User login
Common Herbicide a Player in Neurodegeneration?
new research showed.
Researchers found that glyphosate exposure even at regulated levels was associated with increased neuroinflammation and accelerated Alzheimer’s disease–like pathology in mice — an effect that persisted 6 months after a recovery period when exposure was stopped.
“More research is needed to understand the consequences of glyphosate exposure to the brain in humans and to understand the appropriate dose of exposure to limit detrimental outcomes,” said co–senior author Ramon Velazquez, PhD, with Arizona State University, Tempe.
The study was published online in The Journal of Neuroinflammation.
Persistent Accumulation Within the Brain
Glyphosate is the most heavily applied herbicide in the United States, with roughly 300 million pounds used annually in agricultural communities throughout the United States. It is also used for weed control in parks, residential areas, and personal gardens.
The Environmental Protection Agency (EPA) has determined that glyphosate poses no risks to human health when used as directed. But the World Health Organization’s International Agency for Research on Cancer disagrees, classifying the herbicide as “possibly carcinogenic to humans.”
In addition to the possible cancer risk, multiple reports have also suggested potential harmful effects of glyphosate exposure on the brain.
In earlier work, Velazquez and colleagues showed that glyphosate crosses the blood-brain barrier and infiltrates the brains of mice, contributing to neuroinflammation and other detrimental effects on brain function.
In their latest study, they examined the long-term effects of glyphosate exposure on neuroinflammation and Alzheimer’s disease–like pathology using a mouse model.
They dosed 4.5-month-old mice genetically predisposed to Alzheimer’s disease and non-transgenic control mice with either 0, 50, or 500 mg/kg of glyphosate daily for 13 weeks followed by a 6-month recovery period.
The high dose is similar to levels used in earlier research, and the low dose is close to the limit used to establish the current EPA acceptable dose in humans.
Glyphosate’s metabolite, aminomethylphosphonic acid, was detectable and persisted in mouse brain tissue even 6 months after exposure ceased, the researchers reported.
Additionally, there was a significant increase in soluble and insoluble fractions of amyloid-beta (Abeta), Abeta42 plaque load and plaque size, and phosphorylated tau at Threonine 181 and Serine 396 in hippocampus and cortex brain tissue from glyphosate-exposed mice, “highlighting an exacerbation of hallmark Alzheimer’s disease–like proteinopathies,” they noted.
Glyphosate exposure was also associated with significant elevations in both pro- and anti-inflammatory cytokines and chemokines in brain tissue of transgenic and normal mice and in peripheral blood plasma of transgenic mice.
Glyphosate-exposed transgenic mice also showed heightened anxiety-like behaviors and reduced survival.
“These findings highlight that many chemicals we regularly encounter, previously considered safe, may pose potential health risks,” co–senior author Patrick Pirrotte, PhD, with the Translational Genomics Research Institute, Phoenix, Arizona, said in a statement.
“However, further research is needed to fully assess the public health impact and identify safer alternatives,” Pirrotte added.
Funding for the study was provided by the National Institutes on Aging, National Cancer Institute and the Arizona State University (ASU) Biodesign Institute. The authors have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
new research showed.
Researchers found that glyphosate exposure even at regulated levels was associated with increased neuroinflammation and accelerated Alzheimer’s disease–like pathology in mice — an effect that persisted 6 months after a recovery period when exposure was stopped.
“More research is needed to understand the consequences of glyphosate exposure to the brain in humans and to understand the appropriate dose of exposure to limit detrimental outcomes,” said co–senior author Ramon Velazquez, PhD, with Arizona State University, Tempe.
The study was published online in The Journal of Neuroinflammation.
Persistent Accumulation Within the Brain
Glyphosate is the most heavily applied herbicide in the United States, with roughly 300 million pounds used annually in agricultural communities throughout the United States. It is also used for weed control in parks, residential areas, and personal gardens.
The Environmental Protection Agency (EPA) has determined that glyphosate poses no risks to human health when used as directed. But the World Health Organization’s International Agency for Research on Cancer disagrees, classifying the herbicide as “possibly carcinogenic to humans.”
In addition to the possible cancer risk, multiple reports have also suggested potential harmful effects of glyphosate exposure on the brain.
In earlier work, Velazquez and colleagues showed that glyphosate crosses the blood-brain barrier and infiltrates the brains of mice, contributing to neuroinflammation and other detrimental effects on brain function.
In their latest study, they examined the long-term effects of glyphosate exposure on neuroinflammation and Alzheimer’s disease–like pathology using a mouse model.
They dosed 4.5-month-old mice genetically predisposed to Alzheimer’s disease and non-transgenic control mice with either 0, 50, or 500 mg/kg of glyphosate daily for 13 weeks followed by a 6-month recovery period.
The high dose is similar to levels used in earlier research, and the low dose is close to the limit used to establish the current EPA acceptable dose in humans.
Glyphosate’s metabolite, aminomethylphosphonic acid, was detectable and persisted in mouse brain tissue even 6 months after exposure ceased, the researchers reported.
Additionally, there was a significant increase in soluble and insoluble fractions of amyloid-beta (Abeta), Abeta42 plaque load and plaque size, and phosphorylated tau at Threonine 181 and Serine 396 in hippocampus and cortex brain tissue from glyphosate-exposed mice, “highlighting an exacerbation of hallmark Alzheimer’s disease–like proteinopathies,” they noted.
Glyphosate exposure was also associated with significant elevations in both pro- and anti-inflammatory cytokines and chemokines in brain tissue of transgenic and normal mice and in peripheral blood plasma of transgenic mice.
Glyphosate-exposed transgenic mice also showed heightened anxiety-like behaviors and reduced survival.
“These findings highlight that many chemicals we regularly encounter, previously considered safe, may pose potential health risks,” co–senior author Patrick Pirrotte, PhD, with the Translational Genomics Research Institute, Phoenix, Arizona, said in a statement.
“However, further research is needed to fully assess the public health impact and identify safer alternatives,” Pirrotte added.
Funding for the study was provided by the National Institutes on Aging, National Cancer Institute and the Arizona State University (ASU) Biodesign Institute. The authors have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
new research showed.
Researchers found that glyphosate exposure even at regulated levels was associated with increased neuroinflammation and accelerated Alzheimer’s disease–like pathology in mice — an effect that persisted 6 months after a recovery period when exposure was stopped.
“More research is needed to understand the consequences of glyphosate exposure to the brain in humans and to understand the appropriate dose of exposure to limit detrimental outcomes,” said co–senior author Ramon Velazquez, PhD, with Arizona State University, Tempe.
The study was published online in The Journal of Neuroinflammation.
Persistent Accumulation Within the Brain
Glyphosate is the most heavily applied herbicide in the United States, with roughly 300 million pounds used annually in agricultural communities throughout the United States. It is also used for weed control in parks, residential areas, and personal gardens.
The Environmental Protection Agency (EPA) has determined that glyphosate poses no risks to human health when used as directed. But the World Health Organization’s International Agency for Research on Cancer disagrees, classifying the herbicide as “possibly carcinogenic to humans.”
In addition to the possible cancer risk, multiple reports have also suggested potential harmful effects of glyphosate exposure on the brain.
In earlier work, Velazquez and colleagues showed that glyphosate crosses the blood-brain barrier and infiltrates the brains of mice, contributing to neuroinflammation and other detrimental effects on brain function.
In their latest study, they examined the long-term effects of glyphosate exposure on neuroinflammation and Alzheimer’s disease–like pathology using a mouse model.
They dosed 4.5-month-old mice genetically predisposed to Alzheimer’s disease and non-transgenic control mice with either 0, 50, or 500 mg/kg of glyphosate daily for 13 weeks followed by a 6-month recovery period.
The high dose is similar to levels used in earlier research, and the low dose is close to the limit used to establish the current EPA acceptable dose in humans.
Glyphosate’s metabolite, aminomethylphosphonic acid, was detectable and persisted in mouse brain tissue even 6 months after exposure ceased, the researchers reported.
Additionally, there was a significant increase in soluble and insoluble fractions of amyloid-beta (Abeta), Abeta42 plaque load and plaque size, and phosphorylated tau at Threonine 181 and Serine 396 in hippocampus and cortex brain tissue from glyphosate-exposed mice, “highlighting an exacerbation of hallmark Alzheimer’s disease–like proteinopathies,” they noted.
Glyphosate exposure was also associated with significant elevations in both pro- and anti-inflammatory cytokines and chemokines in brain tissue of transgenic and normal mice and in peripheral blood plasma of transgenic mice.
Glyphosate-exposed transgenic mice also showed heightened anxiety-like behaviors and reduced survival.
“These findings highlight that many chemicals we regularly encounter, previously considered safe, may pose potential health risks,” co–senior author Patrick Pirrotte, PhD, with the Translational Genomics Research Institute, Phoenix, Arizona, said in a statement.
“However, further research is needed to fully assess the public health impact and identify safer alternatives,” Pirrotte added.
Funding for the study was provided by the National Institutes on Aging, National Cancer Institute and the Arizona State University (ASU) Biodesign Institute. The authors have declared no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF NEUROINFLAMMATION
BCG Vaccine May Protect Against Long COVID Symptoms
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A phase 3 clinical trial initiated in early 2020 investigated the effect of the BCG vaccine injected during active infection on COVID-19 progression in adults with mild or moderate COVID-19. The current study summarizes the 6- and 12-month follow-up data with a focus on long-COVID symptoms.
- Patients who tested positive for severe acute respiratory syndrome coronavirus 2 were randomly assigned to receive either 0.1 mL of intradermal BCG (n = 191) or 0.9% saline placebo (n = 202) within 14 days of symptom onset and were followed up at 7, 14, 21, and 45 days and at 6 and 12 months postinjection.
- Overall, 157 BCG (median age, 40 years; 54.1% women) and 142 placebo (median age, 41 years; 65.5% women) recipients completed the 6-month follow-up, and 97 BCG (median age, 37 years; 49.5% women) and 95 placebo (median age, 40 years; 67.4% women) recipients completed the 12-month follow-up.
- The researchers primarily assessed the effect of the BCG vaccine on the development of the symptoms of long COVID at 6 and 12 months.
TAKEAWAY:
- Hearing problems were less frequent among BCG recipients at 6 months compared with those who received placebo (odds ratio [OR], 0.26; 95% CI, 0.045-1.0; P = .044).
- At 12 months, participants who received the BCG vaccine exhibited fewer issues with sleeping (P = .027), concentration (P = .009), memory (P = .009), and vision (P = .022) along with a lower long-COVID score (one-sided Wilcoxon test, P = .002) than those who received placebo.
- At 6 months, BCG demonstrated a sex-specific paradoxical effect on hair loss, decreasing it in men (P = .031), while causing a slight, though statistically nonsignificant, increase in women.
- Male sex was the strongest predictive factor for long COVID, cognitive dysfunction, and cardiopulmonary scores at both follow-up assessments.
IN PRACTICE:
“[The study] findings suggest that BCG immunotherapy for an existing ailment may be superior to prophylaxis in healthy individuals,” the authors wrote.
SOURCE:
The study was led by Mehrsa Jalalizadeh and Keini Buosi, UroScience, State University of Campinas, Unicamp, São Paulo, Brazil. It was published online on November 19, 2024, in the Journal of Internal Medicine.
LIMITATIONS:
Previous mycobacterial exposure was not tested among the study participants. A notable loss to follow-up, particularly at 12 months, may have introduced bias into the results.
DISCLOSURES:
The study was supported by the Coordination for the Improvement of Higher Education Personnel, Federal Government of Brazil, the General Coordination of the National Immunization Program, Ministry of Health (Brazil), and the National Council for Scientific and Technological Development-Research Productivity. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
How Metals Affect the Brain
This transcript has been edited for clarity.
It has always amazed me that our bodies require these tiny amounts of incredibly rare substances to function. Sure, we need oxygen. We need water. But we also need molybdenum, which makes up just 1.2 parts per million of the Earth’s crust.
Without adequate molybdenum intake, we develop seizures, developmental delays, death. Fortunately, we need so little molybdenum that true molybdenum deficiency is incredibly rare — seen only in people on total parenteral nutrition without supplementation or those with certain rare genetic conditions. But still, molybdenum is necessary for life.
Many metals are. Figure 1 colors the essential minerals on the periodic table. You can see that to stay alive, we humans need not only things like sodium, but selenium, bromine, zinc, copper, and cobalt.

Some metals are very clearly not essential; we can all do without lead and mercury, and probably should.
But just because something is essential for life does not mean that more is better. The dose is the poison, as they say. And this week, we explore whether metals — even essential metals — might be adversely affecting our brains.
It’s not a stretch to think that metal intake could have weird effects on our nervous system. Lead exposure, primarily due to leaded gasoline, has been blamed for an average reduction of about 3 points in our national IQ, for example . But not all metals are created equal. Researchers set out to find out which might be more strongly associated with performance on cognitive tests and dementia, and reported their results in this study in JAMA Network Open.
To do this, they leveraged the MESA cohort study. This is a longitudinal study of a relatively diverse group of 6300 adults who were enrolled from 2000 to 2002 around the United States. At enrollment, they gave a urine sample and took a variety of cognitive tests. Important for this study was the digit symbol substitution test, where participants are provided a code and need to replace a list of numbers with symbols as per that code. Performance on this test worsens with age, depression, and cognitive impairment.
Participants were followed for more than a decade, and over that time, 559 (about 9%) were diagnosed with dementia.
Those baseline urine samples were assayed for a variety of metals — some essential, some very much not, as you can see in Figure 2.

Now, I have to put my kidney doctor hat on for a second and talk about urine measurement ... of anything. The problem with urine is that the concentration can change a lot — by more than 10-fold, in fact — based on how much water you drank recently. Researchers correct for this, and in the case of this study, they do what a lot of researchers do: divide the measured concentration by the urine creatinine level.

This introduces a bit of a problem. Take two people with exactly the same kidney function, who drank exactly the same water, whose urine is exactly the same concentration. The person with more muscle mass will have more creatinine in that urine sample, since creatinine is a byproduct of muscle metabolism. Because people with more muscle mass are generally healthier, when you divide your metal concentration by urine creatinine, you get a lower number, which might lead you to believe that lower levels of the metal in the urine are protective. But in fact, what you’re seeing is that higher levels of creatinine are protective. I see this issue all the time and it will always color results of studies like this.
Okay, I am doffing my kidney doctor hat now to show you the results.
The researchers first looked at the relationship between metal concentrations in the urine and performance on cognitive tests. The results were fairly equivocal, save for that digit substitution test which is shown in Figure 4.
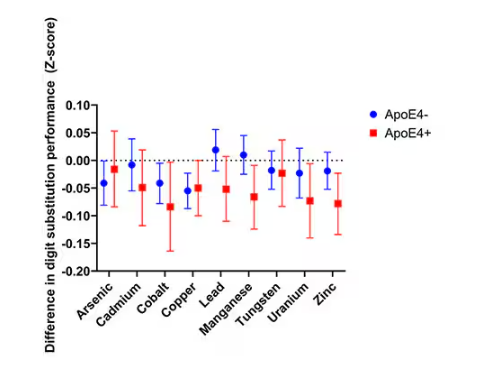
Even these results don’t ring major alarm bells for me. What you’re seeing here is the change in scores on the digit substitution test for each 25-percentile increase in urinary metal level — a pretty big change. And yet, you see really minor changes in the performance on the test. The digit substitution test is not an IQ test; but to give you a feeling for the magnitude of this change, if we looked at copper level, moving from the 25th to the 50th percentile would be associated with a loss of nine tenths of an IQ point.
You see two colors on the Figure 4 graph, by the way. That’s because the researchers stratified their findings based on whether the individual carried the ApoE4 gene allele, which is a risk factor for the development of dementia. There are reasons to believe that neurotoxic metals might be worse in this population, and I suppose you do see generally more adverse effects on scores in the red lines compared with the blue lines. But still, we’re not talking about a huge effect size here.
Let’s look at the relationship between these metals and the development of dementia itself, a clearly more important outcome than how well you can replace numeric digits with symbols. I’ll highlight a few of the results that are particularly telling.
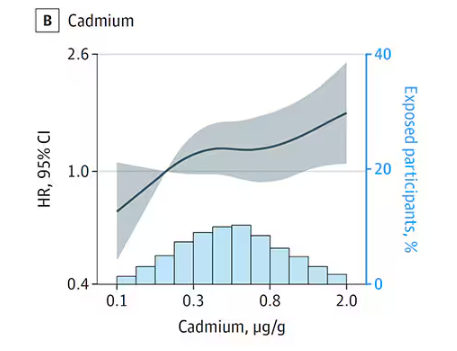
First, the nonessential mineral cadmium, which displays the type of relationship we would expect if the metal were neurotoxic: a clear, roughly linear increase in risk for dementia as urinary concentration increases.
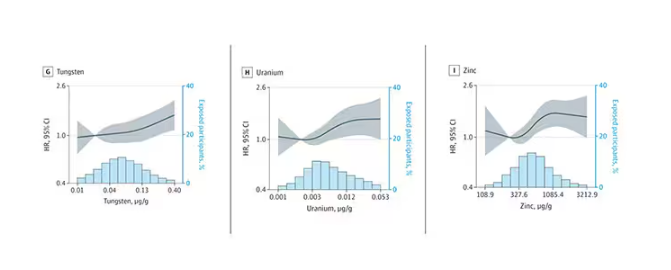
We see roughly similar patterns with the nonessential minerals tungsten and uranium, and the essential mineral zinc (beloved of respiratory-virus avoiders everywhere).
But it is very much not what we see for all metals. Strangest of all, look at lead, which shows basically no relationship with dementia.
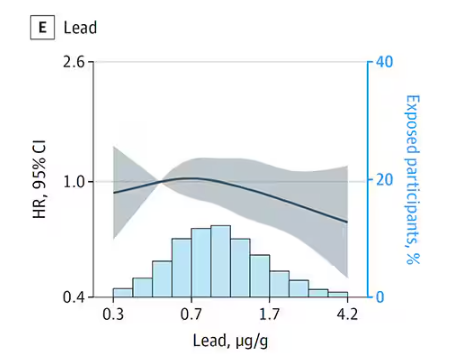
This concerns me a bit. Earlier, I discussed the issue of measuring stuff in urine and how standardizing levels to the urine creatinine level introduces a bias due to muscle mass. One way around this is to standardize urine levels to some other marker of urine dilution, like osmolality. But more fundamental than that, I like to see positive and negative controls in studies like this. For example, lead strikes me as a good positive control here. If the experimental framework were valid, I would think we’d see a relationship between lead level and dementia.
For a negative control? Well, something we are quite sure is not neurotoxic — something like sulfur, which is relatively ubiquitous, used in a variety of biological processes, and efficiently eliminated. We don’t have that in this study.
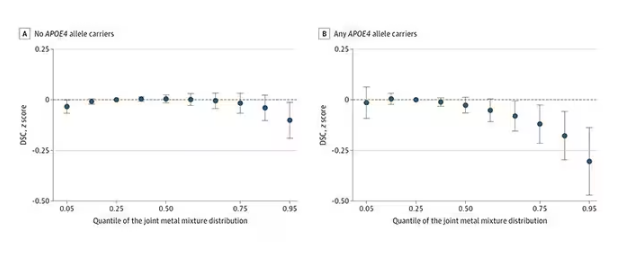
The authors close their case by creating a model that combines all the metal levels, asking the question of whether higher levels of metals in the urine in general worsen cognitive scores. And they find that the relationship exists, as you can see in Figure 8, both in carriers and noncarriers of ApoE4. But, to me, this is even more argument for the creatinine problem. If it’s not a specific metal but just the sort of general concentration of all metals, the risk for confounding by muscle mass is even higher.
So should we worry about ingesting metals? I suppose the answer is ... kind of.
I am sure we should be avoiding lead, despite the results of this study. It’s probably best to stay away from uranium too.
As for the essential metals, I’m sure there is some toxic dose; there’s a toxic dose for everything at some point. But I don’t see evidence in this study to make me worry that a significant chunk of the population is anywhere close to that.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It has always amazed me that our bodies require these tiny amounts of incredibly rare substances to function. Sure, we need oxygen. We need water. But we also need molybdenum, which makes up just 1.2 parts per million of the Earth’s crust.
Without adequate molybdenum intake, we develop seizures, developmental delays, death. Fortunately, we need so little molybdenum that true molybdenum deficiency is incredibly rare — seen only in people on total parenteral nutrition without supplementation or those with certain rare genetic conditions. But still, molybdenum is necessary for life.
Many metals are. Figure 1 colors the essential minerals on the periodic table. You can see that to stay alive, we humans need not only things like sodium, but selenium, bromine, zinc, copper, and cobalt.

Some metals are very clearly not essential; we can all do without lead and mercury, and probably should.
But just because something is essential for life does not mean that more is better. The dose is the poison, as they say. And this week, we explore whether metals — even essential metals — might be adversely affecting our brains.
It’s not a stretch to think that metal intake could have weird effects on our nervous system. Lead exposure, primarily due to leaded gasoline, has been blamed for an average reduction of about 3 points in our national IQ, for example . But not all metals are created equal. Researchers set out to find out which might be more strongly associated with performance on cognitive tests and dementia, and reported their results in this study in JAMA Network Open.
To do this, they leveraged the MESA cohort study. This is a longitudinal study of a relatively diverse group of 6300 adults who were enrolled from 2000 to 2002 around the United States. At enrollment, they gave a urine sample and took a variety of cognitive tests. Important for this study was the digit symbol substitution test, where participants are provided a code and need to replace a list of numbers with symbols as per that code. Performance on this test worsens with age, depression, and cognitive impairment.
Participants were followed for more than a decade, and over that time, 559 (about 9%) were diagnosed with dementia.
Those baseline urine samples were assayed for a variety of metals — some essential, some very much not, as you can see in Figure 2.

Now, I have to put my kidney doctor hat on for a second and talk about urine measurement ... of anything. The problem with urine is that the concentration can change a lot — by more than 10-fold, in fact — based on how much water you drank recently. Researchers correct for this, and in the case of this study, they do what a lot of researchers do: divide the measured concentration by the urine creatinine level.

This introduces a bit of a problem. Take two people with exactly the same kidney function, who drank exactly the same water, whose urine is exactly the same concentration. The person with more muscle mass will have more creatinine in that urine sample, since creatinine is a byproduct of muscle metabolism. Because people with more muscle mass are generally healthier, when you divide your metal concentration by urine creatinine, you get a lower number, which might lead you to believe that lower levels of the metal in the urine are protective. But in fact, what you’re seeing is that higher levels of creatinine are protective. I see this issue all the time and it will always color results of studies like this.
Okay, I am doffing my kidney doctor hat now to show you the results.
The researchers first looked at the relationship between metal concentrations in the urine and performance on cognitive tests. The results were fairly equivocal, save for that digit substitution test which is shown in Figure 4.

Even these results don’t ring major alarm bells for me. What you’re seeing here is the change in scores on the digit substitution test for each 25-percentile increase in urinary metal level — a pretty big change. And yet, you see really minor changes in the performance on the test. The digit substitution test is not an IQ test; but to give you a feeling for the magnitude of this change, if we looked at copper level, moving from the 25th to the 50th percentile would be associated with a loss of nine tenths of an IQ point.
You see two colors on the Figure 4 graph, by the way. That’s because the researchers stratified their findings based on whether the individual carried the ApoE4 gene allele, which is a risk factor for the development of dementia. There are reasons to believe that neurotoxic metals might be worse in this population, and I suppose you do see generally more adverse effects on scores in the red lines compared with the blue lines. But still, we’re not talking about a huge effect size here.
Let’s look at the relationship between these metals and the development of dementia itself, a clearly more important outcome than how well you can replace numeric digits with symbols. I’ll highlight a few of the results that are particularly telling.
First, the nonessential mineral cadmium, which displays the type of relationship we would expect if the metal were neurotoxic: a clear, roughly linear increase in risk for dementia as urinary concentration increases.

We see roughly similar patterns with the nonessential minerals tungsten and uranium, and the essential mineral zinc (beloved of respiratory-virus avoiders everywhere).

But it is very much not what we see for all metals. Strangest of all, look at lead, which shows basically no relationship with dementia.

This concerns me a bit. Earlier, I discussed the issue of measuring stuff in urine and how standardizing levels to the urine creatinine level introduces a bias due to muscle mass. One way around this is to standardize urine levels to some other marker of urine dilution, like osmolality. But more fundamental than that, I like to see positive and negative controls in studies like this. For example, lead strikes me as a good positive control here. If the experimental framework were valid, I would think we’d see a relationship between lead level and dementia.
For a negative control? Well, something we are quite sure is not neurotoxic — something like sulfur, which is relatively ubiquitous, used in a variety of biological processes, and efficiently eliminated. We don’t have that in this study.
The authors close their case by creating a model that combines all the metal levels, asking the question of whether higher levels of metals in the urine in general worsen cognitive scores. And they find that the relationship exists, as you can see in Figure 8, both in carriers and noncarriers of ApoE4. But, to me, this is even more argument for the creatinine problem. If it’s not a specific metal but just the sort of general concentration of all metals, the risk for confounding by muscle mass is even higher.

So should we worry about ingesting metals? I suppose the answer is ... kind of.
I am sure we should be avoiding lead, despite the results of this study. It’s probably best to stay away from uranium too.
As for the essential metals, I’m sure there is some toxic dose; there’s a toxic dose for everything at some point. But I don’t see evidence in this study to make me worry that a significant chunk of the population is anywhere close to that.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
It has always amazed me that our bodies require these tiny amounts of incredibly rare substances to function. Sure, we need oxygen. We need water. But we also need molybdenum, which makes up just 1.2 parts per million of the Earth’s crust.
Without adequate molybdenum intake, we develop seizures, developmental delays, death. Fortunately, we need so little molybdenum that true molybdenum deficiency is incredibly rare — seen only in people on total parenteral nutrition without supplementation or those with certain rare genetic conditions. But still, molybdenum is necessary for life.
Many metals are. Figure 1 colors the essential minerals on the periodic table. You can see that to stay alive, we humans need not only things like sodium, but selenium, bromine, zinc, copper, and cobalt.

Some metals are very clearly not essential; we can all do without lead and mercury, and probably should.
But just because something is essential for life does not mean that more is better. The dose is the poison, as they say. And this week, we explore whether metals — even essential metals — might be adversely affecting our brains.
It’s not a stretch to think that metal intake could have weird effects on our nervous system. Lead exposure, primarily due to leaded gasoline, has been blamed for an average reduction of about 3 points in our national IQ, for example . But not all metals are created equal. Researchers set out to find out which might be more strongly associated with performance on cognitive tests and dementia, and reported their results in this study in JAMA Network Open.
To do this, they leveraged the MESA cohort study. This is a longitudinal study of a relatively diverse group of 6300 adults who were enrolled from 2000 to 2002 around the United States. At enrollment, they gave a urine sample and took a variety of cognitive tests. Important for this study was the digit symbol substitution test, where participants are provided a code and need to replace a list of numbers with symbols as per that code. Performance on this test worsens with age, depression, and cognitive impairment.
Participants were followed for more than a decade, and over that time, 559 (about 9%) were diagnosed with dementia.
Those baseline urine samples were assayed for a variety of metals — some essential, some very much not, as you can see in Figure 2.

Now, I have to put my kidney doctor hat on for a second and talk about urine measurement ... of anything. The problem with urine is that the concentration can change a lot — by more than 10-fold, in fact — based on how much water you drank recently. Researchers correct for this, and in the case of this study, they do what a lot of researchers do: divide the measured concentration by the urine creatinine level.

This introduces a bit of a problem. Take two people with exactly the same kidney function, who drank exactly the same water, whose urine is exactly the same concentration. The person with more muscle mass will have more creatinine in that urine sample, since creatinine is a byproduct of muscle metabolism. Because people with more muscle mass are generally healthier, when you divide your metal concentration by urine creatinine, you get a lower number, which might lead you to believe that lower levels of the metal in the urine are protective. But in fact, what you’re seeing is that higher levels of creatinine are protective. I see this issue all the time and it will always color results of studies like this.
Okay, I am doffing my kidney doctor hat now to show you the results.
The researchers first looked at the relationship between metal concentrations in the urine and performance on cognitive tests. The results were fairly equivocal, save for that digit substitution test which is shown in Figure 4.

Even these results don’t ring major alarm bells for me. What you’re seeing here is the change in scores on the digit substitution test for each 25-percentile increase in urinary metal level — a pretty big change. And yet, you see really minor changes in the performance on the test. The digit substitution test is not an IQ test; but to give you a feeling for the magnitude of this change, if we looked at copper level, moving from the 25th to the 50th percentile would be associated with a loss of nine tenths of an IQ point.
You see two colors on the Figure 4 graph, by the way. That’s because the researchers stratified their findings based on whether the individual carried the ApoE4 gene allele, which is a risk factor for the development of dementia. There are reasons to believe that neurotoxic metals might be worse in this population, and I suppose you do see generally more adverse effects on scores in the red lines compared with the blue lines. But still, we’re not talking about a huge effect size here.
Let’s look at the relationship between these metals and the development of dementia itself, a clearly more important outcome than how well you can replace numeric digits with symbols. I’ll highlight a few of the results that are particularly telling.
First, the nonessential mineral cadmium, which displays the type of relationship we would expect if the metal were neurotoxic: a clear, roughly linear increase in risk for dementia as urinary concentration increases.

We see roughly similar patterns with the nonessential minerals tungsten and uranium, and the essential mineral zinc (beloved of respiratory-virus avoiders everywhere).

But it is very much not what we see for all metals. Strangest of all, look at lead, which shows basically no relationship with dementia.

This concerns me a bit. Earlier, I discussed the issue of measuring stuff in urine and how standardizing levels to the urine creatinine level introduces a bias due to muscle mass. One way around this is to standardize urine levels to some other marker of urine dilution, like osmolality. But more fundamental than that, I like to see positive and negative controls in studies like this. For example, lead strikes me as a good positive control here. If the experimental framework were valid, I would think we’d see a relationship between lead level and dementia.
For a negative control? Well, something we are quite sure is not neurotoxic — something like sulfur, which is relatively ubiquitous, used in a variety of biological processes, and efficiently eliminated. We don’t have that in this study.
The authors close their case by creating a model that combines all the metal levels, asking the question of whether higher levels of metals in the urine in general worsen cognitive scores. And they find that the relationship exists, as you can see in Figure 8, both in carriers and noncarriers of ApoE4. But, to me, this is even more argument for the creatinine problem. If it’s not a specific metal but just the sort of general concentration of all metals, the risk for confounding by muscle mass is even higher.

So should we worry about ingesting metals? I suppose the answer is ... kind of.
I am sure we should be avoiding lead, despite the results of this study. It’s probably best to stay away from uranium too.
As for the essential metals, I’m sure there is some toxic dose; there’s a toxic dose for everything at some point. But I don’t see evidence in this study to make me worry that a significant chunk of the population is anywhere close to that.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Microplastics Have Been Found in the Human Brain. Now What?
In a recent case series study that examined olfactory bulb tissue from deceased individuals, 8 of the 15 decedent brains showed the presence of microplastics, most commonly polypropylene, a plastic typically used in food packaging and water bottles.
Measuring less than 5 mm in size, microplastics are formed over time as plastic materials break down but don’t biodegrade. Exposure to these substances can come through food, air, and skin absorption.
While scientists are learning more about how these substances are absorbed by the body, questions remain about how much exposure is safe, what effect — if any — microplastics could have on brain function, and what clinicians should tell their patients.
What Are the Major Health Concerns?
The Plastic Health Council estimates that more than 500 million metric tons of plastic are produced worldwide each year. In addition, it reports that plastic products can contain more than 16,000 chemicals, about a quarter of which have been found to be hazardous to human health and the environment. Microplastics and nanoplastics can enter the body through the air, in food, or absorption through the skin.
A study published in March showed that patients with carotid plaques and the presence of microplastics and nanoplastics were at an increased risk for death or major cardiovascular events.
Other studies have shown a link between these substances and placental inflammation and preterm births, reduced male fertility, and endocrine disruption — as well as accelerated spread of cancer cells in the gut.
There is also evidence suggesting that microplastics may facilitate the development of antibiotic resistance in bacteria and could contribute to the rise in food allergies.
And now, Thais Mauad, MD, PhD, and colleagues have found the substances in the brain.
How Is the Brain Affected?
The investigators examined olfactory bulb tissues from 15 deceased Sao Paulo, Brazil, residents ranging in age from 33 to 100 years who underwent routine coroner autopsies. All but three of the participants were men.
Exclusion criteria included having undergone previous neurosurgical interventions. The tissues were analyzed using micro–Fourier transform infrared spectroscopy (µFTIR).
In addition, the researchers practiced a “plastic-free approach” in their analysis, which included using filters and covering glassware and samples with aluminum foil.
Study findings showed microplastics in 8 of the 15 participants — including in the centenarian. In total, there were 16 synthetic polymer particles and fibers detected, with up to four microplastics detected per olfactory bulb. Polypropylene was the most common polymer found (44%), followed by polyamide, nylon, and polyethylene vinyl acetate. These substances are commonly used in a wide range of products, including food packaging, textiles, kitchen utensils, medical devices, and adhesives.
The microplastic particles ranged in length from 5.5 to 26 microns (one millionth of a meter), with a width that ranged from 3 to 25 microns. The mean fiber length and width was 21 and 4 microns, respectively. For comparison, the diameter of one human hair averages about 70 microns, according to the US Food and Drug Administration (FDA).
“To our knowledge, this is the first study in which the presence of microplastics in the human brain was identified and characterized using µFTIR,” the researchers wrote.
How Do Microplastics Reach the Brain?
Although the possibility of microplastics crossing the blood-brain barrier has been questioned, senior investigator Mauad, associate professor in the Department of Pathology, the University of Sao Paulo in Brazil, noted that the olfactory pathway could offer an entry route through inhalation of the particles.
This means that “breathing within indoor environments could be a major source of plastic pollution in the brain,” she said in a press release.
“With much smaller nanoplastics entering the body with greater ease, the total level of plastic particles may be much higher. What is worrying is the capacity of such particles to be internalized by cells and alter how our bodies function,” she added.
Mauad said that although questions remain regarding the health implications of their findings, some animal studies have shown that the presence of microplastics in the brain is linked to neurotoxic effects, including oxidative stress.
In addition, exposure to particulate matter has been linked previously to such neurologic conditions as dementia and neurodegenerative conditions such as Parkinson’s disease “seem to have a connection with nasal abnormalities as initial symptoms,” the investigators noted.
While the olfactory pathway appears to be a likely route of exposure the researchers noted that other potential entry routes, including through blood circulation, may also be involved.
The research suggests that inhaling microplastics while indoors may be unavoidable, Mauad said, making it unlikely individuals can eliminate exposure to these substances.
“Everything that surrounds us is plastic. So we can’t really get rid of it,” she said.
Are Microplastics Regulated?
The most effective solution would be stricter regulations, Mauad said.
“The industry has chosen to sell many things in plastic, and I think this has to change. We need more policies to decrease plastic production — especially single-use plastic,” she said.
Federal, state, and local regulations for microplastics are “virtually nonexistent,” reported the Interstate Technology and Regulatory Council (ITRC), a state-led coalition that produces documents and trainings related to regulatory issues.
In 2021, the ITRC sent a survey to all US states asking about microplastics regulations. Of the 26 states that responded, only 4 said they had conducted sampling for microplastics. None of the responders indicated they had established any criteria or standards for microplastics, although eight states indicated they had plans to pursue them in the future.
Although federal regulations include the Microbead-Free Waters Act of 2015 and the Save Our Seas Act 2.0, the rules don’t directly pertain to microplastics.
There are also no regulations currently in place regarding microplastics or nanoplastics in food. A report issued in July by the FDA claimed that “the overall scientific evidence does not demonstrate that levels of microplastics or nanoplastics found in foods pose a risk to human health.”
International efforts to regulate microplastics are much further along. First created in 2022, the treaty would forge an international, legally binding agreement.
While it is a step in the right direction, the Plastic Health Council has cautioned about “the omission of measures in draft provisions that fully address the impact of plastic pollution on human health.” The treaty should reduce plastic production, eliminate single-use plastic items, and call for testing of all chemicals in plastics, the council argues.
The final round of negotiations for the UN Global Plastic Treaty is set for completion before the end of the year.
What Should Clinicians Know?
Much remains unknown about the potential health effects of microplastic exposure. So how can clinicians respond to questions from concerned patients?
“We don’t yet have enough evidence about the plastic particle itself, like those highlighted in the current study — and even more so when it comes to nanoplastics, which are a thousand times smaller,” said Phoebe Stapleton, PhD, associated professor in the Department of Pharmacology and Toxicology at the Ernest Mario School of Pharmacy at Rutgers University, Piscataway, New Jersey.
“But we do have a lot of evidence about the chemicals that are used to make plastics, and we’ve already seen regulation there from the EPA. That’s one conversation that clinicians could have with patients: about those chemicals,” she added.
Stapleton recommended clinicians stay current on the latest research and be ready to respond should a patient raise the issue. She also noted the importance of exercising caution when interpreting these new findings.
While the study is important — especially because it highlights inhalation as a viable route of entry — exposure through the olfactory area is still just a theory and hasn’t yet been fully proven.
In addition, Stapleton wonders whether there are tissues where these substances are not found. A discovery like that “would be really exciting because that means that that tissue has mechanisms protecting it, and maybe, we could learn more about how to keep microplastics out,” she said.
She would also like to see more studies on specific adverse health effects from microplastics in the body.
Mauad agreed.
“That’s the next set of questions: What are the toxicities or lack thereof in those tissues? That will give us more information as it pertains to human health. It doesn’t feel good to know they’re in our tissues, but we still don’t have a real understanding of what they’re doing when they’re there,” she said.
The current study was funded by the Alexander von Humboldt Foundation and by grants from the Brazilian Research Council and the Soa State Research Agency. It was also funded by the Plastic Soup Foundation — which, together with A Plastic Planet, forms the Plastic Health Council. The investigators and Stapleton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a recent case series study that examined olfactory bulb tissue from deceased individuals, 8 of the 15 decedent brains showed the presence of microplastics, most commonly polypropylene, a plastic typically used in food packaging and water bottles.
Measuring less than 5 mm in size, microplastics are formed over time as plastic materials break down but don’t biodegrade. Exposure to these substances can come through food, air, and skin absorption.
While scientists are learning more about how these substances are absorbed by the body, questions remain about how much exposure is safe, what effect — if any — microplastics could have on brain function, and what clinicians should tell their patients.
What Are the Major Health Concerns?
The Plastic Health Council estimates that more than 500 million metric tons of plastic are produced worldwide each year. In addition, it reports that plastic products can contain more than 16,000 chemicals, about a quarter of which have been found to be hazardous to human health and the environment. Microplastics and nanoplastics can enter the body through the air, in food, or absorption through the skin.
A study published in March showed that patients with carotid plaques and the presence of microplastics and nanoplastics were at an increased risk for death or major cardiovascular events.
Other studies have shown a link between these substances and placental inflammation and preterm births, reduced male fertility, and endocrine disruption — as well as accelerated spread of cancer cells in the gut.
There is also evidence suggesting that microplastics may facilitate the development of antibiotic resistance in bacteria and could contribute to the rise in food allergies.
And now, Thais Mauad, MD, PhD, and colleagues have found the substances in the brain.
How Is the Brain Affected?
The investigators examined olfactory bulb tissues from 15 deceased Sao Paulo, Brazil, residents ranging in age from 33 to 100 years who underwent routine coroner autopsies. All but three of the participants were men.
Exclusion criteria included having undergone previous neurosurgical interventions. The tissues were analyzed using micro–Fourier transform infrared spectroscopy (µFTIR).
In addition, the researchers practiced a “plastic-free approach” in their analysis, which included using filters and covering glassware and samples with aluminum foil.
Study findings showed microplastics in 8 of the 15 participants — including in the centenarian. In total, there were 16 synthetic polymer particles and fibers detected, with up to four microplastics detected per olfactory bulb. Polypropylene was the most common polymer found (44%), followed by polyamide, nylon, and polyethylene vinyl acetate. These substances are commonly used in a wide range of products, including food packaging, textiles, kitchen utensils, medical devices, and adhesives.
The microplastic particles ranged in length from 5.5 to 26 microns (one millionth of a meter), with a width that ranged from 3 to 25 microns. The mean fiber length and width was 21 and 4 microns, respectively. For comparison, the diameter of one human hair averages about 70 microns, according to the US Food and Drug Administration (FDA).
“To our knowledge, this is the first study in which the presence of microplastics in the human brain was identified and characterized using µFTIR,” the researchers wrote.
How Do Microplastics Reach the Brain?
Although the possibility of microplastics crossing the blood-brain barrier has been questioned, senior investigator Mauad, associate professor in the Department of Pathology, the University of Sao Paulo in Brazil, noted that the olfactory pathway could offer an entry route through inhalation of the particles.
This means that “breathing within indoor environments could be a major source of plastic pollution in the brain,” she said in a press release.
“With much smaller nanoplastics entering the body with greater ease, the total level of plastic particles may be much higher. What is worrying is the capacity of such particles to be internalized by cells and alter how our bodies function,” she added.
Mauad said that although questions remain regarding the health implications of their findings, some animal studies have shown that the presence of microplastics in the brain is linked to neurotoxic effects, including oxidative stress.
In addition, exposure to particulate matter has been linked previously to such neurologic conditions as dementia and neurodegenerative conditions such as Parkinson’s disease “seem to have a connection with nasal abnormalities as initial symptoms,” the investigators noted.
While the olfactory pathway appears to be a likely route of exposure the researchers noted that other potential entry routes, including through blood circulation, may also be involved.
The research suggests that inhaling microplastics while indoors may be unavoidable, Mauad said, making it unlikely individuals can eliminate exposure to these substances.
“Everything that surrounds us is plastic. So we can’t really get rid of it,” she said.
Are Microplastics Regulated?
The most effective solution would be stricter regulations, Mauad said.
“The industry has chosen to sell many things in plastic, and I think this has to change. We need more policies to decrease plastic production — especially single-use plastic,” she said.
Federal, state, and local regulations for microplastics are “virtually nonexistent,” reported the Interstate Technology and Regulatory Council (ITRC), a state-led coalition that produces documents and trainings related to regulatory issues.
In 2021, the ITRC sent a survey to all US states asking about microplastics regulations. Of the 26 states that responded, only 4 said they had conducted sampling for microplastics. None of the responders indicated they had established any criteria or standards for microplastics, although eight states indicated they had plans to pursue them in the future.
Although federal regulations include the Microbead-Free Waters Act of 2015 and the Save Our Seas Act 2.0, the rules don’t directly pertain to microplastics.
There are also no regulations currently in place regarding microplastics or nanoplastics in food. A report issued in July by the FDA claimed that “the overall scientific evidence does not demonstrate that levels of microplastics or nanoplastics found in foods pose a risk to human health.”
International efforts to regulate microplastics are much further along. First created in 2022, the treaty would forge an international, legally binding agreement.
While it is a step in the right direction, the Plastic Health Council has cautioned about “the omission of measures in draft provisions that fully address the impact of plastic pollution on human health.” The treaty should reduce plastic production, eliminate single-use plastic items, and call for testing of all chemicals in plastics, the council argues.
The final round of negotiations for the UN Global Plastic Treaty is set for completion before the end of the year.
What Should Clinicians Know?
Much remains unknown about the potential health effects of microplastic exposure. So how can clinicians respond to questions from concerned patients?
“We don’t yet have enough evidence about the plastic particle itself, like those highlighted in the current study — and even more so when it comes to nanoplastics, which are a thousand times smaller,” said Phoebe Stapleton, PhD, associated professor in the Department of Pharmacology and Toxicology at the Ernest Mario School of Pharmacy at Rutgers University, Piscataway, New Jersey.
“But we do have a lot of evidence about the chemicals that are used to make plastics, and we’ve already seen regulation there from the EPA. That’s one conversation that clinicians could have with patients: about those chemicals,” she added.
Stapleton recommended clinicians stay current on the latest research and be ready to respond should a patient raise the issue. She also noted the importance of exercising caution when interpreting these new findings.
While the study is important — especially because it highlights inhalation as a viable route of entry — exposure through the olfactory area is still just a theory and hasn’t yet been fully proven.
In addition, Stapleton wonders whether there are tissues where these substances are not found. A discovery like that “would be really exciting because that means that that tissue has mechanisms protecting it, and maybe, we could learn more about how to keep microplastics out,” she said.
She would also like to see more studies on specific adverse health effects from microplastics in the body.
Mauad agreed.
“That’s the next set of questions: What are the toxicities or lack thereof in those tissues? That will give us more information as it pertains to human health. It doesn’t feel good to know they’re in our tissues, but we still don’t have a real understanding of what they’re doing when they’re there,” she said.
The current study was funded by the Alexander von Humboldt Foundation and by grants from the Brazilian Research Council and the Soa State Research Agency. It was also funded by the Plastic Soup Foundation — which, together with A Plastic Planet, forms the Plastic Health Council. The investigators and Stapleton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a recent case series study that examined olfactory bulb tissue from deceased individuals, 8 of the 15 decedent brains showed the presence of microplastics, most commonly polypropylene, a plastic typically used in food packaging and water bottles.
Measuring less than 5 mm in size, microplastics are formed over time as plastic materials break down but don’t biodegrade. Exposure to these substances can come through food, air, and skin absorption.
While scientists are learning more about how these substances are absorbed by the body, questions remain about how much exposure is safe, what effect — if any — microplastics could have on brain function, and what clinicians should tell their patients.
What Are the Major Health Concerns?
The Plastic Health Council estimates that more than 500 million metric tons of plastic are produced worldwide each year. In addition, it reports that plastic products can contain more than 16,000 chemicals, about a quarter of which have been found to be hazardous to human health and the environment. Microplastics and nanoplastics can enter the body through the air, in food, or absorption through the skin.
A study published in March showed that patients with carotid plaques and the presence of microplastics and nanoplastics were at an increased risk for death or major cardiovascular events.
Other studies have shown a link between these substances and placental inflammation and preterm births, reduced male fertility, and endocrine disruption — as well as accelerated spread of cancer cells in the gut.
There is also evidence suggesting that microplastics may facilitate the development of antibiotic resistance in bacteria and could contribute to the rise in food allergies.
And now, Thais Mauad, MD, PhD, and colleagues have found the substances in the brain.
How Is the Brain Affected?
The investigators examined olfactory bulb tissues from 15 deceased Sao Paulo, Brazil, residents ranging in age from 33 to 100 years who underwent routine coroner autopsies. All but three of the participants were men.
Exclusion criteria included having undergone previous neurosurgical interventions. The tissues were analyzed using micro–Fourier transform infrared spectroscopy (µFTIR).
In addition, the researchers practiced a “plastic-free approach” in their analysis, which included using filters and covering glassware and samples with aluminum foil.
Study findings showed microplastics in 8 of the 15 participants — including in the centenarian. In total, there were 16 synthetic polymer particles and fibers detected, with up to four microplastics detected per olfactory bulb. Polypropylene was the most common polymer found (44%), followed by polyamide, nylon, and polyethylene vinyl acetate. These substances are commonly used in a wide range of products, including food packaging, textiles, kitchen utensils, medical devices, and adhesives.
The microplastic particles ranged in length from 5.5 to 26 microns (one millionth of a meter), with a width that ranged from 3 to 25 microns. The mean fiber length and width was 21 and 4 microns, respectively. For comparison, the diameter of one human hair averages about 70 microns, according to the US Food and Drug Administration (FDA).
“To our knowledge, this is the first study in which the presence of microplastics in the human brain was identified and characterized using µFTIR,” the researchers wrote.
How Do Microplastics Reach the Brain?
Although the possibility of microplastics crossing the blood-brain barrier has been questioned, senior investigator Mauad, associate professor in the Department of Pathology, the University of Sao Paulo in Brazil, noted that the olfactory pathway could offer an entry route through inhalation of the particles.
This means that “breathing within indoor environments could be a major source of plastic pollution in the brain,” she said in a press release.
“With much smaller nanoplastics entering the body with greater ease, the total level of plastic particles may be much higher. What is worrying is the capacity of such particles to be internalized by cells and alter how our bodies function,” she added.
Mauad said that although questions remain regarding the health implications of their findings, some animal studies have shown that the presence of microplastics in the brain is linked to neurotoxic effects, including oxidative stress.
In addition, exposure to particulate matter has been linked previously to such neurologic conditions as dementia and neurodegenerative conditions such as Parkinson’s disease “seem to have a connection with nasal abnormalities as initial symptoms,” the investigators noted.
While the olfactory pathway appears to be a likely route of exposure the researchers noted that other potential entry routes, including through blood circulation, may also be involved.
The research suggests that inhaling microplastics while indoors may be unavoidable, Mauad said, making it unlikely individuals can eliminate exposure to these substances.
“Everything that surrounds us is plastic. So we can’t really get rid of it,” she said.
Are Microplastics Regulated?
The most effective solution would be stricter regulations, Mauad said.
“The industry has chosen to sell many things in plastic, and I think this has to change. We need more policies to decrease plastic production — especially single-use plastic,” she said.
Federal, state, and local regulations for microplastics are “virtually nonexistent,” reported the Interstate Technology and Regulatory Council (ITRC), a state-led coalition that produces documents and trainings related to regulatory issues.
In 2021, the ITRC sent a survey to all US states asking about microplastics regulations. Of the 26 states that responded, only 4 said they had conducted sampling for microplastics. None of the responders indicated they had established any criteria or standards for microplastics, although eight states indicated they had plans to pursue them in the future.
Although federal regulations include the Microbead-Free Waters Act of 2015 and the Save Our Seas Act 2.0, the rules don’t directly pertain to microplastics.
There are also no regulations currently in place regarding microplastics or nanoplastics in food. A report issued in July by the FDA claimed that “the overall scientific evidence does not demonstrate that levels of microplastics or nanoplastics found in foods pose a risk to human health.”
International efforts to regulate microplastics are much further along. First created in 2022, the treaty would forge an international, legally binding agreement.
While it is a step in the right direction, the Plastic Health Council has cautioned about “the omission of measures in draft provisions that fully address the impact of plastic pollution on human health.” The treaty should reduce plastic production, eliminate single-use plastic items, and call for testing of all chemicals in plastics, the council argues.
The final round of negotiations for the UN Global Plastic Treaty is set for completion before the end of the year.
What Should Clinicians Know?
Much remains unknown about the potential health effects of microplastic exposure. So how can clinicians respond to questions from concerned patients?
“We don’t yet have enough evidence about the plastic particle itself, like those highlighted in the current study — and even more so when it comes to nanoplastics, which are a thousand times smaller,” said Phoebe Stapleton, PhD, associated professor in the Department of Pharmacology and Toxicology at the Ernest Mario School of Pharmacy at Rutgers University, Piscataway, New Jersey.
“But we do have a lot of evidence about the chemicals that are used to make plastics, and we’ve already seen regulation there from the EPA. That’s one conversation that clinicians could have with patients: about those chemicals,” she added.
Stapleton recommended clinicians stay current on the latest research and be ready to respond should a patient raise the issue. She also noted the importance of exercising caution when interpreting these new findings.
While the study is important — especially because it highlights inhalation as a viable route of entry — exposure through the olfactory area is still just a theory and hasn’t yet been fully proven.
In addition, Stapleton wonders whether there are tissues where these substances are not found. A discovery like that “would be really exciting because that means that that tissue has mechanisms protecting it, and maybe, we could learn more about how to keep microplastics out,” she said.
She would also like to see more studies on specific adverse health effects from microplastics in the body.
Mauad agreed.
“That’s the next set of questions: What are the toxicities or lack thereof in those tissues? That will give us more information as it pertains to human health. It doesn’t feel good to know they’re in our tissues, but we still don’t have a real understanding of what they’re doing when they’re there,” she said.
The current study was funded by the Alexander von Humboldt Foundation and by grants from the Brazilian Research Council and the Soa State Research Agency. It was also funded by the Plastic Soup Foundation — which, together with A Plastic Planet, forms the Plastic Health Council. The investigators and Stapleton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Six Updates on Stroke Management
This video transcript has been edited for clarity.
Dear colleagues, I am Christoph Diener, from the Faculty of Medicine at the University Duisburg-Essen in Germany. In this video, I would like to cover six publications on stroke, which were published this fall.
The Best Thrombolytic?
Let me start with systemic thrombolysis. We now have two thrombolytic agents available. One is the well-known alteplase, and newly approved for the treatment of stroke is tenecteplase. The ATTEST-2 study in the United Kingdom, published in The Lancet Neurology, compared tenecteplase 0.25 mg/kg body weight as a bolus with alteplase 0.9 mg/kg body weight as an infusion over 60 minutes in the 4.5-hour time window in 1777 patients with ischemic stroke.
There was no significant difference between the two thrombolytics for the primary endpoint of modified Rankin Scale score after 90 days. There was also no difference with respect to mortality, intracranial bleeding, or extracranial bleeding.
We finally have 11 randomized controlled trials that compared tenecteplase and alteplase in acute ischemic stroke. A meta-analysis of these randomized trials was published in Neurology. The analysis included 3700 patients treated with tenecteplase and 3700 patients treated with alteplase. For the primary endpoint, excellent functional outcome defined as modified Rankin Scale score 0-1 after 90 days, there was a significant benefit for tenecteplase (relative risk, 1.05), but the absolute difference was very small, at 3%. There was no difference in mortality or bleeding complications.
In conclusion, I think both substances are great. They are effective. Tenecteplase is most probably the drug which should be used in people who have to transfer from a primary stroke center to a dedicated stroke center that provides thrombectomy. Otherwise, I think it’s a choice of the physician as to which thrombolytic agent to use.
Mobile Stroke Units
A highly debated topic is mobile stroke units. These stroke units have a CT scanner and laboratory on board, and this makes it possible to perform thrombolysis on the way to the hospital. A retrospective, observational study collected data between 2018 and 2023, and included 19,400 patients with acute stroke, of whom 1237, or 6.4%, were treated in a mobile stroke unit. This study was published in JAMA Neurology.
The modified Rankin Scale score at the time of discharge was better in patients treated with a mobile stroke unit, but the absolute benefit was only 0.03 points on the modified Rankin Scale. The question is whether this is cost-effective, and can we really do this at times when there is a traumatic shortage of physicians and nursing staff in the hospital?
DOAC Reversal Agents
Oral anticoagulation, as you know, is usually considered a contraindication for systemic thrombolysis. Idarucizumab, a monoclonal antibody, was developed to reverse the biological activity of dabigatran and then allow systemic thrombolysis.
A recent publication in Neurology analyzed 13 cohort studies with 553 stroke patients on dabigatran who received idarucizumab prior to systemic thrombolysis, and the rate of intracranial hemorrhage was 4%. This means it’s obviously possible to perform thrombolysis when the activity of dabigatran is neutralized by idarucizumab.
Unfortunately, until today, we have no data on whether this can also be done with andexanet alfa in people who are treated with a factor Xa inhibitor like, for example, apixaban, rivaroxaban, or edoxaban.
Anticoagulation in ESUS
My next topic is ESUS, or embolic stroke of undetermined source. We have four large randomized trials and three smaller trials that compared antiplatelet therapy with DOACs in patients with ESUS. A group in Neurology published a meta-analysis of seven randomized controlled studies with, altogether, 14,800 patients with ESUS.
The comparison between antiplatelet therapy and anticoagulants showed no difference for recurrent ischemic stroke, and also not for major subgroups. This means that people with ESUS should receive antiplatelet therapy, most probably aspirin.
Anticoagulation Post–Ischemic Stroke With AF
My final topic is the optimal time to start anticoagulation in people with atrial fibrillation who suffer an ischemic stroke. The OPTIMAS study, published in The Lancet, randomized 3650 patients who were anticoagulated with DOACs early (which means less than 4 days) or delayed (between 7 and 14 days). There was no difference in the primary endpoint, which was recurrent ischemic stroke, intracranial hemorrhage, or systemic embolism at 90 days.
The conclusion is that, in most cases, we can probably initiate anticoagulation in people with ischemic stroke and atrial fibrillation within the first 4 days.
Dear colleagues, this is an exciting time for the stroke field. I presented six new studies that have impact, I think, on the management of patients with ischemic stroke.
Dr. Diener is a professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen in Germany. He reported conflicts of interest with Abbott, AbbVie, Boehringer Ingelheim, Lundbeck, Novartis, Orion Pharma, Teva, WebMD, and The German Research Council. He also serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Dear colleagues, I am Christoph Diener, from the Faculty of Medicine at the University Duisburg-Essen in Germany. In this video, I would like to cover six publications on stroke, which were published this fall.
The Best Thrombolytic?
Let me start with systemic thrombolysis. We now have two thrombolytic agents available. One is the well-known alteplase, and newly approved for the treatment of stroke is tenecteplase. The ATTEST-2 study in the United Kingdom, published in The Lancet Neurology, compared tenecteplase 0.25 mg/kg body weight as a bolus with alteplase 0.9 mg/kg body weight as an infusion over 60 minutes in the 4.5-hour time window in 1777 patients with ischemic stroke.
There was no significant difference between the two thrombolytics for the primary endpoint of modified Rankin Scale score after 90 days. There was also no difference with respect to mortality, intracranial bleeding, or extracranial bleeding.
We finally have 11 randomized controlled trials that compared tenecteplase and alteplase in acute ischemic stroke. A meta-analysis of these randomized trials was published in Neurology. The analysis included 3700 patients treated with tenecteplase and 3700 patients treated with alteplase. For the primary endpoint, excellent functional outcome defined as modified Rankin Scale score 0-1 after 90 days, there was a significant benefit for tenecteplase (relative risk, 1.05), but the absolute difference was very small, at 3%. There was no difference in mortality or bleeding complications.
In conclusion, I think both substances are great. They are effective. Tenecteplase is most probably the drug which should be used in people who have to transfer from a primary stroke center to a dedicated stroke center that provides thrombectomy. Otherwise, I think it’s a choice of the physician as to which thrombolytic agent to use.
Mobile Stroke Units
A highly debated topic is mobile stroke units. These stroke units have a CT scanner and laboratory on board, and this makes it possible to perform thrombolysis on the way to the hospital. A retrospective, observational study collected data between 2018 and 2023, and included 19,400 patients with acute stroke, of whom 1237, or 6.4%, were treated in a mobile stroke unit. This study was published in JAMA Neurology.
The modified Rankin Scale score at the time of discharge was better in patients treated with a mobile stroke unit, but the absolute benefit was only 0.03 points on the modified Rankin Scale. The question is whether this is cost-effective, and can we really do this at times when there is a traumatic shortage of physicians and nursing staff in the hospital?
DOAC Reversal Agents
Oral anticoagulation, as you know, is usually considered a contraindication for systemic thrombolysis. Idarucizumab, a monoclonal antibody, was developed to reverse the biological activity of dabigatran and then allow systemic thrombolysis.
A recent publication in Neurology analyzed 13 cohort studies with 553 stroke patients on dabigatran who received idarucizumab prior to systemic thrombolysis, and the rate of intracranial hemorrhage was 4%. This means it’s obviously possible to perform thrombolysis when the activity of dabigatran is neutralized by idarucizumab.
Unfortunately, until today, we have no data on whether this can also be done with andexanet alfa in people who are treated with a factor Xa inhibitor like, for example, apixaban, rivaroxaban, or edoxaban.
Anticoagulation in ESUS
My next topic is ESUS, or embolic stroke of undetermined source. We have four large randomized trials and three smaller trials that compared antiplatelet therapy with DOACs in patients with ESUS. A group in Neurology published a meta-analysis of seven randomized controlled studies with, altogether, 14,800 patients with ESUS.
The comparison between antiplatelet therapy and anticoagulants showed no difference for recurrent ischemic stroke, and also not for major subgroups. This means that people with ESUS should receive antiplatelet therapy, most probably aspirin.
Anticoagulation Post–Ischemic Stroke With AF
My final topic is the optimal time to start anticoagulation in people with atrial fibrillation who suffer an ischemic stroke. The OPTIMAS study, published in The Lancet, randomized 3650 patients who were anticoagulated with DOACs early (which means less than 4 days) or delayed (between 7 and 14 days). There was no difference in the primary endpoint, which was recurrent ischemic stroke, intracranial hemorrhage, or systemic embolism at 90 days.
The conclusion is that, in most cases, we can probably initiate anticoagulation in people with ischemic stroke and atrial fibrillation within the first 4 days.
Dear colleagues, this is an exciting time for the stroke field. I presented six new studies that have impact, I think, on the management of patients with ischemic stroke.
Dr. Diener is a professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen in Germany. He reported conflicts of interest with Abbott, AbbVie, Boehringer Ingelheim, Lundbeck, Novartis, Orion Pharma, Teva, WebMD, and The German Research Council. He also serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Dear colleagues, I am Christoph Diener, from the Faculty of Medicine at the University Duisburg-Essen in Germany. In this video, I would like to cover six publications on stroke, which were published this fall.
The Best Thrombolytic?
Let me start with systemic thrombolysis. We now have two thrombolytic agents available. One is the well-known alteplase, and newly approved for the treatment of stroke is tenecteplase. The ATTEST-2 study in the United Kingdom, published in The Lancet Neurology, compared tenecteplase 0.25 mg/kg body weight as a bolus with alteplase 0.9 mg/kg body weight as an infusion over 60 minutes in the 4.5-hour time window in 1777 patients with ischemic stroke.
There was no significant difference between the two thrombolytics for the primary endpoint of modified Rankin Scale score after 90 days. There was also no difference with respect to mortality, intracranial bleeding, or extracranial bleeding.
We finally have 11 randomized controlled trials that compared tenecteplase and alteplase in acute ischemic stroke. A meta-analysis of these randomized trials was published in Neurology. The analysis included 3700 patients treated with tenecteplase and 3700 patients treated with alteplase. For the primary endpoint, excellent functional outcome defined as modified Rankin Scale score 0-1 after 90 days, there was a significant benefit for tenecteplase (relative risk, 1.05), but the absolute difference was very small, at 3%. There was no difference in mortality or bleeding complications.
In conclusion, I think both substances are great. They are effective. Tenecteplase is most probably the drug which should be used in people who have to transfer from a primary stroke center to a dedicated stroke center that provides thrombectomy. Otherwise, I think it’s a choice of the physician as to which thrombolytic agent to use.
Mobile Stroke Units
A highly debated topic is mobile stroke units. These stroke units have a CT scanner and laboratory on board, and this makes it possible to perform thrombolysis on the way to the hospital. A retrospective, observational study collected data between 2018 and 2023, and included 19,400 patients with acute stroke, of whom 1237, or 6.4%, were treated in a mobile stroke unit. This study was published in JAMA Neurology.
The modified Rankin Scale score at the time of discharge was better in patients treated with a mobile stroke unit, but the absolute benefit was only 0.03 points on the modified Rankin Scale. The question is whether this is cost-effective, and can we really do this at times when there is a traumatic shortage of physicians and nursing staff in the hospital?
DOAC Reversal Agents
Oral anticoagulation, as you know, is usually considered a contraindication for systemic thrombolysis. Idarucizumab, a monoclonal antibody, was developed to reverse the biological activity of dabigatran and then allow systemic thrombolysis.
A recent publication in Neurology analyzed 13 cohort studies with 553 stroke patients on dabigatran who received idarucizumab prior to systemic thrombolysis, and the rate of intracranial hemorrhage was 4%. This means it’s obviously possible to perform thrombolysis when the activity of dabigatran is neutralized by idarucizumab.
Unfortunately, until today, we have no data on whether this can also be done with andexanet alfa in people who are treated with a factor Xa inhibitor like, for example, apixaban, rivaroxaban, or edoxaban.
Anticoagulation in ESUS
My next topic is ESUS, or embolic stroke of undetermined source. We have four large randomized trials and three smaller trials that compared antiplatelet therapy with DOACs in patients with ESUS. A group in Neurology published a meta-analysis of seven randomized controlled studies with, altogether, 14,800 patients with ESUS.
The comparison between antiplatelet therapy and anticoagulants showed no difference for recurrent ischemic stroke, and also not for major subgroups. This means that people with ESUS should receive antiplatelet therapy, most probably aspirin.
Anticoagulation Post–Ischemic Stroke With AF
My final topic is the optimal time to start anticoagulation in people with atrial fibrillation who suffer an ischemic stroke. The OPTIMAS study, published in The Lancet, randomized 3650 patients who were anticoagulated with DOACs early (which means less than 4 days) or delayed (between 7 and 14 days). There was no difference in the primary endpoint, which was recurrent ischemic stroke, intracranial hemorrhage, or systemic embolism at 90 days.
The conclusion is that, in most cases, we can probably initiate anticoagulation in people with ischemic stroke and atrial fibrillation within the first 4 days.
Dear colleagues, this is an exciting time for the stroke field. I presented six new studies that have impact, I think, on the management of patients with ischemic stroke.
Dr. Diener is a professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen in Germany. He reported conflicts of interest with Abbott, AbbVie, Boehringer Ingelheim, Lundbeck, Novartis, Orion Pharma, Teva, WebMD, and The German Research Council. He also serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs.
A version of this article first appeared on Medscape.com.
Three Vascular Risk Factors May Up Severe Stroke Risk
TOPLINE:
, a global study shows.
METHODOLOGY:
- The INTERSTROKE case-control study included nearly 27,000 participants, half of whom had a first acute stroke (ischemic or hemorrhagic) and the other half acting as age- and sex-matched controls.
- Participants (mean age, 62 years; 40% women) were recruited across 142 centers in 32 countries between 2007 and 2015. Baseline demographics and lifestyle risk factors for stroke were gathered using standardized questionnaires
- Modified Rankin Scale (mRS) scores measured within 72 hours of hospital admission were used to classify stroke severity (0-3, nonsevere stroke; 4-6, severe stroke).
TAKEAWAY:
- Among the participants with acute stroke, 64% had nonsevere stroke and 36% had severe stroke, based on the mRS.
- Hypertension, atrial fibrillation, and smoking showed a significantly stronger association with severe stroke than with nonsevere stroke (odds ratios [ORs], 3.21 vs 2.87, 4.70 vs 3.61, and 1.87 vs 1.65, respectively; all P < .001).
- A high waist-to-hip ratio showed a stronger association with nonsevere stroke than with severe stroke (OR, 1.37 vs 1.11, respectively; P < .001).
- Diabetes, poor diet, physical inactivity, and stress were linked to increased odds of both severe and nonsevere stroke, whereas alcohol consumption and high apolipoprotein B levels were linked to higher odds of only nonsevere stroke. No significant differences in odds were observed between stroke severities in matched individuals.
IN PRACTICE:
“Our findings emphasize the importance of controlling high blood pressure, which is the most important modifiable risk factor for stroke globally,” lead author Catriona Reddin, MB BCh, BAO, MSc, School of Medicine, University of Galway, in Ireland, said in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
The study limitations included potential unmeasured confounders; reliance on the mRS score, which may have underestimated stroke severity; and challenges with recruiting patients with severe stroke in a case-control study. Smoking-related comorbidities and regional or sex-related variations in alcohol intake may also have influenced the results.
DISCLOSURES:
The study was funded by various organizations, including health research councils and foundations from Canada, Sweden, and Scotland, and pharmaceutical companies such as AstraZeneca, Boehringer Ingelheim, Pfizer, and MSD. One investigator reported receiving funding from the Irish Clinical Academic Training Programme, the Wellcome Trust and the Health Research Board, the Health Service Executive, National Doctors Training and Planning, and the Health and Social Care, Research and Development Division in Northern Ireland. No other conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
, a global study shows.
METHODOLOGY:
- The INTERSTROKE case-control study included nearly 27,000 participants, half of whom had a first acute stroke (ischemic or hemorrhagic) and the other half acting as age- and sex-matched controls.
- Participants (mean age, 62 years; 40% women) were recruited across 142 centers in 32 countries between 2007 and 2015. Baseline demographics and lifestyle risk factors for stroke were gathered using standardized questionnaires
- Modified Rankin Scale (mRS) scores measured within 72 hours of hospital admission were used to classify stroke severity (0-3, nonsevere stroke; 4-6, severe stroke).
TAKEAWAY:
- Among the participants with acute stroke, 64% had nonsevere stroke and 36% had severe stroke, based on the mRS.
- Hypertension, atrial fibrillation, and smoking showed a significantly stronger association with severe stroke than with nonsevere stroke (odds ratios [ORs], 3.21 vs 2.87, 4.70 vs 3.61, and 1.87 vs 1.65, respectively; all P < .001).
- A high waist-to-hip ratio showed a stronger association with nonsevere stroke than with severe stroke (OR, 1.37 vs 1.11, respectively; P < .001).
- Diabetes, poor diet, physical inactivity, and stress were linked to increased odds of both severe and nonsevere stroke, whereas alcohol consumption and high apolipoprotein B levels were linked to higher odds of only nonsevere stroke. No significant differences in odds were observed between stroke severities in matched individuals.
IN PRACTICE:
“Our findings emphasize the importance of controlling high blood pressure, which is the most important modifiable risk factor for stroke globally,” lead author Catriona Reddin, MB BCh, BAO, MSc, School of Medicine, University of Galway, in Ireland, said in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
The study limitations included potential unmeasured confounders; reliance on the mRS score, which may have underestimated stroke severity; and challenges with recruiting patients with severe stroke in a case-control study. Smoking-related comorbidities and regional or sex-related variations in alcohol intake may also have influenced the results.
DISCLOSURES:
The study was funded by various organizations, including health research councils and foundations from Canada, Sweden, and Scotland, and pharmaceutical companies such as AstraZeneca, Boehringer Ingelheim, Pfizer, and MSD. One investigator reported receiving funding from the Irish Clinical Academic Training Programme, the Wellcome Trust and the Health Research Board, the Health Service Executive, National Doctors Training and Planning, and the Health and Social Care, Research and Development Division in Northern Ireland. No other conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
, a global study shows.
METHODOLOGY:
- The INTERSTROKE case-control study included nearly 27,000 participants, half of whom had a first acute stroke (ischemic or hemorrhagic) and the other half acting as age- and sex-matched controls.
- Participants (mean age, 62 years; 40% women) were recruited across 142 centers in 32 countries between 2007 and 2015. Baseline demographics and lifestyle risk factors for stroke were gathered using standardized questionnaires
- Modified Rankin Scale (mRS) scores measured within 72 hours of hospital admission were used to classify stroke severity (0-3, nonsevere stroke; 4-6, severe stroke).
TAKEAWAY:
- Among the participants with acute stroke, 64% had nonsevere stroke and 36% had severe stroke, based on the mRS.
- Hypertension, atrial fibrillation, and smoking showed a significantly stronger association with severe stroke than with nonsevere stroke (odds ratios [ORs], 3.21 vs 2.87, 4.70 vs 3.61, and 1.87 vs 1.65, respectively; all P < .001).
- A high waist-to-hip ratio showed a stronger association with nonsevere stroke than with severe stroke (OR, 1.37 vs 1.11, respectively; P < .001).
- Diabetes, poor diet, physical inactivity, and stress were linked to increased odds of both severe and nonsevere stroke, whereas alcohol consumption and high apolipoprotein B levels were linked to higher odds of only nonsevere stroke. No significant differences in odds were observed between stroke severities in matched individuals.
IN PRACTICE:
“Our findings emphasize the importance of controlling high blood pressure, which is the most important modifiable risk factor for stroke globally,” lead author Catriona Reddin, MB BCh, BAO, MSc, School of Medicine, University of Galway, in Ireland, said in a press release.
SOURCE:
The study was published online in Neurology.
LIMITATIONS:
The study limitations included potential unmeasured confounders; reliance on the mRS score, which may have underestimated stroke severity; and challenges with recruiting patients with severe stroke in a case-control study. Smoking-related comorbidities and regional or sex-related variations in alcohol intake may also have influenced the results.
DISCLOSURES:
The study was funded by various organizations, including health research councils and foundations from Canada, Sweden, and Scotland, and pharmaceutical companies such as AstraZeneca, Boehringer Ingelheim, Pfizer, and MSD. One investigator reported receiving funding from the Irish Clinical Academic Training Programme, the Wellcome Trust and the Health Research Board, the Health Service Executive, National Doctors Training and Planning, and the Health and Social Care, Research and Development Division in Northern Ireland. No other conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Food as Medicine: Diet’s Role in Parkinson’s Disease
For 15 years, John Duda, MD, national director of the VA Parkinson’s Disease Research, Education and Clinical Centers, has urged his patients to “keep waiting” for effective treatments to manage both motor and nonmotor symptoms of Parkinson’s disease.
However, Duda, who also serves as director of the Brain Wellness Clinic at the Corporal Michael J. Crescenz VA Medical Center in Philadelphia, Pennsylvania, recognized the persistent lack of effective drugs to address these symptoms. This prompted him to consider what other evidence-based strategies he could use to support his patients.
“I recognized that nutritional approaches within a broader program that includes medication review, stress management, social connections, adequate sleep, and physical exercise could make a real difference,” he said.
Observational studies have shown an inverse association between dietary patterns and Parkinson’s disease risk, age of onset, symptom severity, and mortality rates — particularly with the Mediterranean diet (MeDi) and the MIND diet, which combines elements of MeDi and the Dietary Approaches to Stop Hypertension (DASH) diet. Although randomized controlled trials are still limited, the epidemiologic evidence supporting dietary interventions is “compelling,” said Duda.
For example, a cross-sectional study comparing 167 participants with Parkinson’s disease vs 119 controls showed that later age of Parkinson’s disease onset correlated with adherence to the MIND diet in women, with a difference of up to 17.4 years (P < .001) between low and high dietary tertiles.
The MeDi was correlated with later onset in men, with differences of up to 8.4 years (P = .002). As previously reported, a healthy diet emphasizing vegetables, fruits, nuts, and grains was inversely associated with prodromal features of Parkinson’s disease, including constipation, excessive daytime sleepiness, and depression. In addition, lower rates of Parkinson’s disease have been shown in populations following vegetarian and vegan dietary patterns.
Does Parkinson’s disease Start in the Gut?
Parkinson’s disease is characterized by decreased short-chain fatty acid–producing bacteria and increased pro-inflammatory species linked to intestinal inflammation and alpha-synuclein aggregation. “There are reasons to believe that a-synuclein accumulation may start in the gut,” Duda noted.
Numerous studies implicate gut microbiome dysbiosis as a pathogenic mechanism in Parkinson’s disease, with gastrointestinal symptoms often predating motor symptoms. Dysbiosis might result in a pro-inflammatory state potentially linked to the recurrent gastrointestinal symptoms. Fecal microbiota transplant may restore a healthier gut environment and beneficially affect Parkinson’s disease symptoms, he said.
Some of the benefits conferred by the MeDi and other healthy diets may be mediated by improving the gut microbiome. Duda cited a study that showed that a 14-day ovo-lacto vegetarian diet intervention and a daily fecal enema for 8 days improved not only the microbiome but also Movement Disorder Society Unified Parkinson’s Disease Rating Scale—part III scores.
Duda also reviewed the role of dietary interventions in addressing common Parkinson’s disease symptoms, such as orthostatic hypotension. He recommended that Parkinson’s disease patients with this condition should avoid eating large meals, increase dietary salt intake, increase fluid intake, and decrease alcohol intake.
Malnutrition affects close to 25% of those with Parkinson’s disease, which is partially attributable to diminished olfaction. Because the experience of taste is largely driven by a sense of smell, patients may be less interested in eating. Duda recommended increasing herbs, spices, and other flavors in food. High caloric–density foods, including nuts, nut butters, and seeds, can boost weight, he said. However, he added, any patient with significant weight loss should consult a nutritionist.
Constipation is one of the most debilitating symptoms of Parkinson’s disease, affecting up to 66% of patients. Duda advised increasing fluid intake, exercise, and dietary fiber and use of stool softeners and laxatives. The MeDi may reduce symptoms of constipation and have a beneficial effect on gut microbiota.
Coffee may be helpful for sleepiness in Parkinson’s disease and may also confer neuroprotective, motor, and cognitive benefits. As an adjuvant treatment, caffeine may alter levodopa pharmacokinetics, reduce dyskinesia, improve gait in patients with freezing and may even reduce the risk of developing Parkinson’s disease, with a maximum benefit reached at approximately three cups of coffee daily.
Problematic Foods
There is also a growing body of evidence regarding the deleterious effects of ultraprocessed foods (UPFs), Duda said. He noted that a recent systematic review and meta-analysis of 28 studies showed that higher UPF intake was significantly associated with an enhanced risk for Parkinson’s disease (relative risk, 1.56; 95% CI, 1.21-2.02). As previously reported, UPFs have been tied to a host of adverse neurologic outcomes, including cognitive decline and stroke.
Although protein is a necessary nutrient, incorporating it into the diet of Parkinson’s disease patients taking levodopa is complicated. Levodopa, a large neutral amino acid (LNAA), competes with other LNAAs for transport to the brain from the small intestine, Duda explained.
“Some people notice that carbidopa-levodopa doesn’t work as well if taken with a high-protein meal.” He recommended taking carbidopa-levodopa 30 minutes before or 60 minutes after meals.
Rebecca Gilbert, MD, PhD, chief mission officer of the American Parkinson’s Disease Association, said that patients with Parkinson’s disease might want to avoid eating protein during the day, concentrating instead on carbohydrates and vegetables and saving the protein for the evening, which is closer to bedtime. Some evidence also supports the use of protein redistribution diets to enhance the clinical response to levodopa and reduce motor fluctuations.
What About Supplements?
It’s “hard to prove that one specific supplement can be protective against Parkinson’s disease because diet consists of many different components and the whole diet may be worth more than the sum of its parts,” Gilbert said. The evidence for individual supplements “isn’t robust enough to say they prevent or treat Parkinson’s disease.”
Research on the role of specific nutrients in Parkinson’s disease is conflicting, with no clear evidence supporting or refuting their benefits. For example, a study that followed participants for about 30 years showed no link between reduced Parkinson’s disease risk and vitamin B or folate intake.
On the other hand, there is research suggesting that certain vitamins may help reduce Parkinson’s disease risk, although these nutrients do not operate in isolation. For instance, one recent study showed a connection between vitamins C and E and reduced Parkinson’s disease risk, but factors such as body mass index and coffee consumption appeared to influence the strength of this association.
Consuming polyunsaturated fatty acids along with reducing saturated fatty acid intake has been tied to a reduced risk for Parkinson’s disease.
Additionally, certain foods may offer protective effects, including green and black tea, with consumption of three or more cups per day associated with a delay in motor symptom onset by 7.7 years. Foods high in nicotine content, such as those from the Solanaceae family — including peppers, tomatoes, tomato juice, and potatoes — have also been linked to potential protective benefits.
Diets rich in antioxidants, including carotenoids, lutein, and vitamins E and C, have been robustly linked to a reduced risk for parkinsonism and progression of parkinsonian symptoms in older adults.
Increasing the intake of dietary flavonoids, particularly tea, berry fruits, apples, red wine, and oranges or orange juice, can reduce Parkinson’s disease risk. One study showed that male participants in the highest quintile of total flavonoid consumption had a 40% lower Parkinson’s disease risk compared with those in the lowest quintile. Another study showed that flavonoid-rich foods were also associated with a lower risk for death in patients with Parkinson’s disease.
Food as Medicine
Although recent research shows that the drug development pipeline for Parkinson’s disease is robust, with a wide variety of approaches being developed and evaluated in phase 1 and 2, investigators note that only a limited number of disease-modifying treatments are transitioning to phase 3.
Duda noted that phytochemicals incorporated into the diet might target some of the same mechanisms that are targets of these drugs in development.
“Flavonoids have been shown to stabilize alpha-synuclein in vitro,” he said. “Caffeine, curcumin, resveratrol, and eliminating meat and dairy inhibit mTOR [mammalian target of rapamycin], and mTOR inhibition results in increased autophagy that may help clear alpha-synuclein. Genestein, an isoflavone in soybeans, protects dopaminergic neurons by inhibiting microglia activation. Flavonoids inhibit inflammation by inhibiting release of NO [nitric oxide] and pro-inflammatory cytokines,” he noted.
Ongoing studies of dietary interventions for Parkinson’s disease are exploring various areas, including the potential role of the ketogenic diet in protecting the gut microbiome, optimizing protein intake for muscle preservation and sleep, the effects of psyllium and wheat bran on weight and constipation, and the impact of a gluten-free diet.
Practical Tips for Healthy Eating
Gilbert emphasized that there are no medications or interventions currently available that can delay a Parkinson’s disease diagnosis by up to 17 years, as some dietary patterns have been shown to do, and she noted that it’s not possible to replicate the MeDi diet in a pill. However, she recommended a practical approach to eating that includes a diet low in ultraprocessed foods and high in beneficial nutrients. She encouraged people to shop for “real food” and enjoy a variety of colorful fruits and vegetables.
Duda acknowledged that motivating patients to follow a healthy diet can be difficult. As a result, the focus often shifts to making small adjustments and modifications. For example, he suggested that instead of pairing meat with French fries, people could opt for vegetables or add greens to their meals. Similarly, instead of having eggs and bacon for breakfast, they might choose oatmeal.
Preparing whole-food, plant-based meals may take more time than patients are accustomed to, so Duda suggests that, if possible, patients involve loved ones in both the meal preparation and the meal itself. He explained that a healthy meal can become an opportunity for bonding and that the key is educating them about new meal-related concepts.
Duda reported no relevant financial relationships with the pharmaceutical or food industries. He has received compensation from the Physicians Committee for Responsible Medicine for his lecture delivered at the conference and research grant support from the VA, the National Institutes of Health, the Michael J. Fox Foundation, and the Department of Defense unrelated to this topic. Gilbert reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
For 15 years, John Duda, MD, national director of the VA Parkinson’s Disease Research, Education and Clinical Centers, has urged his patients to “keep waiting” for effective treatments to manage both motor and nonmotor symptoms of Parkinson’s disease.
However, Duda, who also serves as director of the Brain Wellness Clinic at the Corporal Michael J. Crescenz VA Medical Center in Philadelphia, Pennsylvania, recognized the persistent lack of effective drugs to address these symptoms. This prompted him to consider what other evidence-based strategies he could use to support his patients.
“I recognized that nutritional approaches within a broader program that includes medication review, stress management, social connections, adequate sleep, and physical exercise could make a real difference,” he said.
Observational studies have shown an inverse association between dietary patterns and Parkinson’s disease risk, age of onset, symptom severity, and mortality rates — particularly with the Mediterranean diet (MeDi) and the MIND diet, which combines elements of MeDi and the Dietary Approaches to Stop Hypertension (DASH) diet. Although randomized controlled trials are still limited, the epidemiologic evidence supporting dietary interventions is “compelling,” said Duda.
For example, a cross-sectional study comparing 167 participants with Parkinson’s disease vs 119 controls showed that later age of Parkinson’s disease onset correlated with adherence to the MIND diet in women, with a difference of up to 17.4 years (P < .001) between low and high dietary tertiles.
The MeDi was correlated with later onset in men, with differences of up to 8.4 years (P = .002). As previously reported, a healthy diet emphasizing vegetables, fruits, nuts, and grains was inversely associated with prodromal features of Parkinson’s disease, including constipation, excessive daytime sleepiness, and depression. In addition, lower rates of Parkinson’s disease have been shown in populations following vegetarian and vegan dietary patterns.
Does Parkinson’s disease Start in the Gut?
Parkinson’s disease is characterized by decreased short-chain fatty acid–producing bacteria and increased pro-inflammatory species linked to intestinal inflammation and alpha-synuclein aggregation. “There are reasons to believe that a-synuclein accumulation may start in the gut,” Duda noted.
Numerous studies implicate gut microbiome dysbiosis as a pathogenic mechanism in Parkinson’s disease, with gastrointestinal symptoms often predating motor symptoms. Dysbiosis might result in a pro-inflammatory state potentially linked to the recurrent gastrointestinal symptoms. Fecal microbiota transplant may restore a healthier gut environment and beneficially affect Parkinson’s disease symptoms, he said.
Some of the benefits conferred by the MeDi and other healthy diets may be mediated by improving the gut microbiome. Duda cited a study that showed that a 14-day ovo-lacto vegetarian diet intervention and a daily fecal enema for 8 days improved not only the microbiome but also Movement Disorder Society Unified Parkinson’s Disease Rating Scale—part III scores.
Duda also reviewed the role of dietary interventions in addressing common Parkinson’s disease symptoms, such as orthostatic hypotension. He recommended that Parkinson’s disease patients with this condition should avoid eating large meals, increase dietary salt intake, increase fluid intake, and decrease alcohol intake.
Malnutrition affects close to 25% of those with Parkinson’s disease, which is partially attributable to diminished olfaction. Because the experience of taste is largely driven by a sense of smell, patients may be less interested in eating. Duda recommended increasing herbs, spices, and other flavors in food. High caloric–density foods, including nuts, nut butters, and seeds, can boost weight, he said. However, he added, any patient with significant weight loss should consult a nutritionist.
Constipation is one of the most debilitating symptoms of Parkinson’s disease, affecting up to 66% of patients. Duda advised increasing fluid intake, exercise, and dietary fiber and use of stool softeners and laxatives. The MeDi may reduce symptoms of constipation and have a beneficial effect on gut microbiota.
Coffee may be helpful for sleepiness in Parkinson’s disease and may also confer neuroprotective, motor, and cognitive benefits. As an adjuvant treatment, caffeine may alter levodopa pharmacokinetics, reduce dyskinesia, improve gait in patients with freezing and may even reduce the risk of developing Parkinson’s disease, with a maximum benefit reached at approximately three cups of coffee daily.
Problematic Foods
There is also a growing body of evidence regarding the deleterious effects of ultraprocessed foods (UPFs), Duda said. He noted that a recent systematic review and meta-analysis of 28 studies showed that higher UPF intake was significantly associated with an enhanced risk for Parkinson’s disease (relative risk, 1.56; 95% CI, 1.21-2.02). As previously reported, UPFs have been tied to a host of adverse neurologic outcomes, including cognitive decline and stroke.
Although protein is a necessary nutrient, incorporating it into the diet of Parkinson’s disease patients taking levodopa is complicated. Levodopa, a large neutral amino acid (LNAA), competes with other LNAAs for transport to the brain from the small intestine, Duda explained.
“Some people notice that carbidopa-levodopa doesn’t work as well if taken with a high-protein meal.” He recommended taking carbidopa-levodopa 30 minutes before or 60 minutes after meals.
Rebecca Gilbert, MD, PhD, chief mission officer of the American Parkinson’s Disease Association, said that patients with Parkinson’s disease might want to avoid eating protein during the day, concentrating instead on carbohydrates and vegetables and saving the protein for the evening, which is closer to bedtime. Some evidence also supports the use of protein redistribution diets to enhance the clinical response to levodopa and reduce motor fluctuations.
What About Supplements?
It’s “hard to prove that one specific supplement can be protective against Parkinson’s disease because diet consists of many different components and the whole diet may be worth more than the sum of its parts,” Gilbert said. The evidence for individual supplements “isn’t robust enough to say they prevent or treat Parkinson’s disease.”
Research on the role of specific nutrients in Parkinson’s disease is conflicting, with no clear evidence supporting or refuting their benefits. For example, a study that followed participants for about 30 years showed no link between reduced Parkinson’s disease risk and vitamin B or folate intake.
On the other hand, there is research suggesting that certain vitamins may help reduce Parkinson’s disease risk, although these nutrients do not operate in isolation. For instance, one recent study showed a connection between vitamins C and E and reduced Parkinson’s disease risk, but factors such as body mass index and coffee consumption appeared to influence the strength of this association.
Consuming polyunsaturated fatty acids along with reducing saturated fatty acid intake has been tied to a reduced risk for Parkinson’s disease.
Additionally, certain foods may offer protective effects, including green and black tea, with consumption of three or more cups per day associated with a delay in motor symptom onset by 7.7 years. Foods high in nicotine content, such as those from the Solanaceae family — including peppers, tomatoes, tomato juice, and potatoes — have also been linked to potential protective benefits.
Diets rich in antioxidants, including carotenoids, lutein, and vitamins E and C, have been robustly linked to a reduced risk for parkinsonism and progression of parkinsonian symptoms in older adults.
Increasing the intake of dietary flavonoids, particularly tea, berry fruits, apples, red wine, and oranges or orange juice, can reduce Parkinson’s disease risk. One study showed that male participants in the highest quintile of total flavonoid consumption had a 40% lower Parkinson’s disease risk compared with those in the lowest quintile. Another study showed that flavonoid-rich foods were also associated with a lower risk for death in patients with Parkinson’s disease.
Food as Medicine
Although recent research shows that the drug development pipeline for Parkinson’s disease is robust, with a wide variety of approaches being developed and evaluated in phase 1 and 2, investigators note that only a limited number of disease-modifying treatments are transitioning to phase 3.
Duda noted that phytochemicals incorporated into the diet might target some of the same mechanisms that are targets of these drugs in development.
“Flavonoids have been shown to stabilize alpha-synuclein in vitro,” he said. “Caffeine, curcumin, resveratrol, and eliminating meat and dairy inhibit mTOR [mammalian target of rapamycin], and mTOR inhibition results in increased autophagy that may help clear alpha-synuclein. Genestein, an isoflavone in soybeans, protects dopaminergic neurons by inhibiting microglia activation. Flavonoids inhibit inflammation by inhibiting release of NO [nitric oxide] and pro-inflammatory cytokines,” he noted.
Ongoing studies of dietary interventions for Parkinson’s disease are exploring various areas, including the potential role of the ketogenic diet in protecting the gut microbiome, optimizing protein intake for muscle preservation and sleep, the effects of psyllium and wheat bran on weight and constipation, and the impact of a gluten-free diet.
Practical Tips for Healthy Eating
Gilbert emphasized that there are no medications or interventions currently available that can delay a Parkinson’s disease diagnosis by up to 17 years, as some dietary patterns have been shown to do, and she noted that it’s not possible to replicate the MeDi diet in a pill. However, she recommended a practical approach to eating that includes a diet low in ultraprocessed foods and high in beneficial nutrients. She encouraged people to shop for “real food” and enjoy a variety of colorful fruits and vegetables.
Duda acknowledged that motivating patients to follow a healthy diet can be difficult. As a result, the focus often shifts to making small adjustments and modifications. For example, he suggested that instead of pairing meat with French fries, people could opt for vegetables or add greens to their meals. Similarly, instead of having eggs and bacon for breakfast, they might choose oatmeal.
Preparing whole-food, plant-based meals may take more time than patients are accustomed to, so Duda suggests that, if possible, patients involve loved ones in both the meal preparation and the meal itself. He explained that a healthy meal can become an opportunity for bonding and that the key is educating them about new meal-related concepts.
Duda reported no relevant financial relationships with the pharmaceutical or food industries. He has received compensation from the Physicians Committee for Responsible Medicine for his lecture delivered at the conference and research grant support from the VA, the National Institutes of Health, the Michael J. Fox Foundation, and the Department of Defense unrelated to this topic. Gilbert reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
For 15 years, John Duda, MD, national director of the VA Parkinson’s Disease Research, Education and Clinical Centers, has urged his patients to “keep waiting” for effective treatments to manage both motor and nonmotor symptoms of Parkinson’s disease.
However, Duda, who also serves as director of the Brain Wellness Clinic at the Corporal Michael J. Crescenz VA Medical Center in Philadelphia, Pennsylvania, recognized the persistent lack of effective drugs to address these symptoms. This prompted him to consider what other evidence-based strategies he could use to support his patients.
“I recognized that nutritional approaches within a broader program that includes medication review, stress management, social connections, adequate sleep, and physical exercise could make a real difference,” he said.
Observational studies have shown an inverse association between dietary patterns and Parkinson’s disease risk, age of onset, symptom severity, and mortality rates — particularly with the Mediterranean diet (MeDi) and the MIND diet, which combines elements of MeDi and the Dietary Approaches to Stop Hypertension (DASH) diet. Although randomized controlled trials are still limited, the epidemiologic evidence supporting dietary interventions is “compelling,” said Duda.
For example, a cross-sectional study comparing 167 participants with Parkinson’s disease vs 119 controls showed that later age of Parkinson’s disease onset correlated with adherence to the MIND diet in women, with a difference of up to 17.4 years (P < .001) between low and high dietary tertiles.
The MeDi was correlated with later onset in men, with differences of up to 8.4 years (P = .002). As previously reported, a healthy diet emphasizing vegetables, fruits, nuts, and grains was inversely associated with prodromal features of Parkinson’s disease, including constipation, excessive daytime sleepiness, and depression. In addition, lower rates of Parkinson’s disease have been shown in populations following vegetarian and vegan dietary patterns.
Does Parkinson’s disease Start in the Gut?
Parkinson’s disease is characterized by decreased short-chain fatty acid–producing bacteria and increased pro-inflammatory species linked to intestinal inflammation and alpha-synuclein aggregation. “There are reasons to believe that a-synuclein accumulation may start in the gut,” Duda noted.
Numerous studies implicate gut microbiome dysbiosis as a pathogenic mechanism in Parkinson’s disease, with gastrointestinal symptoms often predating motor symptoms. Dysbiosis might result in a pro-inflammatory state potentially linked to the recurrent gastrointestinal symptoms. Fecal microbiota transplant may restore a healthier gut environment and beneficially affect Parkinson’s disease symptoms, he said.
Some of the benefits conferred by the MeDi and other healthy diets may be mediated by improving the gut microbiome. Duda cited a study that showed that a 14-day ovo-lacto vegetarian diet intervention and a daily fecal enema for 8 days improved not only the microbiome but also Movement Disorder Society Unified Parkinson’s Disease Rating Scale—part III scores.
Duda also reviewed the role of dietary interventions in addressing common Parkinson’s disease symptoms, such as orthostatic hypotension. He recommended that Parkinson’s disease patients with this condition should avoid eating large meals, increase dietary salt intake, increase fluid intake, and decrease alcohol intake.
Malnutrition affects close to 25% of those with Parkinson’s disease, which is partially attributable to diminished olfaction. Because the experience of taste is largely driven by a sense of smell, patients may be less interested in eating. Duda recommended increasing herbs, spices, and other flavors in food. High caloric–density foods, including nuts, nut butters, and seeds, can boost weight, he said. However, he added, any patient with significant weight loss should consult a nutritionist.
Constipation is one of the most debilitating symptoms of Parkinson’s disease, affecting up to 66% of patients. Duda advised increasing fluid intake, exercise, and dietary fiber and use of stool softeners and laxatives. The MeDi may reduce symptoms of constipation and have a beneficial effect on gut microbiota.
Coffee may be helpful for sleepiness in Parkinson’s disease and may also confer neuroprotective, motor, and cognitive benefits. As an adjuvant treatment, caffeine may alter levodopa pharmacokinetics, reduce dyskinesia, improve gait in patients with freezing and may even reduce the risk of developing Parkinson’s disease, with a maximum benefit reached at approximately three cups of coffee daily.
Problematic Foods
There is also a growing body of evidence regarding the deleterious effects of ultraprocessed foods (UPFs), Duda said. He noted that a recent systematic review and meta-analysis of 28 studies showed that higher UPF intake was significantly associated with an enhanced risk for Parkinson’s disease (relative risk, 1.56; 95% CI, 1.21-2.02). As previously reported, UPFs have been tied to a host of adverse neurologic outcomes, including cognitive decline and stroke.
Although protein is a necessary nutrient, incorporating it into the diet of Parkinson’s disease patients taking levodopa is complicated. Levodopa, a large neutral amino acid (LNAA), competes with other LNAAs for transport to the brain from the small intestine, Duda explained.
“Some people notice that carbidopa-levodopa doesn’t work as well if taken with a high-protein meal.” He recommended taking carbidopa-levodopa 30 minutes before or 60 minutes after meals.
Rebecca Gilbert, MD, PhD, chief mission officer of the American Parkinson’s Disease Association, said that patients with Parkinson’s disease might want to avoid eating protein during the day, concentrating instead on carbohydrates and vegetables and saving the protein for the evening, which is closer to bedtime. Some evidence also supports the use of protein redistribution diets to enhance the clinical response to levodopa and reduce motor fluctuations.
What About Supplements?
It’s “hard to prove that one specific supplement can be protective against Parkinson’s disease because diet consists of many different components and the whole diet may be worth more than the sum of its parts,” Gilbert said. The evidence for individual supplements “isn’t robust enough to say they prevent or treat Parkinson’s disease.”
Research on the role of specific nutrients in Parkinson’s disease is conflicting, with no clear evidence supporting or refuting their benefits. For example, a study that followed participants for about 30 years showed no link between reduced Parkinson’s disease risk and vitamin B or folate intake.
On the other hand, there is research suggesting that certain vitamins may help reduce Parkinson’s disease risk, although these nutrients do not operate in isolation. For instance, one recent study showed a connection between vitamins C and E and reduced Parkinson’s disease risk, but factors such as body mass index and coffee consumption appeared to influence the strength of this association.
Consuming polyunsaturated fatty acids along with reducing saturated fatty acid intake has been tied to a reduced risk for Parkinson’s disease.
Additionally, certain foods may offer protective effects, including green and black tea, with consumption of three or more cups per day associated with a delay in motor symptom onset by 7.7 years. Foods high in nicotine content, such as those from the Solanaceae family — including peppers, tomatoes, tomato juice, and potatoes — have also been linked to potential protective benefits.
Diets rich in antioxidants, including carotenoids, lutein, and vitamins E and C, have been robustly linked to a reduced risk for parkinsonism and progression of parkinsonian symptoms in older adults.
Increasing the intake of dietary flavonoids, particularly tea, berry fruits, apples, red wine, and oranges or orange juice, can reduce Parkinson’s disease risk. One study showed that male participants in the highest quintile of total flavonoid consumption had a 40% lower Parkinson’s disease risk compared with those in the lowest quintile. Another study showed that flavonoid-rich foods were also associated with a lower risk for death in patients with Parkinson’s disease.
Food as Medicine
Although recent research shows that the drug development pipeline for Parkinson’s disease is robust, with a wide variety of approaches being developed and evaluated in phase 1 and 2, investigators note that only a limited number of disease-modifying treatments are transitioning to phase 3.
Duda noted that phytochemicals incorporated into the diet might target some of the same mechanisms that are targets of these drugs in development.
“Flavonoids have been shown to stabilize alpha-synuclein in vitro,” he said. “Caffeine, curcumin, resveratrol, and eliminating meat and dairy inhibit mTOR [mammalian target of rapamycin], and mTOR inhibition results in increased autophagy that may help clear alpha-synuclein. Genestein, an isoflavone in soybeans, protects dopaminergic neurons by inhibiting microglia activation. Flavonoids inhibit inflammation by inhibiting release of NO [nitric oxide] and pro-inflammatory cytokines,” he noted.
Ongoing studies of dietary interventions for Parkinson’s disease are exploring various areas, including the potential role of the ketogenic diet in protecting the gut microbiome, optimizing protein intake for muscle preservation and sleep, the effects of psyllium and wheat bran on weight and constipation, and the impact of a gluten-free diet.
Practical Tips for Healthy Eating
Gilbert emphasized that there are no medications or interventions currently available that can delay a Parkinson’s disease diagnosis by up to 17 years, as some dietary patterns have been shown to do, and she noted that it’s not possible to replicate the MeDi diet in a pill. However, she recommended a practical approach to eating that includes a diet low in ultraprocessed foods and high in beneficial nutrients. She encouraged people to shop for “real food” and enjoy a variety of colorful fruits and vegetables.
Duda acknowledged that motivating patients to follow a healthy diet can be difficult. As a result, the focus often shifts to making small adjustments and modifications. For example, he suggested that instead of pairing meat with French fries, people could opt for vegetables or add greens to their meals. Similarly, instead of having eggs and bacon for breakfast, they might choose oatmeal.
Preparing whole-food, plant-based meals may take more time than patients are accustomed to, so Duda suggests that, if possible, patients involve loved ones in both the meal preparation and the meal itself. He explained that a healthy meal can become an opportunity for bonding and that the key is educating them about new meal-related concepts.
Duda reported no relevant financial relationships with the pharmaceutical or food industries. He has received compensation from the Physicians Committee for Responsible Medicine for his lecture delivered at the conference and research grant support from the VA, the National Institutes of Health, the Michael J. Fox Foundation, and the Department of Defense unrelated to this topic. Gilbert reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
The Strange Untold Story of How Science Solved Narcolepsy
It was 1996, and Masashi Yanagisawa was on the brink of his next discovery.
The Japanese scientist had arrived at the University of Texas Southwestern in Dallas 5 years earlier, setting up his own lab at age 31. After earning his medical degree, he’d gained notoriety as a PhD student when he discovered endothelin, the body’s most potent vasoconstrictor.
Yanagisawa was about to prove this wasn’t a first-timer’s fluke.
His focus was G-protein–coupled receptors (GPCRs), cell surface receptors that respond to a range of molecules and a popular target for drug discovery. The Human Genome Project had just revealed a slew of newly discovered receptors, or “orphan” GPCRs, and identifying an activating molecule could yield a new drug. (That vasoconstrictor endothelin was one such success story, leading to four new drug approvals in the United States over the past quarter century.)
Yanagisawa and his team created 50 cell lines, each expressing one orphan receptor. They applied animal tissue to every line, along with a calcium-sensitive dye. If the cells glowed under the microscope, they had a hit.
“He was basically doing an elaborate fishing expedition,” said Jon Willie, MD, PhD, an associate professor of neurosurgery at Washington University School of Medicine in St. Louis, Missouri, who would later join Yanagisawa’s team.
It wasn’t long before the neon-green fluorescence signaled a match. After isolating the activating molecule, the scientists realized they were dealing with two neuropeptides.
No one had ever seen these proteins before. And no one knew their discovery would set off a decades-long journey that would finally solve a century-old medical mystery — and may even fix one of the biggest health crises of our time, as revealed by research published earlier in 2024. It’s a story of strange coincidences, serendipitous discoveries, and quirky details. Most of all, it’s a fascinating example of how basic science can revolutionize medicine — and how true breakthroughs happen over time and in real time.
But That’s Basic Science for You
Most basic science studies — the early, foundational research that provides the building blocks for science that follows — don’t lead to medical breakthroughs. But some do, often in surprising ways.
Also called curiosity-driven research, basic science aims to fill knowledge gaps to keep science moving, even if the trajectory isn’t always clear.
“The people working on the basic research that led to discoveries that transformed the modern world had no idea at the time,” said Isobel Ronai, PhD, a postdoctoral fellow in life sciences at Harvard University, Cambridge, Massachusetts. “Often, these stories can only be seen in hindsight,” sometimes decades later.
Case in point: For molecular biology techniques — things like DNA sequencing and gene targeting — the lag between basic science and breakthrough is, on average, 23 years. While many of the resulting techniques have received Nobel Prizes, few of the foundational discoveries have been awarded such accolades.
“The scientific glory is more often associated with the downstream applications,” said Ronai. “The importance of basic research can get lost. But it is the foundation for any future application, such as drug development.”
As funding is increasingly funneled toward applied research, basic science can require a certain persistence. What this under-appreciation can obscure is the pathway to discovery — which is often as compelling as the end result, full of unpredictable twists, turns, and even interpersonal intrigue.
And then there’s the fascinating — and definitely complicated — phenomenon of multiple independent discoveries.
As in: What happens when two independent teams discover the same thing at the same time?
Back to Yanagisawa’s Lab ...
... where he and his team learned a few things about those new neuropeptides. Rat brain studies pinpointed the lateral hypothalamus as the peptides’ area of activity — a region often called the brain’s feeding center.
“If you destroy that part of the brain, animals lose appetite,” said Yanagisawa. So these peptides must control feeding, the scientists thought.
Sure enough, injecting the proteins into rat brains led the rodents to start eating.
Satisfied, the team named them “orexin-A” and “orexin-B,” for the Greek word “orexis,” meaning appetite. The brain receptors became “orexin-1” and “orexin-2.” The team prepared to publish its findings in Cell.
But another group beat them to it.
Introducing the ‘Hypocretins’
In early January 1998, a team of Scripps Research Institute scientists, led by J. Gregor Sutcliffe, PhD, released a paper in the journal PNAS. They described a gene encoding for the precursor to two neuropeptides
As the peptides were in the hypothalamus and structurally like secretin (a gut hormone), they called them “hypocretins.” The hypocretin peptides excited neurons in the hypothalamus, and later that year, the scientists discovered that the neurons’ branches extended, tentacle-like, throughout the brain. “Many of the connected areas were involved in sleep-wake control,” said Thomas Kilduff, PhD, who joined the Sutcliffe lab just weeks before the hypocretin discovery. At the time, however, the significance of this finding was not yet clear.
Weeks later, in February 1998, Yanagisawa’s paper came out.
Somehow, two groups, over 1000 miles apart, had stumbled on the same neuropeptides at the same time.
“I first heard about [Yanagisawa’s] paper on NBC Nightly News,” recalls Kilduff. “I was skiing in the mountains, so I had to wait until Monday to get back to the lab to see what the paper was all about.”
He realized that Yanagisawa’s orexin was his lab’s hypocretin, although the study didn’t mention another team’s discovery.
“There may have been accusations. But as far as I know, it’s because [Yanagisawa] didn’t know [about the other paper],” said Willie. “This was not something he produced in 2 months. This was clearly years of work.”
‘Multiple Discovery’ Happens More Often Than You Think
In the mid-20th century, sociologist Robert Merton described the phenomenon of “multiple discovery,” where many scientific discoveries or inventions are made independently at roughly the same time.
“This happens much more frequently in scientific research than people suppose,” said David Pendlebury, head of research analysis at Clarivate’s Institute for Scientific Information, the analytics company’s research arm. (Last year, Pendlebury flagged the hypocretin/orexin discovery for Clarivate’s prestigious Citations Laureates award, an honor that aims to predict, often successfully, who will go on to win the Nobel Prize.)
“People have this idea of the lone researcher making a brilliant discovery,” Pendlebury said. “But more and more, teams find things at the same time.”
While this can — and does — lead to squabbling about who deserves credit, the desire to be first can also be highly motivating, said Mike Schneider, PhD, an assistant professor of philosophy at the University of Missouri, Columbia, who studies the social dynamics of science, potentially leading to faster scientific advancement.
The downside? If two groups produce the same or similar results, but one publishes first, scientific journals tend to reject the second, citing a lack of novelty.
Yet duplicating research is a key step in confirming the validity of a discovery.
That’s why, in 2018, the journal PLOS Biology created a provision for “scooped” scientists, allowing them to submit their paper within 6 months of the first as a complementary finding. Instead of viewing this as redundancy, the editors believe it adds robustness to the research.
‘What the Heck Is This Mouse Doing?’
Even though he’d been scooped, Yanagisawa forged on to the next challenge: Confirming whether orexin regulated feeding.
He began breeding mice missing the orexin gene. His team expected these “knockout” mice to eat less, resulting in a thinner body than other rodents. To the contrary, “they were on average fatter,” said Willie. “They were eating less but weighed more, indicating a slower metabolism.”
The researchers were befuddled. “We were really disappointed, almost desperate about what to do,” said Yanagisawa.
As nocturnal animals eat more at night, he decided they should study the mice after dark. One of his students, Richard Chemelli, MD, bought an infrared video camera from Radio Shack, filming the first 4 hours of the mice’s active period for several nights.
After watching the footage, “Rick called me and said, ‘Let’s get into the lab,’ ” said Willie. “It was four of us on a Saturday looking at these videos, saying, ‘What the heck is this mouse doing?’ ”
While exploring their habitat, the knockout mice would randomly fall over, pop back up after a minute or so, and resume normal activity. This happened over and over — and the scientists were unsure why.
They began monitoring the mice’s brains during these episodes — and made a startling discovery.
The mice weren’t having seizures. They were shifting directly into REM sleep, bypassing the non-REM stage, then quickly toggling back to wake mode.
“That’s when we knew these animals had something akin to narcolepsy,” said Willie.
The team recruited Thomas Scammell, MD, a Harvard neurologist, to investigate whether modafinil — an anti-narcoleptic drug without a clear mechanism — affected orexin neurons.
Two hours after injecting the mice with the medication, the scientists sacrificed them and stained their brains. Remarkably, the number of neurons showing orexin activity had increased ninefold. It seemed modafinil worked by activating the orexin system.
These findings had the potential to crack open the science of narcolepsy, one of the most mysterious sleep disorders.
Unless, of course, another team did it first.
The Mystery of Narcolepsy
Yet another multiple discovery, narcolepsy was first described by two scientists — one in Germany, the other in France — within a short span in the late 1800s.
It would be more than a hundred years before anyone understood the disorder’s cause, even though it affects about 1 in 2000 people.
“Patients were often labeled as lazy and malingerers,” said Kilduff, “since they were sleepy all the time and had this weird motor behavior called cataplexy” or the sudden loss of muscle tone.
In the early 1970s, William Dement, MD, PhD — “the father of sleep medicine” — was searching for a narcoleptic cat to study. He couldn’t find a feline, but several colleagues mentioned dogs with narcolepsy-like symptoms.
Dement, who died in 2020, had found his newest research subjects.
In 1973, he started a narcoleptic dog colony at Stanford University in Palo Alto, California. At first, he focused on poodles and beagles. After discovering their narcolepsy wasn’t genetic, he pivoted to dobermans and labradors. Their narcolepsy was inherited, so he could breed them to populate the colony.
Although human narcolepsy is rarely genetic, it’s otherwise a lot like the version in these dogs.
Both involve daytime sleepiness, “pathological” bouts of REM sleep, and the loss of muscle tone in response to emotions, often positive ones.
The researchers hoped the canines could unlock a treatment for human narcolepsy. They began laying out a path of dog kibble, then injecting the dogs with drugs such as selective serotonin reuptake inhibitors. They wanted to see what might help them stay awake as they excitedly chowed down.
Kilduff also started a molecular genetics program, trying to identify the genetic defect behind canine narcolepsy. But after a parvovirus outbreak, Kilduff resigned from the project, drained from the strain of seeing so many dogs die.
A decade after his departure from the dog colony, his work would dramatically intersect with that of his successor, Emmanuel Mignot, MD, PhD.
“I thought I had closed the narcolepsy chapter in my life forever,” said Kilduff. “Then in 1998, we described this novel neuropeptide, hypocretin, that turned out to be the key to understanding the disorder.”
Narcoleptic Dogs in California, Mutant Mice in Texas
It was modafinil — the same anti-narcoleptic drug Yanagisawa’s team studied — that brought Emmanuel Mignot to the United States. After training as a pharmacologist in France, his home country sent him to Stanford to study the drug, which was discovered by French scientists, as his required military service.
As Kilduff’s replacement at the dog colony, his goal was to figure out how modafinil worked, hoping to attract a US company to develop the drug.
The plan succeeded. Modafinil became Provigil, a billion-dollar narcolepsy drug, and Mignot became “completely fascinated” with the disorder.
“I realized quickly that there was no way we’d find the cause of narcolepsy by finding the mode of action of this drug,” Mignot said. “Most likely, the drug was acting downstream, not at the cause of the disorder.”
To discover the answer, he needed to become a geneticist. And so began his 11-year odyssey to find the cause of canine narcolepsy.
After mapping the dog genome, Mignot set out to find the smallest stretch of chromosome that the narcoleptic animals had in common. “For a very long time, we were stuck with a relatively large region [of DNA],” he recalls. “It was a no man’s land.”
Within that region was the gene for the hypocretin/orexin-2 receptor — the same receptor that Yanagisawa had identified in his first orexin paper. Mignot didn’t immediately pursue that gene as a possibility — even though his students suggested it. Why?
“The decision was simply: Should we lose time to test a possible candidate [gene] among many?” Mignot said.
As Mignot studied dog DNA in California, Yanagisawa was creating mutant mice in Texas. Unbeknownst to either scientist, their work was about to converge.
What Happened Next Is Somewhat Disputed
After diagnosing his mice with narcolepsy, Yanagisawa opted not to share this finding with Mignot, though he knew about Mignot’s interest in the condition. Instead, he asked a colleague to find out how far along Mignot was in his genetics research.
According to Yanagisawa, his colleague didn’t realize how quickly DNA sequencing could happen once a target gene was identified. At a sleep meeting, “he showed Emmanuel all of our raw data. Almost accidentally, he disclosed our findings,” he said. “It was a shock for me.”
Unsure whether he was part of the orexin group, Mignot decided not to reveal that he’d identified the hypocretin/orexin-2 receptor gene as the faulty one in his narcoleptic dogs.
Although he didn’t share this finding, Mignot said he did offer to speak with the lead researcher to see if their findings were the same. If they were, they could jointly submit their articles. But Mignot never heard back.
Meanwhile, back at his lab, Mignot buckled down. While he wasn’t convinced the mouse data proved anything, it did give him the motivation to move faster.
Within weeks, he submitted his findings to Cell, revealing a mutation in the hypocretin/orexin-2 receptor gene as the cause of canine narcolepsy. According to Yanagisawa, the journal’s editor invited him to peer-review the paper, tipping him off to its existence.
“I told him I had a conflict of interest,” said Yanagisawa. “And then we scrambled to finish our manuscript. We wrote up the paper within almost 5 days.”
For a moment, it seemed both papers would be published together in Cell. Instead, on August 6, 1999, Mignot’s study was splashed solo across the journal’s cover.
“At the time, our team was pissed off, but looking back, what else could Emmanuel have done?” said Willie, who was part of Yanagisawa’s team. “The grant he’d been working on for years was at risk. He had it within his power to do the final experiments. Of course he was going to finish.”
Two weeks later, Yanagisawa’s findings followed, also in Cell.
His paper proposed knockout mice as a model for human narcolepsy and orexin as a key regulator of the sleep/wake cycle. With orexin-activated neurons branching into other areas of the brain, the peptide seemed to promote wakefulness by synchronizing several arousal neurotransmitters, such as serotonin, norepinephrine, and histamine.
“If you don’t have orexin, each of those systems can still function, but they’re not as coordinated,” said Willie. “If you have narcolepsy, you’re capable of wakefulness, and you’re capable of sleep. What you can’t do is prevent inappropriately switching between states.”
Together, the two papers painted a clear picture: Narcolepsy was the result of a dysfunction in the hypocretin/orexin system.
After more than a century, the cause of narcolepsy was starting to come into focus.
“This was blockbuster,” said Willie.
By itself, either finding — one in dogs, one in mice — might have been met with skepticism. But in combination, they offered indisputable evidence about narcolepsy’s cause.
The Human Brains in Your Fridge Hold Secrets
Jerome Siegel had been searching for the cause of human narcolepsy for years. A PhD and professor at the University of California, Los Angeles, he had managed to acquire four human narcoleptic brains. As laughter is often the trigger for the sudden shift to REM sleep in humans, he focused on the amygdala, an area linked to emotion.
“I looked in the amygdala and didn’t see anything,” he said. “So the brains stayed in my refrigerator for probably 10 years.”
Then he was invited to review Yanagisawa’s study in Cell. The lightbulb clicked on: Maybe the hypothalamus — not the amygdala — was the area of abnormality. He and his team dug out the decade-old brains.
When they stained the brains, the massive loss of hypocretin-activated neurons was hard to miss: On average, the narcoleptic brains had only about 7000 of the cells versus 70,000 in the average human brain. The scientists also noticed scar tissue in the hypothalamus, indicating that the neurons had at some point died, rather than being absent from birth.
What Siegel didn’t know: Mignot had also acquired a handful of human narcoleptic brains.
Already, he had coauthored a study showing that hypocretin/orexin was undetectable in the cerebrospinal fluid of the majority of the people with narcolepsy his team tested. It seemed clear that the hypocretin/orexin system was flawed — or even broken — in people with the condition.
“It looked like the cause of narcolepsy in humans was indeed this lack of orexin in the brain,” he said. “That was the hypothesis immediately. To me, this is when we established that narcolepsy in humans was due to a lack of orexin. The next thing was to check that the cells were missing.”
Now he could do exactly that.
As expected, Mignot’s team observed a dramatic loss of hypocretin/orexin cells in the narcoleptic brains. They also noticed that a different cell type in the hypothalamus was unaffected. This implied the damage was specific to the hypocretin-activated cells and supported a hunch they already had: That the deficit was the result not of a genetic defect but of an autoimmune attack. (It’s a hypothesis Mignot has spent the last 15 years proving.)
It wasn’t until a gathering in Hawaii, in late August 2000, that the two realized the overlap of their work.
To celebrate his team’s finding, Mignot had invited a group of researchers to Big Island. With his paper scheduled for publication on September 1, he felt comfortable presenting his findings to his guests, which included Siegel.
Until then, “I didn’t know what he had found, and he didn’t know what I had found, which basically was the same thing,” said Siegel.
In yet another strange twist, the two papers were published just weeks apart, simultaneously revealing that human narcoleptics have a depleted supply of the neurons that bind to hypocretin/orexin. The cause of the disorder was at last a certainty.
“Even if I was first, what does it matter? In the end, you need confirmation,” said Mignot. “You need multiple people to make sure that it’s true. It’s good science when things like this happen.”
How All of This Changed Medicine
Since these groundbreaking discoveries, the diagnosis of narcolepsy has become much simpler. Lab tests can now easily measure hypocretin in cerebrospinal fluid, providing a definitive diagnosis.
But the development of narcolepsy treatments has lagged — even though hypocretin/orexin replacement therapy is the obvious answer.
“Almost 25 years have elapsed, and there’s no such therapeutic on the market,” said Kilduff, who now works for SRI International, a non-profit research and development institute.
That’s partly because agonists — drugs that bind to receptors in the brain — are challenging to create, as this requires mimicking the activating molecule’s structure, like copying the grooves of an intricate key.
Antagonists, by comparison, are easier to develop. These act as a gate, blocking access to the receptors. As a result, drugs that promote sleep by thwarting hypocretin/orexin have emerged more quickly, providing a flurry of new options for people with insomnia. The first, suvorexant, was launched in 2014. Two others followed in recent years.
Researchers are hopeful a hypocretin/orexin agonist is on the horizon.
“This is a very hot area of drug development,” said Kilduff. “It’s just a matter of who’s going to get the drug to market first.”
One More Hypocretin/Orexin Surprise — and It Could Be The Biggest
Several years ago, Siegel’s lab received what was supposed to be a healthy human brain — one they could use as a comparison for narcoleptic brains. But researcher Thomas Thannickal, PhD, lead author of the UCLA study linking hypocretin loss to human narcolepsy, noticed something strange: This brain had significantly more hypocretin neurons than average.
Was this due to a seizure? A traumatic death? Siegel called the brain bank to request the donor’s records. He was told they were missing.
Years later, Siegel happened to be visiting the brain bank for another project and found himself in a room adjacent to the medical records. “Nobody was there,” he said, “so I just opened a drawer.”
Shuffling through the brain bank’s files, Siegel found the medical records he’d been told were lost. In the file was a note from the donor, explaining that he was a former heroin addict.
“I almost fell out of my chair,” said Siegel. “I realized this guy’s heroin addiction likely had something to do with his very unusual brain.”
Obviously, opioids affected the orexin system. But how?
“It’s when people are happy that this peptide is released,” said Siegel. “The hypocretin system is not just related to alertness. It’s related to pleasure.”
As Yanagisawa observed early on, hypocretin/orexin does indeed play a role in eating — just not the one he initially thought. The peptides prompted pleasure seeking. So the rodents ate.
In 2018, after acquiring five more brains, Siegel’s group published a study in Translational Medicine showing 54% more detectable hypocretin neurons in the brains of heroin addicts than in those of control individuals.
In 2022, another breakthrough: His team showed that morphine significantly altered the pathways of hypocretin neurons in mice, sending their axons into brain regions associated with addiction. Then, when they removed the mice’s hypocretin neurons and discontinued their daily morphine dose, the rodents showed no symptoms of opioid withdrawal.
This fits the connection with narcolepsy: Among the standard treatments for the condition are amphetamines and other stimulants, which all have addictive potential. Yet, “narcoleptics never abuse these drugs,” Siegel said. “They seem to be uniquely resistant to addiction.”
This could powerfully change the way opioids are administered.
“If you prevent the hypocretin response to opioids, you may be able to prevent opioid addiction,” said Siegel. In other words, blocking the hypocretin system with a drug like those used to treat insomnia may allow patients to experience the pain-relieving benefits of opioids — without the risk for addiction.
His team is currently investigating treatments targeting the hypocretin/orexin system for opioid addiction.
In a study published in July, they found that mice who received suvorexant — the drug for insomnia — didn’t anticipate their daily dose of opioids the way other rodents did. This suggests the medication prevented addiction, without diminishing the pain-relieving effect of opioids.
If it translates to humans, this discovery could potentially save millions of lives.
“I think it’s just us working on this,” said Siegel.
But with hypocretin/orexin, you never know.
A version of this article appeared on Medscape.com.
It was 1996, and Masashi Yanagisawa was on the brink of his next discovery.
The Japanese scientist had arrived at the University of Texas Southwestern in Dallas 5 years earlier, setting up his own lab at age 31. After earning his medical degree, he’d gained notoriety as a PhD student when he discovered endothelin, the body’s most potent vasoconstrictor.
Yanagisawa was about to prove this wasn’t a first-timer’s fluke.
His focus was G-protein–coupled receptors (GPCRs), cell surface receptors that respond to a range of molecules and a popular target for drug discovery. The Human Genome Project had just revealed a slew of newly discovered receptors, or “orphan” GPCRs, and identifying an activating molecule could yield a new drug. (That vasoconstrictor endothelin was one such success story, leading to four new drug approvals in the United States over the past quarter century.)
Yanagisawa and his team created 50 cell lines, each expressing one orphan receptor. They applied animal tissue to every line, along with a calcium-sensitive dye. If the cells glowed under the microscope, they had a hit.
“He was basically doing an elaborate fishing expedition,” said Jon Willie, MD, PhD, an associate professor of neurosurgery at Washington University School of Medicine in St. Louis, Missouri, who would later join Yanagisawa’s team.
It wasn’t long before the neon-green fluorescence signaled a match. After isolating the activating molecule, the scientists realized they were dealing with two neuropeptides.
No one had ever seen these proteins before. And no one knew their discovery would set off a decades-long journey that would finally solve a century-old medical mystery — and may even fix one of the biggest health crises of our time, as revealed by research published earlier in 2024. It’s a story of strange coincidences, serendipitous discoveries, and quirky details. Most of all, it’s a fascinating example of how basic science can revolutionize medicine — and how true breakthroughs happen over time and in real time.
But That’s Basic Science for You
Most basic science studies — the early, foundational research that provides the building blocks for science that follows — don’t lead to medical breakthroughs. But some do, often in surprising ways.
Also called curiosity-driven research, basic science aims to fill knowledge gaps to keep science moving, even if the trajectory isn’t always clear.
“The people working on the basic research that led to discoveries that transformed the modern world had no idea at the time,” said Isobel Ronai, PhD, a postdoctoral fellow in life sciences at Harvard University, Cambridge, Massachusetts. “Often, these stories can only be seen in hindsight,” sometimes decades later.
Case in point: For molecular biology techniques — things like DNA sequencing and gene targeting — the lag between basic science and breakthrough is, on average, 23 years. While many of the resulting techniques have received Nobel Prizes, few of the foundational discoveries have been awarded such accolades.
“The scientific glory is more often associated with the downstream applications,” said Ronai. “The importance of basic research can get lost. But it is the foundation for any future application, such as drug development.”
As funding is increasingly funneled toward applied research, basic science can require a certain persistence. What this under-appreciation can obscure is the pathway to discovery — which is often as compelling as the end result, full of unpredictable twists, turns, and even interpersonal intrigue.
And then there’s the fascinating — and definitely complicated — phenomenon of multiple independent discoveries.
As in: What happens when two independent teams discover the same thing at the same time?
Back to Yanagisawa’s Lab ...
... where he and his team learned a few things about those new neuropeptides. Rat brain studies pinpointed the lateral hypothalamus as the peptides’ area of activity — a region often called the brain’s feeding center.
“If you destroy that part of the brain, animals lose appetite,” said Yanagisawa. So these peptides must control feeding, the scientists thought.
Sure enough, injecting the proteins into rat brains led the rodents to start eating.
Satisfied, the team named them “orexin-A” and “orexin-B,” for the Greek word “orexis,” meaning appetite. The brain receptors became “orexin-1” and “orexin-2.” The team prepared to publish its findings in Cell.
But another group beat them to it.
Introducing the ‘Hypocretins’
In early January 1998, a team of Scripps Research Institute scientists, led by J. Gregor Sutcliffe, PhD, released a paper in the journal PNAS. They described a gene encoding for the precursor to two neuropeptides
As the peptides were in the hypothalamus and structurally like secretin (a gut hormone), they called them “hypocretins.” The hypocretin peptides excited neurons in the hypothalamus, and later that year, the scientists discovered that the neurons’ branches extended, tentacle-like, throughout the brain. “Many of the connected areas were involved in sleep-wake control,” said Thomas Kilduff, PhD, who joined the Sutcliffe lab just weeks before the hypocretin discovery. At the time, however, the significance of this finding was not yet clear.
Weeks later, in February 1998, Yanagisawa’s paper came out.
Somehow, two groups, over 1000 miles apart, had stumbled on the same neuropeptides at the same time.
“I first heard about [Yanagisawa’s] paper on NBC Nightly News,” recalls Kilduff. “I was skiing in the mountains, so I had to wait until Monday to get back to the lab to see what the paper was all about.”
He realized that Yanagisawa’s orexin was his lab’s hypocretin, although the study didn’t mention another team’s discovery.
“There may have been accusations. But as far as I know, it’s because [Yanagisawa] didn’t know [about the other paper],” said Willie. “This was not something he produced in 2 months. This was clearly years of work.”
‘Multiple Discovery’ Happens More Often Than You Think
In the mid-20th century, sociologist Robert Merton described the phenomenon of “multiple discovery,” where many scientific discoveries or inventions are made independently at roughly the same time.
“This happens much more frequently in scientific research than people suppose,” said David Pendlebury, head of research analysis at Clarivate’s Institute for Scientific Information, the analytics company’s research arm. (Last year, Pendlebury flagged the hypocretin/orexin discovery for Clarivate’s prestigious Citations Laureates award, an honor that aims to predict, often successfully, who will go on to win the Nobel Prize.)
“People have this idea of the lone researcher making a brilliant discovery,” Pendlebury said. “But more and more, teams find things at the same time.”
While this can — and does — lead to squabbling about who deserves credit, the desire to be first can also be highly motivating, said Mike Schneider, PhD, an assistant professor of philosophy at the University of Missouri, Columbia, who studies the social dynamics of science, potentially leading to faster scientific advancement.
The downside? If two groups produce the same or similar results, but one publishes first, scientific journals tend to reject the second, citing a lack of novelty.
Yet duplicating research is a key step in confirming the validity of a discovery.
That’s why, in 2018, the journal PLOS Biology created a provision for “scooped” scientists, allowing them to submit their paper within 6 months of the first as a complementary finding. Instead of viewing this as redundancy, the editors believe it adds robustness to the research.
‘What the Heck Is This Mouse Doing?’
Even though he’d been scooped, Yanagisawa forged on to the next challenge: Confirming whether orexin regulated feeding.
He began breeding mice missing the orexin gene. His team expected these “knockout” mice to eat less, resulting in a thinner body than other rodents. To the contrary, “they were on average fatter,” said Willie. “They were eating less but weighed more, indicating a slower metabolism.”
The researchers were befuddled. “We were really disappointed, almost desperate about what to do,” said Yanagisawa.
As nocturnal animals eat more at night, he decided they should study the mice after dark. One of his students, Richard Chemelli, MD, bought an infrared video camera from Radio Shack, filming the first 4 hours of the mice’s active period for several nights.
After watching the footage, “Rick called me and said, ‘Let’s get into the lab,’ ” said Willie. “It was four of us on a Saturday looking at these videos, saying, ‘What the heck is this mouse doing?’ ”
While exploring their habitat, the knockout mice would randomly fall over, pop back up after a minute or so, and resume normal activity. This happened over and over — and the scientists were unsure why.
They began monitoring the mice’s brains during these episodes — and made a startling discovery.
The mice weren’t having seizures. They were shifting directly into REM sleep, bypassing the non-REM stage, then quickly toggling back to wake mode.
“That’s when we knew these animals had something akin to narcolepsy,” said Willie.
The team recruited Thomas Scammell, MD, a Harvard neurologist, to investigate whether modafinil — an anti-narcoleptic drug without a clear mechanism — affected orexin neurons.
Two hours after injecting the mice with the medication, the scientists sacrificed them and stained their brains. Remarkably, the number of neurons showing orexin activity had increased ninefold. It seemed modafinil worked by activating the orexin system.
These findings had the potential to crack open the science of narcolepsy, one of the most mysterious sleep disorders.
Unless, of course, another team did it first.
The Mystery of Narcolepsy
Yet another multiple discovery, narcolepsy was first described by two scientists — one in Germany, the other in France — within a short span in the late 1800s.
It would be more than a hundred years before anyone understood the disorder’s cause, even though it affects about 1 in 2000 people.
“Patients were often labeled as lazy and malingerers,” said Kilduff, “since they were sleepy all the time and had this weird motor behavior called cataplexy” or the sudden loss of muscle tone.
In the early 1970s, William Dement, MD, PhD — “the father of sleep medicine” — was searching for a narcoleptic cat to study. He couldn’t find a feline, but several colleagues mentioned dogs with narcolepsy-like symptoms.
Dement, who died in 2020, had found his newest research subjects.
In 1973, he started a narcoleptic dog colony at Stanford University in Palo Alto, California. At first, he focused on poodles and beagles. After discovering their narcolepsy wasn’t genetic, he pivoted to dobermans and labradors. Their narcolepsy was inherited, so he could breed them to populate the colony.
Although human narcolepsy is rarely genetic, it’s otherwise a lot like the version in these dogs.
Both involve daytime sleepiness, “pathological” bouts of REM sleep, and the loss of muscle tone in response to emotions, often positive ones.
The researchers hoped the canines could unlock a treatment for human narcolepsy. They began laying out a path of dog kibble, then injecting the dogs with drugs such as selective serotonin reuptake inhibitors. They wanted to see what might help them stay awake as they excitedly chowed down.
Kilduff also started a molecular genetics program, trying to identify the genetic defect behind canine narcolepsy. But after a parvovirus outbreak, Kilduff resigned from the project, drained from the strain of seeing so many dogs die.
A decade after his departure from the dog colony, his work would dramatically intersect with that of his successor, Emmanuel Mignot, MD, PhD.
“I thought I had closed the narcolepsy chapter in my life forever,” said Kilduff. “Then in 1998, we described this novel neuropeptide, hypocretin, that turned out to be the key to understanding the disorder.”
Narcoleptic Dogs in California, Mutant Mice in Texas
It was modafinil — the same anti-narcoleptic drug Yanagisawa’s team studied — that brought Emmanuel Mignot to the United States. After training as a pharmacologist in France, his home country sent him to Stanford to study the drug, which was discovered by French scientists, as his required military service.
As Kilduff’s replacement at the dog colony, his goal was to figure out how modafinil worked, hoping to attract a US company to develop the drug.
The plan succeeded. Modafinil became Provigil, a billion-dollar narcolepsy drug, and Mignot became “completely fascinated” with the disorder.
“I realized quickly that there was no way we’d find the cause of narcolepsy by finding the mode of action of this drug,” Mignot said. “Most likely, the drug was acting downstream, not at the cause of the disorder.”
To discover the answer, he needed to become a geneticist. And so began his 11-year odyssey to find the cause of canine narcolepsy.
After mapping the dog genome, Mignot set out to find the smallest stretch of chromosome that the narcoleptic animals had in common. “For a very long time, we were stuck with a relatively large region [of DNA],” he recalls. “It was a no man’s land.”
Within that region was the gene for the hypocretin/orexin-2 receptor — the same receptor that Yanagisawa had identified in his first orexin paper. Mignot didn’t immediately pursue that gene as a possibility — even though his students suggested it. Why?
“The decision was simply: Should we lose time to test a possible candidate [gene] among many?” Mignot said.
As Mignot studied dog DNA in California, Yanagisawa was creating mutant mice in Texas. Unbeknownst to either scientist, their work was about to converge.
What Happened Next Is Somewhat Disputed
After diagnosing his mice with narcolepsy, Yanagisawa opted not to share this finding with Mignot, though he knew about Mignot’s interest in the condition. Instead, he asked a colleague to find out how far along Mignot was in his genetics research.
According to Yanagisawa, his colleague didn’t realize how quickly DNA sequencing could happen once a target gene was identified. At a sleep meeting, “he showed Emmanuel all of our raw data. Almost accidentally, he disclosed our findings,” he said. “It was a shock for me.”
Unsure whether he was part of the orexin group, Mignot decided not to reveal that he’d identified the hypocretin/orexin-2 receptor gene as the faulty one in his narcoleptic dogs.
Although he didn’t share this finding, Mignot said he did offer to speak with the lead researcher to see if their findings were the same. If they were, they could jointly submit their articles. But Mignot never heard back.
Meanwhile, back at his lab, Mignot buckled down. While he wasn’t convinced the mouse data proved anything, it did give him the motivation to move faster.
Within weeks, he submitted his findings to Cell, revealing a mutation in the hypocretin/orexin-2 receptor gene as the cause of canine narcolepsy. According to Yanagisawa, the journal’s editor invited him to peer-review the paper, tipping him off to its existence.
“I told him I had a conflict of interest,” said Yanagisawa. “And then we scrambled to finish our manuscript. We wrote up the paper within almost 5 days.”
For a moment, it seemed both papers would be published together in Cell. Instead, on August 6, 1999, Mignot’s study was splashed solo across the journal’s cover.
“At the time, our team was pissed off, but looking back, what else could Emmanuel have done?” said Willie, who was part of Yanagisawa’s team. “The grant he’d been working on for years was at risk. He had it within his power to do the final experiments. Of course he was going to finish.”
Two weeks later, Yanagisawa’s findings followed, also in Cell.
His paper proposed knockout mice as a model for human narcolepsy and orexin as a key regulator of the sleep/wake cycle. With orexin-activated neurons branching into other areas of the brain, the peptide seemed to promote wakefulness by synchronizing several arousal neurotransmitters, such as serotonin, norepinephrine, and histamine.
“If you don’t have orexin, each of those systems can still function, but they’re not as coordinated,” said Willie. “If you have narcolepsy, you’re capable of wakefulness, and you’re capable of sleep. What you can’t do is prevent inappropriately switching between states.”
Together, the two papers painted a clear picture: Narcolepsy was the result of a dysfunction in the hypocretin/orexin system.
After more than a century, the cause of narcolepsy was starting to come into focus.
“This was blockbuster,” said Willie.
By itself, either finding — one in dogs, one in mice — might have been met with skepticism. But in combination, they offered indisputable evidence about narcolepsy’s cause.
The Human Brains in Your Fridge Hold Secrets
Jerome Siegel had been searching for the cause of human narcolepsy for years. A PhD and professor at the University of California, Los Angeles, he had managed to acquire four human narcoleptic brains. As laughter is often the trigger for the sudden shift to REM sleep in humans, he focused on the amygdala, an area linked to emotion.
“I looked in the amygdala and didn’t see anything,” he said. “So the brains stayed in my refrigerator for probably 10 years.”
Then he was invited to review Yanagisawa’s study in Cell. The lightbulb clicked on: Maybe the hypothalamus — not the amygdala — was the area of abnormality. He and his team dug out the decade-old brains.
When they stained the brains, the massive loss of hypocretin-activated neurons was hard to miss: On average, the narcoleptic brains had only about 7000 of the cells versus 70,000 in the average human brain. The scientists also noticed scar tissue in the hypothalamus, indicating that the neurons had at some point died, rather than being absent from birth.
What Siegel didn’t know: Mignot had also acquired a handful of human narcoleptic brains.
Already, he had coauthored a study showing that hypocretin/orexin was undetectable in the cerebrospinal fluid of the majority of the people with narcolepsy his team tested. It seemed clear that the hypocretin/orexin system was flawed — or even broken — in people with the condition.
“It looked like the cause of narcolepsy in humans was indeed this lack of orexin in the brain,” he said. “That was the hypothesis immediately. To me, this is when we established that narcolepsy in humans was due to a lack of orexin. The next thing was to check that the cells were missing.”
Now he could do exactly that.
As expected, Mignot’s team observed a dramatic loss of hypocretin/orexin cells in the narcoleptic brains. They also noticed that a different cell type in the hypothalamus was unaffected. This implied the damage was specific to the hypocretin-activated cells and supported a hunch they already had: That the deficit was the result not of a genetic defect but of an autoimmune attack. (It’s a hypothesis Mignot has spent the last 15 years proving.)
It wasn’t until a gathering in Hawaii, in late August 2000, that the two realized the overlap of their work.
To celebrate his team’s finding, Mignot had invited a group of researchers to Big Island. With his paper scheduled for publication on September 1, he felt comfortable presenting his findings to his guests, which included Siegel.
Until then, “I didn’t know what he had found, and he didn’t know what I had found, which basically was the same thing,” said Siegel.
In yet another strange twist, the two papers were published just weeks apart, simultaneously revealing that human narcoleptics have a depleted supply of the neurons that bind to hypocretin/orexin. The cause of the disorder was at last a certainty.
“Even if I was first, what does it matter? In the end, you need confirmation,” said Mignot. “You need multiple people to make sure that it’s true. It’s good science when things like this happen.”
How All of This Changed Medicine
Since these groundbreaking discoveries, the diagnosis of narcolepsy has become much simpler. Lab tests can now easily measure hypocretin in cerebrospinal fluid, providing a definitive diagnosis.
But the development of narcolepsy treatments has lagged — even though hypocretin/orexin replacement therapy is the obvious answer.
“Almost 25 years have elapsed, and there’s no such therapeutic on the market,” said Kilduff, who now works for SRI International, a non-profit research and development institute.
That’s partly because agonists — drugs that bind to receptors in the brain — are challenging to create, as this requires mimicking the activating molecule’s structure, like copying the grooves of an intricate key.
Antagonists, by comparison, are easier to develop. These act as a gate, blocking access to the receptors. As a result, drugs that promote sleep by thwarting hypocretin/orexin have emerged more quickly, providing a flurry of new options for people with insomnia. The first, suvorexant, was launched in 2014. Two others followed in recent years.
Researchers are hopeful a hypocretin/orexin agonist is on the horizon.
“This is a very hot area of drug development,” said Kilduff. “It’s just a matter of who’s going to get the drug to market first.”
One More Hypocretin/Orexin Surprise — and It Could Be The Biggest
Several years ago, Siegel’s lab received what was supposed to be a healthy human brain — one they could use as a comparison for narcoleptic brains. But researcher Thomas Thannickal, PhD, lead author of the UCLA study linking hypocretin loss to human narcolepsy, noticed something strange: This brain had significantly more hypocretin neurons than average.
Was this due to a seizure? A traumatic death? Siegel called the brain bank to request the donor’s records. He was told they were missing.
Years later, Siegel happened to be visiting the brain bank for another project and found himself in a room adjacent to the medical records. “Nobody was there,” he said, “so I just opened a drawer.”
Shuffling through the brain bank’s files, Siegel found the medical records he’d been told were lost. In the file was a note from the donor, explaining that he was a former heroin addict.
“I almost fell out of my chair,” said Siegel. “I realized this guy’s heroin addiction likely had something to do with his very unusual brain.”
Obviously, opioids affected the orexin system. But how?
“It’s when people are happy that this peptide is released,” said Siegel. “The hypocretin system is not just related to alertness. It’s related to pleasure.”
As Yanagisawa observed early on, hypocretin/orexin does indeed play a role in eating — just not the one he initially thought. The peptides prompted pleasure seeking. So the rodents ate.
In 2018, after acquiring five more brains, Siegel’s group published a study in Translational Medicine showing 54% more detectable hypocretin neurons in the brains of heroin addicts than in those of control individuals.
In 2022, another breakthrough: His team showed that morphine significantly altered the pathways of hypocretin neurons in mice, sending their axons into brain regions associated with addiction. Then, when they removed the mice’s hypocretin neurons and discontinued their daily morphine dose, the rodents showed no symptoms of opioid withdrawal.
This fits the connection with narcolepsy: Among the standard treatments for the condition are amphetamines and other stimulants, which all have addictive potential. Yet, “narcoleptics never abuse these drugs,” Siegel said. “They seem to be uniquely resistant to addiction.”
This could powerfully change the way opioids are administered.
“If you prevent the hypocretin response to opioids, you may be able to prevent opioid addiction,” said Siegel. In other words, blocking the hypocretin system with a drug like those used to treat insomnia may allow patients to experience the pain-relieving benefits of opioids — without the risk for addiction.
His team is currently investigating treatments targeting the hypocretin/orexin system for opioid addiction.
In a study published in July, they found that mice who received suvorexant — the drug for insomnia — didn’t anticipate their daily dose of opioids the way other rodents did. This suggests the medication prevented addiction, without diminishing the pain-relieving effect of opioids.
If it translates to humans, this discovery could potentially save millions of lives.
“I think it’s just us working on this,” said Siegel.
But with hypocretin/orexin, you never know.
A version of this article appeared on Medscape.com.
It was 1996, and Masashi Yanagisawa was on the brink of his next discovery.
The Japanese scientist had arrived at the University of Texas Southwestern in Dallas 5 years earlier, setting up his own lab at age 31. After earning his medical degree, he’d gained notoriety as a PhD student when he discovered endothelin, the body’s most potent vasoconstrictor.
Yanagisawa was about to prove this wasn’t a first-timer’s fluke.
His focus was G-protein–coupled receptors (GPCRs), cell surface receptors that respond to a range of molecules and a popular target for drug discovery. The Human Genome Project had just revealed a slew of newly discovered receptors, or “orphan” GPCRs, and identifying an activating molecule could yield a new drug. (That vasoconstrictor endothelin was one such success story, leading to four new drug approvals in the United States over the past quarter century.)
Yanagisawa and his team created 50 cell lines, each expressing one orphan receptor. They applied animal tissue to every line, along with a calcium-sensitive dye. If the cells glowed under the microscope, they had a hit.
“He was basically doing an elaborate fishing expedition,” said Jon Willie, MD, PhD, an associate professor of neurosurgery at Washington University School of Medicine in St. Louis, Missouri, who would later join Yanagisawa’s team.
It wasn’t long before the neon-green fluorescence signaled a match. After isolating the activating molecule, the scientists realized they were dealing with two neuropeptides.
No one had ever seen these proteins before. And no one knew their discovery would set off a decades-long journey that would finally solve a century-old medical mystery — and may even fix one of the biggest health crises of our time, as revealed by research published earlier in 2024. It’s a story of strange coincidences, serendipitous discoveries, and quirky details. Most of all, it’s a fascinating example of how basic science can revolutionize medicine — and how true breakthroughs happen over time and in real time.
But That’s Basic Science for You
Most basic science studies — the early, foundational research that provides the building blocks for science that follows — don’t lead to medical breakthroughs. But some do, often in surprising ways.
Also called curiosity-driven research, basic science aims to fill knowledge gaps to keep science moving, even if the trajectory isn’t always clear.
“The people working on the basic research that led to discoveries that transformed the modern world had no idea at the time,” said Isobel Ronai, PhD, a postdoctoral fellow in life sciences at Harvard University, Cambridge, Massachusetts. “Often, these stories can only be seen in hindsight,” sometimes decades later.
Case in point: For molecular biology techniques — things like DNA sequencing and gene targeting — the lag between basic science and breakthrough is, on average, 23 years. While many of the resulting techniques have received Nobel Prizes, few of the foundational discoveries have been awarded such accolades.
“The scientific glory is more often associated with the downstream applications,” said Ronai. “The importance of basic research can get lost. But it is the foundation for any future application, such as drug development.”
As funding is increasingly funneled toward applied research, basic science can require a certain persistence. What this under-appreciation can obscure is the pathway to discovery — which is often as compelling as the end result, full of unpredictable twists, turns, and even interpersonal intrigue.
And then there’s the fascinating — and definitely complicated — phenomenon of multiple independent discoveries.
As in: What happens when two independent teams discover the same thing at the same time?
Back to Yanagisawa’s Lab ...
... where he and his team learned a few things about those new neuropeptides. Rat brain studies pinpointed the lateral hypothalamus as the peptides’ area of activity — a region often called the brain’s feeding center.
“If you destroy that part of the brain, animals lose appetite,” said Yanagisawa. So these peptides must control feeding, the scientists thought.
Sure enough, injecting the proteins into rat brains led the rodents to start eating.
Satisfied, the team named them “orexin-A” and “orexin-B,” for the Greek word “orexis,” meaning appetite. The brain receptors became “orexin-1” and “orexin-2.” The team prepared to publish its findings in Cell.
But another group beat them to it.
Introducing the ‘Hypocretins’
In early January 1998, a team of Scripps Research Institute scientists, led by J. Gregor Sutcliffe, PhD, released a paper in the journal PNAS. They described a gene encoding for the precursor to two neuropeptides
As the peptides were in the hypothalamus and structurally like secretin (a gut hormone), they called them “hypocretins.” The hypocretin peptides excited neurons in the hypothalamus, and later that year, the scientists discovered that the neurons’ branches extended, tentacle-like, throughout the brain. “Many of the connected areas were involved in sleep-wake control,” said Thomas Kilduff, PhD, who joined the Sutcliffe lab just weeks before the hypocretin discovery. At the time, however, the significance of this finding was not yet clear.
Weeks later, in February 1998, Yanagisawa’s paper came out.
Somehow, two groups, over 1000 miles apart, had stumbled on the same neuropeptides at the same time.
“I first heard about [Yanagisawa’s] paper on NBC Nightly News,” recalls Kilduff. “I was skiing in the mountains, so I had to wait until Monday to get back to the lab to see what the paper was all about.”
He realized that Yanagisawa’s orexin was his lab’s hypocretin, although the study didn’t mention another team’s discovery.
“There may have been accusations. But as far as I know, it’s because [Yanagisawa] didn’t know [about the other paper],” said Willie. “This was not something he produced in 2 months. This was clearly years of work.”
‘Multiple Discovery’ Happens More Often Than You Think
In the mid-20th century, sociologist Robert Merton described the phenomenon of “multiple discovery,” where many scientific discoveries or inventions are made independently at roughly the same time.
“This happens much more frequently in scientific research than people suppose,” said David Pendlebury, head of research analysis at Clarivate’s Institute for Scientific Information, the analytics company’s research arm. (Last year, Pendlebury flagged the hypocretin/orexin discovery for Clarivate’s prestigious Citations Laureates award, an honor that aims to predict, often successfully, who will go on to win the Nobel Prize.)
“People have this idea of the lone researcher making a brilliant discovery,” Pendlebury said. “But more and more, teams find things at the same time.”
While this can — and does — lead to squabbling about who deserves credit, the desire to be first can also be highly motivating, said Mike Schneider, PhD, an assistant professor of philosophy at the University of Missouri, Columbia, who studies the social dynamics of science, potentially leading to faster scientific advancement.
The downside? If two groups produce the same or similar results, but one publishes first, scientific journals tend to reject the second, citing a lack of novelty.
Yet duplicating research is a key step in confirming the validity of a discovery.
That’s why, in 2018, the journal PLOS Biology created a provision for “scooped” scientists, allowing them to submit their paper within 6 months of the first as a complementary finding. Instead of viewing this as redundancy, the editors believe it adds robustness to the research.
‘What the Heck Is This Mouse Doing?’
Even though he’d been scooped, Yanagisawa forged on to the next challenge: Confirming whether orexin regulated feeding.
He began breeding mice missing the orexin gene. His team expected these “knockout” mice to eat less, resulting in a thinner body than other rodents. To the contrary, “they were on average fatter,” said Willie. “They were eating less but weighed more, indicating a slower metabolism.”
The researchers were befuddled. “We were really disappointed, almost desperate about what to do,” said Yanagisawa.
As nocturnal animals eat more at night, he decided they should study the mice after dark. One of his students, Richard Chemelli, MD, bought an infrared video camera from Radio Shack, filming the first 4 hours of the mice’s active period for several nights.
After watching the footage, “Rick called me and said, ‘Let’s get into the lab,’ ” said Willie. “It was four of us on a Saturday looking at these videos, saying, ‘What the heck is this mouse doing?’ ”
While exploring their habitat, the knockout mice would randomly fall over, pop back up after a minute or so, and resume normal activity. This happened over and over — and the scientists were unsure why.
They began monitoring the mice’s brains during these episodes — and made a startling discovery.
The mice weren’t having seizures. They were shifting directly into REM sleep, bypassing the non-REM stage, then quickly toggling back to wake mode.
“That’s when we knew these animals had something akin to narcolepsy,” said Willie.
The team recruited Thomas Scammell, MD, a Harvard neurologist, to investigate whether modafinil — an anti-narcoleptic drug without a clear mechanism — affected orexin neurons.
Two hours after injecting the mice with the medication, the scientists sacrificed them and stained their brains. Remarkably, the number of neurons showing orexin activity had increased ninefold. It seemed modafinil worked by activating the orexin system.
These findings had the potential to crack open the science of narcolepsy, one of the most mysterious sleep disorders.
Unless, of course, another team did it first.
The Mystery of Narcolepsy
Yet another multiple discovery, narcolepsy was first described by two scientists — one in Germany, the other in France — within a short span in the late 1800s.
It would be more than a hundred years before anyone understood the disorder’s cause, even though it affects about 1 in 2000 people.
“Patients were often labeled as lazy and malingerers,” said Kilduff, “since they were sleepy all the time and had this weird motor behavior called cataplexy” or the sudden loss of muscle tone.
In the early 1970s, William Dement, MD, PhD — “the father of sleep medicine” — was searching for a narcoleptic cat to study. He couldn’t find a feline, but several colleagues mentioned dogs with narcolepsy-like symptoms.
Dement, who died in 2020, had found his newest research subjects.
In 1973, he started a narcoleptic dog colony at Stanford University in Palo Alto, California. At first, he focused on poodles and beagles. After discovering their narcolepsy wasn’t genetic, he pivoted to dobermans and labradors. Their narcolepsy was inherited, so he could breed them to populate the colony.
Although human narcolepsy is rarely genetic, it’s otherwise a lot like the version in these dogs.
Both involve daytime sleepiness, “pathological” bouts of REM sleep, and the loss of muscle tone in response to emotions, often positive ones.
The researchers hoped the canines could unlock a treatment for human narcolepsy. They began laying out a path of dog kibble, then injecting the dogs with drugs such as selective serotonin reuptake inhibitors. They wanted to see what might help them stay awake as they excitedly chowed down.
Kilduff also started a molecular genetics program, trying to identify the genetic defect behind canine narcolepsy. But after a parvovirus outbreak, Kilduff resigned from the project, drained from the strain of seeing so many dogs die.
A decade after his departure from the dog colony, his work would dramatically intersect with that of his successor, Emmanuel Mignot, MD, PhD.
“I thought I had closed the narcolepsy chapter in my life forever,” said Kilduff. “Then in 1998, we described this novel neuropeptide, hypocretin, that turned out to be the key to understanding the disorder.”
Narcoleptic Dogs in California, Mutant Mice in Texas
It was modafinil — the same anti-narcoleptic drug Yanagisawa’s team studied — that brought Emmanuel Mignot to the United States. After training as a pharmacologist in France, his home country sent him to Stanford to study the drug, which was discovered by French scientists, as his required military service.
As Kilduff’s replacement at the dog colony, his goal was to figure out how modafinil worked, hoping to attract a US company to develop the drug.
The plan succeeded. Modafinil became Provigil, a billion-dollar narcolepsy drug, and Mignot became “completely fascinated” with the disorder.
“I realized quickly that there was no way we’d find the cause of narcolepsy by finding the mode of action of this drug,” Mignot said. “Most likely, the drug was acting downstream, not at the cause of the disorder.”
To discover the answer, he needed to become a geneticist. And so began his 11-year odyssey to find the cause of canine narcolepsy.
After mapping the dog genome, Mignot set out to find the smallest stretch of chromosome that the narcoleptic animals had in common. “For a very long time, we were stuck with a relatively large region [of DNA],” he recalls. “It was a no man’s land.”
Within that region was the gene for the hypocretin/orexin-2 receptor — the same receptor that Yanagisawa had identified in his first orexin paper. Mignot didn’t immediately pursue that gene as a possibility — even though his students suggested it. Why?
“The decision was simply: Should we lose time to test a possible candidate [gene] among many?” Mignot said.
As Mignot studied dog DNA in California, Yanagisawa was creating mutant mice in Texas. Unbeknownst to either scientist, their work was about to converge.
What Happened Next Is Somewhat Disputed
After diagnosing his mice with narcolepsy, Yanagisawa opted not to share this finding with Mignot, though he knew about Mignot’s interest in the condition. Instead, he asked a colleague to find out how far along Mignot was in his genetics research.
According to Yanagisawa, his colleague didn’t realize how quickly DNA sequencing could happen once a target gene was identified. At a sleep meeting, “he showed Emmanuel all of our raw data. Almost accidentally, he disclosed our findings,” he said. “It was a shock for me.”
Unsure whether he was part of the orexin group, Mignot decided not to reveal that he’d identified the hypocretin/orexin-2 receptor gene as the faulty one in his narcoleptic dogs.
Although he didn’t share this finding, Mignot said he did offer to speak with the lead researcher to see if their findings were the same. If they were, they could jointly submit their articles. But Mignot never heard back.
Meanwhile, back at his lab, Mignot buckled down. While he wasn’t convinced the mouse data proved anything, it did give him the motivation to move faster.
Within weeks, he submitted his findings to Cell, revealing a mutation in the hypocretin/orexin-2 receptor gene as the cause of canine narcolepsy. According to Yanagisawa, the journal’s editor invited him to peer-review the paper, tipping him off to its existence.
“I told him I had a conflict of interest,” said Yanagisawa. “And then we scrambled to finish our manuscript. We wrote up the paper within almost 5 days.”
For a moment, it seemed both papers would be published together in Cell. Instead, on August 6, 1999, Mignot’s study was splashed solo across the journal’s cover.
“At the time, our team was pissed off, but looking back, what else could Emmanuel have done?” said Willie, who was part of Yanagisawa’s team. “The grant he’d been working on for years was at risk. He had it within his power to do the final experiments. Of course he was going to finish.”
Two weeks later, Yanagisawa’s findings followed, also in Cell.
His paper proposed knockout mice as a model for human narcolepsy and orexin as a key regulator of the sleep/wake cycle. With orexin-activated neurons branching into other areas of the brain, the peptide seemed to promote wakefulness by synchronizing several arousal neurotransmitters, such as serotonin, norepinephrine, and histamine.
“If you don’t have orexin, each of those systems can still function, but they’re not as coordinated,” said Willie. “If you have narcolepsy, you’re capable of wakefulness, and you’re capable of sleep. What you can’t do is prevent inappropriately switching between states.”
Together, the two papers painted a clear picture: Narcolepsy was the result of a dysfunction in the hypocretin/orexin system.
After more than a century, the cause of narcolepsy was starting to come into focus.
“This was blockbuster,” said Willie.
By itself, either finding — one in dogs, one in mice — might have been met with skepticism. But in combination, they offered indisputable evidence about narcolepsy’s cause.
The Human Brains in Your Fridge Hold Secrets
Jerome Siegel had been searching for the cause of human narcolepsy for years. A PhD and professor at the University of California, Los Angeles, he had managed to acquire four human narcoleptic brains. As laughter is often the trigger for the sudden shift to REM sleep in humans, he focused on the amygdala, an area linked to emotion.
“I looked in the amygdala and didn’t see anything,” he said. “So the brains stayed in my refrigerator for probably 10 years.”
Then he was invited to review Yanagisawa’s study in Cell. The lightbulb clicked on: Maybe the hypothalamus — not the amygdala — was the area of abnormality. He and his team dug out the decade-old brains.
When they stained the brains, the massive loss of hypocretin-activated neurons was hard to miss: On average, the narcoleptic brains had only about 7000 of the cells versus 70,000 in the average human brain. The scientists also noticed scar tissue in the hypothalamus, indicating that the neurons had at some point died, rather than being absent from birth.
What Siegel didn’t know: Mignot had also acquired a handful of human narcoleptic brains.
Already, he had coauthored a study showing that hypocretin/orexin was undetectable in the cerebrospinal fluid of the majority of the people with narcolepsy his team tested. It seemed clear that the hypocretin/orexin system was flawed — or even broken — in people with the condition.
“It looked like the cause of narcolepsy in humans was indeed this lack of orexin in the brain,” he said. “That was the hypothesis immediately. To me, this is when we established that narcolepsy in humans was due to a lack of orexin. The next thing was to check that the cells were missing.”
Now he could do exactly that.
As expected, Mignot’s team observed a dramatic loss of hypocretin/orexin cells in the narcoleptic brains. They also noticed that a different cell type in the hypothalamus was unaffected. This implied the damage was specific to the hypocretin-activated cells and supported a hunch they already had: That the deficit was the result not of a genetic defect but of an autoimmune attack. (It’s a hypothesis Mignot has spent the last 15 years proving.)
It wasn’t until a gathering in Hawaii, in late August 2000, that the two realized the overlap of their work.
To celebrate his team’s finding, Mignot had invited a group of researchers to Big Island. With his paper scheduled for publication on September 1, he felt comfortable presenting his findings to his guests, which included Siegel.
Until then, “I didn’t know what he had found, and he didn’t know what I had found, which basically was the same thing,” said Siegel.
In yet another strange twist, the two papers were published just weeks apart, simultaneously revealing that human narcoleptics have a depleted supply of the neurons that bind to hypocretin/orexin. The cause of the disorder was at last a certainty.
“Even if I was first, what does it matter? In the end, you need confirmation,” said Mignot. “You need multiple people to make sure that it’s true. It’s good science when things like this happen.”
How All of This Changed Medicine
Since these groundbreaking discoveries, the diagnosis of narcolepsy has become much simpler. Lab tests can now easily measure hypocretin in cerebrospinal fluid, providing a definitive diagnosis.
But the development of narcolepsy treatments has lagged — even though hypocretin/orexin replacement therapy is the obvious answer.
“Almost 25 years have elapsed, and there’s no such therapeutic on the market,” said Kilduff, who now works for SRI International, a non-profit research and development institute.
That’s partly because agonists — drugs that bind to receptors in the brain — are challenging to create, as this requires mimicking the activating molecule’s structure, like copying the grooves of an intricate key.
Antagonists, by comparison, are easier to develop. These act as a gate, blocking access to the receptors. As a result, drugs that promote sleep by thwarting hypocretin/orexin have emerged more quickly, providing a flurry of new options for people with insomnia. The first, suvorexant, was launched in 2014. Two others followed in recent years.
Researchers are hopeful a hypocretin/orexin agonist is on the horizon.
“This is a very hot area of drug development,” said Kilduff. “It’s just a matter of who’s going to get the drug to market first.”
One More Hypocretin/Orexin Surprise — and It Could Be The Biggest
Several years ago, Siegel’s lab received what was supposed to be a healthy human brain — one they could use as a comparison for narcoleptic brains. But researcher Thomas Thannickal, PhD, lead author of the UCLA study linking hypocretin loss to human narcolepsy, noticed something strange: This brain had significantly more hypocretin neurons than average.
Was this due to a seizure? A traumatic death? Siegel called the brain bank to request the donor’s records. He was told they were missing.
Years later, Siegel happened to be visiting the brain bank for another project and found himself in a room adjacent to the medical records. “Nobody was there,” he said, “so I just opened a drawer.”
Shuffling through the brain bank’s files, Siegel found the medical records he’d been told were lost. In the file was a note from the donor, explaining that he was a former heroin addict.
“I almost fell out of my chair,” said Siegel. “I realized this guy’s heroin addiction likely had something to do with his very unusual brain.”
Obviously, opioids affected the orexin system. But how?
“It’s when people are happy that this peptide is released,” said Siegel. “The hypocretin system is not just related to alertness. It’s related to pleasure.”
As Yanagisawa observed early on, hypocretin/orexin does indeed play a role in eating — just not the one he initially thought. The peptides prompted pleasure seeking. So the rodents ate.
In 2018, after acquiring five more brains, Siegel’s group published a study in Translational Medicine showing 54% more detectable hypocretin neurons in the brains of heroin addicts than in those of control individuals.
In 2022, another breakthrough: His team showed that morphine significantly altered the pathways of hypocretin neurons in mice, sending their axons into brain regions associated with addiction. Then, when they removed the mice’s hypocretin neurons and discontinued their daily morphine dose, the rodents showed no symptoms of opioid withdrawal.
This fits the connection with narcolepsy: Among the standard treatments for the condition are amphetamines and other stimulants, which all have addictive potential. Yet, “narcoleptics never abuse these drugs,” Siegel said. “They seem to be uniquely resistant to addiction.”
This could powerfully change the way opioids are administered.
“If you prevent the hypocretin response to opioids, you may be able to prevent opioid addiction,” said Siegel. In other words, blocking the hypocretin system with a drug like those used to treat insomnia may allow patients to experience the pain-relieving benefits of opioids — without the risk for addiction.
His team is currently investigating treatments targeting the hypocretin/orexin system for opioid addiction.
In a study published in July, they found that mice who received suvorexant — the drug for insomnia — didn’t anticipate their daily dose of opioids the way other rodents did. This suggests the medication prevented addiction, without diminishing the pain-relieving effect of opioids.
If it translates to humans, this discovery could potentially save millions of lives.
“I think it’s just us working on this,” said Siegel.
But with hypocretin/orexin, you never know.
A version of this article appeared on Medscape.com.
Cancer Mortality Not Higher for Patients With Autoimmune Disease on Checkpoint Inhibitors
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ACR 2024
Two Brain Stim Methods Better Than One for Depression?
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
a new study showed.
METHODOLOGY:
- Researchers conducted a double-blind, sham-controlled randomized clinical trial from 2021 to 2023 at three hospitals in China with 240 participants with MDD (mean age, 32.5 years; 58% women).
- Participants received active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, or sham tDCS + sham rTMS with treatments administered five times per week for 2 weeks.
- tDCS was administered in 20-minute sessions using a 2-mA direct current stimulator, whereas rTMS involved 1600 pulses of 10-Hz stimulation targeting the left dorsolateral prefrontal cortex. Sham treatments used a pseudostimulation coil and only emitted sound.
- The primary outcome was change in the 24-item Hamilton Depression Rating Scale (HDRS-24) total score from baseline to week 2.
- Secondary outcomes included HDRS-24 total score change at week 4, remission rate (HDRS-24 total score ≤ 9), response rate (≥ 50% reduction in HDRS-24 total score), and adverse events.
TAKEAWAY:
- The active tDCS + active rTMS group demonstrated the greatest reduction in mean HDRS-24 score (18.33 ± 5.39) at week 2 compared with sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS (P < .001).
- Response rates at week 2 were notably higher in the active tDCS + active rTMS group (85%) than in the active tDCS + sham rTMS (30%) and sham tDCS + sham rTMS groups (32%).
- The remission rate at week 4 reached 83% in the active tDCS + active rTMS group, which was significantly higher than the remission rates with the other interventions (P < .001).
- The treatments were well tolerated, with no serious adverse events, seizures, or manic symptoms reported across all intervention groups.
IN PRACTICE:
This trial “was the first to evaluate the safety, feasibility, and efficacy of combining tDCS and rTMS in treating depression. Future studies should focus on investigating the mechanism of this synergistic effect and improving the stimulation parameters to optimize the therapeutic effect,” the investigators wrote.
SOURCE:
This study was led by Dongsheng Zhou, MD, Ningbo Kangning Hospital, Ningbo, China. It was published online in JAMA Network Open.
LIMITATIONS:
The brief treatment duration involving 10 sessions may have been insufficient for tDCS and rTMS to demonstrate their full antidepressant potential. The inability to regulate participants’ antidepressant medications throughout the study period presented another limitation. Additionally, the lack of stratified randomization and adjustment for center effects may have introduced variability in the results.
DISCLOSURES:
This study received support from multiple grants, including from the Natural Science Foundation of Zhejiang Province, Basic Public Welfare Research Project of Zhejiang Province, Ningbo Medical and Health Brand Discipline, Ningbo Clinical Medical Research Centre for Mental Health, Ningbo Top Medical and Health Research Program, and the Zhejiang Medical and Health Science and Technology Plan Project. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.