User login
Lipidologists welcome bempedoic acid as new lipid-lowering option
Bempedoic acid, the first agent in a new class of drugs that reduce LDL cholesterol, received Food and Drug Administration approval on Feb. 21 for treating selected hypercholesterolemic patients and is a welcome addition to the medicine cabinet, say lipid experts.
However, it is a tertiary option at least until results from a 14,000 patient clinical-outcome trial of bempedoic acid come out, likely in 2022, they agreed.
“I’m excited to have a new tool in the toolkit for treating high-risk patients, but I will always reach first for the drugs proven to reduce clinical outcomes,” said Erin D. Michos, MD, director of Women’s Cardiovascular Health and associate director of Preventive Cardiology at Johns Hopkins Medicine in Baltimore. That sentiment, shared by other experts, should for the time being relegate bempedoic acid (Nexletol) to a backup role behind statins, ezetimibe, and the PCSK9 inhibitor antibodies that are all now on the U.S. market and all buttressed with evidence of their ability to cut cardiovascular disease death and other CVD outcomes from large outcome studies.
The existing evidence base for bempedoic acid rests primarily two multicenter, randomized, placebo-controlled clinical trials of bempedoic acid in patients with LDL levels above 70 mg/dL while on maximally tolerated lipid-lowering therapy. In CLEAR Harmony, results showed that treatment with bempedoic acid cut LDL-cholesterol levels by an average of 18% more compared with placebo (N Engl J Med 2019;380:1022-32). In CLEAR Wisdom, bempedoic acid reduced LDL cholesterol levels by 17% (JAMA. 2019;322[18]:1780-8).
While those two trials proved the drug’s ability to lower levels of LDL cholesterol, they lacked the power to address whether this effect cut the incidence of CVD events, a question that the CLEAR Outcomes trial aims to answer.
“I believe in the lipid hypothesis, but the main thing we need to see is whether bempedoic acid leads to a meaningful reduction in CVD events. The window for bempedoic acid will remain narrow until we see the outcomes results,” Dr. Michos said in an interview.
Bempedoic acid is a prodrug that’s activated in liver and targets the same cholesterol synthesis pathway as statins by inhibition of ATP-citrate lyase, an enzyme that’s upstream of HMG-CoA reductase, thereby enhancing LDL cholesterol clearance via up-regulation of LDL receptors
.
“,” said Jennifer G. Robinson, MD, professor of epidemiology and medicine and director of the Prevention Intervention Center of the University of Iowa in Iowa City. That would be just a portion of the newly labeled target population. The FDA’s approved label for bempedoic acid cites the drug as an “adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional lowering of LDL-C.”
The current lack of outcomes evidence for bempedoic acid was not an issue for Robert H. Eckel, MD, an endocrinologist and lipid management specialist at the University of Colorado at Denver in Aurora. Having results from CLEAR Outcomes “may be helpful, but LDL cholesterol lowering in the range where the FDA has indicated using bempedoic acid seems all we need for now,” he said in an interview. Viewing bempedoic acid as potentially useful for both HeFH and ASCVD patients, Dr. Eckel particularly cited the possibility of using the new drug in combination with ezetimibe, another oral, once-daily agent with a moderate but additive effect for cutting LDL cholesterol.
Combined treatment with bempedoic acid and ezetimibe “may be successful in avoiding [using] a PCSK9 inhibitor in some patients, and in particular patients with HeFH or those who are statin intolerant.” But like his colleagues, Dr. Eckel agreed that, for the moment, ezetimibe has an edge over bempedoic acid because of its more extensive evidence base. “If the combination of bempedoic acid and ezetimibe is not needed, the decision [of which one of these to use] needs to depend on the outcome trial results for ezetimibe,” he said. Other factors clinicians could apply if faced with choosing between these two agents include the significant reduction in high-sensitivity C-reactive protein that bempedoic acid produces; the downside that bempedoic acid can cause in some patients an early and persistent rise in serum uric acid levels that can trigger gout flares in patients with a history of gout or at risk for gout; and cost, he said.
Cost is the room-dwelling elephant that colors many decisions about which lipid-lowering drug to use for patients, with options running the price gamut from the generic and uniformly affordable statins and ezetimibe, to the notoriously pricey PCSK9 inhibitors that remain for many patients either prohibitively expensive or hard to get covered by some insurers. Bempedoic acid seems on track to fall somewhere between these two poles, although staff members from Esperion, the company that developed and will market bempedoic acid as Nexletol starting on March 30, declared in a conference call on Feb. 24 that “cost will not be an issue,” for indicated patients prescribed the drug. Company representatives cited a program of coupons, discounts, and rebates they have planned that they anticipate will allow patients who meet the labeled indications to have an out-of-pocket cost for bempedoic acid of “as low as” $10 for a 90-pill supply. They also noted their goal of getting bempedoic acid onto the lowest tier of the Medicare formulary.
How these steps actually play out in the fun house of U.S. prescription drug pricing and preauthorizations remains to be seen. “Out-of-pocket costs are not the real drivers” of drug access, noted Dr. Robinson. “Insurers will likely start with restricted access and prior authorization requirements, just as they did with ezetimibe when it was on patent and prior to having the results from a CVD outcomes trial.” For the time being, bempedoic acid can generally be seen as “expensive ezetimibe,” summed up Dr. Robinson.
Despite that somewhat dismissive characterization, experts are intrigued by the possibility of combining two moderately potent, oral, and safe lipid-lowering drugs in selected patients as a potential alternative to the still financially challenging PCSK9 inhibitors. Combining bempedoic acid and ezetimibe “has a lot of appeal,” said Dr. Michos. “Even though preauthorization has gotten better, it’s still a challenge to get a PCSK9 inhibitor approved.”
Much of her enthusiasm stems from a study reported last year that randomized 301 patients to treatment with bempedoic acid, ezetimibe, or both. The results showed that combined treatment has a similar safety profile to treatment with either drug alone, and produced a cut in LDL cholesterol that was roughly additive for the reductions produced by each drug by itself: Ezetimibe alone cut LDL by about 23%, bempedoic acid alone by about 17%, and the two dosed together once daily resulted in an average 36% drop (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864671). The results showed that, “in patients requiring intensive LDL cholesterol lowering, who cannot afford PCSK9 inhibitors, or have statin intolerance, bempedoic acid and ezetimibe are stronger together and can serve as an alternative approach for lipid management in ASCVD prevention,” wrote Dr. Michos and a coauthor in a commentary that appeared with the study results (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864672).
The concept of combined bempedoic acid and ezetimibe treatment is so appealing that the bempedoic acid manufacturer, Esperion, has already developed a single-pill formulation of the two drugs that received FDA marketing approval on February 26. A company statement said that marketing of this combined formulation, Nexlizet, will start in July 2020.
Although interest in bempedoic acid seems running high for patients included in the new FDA indication, Dr. Michos and others see possibly greater potential for what would now be off-label use for primary prevention in high-risk patients without HeFH, patients who generally don’t qualify for insurance coverage of a PCSK9 inhibitor.
“Use in primary prevention in [non-HeFH] patients with insufficient lowering of LDL cholesterol wouldn’t surprise me,” but a big concern will be out-of-pocket cost when off-label use precludes insurance coverage or discount eligibility, noted Dr. Eckel. An Esperion spokesperson said that the undiscounted, wholesale acquisition cost for bempedoic acid is expected to be roughly $10/pill, or about $300 for a 30-day supply, positioning it more or less midway between generic statins and ezetimibe and the list price for a PCSk9 inhibitor of roughly $500/month.
“I’m most excited about bempedoic acid in the off-label space, for patients who can’t get approved for a PCSK9 inhibitor, for treating patients with subclinical ASCVD, or really high-risk patients with multiple risk factors including diabetes,” especially when these patients are intolerant of a high-intensity statin regimen, said Dr. Michos. “I have a clinic full of patients” who can’t take their full, indicated dosage of a high-intensity statin, and when those patients also can’t get on treatment with a PCSK9 inhibitor then bempedoic acid will be an important part of their alternative regimen, she explained.
Dr. Michos had no disclosures. Dr. Robinson has received research funding from Esperion and from several other companies, and she has been a consultant to Amgen, Merck, Novartis, Novo Nordisk, Pfizer, Regeneron, and Sanofi. Dr. Eckel has received honoraria from Kowa, Merck, Novo Nordisk, and Sanofi/Regeneron.
This article was updated 2/27/20.
Bempedoic acid, the first agent in a new class of drugs that reduce LDL cholesterol, received Food and Drug Administration approval on Feb. 21 for treating selected hypercholesterolemic patients and is a welcome addition to the medicine cabinet, say lipid experts.
However, it is a tertiary option at least until results from a 14,000 patient clinical-outcome trial of bempedoic acid come out, likely in 2022, they agreed.
“I’m excited to have a new tool in the toolkit for treating high-risk patients, but I will always reach first for the drugs proven to reduce clinical outcomes,” said Erin D. Michos, MD, director of Women’s Cardiovascular Health and associate director of Preventive Cardiology at Johns Hopkins Medicine in Baltimore. That sentiment, shared by other experts, should for the time being relegate bempedoic acid (Nexletol) to a backup role behind statins, ezetimibe, and the PCSK9 inhibitor antibodies that are all now on the U.S. market and all buttressed with evidence of their ability to cut cardiovascular disease death and other CVD outcomes from large outcome studies.
The existing evidence base for bempedoic acid rests primarily two multicenter, randomized, placebo-controlled clinical trials of bempedoic acid in patients with LDL levels above 70 mg/dL while on maximally tolerated lipid-lowering therapy. In CLEAR Harmony, results showed that treatment with bempedoic acid cut LDL-cholesterol levels by an average of 18% more compared with placebo (N Engl J Med 2019;380:1022-32). In CLEAR Wisdom, bempedoic acid reduced LDL cholesterol levels by 17% (JAMA. 2019;322[18]:1780-8).
While those two trials proved the drug’s ability to lower levels of LDL cholesterol, they lacked the power to address whether this effect cut the incidence of CVD events, a question that the CLEAR Outcomes trial aims to answer.
“I believe in the lipid hypothesis, but the main thing we need to see is whether bempedoic acid leads to a meaningful reduction in CVD events. The window for bempedoic acid will remain narrow until we see the outcomes results,” Dr. Michos said in an interview.
Bempedoic acid is a prodrug that’s activated in liver and targets the same cholesterol synthesis pathway as statins by inhibition of ATP-citrate lyase, an enzyme that’s upstream of HMG-CoA reductase, thereby enhancing LDL cholesterol clearance via up-regulation of LDL receptors
.
“,” said Jennifer G. Robinson, MD, professor of epidemiology and medicine and director of the Prevention Intervention Center of the University of Iowa in Iowa City. That would be just a portion of the newly labeled target population. The FDA’s approved label for bempedoic acid cites the drug as an “adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional lowering of LDL-C.”
The current lack of outcomes evidence for bempedoic acid was not an issue for Robert H. Eckel, MD, an endocrinologist and lipid management specialist at the University of Colorado at Denver in Aurora. Having results from CLEAR Outcomes “may be helpful, but LDL cholesterol lowering in the range where the FDA has indicated using bempedoic acid seems all we need for now,” he said in an interview. Viewing bempedoic acid as potentially useful for both HeFH and ASCVD patients, Dr. Eckel particularly cited the possibility of using the new drug in combination with ezetimibe, another oral, once-daily agent with a moderate but additive effect for cutting LDL cholesterol.
Combined treatment with bempedoic acid and ezetimibe “may be successful in avoiding [using] a PCSK9 inhibitor in some patients, and in particular patients with HeFH or those who are statin intolerant.” But like his colleagues, Dr. Eckel agreed that, for the moment, ezetimibe has an edge over bempedoic acid because of its more extensive evidence base. “If the combination of bempedoic acid and ezetimibe is not needed, the decision [of which one of these to use] needs to depend on the outcome trial results for ezetimibe,” he said. Other factors clinicians could apply if faced with choosing between these two agents include the significant reduction in high-sensitivity C-reactive protein that bempedoic acid produces; the downside that bempedoic acid can cause in some patients an early and persistent rise in serum uric acid levels that can trigger gout flares in patients with a history of gout or at risk for gout; and cost, he said.
Cost is the room-dwelling elephant that colors many decisions about which lipid-lowering drug to use for patients, with options running the price gamut from the generic and uniformly affordable statins and ezetimibe, to the notoriously pricey PCSK9 inhibitors that remain for many patients either prohibitively expensive or hard to get covered by some insurers. Bempedoic acid seems on track to fall somewhere between these two poles, although staff members from Esperion, the company that developed and will market bempedoic acid as Nexletol starting on March 30, declared in a conference call on Feb. 24 that “cost will not be an issue,” for indicated patients prescribed the drug. Company representatives cited a program of coupons, discounts, and rebates they have planned that they anticipate will allow patients who meet the labeled indications to have an out-of-pocket cost for bempedoic acid of “as low as” $10 for a 90-pill supply. They also noted their goal of getting bempedoic acid onto the lowest tier of the Medicare formulary.
How these steps actually play out in the fun house of U.S. prescription drug pricing and preauthorizations remains to be seen. “Out-of-pocket costs are not the real drivers” of drug access, noted Dr. Robinson. “Insurers will likely start with restricted access and prior authorization requirements, just as they did with ezetimibe when it was on patent and prior to having the results from a CVD outcomes trial.” For the time being, bempedoic acid can generally be seen as “expensive ezetimibe,” summed up Dr. Robinson.
Despite that somewhat dismissive characterization, experts are intrigued by the possibility of combining two moderately potent, oral, and safe lipid-lowering drugs in selected patients as a potential alternative to the still financially challenging PCSK9 inhibitors. Combining bempedoic acid and ezetimibe “has a lot of appeal,” said Dr. Michos. “Even though preauthorization has gotten better, it’s still a challenge to get a PCSK9 inhibitor approved.”
Much of her enthusiasm stems from a study reported last year that randomized 301 patients to treatment with bempedoic acid, ezetimibe, or both. The results showed that combined treatment has a similar safety profile to treatment with either drug alone, and produced a cut in LDL cholesterol that was roughly additive for the reductions produced by each drug by itself: Ezetimibe alone cut LDL by about 23%, bempedoic acid alone by about 17%, and the two dosed together once daily resulted in an average 36% drop (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864671). The results showed that, “in patients requiring intensive LDL cholesterol lowering, who cannot afford PCSK9 inhibitors, or have statin intolerance, bempedoic acid and ezetimibe are stronger together and can serve as an alternative approach for lipid management in ASCVD prevention,” wrote Dr. Michos and a coauthor in a commentary that appeared with the study results (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864672).
The concept of combined bempedoic acid and ezetimibe treatment is so appealing that the bempedoic acid manufacturer, Esperion, has already developed a single-pill formulation of the two drugs that received FDA marketing approval on February 26. A company statement said that marketing of this combined formulation, Nexlizet, will start in July 2020.
Although interest in bempedoic acid seems running high for patients included in the new FDA indication, Dr. Michos and others see possibly greater potential for what would now be off-label use for primary prevention in high-risk patients without HeFH, patients who generally don’t qualify for insurance coverage of a PCSK9 inhibitor.
“Use in primary prevention in [non-HeFH] patients with insufficient lowering of LDL cholesterol wouldn’t surprise me,” but a big concern will be out-of-pocket cost when off-label use precludes insurance coverage or discount eligibility, noted Dr. Eckel. An Esperion spokesperson said that the undiscounted, wholesale acquisition cost for bempedoic acid is expected to be roughly $10/pill, or about $300 for a 30-day supply, positioning it more or less midway between generic statins and ezetimibe and the list price for a PCSk9 inhibitor of roughly $500/month.
“I’m most excited about bempedoic acid in the off-label space, for patients who can’t get approved for a PCSK9 inhibitor, for treating patients with subclinical ASCVD, or really high-risk patients with multiple risk factors including diabetes,” especially when these patients are intolerant of a high-intensity statin regimen, said Dr. Michos. “I have a clinic full of patients” who can’t take their full, indicated dosage of a high-intensity statin, and when those patients also can’t get on treatment with a PCSK9 inhibitor then bempedoic acid will be an important part of their alternative regimen, she explained.
Dr. Michos had no disclosures. Dr. Robinson has received research funding from Esperion and from several other companies, and she has been a consultant to Amgen, Merck, Novartis, Novo Nordisk, Pfizer, Regeneron, and Sanofi. Dr. Eckel has received honoraria from Kowa, Merck, Novo Nordisk, and Sanofi/Regeneron.
This article was updated 2/27/20.
Bempedoic acid, the first agent in a new class of drugs that reduce LDL cholesterol, received Food and Drug Administration approval on Feb. 21 for treating selected hypercholesterolemic patients and is a welcome addition to the medicine cabinet, say lipid experts.
However, it is a tertiary option at least until results from a 14,000 patient clinical-outcome trial of bempedoic acid come out, likely in 2022, they agreed.
“I’m excited to have a new tool in the toolkit for treating high-risk patients, but I will always reach first for the drugs proven to reduce clinical outcomes,” said Erin D. Michos, MD, director of Women’s Cardiovascular Health and associate director of Preventive Cardiology at Johns Hopkins Medicine in Baltimore. That sentiment, shared by other experts, should for the time being relegate bempedoic acid (Nexletol) to a backup role behind statins, ezetimibe, and the PCSK9 inhibitor antibodies that are all now on the U.S. market and all buttressed with evidence of their ability to cut cardiovascular disease death and other CVD outcomes from large outcome studies.
The existing evidence base for bempedoic acid rests primarily two multicenter, randomized, placebo-controlled clinical trials of bempedoic acid in patients with LDL levels above 70 mg/dL while on maximally tolerated lipid-lowering therapy. In CLEAR Harmony, results showed that treatment with bempedoic acid cut LDL-cholesterol levels by an average of 18% more compared with placebo (N Engl J Med 2019;380:1022-32). In CLEAR Wisdom, bempedoic acid reduced LDL cholesterol levels by 17% (JAMA. 2019;322[18]:1780-8).
While those two trials proved the drug’s ability to lower levels of LDL cholesterol, they lacked the power to address whether this effect cut the incidence of CVD events, a question that the CLEAR Outcomes trial aims to answer.
“I believe in the lipid hypothesis, but the main thing we need to see is whether bempedoic acid leads to a meaningful reduction in CVD events. The window for bempedoic acid will remain narrow until we see the outcomes results,” Dr. Michos said in an interview.
Bempedoic acid is a prodrug that’s activated in liver and targets the same cholesterol synthesis pathway as statins by inhibition of ATP-citrate lyase, an enzyme that’s upstream of HMG-CoA reductase, thereby enhancing LDL cholesterol clearance via up-regulation of LDL receptors
.
“,” said Jennifer G. Robinson, MD, professor of epidemiology and medicine and director of the Prevention Intervention Center of the University of Iowa in Iowa City. That would be just a portion of the newly labeled target population. The FDA’s approved label for bempedoic acid cites the drug as an “adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional lowering of LDL-C.”
The current lack of outcomes evidence for bempedoic acid was not an issue for Robert H. Eckel, MD, an endocrinologist and lipid management specialist at the University of Colorado at Denver in Aurora. Having results from CLEAR Outcomes “may be helpful, but LDL cholesterol lowering in the range where the FDA has indicated using bempedoic acid seems all we need for now,” he said in an interview. Viewing bempedoic acid as potentially useful for both HeFH and ASCVD patients, Dr. Eckel particularly cited the possibility of using the new drug in combination with ezetimibe, another oral, once-daily agent with a moderate but additive effect for cutting LDL cholesterol.
Combined treatment with bempedoic acid and ezetimibe “may be successful in avoiding [using] a PCSK9 inhibitor in some patients, and in particular patients with HeFH or those who are statin intolerant.” But like his colleagues, Dr. Eckel agreed that, for the moment, ezetimibe has an edge over bempedoic acid because of its more extensive evidence base. “If the combination of bempedoic acid and ezetimibe is not needed, the decision [of which one of these to use] needs to depend on the outcome trial results for ezetimibe,” he said. Other factors clinicians could apply if faced with choosing between these two agents include the significant reduction in high-sensitivity C-reactive protein that bempedoic acid produces; the downside that bempedoic acid can cause in some patients an early and persistent rise in serum uric acid levels that can trigger gout flares in patients with a history of gout or at risk for gout; and cost, he said.
Cost is the room-dwelling elephant that colors many decisions about which lipid-lowering drug to use for patients, with options running the price gamut from the generic and uniformly affordable statins and ezetimibe, to the notoriously pricey PCSK9 inhibitors that remain for many patients either prohibitively expensive or hard to get covered by some insurers. Bempedoic acid seems on track to fall somewhere between these two poles, although staff members from Esperion, the company that developed and will market bempedoic acid as Nexletol starting on March 30, declared in a conference call on Feb. 24 that “cost will not be an issue,” for indicated patients prescribed the drug. Company representatives cited a program of coupons, discounts, and rebates they have planned that they anticipate will allow patients who meet the labeled indications to have an out-of-pocket cost for bempedoic acid of “as low as” $10 for a 90-pill supply. They also noted their goal of getting bempedoic acid onto the lowest tier of the Medicare formulary.
How these steps actually play out in the fun house of U.S. prescription drug pricing and preauthorizations remains to be seen. “Out-of-pocket costs are not the real drivers” of drug access, noted Dr. Robinson. “Insurers will likely start with restricted access and prior authorization requirements, just as they did with ezetimibe when it was on patent and prior to having the results from a CVD outcomes trial.” For the time being, bempedoic acid can generally be seen as “expensive ezetimibe,” summed up Dr. Robinson.
Despite that somewhat dismissive characterization, experts are intrigued by the possibility of combining two moderately potent, oral, and safe lipid-lowering drugs in selected patients as a potential alternative to the still financially challenging PCSK9 inhibitors. Combining bempedoic acid and ezetimibe “has a lot of appeal,” said Dr. Michos. “Even though preauthorization has gotten better, it’s still a challenge to get a PCSK9 inhibitor approved.”
Much of her enthusiasm stems from a study reported last year that randomized 301 patients to treatment with bempedoic acid, ezetimibe, or both. The results showed that combined treatment has a similar safety profile to treatment with either drug alone, and produced a cut in LDL cholesterol that was roughly additive for the reductions produced by each drug by itself: Ezetimibe alone cut LDL by about 23%, bempedoic acid alone by about 17%, and the two dosed together once daily resulted in an average 36% drop (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864671). The results showed that, “in patients requiring intensive LDL cholesterol lowering, who cannot afford PCSK9 inhibitors, or have statin intolerance, bempedoic acid and ezetimibe are stronger together and can serve as an alternative approach for lipid management in ASCVD prevention,” wrote Dr. Michos and a coauthor in a commentary that appeared with the study results (Eur J Prev Cardiol. 2019 Jul 29. doi: 10.1177/2047487319864672).
The concept of combined bempedoic acid and ezetimibe treatment is so appealing that the bempedoic acid manufacturer, Esperion, has already developed a single-pill formulation of the two drugs that received FDA marketing approval on February 26. A company statement said that marketing of this combined formulation, Nexlizet, will start in July 2020.
Although interest in bempedoic acid seems running high for patients included in the new FDA indication, Dr. Michos and others see possibly greater potential for what would now be off-label use for primary prevention in high-risk patients without HeFH, patients who generally don’t qualify for insurance coverage of a PCSK9 inhibitor.
“Use in primary prevention in [non-HeFH] patients with insufficient lowering of LDL cholesterol wouldn’t surprise me,” but a big concern will be out-of-pocket cost when off-label use precludes insurance coverage or discount eligibility, noted Dr. Eckel. An Esperion spokesperson said that the undiscounted, wholesale acquisition cost for bempedoic acid is expected to be roughly $10/pill, or about $300 for a 30-day supply, positioning it more or less midway between generic statins and ezetimibe and the list price for a PCSk9 inhibitor of roughly $500/month.
“I’m most excited about bempedoic acid in the off-label space, for patients who can’t get approved for a PCSK9 inhibitor, for treating patients with subclinical ASCVD, or really high-risk patients with multiple risk factors including diabetes,” especially when these patients are intolerant of a high-intensity statin regimen, said Dr. Michos. “I have a clinic full of patients” who can’t take their full, indicated dosage of a high-intensity statin, and when those patients also can’t get on treatment with a PCSK9 inhibitor then bempedoic acid will be an important part of their alternative regimen, she explained.
Dr. Michos had no disclosures. Dr. Robinson has received research funding from Esperion and from several other companies, and she has been a consultant to Amgen, Merck, Novartis, Novo Nordisk, Pfizer, Regeneron, and Sanofi. Dr. Eckel has received honoraria from Kowa, Merck, Novo Nordisk, and Sanofi/Regeneron.
This article was updated 2/27/20.
Study implicates gut bacteria in PAH
Model finds microbiota highly predictive
A unique collection of bacteria in the gut may have a strong association with pulmonary arterial hypertension and could be highly predictive of the disease in undiagnosed patients, according to a study published in the journal Hypertension.
This is the first study to show that people with PAH have a common specific gut microbiota profile, wrote lead study author Mohan Raizada, PhD, distinguished professor in the department of physiology and functional genomics at the University of Florida, Gainesville.
The findings have the potential to change how cardiologists diagnose and treat PAH, he added. “While current PAH treatments focus on the lungs, looking at the lung/gut axis could open the door to new therapies centered in the digestive system,” Dr. Raizada said.
The researchers developed a model that found the specific microbiota profile was 83% accurate in predicting the presence or absence of PAH. If a larger study can validate the findings, the researchers wrote, this could lead to a new test for diagnosing PAH that’s less invasive than cardiac catheterization. It could also lead to new treatments that target the gut microbiome.
Study investigators collected stool samples from 18 PAH patients and 12 people without a history of cardiopulmonary disease. The microbiota DNA from the stool samples were isolated and sequenced. The analysis revealed that PAH patients had reduced richness and evenness of the gut bacteria, known as alpha diversity. They had increased levels of bacteria associated with atherosclerosis, and healthy patients had increased levels of bacteria that produced short-chain fatty acids.
Although recent studies have begun to show potential associations between the gut microbiome and cardiovascular diseases, this research is in its infancy, Mariell Jessup, MD, commented. “Even though the study by Dr. Raizada and colleagues predicted pulmonary arterial hypertension based on an individual’s microbiome with some accuracy, it is an observational study, so it does not prove cause and effect. Many other factors, especially diet, affect the gut microbiome,” added Dr. Jessup, Chief Science and Medical Officer for the American Heart Association.
She stressed that, “In addition, even if studies confirm an association between the gut microbiome and cardiovascular diseases such as PAH, more research is needed to determine if improving gut microbiota could directly impact PAH or other cardiovascular diseases. The findings of this study will not impact clinical practice.”
Dr. Raizada and his coinvestigators offered two possible mechanisms through which the gut microbiome influences pulmonary physiology. One is that lower levels of bacteria that produce the short-chain fatty acid butyrate, such as Coprococcus, Butyrivibrio, Lachnospiraceae, and Eubacterium, along with Clostridia in the gut of PAH patients, may increase gut permeability. Reduced butyrate weakens gut barrier function and can induce inflammation and leakage. This can allow microbial metabolites to enter the circulatory system, disrupting metabolism and immunity and affecting pulmonary vessels.
The second potential mechanism is that increased Collinsella in the PAH cohort may be the culprit that increases gut permeability, resulting in the ensuing gut barrier dysfunction and inflammation. The study noted Collinsella contributed most of the increased genes for the biosynthesis on the amino acid proline in these patients, and that a previous study implicated Collinsella and its parent, Cariobacteriales, in trimethylamine/trimethylamine N-oxide production (TMA/TMAO) in atherosclerosis (Cell. 2015;163[7]:1585-95). The non-PAH patients had higher levels of bacteria that had a low correlation with TMA/TMAO.
“We were very surprised to see such an association within a small group of study subjects,” wrote Dr. Raizada and associates. “It usually requires hundreds of patients to achieve such significance.”
More research is needed to determine if the specific microbiota associated with PAH causes the disease or is a result of it, they concluded.
The study was funded by grants from the National Institutes of Health, the NIH National Center for Research Resources, and the U.S. Department of Defense. Dr. Raizada and coauthors reported no relevant financial relationships.
SOURCE: Raizada MK et al. Hypertension. 2020. doi: 10.1161/HYPERTENSIONAHA.119.14294.
Model finds microbiota highly predictive
Model finds microbiota highly predictive
A unique collection of bacteria in the gut may have a strong association with pulmonary arterial hypertension and could be highly predictive of the disease in undiagnosed patients, according to a study published in the journal Hypertension.
This is the first study to show that people with PAH have a common specific gut microbiota profile, wrote lead study author Mohan Raizada, PhD, distinguished professor in the department of physiology and functional genomics at the University of Florida, Gainesville.
The findings have the potential to change how cardiologists diagnose and treat PAH, he added. “While current PAH treatments focus on the lungs, looking at the lung/gut axis could open the door to new therapies centered in the digestive system,” Dr. Raizada said.
The researchers developed a model that found the specific microbiota profile was 83% accurate in predicting the presence or absence of PAH. If a larger study can validate the findings, the researchers wrote, this could lead to a new test for diagnosing PAH that’s less invasive than cardiac catheterization. It could also lead to new treatments that target the gut microbiome.
Study investigators collected stool samples from 18 PAH patients and 12 people without a history of cardiopulmonary disease. The microbiota DNA from the stool samples were isolated and sequenced. The analysis revealed that PAH patients had reduced richness and evenness of the gut bacteria, known as alpha diversity. They had increased levels of bacteria associated with atherosclerosis, and healthy patients had increased levels of bacteria that produced short-chain fatty acids.
Although recent studies have begun to show potential associations between the gut microbiome and cardiovascular diseases, this research is in its infancy, Mariell Jessup, MD, commented. “Even though the study by Dr. Raizada and colleagues predicted pulmonary arterial hypertension based on an individual’s microbiome with some accuracy, it is an observational study, so it does not prove cause and effect. Many other factors, especially diet, affect the gut microbiome,” added Dr. Jessup, Chief Science and Medical Officer for the American Heart Association.
She stressed that, “In addition, even if studies confirm an association between the gut microbiome and cardiovascular diseases such as PAH, more research is needed to determine if improving gut microbiota could directly impact PAH or other cardiovascular diseases. The findings of this study will not impact clinical practice.”
Dr. Raizada and his coinvestigators offered two possible mechanisms through which the gut microbiome influences pulmonary physiology. One is that lower levels of bacteria that produce the short-chain fatty acid butyrate, such as Coprococcus, Butyrivibrio, Lachnospiraceae, and Eubacterium, along with Clostridia in the gut of PAH patients, may increase gut permeability. Reduced butyrate weakens gut barrier function and can induce inflammation and leakage. This can allow microbial metabolites to enter the circulatory system, disrupting metabolism and immunity and affecting pulmonary vessels.
The second potential mechanism is that increased Collinsella in the PAH cohort may be the culprit that increases gut permeability, resulting in the ensuing gut barrier dysfunction and inflammation. The study noted Collinsella contributed most of the increased genes for the biosynthesis on the amino acid proline in these patients, and that a previous study implicated Collinsella and its parent, Cariobacteriales, in trimethylamine/trimethylamine N-oxide production (TMA/TMAO) in atherosclerosis (Cell. 2015;163[7]:1585-95). The non-PAH patients had higher levels of bacteria that had a low correlation with TMA/TMAO.
“We were very surprised to see such an association within a small group of study subjects,” wrote Dr. Raizada and associates. “It usually requires hundreds of patients to achieve such significance.”
More research is needed to determine if the specific microbiota associated with PAH causes the disease or is a result of it, they concluded.
The study was funded by grants from the National Institutes of Health, the NIH National Center for Research Resources, and the U.S. Department of Defense. Dr. Raizada and coauthors reported no relevant financial relationships.
SOURCE: Raizada MK et al. Hypertension. 2020. doi: 10.1161/HYPERTENSIONAHA.119.14294.
A unique collection of bacteria in the gut may have a strong association with pulmonary arterial hypertension and could be highly predictive of the disease in undiagnosed patients, according to a study published in the journal Hypertension.
This is the first study to show that people with PAH have a common specific gut microbiota profile, wrote lead study author Mohan Raizada, PhD, distinguished professor in the department of physiology and functional genomics at the University of Florida, Gainesville.
The findings have the potential to change how cardiologists diagnose and treat PAH, he added. “While current PAH treatments focus on the lungs, looking at the lung/gut axis could open the door to new therapies centered in the digestive system,” Dr. Raizada said.
The researchers developed a model that found the specific microbiota profile was 83% accurate in predicting the presence or absence of PAH. If a larger study can validate the findings, the researchers wrote, this could lead to a new test for diagnosing PAH that’s less invasive than cardiac catheterization. It could also lead to new treatments that target the gut microbiome.
Study investigators collected stool samples from 18 PAH patients and 12 people without a history of cardiopulmonary disease. The microbiota DNA from the stool samples were isolated and sequenced. The analysis revealed that PAH patients had reduced richness and evenness of the gut bacteria, known as alpha diversity. They had increased levels of bacteria associated with atherosclerosis, and healthy patients had increased levels of bacteria that produced short-chain fatty acids.
Although recent studies have begun to show potential associations between the gut microbiome and cardiovascular diseases, this research is in its infancy, Mariell Jessup, MD, commented. “Even though the study by Dr. Raizada and colleagues predicted pulmonary arterial hypertension based on an individual’s microbiome with some accuracy, it is an observational study, so it does not prove cause and effect. Many other factors, especially diet, affect the gut microbiome,” added Dr. Jessup, Chief Science and Medical Officer for the American Heart Association.
She stressed that, “In addition, even if studies confirm an association between the gut microbiome and cardiovascular diseases such as PAH, more research is needed to determine if improving gut microbiota could directly impact PAH or other cardiovascular diseases. The findings of this study will not impact clinical practice.”
Dr. Raizada and his coinvestigators offered two possible mechanisms through which the gut microbiome influences pulmonary physiology. One is that lower levels of bacteria that produce the short-chain fatty acid butyrate, such as Coprococcus, Butyrivibrio, Lachnospiraceae, and Eubacterium, along with Clostridia in the gut of PAH patients, may increase gut permeability. Reduced butyrate weakens gut barrier function and can induce inflammation and leakage. This can allow microbial metabolites to enter the circulatory system, disrupting metabolism and immunity and affecting pulmonary vessels.
The second potential mechanism is that increased Collinsella in the PAH cohort may be the culprit that increases gut permeability, resulting in the ensuing gut barrier dysfunction and inflammation. The study noted Collinsella contributed most of the increased genes for the biosynthesis on the amino acid proline in these patients, and that a previous study implicated Collinsella and its parent, Cariobacteriales, in trimethylamine/trimethylamine N-oxide production (TMA/TMAO) in atherosclerosis (Cell. 2015;163[7]:1585-95). The non-PAH patients had higher levels of bacteria that had a low correlation with TMA/TMAO.
“We were very surprised to see such an association within a small group of study subjects,” wrote Dr. Raizada and associates. “It usually requires hundreds of patients to achieve such significance.”
More research is needed to determine if the specific microbiota associated with PAH causes the disease or is a result of it, they concluded.
The study was funded by grants from the National Institutes of Health, the NIH National Center for Research Resources, and the U.S. Department of Defense. Dr. Raizada and coauthors reported no relevant financial relationships.
SOURCE: Raizada MK et al. Hypertension. 2020. doi: 10.1161/HYPERTENSIONAHA.119.14294.
FROM HYPERTENSION
First clinical evidence of neuroprotection in acute stroke?
LOS ANGELES – A new potential neuroprotectant agent has been found to be beneficial for patients with acute ischemic stroke undergoing endovascular thrombectomy in a large placebo-controlled trial, but only for those patients who did not also receive thrombolysis.
There was no difference between groups on the primary outcome in the main analysis of the trial, lead author Michael Hill, MD, reported.
However, “In our study, we found a dramatic interaction of nerinetide with alteplase. There was a large benefit of nerinetide in patients not given thrombolysis, but in patients who received alteplase, this benefit was completely obliterated,” Dr. Hill said in an interview.
“In patients not treated with thrombolysis, we found a large effect size with a 9.5% absolute improvement in patients having an independent outcome (modified Rankin Score [mRS] 0-2) and a number need to treat of 10 to 11,” he said. “We also found a mortality benefit and a reduction in the size of strokes, with all other secondary outcomes going in the right direction.
“The drug works really well in patients who do not get thrombolysis, but it doesn’t work at all in patients who have had thrombolysis. The thrombolytic appears to break the peptide down so it is inactive,” he added.
“This is the first evidence that neuroprotection is possible in human stroke. This has never been shown before,” Dr. Hill noted. “Many previous clinical trials of potential neuroprotectants have been negative. We think this is a major breakthrough. This is pretty exciting stuff with really tantalizing results.”
Dr. Hill, professor of neurology at the University of Calgary (Alta.), presented results of the ESCAPE-NA1 trial on Feb. 20 at the International Stroke Conference (ISC) 2020. The trial was also simultaneously published online (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30258-0).
Endogenous nitric oxide
The new agent – known as NA1 or nerinetide – is a 20-amino-acid peptide with a novel mechanism of action; it inhibits signaling that leads to neuronal excitotoxicity. “It reduces endogenous nitric oxide generated inside the cell during ischemia, which is one of the main biochemical processes contributing to cell death,” Dr. Hill explained. In a primate model of ischemia reperfusion that was published in Nature in 2012, it was highly protective, he added.
The drug is given just once at the time of thrombectomy. It is short lived in the blood but detectable in the brain for up to 24 hours, he said.
The trial included 1,105 patients who had experienced acute ischemic stroke due to large-vessel occlusion within a 12-hour treatment window and for whom imaging results suitable for thrombectomy were available. The patients were randomly assigned to receive either intravenous nerinetide in a single dose of 2.6 mg/kg or saline placebo at the time of thrombectomy.
Patients were stratified by intravenous alteplase treatment and by declared endovascular device choice.
The primary outcome was a favorable functional outcome 90 days after randomization, defined as an mRS score of 0-2. In the main analysis of the whole population, this favorable outcome was achieved for 61.4% of the group that received nerinetide and for 59.2% of the placebo group, a nonsignificant difference. Secondary outcomes were also similar between the two groups.
But an exploratory analysis showed evidence that nerinetide’s treatment effect was modified by alteplase treatment. Among the patients who did not receive alteplase, use of nerinetide was associated with improved outcomes, whereas no benefit was found in the alteplase stratum. The difference in absolute risk slightly but not significantly favored placebo.
In the stratum that did not receive alteplase (40% of the trial population), the favorable mRS outcome was achieved by 59.3% of patients who received nerinetide, compared with 49.8% of those given placebo – a significant difference (adjusted risk ratio, 1.18; 95% confidence interval, 1.01-1.38).
There was also a 7.5% absolute risk reduction in mortality at 90 days post treatment with nerinetide for the patients who did not receive thrombolysis. This resulted in an approximate halving of the hazard of death (adjusted hazard ratio, 0.56).
In addition, infarct size was reduced in those patients who received nerinetide but not thrombolysis.
Among the patients who received alteplase, the proportion of patients who achieved an mRS of 0-2 was similar between groups, as were median infarct volumes.
The observed treatment effect modification by alteplase was supported by reductions in peak plasma nerinetide concentrations in the alteplase stratum, the researchers reported.
They said that the combination of the clinical results in the no-thrombolytic stratum and subsequent tests documenting that nerinetide is broken down by plasmin (which is generated by alteplase) “provide evidence that the clinical observation of effect modification is not a chance finding.” But they added: “This novel observation will require additional confirmation, and we cannot draw a definitive conclusion on treatment effect in this study.”
“Shaking up the field”
There is still more work to do, Dr. Hill said. “We don’t fully understand the pharmacology, and we will certainly have to do another trial, but we believe this agent is going to shake the field up. This is a totally new drug, and we have to think carefully about where it could fit in.”
“The obvious first group is those patients who do not receive thrombolysis. This is a large group, as most patients do not present in time for thrombolysis. Then we can work on the biochemistry and see if we can develop a version of nerinetide that is resistant to breakdown by thrombolysis,” he said.
Another possibility would be to withhold thrombolysis and give nerinetide instead. “It may be that thrombolysis is not needed if patients are receiving thrombectomy – this is being suggested now in initial studies,” Hill stated.
They also chose a very select group of patients – those undergoing thrombectomy, who represent only 10% to 15% of stroke patients. “We have to work out how to expand that population,” he said.
Hill noted that there have been many examples in the past of potential neuroprotectant agents that have worked in animal models of ischemia-reperfusion but that failed in humans with acute stroke.
“Until recently, we have not had a reliable ischemia-reperfusion model in humans, but now with endovascular therapy, we have a situation where the blood flow is reliably restored, which is an ideal situation to test new neuroprotectant agents. That may be another factor that has contributed to our positive findings,” he said.
In an accompanying comment in The Lancet, Graeme J. Hankey, MD, of the University of Western Australia, Perth, noted that although endovascular thrombectomy after use of intravenous alteplase improves reperfusion and clinical outcomes for a fifth of patients with ischemic stroke caused by large-artery occlusion, half of patients do not recover an independent lifestyle. Cytoprotection aims to augment the resilience of neurons, neurovascular units, and white matter during ischemia until perfusion is restored (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30316-0).
Dr. Hankey also pointed out that numerous cytoprotection strategies have been reported to reduce brain infarction in preclinical models of ischemic stroke but have not been found to improve clinical outcomes in clinical trials involving patients with ischemic stroke.
The advent of thrombectomy provides an opportunity to reassess cytoprotection as an adjunctive therapy for patients with types of temporary brain ischemia that align more closely with successful preclinical models of ischemia, cytoprotection, and reperfusion, he added.
On the results of the current study and the benefit in the no-thrombolysis group, Dr. Hankey stated: “Although this result might be a chance finding or confounded by the indication for alteplase, complementary pharmacokinetic data in a small number of patients treated with nerinetide showed that alteplase lowered plasma concentrations of nerinetide, probably by converting plasminogen to plasmin, which cleaves peptide bonds not only in fibrin but also in the eicosapeptide nerinetide.”
He said the ESCAPE-NA1 trial “informs the study of cytoprotection as an adjunct therapy to reperfusion in acute ischemic stroke” and suggested that researchers who have reported encouraging results of other cytoprotective therapies for ischemic stroke should test their compounds for interactions with concurrent thrombolytic therapies.
The ESCAPE-NA1 trial was sponsored by NoNO, the company developing nerinetide. Dr. Hill has received grants from NoNO for the conduct of the study, is named on a U.S. patent for systems and methods for assisting in decision making and triaging for acute stroke patients, and owns stock in Calgary Scientific. Other coauthors are employees of NoNO or have stock options in the company. Dr. Hankey has received personal honoraria from the American Heart Association, AC Immune, Bayer, Bristol-Myers Squibb, and Medscape outside the area of work that he commented on.
This article first appeared on Medscape.com.
LOS ANGELES – A new potential neuroprotectant agent has been found to be beneficial for patients with acute ischemic stroke undergoing endovascular thrombectomy in a large placebo-controlled trial, but only for those patients who did not also receive thrombolysis.
There was no difference between groups on the primary outcome in the main analysis of the trial, lead author Michael Hill, MD, reported.
However, “In our study, we found a dramatic interaction of nerinetide with alteplase. There was a large benefit of nerinetide in patients not given thrombolysis, but in patients who received alteplase, this benefit was completely obliterated,” Dr. Hill said in an interview.
“In patients not treated with thrombolysis, we found a large effect size with a 9.5% absolute improvement in patients having an independent outcome (modified Rankin Score [mRS] 0-2) and a number need to treat of 10 to 11,” he said. “We also found a mortality benefit and a reduction in the size of strokes, with all other secondary outcomes going in the right direction.
“The drug works really well in patients who do not get thrombolysis, but it doesn’t work at all in patients who have had thrombolysis. The thrombolytic appears to break the peptide down so it is inactive,” he added.
“This is the first evidence that neuroprotection is possible in human stroke. This has never been shown before,” Dr. Hill noted. “Many previous clinical trials of potential neuroprotectants have been negative. We think this is a major breakthrough. This is pretty exciting stuff with really tantalizing results.”
Dr. Hill, professor of neurology at the University of Calgary (Alta.), presented results of the ESCAPE-NA1 trial on Feb. 20 at the International Stroke Conference (ISC) 2020. The trial was also simultaneously published online (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30258-0).
Endogenous nitric oxide
The new agent – known as NA1 or nerinetide – is a 20-amino-acid peptide with a novel mechanism of action; it inhibits signaling that leads to neuronal excitotoxicity. “It reduces endogenous nitric oxide generated inside the cell during ischemia, which is one of the main biochemical processes contributing to cell death,” Dr. Hill explained. In a primate model of ischemia reperfusion that was published in Nature in 2012, it was highly protective, he added.
The drug is given just once at the time of thrombectomy. It is short lived in the blood but detectable in the brain for up to 24 hours, he said.
The trial included 1,105 patients who had experienced acute ischemic stroke due to large-vessel occlusion within a 12-hour treatment window and for whom imaging results suitable for thrombectomy were available. The patients were randomly assigned to receive either intravenous nerinetide in a single dose of 2.6 mg/kg or saline placebo at the time of thrombectomy.
Patients were stratified by intravenous alteplase treatment and by declared endovascular device choice.
The primary outcome was a favorable functional outcome 90 days after randomization, defined as an mRS score of 0-2. In the main analysis of the whole population, this favorable outcome was achieved for 61.4% of the group that received nerinetide and for 59.2% of the placebo group, a nonsignificant difference. Secondary outcomes were also similar between the two groups.
But an exploratory analysis showed evidence that nerinetide’s treatment effect was modified by alteplase treatment. Among the patients who did not receive alteplase, use of nerinetide was associated with improved outcomes, whereas no benefit was found in the alteplase stratum. The difference in absolute risk slightly but not significantly favored placebo.
In the stratum that did not receive alteplase (40% of the trial population), the favorable mRS outcome was achieved by 59.3% of patients who received nerinetide, compared with 49.8% of those given placebo – a significant difference (adjusted risk ratio, 1.18; 95% confidence interval, 1.01-1.38).
There was also a 7.5% absolute risk reduction in mortality at 90 days post treatment with nerinetide for the patients who did not receive thrombolysis. This resulted in an approximate halving of the hazard of death (adjusted hazard ratio, 0.56).
In addition, infarct size was reduced in those patients who received nerinetide but not thrombolysis.
Among the patients who received alteplase, the proportion of patients who achieved an mRS of 0-2 was similar between groups, as were median infarct volumes.
The observed treatment effect modification by alteplase was supported by reductions in peak plasma nerinetide concentrations in the alteplase stratum, the researchers reported.
They said that the combination of the clinical results in the no-thrombolytic stratum and subsequent tests documenting that nerinetide is broken down by plasmin (which is generated by alteplase) “provide evidence that the clinical observation of effect modification is not a chance finding.” But they added: “This novel observation will require additional confirmation, and we cannot draw a definitive conclusion on treatment effect in this study.”
“Shaking up the field”
There is still more work to do, Dr. Hill said. “We don’t fully understand the pharmacology, and we will certainly have to do another trial, but we believe this agent is going to shake the field up. This is a totally new drug, and we have to think carefully about where it could fit in.”
“The obvious first group is those patients who do not receive thrombolysis. This is a large group, as most patients do not present in time for thrombolysis. Then we can work on the biochemistry and see if we can develop a version of nerinetide that is resistant to breakdown by thrombolysis,” he said.
Another possibility would be to withhold thrombolysis and give nerinetide instead. “It may be that thrombolysis is not needed if patients are receiving thrombectomy – this is being suggested now in initial studies,” Hill stated.
They also chose a very select group of patients – those undergoing thrombectomy, who represent only 10% to 15% of stroke patients. “We have to work out how to expand that population,” he said.
Hill noted that there have been many examples in the past of potential neuroprotectant agents that have worked in animal models of ischemia-reperfusion but that failed in humans with acute stroke.
“Until recently, we have not had a reliable ischemia-reperfusion model in humans, but now with endovascular therapy, we have a situation where the blood flow is reliably restored, which is an ideal situation to test new neuroprotectant agents. That may be another factor that has contributed to our positive findings,” he said.
In an accompanying comment in The Lancet, Graeme J. Hankey, MD, of the University of Western Australia, Perth, noted that although endovascular thrombectomy after use of intravenous alteplase improves reperfusion and clinical outcomes for a fifth of patients with ischemic stroke caused by large-artery occlusion, half of patients do not recover an independent lifestyle. Cytoprotection aims to augment the resilience of neurons, neurovascular units, and white matter during ischemia until perfusion is restored (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30316-0).
Dr. Hankey also pointed out that numerous cytoprotection strategies have been reported to reduce brain infarction in preclinical models of ischemic stroke but have not been found to improve clinical outcomes in clinical trials involving patients with ischemic stroke.
The advent of thrombectomy provides an opportunity to reassess cytoprotection as an adjunctive therapy for patients with types of temporary brain ischemia that align more closely with successful preclinical models of ischemia, cytoprotection, and reperfusion, he added.
On the results of the current study and the benefit in the no-thrombolysis group, Dr. Hankey stated: “Although this result might be a chance finding or confounded by the indication for alteplase, complementary pharmacokinetic data in a small number of patients treated with nerinetide showed that alteplase lowered plasma concentrations of nerinetide, probably by converting plasminogen to plasmin, which cleaves peptide bonds not only in fibrin but also in the eicosapeptide nerinetide.”
He said the ESCAPE-NA1 trial “informs the study of cytoprotection as an adjunct therapy to reperfusion in acute ischemic stroke” and suggested that researchers who have reported encouraging results of other cytoprotective therapies for ischemic stroke should test their compounds for interactions with concurrent thrombolytic therapies.
The ESCAPE-NA1 trial was sponsored by NoNO, the company developing nerinetide. Dr. Hill has received grants from NoNO for the conduct of the study, is named on a U.S. patent for systems and methods for assisting in decision making and triaging for acute stroke patients, and owns stock in Calgary Scientific. Other coauthors are employees of NoNO or have stock options in the company. Dr. Hankey has received personal honoraria from the American Heart Association, AC Immune, Bayer, Bristol-Myers Squibb, and Medscape outside the area of work that he commented on.
This article first appeared on Medscape.com.
LOS ANGELES – A new potential neuroprotectant agent has been found to be beneficial for patients with acute ischemic stroke undergoing endovascular thrombectomy in a large placebo-controlled trial, but only for those patients who did not also receive thrombolysis.
There was no difference between groups on the primary outcome in the main analysis of the trial, lead author Michael Hill, MD, reported.
However, “In our study, we found a dramatic interaction of nerinetide with alteplase. There was a large benefit of nerinetide in patients not given thrombolysis, but in patients who received alteplase, this benefit was completely obliterated,” Dr. Hill said in an interview.
“In patients not treated with thrombolysis, we found a large effect size with a 9.5% absolute improvement in patients having an independent outcome (modified Rankin Score [mRS] 0-2) and a number need to treat of 10 to 11,” he said. “We also found a mortality benefit and a reduction in the size of strokes, with all other secondary outcomes going in the right direction.
“The drug works really well in patients who do not get thrombolysis, but it doesn’t work at all in patients who have had thrombolysis. The thrombolytic appears to break the peptide down so it is inactive,” he added.
“This is the first evidence that neuroprotection is possible in human stroke. This has never been shown before,” Dr. Hill noted. “Many previous clinical trials of potential neuroprotectants have been negative. We think this is a major breakthrough. This is pretty exciting stuff with really tantalizing results.”
Dr. Hill, professor of neurology at the University of Calgary (Alta.), presented results of the ESCAPE-NA1 trial on Feb. 20 at the International Stroke Conference (ISC) 2020. The trial was also simultaneously published online (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30258-0).
Endogenous nitric oxide
The new agent – known as NA1 or nerinetide – is a 20-amino-acid peptide with a novel mechanism of action; it inhibits signaling that leads to neuronal excitotoxicity. “It reduces endogenous nitric oxide generated inside the cell during ischemia, which is one of the main biochemical processes contributing to cell death,” Dr. Hill explained. In a primate model of ischemia reperfusion that was published in Nature in 2012, it was highly protective, he added.
The drug is given just once at the time of thrombectomy. It is short lived in the blood but detectable in the brain for up to 24 hours, he said.
The trial included 1,105 patients who had experienced acute ischemic stroke due to large-vessel occlusion within a 12-hour treatment window and for whom imaging results suitable for thrombectomy were available. The patients were randomly assigned to receive either intravenous nerinetide in a single dose of 2.6 mg/kg or saline placebo at the time of thrombectomy.
Patients were stratified by intravenous alteplase treatment and by declared endovascular device choice.
The primary outcome was a favorable functional outcome 90 days after randomization, defined as an mRS score of 0-2. In the main analysis of the whole population, this favorable outcome was achieved for 61.4% of the group that received nerinetide and for 59.2% of the placebo group, a nonsignificant difference. Secondary outcomes were also similar between the two groups.
But an exploratory analysis showed evidence that nerinetide’s treatment effect was modified by alteplase treatment. Among the patients who did not receive alteplase, use of nerinetide was associated with improved outcomes, whereas no benefit was found in the alteplase stratum. The difference in absolute risk slightly but not significantly favored placebo.
In the stratum that did not receive alteplase (40% of the trial population), the favorable mRS outcome was achieved by 59.3% of patients who received nerinetide, compared with 49.8% of those given placebo – a significant difference (adjusted risk ratio, 1.18; 95% confidence interval, 1.01-1.38).
There was also a 7.5% absolute risk reduction in mortality at 90 days post treatment with nerinetide for the patients who did not receive thrombolysis. This resulted in an approximate halving of the hazard of death (adjusted hazard ratio, 0.56).
In addition, infarct size was reduced in those patients who received nerinetide but not thrombolysis.
Among the patients who received alteplase, the proportion of patients who achieved an mRS of 0-2 was similar between groups, as were median infarct volumes.
The observed treatment effect modification by alteplase was supported by reductions in peak plasma nerinetide concentrations in the alteplase stratum, the researchers reported.
They said that the combination of the clinical results in the no-thrombolytic stratum and subsequent tests documenting that nerinetide is broken down by plasmin (which is generated by alteplase) “provide evidence that the clinical observation of effect modification is not a chance finding.” But they added: “This novel observation will require additional confirmation, and we cannot draw a definitive conclusion on treatment effect in this study.”
“Shaking up the field”
There is still more work to do, Dr. Hill said. “We don’t fully understand the pharmacology, and we will certainly have to do another trial, but we believe this agent is going to shake the field up. This is a totally new drug, and we have to think carefully about where it could fit in.”
“The obvious first group is those patients who do not receive thrombolysis. This is a large group, as most patients do not present in time for thrombolysis. Then we can work on the biochemistry and see if we can develop a version of nerinetide that is resistant to breakdown by thrombolysis,” he said.
Another possibility would be to withhold thrombolysis and give nerinetide instead. “It may be that thrombolysis is not needed if patients are receiving thrombectomy – this is being suggested now in initial studies,” Hill stated.
They also chose a very select group of patients – those undergoing thrombectomy, who represent only 10% to 15% of stroke patients. “We have to work out how to expand that population,” he said.
Hill noted that there have been many examples in the past of potential neuroprotectant agents that have worked in animal models of ischemia-reperfusion but that failed in humans with acute stroke.
“Until recently, we have not had a reliable ischemia-reperfusion model in humans, but now with endovascular therapy, we have a situation where the blood flow is reliably restored, which is an ideal situation to test new neuroprotectant agents. That may be another factor that has contributed to our positive findings,” he said.
In an accompanying comment in The Lancet, Graeme J. Hankey, MD, of the University of Western Australia, Perth, noted that although endovascular thrombectomy after use of intravenous alteplase improves reperfusion and clinical outcomes for a fifth of patients with ischemic stroke caused by large-artery occlusion, half of patients do not recover an independent lifestyle. Cytoprotection aims to augment the resilience of neurons, neurovascular units, and white matter during ischemia until perfusion is restored (Lancet. 2020 Feb 20; doi: 10.1016/S0140-6736(20)30316-0).
Dr. Hankey also pointed out that numerous cytoprotection strategies have been reported to reduce brain infarction in preclinical models of ischemic stroke but have not been found to improve clinical outcomes in clinical trials involving patients with ischemic stroke.
The advent of thrombectomy provides an opportunity to reassess cytoprotection as an adjunctive therapy for patients with types of temporary brain ischemia that align more closely with successful preclinical models of ischemia, cytoprotection, and reperfusion, he added.
On the results of the current study and the benefit in the no-thrombolysis group, Dr. Hankey stated: “Although this result might be a chance finding or confounded by the indication for alteplase, complementary pharmacokinetic data in a small number of patients treated with nerinetide showed that alteplase lowered plasma concentrations of nerinetide, probably by converting plasminogen to plasmin, which cleaves peptide bonds not only in fibrin but also in the eicosapeptide nerinetide.”
He said the ESCAPE-NA1 trial “informs the study of cytoprotection as an adjunct therapy to reperfusion in acute ischemic stroke” and suggested that researchers who have reported encouraging results of other cytoprotective therapies for ischemic stroke should test their compounds for interactions with concurrent thrombolytic therapies.
The ESCAPE-NA1 trial was sponsored by NoNO, the company developing nerinetide. Dr. Hill has received grants from NoNO for the conduct of the study, is named on a U.S. patent for systems and methods for assisting in decision making and triaging for acute stroke patients, and owns stock in Calgary Scientific. Other coauthors are employees of NoNO or have stock options in the company. Dr. Hankey has received personal honoraria from the American Heart Association, AC Immune, Bayer, Bristol-Myers Squibb, and Medscape outside the area of work that he commented on.
This article first appeared on Medscape.com.
New lipid-lowering drug earns FDA approval
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The Food and Drug Administration has approved bempedoic acid (Nexletol) for the treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or established atherosclerotic cardiovascular disease (ASCVD) who require additional LDL cholesterol lowering.
The oral adenosine triphosphate–citrate lyase (ACL) inhibitor is indicated as an adjunct to diet and maximally tolerated statin therapy in these patients, and approved at the 180 mg once daily dose, the agency announced today.
The safety and efficacy of bempedoic acid were demonstrated over 52 weeks in two multicenter randomized, clinical trials involving 3,009 adults with HeFH or established ASCVD on maximally tolerated statin therapy.
The difference between bempedoic acid and placebo for the primary outcome of change in LDL cholesterol from baseline to week 12 was –18% in the first trial, CLEAR Harmony (95% confidence interval, –20% to –16%; P less than .001), and –17% in the second trial, CLEAR Wisdom (95% CI, –21% to –14%; P less than .001).
The label notes that the effect on cardiovascular morbidity and mortality has not been determined. The label also includes warnings stating that bempedoic acid may increase blood uric acid levels and is associated with an increased risk of tendon rupture or injury.
In clinical trials, 26% of bempedoic acid–treated patients with normal baseline uric acid values versus 9.5% of placebo-treated patients experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction versus 1.1% with placebo, according to the label. Gout was reported in 1.5% of patients treated with bempedoic acid and 0.4% of those treated with placebo.
Also in clinical trials, the risk of tendon rupture was 0.5% with bempedoic acid and 0% with placebo. Tendon rupture involved the rotator cuff, biceps tendon, or Achilles tendon, and occurred within weeks to months of starting the drug. Rupture may “occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders,” the label states.
The label also advises that patients avoid concomitant use of bempedoic acid with simvastatin greater than 20 mg or pravastatin greater than 40 mg because it causes an increase in statin concentrations and may increase the risk of related myopathy.
A decision is expected shortly on a new drug application submitted by Esperion for an LDL cholesterol–lowering indication for bempedoic acid 180 mg/ezetimibe 10 mg combination tablet.
Full prescribing information is available online.
This article first appeared on Medscape.com.
Higher endovascular thrombectomy volumes yield better stroke outcomes
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
The results reported by Dr. Stein raise issues about balancing the access to certain therapies with the outcomes of those therapies. Having procedures like endovascular thrombectomy for acute ischemic stroke done primarily at high-volume centers might improve procedural outcomes, but having more centers offering this treatment across wider geographical areas would make this treatment more broadly available to more people.
For endovascular thrombectomy, center volume and experience may be much more important than proceduralist volume because having a smoothly functioning system in place is so important for rapid stroke assessment and treatment. It’s also important for programs to provide experienced and comprehensive postthrombectomy care. Success in endovascular thrombectomy involves much more than just taking a clot out. It means quickly and smoothly moving patients through the steps that precede thrombectomy and then following the intervention with a range of services that optimize recovery.
Ashutosh P. Jadhav, MD, PhD , is director of the comprehensive stroke center at the University of Pittsburgh. He had no relevant disclosures. He made these comments in an interview.
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
LOS ANGELES – Higher case volumes matter for getting better outcomes in acute ischemic stroke patients treated with endovascular thrombectomy, according to data from more than 13,000 Medicare patients treated during 2016 and 2017.
That’s hardly surprising, given that it’s consistent with what’s already been reported for several other types of endovascular and transcatheter procedures: The more cases a center or individual proceduralist performs, the better their patients do. Routine use of endovascular thrombectomy to treat selected acute ischemic stroke patients is a new-enough paradigm that until now few reports have come out that looked at this issue (Stroke. 2019 May;50[5]:1178-83).
The new analysis of Medicare data “is one of the first contemporary studies of the volume-outcome relationship in endovascular thrombectomy,” Laura K. Stein, MD, said at the International Stroke Conference sponsored by the American Heart Association. The analysis showed that, when the researchers adjusted the Medicare data to better reflect overall case volumes (Medicare patients represent just 59% of all endovascular thrombectomies performed on U.S. acute ischemic stroke patients), the minimum case number for a stroke center to have statistically better in-hospital survival than lower volume centers was 24 cases/year, and 29 cases/year to have a statistically significant higher rate of “good” outcomes than lower-volume centers, reported Dr. Stein, a stroke neurologist with the Mount Sinai Health System in New York. For individual proceduralists, the minimum, adjusted case number to have statistically better acute patient survival was 4 cases/year, and 19 cases/year to have a statistically better rate of good outcomes.
For this analysis, good outcomes were defined as cases when patients left the hospital following their acute care and returned home with either self care or a home health care service, and also patients discharged to rehabilitation. “Bad” outcomes for this analysis were discharges to a skilled nursing facility or hospice, as well as patients who died during their acute hospitalization.
The analyses also showed no plateau to the volume effect for any of the four parameters examined: in-hospital mortality by center and by proceduralist, and the rates of good outcomes by center and by proceduralist. For each of these measures, as case volume increased above the minimum number needed to produce statistically better outcomes, the rate of good outcomes continued to steadily rise and acute mortality continued to steadily fall.
The study run by Dr. Stein and associates used data collected by the Center for Medicare & Medicaid Services on 13,311 Medicare patients who underwent endovascular thrombectomy for acute ischemic stroke at any of 641 U.S. hospitals and received treatment from any of 2,754 thrombectomy proceduralists. Outcomes rated as good occurred in 56% of the patients. The statistical adjustments that the researchers applied to calculate the incremental effect of increasing case volume took into account the variables of patient age, sex, and comorbidities measured by the Charlson Comorbidity Index.
The analysis also showed that, during this 2-year period, the average number of endovascular thrombectomy cases among Medicare patients was just under 21 cases per center, with a range of 1-160 cases; for individual proceduralists, the average was just under 5 cases, with a range of 1-82 cases.
The 19 case/year volume minimum that the analysis identified for an individual proceduralist to have a statistically significant higher rate of good outcomes, compared with lower-volume proceduralists, came close to the 15 cases/year minimum set by the Joint Commission in 2019 for individual operators at centers seeking accreditation from the Joint Commission as either a Thrombectomy-Capable Stroke Center or a Comprehensive Stroke Center. The CMS has not yet set thrombectomy case-load requirements for centers or operators to qualify for Medicare reimbursements, although CMS has set such standards for other endovascular procedures, such as transcatheter aortic valve replacement. When setting such standards, CMS has cited its need to balance the better outcomes produced by higher-volume centers against a societal interest in facilitating access to vital medical services, a balance that Dr. Stein also highlighted in her talk.
“We want to optimize access as well as outcomes for every patient,” she said. “These data support certification volume standards,” but they are “in no way an argument for limiting access based on volume.”
Dr. Stein had no disclosures.
SOURCE: Stein LK et al. ISC 2020, Abstract LB11.
REPORTING FROM ISC 2020
TNK dose in large-vessel stroke: 0.25 mg/kg is sufficient
A new study suggests that the 0.25-mg/kg dose of the thrombolytic tenecteplase (TNK) is just as good at facilitating reperfusion of the blocked artery in patients with ischemic large-vessel stroke prior to planned thrombectomy as the higher 0.4-mg/kg dose.
The EXTEND-IA TNK Part 2 trial was presented today at the American Stroke Association’s International Stroke Conference (ISC) 2020 in Los Angeles and was published online simultaneously (JAMA. 2020 Feb 20. doi: 10.1001/jama.2020.1511).
“We found the 0.4-mg/kg dose was no better than 0.25 mg/kg. There was absolutely no perceptible difference, so it appears that 0.25 mg/kg is enough,” lead investigator Bruce Campbell, MBBS, PhD, said in an interview.
“Our study was conducted in patients with large-vessel occlusions heading for thrombectomy, but I think the results can be extrapolated to patients with smaller occlusions too,” he added.
The study also showed that one-fifth of patients given tenecteplase experienced reperfusion before thrombectomy was performed. The percentage rose to one-third among patients from rural areas, whose longer times in transport led to an increase in the time between thrombolysis and thrombectomy.
“I think these data are as good as we’re going to get on the optimal dose of TNK. Our endpoint was reperfusion rates – a good, solid biological marker of benefit – but if a difference in clinical outcomes is wanted, that would take a trial of several thousand patients, which is never likely to be done,” said Dr. Campbell, who is from the Department of Neurology at the Royal Melbourne Hospital, Australia.
The researchers note that tenecteplase has a practical advantage over alteplase in that it is given as a bolus injection, whereas alteplase is given as bolus followed by a 1-hour infusion.
Results from the first EXTEND-IA TNK study suggested that tenecteplase 0.25 mg/kg produced higher reperfusion rates than alteplase (N Engl J Med. 2018;378:1573-82). However, the larger NOR-TEST study found no difference in efficacy or safety between a 0.4-mg/kg dose of tenecteplase and alteplase in patients with mild stroke (Lancet Neurol. 2017 Oct;16[10]:781-8).
TNK use in stroke varies around the world. The drug is not licensed for use in stroke anywhere, which Dr. Campbell attributes to a lack of incentive for the manufacturer, Genentech/Boehringer Ingelheim. That company also markets alteplase, the main thrombolytic used in stroke.
But many countries have now included TNK in their stroke guidelines, Dr. Campbell noted. “This has only recently occurred in the U.S., where it has a 2b recommendation, and the dose recommendations are somewhat confusing, advocating 0.25 mg/kg in large-vessel occlusions [as was used in the first EXTEND IA study] and 0.4 mg/kg in non–large vessel occlusions [from the NOR-TEST trial].
“This makes no biological sense whatsoever, recommending a higher dose for smaller occlusions, but that is just a literal translation of the design of the two major studies. I’m hoping our current results will help clarify the dosage issue and that might encourage more use of TNK altogether,” he commented.
For the current study, conducted in Australia and New Zealand, 300 patients who had experienced ischemic large-vessel stroke within 4.5 hours of symptom onset and who were scheduled for endovascular thrombectomy were randomly assigned to receive open-label thrombolysis with tenecteplase 0.4 mg/kg or 0.25 mg/kg.
The primary outcome, reperfusion of greater than 50% of the involved ischemic territory prior to thrombectomy, occurred in 19.3% of both groups. There was also no difference in any of the functional-outcome secondary endpoints or all-cause mortality between the two doses.
“While we didn’t find any extra benefit of the 0.4-mg/kg dose over the 0.25-mg/kg dose, we also didn’t find any extra harm, and this gives us reassurance in the emergency situation if the weight of the patient is overestimated; then we have a window of safety,” Dr. Campbell commented. “While there was a nonsignificant numerical increase in intracranial hemorrhage in the 0.4-mg/kg group, the excess bleeds were caused by puncturing of the vessels during thrombectomy, so I don’t think we can blame the TNK dose for that.
Better reperfusion than with alteplase?
Noting that the original EXTEND-IA TNK study showed higher reperfusion rates with tenecteplase vs alteplase and a trend toward better outcomes on the mRS scale, Campbell reported that a pooled analysis of the TNK results from the current study with those from the first study confirmed these findings.
“We found a doubling in the rate of reperfusion with TNK vs. alteplase, and the [modified Rankin Scale] shift analysis remained positive,” he said.
“I think we say with confidence that TNK is at least as good as alteplase and probably better, but further studies comparing the two agents are ongoing,” he added.
Of note, for the 41 patients from rural areas in the current study, in whom the time from thrombolysis to thrombectomy was longer (152 min vs. 41 min for patients from urban areas), reperfusion rates were higher (34% vs 17%), and there was no difference in dosage between the two groups.
Commenting on these latest results in an interview, Nicola Logallo, MD, of Haukeland University Hospital, Bergen, Norway, who was part of the NOR-TEST trial, said: “There is some evidence supporting the use of TNK 0.4 mg/kg in mild stroke patients, based mainly on the results from the NOR-TEST trial, and the use of TNK 0.25 mg/kg in patients undergoing thrombectomy, based on Dr. Campbell’s previous EXTEND-TNK trial. Dr. Campbell’s new study confirms that probably the higher dose of TNK does not add any advantages in terms of clinical outcome.”
Hemorrhagic complications appear to be similar in the two groups, Dr. Logallo said. “Overall, the 0.25-mg/kg TNK dose could therefore be considered as the most convenient and sensible, at least in patients undergoing thrombectomy. When it comes to the remaining stroke patients receiving thrombolysis, it remains unclear which is the best dose, but studies such as TASTE, NOR-TEST 2, AcT, and ATTEST-2 will hopefully answer this question within the next years.”
Also commenting on the study, Michael Hill, MD, professor of neurology at University of Calgary, Alberta, Canada, said the results “confirm that a good proportion of patients given TNK reperfuse before the angiogram and clarifies the dose. This is useful information.”
Dr. Hill said TNK is used routinely in some countries – mainly in Australia and Norway, where the studies have been conducted – but there is now a movement toward use of TNK in North America, too.
“Studies so far suggest that it could be more effective than alteplase, and as it is more fibrin specific, it could be safer. It is also easier to give with a bolus dose, but perhaps the biggest driver might be that it is cheaper than alteplase. Momentum is building, and many leading investigators are now conducting new studies with TNK with several more studies coming out in the next year or so,” Dr. Hill added.
The EXTEND-IA TNK Part 2 trial was supported by grants from the National Health and Medical Research Council of Australia and the National Heart Foundation of Australia. Campbell reports receiving grants from both institutions during the conduct of the study.
This article first appeared on Medscape.com.
A new study suggests that the 0.25-mg/kg dose of the thrombolytic tenecteplase (TNK) is just as good at facilitating reperfusion of the blocked artery in patients with ischemic large-vessel stroke prior to planned thrombectomy as the higher 0.4-mg/kg dose.
The EXTEND-IA TNK Part 2 trial was presented today at the American Stroke Association’s International Stroke Conference (ISC) 2020 in Los Angeles and was published online simultaneously (JAMA. 2020 Feb 20. doi: 10.1001/jama.2020.1511).
“We found the 0.4-mg/kg dose was no better than 0.25 mg/kg. There was absolutely no perceptible difference, so it appears that 0.25 mg/kg is enough,” lead investigator Bruce Campbell, MBBS, PhD, said in an interview.
“Our study was conducted in patients with large-vessel occlusions heading for thrombectomy, but I think the results can be extrapolated to patients with smaller occlusions too,” he added.
The study also showed that one-fifth of patients given tenecteplase experienced reperfusion before thrombectomy was performed. The percentage rose to one-third among patients from rural areas, whose longer times in transport led to an increase in the time between thrombolysis and thrombectomy.
“I think these data are as good as we’re going to get on the optimal dose of TNK. Our endpoint was reperfusion rates – a good, solid biological marker of benefit – but if a difference in clinical outcomes is wanted, that would take a trial of several thousand patients, which is never likely to be done,” said Dr. Campbell, who is from the Department of Neurology at the Royal Melbourne Hospital, Australia.
The researchers note that tenecteplase has a practical advantage over alteplase in that it is given as a bolus injection, whereas alteplase is given as bolus followed by a 1-hour infusion.
Results from the first EXTEND-IA TNK study suggested that tenecteplase 0.25 mg/kg produced higher reperfusion rates than alteplase (N Engl J Med. 2018;378:1573-82). However, the larger NOR-TEST study found no difference in efficacy or safety between a 0.4-mg/kg dose of tenecteplase and alteplase in patients with mild stroke (Lancet Neurol. 2017 Oct;16[10]:781-8).
TNK use in stroke varies around the world. The drug is not licensed for use in stroke anywhere, which Dr. Campbell attributes to a lack of incentive for the manufacturer, Genentech/Boehringer Ingelheim. That company also markets alteplase, the main thrombolytic used in stroke.
But many countries have now included TNK in their stroke guidelines, Dr. Campbell noted. “This has only recently occurred in the U.S., where it has a 2b recommendation, and the dose recommendations are somewhat confusing, advocating 0.25 mg/kg in large-vessel occlusions [as was used in the first EXTEND IA study] and 0.4 mg/kg in non–large vessel occlusions [from the NOR-TEST trial].
“This makes no biological sense whatsoever, recommending a higher dose for smaller occlusions, but that is just a literal translation of the design of the two major studies. I’m hoping our current results will help clarify the dosage issue and that might encourage more use of TNK altogether,” he commented.
For the current study, conducted in Australia and New Zealand, 300 patients who had experienced ischemic large-vessel stroke within 4.5 hours of symptom onset and who were scheduled for endovascular thrombectomy were randomly assigned to receive open-label thrombolysis with tenecteplase 0.4 mg/kg or 0.25 mg/kg.
The primary outcome, reperfusion of greater than 50% of the involved ischemic territory prior to thrombectomy, occurred in 19.3% of both groups. There was also no difference in any of the functional-outcome secondary endpoints or all-cause mortality between the two doses.
“While we didn’t find any extra benefit of the 0.4-mg/kg dose over the 0.25-mg/kg dose, we also didn’t find any extra harm, and this gives us reassurance in the emergency situation if the weight of the patient is overestimated; then we have a window of safety,” Dr. Campbell commented. “While there was a nonsignificant numerical increase in intracranial hemorrhage in the 0.4-mg/kg group, the excess bleeds were caused by puncturing of the vessels during thrombectomy, so I don’t think we can blame the TNK dose for that.
Better reperfusion than with alteplase?
Noting that the original EXTEND-IA TNK study showed higher reperfusion rates with tenecteplase vs alteplase and a trend toward better outcomes on the mRS scale, Campbell reported that a pooled analysis of the TNK results from the current study with those from the first study confirmed these findings.
“We found a doubling in the rate of reperfusion with TNK vs. alteplase, and the [modified Rankin Scale] shift analysis remained positive,” he said.
“I think we say with confidence that TNK is at least as good as alteplase and probably better, but further studies comparing the two agents are ongoing,” he added.
Of note, for the 41 patients from rural areas in the current study, in whom the time from thrombolysis to thrombectomy was longer (152 min vs. 41 min for patients from urban areas), reperfusion rates were higher (34% vs 17%), and there was no difference in dosage between the two groups.
Commenting on these latest results in an interview, Nicola Logallo, MD, of Haukeland University Hospital, Bergen, Norway, who was part of the NOR-TEST trial, said: “There is some evidence supporting the use of TNK 0.4 mg/kg in mild stroke patients, based mainly on the results from the NOR-TEST trial, and the use of TNK 0.25 mg/kg in patients undergoing thrombectomy, based on Dr. Campbell’s previous EXTEND-TNK trial. Dr. Campbell’s new study confirms that probably the higher dose of TNK does not add any advantages in terms of clinical outcome.”
Hemorrhagic complications appear to be similar in the two groups, Dr. Logallo said. “Overall, the 0.25-mg/kg TNK dose could therefore be considered as the most convenient and sensible, at least in patients undergoing thrombectomy. When it comes to the remaining stroke patients receiving thrombolysis, it remains unclear which is the best dose, but studies such as TASTE, NOR-TEST 2, AcT, and ATTEST-2 will hopefully answer this question within the next years.”
Also commenting on the study, Michael Hill, MD, professor of neurology at University of Calgary, Alberta, Canada, said the results “confirm that a good proportion of patients given TNK reperfuse before the angiogram and clarifies the dose. This is useful information.”
Dr. Hill said TNK is used routinely in some countries – mainly in Australia and Norway, where the studies have been conducted – but there is now a movement toward use of TNK in North America, too.
“Studies so far suggest that it could be more effective than alteplase, and as it is more fibrin specific, it could be safer. It is also easier to give with a bolus dose, but perhaps the biggest driver might be that it is cheaper than alteplase. Momentum is building, and many leading investigators are now conducting new studies with TNK with several more studies coming out in the next year or so,” Dr. Hill added.
The EXTEND-IA TNK Part 2 trial was supported by grants from the National Health and Medical Research Council of Australia and the National Heart Foundation of Australia. Campbell reports receiving grants from both institutions during the conduct of the study.
This article first appeared on Medscape.com.
A new study suggests that the 0.25-mg/kg dose of the thrombolytic tenecteplase (TNK) is just as good at facilitating reperfusion of the blocked artery in patients with ischemic large-vessel stroke prior to planned thrombectomy as the higher 0.4-mg/kg dose.
The EXTEND-IA TNK Part 2 trial was presented today at the American Stroke Association’s International Stroke Conference (ISC) 2020 in Los Angeles and was published online simultaneously (JAMA. 2020 Feb 20. doi: 10.1001/jama.2020.1511).
“We found the 0.4-mg/kg dose was no better than 0.25 mg/kg. There was absolutely no perceptible difference, so it appears that 0.25 mg/kg is enough,” lead investigator Bruce Campbell, MBBS, PhD, said in an interview.
“Our study was conducted in patients with large-vessel occlusions heading for thrombectomy, but I think the results can be extrapolated to patients with smaller occlusions too,” he added.
The study also showed that one-fifth of patients given tenecteplase experienced reperfusion before thrombectomy was performed. The percentage rose to one-third among patients from rural areas, whose longer times in transport led to an increase in the time between thrombolysis and thrombectomy.
“I think these data are as good as we’re going to get on the optimal dose of TNK. Our endpoint was reperfusion rates – a good, solid biological marker of benefit – but if a difference in clinical outcomes is wanted, that would take a trial of several thousand patients, which is never likely to be done,” said Dr. Campbell, who is from the Department of Neurology at the Royal Melbourne Hospital, Australia.
The researchers note that tenecteplase has a practical advantage over alteplase in that it is given as a bolus injection, whereas alteplase is given as bolus followed by a 1-hour infusion.
Results from the first EXTEND-IA TNK study suggested that tenecteplase 0.25 mg/kg produced higher reperfusion rates than alteplase (N Engl J Med. 2018;378:1573-82). However, the larger NOR-TEST study found no difference in efficacy or safety between a 0.4-mg/kg dose of tenecteplase and alteplase in patients with mild stroke (Lancet Neurol. 2017 Oct;16[10]:781-8).
TNK use in stroke varies around the world. The drug is not licensed for use in stroke anywhere, which Dr. Campbell attributes to a lack of incentive for the manufacturer, Genentech/Boehringer Ingelheim. That company also markets alteplase, the main thrombolytic used in stroke.
But many countries have now included TNK in their stroke guidelines, Dr. Campbell noted. “This has only recently occurred in the U.S., where it has a 2b recommendation, and the dose recommendations are somewhat confusing, advocating 0.25 mg/kg in large-vessel occlusions [as was used in the first EXTEND IA study] and 0.4 mg/kg in non–large vessel occlusions [from the NOR-TEST trial].
“This makes no biological sense whatsoever, recommending a higher dose for smaller occlusions, but that is just a literal translation of the design of the two major studies. I’m hoping our current results will help clarify the dosage issue and that might encourage more use of TNK altogether,” he commented.
For the current study, conducted in Australia and New Zealand, 300 patients who had experienced ischemic large-vessel stroke within 4.5 hours of symptom onset and who were scheduled for endovascular thrombectomy were randomly assigned to receive open-label thrombolysis with tenecteplase 0.4 mg/kg or 0.25 mg/kg.
The primary outcome, reperfusion of greater than 50% of the involved ischemic territory prior to thrombectomy, occurred in 19.3% of both groups. There was also no difference in any of the functional-outcome secondary endpoints or all-cause mortality between the two doses.
“While we didn’t find any extra benefit of the 0.4-mg/kg dose over the 0.25-mg/kg dose, we also didn’t find any extra harm, and this gives us reassurance in the emergency situation if the weight of the patient is overestimated; then we have a window of safety,” Dr. Campbell commented. “While there was a nonsignificant numerical increase in intracranial hemorrhage in the 0.4-mg/kg group, the excess bleeds were caused by puncturing of the vessels during thrombectomy, so I don’t think we can blame the TNK dose for that.
Better reperfusion than with alteplase?
Noting that the original EXTEND-IA TNK study showed higher reperfusion rates with tenecteplase vs alteplase and a trend toward better outcomes on the mRS scale, Campbell reported that a pooled analysis of the TNK results from the current study with those from the first study confirmed these findings.
“We found a doubling in the rate of reperfusion with TNK vs. alteplase, and the [modified Rankin Scale] shift analysis remained positive,” he said.
“I think we say with confidence that TNK is at least as good as alteplase and probably better, but further studies comparing the two agents are ongoing,” he added.
Of note, for the 41 patients from rural areas in the current study, in whom the time from thrombolysis to thrombectomy was longer (152 min vs. 41 min for patients from urban areas), reperfusion rates were higher (34% vs 17%), and there was no difference in dosage between the two groups.
Commenting on these latest results in an interview, Nicola Logallo, MD, of Haukeland University Hospital, Bergen, Norway, who was part of the NOR-TEST trial, said: “There is some evidence supporting the use of TNK 0.4 mg/kg in mild stroke patients, based mainly on the results from the NOR-TEST trial, and the use of TNK 0.25 mg/kg in patients undergoing thrombectomy, based on Dr. Campbell’s previous EXTEND-TNK trial. Dr. Campbell’s new study confirms that probably the higher dose of TNK does not add any advantages in terms of clinical outcome.”
Hemorrhagic complications appear to be similar in the two groups, Dr. Logallo said. “Overall, the 0.25-mg/kg TNK dose could therefore be considered as the most convenient and sensible, at least in patients undergoing thrombectomy. When it comes to the remaining stroke patients receiving thrombolysis, it remains unclear which is the best dose, but studies such as TASTE, NOR-TEST 2, AcT, and ATTEST-2 will hopefully answer this question within the next years.”
Also commenting on the study, Michael Hill, MD, professor of neurology at University of Calgary, Alberta, Canada, said the results “confirm that a good proportion of patients given TNK reperfuse before the angiogram and clarifies the dose. This is useful information.”
Dr. Hill said TNK is used routinely in some countries – mainly in Australia and Norway, where the studies have been conducted – but there is now a movement toward use of TNK in North America, too.
“Studies so far suggest that it could be more effective than alteplase, and as it is more fibrin specific, it could be safer. It is also easier to give with a bolus dose, but perhaps the biggest driver might be that it is cheaper than alteplase. Momentum is building, and many leading investigators are now conducting new studies with TNK with several more studies coming out in the next year or so,” Dr. Hill added.
The EXTEND-IA TNK Part 2 trial was supported by grants from the National Health and Medical Research Council of Australia and the National Heart Foundation of Australia. Campbell reports receiving grants from both institutions during the conduct of the study.
This article first appeared on Medscape.com.
ARCADIA: Predicting risk of atrial cardiopathy poststroke
LOS ANGELES – Older age, female sex, black race, relative anemia, and a history of cardiovascular disease are associated with greater risk for atrial cardiopathy among people who experienced an embolic stroke of undetermined source (ESUS), new evidence suggests.
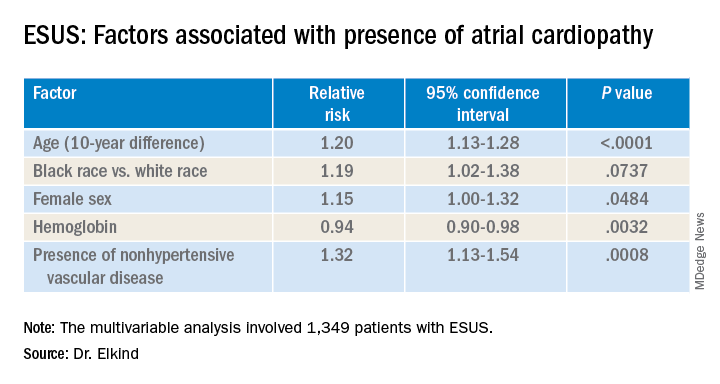
Atrial cardiopathy is a suspected cause of ESUS independent of atrial fibrillation. However, clinical predictors to help physicians identify which ESUS patients are at increased risk remain unknown.
The risk for atrial cardiopathy was 34% higher for women versus men with ESUS in this analysis. In addition, black participants had a 29% increased risk, compared with others, and each 10 years of age increased risk for atrial cardiopathy by 30% in an univariable analysis.
“Modest effects of these associations suggest that all ESUS patients, regardless of underlying demographic and risk factors, may have atrial cardiopathy,” principal investigator Mitchell S.V. Elkind, MD, of Columbia University, New York, said when presenting results at the 2020 International Stroke Conference, sponsored by the American Heart Association.
For this reason, he added, all people with ESUS should be considered for recruitment into the ongoing ARCADIA (AtRial Cardiopathy and Antithrombotic Drugs In Prevention After Cryptogenic Stroke) trial, of which he is one of the principal investigators.
ESUS is a heterogeneous condition, and some patients may be responsive to anticoagulants and some might not, Elkind said. This observation “led us to consider alternative ways for ischemic disease to lead to stroke. We would hypothesize that the underlying atrium can be a risk for stroke by itself.”
Not yet available is the primary efficacy outcome of the multicenter, randomized ARCADIA trial comparing apixaban with aspirin in reducing risk for recurrent stroke of any type. However, Dr. Elkind and colleagues have recruited 1,505 patients to date, enough to analyze factors that predict risk for recurrent stroke among people with evidence of atrial cardiopathy.
All ARCADIA participants are 45 years of age or older and have no history of atrial fibrillation. Atrial cardiopathy was defined by presence of at least one of three biomarkers: N-terminal pro-brain natriuretic peptide (NT-proBNP), P wave terminal force velocity, or evidence of a left atrial diameter of 3 cm/m2 or larger on echocardiography.
Of the 1,349 ARCADIA participants eligible for the current analysis, approximately one-third met one or more of these criteria for atrial cardiopathy.
Those with atrial cardiopathy were “more likely to be black and be women, and tended to have shorter time from stroke to screening,” Dr. Elkind said. In addition, heart failure, hypertension, and peripheral artery disease were more common in those with atrial cardiopathy. This group also was more likely to have an elevation in creatinine and lower hemoglobin and hematocrit levels.
“Heart disease, ischemic heart disease and non-hypertensive vascular disease were significant risk factors” for recurrent stroke in the study, Dr. Elkind added.
Elkind said that, surprisingly, there was no independent association between the time to measurement of NT-proBNP and risk, suggesting that this biomarker “does not rise simply in response to stroke, but reflects a stable condition.”
The multicenter ARCADIA trial is recruiting additional participants at 142 sites now, Dr. Elkind said, “and we are still looking for more sites.”
Which comes first?
“He is looking at what the predictors are for cardiopathy in these patients, which is fascinating for all of us,” session moderator Michelle Christina Johansen, MD, assistant professor of neurology at Johns Hopkins University, Baltimore, said in an interview when asked to comment.
There is always the conundrum of what came first — the chicken or the egg, Johansen said. Do these patients have stroke that then somehow led to a state that predisposes them to have atrial cardiopathy? Or, rather, was it an atrial cardiopathy state independent of atrial fibrillation that then led to stroke?
“That is why looking at predictors in this population is of such interest,” she said. The study could help identify a subgroup of patients at higher risk for atrial cardiopathy and guide clinical decision-making when patients present with ESUS.
“One of the things I found interesting was that he found that atrial cardiopathy patients were older [a mean 69 years]. This was amazing, because ESUS patients in general tend to be younger,” Dr. Johansen said.
“And there is about a 4-5% risk of recurrence with these patients. So. it was interesting that prior stroke or [transient ischemic attack] was not associated.”*
The National Institute of Neurological Disorders and Stroke, the BMS-Pfizer Alliance, and Roche provide funding for ARCADIA. Dr. Elkind and Dr. Johansen disclosed no relevant financial relationships.
SOURCE: Elkind M et al. ISC 2020, Abstract 26.
This article first appeared on Medscape.com.
*Correction, 4/28/20: An earlier version of this article misstated the risk of recurrence.
LOS ANGELES – Older age, female sex, black race, relative anemia, and a history of cardiovascular disease are associated with greater risk for atrial cardiopathy among people who experienced an embolic stroke of undetermined source (ESUS), new evidence suggests.

Atrial cardiopathy is a suspected cause of ESUS independent of atrial fibrillation. However, clinical predictors to help physicians identify which ESUS patients are at increased risk remain unknown.
The risk for atrial cardiopathy was 34% higher for women versus men with ESUS in this analysis. In addition, black participants had a 29% increased risk, compared with others, and each 10 years of age increased risk for atrial cardiopathy by 30% in an univariable analysis.
“Modest effects of these associations suggest that all ESUS patients, regardless of underlying demographic and risk factors, may have atrial cardiopathy,” principal investigator Mitchell S.V. Elkind, MD, of Columbia University, New York, said when presenting results at the 2020 International Stroke Conference, sponsored by the American Heart Association.
For this reason, he added, all people with ESUS should be considered for recruitment into the ongoing ARCADIA (AtRial Cardiopathy and Antithrombotic Drugs In Prevention After Cryptogenic Stroke) trial, of which he is one of the principal investigators.
ESUS is a heterogeneous condition, and some patients may be responsive to anticoagulants and some might not, Elkind said. This observation “led us to consider alternative ways for ischemic disease to lead to stroke. We would hypothesize that the underlying atrium can be a risk for stroke by itself.”
Not yet available is the primary efficacy outcome of the multicenter, randomized ARCADIA trial comparing apixaban with aspirin in reducing risk for recurrent stroke of any type. However, Dr. Elkind and colleagues have recruited 1,505 patients to date, enough to analyze factors that predict risk for recurrent stroke among people with evidence of atrial cardiopathy.
All ARCADIA participants are 45 years of age or older and have no history of atrial fibrillation. Atrial cardiopathy was defined by presence of at least one of three biomarkers: N-terminal pro-brain natriuretic peptide (NT-proBNP), P wave terminal force velocity, or evidence of a left atrial diameter of 3 cm/m2 or larger on echocardiography.
Of the 1,349 ARCADIA participants eligible for the current analysis, approximately one-third met one or more of these criteria for atrial cardiopathy.
Those with atrial cardiopathy were “more likely to be black and be women, and tended to have shorter time from stroke to screening,” Dr. Elkind said. In addition, heart failure, hypertension, and peripheral artery disease were more common in those with atrial cardiopathy. This group also was more likely to have an elevation in creatinine and lower hemoglobin and hematocrit levels.
“Heart disease, ischemic heart disease and non-hypertensive vascular disease were significant risk factors” for recurrent stroke in the study, Dr. Elkind added.
Elkind said that, surprisingly, there was no independent association between the time to measurement of NT-proBNP and risk, suggesting that this biomarker “does not rise simply in response to stroke, but reflects a stable condition.”
The multicenter ARCADIA trial is recruiting additional participants at 142 sites now, Dr. Elkind said, “and we are still looking for more sites.”
Which comes first?
“He is looking at what the predictors are for cardiopathy in these patients, which is fascinating for all of us,” session moderator Michelle Christina Johansen, MD, assistant professor of neurology at Johns Hopkins University, Baltimore, said in an interview when asked to comment.
There is always the conundrum of what came first — the chicken or the egg, Johansen said. Do these patients have stroke that then somehow led to a state that predisposes them to have atrial cardiopathy? Or, rather, was it an atrial cardiopathy state independent of atrial fibrillation that then led to stroke?
“That is why looking at predictors in this population is of such interest,” she said. The study could help identify a subgroup of patients at higher risk for atrial cardiopathy and guide clinical decision-making when patients present with ESUS.
“One of the things I found interesting was that he found that atrial cardiopathy patients were older [a mean 69 years]. This was amazing, because ESUS patients in general tend to be younger,” Dr. Johansen said.
“And there is about a 4-5% risk of recurrence with these patients. So. it was interesting that prior stroke or [transient ischemic attack] was not associated.”*
The National Institute of Neurological Disorders and Stroke, the BMS-Pfizer Alliance, and Roche provide funding for ARCADIA. Dr. Elkind and Dr. Johansen disclosed no relevant financial relationships.
SOURCE: Elkind M et al. ISC 2020, Abstract 26.
This article first appeared on Medscape.com.
*Correction, 4/28/20: An earlier version of this article misstated the risk of recurrence.
LOS ANGELES – Older age, female sex, black race, relative anemia, and a history of cardiovascular disease are associated with greater risk for atrial cardiopathy among people who experienced an embolic stroke of undetermined source (ESUS), new evidence suggests.

Atrial cardiopathy is a suspected cause of ESUS independent of atrial fibrillation. However, clinical predictors to help physicians identify which ESUS patients are at increased risk remain unknown.
The risk for atrial cardiopathy was 34% higher for women versus men with ESUS in this analysis. In addition, black participants had a 29% increased risk, compared with others, and each 10 years of age increased risk for atrial cardiopathy by 30% in an univariable analysis.
“Modest effects of these associations suggest that all ESUS patients, regardless of underlying demographic and risk factors, may have atrial cardiopathy,” principal investigator Mitchell S.V. Elkind, MD, of Columbia University, New York, said when presenting results at the 2020 International Stroke Conference, sponsored by the American Heart Association.
For this reason, he added, all people with ESUS should be considered for recruitment into the ongoing ARCADIA (AtRial Cardiopathy and Antithrombotic Drugs In Prevention After Cryptogenic Stroke) trial, of which he is one of the principal investigators.
ESUS is a heterogeneous condition, and some patients may be responsive to anticoagulants and some might not, Elkind said. This observation “led us to consider alternative ways for ischemic disease to lead to stroke. We would hypothesize that the underlying atrium can be a risk for stroke by itself.”
Not yet available is the primary efficacy outcome of the multicenter, randomized ARCADIA trial comparing apixaban with aspirin in reducing risk for recurrent stroke of any type. However, Dr. Elkind and colleagues have recruited 1,505 patients to date, enough to analyze factors that predict risk for recurrent stroke among people with evidence of atrial cardiopathy.
All ARCADIA participants are 45 years of age or older and have no history of atrial fibrillation. Atrial cardiopathy was defined by presence of at least one of three biomarkers: N-terminal pro-brain natriuretic peptide (NT-proBNP), P wave terminal force velocity, or evidence of a left atrial diameter of 3 cm/m2 or larger on echocardiography.
Of the 1,349 ARCADIA participants eligible for the current analysis, approximately one-third met one or more of these criteria for atrial cardiopathy.
Those with atrial cardiopathy were “more likely to be black and be women, and tended to have shorter time from stroke to screening,” Dr. Elkind said. In addition, heart failure, hypertension, and peripheral artery disease were more common in those with atrial cardiopathy. This group also was more likely to have an elevation in creatinine and lower hemoglobin and hematocrit levels.
“Heart disease, ischemic heart disease and non-hypertensive vascular disease were significant risk factors” for recurrent stroke in the study, Dr. Elkind added.
Elkind said that, surprisingly, there was no independent association between the time to measurement of NT-proBNP and risk, suggesting that this biomarker “does not rise simply in response to stroke, but reflects a stable condition.”
The multicenter ARCADIA trial is recruiting additional participants at 142 sites now, Dr. Elkind said, “and we are still looking for more sites.”
Which comes first?
“He is looking at what the predictors are for cardiopathy in these patients, which is fascinating for all of us,” session moderator Michelle Christina Johansen, MD, assistant professor of neurology at Johns Hopkins University, Baltimore, said in an interview when asked to comment.
There is always the conundrum of what came first — the chicken or the egg, Johansen said. Do these patients have stroke that then somehow led to a state that predisposes them to have atrial cardiopathy? Or, rather, was it an atrial cardiopathy state independent of atrial fibrillation that then led to stroke?
“That is why looking at predictors in this population is of such interest,” she said. The study could help identify a subgroup of patients at higher risk for atrial cardiopathy and guide clinical decision-making when patients present with ESUS.
“One of the things I found interesting was that he found that atrial cardiopathy patients were older [a mean 69 years]. This was amazing, because ESUS patients in general tend to be younger,” Dr. Johansen said.
“And there is about a 4-5% risk of recurrence with these patients. So. it was interesting that prior stroke or [transient ischemic attack] was not associated.”*
The National Institute of Neurological Disorders and Stroke, the BMS-Pfizer Alliance, and Roche provide funding for ARCADIA. Dr. Elkind and Dr. Johansen disclosed no relevant financial relationships.
SOURCE: Elkind M et al. ISC 2020, Abstract 26.
This article first appeared on Medscape.com.
*Correction, 4/28/20: An earlier version of this article misstated the risk of recurrence.
REPORTING FROM ISC 2020
Shingles vaccine linked to lower stroke risk
LOS ANGELES – Prevention of shingles with the Zoster Vaccine Live may reduce the risk of subsequent stroke among older adults as well, the first study to examine this association suggests. Shingles vaccination was linked to a 20% decrease in stroke risk in people younger than 80 years of age in the large Medicare cohort study. Older participants showed a 10% reduced risk, according to data released in advance of formal presentation at this week’s International Stroke Conference, sponsored by the American Heart Association.
Reductions were seen for both ischemic and hemorrhagic events.
“Our findings might encourage people age 50 or older to get vaccinated against shingles and to prevent shingles-associated stroke risk,” Quanhe Yang, PhD, lead study author and senior scientist at the Centers for Disease Control and Prevention, said in an interview.
Dr. Yang and colleagues evaluated the only shingles vaccine available at the time of the study, Zoster Vaccine Live (Zostavax). However, the CDC now calls an adjuvanted, nonlive recombinant vaccine (Shingrix) the preferred shingles vaccine for healthy adults aged 50 years and older. Shingrix was approved in 2017. Zostavax, approved in 2006, can still be used in healthy adults aged 60 years and older, the agency states.
A reduction in inflammation from Zoster Vaccine Live may be the mechanism by which stroke risk is reduced, Dr. Yang said. The newer vaccine, which the CDC notes is more than 90% effective, might provide even greater protection against stroke, although more research is needed, he added.
Interestingly, prior research suggested that, once a person develops shingles, it may be too late. Dr. Yang and colleagues showed vaccination or antiviral treatment after a shingles episode was not effective at reducing stroke risk in research presented at the 2019 International Stroke Conference.
Shingles can present as a painful reactivation of chickenpox, also known as the varicella-zoster virus. Shingles is also common; Dr. Yang estimated one in three people who had chickenpox will develop the condition at some point in their lifetime. In addition, researchers have linked shingles to an elevated risk of stroke.
To assess the vaccine’s protective effect on stroke, Dr. Yang and colleagues reviewed health records for 1.38 million Medicare recipients. All participants were aged 66 years or older, had no history of stroke at baseline, and received the Zoster Vaccine Live during 2008-2016. The investigators compared the stroke rate in this vaccinated group with the rate in a matched control group of the same number of Medicare fee-for-service beneficiaries who did not receive the vaccination. They adjusted their analysis for age, sex, race, medications, and comorbidities.
The overall decrease of 16% in stroke risk associated with vaccination included a 12% drop in hemorrhagic stroke and 18% decrease in ischemic stroke over a median follow-up of 3.9 years follow-up (interquartile range, 2.7-5.4).
The adjusted hazard ratios comparing the vaccinated with control groups were 0.84 (95% confidence interval, 0.83-0.85) for all stroke; 0.82 (95% CI, 0.81-0.83) for acute ischemic stroke; and 0.88 (95% CI, 0.84-0.91) for hemorrhagic stroke.
The vaccinated group experienced 42,267 stroke events during that time. This rate included 33,510 acute ischemic strokes and 4,318 hemorrhagic strokes. At the same time, 48,139 strokes occurred in the control group. The breakdown included 39,334 ischemic and 4,713 hemorrhagic events.
“Approximately 1 million people in the United States get shingles each year, yet there is a vaccine to help prevent it,” Dr. Yang stated in a news release. “Our study results may encourage people ages 50 and older to follow the recommendation and get vaccinated against shingles. You are reducing the risk of shingles, and at the same time, you may be reducing your risk of stroke.”
“Further studies are needed to confirm our findings of association between Zostavax vaccine and risk of stroke,” Dr. Yang said.
Because the CDC Advisory Committee on Immunization Practices recommended Shingrix vaccine only for healthy adults 50 years and older in 2017, there were insufficient data in Medicare to study the association between that vaccine and risk of stroke at the time of the current study.
“However, two doses of Shingrix are more than 90% effective at preventing shingles and postherpetic neuralgia, and higher than that of Zostavax,” Dr. Yang said.
‘Very intriguing’ research
“This is a very interesting study,” Ralph L. Sacco, MD, past president of the American Heart Association, said in a video commentary released in advance of the conference. It was a very large sample, he noted, and those older than age 60 years who had the vaccine were protected with a lower risk for both ischemic and hemorrhagic stroke.
“So it is very intriguing,” added Dr. Sacco, chairman of the department of neurology at the University of Miami. “We know things like shingles can increase inflammation and increase the risk of stroke,” Dr. Sacco said, “but this is the first time in a very large Medicare database that it was shown that those who had the vaccine had a lower risk of stroke.”
The CDC funded this study. Dr. Yang and Dr. Sacco have disclosed no relevant financial relationships.
SOURCE: Yang Q et al. ISC 2020, Abstract TP493.
This article first appeared on Medscape.com.
LOS ANGELES – Prevention of shingles with the Zoster Vaccine Live may reduce the risk of subsequent stroke among older adults as well, the first study to examine this association suggests. Shingles vaccination was linked to a 20% decrease in stroke risk in people younger than 80 years of age in the large Medicare cohort study. Older participants showed a 10% reduced risk, according to data released in advance of formal presentation at this week’s International Stroke Conference, sponsored by the American Heart Association.
Reductions were seen for both ischemic and hemorrhagic events.
“Our findings might encourage people age 50 or older to get vaccinated against shingles and to prevent shingles-associated stroke risk,” Quanhe Yang, PhD, lead study author and senior scientist at the Centers for Disease Control and Prevention, said in an interview.
Dr. Yang and colleagues evaluated the only shingles vaccine available at the time of the study, Zoster Vaccine Live (Zostavax). However, the CDC now calls an adjuvanted, nonlive recombinant vaccine (Shingrix) the preferred shingles vaccine for healthy adults aged 50 years and older. Shingrix was approved in 2017. Zostavax, approved in 2006, can still be used in healthy adults aged 60 years and older, the agency states.
A reduction in inflammation from Zoster Vaccine Live may be the mechanism by which stroke risk is reduced, Dr. Yang said. The newer vaccine, which the CDC notes is more than 90% effective, might provide even greater protection against stroke, although more research is needed, he added.
Interestingly, prior research suggested that, once a person develops shingles, it may be too late. Dr. Yang and colleagues showed vaccination or antiviral treatment after a shingles episode was not effective at reducing stroke risk in research presented at the 2019 International Stroke Conference.
Shingles can present as a painful reactivation of chickenpox, also known as the varicella-zoster virus. Shingles is also common; Dr. Yang estimated one in three people who had chickenpox will develop the condition at some point in their lifetime. In addition, researchers have linked shingles to an elevated risk of stroke.
To assess the vaccine’s protective effect on stroke, Dr. Yang and colleagues reviewed health records for 1.38 million Medicare recipients. All participants were aged 66 years or older, had no history of stroke at baseline, and received the Zoster Vaccine Live during 2008-2016. The investigators compared the stroke rate in this vaccinated group with the rate in a matched control group of the same number of Medicare fee-for-service beneficiaries who did not receive the vaccination. They adjusted their analysis for age, sex, race, medications, and comorbidities.
The overall decrease of 16% in stroke risk associated with vaccination included a 12% drop in hemorrhagic stroke and 18% decrease in ischemic stroke over a median follow-up of 3.9 years follow-up (interquartile range, 2.7-5.4).
The adjusted hazard ratios comparing the vaccinated with control groups were 0.84 (95% confidence interval, 0.83-0.85) for all stroke; 0.82 (95% CI, 0.81-0.83) for acute ischemic stroke; and 0.88 (95% CI, 0.84-0.91) for hemorrhagic stroke.
The vaccinated group experienced 42,267 stroke events during that time. This rate included 33,510 acute ischemic strokes and 4,318 hemorrhagic strokes. At the same time, 48,139 strokes occurred in the control group. The breakdown included 39,334 ischemic and 4,713 hemorrhagic events.
“Approximately 1 million people in the United States get shingles each year, yet there is a vaccine to help prevent it,” Dr. Yang stated in a news release. “Our study results may encourage people ages 50 and older to follow the recommendation and get vaccinated against shingles. You are reducing the risk of shingles, and at the same time, you may be reducing your risk of stroke.”
“Further studies are needed to confirm our findings of association between Zostavax vaccine and risk of stroke,” Dr. Yang said.
Because the CDC Advisory Committee on Immunization Practices recommended Shingrix vaccine only for healthy adults 50 years and older in 2017, there were insufficient data in Medicare to study the association between that vaccine and risk of stroke at the time of the current study.
“However, two doses of Shingrix are more than 90% effective at preventing shingles and postherpetic neuralgia, and higher than that of Zostavax,” Dr. Yang said.
‘Very intriguing’ research
“This is a very interesting study,” Ralph L. Sacco, MD, past president of the American Heart Association, said in a video commentary released in advance of the conference. It was a very large sample, he noted, and those older than age 60 years who had the vaccine were protected with a lower risk for both ischemic and hemorrhagic stroke.
“So it is very intriguing,” added Dr. Sacco, chairman of the department of neurology at the University of Miami. “We know things like shingles can increase inflammation and increase the risk of stroke,” Dr. Sacco said, “but this is the first time in a very large Medicare database that it was shown that those who had the vaccine had a lower risk of stroke.”
The CDC funded this study. Dr. Yang and Dr. Sacco have disclosed no relevant financial relationships.
SOURCE: Yang Q et al. ISC 2020, Abstract TP493.
This article first appeared on Medscape.com.
LOS ANGELES – Prevention of shingles with the Zoster Vaccine Live may reduce the risk of subsequent stroke among older adults as well, the first study to examine this association suggests. Shingles vaccination was linked to a 20% decrease in stroke risk in people younger than 80 years of age in the large Medicare cohort study. Older participants showed a 10% reduced risk, according to data released in advance of formal presentation at this week’s International Stroke Conference, sponsored by the American Heart Association.
Reductions were seen for both ischemic and hemorrhagic events.
“Our findings might encourage people age 50 or older to get vaccinated against shingles and to prevent shingles-associated stroke risk,” Quanhe Yang, PhD, lead study author and senior scientist at the Centers for Disease Control and Prevention, said in an interview.
Dr. Yang and colleagues evaluated the only shingles vaccine available at the time of the study, Zoster Vaccine Live (Zostavax). However, the CDC now calls an adjuvanted, nonlive recombinant vaccine (Shingrix) the preferred shingles vaccine for healthy adults aged 50 years and older. Shingrix was approved in 2017. Zostavax, approved in 2006, can still be used in healthy adults aged 60 years and older, the agency states.
A reduction in inflammation from Zoster Vaccine Live may be the mechanism by which stroke risk is reduced, Dr. Yang said. The newer vaccine, which the CDC notes is more than 90% effective, might provide even greater protection against stroke, although more research is needed, he added.
Interestingly, prior research suggested that, once a person develops shingles, it may be too late. Dr. Yang and colleagues showed vaccination or antiviral treatment after a shingles episode was not effective at reducing stroke risk in research presented at the 2019 International Stroke Conference.
Shingles can present as a painful reactivation of chickenpox, also known as the varicella-zoster virus. Shingles is also common; Dr. Yang estimated one in three people who had chickenpox will develop the condition at some point in their lifetime. In addition, researchers have linked shingles to an elevated risk of stroke.
To assess the vaccine’s protective effect on stroke, Dr. Yang and colleagues reviewed health records for 1.38 million Medicare recipients. All participants were aged 66 years or older, had no history of stroke at baseline, and received the Zoster Vaccine Live during 2008-2016. The investigators compared the stroke rate in this vaccinated group with the rate in a matched control group of the same number of Medicare fee-for-service beneficiaries who did not receive the vaccination. They adjusted their analysis for age, sex, race, medications, and comorbidities.
The overall decrease of 16% in stroke risk associated with vaccination included a 12% drop in hemorrhagic stroke and 18% decrease in ischemic stroke over a median follow-up of 3.9 years follow-up (interquartile range, 2.7-5.4).
The adjusted hazard ratios comparing the vaccinated with control groups were 0.84 (95% confidence interval, 0.83-0.85) for all stroke; 0.82 (95% CI, 0.81-0.83) for acute ischemic stroke; and 0.88 (95% CI, 0.84-0.91) for hemorrhagic stroke.
The vaccinated group experienced 42,267 stroke events during that time. This rate included 33,510 acute ischemic strokes and 4,318 hemorrhagic strokes. At the same time, 48,139 strokes occurred in the control group. The breakdown included 39,334 ischemic and 4,713 hemorrhagic events.
“Approximately 1 million people in the United States get shingles each year, yet there is a vaccine to help prevent it,” Dr. Yang stated in a news release. “Our study results may encourage people ages 50 and older to follow the recommendation and get vaccinated against shingles. You are reducing the risk of shingles, and at the same time, you may be reducing your risk of stroke.”
“Further studies are needed to confirm our findings of association between Zostavax vaccine and risk of stroke,” Dr. Yang said.
Because the CDC Advisory Committee on Immunization Practices recommended Shingrix vaccine only for healthy adults 50 years and older in 2017, there were insufficient data in Medicare to study the association between that vaccine and risk of stroke at the time of the current study.
“However, two doses of Shingrix are more than 90% effective at preventing shingles and postherpetic neuralgia, and higher than that of Zostavax,” Dr. Yang said.
‘Very intriguing’ research
“This is a very interesting study,” Ralph L. Sacco, MD, past president of the American Heart Association, said in a video commentary released in advance of the conference. It was a very large sample, he noted, and those older than age 60 years who had the vaccine were protected with a lower risk for both ischemic and hemorrhagic stroke.
“So it is very intriguing,” added Dr. Sacco, chairman of the department of neurology at the University of Miami. “We know things like shingles can increase inflammation and increase the risk of stroke,” Dr. Sacco said, “but this is the first time in a very large Medicare database that it was shown that those who had the vaccine had a lower risk of stroke.”
The CDC funded this study. Dr. Yang and Dr. Sacco have disclosed no relevant financial relationships.
SOURCE: Yang Q et al. ISC 2020, Abstract TP493.
This article first appeared on Medscape.com.
REPORTING FROM ISC 2020
Stroke risk tied to diabetic retinopathy may not be modifiable
LOS ANGELES – Evidence continues to mount that diabetic retinopathy predicts elevated risk for stroke.
In a new study with nearly 3,000 people, those with diabetic retinopathy were 60% more likely than others with diabetes to develop an incident stroke over time. Investigators also found that addressing glucose, lipids, and blood pressure levels did not mitigate this risk in this secondary analysis of the ACCORD Eye Study.
“We are not surprised with the finding that diabetic retinopathy increases the risk of stroke — as diabetic retinopathy is common microvascular disease that is an established risk factor for cardiovascular disease,” lead author Ka-Ho Wong, BS, MBA, said in an interview.
However, “we were surprised that none of the trial interventions mitigated this risk, in particular the intensive blood pressure reduction, because hypertension is the most important cause of microvascular disease,” he said. Mr. Wong is clinical research coordinator and lab manager of the de Havenon Lab at the University of Utah Health Hospitals and Clinics in Salt Lake City.
The study findings were released Feb. 12, 2020, in advance of formal presentation at the International Stroke Conference sponsored by the American Heart Association.
Common predictor of vascular disease
Diabetic retinopathy is the most common complication of diabetes mellitus, affecting up to 50% of people living with type 1 and type 2 diabetes. In addition, previous research suggests that macrovascular diabetes complications, including stroke, could share a common or synergistic pathway.
This small vessel damage in the eye also has been linked to an increased risk of adverse cardiac events, including heart failure, as previously reported by Medscape Medical News.
To find out more, Mr. Wong and colleagues analyzed 2,828 participants in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study. They compared the stroke risk between 874 people with diabetic retinopathy and another 1,954 diabetics without this complication. The average age was 62 years and 62% were men.
Diabetic neuropathy at baseline was diagnosed using the Early Treatment Diabetic Retinopathy Study Severity Scale using seven-field stereoscopic fundus photographs.
A total of 117 participants experienced a stroke during a mean follow-up of 5.4 years.
The investigators found that diabetic retinopathy was more common among patients who had a stroke (41%) versus 31% of those without a stroke (P = .016). The link between diabetic retinopathy and stroke remained in an analysis adjusted for multiple factors, including baseline age, gender, race, total cholesterol, A1c, smoking, and more. Risk remained elevated, with a hazard ratio of 1.60 (95% confidence interval, 1.10-2.32; P = .015).
Regarding the potential for modifying this risk, the association was unaffected among participants randomly assigned to the ACCORD glucose intervention (P = .305), lipid intervention (P = .546), or blood pressure intervention (P = .422).
The study was a secondary analysis, so information on stroke type and location were unavailable.
The big picture
“Diabetic retinopathy is associated with an increased risk of stroke, which suggests that the microvascular pathology inherent to diabetic retinopathy has larger cardiovascular implications,” the researchers noted.
Despite these findings, the researchers suggest that patients with diabetic retinopathy receive aggressive medical management to try to reduce their stroke risk.
“It’s important for everyone with diabetes to maintain good blood glucose control, and those with established diabetic retinopathy should pay particular attention to meeting all the stroke prevention guidelines that are established by the American Stroke Association,” said Mr. Wong.
“Patients with established diabetic retinopathy should pay particular attention to meeting all stroke prevention guidelines established by the [American Heart Association],” he added.
Mr. Wong and colleagues would like to expand on these findings. Pending grant application and funding support, they propose conducting a prospective, observational trial in stroke patients with baseline diabetic retinopathy. One aim would be to identify the most common mechanisms leading to stroke in this population, “which would have important implications for prevention efforts,” he said.
Consistent Findings
“The results of the study showing that having diabetic retinopathy is also associated with an increase in stroke really isn’t surprising. There have been other studies, population-based studies, done in the past, that have found a similar relationship,” Larry B. Goldstein, MD, said in a video commentary on the findings.
“The results are actually quite consistent with several other studies that have evaluated the same relationship,” added Dr. Goldstein, who is chair of the department of neurology and codirector of the Kentucky Neuroscience Institute, University of Kentucky HealthCare, Lexington.
Mr. Wong and Dr. Goldstein have disclosed no relevant financial relationships. The NIH’s National Institute of Neurological Disorders and Stroke funded the study.
This article first appeared on Medscape.com.
LOS ANGELES – Evidence continues to mount that diabetic retinopathy predicts elevated risk for stroke.
In a new study with nearly 3,000 people, those with diabetic retinopathy were 60% more likely than others with diabetes to develop an incident stroke over time. Investigators also found that addressing glucose, lipids, and blood pressure levels did not mitigate this risk in this secondary analysis of the ACCORD Eye Study.
“We are not surprised with the finding that diabetic retinopathy increases the risk of stroke — as diabetic retinopathy is common microvascular disease that is an established risk factor for cardiovascular disease,” lead author Ka-Ho Wong, BS, MBA, said in an interview.
However, “we were surprised that none of the trial interventions mitigated this risk, in particular the intensive blood pressure reduction, because hypertension is the most important cause of microvascular disease,” he said. Mr. Wong is clinical research coordinator and lab manager of the de Havenon Lab at the University of Utah Health Hospitals and Clinics in Salt Lake City.
The study findings were released Feb. 12, 2020, in advance of formal presentation at the International Stroke Conference sponsored by the American Heart Association.
Common predictor of vascular disease
Diabetic retinopathy is the most common complication of diabetes mellitus, affecting up to 50% of people living with type 1 and type 2 diabetes. In addition, previous research suggests that macrovascular diabetes complications, including stroke, could share a common or synergistic pathway.
This small vessel damage in the eye also has been linked to an increased risk of adverse cardiac events, including heart failure, as previously reported by Medscape Medical News.
To find out more, Mr. Wong and colleagues analyzed 2,828 participants in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study. They compared the stroke risk between 874 people with diabetic retinopathy and another 1,954 diabetics without this complication. The average age was 62 years and 62% were men.
Diabetic neuropathy at baseline was diagnosed using the Early Treatment Diabetic Retinopathy Study Severity Scale using seven-field stereoscopic fundus photographs.
A total of 117 participants experienced a stroke during a mean follow-up of 5.4 years.
The investigators found that diabetic retinopathy was more common among patients who had a stroke (41%) versus 31% of those without a stroke (P = .016). The link between diabetic retinopathy and stroke remained in an analysis adjusted for multiple factors, including baseline age, gender, race, total cholesterol, A1c, smoking, and more. Risk remained elevated, with a hazard ratio of 1.60 (95% confidence interval, 1.10-2.32; P = .015).
Regarding the potential for modifying this risk, the association was unaffected among participants randomly assigned to the ACCORD glucose intervention (P = .305), lipid intervention (P = .546), or blood pressure intervention (P = .422).
The study was a secondary analysis, so information on stroke type and location were unavailable.
The big picture
“Diabetic retinopathy is associated with an increased risk of stroke, which suggests that the microvascular pathology inherent to diabetic retinopathy has larger cardiovascular implications,” the researchers noted.
Despite these findings, the researchers suggest that patients with diabetic retinopathy receive aggressive medical management to try to reduce their stroke risk.
“It’s important for everyone with diabetes to maintain good blood glucose control, and those with established diabetic retinopathy should pay particular attention to meeting all the stroke prevention guidelines that are established by the American Stroke Association,” said Mr. Wong.
“Patients with established diabetic retinopathy should pay particular attention to meeting all stroke prevention guidelines established by the [American Heart Association],” he added.
Mr. Wong and colleagues would like to expand on these findings. Pending grant application and funding support, they propose conducting a prospective, observational trial in stroke patients with baseline diabetic retinopathy. One aim would be to identify the most common mechanisms leading to stroke in this population, “which would have important implications for prevention efforts,” he said.
Consistent Findings
“The results of the study showing that having diabetic retinopathy is also associated with an increase in stroke really isn’t surprising. There have been other studies, population-based studies, done in the past, that have found a similar relationship,” Larry B. Goldstein, MD, said in a video commentary on the findings.
“The results are actually quite consistent with several other studies that have evaluated the same relationship,” added Dr. Goldstein, who is chair of the department of neurology and codirector of the Kentucky Neuroscience Institute, University of Kentucky HealthCare, Lexington.
Mr. Wong and Dr. Goldstein have disclosed no relevant financial relationships. The NIH’s National Institute of Neurological Disorders and Stroke funded the study.
This article first appeared on Medscape.com.
LOS ANGELES – Evidence continues to mount that diabetic retinopathy predicts elevated risk for stroke.
In a new study with nearly 3,000 people, those with diabetic retinopathy were 60% more likely than others with diabetes to develop an incident stroke over time. Investigators also found that addressing glucose, lipids, and blood pressure levels did not mitigate this risk in this secondary analysis of the ACCORD Eye Study.
“We are not surprised with the finding that diabetic retinopathy increases the risk of stroke — as diabetic retinopathy is common microvascular disease that is an established risk factor for cardiovascular disease,” lead author Ka-Ho Wong, BS, MBA, said in an interview.
However, “we were surprised that none of the trial interventions mitigated this risk, in particular the intensive blood pressure reduction, because hypertension is the most important cause of microvascular disease,” he said. Mr. Wong is clinical research coordinator and lab manager of the de Havenon Lab at the University of Utah Health Hospitals and Clinics in Salt Lake City.
The study findings were released Feb. 12, 2020, in advance of formal presentation at the International Stroke Conference sponsored by the American Heart Association.
Common predictor of vascular disease
Diabetic retinopathy is the most common complication of diabetes mellitus, affecting up to 50% of people living with type 1 and type 2 diabetes. In addition, previous research suggests that macrovascular diabetes complications, including stroke, could share a common or synergistic pathway.
This small vessel damage in the eye also has been linked to an increased risk of adverse cardiac events, including heart failure, as previously reported by Medscape Medical News.
To find out more, Mr. Wong and colleagues analyzed 2,828 participants in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study. They compared the stroke risk between 874 people with diabetic retinopathy and another 1,954 diabetics without this complication. The average age was 62 years and 62% were men.
Diabetic neuropathy at baseline was diagnosed using the Early Treatment Diabetic Retinopathy Study Severity Scale using seven-field stereoscopic fundus photographs.
A total of 117 participants experienced a stroke during a mean follow-up of 5.4 years.
The investigators found that diabetic retinopathy was more common among patients who had a stroke (41%) versus 31% of those without a stroke (P = .016). The link between diabetic retinopathy and stroke remained in an analysis adjusted for multiple factors, including baseline age, gender, race, total cholesterol, A1c, smoking, and more. Risk remained elevated, with a hazard ratio of 1.60 (95% confidence interval, 1.10-2.32; P = .015).
Regarding the potential for modifying this risk, the association was unaffected among participants randomly assigned to the ACCORD glucose intervention (P = .305), lipid intervention (P = .546), or blood pressure intervention (P = .422).
The study was a secondary analysis, so information on stroke type and location were unavailable.
The big picture
“Diabetic retinopathy is associated with an increased risk of stroke, which suggests that the microvascular pathology inherent to diabetic retinopathy has larger cardiovascular implications,” the researchers noted.
Despite these findings, the researchers suggest that patients with diabetic retinopathy receive aggressive medical management to try to reduce their stroke risk.
“It’s important for everyone with diabetes to maintain good blood glucose control, and those with established diabetic retinopathy should pay particular attention to meeting all the stroke prevention guidelines that are established by the American Stroke Association,” said Mr. Wong.
“Patients with established diabetic retinopathy should pay particular attention to meeting all stroke prevention guidelines established by the [American Heart Association],” he added.
Mr. Wong and colleagues would like to expand on these findings. Pending grant application and funding support, they propose conducting a prospective, observational trial in stroke patients with baseline diabetic retinopathy. One aim would be to identify the most common mechanisms leading to stroke in this population, “which would have important implications for prevention efforts,” he said.
Consistent Findings
“The results of the study showing that having diabetic retinopathy is also associated with an increase in stroke really isn’t surprising. There have been other studies, population-based studies, done in the past, that have found a similar relationship,” Larry B. Goldstein, MD, said in a video commentary on the findings.
“The results are actually quite consistent with several other studies that have evaluated the same relationship,” added Dr. Goldstein, who is chair of the department of neurology and codirector of the Kentucky Neuroscience Institute, University of Kentucky HealthCare, Lexington.
Mr. Wong and Dr. Goldstein have disclosed no relevant financial relationships. The NIH’s National Institute of Neurological Disorders and Stroke funded the study.
This article first appeared on Medscape.com.
REPORTING FROM ISC 2020
Carotid endarterectomy surpasses stenting in elderly, asymptomatic patients
LOS ANGELES – Carotid artery stenting in older, asymptomatic patients with severe carotid artery stenosis is, in general, as bad an idea as it has already proven to be in symptomatic patients, with a multifold increase in adverse short- and mid-term outcomes, compared with similar older, asymptomatic patients who underwent endarterectomy, according to a combined-study analysis with more than 2,500 patients.
The risk for poor outcomes in patients with severe but asymptomatic carotid artery disease who underwent carotid artery stenting (CAS), compared with patients who instead underwent carotid endarterectomy (CEA) “abruptly increased around age 75,” in an analysis that combined data from the two major, published, randomized trials that compared these two interventions in this patient population, Jenifer H. Voeks, PhD said at the International Stroke Conference sponsored by the American Heart Association.
These results “largely mirror” the findings from a similar combined analysis of data from four major, randomized trials that compared CEA and CAS in patients with symptomatic carotid disease, she noted (Lancet. 2016 Mar 26;387[10025]:1305-11). The new findings in an expanded population of asymptomatic patients derived from two separate studies showed that, in patients aged 70 years or less, “CAS appears to be a reasonable alternative to CEA, but above age 70, and certainly above age 75, age-related risk factors such as cerebrovascular anatomy and underlying cerebral pathology should be carefully considered before selecting patients for CAS,” said Dr. Voeks, a neurology researcher at the Medical University of South Carolina, Charleston. Many experts also believe that, for asymptomatic patients, intensive medical management may have returned as an alternative to either of these invasive approaches for treating severe carotid stenosis and has achieved a level of equipoise that led to the launch of CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial). CREST 2 is comparing CEA and CAS with medical management, and is scheduled to report results in 2021.
The data for this analysis in asymptomatic patients came from the first CREST (Carotid Revascularization Endarterectomy Versus Stenting Trial; N Engl J Med. 2010 Jul 1;363[1]:11-23), which included 1,181 asymptomatic patients (nearly half the total enrollment, with symptomatic patients making up the balance) and had no age ceiling, as well as all 1,453 patients from the ACT 1 trial, which enrolled exclusively asymptomatic patients and limited enrollment to patients aged 79 years or less (N Engl J Med. 2016 Mar 17;374[11]: 1011-20). Because the maximum age of patients in ACT 1 was 79 years, for this analysis Dr. Voeks and associates only included the 1,091 asymptomatic CREST patients who also were within the same age ceiling. The resulting cohort of 2,544 included 1,637 patients who underwent CAS and 907 who underwent CEA (because of a 3:1 randomization ratio in ACT 1), creating the largest data set to compare CAS and CEA by age in asymptomatic patients, Dr. Voeks noted. When subdivided by age, 30% of the cohort was younger that 65 years, 54% were 65-74, and 16% were 75-79.
The primary outcome the researchers used for their analysis was the combined incidence of periprocedural stroke, MI, or death, plus the incidence of ipsilateral stroke during 4 years of follow-up post procedure. Among patients who underwent CAS, this outcome occurred in roughly 9% of patients aged 75-79 years and in about 3% of those younger than 65 years, a hazard ratio of 2.9 that was statistically significant. In contrast, the incidence of the primary outcome among patients aged 65-74 years was just 30% higher, compared with patients aged less than 65 years, a difference that was not statistically significant.
Patients who underwent CEA showed no similar relationship between age and outcome. The incidence of the primary outcome among the CEA patients was roughly the same, about 3.5%, regardless of their age.
A second analysis that considered age as a continuous variable showed a sharply spiked increase in the risk for CAS patients, compared with CEA patients once they reached about age 73-75 years. Until about age 72, the rate of the primary outcome was nearly the same regardless of whether patients underwent CAS or CEA, but the risk for adverse outcomes rose “steeply” starting at about age 75 so that by age 79 the rate of the primary outcome approached 300% higher among the CAS patients compared with CEA patients, Dr. Voeks said.
She cautioned that the analysis included just 115 total primary-outcome events, which makes the incidence rate estimates somewhat imprecise, and that the data reflect outcomes in patients who were treated more than a decade ago, but these data remain the only reported results from large randomized trials that compared CAS and CEA in asymptomatic patients.
Dr. Voeks reported no disclosures.
SOURCE: Voeks JH al. Stroke. 2020 Feb 12;51[suppl 1], Abstract 70.
The role for carotid intervention in asymptomatic patients with severe carotid stenosis, usually defined as a stenosis that obstructs at least 70% of the carotid lumen, is controversial right now because intensive medical management has not been compared with invasive treatments, such as carotid endarterectomy and carotid stenting, for well over a decade. New drugs and new regimens have become treatment options for patients with advanced atherosclerotic carotid artery disease, and this has returned us to a state of equipoise for medical versus interventional management. That’s the premise behind CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial), which is comparing medical treatment against endarterectomy and against carotid stenting in a randomized study. The results may be available in 2021.
The new findings are very important for helping patients and their families make informed decisions. CAS is often perceived as the safer option for older patients because it is less traumatic and invasive than CEA. The data that Dr. Voeks reported show once again that this intuitive impression about CAS in the elderly is belied by the evidence. But the findings also require cautious interpretation because they came from a post hoc, subgroup analysis.
Mai N. Nguyen-Huynh, MD , is a vascular neurologist with Kaiser Permanente Northern California in Oakland. She had no relevant disclosures. She made these comments in an interview.
The role for carotid intervention in asymptomatic patients with severe carotid stenosis, usually defined as a stenosis that obstructs at least 70% of the carotid lumen, is controversial right now because intensive medical management has not been compared with invasive treatments, such as carotid endarterectomy and carotid stenting, for well over a decade. New drugs and new regimens have become treatment options for patients with advanced atherosclerotic carotid artery disease, and this has returned us to a state of equipoise for medical versus interventional management. That’s the premise behind CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial), which is comparing medical treatment against endarterectomy and against carotid stenting in a randomized study. The results may be available in 2021.
The new findings are very important for helping patients and their families make informed decisions. CAS is often perceived as the safer option for older patients because it is less traumatic and invasive than CEA. The data that Dr. Voeks reported show once again that this intuitive impression about CAS in the elderly is belied by the evidence. But the findings also require cautious interpretation because they came from a post hoc, subgroup analysis.
Mai N. Nguyen-Huynh, MD , is a vascular neurologist with Kaiser Permanente Northern California in Oakland. She had no relevant disclosures. She made these comments in an interview.
The role for carotid intervention in asymptomatic patients with severe carotid stenosis, usually defined as a stenosis that obstructs at least 70% of the carotid lumen, is controversial right now because intensive medical management has not been compared with invasive treatments, such as carotid endarterectomy and carotid stenting, for well over a decade. New drugs and new regimens have become treatment options for patients with advanced atherosclerotic carotid artery disease, and this has returned us to a state of equipoise for medical versus interventional management. That’s the premise behind CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial), which is comparing medical treatment against endarterectomy and against carotid stenting in a randomized study. The results may be available in 2021.
The new findings are very important for helping patients and their families make informed decisions. CAS is often perceived as the safer option for older patients because it is less traumatic and invasive than CEA. The data that Dr. Voeks reported show once again that this intuitive impression about CAS in the elderly is belied by the evidence. But the findings also require cautious interpretation because they came from a post hoc, subgroup analysis.
Mai N. Nguyen-Huynh, MD , is a vascular neurologist with Kaiser Permanente Northern California in Oakland. She had no relevant disclosures. She made these comments in an interview.
LOS ANGELES – Carotid artery stenting in older, asymptomatic patients with severe carotid artery stenosis is, in general, as bad an idea as it has already proven to be in symptomatic patients, with a multifold increase in adverse short- and mid-term outcomes, compared with similar older, asymptomatic patients who underwent endarterectomy, according to a combined-study analysis with more than 2,500 patients.
The risk for poor outcomes in patients with severe but asymptomatic carotid artery disease who underwent carotid artery stenting (CAS), compared with patients who instead underwent carotid endarterectomy (CEA) “abruptly increased around age 75,” in an analysis that combined data from the two major, published, randomized trials that compared these two interventions in this patient population, Jenifer H. Voeks, PhD said at the International Stroke Conference sponsored by the American Heart Association.
These results “largely mirror” the findings from a similar combined analysis of data from four major, randomized trials that compared CEA and CAS in patients with symptomatic carotid disease, she noted (Lancet. 2016 Mar 26;387[10025]:1305-11). The new findings in an expanded population of asymptomatic patients derived from two separate studies showed that, in patients aged 70 years or less, “CAS appears to be a reasonable alternative to CEA, but above age 70, and certainly above age 75, age-related risk factors such as cerebrovascular anatomy and underlying cerebral pathology should be carefully considered before selecting patients for CAS,” said Dr. Voeks, a neurology researcher at the Medical University of South Carolina, Charleston. Many experts also believe that, for asymptomatic patients, intensive medical management may have returned as an alternative to either of these invasive approaches for treating severe carotid stenosis and has achieved a level of equipoise that led to the launch of CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial). CREST 2 is comparing CEA and CAS with medical management, and is scheduled to report results in 2021.
The data for this analysis in asymptomatic patients came from the first CREST (Carotid Revascularization Endarterectomy Versus Stenting Trial; N Engl J Med. 2010 Jul 1;363[1]:11-23), which included 1,181 asymptomatic patients (nearly half the total enrollment, with symptomatic patients making up the balance) and had no age ceiling, as well as all 1,453 patients from the ACT 1 trial, which enrolled exclusively asymptomatic patients and limited enrollment to patients aged 79 years or less (N Engl J Med. 2016 Mar 17;374[11]: 1011-20). Because the maximum age of patients in ACT 1 was 79 years, for this analysis Dr. Voeks and associates only included the 1,091 asymptomatic CREST patients who also were within the same age ceiling. The resulting cohort of 2,544 included 1,637 patients who underwent CAS and 907 who underwent CEA (because of a 3:1 randomization ratio in ACT 1), creating the largest data set to compare CAS and CEA by age in asymptomatic patients, Dr. Voeks noted. When subdivided by age, 30% of the cohort was younger that 65 years, 54% were 65-74, and 16% were 75-79.
The primary outcome the researchers used for their analysis was the combined incidence of periprocedural stroke, MI, or death, plus the incidence of ipsilateral stroke during 4 years of follow-up post procedure. Among patients who underwent CAS, this outcome occurred in roughly 9% of patients aged 75-79 years and in about 3% of those younger than 65 years, a hazard ratio of 2.9 that was statistically significant. In contrast, the incidence of the primary outcome among patients aged 65-74 years was just 30% higher, compared with patients aged less than 65 years, a difference that was not statistically significant.
Patients who underwent CEA showed no similar relationship between age and outcome. The incidence of the primary outcome among the CEA patients was roughly the same, about 3.5%, regardless of their age.
A second analysis that considered age as a continuous variable showed a sharply spiked increase in the risk for CAS patients, compared with CEA patients once they reached about age 73-75 years. Until about age 72, the rate of the primary outcome was nearly the same regardless of whether patients underwent CAS or CEA, but the risk for adverse outcomes rose “steeply” starting at about age 75 so that by age 79 the rate of the primary outcome approached 300% higher among the CAS patients compared with CEA patients, Dr. Voeks said.
She cautioned that the analysis included just 115 total primary-outcome events, which makes the incidence rate estimates somewhat imprecise, and that the data reflect outcomes in patients who were treated more than a decade ago, but these data remain the only reported results from large randomized trials that compared CAS and CEA in asymptomatic patients.
Dr. Voeks reported no disclosures.
SOURCE: Voeks JH al. Stroke. 2020 Feb 12;51[suppl 1], Abstract 70.
LOS ANGELES – Carotid artery stenting in older, asymptomatic patients with severe carotid artery stenosis is, in general, as bad an idea as it has already proven to be in symptomatic patients, with a multifold increase in adverse short- and mid-term outcomes, compared with similar older, asymptomatic patients who underwent endarterectomy, according to a combined-study analysis with more than 2,500 patients.
The risk for poor outcomes in patients with severe but asymptomatic carotid artery disease who underwent carotid artery stenting (CAS), compared with patients who instead underwent carotid endarterectomy (CEA) “abruptly increased around age 75,” in an analysis that combined data from the two major, published, randomized trials that compared these two interventions in this patient population, Jenifer H. Voeks, PhD said at the International Stroke Conference sponsored by the American Heart Association.
These results “largely mirror” the findings from a similar combined analysis of data from four major, randomized trials that compared CEA and CAS in patients with symptomatic carotid disease, she noted (Lancet. 2016 Mar 26;387[10025]:1305-11). The new findings in an expanded population of asymptomatic patients derived from two separate studies showed that, in patients aged 70 years or less, “CAS appears to be a reasonable alternative to CEA, but above age 70, and certainly above age 75, age-related risk factors such as cerebrovascular anatomy and underlying cerebral pathology should be carefully considered before selecting patients for CAS,” said Dr. Voeks, a neurology researcher at the Medical University of South Carolina, Charleston. Many experts also believe that, for asymptomatic patients, intensive medical management may have returned as an alternative to either of these invasive approaches for treating severe carotid stenosis and has achieved a level of equipoise that led to the launch of CREST 2 (Carotid Revascularization and Medical Management for Asymptomatic Carotid Stenosis Trial). CREST 2 is comparing CEA and CAS with medical management, and is scheduled to report results in 2021.
The data for this analysis in asymptomatic patients came from the first CREST (Carotid Revascularization Endarterectomy Versus Stenting Trial; N Engl J Med. 2010 Jul 1;363[1]:11-23), which included 1,181 asymptomatic patients (nearly half the total enrollment, with symptomatic patients making up the balance) and had no age ceiling, as well as all 1,453 patients from the ACT 1 trial, which enrolled exclusively asymptomatic patients and limited enrollment to patients aged 79 years or less (N Engl J Med. 2016 Mar 17;374[11]: 1011-20). Because the maximum age of patients in ACT 1 was 79 years, for this analysis Dr. Voeks and associates only included the 1,091 asymptomatic CREST patients who also were within the same age ceiling. The resulting cohort of 2,544 included 1,637 patients who underwent CAS and 907 who underwent CEA (because of a 3:1 randomization ratio in ACT 1), creating the largest data set to compare CAS and CEA by age in asymptomatic patients, Dr. Voeks noted. When subdivided by age, 30% of the cohort was younger that 65 years, 54% were 65-74, and 16% were 75-79.
The primary outcome the researchers used for their analysis was the combined incidence of periprocedural stroke, MI, or death, plus the incidence of ipsilateral stroke during 4 years of follow-up post procedure. Among patients who underwent CAS, this outcome occurred in roughly 9% of patients aged 75-79 years and in about 3% of those younger than 65 years, a hazard ratio of 2.9 that was statistically significant. In contrast, the incidence of the primary outcome among patients aged 65-74 years was just 30% higher, compared with patients aged less than 65 years, a difference that was not statistically significant.
Patients who underwent CEA showed no similar relationship between age and outcome. The incidence of the primary outcome among the CEA patients was roughly the same, about 3.5%, regardless of their age.
A second analysis that considered age as a continuous variable showed a sharply spiked increase in the risk for CAS patients, compared with CEA patients once they reached about age 73-75 years. Until about age 72, the rate of the primary outcome was nearly the same regardless of whether patients underwent CAS or CEA, but the risk for adverse outcomes rose “steeply” starting at about age 75 so that by age 79 the rate of the primary outcome approached 300% higher among the CAS patients compared with CEA patients, Dr. Voeks said.
She cautioned that the analysis included just 115 total primary-outcome events, which makes the incidence rate estimates somewhat imprecise, and that the data reflect outcomes in patients who were treated more than a decade ago, but these data remain the only reported results from large randomized trials that compared CAS and CEA in asymptomatic patients.
Dr. Voeks reported no disclosures.
SOURCE: Voeks JH al. Stroke. 2020 Feb 12;51[suppl 1], Abstract 70.
REPORTING FROM ISC 2020
















