secukinumab), and 2 (6.7%) received biologics that inhibited IL-12/IL-23 activity (ustekinumab)(Table 1).

For the primary end point, 52.4% (11/21) of patients reached the TTT goal (BSA involvement ≤1% after 12 weeks of treatment with tapinarof cream added to a prescribed biologic). The proportion of patients reaching the TTT goal increased over time with the combined treatment (eFigure 1). Additionally, the mean percentage of BSA involvement (eFigure 2) as well as the mean values for PGA (eFigure 3) and PGA×BSA decreased over time. The mean percentage of BSA involvement was 5.0% at baseline and dropped to 2.0% by week 12. Similar reductions were observed for PGA and PGA×BSA scores at week 12.
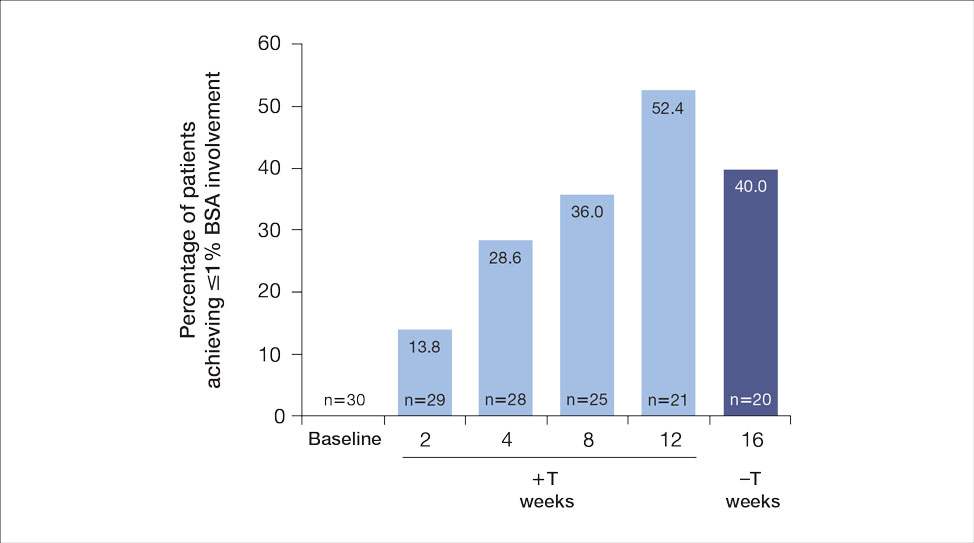
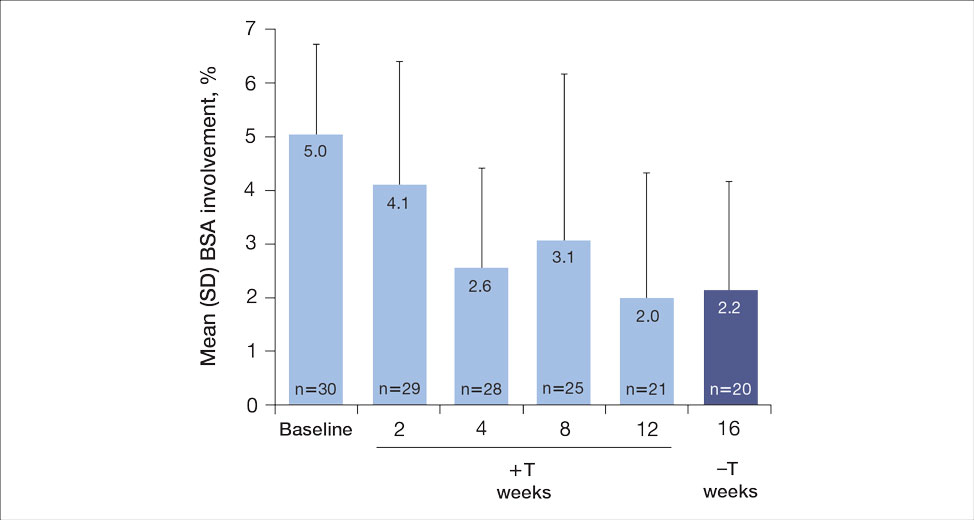
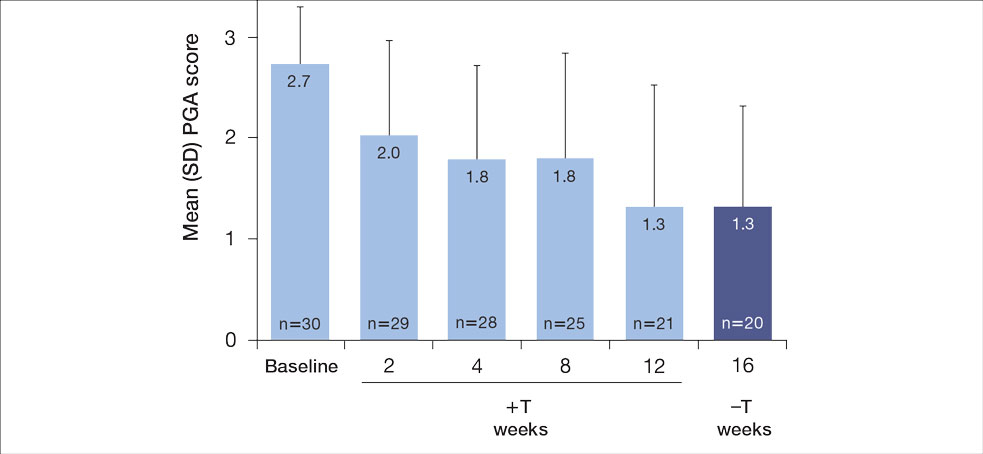
After discontinuing tapinarof cream at week 12 and receiving only the biologic for 4 weeks, the proportion of patients maintaining 1% or less BSA involvement fell to 40.0% (8/20) at week 16, which was closer to that observed at week 8 (36% [9/25]) than at week 12 (52.4% [11/21])(eFigure 1).
The mean PASI score was 5.5 at baseline, then decreased over time when tapinarof cream was combined with a biologic (eFigure 4), falling to 3.1 by week 2 and 1.6 by week 12; it was maintained at 1.7 at week 16. Nine (30.0%) participants had psoriasis on the scalp at baseline with a mean PSSI score of 2.6, which decreased to 0.83 by week 2. By week 12, the mean PSSI score remained stable at 0.95 in the 2 (9.5%) participants who still had scalp involvement. The mean PSSI score increased slightly to 1.45 after patients received only the biologic for 4 weeks. At baseline, 3 (10.0%) patients had genital involvement (mean Static Physician’s Global Assessment of Genitalia score, 0.27). Symptoms resolved in 2 (66.7%) of these patients at week 2 and stayed consistent until week 16; the third patient withdrew at week 2.
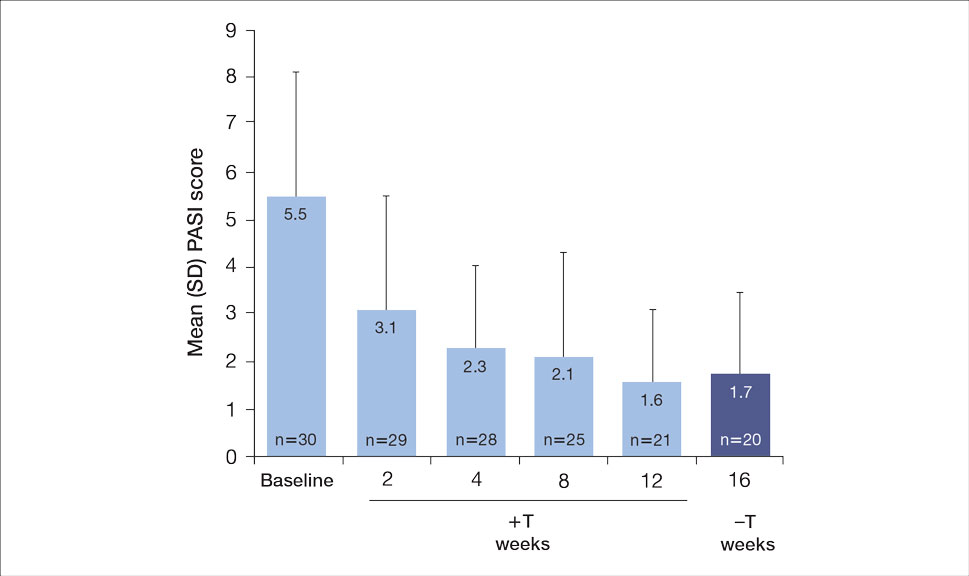
Both DLQI and WI-NRS scores decreased with use of tapinarof cream added to a biologic up to week 12 (eFigures 5 and 6). Mean DLQI scores were 5.3 at baseline and 3.1 at week 12. At week 16, the mean DLQI score remained stable at 2.8. Mean WI-NRS scores decreased from 4.0 at baseline to 2.7 at week 12 with the therapy combination; at week 16, the mean WI-NRS score fell further to 1.8.
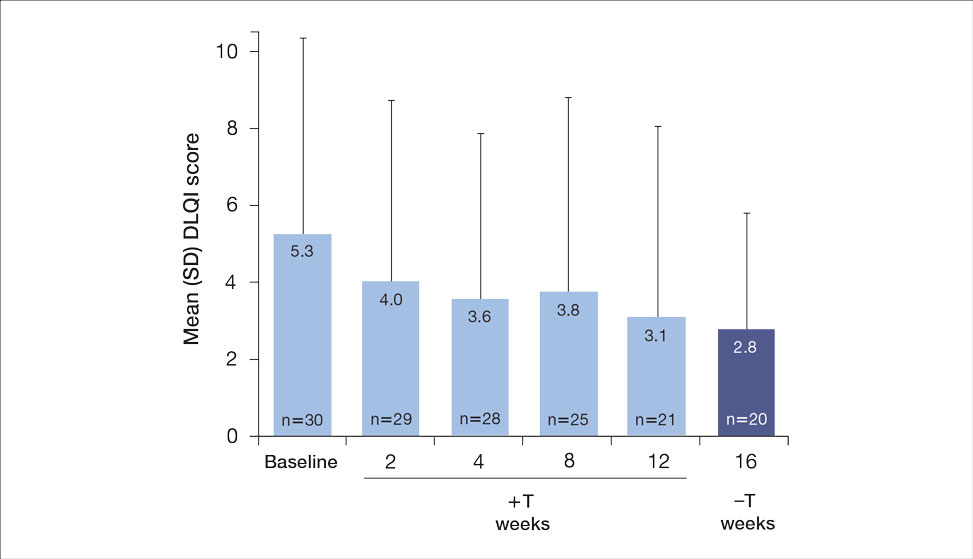

A total of 6 AEs were reported in 5 (16.7%) patients (Table 2). The majority (4/6 [67.0%]) of AEs were considered mild. Two reported cases of COVID-19 were both considered mild and unrelated. Mild folliculitis and moderate worsening of psoriasis in 2 (6.7%) different patients were the only AEs considered related to treatment. No serious AEs were reported, and no patient withdrew from the study due to an AE.
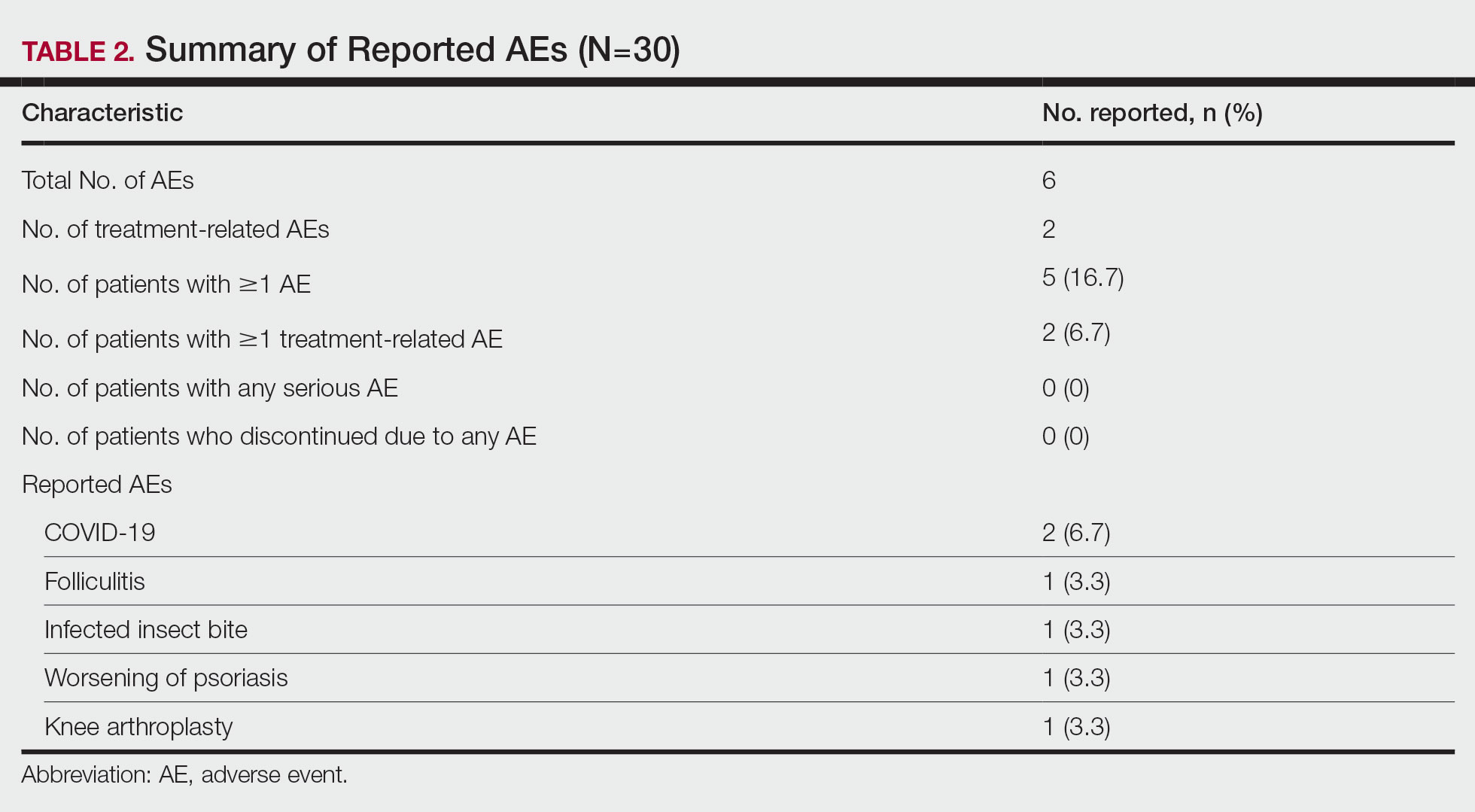
Comment
Disease activity improvements we observed with the nonsteroidal tapinarof cream were consistent with those reported when topical steroidal therapies were given to patients responding poorly to their current biologic. Our primary end point (proportion of patients with BSA involvement ≤1% after 12 weeks) showed that half (52% [11/21]) of patients whose BSA involvement was 3% or greater with a biologic for 24 weeks or more reached the TTT goal after 12 weeks of tapinarof-biologic treatment. Other studies of halobetasol propionate–tazarotene lotion16 and calcipotriene/betamethasone dipropionate foam17,18 added to the current biologic of poor responders found 60% to 68% of patients had reductions in their percentage BSA to 1% or lower at 12 to 16 weeks of treatment. Randomized studies showed etanercept plus topical clobetasol propionate foam20 or adalimumab plus calcipotriene/betamethasone dipropionate foam21 similarly enhanced treatment effects vs biologic alone.
A phase 3 PSOARING trial demonstrated benefit from treatment with tapinarof alone, with a remittive effect of approximately 4 months after discontinuation.25 Our data are consistent with these findings, with 40% (8/20) of patients demonstrating a remittive effect 4 weeks after discontinuing tapinarof while receiving a biologic. A similar maintenance effect was reported in another study in 50% (9/18) of patients treated with a biologic plus halobetasol propionate–tazarotene lotion.16 Additionally, when halcinonide ointment was given to patients receiving tildrakizumab, mean percentage of BSA involvement, PGA scores, PGA×BSA, and DLQI scores improved and were maintained 4 weeks after halcinonide ointment was stopped.19 Thus, topical therapy can augment and extend a biologic’s effect for up to 4 weeks.
In our study, tapinarof cream added to a biologic had a good safety and tolerability profile. Few AEs were recorded, with most being mild in nature, and no serious AEs or discontinuations due to AEs were reported. Only 1 case of mild folliculitis and 1 case of moderate worsening of psoriasis were considered treatment related. Further, no unexpected or new safety signals with the tapinarof-biologic combination were observed compared with tapinarof alone.27Prior studies have found that supplementing a biologic with topical therapy can reduce the probability of patients switching to another biologic.16,19 We previously found that adding halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17 to a biologic helped reduce the probability of switching biologics from 88% to 90% at baseline to 12% to 24% after 12 weeks of combined therapy. Such combinations also could prevent a less responsive patient from being prescribed a higher biologic dose.19 These are important research findings, as patients—even when not responding well to their current biologic—are more likely to be tolerating that biologic well, and switching to a new biologic may introduce new safety or tolerability concerns. Thus, by enhancing the effect of a biologic with a topical therapy, one can avoid increasing the dose of the current biologic or switching to a new biologic, either of which may increase safety and/or tolerability risks. Switching biologics also has increased cost implications to the health care system and/or the patient. When comparing the cost of adding halobetasol propionate–tazarotene lotion to a biologic compared with switching to another biologic, the cost was 1.2 to 2.9 times higher to switch, depending on the biologic, compared with a smaller incremental cost increase to add a topical to the current biologic.16 Similar observations were reported with calcipotriene/betamethasone dipropionate foam plus a biologic.17 Although we did not evaluate biologic switching here, we anticipate a similar clinical scenario with a tapinarof-biologic combination.
Limitations of our study included the open-label design, lack of a control arm, and the relatively small study population; however, for studies investigating the safety and effectiveness of a treatment in a real-world setting, these limitations are common and are not unexpected. Our results also are consistent with the overall improvement seen in other studies16-21 examining the effects of adding a topical to a biologic. Future research is warranted to investigate a longer remittive effect and potential health care system and patient cost savings without having to switch biologics due to lack of effectiveness.
Conclusion
This study demonstrated that adjunctive use of nonsteroidal tapinarof cream 1% may enhance a biologic treatment effect in patients with moderate to severe plaque psoriasis, providing an adequate response for many patients who were not responding well to a biologic alone. Clinical outcomes improved with the tapinarof-biologic combination, and a remittive effect was noted 4 weeks after tapinarof discontinuation without any new safety signals. Adding tapinarof cream to a biologic also may prevent the need to switch biologics when patients do not sufficiently respond, preserving the safety and cost associated with a patient’s current biologic.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804. doi:10.1016/j.jaad.2019.04.042
- Elmets CA, Korman NJ, Prater EF, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with topical therapy and alternative medicine modalities for psoriasis severity measures. J Am Acad Dermatol. 2021;84:432-470. doi:10.1016/j.jaad.2020.07.087
- Menter A, Gelfand JM, Connor C, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management of psoriasis with systemic nonbiological therapies. J Am Acad Dermatol. 2020;82:1445-1486. doi:10.1016/j.jaad.2020.02.044
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072. doi:10.1016/j.jaad.2018.11.057
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis.J Am Acad Dermatol. 2017;76:290-298. doi:10.1016/j.jaad.2016.10.017
- Taltz. Prescribing information. Eli Lilly and Company; 2024.
- Cosentyx. Prescribing information. Novartis Pharmaceuticals Corporation; 2023.
- Tremfya. Prescribing information. Janssen Biotech, Inc; 2023.
- Skyrizi. Prescribing information. AbbVie Inc; 2024.
- Ilumya. Prescribing information. Sun Pharmaceutical Industries, Inc; 2020.
- Stelara. Prescribing information. Janssen Biotech, Inc; 2022.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Jensen JD, Delcambre MR, Nguyen G, et al. Biologic therapy with or without topical treatment in psoriasis: what does the current evidence say? Am J Clin Dermatol. 2014;15:379-385. doi:10.1007/s40257-014-0089-1
- Gustafson CJ, Watkins C, Hix E, et al. Combination therapy in psoriasis: an evidence-based review. Am J Clin Dermatol. 2013;14:9-25. doi:10.1007/s40257-012-0003-7
- Bagel J, Novak K, Nelson E. Adjunctive use of halobetasol propionate-tazarotene in biologic-experienced patients with psoriasis. Cutis. 2022;109:103-109. doi:10.12788/cutis.0451
- Bagel J, Nelson E, Zapata J, et al. Adjunctive use of calcipotriene/betamethasone dipropionate foam in a real-world setting curtails the cost of biologics without reducing efficacy in psoriasis. Dermatol Ther (Heidelb). 2020;10:1383-1396. doi:10.1007/s13555-020-00454-z
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:611-616.
- Bagel J, Novak K, Nelson E. Tildrakizumab in combination with topical halcinonide 0.1% ointment for treating moderate to severe plaque psoriasis. J Drugs Dermatol. 2023;22:766-772. doi:10.36849/jdd.6830
- Lebwohl MG, Kircik L, Callis Duffin K, et al. A randomized study to evaluate the efficacy and safety of adding topical therapy to etanercept in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2013;69:385-392. doi:10.1016/j.jaad.2013.03.031
- Thaci D, Ortonne JP, Chimenti S, et al. A phase IIIb, multicentre, randomized, double-blind, vehicle-controlled study of the efficacy and safety of adalimumab with and without calcipotriol/betamethasone topical treatment in patients with moderate to severe psoriasis: the BELIEVE study. Br J Dermatol. 2010;163:402-411. doi:10.1111/j.1365-2133.2010.09791.x
- Vtama. Prescribing information. Dermavant Sciences, Inc; 2022.
- Bobonich M, Gorelick J, Aldredge L, et al. Tapinarof, a novel, first-in-class, topical therapeutic aryl hydrocarbon receptor agonist for the management of psoriasis. J Drugs Dermatol. 2023;22:779-784. doi:10.36849/jdd.7317
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Strober B, Stein Gold L, Bissonnette R, et al. One-year safety and efficacy of tapinarof cream for the treatment of plaque psoriasis: results from the PSOARING 3 trial. J Am Acad Dermatol. 2022;87:800-806. doi:10.1016/j.jaad.2022.06.1171
- Kircik L, Zirwas M, Kwatra SG, et al. Rapid improvements in itch with tapinarof cream 1% once daily in two phase 3 trials in adults with mild to severe plaque psoriasis. Dermatol Ther (Heidelb). 2024;14:201-211. doi:10.1007/s13555-023-01068-x
- Bagel J, Gold LS, Del Rosso J, et al. Tapinarof cream 1% once daily for the treatment of plaque psoriasis: patient-reported outcomes from the PSOARING 3 trial. J Am Acad Dermatol. 2023;89:936-944. doi:10.1016/j.jaad.2023.04.061
- Abdin R, Kircik L, Issa NT. First use of combination oral deucravacitinib with tapinarof cream for treatment of severe plaque psoriasis. J Drugs Dermatol. 2024;23:192-194. doi:10.36849/jdd.8091
The estimated prevalence of psoriasis in individuals older than 20 years in the United States has been reported at approximately 3%, or more than 7.5 million people.1 There currently is no cure for psoriasis, and available therapeutics, including phototherapy,2 topical therapies,3 systemic medications,4 and biologic agents,5 are focused only on controlling symptoms. The National Psoriasis Foundation defines an acceptable treatment response for plaque psoriasis as 3% or lower body surface area (BSA) involvement after 3 months of therapy, with a treat-to-target (TTT) goal of 1% or less BSA involvement.6
Cytokines are known to mediate psoriasis pathology, and biologic therapies target the signaling cascade of various cytokines. Biologics approved to treat moderate to severe plaque psoriasis include IgG monoclonal antibodies binding and inhibiting the activity of interleukin (IL)-17 (ixekizumab,7 secukinumab8), IL-23 (guselkumab,9 risankizumab,10 tildrakizumab11), and IL-12/23 (ustekinumab12). Despite targeting these cytokines, biologics may not sufficiently suppress the symptoms of psoriatic disease and their severity in all patients. Adding a topical treatment to biologic therapy can augment clinical response without increasing the incidence of adverse effects13-15 and may reduce the need to switch biologics due to ineffectiveness. Switching biologics likely would increase cost burden to the health care system and/or patient depending on their insurance plan and possibly introduce new safety and/or tolerability issues.16,17
In patients who do not adequately respond to biologics, better responses were reported when topical medications including halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17,18 were administered. In randomized or open-label, real-world studies, patients with psoriasis responded well when topical medications were added to a biologic, such as tildrakizumab combined with halcinonide ointment 0.1%,19 etanercept combined with topical clobetasol propionate foam,20 or adalimumab combined with calcipotriene/betamethasone dipropionate foam.21 No additional safety concerns were observed with the topical add-ons in any of these studies.
Tapinarof is an aryl hydrocarbon receptor agonist approved by the US Food and Drug Administration for topical treatment of plaque psoriasis in adults.22 It is a first-in-class small molecule with a novel mechanism of action that downregulates IL-17A and IL-17F and normalizes the skin barrier through expression of filaggrin, loricrin, and involucrin; it also has antioxidant activity.23 In the phase 3 PSOARING 1 and 2 trials, daily application of tapinarof cream was safe and efficacious in patients with plaque psoriasis,24,25 with a remittive (maintenance) effect of a median of approximately 4 months after discontinuation.25 In these 2 phase 3 studies, tapinarof significantly (P<0.01 at week 12) relieved itch, which was seen rapidly (P<0.05 at week 2),26 improved quality of life,27 and led to high patient satisfaction.27 When tapinarof cream was combined with deucravacitinib in a patient with severe plaque psoriasis, symptoms rapidly cleared, with a 75% decrease in disease severity after 4 weeks.28
The objective of this prospective, open-label, real-world, single-center study was to assess the effectiveness, safety, and remittive (or maintenance) effect of nonsteroidal tapinarof cream 1% added to ongoing biologic therapy in patients with plaque psoriasis who were not adequately responding to a biologic alone.
Methods
Study Design and Participants—This prospective, open-label, real-world, single-center study assessed the safety and effectiveness of
Eligible participants were otherwise healthy males and females aged 18 years and older with moderate to severe plaque psoriasis (BSA involvement ≥3%) who had been treated with a biologic for 24 weeks or more. Patients were recruited from the Psoriasis Treatment Center of New Jersey (East Windsor, New Jersey). Exclusion criteria were recent use of oral systemic therapies (within 4 weeks of baseline) or topical therapies (within 2 weeks) to treat psoriasis, recent use of UVB (within 2 weeks) or psoralen plus UVA (within 4 weeks) phototherapy, or use of any investigational drug within 4 weeks of baseline (or within 5 pharmacokinetic/pharmacodynamic half-lives, whichever was longer). Patients who were pregnant or breastfeeding or who had any known hypersensitivity to the excipients of tapinarof cream also were excluded from the study.
Eligible participants received tapinarof cream 1% once daily plus their ongoing biologic for 12 weeks, after which tapinarof was discontinued and the biologic was continued for an additional 4 weeks. A remittive (maintenance) effect was assessed at week 16.
Study Outcomes—Safety and efficacy were evaluated at baseline and weeks 2, 4, 8, 12, and 16. The primary end point was the proportion of patients who reached the TTT goal of 1% or less BSA involvement at week 12. Secondary end points included the proportion of patients with 1% or less BSA involvement at weeks 2, 4, 8, and 16; and PGA scores, composite PGA multiplied by mean percentage of BSA involvement (PGA×BSA), and PASI scores at baseline and weeks 2, 4, 8, 12, and 16. The patient-reported outcomes of Dermatology Life Quality Index (DLQI) and Worst Itch Numeric Rating Scale (WI-NRS) scores also were evaluated at baseline and weeks 2, 4, 8, 12, and 16. In patients who had disease involvement on the scalp or genital region at baseline, Psoriasis Scalp Severity Index (PSSI) and Static Physician’s Global Assessment of Genitalia scores, respectively, were assessed at baseline and weeks 2, 4, 8, 12, and 16. Safety was determined by the incidence, severity, and relatedness of adverse events (AEs) and serious AEs.
Statistical Analysis—Approximately 30 participants were planned for enrollment and recruited consecutively as they were identified during screening against inclusion and exclusion criteria. Changes from baseline in all outcomes were summarized descriptively. Missing data were not imputed. Given the sample size, no formal statistical analyses were conducted. Safety was summarized by descriptively collating AEs and serious AEs, including their frequency, severity, and treatment relatedness.
Results
Thirty participants were enrolled in the study, and 20 fully completed the study. Nine discontinued treatment before week 12 (6 were lost to follow-up, 2 were terminated early by the investigators, and 1 voluntarily withdrew); 1 additional participant was lost to follow-up after week 12. Patients were predominantly male (20/30 [66.7%]) and White (21/30 [70.0%]); the mean age of all participants was 55.4 years, and the mean (SD) duration of psoriasis was 21.4 (15.0) years (Table 1). The mean baseline percentage of BSA involvement and mean baseline PGA, PASI, and DLQI scores are shown in Table 1. Most (19/30 [63.3%]) patients received biologics that inhibited IL-23 activity (guselkumab, risankizumab, tildrakizumab), approximately one-third (9/30 [30.0%]) received biologics that inhibited IL-17 activity (ixekizumab, secukinumab), and 2 (6.7%) received biologics that inhibited IL-12/IL-23 activity (ustekinumab)(Table 1).

For the primary end point, 52.4% (11/21) of patients reached the TTT goal (BSA involvement ≤1% after 12 weeks of treatment with tapinarof cream added to a prescribed biologic). The proportion of patients reaching the TTT goal increased over time with the combined treatment (eFigure 1). Additionally, the mean percentage of BSA involvement (eFigure 2) as well as the mean values for PGA (eFigure 3) and PGA×BSA decreased over time. The mean percentage of BSA involvement was 5.0% at baseline and dropped to 2.0% by week 12. Similar reductions were observed for PGA and PGA×BSA scores at week 12.



After discontinuing tapinarof cream at week 12 and receiving only the biologic for 4 weeks, the proportion of patients maintaining 1% or less BSA involvement fell to 40.0% (8/20) at week 16, which was closer to that observed at week 8 (36% [9/25]) than at week 12 (52.4% [11/21])(eFigure 1).
The mean PASI score was 5.5 at baseline, then decreased over time when tapinarof cream was combined with a biologic (eFigure 4), falling to 3.1 by week 2 and 1.6 by week 12; it was maintained at 1.7 at week 16. Nine (30.0%) participants had psoriasis on the scalp at baseline with a mean PSSI score of 2.6, which decreased to 0.83 by week 2. By week 12, the mean PSSI score remained stable at 0.95 in the 2 (9.5%) participants who still had scalp involvement. The mean PSSI score increased slightly to 1.45 after patients received only the biologic for 4 weeks. At baseline, 3 (10.0%) patients had genital involvement (mean Static Physician’s Global Assessment of Genitalia score, 0.27). Symptoms resolved in 2 (66.7%) of these patients at week 2 and stayed consistent until week 16; the third patient withdrew at week 2.

Both DLQI and WI-NRS scores decreased with use of tapinarof cream added to a biologic up to week 12 (eFigures 5 and 6). Mean DLQI scores were 5.3 at baseline and 3.1 at week 12. At week 16, the mean DLQI score remained stable at 2.8. Mean WI-NRS scores decreased from 4.0 at baseline to 2.7 at week 12 with the therapy combination; at week 16, the mean WI-NRS score fell further to 1.8.


A total of 6 AEs were reported in 5 (16.7%) patients (Table 2). The majority (4/6 [67.0%]) of AEs were considered mild. Two reported cases of COVID-19 were both considered mild and unrelated. Mild folliculitis and moderate worsening of psoriasis in 2 (6.7%) different patients were the only AEs considered related to treatment. No serious AEs were reported, and no patient withdrew from the study due to an AE.

Comment
Disease activity improvements we observed with the nonsteroidal tapinarof cream were consistent with those reported when topical steroidal therapies were given to patients responding poorly to their current biologic. Our primary end point (proportion of patients with BSA involvement ≤1% after 12 weeks) showed that half (52% [11/21]) of patients whose BSA involvement was 3% or greater with a biologic for 24 weeks or more reached the TTT goal after 12 weeks of tapinarof-biologic treatment. Other studies of halobetasol propionate–tazarotene lotion16 and calcipotriene/betamethasone dipropionate foam17,18 added to the current biologic of poor responders found 60% to 68% of patients had reductions in their percentage BSA to 1% or lower at 12 to 16 weeks of treatment. Randomized studies showed etanercept plus topical clobetasol propionate foam20 or adalimumab plus calcipotriene/betamethasone dipropionate foam21 similarly enhanced treatment effects vs biologic alone.
A phase 3 PSOARING trial demonstrated benefit from treatment with tapinarof alone, with a remittive effect of approximately 4 months after discontinuation.25 Our data are consistent with these findings, with 40% (8/20) of patients demonstrating a remittive effect 4 weeks after discontinuing tapinarof while receiving a biologic. A similar maintenance effect was reported in another study in 50% (9/18) of patients treated with a biologic plus halobetasol propionate–tazarotene lotion.16 Additionally, when halcinonide ointment was given to patients receiving tildrakizumab, mean percentage of BSA involvement, PGA scores, PGA×BSA, and DLQI scores improved and were maintained 4 weeks after halcinonide ointment was stopped.19 Thus, topical therapy can augment and extend a biologic’s effect for up to 4 weeks.
In our study, tapinarof cream added to a biologic had a good safety and tolerability profile. Few AEs were recorded, with most being mild in nature, and no serious AEs or discontinuations due to AEs were reported. Only 1 case of mild folliculitis and 1 case of moderate worsening of psoriasis were considered treatment related. Further, no unexpected or new safety signals with the tapinarof-biologic combination were observed compared with tapinarof alone.27Prior studies have found that supplementing a biologic with topical therapy can reduce the probability of patients switching to another biologic.16,19 We previously found that adding halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17 to a biologic helped reduce the probability of switching biologics from 88% to 90% at baseline to 12% to 24% after 12 weeks of combined therapy. Such combinations also could prevent a less responsive patient from being prescribed a higher biologic dose.19 These are important research findings, as patients—even when not responding well to their current biologic—are more likely to be tolerating that biologic well, and switching to a new biologic may introduce new safety or tolerability concerns. Thus, by enhancing the effect of a biologic with a topical therapy, one can avoid increasing the dose of the current biologic or switching to a new biologic, either of which may increase safety and/or tolerability risks. Switching biologics also has increased cost implications to the health care system and/or the patient. When comparing the cost of adding halobetasol propionate–tazarotene lotion to a biologic compared with switching to another biologic, the cost was 1.2 to 2.9 times higher to switch, depending on the biologic, compared with a smaller incremental cost increase to add a topical to the current biologic.16 Similar observations were reported with calcipotriene/betamethasone dipropionate foam plus a biologic.17 Although we did not evaluate biologic switching here, we anticipate a similar clinical scenario with a tapinarof-biologic combination.
Limitations of our study included the open-label design, lack of a control arm, and the relatively small study population; however, for studies investigating the safety and effectiveness of a treatment in a real-world setting, these limitations are common and are not unexpected. Our results also are consistent with the overall improvement seen in other studies16-21 examining the effects of adding a topical to a biologic. Future research is warranted to investigate a longer remittive effect and potential health care system and patient cost savings without having to switch biologics due to lack of effectiveness.
Conclusion
This study demonstrated that adjunctive use of nonsteroidal tapinarof cream 1% may enhance a biologic treatment effect in patients with moderate to severe plaque psoriasis, providing an adequate response for many patients who were not responding well to a biologic alone. Clinical outcomes improved with the tapinarof-biologic combination, and a remittive effect was noted 4 weeks after tapinarof discontinuation without any new safety signals. Adding tapinarof cream to a biologic also may prevent the need to switch biologics when patients do not sufficiently respond, preserving the safety and cost associated with a patient’s current biologic.
The estimated prevalence of psoriasis in individuals older than 20 years in the United States has been reported at approximately 3%, or more than 7.5 million people.1 There currently is no cure for psoriasis, and available therapeutics, including phototherapy,2 topical therapies,3 systemic medications,4 and biologic agents,5 are focused only on controlling symptoms. The National Psoriasis Foundation defines an acceptable treatment response for plaque psoriasis as 3% or lower body surface area (BSA) involvement after 3 months of therapy, with a treat-to-target (TTT) goal of 1% or less BSA involvement.6
Cytokines are known to mediate psoriasis pathology, and biologic therapies target the signaling cascade of various cytokines. Biologics approved to treat moderate to severe plaque psoriasis include IgG monoclonal antibodies binding and inhibiting the activity of interleukin (IL)-17 (ixekizumab,7 secukinumab8), IL-23 (guselkumab,9 risankizumab,10 tildrakizumab11), and IL-12/23 (ustekinumab12). Despite targeting these cytokines, biologics may not sufficiently suppress the symptoms of psoriatic disease and their severity in all patients. Adding a topical treatment to biologic therapy can augment clinical response without increasing the incidence of adverse effects13-15 and may reduce the need to switch biologics due to ineffectiveness. Switching biologics likely would increase cost burden to the health care system and/or patient depending on their insurance plan and possibly introduce new safety and/or tolerability issues.16,17
In patients who do not adequately respond to biologics, better responses were reported when topical medications including halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17,18 were administered. In randomized or open-label, real-world studies, patients with psoriasis responded well when topical medications were added to a biologic, such as tildrakizumab combined with halcinonide ointment 0.1%,19 etanercept combined with topical clobetasol propionate foam,20 or adalimumab combined with calcipotriene/betamethasone dipropionate foam.21 No additional safety concerns were observed with the topical add-ons in any of these studies.
Tapinarof is an aryl hydrocarbon receptor agonist approved by the US Food and Drug Administration for topical treatment of plaque psoriasis in adults.22 It is a first-in-class small molecule with a novel mechanism of action that downregulates IL-17A and IL-17F and normalizes the skin barrier through expression of filaggrin, loricrin, and involucrin; it also has antioxidant activity.23 In the phase 3 PSOARING 1 and 2 trials, daily application of tapinarof cream was safe and efficacious in patients with plaque psoriasis,24,25 with a remittive (maintenance) effect of a median of approximately 4 months after discontinuation.25 In these 2 phase 3 studies, tapinarof significantly (P<0.01 at week 12) relieved itch, which was seen rapidly (P<0.05 at week 2),26 improved quality of life,27 and led to high patient satisfaction.27 When tapinarof cream was combined with deucravacitinib in a patient with severe plaque psoriasis, symptoms rapidly cleared, with a 75% decrease in disease severity after 4 weeks.28
The objective of this prospective, open-label, real-world, single-center study was to assess the effectiveness, safety, and remittive (or maintenance) effect of nonsteroidal tapinarof cream 1% added to ongoing biologic therapy in patients with plaque psoriasis who were not adequately responding to a biologic alone.
Methods
Study Design and Participants—This prospective, open-label, real-world, single-center study assessed the safety and effectiveness of
Eligible participants were otherwise healthy males and females aged 18 years and older with moderate to severe plaque psoriasis (BSA involvement ≥3%) who had been treated with a biologic for 24 weeks or more. Patients were recruited from the Psoriasis Treatment Center of New Jersey (East Windsor, New Jersey). Exclusion criteria were recent use of oral systemic therapies (within 4 weeks of baseline) or topical therapies (within 2 weeks) to treat psoriasis, recent use of UVB (within 2 weeks) or psoralen plus UVA (within 4 weeks) phototherapy, or use of any investigational drug within 4 weeks of baseline (or within 5 pharmacokinetic/pharmacodynamic half-lives, whichever was longer). Patients who were pregnant or breastfeeding or who had any known hypersensitivity to the excipients of tapinarof cream also were excluded from the study.
Eligible participants received tapinarof cream 1% once daily plus their ongoing biologic for 12 weeks, after which tapinarof was discontinued and the biologic was continued for an additional 4 weeks. A remittive (maintenance) effect was assessed at week 16.
Study Outcomes—Safety and efficacy were evaluated at baseline and weeks 2, 4, 8, 12, and 16. The primary end point was the proportion of patients who reached the TTT goal of 1% or less BSA involvement at week 12. Secondary end points included the proportion of patients with 1% or less BSA involvement at weeks 2, 4, 8, and 16; and PGA scores, composite PGA multiplied by mean percentage of BSA involvement (PGA×BSA), and PASI scores at baseline and weeks 2, 4, 8, 12, and 16. The patient-reported outcomes of Dermatology Life Quality Index (DLQI) and Worst Itch Numeric Rating Scale (WI-NRS) scores also were evaluated at baseline and weeks 2, 4, 8, 12, and 16. In patients who had disease involvement on the scalp or genital region at baseline, Psoriasis Scalp Severity Index (PSSI) and Static Physician’s Global Assessment of Genitalia scores, respectively, were assessed at baseline and weeks 2, 4, 8, 12, and 16. Safety was determined by the incidence, severity, and relatedness of adverse events (AEs) and serious AEs.
Statistical Analysis—Approximately 30 participants were planned for enrollment and recruited consecutively as they were identified during screening against inclusion and exclusion criteria. Changes from baseline in all outcomes were summarized descriptively. Missing data were not imputed. Given the sample size, no formal statistical analyses were conducted. Safety was summarized by descriptively collating AEs and serious AEs, including their frequency, severity, and treatment relatedness.
Results
Thirty participants were enrolled in the study, and 20 fully completed the study. Nine discontinued treatment before week 12 (6 were lost to follow-up, 2 were terminated early by the investigators, and 1 voluntarily withdrew); 1 additional participant was lost to follow-up after week 12. Patients were predominantly male (20/30 [66.7%]) and White (21/30 [70.0%]); the mean age of all participants was 55.4 years, and the mean (SD) duration of psoriasis was 21.4 (15.0) years (Table 1). The mean baseline percentage of BSA involvement and mean baseline PGA, PASI, and DLQI scores are shown in Table 1. Most (19/30 [63.3%]) patients received biologics that inhibited IL-23 activity (guselkumab, risankizumab, tildrakizumab), approximately one-third (9/30 [30.0%]) received biologics that inhibited IL-17 activity (ixekizumab, secukinumab), and 2 (6.7%) received biologics that inhibited IL-12/IL-23 activity (ustekinumab)(Table 1).

For the primary end point, 52.4% (11/21) of patients reached the TTT goal (BSA involvement ≤1% after 12 weeks of treatment with tapinarof cream added to a prescribed biologic). The proportion of patients reaching the TTT goal increased over time with the combined treatment (eFigure 1). Additionally, the mean percentage of BSA involvement (eFigure 2) as well as the mean values for PGA (eFigure 3) and PGA×BSA decreased over time. The mean percentage of BSA involvement was 5.0% at baseline and dropped to 2.0% by week 12. Similar reductions were observed for PGA and PGA×BSA scores at week 12.



After discontinuing tapinarof cream at week 12 and receiving only the biologic for 4 weeks, the proportion of patients maintaining 1% or less BSA involvement fell to 40.0% (8/20) at week 16, which was closer to that observed at week 8 (36% [9/25]) than at week 12 (52.4% [11/21])(eFigure 1).
The mean PASI score was 5.5 at baseline, then decreased over time when tapinarof cream was combined with a biologic (eFigure 4), falling to 3.1 by week 2 and 1.6 by week 12; it was maintained at 1.7 at week 16. Nine (30.0%) participants had psoriasis on the scalp at baseline with a mean PSSI score of 2.6, which decreased to 0.83 by week 2. By week 12, the mean PSSI score remained stable at 0.95 in the 2 (9.5%) participants who still had scalp involvement. The mean PSSI score increased slightly to 1.45 after patients received only the biologic for 4 weeks. At baseline, 3 (10.0%) patients had genital involvement (mean Static Physician’s Global Assessment of Genitalia score, 0.27). Symptoms resolved in 2 (66.7%) of these patients at week 2 and stayed consistent until week 16; the third patient withdrew at week 2.

Both DLQI and WI-NRS scores decreased with use of tapinarof cream added to a biologic up to week 12 (eFigures 5 and 6). Mean DLQI scores were 5.3 at baseline and 3.1 at week 12. At week 16, the mean DLQI score remained stable at 2.8. Mean WI-NRS scores decreased from 4.0 at baseline to 2.7 at week 12 with the therapy combination; at week 16, the mean WI-NRS score fell further to 1.8.


A total of 6 AEs were reported in 5 (16.7%) patients (Table 2). The majority (4/6 [67.0%]) of AEs were considered mild. Two reported cases of COVID-19 were both considered mild and unrelated. Mild folliculitis and moderate worsening of psoriasis in 2 (6.7%) different patients were the only AEs considered related to treatment. No serious AEs were reported, and no patient withdrew from the study due to an AE.

Comment
Disease activity improvements we observed with the nonsteroidal tapinarof cream were consistent with those reported when topical steroidal therapies were given to patients responding poorly to their current biologic. Our primary end point (proportion of patients with BSA involvement ≤1% after 12 weeks) showed that half (52% [11/21]) of patients whose BSA involvement was 3% or greater with a biologic for 24 weeks or more reached the TTT goal after 12 weeks of tapinarof-biologic treatment. Other studies of halobetasol propionate–tazarotene lotion16 and calcipotriene/betamethasone dipropionate foam17,18 added to the current biologic of poor responders found 60% to 68% of patients had reductions in their percentage BSA to 1% or lower at 12 to 16 weeks of treatment. Randomized studies showed etanercept plus topical clobetasol propionate foam20 or adalimumab plus calcipotriene/betamethasone dipropionate foam21 similarly enhanced treatment effects vs biologic alone.
A phase 3 PSOARING trial demonstrated benefit from treatment with tapinarof alone, with a remittive effect of approximately 4 months after discontinuation.25 Our data are consistent with these findings, with 40% (8/20) of patients demonstrating a remittive effect 4 weeks after discontinuing tapinarof while receiving a biologic. A similar maintenance effect was reported in another study in 50% (9/18) of patients treated with a biologic plus halobetasol propionate–tazarotene lotion.16 Additionally, when halcinonide ointment was given to patients receiving tildrakizumab, mean percentage of BSA involvement, PGA scores, PGA×BSA, and DLQI scores improved and were maintained 4 weeks after halcinonide ointment was stopped.19 Thus, topical therapy can augment and extend a biologic’s effect for up to 4 weeks.
In our study, tapinarof cream added to a biologic had a good safety and tolerability profile. Few AEs were recorded, with most being mild in nature, and no serious AEs or discontinuations due to AEs were reported. Only 1 case of mild folliculitis and 1 case of moderate worsening of psoriasis were considered treatment related. Further, no unexpected or new safety signals with the tapinarof-biologic combination were observed compared with tapinarof alone.27Prior studies have found that supplementing a biologic with topical therapy can reduce the probability of patients switching to another biologic.16,19 We previously found that adding halobetasol propionate–tazarotene lotion16 or calcipotriene/betamethasone dipropionate foam17 to a biologic helped reduce the probability of switching biologics from 88% to 90% at baseline to 12% to 24% after 12 weeks of combined therapy. Such combinations also could prevent a less responsive patient from being prescribed a higher biologic dose.19 These are important research findings, as patients—even when not responding well to their current biologic—are more likely to be tolerating that biologic well, and switching to a new biologic may introduce new safety or tolerability concerns. Thus, by enhancing the effect of a biologic with a topical therapy, one can avoid increasing the dose of the current biologic or switching to a new biologic, either of which may increase safety and/or tolerability risks. Switching biologics also has increased cost implications to the health care system and/or the patient. When comparing the cost of adding halobetasol propionate–tazarotene lotion to a biologic compared with switching to another biologic, the cost was 1.2 to 2.9 times higher to switch, depending on the biologic, compared with a smaller incremental cost increase to add a topical to the current biologic.16 Similar observations were reported with calcipotriene/betamethasone dipropionate foam plus a biologic.17 Although we did not evaluate biologic switching here, we anticipate a similar clinical scenario with a tapinarof-biologic combination.
Limitations of our study included the open-label design, lack of a control arm, and the relatively small study population; however, for studies investigating the safety and effectiveness of a treatment in a real-world setting, these limitations are common and are not unexpected. Our results also are consistent with the overall improvement seen in other studies16-21 examining the effects of adding a topical to a biologic. Future research is warranted to investigate a longer remittive effect and potential health care system and patient cost savings without having to switch biologics due to lack of effectiveness.
Conclusion
This study demonstrated that adjunctive use of nonsteroidal tapinarof cream 1% may enhance a biologic treatment effect in patients with moderate to severe plaque psoriasis, providing an adequate response for many patients who were not responding well to a biologic alone. Clinical outcomes improved with the tapinarof-biologic combination, and a remittive effect was noted 4 weeks after tapinarof discontinuation without any new safety signals. Adding tapinarof cream to a biologic also may prevent the need to switch biologics when patients do not sufficiently respond, preserving the safety and cost associated with a patient’s current biologic.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804. doi:10.1016/j.jaad.2019.04.042
- Elmets CA, Korman NJ, Prater EF, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with topical therapy and alternative medicine modalities for psoriasis severity measures. J Am Acad Dermatol. 2021;84:432-470. doi:10.1016/j.jaad.2020.07.087
- Menter A, Gelfand JM, Connor C, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management of psoriasis with systemic nonbiological therapies. J Am Acad Dermatol. 2020;82:1445-1486. doi:10.1016/j.jaad.2020.02.044
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072. doi:10.1016/j.jaad.2018.11.057
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis.J Am Acad Dermatol. 2017;76:290-298. doi:10.1016/j.jaad.2016.10.017
- Taltz. Prescribing information. Eli Lilly and Company; 2024.
- Cosentyx. Prescribing information. Novartis Pharmaceuticals Corporation; 2023.
- Tremfya. Prescribing information. Janssen Biotech, Inc; 2023.
- Skyrizi. Prescribing information. AbbVie Inc; 2024.
- Ilumya. Prescribing information. Sun Pharmaceutical Industries, Inc; 2020.
- Stelara. Prescribing information. Janssen Biotech, Inc; 2022.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Jensen JD, Delcambre MR, Nguyen G, et al. Biologic therapy with or without topical treatment in psoriasis: what does the current evidence say? Am J Clin Dermatol. 2014;15:379-385. doi:10.1007/s40257-014-0089-1
- Gustafson CJ, Watkins C, Hix E, et al. Combination therapy in psoriasis: an evidence-based review. Am J Clin Dermatol. 2013;14:9-25. doi:10.1007/s40257-012-0003-7
- Bagel J, Novak K, Nelson E. Adjunctive use of halobetasol propionate-tazarotene in biologic-experienced patients with psoriasis. Cutis. 2022;109:103-109. doi:10.12788/cutis.0451
- Bagel J, Nelson E, Zapata J, et al. Adjunctive use of calcipotriene/betamethasone dipropionate foam in a real-world setting curtails the cost of biologics without reducing efficacy in psoriasis. Dermatol Ther (Heidelb). 2020;10:1383-1396. doi:10.1007/s13555-020-00454-z
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:611-616.
- Bagel J, Novak K, Nelson E. Tildrakizumab in combination with topical halcinonide 0.1% ointment for treating moderate to severe plaque psoriasis. J Drugs Dermatol. 2023;22:766-772. doi:10.36849/jdd.6830
- Lebwohl MG, Kircik L, Callis Duffin K, et al. A randomized study to evaluate the efficacy and safety of adding topical therapy to etanercept in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2013;69:385-392. doi:10.1016/j.jaad.2013.03.031
- Thaci D, Ortonne JP, Chimenti S, et al. A phase IIIb, multicentre, randomized, double-blind, vehicle-controlled study of the efficacy and safety of adalimumab with and without calcipotriol/betamethasone topical treatment in patients with moderate to severe psoriasis: the BELIEVE study. Br J Dermatol. 2010;163:402-411. doi:10.1111/j.1365-2133.2010.09791.x
- Vtama. Prescribing information. Dermavant Sciences, Inc; 2022.
- Bobonich M, Gorelick J, Aldredge L, et al. Tapinarof, a novel, first-in-class, topical therapeutic aryl hydrocarbon receptor agonist for the management of psoriasis. J Drugs Dermatol. 2023;22:779-784. doi:10.36849/jdd.7317
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Strober B, Stein Gold L, Bissonnette R, et al. One-year safety and efficacy of tapinarof cream for the treatment of plaque psoriasis: results from the PSOARING 3 trial. J Am Acad Dermatol. 2022;87:800-806. doi:10.1016/j.jaad.2022.06.1171
- Kircik L, Zirwas M, Kwatra SG, et al. Rapid improvements in itch with tapinarof cream 1% once daily in two phase 3 trials in adults with mild to severe plaque psoriasis. Dermatol Ther (Heidelb). 2024;14:201-211. doi:10.1007/s13555-023-01068-x
- Bagel J, Gold LS, Del Rosso J, et al. Tapinarof cream 1% once daily for the treatment of plaque psoriasis: patient-reported outcomes from the PSOARING 3 trial. J Am Acad Dermatol. 2023;89:936-944. doi:10.1016/j.jaad.2023.04.061
- Abdin R, Kircik L, Issa NT. First use of combination oral deucravacitinib with tapinarof cream for treatment of severe plaque psoriasis. J Drugs Dermatol. 2024;23:192-194. doi:10.36849/jdd.8091
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804. doi:10.1016/j.jaad.2019.04.042
- Elmets CA, Korman NJ, Prater EF, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with topical therapy and alternative medicine modalities for psoriasis severity measures. J Am Acad Dermatol. 2021;84:432-470. doi:10.1016/j.jaad.2020.07.087
- Menter A, Gelfand JM, Connor C, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management of psoriasis with systemic nonbiological therapies. J Am Acad Dermatol. 2020;82:1445-1486. doi:10.1016/j.jaad.2020.02.044
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80:1029-1072. doi:10.1016/j.jaad.2018.11.057
- Armstrong AW, Siegel MP, Bagel J, et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis.J Am Acad Dermatol. 2017;76:290-298. doi:10.1016/j.jaad.2016.10.017
- Taltz. Prescribing information. Eli Lilly and Company; 2024.
- Cosentyx. Prescribing information. Novartis Pharmaceuticals Corporation; 2023.
- Tremfya. Prescribing information. Janssen Biotech, Inc; 2023.
- Skyrizi. Prescribing information. AbbVie Inc; 2024.
- Ilumya. Prescribing information. Sun Pharmaceutical Industries, Inc; 2020.
- Stelara. Prescribing information. Janssen Biotech, Inc; 2022.
- Bagel J, Gold LS. Combining topical psoriasis treatment to enhance systemic and phototherapy: a review of the literature. J Drugs Dermatol. 2017;16:1209-1222.
- Jensen JD, Delcambre MR, Nguyen G, et al. Biologic therapy with or without topical treatment in psoriasis: what does the current evidence say? Am J Clin Dermatol. 2014;15:379-385. doi:10.1007/s40257-014-0089-1
- Gustafson CJ, Watkins C, Hix E, et al. Combination therapy in psoriasis: an evidence-based review. Am J Clin Dermatol. 2013;14:9-25. doi:10.1007/s40257-012-0003-7
- Bagel J, Novak K, Nelson E. Adjunctive use of halobetasol propionate-tazarotene in biologic-experienced patients with psoriasis. Cutis. 2022;109:103-109. doi:10.12788/cutis.0451
- Bagel J, Nelson E, Zapata J, et al. Adjunctive use of calcipotriene/betamethasone dipropionate foam in a real-world setting curtails the cost of biologics without reducing efficacy in psoriasis. Dermatol Ther (Heidelb). 2020;10:1383-1396. doi:10.1007/s13555-020-00454-z
- Bagel J, Zapata J, Nelson E. A prospective, open-label study evaluating adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% foam in psoriasis patients with inadequate response to biologic therapy. J Drugs Dermatol. 2018;17:611-616.
- Bagel J, Novak K, Nelson E. Tildrakizumab in combination with topical halcinonide 0.1% ointment for treating moderate to severe plaque psoriasis. J Drugs Dermatol. 2023;22:766-772. doi:10.36849/jdd.6830
- Lebwohl MG, Kircik L, Callis Duffin K, et al. A randomized study to evaluate the efficacy and safety of adding topical therapy to etanercept in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2013;69:385-392. doi:10.1016/j.jaad.2013.03.031
- Thaci D, Ortonne JP, Chimenti S, et al. A phase IIIb, multicentre, randomized, double-blind, vehicle-controlled study of the efficacy and safety of adalimumab with and without calcipotriol/betamethasone topical treatment in patients with moderate to severe psoriasis: the BELIEVE study. Br J Dermatol. 2010;163:402-411. doi:10.1111/j.1365-2133.2010.09791.x
- Vtama. Prescribing information. Dermavant Sciences, Inc; 2022.
- Bobonich M, Gorelick J, Aldredge L, et al. Tapinarof, a novel, first-in-class, topical therapeutic aryl hydrocarbon receptor agonist for the management of psoriasis. J Drugs Dermatol. 2023;22:779-784. doi:10.36849/jdd.7317
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Strober B, Stein Gold L, Bissonnette R, et al. One-year safety and efficacy of tapinarof cream for the treatment of plaque psoriasis: results from the PSOARING 3 trial. J Am Acad Dermatol. 2022;87:800-806. doi:10.1016/j.jaad.2022.06.1171
- Kircik L, Zirwas M, Kwatra SG, et al. Rapid improvements in itch with tapinarof cream 1% once daily in two phase 3 trials in adults with mild to severe plaque psoriasis. Dermatol Ther (Heidelb). 2024;14:201-211. doi:10.1007/s13555-023-01068-x
- Bagel J, Gold LS, Del Rosso J, et al. Tapinarof cream 1% once daily for the treatment of plaque psoriasis: patient-reported outcomes from the PSOARING 3 trial. J Am Acad Dermatol. 2023;89:936-944. doi:10.1016/j.jaad.2023.04.061
- Abdin R, Kircik L, Issa NT. First use of combination oral deucravacitinib with tapinarof cream for treatment of severe plaque psoriasis. J Drugs Dermatol. 2024;23:192-194. doi:10.36849/jdd.8091
Safety and Effectiveness of Nonsteroidal Tapinarof Cream 1% Added to Ongoing Biologic Therapy for Treatment of Moderate to Severe Plaque Psoriasis
Safety and Effectiveness of Nonsteroidal Tapinarof Cream 1% Added to Ongoing Biologic Therapy for Treatment of Moderate to Severe Plaque Psoriasis
Practice Points
- Patients with moderate to severe psoriasis do not always reach treatment goals with biologic therapy alone.
- Adjunctive use of nonsteroidal tapinarof cream 1% may enhance the effects of ongoing biologic therapy in patients with moderate to severe plaque psoriasis, possibly avoiding the need to switch to another biologic.
- Patients with moderate to severe plaque psoriasis who are not adequately responding to biologics may benefit from adding tapinarof cream 1% to their current regimen.
Pathogenic Significance of Serum Syndecan-1 and Syndecan-4 in Psoriasis
Pathogenic Significance of Serum Syndecan-1 and Syndecan-4 in Psoriasis
Psoriasis, one of the most researched diseases in dermatology, has a complex pathogenesis that is not yet fully understood. One of the most important stages of psoriasis pathogenesis is the proliferation of T helper (Th) 17 cells by IL-23 released from myeloid dendritic cells. Cytokines such as tumor necrosis factor (TNF) α released from Th1 cells and IL-17 and IL-22 released from Th17 cells are known to induce the proliferation of keratinocytes and the release of chemokines responsible for neutrophil chemotaxis.1
Although secondary messengers such as cytokines and chemokines, which provide cell interaction with the extracellular matrix (ECM), have their own specific receptors, it is known that syndecans (SDCs) play a role in ECM and cell interactions and have receptor or coreceptor functions.2 In humans, 4 types of SDCs have been identified (SDC1-SDC4), which are type I transmembrane proteoglycans found in all nucleated cells. Syndecans consist of heparan sulfate glycosaminoglycan chains that are structurally linked to a core protein sequence. The molecule has cytoplasmic, transmembrane, and extracellular domains.2,3 While SDCs often are described as coreceptors for integrins and growth factor and hormone receptors, they also are capable of acting as signaling receptors by engaging intracellular messengers, including actin-related proteins and protein kinases.4
Prior research has indicated that the release of heparanase from the lysosomes of leukocytes during infection, inflammation, and endothelial damage causes cleavage of heparan sulfate glycosaminoglycans from the extracellular domains of SDCs. The peptide chains at the SDC core then are separated by matrix metalloproteinases in a process known as shedding. The shed SDCs may have either a stimulating or a suppressive effect on their receptor activity. Several cytokines are known to cause SDC shedding.5,6 Many studies in recent years have reported that SDCs play a role in the pathogenesis of inflammatory diseases, for which serum levels of soluble SDCs can be biomarkers.7
In this study, we aimed to evaluate and compare serum SDC1, SDC4, TNF-α, and IL-17A levels in patients with psoriasis vs healthy controls. Additionally, by reviewing the literature data, we analyzed whether SDCs can be implicated in the pathogenesis of psoriasis and their potential role in this process.
Methods
The study population consisted of 40 patients with psoriasis and 40 healthy controls. Age and sex characteristics were similar between the 2 groups, but weight distribution was not. The psoriasis group included patients older than 18 years who had received a clinical and/or histologic diagnosis, had no systemic disease other than psoriasis in their medical history, and had not used any systemic treatment or phototherapy for the past 3 months. Healthy patients older than 18 years who had no medical history of inflammatory disease were included in the control group. Participants provided signed consent.
Data such as medical history, laboratory findings, and physical specifications were recorded. A Psoriasis Area and Severity Index (PASI) score of 10 or lower was considered mild disease, and a score higher than 10 was considered moderate to severe disease. An enzyme-linked immunosorbent assay was used to measure SDC1, SDC4, TNF-α, and IL-17A levels.
The data were evaluated using the IBM SPSS Statistics V22.0 statistical package program. A P value of <.05 was considered statistically significant. The conformity of the data to a normal distribution was examined using a Shapiro-Wilk test. Normally distributed variables were expressed as mean (SD) and nonnormally distributed variables were expressed as median (interquartile range [IQR]). Data were compared between the 2 study groups using either a student t test (normal distribution) or Mann-Whitney U test (nonnormal distribution). Categorical variables were expressed as numbers and percentages. Categorical data were compared using a χ2 test. Associations among SDC1, SDC4, TNF-α, IL-17A, and other variables were assessed using Spearman rank correlation. A binary logistic regression analysis was used to determine whether serum SDC1 and SDC4 levels were independent risk factors for psoriasis.
Results
The 2 study groups showed similar demographic characteristics in terms of sex (P=.67) and age (P=.22) distribution. The mean (SD) PASI score in the psoriasis group was 12.33 (7.62); the mean (SD) disease duration was 11.10 (8.00) years. Body weight and BMI were both significantly higher in the psoriasis group (P=.027 and P=.029, respectively) compared with the control group (eTable 1).
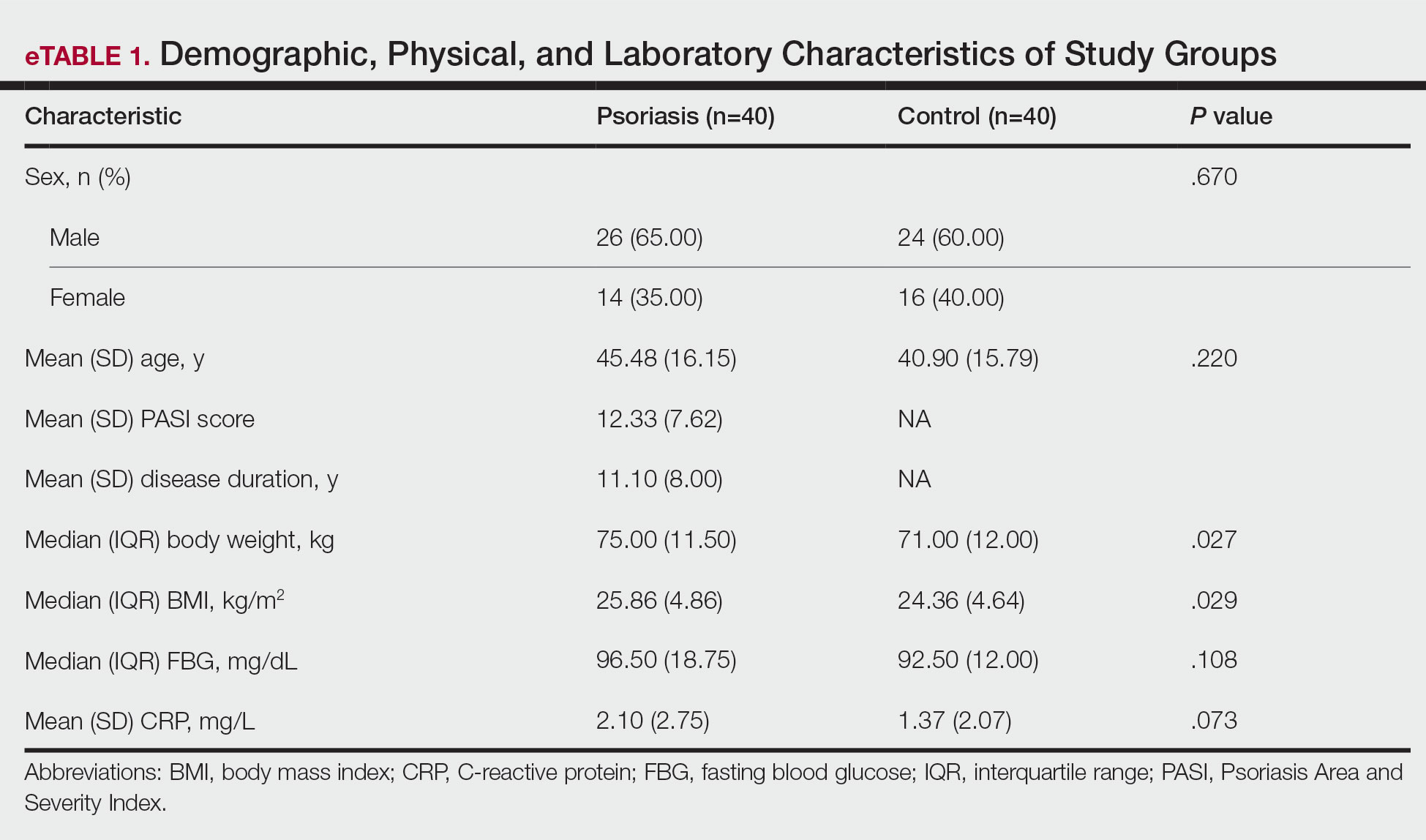
The mean (SD) serum SDC1 level was 119.52 ng/mL (69.53 ng/mL) in the psoriasis group, which was significantly higher than the control group (82.81 ng/mL [51.85 ng/mL])(P=.011)(eTable 2)(eFigure 1). The median (IQR) serum SDC4 level also was significantly higher in the psoriasis group compared with the control group (5.78 ng/mL [7.09 ng/mL] vs 3.92 ng/mL [2.88 ng/mL])(P=.030)(eTable 2)(eFigure 2). The median (IQR) IL-17A value was 59.94 pg/mL (12.97 pg/mL) in the psoriasis group, which was significantly higher than the control group (37.74 pg/mL [15.10 pg/mL])(P<.001)(eTable 2)(eFigure 3). The median (IQR) serum TNF-α level was 25.07 pg/mL (41.70 pg/mL) in the psoriasis group and 18.21 pg/mL (48.51 pg/mL) in the control group; however, the difference was not statistically significance (P=.444)(eTable 2)(eFigure 4).
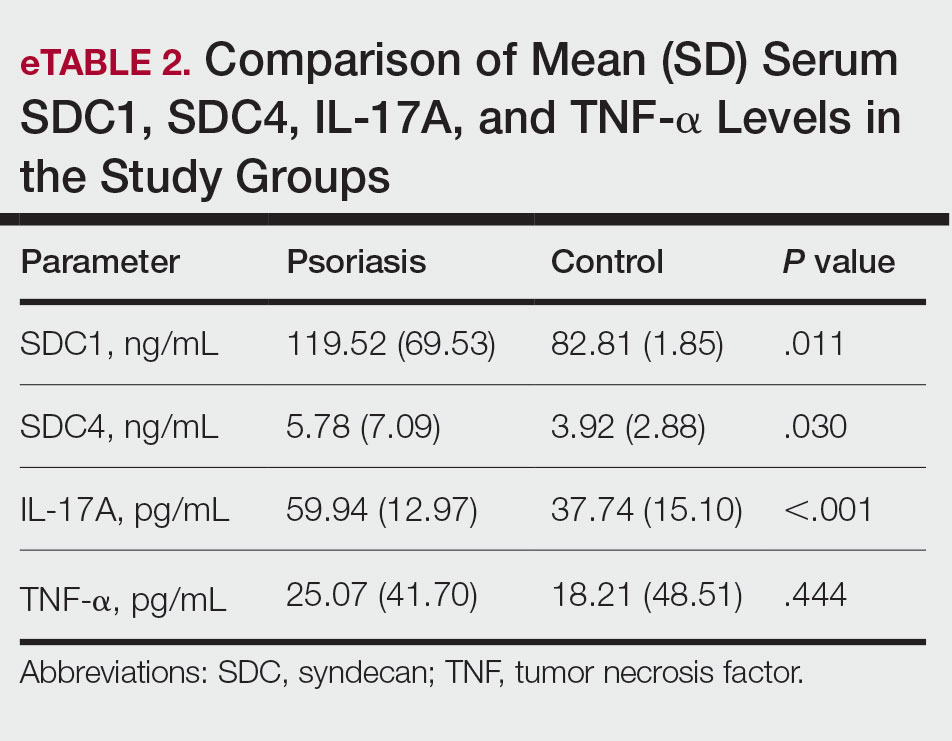
A significant positive correlation was found between serum SDC1 and PASI score (p=0.064; P=.03). Furthermore, significant positive correlations were identified between serum SDC1 and body weight (p=0.404; P<.001), disease duration (p=0.377; P=.008), and C-reactive protein (p=0.327; P=.002). A significant positive correlation also was identified between SDC4 and IL-17A (p=0.265; P=.009). Serum TNF-α was positively correlated with IL-17A (p=0.384; P<.001) and BMI (p=0.234; P=.020)(eTable 3).
Logistic regression analysis showed that high SDC1 levels were independently associated with the development of psoriasis (odds ratio [OR], 1.009; 95% CI, 1.000-1.017; P=.049)(eTable 4).
Comment
Tumor necrosis factor α and IL-17A are key cytokines whose roles in the pathogenesis of psoriasis are well established. Arican et al,8 Kyriakou et al,9 and Xuan et al10 previously reported a lack of any correlation between TNF-α and IL-17A in the pathogenesis of psoriasis; however, we observed a positive correlation between TNF-α and IL-17A in our study. This finding may be due to the abundant TNF-α production by myeloid dendritic cells involved in the transformation of naive T lymphocytes into IL-17A–secreting Th17 lymphocytes, which can also secrete TNF-α.
After the molecular cloning of SDCs by Saunders et al11 in 1989, SDCs gained attention and have been the focus of many studies for their part in the pathogenesis of conditions such as inflammatory diseases, carcinogenesis, infections, sepsis, and trauma.6,12 Among the inflammatory diseases sharing similar pathogenetic features to psoriasis, serum SDC4 levels are found to be elevated in rheumatoid arthritis and are correlated with disease activity.13 Cekic et al14 reported that serum SDC1 levels were significantly higher in patients with Crohn disease than controls (P=.03). Additionally, serum SDC1 levels were higher in patients with active disease compared with those who were in remission. Correlations between SDC1 and disease severity and C-reactive protein also have been found.14 Serum SDC-1 levels found to be elevated in patients with systemic lupus erythematosus were compared to the controls and were correlated with disease activity.15 Nakao et al16 reported that the serum SDC4 levels were significantly higher in patients with atopic dermatitis compared to controls (P<.01); further, SDC4 levels were correlated with severity of the disease.
Jaiswal et al17 reported that SDC1 is abundant on the surface of IL-17A–secreting γδ T lymphocytes (Tγδ17), whose contribution to psoriasis pathogenesis is known. When subjected to treatment with imiquimod, SDC1-suppressed mice displayed increased psoriasiform dermatitis compared with wild-type counterparts. The authors stated that SDC1 may play a role in controlling homeostasis of Tγδ17
In a study examining changes in the ECM in patients with psoriasis, it was observed that the expression of
A study conducted by Koliakou et al20 showed that, in healthy skin, SDC1 was expressed in almost the full thickness of the epidermis, but lowest expression was in the basal-layer keratinocytes. In a psoriatic epidermis, unlike the normal epidermis, SDC1 was found to be more intensely expressed in the keratinocytes of the basal layer, where keratinocyte proliferation occurs. In this study, SDC4 was expressed mainly at lower levels in a healthy epidermis, especially in the spinous and the basal layers. In a psoriatic epidermis, SDC4 was absent from all the layers. In the same study, gelatin-based carriers containing anti–TNF-α and anti–IL-17A were applied to a full-thickness epidermis with psoriatic lesions, after which SDC1 expression was observed to decrease almost completely in the psoriatic epidermis; there was no change in SDC4 expression, which also was not seen in the psoriatic epidermis. The authors claimed the application of these gelatin-based carriers could be a possible treatment modality for psoriasis, and the study provides evidence for the involvement of SDC1 and/or SDC4 in the pathogenesis of psoriasis
Limitations of the current study include small sample size, lack of longitudinal data, lack of tissue testing of these molecules, and lack of external validation.
Conclusion
Overall, research has shown that SDCs play important roles in inflammatory processes, and more widespread inflammation has been associated with increased shedding of these molecules into the ECM and higher serum levels. In our study, serum SDC1, SDC4, and IL-17A levels were increased in patients with psoriasis compared to the healthy controls. A logistic regression analysis indicated that high serum SDC1 levels may be an independent risk factor for development of psoriasis. The increase in serum SDC1 and SDC4 levels and the positive correlation between SDC1 levels and disease severity observed in our study strongly implicate SDCs in the inflammatory disease psoriasis. The precise role of SDCs in the pathogenesis of psoriasis and the implications of targeting these molecules are the subject of more in-depth studies in the future.
Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
Uings IJ, Farrow SN. Cell receptors and cell signaling. Mol Pathol. 2000;53:295-299.
Kirkpatrick CA, Selleck SB. Heparan sulfate proteoglycans at a glance.J Cell Sci. 2007;120:1829-1832.
Stepp MA, Pal-Ghosh S, Tadvalkar G, et al. Syndecan-1 and its expanding list of contacts. Adv Wound Care (New Rochelle). 2015;4:235-249.
Rangarajan S, Richter JR, Richter RP, et al. Heparanase-enhanced shedding of syndecan-1 and its role in driving disease pathogenesis and progression. J Histochem Cytochem. 2020;68:823-840.
Gopal S, Arokiasamy S, Pataki C, et al. Syndecan receptors: pericellular regulators in development and inflammatory disease. Open Biol. 2021;11:200377.
Bertrand J, Bollmann M. Soluble syndecans: biomarkers for diseases and therapeutic options. Br J Pharmacol. 2019;176:67-81.
Arican O, Aral M, Sasmaz S, et al. Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm. 2005;2005:273-279.
Kyriakou A, Patsatsi A, Vyzantiadis TA, et al. Serum levels of TNF-α, IL12/23 p40, and IL-17 in psoriatic patients with and without nail psoriasis: a cross-sectional study. ScientificWorldJournal. 2014;2014:508178.
Xuan ML, Lu CJ, Han L, et al. Circulating levels of inflammatory cytokines in patients with psoriasis vulgaris of different Chinese medicine syndromes. Chin J Integr Med. 2015;21:108-114.
Saunders S, Jalkanen M, O’Farrell S, et al. Molecular cloning of syndecan, an integral membrane proteoglycan. J Cell Biol. 1989;108:1547-1556.
Manon-Jensen T, Itoh Y, Couchman JR. Proteoglycans in health and disease: the multiple roles of syndecan shedding. FEBS J. 2010;277:3876-3889.
Zhao J, Ye X, Zhang Z. Syndecan-4 is correlated with disease activity and serological characteristic of rheumatoid arthritis. Adv Rheumatol. 2022;62:21.
Cekic C, Kırcı A, Vatansever S, et al. Serum syndecan-1 levels and its relationship to disease activity in patients with Crohn’s disease. Gastroenterol Res Pract. 2015;2015:850351.
Minowa K, Amano H, Nakano S, et al. Elevated serum level of circulating syndecan-1 (CD138) in active systemic lupus erythematosus. Autoimmunity. 2011;44:357-362.
Nakao M, Sugaya M, Takahashi N, et al. Increased syndecan-4 expression in sera and skin of patients with atopic dermatitis. Arch Dermatol Res. 2016;308:655-660.
Jaiswal AK, Sadasivam M, Archer NK, et al. Syndecan-1 regulates psoriasiform dermatitis by controlling homeostasis of IL-17-producing γδ T cells. J Immunol. 2018;201:1651-1661
Wagner MFMG, Theodoro TR, Filho CASM, et al. Extracellular matrix alterations in the skin of patients affected by psoriasis. BMC Mol Cell Biol. 2021;22:55.
Peters F, Rahn S, Mengel M, et al. Syndecan-1 shedding by meprin β impairs keratinocyte adhesion and differentiation in hyperkeratosis. Matrix Biol. 2021;102:37-69.
Koliakou E, Eleni MM, Koumentakou I, et al. Altered distribution and expression of syndecan-1 and -4 as an additional hallmark in psoriasis. Int J Mol Sci. 2022;23:6511.
Doss RW, El-Rifaie AA, Said AN, et al. Cutaneous syndecan-1 expression before and after phototherapy in psoriasis. Indian J Dermatol Venereol Leprol. 2020;86:439-440.
Psoriasis, one of the most researched diseases in dermatology, has a complex pathogenesis that is not yet fully understood. One of the most important stages of psoriasis pathogenesis is the proliferation of T helper (Th) 17 cells by IL-23 released from myeloid dendritic cells. Cytokines such as tumor necrosis factor (TNF) α released from Th1 cells and IL-17 and IL-22 released from Th17 cells are known to induce the proliferation of keratinocytes and the release of chemokines responsible for neutrophil chemotaxis.1
Although secondary messengers such as cytokines and chemokines, which provide cell interaction with the extracellular matrix (ECM), have their own specific receptors, it is known that syndecans (SDCs) play a role in ECM and cell interactions and have receptor or coreceptor functions.2 In humans, 4 types of SDCs have been identified (SDC1-SDC4), which are type I transmembrane proteoglycans found in all nucleated cells. Syndecans consist of heparan sulfate glycosaminoglycan chains that are structurally linked to a core protein sequence. The molecule has cytoplasmic, transmembrane, and extracellular domains.2,3 While SDCs often are described as coreceptors for integrins and growth factor and hormone receptors, they also are capable of acting as signaling receptors by engaging intracellular messengers, including actin-related proteins and protein kinases.4
Prior research has indicated that the release of heparanase from the lysosomes of leukocytes during infection, inflammation, and endothelial damage causes cleavage of heparan sulfate glycosaminoglycans from the extracellular domains of SDCs. The peptide chains at the SDC core then are separated by matrix metalloproteinases in a process known as shedding. The shed SDCs may have either a stimulating or a suppressive effect on their receptor activity. Several cytokines are known to cause SDC shedding.5,6 Many studies in recent years have reported that SDCs play a role in the pathogenesis of inflammatory diseases, for which serum levels of soluble SDCs can be biomarkers.7
In this study, we aimed to evaluate and compare serum SDC1, SDC4, TNF-α, and IL-17A levels in patients with psoriasis vs healthy controls. Additionally, by reviewing the literature data, we analyzed whether SDCs can be implicated in the pathogenesis of psoriasis and their potential role in this process.
Methods
The study population consisted of 40 patients with psoriasis and 40 healthy controls. Age and sex characteristics were similar between the 2 groups, but weight distribution was not. The psoriasis group included patients older than 18 years who had received a clinical and/or histologic diagnosis, had no systemic disease other than psoriasis in their medical history, and had not used any systemic treatment or phototherapy for the past 3 months. Healthy patients older than 18 years who had no medical history of inflammatory disease were included in the control group. Participants provided signed consent.
Data such as medical history, laboratory findings, and physical specifications were recorded. A Psoriasis Area and Severity Index (PASI) score of 10 or lower was considered mild disease, and a score higher than 10 was considered moderate to severe disease. An enzyme-linked immunosorbent assay was used to measure SDC1, SDC4, TNF-α, and IL-17A levels.
The data were evaluated using the IBM SPSS Statistics V22.0 statistical package program. A P value of <.05 was considered statistically significant. The conformity of the data to a normal distribution was examined using a Shapiro-Wilk test. Normally distributed variables were expressed as mean (SD) and nonnormally distributed variables were expressed as median (interquartile range [IQR]). Data were compared between the 2 study groups using either a student t test (normal distribution) or Mann-Whitney U test (nonnormal distribution). Categorical variables were expressed as numbers and percentages. Categorical data were compared using a χ2 test. Associations among SDC1, SDC4, TNF-α, IL-17A, and other variables were assessed using Spearman rank correlation. A binary logistic regression analysis was used to determine whether serum SDC1 and SDC4 levels were independent risk factors for psoriasis.
Results
The 2 study groups showed similar demographic characteristics in terms of sex (P=.67) and age (P=.22) distribution. The mean (SD) PASI score in the psoriasis group was 12.33 (7.62); the mean (SD) disease duration was 11.10 (8.00) years. Body weight and BMI were both significantly higher in the psoriasis group (P=.027 and P=.029, respectively) compared with the control group (eTable 1).

The mean (SD) serum SDC1 level was 119.52 ng/mL (69.53 ng/mL) in the psoriasis group, which was significantly higher than the control group (82.81 ng/mL [51.85 ng/mL])(P=.011)(eTable 2)(eFigure 1). The median (IQR) serum SDC4 level also was significantly higher in the psoriasis group compared with the control group (5.78 ng/mL [7.09 ng/mL] vs 3.92 ng/mL [2.88 ng/mL])(P=.030)(eTable 2)(eFigure 2). The median (IQR) IL-17A value was 59.94 pg/mL (12.97 pg/mL) in the psoriasis group, which was significantly higher than the control group (37.74 pg/mL [15.10 pg/mL])(P<.001)(eTable 2)(eFigure 3). The median (IQR) serum TNF-α level was 25.07 pg/mL (41.70 pg/mL) in the psoriasis group and 18.21 pg/mL (48.51 pg/mL) in the control group; however, the difference was not statistically significance (P=.444)(eTable 2)(eFigure 4).





A significant positive correlation was found between serum SDC1 and PASI score (p=0.064; P=.03). Furthermore, significant positive correlations were identified between serum SDC1 and body weight (p=0.404; P<.001), disease duration (p=0.377; P=.008), and C-reactive protein (p=0.327; P=.002). A significant positive correlation also was identified between SDC4 and IL-17A (p=0.265; P=.009). Serum TNF-α was positively correlated with IL-17A (p=0.384; P<.001) and BMI (p=0.234; P=.020)(eTable 3).

Logistic regression analysis showed that high SDC1 levels were independently associated with the development of psoriasis (odds ratio [OR], 1.009; 95% CI, 1.000-1.017; P=.049)(eTable 4).

Comment
Tumor necrosis factor α and IL-17A are key cytokines whose roles in the pathogenesis of psoriasis are well established. Arican et al,8 Kyriakou et al,9 and Xuan et al10 previously reported a lack of any correlation between TNF-α and IL-17A in the pathogenesis of psoriasis; however, we observed a positive correlation between TNF-α and IL-17A in our study. This finding may be due to the abundant TNF-α production by myeloid dendritic cells involved in the transformation of naive T lymphocytes into IL-17A–secreting Th17 lymphocytes, which can also secrete TNF-α.
After the molecular cloning of SDCs by Saunders et al11 in 1989, SDCs gained attention and have been the focus of many studies for their part in the pathogenesis of conditions such as inflammatory diseases, carcinogenesis, infections, sepsis, and trauma.6,12 Among the inflammatory diseases sharing similar pathogenetic features to psoriasis, serum SDC4 levels are found to be elevated in rheumatoid arthritis and are correlated with disease activity.13 Cekic et al14 reported that serum SDC1 levels were significantly higher in patients with Crohn disease than controls (P=.03). Additionally, serum SDC1 levels were higher in patients with active disease compared with those who were in remission. Correlations between SDC1 and disease severity and C-reactive protein also have been found.14 Serum SDC-1 levels found to be elevated in patients with systemic lupus erythematosus were compared to the controls and were correlated with disease activity.15 Nakao et al16 reported that the serum SDC4 levels were significantly higher in patients with atopic dermatitis compared to controls (P<.01); further, SDC4 levels were correlated with severity of the disease.
Jaiswal et al17 reported that SDC1 is abundant on the surface of IL-17A–secreting γδ T lymphocytes (Tγδ17), whose contribution to psoriasis pathogenesis is known. When subjected to treatment with imiquimod, SDC1-suppressed mice displayed increased psoriasiform dermatitis compared with wild-type counterparts. The authors stated that SDC1 may play a role in controlling homeostasis of Tγδ17
In a study examining changes in the ECM in patients with psoriasis, it was observed that the expression of
A study conducted by Koliakou et al20 showed that, in healthy skin, SDC1 was expressed in almost the full thickness of the epidermis, but lowest expression was in the basal-layer keratinocytes. In a psoriatic epidermis, unlike the normal epidermis, SDC1 was found to be more intensely expressed in the keratinocytes of the basal layer, where keratinocyte proliferation occurs. In this study, SDC4 was expressed mainly at lower levels in a healthy epidermis, especially in the spinous and the basal layers. In a psoriatic epidermis, SDC4 was absent from all the layers. In the same study, gelatin-based carriers containing anti–TNF-α and anti–IL-17A were applied to a full-thickness epidermis with psoriatic lesions, after which SDC1 expression was observed to decrease almost completely in the psoriatic epidermis; there was no change in SDC4 expression, which also was not seen in the psoriatic epidermis. The authors claimed the application of these gelatin-based carriers could be a possible treatment modality for psoriasis, and the study provides evidence for the involvement of SDC1 and/or SDC4 in the pathogenesis of psoriasis
Limitations of the current study include small sample size, lack of longitudinal data, lack of tissue testing of these molecules, and lack of external validation.
Conclusion
Overall, research has shown that SDCs play important roles in inflammatory processes, and more widespread inflammation has been associated with increased shedding of these molecules into the ECM and higher serum levels. In our study, serum SDC1, SDC4, and IL-17A levels were increased in patients with psoriasis compared to the healthy controls. A logistic regression analysis indicated that high serum SDC1 levels may be an independent risk factor for development of psoriasis. The increase in serum SDC1 and SDC4 levels and the positive correlation between SDC1 levels and disease severity observed in our study strongly implicate SDCs in the inflammatory disease psoriasis. The precise role of SDCs in the pathogenesis of psoriasis and the implications of targeting these molecules are the subject of more in-depth studies in the future.
Psoriasis, one of the most researched diseases in dermatology, has a complex pathogenesis that is not yet fully understood. One of the most important stages of psoriasis pathogenesis is the proliferation of T helper (Th) 17 cells by IL-23 released from myeloid dendritic cells. Cytokines such as tumor necrosis factor (TNF) α released from Th1 cells and IL-17 and IL-22 released from Th17 cells are known to induce the proliferation of keratinocytes and the release of chemokines responsible for neutrophil chemotaxis.1
Although secondary messengers such as cytokines and chemokines, which provide cell interaction with the extracellular matrix (ECM), have their own specific receptors, it is known that syndecans (SDCs) play a role in ECM and cell interactions and have receptor or coreceptor functions.2 In humans, 4 types of SDCs have been identified (SDC1-SDC4), which are type I transmembrane proteoglycans found in all nucleated cells. Syndecans consist of heparan sulfate glycosaminoglycan chains that are structurally linked to a core protein sequence. The molecule has cytoplasmic, transmembrane, and extracellular domains.2,3 While SDCs often are described as coreceptors for integrins and growth factor and hormone receptors, they also are capable of acting as signaling receptors by engaging intracellular messengers, including actin-related proteins and protein kinases.4
Prior research has indicated that the release of heparanase from the lysosomes of leukocytes during infection, inflammation, and endothelial damage causes cleavage of heparan sulfate glycosaminoglycans from the extracellular domains of SDCs. The peptide chains at the SDC core then are separated by matrix metalloproteinases in a process known as shedding. The shed SDCs may have either a stimulating or a suppressive effect on their receptor activity. Several cytokines are known to cause SDC shedding.5,6 Many studies in recent years have reported that SDCs play a role in the pathogenesis of inflammatory diseases, for which serum levels of soluble SDCs can be biomarkers.7
In this study, we aimed to evaluate and compare serum SDC1, SDC4, TNF-α, and IL-17A levels in patients with psoriasis vs healthy controls. Additionally, by reviewing the literature data, we analyzed whether SDCs can be implicated in the pathogenesis of psoriasis and their potential role in this process.
Methods
The study population consisted of 40 patients with psoriasis and 40 healthy controls. Age and sex characteristics were similar between the 2 groups, but weight distribution was not. The psoriasis group included patients older than 18 years who had received a clinical and/or histologic diagnosis, had no systemic disease other than psoriasis in their medical history, and had not used any systemic treatment or phototherapy for the past 3 months. Healthy patients older than 18 years who had no medical history of inflammatory disease were included in the control group. Participants provided signed consent.
Data such as medical history, laboratory findings, and physical specifications were recorded. A Psoriasis Area and Severity Index (PASI) score of 10 or lower was considered mild disease, and a score higher than 10 was considered moderate to severe disease. An enzyme-linked immunosorbent assay was used to measure SDC1, SDC4, TNF-α, and IL-17A levels.
The data were evaluated using the IBM SPSS Statistics V22.0 statistical package program. A P value of <.05 was considered statistically significant. The conformity of the data to a normal distribution was examined using a Shapiro-Wilk test. Normally distributed variables were expressed as mean (SD) and nonnormally distributed variables were expressed as median (interquartile range [IQR]). Data were compared between the 2 study groups using either a student t test (normal distribution) or Mann-Whitney U test (nonnormal distribution). Categorical variables were expressed as numbers and percentages. Categorical data were compared using a χ2 test. Associations among SDC1, SDC4, TNF-α, IL-17A, and other variables were assessed using Spearman rank correlation. A binary logistic regression analysis was used to determine whether serum SDC1 and SDC4 levels were independent risk factors for psoriasis.
Results
The 2 study groups showed similar demographic characteristics in terms of sex (P=.67) and age (P=.22) distribution. The mean (SD) PASI score in the psoriasis group was 12.33 (7.62); the mean (SD) disease duration was 11.10 (8.00) years. Body weight and BMI were both significantly higher in the psoriasis group (P=.027 and P=.029, respectively) compared with the control group (eTable 1).

The mean (SD) serum SDC1 level was 119.52 ng/mL (69.53 ng/mL) in the psoriasis group, which was significantly higher than the control group (82.81 ng/mL [51.85 ng/mL])(P=.011)(eTable 2)(eFigure 1). The median (IQR) serum SDC4 level also was significantly higher in the psoriasis group compared with the control group (5.78 ng/mL [7.09 ng/mL] vs 3.92 ng/mL [2.88 ng/mL])(P=.030)(eTable 2)(eFigure 2). The median (IQR) IL-17A value was 59.94 pg/mL (12.97 pg/mL) in the psoriasis group, which was significantly higher than the control group (37.74 pg/mL [15.10 pg/mL])(P<.001)(eTable 2)(eFigure 3). The median (IQR) serum TNF-α level was 25.07 pg/mL (41.70 pg/mL) in the psoriasis group and 18.21 pg/mL (48.51 pg/mL) in the control group; however, the difference was not statistically significance (P=.444)(eTable 2)(eFigure 4).





A significant positive correlation was found between serum SDC1 and PASI score (p=0.064; P=.03). Furthermore, significant positive correlations were identified between serum SDC1 and body weight (p=0.404; P<.001), disease duration (p=0.377; P=.008), and C-reactive protein (p=0.327; P=.002). A significant positive correlation also was identified between SDC4 and IL-17A (p=0.265; P=.009). Serum TNF-α was positively correlated with IL-17A (p=0.384; P<.001) and BMI (p=0.234; P=.020)(eTable 3).

Logistic regression analysis showed that high SDC1 levels were independently associated with the development of psoriasis (odds ratio [OR], 1.009; 95% CI, 1.000-1.017; P=.049)(eTable 4).

Comment
Tumor necrosis factor α and IL-17A are key cytokines whose roles in the pathogenesis of psoriasis are well established. Arican et al,8 Kyriakou et al,9 and Xuan et al10 previously reported a lack of any correlation between TNF-α and IL-17A in the pathogenesis of psoriasis; however, we observed a positive correlation between TNF-α and IL-17A in our study. This finding may be due to the abundant TNF-α production by myeloid dendritic cells involved in the transformation of naive T lymphocytes into IL-17A–secreting Th17 lymphocytes, which can also secrete TNF-α.
After the molecular cloning of SDCs by Saunders et al11 in 1989, SDCs gained attention and have been the focus of many studies for their part in the pathogenesis of conditions such as inflammatory diseases, carcinogenesis, infections, sepsis, and trauma.6,12 Among the inflammatory diseases sharing similar pathogenetic features to psoriasis, serum SDC4 levels are found to be elevated in rheumatoid arthritis and are correlated with disease activity.13 Cekic et al14 reported that serum SDC1 levels were significantly higher in patients with Crohn disease than controls (P=.03). Additionally, serum SDC1 levels were higher in patients with active disease compared with those who were in remission. Correlations between SDC1 and disease severity and C-reactive protein also have been found.14 Serum SDC-1 levels found to be elevated in patients with systemic lupus erythematosus were compared to the controls and were correlated with disease activity.15 Nakao et al16 reported that the serum SDC4 levels were significantly higher in patients with atopic dermatitis compared to controls (P<.01); further, SDC4 levels were correlated with severity of the disease.
Jaiswal et al17 reported that SDC1 is abundant on the surface of IL-17A–secreting γδ T lymphocytes (Tγδ17), whose contribution to psoriasis pathogenesis is known. When subjected to treatment with imiquimod, SDC1-suppressed mice displayed increased psoriasiform dermatitis compared with wild-type counterparts. The authors stated that SDC1 may play a role in controlling homeostasis of Tγδ17
In a study examining changes in the ECM in patients with psoriasis, it was observed that the expression of
A study conducted by Koliakou et al20 showed that, in healthy skin, SDC1 was expressed in almost the full thickness of the epidermis, but lowest expression was in the basal-layer keratinocytes. In a psoriatic epidermis, unlike the normal epidermis, SDC1 was found to be more intensely expressed in the keratinocytes of the basal layer, where keratinocyte proliferation occurs. In this study, SDC4 was expressed mainly at lower levels in a healthy epidermis, especially in the spinous and the basal layers. In a psoriatic epidermis, SDC4 was absent from all the layers. In the same study, gelatin-based carriers containing anti–TNF-α and anti–IL-17A were applied to a full-thickness epidermis with psoriatic lesions, after which SDC1 expression was observed to decrease almost completely in the psoriatic epidermis; there was no change in SDC4 expression, which also was not seen in the psoriatic epidermis. The authors claimed the application of these gelatin-based carriers could be a possible treatment modality for psoriasis, and the study provides evidence for the involvement of SDC1 and/or SDC4 in the pathogenesis of psoriasis
Limitations of the current study include small sample size, lack of longitudinal data, lack of tissue testing of these molecules, and lack of external validation.
Conclusion
Overall, research has shown that SDCs play important roles in inflammatory processes, and more widespread inflammation has been associated with increased shedding of these molecules into the ECM and higher serum levels. In our study, serum SDC1, SDC4, and IL-17A levels were increased in patients with psoriasis compared to the healthy controls. A logistic regression analysis indicated that high serum SDC1 levels may be an independent risk factor for development of psoriasis. The increase in serum SDC1 and SDC4 levels and the positive correlation between SDC1 levels and disease severity observed in our study strongly implicate SDCs in the inflammatory disease psoriasis. The precise role of SDCs in the pathogenesis of psoriasis and the implications of targeting these molecules are the subject of more in-depth studies in the future.
Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
Uings IJ, Farrow SN. Cell receptors and cell signaling. Mol Pathol. 2000;53:295-299.
Kirkpatrick CA, Selleck SB. Heparan sulfate proteoglycans at a glance.J Cell Sci. 2007;120:1829-1832.
Stepp MA, Pal-Ghosh S, Tadvalkar G, et al. Syndecan-1 and its expanding list of contacts. Adv Wound Care (New Rochelle). 2015;4:235-249.
Rangarajan S, Richter JR, Richter RP, et al. Heparanase-enhanced shedding of syndecan-1 and its role in driving disease pathogenesis and progression. J Histochem Cytochem. 2020;68:823-840.
Gopal S, Arokiasamy S, Pataki C, et al. Syndecan receptors: pericellular regulators in development and inflammatory disease. Open Biol. 2021;11:200377.
Bertrand J, Bollmann M. Soluble syndecans: biomarkers for diseases and therapeutic options. Br J Pharmacol. 2019;176:67-81.
Arican O, Aral M, Sasmaz S, et al. Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm. 2005;2005:273-279.
Kyriakou A, Patsatsi A, Vyzantiadis TA, et al. Serum levels of TNF-α, IL12/23 p40, and IL-17 in psoriatic patients with and without nail psoriasis: a cross-sectional study. ScientificWorldJournal. 2014;2014:508178.
Xuan ML, Lu CJ, Han L, et al. Circulating levels of inflammatory cytokines in patients with psoriasis vulgaris of different Chinese medicine syndromes. Chin J Integr Med. 2015;21:108-114.
Saunders S, Jalkanen M, O’Farrell S, et al. Molecular cloning of syndecan, an integral membrane proteoglycan. J Cell Biol. 1989;108:1547-1556.
Manon-Jensen T, Itoh Y, Couchman JR. Proteoglycans in health and disease: the multiple roles of syndecan shedding. FEBS J. 2010;277:3876-3889.
Zhao J, Ye X, Zhang Z. Syndecan-4 is correlated with disease activity and serological characteristic of rheumatoid arthritis. Adv Rheumatol. 2022;62:21.
Cekic C, Kırcı A, Vatansever S, et al. Serum syndecan-1 levels and its relationship to disease activity in patients with Crohn’s disease. Gastroenterol Res Pract. 2015;2015:850351.
Minowa K, Amano H, Nakano S, et al. Elevated serum level of circulating syndecan-1 (CD138) in active systemic lupus erythematosus. Autoimmunity. 2011;44:357-362.
Nakao M, Sugaya M, Takahashi N, et al. Increased syndecan-4 expression in sera and skin of patients with atopic dermatitis. Arch Dermatol Res. 2016;308:655-660.
Jaiswal AK, Sadasivam M, Archer NK, et al. Syndecan-1 regulates psoriasiform dermatitis by controlling homeostasis of IL-17-producing γδ T cells. J Immunol. 2018;201:1651-1661
Wagner MFMG, Theodoro TR, Filho CASM, et al. Extracellular matrix alterations in the skin of patients affected by psoriasis. BMC Mol Cell Biol. 2021;22:55.
Peters F, Rahn S, Mengel M, et al. Syndecan-1 shedding by meprin β impairs keratinocyte adhesion and differentiation in hyperkeratosis. Matrix Biol. 2021;102:37-69.
Koliakou E, Eleni MM, Koumentakou I, et al. Altered distribution and expression of syndecan-1 and -4 as an additional hallmark in psoriasis. Int J Mol Sci. 2022;23:6511.
Doss RW, El-Rifaie AA, Said AN, et al. Cutaneous syndecan-1 expression before and after phototherapy in psoriasis. Indian J Dermatol Venereol Leprol. 2020;86:439-440.
Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
Uings IJ, Farrow SN. Cell receptors and cell signaling. Mol Pathol. 2000;53:295-299.
Kirkpatrick CA, Selleck SB. Heparan sulfate proteoglycans at a glance.J Cell Sci. 2007;120:1829-1832.
Stepp MA, Pal-Ghosh S, Tadvalkar G, et al. Syndecan-1 and its expanding list of contacts. Adv Wound Care (New Rochelle). 2015;4:235-249.
Rangarajan S, Richter JR, Richter RP, et al. Heparanase-enhanced shedding of syndecan-1 and its role in driving disease pathogenesis and progression. J Histochem Cytochem. 2020;68:823-840.
Gopal S, Arokiasamy S, Pataki C, et al. Syndecan receptors: pericellular regulators in development and inflammatory disease. Open Biol. 2021;11:200377.
Bertrand J, Bollmann M. Soluble syndecans: biomarkers for diseases and therapeutic options. Br J Pharmacol. 2019;176:67-81.
Arican O, Aral M, Sasmaz S, et al. Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm. 2005;2005:273-279.
Kyriakou A, Patsatsi A, Vyzantiadis TA, et al. Serum levels of TNF-α, IL12/23 p40, and IL-17 in psoriatic patients with and without nail psoriasis: a cross-sectional study. ScientificWorldJournal. 2014;2014:508178.
Xuan ML, Lu CJ, Han L, et al. Circulating levels of inflammatory cytokines in patients with psoriasis vulgaris of different Chinese medicine syndromes. Chin J Integr Med. 2015;21:108-114.
Saunders S, Jalkanen M, O’Farrell S, et al. Molecular cloning of syndecan, an integral membrane proteoglycan. J Cell Biol. 1989;108:1547-1556.
Manon-Jensen T, Itoh Y, Couchman JR. Proteoglycans in health and disease: the multiple roles of syndecan shedding. FEBS J. 2010;277:3876-3889.
Zhao J, Ye X, Zhang Z. Syndecan-4 is correlated with disease activity and serological characteristic of rheumatoid arthritis. Adv Rheumatol. 2022;62:21.
Cekic C, Kırcı A, Vatansever S, et al. Serum syndecan-1 levels and its relationship to disease activity in patients with Crohn’s disease. Gastroenterol Res Pract. 2015;2015:850351.
Minowa K, Amano H, Nakano S, et al. Elevated serum level of circulating syndecan-1 (CD138) in active systemic lupus erythematosus. Autoimmunity. 2011;44:357-362.
Nakao M, Sugaya M, Takahashi N, et al. Increased syndecan-4 expression in sera and skin of patients with atopic dermatitis. Arch Dermatol Res. 2016;308:655-660.
Jaiswal AK, Sadasivam M, Archer NK, et al. Syndecan-1 regulates psoriasiform dermatitis by controlling homeostasis of IL-17-producing γδ T cells. J Immunol. 2018;201:1651-1661
Wagner MFMG, Theodoro TR, Filho CASM, et al. Extracellular matrix alterations in the skin of patients affected by psoriasis. BMC Mol Cell Biol. 2021;22:55.
Peters F, Rahn S, Mengel M, et al. Syndecan-1 shedding by meprin β impairs keratinocyte adhesion and differentiation in hyperkeratosis. Matrix Biol. 2021;102:37-69.
Koliakou E, Eleni MM, Koumentakou I, et al. Altered distribution and expression of syndecan-1 and -4 as an additional hallmark in psoriasis. Int J Mol Sci. 2022;23:6511.
Doss RW, El-Rifaie AA, Said AN, et al. Cutaneous syndecan-1 expression before and after phototherapy in psoriasis. Indian J Dermatol Venereol Leprol. 2020;86:439-440.
Pathogenic Significance of Serum Syndecan-1 and Syndecan-4 in Psoriasis
Pathogenic Significance of Serum Syndecan-1 and Syndecan-4 in Psoriasis
PRACTICE POINTS
- Improved understanding of psoriasis pathogenesis has enabled the development of targeted treatments, although the mediators driving the disease have not yet been fully identified.
- Based on the findings of this study and existing literature, we suggest that syndecan-1 and syndecan-4 may play a role in the pathogenesis of psoriasis; however, further studies are needed to elucidate their precise mechanisms of action.
Acute Generalized Exanthematous Pustulosis Secondary to Application of Tapinarof Cream 1%
Acute Generalized Exanthematous Pustulosis Secondary to Application of Tapinarof Cream 1%
To the Editor:
For many years, topical treatment of plaque psoriasis was limited to steroids, calcineurin inhibitors, vitamin D analogs, retinoids, coal tar products, and anthralin. In recent years, 2 new nonsteroidal treatment options with alternative mechanisms of action, roflumilast 0.3% and tapinarof 1%, have been approved by the US Food and Drug Administration.1 Roflumilast 0.3%, a topical phosphodiesterase 4 inhibitor, was shown in phase 3 clinical trials to reach an Investigator Global Assessment response of 37.5% to 42.2% in 8 weeks using once-daily application with minimal cutaneous adverse effects.1 Furthermore, it has demonstrated efficacy in treating psoriasis in intertriginous areas in subset analyses.1 Tapinarof is an aryl hydrocarbon receptor agonist that suppresses Th17 cell differentiation by downregulating IL-17, IL-22, and IL-23.1 In phase 3 clinical trials, 35% to 40% of patients who used tapinarof cream 1% once daily demonstrated improvement in psoriasis compared with 6% who used the vehicle alone.2 In these studies, 18% to 24% of patients who used tapinarof cream 1% experienced folliculitis.2
Acute generalized exanthematous pustulosis (AGEP) is a nonfollicular pustular drug reaction with systemic symptoms that typically occurs within 2 weeks of exposure to an inciting medication. Systemic antibiotics are the most commonly reported cause of AGEP.3 There are few reports in the literature of AGEP induced by topical agents.4,5 We report a case of AGEP in a young man following the use of tapinarof cream 1%.
A 23-year-old man with a history of psoriasis presented to the emergency department with fever and a pustular rash. One week prior to presentation, he developed a pustular eruption around plaques of psoriasis on the arms and legs. The patient had been prescribed tapinarof cream 1% by an outside dermatologist and was applying the medication to the affected areas once daily for 1 month prior to onset of symptoms. He discontinued tapinarof a few days prior to the eruption starting, but the rash progressed centrifugally and was associated with fevers and fatigue despite treatment with a brief course of empiric cephalexin prescribed by his primary care provider.
At presentation to our institution, the patient had widespread erythematous patches studded with pustules located on the arms, legs, and flexural areas as well as plaques of psoriasis involving approximately 20% of the body surface area (Figure 1). Furthermore, the patient was noted to have large noninflammatory bullae along the legs. The new eruption occurred on areas that were both treated and spared from the tapinarof cream 1%. Laboratory evaluation showed neutrophil-predominant leukocytosis (white blood cell count, 15.9×103/µL [reference range, 4.0-11.0×103/µL]; absolute neutrophil count, 10.3×103/µL [reference range, 1.5-8.0×103/µL]), absolute eosinophilia (1930/µL [reference range, 0-0.5×103/µL]), hypocalcemia (8.4 mg/dL [reference range, 8.5-10.5 mg/dL]), and a mild transaminitis (aspartate aminotransferase, 37 IU/L [reference range, 10-40 IU/L]; alanine aminotransferase, 53 IU/L [reference range, 7-56 U/L]). Histopathology demonstrated spongiosis with subcorneal and intraepidermal pustules and mixed dermal inflammation containing eosinophils (Figure 2). Direct immunofluorescence revealed mild granular staining of C3 at the basement membrane zone.
The patient was started on 1 mg/kg/d of prednisone tapered over 20 days, and he rapidly improved. Alanine aminotransferase levels peaked at 120 IU/L 2 weeks later. At that time, he had complete resolution of the original eruption and was transitioned to topical steroids for continued management of the psoriasis (Figure 3).
The differential diagnosis for our patient included AGEP, generalized pustular psoriasis (GPP), miliaria pustulosa, generalized cutaneous candidiasis, exuberant allergic contact dermatitis (ACD), and linear IgA bullous dermatosis (LABD). Based on the clinical manifestations, laboratory results, and histopathologic evaluation, we made the diagnosis of AGEP secondary to tapinarof with systemic absorption. Acute generalized exanthematous pustulosis has been reported with topical use of morphine and diphenhydramine, among other agents.4,5 To our knowledge, AGEP due to tapinarof cream 1% has not been reported. In the original clinical trials of tapinarof, folliculitis was contained to sites of application.2 Our patient developed pustules at sites distant to areas of application, as well as systemic symptoms and laboratory abnormalities, indicating a systemic reaction. It can be difficult to distinguish AGEP clinically and histologically from GPP. Both conditions can manifest with fever, hypocalcemia, and sterile pustules on a background of erythema that favors intertriginous areas.6 Infection, rapid oral steroid withdrawal, pregnancy, and rarely oral medications have been reported causes of GPP.6 Our patient did not have any of these exposures. There is overlap in the histology of AGEP and GPP. One retrospective series compared histologic samples to help distinguish these 2 entities. Reliable markers that favored AGEP over GPP included eosinophilic spongiosis, interface dermatitis, and dermal eosinophilia (>2/mm2).7 In contrast, the presence of CD161 positivity in the dermis with at least 10 cells favored a diagnosis of GPP.7 In our case, the presence of spongiosis with eosinophils in the dermis favored a diagnosis of AGEP over GPP.
Miliaria pustulosa is a benign condition caused by the occlusion of the epidermal portion of eccrine glands related to either high fever or hot and humid environmental conditions. While it can be present in intertriginous areas like AGEP, miliaria pustulosa can be seen extensively on the back, most commonly in immobile hospitalized patients.8 Generalized cutaneous candidiasis usually is caused by the yeast Candida albicans and can take on multiple morphologies, including folliculitis.9 The eruption may be disseminated but often is accentuated in intertriginous areas and the anogenital folds. Predisposing factors include immunosuppression, endocrinopathies, recent use of systemic antibiotics or steroids, chemotherapy, and indwelling catheters.9 Outside of recent antibiotic use, our patient did not have any risk factors for miliaria pustulosa, making this diagnosis unlikely.
Given the presence of overlapping bullae along the lower extremities, an exuberant ACD and LABD were considered. Bullae formation can occur in ACD secondary to robust inflammation and edema leading to acantholysis.10 While a delayed hypersensitivity reaction to topical tapinarof cream 1% was considered given that the patient used the medication for approximately 1 month prior to the onset of symptoms, it would be unlikely for ACD to present with a concomitant pustular eruption. Linear IgA bullous dermatosis is an autoimmune blistering disease in which antibodies target bullous pemphigoid antigen 2, and there is characteristically linear deposition of IgA at the dermal-epidermal junction that leads to subepidermal blistering.11 This often manifests clinically as widespread tense vesicles in an annular or string-of-pearls appearance. However, morphologies can vary, and large bullae may be seen. In adults, LABD typically is associated with inflammatory bowel disease, malignancy, or medications, notably vancomycin.11,12 Our patient did not have any of these predisposing factors, and his biopsy for direct immunofluorescence did not reveal the classic pattern described above.
Interestingly, there have been reports in the literature of bullous AGEP in the setting of oral anti-infectives. One report described a 62-year-old woman who developed widespread nonfollicular pustules with multiple tense serous blisters 24 hours after taking oral terbinafine.13 Another case described an 80-year-old woman with a similar presentation following a course of ciprofloxacin (although the timeline of medication administration was not described).14 In this case, patch testing to the culprit medication reproduced the response.14 In both cases, a biopsy revealed subcorneal and intraepidermal pustules with marked dermal edema.13,14 As previously described, spongiosis is a common feature of AGEP. We hypothesize that, similar to these reports, our patient had a robust inflammatory response leading to spongiosis, acantholysis, and blister formation secondary to AGEP.
Dermatologists should be aware of this case of AGEP secondary to tapinarof cream 1%, as reports in the literature are rare and it is a reminder that topical medications can cause serious systemic reactions.
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Szatkowski J, Schwartz RA. Acute generalized exanthematous pustulosis (AGEP): a review and update. J Am Acad Dermatol. 2015;73:843-848. doi:10.1016/j.jaad.2015.07.017
- Ghazawi FM, Colantonio S, Bradshaw S, et al. Acute generalized exanthematous pustulosis induced by topical morphine and confirmed by patch testing. Dermat Contact Atopic Occup Drug. 2020;31:E22-E23. doi:10.1097/DER.0000000000000573
- Hanafusa T, Igawa K, Azukizawa H, et al. Acute generalized exanthematous pustulosis induced by topical diphenhydramine. Eur J Dermatol. 2011;21:994-995. doi:10.1684/ejd.2011.1500
- Reynolds KA, Pithadia DJ, Lee EB, et al. Generalized pustular psoriasis: a review of the pathophysiology, clinical manifestations,diagnosis, and treatment. Cutis. 2022;110:19-25. doi:10.12788/cutis.0579
- Isom J, Braswell DS, Siroy A, et al. Clinical and histopathologic features differentiating acute generalized exanthematous pustulosis and pustular psoriasis: a retrospective series. J Am Acad Dermatol. 2020;83:265-267. doi:10.1016/j.jaad.2020.03.015
- Fealey RD, Hebert AA. Disorders of the eccrine sweat glands and sweating. In: Goldsmith LA, Katz SI, Gilchrest BA, et al, eds. Fitzpatrick’s Dermatology in General Medicine.8th ed. McGraw-Hill; 2012:946.
- Elewski BE, Hughey LC, Marchiony Hunt K, et al. Fungal diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:1329-1363.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192. doi:10.1016/j.clindermatol.2019.10.003
- Hull CM, Zone JZ. Dermatitis herpetiforms and linear IgA bullous dermatosis. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:527-537.
- Yamagami J, Nakamura Y, Nagao K, et al. Vancomycin mediates IgA autoreactivity in drug-induced linear IgA bullous dermatosis. J Invest Dermatol. 2018;138:1473-1480.
- Bullous acute generalized exanthematous pustulosis due to oral terbinafine. J Am Acad Dermatol. 2005;52:P115. doi:10.1016/j.jaad.2004.10.468
- Hausermann P, Scherer K, Weber M, et al. Ciprofloxacin-induced acute generalized exanthematous pustulosis mimicking bullous drug eruption confirmed by a positive patch test. Dermatology. 2005;211:277-280. doi:10.1159/000087024
To the Editor:
For many years, topical treatment of plaque psoriasis was limited to steroids, calcineurin inhibitors, vitamin D analogs, retinoids, coal tar products, and anthralin. In recent years, 2 new nonsteroidal treatment options with alternative mechanisms of action, roflumilast 0.3% and tapinarof 1%, have been approved by the US Food and Drug Administration.1 Roflumilast 0.3%, a topical phosphodiesterase 4 inhibitor, was shown in phase 3 clinical trials to reach an Investigator Global Assessment response of 37.5% to 42.2% in 8 weeks using once-daily application with minimal cutaneous adverse effects.1 Furthermore, it has demonstrated efficacy in treating psoriasis in intertriginous areas in subset analyses.1 Tapinarof is an aryl hydrocarbon receptor agonist that suppresses Th17 cell differentiation by downregulating IL-17, IL-22, and IL-23.1 In phase 3 clinical trials, 35% to 40% of patients who used tapinarof cream 1% once daily demonstrated improvement in psoriasis compared with 6% who used the vehicle alone.2 In these studies, 18% to 24% of patients who used tapinarof cream 1% experienced folliculitis.2
Acute generalized exanthematous pustulosis (AGEP) is a nonfollicular pustular drug reaction with systemic symptoms that typically occurs within 2 weeks of exposure to an inciting medication. Systemic antibiotics are the most commonly reported cause of AGEP.3 There are few reports in the literature of AGEP induced by topical agents.4,5 We report a case of AGEP in a young man following the use of tapinarof cream 1%.
A 23-year-old man with a history of psoriasis presented to the emergency department with fever and a pustular rash. One week prior to presentation, he developed a pustular eruption around plaques of psoriasis on the arms and legs. The patient had been prescribed tapinarof cream 1% by an outside dermatologist and was applying the medication to the affected areas once daily for 1 month prior to onset of symptoms. He discontinued tapinarof a few days prior to the eruption starting, but the rash progressed centrifugally and was associated with fevers and fatigue despite treatment with a brief course of empiric cephalexin prescribed by his primary care provider.
At presentation to our institution, the patient had widespread erythematous patches studded with pustules located on the arms, legs, and flexural areas as well as plaques of psoriasis involving approximately 20% of the body surface area (Figure 1). Furthermore, the patient was noted to have large noninflammatory bullae along the legs. The new eruption occurred on areas that were both treated and spared from the tapinarof cream 1%. Laboratory evaluation showed neutrophil-predominant leukocytosis (white blood cell count, 15.9×103/µL [reference range, 4.0-11.0×103/µL]; absolute neutrophil count, 10.3×103/µL [reference range, 1.5-8.0×103/µL]), absolute eosinophilia (1930/µL [reference range, 0-0.5×103/µL]), hypocalcemia (8.4 mg/dL [reference range, 8.5-10.5 mg/dL]), and a mild transaminitis (aspartate aminotransferase, 37 IU/L [reference range, 10-40 IU/L]; alanine aminotransferase, 53 IU/L [reference range, 7-56 U/L]). Histopathology demonstrated spongiosis with subcorneal and intraepidermal pustules and mixed dermal inflammation containing eosinophils (Figure 2). Direct immunofluorescence revealed mild granular staining of C3 at the basement membrane zone.


The patient was started on 1 mg/kg/d of prednisone tapered over 20 days, and he rapidly improved. Alanine aminotransferase levels peaked at 120 IU/L 2 weeks later. At that time, he had complete resolution of the original eruption and was transitioned to topical steroids for continued management of the psoriasis (Figure 3).

The differential diagnosis for our patient included AGEP, generalized pustular psoriasis (GPP), miliaria pustulosa, generalized cutaneous candidiasis, exuberant allergic contact dermatitis (ACD), and linear IgA bullous dermatosis (LABD). Based on the clinical manifestations, laboratory results, and histopathologic evaluation, we made the diagnosis of AGEP secondary to tapinarof with systemic absorption. Acute generalized exanthematous pustulosis has been reported with topical use of morphine and diphenhydramine, among other agents.4,5 To our knowledge, AGEP due to tapinarof cream 1% has not been reported. In the original clinical trials of tapinarof, folliculitis was contained to sites of application.2 Our patient developed pustules at sites distant to areas of application, as well as systemic symptoms and laboratory abnormalities, indicating a systemic reaction. It can be difficult to distinguish AGEP clinically and histologically from GPP. Both conditions can manifest with fever, hypocalcemia, and sterile pustules on a background of erythema that favors intertriginous areas.6 Infection, rapid oral steroid withdrawal, pregnancy, and rarely oral medications have been reported causes of GPP.6 Our patient did not have any of these exposures. There is overlap in the histology of AGEP and GPP. One retrospective series compared histologic samples to help distinguish these 2 entities. Reliable markers that favored AGEP over GPP included eosinophilic spongiosis, interface dermatitis, and dermal eosinophilia (>2/mm2).7 In contrast, the presence of CD161 positivity in the dermis with at least 10 cells favored a diagnosis of GPP.7 In our case, the presence of spongiosis with eosinophils in the dermis favored a diagnosis of AGEP over GPP.
Miliaria pustulosa is a benign condition caused by the occlusion of the epidermal portion of eccrine glands related to either high fever or hot and humid environmental conditions. While it can be present in intertriginous areas like AGEP, miliaria pustulosa can be seen extensively on the back, most commonly in immobile hospitalized patients.8 Generalized cutaneous candidiasis usually is caused by the yeast Candida albicans and can take on multiple morphologies, including folliculitis.9 The eruption may be disseminated but often is accentuated in intertriginous areas and the anogenital folds. Predisposing factors include immunosuppression, endocrinopathies, recent use of systemic antibiotics or steroids, chemotherapy, and indwelling catheters.9 Outside of recent antibiotic use, our patient did not have any risk factors for miliaria pustulosa, making this diagnosis unlikely.
Given the presence of overlapping bullae along the lower extremities, an exuberant ACD and LABD were considered. Bullae formation can occur in ACD secondary to robust inflammation and edema leading to acantholysis.10 While a delayed hypersensitivity reaction to topical tapinarof cream 1% was considered given that the patient used the medication for approximately 1 month prior to the onset of symptoms, it would be unlikely for ACD to present with a concomitant pustular eruption. Linear IgA bullous dermatosis is an autoimmune blistering disease in which antibodies target bullous pemphigoid antigen 2, and there is characteristically linear deposition of IgA at the dermal-epidermal junction that leads to subepidermal blistering.11 This often manifests clinically as widespread tense vesicles in an annular or string-of-pearls appearance. However, morphologies can vary, and large bullae may be seen. In adults, LABD typically is associated with inflammatory bowel disease, malignancy, or medications, notably vancomycin.11,12 Our patient did not have any of these predisposing factors, and his biopsy for direct immunofluorescence did not reveal the classic pattern described above.
Interestingly, there have been reports in the literature of bullous AGEP in the setting of oral anti-infectives. One report described a 62-year-old woman who developed widespread nonfollicular pustules with multiple tense serous blisters 24 hours after taking oral terbinafine.13 Another case described an 80-year-old woman with a similar presentation following a course of ciprofloxacin (although the timeline of medication administration was not described).14 In this case, patch testing to the culprit medication reproduced the response.14 In both cases, a biopsy revealed subcorneal and intraepidermal pustules with marked dermal edema.13,14 As previously described, spongiosis is a common feature of AGEP. We hypothesize that, similar to these reports, our patient had a robust inflammatory response leading to spongiosis, acantholysis, and blister formation secondary to AGEP.
Dermatologists should be aware of this case of AGEP secondary to tapinarof cream 1%, as reports in the literature are rare and it is a reminder that topical medications can cause serious systemic reactions.
To the Editor:
For many years, topical treatment of plaque psoriasis was limited to steroids, calcineurin inhibitors, vitamin D analogs, retinoids, coal tar products, and anthralin. In recent years, 2 new nonsteroidal treatment options with alternative mechanisms of action, roflumilast 0.3% and tapinarof 1%, have been approved by the US Food and Drug Administration.1 Roflumilast 0.3%, a topical phosphodiesterase 4 inhibitor, was shown in phase 3 clinical trials to reach an Investigator Global Assessment response of 37.5% to 42.2% in 8 weeks using once-daily application with minimal cutaneous adverse effects.1 Furthermore, it has demonstrated efficacy in treating psoriasis in intertriginous areas in subset analyses.1 Tapinarof is an aryl hydrocarbon receptor agonist that suppresses Th17 cell differentiation by downregulating IL-17, IL-22, and IL-23.1 In phase 3 clinical trials, 35% to 40% of patients who used tapinarof cream 1% once daily demonstrated improvement in psoriasis compared with 6% who used the vehicle alone.2 In these studies, 18% to 24% of patients who used tapinarof cream 1% experienced folliculitis.2
Acute generalized exanthematous pustulosis (AGEP) is a nonfollicular pustular drug reaction with systemic symptoms that typically occurs within 2 weeks of exposure to an inciting medication. Systemic antibiotics are the most commonly reported cause of AGEP.3 There are few reports in the literature of AGEP induced by topical agents.4,5 We report a case of AGEP in a young man following the use of tapinarof cream 1%.
A 23-year-old man with a history of psoriasis presented to the emergency department with fever and a pustular rash. One week prior to presentation, he developed a pustular eruption around plaques of psoriasis on the arms and legs. The patient had been prescribed tapinarof cream 1% by an outside dermatologist and was applying the medication to the affected areas once daily for 1 month prior to onset of symptoms. He discontinued tapinarof a few days prior to the eruption starting, but the rash progressed centrifugally and was associated with fevers and fatigue despite treatment with a brief course of empiric cephalexin prescribed by his primary care provider.
At presentation to our institution, the patient had widespread erythematous patches studded with pustules located on the arms, legs, and flexural areas as well as plaques of psoriasis involving approximately 20% of the body surface area (Figure 1). Furthermore, the patient was noted to have large noninflammatory bullae along the legs. The new eruption occurred on areas that were both treated and spared from the tapinarof cream 1%. Laboratory evaluation showed neutrophil-predominant leukocytosis (white blood cell count, 15.9×103/µL [reference range, 4.0-11.0×103/µL]; absolute neutrophil count, 10.3×103/µL [reference range, 1.5-8.0×103/µL]), absolute eosinophilia (1930/µL [reference range, 0-0.5×103/µL]), hypocalcemia (8.4 mg/dL [reference range, 8.5-10.5 mg/dL]), and a mild transaminitis (aspartate aminotransferase, 37 IU/L [reference range, 10-40 IU/L]; alanine aminotransferase, 53 IU/L [reference range, 7-56 U/L]). Histopathology demonstrated spongiosis with subcorneal and intraepidermal pustules and mixed dermal inflammation containing eosinophils (Figure 2). Direct immunofluorescence revealed mild granular staining of C3 at the basement membrane zone.


The patient was started on 1 mg/kg/d of prednisone tapered over 20 days, and he rapidly improved. Alanine aminotransferase levels peaked at 120 IU/L 2 weeks later. At that time, he had complete resolution of the original eruption and was transitioned to topical steroids for continued management of the psoriasis (Figure 3).

The differential diagnosis for our patient included AGEP, generalized pustular psoriasis (GPP), miliaria pustulosa, generalized cutaneous candidiasis, exuberant allergic contact dermatitis (ACD), and linear IgA bullous dermatosis (LABD). Based on the clinical manifestations, laboratory results, and histopathologic evaluation, we made the diagnosis of AGEP secondary to tapinarof with systemic absorption. Acute generalized exanthematous pustulosis has been reported with topical use of morphine and diphenhydramine, among other agents.4,5 To our knowledge, AGEP due to tapinarof cream 1% has not been reported. In the original clinical trials of tapinarof, folliculitis was contained to sites of application.2 Our patient developed pustules at sites distant to areas of application, as well as systemic symptoms and laboratory abnormalities, indicating a systemic reaction. It can be difficult to distinguish AGEP clinically and histologically from GPP. Both conditions can manifest with fever, hypocalcemia, and sterile pustules on a background of erythema that favors intertriginous areas.6 Infection, rapid oral steroid withdrawal, pregnancy, and rarely oral medications have been reported causes of GPP.6 Our patient did not have any of these exposures. There is overlap in the histology of AGEP and GPP. One retrospective series compared histologic samples to help distinguish these 2 entities. Reliable markers that favored AGEP over GPP included eosinophilic spongiosis, interface dermatitis, and dermal eosinophilia (>2/mm2).7 In contrast, the presence of CD161 positivity in the dermis with at least 10 cells favored a diagnosis of GPP.7 In our case, the presence of spongiosis with eosinophils in the dermis favored a diagnosis of AGEP over GPP.
Miliaria pustulosa is a benign condition caused by the occlusion of the epidermal portion of eccrine glands related to either high fever or hot and humid environmental conditions. While it can be present in intertriginous areas like AGEP, miliaria pustulosa can be seen extensively on the back, most commonly in immobile hospitalized patients.8 Generalized cutaneous candidiasis usually is caused by the yeast Candida albicans and can take on multiple morphologies, including folliculitis.9 The eruption may be disseminated but often is accentuated in intertriginous areas and the anogenital folds. Predisposing factors include immunosuppression, endocrinopathies, recent use of systemic antibiotics or steroids, chemotherapy, and indwelling catheters.9 Outside of recent antibiotic use, our patient did not have any risk factors for miliaria pustulosa, making this diagnosis unlikely.
Given the presence of overlapping bullae along the lower extremities, an exuberant ACD and LABD were considered. Bullae formation can occur in ACD secondary to robust inflammation and edema leading to acantholysis.10 While a delayed hypersensitivity reaction to topical tapinarof cream 1% was considered given that the patient used the medication for approximately 1 month prior to the onset of symptoms, it would be unlikely for ACD to present with a concomitant pustular eruption. Linear IgA bullous dermatosis is an autoimmune blistering disease in which antibodies target bullous pemphigoid antigen 2, and there is characteristically linear deposition of IgA at the dermal-epidermal junction that leads to subepidermal blistering.11 This often manifests clinically as widespread tense vesicles in an annular or string-of-pearls appearance. However, morphologies can vary, and large bullae may be seen. In adults, LABD typically is associated with inflammatory bowel disease, malignancy, or medications, notably vancomycin.11,12 Our patient did not have any of these predisposing factors, and his biopsy for direct immunofluorescence did not reveal the classic pattern described above.
Interestingly, there have been reports in the literature of bullous AGEP in the setting of oral anti-infectives. One report described a 62-year-old woman who developed widespread nonfollicular pustules with multiple tense serous blisters 24 hours after taking oral terbinafine.13 Another case described an 80-year-old woman with a similar presentation following a course of ciprofloxacin (although the timeline of medication administration was not described).14 In this case, patch testing to the culprit medication reproduced the response.14 In both cases, a biopsy revealed subcorneal and intraepidermal pustules with marked dermal edema.13,14 As previously described, spongiosis is a common feature of AGEP. We hypothesize that, similar to these reports, our patient had a robust inflammatory response leading to spongiosis, acantholysis, and blister formation secondary to AGEP.
Dermatologists should be aware of this case of AGEP secondary to tapinarof cream 1%, as reports in the literature are rare and it is a reminder that topical medications can cause serious systemic reactions.
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Szatkowski J, Schwartz RA. Acute generalized exanthematous pustulosis (AGEP): a review and update. J Am Acad Dermatol. 2015;73:843-848. doi:10.1016/j.jaad.2015.07.017
- Ghazawi FM, Colantonio S, Bradshaw S, et al. Acute generalized exanthematous pustulosis induced by topical morphine and confirmed by patch testing. Dermat Contact Atopic Occup Drug. 2020;31:E22-E23. doi:10.1097/DER.0000000000000573
- Hanafusa T, Igawa K, Azukizawa H, et al. Acute generalized exanthematous pustulosis induced by topical diphenhydramine. Eur J Dermatol. 2011;21:994-995. doi:10.1684/ejd.2011.1500
- Reynolds KA, Pithadia DJ, Lee EB, et al. Generalized pustular psoriasis: a review of the pathophysiology, clinical manifestations,diagnosis, and treatment. Cutis. 2022;110:19-25. doi:10.12788/cutis.0579
- Isom J, Braswell DS, Siroy A, et al. Clinical and histopathologic features differentiating acute generalized exanthematous pustulosis and pustular psoriasis: a retrospective series. J Am Acad Dermatol. 2020;83:265-267. doi:10.1016/j.jaad.2020.03.015
- Fealey RD, Hebert AA. Disorders of the eccrine sweat glands and sweating. In: Goldsmith LA, Katz SI, Gilchrest BA, et al, eds. Fitzpatrick’s Dermatology in General Medicine.8th ed. McGraw-Hill; 2012:946.
- Elewski BE, Hughey LC, Marchiony Hunt K, et al. Fungal diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:1329-1363.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192. doi:10.1016/j.clindermatol.2019.10.003
- Hull CM, Zone JZ. Dermatitis herpetiforms and linear IgA bullous dermatosis. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:527-537.
- Yamagami J, Nakamura Y, Nagao K, et al. Vancomycin mediates IgA autoreactivity in drug-induced linear IgA bullous dermatosis. J Invest Dermatol. 2018;138:1473-1480.
- Bullous acute generalized exanthematous pustulosis due to oral terbinafine. J Am Acad Dermatol. 2005;52:P115. doi:10.1016/j.jaad.2004.10.468
- Hausermann P, Scherer K, Weber M, et al. Ciprofloxacin-induced acute generalized exanthematous pustulosis mimicking bullous drug eruption confirmed by a positive patch test. Dermatology. 2005;211:277-280. doi:10.1159/000087024
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Stein Gold L, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- Szatkowski J, Schwartz RA. Acute generalized exanthematous pustulosis (AGEP): a review and update. J Am Acad Dermatol. 2015;73:843-848. doi:10.1016/j.jaad.2015.07.017
- Ghazawi FM, Colantonio S, Bradshaw S, et al. Acute generalized exanthematous pustulosis induced by topical morphine and confirmed by patch testing. Dermat Contact Atopic Occup Drug. 2020;31:E22-E23. doi:10.1097/DER.0000000000000573
- Hanafusa T, Igawa K, Azukizawa H, et al. Acute generalized exanthematous pustulosis induced by topical diphenhydramine. Eur J Dermatol. 2011;21:994-995. doi:10.1684/ejd.2011.1500
- Reynolds KA, Pithadia DJ, Lee EB, et al. Generalized pustular psoriasis: a review of the pathophysiology, clinical manifestations,diagnosis, and treatment. Cutis. 2022;110:19-25. doi:10.12788/cutis.0579
- Isom J, Braswell DS, Siroy A, et al. Clinical and histopathologic features differentiating acute generalized exanthematous pustulosis and pustular psoriasis: a retrospective series. J Am Acad Dermatol. 2020;83:265-267. doi:10.1016/j.jaad.2020.03.015
- Fealey RD, Hebert AA. Disorders of the eccrine sweat glands and sweating. In: Goldsmith LA, Katz SI, Gilchrest BA, et al, eds. Fitzpatrick’s Dermatology in General Medicine.8th ed. McGraw-Hill; 2012:946.
- Elewski BE, Hughey LC, Marchiony Hunt K, et al. Fungal diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:1329-1363.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192. doi:10.1016/j.clindermatol.2019.10.003
- Hull CM, Zone JZ. Dermatitis herpetiforms and linear IgA bullous dermatosis. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017:527-537.
- Yamagami J, Nakamura Y, Nagao K, et al. Vancomycin mediates IgA autoreactivity in drug-induced linear IgA bullous dermatosis. J Invest Dermatol. 2018;138:1473-1480.
- Bullous acute generalized exanthematous pustulosis due to oral terbinafine. J Am Acad Dermatol. 2005;52:P115. doi:10.1016/j.jaad.2004.10.468
- Hausermann P, Scherer K, Weber M, et al. Ciprofloxacin-induced acute generalized exanthematous pustulosis mimicking bullous drug eruption confirmed by a positive patch test. Dermatology. 2005;211:277-280. doi:10.1159/000087024
Acute Generalized Exanthematous Pustulosis Secondary to Application of Tapinarof Cream 1%
Acute Generalized Exanthematous Pustulosis Secondary to Application of Tapinarof Cream 1%
PRACTICE POINTS
- Tapinarof cream 1% can be absorbed systemically and cause acute generalized exanthematous pustulosis (AGEP).
- Clinical configuration and histology can be useful to distinguish AGEP from mimickers.
- Topical application of drugs in general, particularly over large body surface areas, may lead to systemic drug eruptions.
Exploring the Relationship Between Psoriasis and Mobility Among US Adults
Exploring the Relationship Between Psoriasis and Mobility Among US Adults
To the Editor:
Psoriasis is a chronic inflammatory condition that affects individuals in various extracutaneous ways.1 Prior studies have documented a decrease in exercise intensity among patients with psoriasis2; however, few studies have specifically investigated baseline mobility in this population. Baseline mobility denotes an individual’s fundamental ability to walk or move around without assistance of any kind. Impaired mobility—when baseline mobility is compromised—is an aspect of the wider diversity, equity, and inclusion framework that underscores the significance of recognizing challenges and promoting inclusive measures, both at the point of care and in research.3 study sought to analyze the relationship between psoriasis and baseline mobility among US adults (aged 45 to 80 years) utilizing the latest data from the National Health and Nutrition Examination Survey (NHANES) database for psoriasis.4 We used three 2-year cycles of NHANES data to create a 2009-2014 dataset.
The overall NHANES response rate among adults aged 45 to 80 years between 2009 and 2014 was 67.9%. Patients were categorized as having impaired mobility if they responded “yes” to the following question: “Because of a health problem, do you have difficulty walking without using any special equipment?” Psoriasis status was assessed by the following question: “Have you ever been told by a doctor or other health professional that you had psoriasis?” Multivariable logistic regression analyses were performed using Stata/SE 18.0 software (StataCorp LLC) to assess the relationship between psoriasis and impaired mobility. Age, income, education, sex, race, tobacco use, diabetes status, body mass index, and arthritis status were controlled for in our models.
Our analysis initially included 9982 participants; 14 did not respond to questions assessing psoriasis and impaired mobility and were excluded. The prevalence of impaired mobility in patients with psoriasis was 17.1% compared with 10.9% among those without psoriasis (Table 1). There was a significant association between psoriasis and impaired mobility among patients aged 45 to 80 years after adjusting for potential confounding variables (adjusted odds ratio [AOR], 1.54; 95% CI, 1.04- 2.29; P=.032)(Table 2). Analyses of subgroups yielded no statistically significant results.
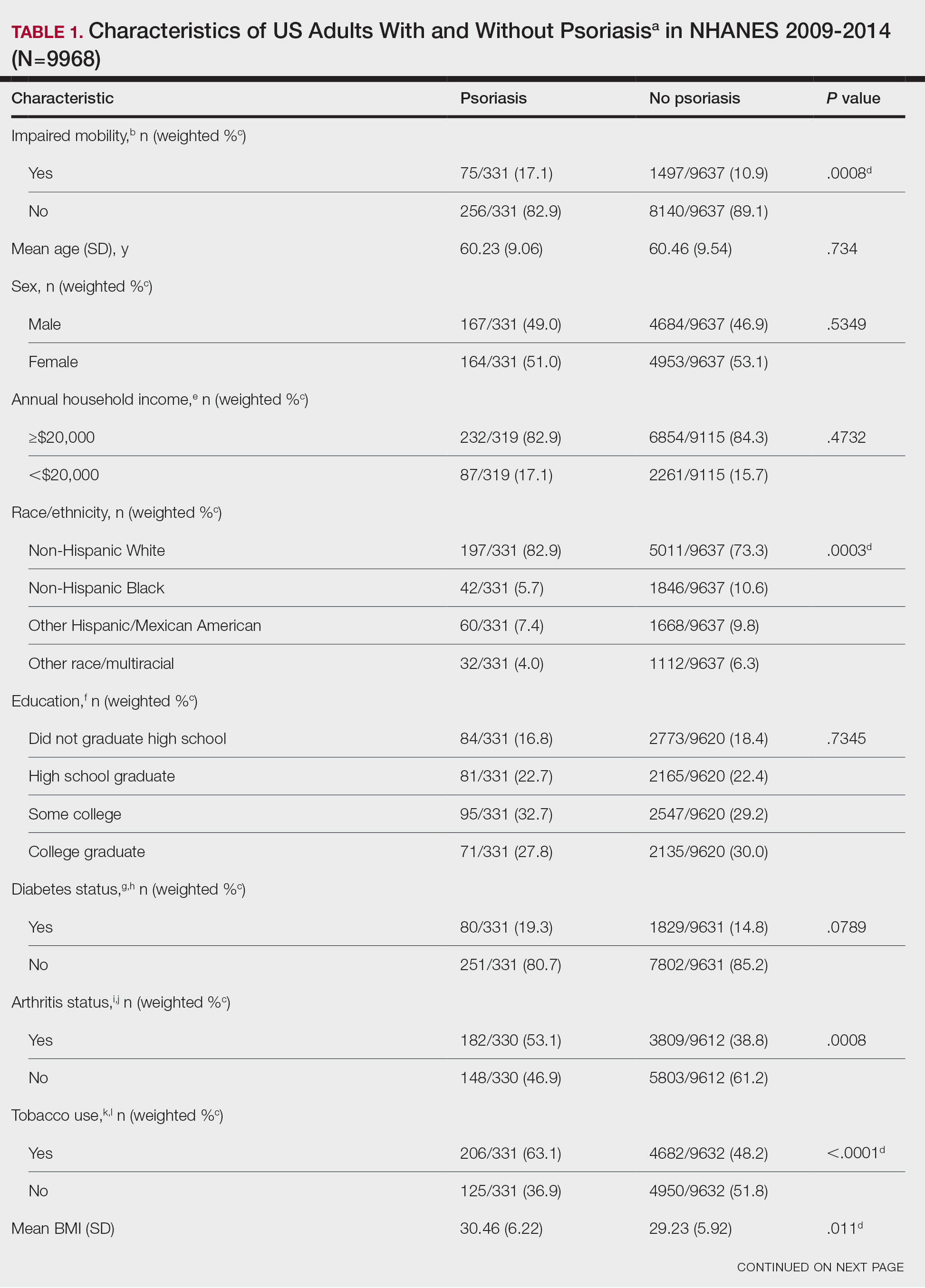

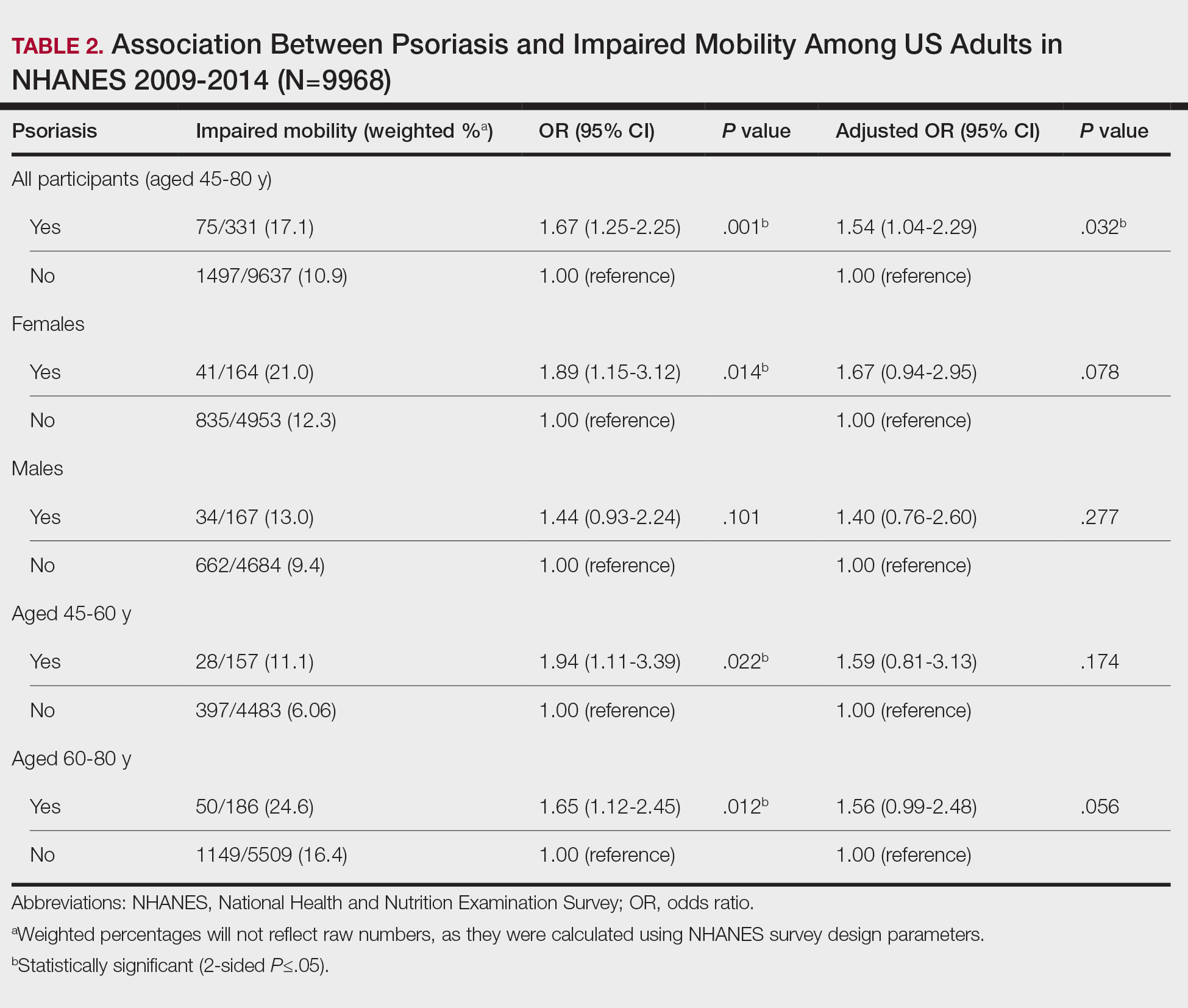
Our study demonstrated a statistically significant difference in mobility between individuals with psoriasis compared with the general population, which remained significant when controlling for arthritis, obesity, and diabetes (P=.032). This may be the result of several influences. First, the location of the psoriasis may impact mobility. Plantar psoriasis—a manifestation on the soles of the feet—can cause discomfort and pain, which can hinder walking and standing.5 Second, a study by Lasselin et al6 found that systemic inflammation contributes to mobility impairment through alterations in gait and posture, which suggests that the inflammatory processes inherent in psoriasis could intrinsically modify walking speed and stride, potentially exacerbating mobility difficulties independent of other comorbid conditions. These findings suggest that psoriasis may disproportionately affect individuals with impaired mobility, independent of comorbid arthritis, obesity, and diabetes.
These findings have broad implications for diversity, equity, and inclusion. They should prompt us to consider the practical challenges faced by this patient population and the ways that we can address barriers to care. Offering telehealth appointments, making primary care referrals for impaired mobility workups, and advising patients of direct-to-home delivery of prescriptions are good places to start.
Limitations to our study include the lack of specificity in the survey question, self-reporting bias, and the inability to control for the psoriasis location. Further investigations are warranted in large, representative US adult populations to assess the implications of impaired mobility in patients with psoriasis.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi: 10.1016/j.jaad.2018.11.058
- Zheng Q, Sun XY, Miao X, et al. Association between physical activity and risk of prevalent psoriasis: A MOOSE-compliant meta-analysis. Medicine (Baltimore). 2018;97:e11394. doi: 10.1097 /MD.0000000000011394
- Mullin AE, Coe IR, Gooden EA, et al. Inclusion, diversity, equity, and accessibility: from organizational responsibility to leadership competency. Healthc Manage Forum. 2021;34311-315. doi: 10.1177/08404704211038232
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed October 21, 2023. https://wwwn.cdc.gov/nchs/nhanes/
- Romani M, Biela G, Farr K, et al. Plantar psoriasis: a review of the literature. Clin Podiatr Med Surg. 2021;38:541-552. doi: 10.1016 /j.cpm.2021.06.009
- Lasselin J, Sundelin T, Wayne PM, et al. Biological motion during inflammation in humans. Brain Behav Immun. 2020;84:147-153. doi: 10.1016/j.bbi.2019.11.019
To the Editor:
Psoriasis is a chronic inflammatory condition that affects individuals in various extracutaneous ways.1 Prior studies have documented a decrease in exercise intensity among patients with psoriasis2; however, few studies have specifically investigated baseline mobility in this population. Baseline mobility denotes an individual’s fundamental ability to walk or move around without assistance of any kind. Impaired mobility—when baseline mobility is compromised—is an aspect of the wider diversity, equity, and inclusion framework that underscores the significance of recognizing challenges and promoting inclusive measures, both at the point of care and in research.3 study sought to analyze the relationship between psoriasis and baseline mobility among US adults (aged 45 to 80 years) utilizing the latest data from the National Health and Nutrition Examination Survey (NHANES) database for psoriasis.4 We used three 2-year cycles of NHANES data to create a 2009-2014 dataset.
The overall NHANES response rate among adults aged 45 to 80 years between 2009 and 2014 was 67.9%. Patients were categorized as having impaired mobility if they responded “yes” to the following question: “Because of a health problem, do you have difficulty walking without using any special equipment?” Psoriasis status was assessed by the following question: “Have you ever been told by a doctor or other health professional that you had psoriasis?” Multivariable logistic regression analyses were performed using Stata/SE 18.0 software (StataCorp LLC) to assess the relationship between psoriasis and impaired mobility. Age, income, education, sex, race, tobacco use, diabetes status, body mass index, and arthritis status were controlled for in our models.
Our analysis initially included 9982 participants; 14 did not respond to questions assessing psoriasis and impaired mobility and were excluded. The prevalence of impaired mobility in patients with psoriasis was 17.1% compared with 10.9% among those without psoriasis (Table 1). There was a significant association between psoriasis and impaired mobility among patients aged 45 to 80 years after adjusting for potential confounding variables (adjusted odds ratio [AOR], 1.54; 95% CI, 1.04- 2.29; P=.032)(Table 2). Analyses of subgroups yielded no statistically significant results.



Our study demonstrated a statistically significant difference in mobility between individuals with psoriasis compared with the general population, which remained significant when controlling for arthritis, obesity, and diabetes (P=.032). This may be the result of several influences. First, the location of the psoriasis may impact mobility. Plantar psoriasis—a manifestation on the soles of the feet—can cause discomfort and pain, which can hinder walking and standing.5 Second, a study by Lasselin et al6 found that systemic inflammation contributes to mobility impairment through alterations in gait and posture, which suggests that the inflammatory processes inherent in psoriasis could intrinsically modify walking speed and stride, potentially exacerbating mobility difficulties independent of other comorbid conditions. These findings suggest that psoriasis may disproportionately affect individuals with impaired mobility, independent of comorbid arthritis, obesity, and diabetes.
These findings have broad implications for diversity, equity, and inclusion. They should prompt us to consider the practical challenges faced by this patient population and the ways that we can address barriers to care. Offering telehealth appointments, making primary care referrals for impaired mobility workups, and advising patients of direct-to-home delivery of prescriptions are good places to start.
Limitations to our study include the lack of specificity in the survey question, self-reporting bias, and the inability to control for the psoriasis location. Further investigations are warranted in large, representative US adult populations to assess the implications of impaired mobility in patients with psoriasis.
To the Editor:
Psoriasis is a chronic inflammatory condition that affects individuals in various extracutaneous ways.1 Prior studies have documented a decrease in exercise intensity among patients with psoriasis2; however, few studies have specifically investigated baseline mobility in this population. Baseline mobility denotes an individual’s fundamental ability to walk or move around without assistance of any kind. Impaired mobility—when baseline mobility is compromised—is an aspect of the wider diversity, equity, and inclusion framework that underscores the significance of recognizing challenges and promoting inclusive measures, both at the point of care and in research.3 study sought to analyze the relationship between psoriasis and baseline mobility among US adults (aged 45 to 80 years) utilizing the latest data from the National Health and Nutrition Examination Survey (NHANES) database for psoriasis.4 We used three 2-year cycles of NHANES data to create a 2009-2014 dataset.
The overall NHANES response rate among adults aged 45 to 80 years between 2009 and 2014 was 67.9%. Patients were categorized as having impaired mobility if they responded “yes” to the following question: “Because of a health problem, do you have difficulty walking without using any special equipment?” Psoriasis status was assessed by the following question: “Have you ever been told by a doctor or other health professional that you had psoriasis?” Multivariable logistic regression analyses were performed using Stata/SE 18.0 software (StataCorp LLC) to assess the relationship between psoriasis and impaired mobility. Age, income, education, sex, race, tobacco use, diabetes status, body mass index, and arthritis status were controlled for in our models.
Our analysis initially included 9982 participants; 14 did not respond to questions assessing psoriasis and impaired mobility and were excluded. The prevalence of impaired mobility in patients with psoriasis was 17.1% compared with 10.9% among those without psoriasis (Table 1). There was a significant association between psoriasis and impaired mobility among patients aged 45 to 80 years after adjusting for potential confounding variables (adjusted odds ratio [AOR], 1.54; 95% CI, 1.04- 2.29; P=.032)(Table 2). Analyses of subgroups yielded no statistically significant results.



Our study demonstrated a statistically significant difference in mobility between individuals with psoriasis compared with the general population, which remained significant when controlling for arthritis, obesity, and diabetes (P=.032). This may be the result of several influences. First, the location of the psoriasis may impact mobility. Plantar psoriasis—a manifestation on the soles of the feet—can cause discomfort and pain, which can hinder walking and standing.5 Second, a study by Lasselin et al6 found that systemic inflammation contributes to mobility impairment through alterations in gait and posture, which suggests that the inflammatory processes inherent in psoriasis could intrinsically modify walking speed and stride, potentially exacerbating mobility difficulties independent of other comorbid conditions. These findings suggest that psoriasis may disproportionately affect individuals with impaired mobility, independent of comorbid arthritis, obesity, and diabetes.
These findings have broad implications for diversity, equity, and inclusion. They should prompt us to consider the practical challenges faced by this patient population and the ways that we can address barriers to care. Offering telehealth appointments, making primary care referrals for impaired mobility workups, and advising patients of direct-to-home delivery of prescriptions are good places to start.
Limitations to our study include the lack of specificity in the survey question, self-reporting bias, and the inability to control for the psoriasis location. Further investigations are warranted in large, representative US adult populations to assess the implications of impaired mobility in patients with psoriasis.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi: 10.1016/j.jaad.2018.11.058
- Zheng Q, Sun XY, Miao X, et al. Association between physical activity and risk of prevalent psoriasis: A MOOSE-compliant meta-analysis. Medicine (Baltimore). 2018;97:e11394. doi: 10.1097 /MD.0000000000011394
- Mullin AE, Coe IR, Gooden EA, et al. Inclusion, diversity, equity, and accessibility: from organizational responsibility to leadership competency. Healthc Manage Forum. 2021;34311-315. doi: 10.1177/08404704211038232
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed October 21, 2023. https://wwwn.cdc.gov/nchs/nhanes/
- Romani M, Biela G, Farr K, et al. Plantar psoriasis: a review of the literature. Clin Podiatr Med Surg. 2021;38:541-552. doi: 10.1016 /j.cpm.2021.06.009
- Lasselin J, Sundelin T, Wayne PM, et al. Biological motion during inflammation in humans. Brain Behav Immun. 2020;84:147-153. doi: 10.1016/j.bbi.2019.11.019
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi: 10.1016/j.jaad.2018.11.058
- Zheng Q, Sun XY, Miao X, et al. Association between physical activity and risk of prevalent psoriasis: A MOOSE-compliant meta-analysis. Medicine (Baltimore). 2018;97:e11394. doi: 10.1097 /MD.0000000000011394
- Mullin AE, Coe IR, Gooden EA, et al. Inclusion, diversity, equity, and accessibility: from organizational responsibility to leadership competency. Healthc Manage Forum. 2021;34311-315. doi: 10.1177/08404704211038232
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed October 21, 2023. https://wwwn.cdc.gov/nchs/nhanes/
- Romani M, Biela G, Farr K, et al. Plantar psoriasis: a review of the literature. Clin Podiatr Med Surg. 2021;38:541-552. doi: 10.1016 /j.cpm.2021.06.009
- Lasselin J, Sundelin T, Wayne PM, et al. Biological motion during inflammation in humans. Brain Behav Immun. 2020;84:147-153. doi: 10.1016/j.bbi.2019.11.019
Exploring the Relationship Between Psoriasis and Mobility Among US Adults
Exploring the Relationship Between Psoriasis and Mobility Among US Adults
PRACTICE POINTS
- Mobility issues are more common in patients who have psoriasis than in those who do not.
- It is important to assess patients with psoriasis for mobility issues regardless of age or comorbid conditions such as arthritis, obesity, and diabetes.
- Dermatologists can help patients with psoriasis and impaired mobility overcome potential barriers to care by incorporating telehealth services into their practices and informing patients of direct-to-home delivery of prescriptions.
Cyclically Bleeding Umbilical Papules
Cyclically Bleeding Umbilical Papules
THE DIAGNOSIS: Cutaneous Endometriosis
On histopathology, a biopsy specimen of an umbilical papule showed a dermal lymphohistiocyticrich infiltrate, hemorrhage, and ectopic endometrial glands consistent with cutaneous endometriosis (CE)(Figure). Cutaneous endometriosis is a rare condition that typically affects females of reproductive potential and is characterized by endometrial glands and stroma within the dermis and hypodermis. Cutaneous endometriosis is classified as primary or secondary. There is no surgical history of the abdomen or pelvis in primary CE. In contrast, a history of abdominopelvic surgery is the defining characteristic of secondary CE, which is more common than primary CE and typically manifests as painful red, brown, or purple papules along preexisting surgical scars of the umbilicus, lower abdomen, or pelvic region.1 Our patient may have developed secondary CE related to the laparoscopic cholecystectomy performed 10 years prior. Surgical excision is considered the definitive treatment for CE, and hormonal therapy with danazol or leuprolide may help ameliorate symptoms.1 Our patient deferred any hormonal or surgical interventions to undergo fertility treatments for pregnancy.
Cyclical bleeding and pain that coincides with menstruation is consistent with CE; however, cyclical symptoms are not always present, which can lead to delayed or incorrect diagnosis. Biopsy and histopathologic analysis are required for definitive diagnosis and are critical for distinguishing CE from other conditions. The differential diagnosis in our patient included pyogenic granuloma, dermatofibrosarcoma protuberans, keloid, and cutaneous metastasis of a primary malignancy. Vascular lesions such as pyogenic granuloma can manifest with bleeding but have a characteristic histopathologic lobular capillary arrangement that was not present in our patient.
Dermatofibrosarcoma protuberans is a rare, slow-growing, malignant soft-tissue sarcoma that most commonly manifests on the trunk, arms, and legs.2 It is characterized by a slow-growing, indurated plaque that often is present for years and may suddenly progress into a smooth, red-brown, multinodular mass. Histopathology typically shows spindle cells infiltrating the dermis and subcutaneous tissue in storiform or whorled pattern with variations based on the tumor stage, as well as diffuse CD34 immunoreactivity.2
Keloids are dense, raised, hyperpigmented, fibrous nodules—sometimes with accompanying telangiectasias—that typically grow secondary to trauma and project past the boundaries of the initial trauma site.1 Keloids are more commonly seen in individuals with darker skin types and tend to grow larger in this population. Histopathology reveals thickened hyalinized collagen bundles, which were not seen in our patient.1
Metastatic skin lesions of the umbilicus are rare but can arise from internal malignancies including cancers of the lung, colon, and breast.3 We considered Sister Mary Joseph nodule, which is caused most commonly by metastasis of a primary gastrointestinal cancer and signifies poor prognosis. The histopathology of metastatic lesions would reveal the presence of atypical cells with cancer-specific markers. Histopathology along with the patient’s personal and family history, a comprehensive review of symptoms, and cancer screening may help with reaching the correct diagnosis.
The average duration between abdominopelvic surgery and onset of secondary CE symptoms is 3.7 to 5.3 years.4 Our patient presented 10 years post surgery and after cessation of oral contraception, which may suggest a potential role of hormonal contraception in delayed CE onset. Diagnosis of CE can be challenging due to atypical signs or symptoms, delayed onset, and lack of awareness among health care professionals. Patients with delayed diagnosis may endure multiple procedures, prolonged physical pain, and emotional distress. Furthermore, 30% to 50% of females with endometriosis experience infertility. Delayed diagnosis of CE compounded with associated age-related increase in oocyte atresia could potentially worsen fecundity as patients age.5 It is important to consider CE in the differential diagnosis of females of reproductive age who present with cyclical bleeding and abdominal or umbilical nodules.
- James WD, Elston D, Treat JR, et al. Andrews Diseases of the Skin: Clinical Dermatology. 13th ed. Elsevier; 2019. Accessed March 19, 2024. https://search.worldcat.org/title/1084979207
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Komurcugil I, Arslan Z, Bal ZI, et al. Cutaneous metastases different clinical presentations: case series and review of the literature. Dermatol Reports. 2022;15:9553.
- Marras S, Pluchino N, Petignat P, et al. Abdominal wall endometriosis: an 11-year retrospective observational cohort study. Published online September 16, 2019. Eur J Obstet Gynecol Reprod Biol X.
- Missmer SA, Hankinson SE, Spiegelman D, et al. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am J Epidemiol. 2004;160:784-796.
THE DIAGNOSIS: Cutaneous Endometriosis
On histopathology, a biopsy specimen of an umbilical papule showed a dermal lymphohistiocyticrich infiltrate, hemorrhage, and ectopic endometrial glands consistent with cutaneous endometriosis (CE)(Figure). Cutaneous endometriosis is a rare condition that typically affects females of reproductive potential and is characterized by endometrial glands and stroma within the dermis and hypodermis. Cutaneous endometriosis is classified as primary or secondary. There is no surgical history of the abdomen or pelvis in primary CE. In contrast, a history of abdominopelvic surgery is the defining characteristic of secondary CE, which is more common than primary CE and typically manifests as painful red, brown, or purple papules along preexisting surgical scars of the umbilicus, lower abdomen, or pelvic region.1 Our patient may have developed secondary CE related to the laparoscopic cholecystectomy performed 10 years prior. Surgical excision is considered the definitive treatment for CE, and hormonal therapy with danazol or leuprolide may help ameliorate symptoms.1 Our patient deferred any hormonal or surgical interventions to undergo fertility treatments for pregnancy.

Cyclical bleeding and pain that coincides with menstruation is consistent with CE; however, cyclical symptoms are not always present, which can lead to delayed or incorrect diagnosis. Biopsy and histopathologic analysis are required for definitive diagnosis and are critical for distinguishing CE from other conditions. The differential diagnosis in our patient included pyogenic granuloma, dermatofibrosarcoma protuberans, keloid, and cutaneous metastasis of a primary malignancy. Vascular lesions such as pyogenic granuloma can manifest with bleeding but have a characteristic histopathologic lobular capillary arrangement that was not present in our patient.
Dermatofibrosarcoma protuberans is a rare, slow-growing, malignant soft-tissue sarcoma that most commonly manifests on the trunk, arms, and legs.2 It is characterized by a slow-growing, indurated plaque that often is present for years and may suddenly progress into a smooth, red-brown, multinodular mass. Histopathology typically shows spindle cells infiltrating the dermis and subcutaneous tissue in storiform or whorled pattern with variations based on the tumor stage, as well as diffuse CD34 immunoreactivity.2
Keloids are dense, raised, hyperpigmented, fibrous nodules—sometimes with accompanying telangiectasias—that typically grow secondary to trauma and project past the boundaries of the initial trauma site.1 Keloids are more commonly seen in individuals with darker skin types and tend to grow larger in this population. Histopathology reveals thickened hyalinized collagen bundles, which were not seen in our patient.1
Metastatic skin lesions of the umbilicus are rare but can arise from internal malignancies including cancers of the lung, colon, and breast.3 We considered Sister Mary Joseph nodule, which is caused most commonly by metastasis of a primary gastrointestinal cancer and signifies poor prognosis. The histopathology of metastatic lesions would reveal the presence of atypical cells with cancer-specific markers. Histopathology along with the patient’s personal and family history, a comprehensive review of symptoms, and cancer screening may help with reaching the correct diagnosis.
The average duration between abdominopelvic surgery and onset of secondary CE symptoms is 3.7 to 5.3 years.4 Our patient presented 10 years post surgery and after cessation of oral contraception, which may suggest a potential role of hormonal contraception in delayed CE onset. Diagnosis of CE can be challenging due to atypical signs or symptoms, delayed onset, and lack of awareness among health care professionals. Patients with delayed diagnosis may endure multiple procedures, prolonged physical pain, and emotional distress. Furthermore, 30% to 50% of females with endometriosis experience infertility. Delayed diagnosis of CE compounded with associated age-related increase in oocyte atresia could potentially worsen fecundity as patients age.5 It is important to consider CE in the differential diagnosis of females of reproductive age who present with cyclical bleeding and abdominal or umbilical nodules.
THE DIAGNOSIS: Cutaneous Endometriosis
On histopathology, a biopsy specimen of an umbilical papule showed a dermal lymphohistiocyticrich infiltrate, hemorrhage, and ectopic endometrial glands consistent with cutaneous endometriosis (CE)(Figure). Cutaneous endometriosis is a rare condition that typically affects females of reproductive potential and is characterized by endometrial glands and stroma within the dermis and hypodermis. Cutaneous endometriosis is classified as primary or secondary. There is no surgical history of the abdomen or pelvis in primary CE. In contrast, a history of abdominopelvic surgery is the defining characteristic of secondary CE, which is more common than primary CE and typically manifests as painful red, brown, or purple papules along preexisting surgical scars of the umbilicus, lower abdomen, or pelvic region.1 Our patient may have developed secondary CE related to the laparoscopic cholecystectomy performed 10 years prior. Surgical excision is considered the definitive treatment for CE, and hormonal therapy with danazol or leuprolide may help ameliorate symptoms.1 Our patient deferred any hormonal or surgical interventions to undergo fertility treatments for pregnancy.

Cyclical bleeding and pain that coincides with menstruation is consistent with CE; however, cyclical symptoms are not always present, which can lead to delayed or incorrect diagnosis. Biopsy and histopathologic analysis are required for definitive diagnosis and are critical for distinguishing CE from other conditions. The differential diagnosis in our patient included pyogenic granuloma, dermatofibrosarcoma protuberans, keloid, and cutaneous metastasis of a primary malignancy. Vascular lesions such as pyogenic granuloma can manifest with bleeding but have a characteristic histopathologic lobular capillary arrangement that was not present in our patient.
Dermatofibrosarcoma protuberans is a rare, slow-growing, malignant soft-tissue sarcoma that most commonly manifests on the trunk, arms, and legs.2 It is characterized by a slow-growing, indurated plaque that often is present for years and may suddenly progress into a smooth, red-brown, multinodular mass. Histopathology typically shows spindle cells infiltrating the dermis and subcutaneous tissue in storiform or whorled pattern with variations based on the tumor stage, as well as diffuse CD34 immunoreactivity.2
Keloids are dense, raised, hyperpigmented, fibrous nodules—sometimes with accompanying telangiectasias—that typically grow secondary to trauma and project past the boundaries of the initial trauma site.1 Keloids are more commonly seen in individuals with darker skin types and tend to grow larger in this population. Histopathology reveals thickened hyalinized collagen bundles, which were not seen in our patient.1
Metastatic skin lesions of the umbilicus are rare but can arise from internal malignancies including cancers of the lung, colon, and breast.3 We considered Sister Mary Joseph nodule, which is caused most commonly by metastasis of a primary gastrointestinal cancer and signifies poor prognosis. The histopathology of metastatic lesions would reveal the presence of atypical cells with cancer-specific markers. Histopathology along with the patient’s personal and family history, a comprehensive review of symptoms, and cancer screening may help with reaching the correct diagnosis.
The average duration between abdominopelvic surgery and onset of secondary CE symptoms is 3.7 to 5.3 years.4 Our patient presented 10 years post surgery and after cessation of oral contraception, which may suggest a potential role of hormonal contraception in delayed CE onset. Diagnosis of CE can be challenging due to atypical signs or symptoms, delayed onset, and lack of awareness among health care professionals. Patients with delayed diagnosis may endure multiple procedures, prolonged physical pain, and emotional distress. Furthermore, 30% to 50% of females with endometriosis experience infertility. Delayed diagnosis of CE compounded with associated age-related increase in oocyte atresia could potentially worsen fecundity as patients age.5 It is important to consider CE in the differential diagnosis of females of reproductive age who present with cyclical bleeding and abdominal or umbilical nodules.
- James WD, Elston D, Treat JR, et al. Andrews Diseases of the Skin: Clinical Dermatology. 13th ed. Elsevier; 2019. Accessed March 19, 2024. https://search.worldcat.org/title/1084979207
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Komurcugil I, Arslan Z, Bal ZI, et al. Cutaneous metastases different clinical presentations: case series and review of the literature. Dermatol Reports. 2022;15:9553.
- Marras S, Pluchino N, Petignat P, et al. Abdominal wall endometriosis: an 11-year retrospective observational cohort study. Published online September 16, 2019. Eur J Obstet Gynecol Reprod Biol X.
- Missmer SA, Hankinson SE, Spiegelman D, et al. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am J Epidemiol. 2004;160:784-796.
- James WD, Elston D, Treat JR, et al. Andrews Diseases of the Skin: Clinical Dermatology. 13th ed. Elsevier; 2019. Accessed March 19, 2024. https://search.worldcat.org/title/1084979207
- Hao X, Billings SD, Wu F, et al. Dermatofibrosarcoma protuberans: update on the diagnosis and treatment. J Clin Med. 2020;9:1752.
- Komurcugil I, Arslan Z, Bal ZI, et al. Cutaneous metastases different clinical presentations: case series and review of the literature. Dermatol Reports. 2022;15:9553.
- Marras S, Pluchino N, Petignat P, et al. Abdominal wall endometriosis: an 11-year retrospective observational cohort study. Published online September 16, 2019. Eur J Obstet Gynecol Reprod Biol X.
- Missmer SA, Hankinson SE, Spiegelman D, et al. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am J Epidemiol. 2004;160:784-796.
Cyclically Bleeding Umbilical Papules
Cyclically Bleeding Umbilical Papules
A 38-year-old nulligravid female with menorrhagia and dysmenorrhea presented with cyclical umbilical bleeding of 1 year’s duration. Shortly before the onset of symptoms, the patient had discontinued oral contraceptive therapy with the intent to become pregnant. She had an uncomplicated laparoscopic cholecystectomy 10 years prior, but her medical history was otherwise unremarkable. At the current presentation, physical examination revealed multilobular brown papules with serosanguineous crusting in the umbilicus.
Apremilast Treatment Outcomes and Adverse Events in Psoriasis Patients With HIV
Apremilast Treatment Outcomes and Adverse Events in Psoriasis Patients With HIV
To the Editor:
Psoriasis is a chronic systemic inflammatory disease that affects 1% to 3% of the global population.1,2 Due to dysregulation of the immune system, patients with HIV who have concurrent moderate to severe psoriasis present a clinical therapeutic challenge for dermatologists. Recent guidelines from the American Academy of Dermatology recommended avoiding certain systemic treatments (eg, methotrexate, cyclosporine) in patients who are HIV positive due to their immunosuppressive effects, as well as cautious use of certain biologics in populations with HIV.3 Traditional therapies for managing psoriasis in patients with HIV have included topical agents, antiretroviral therapy (ART), phototherapy, and acitretin; however, phototherapy can be logistically cumbersome for patients, and in the setting of ART, acitretin has the potential to exacerbate hypertriglyceridemia as well as other undesirable adverse effects.3
Apremilast is a phosphodiesterase 4 inhibitor that has emerged as a promising alternative in patients with HIV who require treatment for psoriasis. It has demonstrated clinical efficacy in psoriasis and has minimal immunosuppressive risk.4 Despite its potential in this population, reports of apremilast used in patients who are HIV positive are rare, and these patients often are excluded from larges studies. In this study, we reviewed the literature to evaluate outcomes and adverse events in patients with HIV who underwent psoriasis treatment with apremilast.
A search of PubMed articles indexed for MEDLINE from the inception of the database through January 2023 was conducted using the terms psoriasis, human immunodeficiency virus, acquired immunodeficiency syndrome, therapy, apremilast, and adverse events. The inclusion criteria were articles that reported patients with HIV and psoriasis undergoing treatment with apremilast with subsequent follow-up to delineate potential outcomes and adverse effects. Non–English language articles were excluded.
Our search of the literature yielded 7 patients with HIV and psoriasis who were treated with apremilast (eTable).5-11 All of the patients were male and ranged in age from 31 to 55 years, and all had pretreatment CD4 cell counts greater than 450 cells/mm3. All but 1 patient were confirmed to have undergone ART prior to treatment with apremilast, and all were treated using the traditional apremilast titration from 10 mg to 30 mg orally twice daily.
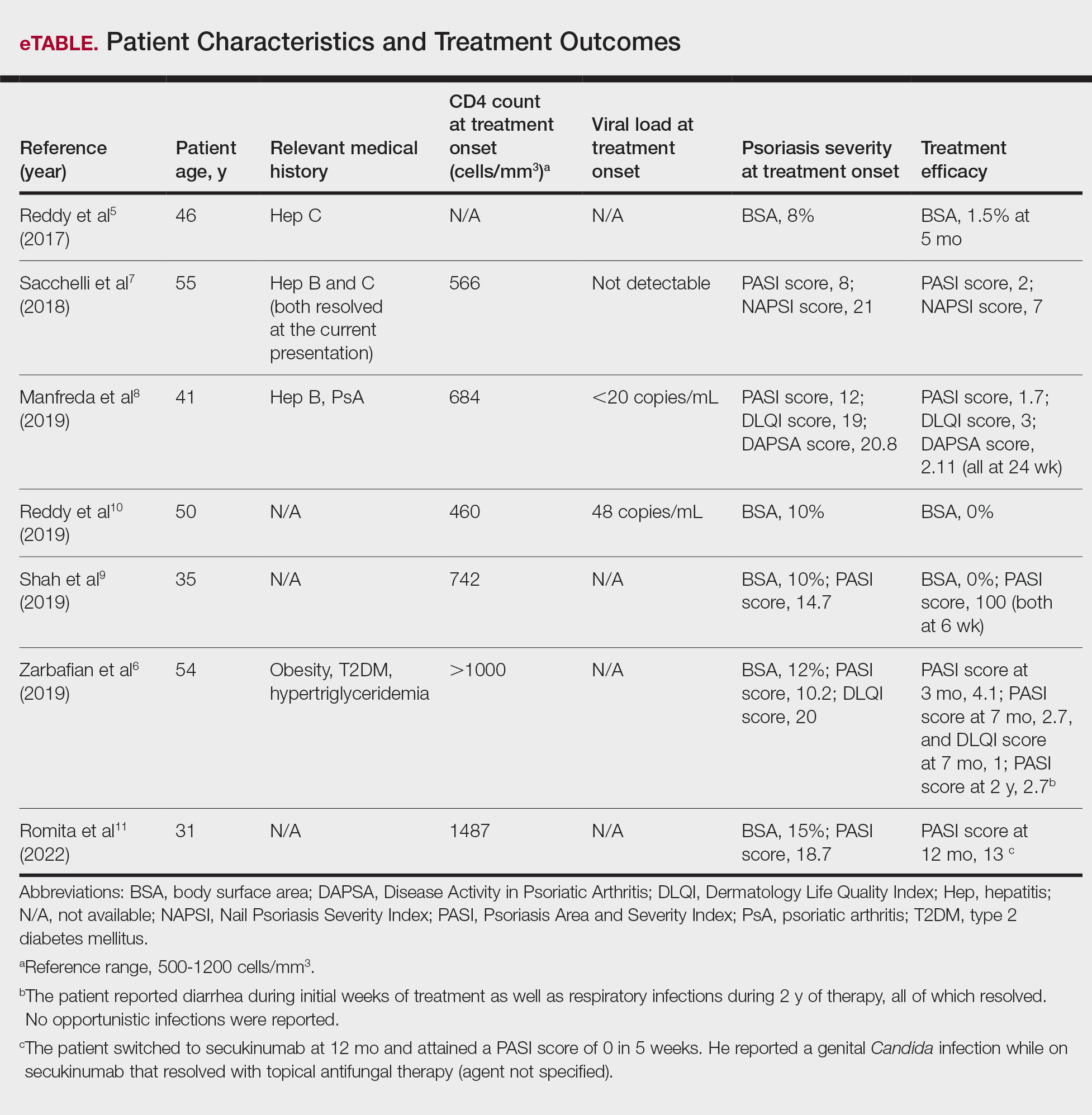
The mean pretreatment Psoriasis Area and Severity Index (PASI) score in the patients we evaluated was 12.2, with an average reduction in PASI score of 9.3. This equated to achievement of PASI 75 or greater (ie, representing at least a 75% improvement in psoriasis) in 4 (57.1%) patients, with clinical improvement confirmed in all 7 patients (100.0%)(eTable). The average follow-up time was 9.7 months (range, 6 weeks to 24 months). Only 1 (14.3%) patient experienced any adverse effects, which included self-resolving diarrhea and respiratory infections (nonopportunistic) over a follow-up period of 2 years.6 Of note, gastrointestinal upset is common with apremilast and usually improves over time.12
Apremilast represents a safe and effective alternative systemic therapy for patients with HIV and psoriasis.4 As a phosphodiesterase 4 inhibitor, apremilast leads to increased levels of cyclic adenosine monophosphate, which restores an equilibrium between proinflammatory (eg, tumor necrosis factors, interferons, IL-2, IL-6, IL-12, IL-23) and anti-inflammatory (eg, IL-10) cytokines.13 Unlike most biologics that target and inhibit a specific proinflammatory cytokine, apremilast’s homeostatic mechanism may explain its minimal immunosuppressive adverse effects.
In the majority of patients we evaluated, initiation of apremilast led to documented clinical improvement. It is worth noting that some patients presented with a relevant medical history and/or comorbidities such as hepatitis and metabolic conditions (eg, obesity, type 2 diabetes mellitus, hypertriglyceridemia). Despite these comorbidities, initiation of apremilast therapy in these patients led to clinical improvement of psoriasis overall. Notable cases from our study included a 41-year-old man with concurrent hepatitis B and psoriatic arthritis who achieved PASI 90 after 24 weeks of apremilast therapy8; a 46-year-old man with concurrent hepatitis C who went from 8% to 1.5% body surface area affected after 5 months of treatment with apremilast5; and a 54-year-old man with concurrent obesity, type 2 diabetes mellitus, and hypertriglyceridemia who went from a PASI score of 10.2 to 4.1 after 3 months of apremilast treatment and maintained a PASI score of 2.7 at 2 years’ follow up (eTable).6
Limitations of this study included the small sample size and homogeneous demographic consisting only of adult males, which restrict the external validity of the findings. Despite limitations, apremilast was utilized effectively for patients with both psoriasis and psoriatic arthritis. The observed effectiveness of apremilast in multiple forms of psoriasis provides valuable insights into the drug’s versatility in this patient population.
The use of apremilast for treatment of psoriasis in patients with HIV represents an important therapeutic development. Its effectiveness in reducing psoriasis symptoms in these immunocompromised patients makes it a viable alternative to traditional systemic therapies that might be contraindicated in this population. While larger studies would be ideal, the exclusion of patients with HIV from clinical trials presents an obstacle and therefore makes case series and reviews helpful for clinicians in bridging the gap with respect to treatment options for these patients. Apremilast may be a safe and effective medication for patients with HIV and psoriasis who require systemic therapy to treat their skin disease.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516. doi:10.1016/j.jaad.2013.11.013
- Parisi R, Symmons DP, Griffiths CE, et al; Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53. doi:10.1016/j.jaad.2018.06.056
- Crowley J, Thaci D, Joly P, et al. Long-term safety and tolerability of apremilast in patients with psoriasis: pooled safety analysis for >156 weeks from 2 phase 3, randomized, controlled trials (ESTEEM 1 and 2). J Am Acad Dermatol. 2017;77:310-317.e1.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:E481-E482. doi:10.1111/jdv.14301
- Zarbafian M, Cote B, Richer V. Treatment of moderate to severe psoriasis with apremilast over 2 years in the context of long-term treated HIV infection: a case report. SAGE Open Med Case Rep. 2019;7:2050313X19845193. doi:10.1177/2050313X19845193 doi:10.1016/j.jaad.2017.01.052
- Sacchelli L, Patrizi A, Ferrara F, et al. Apremilast as therapeutic option in a HIV positive patient with severe psoriasis. Dermatol Ther. 2018;31:E12719. doi:10.1111/dth.12719
- Manfreda V, Esposito M, Campione E, et al. Apremilast efficacy and safety in a psoriatic arthritis patient affected by HIV and HBV virus infections. Postgrad Med. 2019;131:239-240. doi:10.1080/00325481.2019 .1575613
- Shah BJ, Mistry D, Chaudhary N. Apremilast in people living with HIV with psoriasis vulgaris: a case report. Indian J Dermatol. 2019;64:242- 244. doi:10.4103/ijd.IJD_633_18
- Reddy SP, Lee E, Wu JJ. Apremilast and phototherapy for treatment of psoriasis in a patient with human immunodeficiency virus. Cutis. 2019;103:E6-E7.
- Romita P, Foti C, Calianno G, et al. Successful treatment with secukinumab in an HIV-positive psoriatic patient after failure of apremilast. Dermatol Ther. 2022;35:E15610. doi:10.1111/dth.15610
- Zeb L, Mhaskar R, Lewis S, et al. Real-world drug survival and reasons for treatment discontinuation of biologics and apremilast in patients with psoriasis in an academic center. Dermatol Ther. 2021;34:E14826. doi:10.1111/dth.14826
- Schafer P. Apremilast mechanism of action and application to psoriasis and psoriatic arthritis. Biochem Pharmacol. 2012;83:1583-1590. doi:10.1016/j.bcp.2012.01.001
To the Editor:
Psoriasis is a chronic systemic inflammatory disease that affects 1% to 3% of the global population.1,2 Due to dysregulation of the immune system, patients with HIV who have concurrent moderate to severe psoriasis present a clinical therapeutic challenge for dermatologists. Recent guidelines from the American Academy of Dermatology recommended avoiding certain systemic treatments (eg, methotrexate, cyclosporine) in patients who are HIV positive due to their immunosuppressive effects, as well as cautious use of certain biologics in populations with HIV.3 Traditional therapies for managing psoriasis in patients with HIV have included topical agents, antiretroviral therapy (ART), phototherapy, and acitretin; however, phototherapy can be logistically cumbersome for patients, and in the setting of ART, acitretin has the potential to exacerbate hypertriglyceridemia as well as other undesirable adverse effects.3
Apremilast is a phosphodiesterase 4 inhibitor that has emerged as a promising alternative in patients with HIV who require treatment for psoriasis. It has demonstrated clinical efficacy in psoriasis and has minimal immunosuppressive risk.4 Despite its potential in this population, reports of apremilast used in patients who are HIV positive are rare, and these patients often are excluded from larges studies. In this study, we reviewed the literature to evaluate outcomes and adverse events in patients with HIV who underwent psoriasis treatment with apremilast.
A search of PubMed articles indexed for MEDLINE from the inception of the database through January 2023 was conducted using the terms psoriasis, human immunodeficiency virus, acquired immunodeficiency syndrome, therapy, apremilast, and adverse events. The inclusion criteria were articles that reported patients with HIV and psoriasis undergoing treatment with apremilast with subsequent follow-up to delineate potential outcomes and adverse effects. Non–English language articles were excluded.
Our search of the literature yielded 7 patients with HIV and psoriasis who were treated with apremilast (eTable).5-11 All of the patients were male and ranged in age from 31 to 55 years, and all had pretreatment CD4 cell counts greater than 450 cells/mm3. All but 1 patient were confirmed to have undergone ART prior to treatment with apremilast, and all were treated using the traditional apremilast titration from 10 mg to 30 mg orally twice daily.

The mean pretreatment Psoriasis Area and Severity Index (PASI) score in the patients we evaluated was 12.2, with an average reduction in PASI score of 9.3. This equated to achievement of PASI 75 or greater (ie, representing at least a 75% improvement in psoriasis) in 4 (57.1%) patients, with clinical improvement confirmed in all 7 patients (100.0%)(eTable). The average follow-up time was 9.7 months (range, 6 weeks to 24 months). Only 1 (14.3%) patient experienced any adverse effects, which included self-resolving diarrhea and respiratory infections (nonopportunistic) over a follow-up period of 2 years.6 Of note, gastrointestinal upset is common with apremilast and usually improves over time.12
Apremilast represents a safe and effective alternative systemic therapy for patients with HIV and psoriasis.4 As a phosphodiesterase 4 inhibitor, apremilast leads to increased levels of cyclic adenosine monophosphate, which restores an equilibrium between proinflammatory (eg, tumor necrosis factors, interferons, IL-2, IL-6, IL-12, IL-23) and anti-inflammatory (eg, IL-10) cytokines.13 Unlike most biologics that target and inhibit a specific proinflammatory cytokine, apremilast’s homeostatic mechanism may explain its minimal immunosuppressive adverse effects.
In the majority of patients we evaluated, initiation of apremilast led to documented clinical improvement. It is worth noting that some patients presented with a relevant medical history and/or comorbidities such as hepatitis and metabolic conditions (eg, obesity, type 2 diabetes mellitus, hypertriglyceridemia). Despite these comorbidities, initiation of apremilast therapy in these patients led to clinical improvement of psoriasis overall. Notable cases from our study included a 41-year-old man with concurrent hepatitis B and psoriatic arthritis who achieved PASI 90 after 24 weeks of apremilast therapy8; a 46-year-old man with concurrent hepatitis C who went from 8% to 1.5% body surface area affected after 5 months of treatment with apremilast5; and a 54-year-old man with concurrent obesity, type 2 diabetes mellitus, and hypertriglyceridemia who went from a PASI score of 10.2 to 4.1 after 3 months of apremilast treatment and maintained a PASI score of 2.7 at 2 years’ follow up (eTable).6
Limitations of this study included the small sample size and homogeneous demographic consisting only of adult males, which restrict the external validity of the findings. Despite limitations, apremilast was utilized effectively for patients with both psoriasis and psoriatic arthritis. The observed effectiveness of apremilast in multiple forms of psoriasis provides valuable insights into the drug’s versatility in this patient population.
The use of apremilast for treatment of psoriasis in patients with HIV represents an important therapeutic development. Its effectiveness in reducing psoriasis symptoms in these immunocompromised patients makes it a viable alternative to traditional systemic therapies that might be contraindicated in this population. While larger studies would be ideal, the exclusion of patients with HIV from clinical trials presents an obstacle and therefore makes case series and reviews helpful for clinicians in bridging the gap with respect to treatment options for these patients. Apremilast may be a safe and effective medication for patients with HIV and psoriasis who require systemic therapy to treat their skin disease.
To the Editor:
Psoriasis is a chronic systemic inflammatory disease that affects 1% to 3% of the global population.1,2 Due to dysregulation of the immune system, patients with HIV who have concurrent moderate to severe psoriasis present a clinical therapeutic challenge for dermatologists. Recent guidelines from the American Academy of Dermatology recommended avoiding certain systemic treatments (eg, methotrexate, cyclosporine) in patients who are HIV positive due to their immunosuppressive effects, as well as cautious use of certain biologics in populations with HIV.3 Traditional therapies for managing psoriasis in patients with HIV have included topical agents, antiretroviral therapy (ART), phototherapy, and acitretin; however, phototherapy can be logistically cumbersome for patients, and in the setting of ART, acitretin has the potential to exacerbate hypertriglyceridemia as well as other undesirable adverse effects.3
Apremilast is a phosphodiesterase 4 inhibitor that has emerged as a promising alternative in patients with HIV who require treatment for psoriasis. It has demonstrated clinical efficacy in psoriasis and has minimal immunosuppressive risk.4 Despite its potential in this population, reports of apremilast used in patients who are HIV positive are rare, and these patients often are excluded from larges studies. In this study, we reviewed the literature to evaluate outcomes and adverse events in patients with HIV who underwent psoriasis treatment with apremilast.
A search of PubMed articles indexed for MEDLINE from the inception of the database through January 2023 was conducted using the terms psoriasis, human immunodeficiency virus, acquired immunodeficiency syndrome, therapy, apremilast, and adverse events. The inclusion criteria were articles that reported patients with HIV and psoriasis undergoing treatment with apremilast with subsequent follow-up to delineate potential outcomes and adverse effects. Non–English language articles were excluded.
Our search of the literature yielded 7 patients with HIV and psoriasis who were treated with apremilast (eTable).5-11 All of the patients were male and ranged in age from 31 to 55 years, and all had pretreatment CD4 cell counts greater than 450 cells/mm3. All but 1 patient were confirmed to have undergone ART prior to treatment with apremilast, and all were treated using the traditional apremilast titration from 10 mg to 30 mg orally twice daily.

The mean pretreatment Psoriasis Area and Severity Index (PASI) score in the patients we evaluated was 12.2, with an average reduction in PASI score of 9.3. This equated to achievement of PASI 75 or greater (ie, representing at least a 75% improvement in psoriasis) in 4 (57.1%) patients, with clinical improvement confirmed in all 7 patients (100.0%)(eTable). The average follow-up time was 9.7 months (range, 6 weeks to 24 months). Only 1 (14.3%) patient experienced any adverse effects, which included self-resolving diarrhea and respiratory infections (nonopportunistic) over a follow-up period of 2 years.6 Of note, gastrointestinal upset is common with apremilast and usually improves over time.12
Apremilast represents a safe and effective alternative systemic therapy for patients with HIV and psoriasis.4 As a phosphodiesterase 4 inhibitor, apremilast leads to increased levels of cyclic adenosine monophosphate, which restores an equilibrium between proinflammatory (eg, tumor necrosis factors, interferons, IL-2, IL-6, IL-12, IL-23) and anti-inflammatory (eg, IL-10) cytokines.13 Unlike most biologics that target and inhibit a specific proinflammatory cytokine, apremilast’s homeostatic mechanism may explain its minimal immunosuppressive adverse effects.
In the majority of patients we evaluated, initiation of apremilast led to documented clinical improvement. It is worth noting that some patients presented with a relevant medical history and/or comorbidities such as hepatitis and metabolic conditions (eg, obesity, type 2 diabetes mellitus, hypertriglyceridemia). Despite these comorbidities, initiation of apremilast therapy in these patients led to clinical improvement of psoriasis overall. Notable cases from our study included a 41-year-old man with concurrent hepatitis B and psoriatic arthritis who achieved PASI 90 after 24 weeks of apremilast therapy8; a 46-year-old man with concurrent hepatitis C who went from 8% to 1.5% body surface area affected after 5 months of treatment with apremilast5; and a 54-year-old man with concurrent obesity, type 2 diabetes mellitus, and hypertriglyceridemia who went from a PASI score of 10.2 to 4.1 after 3 months of apremilast treatment and maintained a PASI score of 2.7 at 2 years’ follow up (eTable).6
Limitations of this study included the small sample size and homogeneous demographic consisting only of adult males, which restrict the external validity of the findings. Despite limitations, apremilast was utilized effectively for patients with both psoriasis and psoriatic arthritis. The observed effectiveness of apremilast in multiple forms of psoriasis provides valuable insights into the drug’s versatility in this patient population.
The use of apremilast for treatment of psoriasis in patients with HIV represents an important therapeutic development. Its effectiveness in reducing psoriasis symptoms in these immunocompromised patients makes it a viable alternative to traditional systemic therapies that might be contraindicated in this population. While larger studies would be ideal, the exclusion of patients with HIV from clinical trials presents an obstacle and therefore makes case series and reviews helpful for clinicians in bridging the gap with respect to treatment options for these patients. Apremilast may be a safe and effective medication for patients with HIV and psoriasis who require systemic therapy to treat their skin disease.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516. doi:10.1016/j.jaad.2013.11.013
- Parisi R, Symmons DP, Griffiths CE, et al; Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53. doi:10.1016/j.jaad.2018.06.056
- Crowley J, Thaci D, Joly P, et al. Long-term safety and tolerability of apremilast in patients with psoriasis: pooled safety analysis for >156 weeks from 2 phase 3, randomized, controlled trials (ESTEEM 1 and 2). J Am Acad Dermatol. 2017;77:310-317.e1.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:E481-E482. doi:10.1111/jdv.14301
- Zarbafian M, Cote B, Richer V. Treatment of moderate to severe psoriasis with apremilast over 2 years in the context of long-term treated HIV infection: a case report. SAGE Open Med Case Rep. 2019;7:2050313X19845193. doi:10.1177/2050313X19845193 doi:10.1016/j.jaad.2017.01.052
- Sacchelli L, Patrizi A, Ferrara F, et al. Apremilast as therapeutic option in a HIV positive patient with severe psoriasis. Dermatol Ther. 2018;31:E12719. doi:10.1111/dth.12719
- Manfreda V, Esposito M, Campione E, et al. Apremilast efficacy and safety in a psoriatic arthritis patient affected by HIV and HBV virus infections. Postgrad Med. 2019;131:239-240. doi:10.1080/00325481.2019 .1575613
- Shah BJ, Mistry D, Chaudhary N. Apremilast in people living with HIV with psoriasis vulgaris: a case report. Indian J Dermatol. 2019;64:242- 244. doi:10.4103/ijd.IJD_633_18
- Reddy SP, Lee E, Wu JJ. Apremilast and phototherapy for treatment of psoriasis in a patient with human immunodeficiency virus. Cutis. 2019;103:E6-E7.
- Romita P, Foti C, Calianno G, et al. Successful treatment with secukinumab in an HIV-positive psoriatic patient after failure of apremilast. Dermatol Ther. 2022;35:E15610. doi:10.1111/dth.15610
- Zeb L, Mhaskar R, Lewis S, et al. Real-world drug survival and reasons for treatment discontinuation of biologics and apremilast in patients with psoriasis in an academic center. Dermatol Ther. 2021;34:E14826. doi:10.1111/dth.14826
- Schafer P. Apremilast mechanism of action and application to psoriasis and psoriatic arthritis. Biochem Pharmacol. 2012;83:1583-1590. doi:10.1016/j.bcp.2012.01.001
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516. doi:10.1016/j.jaad.2013.11.013
- Parisi R, Symmons DP, Griffiths CE, et al; Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol. 2013;133:377-385. doi:10.1038/jid.2012.339
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections. J Am Acad Dermatol. 2019;80:43-53. doi:10.1016/j.jaad.2018.06.056
- Crowley J, Thaci D, Joly P, et al. Long-term safety and tolerability of apremilast in patients with psoriasis: pooled safety analysis for >156 weeks from 2 phase 3, randomized, controlled trials (ESTEEM 1 and 2). J Am Acad Dermatol. 2017;77:310-317.e1.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:E481-E482. doi:10.1111/jdv.14301
- Zarbafian M, Cote B, Richer V. Treatment of moderate to severe psoriasis with apremilast over 2 years in the context of long-term treated HIV infection: a case report. SAGE Open Med Case Rep. 2019;7:2050313X19845193. doi:10.1177/2050313X19845193 doi:10.1016/j.jaad.2017.01.052
- Sacchelli L, Patrizi A, Ferrara F, et al. Apremilast as therapeutic option in a HIV positive patient with severe psoriasis. Dermatol Ther. 2018;31:E12719. doi:10.1111/dth.12719
- Manfreda V, Esposito M, Campione E, et al. Apremilast efficacy and safety in a psoriatic arthritis patient affected by HIV and HBV virus infections. Postgrad Med. 2019;131:239-240. doi:10.1080/00325481.2019 .1575613
- Shah BJ, Mistry D, Chaudhary N. Apremilast in people living with HIV with psoriasis vulgaris: a case report. Indian J Dermatol. 2019;64:242- 244. doi:10.4103/ijd.IJD_633_18
- Reddy SP, Lee E, Wu JJ. Apremilast and phototherapy for treatment of psoriasis in a patient with human immunodeficiency virus. Cutis. 2019;103:E6-E7.
- Romita P, Foti C, Calianno G, et al. Successful treatment with secukinumab in an HIV-positive psoriatic patient after failure of apremilast. Dermatol Ther. 2022;35:E15610. doi:10.1111/dth.15610
- Zeb L, Mhaskar R, Lewis S, et al. Real-world drug survival and reasons for treatment discontinuation of biologics and apremilast in patients with psoriasis in an academic center. Dermatol Ther. 2021;34:E14826. doi:10.1111/dth.14826
- Schafer P. Apremilast mechanism of action and application to psoriasis and psoriatic arthritis. Biochem Pharmacol. 2012;83:1583-1590. doi:10.1016/j.bcp.2012.01.001
Apremilast Treatment Outcomes and Adverse Events in Psoriasis Patients With HIV
Apremilast Treatment Outcomes and Adverse Events in Psoriasis Patients With HIV
PRACTICE POINT
- For patients with HIV who require systemic therapy for psoriasis, apremilast may provide an effective and safe therapeutic option, with minimal immunosuppressive adverse effects.