User login
Tularemia: A Rare But Nationally Notifiable Disease
The pediatrician’s first patient of the day was an 8-year-old boy, accompanied by both of his parents. It was the boy’s third visit in just over a week for fever and left-sided neck swelling, and the family was understandably anxious for answers.
“The antibiotics don’t seem to be working,” the mother explained. “He still has fever every day, as high as 104, and his neck looks just as swollen.”
A quick review of the chart revealed the boy’s initial diagnosis had been bacterial lymphadenitis, for which amoxicillin-clavulanate had been prescribed. Three days later, given lack of clinical improvement, therapy was transitioned to clindamycin. On examination, the boy was febrile and ill-appearing with a 3-cm by 5-cm tender, non-fluctuant swelling over the left sternocleidomastoid muscle.
The pediatrician ran through a quick mental checklist of diagnostic possibilities for his patient’s continued symptoms. Staphylococcal lymphadenitis still seemed possible. Could the boy be infected with methicillin-resistant Staphylococcus aureus that was also clindamycin resistant? Alternately, perhaps the problem was “source control” and the boy had developed an occult neck abscess that needed to be drained. An ultrasound could help sort that out. Finally, the pediatrician considered less common bacterial causes of lymph node swelling and fever. He placed Bartonella henselae, the cause of cat scratch disease, near the top of his list. “I’ve never seen it,” he told the parents, “But we could also consider tularemia.”
On average, 200 cases of tularemia are reported in the United States each year, and the incidence of disease is increasing, according to a surveillance report released by the Centers for Disease Control and Prevention in December 2023.1
Between 2011 and 2022, 2462 tularemia cases were reported in the United States. That translated to an average annual incidence of 0.064 per 100,000 population, an increase of 56% compared with 2001-2010. Forty-seven states reported at least one case of tularemia, although half of all reported cases came from four states — Arkansas (18%), Kansas (11%), Missouri (11%), and Oklahoma (10%). The incidence of tularemia was highest in children ages 5-9 years old, older men, and American Indian or Alaska Natives individuals. Although cases occurred year-round, 78% had symptom onset May through September.
In the United States, most human cases of tularemia have been arthropod borne, transmitted by the bite of an infected tick or deer fly. Infection also can be spread through contact with infected animals or animal tissue, particularly rabbits, hares, muskrats, prairie dogs, and other rodents, including hamsters. Outbreaks of tularemia have occurred among pet store hamsters, and at least one child in the United States developed tularemia after being bitten by a pet hamster.
Tularemia is almost always associated with fever but other clinical manifestations vary by the type of exposure. Ulceroglandular disease occurs after a tick or deer fly bite or after handling an infected animal. An ulcer develops at the site where the bacteria entered the body, along with enlargement of regional lymph nodes. Less commonly, lymph node swelling can occur without the development of an ulcer. If the bacteria enter through the eye, symptoms include conjunctivitis and swelling of pre-auricular lymph nodes. Eating or drinking contaminated food or water is associated with sore throat, mouth ulcers, tonsillitis, and swelling of lymph glands in the neck. Pneumonic tularemia, the most serious form of the disease, typically happens after inhaling bacteria-containing dust or aerosols and is associated with cough, chest pain, and difficulty breathing. Pneumonic tularemia can develop if other forms of tularemia are untreated, and the bacteria spread to the lung.
Back in the exam room, the pediatrician carefully re-examined the boy’s scalp. A 1-cm poorly healing ulcer on the left occiput added support for the diagnosis of ulceroglandular tularemia, the most common form of the disease in children. Serologic testing ultimately confirmed the diagnosis and the boy’s symptoms resolved with treatment.
Gentamicin administered intravenously or intramuscularly is the drug of choice for the treatment of tularemia in children. Ciprofloxacin is considered an alternative but is not approved by the U.S. Food and Drug Administration for this indication.
The pediatrician reported the case of tularemia to his local health department. Tularemia is a nationally notifiable disease in the United States; state health departments report to the CDC through the National Notifiable Diseases Surveillance System. In turn, public health authorities shared information to prevent tularemia. Steps to prevent tick and deer fly bites include the use of an Environmental Protection Agency–registered insect repellent. Individuals who hunt, trap, or skin animals are encouraged to wear gloves when handling animals —especially rabbits, muskrats, and prairie dogs — and cook game meat thoroughly. Tularemia can be inadvertently aerosolized if an infected animal or carcass is run over with a tractor or lawnmower. Checking for carcasses before mowing may reduce the risk.
Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at pdnews@mdedge.com. (Also kristina.bryant@louisville.edu.)
Reference
1. Rich SN et al. Tularemia—United States, 2011-2022. MMWR Morb Mortal Wkly Rep 2025;73:1152–1156. doi:
The pediatrician’s first patient of the day was an 8-year-old boy, accompanied by both of his parents. It was the boy’s third visit in just over a week for fever and left-sided neck swelling, and the family was understandably anxious for answers.
“The antibiotics don’t seem to be working,” the mother explained. “He still has fever every day, as high as 104, and his neck looks just as swollen.”
A quick review of the chart revealed the boy’s initial diagnosis had been bacterial lymphadenitis, for which amoxicillin-clavulanate had been prescribed. Three days later, given lack of clinical improvement, therapy was transitioned to clindamycin. On examination, the boy was febrile and ill-appearing with a 3-cm by 5-cm tender, non-fluctuant swelling over the left sternocleidomastoid muscle.
The pediatrician ran through a quick mental checklist of diagnostic possibilities for his patient’s continued symptoms. Staphylococcal lymphadenitis still seemed possible. Could the boy be infected with methicillin-resistant Staphylococcus aureus that was also clindamycin resistant? Alternately, perhaps the problem was “source control” and the boy had developed an occult neck abscess that needed to be drained. An ultrasound could help sort that out. Finally, the pediatrician considered less common bacterial causes of lymph node swelling and fever. He placed Bartonella henselae, the cause of cat scratch disease, near the top of his list. “I’ve never seen it,” he told the parents, “But we could also consider tularemia.”
On average, 200 cases of tularemia are reported in the United States each year, and the incidence of disease is increasing, according to a surveillance report released by the Centers for Disease Control and Prevention in December 2023.1
Between 2011 and 2022, 2462 tularemia cases were reported in the United States. That translated to an average annual incidence of 0.064 per 100,000 population, an increase of 56% compared with 2001-2010. Forty-seven states reported at least one case of tularemia, although half of all reported cases came from four states — Arkansas (18%), Kansas (11%), Missouri (11%), and Oklahoma (10%). The incidence of tularemia was highest in children ages 5-9 years old, older men, and American Indian or Alaska Natives individuals. Although cases occurred year-round, 78% had symptom onset May through September.
In the United States, most human cases of tularemia have been arthropod borne, transmitted by the bite of an infected tick or deer fly. Infection also can be spread through contact with infected animals or animal tissue, particularly rabbits, hares, muskrats, prairie dogs, and other rodents, including hamsters. Outbreaks of tularemia have occurred among pet store hamsters, and at least one child in the United States developed tularemia after being bitten by a pet hamster.
Tularemia is almost always associated with fever but other clinical manifestations vary by the type of exposure. Ulceroglandular disease occurs after a tick or deer fly bite or after handling an infected animal. An ulcer develops at the site where the bacteria entered the body, along with enlargement of regional lymph nodes. Less commonly, lymph node swelling can occur without the development of an ulcer. If the bacteria enter through the eye, symptoms include conjunctivitis and swelling of pre-auricular lymph nodes. Eating or drinking contaminated food or water is associated with sore throat, mouth ulcers, tonsillitis, and swelling of lymph glands in the neck. Pneumonic tularemia, the most serious form of the disease, typically happens after inhaling bacteria-containing dust or aerosols and is associated with cough, chest pain, and difficulty breathing. Pneumonic tularemia can develop if other forms of tularemia are untreated, and the bacteria spread to the lung.
Back in the exam room, the pediatrician carefully re-examined the boy’s scalp. A 1-cm poorly healing ulcer on the left occiput added support for the diagnosis of ulceroglandular tularemia, the most common form of the disease in children. Serologic testing ultimately confirmed the diagnosis and the boy’s symptoms resolved with treatment.
Gentamicin administered intravenously or intramuscularly is the drug of choice for the treatment of tularemia in children. Ciprofloxacin is considered an alternative but is not approved by the U.S. Food and Drug Administration for this indication.
The pediatrician reported the case of tularemia to his local health department. Tularemia is a nationally notifiable disease in the United States; state health departments report to the CDC through the National Notifiable Diseases Surveillance System. In turn, public health authorities shared information to prevent tularemia. Steps to prevent tick and deer fly bites include the use of an Environmental Protection Agency–registered insect repellent. Individuals who hunt, trap, or skin animals are encouraged to wear gloves when handling animals —especially rabbits, muskrats, and prairie dogs — and cook game meat thoroughly. Tularemia can be inadvertently aerosolized if an infected animal or carcass is run over with a tractor or lawnmower. Checking for carcasses before mowing may reduce the risk.
Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at pdnews@mdedge.com. (Also kristina.bryant@louisville.edu.)
Reference
1. Rich SN et al. Tularemia—United States, 2011-2022. MMWR Morb Mortal Wkly Rep 2025;73:1152–1156. doi:
The pediatrician’s first patient of the day was an 8-year-old boy, accompanied by both of his parents. It was the boy’s third visit in just over a week for fever and left-sided neck swelling, and the family was understandably anxious for answers.
“The antibiotics don’t seem to be working,” the mother explained. “He still has fever every day, as high as 104, and his neck looks just as swollen.”
A quick review of the chart revealed the boy’s initial diagnosis had been bacterial lymphadenitis, for which amoxicillin-clavulanate had been prescribed. Three days later, given lack of clinical improvement, therapy was transitioned to clindamycin. On examination, the boy was febrile and ill-appearing with a 3-cm by 5-cm tender, non-fluctuant swelling over the left sternocleidomastoid muscle.
The pediatrician ran through a quick mental checklist of diagnostic possibilities for his patient’s continued symptoms. Staphylococcal lymphadenitis still seemed possible. Could the boy be infected with methicillin-resistant Staphylococcus aureus that was also clindamycin resistant? Alternately, perhaps the problem was “source control” and the boy had developed an occult neck abscess that needed to be drained. An ultrasound could help sort that out. Finally, the pediatrician considered less common bacterial causes of lymph node swelling and fever. He placed Bartonella henselae, the cause of cat scratch disease, near the top of his list. “I’ve never seen it,” he told the parents, “But we could also consider tularemia.”
On average, 200 cases of tularemia are reported in the United States each year, and the incidence of disease is increasing, according to a surveillance report released by the Centers for Disease Control and Prevention in December 2023.1
Between 2011 and 2022, 2462 tularemia cases were reported in the United States. That translated to an average annual incidence of 0.064 per 100,000 population, an increase of 56% compared with 2001-2010. Forty-seven states reported at least one case of tularemia, although half of all reported cases came from four states — Arkansas (18%), Kansas (11%), Missouri (11%), and Oklahoma (10%). The incidence of tularemia was highest in children ages 5-9 years old, older men, and American Indian or Alaska Natives individuals. Although cases occurred year-round, 78% had symptom onset May through September.
In the United States, most human cases of tularemia have been arthropod borne, transmitted by the bite of an infected tick or deer fly. Infection also can be spread through contact with infected animals or animal tissue, particularly rabbits, hares, muskrats, prairie dogs, and other rodents, including hamsters. Outbreaks of tularemia have occurred among pet store hamsters, and at least one child in the United States developed tularemia after being bitten by a pet hamster.
Tularemia is almost always associated with fever but other clinical manifestations vary by the type of exposure. Ulceroglandular disease occurs after a tick or deer fly bite or after handling an infected animal. An ulcer develops at the site where the bacteria entered the body, along with enlargement of regional lymph nodes. Less commonly, lymph node swelling can occur without the development of an ulcer. If the bacteria enter through the eye, symptoms include conjunctivitis and swelling of pre-auricular lymph nodes. Eating or drinking contaminated food or water is associated with sore throat, mouth ulcers, tonsillitis, and swelling of lymph glands in the neck. Pneumonic tularemia, the most serious form of the disease, typically happens after inhaling bacteria-containing dust or aerosols and is associated with cough, chest pain, and difficulty breathing. Pneumonic tularemia can develop if other forms of tularemia are untreated, and the bacteria spread to the lung.
Back in the exam room, the pediatrician carefully re-examined the boy’s scalp. A 1-cm poorly healing ulcer on the left occiput added support for the diagnosis of ulceroglandular tularemia, the most common form of the disease in children. Serologic testing ultimately confirmed the diagnosis and the boy’s symptoms resolved with treatment.
Gentamicin administered intravenously or intramuscularly is the drug of choice for the treatment of tularemia in children. Ciprofloxacin is considered an alternative but is not approved by the U.S. Food and Drug Administration for this indication.
The pediatrician reported the case of tularemia to his local health department. Tularemia is a nationally notifiable disease in the United States; state health departments report to the CDC through the National Notifiable Diseases Surveillance System. In turn, public health authorities shared information to prevent tularemia. Steps to prevent tick and deer fly bites include the use of an Environmental Protection Agency–registered insect repellent. Individuals who hunt, trap, or skin animals are encouraged to wear gloves when handling animals —especially rabbits, muskrats, and prairie dogs — and cook game meat thoroughly. Tularemia can be inadvertently aerosolized if an infected animal or carcass is run over with a tractor or lawnmower. Checking for carcasses before mowing may reduce the risk.
Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at pdnews@mdedge.com. (Also kristina.bryant@louisville.edu.)
Reference
1. Rich SN et al. Tularemia—United States, 2011-2022. MMWR Morb Mortal Wkly Rep 2025;73:1152–1156. doi:
Traumatic Brain Injury May Reactivate Herpes Virus Leading to Neurodegeneration
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
FROM SCIENCE SIGNALING
RSV Vaccines and Treatments Face Global Access Hurdles
Almost 70 years after the discovery of the respiratory syncytial virus (RSV), vaccines and preventive treatments are giving babies a chance to beat the potentially deadly childhood infection.
As doctors turn to monoclonal antibody therapies and governments plan vaccination programs, clinical researchers are asking whether these measures will reduce the spread of the virus. Will fewer babies die from RSV, and fewer children develop permanent wheezing?
Recent studies offer clues.
Fabio Midulla, an associate professor of pediatrics at Sapienza University of Rome in Rome, Italy, said that the pharmaceutical industry is poised to push governments to use vaccines and monoclonal antibodies for even more children. “Such a push might work,” he said at the European Respiratory Society (ERS) 2024 Congress, “given that several studies have already demonstrated that their use can improve outcomes for children who do become infected and reduce societal costs by reducing hospitalizations.”
But Mariëlle WH Pijnenburg, a pulmonary specialist at Erasmus University Rotterdam in the Netherlands, said at the Congress that greater rollout would require governments to force industry to lower prices. If treatments remain beyond the reach of lower-income countries — where the burden of RSV is the greatest — the death toll from this common childhood infection will remain stubbornly high, and the prospect of global elimination will remain forever out of reach, she said.
New Tools in the Fight Against RSV
Nirsevimab, a long-acting monoclonal antibody given to newborns to prevent severe infection, was approved by the European Medicines Agency (EMA) in October 2022 and the US Food and Drug Administration (FDA) in July 2023. And Abrysvo, a vaccine given to older adults and pregnant women to stop them from passing the virus to babies from birth through 6 months of age, was approved by the FDA and the EMA in 2023.
RSV is responsible for over 33 million lung infections in children younger than 5 years annually, with more than 4 million hospitalizations and nearly 200,000 deaths. According to the Centers for Disease Control and Prevention, every year, 2.1 million children younger than 5 years old visit a healthcare provider because of an RSV infection and between 58,000 and 80,000 children younger than 5 years old are hospitalized in the United States. The burden of severe RSV disease is also high among adults, with an estimated 123,000-193,000 hospitalizations, 24,400-34,900 ICU admissions, and 4680-8620 in-hospital deaths occurring annually among US adults.
Infection in infancy can lead to later complications, such as the development of wheezing, a condition that causes breathlessness and a feeling of tightening in the chest, and possibly also asthma.
Studies have shown that children and preterm infants infected with RSV who were given monoclonal antibodies experienced less post-infection wheezing, suggesting that RSV prophylaxis could prevent the development of wheezing bronchitis.
A study conducted in Galicia, Spain, showed that only 0.3% of infants who received prophylaxis with Nirsevimab were hospitalized for RSV-related lower respiratory tract infections. “This is very promising,” Yvonne Maldonado, MD, professor of pediatrics and epidemiology and population health at Stanford University in Stanford, California, told Medscape Medical News. “But this virus is ubiquitous. It’s found everywhere. It comes around every winter season. And immunity is not long-lasting.”
Older children who are not receiving monoclonal antibodies still experience RSV-related hospitalizations, suggesting the virus continues to circulate at high enough levels in the community. “The vaccine and monoclonal antibodies can reduce the risk of hospitalization and more severe disease in young kids, but they won’t eliminate the virus,” Maldonado said. “Right now, the goal is to prevent serious infection, not to prevent the spread of the virus completely.”
Expanding Access to RSV Prevention in Low-Income Countries
Currently, the RSV vaccine and monoclonal antibodies are only given in the United States, Europe, United Kingdom, and Canada to newborns, children at risk for severe disease, and pregnant women. However, Midulla said that pharmaceutical companies are pushing to broaden the rollout to a broader population within these countries. Yet, he said, over 99% of RSV infection–related deaths occur in the Global South.
No pharmaceutical company has sought approval in low-income countries such as those in Africa. “Unless they see there being a market in a country, they’re not going to go through the onerous process of getting [a vaccine] licensed,” Shabir Madhi, dean of the faculty of health sciences and a professor of vaccinology at the University of the Witwatersrand, Johannesburg, South Africa, told Medscape Medical News.
He highlighted that almost 50% of RSV-related deaths occur in African children younger than 5 years, despite these children comprising just one fifth of the global under-5 population. The high burden of RSV mortality in the Global South is mainly due to poor access to healthcare and supportive treatments, such as supplemental oxygen, which can help children recover from severe RSV infection.
Companies are unlikely to pursue regulatory approval and licensing in low- and middle-income countries until GAVI, the global vaccine alliance, commits to procuring and funding the vaccines for these regions. GAVI’s decision would provide the necessary market incentive for manufacturers to seek approval.
Madhi suggested that GAVI’s decision on RSV vaccine procurement is imminent, likely early next year, following the World Health Organization’s Strategic Advisory Group of Experts on Immunization recommendation to vaccinate all pregnant women with the RSV vaccine, regardless of whether they are in high-income or low-income countries.
Nevertheless, even if vaccines become available, many African countries may still struggle to afford them. Madhi said that these countries would likely depend on GAVI and organizations like UNICEF to procure the vaccines at affordable prices. “The unfortunate reality is that many countries — especially in Africa — still wouldn’t be able to afford it, even if the vaccine cost as little as $5,” said Madhi. “But that’s where they would have the greatest impact.”
Midulla, Pijnenburg reported no relevant financial relationships. Madhi’s research unit, the Vaccines and Infectious Disease Analytics Unit, was involved in the clinical trials for the Pfizer RSV vaccine, the GSK RSV vaccine (which was terminated), as well as the MEDLEY trial of palivizumab. All funding for these studies went to his institution, the University of the Witwatersrand. Maldonado was Stanford principal investigator for the Pfizer RSV vaccine.
A version of this article appeared on Medscape.com.
Almost 70 years after the discovery of the respiratory syncytial virus (RSV), vaccines and preventive treatments are giving babies a chance to beat the potentially deadly childhood infection.
As doctors turn to monoclonal antibody therapies and governments plan vaccination programs, clinical researchers are asking whether these measures will reduce the spread of the virus. Will fewer babies die from RSV, and fewer children develop permanent wheezing?
Recent studies offer clues.
Fabio Midulla, an associate professor of pediatrics at Sapienza University of Rome in Rome, Italy, said that the pharmaceutical industry is poised to push governments to use vaccines and monoclonal antibodies for even more children. “Such a push might work,” he said at the European Respiratory Society (ERS) 2024 Congress, “given that several studies have already demonstrated that their use can improve outcomes for children who do become infected and reduce societal costs by reducing hospitalizations.”
But Mariëlle WH Pijnenburg, a pulmonary specialist at Erasmus University Rotterdam in the Netherlands, said at the Congress that greater rollout would require governments to force industry to lower prices. If treatments remain beyond the reach of lower-income countries — where the burden of RSV is the greatest — the death toll from this common childhood infection will remain stubbornly high, and the prospect of global elimination will remain forever out of reach, she said.
New Tools in the Fight Against RSV
Nirsevimab, a long-acting monoclonal antibody given to newborns to prevent severe infection, was approved by the European Medicines Agency (EMA) in October 2022 and the US Food and Drug Administration (FDA) in July 2023. And Abrysvo, a vaccine given to older adults and pregnant women to stop them from passing the virus to babies from birth through 6 months of age, was approved by the FDA and the EMA in 2023.
RSV is responsible for over 33 million lung infections in children younger than 5 years annually, with more than 4 million hospitalizations and nearly 200,000 deaths. According to the Centers for Disease Control and Prevention, every year, 2.1 million children younger than 5 years old visit a healthcare provider because of an RSV infection and between 58,000 and 80,000 children younger than 5 years old are hospitalized in the United States. The burden of severe RSV disease is also high among adults, with an estimated 123,000-193,000 hospitalizations, 24,400-34,900 ICU admissions, and 4680-8620 in-hospital deaths occurring annually among US adults.
Infection in infancy can lead to later complications, such as the development of wheezing, a condition that causes breathlessness and a feeling of tightening in the chest, and possibly also asthma.
Studies have shown that children and preterm infants infected with RSV who were given monoclonal antibodies experienced less post-infection wheezing, suggesting that RSV prophylaxis could prevent the development of wheezing bronchitis.
A study conducted in Galicia, Spain, showed that only 0.3% of infants who received prophylaxis with Nirsevimab were hospitalized for RSV-related lower respiratory tract infections. “This is very promising,” Yvonne Maldonado, MD, professor of pediatrics and epidemiology and population health at Stanford University in Stanford, California, told Medscape Medical News. “But this virus is ubiquitous. It’s found everywhere. It comes around every winter season. And immunity is not long-lasting.”
Older children who are not receiving monoclonal antibodies still experience RSV-related hospitalizations, suggesting the virus continues to circulate at high enough levels in the community. “The vaccine and monoclonal antibodies can reduce the risk of hospitalization and more severe disease in young kids, but they won’t eliminate the virus,” Maldonado said. “Right now, the goal is to prevent serious infection, not to prevent the spread of the virus completely.”
Expanding Access to RSV Prevention in Low-Income Countries
Currently, the RSV vaccine and monoclonal antibodies are only given in the United States, Europe, United Kingdom, and Canada to newborns, children at risk for severe disease, and pregnant women. However, Midulla said that pharmaceutical companies are pushing to broaden the rollout to a broader population within these countries. Yet, he said, over 99% of RSV infection–related deaths occur in the Global South.
No pharmaceutical company has sought approval in low-income countries such as those in Africa. “Unless they see there being a market in a country, they’re not going to go through the onerous process of getting [a vaccine] licensed,” Shabir Madhi, dean of the faculty of health sciences and a professor of vaccinology at the University of the Witwatersrand, Johannesburg, South Africa, told Medscape Medical News.
He highlighted that almost 50% of RSV-related deaths occur in African children younger than 5 years, despite these children comprising just one fifth of the global under-5 population. The high burden of RSV mortality in the Global South is mainly due to poor access to healthcare and supportive treatments, such as supplemental oxygen, which can help children recover from severe RSV infection.
Companies are unlikely to pursue regulatory approval and licensing in low- and middle-income countries until GAVI, the global vaccine alliance, commits to procuring and funding the vaccines for these regions. GAVI’s decision would provide the necessary market incentive for manufacturers to seek approval.
Madhi suggested that GAVI’s decision on RSV vaccine procurement is imminent, likely early next year, following the World Health Organization’s Strategic Advisory Group of Experts on Immunization recommendation to vaccinate all pregnant women with the RSV vaccine, regardless of whether they are in high-income or low-income countries.
Nevertheless, even if vaccines become available, many African countries may still struggle to afford them. Madhi said that these countries would likely depend on GAVI and organizations like UNICEF to procure the vaccines at affordable prices. “The unfortunate reality is that many countries — especially in Africa — still wouldn’t be able to afford it, even if the vaccine cost as little as $5,” said Madhi. “But that’s where they would have the greatest impact.”
Midulla, Pijnenburg reported no relevant financial relationships. Madhi’s research unit, the Vaccines and Infectious Disease Analytics Unit, was involved in the clinical trials for the Pfizer RSV vaccine, the GSK RSV vaccine (which was terminated), as well as the MEDLEY trial of palivizumab. All funding for these studies went to his institution, the University of the Witwatersrand. Maldonado was Stanford principal investigator for the Pfizer RSV vaccine.
A version of this article appeared on Medscape.com.
Almost 70 years after the discovery of the respiratory syncytial virus (RSV), vaccines and preventive treatments are giving babies a chance to beat the potentially deadly childhood infection.
As doctors turn to monoclonal antibody therapies and governments plan vaccination programs, clinical researchers are asking whether these measures will reduce the spread of the virus. Will fewer babies die from RSV, and fewer children develop permanent wheezing?
Recent studies offer clues.
Fabio Midulla, an associate professor of pediatrics at Sapienza University of Rome in Rome, Italy, said that the pharmaceutical industry is poised to push governments to use vaccines and monoclonal antibodies for even more children. “Such a push might work,” he said at the European Respiratory Society (ERS) 2024 Congress, “given that several studies have already demonstrated that their use can improve outcomes for children who do become infected and reduce societal costs by reducing hospitalizations.”
But Mariëlle WH Pijnenburg, a pulmonary specialist at Erasmus University Rotterdam in the Netherlands, said at the Congress that greater rollout would require governments to force industry to lower prices. If treatments remain beyond the reach of lower-income countries — where the burden of RSV is the greatest — the death toll from this common childhood infection will remain stubbornly high, and the prospect of global elimination will remain forever out of reach, she said.
New Tools in the Fight Against RSV
Nirsevimab, a long-acting monoclonal antibody given to newborns to prevent severe infection, was approved by the European Medicines Agency (EMA) in October 2022 and the US Food and Drug Administration (FDA) in July 2023. And Abrysvo, a vaccine given to older adults and pregnant women to stop them from passing the virus to babies from birth through 6 months of age, was approved by the FDA and the EMA in 2023.
RSV is responsible for over 33 million lung infections in children younger than 5 years annually, with more than 4 million hospitalizations and nearly 200,000 deaths. According to the Centers for Disease Control and Prevention, every year, 2.1 million children younger than 5 years old visit a healthcare provider because of an RSV infection and between 58,000 and 80,000 children younger than 5 years old are hospitalized in the United States. The burden of severe RSV disease is also high among adults, with an estimated 123,000-193,000 hospitalizations, 24,400-34,900 ICU admissions, and 4680-8620 in-hospital deaths occurring annually among US adults.
Infection in infancy can lead to later complications, such as the development of wheezing, a condition that causes breathlessness and a feeling of tightening in the chest, and possibly also asthma.
Studies have shown that children and preterm infants infected with RSV who were given monoclonal antibodies experienced less post-infection wheezing, suggesting that RSV prophylaxis could prevent the development of wheezing bronchitis.
A study conducted in Galicia, Spain, showed that only 0.3% of infants who received prophylaxis with Nirsevimab were hospitalized for RSV-related lower respiratory tract infections. “This is very promising,” Yvonne Maldonado, MD, professor of pediatrics and epidemiology and population health at Stanford University in Stanford, California, told Medscape Medical News. “But this virus is ubiquitous. It’s found everywhere. It comes around every winter season. And immunity is not long-lasting.”
Older children who are not receiving monoclonal antibodies still experience RSV-related hospitalizations, suggesting the virus continues to circulate at high enough levels in the community. “The vaccine and monoclonal antibodies can reduce the risk of hospitalization and more severe disease in young kids, but they won’t eliminate the virus,” Maldonado said. “Right now, the goal is to prevent serious infection, not to prevent the spread of the virus completely.”
Expanding Access to RSV Prevention in Low-Income Countries
Currently, the RSV vaccine and monoclonal antibodies are only given in the United States, Europe, United Kingdom, and Canada to newborns, children at risk for severe disease, and pregnant women. However, Midulla said that pharmaceutical companies are pushing to broaden the rollout to a broader population within these countries. Yet, he said, over 99% of RSV infection–related deaths occur in the Global South.
No pharmaceutical company has sought approval in low-income countries such as those in Africa. “Unless they see there being a market in a country, they’re not going to go through the onerous process of getting [a vaccine] licensed,” Shabir Madhi, dean of the faculty of health sciences and a professor of vaccinology at the University of the Witwatersrand, Johannesburg, South Africa, told Medscape Medical News.
He highlighted that almost 50% of RSV-related deaths occur in African children younger than 5 years, despite these children comprising just one fifth of the global under-5 population. The high burden of RSV mortality in the Global South is mainly due to poor access to healthcare and supportive treatments, such as supplemental oxygen, which can help children recover from severe RSV infection.
Companies are unlikely to pursue regulatory approval and licensing in low- and middle-income countries until GAVI, the global vaccine alliance, commits to procuring and funding the vaccines for these regions. GAVI’s decision would provide the necessary market incentive for manufacturers to seek approval.
Madhi suggested that GAVI’s decision on RSV vaccine procurement is imminent, likely early next year, following the World Health Organization’s Strategic Advisory Group of Experts on Immunization recommendation to vaccinate all pregnant women with the RSV vaccine, regardless of whether they are in high-income or low-income countries.
Nevertheless, even if vaccines become available, many African countries may still struggle to afford them. Madhi said that these countries would likely depend on GAVI and organizations like UNICEF to procure the vaccines at affordable prices. “The unfortunate reality is that many countries — especially in Africa — still wouldn’t be able to afford it, even if the vaccine cost as little as $5,” said Madhi. “But that’s where they would have the greatest impact.”
Midulla, Pijnenburg reported no relevant financial relationships. Madhi’s research unit, the Vaccines and Infectious Disease Analytics Unit, was involved in the clinical trials for the Pfizer RSV vaccine, the GSK RSV vaccine (which was terminated), as well as the MEDLEY trial of palivizumab. All funding for these studies went to his institution, the University of the Witwatersrand. Maldonado was Stanford principal investigator for the Pfizer RSV vaccine.
A version of this article appeared on Medscape.com.
Hypermucoviscous K pneumoniae Shows Reduced Drug Resistance
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
First Human Bird Flu Death Confirmed in US; Overall Risk Remains Low
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Group A Streptococcal Pharyngitis Diagnosis
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)
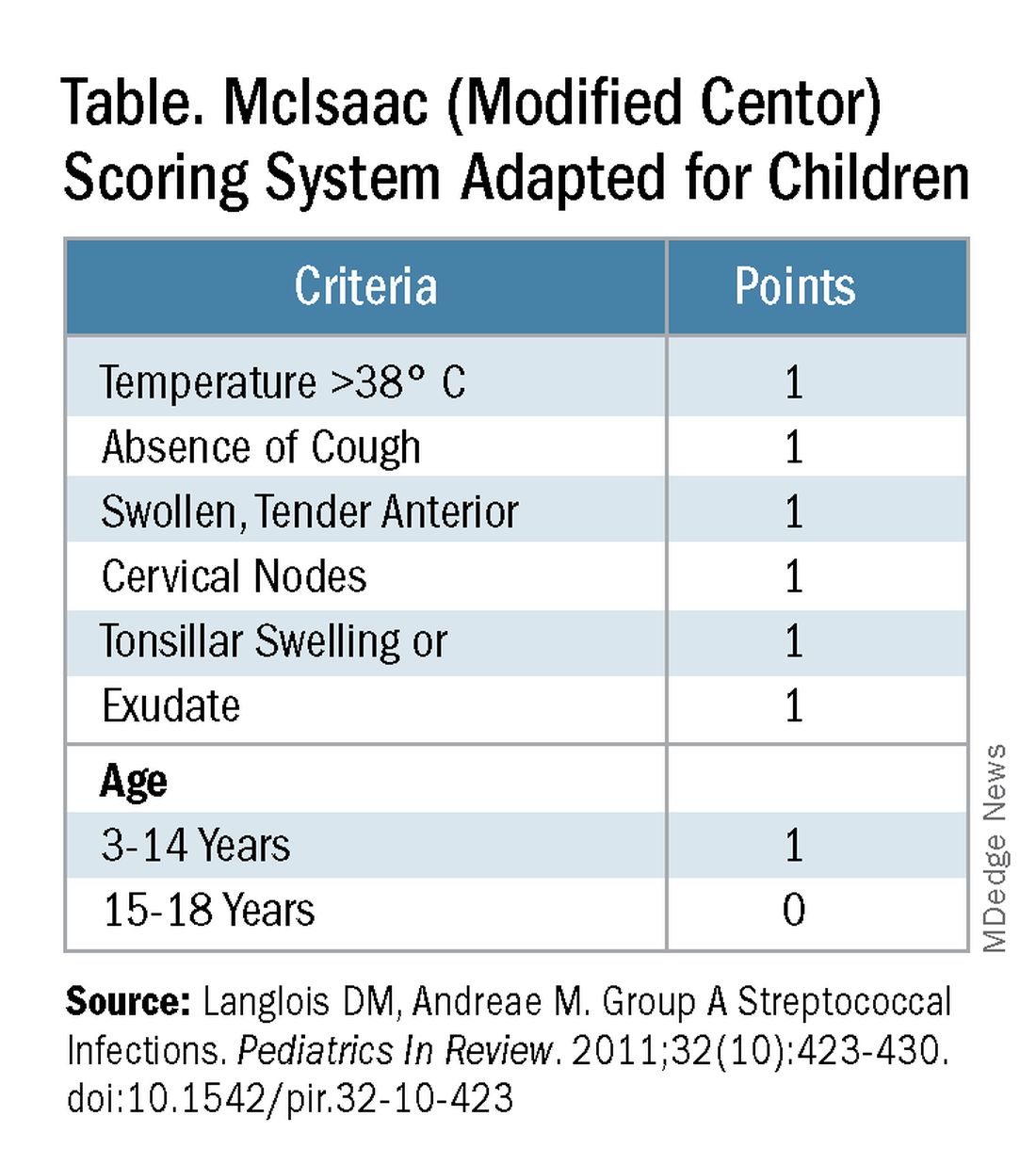
However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)
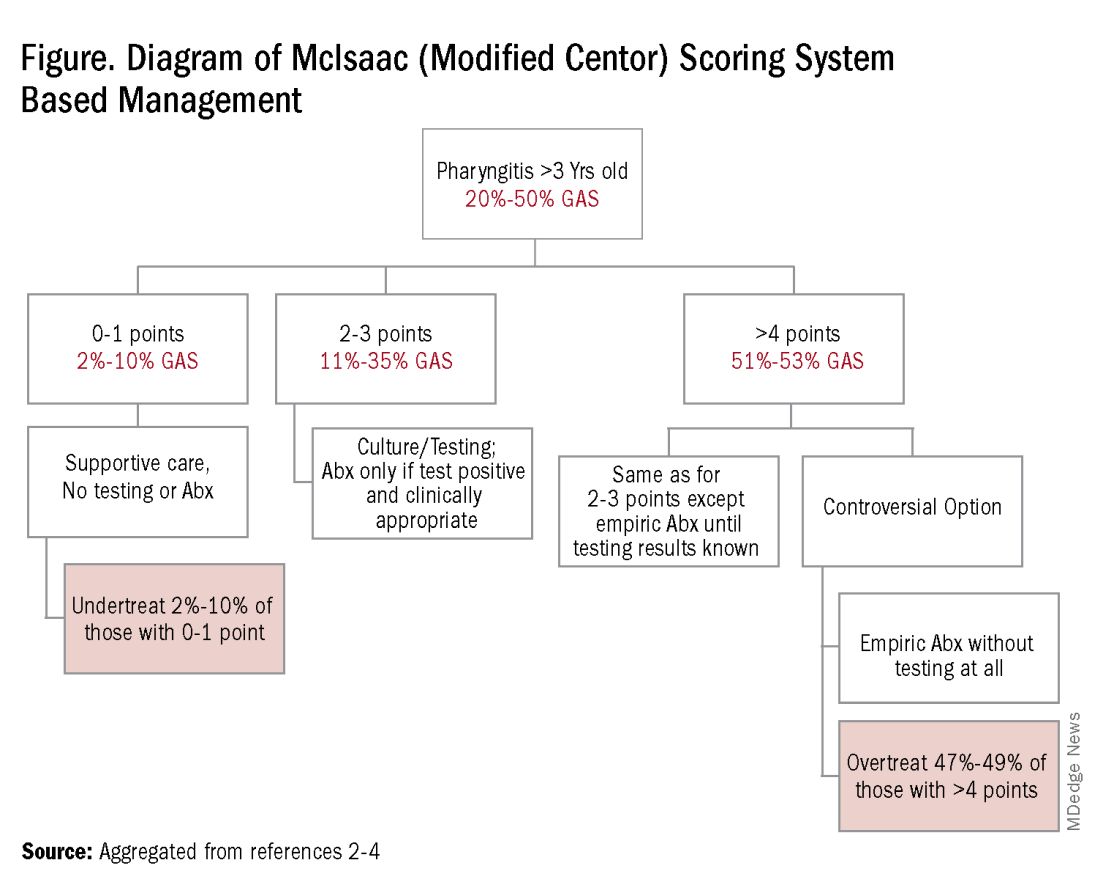
Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)

However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)

Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)

However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)

Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at pdnews@mdedge.com.
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
Central Line Skin Reactions in Children: Survey Addresses Treatment Protocols in Use
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Imipenem-Cilastatin-Relebactam, the New Go-To for Pneumonia?
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Doxycycline Kits Boost Chlamydia Treatment in ED
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Common Gut Infection Tied to Alzheimer’s Disease
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S & DEMENTIA

