User login
Kidney function may help docs pick antiplatelet mix after stroke
Renal function should be considered when determining whether to pick ticagrelor-aspirin or clopidogrel-aspirin as the antiplatelet therapy for patients with minor stroke, according to new research.
The study, which was conducted in 202 centers in China and published in Annals of Internal Medicine, indicates that when patients had normal kidney function, ticagrelor-aspirin, compared with clopidogrel-aspirin, substantially reduced the risk for recurrent stroke within 90 days of follow-up.
However, this effect was not seen in patients with mildly, moderately or severely decreased kidney function.
Rates of severe or moderate bleeding did not differ substantially between the two treatments.
Results gleaned from CHANCE-2 data
The researchers, led by Anxin Wang, PhD, from Capital Medical University in Beijing, conducted a post hoc analysis of the CHANCE-2 (Clopidogrel in High-Risk Patients with Acute Nondisabling Cerebrovascular Events-II) trial.
The trial included 6,378 patients who carried cytochrome P450 2C19 (CYP2C19) loss-of-function (LOF) alleles who had experienced a minor stroke or transient ischemic attack.
Patients received either ticagrelor-aspirin or clopidogrel-aspirin, and their renal function was measured by estimated glomerular filtration rate. The authors listed as a limitation that no data were available on the presence of albuminuria or proteinuria.
The researchers investigated what effect renal function had on the efficacy and safety of the therapies.
Differences in the therapies
Clopidogrel-aspirin is often recommended for preventing stroke. It can reduce thrombotic risk in patients with impaired kidney function, the authors noted. Ticagrelor can provide greater, faster, and more consistent P2Y12 inhibition than clopidogrel, and evidence shows it is effective in preventing stroke recurrence, particularly in people carrying CYP2C19 LOF alleles.
When people have reduced kidney function, clopidogrel may be harder to clear than ticagrelor and there may be increased plasma concentrations, so function is important to consider when choosing an antiplatelet therapy, the authors wrote.
Choice may come down to cost
Geoffrey Barnes, MD, MSc, associate professor of vascular and cardiovascular medicine at University of Michigan Medicine in Ann Arbor, said in an interview that there has been momentum toward ticagrelor as a more potent choice than clopidogrel not just in populations with minor stroke but for people with MI and coronary stents.
He said he found the results surprising and was intrigued that this paper suggests looking more skeptically at ticagrelor when kidney function is impaired.
Still, the choice may also come down to what the patient can afford at the pharmacy, he said.
“The reality is many patients still get clopidogrel either because that’s what their physicians have been prescribing for well over a decade or because of cost issues, and clopidogrel, for many patients, can be less expensive,” Dr. Barnes noted.
He said he would like to see more study in different populations as the prevalence of people carrying CYP2C19 allele differs by race and results might be different in a non-Asian population. That allele is thought to affect how clopidogrel is metabolized.
Study should spur more research
Nada El Husseini, MD, associate professor of neurology and Duke Telestroke Medical Director at Duke University Medical Center, Durham, N.C., said the study is hypothesis generating, but shouldn’t be thought of as the last word on the subject.
She pointed out some additional limitations of the study, including that it was a post hoc analysis. She explained that the question researchers asked in this study – about effect of kidney function on the safety and efficacy of the therapies – was not the focus of the original CHANCE-2 study, and, as such, the post hoc study may have been underpowered to answer the renal function question.
The authors acknowledged that limitation, noting that “the proportion of patients with severely decreased renal function was low.”
Among 6,378 patients, 4,050 (63.5%) had normal kidney function, 2,010 (31.5%) had mildly decreased function, and 318 (5.0%) had moderately to severely decreased function.
The study was funded by the Ministry of Science and Technology of the People’s Republic of China, the Beijing Municipal Science and Technology Commission, the Chinese Stroke Association, the National Science and Technology Major Project and the Beijing Municipal Administration of Hospitals Incubating Program). Salubris Pharmaceuticals contributed ticagrelor and, clopidogrel at no cost and with no restrictions. Dr. Wang reported no relevant financial relationships. Dr. Barnes and Dr. El Husseini reported no relevant financial relationships.
Renal function should be considered when determining whether to pick ticagrelor-aspirin or clopidogrel-aspirin as the antiplatelet therapy for patients with minor stroke, according to new research.
The study, which was conducted in 202 centers in China and published in Annals of Internal Medicine, indicates that when patients had normal kidney function, ticagrelor-aspirin, compared with clopidogrel-aspirin, substantially reduced the risk for recurrent stroke within 90 days of follow-up.
However, this effect was not seen in patients with mildly, moderately or severely decreased kidney function.
Rates of severe or moderate bleeding did not differ substantially between the two treatments.
Results gleaned from CHANCE-2 data
The researchers, led by Anxin Wang, PhD, from Capital Medical University in Beijing, conducted a post hoc analysis of the CHANCE-2 (Clopidogrel in High-Risk Patients with Acute Nondisabling Cerebrovascular Events-II) trial.
The trial included 6,378 patients who carried cytochrome P450 2C19 (CYP2C19) loss-of-function (LOF) alleles who had experienced a minor stroke or transient ischemic attack.
Patients received either ticagrelor-aspirin or clopidogrel-aspirin, and their renal function was measured by estimated glomerular filtration rate. The authors listed as a limitation that no data were available on the presence of albuminuria or proteinuria.
The researchers investigated what effect renal function had on the efficacy and safety of the therapies.
Differences in the therapies
Clopidogrel-aspirin is often recommended for preventing stroke. It can reduce thrombotic risk in patients with impaired kidney function, the authors noted. Ticagrelor can provide greater, faster, and more consistent P2Y12 inhibition than clopidogrel, and evidence shows it is effective in preventing stroke recurrence, particularly in people carrying CYP2C19 LOF alleles.
When people have reduced kidney function, clopidogrel may be harder to clear than ticagrelor and there may be increased plasma concentrations, so function is important to consider when choosing an antiplatelet therapy, the authors wrote.
Choice may come down to cost
Geoffrey Barnes, MD, MSc, associate professor of vascular and cardiovascular medicine at University of Michigan Medicine in Ann Arbor, said in an interview that there has been momentum toward ticagrelor as a more potent choice than clopidogrel not just in populations with minor stroke but for people with MI and coronary stents.
He said he found the results surprising and was intrigued that this paper suggests looking more skeptically at ticagrelor when kidney function is impaired.
Still, the choice may also come down to what the patient can afford at the pharmacy, he said.
“The reality is many patients still get clopidogrel either because that’s what their physicians have been prescribing for well over a decade or because of cost issues, and clopidogrel, for many patients, can be less expensive,” Dr. Barnes noted.
He said he would like to see more study in different populations as the prevalence of people carrying CYP2C19 allele differs by race and results might be different in a non-Asian population. That allele is thought to affect how clopidogrel is metabolized.
Study should spur more research
Nada El Husseini, MD, associate professor of neurology and Duke Telestroke Medical Director at Duke University Medical Center, Durham, N.C., said the study is hypothesis generating, but shouldn’t be thought of as the last word on the subject.
She pointed out some additional limitations of the study, including that it was a post hoc analysis. She explained that the question researchers asked in this study – about effect of kidney function on the safety and efficacy of the therapies – was not the focus of the original CHANCE-2 study, and, as such, the post hoc study may have been underpowered to answer the renal function question.
The authors acknowledged that limitation, noting that “the proportion of patients with severely decreased renal function was low.”
Among 6,378 patients, 4,050 (63.5%) had normal kidney function, 2,010 (31.5%) had mildly decreased function, and 318 (5.0%) had moderately to severely decreased function.
The study was funded by the Ministry of Science and Technology of the People’s Republic of China, the Beijing Municipal Science and Technology Commission, the Chinese Stroke Association, the National Science and Technology Major Project and the Beijing Municipal Administration of Hospitals Incubating Program). Salubris Pharmaceuticals contributed ticagrelor and, clopidogrel at no cost and with no restrictions. Dr. Wang reported no relevant financial relationships. Dr. Barnes and Dr. El Husseini reported no relevant financial relationships.
Renal function should be considered when determining whether to pick ticagrelor-aspirin or clopidogrel-aspirin as the antiplatelet therapy for patients with minor stroke, according to new research.
The study, which was conducted in 202 centers in China and published in Annals of Internal Medicine, indicates that when patients had normal kidney function, ticagrelor-aspirin, compared with clopidogrel-aspirin, substantially reduced the risk for recurrent stroke within 90 days of follow-up.
However, this effect was not seen in patients with mildly, moderately or severely decreased kidney function.
Rates of severe or moderate bleeding did not differ substantially between the two treatments.
Results gleaned from CHANCE-2 data
The researchers, led by Anxin Wang, PhD, from Capital Medical University in Beijing, conducted a post hoc analysis of the CHANCE-2 (Clopidogrel in High-Risk Patients with Acute Nondisabling Cerebrovascular Events-II) trial.
The trial included 6,378 patients who carried cytochrome P450 2C19 (CYP2C19) loss-of-function (LOF) alleles who had experienced a minor stroke or transient ischemic attack.
Patients received either ticagrelor-aspirin or clopidogrel-aspirin, and their renal function was measured by estimated glomerular filtration rate. The authors listed as a limitation that no data were available on the presence of albuminuria or proteinuria.
The researchers investigated what effect renal function had on the efficacy and safety of the therapies.
Differences in the therapies
Clopidogrel-aspirin is often recommended for preventing stroke. It can reduce thrombotic risk in patients with impaired kidney function, the authors noted. Ticagrelor can provide greater, faster, and more consistent P2Y12 inhibition than clopidogrel, and evidence shows it is effective in preventing stroke recurrence, particularly in people carrying CYP2C19 LOF alleles.
When people have reduced kidney function, clopidogrel may be harder to clear than ticagrelor and there may be increased plasma concentrations, so function is important to consider when choosing an antiplatelet therapy, the authors wrote.
Choice may come down to cost
Geoffrey Barnes, MD, MSc, associate professor of vascular and cardiovascular medicine at University of Michigan Medicine in Ann Arbor, said in an interview that there has been momentum toward ticagrelor as a more potent choice than clopidogrel not just in populations with minor stroke but for people with MI and coronary stents.
He said he found the results surprising and was intrigued that this paper suggests looking more skeptically at ticagrelor when kidney function is impaired.
Still, the choice may also come down to what the patient can afford at the pharmacy, he said.
“The reality is many patients still get clopidogrel either because that’s what their physicians have been prescribing for well over a decade or because of cost issues, and clopidogrel, for many patients, can be less expensive,” Dr. Barnes noted.
He said he would like to see more study in different populations as the prevalence of people carrying CYP2C19 allele differs by race and results might be different in a non-Asian population. That allele is thought to affect how clopidogrel is metabolized.
Study should spur more research
Nada El Husseini, MD, associate professor of neurology and Duke Telestroke Medical Director at Duke University Medical Center, Durham, N.C., said the study is hypothesis generating, but shouldn’t be thought of as the last word on the subject.
She pointed out some additional limitations of the study, including that it was a post hoc analysis. She explained that the question researchers asked in this study – about effect of kidney function on the safety and efficacy of the therapies – was not the focus of the original CHANCE-2 study, and, as such, the post hoc study may have been underpowered to answer the renal function question.
The authors acknowledged that limitation, noting that “the proportion of patients with severely decreased renal function was low.”
Among 6,378 patients, 4,050 (63.5%) had normal kidney function, 2,010 (31.5%) had mildly decreased function, and 318 (5.0%) had moderately to severely decreased function.
The study was funded by the Ministry of Science and Technology of the People’s Republic of China, the Beijing Municipal Science and Technology Commission, the Chinese Stroke Association, the National Science and Technology Major Project and the Beijing Municipal Administration of Hospitals Incubating Program). Salubris Pharmaceuticals contributed ticagrelor and, clopidogrel at no cost and with no restrictions. Dr. Wang reported no relevant financial relationships. Dr. Barnes and Dr. El Husseini reported no relevant financial relationships.
FROM ANNALS OF INTERNAL MEDICINE
Terlipressin decreases need for renal replacement therapy in liver transplant recipients
In a subgroup of patients with hepatorenal syndrome type 1 (HRS) who received a liver transplant, terlipressin treatment appears to reduce the need for renal replacement therapy (RRT) through 12 months of follow-up, according to a study presented at the annual meeting of the American College of Gastroenterology.
Among transplant recipients, overall 12-month survival was 11% higher for those treated with terlipressin compared with placebo, said K. Rajender Reddy, MD, director of hepatology and medical director of liver transplantation at the University of Pennsylvania, Philadelphia.
“Hepatorenal syndrome type 1 is a potentially reversible form of acute kidney injury that occurs in the setting of end-stage liver disease,” he said.
Liver transplantation, which eliminates end-stage liver disease, is the only definitive treatment for HRS. However, renal replacement therapy is common and associated with poor clinical outcomes and low patient survival rates in both the pretransplant and posttransplant settings, he noted.
Terlipressin, an injectable synthetic vasopressin analogue, restores renal blood flow and reverses HRS in 20%-40% of patients, Dr. Reddy said. In September, the U.S. Food and Drug Administration approved terlipressin (Terlivaz) for patients with HRS type 1. The label has a boxed warning for serious or fatal respiratory failure.
The safety and efficacy were assessed in the phase 3 CONFIRM trial, which Dr. Reddy and colleagues previously published. The randomized, placebo-controlled study demonstrated that terlipressin reversed HRS and reduced the need for RRT through day 30. The reversal of HRS with terlipressin did not improve 90-day survival as compared with placebo, which researchers attributed to a higher death rate within 90 days after the first dose despite improved kidney function.
A closer look at the liver transplant patients
In the subgroup analysis of the CONFIRM study, Dr. Reddy and colleagues analyzed the clinical outcomes through 12 months of follow-up in patients with HRS who received a liver transplant. They looked at the incidence of verified HRS reversal, HRS reversal, need for RRT, and overall survival.
Verified HRS reversal was defined as two consecutive serum creatinine measurements of 1.5 mg/dL or less at least 2 hours apart up to day 14 and survival without RRT for at least 10 days. HRS reversal was defined as a serum creatinine level of 1.5 mg/dL or less while on treatment. In addition, the need for RRT and overall survival were assessed at days 30, 60, 90, 180, and 365.
RRT was defined as any procedure that replaced nonendocrine kidney function, including continuous hemofiltration and hemodialysis, intermittent hemodialysis, peritoneal dialysis, ultrafiltration, or other dialysis and filtration techniques.
In the CONFIRM study, 199 patients with HRS were treated with terlipressin plus albumin, and 101 patients were treated with placebo plus albumin for up to 14 days. In the terlipressin group, 46 patients received liver transplants within the first 2 months of the study, as did 29 in the placebo group. Two patients in the terlipressin group and one in the placebo group received a simultaneous liver-kidney transplant.
Meaningful clinical outcomes
In the 12-month follow-up subgroup analysis, verified HRS reversal was statistically comparable between the groups, with a 30% decrease in the terlipressin group and 17% decrease in the placebo group, Dr. Reddy reported.
HRS reversal was higher in the terlipressin group, at 37%, as compared with 14% in the placebo group.
The pretransplant need for RRT was lower in the terlipressin group, at 30%, as compared with 62% in the placebo group. The posttransplant need for RRT remained numerically lower in the terlipressin group at all time points and was significantly lower at day 180 and day 365.
Overall survival for transplant recipients in the terlipressin group was 94%, as compared with 83% in the placebo group. Posttreatment adverse events and severe adverse events were similar between the groups.
“Collectively, these data indicate that terlipressin treatment in patients with HRS led to better long-term clinical outcomes in those who received a liver transplant,” Dr. Reddy said.
The study was funded by Mallinckrodt Pharmaceuticals, which manufactures terlipressin. One author is an employee of Mallinckrodt, and the other authors have served in an advisory role or received grant support from Mallinckrodt. The authors also disclosed consultant roles and research support from several other pharmaceutical companies.
In a subgroup of patients with hepatorenal syndrome type 1 (HRS) who received a liver transplant, terlipressin treatment appears to reduce the need for renal replacement therapy (RRT) through 12 months of follow-up, according to a study presented at the annual meeting of the American College of Gastroenterology.
Among transplant recipients, overall 12-month survival was 11% higher for those treated with terlipressin compared with placebo, said K. Rajender Reddy, MD, director of hepatology and medical director of liver transplantation at the University of Pennsylvania, Philadelphia.
“Hepatorenal syndrome type 1 is a potentially reversible form of acute kidney injury that occurs in the setting of end-stage liver disease,” he said.
Liver transplantation, which eliminates end-stage liver disease, is the only definitive treatment for HRS. However, renal replacement therapy is common and associated with poor clinical outcomes and low patient survival rates in both the pretransplant and posttransplant settings, he noted.
Terlipressin, an injectable synthetic vasopressin analogue, restores renal blood flow and reverses HRS in 20%-40% of patients, Dr. Reddy said. In September, the U.S. Food and Drug Administration approved terlipressin (Terlivaz) for patients with HRS type 1. The label has a boxed warning for serious or fatal respiratory failure.
The safety and efficacy were assessed in the phase 3 CONFIRM trial, which Dr. Reddy and colleagues previously published. The randomized, placebo-controlled study demonstrated that terlipressin reversed HRS and reduced the need for RRT through day 30. The reversal of HRS with terlipressin did not improve 90-day survival as compared with placebo, which researchers attributed to a higher death rate within 90 days after the first dose despite improved kidney function.
A closer look at the liver transplant patients
In the subgroup analysis of the CONFIRM study, Dr. Reddy and colleagues analyzed the clinical outcomes through 12 months of follow-up in patients with HRS who received a liver transplant. They looked at the incidence of verified HRS reversal, HRS reversal, need for RRT, and overall survival.
Verified HRS reversal was defined as two consecutive serum creatinine measurements of 1.5 mg/dL or less at least 2 hours apart up to day 14 and survival without RRT for at least 10 days. HRS reversal was defined as a serum creatinine level of 1.5 mg/dL or less while on treatment. In addition, the need for RRT and overall survival were assessed at days 30, 60, 90, 180, and 365.
RRT was defined as any procedure that replaced nonendocrine kidney function, including continuous hemofiltration and hemodialysis, intermittent hemodialysis, peritoneal dialysis, ultrafiltration, or other dialysis and filtration techniques.
In the CONFIRM study, 199 patients with HRS were treated with terlipressin plus albumin, and 101 patients were treated with placebo plus albumin for up to 14 days. In the terlipressin group, 46 patients received liver transplants within the first 2 months of the study, as did 29 in the placebo group. Two patients in the terlipressin group and one in the placebo group received a simultaneous liver-kidney transplant.
Meaningful clinical outcomes
In the 12-month follow-up subgroup analysis, verified HRS reversal was statistically comparable between the groups, with a 30% decrease in the terlipressin group and 17% decrease in the placebo group, Dr. Reddy reported.
HRS reversal was higher in the terlipressin group, at 37%, as compared with 14% in the placebo group.
The pretransplant need for RRT was lower in the terlipressin group, at 30%, as compared with 62% in the placebo group. The posttransplant need for RRT remained numerically lower in the terlipressin group at all time points and was significantly lower at day 180 and day 365.
Overall survival for transplant recipients in the terlipressin group was 94%, as compared with 83% in the placebo group. Posttreatment adverse events and severe adverse events were similar between the groups.
“Collectively, these data indicate that terlipressin treatment in patients with HRS led to better long-term clinical outcomes in those who received a liver transplant,” Dr. Reddy said.
The study was funded by Mallinckrodt Pharmaceuticals, which manufactures terlipressin. One author is an employee of Mallinckrodt, and the other authors have served in an advisory role or received grant support from Mallinckrodt. The authors also disclosed consultant roles and research support from several other pharmaceutical companies.
In a subgroup of patients with hepatorenal syndrome type 1 (HRS) who received a liver transplant, terlipressin treatment appears to reduce the need for renal replacement therapy (RRT) through 12 months of follow-up, according to a study presented at the annual meeting of the American College of Gastroenterology.
Among transplant recipients, overall 12-month survival was 11% higher for those treated with terlipressin compared with placebo, said K. Rajender Reddy, MD, director of hepatology and medical director of liver transplantation at the University of Pennsylvania, Philadelphia.
“Hepatorenal syndrome type 1 is a potentially reversible form of acute kidney injury that occurs in the setting of end-stage liver disease,” he said.
Liver transplantation, which eliminates end-stage liver disease, is the only definitive treatment for HRS. However, renal replacement therapy is common and associated with poor clinical outcomes and low patient survival rates in both the pretransplant and posttransplant settings, he noted.
Terlipressin, an injectable synthetic vasopressin analogue, restores renal blood flow and reverses HRS in 20%-40% of patients, Dr. Reddy said. In September, the U.S. Food and Drug Administration approved terlipressin (Terlivaz) for patients with HRS type 1. The label has a boxed warning for serious or fatal respiratory failure.
The safety and efficacy were assessed in the phase 3 CONFIRM trial, which Dr. Reddy and colleagues previously published. The randomized, placebo-controlled study demonstrated that terlipressin reversed HRS and reduced the need for RRT through day 30. The reversal of HRS with terlipressin did not improve 90-day survival as compared with placebo, which researchers attributed to a higher death rate within 90 days after the first dose despite improved kidney function.
A closer look at the liver transplant patients
In the subgroup analysis of the CONFIRM study, Dr. Reddy and colleagues analyzed the clinical outcomes through 12 months of follow-up in patients with HRS who received a liver transplant. They looked at the incidence of verified HRS reversal, HRS reversal, need for RRT, and overall survival.
Verified HRS reversal was defined as two consecutive serum creatinine measurements of 1.5 mg/dL or less at least 2 hours apart up to day 14 and survival without RRT for at least 10 days. HRS reversal was defined as a serum creatinine level of 1.5 mg/dL or less while on treatment. In addition, the need for RRT and overall survival were assessed at days 30, 60, 90, 180, and 365.
RRT was defined as any procedure that replaced nonendocrine kidney function, including continuous hemofiltration and hemodialysis, intermittent hemodialysis, peritoneal dialysis, ultrafiltration, or other dialysis and filtration techniques.
In the CONFIRM study, 199 patients with HRS were treated with terlipressin plus albumin, and 101 patients were treated with placebo plus albumin for up to 14 days. In the terlipressin group, 46 patients received liver transplants within the first 2 months of the study, as did 29 in the placebo group. Two patients in the terlipressin group and one in the placebo group received a simultaneous liver-kidney transplant.
Meaningful clinical outcomes
In the 12-month follow-up subgroup analysis, verified HRS reversal was statistically comparable between the groups, with a 30% decrease in the terlipressin group and 17% decrease in the placebo group, Dr. Reddy reported.
HRS reversal was higher in the terlipressin group, at 37%, as compared with 14% in the placebo group.
The pretransplant need for RRT was lower in the terlipressin group, at 30%, as compared with 62% in the placebo group. The posttransplant need for RRT remained numerically lower in the terlipressin group at all time points and was significantly lower at day 180 and day 365.
Overall survival for transplant recipients in the terlipressin group was 94%, as compared with 83% in the placebo group. Posttreatment adverse events and severe adverse events were similar between the groups.
“Collectively, these data indicate that terlipressin treatment in patients with HRS led to better long-term clinical outcomes in those who received a liver transplant,” Dr. Reddy said.
The study was funded by Mallinckrodt Pharmaceuticals, which manufactures terlipressin. One author is an employee of Mallinckrodt, and the other authors have served in an advisory role or received grant support from Mallinckrodt. The authors also disclosed consultant roles and research support from several other pharmaceutical companies.
FROM ACG 2022
Goodbye ‘diabetes insipidus’, hello ‘AVP-D’ and ‘AVP-R’
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
Islet transplants in type 1 diabetes durable up to 8 years
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE
‘Too good to be true’? Ultrasound safely treats kidney stones
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
FROM THE JOURNAL OF UROLOGY
Finerenone benefits T2D across spectrum of renal function
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
FROM JACC: HEART FAILURE
Ultrasonic renal denervation passes 2-month test in uncontrolled HTN: RADIANCE II
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.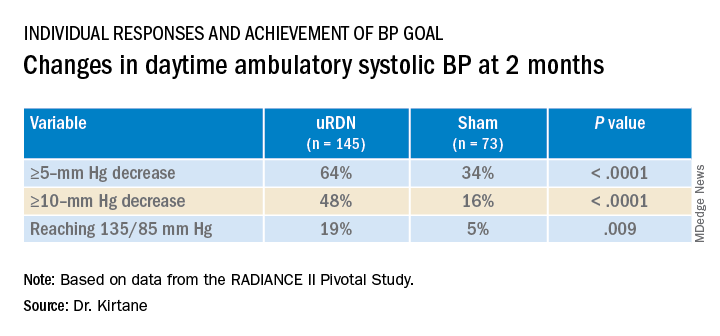
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).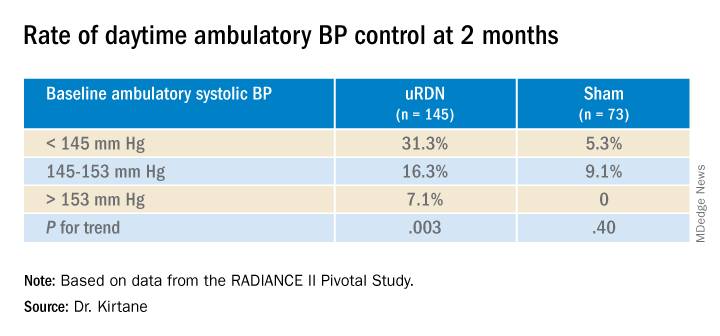
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
Systolic blood pressure went down safely and consistently 2 months after renal denervation achieved by ultrasound ablation in patients with uncontrolled, mild to moderate hypertension (HTN) in a key sham-controlled test of the balloon-equipped catheter.
The BP reductions were significant almost regardless of how they were measured – at home, in the office, during the day, at night, or over 24 hours – and weren’t dependent on baseline BP levels.
The 224-patient RADIANCE II Pivotal Study follows two earlier successful sham-controlled trials that used the same renal denervation catheter in other types of patients with HTN. They were RADIANCE-HTN SOLO, which entered patients with mild to moderate HTN not taking medication, and RADIANCE-HTN TRIO, which included patients with HTN despite fixed-dose, single-pill, triple-antihypertensive therapy.
The consistent results of all three trials suggest that the ultrasound renal denervation (uRDN) technique “lowers blood pressure across the spectrum of hypertension,” concluded co–principal investigator Ajay J. Kirtane, MD, SM, Columbia University Irving Medical Center, New York–Presbyterian Hospital, when presenting RADIANCE II at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation.
RADIANCE II, the largest of the three studies, met its prespecified primary efficacy endpoint of change in daytime ambulatory systolic BP at 2 months by showing a significant 6.3–mm Hg greater reduction in the uRDN group, compared with the sham-control group. There were no major adverse events at 30 days in either group.
The trial was similarly successful for the secondary endpoints of change in systolic BP measured in various other settings, including over 24 hours. Reductions after uRDN averaged 5-7 mm Hg greater than in the control group.
Sparse top-line results of the RADIANCE II pivotal trial were announced in July by the study’s sponsor, ReCor Medical.
Dr. Kirtane stressed in an interview that uRDN and likely any form of HTN renal denervation therapy is not a substitute for standard management. “This is really for patients in whom you’ve made best efforts to do the traditional things – lifestyle modification, medications, all of that – and yet they’re still uncontrolled.” At that point, assuming denervation therapy is available in practice, “it would be something to potentially consider.”
As a panelist after Dr. Kirtane’s formal presentation of RADIANCE II at the conference, Naomi D. Fisher, MD, who was a RADIANCE-HTN TRIO investigator, described how the treatment’s perceived intended patient population evolved over time.
“We all began with the idea that we were going to treat patients with resistant hypertension, that was going to be the first target. We have learned that those patients are far fewer than we thought,” said Dr. Fisher, who directs the hypertension service at Brigham and Women’s Hospital, Boston.
Initial estimates were that such patients with the resistant form, “meaning they require more than three drugs to control their blood pressure,” would represent 15%-20% of patients with HTN.
“We learned from our TRIO data that if you give these patients one single combined pill, lo and behold, many of them become controlled,” she said. “There is so much nonadherence out there in the world, about 50% of our patients aren’t taking their pills. It’s a hard and true fact.”
Exclude patients who aren’t adherent and “our true resistance population becomes minuscule. So, I don’t think that’s going to be the main population” for renal denervation therapy.
More likely, she said, it would be “patients who are uncontrolled and unable to take their medications. So that is going to include nonadherence, intolerance. It’s a very large category of patients. And the priorities can be stacked in favor of those who have higher cardiovascular risk.”
RADIANCE II can show the persistence of uRDN’s BP-lowering effect only out to 2 months so far, but the effect’s durability based on the RADIANCE program’s combined experience appears to be at least 2 years, Dr. Kirtane said in an interview.
“The RADIANCE II pivotal trial is a powerful, well-designed study attesting to the efficacy of renal denervation in BP lowering,” Franz H. Messerli, MD, Swiss Cardiovascular Center, University Hospital Bern, said in an interview.
The trial “shows the well-known unpredictability of antihypertensive response. We cannot predict who responds to renal denervation and who does not, and who even has a paradoxical increase in BP,” Dr. Messerli, an international hypertension expert not associated with the trial, said in an interview.
“As long as we cannot predict the antihypertensive response to renal denervation therapy, potential synergism/antagonism with drug therapy remains an educated guess,” he said.
“Hypertension is a disease that lasts years and decades. As impressive as RADIANCE II’s 2-month snapshot is, I look forward to similar or better BP data 12 and 24 months after renal denervation,” Dr. Messerli added.
RADIANCE II entered patients with mild to moderate uncontrolled HTN, that is, a systolic BP at least 140/90 mm Hg and less than 180/120 mm Hg, who were receiving no more than two antihypertensive medications. They could have no history of cardiovascular or cerebrovascular events or uncontrolled diabetes, and their estimated glomerular filtration rate (eGFR) had to be at least 40 mL/min per 1.73 m2.
After a 4-week drug washout period, patients who were clinically stable with an ambulatory BP of at least 135/85 mm Hg and less than 170/105 mm Hg underwent CT and renal angiography. Then, the 224 patients still anatomically eligible for the procedure were randomly assigned 2:1 to uRDN or a sham-control procedure: 150 and 74 patients, respectively.
At 2 months, daytime ambulatory systolic BP on average fell 7.9 mm Hg in the uRDN group and 1.8 mm Hg in the sham-control group, for a drop that was steeper by 6.3 mm Hg (P < .0001) after uRDN.
Also in the uRDN group, there was a 6.2–mm Hg larger decrease in 24-hour ambulatory systolic BP (P < .0001), a 5.8–mm Hg greater decline in nighttime ambulatory systolic BP (P < .0004), a 7.6–mm Hg steeper drop in mean home systolic BP (P < .0001), and 5.4 mm Hg more of a decrease in office-based systolic BP (P = .0035).
No significant differences were seen in subgroup analyses by sex, age, higher versus lower baseline systolic pressures, high versus low baseline eGFR, degree of abdominal obesity, U.S. versus European site, or whether patients entered before or during the COVID pandemic
Regulators have been accepting change in systolic BP as a surrogate for clinical endpoints in trials of antihypertensive therapy, whether pharmacologic or interventional, under consideration for approval. “That’s why safety endpoints are important to investigate” in these clinical trials, especially for invasive therapies like renal denervation, Dr. Kirtane observed.
That said, “in the longer-term follow-ups of the renal denervation therapies that are out there, including this one, there does not appear to be an appreciable decline in glomerular filtration rate, or any adverse safety signals that we see to date,” Dr. Kirtane said in an interview. “But we know that these are low-frequency events, so we have to be very vigilant, and we can’t get complacent about it.”
In RADIANCE II, there were zero adverse events within 30 days in both groups; the endpoint included death, new myocardial infarction, renal artery complications requiring invasive intervention, and hospitalization for major cardiovascular or hemodynamic-related events. Nor were there instances of new-onset renal artery stenosis greater than 70% documented by imaging at 6 months.
The ReCor uRDN catheter uses ultrasound energy to disrupt renal nerve signaling, a technology thought to deliver safer “burns,” compared with other renal denervation catheter technologies. It features an axially stabilizing balloon that transmits ultrasound energy – two to three sonications, each lasting 7 seconds, Dr. Kirtane said – outward through the arterial wall. The design is intended to ensure consistently circumferential ablation. Circulating saline within the balloon, Kirtane noted, directly cools the adjacent vessel wall to help it avoid thermal damage.
Dr. Kirtane reported receiving institutional funding from Medtronic, Boston Scientific, Abbott Vascular, Amgen, CSI, Philips, ReCor Medical, Neurotronic, Biotronik, Chiesi, Bolt Medical, Magenta Medical, Canon, SoniVie, Shockwave Medical, and Merck; consulting for IMDS; and receiving travel and meal expenses from Medtronic, Boston Scientific, Abbott Vascular, CSI, Siemens, Philips, ReCor Medical, Chiesi, OpSens, Zoll, and Regeneron. Dr. Fisher disclosed receiving honoraria or fees for consulting or serving on a speaker’s bureau for Medtronic, ReCor Medical, and Aktiia and receiving grant support or holding research contracts for Recor Medical and Aktiia. Dr. Messerli disclosed receiving honoraria from Medtronic, Menarini, Krka, and Ipca.
A version of this article first appeared on Medscape.com.
FROM TCT 2022
No invasive strategy benefit at 5 years in ISCHEMIA-CKD extension study
A trip to the cath lab for possible revascularization after a positive stress test, compared with a wait-and-see approach backed by optimal medications, did not improve 5-year survival for patients with advanced chronic kidney disease (CKD) in the ISCHEMIA-CKD trial’s extension study, ISCHEMIA-CKD EXTEND.
The long-term results, from the same 777 patients followed for an average of 2.2 years in the main trial, are consistent with the overall findings of no survival advantage with an initially invasive strategy, compared with one that is initially conservative. The finding applies to patients like those in the trial who had moderate to severe ischemia at stress testing and whose CKD put them in an especially high-risk and little-studied coronary artery disease (CAD) category.
Indeed, in a reflection of that high-risk status, 5-year all-cause mortality reached about 40% and cardiovascular (CV) mortality approached 30%, with no significant differences between patients in the invasive- and conservative-strategy groups.
Those numbers arguably put CKD’s effect on CAD survival in about the same league as an ejection fraction (EF) of 35% or less. For context, all-cause mortality over 3-4 years was about 32% in the REVIVED-BCIS2 trial of such patients with ischemic reduced-EF cardiomyopathy, whether or not they were revascularized, observed Sripal Bangalore, MD, MHA.
Yet in ISCHEMIA-CKD EXTEND, “you’re seeing in a group of patients, with largely preserved EF but advanced CKD, a mortality rate close to 40% at 5 years,” said Dr. Bangalore of New York University.
Although the study doesn’t show benefit from the initially invasive approach in CKD patients with stable CAD, for those with acute coronary syndromes (ACS), it seems to suggest that “at least the invasive strategy is safe,” Dr. Bangalore said during a press conference preceding his presentation of the study Aug. 29 at the annual congress of the European Society of Cardiology, held in Barcelona.
REVIVED-BCIS2 was also presented at the ESC sessions on Aug. 27, as reported by this news organization.
ISCHEMIA-CKD EXTEND “is a large trial and a very well-done trial. The results are robust, and they should influence clinical practice,” Deepak L. Bhatt, MD, MPH, Brigham and Women’s Hospital Heart & Vascular Center, Boston, said as the invited discussant after Dr. Bangalore’s presentation.
“The main message here, really, is don’t just go looking for ischemia, at least with the modalities used in this trial, in your CKD patients as a routine practice, and then try to stomp out that ischemia with revascularization,” Dr. Bhatt said. “The right thing to do in these high-risk patients is to focus on lifestyle modification and intensive medical therapy.”
A caveat, he said, is that the trial’s results don’t apply to the types of patients excluded from it, including those with recent ACS and those who are highly symptomatic or have an EF of less than 35%.
“Those CKD patients likely benefit from an invasive strategy with anatomically appropriate revascularization,” whether percutaneous coronary intervention (PCI) or coronary bypass surgery, Dr. Bhatt said.
At a median follow-up of 5 years in the extension study, the rates of death from any cause were 40.6% for patients in the invasive-strategy group and 37.4% for those in the conservative-strategy group. That yielded a hazard ratio of 1.12 (95% confidence interval, 0.89-1.41; P = .32) after adjustment for age, sex, diabetes status, EF, dialysis status, and – for patients not on dialysis – baseline estimated glomerular filtration rate.
The rates of CV death were 29% for patients managed invasively and 27% for those initially managed conservatively, for a similarly adjusted HR of 1.04 (95% CI, 0.80-1.37; P = .75).
In subgroup analyses, Dr. Bangalore reported, there were no significant differences in all-cause or CV mortality by diabetes status, by severity of baseline ischemia, or by whether the patient had recently experienced new or more frequent angina at study entry, was on guideline-directed medical therapy at baseline, or was on dialysis.
Among the contributions of ISCHEMIA-CKD and its 5-year extension study, Dr. Bangalore told this news organization, is that the relative safety of revascularization they showed may help to counter “renalism,” that is, the aversion to invasive intervention in patients with advanced CKD in clinical practice.
For example, if a patient with advanced CKD presents with an acute myocardial infarction, “people are hesitant to take them to the cath lab,” Dr. Bangalore said. But “if you follow protocols, if you follow strategies to minimize the risk, you can safely go ahead and do it.”
But in patients with stable CAD, as the ISCHEMIA-CKD studies show, “routinely revascularizing them may not have significant benefits.”
ISCHEMIC-CKD and its extension study were funded by the National Heart, Lung, and Blood Institute. Dr. Bangalore discloses receiving research grants from NHLBI and serving as a consultant for Abbott Vascular, Biotronik, Boston Scientific, Amgen, Pfizer, Merck, and Reata. Dr. Bhatt has disclosed grants and/or personal fees from many companies; personal fees from WebMD and other publications or organizations; and having other relationships with Medscape Cardiology and other publications or organizations.
A version of this article first appeared on Medscape.com.
A trip to the cath lab for possible revascularization after a positive stress test, compared with a wait-and-see approach backed by optimal medications, did not improve 5-year survival for patients with advanced chronic kidney disease (CKD) in the ISCHEMIA-CKD trial’s extension study, ISCHEMIA-CKD EXTEND.
The long-term results, from the same 777 patients followed for an average of 2.2 years in the main trial, are consistent with the overall findings of no survival advantage with an initially invasive strategy, compared with one that is initially conservative. The finding applies to patients like those in the trial who had moderate to severe ischemia at stress testing and whose CKD put them in an especially high-risk and little-studied coronary artery disease (CAD) category.
Indeed, in a reflection of that high-risk status, 5-year all-cause mortality reached about 40% and cardiovascular (CV) mortality approached 30%, with no significant differences between patients in the invasive- and conservative-strategy groups.
Those numbers arguably put CKD’s effect on CAD survival in about the same league as an ejection fraction (EF) of 35% or less. For context, all-cause mortality over 3-4 years was about 32% in the REVIVED-BCIS2 trial of such patients with ischemic reduced-EF cardiomyopathy, whether or not they were revascularized, observed Sripal Bangalore, MD, MHA.
Yet in ISCHEMIA-CKD EXTEND, “you’re seeing in a group of patients, with largely preserved EF but advanced CKD, a mortality rate close to 40% at 5 years,” said Dr. Bangalore of New York University.
Although the study doesn’t show benefit from the initially invasive approach in CKD patients with stable CAD, for those with acute coronary syndromes (ACS), it seems to suggest that “at least the invasive strategy is safe,” Dr. Bangalore said during a press conference preceding his presentation of the study Aug. 29 at the annual congress of the European Society of Cardiology, held in Barcelona.
REVIVED-BCIS2 was also presented at the ESC sessions on Aug. 27, as reported by this news organization.
ISCHEMIA-CKD EXTEND “is a large trial and a very well-done trial. The results are robust, and they should influence clinical practice,” Deepak L. Bhatt, MD, MPH, Brigham and Women’s Hospital Heart & Vascular Center, Boston, said as the invited discussant after Dr. Bangalore’s presentation.
“The main message here, really, is don’t just go looking for ischemia, at least with the modalities used in this trial, in your CKD patients as a routine practice, and then try to stomp out that ischemia with revascularization,” Dr. Bhatt said. “The right thing to do in these high-risk patients is to focus on lifestyle modification and intensive medical therapy.”
A caveat, he said, is that the trial’s results don’t apply to the types of patients excluded from it, including those with recent ACS and those who are highly symptomatic or have an EF of less than 35%.
“Those CKD patients likely benefit from an invasive strategy with anatomically appropriate revascularization,” whether percutaneous coronary intervention (PCI) or coronary bypass surgery, Dr. Bhatt said.
At a median follow-up of 5 years in the extension study, the rates of death from any cause were 40.6% for patients in the invasive-strategy group and 37.4% for those in the conservative-strategy group. That yielded a hazard ratio of 1.12 (95% confidence interval, 0.89-1.41; P = .32) after adjustment for age, sex, diabetes status, EF, dialysis status, and – for patients not on dialysis – baseline estimated glomerular filtration rate.
The rates of CV death were 29% for patients managed invasively and 27% for those initially managed conservatively, for a similarly adjusted HR of 1.04 (95% CI, 0.80-1.37; P = .75).
In subgroup analyses, Dr. Bangalore reported, there were no significant differences in all-cause or CV mortality by diabetes status, by severity of baseline ischemia, or by whether the patient had recently experienced new or more frequent angina at study entry, was on guideline-directed medical therapy at baseline, or was on dialysis.
Among the contributions of ISCHEMIA-CKD and its 5-year extension study, Dr. Bangalore told this news organization, is that the relative safety of revascularization they showed may help to counter “renalism,” that is, the aversion to invasive intervention in patients with advanced CKD in clinical practice.
For example, if a patient with advanced CKD presents with an acute myocardial infarction, “people are hesitant to take them to the cath lab,” Dr. Bangalore said. But “if you follow protocols, if you follow strategies to minimize the risk, you can safely go ahead and do it.”
But in patients with stable CAD, as the ISCHEMIA-CKD studies show, “routinely revascularizing them may not have significant benefits.”
ISCHEMIC-CKD and its extension study were funded by the National Heart, Lung, and Blood Institute. Dr. Bangalore discloses receiving research grants from NHLBI and serving as a consultant for Abbott Vascular, Biotronik, Boston Scientific, Amgen, Pfizer, Merck, and Reata. Dr. Bhatt has disclosed grants and/or personal fees from many companies; personal fees from WebMD and other publications or organizations; and having other relationships with Medscape Cardiology and other publications or organizations.
A version of this article first appeared on Medscape.com.
A trip to the cath lab for possible revascularization after a positive stress test, compared with a wait-and-see approach backed by optimal medications, did not improve 5-year survival for patients with advanced chronic kidney disease (CKD) in the ISCHEMIA-CKD trial’s extension study, ISCHEMIA-CKD EXTEND.
The long-term results, from the same 777 patients followed for an average of 2.2 years in the main trial, are consistent with the overall findings of no survival advantage with an initially invasive strategy, compared with one that is initially conservative. The finding applies to patients like those in the trial who had moderate to severe ischemia at stress testing and whose CKD put them in an especially high-risk and little-studied coronary artery disease (CAD) category.
Indeed, in a reflection of that high-risk status, 5-year all-cause mortality reached about 40% and cardiovascular (CV) mortality approached 30%, with no significant differences between patients in the invasive- and conservative-strategy groups.
Those numbers arguably put CKD’s effect on CAD survival in about the same league as an ejection fraction (EF) of 35% or less. For context, all-cause mortality over 3-4 years was about 32% in the REVIVED-BCIS2 trial of such patients with ischemic reduced-EF cardiomyopathy, whether or not they were revascularized, observed Sripal Bangalore, MD, MHA.
Yet in ISCHEMIA-CKD EXTEND, “you’re seeing in a group of patients, with largely preserved EF but advanced CKD, a mortality rate close to 40% at 5 years,” said Dr. Bangalore of New York University.
Although the study doesn’t show benefit from the initially invasive approach in CKD patients with stable CAD, for those with acute coronary syndromes (ACS), it seems to suggest that “at least the invasive strategy is safe,” Dr. Bangalore said during a press conference preceding his presentation of the study Aug. 29 at the annual congress of the European Society of Cardiology, held in Barcelona.
REVIVED-BCIS2 was also presented at the ESC sessions on Aug. 27, as reported by this news organization.
ISCHEMIA-CKD EXTEND “is a large trial and a very well-done trial. The results are robust, and they should influence clinical practice,” Deepak L. Bhatt, MD, MPH, Brigham and Women’s Hospital Heart & Vascular Center, Boston, said as the invited discussant after Dr. Bangalore’s presentation.
“The main message here, really, is don’t just go looking for ischemia, at least with the modalities used in this trial, in your CKD patients as a routine practice, and then try to stomp out that ischemia with revascularization,” Dr. Bhatt said. “The right thing to do in these high-risk patients is to focus on lifestyle modification and intensive medical therapy.”
A caveat, he said, is that the trial’s results don’t apply to the types of patients excluded from it, including those with recent ACS and those who are highly symptomatic or have an EF of less than 35%.
“Those CKD patients likely benefit from an invasive strategy with anatomically appropriate revascularization,” whether percutaneous coronary intervention (PCI) or coronary bypass surgery, Dr. Bhatt said.
At a median follow-up of 5 years in the extension study, the rates of death from any cause were 40.6% for patients in the invasive-strategy group and 37.4% for those in the conservative-strategy group. That yielded a hazard ratio of 1.12 (95% confidence interval, 0.89-1.41; P = .32) after adjustment for age, sex, diabetes status, EF, dialysis status, and – for patients not on dialysis – baseline estimated glomerular filtration rate.
The rates of CV death were 29% for patients managed invasively and 27% for those initially managed conservatively, for a similarly adjusted HR of 1.04 (95% CI, 0.80-1.37; P = .75).
In subgroup analyses, Dr. Bangalore reported, there were no significant differences in all-cause or CV mortality by diabetes status, by severity of baseline ischemia, or by whether the patient had recently experienced new or more frequent angina at study entry, was on guideline-directed medical therapy at baseline, or was on dialysis.
Among the contributions of ISCHEMIA-CKD and its 5-year extension study, Dr. Bangalore told this news organization, is that the relative safety of revascularization they showed may help to counter “renalism,” that is, the aversion to invasive intervention in patients with advanced CKD in clinical practice.
For example, if a patient with advanced CKD presents with an acute myocardial infarction, “people are hesitant to take them to the cath lab,” Dr. Bangalore said. But “if you follow protocols, if you follow strategies to minimize the risk, you can safely go ahead and do it.”
But in patients with stable CAD, as the ISCHEMIA-CKD studies show, “routinely revascularizing them may not have significant benefits.”
ISCHEMIC-CKD and its extension study were funded by the National Heart, Lung, and Blood Institute. Dr. Bangalore discloses receiving research grants from NHLBI and serving as a consultant for Abbott Vascular, Biotronik, Boston Scientific, Amgen, Pfizer, Merck, and Reata. Dr. Bhatt has disclosed grants and/or personal fees from many companies; personal fees from WebMD and other publications or organizations; and having other relationships with Medscape Cardiology and other publications or organizations.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
Gastrointestinal Stromal Tumor Arising From the Small Intestine in a Heart Transplant Recipient on Hemodialysis and Chronic Immunosuppression: A Case Report
Background
Gastrointestinal stromal tumors (GISTs) are rare mesenchymal tumors with worse prognosis if arising from the small bowel. Surgery remains the mainstay of treatment for patients with resectable tumors. Imatinib has become the standard treatment in cKIT-positive GISTs with significant morbidity in neoadjuvant, adjuvant, and palliative settings. There are limited data on efficacy and safety of imatinib in dialysis patients, and chemotherapy dosing is challenging in dialysis patients with multiple comorbidities.
Presentation
A 68-year-old male with a history of orthotopic heart transplantation on sirolimus with prednisone, cardiac allograft vasculopathy, plus ESRD on peritoneal dialysis (PD), presented with lower abdominal pain and fever. Abdominal imaging revealed a right lower quadrant (RLQ) mass with concern for bowel perforation.
Diagnosis and Treatment
The patient underwent exploratory laparoscopy with small bowel resection, excision of the mesenteric small bowel mass, drainage and washout of intraabdominal abscess, removal of PD catheter, and transition to hemodialysis. Pathology revealed a 14.5-cm high-grade GIST with mixed spindle and epithelioid types involving the ileal wall and mesentery, consistent with pT4 primary tumor and stage IIIB disease. Molecular testing was positive for c-KIT and DOG-1 mutations.
After a prolonged recovery, repeat abdominal imaging demonstrated metastatic liver disease and a new RLQ lesion. The patient was started on palliative imatinib 100 mg daily with subsequent increase to 200 mg daily. He was monitored closely for toxicities but reported only mild nausea controlled with ondansetron. Hemodialysis was continued 3 times per week. Follow up scans 3 months later showed improvement in RLQ mass and hepatic lesions. The patient remains on the current dose 15 months after the diagnosis.
Conclusion
To our knowledge, this is the first case of a small intestinal GIST in a heart transplant recipient treated with dose-reduced imatinib with concurrent dialysis and immunosuppression. Treatment decision-making was complex given concern for cardiotoxicity with pre-existing cardiac disease and drug-drug interactions with immunosuppressive agents. While some literature suggests standard dose imatinib with dialysis, no large-scale studies evaluated pharmacokinetics of imatinib with creatinine clearance < 20 mL/min. There is a need for further studies to determine dosing strategies for such patients.
Background
Gastrointestinal stromal tumors (GISTs) are rare mesenchymal tumors with worse prognosis if arising from the small bowel. Surgery remains the mainstay of treatment for patients with resectable tumors. Imatinib has become the standard treatment in cKIT-positive GISTs with significant morbidity in neoadjuvant, adjuvant, and palliative settings. There are limited data on efficacy and safety of imatinib in dialysis patients, and chemotherapy dosing is challenging in dialysis patients with multiple comorbidities.
Presentation
A 68-year-old male with a history of orthotopic heart transplantation on sirolimus with prednisone, cardiac allograft vasculopathy, plus ESRD on peritoneal dialysis (PD), presented with lower abdominal pain and fever. Abdominal imaging revealed a right lower quadrant (RLQ) mass with concern for bowel perforation.
Diagnosis and Treatment
The patient underwent exploratory laparoscopy with small bowel resection, excision of the mesenteric small bowel mass, drainage and washout of intraabdominal abscess, removal of PD catheter, and transition to hemodialysis. Pathology revealed a 14.5-cm high-grade GIST with mixed spindle and epithelioid types involving the ileal wall and mesentery, consistent with pT4 primary tumor and stage IIIB disease. Molecular testing was positive for c-KIT and DOG-1 mutations.
After a prolonged recovery, repeat abdominal imaging demonstrated metastatic liver disease and a new RLQ lesion. The patient was started on palliative imatinib 100 mg daily with subsequent increase to 200 mg daily. He was monitored closely for toxicities but reported only mild nausea controlled with ondansetron. Hemodialysis was continued 3 times per week. Follow up scans 3 months later showed improvement in RLQ mass and hepatic lesions. The patient remains on the current dose 15 months after the diagnosis.
Conclusion
To our knowledge, this is the first case of a small intestinal GIST in a heart transplant recipient treated with dose-reduced imatinib with concurrent dialysis and immunosuppression. Treatment decision-making was complex given concern for cardiotoxicity with pre-existing cardiac disease and drug-drug interactions with immunosuppressive agents. While some literature suggests standard dose imatinib with dialysis, no large-scale studies evaluated pharmacokinetics of imatinib with creatinine clearance < 20 mL/min. There is a need for further studies to determine dosing strategies for such patients.
Background
Gastrointestinal stromal tumors (GISTs) are rare mesenchymal tumors with worse prognosis if arising from the small bowel. Surgery remains the mainstay of treatment for patients with resectable tumors. Imatinib has become the standard treatment in cKIT-positive GISTs with significant morbidity in neoadjuvant, adjuvant, and palliative settings. There are limited data on efficacy and safety of imatinib in dialysis patients, and chemotherapy dosing is challenging in dialysis patients with multiple comorbidities.
Presentation
A 68-year-old male with a history of orthotopic heart transplantation on sirolimus with prednisone, cardiac allograft vasculopathy, plus ESRD on peritoneal dialysis (PD), presented with lower abdominal pain and fever. Abdominal imaging revealed a right lower quadrant (RLQ) mass with concern for bowel perforation.
Diagnosis and Treatment
The patient underwent exploratory laparoscopy with small bowel resection, excision of the mesenteric small bowel mass, drainage and washout of intraabdominal abscess, removal of PD catheter, and transition to hemodialysis. Pathology revealed a 14.5-cm high-grade GIST with mixed spindle and epithelioid types involving the ileal wall and mesentery, consistent with pT4 primary tumor and stage IIIB disease. Molecular testing was positive for c-KIT and DOG-1 mutations.
After a prolonged recovery, repeat abdominal imaging demonstrated metastatic liver disease and a new RLQ lesion. The patient was started on palliative imatinib 100 mg daily with subsequent increase to 200 mg daily. He was monitored closely for toxicities but reported only mild nausea controlled with ondansetron. Hemodialysis was continued 3 times per week. Follow up scans 3 months later showed improvement in RLQ mass and hepatic lesions. The patient remains on the current dose 15 months after the diagnosis.
Conclusion
To our knowledge, this is the first case of a small intestinal GIST in a heart transplant recipient treated with dose-reduced imatinib with concurrent dialysis and immunosuppression. Treatment decision-making was complex given concern for cardiotoxicity with pre-existing cardiac disease and drug-drug interactions with immunosuppressive agents. While some literature suggests standard dose imatinib with dialysis, no large-scale studies evaluated pharmacokinetics of imatinib with creatinine clearance < 20 mL/min. There is a need for further studies to determine dosing strategies for such patients.
Albuminuria linked to higher CVD risk in diabetes
BARCELONA – Fewer than half the adults in Denmark with type 2 diabetes in 2015 had a recent assessment for albuminuria, and those who underwent testing and had albuminuria had a greater than 50% increased rate of incident heart failure, MI, stroke, or all-cause death during 4-year follow-up, in a study of more than 74,000 Danish residents.
Even those in this study with type 2 diabetes but without albuminuria had a 19% rate of these adverse outcomes, highlighting the “substantial” cardiovascular disease risk faced by people with type 2 diabetes even without a clear indication of nephropathy, Saaima Parveen, MD, a cardiology researcher at Herlev and Gentofte University Hospital in Copenhagen, said at the annual congress of the European Society of Cardiology.
This high rate of heart failure, MI, stroke, or death even in the absence of what is conventionally defined as albuminuria – a urinary albumin-to-creatinine ratio (UACR) of at least 30 mg/g – suggests that this threshold for albuminuria may be too high, commented Luis M. Ruilope, MD, professor of public health and preventive medicine at Autonoma University, Madrid, who was not involved with the Danish study.
The study reported by Dr. Parveen “is very important because it shows that the risk of events is high not only in people with diabetes with albuminuria, but also in those without albuminuria,” Dr. Ruilope said in an interview.
The profile of albuminuria as a risk marker for people with type 2 diabetes spiked following the 2021 U.S. approval of finerenone (Kerendia) as an agent specifically targeted to adults with type 2 diabetes and albuminuria. (Finerenone gained marketing approval by in Europe in February 2022 under the same brand name.)
A lower threshold for albuminuria?
“Even patients with a UACR of 10-29 mg/g have risk and should be considered for finerenone treatment, said Dr. Ruilope. “People with type 2 diabetes with a UACR of 10-29 mg/g could explain” the high background risk shown by Dr. Parveen in her reported data. “In people with type 2 diabetes and a UACR of 10-29 mg/g we also see progression of kidney disease, but it’s slower” than in those who meet the current, standard threshold for albuminuria.
Dr. Ruilope was a coinvestigator for both of the finerenone pivotal trials, FIDELIO-DKD and FIGARO-DKD. Although the design of both these studies specified enrollment of people with type 2 diabetes and a UACR of at least 30 mg/g, a few hundred of the total combined enrollment of more than 13,000 patients had UACR values below this level, and analysis of this subgroup could provide some important insights into the value of finerenone for people with “high normal” albuminuria, he said.
The study led by Dr. Parveen used data routinely collected in Danish national records and focused on all Danish adults diagnosed with type 2 diabetes as of Jan. 1, 2015, who also had information in their records for a UACR and an estimated glomerular filtration rate (eGFR) within the preceding year.
The records showed that only 47% of these people had a UACR value during this time frame, and that 57% had a recent measure of their eGFR, despite prevailing recommendations for routine and regular measurements of these parameters for all people with type 2 diabetes.
Dr. Parveen hypothesized that UACR measurement may lag for several reasons, such as reliance by primary care physicians on urine dipstick assessments, which preclude calculation of a UACR, poor adherence to regular medical assessment by people in low socioeconomic groups, and medical examination done outside of morning time periods, which is the best time of day for assessing UACR.
More albuminuria measurement needed in primary care
“Measurement of albuminuria in people with type 2 diabetes is improving in Europe, but is not yet at the level that’s needed,” commented Dr. Ruilope. “We are pushing to have it done more often in primary care practices,” he said.
Among the 74,014 people with type 2 diabetes who had the measurement records that allowed for their inclusion in the study, 40% had albuminuria and 60% did not.
During 4 years of follow-up, the incidence of heart failure, MI, stroke, or all-cause death was 28.6% in those with albuminuria and 18.7% among those without albuminuria, reported Dr. Parveen.
The rates for each event type in those with albuminuria were 7.0% for heart failure, 4.4% for MI, 7.6% for stroke, and 16.6% for all-cause death (each patient could tally more than one type of event). Among those without albuminuria, the rates were 4.0%, 3.2%, 5.5%, and 9.3%, respectively.
The study received no commercial funding. Dr. Parveen and Dr. Ruilope had no disclosures.
BARCELONA – Fewer than half the adults in Denmark with type 2 diabetes in 2015 had a recent assessment for albuminuria, and those who underwent testing and had albuminuria had a greater than 50% increased rate of incident heart failure, MI, stroke, or all-cause death during 4-year follow-up, in a study of more than 74,000 Danish residents.
Even those in this study with type 2 diabetes but without albuminuria had a 19% rate of these adverse outcomes, highlighting the “substantial” cardiovascular disease risk faced by people with type 2 diabetes even without a clear indication of nephropathy, Saaima Parveen, MD, a cardiology researcher at Herlev and Gentofte University Hospital in Copenhagen, said at the annual congress of the European Society of Cardiology.
This high rate of heart failure, MI, stroke, or death even in the absence of what is conventionally defined as albuminuria – a urinary albumin-to-creatinine ratio (UACR) of at least 30 mg/g – suggests that this threshold for albuminuria may be too high, commented Luis M. Ruilope, MD, professor of public health and preventive medicine at Autonoma University, Madrid, who was not involved with the Danish study.
The study reported by Dr. Parveen “is very important because it shows that the risk of events is high not only in people with diabetes with albuminuria, but also in those without albuminuria,” Dr. Ruilope said in an interview.
The profile of albuminuria as a risk marker for people with type 2 diabetes spiked following the 2021 U.S. approval of finerenone (Kerendia) as an agent specifically targeted to adults with type 2 diabetes and albuminuria. (Finerenone gained marketing approval by in Europe in February 2022 under the same brand name.)
A lower threshold for albuminuria?
“Even patients with a UACR of 10-29 mg/g have risk and should be considered for finerenone treatment, said Dr. Ruilope. “People with type 2 diabetes with a UACR of 10-29 mg/g could explain” the high background risk shown by Dr. Parveen in her reported data. “In people with type 2 diabetes and a UACR of 10-29 mg/g we also see progression of kidney disease, but it’s slower” than in those who meet the current, standard threshold for albuminuria.
Dr. Ruilope was a coinvestigator for both of the finerenone pivotal trials, FIDELIO-DKD and FIGARO-DKD. Although the design of both these studies specified enrollment of people with type 2 diabetes and a UACR of at least 30 mg/g, a few hundred of the total combined enrollment of more than 13,000 patients had UACR values below this level, and analysis of this subgroup could provide some important insights into the value of finerenone for people with “high normal” albuminuria, he said.
The study led by Dr. Parveen used data routinely collected in Danish national records and focused on all Danish adults diagnosed with type 2 diabetes as of Jan. 1, 2015, who also had information in their records for a UACR and an estimated glomerular filtration rate (eGFR) within the preceding year.
The records showed that only 47% of these people had a UACR value during this time frame, and that 57% had a recent measure of their eGFR, despite prevailing recommendations for routine and regular measurements of these parameters for all people with type 2 diabetes.
Dr. Parveen hypothesized that UACR measurement may lag for several reasons, such as reliance by primary care physicians on urine dipstick assessments, which preclude calculation of a UACR, poor adherence to regular medical assessment by people in low socioeconomic groups, and medical examination done outside of morning time periods, which is the best time of day for assessing UACR.
More albuminuria measurement needed in primary care
“Measurement of albuminuria in people with type 2 diabetes is improving in Europe, but is not yet at the level that’s needed,” commented Dr. Ruilope. “We are pushing to have it done more often in primary care practices,” he said.
Among the 74,014 people with type 2 diabetes who had the measurement records that allowed for their inclusion in the study, 40% had albuminuria and 60% did not.
During 4 years of follow-up, the incidence of heart failure, MI, stroke, or all-cause death was 28.6% in those with albuminuria and 18.7% among those without albuminuria, reported Dr. Parveen.
The rates for each event type in those with albuminuria were 7.0% for heart failure, 4.4% for MI, 7.6% for stroke, and 16.6% for all-cause death (each patient could tally more than one type of event). Among those without albuminuria, the rates were 4.0%, 3.2%, 5.5%, and 9.3%, respectively.
The study received no commercial funding. Dr. Parveen and Dr. Ruilope had no disclosures.
BARCELONA – Fewer than half the adults in Denmark with type 2 diabetes in 2015 had a recent assessment for albuminuria, and those who underwent testing and had albuminuria had a greater than 50% increased rate of incident heart failure, MI, stroke, or all-cause death during 4-year follow-up, in a study of more than 74,000 Danish residents.
Even those in this study with type 2 diabetes but without albuminuria had a 19% rate of these adverse outcomes, highlighting the “substantial” cardiovascular disease risk faced by people with type 2 diabetes even without a clear indication of nephropathy, Saaima Parveen, MD, a cardiology researcher at Herlev and Gentofte University Hospital in Copenhagen, said at the annual congress of the European Society of Cardiology.
This high rate of heart failure, MI, stroke, or death even in the absence of what is conventionally defined as albuminuria – a urinary albumin-to-creatinine ratio (UACR) of at least 30 mg/g – suggests that this threshold for albuminuria may be too high, commented Luis M. Ruilope, MD, professor of public health and preventive medicine at Autonoma University, Madrid, who was not involved with the Danish study.
The study reported by Dr. Parveen “is very important because it shows that the risk of events is high not only in people with diabetes with albuminuria, but also in those without albuminuria,” Dr. Ruilope said in an interview.
The profile of albuminuria as a risk marker for people with type 2 diabetes spiked following the 2021 U.S. approval of finerenone (Kerendia) as an agent specifically targeted to adults with type 2 diabetes and albuminuria. (Finerenone gained marketing approval by in Europe in February 2022 under the same brand name.)
A lower threshold for albuminuria?
“Even patients with a UACR of 10-29 mg/g have risk and should be considered for finerenone treatment, said Dr. Ruilope. “People with type 2 diabetes with a UACR of 10-29 mg/g could explain” the high background risk shown by Dr. Parveen in her reported data. “In people with type 2 diabetes and a UACR of 10-29 mg/g we also see progression of kidney disease, but it’s slower” than in those who meet the current, standard threshold for albuminuria.
Dr. Ruilope was a coinvestigator for both of the finerenone pivotal trials, FIDELIO-DKD and FIGARO-DKD. Although the design of both these studies specified enrollment of people with type 2 diabetes and a UACR of at least 30 mg/g, a few hundred of the total combined enrollment of more than 13,000 patients had UACR values below this level, and analysis of this subgroup could provide some important insights into the value of finerenone for people with “high normal” albuminuria, he said.
The study led by Dr. Parveen used data routinely collected in Danish national records and focused on all Danish adults diagnosed with type 2 diabetes as of Jan. 1, 2015, who also had information in their records for a UACR and an estimated glomerular filtration rate (eGFR) within the preceding year.
The records showed that only 47% of these people had a UACR value during this time frame, and that 57% had a recent measure of their eGFR, despite prevailing recommendations for routine and regular measurements of these parameters for all people with type 2 diabetes.
Dr. Parveen hypothesized that UACR measurement may lag for several reasons, such as reliance by primary care physicians on urine dipstick assessments, which preclude calculation of a UACR, poor adherence to regular medical assessment by people in low socioeconomic groups, and medical examination done outside of morning time periods, which is the best time of day for assessing UACR.
More albuminuria measurement needed in primary care
“Measurement of albuminuria in people with type 2 diabetes is improving in Europe, but is not yet at the level that’s needed,” commented Dr. Ruilope. “We are pushing to have it done more often in primary care practices,” he said.
Among the 74,014 people with type 2 diabetes who had the measurement records that allowed for their inclusion in the study, 40% had albuminuria and 60% did not.
During 4 years of follow-up, the incidence of heart failure, MI, stroke, or all-cause death was 28.6% in those with albuminuria and 18.7% among those without albuminuria, reported Dr. Parveen.
The rates for each event type in those with albuminuria were 7.0% for heart failure, 4.4% for MI, 7.6% for stroke, and 16.6% for all-cause death (each patient could tally more than one type of event). Among those without albuminuria, the rates were 4.0%, 3.2%, 5.5%, and 9.3%, respectively.
The study received no commercial funding. Dr. Parveen and Dr. Ruilope had no disclosures.
AT ESC CONGRESS 2022











