User login
Can Peripheral Nerve Blocks Improve Joint Replacement Outcomes?
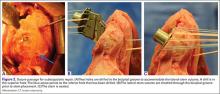
Patients who receive a peripheral nerve block during hip or knee replacement surgery are less likely to experience complications, according to a recent large retrospective study.
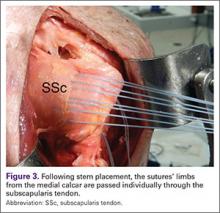
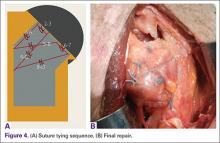
Researchers reviewed more than 1 million hip and knee replacements performed between 2006 and 2013 using data from approximately 3,000 hospitals in the United States.
Investigators compiled information on cardiac, pulmonary, gastrointestinal, and renal complications. They also determined the rate of infections, wound complications, and inpatient falls. In addition, they analyzed data on resource utilization, which included the number of blood transfusions needed, admission to an intensive care unit, opioid consumption, length of hospital stay, and the cost of hospitalization.
They looked at data for 342,726 patients who had hip replacement surgery and 719,426 who had knee replacement surgery.
Overall, 18% of the patients received a peripheral nerve block. They found the rate peripheral nerve block use among patients with knee replacement increased from 15.2% in 2006 to 24.5% in 2013. The use of peripheral nerve blocks was associated with significantly lower odds for almost all complications.
A strong effect was seen for cardiac complications in patients with knee replacement and for wound complications in people who had hip replacement. Similar patterns were observed for resource utilization, particularly in length of hospital stay among patients with hip replacement.
Patients who receive a peripheral nerve block during hip or knee replacement surgery are less likely to experience complications, according to a recent large retrospective study.
Researchers reviewed more than 1 million hip and knee replacements performed between 2006 and 2013 using data from approximately 3,000 hospitals in the United States.
Investigators compiled information on cardiac, pulmonary, gastrointestinal, and renal complications. They also determined the rate of infections, wound complications, and inpatient falls. In addition, they analyzed data on resource utilization, which included the number of blood transfusions needed, admission to an intensive care unit, opioid consumption, length of hospital stay, and the cost of hospitalization.
They looked at data for 342,726 patients who had hip replacement surgery and 719,426 who had knee replacement surgery.
Overall, 18% of the patients received a peripheral nerve block. They found the rate peripheral nerve block use among patients with knee replacement increased from 15.2% in 2006 to 24.5% in 2013. The use of peripheral nerve blocks was associated with significantly lower odds for almost all complications.
A strong effect was seen for cardiac complications in patients with knee replacement and for wound complications in people who had hip replacement. Similar patterns were observed for resource utilization, particularly in length of hospital stay among patients with hip replacement.
Patients who receive a peripheral nerve block during hip or knee replacement surgery are less likely to experience complications, according to a recent large retrospective study.
Researchers reviewed more than 1 million hip and knee replacements performed between 2006 and 2013 using data from approximately 3,000 hospitals in the United States.
Investigators compiled information on cardiac, pulmonary, gastrointestinal, and renal complications. They also determined the rate of infections, wound complications, and inpatient falls. In addition, they analyzed data on resource utilization, which included the number of blood transfusions needed, admission to an intensive care unit, opioid consumption, length of hospital stay, and the cost of hospitalization.
They looked at data for 342,726 patients who had hip replacement surgery and 719,426 who had knee replacement surgery.
Overall, 18% of the patients received a peripheral nerve block. They found the rate peripheral nerve block use among patients with knee replacement increased from 15.2% in 2006 to 24.5% in 2013. The use of peripheral nerve blocks was associated with significantly lower odds for almost all complications.
A strong effect was seen for cardiac complications in patients with knee replacement and for wound complications in people who had hip replacement. Similar patterns were observed for resource utilization, particularly in length of hospital stay among patients with hip replacement.
Periprocedural Management of Chronically Anticoagulated Patients: A Practical Approach to Use of Novel Anticoagulants in Orthopedic Surgery
Chronic anticoagulation is a common preexisting condition in patients undergoing total joint arthroplasty (TJA). Atrial fibrillation (AF), the most common underlying disorder requiring chronic anticoagulation, affects more than 3 million patients in the United States—a number that is projected to increase to 16 million by 2050.1,2 Other common indications for anticoagulation are deep vein thrombosis (DVT) treatment, presence of a prosthetic heart valve, and venous thromboembolism (VTE) prevention after hip or knee arthroplasty. These patients face the additional risks of hemorrhage, persistent wound drainage, hematoma formation, transfusion requirements, periprosthetic joint infection, and longer hospital stay.1 Chronic anticoagulation traditionally has been managed with warfarin, which inhibits production of the vitamin K–dependent clotting factors II, VII, IX, and X. However, the new novel oral anticoagulants (NOACs), which target individual factors in the clotting cascade, are gaining favor as chronic anticoagulant agents because of their ease of use and improved efficacy and safety. These agents include the factor IIA inhibitor dabigatran (Pradaxa) and the direct factor Xa inhibitors rivaroxaban (Xarelto) and apixaban (Eliquis).
Management of patients at risk for thromboembolism and bleeding issues, particularly within the context of elective, urgent, or emergent orthopedic surgeries, is an evolving area. Understanding the pharmacokinetics, conventional laboratory tests, dosing, and reversal methods for NOACs is important, especially because clinical data are limited and the treatment itself can cause clinically significant harm.
In this article, we review the medical literature on these medications, their mechanism of action, and their reversal agents, and outline a practical approach for managing patients during the perioperative period.
Dabigatran
In October 2010, dabigatran became the first NOAC approved by the US Food and Drug Administration (FDA) for the prevention of arterial thromboembolic events in patients with nonvalvular AF, on the basis of the results of the RELY (Randomized Evaluation of Long-Term Anticoagulation Therapy) trial. Dabigatran is an oral factor IIA (thrombin) inhibitor. From time of ingestion, dabigatran takes 1.25 to 3 hours to reach peak plasma concentration. It has a half-life of 12 to 14 hours, is excreted predominantly by the kidneys (80%), and is renally dosed. The usual dose is 150 mg 2 times daily if creatinine clearance (CrCl) is >30 mL/minute, or 75 mg 2 times daily if CrCl is 15 to 30 mL/minute.3 Dabigatran is not recommended for patients with CrCl <15 mL/minute.
Dabigatran affects prothrombin time (PT), activated partial thromboplastin time (aPTT), ecarin clotting time, and thrombin time, with the latter 2 providing the most accurate means of monitoring appropriate drug levels.3,4 Of the tests commonly used to assess coagulation hemostasis in hospitals, normalization of thrombin time and aPTT provide the most accurate results (Table 1). The pharmacokinetics of dabigatran mandate consideration of dose, time of ingestion relative to time of blood sampling, and renal function in the assessment of coagulation hemostasis.
For elective surgeries, the periprocedure recommendation for patients being treated with dabigatran is to discontinue the medication 3 to 4 days before an operation if CrCl is ≥50 mL/minute, or 4 to 5 days beforehand if CrCl is <50 mL/minute.3 There is no antidote for dabigatran. In an in vitro model, activated charcoal reduced 99.9% of dabigatran absorption after recent ingestion.3 According to case reports, acute hemodialysis successfully removed 60% of the medication after 6 hours.5 In patients with end-stage renal disease, hemodialysis removed up to 68% of active dabigatran after 4 hours.3
Pernod and colleagues6 proposed that urgent surgeries can proceed if the concentration of dabigatran is ≤30 ng/mL—equivalent to normal aPTT. Their dictum was extrapolated from the data of patients who underwent elective surgeries while being treated with dabigatran, as recorded during the RELY trial. According to Pernod and colleagues,6 if aPTT is increased (probable drug level, ≥30 ng/mL), surgery should be postponed for up to 12 hours, with aPTT checked again and the process repeated if the concentration of dabigatran is still elevated and surgery can continue to be delayed. In patients who require urgent surgical interventions, we previously utilized nanofiltered activated prothrombin complex concentrate (aPCC; Feiba NF) 30 to 50 IU/kg over prothrombin complex concentrate (PCC; Kcentra or Bebulin) 25 to 50 IU/kg, as supported by in vitro and animal model studies and anecdotal case reports. However, neither aPCC nor PCC fully corrects the abnormalities evident on hemostasis tests.3,6 In October 2015, the FDA approved Idarucizumab (Praxbind), an injectable monoclonal antibody fragment that binds to dabigatran, as a reversing agent for use in urgent/emergent settings. Recommendation is to administer two 50-ml bolus infusions, each containing 2.5 g of idarucizumab, no more than 15 minutes apart.7 Additionally, hemodialysis could be discussed before surgery, with the understanding that it will take a long time to reach the threshold of 30 ng/mL in these patients (Table 2).
Rivaroxaban
Rivaroxaban is an oral direct factor Xa inhibitor that was initially approved in November 2011 for the prevention of stroke and systemic embolism in patients with nonvalvular AF. Since then, clinical use of rivaroxaban has been expanded to include prevention of VTE after elective hip or knee arthroplasty as well as treatment of DVT and prevention of recurrent VTE after acute DVT. In the phase 3 ROCKET AF (Rivaroxaban Once-Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation) study, rivaroxaban 20 mg daily (CrCl, ≥50 mL/min) and rivaroxaban 15 mg daily (CrCl, 15-49 mL/min) were equally effective as warfarin. Compared with warfarin, rivaroxaban had a similar safety rate for bleeding and adverse events but fewer intracranial hemorrhage and fatal bleeding events.8 On the basis of the outcomes of the RECORD (Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Venous Thrombosis and Pulmonary Embolism) studies comparing rivaroxaban and enoxaparin sodium, rivaroxaban 10 mg daily was approved for the prevention of VTE and pulmonary embolism after elective hip or knee arthroplasty.8
The half-life of rivaroxaban is 5 to 9 hours in the young and 11 to 13 hours in the elderly.8 As rivaroxaban takes 2 to 4 hours after ingestion to reach peak plasma concentration, it is important to know the timing and the dose taken. Because of the short half-life and rapid onset of action of this medication, bridging with another anticoagulant is not required when rivaroxaban is discontinued before surgery or initiated after surgery.8 The recommendation is to withhold rivaroxaban for 24 to 48 hours before surgery and then to administer the first postoperative dose 6 to 10 hours after surgery, or when hemostasis is achieved (Table 1).
PT is recommended for rivaroxaban detection. Conventional assays are not sensitive at low concentrations, and degree of prolongation does not reliably predict amount of medication present.3,9 However, normal PT corresponds to a drug concentration of about 30 ng/mL and is considered safe for patients undergoing surgical intervention without increased risk for bleeding.6 This recommendation was extrapolated from data in the ROCKET AF study of patients who underwent elective surgeries while on rivaroxaban.6 Commercially available chromogenic anti–factor Xa assays, used with a rivaroxaban calibration curve, are sensitive and specific for rivaroxaban plasma concentrations.3,8 However, these assays are not widely available.
If a bleeding complication occurs in a patient who is being treated with rivaroxaban, the next rivaroxaban dose should be delayed, or treatment should be discontinued, as appropriate.8 Urgency of surgery should be weighed against risk for bleeding complications on a case-by-case basis. This decision is deferred to the clinical judgment of the surgeon. In the case of a patient with severe, life-threatening bleeding or a patient who requires emergent surgery, PCC 25-50 IU/kg is the recommended reversal agent.9 Recombinant factor VIIa and aPCC have been used in experimental settings, but there is concern about the greater prothrombotic potential of these agents compared with PCC8 (Table 2).
Apixaban
Apixaban is the second factor Xa inhibitor introduced in the United States and the first to show—in the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) study—efficacy superior to that of warfarin for the prevention of stroke and systemic embolism, all-cause mortality, and major bleeding. Furthermore, in the AVERROES (Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment) study, apixaban used in AF patients who were deemed not suitable for warfarin proved to be more effective than aspirin for stroke prevention, and had a similar rate of major bleeding.10 Apixaban is administered in a 5-mg dose 2 times daily. It has a half-life of 10 to 14 hours, is highly protein-bound, and has predominantly fecal excretion (27% is renal). Apixaban can prolong PT, but the correlation is nonlinear. Barrett and colleagues11 found that chromogenic anti–factor Xa assays provided the most accurate readings of apixaban plasma concentrations. Normal anti–factor Xa activity in patients being treated with apixaban suggests low drug levels and an intact hemostatic function, which are indicators of low bleeding risk with surgical intervention3 (Table 1).
Similar to other NOACs, apixaban has no antidote. In vitro testing showed that PCC improved thrombin generation when added to the blood of healthy donors who had received apixaban. Despite the lack of clinical experience, use of PCC 50 IU/kg may be reasonable for apixaban patients with severe or life-threatening bleeding3 (Table 2). Unlike dabigatran, apixaban cannot be eliminated with dialysis because of its high degree of protein binding. In nonemergent circumstances, delaying surgery 24 to 48 hours is considered effective in reducing the concentration of apixaban to a range that does not cause additional risk for bleeding.
Conclusion
Compared with warfarin, the NOACs dabigatran, rivaroxaban, and apixaban are efficacious and safe. Because of their steady pharmacokinetics, they do not require regular coagulation testing, as is the case with warfarin. These NOACs have been approved for the prevention of stroke and thromboembolic events in patients with nonvalvular AF; rivaroxaban has also been approved for VTE prevention after total hip or knee arthroplasty, for DVT treatment, and for prevention of recurrent VTE after acute DVT. Other options for VTE prophylaxis after hip and knee surgery are addressed in the guidelines issued by the American Academy of Orthopaedic Surgeons in 2011.12 As the incidence of chronic anticoagulation continues to increase among patients undergoing TJA, orthopedic surgeons need to be aware of the mechanism of action of these NOACs, as well as their pharmacokinetics and available reversal agents. Aggarwal and colleagues1 found that AF patients undergoing TJA had longer hospital stays, increased transfusion requirements, and increased risk for periprosthetic joint infection and unplanned hospital readmission.
The anticoagulation tests recommended for evaluation of hemostasis and drug reversal are normalization of aPTT for dabigatran; PT for rivaroxaban; and chromogenic anti–factor Xa activity for apixaban3 (Table 2). Although several research projects are being planned to develop an antidote for these medications, no antidote has been approved for human trials. The coagulation agents currently being used for reversal of NOACs are nonactivated PCC (Kcentra, Bebulin) and aPCC. Kcentra is a 4-factor PCC (II, VII, IX, X), and Bebulin is a 3-factor PCC (II, IX, X). Most authors recommend using 4-factor PCC 25 to 50 IU/kg. In vivo studies and animal studies have shown that nanofiltered aPCC (Feiba NF) at doses of 30 to 50 IU/kg can to some extent reverse anticoagulation in patients receiving NOACs. The current, limited data support use of reversal agent PCC for rivaroxaban and apixaban (no human studies for apixaban) and use of aPCC for dabigatran.3,6,8 Activated charcoal can be used for patients who have taken dabigatran <6 hours before presentation.3 Hemodialysis is another option for dabigatran removal. Hemodialysis, however, takes 4 to 6 hours or longer to remove about 60% of the medication (Table 2).3,5
In major orthopedic surgeries, such as TJA, bleeding is a critical concern. Using reversal agents to overcome the anticoagulation effect adds to the potential concern for thromboembolism secondary to these agents. Therefore, in cases in which surgery cannot be delayed any longer, the decision to use reversal agents should be made on a case-by-case basis. For most patients on rivaroxaban or apixaban, it is sufficient to delay for 24 to 48 hours before proceeding safely with surgery; for dabigatran, a delay of 3 to 4 days is recommended. Delay before surgery may need to be extended for the elderly and for patients with renal failure. The pharmacokinetics of these medications is summarized in Table 1.
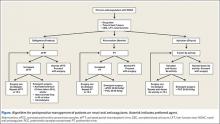
There are no guidelines for perioperative management of patients undergoing elective, urgent, or emergent surgeries while on NOACs. As discussed, Pernod and colleagues6 proposed better perioperative management of major bleeding risks in patients receiving rivaroxaban or dabigatran. Adapting their approach, and using the data available from the medical literature, we propose a perioperative algorithm that can guide practicing orthopedic surgeons performing urgent and emergent surgeries (Figure).
The population of patients receiving chronic anticoagulation therapy is growing, and anticoagulant and antiplatelet options are increasing in the United States and around the world. We propose a team approach for patient care, with orthopedic surgeon and cardiologist or vascular medicine specialist collaborating to ensure the safety and effectiveness of this treatment.
1. Aggarwal VK, Tischler EH, Post ZD, Kane I, Orozco FR, Ong A. Patients with atrial fibrillation undergoing total joint arthroplasty increase hospital burden. J Bone Joint Surg Am. 2013;95(17):1606-1611.
2. Curtis AB. Practice implications of the atrial fibrillation guidelines. Am J Cardiol. 2013;111(11):1660-1670.
3. Siegal DM, Crowther MA. Acute management of bleeding in patients on novel oral anticoagulants. Eur Heart J. 2013;34(7):489-498b.
4. van Ryn J, Stangier J, Haertter S, et al. Dabigatran etexilate—a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity. Thromb Haemost. 2010;103(6):1116-1127.
5. Lillo-Le Louët A, Wolf M, Soufir L, et al. Life-threatening bleeding in four patients with an unusual excessive response to dabigatran: implications for emergency surgery and resuscitation. Thromb Haemost. 2012;108(3):583-585.
6. Pernod G, Albaladejo P, Godier A, et al; Working Group on Perioperative Haemostasis. Management of major bleeding complications and emergency surgery in patients on long-term treatment with direct oral anticoagulants, thrombin or factor-Xa inhibitors: proposals of the Working Group on Perioperative Haemostasis (GIHP) - March 2013. Arch Cardiovasc Dis. 2013;106(6-7):382-393.
7. Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med. 2015;373(6):511-520.
8. Turpie AG, Kreutz R, Llau J, Norrving B, Haas S. Management consensus guidance for the use of rivaroxaban: an oral, direct factor Xa inhibitor. Thromb Haemost. 2012;108(5):876-886.
9. Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011;124(14):1573-1579.
10. Yates SW. Apixaban for stroke prevention in atrial fibrillation: a review of the clinical trial evidence. Hosp Pract. 2011;39(4):7-16.
11. Barrett YC, Wang Z, Frost C, Shenker A. Clinical laboratory measurement of direct factor Xa inhibitors: anti-Xa assay is preferable to prothrombin time assay. Thromb Haemost. 2010;104(6):1263-1271.
12. American Academy of Orthopaedic Surgeons. American Academy of Orthopaedic Surgeons clinical practice guideline on preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. Agency for Healthcare Research and Quality website. http://www.guideline.gov/content.aspx?id=35173. Released 2007. Revised 2011. Accessed March 21, 2016.
Chronic anticoagulation is a common preexisting condition in patients undergoing total joint arthroplasty (TJA). Atrial fibrillation (AF), the most common underlying disorder requiring chronic anticoagulation, affects more than 3 million patients in the United States—a number that is projected to increase to 16 million by 2050.1,2 Other common indications for anticoagulation are deep vein thrombosis (DVT) treatment, presence of a prosthetic heart valve, and venous thromboembolism (VTE) prevention after hip or knee arthroplasty. These patients face the additional risks of hemorrhage, persistent wound drainage, hematoma formation, transfusion requirements, periprosthetic joint infection, and longer hospital stay.1 Chronic anticoagulation traditionally has been managed with warfarin, which inhibits production of the vitamin K–dependent clotting factors II, VII, IX, and X. However, the new novel oral anticoagulants (NOACs), which target individual factors in the clotting cascade, are gaining favor as chronic anticoagulant agents because of their ease of use and improved efficacy and safety. These agents include the factor IIA inhibitor dabigatran (Pradaxa) and the direct factor Xa inhibitors rivaroxaban (Xarelto) and apixaban (Eliquis).
Management of patients at risk for thromboembolism and bleeding issues, particularly within the context of elective, urgent, or emergent orthopedic surgeries, is an evolving area. Understanding the pharmacokinetics, conventional laboratory tests, dosing, and reversal methods for NOACs is important, especially because clinical data are limited and the treatment itself can cause clinically significant harm.
In this article, we review the medical literature on these medications, their mechanism of action, and their reversal agents, and outline a practical approach for managing patients during the perioperative period.
Dabigatran
In October 2010, dabigatran became the first NOAC approved by the US Food and Drug Administration (FDA) for the prevention of arterial thromboembolic events in patients with nonvalvular AF, on the basis of the results of the RELY (Randomized Evaluation of Long-Term Anticoagulation Therapy) trial. Dabigatran is an oral factor IIA (thrombin) inhibitor. From time of ingestion, dabigatran takes 1.25 to 3 hours to reach peak plasma concentration. It has a half-life of 12 to 14 hours, is excreted predominantly by the kidneys (80%), and is renally dosed. The usual dose is 150 mg 2 times daily if creatinine clearance (CrCl) is >30 mL/minute, or 75 mg 2 times daily if CrCl is 15 to 30 mL/minute.3 Dabigatran is not recommended for patients with CrCl <15 mL/minute.
Dabigatran affects prothrombin time (PT), activated partial thromboplastin time (aPTT), ecarin clotting time, and thrombin time, with the latter 2 providing the most accurate means of monitoring appropriate drug levels.3,4 Of the tests commonly used to assess coagulation hemostasis in hospitals, normalization of thrombin time and aPTT provide the most accurate results (Table 1). The pharmacokinetics of dabigatran mandate consideration of dose, time of ingestion relative to time of blood sampling, and renal function in the assessment of coagulation hemostasis.
For elective surgeries, the periprocedure recommendation for patients being treated with dabigatran is to discontinue the medication 3 to 4 days before an operation if CrCl is ≥50 mL/minute, or 4 to 5 days beforehand if CrCl is <50 mL/minute.3 There is no antidote for dabigatran. In an in vitro model, activated charcoal reduced 99.9% of dabigatran absorption after recent ingestion.3 According to case reports, acute hemodialysis successfully removed 60% of the medication after 6 hours.5 In patients with end-stage renal disease, hemodialysis removed up to 68% of active dabigatran after 4 hours.3
Pernod and colleagues6 proposed that urgent surgeries can proceed if the concentration of dabigatran is ≤30 ng/mL—equivalent to normal aPTT. Their dictum was extrapolated from the data of patients who underwent elective surgeries while being treated with dabigatran, as recorded during the RELY trial. According to Pernod and colleagues,6 if aPTT is increased (probable drug level, ≥30 ng/mL), surgery should be postponed for up to 12 hours, with aPTT checked again and the process repeated if the concentration of dabigatran is still elevated and surgery can continue to be delayed. In patients who require urgent surgical interventions, we previously utilized nanofiltered activated prothrombin complex concentrate (aPCC; Feiba NF) 30 to 50 IU/kg over prothrombin complex concentrate (PCC; Kcentra or Bebulin) 25 to 50 IU/kg, as supported by in vitro and animal model studies and anecdotal case reports. However, neither aPCC nor PCC fully corrects the abnormalities evident on hemostasis tests.3,6 In October 2015, the FDA approved Idarucizumab (Praxbind), an injectable monoclonal antibody fragment that binds to dabigatran, as a reversing agent for use in urgent/emergent settings. Recommendation is to administer two 50-ml bolus infusions, each containing 2.5 g of idarucizumab, no more than 15 minutes apart.7 Additionally, hemodialysis could be discussed before surgery, with the understanding that it will take a long time to reach the threshold of 30 ng/mL in these patients (Table 2).
Rivaroxaban
Rivaroxaban is an oral direct factor Xa inhibitor that was initially approved in November 2011 for the prevention of stroke and systemic embolism in patients with nonvalvular AF. Since then, clinical use of rivaroxaban has been expanded to include prevention of VTE after elective hip or knee arthroplasty as well as treatment of DVT and prevention of recurrent VTE after acute DVT. In the phase 3 ROCKET AF (Rivaroxaban Once-Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation) study, rivaroxaban 20 mg daily (CrCl, ≥50 mL/min) and rivaroxaban 15 mg daily (CrCl, 15-49 mL/min) were equally effective as warfarin. Compared with warfarin, rivaroxaban had a similar safety rate for bleeding and adverse events but fewer intracranial hemorrhage and fatal bleeding events.8 On the basis of the outcomes of the RECORD (Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Venous Thrombosis and Pulmonary Embolism) studies comparing rivaroxaban and enoxaparin sodium, rivaroxaban 10 mg daily was approved for the prevention of VTE and pulmonary embolism after elective hip or knee arthroplasty.8
The half-life of rivaroxaban is 5 to 9 hours in the young and 11 to 13 hours in the elderly.8 As rivaroxaban takes 2 to 4 hours after ingestion to reach peak plasma concentration, it is important to know the timing and the dose taken. Because of the short half-life and rapid onset of action of this medication, bridging with another anticoagulant is not required when rivaroxaban is discontinued before surgery or initiated after surgery.8 The recommendation is to withhold rivaroxaban for 24 to 48 hours before surgery and then to administer the first postoperative dose 6 to 10 hours after surgery, or when hemostasis is achieved (Table 1).
PT is recommended for rivaroxaban detection. Conventional assays are not sensitive at low concentrations, and degree of prolongation does not reliably predict amount of medication present.3,9 However, normal PT corresponds to a drug concentration of about 30 ng/mL and is considered safe for patients undergoing surgical intervention without increased risk for bleeding.6 This recommendation was extrapolated from data in the ROCKET AF study of patients who underwent elective surgeries while on rivaroxaban.6 Commercially available chromogenic anti–factor Xa assays, used with a rivaroxaban calibration curve, are sensitive and specific for rivaroxaban plasma concentrations.3,8 However, these assays are not widely available.
If a bleeding complication occurs in a patient who is being treated with rivaroxaban, the next rivaroxaban dose should be delayed, or treatment should be discontinued, as appropriate.8 Urgency of surgery should be weighed against risk for bleeding complications on a case-by-case basis. This decision is deferred to the clinical judgment of the surgeon. In the case of a patient with severe, life-threatening bleeding or a patient who requires emergent surgery, PCC 25-50 IU/kg is the recommended reversal agent.9 Recombinant factor VIIa and aPCC have been used in experimental settings, but there is concern about the greater prothrombotic potential of these agents compared with PCC8 (Table 2).
Apixaban
Apixaban is the second factor Xa inhibitor introduced in the United States and the first to show—in the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) study—efficacy superior to that of warfarin for the prevention of stroke and systemic embolism, all-cause mortality, and major bleeding. Furthermore, in the AVERROES (Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment) study, apixaban used in AF patients who were deemed not suitable for warfarin proved to be more effective than aspirin for stroke prevention, and had a similar rate of major bleeding.10 Apixaban is administered in a 5-mg dose 2 times daily. It has a half-life of 10 to 14 hours, is highly protein-bound, and has predominantly fecal excretion (27% is renal). Apixaban can prolong PT, but the correlation is nonlinear. Barrett and colleagues11 found that chromogenic anti–factor Xa assays provided the most accurate readings of apixaban plasma concentrations. Normal anti–factor Xa activity in patients being treated with apixaban suggests low drug levels and an intact hemostatic function, which are indicators of low bleeding risk with surgical intervention3 (Table 1).
Similar to other NOACs, apixaban has no antidote. In vitro testing showed that PCC improved thrombin generation when added to the blood of healthy donors who had received apixaban. Despite the lack of clinical experience, use of PCC 50 IU/kg may be reasonable for apixaban patients with severe or life-threatening bleeding3 (Table 2). Unlike dabigatran, apixaban cannot be eliminated with dialysis because of its high degree of protein binding. In nonemergent circumstances, delaying surgery 24 to 48 hours is considered effective in reducing the concentration of apixaban to a range that does not cause additional risk for bleeding.
Conclusion
Compared with warfarin, the NOACs dabigatran, rivaroxaban, and apixaban are efficacious and safe. Because of their steady pharmacokinetics, they do not require regular coagulation testing, as is the case with warfarin. These NOACs have been approved for the prevention of stroke and thromboembolic events in patients with nonvalvular AF; rivaroxaban has also been approved for VTE prevention after total hip or knee arthroplasty, for DVT treatment, and for prevention of recurrent VTE after acute DVT. Other options for VTE prophylaxis after hip and knee surgery are addressed in the guidelines issued by the American Academy of Orthopaedic Surgeons in 2011.12 As the incidence of chronic anticoagulation continues to increase among patients undergoing TJA, orthopedic surgeons need to be aware of the mechanism of action of these NOACs, as well as their pharmacokinetics and available reversal agents. Aggarwal and colleagues1 found that AF patients undergoing TJA had longer hospital stays, increased transfusion requirements, and increased risk for periprosthetic joint infection and unplanned hospital readmission.
The anticoagulation tests recommended for evaluation of hemostasis and drug reversal are normalization of aPTT for dabigatran; PT for rivaroxaban; and chromogenic anti–factor Xa activity for apixaban3 (Table 2). Although several research projects are being planned to develop an antidote for these medications, no antidote has been approved for human trials. The coagulation agents currently being used for reversal of NOACs are nonactivated PCC (Kcentra, Bebulin) and aPCC. Kcentra is a 4-factor PCC (II, VII, IX, X), and Bebulin is a 3-factor PCC (II, IX, X). Most authors recommend using 4-factor PCC 25 to 50 IU/kg. In vivo studies and animal studies have shown that nanofiltered aPCC (Feiba NF) at doses of 30 to 50 IU/kg can to some extent reverse anticoagulation in patients receiving NOACs. The current, limited data support use of reversal agent PCC for rivaroxaban and apixaban (no human studies for apixaban) and use of aPCC for dabigatran.3,6,8 Activated charcoal can be used for patients who have taken dabigatran <6 hours before presentation.3 Hemodialysis is another option for dabigatran removal. Hemodialysis, however, takes 4 to 6 hours or longer to remove about 60% of the medication (Table 2).3,5
In major orthopedic surgeries, such as TJA, bleeding is a critical concern. Using reversal agents to overcome the anticoagulation effect adds to the potential concern for thromboembolism secondary to these agents. Therefore, in cases in which surgery cannot be delayed any longer, the decision to use reversal agents should be made on a case-by-case basis. For most patients on rivaroxaban or apixaban, it is sufficient to delay for 24 to 48 hours before proceeding safely with surgery; for dabigatran, a delay of 3 to 4 days is recommended. Delay before surgery may need to be extended for the elderly and for patients with renal failure. The pharmacokinetics of these medications is summarized in Table 1.
There are no guidelines for perioperative management of patients undergoing elective, urgent, or emergent surgeries while on NOACs. As discussed, Pernod and colleagues6 proposed better perioperative management of major bleeding risks in patients receiving rivaroxaban or dabigatran. Adapting their approach, and using the data available from the medical literature, we propose a perioperative algorithm that can guide practicing orthopedic surgeons performing urgent and emergent surgeries (Figure).
The population of patients receiving chronic anticoagulation therapy is growing, and anticoagulant and antiplatelet options are increasing in the United States and around the world. We propose a team approach for patient care, with orthopedic surgeon and cardiologist or vascular medicine specialist collaborating to ensure the safety and effectiveness of this treatment.
Chronic anticoagulation is a common preexisting condition in patients undergoing total joint arthroplasty (TJA). Atrial fibrillation (AF), the most common underlying disorder requiring chronic anticoagulation, affects more than 3 million patients in the United States—a number that is projected to increase to 16 million by 2050.1,2 Other common indications for anticoagulation are deep vein thrombosis (DVT) treatment, presence of a prosthetic heart valve, and venous thromboembolism (VTE) prevention after hip or knee arthroplasty. These patients face the additional risks of hemorrhage, persistent wound drainage, hematoma formation, transfusion requirements, periprosthetic joint infection, and longer hospital stay.1 Chronic anticoagulation traditionally has been managed with warfarin, which inhibits production of the vitamin K–dependent clotting factors II, VII, IX, and X. However, the new novel oral anticoagulants (NOACs), which target individual factors in the clotting cascade, are gaining favor as chronic anticoagulant agents because of their ease of use and improved efficacy and safety. These agents include the factor IIA inhibitor dabigatran (Pradaxa) and the direct factor Xa inhibitors rivaroxaban (Xarelto) and apixaban (Eliquis).
Management of patients at risk for thromboembolism and bleeding issues, particularly within the context of elective, urgent, or emergent orthopedic surgeries, is an evolving area. Understanding the pharmacokinetics, conventional laboratory tests, dosing, and reversal methods for NOACs is important, especially because clinical data are limited and the treatment itself can cause clinically significant harm.
In this article, we review the medical literature on these medications, their mechanism of action, and their reversal agents, and outline a practical approach for managing patients during the perioperative period.
Dabigatran
In October 2010, dabigatran became the first NOAC approved by the US Food and Drug Administration (FDA) for the prevention of arterial thromboembolic events in patients with nonvalvular AF, on the basis of the results of the RELY (Randomized Evaluation of Long-Term Anticoagulation Therapy) trial. Dabigatran is an oral factor IIA (thrombin) inhibitor. From time of ingestion, dabigatran takes 1.25 to 3 hours to reach peak plasma concentration. It has a half-life of 12 to 14 hours, is excreted predominantly by the kidneys (80%), and is renally dosed. The usual dose is 150 mg 2 times daily if creatinine clearance (CrCl) is >30 mL/minute, or 75 mg 2 times daily if CrCl is 15 to 30 mL/minute.3 Dabigatran is not recommended for patients with CrCl <15 mL/minute.
Dabigatran affects prothrombin time (PT), activated partial thromboplastin time (aPTT), ecarin clotting time, and thrombin time, with the latter 2 providing the most accurate means of monitoring appropriate drug levels.3,4 Of the tests commonly used to assess coagulation hemostasis in hospitals, normalization of thrombin time and aPTT provide the most accurate results (Table 1). The pharmacokinetics of dabigatran mandate consideration of dose, time of ingestion relative to time of blood sampling, and renal function in the assessment of coagulation hemostasis.
For elective surgeries, the periprocedure recommendation for patients being treated with dabigatran is to discontinue the medication 3 to 4 days before an operation if CrCl is ≥50 mL/minute, or 4 to 5 days beforehand if CrCl is <50 mL/minute.3 There is no antidote for dabigatran. In an in vitro model, activated charcoal reduced 99.9% of dabigatran absorption after recent ingestion.3 According to case reports, acute hemodialysis successfully removed 60% of the medication after 6 hours.5 In patients with end-stage renal disease, hemodialysis removed up to 68% of active dabigatran after 4 hours.3
Pernod and colleagues6 proposed that urgent surgeries can proceed if the concentration of dabigatran is ≤30 ng/mL—equivalent to normal aPTT. Their dictum was extrapolated from the data of patients who underwent elective surgeries while being treated with dabigatran, as recorded during the RELY trial. According to Pernod and colleagues,6 if aPTT is increased (probable drug level, ≥30 ng/mL), surgery should be postponed for up to 12 hours, with aPTT checked again and the process repeated if the concentration of dabigatran is still elevated and surgery can continue to be delayed. In patients who require urgent surgical interventions, we previously utilized nanofiltered activated prothrombin complex concentrate (aPCC; Feiba NF) 30 to 50 IU/kg over prothrombin complex concentrate (PCC; Kcentra or Bebulin) 25 to 50 IU/kg, as supported by in vitro and animal model studies and anecdotal case reports. However, neither aPCC nor PCC fully corrects the abnormalities evident on hemostasis tests.3,6 In October 2015, the FDA approved Idarucizumab (Praxbind), an injectable monoclonal antibody fragment that binds to dabigatran, as a reversing agent for use in urgent/emergent settings. Recommendation is to administer two 50-ml bolus infusions, each containing 2.5 g of idarucizumab, no more than 15 minutes apart.7 Additionally, hemodialysis could be discussed before surgery, with the understanding that it will take a long time to reach the threshold of 30 ng/mL in these patients (Table 2).
Rivaroxaban
Rivaroxaban is an oral direct factor Xa inhibitor that was initially approved in November 2011 for the prevention of stroke and systemic embolism in patients with nonvalvular AF. Since then, clinical use of rivaroxaban has been expanded to include prevention of VTE after elective hip or knee arthroplasty as well as treatment of DVT and prevention of recurrent VTE after acute DVT. In the phase 3 ROCKET AF (Rivaroxaban Once-Daily Oral Direct Factor Xa Inhibition Compared With Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation) study, rivaroxaban 20 mg daily (CrCl, ≥50 mL/min) and rivaroxaban 15 mg daily (CrCl, 15-49 mL/min) were equally effective as warfarin. Compared with warfarin, rivaroxaban had a similar safety rate for bleeding and adverse events but fewer intracranial hemorrhage and fatal bleeding events.8 On the basis of the outcomes of the RECORD (Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Venous Thrombosis and Pulmonary Embolism) studies comparing rivaroxaban and enoxaparin sodium, rivaroxaban 10 mg daily was approved for the prevention of VTE and pulmonary embolism after elective hip or knee arthroplasty.8
The half-life of rivaroxaban is 5 to 9 hours in the young and 11 to 13 hours in the elderly.8 As rivaroxaban takes 2 to 4 hours after ingestion to reach peak plasma concentration, it is important to know the timing and the dose taken. Because of the short half-life and rapid onset of action of this medication, bridging with another anticoagulant is not required when rivaroxaban is discontinued before surgery or initiated after surgery.8 The recommendation is to withhold rivaroxaban for 24 to 48 hours before surgery and then to administer the first postoperative dose 6 to 10 hours after surgery, or when hemostasis is achieved (Table 1).
PT is recommended for rivaroxaban detection. Conventional assays are not sensitive at low concentrations, and degree of prolongation does not reliably predict amount of medication present.3,9 However, normal PT corresponds to a drug concentration of about 30 ng/mL and is considered safe for patients undergoing surgical intervention without increased risk for bleeding.6 This recommendation was extrapolated from data in the ROCKET AF study of patients who underwent elective surgeries while on rivaroxaban.6 Commercially available chromogenic anti–factor Xa assays, used with a rivaroxaban calibration curve, are sensitive and specific for rivaroxaban plasma concentrations.3,8 However, these assays are not widely available.
If a bleeding complication occurs in a patient who is being treated with rivaroxaban, the next rivaroxaban dose should be delayed, or treatment should be discontinued, as appropriate.8 Urgency of surgery should be weighed against risk for bleeding complications on a case-by-case basis. This decision is deferred to the clinical judgment of the surgeon. In the case of a patient with severe, life-threatening bleeding or a patient who requires emergent surgery, PCC 25-50 IU/kg is the recommended reversal agent.9 Recombinant factor VIIa and aPCC have been used in experimental settings, but there is concern about the greater prothrombotic potential of these agents compared with PCC8 (Table 2).
Apixaban
Apixaban is the second factor Xa inhibitor introduced in the United States and the first to show—in the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) study—efficacy superior to that of warfarin for the prevention of stroke and systemic embolism, all-cause mortality, and major bleeding. Furthermore, in the AVERROES (Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment) study, apixaban used in AF patients who were deemed not suitable for warfarin proved to be more effective than aspirin for stroke prevention, and had a similar rate of major bleeding.10 Apixaban is administered in a 5-mg dose 2 times daily. It has a half-life of 10 to 14 hours, is highly protein-bound, and has predominantly fecal excretion (27% is renal). Apixaban can prolong PT, but the correlation is nonlinear. Barrett and colleagues11 found that chromogenic anti–factor Xa assays provided the most accurate readings of apixaban plasma concentrations. Normal anti–factor Xa activity in patients being treated with apixaban suggests low drug levels and an intact hemostatic function, which are indicators of low bleeding risk with surgical intervention3 (Table 1).
Similar to other NOACs, apixaban has no antidote. In vitro testing showed that PCC improved thrombin generation when added to the blood of healthy donors who had received apixaban. Despite the lack of clinical experience, use of PCC 50 IU/kg may be reasonable for apixaban patients with severe or life-threatening bleeding3 (Table 2). Unlike dabigatran, apixaban cannot be eliminated with dialysis because of its high degree of protein binding. In nonemergent circumstances, delaying surgery 24 to 48 hours is considered effective in reducing the concentration of apixaban to a range that does not cause additional risk for bleeding.
Conclusion
Compared with warfarin, the NOACs dabigatran, rivaroxaban, and apixaban are efficacious and safe. Because of their steady pharmacokinetics, they do not require regular coagulation testing, as is the case with warfarin. These NOACs have been approved for the prevention of stroke and thromboembolic events in patients with nonvalvular AF; rivaroxaban has also been approved for VTE prevention after total hip or knee arthroplasty, for DVT treatment, and for prevention of recurrent VTE after acute DVT. Other options for VTE prophylaxis after hip and knee surgery are addressed in the guidelines issued by the American Academy of Orthopaedic Surgeons in 2011.12 As the incidence of chronic anticoagulation continues to increase among patients undergoing TJA, orthopedic surgeons need to be aware of the mechanism of action of these NOACs, as well as their pharmacokinetics and available reversal agents. Aggarwal and colleagues1 found that AF patients undergoing TJA had longer hospital stays, increased transfusion requirements, and increased risk for periprosthetic joint infection and unplanned hospital readmission.
The anticoagulation tests recommended for evaluation of hemostasis and drug reversal are normalization of aPTT for dabigatran; PT for rivaroxaban; and chromogenic anti–factor Xa activity for apixaban3 (Table 2). Although several research projects are being planned to develop an antidote for these medications, no antidote has been approved for human trials. The coagulation agents currently being used for reversal of NOACs are nonactivated PCC (Kcentra, Bebulin) and aPCC. Kcentra is a 4-factor PCC (II, VII, IX, X), and Bebulin is a 3-factor PCC (II, IX, X). Most authors recommend using 4-factor PCC 25 to 50 IU/kg. In vivo studies and animal studies have shown that nanofiltered aPCC (Feiba NF) at doses of 30 to 50 IU/kg can to some extent reverse anticoagulation in patients receiving NOACs. The current, limited data support use of reversal agent PCC for rivaroxaban and apixaban (no human studies for apixaban) and use of aPCC for dabigatran.3,6,8 Activated charcoal can be used for patients who have taken dabigatran <6 hours before presentation.3 Hemodialysis is another option for dabigatran removal. Hemodialysis, however, takes 4 to 6 hours or longer to remove about 60% of the medication (Table 2).3,5
In major orthopedic surgeries, such as TJA, bleeding is a critical concern. Using reversal agents to overcome the anticoagulation effect adds to the potential concern for thromboembolism secondary to these agents. Therefore, in cases in which surgery cannot be delayed any longer, the decision to use reversal agents should be made on a case-by-case basis. For most patients on rivaroxaban or apixaban, it is sufficient to delay for 24 to 48 hours before proceeding safely with surgery; for dabigatran, a delay of 3 to 4 days is recommended. Delay before surgery may need to be extended for the elderly and for patients with renal failure. The pharmacokinetics of these medications is summarized in Table 1.
There are no guidelines for perioperative management of patients undergoing elective, urgent, or emergent surgeries while on NOACs. As discussed, Pernod and colleagues6 proposed better perioperative management of major bleeding risks in patients receiving rivaroxaban or dabigatran. Adapting their approach, and using the data available from the medical literature, we propose a perioperative algorithm that can guide practicing orthopedic surgeons performing urgent and emergent surgeries (Figure).
The population of patients receiving chronic anticoagulation therapy is growing, and anticoagulant and antiplatelet options are increasing in the United States and around the world. We propose a team approach for patient care, with orthopedic surgeon and cardiologist or vascular medicine specialist collaborating to ensure the safety and effectiveness of this treatment.
1. Aggarwal VK, Tischler EH, Post ZD, Kane I, Orozco FR, Ong A. Patients with atrial fibrillation undergoing total joint arthroplasty increase hospital burden. J Bone Joint Surg Am. 2013;95(17):1606-1611.
2. Curtis AB. Practice implications of the atrial fibrillation guidelines. Am J Cardiol. 2013;111(11):1660-1670.
3. Siegal DM, Crowther MA. Acute management of bleeding in patients on novel oral anticoagulants. Eur Heart J. 2013;34(7):489-498b.
4. van Ryn J, Stangier J, Haertter S, et al. Dabigatran etexilate—a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity. Thromb Haemost. 2010;103(6):1116-1127.
5. Lillo-Le Louët A, Wolf M, Soufir L, et al. Life-threatening bleeding in four patients with an unusual excessive response to dabigatran: implications for emergency surgery and resuscitation. Thromb Haemost. 2012;108(3):583-585.
6. Pernod G, Albaladejo P, Godier A, et al; Working Group on Perioperative Haemostasis. Management of major bleeding complications and emergency surgery in patients on long-term treatment with direct oral anticoagulants, thrombin or factor-Xa inhibitors: proposals of the Working Group on Perioperative Haemostasis (GIHP) - March 2013. Arch Cardiovasc Dis. 2013;106(6-7):382-393.
7. Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med. 2015;373(6):511-520.
8. Turpie AG, Kreutz R, Llau J, Norrving B, Haas S. Management consensus guidance for the use of rivaroxaban: an oral, direct factor Xa inhibitor. Thromb Haemost. 2012;108(5):876-886.
9. Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011;124(14):1573-1579.
10. Yates SW. Apixaban for stroke prevention in atrial fibrillation: a review of the clinical trial evidence. Hosp Pract. 2011;39(4):7-16.
11. Barrett YC, Wang Z, Frost C, Shenker A. Clinical laboratory measurement of direct factor Xa inhibitors: anti-Xa assay is preferable to prothrombin time assay. Thromb Haemost. 2010;104(6):1263-1271.
12. American Academy of Orthopaedic Surgeons. American Academy of Orthopaedic Surgeons clinical practice guideline on preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. Agency for Healthcare Research and Quality website. http://www.guideline.gov/content.aspx?id=35173. Released 2007. Revised 2011. Accessed March 21, 2016.
1. Aggarwal VK, Tischler EH, Post ZD, Kane I, Orozco FR, Ong A. Patients with atrial fibrillation undergoing total joint arthroplasty increase hospital burden. J Bone Joint Surg Am. 2013;95(17):1606-1611.
2. Curtis AB. Practice implications of the atrial fibrillation guidelines. Am J Cardiol. 2013;111(11):1660-1670.
3. Siegal DM, Crowther MA. Acute management of bleeding in patients on novel oral anticoagulants. Eur Heart J. 2013;34(7):489-498b.
4. van Ryn J, Stangier J, Haertter S, et al. Dabigatran etexilate—a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity. Thromb Haemost. 2010;103(6):1116-1127.
5. Lillo-Le Louët A, Wolf M, Soufir L, et al. Life-threatening bleeding in four patients with an unusual excessive response to dabigatran: implications for emergency surgery and resuscitation. Thromb Haemost. 2012;108(3):583-585.
6. Pernod G, Albaladejo P, Godier A, et al; Working Group on Perioperative Haemostasis. Management of major bleeding complications and emergency surgery in patients on long-term treatment with direct oral anticoagulants, thrombin or factor-Xa inhibitors: proposals of the Working Group on Perioperative Haemostasis (GIHP) - March 2013. Arch Cardiovasc Dis. 2013;106(6-7):382-393.
7. Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med. 2015;373(6):511-520.
8. Turpie AG, Kreutz R, Llau J, Norrving B, Haas S. Management consensus guidance for the use of rivaroxaban: an oral, direct factor Xa inhibitor. Thromb Haemost. 2012;108(5):876-886.
9. Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011;124(14):1573-1579.
10. Yates SW. Apixaban for stroke prevention in atrial fibrillation: a review of the clinical trial evidence. Hosp Pract. 2011;39(4):7-16.
11. Barrett YC, Wang Z, Frost C, Shenker A. Clinical laboratory measurement of direct factor Xa inhibitors: anti-Xa assay is preferable to prothrombin time assay. Thromb Haemost. 2010;104(6):1263-1271.
12. American Academy of Orthopaedic Surgeons. American Academy of Orthopaedic Surgeons clinical practice guideline on preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. Agency for Healthcare Research and Quality website. http://www.guideline.gov/content.aspx?id=35173. Released 2007. Revised 2011. Accessed March 21, 2016.
Total Hip Arthroplasty After Proximal Femoral Osteotomy: A Technique That Can Be Used to Address Presence of a Retained Intracortical Plate
Total hip arthroplasty (THA) is an effective treatment for advanced hip arthritis from a variety of causes, including osteoarthritis, inflammatory arthritis, posttraumatic arthritis, and sequelae of developmental disorders. It is not uncommon to perform THA in the presence of a previous proximal femoral osteotomy that may have been performed for slipped capital femoral epiphysis (SCFE), Legg-Calvé-Perthes disease, or developmental dysplasia of the hip, among other conditions. These osteotomies are commonly combined with internal fixation, a plate-and-screw device. These patients are at risk for developing degenerative arthritis at an earlier age than patients with other types of arthritis and subsequently may undergo THA at a younger age.1-3 Presence of a plate can pose a technical challenge during THA surgery. THA performed after intertrochanteric osteotomy has higher rates of perioperative and postoperative complications.4 Ferguson and colleagues4 noted difficulty during hardware removal in 24% of cases. Among the complications encountered were broken hardware, stripped screws, greater trochanteric fracture, stress risers from previous screw holes, canal narrowing from endosteal hypertrophy around hardware, and lateral cortical deficiency after removal of the side plate. As intertrochanteric osteotomies are often performed in patients who have yet to reach skeletal maturity, cortical hypertrophy can lead to complete coverage of the side plate and an “intracortical” position.
This article reports on 2 THA cases in which a technique was used to avoid intracortical plate removal and the resulting problems of lateral cortical deficiency. During each THA, the plate was left in place to avoid compromise of the lateral femoral cortex. The patients provided written informed consent for print and electronic publication of these case reports.
Case Reports
Case 1
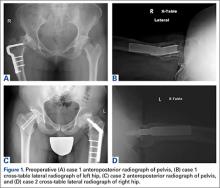
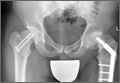
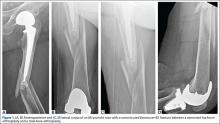
An adolescent with bilateral SCFE was treated first with internal fixation of the right hip and subsequently with left proximal femoral osteotomy with internal fixation. He did well until age 31 years, when he developed progressively worsening pain about the left hip. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the left hip. Radiographs showed a sliding hip screw in place, with proximal femoral deformity consisting of femoral neck shortening and posterior angulation (Figures 1A, 1B). Preoperative Harris Hip Score was 54.5.
Case 2
A 51-year-old woman presented with a history of right hip problems dating back to age 13 years, when she sustained a fracture of the right hip and was treated with internal fixation. At age 15 years, she underwent proximal femoral osteotomy to correct residual deformity. She did well until age 45 years, when she developed worsening hip symptoms. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the right hip. Radiographs showed a fixed-angle blade plate in the proximal femur, with significant proximal femoral deformity (Figures 1C, 1D). Preoperative Harris Hip Score was 53.6.
Surgical Technique
In both cases, a standard series of radiographs was obtained—an anteroposterior (AP) radiograph of the pelvis and AP and cross-table lateral radiographs of the operative hip (Figure 1). Computed tomography (CT) with a metal-artifact-reducing technique may be useful in determining amount of cortical bone remaining under the plate. CT showed limited lateral cortex beneath the side plate and bony overgrowth covering the side plate. Preoperative templating was performed using previously described techniques.5
During THA, before removing any portion of any retained hardware, the surgeon should perform 3 important actions: Dislocate the hip, perform all appropriate capsular releases, and reduce the hip. Dislocating the hip before hardware removal significantly decreases the risk for fracture caused by stress risers, as the force required for dislocation is much more controlled because of the capsular releases. After hardware removal, the hip can be easily redislocated, and the femoral neck osteotomy can be performed.
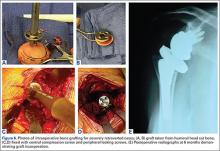
When plate and screws are in an intracortical position, the screws can be removed only after removing the small shell of cortical bone covering them. The amount of bone to be removed is minimal. After the screws are removed, the plate remains in place. A motorized device with a metal-cutting attachment is used to transect the construct at the junction of the plate and barrel (case 1) or at the bend of a fixed-angle device (case 2). Laparotomy sponges are placed around the proximal femur to minimize the amount of soft tissue that could be exposed to metal shavings. Copious irrigation is used throughout this part of the procedure. Osteotomes are used to elevate the proximal portion of the plate and the barrel, preserving the distal portion of the plate on the lateral cortex of the femoral shaft.
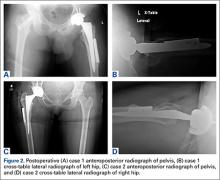
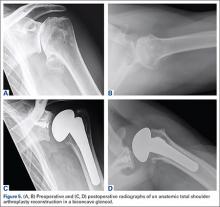
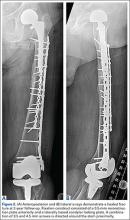
After the head is removed, the rest of the THA can be performed using standard press-fit insertion technique (Figures 2A-2D). Care must be taken to ensure that the distal aspect of the femoral stem bypasses the most distal screw hole by at least 2 cortical diameters in order to reduce the risk for periprosthetic fracture.
By 2-year follow-up, both patients had regained excellent range of motion, ambulation, and overall function. Postoperative Harris Hip Scores were 86.6 and 83.8, respectively. There were no radiographic signs of complications.
Discussion
THA can be challenging in the setting of previously placed internal fixation devices, particularly devices inserted during a patient’s adolescence, as significant bony overgrowth can occur. The standard approach has been to remove the internal fixation device and then perform the THA. In most cases, and particularly when the internal fixation device is in an intracortical position, the result is significant compromise of bone. This article describes a technique in which a portion of the hardware is retained to avoid compromise of the lateral femoral cortex, thereby allowing insertion of a noncemented femoral component.
THA is the most effective procedure for reducing hip pain and disability in the setting of degenerative changes.6 Patients with SCFE, Legg-Calvé-Perthes disease, or developmental dysplasia of the hip generally are younger at the time they may be sufficiently symptomatic to consider THA.7,8 Many have had previous surgery using internal fixation devices. THAs after previous osteotomies with internal fixation devices are more technically demanding, require more operative time, are subject to more blood loss, and have a higher rate of complications, including femoral fracture. Ferguson and colleagues4 and Boos and colleagues9 found these surgeries were more difficult 33.8% and 36.8% of the time, respectively. For these reasons, some authors have recommended removing the internal fixation device as soon as the osteotomy is healed.4 However, this has not become the standard of care, and surgeons continue to perform THAs in the presence of a previous osteotomy with an internal fixation device in place.
The technique described in this article was used successfully in 2 cases. In each case, leaving the intracortical plate in place avoided compromise of the lateral femoral cortex and allowed insertion of a noncemented femoral component without complication. Of course, with the screw holes representing stress risers, careful insertion of the femoral component was required. Retaining the intracortical plate allowed it to function as part of the lateral femoral cortex, thereby maintaining the structural integrity of the femoral canal. As has been described for the 2 cases, a blade plate and plate and barrel were converted to a limited intracortical plate by removing the proximal portion of the plates—a modification that could be applied to other types of internal fixation devices that extend into the femoral neck as long as appropriate cutting tools are available.
Conclusion
THA in the setting of a retained internal fixation device is relatively common. This article describes a technique that can be used when a plate applied to the lateral femoral cortex has become intracortical as a result of extensive bony overgrowth. In using this technique to avoid plate removal, the surgeon eliminates the need for more extensive procedures aimed at compensating for deficiency of the femoral cortex in the area of plate removal. Although only 2 cases are presented here, this technique potentially can be used more broadly in these specific clinical situations.
1. Engesæter LB, Engesæter IØ, Fenstad AM, et al. Low revision rate after total hip arthroplasty in patients with pediatric hip diseases. Acta Orthop. 2012;83(5):436-441.
2. Froberg L, Christensen F, Pedersen NW, Overgaard S. The need for total hip arthroplasty in Perthes disease: a long-term study. Clin Orthop Relat Res. 2011;469(4):1134-1140.
3. Furnes O, Lie SA, Espehaug B, Vollset SE, Engesæter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586.
4. Ferguson GM, Cabanela ME, Ilstrup DM. Total hip arthroplasty after failed intertrochanteric osteotomy. J Bone Joint Surg Br. 1994;76(2):252-257.
5. Scheerlinck T. Primary hip arthroplasty templating on standard radiographs. A stepwise approach. Acta Orthop Belg. 2010;76(4):432-442.
6. Wroblewski BM, Siney PD. Charnley low-friction arthroplasty of the hip. Long-term results. Clin Orthop Relat Res. 1993;(292):191-201.
7. Chandler HP, Reineck FT, Wixson RL, McCarthy JC. Total hip replacement in patients younger than thirty years old. A five-year follow-up study. J Bone Joint Surg Am. 1981;63(9):1426-1434.
8. Dorr LD, Luckett M, Conaty JP. Total hip arthroplasties in patients younger than 45 years. A nine- to ten-year follow-up study. Clin Orthop Relat Res. 1990;(260):215-219.
9. Boos N, Krushell R, Ganz R, Müller ME. Total hip arthroplasty after previous proximal femoral osteotomy. J Bone Joint Surg Br. 1997;79(2):247-253.
Total hip arthroplasty (THA) is an effective treatment for advanced hip arthritis from a variety of causes, including osteoarthritis, inflammatory arthritis, posttraumatic arthritis, and sequelae of developmental disorders. It is not uncommon to perform THA in the presence of a previous proximal femoral osteotomy that may have been performed for slipped capital femoral epiphysis (SCFE), Legg-Calvé-Perthes disease, or developmental dysplasia of the hip, among other conditions. These osteotomies are commonly combined with internal fixation, a plate-and-screw device. These patients are at risk for developing degenerative arthritis at an earlier age than patients with other types of arthritis and subsequently may undergo THA at a younger age.1-3 Presence of a plate can pose a technical challenge during THA surgery. THA performed after intertrochanteric osteotomy has higher rates of perioperative and postoperative complications.4 Ferguson and colleagues4 noted difficulty during hardware removal in 24% of cases. Among the complications encountered were broken hardware, stripped screws, greater trochanteric fracture, stress risers from previous screw holes, canal narrowing from endosteal hypertrophy around hardware, and lateral cortical deficiency after removal of the side plate. As intertrochanteric osteotomies are often performed in patients who have yet to reach skeletal maturity, cortical hypertrophy can lead to complete coverage of the side plate and an “intracortical” position.
This article reports on 2 THA cases in which a technique was used to avoid intracortical plate removal and the resulting problems of lateral cortical deficiency. During each THA, the plate was left in place to avoid compromise of the lateral femoral cortex. The patients provided written informed consent for print and electronic publication of these case reports.
Case Reports
Case 1
An adolescent with bilateral SCFE was treated first with internal fixation of the right hip and subsequently with left proximal femoral osteotomy with internal fixation. He did well until age 31 years, when he developed progressively worsening pain about the left hip. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the left hip. Radiographs showed a sliding hip screw in place, with proximal femoral deformity consisting of femoral neck shortening and posterior angulation (Figures 1A, 1B). Preoperative Harris Hip Score was 54.5.
Case 2
A 51-year-old woman presented with a history of right hip problems dating back to age 13 years, when she sustained a fracture of the right hip and was treated with internal fixation. At age 15 years, she underwent proximal femoral osteotomy to correct residual deformity. She did well until age 45 years, when she developed worsening hip symptoms. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the right hip. Radiographs showed a fixed-angle blade plate in the proximal femur, with significant proximal femoral deformity (Figures 1C, 1D). Preoperative Harris Hip Score was 53.6.
Surgical Technique
In both cases, a standard series of radiographs was obtained—an anteroposterior (AP) radiograph of the pelvis and AP and cross-table lateral radiographs of the operative hip (Figure 1). Computed tomography (CT) with a metal-artifact-reducing technique may be useful in determining amount of cortical bone remaining under the plate. CT showed limited lateral cortex beneath the side plate and bony overgrowth covering the side plate. Preoperative templating was performed using previously described techniques.5
During THA, before removing any portion of any retained hardware, the surgeon should perform 3 important actions: Dislocate the hip, perform all appropriate capsular releases, and reduce the hip. Dislocating the hip before hardware removal significantly decreases the risk for fracture caused by stress risers, as the force required for dislocation is much more controlled because of the capsular releases. After hardware removal, the hip can be easily redislocated, and the femoral neck osteotomy can be performed.
When plate and screws are in an intracortical position, the screws can be removed only after removing the small shell of cortical bone covering them. The amount of bone to be removed is minimal. After the screws are removed, the plate remains in place. A motorized device with a metal-cutting attachment is used to transect the construct at the junction of the plate and barrel (case 1) or at the bend of a fixed-angle device (case 2). Laparotomy sponges are placed around the proximal femur to minimize the amount of soft tissue that could be exposed to metal shavings. Copious irrigation is used throughout this part of the procedure. Osteotomes are used to elevate the proximal portion of the plate and the barrel, preserving the distal portion of the plate on the lateral cortex of the femoral shaft.
After the head is removed, the rest of the THA can be performed using standard press-fit insertion technique (Figures 2A-2D). Care must be taken to ensure that the distal aspect of the femoral stem bypasses the most distal screw hole by at least 2 cortical diameters in order to reduce the risk for periprosthetic fracture.
By 2-year follow-up, both patients had regained excellent range of motion, ambulation, and overall function. Postoperative Harris Hip Scores were 86.6 and 83.8, respectively. There were no radiographic signs of complications.
Discussion
THA can be challenging in the setting of previously placed internal fixation devices, particularly devices inserted during a patient’s adolescence, as significant bony overgrowth can occur. The standard approach has been to remove the internal fixation device and then perform the THA. In most cases, and particularly when the internal fixation device is in an intracortical position, the result is significant compromise of bone. This article describes a technique in which a portion of the hardware is retained to avoid compromise of the lateral femoral cortex, thereby allowing insertion of a noncemented femoral component.
THA is the most effective procedure for reducing hip pain and disability in the setting of degenerative changes.6 Patients with SCFE, Legg-Calvé-Perthes disease, or developmental dysplasia of the hip generally are younger at the time they may be sufficiently symptomatic to consider THA.7,8 Many have had previous surgery using internal fixation devices. THAs after previous osteotomies with internal fixation devices are more technically demanding, require more operative time, are subject to more blood loss, and have a higher rate of complications, including femoral fracture. Ferguson and colleagues4 and Boos and colleagues9 found these surgeries were more difficult 33.8% and 36.8% of the time, respectively. For these reasons, some authors have recommended removing the internal fixation device as soon as the osteotomy is healed.4 However, this has not become the standard of care, and surgeons continue to perform THAs in the presence of a previous osteotomy with an internal fixation device in place.
The technique described in this article was used successfully in 2 cases. In each case, leaving the intracortical plate in place avoided compromise of the lateral femoral cortex and allowed insertion of a noncemented femoral component without complication. Of course, with the screw holes representing stress risers, careful insertion of the femoral component was required. Retaining the intracortical plate allowed it to function as part of the lateral femoral cortex, thereby maintaining the structural integrity of the femoral canal. As has been described for the 2 cases, a blade plate and plate and barrel were converted to a limited intracortical plate by removing the proximal portion of the plates—a modification that could be applied to other types of internal fixation devices that extend into the femoral neck as long as appropriate cutting tools are available.
Conclusion
THA in the setting of a retained internal fixation device is relatively common. This article describes a technique that can be used when a plate applied to the lateral femoral cortex has become intracortical as a result of extensive bony overgrowth. In using this technique to avoid plate removal, the surgeon eliminates the need for more extensive procedures aimed at compensating for deficiency of the femoral cortex in the area of plate removal. Although only 2 cases are presented here, this technique potentially can be used more broadly in these specific clinical situations.
Total hip arthroplasty (THA) is an effective treatment for advanced hip arthritis from a variety of causes, including osteoarthritis, inflammatory arthritis, posttraumatic arthritis, and sequelae of developmental disorders. It is not uncommon to perform THA in the presence of a previous proximal femoral osteotomy that may have been performed for slipped capital femoral epiphysis (SCFE), Legg-Calvé-Perthes disease, or developmental dysplasia of the hip, among other conditions. These osteotomies are commonly combined with internal fixation, a plate-and-screw device. These patients are at risk for developing degenerative arthritis at an earlier age than patients with other types of arthritis and subsequently may undergo THA at a younger age.1-3 Presence of a plate can pose a technical challenge during THA surgery. THA performed after intertrochanteric osteotomy has higher rates of perioperative and postoperative complications.4 Ferguson and colleagues4 noted difficulty during hardware removal in 24% of cases. Among the complications encountered were broken hardware, stripped screws, greater trochanteric fracture, stress risers from previous screw holes, canal narrowing from endosteal hypertrophy around hardware, and lateral cortical deficiency after removal of the side plate. As intertrochanteric osteotomies are often performed in patients who have yet to reach skeletal maturity, cortical hypertrophy can lead to complete coverage of the side plate and an “intracortical” position.
This article reports on 2 THA cases in which a technique was used to avoid intracortical plate removal and the resulting problems of lateral cortical deficiency. During each THA, the plate was left in place to avoid compromise of the lateral femoral cortex. The patients provided written informed consent for print and electronic publication of these case reports.
Case Reports
Case 1
An adolescent with bilateral SCFE was treated first with internal fixation of the right hip and subsequently with left proximal femoral osteotomy with internal fixation. He did well until age 31 years, when he developed progressively worsening pain about the left hip. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the left hip. Radiographs showed a sliding hip screw in place, with proximal femoral deformity consisting of femoral neck shortening and posterior angulation (Figures 1A, 1B). Preoperative Harris Hip Score was 54.5.
Case 2
A 51-year-old woman presented with a history of right hip problems dating back to age 13 years, when she sustained a fracture of the right hip and was treated with internal fixation. At age 15 years, she underwent proximal femoral osteotomy to correct residual deformity. She did well until age 45 years, when she developed worsening hip symptoms. Clinical findings and imaging studies were consistent with advanced degenerative arthritis of the right hip. Radiographs showed a fixed-angle blade plate in the proximal femur, with significant proximal femoral deformity (Figures 1C, 1D). Preoperative Harris Hip Score was 53.6.
Surgical Technique
In both cases, a standard series of radiographs was obtained—an anteroposterior (AP) radiograph of the pelvis and AP and cross-table lateral radiographs of the operative hip (Figure 1). Computed tomography (CT) with a metal-artifact-reducing technique may be useful in determining amount of cortical bone remaining under the plate. CT showed limited lateral cortex beneath the side plate and bony overgrowth covering the side plate. Preoperative templating was performed using previously described techniques.5
During THA, before removing any portion of any retained hardware, the surgeon should perform 3 important actions: Dislocate the hip, perform all appropriate capsular releases, and reduce the hip. Dislocating the hip before hardware removal significantly decreases the risk for fracture caused by stress risers, as the force required for dislocation is much more controlled because of the capsular releases. After hardware removal, the hip can be easily redislocated, and the femoral neck osteotomy can be performed.
When plate and screws are in an intracortical position, the screws can be removed only after removing the small shell of cortical bone covering them. The amount of bone to be removed is minimal. After the screws are removed, the plate remains in place. A motorized device with a metal-cutting attachment is used to transect the construct at the junction of the plate and barrel (case 1) or at the bend of a fixed-angle device (case 2). Laparotomy sponges are placed around the proximal femur to minimize the amount of soft tissue that could be exposed to metal shavings. Copious irrigation is used throughout this part of the procedure. Osteotomes are used to elevate the proximal portion of the plate and the barrel, preserving the distal portion of the plate on the lateral cortex of the femoral shaft.
After the head is removed, the rest of the THA can be performed using standard press-fit insertion technique (Figures 2A-2D). Care must be taken to ensure that the distal aspect of the femoral stem bypasses the most distal screw hole by at least 2 cortical diameters in order to reduce the risk for periprosthetic fracture.
By 2-year follow-up, both patients had regained excellent range of motion, ambulation, and overall function. Postoperative Harris Hip Scores were 86.6 and 83.8, respectively. There were no radiographic signs of complications.
Discussion
THA can be challenging in the setting of previously placed internal fixation devices, particularly devices inserted during a patient’s adolescence, as significant bony overgrowth can occur. The standard approach has been to remove the internal fixation device and then perform the THA. In most cases, and particularly when the internal fixation device is in an intracortical position, the result is significant compromise of bone. This article describes a technique in which a portion of the hardware is retained to avoid compromise of the lateral femoral cortex, thereby allowing insertion of a noncemented femoral component.
THA is the most effective procedure for reducing hip pain and disability in the setting of degenerative changes.6 Patients with SCFE, Legg-Calvé-Perthes disease, or developmental dysplasia of the hip generally are younger at the time they may be sufficiently symptomatic to consider THA.7,8 Many have had previous surgery using internal fixation devices. THAs after previous osteotomies with internal fixation devices are more technically demanding, require more operative time, are subject to more blood loss, and have a higher rate of complications, including femoral fracture. Ferguson and colleagues4 and Boos and colleagues9 found these surgeries were more difficult 33.8% and 36.8% of the time, respectively. For these reasons, some authors have recommended removing the internal fixation device as soon as the osteotomy is healed.4 However, this has not become the standard of care, and surgeons continue to perform THAs in the presence of a previous osteotomy with an internal fixation device in place.
The technique described in this article was used successfully in 2 cases. In each case, leaving the intracortical plate in place avoided compromise of the lateral femoral cortex and allowed insertion of a noncemented femoral component without complication. Of course, with the screw holes representing stress risers, careful insertion of the femoral component was required. Retaining the intracortical plate allowed it to function as part of the lateral femoral cortex, thereby maintaining the structural integrity of the femoral canal. As has been described for the 2 cases, a blade plate and plate and barrel were converted to a limited intracortical plate by removing the proximal portion of the plates—a modification that could be applied to other types of internal fixation devices that extend into the femoral neck as long as appropriate cutting tools are available.
Conclusion
THA in the setting of a retained internal fixation device is relatively common. This article describes a technique that can be used when a plate applied to the lateral femoral cortex has become intracortical as a result of extensive bony overgrowth. In using this technique to avoid plate removal, the surgeon eliminates the need for more extensive procedures aimed at compensating for deficiency of the femoral cortex in the area of plate removal. Although only 2 cases are presented here, this technique potentially can be used more broadly in these specific clinical situations.
1. Engesæter LB, Engesæter IØ, Fenstad AM, et al. Low revision rate after total hip arthroplasty in patients with pediatric hip diseases. Acta Orthop. 2012;83(5):436-441.
2. Froberg L, Christensen F, Pedersen NW, Overgaard S. The need for total hip arthroplasty in Perthes disease: a long-term study. Clin Orthop Relat Res. 2011;469(4):1134-1140.
3. Furnes O, Lie SA, Espehaug B, Vollset SE, Engesæter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586.
4. Ferguson GM, Cabanela ME, Ilstrup DM. Total hip arthroplasty after failed intertrochanteric osteotomy. J Bone Joint Surg Br. 1994;76(2):252-257.
5. Scheerlinck T. Primary hip arthroplasty templating on standard radiographs. A stepwise approach. Acta Orthop Belg. 2010;76(4):432-442.
6. Wroblewski BM, Siney PD. Charnley low-friction arthroplasty of the hip. Long-term results. Clin Orthop Relat Res. 1993;(292):191-201.
7. Chandler HP, Reineck FT, Wixson RL, McCarthy JC. Total hip replacement in patients younger than thirty years old. A five-year follow-up study. J Bone Joint Surg Am. 1981;63(9):1426-1434.
8. Dorr LD, Luckett M, Conaty JP. Total hip arthroplasties in patients younger than 45 years. A nine- to ten-year follow-up study. Clin Orthop Relat Res. 1990;(260):215-219.
9. Boos N, Krushell R, Ganz R, Müller ME. Total hip arthroplasty after previous proximal femoral osteotomy. J Bone Joint Surg Br. 1997;79(2):247-253.
1. Engesæter LB, Engesæter IØ, Fenstad AM, et al. Low revision rate after total hip arthroplasty in patients with pediatric hip diseases. Acta Orthop. 2012;83(5):436-441.
2. Froberg L, Christensen F, Pedersen NW, Overgaard S. The need for total hip arthroplasty in Perthes disease: a long-term study. Clin Orthop Relat Res. 2011;469(4):1134-1140.
3. Furnes O, Lie SA, Espehaug B, Vollset SE, Engesæter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586.
4. Ferguson GM, Cabanela ME, Ilstrup DM. Total hip arthroplasty after failed intertrochanteric osteotomy. J Bone Joint Surg Br. 1994;76(2):252-257.
5. Scheerlinck T. Primary hip arthroplasty templating on standard radiographs. A stepwise approach. Acta Orthop Belg. 2010;76(4):432-442.
6. Wroblewski BM, Siney PD. Charnley low-friction arthroplasty of the hip. Long-term results. Clin Orthop Relat Res. 1993;(292):191-201.
7. Chandler HP, Reineck FT, Wixson RL, McCarthy JC. Total hip replacement in patients younger than thirty years old. A five-year follow-up study. J Bone Joint Surg Am. 1981;63(9):1426-1434.
8. Dorr LD, Luckett M, Conaty JP. Total hip arthroplasties in patients younger than 45 years. A nine- to ten-year follow-up study. Clin Orthop Relat Res. 1990;(260):215-219.
9. Boos N, Krushell R, Ganz R, Müller ME. Total hip arthroplasty after previous proximal femoral osteotomy. J Bone Joint Surg Br. 1997;79(2):247-253.
A Retrospective Analysis of Hemostatic Techniques in Primary Total Knee Arthroplasty: Traditional Electrocautery, Bipolar Sealer, and Argon Beam Coagulation
Total knee arthroplasty (TKA) is a reliable and successful treatment for end-stage degenerative joint disease of the knee. Given the reproducibility of its generally excellent outcomes, TKA is increasingly being performed.1 However, the potential complications of this procedure can be devastating.2-4 The arthroplasty literature has shed light on the detrimental effects of postoperative blood loss and anemia.5,6 In addition, the increase in transfusion burden among patients is not without risk.7 Given these concerns, surgeons have been tasked with determining the ideal methods for minimizing blood transfusions and postoperative hematomas and anemia. Several strategies have been described.8-11 Hemostasis can be achieved with use of intravenous medications, intra-articular agents, or electrocautery devices. Electrocautery technologies include traditional electrocautery (TE), saline-coupled bipolar sealer (BS), and argon beam coagulation (ABC). There is controversy as to whether outcomes are better with one hemostasis method over another and whether these methods are worth the additional cost.
In traditional (Bovie) electrocautery, a unipolar device delivers an electrical current to tissues through a pencil-like instrument. Intraoperative tissue temperatures can exceed 400°C.12 In BS, radiofrequency energy is delivered through a saline medium, which increases the contact area, acts as an electrode, and maintains a cooler environment during electrocautery. Proposed advantages are reduced tissue destruction and absence of smoke.12 There is evidence both for10,12-16 and against17-20 use of BS in total joint arthroplasty. ABC, a novel hemostasis method, has been studied in the context of orthopedics21,22 but not TKA specifically. ABC establishes a monopolar electric circuit between a handheld device and the target tissues by channeling electrons through ionized argon gas. Hemostasis is achieved through thermal coagulation. Tissue penetration can be adjusted by changing power, probe-to-target distance, and duration of use.23 We conducted a study to assess the efficacy of all 3 electrocautery methods during TKA. We hypothesized the 3 methods would be clinically equivalent with respect to estimated blood loss (EBL), 48-hour wound drainage, operative time, and change from preoperative hemoglobin (Hb) level.
Methods
We conducted a retrospective cohort study of consecutive primary TKAs performed by Dr. Levine between October 2010 and November 2011. Patients were identified by querying an internal database. Exclusion criteria were prior ipsilateral open knee procedure, prior fracture, nonuse of our standard hemostatic protocol, and either tourniquet time under 40 minutes or intraoperative documentation of tourniquet failure. As only 9 patients were initially identified for the TE cohort, the same database was used to add 32 patients treated between April 2009 and October 2009 (before our institution began using BS and ABC).
Clinical charts were reviewed, and baseline demographics (age, body mass index [BMI], preoperative Hb level) were abstracted, as were outcome metrics (EBL, 48-hour wound drainage, operative time, postoperative transfusions, adverse events (AEs) before discharge, and change in Hb level from before surgery to after surgery, in recovery room and on discharge). Statistical analyses were performed with JMP Version 10.0.0 (SAS Institute). Given the hypothesis that the 3 hemostasis methods would be clinically equivalent, 2 one-sided tests (TOSTs) of equivalence were performed with an α of 0.05. With TOST, the traditional null and alternative hypotheses are reversed; thus, P < .05 identifies statistical equivalence. The advantage of this study design is that equivalence can be identified, whereas traditional study designs can identify only a lack of statistical difference.24 We used our consensus opinions to set clinical insignificance thresholds for EBL (150 mL), wound drainage (150 mL), decrease from postoperative Hb level (1 g/dL), and operative time (10 minutes). Patients who received a blood transfusion were subsequently excluded from analysis in order to avoid skewing Hb-level depreciation calculations. Analysis of variance (ANOVA) and χ2 tests were used to compare preoperative variables, transfusion requirements, hospital length of stay, and AE rates by hemostasis type.
Cautery Technique
In all cases, TE was used for surgical dissection, which followed a standard midvastus approach. Then, for meniscal excision, the capsule and meniscal attachment sites were treated with TE, BS, or ABC. During cement hardening, an available supplemental cautery option was used to achieve hemostasis of the suprapatellar fat pad and visible meniscal attachment sites. All other aspects of the procedure and the postoperative protocols—including the anticoagulation and rapid rehabilitation (early ambulation and therapy) protocols—were similar for all patients. The standard anticoagulation protocol was to use low-molecular-weight heparin, unless contraindicated. Tranexamic acid was not used at our institution during the study period.
Results
For the study period, 280 cases (41 TE, 203 BS, 36 ABC) met the inclusion criteria. Of the 280 TKAs, 261 (93.21%) were performed for degenerative arthritis. There was no statistically significant difference among cohorts in indication (χ2 = 1.841, P = .398) or sex (χ2 = 1.176, P= .555).
Table 1 lists the cohorts’ baseline demographics (mean age, BMI, preoperative Hb level) and comparative ANOVA results. TOSTs of equivalence were performed to compare operative time, EBL, 48-hour wound drainage, and postoperative Hb-level depreciation among hemostasis types. Changes in Hb level were calculated for the immediate postoperative period and time of discharge (Table 2). ANOVA of hospital length of stay demonstrated no significant difference in means among groups (P = .09).
The cohorts were compared with respect to use of postoperative transfusions and incidence of postoperative AEs (Table 3). The TE cohort did not have any AEs. Of the 203 BS patients, 14 (7%) had 1 or more AEs, which included acute kidney injury (3 cases), electrolyte disturbance (3), urinary tract infection (2), oxygen desaturation (2), altered mental status (1), pneumonia (1), arrhythmia (1), congestive heart failure exacerbation (1), dehiscence (1), pulmonary embolism (2), and hypotension (1). Of the 36 ABC patients, 1 (3%) had arrhythmia, pneumonia, sepsis, and altered mental status.
Discussion
With the population aging, the demand for TKA is greater than ever.1 As surgical volume increases, the ability to minimize the rates of intraoperative bleeding, postoperative anemia, and transfusion is becoming increasingly important to patients and the healthcare system. There is no consensus as to which cautery method is ideal. Other investigators have identified differences in clinical outcomes between cautery systems, but reported results are largely conflicting.10,12-20 In addition, no one has studied the utility of ABC in TKA. In the present retrospective cohort analysis, we hypothesized that TE, BS, and ABC would be clinically equivalent in primary TKA with respect to EBL, 48-hour wound drainage, operative time, and change from preoperative Hb level.
The data on hemostatic technology in primary TKA are inconclusive. In an age- and sex-matched study comparing TE and BS in primary TKA, BS used with shed blood autotransfusion reduced homologous blood transfusions by a factor of 5.16 In addition, BS patients lost significantly less total visible blood (intraoperative EBL, postoperative drain output), and their magnitude of postoperative Hb-level depreciations at time of discharge was significantly lower. In a multicenter, prospective randomized trial comparing TE with BS, adjusted blood loss and need for autologous blood transfusions were lower in BS patients,10 though there was no significant difference in Knee Society Scale scores between the 2 treatment arms. However, analysis was potentially biased in that multiple authors had financial ties to Salient Surgical Technologies, the manufacturer of the BS device used in the study. Other prospective randomized trials of patients who had primary TKA with either TE or BS did not find any significant difference in postoperative Hb level, postoperative drainage, or transfusion requirements.19 ABC has been studied in the context of orthopedics but not joint arthroplasty specifically. This technology was anecdotally identified as a means of attaining hemostasis in foot and ankle surgery after failure of TE and other conventional means.22 ABC has also been identified as a successful adjuvant to curettage in the treatment of aneurysmal bone cysts.21 However, ABC has not been compared with TE or BS in the orthopedic literature.
In the present study, analysis of preoperative variables revealed a statistically but not clinically significant difference in BMI among cohorts. Mean (SD) BMI was 35.6 (6.5) for TE patients, 35.8 (9.7) for BS patients, and 40.9 (11.3) for ABC patients. (Previously, BMI did not correlate with intraoperative blood loss in TKA.25) Analysis also revealed a statistically significant but clinically insignificant and inconsequential difference in Hb level among cohorts. Mean (SD) preoperative Hb level was 13.5 (1.6) g/dL for TE patients, 12.8 (1.4) g/dL for BS patients, and 13.0 (1.6) g/dL for ABC patients. As decreases from preoperative baseline Hb levels were the intended focus of analysis—not absolute Hb levels—this finding does not refute postoperative analyses.
Our results suggest that, though TE may have relatively longer operative times in primary TKA, it is clinically equivalent to BS and ABC with respect to EBL and postoperative change in Hb levels. In addition, postoperative drainage was lower in TE than in BS and ABC, which were equivalent. No significant differences were found among hemostasis types with respect to postoperative transfusion requirements.
The prevalence distribution of predischarge AEs trended toward significance (χ2 = 5.957, P = .051), despite not meeting the predetermined α level. Rates of predischarge AEs were 0% (0/41) for TE patients, 7% (14/203) for BS patients, and 3% (1/36) for ABC patients. AEs included acute kidney injuries, electrolyte disturbances, urinary tract infections, oxygen desaturation, altered mental status, sepsis/infections, arrhythmias, congestive heart failure exacerbation, dehiscence, pulmonary embolism, and hypotension. Clearly, many of these AEs are not attributable to the hemostasis system used.
Limitations of this study include its retrospective design, documentation inadequate to account for drainage amount reinfused, and limited data on which clinical insignificance thresholds were based. In addition, reliance on historical data may have introduced bias into the analysis. The historical data used to increase the size of the TE cohort may reflect a period of relative inexperience and may have contributed to the longer operative times relative to those of the ABC cohort (Dr. Levine used ABC later in his career).
Traditional electrocautery remains a viable option in primary TKA. With its low cost and hemostasis equivalent to that of BS and ABC, TE deserves consideration equal to that given to these more modern hemostasis technologies. Cost per case is about $10 for TE versus $500 for BS and $110 for ABC.17 Soaring healthcare expenditures may warrant returning to TE or combining cautery techniques and other agents in primary TKA in order to reduce the number of transfusions and associated surgical costs.
1. Cram P, Lu X, Kates SL, Singh JA, Li Y, Wolf BR. Total knee arthroplasty volume, utilization, and outcomes among Medicare beneficiaries, 1991-2010. JAMA. 2012;308(12):1227-1236.
2. Leijtens B, Kremers van de Hei K, Jansen J, Koëter S. High complication rate after total knee and hip replacement due to perioperative bridging of anticoagulant therapy based on the 2012 ACCP guideline. Arch Orthop Trauma Surg. 2014;134(9):1335-1341.
3. Park CH, Lee SH, Kang DG, Cho KY, Lee SH, Kim KI. Compartment syndrome following total knee arthroplasty: clinical results of late fasciotomy. Knee Surg Relat Res. 2014;26(3):177-181.
4. Pedersen AB, Mehnert F, Sorensen HT, Emmeluth C, Overgaard S, Johnsen SP. The risk of venous thromboembolism, myocardial infarction, stroke, major bleeding and death in patients undergoing total hip and knee replacement: a 15-year retrospective cohort study of routine clinical practice. Bone Joint J. 2014;96-B(4):479-485.
5. Carson JL, Poses RM, Spence RK, Bonavita G. Severity of anaemia and operative mortality and morbidity. Lancet. 1988;1(8588):727-729.
6. Carson JL, Duff A, Poses RM, et al. Effect of anaemia and cardiovascular disease on surgical mortality and morbidity. Lancet. 1996;348(9034):1055-1060.
7. Dodd RY. Current risk for transfusion transmitted infections. Curr Opin Hematol. 2007;14(6):671-676.
8. Kang DG, Khurana S, Baek JH, Park YS, Lee SH, Kim KI. Efficacy and safety using autotransfusion system with postoperative shed blood following total knee arthroplasty in haemophilia. Haemophilia. 2014;20(1):129-132.
9. Aguilera X, Martinez-Zapata MJ, Bosch A, et al. Efficacy and safety of fibrin glue and tranexamic acid to prevent postoperative blood loss in total knee arthroplasty: a randomized controlled clinical trial. J Bone Joint Surg Am. 2013;95(22):2001-2007.
10. Marulanda GA, Krebs VE, Bierbaum BE, et al. Hemostasis using a bipolar sealer in primary unilateral total knee arthroplasty. Am J Orthop. 2009;38(12):E179-E183.
11. Katkhouda N, Friedlander M, Darehzereshki A, et al. Argon beam coagulation versus fibrin sealant for hemostasis following liver resection: a randomized study in a porcine model. Hepatogastroenterology. 2013;60(125):1110-1116.
12. Marulanda GA, Ulrich SD, Seyler TM, Delanois RE, Mont MA. Reductions in blood loss with a bipolar sealer in total hip arthroplasty. Expert Rev Med Devices. 2008;5(2):125-131.
13. Morris MJ, Berend KR, Lombardi AV Jr. Hemostasis in anterior supine intermuscular total hip arthroplasty: pilot study comparing standard electrocautery and a bipolar sealer. Surg Technol Int. 2010;20:352-356.
14. Clement RC, Kamath AF, Derman PB, Garino JP, Lee GC. Bipolar sealing in revision total hip arthroplasty for infection: efficacy and cost analysis. J Arthroplasty. 2012;27(7):1376-1381.
15. Rosenberg AG. Reducing blood loss in total joint surgery with a saline-coupled bipolar sealing technology. J Arthroplasty. 2007;22(4 suppl 1):82-85.
16. Pfeiffer M, Bräutigam H, Draws D, Sigg A. A new bipolar blood sealing system embedded in perioperative strategies vs. a conventional regimen for total knee arthroplasty: results of a matched-pair study. Ger Med Sci. 2005;3:Doc10.
17. Morris MJ, Barrett M, Lombardi AV Jr, Tucker TL, Berend KR. Randomized blinded study comparing a bipolar sealer and standard electrocautery in reducing transfusion requirements in anterior supine intermuscular total hip arthroplasty. J Arthroplasty. 2013;28(9):1614-1617.
18. Barsoum WK, Klika AK, Murray TG, Higuera C, Lee HH, Krebs VE. Prospective randomized evaluation of the need for blood transfusion during primary total hip arthroplasty with use of a bipolar sealer. J Bone Joint Surg Am. 2011;93(6):513-518.
19. Plymale MF, Capogna BM, Lovy AJ, Adler ML, Hirsh DM, Kim SJ. Unipolar vs bipolar hemostasis in total knee arthroplasty: a prospective randomized trial. J Arthroplasty. 2012;27(6):1133-1137.e1.
20. Zeh A, Messer J, Davis J, Vasarhelyi A, Wohlrab D. The Aquamantys system—an alternative to reduce blood loss in primary total hip arthroplasty? J Arthroplasty. 2010;25(7):1072-1077.
21. Cummings JE, Smith RA, Heck RK Jr. Argon beam coagulation as adjuvant treatment after curettage of aneurysmal bone cysts: a preliminary study. Clin Orthop Relat Res. 2010;468(1):231-237.
22. Adams ML, Steinberg JS. Argon beam coagulation in foot and ankle surgery. J Foot Ankle Surg. 2011;50(6):780-782.
23. Neumayer L, Vargo D. Principles of preoperative and operative surgery. In: Townsend CM Jr, Beauchamp RD, Evers BM, Mattox KL, eds. Sabiston Textbook of Surgery. 19th ed. Philadelphia, PA: Elsevier Saunders; 2012:211-239.
24. Walker E, Nowacki AS. Understanding equivalence and noninferiority testing. J Gen Intern Med. 2011;26(2):192-196.
25. Hrnack SA, Skeen N, Xu T, Rosenstein AD. Correlation of body mass index and blood loss during total knee and total hip arthroplasty. Am J Orthop. 2012;41(10):467-471.
Total knee arthroplasty (TKA) is a reliable and successful treatment for end-stage degenerative joint disease of the knee. Given the reproducibility of its generally excellent outcomes, TKA is increasingly being performed.1 However, the potential complications of this procedure can be devastating.2-4 The arthroplasty literature has shed light on the detrimental effects of postoperative blood loss and anemia.5,6 In addition, the increase in transfusion burden among patients is not without risk.7 Given these concerns, surgeons have been tasked with determining the ideal methods for minimizing blood transfusions and postoperative hematomas and anemia. Several strategies have been described.8-11 Hemostasis can be achieved with use of intravenous medications, intra-articular agents, or electrocautery devices. Electrocautery technologies include traditional electrocautery (TE), saline-coupled bipolar sealer (BS), and argon beam coagulation (ABC). There is controversy as to whether outcomes are better with one hemostasis method over another and whether these methods are worth the additional cost.
In traditional (Bovie) electrocautery, a unipolar device delivers an electrical current to tissues through a pencil-like instrument. Intraoperative tissue temperatures can exceed 400°C.12 In BS, radiofrequency energy is delivered through a saline medium, which increases the contact area, acts as an electrode, and maintains a cooler environment during electrocautery. Proposed advantages are reduced tissue destruction and absence of smoke.12 There is evidence both for10,12-16 and against17-20 use of BS in total joint arthroplasty. ABC, a novel hemostasis method, has been studied in the context of orthopedics21,22 but not TKA specifically. ABC establishes a monopolar electric circuit between a handheld device and the target tissues by channeling electrons through ionized argon gas. Hemostasis is achieved through thermal coagulation. Tissue penetration can be adjusted by changing power, probe-to-target distance, and duration of use.23 We conducted a study to assess the efficacy of all 3 electrocautery methods during TKA. We hypothesized the 3 methods would be clinically equivalent with respect to estimated blood loss (EBL), 48-hour wound drainage, operative time, and change from preoperative hemoglobin (Hb) level.
Methods
We conducted a retrospective cohort study of consecutive primary TKAs performed by Dr. Levine between October 2010 and November 2011. Patients were identified by querying an internal database. Exclusion criteria were prior ipsilateral open knee procedure, prior fracture, nonuse of our standard hemostatic protocol, and either tourniquet time under 40 minutes or intraoperative documentation of tourniquet failure. As only 9 patients were initially identified for the TE cohort, the same database was used to add 32 patients treated between April 2009 and October 2009 (before our institution began using BS and ABC).
Clinical charts were reviewed, and baseline demographics (age, body mass index [BMI], preoperative Hb level) were abstracted, as were outcome metrics (EBL, 48-hour wound drainage, operative time, postoperative transfusions, adverse events (AEs) before discharge, and change in Hb level from before surgery to after surgery, in recovery room and on discharge). Statistical analyses were performed with JMP Version 10.0.0 (SAS Institute). Given the hypothesis that the 3 hemostasis methods would be clinically equivalent, 2 one-sided tests (TOSTs) of equivalence were performed with an α of 0.05. With TOST, the traditional null and alternative hypotheses are reversed; thus, P < .05 identifies statistical equivalence. The advantage of this study design is that equivalence can be identified, whereas traditional study designs can identify only a lack of statistical difference.24 We used our consensus opinions to set clinical insignificance thresholds for EBL (150 mL), wound drainage (150 mL), decrease from postoperative Hb level (1 g/dL), and operative time (10 minutes). Patients who received a blood transfusion were subsequently excluded from analysis in order to avoid skewing Hb-level depreciation calculations. Analysis of variance (ANOVA) and χ2 tests were used to compare preoperative variables, transfusion requirements, hospital length of stay, and AE rates by hemostasis type.
Cautery Technique
In all cases, TE was used for surgical dissection, which followed a standard midvastus approach. Then, for meniscal excision, the capsule and meniscal attachment sites were treated with TE, BS, or ABC. During cement hardening, an available supplemental cautery option was used to achieve hemostasis of the suprapatellar fat pad and visible meniscal attachment sites. All other aspects of the procedure and the postoperative protocols—including the anticoagulation and rapid rehabilitation (early ambulation and therapy) protocols—were similar for all patients. The standard anticoagulation protocol was to use low-molecular-weight heparin, unless contraindicated. Tranexamic acid was not used at our institution during the study period.
Results
For the study period, 280 cases (41 TE, 203 BS, 36 ABC) met the inclusion criteria. Of the 280 TKAs, 261 (93.21%) were performed for degenerative arthritis. There was no statistically significant difference among cohorts in indication (χ2 = 1.841, P = .398) or sex (χ2 = 1.176, P= .555).
Table 1 lists the cohorts’ baseline demographics (mean age, BMI, preoperative Hb level) and comparative ANOVA results. TOSTs of equivalence were performed to compare operative time, EBL, 48-hour wound drainage, and postoperative Hb-level depreciation among hemostasis types. Changes in Hb level were calculated for the immediate postoperative period and time of discharge (Table 2). ANOVA of hospital length of stay demonstrated no significant difference in means among groups (P = .09).
The cohorts were compared with respect to use of postoperative transfusions and incidence of postoperative AEs (Table 3). The TE cohort did not have any AEs. Of the 203 BS patients, 14 (7%) had 1 or more AEs, which included acute kidney injury (3 cases), electrolyte disturbance (3), urinary tract infection (2), oxygen desaturation (2), altered mental status (1), pneumonia (1), arrhythmia (1), congestive heart failure exacerbation (1), dehiscence (1), pulmonary embolism (2), and hypotension (1). Of the 36 ABC patients, 1 (3%) had arrhythmia, pneumonia, sepsis, and altered mental status.
Discussion
With the population aging, the demand for TKA is greater than ever.1 As surgical volume increases, the ability to minimize the rates of intraoperative bleeding, postoperative anemia, and transfusion is becoming increasingly important to patients and the healthcare system. There is no consensus as to which cautery method is ideal. Other investigators have identified differences in clinical outcomes between cautery systems, but reported results are largely conflicting.10,12-20 In addition, no one has studied the utility of ABC in TKA. In the present retrospective cohort analysis, we hypothesized that TE, BS, and ABC would be clinically equivalent in primary TKA with respect to EBL, 48-hour wound drainage, operative time, and change from preoperative Hb level.
The data on hemostatic technology in primary TKA are inconclusive. In an age- and sex-matched study comparing TE and BS in primary TKA, BS used with shed blood autotransfusion reduced homologous blood transfusions by a factor of 5.16 In addition, BS patients lost significantly less total visible blood (intraoperative EBL, postoperative drain output), and their magnitude of postoperative Hb-level depreciations at time of discharge was significantly lower. In a multicenter, prospective randomized trial comparing TE with BS, adjusted blood loss and need for autologous blood transfusions were lower in BS patients,10 though there was no significant difference in Knee Society Scale scores between the 2 treatment arms. However, analysis was potentially biased in that multiple authors had financial ties to Salient Surgical Technologies, the manufacturer of the BS device used in the study. Other prospective randomized trials of patients who had primary TKA with either TE or BS did not find any significant difference in postoperative Hb level, postoperative drainage, or transfusion requirements.19 ABC has been studied in the context of orthopedics but not joint arthroplasty specifically. This technology was anecdotally identified as a means of attaining hemostasis in foot and ankle surgery after failure of TE and other conventional means.22 ABC has also been identified as a successful adjuvant to curettage in the treatment of aneurysmal bone cysts.21 However, ABC has not been compared with TE or BS in the orthopedic literature.
In the present study, analysis of preoperative variables revealed a statistically but not clinically significant difference in BMI among cohorts. Mean (SD) BMI was 35.6 (6.5) for TE patients, 35.8 (9.7) for BS patients, and 40.9 (11.3) for ABC patients. (Previously, BMI did not correlate with intraoperative blood loss in TKA.25) Analysis also revealed a statistically significant but clinically insignificant and inconsequential difference in Hb level among cohorts. Mean (SD) preoperative Hb level was 13.5 (1.6) g/dL for TE patients, 12.8 (1.4) g/dL for BS patients, and 13.0 (1.6) g/dL for ABC patients. As decreases from preoperative baseline Hb levels were the intended focus of analysis—not absolute Hb levels—this finding does not refute postoperative analyses.
Our results suggest that, though TE may have relatively longer operative times in primary TKA, it is clinically equivalent to BS and ABC with respect to EBL and postoperative change in Hb levels. In addition, postoperative drainage was lower in TE than in BS and ABC, which were equivalent. No significant differences were found among hemostasis types with respect to postoperative transfusion requirements.
The prevalence distribution of predischarge AEs trended toward significance (χ2 = 5.957, P = .051), despite not meeting the predetermined α level. Rates of predischarge AEs were 0% (0/41) for TE patients, 7% (14/203) for BS patients, and 3% (1/36) for ABC patients. AEs included acute kidney injuries, electrolyte disturbances, urinary tract infections, oxygen desaturation, altered mental status, sepsis/infections, arrhythmias, congestive heart failure exacerbation, dehiscence, pulmonary embolism, and hypotension. Clearly, many of these AEs are not attributable to the hemostasis system used.
Limitations of this study include its retrospective design, documentation inadequate to account for drainage amount reinfused, and limited data on which clinical insignificance thresholds were based. In addition, reliance on historical data may have introduced bias into the analysis. The historical data used to increase the size of the TE cohort may reflect a period of relative inexperience and may have contributed to the longer operative times relative to those of the ABC cohort (Dr. Levine used ABC later in his career).
Traditional electrocautery remains a viable option in primary TKA. With its low cost and hemostasis equivalent to that of BS and ABC, TE deserves consideration equal to that given to these more modern hemostasis technologies. Cost per case is about $10 for TE versus $500 for BS and $110 for ABC.17 Soaring healthcare expenditures may warrant returning to TE or combining cautery techniques and other agents in primary TKA in order to reduce the number of transfusions and associated surgical costs.
Total knee arthroplasty (TKA) is a reliable and successful treatment for end-stage degenerative joint disease of the knee. Given the reproducibility of its generally excellent outcomes, TKA is increasingly being performed.1 However, the potential complications of this procedure can be devastating.2-4 The arthroplasty literature has shed light on the detrimental effects of postoperative blood loss and anemia.5,6 In addition, the increase in transfusion burden among patients is not without risk.7 Given these concerns, surgeons have been tasked with determining the ideal methods for minimizing blood transfusions and postoperative hematomas and anemia. Several strategies have been described.8-11 Hemostasis can be achieved with use of intravenous medications, intra-articular agents, or electrocautery devices. Electrocautery technologies include traditional electrocautery (TE), saline-coupled bipolar sealer (BS), and argon beam coagulation (ABC). There is controversy as to whether outcomes are better with one hemostasis method over another and whether these methods are worth the additional cost.
In traditional (Bovie) electrocautery, a unipolar device delivers an electrical current to tissues through a pencil-like instrument. Intraoperative tissue temperatures can exceed 400°C.12 In BS, radiofrequency energy is delivered through a saline medium, which increases the contact area, acts as an electrode, and maintains a cooler environment during electrocautery. Proposed advantages are reduced tissue destruction and absence of smoke.12 There is evidence both for10,12-16 and against17-20 use of BS in total joint arthroplasty. ABC, a novel hemostasis method, has been studied in the context of orthopedics21,22 but not TKA specifically. ABC establishes a monopolar electric circuit between a handheld device and the target tissues by channeling electrons through ionized argon gas. Hemostasis is achieved through thermal coagulation. Tissue penetration can be adjusted by changing power, probe-to-target distance, and duration of use.23 We conducted a study to assess the efficacy of all 3 electrocautery methods during TKA. We hypothesized the 3 methods would be clinically equivalent with respect to estimated blood loss (EBL), 48-hour wound drainage, operative time, and change from preoperative hemoglobin (Hb) level.
Methods
We conducted a retrospective cohort study of consecutive primary TKAs performed by Dr. Levine between October 2010 and November 2011. Patients were identified by querying an internal database. Exclusion criteria were prior ipsilateral open knee procedure, prior fracture, nonuse of our standard hemostatic protocol, and either tourniquet time under 40 minutes or intraoperative documentation of tourniquet failure. As only 9 patients were initially identified for the TE cohort, the same database was used to add 32 patients treated between April 2009 and October 2009 (before our institution began using BS and ABC).
Clinical charts were reviewed, and baseline demographics (age, body mass index [BMI], preoperative Hb level) were abstracted, as were outcome metrics (EBL, 48-hour wound drainage, operative time, postoperative transfusions, adverse events (AEs) before discharge, and change in Hb level from before surgery to after surgery, in recovery room and on discharge). Statistical analyses were performed with JMP Version 10.0.0 (SAS Institute). Given the hypothesis that the 3 hemostasis methods would be clinically equivalent, 2 one-sided tests (TOSTs) of equivalence were performed with an α of 0.05. With TOST, the traditional null and alternative hypotheses are reversed; thus, P < .05 identifies statistical equivalence. The advantage of this study design is that equivalence can be identified, whereas traditional study designs can identify only a lack of statistical difference.24 We used our consensus opinions to set clinical insignificance thresholds for EBL (150 mL), wound drainage (150 mL), decrease from postoperative Hb level (1 g/dL), and operative time (10 minutes). Patients who received a blood transfusion were subsequently excluded from analysis in order to avoid skewing Hb-level depreciation calculations. Analysis of variance (ANOVA) and χ2 tests were used to compare preoperative variables, transfusion requirements, hospital length of stay, and AE rates by hemostasis type.
Cautery Technique
In all cases, TE was used for surgical dissection, which followed a standard midvastus approach. Then, for meniscal excision, the capsule and meniscal attachment sites were treated with TE, BS, or ABC. During cement hardening, an available supplemental cautery option was used to achieve hemostasis of the suprapatellar fat pad and visible meniscal attachment sites. All other aspects of the procedure and the postoperative protocols—including the anticoagulation and rapid rehabilitation (early ambulation and therapy) protocols—were similar for all patients. The standard anticoagulation protocol was to use low-molecular-weight heparin, unless contraindicated. Tranexamic acid was not used at our institution during the study period.
Results
For the study period, 280 cases (41 TE, 203 BS, 36 ABC) met the inclusion criteria. Of the 280 TKAs, 261 (93.21%) were performed for degenerative arthritis. There was no statistically significant difference among cohorts in indication (χ2 = 1.841, P = .398) or sex (χ2 = 1.176, P= .555).
Table 1 lists the cohorts’ baseline demographics (mean age, BMI, preoperative Hb level) and comparative ANOVA results. TOSTs of equivalence were performed to compare operative time, EBL, 48-hour wound drainage, and postoperative Hb-level depreciation among hemostasis types. Changes in Hb level were calculated for the immediate postoperative period and time of discharge (Table 2). ANOVA of hospital length of stay demonstrated no significant difference in means among groups (P = .09).
The cohorts were compared with respect to use of postoperative transfusions and incidence of postoperative AEs (Table 3). The TE cohort did not have any AEs. Of the 203 BS patients, 14 (7%) had 1 or more AEs, which included acute kidney injury (3 cases), electrolyte disturbance (3), urinary tract infection (2), oxygen desaturation (2), altered mental status (1), pneumonia (1), arrhythmia (1), congestive heart failure exacerbation (1), dehiscence (1), pulmonary embolism (2), and hypotension (1). Of the 36 ABC patients, 1 (3%) had arrhythmia, pneumonia, sepsis, and altered mental status.
Discussion
With the population aging, the demand for TKA is greater than ever.1 As surgical volume increases, the ability to minimize the rates of intraoperative bleeding, postoperative anemia, and transfusion is becoming increasingly important to patients and the healthcare system. There is no consensus as to which cautery method is ideal. Other investigators have identified differences in clinical outcomes between cautery systems, but reported results are largely conflicting.10,12-20 In addition, no one has studied the utility of ABC in TKA. In the present retrospective cohort analysis, we hypothesized that TE, BS, and ABC would be clinically equivalent in primary TKA with respect to EBL, 48-hour wound drainage, operative time, and change from preoperative Hb level.
The data on hemostatic technology in primary TKA are inconclusive. In an age- and sex-matched study comparing TE and BS in primary TKA, BS used with shed blood autotransfusion reduced homologous blood transfusions by a factor of 5.16 In addition, BS patients lost significantly less total visible blood (intraoperative EBL, postoperative drain output), and their magnitude of postoperative Hb-level depreciations at time of discharge was significantly lower. In a multicenter, prospective randomized trial comparing TE with BS, adjusted blood loss and need for autologous blood transfusions were lower in BS patients,10 though there was no significant difference in Knee Society Scale scores between the 2 treatment arms. However, analysis was potentially biased in that multiple authors had financial ties to Salient Surgical Technologies, the manufacturer of the BS device used in the study. Other prospective randomized trials of patients who had primary TKA with either TE or BS did not find any significant difference in postoperative Hb level, postoperative drainage, or transfusion requirements.19 ABC has been studied in the context of orthopedics but not joint arthroplasty specifically. This technology was anecdotally identified as a means of attaining hemostasis in foot and ankle surgery after failure of TE and other conventional means.22 ABC has also been identified as a successful adjuvant to curettage in the treatment of aneurysmal bone cysts.21 However, ABC has not been compared with TE or BS in the orthopedic literature.
In the present study, analysis of preoperative variables revealed a statistically but not clinically significant difference in BMI among cohorts. Mean (SD) BMI was 35.6 (6.5) for TE patients, 35.8 (9.7) for BS patients, and 40.9 (11.3) for ABC patients. (Previously, BMI did not correlate with intraoperative blood loss in TKA.25) Analysis also revealed a statistically significant but clinically insignificant and inconsequential difference in Hb level among cohorts. Mean (SD) preoperative Hb level was 13.5 (1.6) g/dL for TE patients, 12.8 (1.4) g/dL for BS patients, and 13.0 (1.6) g/dL for ABC patients. As decreases from preoperative baseline Hb levels were the intended focus of analysis—not absolute Hb levels—this finding does not refute postoperative analyses.
Our results suggest that, though TE may have relatively longer operative times in primary TKA, it is clinically equivalent to BS and ABC with respect to EBL and postoperative change in Hb levels. In addition, postoperative drainage was lower in TE than in BS and ABC, which were equivalent. No significant differences were found among hemostasis types with respect to postoperative transfusion requirements.
The prevalence distribution of predischarge AEs trended toward significance (χ2 = 5.957, P = .051), despite not meeting the predetermined α level. Rates of predischarge AEs were 0% (0/41) for TE patients, 7% (14/203) for BS patients, and 3% (1/36) for ABC patients. AEs included acute kidney injuries, electrolyte disturbances, urinary tract infections, oxygen desaturation, altered mental status, sepsis/infections, arrhythmias, congestive heart failure exacerbation, dehiscence, pulmonary embolism, and hypotension. Clearly, many of these AEs are not attributable to the hemostasis system used.
Limitations of this study include its retrospective design, documentation inadequate to account for drainage amount reinfused, and limited data on which clinical insignificance thresholds were based. In addition, reliance on historical data may have introduced bias into the analysis. The historical data used to increase the size of the TE cohort may reflect a period of relative inexperience and may have contributed to the longer operative times relative to those of the ABC cohort (Dr. Levine used ABC later in his career).
Traditional electrocautery remains a viable option in primary TKA. With its low cost and hemostasis equivalent to that of BS and ABC, TE deserves consideration equal to that given to these more modern hemostasis technologies. Cost per case is about $10 for TE versus $500 for BS and $110 for ABC.17 Soaring healthcare expenditures may warrant returning to TE or combining cautery techniques and other agents in primary TKA in order to reduce the number of transfusions and associated surgical costs.
1. Cram P, Lu X, Kates SL, Singh JA, Li Y, Wolf BR. Total knee arthroplasty volume, utilization, and outcomes among Medicare beneficiaries, 1991-2010. JAMA. 2012;308(12):1227-1236.
2. Leijtens B, Kremers van de Hei K, Jansen J, Koëter S. High complication rate after total knee and hip replacement due to perioperative bridging of anticoagulant therapy based on the 2012 ACCP guideline. Arch Orthop Trauma Surg. 2014;134(9):1335-1341.
3. Park CH, Lee SH, Kang DG, Cho KY, Lee SH, Kim KI. Compartment syndrome following total knee arthroplasty: clinical results of late fasciotomy. Knee Surg Relat Res. 2014;26(3):177-181.
4. Pedersen AB, Mehnert F, Sorensen HT, Emmeluth C, Overgaard S, Johnsen SP. The risk of venous thromboembolism, myocardial infarction, stroke, major bleeding and death in patients undergoing total hip and knee replacement: a 15-year retrospective cohort study of routine clinical practice. Bone Joint J. 2014;96-B(4):479-485.
5. Carson JL, Poses RM, Spence RK, Bonavita G. Severity of anaemia and operative mortality and morbidity. Lancet. 1988;1(8588):727-729.
6. Carson JL, Duff A, Poses RM, et al. Effect of anaemia and cardiovascular disease on surgical mortality and morbidity. Lancet. 1996;348(9034):1055-1060.
7. Dodd RY. Current risk for transfusion transmitted infections. Curr Opin Hematol. 2007;14(6):671-676.
8. Kang DG, Khurana S, Baek JH, Park YS, Lee SH, Kim KI. Efficacy and safety using autotransfusion system with postoperative shed blood following total knee arthroplasty in haemophilia. Haemophilia. 2014;20(1):129-132.
9. Aguilera X, Martinez-Zapata MJ, Bosch A, et al. Efficacy and safety of fibrin glue and tranexamic acid to prevent postoperative blood loss in total knee arthroplasty: a randomized controlled clinical trial. J Bone Joint Surg Am. 2013;95(22):2001-2007.
10. Marulanda GA, Krebs VE, Bierbaum BE, et al. Hemostasis using a bipolar sealer in primary unilateral total knee arthroplasty. Am J Orthop. 2009;38(12):E179-E183.
11. Katkhouda N, Friedlander M, Darehzereshki A, et al. Argon beam coagulation versus fibrin sealant for hemostasis following liver resection: a randomized study in a porcine model. Hepatogastroenterology. 2013;60(125):1110-1116.
12. Marulanda GA, Ulrich SD, Seyler TM, Delanois RE, Mont MA. Reductions in blood loss with a bipolar sealer in total hip arthroplasty. Expert Rev Med Devices. 2008;5(2):125-131.
13. Morris MJ, Berend KR, Lombardi AV Jr. Hemostasis in anterior supine intermuscular total hip arthroplasty: pilot study comparing standard electrocautery and a bipolar sealer. Surg Technol Int. 2010;20:352-356.
14. Clement RC, Kamath AF, Derman PB, Garino JP, Lee GC. Bipolar sealing in revision total hip arthroplasty for infection: efficacy and cost analysis. J Arthroplasty. 2012;27(7):1376-1381.
15. Rosenberg AG. Reducing blood loss in total joint surgery with a saline-coupled bipolar sealing technology. J Arthroplasty. 2007;22(4 suppl 1):82-85.
16. Pfeiffer M, Bräutigam H, Draws D, Sigg A. A new bipolar blood sealing system embedded in perioperative strategies vs. a conventional regimen for total knee arthroplasty: results of a matched-pair study. Ger Med Sci. 2005;3:Doc10.
17. Morris MJ, Barrett M, Lombardi AV Jr, Tucker TL, Berend KR. Randomized blinded study comparing a bipolar sealer and standard electrocautery in reducing transfusion requirements in anterior supine intermuscular total hip arthroplasty. J Arthroplasty. 2013;28(9):1614-1617.
18. Barsoum WK, Klika AK, Murray TG, Higuera C, Lee HH, Krebs VE. Prospective randomized evaluation of the need for blood transfusion during primary total hip arthroplasty with use of a bipolar sealer. J Bone Joint Surg Am. 2011;93(6):513-518.
19. Plymale MF, Capogna BM, Lovy AJ, Adler ML, Hirsh DM, Kim SJ. Unipolar vs bipolar hemostasis in total knee arthroplasty: a prospective randomized trial. J Arthroplasty. 2012;27(6):1133-1137.e1.
20. Zeh A, Messer J, Davis J, Vasarhelyi A, Wohlrab D. The Aquamantys system—an alternative to reduce blood loss in primary total hip arthroplasty? J Arthroplasty. 2010;25(7):1072-1077.
21. Cummings JE, Smith RA, Heck RK Jr. Argon beam coagulation as adjuvant treatment after curettage of aneurysmal bone cysts: a preliminary study. Clin Orthop Relat Res. 2010;468(1):231-237.
22. Adams ML, Steinberg JS. Argon beam coagulation in foot and ankle surgery. J Foot Ankle Surg. 2011;50(6):780-782.
23. Neumayer L, Vargo D. Principles of preoperative and operative surgery. In: Townsend CM Jr, Beauchamp RD, Evers BM, Mattox KL, eds. Sabiston Textbook of Surgery. 19th ed. Philadelphia, PA: Elsevier Saunders; 2012:211-239.
24. Walker E, Nowacki AS. Understanding equivalence and noninferiority testing. J Gen Intern Med. 2011;26(2):192-196.
25. Hrnack SA, Skeen N, Xu T, Rosenstein AD. Correlation of body mass index and blood loss during total knee and total hip arthroplasty. Am J Orthop. 2012;41(10):467-471.
1. Cram P, Lu X, Kates SL, Singh JA, Li Y, Wolf BR. Total knee arthroplasty volume, utilization, and outcomes among Medicare beneficiaries, 1991-2010. JAMA. 2012;308(12):1227-1236.
2. Leijtens B, Kremers van de Hei K, Jansen J, Koëter S. High complication rate after total knee and hip replacement due to perioperative bridging of anticoagulant therapy based on the 2012 ACCP guideline. Arch Orthop Trauma Surg. 2014;134(9):1335-1341.
3. Park CH, Lee SH, Kang DG, Cho KY, Lee SH, Kim KI. Compartment syndrome following total knee arthroplasty: clinical results of late fasciotomy. Knee Surg Relat Res. 2014;26(3):177-181.
4. Pedersen AB, Mehnert F, Sorensen HT, Emmeluth C, Overgaard S, Johnsen SP. The risk of venous thromboembolism, myocardial infarction, stroke, major bleeding and death in patients undergoing total hip and knee replacement: a 15-year retrospective cohort study of routine clinical practice. Bone Joint J. 2014;96-B(4):479-485.
5. Carson JL, Poses RM, Spence RK, Bonavita G. Severity of anaemia and operative mortality and morbidity. Lancet. 1988;1(8588):727-729.
6. Carson JL, Duff A, Poses RM, et al. Effect of anaemia and cardiovascular disease on surgical mortality and morbidity. Lancet. 1996;348(9034):1055-1060.
7. Dodd RY. Current risk for transfusion transmitted infections. Curr Opin Hematol. 2007;14(6):671-676.
8. Kang DG, Khurana S, Baek JH, Park YS, Lee SH, Kim KI. Efficacy and safety using autotransfusion system with postoperative shed blood following total knee arthroplasty in haemophilia. Haemophilia. 2014;20(1):129-132.
9. Aguilera X, Martinez-Zapata MJ, Bosch A, et al. Efficacy and safety of fibrin glue and tranexamic acid to prevent postoperative blood loss in total knee arthroplasty: a randomized controlled clinical trial. J Bone Joint Surg Am. 2013;95(22):2001-2007.
10. Marulanda GA, Krebs VE, Bierbaum BE, et al. Hemostasis using a bipolar sealer in primary unilateral total knee arthroplasty. Am J Orthop. 2009;38(12):E179-E183.
11. Katkhouda N, Friedlander M, Darehzereshki A, et al. Argon beam coagulation versus fibrin sealant for hemostasis following liver resection: a randomized study in a porcine model. Hepatogastroenterology. 2013;60(125):1110-1116.
12. Marulanda GA, Ulrich SD, Seyler TM, Delanois RE, Mont MA. Reductions in blood loss with a bipolar sealer in total hip arthroplasty. Expert Rev Med Devices. 2008;5(2):125-131.
13. Morris MJ, Berend KR, Lombardi AV Jr. Hemostasis in anterior supine intermuscular total hip arthroplasty: pilot study comparing standard electrocautery and a bipolar sealer. Surg Technol Int. 2010;20:352-356.
14. Clement RC, Kamath AF, Derman PB, Garino JP, Lee GC. Bipolar sealing in revision total hip arthroplasty for infection: efficacy and cost analysis. J Arthroplasty. 2012;27(7):1376-1381.
15. Rosenberg AG. Reducing blood loss in total joint surgery with a saline-coupled bipolar sealing technology. J Arthroplasty. 2007;22(4 suppl 1):82-85.
16. Pfeiffer M, Bräutigam H, Draws D, Sigg A. A new bipolar blood sealing system embedded in perioperative strategies vs. a conventional regimen for total knee arthroplasty: results of a matched-pair study. Ger Med Sci. 2005;3:Doc10.
17. Morris MJ, Barrett M, Lombardi AV Jr, Tucker TL, Berend KR. Randomized blinded study comparing a bipolar sealer and standard electrocautery in reducing transfusion requirements in anterior supine intermuscular total hip arthroplasty. J Arthroplasty. 2013;28(9):1614-1617.
18. Barsoum WK, Klika AK, Murray TG, Higuera C, Lee HH, Krebs VE. Prospective randomized evaluation of the need for blood transfusion during primary total hip arthroplasty with use of a bipolar sealer. J Bone Joint Surg Am. 2011;93(6):513-518.
19. Plymale MF, Capogna BM, Lovy AJ, Adler ML, Hirsh DM, Kim SJ. Unipolar vs bipolar hemostasis in total knee arthroplasty: a prospective randomized trial. J Arthroplasty. 2012;27(6):1133-1137.e1.
20. Zeh A, Messer J, Davis J, Vasarhelyi A, Wohlrab D. The Aquamantys system—an alternative to reduce blood loss in primary total hip arthroplasty? J Arthroplasty. 2010;25(7):1072-1077.
21. Cummings JE, Smith RA, Heck RK Jr. Argon beam coagulation as adjuvant treatment after curettage of aneurysmal bone cysts: a preliminary study. Clin Orthop Relat Res. 2010;468(1):231-237.
22. Adams ML, Steinberg JS. Argon beam coagulation in foot and ankle surgery. J Foot Ankle Surg. 2011;50(6):780-782.
23. Neumayer L, Vargo D. Principles of preoperative and operative surgery. In: Townsend CM Jr, Beauchamp RD, Evers BM, Mattox KL, eds. Sabiston Textbook of Surgery. 19th ed. Philadelphia, PA: Elsevier Saunders; 2012:211-239.
24. Walker E, Nowacki AS. Understanding equivalence and noninferiority testing. J Gen Intern Med. 2011;26(2):192-196.
25. Hrnack SA, Skeen N, Xu T, Rosenstein AD. Correlation of body mass index and blood loss during total knee and total hip arthroplasty. Am J Orthop. 2012;41(10):467-471.
Obesity Has Minimal Impact on Short-Term Functional Scores After Reverse Shoulder Arthroplasty for Rotator Cuff Tear Arthropathy
Body mass index (BMI) is thought to be a predictor of body composition, with higher values indicating more adipose tissue. BMI is a measure of mass with respect to height. The World Health Organization1 has established health categories based on BMI measurements. Values from 18.5 to 24.9 kg/m2 are deemed to represent normal weight; those from 25 to 30 kg/m2, overweight; and those higher than 30 kg/m2, obesity. BMI is not a perfect tool, but it is the most widely used tool in clinical and research practice because of its relative reliability and ease of use.2 Being overweight or obese (according to BMI) is increasingly common among adults worldwide, and particularly in the United States. An estimated 39% of adults worldwide are overweight, and 13% are obese.1 An estimated 69% of US adults are overweight, including 35.1% who are obese.2
Various pathologies have been treated with reverse shoulder arthroplasty (RSA), and results have been promising,3-9 but little is known about patient demographic and clinical factors that may adversely affect outcomes. Recent work suggests younger age7 and failed prior arthroplasty may adversely affect RSA outcomes.10 Higher BMI has also been implicated as a cause of increased perioperative and immediate postoperative complications of RSA with minimum 90-day follow-up, but no one has examined shoulder function scores at minimum 2-year follow-up.11,12
We conducted a study to examine shoulder function scores, mobility, patient satisfaction, and complications at minimum 2-year follow-up in normal-weight, overweight, and obese patients who underwent RSA. We hypothesized that, compared with normal-weight patients, obese patients would have worse shoulder function scores, worse mobility, and more complications.
Materials and Methods
Inclusion Criteria and Demographics
After obtaining Institutional Review Board approval for this study, we used a prospective shoulder arthroplasty registry to identify patients (N = 77) who had rotator cuff tear arthropathy (RCTA) treated with primary RSA and then had minimum 2-year follow-up. The study period was 2004-2011. All patients had RCTA diagnosed with physical examination findings and anteroposterior, scapular Y, and axillary radiographs. RCTA was graded 1 to 5 using the classification system of Hamada and colleagues.13 Rotator cuff status was determined with preoperative computed tomography arthrogram (CTA) or magnetic resonance imaging (MRI) and confirmed at time of surgery. BMI calculations were based on height and weight measured at initial office visit. Thirty-four patients had normal weight (BMI <25 kg/m2), 21 were overweight (BMI 25-30 kg/m2), and 22 were obese (BMI >30 kg/m2). Patient demographic and clinical characteristics reviewed also included age, sex, follow-up duration, arm dominance, complications, prevalence of depression, and prevalence of diabetes. All RSAs were performed by the same surgeon (Dr. Edwards) at a single high-volume shoulder arthroplasty center.
Shoulder function scores evaluated before surgery and at final follow-up included Constant score,14 American Shoulder and Elbow Surgeons (ASES) score,15 Western Ontario Osteoarthritis Shoulder (WOOS) index,16 Single Assessment Numeric Evaluation (SANE),17 and mobility. Satisfaction was assessed by having patients describe themselves as very dissatisfied, dissatisfied, satisfied, or very satisfied. All intraoperative and postoperative complications were recorded.
Surgical Technique and Postoperative Rehabilitation
The Aequalis RSA system (Tornier) was used for all patients during the study period. The RSA technique used has been well described.18,19 A standard postoperative rehabilitation protocol was followed.19,20
Clinical and Radiographic Assessment
Patients were prospectively enrolled in a shoulder arthroplasty outcomes registry and followed clinically. Mean follow-up was 3.16 years (range, 2-8 years). Before surgery, patients were examined by the surgeon. Examinations were repeated 1 week, 6 weeks, 3 months, 6 months, and 12 months after surgery and annually thereafter. Mobility (active range of motion) was determined with a handheld goniometer. Strength of abduction was measured with a handheld digital dynamometer (Chatillon digital force gauge, 200 lbf; Ametek). Anteroposterior in plane of scapula, scapular Y, and axillary radiographs were obtained at each clinic appointment.
Before surgery, the surgeon reviewed all radiographs. Each RCTA was given a Hamada grade (1-5).13 Glenoid erosion in the coronal plane was classified (E0, E1, E2, E3) according to Sirveaux and colleagues.21 Hamada grades and glenoid erosion types are listed in Table 1. The overall trend in classification by BMI group was statistically significant for Hamada grade (P = .004) but not glenoid erosion type (P = .153).
Before surgery, the surgeon also evaluated rotator cuff status using CTA or MRI. All patients had full-thickness tears of the supraspinatus and infraspinatus. The subscapularis was variably present, and subscapularis repair was performed when the subscapularis was intact. Rotator cuff status is listed in Table 2. There were no significant differences in the distribution of intact subscapularis (P = .402) or teres minor (P = .188) among the normal-weight, overweight, and obese groups. No patient had a latissimus dorsi transfer at time of RSA.
Statistical Analysis
Independent-samples t tests assuming unequal variances were used to compare the 3 BMI groups on age, follow-up duration, preoperative shoulder function scores, and mobility. Chi-square tests were used to identify any significant group differences in comorbidities (eg, complications, arm dominance, prevalence of depression, prevalence of diabetes) and patient satisfaction. Repeated-measures analysis of variance was used to evaluate main effects, changes from before surgery to final follow-up, and BMI group differences, as well as differences in changes from before surgery to final follow-up among the 3 BMI groups.
Results
Among BMI groups (<25 kg/m2, 25-30 kg/m2, >30 kg/m2), there were no statistically significant preoperative differences in age, sex, follow-up duration, complications, arm dominance, prevalence of depression, or prevalence of diabetes (P >. 05) (Table 3). Table 4 lists the groups’ preoperative and final follow-up data (Constant score, ASES score, WOOS index, SANE, mobility). There were no statistically significant preoperative group differences in Constant score, ASES score, WOOS index, SANE, mobility, or patient satisfaction (P > .05) (Tables 5, 6).
All groups’ shoulder function scores and mobility improved significantly from before surgery to final follow-up (P < .001) (Table 5). The groups’ magnitudes of change (improvement) from before surgery to final follow-up were almost identical, with no significant differences in shoulder function scores or mobility (Table 5). The only significant difference was in Constant–Strength, which was higher in the obese group (P = .017) (Table 5). Patient satisfaction ratings improved after surgery, with 79% of the normal-weight group reporting being satisfied or very satisfied with their shoulders (Table 6). The overweight and obese groups gave similar satisfied (81%) and very satisfied (82%) ratings. The small differences between group satisfaction scores were nonsignificant (P = .967).
Complications
The normal-weight group had 4 complications: periprosthetic infection (2 cases), intraoperative humeral fracture (1), and scapular spine stress fracture (1). The overweight group had 1 complication, an acromial stress fracture that was successfully treated with conservative measures. The obese group had 1 patient with 2 postoperative dislocations. The first dislocation was treated with closed reduction and bracing, and the second required revision surgery. There was no statistical difference in complications among the groups (P = .680).
Discussion
To our knowledge, this is the first study of the effects of varying BMI on functional outcomes of RSA with minimum 2-year follow-up. There appears to be minimal impact on shoulder function scores, complications, and patient satisfaction among normal-weight, overweight, and obese patients with RCTA treated by the same surgeon using similar techniques.
The relationship between obesity and increased perioperative risks or poorer surgical outcomes has been well established in orthopedic surgery. In a systematic review, Falagas and Kompoti22 found increased risk for perioperative and nosocomial infections in obese patients. Schoenfeld and colleagues23 and Jiang and colleagues24 reported increased risk for complications in spinal surgery. The total joint arthroplasty literature is rife with evidence suggesting higher BMI leads to increased risk for complications, including infection and deep venous thrombosis, as well as decreased functional outcome scores.25-29 Recent studies on shoulder surgery have shown worse outcomes in rotator cuff repair30 and a higher revision rate in hemiarthroplasy.31
Other RSA studies have examined short-term complications or perioperative risk factors associated with BMI. In a study using slightly different BMI groupings, Gupta and colleagues12 reported significantly higher complication rates for RSA patients with BMI higher than 35 kg/m2 compared to patients with BMI of 25 to 35 kg/m2 and compared to patients with BMI lower than 25 kg/m2. Their study highlighted medical and surgical complications and used a short follow-up period (minimum, 90 days). It did not assess shoulder function scores, and included multiple indications for RSA (eg, RCTA, proximal humerus fracture, inflammatory arthropathy). In another study, higher BMI was reported as a risk factor for early dislocation after RSA, but only 11 patients with a history of dislocation were assessed, and minimum follow-up was 6 months.32 We know of only one study that addressed RSA outcomes in obese patients and used minimum 2-year follow-up, but its primary endpoint was rate of complications, and it did not report shoulder function scores.11 Li and colleagues33 conducted a study similar to ours, but with primary total shoulder arthroplasty (TSA) patients, and reported similar results. Relative to normal BMI, higher BMI did not have a detrimental effect on short-term improvement in shoulder function after TSA.
Given the US obesity epidemic, our study results are encouraging. Depending on many factors, obesity remains a risk factor for poor outcomes in patients who undergo orthopedic surgery. As our results show, however, patients with higher BMI can obtain functional outcomes similar to those experienced by patients with normal-weight BMI after RSA for RCTA.
The primary limitation of this study was its retrospective design. Strengths of the study included its having a single surgeon and a single diagnosis: RCTA. In addition, each group was robust in size, a standard operative/postoperative protocol was used, and clinical results were measured with multiple validated shoulder function scores.
Conclusion
Improved shoulder function scores, mobility, and patient satisfaction can be expected after RSA for RCTA in patients with BMI higher than 30 kg/m2. These patients did not exhibit an increase in complications at short-term follow-up.
1. World Health Organization. Obesity and overweight [factsheet 311]. Updated January 2015. http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed March 27, 2016.
2. National Center for Health Statistics, Centers for Disease Control and Prevention. Obesity and overweight. Updated February 25, 2016. http://www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed March 27, 2016.
3. Boileau P, Gonzalez JF, Chuinard C, Bicknell R, Walch G. Reverse total shoulder arthroplasty after failed rotator cuff surgery. J Shoulder Elbow Surg. 2009;18(4):600-606.
4. Drake GN, O’Connor DP, Edwards TB. Indications for reverse total shoulder arthroplasty in rotator cuff disease. Clin Orthop Relat Res. 2010;468(6):1526-1533.
5. Gerber C, Pennington SD, Nyffeler RW. Reverse total shoulder arthroplasty. J Am Acad Orthop Surg. 2009;17(5):284-289.
6. Lenarz C, Shishani Y, McCrum C, Nowinski RJ, Edwards TB, Gobezie R. Is reverse shoulder arthroplasty appropriate for the treatment of fractures in the older patient? Early observations. Clin Orthop Relat Res. 2011;469(12):3324-3331.
7. Muh SJ, Streit JJ, Wanner JP, et al. Early follow-up of reverse total shoulder arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2013;95(20):1877-1883.
8. Wall B, Nové-Josserand L, O’Connor DP, Edwards TB, Walch G. Reverse total shoulder arthroplasty: a review of results according to etiology. J Bone Joint Surg Am. 2007;89(7):1476-1485.
9. Young AA, Smith MM, Bacle G, Moraga C, Walch G. Early results of reverse shoulder arthroplasty in patients with rheumatoid arthritis. J Bone Joint Surg Am. 2011;93(20):1915-1923.
10. Boileau P, Melis B, Duperron D, Moineau G, Rumian AP, Han Y. Revision surgery of reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2013;22(10):1359-1370.
11. Beck JD, Irgit KS, Andreychik CM, Maloney PJ, Tang X, Harter GD. Reverse total shoulder arthroplasty in obese patients. J Hand Surg Am. 2013;38(5):965-970.
12. Gupta AK, Chalmers PN, Rahman Z, et al. Reverse total shoulder arthroplasty in patients of varying body mass index. J Shoulder Elbow Surg. 2014;23(1):35-42.
13. Hamada K, Fukuda H, Mikasa M, Kobayashi Y. Roentgenographic findings in massive rotator cuff tears. A long-term observation. Clin Orthop Relat Res. 1990;(254):92-96.
14. Constant CR, Murley AH. A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res. 1987;(214):160-164.
15. Michener LA, McClure PW, Sennett BJ. American Shoulder and Elbow Surgeons standardized shoulder assessment form, patient self-report section: reliability, validity, and responsiveness. J Shoulder Elbow Surg. 2002;11(6):587-594.
16. Lo IK, Griffin S, Kirkley A. The development of a disease-specific quality of life measurement tool for osteoarthritis of the shoulder: the Western Ontario Osteoarthritis of the Shoulder (WOOS) index. Osteoarthritis Cartilage. 2001;9(8):771-778.
17. Williams GN, Gangel TJ, Arciero RA, Uhorchak JM, Taylor DC. Comparison of the Single Assessment Numeric Evaluation method and two shoulder rating scales. Outcomes measures after shoulder surgery. Am J Sports Med. 1999;27(2):214-221.
18. Gartsman GM, Edwards TB. Shoulder Arthroplasty. Philadelpia, PA: Saunders Elsevier; 2008.
19. Liotard JP, Edwards TB, Padey A, Walch G, Boulahia A. Hydrotherapy rehabilitation after shoulder surgery. Tech Shoulder Elbow Surg. 2003;4:44-49.
20. Trappey GJ 4th, O’Connor DP, Edwards TB. What are the instability and infection rates after reverse shoulder arthroplasty? Clin Orthop Relat Res. 2011;469(9):2505-2511.
21. Sirveaux F, Favard L, Oudet D, Huquet D, Walch G, Molé D. Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Joint Surg Br. 2004;86(3):388-395.
22. Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6(7):438-446.
23. Schoenfeld AJ, Carey PA, Cleveland AW 3rd, Bader JO, Bono CM. Patient factors, comorbidities, and surgical characteristics that increase mortality and complication risk after spinal arthrodesis: a prognostic study based on 5,887 patients. Spine J. 2013;13(10):1171-1179.
24. Jiang J, Teng Y, Fan Z, Khan S, Xia Y. Does obesity affect the surgical outcome and complication rates of spinal surgery? A meta-analysis. Clin Orthop Relat Res. 2014;472(3):968-975.
25. Bozic KJ, Lau E, Kurtz S, et al. Patient-related risk factors for periprosthetic joint infection and postoperative mortality following total hip arthroplasty in Medicare patients. J Bone Joint Surg Am. 2012;94(9):794-800.
26. Franklin PD, Li W, Ayers DC. The Chitranjan Ranawat Award: functional outcome after total knee replacement varies with patient attributes. Clin Orthop Relat Res. 2008;466(11):2597-2604.
27. Huddleston JI, Wang Y, Uquillas C, Herndon JH, Maloney WJ. Age and obesity are risk factors for adverse events after total hip arthroplasty. Clin Orthop Relat Res. 2012;470(2):490-496.
28. Jämsen E, Nevalainen P, Eskelinen A, Huotari K, Kalliovalkama J, Moilanen T. Obesity, diabetes, and preoperative hyperglycemia as predictors of periprosthetic joint infection: a single-center analysis of 7181 primary hip and knee replacements for osteoarthritis. J Bone Joint Surg Am. 2012;94(14):e101.
29. Naziri Q, Issa K, Malkani AL, Bonutti PM, Harwin SF, Mont MA. Bariatric orthopaedics: total knee arthroplasty in super-obese patients (BMI > 50 kg/m2). Survivorship and complications. Clin Orthop Relat Res. 2013;471(11):3523-3530.
30. Warrender WJ, Brown OL, Abboud JA. Outcomes of arthroscopic rotator cuff repairs in obese patients. J Shoulder Elbow Surg. 2011;20(6):961-967.
31. Singh JA, Sperling JW, Cofield RH. Risk factors for revision surgery after humeral head replacement: 1,431 shoulders over 3 decades. J Shoulder Elbow Surg. 2012;21(8):1039-1044.
32. Chalmers PN, Rahman Z, Romeo AA, Nicholson GP. Early dislocation after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(5):737-744.
33. Li X, Williams PN, Nguyen JT, Craig EV, Warren RF, Gulotta LV. Functional outcomes after total shoulder arthroplasty in obese patients. J Bone Joint Surg Am. 2013;95(21):e160.
Body mass index (BMI) is thought to be a predictor of body composition, with higher values indicating more adipose tissue. BMI is a measure of mass with respect to height. The World Health Organization1 has established health categories based on BMI measurements. Values from 18.5 to 24.9 kg/m2 are deemed to represent normal weight; those from 25 to 30 kg/m2, overweight; and those higher than 30 kg/m2, obesity. BMI is not a perfect tool, but it is the most widely used tool in clinical and research practice because of its relative reliability and ease of use.2 Being overweight or obese (according to BMI) is increasingly common among adults worldwide, and particularly in the United States. An estimated 39% of adults worldwide are overweight, and 13% are obese.1 An estimated 69% of US adults are overweight, including 35.1% who are obese.2
Various pathologies have been treated with reverse shoulder arthroplasty (RSA), and results have been promising,3-9 but little is known about patient demographic and clinical factors that may adversely affect outcomes. Recent work suggests younger age7 and failed prior arthroplasty may adversely affect RSA outcomes.10 Higher BMI has also been implicated as a cause of increased perioperative and immediate postoperative complications of RSA with minimum 90-day follow-up, but no one has examined shoulder function scores at minimum 2-year follow-up.11,12
We conducted a study to examine shoulder function scores, mobility, patient satisfaction, and complications at minimum 2-year follow-up in normal-weight, overweight, and obese patients who underwent RSA. We hypothesized that, compared with normal-weight patients, obese patients would have worse shoulder function scores, worse mobility, and more complications.
Materials and Methods
Inclusion Criteria and Demographics
After obtaining Institutional Review Board approval for this study, we used a prospective shoulder arthroplasty registry to identify patients (N = 77) who had rotator cuff tear arthropathy (RCTA) treated with primary RSA and then had minimum 2-year follow-up. The study period was 2004-2011. All patients had RCTA diagnosed with physical examination findings and anteroposterior, scapular Y, and axillary radiographs. RCTA was graded 1 to 5 using the classification system of Hamada and colleagues.13 Rotator cuff status was determined with preoperative computed tomography arthrogram (CTA) or magnetic resonance imaging (MRI) and confirmed at time of surgery. BMI calculations were based on height and weight measured at initial office visit. Thirty-four patients had normal weight (BMI <25 kg/m2), 21 were overweight (BMI 25-30 kg/m2), and 22 were obese (BMI >30 kg/m2). Patient demographic and clinical characteristics reviewed also included age, sex, follow-up duration, arm dominance, complications, prevalence of depression, and prevalence of diabetes. All RSAs were performed by the same surgeon (Dr. Edwards) at a single high-volume shoulder arthroplasty center.
Shoulder function scores evaluated before surgery and at final follow-up included Constant score,14 American Shoulder and Elbow Surgeons (ASES) score,15 Western Ontario Osteoarthritis Shoulder (WOOS) index,16 Single Assessment Numeric Evaluation (SANE),17 and mobility. Satisfaction was assessed by having patients describe themselves as very dissatisfied, dissatisfied, satisfied, or very satisfied. All intraoperative and postoperative complications were recorded.
Surgical Technique and Postoperative Rehabilitation
The Aequalis RSA system (Tornier) was used for all patients during the study period. The RSA technique used has been well described.18,19 A standard postoperative rehabilitation protocol was followed.19,20
Clinical and Radiographic Assessment
Patients were prospectively enrolled in a shoulder arthroplasty outcomes registry and followed clinically. Mean follow-up was 3.16 years (range, 2-8 years). Before surgery, patients were examined by the surgeon. Examinations were repeated 1 week, 6 weeks, 3 months, 6 months, and 12 months after surgery and annually thereafter. Mobility (active range of motion) was determined with a handheld goniometer. Strength of abduction was measured with a handheld digital dynamometer (Chatillon digital force gauge, 200 lbf; Ametek). Anteroposterior in plane of scapula, scapular Y, and axillary radiographs were obtained at each clinic appointment.
Before surgery, the surgeon reviewed all radiographs. Each RCTA was given a Hamada grade (1-5).13 Glenoid erosion in the coronal plane was classified (E0, E1, E2, E3) according to Sirveaux and colleagues.21 Hamada grades and glenoid erosion types are listed in Table 1. The overall trend in classification by BMI group was statistically significant for Hamada grade (P = .004) but not glenoid erosion type (P = .153).
Before surgery, the surgeon also evaluated rotator cuff status using CTA or MRI. All patients had full-thickness tears of the supraspinatus and infraspinatus. The subscapularis was variably present, and subscapularis repair was performed when the subscapularis was intact. Rotator cuff status is listed in Table 2. There were no significant differences in the distribution of intact subscapularis (P = .402) or teres minor (P = .188) among the normal-weight, overweight, and obese groups. No patient had a latissimus dorsi transfer at time of RSA.
Statistical Analysis
Independent-samples t tests assuming unequal variances were used to compare the 3 BMI groups on age, follow-up duration, preoperative shoulder function scores, and mobility. Chi-square tests were used to identify any significant group differences in comorbidities (eg, complications, arm dominance, prevalence of depression, prevalence of diabetes) and patient satisfaction. Repeated-measures analysis of variance was used to evaluate main effects, changes from before surgery to final follow-up, and BMI group differences, as well as differences in changes from before surgery to final follow-up among the 3 BMI groups.
Results
Among BMI groups (<25 kg/m2, 25-30 kg/m2, >30 kg/m2), there were no statistically significant preoperative differences in age, sex, follow-up duration, complications, arm dominance, prevalence of depression, or prevalence of diabetes (P >. 05) (Table 3). Table 4 lists the groups’ preoperative and final follow-up data (Constant score, ASES score, WOOS index, SANE, mobility). There were no statistically significant preoperative group differences in Constant score, ASES score, WOOS index, SANE, mobility, or patient satisfaction (P > .05) (Tables 5, 6).
All groups’ shoulder function scores and mobility improved significantly from before surgery to final follow-up (P < .001) (Table 5). The groups’ magnitudes of change (improvement) from before surgery to final follow-up were almost identical, with no significant differences in shoulder function scores or mobility (Table 5). The only significant difference was in Constant–Strength, which was higher in the obese group (P = .017) (Table 5). Patient satisfaction ratings improved after surgery, with 79% of the normal-weight group reporting being satisfied or very satisfied with their shoulders (Table 6). The overweight and obese groups gave similar satisfied (81%) and very satisfied (82%) ratings. The small differences between group satisfaction scores were nonsignificant (P = .967).
Complications
The normal-weight group had 4 complications: periprosthetic infection (2 cases), intraoperative humeral fracture (1), and scapular spine stress fracture (1). The overweight group had 1 complication, an acromial stress fracture that was successfully treated with conservative measures. The obese group had 1 patient with 2 postoperative dislocations. The first dislocation was treated with closed reduction and bracing, and the second required revision surgery. There was no statistical difference in complications among the groups (P = .680).
Discussion
To our knowledge, this is the first study of the effects of varying BMI on functional outcomes of RSA with minimum 2-year follow-up. There appears to be minimal impact on shoulder function scores, complications, and patient satisfaction among normal-weight, overweight, and obese patients with RCTA treated by the same surgeon using similar techniques.
The relationship between obesity and increased perioperative risks or poorer surgical outcomes has been well established in orthopedic surgery. In a systematic review, Falagas and Kompoti22 found increased risk for perioperative and nosocomial infections in obese patients. Schoenfeld and colleagues23 and Jiang and colleagues24 reported increased risk for complications in spinal surgery. The total joint arthroplasty literature is rife with evidence suggesting higher BMI leads to increased risk for complications, including infection and deep venous thrombosis, as well as decreased functional outcome scores.25-29 Recent studies on shoulder surgery have shown worse outcomes in rotator cuff repair30 and a higher revision rate in hemiarthroplasy.31
Other RSA studies have examined short-term complications or perioperative risk factors associated with BMI. In a study using slightly different BMI groupings, Gupta and colleagues12 reported significantly higher complication rates for RSA patients with BMI higher than 35 kg/m2 compared to patients with BMI of 25 to 35 kg/m2 and compared to patients with BMI lower than 25 kg/m2. Their study highlighted medical and surgical complications and used a short follow-up period (minimum, 90 days). It did not assess shoulder function scores, and included multiple indications for RSA (eg, RCTA, proximal humerus fracture, inflammatory arthropathy). In another study, higher BMI was reported as a risk factor for early dislocation after RSA, but only 11 patients with a history of dislocation were assessed, and minimum follow-up was 6 months.32 We know of only one study that addressed RSA outcomes in obese patients and used minimum 2-year follow-up, but its primary endpoint was rate of complications, and it did not report shoulder function scores.11 Li and colleagues33 conducted a study similar to ours, but with primary total shoulder arthroplasty (TSA) patients, and reported similar results. Relative to normal BMI, higher BMI did not have a detrimental effect on short-term improvement in shoulder function after TSA.
Given the US obesity epidemic, our study results are encouraging. Depending on many factors, obesity remains a risk factor for poor outcomes in patients who undergo orthopedic surgery. As our results show, however, patients with higher BMI can obtain functional outcomes similar to those experienced by patients with normal-weight BMI after RSA for RCTA.
The primary limitation of this study was its retrospective design. Strengths of the study included its having a single surgeon and a single diagnosis: RCTA. In addition, each group was robust in size, a standard operative/postoperative protocol was used, and clinical results were measured with multiple validated shoulder function scores.
Conclusion
Improved shoulder function scores, mobility, and patient satisfaction can be expected after RSA for RCTA in patients with BMI higher than 30 kg/m2. These patients did not exhibit an increase in complications at short-term follow-up.
Body mass index (BMI) is thought to be a predictor of body composition, with higher values indicating more adipose tissue. BMI is a measure of mass with respect to height. The World Health Organization1 has established health categories based on BMI measurements. Values from 18.5 to 24.9 kg/m2 are deemed to represent normal weight; those from 25 to 30 kg/m2, overweight; and those higher than 30 kg/m2, obesity. BMI is not a perfect tool, but it is the most widely used tool in clinical and research practice because of its relative reliability and ease of use.2 Being overweight or obese (according to BMI) is increasingly common among adults worldwide, and particularly in the United States. An estimated 39% of adults worldwide are overweight, and 13% are obese.1 An estimated 69% of US adults are overweight, including 35.1% who are obese.2
Various pathologies have been treated with reverse shoulder arthroplasty (RSA), and results have been promising,3-9 but little is known about patient demographic and clinical factors that may adversely affect outcomes. Recent work suggests younger age7 and failed prior arthroplasty may adversely affect RSA outcomes.10 Higher BMI has also been implicated as a cause of increased perioperative and immediate postoperative complications of RSA with minimum 90-day follow-up, but no one has examined shoulder function scores at minimum 2-year follow-up.11,12
We conducted a study to examine shoulder function scores, mobility, patient satisfaction, and complications at minimum 2-year follow-up in normal-weight, overweight, and obese patients who underwent RSA. We hypothesized that, compared with normal-weight patients, obese patients would have worse shoulder function scores, worse mobility, and more complications.
Materials and Methods
Inclusion Criteria and Demographics
After obtaining Institutional Review Board approval for this study, we used a prospective shoulder arthroplasty registry to identify patients (N = 77) who had rotator cuff tear arthropathy (RCTA) treated with primary RSA and then had minimum 2-year follow-up. The study period was 2004-2011. All patients had RCTA diagnosed with physical examination findings and anteroposterior, scapular Y, and axillary radiographs. RCTA was graded 1 to 5 using the classification system of Hamada and colleagues.13 Rotator cuff status was determined with preoperative computed tomography arthrogram (CTA) or magnetic resonance imaging (MRI) and confirmed at time of surgery. BMI calculations were based on height and weight measured at initial office visit. Thirty-four patients had normal weight (BMI <25 kg/m2), 21 were overweight (BMI 25-30 kg/m2), and 22 were obese (BMI >30 kg/m2). Patient demographic and clinical characteristics reviewed also included age, sex, follow-up duration, arm dominance, complications, prevalence of depression, and prevalence of diabetes. All RSAs were performed by the same surgeon (Dr. Edwards) at a single high-volume shoulder arthroplasty center.
Shoulder function scores evaluated before surgery and at final follow-up included Constant score,14 American Shoulder and Elbow Surgeons (ASES) score,15 Western Ontario Osteoarthritis Shoulder (WOOS) index,16 Single Assessment Numeric Evaluation (SANE),17 and mobility. Satisfaction was assessed by having patients describe themselves as very dissatisfied, dissatisfied, satisfied, or very satisfied. All intraoperative and postoperative complications were recorded.
Surgical Technique and Postoperative Rehabilitation
The Aequalis RSA system (Tornier) was used for all patients during the study period. The RSA technique used has been well described.18,19 A standard postoperative rehabilitation protocol was followed.19,20
Clinical and Radiographic Assessment
Patients were prospectively enrolled in a shoulder arthroplasty outcomes registry and followed clinically. Mean follow-up was 3.16 years (range, 2-8 years). Before surgery, patients were examined by the surgeon. Examinations were repeated 1 week, 6 weeks, 3 months, 6 months, and 12 months after surgery and annually thereafter. Mobility (active range of motion) was determined with a handheld goniometer. Strength of abduction was measured with a handheld digital dynamometer (Chatillon digital force gauge, 200 lbf; Ametek). Anteroposterior in plane of scapula, scapular Y, and axillary radiographs were obtained at each clinic appointment.
Before surgery, the surgeon reviewed all radiographs. Each RCTA was given a Hamada grade (1-5).13 Glenoid erosion in the coronal plane was classified (E0, E1, E2, E3) according to Sirveaux and colleagues.21 Hamada grades and glenoid erosion types are listed in Table 1. The overall trend in classification by BMI group was statistically significant for Hamada grade (P = .004) but not glenoid erosion type (P = .153).
Before surgery, the surgeon also evaluated rotator cuff status using CTA or MRI. All patients had full-thickness tears of the supraspinatus and infraspinatus. The subscapularis was variably present, and subscapularis repair was performed when the subscapularis was intact. Rotator cuff status is listed in Table 2. There were no significant differences in the distribution of intact subscapularis (P = .402) or teres minor (P = .188) among the normal-weight, overweight, and obese groups. No patient had a latissimus dorsi transfer at time of RSA.
Statistical Analysis
Independent-samples t tests assuming unequal variances were used to compare the 3 BMI groups on age, follow-up duration, preoperative shoulder function scores, and mobility. Chi-square tests were used to identify any significant group differences in comorbidities (eg, complications, arm dominance, prevalence of depression, prevalence of diabetes) and patient satisfaction. Repeated-measures analysis of variance was used to evaluate main effects, changes from before surgery to final follow-up, and BMI group differences, as well as differences in changes from before surgery to final follow-up among the 3 BMI groups.
Results
Among BMI groups (<25 kg/m2, 25-30 kg/m2, >30 kg/m2), there were no statistically significant preoperative differences in age, sex, follow-up duration, complications, arm dominance, prevalence of depression, or prevalence of diabetes (P >. 05) (Table 3). Table 4 lists the groups’ preoperative and final follow-up data (Constant score, ASES score, WOOS index, SANE, mobility). There were no statistically significant preoperative group differences in Constant score, ASES score, WOOS index, SANE, mobility, or patient satisfaction (P > .05) (Tables 5, 6).
All groups’ shoulder function scores and mobility improved significantly from before surgery to final follow-up (P < .001) (Table 5). The groups’ magnitudes of change (improvement) from before surgery to final follow-up were almost identical, with no significant differences in shoulder function scores or mobility (Table 5). The only significant difference was in Constant–Strength, which was higher in the obese group (P = .017) (Table 5). Patient satisfaction ratings improved after surgery, with 79% of the normal-weight group reporting being satisfied or very satisfied with their shoulders (Table 6). The overweight and obese groups gave similar satisfied (81%) and very satisfied (82%) ratings. The small differences between group satisfaction scores were nonsignificant (P = .967).
Complications
The normal-weight group had 4 complications: periprosthetic infection (2 cases), intraoperative humeral fracture (1), and scapular spine stress fracture (1). The overweight group had 1 complication, an acromial stress fracture that was successfully treated with conservative measures. The obese group had 1 patient with 2 postoperative dislocations. The first dislocation was treated with closed reduction and bracing, and the second required revision surgery. There was no statistical difference in complications among the groups (P = .680).
Discussion
To our knowledge, this is the first study of the effects of varying BMI on functional outcomes of RSA with minimum 2-year follow-up. There appears to be minimal impact on shoulder function scores, complications, and patient satisfaction among normal-weight, overweight, and obese patients with RCTA treated by the same surgeon using similar techniques.
The relationship between obesity and increased perioperative risks or poorer surgical outcomes has been well established in orthopedic surgery. In a systematic review, Falagas and Kompoti22 found increased risk for perioperative and nosocomial infections in obese patients. Schoenfeld and colleagues23 and Jiang and colleagues24 reported increased risk for complications in spinal surgery. The total joint arthroplasty literature is rife with evidence suggesting higher BMI leads to increased risk for complications, including infection and deep venous thrombosis, as well as decreased functional outcome scores.25-29 Recent studies on shoulder surgery have shown worse outcomes in rotator cuff repair30 and a higher revision rate in hemiarthroplasy.31
Other RSA studies have examined short-term complications or perioperative risk factors associated with BMI. In a study using slightly different BMI groupings, Gupta and colleagues12 reported significantly higher complication rates for RSA patients with BMI higher than 35 kg/m2 compared to patients with BMI of 25 to 35 kg/m2 and compared to patients with BMI lower than 25 kg/m2. Their study highlighted medical and surgical complications and used a short follow-up period (minimum, 90 days). It did not assess shoulder function scores, and included multiple indications for RSA (eg, RCTA, proximal humerus fracture, inflammatory arthropathy). In another study, higher BMI was reported as a risk factor for early dislocation after RSA, but only 11 patients with a history of dislocation were assessed, and minimum follow-up was 6 months.32 We know of only one study that addressed RSA outcomes in obese patients and used minimum 2-year follow-up, but its primary endpoint was rate of complications, and it did not report shoulder function scores.11 Li and colleagues33 conducted a study similar to ours, but with primary total shoulder arthroplasty (TSA) patients, and reported similar results. Relative to normal BMI, higher BMI did not have a detrimental effect on short-term improvement in shoulder function after TSA.
Given the US obesity epidemic, our study results are encouraging. Depending on many factors, obesity remains a risk factor for poor outcomes in patients who undergo orthopedic surgery. As our results show, however, patients with higher BMI can obtain functional outcomes similar to those experienced by patients with normal-weight BMI after RSA for RCTA.
The primary limitation of this study was its retrospective design. Strengths of the study included its having a single surgeon and a single diagnosis: RCTA. In addition, each group was robust in size, a standard operative/postoperative protocol was used, and clinical results were measured with multiple validated shoulder function scores.
Conclusion
Improved shoulder function scores, mobility, and patient satisfaction can be expected after RSA for RCTA in patients with BMI higher than 30 kg/m2. These patients did not exhibit an increase in complications at short-term follow-up.
1. World Health Organization. Obesity and overweight [factsheet 311]. Updated January 2015. http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed March 27, 2016.
2. National Center for Health Statistics, Centers for Disease Control and Prevention. Obesity and overweight. Updated February 25, 2016. http://www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed March 27, 2016.
3. Boileau P, Gonzalez JF, Chuinard C, Bicknell R, Walch G. Reverse total shoulder arthroplasty after failed rotator cuff surgery. J Shoulder Elbow Surg. 2009;18(4):600-606.
4. Drake GN, O’Connor DP, Edwards TB. Indications for reverse total shoulder arthroplasty in rotator cuff disease. Clin Orthop Relat Res. 2010;468(6):1526-1533.
5. Gerber C, Pennington SD, Nyffeler RW. Reverse total shoulder arthroplasty. J Am Acad Orthop Surg. 2009;17(5):284-289.
6. Lenarz C, Shishani Y, McCrum C, Nowinski RJ, Edwards TB, Gobezie R. Is reverse shoulder arthroplasty appropriate for the treatment of fractures in the older patient? Early observations. Clin Orthop Relat Res. 2011;469(12):3324-3331.
7. Muh SJ, Streit JJ, Wanner JP, et al. Early follow-up of reverse total shoulder arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2013;95(20):1877-1883.
8. Wall B, Nové-Josserand L, O’Connor DP, Edwards TB, Walch G. Reverse total shoulder arthroplasty: a review of results according to etiology. J Bone Joint Surg Am. 2007;89(7):1476-1485.
9. Young AA, Smith MM, Bacle G, Moraga C, Walch G. Early results of reverse shoulder arthroplasty in patients with rheumatoid arthritis. J Bone Joint Surg Am. 2011;93(20):1915-1923.
10. Boileau P, Melis B, Duperron D, Moineau G, Rumian AP, Han Y. Revision surgery of reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2013;22(10):1359-1370.
11. Beck JD, Irgit KS, Andreychik CM, Maloney PJ, Tang X, Harter GD. Reverse total shoulder arthroplasty in obese patients. J Hand Surg Am. 2013;38(5):965-970.
12. Gupta AK, Chalmers PN, Rahman Z, et al. Reverse total shoulder arthroplasty in patients of varying body mass index. J Shoulder Elbow Surg. 2014;23(1):35-42.
13. Hamada K, Fukuda H, Mikasa M, Kobayashi Y. Roentgenographic findings in massive rotator cuff tears. A long-term observation. Clin Orthop Relat Res. 1990;(254):92-96.
14. Constant CR, Murley AH. A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res. 1987;(214):160-164.
15. Michener LA, McClure PW, Sennett BJ. American Shoulder and Elbow Surgeons standardized shoulder assessment form, patient self-report section: reliability, validity, and responsiveness. J Shoulder Elbow Surg. 2002;11(6):587-594.
16. Lo IK, Griffin S, Kirkley A. The development of a disease-specific quality of life measurement tool for osteoarthritis of the shoulder: the Western Ontario Osteoarthritis of the Shoulder (WOOS) index. Osteoarthritis Cartilage. 2001;9(8):771-778.
17. Williams GN, Gangel TJ, Arciero RA, Uhorchak JM, Taylor DC. Comparison of the Single Assessment Numeric Evaluation method and two shoulder rating scales. Outcomes measures after shoulder surgery. Am J Sports Med. 1999;27(2):214-221.
18. Gartsman GM, Edwards TB. Shoulder Arthroplasty. Philadelpia, PA: Saunders Elsevier; 2008.
19. Liotard JP, Edwards TB, Padey A, Walch G, Boulahia A. Hydrotherapy rehabilitation after shoulder surgery. Tech Shoulder Elbow Surg. 2003;4:44-49.
20. Trappey GJ 4th, O’Connor DP, Edwards TB. What are the instability and infection rates after reverse shoulder arthroplasty? Clin Orthop Relat Res. 2011;469(9):2505-2511.
21. Sirveaux F, Favard L, Oudet D, Huquet D, Walch G, Molé D. Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Joint Surg Br. 2004;86(3):388-395.
22. Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6(7):438-446.
23. Schoenfeld AJ, Carey PA, Cleveland AW 3rd, Bader JO, Bono CM. Patient factors, comorbidities, and surgical characteristics that increase mortality and complication risk after spinal arthrodesis: a prognostic study based on 5,887 patients. Spine J. 2013;13(10):1171-1179.
24. Jiang J, Teng Y, Fan Z, Khan S, Xia Y. Does obesity affect the surgical outcome and complication rates of spinal surgery? A meta-analysis. Clin Orthop Relat Res. 2014;472(3):968-975.
25. Bozic KJ, Lau E, Kurtz S, et al. Patient-related risk factors for periprosthetic joint infection and postoperative mortality following total hip arthroplasty in Medicare patients. J Bone Joint Surg Am. 2012;94(9):794-800.
26. Franklin PD, Li W, Ayers DC. The Chitranjan Ranawat Award: functional outcome after total knee replacement varies with patient attributes. Clin Orthop Relat Res. 2008;466(11):2597-2604.
27. Huddleston JI, Wang Y, Uquillas C, Herndon JH, Maloney WJ. Age and obesity are risk factors for adverse events after total hip arthroplasty. Clin Orthop Relat Res. 2012;470(2):490-496.
28. Jämsen E, Nevalainen P, Eskelinen A, Huotari K, Kalliovalkama J, Moilanen T. Obesity, diabetes, and preoperative hyperglycemia as predictors of periprosthetic joint infection: a single-center analysis of 7181 primary hip and knee replacements for osteoarthritis. J Bone Joint Surg Am. 2012;94(14):e101.
29. Naziri Q, Issa K, Malkani AL, Bonutti PM, Harwin SF, Mont MA. Bariatric orthopaedics: total knee arthroplasty in super-obese patients (BMI > 50 kg/m2). Survivorship and complications. Clin Orthop Relat Res. 2013;471(11):3523-3530.
30. Warrender WJ, Brown OL, Abboud JA. Outcomes of arthroscopic rotator cuff repairs in obese patients. J Shoulder Elbow Surg. 2011;20(6):961-967.
31. Singh JA, Sperling JW, Cofield RH. Risk factors for revision surgery after humeral head replacement: 1,431 shoulders over 3 decades. J Shoulder Elbow Surg. 2012;21(8):1039-1044.
32. Chalmers PN, Rahman Z, Romeo AA, Nicholson GP. Early dislocation after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(5):737-744.
33. Li X, Williams PN, Nguyen JT, Craig EV, Warren RF, Gulotta LV. Functional outcomes after total shoulder arthroplasty in obese patients. J Bone Joint Surg Am. 2013;95(21):e160.
1. World Health Organization. Obesity and overweight [factsheet 311]. Updated January 2015. http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed March 27, 2016.
2. National Center for Health Statistics, Centers for Disease Control and Prevention. Obesity and overweight. Updated February 25, 2016. http://www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed March 27, 2016.
3. Boileau P, Gonzalez JF, Chuinard C, Bicknell R, Walch G. Reverse total shoulder arthroplasty after failed rotator cuff surgery. J Shoulder Elbow Surg. 2009;18(4):600-606.
4. Drake GN, O’Connor DP, Edwards TB. Indications for reverse total shoulder arthroplasty in rotator cuff disease. Clin Orthop Relat Res. 2010;468(6):1526-1533.
5. Gerber C, Pennington SD, Nyffeler RW. Reverse total shoulder arthroplasty. J Am Acad Orthop Surg. 2009;17(5):284-289.
6. Lenarz C, Shishani Y, McCrum C, Nowinski RJ, Edwards TB, Gobezie R. Is reverse shoulder arthroplasty appropriate for the treatment of fractures in the older patient? Early observations. Clin Orthop Relat Res. 2011;469(12):3324-3331.
7. Muh SJ, Streit JJ, Wanner JP, et al. Early follow-up of reverse total shoulder arthroplasty in patients sixty years of age or younger. J Bone Joint Surg Am. 2013;95(20):1877-1883.
8. Wall B, Nové-Josserand L, O’Connor DP, Edwards TB, Walch G. Reverse total shoulder arthroplasty: a review of results according to etiology. J Bone Joint Surg Am. 2007;89(7):1476-1485.
9. Young AA, Smith MM, Bacle G, Moraga C, Walch G. Early results of reverse shoulder arthroplasty in patients with rheumatoid arthritis. J Bone Joint Surg Am. 2011;93(20):1915-1923.
10. Boileau P, Melis B, Duperron D, Moineau G, Rumian AP, Han Y. Revision surgery of reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2013;22(10):1359-1370.
11. Beck JD, Irgit KS, Andreychik CM, Maloney PJ, Tang X, Harter GD. Reverse total shoulder arthroplasty in obese patients. J Hand Surg Am. 2013;38(5):965-970.
12. Gupta AK, Chalmers PN, Rahman Z, et al. Reverse total shoulder arthroplasty in patients of varying body mass index. J Shoulder Elbow Surg. 2014;23(1):35-42.
13. Hamada K, Fukuda H, Mikasa M, Kobayashi Y. Roentgenographic findings in massive rotator cuff tears. A long-term observation. Clin Orthop Relat Res. 1990;(254):92-96.
14. Constant CR, Murley AH. A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res. 1987;(214):160-164.
15. Michener LA, McClure PW, Sennett BJ. American Shoulder and Elbow Surgeons standardized shoulder assessment form, patient self-report section: reliability, validity, and responsiveness. J Shoulder Elbow Surg. 2002;11(6):587-594.
16. Lo IK, Griffin S, Kirkley A. The development of a disease-specific quality of life measurement tool for osteoarthritis of the shoulder: the Western Ontario Osteoarthritis of the Shoulder (WOOS) index. Osteoarthritis Cartilage. 2001;9(8):771-778.
17. Williams GN, Gangel TJ, Arciero RA, Uhorchak JM, Taylor DC. Comparison of the Single Assessment Numeric Evaluation method and two shoulder rating scales. Outcomes measures after shoulder surgery. Am J Sports Med. 1999;27(2):214-221.
18. Gartsman GM, Edwards TB. Shoulder Arthroplasty. Philadelpia, PA: Saunders Elsevier; 2008.
19. Liotard JP, Edwards TB, Padey A, Walch G, Boulahia A. Hydrotherapy rehabilitation after shoulder surgery. Tech Shoulder Elbow Surg. 2003;4:44-49.
20. Trappey GJ 4th, O’Connor DP, Edwards TB. What are the instability and infection rates after reverse shoulder arthroplasty? Clin Orthop Relat Res. 2011;469(9):2505-2511.
21. Sirveaux F, Favard L, Oudet D, Huquet D, Walch G, Molé D. Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Joint Surg Br. 2004;86(3):388-395.
22. Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis. 2006;6(7):438-446.
23. Schoenfeld AJ, Carey PA, Cleveland AW 3rd, Bader JO, Bono CM. Patient factors, comorbidities, and surgical characteristics that increase mortality and complication risk after spinal arthrodesis: a prognostic study based on 5,887 patients. Spine J. 2013;13(10):1171-1179.
24. Jiang J, Teng Y, Fan Z, Khan S, Xia Y. Does obesity affect the surgical outcome and complication rates of spinal surgery? A meta-analysis. Clin Orthop Relat Res. 2014;472(3):968-975.
25. Bozic KJ, Lau E, Kurtz S, et al. Patient-related risk factors for periprosthetic joint infection and postoperative mortality following total hip arthroplasty in Medicare patients. J Bone Joint Surg Am. 2012;94(9):794-800.
26. Franklin PD, Li W, Ayers DC. The Chitranjan Ranawat Award: functional outcome after total knee replacement varies with patient attributes. Clin Orthop Relat Res. 2008;466(11):2597-2604.
27. Huddleston JI, Wang Y, Uquillas C, Herndon JH, Maloney WJ. Age and obesity are risk factors for adverse events after total hip arthroplasty. Clin Orthop Relat Res. 2012;470(2):490-496.
28. Jämsen E, Nevalainen P, Eskelinen A, Huotari K, Kalliovalkama J, Moilanen T. Obesity, diabetes, and preoperative hyperglycemia as predictors of periprosthetic joint infection: a single-center analysis of 7181 primary hip and knee replacements for osteoarthritis. J Bone Joint Surg Am. 2012;94(14):e101.
29. Naziri Q, Issa K, Malkani AL, Bonutti PM, Harwin SF, Mont MA. Bariatric orthopaedics: total knee arthroplasty in super-obese patients (BMI > 50 kg/m2). Survivorship and complications. Clin Orthop Relat Res. 2013;471(11):3523-3530.
30. Warrender WJ, Brown OL, Abboud JA. Outcomes of arthroscopic rotator cuff repairs in obese patients. J Shoulder Elbow Surg. 2011;20(6):961-967.
31. Singh JA, Sperling JW, Cofield RH. Risk factors for revision surgery after humeral head replacement: 1,431 shoulders over 3 decades. J Shoulder Elbow Surg. 2012;21(8):1039-1044.
32. Chalmers PN, Rahman Z, Romeo AA, Nicholson GP. Early dislocation after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(5):737-744.
33. Li X, Williams PN, Nguyen JT, Craig EV, Warren RF, Gulotta LV. Functional outcomes after total shoulder arthroplasty in obese patients. J Bone Joint Surg Am. 2013;95(21):e160.
Use of an Anti-Gravity Treadmill for Early Postoperative Rehabilitation After Total Knee Replacement: A Pilot Study to Determine Safety and Feasibility
Patients undergoing total knee arthroplasty (TKA) may benefit from focused postoperative rehabilitation. Although there is limited research comparing different rehabilitation protocols after TKA,1 any type of rehabilitation often helps to optimize range of motion (ROM), strength, balance, and ambulation.2 Early mobilization and rehabilitation after TKA reduces pain, fear, anxiety, and risk of postoperative venous thromboembolic disease.3 Earlier discharge to home or community settings deceases time for inpatient rehabilitation, patient and family education, and gait training, which places a greater emphasis on outpatient rehabilitation.4
Although rapid rehabilitation protocols have gained wide acceptance, concern remains that a higher intensity intervention initiated immediately after hospital discharge could lead to an increased incidence of pain and swelling, and to poorer ROM and functional outcomes.5 Progressive weight-bearing activities, such as walking, are routinely recommended during rehabilitation to facilitate return to normal function. Not all patients are capable of full weight-bearing activity in the early postoperative period and assistive devices (ADs), such as walkers, crutches, and canes, are routinely employed. An opportunity to enhance early TKA rehabilitation exists with devices that allow functional gait training while modifying weight-bearing forces across the joint. Assistive devices, hydrotherapy (walking in water),6,7 and lower body positive-pressure chambers8 can reduce the forces at the knee during weight-bearing exercise.
Lower body positive-pressure devices have been extensively studied in physiological response of healthy humans;9-12 in disease states such as cerebral palsy13 and obesity;14 and in other postoperative orthopedic conditions, such as anterior cruciate ligament reconstruction, meniscectomy,8 microfracture,15 TKA,16 and Achilles tendon repair.17 These studies demonstrate that a lower body positive-pressure treadmill is associated with minimal cardiovascular effect while producing a significant decrease in ground reaction forces without altering gait kinematics.
We postulated that an anti-gravity treadmill may be safe and effective for gait training during rehabilitation following TKA. The primary objective was to determine the safety and feasibility of using the AlterG® Anti-Gravity Treadmill® device for postoperative gait training during rehabilitation following TKA. The secondary objective was to determine the effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on subjective patient outcomes assessed by Knee Injury and Osteoarthritis Outcome Score (KOOS), mobility assessed by the Timed Up and Go test (TUG), and pain assessed by a Numerical Rating Scale (NRS) to conduct a power analysis to determine sample sizes for efficacy studies based on these preliminary findings.
MethodsParticipants/Patient Enrollment and Study Overview
After signing an Institutional Review Board-approved consent, 30 patients were enrolled, and TKA surgeries were performed by 1 of 5 surgeons at 1 hospital. To be enrolled in the study, subjects must have (1) had a unilateral primary TKA, (2) been discharged from the hospital to home (not to a skilled nursing facility), (3) had only 3 to 4 home physical therapy (PT) sessions, (4) agreed to further outpatient PT at a single site, and (5) agreed to complete patient questionnaires. Exclusion criteria included (1) inability to meet inclusion criteria, (2) gross musculoskeletal deformity, (3) uncontrolled chronic or systemic disease, and (4) inability to follow instructions because of mental impairment, substance abuse, or addiction. Home PT was conducted for 3 to 4 sessions after surgery, and outpatient PT was continued at the study site per protocol for 4 weeks; subjects were asked to return for follow-up 3 months postoperatively. Patients were randomized on the first day of their outpatient PT to either a land-based (control) or an anti-gravity-based group using the AlterG Anti-Gravity Treadmill (AlterG group) gait training during outpatient PT sessions. Patients attended outpatient PT 2 days per week for 4 weeks for a total of 8 sessions. Therapy sessions lasted 45 to 60 minutes and included manual therapy, gait training, and therapeutic exercises/activities. The KOOS18,19 and TUG20 scores were evaluated at baseline (ie, first therapy session), end of physical therapy (EOPT) (ie, at final therapy session), and end of study (EOS) (ie, 3 months postoperatively). The NRS for pain was evaluated at baseline and at EOPT. Physical therapists were questioned for satisfaction with the anti-gravity rehabilitation protocol at EOPT.
Physical Therapy Protocols
All patients were treated consistently by 1 of 5 physical therapists at 1 outpatient setting; physical therapists averaged 11 years of experience in treating orthopedic conditions. Care was delivered in accordance with professional standards and the therapist’s assessment of medical necessity. Considerations included, but were not limited to, overall general health, any medical comorbidity, support system, and an ongoing assessment of ROM, strength, pain, and functional status. Each PT session started with a 5- to 10-minute warm-up on a standard cycle ergometer and was followed by manual therapy, gait training (land-based vs anti-gravity), therapeutic exercises/activities, and treatment modalities.
The time spent, activities selected, and modalities or physical agents chosen during the PT session were based on the patient’s needs and progress toward his/her functional goals. Manual therapy techniques consisted of soft-tissue mobilization, passive ROM, joint mobilization, passive stretching, scar mobilization, manual resistive exercises, and proprioceptive neuromuscular facilitation techniques. Therapeutic exercises/activities consisted of lower extremity resistance exercises (weight bearing and non-weight bearing), ROM exercises, stretching, balance, stair training, agility, activities of daily life (ADL) training, and a comprehensive home exercise program. Modalities or physical agents used during this study included moist hot packs, cold packs, ultrasound, electrical stimulation, and Kinesio Tape. Physical agents were incorporated into the individual’s plan of care based on medical necessity when deemed appropriate by the treating therapist. The exercise prescription was based on an individual’s status and tolerance and the number of sets and repetitions were based on fatigue.
Gait Training
The patients were randomized (1:1) to either land-based or anti-gravity gait training. For the control group, land-based gait training was performed with or without an appropriate AD and appropriate assistance, tactile cueing, and verbal cueing from a physical therapist. Duration (minutes) and gait-training progression were dependent on the participant’s functional goals, pain level (assessed throughout treatment), and level of fatigue. For the AlterG group, gait training was performed in the AlterG Anti-Gravity Treadmill, M320 (Alter-G; Figure 1). On day 1, the AlterG pressure chamber was set to allow only 50% of the patient’s body weight to be transmitted to the treadmill floor, and speed was controlled by the patient according to his/her comfort level. The percentage of body weight was adjusted to allow for a safe and normalized gait pattern with a pain level no greater than 5 (0 to 10 scale) throughout the PT session. A report card was recorded at each PT session, including body-weight setting (%), speed (miles per hour), incline (%), and duration (minutes) (Figure 2). For subsequent visits, the body-weight setting was started from the end point of the previous session.
Data Collection and Analysis
SPSS version 12.0 (SPSS Inc.) was used for all analyses, and an alpha level of .05 determined statistical significance when comparing group differences. The safety and feasibility of the anti-gravity (AlterG) vs land-based (control) gait training was assessed by the presence (or absence) of adverse events (AEs) and complications, and the date the patient discontinued use of his/her AD. A chi-square test was used to assess differences between control and AlterG groups regarding patient discontinuance of an AD. Additionally, for patients randomized to AlterG, a report card summarized means and frequencies for body weight, speed, incline, and duration. At EOPT, the frequency of therapists who were satisfied with the AlterG Anti-Gravity Treadmill as part of the rehabilitation protocol was reported. The preliminary effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on functional outcomes (subjective patient outcomes assessed by KOOS, mobility assessed by the TUG test, and pain assessed by a NRS) were evaluated by independent sample t tests. Paired sample t tests were used to compare each of the functional outcomes at EOPT or EOS to the baseline value.
Results
Of the 30 patients enrolled, 29 (96.7%; 29/30) patients completed the study; 1 patient, who could not complete all PT sessions because of medical and transportation issues, was excluded. The remaining 29 patients comprised the study population (control = 15; AlterG = 14). All patients were compliant with PT protocols.
Patient demographics were similar between the control and AlterG groups (Table 1). The control group comprised 9 women (60%; 9/15) and 6 men (40%; 6/15), age 69.9 ± 7.8 years and a body mass index of 28.8 ± 4.2. Similarly, the AlterG group comprised 7 women (50%; 7/14) and 7 men (50%; 7/14), age 66.5 ± 7.8 years and a body mass index of 28.4 ± 5.2.
At the baseline PT visit, patients in the control and AlterG groups had similar KOOS, TUG, and NRS scores. At baseline, mean KOOS for symptoms, pain, sports/recreation, ADL, and quality of life were 52.7, 52.9, 22.7, 64, and 31.8, respectively, although 50% of patients did not complete the sports/recreation subset of the KOOS. In addition, the mean time to complete the TUG test was 14.5 seconds, which was within the normal limits for disabled patients. This was slightly longer than normal mobility (TUG <10 seconds),20 but patients had relatively low levels of pain (mean NRS = 2.5, on a scale of 0-11).
All patients completed the PT protocols without indication of injury or AEs related to their operative knee. Three patients (10.3%; 3/29) experienced a deep venous thromboembolism (DVT), 2 in the control group (13.3%; 2/15), and 1 in the AlterG group (7.1%; 1/14). Venous thromboembolism protocol of enoxaparin 30 mg twice daily while in the hospital and enoxaparin 40 mg once daily for 10 days after discharge was followed for all patients.
Overall, more than half of patients (55.2%; 16/29) discontinued their AD during the 4-week PT period, with the remaining discontinuing prior to EOPT (24.1%; 7/29) or after EOPT (20.7%; 6/29). No statistically significant differences were found between the control and AlterG groups regarding discontinuance of AD.
Among those randomized to the AlterG group, all patients performed within the protocol established for the device for body-weight setting, treadmill speed, and duration of walking. The average body-weight treadmill setting increased by ~30% over the treatment period, from 55% at baseline to 84% at EOPT. The average speed increased by ~70%, from 1.6 mph at baseline to 2.7 mph at EOPT. The mean duration of AlterG use increased by ~75%, from 7.2 minutes at baseline to 12.7 minutes at EOPT. All physical therapists (100%) reported satisfaction with the AlterG for use in early postoperative rehabilitation and reported that patients’ treatment progressed positively.
While functional outcomes (KOOS, TUG, or NRS) did not vary with the type of gait training (P > .2 for land-based vs anti-gravity), functional outcomes improved over time (all P < .01 from baseline to EOPT and all P < .01 from baseline to EOS).
The KOOS scores improved from baseline to EOPT and from baseline to EOS (ie, 3-month follow-up visit) for both treatment groups (Figure 3). More patients completed the sports/recreation portion of the KOOS scores at EOPT and EOS compared to baseline. Forty-three percent and 25% of patients did not complete KOOS sports/recreation questions at EOPT and EOS, respectively, compared to 50% at baseline. This suggests that patients were improving to a level where sports/recreation scores were more applicable than directly after TKA surgery. The TUG scores had the greatest improvement from baseline to EOPT, with a decrease in time of 5 seconds and 7 seconds for the control and AlterG groups, respectively, and slight improvement from EOPT to EOS, with a decrease in time of 1 second and 2 seconds for the control and AlterG groups, respectively (Table 2). By the EOS, the values for the TUG tests for both treatment groups were within normal (<10 seconds) range.20 The NRS scores improved from baseline to EOPT with a score of 1 ± 1 in both control and AlterG groups.
Using these preliminary efficacy results, a post-hoc power analysis (α = .05 and 1β = 80%) was performed with the ADL domain of KOOS as the primary endpoint. Based on a standard deviation of 20 points and an effect size of 5 points, the sample size was estimated to be N = 250 per treatment group.
Discussion
We conducted a pilot study to assess, primarily, the feasibility and safety, and, secondarily, the efficacy, of a lower body positive-pressure treadmill for rehabilitation of patients after TKA. This small study showed that use of the AlterG Anti-Gravity Treadmill was not only safe and feasible during postoperative TKA rehabilitation, but also was well tolerated by patients and was rated highly satisfactory by physical therapists. Patients who used AlterG during gait training improved functionally (in terms of KOOS, TUG, and NRS) after 8 treatment sessions compared to baseline. However, there were no statistical differences between groups (control vs AlterG). Thus, these results suggest that an anti-gravity device for gait training may be a useful adjunct for postoperative TKA rehabilitation, but further studies are needed to determine the efficacy of anti-gravity compared to traditional land-based gait training.
The study of rehabilitation protocols during postoperative PT involved consideration of a number of issues. First, differences in functional outcomes compared to traditional rehabilitation could not be detected in this study because of the small number of patients, but the patients treated with anti-gravity gait training showed improvement in functional outcomes over time and did not report any added complications. Given that the primary outcome of this study was safety and feasibility, these added efficacy results are supplemental and useful in helping to plan studies. Second, the functional outcomes used to measure the efficacy of the anti-gravity treadmill may not be sensitive enough to detect differences between rehabilitation protocols. Use of a treadmill to measure speed improvement, endurance, and tolerance in both groups could be valuable in future studies. More studies may need to refine characteristics that are important to postoperative rehabilitation success, and quantitative and subjective measures that must be defined.
The results reported here using an anti-gravity treadmill for postoperative TKA rehabilitation support the safety and feasibility that has been reported in other orthopedic rehabilitation settings. Anti-gravity treadmills, which have been used to study patients after meniscectomy or anterior cruciate ligament reconstruction8 and Achilles repair,17 have demonstrated predictable decreases in ground reaction forces with increasing positive-pressure unweighting, reductions in pain with ambulation, and allowance of earlier institution of walking and jogging during rehabilitation.17
Patient safety is an important attribute for any postoperative rehabilitation protocol, especially in an elderly population undergoing major surgery. One of our important goals was to assess the safety of AlterG. We noted no AEs attributable to the device, which was supported by work indicating no adverse impact on systemic cardiovascular parameters in a similar lower body positive-pressure environment.9 Although 3 patients (10%) developed symptomatic DVT, there were no differences between the groups in the incidence of DVT. Use of an anti-gravity treadmill has also been examined for cardiovascular responses in TKA patients. In a study of 24 adults with TKA, researchers found that anti-gravity support allowed TKA patients to walk at faster speeds and tolerate greater inclines with lower heart rate, blood pressure, and oxygen consumption.21 With respect to efficacy of the rehabilitation intervention, we demonstrated significant improvements in all functional outcomes in both groups but no differences between the study groups. We concluded that AlterG was at least as effective as standard therapy in this small cohort. TKA is a very successful procedure, and the improvement in pain and function after surgery is fairly dramatic in most patients, regardless of specific rehabilitation protocols. Therefore, the substantial improvement in clinical outcomes may overshadow any enhanced benefits of the anti-gravity treadmill. Further investigations into the efficacy of AlterG are needed in a larger cohort to determine if this type of treatment is more beneficial than traditional land-based gait training.
Standard scoring systems such as KOOS, TUG, and NRS may not be sensitive enough to detect differences between treatment groups with small sample sizes. Given the results of the post hoc power analysis, a large number of patients (N = 250/group) would be necessary to detect any potential difference in clinical outcomes between the 2 groups. Larger studies are required to answer relevant questions, and additional outcome measures may be needed to detect differences between treatment groups. Relevant questions include whether earlier institution of the anti-gravity device during the immediate TKA postoperative period would be beneficial compared to standard postoperative PT, and whether PT enhanced with the anti-gravity device has incremental benefit in functional outcomes and in time to reach those goals. Finally, given the present attention to healthcare expenses, a cost-benefit analysis of anti-gravity device treatment vs traditional PT would be useful. Once the patient has become familiar with the function of an anti-gravity treadmill, gait therapy could proceed without the direct intervention of the therapist, potentially improving efficient delivery of rehabilitation services.
Studying the effect of different postoperative rehabilitation protocols after orthopedic surgeries can be challenging. In a large (N > 350) randomized controlled trial to study the effect of ergometer cycling after hip and knee replacement, patients who used the cycle ergometer had a higher Western Ontario and McMaster Universities Arthritis Index and greater satisfaction than those who did not after hip arthroplasty, but not after TKA.22 Improvements in muscular coordination and proprioception with the cycle ergometer may have been offset by increases in edema, joint effusion, and pain from the loading of the joint and the relatively fast rate of cycling compared to passive motion or ambulation. While many therapists and surgeons advocate cycling for rehabilitation after knee surgery, the need remains for a better definition of an optimal TKA rehabilitation program. A study of 82 patients comparing early progressive strength training to no early strength training showed no difference in the 6-minute walk test at 8 weeks.23 A systematic review of progressive resistance training (PRT) found that although postoperative PRT is safe and feasible, the methodological quality of existing studies is too low to allow conclusions regarding its efficacy.24 Gait training in an environment where weight-bearing loads can be closely controlled, monitored, and individualized may be an ideal methodology to enhance rehabilitation and return to function for knee replacement surgery.
This current study showed that the use of AlterG as an adjunct for postoperative rehabilitation is safe, accepted by patients and therapists, and leads to clinical functional outcomes that are at least as good as traditional postoperative TKA rehabilitation. We conclude that AlterG demonstrates utility and a potential for innovation in TKA rehabilitation.
1. NIH Consensus Statement on total knee arthroplasty. NIH Consensus State Sci Statements. 2003;20(1):1-34.
2. Jones CA, Voaklander DC, Suarez-Almazor ME. Determinants of function after total knee arthroplasty. Phys Ther. 2003;83(8):696-706.
3. Pearse EO, Caldwell BF, Lockwood RJ, Hollard J. Early mobilisation after conventional knee replacement may reduce the risk of post-operative venous thromboembolism. J Bone Joint Surg Br. 2007;89(3):316-322.
4. Westby MD, Kennedy D, Jones D, Jones A, Doyle-Waters MM, Backman C. Post-acute physiotherapy for primary total knee arthroplasty. Cochrane Database Syst Rev. 2008. doi.10.1002/14651858.CD007099
5. Bade MJ, Stevens-Lapsley JE. Early high-intensity rehabilitation following total knee arthroplasty improves outcomes. J Orthop Sports Phys Ther. 2011;41(12):932-941.
6. Ivanenko YP, Grasso R, Macellari V, Lacquaniti F. Control of foot trajectory in human locomotion: role of ground contact forces in simulated reduced gravity. J Neurophysiol. 2002;87(6):3070-3089.
7. Pöyhönen T, Keskinen KL, Kyröläinen H, Hautala A, Savolainen J, Mälkiä E. Neuromuscular function during therapeutic knee exercise under water and on dry land. Arch Phys Med Rehabil. 2001;82(10):1446-1452.
8. Eastlack RK, Hargens AR, Groppo ER, Steinbach GC, White KK, Pedowitz RA. Lower body positive-pressure exercise after knee surgery. Clin Orthop Rel Res. 2005;431:213-219.
9. Cutuk A, Groppo ER, Quigley EJ, White KW, Pedowitz RA, Hargens AR. Ambulation in simulated fractional gravity using lower body positive pressure: cardiovascular safety and gait analyses. J Appl Physiol. 2006;101(3):771-777.
10. Gojanovic B, Cutti P, Shultz R, Matheson GO. Maximal physiological parameters during partial body-weight support treadmill testing. Med Sci Sports Exerc. 2012;44(10):1935-1941.
11. Figueroa MA, Manning J, Escamilla P. Physiological responses to the AlterG Anti-Gravity Treadmill. Int J Applied Sci Tech. 2011;1:92-97.
12. Hoffman MD, Donaghe HE. Physiological responses to body weight-supported treadmill exercise in healthy adults. Arch Phys Med Rehabil. 2011;92(6):960-966.
13. Kurz MJ, Corr B, Stuberg W, Volkman KG, Smith N. Evaluation of lower body positive pressure supported treadmill training for children with cerebral palsy. Pediatr Phys Ther. 2011;23(3):232-239.
14. Christian M. Managing knee osteoarthritis: the effects of anti-gravity treadmill exercise on joint pain and physical function. Available at: http://mspace.lib.umanitoba.ca/handle/1993/8580. Accessed March 31, 2016.
15. Wilk KE, Macrina LC, Reinhold MM. Rehabilitation following microfracture of the knee. Cartilage. 2010;1(2):96-107.
16. Patil SS, Branovacki G, Martin MR, Pulido PA, Levy YD, Colwell CW Jr. 14-year median follow-up using the press-fit condylar sigma design for total knee arthroplasty. J Arthroplasty. 2013;28(8):1286-1290.
17. Saxena A, Granot A. Use of an anti-gravity treadmill in the rehabilitation of the operated achilles tendon: a pilot study. J Foot Ankle Surg. 2011;50(5):558-561.
18. Roos EM, Roos HP, Ekdahl C, Lohmander LS. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation of a Swedish version. Scand J Med Sci Sports. 1998;8(6):439-448.
19. Roos EM, Toksvig-Larsen S. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation and comparison to the WOMAC in total knee replacement. Health Qual Life Outcomes. 2003;1:17.
20. Timed Up and Go (TUG). Available at: http://www.rheumatology.org/I-Am-A/Rheumatologist/Research/Clinician-Researchers/Timed-Up-Go-TUG Accessed: March 15, 2016.
21. Webber SC, Horvey KJ, Yurach Pikaluk MT, Butcher SJ. Cardiovascular responses in older adults with total knee arthroplasty at rest and with exercise on a positive pressure treadmill. Eur J Appl Physiol. 2014;114(3):653-662.
22. Liebs TR, Herzberg W, Ruther W, Haasters J, Russlies M, Hassenpflug J. Ergometer cycling after hip and knee replacement surgery: a randomized control trial. J Bone Joint Surg Am. 2010;92(4):814-822.
23. Jakobsen TL, Kehlet H, Husted H, Petersen J, Bandholm T. Early progressive strength training to enhance recovery after fast-track total knee arthroplasty: a randomized controlled trial. Arthritis Care Res. 2014;66(12):1856-1866.
24. Skoffer B, Dalgas U, Mechlenburg I. Progressive resistance training before and after total hip and knee arthroplasty: a systematic review. Clin Rehabil. 2015;29(1):14-29.
Patients undergoing total knee arthroplasty (TKA) may benefit from focused postoperative rehabilitation. Although there is limited research comparing different rehabilitation protocols after TKA,1 any type of rehabilitation often helps to optimize range of motion (ROM), strength, balance, and ambulation.2 Early mobilization and rehabilitation after TKA reduces pain, fear, anxiety, and risk of postoperative venous thromboembolic disease.3 Earlier discharge to home or community settings deceases time for inpatient rehabilitation, patient and family education, and gait training, which places a greater emphasis on outpatient rehabilitation.4
Although rapid rehabilitation protocols have gained wide acceptance, concern remains that a higher intensity intervention initiated immediately after hospital discharge could lead to an increased incidence of pain and swelling, and to poorer ROM and functional outcomes.5 Progressive weight-bearing activities, such as walking, are routinely recommended during rehabilitation to facilitate return to normal function. Not all patients are capable of full weight-bearing activity in the early postoperative period and assistive devices (ADs), such as walkers, crutches, and canes, are routinely employed. An opportunity to enhance early TKA rehabilitation exists with devices that allow functional gait training while modifying weight-bearing forces across the joint. Assistive devices, hydrotherapy (walking in water),6,7 and lower body positive-pressure chambers8 can reduce the forces at the knee during weight-bearing exercise.
Lower body positive-pressure devices have been extensively studied in physiological response of healthy humans;9-12 in disease states such as cerebral palsy13 and obesity;14 and in other postoperative orthopedic conditions, such as anterior cruciate ligament reconstruction, meniscectomy,8 microfracture,15 TKA,16 and Achilles tendon repair.17 These studies demonstrate that a lower body positive-pressure treadmill is associated with minimal cardiovascular effect while producing a significant decrease in ground reaction forces without altering gait kinematics.
We postulated that an anti-gravity treadmill may be safe and effective for gait training during rehabilitation following TKA. The primary objective was to determine the safety and feasibility of using the AlterG® Anti-Gravity Treadmill® device for postoperative gait training during rehabilitation following TKA. The secondary objective was to determine the effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on subjective patient outcomes assessed by Knee Injury and Osteoarthritis Outcome Score (KOOS), mobility assessed by the Timed Up and Go test (TUG), and pain assessed by a Numerical Rating Scale (NRS) to conduct a power analysis to determine sample sizes for efficacy studies based on these preliminary findings.
MethodsParticipants/Patient Enrollment and Study Overview
After signing an Institutional Review Board-approved consent, 30 patients were enrolled, and TKA surgeries were performed by 1 of 5 surgeons at 1 hospital. To be enrolled in the study, subjects must have (1) had a unilateral primary TKA, (2) been discharged from the hospital to home (not to a skilled nursing facility), (3) had only 3 to 4 home physical therapy (PT) sessions, (4) agreed to further outpatient PT at a single site, and (5) agreed to complete patient questionnaires. Exclusion criteria included (1) inability to meet inclusion criteria, (2) gross musculoskeletal deformity, (3) uncontrolled chronic or systemic disease, and (4) inability to follow instructions because of mental impairment, substance abuse, or addiction. Home PT was conducted for 3 to 4 sessions after surgery, and outpatient PT was continued at the study site per protocol for 4 weeks; subjects were asked to return for follow-up 3 months postoperatively. Patients were randomized on the first day of their outpatient PT to either a land-based (control) or an anti-gravity-based group using the AlterG Anti-Gravity Treadmill (AlterG group) gait training during outpatient PT sessions. Patients attended outpatient PT 2 days per week for 4 weeks for a total of 8 sessions. Therapy sessions lasted 45 to 60 minutes and included manual therapy, gait training, and therapeutic exercises/activities. The KOOS18,19 and TUG20 scores were evaluated at baseline (ie, first therapy session), end of physical therapy (EOPT) (ie, at final therapy session), and end of study (EOS) (ie, 3 months postoperatively). The NRS for pain was evaluated at baseline and at EOPT. Physical therapists were questioned for satisfaction with the anti-gravity rehabilitation protocol at EOPT.
Physical Therapy Protocols
All patients were treated consistently by 1 of 5 physical therapists at 1 outpatient setting; physical therapists averaged 11 years of experience in treating orthopedic conditions. Care was delivered in accordance with professional standards and the therapist’s assessment of medical necessity. Considerations included, but were not limited to, overall general health, any medical comorbidity, support system, and an ongoing assessment of ROM, strength, pain, and functional status. Each PT session started with a 5- to 10-minute warm-up on a standard cycle ergometer and was followed by manual therapy, gait training (land-based vs anti-gravity), therapeutic exercises/activities, and treatment modalities.
The time spent, activities selected, and modalities or physical agents chosen during the PT session were based on the patient’s needs and progress toward his/her functional goals. Manual therapy techniques consisted of soft-tissue mobilization, passive ROM, joint mobilization, passive stretching, scar mobilization, manual resistive exercises, and proprioceptive neuromuscular facilitation techniques. Therapeutic exercises/activities consisted of lower extremity resistance exercises (weight bearing and non-weight bearing), ROM exercises, stretching, balance, stair training, agility, activities of daily life (ADL) training, and a comprehensive home exercise program. Modalities or physical agents used during this study included moist hot packs, cold packs, ultrasound, electrical stimulation, and Kinesio Tape. Physical agents were incorporated into the individual’s plan of care based on medical necessity when deemed appropriate by the treating therapist. The exercise prescription was based on an individual’s status and tolerance and the number of sets and repetitions were based on fatigue.
Gait Training
The patients were randomized (1:1) to either land-based or anti-gravity gait training. For the control group, land-based gait training was performed with or without an appropriate AD and appropriate assistance, tactile cueing, and verbal cueing from a physical therapist. Duration (minutes) and gait-training progression were dependent on the participant’s functional goals, pain level (assessed throughout treatment), and level of fatigue. For the AlterG group, gait training was performed in the AlterG Anti-Gravity Treadmill, M320 (Alter-G; Figure 1). On day 1, the AlterG pressure chamber was set to allow only 50% of the patient’s body weight to be transmitted to the treadmill floor, and speed was controlled by the patient according to his/her comfort level. The percentage of body weight was adjusted to allow for a safe and normalized gait pattern with a pain level no greater than 5 (0 to 10 scale) throughout the PT session. A report card was recorded at each PT session, including body-weight setting (%), speed (miles per hour), incline (%), and duration (minutes) (Figure 2). For subsequent visits, the body-weight setting was started from the end point of the previous session.
Data Collection and Analysis
SPSS version 12.0 (SPSS Inc.) was used for all analyses, and an alpha level of .05 determined statistical significance when comparing group differences. The safety and feasibility of the anti-gravity (AlterG) vs land-based (control) gait training was assessed by the presence (or absence) of adverse events (AEs) and complications, and the date the patient discontinued use of his/her AD. A chi-square test was used to assess differences between control and AlterG groups regarding patient discontinuance of an AD. Additionally, for patients randomized to AlterG, a report card summarized means and frequencies for body weight, speed, incline, and duration. At EOPT, the frequency of therapists who were satisfied with the AlterG Anti-Gravity Treadmill as part of the rehabilitation protocol was reported. The preliminary effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on functional outcomes (subjective patient outcomes assessed by KOOS, mobility assessed by the TUG test, and pain assessed by a NRS) were evaluated by independent sample t tests. Paired sample t tests were used to compare each of the functional outcomes at EOPT or EOS to the baseline value.
Results
Of the 30 patients enrolled, 29 (96.7%; 29/30) patients completed the study; 1 patient, who could not complete all PT sessions because of medical and transportation issues, was excluded. The remaining 29 patients comprised the study population (control = 15; AlterG = 14). All patients were compliant with PT protocols.
Patient demographics were similar between the control and AlterG groups (Table 1). The control group comprised 9 women (60%; 9/15) and 6 men (40%; 6/15), age 69.9 ± 7.8 years and a body mass index of 28.8 ± 4.2. Similarly, the AlterG group comprised 7 women (50%; 7/14) and 7 men (50%; 7/14), age 66.5 ± 7.8 years and a body mass index of 28.4 ± 5.2.
At the baseline PT visit, patients in the control and AlterG groups had similar KOOS, TUG, and NRS scores. At baseline, mean KOOS for symptoms, pain, sports/recreation, ADL, and quality of life were 52.7, 52.9, 22.7, 64, and 31.8, respectively, although 50% of patients did not complete the sports/recreation subset of the KOOS. In addition, the mean time to complete the TUG test was 14.5 seconds, which was within the normal limits for disabled patients. This was slightly longer than normal mobility (TUG <10 seconds),20 but patients had relatively low levels of pain (mean NRS = 2.5, on a scale of 0-11).
All patients completed the PT protocols without indication of injury or AEs related to their operative knee. Three patients (10.3%; 3/29) experienced a deep venous thromboembolism (DVT), 2 in the control group (13.3%; 2/15), and 1 in the AlterG group (7.1%; 1/14). Venous thromboembolism protocol of enoxaparin 30 mg twice daily while in the hospital and enoxaparin 40 mg once daily for 10 days after discharge was followed for all patients.
Overall, more than half of patients (55.2%; 16/29) discontinued their AD during the 4-week PT period, with the remaining discontinuing prior to EOPT (24.1%; 7/29) or after EOPT (20.7%; 6/29). No statistically significant differences were found between the control and AlterG groups regarding discontinuance of AD.
Among those randomized to the AlterG group, all patients performed within the protocol established for the device for body-weight setting, treadmill speed, and duration of walking. The average body-weight treadmill setting increased by ~30% over the treatment period, from 55% at baseline to 84% at EOPT. The average speed increased by ~70%, from 1.6 mph at baseline to 2.7 mph at EOPT. The mean duration of AlterG use increased by ~75%, from 7.2 minutes at baseline to 12.7 minutes at EOPT. All physical therapists (100%) reported satisfaction with the AlterG for use in early postoperative rehabilitation and reported that patients’ treatment progressed positively.
While functional outcomes (KOOS, TUG, or NRS) did not vary with the type of gait training (P > .2 for land-based vs anti-gravity), functional outcomes improved over time (all P < .01 from baseline to EOPT and all P < .01 from baseline to EOS).
The KOOS scores improved from baseline to EOPT and from baseline to EOS (ie, 3-month follow-up visit) for both treatment groups (Figure 3). More patients completed the sports/recreation portion of the KOOS scores at EOPT and EOS compared to baseline. Forty-three percent and 25% of patients did not complete KOOS sports/recreation questions at EOPT and EOS, respectively, compared to 50% at baseline. This suggests that patients were improving to a level where sports/recreation scores were more applicable than directly after TKA surgery. The TUG scores had the greatest improvement from baseline to EOPT, with a decrease in time of 5 seconds and 7 seconds for the control and AlterG groups, respectively, and slight improvement from EOPT to EOS, with a decrease in time of 1 second and 2 seconds for the control and AlterG groups, respectively (Table 2). By the EOS, the values for the TUG tests for both treatment groups were within normal (<10 seconds) range.20 The NRS scores improved from baseline to EOPT with a score of 1 ± 1 in both control and AlterG groups.
Using these preliminary efficacy results, a post-hoc power analysis (α = .05 and 1β = 80%) was performed with the ADL domain of KOOS as the primary endpoint. Based on a standard deviation of 20 points and an effect size of 5 points, the sample size was estimated to be N = 250 per treatment group.
Discussion
We conducted a pilot study to assess, primarily, the feasibility and safety, and, secondarily, the efficacy, of a lower body positive-pressure treadmill for rehabilitation of patients after TKA. This small study showed that use of the AlterG Anti-Gravity Treadmill was not only safe and feasible during postoperative TKA rehabilitation, but also was well tolerated by patients and was rated highly satisfactory by physical therapists. Patients who used AlterG during gait training improved functionally (in terms of KOOS, TUG, and NRS) after 8 treatment sessions compared to baseline. However, there were no statistical differences between groups (control vs AlterG). Thus, these results suggest that an anti-gravity device for gait training may be a useful adjunct for postoperative TKA rehabilitation, but further studies are needed to determine the efficacy of anti-gravity compared to traditional land-based gait training.
The study of rehabilitation protocols during postoperative PT involved consideration of a number of issues. First, differences in functional outcomes compared to traditional rehabilitation could not be detected in this study because of the small number of patients, but the patients treated with anti-gravity gait training showed improvement in functional outcomes over time and did not report any added complications. Given that the primary outcome of this study was safety and feasibility, these added efficacy results are supplemental and useful in helping to plan studies. Second, the functional outcomes used to measure the efficacy of the anti-gravity treadmill may not be sensitive enough to detect differences between rehabilitation protocols. Use of a treadmill to measure speed improvement, endurance, and tolerance in both groups could be valuable in future studies. More studies may need to refine characteristics that are important to postoperative rehabilitation success, and quantitative and subjective measures that must be defined.
The results reported here using an anti-gravity treadmill for postoperative TKA rehabilitation support the safety and feasibility that has been reported in other orthopedic rehabilitation settings. Anti-gravity treadmills, which have been used to study patients after meniscectomy or anterior cruciate ligament reconstruction8 and Achilles repair,17 have demonstrated predictable decreases in ground reaction forces with increasing positive-pressure unweighting, reductions in pain with ambulation, and allowance of earlier institution of walking and jogging during rehabilitation.17
Patient safety is an important attribute for any postoperative rehabilitation protocol, especially in an elderly population undergoing major surgery. One of our important goals was to assess the safety of AlterG. We noted no AEs attributable to the device, which was supported by work indicating no adverse impact on systemic cardiovascular parameters in a similar lower body positive-pressure environment.9 Although 3 patients (10%) developed symptomatic DVT, there were no differences between the groups in the incidence of DVT. Use of an anti-gravity treadmill has also been examined for cardiovascular responses in TKA patients. In a study of 24 adults with TKA, researchers found that anti-gravity support allowed TKA patients to walk at faster speeds and tolerate greater inclines with lower heart rate, blood pressure, and oxygen consumption.21 With respect to efficacy of the rehabilitation intervention, we demonstrated significant improvements in all functional outcomes in both groups but no differences between the study groups. We concluded that AlterG was at least as effective as standard therapy in this small cohort. TKA is a very successful procedure, and the improvement in pain and function after surgery is fairly dramatic in most patients, regardless of specific rehabilitation protocols. Therefore, the substantial improvement in clinical outcomes may overshadow any enhanced benefits of the anti-gravity treadmill. Further investigations into the efficacy of AlterG are needed in a larger cohort to determine if this type of treatment is more beneficial than traditional land-based gait training.
Standard scoring systems such as KOOS, TUG, and NRS may not be sensitive enough to detect differences between treatment groups with small sample sizes. Given the results of the post hoc power analysis, a large number of patients (N = 250/group) would be necessary to detect any potential difference in clinical outcomes between the 2 groups. Larger studies are required to answer relevant questions, and additional outcome measures may be needed to detect differences between treatment groups. Relevant questions include whether earlier institution of the anti-gravity device during the immediate TKA postoperative period would be beneficial compared to standard postoperative PT, and whether PT enhanced with the anti-gravity device has incremental benefit in functional outcomes and in time to reach those goals. Finally, given the present attention to healthcare expenses, a cost-benefit analysis of anti-gravity device treatment vs traditional PT would be useful. Once the patient has become familiar with the function of an anti-gravity treadmill, gait therapy could proceed without the direct intervention of the therapist, potentially improving efficient delivery of rehabilitation services.
Studying the effect of different postoperative rehabilitation protocols after orthopedic surgeries can be challenging. In a large (N > 350) randomized controlled trial to study the effect of ergometer cycling after hip and knee replacement, patients who used the cycle ergometer had a higher Western Ontario and McMaster Universities Arthritis Index and greater satisfaction than those who did not after hip arthroplasty, but not after TKA.22 Improvements in muscular coordination and proprioception with the cycle ergometer may have been offset by increases in edema, joint effusion, and pain from the loading of the joint and the relatively fast rate of cycling compared to passive motion or ambulation. While many therapists and surgeons advocate cycling for rehabilitation after knee surgery, the need remains for a better definition of an optimal TKA rehabilitation program. A study of 82 patients comparing early progressive strength training to no early strength training showed no difference in the 6-minute walk test at 8 weeks.23 A systematic review of progressive resistance training (PRT) found that although postoperative PRT is safe and feasible, the methodological quality of existing studies is too low to allow conclusions regarding its efficacy.24 Gait training in an environment where weight-bearing loads can be closely controlled, monitored, and individualized may be an ideal methodology to enhance rehabilitation and return to function for knee replacement surgery.
This current study showed that the use of AlterG as an adjunct for postoperative rehabilitation is safe, accepted by patients and therapists, and leads to clinical functional outcomes that are at least as good as traditional postoperative TKA rehabilitation. We conclude that AlterG demonstrates utility and a potential for innovation in TKA rehabilitation.
Patients undergoing total knee arthroplasty (TKA) may benefit from focused postoperative rehabilitation. Although there is limited research comparing different rehabilitation protocols after TKA,1 any type of rehabilitation often helps to optimize range of motion (ROM), strength, balance, and ambulation.2 Early mobilization and rehabilitation after TKA reduces pain, fear, anxiety, and risk of postoperative venous thromboembolic disease.3 Earlier discharge to home or community settings deceases time for inpatient rehabilitation, patient and family education, and gait training, which places a greater emphasis on outpatient rehabilitation.4
Although rapid rehabilitation protocols have gained wide acceptance, concern remains that a higher intensity intervention initiated immediately after hospital discharge could lead to an increased incidence of pain and swelling, and to poorer ROM and functional outcomes.5 Progressive weight-bearing activities, such as walking, are routinely recommended during rehabilitation to facilitate return to normal function. Not all patients are capable of full weight-bearing activity in the early postoperative period and assistive devices (ADs), such as walkers, crutches, and canes, are routinely employed. An opportunity to enhance early TKA rehabilitation exists with devices that allow functional gait training while modifying weight-bearing forces across the joint. Assistive devices, hydrotherapy (walking in water),6,7 and lower body positive-pressure chambers8 can reduce the forces at the knee during weight-bearing exercise.
Lower body positive-pressure devices have been extensively studied in physiological response of healthy humans;9-12 in disease states such as cerebral palsy13 and obesity;14 and in other postoperative orthopedic conditions, such as anterior cruciate ligament reconstruction, meniscectomy,8 microfracture,15 TKA,16 and Achilles tendon repair.17 These studies demonstrate that a lower body positive-pressure treadmill is associated with minimal cardiovascular effect while producing a significant decrease in ground reaction forces without altering gait kinematics.
We postulated that an anti-gravity treadmill may be safe and effective for gait training during rehabilitation following TKA. The primary objective was to determine the safety and feasibility of using the AlterG® Anti-Gravity Treadmill® device for postoperative gait training during rehabilitation following TKA. The secondary objective was to determine the effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on subjective patient outcomes assessed by Knee Injury and Osteoarthritis Outcome Score (KOOS), mobility assessed by the Timed Up and Go test (TUG), and pain assessed by a Numerical Rating Scale (NRS) to conduct a power analysis to determine sample sizes for efficacy studies based on these preliminary findings.
MethodsParticipants/Patient Enrollment and Study Overview
After signing an Institutional Review Board-approved consent, 30 patients were enrolled, and TKA surgeries were performed by 1 of 5 surgeons at 1 hospital. To be enrolled in the study, subjects must have (1) had a unilateral primary TKA, (2) been discharged from the hospital to home (not to a skilled nursing facility), (3) had only 3 to 4 home physical therapy (PT) sessions, (4) agreed to further outpatient PT at a single site, and (5) agreed to complete patient questionnaires. Exclusion criteria included (1) inability to meet inclusion criteria, (2) gross musculoskeletal deformity, (3) uncontrolled chronic or systemic disease, and (4) inability to follow instructions because of mental impairment, substance abuse, or addiction. Home PT was conducted for 3 to 4 sessions after surgery, and outpatient PT was continued at the study site per protocol for 4 weeks; subjects were asked to return for follow-up 3 months postoperatively. Patients were randomized on the first day of their outpatient PT to either a land-based (control) or an anti-gravity-based group using the AlterG Anti-Gravity Treadmill (AlterG group) gait training during outpatient PT sessions. Patients attended outpatient PT 2 days per week for 4 weeks for a total of 8 sessions. Therapy sessions lasted 45 to 60 minutes and included manual therapy, gait training, and therapeutic exercises/activities. The KOOS18,19 and TUG20 scores were evaluated at baseline (ie, first therapy session), end of physical therapy (EOPT) (ie, at final therapy session), and end of study (EOS) (ie, 3 months postoperatively). The NRS for pain was evaluated at baseline and at EOPT. Physical therapists were questioned for satisfaction with the anti-gravity rehabilitation protocol at EOPT.
Physical Therapy Protocols
All patients were treated consistently by 1 of 5 physical therapists at 1 outpatient setting; physical therapists averaged 11 years of experience in treating orthopedic conditions. Care was delivered in accordance with professional standards and the therapist’s assessment of medical necessity. Considerations included, but were not limited to, overall general health, any medical comorbidity, support system, and an ongoing assessment of ROM, strength, pain, and functional status. Each PT session started with a 5- to 10-minute warm-up on a standard cycle ergometer and was followed by manual therapy, gait training (land-based vs anti-gravity), therapeutic exercises/activities, and treatment modalities.
The time spent, activities selected, and modalities or physical agents chosen during the PT session were based on the patient’s needs and progress toward his/her functional goals. Manual therapy techniques consisted of soft-tissue mobilization, passive ROM, joint mobilization, passive stretching, scar mobilization, manual resistive exercises, and proprioceptive neuromuscular facilitation techniques. Therapeutic exercises/activities consisted of lower extremity resistance exercises (weight bearing and non-weight bearing), ROM exercises, stretching, balance, stair training, agility, activities of daily life (ADL) training, and a comprehensive home exercise program. Modalities or physical agents used during this study included moist hot packs, cold packs, ultrasound, electrical stimulation, and Kinesio Tape. Physical agents were incorporated into the individual’s plan of care based on medical necessity when deemed appropriate by the treating therapist. The exercise prescription was based on an individual’s status and tolerance and the number of sets and repetitions were based on fatigue.
Gait Training
The patients were randomized (1:1) to either land-based or anti-gravity gait training. For the control group, land-based gait training was performed with or without an appropriate AD and appropriate assistance, tactile cueing, and verbal cueing from a physical therapist. Duration (minutes) and gait-training progression were dependent on the participant’s functional goals, pain level (assessed throughout treatment), and level of fatigue. For the AlterG group, gait training was performed in the AlterG Anti-Gravity Treadmill, M320 (Alter-G; Figure 1). On day 1, the AlterG pressure chamber was set to allow only 50% of the patient’s body weight to be transmitted to the treadmill floor, and speed was controlled by the patient according to his/her comfort level. The percentage of body weight was adjusted to allow for a safe and normalized gait pattern with a pain level no greater than 5 (0 to 10 scale) throughout the PT session. A report card was recorded at each PT session, including body-weight setting (%), speed (miles per hour), incline (%), and duration (minutes) (Figure 2). For subsequent visits, the body-weight setting was started from the end point of the previous session.
Data Collection and Analysis
SPSS version 12.0 (SPSS Inc.) was used for all analyses, and an alpha level of .05 determined statistical significance when comparing group differences. The safety and feasibility of the anti-gravity (AlterG) vs land-based (control) gait training was assessed by the presence (or absence) of adverse events (AEs) and complications, and the date the patient discontinued use of his/her AD. A chi-square test was used to assess differences between control and AlterG groups regarding patient discontinuance of an AD. Additionally, for patients randomized to AlterG, a report card summarized means and frequencies for body weight, speed, incline, and duration. At EOPT, the frequency of therapists who were satisfied with the AlterG Anti-Gravity Treadmill as part of the rehabilitation protocol was reported. The preliminary effects of gait training (land-based vs anti-gravity) during postoperative rehabilitation on functional outcomes (subjective patient outcomes assessed by KOOS, mobility assessed by the TUG test, and pain assessed by a NRS) were evaluated by independent sample t tests. Paired sample t tests were used to compare each of the functional outcomes at EOPT or EOS to the baseline value.
Results
Of the 30 patients enrolled, 29 (96.7%; 29/30) patients completed the study; 1 patient, who could not complete all PT sessions because of medical and transportation issues, was excluded. The remaining 29 patients comprised the study population (control = 15; AlterG = 14). All patients were compliant with PT protocols.
Patient demographics were similar between the control and AlterG groups (Table 1). The control group comprised 9 women (60%; 9/15) and 6 men (40%; 6/15), age 69.9 ± 7.8 years and a body mass index of 28.8 ± 4.2. Similarly, the AlterG group comprised 7 women (50%; 7/14) and 7 men (50%; 7/14), age 66.5 ± 7.8 years and a body mass index of 28.4 ± 5.2.
At the baseline PT visit, patients in the control and AlterG groups had similar KOOS, TUG, and NRS scores. At baseline, mean KOOS for symptoms, pain, sports/recreation, ADL, and quality of life were 52.7, 52.9, 22.7, 64, and 31.8, respectively, although 50% of patients did not complete the sports/recreation subset of the KOOS. In addition, the mean time to complete the TUG test was 14.5 seconds, which was within the normal limits for disabled patients. This was slightly longer than normal mobility (TUG <10 seconds),20 but patients had relatively low levels of pain (mean NRS = 2.5, on a scale of 0-11).
All patients completed the PT protocols without indication of injury or AEs related to their operative knee. Three patients (10.3%; 3/29) experienced a deep venous thromboembolism (DVT), 2 in the control group (13.3%; 2/15), and 1 in the AlterG group (7.1%; 1/14). Venous thromboembolism protocol of enoxaparin 30 mg twice daily while in the hospital and enoxaparin 40 mg once daily for 10 days after discharge was followed for all patients.
Overall, more than half of patients (55.2%; 16/29) discontinued their AD during the 4-week PT period, with the remaining discontinuing prior to EOPT (24.1%; 7/29) or after EOPT (20.7%; 6/29). No statistically significant differences were found between the control and AlterG groups regarding discontinuance of AD.
Among those randomized to the AlterG group, all patients performed within the protocol established for the device for body-weight setting, treadmill speed, and duration of walking. The average body-weight treadmill setting increased by ~30% over the treatment period, from 55% at baseline to 84% at EOPT. The average speed increased by ~70%, from 1.6 mph at baseline to 2.7 mph at EOPT. The mean duration of AlterG use increased by ~75%, from 7.2 minutes at baseline to 12.7 minutes at EOPT. All physical therapists (100%) reported satisfaction with the AlterG for use in early postoperative rehabilitation and reported that patients’ treatment progressed positively.
While functional outcomes (KOOS, TUG, or NRS) did not vary with the type of gait training (P > .2 for land-based vs anti-gravity), functional outcomes improved over time (all P < .01 from baseline to EOPT and all P < .01 from baseline to EOS).
The KOOS scores improved from baseline to EOPT and from baseline to EOS (ie, 3-month follow-up visit) for both treatment groups (Figure 3). More patients completed the sports/recreation portion of the KOOS scores at EOPT and EOS compared to baseline. Forty-three percent and 25% of patients did not complete KOOS sports/recreation questions at EOPT and EOS, respectively, compared to 50% at baseline. This suggests that patients were improving to a level where sports/recreation scores were more applicable than directly after TKA surgery. The TUG scores had the greatest improvement from baseline to EOPT, with a decrease in time of 5 seconds and 7 seconds for the control and AlterG groups, respectively, and slight improvement from EOPT to EOS, with a decrease in time of 1 second and 2 seconds for the control and AlterG groups, respectively (Table 2). By the EOS, the values for the TUG tests for both treatment groups were within normal (<10 seconds) range.20 The NRS scores improved from baseline to EOPT with a score of 1 ± 1 in both control and AlterG groups.
Using these preliminary efficacy results, a post-hoc power analysis (α = .05 and 1β = 80%) was performed with the ADL domain of KOOS as the primary endpoint. Based on a standard deviation of 20 points and an effect size of 5 points, the sample size was estimated to be N = 250 per treatment group.
Discussion
We conducted a pilot study to assess, primarily, the feasibility and safety, and, secondarily, the efficacy, of a lower body positive-pressure treadmill for rehabilitation of patients after TKA. This small study showed that use of the AlterG Anti-Gravity Treadmill was not only safe and feasible during postoperative TKA rehabilitation, but also was well tolerated by patients and was rated highly satisfactory by physical therapists. Patients who used AlterG during gait training improved functionally (in terms of KOOS, TUG, and NRS) after 8 treatment sessions compared to baseline. However, there were no statistical differences between groups (control vs AlterG). Thus, these results suggest that an anti-gravity device for gait training may be a useful adjunct for postoperative TKA rehabilitation, but further studies are needed to determine the efficacy of anti-gravity compared to traditional land-based gait training.
The study of rehabilitation protocols during postoperative PT involved consideration of a number of issues. First, differences in functional outcomes compared to traditional rehabilitation could not be detected in this study because of the small number of patients, but the patients treated with anti-gravity gait training showed improvement in functional outcomes over time and did not report any added complications. Given that the primary outcome of this study was safety and feasibility, these added efficacy results are supplemental and useful in helping to plan studies. Second, the functional outcomes used to measure the efficacy of the anti-gravity treadmill may not be sensitive enough to detect differences between rehabilitation protocols. Use of a treadmill to measure speed improvement, endurance, and tolerance in both groups could be valuable in future studies. More studies may need to refine characteristics that are important to postoperative rehabilitation success, and quantitative and subjective measures that must be defined.
The results reported here using an anti-gravity treadmill for postoperative TKA rehabilitation support the safety and feasibility that has been reported in other orthopedic rehabilitation settings. Anti-gravity treadmills, which have been used to study patients after meniscectomy or anterior cruciate ligament reconstruction8 and Achilles repair,17 have demonstrated predictable decreases in ground reaction forces with increasing positive-pressure unweighting, reductions in pain with ambulation, and allowance of earlier institution of walking and jogging during rehabilitation.17
Patient safety is an important attribute for any postoperative rehabilitation protocol, especially in an elderly population undergoing major surgery. One of our important goals was to assess the safety of AlterG. We noted no AEs attributable to the device, which was supported by work indicating no adverse impact on systemic cardiovascular parameters in a similar lower body positive-pressure environment.9 Although 3 patients (10%) developed symptomatic DVT, there were no differences between the groups in the incidence of DVT. Use of an anti-gravity treadmill has also been examined for cardiovascular responses in TKA patients. In a study of 24 adults with TKA, researchers found that anti-gravity support allowed TKA patients to walk at faster speeds and tolerate greater inclines with lower heart rate, blood pressure, and oxygen consumption.21 With respect to efficacy of the rehabilitation intervention, we demonstrated significant improvements in all functional outcomes in both groups but no differences between the study groups. We concluded that AlterG was at least as effective as standard therapy in this small cohort. TKA is a very successful procedure, and the improvement in pain and function after surgery is fairly dramatic in most patients, regardless of specific rehabilitation protocols. Therefore, the substantial improvement in clinical outcomes may overshadow any enhanced benefits of the anti-gravity treadmill. Further investigations into the efficacy of AlterG are needed in a larger cohort to determine if this type of treatment is more beneficial than traditional land-based gait training.
Standard scoring systems such as KOOS, TUG, and NRS may not be sensitive enough to detect differences between treatment groups with small sample sizes. Given the results of the post hoc power analysis, a large number of patients (N = 250/group) would be necessary to detect any potential difference in clinical outcomes between the 2 groups. Larger studies are required to answer relevant questions, and additional outcome measures may be needed to detect differences between treatment groups. Relevant questions include whether earlier institution of the anti-gravity device during the immediate TKA postoperative period would be beneficial compared to standard postoperative PT, and whether PT enhanced with the anti-gravity device has incremental benefit in functional outcomes and in time to reach those goals. Finally, given the present attention to healthcare expenses, a cost-benefit analysis of anti-gravity device treatment vs traditional PT would be useful. Once the patient has become familiar with the function of an anti-gravity treadmill, gait therapy could proceed without the direct intervention of the therapist, potentially improving efficient delivery of rehabilitation services.
Studying the effect of different postoperative rehabilitation protocols after orthopedic surgeries can be challenging. In a large (N > 350) randomized controlled trial to study the effect of ergometer cycling after hip and knee replacement, patients who used the cycle ergometer had a higher Western Ontario and McMaster Universities Arthritis Index and greater satisfaction than those who did not after hip arthroplasty, but not after TKA.22 Improvements in muscular coordination and proprioception with the cycle ergometer may have been offset by increases in edema, joint effusion, and pain from the loading of the joint and the relatively fast rate of cycling compared to passive motion or ambulation. While many therapists and surgeons advocate cycling for rehabilitation after knee surgery, the need remains for a better definition of an optimal TKA rehabilitation program. A study of 82 patients comparing early progressive strength training to no early strength training showed no difference in the 6-minute walk test at 8 weeks.23 A systematic review of progressive resistance training (PRT) found that although postoperative PRT is safe and feasible, the methodological quality of existing studies is too low to allow conclusions regarding its efficacy.24 Gait training in an environment where weight-bearing loads can be closely controlled, monitored, and individualized may be an ideal methodology to enhance rehabilitation and return to function for knee replacement surgery.
This current study showed that the use of AlterG as an adjunct for postoperative rehabilitation is safe, accepted by patients and therapists, and leads to clinical functional outcomes that are at least as good as traditional postoperative TKA rehabilitation. We conclude that AlterG demonstrates utility and a potential for innovation in TKA rehabilitation.
1. NIH Consensus Statement on total knee arthroplasty. NIH Consensus State Sci Statements. 2003;20(1):1-34.
2. Jones CA, Voaklander DC, Suarez-Almazor ME. Determinants of function after total knee arthroplasty. Phys Ther. 2003;83(8):696-706.
3. Pearse EO, Caldwell BF, Lockwood RJ, Hollard J. Early mobilisation after conventional knee replacement may reduce the risk of post-operative venous thromboembolism. J Bone Joint Surg Br. 2007;89(3):316-322.
4. Westby MD, Kennedy D, Jones D, Jones A, Doyle-Waters MM, Backman C. Post-acute physiotherapy for primary total knee arthroplasty. Cochrane Database Syst Rev. 2008. doi.10.1002/14651858.CD007099
5. Bade MJ, Stevens-Lapsley JE. Early high-intensity rehabilitation following total knee arthroplasty improves outcomes. J Orthop Sports Phys Ther. 2011;41(12):932-941.
6. Ivanenko YP, Grasso R, Macellari V, Lacquaniti F. Control of foot trajectory in human locomotion: role of ground contact forces in simulated reduced gravity. J Neurophysiol. 2002;87(6):3070-3089.
7. Pöyhönen T, Keskinen KL, Kyröläinen H, Hautala A, Savolainen J, Mälkiä E. Neuromuscular function during therapeutic knee exercise under water and on dry land. Arch Phys Med Rehabil. 2001;82(10):1446-1452.
8. Eastlack RK, Hargens AR, Groppo ER, Steinbach GC, White KK, Pedowitz RA. Lower body positive-pressure exercise after knee surgery. Clin Orthop Rel Res. 2005;431:213-219.
9. Cutuk A, Groppo ER, Quigley EJ, White KW, Pedowitz RA, Hargens AR. Ambulation in simulated fractional gravity using lower body positive pressure: cardiovascular safety and gait analyses. J Appl Physiol. 2006;101(3):771-777.
10. Gojanovic B, Cutti P, Shultz R, Matheson GO. Maximal physiological parameters during partial body-weight support treadmill testing. Med Sci Sports Exerc. 2012;44(10):1935-1941.
11. Figueroa MA, Manning J, Escamilla P. Physiological responses to the AlterG Anti-Gravity Treadmill. Int J Applied Sci Tech. 2011;1:92-97.
12. Hoffman MD, Donaghe HE. Physiological responses to body weight-supported treadmill exercise in healthy adults. Arch Phys Med Rehabil. 2011;92(6):960-966.
13. Kurz MJ, Corr B, Stuberg W, Volkman KG, Smith N. Evaluation of lower body positive pressure supported treadmill training for children with cerebral palsy. Pediatr Phys Ther. 2011;23(3):232-239.
14. Christian M. Managing knee osteoarthritis: the effects of anti-gravity treadmill exercise on joint pain and physical function. Available at: http://mspace.lib.umanitoba.ca/handle/1993/8580. Accessed March 31, 2016.
15. Wilk KE, Macrina LC, Reinhold MM. Rehabilitation following microfracture of the knee. Cartilage. 2010;1(2):96-107.
16. Patil SS, Branovacki G, Martin MR, Pulido PA, Levy YD, Colwell CW Jr. 14-year median follow-up using the press-fit condylar sigma design for total knee arthroplasty. J Arthroplasty. 2013;28(8):1286-1290.
17. Saxena A, Granot A. Use of an anti-gravity treadmill in the rehabilitation of the operated achilles tendon: a pilot study. J Foot Ankle Surg. 2011;50(5):558-561.
18. Roos EM, Roos HP, Ekdahl C, Lohmander LS. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation of a Swedish version. Scand J Med Sci Sports. 1998;8(6):439-448.
19. Roos EM, Toksvig-Larsen S. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation and comparison to the WOMAC in total knee replacement. Health Qual Life Outcomes. 2003;1:17.
20. Timed Up and Go (TUG). Available at: http://www.rheumatology.org/I-Am-A/Rheumatologist/Research/Clinician-Researchers/Timed-Up-Go-TUG Accessed: March 15, 2016.
21. Webber SC, Horvey KJ, Yurach Pikaluk MT, Butcher SJ. Cardiovascular responses in older adults with total knee arthroplasty at rest and with exercise on a positive pressure treadmill. Eur J Appl Physiol. 2014;114(3):653-662.
22. Liebs TR, Herzberg W, Ruther W, Haasters J, Russlies M, Hassenpflug J. Ergometer cycling after hip and knee replacement surgery: a randomized control trial. J Bone Joint Surg Am. 2010;92(4):814-822.
23. Jakobsen TL, Kehlet H, Husted H, Petersen J, Bandholm T. Early progressive strength training to enhance recovery after fast-track total knee arthroplasty: a randomized controlled trial. Arthritis Care Res. 2014;66(12):1856-1866.
24. Skoffer B, Dalgas U, Mechlenburg I. Progressive resistance training before and after total hip and knee arthroplasty: a systematic review. Clin Rehabil. 2015;29(1):14-29.
1. NIH Consensus Statement on total knee arthroplasty. NIH Consensus State Sci Statements. 2003;20(1):1-34.
2. Jones CA, Voaklander DC, Suarez-Almazor ME. Determinants of function after total knee arthroplasty. Phys Ther. 2003;83(8):696-706.
3. Pearse EO, Caldwell BF, Lockwood RJ, Hollard J. Early mobilisation after conventional knee replacement may reduce the risk of post-operative venous thromboembolism. J Bone Joint Surg Br. 2007;89(3):316-322.
4. Westby MD, Kennedy D, Jones D, Jones A, Doyle-Waters MM, Backman C. Post-acute physiotherapy for primary total knee arthroplasty. Cochrane Database Syst Rev. 2008. doi.10.1002/14651858.CD007099
5. Bade MJ, Stevens-Lapsley JE. Early high-intensity rehabilitation following total knee arthroplasty improves outcomes. J Orthop Sports Phys Ther. 2011;41(12):932-941.
6. Ivanenko YP, Grasso R, Macellari V, Lacquaniti F. Control of foot trajectory in human locomotion: role of ground contact forces in simulated reduced gravity. J Neurophysiol. 2002;87(6):3070-3089.
7. Pöyhönen T, Keskinen KL, Kyröläinen H, Hautala A, Savolainen J, Mälkiä E. Neuromuscular function during therapeutic knee exercise under water and on dry land. Arch Phys Med Rehabil. 2001;82(10):1446-1452.
8. Eastlack RK, Hargens AR, Groppo ER, Steinbach GC, White KK, Pedowitz RA. Lower body positive-pressure exercise after knee surgery. Clin Orthop Rel Res. 2005;431:213-219.
9. Cutuk A, Groppo ER, Quigley EJ, White KW, Pedowitz RA, Hargens AR. Ambulation in simulated fractional gravity using lower body positive pressure: cardiovascular safety and gait analyses. J Appl Physiol. 2006;101(3):771-777.
10. Gojanovic B, Cutti P, Shultz R, Matheson GO. Maximal physiological parameters during partial body-weight support treadmill testing. Med Sci Sports Exerc. 2012;44(10):1935-1941.
11. Figueroa MA, Manning J, Escamilla P. Physiological responses to the AlterG Anti-Gravity Treadmill. Int J Applied Sci Tech. 2011;1:92-97.
12. Hoffman MD, Donaghe HE. Physiological responses to body weight-supported treadmill exercise in healthy adults. Arch Phys Med Rehabil. 2011;92(6):960-966.
13. Kurz MJ, Corr B, Stuberg W, Volkman KG, Smith N. Evaluation of lower body positive pressure supported treadmill training for children with cerebral palsy. Pediatr Phys Ther. 2011;23(3):232-239.
14. Christian M. Managing knee osteoarthritis: the effects of anti-gravity treadmill exercise on joint pain and physical function. Available at: http://mspace.lib.umanitoba.ca/handle/1993/8580. Accessed March 31, 2016.
15. Wilk KE, Macrina LC, Reinhold MM. Rehabilitation following microfracture of the knee. Cartilage. 2010;1(2):96-107.
16. Patil SS, Branovacki G, Martin MR, Pulido PA, Levy YD, Colwell CW Jr. 14-year median follow-up using the press-fit condylar sigma design for total knee arthroplasty. J Arthroplasty. 2013;28(8):1286-1290.
17. Saxena A, Granot A. Use of an anti-gravity treadmill in the rehabilitation of the operated achilles tendon: a pilot study. J Foot Ankle Surg. 2011;50(5):558-561.
18. Roos EM, Roos HP, Ekdahl C, Lohmander LS. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation of a Swedish version. Scand J Med Sci Sports. 1998;8(6):439-448.
19. Roos EM, Toksvig-Larsen S. Knee injury and Osteoarthritis Outcome Score (KOOS) - validation and comparison to the WOMAC in total knee replacement. Health Qual Life Outcomes. 2003;1:17.
20. Timed Up and Go (TUG). Available at: http://www.rheumatology.org/I-Am-A/Rheumatologist/Research/Clinician-Researchers/Timed-Up-Go-TUG Accessed: March 15, 2016.
21. Webber SC, Horvey KJ, Yurach Pikaluk MT, Butcher SJ. Cardiovascular responses in older adults with total knee arthroplasty at rest and with exercise on a positive pressure treadmill. Eur J Appl Physiol. 2014;114(3):653-662.
22. Liebs TR, Herzberg W, Ruther W, Haasters J, Russlies M, Hassenpflug J. Ergometer cycling after hip and knee replacement surgery: a randomized control trial. J Bone Joint Surg Am. 2010;92(4):814-822.
23. Jakobsen TL, Kehlet H, Husted H, Petersen J, Bandholm T. Early progressive strength training to enhance recovery after fast-track total knee arthroplasty: a randomized controlled trial. Arthritis Care Res. 2014;66(12):1856-1866.
24. Skoffer B, Dalgas U, Mechlenburg I. Progressive resistance training before and after total hip and knee arthroplasty: a systematic review. Clin Rehabil. 2015;29(1):14-29.
The Cruciate Ligaments in Total Knee Arthroplasty
Hinge knee arthroplasty was introduced in the 1950s.1 All 4 major ligaments were replaced by the hinge, which provided stabilization while allowing sagittal plane motion. Its goal was stability, not replication of normal kinematics. The addition of methyl methacrylate cement improved fixation and allowed surface design modifications that addressed normal articular motion. Implants such as the Gunston Polycentric,2 the Duocondylar,3 and the Geometric4 resurfaced the medial and lateral compartments of the knee while preserving the cruciate ligaments. The implants were subject to greater translational forces without the hinge and loosening became a major problem despite the advances in cementing. It became evident in the 1970s that preservation of the cruciates complicated the procedure. Cruciate resection simplified the operation and allowed improved fixation. The ICLH prosthesis resected the cruciates and used the articular surface design to give stability to the knee.5,6 The total condylar prosthesis had a “tibial” imminence that mimicked the shape of the tibial surface but also sacrificed both of the cruciate ligaments (Figure 1).
Designers recognized that the cruciate ligaments affected knee kinematics; however, they elected to sacrifice the anterior cruciate ligament (ACL) for surgical simplicity and implant longevity.6 In the early 1980s, both the cruciate-retaining (CR) total knee arthroplasty (TKA) (Figure 2) and posterior-stabilized (PS) TKA (Figure 3) designs addressed the posterior cruciate ligament (PCL) function. The PCL was preserved in the “cruciate-retaining” TKA, substituted in the “posterior-stabilized” TKA using a cam-post mechanism. The CR TKA designers believed that PCL preservation produced a more balanced knee with a more anatomical result, a more normal joint line, and better function, especially on stair climbing. The PS TKA designers admitted the value of posterior stabilization but argued that it was too difficult to consistently save the PCL in all cases, and that the PS knee was easier for surgeons to implant with more reliable roll back.7
The Geometric knee was developed in the 1970s to retain both cruciate ligaments.4 Unfortunately, it created a kinematic conflict by using a constrained articular surface design that prevented the motion required by the cruciate ligaments. This conflict resulted in tibial loosening and early failures. The compromised results decreased interest in the bicruciate-retaining (BCR) TKA designs, allowing the CR TKA and PS TKA designs to flourish for the next 20 years with little or no attempts to retain the ACL.
In the 1980s the BCR TKA design was pursued by Townley8 and Cartier.9 Townley8 believed that cruciate resection was a concession to “improper joint synchronization”8 and Cartier9 thought that cruciate preservation permitted more normal proprioception.9 Unlike prior BCR TKA designs, the mid-term clinical results were equal to or better than the standard CR TKA or PS TKA of the time, and 9- to 11-year follow-up demonstrated comparable outcomes.8 While these results highlighted the possibility of a BCR TKA, the surgical technique and failures of the Geometric knee discouraged surgeons from pursuing the BCR TKA.
Interest in cruciate-preserving knee arthroplasty returned with partial knee replacements, with patients reporting more normal proprioception and motion.10 The techniques became more popular with the introduction of the minimally invasive surgeries in the early 2000s and cruciate ligament preservation became a more interesting concept.11,12 Some surgeons preserved the cruciates by using separate implants for the medial, lateral, and patellofemoral surfaces.10 These results were acceptable for the time but required considerable surgical talent and did not report 20-year results similar to the CR and PS knees.
Most prosthetic designs attempt to copy the normal knee anatomy. Using fluoroscopic studies and computer analysis, designers began to investigate the motion (or kinematics) of the normal knee and realized that despite the fact the TKA looked like the human knee, the designs were not kinematically correct.13
Although TKA successfully treats pain secondary to degenerative joint disease, many patients are unable to return to their prior level of function, with up to 20% reporting dissatisfaction with their level of activity.14 The observed differences in kinematics between a normal knee and a TKA may explain part of this discrepancy.
Normal Knee Motion
The tibiofemoral articulation in a normal knee follows a reproducible pattern of motion as the knee moves from extension to flexion. The lateral femoral condyle (LFC) translates posteriorly with a combination of rolling and sliding motion, while the medial femoral condyle (MFC) has minimal posterior translation and thus acts as a pivot for knee motion. The MFC is larger, less curved, and has a biphasic shape with 2 distinct radiuses of curvature that correspond to an “extension” and “flexion” facet. The transition between the MFC facets occurs at approximately 30° of flexion, whereby the contact point transfers posteriorly with little condylar translation.15-17 In contrast, the LFC is smaller, has a single radius of curvature, and gradually translates posteriorly throughout flexion. Static magnetic resonance imaging of the knee from 0° to 120° shows an average of 19 mm posterior translation for the LFC and 2 mm for the MFC.15-20
In deep flexion, beyond 130°, posterior translation continues for both condyles. The LFC experiences enough excursion to cause loss of joint congruity and partial posterior subluxation.19,20 The MFC shows little additional posterior translation, yet it too loses joint congruity through condylar lift-off. Contact between the posterior horn of the medial meniscus and the posterior femoral condyle limits further flexion.16,21
The difference in motion between the condyles leads to internal tibial rotation during flexion. The initial 10° of knee flexion produces 5° of internal rotation, and an additional 15° of internal tibial rotation occurs throughout the remainder of knee flexion.
Fluoroscopic imaging with computed tomography (CT)- or magnetic resonance (MR)-based modeling has shown the dynamic in vivo relationship of the tibiofemoral joint. Studies have confirmed significantly greater LFC posterior translation as compared to the MFC;22 however, in vivo studies have also shown notable variability in articular rotation and translation based on activity. This highlights the role of ligamentous tension and muscle contraction in kinematics.21-23
The ACL in TKA
The majority of current TKA designs sacrifice the ACL without substituting for its function. The loss of the ACL has significant effects upon the kinematics of the knee.
The ACL is composed of 2 bundles, the anteromedial and posterolateral bundles, which originate on the LFC and insert broadly onto the tibial intercondylar eminence. Its primary role is to resist anterior tibial translation, particularly from 0° to 30° of flexion, which corresponds to the peak quadriceps force that pulls the tibia anteriorly.24 ACL deficiency causes anterior tibial translation during early flexion and abnormal internal tibial rotation.25-27 ACL deficient knees demonstrate a posterior femoral position in full extension, and increased MFC translation during knee flexion.28-32
The role of the ACL in knee arthroplasty has been evaluated by comparing unicompartmental knee arthroplasty (UKA) with TKA, as a reflection of ACL preserving vs sacrificing procedures.33-35 Sagittal plane translation is similar between UKA and normal knees,33,34 while the CR TKA and PS TKA designs show anterior tibia subluxation in full extension.33-35 The difference between UKA and TKA is greatest in extension, corresponding to the ACL functional range. These findings highlight kinematic similarities between TKA designs and the ACL deficient knee.
The majority of UKAs demonstrate near-normal kinematics. A small percentage of the study group demonstrated aberrant anterior tibial motion, highlighting a concern over ACL attenuation with time. Additionally, studies that evaluate the ACL in osteoarthritic knees have questioned the baseline integrity of the ACL.36 Yet the long-term outcomes in UKA design have shown preservation of kinematics due to intact cruciates.37
The PCL in TKA
Because the majority of TKA designs sacrifice the ACL, the classic debate has focused on the utility of the native PCL. Both the CR and PS TKA are designed to offer posterior stabilization; however, kinematic studies have demonstrated notable differences.38,39
The CR TKA design relies on the PCL to resist posterior sag and to prevent the hamstring musculature from pulling the tibia posteriorly during flexion. Studies have shown paradoxical anterior translation of both femoral condyles during flexion, particularly on the medial side of the knee.40 There is also increased variability in femoral rollback. It is unclear whether the PCL can function normally in the absence of the ACL, which causes the PCL to adapt a less anatomic vertical position. The PCL may also be unable to function significantly without the ACL because of pre-existing degenerative histological changes.41
The PS TKA utilizes a cam-post mechanism for posterior stabilization. In contrast to normal knee kinematics, this mechanism creates equal MFC and LFC posterior translation, 8 mm on average at 90° flexion.40 The equivalent translation in PS designs contributes to decreased internal tibial rotation and an increased polyethylene wear at the post.
Role of Surface Geometry
The articular geometry of the knee plays an important role in normal knee kinematics. Initial TKA designs used a femoral component with a single radius of curvature for both femoral condyles.42Current TKA designs that match the femoral component to the native femoral anatomy, by differing the medial and lateral condyle geometry, have demonstrated kinematics that better resemble a native knee.43 Additional changes to the radius of curvature along the posterior facet of the femoral condyles may reduce impingement during deep flexion. These “high flex” designs have demonstrated equivalent range of motion in some studies44 and improved weight-bearing motion in others.45 Surface geometry is important but is not the entire answer to kinematics.
Advances in TKA Design
Knee motion is guided by multiple factors, including the tibiofemoral articular geometry, the surrounding soft tissue tension, and muscle tone. Bicruciate-substituting (BCS) TKA and BCR TKA are forms of evolution from the CR and PS TKA and attempt to respect the function of both cruciate ligaments and provide better kinematics.
The BCS TKA utilizes a modified cam-post articulation to provide both anterior and posterior stabilization (Figure 4).46 The surgical approach remains the same and the implant geometry affects the motion. The BCS TKA design demonstrates femoral rollback at 90° with an average of 14 mm for the MFC and 23 mm for the LFC, and 10° internal tibial rotation.46,47 Additionally, it provides increased sagittal stability during early flexion and an improved pivot shift (indicating improved anterior stabilization).
The BCR designs preserve both cruciates and provide anterior and posterior stabilization. Fluoroscopic imaging has demonstrated contact points in full extension, and posterior rollback at 90° flexion that more closely replicates the normal knee.48
Design and Surgical Techniques for Bicruciate Knee Replacements
If all of the ligaments are preserved, the TKA surfaces must allow motion to be driven by the ligaments in combination with the surfaces alone. The femur can be designed anatomically with asymmetric condyles. The femoral box must allow for preservation of the tibial bone island without impinging upon the cruciate ligaments. The tibial surface must be minimally constrained with concavity medially and convexity laterally.
The bone island preservation does not permit a single-piece tibial polyethylene insert. Therefore, the inserts will replicate the UKA designs (Figure 5). The knee should allow greater range of motion with the possibility of heel to buttocks contact. This increased motion will lead to greater roll back of the femur on the tibia and can lead to subluxation of the femoral runner off of the tibial surface on the lateral side, mimicking the normal knee. This subluxation is desirable but may lead to increased wear of the polyethylene on the lateral side of the knee.
The instruments should be specific for the design but must also be user-friendly. The 2 major issues with the surgery are balancing the knee in full extension and flexion, and preservation of the tibial bone island. The preexisting knee deformity should be <10° in all planes to limit the amount of collateral ligament releases. The collaterals must be balanced in a similar fashion to the standard TKA. Flexion contracture can be treated with posterior capsular release around the cruciates or with an increased distal femoral resection (2 mm at the maximum).
It is important to size the femur correctly because it will be difficult to adjust the flexion gap on the tibial side. A 9-mm posterior medial femoral condyle resection is a reasonable guide if the condyle is not atrophic. However, the exact resection thickness will be implant-specific and should be correlated with the dimensions of the prosthesis being implanted. The tibial bone island must be properly rotated with respect to the center line (Akagi’s line)49 and must not be undercut. The tibial instrument should include pins or blocks to prevent the sawblades from undercutting the island (Figure 6), as undermining leads to fracture in full extension. If undermining occurs, it may be possible to place a cancellous screw through the island and still preserve the ligaments. The integrity of the island is best tested by bringing the knee to full extension and checking for liftoff of the bone. If there is significant compromise of the island, the bone should be resected and either a CR or PS TKA can be implanted. Della Valle and colleagues50 reported a 9.2% incidence (11 of 119 cases) of bone island fracture in their early experience with a BCR TKA and improved this to 1.9% (5/258 cases) after reassessing their technique.
The gap tension should be evaluated either with traditional spacer blocks or with tensioning devices on the medial and lateral side of the knee after the tibial resections are completed. The polyethylene inserts are anatomically different. It may be possible to vary the thickness from medial to lateral, but not in excess of 2 mm.
As the BCR surgical techniques evolve, the balancing and tibial resection may be refined through specialized instrumentation. Such “smart instruments” that incorporate gyros may expedite tibial alignment, and sensor devices may assist with gap balancing. Haptic surgical robotic guides may assist in the tibial resection, facilitating bone island preservation by avoiding any possibility of undermining. At present these assistive aides are not necessary for the operation but may play a future role.
Clinical Results of Knee Arthroplasties
The results of knee replacements improved steadily from the 1970s through the 1990s. The scoring systems were somewhat limited and there was little data on the perception of the patients. The prosthetic designs stabilized at the end of the 1990s with only minor modifications since the year 2000. The 20-year results show similar findings for both the CR and the PS designs. There is little evidence to suggest a clinical correlation with the observed kinematic differences between CR and PS TKA designs.40,51-58 Multiple studies have demonstrated equivalent range of motion38,39,59 and subjective outcome measures (Table 1).60 A randomized prospective trial that compared kinematics and functional scores between the 2 designs failed to observe significant differences in function despite differences in kinematics.46 Equivalence in clinical outcome was further supported by a Cochrane Review meta-analysis that evaluated 1810 patients in 17 selected studies.61 The Knee Society scores have all been in the 92% to 95% ratings with survivals between 90% and 95%.
However, only 80% to 90% of patients are fully satisfied with their implants. The reasons for the dissatisfaction include unexplained anterior knee pain, stiffness, unexplained swelling, loss of range of motion, changes in proprioception, and loss of preoperative functions.14
The mid-term results of the BCR knees that were performed in the 1980s showed similar results to the CR and PS knees. Townley8 reported excellent clinical results with only 2% loosening at 2 to 11 years after surgery. Cloutier and colleagues9 reported 95% survival with improved proprioception at 9 to 11 years after surgery(Table 2).62,63
Studies comparing traditional TKA designs with cruciate preserving designs, both UKA and BCR, have found differences in subjective outcomes.62,64 Comparison of UKA and TKA in the same patient demonstrated significant preference for UKA, particularly with stair-climbing.65 Similarly, comparison between BCR and PS TKA or CR TKA demonstrated preference for BCR in 85% of patients.62
The new BCR knee designs have just started to come to the market.50 The surgical techniques are much improved over the 1980s and cruciate preservation is certainly much easier now. The new designs can produce full range of motion with kinematics that are almost identical to the normal knee in the cadaver laboratory and in computer analyses. These designs certainly should have a similar 20-year survival to the original BCR knees. However, the critical evaluation will be the patient satisfaction scores. With greater motion, better kinematics, and more precise balancing the scores would improve with these designs.
Conclusion
The cruciate ligaments of the knee are central to control of the motion of the normal knee. TKA is a successful operation with at least a 40- to 50-year history. The techniques have continued to develop but 15% to 20% of patients are dissatisfied with the results.14 Evaluations of the prostheses are more sophisticated and kinematics appears to have a central position in the evaluation. If the knee is to move more anatomically correctly, all of the ligaments must be preserved. Proprioception certainly plays a role in the patient’s judgment of the result. History has shown that a BCR knee can be implanted with good mid-term results and it should certainly be possible to build on these results and design a knee that will incorporate all of the ligaments with full range of motion and increased levels of activity.
1. Walldius B. Arthroplasty of the knee with an endoprosthesis. Acta Chir Scand. 1957;113(6):445-446.
2. Gunston FH. Polycentric knee arthroplasty. Prosthetic simulation of normal knee movement. J Bone Joint Surg Br. 1971;53(2):272-277.
3. Insall JN, Ranawat CS, Aglietti P, Shine J. A comparison of four models of total knee-replacement prostheses. J Bone Joint Surg Am. 1976;58(6):754-765.
4. Coventry MB, Finerman GA, Riley LH, Turner RH, Upshaw JE. A new geometric knee for total knee arthroplasty. Clin Orthop Relat Res.1972;83:157-162.
5. Freeman MA, Sculco T, Todd RC. Replacement of the severely damaged arthritic knee by the ICLH (Freeman-Swanson) arthroplasty. J Bone Joint Surg Br. 1977;59(1):64-71.
6. Freeman MA, Insall JN, Besser W, Walker PS, Hallel T. Excision of the cruciate ligaments in total knee replacement. Clin Orthop Relat Res. 1977(126):209-212.
7. Pagnano MW, Cushner FD, Scott WN. Role of the posterior cruciate ligament in total knee arthroplasty. J Am Acad Orthop Surg. 1998;6(3):176-187.
8. Townley CO. The anatomic total knee resurfacing arthroplasty. Clin Orthop Relat Res. 1985(192):82-96.
9. Cloutier JM, Sabouret P, Deghrar A. Total knee arthroplasty with retention of both cruciate ligaments. A nine to eleven-year follow-up study. J Bone Joint Surg Am. 1999; 81(5):697-702.
10. Banks SA, Fregly BJ, Boniforti F, Reinschmidt C, Romagnoli S. Comparing in vivo kinematics of unicondylar and bi-unicondylar knee replacements. Knee Surg Sports Traumatol Arthrosc. 2005;13(7):551-556.
11. Repicci JA, Eberle RW. Minimally invasive surgical technique for unicondylar knee arthroplasty. J South Orthop Assoc. 1999;8(1):20-27; discussion 27.
12. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
13. Banks SA, Markovich GD, Hodge WA. In vivo kinematics of cruciate-retaining and -substituting knee arthroplasties. J Arthroplasty. 1997;12(3):297-304.
14. Nam D, Nunley RM, Barrack RL. Patient dissatisfaction following total knee replacement: a growing concern? Bone Joint J. 2014;96-B(11 Supple A):96-100.
15. Iwaki H, Pinskerova V, Freeman MA. Tibiofemoral movement 1: the shapes and relative movements of the femur and tibia in the unloaded cadaver knee. J Bone Joint Surg Br. 2000;82(8):1189-1195.
16. Johal P, Williams A, Wragg P, Hunt D, Gedroyc W. Tibio-femoral movement in the living knee. A study of weight bearing and non-weight bearing knee kinematics using ‘interventional’ MRI. J Biomech. 2005;38(2):269-276.
17. Pinskerova V, Johal P, Nakagawa S, et al. Does the femur roll-back with flexion? J Bone Joint Surg Br. 2004;86(6):925-931.
18. Hill PF, Vedi V, Williams A, Pinskerova V, Freeman MA. Tibiofemoral movement 2: the loaded and unloaded living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1196-1198.
19. Nakagawa S, Kadoya Y, Todo S, et al. Tibiofemoral movement 3: full flexion in the living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1199-1200.
20. Freeman MA, Pinskerova V. The movement of the knee studied by magnetic resonance imaging. Clin Orthop Relat Res. 2003(410):35-43.
21. Moro-oka TA, Hamai S, Miura H, et al. Dynamic activity dependence of in vivo normal knee kinematics. J Orthop Res. 2008;26(4):428-434.
22. Komistek RD, Dennis DA, Mahfouz M. In vivo fluoroscopic analysis of the normal human knee. Clin Orthop Relat Res. 2003(410):69-81.
23. Li G, DeFrate LE, Park SE, Gill TJ, Rubash HE. In vivo articular cartilage contact kinematics of the knee: an investigation using dual-orthogonal fluoroscopy and magnetic resonance image-based computer models. Am J Sports Med. 2005;33(1):102-107.
24. Grood ES, Suntay WJ, Noyes FR, Butler DL. Biomechanics of the knee-extension exercise. Effect of cutting the anterior cruciate ligament. J Bone Joint Surg Am. 1984;66(5):725-734.
25. Noyes FR, Jetter AW, Grood ES, Harms SP, Gardner EJ, Levy MS. Anterior cruciate ligament function in providing rotational stability assessed by medial and lateral tibiofemoral compartment translations and subluxations. Am J Sports Med. 2015;43(3):683-692.
26. Good L, Askew MJ, Boom A, Melby A 3rd. Kinematic in-vitro comparison between the normal knee and two techniques for reconstruction of the anterior cruciate ligament. Clin Biomech (Bristol, Avon). 1993;8(5):243-249.
27. Beard DJ, Murray DW, Gill HS. Reconstruction does not reduce tibial translation in the cruciate-deficient knee an in vivo study. J Bone Joint Surg Br. 2001;83(8):1098-1103.
28. Dennis DA, Mahfouz MR, Komistek RD, Hoff W. In vivo determination of normal and anterior cruciate ligament-deficient knee kinematics. J Biomech. 2005;38(2):241-253.
29. Beynnon BD, Fleming BC, Labovitch R, Parsons B. Chronic anterior cruciate ligament deficiency is associated with increased anterior translation of the tibia during the transition from non-weightbearing to weightbearing. J Orthop Res. 2002;20(2):332-337.
30. Brandsson S, Karlsson J, Eriksson BI, Kärrholm J. Kinematics after tear in the anterior cruciate ligament: dynamic bilateral radiostereometric studies in 11 patients. Acta Orthop Scand. 2001;72(4):372-378.
31. Andriacchi TP, Briant PL, Bevill SL, Koo S. Rotational changes at the knee after ACL injury cause cartilage thinning. Clin Orthop Relat Res. 2006;442:39-44.
32. Scarvell JM, Smith PN, Refshauge KM, Galloway HR, Woods KR. Comparison of kinematic analysis by mapping tibiofemoral contact with movement of the femoral condylar centres in healthy and anterior cruciate ligament injured knees. J Orthop Res. 2004;22(5):955-962.
33. Miller RK, Goodfellow JW, Murray DW, O’Connor JJ. In vitro measurement of patellofemoral force after three types of knee replacement. J Bone Joint Surg Br. 1998;80(5):900-906.
34. Price AJ, Rees JL, Beard DL, Gill RH, Dodd CA, Murray DM. Sagittal plane kinematics of a mobile-bearing unicompartmental knee arthroplasty at 10 years: a comparative in vivo fluoroscopic analysis. J Arthroplasty. 2004;19(5):590-597.
35. Dennis D, Komistek R, Scuderi G, et al. In vivo three-dimensional determination of kinematics for subjects with a normal knee or a unicompartmental or total knee replacement. J Bone Joint Surg Am. 2001;83-A Suppl 2 Pt 2:104-115.
36. Arbuthnot JE, Brink RB. Assessment of the antero-posterior and rotational stability of the anterior cruciate ligament analogue in a guided motion bi-cruciate stabilized total knee arthroplasty. J Med Eng Technol. 2009;33(8):610-615.
37. Hollinghurst D, Stoney J, Ward T, et al. No deterioration of kinematics and cruciate function 10 years after medial unicompartmental arthroplasty. Knee. 2006;13(6):440-444.
38. Dennis DA, Komistek RD, Colwell CE Jr, et al. In vivo anteroposterior femorotibial translation of total knee arthroplasty: a multicenter analysis. Clin Orthop Relat Res. 1998(356):47-57.
39. Dennis DA, Komistek RD, Hoff WA, Gabriel SM. In vivo knee kinematics derived using an inverse perspective technique. Clin Orthop Relat Res. 1996;(331):107-117.
40. Yoshiya S, Matsui N, Komistek RD, Dennis DA, Mahfouz M, Kurosaka M. In vivo kinematic comparison of posterior cruciate-retaining and posterior stabilized total knee arthroplasties under passive and weight-bearing conditions. J Arthroplasty. 2005;20(6):777-783.
41. Kleinbart FA, Bryk E, Evangelista J, Scott WN, Vigorita VJ. Histologic comparison of posterior cruciate ligaments from arthritic and age-matched knee specimens. J Arthroplasty. 1996;11(6):726-731.
42. Bull AM, Kessler O, Alam M, Amis AA. Changes in knee kinematics reflect the articular geometry after arthroplasty. Clin Orthop Relat Res. 2008;466(10):2491-2499.
43. Komistek RD, Mahfouz MR, Bertin KC, Rosenberg A, Kennedy W. In vivo determination of total knee arthroplasty kinematics: a multicenter analysis of an asymmetrical posterior cruciate retaining total knee arthroplasty. J Arthroplasty. 2008;23(1):41-50.
44. Mehin R, Burnett RS, Brasher PM. Does the new generation of high-flex knee prostheses improve the post-operative range of movement?: a meta-analysis. J Bone Joint Surg Br. 2010;92(10):1429-1434.
45. Dennis DA, Heekin RD, Clark CR, Murphy JA, O’Dell TL, Dwyer KA. Effect of implant design on knee flexion. J Arthroplasty. 2013;28(3):429-438.
46. Victor J, Mueller JK, Komistek RD, Sharma A, Nadaud MC, Bellemans J. In vivo kinematics after a cruciate-substituting TKA. Clin Orthop Relat Res. 2010;468(3):807-814.
47. Catani F, Ensini A, Belvedere C, et al. In vivo kinematics and kinetics of a bi-cruciate substituting total knee arthroplasty: a combined fluoroscopic and gait analysis study. J Orthop Res. 2009;27(12):1569-1575.
48. Stiehl JB, Komistek RD, Cloutier JM, Dennis DA. The cruciate ligaments in total knee arthroplasty: a kinematic analysis of 2 total knee arthroplasties. J Arthroplasty. 2000;15(5):545-550.
49. Akagi M, Oh M, Nonaka T, Tsujimoto H, Asano T, Hamanishi C. An anteroposterior axis of the tibia for total knee arthroplasty. Clin Orthop Relat Res. 2004;(420):213-219.
50. Della Valle CJ, Andriacchi TP, Berend KR, DeClaire JH, Lombardi AV Jr, Peters CL. Early experience with bi-cruciate retaining TKA. Poster presented at: American Academy of Orthopaedic Surgeons 2015 Annual Meeting; March 24-28, 2015; Las Vegas, NV.
51. Udomkiat P, Meng BJ, Dorr LD, Wan Z. Functional comparison of posterior cruciate retention and substitution knee replacement. Clin Orthop Relat Res. 2000;(378):192-201.
52. Tanzer M, Smith K, Burnett S. Posterior-stabilized versus cruciate-retaining total knee arthroplasty: balancing the gap. J Arthroplasty. 2002;17(7):813-819.
53. Maruyama S, Yoshiya S, Matsui N, Kuroda R, Kurosaka M. Functional comparison of posterior cruciate-retaining versus posterior stabilized total knee arthroplasty. J Arthroplasty. 2004;19(3):349-53.
54. Clark CR, Rorabeck CH, MacDonald S, MacDonald D, Swafford J, Cleland D. Posterior-stabilized and cruciate-retaining total knee replacement: a randomized study. Clin Orthop Relat Res. 2001;(392):208-212.
55. Swanik CB, Lephart SM, Rubash HE. Proprioception, kinesthesia, and balance after total knee arthroplasty with cruciate-retaining and posterior stabilized prostheses. J Bone Joint Surg Am. 2004;86-A(2):328-334.
56. Harato K, Bourne RB, Victor J, Snyder M, Hart J, Ries MD. Midterm comparison of posterior cruciate-retaining versus -substituting total knee arthroplasty using the Genesis II prosthesis. A multicenter prospective randomized clinical trial. Knee. 2008;15(3):217-221.
57. Catani F, Leardini A, Ensini A, et al. The stability of the cemented tibial component of total knee arthroplasty: posterior cruciate-retaining versus posterior-stabilized design. J Arthroplasty. 2004;19(6):775-782.
58. Dennis DA, Komistek RD, Stiehl JB, Walker SA, Dennis KN. Range of motion after total knee arthroplasty: the effect of implant design and weight-bearing conditions. J Arthroplasty. 1998;13(7):748-752.
59. Becker MW, Insall JN, Faris PM. Bilateral total knee arthroplasty. One cruciate retaining and one cruciate substituting. Clin Orthop Relat Res. 1991;(271):122-124.
60. Kim YH, Choi Y, Kwon OR, Kim JS. Functional outcome and range of motion of high-flexion posterior cruciate-retaining and high-flexion posterior cruciate-substituting total knee prostheses. A prospective, randomized study. J Bone Joint Surg Am. 2009;91(4):753-760.
61. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
62. Pritchett JW. Patients prefer a bicruciate-retaining or the medial pivot total knee prosthesis. J Arthroplasty. 2011;26(2):224-228.
63. Sabouret P, Lavoie F, Cloutier JM. Total knee replacement with retention of both cruciate ligaments: a 22-year follow-up study. Bone Joint J. 2013;95-B(7):917-922.
64. Andriacchi TP, Galante JO, Fermier RW. The influence of total knee-replacement design on walking and stair-climbing. J Bone Joint Surg Am. 1982;64(9):1328-1335.
65. Laurencin CT, Zelicof SB, Scott RD, Ewald FC. Unicompartmental versus total knee arthroplasty in the same patient. A comparative study. Clin Orthop Relat Res. 1991;(273):151-156.
66. Victor J, Banks S, Bellemans J. Kinematics of posterior cruciate ligament-retaining and -substituting total knee arthroplasty: a prospective randomised outcome study. J Bone Joint Surg Br. 2005;87(5):646-655.
Hinge knee arthroplasty was introduced in the 1950s.1 All 4 major ligaments were replaced by the hinge, which provided stabilization while allowing sagittal plane motion. Its goal was stability, not replication of normal kinematics. The addition of methyl methacrylate cement improved fixation and allowed surface design modifications that addressed normal articular motion. Implants such as the Gunston Polycentric,2 the Duocondylar,3 and the Geometric4 resurfaced the medial and lateral compartments of the knee while preserving the cruciate ligaments. The implants were subject to greater translational forces without the hinge and loosening became a major problem despite the advances in cementing. It became evident in the 1970s that preservation of the cruciates complicated the procedure. Cruciate resection simplified the operation and allowed improved fixation. The ICLH prosthesis resected the cruciates and used the articular surface design to give stability to the knee.5,6 The total condylar prosthesis had a “tibial” imminence that mimicked the shape of the tibial surface but also sacrificed both of the cruciate ligaments (Figure 1).
Designers recognized that the cruciate ligaments affected knee kinematics; however, they elected to sacrifice the anterior cruciate ligament (ACL) for surgical simplicity and implant longevity.6 In the early 1980s, both the cruciate-retaining (CR) total knee arthroplasty (TKA) (Figure 2) and posterior-stabilized (PS) TKA (Figure 3) designs addressed the posterior cruciate ligament (PCL) function. The PCL was preserved in the “cruciate-retaining” TKA, substituted in the “posterior-stabilized” TKA using a cam-post mechanism. The CR TKA designers believed that PCL preservation produced a more balanced knee with a more anatomical result, a more normal joint line, and better function, especially on stair climbing. The PS TKA designers admitted the value of posterior stabilization but argued that it was too difficult to consistently save the PCL in all cases, and that the PS knee was easier for surgeons to implant with more reliable roll back.7
The Geometric knee was developed in the 1970s to retain both cruciate ligaments.4 Unfortunately, it created a kinematic conflict by using a constrained articular surface design that prevented the motion required by the cruciate ligaments. This conflict resulted in tibial loosening and early failures. The compromised results decreased interest in the bicruciate-retaining (BCR) TKA designs, allowing the CR TKA and PS TKA designs to flourish for the next 20 years with little or no attempts to retain the ACL.
In the 1980s the BCR TKA design was pursued by Townley8 and Cartier.9 Townley8 believed that cruciate resection was a concession to “improper joint synchronization”8 and Cartier9 thought that cruciate preservation permitted more normal proprioception.9 Unlike prior BCR TKA designs, the mid-term clinical results were equal to or better than the standard CR TKA or PS TKA of the time, and 9- to 11-year follow-up demonstrated comparable outcomes.8 While these results highlighted the possibility of a BCR TKA, the surgical technique and failures of the Geometric knee discouraged surgeons from pursuing the BCR TKA.
Interest in cruciate-preserving knee arthroplasty returned with partial knee replacements, with patients reporting more normal proprioception and motion.10 The techniques became more popular with the introduction of the minimally invasive surgeries in the early 2000s and cruciate ligament preservation became a more interesting concept.11,12 Some surgeons preserved the cruciates by using separate implants for the medial, lateral, and patellofemoral surfaces.10 These results were acceptable for the time but required considerable surgical talent and did not report 20-year results similar to the CR and PS knees.
Most prosthetic designs attempt to copy the normal knee anatomy. Using fluoroscopic studies and computer analysis, designers began to investigate the motion (or kinematics) of the normal knee and realized that despite the fact the TKA looked like the human knee, the designs were not kinematically correct.13
Although TKA successfully treats pain secondary to degenerative joint disease, many patients are unable to return to their prior level of function, with up to 20% reporting dissatisfaction with their level of activity.14 The observed differences in kinematics between a normal knee and a TKA may explain part of this discrepancy.
Normal Knee Motion
The tibiofemoral articulation in a normal knee follows a reproducible pattern of motion as the knee moves from extension to flexion. The lateral femoral condyle (LFC) translates posteriorly with a combination of rolling and sliding motion, while the medial femoral condyle (MFC) has minimal posterior translation and thus acts as a pivot for knee motion. The MFC is larger, less curved, and has a biphasic shape with 2 distinct radiuses of curvature that correspond to an “extension” and “flexion” facet. The transition between the MFC facets occurs at approximately 30° of flexion, whereby the contact point transfers posteriorly with little condylar translation.15-17 In contrast, the LFC is smaller, has a single radius of curvature, and gradually translates posteriorly throughout flexion. Static magnetic resonance imaging of the knee from 0° to 120° shows an average of 19 mm posterior translation for the LFC and 2 mm for the MFC.15-20
In deep flexion, beyond 130°, posterior translation continues for both condyles. The LFC experiences enough excursion to cause loss of joint congruity and partial posterior subluxation.19,20 The MFC shows little additional posterior translation, yet it too loses joint congruity through condylar lift-off. Contact between the posterior horn of the medial meniscus and the posterior femoral condyle limits further flexion.16,21
The difference in motion between the condyles leads to internal tibial rotation during flexion. The initial 10° of knee flexion produces 5° of internal rotation, and an additional 15° of internal tibial rotation occurs throughout the remainder of knee flexion.
Fluoroscopic imaging with computed tomography (CT)- or magnetic resonance (MR)-based modeling has shown the dynamic in vivo relationship of the tibiofemoral joint. Studies have confirmed significantly greater LFC posterior translation as compared to the MFC;22 however, in vivo studies have also shown notable variability in articular rotation and translation based on activity. This highlights the role of ligamentous tension and muscle contraction in kinematics.21-23
The ACL in TKA
The majority of current TKA designs sacrifice the ACL without substituting for its function. The loss of the ACL has significant effects upon the kinematics of the knee.
The ACL is composed of 2 bundles, the anteromedial and posterolateral bundles, which originate on the LFC and insert broadly onto the tibial intercondylar eminence. Its primary role is to resist anterior tibial translation, particularly from 0° to 30° of flexion, which corresponds to the peak quadriceps force that pulls the tibia anteriorly.24 ACL deficiency causes anterior tibial translation during early flexion and abnormal internal tibial rotation.25-27 ACL deficient knees demonstrate a posterior femoral position in full extension, and increased MFC translation during knee flexion.28-32
The role of the ACL in knee arthroplasty has been evaluated by comparing unicompartmental knee arthroplasty (UKA) with TKA, as a reflection of ACL preserving vs sacrificing procedures.33-35 Sagittal plane translation is similar between UKA and normal knees,33,34 while the CR TKA and PS TKA designs show anterior tibia subluxation in full extension.33-35 The difference between UKA and TKA is greatest in extension, corresponding to the ACL functional range. These findings highlight kinematic similarities between TKA designs and the ACL deficient knee.
The majority of UKAs demonstrate near-normal kinematics. A small percentage of the study group demonstrated aberrant anterior tibial motion, highlighting a concern over ACL attenuation with time. Additionally, studies that evaluate the ACL in osteoarthritic knees have questioned the baseline integrity of the ACL.36 Yet the long-term outcomes in UKA design have shown preservation of kinematics due to intact cruciates.37
The PCL in TKA
Because the majority of TKA designs sacrifice the ACL, the classic debate has focused on the utility of the native PCL. Both the CR and PS TKA are designed to offer posterior stabilization; however, kinematic studies have demonstrated notable differences.38,39
The CR TKA design relies on the PCL to resist posterior sag and to prevent the hamstring musculature from pulling the tibia posteriorly during flexion. Studies have shown paradoxical anterior translation of both femoral condyles during flexion, particularly on the medial side of the knee.40 There is also increased variability in femoral rollback. It is unclear whether the PCL can function normally in the absence of the ACL, which causes the PCL to adapt a less anatomic vertical position. The PCL may also be unable to function significantly without the ACL because of pre-existing degenerative histological changes.41
The PS TKA utilizes a cam-post mechanism for posterior stabilization. In contrast to normal knee kinematics, this mechanism creates equal MFC and LFC posterior translation, 8 mm on average at 90° flexion.40 The equivalent translation in PS designs contributes to decreased internal tibial rotation and an increased polyethylene wear at the post.
Role of Surface Geometry
The articular geometry of the knee plays an important role in normal knee kinematics. Initial TKA designs used a femoral component with a single radius of curvature for both femoral condyles.42Current TKA designs that match the femoral component to the native femoral anatomy, by differing the medial and lateral condyle geometry, have demonstrated kinematics that better resemble a native knee.43 Additional changes to the radius of curvature along the posterior facet of the femoral condyles may reduce impingement during deep flexion. These “high flex” designs have demonstrated equivalent range of motion in some studies44 and improved weight-bearing motion in others.45 Surface geometry is important but is not the entire answer to kinematics.
Advances in TKA Design
Knee motion is guided by multiple factors, including the tibiofemoral articular geometry, the surrounding soft tissue tension, and muscle tone. Bicruciate-substituting (BCS) TKA and BCR TKA are forms of evolution from the CR and PS TKA and attempt to respect the function of both cruciate ligaments and provide better kinematics.
The BCS TKA utilizes a modified cam-post articulation to provide both anterior and posterior stabilization (Figure 4).46 The surgical approach remains the same and the implant geometry affects the motion. The BCS TKA design demonstrates femoral rollback at 90° with an average of 14 mm for the MFC and 23 mm for the LFC, and 10° internal tibial rotation.46,47 Additionally, it provides increased sagittal stability during early flexion and an improved pivot shift (indicating improved anterior stabilization).
The BCR designs preserve both cruciates and provide anterior and posterior stabilization. Fluoroscopic imaging has demonstrated contact points in full extension, and posterior rollback at 90° flexion that more closely replicates the normal knee.48
Design and Surgical Techniques for Bicruciate Knee Replacements
If all of the ligaments are preserved, the TKA surfaces must allow motion to be driven by the ligaments in combination with the surfaces alone. The femur can be designed anatomically with asymmetric condyles. The femoral box must allow for preservation of the tibial bone island without impinging upon the cruciate ligaments. The tibial surface must be minimally constrained with concavity medially and convexity laterally.
The bone island preservation does not permit a single-piece tibial polyethylene insert. Therefore, the inserts will replicate the UKA designs (Figure 5). The knee should allow greater range of motion with the possibility of heel to buttocks contact. This increased motion will lead to greater roll back of the femur on the tibia and can lead to subluxation of the femoral runner off of the tibial surface on the lateral side, mimicking the normal knee. This subluxation is desirable but may lead to increased wear of the polyethylene on the lateral side of the knee.
The instruments should be specific for the design but must also be user-friendly. The 2 major issues with the surgery are balancing the knee in full extension and flexion, and preservation of the tibial bone island. The preexisting knee deformity should be <10° in all planes to limit the amount of collateral ligament releases. The collaterals must be balanced in a similar fashion to the standard TKA. Flexion contracture can be treated with posterior capsular release around the cruciates or with an increased distal femoral resection (2 mm at the maximum).
It is important to size the femur correctly because it will be difficult to adjust the flexion gap on the tibial side. A 9-mm posterior medial femoral condyle resection is a reasonable guide if the condyle is not atrophic. However, the exact resection thickness will be implant-specific and should be correlated with the dimensions of the prosthesis being implanted. The tibial bone island must be properly rotated with respect to the center line (Akagi’s line)49 and must not be undercut. The tibial instrument should include pins or blocks to prevent the sawblades from undercutting the island (Figure 6), as undermining leads to fracture in full extension. If undermining occurs, it may be possible to place a cancellous screw through the island and still preserve the ligaments. The integrity of the island is best tested by bringing the knee to full extension and checking for liftoff of the bone. If there is significant compromise of the island, the bone should be resected and either a CR or PS TKA can be implanted. Della Valle and colleagues50 reported a 9.2% incidence (11 of 119 cases) of bone island fracture in their early experience with a BCR TKA and improved this to 1.9% (5/258 cases) after reassessing their technique.
The gap tension should be evaluated either with traditional spacer blocks or with tensioning devices on the medial and lateral side of the knee after the tibial resections are completed. The polyethylene inserts are anatomically different. It may be possible to vary the thickness from medial to lateral, but not in excess of 2 mm.
As the BCR surgical techniques evolve, the balancing and tibial resection may be refined through specialized instrumentation. Such “smart instruments” that incorporate gyros may expedite tibial alignment, and sensor devices may assist with gap balancing. Haptic surgical robotic guides may assist in the tibial resection, facilitating bone island preservation by avoiding any possibility of undermining. At present these assistive aides are not necessary for the operation but may play a future role.
Clinical Results of Knee Arthroplasties
The results of knee replacements improved steadily from the 1970s through the 1990s. The scoring systems were somewhat limited and there was little data on the perception of the patients. The prosthetic designs stabilized at the end of the 1990s with only minor modifications since the year 2000. The 20-year results show similar findings for both the CR and the PS designs. There is little evidence to suggest a clinical correlation with the observed kinematic differences between CR and PS TKA designs.40,51-58 Multiple studies have demonstrated equivalent range of motion38,39,59 and subjective outcome measures (Table 1).60 A randomized prospective trial that compared kinematics and functional scores between the 2 designs failed to observe significant differences in function despite differences in kinematics.46 Equivalence in clinical outcome was further supported by a Cochrane Review meta-analysis that evaluated 1810 patients in 17 selected studies.61 The Knee Society scores have all been in the 92% to 95% ratings with survivals between 90% and 95%.
However, only 80% to 90% of patients are fully satisfied with their implants. The reasons for the dissatisfaction include unexplained anterior knee pain, stiffness, unexplained swelling, loss of range of motion, changes in proprioception, and loss of preoperative functions.14
The mid-term results of the BCR knees that were performed in the 1980s showed similar results to the CR and PS knees. Townley8 reported excellent clinical results with only 2% loosening at 2 to 11 years after surgery. Cloutier and colleagues9 reported 95% survival with improved proprioception at 9 to 11 years after surgery(Table 2).62,63
Studies comparing traditional TKA designs with cruciate preserving designs, both UKA and BCR, have found differences in subjective outcomes.62,64 Comparison of UKA and TKA in the same patient demonstrated significant preference for UKA, particularly with stair-climbing.65 Similarly, comparison between BCR and PS TKA or CR TKA demonstrated preference for BCR in 85% of patients.62
The new BCR knee designs have just started to come to the market.50 The surgical techniques are much improved over the 1980s and cruciate preservation is certainly much easier now. The new designs can produce full range of motion with kinematics that are almost identical to the normal knee in the cadaver laboratory and in computer analyses. These designs certainly should have a similar 20-year survival to the original BCR knees. However, the critical evaluation will be the patient satisfaction scores. With greater motion, better kinematics, and more precise balancing the scores would improve with these designs.
Conclusion
The cruciate ligaments of the knee are central to control of the motion of the normal knee. TKA is a successful operation with at least a 40- to 50-year history. The techniques have continued to develop but 15% to 20% of patients are dissatisfied with the results.14 Evaluations of the prostheses are more sophisticated and kinematics appears to have a central position in the evaluation. If the knee is to move more anatomically correctly, all of the ligaments must be preserved. Proprioception certainly plays a role in the patient’s judgment of the result. History has shown that a BCR knee can be implanted with good mid-term results and it should certainly be possible to build on these results and design a knee that will incorporate all of the ligaments with full range of motion and increased levels of activity.
Hinge knee arthroplasty was introduced in the 1950s.1 All 4 major ligaments were replaced by the hinge, which provided stabilization while allowing sagittal plane motion. Its goal was stability, not replication of normal kinematics. The addition of methyl methacrylate cement improved fixation and allowed surface design modifications that addressed normal articular motion. Implants such as the Gunston Polycentric,2 the Duocondylar,3 and the Geometric4 resurfaced the medial and lateral compartments of the knee while preserving the cruciate ligaments. The implants were subject to greater translational forces without the hinge and loosening became a major problem despite the advances in cementing. It became evident in the 1970s that preservation of the cruciates complicated the procedure. Cruciate resection simplified the operation and allowed improved fixation. The ICLH prosthesis resected the cruciates and used the articular surface design to give stability to the knee.5,6 The total condylar prosthesis had a “tibial” imminence that mimicked the shape of the tibial surface but also sacrificed both of the cruciate ligaments (Figure 1).
Designers recognized that the cruciate ligaments affected knee kinematics; however, they elected to sacrifice the anterior cruciate ligament (ACL) for surgical simplicity and implant longevity.6 In the early 1980s, both the cruciate-retaining (CR) total knee arthroplasty (TKA) (Figure 2) and posterior-stabilized (PS) TKA (Figure 3) designs addressed the posterior cruciate ligament (PCL) function. The PCL was preserved in the “cruciate-retaining” TKA, substituted in the “posterior-stabilized” TKA using a cam-post mechanism. The CR TKA designers believed that PCL preservation produced a more balanced knee with a more anatomical result, a more normal joint line, and better function, especially on stair climbing. The PS TKA designers admitted the value of posterior stabilization but argued that it was too difficult to consistently save the PCL in all cases, and that the PS knee was easier for surgeons to implant with more reliable roll back.7
The Geometric knee was developed in the 1970s to retain both cruciate ligaments.4 Unfortunately, it created a kinematic conflict by using a constrained articular surface design that prevented the motion required by the cruciate ligaments. This conflict resulted in tibial loosening and early failures. The compromised results decreased interest in the bicruciate-retaining (BCR) TKA designs, allowing the CR TKA and PS TKA designs to flourish for the next 20 years with little or no attempts to retain the ACL.
In the 1980s the BCR TKA design was pursued by Townley8 and Cartier.9 Townley8 believed that cruciate resection was a concession to “improper joint synchronization”8 and Cartier9 thought that cruciate preservation permitted more normal proprioception.9 Unlike prior BCR TKA designs, the mid-term clinical results were equal to or better than the standard CR TKA or PS TKA of the time, and 9- to 11-year follow-up demonstrated comparable outcomes.8 While these results highlighted the possibility of a BCR TKA, the surgical technique and failures of the Geometric knee discouraged surgeons from pursuing the BCR TKA.
Interest in cruciate-preserving knee arthroplasty returned with partial knee replacements, with patients reporting more normal proprioception and motion.10 The techniques became more popular with the introduction of the minimally invasive surgeries in the early 2000s and cruciate ligament preservation became a more interesting concept.11,12 Some surgeons preserved the cruciates by using separate implants for the medial, lateral, and patellofemoral surfaces.10 These results were acceptable for the time but required considerable surgical talent and did not report 20-year results similar to the CR and PS knees.
Most prosthetic designs attempt to copy the normal knee anatomy. Using fluoroscopic studies and computer analysis, designers began to investigate the motion (or kinematics) of the normal knee and realized that despite the fact the TKA looked like the human knee, the designs were not kinematically correct.13
Although TKA successfully treats pain secondary to degenerative joint disease, many patients are unable to return to their prior level of function, with up to 20% reporting dissatisfaction with their level of activity.14 The observed differences in kinematics between a normal knee and a TKA may explain part of this discrepancy.
Normal Knee Motion
The tibiofemoral articulation in a normal knee follows a reproducible pattern of motion as the knee moves from extension to flexion. The lateral femoral condyle (LFC) translates posteriorly with a combination of rolling and sliding motion, while the medial femoral condyle (MFC) has minimal posterior translation and thus acts as a pivot for knee motion. The MFC is larger, less curved, and has a biphasic shape with 2 distinct radiuses of curvature that correspond to an “extension” and “flexion” facet. The transition between the MFC facets occurs at approximately 30° of flexion, whereby the contact point transfers posteriorly with little condylar translation.15-17 In contrast, the LFC is smaller, has a single radius of curvature, and gradually translates posteriorly throughout flexion. Static magnetic resonance imaging of the knee from 0° to 120° shows an average of 19 mm posterior translation for the LFC and 2 mm for the MFC.15-20
In deep flexion, beyond 130°, posterior translation continues for both condyles. The LFC experiences enough excursion to cause loss of joint congruity and partial posterior subluxation.19,20 The MFC shows little additional posterior translation, yet it too loses joint congruity through condylar lift-off. Contact between the posterior horn of the medial meniscus and the posterior femoral condyle limits further flexion.16,21
The difference in motion between the condyles leads to internal tibial rotation during flexion. The initial 10° of knee flexion produces 5° of internal rotation, and an additional 15° of internal tibial rotation occurs throughout the remainder of knee flexion.
Fluoroscopic imaging with computed tomography (CT)- or magnetic resonance (MR)-based modeling has shown the dynamic in vivo relationship of the tibiofemoral joint. Studies have confirmed significantly greater LFC posterior translation as compared to the MFC;22 however, in vivo studies have also shown notable variability in articular rotation and translation based on activity. This highlights the role of ligamentous tension and muscle contraction in kinematics.21-23
The ACL in TKA
The majority of current TKA designs sacrifice the ACL without substituting for its function. The loss of the ACL has significant effects upon the kinematics of the knee.
The ACL is composed of 2 bundles, the anteromedial and posterolateral bundles, which originate on the LFC and insert broadly onto the tibial intercondylar eminence. Its primary role is to resist anterior tibial translation, particularly from 0° to 30° of flexion, which corresponds to the peak quadriceps force that pulls the tibia anteriorly.24 ACL deficiency causes anterior tibial translation during early flexion and abnormal internal tibial rotation.25-27 ACL deficient knees demonstrate a posterior femoral position in full extension, and increased MFC translation during knee flexion.28-32
The role of the ACL in knee arthroplasty has been evaluated by comparing unicompartmental knee arthroplasty (UKA) with TKA, as a reflection of ACL preserving vs sacrificing procedures.33-35 Sagittal plane translation is similar between UKA and normal knees,33,34 while the CR TKA and PS TKA designs show anterior tibia subluxation in full extension.33-35 The difference between UKA and TKA is greatest in extension, corresponding to the ACL functional range. These findings highlight kinematic similarities between TKA designs and the ACL deficient knee.
The majority of UKAs demonstrate near-normal kinematics. A small percentage of the study group demonstrated aberrant anterior tibial motion, highlighting a concern over ACL attenuation with time. Additionally, studies that evaluate the ACL in osteoarthritic knees have questioned the baseline integrity of the ACL.36 Yet the long-term outcomes in UKA design have shown preservation of kinematics due to intact cruciates.37
The PCL in TKA
Because the majority of TKA designs sacrifice the ACL, the classic debate has focused on the utility of the native PCL. Both the CR and PS TKA are designed to offer posterior stabilization; however, kinematic studies have demonstrated notable differences.38,39
The CR TKA design relies on the PCL to resist posterior sag and to prevent the hamstring musculature from pulling the tibia posteriorly during flexion. Studies have shown paradoxical anterior translation of both femoral condyles during flexion, particularly on the medial side of the knee.40 There is also increased variability in femoral rollback. It is unclear whether the PCL can function normally in the absence of the ACL, which causes the PCL to adapt a less anatomic vertical position. The PCL may also be unable to function significantly without the ACL because of pre-existing degenerative histological changes.41
The PS TKA utilizes a cam-post mechanism for posterior stabilization. In contrast to normal knee kinematics, this mechanism creates equal MFC and LFC posterior translation, 8 mm on average at 90° flexion.40 The equivalent translation in PS designs contributes to decreased internal tibial rotation and an increased polyethylene wear at the post.
Role of Surface Geometry
The articular geometry of the knee plays an important role in normal knee kinematics. Initial TKA designs used a femoral component with a single radius of curvature for both femoral condyles.42Current TKA designs that match the femoral component to the native femoral anatomy, by differing the medial and lateral condyle geometry, have demonstrated kinematics that better resemble a native knee.43 Additional changes to the radius of curvature along the posterior facet of the femoral condyles may reduce impingement during deep flexion. These “high flex” designs have demonstrated equivalent range of motion in some studies44 and improved weight-bearing motion in others.45 Surface geometry is important but is not the entire answer to kinematics.
Advances in TKA Design
Knee motion is guided by multiple factors, including the tibiofemoral articular geometry, the surrounding soft tissue tension, and muscle tone. Bicruciate-substituting (BCS) TKA and BCR TKA are forms of evolution from the CR and PS TKA and attempt to respect the function of both cruciate ligaments and provide better kinematics.
The BCS TKA utilizes a modified cam-post articulation to provide both anterior and posterior stabilization (Figure 4).46 The surgical approach remains the same and the implant geometry affects the motion. The BCS TKA design demonstrates femoral rollback at 90° with an average of 14 mm for the MFC and 23 mm for the LFC, and 10° internal tibial rotation.46,47 Additionally, it provides increased sagittal stability during early flexion and an improved pivot shift (indicating improved anterior stabilization).
The BCR designs preserve both cruciates and provide anterior and posterior stabilization. Fluoroscopic imaging has demonstrated contact points in full extension, and posterior rollback at 90° flexion that more closely replicates the normal knee.48
Design and Surgical Techniques for Bicruciate Knee Replacements
If all of the ligaments are preserved, the TKA surfaces must allow motion to be driven by the ligaments in combination with the surfaces alone. The femur can be designed anatomically with asymmetric condyles. The femoral box must allow for preservation of the tibial bone island without impinging upon the cruciate ligaments. The tibial surface must be minimally constrained with concavity medially and convexity laterally.
The bone island preservation does not permit a single-piece tibial polyethylene insert. Therefore, the inserts will replicate the UKA designs (Figure 5). The knee should allow greater range of motion with the possibility of heel to buttocks contact. This increased motion will lead to greater roll back of the femur on the tibia and can lead to subluxation of the femoral runner off of the tibial surface on the lateral side, mimicking the normal knee. This subluxation is desirable but may lead to increased wear of the polyethylene on the lateral side of the knee.
The instruments should be specific for the design but must also be user-friendly. The 2 major issues with the surgery are balancing the knee in full extension and flexion, and preservation of the tibial bone island. The preexisting knee deformity should be <10° in all planes to limit the amount of collateral ligament releases. The collaterals must be balanced in a similar fashion to the standard TKA. Flexion contracture can be treated with posterior capsular release around the cruciates or with an increased distal femoral resection (2 mm at the maximum).
It is important to size the femur correctly because it will be difficult to adjust the flexion gap on the tibial side. A 9-mm posterior medial femoral condyle resection is a reasonable guide if the condyle is not atrophic. However, the exact resection thickness will be implant-specific and should be correlated with the dimensions of the prosthesis being implanted. The tibial bone island must be properly rotated with respect to the center line (Akagi’s line)49 and must not be undercut. The tibial instrument should include pins or blocks to prevent the sawblades from undercutting the island (Figure 6), as undermining leads to fracture in full extension. If undermining occurs, it may be possible to place a cancellous screw through the island and still preserve the ligaments. The integrity of the island is best tested by bringing the knee to full extension and checking for liftoff of the bone. If there is significant compromise of the island, the bone should be resected and either a CR or PS TKA can be implanted. Della Valle and colleagues50 reported a 9.2% incidence (11 of 119 cases) of bone island fracture in their early experience with a BCR TKA and improved this to 1.9% (5/258 cases) after reassessing their technique.
The gap tension should be evaluated either with traditional spacer blocks or with tensioning devices on the medial and lateral side of the knee after the tibial resections are completed. The polyethylene inserts are anatomically different. It may be possible to vary the thickness from medial to lateral, but not in excess of 2 mm.
As the BCR surgical techniques evolve, the balancing and tibial resection may be refined through specialized instrumentation. Such “smart instruments” that incorporate gyros may expedite tibial alignment, and sensor devices may assist with gap balancing. Haptic surgical robotic guides may assist in the tibial resection, facilitating bone island preservation by avoiding any possibility of undermining. At present these assistive aides are not necessary for the operation but may play a future role.
Clinical Results of Knee Arthroplasties
The results of knee replacements improved steadily from the 1970s through the 1990s. The scoring systems were somewhat limited and there was little data on the perception of the patients. The prosthetic designs stabilized at the end of the 1990s with only minor modifications since the year 2000. The 20-year results show similar findings for both the CR and the PS designs. There is little evidence to suggest a clinical correlation with the observed kinematic differences between CR and PS TKA designs.40,51-58 Multiple studies have demonstrated equivalent range of motion38,39,59 and subjective outcome measures (Table 1).60 A randomized prospective trial that compared kinematics and functional scores between the 2 designs failed to observe significant differences in function despite differences in kinematics.46 Equivalence in clinical outcome was further supported by a Cochrane Review meta-analysis that evaluated 1810 patients in 17 selected studies.61 The Knee Society scores have all been in the 92% to 95% ratings with survivals between 90% and 95%.
However, only 80% to 90% of patients are fully satisfied with their implants. The reasons for the dissatisfaction include unexplained anterior knee pain, stiffness, unexplained swelling, loss of range of motion, changes in proprioception, and loss of preoperative functions.14
The mid-term results of the BCR knees that were performed in the 1980s showed similar results to the CR and PS knees. Townley8 reported excellent clinical results with only 2% loosening at 2 to 11 years after surgery. Cloutier and colleagues9 reported 95% survival with improved proprioception at 9 to 11 years after surgery(Table 2).62,63
Studies comparing traditional TKA designs with cruciate preserving designs, both UKA and BCR, have found differences in subjective outcomes.62,64 Comparison of UKA and TKA in the same patient demonstrated significant preference for UKA, particularly with stair-climbing.65 Similarly, comparison between BCR and PS TKA or CR TKA demonstrated preference for BCR in 85% of patients.62
The new BCR knee designs have just started to come to the market.50 The surgical techniques are much improved over the 1980s and cruciate preservation is certainly much easier now. The new designs can produce full range of motion with kinematics that are almost identical to the normal knee in the cadaver laboratory and in computer analyses. These designs certainly should have a similar 20-year survival to the original BCR knees. However, the critical evaluation will be the patient satisfaction scores. With greater motion, better kinematics, and more precise balancing the scores would improve with these designs.
Conclusion
The cruciate ligaments of the knee are central to control of the motion of the normal knee. TKA is a successful operation with at least a 40- to 50-year history. The techniques have continued to develop but 15% to 20% of patients are dissatisfied with the results.14 Evaluations of the prostheses are more sophisticated and kinematics appears to have a central position in the evaluation. If the knee is to move more anatomically correctly, all of the ligaments must be preserved. Proprioception certainly plays a role in the patient’s judgment of the result. History has shown that a BCR knee can be implanted with good mid-term results and it should certainly be possible to build on these results and design a knee that will incorporate all of the ligaments with full range of motion and increased levels of activity.
1. Walldius B. Arthroplasty of the knee with an endoprosthesis. Acta Chir Scand. 1957;113(6):445-446.
2. Gunston FH. Polycentric knee arthroplasty. Prosthetic simulation of normal knee movement. J Bone Joint Surg Br. 1971;53(2):272-277.
3. Insall JN, Ranawat CS, Aglietti P, Shine J. A comparison of four models of total knee-replacement prostheses. J Bone Joint Surg Am. 1976;58(6):754-765.
4. Coventry MB, Finerman GA, Riley LH, Turner RH, Upshaw JE. A new geometric knee for total knee arthroplasty. Clin Orthop Relat Res.1972;83:157-162.
5. Freeman MA, Sculco T, Todd RC. Replacement of the severely damaged arthritic knee by the ICLH (Freeman-Swanson) arthroplasty. J Bone Joint Surg Br. 1977;59(1):64-71.
6. Freeman MA, Insall JN, Besser W, Walker PS, Hallel T. Excision of the cruciate ligaments in total knee replacement. Clin Orthop Relat Res. 1977(126):209-212.
7. Pagnano MW, Cushner FD, Scott WN. Role of the posterior cruciate ligament in total knee arthroplasty. J Am Acad Orthop Surg. 1998;6(3):176-187.
8. Townley CO. The anatomic total knee resurfacing arthroplasty. Clin Orthop Relat Res. 1985(192):82-96.
9. Cloutier JM, Sabouret P, Deghrar A. Total knee arthroplasty with retention of both cruciate ligaments. A nine to eleven-year follow-up study. J Bone Joint Surg Am. 1999; 81(5):697-702.
10. Banks SA, Fregly BJ, Boniforti F, Reinschmidt C, Romagnoli S. Comparing in vivo kinematics of unicondylar and bi-unicondylar knee replacements. Knee Surg Sports Traumatol Arthrosc. 2005;13(7):551-556.
11. Repicci JA, Eberle RW. Minimally invasive surgical technique for unicondylar knee arthroplasty. J South Orthop Assoc. 1999;8(1):20-27; discussion 27.
12. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
13. Banks SA, Markovich GD, Hodge WA. In vivo kinematics of cruciate-retaining and -substituting knee arthroplasties. J Arthroplasty. 1997;12(3):297-304.
14. Nam D, Nunley RM, Barrack RL. Patient dissatisfaction following total knee replacement: a growing concern? Bone Joint J. 2014;96-B(11 Supple A):96-100.
15. Iwaki H, Pinskerova V, Freeman MA. Tibiofemoral movement 1: the shapes and relative movements of the femur and tibia in the unloaded cadaver knee. J Bone Joint Surg Br. 2000;82(8):1189-1195.
16. Johal P, Williams A, Wragg P, Hunt D, Gedroyc W. Tibio-femoral movement in the living knee. A study of weight bearing and non-weight bearing knee kinematics using ‘interventional’ MRI. J Biomech. 2005;38(2):269-276.
17. Pinskerova V, Johal P, Nakagawa S, et al. Does the femur roll-back with flexion? J Bone Joint Surg Br. 2004;86(6):925-931.
18. Hill PF, Vedi V, Williams A, Pinskerova V, Freeman MA. Tibiofemoral movement 2: the loaded and unloaded living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1196-1198.
19. Nakagawa S, Kadoya Y, Todo S, et al. Tibiofemoral movement 3: full flexion in the living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1199-1200.
20. Freeman MA, Pinskerova V. The movement of the knee studied by magnetic resonance imaging. Clin Orthop Relat Res. 2003(410):35-43.
21. Moro-oka TA, Hamai S, Miura H, et al. Dynamic activity dependence of in vivo normal knee kinematics. J Orthop Res. 2008;26(4):428-434.
22. Komistek RD, Dennis DA, Mahfouz M. In vivo fluoroscopic analysis of the normal human knee. Clin Orthop Relat Res. 2003(410):69-81.
23. Li G, DeFrate LE, Park SE, Gill TJ, Rubash HE. In vivo articular cartilage contact kinematics of the knee: an investigation using dual-orthogonal fluoroscopy and magnetic resonance image-based computer models. Am J Sports Med. 2005;33(1):102-107.
24. Grood ES, Suntay WJ, Noyes FR, Butler DL. Biomechanics of the knee-extension exercise. Effect of cutting the anterior cruciate ligament. J Bone Joint Surg Am. 1984;66(5):725-734.
25. Noyes FR, Jetter AW, Grood ES, Harms SP, Gardner EJ, Levy MS. Anterior cruciate ligament function in providing rotational stability assessed by medial and lateral tibiofemoral compartment translations and subluxations. Am J Sports Med. 2015;43(3):683-692.
26. Good L, Askew MJ, Boom A, Melby A 3rd. Kinematic in-vitro comparison between the normal knee and two techniques for reconstruction of the anterior cruciate ligament. Clin Biomech (Bristol, Avon). 1993;8(5):243-249.
27. Beard DJ, Murray DW, Gill HS. Reconstruction does not reduce tibial translation in the cruciate-deficient knee an in vivo study. J Bone Joint Surg Br. 2001;83(8):1098-1103.
28. Dennis DA, Mahfouz MR, Komistek RD, Hoff W. In vivo determination of normal and anterior cruciate ligament-deficient knee kinematics. J Biomech. 2005;38(2):241-253.
29. Beynnon BD, Fleming BC, Labovitch R, Parsons B. Chronic anterior cruciate ligament deficiency is associated with increased anterior translation of the tibia during the transition from non-weightbearing to weightbearing. J Orthop Res. 2002;20(2):332-337.
30. Brandsson S, Karlsson J, Eriksson BI, Kärrholm J. Kinematics after tear in the anterior cruciate ligament: dynamic bilateral radiostereometric studies in 11 patients. Acta Orthop Scand. 2001;72(4):372-378.
31. Andriacchi TP, Briant PL, Bevill SL, Koo S. Rotational changes at the knee after ACL injury cause cartilage thinning. Clin Orthop Relat Res. 2006;442:39-44.
32. Scarvell JM, Smith PN, Refshauge KM, Galloway HR, Woods KR. Comparison of kinematic analysis by mapping tibiofemoral contact with movement of the femoral condylar centres in healthy and anterior cruciate ligament injured knees. J Orthop Res. 2004;22(5):955-962.
33. Miller RK, Goodfellow JW, Murray DW, O’Connor JJ. In vitro measurement of patellofemoral force after three types of knee replacement. J Bone Joint Surg Br. 1998;80(5):900-906.
34. Price AJ, Rees JL, Beard DL, Gill RH, Dodd CA, Murray DM. Sagittal plane kinematics of a mobile-bearing unicompartmental knee arthroplasty at 10 years: a comparative in vivo fluoroscopic analysis. J Arthroplasty. 2004;19(5):590-597.
35. Dennis D, Komistek R, Scuderi G, et al. In vivo three-dimensional determination of kinematics for subjects with a normal knee or a unicompartmental or total knee replacement. J Bone Joint Surg Am. 2001;83-A Suppl 2 Pt 2:104-115.
36. Arbuthnot JE, Brink RB. Assessment of the antero-posterior and rotational stability of the anterior cruciate ligament analogue in a guided motion bi-cruciate stabilized total knee arthroplasty. J Med Eng Technol. 2009;33(8):610-615.
37. Hollinghurst D, Stoney J, Ward T, et al. No deterioration of kinematics and cruciate function 10 years after medial unicompartmental arthroplasty. Knee. 2006;13(6):440-444.
38. Dennis DA, Komistek RD, Colwell CE Jr, et al. In vivo anteroposterior femorotibial translation of total knee arthroplasty: a multicenter analysis. Clin Orthop Relat Res. 1998(356):47-57.
39. Dennis DA, Komistek RD, Hoff WA, Gabriel SM. In vivo knee kinematics derived using an inverse perspective technique. Clin Orthop Relat Res. 1996;(331):107-117.
40. Yoshiya S, Matsui N, Komistek RD, Dennis DA, Mahfouz M, Kurosaka M. In vivo kinematic comparison of posterior cruciate-retaining and posterior stabilized total knee arthroplasties under passive and weight-bearing conditions. J Arthroplasty. 2005;20(6):777-783.
41. Kleinbart FA, Bryk E, Evangelista J, Scott WN, Vigorita VJ. Histologic comparison of posterior cruciate ligaments from arthritic and age-matched knee specimens. J Arthroplasty. 1996;11(6):726-731.
42. Bull AM, Kessler O, Alam M, Amis AA. Changes in knee kinematics reflect the articular geometry after arthroplasty. Clin Orthop Relat Res. 2008;466(10):2491-2499.
43. Komistek RD, Mahfouz MR, Bertin KC, Rosenberg A, Kennedy W. In vivo determination of total knee arthroplasty kinematics: a multicenter analysis of an asymmetrical posterior cruciate retaining total knee arthroplasty. J Arthroplasty. 2008;23(1):41-50.
44. Mehin R, Burnett RS, Brasher PM. Does the new generation of high-flex knee prostheses improve the post-operative range of movement?: a meta-analysis. J Bone Joint Surg Br. 2010;92(10):1429-1434.
45. Dennis DA, Heekin RD, Clark CR, Murphy JA, O’Dell TL, Dwyer KA. Effect of implant design on knee flexion. J Arthroplasty. 2013;28(3):429-438.
46. Victor J, Mueller JK, Komistek RD, Sharma A, Nadaud MC, Bellemans J. In vivo kinematics after a cruciate-substituting TKA. Clin Orthop Relat Res. 2010;468(3):807-814.
47. Catani F, Ensini A, Belvedere C, et al. In vivo kinematics and kinetics of a bi-cruciate substituting total knee arthroplasty: a combined fluoroscopic and gait analysis study. J Orthop Res. 2009;27(12):1569-1575.
48. Stiehl JB, Komistek RD, Cloutier JM, Dennis DA. The cruciate ligaments in total knee arthroplasty: a kinematic analysis of 2 total knee arthroplasties. J Arthroplasty. 2000;15(5):545-550.
49. Akagi M, Oh M, Nonaka T, Tsujimoto H, Asano T, Hamanishi C. An anteroposterior axis of the tibia for total knee arthroplasty. Clin Orthop Relat Res. 2004;(420):213-219.
50. Della Valle CJ, Andriacchi TP, Berend KR, DeClaire JH, Lombardi AV Jr, Peters CL. Early experience with bi-cruciate retaining TKA. Poster presented at: American Academy of Orthopaedic Surgeons 2015 Annual Meeting; March 24-28, 2015; Las Vegas, NV.
51. Udomkiat P, Meng BJ, Dorr LD, Wan Z. Functional comparison of posterior cruciate retention and substitution knee replacement. Clin Orthop Relat Res. 2000;(378):192-201.
52. Tanzer M, Smith K, Burnett S. Posterior-stabilized versus cruciate-retaining total knee arthroplasty: balancing the gap. J Arthroplasty. 2002;17(7):813-819.
53. Maruyama S, Yoshiya S, Matsui N, Kuroda R, Kurosaka M. Functional comparison of posterior cruciate-retaining versus posterior stabilized total knee arthroplasty. J Arthroplasty. 2004;19(3):349-53.
54. Clark CR, Rorabeck CH, MacDonald S, MacDonald D, Swafford J, Cleland D. Posterior-stabilized and cruciate-retaining total knee replacement: a randomized study. Clin Orthop Relat Res. 2001;(392):208-212.
55. Swanik CB, Lephart SM, Rubash HE. Proprioception, kinesthesia, and balance after total knee arthroplasty with cruciate-retaining and posterior stabilized prostheses. J Bone Joint Surg Am. 2004;86-A(2):328-334.
56. Harato K, Bourne RB, Victor J, Snyder M, Hart J, Ries MD. Midterm comparison of posterior cruciate-retaining versus -substituting total knee arthroplasty using the Genesis II prosthesis. A multicenter prospective randomized clinical trial. Knee. 2008;15(3):217-221.
57. Catani F, Leardini A, Ensini A, et al. The stability of the cemented tibial component of total knee arthroplasty: posterior cruciate-retaining versus posterior-stabilized design. J Arthroplasty. 2004;19(6):775-782.
58. Dennis DA, Komistek RD, Stiehl JB, Walker SA, Dennis KN. Range of motion after total knee arthroplasty: the effect of implant design and weight-bearing conditions. J Arthroplasty. 1998;13(7):748-752.
59. Becker MW, Insall JN, Faris PM. Bilateral total knee arthroplasty. One cruciate retaining and one cruciate substituting. Clin Orthop Relat Res. 1991;(271):122-124.
60. Kim YH, Choi Y, Kwon OR, Kim JS. Functional outcome and range of motion of high-flexion posterior cruciate-retaining and high-flexion posterior cruciate-substituting total knee prostheses. A prospective, randomized study. J Bone Joint Surg Am. 2009;91(4):753-760.
61. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
62. Pritchett JW. Patients prefer a bicruciate-retaining or the medial pivot total knee prosthesis. J Arthroplasty. 2011;26(2):224-228.
63. Sabouret P, Lavoie F, Cloutier JM. Total knee replacement with retention of both cruciate ligaments: a 22-year follow-up study. Bone Joint J. 2013;95-B(7):917-922.
64. Andriacchi TP, Galante JO, Fermier RW. The influence of total knee-replacement design on walking and stair-climbing. J Bone Joint Surg Am. 1982;64(9):1328-1335.
65. Laurencin CT, Zelicof SB, Scott RD, Ewald FC. Unicompartmental versus total knee arthroplasty in the same patient. A comparative study. Clin Orthop Relat Res. 1991;(273):151-156.
66. Victor J, Banks S, Bellemans J. Kinematics of posterior cruciate ligament-retaining and -substituting total knee arthroplasty: a prospective randomised outcome study. J Bone Joint Surg Br. 2005;87(5):646-655.
1. Walldius B. Arthroplasty of the knee with an endoprosthesis. Acta Chir Scand. 1957;113(6):445-446.
2. Gunston FH. Polycentric knee arthroplasty. Prosthetic simulation of normal knee movement. J Bone Joint Surg Br. 1971;53(2):272-277.
3. Insall JN, Ranawat CS, Aglietti P, Shine J. A comparison of four models of total knee-replacement prostheses. J Bone Joint Surg Am. 1976;58(6):754-765.
4. Coventry MB, Finerman GA, Riley LH, Turner RH, Upshaw JE. A new geometric knee for total knee arthroplasty. Clin Orthop Relat Res.1972;83:157-162.
5. Freeman MA, Sculco T, Todd RC. Replacement of the severely damaged arthritic knee by the ICLH (Freeman-Swanson) arthroplasty. J Bone Joint Surg Br. 1977;59(1):64-71.
6. Freeman MA, Insall JN, Besser W, Walker PS, Hallel T. Excision of the cruciate ligaments in total knee replacement. Clin Orthop Relat Res. 1977(126):209-212.
7. Pagnano MW, Cushner FD, Scott WN. Role of the posterior cruciate ligament in total knee arthroplasty. J Am Acad Orthop Surg. 1998;6(3):176-187.
8. Townley CO. The anatomic total knee resurfacing arthroplasty. Clin Orthop Relat Res. 1985(192):82-96.
9. Cloutier JM, Sabouret P, Deghrar A. Total knee arthroplasty with retention of both cruciate ligaments. A nine to eleven-year follow-up study. J Bone Joint Surg Am. 1999; 81(5):697-702.
10. Banks SA, Fregly BJ, Boniforti F, Reinschmidt C, Romagnoli S. Comparing in vivo kinematics of unicondylar and bi-unicondylar knee replacements. Knee Surg Sports Traumatol Arthrosc. 2005;13(7):551-556.
11. Repicci JA, Eberle RW. Minimally invasive surgical technique for unicondylar knee arthroplasty. J South Orthop Assoc. 1999;8(1):20-27; discussion 27.
12. Romanowski MR, Repicci JA. Minimally invasive unicondylar arthroplasty: eight-year follow-up. J Knee Surg. 2002;15(1):17-22.
13. Banks SA, Markovich GD, Hodge WA. In vivo kinematics of cruciate-retaining and -substituting knee arthroplasties. J Arthroplasty. 1997;12(3):297-304.
14. Nam D, Nunley RM, Barrack RL. Patient dissatisfaction following total knee replacement: a growing concern? Bone Joint J. 2014;96-B(11 Supple A):96-100.
15. Iwaki H, Pinskerova V, Freeman MA. Tibiofemoral movement 1: the shapes and relative movements of the femur and tibia in the unloaded cadaver knee. J Bone Joint Surg Br. 2000;82(8):1189-1195.
16. Johal P, Williams A, Wragg P, Hunt D, Gedroyc W. Tibio-femoral movement in the living knee. A study of weight bearing and non-weight bearing knee kinematics using ‘interventional’ MRI. J Biomech. 2005;38(2):269-276.
17. Pinskerova V, Johal P, Nakagawa S, et al. Does the femur roll-back with flexion? J Bone Joint Surg Br. 2004;86(6):925-931.
18. Hill PF, Vedi V, Williams A, Pinskerova V, Freeman MA. Tibiofemoral movement 2: the loaded and unloaded living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1196-1198.
19. Nakagawa S, Kadoya Y, Todo S, et al. Tibiofemoral movement 3: full flexion in the living knee studied by MRI. J Bone Joint Surg Br. 2000;82(8):1199-1200.
20. Freeman MA, Pinskerova V. The movement of the knee studied by magnetic resonance imaging. Clin Orthop Relat Res. 2003(410):35-43.
21. Moro-oka TA, Hamai S, Miura H, et al. Dynamic activity dependence of in vivo normal knee kinematics. J Orthop Res. 2008;26(4):428-434.
22. Komistek RD, Dennis DA, Mahfouz M. In vivo fluoroscopic analysis of the normal human knee. Clin Orthop Relat Res. 2003(410):69-81.
23. Li G, DeFrate LE, Park SE, Gill TJ, Rubash HE. In vivo articular cartilage contact kinematics of the knee: an investigation using dual-orthogonal fluoroscopy and magnetic resonance image-based computer models. Am J Sports Med. 2005;33(1):102-107.
24. Grood ES, Suntay WJ, Noyes FR, Butler DL. Biomechanics of the knee-extension exercise. Effect of cutting the anterior cruciate ligament. J Bone Joint Surg Am. 1984;66(5):725-734.
25. Noyes FR, Jetter AW, Grood ES, Harms SP, Gardner EJ, Levy MS. Anterior cruciate ligament function in providing rotational stability assessed by medial and lateral tibiofemoral compartment translations and subluxations. Am J Sports Med. 2015;43(3):683-692.
26. Good L, Askew MJ, Boom A, Melby A 3rd. Kinematic in-vitro comparison between the normal knee and two techniques for reconstruction of the anterior cruciate ligament. Clin Biomech (Bristol, Avon). 1993;8(5):243-249.
27. Beard DJ, Murray DW, Gill HS. Reconstruction does not reduce tibial translation in the cruciate-deficient knee an in vivo study. J Bone Joint Surg Br. 2001;83(8):1098-1103.
28. Dennis DA, Mahfouz MR, Komistek RD, Hoff W. In vivo determination of normal and anterior cruciate ligament-deficient knee kinematics. J Biomech. 2005;38(2):241-253.
29. Beynnon BD, Fleming BC, Labovitch R, Parsons B. Chronic anterior cruciate ligament deficiency is associated with increased anterior translation of the tibia during the transition from non-weightbearing to weightbearing. J Orthop Res. 2002;20(2):332-337.
30. Brandsson S, Karlsson J, Eriksson BI, Kärrholm J. Kinematics after tear in the anterior cruciate ligament: dynamic bilateral radiostereometric studies in 11 patients. Acta Orthop Scand. 2001;72(4):372-378.
31. Andriacchi TP, Briant PL, Bevill SL, Koo S. Rotational changes at the knee after ACL injury cause cartilage thinning. Clin Orthop Relat Res. 2006;442:39-44.
32. Scarvell JM, Smith PN, Refshauge KM, Galloway HR, Woods KR. Comparison of kinematic analysis by mapping tibiofemoral contact with movement of the femoral condylar centres in healthy and anterior cruciate ligament injured knees. J Orthop Res. 2004;22(5):955-962.
33. Miller RK, Goodfellow JW, Murray DW, O’Connor JJ. In vitro measurement of patellofemoral force after three types of knee replacement. J Bone Joint Surg Br. 1998;80(5):900-906.
34. Price AJ, Rees JL, Beard DL, Gill RH, Dodd CA, Murray DM. Sagittal plane kinematics of a mobile-bearing unicompartmental knee arthroplasty at 10 years: a comparative in vivo fluoroscopic analysis. J Arthroplasty. 2004;19(5):590-597.
35. Dennis D, Komistek R, Scuderi G, et al. In vivo three-dimensional determination of kinematics for subjects with a normal knee or a unicompartmental or total knee replacement. J Bone Joint Surg Am. 2001;83-A Suppl 2 Pt 2:104-115.
36. Arbuthnot JE, Brink RB. Assessment of the antero-posterior and rotational stability of the anterior cruciate ligament analogue in a guided motion bi-cruciate stabilized total knee arthroplasty. J Med Eng Technol. 2009;33(8):610-615.
37. Hollinghurst D, Stoney J, Ward T, et al. No deterioration of kinematics and cruciate function 10 years after medial unicompartmental arthroplasty. Knee. 2006;13(6):440-444.
38. Dennis DA, Komistek RD, Colwell CE Jr, et al. In vivo anteroposterior femorotibial translation of total knee arthroplasty: a multicenter analysis. Clin Orthop Relat Res. 1998(356):47-57.
39. Dennis DA, Komistek RD, Hoff WA, Gabriel SM. In vivo knee kinematics derived using an inverse perspective technique. Clin Orthop Relat Res. 1996;(331):107-117.
40. Yoshiya S, Matsui N, Komistek RD, Dennis DA, Mahfouz M, Kurosaka M. In vivo kinematic comparison of posterior cruciate-retaining and posterior stabilized total knee arthroplasties under passive and weight-bearing conditions. J Arthroplasty. 2005;20(6):777-783.
41. Kleinbart FA, Bryk E, Evangelista J, Scott WN, Vigorita VJ. Histologic comparison of posterior cruciate ligaments from arthritic and age-matched knee specimens. J Arthroplasty. 1996;11(6):726-731.
42. Bull AM, Kessler O, Alam M, Amis AA. Changes in knee kinematics reflect the articular geometry after arthroplasty. Clin Orthop Relat Res. 2008;466(10):2491-2499.
43. Komistek RD, Mahfouz MR, Bertin KC, Rosenberg A, Kennedy W. In vivo determination of total knee arthroplasty kinematics: a multicenter analysis of an asymmetrical posterior cruciate retaining total knee arthroplasty. J Arthroplasty. 2008;23(1):41-50.
44. Mehin R, Burnett RS, Brasher PM. Does the new generation of high-flex knee prostheses improve the post-operative range of movement?: a meta-analysis. J Bone Joint Surg Br. 2010;92(10):1429-1434.
45. Dennis DA, Heekin RD, Clark CR, Murphy JA, O’Dell TL, Dwyer KA. Effect of implant design on knee flexion. J Arthroplasty. 2013;28(3):429-438.
46. Victor J, Mueller JK, Komistek RD, Sharma A, Nadaud MC, Bellemans J. In vivo kinematics after a cruciate-substituting TKA. Clin Orthop Relat Res. 2010;468(3):807-814.
47. Catani F, Ensini A, Belvedere C, et al. In vivo kinematics and kinetics of a bi-cruciate substituting total knee arthroplasty: a combined fluoroscopic and gait analysis study. J Orthop Res. 2009;27(12):1569-1575.
48. Stiehl JB, Komistek RD, Cloutier JM, Dennis DA. The cruciate ligaments in total knee arthroplasty: a kinematic analysis of 2 total knee arthroplasties. J Arthroplasty. 2000;15(5):545-550.
49. Akagi M, Oh M, Nonaka T, Tsujimoto H, Asano T, Hamanishi C. An anteroposterior axis of the tibia for total knee arthroplasty. Clin Orthop Relat Res. 2004;(420):213-219.
50. Della Valle CJ, Andriacchi TP, Berend KR, DeClaire JH, Lombardi AV Jr, Peters CL. Early experience with bi-cruciate retaining TKA. Poster presented at: American Academy of Orthopaedic Surgeons 2015 Annual Meeting; March 24-28, 2015; Las Vegas, NV.
51. Udomkiat P, Meng BJ, Dorr LD, Wan Z. Functional comparison of posterior cruciate retention and substitution knee replacement. Clin Orthop Relat Res. 2000;(378):192-201.
52. Tanzer M, Smith K, Burnett S. Posterior-stabilized versus cruciate-retaining total knee arthroplasty: balancing the gap. J Arthroplasty. 2002;17(7):813-819.
53. Maruyama S, Yoshiya S, Matsui N, Kuroda R, Kurosaka M. Functional comparison of posterior cruciate-retaining versus posterior stabilized total knee arthroplasty. J Arthroplasty. 2004;19(3):349-53.
54. Clark CR, Rorabeck CH, MacDonald S, MacDonald D, Swafford J, Cleland D. Posterior-stabilized and cruciate-retaining total knee replacement: a randomized study. Clin Orthop Relat Res. 2001;(392):208-212.
55. Swanik CB, Lephart SM, Rubash HE. Proprioception, kinesthesia, and balance after total knee arthroplasty with cruciate-retaining and posterior stabilized prostheses. J Bone Joint Surg Am. 2004;86-A(2):328-334.
56. Harato K, Bourne RB, Victor J, Snyder M, Hart J, Ries MD. Midterm comparison of posterior cruciate-retaining versus -substituting total knee arthroplasty using the Genesis II prosthesis. A multicenter prospective randomized clinical trial. Knee. 2008;15(3):217-221.
57. Catani F, Leardini A, Ensini A, et al. The stability of the cemented tibial component of total knee arthroplasty: posterior cruciate-retaining versus posterior-stabilized design. J Arthroplasty. 2004;19(6):775-782.
58. Dennis DA, Komistek RD, Stiehl JB, Walker SA, Dennis KN. Range of motion after total knee arthroplasty: the effect of implant design and weight-bearing conditions. J Arthroplasty. 1998;13(7):748-752.
59. Becker MW, Insall JN, Faris PM. Bilateral total knee arthroplasty. One cruciate retaining and one cruciate substituting. Clin Orthop Relat Res. 1991;(271):122-124.
60. Kim YH, Choi Y, Kwon OR, Kim JS. Functional outcome and range of motion of high-flexion posterior cruciate-retaining and high-flexion posterior cruciate-substituting total knee prostheses. A prospective, randomized study. J Bone Joint Surg Am. 2009;91(4):753-760.
61. Verra WC, van den Boom LG, Jacobs W, Clement DJ, Wymenga AA, Nelissen RG. Retention versus sacrifice of the posterior cruciate ligament in total knee arthroplasty for treating osteoarthritis. Cochrane Database Syst Rev. 2013;10:CD004803.
62. Pritchett JW. Patients prefer a bicruciate-retaining or the medial pivot total knee prosthesis. J Arthroplasty. 2011;26(2):224-228.
63. Sabouret P, Lavoie F, Cloutier JM. Total knee replacement with retention of both cruciate ligaments: a 22-year follow-up study. Bone Joint J. 2013;95-B(7):917-922.
64. Andriacchi TP, Galante JO, Fermier RW. The influence of total knee-replacement design on walking and stair-climbing. J Bone Joint Surg Am. 1982;64(9):1328-1335.
65. Laurencin CT, Zelicof SB, Scott RD, Ewald FC. Unicompartmental versus total knee arthroplasty in the same patient. A comparative study. Clin Orthop Relat Res. 1991;(273):151-156.
66. Victor J, Banks S, Bellemans J. Kinematics of posterior cruciate ligament-retaining and -substituting total knee arthroplasty: a prospective randomised outcome study. J Bone Joint Surg Br. 2005;87(5):646-655.
Management of the Biconcave (B2) Glenoid in Shoulder Arthroplasty: Technical Considerations
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
Total shoulder arthroplasty (TSA) has demonstrated excellent long-term clinical outcomes for the treatment of advanced glenohumeral osteoarthritis (OA).1-5 Glenohumeral OA is characterized by a broad spectrum of glenoid pathology. Both the morphology of the glenoid and humeral head subluxation are important preoperative factors to evaluate, as these have been shown to adversely impact shoulder arthroplasty outcomes.6,7
Walch and colleagues8 have previously classified glenoid morphology in cases of advanced glenohumeral arthritis based on the preoperative computed tomography (CT) scans of individuals undergoing shoulder arthroplasty (Figures 1A-1E). The biconcave (B2) glenoid is characterized by asymmetric posterior bone loss and a posterior translated humeral head that is seated in a biconcave glenoid. The degree and extent of bone loss in the B2 glenoid can be highly variable, ranging from the classic interpretation, in which 50% of the native glenoid fossa is preserved, to the more extreme case with little remaining native anterior glenoid. Scalise and colleagues9 have reported that determining the premorbid native glenoid version with a 3-dimensional (3D) glenoid vault model can aid in differentiating a pathologic B2 glenoid from a nonpathologic type C glenoid.
The B2 glenoid in particular has been associated with poor shoulder arthroplasty outcomes and component survivorship.6,10-12 There are many factors that are thought to contribute to this problem, such as glenoid component malposition, or undercorrection of the pathologic retroversion.6,13,14 Walch and colleagues10 reported that if the neoglenoid retroversion was greater than 27°, there was a 44% incidence of loosening and/or instability and 60% of the dislocations were observed when the humeral head subluxation was greater than 80%. Cases with severe posterior glenoid bone deficiency present a unique challenge to the surgeon, and the ability to accurately and securely place an implant in the correct anatomic position can be compromised. Standard TSA has proven excellent outcomes in the setting of typical glenohumeral OA, but in the B2 glenoid with significant posterior bone erosion, additional attention must be given to ensure adequate correction of the bony deformity, soft tissue balancing, and implant stability.
Several strategies that have been proposed to address extreme bone loss in the B2 glenoid will be discussed in this review. These include hemiarthroplasty, TSA with asymmetric reaming of the high side, TSA with bone grafting of the posterior glenoid bone loss, TSA with an augmented glenoid component, and reverse shoulder arthroplasty (RSA). Importantly, while these techniques have been proposed for managing the B2 glenoid, currently there is no gold standard consensus for the treatment of this condition. The purpose of this review is to highlight important characteristics of the B2 glenoid morphology on clinical outcomes and discuss the current surgical management options for this condition.
Preoperative Planning
Being able to accurately determine the amount of retroversion is critical for preoperative planning. Friedman and colleagues15 initially described a method to measure glenoid retroversion; however, this is less accurate in B2 glenoids (Figures 2A, 2B). More recently, Rouleau and colleagues16 have validated and published methods to measure glenoid retroversion and subluxation in the B2 glenoid using 3 reference lines: the paleoglenoid (native glenoid surface), intermediate glenoid (line from anterior and posterior edge), and neoglenoid (eroded posterior surface) (Figure 2).
Preoperative evaluation starts with plain radiographs; however, additional imaging is needed, as the axillary view has shown to overestimate retroversion in 86% of patients (Figures 3A-3E).17 For a detailed evaluation of the glenoid retroversion and bone deficiency, CT scans with 3D reconstructions are useful.18,19 The surgical plan should be guided by the location and extent of glenoid bone loss. One tool that has been developed to help in predicting premorbid glenoid version, inclination, and position of the joint line is the 3D virtual glenoid vault model.9,20,21 This helps determine accurate premorbid glenoid anatomy and has been shown to assist in the selection of the optimal implant in an attempt to restore native glenoid anatomy, and avoid peg perforation.21 Patient-specific instrumentation (PSI) for shoulder arthroplasty is being used more frequently and has shown promise for more accurate glenoid component placement, particularly in the complex glenoid with severe bone deficiency. PSI involves creating a custom-fitted guide that is referenced to surface anatomy derived from the preoperative CT scan, which can then direct the surgeon toward optimal implant position with regard to glenoid component location, version and inclination (Figures 4A, 4B). Early reports show that PSI has resulted in a significant reduction in the frequency of malpositioned glenoid implants, with the greatest benefit observed in patients with retroversion in excess of 16°.22
Surgical Management
Hemiarthroplasty
Shoulder hemiarthroplasty has been traditionally described as an option for younger, more active patients in whom longevity of the glenoid component is a concern, or in patients with inadequate glenoid bone stock to tolerate a glenoid component. While there are no reports of hemiarthroplasty specifically for patients with B2 glenoids, one study has examined the effect of glenoid morphology on the outcomes of hemiarthroplasty for shoulder osteoarthritis. Levine and colleagues7 reported inferior clinical outcomes after shoulder hemiarthroplasty in patients with eccentric posterior glenoid wear. Several authors have advocated a “ream-and-run” technique to create a concentric glenoid and re-center the humeral head while still maintaining the native glenoid.23,24 However, in a recent series of 162 ream-and-run procedures, Gilmer and colleagues25 reported that only 23% of patients with B2 glenoid geometry achieved a minimal clinically important change in patient-reported outcome scores and 14% required revision. Furthermore, Lynch and colleagues26 found that progressive medial erosion and recurrent posterior glenoid erosion occur in a significant percentage of patients at early follow-up. Given these recent findings, the use of hemiarthroplasty alone or a ream-and-run procedure for patients with B2 glenoid morphology should be approached with caution.
Total Shoulder Arthroplasty
As with any TSA, the primary goals in treating patients with B2 glenoid defects are to provide the patient with a pain-free, stable, and functional shoulder (Figures 5A-5D). There are, however, a few challenges that are unique to TSA in the setting of B2 glenoid defects. Because the humeral head is often subluxated posteriorly into the defect, the anterior capsule and rotator cuff can tighten while the posterior aspect of the joint becomes lax. These soft tissues must be balanced during TSA in order to stabilize the shoulder and restore the appropriate length-tension relationship of the rotator cuff. The other primary concern is restoration of appropriate glenoid version and lateralization. To accomplish this, the most common techniques utilized are asymmetric reaming, bone graft augmentation, and glenoid component augmentation.27,28
Asymmetric Reaming. One of the more readily utilized techniques for addressing the B2 glenoid during TSA is eccentric or asymmetric reaming. During this process, the anterior glenoid is preferentially reamed while little to no bone is removed posteriorly. This technique is generally felt to be sufficient to treat posterior defects up to 5 mm to 8 mm or retroversion up to 15°.28 These upper limits have been confirmed in a number of cadaveric and simulated models.29-31
The success of this technique hinges on excellent glenoid exposure. With appropriate retractors in place, the anterior capsulolabral complex, including the biceps insertion, is resected to improve visualization. The inferior capsule must be resected carefully to ensure exposure and better motion postoperatively. On the other hand, it is imperative to protect the posterior capsulolabral attachments because of the increased risk of posterior instability in patients with B2 glenoids.
Detailed imaging such as CT scans with 3D reconstructions have improved our understanding of the degree of the deformities in all directions, which can better guide the reaming. PSI and planning software developed to improve the surgeon’s ability to place the glenoid component centrally in the best possible position after version correction can be even more helpful. We find that using a burr to provisionally lower the high side (anterior) provides a more en face view, which subsequently makes the eccentric reaming easier. As a guide, we will not ream more than 1 cm of anterior bone or attempt to correct more than ~20° of retroversion. The goal should be to create a glenoid surface that is more neutral and congruent to the posterior surface of the glenoid component while not overmedializing the component.
Although eccentric reaming may be one of the more straightforward methods for addressing posterior glenoid erosion, it is not without a number of potential downsides. When attempting to correct defects >10 mm or retroversion beyond 15°, excessive medialization of the implant can occur. Although increasing the thickness of the glenoid component can compensate for small amounts of medialization, excessive medialization can lead to a number of issues.27,28,32 As reaming progresses medially, the risk of keel penetration increases as the glenoid vault narrows.30,32 Further medialization decreases posterior cortical support for the implant, which increases the risk of component loosening and subsidence.33-35 The more medial the implant is placed, the smaller the surface of available bone for implant fixation. This often requires utilization of a smaller sized glenoid component that may result in component mismatch with the humeral implant. Finally, excessive medialization has the potential to under tension the rotator cuff, leading to decreased shoulder stability, strength, and function.
Bone Graft Augmentation. When posterior erosion becomes too excessive to address with eccentric reaming alone, defect augmentation is another option to consider (Figures 6A-6E). While technically more demanding, bone graft also provides the advantage of better re-creating the natural joint line and center of rotation of the glenohumeral joint.
For most defects, the resected humeral head provides the ideal source of graft. After initial reaming of the anterior glenoid, the defect must be sized and measured. We then recommend using a guided, cannulated system to place a central pin, lying perpendicular to the glenoid axis in neutral position. The anterior glenoid is then reamed enough to create a flat surface on which to attach the bone graft. The posterior surface is then gently burred to create a bleeding surface to enhance graft incorporation. The graft is then contoured to the defect and placed flush with the anterior glenoid. Cannulated screws are placed over guidewires to fix the graft. Using an arthroscopic cannula inserted posteriorly allows for easier placement of the guidewires and easier implantation of the screws. Although a reamer or burr can be used to contour the graft once it is fixed in place, this should be minimized to prevent loss of fixation. When the graft is fixed, we then cement the glenoid component into place.
Although good clinical results have been obtained with this technique, there is concern of incomplete graft healing and component loosening in the long term. Even in clinically asymptomatic and well functioning patients, some degree of radiographic lucency may be present in over 50% of cases.31,36,37 Glenoid Component Augmentation. To address the issues related to lucency and nonunion of bone graft augmentation, several augmented glenoid components have been developed. Augmented glenoid components have the benefit of filling posterior defects and stabilizing the shoulder without requiring excessive medialization (as often occurs with eccentric reaming) or union of a bone-to-bone interface (as is required in bone graft augmentation).38 Although many of the metal back designs experienced undesirably high failure rates and have since been recalled,39 more modern all-polyethylene components hold promise. The 2 most commonly utilized designs are the posterior step augment (DePuy) and the posterior wedge (Exactech). Although biomechanical analyses of both designs have demonstrated increased stability during loading in cadaveric and simulation models, the step augment (DePuy) has demonstrated increased stability and resistance to loosening.40,41 Although midterm results are not yet available for this newest generation of augmented components, short-term results with 2 to 3 years of follow-up have demonstrated excellent clinical outcomes.28
Reverse Total Shoulder Arthroplasty
While most commonly indicated for patients with rotator cuff tear arthropathy, RSA has recently been advocated for older patients with osteoarthritis and B2 glenoids in the setting of an intact rotator cuff. The semi-constrained design of the RSA is a potential solution to the static posterior humeral head subluxation seen in patients with B2 glenoid geometry (Figure 6E).
Technically, RSA is often an easier solution than a TSA with bone grafting because there is usually enough glenoid bone stock for fixation. That said, we always get a CT scan with 3D reconstructions to better appreciate the anatomy. Note that in B2 glenoids, the bone loss is typically posterior and inferior. RSA in the setting of a B2 glenoid is one of the ideal indications to use PSI to ensure ideal placement of the central pin, which is the key to glenoid baseplate positioning. Even when using a RSA, eccentric reaming and/or bone grafting allow for more ideal component placement. Using the same eccentric reaming techniques described above, one should try to ream to place the baseplate at 10° of retroversion. In cases where retroversion cannot be corrected to 10°, graft can be taken from the humeral head, iliac crest, or allograft. A benefit to using bone graft with RSA as opposed to TSA is that the graft can be fashioned to the baseplate, impacted/compressed into the B2 glenoid, and then secured with a central compression screw and peripheral locking screws.
Mizuno and colleagues41 reported a retrospective series of 27 RSAs performed for primary glenohumeral osteoarthritis and biconcave glenoid. At a mean follow-up of nearly 5 years, the authors noted significant improvement in Constant scores and shoulder motion with minimal complications. There was no recurrence of posterior instability observed by the time of final follow-up.41
RSA is a promising treatment for primary glenohumeral arthritis with posterior glenoid bone loss and static posterior subluxation in elderly or less active patients, but the longevity of these implants has yet to be established for younger, more active patients and requires further study.
Conclusion
Reconstruction of the B2 glenoid presents a challenging clinical problem that has been associated with poor clinical outcomes and implant survivorship. The high failure rate from glenoid component loosening and subsequent premature implant failure can be substantially decreased with accurate glenoid component positioning and appropriate correction of the pathologic glenoid retroversion. Careful preoperative planning is essential for accurate preparation and execution of the optimal surgical plan. There are many surgical strategies to address the B2 glenoid, but no consensus on the optimal method exists, as the technique should be uniquely customized to the individual’s pathology and surgeon preference (Table). Cases with mild deformity may be corrected with eccentric reaming and TSA, while the more severe deformities may require posterior glenoid bone grafting and/or augmented implants to restore native version. Finally, the RSA is a reliable option to restore stability and address bone deficiency for the severe B2 glenoid in an older, lower demand patient.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
1. Barrett WP, Franklin JL, Jackins SE, Wyss CR, Matsen FA 3rd. Total shoulder arthroplasty. J Bone Joint Surg Am. 1987;69(6):865-872.
2. Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A. A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Joint Surg Am. 2005;87(9):1947-1956.
3. Matsen FA 3rd. Early effectiveness of shoulder arthroplasty for patients who have primary glenohumeral degenerative joint disease. J Bone Joint Surg Am. 1996;78(2):260-264.
4. Fenlin JM Jr, Frieman BG. Indications, technique, and results of total shoulder arthroplasty in osteoarthritis. Orthop Clin North Am. 1998;29(3):423-434.
5. Singh JA, Sperling JW, Cofield RH. Revision surgery following total shoulder arthroplasty: Analysis of 2588 shoulders over three decades (1976 to 2008). J Bone Joint Surg Br. 2011;93(11):1513-1517.
6. Iannotti JP, Norris TR. Influence of preoperative factors on outcome of shoulder arthroplasty for glenohumeral osteoarthritis. J Bone Joint Surg Am. 2003;85-A(2):251-258.
7. Levine WN, Djurasovic M, Glasson JM, Pollock RG, Flatow EL, Bigliani LU. Hemiarthroplasty for glenohumeral osteoarthritis: Results correlated to degree of glenoid wear. J Shoulder Elbow Surg. 1997;6(5):449-454.
8. Walch G, Badet R, Boulahia A, Khoury A. Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty. 1999;14(6):756-760.
9. Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Shoulder Elbow Surg. 2008;17(3):487-491.
10. Walch G, Moraga C, Young A, Castellanos-Rosas J. Results of anatomic nonconstrained prosthesis in primary osteoarthritis with biconcave glenoid. J Shoulder Elbow Surg. 2012;21(11):1526-1533.
11. Kany J, Katz D. How to deal with glenoid type B2 or C? How to prevent mistakes in implantation of glenoid component? Eur J Orthop Surg Traumatol. 2013;23(4):379-385.
12. Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Shoulder Elbow Surg. 2013;22(11):1589-1598.
13. Iannotti JP, Greeson C, Downing D, Sabesan V, Bryan JA. Effect of glenoid deformity on glenoid component placement in primary shoulder arthroplasty. J Shoulder Elbow Surg. 2012;21(1):48-55.
14. Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95(12):e82.
15. Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74(7):1032-1037.
16. Rouleau DM, Kidder JF, Pons-Villanueva J, Dynamidis S, Defranco M, Walch G. Glenoid version: How to measure it? Validity of different methods in two-dimensional computed tomography scans. J Shoulder Elbow Surg. 2010;19(8):1230-1237.
17. Nyffeler RW, Jost B, Pfirrmann CW, Gerber C. Measurement of glenoid version: Conventional radiographs versus computed tomography scans. J Shoulder Elbow Surg. 2003;12(5):493-496.
18. Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Shoulder Elbow Surg. 2011;20(4):577-583.
19. Bokor DJ, O’Sullivan MD, Hazan GJ. Variability of measurement of glenoid version on computed tomography scan. J Shoulder Elbow Surg. 1999;8(6):595-598.
20. Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: A comparison of 4 methods in 2- and 3-dimensional models. J Shoulder Elbow Surg. 2011;20(2):234-244.
21. Ricchetti ET, Hendel MD, Collins DN, Iannotti JP. Is premorbid glenoid anatomy altered in patients with glenohumeral osteoarthritis? Clin Orthop Relat Res. 2013;471(9):2932-2939.
22. Hendel MD, Bryan JA, Barsoum WK, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position: A randomized prospective clinical trial. J Bone Joint Surg Am. 2012;94(23):2167-2175.
23. Matsen FA 3rd, Warme WJ, Jackins SE. Can the ream and run procedure improve glenohumeral relationships and function for shoulders with the arthritic triad? Clin Orthop Relat Res. 2015;473(6):2088-2096.
24. Saltzman MD, Chamberlain AM, Mercer DM, Warme WJ, Bertelsen AL, Matsen FA 3rd. Shoulder hemiarthroplasty with concentric glenoid reaming in patients 55 years old or less. J Shoulder Elbow Surg. 2011;20(4):609-615.
25. Gilmer BB, Comstock BA, Jette JL, Warme WJ, Jackins SE, Matsen FA. The prognosis for improvement in comfort and function after the ream-and-run arthroplasty for glenohumeral arthritis: An analysis of 176 consecutive cases. J Bone Joint Surg Am. 2012;94(14):e102.
26. Lynch JR, Franta AK, Montgomery WH Jr, Lenters TR, Mounce D, Matsen FA 3rd. Self-assessed outcome at two to four years after shoulder hemiarthroplasty with concentric glenoid reaming. J Bone Joint Surg Am. 2007;89(6):1284-1292.
27. Donohue KW, Ricchetti ET, Iannotti JP. Surgical management of the biconcave (B2) glenoid. Curr Rev Musculoskelet Med. 2016;9(1):30-39.
28. Clavert P, Millett PJ, Warner JJ. Glenoid resurfacing: What are the limits to asymmetric reaming for posterior erosion? J Shoulder Elbow Surg. 2007;16(6):843-848.
29. Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: A cadaveric study. Orthopedics. 2009;32(1):21.
30. Neer CS 2nd, Morrison DS. Glenoid bone-grafting in total shoulder arthroplasty. J Bone Joint Surg Am. 1988;70(8):1154-1162.
31. Nowak DD, Bahu MJ, Gardner TR, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: The amount of glenoid retroversion that can be corrected. J Shoulder Elbow Surg. 2009;18(5):680-688.
32. Strauss EJ, Roche C, Flurin PH, Wright T, Zuckerman JD. The glenoid in shoulder arthroplasty. J Shoulder Elbow Surg. 2009;18(5):819-833.
33. Walch G, Young AA, Boileau P, Loew M, Gazielly D, Mole D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: Results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94(2):145-150.
34. Walch G, Young AA, Melis B, Gazielly D, Loew M, Boileau P. Results of a convex-back cemented keeled glenoid component in primary osteoarthritis: Multicenter study with a follow-up greater than 5 years. J Shoulder Elbow Surg. 2011;20(3):385-394.
35. Klika BJ, Wooten CW, Sperling JW, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):1066-1072.
36. Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Shoulder Elbow Surg. 2014;23(7):964-973.
37. Steinmann SP, Cofield RH. Bone grafting for glenoid deficiency in total shoulder replacement. J Shoulder Elbow Surg. 2000;9(5):361-367.
38. Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Shoulder Elbow Surg. 2014;23(7):e149-e157.
39. Iannotti JP, Lappin KE, Klotz CL, Reber EW, Swope SW. Liftoff resistance of augmented glenoid components during cyclic fatigue loading in the posterior-superior direction. J Shoulder Elbow Surg. 2013;22(11):1530-1536.
40. Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: A computational comparison by volume of bone removal and quality of remaining bone. J Shoulder Elbow Surg. 2015;24(8):1218-1226.
41. Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95(14):1297-1304.
Stem-Based Repair of the Subscapularis in Total Shoulder Arthroplasty
Subscapularis integrity following total shoulder arthroplasty (TSA) is important to maintaining glenohumeral joint stability and functional outcome. In recent years increased emphasis has been placed on the management of the subscapularis during TSA. Options for management of the subscapularis during TSA include tenotomy, release of the tendon from the bone (peel technique), or a lesser tuberosity osteotomy (LTO). Several studies have demonstrated that subscapularis integrity is often impaired with a traditional tenotomy approach.1,2 Based on these studies, a subscapularis peel or LTO approach have gained popularity.3 This technical article describes a subscapularis peel repair technique that is integrated into a press-fit anatomical short-stem during TSA.
Technique
The repair technique demonstrated in this article features the Univers Apex (Arthrex) humeral stem, but it can be adapted to other stems with features that allow for the incorporation of sutures.
A standard deltopectoral approach is used to gain access to the shoulder. The biceps tendon is released or tenotomized to gain access to the bicipital groove. The rotator interval is then opened beginning at the superior subscapularis by following the course of the anterior side of the proximal biceps and then directing the release toward the base of the coracoid in order to protect the supraspinatus tendon. Next, the subscapularis is sharply released from the lesser tuberosity. The tendon and capsule are released as a unit and a 3-sided release of the subscapularis is performed.
The humeral canal is opened with a reamer and broached to accommodate an appropriately sized press-fit component. A polyethylene glenoid component is placed and then attention is returned to the humerus.
Prior to placement of the humeral stem, 6 No. 2 or No. 5 FiberWire (Arthrex) sutures are pre-placed through suture holes in the stem (Figure 1). Four sutures are passed by hand through the medial calcar component and 2 sutures are placed through holes in the lateral portion of the stem. A 2.0-mm or 2.5-mm drill is used to create 2 holes in the bicipital groove: 1 at the superior aspect of the lesser tuberosity, and 1 at the inferior aspect of the lesser tuberosity (Figure 2A). Prior to impacting the stem, the 4 lateral suture limbs (limbs A through D) are shuttled through the holes in the bicipital groove (Figure 2B). Then the stem is impacted and secured, the final humeral head is placed, the joint is reduced, and the subscapularis is repaired (Figure 2C).
The 4 sutures passing through the medial calcar of the stem result in 8 suture limbs (limbs 1 through 8). Each limb is separately passed through the subscapularis tendon with a free needle, moving obliquely from inferior-medial to superior-lateral (Figure 3). Note: A variation is to pass 2 suture limbs at a time, but this technique has not been biomechanically investigated at the time of this writing.
Prior to tying the sutures, it is helpful to place a stitch between the superolateral corner of the subscapularis and the anterior supraspinatus in order to facilitate reduction. The suture limbs are then tied with a specific sequence to create a suture-bridging construct with 2 additional medial mattress sutures as follows (Figures 4A, 4B):
1 to A
4 to C
5 to B
8 to D
2 to 3
6 to 7
In this technique, each suture limb is tied to a limb from another suture. When the last 2 pairs are tied (2 to 3 and 6 to 7), they are tensioned to remove any slack from the repair and equalize tension within all suture pairs. After the sutures are tied, the rotator interval may be closed with simple sutures if desired. The patient is immobilized in a sling for 4 to 6 weeks. Immediate passive forward flexion is allowed as well as external rotation to 30°. Strengthening is initiated at 8 weeks.
Discussion
The incidence of TSA has increased dramatically in the last decade and is projected to continue in the coming years.4 In the majority of cases, TSA leads to improvement in pain and function. However, failures continue to exist. In addition to glenoid loosening, prosthetic instability and rotator cuff insufficiency are the most common causes of failure.5 The latter 2 are intimately related since glenohumeral stability depends largely upon the rotator cuff. Therefore, optimization of outcome following TSA depends largely upon maintaining integrity of the rotator cuff. While the incidence of preoperative rotator cuff tears and fatty degeneration of the rotator are not modifiable, the management of the subscapularis is in the hands of the surgeon.
While subscapularis tenotomy has historically been used to access the glenohumeral joint during TSA, this approach is associated with an alarmingly high failure rate. Jackson and colleagues1 reported that 7 out of 15 (47%) of subscapularis tendons managed with tenotomy during TSA were completely torn on postoperative ultrasound. The patients with postoperative rupture had decreased internal rotation strength and DASH scores (4.6 intact vs. 25 ruptured; P = .04) compared to the patients with an intact tendon. Scalise and colleagues2 retrospectively compared a tenotomy approach to a LTO. They reported that 7 out of 15 subscapularis tenotomies were ruptured or attenuated postoperatively. By comparison, 18 out of 20 LTOs were healed. Regardless of approach, functional outcome was higher at 1 year postoperative when the subscapularis was intact.
The high failure rate with tendon-to-tendon healing following tenotomy has led to interest in a subscapularis peel to achieve tendon-to-bone healing or an LTO approach to achieve bone-to-bone healing. Lapner and colleagues3 compared a peel to an LTO in a randomized controlled trial of 87 patients. At 2 years postoperative, there was no difference in functional outcome between the 2 groups.
While both a peel and an LTO approach can be repaired with the technique described in this article, there are advantages to a peel approach. First, a peel approach may be considered more reproducible, particularly for surgeons who do a limited amount of shoulder arthroplasty. Whereas an LTO can vary in size, the subscapularis can nearly always be reproducibly peeled from the lesser tuberosity. Second, this technique uses a short stem, which relies upon proximal fixation. While this approach is bone-preserving, a large osteotomy has the potential to compromise fixation of the stem. Therefore, while one of us (PJD) uses a fleck LTO with a short stem, we advise a peel technique in most cases.
In summary, the subscapularis repair technique described here provides a reproducible and biomechanically sound approach to managing the subscapularis during TSA.
1. Jackson JD, Cil A, Smith J, Steinmann SP. Integrity and function of the subscapularis after total shoulder arthroplasty. J Shoulder Elbow Surg. 2010;19(7):1085-1090.
2. Scalise JJ, Ciccone J, Iannotti JP. Clinical, radiographic, and ultrasonographic comparison of subscapularis tenotomy and lesser tuberosity osteotomy for total shoulder arthroplasty. J Bone Joint Surg Am. 2010;92(7):1627-1634.
3. Lapner PL, Sabri E, Rakhra K, Bell K, Athwal GS. Comparison of lesser tuberosity osteotomy to subscapularis peel in shoulder arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(24):2239-2246.
4. Kim SH, Wise BL, Zhang Y, Szabo RM. Increasing incidence of shoulder arthroplasty in the United States. J Bone Joint Surg Am. 2011;93(24):2249-2254.
5. Australian Orthopaedic Association National Joint Replacement Registry. Shoulder Arthroplasty 2015 Annual Report. https://aoanjrr.sahmri.com/documents/10180/217645/Shoulder%20Arthroplasty. Accessed April 7, 2016.
Subscapularis integrity following total shoulder arthroplasty (TSA) is important to maintaining glenohumeral joint stability and functional outcome. In recent years increased emphasis has been placed on the management of the subscapularis during TSA. Options for management of the subscapularis during TSA include tenotomy, release of the tendon from the bone (peel technique), or a lesser tuberosity osteotomy (LTO). Several studies have demonstrated that subscapularis integrity is often impaired with a traditional tenotomy approach.1,2 Based on these studies, a subscapularis peel or LTO approach have gained popularity.3 This technical article describes a subscapularis peel repair technique that is integrated into a press-fit anatomical short-stem during TSA.
Technique
The repair technique demonstrated in this article features the Univers Apex (Arthrex) humeral stem, but it can be adapted to other stems with features that allow for the incorporation of sutures.
A standard deltopectoral approach is used to gain access to the shoulder. The biceps tendon is released or tenotomized to gain access to the bicipital groove. The rotator interval is then opened beginning at the superior subscapularis by following the course of the anterior side of the proximal biceps and then directing the release toward the base of the coracoid in order to protect the supraspinatus tendon. Next, the subscapularis is sharply released from the lesser tuberosity. The tendon and capsule are released as a unit and a 3-sided release of the subscapularis is performed.
The humeral canal is opened with a reamer and broached to accommodate an appropriately sized press-fit component. A polyethylene glenoid component is placed and then attention is returned to the humerus.
Prior to placement of the humeral stem, 6 No. 2 or No. 5 FiberWire (Arthrex) sutures are pre-placed through suture holes in the stem (Figure 1). Four sutures are passed by hand through the medial calcar component and 2 sutures are placed through holes in the lateral portion of the stem. A 2.0-mm or 2.5-mm drill is used to create 2 holes in the bicipital groove: 1 at the superior aspect of the lesser tuberosity, and 1 at the inferior aspect of the lesser tuberosity (Figure 2A). Prior to impacting the stem, the 4 lateral suture limbs (limbs A through D) are shuttled through the holes in the bicipital groove (Figure 2B). Then the stem is impacted and secured, the final humeral head is placed, the joint is reduced, and the subscapularis is repaired (Figure 2C).
The 4 sutures passing through the medial calcar of the stem result in 8 suture limbs (limbs 1 through 8). Each limb is separately passed through the subscapularis tendon with a free needle, moving obliquely from inferior-medial to superior-lateral (Figure 3). Note: A variation is to pass 2 suture limbs at a time, but this technique has not been biomechanically investigated at the time of this writing.
Prior to tying the sutures, it is helpful to place a stitch between the superolateral corner of the subscapularis and the anterior supraspinatus in order to facilitate reduction. The suture limbs are then tied with a specific sequence to create a suture-bridging construct with 2 additional medial mattress sutures as follows (Figures 4A, 4B):
1 to A
4 to C
5 to B
8 to D
2 to 3
6 to 7
In this technique, each suture limb is tied to a limb from another suture. When the last 2 pairs are tied (2 to 3 and 6 to 7), they are tensioned to remove any slack from the repair and equalize tension within all suture pairs. After the sutures are tied, the rotator interval may be closed with simple sutures if desired. The patient is immobilized in a sling for 4 to 6 weeks. Immediate passive forward flexion is allowed as well as external rotation to 30°. Strengthening is initiated at 8 weeks.
Discussion
The incidence of TSA has increased dramatically in the last decade and is projected to continue in the coming years.4 In the majority of cases, TSA leads to improvement in pain and function. However, failures continue to exist. In addition to glenoid loosening, prosthetic instability and rotator cuff insufficiency are the most common causes of failure.5 The latter 2 are intimately related since glenohumeral stability depends largely upon the rotator cuff. Therefore, optimization of outcome following TSA depends largely upon maintaining integrity of the rotator cuff. While the incidence of preoperative rotator cuff tears and fatty degeneration of the rotator are not modifiable, the management of the subscapularis is in the hands of the surgeon.
While subscapularis tenotomy has historically been used to access the glenohumeral joint during TSA, this approach is associated with an alarmingly high failure rate. Jackson and colleagues1 reported that 7 out of 15 (47%) of subscapularis tendons managed with tenotomy during TSA were completely torn on postoperative ultrasound. The patients with postoperative rupture had decreased internal rotation strength and DASH scores (4.6 intact vs. 25 ruptured; P = .04) compared to the patients with an intact tendon. Scalise and colleagues2 retrospectively compared a tenotomy approach to a LTO. They reported that 7 out of 15 subscapularis tenotomies were ruptured or attenuated postoperatively. By comparison, 18 out of 20 LTOs were healed. Regardless of approach, functional outcome was higher at 1 year postoperative when the subscapularis was intact.
The high failure rate with tendon-to-tendon healing following tenotomy has led to interest in a subscapularis peel to achieve tendon-to-bone healing or an LTO approach to achieve bone-to-bone healing. Lapner and colleagues3 compared a peel to an LTO in a randomized controlled trial of 87 patients. At 2 years postoperative, there was no difference in functional outcome between the 2 groups.
While both a peel and an LTO approach can be repaired with the technique described in this article, there are advantages to a peel approach. First, a peel approach may be considered more reproducible, particularly for surgeons who do a limited amount of shoulder arthroplasty. Whereas an LTO can vary in size, the subscapularis can nearly always be reproducibly peeled from the lesser tuberosity. Second, this technique uses a short stem, which relies upon proximal fixation. While this approach is bone-preserving, a large osteotomy has the potential to compromise fixation of the stem. Therefore, while one of us (PJD) uses a fleck LTO with a short stem, we advise a peel technique in most cases.
In summary, the subscapularis repair technique described here provides a reproducible and biomechanically sound approach to managing the subscapularis during TSA.
Subscapularis integrity following total shoulder arthroplasty (TSA) is important to maintaining glenohumeral joint stability and functional outcome. In recent years increased emphasis has been placed on the management of the subscapularis during TSA. Options for management of the subscapularis during TSA include tenotomy, release of the tendon from the bone (peel technique), or a lesser tuberosity osteotomy (LTO). Several studies have demonstrated that subscapularis integrity is often impaired with a traditional tenotomy approach.1,2 Based on these studies, a subscapularis peel or LTO approach have gained popularity.3 This technical article describes a subscapularis peel repair technique that is integrated into a press-fit anatomical short-stem during TSA.
Technique
The repair technique demonstrated in this article features the Univers Apex (Arthrex) humeral stem, but it can be adapted to other stems with features that allow for the incorporation of sutures.
A standard deltopectoral approach is used to gain access to the shoulder. The biceps tendon is released or tenotomized to gain access to the bicipital groove. The rotator interval is then opened beginning at the superior subscapularis by following the course of the anterior side of the proximal biceps and then directing the release toward the base of the coracoid in order to protect the supraspinatus tendon. Next, the subscapularis is sharply released from the lesser tuberosity. The tendon and capsule are released as a unit and a 3-sided release of the subscapularis is performed.
The humeral canal is opened with a reamer and broached to accommodate an appropriately sized press-fit component. A polyethylene glenoid component is placed and then attention is returned to the humerus.
Prior to placement of the humeral stem, 6 No. 2 or No. 5 FiberWire (Arthrex) sutures are pre-placed through suture holes in the stem (Figure 1). Four sutures are passed by hand through the medial calcar component and 2 sutures are placed through holes in the lateral portion of the stem. A 2.0-mm or 2.5-mm drill is used to create 2 holes in the bicipital groove: 1 at the superior aspect of the lesser tuberosity, and 1 at the inferior aspect of the lesser tuberosity (Figure 2A). Prior to impacting the stem, the 4 lateral suture limbs (limbs A through D) are shuttled through the holes in the bicipital groove (Figure 2B). Then the stem is impacted and secured, the final humeral head is placed, the joint is reduced, and the subscapularis is repaired (Figure 2C).
The 4 sutures passing through the medial calcar of the stem result in 8 suture limbs (limbs 1 through 8). Each limb is separately passed through the subscapularis tendon with a free needle, moving obliquely from inferior-medial to superior-lateral (Figure 3). Note: A variation is to pass 2 suture limbs at a time, but this technique has not been biomechanically investigated at the time of this writing.
Prior to tying the sutures, it is helpful to place a stitch between the superolateral corner of the subscapularis and the anterior supraspinatus in order to facilitate reduction. The suture limbs are then tied with a specific sequence to create a suture-bridging construct with 2 additional medial mattress sutures as follows (Figures 4A, 4B):
1 to A
4 to C
5 to B
8 to D
2 to 3
6 to 7
In this technique, each suture limb is tied to a limb from another suture. When the last 2 pairs are tied (2 to 3 and 6 to 7), they are tensioned to remove any slack from the repair and equalize tension within all suture pairs. After the sutures are tied, the rotator interval may be closed with simple sutures if desired. The patient is immobilized in a sling for 4 to 6 weeks. Immediate passive forward flexion is allowed as well as external rotation to 30°. Strengthening is initiated at 8 weeks.
Discussion
The incidence of TSA has increased dramatically in the last decade and is projected to continue in the coming years.4 In the majority of cases, TSA leads to improvement in pain and function. However, failures continue to exist. In addition to glenoid loosening, prosthetic instability and rotator cuff insufficiency are the most common causes of failure.5 The latter 2 are intimately related since glenohumeral stability depends largely upon the rotator cuff. Therefore, optimization of outcome following TSA depends largely upon maintaining integrity of the rotator cuff. While the incidence of preoperative rotator cuff tears and fatty degeneration of the rotator are not modifiable, the management of the subscapularis is in the hands of the surgeon.
While subscapularis tenotomy has historically been used to access the glenohumeral joint during TSA, this approach is associated with an alarmingly high failure rate. Jackson and colleagues1 reported that 7 out of 15 (47%) of subscapularis tendons managed with tenotomy during TSA were completely torn on postoperative ultrasound. The patients with postoperative rupture had decreased internal rotation strength and DASH scores (4.6 intact vs. 25 ruptured; P = .04) compared to the patients with an intact tendon. Scalise and colleagues2 retrospectively compared a tenotomy approach to a LTO. They reported that 7 out of 15 subscapularis tenotomies were ruptured or attenuated postoperatively. By comparison, 18 out of 20 LTOs were healed. Regardless of approach, functional outcome was higher at 1 year postoperative when the subscapularis was intact.
The high failure rate with tendon-to-tendon healing following tenotomy has led to interest in a subscapularis peel to achieve tendon-to-bone healing or an LTO approach to achieve bone-to-bone healing. Lapner and colleagues3 compared a peel to an LTO in a randomized controlled trial of 87 patients. At 2 years postoperative, there was no difference in functional outcome between the 2 groups.
While both a peel and an LTO approach can be repaired with the technique described in this article, there are advantages to a peel approach. First, a peel approach may be considered more reproducible, particularly for surgeons who do a limited amount of shoulder arthroplasty. Whereas an LTO can vary in size, the subscapularis can nearly always be reproducibly peeled from the lesser tuberosity. Second, this technique uses a short stem, which relies upon proximal fixation. While this approach is bone-preserving, a large osteotomy has the potential to compromise fixation of the stem. Therefore, while one of us (PJD) uses a fleck LTO with a short stem, we advise a peel technique in most cases.
In summary, the subscapularis repair technique described here provides a reproducible and biomechanically sound approach to managing the subscapularis during TSA.
1. Jackson JD, Cil A, Smith J, Steinmann SP. Integrity and function of the subscapularis after total shoulder arthroplasty. J Shoulder Elbow Surg. 2010;19(7):1085-1090.
2. Scalise JJ, Ciccone J, Iannotti JP. Clinical, radiographic, and ultrasonographic comparison of subscapularis tenotomy and lesser tuberosity osteotomy for total shoulder arthroplasty. J Bone Joint Surg Am. 2010;92(7):1627-1634.
3. Lapner PL, Sabri E, Rakhra K, Bell K, Athwal GS. Comparison of lesser tuberosity osteotomy to subscapularis peel in shoulder arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(24):2239-2246.
4. Kim SH, Wise BL, Zhang Y, Szabo RM. Increasing incidence of shoulder arthroplasty in the United States. J Bone Joint Surg Am. 2011;93(24):2249-2254.
5. Australian Orthopaedic Association National Joint Replacement Registry. Shoulder Arthroplasty 2015 Annual Report. https://aoanjrr.sahmri.com/documents/10180/217645/Shoulder%20Arthroplasty. Accessed April 7, 2016.
1. Jackson JD, Cil A, Smith J, Steinmann SP. Integrity and function of the subscapularis after total shoulder arthroplasty. J Shoulder Elbow Surg. 2010;19(7):1085-1090.
2. Scalise JJ, Ciccone J, Iannotti JP. Clinical, radiographic, and ultrasonographic comparison of subscapularis tenotomy and lesser tuberosity osteotomy for total shoulder arthroplasty. J Bone Joint Surg Am. 2010;92(7):1627-1634.
3. Lapner PL, Sabri E, Rakhra K, Bell K, Athwal GS. Comparison of lesser tuberosity osteotomy to subscapularis peel in shoulder arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012;94(24):2239-2246.
4. Kim SH, Wise BL, Zhang Y, Szabo RM. Increasing incidence of shoulder arthroplasty in the United States. J Bone Joint Surg Am. 2011;93(24):2249-2254.
5. Australian Orthopaedic Association National Joint Replacement Registry. Shoulder Arthroplasty 2015 Annual Report. https://aoanjrr.sahmri.com/documents/10180/217645/Shoulder%20Arthroplasty. Accessed April 7, 2016.
Proximal Periprosthetic Femur Fractures: Strategies for Internal Fixation
The rate of total hip arthroplasty (THA) is rising and demand is expected to increase by 174% to 572,000 by 2030.1 The rate of periprosthetic fracture around primary THA is frequently reported at around 1%,2-4 though a recent study of over 32,000 THAs quotes the 20-year probability of periprosthetic fracture at 3.5%.5 Revision THA is also increasing in frequency and associated rates of periprosthetic fracture range from 1.5% to 7.8% following revision THA,3,4,6 with the probability of fracture at 20 years of 11%.7 Projection models predict that the number of periprosthetic fractures will rise by 4.6% per decade over the next 30 years.8
Broadly, treatment options include open reduction internal fixation (ORIF), revision THA, and combined approaches. The Vancouver classification, based on fracture location, stem stability, and bone loss, is often used to guide fracture treatment, with stable implants treated with ORIF and unstable implants requiring revision arthroplasty.
Fixation strategies for treatment of periprosthetic fracture around a well-fixed arthroplasty stem have evolved over time, and there continue to be a variety of available internal fixation options with no clear consensus on the optimal strategy.9 Rates of reoperation following ORIF of periprosthetic femur fracture are reported from 13% to 23%,8,10-12 confirming that there remains room for improvement in management of these injuries.
Locking Plate Fixation
Early fixation strategies included allograft and cables alone as well as nonlocked plate and cerclage constructs. In response to the complication and reoperation rate for nonlocked plate constructs, reported at 33%,13 locking plates were introduced as a treatment option, allowing for both improved osseous vascularity and added screw options.14 When compared to the traditional nonlocked Ogden construct, locking plate constructs are more resistant to axial and torsional load.15 Clinically, the relative risk of nonunion after nonlocking plate fixation is reported at 11.9 times that of fixation with locking plate technology.16
Successful use of lateral locking plate fixation for treatment of this injury has been reported on in several clinical series.17-20 Froberg and colleagues12 evaluated 60 Vancouver B1 and C fractures treated by locking plate osteosynthesis and reported no nonunions, an improvement from previous constructs. However, 8 out of 60 patients with 2-year follow-up required reoperation—4 for infection, 3 for refracture, and 1 for stem loosening—making it clear that the locking plate alone was not a panacea.
With locking plate fixation a mainstay of modern treatment of periprosthetic femur fractures, many questions still remain.
Proximal Fixation
Even with the introduction of locked plates, treatment success after ORIF of Vancouver B1 fractures relies on adequate proximal fixation. Options for proximal fixation around the stem include cerclage wires or cables, unicortical locked screws, obliquely directed bicortical screws, and use of the locking attachment plate to insert bicortical locked screws. These strategies can be used in the presence of cemented or uncemented stems, with biomechanical evidence that screw fixation through the cement mantle does not cause failure.21
Several biomechanical studies address the stiffness and strength of varying proximal fixation strategies. While early fixation relied heavily on cables, the use of cables alone as proximal fixation has been linked to significantly higher rates of failure when compared to other constructs in a large clinical series.11 Multiple biomechanical studies have shown that newer methods of proximal fixation provide more rigid constructs.22,23
Unicortical locked screws appear to outperform cables biomechanically. The use of unicortical screws in lieu of or in addition to cables provides added resistance to lateral bending as well as torsion when compared to cables alone.24 A second group found that unicortical locked screws alone were superior to combined fixation with cerclage wires and unicortical locked screws.25
Added stability can be demonstrated by bicortical fixation strategies, which offer increased rigidity when compared to cables or unicortical screws.22 In vitro work has shown enhanced fixation stability with bicortical screw fixation using the locking attachment plate when compared to cerclage wires alone.23,26 Clinically, some authors have demonstrated success with the use of reversed distal femoral locking plates in order to enhance proximal locking options and allow for bicortical fixation around the stem.19 As noted above, the data favor the opinion that clinical failure rates with cerclage wires alone are high, and biomechanically, bicortical fixation around the femoral stem appears to be superior to unicortical locked screw fixation or cerclage wires. If rigid proximal fixation is desired, an effort should be made to obtain bicortical fixation around the femoral stem.
Allograft
Allograft strut, either alone or in addition to plate osteosynthesis, has long been used in treatment of periprosthetic fractures. Proponents of this technique cite improved biomechanical stability17 and allograft incorporation resulting in restoration of bone stock.
Early treatment of periprosthetic femur fractures consisted solely of allograft and cable fixation, but data on the technique is limited. A small series reported reasonable success, with only 2 out of 19 patients developing nonunion.27 More recently Haddad and colleagues28 reported malunions in 3 out of 19 patients treated with allograft and cables alone. Allograft alone has been largely abandoned in favor of plate fixation, and biomechanical evidence shows that plate and screw or cerclage constructs are more resistant to torsion and lateral bending than allograft with cables alone.29
However, the role of allograft in treatment of periprosthetic femur fractures is not clearly defined. Some authors advocate routinely supplementing plate fixation with allograft28,30 and others go as far as to suggest superior union rates of strut allograft augmented plate fixation when compared to plate fixation alone for periprosthetic fractures around a stable femoral stem.31 However, in that series, the failure rate of 5/11 patients treated with plate alone is higher than current series,12 and others have demonstrated good success without allograft, even with nonlocked plates.32
As recently as 2016, a lateral locking plate supplemented with allograft has been described as a successful technique, with no nonunions reported in a small series.30 However, without a comparison group, it is unclear what role the allograft plays in success in that construct.
Despite some proposed benefits, the additional soft tissue stripping required to place allograft has raised the question of delayed healing and increased infection rate as a result of this technique. A systematic review by Moore and colleagues33 looking at the use of allograft strut in Vancouver B1 fractures found increased time to union (4.4 vs 6.6 months) and deep infection rate (3.8% vs 8.3%) with the use of allograft strut, leading them to recommend cautious use of allograft when treating Vancouver B1 fractures.
With improved fixation strategies available, the role of allograft may be best reserved for patients with inadequate bone stock.
Dual Plate Fixation
Dual plate fixation has been proposed as one mechanism to increase construct strength. A periprosthetic fracture model has shown that, biomechanically, orthogonal plates have higher bending stiffness, torsional stiffness, cycles to failure, and load to failure when compared to a single lateral plate with use of a locking attachment plate proximally.34 Choi and colleagues35 compared lateral locking plates alone, lateral locking plates with allograft, and lateral locking plates with an orthogonal anterior plate and found the addition of an anterior plate resulted in the strongest construct.
Clinically, Müller and colleagues36 reported on a series of 10 patients treated with orthogonal (anterior and lateral) plating for periprosthetic femur fractures, including 3 nonunions. In their series, there was 1 plate failure and they conclude that dual plating is not associated with an increased risk of complications, and can also be used as a salvage procedure.
While the evidence for dual plating is limited, it may provide needed additional stability in certain cases without the added cost and exposure required for allograft.
Minimally Invasive Plate Osteosynthesis
Contrary to the extensive exposure required to place allograft, minimally invasive plate osteosynthesis (MIPO) of periprosthetic femur fractures is advocated by some authors.18,20 Ricci and colleagues18 reported no nonunions in 50 patients treated with indirect reduction techniques and laterally based plating alone without use of allograft. A combination of cables, locking, and nonlocking screws were used. Critical to their technique was preservation of the soft tissue envelope at the level of the fracture.
In further support of MIPO techniques, a systematic review of 1571 periprosthetic hip fractures reported significantly increased risk of nonunion with open approaches when compared to minimally invasive osteosynthesis,16 emphasizing the role of preservation of vascularity in treating these fractures.
Length of Fixation
For some time it was recommended that fixation of Vancouver B1 fractures end 2 cortical diameters below the level of the fracture.37,38 More recently there has been interest in the potential benefits of increased length of fixation.
A biomechanical study comparing long (20-hole) and short (12-hole) plates for periprosthetic fracture with regard to failure found no difference in failure rates between groups.39 While plate length did not appear to affect construct stiffness, the issue of subsequent fracture distal to the construct remains.
Moloney and colleagues40 proposed fixation of Vancouver B1 fractures using plates that span the length of the femur to the level of the femoral condyles to minimize peri-implant failures in osteoporotic patients. In 36 patients treated with standard-length plates, there were 2 fractures distal to the previous fixation compared to no subsequent fractures in 21 patients treated with spanning fixation.
Similarly, in Vancouver C fractures there is some evidence that fixation should span the femoral stem, regardless of available bone for fixation proximal to the fracture. Kubiak and colleagues41 found increasing load to failure and decreased cortical strain in a biomechanical model comparing plates that stop short of the femoral stem with those that span the stem.
Clinically, this concept is supported by Froberg and colleagues.12 In their series of 60 Vancouver B1 and C fractures treated with laterally based locked plating, 3 patients went on to refracture. All of these fractures occurred in patients with Vancouver C fractures treated with plates overlapping the preexisting stem by <50%. The fractures all occurred at the high stress area between the tip of the stem and the end of the plate.
Further support of extended plate length comes from Drew and colleagues,8 who demonstrated a significantly decreased risk of reoperation following ORIF of periprosthetic femur fracture when >75% of the length of the femur was spanned compared to <50%. Although in some settings short fixation may produce satisfactory results, consideration should be given to extending the length of fixation, especially in the osteoporotic population.
Interprosthetic Fractures
With a rising number of patients with ipsilateral hip and knee arthroplasty, the rate of interprosthetic fractures is rising. These fractures present additional challenges given preexisting implants above and below the level of the fracture. The use of a single precontoured laterally based locked plate has been reported with good union rates approaching 90%.42,43 In one series, all nonunions occurred in Vancouver B1 fractures,43 again bringing to light the challenging nature of the B1 fracture.
Nonunion
Success in treating periprosthetic femur fractures has improved with improved fixation methods and understanding of technique. However, current rates of nonunion are still reported up to 27% for B1 and C fractures.44
There is limited evidence on the treatment of periprosthetic femur fracture nonunion. However, treatment is difficult and complication rates are high. Crockarell and colleagues45 reported a 52% overall complication rate in their series of 23 periprosthetic femur fracture nonunions.
Nonunions of the femur near a prosthesis can be treated by revision of the fracture fixation using compression and grafting to achieve bone healing vs revision of the joint prosthesis to span the area of the nonunited bone. Case-by-case decision-making is based on the remaining bone stock and the type of revision prosthesis necessary to span the problem area. Given the challenges associated with their treatment, a focus on prevention of nonunion is of paramount importance.
Authors’ Preferred Treatment
Our treatment of periprosthetic femur fractures with a well-fixed hip arthroplasty stem adheres to the principles supported in the literature (Figures 1A-1D and Figures 2A, 2B).
- Soft tissue friendly dissection with limited exposure at the fracture site is preferred as the fracture allows, particularly in cases with comminution where a direct assessment of the reduction is not available.
- Plate fixation strategy is dictated by the characteristics of the fracture. Fracture patterns amenable to anatomic reduction receive interfragmentary compression and absolute stability constructs. Highly comminuted fractures receive relatively stable bridging constructs to encourage callous.
- Locking screws are used rarely in diaphyseal fracture patterns, and when employed, are applied to only one side of the fracture to limit “over stiffening” the construct.
- Liberal use of dual plating, both as a method of maintaining fracture reduction while a structural plate is applied and increasing construct rigidity.
- Proximal fixation relies heavily on bicortical screws placed through the holes of the lateral plate. Cerclage wires and unicortical screws are rarely used in our practice. In the case of larger stems, a bicortical 3.5-mm screw can be placed through a 4.5-mm plate using a reduction washer.
Summary
Techniques for treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty stem are constantly evolving. Several principles have emerged to decrease rates of treatment failure and subsequent reoperation. While there are several methods to do so, it is critical to achieve stable proximal fixation. Long spanning fixation constructs are linked to lower failure and reoperation rates in both B1 and C type fractures. Additionally, the importance of soft tissue management and maintenance of local vascularity should not be underestimated.
1. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
2. Lewallen DG, Berry DJ. Periprosthetic fracture of the femur after total hip arthroplasty: treatment and results to date. Instr Course Lect. 1998;47:243-249.
3. Kavanagh BF. Femoral fractures associated with total hip arthroplasty. Orthop Clin North Am. 1992;23(2):249-257.
4. Meek RM, Norwood T, Smith R, Brenkel IJ, Howie CR. The risk of peri-prosthetic fracture after primary and revision total hip and knee replacement. J Bone Joint Surg Br. 2011;93(1):96-101.
5. Abdel MP, Watts CD, Houdek MT, Lewallen DG, Berry DJ. Epidemiology of periprosthetic fracture of the femur in 32 644 primary total hip arthroplasties: a 40-year experience. Bone Joint J. 2016;98-B(4):461-467.
6. Berry DJ. Epidemiology: hip and knee. Orthop Clin North Am. 1999;30(2):183-190.
7. Abdel MP, Houdek MT, Watts CD, Lewallen DG, Berry DJ. Epidemiology of periprosthetic femoral fractures in 5417 revision total hip arthrolasties: a 40-year experience. Bone Joint J. 2016;98-B(4):468-474.
8. Drew JM, Griffin WL, Odum SM, Van Doren B, Weston BT, Stryker LS. Survivorship after periprosthetic femur fracture: factors affecting outcome. J Arthroplasty. 2015. [Epub ahead of print]
9. Dehghan N, McKee MD, Nauth A, Ristevski B, Schemitsch EH. Surgical fixation of Vancouver type B1 periprosthetic femur fractures: a systematic review. J Orthop Trauma. 2014;28(12):721-727.
10. Mukundan C, Rayan F, Kheir E, Macdonald D. Management of late periprosthetic femur fractures: a retrospective cohort of 72 patients. Int Orthop. 2010;34(4):485-489.
11. Lindahl H, Malchau H, Odén A, Garellick G. Risk factors for failure after treatment of a periprosthetic fracture of the femur. J Bone Joint Surg Br. 2006;88(1):26-30.
12. Froberg L, Troelsen A, Brix M. Periprosthetic Vancouver type B1 and C fractures treated by locking-plate osteosynthesis: fracture union and reoperations in 60 consecutive fractures. Acta Orthop. 2012;83(6):648-652.
13. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996(327):238-246.
14. Perren SM. Evolution of the internal fixation of long bone fractures. The scientific basis of biological internal fixation: choosing a new balance between stability and biology. J Bone Joint Surg Br. 2002;84(8):1093-1110.
15. Fulkerson E, Koval K, Preston CF, Iesaka K, Kummer FJ, Egol KA. Fixation of periprosthetic femoral shaft fractures associated with cemented femoral stems: a biomechanical comparison of locked plating and conventional cable plates. J Orthop Trauma. 2006;20(2):89-93.
16. Stoffel K, Sommer C, Kalampoki V, Blumenthal A, Joeris A. The influence of the operation technique and implant used in the treatment of periprosthetic hip and interprosthetic femur fractures: a systematic literature review of 1571 cases. Arch Orthop Trauma Surg. 2016;136(4):553-561.
17. Fulkerson E, Tejwani N, Stuchin S, Egol K. Management of periprosthetic femur fractures with a first generation locking plate. Injury. 2007;38(8):965-972.
18. Ricci WM, Bolhofner BR, Loftus T, Cox C, Mitchell S, Borrelli J Jr. Indirect reduction and plate fixation, without grafting, for periprosthetic femoral shaft fractures about a stable intramedullary implant. Surgical technique. J Bone Joint Surg Am. 2006;88 Suppl 1 Pt 2:275-282.
19. Ebraheim NA, Gomez C, Ramineni SK, Liu J. Fixation of periprosthetic femoral shaft fractures adjacent to a well-fixed femoral stem with reversed distal femoral locking plate. J Trauma. 2009;66(4):1152-1157.
20. Bryant GK, Morshed S, Agel J, et al. Isolated locked compression plating for Vancouver Type B1 periprosthetic femoral fractures. Injury. 2009;40(11):1180-1186.
21. Giesinger K, Ebneter L, Day RE, Stoffel KK, Yates PJ, Kuster MS. Can plate osteosynthesis of periprosthethic femoral fractures cause cement mantle failure around a stable hip stem? A biomechanical analysis. J Arthroplasty. 2014;29(6):1308-1312.
22. Lewis GS, Caroom CT, Wee H, et al. Tangential bicortical locked fixation improves stability in vancouver B1 periprosthetic femur fractures: a biomechanical study. J Orthop Trauma. 2015;29(10):e364-e370.
23. Lenz M, Perren SM, Gueorguiev B, et al. A biomechanical study on proximal plate fixation techniques in periprosthetic femur fractures. Injury. 2014;45 Suppl 1:S71-S75.
24. Dennis MG, Simon JA, Kummer FJ, Koval KJ, DiCesare PE. Fixation of periprosthetic femoral shaft fractures occurring at the tip of the stem: a biomechanical study of 5 techniques. J Arthroplasty. 2000;15(4):523-528.
25. Graham SM, Mak JH, Moazen M, et al. Periprosthetic femoral fracture fixation: a biomechanical comparison between proximal locking screws and cables. J Orthop Sci. 2015;20(5):875-880.
26. Griffiths JT, Taheri A, Day RE, Yates PJ. Better axial stiffness of a bicortical screw construct compared to a cable construct for comminuted Vancouver B1 proximal femoral fractures. J Arthroplasty. 2015;30(12):2333-2337.
27. Chandler HP, King D, Limbird R, et al. The use of cortical allograft struts for fixation of fractures associated with well-fixed total joint prostheses. Semin Arthroplasty. 1993;4(2):99-107.
28. Haddad FS, Duncan CP, Berry DJ, Lewallen DG, Gross AE, Chandler HP. Periprosthetic femoral fractures around well-fixed implants: use of cortical onlay allografts with or without a plate. J Bone Joint Surg Am. 2002;84-A(6):945-950.
29. Dennis MG, Simon JA, Kummer FJ, Koval KJ, Di Cesare PE. Fixation of periprosthetic femoral shaft fractures: a biomechanical comparison of two techniques. J Orthop Trauma. 2001;15(3):177-180.
30. Yeo I, Rhyu KH, Kim SM, Park YS, Lim SJ. High union rates of locking compression plating with cortical strut allograft for type B1 periprosthetic femoral fractures. Int Orthop. 2016. [Epub ahead of print]
31. Khashan M, Amar E, Drexler M, Chechik Ok, Cohen Z, Steinberg EL. Superior outcome of strut allograft-augmented plate fixation for the treatment of periprosthetic fractures around a stable femoral stem. Injury. 2013;44(11):1556-1560.
32. Old AB, McGrory BJ, White RR, Babikian GM. Fixation of Vancouver B1 peri-prosthetic fractures by broad metal plates without the application of strut allografts. J Bone Joint Surg Br. 2006;88(11):1425-1429.
33. Moore RE, Baldwin K, Austin MS, Mehta S. A systematic review of open reduction and internal fixation of periprosthetic femur fractures with or without allograft strut, cerclage, and locked plates. J Arthroplasty. 2014;29(5):872-876.
34. Lenz M, Stoffel K, Gueorguiev B, Klos K, Kielstein H, Hofmann GO. Enhancing fixation strength in periprosthetic femur fractures by orthogonal plating-a biomechanical study. J Orthop Res. 2016;34(4):591-596.
35. Choi JK, Gardner TR, Yoon E, Morrison TA, Macaulay WB, Geller JA. The effect of fixation technique on the stiffness of comminuted Vancouver B1 periprosthetic femur fractures. J Arthroplasty. 2010;25(6 Suppl):124-128.
36. Müller FJ, Galler M Füchtmeier B. Clinical and radiological results of patients treated with orthogonal double plating for periprosthetic femoral fractures. Int Orthop. 2014;38(12):2469-2472.
37. Pike J, Davidson D, Garbuz D, Duncan CP, O’Brien PJ, Masri BA. Principles of treatment for periprosthetic femoral shaft fractures around well-fixed total hip arthroplasty. J Am Acad Orthop Surg. 2009;17(11):677-688.
38. Serocki JH, Chandler RW, Dorr LD. Treatment of fractures about hip prostheses with compression plating. J Arthroplasty. 1992;7(2):129-135.
39. Pletka JD, Marsland D, Belkoff SM, Mears SC, Kates SL. Biomechanical comparison of 2 different locking plate fixation methods in vancouver b1 periprosthetic femur fractures. Geriatr Orthop Surg Rehabil. 2011;2(2):51-55.
40. Moloney GB, Westrick ER, Siska PA, Tarkin IS. Treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty implant: span the whole bone. Arch Orthop Trauma Surg. 2014;134(1):9-14.
41. Kubiak EN, Haller JM, Kemper DD, Presson AP, Higgins TF, Horowitz DS. Does the lateral plate need to overlap the stem to mitigate stress concentration when treating Vancouver C periprosthetic supracondylar femur fracture? J Arthroplasty. 2015;30(1):104-108.
42. Sah AP, Marshall A, Virkus WV, Estok DM 2nd, Della Valle CJ. Interprosthetic fractures of the femur: treatment with a single-locked plate. J Arthroplasty. 2010;25(2):280-286.
43. Hoffmann MF, Lotzien S, Schildhauer TA. Clinical outcome of interprosthetic femoral fractures treated with polyaxial locking plates. Injury. 2016. [Epub ahead of print]
44. Holder N, Papp S, Gofton W, Beaulé PE. Outcomes following surgical treatment of periprosthetic femur fractures: a single centre series. Can J Surg. 2014;57(3):209-213.
45. Crockarell JR Jr, Berry DJ, Lewallen DG. Nonunion after periprosthetic femoral fracture associated with total hip arthroplasty. J Bone Joint Surg Am. 1999;81(8):1073-1079.
The rate of total hip arthroplasty (THA) is rising and demand is expected to increase by 174% to 572,000 by 2030.1 The rate of periprosthetic fracture around primary THA is frequently reported at around 1%,2-4 though a recent study of over 32,000 THAs quotes the 20-year probability of periprosthetic fracture at 3.5%.5 Revision THA is also increasing in frequency and associated rates of periprosthetic fracture range from 1.5% to 7.8% following revision THA,3,4,6 with the probability of fracture at 20 years of 11%.7 Projection models predict that the number of periprosthetic fractures will rise by 4.6% per decade over the next 30 years.8
Broadly, treatment options include open reduction internal fixation (ORIF), revision THA, and combined approaches. The Vancouver classification, based on fracture location, stem stability, and bone loss, is often used to guide fracture treatment, with stable implants treated with ORIF and unstable implants requiring revision arthroplasty.
Fixation strategies for treatment of periprosthetic fracture around a well-fixed arthroplasty stem have evolved over time, and there continue to be a variety of available internal fixation options with no clear consensus on the optimal strategy.9 Rates of reoperation following ORIF of periprosthetic femur fracture are reported from 13% to 23%,8,10-12 confirming that there remains room for improvement in management of these injuries.
Locking Plate Fixation
Early fixation strategies included allograft and cables alone as well as nonlocked plate and cerclage constructs. In response to the complication and reoperation rate for nonlocked plate constructs, reported at 33%,13 locking plates were introduced as a treatment option, allowing for both improved osseous vascularity and added screw options.14 When compared to the traditional nonlocked Ogden construct, locking plate constructs are more resistant to axial and torsional load.15 Clinically, the relative risk of nonunion after nonlocking plate fixation is reported at 11.9 times that of fixation with locking plate technology.16
Successful use of lateral locking plate fixation for treatment of this injury has been reported on in several clinical series.17-20 Froberg and colleagues12 evaluated 60 Vancouver B1 and C fractures treated by locking plate osteosynthesis and reported no nonunions, an improvement from previous constructs. However, 8 out of 60 patients with 2-year follow-up required reoperation—4 for infection, 3 for refracture, and 1 for stem loosening—making it clear that the locking plate alone was not a panacea.
With locking plate fixation a mainstay of modern treatment of periprosthetic femur fractures, many questions still remain.
Proximal Fixation
Even with the introduction of locked plates, treatment success after ORIF of Vancouver B1 fractures relies on adequate proximal fixation. Options for proximal fixation around the stem include cerclage wires or cables, unicortical locked screws, obliquely directed bicortical screws, and use of the locking attachment plate to insert bicortical locked screws. These strategies can be used in the presence of cemented or uncemented stems, with biomechanical evidence that screw fixation through the cement mantle does not cause failure.21
Several biomechanical studies address the stiffness and strength of varying proximal fixation strategies. While early fixation relied heavily on cables, the use of cables alone as proximal fixation has been linked to significantly higher rates of failure when compared to other constructs in a large clinical series.11 Multiple biomechanical studies have shown that newer methods of proximal fixation provide more rigid constructs.22,23
Unicortical locked screws appear to outperform cables biomechanically. The use of unicortical screws in lieu of or in addition to cables provides added resistance to lateral bending as well as torsion when compared to cables alone.24 A second group found that unicortical locked screws alone were superior to combined fixation with cerclage wires and unicortical locked screws.25
Added stability can be demonstrated by bicortical fixation strategies, which offer increased rigidity when compared to cables or unicortical screws.22 In vitro work has shown enhanced fixation stability with bicortical screw fixation using the locking attachment plate when compared to cerclage wires alone.23,26 Clinically, some authors have demonstrated success with the use of reversed distal femoral locking plates in order to enhance proximal locking options and allow for bicortical fixation around the stem.19 As noted above, the data favor the opinion that clinical failure rates with cerclage wires alone are high, and biomechanically, bicortical fixation around the femoral stem appears to be superior to unicortical locked screw fixation or cerclage wires. If rigid proximal fixation is desired, an effort should be made to obtain bicortical fixation around the femoral stem.
Allograft
Allograft strut, either alone or in addition to plate osteosynthesis, has long been used in treatment of periprosthetic fractures. Proponents of this technique cite improved biomechanical stability17 and allograft incorporation resulting in restoration of bone stock.
Early treatment of periprosthetic femur fractures consisted solely of allograft and cable fixation, but data on the technique is limited. A small series reported reasonable success, with only 2 out of 19 patients developing nonunion.27 More recently Haddad and colleagues28 reported malunions in 3 out of 19 patients treated with allograft and cables alone. Allograft alone has been largely abandoned in favor of plate fixation, and biomechanical evidence shows that plate and screw or cerclage constructs are more resistant to torsion and lateral bending than allograft with cables alone.29
However, the role of allograft in treatment of periprosthetic femur fractures is not clearly defined. Some authors advocate routinely supplementing plate fixation with allograft28,30 and others go as far as to suggest superior union rates of strut allograft augmented plate fixation when compared to plate fixation alone for periprosthetic fractures around a stable femoral stem.31 However, in that series, the failure rate of 5/11 patients treated with plate alone is higher than current series,12 and others have demonstrated good success without allograft, even with nonlocked plates.32
As recently as 2016, a lateral locking plate supplemented with allograft has been described as a successful technique, with no nonunions reported in a small series.30 However, without a comparison group, it is unclear what role the allograft plays in success in that construct.
Despite some proposed benefits, the additional soft tissue stripping required to place allograft has raised the question of delayed healing and increased infection rate as a result of this technique. A systematic review by Moore and colleagues33 looking at the use of allograft strut in Vancouver B1 fractures found increased time to union (4.4 vs 6.6 months) and deep infection rate (3.8% vs 8.3%) with the use of allograft strut, leading them to recommend cautious use of allograft when treating Vancouver B1 fractures.
With improved fixation strategies available, the role of allograft may be best reserved for patients with inadequate bone stock.
Dual Plate Fixation
Dual plate fixation has been proposed as one mechanism to increase construct strength. A periprosthetic fracture model has shown that, biomechanically, orthogonal plates have higher bending stiffness, torsional stiffness, cycles to failure, and load to failure when compared to a single lateral plate with use of a locking attachment plate proximally.34 Choi and colleagues35 compared lateral locking plates alone, lateral locking plates with allograft, and lateral locking plates with an orthogonal anterior plate and found the addition of an anterior plate resulted in the strongest construct.
Clinically, Müller and colleagues36 reported on a series of 10 patients treated with orthogonal (anterior and lateral) plating for periprosthetic femur fractures, including 3 nonunions. In their series, there was 1 plate failure and they conclude that dual plating is not associated with an increased risk of complications, and can also be used as a salvage procedure.
While the evidence for dual plating is limited, it may provide needed additional stability in certain cases without the added cost and exposure required for allograft.
Minimally Invasive Plate Osteosynthesis
Contrary to the extensive exposure required to place allograft, minimally invasive plate osteosynthesis (MIPO) of periprosthetic femur fractures is advocated by some authors.18,20 Ricci and colleagues18 reported no nonunions in 50 patients treated with indirect reduction techniques and laterally based plating alone without use of allograft. A combination of cables, locking, and nonlocking screws were used. Critical to their technique was preservation of the soft tissue envelope at the level of the fracture.
In further support of MIPO techniques, a systematic review of 1571 periprosthetic hip fractures reported significantly increased risk of nonunion with open approaches when compared to minimally invasive osteosynthesis,16 emphasizing the role of preservation of vascularity in treating these fractures.
Length of Fixation
For some time it was recommended that fixation of Vancouver B1 fractures end 2 cortical diameters below the level of the fracture.37,38 More recently there has been interest in the potential benefits of increased length of fixation.
A biomechanical study comparing long (20-hole) and short (12-hole) plates for periprosthetic fracture with regard to failure found no difference in failure rates between groups.39 While plate length did not appear to affect construct stiffness, the issue of subsequent fracture distal to the construct remains.
Moloney and colleagues40 proposed fixation of Vancouver B1 fractures using plates that span the length of the femur to the level of the femoral condyles to minimize peri-implant failures in osteoporotic patients. In 36 patients treated with standard-length plates, there were 2 fractures distal to the previous fixation compared to no subsequent fractures in 21 patients treated with spanning fixation.
Similarly, in Vancouver C fractures there is some evidence that fixation should span the femoral stem, regardless of available bone for fixation proximal to the fracture. Kubiak and colleagues41 found increasing load to failure and decreased cortical strain in a biomechanical model comparing plates that stop short of the femoral stem with those that span the stem.
Clinically, this concept is supported by Froberg and colleagues.12 In their series of 60 Vancouver B1 and C fractures treated with laterally based locked plating, 3 patients went on to refracture. All of these fractures occurred in patients with Vancouver C fractures treated with plates overlapping the preexisting stem by <50%. The fractures all occurred at the high stress area between the tip of the stem and the end of the plate.
Further support of extended plate length comes from Drew and colleagues,8 who demonstrated a significantly decreased risk of reoperation following ORIF of periprosthetic femur fracture when >75% of the length of the femur was spanned compared to <50%. Although in some settings short fixation may produce satisfactory results, consideration should be given to extending the length of fixation, especially in the osteoporotic population.
Interprosthetic Fractures
With a rising number of patients with ipsilateral hip and knee arthroplasty, the rate of interprosthetic fractures is rising. These fractures present additional challenges given preexisting implants above and below the level of the fracture. The use of a single precontoured laterally based locked plate has been reported with good union rates approaching 90%.42,43 In one series, all nonunions occurred in Vancouver B1 fractures,43 again bringing to light the challenging nature of the B1 fracture.
Nonunion
Success in treating periprosthetic femur fractures has improved with improved fixation methods and understanding of technique. However, current rates of nonunion are still reported up to 27% for B1 and C fractures.44
There is limited evidence on the treatment of periprosthetic femur fracture nonunion. However, treatment is difficult and complication rates are high. Crockarell and colleagues45 reported a 52% overall complication rate in their series of 23 periprosthetic femur fracture nonunions.
Nonunions of the femur near a prosthesis can be treated by revision of the fracture fixation using compression and grafting to achieve bone healing vs revision of the joint prosthesis to span the area of the nonunited bone. Case-by-case decision-making is based on the remaining bone stock and the type of revision prosthesis necessary to span the problem area. Given the challenges associated with their treatment, a focus on prevention of nonunion is of paramount importance.
Authors’ Preferred Treatment
Our treatment of periprosthetic femur fractures with a well-fixed hip arthroplasty stem adheres to the principles supported in the literature (Figures 1A-1D and Figures 2A, 2B).
- Soft tissue friendly dissection with limited exposure at the fracture site is preferred as the fracture allows, particularly in cases with comminution where a direct assessment of the reduction is not available.
- Plate fixation strategy is dictated by the characteristics of the fracture. Fracture patterns amenable to anatomic reduction receive interfragmentary compression and absolute stability constructs. Highly comminuted fractures receive relatively stable bridging constructs to encourage callous.
- Locking screws are used rarely in diaphyseal fracture patterns, and when employed, are applied to only one side of the fracture to limit “over stiffening” the construct.
- Liberal use of dual plating, both as a method of maintaining fracture reduction while a structural plate is applied and increasing construct rigidity.
- Proximal fixation relies heavily on bicortical screws placed through the holes of the lateral plate. Cerclage wires and unicortical screws are rarely used in our practice. In the case of larger stems, a bicortical 3.5-mm screw can be placed through a 4.5-mm plate using a reduction washer.
Summary
Techniques for treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty stem are constantly evolving. Several principles have emerged to decrease rates of treatment failure and subsequent reoperation. While there are several methods to do so, it is critical to achieve stable proximal fixation. Long spanning fixation constructs are linked to lower failure and reoperation rates in both B1 and C type fractures. Additionally, the importance of soft tissue management and maintenance of local vascularity should not be underestimated.
The rate of total hip arthroplasty (THA) is rising and demand is expected to increase by 174% to 572,000 by 2030.1 The rate of periprosthetic fracture around primary THA is frequently reported at around 1%,2-4 though a recent study of over 32,000 THAs quotes the 20-year probability of periprosthetic fracture at 3.5%.5 Revision THA is also increasing in frequency and associated rates of periprosthetic fracture range from 1.5% to 7.8% following revision THA,3,4,6 with the probability of fracture at 20 years of 11%.7 Projection models predict that the number of periprosthetic fractures will rise by 4.6% per decade over the next 30 years.8
Broadly, treatment options include open reduction internal fixation (ORIF), revision THA, and combined approaches. The Vancouver classification, based on fracture location, stem stability, and bone loss, is often used to guide fracture treatment, with stable implants treated with ORIF and unstable implants requiring revision arthroplasty.
Fixation strategies for treatment of periprosthetic fracture around a well-fixed arthroplasty stem have evolved over time, and there continue to be a variety of available internal fixation options with no clear consensus on the optimal strategy.9 Rates of reoperation following ORIF of periprosthetic femur fracture are reported from 13% to 23%,8,10-12 confirming that there remains room for improvement in management of these injuries.
Locking Plate Fixation
Early fixation strategies included allograft and cables alone as well as nonlocked plate and cerclage constructs. In response to the complication and reoperation rate for nonlocked plate constructs, reported at 33%,13 locking plates were introduced as a treatment option, allowing for both improved osseous vascularity and added screw options.14 When compared to the traditional nonlocked Ogden construct, locking plate constructs are more resistant to axial and torsional load.15 Clinically, the relative risk of nonunion after nonlocking plate fixation is reported at 11.9 times that of fixation with locking plate technology.16
Successful use of lateral locking plate fixation for treatment of this injury has been reported on in several clinical series.17-20 Froberg and colleagues12 evaluated 60 Vancouver B1 and C fractures treated by locking plate osteosynthesis and reported no nonunions, an improvement from previous constructs. However, 8 out of 60 patients with 2-year follow-up required reoperation—4 for infection, 3 for refracture, and 1 for stem loosening—making it clear that the locking plate alone was not a panacea.
With locking plate fixation a mainstay of modern treatment of periprosthetic femur fractures, many questions still remain.
Proximal Fixation
Even with the introduction of locked plates, treatment success after ORIF of Vancouver B1 fractures relies on adequate proximal fixation. Options for proximal fixation around the stem include cerclage wires or cables, unicortical locked screws, obliquely directed bicortical screws, and use of the locking attachment plate to insert bicortical locked screws. These strategies can be used in the presence of cemented or uncemented stems, with biomechanical evidence that screw fixation through the cement mantle does not cause failure.21
Several biomechanical studies address the stiffness and strength of varying proximal fixation strategies. While early fixation relied heavily on cables, the use of cables alone as proximal fixation has been linked to significantly higher rates of failure when compared to other constructs in a large clinical series.11 Multiple biomechanical studies have shown that newer methods of proximal fixation provide more rigid constructs.22,23
Unicortical locked screws appear to outperform cables biomechanically. The use of unicortical screws in lieu of or in addition to cables provides added resistance to lateral bending as well as torsion when compared to cables alone.24 A second group found that unicortical locked screws alone were superior to combined fixation with cerclage wires and unicortical locked screws.25
Added stability can be demonstrated by bicortical fixation strategies, which offer increased rigidity when compared to cables or unicortical screws.22 In vitro work has shown enhanced fixation stability with bicortical screw fixation using the locking attachment plate when compared to cerclage wires alone.23,26 Clinically, some authors have demonstrated success with the use of reversed distal femoral locking plates in order to enhance proximal locking options and allow for bicortical fixation around the stem.19 As noted above, the data favor the opinion that clinical failure rates with cerclage wires alone are high, and biomechanically, bicortical fixation around the femoral stem appears to be superior to unicortical locked screw fixation or cerclage wires. If rigid proximal fixation is desired, an effort should be made to obtain bicortical fixation around the femoral stem.
Allograft
Allograft strut, either alone or in addition to plate osteosynthesis, has long been used in treatment of periprosthetic fractures. Proponents of this technique cite improved biomechanical stability17 and allograft incorporation resulting in restoration of bone stock.
Early treatment of periprosthetic femur fractures consisted solely of allograft and cable fixation, but data on the technique is limited. A small series reported reasonable success, with only 2 out of 19 patients developing nonunion.27 More recently Haddad and colleagues28 reported malunions in 3 out of 19 patients treated with allograft and cables alone. Allograft alone has been largely abandoned in favor of plate fixation, and biomechanical evidence shows that plate and screw or cerclage constructs are more resistant to torsion and lateral bending than allograft with cables alone.29
However, the role of allograft in treatment of periprosthetic femur fractures is not clearly defined. Some authors advocate routinely supplementing plate fixation with allograft28,30 and others go as far as to suggest superior union rates of strut allograft augmented plate fixation when compared to plate fixation alone for periprosthetic fractures around a stable femoral stem.31 However, in that series, the failure rate of 5/11 patients treated with plate alone is higher than current series,12 and others have demonstrated good success without allograft, even with nonlocked plates.32
As recently as 2016, a lateral locking plate supplemented with allograft has been described as a successful technique, with no nonunions reported in a small series.30 However, without a comparison group, it is unclear what role the allograft plays in success in that construct.
Despite some proposed benefits, the additional soft tissue stripping required to place allograft has raised the question of delayed healing and increased infection rate as a result of this technique. A systematic review by Moore and colleagues33 looking at the use of allograft strut in Vancouver B1 fractures found increased time to union (4.4 vs 6.6 months) and deep infection rate (3.8% vs 8.3%) with the use of allograft strut, leading them to recommend cautious use of allograft when treating Vancouver B1 fractures.
With improved fixation strategies available, the role of allograft may be best reserved for patients with inadequate bone stock.
Dual Plate Fixation
Dual plate fixation has been proposed as one mechanism to increase construct strength. A periprosthetic fracture model has shown that, biomechanically, orthogonal plates have higher bending stiffness, torsional stiffness, cycles to failure, and load to failure when compared to a single lateral plate with use of a locking attachment plate proximally.34 Choi and colleagues35 compared lateral locking plates alone, lateral locking plates with allograft, and lateral locking plates with an orthogonal anterior plate and found the addition of an anterior plate resulted in the strongest construct.
Clinically, Müller and colleagues36 reported on a series of 10 patients treated with orthogonal (anterior and lateral) plating for periprosthetic femur fractures, including 3 nonunions. In their series, there was 1 plate failure and they conclude that dual plating is not associated with an increased risk of complications, and can also be used as a salvage procedure.
While the evidence for dual plating is limited, it may provide needed additional stability in certain cases without the added cost and exposure required for allograft.
Minimally Invasive Plate Osteosynthesis
Contrary to the extensive exposure required to place allograft, minimally invasive plate osteosynthesis (MIPO) of periprosthetic femur fractures is advocated by some authors.18,20 Ricci and colleagues18 reported no nonunions in 50 patients treated with indirect reduction techniques and laterally based plating alone without use of allograft. A combination of cables, locking, and nonlocking screws were used. Critical to their technique was preservation of the soft tissue envelope at the level of the fracture.
In further support of MIPO techniques, a systematic review of 1571 periprosthetic hip fractures reported significantly increased risk of nonunion with open approaches when compared to minimally invasive osteosynthesis,16 emphasizing the role of preservation of vascularity in treating these fractures.
Length of Fixation
For some time it was recommended that fixation of Vancouver B1 fractures end 2 cortical diameters below the level of the fracture.37,38 More recently there has been interest in the potential benefits of increased length of fixation.
A biomechanical study comparing long (20-hole) and short (12-hole) plates for periprosthetic fracture with regard to failure found no difference in failure rates between groups.39 While plate length did not appear to affect construct stiffness, the issue of subsequent fracture distal to the construct remains.
Moloney and colleagues40 proposed fixation of Vancouver B1 fractures using plates that span the length of the femur to the level of the femoral condyles to minimize peri-implant failures in osteoporotic patients. In 36 patients treated with standard-length plates, there were 2 fractures distal to the previous fixation compared to no subsequent fractures in 21 patients treated with spanning fixation.
Similarly, in Vancouver C fractures there is some evidence that fixation should span the femoral stem, regardless of available bone for fixation proximal to the fracture. Kubiak and colleagues41 found increasing load to failure and decreased cortical strain in a biomechanical model comparing plates that stop short of the femoral stem with those that span the stem.
Clinically, this concept is supported by Froberg and colleagues.12 In their series of 60 Vancouver B1 and C fractures treated with laterally based locked plating, 3 patients went on to refracture. All of these fractures occurred in patients with Vancouver C fractures treated with plates overlapping the preexisting stem by <50%. The fractures all occurred at the high stress area between the tip of the stem and the end of the plate.
Further support of extended plate length comes from Drew and colleagues,8 who demonstrated a significantly decreased risk of reoperation following ORIF of periprosthetic femur fracture when >75% of the length of the femur was spanned compared to <50%. Although in some settings short fixation may produce satisfactory results, consideration should be given to extending the length of fixation, especially in the osteoporotic population.
Interprosthetic Fractures
With a rising number of patients with ipsilateral hip and knee arthroplasty, the rate of interprosthetic fractures is rising. These fractures present additional challenges given preexisting implants above and below the level of the fracture. The use of a single precontoured laterally based locked plate has been reported with good union rates approaching 90%.42,43 In one series, all nonunions occurred in Vancouver B1 fractures,43 again bringing to light the challenging nature of the B1 fracture.
Nonunion
Success in treating periprosthetic femur fractures has improved with improved fixation methods and understanding of technique. However, current rates of nonunion are still reported up to 27% for B1 and C fractures.44
There is limited evidence on the treatment of periprosthetic femur fracture nonunion. However, treatment is difficult and complication rates are high. Crockarell and colleagues45 reported a 52% overall complication rate in their series of 23 periprosthetic femur fracture nonunions.
Nonunions of the femur near a prosthesis can be treated by revision of the fracture fixation using compression and grafting to achieve bone healing vs revision of the joint prosthesis to span the area of the nonunited bone. Case-by-case decision-making is based on the remaining bone stock and the type of revision prosthesis necessary to span the problem area. Given the challenges associated with their treatment, a focus on prevention of nonunion is of paramount importance.
Authors’ Preferred Treatment
Our treatment of periprosthetic femur fractures with a well-fixed hip arthroplasty stem adheres to the principles supported in the literature (Figures 1A-1D and Figures 2A, 2B).
- Soft tissue friendly dissection with limited exposure at the fracture site is preferred as the fracture allows, particularly in cases with comminution where a direct assessment of the reduction is not available.
- Plate fixation strategy is dictated by the characteristics of the fracture. Fracture patterns amenable to anatomic reduction receive interfragmentary compression and absolute stability constructs. Highly comminuted fractures receive relatively stable bridging constructs to encourage callous.
- Locking screws are used rarely in diaphyseal fracture patterns, and when employed, are applied to only one side of the fracture to limit “over stiffening” the construct.
- Liberal use of dual plating, both as a method of maintaining fracture reduction while a structural plate is applied and increasing construct rigidity.
- Proximal fixation relies heavily on bicortical screws placed through the holes of the lateral plate. Cerclage wires and unicortical screws are rarely used in our practice. In the case of larger stems, a bicortical 3.5-mm screw can be placed through a 4.5-mm plate using a reduction washer.
Summary
Techniques for treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty stem are constantly evolving. Several principles have emerged to decrease rates of treatment failure and subsequent reoperation. While there are several methods to do so, it is critical to achieve stable proximal fixation. Long spanning fixation constructs are linked to lower failure and reoperation rates in both B1 and C type fractures. Additionally, the importance of soft tissue management and maintenance of local vascularity should not be underestimated.
1. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
2. Lewallen DG, Berry DJ. Periprosthetic fracture of the femur after total hip arthroplasty: treatment and results to date. Instr Course Lect. 1998;47:243-249.
3. Kavanagh BF. Femoral fractures associated with total hip arthroplasty. Orthop Clin North Am. 1992;23(2):249-257.
4. Meek RM, Norwood T, Smith R, Brenkel IJ, Howie CR. The risk of peri-prosthetic fracture after primary and revision total hip and knee replacement. J Bone Joint Surg Br. 2011;93(1):96-101.
5. Abdel MP, Watts CD, Houdek MT, Lewallen DG, Berry DJ. Epidemiology of periprosthetic fracture of the femur in 32 644 primary total hip arthroplasties: a 40-year experience. Bone Joint J. 2016;98-B(4):461-467.
6. Berry DJ. Epidemiology: hip and knee. Orthop Clin North Am. 1999;30(2):183-190.
7. Abdel MP, Houdek MT, Watts CD, Lewallen DG, Berry DJ. Epidemiology of periprosthetic femoral fractures in 5417 revision total hip arthrolasties: a 40-year experience. Bone Joint J. 2016;98-B(4):468-474.
8. Drew JM, Griffin WL, Odum SM, Van Doren B, Weston BT, Stryker LS. Survivorship after periprosthetic femur fracture: factors affecting outcome. J Arthroplasty. 2015. [Epub ahead of print]
9. Dehghan N, McKee MD, Nauth A, Ristevski B, Schemitsch EH. Surgical fixation of Vancouver type B1 periprosthetic femur fractures: a systematic review. J Orthop Trauma. 2014;28(12):721-727.
10. Mukundan C, Rayan F, Kheir E, Macdonald D. Management of late periprosthetic femur fractures: a retrospective cohort of 72 patients. Int Orthop. 2010;34(4):485-489.
11. Lindahl H, Malchau H, Odén A, Garellick G. Risk factors for failure after treatment of a periprosthetic fracture of the femur. J Bone Joint Surg Br. 2006;88(1):26-30.
12. Froberg L, Troelsen A, Brix M. Periprosthetic Vancouver type B1 and C fractures treated by locking-plate osteosynthesis: fracture union and reoperations in 60 consecutive fractures. Acta Orthop. 2012;83(6):648-652.
13. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996(327):238-246.
14. Perren SM. Evolution of the internal fixation of long bone fractures. The scientific basis of biological internal fixation: choosing a new balance between stability and biology. J Bone Joint Surg Br. 2002;84(8):1093-1110.
15. Fulkerson E, Koval K, Preston CF, Iesaka K, Kummer FJ, Egol KA. Fixation of periprosthetic femoral shaft fractures associated with cemented femoral stems: a biomechanical comparison of locked plating and conventional cable plates. J Orthop Trauma. 2006;20(2):89-93.
16. Stoffel K, Sommer C, Kalampoki V, Blumenthal A, Joeris A. The influence of the operation technique and implant used in the treatment of periprosthetic hip and interprosthetic femur fractures: a systematic literature review of 1571 cases. Arch Orthop Trauma Surg. 2016;136(4):553-561.
17. Fulkerson E, Tejwani N, Stuchin S, Egol K. Management of periprosthetic femur fractures with a first generation locking plate. Injury. 2007;38(8):965-972.
18. Ricci WM, Bolhofner BR, Loftus T, Cox C, Mitchell S, Borrelli J Jr. Indirect reduction and plate fixation, without grafting, for periprosthetic femoral shaft fractures about a stable intramedullary implant. Surgical technique. J Bone Joint Surg Am. 2006;88 Suppl 1 Pt 2:275-282.
19. Ebraheim NA, Gomez C, Ramineni SK, Liu J. Fixation of periprosthetic femoral shaft fractures adjacent to a well-fixed femoral stem with reversed distal femoral locking plate. J Trauma. 2009;66(4):1152-1157.
20. Bryant GK, Morshed S, Agel J, et al. Isolated locked compression plating for Vancouver Type B1 periprosthetic femoral fractures. Injury. 2009;40(11):1180-1186.
21. Giesinger K, Ebneter L, Day RE, Stoffel KK, Yates PJ, Kuster MS. Can plate osteosynthesis of periprosthethic femoral fractures cause cement mantle failure around a stable hip stem? A biomechanical analysis. J Arthroplasty. 2014;29(6):1308-1312.
22. Lewis GS, Caroom CT, Wee H, et al. Tangential bicortical locked fixation improves stability in vancouver B1 periprosthetic femur fractures: a biomechanical study. J Orthop Trauma. 2015;29(10):e364-e370.
23. Lenz M, Perren SM, Gueorguiev B, et al. A biomechanical study on proximal plate fixation techniques in periprosthetic femur fractures. Injury. 2014;45 Suppl 1:S71-S75.
24. Dennis MG, Simon JA, Kummer FJ, Koval KJ, DiCesare PE. Fixation of periprosthetic femoral shaft fractures occurring at the tip of the stem: a biomechanical study of 5 techniques. J Arthroplasty. 2000;15(4):523-528.
25. Graham SM, Mak JH, Moazen M, et al. Periprosthetic femoral fracture fixation: a biomechanical comparison between proximal locking screws and cables. J Orthop Sci. 2015;20(5):875-880.
26. Griffiths JT, Taheri A, Day RE, Yates PJ. Better axial stiffness of a bicortical screw construct compared to a cable construct for comminuted Vancouver B1 proximal femoral fractures. J Arthroplasty. 2015;30(12):2333-2337.
27. Chandler HP, King D, Limbird R, et al. The use of cortical allograft struts for fixation of fractures associated with well-fixed total joint prostheses. Semin Arthroplasty. 1993;4(2):99-107.
28. Haddad FS, Duncan CP, Berry DJ, Lewallen DG, Gross AE, Chandler HP. Periprosthetic femoral fractures around well-fixed implants: use of cortical onlay allografts with or without a plate. J Bone Joint Surg Am. 2002;84-A(6):945-950.
29. Dennis MG, Simon JA, Kummer FJ, Koval KJ, Di Cesare PE. Fixation of periprosthetic femoral shaft fractures: a biomechanical comparison of two techniques. J Orthop Trauma. 2001;15(3):177-180.
30. Yeo I, Rhyu KH, Kim SM, Park YS, Lim SJ. High union rates of locking compression plating with cortical strut allograft for type B1 periprosthetic femoral fractures. Int Orthop. 2016. [Epub ahead of print]
31. Khashan M, Amar E, Drexler M, Chechik Ok, Cohen Z, Steinberg EL. Superior outcome of strut allograft-augmented plate fixation for the treatment of periprosthetic fractures around a stable femoral stem. Injury. 2013;44(11):1556-1560.
32. Old AB, McGrory BJ, White RR, Babikian GM. Fixation of Vancouver B1 peri-prosthetic fractures by broad metal plates without the application of strut allografts. J Bone Joint Surg Br. 2006;88(11):1425-1429.
33. Moore RE, Baldwin K, Austin MS, Mehta S. A systematic review of open reduction and internal fixation of periprosthetic femur fractures with or without allograft strut, cerclage, and locked plates. J Arthroplasty. 2014;29(5):872-876.
34. Lenz M, Stoffel K, Gueorguiev B, Klos K, Kielstein H, Hofmann GO. Enhancing fixation strength in periprosthetic femur fractures by orthogonal plating-a biomechanical study. J Orthop Res. 2016;34(4):591-596.
35. Choi JK, Gardner TR, Yoon E, Morrison TA, Macaulay WB, Geller JA. The effect of fixation technique on the stiffness of comminuted Vancouver B1 periprosthetic femur fractures. J Arthroplasty. 2010;25(6 Suppl):124-128.
36. Müller FJ, Galler M Füchtmeier B. Clinical and radiological results of patients treated with orthogonal double plating for periprosthetic femoral fractures. Int Orthop. 2014;38(12):2469-2472.
37. Pike J, Davidson D, Garbuz D, Duncan CP, O’Brien PJ, Masri BA. Principles of treatment for periprosthetic femoral shaft fractures around well-fixed total hip arthroplasty. J Am Acad Orthop Surg. 2009;17(11):677-688.
38. Serocki JH, Chandler RW, Dorr LD. Treatment of fractures about hip prostheses with compression plating. J Arthroplasty. 1992;7(2):129-135.
39. Pletka JD, Marsland D, Belkoff SM, Mears SC, Kates SL. Biomechanical comparison of 2 different locking plate fixation methods in vancouver b1 periprosthetic femur fractures. Geriatr Orthop Surg Rehabil. 2011;2(2):51-55.
40. Moloney GB, Westrick ER, Siska PA, Tarkin IS. Treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty implant: span the whole bone. Arch Orthop Trauma Surg. 2014;134(1):9-14.
41. Kubiak EN, Haller JM, Kemper DD, Presson AP, Higgins TF, Horowitz DS. Does the lateral plate need to overlap the stem to mitigate stress concentration when treating Vancouver C periprosthetic supracondylar femur fracture? J Arthroplasty. 2015;30(1):104-108.
42. Sah AP, Marshall A, Virkus WV, Estok DM 2nd, Della Valle CJ. Interprosthetic fractures of the femur: treatment with a single-locked plate. J Arthroplasty. 2010;25(2):280-286.
43. Hoffmann MF, Lotzien S, Schildhauer TA. Clinical outcome of interprosthetic femoral fractures treated with polyaxial locking plates. Injury. 2016. [Epub ahead of print]
44. Holder N, Papp S, Gofton W, Beaulé PE. Outcomes following surgical treatment of periprosthetic femur fractures: a single centre series. Can J Surg. 2014;57(3):209-213.
45. Crockarell JR Jr, Berry DJ, Lewallen DG. Nonunion after periprosthetic femoral fracture associated with total hip arthroplasty. J Bone Joint Surg Am. 1999;81(8):1073-1079.
1. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
2. Lewallen DG, Berry DJ. Periprosthetic fracture of the femur after total hip arthroplasty: treatment and results to date. Instr Course Lect. 1998;47:243-249.
3. Kavanagh BF. Femoral fractures associated with total hip arthroplasty. Orthop Clin North Am. 1992;23(2):249-257.
4. Meek RM, Norwood T, Smith R, Brenkel IJ, Howie CR. The risk of peri-prosthetic fracture after primary and revision total hip and knee replacement. J Bone Joint Surg Br. 2011;93(1):96-101.
5. Abdel MP, Watts CD, Houdek MT, Lewallen DG, Berry DJ. Epidemiology of periprosthetic fracture of the femur in 32 644 primary total hip arthroplasties: a 40-year experience. Bone Joint J. 2016;98-B(4):461-467.
6. Berry DJ. Epidemiology: hip and knee. Orthop Clin North Am. 1999;30(2):183-190.
7. Abdel MP, Houdek MT, Watts CD, Lewallen DG, Berry DJ. Epidemiology of periprosthetic femoral fractures in 5417 revision total hip arthrolasties: a 40-year experience. Bone Joint J. 2016;98-B(4):468-474.
8. Drew JM, Griffin WL, Odum SM, Van Doren B, Weston BT, Stryker LS. Survivorship after periprosthetic femur fracture: factors affecting outcome. J Arthroplasty. 2015. [Epub ahead of print]
9. Dehghan N, McKee MD, Nauth A, Ristevski B, Schemitsch EH. Surgical fixation of Vancouver type B1 periprosthetic femur fractures: a systematic review. J Orthop Trauma. 2014;28(12):721-727.
10. Mukundan C, Rayan F, Kheir E, Macdonald D. Management of late periprosthetic femur fractures: a retrospective cohort of 72 patients. Int Orthop. 2010;34(4):485-489.
11. Lindahl H, Malchau H, Odén A, Garellick G. Risk factors for failure after treatment of a periprosthetic fracture of the femur. J Bone Joint Surg Br. 2006;88(1):26-30.
12. Froberg L, Troelsen A, Brix M. Periprosthetic Vancouver type B1 and C fractures treated by locking-plate osteosynthesis: fracture union and reoperations in 60 consecutive fractures. Acta Orthop. 2012;83(6):648-652.
13. Beals RK, Tower SS. Periprosthetic fractures of the femur. An analysis of 93 fractures. Clin Orthop Relat Res. 1996(327):238-246.
14. Perren SM. Evolution of the internal fixation of long bone fractures. The scientific basis of biological internal fixation: choosing a new balance between stability and biology. J Bone Joint Surg Br. 2002;84(8):1093-1110.
15. Fulkerson E, Koval K, Preston CF, Iesaka K, Kummer FJ, Egol KA. Fixation of periprosthetic femoral shaft fractures associated with cemented femoral stems: a biomechanical comparison of locked plating and conventional cable plates. J Orthop Trauma. 2006;20(2):89-93.
16. Stoffel K, Sommer C, Kalampoki V, Blumenthal A, Joeris A. The influence of the operation technique and implant used in the treatment of periprosthetic hip and interprosthetic femur fractures: a systematic literature review of 1571 cases. Arch Orthop Trauma Surg. 2016;136(4):553-561.
17. Fulkerson E, Tejwani N, Stuchin S, Egol K. Management of periprosthetic femur fractures with a first generation locking plate. Injury. 2007;38(8):965-972.
18. Ricci WM, Bolhofner BR, Loftus T, Cox C, Mitchell S, Borrelli J Jr. Indirect reduction and plate fixation, without grafting, for periprosthetic femoral shaft fractures about a stable intramedullary implant. Surgical technique. J Bone Joint Surg Am. 2006;88 Suppl 1 Pt 2:275-282.
19. Ebraheim NA, Gomez C, Ramineni SK, Liu J. Fixation of periprosthetic femoral shaft fractures adjacent to a well-fixed femoral stem with reversed distal femoral locking plate. J Trauma. 2009;66(4):1152-1157.
20. Bryant GK, Morshed S, Agel J, et al. Isolated locked compression plating for Vancouver Type B1 periprosthetic femoral fractures. Injury. 2009;40(11):1180-1186.
21. Giesinger K, Ebneter L, Day RE, Stoffel KK, Yates PJ, Kuster MS. Can plate osteosynthesis of periprosthethic femoral fractures cause cement mantle failure around a stable hip stem? A biomechanical analysis. J Arthroplasty. 2014;29(6):1308-1312.
22. Lewis GS, Caroom CT, Wee H, et al. Tangential bicortical locked fixation improves stability in vancouver B1 periprosthetic femur fractures: a biomechanical study. J Orthop Trauma. 2015;29(10):e364-e370.
23. Lenz M, Perren SM, Gueorguiev B, et al. A biomechanical study on proximal plate fixation techniques in periprosthetic femur fractures. Injury. 2014;45 Suppl 1:S71-S75.
24. Dennis MG, Simon JA, Kummer FJ, Koval KJ, DiCesare PE. Fixation of periprosthetic femoral shaft fractures occurring at the tip of the stem: a biomechanical study of 5 techniques. J Arthroplasty. 2000;15(4):523-528.
25. Graham SM, Mak JH, Moazen M, et al. Periprosthetic femoral fracture fixation: a biomechanical comparison between proximal locking screws and cables. J Orthop Sci. 2015;20(5):875-880.
26. Griffiths JT, Taheri A, Day RE, Yates PJ. Better axial stiffness of a bicortical screw construct compared to a cable construct for comminuted Vancouver B1 proximal femoral fractures. J Arthroplasty. 2015;30(12):2333-2337.
27. Chandler HP, King D, Limbird R, et al. The use of cortical allograft struts for fixation of fractures associated with well-fixed total joint prostheses. Semin Arthroplasty. 1993;4(2):99-107.
28. Haddad FS, Duncan CP, Berry DJ, Lewallen DG, Gross AE, Chandler HP. Periprosthetic femoral fractures around well-fixed implants: use of cortical onlay allografts with or without a plate. J Bone Joint Surg Am. 2002;84-A(6):945-950.
29. Dennis MG, Simon JA, Kummer FJ, Koval KJ, Di Cesare PE. Fixation of periprosthetic femoral shaft fractures: a biomechanical comparison of two techniques. J Orthop Trauma. 2001;15(3):177-180.
30. Yeo I, Rhyu KH, Kim SM, Park YS, Lim SJ. High union rates of locking compression plating with cortical strut allograft for type B1 periprosthetic femoral fractures. Int Orthop. 2016. [Epub ahead of print]
31. Khashan M, Amar E, Drexler M, Chechik Ok, Cohen Z, Steinberg EL. Superior outcome of strut allograft-augmented plate fixation for the treatment of periprosthetic fractures around a stable femoral stem. Injury. 2013;44(11):1556-1560.
32. Old AB, McGrory BJ, White RR, Babikian GM. Fixation of Vancouver B1 peri-prosthetic fractures by broad metal plates without the application of strut allografts. J Bone Joint Surg Br. 2006;88(11):1425-1429.
33. Moore RE, Baldwin K, Austin MS, Mehta S. A systematic review of open reduction and internal fixation of periprosthetic femur fractures with or without allograft strut, cerclage, and locked plates. J Arthroplasty. 2014;29(5):872-876.
34. Lenz M, Stoffel K, Gueorguiev B, Klos K, Kielstein H, Hofmann GO. Enhancing fixation strength in periprosthetic femur fractures by orthogonal plating-a biomechanical study. J Orthop Res. 2016;34(4):591-596.
35. Choi JK, Gardner TR, Yoon E, Morrison TA, Macaulay WB, Geller JA. The effect of fixation technique on the stiffness of comminuted Vancouver B1 periprosthetic femur fractures. J Arthroplasty. 2010;25(6 Suppl):124-128.
36. Müller FJ, Galler M Füchtmeier B. Clinical and radiological results of patients treated with orthogonal double plating for periprosthetic femoral fractures. Int Orthop. 2014;38(12):2469-2472.
37. Pike J, Davidson D, Garbuz D, Duncan CP, O’Brien PJ, Masri BA. Principles of treatment for periprosthetic femoral shaft fractures around well-fixed total hip arthroplasty. J Am Acad Orthop Surg. 2009;17(11):677-688.
38. Serocki JH, Chandler RW, Dorr LD. Treatment of fractures about hip prostheses with compression plating. J Arthroplasty. 1992;7(2):129-135.
39. Pletka JD, Marsland D, Belkoff SM, Mears SC, Kates SL. Biomechanical comparison of 2 different locking plate fixation methods in vancouver b1 periprosthetic femur fractures. Geriatr Orthop Surg Rehabil. 2011;2(2):51-55.
40. Moloney GB, Westrick ER, Siska PA, Tarkin IS. Treatment of periprosthetic femur fractures around a well-fixed hip arthroplasty implant: span the whole bone. Arch Orthop Trauma Surg. 2014;134(1):9-14.
41. Kubiak EN, Haller JM, Kemper DD, Presson AP, Higgins TF, Horowitz DS. Does the lateral plate need to overlap the stem to mitigate stress concentration when treating Vancouver C periprosthetic supracondylar femur fracture? J Arthroplasty. 2015;30(1):104-108.
42. Sah AP, Marshall A, Virkus WV, Estok DM 2nd, Della Valle CJ. Interprosthetic fractures of the femur: treatment with a single-locked plate. J Arthroplasty. 2010;25(2):280-286.
43. Hoffmann MF, Lotzien S, Schildhauer TA. Clinical outcome of interprosthetic femoral fractures treated with polyaxial locking plates. Injury. 2016. [Epub ahead of print]
44. Holder N, Papp S, Gofton W, Beaulé PE. Outcomes following surgical treatment of periprosthetic femur fractures: a single centre series. Can J Surg. 2014;57(3):209-213.
45. Crockarell JR Jr, Berry DJ, Lewallen DG. Nonunion after periprosthetic femoral fracture associated with total hip arthroplasty. J Bone Joint Surg Am. 1999;81(8):1073-1079.