User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Is This Journal Legit? Predatory Publishers
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Online Diagnosis of Sexually Transmitted Infections? Ethicist Says We Are Nowhere Close
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There has been a large amount of news lately about dating online and dating apps. Probably the most common way younger people find potential partners is to go online and see who’s there that they might want to meet.
Online dating is also notorious for being full of scammers. There are all kinds of people out there that you have to be careful of, who are trying to rip you off by saying, “Send me money, I’m in trouble,” or “Now that we have a relationship, will you support my particular entrepreneurial idea?” Certainly, dangers are there.
Another danger we don’t talk much about is meeting people who have sexually transmitted diseases. That’s been a problem before websites and before dating apps. I think the opportunity of meeting more people — strangers, people you don’t really know — who may not tell you the truth about their health, and particularly their sexual health, is really out there.
It’s always good medical advice to tell people to practice safe sex, and that often involves a man wearing a condom. It certainly is the case that we want to attend not just to the prevention of unwanted pregnancy but also to the transmission of diseases. I think it’s very important to tell women of reproductive age to get their HPV shot to try to reduce cancers in their reproductive systems, or sometimes in men — anal cancers, or even being a transmitter of disease.
Even then, certainly one wants to recommend that, in an age where some people are going to meet many partners that they don’t know well or don’t have much background with, it’s wise to try to prevent diseases using the vaccines we’ve got, using the contraceptive methods that will prevent disease transmission, and reminding people to ask about sex life.
I did come across a website that just startled me. It’s called HeHealth, and basically it says to men, if you are conscientious about your sex life, take a picture of your penis, send it to us, and we have doctors — I presume they’re US doctors but I don’t know — who will diagnose venereal diseases based on that picture. I presume women could also say, “Before we have sex, or now that we’re approaching that possibility, I want you to send a picture to this company on this website.”
Now, a couple of reminders. I think we all know this, but just because you’re not manifesting symptoms on your reproductive organs doesn’t mean you don’t have a sexual disease. It’s not a reliable measure. Yes, maybe you could have somebody say: “Oh, that looks nasty. I’m not sure you ought to have sex right now, and maybe you should go get some treatment.” This is going to miss many cases and is not a reliable indicator that your partner is safe in terms of not transmitting diseases to you.
It also isn’t clear what they do with these images. Do they keep them? Who can see them? Could they resell them? What sort of privacy protection have you got if you decide to use this?
There’s another issue here, which is, if they misdiagnose someone and you do catch a sexual disease, who’s liable? Can you go after them for using doctors who weren’t competent or transmitting images that weren’t really adequate because you didn’t know how to take that picture properly when you sent that off to them? There are many unknowns.
The bottom line is that we’re in a different world, I think, of romance. We’re in a world where some people are going to meet more partners. Some people are going to meet more strangers. One approach is to have us take pictures of ourselves, send them off to who knows where, and ask for a green light to go ahead and have sexual relations. I don’t think we’re anywhere close to being able to rely on that as a way to avoid the risks of unprotected sexual behavior.
We do know what to do in dealing with patients who are sexually active. First, we have to ask them. Then we’ve got to recommend available vaccinations to prevent the transmission of some cancers, the HPV vaccine. Then they need that reminder about safe sexual practices not only to protect against unwanted pregnancy, but still, in this day and age, to protect against syphilis, which is on the rise, plus HIV, gonorrhea, chlamydia, and other sexually transmissible diseases.
I’m not going to rely on the penis picture to make the world safe for sex. I think we have to still use the old-fashioned techniques of education and prevention to do the best we can.
Dr. Caplan is director of the Division of Medical Ethics at New York University Langone Medical Center, New York City. He reported conflicts of interest with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
See the Medical World Through Neurodivergent Doctors’ Eyes
Some 15%-20% of the world’s population are neurodivergent, with conditions such as autism, dyslexia, Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), and others. With different strengths and challenges around learning, engaging socially, or completing certain tasks, neurodivergent people can face barriers in the workforce.
Meanwhile, studies suggest that neurodivergent people may be overrepresented in STEM fields such as medicine. The medical field may self-select for traits associated with neurodivergent conditions, researchers say, including a hyperfocus on intense interests, pattern recognition, increased curiosity and empathy, and thinking quickly under pressure.
But . They struggle with stigma, a culture of nondisclosure, and lack of accommodations, which can lead to burnout and poor mental health.
“The medical system and the mental health system are some of the spaces that are holding on tightly to some of the outdated understandings of things like autism and ADHD,” says Megan Anna Neff, PsyD, a psychologist with autism and ADHD based in Portland, Oregon.
Situations can get dire: A 2023 survey of more than 200 autistic doctors from several countries found that 77% had considered suicide and 24% had attempted it.
But here’s the crux of it: Many neurodivergent doctors believe their unique ways of thinking and outside-the-box creativity are skills and strengths that can benefit the field. And they say making medicine more inclusive — and better understanding how a neurodivergent physician’s brain works — would allow them to thrive.
Blending In and Breaking Down
The exact number of neurodivergent physicians in the workforce remains unknown. Existing studies are small and focus mainly on autism. But researchers believe the percentage could be higher than we think, because neurodiversity can be underidentified.
Although autism can sometimes be diagnosed as early as 18 months, it’s not uncommon to receive a diagnosis well into adulthood. “Like many late-identified autistic adults, I got my autism diagnosis in the context of autistic burnout,” says Melissa Houser, MD, a primary care physician who received a diagnosis in 2021. Dr. Houser, who uses the pronouns she/they, explains that her experience is common, “a consequence of chronically having your life’s demands exceed your capacity.”
Dr. Houser, who also has ADHD and dyslexia, among other neurodivergent conditions, says that before her diagnosis, she worked in a traditional practice setting. Eventually, she began to notice intense dysregulation and fatigue. “I began to have a lot more difficulties with communication and my motor planning and sequencing,” Dr. Houser says. “I was sleep-deprived, and my needs were not being met. I was in a situation where I had a complete lack of autonomy over my practice.”
Deep in burnout, Dr. Houser says she lost her ability to “mask,” a term used to describe how some neurodivergent people work to “blend in” with societal expectations. This led to further communication breakdowns with her supervisor. Finally, Dr. Houser saw a psychiatrist.
Shortly after her diagnosis, Dr. Houser quit her job and founded All Brains Belong, a nonprofit that provides neurodiversity-affirming medical care, education, and advocacy. Research has found that people with autism are at increased risk for physical health conditions, including immune conditions, gastrointestinal disorders, metabolic conditions, and increased mortality in hospital settings. Understanding these connections can “mean the difference between life and death” for neurodivergent patients, Dr. Houser says.
Yet, in a 2015 study that assessed providers’ ability to recognize autism, a high proportion were not aware that they had patients with autism spectrum disorder, and most reported lacking both the skills and the tools to care for them.
Different as a Doctor and a Patient
Bernadette Grosjean, MD, a retired associate professor of psychiatry at David Geffen School of Medicine at UCLA and a distinguished Fellow of the American Psychiatric Association, also found insight into lifelong experiences as both a doctor and a patient with her autism diagnosis, which came when she was 61.
“Looking back, I was a smart kid but kind of clumsy and different in other ways,” Dr. Grosjean says. According to a 2021 survey by Cambridge University, autistic individuals are significantly more likely to identify as LGBTQ+, and Dr. Grosjean, who is gay, says that not being fully accepted by family or friends played a role in her struggles with mental health issues.
Throughout her mental health treatment, Dr. Grosjean felt as though her providers “were expecting from me things that I didn’t know how to do or fix. I didn’t know how to be a ‘good’ patient,” she recalls.
As a psychiatrist, Dr. Grosjean started to notice that many of the women she treated for borderline personality disorder, which is categorized by unstable relationships and emotions, were autistic. “I then started asking lots of questions about myself — the fact that I’ve always been very sensitive or that I’ve been accused of being both hypersensitive and not having emotions, and I understood a lot.”
When Dr. Grosjean came across Autistic Doctors International, a group of over 800 autistic doctors worldwide, she says, “I found my tribe.” She now serves as the US lead for psychiatry for the group, which is focused on support, advocacy, research, and education around neurodiversity.
Psychiatric comorbidities can accompany neurodivergent conditions. But a growing body of research, including a 2022 study published in the European Archives of Psychiatry and Clinical Neuroscience, indicates that autism and ADHD are frequently misdiagnosed as depression or anxiety.
Dr. Neff was unaware of her conditions until one of her children was diagnosed with autism in 2021. She started to research it. “As I was learning about autism and girls, I was like, ‘Oh, my gosh, this is me,’ ” Dr. Neff recalls. Within a few weeks, she had her own diagnosis.
In hindsight, Dr. Neff has more clarity regarding her struggles in the traditional medical space. She had found it difficult to fit patients into short appointment windows and keep their notes concise. Although she loved hospital work, the environment had been overwhelming and led to burnout.
‘A Deficit-Based Lens’
Dr. Houser believes that too often, autism is viewed through a “deficit-based lens.” Stressors like sensory overload, changes in routine, or unexpected events can exacerbate behavioral challenges for neurodivergent people in the workplace. The DSM-5 criteria for autism, she points out, are largely based on autistic “stress behaviors.”
The result, Dr. Houser says, is that neurodivergent doctors are judged by their response to stressors that put them at a disadvantage rather than their capabilities under more positive circumstances. “The more dysregulated someone is,” she says, “the more likely they are to manifest those observable behaviors.”
Dr. Neff notes that medicine is a very “sensory overwhelming work environment.” Working in ob.gyn. and primary care clinics, she remembers often coming home with a headache and a low-grade fever. “I had no idea why, but I now realize it’s because I was so sensory sick.”
Fearing for her job, Dr. Neff intentionally waited until she was in private practice to disclose her neurodiversity. “I don’t think it would have been received well if I was in a hospital system,” she says. “There’s a lot of invalidation that can come when someone chooses to self-disclose, and their colleagues don’t have a framework in mind to understand.” In one instance, after revealing her diagnosis, she remembers a well-known researcher telling her she wasn’t autistic.
Dr. Grosjean has also had former colleagues invalidate her diagnosis, something she says “keeps people quiet.”
Understanding the Neurodivergent Brain
The general lack of education on how neurodivergent brains work, physicians with these conditions say, means they are not often recognized for how they can function with certain accommodations and how they could contribute in unique ways if their workplace challenges were reduced.
“What we know about autistic brains is that we are systems-thinking pattern matchers,” says Dr. Houser, who formed an interdisciplinary task force to explore medical conditions that are more common in autistic people. Through that comprehensive approach, she has worked to find best practices to treat the constellation of conditions that can arise among these patients. “My autistic brain allowed me to do that,” Dr. Houser says.
Catriona McVey, a medical student in the United Kingdom and creator of the blog Attention Deficit Doctor, points out that “ADHD brains are interest-driven; they can be very focused when you’re doing something enjoyable or new due to increased dopaminergic stimulation.” Ms. McVey speaks from personal experience. “I’ve hyperfocused before on an essay that interested me for over 10 hours,” she recalls, “so I imagine if I was interested in surgery, I could easily hyperfocus on a long operation.”
Empathy is another key part of medical practice. Contrary to stereotypes of neurodivergent people lacking empathy, current research suggests this isn’t true. A concept known as the “double empathy problem,” a term coined by British researcher Damian Milton in 2012, challenges the misconception that autistic people do not have empathy, explains Dr. Grosjean.
Mr. Milton theorized that there are two types of empathy: emotional, when you feel someone else’s pain, and cognitive, which involves critical thinking to understand someone’s emotions or thoughts. “Autistic people have, in general, a lot of emotional empathy,” Dr. Grosjean says, “but the cognitive empathy they don’t have as well.”
Dr. Neff has experienced this in her practice. “I will often feel what my clients are feeling as they’re feeling it,” she says, adding that she has always had an innate ability to analyze and connect with clients. She’s good at observing the interplay of health conditions, incorporating biology, psychology, and social conceptualizations of issues, with nuance. She feels that recognizing behavioral patterns or psychological triggers in her patients helps her see them holistically and provide better care. “That was a skill even before I realized I was autistic, but I always thought it was just intuitive to everyone,” she says.
Support Can Lead to Success
The Americans with Disabilities Act requires employers to provide reasonable accommodations to neurodivergent employees. However, getting those accommodations involves disclosure, which many physicians have reasons to avoid.
It also means more work. Requesting and putting adjustments in place can take a lot of time and energy to organize. Ms. McVey says they can be “long-winded, multistep tasks” that are not very compatible with ADHD. “Some doctors report that service pressures and funding are used as excuses to refuse adjustments,” she adds.
Ms. McVey lists several workplace accommodations that could be helpful, including flexible working hours, a quiet space to complete paperwork, dictation software, and extra time for medical students to complete written exams.
Neurodivergent physicians have also called for increased diversity of senior leadership and utilizing “cognitive apprenticeship models,” where employees explain their thought processes and receive timely feedback.
But far too often, there is little intervention until a doctor reaches a crisis point. “I look forward to the day when we don’t have to wait until people are profoundly depleted to discover how their brains work,” says Dr. Houser.
Beyond logistical and structural changes in the medical field, Dr. Grosjean speaks of the simple need to listen to colleagues with an open mind and believe them when they express their feelings and experiences. “Everyone has a role to play in challenging stigma, misconceptions, and stereotypes,” Ms. McVey agrees. Ask yourself the old question, she suggests: “If not me, then who? If not now, then when?”
A version of this article first appeared on Medscape.com.
Some 15%-20% of the world’s population are neurodivergent, with conditions such as autism, dyslexia, Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), and others. With different strengths and challenges around learning, engaging socially, or completing certain tasks, neurodivergent people can face barriers in the workforce.
Meanwhile, studies suggest that neurodivergent people may be overrepresented in STEM fields such as medicine. The medical field may self-select for traits associated with neurodivergent conditions, researchers say, including a hyperfocus on intense interests, pattern recognition, increased curiosity and empathy, and thinking quickly under pressure.
But . They struggle with stigma, a culture of nondisclosure, and lack of accommodations, which can lead to burnout and poor mental health.
“The medical system and the mental health system are some of the spaces that are holding on tightly to some of the outdated understandings of things like autism and ADHD,” says Megan Anna Neff, PsyD, a psychologist with autism and ADHD based in Portland, Oregon.
Situations can get dire: A 2023 survey of more than 200 autistic doctors from several countries found that 77% had considered suicide and 24% had attempted it.
But here’s the crux of it: Many neurodivergent doctors believe their unique ways of thinking and outside-the-box creativity are skills and strengths that can benefit the field. And they say making medicine more inclusive — and better understanding how a neurodivergent physician’s brain works — would allow them to thrive.
Blending In and Breaking Down
The exact number of neurodivergent physicians in the workforce remains unknown. Existing studies are small and focus mainly on autism. But researchers believe the percentage could be higher than we think, because neurodiversity can be underidentified.
Although autism can sometimes be diagnosed as early as 18 months, it’s not uncommon to receive a diagnosis well into adulthood. “Like many late-identified autistic adults, I got my autism diagnosis in the context of autistic burnout,” says Melissa Houser, MD, a primary care physician who received a diagnosis in 2021. Dr. Houser, who uses the pronouns she/they, explains that her experience is common, “a consequence of chronically having your life’s demands exceed your capacity.”
Dr. Houser, who also has ADHD and dyslexia, among other neurodivergent conditions, says that before her diagnosis, she worked in a traditional practice setting. Eventually, she began to notice intense dysregulation and fatigue. “I began to have a lot more difficulties with communication and my motor planning and sequencing,” Dr. Houser says. “I was sleep-deprived, and my needs were not being met. I was in a situation where I had a complete lack of autonomy over my practice.”
Deep in burnout, Dr. Houser says she lost her ability to “mask,” a term used to describe how some neurodivergent people work to “blend in” with societal expectations. This led to further communication breakdowns with her supervisor. Finally, Dr. Houser saw a psychiatrist.
Shortly after her diagnosis, Dr. Houser quit her job and founded All Brains Belong, a nonprofit that provides neurodiversity-affirming medical care, education, and advocacy. Research has found that people with autism are at increased risk for physical health conditions, including immune conditions, gastrointestinal disorders, metabolic conditions, and increased mortality in hospital settings. Understanding these connections can “mean the difference between life and death” for neurodivergent patients, Dr. Houser says.
Yet, in a 2015 study that assessed providers’ ability to recognize autism, a high proportion were not aware that they had patients with autism spectrum disorder, and most reported lacking both the skills and the tools to care for them.
Different as a Doctor and a Patient
Bernadette Grosjean, MD, a retired associate professor of psychiatry at David Geffen School of Medicine at UCLA and a distinguished Fellow of the American Psychiatric Association, also found insight into lifelong experiences as both a doctor and a patient with her autism diagnosis, which came when she was 61.
“Looking back, I was a smart kid but kind of clumsy and different in other ways,” Dr. Grosjean says. According to a 2021 survey by Cambridge University, autistic individuals are significantly more likely to identify as LGBTQ+, and Dr. Grosjean, who is gay, says that not being fully accepted by family or friends played a role in her struggles with mental health issues.
Throughout her mental health treatment, Dr. Grosjean felt as though her providers “were expecting from me things that I didn’t know how to do or fix. I didn’t know how to be a ‘good’ patient,” she recalls.
As a psychiatrist, Dr. Grosjean started to notice that many of the women she treated for borderline personality disorder, which is categorized by unstable relationships and emotions, were autistic. “I then started asking lots of questions about myself — the fact that I’ve always been very sensitive or that I’ve been accused of being both hypersensitive and not having emotions, and I understood a lot.”
When Dr. Grosjean came across Autistic Doctors International, a group of over 800 autistic doctors worldwide, she says, “I found my tribe.” She now serves as the US lead for psychiatry for the group, which is focused on support, advocacy, research, and education around neurodiversity.
Psychiatric comorbidities can accompany neurodivergent conditions. But a growing body of research, including a 2022 study published in the European Archives of Psychiatry and Clinical Neuroscience, indicates that autism and ADHD are frequently misdiagnosed as depression or anxiety.
Dr. Neff was unaware of her conditions until one of her children was diagnosed with autism in 2021. She started to research it. “As I was learning about autism and girls, I was like, ‘Oh, my gosh, this is me,’ ” Dr. Neff recalls. Within a few weeks, she had her own diagnosis.
In hindsight, Dr. Neff has more clarity regarding her struggles in the traditional medical space. She had found it difficult to fit patients into short appointment windows and keep their notes concise. Although she loved hospital work, the environment had been overwhelming and led to burnout.
‘A Deficit-Based Lens’
Dr. Houser believes that too often, autism is viewed through a “deficit-based lens.” Stressors like sensory overload, changes in routine, or unexpected events can exacerbate behavioral challenges for neurodivergent people in the workplace. The DSM-5 criteria for autism, she points out, are largely based on autistic “stress behaviors.”
The result, Dr. Houser says, is that neurodivergent doctors are judged by their response to stressors that put them at a disadvantage rather than their capabilities under more positive circumstances. “The more dysregulated someone is,” she says, “the more likely they are to manifest those observable behaviors.”
Dr. Neff notes that medicine is a very “sensory overwhelming work environment.” Working in ob.gyn. and primary care clinics, she remembers often coming home with a headache and a low-grade fever. “I had no idea why, but I now realize it’s because I was so sensory sick.”
Fearing for her job, Dr. Neff intentionally waited until she was in private practice to disclose her neurodiversity. “I don’t think it would have been received well if I was in a hospital system,” she says. “There’s a lot of invalidation that can come when someone chooses to self-disclose, and their colleagues don’t have a framework in mind to understand.” In one instance, after revealing her diagnosis, she remembers a well-known researcher telling her she wasn’t autistic.
Dr. Grosjean has also had former colleagues invalidate her diagnosis, something she says “keeps people quiet.”
Understanding the Neurodivergent Brain
The general lack of education on how neurodivergent brains work, physicians with these conditions say, means they are not often recognized for how they can function with certain accommodations and how they could contribute in unique ways if their workplace challenges were reduced.
“What we know about autistic brains is that we are systems-thinking pattern matchers,” says Dr. Houser, who formed an interdisciplinary task force to explore medical conditions that are more common in autistic people. Through that comprehensive approach, she has worked to find best practices to treat the constellation of conditions that can arise among these patients. “My autistic brain allowed me to do that,” Dr. Houser says.
Catriona McVey, a medical student in the United Kingdom and creator of the blog Attention Deficit Doctor, points out that “ADHD brains are interest-driven; they can be very focused when you’re doing something enjoyable or new due to increased dopaminergic stimulation.” Ms. McVey speaks from personal experience. “I’ve hyperfocused before on an essay that interested me for over 10 hours,” she recalls, “so I imagine if I was interested in surgery, I could easily hyperfocus on a long operation.”
Empathy is another key part of medical practice. Contrary to stereotypes of neurodivergent people lacking empathy, current research suggests this isn’t true. A concept known as the “double empathy problem,” a term coined by British researcher Damian Milton in 2012, challenges the misconception that autistic people do not have empathy, explains Dr. Grosjean.
Mr. Milton theorized that there are two types of empathy: emotional, when you feel someone else’s pain, and cognitive, which involves critical thinking to understand someone’s emotions or thoughts. “Autistic people have, in general, a lot of emotional empathy,” Dr. Grosjean says, “but the cognitive empathy they don’t have as well.”
Dr. Neff has experienced this in her practice. “I will often feel what my clients are feeling as they’re feeling it,” she says, adding that she has always had an innate ability to analyze and connect with clients. She’s good at observing the interplay of health conditions, incorporating biology, psychology, and social conceptualizations of issues, with nuance. She feels that recognizing behavioral patterns or psychological triggers in her patients helps her see them holistically and provide better care. “That was a skill even before I realized I was autistic, but I always thought it was just intuitive to everyone,” she says.
Support Can Lead to Success
The Americans with Disabilities Act requires employers to provide reasonable accommodations to neurodivergent employees. However, getting those accommodations involves disclosure, which many physicians have reasons to avoid.
It also means more work. Requesting and putting adjustments in place can take a lot of time and energy to organize. Ms. McVey says they can be “long-winded, multistep tasks” that are not very compatible with ADHD. “Some doctors report that service pressures and funding are used as excuses to refuse adjustments,” she adds.
Ms. McVey lists several workplace accommodations that could be helpful, including flexible working hours, a quiet space to complete paperwork, dictation software, and extra time for medical students to complete written exams.
Neurodivergent physicians have also called for increased diversity of senior leadership and utilizing “cognitive apprenticeship models,” where employees explain their thought processes and receive timely feedback.
But far too often, there is little intervention until a doctor reaches a crisis point. “I look forward to the day when we don’t have to wait until people are profoundly depleted to discover how their brains work,” says Dr. Houser.
Beyond logistical and structural changes in the medical field, Dr. Grosjean speaks of the simple need to listen to colleagues with an open mind and believe them when they express their feelings and experiences. “Everyone has a role to play in challenging stigma, misconceptions, and stereotypes,” Ms. McVey agrees. Ask yourself the old question, she suggests: “If not me, then who? If not now, then when?”
A version of this article first appeared on Medscape.com.
Some 15%-20% of the world’s population are neurodivergent, with conditions such as autism, dyslexia, Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), and others. With different strengths and challenges around learning, engaging socially, or completing certain tasks, neurodivergent people can face barriers in the workforce.
Meanwhile, studies suggest that neurodivergent people may be overrepresented in STEM fields such as medicine. The medical field may self-select for traits associated with neurodivergent conditions, researchers say, including a hyperfocus on intense interests, pattern recognition, increased curiosity and empathy, and thinking quickly under pressure.
But . They struggle with stigma, a culture of nondisclosure, and lack of accommodations, which can lead to burnout and poor mental health.
“The medical system and the mental health system are some of the spaces that are holding on tightly to some of the outdated understandings of things like autism and ADHD,” says Megan Anna Neff, PsyD, a psychologist with autism and ADHD based in Portland, Oregon.
Situations can get dire: A 2023 survey of more than 200 autistic doctors from several countries found that 77% had considered suicide and 24% had attempted it.
But here’s the crux of it: Many neurodivergent doctors believe their unique ways of thinking and outside-the-box creativity are skills and strengths that can benefit the field. And they say making medicine more inclusive — and better understanding how a neurodivergent physician’s brain works — would allow them to thrive.
Blending In and Breaking Down
The exact number of neurodivergent physicians in the workforce remains unknown. Existing studies are small and focus mainly on autism. But researchers believe the percentage could be higher than we think, because neurodiversity can be underidentified.
Although autism can sometimes be diagnosed as early as 18 months, it’s not uncommon to receive a diagnosis well into adulthood. “Like many late-identified autistic adults, I got my autism diagnosis in the context of autistic burnout,” says Melissa Houser, MD, a primary care physician who received a diagnosis in 2021. Dr. Houser, who uses the pronouns she/they, explains that her experience is common, “a consequence of chronically having your life’s demands exceed your capacity.”
Dr. Houser, who also has ADHD and dyslexia, among other neurodivergent conditions, says that before her diagnosis, she worked in a traditional practice setting. Eventually, she began to notice intense dysregulation and fatigue. “I began to have a lot more difficulties with communication and my motor planning and sequencing,” Dr. Houser says. “I was sleep-deprived, and my needs were not being met. I was in a situation where I had a complete lack of autonomy over my practice.”
Deep in burnout, Dr. Houser says she lost her ability to “mask,” a term used to describe how some neurodivergent people work to “blend in” with societal expectations. This led to further communication breakdowns with her supervisor. Finally, Dr. Houser saw a psychiatrist.
Shortly after her diagnosis, Dr. Houser quit her job and founded All Brains Belong, a nonprofit that provides neurodiversity-affirming medical care, education, and advocacy. Research has found that people with autism are at increased risk for physical health conditions, including immune conditions, gastrointestinal disorders, metabolic conditions, and increased mortality in hospital settings. Understanding these connections can “mean the difference between life and death” for neurodivergent patients, Dr. Houser says.
Yet, in a 2015 study that assessed providers’ ability to recognize autism, a high proportion were not aware that they had patients with autism spectrum disorder, and most reported lacking both the skills and the tools to care for them.
Different as a Doctor and a Patient
Bernadette Grosjean, MD, a retired associate professor of psychiatry at David Geffen School of Medicine at UCLA and a distinguished Fellow of the American Psychiatric Association, also found insight into lifelong experiences as both a doctor and a patient with her autism diagnosis, which came when she was 61.
“Looking back, I was a smart kid but kind of clumsy and different in other ways,” Dr. Grosjean says. According to a 2021 survey by Cambridge University, autistic individuals are significantly more likely to identify as LGBTQ+, and Dr. Grosjean, who is gay, says that not being fully accepted by family or friends played a role in her struggles with mental health issues.
Throughout her mental health treatment, Dr. Grosjean felt as though her providers “were expecting from me things that I didn’t know how to do or fix. I didn’t know how to be a ‘good’ patient,” she recalls.
As a psychiatrist, Dr. Grosjean started to notice that many of the women she treated for borderline personality disorder, which is categorized by unstable relationships and emotions, were autistic. “I then started asking lots of questions about myself — the fact that I’ve always been very sensitive or that I’ve been accused of being both hypersensitive and not having emotions, and I understood a lot.”
When Dr. Grosjean came across Autistic Doctors International, a group of over 800 autistic doctors worldwide, she says, “I found my tribe.” She now serves as the US lead for psychiatry for the group, which is focused on support, advocacy, research, and education around neurodiversity.
Psychiatric comorbidities can accompany neurodivergent conditions. But a growing body of research, including a 2022 study published in the European Archives of Psychiatry and Clinical Neuroscience, indicates that autism and ADHD are frequently misdiagnosed as depression or anxiety.
Dr. Neff was unaware of her conditions until one of her children was diagnosed with autism in 2021. She started to research it. “As I was learning about autism and girls, I was like, ‘Oh, my gosh, this is me,’ ” Dr. Neff recalls. Within a few weeks, she had her own diagnosis.
In hindsight, Dr. Neff has more clarity regarding her struggles in the traditional medical space. She had found it difficult to fit patients into short appointment windows and keep their notes concise. Although she loved hospital work, the environment had been overwhelming and led to burnout.
‘A Deficit-Based Lens’
Dr. Houser believes that too often, autism is viewed through a “deficit-based lens.” Stressors like sensory overload, changes in routine, or unexpected events can exacerbate behavioral challenges for neurodivergent people in the workplace. The DSM-5 criteria for autism, she points out, are largely based on autistic “stress behaviors.”
The result, Dr. Houser says, is that neurodivergent doctors are judged by their response to stressors that put them at a disadvantage rather than their capabilities under more positive circumstances. “The more dysregulated someone is,” she says, “the more likely they are to manifest those observable behaviors.”
Dr. Neff notes that medicine is a very “sensory overwhelming work environment.” Working in ob.gyn. and primary care clinics, she remembers often coming home with a headache and a low-grade fever. “I had no idea why, but I now realize it’s because I was so sensory sick.”
Fearing for her job, Dr. Neff intentionally waited until she was in private practice to disclose her neurodiversity. “I don’t think it would have been received well if I was in a hospital system,” she says. “There’s a lot of invalidation that can come when someone chooses to self-disclose, and their colleagues don’t have a framework in mind to understand.” In one instance, after revealing her diagnosis, she remembers a well-known researcher telling her she wasn’t autistic.
Dr. Grosjean has also had former colleagues invalidate her diagnosis, something she says “keeps people quiet.”
Understanding the Neurodivergent Brain
The general lack of education on how neurodivergent brains work, physicians with these conditions say, means they are not often recognized for how they can function with certain accommodations and how they could contribute in unique ways if their workplace challenges were reduced.
“What we know about autistic brains is that we are systems-thinking pattern matchers,” says Dr. Houser, who formed an interdisciplinary task force to explore medical conditions that are more common in autistic people. Through that comprehensive approach, she has worked to find best practices to treat the constellation of conditions that can arise among these patients. “My autistic brain allowed me to do that,” Dr. Houser says.
Catriona McVey, a medical student in the United Kingdom and creator of the blog Attention Deficit Doctor, points out that “ADHD brains are interest-driven; they can be very focused when you’re doing something enjoyable or new due to increased dopaminergic stimulation.” Ms. McVey speaks from personal experience. “I’ve hyperfocused before on an essay that interested me for over 10 hours,” she recalls, “so I imagine if I was interested in surgery, I could easily hyperfocus on a long operation.”
Empathy is another key part of medical practice. Contrary to stereotypes of neurodivergent people lacking empathy, current research suggests this isn’t true. A concept known as the “double empathy problem,” a term coined by British researcher Damian Milton in 2012, challenges the misconception that autistic people do not have empathy, explains Dr. Grosjean.
Mr. Milton theorized that there are two types of empathy: emotional, when you feel someone else’s pain, and cognitive, which involves critical thinking to understand someone’s emotions or thoughts. “Autistic people have, in general, a lot of emotional empathy,” Dr. Grosjean says, “but the cognitive empathy they don’t have as well.”
Dr. Neff has experienced this in her practice. “I will often feel what my clients are feeling as they’re feeling it,” she says, adding that she has always had an innate ability to analyze and connect with clients. She’s good at observing the interplay of health conditions, incorporating biology, psychology, and social conceptualizations of issues, with nuance. She feels that recognizing behavioral patterns or psychological triggers in her patients helps her see them holistically and provide better care. “That was a skill even before I realized I was autistic, but I always thought it was just intuitive to everyone,” she says.
Support Can Lead to Success
The Americans with Disabilities Act requires employers to provide reasonable accommodations to neurodivergent employees. However, getting those accommodations involves disclosure, which many physicians have reasons to avoid.
It also means more work. Requesting and putting adjustments in place can take a lot of time and energy to organize. Ms. McVey says they can be “long-winded, multistep tasks” that are not very compatible with ADHD. “Some doctors report that service pressures and funding are used as excuses to refuse adjustments,” she adds.
Ms. McVey lists several workplace accommodations that could be helpful, including flexible working hours, a quiet space to complete paperwork, dictation software, and extra time for medical students to complete written exams.
Neurodivergent physicians have also called for increased diversity of senior leadership and utilizing “cognitive apprenticeship models,” where employees explain their thought processes and receive timely feedback.
But far too often, there is little intervention until a doctor reaches a crisis point. “I look forward to the day when we don’t have to wait until people are profoundly depleted to discover how their brains work,” says Dr. Houser.
Beyond logistical and structural changes in the medical field, Dr. Grosjean speaks of the simple need to listen to colleagues with an open mind and believe them when they express their feelings and experiences. “Everyone has a role to play in challenging stigma, misconceptions, and stereotypes,” Ms. McVey agrees. Ask yourself the old question, she suggests: “If not me, then who? If not now, then when?”
A version of this article first appeared on Medscape.com.
US Hospitals Prone to Cyberattacks Like One That Impacted Patient Care at Ascension, Experts Say
In the wake of a debilitating cyberattack against one of the nation’s largest health care systems, Marvin Ruckle, a nurse at an Ascension hospital in Wichita, Kansas, said he had a frightening experience: He nearly gave a baby “the wrong dose of narcotic” because of confusing paperwork.
A May 8 ransomware attack against Ascension, a Catholic health system with 140 hospitals in at least 10 states, locked providers out of systems that track and coordinate nearly every aspect of patient care. They include its systems for electronic health records, some phones, and ones “utilized to order certain tests, procedures and medications,” the company said in a May 9 statement.
More than a dozen doctors and nurses who work for the sprawling health system told Michigan Public and KFF Health News that patient care at its hospitals across the nation was compromised in the fallout of the cyberattack over the past several weeks. Clinicians working for hospitals in three states described harrowing lapses, including delayed or lost lab results, medication errors, and an absence of routine safety checks via technology to prevent potentially fatal mistakes.
Despite a precipitous rise in cyberattacks against the health sector in recent years, a weeks-long disruption of this magnitude is beyond what most health systems are prepared for, said John S. Clark, an associate chief pharmacy officer at the University of Michigan health system.
“I don’t believe that anyone is fully prepared,” he said. Most emergency management plans “are designed around long-term downtimes that are into one, two, or three days.”
Ascension in a public statement May 9 said its care teams were “trained for these kinds of disruptions,” but did not respond to questions in early June about whether it had prepared for longer periods of downtime. Ascension said June 14 it had restored access to electronic health records across its network, but that patient “medical records and other information collected between May 8” and when the service was restored “may be temporarily inaccessible as we work to update the portal with information collected during the system downtime.”
Ruckle said he “had no training” for the cyberattack.
Back to Paper
Lisa Watson, an intensive care unit nurse at Ascension Via Christi St. Francis hospital in Wichita, described her own close call. She said she nearly administered the wrong medication to a critically ill patient because she couldn’t scan it as she normally would. “My patient probably would have passed away had I not caught it,” she said.
Watson is no stranger to using paper for patients’ medical charts, saying she did so “for probably half of my career,” before electronic health records became ubiquitous in hospitals. What happened after the cyberattack was “by no means the same.”
“When we paper-charted, we had systems in place to get those orders to other departments in a timely manner,” she said, “and those have all gone away.”
Melissa LaRue, an ICU nurse at Ascension Saint Agnes Hospital in Baltimore, described a close call with “administering the wrong dosage” of a patient’s blood pressure medication. “Luckily,” she said, it was “triple-checked and remedied before that could happen. But I think the potential for harm is there when you have so much information and paperwork that you have to go through.”
Clinicians say their hospitals have relied on slapdash workarounds, using handwritten notes, faxes, sticky notes, and basic computer spreadsheets — many devised on the fly by doctors and nurses — to care for patients.
More than a dozen other nurses and doctors, some of them without union protections, at Ascension hospitals in Michigan recounted situations in which they say patient care was compromised. Those clinicians spoke on the condition that they not be named for fear of retaliation by their employer.
An Ascension hospital emergency room doctor in Detroit said a man on the city’s east side was given a dangerous narcotic intended for another patient because of a paperwork mix-up. As a result, the patient’s breathing slowed to the point that he had to be put on a ventilator. “We intubated him and we sent him to the ICU because he got the wrong medication.”
A nurse in a Michigan Ascension hospital ER said a woman with low blood sugar and “altered mental status” went into cardiac arrest and died after staff said they waited four hours for lab results they needed to determine how to treat her, but never received. “If I started having crushing chest pain in the middle of work and thought I was having a big one, I would grab someone to drive me down the street to another hospital,” the same ER nurse said.
Similar concerns reportedly led a travel nurse at an Ascension hospital in Indiana to quit. “I just want to warn those patients that are coming to any of the Ascension facilities that there will be delays in care. There is potential for error and for harm,” Justin Neisser told CBS4 in Indianapolis in May.
Several nurses and doctors at Ascension hospitals said they feared the errors they’ve witnessed since the cyberattack began could threaten their professional licenses. “This is how a RaDonda Vaught happens,” one nurse said, referring to the Tennessee nurse who was convicted of criminally negligent homicide in 2022 for a fatal drug error.
Reporters were not able to review records to verify clinicians’ claims because of privacy laws surrounding patients’ medical information that apply to health care professionals.
Ascension declined to answer questions about claims that care has been affected by the ransomware attack. “As we have made clear throughout this cyber attack which has impacted our system and our dedicated clinical providers, caring for our patients is our highest priority,” Sean Fitzpatrick, Ascension’s vice president of external communications, said via email on June 3. “We are confident that our care providers in our hospitals and facilities continue to provide quality medical care.”
The federal government requires hospitals to protect patients’ sensitive health data, according to cybersecurity experts. However, there are no federal requirements for hospitals to prevent or prepare for cyberattacks that could compromise their electronic systems.
Hospitals: ‘The No.1 Target of Ransomware’
“We’ve started to think about these as public health issues and disasters on the scale of earthquakes or hurricanes,” said Jeff Tully, a co-director of the Center for Healthcare Cybersecurity at the University of California-San Diego. “These types of cybersecurity incidents should be thought of as a matter of when, and not if.”
Josh Corman, a cybersecurity expert and advocate, said ransom crews regard hospitals as the perfect prey: “They have terrible security and they’ll pay. So almost immediately, hospitals went to the No. 1 target of ransomware.”
In 2023, the health sector experienced the largest share of ransomware attacks of 16 infrastructure sectors considered vital to national security or safety, according to an FBI report on internet crimes. In March, the federal Department of Health and Human Services said reported large breaches involving ransomware had jumped by 264% over the past five years.
A cyberattack this year on Change Healthcare, a unit of UnitedHealth Group’s Optum division that processes billions of health care transactions every year, crippled the business of providers, pharmacies, and hospitals.
In May, UnitedHealth Group CEO Andrew Witty told lawmakers the company paid a $22 million ransom as a result of the Change Healthcare attack — which occurred after hackers accessed a company portal that didn’t have multifactor authentication, a basic cybersecurity tool.
The Biden administration in recent months has pushed to bolster health care cybersecurity standards, but it’s not clear which new measures will be required.
In January, HHS nudged companies to improve email security, add multifactor authentication, and institute cybersecurity training and testing, among other voluntary measures. The Centers for Medicare & Medicaid Services is expected to release new requirements for hospitals, but the scope and timing are unclear. The same is true of an update HHS is expected to make to patient privacy regulations.
HHS said the voluntary measures “will inform the creation of new enforceable cybersecurity standards,” department spokesperson Jeff Nesbit said in a statement.
“The recent cyberattack at Ascension only underscores the need for everyone in the health care ecosystem to do their part to secure their systems and protect patients,” Nesbit said.
Meanwhile, lobbyists for the hospital industry contend cybersecurity mandates or penalties are misplaced and would curtail hospitals’ resources to fend off attacks.
“Hospitals and health systems are not the primary source of cyber risk exposure facing the health care sector,” the American Hospital Association, the largest lobbying group for U.S. hospitals, said in an April statement prepared for U.S. House lawmakers. Most large data breaches that hit hospitals in 2023 originated with third-party “business associates” or other health entities, including CMS itself, the AHA statement said.
Hospitals consolidating into large multistate health systems face increased risk of data breaches and ransomware attacks, according to one study. Ascension in 2022 was the third-largest hospital chain in the U.S. by number of beds, according to the most recent data from the federal Agency for Healthcare Research and Quality.
And while cybersecurity regulations can quickly become outdated, they can at least make it clear that if health systems fail to implement basic protections there “should be consequences for that,” Jim Bagian, a former director of the National Center for Patient Safety at the Veterans Health Administration, told Michigan Public’s Stateside.
Patients can pay the price when lapses occur. Those in hospital care face a greater likelihood of death during a cyberattack, according to researchers at the University of Minnesota School of Public Health.
Workers concerned about patient safety at Ascension hospitals in Michigan have called for the company to make changes.
“We implore Ascension to recognize the internal problems that continue to plague its hospitals, both publicly and transparently,” said Dina Carlisle, a nurse and the president of the OPEIU Local 40 union, which represents nurses at Ascension Providence Rochester. At least 125 staff members at that Ascension hospital have signed a petition asking administrators to temporarily reduce elective surgeries and nonemergency patient admissions, like under the protocols many hospitals adopted early in the covid-19 pandemic.
Watson, the Kansas ICU nurse, said in late May that nurses had urged management to bring in more nurses to help manage the workflow. “Everything that we say has fallen on deaf ears,” she said.
“It is very hard to be a nurse at Ascension right now,” Watson said in late May. “It is very hard to be a patient at Ascension right now.”
If you’re a patient or worker at an Ascension hospital and would like to tell KFF Health News about your experiences, click here to share your story with us.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
In the wake of a debilitating cyberattack against one of the nation’s largest health care systems, Marvin Ruckle, a nurse at an Ascension hospital in Wichita, Kansas, said he had a frightening experience: He nearly gave a baby “the wrong dose of narcotic” because of confusing paperwork.
A May 8 ransomware attack against Ascension, a Catholic health system with 140 hospitals in at least 10 states, locked providers out of systems that track and coordinate nearly every aspect of patient care. They include its systems for electronic health records, some phones, and ones “utilized to order certain tests, procedures and medications,” the company said in a May 9 statement.
More than a dozen doctors and nurses who work for the sprawling health system told Michigan Public and KFF Health News that patient care at its hospitals across the nation was compromised in the fallout of the cyberattack over the past several weeks. Clinicians working for hospitals in three states described harrowing lapses, including delayed or lost lab results, medication errors, and an absence of routine safety checks via technology to prevent potentially fatal mistakes.
Despite a precipitous rise in cyberattacks against the health sector in recent years, a weeks-long disruption of this magnitude is beyond what most health systems are prepared for, said John S. Clark, an associate chief pharmacy officer at the University of Michigan health system.
“I don’t believe that anyone is fully prepared,” he said. Most emergency management plans “are designed around long-term downtimes that are into one, two, or three days.”
Ascension in a public statement May 9 said its care teams were “trained for these kinds of disruptions,” but did not respond to questions in early June about whether it had prepared for longer periods of downtime. Ascension said June 14 it had restored access to electronic health records across its network, but that patient “medical records and other information collected between May 8” and when the service was restored “may be temporarily inaccessible as we work to update the portal with information collected during the system downtime.”
Ruckle said he “had no training” for the cyberattack.
Back to Paper
Lisa Watson, an intensive care unit nurse at Ascension Via Christi St. Francis hospital in Wichita, described her own close call. She said she nearly administered the wrong medication to a critically ill patient because she couldn’t scan it as she normally would. “My patient probably would have passed away had I not caught it,” she said.
Watson is no stranger to using paper for patients’ medical charts, saying she did so “for probably half of my career,” before electronic health records became ubiquitous in hospitals. What happened after the cyberattack was “by no means the same.”
“When we paper-charted, we had systems in place to get those orders to other departments in a timely manner,” she said, “and those have all gone away.”
Melissa LaRue, an ICU nurse at Ascension Saint Agnes Hospital in Baltimore, described a close call with “administering the wrong dosage” of a patient’s blood pressure medication. “Luckily,” she said, it was “triple-checked and remedied before that could happen. But I think the potential for harm is there when you have so much information and paperwork that you have to go through.”
Clinicians say their hospitals have relied on slapdash workarounds, using handwritten notes, faxes, sticky notes, and basic computer spreadsheets — many devised on the fly by doctors and nurses — to care for patients.
More than a dozen other nurses and doctors, some of them without union protections, at Ascension hospitals in Michigan recounted situations in which they say patient care was compromised. Those clinicians spoke on the condition that they not be named for fear of retaliation by their employer.
An Ascension hospital emergency room doctor in Detroit said a man on the city’s east side was given a dangerous narcotic intended for another patient because of a paperwork mix-up. As a result, the patient’s breathing slowed to the point that he had to be put on a ventilator. “We intubated him and we sent him to the ICU because he got the wrong medication.”
A nurse in a Michigan Ascension hospital ER said a woman with low blood sugar and “altered mental status” went into cardiac arrest and died after staff said they waited four hours for lab results they needed to determine how to treat her, but never received. “If I started having crushing chest pain in the middle of work and thought I was having a big one, I would grab someone to drive me down the street to another hospital,” the same ER nurse said.
Similar concerns reportedly led a travel nurse at an Ascension hospital in Indiana to quit. “I just want to warn those patients that are coming to any of the Ascension facilities that there will be delays in care. There is potential for error and for harm,” Justin Neisser told CBS4 in Indianapolis in May.
Several nurses and doctors at Ascension hospitals said they feared the errors they’ve witnessed since the cyberattack began could threaten their professional licenses. “This is how a RaDonda Vaught happens,” one nurse said, referring to the Tennessee nurse who was convicted of criminally negligent homicide in 2022 for a fatal drug error.
Reporters were not able to review records to verify clinicians’ claims because of privacy laws surrounding patients’ medical information that apply to health care professionals.
Ascension declined to answer questions about claims that care has been affected by the ransomware attack. “As we have made clear throughout this cyber attack which has impacted our system and our dedicated clinical providers, caring for our patients is our highest priority,” Sean Fitzpatrick, Ascension’s vice president of external communications, said via email on June 3. “We are confident that our care providers in our hospitals and facilities continue to provide quality medical care.”
The federal government requires hospitals to protect patients’ sensitive health data, according to cybersecurity experts. However, there are no federal requirements for hospitals to prevent or prepare for cyberattacks that could compromise their electronic systems.
Hospitals: ‘The No.1 Target of Ransomware’
“We’ve started to think about these as public health issues and disasters on the scale of earthquakes or hurricanes,” said Jeff Tully, a co-director of the Center for Healthcare Cybersecurity at the University of California-San Diego. “These types of cybersecurity incidents should be thought of as a matter of when, and not if.”
Josh Corman, a cybersecurity expert and advocate, said ransom crews regard hospitals as the perfect prey: “They have terrible security and they’ll pay. So almost immediately, hospitals went to the No. 1 target of ransomware.”
In 2023, the health sector experienced the largest share of ransomware attacks of 16 infrastructure sectors considered vital to national security or safety, according to an FBI report on internet crimes. In March, the federal Department of Health and Human Services said reported large breaches involving ransomware had jumped by 264% over the past five years.
A cyberattack this year on Change Healthcare, a unit of UnitedHealth Group’s Optum division that processes billions of health care transactions every year, crippled the business of providers, pharmacies, and hospitals.
In May, UnitedHealth Group CEO Andrew Witty told lawmakers the company paid a $22 million ransom as a result of the Change Healthcare attack — which occurred after hackers accessed a company portal that didn’t have multifactor authentication, a basic cybersecurity tool.
The Biden administration in recent months has pushed to bolster health care cybersecurity standards, but it’s not clear which new measures will be required.
In January, HHS nudged companies to improve email security, add multifactor authentication, and institute cybersecurity training and testing, among other voluntary measures. The Centers for Medicare & Medicaid Services is expected to release new requirements for hospitals, but the scope and timing are unclear. The same is true of an update HHS is expected to make to patient privacy regulations.
HHS said the voluntary measures “will inform the creation of new enforceable cybersecurity standards,” department spokesperson Jeff Nesbit said in a statement.
“The recent cyberattack at Ascension only underscores the need for everyone in the health care ecosystem to do their part to secure their systems and protect patients,” Nesbit said.
Meanwhile, lobbyists for the hospital industry contend cybersecurity mandates or penalties are misplaced and would curtail hospitals’ resources to fend off attacks.
“Hospitals and health systems are not the primary source of cyber risk exposure facing the health care sector,” the American Hospital Association, the largest lobbying group for U.S. hospitals, said in an April statement prepared for U.S. House lawmakers. Most large data breaches that hit hospitals in 2023 originated with third-party “business associates” or other health entities, including CMS itself, the AHA statement said.
Hospitals consolidating into large multistate health systems face increased risk of data breaches and ransomware attacks, according to one study. Ascension in 2022 was the third-largest hospital chain in the U.S. by number of beds, according to the most recent data from the federal Agency for Healthcare Research and Quality.
And while cybersecurity regulations can quickly become outdated, they can at least make it clear that if health systems fail to implement basic protections there “should be consequences for that,” Jim Bagian, a former director of the National Center for Patient Safety at the Veterans Health Administration, told Michigan Public’s Stateside.
Patients can pay the price when lapses occur. Those in hospital care face a greater likelihood of death during a cyberattack, according to researchers at the University of Minnesota School of Public Health.
Workers concerned about patient safety at Ascension hospitals in Michigan have called for the company to make changes.
“We implore Ascension to recognize the internal problems that continue to plague its hospitals, both publicly and transparently,” said Dina Carlisle, a nurse and the president of the OPEIU Local 40 union, which represents nurses at Ascension Providence Rochester. At least 125 staff members at that Ascension hospital have signed a petition asking administrators to temporarily reduce elective surgeries and nonemergency patient admissions, like under the protocols many hospitals adopted early in the covid-19 pandemic.
Watson, the Kansas ICU nurse, said in late May that nurses had urged management to bring in more nurses to help manage the workflow. “Everything that we say has fallen on deaf ears,” she said.
“It is very hard to be a nurse at Ascension right now,” Watson said in late May. “It is very hard to be a patient at Ascension right now.”
If you’re a patient or worker at an Ascension hospital and would like to tell KFF Health News about your experiences, click here to share your story with us.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
In the wake of a debilitating cyberattack against one of the nation’s largest health care systems, Marvin Ruckle, a nurse at an Ascension hospital in Wichita, Kansas, said he had a frightening experience: He nearly gave a baby “the wrong dose of narcotic” because of confusing paperwork.
A May 8 ransomware attack against Ascension, a Catholic health system with 140 hospitals in at least 10 states, locked providers out of systems that track and coordinate nearly every aspect of patient care. They include its systems for electronic health records, some phones, and ones “utilized to order certain tests, procedures and medications,” the company said in a May 9 statement.
More than a dozen doctors and nurses who work for the sprawling health system told Michigan Public and KFF Health News that patient care at its hospitals across the nation was compromised in the fallout of the cyberattack over the past several weeks. Clinicians working for hospitals in three states described harrowing lapses, including delayed or lost lab results, medication errors, and an absence of routine safety checks via technology to prevent potentially fatal mistakes.
Despite a precipitous rise in cyberattacks against the health sector in recent years, a weeks-long disruption of this magnitude is beyond what most health systems are prepared for, said John S. Clark, an associate chief pharmacy officer at the University of Michigan health system.
“I don’t believe that anyone is fully prepared,” he said. Most emergency management plans “are designed around long-term downtimes that are into one, two, or three days.”
Ascension in a public statement May 9 said its care teams were “trained for these kinds of disruptions,” but did not respond to questions in early June about whether it had prepared for longer periods of downtime. Ascension said June 14 it had restored access to electronic health records across its network, but that patient “medical records and other information collected between May 8” and when the service was restored “may be temporarily inaccessible as we work to update the portal with information collected during the system downtime.”
Ruckle said he “had no training” for the cyberattack.
Back to Paper
Lisa Watson, an intensive care unit nurse at Ascension Via Christi St. Francis hospital in Wichita, described her own close call. She said she nearly administered the wrong medication to a critically ill patient because she couldn’t scan it as she normally would. “My patient probably would have passed away had I not caught it,” she said.
Watson is no stranger to using paper for patients’ medical charts, saying she did so “for probably half of my career,” before electronic health records became ubiquitous in hospitals. What happened after the cyberattack was “by no means the same.”
“When we paper-charted, we had systems in place to get those orders to other departments in a timely manner,” she said, “and those have all gone away.”
Melissa LaRue, an ICU nurse at Ascension Saint Agnes Hospital in Baltimore, described a close call with “administering the wrong dosage” of a patient’s blood pressure medication. “Luckily,” she said, it was “triple-checked and remedied before that could happen. But I think the potential for harm is there when you have so much information and paperwork that you have to go through.”
Clinicians say their hospitals have relied on slapdash workarounds, using handwritten notes, faxes, sticky notes, and basic computer spreadsheets — many devised on the fly by doctors and nurses — to care for patients.
More than a dozen other nurses and doctors, some of them without union protections, at Ascension hospitals in Michigan recounted situations in which they say patient care was compromised. Those clinicians spoke on the condition that they not be named for fear of retaliation by their employer.
An Ascension hospital emergency room doctor in Detroit said a man on the city’s east side was given a dangerous narcotic intended for another patient because of a paperwork mix-up. As a result, the patient’s breathing slowed to the point that he had to be put on a ventilator. “We intubated him and we sent him to the ICU because he got the wrong medication.”
A nurse in a Michigan Ascension hospital ER said a woman with low blood sugar and “altered mental status” went into cardiac arrest and died after staff said they waited four hours for lab results they needed to determine how to treat her, but never received. “If I started having crushing chest pain in the middle of work and thought I was having a big one, I would grab someone to drive me down the street to another hospital,” the same ER nurse said.
Similar concerns reportedly led a travel nurse at an Ascension hospital in Indiana to quit. “I just want to warn those patients that are coming to any of the Ascension facilities that there will be delays in care. There is potential for error and for harm,” Justin Neisser told CBS4 in Indianapolis in May.
Several nurses and doctors at Ascension hospitals said they feared the errors they’ve witnessed since the cyberattack began could threaten their professional licenses. “This is how a RaDonda Vaught happens,” one nurse said, referring to the Tennessee nurse who was convicted of criminally negligent homicide in 2022 for a fatal drug error.
Reporters were not able to review records to verify clinicians’ claims because of privacy laws surrounding patients’ medical information that apply to health care professionals.
Ascension declined to answer questions about claims that care has been affected by the ransomware attack. “As we have made clear throughout this cyber attack which has impacted our system and our dedicated clinical providers, caring for our patients is our highest priority,” Sean Fitzpatrick, Ascension’s vice president of external communications, said via email on June 3. “We are confident that our care providers in our hospitals and facilities continue to provide quality medical care.”
The federal government requires hospitals to protect patients’ sensitive health data, according to cybersecurity experts. However, there are no federal requirements for hospitals to prevent or prepare for cyberattacks that could compromise their electronic systems.
Hospitals: ‘The No.1 Target of Ransomware’
“We’ve started to think about these as public health issues and disasters on the scale of earthquakes or hurricanes,” said Jeff Tully, a co-director of the Center for Healthcare Cybersecurity at the University of California-San Diego. “These types of cybersecurity incidents should be thought of as a matter of when, and not if.”
Josh Corman, a cybersecurity expert and advocate, said ransom crews regard hospitals as the perfect prey: “They have terrible security and they’ll pay. So almost immediately, hospitals went to the No. 1 target of ransomware.”
In 2023, the health sector experienced the largest share of ransomware attacks of 16 infrastructure sectors considered vital to national security or safety, according to an FBI report on internet crimes. In March, the federal Department of Health and Human Services said reported large breaches involving ransomware had jumped by 264% over the past five years.
A cyberattack this year on Change Healthcare, a unit of UnitedHealth Group’s Optum division that processes billions of health care transactions every year, crippled the business of providers, pharmacies, and hospitals.
In May, UnitedHealth Group CEO Andrew Witty told lawmakers the company paid a $22 million ransom as a result of the Change Healthcare attack — which occurred after hackers accessed a company portal that didn’t have multifactor authentication, a basic cybersecurity tool.
The Biden administration in recent months has pushed to bolster health care cybersecurity standards, but it’s not clear which new measures will be required.
In January, HHS nudged companies to improve email security, add multifactor authentication, and institute cybersecurity training and testing, among other voluntary measures. The Centers for Medicare & Medicaid Services is expected to release new requirements for hospitals, but the scope and timing are unclear. The same is true of an update HHS is expected to make to patient privacy regulations.
HHS said the voluntary measures “will inform the creation of new enforceable cybersecurity standards,” department spokesperson Jeff Nesbit said in a statement.
“The recent cyberattack at Ascension only underscores the need for everyone in the health care ecosystem to do their part to secure their systems and protect patients,” Nesbit said.
Meanwhile, lobbyists for the hospital industry contend cybersecurity mandates or penalties are misplaced and would curtail hospitals’ resources to fend off attacks.
“Hospitals and health systems are not the primary source of cyber risk exposure facing the health care sector,” the American Hospital Association, the largest lobbying group for U.S. hospitals, said in an April statement prepared for U.S. House lawmakers. Most large data breaches that hit hospitals in 2023 originated with third-party “business associates” or other health entities, including CMS itself, the AHA statement said.
Hospitals consolidating into large multistate health systems face increased risk of data breaches and ransomware attacks, according to one study. Ascension in 2022 was the third-largest hospital chain in the U.S. by number of beds, according to the most recent data from the federal Agency for Healthcare Research and Quality.
And while cybersecurity regulations can quickly become outdated, they can at least make it clear that if health systems fail to implement basic protections there “should be consequences for that,” Jim Bagian, a former director of the National Center for Patient Safety at the Veterans Health Administration, told Michigan Public’s Stateside.
Patients can pay the price when lapses occur. Those in hospital care face a greater likelihood of death during a cyberattack, according to researchers at the University of Minnesota School of Public Health.
Workers concerned about patient safety at Ascension hospitals in Michigan have called for the company to make changes.
“We implore Ascension to recognize the internal problems that continue to plague its hospitals, both publicly and transparently,” said Dina Carlisle, a nurse and the president of the OPEIU Local 40 union, which represents nurses at Ascension Providence Rochester. At least 125 staff members at that Ascension hospital have signed a petition asking administrators to temporarily reduce elective surgeries and nonemergency patient admissions, like under the protocols many hospitals adopted early in the covid-19 pandemic.
Watson, the Kansas ICU nurse, said in late May that nurses had urged management to bring in more nurses to help manage the workflow. “Everything that we say has fallen on deaf ears,” she said.
“It is very hard to be a nurse at Ascension right now,” Watson said in late May. “It is very hard to be a patient at Ascension right now.”
If you’re a patient or worker at an Ascension hospital and would like to tell KFF Health News about your experiences, click here to share your story with us.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
Experts Expect New Human Cases of Avian Flu
With avian influenza spreading quickly around the globe, the virus has more opportunities to mutate and cause problems for people. By some calculations, H5N1 bird flu is still at least two mutations away from widespread human infections, but experts warn that new flu symptoms in individuals at high risk are likely to start turning up in health systems this summer.
“Some of this will not be obvious or at the forefront of our minds.”
Dr. Dugan is leading the team of CDC scientists that is working with partners from the US Department of Agriculture, the US Food and Drug Administration (FDA), and state and local health departments to track and respond to the H5N1 bird flu outbreak currently sweeping through the United States.
Since 2022, avian influenza A viruses have been detected in more than 9300 wild birds in 50 states and territories and in commercial and backyard flocks.
“It’s a bad situation,” said Florian Krammer, PhD, professor of vaccinology at the Icahn School of Medicine at Mount Sinai in New York. “Globally, we’ve seen tons of exposure in cities around the world and even in the birds here in New York City where I am.”
Birds shed the virus in their saliva, mucous, and feces, so people or other animals with close, unprotected contact with infected birds or their contaminated environments can be infected.
And for the first time in March 2024, H5N1 bird flu was reported in dairy cows. The US Department of Agriculture said that at last count, 101 dairy herds in 12 states had been infected, with several cases also found in dairy workers.
From Birds to Cattle and Farm Workers
The National Veterinary Services Laboratories confirmed the infections were highly pathogenic avian influenza H5N1 clade 2.3.4.4b of Eurasian lineage. Also known as the goose, Guangdong clade from China, phylogenetic analysis and epidemiology suggests a single introduction into cows followed by onward transmission.
“I was surprised when H5 was introduced to dairy cattle in this way,” Dr. Dugan said. “Influenza viruses are always surprising us and it reminds me to stay humble and keep an open mind when dealing with them.”
People rarely inhale or get sufficient virus in their eyes or mouth to get sick, Dr. Dugan said, but those in close contact with animals are still at risk for infection, which could lead to upper respiratory tract symptoms such as shortness of breath, cough, sore throat, or runny or stuffy nose.
Like with other viruses, people can also experience muscle or body aches, headache, fatigue, fever or, as was seen in farm workers, conjunctivitis.
But there are less-common symptoms too like diarrhea, nausea, and vomiting — and sometimes, even seizures.
The risk to the general public is still low, Dr. Dugan said, but authorities recommend that people working with animals wash their hands with soap and water and wear personal protective equipment that includes fluid-resistant coveralls, a waterproof apron, a safety-approved respirator, properly fitted goggles or face shield, a head or hair cover, gloves, and boots.
Dr. Dugan said that health care providers often don’t take a history of occupational exposures when a patient presents with flu. But with rising rates of bird flu in new animal hosts, “this will be an important next step.”
Asking Unusual Questions
This approach is not standardized on most electronic health records, so these are questions that clinicians will need to initiate themselves.
“Physicians should ask about work,” said Meghan Davis, PhD, associate professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore. “If it’s not already on the radar, asking about any direct contact with dairy cows, poultry, pigs, wild birds, or wild mammals is important.”
Dr. Davis says she’s worried about a new study tracking risk factors for farm-to-farm transmission because it shows that farms testing positive for avian influenza often have workers with a family member also employed on another farm. “This suggests that we might need to be on the lookout for possible transmission within families,” she said. Now, we have to ask “not just if the person with symptoms has contact with or works on a dairy farm, milk processing plant, or slaughterhouse, but also if any family member does.”
Dr. Davis said that it’s important to bear in mind when taking these histories that there may be younger workers on farms and in slaughter and processing facilities due to exemptions or illegal work.
What is important now is to get the situation under control this season in dairy cattle, Dr. Krammer said. “This will be easier to stop in cows than humans, so this is the time to stop moving dairy cattle and start vaccinating them.”
Spotting New Cases
Since April 2024, there have been three human cases of avian influenza after exposure to dairy cows reported. “And what we don’t want to see this summer is an unusual human cluster of influenza. It’s important we keep a close, watchful eye for this,” Dr. Krammer said.
“Influenza viruses do very interesting things and as we head into fall and winter flu season, we don’t want new human co-infections that could cause major problems for us,” he said.
If people become mixing vessels of a seasonal cocktail of multiple viruses, that could empower H5N1 to mutate again into something more dangerous, sparking a new pandemic.
“It wasn’t all that long ago that we were asking China difficult questions about the steps Chinese authorities took to protect human lives from SARS-CoV-2 in the COVID pandemic. Now, we must ask ourselves many of these questions,” Dr. Krammer said. “We are at a crucial crossroad where we will either elude a new pandemic or see one take off, risking 10 to 20 million lives.”
There is a precedent for safely evading more trouble, Dr. Krammer pointed out. Government agencies have already been working with the poultry industry for a couple of years now. “And here, we have successfully stopped H5N1 with new regulations and policies.”
But moving from poultry farms to cattle has not been an easy transition, Dr. Dugan said. Cattle farms have no experience with bird flu or tactics to contain it with regulations, and officials too are working in new, unfamiliar terrain.
“What we have now isn’t a science problem, it’s a policy issue, and it hasn’t always been clear who is in charge,” Dr. Krammer said.
“Agencies are working together at the state, federal, and global level,” said Dr. Dugan. “We are increasing our transparency and are working to share what we know, when we know it.”
The infrastructure built during the COVID pandemic has helped teams prepare for this new crisis, Dr. Dugan said. Year-round, layered monitoring has clinical labs reporting seasonal influenza and novel cases.
“Laboratories are ready to help with testing,” Dr. Dugan said.
Specimens should be collected as soon as possible from patients with flu symptoms. A nasopharyngeal swab is recommended with a nasal swab combined with an oropharyngeal swab. If a patient has conjunctivitis with or without respiratory symptoms, both a conjunctival swab and a nasopharyngeal swab should be collected.
People with severe respiratory disease should also have lower respiratory tract specimens collected.
Standard, contact, and airborne precautions are recommended for patients presenting for medical care who have illness consistent with influenza and recent exposure to birds or other animals.
Antiviral Drugs
There are four FDA-approved antivirals for influenza: Oseltamivir phosphate (available as a generic drug or by the trade name Tamiflu), zanamivir (Relenza), peramivir (Rapivab) , and baloxavir (Xofluza).
For people with suspected or confirmed avian influenza, treatment is recommended as soon as possible.
There are no clinical trials measuring the outcome of antivirals in people infected with avian influenza. However, data from animal models and human observational studies suggest a benefit.
“We can’t afford to wait this summer,” Dr. Krammer said. “We have an opportunity right now to stop this in cows before we risk infecting more people. I hope we do.”
A version of this article first appeared on Medscape.com.
With avian influenza spreading quickly around the globe, the virus has more opportunities to mutate and cause problems for people. By some calculations, H5N1 bird flu is still at least two mutations away from widespread human infections, but experts warn that new flu symptoms in individuals at high risk are likely to start turning up in health systems this summer.
“Some of this will not be obvious or at the forefront of our minds.”
Dr. Dugan is leading the team of CDC scientists that is working with partners from the US Department of Agriculture, the US Food and Drug Administration (FDA), and state and local health departments to track and respond to the H5N1 bird flu outbreak currently sweeping through the United States.
Since 2022, avian influenza A viruses have been detected in more than 9300 wild birds in 50 states and territories and in commercial and backyard flocks.
“It’s a bad situation,” said Florian Krammer, PhD, professor of vaccinology at the Icahn School of Medicine at Mount Sinai in New York. “Globally, we’ve seen tons of exposure in cities around the world and even in the birds here in New York City where I am.”
Birds shed the virus in their saliva, mucous, and feces, so people or other animals with close, unprotected contact with infected birds or their contaminated environments can be infected.
And for the first time in March 2024, H5N1 bird flu was reported in dairy cows. The US Department of Agriculture said that at last count, 101 dairy herds in 12 states had been infected, with several cases also found in dairy workers.
From Birds to Cattle and Farm Workers
The National Veterinary Services Laboratories confirmed the infections were highly pathogenic avian influenza H5N1 clade 2.3.4.4b of Eurasian lineage. Also known as the goose, Guangdong clade from China, phylogenetic analysis and epidemiology suggests a single introduction into cows followed by onward transmission.
“I was surprised when H5 was introduced to dairy cattle in this way,” Dr. Dugan said. “Influenza viruses are always surprising us and it reminds me to stay humble and keep an open mind when dealing with them.”
People rarely inhale or get sufficient virus in their eyes or mouth to get sick, Dr. Dugan said, but those in close contact with animals are still at risk for infection, which could lead to upper respiratory tract symptoms such as shortness of breath, cough, sore throat, or runny or stuffy nose.
Like with other viruses, people can also experience muscle or body aches, headache, fatigue, fever or, as was seen in farm workers, conjunctivitis.
But there are less-common symptoms too like diarrhea, nausea, and vomiting — and sometimes, even seizures.
The risk to the general public is still low, Dr. Dugan said, but authorities recommend that people working with animals wash their hands with soap and water and wear personal protective equipment that includes fluid-resistant coveralls, a waterproof apron, a safety-approved respirator, properly fitted goggles or face shield, a head or hair cover, gloves, and boots.
Dr. Dugan said that health care providers often don’t take a history of occupational exposures when a patient presents with flu. But with rising rates of bird flu in new animal hosts, “this will be an important next step.”
Asking Unusual Questions
This approach is not standardized on most electronic health records, so these are questions that clinicians will need to initiate themselves.
“Physicians should ask about work,” said Meghan Davis, PhD, associate professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore. “If it’s not already on the radar, asking about any direct contact with dairy cows, poultry, pigs, wild birds, or wild mammals is important.”
Dr. Davis says she’s worried about a new study tracking risk factors for farm-to-farm transmission because it shows that farms testing positive for avian influenza often have workers with a family member also employed on another farm. “This suggests that we might need to be on the lookout for possible transmission within families,” she said. Now, we have to ask “not just if the person with symptoms has contact with or works on a dairy farm, milk processing plant, or slaughterhouse, but also if any family member does.”
Dr. Davis said that it’s important to bear in mind when taking these histories that there may be younger workers on farms and in slaughter and processing facilities due to exemptions or illegal work.
What is important now is to get the situation under control this season in dairy cattle, Dr. Krammer said. “This will be easier to stop in cows than humans, so this is the time to stop moving dairy cattle and start vaccinating them.”
Spotting New Cases
Since April 2024, there have been three human cases of avian influenza after exposure to dairy cows reported. “And what we don’t want to see this summer is an unusual human cluster of influenza. It’s important we keep a close, watchful eye for this,” Dr. Krammer said.
“Influenza viruses do very interesting things and as we head into fall and winter flu season, we don’t want new human co-infections that could cause major problems for us,” he said.
If people become mixing vessels of a seasonal cocktail of multiple viruses, that could empower H5N1 to mutate again into something more dangerous, sparking a new pandemic.
“It wasn’t all that long ago that we were asking China difficult questions about the steps Chinese authorities took to protect human lives from SARS-CoV-2 in the COVID pandemic. Now, we must ask ourselves many of these questions,” Dr. Krammer said. “We are at a crucial crossroad where we will either elude a new pandemic or see one take off, risking 10 to 20 million lives.”
There is a precedent for safely evading more trouble, Dr. Krammer pointed out. Government agencies have already been working with the poultry industry for a couple of years now. “And here, we have successfully stopped H5N1 with new regulations and policies.”
But moving from poultry farms to cattle has not been an easy transition, Dr. Dugan said. Cattle farms have no experience with bird flu or tactics to contain it with regulations, and officials too are working in new, unfamiliar terrain.
“What we have now isn’t a science problem, it’s a policy issue, and it hasn’t always been clear who is in charge,” Dr. Krammer said.
“Agencies are working together at the state, federal, and global level,” said Dr. Dugan. “We are increasing our transparency and are working to share what we know, when we know it.”
The infrastructure built during the COVID pandemic has helped teams prepare for this new crisis, Dr. Dugan said. Year-round, layered monitoring has clinical labs reporting seasonal influenza and novel cases.
“Laboratories are ready to help with testing,” Dr. Dugan said.
Specimens should be collected as soon as possible from patients with flu symptoms. A nasopharyngeal swab is recommended with a nasal swab combined with an oropharyngeal swab. If a patient has conjunctivitis with or without respiratory symptoms, both a conjunctival swab and a nasopharyngeal swab should be collected.
People with severe respiratory disease should also have lower respiratory tract specimens collected.
Standard, contact, and airborne precautions are recommended for patients presenting for medical care who have illness consistent with influenza and recent exposure to birds or other animals.
Antiviral Drugs
There are four FDA-approved antivirals for influenza: Oseltamivir phosphate (available as a generic drug or by the trade name Tamiflu), zanamivir (Relenza), peramivir (Rapivab) , and baloxavir (Xofluza).
For people with suspected or confirmed avian influenza, treatment is recommended as soon as possible.
There are no clinical trials measuring the outcome of antivirals in people infected with avian influenza. However, data from animal models and human observational studies suggest a benefit.
“We can’t afford to wait this summer,” Dr. Krammer said. “We have an opportunity right now to stop this in cows before we risk infecting more people. I hope we do.”
A version of this article first appeared on Medscape.com.
With avian influenza spreading quickly around the globe, the virus has more opportunities to mutate and cause problems for people. By some calculations, H5N1 bird flu is still at least two mutations away from widespread human infections, but experts warn that new flu symptoms in individuals at high risk are likely to start turning up in health systems this summer.
“Some of this will not be obvious or at the forefront of our minds.”
Dr. Dugan is leading the team of CDC scientists that is working with partners from the US Department of Agriculture, the US Food and Drug Administration (FDA), and state and local health departments to track and respond to the H5N1 bird flu outbreak currently sweeping through the United States.
Since 2022, avian influenza A viruses have been detected in more than 9300 wild birds in 50 states and territories and in commercial and backyard flocks.
“It’s a bad situation,” said Florian Krammer, PhD, professor of vaccinology at the Icahn School of Medicine at Mount Sinai in New York. “Globally, we’ve seen tons of exposure in cities around the world and even in the birds here in New York City where I am.”
Birds shed the virus in their saliva, mucous, and feces, so people or other animals with close, unprotected contact with infected birds or their contaminated environments can be infected.
And for the first time in March 2024, H5N1 bird flu was reported in dairy cows. The US Department of Agriculture said that at last count, 101 dairy herds in 12 states had been infected, with several cases also found in dairy workers.
From Birds to Cattle and Farm Workers
The National Veterinary Services Laboratories confirmed the infections were highly pathogenic avian influenza H5N1 clade 2.3.4.4b of Eurasian lineage. Also known as the goose, Guangdong clade from China, phylogenetic analysis and epidemiology suggests a single introduction into cows followed by onward transmission.
“I was surprised when H5 was introduced to dairy cattle in this way,” Dr. Dugan said. “Influenza viruses are always surprising us and it reminds me to stay humble and keep an open mind when dealing with them.”
People rarely inhale or get sufficient virus in their eyes or mouth to get sick, Dr. Dugan said, but those in close contact with animals are still at risk for infection, which could lead to upper respiratory tract symptoms such as shortness of breath, cough, sore throat, or runny or stuffy nose.
Like with other viruses, people can also experience muscle or body aches, headache, fatigue, fever or, as was seen in farm workers, conjunctivitis.
But there are less-common symptoms too like diarrhea, nausea, and vomiting — and sometimes, even seizures.
The risk to the general public is still low, Dr. Dugan said, but authorities recommend that people working with animals wash their hands with soap and water and wear personal protective equipment that includes fluid-resistant coveralls, a waterproof apron, a safety-approved respirator, properly fitted goggles or face shield, a head or hair cover, gloves, and boots.
Dr. Dugan said that health care providers often don’t take a history of occupational exposures when a patient presents with flu. But with rising rates of bird flu in new animal hosts, “this will be an important next step.”
Asking Unusual Questions
This approach is not standardized on most electronic health records, so these are questions that clinicians will need to initiate themselves.
“Physicians should ask about work,” said Meghan Davis, PhD, associate professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore. “If it’s not already on the radar, asking about any direct contact with dairy cows, poultry, pigs, wild birds, or wild mammals is important.”
Dr. Davis says she’s worried about a new study tracking risk factors for farm-to-farm transmission because it shows that farms testing positive for avian influenza often have workers with a family member also employed on another farm. “This suggests that we might need to be on the lookout for possible transmission within families,” she said. Now, we have to ask “not just if the person with symptoms has contact with or works on a dairy farm, milk processing plant, or slaughterhouse, but also if any family member does.”
Dr. Davis said that it’s important to bear in mind when taking these histories that there may be younger workers on farms and in slaughter and processing facilities due to exemptions or illegal work.
What is important now is to get the situation under control this season in dairy cattle, Dr. Krammer said. “This will be easier to stop in cows than humans, so this is the time to stop moving dairy cattle and start vaccinating them.”
Spotting New Cases
Since April 2024, there have been three human cases of avian influenza after exposure to dairy cows reported. “And what we don’t want to see this summer is an unusual human cluster of influenza. It’s important we keep a close, watchful eye for this,” Dr. Krammer said.
“Influenza viruses do very interesting things and as we head into fall and winter flu season, we don’t want new human co-infections that could cause major problems for us,” he said.
If people become mixing vessels of a seasonal cocktail of multiple viruses, that could empower H5N1 to mutate again into something more dangerous, sparking a new pandemic.
“It wasn’t all that long ago that we were asking China difficult questions about the steps Chinese authorities took to protect human lives from SARS-CoV-2 in the COVID pandemic. Now, we must ask ourselves many of these questions,” Dr. Krammer said. “We are at a crucial crossroad where we will either elude a new pandemic or see one take off, risking 10 to 20 million lives.”
There is a precedent for safely evading more trouble, Dr. Krammer pointed out. Government agencies have already been working with the poultry industry for a couple of years now. “And here, we have successfully stopped H5N1 with new regulations and policies.”
But moving from poultry farms to cattle has not been an easy transition, Dr. Dugan said. Cattle farms have no experience with bird flu or tactics to contain it with regulations, and officials too are working in new, unfamiliar terrain.
“What we have now isn’t a science problem, it’s a policy issue, and it hasn’t always been clear who is in charge,” Dr. Krammer said.
“Agencies are working together at the state, federal, and global level,” said Dr. Dugan. “We are increasing our transparency and are working to share what we know, when we know it.”
The infrastructure built during the COVID pandemic has helped teams prepare for this new crisis, Dr. Dugan said. Year-round, layered monitoring has clinical labs reporting seasonal influenza and novel cases.
“Laboratories are ready to help with testing,” Dr. Dugan said.
Specimens should be collected as soon as possible from patients with flu symptoms. A nasopharyngeal swab is recommended with a nasal swab combined with an oropharyngeal swab. If a patient has conjunctivitis with or without respiratory symptoms, both a conjunctival swab and a nasopharyngeal swab should be collected.
People with severe respiratory disease should also have lower respiratory tract specimens collected.
Standard, contact, and airborne precautions are recommended for patients presenting for medical care who have illness consistent with influenza and recent exposure to birds or other animals.
Antiviral Drugs
There are four FDA-approved antivirals for influenza: Oseltamivir phosphate (available as a generic drug or by the trade name Tamiflu), zanamivir (Relenza), peramivir (Rapivab) , and baloxavir (Xofluza).
For people with suspected or confirmed avian influenza, treatment is recommended as soon as possible.
There are no clinical trials measuring the outcome of antivirals in people infected with avian influenza. However, data from animal models and human observational studies suggest a benefit.
“We can’t afford to wait this summer,” Dr. Krammer said. “We have an opportunity right now to stop this in cows before we risk infecting more people. I hope we do.”
A version of this article first appeared on Medscape.com.
Long COVID Can’t Be Solved Until We Decide What It Is
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.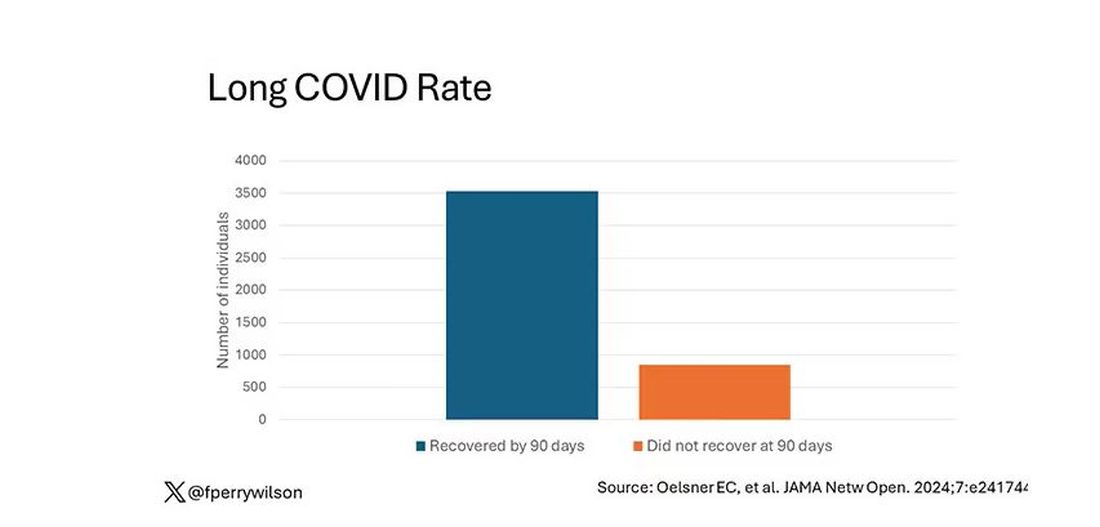
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.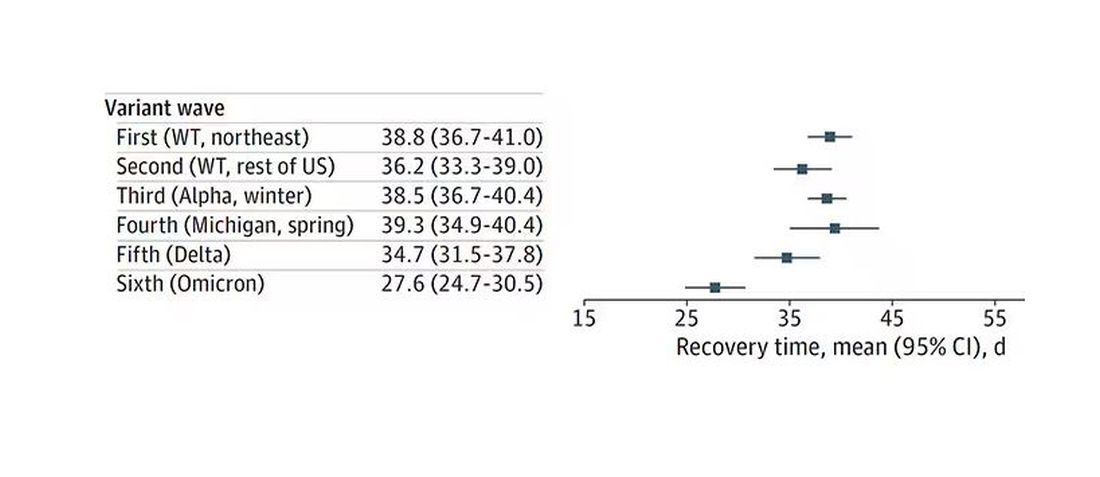
Recovery times were longer for smokers, those with diabetes, and those who were obese.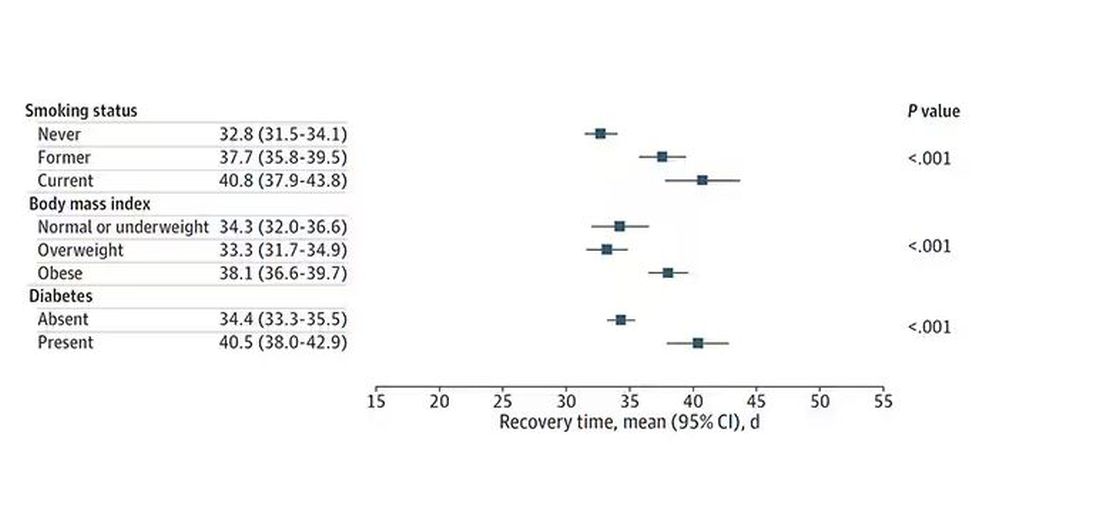
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.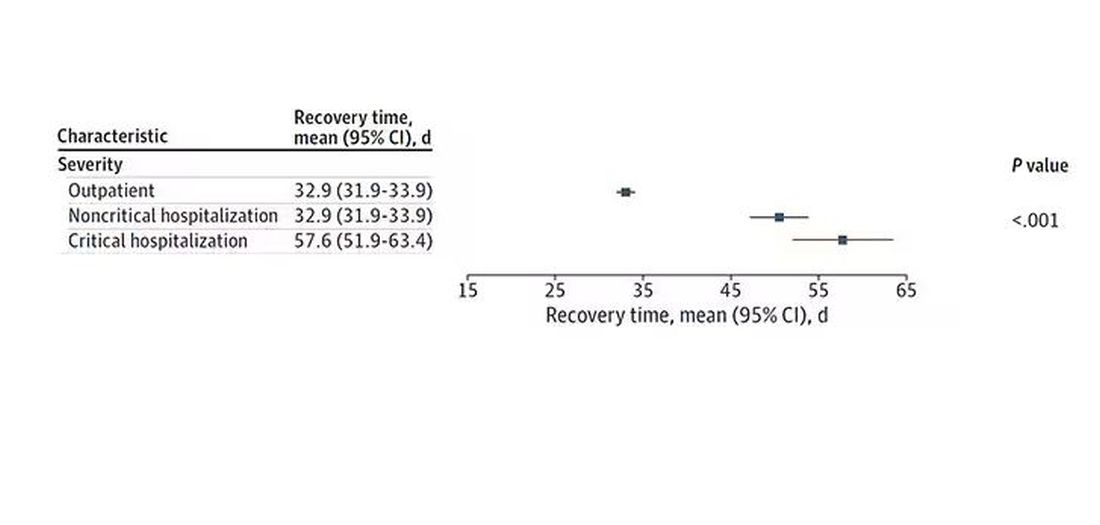
Vaccination was associated with shorter recovery times, as you can see here. 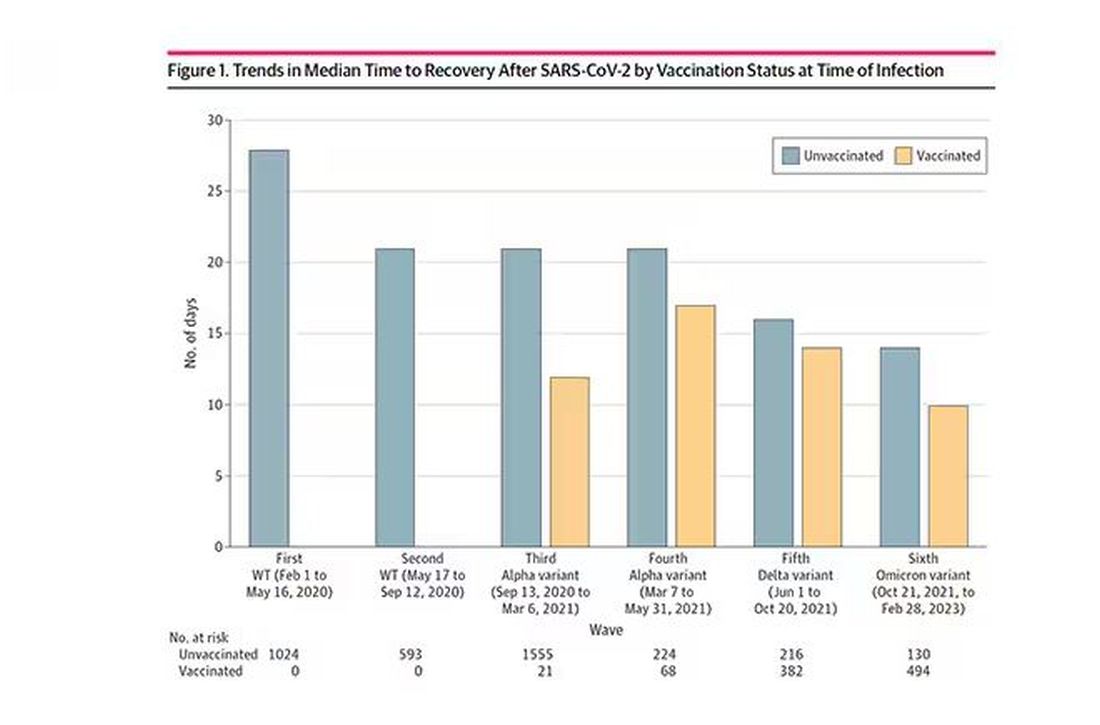
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.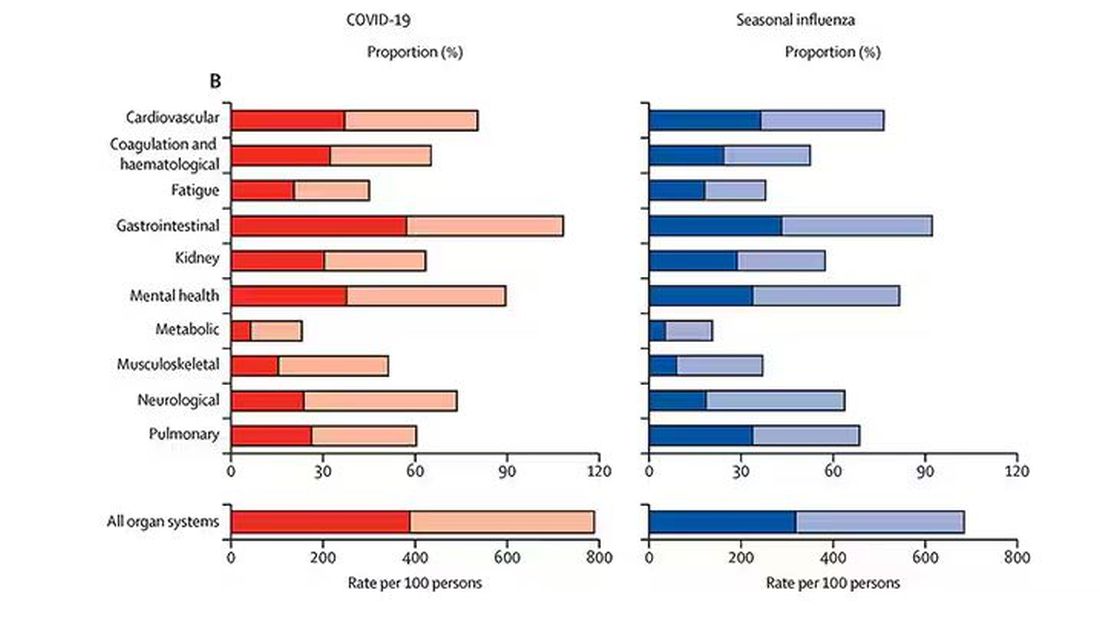
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.
Recovery times were longer for smokers, those with diabetes, and those who were obese.
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.
Vaccination was associated with shorter recovery times, as you can see here. 
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.
Recovery times were longer for smokers, those with diabetes, and those who were obese.
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.
Vaccination was associated with shorter recovery times, as you can see here. 
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FDA Approves New Pneumococcal Vaccine
A new vaccine to prevent invasive pneumococcal disease and pneumococcal pneumonia in adults has been approved by the Food and Drug Administration.
The injectable drug, Capvaxive (Pneumococcal 21-valent Conjugate Vaccine), protects against 22 serotypes that cause invasive pneumococcal disease in adults, the company said in a news release. These strains account for about 84% of invasive pneumococcal disease cases among adults aged 50 years or older and about 85% of these cases in adults aged 65 years or older.
The drug company said about 150,000 adults in the United States are hospitalized annually because of pneumococcal pneumonia.
“Many cases of adult disease are caused by serotypes not included in other approved pneumococcal conjugate vaccines,” Walter Orenstein, MD, a professor emeritus of medicine, epidemiology, global health, and pediatrics at Emory University, Atlanta, Georgia, and a member of Merck’s Scientific Advisory Committee, said in the release.
A draft agenda shows a Centers for Disease Control and Prevention (CDC) advisory panel will meet on June 27 to discuss the vaccine. If the committee votes to approve Capvaxive, the CDC director will decide whether to make it available across the country.
Testing showed that Capvaxive was well tolerated by people it was tested on, with the main reports being pain where they got the shot, fatigue, headaches, and muscle aches, Merck said.
The eight unique serotypes included in CAPVAXIVE will protect against invasive pneumococcal disease and pneumococcal pneumonia, not just pneumonia.
According to Reuters, Merck said Capvaxive has a wholesale acquisition price of $287 per dose, but most people will probably have access to it at no cost if the drug receives a routine CDC recommendation. Capvaxive’s main competition is expected to be Pfizer’s shot, Prevnar 20, which was approved in 2021 for use in adults aged 18 years or older, Reuters reported.
A version of this article appeared on Medscape.com.
A new vaccine to prevent invasive pneumococcal disease and pneumococcal pneumonia in adults has been approved by the Food and Drug Administration.
The injectable drug, Capvaxive (Pneumococcal 21-valent Conjugate Vaccine), protects against 22 serotypes that cause invasive pneumococcal disease in adults, the company said in a news release. These strains account for about 84% of invasive pneumococcal disease cases among adults aged 50 years or older and about 85% of these cases in adults aged 65 years or older.
The drug company said about 150,000 adults in the United States are hospitalized annually because of pneumococcal pneumonia.
“Many cases of adult disease are caused by serotypes not included in other approved pneumococcal conjugate vaccines,” Walter Orenstein, MD, a professor emeritus of medicine, epidemiology, global health, and pediatrics at Emory University, Atlanta, Georgia, and a member of Merck’s Scientific Advisory Committee, said in the release.
A draft agenda shows a Centers for Disease Control and Prevention (CDC) advisory panel will meet on June 27 to discuss the vaccine. If the committee votes to approve Capvaxive, the CDC director will decide whether to make it available across the country.
Testing showed that Capvaxive was well tolerated by people it was tested on, with the main reports being pain where they got the shot, fatigue, headaches, and muscle aches, Merck said.
The eight unique serotypes included in CAPVAXIVE will protect against invasive pneumococcal disease and pneumococcal pneumonia, not just pneumonia.
According to Reuters, Merck said Capvaxive has a wholesale acquisition price of $287 per dose, but most people will probably have access to it at no cost if the drug receives a routine CDC recommendation. Capvaxive’s main competition is expected to be Pfizer’s shot, Prevnar 20, which was approved in 2021 for use in adults aged 18 years or older, Reuters reported.
A version of this article appeared on Medscape.com.
A new vaccine to prevent invasive pneumococcal disease and pneumococcal pneumonia in adults has been approved by the Food and Drug Administration.
The injectable drug, Capvaxive (Pneumococcal 21-valent Conjugate Vaccine), protects against 22 serotypes that cause invasive pneumococcal disease in adults, the company said in a news release. These strains account for about 84% of invasive pneumococcal disease cases among adults aged 50 years or older and about 85% of these cases in adults aged 65 years or older.
The drug company said about 150,000 adults in the United States are hospitalized annually because of pneumococcal pneumonia.
“Many cases of adult disease are caused by serotypes not included in other approved pneumococcal conjugate vaccines,” Walter Orenstein, MD, a professor emeritus of medicine, epidemiology, global health, and pediatrics at Emory University, Atlanta, Georgia, and a member of Merck’s Scientific Advisory Committee, said in the release.
A draft agenda shows a Centers for Disease Control and Prevention (CDC) advisory panel will meet on June 27 to discuss the vaccine. If the committee votes to approve Capvaxive, the CDC director will decide whether to make it available across the country.
Testing showed that Capvaxive was well tolerated by people it was tested on, with the main reports being pain where they got the shot, fatigue, headaches, and muscle aches, Merck said.
The eight unique serotypes included in CAPVAXIVE will protect against invasive pneumococcal disease and pneumococcal pneumonia, not just pneumonia.
According to Reuters, Merck said Capvaxive has a wholesale acquisition price of $287 per dose, but most people will probably have access to it at no cost if the drug receives a routine CDC recommendation. Capvaxive’s main competition is expected to be Pfizer’s shot, Prevnar 20, which was approved in 2021 for use in adults aged 18 years or older, Reuters reported.
A version of this article appeared on Medscape.com.
Acute Sore Throat in Primary Care: When to Reach for the Antibiotics
This transcript has been edited for clarity.
There is a helpful consensus from experts on the best management of patients with acute sore throat. This is a common problem in primary care, and one for which there is a lot of evidence, opinion, and ultimately overprescribing of antibiotics. This consensus presents a pragmatic clinical approach aimed at decreasing overprescribing, yet detecting which patients are likely to benefit from treatment with antibiotics.
Let’s first go over the evidence that forms the basis for the recommendations, then the recommended approach. First, a sore throat can be caused by many different viruses, as well as group A streptococcus (GAS), the group C streptococcus S dysgalactiae, and fusobacterium. We sometimes think of throat cultures as telling us the definitive etiology of a sore throat. In fact, children commonly are colonized with GAS even when not infected — 35% of the time, when GAS is detected on throat swab in a child, GAS is not the cause of the sore throat. Very few adults are colonized with GAS.
Sore throats are usually self-limited, whether they are treated with antibiotics or not, but occasionally complications can occur. Suppurative complications include peritonsillar abscess, sinusitis and sepsis. Nonsuppurative complications are primarily glomerulonephritis and rheumatic fever, which can lead to rheumatic heart disease.
Antibiotics. Antibiotics have three potential benefits in acute sore throat: to reduce the risk of developing rheumatic heart disease, reduce the duration and severity of symptoms, and treat suppurative complications. The risk for rheumatic heart disease has almost vanished in high-income countries, but not in low-income countries. Thus, antibiotic treatment of acute sore throat due to GAS may benefit those in living in, and those who recently emigrated from, low-income countries.
Patients with suppurative complications should be identified because antibiotics are important for this group. Although antibiotics are prescribed primarily to prevent rheumatic fever in this population, they may be mildly helpful in reducing a patient’s symptoms.
Testing. The sensitivity and specificity of high-quality point-of-care tests (POCTs) are on par with those of cultures, with the advantage that the results are available within minutes. Negative tests reduce unneeded antibiotic prescriptions.
Given this evidence, the authors recommend an approach that puts a lot of emphasis on two major things: the risk for rheumatic fever, and clinical assessment. On the basis of these factors, a decision is made about the utility of POCTs and treatment with antibiotics for GAS. The risk for rheumatic fever is based on epidemiology: If the patient is in a low-income country or has recently immigrated from one, then the risk is high, and if not, the risk is low.
Complicated vs uncomplicated? This is determined by clinical assessment of the severity of the patient’s illness, including general appearance. Uncomplicated sore throat means that the patient:
- Is not getting worse after 3 days of illness
- Has a duration of illness ≤ 5 days or is getting better after day 5
- Has mild to moderate symptom severity (bilateral throat pain, the ability to open the mouth fully, and absence of a sandpaper or scarlatiniform rash or strawberry tongue)
For patients with uncomplicated sore throat and low risk for rheumatic fever, the main goals are to reduce antibiotic use and provide symptomatic relief. For these patients, an assessment such as the Centor score can be done. Those with a low Centor score (0-2) can be treated with analgesics and there is no need for a POCT.
In patients with a higher Centor score, the consensus gives two choices: They can either be tested (and treated if the testing is positive), or it is reasonable to forgo testing and use a wait-and-see strategy, with reevaluation if they are getting worse after day 3 or not improving after day 5 days of their illness. Illnesses that last longer than 5 days with sore throat and fatigue should prompt consideration of alternative diagnoses, such as infectious mononucleosis.
For patients with potentially complicated sore throat — including indicators such as worsening symptoms after 3 days or worsening after initiation of antibiotics, inability to open the mouth fully, unilateral neck pain or swelling, or rigors — should undergo a careful evaluation. The need for further testing in these patients, including labs and imaging, should be decided on a case-by-case basis. If the patient appears seriously ill, don’t rely solely on POCT for GAS, but think about other diagnoses.
Rheumatic fever. The approach is very different in patients at high risk for rheumatic fever. POCT for GAS is recommended irrespective of their clinical score, and antibiotics should be prescribed if it’s positive for GAS. If a POCT is unavailable, then the consensus recommends prescribing antibiotics for all high-risk patients who have acute sore throat.
This approach is sensible and puts a lot of emphasis on clinical evaluation, though it should be noted that this approach is considerably different from that in the 2012 Infectious Diseases Society of America guidelines.
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly and Company, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
There is a helpful consensus from experts on the best management of patients with acute sore throat. This is a common problem in primary care, and one for which there is a lot of evidence, opinion, and ultimately overprescribing of antibiotics. This consensus presents a pragmatic clinical approach aimed at decreasing overprescribing, yet detecting which patients are likely to benefit from treatment with antibiotics.
Let’s first go over the evidence that forms the basis for the recommendations, then the recommended approach. First, a sore throat can be caused by many different viruses, as well as group A streptococcus (GAS), the group C streptococcus S dysgalactiae, and fusobacterium. We sometimes think of throat cultures as telling us the definitive etiology of a sore throat. In fact, children commonly are colonized with GAS even when not infected — 35% of the time, when GAS is detected on throat swab in a child, GAS is not the cause of the sore throat. Very few adults are colonized with GAS.
Sore throats are usually self-limited, whether they are treated with antibiotics or not, but occasionally complications can occur. Suppurative complications include peritonsillar abscess, sinusitis and sepsis. Nonsuppurative complications are primarily glomerulonephritis and rheumatic fever, which can lead to rheumatic heart disease.
Antibiotics. Antibiotics have three potential benefits in acute sore throat: to reduce the risk of developing rheumatic heart disease, reduce the duration and severity of symptoms, and treat suppurative complications. The risk for rheumatic heart disease has almost vanished in high-income countries, but not in low-income countries. Thus, antibiotic treatment of acute sore throat due to GAS may benefit those in living in, and those who recently emigrated from, low-income countries.
Patients with suppurative complications should be identified because antibiotics are important for this group. Although antibiotics are prescribed primarily to prevent rheumatic fever in this population, they may be mildly helpful in reducing a patient’s symptoms.
Testing. The sensitivity and specificity of high-quality point-of-care tests (POCTs) are on par with those of cultures, with the advantage that the results are available within minutes. Negative tests reduce unneeded antibiotic prescriptions.
Given this evidence, the authors recommend an approach that puts a lot of emphasis on two major things: the risk for rheumatic fever, and clinical assessment. On the basis of these factors, a decision is made about the utility of POCTs and treatment with antibiotics for GAS. The risk for rheumatic fever is based on epidemiology: If the patient is in a low-income country or has recently immigrated from one, then the risk is high, and if not, the risk is low.
Complicated vs uncomplicated? This is determined by clinical assessment of the severity of the patient’s illness, including general appearance. Uncomplicated sore throat means that the patient:
- Is not getting worse after 3 days of illness
- Has a duration of illness ≤ 5 days or is getting better after day 5
- Has mild to moderate symptom severity (bilateral throat pain, the ability to open the mouth fully, and absence of a sandpaper or scarlatiniform rash or strawberry tongue)
For patients with uncomplicated sore throat and low risk for rheumatic fever, the main goals are to reduce antibiotic use and provide symptomatic relief. For these patients, an assessment such as the Centor score can be done. Those with a low Centor score (0-2) can be treated with analgesics and there is no need for a POCT.
In patients with a higher Centor score, the consensus gives two choices: They can either be tested (and treated if the testing is positive), or it is reasonable to forgo testing and use a wait-and-see strategy, with reevaluation if they are getting worse after day 3 or not improving after day 5 days of their illness. Illnesses that last longer than 5 days with sore throat and fatigue should prompt consideration of alternative diagnoses, such as infectious mononucleosis.
For patients with potentially complicated sore throat — including indicators such as worsening symptoms after 3 days or worsening after initiation of antibiotics, inability to open the mouth fully, unilateral neck pain or swelling, or rigors — should undergo a careful evaluation. The need for further testing in these patients, including labs and imaging, should be decided on a case-by-case basis. If the patient appears seriously ill, don’t rely solely on POCT for GAS, but think about other diagnoses.
Rheumatic fever. The approach is very different in patients at high risk for rheumatic fever. POCT for GAS is recommended irrespective of their clinical score, and antibiotics should be prescribed if it’s positive for GAS. If a POCT is unavailable, then the consensus recommends prescribing antibiotics for all high-risk patients who have acute sore throat.
This approach is sensible and puts a lot of emphasis on clinical evaluation, though it should be noted that this approach is considerably different from that in the 2012 Infectious Diseases Society of America guidelines.
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly and Company, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
There is a helpful consensus from experts on the best management of patients with acute sore throat. This is a common problem in primary care, and one for which there is a lot of evidence, opinion, and ultimately overprescribing of antibiotics. This consensus presents a pragmatic clinical approach aimed at decreasing overprescribing, yet detecting which patients are likely to benefit from treatment with antibiotics.
Let’s first go over the evidence that forms the basis for the recommendations, then the recommended approach. First, a sore throat can be caused by many different viruses, as well as group A streptococcus (GAS), the group C streptococcus S dysgalactiae, and fusobacterium. We sometimes think of throat cultures as telling us the definitive etiology of a sore throat. In fact, children commonly are colonized with GAS even when not infected — 35% of the time, when GAS is detected on throat swab in a child, GAS is not the cause of the sore throat. Very few adults are colonized with GAS.
Sore throats are usually self-limited, whether they are treated with antibiotics or not, but occasionally complications can occur. Suppurative complications include peritonsillar abscess, sinusitis and sepsis. Nonsuppurative complications are primarily glomerulonephritis and rheumatic fever, which can lead to rheumatic heart disease.
Antibiotics. Antibiotics have three potential benefits in acute sore throat: to reduce the risk of developing rheumatic heart disease, reduce the duration and severity of symptoms, and treat suppurative complications. The risk for rheumatic heart disease has almost vanished in high-income countries, but not in low-income countries. Thus, antibiotic treatment of acute sore throat due to GAS may benefit those in living in, and those who recently emigrated from, low-income countries.
Patients with suppurative complications should be identified because antibiotics are important for this group. Although antibiotics are prescribed primarily to prevent rheumatic fever in this population, they may be mildly helpful in reducing a patient’s symptoms.
Testing. The sensitivity and specificity of high-quality point-of-care tests (POCTs) are on par with those of cultures, with the advantage that the results are available within minutes. Negative tests reduce unneeded antibiotic prescriptions.
Given this evidence, the authors recommend an approach that puts a lot of emphasis on two major things: the risk for rheumatic fever, and clinical assessment. On the basis of these factors, a decision is made about the utility of POCTs and treatment with antibiotics for GAS. The risk for rheumatic fever is based on epidemiology: If the patient is in a low-income country or has recently immigrated from one, then the risk is high, and if not, the risk is low.
Complicated vs uncomplicated? This is determined by clinical assessment of the severity of the patient’s illness, including general appearance. Uncomplicated sore throat means that the patient:
- Is not getting worse after 3 days of illness
- Has a duration of illness ≤ 5 days or is getting better after day 5
- Has mild to moderate symptom severity (bilateral throat pain, the ability to open the mouth fully, and absence of a sandpaper or scarlatiniform rash or strawberry tongue)
For patients with uncomplicated sore throat and low risk for rheumatic fever, the main goals are to reduce antibiotic use and provide symptomatic relief. For these patients, an assessment such as the Centor score can be done. Those with a low Centor score (0-2) can be treated with analgesics and there is no need for a POCT.
In patients with a higher Centor score, the consensus gives two choices: They can either be tested (and treated if the testing is positive), or it is reasonable to forgo testing and use a wait-and-see strategy, with reevaluation if they are getting worse after day 3 or not improving after day 5 days of their illness. Illnesses that last longer than 5 days with sore throat and fatigue should prompt consideration of alternative diagnoses, such as infectious mononucleosis.
For patients with potentially complicated sore throat — including indicators such as worsening symptoms after 3 days or worsening after initiation of antibiotics, inability to open the mouth fully, unilateral neck pain or swelling, or rigors — should undergo a careful evaluation. The need for further testing in these patients, including labs and imaging, should be decided on a case-by-case basis. If the patient appears seriously ill, don’t rely solely on POCT for GAS, but think about other diagnoses.
Rheumatic fever. The approach is very different in patients at high risk for rheumatic fever. POCT for GAS is recommended irrespective of their clinical score, and antibiotics should be prescribed if it’s positive for GAS. If a POCT is unavailable, then the consensus recommends prescribing antibiotics for all high-risk patients who have acute sore throat.
This approach is sensible and puts a lot of emphasis on clinical evaluation, though it should be noted that this approach is considerably different from that in the 2012 Infectious Diseases Society of America guidelines.
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College, Thomas Jefferson University, Philadelphia, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly and Company, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
Recurrent UTI Rates High Among Older Women, Diagnosing Accurately Is Complicated
TOPLINE:
Accurately diagnosing recurrent urinary tract infections (rUTIs) in older women is challenging and requires careful weighing of the risks and benefits of various treatments, according to a new clinical insight published in JAMA Internal Medicine.
METHODOLOGY:
- Women aged > 65 years have double the rUTI rates compared with younger women, but detecting the condition is more complicated due to age-related conditions, such as overactive bladder related to menopause.
- Overuse of antibiotics can increase their risk of contracting antibiotic-resistant organisms and can lead to pulmonary or hepatic toxic effects in women with reduced kidney function.
- Up to 20% of older women have bacteria in their urine, which may or may not reflect a rUTI.
- Diagnosing rUTIs is complicated if women have dementia or cognitive decline, which can hinder recollection of symptoms.
TAKEAWAYS:
- Clinicians should consider only testing older female patients for rUTIs when symptoms are present and consider all possibilities before making a diagnosis.
- Vaginal estrogen may be an effective treatment, although the authors of the clinical review note a lack of a uniform formulation to recommend. However, oral estrogen use is not supported by evidence, and clinicians should instead consider vaginal creams or rings.
- The drug methenamine may be as effective as antibiotics but may not be safe for women with comorbidities. Evidence supports daily use at 1 g.
- Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
IN PRACTICE:
“Shared decision-making is especially important when diagnosis of an rUTI episode in older women is unclear ... in these cases, clinicians should acknowledge limitations in the evidence and invite patients or their caregivers to discuss preferences about presumptive treatment, weighing the possibility of earlier symptom relief or decreased UTI complications against the risk of adverse drug effects or multidrug resistance.”
SOURCE:
The paper was led by Alison J. Huang, MD, MAS, an internal medicine specialist and researcher in the Department of Medicine at the University of California, San Francisco.
LIMITATIONS:
The authors reported no limitations.
DISCLOSURES:
Dr. Huang received grants from the National Institutes of Health. Other authors reported receiving grants from the Agency for Healthcare Research and Quality, the US Department of Veterans Affairs, the Kahn Foundation, and Nanovibronix.
Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
A version of this article first appeared on Medscape.com.
TOPLINE:
Accurately diagnosing recurrent urinary tract infections (rUTIs) in older women is challenging and requires careful weighing of the risks and benefits of various treatments, according to a new clinical insight published in JAMA Internal Medicine.
METHODOLOGY:
- Women aged > 65 years have double the rUTI rates compared with younger women, but detecting the condition is more complicated due to age-related conditions, such as overactive bladder related to menopause.
- Overuse of antibiotics can increase their risk of contracting antibiotic-resistant organisms and can lead to pulmonary or hepatic toxic effects in women with reduced kidney function.
- Up to 20% of older women have bacteria in their urine, which may or may not reflect a rUTI.
- Diagnosing rUTIs is complicated if women have dementia or cognitive decline, which can hinder recollection of symptoms.
TAKEAWAYS:
- Clinicians should consider only testing older female patients for rUTIs when symptoms are present and consider all possibilities before making a diagnosis.
- Vaginal estrogen may be an effective treatment, although the authors of the clinical review note a lack of a uniform formulation to recommend. However, oral estrogen use is not supported by evidence, and clinicians should instead consider vaginal creams or rings.
- The drug methenamine may be as effective as antibiotics but may not be safe for women with comorbidities. Evidence supports daily use at 1 g.
- Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
IN PRACTICE:
“Shared decision-making is especially important when diagnosis of an rUTI episode in older women is unclear ... in these cases, clinicians should acknowledge limitations in the evidence and invite patients or their caregivers to discuss preferences about presumptive treatment, weighing the possibility of earlier symptom relief or decreased UTI complications against the risk of adverse drug effects or multidrug resistance.”
SOURCE:
The paper was led by Alison J. Huang, MD, MAS, an internal medicine specialist and researcher in the Department of Medicine at the University of California, San Francisco.
LIMITATIONS:
The authors reported no limitations.
DISCLOSURES:
Dr. Huang received grants from the National Institutes of Health. Other authors reported receiving grants from the Agency for Healthcare Research and Quality, the US Department of Veterans Affairs, the Kahn Foundation, and Nanovibronix.
Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
A version of this article first appeared on Medscape.com.
TOPLINE:
Accurately diagnosing recurrent urinary tract infections (rUTIs) in older women is challenging and requires careful weighing of the risks and benefits of various treatments, according to a new clinical insight published in JAMA Internal Medicine.
METHODOLOGY:
- Women aged > 65 years have double the rUTI rates compared with younger women, but detecting the condition is more complicated due to age-related conditions, such as overactive bladder related to menopause.
- Overuse of antibiotics can increase their risk of contracting antibiotic-resistant organisms and can lead to pulmonary or hepatic toxic effects in women with reduced kidney function.
- Up to 20% of older women have bacteria in their urine, which may or may not reflect a rUTI.
- Diagnosing rUTIs is complicated if women have dementia or cognitive decline, which can hinder recollection of symptoms.
TAKEAWAYS:
- Clinicians should consider only testing older female patients for rUTIs when symptoms are present and consider all possibilities before making a diagnosis.
- Vaginal estrogen may be an effective treatment, although the authors of the clinical review note a lack of a uniform formulation to recommend. However, oral estrogen use is not supported by evidence, and clinicians should instead consider vaginal creams or rings.
- The drug methenamine may be as effective as antibiotics but may not be safe for women with comorbidities. Evidence supports daily use at 1 g.
- Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
IN PRACTICE:
“Shared decision-making is especially important when diagnosis of an rUTI episode in older women is unclear ... in these cases, clinicians should acknowledge limitations in the evidence and invite patients or their caregivers to discuss preferences about presumptive treatment, weighing the possibility of earlier symptom relief or decreased UTI complications against the risk of adverse drug effects or multidrug resistance.”
SOURCE:
The paper was led by Alison J. Huang, MD, MAS, an internal medicine specialist and researcher in the Department of Medicine at the University of California, San Francisco.
LIMITATIONS:
The authors reported no limitations.
DISCLOSURES:
Dr. Huang received grants from the National Institutes of Health. Other authors reported receiving grants from the Agency for Healthcare Research and Quality, the US Department of Veterans Affairs, the Kahn Foundation, and Nanovibronix.
Cranberry supplements and behavioral changes may be helpful, but evidence is limited, including among women living in long-term care facilities.
A version of this article first appeared on Medscape.com.
Predicting and Understanding Vaccine Response Determinants
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.
In this column, I recently discussed the impact of the microbiome on childhood vaccine responses. My group has been expanding our research on the topic of childhood vaccine response and its relationship to infection proneness. Therefore, I want to share new research findings.
Immune responsiveness to vaccines varies among children, leaving some susceptible to infections. We also have evidence that the immune deficiencies that contribute to poor vaccine responsiveness also manifest in children as respiratory infection proneness.
Predicting Vaccine Response in the Neonatal Period
The first 100 days of life is an amazing transition time in early life. During that time, the immune system is highly influenced by environmental factors that generate epigenetic changes affecting vaccine responsiveness. Some publications have used the term “window of opportunity,” because it is thought that interventions to change a negative trajectory to a positive one for vaccine responsiveness have a better potential to be effective. Predicting which children will be poorly responsive to vaccines would be desirable, so those children could be specifically identified for intervention. Doing so in the neonatal age time frame using easy-to-obtain clinical samples would be a bonus.
In our most recent study, we sought to identify cytokine biosignatures in the neonatal period, measured in convenient nasopharyngeal secretions, that predict vaccine responses, measured as antibody levels to various vaccines at 1 year of life. Secondly, we assessed the effect of antibiotic exposures on vaccine responses in the study cohort. Third, we tested for induction of CD4+ T-cell vaccine-specific immune memory at infant age 1 year. Fourth, we studied antigen presenting cells (APCs) at rest and in response to an adjuvant called R848, known to stimulate toll-like receptor (TLR) 7/8 agonist, to assess its effects on the immune cells of low vaccine responder children, compared with other children.1
The study population consisted of 101 infants recruited from two primary care pediatric practices in/near Rochester, New York. Children lived in suburban and rural environments. Enrollment and sampling occurred during 2017-2020. All participants received regularly scheduled childhood vaccinations according to the recommendations by US Centers for Disease Control. Nasopharyngeal swabs were used to collect nasal secretions. Antibody titers against six antigens were measured at approximately 1 year of age from all 72 available blood samples. The protective threshold of the corresponding vaccine antigen divided each vaccine-induced antibody level and the ratio considered a normalized titer. The normalized antibody titers were used to define vaccine responsiveness groups as Low Vaccine Responder (bottom 25th percentile of vaccine responders, n = 18 children), as Normal Vaccine Responder (25-75th percentile of vaccine responders, n = 36 children) and as High Vaccine Responder (top 25th percentile of vaccine responders, n = 18 children).
We found that specific nasal cytokine levels measured at newborn age 1 week old, 2 weeks old, and 3 weeks old were predictive of the vaccine response groupings measured at child age 1 year old, following their primary series of vaccinations. The P values varied between less than .05 to .001.
Five newborns had antibiotic exposure at/near the time of birth; 4 [80%] of the 5 were Low Vaccine Responders vs 1 [2%] of 60 Normal+High Vaccine Responder children, P = .006. Also, the cumulative days of antibiotic exposure up to 1 year was highly associated with low vaccine responders, compared with Normal+High Vaccine Responder children (P = 2 x 10-16).
We found that Low Vaccine Responder infants had reduced vaccine-specific T-helper memory cells producing INFg and IL-2 (Th1 cytokines) and IL-4 (Th2 cytokines), compared with Normal+High Vaccine Responder children. In the absence of sufficient numbers of antigen-specific memory CD4+ T-cells, a child would become unprotected from the target infection that the vaccines were intended to prevent after the antibody levels wane.
We found that Low Vaccine Responder antigen-presenting cells are different from those in normal vaccine responders and they can be distinguished when at rest and when stimulated by a specific adjuvant — R848. Our previous findings suggested that Low Vaccine Responder children have a prolonged neonatal-like immune profile (PNIP).2 Therefore, stimulating the immune system of a Low Vaccine Responder could shift their cellular immune responses to behave like cells of Normal+High Vaccine Responder children.
In summary, we identified cytokine biosignatures measured in nasopharyngeal secretions in the neonatal period that predicted vaccine response groups measured as antibody levels at 1 year of life. We showed that reduced vaccine responsiveness was associated with antibiotic exposure at/near birth and with cumulative exposure during the first year of life. We found that Low Vaccine Responder children at 1 year old have fewer vaccine-specific memory CD4+ Th1 and Th2-cells and that antigen-presenting cells at rest and in response to R848 antigen stimulation differ, compared with Normal+High Vaccine Responder children.
Future work by our group will focus on exploring early-life risk factors that influence differences in vaccine responsiveness and interventions that might shift a child’s responsiveness from low to normal or high.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (New York) General Hospital. He has no conflicts of interest to declare.
References
1. Pichichero ME et al. Variability of Vaccine Responsiveness in Young Children. J Infect Dis. 2023 Nov 22:jiad524. doi: 10.1093/infdis/jiad524.
2. Pichichero ME et al. Functional Immune Cell Differences Associated with Low Vaccine Responses in Infants. J Infect Dis. 2016 Jun 15;213(12):2014-2019. doi: 10.1093/infdis/jiw053.

