User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Clozapine and Respiratory Infection Risk: What to Know
Clozapine is considered the drug of choice for treatment-resistant schizophrenia in guidelines globally, but it remains significantly underutilized. This is largely due to its range of side effects, particularly its increased infection risk which prompted the US Food and Drug Administration (FDA) to mandate regular blood testing to monitor neutrophil counts.
The COVID-19 pandemic raised new concerns about the care of clozapine-treated patients, leading clinicians and patients to urge the FDA to relax prescription requirements for the drug under the Risk Evaluation and Mitigation Strategy (REMS) program.
As the FDA prepares for a public hearing in November on proposed adjustments to the drug’s REMS criteria, a growing body of research is challenging the previous understanding of clozapine and infection risk.
Clarifying the Risk
Research on the link between clozapine and respiratory infections has produced conflicting results. Some studies indicate little to no increased risk for mild COVID-19 and other respiratory illnesses, while others have shown a higher likelihood of severe infection.
A recent nationwide Danish registry study of respiratory infections in people with a schizophrenia spectrum disorder could bring some clarity, Maxime Taquet, MD, a clinical lecturer at the University of Oxford, Warneford Hospital, Oxford, England, told this news organization.
By tracking periods when patients were on and off clozapine and other antipsychotics, the study offers more precise risk estimates, distinguishing the risks associated with the antipsychotic from those related to underlying schizophrenia, said Dr. Taquet, who authored an accompanying editorial on the study.
“It’s very important to try to disentangle the effects of schizophrenia, its severity, from the medication,” Dr. Taquet said. “I think that the Danish study is the first to try and really do that with as much precision as possible.”
After adjusting for key confounders including economic status and COVID-19 vaccination status, the researchers found that individuals taking antipsychotics had lower odds of testing positive for SARS-CoV-2 and similar rates of filled anti-infective prescriptions as those not taking the drugs.
Although antipsychotic use was not linked to higher rates of mild infection, it was linked to an increased risk for COVID-19 hospitalization in individuals older than 70 years, as well as hospitalization and death from other respiratory infections, mainly pneumonia, in those older than 40 years.
Notably, there was no excess risk for any outcome with clozapine vs other antipsychotics.
Strong Link to Pneumonia Risk
Results from a longitudinal Finnish study, just published in The American Journal of Psychiatry, also show an increased risk for severe outcomes from ileus and pneumonia among more than 2600 patients with schizophrenia taking clozapine.
Twenty years after initiating clozapine, the cumulative incidence estimate for ileus was 5.3% — more than sixfold higher than previously reported. The incidence of pneumonia was also high, at 29.5%.
Both illnesses were significantly associated with mortality, with odds ratios of 4.5 and 2.8, respectively.
These findings align with previous pharmacovigilance studies, with reported mortality rates for gastrointestinal hypomotility and pneumonia that were 4-10 times higher than those for agranulocytosis, the researchers said.
The study “really adds to a growing body of research suggesting a connection between clozapine use and a higher risk of developing pneumonia,” Robert O. Cotes, MD, a professor of psychiatry and behavioral sciences at Emory University, Atlanta, who specializes in the use of clozapine, told this news organization.
“Additionally, when people on clozapine do contract pneumonia, there’s concern the condition may be more dangerous,” he added.
A Closer Look at Neutropenia Risk
Neutropenia receives the lion’s share of attention among clozapine’s potential side effects, but this focus may need to be re-evaluated, Dr. Cotes said.
He pointed out that recent data suggest the risk for severe neutropenia, 2-3 years after initiating clozapine, is comparable to that of other antipsychotics.
A study of 26,630 clozapine users in Australia and New Zealand showed that most cases of severe neutropenia leading to clozapine cessation peaked within 18 weeks and was negligible after 2 years. This suggests weekly hematologic monitoring could potentially be discontinued after the 2-year mark.
Another study reported earlier this year by this news organization showed a low risk for mild or moderate neutropenia and no severe cases in nearly 1000 people taking clozapine.
“I worry that we may be missing the forest for the trees by hyperfocusing on neutropenia and not considering clozapine’s other potential serious side effects like pneumonia, myocarditis, and gastrointestinal hypermotility,” Dr. Cotes said.
Importance of Vaccines
The findings of these studies highlight the importance of vaccines in this at-risk group, said Dr. Taquet, a point emphasized by investigators of the Danish study he reviewed.
“Inspired by the experience of COVID-19 vaccine prioritization in severe mental illness and based on our findings, there is momentum for preventive action,” the authors wrote. “Our findings do not suggest the avoidance of specific antipsychotics but rather a call for increased vigilance regarding this at-risk group.”
This includes recommending pneumococcal, influenza, COVID-19, and other anti-infective vaccines in those older than 40 years treated with, or due to start, an antipsychotic.
“It’s not mandatory, but we do recommend that patients on clozapine get the regular vaccines,” Dr. Taquet said.
Pointing to the recent study on pneumonia risk, Dr. Cotes said addressing underlying risk factors, such as smoking, obesity, and possibly sedation and excessive salivation caused by clozapine, is key.
“And to make sure that vaccinations are up to date, particularly heading into this fall,” he added.
Rethinking Clozapine REMS
One of the most challenging issues facing clinicians and researchers is how to help people understand the safety profile of clozapine and to use it with more confidence, Dr. Cotes said.
“A lot of people hear about clozapine and they think about neutropenia, they think about side effects, the REMS system, and all of these factors really drive down clozapine utilization,” he said.
Treatment-resistant schizophrenia affects about a quarter of those with schizophrenia, yet only 4% of these patients receive clozapine in the United States, Dr. Cotes said. That number may be even lower for its other indication of reducing suicidal behavior in patients with schizophrenia or schizoaffective disorder.
The clozapine REMS is viewed as a major barrier to utilization and requires certification of pharmacists and physicians and use of a central system to monitor absolute neutrophil counts for neutropenia in patients.
As previously reported by this news organization in November 2022, the FDA opted to temporarily exercise enforcement discretion for certain aspects of the drug safety program to ensure continuity of care for patients after concerns were raised by the American Psychiatric Association (APA) along with other professional organizations.
Even with that temporary enforcement discretion, “reports have shown that over half of those prescribed clozapine have trouble accessing the medication because of the REMS program,” a spokesperson for the APA told this news organization.
“Not only are patients having trouble accessing the medication, many have trouble finding a prescriber in their geographic locations and others because of the monitoring requirements have their treatment discontinued leading to negative outcomes,” the spokesperson said.
The FDA is currently reviewing the clozapine REMS and is holding a joint advisory committee meeting on November 19 to discuss the review and “possible changes to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of clozapine.”
The APA plans to submit written and oral comments to the advisory committees.
“We are hopeful that the re-evaluation meeting in November will remove barriers and increase access to clozapine, which is currently highly underutilized, especially in marginalized communities,” the spokesperson said.
Dr. Cotes reported serving as a speaker and consultant for Saladax Biomedical and as a consultant for Syneos Health. Dr. Taquet reported having no competing interests.
A version of this article first appeared on Medscape.com.
Clozapine is considered the drug of choice for treatment-resistant schizophrenia in guidelines globally, but it remains significantly underutilized. This is largely due to its range of side effects, particularly its increased infection risk which prompted the US Food and Drug Administration (FDA) to mandate regular blood testing to monitor neutrophil counts.
The COVID-19 pandemic raised new concerns about the care of clozapine-treated patients, leading clinicians and patients to urge the FDA to relax prescription requirements for the drug under the Risk Evaluation and Mitigation Strategy (REMS) program.
As the FDA prepares for a public hearing in November on proposed adjustments to the drug’s REMS criteria, a growing body of research is challenging the previous understanding of clozapine and infection risk.
Clarifying the Risk
Research on the link between clozapine and respiratory infections has produced conflicting results. Some studies indicate little to no increased risk for mild COVID-19 and other respiratory illnesses, while others have shown a higher likelihood of severe infection.
A recent nationwide Danish registry study of respiratory infections in people with a schizophrenia spectrum disorder could bring some clarity, Maxime Taquet, MD, a clinical lecturer at the University of Oxford, Warneford Hospital, Oxford, England, told this news organization.
By tracking periods when patients were on and off clozapine and other antipsychotics, the study offers more precise risk estimates, distinguishing the risks associated with the antipsychotic from those related to underlying schizophrenia, said Dr. Taquet, who authored an accompanying editorial on the study.
“It’s very important to try to disentangle the effects of schizophrenia, its severity, from the medication,” Dr. Taquet said. “I think that the Danish study is the first to try and really do that with as much precision as possible.”
After adjusting for key confounders including economic status and COVID-19 vaccination status, the researchers found that individuals taking antipsychotics had lower odds of testing positive for SARS-CoV-2 and similar rates of filled anti-infective prescriptions as those not taking the drugs.
Although antipsychotic use was not linked to higher rates of mild infection, it was linked to an increased risk for COVID-19 hospitalization in individuals older than 70 years, as well as hospitalization and death from other respiratory infections, mainly pneumonia, in those older than 40 years.
Notably, there was no excess risk for any outcome with clozapine vs other antipsychotics.
Strong Link to Pneumonia Risk
Results from a longitudinal Finnish study, just published in The American Journal of Psychiatry, also show an increased risk for severe outcomes from ileus and pneumonia among more than 2600 patients with schizophrenia taking clozapine.
Twenty years after initiating clozapine, the cumulative incidence estimate for ileus was 5.3% — more than sixfold higher than previously reported. The incidence of pneumonia was also high, at 29.5%.
Both illnesses were significantly associated with mortality, with odds ratios of 4.5 and 2.8, respectively.
These findings align with previous pharmacovigilance studies, with reported mortality rates for gastrointestinal hypomotility and pneumonia that were 4-10 times higher than those for agranulocytosis, the researchers said.
The study “really adds to a growing body of research suggesting a connection between clozapine use and a higher risk of developing pneumonia,” Robert O. Cotes, MD, a professor of psychiatry and behavioral sciences at Emory University, Atlanta, who specializes in the use of clozapine, told this news organization.
“Additionally, when people on clozapine do contract pneumonia, there’s concern the condition may be more dangerous,” he added.
A Closer Look at Neutropenia Risk
Neutropenia receives the lion’s share of attention among clozapine’s potential side effects, but this focus may need to be re-evaluated, Dr. Cotes said.
He pointed out that recent data suggest the risk for severe neutropenia, 2-3 years after initiating clozapine, is comparable to that of other antipsychotics.
A study of 26,630 clozapine users in Australia and New Zealand showed that most cases of severe neutropenia leading to clozapine cessation peaked within 18 weeks and was negligible after 2 years. This suggests weekly hematologic monitoring could potentially be discontinued after the 2-year mark.
Another study reported earlier this year by this news organization showed a low risk for mild or moderate neutropenia and no severe cases in nearly 1000 people taking clozapine.
“I worry that we may be missing the forest for the trees by hyperfocusing on neutropenia and not considering clozapine’s other potential serious side effects like pneumonia, myocarditis, and gastrointestinal hypermotility,” Dr. Cotes said.
Importance of Vaccines
The findings of these studies highlight the importance of vaccines in this at-risk group, said Dr. Taquet, a point emphasized by investigators of the Danish study he reviewed.
“Inspired by the experience of COVID-19 vaccine prioritization in severe mental illness and based on our findings, there is momentum for preventive action,” the authors wrote. “Our findings do not suggest the avoidance of specific antipsychotics but rather a call for increased vigilance regarding this at-risk group.”
This includes recommending pneumococcal, influenza, COVID-19, and other anti-infective vaccines in those older than 40 years treated with, or due to start, an antipsychotic.
“It’s not mandatory, but we do recommend that patients on clozapine get the regular vaccines,” Dr. Taquet said.
Pointing to the recent study on pneumonia risk, Dr. Cotes said addressing underlying risk factors, such as smoking, obesity, and possibly sedation and excessive salivation caused by clozapine, is key.
“And to make sure that vaccinations are up to date, particularly heading into this fall,” he added.
Rethinking Clozapine REMS
One of the most challenging issues facing clinicians and researchers is how to help people understand the safety profile of clozapine and to use it with more confidence, Dr. Cotes said.
“A lot of people hear about clozapine and they think about neutropenia, they think about side effects, the REMS system, and all of these factors really drive down clozapine utilization,” he said.
Treatment-resistant schizophrenia affects about a quarter of those with schizophrenia, yet only 4% of these patients receive clozapine in the United States, Dr. Cotes said. That number may be even lower for its other indication of reducing suicidal behavior in patients with schizophrenia or schizoaffective disorder.
The clozapine REMS is viewed as a major barrier to utilization and requires certification of pharmacists and physicians and use of a central system to monitor absolute neutrophil counts for neutropenia in patients.
As previously reported by this news organization in November 2022, the FDA opted to temporarily exercise enforcement discretion for certain aspects of the drug safety program to ensure continuity of care for patients after concerns were raised by the American Psychiatric Association (APA) along with other professional organizations.
Even with that temporary enforcement discretion, “reports have shown that over half of those prescribed clozapine have trouble accessing the medication because of the REMS program,” a spokesperson for the APA told this news organization.
“Not only are patients having trouble accessing the medication, many have trouble finding a prescriber in their geographic locations and others because of the monitoring requirements have their treatment discontinued leading to negative outcomes,” the spokesperson said.
The FDA is currently reviewing the clozapine REMS and is holding a joint advisory committee meeting on November 19 to discuss the review and “possible changes to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of clozapine.”
The APA plans to submit written and oral comments to the advisory committees.
“We are hopeful that the re-evaluation meeting in November will remove barriers and increase access to clozapine, which is currently highly underutilized, especially in marginalized communities,” the spokesperson said.
Dr. Cotes reported serving as a speaker and consultant for Saladax Biomedical and as a consultant for Syneos Health. Dr. Taquet reported having no competing interests.
A version of this article first appeared on Medscape.com.
Clozapine is considered the drug of choice for treatment-resistant schizophrenia in guidelines globally, but it remains significantly underutilized. This is largely due to its range of side effects, particularly its increased infection risk which prompted the US Food and Drug Administration (FDA) to mandate regular blood testing to monitor neutrophil counts.
The COVID-19 pandemic raised new concerns about the care of clozapine-treated patients, leading clinicians and patients to urge the FDA to relax prescription requirements for the drug under the Risk Evaluation and Mitigation Strategy (REMS) program.
As the FDA prepares for a public hearing in November on proposed adjustments to the drug’s REMS criteria, a growing body of research is challenging the previous understanding of clozapine and infection risk.
Clarifying the Risk
Research on the link between clozapine and respiratory infections has produced conflicting results. Some studies indicate little to no increased risk for mild COVID-19 and other respiratory illnesses, while others have shown a higher likelihood of severe infection.
A recent nationwide Danish registry study of respiratory infections in people with a schizophrenia spectrum disorder could bring some clarity, Maxime Taquet, MD, a clinical lecturer at the University of Oxford, Warneford Hospital, Oxford, England, told this news organization.
By tracking periods when patients were on and off clozapine and other antipsychotics, the study offers more precise risk estimates, distinguishing the risks associated with the antipsychotic from those related to underlying schizophrenia, said Dr. Taquet, who authored an accompanying editorial on the study.
“It’s very important to try to disentangle the effects of schizophrenia, its severity, from the medication,” Dr. Taquet said. “I think that the Danish study is the first to try and really do that with as much precision as possible.”
After adjusting for key confounders including economic status and COVID-19 vaccination status, the researchers found that individuals taking antipsychotics had lower odds of testing positive for SARS-CoV-2 and similar rates of filled anti-infective prescriptions as those not taking the drugs.
Although antipsychotic use was not linked to higher rates of mild infection, it was linked to an increased risk for COVID-19 hospitalization in individuals older than 70 years, as well as hospitalization and death from other respiratory infections, mainly pneumonia, in those older than 40 years.
Notably, there was no excess risk for any outcome with clozapine vs other antipsychotics.
Strong Link to Pneumonia Risk
Results from a longitudinal Finnish study, just published in The American Journal of Psychiatry, also show an increased risk for severe outcomes from ileus and pneumonia among more than 2600 patients with schizophrenia taking clozapine.
Twenty years after initiating clozapine, the cumulative incidence estimate for ileus was 5.3% — more than sixfold higher than previously reported. The incidence of pneumonia was also high, at 29.5%.
Both illnesses were significantly associated with mortality, with odds ratios of 4.5 and 2.8, respectively.
These findings align with previous pharmacovigilance studies, with reported mortality rates for gastrointestinal hypomotility and pneumonia that were 4-10 times higher than those for agranulocytosis, the researchers said.
The study “really adds to a growing body of research suggesting a connection between clozapine use and a higher risk of developing pneumonia,” Robert O. Cotes, MD, a professor of psychiatry and behavioral sciences at Emory University, Atlanta, who specializes in the use of clozapine, told this news organization.
“Additionally, when people on clozapine do contract pneumonia, there’s concern the condition may be more dangerous,” he added.
A Closer Look at Neutropenia Risk
Neutropenia receives the lion’s share of attention among clozapine’s potential side effects, but this focus may need to be re-evaluated, Dr. Cotes said.
He pointed out that recent data suggest the risk for severe neutropenia, 2-3 years after initiating clozapine, is comparable to that of other antipsychotics.
A study of 26,630 clozapine users in Australia and New Zealand showed that most cases of severe neutropenia leading to clozapine cessation peaked within 18 weeks and was negligible after 2 years. This suggests weekly hematologic monitoring could potentially be discontinued after the 2-year mark.
Another study reported earlier this year by this news organization showed a low risk for mild or moderate neutropenia and no severe cases in nearly 1000 people taking clozapine.
“I worry that we may be missing the forest for the trees by hyperfocusing on neutropenia and not considering clozapine’s other potential serious side effects like pneumonia, myocarditis, and gastrointestinal hypermotility,” Dr. Cotes said.
Importance of Vaccines
The findings of these studies highlight the importance of vaccines in this at-risk group, said Dr. Taquet, a point emphasized by investigators of the Danish study he reviewed.
“Inspired by the experience of COVID-19 vaccine prioritization in severe mental illness and based on our findings, there is momentum for preventive action,” the authors wrote. “Our findings do not suggest the avoidance of specific antipsychotics but rather a call for increased vigilance regarding this at-risk group.”
This includes recommending pneumococcal, influenza, COVID-19, and other anti-infective vaccines in those older than 40 years treated with, or due to start, an antipsychotic.
“It’s not mandatory, but we do recommend that patients on clozapine get the regular vaccines,” Dr. Taquet said.
Pointing to the recent study on pneumonia risk, Dr. Cotes said addressing underlying risk factors, such as smoking, obesity, and possibly sedation and excessive salivation caused by clozapine, is key.
“And to make sure that vaccinations are up to date, particularly heading into this fall,” he added.
Rethinking Clozapine REMS
One of the most challenging issues facing clinicians and researchers is how to help people understand the safety profile of clozapine and to use it with more confidence, Dr. Cotes said.
“A lot of people hear about clozapine and they think about neutropenia, they think about side effects, the REMS system, and all of these factors really drive down clozapine utilization,” he said.
Treatment-resistant schizophrenia affects about a quarter of those with schizophrenia, yet only 4% of these patients receive clozapine in the United States, Dr. Cotes said. That number may be even lower for its other indication of reducing suicidal behavior in patients with schizophrenia or schizoaffective disorder.
The clozapine REMS is viewed as a major barrier to utilization and requires certification of pharmacists and physicians and use of a central system to monitor absolute neutrophil counts for neutropenia in patients.
As previously reported by this news organization in November 2022, the FDA opted to temporarily exercise enforcement discretion for certain aspects of the drug safety program to ensure continuity of care for patients after concerns were raised by the American Psychiatric Association (APA) along with other professional organizations.
Even with that temporary enforcement discretion, “reports have shown that over half of those prescribed clozapine have trouble accessing the medication because of the REMS program,” a spokesperson for the APA told this news organization.
“Not only are patients having trouble accessing the medication, many have trouble finding a prescriber in their geographic locations and others because of the monitoring requirements have their treatment discontinued leading to negative outcomes,” the spokesperson said.
The FDA is currently reviewing the clozapine REMS and is holding a joint advisory committee meeting on November 19 to discuss the review and “possible changes to minimize burden on patients, pharmacies, and prescribers while maintaining safe use of clozapine.”
The APA plans to submit written and oral comments to the advisory committees.
“We are hopeful that the re-evaluation meeting in November will remove barriers and increase access to clozapine, which is currently highly underutilized, especially in marginalized communities,” the spokesperson said.
Dr. Cotes reported serving as a speaker and consultant for Saladax Biomedical and as a consultant for Syneos Health. Dr. Taquet reported having no competing interests.
A version of this article first appeared on Medscape.com.
Anticipated Effects of Pneumococcal Vaccines on Otitis
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).
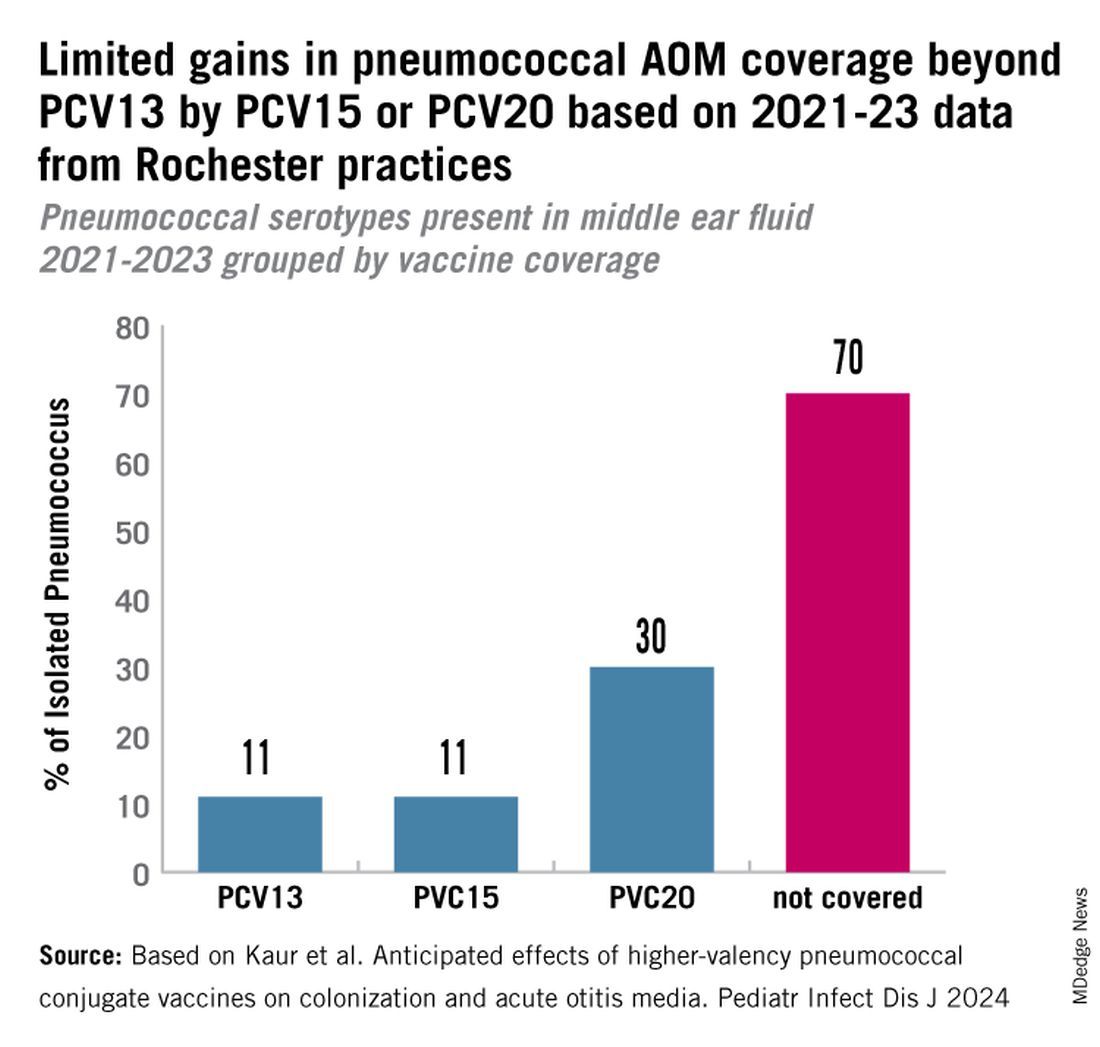
Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).

Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).

Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.
Reduced Vaccination Rates Contribute to Rising Pertussis Numbers
New data from the Centers for Disease Control and Prevention (CDC) show significant spikes in pertussis cases compared with last year, especially in several urban areas including New York, Illinois, Florida, and Colorado.
Notably, the current pertussis case count in Illinois as of September 21, 2024, was five times higher than the total cases in 2023 (1058 vs 50). New York City alone had reported 624 cases as of September 21, compared with 38 cases in 2023.
Additional data from the CDC on vaccination coverage and exemptions of school-aged children showed an increase from 3.0% last year to 3.3% in 2024 of children who were exempted from recommended vaccination requirements. Although nearly 93% of kindergarteners in the United States received recommended vaccines (including Tdap), similar to last year, this number shows a steady decline from 94% in the 2021-2021 school year and 93% in the 2021-2022 school year, according to previous CDC reports.
What’s Happening in the Clinic
Clinical experience and the most recent CDC data point to under vaccination as a driver of the increased pertussis cases this year, David J. Cennimo, MD, associate professor of medicine and pediatrics in the division of infectious disease at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
Although the pertussis vaccination rates in infancy are still very good, clinicians are seeing a drop-off in school-aged children and adults, and the lingering anti-vaccine efforts from the COVID-19 pandemic period are undoubtedly playing a part, said Dr. Cennimo. “Unfortunately, pertussis is contagious, and the vaccine effectiveness wears off. Having decreased numbers of people protected results in more rapid spread,” he said.
Dr. Cennimo agreed that the number of cases in the United States is underreported, and even higher than the data suggest. “I’m sure of it; the initial clinical presentation may be mistaken for a viral upper respiratory tract infection (common cold),” he told this news organization.
Many older children and adults with pertussis do not manifest the classic “whooping cough” seen in infants and young children, so making a clinical diagnosis can be difficult, he said. “One classical component of the illness is a prolonged cough. I have wondered if some people now reporting a lingering cough had pertussis that was missed,” Dr. Cennimo noted.
“Clinicians should stress the value of boosters in a vaccine-preventable illness where we know immunity wanes overtime,” Dr. Cennimo said. “We have a great remedy in the Tdap vaccine, which we should all be getting very 10 years,” he said.
He also emphasized that clinicians remind pregnant women of the current recommendations to receive the Tdap vaccine for every pregnancy. “Vaccination during pregnancy is the best way to protect both the pregnant person and the newborn.
Even for the vaccine hesitant, this vaccine has a long track record of safety so should not be a significant concern,” he said.
The ultimate take-home message is not a new one, and applies to all illnesses, Dr. Cennimo told this news organization. Simply put, “Stay home if you are sick. Social distancing is not just for COVID-19,” he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
New data from the Centers for Disease Control and Prevention (CDC) show significant spikes in pertussis cases compared with last year, especially in several urban areas including New York, Illinois, Florida, and Colorado.
Notably, the current pertussis case count in Illinois as of September 21, 2024, was five times higher than the total cases in 2023 (1058 vs 50). New York City alone had reported 624 cases as of September 21, compared with 38 cases in 2023.
Additional data from the CDC on vaccination coverage and exemptions of school-aged children showed an increase from 3.0% last year to 3.3% in 2024 of children who were exempted from recommended vaccination requirements. Although nearly 93% of kindergarteners in the United States received recommended vaccines (including Tdap), similar to last year, this number shows a steady decline from 94% in the 2021-2021 school year and 93% in the 2021-2022 school year, according to previous CDC reports.
What’s Happening in the Clinic
Clinical experience and the most recent CDC data point to under vaccination as a driver of the increased pertussis cases this year, David J. Cennimo, MD, associate professor of medicine and pediatrics in the division of infectious disease at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
Although the pertussis vaccination rates in infancy are still very good, clinicians are seeing a drop-off in school-aged children and adults, and the lingering anti-vaccine efforts from the COVID-19 pandemic period are undoubtedly playing a part, said Dr. Cennimo. “Unfortunately, pertussis is contagious, and the vaccine effectiveness wears off. Having decreased numbers of people protected results in more rapid spread,” he said.
Dr. Cennimo agreed that the number of cases in the United States is underreported, and even higher than the data suggest. “I’m sure of it; the initial clinical presentation may be mistaken for a viral upper respiratory tract infection (common cold),” he told this news organization.
Many older children and adults with pertussis do not manifest the classic “whooping cough” seen in infants and young children, so making a clinical diagnosis can be difficult, he said. “One classical component of the illness is a prolonged cough. I have wondered if some people now reporting a lingering cough had pertussis that was missed,” Dr. Cennimo noted.
“Clinicians should stress the value of boosters in a vaccine-preventable illness where we know immunity wanes overtime,” Dr. Cennimo said. “We have a great remedy in the Tdap vaccine, which we should all be getting very 10 years,” he said.
He also emphasized that clinicians remind pregnant women of the current recommendations to receive the Tdap vaccine for every pregnancy. “Vaccination during pregnancy is the best way to protect both the pregnant person and the newborn.
Even for the vaccine hesitant, this vaccine has a long track record of safety so should not be a significant concern,” he said.
The ultimate take-home message is not a new one, and applies to all illnesses, Dr. Cennimo told this news organization. Simply put, “Stay home if you are sick. Social distancing is not just for COVID-19,” he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
New data from the Centers for Disease Control and Prevention (CDC) show significant spikes in pertussis cases compared with last year, especially in several urban areas including New York, Illinois, Florida, and Colorado.
Notably, the current pertussis case count in Illinois as of September 21, 2024, was five times higher than the total cases in 2023 (1058 vs 50). New York City alone had reported 624 cases as of September 21, compared with 38 cases in 2023.
Additional data from the CDC on vaccination coverage and exemptions of school-aged children showed an increase from 3.0% last year to 3.3% in 2024 of children who were exempted from recommended vaccination requirements. Although nearly 93% of kindergarteners in the United States received recommended vaccines (including Tdap), similar to last year, this number shows a steady decline from 94% in the 2021-2021 school year and 93% in the 2021-2022 school year, according to previous CDC reports.
What’s Happening in the Clinic
Clinical experience and the most recent CDC data point to under vaccination as a driver of the increased pertussis cases this year, David J. Cennimo, MD, associate professor of medicine and pediatrics in the division of infectious disease at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
Although the pertussis vaccination rates in infancy are still very good, clinicians are seeing a drop-off in school-aged children and adults, and the lingering anti-vaccine efforts from the COVID-19 pandemic period are undoubtedly playing a part, said Dr. Cennimo. “Unfortunately, pertussis is contagious, and the vaccine effectiveness wears off. Having decreased numbers of people protected results in more rapid spread,” he said.
Dr. Cennimo agreed that the number of cases in the United States is underreported, and even higher than the data suggest. “I’m sure of it; the initial clinical presentation may be mistaken for a viral upper respiratory tract infection (common cold),” he told this news organization.
Many older children and adults with pertussis do not manifest the classic “whooping cough” seen in infants and young children, so making a clinical diagnosis can be difficult, he said. “One classical component of the illness is a prolonged cough. I have wondered if some people now reporting a lingering cough had pertussis that was missed,” Dr. Cennimo noted.
“Clinicians should stress the value of boosters in a vaccine-preventable illness where we know immunity wanes overtime,” Dr. Cennimo said. “We have a great remedy in the Tdap vaccine, which we should all be getting very 10 years,” he said.
He also emphasized that clinicians remind pregnant women of the current recommendations to receive the Tdap vaccine for every pregnancy. “Vaccination during pregnancy is the best way to protect both the pregnant person and the newborn.
Even for the vaccine hesitant, this vaccine has a long track record of safety so should not be a significant concern,” he said.
The ultimate take-home message is not a new one, and applies to all illnesses, Dr. Cennimo told this news organization. Simply put, “Stay home if you are sick. Social distancing is not just for COVID-19,” he said.
Dr. Cennimo had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Public Health, Not Politics, Should Drive Mask Policies, Says Ethicist
This transcript has been edited for clarity.
I recently saw a ban that has me very worried, concerned, and strongly in opposition.
Basically, the standard kind of medical mask would be captured, although I think their aim in doing this was to try to discourage people at political protests from being able to wear masks and hide their identity. They’re basically trying to discourage that. This is particularly triggered by, I think, protests about the invasion of Israel, the war that resulted in Gaza, and the demonstrations that have gone on around the country, with many people masked.
There may be issues about what is acceptable to wear when you go to a demonstration. I don’t claim to know about the civil rights of that.
In a time at which COVID-19 is flourishing, really on the rebound, expanding fast, and still causing 600 deaths a week; the flu season is going to be upon us soon enough; and there are also concerns about the possibility of avian flu jumping into the human population, it is absolutely the wrong time to single out those who are trying to mask for health reasons.
Basically, there are two strong reasons. One, there are people out there who wear a medical mask or mask for a medical reason because they have an underlying disease. They may have had a transplant or they may feel they’re immunocompromised for some reason. They worry that, if they don’t wear a mask, they’re going to get an infection from something like COVID-19 or flu, which could really be super-dangerous for them.
The other reason people mask is to protect their family members. They may have someone who’s immunocompromised in the family, or they’re doing it kindly and altruistically to protect the rest of us and to stop viruses from circulating.
These bans are not taking into account public health. They’re being brought forward in the midst of political heat about demonstrations and political issues. I think they should be opposed. I do not think they should be enacted.
I think the medical rights of people with disabilities and immunologic disorders, and those who want to mask to prevent getting sick at a time at which infectious diseases are still circulating and killing people, ought to take priority. Public health, in this case, should drive our policies about masks.
Dr. Caplan, director, Division of Medical Ethics, New York University Langone Medical Center, New York, NY, served on Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position), and is a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently saw a ban that has me very worried, concerned, and strongly in opposition.
Basically, the standard kind of medical mask would be captured, although I think their aim in doing this was to try to discourage people at political protests from being able to wear masks and hide their identity. They’re basically trying to discourage that. This is particularly triggered by, I think, protests about the invasion of Israel, the war that resulted in Gaza, and the demonstrations that have gone on around the country, with many people masked.
There may be issues about what is acceptable to wear when you go to a demonstration. I don’t claim to know about the civil rights of that.
In a time at which COVID-19 is flourishing, really on the rebound, expanding fast, and still causing 600 deaths a week; the flu season is going to be upon us soon enough; and there are also concerns about the possibility of avian flu jumping into the human population, it is absolutely the wrong time to single out those who are trying to mask for health reasons.
Basically, there are two strong reasons. One, there are people out there who wear a medical mask or mask for a medical reason because they have an underlying disease. They may have had a transplant or they may feel they’re immunocompromised for some reason. They worry that, if they don’t wear a mask, they’re going to get an infection from something like COVID-19 or flu, which could really be super-dangerous for them.
The other reason people mask is to protect their family members. They may have someone who’s immunocompromised in the family, or they’re doing it kindly and altruistically to protect the rest of us and to stop viruses from circulating.
These bans are not taking into account public health. They’re being brought forward in the midst of political heat about demonstrations and political issues. I think they should be opposed. I do not think they should be enacted.
I think the medical rights of people with disabilities and immunologic disorders, and those who want to mask to prevent getting sick at a time at which infectious diseases are still circulating and killing people, ought to take priority. Public health, in this case, should drive our policies about masks.
Dr. Caplan, director, Division of Medical Ethics, New York University Langone Medical Center, New York, NY, served on Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position), and is a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently saw a ban that has me very worried, concerned, and strongly in opposition.
Basically, the standard kind of medical mask would be captured, although I think their aim in doing this was to try to discourage people at political protests from being able to wear masks and hide their identity. They’re basically trying to discourage that. This is particularly triggered by, I think, protests about the invasion of Israel, the war that resulted in Gaza, and the demonstrations that have gone on around the country, with many people masked.
There may be issues about what is acceptable to wear when you go to a demonstration. I don’t claim to know about the civil rights of that.
In a time at which COVID-19 is flourishing, really on the rebound, expanding fast, and still causing 600 deaths a week; the flu season is going to be upon us soon enough; and there are also concerns about the possibility of avian flu jumping into the human population, it is absolutely the wrong time to single out those who are trying to mask for health reasons.
Basically, there are two strong reasons. One, there are people out there who wear a medical mask or mask for a medical reason because they have an underlying disease. They may have had a transplant or they may feel they’re immunocompromised for some reason. They worry that, if they don’t wear a mask, they’re going to get an infection from something like COVID-19 or flu, which could really be super-dangerous for them.
The other reason people mask is to protect their family members. They may have someone who’s immunocompromised in the family, or they’re doing it kindly and altruistically to protect the rest of us and to stop viruses from circulating.
These bans are not taking into account public health. They’re being brought forward in the midst of political heat about demonstrations and political issues. I think they should be opposed. I do not think they should be enacted.
I think the medical rights of people with disabilities and immunologic disorders, and those who want to mask to prevent getting sick at a time at which infectious diseases are still circulating and killing people, ought to take priority. Public health, in this case, should drive our policies about masks.
Dr. Caplan, director, Division of Medical Ethics, New York University Langone Medical Center, New York, NY, served on Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position), and is a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
Six Tips on Coronavirus Testing for Doctors and Patients
according to the Robert Koch Institute, Germany. If a patient has a fever and cough and feels exhausted, it could be COVID-19. What significance do rapid tests have? And when should doctors advise their patients about them?
When to Test
People at a higher risk for severe COVID-19 benefit from tests. This population includes the following groups:
- Older patients
- Immunocompromised patients
- Patients with respiratory diseases
- Patients with cardiovascular diseases
- Patients with liver and kidney diseases
- Patients with neurological diseases
- Patients with obesity
If doctors detect SARS-CoV-2 infection early, they can prescribe Paxlovid, for example, to reduce morbidity and mortality risks. Conversely, people without specific risks should test themselves if they plan to visit vulnerable individuals.
Detecting New Variants
A comprehensive study from the fall of 2022 provides evidence that antigen tests targeting the nucleocapsid (N) protein of SARS-CoV-2 also detect new variants.
The researchers built a library of various versions of the SARS-CoV-2 N protein. Their collection included nearly 8000 individual amino acid substitutions, representing more than 99.5% of all statistically possible mutations of the N protein.
They then examined how these N proteins interacted with 17 antibodies used in 11 commercially available antigen rapid tests.
All antibodies were able to recognize altered N proteins. Since the researchers successfully investigated diagnostic antibodies against nearly all possible N-protein mutations, rapid tests should be able to detect future virus variants. However, sensitivity and specificity may still change.
Test Timing
Uncertainty about what time of day to test can be mitigated by performing multiple COVID-19 rapid tests over time. The Food and Drug Administration (FDA) and similar organizations make this recommendation. Studies of symptomatic individuals show that serial tests increase accuracy.
In the early stages of infection, swabs may contain too little virus material because of widespread immunity against SARS-CoV-2. That is, they may contain inadequate levels of the relevant antigen. Especially in asymptomatic individuals or patients in the incubation phase, a single test may therefore yield a false-negative result. Therefore, the FDA recommends conducting at least two additional tests 48 hours apart in case of a negative test result.
Costs of Rapid Tests
The days of free tests are long gone. In Germany, the distribution of free preventive coronavirus tests was discontinued on March 1, 2023.
Test kits are still available in pharmacies or drugstores. In packages with 5-10 tests, the individual test costs between €0.90 and €1.50, depending on the provider. If a patient still has old rapid coronavirus tests in his or her medicine cabinet, are they still suitable?
Expired Tests
Properly stored tests that have not passed their expiration dates can still be used. But microbiologist and pathologist Daniel Rhoads, MD, from the Cleveland Clinic in Ohio warns against expired rapid tests.
The chemicals may have decomposed, the solvent may have evaporated, or antibodies may have lost their effectiveness, thus making false negative results more likely. “These are proteins that can decompose over time,” said Dr. Rhoads.
Ordering PCR Tests
The polymerase chain reaction (PCR) test remains the gold standard for diagnosing COVID-19. It is still available within statutory health insurance coverage. As Germany’s National Association of Statutory Health Insurance Physicians observes, form Muster 10 is used to order the test in that country.
The fee for the swab is included in the insured patient’s basic flat rate. Laboratories bill the PCR test using fee schedule position (GOP) 32816, according to the Uniform Value Scale (EBM).
There is no possibility for billing rapid tests for SARS-CoV-2 in medical practices within the EBM. A laboratory-based SARS-CoV-2 antigen detection test (GOP 32779) can be requested via the Muster 10 form.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
according to the Robert Koch Institute, Germany. If a patient has a fever and cough and feels exhausted, it could be COVID-19. What significance do rapid tests have? And when should doctors advise their patients about them?
When to Test
People at a higher risk for severe COVID-19 benefit from tests. This population includes the following groups:
- Older patients
- Immunocompromised patients
- Patients with respiratory diseases
- Patients with cardiovascular diseases
- Patients with liver and kidney diseases
- Patients with neurological diseases
- Patients with obesity
If doctors detect SARS-CoV-2 infection early, they can prescribe Paxlovid, for example, to reduce morbidity and mortality risks. Conversely, people without specific risks should test themselves if they plan to visit vulnerable individuals.
Detecting New Variants
A comprehensive study from the fall of 2022 provides evidence that antigen tests targeting the nucleocapsid (N) protein of SARS-CoV-2 also detect new variants.
The researchers built a library of various versions of the SARS-CoV-2 N protein. Their collection included nearly 8000 individual amino acid substitutions, representing more than 99.5% of all statistically possible mutations of the N protein.
They then examined how these N proteins interacted with 17 antibodies used in 11 commercially available antigen rapid tests.
All antibodies were able to recognize altered N proteins. Since the researchers successfully investigated diagnostic antibodies against nearly all possible N-protein mutations, rapid tests should be able to detect future virus variants. However, sensitivity and specificity may still change.
Test Timing
Uncertainty about what time of day to test can be mitigated by performing multiple COVID-19 rapid tests over time. The Food and Drug Administration (FDA) and similar organizations make this recommendation. Studies of symptomatic individuals show that serial tests increase accuracy.
In the early stages of infection, swabs may contain too little virus material because of widespread immunity against SARS-CoV-2. That is, they may contain inadequate levels of the relevant antigen. Especially in asymptomatic individuals or patients in the incubation phase, a single test may therefore yield a false-negative result. Therefore, the FDA recommends conducting at least two additional tests 48 hours apart in case of a negative test result.
Costs of Rapid Tests
The days of free tests are long gone. In Germany, the distribution of free preventive coronavirus tests was discontinued on March 1, 2023.
Test kits are still available in pharmacies or drugstores. In packages with 5-10 tests, the individual test costs between €0.90 and €1.50, depending on the provider. If a patient still has old rapid coronavirus tests in his or her medicine cabinet, are they still suitable?
Expired Tests
Properly stored tests that have not passed their expiration dates can still be used. But microbiologist and pathologist Daniel Rhoads, MD, from the Cleveland Clinic in Ohio warns against expired rapid tests.
The chemicals may have decomposed, the solvent may have evaporated, or antibodies may have lost their effectiveness, thus making false negative results more likely. “These are proteins that can decompose over time,” said Dr. Rhoads.
Ordering PCR Tests
The polymerase chain reaction (PCR) test remains the gold standard for diagnosing COVID-19. It is still available within statutory health insurance coverage. As Germany’s National Association of Statutory Health Insurance Physicians observes, form Muster 10 is used to order the test in that country.
The fee for the swab is included in the insured patient’s basic flat rate. Laboratories bill the PCR test using fee schedule position (GOP) 32816, according to the Uniform Value Scale (EBM).
There is no possibility for billing rapid tests for SARS-CoV-2 in medical practices within the EBM. A laboratory-based SARS-CoV-2 antigen detection test (GOP 32779) can be requested via the Muster 10 form.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
according to the Robert Koch Institute, Germany. If a patient has a fever and cough and feels exhausted, it could be COVID-19. What significance do rapid tests have? And when should doctors advise their patients about them?
When to Test
People at a higher risk for severe COVID-19 benefit from tests. This population includes the following groups:
- Older patients
- Immunocompromised patients
- Patients with respiratory diseases
- Patients with cardiovascular diseases
- Patients with liver and kidney diseases
- Patients with neurological diseases
- Patients with obesity
If doctors detect SARS-CoV-2 infection early, they can prescribe Paxlovid, for example, to reduce morbidity and mortality risks. Conversely, people without specific risks should test themselves if they plan to visit vulnerable individuals.
Detecting New Variants
A comprehensive study from the fall of 2022 provides evidence that antigen tests targeting the nucleocapsid (N) protein of SARS-CoV-2 also detect new variants.
The researchers built a library of various versions of the SARS-CoV-2 N protein. Their collection included nearly 8000 individual amino acid substitutions, representing more than 99.5% of all statistically possible mutations of the N protein.
They then examined how these N proteins interacted with 17 antibodies used in 11 commercially available antigen rapid tests.
All antibodies were able to recognize altered N proteins. Since the researchers successfully investigated diagnostic antibodies against nearly all possible N-protein mutations, rapid tests should be able to detect future virus variants. However, sensitivity and specificity may still change.
Test Timing
Uncertainty about what time of day to test can be mitigated by performing multiple COVID-19 rapid tests over time. The Food and Drug Administration (FDA) and similar organizations make this recommendation. Studies of symptomatic individuals show that serial tests increase accuracy.
In the early stages of infection, swabs may contain too little virus material because of widespread immunity against SARS-CoV-2. That is, they may contain inadequate levels of the relevant antigen. Especially in asymptomatic individuals or patients in the incubation phase, a single test may therefore yield a false-negative result. Therefore, the FDA recommends conducting at least two additional tests 48 hours apart in case of a negative test result.
Costs of Rapid Tests
The days of free tests are long gone. In Germany, the distribution of free preventive coronavirus tests was discontinued on March 1, 2023.
Test kits are still available in pharmacies or drugstores. In packages with 5-10 tests, the individual test costs between €0.90 and €1.50, depending on the provider. If a patient still has old rapid coronavirus tests in his or her medicine cabinet, are they still suitable?
Expired Tests
Properly stored tests that have not passed their expiration dates can still be used. But microbiologist and pathologist Daniel Rhoads, MD, from the Cleveland Clinic in Ohio warns against expired rapid tests.
The chemicals may have decomposed, the solvent may have evaporated, or antibodies may have lost their effectiveness, thus making false negative results more likely. “These are proteins that can decompose over time,” said Dr. Rhoads.
Ordering PCR Tests
The polymerase chain reaction (PCR) test remains the gold standard for diagnosing COVID-19. It is still available within statutory health insurance coverage. As Germany’s National Association of Statutory Health Insurance Physicians observes, form Muster 10 is used to order the test in that country.
The fee for the swab is included in the insured patient’s basic flat rate. Laboratories bill the PCR test using fee schedule position (GOP) 32816, according to the Uniform Value Scale (EBM).
There is no possibility for billing rapid tests for SARS-CoV-2 in medical practices within the EBM. A laboratory-based SARS-CoV-2 antigen detection test (GOP 32779) can be requested via the Muster 10 form.
This story was translated from the Medscape German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
COVID Levels Start to Dip, New Variant Emerges
A new COVID-19 variant called XEC is on the rise, and it has experts who track variants on alert.
Each time a new variant makes a grand entrance onto tracker lists, health officials take notice because it may mean there’s an important change in behavior of SARS-CoV-2, the virus that causes COVID.
Countries reporting rising detections of XEC include Germany, the United Kingdom, and the Netherlands, Australian data scientist Mike Honey posted on the platform X this past week.
XEC’s “characteristic mutations” have been detected in at least 25 states, CBS News reported, with New Jersey, California, and Virginia labs reporting 10 or more cases each. New Jersey detections at least in part stem from the CDC’s testing program for international travelers at Newark Liberty International Airport.
Still, XEC hasn’t gained enough traction in Europe, the United States, or any other part of the world for it to be listed as a standalone variant on official watchlists maintained by the CDC, European Union, or World Health Organization.
However, Eric Topol, MD, executive vice president of Scripps Research and editor-at-large for Medscape, believes XEC is the next variant “to get legs.”
The rate at which a new variant takes the stage doesn’t always predict how severe it will be. Around this time last year, health officials sounded alarms about another Omicron variant called BA.2.86, dubbed Pirola, that ultimately didn’t make major waves.
“CDC is not aware of any specific symptoms associated with XEC or any other co-circulating SARS-CoV-2 lineage,” a CDC spokesperson said in a statement to CBS News.
Its parent lineages are KP.2 and KP.3, and all of these belong to the Omicron family. The SARS-CoV-2 virus mutates over time, and scientists use the names and labels to identify groups of viral variants based on their similarities and on which strains a mutated descendant came from.
A version of this article appeared on WebMD.com.
A new COVID-19 variant called XEC is on the rise, and it has experts who track variants on alert.
Each time a new variant makes a grand entrance onto tracker lists, health officials take notice because it may mean there’s an important change in behavior of SARS-CoV-2, the virus that causes COVID.
Countries reporting rising detections of XEC include Germany, the United Kingdom, and the Netherlands, Australian data scientist Mike Honey posted on the platform X this past week.
XEC’s “characteristic mutations” have been detected in at least 25 states, CBS News reported, with New Jersey, California, and Virginia labs reporting 10 or more cases each. New Jersey detections at least in part stem from the CDC’s testing program for international travelers at Newark Liberty International Airport.
Still, XEC hasn’t gained enough traction in Europe, the United States, or any other part of the world for it to be listed as a standalone variant on official watchlists maintained by the CDC, European Union, or World Health Organization.
However, Eric Topol, MD, executive vice president of Scripps Research and editor-at-large for Medscape, believes XEC is the next variant “to get legs.”
The rate at which a new variant takes the stage doesn’t always predict how severe it will be. Around this time last year, health officials sounded alarms about another Omicron variant called BA.2.86, dubbed Pirola, that ultimately didn’t make major waves.
“CDC is not aware of any specific symptoms associated with XEC or any other co-circulating SARS-CoV-2 lineage,” a CDC spokesperson said in a statement to CBS News.
Its parent lineages are KP.2 and KP.3, and all of these belong to the Omicron family. The SARS-CoV-2 virus mutates over time, and scientists use the names and labels to identify groups of viral variants based on their similarities and on which strains a mutated descendant came from.
A version of this article appeared on WebMD.com.
A new COVID-19 variant called XEC is on the rise, and it has experts who track variants on alert.
Each time a new variant makes a grand entrance onto tracker lists, health officials take notice because it may mean there’s an important change in behavior of SARS-CoV-2, the virus that causes COVID.
Countries reporting rising detections of XEC include Germany, the United Kingdom, and the Netherlands, Australian data scientist Mike Honey posted on the platform X this past week.
XEC’s “characteristic mutations” have been detected in at least 25 states, CBS News reported, with New Jersey, California, and Virginia labs reporting 10 or more cases each. New Jersey detections at least in part stem from the CDC’s testing program for international travelers at Newark Liberty International Airport.
Still, XEC hasn’t gained enough traction in Europe, the United States, or any other part of the world for it to be listed as a standalone variant on official watchlists maintained by the CDC, European Union, or World Health Organization.
However, Eric Topol, MD, executive vice president of Scripps Research and editor-at-large for Medscape, believes XEC is the next variant “to get legs.”
The rate at which a new variant takes the stage doesn’t always predict how severe it will be. Around this time last year, health officials sounded alarms about another Omicron variant called BA.2.86, dubbed Pirola, that ultimately didn’t make major waves.
“CDC is not aware of any specific symptoms associated with XEC or any other co-circulating SARS-CoV-2 lineage,” a CDC spokesperson said in a statement to CBS News.
Its parent lineages are KP.2 and KP.3, and all of these belong to the Omicron family. The SARS-CoV-2 virus mutates over time, and scientists use the names and labels to identify groups of viral variants based on their similarities and on which strains a mutated descendant came from.
A version of this article appeared on WebMD.com.
Millennial Clinicians Face Pay Disparities by Specialty, Other Factors
Salaries for millennial physicians are slightly increasing, but clinicians still face pay disparities across location, practice type, and gender.
Medscape Medical News reviewed survey data from more than 1200 practicing doctors under age 40 across 29 specialties over a 4-month period starting in October 2023.
The average annual total compensation (including any bonuses) for young clinicians rose from $326,000 to $338,000, about 4%, between 2022 and 2023. Among millennials, primary care physicians saw a 5% increase. But a large pay gap exists between fields: Specialists under age 40 earned an average of $357,000 in 2023, compared with the average primary care clinician salary of $271,000.
“Procedures are reimbursed too high, while very little value is placed on primary care,” one survey respondent complained.
The type of practice plays a major part in compensation. Millennial doctors in office-based, single-specialty group practices earned an average of $358,000 per year, followed by those in office-based multispecialty group practices at 355,000 per year. Those in outpatient clinics earned $278,000 per year.
“I believe the practice situation is a huge portion of compensation,” said Tiffany Di Pietro, DO, a cardiologist and internal medicine physician in Fort Lauderdale, Florida. “Owning your own private practice is generally more lucrative (if you have good business sense), but it is also quite a bit more time-consuming, whereas employed physicians usually make less but have fewer concerns with staffing and overhead.”
Like in previous years, a gender pay gap equated to men outearning women. Female physicians under age 40 of any kind earned about $302,000 per year, 24% less than their male counterparts, on average.
Millennial doctors in the Midwest brought home the biggest earnings, with an average salary of $343,000 vs $332,000 on the West Coast.
Millennial physicians also reported higher levels of dissatisfaction. In the 2022 report, 46% said they were not paid fairly. That figure rose to 49%. Just 68% of millennial doctors would choose medicine again if they could do things over, down from 76% in the 2021 report.
“Doctors go through multiple years of school and then have to act like we are working at Dunkin’ Donuts — like we’re on an assembly line,” one survey respondent said. “We should not have to be paid per patient seen but valued for 8-9 years of training.”
Despite these complaints, close to 7 out of 10 millennial respondents said pay was not a major factor in what area of medicine they chose, with 29% saying it played no role at all in their decision.
Psychiatrists and anesthesiologists were the happiest with their earnings, with 61% of both specialties reporting that they felt fairly paid. They were followed by dermatologists and emergency medicine doctors, both of whom 60% reported fair earnings.
Many millennial doctors are finding ways to make money outside of their practice, with 18% securing other medical-related work, 15% doing medical moonlighting, and 5% taking on non–medical-related work.
A version of this article first appeared on Medscape.com.
Salaries for millennial physicians are slightly increasing, but clinicians still face pay disparities across location, practice type, and gender.
Medscape Medical News reviewed survey data from more than 1200 practicing doctors under age 40 across 29 specialties over a 4-month period starting in October 2023.
The average annual total compensation (including any bonuses) for young clinicians rose from $326,000 to $338,000, about 4%, between 2022 and 2023. Among millennials, primary care physicians saw a 5% increase. But a large pay gap exists between fields: Specialists under age 40 earned an average of $357,000 in 2023, compared with the average primary care clinician salary of $271,000.
“Procedures are reimbursed too high, while very little value is placed on primary care,” one survey respondent complained.
The type of practice plays a major part in compensation. Millennial doctors in office-based, single-specialty group practices earned an average of $358,000 per year, followed by those in office-based multispecialty group practices at 355,000 per year. Those in outpatient clinics earned $278,000 per year.
“I believe the practice situation is a huge portion of compensation,” said Tiffany Di Pietro, DO, a cardiologist and internal medicine physician in Fort Lauderdale, Florida. “Owning your own private practice is generally more lucrative (if you have good business sense), but it is also quite a bit more time-consuming, whereas employed physicians usually make less but have fewer concerns with staffing and overhead.”
Like in previous years, a gender pay gap equated to men outearning women. Female physicians under age 40 of any kind earned about $302,000 per year, 24% less than their male counterparts, on average.
Millennial doctors in the Midwest brought home the biggest earnings, with an average salary of $343,000 vs $332,000 on the West Coast.
Millennial physicians also reported higher levels of dissatisfaction. In the 2022 report, 46% said they were not paid fairly. That figure rose to 49%. Just 68% of millennial doctors would choose medicine again if they could do things over, down from 76% in the 2021 report.
“Doctors go through multiple years of school and then have to act like we are working at Dunkin’ Donuts — like we’re on an assembly line,” one survey respondent said. “We should not have to be paid per patient seen but valued for 8-9 years of training.”
Despite these complaints, close to 7 out of 10 millennial respondents said pay was not a major factor in what area of medicine they chose, with 29% saying it played no role at all in their decision.
Psychiatrists and anesthesiologists were the happiest with their earnings, with 61% of both specialties reporting that they felt fairly paid. They were followed by dermatologists and emergency medicine doctors, both of whom 60% reported fair earnings.
Many millennial doctors are finding ways to make money outside of their practice, with 18% securing other medical-related work, 15% doing medical moonlighting, and 5% taking on non–medical-related work.
A version of this article first appeared on Medscape.com.
Salaries for millennial physicians are slightly increasing, but clinicians still face pay disparities across location, practice type, and gender.
Medscape Medical News reviewed survey data from more than 1200 practicing doctors under age 40 across 29 specialties over a 4-month period starting in October 2023.
The average annual total compensation (including any bonuses) for young clinicians rose from $326,000 to $338,000, about 4%, between 2022 and 2023. Among millennials, primary care physicians saw a 5% increase. But a large pay gap exists between fields: Specialists under age 40 earned an average of $357,000 in 2023, compared with the average primary care clinician salary of $271,000.
“Procedures are reimbursed too high, while very little value is placed on primary care,” one survey respondent complained.
The type of practice plays a major part in compensation. Millennial doctors in office-based, single-specialty group practices earned an average of $358,000 per year, followed by those in office-based multispecialty group practices at 355,000 per year. Those in outpatient clinics earned $278,000 per year.
“I believe the practice situation is a huge portion of compensation,” said Tiffany Di Pietro, DO, a cardiologist and internal medicine physician in Fort Lauderdale, Florida. “Owning your own private practice is generally more lucrative (if you have good business sense), but it is also quite a bit more time-consuming, whereas employed physicians usually make less but have fewer concerns with staffing and overhead.”
Like in previous years, a gender pay gap equated to men outearning women. Female physicians under age 40 of any kind earned about $302,000 per year, 24% less than their male counterparts, on average.
Millennial doctors in the Midwest brought home the biggest earnings, with an average salary of $343,000 vs $332,000 on the West Coast.
Millennial physicians also reported higher levels of dissatisfaction. In the 2022 report, 46% said they were not paid fairly. That figure rose to 49%. Just 68% of millennial doctors would choose medicine again if they could do things over, down from 76% in the 2021 report.
“Doctors go through multiple years of school and then have to act like we are working at Dunkin’ Donuts — like we’re on an assembly line,” one survey respondent said. “We should not have to be paid per patient seen but valued for 8-9 years of training.”
Despite these complaints, close to 7 out of 10 millennial respondents said pay was not a major factor in what area of medicine they chose, with 29% saying it played no role at all in their decision.
Psychiatrists and anesthesiologists were the happiest with their earnings, with 61% of both specialties reporting that they felt fairly paid. They were followed by dermatologists and emergency medicine doctors, both of whom 60% reported fair earnings.
Many millennial doctors are finding ways to make money outside of their practice, with 18% securing other medical-related work, 15% doing medical moonlighting, and 5% taking on non–medical-related work.
A version of this article first appeared on Medscape.com.
How Experts Predicts This COVID and Flu Season Will Unfold
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
What’s the outlook for COVID-19 and flu this fall and winter? It’ll probably be a lot like last year, experts say.
“We currently expect this flu season to be comparable to last year’s season,” said Adrienne Keen, PhD, of the Centers for Disease Control and Prevention’s (CDC) Center for Forecasting and Outbreak Analytics. “We expect this year’s COVID-19 season peak to be similar to last year’s or lower.” The CDC is still analyzing COVID surveillance data from the summer and will update the forecast as more is learned.
For COVID, that means it won’t be as bad as the pandemic years, and for the flu, it’s a typical pre-pandemic season. But status quo does not mean great.
Between October 2023 and April 2024, as many as 75 million people got the flu in the United States, according to CDC estimates, resulting in up to 900,000 hospitalizations and between 17,000 and 100,000 deaths. In 2023, about 900,000 Americans were hospitalized with COVID and 75,000 died.
Getting vaccinated remains crucial, public health officials stressed.
Predicting COVID
Two key predictors of how bad an upcoming COVID season will be are the cycling of new variants and the population’s immunity (protection from an infectious disease that happens when a population is immune through vaccination or previous infection).
When new variants go up and immunity goes down, “we tend to see the increase in cases,” said Michael T. Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy and a professor of public health at the University of Minnesota, Minneapolis. But if the number of variants goes down and immunity levels go up, the outlook is more favorable.
The new COVID variant called XEC has been found in at least 25 states. On September 27, the CDC added the variant to the COVID tracker. It now accounts for 6% of US cases. This was expected, as the variant has been circulating in Europe, said Amesh Adalja, MD, a senior scholar and infectious disease expert at the Center for Health Security at Johns Hopkins University, Baltimore, Maryland.
“There will always be a new variant appearing, and one falling,” he said. “So the fact that this is happening is not surprising.”
Meanwhile, the summer COVID surge has provided postinfection immunity for some people. “What’s likely is, we are going to see substantial protection of the population for several months based on previous infection and in some cases vaccination,” Dr. Osterholm said. That means protection from serious illness, hospitalizations, and deaths (but not necessarily infection). That protection could last through the year or into early 2025.
The timing of 2024’s winter surge will likely be a bit later than 2023’s, said Andrew Pekosz, PhD, a professor and vice chair of molecular microbiology and immunology at Johns Hopkins University, Baltimore, “peaking just after the Christmas/New Year holiday.”
During the 2023-2024 season, weekly COVID hospitalizations peaked the week of Dec. 30, said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill and a member of the COVID-19 Scenario Modeling Hub.
But variants are unpredictable. “There’s a chance that the XEC variant may take off and spread, or might not,” said Dr. Adalja. As of September 28, the Omicron variant KP.3.1.1 was leading, accounting for 58.7% of US cases, according to the CDC.
While Dr. Adalja agreed that 2024’s COVID season will probably be like 2023’s, “we have to be prepared for cases and hospitalizations going up,” he said, “but not to the point of a crisis.” A return to lockdowns and social distancing is unlikely.
Still, older adults and others at higher risk of getting very sick from COVID should consider masking during travel, said Rajendram Rajnarayanan, PhD, MSc, an associate professor at the New York Institute of Technology College of Osteopathic Medicine at Arkansas State University, Jonesboro.
Flu Forecasts
Predicting flu season this early is hard, said Jeffrey Shaman, PhD, a professor of environmental health sciences and professor of climate at Colombia University, New York.
“You can look at the CDC forecast and use it as a very loose guide right now,” said Dr. Shaman, who won the CDC’s first “Predict the Influenza Season Challenge” in 2014. “Until there is actually flu, it’s like trying to predict the landfall of a hurricane.” Flu activity remained low as of September 14 (the most current data available), according to the CDC.
When flu activity picks up, typically in mid-October or November, experts look at the dominant strain, exposure to similar strains in previous years, and how well-matched the current flu vaccine is to that dominant strain, Dr. Shaman said. Vaccine makers must make an educated guess months in advance regarding which strain to target, to allow time for production.
The vaccination rate plays a role, too, but that tends to remain constant, Dr. Shaman said. According to the CDC, less than half of adults age 18 and up got a flu vaccination last year.
Experts also consider flu patterns in the Southern Hemisphere, where 2024 flu activity has mostly involved two subtypes of influenza A — H1N1 and H3N2 — and some influenza B, the CDC found.
How Well Do This Year’s Vaccines and Viruses Match Up?
The FDA has authorized three updated COVID vaccines for this fall. Novavax targets the JN.1 strain of SARS-CoV-2, the virus that causes COVID-19. Both mRNA vaccines, Moderna and Pfizer, target KP.2, a descendant of JN.1. All three target current predominant variants, and any one of them is recommended by the CDC.
The vaccines are a good “though not perfect match to virtually all the circulating variants of SARS-CoV-2,” said Dr. Pekosz.
Experts said that the shots will protect against the XEC variant.
“XEC and its sublineages are expected to be the dominant fall/winter variant group,” said Dr. Rajnarayanan.
This year’s flu vaccines, all trivalent (protecting against three viruses), will target the three strains expected to circulate — H1N1, H3N2, and influenza B (Victoria), according to the CDC.
People should still get vaccinated, Dr. Adalja said, and use home tests for flu and COVID and take antivirals promptly when needed. The goal should not be status quo but rather fewer COVID and flu hospitalizations and deaths.
A version of this article first appeared on WebMD.com.
A Few Rural Towns Are Bucking the Trend and Building New Hospitals
There’s a new morning ritual in Pinedale, Wyoming, a town of about 2000, nestled against the Wind River Mountains.
Friends and neighbors in the oil- and gas-rich community “take their morning coffee and pull up” to watch workers building the county’s first hospital, said Kari DeWitt, the project’s public relations director.
“I think it’s just gratitude,” Ms. DeWitt said.
Sublette County is the only one in Wyoming — where counties span thousands of square miles — without a hospital. The 10-bed, 40,000-square-foot hospital, with a similarly sized attached long-term care facility, is slated to open by the summer of 2025.
Ms. DeWitt, who also is executive director of the Sublette County Health Foundation, has an office at the town’s health clinic with a window view of the construction.
Pinedale’s residents have good reason to be excited. New full-service hospitals with inpatient beds are rare in rural America, where declining population has spurred decades of downsizing and closures. Yet, a few communities in Wyoming and others in Kansas and Georgia are defying the trend.
“To be honest with you, it even seems strange to me,” said Wyoming Hospital Association President Eric Boley. Small rural “hospitals are really struggling all across the country,” he said.
There is no official tally of new hospitals being built in rural America, but industry experts such as Mr. Boley said they’re rare. Typically, health-related construction projects in rural areas are for smaller urgent care centers or stand-alone emergency facilities or are replacements for old hospitals.
About half of rural hospitals lost money in the prior year, according to Chartis, a health analytics and consulting firm. And nearly 150 rural hospitals have closed or converted to smaller operations since 2010, according to data collected by the University of North Carolina’s Cecil G. Sheps Center for Health Services Research.
To stem the tide of closures, Congress created a new rural emergency hospital designation that allowed struggling hospitals to close their inpatient units and provide only outpatient and emergency services. Since January 2023, when the program took effect, 32 of the more than 1700 eligible rural hospitals — from Georgia to New Mexico — have joined the program, according to data from the Centers for Medicare & Medicaid Services.
Tony Breitlow is healthcare studio director for EUA, which has extensive experience working for rural health care systems. Mr. Breitlow said his national architecture and engineering firm’s work expands, replaces, or revamps older buildings, many of which were constructed during the middle of the last century.
The work, Mr. Breitlow said, is part of health care “systems figuring out how to remain robust and viable.”
Freeman Health System, based in Joplin, Missouri, announced plans last year to build a new 50-bed hospital across the state line in Kansas. Paula Baker, Freeman’s president and chief executive, said the system is building for patients in the southeastern corner of the state who travel 45 minutes or more to its bigger Joplin facilities for care.
Freeman’s new hospital, with construction on the building expected to begin in the spring, will be less than 10 miles away from an older, 64-bed hospital that has existed for decades. Kansas is one of more than a dozen states with no “certificate of need” law that would require health providers to obtain approval from the state before offering new services or building or expanding facilities.
Ms. Baker also said Freeman plans to operate emergency services and a small 10-bed outpost in Fort Scott, Kansas, opening early next year in a corner of a hospital that closed in late 2018. Residents there “cried, they cheered, they hugged me,” Ms. Baker said, adding that the “level of appreciation and gratitude that they felt and they displayed was overwhelming to me.”
Michael Topchik, executive director of the Chartis Center for Rural Health, said regional healthcare systems in the Upper Midwest have been particularly active in competing for patients by, among other things, building new hospitals.
And while private corporate money can drive construction, many rural hospital projects tap government programs, especially those supported by the US Department of Agriculture, Mr. Topchik said. That, he said, “surprises a lot of people.”
Since 2021, the USDA’s rural Community Facilities Programs have awarded $2.24 billion in loans and grants to 68 rural hospitals for work that was not related to an emergency or disaster, according to data analyzed by KFF Health News and confirmed by the agency. The federal program is funded through what is often known as the farm bill, which faces a September congressional renewal deadline.
Nearly all the projects are replacements or expansions and updates of older facilities.
The USDA confirmed that three new or planned Wyoming hospitals received federal funding. Hospital projects in Riverton and Saratoga received loans of $37.2 million and $18.3 million, respectively. Pinedale’s hospital received a $29.2 million loan from the agency.
Wyoming’s new construction is rare in a state where more than 80% of rural hospitals reported losses in the third quarter of 2023, according to Chartis. The state association’s Mr. Boley said he worries about several hospitals that have less than 10 days’ cash on hand “day and night.”
Pinedale’s project loan was approved after the community submitted a feasibility study to the USDA that included local clinics and a long-term care facility. “It’s pretty remote and right up in the mountains,” Mr. Boley said.
Pinedale’s Ms. DeWitt said the community was missing key services, such as blood transfusions, which are often necessary when there is a trauma like a car crash or if a pregnant woman faces severe complications. Local ambulances drove 94,000 miles last year, she said.
Ms. DeWitt began working to raise support for the new hospital after her own pregnancy-related trauma in 2014. She was bleeding heavily and arrived at the local health clinic believing it operated like a hospital.
“It was shocking to hear, ‘No, we’re not a hospital. We can’t do blood transfusions. We’re just going to have to pray you live for the next 45 minutes,’ ” Ms. DeWitt said.
Ms. DeWitt had to be airlifted to Idaho, where she delivered a few minutes after landing. When the hospital financing went on the ballot in 2020, Ms. DeWitt — fully recovered, with healthy grade-schoolers at home — began making five calls a night to rally support for a county tax increase to help fund the hospital.
“By improving health care, I think we improve everybody’s chances of survival. You know, it’s pretty basic,” Ms. DeWitt said.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
There’s a new morning ritual in Pinedale, Wyoming, a town of about 2000, nestled against the Wind River Mountains.
Friends and neighbors in the oil- and gas-rich community “take their morning coffee and pull up” to watch workers building the county’s first hospital, said Kari DeWitt, the project’s public relations director.
“I think it’s just gratitude,” Ms. DeWitt said.
Sublette County is the only one in Wyoming — where counties span thousands of square miles — without a hospital. The 10-bed, 40,000-square-foot hospital, with a similarly sized attached long-term care facility, is slated to open by the summer of 2025.
Ms. DeWitt, who also is executive director of the Sublette County Health Foundation, has an office at the town’s health clinic with a window view of the construction.
Pinedale’s residents have good reason to be excited. New full-service hospitals with inpatient beds are rare in rural America, where declining population has spurred decades of downsizing and closures. Yet, a few communities in Wyoming and others in Kansas and Georgia are defying the trend.
“To be honest with you, it even seems strange to me,” said Wyoming Hospital Association President Eric Boley. Small rural “hospitals are really struggling all across the country,” he said.
There is no official tally of new hospitals being built in rural America, but industry experts such as Mr. Boley said they’re rare. Typically, health-related construction projects in rural areas are for smaller urgent care centers or stand-alone emergency facilities or are replacements for old hospitals.
About half of rural hospitals lost money in the prior year, according to Chartis, a health analytics and consulting firm. And nearly 150 rural hospitals have closed or converted to smaller operations since 2010, according to data collected by the University of North Carolina’s Cecil G. Sheps Center for Health Services Research.
To stem the tide of closures, Congress created a new rural emergency hospital designation that allowed struggling hospitals to close their inpatient units and provide only outpatient and emergency services. Since January 2023, when the program took effect, 32 of the more than 1700 eligible rural hospitals — from Georgia to New Mexico — have joined the program, according to data from the Centers for Medicare & Medicaid Services.
Tony Breitlow is healthcare studio director for EUA, which has extensive experience working for rural health care systems. Mr. Breitlow said his national architecture and engineering firm’s work expands, replaces, or revamps older buildings, many of which were constructed during the middle of the last century.
The work, Mr. Breitlow said, is part of health care “systems figuring out how to remain robust and viable.”
Freeman Health System, based in Joplin, Missouri, announced plans last year to build a new 50-bed hospital across the state line in Kansas. Paula Baker, Freeman’s president and chief executive, said the system is building for patients in the southeastern corner of the state who travel 45 minutes or more to its bigger Joplin facilities for care.
Freeman’s new hospital, with construction on the building expected to begin in the spring, will be less than 10 miles away from an older, 64-bed hospital that has existed for decades. Kansas is one of more than a dozen states with no “certificate of need” law that would require health providers to obtain approval from the state before offering new services or building or expanding facilities.
Ms. Baker also said Freeman plans to operate emergency services and a small 10-bed outpost in Fort Scott, Kansas, opening early next year in a corner of a hospital that closed in late 2018. Residents there “cried, they cheered, they hugged me,” Ms. Baker said, adding that the “level of appreciation and gratitude that they felt and they displayed was overwhelming to me.”
Michael Topchik, executive director of the Chartis Center for Rural Health, said regional healthcare systems in the Upper Midwest have been particularly active in competing for patients by, among other things, building new hospitals.
And while private corporate money can drive construction, many rural hospital projects tap government programs, especially those supported by the US Department of Agriculture, Mr. Topchik said. That, he said, “surprises a lot of people.”
Since 2021, the USDA’s rural Community Facilities Programs have awarded $2.24 billion in loans and grants to 68 rural hospitals for work that was not related to an emergency or disaster, according to data analyzed by KFF Health News and confirmed by the agency. The federal program is funded through what is often known as the farm bill, which faces a September congressional renewal deadline.
Nearly all the projects are replacements or expansions and updates of older facilities.
The USDA confirmed that three new or planned Wyoming hospitals received federal funding. Hospital projects in Riverton and Saratoga received loans of $37.2 million and $18.3 million, respectively. Pinedale’s hospital received a $29.2 million loan from the agency.
Wyoming’s new construction is rare in a state where more than 80% of rural hospitals reported losses in the third quarter of 2023, according to Chartis. The state association’s Mr. Boley said he worries about several hospitals that have less than 10 days’ cash on hand “day and night.”
Pinedale’s project loan was approved after the community submitted a feasibility study to the USDA that included local clinics and a long-term care facility. “It’s pretty remote and right up in the mountains,” Mr. Boley said.
Pinedale’s Ms. DeWitt said the community was missing key services, such as blood transfusions, which are often necessary when there is a trauma like a car crash or if a pregnant woman faces severe complications. Local ambulances drove 94,000 miles last year, she said.
Ms. DeWitt began working to raise support for the new hospital after her own pregnancy-related trauma in 2014. She was bleeding heavily and arrived at the local health clinic believing it operated like a hospital.
“It was shocking to hear, ‘No, we’re not a hospital. We can’t do blood transfusions. We’re just going to have to pray you live for the next 45 minutes,’ ” Ms. DeWitt said.
Ms. DeWitt had to be airlifted to Idaho, where she delivered a few minutes after landing. When the hospital financing went on the ballot in 2020, Ms. DeWitt — fully recovered, with healthy grade-schoolers at home — began making five calls a night to rally support for a county tax increase to help fund the hospital.
“By improving health care, I think we improve everybody’s chances of survival. You know, it’s pretty basic,” Ms. DeWitt said.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
There’s a new morning ritual in Pinedale, Wyoming, a town of about 2000, nestled against the Wind River Mountains.
Friends and neighbors in the oil- and gas-rich community “take their morning coffee and pull up” to watch workers building the county’s first hospital, said Kari DeWitt, the project’s public relations director.
“I think it’s just gratitude,” Ms. DeWitt said.
Sublette County is the only one in Wyoming — where counties span thousands of square miles — without a hospital. The 10-bed, 40,000-square-foot hospital, with a similarly sized attached long-term care facility, is slated to open by the summer of 2025.
Ms. DeWitt, who also is executive director of the Sublette County Health Foundation, has an office at the town’s health clinic with a window view of the construction.
Pinedale’s residents have good reason to be excited. New full-service hospitals with inpatient beds are rare in rural America, where declining population has spurred decades of downsizing and closures. Yet, a few communities in Wyoming and others in Kansas and Georgia are defying the trend.
“To be honest with you, it even seems strange to me,” said Wyoming Hospital Association President Eric Boley. Small rural “hospitals are really struggling all across the country,” he said.
There is no official tally of new hospitals being built in rural America, but industry experts such as Mr. Boley said they’re rare. Typically, health-related construction projects in rural areas are for smaller urgent care centers or stand-alone emergency facilities or are replacements for old hospitals.
About half of rural hospitals lost money in the prior year, according to Chartis, a health analytics and consulting firm. And nearly 150 rural hospitals have closed or converted to smaller operations since 2010, according to data collected by the University of North Carolina’s Cecil G. Sheps Center for Health Services Research.
To stem the tide of closures, Congress created a new rural emergency hospital designation that allowed struggling hospitals to close their inpatient units and provide only outpatient and emergency services. Since January 2023, when the program took effect, 32 of the more than 1700 eligible rural hospitals — from Georgia to New Mexico — have joined the program, according to data from the Centers for Medicare & Medicaid Services.
Tony Breitlow is healthcare studio director for EUA, which has extensive experience working for rural health care systems. Mr. Breitlow said his national architecture and engineering firm’s work expands, replaces, or revamps older buildings, many of which were constructed during the middle of the last century.
The work, Mr. Breitlow said, is part of health care “systems figuring out how to remain robust and viable.”
Freeman Health System, based in Joplin, Missouri, announced plans last year to build a new 50-bed hospital across the state line in Kansas. Paula Baker, Freeman’s president and chief executive, said the system is building for patients in the southeastern corner of the state who travel 45 minutes or more to its bigger Joplin facilities for care.
Freeman’s new hospital, with construction on the building expected to begin in the spring, will be less than 10 miles away from an older, 64-bed hospital that has existed for decades. Kansas is one of more than a dozen states with no “certificate of need” law that would require health providers to obtain approval from the state before offering new services or building or expanding facilities.
Ms. Baker also said Freeman plans to operate emergency services and a small 10-bed outpost in Fort Scott, Kansas, opening early next year in a corner of a hospital that closed in late 2018. Residents there “cried, they cheered, they hugged me,” Ms. Baker said, adding that the “level of appreciation and gratitude that they felt and they displayed was overwhelming to me.”
Michael Topchik, executive director of the Chartis Center for Rural Health, said regional healthcare systems in the Upper Midwest have been particularly active in competing for patients by, among other things, building new hospitals.
And while private corporate money can drive construction, many rural hospital projects tap government programs, especially those supported by the US Department of Agriculture, Mr. Topchik said. That, he said, “surprises a lot of people.”
Since 2021, the USDA’s rural Community Facilities Programs have awarded $2.24 billion in loans and grants to 68 rural hospitals for work that was not related to an emergency or disaster, according to data analyzed by KFF Health News and confirmed by the agency. The federal program is funded through what is often known as the farm bill, which faces a September congressional renewal deadline.
Nearly all the projects are replacements or expansions and updates of older facilities.
The USDA confirmed that three new or planned Wyoming hospitals received federal funding. Hospital projects in Riverton and Saratoga received loans of $37.2 million and $18.3 million, respectively. Pinedale’s hospital received a $29.2 million loan from the agency.
Wyoming’s new construction is rare in a state where more than 80% of rural hospitals reported losses in the third quarter of 2023, according to Chartis. The state association’s Mr. Boley said he worries about several hospitals that have less than 10 days’ cash on hand “day and night.”
Pinedale’s project loan was approved after the community submitted a feasibility study to the USDA that included local clinics and a long-term care facility. “It’s pretty remote and right up in the mountains,” Mr. Boley said.
Pinedale’s Ms. DeWitt said the community was missing key services, such as blood transfusions, which are often necessary when there is a trauma like a car crash or if a pregnant woman faces severe complications. Local ambulances drove 94,000 miles last year, she said.
Ms. DeWitt began working to raise support for the new hospital after her own pregnancy-related trauma in 2014. She was bleeding heavily and arrived at the local health clinic believing it operated like a hospital.
“It was shocking to hear, ‘No, we’re not a hospital. We can’t do blood transfusions. We’re just going to have to pray you live for the next 45 minutes,’ ” Ms. DeWitt said.
Ms. DeWitt had to be airlifted to Idaho, where she delivered a few minutes after landing. When the hospital financing went on the ballot in 2020, Ms. DeWitt — fully recovered, with healthy grade-schoolers at home — began making five calls a night to rally support for a county tax increase to help fund the hospital.
“By improving health care, I think we improve everybody’s chances of survival. You know, it’s pretty basic,” Ms. DeWitt said.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
Guidance for Practicing Primary Care: World Health Organization’s Updated Influenza Guidelines for 2024
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.
As primary care physicians, we are often the first ones patients see when they become infected with influenza. According to Centers for Disease Control and Prevention statistics, approximately 5%-20% of the US population will be infected with influenza every year. Additionally, more than 200,000 of these patients will be hospitalized because of complications related to influenza.
Earlier in September, the World Health Organization (WHO) issued its latest clinical practice guidelines for influenza for the 2024-2025 season. This is a 213-page document aimed at healthcare providers who treat patients infected with influenza. It includes treatment for those with severe and nonsevere influenza infections, those in both the outpatient and hospitalized setting, as well as medication prophylaxis for those exposed to the virus. Additionally, it defines risk estimates for those who are at risk of being hospitalized or dying. In contrast, previous updates focused on management of severe influenza or those at risk of severe influenza.
These guidelines cover recommendations regarding all the antiviral medications for treating influenza used around the world. For the purpose of this article, we will focus on those most commonly used in the United States.
A newer medication discussed was baloxavir. It is recommended to be used for patients with nonsevere influenza who are at high risk for progression to severe disease. The advice is to not use it for those with little risk of progression to severe disease. Oseltamivir is recommended for those with severe infection.
The guidelines recommend against using antibiotics for those who have a low likelihood of having a bacterial coinfection. As primary care doctors, we often prescribe medications to help with symptoms. These guidelines recommend against the use of corticosteroids and antibiotics but did advise that NSAIDs could be used for symptom relief.
One of the important parts of these guidelines is prevention in patients who have been exposed but are asymptomatic. They recommend baloxavir or oseltamivir but only for those patients who are at high risk of being hospitalized if they were to become infected. Any of the antivirals can be used for patients who are exposed to the novel influenza A, which is associated with a higher mortality rate. Caution when prescribing antivirals is recommended in immunocompromised patients because there is more drug resistance seen in these patients.
These updates also discuss the use of different influenza tests. In the outpatient setting, primary doctors don’t have time for test results that may take 2 days to come back. Only rapid tests make the sense in the primary care setting. Additionally, in the age of COVID, it is important to make an accurate diagnosis so we should be testing patients. There is resistance seen with the antivirals we prescribe for influenza so prescribing them empirically without a confirmed diagnosis of influenza may be doing more harm than good.
One gap in these recommendations is vaccination. This topic was not covered at all. It would be helpful to have a strategy in place to prevent infection in populations rather than focusing just on exposed individuals. A discussion of when and who and to vaccinate would be helpful. Research into the effectiveness of vaccines is key and more accurate development of a season’s influenza vaccine would be beneficial. Currently, there is much vaccine misinformation being spread around. Education and information regarding influenza vaccines, especially coming from WHO, is crucial.
Another failure of these recommendations is that the guidelines apply only to those who present within a few days of becoming symptomatic. As family doctors, we know many of our patients self-treat or consult Google. They often don’t come for medical care until they’ve been sick for a week or longer. There are no guidelines for these patients.
In general, these guidelines are comprehensive and do a great job discussing the current medications available. However, more is needed to increase vaccination rates. Patients need to know that if they may be sick with influenza, they need to seek medical care as soon as possible. We, as family doctors, need to do a better job of risk-stratifying our patients and prescribing prophylactic medication when suitable. Every infection we prevent aids in the health of our community and the global population at large.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no relevant conflicts of interest.


