User login
Nonhealing Ulcer on the Lower Lip
Nonhealing Ulcer on the Lower Lip
THE DIAGNOSIS: Syphilis
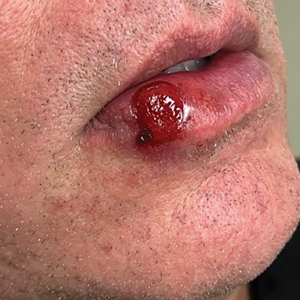
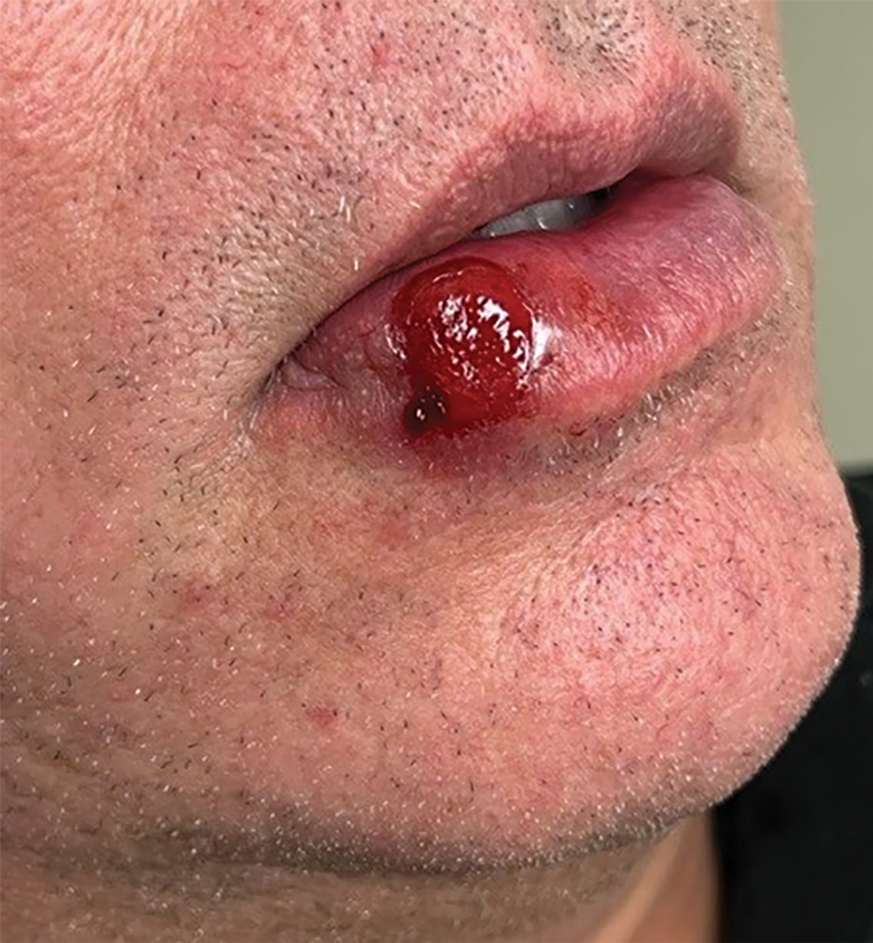
The differential diagnosis of oral lesions can be complex; in our patient, we considered conditions such as pyogenic granuloma, herpes simplex virus, and syphilis, despite the presence of pain. Immunohistochemical staining for spirochete antigens was positive, and serologic confirmation through a positive rapid plasma reagin (RPR) test confirmed the diagnosis of primary syphilis. The patient was promptly referred back to the primary care physician for treatment with intramuscular penicillin, leading to resolution of the lesion. At 3 months’ follow-up in our clinic, the lesion was fully resolved.
A primary syphilitic chancre is the initial lesion caused by Treponema pallidum, typically manifesting as a painless ulcer at the infection site, usually in the genital area; however, chancres also may manifest in other locations (eg, the anus or oral cavity) due to direct contact with infectious lesions on another individual. Our case represents an atypical presentation of an oral syphilitic chancre.
Syphilis is a sexually transmitted infection with various clinical manifestations. It is crucial to consider syphilis in the differential diagnosis of ulcerative lesions even when pain is present, especially in high-risk individuals such as those who engage in unprotected sex.1,2 Oral syphilitic chancres have been documented in the medical literature for more than a century, underscoring the importance of maintaining a high index of suspicion for diagnosis and a low threshold for obtaining an RPR test to facilitate early detection and treatment.2,3 Notably, the prevalence of syphilis is higher in men who have sex with men, particularly among those who engage in unprotected oral and anal sex. Increased screening and early treatment are essential to control the spread of disease within all populations. Doxycycline postexposure prophylaxis (doxyPEP) is used as a preventive measure for syphilis, chlamydia, and gonorrhea.4 This regimen consists of 200 mg of doxycycline taken within 24 hours but no later than 72 hours after unprotected anal, vaginal, or oral sex.
Our case highlights the importance of considering the differential diagnosis of oral ulcers, particularly in high-risk populations such as men who have sex with men. Prompt diagnosis, effective treatment, and preventive strategies such as doxyPEP are essential for controlling syphilis. Comprehensive patient education and regular follow-up appointments are critical components of successful management.
The United States has experienced a considerable rise in primary and congenital syphilis cases, with an 80% increase between 2018 and 2022.6 Serologic testing is the primary method for diagnosing, staging, and managing syphilis. Sexually active patients with suspected syphilis or unexplained symptoms should undergo testing. Prompt diagnosis and treatment can prevent systemic complications, including ocular involvement and permanent blindness.
Syphilis is transmitted through direct contact with a syphilitic ulcer or saliva or blood from an infected individual. Oral syphilitic ulcers can develop on the lips, tongue, oral mucosa, and tonsils. Chancres can range from a few millimeters to several centimeters, with an incubation period of 10 to 90 days (average, 21 days). The chancre lasts 3 to 6 weeks and heals spontaneously. Without treatment, primary syphilis can progress to secondary syphilis, characterized by a papulosquamous eruption and mucosal involvement, and potentially tertiary syphilis, which can affect the central nervous system, heart, bones, and skin.7
Immunocompromised patients, especially those diagnosed with HIV, face increased risks including altered clinical presentations (eg, multiple or deep chancres), delayed healing, overlapping stages of disease, and increased severity of organ involvement. All sexually active individuals should be screened for syphilis every 3 to 6 months, particularly those with unexplained oral ulcers.
Serologic testing is fundamental for syphilis diagnosis and management. Nontreponemal tests such as RPR and treponemal tests such as the fluorescent treponemal antibody absorption test provide comprehensive diagnostic information. Early diagnosis and empiric treatment are crucial in suspected cases. Ocular screening is recommended for suspected or confirmed syphilis cases.7
Management of syphilis includes treating all sexual partners and providing thorough patient education on the disease. Monitoring for the Jarisch-Herxheimer reaction—an acute febrile reaction following penicillin therapy—is important, especially in pregnant patients.5 Serologic evaluation at 6 and 12 months posttreatment is recommended, with more frequent evaluations if follow-up is uncertain, particularly for those with inconsistent access to health care or in whom reinfection is suspected. Guidelines from the Centers for Disease Control and Prevention advocate for intramuscular penicillin G benzathine as the preferred treatment, with specific dosing for adults and children.7 Due to the ongoing bicillin shortage, alternatives such as extencilline have temporarily been allowed for use in the United States.8
The rising incidence of syphilis in the United States underscores the critical need for enhanced public health initiatives focusing on education, screening, and early intervention. Comprehensive sexual education that includes information about syphilis and other sexually transmitted infections, proper use of prophylactic measures such as condoms, and the benefits of doxyPEP can considerably reduce transmission rates. Health care providers should routinely discuss these preventive measures with their patients, especially those in high-risk groups.
Our case highlights the importance of considering syphilis in the differential diagnosis of oral ulcers, particularly in high-risk populations. Timely diagnosis, effective treatment, and preventive measures such as doxyPEP are essential for managing and controlling syphilis. The rising incidence of syphilis in the United States warrants increased screening, patient education, and public health interventions to address this notable health challenge. The syphilis crisis calls for coordinated efforts from health care providers, public health officials, and community leaders to curb the spread of this infection and protect public health.
- Mayer KH, Traeger M, Marcus JL. Doxycycline postexposure prophylaxis and sexually transmitted infections. JAMA. 2023;330:1381-1382. doi:10.1001/jama.2023.16416
- Cossman JP, Fournier JB. Frequency of syphilis diagnoses by dermatologists. JAMA Dermatol. 2017;153:718-719. doi:10.1001 /jamadermatol.2017.0460
- Porterfield C, Brodell D, Dolohanty L, et al. Primary syphilis presenting as a chronic lip ulcer. Cureus. 2020;12:E7086. doi:10.7759 /cureus.7086
- Schamberg JF. An epidemic of chancres of the lip from kissing. JAMA. 1911;LVII:783-784. doi:10.1001/jama.1911.04260090005002
- Farmer TW. Jarisch-Herxheimer reaction in early syphilis. JAMA. 1948;138:480–485. doi:10.1001/jama.1948.02900070012003
- Winney A. Why is syphilis spiking in the U.S.? Johns Hopkins Bloomberg School of Public Health. Johns Hopkins Bloomberg School of Public Health. Published March 13, 2024. Accessed April 30, 2025. https://publichealth.jhu.edu/why-is-syphilis-spiking-in-the-us
- Koundanya VV, Tripathy K. Syphilis ocular manifestations. StatPearls Publishing; 2021. Updated August 25, 2023. Accessed May 6, 2025. https://www.ncbi.nlm.nih.gov/books/NBK558957/
- CDC. FDA announcement on availability of extencilline. National Center for HIV, Viral Hepatitis, STD, and Tuberculosis Prevention. Published July 19, 2024. Accessed April 30, 2025. https://www.cdc.gov/nchhstp/director-letters/extencilline-during-bicillin-l-a-shortage.html
THE DIAGNOSIS: Syphilis
The differential diagnosis of oral lesions can be complex; in our patient, we considered conditions such as pyogenic granuloma, herpes simplex virus, and syphilis, despite the presence of pain. Immunohistochemical staining for spirochete antigens was positive, and serologic confirmation through a positive rapid plasma reagin (RPR) test confirmed the diagnosis of primary syphilis. The patient was promptly referred back to the primary care physician for treatment with intramuscular penicillin, leading to resolution of the lesion. At 3 months’ follow-up in our clinic, the lesion was fully resolved.
A primary syphilitic chancre is the initial lesion caused by Treponema pallidum, typically manifesting as a painless ulcer at the infection site, usually in the genital area; however, chancres also may manifest in other locations (eg, the anus or oral cavity) due to direct contact with infectious lesions on another individual. Our case represents an atypical presentation of an oral syphilitic chancre.
Syphilis is a sexually transmitted infection with various clinical manifestations. It is crucial to consider syphilis in the differential diagnosis of ulcerative lesions even when pain is present, especially in high-risk individuals such as those who engage in unprotected sex.1,2 Oral syphilitic chancres have been documented in the medical literature for more than a century, underscoring the importance of maintaining a high index of suspicion for diagnosis and a low threshold for obtaining an RPR test to facilitate early detection and treatment.2,3 Notably, the prevalence of syphilis is higher in men who have sex with men, particularly among those who engage in unprotected oral and anal sex. Increased screening and early treatment are essential to control the spread of disease within all populations. Doxycycline postexposure prophylaxis (doxyPEP) is used as a preventive measure for syphilis, chlamydia, and gonorrhea.4 This regimen consists of 200 mg of doxycycline taken within 24 hours but no later than 72 hours after unprotected anal, vaginal, or oral sex.
Our case highlights the importance of considering the differential diagnosis of oral ulcers, particularly in high-risk populations such as men who have sex with men. Prompt diagnosis, effective treatment, and preventive strategies such as doxyPEP are essential for controlling syphilis. Comprehensive patient education and regular follow-up appointments are critical components of successful management.
The United States has experienced a considerable rise in primary and congenital syphilis cases, with an 80% increase between 2018 and 2022.6 Serologic testing is the primary method for diagnosing, staging, and managing syphilis. Sexually active patients with suspected syphilis or unexplained symptoms should undergo testing. Prompt diagnosis and treatment can prevent systemic complications, including ocular involvement and permanent blindness.
Syphilis is transmitted through direct contact with a syphilitic ulcer or saliva or blood from an infected individual. Oral syphilitic ulcers can develop on the lips, tongue, oral mucosa, and tonsils. Chancres can range from a few millimeters to several centimeters, with an incubation period of 10 to 90 days (average, 21 days). The chancre lasts 3 to 6 weeks and heals spontaneously. Without treatment, primary syphilis can progress to secondary syphilis, characterized by a papulosquamous eruption and mucosal involvement, and potentially tertiary syphilis, which can affect the central nervous system, heart, bones, and skin.7
Immunocompromised patients, especially those diagnosed with HIV, face increased risks including altered clinical presentations (eg, multiple or deep chancres), delayed healing, overlapping stages of disease, and increased severity of organ involvement. All sexually active individuals should be screened for syphilis every 3 to 6 months, particularly those with unexplained oral ulcers.
Serologic testing is fundamental for syphilis diagnosis and management. Nontreponemal tests such as RPR and treponemal tests such as the fluorescent treponemal antibody absorption test provide comprehensive diagnostic information. Early diagnosis and empiric treatment are crucial in suspected cases. Ocular screening is recommended for suspected or confirmed syphilis cases.7
Management of syphilis includes treating all sexual partners and providing thorough patient education on the disease. Monitoring for the Jarisch-Herxheimer reaction—an acute febrile reaction following penicillin therapy—is important, especially in pregnant patients.5 Serologic evaluation at 6 and 12 months posttreatment is recommended, with more frequent evaluations if follow-up is uncertain, particularly for those with inconsistent access to health care or in whom reinfection is suspected. Guidelines from the Centers for Disease Control and Prevention advocate for intramuscular penicillin G benzathine as the preferred treatment, with specific dosing for adults and children.7 Due to the ongoing bicillin shortage, alternatives such as extencilline have temporarily been allowed for use in the United States.8
The rising incidence of syphilis in the United States underscores the critical need for enhanced public health initiatives focusing on education, screening, and early intervention. Comprehensive sexual education that includes information about syphilis and other sexually transmitted infections, proper use of prophylactic measures such as condoms, and the benefits of doxyPEP can considerably reduce transmission rates. Health care providers should routinely discuss these preventive measures with their patients, especially those in high-risk groups.
Our case highlights the importance of considering syphilis in the differential diagnosis of oral ulcers, particularly in high-risk populations. Timely diagnosis, effective treatment, and preventive measures such as doxyPEP are essential for managing and controlling syphilis. The rising incidence of syphilis in the United States warrants increased screening, patient education, and public health interventions to address this notable health challenge. The syphilis crisis calls for coordinated efforts from health care providers, public health officials, and community leaders to curb the spread of this infection and protect public health.
THE DIAGNOSIS: Syphilis
The differential diagnosis of oral lesions can be complex; in our patient, we considered conditions such as pyogenic granuloma, herpes simplex virus, and syphilis, despite the presence of pain. Immunohistochemical staining for spirochete antigens was positive, and serologic confirmation through a positive rapid plasma reagin (RPR) test confirmed the diagnosis of primary syphilis. The patient was promptly referred back to the primary care physician for treatment with intramuscular penicillin, leading to resolution of the lesion. At 3 months’ follow-up in our clinic, the lesion was fully resolved.
A primary syphilitic chancre is the initial lesion caused by Treponema pallidum, typically manifesting as a painless ulcer at the infection site, usually in the genital area; however, chancres also may manifest in other locations (eg, the anus or oral cavity) due to direct contact with infectious lesions on another individual. Our case represents an atypical presentation of an oral syphilitic chancre.
Syphilis is a sexually transmitted infection with various clinical manifestations. It is crucial to consider syphilis in the differential diagnosis of ulcerative lesions even when pain is present, especially in high-risk individuals such as those who engage in unprotected sex.1,2 Oral syphilitic chancres have been documented in the medical literature for more than a century, underscoring the importance of maintaining a high index of suspicion for diagnosis and a low threshold for obtaining an RPR test to facilitate early detection and treatment.2,3 Notably, the prevalence of syphilis is higher in men who have sex with men, particularly among those who engage in unprotected oral and anal sex. Increased screening and early treatment are essential to control the spread of disease within all populations. Doxycycline postexposure prophylaxis (doxyPEP) is used as a preventive measure for syphilis, chlamydia, and gonorrhea.4 This regimen consists of 200 mg of doxycycline taken within 24 hours but no later than 72 hours after unprotected anal, vaginal, or oral sex.
Our case highlights the importance of considering the differential diagnosis of oral ulcers, particularly in high-risk populations such as men who have sex with men. Prompt diagnosis, effective treatment, and preventive strategies such as doxyPEP are essential for controlling syphilis. Comprehensive patient education and regular follow-up appointments are critical components of successful management.
The United States has experienced a considerable rise in primary and congenital syphilis cases, with an 80% increase between 2018 and 2022.6 Serologic testing is the primary method for diagnosing, staging, and managing syphilis. Sexually active patients with suspected syphilis or unexplained symptoms should undergo testing. Prompt diagnosis and treatment can prevent systemic complications, including ocular involvement and permanent blindness.
Syphilis is transmitted through direct contact with a syphilitic ulcer or saliva or blood from an infected individual. Oral syphilitic ulcers can develop on the lips, tongue, oral mucosa, and tonsils. Chancres can range from a few millimeters to several centimeters, with an incubation period of 10 to 90 days (average, 21 days). The chancre lasts 3 to 6 weeks and heals spontaneously. Without treatment, primary syphilis can progress to secondary syphilis, characterized by a papulosquamous eruption and mucosal involvement, and potentially tertiary syphilis, which can affect the central nervous system, heart, bones, and skin.7
Immunocompromised patients, especially those diagnosed with HIV, face increased risks including altered clinical presentations (eg, multiple or deep chancres), delayed healing, overlapping stages of disease, and increased severity of organ involvement. All sexually active individuals should be screened for syphilis every 3 to 6 months, particularly those with unexplained oral ulcers.
Serologic testing is fundamental for syphilis diagnosis and management. Nontreponemal tests such as RPR and treponemal tests such as the fluorescent treponemal antibody absorption test provide comprehensive diagnostic information. Early diagnosis and empiric treatment are crucial in suspected cases. Ocular screening is recommended for suspected or confirmed syphilis cases.7
Management of syphilis includes treating all sexual partners and providing thorough patient education on the disease. Monitoring for the Jarisch-Herxheimer reaction—an acute febrile reaction following penicillin therapy—is important, especially in pregnant patients.5 Serologic evaluation at 6 and 12 months posttreatment is recommended, with more frequent evaluations if follow-up is uncertain, particularly for those with inconsistent access to health care or in whom reinfection is suspected. Guidelines from the Centers for Disease Control and Prevention advocate for intramuscular penicillin G benzathine as the preferred treatment, with specific dosing for adults and children.7 Due to the ongoing bicillin shortage, alternatives such as extencilline have temporarily been allowed for use in the United States.8
The rising incidence of syphilis in the United States underscores the critical need for enhanced public health initiatives focusing on education, screening, and early intervention. Comprehensive sexual education that includes information about syphilis and other sexually transmitted infections, proper use of prophylactic measures such as condoms, and the benefits of doxyPEP can considerably reduce transmission rates. Health care providers should routinely discuss these preventive measures with their patients, especially those in high-risk groups.
Our case highlights the importance of considering syphilis in the differential diagnosis of oral ulcers, particularly in high-risk populations. Timely diagnosis, effective treatment, and preventive measures such as doxyPEP are essential for managing and controlling syphilis. The rising incidence of syphilis in the United States warrants increased screening, patient education, and public health interventions to address this notable health challenge. The syphilis crisis calls for coordinated efforts from health care providers, public health officials, and community leaders to curb the spread of this infection and protect public health.
- Mayer KH, Traeger M, Marcus JL. Doxycycline postexposure prophylaxis and sexually transmitted infections. JAMA. 2023;330:1381-1382. doi:10.1001/jama.2023.16416
- Cossman JP, Fournier JB. Frequency of syphilis diagnoses by dermatologists. JAMA Dermatol. 2017;153:718-719. doi:10.1001 /jamadermatol.2017.0460
- Porterfield C, Brodell D, Dolohanty L, et al. Primary syphilis presenting as a chronic lip ulcer. Cureus. 2020;12:E7086. doi:10.7759 /cureus.7086
- Schamberg JF. An epidemic of chancres of the lip from kissing. JAMA. 1911;LVII:783-784. doi:10.1001/jama.1911.04260090005002
- Farmer TW. Jarisch-Herxheimer reaction in early syphilis. JAMA. 1948;138:480–485. doi:10.1001/jama.1948.02900070012003
- Winney A. Why is syphilis spiking in the U.S.? Johns Hopkins Bloomberg School of Public Health. Johns Hopkins Bloomberg School of Public Health. Published March 13, 2024. Accessed April 30, 2025. https://publichealth.jhu.edu/why-is-syphilis-spiking-in-the-us
- Koundanya VV, Tripathy K. Syphilis ocular manifestations. StatPearls Publishing; 2021. Updated August 25, 2023. Accessed May 6, 2025. https://www.ncbi.nlm.nih.gov/books/NBK558957/
- CDC. FDA announcement on availability of extencilline. National Center for HIV, Viral Hepatitis, STD, and Tuberculosis Prevention. Published July 19, 2024. Accessed April 30, 2025. https://www.cdc.gov/nchhstp/director-letters/extencilline-during-bicillin-l-a-shortage.html
- Mayer KH, Traeger M, Marcus JL. Doxycycline postexposure prophylaxis and sexually transmitted infections. JAMA. 2023;330:1381-1382. doi:10.1001/jama.2023.16416
- Cossman JP, Fournier JB. Frequency of syphilis diagnoses by dermatologists. JAMA Dermatol. 2017;153:718-719. doi:10.1001 /jamadermatol.2017.0460
- Porterfield C, Brodell D, Dolohanty L, et al. Primary syphilis presenting as a chronic lip ulcer. Cureus. 2020;12:E7086. doi:10.7759 /cureus.7086
- Schamberg JF. An epidemic of chancres of the lip from kissing. JAMA. 1911;LVII:783-784. doi:10.1001/jama.1911.04260090005002
- Farmer TW. Jarisch-Herxheimer reaction in early syphilis. JAMA. 1948;138:480–485. doi:10.1001/jama.1948.02900070012003
- Winney A. Why is syphilis spiking in the U.S.? Johns Hopkins Bloomberg School of Public Health. Johns Hopkins Bloomberg School of Public Health. Published March 13, 2024. Accessed April 30, 2025. https://publichealth.jhu.edu/why-is-syphilis-spiking-in-the-us
- Koundanya VV, Tripathy K. Syphilis ocular manifestations. StatPearls Publishing; 2021. Updated August 25, 2023. Accessed May 6, 2025. https://www.ncbi.nlm.nih.gov/books/NBK558957/
- CDC. FDA announcement on availability of extencilline. National Center for HIV, Viral Hepatitis, STD, and Tuberculosis Prevention. Published July 19, 2024. Accessed April 30, 2025. https://www.cdc.gov/nchhstp/director-letters/extencilline-during-bicillin-l-a-shortage.html
Nonhealing Ulcer on the Lower Lip
Nonhealing Ulcer on the Lower Lip
A 54-year-old HIV-negative man with a history of having sex with men presented to his primary care physician with an ulcer on the lower lip of 3 weeks’ duration. The patient reported that the lesion had appeared as a typical cold sore with pain in the area. A 9-day course of oral valacyclovir prescribed by the primary care physician provided no relief or improvement. A 2-mm punch biopsy was performed.
The Rise of Antifungal-Resistant Dermatophyte Infections: What Dermatologists Need to Know
The Rise of Antifungal-Resistant Dermatophyte Infections: What Dermatologists Need to Know
Worldwide, it is estimated that up to 1 in 5 individuals will experience a dermatophyte infection (commonly called ringworm or tinea infection) in their lifetime.1 Historically, dermatophyte infections have been considered relatively minor conditions usually treated with short courses of topical antifungals.2 Oral antifungals historically were needed only for patients with nail or hair shaft infections or extensive cutaneous fungal infections, which typically occurred in immunosuppressed patients.2 However, the landscape is changing rapidly due to the global emergence of severe dermatophyte infections that frequently are resistant to first-line antifungal medications.3-5 In this article, we aimed to review the epidemiology of emerging dermatophyte infections and provide dermatologists with information needed for effective diagnosis and management.
Emergence of Trichophyton indotineae
In recent decades, public health officials and dermatologists have noted with concern the spread of the recently emerged dermatophyte species Trichophyton indotineae in South Asia.3,6 This species (previously known as Trichophyton mentagrophytes genotype VIII) usually is transmitted from person to person, either through direct skin-to-skin contact or by fomites.4,6 Potential sexual transmission of T indotineae infections also has been reported,7 and it is possible that animals may serve as reservoirs for this pathogen, although there are no known reports of direct spread from animals to humans.8,9 Major outbreaks of T indotineae are ongoing in South Asia, and cases have been documented in 6 continents.10-12 In the United States, most but not all cases have occurred in immigrants from or recently returned travelers to South Asia.6,13 The emergence and spread of T indotineae is hypothesized to be promoted by the misuse and overuse of topical antifungal products, particularly those containing combinations of potent corticosteroids with other antimicrobial drugs.14,15
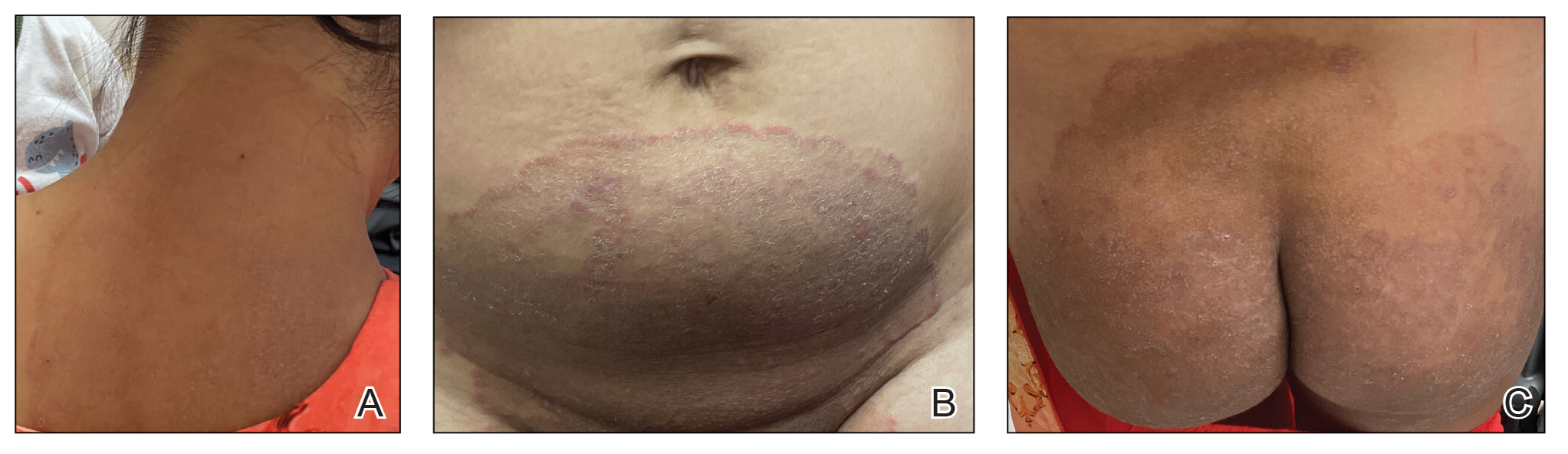
Cutaneous manifestations of T indotineae infections tend to cover large body surface areas, recur frequently, and pose substantial treatment challenges.6,13,16 Several clinical presentations have been documented, including erythematous, scaly concentric plaques; papulosquamous lesions; pustular forms; and corticosteroid-modified disease (Figure 1).6,16 Affected patients seldom are immunocompromised and often have a history of multiple failed courses of topical or oral antifungals, including oral terbinafine.13 Many also have been prescribed topical corticosteroids or have used over-the-counter topical corticosteroids, which worsen the rash.17
Direct microscopy with potassium hydroxide could be used to confirm the diagnosis of dermatophyte infection, but it does not distinguish T indotineae from other dermatophyte species.2,6 Importantly, culture-based testing usually will misidentify T indotineae as other Trichophyton species such as the more common T mentagrophytes or Trichophyton interdigitale. Definitive identification of T indotineae requires advanced molecular techniques that are available only at select laboratories.6 Unfortunately, availability of such testing is limited (Table), and results may take several weeks; therefore, it is suggested that dermatologists who suspect T indotineae infections based on the patient’s history and clinical presentation begin antifungal treatment after confirmation of dermatophyte infection but not wait for definitive confirmation of the causative organism.16
Itraconazole is considered the first-line therapy for T indotineae infection, as terbinafine usually is ineffective due to mutations in the squalene epoxidase gene.16 Dermatologists should be aware that itraconazole is available in different formulations that can affect absorption. The oral solution has greater bioavailability and should be taken on an empty stomach, whereas the capsules are required to be taken with food for effective absorption; the capsules also should be taken with an acidic beverage such as orange juice. Dermatologists should carefully assess for drug-drug interactions when prescribing itraconazole, given its extensive interaction profile with numerous other medications. Patients may require treatment with itraconazole (100 mg/d or 200 mg/d) for a minimum of 6 to 8 weeks until complete clearance has been achieved and ideally a negative potassium hydroxide preparation of skin scrapings has been obtained. A longer treatment period (eg, ≥3 months) frequently is needed, and relapses are common.6,16,18 Regular follow-up is needed to monitor for infection clearance and recurrences. It is important to note that cases of itraconazole resistance have been reported, although this currently appears to be uncommon.19,20
Other Emerging Dermatophytes to Watch
Trichophyton rubrum is the most common cause of dermatophyte infections among humans,21 and cases of terbinafine-resistant T rubrum infections have been reported increasingly in the United States and Canada.5,22-24 Onychomycosis caused by terbinafine-resistant T rubrum has been documented, and patients may have infections that do not respond to terbinafine given at the standard dose and duration.22,23 Case reports have indicated successful treatment using itraconazole 200 mg/d and posaconazole 300 mg/d.5,23
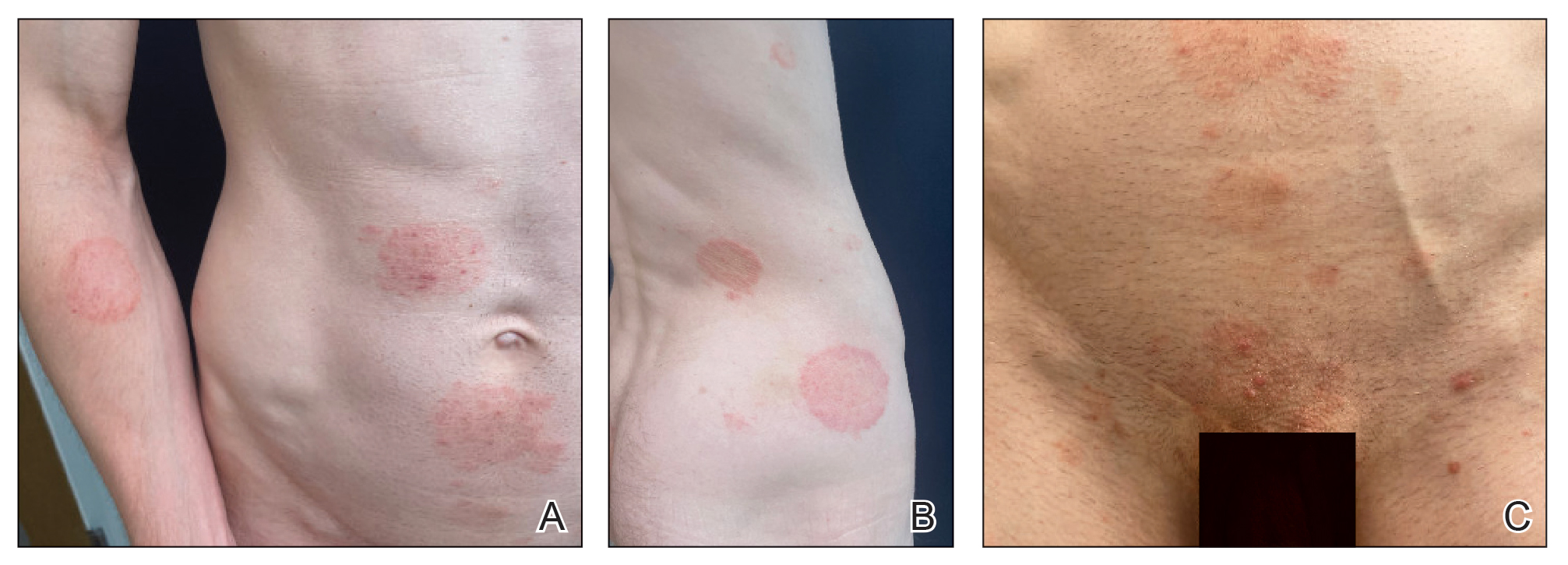
Trichophyton mentagrophytes genotype VII (TMVII) is an emerging dermatophyte that recently has been reported as a cause of sexually transmitted dermatophyte infections in Europe and the United States primarily affecting men who have sex with men.25-27 Patients may present with pruritic, annular, scaly patches and plaques involving the trunk, groin, genital region, or face (Figure 2). Although closely related to T indotineae, TMVII differs in that it more often affects the genital region, generally is susceptible to terbinafine, and in the United States and Europe usually is not related to travel or immigration involving South Asia.26 Although TMVII has not been associated with antifungal resistance, awareness among dermatologists is important because patients may experience inflamed, painful, and persistent rashes that can lead to secondary bacterial infection or scarring, and physicians might mistake it for mimics including eczema or psoriasis.25,26
Importance of Judicious Antifungal Use
Optimizing the use of antifungals is critical to improving patient outcomes and preserving available treatment options.28,29 A retrospective analysis of commercial health insurance data estimated that topical antifungal prescriptions were potentially unnecessary for more than half of the more than 560,000 patients who were prescribed these medications in 2023. In this study, it also was observed that only 16% of patients prescribed a topical antifungal had received diagnostic testing, with low rates across specialties.30 This is concerning because even among board-certified dermatologists, incorrect diagnosis of suspected fungal skin infections can occur; in one survey-based study of board-certified dermatologists who were presented with dermatomycosis images, respondents categorized cases with greater than 75% accuracy in only 31% (4/13) of instances.31 Clotrimazole-betamethasone is among the most commonly prescribed topical antifungals in the United States,14,32 and 2 recent retrospective analyses highlighted that the majority of patients prescribed this medication did not receive any fungal diagnostic testing.33,34
Final Thoughts
In an era of emerging antifungal-resistant dermatophyte infections, it is important for dermatologists to educate nondermatologists about the importance of using diagnostic testing for suspected dermatophyte infections.14,28 Dermatologists also can educate nondermatologist colleagues on the importance of avoiding the use of topical combination antifungal/corticosteroid medications and referring for dermatologic evaluation when diagnoses are uncertain.33,34 Strategies for education by dermatologists could include giving workshops, creating educational materials, and fostering open communication about optimal treatment practices and referral parameters for suspected dermatophyte infections.
- Noble SL, Forbes RC, Stamm PL. Diagnosis and management of common tinea infections. Am Fam Physician. 1998;58:163-174, 177-168.
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
- Uhrlaß S, Verma SB, Gräser Y, et al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: I. epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021;87:154-175.
- Chen E, Ghannoum M, Elewski BE. Treatment]resistant tinea corporis, a potential public health issue. Br J Dermatol. 2021;184:164-165.
- Caplan AS. Notes from the field: first reported US cases of tinea caused by Trichophyton indotineae—New York City, December 2021–March 2023. MMWR Morbidity and Mortality Weekly Report. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Spivack S, Gold JA, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807.
- Jabet A, Brun S, Normand AC, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233.
- Thakur S, Spruijtenburg B, Abhishek, et al. Whole genome sequence analysis of terbinafine resistant and susceptible Trichophyton isolates from human and animal origin. Mycopathologia. 2025;190:13.
- Lockhart SR, Chowdhary A, Gold JA. The rapid emergence of antifungal-resistant human-pathogenic fungi. Nat Rev Microbiol. 2023;21:818-832.
- Mosam A, Shuping L, Naicker S, et al. A case of antifungal-resistant ringworm infection in KwaZulu-Natal Province, South Africa, caused by Trichophyton indotineae. Public Health Bulletin South Africa. Accessed April 4, 2025. https://www.phbsa.ac.za/wp-content/uploads/2023/12PHBSA-Ringworm-Article-2023.pdf
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:E0056223
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709. doi:10.1001/jamadermatol.2024.1126
- Benedict K. Topical antifungal prescribing for Medicare Part D beneficiaries—United States, 2021. MMWR Morb Mortal Wkly Rep. 2024;73:1-5.
- Verma SB. Emergence of recalcitrant dermatophytosis in India. Lancet Infect Dis. 2018;18:718-719.
- Khurana A, Sharath S, Sardana K, et al. Clinico-mycological and therapeutic updates on cutaneous dermatophytic infections in the era of Trichophyton indotineae. J Am Acad Dermatol. 2024;91:315-323. doi:10.1016/j.jaad.2024.03.024
- Verma S. Steroid modified tinea. BMJ. 2017;356:j973.
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278.
- Burmester A, Hipler UC, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180.
- Bhuiyan MSI, Verma SB, Illigner GM, et al. Trichophyton mentagrophytes ITS genotype VIII/Trichophyton indotineae infection and antifungal resistance in Bangladesh. J Fungi (Basel). 2024;10:768. doi:10.3390 /jof10110768
- Hay RJ. Chapter 82: superficial mycoses. In: Ryan ET, Hill DR, Solomon T, et al, eds. Hunter’s Tropical Medicine and Emerging Infectious Diseases. 10th ed. Elsevier; 2020:648-652.
- Gupta AK, Cooper EA, Wang T, et al. Detection of squalene epoxidase mutations in United States patients with onychomycosis: implications for management. J Invest Dermatol. 2023;143:2476-2483.E2477.
- Hwang JK, Bakotic WL, Gold JA, et al. Isolation of terbinafine-resistant Trichophyton rubrum from onychomycosis patients who failed treatment at an academic center in New York, United States. J Fungi. 2023;9:710.
- Gu D, Hatch M, Ghannoum M, et al. Treatment-resistant dermatophytosis: a representative case highlighting an emerging public health threat. JAAD Case Rep. 2020;6:1153-1155.
- Jabet A, Dellière S, Seang S, et al. Sexually transmitted Trichophyton mentagrophytes genotype VII infection among men who have sex with men. Emerg Infect Dis. 2023;29:1411-1414.
- Zucker J, Caplan AS, Gunaratne SH, et al. Notes from the field: Trichophyton mentagrophytes genotype VII—New York City, April-July 2024. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Caplan AS, Gold JA, Smith DJ, et al. Improving antifungal stewardship in dermatology in an era of emerging dermatophyte resistance. JAAD International. 2024;15:168-169.
- Elewski B. A call for antifungal stewardship. Br J Dermatol. 2020; 183:798-799.
- Gold JAW, Benedict K, Caplan AS, et al. High rates of potentially unnecessary topical antifungal prescribing in a large commercial health insurance claims database, United States. J Am Acad Dermatol. 2025:S0190-9622(25)00098-2. doi:10.1016/j.jaad.2025.01.022
- Yadgar RJ, Bhatia N, Friedman A. Cutaneous fungal infections are commonly misdiagnosed: a survey-based study. J Am Acad Dermatol. 2017;76:562-563.
- Flint ND, Rhoads JLW, Carlisle R, et al. The continued inappropriate use and overuse of combination topical clotrimazole-betamethasone. Dermatol Online J. 2021;27. doi:10.5070/D327854686
- Currie DW, Caplan AS, Benedict K, et al. Prescribing of clotrimazolebetamethasone dipropionate, a topical combination corticosteroidantifungal product, for Medicare part D beneficiaries, United States, 2016–2022. Antimicrob Steward Healthc Epidemiol. 2024;4:E174.
- Gold JA, Caplan AS, Benedict K, et al. Clotrimazole-betamethasone dipropionate prescribing for nonfungal skin conditions. JAMA Network Open. 2024;7:E2411721-E2411721.
Worldwide, it is estimated that up to 1 in 5 individuals will experience a dermatophyte infection (commonly called ringworm or tinea infection) in their lifetime.1 Historically, dermatophyte infections have been considered relatively minor conditions usually treated with short courses of topical antifungals.2 Oral antifungals historically were needed only for patients with nail or hair shaft infections or extensive cutaneous fungal infections, which typically occurred in immunosuppressed patients.2 However, the landscape is changing rapidly due to the global emergence of severe dermatophyte infections that frequently are resistant to first-line antifungal medications.3-5 In this article, we aimed to review the epidemiology of emerging dermatophyte infections and provide dermatologists with information needed for effective diagnosis and management.
Emergence of Trichophyton indotineae
In recent decades, public health officials and dermatologists have noted with concern the spread of the recently emerged dermatophyte species Trichophyton indotineae in South Asia.3,6 This species (previously known as Trichophyton mentagrophytes genotype VIII) usually is transmitted from person to person, either through direct skin-to-skin contact or by fomites.4,6 Potential sexual transmission of T indotineae infections also has been reported,7 and it is possible that animals may serve as reservoirs for this pathogen, although there are no known reports of direct spread from animals to humans.8,9 Major outbreaks of T indotineae are ongoing in South Asia, and cases have been documented in 6 continents.10-12 In the United States, most but not all cases have occurred in immigrants from or recently returned travelers to South Asia.6,13 The emergence and spread of T indotineae is hypothesized to be promoted by the misuse and overuse of topical antifungal products, particularly those containing combinations of potent corticosteroids with other antimicrobial drugs.14,15
Cutaneous manifestations of T indotineae infections tend to cover large body surface areas, recur frequently, and pose substantial treatment challenges.6,13,16 Several clinical presentations have been documented, including erythematous, scaly concentric plaques; papulosquamous lesions; pustular forms; and corticosteroid-modified disease (Figure 1).6,16 Affected patients seldom are immunocompromised and often have a history of multiple failed courses of topical or oral antifungals, including oral terbinafine.13 Many also have been prescribed topical corticosteroids or have used over-the-counter topical corticosteroids, which worsen the rash.17

Direct microscopy with potassium hydroxide could be used to confirm the diagnosis of dermatophyte infection, but it does not distinguish T indotineae from other dermatophyte species.2,6 Importantly, culture-based testing usually will misidentify T indotineae as other Trichophyton species such as the more common T mentagrophytes or Trichophyton interdigitale. Definitive identification of T indotineae requires advanced molecular techniques that are available only at select laboratories.6 Unfortunately, availability of such testing is limited (Table), and results may take several weeks; therefore, it is suggested that dermatologists who suspect T indotineae infections based on the patient’s history and clinical presentation begin antifungal treatment after confirmation of dermatophyte infection but not wait for definitive confirmation of the causative organism.16

Itraconazole is considered the first-line therapy for T indotineae infection, as terbinafine usually is ineffective due to mutations in the squalene epoxidase gene.16 Dermatologists should be aware that itraconazole is available in different formulations that can affect absorption. The oral solution has greater bioavailability and should be taken on an empty stomach, whereas the capsules are required to be taken with food for effective absorption; the capsules also should be taken with an acidic beverage such as orange juice. Dermatologists should carefully assess for drug-drug interactions when prescribing itraconazole, given its extensive interaction profile with numerous other medications. Patients may require treatment with itraconazole (100 mg/d or 200 mg/d) for a minimum of 6 to 8 weeks until complete clearance has been achieved and ideally a negative potassium hydroxide preparation of skin scrapings has been obtained. A longer treatment period (eg, ≥3 months) frequently is needed, and relapses are common.6,16,18 Regular follow-up is needed to monitor for infection clearance and recurrences. It is important to note that cases of itraconazole resistance have been reported, although this currently appears to be uncommon.19,20
Other Emerging Dermatophytes to Watch
Trichophyton rubrum is the most common cause of dermatophyte infections among humans,21 and cases of terbinafine-resistant T rubrum infections have been reported increasingly in the United States and Canada.5,22-24 Onychomycosis caused by terbinafine-resistant T rubrum has been documented, and patients may have infections that do not respond to terbinafine given at the standard dose and duration.22,23 Case reports have indicated successful treatment using itraconazole 200 mg/d and posaconazole 300 mg/d.5,23
Trichophyton mentagrophytes genotype VII (TMVII) is an emerging dermatophyte that recently has been reported as a cause of sexually transmitted dermatophyte infections in Europe and the United States primarily affecting men who have sex with men.25-27 Patients may present with pruritic, annular, scaly patches and plaques involving the trunk, groin, genital region, or face (Figure 2). Although closely related to T indotineae, TMVII differs in that it more often affects the genital region, generally is susceptible to terbinafine, and in the United States and Europe usually is not related to travel or immigration involving South Asia.26 Although TMVII has not been associated with antifungal resistance, awareness among dermatologists is important because patients may experience inflamed, painful, and persistent rashes that can lead to secondary bacterial infection or scarring, and physicians might mistake it for mimics including eczema or psoriasis.25,26

Importance of Judicious Antifungal Use
Optimizing the use of antifungals is critical to improving patient outcomes and preserving available treatment options.28,29 A retrospective analysis of commercial health insurance data estimated that topical antifungal prescriptions were potentially unnecessary for more than half of the more than 560,000 patients who were prescribed these medications in 2023. In this study, it also was observed that only 16% of patients prescribed a topical antifungal had received diagnostic testing, with low rates across specialties.30 This is concerning because even among board-certified dermatologists, incorrect diagnosis of suspected fungal skin infections can occur; in one survey-based study of board-certified dermatologists who were presented with dermatomycosis images, respondents categorized cases with greater than 75% accuracy in only 31% (4/13) of instances.31 Clotrimazole-betamethasone is among the most commonly prescribed topical antifungals in the United States,14,32 and 2 recent retrospective analyses highlighted that the majority of patients prescribed this medication did not receive any fungal diagnostic testing.33,34
Final Thoughts
In an era of emerging antifungal-resistant dermatophyte infections, it is important for dermatologists to educate nondermatologists about the importance of using diagnostic testing for suspected dermatophyte infections.14,28 Dermatologists also can educate nondermatologist colleagues on the importance of avoiding the use of topical combination antifungal/corticosteroid medications and referring for dermatologic evaluation when diagnoses are uncertain.33,34 Strategies for education by dermatologists could include giving workshops, creating educational materials, and fostering open communication about optimal treatment practices and referral parameters for suspected dermatophyte infections.
Worldwide, it is estimated that up to 1 in 5 individuals will experience a dermatophyte infection (commonly called ringworm or tinea infection) in their lifetime.1 Historically, dermatophyte infections have been considered relatively minor conditions usually treated with short courses of topical antifungals.2 Oral antifungals historically were needed only for patients with nail or hair shaft infections or extensive cutaneous fungal infections, which typically occurred in immunosuppressed patients.2 However, the landscape is changing rapidly due to the global emergence of severe dermatophyte infections that frequently are resistant to first-line antifungal medications.3-5 In this article, we aimed to review the epidemiology of emerging dermatophyte infections and provide dermatologists with information needed for effective diagnosis and management.
Emergence of Trichophyton indotineae
In recent decades, public health officials and dermatologists have noted with concern the spread of the recently emerged dermatophyte species Trichophyton indotineae in South Asia.3,6 This species (previously known as Trichophyton mentagrophytes genotype VIII) usually is transmitted from person to person, either through direct skin-to-skin contact or by fomites.4,6 Potential sexual transmission of T indotineae infections also has been reported,7 and it is possible that animals may serve as reservoirs for this pathogen, although there are no known reports of direct spread from animals to humans.8,9 Major outbreaks of T indotineae are ongoing in South Asia, and cases have been documented in 6 continents.10-12 In the United States, most but not all cases have occurred in immigrants from or recently returned travelers to South Asia.6,13 The emergence and spread of T indotineae is hypothesized to be promoted by the misuse and overuse of topical antifungal products, particularly those containing combinations of potent corticosteroids with other antimicrobial drugs.14,15
Cutaneous manifestations of T indotineae infections tend to cover large body surface areas, recur frequently, and pose substantial treatment challenges.6,13,16 Several clinical presentations have been documented, including erythematous, scaly concentric plaques; papulosquamous lesions; pustular forms; and corticosteroid-modified disease (Figure 1).6,16 Affected patients seldom are immunocompromised and often have a history of multiple failed courses of topical or oral antifungals, including oral terbinafine.13 Many also have been prescribed topical corticosteroids or have used over-the-counter topical corticosteroids, which worsen the rash.17

Direct microscopy with potassium hydroxide could be used to confirm the diagnosis of dermatophyte infection, but it does not distinguish T indotineae from other dermatophyte species.2,6 Importantly, culture-based testing usually will misidentify T indotineae as other Trichophyton species such as the more common T mentagrophytes or Trichophyton interdigitale. Definitive identification of T indotineae requires advanced molecular techniques that are available only at select laboratories.6 Unfortunately, availability of such testing is limited (Table), and results may take several weeks; therefore, it is suggested that dermatologists who suspect T indotineae infections based on the patient’s history and clinical presentation begin antifungal treatment after confirmation of dermatophyte infection but not wait for definitive confirmation of the causative organism.16

Itraconazole is considered the first-line therapy for T indotineae infection, as terbinafine usually is ineffective due to mutations in the squalene epoxidase gene.16 Dermatologists should be aware that itraconazole is available in different formulations that can affect absorption. The oral solution has greater bioavailability and should be taken on an empty stomach, whereas the capsules are required to be taken with food for effective absorption; the capsules also should be taken with an acidic beverage such as orange juice. Dermatologists should carefully assess for drug-drug interactions when prescribing itraconazole, given its extensive interaction profile with numerous other medications. Patients may require treatment with itraconazole (100 mg/d or 200 mg/d) for a minimum of 6 to 8 weeks until complete clearance has been achieved and ideally a negative potassium hydroxide preparation of skin scrapings has been obtained. A longer treatment period (eg, ≥3 months) frequently is needed, and relapses are common.6,16,18 Regular follow-up is needed to monitor for infection clearance and recurrences. It is important to note that cases of itraconazole resistance have been reported, although this currently appears to be uncommon.19,20
Other Emerging Dermatophytes to Watch
Trichophyton rubrum is the most common cause of dermatophyte infections among humans,21 and cases of terbinafine-resistant T rubrum infections have been reported increasingly in the United States and Canada.5,22-24 Onychomycosis caused by terbinafine-resistant T rubrum has been documented, and patients may have infections that do not respond to terbinafine given at the standard dose and duration.22,23 Case reports have indicated successful treatment using itraconazole 200 mg/d and posaconazole 300 mg/d.5,23
Trichophyton mentagrophytes genotype VII (TMVII) is an emerging dermatophyte that recently has been reported as a cause of sexually transmitted dermatophyte infections in Europe and the United States primarily affecting men who have sex with men.25-27 Patients may present with pruritic, annular, scaly patches and plaques involving the trunk, groin, genital region, or face (Figure 2). Although closely related to T indotineae, TMVII differs in that it more often affects the genital region, generally is susceptible to terbinafine, and in the United States and Europe usually is not related to travel or immigration involving South Asia.26 Although TMVII has not been associated with antifungal resistance, awareness among dermatologists is important because patients may experience inflamed, painful, and persistent rashes that can lead to secondary bacterial infection or scarring, and physicians might mistake it for mimics including eczema or psoriasis.25,26

Importance of Judicious Antifungal Use
Optimizing the use of antifungals is critical to improving patient outcomes and preserving available treatment options.28,29 A retrospective analysis of commercial health insurance data estimated that topical antifungal prescriptions were potentially unnecessary for more than half of the more than 560,000 patients who were prescribed these medications in 2023. In this study, it also was observed that only 16% of patients prescribed a topical antifungal had received diagnostic testing, with low rates across specialties.30 This is concerning because even among board-certified dermatologists, incorrect diagnosis of suspected fungal skin infections can occur; in one survey-based study of board-certified dermatologists who were presented with dermatomycosis images, respondents categorized cases with greater than 75% accuracy in only 31% (4/13) of instances.31 Clotrimazole-betamethasone is among the most commonly prescribed topical antifungals in the United States,14,32 and 2 recent retrospective analyses highlighted that the majority of patients prescribed this medication did not receive any fungal diagnostic testing.33,34
Final Thoughts
In an era of emerging antifungal-resistant dermatophyte infections, it is important for dermatologists to educate nondermatologists about the importance of using diagnostic testing for suspected dermatophyte infections.14,28 Dermatologists also can educate nondermatologist colleagues on the importance of avoiding the use of topical combination antifungal/corticosteroid medications and referring for dermatologic evaluation when diagnoses are uncertain.33,34 Strategies for education by dermatologists could include giving workshops, creating educational materials, and fostering open communication about optimal treatment practices and referral parameters for suspected dermatophyte infections.
- Noble SL, Forbes RC, Stamm PL. Diagnosis and management of common tinea infections. Am Fam Physician. 1998;58:163-174, 177-168.
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
- Uhrlaß S, Verma SB, Gräser Y, et al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: I. epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021;87:154-175.
- Chen E, Ghannoum M, Elewski BE. Treatment]resistant tinea corporis, a potential public health issue. Br J Dermatol. 2021;184:164-165.
- Caplan AS. Notes from the field: first reported US cases of tinea caused by Trichophyton indotineae—New York City, December 2021–March 2023. MMWR Morbidity and Mortality Weekly Report. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Spivack S, Gold JA, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807.
- Jabet A, Brun S, Normand AC, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233.
- Thakur S, Spruijtenburg B, Abhishek, et al. Whole genome sequence analysis of terbinafine resistant and susceptible Trichophyton isolates from human and animal origin. Mycopathologia. 2025;190:13.
- Lockhart SR, Chowdhary A, Gold JA. The rapid emergence of antifungal-resistant human-pathogenic fungi. Nat Rev Microbiol. 2023;21:818-832.
- Mosam A, Shuping L, Naicker S, et al. A case of antifungal-resistant ringworm infection in KwaZulu-Natal Province, South Africa, caused by Trichophyton indotineae. Public Health Bulletin South Africa. Accessed April 4, 2025. https://www.phbsa.ac.za/wp-content/uploads/2023/12PHBSA-Ringworm-Article-2023.pdf
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:E0056223
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709. doi:10.1001/jamadermatol.2024.1126
- Benedict K. Topical antifungal prescribing for Medicare Part D beneficiaries—United States, 2021. MMWR Morb Mortal Wkly Rep. 2024;73:1-5.
- Verma SB. Emergence of recalcitrant dermatophytosis in India. Lancet Infect Dis. 2018;18:718-719.
- Khurana A, Sharath S, Sardana K, et al. Clinico-mycological and therapeutic updates on cutaneous dermatophytic infections in the era of Trichophyton indotineae. J Am Acad Dermatol. 2024;91:315-323. doi:10.1016/j.jaad.2024.03.024
- Verma S. Steroid modified tinea. BMJ. 2017;356:j973.
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278.
- Burmester A, Hipler UC, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180.
- Bhuiyan MSI, Verma SB, Illigner GM, et al. Trichophyton mentagrophytes ITS genotype VIII/Trichophyton indotineae infection and antifungal resistance in Bangladesh. J Fungi (Basel). 2024;10:768. doi:10.3390 /jof10110768
- Hay RJ. Chapter 82: superficial mycoses. In: Ryan ET, Hill DR, Solomon T, et al, eds. Hunter’s Tropical Medicine and Emerging Infectious Diseases. 10th ed. Elsevier; 2020:648-652.
- Gupta AK, Cooper EA, Wang T, et al. Detection of squalene epoxidase mutations in United States patients with onychomycosis: implications for management. J Invest Dermatol. 2023;143:2476-2483.E2477.
- Hwang JK, Bakotic WL, Gold JA, et al. Isolation of terbinafine-resistant Trichophyton rubrum from onychomycosis patients who failed treatment at an academic center in New York, United States. J Fungi. 2023;9:710.
- Gu D, Hatch M, Ghannoum M, et al. Treatment-resistant dermatophytosis: a representative case highlighting an emerging public health threat. JAAD Case Rep. 2020;6:1153-1155.
- Jabet A, Dellière S, Seang S, et al. Sexually transmitted Trichophyton mentagrophytes genotype VII infection among men who have sex with men. Emerg Infect Dis. 2023;29:1411-1414.
- Zucker J, Caplan AS, Gunaratne SH, et al. Notes from the field: Trichophyton mentagrophytes genotype VII—New York City, April-July 2024. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Caplan AS, Gold JA, Smith DJ, et al. Improving antifungal stewardship in dermatology in an era of emerging dermatophyte resistance. JAAD International. 2024;15:168-169.
- Elewski B. A call for antifungal stewardship. Br J Dermatol. 2020; 183:798-799.
- Gold JAW, Benedict K, Caplan AS, et al. High rates of potentially unnecessary topical antifungal prescribing in a large commercial health insurance claims database, United States. J Am Acad Dermatol. 2025:S0190-9622(25)00098-2. doi:10.1016/j.jaad.2025.01.022
- Yadgar RJ, Bhatia N, Friedman A. Cutaneous fungal infections are commonly misdiagnosed: a survey-based study. J Am Acad Dermatol. 2017;76:562-563.
- Flint ND, Rhoads JLW, Carlisle R, et al. The continued inappropriate use and overuse of combination topical clotrimazole-betamethasone. Dermatol Online J. 2021;27. doi:10.5070/D327854686
- Currie DW, Caplan AS, Benedict K, et al. Prescribing of clotrimazolebetamethasone dipropionate, a topical combination corticosteroidantifungal product, for Medicare part D beneficiaries, United States, 2016–2022. Antimicrob Steward Healthc Epidemiol. 2024;4:E174.
- Gold JA, Caplan AS, Benedict K, et al. Clotrimazole-betamethasone dipropionate prescribing for nonfungal skin conditions. JAMA Network Open. 2024;7:E2411721-E2411721.
- Noble SL, Forbes RC, Stamm PL. Diagnosis and management of common tinea infections. Am Fam Physician. 1998;58:163-174, 177-168.
- Ely JW, Rosenfeld S, Seabury Stone M. Diagnosis and management of tinea infections. Am Fam Physician. 2014;90:702-710.
- Uhrlaß S, Verma SB, Gräser Y, et al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: I. epidemiology, risk factors and clinical features. Indian J Dermatol Venereol Leprol. 2021;87:154-175.
- Chen E, Ghannoum M, Elewski BE. Treatment]resistant tinea corporis, a potential public health issue. Br J Dermatol. 2021;184:164-165.
- Caplan AS. Notes from the field: first reported US cases of tinea caused by Trichophyton indotineae—New York City, December 2021–March 2023. MMWR Morbidity and Mortality Weekly Report. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Spivack S, Gold JA, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807.
- Jabet A, Brun S, Normand AC, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233.
- Thakur S, Spruijtenburg B, Abhishek, et al. Whole genome sequence analysis of terbinafine resistant and susceptible Trichophyton isolates from human and animal origin. Mycopathologia. 2025;190:13.
- Lockhart SR, Chowdhary A, Gold JA. The rapid emergence of antifungal-resistant human-pathogenic fungi. Nat Rev Microbiol. 2023;21:818-832.
- Mosam A, Shuping L, Naicker S, et al. A case of antifungal-resistant ringworm infection in KwaZulu-Natal Province, South Africa, caused by Trichophyton indotineae. Public Health Bulletin South Africa. Accessed April 4, 2025. https://www.phbsa.ac.za/wp-content/uploads/2023/12PHBSA-Ringworm-Article-2023.pdf
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:E0056223
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709. doi:10.1001/jamadermatol.2024.1126
- Benedict K. Topical antifungal prescribing for Medicare Part D beneficiaries—United States, 2021. MMWR Morb Mortal Wkly Rep. 2024;73:1-5.
- Verma SB. Emergence of recalcitrant dermatophytosis in India. Lancet Infect Dis. 2018;18:718-719.
- Khurana A, Sharath S, Sardana K, et al. Clinico-mycological and therapeutic updates on cutaneous dermatophytic infections in the era of Trichophyton indotineae. J Am Acad Dermatol. 2024;91:315-323. doi:10.1016/j.jaad.2024.03.024
- Verma S. Steroid modified tinea. BMJ. 2017;356:j973.
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278.
- Burmester A, Hipler UC, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180.
- Bhuiyan MSI, Verma SB, Illigner GM, et al. Trichophyton mentagrophytes ITS genotype VIII/Trichophyton indotineae infection and antifungal resistance in Bangladesh. J Fungi (Basel). 2024;10:768. doi:10.3390 /jof10110768
- Hay RJ. Chapter 82: superficial mycoses. In: Ryan ET, Hill DR, Solomon T, et al, eds. Hunter’s Tropical Medicine and Emerging Infectious Diseases. 10th ed. Elsevier; 2020:648-652.
- Gupta AK, Cooper EA, Wang T, et al. Detection of squalene epoxidase mutations in United States patients with onychomycosis: implications for management. J Invest Dermatol. 2023;143:2476-2483.E2477.
- Hwang JK, Bakotic WL, Gold JA, et al. Isolation of terbinafine-resistant Trichophyton rubrum from onychomycosis patients who failed treatment at an academic center in New York, United States. J Fungi. 2023;9:710.
- Gu D, Hatch M, Ghannoum M, et al. Treatment-resistant dermatophytosis: a representative case highlighting an emerging public health threat. JAAD Case Rep. 2020;6:1153-1155.
- Jabet A, Dellière S, Seang S, et al. Sexually transmitted Trichophyton mentagrophytes genotype VII infection among men who have sex with men. Emerg Infect Dis. 2023;29:1411-1414.
- Zucker J, Caplan AS, Gunaratne SH, et al. Notes from the field: Trichophyton mentagrophytes genotype VII—New York City, April-July 2024. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Caplan AS, Gold JA, Smith DJ, et al. Improving antifungal stewardship in dermatology in an era of emerging dermatophyte resistance. JAAD International. 2024;15:168-169.
- Elewski B. A call for antifungal stewardship. Br J Dermatol. 2020; 183:798-799.
- Gold JAW, Benedict K, Caplan AS, et al. High rates of potentially unnecessary topical antifungal prescribing in a large commercial health insurance claims database, United States. J Am Acad Dermatol. 2025:S0190-9622(25)00098-2. doi:10.1016/j.jaad.2025.01.022
- Yadgar RJ, Bhatia N, Friedman A. Cutaneous fungal infections are commonly misdiagnosed: a survey-based study. J Am Acad Dermatol. 2017;76:562-563.
- Flint ND, Rhoads JLW, Carlisle R, et al. The continued inappropriate use and overuse of combination topical clotrimazole-betamethasone. Dermatol Online J. 2021;27. doi:10.5070/D327854686
- Currie DW, Caplan AS, Benedict K, et al. Prescribing of clotrimazolebetamethasone dipropionate, a topical combination corticosteroidantifungal product, for Medicare part D beneficiaries, United States, 2016–2022. Antimicrob Steward Healthc Epidemiol. 2024;4:E174.
- Gold JA, Caplan AS, Benedict K, et al. Clotrimazole-betamethasone dipropionate prescribing for nonfungal skin conditions. JAMA Network Open. 2024;7:E2411721-E2411721.
The Rise of Antifungal-Resistant Dermatophyte Infections: What Dermatologists Need to Know
The Rise of Antifungal-Resistant Dermatophyte Infections: What Dermatologists Need to Know
PRACTICE POINTS
- Recently emerged dermatophyte species pose a global public health concern because of infection severity, frequent resistance to terbinafine, and easy person-to-person transmission.
- Prolonged itraconazole therapy is considered the firstline treatment for infections caused by Trichophyton indotineae, a globally emerging and frequently terbinafine-resistant dermatophyte.
- Dermatologists can educate nondermatologists on the importance of mycologic confirmation and avoidance of the use of topical antifungal/ corticosteroid products, which are hypothesized to contribute to emergence and spread of resistance.
Pink Ulcerated Nodule on the Forearm
Pink Ulcerated Nodule on the Forearm
THE DIAGNOSIS: Cutaneous Cryptococcosis
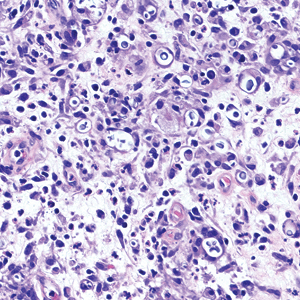
Biopsy of the ulcerated nodule showed numerous yeastlike organisms within clear mucinous capsules and with some surrounding inflammation. On Grocott methenamine silver staining, the organisms stained black. Workup for disseminated cryptococcus was negative, leading to a diagnosis of primary cutaneous cryptococcosis in the setting of immunosuppression. Notably, cryptococcosis infection has been reported in patients taking fingolimod (a sphingosine-1-phosphate receptor) for multiple sclerosis, which was the case for our patient.1
The genus Cryptococcus comprises more than 30 species of encapsulated basidiomycetous fungi distributed ubiquitously in nature. Currently, only 2 species are known to cause infectious disease in humans: Cryptococcus neoformans, which affects both immunocompromised and immunocompetent patients and frequently is isolated from pigeon droppings, as well as Cryptococcus gatti, which primarily affects immunocompetent patients and is more commonly isolated from soil and decaying wood.2
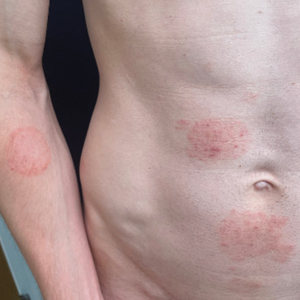
Primary cutaneous cryptococcosis (PCC), characterized by direct inoculation of C neoformans or C gatti via skin injury, is rare and typically is seen in patients with decreased cell-mediated immunity, such as those on chronic corticosteroid therapy, solid-organ transplant recipients, and those with HIV.3 Primary cutaneous cryptococcosis typically manifests as a solitary or confined lesion on exposed areas of the skin and often is accompanied by regional lymphadenopathy.4,5 The most common cutaneous findings associated with PCC include ulceration, cellulitis, and whitlow.5 In immunocompetent hosts, frequently affected sites include the arms, fingers, and face, while the trunk and lower extremities are more commonly affected in immunocompromised hosts.3 Secondary cutaneous cryptococcosis occurs through hematologic spread in patients with disseminated cryptococcosis after inhalation of Cryptococcosis spores and differs from PCC in that it typically manifests as multiple lesions scattered on both exposed and covered areas of the skin. Patients also may have signs and symptoms of disseminated cryptococcosis such as pneumonia and/or meningitis at presentation.5
Despite the difference between PCC and secondary cutaneous cryptococcosis, almost every type of skin lesion has been observed in cryptococcosis, including pustules, nodules, vesicles, acneform lesions, purpura, ulcers, abscesses, molluscumlike lesions, granulomas, draining sinuses, and cellulitis.6,7
Cutaneous cryptococcosis generally is associated with 2 types of histologic reactions: gelatinous and granulomatous. The gelatinous reaction shows numerous yeastlike organisms ranging from 4 μm to 12 μm in diameter with large mucinous polysaccharide capsules and scant inflammation. Organisms may be seen in mucoid sheets.8 The granulomatous type shows a more pronounced reaction with fewer organisms ranging from 2 μm to 4 μm in diameter found within giant cells, histiocytes, and lymphocytes.6,9 Areas of necrosis occasionally can be observed.8
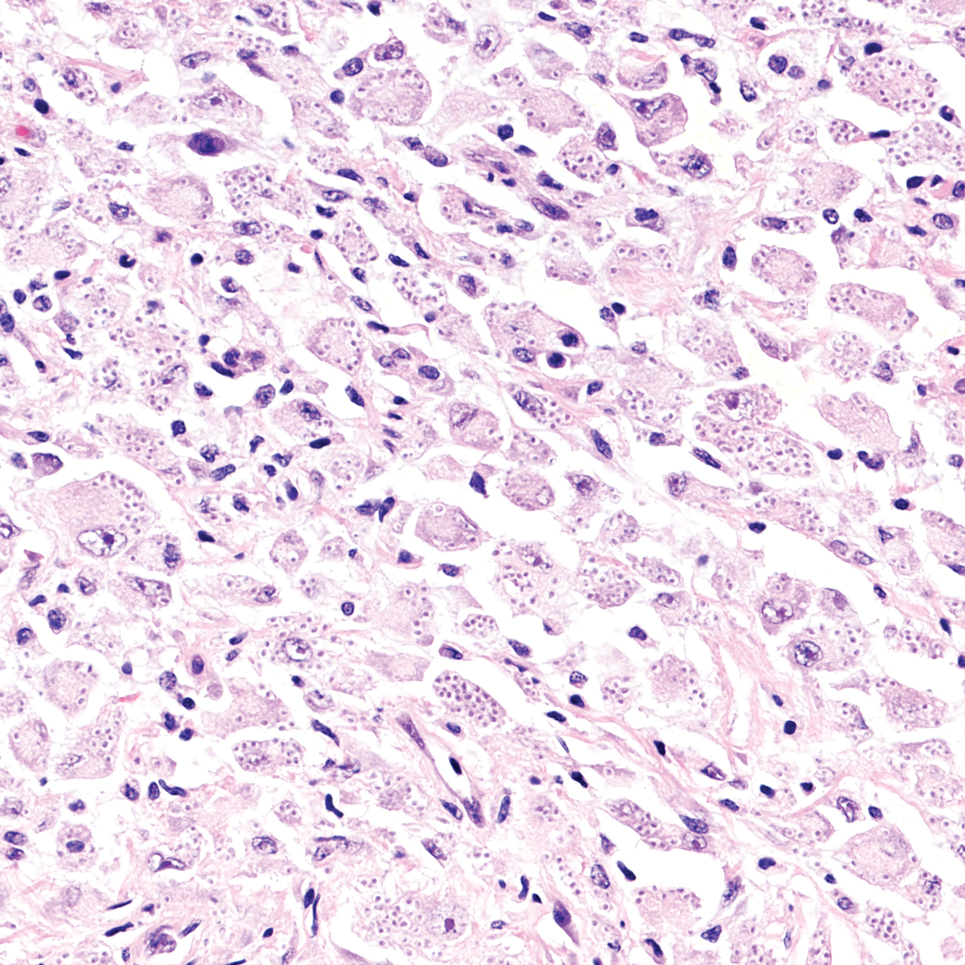
It is important to consider infection with Blastomyces dermatitidis and Histoplasma capsulatum in the differential Both entities can manifest as necrotizing granulomas on histology (Figures 1 and 2).10 Microscopic morphology can help differentiate these pathogenic fungi from Cryptococcus diagnosis of cryptococcosis. species which show pleomorphic, narrow-based budding yeast with wide capsules. In contrast, H capsulatum is characterized by small, intracellular, yeastlike cells with microconidia and macroconidia, while B dermatitidis is distinguished by spherical, thick-walled cells with broad-based budding.11 Capsular material also can help distinguish Cryptococcus from other pathogenic fungi. Special stains highlighting the polysaccharide capsule of Cryptococcus can best identify the yeast. The capsule stains red with periodic acid–Schiff, blue with Alcian blue, and black with Grocott methenamine silver. Mucicarmine is especially useful as it can stain the mucinous capsule pinkish red and typically does not stain other pathogenic fungi.12 Capsule-deficient organisms can lead to considerable difficulties in diagnosis given the organisms can vary in size and may mimic H capsulatum or B dermatitidis. The Fontana-Masson stain is a valuable tool in identifying capsule-deficient organisms, as melanin is found in Cryptococcus cell walls; thus, positive staining excludes H capsulatum and B dermatitidis.13
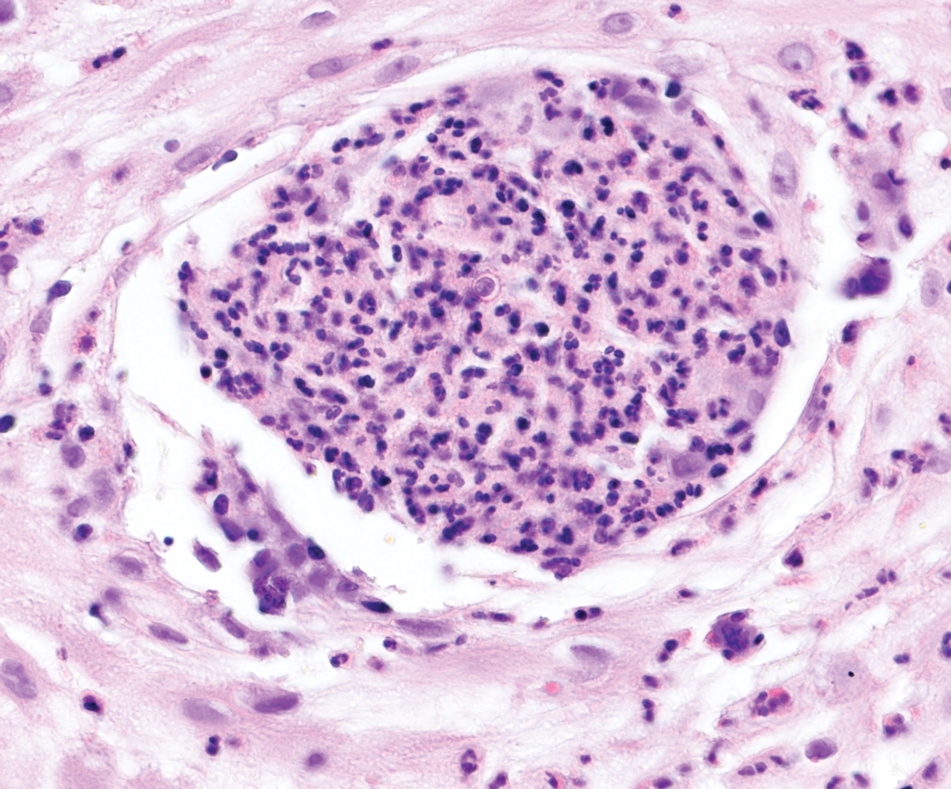
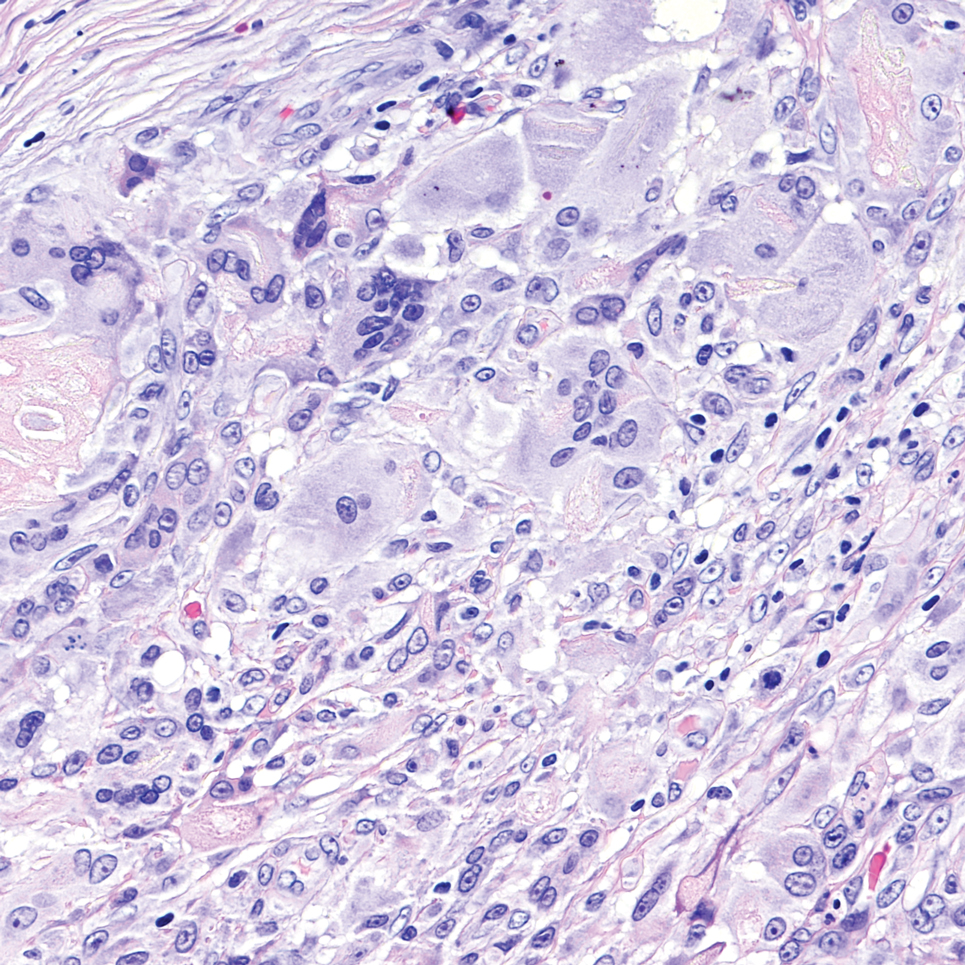
Cutaneous foreign body granuloma, which refers to a granulomatous inflammatory reaction to a foreign body in the skin, is another differential diagnosis that is important to distinguish from cutaneous cryptococcosis. On histology, a collection of histiocytes surround the inert material, forming giant cells without an immune response (Figure 3).10 In contrast, granulomas caused by infectious etiologies (eg, Cryptococcus species) have an associated adaptive immune response and can be further classified as necrotizing or non-necrotizing. Necrotizing granulomas have a distinct central necrosis with a surrounding lymphohistiocytic reaction with peripheral chronic inflammation.10
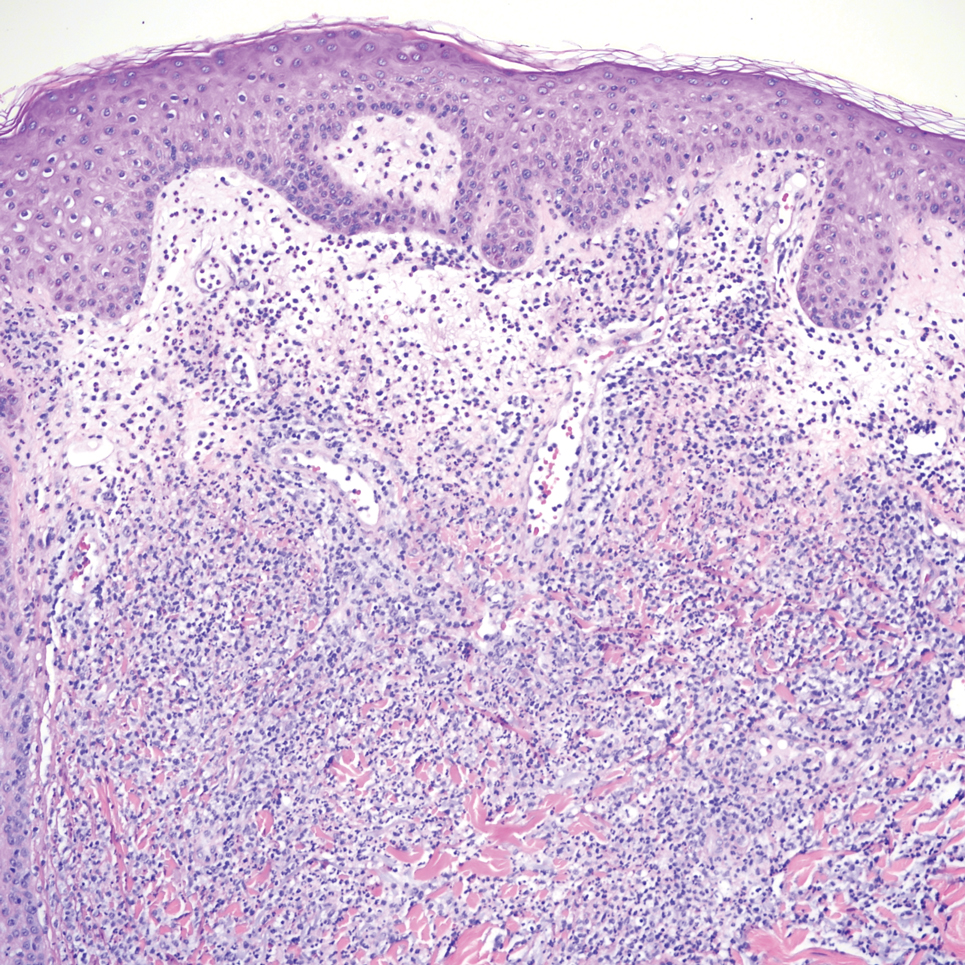
Sweet syndrome is another mimicker of cutaneous cryptococcosis. A histologic variant of Sweet syndrome has been reported that has characteristic cutaneous lesions clinically but shows basophilic bodies with a surrounding halo on pathology that can be mistaken for Cryptococcus yeast. Classic histopathology of Sweet syndrome features papillary dermal edema with neutrophil or histiocytelike inflammatory infiltrate (Figure 4). Identification of Sweet syndrome can be aided by positive myeloperoxidase staining and negative periodic acid–Schiff staining.14,15
- Lehmann NM, Kammeyer JA. Cerebral venous thrombosis due to Cryptococcus in a multiple sclerosis patient on fingolimod. Case Rep Neurol. 2022; 14:286-290. doi:10.1159/000524359
- Maziarz EK, Perfect JR. Cryptococcosis. Infect Dis Clin North Am. 2016;30:179-206. doi:10.1016/j.idc.2015.10.006.
- Christianson JC, Engber W, Andes D. Primary cutaneous cryptococcosis in immunocompetent and immunocompromised hosts. Med Mycol. 2003;41:177-188. doi:10.1080/1369378031000137224
- Tilak R, Prakash P, Nigam C, et al. Cryptococcal meningitis with an antecedent cutaneous Cryptococcal lesion. Dermatol Online J. 2009;15:12.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347. doi:10.1086/345956
- Dimino-Emme L, Gurevitch AW. Cutaneous manifestations of disseminated cryptococcosis. J Am Acad Dermatol. 1995;32:844-850.
- Anderson DJ, Schmidt C, Goodman J, Pomeroy C. Cryptococcal disease presenting as cellulitis. Clin Infect Dis. 1992;14:666-672. doi:10.1093/clinids/14.3.666
- Moore M. Cryptococcosis with cutaneous manifestations: four cases with a review of published reports. J Invest Dermatol. 1957;28(2):159-182. doi: 10.1038/jid.1957.17
- Phan NQ, Tirado M, Moeckel SMC, et al. Cutaneous and pulmonary cryptococcosis in an immunocompetent patient. J Dtsch Dermatol Ges. 2019;17:1283-1286. doi:10.1111/ddg.13997.
- Shah KK, Pritt BS, Alexander MP. Histopathologic review of granulomatous inflammation. J Clin Tuberc Other Mycobact Dis. 2017;7:1-12. doi: 10.1016/j.jctube.2017.02.001
- Fridlington E, Colome-Grimmer M, Kelly E, et al. Tzanck smear as a rapid diagnostic tool for disseminated cryptococcal infection. Arch Dermatol. 2006;142:25-27. doi: 10.1001/archderm.142.1.25
- Hernandez AD. Cutaneous Cryptococcosis. Dermatol Clin. 1989; 7:269-274.
- Ro JY, Lee SS, Ayala AG. Advantage of Fontana-Masson stain in capsule-deficient cryptococcal infection. Arch Pathol Lab Med. 1987;111:53-57.
- Jordan AA, Graciaa DS, Gopalsamy SN, et al. Sweet syndrome imitating cutaneous cryptococcal disease. Open Forum Infect Dis. 2022;9:ofac608. doi: 10.1093/ofid/ofac608
- Ko JS, Fernandez AP, Anderson KA, et al. Morphologic mimickers of Cryptococcus occurring within inflammatory infiltrates in the setting of neutrophilic dermatitis: a series of three cases highlighting clinical dilemmas associated with a novel histopathologic pitfall. J Cutan Pathol. 2013;40:38-45. doi: 10.1111/cup.12019
THE DIAGNOSIS: Cutaneous Cryptococcosis
Biopsy of the ulcerated nodule showed numerous yeastlike organisms within clear mucinous capsules and with some surrounding inflammation. On Grocott methenamine silver staining, the organisms stained black. Workup for disseminated cryptococcus was negative, leading to a diagnosis of primary cutaneous cryptococcosis in the setting of immunosuppression. Notably, cryptococcosis infection has been reported in patients taking fingolimod (a sphingosine-1-phosphate receptor) for multiple sclerosis, which was the case for our patient.1
The genus Cryptococcus comprises more than 30 species of encapsulated basidiomycetous fungi distributed ubiquitously in nature. Currently, only 2 species are known to cause infectious disease in humans: Cryptococcus neoformans, which affects both immunocompromised and immunocompetent patients and frequently is isolated from pigeon droppings, as well as Cryptococcus gatti, which primarily affects immunocompetent patients and is more commonly isolated from soil and decaying wood.2
Primary cutaneous cryptococcosis (PCC), characterized by direct inoculation of C neoformans or C gatti via skin injury, is rare and typically is seen in patients with decreased cell-mediated immunity, such as those on chronic corticosteroid therapy, solid-organ transplant recipients, and those with HIV.3 Primary cutaneous cryptococcosis typically manifests as a solitary or confined lesion on exposed areas of the skin and often is accompanied by regional lymphadenopathy.4,5 The most common cutaneous findings associated with PCC include ulceration, cellulitis, and whitlow.5 In immunocompetent hosts, frequently affected sites include the arms, fingers, and face, while the trunk and lower extremities are more commonly affected in immunocompromised hosts.3 Secondary cutaneous cryptococcosis occurs through hematologic spread in patients with disseminated cryptococcosis after inhalation of Cryptococcosis spores and differs from PCC in that it typically manifests as multiple lesions scattered on both exposed and covered areas of the skin. Patients also may have signs and symptoms of disseminated cryptococcosis such as pneumonia and/or meningitis at presentation.5
Despite the difference between PCC and secondary cutaneous cryptococcosis, almost every type of skin lesion has been observed in cryptococcosis, including pustules, nodules, vesicles, acneform lesions, purpura, ulcers, abscesses, molluscumlike lesions, granulomas, draining sinuses, and cellulitis.6,7
Cutaneous cryptococcosis generally is associated with 2 types of histologic reactions: gelatinous and granulomatous. The gelatinous reaction shows numerous yeastlike organisms ranging from 4 μm to 12 μm in diameter with large mucinous polysaccharide capsules and scant inflammation. Organisms may be seen in mucoid sheets.8 The granulomatous type shows a more pronounced reaction with fewer organisms ranging from 2 μm to 4 μm in diameter found within giant cells, histiocytes, and lymphocytes.6,9 Areas of necrosis occasionally can be observed.8
It is important to consider infection with Blastomyces dermatitidis and Histoplasma capsulatum in the differential Both entities can manifest as necrotizing granulomas on histology (Figures 1 and 2).10 Microscopic morphology can help differentiate these pathogenic fungi from Cryptococcus diagnosis of cryptococcosis. species which show pleomorphic, narrow-based budding yeast with wide capsules. In contrast, H capsulatum is characterized by small, intracellular, yeastlike cells with microconidia and macroconidia, while B dermatitidis is distinguished by spherical, thick-walled cells with broad-based budding.11 Capsular material also can help distinguish Cryptococcus from other pathogenic fungi. Special stains highlighting the polysaccharide capsule of Cryptococcus can best identify the yeast. The capsule stains red with periodic acid–Schiff, blue with Alcian blue, and black with Grocott methenamine silver. Mucicarmine is especially useful as it can stain the mucinous capsule pinkish red and typically does not stain other pathogenic fungi.12 Capsule-deficient organisms can lead to considerable difficulties in diagnosis given the organisms can vary in size and may mimic H capsulatum or B dermatitidis. The Fontana-Masson stain is a valuable tool in identifying capsule-deficient organisms, as melanin is found in Cryptococcus cell walls; thus, positive staining excludes H capsulatum and B dermatitidis.13


Cutaneous foreign body granuloma, which refers to a granulomatous inflammatory reaction to a foreign body in the skin, is another differential diagnosis that is important to distinguish from cutaneous cryptococcosis. On histology, a collection of histiocytes surround the inert material, forming giant cells without an immune response (Figure 3).10 In contrast, granulomas caused by infectious etiologies (eg, Cryptococcus species) have an associated adaptive immune response and can be further classified as necrotizing or non-necrotizing. Necrotizing granulomas have a distinct central necrosis with a surrounding lymphohistiocytic reaction with peripheral chronic inflammation.10

Sweet syndrome is another mimicker of cutaneous cryptococcosis. A histologic variant of Sweet syndrome has been reported that has characteristic cutaneous lesions clinically but shows basophilic bodies with a surrounding halo on pathology that can be mistaken for Cryptococcus yeast. Classic histopathology of Sweet syndrome features papillary dermal edema with neutrophil or histiocytelike inflammatory infiltrate (Figure 4). Identification of Sweet syndrome can be aided by positive myeloperoxidase staining and negative periodic acid–Schiff staining.14,15

THE DIAGNOSIS: Cutaneous Cryptococcosis
Biopsy of the ulcerated nodule showed numerous yeastlike organisms within clear mucinous capsules and with some surrounding inflammation. On Grocott methenamine silver staining, the organisms stained black. Workup for disseminated cryptococcus was negative, leading to a diagnosis of primary cutaneous cryptococcosis in the setting of immunosuppression. Notably, cryptococcosis infection has been reported in patients taking fingolimod (a sphingosine-1-phosphate receptor) for multiple sclerosis, which was the case for our patient.1
The genus Cryptococcus comprises more than 30 species of encapsulated basidiomycetous fungi distributed ubiquitously in nature. Currently, only 2 species are known to cause infectious disease in humans: Cryptococcus neoformans, which affects both immunocompromised and immunocompetent patients and frequently is isolated from pigeon droppings, as well as Cryptococcus gatti, which primarily affects immunocompetent patients and is more commonly isolated from soil and decaying wood.2
Primary cutaneous cryptococcosis (PCC), characterized by direct inoculation of C neoformans or C gatti via skin injury, is rare and typically is seen in patients with decreased cell-mediated immunity, such as those on chronic corticosteroid therapy, solid-organ transplant recipients, and those with HIV.3 Primary cutaneous cryptococcosis typically manifests as a solitary or confined lesion on exposed areas of the skin and often is accompanied by regional lymphadenopathy.4,5 The most common cutaneous findings associated with PCC include ulceration, cellulitis, and whitlow.5 In immunocompetent hosts, frequently affected sites include the arms, fingers, and face, while the trunk and lower extremities are more commonly affected in immunocompromised hosts.3 Secondary cutaneous cryptococcosis occurs through hematologic spread in patients with disseminated cryptococcosis after inhalation of Cryptococcosis spores and differs from PCC in that it typically manifests as multiple lesions scattered on both exposed and covered areas of the skin. Patients also may have signs and symptoms of disseminated cryptococcosis such as pneumonia and/or meningitis at presentation.5
Despite the difference between PCC and secondary cutaneous cryptococcosis, almost every type of skin lesion has been observed in cryptococcosis, including pustules, nodules, vesicles, acneform lesions, purpura, ulcers, abscesses, molluscumlike lesions, granulomas, draining sinuses, and cellulitis.6,7
Cutaneous cryptococcosis generally is associated with 2 types of histologic reactions: gelatinous and granulomatous. The gelatinous reaction shows numerous yeastlike organisms ranging from 4 μm to 12 μm in diameter with large mucinous polysaccharide capsules and scant inflammation. Organisms may be seen in mucoid sheets.8 The granulomatous type shows a more pronounced reaction with fewer organisms ranging from 2 μm to 4 μm in diameter found within giant cells, histiocytes, and lymphocytes.6,9 Areas of necrosis occasionally can be observed.8
It is important to consider infection with Blastomyces dermatitidis and Histoplasma capsulatum in the differential Both entities can manifest as necrotizing granulomas on histology (Figures 1 and 2).10 Microscopic morphology can help differentiate these pathogenic fungi from Cryptococcus diagnosis of cryptococcosis. species which show pleomorphic, narrow-based budding yeast with wide capsules. In contrast, H capsulatum is characterized by small, intracellular, yeastlike cells with microconidia and macroconidia, while B dermatitidis is distinguished by spherical, thick-walled cells with broad-based budding.11 Capsular material also can help distinguish Cryptococcus from other pathogenic fungi. Special stains highlighting the polysaccharide capsule of Cryptococcus can best identify the yeast. The capsule stains red with periodic acid–Schiff, blue with Alcian blue, and black with Grocott methenamine silver. Mucicarmine is especially useful as it can stain the mucinous capsule pinkish red and typically does not stain other pathogenic fungi.12 Capsule-deficient organisms can lead to considerable difficulties in diagnosis given the organisms can vary in size and may mimic H capsulatum or B dermatitidis. The Fontana-Masson stain is a valuable tool in identifying capsule-deficient organisms, as melanin is found in Cryptococcus cell walls; thus, positive staining excludes H capsulatum and B dermatitidis.13


Cutaneous foreign body granuloma, which refers to a granulomatous inflammatory reaction to a foreign body in the skin, is another differential diagnosis that is important to distinguish from cutaneous cryptococcosis. On histology, a collection of histiocytes surround the inert material, forming giant cells without an immune response (Figure 3).10 In contrast, granulomas caused by infectious etiologies (eg, Cryptococcus species) have an associated adaptive immune response and can be further classified as necrotizing or non-necrotizing. Necrotizing granulomas have a distinct central necrosis with a surrounding lymphohistiocytic reaction with peripheral chronic inflammation.10

Sweet syndrome is another mimicker of cutaneous cryptococcosis. A histologic variant of Sweet syndrome has been reported that has characteristic cutaneous lesions clinically but shows basophilic bodies with a surrounding halo on pathology that can be mistaken for Cryptococcus yeast. Classic histopathology of Sweet syndrome features papillary dermal edema with neutrophil or histiocytelike inflammatory infiltrate (Figure 4). Identification of Sweet syndrome can be aided by positive myeloperoxidase staining and negative periodic acid–Schiff staining.14,15

- Lehmann NM, Kammeyer JA. Cerebral venous thrombosis due to Cryptococcus in a multiple sclerosis patient on fingolimod. Case Rep Neurol. 2022; 14:286-290. doi:10.1159/000524359
- Maziarz EK, Perfect JR. Cryptococcosis. Infect Dis Clin North Am. 2016;30:179-206. doi:10.1016/j.idc.2015.10.006.
- Christianson JC, Engber W, Andes D. Primary cutaneous cryptococcosis in immunocompetent and immunocompromised hosts. Med Mycol. 2003;41:177-188. doi:10.1080/1369378031000137224
- Tilak R, Prakash P, Nigam C, et al. Cryptococcal meningitis with an antecedent cutaneous Cryptococcal lesion. Dermatol Online J. 2009;15:12.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347. doi:10.1086/345956
- Dimino-Emme L, Gurevitch AW. Cutaneous manifestations of disseminated cryptococcosis. J Am Acad Dermatol. 1995;32:844-850.
- Anderson DJ, Schmidt C, Goodman J, Pomeroy C. Cryptococcal disease presenting as cellulitis. Clin Infect Dis. 1992;14:666-672. doi:10.1093/clinids/14.3.666
- Moore M. Cryptococcosis with cutaneous manifestations: four cases with a review of published reports. J Invest Dermatol. 1957;28(2):159-182. doi: 10.1038/jid.1957.17
- Phan NQ, Tirado M, Moeckel SMC, et al. Cutaneous and pulmonary cryptococcosis in an immunocompetent patient. J Dtsch Dermatol Ges. 2019;17:1283-1286. doi:10.1111/ddg.13997.
- Shah KK, Pritt BS, Alexander MP. Histopathologic review of granulomatous inflammation. J Clin Tuberc Other Mycobact Dis. 2017;7:1-12. doi: 10.1016/j.jctube.2017.02.001
- Fridlington E, Colome-Grimmer M, Kelly E, et al. Tzanck smear as a rapid diagnostic tool for disseminated cryptococcal infection. Arch Dermatol. 2006;142:25-27. doi: 10.1001/archderm.142.1.25
- Hernandez AD. Cutaneous Cryptococcosis. Dermatol Clin. 1989; 7:269-274.
- Ro JY, Lee SS, Ayala AG. Advantage of Fontana-Masson stain in capsule-deficient cryptococcal infection. Arch Pathol Lab Med. 1987;111:53-57.
- Jordan AA, Graciaa DS, Gopalsamy SN, et al. Sweet syndrome imitating cutaneous cryptococcal disease. Open Forum Infect Dis. 2022;9:ofac608. doi: 10.1093/ofid/ofac608
- Ko JS, Fernandez AP, Anderson KA, et al. Morphologic mimickers of Cryptococcus occurring within inflammatory infiltrates in the setting of neutrophilic dermatitis: a series of three cases highlighting clinical dilemmas associated with a novel histopathologic pitfall. J Cutan Pathol. 2013;40:38-45. doi: 10.1111/cup.12019
- Lehmann NM, Kammeyer JA. Cerebral venous thrombosis due to Cryptococcus in a multiple sclerosis patient on fingolimod. Case Rep Neurol. 2022; 14:286-290. doi:10.1159/000524359
- Maziarz EK, Perfect JR. Cryptococcosis. Infect Dis Clin North Am. 2016;30:179-206. doi:10.1016/j.idc.2015.10.006.
- Christianson JC, Engber W, Andes D. Primary cutaneous cryptococcosis in immunocompetent and immunocompromised hosts. Med Mycol. 2003;41:177-188. doi:10.1080/1369378031000137224
- Tilak R, Prakash P, Nigam C, et al. Cryptococcal meningitis with an antecedent cutaneous Cryptococcal lesion. Dermatol Online J. 2009;15:12.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347. doi:10.1086/345956
- Dimino-Emme L, Gurevitch AW. Cutaneous manifestations of disseminated cryptococcosis. J Am Acad Dermatol. 1995;32:844-850.
- Anderson DJ, Schmidt C, Goodman J, Pomeroy C. Cryptococcal disease presenting as cellulitis. Clin Infect Dis. 1992;14:666-672. doi:10.1093/clinids/14.3.666
- Moore M. Cryptococcosis with cutaneous manifestations: four cases with a review of published reports. J Invest Dermatol. 1957;28(2):159-182. doi: 10.1038/jid.1957.17
- Phan NQ, Tirado M, Moeckel SMC, et al. Cutaneous and pulmonary cryptococcosis in an immunocompetent patient. J Dtsch Dermatol Ges. 2019;17:1283-1286. doi:10.1111/ddg.13997.
- Shah KK, Pritt BS, Alexander MP. Histopathologic review of granulomatous inflammation. J Clin Tuberc Other Mycobact Dis. 2017;7:1-12. doi: 10.1016/j.jctube.2017.02.001
- Fridlington E, Colome-Grimmer M, Kelly E, et al. Tzanck smear as a rapid diagnostic tool for disseminated cryptococcal infection. Arch Dermatol. 2006;142:25-27. doi: 10.1001/archderm.142.1.25
- Hernandez AD. Cutaneous Cryptococcosis. Dermatol Clin. 1989; 7:269-274.
- Ro JY, Lee SS, Ayala AG. Advantage of Fontana-Masson stain in capsule-deficient cryptococcal infection. Arch Pathol Lab Med. 1987;111:53-57.
- Jordan AA, Graciaa DS, Gopalsamy SN, et al. Sweet syndrome imitating cutaneous cryptococcal disease. Open Forum Infect Dis. 2022;9:ofac608. doi: 10.1093/ofid/ofac608
- Ko JS, Fernandez AP, Anderson KA, et al. Morphologic mimickers of Cryptococcus occurring within inflammatory infiltrates in the setting of neutrophilic dermatitis: a series of three cases highlighting clinical dilemmas associated with a novel histopathologic pitfall. J Cutan Pathol. 2013;40:38-45. doi: 10.1111/cup.12019
Pink Ulcerated Nodule on the Forearm
Pink Ulcerated Nodule on the Forearm
A 51-year-old man with a history of multiple sclerosis treated with fingolimod presented to the dermatology department with an ulcerated lesion on the left forearm of 2 to 3 months’ duration. The patient reported that he recently presented to the emergency department for drainage of the lesion, which was unsuccessful. Shortly after, he traumatized the lesion at his construction job. At the current presentation, physical examination revealed a 1-cm, flesh-colored to faintly pink, ulcerated nodule on the left forearm. A biopsy was performed.
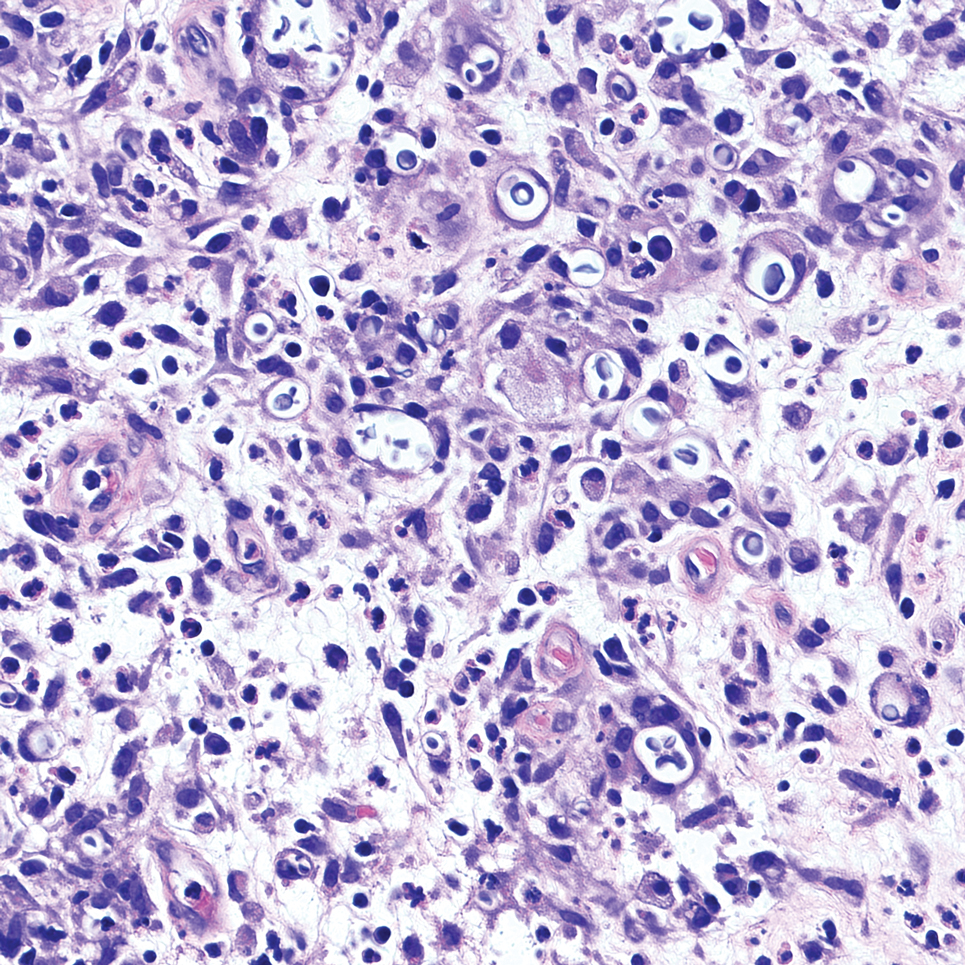
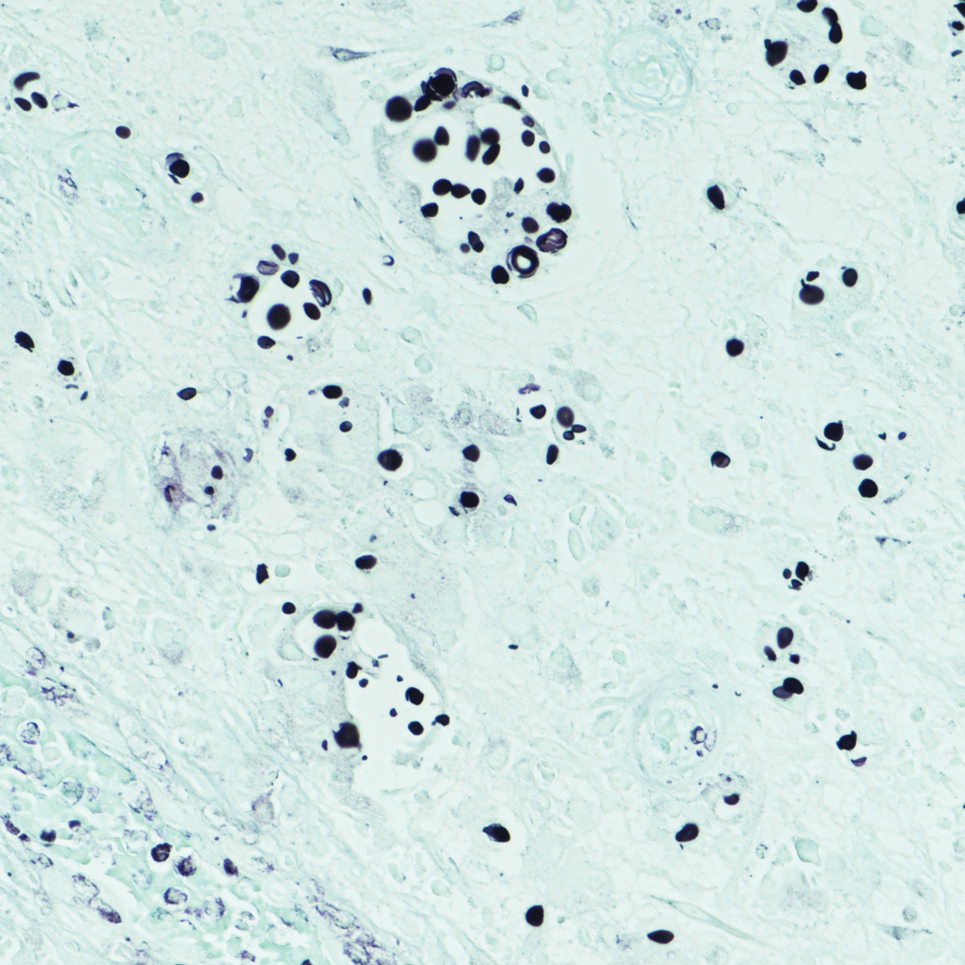
New RSV Vaccine Shows Strong Protection in Veterans
TOPLINE:
A single dose of the recombinant respiratory syncytial virus (RSV) vaccine demonstrates effectiveness against infections and associated hospitalizations in veterans aged 60 years or older during the 2023-2024 respiratory illness season. This protection extends across age groups and immunocompromised individuals.
METHODOLOGY:
Researchers conducted a target trial emulation study to evaluate the real-world effectiveness of a single dose of recombinant RSV vaccine (RSVPreF3 or RSVpreF) among veterans enrolled in the Veterans Health Administration in the United States between September 1 and December 31, 2023.
They analyzed 146,852 vaccinated veterans (69.2%, RSVPreF; 29.9%, RSVPreF3) propensity matched with 582,936 unvaccinated ones (median age, ~76 years; ~94% men; immunocompromised individuals, 11.2%) who were followed up for a median of 124 days.
The primary outcome was any positive RSV test result obtained from day 14 after vaccination.
The secondary outcomes were RSV-associated emergency department or urgent care visits, hospitalizations, intensive care unit (ICU) admissions, and death.
TAKEAWAY:
Vaccine effectiveness against documented RSV infections was 78.1% (95% CI, 72.6-83.5), with incidence rates of infections lower in the vaccinated group than in the unvaccinated group (1.7 vs 7.3 per 1000 person-years).
Likewise, vaccine effectiveness against RSV-associated emergency department or urgent care visits was 78.7% (95% CI, 72.2-84.8), with rates of infections lower in the vaccinated group than in the unvaccinated group (1.3 vs 5.7 per 1000 person-years).
Immunocompromised veterans demonstrated a lower vaccine effectiveness of 71.6% (95% CI, 55.4-85.2); however, infection rates remained lower in the vaccinated group than in the unvaccinated group (5.8 vs 19.9 per 1000 person-years).
Hospitalizations, ICU admission rates, and mortality rates were also lower in the vaccinated group than in the unvaccinated group.
IN PRACTICE:
“These results give confidence that an RSV vaccine for older adults is likely to provide protection against RSV infection and RSV disease, at least in the first season following vaccination,” wrote the author of an accompanying comment.
SOURCE:
The study was funded by the US Department of Veterans Affairs Cooperative Studies Program. It was published online on January 20, 2025, in The Lancet Infectious Diseases (2025 Jan 20. doi:10.1016/S1473-3099(24)00796-5)
LIMITATIONS:
This study did not account for veterans who sought care outside of the Veterans Health Administration. While the study employed rigorous matching to ensure the similarity of demographic, geographic, and clinical characteristics, there could still have been residual confounding. Also, the study was not designed to estimate the protective effect of the vaccine against mild RSV illness.
DISCLOSURES:
This study was supported by the US Department of Veterans Affairs Cooperative Studies Program and funded in part by the US Department of Health and Human Services Biomedical Advanced Research and Development Authority and US Food and Drug Administration. One of the authors reported receiving consulting support from Van-Breemen & Hynes and having a subcontract at Oregon State University for a Patient-Centered Outcomes Research Institute grant. Others reported no conflicts of interest.■
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A single dose of the recombinant respiratory syncytial virus (RSV) vaccine demonstrates effectiveness against infections and associated hospitalizations in veterans aged 60 years or older during the 2023-2024 respiratory illness season. This protection extends across age groups and immunocompromised individuals.
METHODOLOGY:
Researchers conducted a target trial emulation study to evaluate the real-world effectiveness of a single dose of recombinant RSV vaccine (RSVPreF3 or RSVpreF) among veterans enrolled in the Veterans Health Administration in the United States between September 1 and December 31, 2023.
They analyzed 146,852 vaccinated veterans (69.2%, RSVPreF; 29.9%, RSVPreF3) propensity matched with 582,936 unvaccinated ones (median age, ~76 years; ~94% men; immunocompromised individuals, 11.2%) who were followed up for a median of 124 days.
The primary outcome was any positive RSV test result obtained from day 14 after vaccination.
The secondary outcomes were RSV-associated emergency department or urgent care visits, hospitalizations, intensive care unit (ICU) admissions, and death.
TAKEAWAY:
Vaccine effectiveness against documented RSV infections was 78.1% (95% CI, 72.6-83.5), with incidence rates of infections lower in the vaccinated group than in the unvaccinated group (1.7 vs 7.3 per 1000 person-years).
Likewise, vaccine effectiveness against RSV-associated emergency department or urgent care visits was 78.7% (95% CI, 72.2-84.8), with rates of infections lower in the vaccinated group than in the unvaccinated group (1.3 vs 5.7 per 1000 person-years).
Immunocompromised veterans demonstrated a lower vaccine effectiveness of 71.6% (95% CI, 55.4-85.2); however, infection rates remained lower in the vaccinated group than in the unvaccinated group (5.8 vs 19.9 per 1000 person-years).
Hospitalizations, ICU admission rates, and mortality rates were also lower in the vaccinated group than in the unvaccinated group.
IN PRACTICE:
“These results give confidence that an RSV vaccine for older adults is likely to provide protection against RSV infection and RSV disease, at least in the first season following vaccination,” wrote the author of an accompanying comment.
SOURCE:
The study was funded by the US Department of Veterans Affairs Cooperative Studies Program. It was published online on January 20, 2025, in The Lancet Infectious Diseases (2025 Jan 20. doi:10.1016/S1473-3099(24)00796-5)
LIMITATIONS:
This study did not account for veterans who sought care outside of the Veterans Health Administration. While the study employed rigorous matching to ensure the similarity of demographic, geographic, and clinical characteristics, there could still have been residual confounding. Also, the study was not designed to estimate the protective effect of the vaccine against mild RSV illness.
DISCLOSURES:
This study was supported by the US Department of Veterans Affairs Cooperative Studies Program and funded in part by the US Department of Health and Human Services Biomedical Advanced Research and Development Authority and US Food and Drug Administration. One of the authors reported receiving consulting support from Van-Breemen & Hynes and having a subcontract at Oregon State University for a Patient-Centered Outcomes Research Institute grant. Others reported no conflicts of interest.■
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A single dose of the recombinant respiratory syncytial virus (RSV) vaccine demonstrates effectiveness against infections and associated hospitalizations in veterans aged 60 years or older during the 2023-2024 respiratory illness season. This protection extends across age groups and immunocompromised individuals.
METHODOLOGY:
Researchers conducted a target trial emulation study to evaluate the real-world effectiveness of a single dose of recombinant RSV vaccine (RSVPreF3 or RSVpreF) among veterans enrolled in the Veterans Health Administration in the United States between September 1 and December 31, 2023.
They analyzed 146,852 vaccinated veterans (69.2%, RSVPreF; 29.9%, RSVPreF3) propensity matched with 582,936 unvaccinated ones (median age, ~76 years; ~94% men; immunocompromised individuals, 11.2%) who were followed up for a median of 124 days.
The primary outcome was any positive RSV test result obtained from day 14 after vaccination.
The secondary outcomes were RSV-associated emergency department or urgent care visits, hospitalizations, intensive care unit (ICU) admissions, and death.
TAKEAWAY:
Vaccine effectiveness against documented RSV infections was 78.1% (95% CI, 72.6-83.5), with incidence rates of infections lower in the vaccinated group than in the unvaccinated group (1.7 vs 7.3 per 1000 person-years).
Likewise, vaccine effectiveness against RSV-associated emergency department or urgent care visits was 78.7% (95% CI, 72.2-84.8), with rates of infections lower in the vaccinated group than in the unvaccinated group (1.3 vs 5.7 per 1000 person-years).
Immunocompromised veterans demonstrated a lower vaccine effectiveness of 71.6% (95% CI, 55.4-85.2); however, infection rates remained lower in the vaccinated group than in the unvaccinated group (5.8 vs 19.9 per 1000 person-years).
Hospitalizations, ICU admission rates, and mortality rates were also lower in the vaccinated group than in the unvaccinated group.
IN PRACTICE:
“These results give confidence that an RSV vaccine for older adults is likely to provide protection against RSV infection and RSV disease, at least in the first season following vaccination,” wrote the author of an accompanying comment.
SOURCE:
The study was funded by the US Department of Veterans Affairs Cooperative Studies Program. It was published online on January 20, 2025, in The Lancet Infectious Diseases (2025 Jan 20. doi:10.1016/S1473-3099(24)00796-5)
LIMITATIONS:
This study did not account for veterans who sought care outside of the Veterans Health Administration. While the study employed rigorous matching to ensure the similarity of demographic, geographic, and clinical characteristics, there could still have been residual confounding. Also, the study was not designed to estimate the protective effect of the vaccine against mild RSV illness.
DISCLOSURES:
This study was supported by the US Department of Veterans Affairs Cooperative Studies Program and funded in part by the US Department of Health and Human Services Biomedical Advanced Research and Development Authority and US Food and Drug Administration. One of the authors reported receiving consulting support from Van-Breemen & Hynes and having a subcontract at Oregon State University for a Patient-Centered Outcomes Research Institute grant. Others reported no conflicts of interest.■
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Comorbidities and Lifestyle Risk Factors Associated With Scabies Infestation
Comorbidities and Lifestyle Risk Factors Associated With Scabies Infestation
To the Editor:
Scabies infestation, which has been recognized as a neglected tropical disease by the World Health Organization since 2017, is caused by the human itch mite (Sarcoptes scabiei var hominis).1 Infected individuals experience a pruritic papular rash when the mite burrows into the epidermis, where it lives and lays eggs.2,3 Infected individuals also may develop bacterial superinfections if the skin barrier becomes compromised, leading to systemic complications and considerable morbidity.3
In countries with high human development indices, scabies outbreaks are linked to densely populated living conditions, such as those found in nursing homes or prisons.3,4 Scabies also is transmitted via sexual contact in adults. Beyond immunosuppression, little is known about other comorbid conditions or lifestyle risk factors associated with scabies infestation.2 Because scabies can mimic a range of other dermatologic conditions such as folliculitis, atopic dermatitis, and arthropod bites, misdiagnosis is common and can lead to delayed treatment and increased transmission risk.4 In this study, we sought to examine comorbid conditions and/or lifestyle risk factors associated with scabies infestation.
A matched case-control study was performed using the Registered Tier dataset of the National Institutes of Health All of Us Research Program Curated Data Repository version 7, which includes more than 400,000 unique participants aged 18 years or older from across the United States. The All of Us Research Program excludes adults who are unable to consent independently as well as incarcerated populations and children younger than 18 years. Participants diagnosed with scabies were identified using SNOMED code 62752005 and compared to a control group matched 1:4 based on age, sex, and selfidentified race. SNOMED codes also were used to identify various comorbidities and lifestyle risk factors, including depression, bipolar disorder, anxiety, schizophrenia, peripheral vascular disease (PVD), HIV, type 2 diabetes mellitus (T2DM), unsheltered status, tobacco use, difficulty with activities of daily living, insurance status, and any recent travel history. Logistic regression models were used to calculate odds ratios (ORs) and estimate effect sizes, with statistical significance set at P<.05.
We identified 691 cases of scabies infestation and 2073 controls. The average age of the patients diagnosed with scabies was 55.1 years. Seventy percent (481/691) identified as female and 32.4% (224/491) identified as Black or African American. Matched controls were similar for all analyzed demographic characteristics (P=1.0)(eTable 1). Patients diagnosed with scabies were more likely to be unsheltered (OR, 2.33 [95% CI, 1.91-2.85]), use tobacco (OR 1.77 [95% CI, 1.48-2.11]) and have a comorbid diagnosis of HIV (OR, 3.08 [95% CI, 2.03-4.66]), T2DM (OR, 2.05 [95% CI, 1.57- 2.66]) or PVD (OR, 2.06 [95% CI, 1.43-2.97]) compared with controls (P<.001). Psychiatric comorbidities were more common in the patients diagnosed with scabies, including depression (OR, 3.07 [95% CI, 2.54-3.72]), anxiety (OR, 2.48 [95% CI, 2.06-2.98]), bipolar disorder (OR, 3.08 [95% CI, 2.34-4.05]), and schizophrenia (OR, 4.68 [95% CI, 2.93-7.49])(P<.001). Difficulties with activities of daily living, including running errands alone (OR, 2.32 [95% CI, 1.43-3.76]) and concentrating (OR, 5.78; 95% CI, 3.86-8.64), were more prevalent in the scabies group compared to controls (both P<.05). In a multivariate logistic regression model including unsheltered status as a covariate, all associations remained statistically significant (P<.05)(eTable 2).
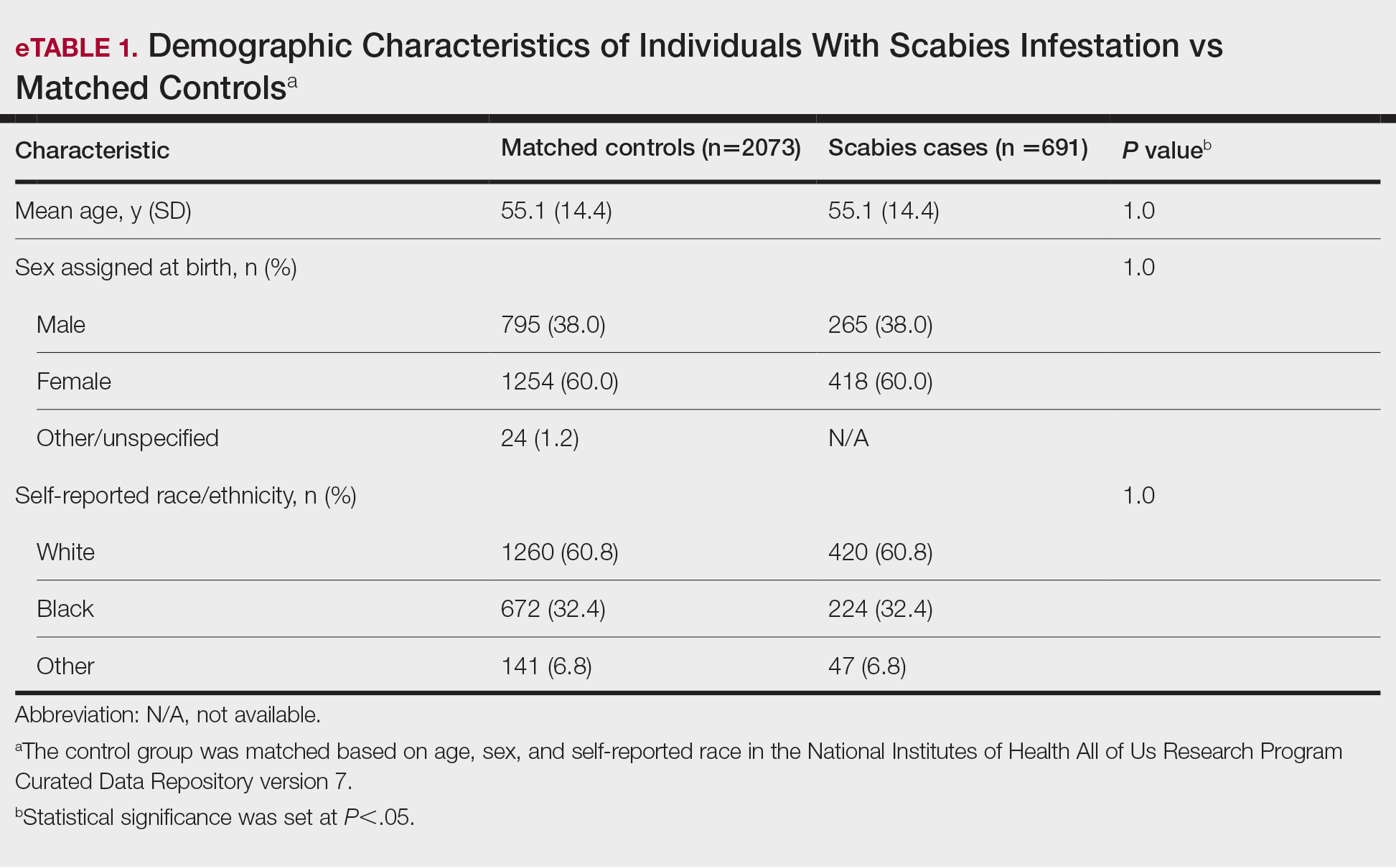
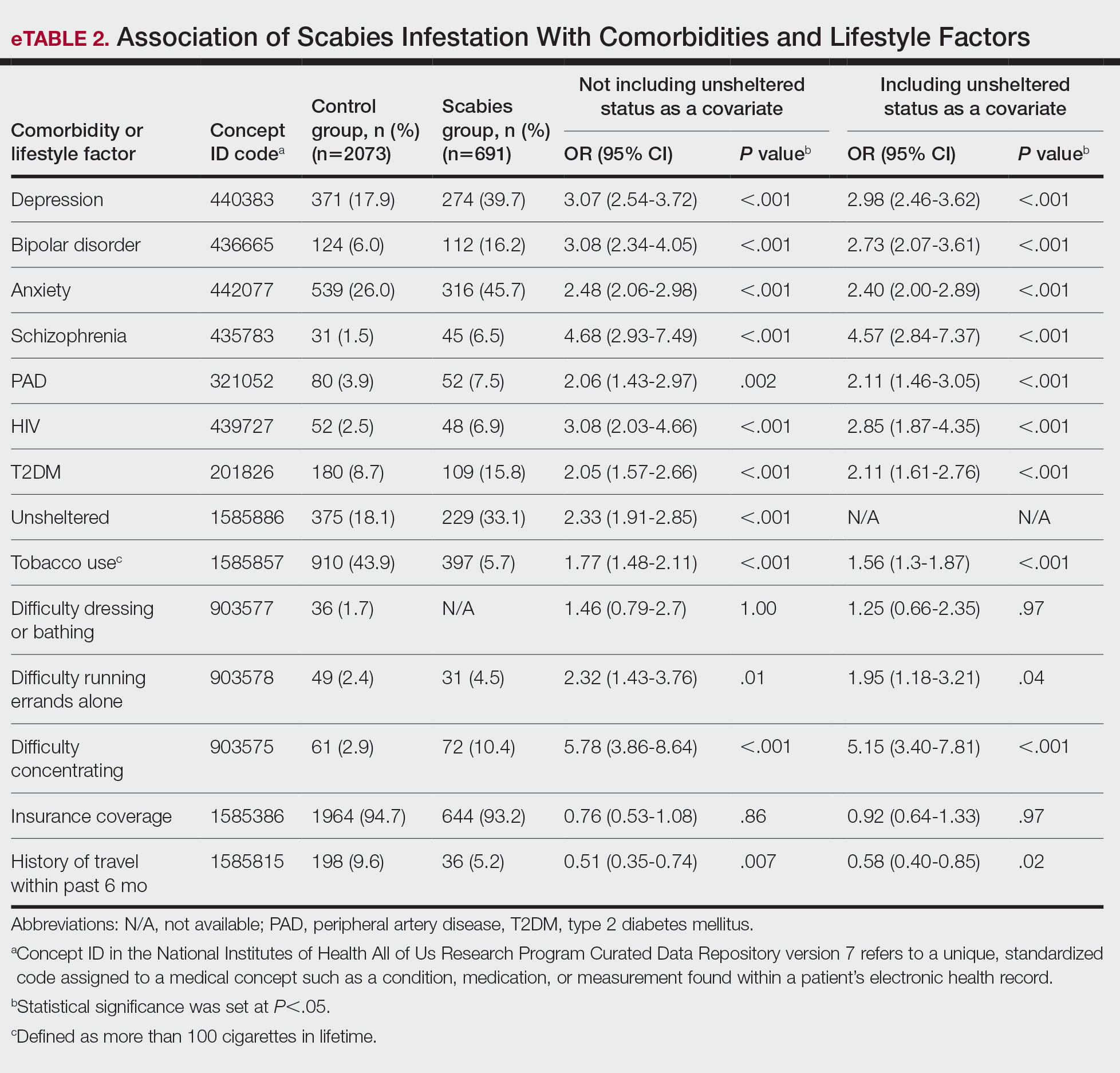
This large diverse study demonstrated an association between scabies infestation and unsheltered status. Previous studies have shown that unsheltered populations are at increased risk for many dermatologic conditions, perhaps due to decreased access to health care and social support, lack of access to hygiene facilities (eg, public showers), and increased prevalence of substance use and psychiatric disorders among this population.5 In a cross-sectional analysis of hospitalized patients, 8.6% of unsheltered patients (n=197) had an ectoparasitic disease (including scabies) compared with 1.0% of patients with stable housing (n=1018), with a 9.43-fold increased risk for ectoparasitic infestation among unsheltered patients (95% CI, 3.79-23.47; P<.001).6 Increased attention to public health initiatives among unsheltered populations— including access to hygiene facilities and increased dermatologic services—are needed, as ectoparasitic infections are both preventable and treatable, and these initiatives could reduce morbidity associated with superimposed bacterial infections for which unsheltered patients are at increased risk.6
Our results also showed that individuals diagnosed with scabies were more likely than the controls to have been diagnosed with HIV, T2DM, and PVD. Our findings are similar to those of a systematic review of immunosuppressive factors associated with crusted scabies (a severe form of scabies infestation) in which 10.2% and 15.7% of patients (n=683) had comorbid HIV and T2DM, respectively.7 A functioning cell-mediated response to scabies mite antigens limits proliferation of the human itch mite; thus, infection with HIV/AIDS, which induces the destruction of CD4+ T cells, limits the immune system’s ability to mount an effective response against these antigens. The association of scabies with T2DM likely is multifactorial; for example, chronic hyperglycemia may lead to immune system impairment, and peripheral neuropathy may reduce the itch sensation, allowing scabies mites to proliferate without removal by scratching.7 In a descriptive epidemiologic study in Japan, 11.7% of patients with scabies (N=857) had comorbid PVD.8 Peripheral vascular disease can lead to the development of ulcers, gangrene, and stasis dermatitis, all of which compromise the skin barrier and increase susceptibility to infection.9 Notably, these associations remained even when unsheltered status was considered as a confounding variable. Because individuals with HIV, T2DM, and PVD may be at higher risk for serious complications of scabies infestation (eg, secondary bacterial infections, invasive group A streptococcal infections), prompt detection and treatment of scabies are crucial in curbing morbidity in these at-risk populations.
Our study also demonstrated that psychiatric comorbidities including depression, anxiety, bipolar disorder, and schizophrenia were associated with scabies infestation, even when controlling for unsheltered status, which may have a bidirectional relationship with mental health disorders.10 In a cross-sectional study of 83 adult patients diagnosed with scabies, 72.2% (60/83) reported moderate to extremely large effect of scabies infestation on quality of life using the Dermatology Life Quality Index, and these scores positively correlated with increased Beck Depression Scale and Beck Anxiety Scale scores (rs=0.448 and rs=0.456 0.456, respectively; both P=.000). The results of this study suggest that scabies negatively impacts quality of life, which might increase symptoms of depression and anxiety.11
Studies are needed to assess whether patients with pre-existing depression and anxiety face increased risk for scabies infestation. In a retrospective case-control study using data from the National Health Insurance Research Database of Taiwan, 0.8% (58/7096) of patients with scabies (n=7096) and 0.4% of controls (n=28,375) were newly diagnosed with bipolar disorder over a 7-year period, indicating a 1.55-fold increased risk for bipolar disorder in patients with scabies compared to those without (95% CI, 1.12-2.09; P<.05).12 Future studies are needed to determine whether the relationship between bipolar disorder and scabies is bidirectional, with pre-existing bipolar disorder evaluated as a risk factor for subsequent scabies infestation. Increased difficulties with activities of daily living, including running errands independently and concentrating, were associated with scabies. These difficulties may reflect sequelae of psychiatric illness or pruritus associated with scabies affecting daily living.
Physician awareness of comorbidities and lifestyle risk factors associated with scabies infestation may improve diagnosis and prevent treatment delays. In a retrospective study at a single dermatology outpatient clinic, 45.3% of patients with scabies (n=428) had previously been misdiagnosed with another dermatologic condition, and the most common erroneous diagnosis was atopic dermatitis.13 Our study provides a framework of comorbidities and lifestyle risk factors associated with scabies infestation that dermatologists can use to stratify patients who may be at greater risk for this condition, allowing dermatologists to select appropriate treatment when clinical signs are ambiguous.
Limitations of our study included the potential for miscoding in the database, lack of information about treatment regimens employed (if any), and lack of information about the temporal relationship between associations.
In summary, it is recommended that patients with pruritus and other characteristic clinical findings of scabies receive appropriate workup for scabies regardless of risk factors; however, the medical and psychiatric comorbidities and lifestyle risk factors identified in this study may help to identify at-risk patients. Our study showed that unsheltered patients are at increased risk for scabies, potentially due to unique dermatologic challenges and lack of access to health care and hygiene facilities. Positive correlations between scabies and HIV, T2DM, and PVD suggest that patients with chronic immunocompromising illnesses who live in group homes or other crowded quarters and present with symptoms could be evaluated for scabies infestation to prevent widespread and difficult- to-control outbreaks in these communities. Based on our findings, scabies also should be included in the differential diagnosis for patients with psychiatric illness and suggestive symptoms. Early identification and treatment of scabies infestation could prevent misdiagnosis and treatment delays.
- World Health Organization. Scabies fact sheet. May 31, 2023. Accessed February 13, 2025. https://www.who.int/news-room/fact-sheets/detail/scabies
- Chandler DJ, Fuller LC. A review of scabies: an infestation more than skin deep. Dermatology. 2019;235:79-90. doi:10.1159/000495290
- Schneider S, Wu J, Tizek L, et al. Prevalence of scabies worldwidean updated systematic literature review in 2022. J Eur Acad Dermatol Venereol. 2023;37:1749-1757. doi:10.1111/jdv.19167
- Thomas C, Coates SJ, Engelman D, et al. Ectoparasites: Scabies. J Am Acad Dermatol. 2020;82:533-548. doi:10.1016/j.jaad.2019.05.109
- Henry T, Khachemoune A. Dermatologic conditions and risk factors in people experiencing homelessness (PEH): systematic review. Arch Dermatol Res. 2023;315:2795-2803. doi:10.1007/s00403-023-02722-2
- Zakaria A, Amerson EH, Kim-Lim P, et al. Characterization of dermatological diagnoses among hospitalized patients experiencing homelessness. Clin Exp Dermatol. 2022;47:117-120. doi:10.1111/ced.14828
- Bergamin G, Hudson J, Currie BJ, et al. A systematic review of immunosuppressive risk factors and comorbidities associated with the development of crusted scabies. Int J Infect Dis. 2024;143:107036. doi:10.1016/j.ijid.2024.107036
- Yamaguchi Y, Murata F, Maeda M, et al. Investigating the epidemiology and outbreaks of scabies in Japanese households, residential care facilities, and hospitals using claims data: the Longevity Improvement & Fair Evidence (LIFE) study. IJID Reg. 2024;11:100353. doi:10.1016 /j.ijregi.2024.03.008
- Raja A, Karch J, Shih AF, et al. Part II: Cutaneous manifestations of peripheral vascular disease. J Am Acad Dermatol. 2023;89:211-226. doi:10.1016/j.jaad.2021.05.077
- Barry R, Anderson J, Tran L, et al. Prevalence of mental health disorders among individuals experiencing homelessness: a systematic review and meta-analysis. JAMA Psychiatry. 2024;81:691-699. doi:10.1001 /jamapsychiatry.2024.0426
- Koc Y.ld.r.m S, Demirel Og. ut N, Erbag. c. E, et al. Scabies affects quality of life in correlation with depression and anxiety. Dermatol Pract Concept. 2023;13:E2023144. doi:10.5826/dpc.1302a144
- Lin CY, Chang FW, Yang JJ, et al. Increased risk of bipolar disorder in patients with scabies: a nationwide population-based matched-cohort study. Psychiatry Res. 2017;257:14-20. doi:10.1016 /j.psychres.2017.07.013
- Anderson KL, Strowd LC. Epidemiology, diagnosis, and treatment of scabies in a dermatology office. J Am Board Fam Med. 2017;30:78-84. doi:10.3122/jabfm.2017.01.160190
To the Editor:
Scabies infestation, which has been recognized as a neglected tropical disease by the World Health Organization since 2017, is caused by the human itch mite (Sarcoptes scabiei var hominis).1 Infected individuals experience a pruritic papular rash when the mite burrows into the epidermis, where it lives and lays eggs.2,3 Infected individuals also may develop bacterial superinfections if the skin barrier becomes compromised, leading to systemic complications and considerable morbidity.3
In countries with high human development indices, scabies outbreaks are linked to densely populated living conditions, such as those found in nursing homes or prisons.3,4 Scabies also is transmitted via sexual contact in adults. Beyond immunosuppression, little is known about other comorbid conditions or lifestyle risk factors associated with scabies infestation.2 Because scabies can mimic a range of other dermatologic conditions such as folliculitis, atopic dermatitis, and arthropod bites, misdiagnosis is common and can lead to delayed treatment and increased transmission risk.4 In this study, we sought to examine comorbid conditions and/or lifestyle risk factors associated with scabies infestation.
A matched case-control study was performed using the Registered Tier dataset of the National Institutes of Health All of Us Research Program Curated Data Repository version 7, which includes more than 400,000 unique participants aged 18 years or older from across the United States. The All of Us Research Program excludes adults who are unable to consent independently as well as incarcerated populations and children younger than 18 years. Participants diagnosed with scabies were identified using SNOMED code 62752005 and compared to a control group matched 1:4 based on age, sex, and selfidentified race. SNOMED codes also were used to identify various comorbidities and lifestyle risk factors, including depression, bipolar disorder, anxiety, schizophrenia, peripheral vascular disease (PVD), HIV, type 2 diabetes mellitus (T2DM), unsheltered status, tobacco use, difficulty with activities of daily living, insurance status, and any recent travel history. Logistic regression models were used to calculate odds ratios (ORs) and estimate effect sizes, with statistical significance set at P<.05.
We identified 691 cases of scabies infestation and 2073 controls. The average age of the patients diagnosed with scabies was 55.1 years. Seventy percent (481/691) identified as female and 32.4% (224/491) identified as Black or African American. Matched controls were similar for all analyzed demographic characteristics (P=1.0)(eTable 1). Patients diagnosed with scabies were more likely to be unsheltered (OR, 2.33 [95% CI, 1.91-2.85]), use tobacco (OR 1.77 [95% CI, 1.48-2.11]) and have a comorbid diagnosis of HIV (OR, 3.08 [95% CI, 2.03-4.66]), T2DM (OR, 2.05 [95% CI, 1.57- 2.66]) or PVD (OR, 2.06 [95% CI, 1.43-2.97]) compared with controls (P<.001). Psychiatric comorbidities were more common in the patients diagnosed with scabies, including depression (OR, 3.07 [95% CI, 2.54-3.72]), anxiety (OR, 2.48 [95% CI, 2.06-2.98]), bipolar disorder (OR, 3.08 [95% CI, 2.34-4.05]), and schizophrenia (OR, 4.68 [95% CI, 2.93-7.49])(P<.001). Difficulties with activities of daily living, including running errands alone (OR, 2.32 [95% CI, 1.43-3.76]) and concentrating (OR, 5.78; 95% CI, 3.86-8.64), were more prevalent in the scabies group compared to controls (both P<.05). In a multivariate logistic regression model including unsheltered status as a covariate, all associations remained statistically significant (P<.05)(eTable 2).


This large diverse study demonstrated an association between scabies infestation and unsheltered status. Previous studies have shown that unsheltered populations are at increased risk for many dermatologic conditions, perhaps due to decreased access to health care and social support, lack of access to hygiene facilities (eg, public showers), and increased prevalence of substance use and psychiatric disorders among this population.5 In a cross-sectional analysis of hospitalized patients, 8.6% of unsheltered patients (n=197) had an ectoparasitic disease (including scabies) compared with 1.0% of patients with stable housing (n=1018), with a 9.43-fold increased risk for ectoparasitic infestation among unsheltered patients (95% CI, 3.79-23.47; P<.001).6 Increased attention to public health initiatives among unsheltered populations— including access to hygiene facilities and increased dermatologic services—are needed, as ectoparasitic infections are both preventable and treatable, and these initiatives could reduce morbidity associated with superimposed bacterial infections for which unsheltered patients are at increased risk.6
Our results also showed that individuals diagnosed with scabies were more likely than the controls to have been diagnosed with HIV, T2DM, and PVD. Our findings are similar to those of a systematic review of immunosuppressive factors associated with crusted scabies (a severe form of scabies infestation) in which 10.2% and 15.7% of patients (n=683) had comorbid HIV and T2DM, respectively.7 A functioning cell-mediated response to scabies mite antigens limits proliferation of the human itch mite; thus, infection with HIV/AIDS, which induces the destruction of CD4+ T cells, limits the immune system’s ability to mount an effective response against these antigens. The association of scabies with T2DM likely is multifactorial; for example, chronic hyperglycemia may lead to immune system impairment, and peripheral neuropathy may reduce the itch sensation, allowing scabies mites to proliferate without removal by scratching.7 In a descriptive epidemiologic study in Japan, 11.7% of patients with scabies (N=857) had comorbid PVD.8 Peripheral vascular disease can lead to the development of ulcers, gangrene, and stasis dermatitis, all of which compromise the skin barrier and increase susceptibility to infection.9 Notably, these associations remained even when unsheltered status was considered as a confounding variable. Because individuals with HIV, T2DM, and PVD may be at higher risk for serious complications of scabies infestation (eg, secondary bacterial infections, invasive group A streptococcal infections), prompt detection and treatment of scabies are crucial in curbing morbidity in these at-risk populations.
Our study also demonstrated that psychiatric comorbidities including depression, anxiety, bipolar disorder, and schizophrenia were associated with scabies infestation, even when controlling for unsheltered status, which may have a bidirectional relationship with mental health disorders.10 In a cross-sectional study of 83 adult patients diagnosed with scabies, 72.2% (60/83) reported moderate to extremely large effect of scabies infestation on quality of life using the Dermatology Life Quality Index, and these scores positively correlated with increased Beck Depression Scale and Beck Anxiety Scale scores (rs=0.448 and rs=0.456 0.456, respectively; both P=.000). The results of this study suggest that scabies negatively impacts quality of life, which might increase symptoms of depression and anxiety.11
Studies are needed to assess whether patients with pre-existing depression and anxiety face increased risk for scabies infestation. In a retrospective case-control study using data from the National Health Insurance Research Database of Taiwan, 0.8% (58/7096) of patients with scabies (n=7096) and 0.4% of controls (n=28,375) were newly diagnosed with bipolar disorder over a 7-year period, indicating a 1.55-fold increased risk for bipolar disorder in patients with scabies compared to those without (95% CI, 1.12-2.09; P<.05).12 Future studies are needed to determine whether the relationship between bipolar disorder and scabies is bidirectional, with pre-existing bipolar disorder evaluated as a risk factor for subsequent scabies infestation. Increased difficulties with activities of daily living, including running errands independently and concentrating, were associated with scabies. These difficulties may reflect sequelae of psychiatric illness or pruritus associated with scabies affecting daily living.
Physician awareness of comorbidities and lifestyle risk factors associated with scabies infestation may improve diagnosis and prevent treatment delays. In a retrospective study at a single dermatology outpatient clinic, 45.3% of patients with scabies (n=428) had previously been misdiagnosed with another dermatologic condition, and the most common erroneous diagnosis was atopic dermatitis.13 Our study provides a framework of comorbidities and lifestyle risk factors associated with scabies infestation that dermatologists can use to stratify patients who may be at greater risk for this condition, allowing dermatologists to select appropriate treatment when clinical signs are ambiguous.
Limitations of our study included the potential for miscoding in the database, lack of information about treatment regimens employed (if any), and lack of information about the temporal relationship between associations.
In summary, it is recommended that patients with pruritus and other characteristic clinical findings of scabies receive appropriate workup for scabies regardless of risk factors; however, the medical and psychiatric comorbidities and lifestyle risk factors identified in this study may help to identify at-risk patients. Our study showed that unsheltered patients are at increased risk for scabies, potentially due to unique dermatologic challenges and lack of access to health care and hygiene facilities. Positive correlations between scabies and HIV, T2DM, and PVD suggest that patients with chronic immunocompromising illnesses who live in group homes or other crowded quarters and present with symptoms could be evaluated for scabies infestation to prevent widespread and difficult- to-control outbreaks in these communities. Based on our findings, scabies also should be included in the differential diagnosis for patients with psychiatric illness and suggestive symptoms. Early identification and treatment of scabies infestation could prevent misdiagnosis and treatment delays.
To the Editor:
Scabies infestation, which has been recognized as a neglected tropical disease by the World Health Organization since 2017, is caused by the human itch mite (Sarcoptes scabiei var hominis).1 Infected individuals experience a pruritic papular rash when the mite burrows into the epidermis, where it lives and lays eggs.2,3 Infected individuals also may develop bacterial superinfections if the skin barrier becomes compromised, leading to systemic complications and considerable morbidity.3
In countries with high human development indices, scabies outbreaks are linked to densely populated living conditions, such as those found in nursing homes or prisons.3,4 Scabies also is transmitted via sexual contact in adults. Beyond immunosuppression, little is known about other comorbid conditions or lifestyle risk factors associated with scabies infestation.2 Because scabies can mimic a range of other dermatologic conditions such as folliculitis, atopic dermatitis, and arthropod bites, misdiagnosis is common and can lead to delayed treatment and increased transmission risk.4 In this study, we sought to examine comorbid conditions and/or lifestyle risk factors associated with scabies infestation.
A matched case-control study was performed using the Registered Tier dataset of the National Institutes of Health All of Us Research Program Curated Data Repository version 7, which includes more than 400,000 unique participants aged 18 years or older from across the United States. The All of Us Research Program excludes adults who are unable to consent independently as well as incarcerated populations and children younger than 18 years. Participants diagnosed with scabies were identified using SNOMED code 62752005 and compared to a control group matched 1:4 based on age, sex, and selfidentified race. SNOMED codes also were used to identify various comorbidities and lifestyle risk factors, including depression, bipolar disorder, anxiety, schizophrenia, peripheral vascular disease (PVD), HIV, type 2 diabetes mellitus (T2DM), unsheltered status, tobacco use, difficulty with activities of daily living, insurance status, and any recent travel history. Logistic regression models were used to calculate odds ratios (ORs) and estimate effect sizes, with statistical significance set at P<.05.
We identified 691 cases of scabies infestation and 2073 controls. The average age of the patients diagnosed with scabies was 55.1 years. Seventy percent (481/691) identified as female and 32.4% (224/491) identified as Black or African American. Matched controls were similar for all analyzed demographic characteristics (P=1.0)(eTable 1). Patients diagnosed with scabies were more likely to be unsheltered (OR, 2.33 [95% CI, 1.91-2.85]), use tobacco (OR 1.77 [95% CI, 1.48-2.11]) and have a comorbid diagnosis of HIV (OR, 3.08 [95% CI, 2.03-4.66]), T2DM (OR, 2.05 [95% CI, 1.57- 2.66]) or PVD (OR, 2.06 [95% CI, 1.43-2.97]) compared with controls (P<.001). Psychiatric comorbidities were more common in the patients diagnosed with scabies, including depression (OR, 3.07 [95% CI, 2.54-3.72]), anxiety (OR, 2.48 [95% CI, 2.06-2.98]), bipolar disorder (OR, 3.08 [95% CI, 2.34-4.05]), and schizophrenia (OR, 4.68 [95% CI, 2.93-7.49])(P<.001). Difficulties with activities of daily living, including running errands alone (OR, 2.32 [95% CI, 1.43-3.76]) and concentrating (OR, 5.78; 95% CI, 3.86-8.64), were more prevalent in the scabies group compared to controls (both P<.05). In a multivariate logistic regression model including unsheltered status as a covariate, all associations remained statistically significant (P<.05)(eTable 2).


This large diverse study demonstrated an association between scabies infestation and unsheltered status. Previous studies have shown that unsheltered populations are at increased risk for many dermatologic conditions, perhaps due to decreased access to health care and social support, lack of access to hygiene facilities (eg, public showers), and increased prevalence of substance use and psychiatric disorders among this population.5 In a cross-sectional analysis of hospitalized patients, 8.6% of unsheltered patients (n=197) had an ectoparasitic disease (including scabies) compared with 1.0% of patients with stable housing (n=1018), with a 9.43-fold increased risk for ectoparasitic infestation among unsheltered patients (95% CI, 3.79-23.47; P<.001).6 Increased attention to public health initiatives among unsheltered populations— including access to hygiene facilities and increased dermatologic services—are needed, as ectoparasitic infections are both preventable and treatable, and these initiatives could reduce morbidity associated with superimposed bacterial infections for which unsheltered patients are at increased risk.6
Our results also showed that individuals diagnosed with scabies were more likely than the controls to have been diagnosed with HIV, T2DM, and PVD. Our findings are similar to those of a systematic review of immunosuppressive factors associated with crusted scabies (a severe form of scabies infestation) in which 10.2% and 15.7% of patients (n=683) had comorbid HIV and T2DM, respectively.7 A functioning cell-mediated response to scabies mite antigens limits proliferation of the human itch mite; thus, infection with HIV/AIDS, which induces the destruction of CD4+ T cells, limits the immune system’s ability to mount an effective response against these antigens. The association of scabies with T2DM likely is multifactorial; for example, chronic hyperglycemia may lead to immune system impairment, and peripheral neuropathy may reduce the itch sensation, allowing scabies mites to proliferate without removal by scratching.7 In a descriptive epidemiologic study in Japan, 11.7% of patients with scabies (N=857) had comorbid PVD.8 Peripheral vascular disease can lead to the development of ulcers, gangrene, and stasis dermatitis, all of which compromise the skin barrier and increase susceptibility to infection.9 Notably, these associations remained even when unsheltered status was considered as a confounding variable. Because individuals with HIV, T2DM, and PVD may be at higher risk for serious complications of scabies infestation (eg, secondary bacterial infections, invasive group A streptococcal infections), prompt detection and treatment of scabies are crucial in curbing morbidity in these at-risk populations.
Our study also demonstrated that psychiatric comorbidities including depression, anxiety, bipolar disorder, and schizophrenia were associated with scabies infestation, even when controlling for unsheltered status, which may have a bidirectional relationship with mental health disorders.10 In a cross-sectional study of 83 adult patients diagnosed with scabies, 72.2% (60/83) reported moderate to extremely large effect of scabies infestation on quality of life using the Dermatology Life Quality Index, and these scores positively correlated with increased Beck Depression Scale and Beck Anxiety Scale scores (rs=0.448 and rs=0.456 0.456, respectively; both P=.000). The results of this study suggest that scabies negatively impacts quality of life, which might increase symptoms of depression and anxiety.11
Studies are needed to assess whether patients with pre-existing depression and anxiety face increased risk for scabies infestation. In a retrospective case-control study using data from the National Health Insurance Research Database of Taiwan, 0.8% (58/7096) of patients with scabies (n=7096) and 0.4% of controls (n=28,375) were newly diagnosed with bipolar disorder over a 7-year period, indicating a 1.55-fold increased risk for bipolar disorder in patients with scabies compared to those without (95% CI, 1.12-2.09; P<.05).12 Future studies are needed to determine whether the relationship between bipolar disorder and scabies is bidirectional, with pre-existing bipolar disorder evaluated as a risk factor for subsequent scabies infestation. Increased difficulties with activities of daily living, including running errands independently and concentrating, were associated with scabies. These difficulties may reflect sequelae of psychiatric illness or pruritus associated with scabies affecting daily living.
Physician awareness of comorbidities and lifestyle risk factors associated with scabies infestation may improve diagnosis and prevent treatment delays. In a retrospective study at a single dermatology outpatient clinic, 45.3% of patients with scabies (n=428) had previously been misdiagnosed with another dermatologic condition, and the most common erroneous diagnosis was atopic dermatitis.13 Our study provides a framework of comorbidities and lifestyle risk factors associated with scabies infestation that dermatologists can use to stratify patients who may be at greater risk for this condition, allowing dermatologists to select appropriate treatment when clinical signs are ambiguous.
Limitations of our study included the potential for miscoding in the database, lack of information about treatment regimens employed (if any), and lack of information about the temporal relationship between associations.
In summary, it is recommended that patients with pruritus and other characteristic clinical findings of scabies receive appropriate workup for scabies regardless of risk factors; however, the medical and psychiatric comorbidities and lifestyle risk factors identified in this study may help to identify at-risk patients. Our study showed that unsheltered patients are at increased risk for scabies, potentially due to unique dermatologic challenges and lack of access to health care and hygiene facilities. Positive correlations between scabies and HIV, T2DM, and PVD suggest that patients with chronic immunocompromising illnesses who live in group homes or other crowded quarters and present with symptoms could be evaluated for scabies infestation to prevent widespread and difficult- to-control outbreaks in these communities. Based on our findings, scabies also should be included in the differential diagnosis for patients with psychiatric illness and suggestive symptoms. Early identification and treatment of scabies infestation could prevent misdiagnosis and treatment delays.
- World Health Organization. Scabies fact sheet. May 31, 2023. Accessed February 13, 2025. https://www.who.int/news-room/fact-sheets/detail/scabies
- Chandler DJ, Fuller LC. A review of scabies: an infestation more than skin deep. Dermatology. 2019;235:79-90. doi:10.1159/000495290
- Schneider S, Wu J, Tizek L, et al. Prevalence of scabies worldwidean updated systematic literature review in 2022. J Eur Acad Dermatol Venereol. 2023;37:1749-1757. doi:10.1111/jdv.19167
- Thomas C, Coates SJ, Engelman D, et al. Ectoparasites: Scabies. J Am Acad Dermatol. 2020;82:533-548. doi:10.1016/j.jaad.2019.05.109
- Henry T, Khachemoune A. Dermatologic conditions and risk factors in people experiencing homelessness (PEH): systematic review. Arch Dermatol Res. 2023;315:2795-2803. doi:10.1007/s00403-023-02722-2
- Zakaria A, Amerson EH, Kim-Lim P, et al. Characterization of dermatological diagnoses among hospitalized patients experiencing homelessness. Clin Exp Dermatol. 2022;47:117-120. doi:10.1111/ced.14828
- Bergamin G, Hudson J, Currie BJ, et al. A systematic review of immunosuppressive risk factors and comorbidities associated with the development of crusted scabies. Int J Infect Dis. 2024;143:107036. doi:10.1016/j.ijid.2024.107036
- Yamaguchi Y, Murata F, Maeda M, et al. Investigating the epidemiology and outbreaks of scabies in Japanese households, residential care facilities, and hospitals using claims data: the Longevity Improvement & Fair Evidence (LIFE) study. IJID Reg. 2024;11:100353. doi:10.1016 /j.ijregi.2024.03.008
- Raja A, Karch J, Shih AF, et al. Part II: Cutaneous manifestations of peripheral vascular disease. J Am Acad Dermatol. 2023;89:211-226. doi:10.1016/j.jaad.2021.05.077
- Barry R, Anderson J, Tran L, et al. Prevalence of mental health disorders among individuals experiencing homelessness: a systematic review and meta-analysis. JAMA Psychiatry. 2024;81:691-699. doi:10.1001 /jamapsychiatry.2024.0426
- Koc Y.ld.r.m S, Demirel Og. ut N, Erbag. c. E, et al. Scabies affects quality of life in correlation with depression and anxiety. Dermatol Pract Concept. 2023;13:E2023144. doi:10.5826/dpc.1302a144
- Lin CY, Chang FW, Yang JJ, et al. Increased risk of bipolar disorder in patients with scabies: a nationwide population-based matched-cohort study. Psychiatry Res. 2017;257:14-20. doi:10.1016 /j.psychres.2017.07.013
- Anderson KL, Strowd LC. Epidemiology, diagnosis, and treatment of scabies in a dermatology office. J Am Board Fam Med. 2017;30:78-84. doi:10.3122/jabfm.2017.01.160190
- World Health Organization. Scabies fact sheet. May 31, 2023. Accessed February 13, 2025. https://www.who.int/news-room/fact-sheets/detail/scabies
- Chandler DJ, Fuller LC. A review of scabies: an infestation more than skin deep. Dermatology. 2019;235:79-90. doi:10.1159/000495290
- Schneider S, Wu J, Tizek L, et al. Prevalence of scabies worldwidean updated systematic literature review in 2022. J Eur Acad Dermatol Venereol. 2023;37:1749-1757. doi:10.1111/jdv.19167
- Thomas C, Coates SJ, Engelman D, et al. Ectoparasites: Scabies. J Am Acad Dermatol. 2020;82:533-548. doi:10.1016/j.jaad.2019.05.109
- Henry T, Khachemoune A. Dermatologic conditions and risk factors in people experiencing homelessness (PEH): systematic review. Arch Dermatol Res. 2023;315:2795-2803. doi:10.1007/s00403-023-02722-2
- Zakaria A, Amerson EH, Kim-Lim P, et al. Characterization of dermatological diagnoses among hospitalized patients experiencing homelessness. Clin Exp Dermatol. 2022;47:117-120. doi:10.1111/ced.14828
- Bergamin G, Hudson J, Currie BJ, et al. A systematic review of immunosuppressive risk factors and comorbidities associated with the development of crusted scabies. Int J Infect Dis. 2024;143:107036. doi:10.1016/j.ijid.2024.107036
- Yamaguchi Y, Murata F, Maeda M, et al. Investigating the epidemiology and outbreaks of scabies in Japanese households, residential care facilities, and hospitals using claims data: the Longevity Improvement & Fair Evidence (LIFE) study. IJID Reg. 2024;11:100353. doi:10.1016 /j.ijregi.2024.03.008
- Raja A, Karch J, Shih AF, et al. Part II: Cutaneous manifestations of peripheral vascular disease. J Am Acad Dermatol. 2023;89:211-226. doi:10.1016/j.jaad.2021.05.077
- Barry R, Anderson J, Tran L, et al. Prevalence of mental health disorders among individuals experiencing homelessness: a systematic review and meta-analysis. JAMA Psychiatry. 2024;81:691-699. doi:10.1001 /jamapsychiatry.2024.0426
- Koc Y.ld.r.m S, Demirel Og. ut N, Erbag. c. E, et al. Scabies affects quality of life in correlation with depression and anxiety. Dermatol Pract Concept. 2023;13:E2023144. doi:10.5826/dpc.1302a144
- Lin CY, Chang FW, Yang JJ, et al. Increased risk of bipolar disorder in patients with scabies: a nationwide population-based matched-cohort study. Psychiatry Res. 2017;257:14-20. doi:10.1016 /j.psychres.2017.07.013
- Anderson KL, Strowd LC. Epidemiology, diagnosis, and treatment of scabies in a dermatology office. J Am Board Fam Med. 2017;30:78-84. doi:10.3122/jabfm.2017.01.160190
Comorbidities and Lifestyle Risk Factors Associated With Scabies Infestation
Comorbidities and Lifestyle Risk Factors Associated With Scabies Infestation
PRACTICE POINTS
- Scabies infestation is caused by the human itch mite (Sarcoptes scabiei var hominis) and can be spread via sexual contact in adults.
- Crowded living conditions are associated with scabies infestation in countries with high human development indices, such as the United States.
- Patients with certain comorbid conditions or lifestyle risk factors should be screened for scabies infestation when presenting with pruritus and other characteristic clinical findings.
Most Kids With COVID-Linked MIS-C Recover by 6 Months
Children who were severely ill with multisystem inflammatory syndrome in children (MIS-C) related to COVID-19 infection appear to show excellent cardiovascular and noncardiovascular outcomes by 6 months, according to data published in JAMA Pediatrics.
MIS-C is a life-threatening complication of COVID-19 infection and data on outcomes are limited, wrote the authors, led by Dongngan T. Truong, MD, MSSI, with Children’s Healthcare of Atlanta Cardiology, Emory University School of Medicine in Atlanta, Georgia. These 6-month results are from the Long-Term Outcomes After the Multisystem Inflammatory Syndrome in Children (MUSIC) study, sponsored by the National Heart, Lung, and Blood Institute.
Researchers found in this cohort study of 1204 participants that by 6 months after hospital discharge, 99% had normalization of left ventricular systolic function, and 92.3% had normalized coronary artery dimensions. More than 95% reported being more than 90% back to baseline health.
Patient-Reported Outcomes Measurement Information Systems (PROMIS) Global Health scores were at least equivalent to prepandemic population normative values. PROMIS Global Health parent/guardian proxy median T scores for fatigue, global health, and pain interference improved significantly from 2 weeks to 6 months: fatigue, 56.1 vs 48.9; global health, 48.8 vs 51.3; pain interference, 53.0 vs 43.3 (P < .001).
The most common symptoms reported at 2 weeks were fatigue (15.9%) and low stamina/energy (9.2%); both decreased to 3.4% and 3.3%, respectively, by 6 months. The most common cardiovascular symptom at 2 weeks was palpitations (1.5%), which decreased to 0.6%.
Chest Pain Increased Over Time
Reports of chest pain, however, reportedly increased over time, with 1.3% reporting chest pain at rest at 2 weeks and 2.2% at 6 months. Although gastrointestinal symptoms were common during the acute MIS-C, only 5.3% of respondents reported those symptoms at 2 weeks.
Children in the cohort had a median age of 9 years, and 60% were men. They self-identified with the following races and ethnicities: American Indian or Alaska Native (0.1%), Asian (3.3%), Black (27.0%), Hawaiian Native or Other Pacific Islander (0.2%), Hispanic or Latino (26.9%), multiracial (2.7%), White (31.2%), other (1.0%), and unknown or refused to specify (7.6%). Authors wrote that the cohort was followed-up to 2 years after illness onset and long-term results are not yet known.
Time to Exhale
David J. Goldberg, MD, with the Cardiac Center, Children’s Hospital of Philadelphia, Pennsylvania, and colleagues, wrote in an accompanying editorial that “the decreased frequency of the disease along (with) the reassuring reports on midterm outcomes can allow the pediatric community a moment of collective exhale.”
The editorialists note that of those who initially presented with myocardial dysfunction, all but one patient evaluated had a normal ejection fraction at follow-up. Energy, sleep, appetite, cognition, and mood also normalized by midterm.
“The results of the MUSIC study add to the emerging midterm outcomes data suggesting a near-complete cardiovascular recovery in the overwhelming majority of patients who develop MIS-C,” Goldberg and colleagues wrote. “Despite initial concerns, driven by the severity of acute presentation at diagnosis and longer-term questions that remain (for example, does coronary microvascular dysfunction persist even after normalization of coronary artery z score?), these data suggest an encouraging outlook for the long-term health of affected children.”
The Centers for Disease Control and Prevention and other agencies have reported a declining overall incidence of MIS-C and highlighted the protective value of vaccination.
The editorialists add, however, that while the drop in MIS-C cases is encouraging, cases are still reported, especially amid high viral activity periods, “and nearly half of affected children continue to require intensive care in the acute phase of illness.”
Truong reported grants from the National Institutes of Health and serving as coprincipal investigator for Pfizer for research on COVID-19 vaccine-associated myocarditis funded by Pfizer and occurring through the framework of the National Heart, Lung, and Blood Institute’s Pediatric Heart Network outside the submitted work. One coauthor reported grants from Pfizer and Boston Scientific outside the submitted work. One coauthor reported receiving grants from Additional Ventures Foundation outside the submitted work. One coauthor reported receiving consultant fees from Amryt Pharma, Chiesi, Esperion, and Ultragenyx outside the submitted work. A coauthor reported receiving consultant fees from Larimar Therapeutics for mitochondrial therapies outside the submitted work. One coauthor reported being an employee of Takeda Pharmaceuticals since July 2023. One editorialist reported grants from Childhood Arthritis and Rheumatology Research Alliance and the Arthritis Foundation, Academy Health, and the Gordon and Betty Moore Foundation during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children who were severely ill with multisystem inflammatory syndrome in children (MIS-C) related to COVID-19 infection appear to show excellent cardiovascular and noncardiovascular outcomes by 6 months, according to data published in JAMA Pediatrics.
MIS-C is a life-threatening complication of COVID-19 infection and data on outcomes are limited, wrote the authors, led by Dongngan T. Truong, MD, MSSI, with Children’s Healthcare of Atlanta Cardiology, Emory University School of Medicine in Atlanta, Georgia. These 6-month results are from the Long-Term Outcomes After the Multisystem Inflammatory Syndrome in Children (MUSIC) study, sponsored by the National Heart, Lung, and Blood Institute.
Researchers found in this cohort study of 1204 participants that by 6 months after hospital discharge, 99% had normalization of left ventricular systolic function, and 92.3% had normalized coronary artery dimensions. More than 95% reported being more than 90% back to baseline health.
Patient-Reported Outcomes Measurement Information Systems (PROMIS) Global Health scores were at least equivalent to prepandemic population normative values. PROMIS Global Health parent/guardian proxy median T scores for fatigue, global health, and pain interference improved significantly from 2 weeks to 6 months: fatigue, 56.1 vs 48.9; global health, 48.8 vs 51.3; pain interference, 53.0 vs 43.3 (P < .001).
The most common symptoms reported at 2 weeks were fatigue (15.9%) and low stamina/energy (9.2%); both decreased to 3.4% and 3.3%, respectively, by 6 months. The most common cardiovascular symptom at 2 weeks was palpitations (1.5%), which decreased to 0.6%.
Chest Pain Increased Over Time
Reports of chest pain, however, reportedly increased over time, with 1.3% reporting chest pain at rest at 2 weeks and 2.2% at 6 months. Although gastrointestinal symptoms were common during the acute MIS-C, only 5.3% of respondents reported those symptoms at 2 weeks.
Children in the cohort had a median age of 9 years, and 60% were men. They self-identified with the following races and ethnicities: American Indian or Alaska Native (0.1%), Asian (3.3%), Black (27.0%), Hawaiian Native or Other Pacific Islander (0.2%), Hispanic or Latino (26.9%), multiracial (2.7%), White (31.2%), other (1.0%), and unknown or refused to specify (7.6%). Authors wrote that the cohort was followed-up to 2 years after illness onset and long-term results are not yet known.
Time to Exhale
David J. Goldberg, MD, with the Cardiac Center, Children’s Hospital of Philadelphia, Pennsylvania, and colleagues, wrote in an accompanying editorial that “the decreased frequency of the disease along (with) the reassuring reports on midterm outcomes can allow the pediatric community a moment of collective exhale.”
The editorialists note that of those who initially presented with myocardial dysfunction, all but one patient evaluated had a normal ejection fraction at follow-up. Energy, sleep, appetite, cognition, and mood also normalized by midterm.
“The results of the MUSIC study add to the emerging midterm outcomes data suggesting a near-complete cardiovascular recovery in the overwhelming majority of patients who develop MIS-C,” Goldberg and colleagues wrote. “Despite initial concerns, driven by the severity of acute presentation at diagnosis and longer-term questions that remain (for example, does coronary microvascular dysfunction persist even after normalization of coronary artery z score?), these data suggest an encouraging outlook for the long-term health of affected children.”
The Centers for Disease Control and Prevention and other agencies have reported a declining overall incidence of MIS-C and highlighted the protective value of vaccination.
The editorialists add, however, that while the drop in MIS-C cases is encouraging, cases are still reported, especially amid high viral activity periods, “and nearly half of affected children continue to require intensive care in the acute phase of illness.”
Truong reported grants from the National Institutes of Health and serving as coprincipal investigator for Pfizer for research on COVID-19 vaccine-associated myocarditis funded by Pfizer and occurring through the framework of the National Heart, Lung, and Blood Institute’s Pediatric Heart Network outside the submitted work. One coauthor reported grants from Pfizer and Boston Scientific outside the submitted work. One coauthor reported receiving grants from Additional Ventures Foundation outside the submitted work. One coauthor reported receiving consultant fees from Amryt Pharma, Chiesi, Esperion, and Ultragenyx outside the submitted work. A coauthor reported receiving consultant fees from Larimar Therapeutics for mitochondrial therapies outside the submitted work. One coauthor reported being an employee of Takeda Pharmaceuticals since July 2023. One editorialist reported grants from Childhood Arthritis and Rheumatology Research Alliance and the Arthritis Foundation, Academy Health, and the Gordon and Betty Moore Foundation during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children who were severely ill with multisystem inflammatory syndrome in children (MIS-C) related to COVID-19 infection appear to show excellent cardiovascular and noncardiovascular outcomes by 6 months, according to data published in JAMA Pediatrics.
MIS-C is a life-threatening complication of COVID-19 infection and data on outcomes are limited, wrote the authors, led by Dongngan T. Truong, MD, MSSI, with Children’s Healthcare of Atlanta Cardiology, Emory University School of Medicine in Atlanta, Georgia. These 6-month results are from the Long-Term Outcomes After the Multisystem Inflammatory Syndrome in Children (MUSIC) study, sponsored by the National Heart, Lung, and Blood Institute.
Researchers found in this cohort study of 1204 participants that by 6 months after hospital discharge, 99% had normalization of left ventricular systolic function, and 92.3% had normalized coronary artery dimensions. More than 95% reported being more than 90% back to baseline health.
Patient-Reported Outcomes Measurement Information Systems (PROMIS) Global Health scores were at least equivalent to prepandemic population normative values. PROMIS Global Health parent/guardian proxy median T scores for fatigue, global health, and pain interference improved significantly from 2 weeks to 6 months: fatigue, 56.1 vs 48.9; global health, 48.8 vs 51.3; pain interference, 53.0 vs 43.3 (P < .001).
The most common symptoms reported at 2 weeks were fatigue (15.9%) and low stamina/energy (9.2%); both decreased to 3.4% and 3.3%, respectively, by 6 months. The most common cardiovascular symptom at 2 weeks was palpitations (1.5%), which decreased to 0.6%.
Chest Pain Increased Over Time
Reports of chest pain, however, reportedly increased over time, with 1.3% reporting chest pain at rest at 2 weeks and 2.2% at 6 months. Although gastrointestinal symptoms were common during the acute MIS-C, only 5.3% of respondents reported those symptoms at 2 weeks.
Children in the cohort had a median age of 9 years, and 60% were men. They self-identified with the following races and ethnicities: American Indian or Alaska Native (0.1%), Asian (3.3%), Black (27.0%), Hawaiian Native or Other Pacific Islander (0.2%), Hispanic or Latino (26.9%), multiracial (2.7%), White (31.2%), other (1.0%), and unknown or refused to specify (7.6%). Authors wrote that the cohort was followed-up to 2 years after illness onset and long-term results are not yet known.
Time to Exhale
David J. Goldberg, MD, with the Cardiac Center, Children’s Hospital of Philadelphia, Pennsylvania, and colleagues, wrote in an accompanying editorial that “the decreased frequency of the disease along (with) the reassuring reports on midterm outcomes can allow the pediatric community a moment of collective exhale.”
The editorialists note that of those who initially presented with myocardial dysfunction, all but one patient evaluated had a normal ejection fraction at follow-up. Energy, sleep, appetite, cognition, and mood also normalized by midterm.
“The results of the MUSIC study add to the emerging midterm outcomes data suggesting a near-complete cardiovascular recovery in the overwhelming majority of patients who develop MIS-C,” Goldberg and colleagues wrote. “Despite initial concerns, driven by the severity of acute presentation at diagnosis and longer-term questions that remain (for example, does coronary microvascular dysfunction persist even after normalization of coronary artery z score?), these data suggest an encouraging outlook for the long-term health of affected children.”
The Centers for Disease Control and Prevention and other agencies have reported a declining overall incidence of MIS-C and highlighted the protective value of vaccination.
The editorialists add, however, that while the drop in MIS-C cases is encouraging, cases are still reported, especially amid high viral activity periods, “and nearly half of affected children continue to require intensive care in the acute phase of illness.”
Truong reported grants from the National Institutes of Health and serving as coprincipal investigator for Pfizer for research on COVID-19 vaccine-associated myocarditis funded by Pfizer and occurring through the framework of the National Heart, Lung, and Blood Institute’s Pediatric Heart Network outside the submitted work. One coauthor reported grants from Pfizer and Boston Scientific outside the submitted work. One coauthor reported receiving grants from Additional Ventures Foundation outside the submitted work. One coauthor reported receiving consultant fees from Amryt Pharma, Chiesi, Esperion, and Ultragenyx outside the submitted work. A coauthor reported receiving consultant fees from Larimar Therapeutics for mitochondrial therapies outside the submitted work. One coauthor reported being an employee of Takeda Pharmaceuticals since July 2023. One editorialist reported grants from Childhood Arthritis and Rheumatology Research Alliance and the Arthritis Foundation, Academy Health, and the Gordon and Betty Moore Foundation during the conduct of the study.
A version of this article first appeared on Medscape.com.
FROM JAMA PEDIATRICS
Valaciclovir Shows Promise in Preventing Herpes Zoster During Anifrolumab Treatment for Lupus
TOPLINE:
The use of valaciclovir as prophylaxis prevents herpes zoster (HZ) in patients with systemic lupus erythematosus (SLE) receiving anifrolumab treatment, with no cases of zoster reported during the follow-up period in patients receiving valaciclovir.
METHODOLOGY:
- Anifrolumab, a human monoclonal antibody binding to type I interferon receptor subunit 1, increases the risk for HZ in patients with SLE; however, specific recommendations to prevent HZ are currently nonexistent for patients with SLE receiving anifrolumab.
- Researchers conducted a multicenter observational study in France from November 2021 to July 2024 to evaluate the prophylactic benefits of valaciclovir in 132 patients with SLE (mean age, 42 years; 92% women) treated with anifrolumab for ≥ 3 months.
- Among these patients, 87 received either 500 mg/d valaciclovir (n = 69) or 1000 mg/d valaciclovir (n = 18) as prophylaxis, whereas 45 did not receive valaciclovir.
- The patients were followed up for a median duration of 234 days under anifrolumab treatment, with monitoring for the development of herpes zoster.
TAKEAWAY:
- The risk for HZ was significantly lower in patients who received valaciclovir than in those who did not (hazard ratio, 0.08; P = .01).
- None of the patients treated with valaciclovir developed HZ during the survey period.
- The frequency of HZ in patients who did not receive valaciclovir increased progressively from 2.2% at 3 months to 6.2% at 6 months, reaching 23% at 12 months.
- None of the reported cases of HZ required hospitalization or led to anifrolumab discontinuation, although one patient developed neuralgia.
IN PRACTICE:
“Prophylactic treatment with valaciclovir is effective for preventing HZ [herpes zoster] infection in SLE patients treated with anifrolumab,” the authors wrote. “This finding is particularly relevant for SLE patients who cannot receive the recombinant HZ vaccine or for whom it is unavailable,” they added.
SOURCE:
The study was led by Ludovic Trefond, MD, PhD, Centre Hospitalier Universitaire de Clermont-Ferrand in France. It was published online on January 4, 2025, in RMD Open.
LIMITATIONS:
The observational design of the study and the low number of herpes zoster events during the follow-up period may have affected the robustness of the findings.
DISCLOSURES:
The authors did not receive any specific grants. Some authors reported having financial relationships with various pharmaceutical companies.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of valaciclovir as prophylaxis prevents herpes zoster (HZ) in patients with systemic lupus erythematosus (SLE) receiving anifrolumab treatment, with no cases of zoster reported during the follow-up period in patients receiving valaciclovir.
METHODOLOGY:
- Anifrolumab, a human monoclonal antibody binding to type I interferon receptor subunit 1, increases the risk for HZ in patients with SLE; however, specific recommendations to prevent HZ are currently nonexistent for patients with SLE receiving anifrolumab.
- Researchers conducted a multicenter observational study in France from November 2021 to July 2024 to evaluate the prophylactic benefits of valaciclovir in 132 patients with SLE (mean age, 42 years; 92% women) treated with anifrolumab for ≥ 3 months.
- Among these patients, 87 received either 500 mg/d valaciclovir (n = 69) or 1000 mg/d valaciclovir (n = 18) as prophylaxis, whereas 45 did not receive valaciclovir.
- The patients were followed up for a median duration of 234 days under anifrolumab treatment, with monitoring for the development of herpes zoster.
TAKEAWAY:
- The risk for HZ was significantly lower in patients who received valaciclovir than in those who did not (hazard ratio, 0.08; P = .01).
- None of the patients treated with valaciclovir developed HZ during the survey period.
- The frequency of HZ in patients who did not receive valaciclovir increased progressively from 2.2% at 3 months to 6.2% at 6 months, reaching 23% at 12 months.
- None of the reported cases of HZ required hospitalization or led to anifrolumab discontinuation, although one patient developed neuralgia.
IN PRACTICE:
“Prophylactic treatment with valaciclovir is effective for preventing HZ [herpes zoster] infection in SLE patients treated with anifrolumab,” the authors wrote. “This finding is particularly relevant for SLE patients who cannot receive the recombinant HZ vaccine or for whom it is unavailable,” they added.
SOURCE:
The study was led by Ludovic Trefond, MD, PhD, Centre Hospitalier Universitaire de Clermont-Ferrand in France. It was published online on January 4, 2025, in RMD Open.
LIMITATIONS:
The observational design of the study and the low number of herpes zoster events during the follow-up period may have affected the robustness of the findings.
DISCLOSURES:
The authors did not receive any specific grants. Some authors reported having financial relationships with various pharmaceutical companies.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The use of valaciclovir as prophylaxis prevents herpes zoster (HZ) in patients with systemic lupus erythematosus (SLE) receiving anifrolumab treatment, with no cases of zoster reported during the follow-up period in patients receiving valaciclovir.
METHODOLOGY:
- Anifrolumab, a human monoclonal antibody binding to type I interferon receptor subunit 1, increases the risk for HZ in patients with SLE; however, specific recommendations to prevent HZ are currently nonexistent for patients with SLE receiving anifrolumab.
- Researchers conducted a multicenter observational study in France from November 2021 to July 2024 to evaluate the prophylactic benefits of valaciclovir in 132 patients with SLE (mean age, 42 years; 92% women) treated with anifrolumab for ≥ 3 months.
- Among these patients, 87 received either 500 mg/d valaciclovir (n = 69) or 1000 mg/d valaciclovir (n = 18) as prophylaxis, whereas 45 did not receive valaciclovir.
- The patients were followed up for a median duration of 234 days under anifrolumab treatment, with monitoring for the development of herpes zoster.
TAKEAWAY:
- The risk for HZ was significantly lower in patients who received valaciclovir than in those who did not (hazard ratio, 0.08; P = .01).
- None of the patients treated with valaciclovir developed HZ during the survey period.
- The frequency of HZ in patients who did not receive valaciclovir increased progressively from 2.2% at 3 months to 6.2% at 6 months, reaching 23% at 12 months.
- None of the reported cases of HZ required hospitalization or led to anifrolumab discontinuation, although one patient developed neuralgia.
IN PRACTICE:
“Prophylactic treatment with valaciclovir is effective for preventing HZ [herpes zoster] infection in SLE patients treated with anifrolumab,” the authors wrote. “This finding is particularly relevant for SLE patients who cannot receive the recombinant HZ vaccine or for whom it is unavailable,” they added.
SOURCE:
The study was led by Ludovic Trefond, MD, PhD, Centre Hospitalier Universitaire de Clermont-Ferrand in France. It was published online on January 4, 2025, in RMD Open.
LIMITATIONS:
The observational design of the study and the low number of herpes zoster events during the follow-up period may have affected the robustness of the findings.
DISCLOSURES:
The authors did not receive any specific grants. Some authors reported having financial relationships with various pharmaceutical companies.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Highly Contagious Norovirus Cases Spike This Season
Current data from the CDC’s NoroSTAT monitoring system show 495 reported outbreaks during the period from August 1, 2024, to December 11, 2024, compared with 363 outbreaks during the same period last year. In addition, the total number of norovirus outbreaks in the current season are higher than those reported in the seasonal years: 2012-2020 and 2021-2024.
Circulating strains of norovirus change over time, which can affect disease burden and potential disease severity, Sara Mirza, MD, an epidemiologist in the CDC’s Division of Viral Diseases, said in an interview.
The numbers for the 2024 norovirus season (considered approximately November to April) have reached or exceeded the case numbers seen before the COVID-19 pandemic, Mirza said.
The increase in cases may be caused in part by a new predominant strain of norovirus. “For the fall/winter of 2024-2025 season, genogroup 2, genotype 17, known as GII.17, has become the most detected genotype (strain) in the US among laboratory confirmed outbreaks reported to CDC,” said Mirza. “At this time, there is no indication that GII.17 causes more severe illness or affects one population more than another, but we are continuing to conduct surveillance to assess,” she added.
Clinical Takeaways
“Norovirus affects all ages, but young children and older adults are most at risk from more severe outcomes,” said Mirza.
“Clinicians treating older patients for acute gastroenteritis should be aware of these elevated risks and be sure to include norovirus as a potentially serious diagnosis, particularly in vulnerable patients with other diseases and those living in congregate settings, such as nursing homes,” she said.
When treating a patient with norovirus during an outbreak, use soap and water for hand hygiene after caring for patients with suspected or confirmed norovirus gastroenteritis, said Mirza. If norovirus infection is suspected, PPE use is recommended for individuals in the patient care area, she added.
Although state, local, and territorial health departments are not required to report individual cases of norovirus to the CDC, healthcare providers are encouraged to report all outbreaks of acute gastroenteritis, including suspected outbreaks of norovirus, to the appropriate state, local, or territorial health department, said Mirza. “Health departments are encouraged to report all suspected and confirmed norovirus outbreaks through the National Outbreak Reporting System and CaliciNet,” she added.
“Infection control measures, such as thorough hand washing, cleaning and disinfecting surfaces with bleach, and patient isolation and contact precautions in congregate or healthcare settings are the best ways to prevent norovirus and keep it from spreading to others,” Mirza said.
Remind patients that alcohol-based hand sanitizer is ineffective against norovirus, because the virus’s protective protein shell prevents the alcohol from penetrating and inactivating the virus, Mirza emphasized. “Soap and water work to remove germs from hands,” she said.
Cruise Ship Considerations
Cruise ships continue to be sources of increased risk for norovirus, according the CDC. The CDC’s Vessel Sanitation Program (VSP) was created to help the cruise industry prevent public health issues such as norovirus outbreaks, and to provide guidance for actions to take in the event of outbreaks.
For example, the most recently reported outbreak of norovirus on a cruise ship reported to the VSP was January 4, 2025, and occurred on a Holland America cruise from December 30, 2024, through January 8, 2025. Overall, 4.0% of passengers and 1.0% of crew members reported illness. Following VSP guidance, the ship reported increased cleaning and disinfection procedures and the collection of stool specimens for testing, and isolation of ill passengers and crew.
Clinical Perspective
In clinical practice, the number of norovirus cases is significantly exceeding previous years, and the trend seems to be consistent nationwide, David J. Cennimo, MD, associate professor of medicine and pediatrics at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
“Norovirus is incredibly contagious and spreads very quickly, which is how you get entire cruise ships infected at once,” he said. Norovirus is notoriously difficult to disinfect or kill, he added.
One possible contributor to the surge in cases is increased travel, especially during the holiday season, when people are coming together and sharing food, Cennimo noted. “We have seen many infections such as pneumonia return to levels approaching the period before the COVID-19 pandemic,” he said.
For norovirus prevention, strict attention to sanitation and handwashing is a must at home or when traveling, said Cennimo. For clinicians, it is important to report outbreaks of GI illness so appropriate control measures can be taken, he said.
Visit the CDC’s website on norovirus prevention for more information.
Mirza and Cennimo had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Current data from the CDC’s NoroSTAT monitoring system show 495 reported outbreaks during the period from August 1, 2024, to December 11, 2024, compared with 363 outbreaks during the same period last year. In addition, the total number of norovirus outbreaks in the current season are higher than those reported in the seasonal years: 2012-2020 and 2021-2024.
Circulating strains of norovirus change over time, which can affect disease burden and potential disease severity, Sara Mirza, MD, an epidemiologist in the CDC’s Division of Viral Diseases, said in an interview.
The numbers for the 2024 norovirus season (considered approximately November to April) have reached or exceeded the case numbers seen before the COVID-19 pandemic, Mirza said.
The increase in cases may be caused in part by a new predominant strain of norovirus. “For the fall/winter of 2024-2025 season, genogroup 2, genotype 17, known as GII.17, has become the most detected genotype (strain) in the US among laboratory confirmed outbreaks reported to CDC,” said Mirza. “At this time, there is no indication that GII.17 causes more severe illness or affects one population more than another, but we are continuing to conduct surveillance to assess,” she added.
Clinical Takeaways
“Norovirus affects all ages, but young children and older adults are most at risk from more severe outcomes,” said Mirza.
“Clinicians treating older patients for acute gastroenteritis should be aware of these elevated risks and be sure to include norovirus as a potentially serious diagnosis, particularly in vulnerable patients with other diseases and those living in congregate settings, such as nursing homes,” she said.
When treating a patient with norovirus during an outbreak, use soap and water for hand hygiene after caring for patients with suspected or confirmed norovirus gastroenteritis, said Mirza. If norovirus infection is suspected, PPE use is recommended for individuals in the patient care area, she added.
Although state, local, and territorial health departments are not required to report individual cases of norovirus to the CDC, healthcare providers are encouraged to report all outbreaks of acute gastroenteritis, including suspected outbreaks of norovirus, to the appropriate state, local, or territorial health department, said Mirza. “Health departments are encouraged to report all suspected and confirmed norovirus outbreaks through the National Outbreak Reporting System and CaliciNet,” she added.
“Infection control measures, such as thorough hand washing, cleaning and disinfecting surfaces with bleach, and patient isolation and contact precautions in congregate or healthcare settings are the best ways to prevent norovirus and keep it from spreading to others,” Mirza said.
Remind patients that alcohol-based hand sanitizer is ineffective against norovirus, because the virus’s protective protein shell prevents the alcohol from penetrating and inactivating the virus, Mirza emphasized. “Soap and water work to remove germs from hands,” she said.
Cruise Ship Considerations
Cruise ships continue to be sources of increased risk for norovirus, according the CDC. The CDC’s Vessel Sanitation Program (VSP) was created to help the cruise industry prevent public health issues such as norovirus outbreaks, and to provide guidance for actions to take in the event of outbreaks.
For example, the most recently reported outbreak of norovirus on a cruise ship reported to the VSP was January 4, 2025, and occurred on a Holland America cruise from December 30, 2024, through January 8, 2025. Overall, 4.0% of passengers and 1.0% of crew members reported illness. Following VSP guidance, the ship reported increased cleaning and disinfection procedures and the collection of stool specimens for testing, and isolation of ill passengers and crew.
Clinical Perspective
In clinical practice, the number of norovirus cases is significantly exceeding previous years, and the trend seems to be consistent nationwide, David J. Cennimo, MD, associate professor of medicine and pediatrics at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
“Norovirus is incredibly contagious and spreads very quickly, which is how you get entire cruise ships infected at once,” he said. Norovirus is notoriously difficult to disinfect or kill, he added.
One possible contributor to the surge in cases is increased travel, especially during the holiday season, when people are coming together and sharing food, Cennimo noted. “We have seen many infections such as pneumonia return to levels approaching the period before the COVID-19 pandemic,” he said.
For norovirus prevention, strict attention to sanitation and handwashing is a must at home or when traveling, said Cennimo. For clinicians, it is important to report outbreaks of GI illness so appropriate control measures can be taken, he said.
Visit the CDC’s website on norovirus prevention for more information.
Mirza and Cennimo had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Current data from the CDC’s NoroSTAT monitoring system show 495 reported outbreaks during the period from August 1, 2024, to December 11, 2024, compared with 363 outbreaks during the same period last year. In addition, the total number of norovirus outbreaks in the current season are higher than those reported in the seasonal years: 2012-2020 and 2021-2024.
Circulating strains of norovirus change over time, which can affect disease burden and potential disease severity, Sara Mirza, MD, an epidemiologist in the CDC’s Division of Viral Diseases, said in an interview.
The numbers for the 2024 norovirus season (considered approximately November to April) have reached or exceeded the case numbers seen before the COVID-19 pandemic, Mirza said.
The increase in cases may be caused in part by a new predominant strain of norovirus. “For the fall/winter of 2024-2025 season, genogroup 2, genotype 17, known as GII.17, has become the most detected genotype (strain) in the US among laboratory confirmed outbreaks reported to CDC,” said Mirza. “At this time, there is no indication that GII.17 causes more severe illness or affects one population more than another, but we are continuing to conduct surveillance to assess,” she added.
Clinical Takeaways
“Norovirus affects all ages, but young children and older adults are most at risk from more severe outcomes,” said Mirza.
“Clinicians treating older patients for acute gastroenteritis should be aware of these elevated risks and be sure to include norovirus as a potentially serious diagnosis, particularly in vulnerable patients with other diseases and those living in congregate settings, such as nursing homes,” she said.
When treating a patient with norovirus during an outbreak, use soap and water for hand hygiene after caring for patients with suspected or confirmed norovirus gastroenteritis, said Mirza. If norovirus infection is suspected, PPE use is recommended for individuals in the patient care area, she added.
Although state, local, and territorial health departments are not required to report individual cases of norovirus to the CDC, healthcare providers are encouraged to report all outbreaks of acute gastroenteritis, including suspected outbreaks of norovirus, to the appropriate state, local, or territorial health department, said Mirza. “Health departments are encouraged to report all suspected and confirmed norovirus outbreaks through the National Outbreak Reporting System and CaliciNet,” she added.
“Infection control measures, such as thorough hand washing, cleaning and disinfecting surfaces with bleach, and patient isolation and contact precautions in congregate or healthcare settings are the best ways to prevent norovirus and keep it from spreading to others,” Mirza said.
Remind patients that alcohol-based hand sanitizer is ineffective against norovirus, because the virus’s protective protein shell prevents the alcohol from penetrating and inactivating the virus, Mirza emphasized. “Soap and water work to remove germs from hands,” she said.
Cruise Ship Considerations
Cruise ships continue to be sources of increased risk for norovirus, according the CDC. The CDC’s Vessel Sanitation Program (VSP) was created to help the cruise industry prevent public health issues such as norovirus outbreaks, and to provide guidance for actions to take in the event of outbreaks.
For example, the most recently reported outbreak of norovirus on a cruise ship reported to the VSP was January 4, 2025, and occurred on a Holland America cruise from December 30, 2024, through January 8, 2025. Overall, 4.0% of passengers and 1.0% of crew members reported illness. Following VSP guidance, the ship reported increased cleaning and disinfection procedures and the collection of stool specimens for testing, and isolation of ill passengers and crew.
Clinical Perspective
In clinical practice, the number of norovirus cases is significantly exceeding previous years, and the trend seems to be consistent nationwide, David J. Cennimo, MD, associate professor of medicine and pediatrics at Rutgers New Jersey Medical School, Newark, New Jersey, said in an interview.
“Norovirus is incredibly contagious and spreads very quickly, which is how you get entire cruise ships infected at once,” he said. Norovirus is notoriously difficult to disinfect or kill, he added.
One possible contributor to the surge in cases is increased travel, especially during the holiday season, when people are coming together and sharing food, Cennimo noted. “We have seen many infections such as pneumonia return to levels approaching the period before the COVID-19 pandemic,” he said.
For norovirus prevention, strict attention to sanitation and handwashing is a must at home or when traveling, said Cennimo. For clinicians, it is important to report outbreaks of GI illness so appropriate control measures can be taken, he said.
Visit the CDC’s website on norovirus prevention for more information.
Mirza and Cennimo had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Do Antibiotics Before Conception Affect Fertility?
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Is there a connection between antibiotics taken before conception and adverse outcomes, such as reduced fertility, miscarriages, and congenital malformations?
A meta-analysis published in the journal eClinicalMedicine suggests a potential link between antibiotics taken before conception and negative outcomes, such as reduced fertility, miscarriages, and congenital malformations. However, a German expert in reproductive toxicology warned against drawing false conclusions.
“It would be fatal if women who want to have children refused necessary antibiotic treatment because they are afraid of infertility, miscarriages, and malformations,” said Wolfgang Paulus, MD, from the Reproductive Toxicology Advisory Center at the University Women’s Hospital in Ulm, Germany. In an interview, the expert criticized not only the authors’ conclusions but also the selection of studies included in the meta-analysis.
Confusion Over Use and Exposure
The meta-analysis, conducted by Bekalu Kassie Alemu, PhD, and colleagues from the Department of Obstetrics and Gynecology at The Chinese University of Hong Kong included 15 studies involving over 1.2 million women to examine how preconception antibiotic use affects fertility and pregnancy outcomes. In most studies (n = 11) that were included in the meta-analysis, fertility was examined as an endpoint, primarily in infertile women. One study involved Danish pharmacy employees who handled antibiotics at work.
“Not only was the therapeutic use of antibiotics not examined in this study, but the biological plausibility is completely lacking in this context,” Paulus noted.
The possible effects of preconception antibiotics on miscarriages were investigated in four studies, while two studies focused on congenital malformations as an endpoint.
Mixed Findings on Infertility
Regarding infertility, the authors reported abnormalities in macrolides and sulfonamides. Women who had received macrolide antibiotics, such as azithromycin, before conception showed a 35% reduction in fertility rates.
However, Paulus questioned whether this was solely because of macrolides. “Macrolide antibiotics are typically used for chlamydia, and chlamydia infection is a significant factor in women with unmet fertility desires,” he explained. Often, the chlamydia has already caused damage, such as inflammatory processes in the fallopian tubes, contributing to infertility that cannot be resolved by administering antibiotics.
The meta-analysis also showed that women who received sulfonamide before conception had a 2.35-fold increased likelihood of infertility. However, this association is not always one-sided. The results for tetracyclines were heterogeneous; while chlortetracycline appeared to increase the risk for infertility, exposure to oxytetracycline appeared to decrease it.
Treatment with oxytetracycline and beta-lactam antibiotics (except penicillin G) was associated with a 64% lower likelihood of infertility. The authors also found that fluoroquinolone antibiotics were associated with a 13% lower likelihood of infertility.
Miscarriage and Malformation Risks
Alemu and colleagues found a significant association between the use of antibiotics before conception and adverse pregnancy outcomes, showing a 34% increased risk for miscarriages and an 85% higher risk for congenital malformations with the use of trimethoprim during preconception. These findings highlight the need for caution regarding antibiotic use in women who are planning to conceive.
“Most antibiotics have half-lives of only a few hours. Therefore, antibiotics administered before conception can hardly have a direct effect on embryonic development,” Paulus noted. He pointed out that extensive data exist on most antibiotic classes included in this meta-analysis regarding childhood anomalies when used during the sensitive phase of organ development. These data do not indicate an increased risk for malformation. Therefore, the increased risk for malformations due to exposure before conception seems less plausible.
Alemu and colleagues assumed that antibiotics might negatively affect female reproductive health by disrupting the gut microbiome. The reasons for the reduced risk for infertility associated with beta-lactams and fluoroquinolones require further investigation. They reach a significant conclusion: “Preconception antibiotics exposure in females increases the risk of infertility, miscarriage, and congenital anomalies.” However, differences exist between the antibiotic classes. While the risk for infertility, spontaneous miscarriages, and congenital malformations increases with the use of macrolide antibiotics, sulfonamides, and trimethoprim, it decreases with the use of beta-lactams and fluoroquinolone antibiotics.
Expert Disagreement
“It is conceivable that the use of antibiotics damages the physiological environment, such as in the vaginal area. This may allow unwanted microbes to establish themselves, leading to more adverse outcomes such as infertility and miscarriages,” Paulus acknowledged.
Disruption of the microbiome due to antibiotic therapy could also result in a deficiency in relevant vitamins and trace elements (eg, folic acid), which could contribute to organogenesis disorders. Therefore, it may be beneficial to stabilize the gut and vaginal flora using probiotics after antibiotic treatment.
However, Paulus disagrees with the study conclusions. First, the studies included in the meta-analysis, which were largely observational, did not allow for the direct effect of antibiotics on the examined outcomes. Second, “quinolone antibiotics are highlighted as positive here, as if they were less problematic for patients trying to have children.”
Quinolone antibiotics are generally “frowned upon,” regardless of whether the patient wants to have children, as they can cause damage to the tendons, muscles, joints, and nervous system. They are currently used only as reserve medications.
“Quinolone antibiotics should not be administered during pregnancy, as they have already caused problems in animal studies, and they should not be used before pregnancy because of their side-effect profile,” Paulus stressed.
Serious Consequences
Paulus clarified:
In these cases, antibiotic treatment is appropriate, and there should be no fear of adverse effects on fertility or pregnancy outcomes. “If antibiotics are not given and the infection worsens, the patient will be even less likely to conceive successfully.”
This story was translated from Medscape’s German edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
FROM ECLINICALMEDICINE
Reducing Risk, One Mask at a Time: What the Science Says
A few items bring back unpleasant memories of COVID-19, such as masks. However, they are among the simplest and most effective ways to prevent the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). If everyone had worn them correctly, the transmission could have been reduced as much as ninefold, according to a theoretical study published in Physical Review E by Richard P. Sear, PhD, from the University of Surrey, Guildford, England.
Study Overcomes Limitations
This study aimed to address the limitations of epidemiological investigations of masks, which can be complex and error-prone. Sear used data obtained from the UK’s COVID-19 app, totaling 7 million contacts, to create a mathematical model of virus transmission, focusing on the correlation between contact duration and infection. The model estimates that if all UK residents had worn masks during every potential exposure, virus transmission would have been approximately nine times lower.
Although this is a mathematical model, it adds to the growing evidence that supports the benefits of masks. Masks are among the best strategies for treating SARS-CoV-2. This conclusion has been supported by several systematic reviews and additional statistical studies. Conversely, the decision to relax and eliminate mask regulations has had consequences that have received little attention.
As expected, removing the mask mandate leads to increased virus transmission, resulting in more hospitalizations and deaths. A 2024 study estimated that in Japan, where cultural factors lead to much higher mask use in public than in Europe, the decline in mask use from 97% of the population in 2022 to 63% in October 2023 may have caused an additional 3500 deaths.
Impact Beyond SARS-CoV-2
One remarkable effect of non-pharmaceutical interventions during the pandemic was the probable extinction of an entire influenza strain (B/Yamagata), which could improve future influenza vaccines and significantly reduce the spread of respiratory syncytial virus. While this was not solely caused by masks, it was also influenced by emergency measures such as lockdowns and social distancing. These behavioral changes can positively alter the landscape of infectious diseases.
Masks play a role in reducing influenza transmission during pandemics. Their effectiveness has been supported by several studies and systematic reviews on a wide range of respiratory viruses. A randomized clinical trial involving 4647 Norwegian participants from February to April 2023, published in May 2024 by the British Medical Journal, suggested that wearing a mask reduces the incidence of respiratory symptoms. Specifically, 8.9% of those who wore masks reported respiratory symptoms during the study period compared with 12.2% of those who did not, representing a relative risk reduction of 27%.
Widespread mask use could also protect against other factors such as fine particulate matter, indirectly reducing the risk for various health conditions. A retrospective study involving 7.8 million residents in the Chinese city of Weifang, published in December 2024 by BMC Public Health, suggested that mask use during the pandemic may have also protected the population from pollution, reducing the number of stroke cases by 38.6% over 33 months of follow-up.
Although there are still voices in bioethics calling for the reintroduction of mask mandates in public places, it is unlikely that, barring emergencies, mask mandates are politically and socially acceptable today. Mask use is also considered a politically polarizing topic in several Western countries. Nevertheless, it is worth considering whether, as we move away from the acute phase of the COVID-19 pandemic, we can more objectively promote the use of masks in public places.
Communicating the importance of public health initiatives and persuading people to support them is a well-known challenge. However, scientific literature offers valuable insights. These include encouraging people to rely on rational thinking rather than emotions and providing information on how masks protect those around them. The fact that East Asian cultures tend to have a more positive relationship with the use of masks shows that, in principle, it is possible to make them acceptable. Data from studies suggest that, as we prepare for potential future pandemics, it may be time to move past polarization and reintroduce masks — not as a universal mandate but as an individual choice for many.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
A few items bring back unpleasant memories of COVID-19, such as masks. However, they are among the simplest and most effective ways to prevent the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). If everyone had worn them correctly, the transmission could have been reduced as much as ninefold, according to a theoretical study published in Physical Review E by Richard P. Sear, PhD, from the University of Surrey, Guildford, England.
Study Overcomes Limitations
This study aimed to address the limitations of epidemiological investigations of masks, which can be complex and error-prone. Sear used data obtained from the UK’s COVID-19 app, totaling 7 million contacts, to create a mathematical model of virus transmission, focusing on the correlation between contact duration and infection. The model estimates that if all UK residents had worn masks during every potential exposure, virus transmission would have been approximately nine times lower.
Although this is a mathematical model, it adds to the growing evidence that supports the benefits of masks. Masks are among the best strategies for treating SARS-CoV-2. This conclusion has been supported by several systematic reviews and additional statistical studies. Conversely, the decision to relax and eliminate mask regulations has had consequences that have received little attention.
As expected, removing the mask mandate leads to increased virus transmission, resulting in more hospitalizations and deaths. A 2024 study estimated that in Japan, where cultural factors lead to much higher mask use in public than in Europe, the decline in mask use from 97% of the population in 2022 to 63% in October 2023 may have caused an additional 3500 deaths.
Impact Beyond SARS-CoV-2
One remarkable effect of non-pharmaceutical interventions during the pandemic was the probable extinction of an entire influenza strain (B/Yamagata), which could improve future influenza vaccines and significantly reduce the spread of respiratory syncytial virus. While this was not solely caused by masks, it was also influenced by emergency measures such as lockdowns and social distancing. These behavioral changes can positively alter the landscape of infectious diseases.
Masks play a role in reducing influenza transmission during pandemics. Their effectiveness has been supported by several studies and systematic reviews on a wide range of respiratory viruses. A randomized clinical trial involving 4647 Norwegian participants from February to April 2023, published in May 2024 by the British Medical Journal, suggested that wearing a mask reduces the incidence of respiratory symptoms. Specifically, 8.9% of those who wore masks reported respiratory symptoms during the study period compared with 12.2% of those who did not, representing a relative risk reduction of 27%.
Widespread mask use could also protect against other factors such as fine particulate matter, indirectly reducing the risk for various health conditions. A retrospective study involving 7.8 million residents in the Chinese city of Weifang, published in December 2024 by BMC Public Health, suggested that mask use during the pandemic may have also protected the population from pollution, reducing the number of stroke cases by 38.6% over 33 months of follow-up.
Although there are still voices in bioethics calling for the reintroduction of mask mandates in public places, it is unlikely that, barring emergencies, mask mandates are politically and socially acceptable today. Mask use is also considered a politically polarizing topic in several Western countries. Nevertheless, it is worth considering whether, as we move away from the acute phase of the COVID-19 pandemic, we can more objectively promote the use of masks in public places.
Communicating the importance of public health initiatives and persuading people to support them is a well-known challenge. However, scientific literature offers valuable insights. These include encouraging people to rely on rational thinking rather than emotions and providing information on how masks protect those around them. The fact that East Asian cultures tend to have a more positive relationship with the use of masks shows that, in principle, it is possible to make them acceptable. Data from studies suggest that, as we prepare for potential future pandemics, it may be time to move past polarization and reintroduce masks — not as a universal mandate but as an individual choice for many.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
A few items bring back unpleasant memories of COVID-19, such as masks. However, they are among the simplest and most effective ways to prevent the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). If everyone had worn them correctly, the transmission could have been reduced as much as ninefold, according to a theoretical study published in Physical Review E by Richard P. Sear, PhD, from the University of Surrey, Guildford, England.
Study Overcomes Limitations
This study aimed to address the limitations of epidemiological investigations of masks, which can be complex and error-prone. Sear used data obtained from the UK’s COVID-19 app, totaling 7 million contacts, to create a mathematical model of virus transmission, focusing on the correlation between contact duration and infection. The model estimates that if all UK residents had worn masks during every potential exposure, virus transmission would have been approximately nine times lower.
Although this is a mathematical model, it adds to the growing evidence that supports the benefits of masks. Masks are among the best strategies for treating SARS-CoV-2. This conclusion has been supported by several systematic reviews and additional statistical studies. Conversely, the decision to relax and eliminate mask regulations has had consequences that have received little attention.
As expected, removing the mask mandate leads to increased virus transmission, resulting in more hospitalizations and deaths. A 2024 study estimated that in Japan, where cultural factors lead to much higher mask use in public than in Europe, the decline in mask use from 97% of the population in 2022 to 63% in October 2023 may have caused an additional 3500 deaths.
Impact Beyond SARS-CoV-2
One remarkable effect of non-pharmaceutical interventions during the pandemic was the probable extinction of an entire influenza strain (B/Yamagata), which could improve future influenza vaccines and significantly reduce the spread of respiratory syncytial virus. While this was not solely caused by masks, it was also influenced by emergency measures such as lockdowns and social distancing. These behavioral changes can positively alter the landscape of infectious diseases.
Masks play a role in reducing influenza transmission during pandemics. Their effectiveness has been supported by several studies and systematic reviews on a wide range of respiratory viruses. A randomized clinical trial involving 4647 Norwegian participants from February to April 2023, published in May 2024 by the British Medical Journal, suggested that wearing a mask reduces the incidence of respiratory symptoms. Specifically, 8.9% of those who wore masks reported respiratory symptoms during the study period compared with 12.2% of those who did not, representing a relative risk reduction of 27%.
Widespread mask use could also protect against other factors such as fine particulate matter, indirectly reducing the risk for various health conditions. A retrospective study involving 7.8 million residents in the Chinese city of Weifang, published in December 2024 by BMC Public Health, suggested that mask use during the pandemic may have also protected the population from pollution, reducing the number of stroke cases by 38.6% over 33 months of follow-up.
Although there are still voices in bioethics calling for the reintroduction of mask mandates in public places, it is unlikely that, barring emergencies, mask mandates are politically and socially acceptable today. Mask use is also considered a politically polarizing topic in several Western countries. Nevertheless, it is worth considering whether, as we move away from the acute phase of the COVID-19 pandemic, we can more objectively promote the use of masks in public places.
Communicating the importance of public health initiatives and persuading people to support them is a well-known challenge. However, scientific literature offers valuable insights. These include encouraging people to rely on rational thinking rather than emotions and providing information on how masks protect those around them. The fact that East Asian cultures tend to have a more positive relationship with the use of masks shows that, in principle, it is possible to make them acceptable. Data from studies suggest that, as we prepare for potential future pandemics, it may be time to move past polarization and reintroduce masks — not as a universal mandate but as an individual choice for many.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.