User login
Study eyes impact of isotretinoin on triglycerides, other lab measures
.
“Isotretinoin is a very effective treatment for severe acne,” Varsha Parthasarathy said at the annual meeting of the Society for Pediatric Dermatology. “However, initiating this medication requires a complex process of laboratory testing,” which includes human chorionic gonadotropin pregnancy testing, because isotretinoin is a teratogen, as well as lipid labs and liver function tests, she noted. “Importantly, triglycerides are measured due to an association in adults between isotretinoin and hypertriglyceridemia-associated pancreatitis. However, these findings in children are limited to case reports, as are findings of retinoid-induced hepatotoxicity.”
To identify the role of isotretinoin on changes in lipids, aspartate aminotransferase (AST), and alanine aminotransferase (ALT), and to determine the impact on treatment course, Ms. Parthasarathy, a 4-year medical student at George Washington University, Washington, and colleagues retrospectively reviewed the charts of 130 patients aged 12-21 years who were cared for at Children’s National Hospital between January 2012 and October 2020. Nearly two-thirds (65%) were male, their average age was 16 years, and the mean time to obtain follow-up labs after starting isotretinoin was 3.25 months.
Between baseline and follow-up, the researchers observed increases in total cholesterol, triglycerides, and LDL (P less than .001 for all associations) and a decrease in HDL (P = .001), but there were no significant changes in AST or ALT levels. These findings were consistent with prior studies in adults examining the utility of these laboratory tests, most notably a 2016 study by Timothy J. Hansen, MD, and colleagues.
Among the 13 patients with elevated triglycerides at baseline, 9 (69%) were overweight or obese. Of the 20 patients with elevated triglycerides at follow-up, 11 patients (55%) were obese. At follow-up, 11 patients had levels of 200-500 mg/dL (grade I elevation), and 2 patients had levels of 501-1,000 mg/dL (grade II elevation). Isotretinoin was stopped in the latter two patients, who also had obesity as a risk factor for their hypertriglyceridemia.
“None of these patients had clinical sequelae from their hypertriglyceridemia, such as pancreatitis at baseline or follow-up,” Ms. Parthasarathy said. “However, since pancreatitis would be expected to be exceedingly rare, the sample size may be limited in identifying this adverse effect.”
She noted that while isotretinoin might cause a significant increase in lipid levels, the mean levels remained within normal limits at both baseline and follow-up. “Of the patients with elevated triglycerides at baseline and follow-up, obesity may have been a potential risk factor,” she said. “This could suggest a possible strategy for reduced testing in nonobese isotretinoin patients, which can be further explored in larger study populations.”
In addition, “there was a lack of significant change in AST and ALT in this study and adult studies, as well as minimal evidence for pediatric retinoid-induced hepatotoxicity, which raises the question of the necessity of baseline and follow-up comprehensive metabolic panel testing,” Ms. Parthasarathy added. “Clinicians must weigh the laboratory values with the costs of laboratory testing, including opportunity costs such as time, monetary costs, and the discomfort of testing for pediatric patients.”
The study’s senior author was A. Yasmine Kirkorian, MD, chief of dermatology at Children’s National Hospital, Washington. The researchers reported having no relevant financial disclosures.
.
“Isotretinoin is a very effective treatment for severe acne,” Varsha Parthasarathy said at the annual meeting of the Society for Pediatric Dermatology. “However, initiating this medication requires a complex process of laboratory testing,” which includes human chorionic gonadotropin pregnancy testing, because isotretinoin is a teratogen, as well as lipid labs and liver function tests, she noted. “Importantly, triglycerides are measured due to an association in adults between isotretinoin and hypertriglyceridemia-associated pancreatitis. However, these findings in children are limited to case reports, as are findings of retinoid-induced hepatotoxicity.”
To identify the role of isotretinoin on changes in lipids, aspartate aminotransferase (AST), and alanine aminotransferase (ALT), and to determine the impact on treatment course, Ms. Parthasarathy, a 4-year medical student at George Washington University, Washington, and colleagues retrospectively reviewed the charts of 130 patients aged 12-21 years who were cared for at Children’s National Hospital between January 2012 and October 2020. Nearly two-thirds (65%) were male, their average age was 16 years, and the mean time to obtain follow-up labs after starting isotretinoin was 3.25 months.
Between baseline and follow-up, the researchers observed increases in total cholesterol, triglycerides, and LDL (P less than .001 for all associations) and a decrease in HDL (P = .001), but there were no significant changes in AST or ALT levels. These findings were consistent with prior studies in adults examining the utility of these laboratory tests, most notably a 2016 study by Timothy J. Hansen, MD, and colleagues.
Among the 13 patients with elevated triglycerides at baseline, 9 (69%) were overweight or obese. Of the 20 patients with elevated triglycerides at follow-up, 11 patients (55%) were obese. At follow-up, 11 patients had levels of 200-500 mg/dL (grade I elevation), and 2 patients had levels of 501-1,000 mg/dL (grade II elevation). Isotretinoin was stopped in the latter two patients, who also had obesity as a risk factor for their hypertriglyceridemia.
“None of these patients had clinical sequelae from their hypertriglyceridemia, such as pancreatitis at baseline or follow-up,” Ms. Parthasarathy said. “However, since pancreatitis would be expected to be exceedingly rare, the sample size may be limited in identifying this adverse effect.”
She noted that while isotretinoin might cause a significant increase in lipid levels, the mean levels remained within normal limits at both baseline and follow-up. “Of the patients with elevated triglycerides at baseline and follow-up, obesity may have been a potential risk factor,” she said. “This could suggest a possible strategy for reduced testing in nonobese isotretinoin patients, which can be further explored in larger study populations.”
In addition, “there was a lack of significant change in AST and ALT in this study and adult studies, as well as minimal evidence for pediatric retinoid-induced hepatotoxicity, which raises the question of the necessity of baseline and follow-up comprehensive metabolic panel testing,” Ms. Parthasarathy added. “Clinicians must weigh the laboratory values with the costs of laboratory testing, including opportunity costs such as time, monetary costs, and the discomfort of testing for pediatric patients.”
The study’s senior author was A. Yasmine Kirkorian, MD, chief of dermatology at Children’s National Hospital, Washington. The researchers reported having no relevant financial disclosures.
.
“Isotretinoin is a very effective treatment for severe acne,” Varsha Parthasarathy said at the annual meeting of the Society for Pediatric Dermatology. “However, initiating this medication requires a complex process of laboratory testing,” which includes human chorionic gonadotropin pregnancy testing, because isotretinoin is a teratogen, as well as lipid labs and liver function tests, she noted. “Importantly, triglycerides are measured due to an association in adults between isotretinoin and hypertriglyceridemia-associated pancreatitis. However, these findings in children are limited to case reports, as are findings of retinoid-induced hepatotoxicity.”
To identify the role of isotretinoin on changes in lipids, aspartate aminotransferase (AST), and alanine aminotransferase (ALT), and to determine the impact on treatment course, Ms. Parthasarathy, a 4-year medical student at George Washington University, Washington, and colleagues retrospectively reviewed the charts of 130 patients aged 12-21 years who were cared for at Children’s National Hospital between January 2012 and October 2020. Nearly two-thirds (65%) were male, their average age was 16 years, and the mean time to obtain follow-up labs after starting isotretinoin was 3.25 months.
Between baseline and follow-up, the researchers observed increases in total cholesterol, triglycerides, and LDL (P less than .001 for all associations) and a decrease in HDL (P = .001), but there were no significant changes in AST or ALT levels. These findings were consistent with prior studies in adults examining the utility of these laboratory tests, most notably a 2016 study by Timothy J. Hansen, MD, and colleagues.
Among the 13 patients with elevated triglycerides at baseline, 9 (69%) were overweight or obese. Of the 20 patients with elevated triglycerides at follow-up, 11 patients (55%) were obese. At follow-up, 11 patients had levels of 200-500 mg/dL (grade I elevation), and 2 patients had levels of 501-1,000 mg/dL (grade II elevation). Isotretinoin was stopped in the latter two patients, who also had obesity as a risk factor for their hypertriglyceridemia.
“None of these patients had clinical sequelae from their hypertriglyceridemia, such as pancreatitis at baseline or follow-up,” Ms. Parthasarathy said. “However, since pancreatitis would be expected to be exceedingly rare, the sample size may be limited in identifying this adverse effect.”
She noted that while isotretinoin might cause a significant increase in lipid levels, the mean levels remained within normal limits at both baseline and follow-up. “Of the patients with elevated triglycerides at baseline and follow-up, obesity may have been a potential risk factor,” she said. “This could suggest a possible strategy for reduced testing in nonobese isotretinoin patients, which can be further explored in larger study populations.”
In addition, “there was a lack of significant change in AST and ALT in this study and adult studies, as well as minimal evidence for pediatric retinoid-induced hepatotoxicity, which raises the question of the necessity of baseline and follow-up comprehensive metabolic panel testing,” Ms. Parthasarathy added. “Clinicians must weigh the laboratory values with the costs of laboratory testing, including opportunity costs such as time, monetary costs, and the discomfort of testing for pediatric patients.”
The study’s senior author was A. Yasmine Kirkorian, MD, chief of dermatology at Children’s National Hospital, Washington. The researchers reported having no relevant financial disclosures.
FROM SPD 2021
Isotretinoin benefits similar in overweight, obese adolescents, and those in normal weight range
a retrospective cohort study found.
“Oral isotretinoin is among the most effective treatments for acne and is indicated for the treatment of severe acne or when first-line regimens have failed,” Maggie Tallmadge said at the annual meeting of the Society for Pediatric Dermatology. In adolescents with acne, isotretinoin is prescribed at a dose of 0.5-1 mg/kg per day “with the goal of reaching a cumulative dose of 120-150 mg/kg and clinical clearance with durable remission,” she said. “Most providers do not prescribe a daily dose over 80 mg due to perceived increased risk of side effects, including xerosis, cheilitis, liver dysfunction, and acne flare. However, many adolescents weigh over 80 kg and are therefore effectively underdosed, prolonging treatment time and possibly increasing the risk of side effects due to prolonged therapy.”
To evaluate differences in treatment courses among normal-weight, overweight, and obese adolescents, and the efficacy and safety of treatment, Ms. Tallmadge, a third-year medical student at the Medical College of Wisconsin, Milwaukee, and colleagues completed a retrospective chart review of 550 dermatology patients at Children’s Wisconsin, also in Milwaukee, who completed at least 2 months of isotretinoin treatment for acne when they were between the ages of 10 and 24, from November 2012 to January 2020. They collected data on age, weight, height, daily dose, cumulative dose, time to acne clearance, side effects, and acne recurrence after treatment, and classified patients as normal weight, overweight, or obese based on their body mass index for age percentile.
Of the 550 patients, 367 (67%) were normal weight, 101 (18%) were overweight, and 82 (15%) were obese. The median age of those in the normal-weight and overweight groups was 16, and was 15 in the obese group.
There was were significant differences in the median cumulative dose in each weight group: 143.7 mg/kg for normal-weight patients, 138.2 mg/kg for overweight patients, and 140.6 mg/kg for obese patients (P < .001).
“Despite achieving different cumulative doses, there was no difference in acne clearance, relapse, and most side effects among the three [body mass index] cohorts,” Ms. Tallmadge said. “Thus, it appears that current treatment strategies may be appropriate for overweight and obese adolescents.”
The proportion of patients with acne clearance did not differ significantly among the three groups of patients: 62% who were in the normal weight range, 60% who were overweight, and 59% who were obese had clearance of facial acne with treatment (P = .84).
Of patients whose treatment course was completed by the time of data collection, the proportion with acne recurrences was similar between the three groups: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients (P > .05). Of patients whose treatment course was completed by the time of data collection, there was no significant differences in acne recurrence: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients.
However, the proportion of patients reporting headaches differed significantly between the groups: 29% of normal-weight patients, compared with 40% of both overweight and obese patients (P = .035). The researchers also observed a significant positive correlation between increased BMI and increased triglyceride and ALT levels during treatment (P < .001 for both associations), yet no elevations required clinical action.
Funding for the study was provided by the MCW Medical Student Summer Research Program and the American Acne & Rosacea Society.
a retrospective cohort study found.
“Oral isotretinoin is among the most effective treatments for acne and is indicated for the treatment of severe acne or when first-line regimens have failed,” Maggie Tallmadge said at the annual meeting of the Society for Pediatric Dermatology. In adolescents with acne, isotretinoin is prescribed at a dose of 0.5-1 mg/kg per day “with the goal of reaching a cumulative dose of 120-150 mg/kg and clinical clearance with durable remission,” she said. “Most providers do not prescribe a daily dose over 80 mg due to perceived increased risk of side effects, including xerosis, cheilitis, liver dysfunction, and acne flare. However, many adolescents weigh over 80 kg and are therefore effectively underdosed, prolonging treatment time and possibly increasing the risk of side effects due to prolonged therapy.”
To evaluate differences in treatment courses among normal-weight, overweight, and obese adolescents, and the efficacy and safety of treatment, Ms. Tallmadge, a third-year medical student at the Medical College of Wisconsin, Milwaukee, and colleagues completed a retrospective chart review of 550 dermatology patients at Children’s Wisconsin, also in Milwaukee, who completed at least 2 months of isotretinoin treatment for acne when they were between the ages of 10 and 24, from November 2012 to January 2020. They collected data on age, weight, height, daily dose, cumulative dose, time to acne clearance, side effects, and acne recurrence after treatment, and classified patients as normal weight, overweight, or obese based on their body mass index for age percentile.
Of the 550 patients, 367 (67%) were normal weight, 101 (18%) were overweight, and 82 (15%) were obese. The median age of those in the normal-weight and overweight groups was 16, and was 15 in the obese group.
There was were significant differences in the median cumulative dose in each weight group: 143.7 mg/kg for normal-weight patients, 138.2 mg/kg for overweight patients, and 140.6 mg/kg for obese patients (P < .001).
“Despite achieving different cumulative doses, there was no difference in acne clearance, relapse, and most side effects among the three [body mass index] cohorts,” Ms. Tallmadge said. “Thus, it appears that current treatment strategies may be appropriate for overweight and obese adolescents.”
The proportion of patients with acne clearance did not differ significantly among the three groups of patients: 62% who were in the normal weight range, 60% who were overweight, and 59% who were obese had clearance of facial acne with treatment (P = .84).
Of patients whose treatment course was completed by the time of data collection, the proportion with acne recurrences was similar between the three groups: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients (P > .05). Of patients whose treatment course was completed by the time of data collection, there was no significant differences in acne recurrence: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients.
However, the proportion of patients reporting headaches differed significantly between the groups: 29% of normal-weight patients, compared with 40% of both overweight and obese patients (P = .035). The researchers also observed a significant positive correlation between increased BMI and increased triglyceride and ALT levels during treatment (P < .001 for both associations), yet no elevations required clinical action.
Funding for the study was provided by the MCW Medical Student Summer Research Program and the American Acne & Rosacea Society.
a retrospective cohort study found.
“Oral isotretinoin is among the most effective treatments for acne and is indicated for the treatment of severe acne or when first-line regimens have failed,” Maggie Tallmadge said at the annual meeting of the Society for Pediatric Dermatology. In adolescents with acne, isotretinoin is prescribed at a dose of 0.5-1 mg/kg per day “with the goal of reaching a cumulative dose of 120-150 mg/kg and clinical clearance with durable remission,” she said. “Most providers do not prescribe a daily dose over 80 mg due to perceived increased risk of side effects, including xerosis, cheilitis, liver dysfunction, and acne flare. However, many adolescents weigh over 80 kg and are therefore effectively underdosed, prolonging treatment time and possibly increasing the risk of side effects due to prolonged therapy.”
To evaluate differences in treatment courses among normal-weight, overweight, and obese adolescents, and the efficacy and safety of treatment, Ms. Tallmadge, a third-year medical student at the Medical College of Wisconsin, Milwaukee, and colleagues completed a retrospective chart review of 550 dermatology patients at Children’s Wisconsin, also in Milwaukee, who completed at least 2 months of isotretinoin treatment for acne when they were between the ages of 10 and 24, from November 2012 to January 2020. They collected data on age, weight, height, daily dose, cumulative dose, time to acne clearance, side effects, and acne recurrence after treatment, and classified patients as normal weight, overweight, or obese based on their body mass index for age percentile.
Of the 550 patients, 367 (67%) were normal weight, 101 (18%) were overweight, and 82 (15%) were obese. The median age of those in the normal-weight and overweight groups was 16, and was 15 in the obese group.
There was were significant differences in the median cumulative dose in each weight group: 143.7 mg/kg for normal-weight patients, 138.2 mg/kg for overweight patients, and 140.6 mg/kg for obese patients (P < .001).
“Despite achieving different cumulative doses, there was no difference in acne clearance, relapse, and most side effects among the three [body mass index] cohorts,” Ms. Tallmadge said. “Thus, it appears that current treatment strategies may be appropriate for overweight and obese adolescents.”
The proportion of patients with acne clearance did not differ significantly among the three groups of patients: 62% who were in the normal weight range, 60% who were overweight, and 59% who were obese had clearance of facial acne with treatment (P = .84).
Of patients whose treatment course was completed by the time of data collection, the proportion with acne recurrences was similar between the three groups: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients (P > .05). Of patients whose treatment course was completed by the time of data collection, there was no significant differences in acne recurrence: 25% of normal-weight patients, 27% of overweight patients, and 35% of obese patients.
However, the proportion of patients reporting headaches differed significantly between the groups: 29% of normal-weight patients, compared with 40% of both overweight and obese patients (P = .035). The researchers also observed a significant positive correlation between increased BMI and increased triglyceride and ALT levels during treatment (P < .001 for both associations), yet no elevations required clinical action.
Funding for the study was provided by the MCW Medical Student Summer Research Program and the American Acne & Rosacea Society.
FROM SPD 2021
Study spanning 2 decades offers insights into pediatric psoriasis trends
, while predictors of moderate to severe disease include morphology, non-White race, and culture-confirmed infection.
Those are among the key findings from a retrospective analysis of pediatric psoriasis patients who were seen at the University of California, San Francisco, over a 24-year period.
“Overall, our data support prior findings of age- and sex-based differences in location and morphology and presents new information demonstrating associations with severity,” presenting study author Carmel Aghdasi said during the annual meeting of the Society for Pediatric Dermatology. “We provide evidence of the increased use of systemic and biologic therapies over time, an important step in ensuring pediatric patients are adequately treated.”
To characterize the demographics, clinical features, comorbidities, and treatments, and to determine predictors of severity and changes in treatment patterns over 2 decades in a large cohort of pediatric psoriasis patients, Ms. Aghdasi, a 4th-year medical student at the University of California, San Francisco, and colleagues retrospectively evaluated the records of 754 pediatric patients up to 18 years of age who were seen at UCSF for psoriasis from 1997 to 2021. They collected demographic, clinical, familial, comorbidity, and treatment data and divided the cohort into two groups by date of last visit.
Group 1 consisted of 332 patients whose last visit was between 2001 and 2011, while the second group included 422 patients whose last visit was between 2012 and 2021. The researchers also divided the cohort into three age groups: infants (0-2 years of age), children (3-12 years of age), and adolescents (13-18 years of age).
Slightly more than half of the patients (55%) were female and 67% presented between ages 3 and 12. (Seventy-four patients were in the youngest category, 0-2 years, when they presented.) The average age of disease onset was 7 years, the average age at presentation to pediatric dermatology was 8.8 years, and 37% of the total cohort were overweight or obese. The top four comorbidities were being overweight or obese (37%), followed by atopic dermatitis (19%), psychiatric disease (7%), and arthritis (4%).
Plaque was the most common morphology (56%), while the most common sites of involvement were the head and neck (69%), extremities (61%), and trunk (44%). About half of the cohort (51%) had mild disease, 15% had culture-confirmed infections (9% had Streptococcal infections), and 66% of patients reported itch as a symptom.
The researchers observed that inverse psoriasis was significantly more common in infants and decreased with age. Anogenital involvement was more common in males and in those aged 0-2, while head and neck involvement was more common in females. Nail involvement was more common in childhood.
Topical therapy was the most common treatment overall and by far the most common among those in the 0-2 age category. “Overall, phototherapy was used in childhood and adolescents but almost never in infancy,” Ms. Aghdasi said. “Looking at changes in systemic treatment over time, conventional systemic use increased in infants and children and decreased in adolescents. Biologic use increased in all ages, most notably in children aged 3-12 years old.”
Multivariate regression analyses revealed that the following independent variables predicted moderate to severe psoriasis: adolescent age (adjusted odds ratio, 1.9; P = .03), guttate morphology (aOR, 2.2; P = .006), plaque and guttate morphology (aOR, 7.6; P less than .001), pustular or erythrodermic morphology (aOR, 5; P = .003), culture-confirmed infection (aOR, 2; P = .007), Black race (aOR, 3.3; P = .007), Asian race (aOR, 1.8; P = .04, and Hispanic race (aOR, 1.9; P = .03).
“Further analysis is needed to elucidate the influence of race on severity and of the clinical utility of infection as a marker of severity,” Ms. Aghdasi said. “Interestingly, we did not find that obesity was a marker of severity in our cohort.”
In an interview, senior study author Kelly M. Cordoro, MD, professor of dermatology and pediatrics at UCSF, noted that this finding conflicts with prior studies showing an association between obesity and severe psoriasis in children.
“Though methodologies and patient populations differ among studies, what is striking,” she said, is the percentage of overweight/obese patients (37%; defined as a body mass index ≥ 85th percentile) “in our 2-decade single institution dataset.” This “is nearly identical” to the percentage of patients with excess adiposity – 37.9% (also defined as BMI ≥ 85th percentile) – in an international cross-sectional study, which also identified an association between obesity (BMI ≥ 95th percentile) and psoriasis severity in children, she noted.
“What is clear is the strong association between obesity and childhood psoriasis, as multiple studies, including ours, confirm obesity as a major comorbidity of pediatric psoriasis,” Dr. Cordoro said. “Both conditions must be adequately managed to reduce the risk of adverse health outcomes for obese patients with psoriasis.”
The other study coauthors were Dana Feigenbaum, MD, and Alana Ju, MD. The work was supported by the UCSF Yearlong Inquiry Program. The researchers reported having no relevant financial disclosures.
, while predictors of moderate to severe disease include morphology, non-White race, and culture-confirmed infection.
Those are among the key findings from a retrospective analysis of pediatric psoriasis patients who were seen at the University of California, San Francisco, over a 24-year period.
“Overall, our data support prior findings of age- and sex-based differences in location and morphology and presents new information demonstrating associations with severity,” presenting study author Carmel Aghdasi said during the annual meeting of the Society for Pediatric Dermatology. “We provide evidence of the increased use of systemic and biologic therapies over time, an important step in ensuring pediatric patients are adequately treated.”
To characterize the demographics, clinical features, comorbidities, and treatments, and to determine predictors of severity and changes in treatment patterns over 2 decades in a large cohort of pediatric psoriasis patients, Ms. Aghdasi, a 4th-year medical student at the University of California, San Francisco, and colleagues retrospectively evaluated the records of 754 pediatric patients up to 18 years of age who were seen at UCSF for psoriasis from 1997 to 2021. They collected demographic, clinical, familial, comorbidity, and treatment data and divided the cohort into two groups by date of last visit.
Group 1 consisted of 332 patients whose last visit was between 2001 and 2011, while the second group included 422 patients whose last visit was between 2012 and 2021. The researchers also divided the cohort into three age groups: infants (0-2 years of age), children (3-12 years of age), and adolescents (13-18 years of age).
Slightly more than half of the patients (55%) were female and 67% presented between ages 3 and 12. (Seventy-four patients were in the youngest category, 0-2 years, when they presented.) The average age of disease onset was 7 years, the average age at presentation to pediatric dermatology was 8.8 years, and 37% of the total cohort were overweight or obese. The top four comorbidities were being overweight or obese (37%), followed by atopic dermatitis (19%), psychiatric disease (7%), and arthritis (4%).
Plaque was the most common morphology (56%), while the most common sites of involvement were the head and neck (69%), extremities (61%), and trunk (44%). About half of the cohort (51%) had mild disease, 15% had culture-confirmed infections (9% had Streptococcal infections), and 66% of patients reported itch as a symptom.
The researchers observed that inverse psoriasis was significantly more common in infants and decreased with age. Anogenital involvement was more common in males and in those aged 0-2, while head and neck involvement was more common in females. Nail involvement was more common in childhood.
Topical therapy was the most common treatment overall and by far the most common among those in the 0-2 age category. “Overall, phototherapy was used in childhood and adolescents but almost never in infancy,” Ms. Aghdasi said. “Looking at changes in systemic treatment over time, conventional systemic use increased in infants and children and decreased in adolescents. Biologic use increased in all ages, most notably in children aged 3-12 years old.”
Multivariate regression analyses revealed that the following independent variables predicted moderate to severe psoriasis: adolescent age (adjusted odds ratio, 1.9; P = .03), guttate morphology (aOR, 2.2; P = .006), plaque and guttate morphology (aOR, 7.6; P less than .001), pustular or erythrodermic morphology (aOR, 5; P = .003), culture-confirmed infection (aOR, 2; P = .007), Black race (aOR, 3.3; P = .007), Asian race (aOR, 1.8; P = .04, and Hispanic race (aOR, 1.9; P = .03).
“Further analysis is needed to elucidate the influence of race on severity and of the clinical utility of infection as a marker of severity,” Ms. Aghdasi said. “Interestingly, we did not find that obesity was a marker of severity in our cohort.”
In an interview, senior study author Kelly M. Cordoro, MD, professor of dermatology and pediatrics at UCSF, noted that this finding conflicts with prior studies showing an association between obesity and severe psoriasis in children.
“Though methodologies and patient populations differ among studies, what is striking,” she said, is the percentage of overweight/obese patients (37%; defined as a body mass index ≥ 85th percentile) “in our 2-decade single institution dataset.” This “is nearly identical” to the percentage of patients with excess adiposity – 37.9% (also defined as BMI ≥ 85th percentile) – in an international cross-sectional study, which also identified an association between obesity (BMI ≥ 95th percentile) and psoriasis severity in children, she noted.
“What is clear is the strong association between obesity and childhood psoriasis, as multiple studies, including ours, confirm obesity as a major comorbidity of pediatric psoriasis,” Dr. Cordoro said. “Both conditions must be adequately managed to reduce the risk of adverse health outcomes for obese patients with psoriasis.”
The other study coauthors were Dana Feigenbaum, MD, and Alana Ju, MD. The work was supported by the UCSF Yearlong Inquiry Program. The researchers reported having no relevant financial disclosures.
, while predictors of moderate to severe disease include morphology, non-White race, and culture-confirmed infection.
Those are among the key findings from a retrospective analysis of pediatric psoriasis patients who were seen at the University of California, San Francisco, over a 24-year period.
“Overall, our data support prior findings of age- and sex-based differences in location and morphology and presents new information demonstrating associations with severity,” presenting study author Carmel Aghdasi said during the annual meeting of the Society for Pediatric Dermatology. “We provide evidence of the increased use of systemic and biologic therapies over time, an important step in ensuring pediatric patients are adequately treated.”
To characterize the demographics, clinical features, comorbidities, and treatments, and to determine predictors of severity and changes in treatment patterns over 2 decades in a large cohort of pediatric psoriasis patients, Ms. Aghdasi, a 4th-year medical student at the University of California, San Francisco, and colleagues retrospectively evaluated the records of 754 pediatric patients up to 18 years of age who were seen at UCSF for psoriasis from 1997 to 2021. They collected demographic, clinical, familial, comorbidity, and treatment data and divided the cohort into two groups by date of last visit.
Group 1 consisted of 332 patients whose last visit was between 2001 and 2011, while the second group included 422 patients whose last visit was between 2012 and 2021. The researchers also divided the cohort into three age groups: infants (0-2 years of age), children (3-12 years of age), and adolescents (13-18 years of age).
Slightly more than half of the patients (55%) were female and 67% presented between ages 3 and 12. (Seventy-four patients were in the youngest category, 0-2 years, when they presented.) The average age of disease onset was 7 years, the average age at presentation to pediatric dermatology was 8.8 years, and 37% of the total cohort were overweight or obese. The top four comorbidities were being overweight or obese (37%), followed by atopic dermatitis (19%), psychiatric disease (7%), and arthritis (4%).
Plaque was the most common morphology (56%), while the most common sites of involvement were the head and neck (69%), extremities (61%), and trunk (44%). About half of the cohort (51%) had mild disease, 15% had culture-confirmed infections (9% had Streptococcal infections), and 66% of patients reported itch as a symptom.
The researchers observed that inverse psoriasis was significantly more common in infants and decreased with age. Anogenital involvement was more common in males and in those aged 0-2, while head and neck involvement was more common in females. Nail involvement was more common in childhood.
Topical therapy was the most common treatment overall and by far the most common among those in the 0-2 age category. “Overall, phototherapy was used in childhood and adolescents but almost never in infancy,” Ms. Aghdasi said. “Looking at changes in systemic treatment over time, conventional systemic use increased in infants and children and decreased in adolescents. Biologic use increased in all ages, most notably in children aged 3-12 years old.”
Multivariate regression analyses revealed that the following independent variables predicted moderate to severe psoriasis: adolescent age (adjusted odds ratio, 1.9; P = .03), guttate morphology (aOR, 2.2; P = .006), plaque and guttate morphology (aOR, 7.6; P less than .001), pustular or erythrodermic morphology (aOR, 5; P = .003), culture-confirmed infection (aOR, 2; P = .007), Black race (aOR, 3.3; P = .007), Asian race (aOR, 1.8; P = .04, and Hispanic race (aOR, 1.9; P = .03).
“Further analysis is needed to elucidate the influence of race on severity and of the clinical utility of infection as a marker of severity,” Ms. Aghdasi said. “Interestingly, we did not find that obesity was a marker of severity in our cohort.”
In an interview, senior study author Kelly M. Cordoro, MD, professor of dermatology and pediatrics at UCSF, noted that this finding conflicts with prior studies showing an association between obesity and severe psoriasis in children.
“Though methodologies and patient populations differ among studies, what is striking,” she said, is the percentage of overweight/obese patients (37%; defined as a body mass index ≥ 85th percentile) “in our 2-decade single institution dataset.” This “is nearly identical” to the percentage of patients with excess adiposity – 37.9% (also defined as BMI ≥ 85th percentile) – in an international cross-sectional study, which also identified an association between obesity (BMI ≥ 95th percentile) and psoriasis severity in children, she noted.
“What is clear is the strong association between obesity and childhood psoriasis, as multiple studies, including ours, confirm obesity as a major comorbidity of pediatric psoriasis,” Dr. Cordoro said. “Both conditions must be adequately managed to reduce the risk of adverse health outcomes for obese patients with psoriasis.”
The other study coauthors were Dana Feigenbaum, MD, and Alana Ju, MD. The work was supported by the UCSF Yearlong Inquiry Program. The researchers reported having no relevant financial disclosures.
FROM SPD 2021
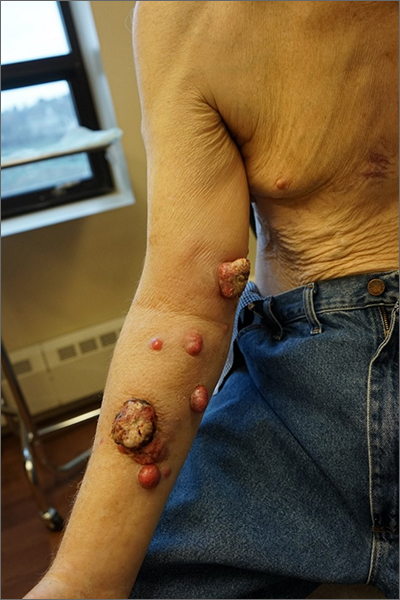
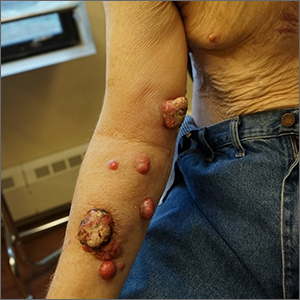
Nodules on the arm
Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155
Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Excisional biopsy was performed on a small tumor and immune staining suggested lung cancer as the primary source.
Metastatic cancer of an unknown primary source accounts for 3% to 5% of invasive cancers.1 Poorly differentiated SCC, as in this case, may arise from skin, lung, or head and neck SCC. Risks of metastatic disease in cutaneous SCC include size greater than 2 cm, depth greater than 2 mm, and location on the ear, hand, lip, or within a burn.2 Recurrent tumors, poorly differentiated tumors, and tumors demonstrating perineural invasion also increase the risk of metastasis.
Having failed both previous treatments, further tissue characterization offered some hope of effective immunotherapy. Specifically, SCC from lung cancer as a primary source may express programmed-death ligand 1 (PD-L1), a transmembrane protein that suppresses adaptive immune responses. Pembrolizumab inhibits this molecule leading to a more aggressive immune response to tumor cells. Additionally, tumor profiling of specific oncogenes can highlight potentially beneficial therapies.
In this case, gene profiling identified several active tumor oncogenes, but PD-L1 was not strongly expressed. Despite the effort, this process did not uncover practical or novel treatment options. The patient was offered an empiric trial of immunotherapy but opted to pursue palliative therapy alone and passed away 2 months later.
This case highlights the role of multidisciplinary care of metastatic disease to the skin and the potential role, and limitations, of skin biopsy in tumor profiling and treatment guidance. Even though the most likely primary site was lung, cutaneous metastases can be equally devastating.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155
1. Pavlidis N, Pentheroudakis G. Cancer of unknown primary site. Lancet. 2012;379:1428-1435. doi: 10.1016/S0140-6736(11)61178-1
2. Brougham NDLS, Dennett ER, Cameron R, et al. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J Surg Oncol. 2012;106:811-815. doi: 10.1002/jso.23155
New analysis puts U.S. psoriasis prevalence at 3%
, according to an analysis of national survey data from 2011 to 2014.
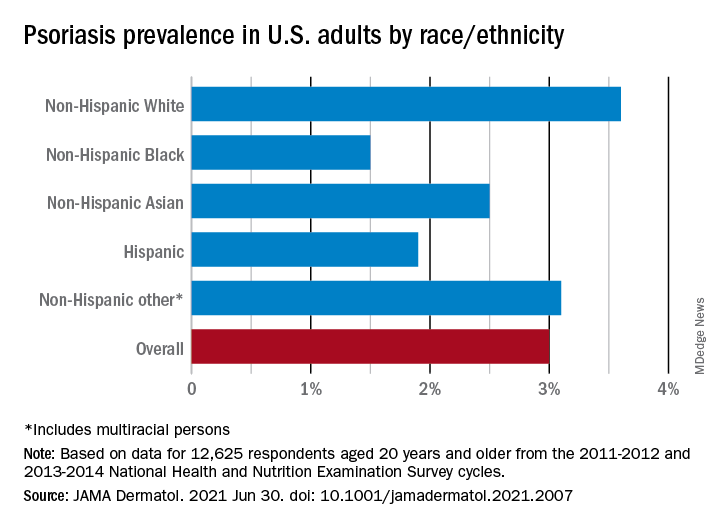
“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
FROM JAMA DERMATOLOGY
Expert shares practical considerations when prescribing dupilumab
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
FROM REVOLUTIONIZING AD 2021
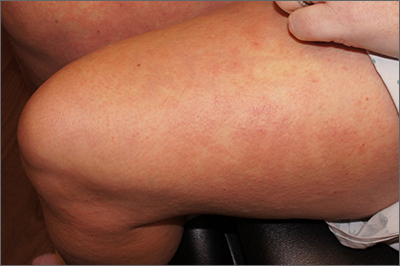
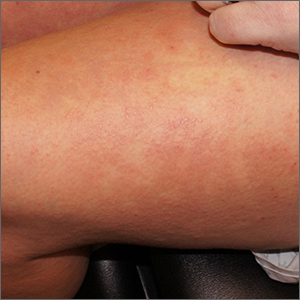
Rash after a medication change
A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.
A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
A punch biopsy revealed lichenoid interface dermatitis with eosinophils and mild spongiosis—consistent with, but not conclusive for, a drug eruption. A complete blood count (CBC) and comprehensive metabolic panel revealed elevated levels of eosinophils and transaminases which raised the possibility of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. In this patient’s case, the transaminitis suggested some mild hepatitis; the liver is the most common organ involved in DRESS.
Diagnostic criteria for DRESS syndrome need not always be met but can include fever, lymphadenopathy, facial edema, and a morbilliform rash presenting 2 to 3 weeks after drug exposure. In this case, a lack of fever, facial edema, and other systemic symptoms favored a less severe drug eruption or what has been described as mini-DRESS.1 DRESS syndrome often merits hospitalization, and in 10% of cases it can be fatal.1
The patient in this case was started on prednisone 60 mg/d and her OCPs were discontinued. One week later, a repeat CBC showed normalized levels of eosinophils and transaminases. However, shortly after a 3-week taper of the prednisone, her levels of eosinophils and transaminases rose again. A repeat prednisone taper finally led to complete resolution of rash and sustained normalization of eosinophil and transaminase levels.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.
Isaacs M, Cardones AR, Rahnama-Moghadam S. DRESS syndrome: clinical myths and pearls. Cutis. 2018;102:322-326.
Baricitinib found effective for moderate to severe AD out to 52 weeks
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
“With long-term therapy, the baricitinib 2 mg response remains stable or slightly improved, compared with week 16 for skin inflammation, itch, sleep, and quality of life,” presenting study author Eric L. Simpson, MD, said during the Revolutionizing Atopic Dermatitis symposium.
Baricitinib is an oral selective Janus kinase 1/JAK2 inhibitor being developed for the treatment of moderate to severe AD in adults who are candidates for systemic therapy. The drug is already approved for AD in Europe at the 2-mg and 4-mg doses. A 16-week placebo-controlled study conducted in North America known as BREEZE-AD5 found that 2 mg of baricitinib improved disease in adults with moderate to severe AD.
For the current analysis, Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland, and colleagues integrated data from BREEZE-AD5 and BREEZE-AD6, an ongoing, open-label study of BREEZE-AD5, to evaluate the long-term efficacy and safety of baricitinib 2 mg in patients with moderate to severe AD.
At week 16, patients from BREEZE-AD5 who were on baricitinib 2 mg could either continue the trial out to week 52, or they could transition to BREEZE-AD6 if they were nonresponders. The use of low-potency corticosteroids was permitted after week 16 in BREEZE-AD5 and throughout BREEZE-AD6. Endpoints of interest at week 52 in both trials were the proportions of patients with 75% or greater improvement from baseline in the Eczema and Severity Index (EASI75), a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1, a Dermatology Life Quality Index (DLQI) score of 5 or less, as well as mean SCORing AD (SCORAD) visual analog scales of itch and sleeplessness scores, and the mean percent change from baseline in EASI score.
Dr. Simpson presented data on 146 patients from both trials who were randomized to baricitinib 2 mg. Their mean age was 40 years, 53% were female, 58% were White, 21% were Black, 15% were Asian, and the remainder were from other backgrounds. Their mean duration of AD was 16 years and their average EASI score was 26.6. At weeks 16, 32, and 52, the proportion of patients who achieved an EASI75 response was 40%, 51%, and 49%, respectively, while the mean percent change from baseline in EASI score was –50%, –59%, and –57%.
At weeks 16, 32, and 52, the vIGA-AD responses of 0 or 1 were observed in 27%, 38%, and 31% of patients. The mean SCORAD pruritus score improved from 7.7 at baseline to 4.8 at week 16 and was maintained at weeks 32 (3.8) and 52 (4.3). The mean SCORAD sleeplessness score also improved from 6.5 at baseline to 3.9 at week 16 and remained stable through weeks 32 (3.4) and 52 (3.7).
Finally, among 129 patients who had a baseline DLQI of greater than 5, 39% had DLQI scores of 5 or lower at week 16, compared with 49% at week 32 and 45% at week 52, indicating a small or no effect of AD on quality of life.
The study was sponsored by Eli Lilly, which is developing baricitinib. Dr. Simpson disclosed that he is a consultant to and/or an investigator for several pharmaceutical companies, including Eli Lilly.
FROM REVOLUTIONIZING AD 2021
In Black patients, acne scarring might not mean what you think
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
Treating the needs of patients of color requires an understanding of differences that may not be readily apparent, a dermatologist told colleagues. For example, of the term that may be misinterpreted in the doctor’s office.
“Scarring is not usually what they’re talking about, although they may have some of that as well. They’re [typically] talking about what we know as postinflammatory hyperpigmentation, not scarring. So right away, you have to clarify,” Amy McMichael, MD, professor and chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C., said in a presentation at the Inaugural Symposium for Inflammatory Skin Disease. “When you’re talking about scarring, do you mean the dark spots? What exactly are you concerned about?”
Dr. McMichael highlighted a 2014 study that reported the results of a survey of 208 women (51% were White; 49% were non-White), which included 51 Black, 23 Hispanic, and 16 Asian women aged 25-45 (mean age, 35) with 25 or more lesions. White women were more troubled by facial acne than were women of color (89% vs. 76%, respectively, P < .05), and they were more likely to say lesion clearance was most important to them (58% vs. 32%, respectively, P < .001).
Meanwhile, non-White women were much more likely than were White women to say that clearance of postinflammatory hyperpigmentation was most important to them (42% vs. 8%, respectively, P < .0001).
“Seventy percent of [non-White women] felt that their race and ethnicity required targeted attention [in treatment], and two-thirds desired acne treatment that was designed to meet the needs of their skin type,” Dr. McMichael said. “If you don’t address the issues, if you don’t talk about the pigmentation with them or explain how you’re going to address it, people don’t feel heard. They don’t feel like they’ve really seen a dermatologist who understands their needs.”
She added that it’s crucial to ask about over-the-counter products. “If you don’t discuss them, they’ll assume that what they’re doing is okay.” She warns her patients against using and exposing their skin and face to cocoa butter and oils such as tea tree oil.
Research has suggested that among people of color, Blacks and Hispanics are most likely to experience dyspigmentation and scarring, Dr. McMichael said. She advised colleagues to be aware of pomade acne in these two groups of patients. Pomade acne appears along the hair line and is caused by the use of hair products. She also cautioned about acne cosmetica, which can be triggered by products such as makeup, used to cover up acne and postinflammatory hyperpigmentation.
As for acne treatments, Dr. McMichael highlighted a long list of familiar topical and oral agents and procedural options. Less familiar strategies include laser and light-based therapies, she said.
As for up-and-coming options, she pointed to topical minocycline, “which allows us to use an anti-inflammatory agent topically rather than orally when we’re trying to get away from using a lot of oral antibiotics.”
Also consider whether female patients have polycystic ovary syndrome, she said. “Then you might consider spironolactone. I certainly use a lot more of that these days to try to avoid long-term oral antibiotics.”
She recommended earlier use of isotretinoin in patients overall, and she urged colleagues to proceed with their standard retinoid approaches. However, she noted that she lets patients know that she’ll focus first on treating the acne itself and then work on the dark spots in later treatments. “If you give people a bleaching agent in the beginning, they’re going to stop using their main products, and they’re going to chase those dark spots. That’s just something that they can’t help doing.”
Dr. McMichael disclosed investigator and consultant relationships with multiple drug makers.
FROM SISD 2021
Ruxolitinib cream for atopic dermatitis found to be effective, safe up to 52 weeks
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
results from a long-term analysis of clinical trial data showed.
“The incidence of application-site reactions was low, and there were no clinically meaningful changes or trends in hematologic parameters,” Kim Papp, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium.
Ruxolitinib cream is a selective Janus kinase 1/JAK2 inhibitor being developed by Incyte for the treatment of atopic dermatitis (AD).
According to a press release from the company, the Food and Drug Administration has extended the New Drug Application review period for the agent by 3 months to September 2021. If approved, it would become first topical JAK inhibitor for use in dermatology.
In two phase 3, randomized studies of identical design involving 1,249 patients aged 12 and older with AD – TRuE-AD1 and TRuE-AD2 – ruxolitinib cream demonstrated anti-inflammatory activity, with rapid and sustained antipruritic action, compared with vehicle. To be eligible for the trials patients with an Investigator’s Global Assessment (IGA) score of 2 or 3 and 3%-20% of affected body surface area (BSA) were randomized (2:2:1) to twice-daily 0.75% ruxolitinib cream, 1.5% ruxolitinib cream, or vehicle cream for 8 continuous weeks.
A recently published report found that significantly more patients in TRuE-AD1 and TRuE-AD2 achieved IGA treatment success with 0.75% (50% vs. 39%, respectively) and 1.5% ruxolitinib cream (53.8% vs. 51.3%), compared with vehicle (15.1% vs. 7.6%; P < .0001) at week 8. In addition, significant reductions in itch, compared with vehicle, were reported within 12 hours of first applying 1.5% ruxolitinib cream (P < .05).
Longterm data
During the symposium, Dr. Papp presented long-term safety data of ruxolitinib cream in patients who were followed for an additional 44 weeks. Those initially randomized to vehicle were rerandomized 1:1 (blinded) to either ruxolitinib cream regimen. They were instructed to treat skin areas with active AD only and to stop treatment 3 days after clearance of lesions, and to restart treatment with ruxolitinib cream at the first sign of recurrence. Safety and tolerability were assessed by frequency and severity of adverse events, while disease control was measured by the proportion of patients with an IGA score of 0 or 1 and the affected BSA.
Dr. Papp, a dermatologist and founder of Probity Medical Research, Waterloo, Ont., reported that 543 patients from TRuE-AD1 and 530 from TRuE-AD2 entered the long-term analysis and that about 78% of these patients completed the study. From weeks 12 to 52, the proportion of patients with an IGA score of 0 or 1 with 0.75% and 1.5% ruxolitinib cream ranged from 62%-77% and 67%-77%, respectively, in TRuE-AD1 to 60%-77% and 72%-80% in TRuE-AD2.
The measured mean total affected BSA was less than 3% throughout the follow-up period in the 1.5% ruxolitinib cream arm in TRuE-AD1 and TRuE-AD2 and was less than 3% in the 0.75% ruxolitinib cream arm during most of the study period.
In a pooled safety analysis, treatment-emergent adverse events (TEAEs) were reported in 60% and 54% of patients who applied 0.75% and 1.5% ruxolitinib cream, respectively, over 44 weeks. The frequency of application-site reactions remained low. Specifically, treatment-related adverse events were reported in 5% of patients who applied 0.75% ruxolitinib cream and in 3% of patients who applied 1.5% ruxolitinib cream; none were serious. TEAEs led to discontinuation in 2% of patients in the 0.75% ruxolitinib cream group, and no patients in the 1.5% ruxolitinib cream group.
“The most common treatment adverse events were upper respiratory tract infections and nasopharyngitis,” Dr. Papp said. “When looking at exposure-adjusted adverse events, we see that there is a high degree of similarity between any of the TEAEs across all of the treatment groups in both studies. We also see that it was patients on the vehicle who experienced the greatest number of application-site reactions.”
Dr. Papp disclosed that he has received honoraria or clinical research grants as a consultant, speaker, scientific officer, advisory board member, and/or steering committee member for several pharmaceutical companies, including Incyte.
FROM REVOLUTIONIZING AD 2021