User login
Docs weigh in on insurance coverage for obesity medications
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
Scientists find the ‘on’ switch for energy-burning brown fat
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
FROM PLOS ONE
Metformin, weight management to stop type 2 diabetes in kids
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
TOPLINE:
Nearly one in five adolescents are living with prediabetes, a condition where blood glucose levels are elevated, but are not high enough for a type 2 diabetes (T2D) diagnosis. According to a new study, higher levels of nonfasting glucose and hemoglobin A1c, and worsening obesity are important predictors of progression to T2D. In addition, metformin and weight stabilization may prove to be important interventions for preventing T2D in kids.
METHODOLOGY:
- Researchers did a retrospective chart review of patient data from Vanderbilt University Medical Center Pediatric Prediabetes Clinic, Nashville, Tenn., from May 2015 to August 2022.
- The study included 552 children with prediabetes, defined as abnormal blood glucose (fasting plasma glucose [FPG] ≥ 100 mg/dL, random glucose ≥ 150 mg/dL), or hemoglobin A1c equal to or greater than 5.9%.
- Based on follow-up visits, patients were classified as having progressed to T2D, or nonprogression.
- Researchers analyzed the patients’ initial visit A1c, fasting C-peptide, 2-hour glucose, fasting glucose, and body mass index (BMI), among other baseline characteristics.
TAKEAWAY:
- Thirty-six children (6.5%) progressed to T2D during the duration of the study period.
- The average time to T2D diagnosis was much longer in patients taking metformin (43 months), compared with those not taking the prescribed medication (28 months).
- Worsening obesity was strongly associated with T2D progression – patients who progressed to T2D had a higher BMI at baseline and had continued weight gain.
- A higher baseline A1c, fasting C-peptide, and 2-hour glucose were also associated with progression to T2D.
- In the multivariable analysis, both A1c and 2-hour glucose were strong independent predictors of progression.
- Fasting plasma glucose was not associated with progression to T2D.
IN PRACTICE:
“Weight stabilization and metformin therapy could be important interventions for diabetes prevention in children,” study author Ashley H. Shoemaker, MD, MSci, a pediatric endocrinologist at Vanderbilt University Medical Center in Nashville, Tenn., said in a press release.
In addition, A1c plus a nonfasting glucose may be a feasible way to identify high-risk pediatric patients in a clinical setting.
SOURCE:
This study was performed by Natasha Belsky, Jaclyn Tamaroff, and Ashley H. Shoemaker of the Vanderbilt University Medical Center and the Vanderbilt University School of Medicine in Nashville, Tenn. It was published October 12, 2023, in the Journal of the Endocrine Society
LIMITATIONS:
Additional patients who developed T2D may have been lost to follow-up, since the authors did not contact patients to confirm their disease status. The authors were also unable to establish racial differences in the progression to T2D because of missing data.
DISCLOSURES:
Funding for this study was provided by the National Center for Advancing Translational Sciences. One author has research contracts with Novo Nordisk and Boehringer Ingelheim.
A version of this article first appeared on Medscape.com.
New ‘twincretin’ pemvidutide: Another option for obesity
HAMBURG, GERMANY – , adding a different type of “twincretin” to a growing mix of incretin-based weight-loss drugs in development that also offer additional benefits.
Pemvidutide (Altimmune Inc) is a long-acting “balanced” dual agonist of both glucagon-like peptide 1 (GLP-1) and glucagon that is in development for the treatment of obesity and nonalcoholic steatohepatitis (NASH) but not type 2 diabetes, as its effect on glucose is neutral. Phase 1 data for pemvidutide’s liver effect were presented in 2022.
In contrast, the dual GLP-1-glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide (Mounjaro, Lilly) has been approved for the treatment of type 2 diabetes. It awaits an indication for obesity.
“When you look [at] the results for any given agent, think about obesity as a series of problems. Some overlap, and some don’t. While about 20%-25% of people with obesity also have type 2 diabetes, not everybody does. So the compounds that don’t lower glucose ... those will be great for others who have [fatty liver disease] or hyperlipidemia. ... It’s not going to be one compound for everybody,” said Louis J. Aronne, MD, director of the center for weight management and metabolic clinical research, Weill Cornell Medicine, New York.
Results of a new 24-week interim analysis of data from the phase 2 pemvidutide trial, called MOMENTUM, were presented at the annual meeting of the European Association for the Study of Diabetes by Dr. Aronne.
Included in that session were encore presentations of data for another GLP-1-glucagon dual agonist, survodutide, as well as data for Eli Lilly’s GLP-1-GIP-glucagon “triagonist,” retatrutide. Retatrutide is in development to induce weight loss, while survodutide (Boehringer Ingelheim and Zealand Pharma), like pemvidutide, is in development to induce weight loss and treat fatty liver disease.
Added Dr. Aronne, “As good as [the triple agonist] retatrutide looks, I doubt that every single person with obesity in the world will be treated with it. ... Think about this as a field, the way you treat diabetes and every other chronic illness.”
Asked to comment, session moderator Rajna Golubic, PhD, of the Oxford (England) Centre for Diabetes, Endocrinology and Metabolism, told this news organization, “We need to think in terms of treating beyond weight loss. ... We need to look at the person holistically and at other aspects of cardiometabolic health and treat in a personalized way and choose treatments according to the comorbidities people have.”
Regarding the dual GLP-1-glucagon agonists, including pemvidutide, Dr. Golubic pointed out that the glucagon agonism does the opposite of glucose-lowering agents but that the compound is “balanced for greater affinity for the GLP-1 receptor vs. glucagon, so that the beneficial effects outweigh the effect for glucose but it still harnesses the benefits of glucagon on liver with a decrease in liver fat, with positive effects on heart, positive effects on kidneys, and other beneficial metabolic effects.”
Pemvidutide lowers weight, LDL cholesterol, triglycerides, and blood pressure
Dr. Aronne began his presentation by noting that dyslipidemia, fatty liver disease, and hypertension are the most significant comorbidities of obesity, occurring in 66%-70%, 58%-75%, and 45%-55% of patients, respectively, while type 2 diabetes is less common, at 19%-23%.
Pemvidutide’s GLP-1 receptor agonism reduces appetite, inflammation, and gastric emptying, while glucagon agonism increases lipolysis, mobilizes fat, and increases energy expenditure, Dr. Aronne explained.
The 48-week phase 2 MOMENTUM trial randomly assigned 320 participants with overweight or obesity and at least one obesity-related comorbidity but not diabetes to receive weekly doses of 1.2 mg, 1.8 mg, or 2.4 mg of pemvidutide or placebo. The two lower pemvidutide doses were initiated immediately without titration, while the 2.4-mg dose was titrated rapidly over 4 weeks.
In a prespecified interim analysis of 160 participants, the percent body weight loss at 24 weeks was 10.7%, 9.4%, and 7.3% with the 2.4-mg, 1.8-mg, and 1.2-mg doses, respectively (P < .001). All weight loss values were significant; weight loss with placebo was a nonsignificant 1%.
The proportions of patients who lost at least 5% of their body weight were 84.6%, 66.7%, and 66.7%, respectively, vs. 25% with placebo. Half of the patients who received the 2.4-mg and 1.8-mg doses lost at least 10% of their body weight. Reductions in waist circumference followed suit; the patients who received the 2.4-mg dose lost an average of 10.2 cm, or “in the U.S., about 4 inches or 4 belt loops. That’s pretty good, you need a new belt,” Dr. Aronne commented.
Significant reductions in total cholesterol and triglyceride levels were also seen at week 24 by 16.5% and 25.0%, respectively, with the 2.4-mg dose. Low-density lipoprotein cholesterol levels also dropped, although not significantly; high-density lipoprotein levels dropped significantly.
Systolic blood pressure dropped by 5.5 mm Hg, and diastolic blood pressure dropped by 1.8 mm Hg in the 2.4-mg group and by lesser degrees among the patients who received lower doses. There were no significant changes in heart rate, Dr. Aronne noted.
Glucose homeostasis was preserved in all groups throughout the 24 weeks.
As with all drugs in the incretin class, gastrointestinal adverse events were common. Severe vomiting occurred in one person in the 1.8-mg group and in four with 2.4 mg. Efforts will be made to reduce that in subsequent trials, Dr. Aronne said.
“We have learned over time that going more gradually in titrating up these agents is a better strategy, allowing dose reduction may be a better strategy, and allowing antiemetics temporarily as we increase the dose is a lesson that many have learned doing these trials and of course in our clinical practices,” he commented.
Dr. Golubic told this news organization that the recent emergence of potent incretin-based weight loss drugs is “a huge paradigm shift. The prevalence of obesity will be 35% or higher by 2035. Bariatric surgery isn’t feasible for everyone, and it’s very expensive, so we need drugs to provide benefits in terms of lowering weight, glucose, and other cardiometabolic risk factors.”
The full 48-week data for MOMENTUM will be announced in the fourth quarter of 2023.
Dr. Aronne has received consulting fees from and serves on advisory boards for Allurion, Altimmune, Atria, Gelesis, Jamieson Wellness, Janssen Pharmaceuticals, Jazz Pharmaceuticals, Novo Nordisk, Pfizer, Optum, Eli Lilly, Senda Biosciences, and Versanis; has received research funding from Allurion, AstraZeneca, Gelesis, Janssen Pharmaceuticals, Novo Nordisk, and Eli Lilly; has equity interests in Allurion, ERX Pharmaceuticals, Gelesis, Intellihealth, Jamieson Wellness, and Myos Corp; and serves on a board of directors for ERX Pharmaceuticals, Intellihealth, and Jamieson Wellness. Dr. Golubic has received research support from AstraZeneca.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – , adding a different type of “twincretin” to a growing mix of incretin-based weight-loss drugs in development that also offer additional benefits.
Pemvidutide (Altimmune Inc) is a long-acting “balanced” dual agonist of both glucagon-like peptide 1 (GLP-1) and glucagon that is in development for the treatment of obesity and nonalcoholic steatohepatitis (NASH) but not type 2 diabetes, as its effect on glucose is neutral. Phase 1 data for pemvidutide’s liver effect were presented in 2022.
In contrast, the dual GLP-1-glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide (Mounjaro, Lilly) has been approved for the treatment of type 2 diabetes. It awaits an indication for obesity.
“When you look [at] the results for any given agent, think about obesity as a series of problems. Some overlap, and some don’t. While about 20%-25% of people with obesity also have type 2 diabetes, not everybody does. So the compounds that don’t lower glucose ... those will be great for others who have [fatty liver disease] or hyperlipidemia. ... It’s not going to be one compound for everybody,” said Louis J. Aronne, MD, director of the center for weight management and metabolic clinical research, Weill Cornell Medicine, New York.
Results of a new 24-week interim analysis of data from the phase 2 pemvidutide trial, called MOMENTUM, were presented at the annual meeting of the European Association for the Study of Diabetes by Dr. Aronne.
Included in that session were encore presentations of data for another GLP-1-glucagon dual agonist, survodutide, as well as data for Eli Lilly’s GLP-1-GIP-glucagon “triagonist,” retatrutide. Retatrutide is in development to induce weight loss, while survodutide (Boehringer Ingelheim and Zealand Pharma), like pemvidutide, is in development to induce weight loss and treat fatty liver disease.
Added Dr. Aronne, “As good as [the triple agonist] retatrutide looks, I doubt that every single person with obesity in the world will be treated with it. ... Think about this as a field, the way you treat diabetes and every other chronic illness.”
Asked to comment, session moderator Rajna Golubic, PhD, of the Oxford (England) Centre for Diabetes, Endocrinology and Metabolism, told this news organization, “We need to think in terms of treating beyond weight loss. ... We need to look at the person holistically and at other aspects of cardiometabolic health and treat in a personalized way and choose treatments according to the comorbidities people have.”
Regarding the dual GLP-1-glucagon agonists, including pemvidutide, Dr. Golubic pointed out that the glucagon agonism does the opposite of glucose-lowering agents but that the compound is “balanced for greater affinity for the GLP-1 receptor vs. glucagon, so that the beneficial effects outweigh the effect for glucose but it still harnesses the benefits of glucagon on liver with a decrease in liver fat, with positive effects on heart, positive effects on kidneys, and other beneficial metabolic effects.”
Pemvidutide lowers weight, LDL cholesterol, triglycerides, and blood pressure
Dr. Aronne began his presentation by noting that dyslipidemia, fatty liver disease, and hypertension are the most significant comorbidities of obesity, occurring in 66%-70%, 58%-75%, and 45%-55% of patients, respectively, while type 2 diabetes is less common, at 19%-23%.
Pemvidutide’s GLP-1 receptor agonism reduces appetite, inflammation, and gastric emptying, while glucagon agonism increases lipolysis, mobilizes fat, and increases energy expenditure, Dr. Aronne explained.
The 48-week phase 2 MOMENTUM trial randomly assigned 320 participants with overweight or obesity and at least one obesity-related comorbidity but not diabetes to receive weekly doses of 1.2 mg, 1.8 mg, or 2.4 mg of pemvidutide or placebo. The two lower pemvidutide doses were initiated immediately without titration, while the 2.4-mg dose was titrated rapidly over 4 weeks.
In a prespecified interim analysis of 160 participants, the percent body weight loss at 24 weeks was 10.7%, 9.4%, and 7.3% with the 2.4-mg, 1.8-mg, and 1.2-mg doses, respectively (P < .001). All weight loss values were significant; weight loss with placebo was a nonsignificant 1%.
The proportions of patients who lost at least 5% of their body weight were 84.6%, 66.7%, and 66.7%, respectively, vs. 25% with placebo. Half of the patients who received the 2.4-mg and 1.8-mg doses lost at least 10% of their body weight. Reductions in waist circumference followed suit; the patients who received the 2.4-mg dose lost an average of 10.2 cm, or “in the U.S., about 4 inches or 4 belt loops. That’s pretty good, you need a new belt,” Dr. Aronne commented.
Significant reductions in total cholesterol and triglyceride levels were also seen at week 24 by 16.5% and 25.0%, respectively, with the 2.4-mg dose. Low-density lipoprotein cholesterol levels also dropped, although not significantly; high-density lipoprotein levels dropped significantly.
Systolic blood pressure dropped by 5.5 mm Hg, and diastolic blood pressure dropped by 1.8 mm Hg in the 2.4-mg group and by lesser degrees among the patients who received lower doses. There were no significant changes in heart rate, Dr. Aronne noted.
Glucose homeostasis was preserved in all groups throughout the 24 weeks.
As with all drugs in the incretin class, gastrointestinal adverse events were common. Severe vomiting occurred in one person in the 1.8-mg group and in four with 2.4 mg. Efforts will be made to reduce that in subsequent trials, Dr. Aronne said.
“We have learned over time that going more gradually in titrating up these agents is a better strategy, allowing dose reduction may be a better strategy, and allowing antiemetics temporarily as we increase the dose is a lesson that many have learned doing these trials and of course in our clinical practices,” he commented.
Dr. Golubic told this news organization that the recent emergence of potent incretin-based weight loss drugs is “a huge paradigm shift. The prevalence of obesity will be 35% or higher by 2035. Bariatric surgery isn’t feasible for everyone, and it’s very expensive, so we need drugs to provide benefits in terms of lowering weight, glucose, and other cardiometabolic risk factors.”
The full 48-week data for MOMENTUM will be announced in the fourth quarter of 2023.
Dr. Aronne has received consulting fees from and serves on advisory boards for Allurion, Altimmune, Atria, Gelesis, Jamieson Wellness, Janssen Pharmaceuticals, Jazz Pharmaceuticals, Novo Nordisk, Pfizer, Optum, Eli Lilly, Senda Biosciences, and Versanis; has received research funding from Allurion, AstraZeneca, Gelesis, Janssen Pharmaceuticals, Novo Nordisk, and Eli Lilly; has equity interests in Allurion, ERX Pharmaceuticals, Gelesis, Intellihealth, Jamieson Wellness, and Myos Corp; and serves on a board of directors for ERX Pharmaceuticals, Intellihealth, and Jamieson Wellness. Dr. Golubic has received research support from AstraZeneca.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – , adding a different type of “twincretin” to a growing mix of incretin-based weight-loss drugs in development that also offer additional benefits.
Pemvidutide (Altimmune Inc) is a long-acting “balanced” dual agonist of both glucagon-like peptide 1 (GLP-1) and glucagon that is in development for the treatment of obesity and nonalcoholic steatohepatitis (NASH) but not type 2 diabetes, as its effect on glucose is neutral. Phase 1 data for pemvidutide’s liver effect were presented in 2022.
In contrast, the dual GLP-1-glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide (Mounjaro, Lilly) has been approved for the treatment of type 2 diabetes. It awaits an indication for obesity.
“When you look [at] the results for any given agent, think about obesity as a series of problems. Some overlap, and some don’t. While about 20%-25% of people with obesity also have type 2 diabetes, not everybody does. So the compounds that don’t lower glucose ... those will be great for others who have [fatty liver disease] or hyperlipidemia. ... It’s not going to be one compound for everybody,” said Louis J. Aronne, MD, director of the center for weight management and metabolic clinical research, Weill Cornell Medicine, New York.
Results of a new 24-week interim analysis of data from the phase 2 pemvidutide trial, called MOMENTUM, were presented at the annual meeting of the European Association for the Study of Diabetes by Dr. Aronne.
Included in that session were encore presentations of data for another GLP-1-glucagon dual agonist, survodutide, as well as data for Eli Lilly’s GLP-1-GIP-glucagon “triagonist,” retatrutide. Retatrutide is in development to induce weight loss, while survodutide (Boehringer Ingelheim and Zealand Pharma), like pemvidutide, is in development to induce weight loss and treat fatty liver disease.
Added Dr. Aronne, “As good as [the triple agonist] retatrutide looks, I doubt that every single person with obesity in the world will be treated with it. ... Think about this as a field, the way you treat diabetes and every other chronic illness.”
Asked to comment, session moderator Rajna Golubic, PhD, of the Oxford (England) Centre for Diabetes, Endocrinology and Metabolism, told this news organization, “We need to think in terms of treating beyond weight loss. ... We need to look at the person holistically and at other aspects of cardiometabolic health and treat in a personalized way and choose treatments according to the comorbidities people have.”
Regarding the dual GLP-1-glucagon agonists, including pemvidutide, Dr. Golubic pointed out that the glucagon agonism does the opposite of glucose-lowering agents but that the compound is “balanced for greater affinity for the GLP-1 receptor vs. glucagon, so that the beneficial effects outweigh the effect for glucose but it still harnesses the benefits of glucagon on liver with a decrease in liver fat, with positive effects on heart, positive effects on kidneys, and other beneficial metabolic effects.”
Pemvidutide lowers weight, LDL cholesterol, triglycerides, and blood pressure
Dr. Aronne began his presentation by noting that dyslipidemia, fatty liver disease, and hypertension are the most significant comorbidities of obesity, occurring in 66%-70%, 58%-75%, and 45%-55% of patients, respectively, while type 2 diabetes is less common, at 19%-23%.
Pemvidutide’s GLP-1 receptor agonism reduces appetite, inflammation, and gastric emptying, while glucagon agonism increases lipolysis, mobilizes fat, and increases energy expenditure, Dr. Aronne explained.
The 48-week phase 2 MOMENTUM trial randomly assigned 320 participants with overweight or obesity and at least one obesity-related comorbidity but not diabetes to receive weekly doses of 1.2 mg, 1.8 mg, or 2.4 mg of pemvidutide or placebo. The two lower pemvidutide doses were initiated immediately without titration, while the 2.4-mg dose was titrated rapidly over 4 weeks.
In a prespecified interim analysis of 160 participants, the percent body weight loss at 24 weeks was 10.7%, 9.4%, and 7.3% with the 2.4-mg, 1.8-mg, and 1.2-mg doses, respectively (P < .001). All weight loss values were significant; weight loss with placebo was a nonsignificant 1%.
The proportions of patients who lost at least 5% of their body weight were 84.6%, 66.7%, and 66.7%, respectively, vs. 25% with placebo. Half of the patients who received the 2.4-mg and 1.8-mg doses lost at least 10% of their body weight. Reductions in waist circumference followed suit; the patients who received the 2.4-mg dose lost an average of 10.2 cm, or “in the U.S., about 4 inches or 4 belt loops. That’s pretty good, you need a new belt,” Dr. Aronne commented.
Significant reductions in total cholesterol and triglyceride levels were also seen at week 24 by 16.5% and 25.0%, respectively, with the 2.4-mg dose. Low-density lipoprotein cholesterol levels also dropped, although not significantly; high-density lipoprotein levels dropped significantly.
Systolic blood pressure dropped by 5.5 mm Hg, and diastolic blood pressure dropped by 1.8 mm Hg in the 2.4-mg group and by lesser degrees among the patients who received lower doses. There were no significant changes in heart rate, Dr. Aronne noted.
Glucose homeostasis was preserved in all groups throughout the 24 weeks.
As with all drugs in the incretin class, gastrointestinal adverse events were common. Severe vomiting occurred in one person in the 1.8-mg group and in four with 2.4 mg. Efforts will be made to reduce that in subsequent trials, Dr. Aronne said.
“We have learned over time that going more gradually in titrating up these agents is a better strategy, allowing dose reduction may be a better strategy, and allowing antiemetics temporarily as we increase the dose is a lesson that many have learned doing these trials and of course in our clinical practices,” he commented.
Dr. Golubic told this news organization that the recent emergence of potent incretin-based weight loss drugs is “a huge paradigm shift. The prevalence of obesity will be 35% or higher by 2035. Bariatric surgery isn’t feasible for everyone, and it’s very expensive, so we need drugs to provide benefits in terms of lowering weight, glucose, and other cardiometabolic risk factors.”
The full 48-week data for MOMENTUM will be announced in the fourth quarter of 2023.
Dr. Aronne has received consulting fees from and serves on advisory boards for Allurion, Altimmune, Atria, Gelesis, Jamieson Wellness, Janssen Pharmaceuticals, Jazz Pharmaceuticals, Novo Nordisk, Pfizer, Optum, Eli Lilly, Senda Biosciences, and Versanis; has received research funding from Allurion, AstraZeneca, Gelesis, Janssen Pharmaceuticals, Novo Nordisk, and Eli Lilly; has equity interests in Allurion, ERX Pharmaceuticals, Gelesis, Intellihealth, Jamieson Wellness, and Myos Corp; and serves on a board of directors for ERX Pharmaceuticals, Intellihealth, and Jamieson Wellness. Dr. Golubic has received research support from AstraZeneca.
A version of this article first appeared on Medscape.com.
AT EASD 2023
Roflumilast side effect benefits patients with psoriasis and overweight/obesity
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
AT THE EADV CONGRESS
GLP-1 agonists linked to higher risk for rare but serious GI complications
Patients taking either of these glucagonlike peptide-1 (GLP-1) receptor agonists had a 9-fold elevation in risk for pancreatitis. They were also 4 times more likely to develop bowel obstruction and over 3.5 times more likely to experience gastroparesis.
The research letter was published online in JAMA.
Investigators say their findings are not about scaring people off the weight-loss drugs, but instead about increasing awareness that these potential adverse outcomes can happen.
“Given the wide use of these drugs, these adverse events, although rare, must be considered by patients thinking about using them for weight loss,” said lead author Mohit Sodhi, MSc, in a news release about the study. Mr. Sodhi is a graduate of the experimental medicine program at the University of British Columbia in Vancouver, and also a 4th-year medical student at UBC.
People taking a GLP-1 agonist to treat diabetes might be more willing to accept the risks, given their potential advantages, especially that of lowering the risk for heart problems, said Mahyar Etminan, PharmD, MSc, the study’s senior author and an expert in drug safety and pharmacoepidemiology at UBC. “But those who are otherwise healthy and just taking them for weight loss might want to be more careful in weighing the risk–benefit equation.”
People taking these drugs for weight loss have an approximately 1%-2% chance of experiencing these events, including a 1% risk for gastroparesis, Dr. Etminan said.
Key findings
The study included 4,144 people taking liraglutide, 613 taking semaglutide, and 654 taking naltrexone/bupropion based on medical records between 2006 and 2020.
They included patients with a recent history of obesity but excluded those with diabetes or who had been prescribed another diabetes medication.
The use of GLP-1 agonists, compared with naltrexone/bupropion, was associated with an increased risk for pancreatitis (adjusted hazard ratio, 9.09; 95% confidence interval, 1.25-66.00), bowel obstruction (HR, 4.22; 95% CI, 1.02-17.40), and gastroparesis (HR, 3.67; 95% CI, 1.15-11.90).
The study also found a higher incidence of biliary disease, but the difference was not statistically significant (HR, 1.50; 95% CI, 0.89-2.53). The incidence of biliary disease (per 1,000 person-years) was 11.7 for semaglutide, 18.6 for liraglutide, and 12.6 for naltrexone/bupropion.
Not the first report of GI issues
“This important paper confirms the safety signals hinted at in previous randomized controlled trials,” said Carel Le Roux, MBChB, PhD, professor of metabolic medicine, Ulster University, Coleraine, Ireland, and professor of experimental pathology at University College Dublin.
“The limitations of the paper are acknowledged but do not detract from the value of the robust data,” Dr. Le Roux said. “Patients should be informed of the low risk of serious complications, such as pancreatitis, gastroparesis, and bowel obstruction, before they start semaglutide or liraglutide.”
This is not the first report of GI issues associated with GLP-1 agonists, but it’s one of the largest. Most reports have been anecdotal. The U.S. Food and Drug Administration announced on Sept. 28 that it would require manufacturers to include a warning about gastrointestinal ileus on the Ozempic (semaglutide) label.
“The results from this study highlight how important it is that patients access these drugs only through trusted medical professionals, and only with ongoing support and monitoring,” noted Simon Cork, PhD, senior lecturer in physiology, Anglia Ruskin University in Cambridge, England.
Dr. Cork added that “it’s important to look at this in the proper context.” Obesity significantly increases the risk for developing cardiovascular disease, type 2 diabetes, cancer, gallbladder disease, and stroke, risks that fall dramatically with clinically meaningful and sustained weight loss, he said.
“For the overwhelming majority of patients for whom these drugs are targeted (those with the most severe forms of obesity), the benefits of weight loss far outweigh the risks,” Dr. Cork said.
The study was independently supported. Mr. Sodhi, Dr. Etminan, and Dr. Cork report no relevant financial relationships. Dr. Le Roux is a consultant and has received research funding and reimbursement of travel expenses from Novo Nordisk.
A version of this article first appeared on Medscape.com.
Patients taking either of these glucagonlike peptide-1 (GLP-1) receptor agonists had a 9-fold elevation in risk for pancreatitis. They were also 4 times more likely to develop bowel obstruction and over 3.5 times more likely to experience gastroparesis.
The research letter was published online in JAMA.
Investigators say their findings are not about scaring people off the weight-loss drugs, but instead about increasing awareness that these potential adverse outcomes can happen.
“Given the wide use of these drugs, these adverse events, although rare, must be considered by patients thinking about using them for weight loss,” said lead author Mohit Sodhi, MSc, in a news release about the study. Mr. Sodhi is a graduate of the experimental medicine program at the University of British Columbia in Vancouver, and also a 4th-year medical student at UBC.
People taking a GLP-1 agonist to treat diabetes might be more willing to accept the risks, given their potential advantages, especially that of lowering the risk for heart problems, said Mahyar Etminan, PharmD, MSc, the study’s senior author and an expert in drug safety and pharmacoepidemiology at UBC. “But those who are otherwise healthy and just taking them for weight loss might want to be more careful in weighing the risk–benefit equation.”
People taking these drugs for weight loss have an approximately 1%-2% chance of experiencing these events, including a 1% risk for gastroparesis, Dr. Etminan said.
Key findings
The study included 4,144 people taking liraglutide, 613 taking semaglutide, and 654 taking naltrexone/bupropion based on medical records between 2006 and 2020.
They included patients with a recent history of obesity but excluded those with diabetes or who had been prescribed another diabetes medication.
The use of GLP-1 agonists, compared with naltrexone/bupropion, was associated with an increased risk for pancreatitis (adjusted hazard ratio, 9.09; 95% confidence interval, 1.25-66.00), bowel obstruction (HR, 4.22; 95% CI, 1.02-17.40), and gastroparesis (HR, 3.67; 95% CI, 1.15-11.90).
The study also found a higher incidence of biliary disease, but the difference was not statistically significant (HR, 1.50; 95% CI, 0.89-2.53). The incidence of biliary disease (per 1,000 person-years) was 11.7 for semaglutide, 18.6 for liraglutide, and 12.6 for naltrexone/bupropion.
Not the first report of GI issues
“This important paper confirms the safety signals hinted at in previous randomized controlled trials,” said Carel Le Roux, MBChB, PhD, professor of metabolic medicine, Ulster University, Coleraine, Ireland, and professor of experimental pathology at University College Dublin.
“The limitations of the paper are acknowledged but do not detract from the value of the robust data,” Dr. Le Roux said. “Patients should be informed of the low risk of serious complications, such as pancreatitis, gastroparesis, and bowel obstruction, before they start semaglutide or liraglutide.”
This is not the first report of GI issues associated with GLP-1 agonists, but it’s one of the largest. Most reports have been anecdotal. The U.S. Food and Drug Administration announced on Sept. 28 that it would require manufacturers to include a warning about gastrointestinal ileus on the Ozempic (semaglutide) label.
“The results from this study highlight how important it is that patients access these drugs only through trusted medical professionals, and only with ongoing support and monitoring,” noted Simon Cork, PhD, senior lecturer in physiology, Anglia Ruskin University in Cambridge, England.
Dr. Cork added that “it’s important to look at this in the proper context.” Obesity significantly increases the risk for developing cardiovascular disease, type 2 diabetes, cancer, gallbladder disease, and stroke, risks that fall dramatically with clinically meaningful and sustained weight loss, he said.
“For the overwhelming majority of patients for whom these drugs are targeted (those with the most severe forms of obesity), the benefits of weight loss far outweigh the risks,” Dr. Cork said.
The study was independently supported. Mr. Sodhi, Dr. Etminan, and Dr. Cork report no relevant financial relationships. Dr. Le Roux is a consultant and has received research funding and reimbursement of travel expenses from Novo Nordisk.
A version of this article first appeared on Medscape.com.
Patients taking either of these glucagonlike peptide-1 (GLP-1) receptor agonists had a 9-fold elevation in risk for pancreatitis. They were also 4 times more likely to develop bowel obstruction and over 3.5 times more likely to experience gastroparesis.
The research letter was published online in JAMA.
Investigators say their findings are not about scaring people off the weight-loss drugs, but instead about increasing awareness that these potential adverse outcomes can happen.
“Given the wide use of these drugs, these adverse events, although rare, must be considered by patients thinking about using them for weight loss,” said lead author Mohit Sodhi, MSc, in a news release about the study. Mr. Sodhi is a graduate of the experimental medicine program at the University of British Columbia in Vancouver, and also a 4th-year medical student at UBC.
People taking a GLP-1 agonist to treat diabetes might be more willing to accept the risks, given their potential advantages, especially that of lowering the risk for heart problems, said Mahyar Etminan, PharmD, MSc, the study’s senior author and an expert in drug safety and pharmacoepidemiology at UBC. “But those who are otherwise healthy and just taking them for weight loss might want to be more careful in weighing the risk–benefit equation.”
People taking these drugs for weight loss have an approximately 1%-2% chance of experiencing these events, including a 1% risk for gastroparesis, Dr. Etminan said.
Key findings
The study included 4,144 people taking liraglutide, 613 taking semaglutide, and 654 taking naltrexone/bupropion based on medical records between 2006 and 2020.
They included patients with a recent history of obesity but excluded those with diabetes or who had been prescribed another diabetes medication.
The use of GLP-1 agonists, compared with naltrexone/bupropion, was associated with an increased risk for pancreatitis (adjusted hazard ratio, 9.09; 95% confidence interval, 1.25-66.00), bowel obstruction (HR, 4.22; 95% CI, 1.02-17.40), and gastroparesis (HR, 3.67; 95% CI, 1.15-11.90).
The study also found a higher incidence of biliary disease, but the difference was not statistically significant (HR, 1.50; 95% CI, 0.89-2.53). The incidence of biliary disease (per 1,000 person-years) was 11.7 for semaglutide, 18.6 for liraglutide, and 12.6 for naltrexone/bupropion.
Not the first report of GI issues
“This important paper confirms the safety signals hinted at in previous randomized controlled trials,” said Carel Le Roux, MBChB, PhD, professor of metabolic medicine, Ulster University, Coleraine, Ireland, and professor of experimental pathology at University College Dublin.
“The limitations of the paper are acknowledged but do not detract from the value of the robust data,” Dr. Le Roux said. “Patients should be informed of the low risk of serious complications, such as pancreatitis, gastroparesis, and bowel obstruction, before they start semaglutide or liraglutide.”
This is not the first report of GI issues associated with GLP-1 agonists, but it’s one of the largest. Most reports have been anecdotal. The U.S. Food and Drug Administration announced on Sept. 28 that it would require manufacturers to include a warning about gastrointestinal ileus on the Ozempic (semaglutide) label.
“The results from this study highlight how important it is that patients access these drugs only through trusted medical professionals, and only with ongoing support and monitoring,” noted Simon Cork, PhD, senior lecturer in physiology, Anglia Ruskin University in Cambridge, England.
Dr. Cork added that “it’s important to look at this in the proper context.” Obesity significantly increases the risk for developing cardiovascular disease, type 2 diabetes, cancer, gallbladder disease, and stroke, risks that fall dramatically with clinically meaningful and sustained weight loss, he said.
“For the overwhelming majority of patients for whom these drugs are targeted (those with the most severe forms of obesity), the benefits of weight loss far outweigh the risks,” Dr. Cork said.
The study was independently supported. Mr. Sodhi, Dr. Etminan, and Dr. Cork report no relevant financial relationships. Dr. Le Roux is a consultant and has received research funding and reimbursement of travel expenses from Novo Nordisk.
A version of this article first appeared on Medscape.com.
FROM JAMA
Obesity linked to multiple ills in MS study
MILAN – Swedish researchers reported at the 9th Joint ECTRIMS-ACTRIMS meeting.
In a group of 3,249 subjects tracked for up to 5 years (74% female; mean age, 37.8 years), patients who were obese at diagnosis were 1.41 times more likely than normal-weight patients to reach an Expanded Disability Status Scale (EDSS) score of 3. About 35% of 355 obese subjects (body mass index > 30 kg/m2) reached that level versus 29% of 713 overweight patients (BMI, 25-30) and 28% of 1,475 normal-weight patients (BMI, 18.5-24.99).
Among subjects whose BMI category didn’t change over follow-up, those who were obese at diagnosis were more likely to develop cognitive worsening than those who weren’t obese (hazard ratio, 1.47, 95% confidence interval, 1.08-2.01).
Lars Alfredsson, PhD, a professor at the Karolinska Institutet, Stockholm, who presented the study findings, said in an interview that they fill a gap in knowledge about obesity and MS. “It is known that obesity around the age of 20 or in adolescence is a risk factor for developing MS. But much less is known in regard to progression, and the studies have been very inconclusive.”
The researchers tracked patients via the Swedish MS registry: 1,475 of normal weight, 713 overweight, and 355 obese. Before adjustment for factors such as age, gender, and baseline EDSS, obese subjects were 1.51 times more likely to reach EDSS score 3 than normal-weight subjects.
Obese subjects whose BMI level didn’t change over time were 1.70 times more likely than the nonobese to develop physical worsening as measured by an increased Multiple Sclerosis Impact Scale physical score of 7.5 points or more, and they were 1.36 times more likely to have psychological worsening as measured by increased MSIS-28 psychological score of 7.5 points or more.
Also, among subjects whose BMI didn’t change over time, the likelihood of cognitive disability worsening was 1.47 times higher among obese participants versus nonobese participants. Worsening was defined as an increased Symbol Digit Modalities Test score of 8 points or more.
The level of excess cognitive decline “will affect people significantly,” Dr. Alfredsson said.
While obesity can counterintuitively provide a protective effect in some diseases, he said there’s no sign of such an effect in the subjects.
As for limitations, Dr. Alfredsson noted in his presentation that BMI data is self-reported, and it’s possible that the researchers didn’t adjust their statistics to reflect important confounders.
A 2023 German study of outcomes in MS patients with obesity came to similar conclusions. It tracked 1,066 subjects for up to 6 years and found that “median time to reach EDSS 3 was 0.99 years for patients with BMI of 30 or higher and 1.46 years for nonobese patients. Risk to reach EDSS 3 over 6 years was significantly increased in patients with BMI of at least 30, compared with patients with BMI less than 30 after adjustment for sex, age, smoking (HR, 1.87; 95% CI, 1.3-2.6; P < .001), and independent of disease-modifying therapies.”
However, the German researchers found no link between obesity and higher levels of relapse, contrast-enhancing MRI lesions, or MRI T2 lesion burden.
Interpretation and commentary
Could obesity be causing worse outcomes? The new study doesn’t provide insight into cause and effect. However, obesity may speed up progression via low-grade inflammation, Dr. Alfredsson said.
What can clinicians do with the information from the study? If patients are obese, it can be a good idea to more carefully monitor them and use reliable tools to improve their progression, Dr. Alfredsson said.
In an interview, Michael D. Kornberg, MD, PhD, an assistant professor of neurology at Johns Hopkins University, Baltimore, who was not involved with the study, agreed with Dr. Alfredsson that other research has linked obesity early in life to higher rates of MS. He added that “a number of studies have shown that comorbidities in general are usually associated with a higher rate of disability.”
Dr. Kornberg said the new research is important, and he noted that it has a “robust” cohort because of its larger size.
Could patients with MS reverse the risk of progression and other poor outcomes by losing weight? “It’s hard to say,” Dr. Kornberg said. “We have to be cautious when we assume causation. There’s a plausible rationale that obesity might worsen progression in MS, but it could just be a marker of some other factor that reflects a different phenotype of MS.”
He doesn’t think it’s likely that weight loss would “dramatically reverse the biology of MS,” but he said reversing the obesity epidemic would still be a good thing. An interventional study could examine the effects of weight-loss intervention on disability measures, he said, “and that’s the next step.”
Also contacted for commentary, Adil Harroud, MD, a neurologist at McGill University who studies obesity in MS, said research suggests that “obesity seems to exacerbate MS disability. While some studies show no effect, the majority indicate a detrimental impact.”
However, “the effect of obesity on MS progression remains unclear. Animal studies suggest that shifts in immune cell subsets and functions may play a role, but the relevance to humans is yet to be determined,” he said.
Dr. Harroud, who did not take part in the new study, said it’s “one of the largest examining the impact of obesity on MS disability.” He added that “the cohort was relatively early in their disease course, suggesting that obesity impacts even the early stages of MS. This underscores the importance of obesity as a modifiable risk factor for disability accumulation.”
As for why obesity affects MS, he said one theory is that obesity plays a role through its impact on vitamin D levels. “However, using a genetic approach, we have demonstrated that, at least for MS risk, the effect of obesity is independent of vitamin D. This is also likely true for MS progression, as recent trials of vitamin D supplementation have not shown a meaningful impact on MS outcomes.”
According to Dr. Harroud, “other theories suggest that obesity leads to a pro-inflammatory immune shift. Additionally, it has been proposed that obesity may influence the response to disease-modifying therapy by reducing drug bioavailability, potentially necessitating weight-based dosing for some therapies.”
Dr. Alfredsson reported receiving grants from the Swedish Research Council, the Swedish Research Council for Health Working Life and Welfare, and the Swedish Brain Foundation and personal fees from Teva and Biogene Idec. Some of the other study authors reported various disclosures. Dr. Kornberg and Dr. Harroud reported no relevant disclosures.
This article was updated 10/20/23.
MILAN – Swedish researchers reported at the 9th Joint ECTRIMS-ACTRIMS meeting.
In a group of 3,249 subjects tracked for up to 5 years (74% female; mean age, 37.8 years), patients who were obese at diagnosis were 1.41 times more likely than normal-weight patients to reach an Expanded Disability Status Scale (EDSS) score of 3. About 35% of 355 obese subjects (body mass index > 30 kg/m2) reached that level versus 29% of 713 overweight patients (BMI, 25-30) and 28% of 1,475 normal-weight patients (BMI, 18.5-24.99).
Among subjects whose BMI category didn’t change over follow-up, those who were obese at diagnosis were more likely to develop cognitive worsening than those who weren’t obese (hazard ratio, 1.47, 95% confidence interval, 1.08-2.01).
Lars Alfredsson, PhD, a professor at the Karolinska Institutet, Stockholm, who presented the study findings, said in an interview that they fill a gap in knowledge about obesity and MS. “It is known that obesity around the age of 20 or in adolescence is a risk factor for developing MS. But much less is known in regard to progression, and the studies have been very inconclusive.”
The researchers tracked patients via the Swedish MS registry: 1,475 of normal weight, 713 overweight, and 355 obese. Before adjustment for factors such as age, gender, and baseline EDSS, obese subjects were 1.51 times more likely to reach EDSS score 3 than normal-weight subjects.
Obese subjects whose BMI level didn’t change over time were 1.70 times more likely than the nonobese to develop physical worsening as measured by an increased Multiple Sclerosis Impact Scale physical score of 7.5 points or more, and they were 1.36 times more likely to have psychological worsening as measured by increased MSIS-28 psychological score of 7.5 points or more.
Also, among subjects whose BMI didn’t change over time, the likelihood of cognitive disability worsening was 1.47 times higher among obese participants versus nonobese participants. Worsening was defined as an increased Symbol Digit Modalities Test score of 8 points or more.
The level of excess cognitive decline “will affect people significantly,” Dr. Alfredsson said.
While obesity can counterintuitively provide a protective effect in some diseases, he said there’s no sign of such an effect in the subjects.
As for limitations, Dr. Alfredsson noted in his presentation that BMI data is self-reported, and it’s possible that the researchers didn’t adjust their statistics to reflect important confounders.
A 2023 German study of outcomes in MS patients with obesity came to similar conclusions. It tracked 1,066 subjects for up to 6 years and found that “median time to reach EDSS 3 was 0.99 years for patients with BMI of 30 or higher and 1.46 years for nonobese patients. Risk to reach EDSS 3 over 6 years was significantly increased in patients with BMI of at least 30, compared with patients with BMI less than 30 after adjustment for sex, age, smoking (HR, 1.87; 95% CI, 1.3-2.6; P < .001), and independent of disease-modifying therapies.”
However, the German researchers found no link between obesity and higher levels of relapse, contrast-enhancing MRI lesions, or MRI T2 lesion burden.
Interpretation and commentary
Could obesity be causing worse outcomes? The new study doesn’t provide insight into cause and effect. However, obesity may speed up progression via low-grade inflammation, Dr. Alfredsson said.
What can clinicians do with the information from the study? If patients are obese, it can be a good idea to more carefully monitor them and use reliable tools to improve their progression, Dr. Alfredsson said.
In an interview, Michael D. Kornberg, MD, PhD, an assistant professor of neurology at Johns Hopkins University, Baltimore, who was not involved with the study, agreed with Dr. Alfredsson that other research has linked obesity early in life to higher rates of MS. He added that “a number of studies have shown that comorbidities in general are usually associated with a higher rate of disability.”
Dr. Kornberg said the new research is important, and he noted that it has a “robust” cohort because of its larger size.
Could patients with MS reverse the risk of progression and other poor outcomes by losing weight? “It’s hard to say,” Dr. Kornberg said. “We have to be cautious when we assume causation. There’s a plausible rationale that obesity might worsen progression in MS, but it could just be a marker of some other factor that reflects a different phenotype of MS.”
He doesn’t think it’s likely that weight loss would “dramatically reverse the biology of MS,” but he said reversing the obesity epidemic would still be a good thing. An interventional study could examine the effects of weight-loss intervention on disability measures, he said, “and that’s the next step.”
Also contacted for commentary, Adil Harroud, MD, a neurologist at McGill University who studies obesity in MS, said research suggests that “obesity seems to exacerbate MS disability. While some studies show no effect, the majority indicate a detrimental impact.”
However, “the effect of obesity on MS progression remains unclear. Animal studies suggest that shifts in immune cell subsets and functions may play a role, but the relevance to humans is yet to be determined,” he said.
Dr. Harroud, who did not take part in the new study, said it’s “one of the largest examining the impact of obesity on MS disability.” He added that “the cohort was relatively early in their disease course, suggesting that obesity impacts even the early stages of MS. This underscores the importance of obesity as a modifiable risk factor for disability accumulation.”
As for why obesity affects MS, he said one theory is that obesity plays a role through its impact on vitamin D levels. “However, using a genetic approach, we have demonstrated that, at least for MS risk, the effect of obesity is independent of vitamin D. This is also likely true for MS progression, as recent trials of vitamin D supplementation have not shown a meaningful impact on MS outcomes.”
According to Dr. Harroud, “other theories suggest that obesity leads to a pro-inflammatory immune shift. Additionally, it has been proposed that obesity may influence the response to disease-modifying therapy by reducing drug bioavailability, potentially necessitating weight-based dosing for some therapies.”
Dr. Alfredsson reported receiving grants from the Swedish Research Council, the Swedish Research Council for Health Working Life and Welfare, and the Swedish Brain Foundation and personal fees from Teva and Biogene Idec. Some of the other study authors reported various disclosures. Dr. Kornberg and Dr. Harroud reported no relevant disclosures.
This article was updated 10/20/23.
MILAN – Swedish researchers reported at the 9th Joint ECTRIMS-ACTRIMS meeting.
In a group of 3,249 subjects tracked for up to 5 years (74% female; mean age, 37.8 years), patients who were obese at diagnosis were 1.41 times more likely than normal-weight patients to reach an Expanded Disability Status Scale (EDSS) score of 3. About 35% of 355 obese subjects (body mass index > 30 kg/m2) reached that level versus 29% of 713 overweight patients (BMI, 25-30) and 28% of 1,475 normal-weight patients (BMI, 18.5-24.99).
Among subjects whose BMI category didn’t change over follow-up, those who were obese at diagnosis were more likely to develop cognitive worsening than those who weren’t obese (hazard ratio, 1.47, 95% confidence interval, 1.08-2.01).
Lars Alfredsson, PhD, a professor at the Karolinska Institutet, Stockholm, who presented the study findings, said in an interview that they fill a gap in knowledge about obesity and MS. “It is known that obesity around the age of 20 or in adolescence is a risk factor for developing MS. But much less is known in regard to progression, and the studies have been very inconclusive.”
The researchers tracked patients via the Swedish MS registry: 1,475 of normal weight, 713 overweight, and 355 obese. Before adjustment for factors such as age, gender, and baseline EDSS, obese subjects were 1.51 times more likely to reach EDSS score 3 than normal-weight subjects.
Obese subjects whose BMI level didn’t change over time were 1.70 times more likely than the nonobese to develop physical worsening as measured by an increased Multiple Sclerosis Impact Scale physical score of 7.5 points or more, and they were 1.36 times more likely to have psychological worsening as measured by increased MSIS-28 psychological score of 7.5 points or more.
Also, among subjects whose BMI didn’t change over time, the likelihood of cognitive disability worsening was 1.47 times higher among obese participants versus nonobese participants. Worsening was defined as an increased Symbol Digit Modalities Test score of 8 points or more.
The level of excess cognitive decline “will affect people significantly,” Dr. Alfredsson said.
While obesity can counterintuitively provide a protective effect in some diseases, he said there’s no sign of such an effect in the subjects.
As for limitations, Dr. Alfredsson noted in his presentation that BMI data is self-reported, and it’s possible that the researchers didn’t adjust their statistics to reflect important confounders.
A 2023 German study of outcomes in MS patients with obesity came to similar conclusions. It tracked 1,066 subjects for up to 6 years and found that “median time to reach EDSS 3 was 0.99 years for patients with BMI of 30 or higher and 1.46 years for nonobese patients. Risk to reach EDSS 3 over 6 years was significantly increased in patients with BMI of at least 30, compared with patients with BMI less than 30 after adjustment for sex, age, smoking (HR, 1.87; 95% CI, 1.3-2.6; P < .001), and independent of disease-modifying therapies.”
However, the German researchers found no link between obesity and higher levels of relapse, contrast-enhancing MRI lesions, or MRI T2 lesion burden.
Interpretation and commentary
Could obesity be causing worse outcomes? The new study doesn’t provide insight into cause and effect. However, obesity may speed up progression via low-grade inflammation, Dr. Alfredsson said.
What can clinicians do with the information from the study? If patients are obese, it can be a good idea to more carefully monitor them and use reliable tools to improve their progression, Dr. Alfredsson said.
In an interview, Michael D. Kornberg, MD, PhD, an assistant professor of neurology at Johns Hopkins University, Baltimore, who was not involved with the study, agreed with Dr. Alfredsson that other research has linked obesity early in life to higher rates of MS. He added that “a number of studies have shown that comorbidities in general are usually associated with a higher rate of disability.”
Dr. Kornberg said the new research is important, and he noted that it has a “robust” cohort because of its larger size.
Could patients with MS reverse the risk of progression and other poor outcomes by losing weight? “It’s hard to say,” Dr. Kornberg said. “We have to be cautious when we assume causation. There’s a plausible rationale that obesity might worsen progression in MS, but it could just be a marker of some other factor that reflects a different phenotype of MS.”
He doesn’t think it’s likely that weight loss would “dramatically reverse the biology of MS,” but he said reversing the obesity epidemic would still be a good thing. An interventional study could examine the effects of weight-loss intervention on disability measures, he said, “and that’s the next step.”
Also contacted for commentary, Adil Harroud, MD, a neurologist at McGill University who studies obesity in MS, said research suggests that “obesity seems to exacerbate MS disability. While some studies show no effect, the majority indicate a detrimental impact.”
However, “the effect of obesity on MS progression remains unclear. Animal studies suggest that shifts in immune cell subsets and functions may play a role, but the relevance to humans is yet to be determined,” he said.
Dr. Harroud, who did not take part in the new study, said it’s “one of the largest examining the impact of obesity on MS disability.” He added that “the cohort was relatively early in their disease course, suggesting that obesity impacts even the early stages of MS. This underscores the importance of obesity as a modifiable risk factor for disability accumulation.”
As for why obesity affects MS, he said one theory is that obesity plays a role through its impact on vitamin D levels. “However, using a genetic approach, we have demonstrated that, at least for MS risk, the effect of obesity is independent of vitamin D. This is also likely true for MS progression, as recent trials of vitamin D supplementation have not shown a meaningful impact on MS outcomes.”
According to Dr. Harroud, “other theories suggest that obesity leads to a pro-inflammatory immune shift. Additionally, it has been proposed that obesity may influence the response to disease-modifying therapy by reducing drug bioavailability, potentially necessitating weight-based dosing for some therapies.”
Dr. Alfredsson reported receiving grants from the Swedish Research Council, the Swedish Research Council for Health Working Life and Welfare, and the Swedish Brain Foundation and personal fees from Teva and Biogene Idec. Some of the other study authors reported various disclosures. Dr. Kornberg and Dr. Harroud reported no relevant disclosures.
This article was updated 10/20/23.
AT ECTRIMS 2023
Are they ‘antiobesity medications’ or ‘weight-loss drugs’?
A simple Google search for the terms “weight-loss pens,” “weight-loss drugs,” and “weight-loss medications” displays seven times more results than a search for terms like “antiobesity medications,” “antiobesity drugs,” or “drugs (or medications) to treat obesity.” The same search applied to academic databases yields the opposite results: fewer than 500 results for “weight-loss drugs/agents/medications” and 19,000 results for “antiobesity agents,” for example.
To highlight the importance of the language used to talk about obesity treatment, researchers affiliated with the Brazilian Society of Endocrinology and Metabolism (SBEM) and the Brazilian Association for the Study of Obesity and Metabolic Syndrome (ABESO) released a statement on the subject at the Brazilian Congress of Update in Endocrinology and Metabolism 2023. On the basis of the study by the ABESO and the SBEM, the statement proposes abandoning the use of the term “weight-loss medications” in scientific publications and, most importantly, in the media.
“Put together, we believe that the common use of the term ‘weight-loss medications’ by media and the public, as well as by doctors and the scientific community, contributes to stigma, and certainly that language matters,” study author Paulo Augusto Carvalho Miranda, MD, PhD, chair of SBEM, said in an interview.
“When we refer to these medications as ‘weight-loss drugs,’ we are using derogatory terms to refer to medications that were extensively studied before their launch onto the market and approved by a regulatory authority to treat a disease called obesity,” said study author Márcio Mancini, MD, PhD, deputy chair of the SBEM’s Obesity Department.
Beyond semantics
Another article published by this news organization presents the initiative of a global task force comprising 60 leaders in the clinical management of obesity, who proposed a new name for the disease. According to the leader of the project, Francesco Rubino, MD, “The word is so stigmatized, with so much misunderstanding and misperception, that some might say the only solution is to change the name.” Following this same logic, the authors of the Brazilian study believe that changing how we refer to medications may improve perceptions of health care professionals and patients toward prevention and treatment strategies for obesity.
According to Dr. Miranda, the first step is “remembering that how we refer to people, diseases, and treatments makes all the difference, especially in situations like obesity, a stigmatized disease loaded with misconceptions. It is not merely an issue of semantics, but also an issue of reducing the stigma surrounding the subject.”
According to Dr. Miranda, the primary purpose of the statement is to highlight the uniqueness of the situation and the importance of encouraging the use of the expressions “antiobesity medications” and “medications to treat obesity” to help reduce the stigma and improve adherence and persistence in obesity treatment.
Impact in practice
The statement also emphasizes that obesity pharmacotherapy is widely underused in patients with obesity and that, in the United States, it is prescribed only for approximately 3% of adults with the disease. Weight management programs for this patient population stress implementing lifestyle changes, and only 1.1% of participants are prescribed medications.
According to the statement, the term “weight-loss medications” contributes to the concept that their use has an aesthetic goal and can be consumed by anyone who desires to lose weight.
In addition to ensuring the correct use of language, Dr. Mancini adds that it is essential for doctors to seek and present pharmacologic treatment for obesity as something that will improve patient health. This means stressing that obesity can be controlled with a 10% loss in body weight, just as other chronic diseases, such as diabetes, can be controlled. Moreover, it is important to point out that medications also have a crucial role in optimizing weight maintenance in the long term.
Another issue Dr. Mancini raised is the prejudice that many doctors have against people with obesity. Health professionals should recognize they are also subject to weight bias and that the way they communicate with patients could have a profound effect on health-related outcomes.
“The stigma surrounding obesity can lead to bullying, even in the patient’s home by their relatives; this is very common. Weight stigma is so strong that it hinders patient health and decreases the likelihood of the patient seeking specialized care,” Dr. Mancini warned.
According to the authors, it is of utmost importance to understand that an individual should not be defined by his or disease (as by the use of the terms “obese” or “diabetic”) but rather understood to live with this disease (“individual with obesity” or “with diabetes”). Dr. Mancini suggests the following strategies that health care professionals can adopt while caring for patients with obesity:
- Speak to patients with empathy and respect, avoiding the use of judgmental words.
- Ask if they would like to discuss the “weight issue,” “BMI issue,” terms that are better received by the public, instead of saying “excess fat” or “excess weight.”
- If the patient agrees to talk about the subject, reinforce that this is a chronic health problem that requires longterm treatment and give him or her short, medium, and longterm options.
Lastly, the authors highlighted the importance of differentiating between regulatory agency–approved medications and over-the-counter drugs and supplements that are often sold as “weight-loss agents” and are responsible for an unacceptably high rate of emergency visits.
This article was translated from the Medscape Portuguese Edition. A version appeared on Medscape.com.
A simple Google search for the terms “weight-loss pens,” “weight-loss drugs,” and “weight-loss medications” displays seven times more results than a search for terms like “antiobesity medications,” “antiobesity drugs,” or “drugs (or medications) to treat obesity.” The same search applied to academic databases yields the opposite results: fewer than 500 results for “weight-loss drugs/agents/medications” and 19,000 results for “antiobesity agents,” for example.
To highlight the importance of the language used to talk about obesity treatment, researchers affiliated with the Brazilian Society of Endocrinology and Metabolism (SBEM) and the Brazilian Association for the Study of Obesity and Metabolic Syndrome (ABESO) released a statement on the subject at the Brazilian Congress of Update in Endocrinology and Metabolism 2023. On the basis of the study by the ABESO and the SBEM, the statement proposes abandoning the use of the term “weight-loss medications” in scientific publications and, most importantly, in the media.
“Put together, we believe that the common use of the term ‘weight-loss medications’ by media and the public, as well as by doctors and the scientific community, contributes to stigma, and certainly that language matters,” study author Paulo Augusto Carvalho Miranda, MD, PhD, chair of SBEM, said in an interview.
“When we refer to these medications as ‘weight-loss drugs,’ we are using derogatory terms to refer to medications that were extensively studied before their launch onto the market and approved by a regulatory authority to treat a disease called obesity,” said study author Márcio Mancini, MD, PhD, deputy chair of the SBEM’s Obesity Department.
Beyond semantics
Another article published by this news organization presents the initiative of a global task force comprising 60 leaders in the clinical management of obesity, who proposed a new name for the disease. According to the leader of the project, Francesco Rubino, MD, “The word is so stigmatized, with so much misunderstanding and misperception, that some might say the only solution is to change the name.” Following this same logic, the authors of the Brazilian study believe that changing how we refer to medications may improve perceptions of health care professionals and patients toward prevention and treatment strategies for obesity.
According to Dr. Miranda, the first step is “remembering that how we refer to people, diseases, and treatments makes all the difference, especially in situations like obesity, a stigmatized disease loaded with misconceptions. It is not merely an issue of semantics, but also an issue of reducing the stigma surrounding the subject.”
According to Dr. Miranda, the primary purpose of the statement is to highlight the uniqueness of the situation and the importance of encouraging the use of the expressions “antiobesity medications” and “medications to treat obesity” to help reduce the stigma and improve adherence and persistence in obesity treatment.
Impact in practice
The statement also emphasizes that obesity pharmacotherapy is widely underused in patients with obesity and that, in the United States, it is prescribed only for approximately 3% of adults with the disease. Weight management programs for this patient population stress implementing lifestyle changes, and only 1.1% of participants are prescribed medications.
According to the statement, the term “weight-loss medications” contributes to the concept that their use has an aesthetic goal and can be consumed by anyone who desires to lose weight.
In addition to ensuring the correct use of language, Dr. Mancini adds that it is essential for doctors to seek and present pharmacologic treatment for obesity as something that will improve patient health. This means stressing that obesity can be controlled with a 10% loss in body weight, just as other chronic diseases, such as diabetes, can be controlled. Moreover, it is important to point out that medications also have a crucial role in optimizing weight maintenance in the long term.
Another issue Dr. Mancini raised is the prejudice that many doctors have against people with obesity. Health professionals should recognize they are also subject to weight bias and that the way they communicate with patients could have a profound effect on health-related outcomes.
“The stigma surrounding obesity can lead to bullying, even in the patient’s home by their relatives; this is very common. Weight stigma is so strong that it hinders patient health and decreases the likelihood of the patient seeking specialized care,” Dr. Mancini warned.
According to the authors, it is of utmost importance to understand that an individual should not be defined by his or disease (as by the use of the terms “obese” or “diabetic”) but rather understood to live with this disease (“individual with obesity” or “with diabetes”). Dr. Mancini suggests the following strategies that health care professionals can adopt while caring for patients with obesity:
- Speak to patients with empathy and respect, avoiding the use of judgmental words.
- Ask if they would like to discuss the “weight issue,” “BMI issue,” terms that are better received by the public, instead of saying “excess fat” or “excess weight.”
- If the patient agrees to talk about the subject, reinforce that this is a chronic health problem that requires longterm treatment and give him or her short, medium, and longterm options.
Lastly, the authors highlighted the importance of differentiating between regulatory agency–approved medications and over-the-counter drugs and supplements that are often sold as “weight-loss agents” and are responsible for an unacceptably high rate of emergency visits.
This article was translated from the Medscape Portuguese Edition. A version appeared on Medscape.com.
A simple Google search for the terms “weight-loss pens,” “weight-loss drugs,” and “weight-loss medications” displays seven times more results than a search for terms like “antiobesity medications,” “antiobesity drugs,” or “drugs (or medications) to treat obesity.” The same search applied to academic databases yields the opposite results: fewer than 500 results for “weight-loss drugs/agents/medications” and 19,000 results for “antiobesity agents,” for example.
To highlight the importance of the language used to talk about obesity treatment, researchers affiliated with the Brazilian Society of Endocrinology and Metabolism (SBEM) and the Brazilian Association for the Study of Obesity and Metabolic Syndrome (ABESO) released a statement on the subject at the Brazilian Congress of Update in Endocrinology and Metabolism 2023. On the basis of the study by the ABESO and the SBEM, the statement proposes abandoning the use of the term “weight-loss medications” in scientific publications and, most importantly, in the media.
“Put together, we believe that the common use of the term ‘weight-loss medications’ by media and the public, as well as by doctors and the scientific community, contributes to stigma, and certainly that language matters,” study author Paulo Augusto Carvalho Miranda, MD, PhD, chair of SBEM, said in an interview.
“When we refer to these medications as ‘weight-loss drugs,’ we are using derogatory terms to refer to medications that were extensively studied before their launch onto the market and approved by a regulatory authority to treat a disease called obesity,” said study author Márcio Mancini, MD, PhD, deputy chair of the SBEM’s Obesity Department.
Beyond semantics
Another article published by this news organization presents the initiative of a global task force comprising 60 leaders in the clinical management of obesity, who proposed a new name for the disease. According to the leader of the project, Francesco Rubino, MD, “The word is so stigmatized, with so much misunderstanding and misperception, that some might say the only solution is to change the name.” Following this same logic, the authors of the Brazilian study believe that changing how we refer to medications may improve perceptions of health care professionals and patients toward prevention and treatment strategies for obesity.
According to Dr. Miranda, the first step is “remembering that how we refer to people, diseases, and treatments makes all the difference, especially in situations like obesity, a stigmatized disease loaded with misconceptions. It is not merely an issue of semantics, but also an issue of reducing the stigma surrounding the subject.”
According to Dr. Miranda, the primary purpose of the statement is to highlight the uniqueness of the situation and the importance of encouraging the use of the expressions “antiobesity medications” and “medications to treat obesity” to help reduce the stigma and improve adherence and persistence in obesity treatment.
Impact in practice
The statement also emphasizes that obesity pharmacotherapy is widely underused in patients with obesity and that, in the United States, it is prescribed only for approximately 3% of adults with the disease. Weight management programs for this patient population stress implementing lifestyle changes, and only 1.1% of participants are prescribed medications.
According to the statement, the term “weight-loss medications” contributes to the concept that their use has an aesthetic goal and can be consumed by anyone who desires to lose weight.
In addition to ensuring the correct use of language, Dr. Mancini adds that it is essential for doctors to seek and present pharmacologic treatment for obesity as something that will improve patient health. This means stressing that obesity can be controlled with a 10% loss in body weight, just as other chronic diseases, such as diabetes, can be controlled. Moreover, it is important to point out that medications also have a crucial role in optimizing weight maintenance in the long term.
Another issue Dr. Mancini raised is the prejudice that many doctors have against people with obesity. Health professionals should recognize they are also subject to weight bias and that the way they communicate with patients could have a profound effect on health-related outcomes.
“The stigma surrounding obesity can lead to bullying, even in the patient’s home by their relatives; this is very common. Weight stigma is so strong that it hinders patient health and decreases the likelihood of the patient seeking specialized care,” Dr. Mancini warned.
According to the authors, it is of utmost importance to understand that an individual should not be defined by his or disease (as by the use of the terms “obese” or “diabetic”) but rather understood to live with this disease (“individual with obesity” or “with diabetes”). Dr. Mancini suggests the following strategies that health care professionals can adopt while caring for patients with obesity:
- Speak to patients with empathy and respect, avoiding the use of judgmental words.
- Ask if they would like to discuss the “weight issue,” “BMI issue,” terms that are better received by the public, instead of saying “excess fat” or “excess weight.”
- If the patient agrees to talk about the subject, reinforce that this is a chronic health problem that requires longterm treatment and give him or her short, medium, and longterm options.
Lastly, the authors highlighted the importance of differentiating between regulatory agency–approved medications and over-the-counter drugs and supplements that are often sold as “weight-loss agents” and are responsible for an unacceptably high rate of emergency visits.
This article was translated from the Medscape Portuguese Edition. A version appeared on Medscape.com.
Inadequate sleep & obesity: Breaking the vicious cycle
Sleep is fundamental to overall health and longevity, with the average person spending about one-third of their life sleeping.1 Adequate sleep is critical for optimal cognition, memory consolidation, mood regulation, metabolism, appetite regulation, and immune and hormone functioning. According to the American Academy of Sleep Medicine and the Sleep Research Society, adults should sleep at least 7 hours per night on a regular basis “to promote optimal health.”2 Yet, between 2013 and 2020, only about 65% of adults in the United States were meeting this amount.3 Insufficient sleep is associated with an increased risk for chronic health conditions, including obesity, diabetes, cardiovascular diseases, and even premature death.4

In a population-based longitudinal study of sleep disorders, short sleep duration was associated with increased body mass index (BMI), low blood levels of leptin, and high ghrelin levels.5 In addition to physical impairments, poor sleep can impair cognitive performance and lead to vehicular accidents and increased accidents at work.4 The potential economic impact that this may have is significant, and includes increased costs and loss of productivity in the workplace.6
Many factors may contribute to short sleep duration: environment, mental and physical condition, and social influences such as occupation, family responsibilities, travel, group activities, and personal care. Furthermore, the rapidly evolving and developing media, communication, and entertainment industries are already strongly implicated in poor sleep quality and quantity, both contributing to excessive daytime sleepiness.7 Poor sleep quality is most notable in modern societies, and it correlates with the increasing prevalence of obesity, likely due to sleep’s effect on food consumption and physical activity.8 Optimizing a person’s sleep will improve overall health and longevity by inhibiting the development of chronic disease.
How insufficient sleep raises the risk for obesity
Not only is sleep beneficial for brain health, memory, learning, and growth, its effect on food consumption and physical activity likely correlates with the increased prevalence of obesity in modern society. Yet the optimal amount of sleep is controversial, and current recommendations of 7 or more hours of sleep per night for adults are derived from expert panels only.2 The recommended sleep duration for children is longer, and it varies by age.9 The quality of sleep and its impact on neuroendocrine hormones, not just the quantity of sleep, needs to be factored into these recommendations.
Sleep restriction activates the orexigenic system via the hormones leptin and ghrelin. These hormones control the food reward system, essentially increasing hunger and food intake. Leptin, created by white adipose tissue, is responsible for satiety and decreased food consumption.10 Ghrelin, made by oxyntic glands in the stomach, is responsible for the sensation of hunger.
In a 2004 study by Spiegel et al,11 leptin and ghrelin levels were measured during 2 days of sleep restriction (4 hours in bed) and sleep extension (10 hours in bed). Sleep restriction was associated with a decrease in leptin levels and an increase in ghrelin levels. The researchers reported that participants experienced an increase in hunger and appetite—especially for calorie-dense foods with high carbohydrate content.
Although research design has limitations with predominantly self-reported sleep data, studies have shown that short sleep time leads to increased food intake by increasing hunger signals and craving of unhealthy foods, and by providing more opportunities to eat while awake. It also may lead to decreased physical activity, creating a sedentary lifestyle that further encourages obesity.8 Reduced sleep is even correlated to decreased efficacy of weight-loss treatments.12
Continue to: Other sleep characteristics weakly correlated with obesity
Other sleep characteristics weakly correlated with obesity are sleep variability, timing, efficiency, quality, and daytime napping.8 Sleep variability causes dysregulation of eating patterns, leading to increased food intake. A shift to later sleep and waking times often results in higher consumption of calories after 8
Poor sleep efficiency and quality decreases N3-stage (deep non-REM) sleep, affects the autonomic nervous system, and has been associated with increased abdominal obesity. Daytime napping, which can cause irregular circadian rhythms and sleep schedules, is associated with increased obesity.15 Thus, each component of sleep needs to be assessed to promote optimal regulation of the orexigenic system.
Another study showed that inadequate sleep not only promotes unhealthy lifestyle habits that can lead to obesity but also decreases the ability to lose weight.16 This small study with 10 overweight patients provided its subjects with a controlled caloric intake over 2 weeks. Patients spent two 14-day periods 3 months apart in the laboratory, divided into 2 time-in-bed arms of 8.5 and 5.5 hours per night. Neuroendocrine changes caused by decreased sleep were associated with a significant lean body mass loss while conserving energy-dense fat.16 This study highlights the importance of sleep hygiene counseling when developing a weight-management plan with patients.
Sleep, and its many components, play an integral role in the prevention and treatment of obesity.17 Poor sleep will increase the risk for obesity and hinder its treatment. Therefore, sleep quality and duration are vital components of obesity management.
The sleep–obesity link in children and the elderly
Childhood obesity is linked to several chronic diseases in adulthood, including type 2 diabetes, cardiovascular disease, nonalcoholic fatty liver disease, asthma, and obstructive sleep apnea (OSA).18 According to 2017-2018 NHANES (National Health and Nutrition Examination Surveys) data, obesity (BMI ≥ 95th percentile) prevalence among children and adolescents was reported at 19.3% and severe obesity (BMI ≥ 120% of the 95th percentile) at 6.1%. Pediatric overweight prevalence (≥ 85th percentile and < 95th percentile) was 16.1%.19
Continue to: Although poor sleep is associated...
Although poor sleep is associated with increased risk for obesity, there is no proven cause-effect relationship.20 Nutrition and physical activity have been identified as 2 critical factors in childhood obesity, but sleep health also needs to be investigated. Shorter sleep duration is strongly associated with the development of obesity. Furthermore, children with obesity are more likely to have shorter sleep duration.21 A short sleep duration alters plasma levels of insulin, low-density lipoprotein, and high-sensitivity C-reactive protein. It is associated with lower diet quality, an increased intake of nutrient-poor foods, and a lower intake of vegetables and fruits.22 Recent studies have shown that interventions to promote earlier bedtimes can improve sleep duration in children.
Older adults have many sleeping issues, including insomnia, circadian rhythm sleep-wake disorders, sleep-related movement disorders, and sleep-breathing disorders. Additionally, the older population has increased sleep latency, decreased sleep efficiency and total sleep time, decreased REM sleep, more frequent nighttime awakenings, and more daytime napping.23 The increased sleep disturbance with age is mainly related to higher risk factors for sleep disorders than the aging process itself. Sleeping 5 or fewer hours is associated with an increased risk for obesity and central abdominal fat compared with those who sleep 7 to 8 hours per night.24 Similar to children and youth, older adults also show a strong correlation between inadequate sleep and obesity.24
The consequence: A vicious cycle
Obesity in turn leads to shorter sleep duration and more disruptions. This negatively affects the orexigenic system, and the resulting hormonal derangement promotes worsening obesity. It is a cycle of poor sleep causing obesity and obesity causing poor sleep. Insomnia, in combination with shorter (and longer) sleep times, also has been linked with obesity.25 These patients experience more daytime sleepiness, fatigue, and nighttime sleep disturbances, all correlated with decreased quality of life and higher prevalence of medical comorbidities.8,26 Additional comorbidities secondary to obesity, including gastroesophageal reflux, depression, and asthma, also have been linked to sleep disturbances.8
OSA is a common sleep complication associated with obesity. With the increasing prevalence of obesity, the prevalence of OSA is rising.8,27 Factors that heighten the risk for OSA are male sex, age 40 to 70 years, postmenopausal status, elevated BMI, and craniofacial and upper airway abnormality.28 However, the US Preventive Services Task Force found insufficient evidence to screen for or treat OSA in asymptomatic adults.28 Signs and symptoms of OSA include nighttime awakenings with choking, loud snoring, and feeling unrefreshed after sleep.29
OSA is caused by the intermittent narrowing and obstruction of the pharyngeal airway due to anatomical and structural irregularities or neuromuscular impairments. Untreated OSA is associated with cardiovascular disease and cardiac arrhythmias such as atrial fibrillation. Even with this correlation between obesity and sleep, it is estimated that 80% of OSA remains undiagnosed.30 Approximately half of primary care clinicians do not screen at-risk patients for OSA, and 90% do not use validated OSA screening tools.31 Screening tools that have been validated are the STOP, STOP-BANG, Epworth Sleepiness Scale, and 4-Variable Screening Tool. However, the US Department of Veterans Affairs and the US Department of Defense have a more recent guideline recommending STOP as an easier-to-administer screen for OSA.32 A positive result with a screening tool should be confirmed with polysomnography.32
Continue to: Intervention for OSA
Intervention for OSA. The longest randomized controlled study to date, Sleep AHEAD, evaluated over a period of 10 years the effect of weight loss on OSA severity achieved with either an intensive lifestyle intervention (ILI) or with diabetes support and education (DSE).33 OSA severity is rated on an Apnea-Hypopnea Index (AHI), with scores reflecting the number of sleep apnea events per hour. This study demonstrated that weight loss was associated with decreased OSA severity. At 4-year follow-up, the greater the weight loss with ILI intervention, the lower the patients’ OSA severity scores. The study found an average decrease in AHI of 0.68 events per hour for every kilogram of weight loss in the ILI group (P < .0001).33,34 Over the follow-up visits, the ILI participants had 7.4 events per hour, a more significantly reduced AHI than the DSE participants (P < .0001).33,34
Additionally, a small cohort of study participants achieved OSA remission (ILI, 34.4%; DSE, 22.2%), indicated by a low AHI score (< 5 events per hour). At the conclusion of the study, OSA severity decreased to a greater degree with ILI intervention.33,34
Alcohol and drug use can negatively influence sleep patterns and obesity. Higher alcohol consumption is associated with poorer sleep quality and higher chances of developing short sleep duration and snoring.35 Alcohol, a muscle relaxant, causes upper airway narrowing and reduced tongue muscle tone, thereby increasing snoring and OSA as demonstrated by increased AHI on polysomnography after alcohol intake. Alcohol also changes sleep architecture by increasing slow-wave sleep, decreasing REM sleep duration, and increasing sleep arousal in the second half of the night.36 Disrupted circadian rhythm after alcohol consumption was correlated with increased adenosine neurotransmitters derived from ethanol metabolism.37 Alcohol dependence may be related to other psychiatric symptoms, and chronic alcohol use eventually alters sleep mechanisms leading to persistent insomnia, further perpetuating adverse outcomes such as suicidal ideation.36 There are positive associations between beer drinking and measures of abdominal adiposity in men, and “the combination of short sleep duration [and] disinhibited eating … is associated with greater alcohol intake and excess weight.”38
Therefore, counsel patients to avoid alcohol since it is a modifiable risk factor with pervasive adverse health effects.
Many drugs have a profound effect on sleep patterns. Illicit drug use in particular can affect the brain’s neurotransmitter serotonin system. For example, ecstasy users have an increased risk for OSA.39 People with cocaine and heroin use disorder tend to have more sleep-maintenance insomnia.40
Continue to: In contrast, those with alcohol...
In contrast, those with alcohol or cannabis use disorder tend to have more sleep-onset insomnia.40 Not only do illicit drugs interrupt sleep, but daily tobacco use also has been correlated with increased insomnia and shorter sleep duration since nicotine is a stimulant.41
Insomnia is commonly treated with sedative antidepressants and hypnotics—eg, mirtazapine and olanzapine—that contribute to weight gain.42 In addition, other common pharmaceuticals used for sleep disorders, such as diphenhydramine, have sedative properties and tend to lead to weight gain.43 Because so many medications affect sleep and weight, carefully review patients’ medication lists and switch offending agents to weight-neutral drugs if possible.
Treatment and tools to improve sleep in patients with obesity
Given the strong correlation between obesity and sleep disorders, validated screening tools should be used to assess sleep quality, including onset and potential symptoms associated with poor sleep (TABLE 144). For weight management to succeed in patients with obesity, it is crucial to address sleep in addition to nutrition and physical activity.17,45
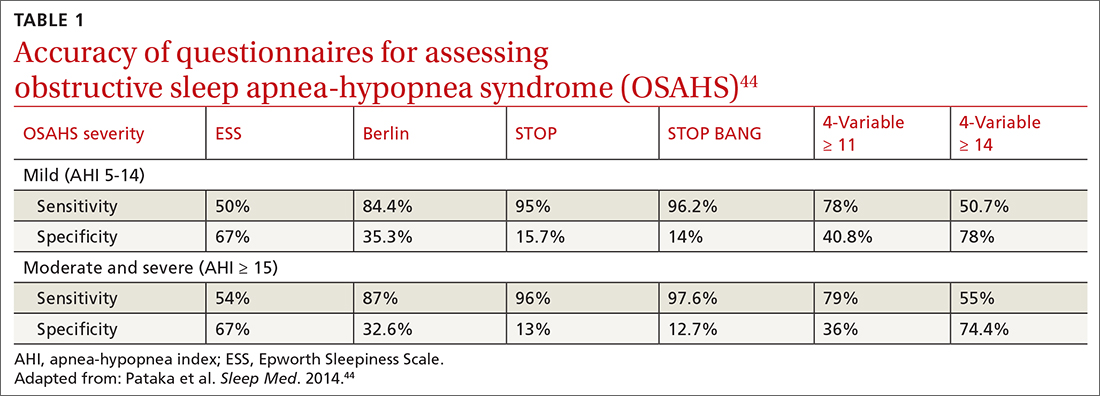
Physical activity has many benefits to overall health, especially for chronic diseases such as type 2 diabetes and hypertension. The Centers for Disease Control and Prevention recommends at least 150 minutes of moderate-intensity aerobic activity or 75 minutes of vigorous-intensity aerobic exercise per week in addition to muscle-strengthening activities 2 or more days per week.46 However, approximately 300 minutes of moderate-
Physical activity and diet in combination are vital, but diet restriction has a more substantial effect on weight loss than physical activity alone.48 Still, physical activity is essential in helping maintain and prevent weight regain.
Continue to: Nonpharmacologic interventions
Nonpharmacologic interventions include promoting greater sleep quality and quantity by emphasizing good sleep hygiene practices. Developing a practical and effective bedtime routine, creating a quiet sleep environment, and practicing healthy daily habits are essential components to sleep hygiene(TABLE 249,50). Relaxation techniques and cognitive behavioral therapy (CBT) also can help. CBT for insomnia (CBT-I) is the first-line intervention for chronic insomnia.51 Sleep restriction is a type of CBT used to treat insomnia, encouraging short-term sleep loss in the hopes of improving insomnia. A trial by Logue et al showed that patients with overweight and obesity randomized to undergo CBT with better sleep hygiene (nonpharmacologic) interventions had a greater mean weight loss percentage (5% vs 2%; P = .04) than did those who received CBT alone.52

Eastern medicine including herbal interventions lack evidence of efficacy and safety. Further studies need to be done on the effects that chamomile, kava, valerian root (Valeriana officinalis), tryptophan, and Wu Ling (from mycelia Xylaria nigripes) might have on sleep.53
Proceed cautiously with medication. The American College of Physicians recommends a shared decision-making approach when considering pharmacologic therapy for chronic insomnia and the American Academy of Sleep Medicine (AASM) offers guidance on options.51,54 However, the evidence behind AASM sleep pharmacologic recommendations is weak, implying a lesser degree of confidence in the outcome and, therefore, in its appropriateness. Thus, it falls upon the clinician and patient to weigh the benefits and burdens of the pharmacologic treatments of insomnia. If indicated, medications suggested to treat sleep onset and sleep maintenance insomnia are eszopiclone, zolpidem, and temazepam. Zaleplon, triazolam, and ramelteon may improve sleep initiation. Suvorexant and doxepin are used for sleep-maintenance insomnia.54 Exploring patient preferences, cost of treatment, health care options, and available resources should all be considered.
CORRESPONDENCE
Ecler Ercole Jaqua, MD, MBA, FAAFP, AGSF, FACLM, DipABOM, Loma Linda University Health, 25455 Barton Road, Suite 206A, Loma Linda, CA 92354; ejaqua@llu.edu
1. Aminoff MJ, Boller F, Swaab DF. We spend about one-third of our life either sleeping or attempting to do so. Handb Clin Neurol. 2011;98:vii. doi: 10.1016/B978-0-444-52006-7.00047-2
2. Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
3. CDC. Sleep and sleep disorders, adults. Accessed September 21, 2023. www.cdc.gov/sleep/data-and-statistics/adults.html
4. Chattu VK, Manzar MD, Kumary S. The global problem of insufficient sleep and its serious public health implications. Healthcare (Basel). 2019;7:1. doi: 10.3390/healthcare7010001
5. Taheri S, Lin L, Austin D, et al. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004;1:e62. doi: 10.1371/journal.pmed.0010062
6. Hafner M, Stepanek M, Taylor J, et al. Why sleep matters—the economic costs of insufficient sleep. Rand Health Q. 2017;6:11.
7. Hisler G, Twenge JM, Krizan Z. Associations between screen time and short sleep duration among adolescents varies by media type: evidence from a cohort study. Sleep Med. 2020;66:92-102. doi: 10.1016/j.sleep.2019.08.007
8. Ogilvie RP, Patel SR. The epidemiology of sleep and obesity. Sleep Health. 2017;3:383-388. doi: 10.1016/j.sleh.2017.07.013
9. CDC. Sleep and sleep disorders: How much sleep do I need? Accessed September 21, 2023. www.cdc.gov/sleep/about_sleep/how_much_sleep.html
10. van Egmond LT, Meth EMS, Engström J, et al. Effects of acute sleep loss on leptin, ghrelin, and adiponectin in adults with healthy weight and obesity: a laboratory study. Obesity (Silver Spring). 2023;31:635-641. doi: 10.1002/oby.23616
11. Spiegel K, Tasali E, Penev P, et al. Brief communication: sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann Intern Med. 2004;141:846-850. doi: 10.7326/0003-4819-141-11-200412070-00008
12. Antza C, Kostopoulos G, Mostafa S, et al. The links between sleep duration, obesity and type 2 diabetes mellitus. J Endocrinol. 2021;252:125-141. doi: 10.1530/JOE-21-0155
13. Baron KG, Reid KJ, Kern AS, et al. Role of sleep timing in caloric intake and BMI. Obesity (Silver Spring). 2011;19:1374-1381. doi: 10.1038/oby.2011.100
14. Liu XY, Zheng CL, Xu C, et al. Nighttime snacking is associated with risk of obesity and hyperglycemia in adults: a cross-sectional survey from Chinese adult teachers J Biomed Res. 2017;31:541-547. doi: 10.7555/JBR.31.20160083
15. Cai Z, Yang Y, Zhang J, et al. The relationship between daytime napping and obesity: a systematic review and meta-analysis. Sci Rep. 2023.13:12124. doi: 10.1038/s41598-023-37883-7
16. Nedeltcheva AV, Kilkus JM, Imperial J, et al. Insufficient sleep undermines dietary efforts to reduce adiposity. Ann Intern Med. 2010;153:435-441. doi: 10.7326/0003-4819-153-7-201010050-00006
17. Chaput JP, Tremblay A. Adequate sleep to improve the treatment of obesity. CMAJ. 2012;184:1975-1976. doi: 10.1503/cmaj.120876
18. Kelsey MM, Zaepfel A, Bjornstad P, et al. Age-related consequences of childhood obesity. Gerontology. 2014;60:222-228. doi: 10.1159/000356023
19. Fryar CD, Carroll MD, Afful J. Prevalence of overweight, obesity, and severe obesity among children and adolescents aged 2-19 years: United States, 1963-1965 through 2017-2018. National Center for Health Statistics Health E-Stats. Updated January 29, 2021. Accessed September 21, 2021. www.cdc.gov/nchs/data/hestat/obesity-child-17-18/overweight-obesity-child-H.pdf
20. Fatima Y, Doi SAR, Mamun AA. Sleep quality and obesity in young subjects: a meta-analysis. Obes Rev. 2016;17:1154-1166. doi: 10.1111/obr.12444
21. Gohil A, Hannon TS. Poor sleep and obesity: concurrent epidemics in adolescent youth. Front Endocrinol. 2018;9:364. doi: 10.3389/fendo.2018.00364
22. Golley RK, Maher CA, Matricciani L, et al. Sleep duration or bedtime? Exploring the association between sleep timing behaviour, diet and BMI in children and adolescents. Int J Obes (Lond). 2013;37:546-551. doi: 10.1038/ijo.2012.212
23. Alessi CA. Sleep issues. In: Harper GM, Lyons WL, Potter JF, eds. Geriatrics Review Syllabus (GRS 10). Updated January 2021. Accessed August 29, 2023. http://geriatricscareonline.org
24. Patel SR, Blackwell T, Redline S, et al. The association between sleep duration and obesity in older adults. Int J Obes (Lond). 2008;32:1825-1834. doi: 10.1038/ijo.2008.198
25. Cai GH, Theorell-Haglöw J, Janson C, et al. Insomnia symptoms and sleep duration and their combined effects in relation to associations with obesity and central obesity. Sleep Med. 2018;46:81-87. doi: 10.1016/j.sleep.2018.03.009
26. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011;14:402-412. doi: 10.1097/MCO.0b013 e3283479109
27. Franklin KA, Lindberg E. Obstructive sleep apnea is a common disorder in the population–a review on the epidemiology of sleep apnea. J Thorac Dis. 2015;7:1311-1322. doi: 10.3978/j.issn.2072-1439.2015.06.11
28. USPSTF. Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for obstructive sleep apnea in adults: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:407-414. doi: 10.1001/jama.2016.20325
29. Goyal M, Johnson J. Obstructive sleep apnea diagnosis and management. Mo Med. 2017;114:120-124.
30. American Academy of Sleep Medicine. Hidden health crisis costing America billions: underdiagnosing and undertreating obstructive sleep apnea draining healthcare system. 2016. Accessed September 25, 2023. https://aasm.org/wp-content/uploads/2017/10/sleep-apnea-economic-crisis.pdf
31. Devaraj, NK. Knowledge, attitude, and practice regarding obstructive sleep apnea among primary care physicians. Sleep Breath. 2020;24:1581-1590. doi: 10.1007/s11325-020-02040-1
32. Mysliwiec V, Martin JL, Ulmer CS, et al. The management of chronic insomnia disorder and obstructive sleep apnea: synopsis of the 2019 U.S. Department of Veterans Affairs and U.S. Department of Defense Clinical Practice Guidelines. Ann Intern Med. 2020;172:325-336. doi: 10.7326/M19-3575
33. Kuna ST, Reboussin DM, Strotmeyer ES, et al. Effects of weight loss on obstructive sleep apnea severity. Ten-year results of the Sleep AHEAD study. Am J Respir Crit Care Med. 2021;203:221-229. doi: 10.1164/rccm.201912-2511OC
34. St-Onge MP, Tasali E. Weight loss is integral to obstructive sleep apnea management. Ten-year follow-up in Sleep AHEAD. Am J Respir Crit Care Med. 2021;203:161-162. doi: 10.1164/rccm.202007-2906ED
35. Zheng D, Yuan X, Ma C, et al. Alcohol consumption and sleep quality: a community-based study. Public Health Nutr. 2021;24:4851-4858. doi: 10.1017/S1368980020004553
36. Chakravorty S, Chaudhary NS, Brower KJ. Alcohol dependence and its relationship with insomnia and other sleep disorders. Alcohol Clin Exp Res. 2016;40:2271-2282. doi: 10.1111/acer.13217
37. Elmenhorst EM, Elmenhorst D, Benderoth S, et al. Cognitive impairments by alcohol and sleep deprivation indicate trait characteristics and a potential role for adenosine A1 receptors. Proc Natl Acad Sci U S A. 2018;115:8009-8014. doi: 10.1073/pnas.1803770115
38. Traversy G, Chaput JP. Alcohol consumption and obesity: an update. Curr Obes Rep. 2015;4:122-130. doi: 10.1007/s13679-014-0129-4
39. McCann UD, Sgambati FP, Schwartz AR, et al. Sleep apnea in young abstinent recreational MDMA (“ecstasy”) consumers. Neurology. 2009;73:2011-2017. doi: 10.1212/WNL.0b013e3181c51a62
40. Grau-López L, Grau-López L, Daigre C, et al. Insomnia symptoms in patients with substance use disorders during detoxification and associated clinical features. Front Psychiatry. 2020;11:540022. doi: 10.3389/fpsyt.2020.540022
41. Boehm MA, Lei QM, Lloyd RM, et al. Depression, anxiety, and tobacco use: overlapping impediments to sleep in a national sample of college students. J Am Coll Health. 2016;64:565-574. doi: 10.1080/07448481.2016.1205073
42. Gracious BL, Meyer AE. Psychotropic-induced weight gain and potential pharmacologic treatment strategies. Psychiatry (Edgmont). 2005;2:36-42.
43. Ratliff JC, Barber JA, Palmese LB, et al. Association of prescription H1 antihistamine use with obesity: results from the National Health and Nutrition Examination Survey. Obesity (Silver Spring). 2010;18:2398-2400. doi: 10.1038/oby.2010.176
44. Pataka A, Daskalopoulou E, Kalamaras G, et al. Evaluation of five different questionnaires for assessing sleep apnea syndrome in a sleep clinic. Sleep Med. 2014;15:776-781. doi: 10.1016/j.sleep.2014.03.012
45. Kline CE, Chasens ER, Bizhanova Z, et al. The association between sleep health and weight change during a 12-month behavioral weight loss intervention. Int J Obes (Lond). 2021;45:639-649. doi: 10.1038/s41366-020-00728-8
46. CDC. How much physical activity do adults need? Accessed August 23, 2023. www.cdc.gov/physicalactivity/basics/adults/index.htm
47. Flack KD, Hays HM, Moreland J, et al. Exercise for weight loss: further evaluating energy compensation with exercise. Med Sci Sports Exerc. 2020;52:2466-2475. doi: 10.1249/MSS.0000000000002376
48. Swift DL, Johannsen NM, Lavie CJ, et al. The role of exercise and physical activity in weight loss and maintenance. Prog Cardiovasc Dis. 2014;56:441-447. doi: 10.1016/j.pcad.2013.09.012
49. Irish LA, Kline CE, Gunn HE, et al. The role of sleep hygiene in promoting public health: a review of empirical evidence. Sleep Med Rev. 2015;22:23-36. doi: 10.1016/j.smrv.2014.10.001
50. CDC. Tips for better sleep. 2022. Accessed August 4, 2023. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html
51. Qaseem A, Kansagara D, Forciea MA, et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165:125-133. doi: 10.7326/M15-2175
52. Logue EE, Bourguet CC, Palmieri PA, et al. The better weight-better sleep study: a pilot intervention in primary care. Am J Health Behav. 2012;36:319-334. doi: 10.5993/AJHB.36.3.4
53. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12. doi: 10.1016/j.smrv.2014.12.003
54. Sateia MJ, Buysse DJ, Krystal AD, et al. Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017;13:307-349. doi: 10.5664/jcsm.6470
Sleep is fundamental to overall health and longevity, with the average person spending about one-third of their life sleeping.1 Adequate sleep is critical for optimal cognition, memory consolidation, mood regulation, metabolism, appetite regulation, and immune and hormone functioning. According to the American Academy of Sleep Medicine and the Sleep Research Society, adults should sleep at least 7 hours per night on a regular basis “to promote optimal health.”2 Yet, between 2013 and 2020, only about 65% of adults in the United States were meeting this amount.3 Insufficient sleep is associated with an increased risk for chronic health conditions, including obesity, diabetes, cardiovascular diseases, and even premature death.4

In a population-based longitudinal study of sleep disorders, short sleep duration was associated with increased body mass index (BMI), low blood levels of leptin, and high ghrelin levels.5 In addition to physical impairments, poor sleep can impair cognitive performance and lead to vehicular accidents and increased accidents at work.4 The potential economic impact that this may have is significant, and includes increased costs and loss of productivity in the workplace.6
Many factors may contribute to short sleep duration: environment, mental and physical condition, and social influences such as occupation, family responsibilities, travel, group activities, and personal care. Furthermore, the rapidly evolving and developing media, communication, and entertainment industries are already strongly implicated in poor sleep quality and quantity, both contributing to excessive daytime sleepiness.7 Poor sleep quality is most notable in modern societies, and it correlates with the increasing prevalence of obesity, likely due to sleep’s effect on food consumption and physical activity.8 Optimizing a person’s sleep will improve overall health and longevity by inhibiting the development of chronic disease.
How insufficient sleep raises the risk for obesity
Not only is sleep beneficial for brain health, memory, learning, and growth, its effect on food consumption and physical activity likely correlates with the increased prevalence of obesity in modern society. Yet the optimal amount of sleep is controversial, and current recommendations of 7 or more hours of sleep per night for adults are derived from expert panels only.2 The recommended sleep duration for children is longer, and it varies by age.9 The quality of sleep and its impact on neuroendocrine hormones, not just the quantity of sleep, needs to be factored into these recommendations.
Sleep restriction activates the orexigenic system via the hormones leptin and ghrelin. These hormones control the food reward system, essentially increasing hunger and food intake. Leptin, created by white adipose tissue, is responsible for satiety and decreased food consumption.10 Ghrelin, made by oxyntic glands in the stomach, is responsible for the sensation of hunger.
In a 2004 study by Spiegel et al,11 leptin and ghrelin levels were measured during 2 days of sleep restriction (4 hours in bed) and sleep extension (10 hours in bed). Sleep restriction was associated with a decrease in leptin levels and an increase in ghrelin levels. The researchers reported that participants experienced an increase in hunger and appetite—especially for calorie-dense foods with high carbohydrate content.
Although research design has limitations with predominantly self-reported sleep data, studies have shown that short sleep time leads to increased food intake by increasing hunger signals and craving of unhealthy foods, and by providing more opportunities to eat while awake. It also may lead to decreased physical activity, creating a sedentary lifestyle that further encourages obesity.8 Reduced sleep is even correlated to decreased efficacy of weight-loss treatments.12
Continue to: Other sleep characteristics weakly correlated with obesity
Other sleep characteristics weakly correlated with obesity are sleep variability, timing, efficiency, quality, and daytime napping.8 Sleep variability causes dysregulation of eating patterns, leading to increased food intake. A shift to later sleep and waking times often results in higher consumption of calories after 8
Poor sleep efficiency and quality decreases N3-stage (deep non-REM) sleep, affects the autonomic nervous system, and has been associated with increased abdominal obesity. Daytime napping, which can cause irregular circadian rhythms and sleep schedules, is associated with increased obesity.15 Thus, each component of sleep needs to be assessed to promote optimal regulation of the orexigenic system.
Another study showed that inadequate sleep not only promotes unhealthy lifestyle habits that can lead to obesity but also decreases the ability to lose weight.16 This small study with 10 overweight patients provided its subjects with a controlled caloric intake over 2 weeks. Patients spent two 14-day periods 3 months apart in the laboratory, divided into 2 time-in-bed arms of 8.5 and 5.5 hours per night. Neuroendocrine changes caused by decreased sleep were associated with a significant lean body mass loss while conserving energy-dense fat.16 This study highlights the importance of sleep hygiene counseling when developing a weight-management plan with patients.
Sleep, and its many components, play an integral role in the prevention and treatment of obesity.17 Poor sleep will increase the risk for obesity and hinder its treatment. Therefore, sleep quality and duration are vital components of obesity management.
The sleep–obesity link in children and the elderly
Childhood obesity is linked to several chronic diseases in adulthood, including type 2 diabetes, cardiovascular disease, nonalcoholic fatty liver disease, asthma, and obstructive sleep apnea (OSA).18 According to 2017-2018 NHANES (National Health and Nutrition Examination Surveys) data, obesity (BMI ≥ 95th percentile) prevalence among children and adolescents was reported at 19.3% and severe obesity (BMI ≥ 120% of the 95th percentile) at 6.1%. Pediatric overweight prevalence (≥ 85th percentile and < 95th percentile) was 16.1%.19
Continue to: Although poor sleep is associated...
Although poor sleep is associated with increased risk for obesity, there is no proven cause-effect relationship.20 Nutrition and physical activity have been identified as 2 critical factors in childhood obesity, but sleep health also needs to be investigated. Shorter sleep duration is strongly associated with the development of obesity. Furthermore, children with obesity are more likely to have shorter sleep duration.21 A short sleep duration alters plasma levels of insulin, low-density lipoprotein, and high-sensitivity C-reactive protein. It is associated with lower diet quality, an increased intake of nutrient-poor foods, and a lower intake of vegetables and fruits.22 Recent studies have shown that interventions to promote earlier bedtimes can improve sleep duration in children.
Older adults have many sleeping issues, including insomnia, circadian rhythm sleep-wake disorders, sleep-related movement disorders, and sleep-breathing disorders. Additionally, the older population has increased sleep latency, decreased sleep efficiency and total sleep time, decreased REM sleep, more frequent nighttime awakenings, and more daytime napping.23 The increased sleep disturbance with age is mainly related to higher risk factors for sleep disorders than the aging process itself. Sleeping 5 or fewer hours is associated with an increased risk for obesity and central abdominal fat compared with those who sleep 7 to 8 hours per night.24 Similar to children and youth, older adults also show a strong correlation between inadequate sleep and obesity.24
The consequence: A vicious cycle
Obesity in turn leads to shorter sleep duration and more disruptions. This negatively affects the orexigenic system, and the resulting hormonal derangement promotes worsening obesity. It is a cycle of poor sleep causing obesity and obesity causing poor sleep. Insomnia, in combination with shorter (and longer) sleep times, also has been linked with obesity.25 These patients experience more daytime sleepiness, fatigue, and nighttime sleep disturbances, all correlated with decreased quality of life and higher prevalence of medical comorbidities.8,26 Additional comorbidities secondary to obesity, including gastroesophageal reflux, depression, and asthma, also have been linked to sleep disturbances.8
OSA is a common sleep complication associated with obesity. With the increasing prevalence of obesity, the prevalence of OSA is rising.8,27 Factors that heighten the risk for OSA are male sex, age 40 to 70 years, postmenopausal status, elevated BMI, and craniofacial and upper airway abnormality.28 However, the US Preventive Services Task Force found insufficient evidence to screen for or treat OSA in asymptomatic adults.28 Signs and symptoms of OSA include nighttime awakenings with choking, loud snoring, and feeling unrefreshed after sleep.29
OSA is caused by the intermittent narrowing and obstruction of the pharyngeal airway due to anatomical and structural irregularities or neuromuscular impairments. Untreated OSA is associated with cardiovascular disease and cardiac arrhythmias such as atrial fibrillation. Even with this correlation between obesity and sleep, it is estimated that 80% of OSA remains undiagnosed.30 Approximately half of primary care clinicians do not screen at-risk patients for OSA, and 90% do not use validated OSA screening tools.31 Screening tools that have been validated are the STOP, STOP-BANG, Epworth Sleepiness Scale, and 4-Variable Screening Tool. However, the US Department of Veterans Affairs and the US Department of Defense have a more recent guideline recommending STOP as an easier-to-administer screen for OSA.32 A positive result with a screening tool should be confirmed with polysomnography.32
Continue to: Intervention for OSA
Intervention for OSA. The longest randomized controlled study to date, Sleep AHEAD, evaluated over a period of 10 years the effect of weight loss on OSA severity achieved with either an intensive lifestyle intervention (ILI) or with diabetes support and education (DSE).33 OSA severity is rated on an Apnea-Hypopnea Index (AHI), with scores reflecting the number of sleep apnea events per hour. This study demonstrated that weight loss was associated with decreased OSA severity. At 4-year follow-up, the greater the weight loss with ILI intervention, the lower the patients’ OSA severity scores. The study found an average decrease in AHI of 0.68 events per hour for every kilogram of weight loss in the ILI group (P < .0001).33,34 Over the follow-up visits, the ILI participants had 7.4 events per hour, a more significantly reduced AHI than the DSE participants (P < .0001).33,34
Additionally, a small cohort of study participants achieved OSA remission (ILI, 34.4%; DSE, 22.2%), indicated by a low AHI score (< 5 events per hour). At the conclusion of the study, OSA severity decreased to a greater degree with ILI intervention.33,34
Alcohol and drug use can negatively influence sleep patterns and obesity. Higher alcohol consumption is associated with poorer sleep quality and higher chances of developing short sleep duration and snoring.35 Alcohol, a muscle relaxant, causes upper airway narrowing and reduced tongue muscle tone, thereby increasing snoring and OSA as demonstrated by increased AHI on polysomnography after alcohol intake. Alcohol also changes sleep architecture by increasing slow-wave sleep, decreasing REM sleep duration, and increasing sleep arousal in the second half of the night.36 Disrupted circadian rhythm after alcohol consumption was correlated with increased adenosine neurotransmitters derived from ethanol metabolism.37 Alcohol dependence may be related to other psychiatric symptoms, and chronic alcohol use eventually alters sleep mechanisms leading to persistent insomnia, further perpetuating adverse outcomes such as suicidal ideation.36 There are positive associations between beer drinking and measures of abdominal adiposity in men, and “the combination of short sleep duration [and] disinhibited eating … is associated with greater alcohol intake and excess weight.”38
Therefore, counsel patients to avoid alcohol since it is a modifiable risk factor with pervasive adverse health effects.
Many drugs have a profound effect on sleep patterns. Illicit drug use in particular can affect the brain’s neurotransmitter serotonin system. For example, ecstasy users have an increased risk for OSA.39 People with cocaine and heroin use disorder tend to have more sleep-maintenance insomnia.40
Continue to: In contrast, those with alcohol...
In contrast, those with alcohol or cannabis use disorder tend to have more sleep-onset insomnia.40 Not only do illicit drugs interrupt sleep, but daily tobacco use also has been correlated with increased insomnia and shorter sleep duration since nicotine is a stimulant.41
Insomnia is commonly treated with sedative antidepressants and hypnotics—eg, mirtazapine and olanzapine—that contribute to weight gain.42 In addition, other common pharmaceuticals used for sleep disorders, such as diphenhydramine, have sedative properties and tend to lead to weight gain.43 Because so many medications affect sleep and weight, carefully review patients’ medication lists and switch offending agents to weight-neutral drugs if possible.
Treatment and tools to improve sleep in patients with obesity
Given the strong correlation between obesity and sleep disorders, validated screening tools should be used to assess sleep quality, including onset and potential symptoms associated with poor sleep (TABLE 144). For weight management to succeed in patients with obesity, it is crucial to address sleep in addition to nutrition and physical activity.17,45

Physical activity has many benefits to overall health, especially for chronic diseases such as type 2 diabetes and hypertension. The Centers for Disease Control and Prevention recommends at least 150 minutes of moderate-intensity aerobic activity or 75 minutes of vigorous-intensity aerobic exercise per week in addition to muscle-strengthening activities 2 or more days per week.46 However, approximately 300 minutes of moderate-
Physical activity and diet in combination are vital, but diet restriction has a more substantial effect on weight loss than physical activity alone.48 Still, physical activity is essential in helping maintain and prevent weight regain.
Continue to: Nonpharmacologic interventions
Nonpharmacologic interventions include promoting greater sleep quality and quantity by emphasizing good sleep hygiene practices. Developing a practical and effective bedtime routine, creating a quiet sleep environment, and practicing healthy daily habits are essential components to sleep hygiene(TABLE 249,50). Relaxation techniques and cognitive behavioral therapy (CBT) also can help. CBT for insomnia (CBT-I) is the first-line intervention for chronic insomnia.51 Sleep restriction is a type of CBT used to treat insomnia, encouraging short-term sleep loss in the hopes of improving insomnia. A trial by Logue et al showed that patients with overweight and obesity randomized to undergo CBT with better sleep hygiene (nonpharmacologic) interventions had a greater mean weight loss percentage (5% vs 2%; P = .04) than did those who received CBT alone.52

Eastern medicine including herbal interventions lack evidence of efficacy and safety. Further studies need to be done on the effects that chamomile, kava, valerian root (Valeriana officinalis), tryptophan, and Wu Ling (from mycelia Xylaria nigripes) might have on sleep.53
Proceed cautiously with medication. The American College of Physicians recommends a shared decision-making approach when considering pharmacologic therapy for chronic insomnia and the American Academy of Sleep Medicine (AASM) offers guidance on options.51,54 However, the evidence behind AASM sleep pharmacologic recommendations is weak, implying a lesser degree of confidence in the outcome and, therefore, in its appropriateness. Thus, it falls upon the clinician and patient to weigh the benefits and burdens of the pharmacologic treatments of insomnia. If indicated, medications suggested to treat sleep onset and sleep maintenance insomnia are eszopiclone, zolpidem, and temazepam. Zaleplon, triazolam, and ramelteon may improve sleep initiation. Suvorexant and doxepin are used for sleep-maintenance insomnia.54 Exploring patient preferences, cost of treatment, health care options, and available resources should all be considered.
CORRESPONDENCE
Ecler Ercole Jaqua, MD, MBA, FAAFP, AGSF, FACLM, DipABOM, Loma Linda University Health, 25455 Barton Road, Suite 206A, Loma Linda, CA 92354; ejaqua@llu.edu
Sleep is fundamental to overall health and longevity, with the average person spending about one-third of their life sleeping.1 Adequate sleep is critical for optimal cognition, memory consolidation, mood regulation, metabolism, appetite regulation, and immune and hormone functioning. According to the American Academy of Sleep Medicine and the Sleep Research Society, adults should sleep at least 7 hours per night on a regular basis “to promote optimal health.”2 Yet, between 2013 and 2020, only about 65% of adults in the United States were meeting this amount.3 Insufficient sleep is associated with an increased risk for chronic health conditions, including obesity, diabetes, cardiovascular diseases, and even premature death.4

In a population-based longitudinal study of sleep disorders, short sleep duration was associated with increased body mass index (BMI), low blood levels of leptin, and high ghrelin levels.5 In addition to physical impairments, poor sleep can impair cognitive performance and lead to vehicular accidents and increased accidents at work.4 The potential economic impact that this may have is significant, and includes increased costs and loss of productivity in the workplace.6
Many factors may contribute to short sleep duration: environment, mental and physical condition, and social influences such as occupation, family responsibilities, travel, group activities, and personal care. Furthermore, the rapidly evolving and developing media, communication, and entertainment industries are already strongly implicated in poor sleep quality and quantity, both contributing to excessive daytime sleepiness.7 Poor sleep quality is most notable in modern societies, and it correlates with the increasing prevalence of obesity, likely due to sleep’s effect on food consumption and physical activity.8 Optimizing a person’s sleep will improve overall health and longevity by inhibiting the development of chronic disease.
How insufficient sleep raises the risk for obesity
Not only is sleep beneficial for brain health, memory, learning, and growth, its effect on food consumption and physical activity likely correlates with the increased prevalence of obesity in modern society. Yet the optimal amount of sleep is controversial, and current recommendations of 7 or more hours of sleep per night for adults are derived from expert panels only.2 The recommended sleep duration for children is longer, and it varies by age.9 The quality of sleep and its impact on neuroendocrine hormones, not just the quantity of sleep, needs to be factored into these recommendations.
Sleep restriction activates the orexigenic system via the hormones leptin and ghrelin. These hormones control the food reward system, essentially increasing hunger and food intake. Leptin, created by white adipose tissue, is responsible for satiety and decreased food consumption.10 Ghrelin, made by oxyntic glands in the stomach, is responsible for the sensation of hunger.
In a 2004 study by Spiegel et al,11 leptin and ghrelin levels were measured during 2 days of sleep restriction (4 hours in bed) and sleep extension (10 hours in bed). Sleep restriction was associated with a decrease in leptin levels and an increase in ghrelin levels. The researchers reported that participants experienced an increase in hunger and appetite—especially for calorie-dense foods with high carbohydrate content.
Although research design has limitations with predominantly self-reported sleep data, studies have shown that short sleep time leads to increased food intake by increasing hunger signals and craving of unhealthy foods, and by providing more opportunities to eat while awake. It also may lead to decreased physical activity, creating a sedentary lifestyle that further encourages obesity.8 Reduced sleep is even correlated to decreased efficacy of weight-loss treatments.12
Continue to: Other sleep characteristics weakly correlated with obesity
Other sleep characteristics weakly correlated with obesity are sleep variability, timing, efficiency, quality, and daytime napping.8 Sleep variability causes dysregulation of eating patterns, leading to increased food intake. A shift to later sleep and waking times often results in higher consumption of calories after 8
Poor sleep efficiency and quality decreases N3-stage (deep non-REM) sleep, affects the autonomic nervous system, and has been associated with increased abdominal obesity. Daytime napping, which can cause irregular circadian rhythms and sleep schedules, is associated with increased obesity.15 Thus, each component of sleep needs to be assessed to promote optimal regulation of the orexigenic system.
Another study showed that inadequate sleep not only promotes unhealthy lifestyle habits that can lead to obesity but also decreases the ability to lose weight.16 This small study with 10 overweight patients provided its subjects with a controlled caloric intake over 2 weeks. Patients spent two 14-day periods 3 months apart in the laboratory, divided into 2 time-in-bed arms of 8.5 and 5.5 hours per night. Neuroendocrine changes caused by decreased sleep were associated with a significant lean body mass loss while conserving energy-dense fat.16 This study highlights the importance of sleep hygiene counseling when developing a weight-management plan with patients.
Sleep, and its many components, play an integral role in the prevention and treatment of obesity.17 Poor sleep will increase the risk for obesity and hinder its treatment. Therefore, sleep quality and duration are vital components of obesity management.
The sleep–obesity link in children and the elderly
Childhood obesity is linked to several chronic diseases in adulthood, including type 2 diabetes, cardiovascular disease, nonalcoholic fatty liver disease, asthma, and obstructive sleep apnea (OSA).18 According to 2017-2018 NHANES (National Health and Nutrition Examination Surveys) data, obesity (BMI ≥ 95th percentile) prevalence among children and adolescents was reported at 19.3% and severe obesity (BMI ≥ 120% of the 95th percentile) at 6.1%. Pediatric overweight prevalence (≥ 85th percentile and < 95th percentile) was 16.1%.19
Continue to: Although poor sleep is associated...
Although poor sleep is associated with increased risk for obesity, there is no proven cause-effect relationship.20 Nutrition and physical activity have been identified as 2 critical factors in childhood obesity, but sleep health also needs to be investigated. Shorter sleep duration is strongly associated with the development of obesity. Furthermore, children with obesity are more likely to have shorter sleep duration.21 A short sleep duration alters plasma levels of insulin, low-density lipoprotein, and high-sensitivity C-reactive protein. It is associated with lower diet quality, an increased intake of nutrient-poor foods, and a lower intake of vegetables and fruits.22 Recent studies have shown that interventions to promote earlier bedtimes can improve sleep duration in children.
Older adults have many sleeping issues, including insomnia, circadian rhythm sleep-wake disorders, sleep-related movement disorders, and sleep-breathing disorders. Additionally, the older population has increased sleep latency, decreased sleep efficiency and total sleep time, decreased REM sleep, more frequent nighttime awakenings, and more daytime napping.23 The increased sleep disturbance with age is mainly related to higher risk factors for sleep disorders than the aging process itself. Sleeping 5 or fewer hours is associated with an increased risk for obesity and central abdominal fat compared with those who sleep 7 to 8 hours per night.24 Similar to children and youth, older adults also show a strong correlation between inadequate sleep and obesity.24
The consequence: A vicious cycle
Obesity in turn leads to shorter sleep duration and more disruptions. This negatively affects the orexigenic system, and the resulting hormonal derangement promotes worsening obesity. It is a cycle of poor sleep causing obesity and obesity causing poor sleep. Insomnia, in combination with shorter (and longer) sleep times, also has been linked with obesity.25 These patients experience more daytime sleepiness, fatigue, and nighttime sleep disturbances, all correlated with decreased quality of life and higher prevalence of medical comorbidities.8,26 Additional comorbidities secondary to obesity, including gastroesophageal reflux, depression, and asthma, also have been linked to sleep disturbances.8
OSA is a common sleep complication associated with obesity. With the increasing prevalence of obesity, the prevalence of OSA is rising.8,27 Factors that heighten the risk for OSA are male sex, age 40 to 70 years, postmenopausal status, elevated BMI, and craniofacial and upper airway abnormality.28 However, the US Preventive Services Task Force found insufficient evidence to screen for or treat OSA in asymptomatic adults.28 Signs and symptoms of OSA include nighttime awakenings with choking, loud snoring, and feeling unrefreshed after sleep.29
OSA is caused by the intermittent narrowing and obstruction of the pharyngeal airway due to anatomical and structural irregularities or neuromuscular impairments. Untreated OSA is associated with cardiovascular disease and cardiac arrhythmias such as atrial fibrillation. Even with this correlation between obesity and sleep, it is estimated that 80% of OSA remains undiagnosed.30 Approximately half of primary care clinicians do not screen at-risk patients for OSA, and 90% do not use validated OSA screening tools.31 Screening tools that have been validated are the STOP, STOP-BANG, Epworth Sleepiness Scale, and 4-Variable Screening Tool. However, the US Department of Veterans Affairs and the US Department of Defense have a more recent guideline recommending STOP as an easier-to-administer screen for OSA.32 A positive result with a screening tool should be confirmed with polysomnography.32
Continue to: Intervention for OSA
Intervention for OSA. The longest randomized controlled study to date, Sleep AHEAD, evaluated over a period of 10 years the effect of weight loss on OSA severity achieved with either an intensive lifestyle intervention (ILI) or with diabetes support and education (DSE).33 OSA severity is rated on an Apnea-Hypopnea Index (AHI), with scores reflecting the number of sleep apnea events per hour. This study demonstrated that weight loss was associated with decreased OSA severity. At 4-year follow-up, the greater the weight loss with ILI intervention, the lower the patients’ OSA severity scores. The study found an average decrease in AHI of 0.68 events per hour for every kilogram of weight loss in the ILI group (P < .0001).33,34 Over the follow-up visits, the ILI participants had 7.4 events per hour, a more significantly reduced AHI than the DSE participants (P < .0001).33,34
Additionally, a small cohort of study participants achieved OSA remission (ILI, 34.4%; DSE, 22.2%), indicated by a low AHI score (< 5 events per hour). At the conclusion of the study, OSA severity decreased to a greater degree with ILI intervention.33,34
Alcohol and drug use can negatively influence sleep patterns and obesity. Higher alcohol consumption is associated with poorer sleep quality and higher chances of developing short sleep duration and snoring.35 Alcohol, a muscle relaxant, causes upper airway narrowing and reduced tongue muscle tone, thereby increasing snoring and OSA as demonstrated by increased AHI on polysomnography after alcohol intake. Alcohol also changes sleep architecture by increasing slow-wave sleep, decreasing REM sleep duration, and increasing sleep arousal in the second half of the night.36 Disrupted circadian rhythm after alcohol consumption was correlated with increased adenosine neurotransmitters derived from ethanol metabolism.37 Alcohol dependence may be related to other psychiatric symptoms, and chronic alcohol use eventually alters sleep mechanisms leading to persistent insomnia, further perpetuating adverse outcomes such as suicidal ideation.36 There are positive associations between beer drinking and measures of abdominal adiposity in men, and “the combination of short sleep duration [and] disinhibited eating … is associated with greater alcohol intake and excess weight.”38
Therefore, counsel patients to avoid alcohol since it is a modifiable risk factor with pervasive adverse health effects.
Many drugs have a profound effect on sleep patterns. Illicit drug use in particular can affect the brain’s neurotransmitter serotonin system. For example, ecstasy users have an increased risk for OSA.39 People with cocaine and heroin use disorder tend to have more sleep-maintenance insomnia.40
Continue to: In contrast, those with alcohol...
In contrast, those with alcohol or cannabis use disorder tend to have more sleep-onset insomnia.40 Not only do illicit drugs interrupt sleep, but daily tobacco use also has been correlated with increased insomnia and shorter sleep duration since nicotine is a stimulant.41
Insomnia is commonly treated with sedative antidepressants and hypnotics—eg, mirtazapine and olanzapine—that contribute to weight gain.42 In addition, other common pharmaceuticals used for sleep disorders, such as diphenhydramine, have sedative properties and tend to lead to weight gain.43 Because so many medications affect sleep and weight, carefully review patients’ medication lists and switch offending agents to weight-neutral drugs if possible.
Treatment and tools to improve sleep in patients with obesity
Given the strong correlation between obesity and sleep disorders, validated screening tools should be used to assess sleep quality, including onset and potential symptoms associated with poor sleep (TABLE 144). For weight management to succeed in patients with obesity, it is crucial to address sleep in addition to nutrition and physical activity.17,45

Physical activity has many benefits to overall health, especially for chronic diseases such as type 2 diabetes and hypertension. The Centers for Disease Control and Prevention recommends at least 150 minutes of moderate-intensity aerobic activity or 75 minutes of vigorous-intensity aerobic exercise per week in addition to muscle-strengthening activities 2 or more days per week.46 However, approximately 300 minutes of moderate-
Physical activity and diet in combination are vital, but diet restriction has a more substantial effect on weight loss than physical activity alone.48 Still, physical activity is essential in helping maintain and prevent weight regain.
Continue to: Nonpharmacologic interventions
Nonpharmacologic interventions include promoting greater sleep quality and quantity by emphasizing good sleep hygiene practices. Developing a practical and effective bedtime routine, creating a quiet sleep environment, and practicing healthy daily habits are essential components to sleep hygiene(TABLE 249,50). Relaxation techniques and cognitive behavioral therapy (CBT) also can help. CBT for insomnia (CBT-I) is the first-line intervention for chronic insomnia.51 Sleep restriction is a type of CBT used to treat insomnia, encouraging short-term sleep loss in the hopes of improving insomnia. A trial by Logue et al showed that patients with overweight and obesity randomized to undergo CBT with better sleep hygiene (nonpharmacologic) interventions had a greater mean weight loss percentage (5% vs 2%; P = .04) than did those who received CBT alone.52

Eastern medicine including herbal interventions lack evidence of efficacy and safety. Further studies need to be done on the effects that chamomile, kava, valerian root (Valeriana officinalis), tryptophan, and Wu Ling (from mycelia Xylaria nigripes) might have on sleep.53
Proceed cautiously with medication. The American College of Physicians recommends a shared decision-making approach when considering pharmacologic therapy for chronic insomnia and the American Academy of Sleep Medicine (AASM) offers guidance on options.51,54 However, the evidence behind AASM sleep pharmacologic recommendations is weak, implying a lesser degree of confidence in the outcome and, therefore, in its appropriateness. Thus, it falls upon the clinician and patient to weigh the benefits and burdens of the pharmacologic treatments of insomnia. If indicated, medications suggested to treat sleep onset and sleep maintenance insomnia are eszopiclone, zolpidem, and temazepam. Zaleplon, triazolam, and ramelteon may improve sleep initiation. Suvorexant and doxepin are used for sleep-maintenance insomnia.54 Exploring patient preferences, cost of treatment, health care options, and available resources should all be considered.
CORRESPONDENCE
Ecler Ercole Jaqua, MD, MBA, FAAFP, AGSF, FACLM, DipABOM, Loma Linda University Health, 25455 Barton Road, Suite 206A, Loma Linda, CA 92354; ejaqua@llu.edu
1. Aminoff MJ, Boller F, Swaab DF. We spend about one-third of our life either sleeping or attempting to do so. Handb Clin Neurol. 2011;98:vii. doi: 10.1016/B978-0-444-52006-7.00047-2
2. Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
3. CDC. Sleep and sleep disorders, adults. Accessed September 21, 2023. www.cdc.gov/sleep/data-and-statistics/adults.html
4. Chattu VK, Manzar MD, Kumary S. The global problem of insufficient sleep and its serious public health implications. Healthcare (Basel). 2019;7:1. doi: 10.3390/healthcare7010001
5. Taheri S, Lin L, Austin D, et al. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004;1:e62. doi: 10.1371/journal.pmed.0010062
6. Hafner M, Stepanek M, Taylor J, et al. Why sleep matters—the economic costs of insufficient sleep. Rand Health Q. 2017;6:11.
7. Hisler G, Twenge JM, Krizan Z. Associations between screen time and short sleep duration among adolescents varies by media type: evidence from a cohort study. Sleep Med. 2020;66:92-102. doi: 10.1016/j.sleep.2019.08.007
8. Ogilvie RP, Patel SR. The epidemiology of sleep and obesity. Sleep Health. 2017;3:383-388. doi: 10.1016/j.sleh.2017.07.013
9. CDC. Sleep and sleep disorders: How much sleep do I need? Accessed September 21, 2023. www.cdc.gov/sleep/about_sleep/how_much_sleep.html
10. van Egmond LT, Meth EMS, Engström J, et al. Effects of acute sleep loss on leptin, ghrelin, and adiponectin in adults with healthy weight and obesity: a laboratory study. Obesity (Silver Spring). 2023;31:635-641. doi: 10.1002/oby.23616
11. Spiegel K, Tasali E, Penev P, et al. Brief communication: sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann Intern Med. 2004;141:846-850. doi: 10.7326/0003-4819-141-11-200412070-00008
12. Antza C, Kostopoulos G, Mostafa S, et al. The links between sleep duration, obesity and type 2 diabetes mellitus. J Endocrinol. 2021;252:125-141. doi: 10.1530/JOE-21-0155
13. Baron KG, Reid KJ, Kern AS, et al. Role of sleep timing in caloric intake and BMI. Obesity (Silver Spring). 2011;19:1374-1381. doi: 10.1038/oby.2011.100
14. Liu XY, Zheng CL, Xu C, et al. Nighttime snacking is associated with risk of obesity and hyperglycemia in adults: a cross-sectional survey from Chinese adult teachers J Biomed Res. 2017;31:541-547. doi: 10.7555/JBR.31.20160083
15. Cai Z, Yang Y, Zhang J, et al. The relationship between daytime napping and obesity: a systematic review and meta-analysis. Sci Rep. 2023.13:12124. doi: 10.1038/s41598-023-37883-7
16. Nedeltcheva AV, Kilkus JM, Imperial J, et al. Insufficient sleep undermines dietary efforts to reduce adiposity. Ann Intern Med. 2010;153:435-441. doi: 10.7326/0003-4819-153-7-201010050-00006
17. Chaput JP, Tremblay A. Adequate sleep to improve the treatment of obesity. CMAJ. 2012;184:1975-1976. doi: 10.1503/cmaj.120876
18. Kelsey MM, Zaepfel A, Bjornstad P, et al. Age-related consequences of childhood obesity. Gerontology. 2014;60:222-228. doi: 10.1159/000356023
19. Fryar CD, Carroll MD, Afful J. Prevalence of overweight, obesity, and severe obesity among children and adolescents aged 2-19 years: United States, 1963-1965 through 2017-2018. National Center for Health Statistics Health E-Stats. Updated January 29, 2021. Accessed September 21, 2021. www.cdc.gov/nchs/data/hestat/obesity-child-17-18/overweight-obesity-child-H.pdf
20. Fatima Y, Doi SAR, Mamun AA. Sleep quality and obesity in young subjects: a meta-analysis. Obes Rev. 2016;17:1154-1166. doi: 10.1111/obr.12444
21. Gohil A, Hannon TS. Poor sleep and obesity: concurrent epidemics in adolescent youth. Front Endocrinol. 2018;9:364. doi: 10.3389/fendo.2018.00364
22. Golley RK, Maher CA, Matricciani L, et al. Sleep duration or bedtime? Exploring the association between sleep timing behaviour, diet and BMI in children and adolescents. Int J Obes (Lond). 2013;37:546-551. doi: 10.1038/ijo.2012.212
23. Alessi CA. Sleep issues. In: Harper GM, Lyons WL, Potter JF, eds. Geriatrics Review Syllabus (GRS 10). Updated January 2021. Accessed August 29, 2023. http://geriatricscareonline.org
24. Patel SR, Blackwell T, Redline S, et al. The association between sleep duration and obesity in older adults. Int J Obes (Lond). 2008;32:1825-1834. doi: 10.1038/ijo.2008.198
25. Cai GH, Theorell-Haglöw J, Janson C, et al. Insomnia symptoms and sleep duration and their combined effects in relation to associations with obesity and central obesity. Sleep Med. 2018;46:81-87. doi: 10.1016/j.sleep.2018.03.009
26. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011;14:402-412. doi: 10.1097/MCO.0b013 e3283479109
27. Franklin KA, Lindberg E. Obstructive sleep apnea is a common disorder in the population–a review on the epidemiology of sleep apnea. J Thorac Dis. 2015;7:1311-1322. doi: 10.3978/j.issn.2072-1439.2015.06.11
28. USPSTF. Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for obstructive sleep apnea in adults: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:407-414. doi: 10.1001/jama.2016.20325
29. Goyal M, Johnson J. Obstructive sleep apnea diagnosis and management. Mo Med. 2017;114:120-124.
30. American Academy of Sleep Medicine. Hidden health crisis costing America billions: underdiagnosing and undertreating obstructive sleep apnea draining healthcare system. 2016. Accessed September 25, 2023. https://aasm.org/wp-content/uploads/2017/10/sleep-apnea-economic-crisis.pdf
31. Devaraj, NK. Knowledge, attitude, and practice regarding obstructive sleep apnea among primary care physicians. Sleep Breath. 2020;24:1581-1590. doi: 10.1007/s11325-020-02040-1
32. Mysliwiec V, Martin JL, Ulmer CS, et al. The management of chronic insomnia disorder and obstructive sleep apnea: synopsis of the 2019 U.S. Department of Veterans Affairs and U.S. Department of Defense Clinical Practice Guidelines. Ann Intern Med. 2020;172:325-336. doi: 10.7326/M19-3575
33. Kuna ST, Reboussin DM, Strotmeyer ES, et al. Effects of weight loss on obstructive sleep apnea severity. Ten-year results of the Sleep AHEAD study. Am J Respir Crit Care Med. 2021;203:221-229. doi: 10.1164/rccm.201912-2511OC
34. St-Onge MP, Tasali E. Weight loss is integral to obstructive sleep apnea management. Ten-year follow-up in Sleep AHEAD. Am J Respir Crit Care Med. 2021;203:161-162. doi: 10.1164/rccm.202007-2906ED
35. Zheng D, Yuan X, Ma C, et al. Alcohol consumption and sleep quality: a community-based study. Public Health Nutr. 2021;24:4851-4858. doi: 10.1017/S1368980020004553
36. Chakravorty S, Chaudhary NS, Brower KJ. Alcohol dependence and its relationship with insomnia and other sleep disorders. Alcohol Clin Exp Res. 2016;40:2271-2282. doi: 10.1111/acer.13217
37. Elmenhorst EM, Elmenhorst D, Benderoth S, et al. Cognitive impairments by alcohol and sleep deprivation indicate trait characteristics and a potential role for adenosine A1 receptors. Proc Natl Acad Sci U S A. 2018;115:8009-8014. doi: 10.1073/pnas.1803770115
38. Traversy G, Chaput JP. Alcohol consumption and obesity: an update. Curr Obes Rep. 2015;4:122-130. doi: 10.1007/s13679-014-0129-4
39. McCann UD, Sgambati FP, Schwartz AR, et al. Sleep apnea in young abstinent recreational MDMA (“ecstasy”) consumers. Neurology. 2009;73:2011-2017. doi: 10.1212/WNL.0b013e3181c51a62
40. Grau-López L, Grau-López L, Daigre C, et al. Insomnia symptoms in patients with substance use disorders during detoxification and associated clinical features. Front Psychiatry. 2020;11:540022. doi: 10.3389/fpsyt.2020.540022
41. Boehm MA, Lei QM, Lloyd RM, et al. Depression, anxiety, and tobacco use: overlapping impediments to sleep in a national sample of college students. J Am Coll Health. 2016;64:565-574. doi: 10.1080/07448481.2016.1205073
42. Gracious BL, Meyer AE. Psychotropic-induced weight gain and potential pharmacologic treatment strategies. Psychiatry (Edgmont). 2005;2:36-42.
43. Ratliff JC, Barber JA, Palmese LB, et al. Association of prescription H1 antihistamine use with obesity: results from the National Health and Nutrition Examination Survey. Obesity (Silver Spring). 2010;18:2398-2400. doi: 10.1038/oby.2010.176
44. Pataka A, Daskalopoulou E, Kalamaras G, et al. Evaluation of five different questionnaires for assessing sleep apnea syndrome in a sleep clinic. Sleep Med. 2014;15:776-781. doi: 10.1016/j.sleep.2014.03.012
45. Kline CE, Chasens ER, Bizhanova Z, et al. The association between sleep health and weight change during a 12-month behavioral weight loss intervention. Int J Obes (Lond). 2021;45:639-649. doi: 10.1038/s41366-020-00728-8
46. CDC. How much physical activity do adults need? Accessed August 23, 2023. www.cdc.gov/physicalactivity/basics/adults/index.htm
47. Flack KD, Hays HM, Moreland J, et al. Exercise for weight loss: further evaluating energy compensation with exercise. Med Sci Sports Exerc. 2020;52:2466-2475. doi: 10.1249/MSS.0000000000002376
48. Swift DL, Johannsen NM, Lavie CJ, et al. The role of exercise and physical activity in weight loss and maintenance. Prog Cardiovasc Dis. 2014;56:441-447. doi: 10.1016/j.pcad.2013.09.012
49. Irish LA, Kline CE, Gunn HE, et al. The role of sleep hygiene in promoting public health: a review of empirical evidence. Sleep Med Rev. 2015;22:23-36. doi: 10.1016/j.smrv.2014.10.001
50. CDC. Tips for better sleep. 2022. Accessed August 4, 2023. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html
51. Qaseem A, Kansagara D, Forciea MA, et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165:125-133. doi: 10.7326/M15-2175
52. Logue EE, Bourguet CC, Palmieri PA, et al. The better weight-better sleep study: a pilot intervention in primary care. Am J Health Behav. 2012;36:319-334. doi: 10.5993/AJHB.36.3.4
53. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12. doi: 10.1016/j.smrv.2014.12.003
54. Sateia MJ, Buysse DJ, Krystal AD, et al. Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017;13:307-349. doi: 10.5664/jcsm.6470
1. Aminoff MJ, Boller F, Swaab DF. We spend about one-third of our life either sleeping or attempting to do so. Handb Clin Neurol. 2011;98:vii. doi: 10.1016/B978-0-444-52006-7.00047-2
2. Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
3. CDC. Sleep and sleep disorders, adults. Accessed September 21, 2023. www.cdc.gov/sleep/data-and-statistics/adults.html
4. Chattu VK, Manzar MD, Kumary S. The global problem of insufficient sleep and its serious public health implications. Healthcare (Basel). 2019;7:1. doi: 10.3390/healthcare7010001
5. Taheri S, Lin L, Austin D, et al. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004;1:e62. doi: 10.1371/journal.pmed.0010062
6. Hafner M, Stepanek M, Taylor J, et al. Why sleep matters—the economic costs of insufficient sleep. Rand Health Q. 2017;6:11.
7. Hisler G, Twenge JM, Krizan Z. Associations between screen time and short sleep duration among adolescents varies by media type: evidence from a cohort study. Sleep Med. 2020;66:92-102. doi: 10.1016/j.sleep.2019.08.007
8. Ogilvie RP, Patel SR. The epidemiology of sleep and obesity. Sleep Health. 2017;3:383-388. doi: 10.1016/j.sleh.2017.07.013
9. CDC. Sleep and sleep disorders: How much sleep do I need? Accessed September 21, 2023. www.cdc.gov/sleep/about_sleep/how_much_sleep.html
10. van Egmond LT, Meth EMS, Engström J, et al. Effects of acute sleep loss on leptin, ghrelin, and adiponectin in adults with healthy weight and obesity: a laboratory study. Obesity (Silver Spring). 2023;31:635-641. doi: 10.1002/oby.23616
11. Spiegel K, Tasali E, Penev P, et al. Brief communication: sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann Intern Med. 2004;141:846-850. doi: 10.7326/0003-4819-141-11-200412070-00008
12. Antza C, Kostopoulos G, Mostafa S, et al. The links between sleep duration, obesity and type 2 diabetes mellitus. J Endocrinol. 2021;252:125-141. doi: 10.1530/JOE-21-0155
13. Baron KG, Reid KJ, Kern AS, et al. Role of sleep timing in caloric intake and BMI. Obesity (Silver Spring). 2011;19:1374-1381. doi: 10.1038/oby.2011.100
14. Liu XY, Zheng CL, Xu C, et al. Nighttime snacking is associated with risk of obesity and hyperglycemia in adults: a cross-sectional survey from Chinese adult teachers J Biomed Res. 2017;31:541-547. doi: 10.7555/JBR.31.20160083
15. Cai Z, Yang Y, Zhang J, et al. The relationship between daytime napping and obesity: a systematic review and meta-analysis. Sci Rep. 2023.13:12124. doi: 10.1038/s41598-023-37883-7
16. Nedeltcheva AV, Kilkus JM, Imperial J, et al. Insufficient sleep undermines dietary efforts to reduce adiposity. Ann Intern Med. 2010;153:435-441. doi: 10.7326/0003-4819-153-7-201010050-00006
17. Chaput JP, Tremblay A. Adequate sleep to improve the treatment of obesity. CMAJ. 2012;184:1975-1976. doi: 10.1503/cmaj.120876
18. Kelsey MM, Zaepfel A, Bjornstad P, et al. Age-related consequences of childhood obesity. Gerontology. 2014;60:222-228. doi: 10.1159/000356023
19. Fryar CD, Carroll MD, Afful J. Prevalence of overweight, obesity, and severe obesity among children and adolescents aged 2-19 years: United States, 1963-1965 through 2017-2018. National Center for Health Statistics Health E-Stats. Updated January 29, 2021. Accessed September 21, 2021. www.cdc.gov/nchs/data/hestat/obesity-child-17-18/overweight-obesity-child-H.pdf
20. Fatima Y, Doi SAR, Mamun AA. Sleep quality and obesity in young subjects: a meta-analysis. Obes Rev. 2016;17:1154-1166. doi: 10.1111/obr.12444
21. Gohil A, Hannon TS. Poor sleep and obesity: concurrent epidemics in adolescent youth. Front Endocrinol. 2018;9:364. doi: 10.3389/fendo.2018.00364
22. Golley RK, Maher CA, Matricciani L, et al. Sleep duration or bedtime? Exploring the association between sleep timing behaviour, diet and BMI in children and adolescents. Int J Obes (Lond). 2013;37:546-551. doi: 10.1038/ijo.2012.212
23. Alessi CA. Sleep issues. In: Harper GM, Lyons WL, Potter JF, eds. Geriatrics Review Syllabus (GRS 10). Updated January 2021. Accessed August 29, 2023. http://geriatricscareonline.org
24. Patel SR, Blackwell T, Redline S, et al. The association between sleep duration and obesity in older adults. Int J Obes (Lond). 2008;32:1825-1834. doi: 10.1038/ijo.2008.198
25. Cai GH, Theorell-Haglöw J, Janson C, et al. Insomnia symptoms and sleep duration and their combined effects in relation to associations with obesity and central obesity. Sleep Med. 2018;46:81-87. doi: 10.1016/j.sleep.2018.03.009
26. Beccuti G, Pannain S. Sleep and obesity. Curr Opin Clin Nutr Metab Care. 2011;14:402-412. doi: 10.1097/MCO.0b013 e3283479109
27. Franklin KA, Lindberg E. Obstructive sleep apnea is a common disorder in the population–a review on the epidemiology of sleep apnea. J Thorac Dis. 2015;7:1311-1322. doi: 10.3978/j.issn.2072-1439.2015.06.11
28. USPSTF. Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for obstructive sleep apnea in adults: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:407-414. doi: 10.1001/jama.2016.20325
29. Goyal M, Johnson J. Obstructive sleep apnea diagnosis and management. Mo Med. 2017;114:120-124.
30. American Academy of Sleep Medicine. Hidden health crisis costing America billions: underdiagnosing and undertreating obstructive sleep apnea draining healthcare system. 2016. Accessed September 25, 2023. https://aasm.org/wp-content/uploads/2017/10/sleep-apnea-economic-crisis.pdf
31. Devaraj, NK. Knowledge, attitude, and practice regarding obstructive sleep apnea among primary care physicians. Sleep Breath. 2020;24:1581-1590. doi: 10.1007/s11325-020-02040-1
32. Mysliwiec V, Martin JL, Ulmer CS, et al. The management of chronic insomnia disorder and obstructive sleep apnea: synopsis of the 2019 U.S. Department of Veterans Affairs and U.S. Department of Defense Clinical Practice Guidelines. Ann Intern Med. 2020;172:325-336. doi: 10.7326/M19-3575
33. Kuna ST, Reboussin DM, Strotmeyer ES, et al. Effects of weight loss on obstructive sleep apnea severity. Ten-year results of the Sleep AHEAD study. Am J Respir Crit Care Med. 2021;203:221-229. doi: 10.1164/rccm.201912-2511OC
34. St-Onge MP, Tasali E. Weight loss is integral to obstructive sleep apnea management. Ten-year follow-up in Sleep AHEAD. Am J Respir Crit Care Med. 2021;203:161-162. doi: 10.1164/rccm.202007-2906ED
35. Zheng D, Yuan X, Ma C, et al. Alcohol consumption and sleep quality: a community-based study. Public Health Nutr. 2021;24:4851-4858. doi: 10.1017/S1368980020004553
36. Chakravorty S, Chaudhary NS, Brower KJ. Alcohol dependence and its relationship with insomnia and other sleep disorders. Alcohol Clin Exp Res. 2016;40:2271-2282. doi: 10.1111/acer.13217
37. Elmenhorst EM, Elmenhorst D, Benderoth S, et al. Cognitive impairments by alcohol and sleep deprivation indicate trait characteristics and a potential role for adenosine A1 receptors. Proc Natl Acad Sci U S A. 2018;115:8009-8014. doi: 10.1073/pnas.1803770115
38. Traversy G, Chaput JP. Alcohol consumption and obesity: an update. Curr Obes Rep. 2015;4:122-130. doi: 10.1007/s13679-014-0129-4
39. McCann UD, Sgambati FP, Schwartz AR, et al. Sleep apnea in young abstinent recreational MDMA (“ecstasy”) consumers. Neurology. 2009;73:2011-2017. doi: 10.1212/WNL.0b013e3181c51a62
40. Grau-López L, Grau-López L, Daigre C, et al. Insomnia symptoms in patients with substance use disorders during detoxification and associated clinical features. Front Psychiatry. 2020;11:540022. doi: 10.3389/fpsyt.2020.540022
41. Boehm MA, Lei QM, Lloyd RM, et al. Depression, anxiety, and tobacco use: overlapping impediments to sleep in a national sample of college students. J Am Coll Health. 2016;64:565-574. doi: 10.1080/07448481.2016.1205073
42. Gracious BL, Meyer AE. Psychotropic-induced weight gain and potential pharmacologic treatment strategies. Psychiatry (Edgmont). 2005;2:36-42.
43. Ratliff JC, Barber JA, Palmese LB, et al. Association of prescription H1 antihistamine use with obesity: results from the National Health and Nutrition Examination Survey. Obesity (Silver Spring). 2010;18:2398-2400. doi: 10.1038/oby.2010.176
44. Pataka A, Daskalopoulou E, Kalamaras G, et al. Evaluation of five different questionnaires for assessing sleep apnea syndrome in a sleep clinic. Sleep Med. 2014;15:776-781. doi: 10.1016/j.sleep.2014.03.012
45. Kline CE, Chasens ER, Bizhanova Z, et al. The association between sleep health and weight change during a 12-month behavioral weight loss intervention. Int J Obes (Lond). 2021;45:639-649. doi: 10.1038/s41366-020-00728-8
46. CDC. How much physical activity do adults need? Accessed August 23, 2023. www.cdc.gov/physicalactivity/basics/adults/index.htm
47. Flack KD, Hays HM, Moreland J, et al. Exercise for weight loss: further evaluating energy compensation with exercise. Med Sci Sports Exerc. 2020;52:2466-2475. doi: 10.1249/MSS.0000000000002376
48. Swift DL, Johannsen NM, Lavie CJ, et al. The role of exercise and physical activity in weight loss and maintenance. Prog Cardiovasc Dis. 2014;56:441-447. doi: 10.1016/j.pcad.2013.09.012
49. Irish LA, Kline CE, Gunn HE, et al. The role of sleep hygiene in promoting public health: a review of empirical evidence. Sleep Med Rev. 2015;22:23-36. doi: 10.1016/j.smrv.2014.10.001
50. CDC. Tips for better sleep. 2022. Accessed August 4, 2023. www.cdc.gov/sleep/about_sleep/sleep_hygiene.html
51. Qaseem A, Kansagara D, Forciea MA, et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;165:125-133. doi: 10.7326/M15-2175
52. Logue EE, Bourguet CC, Palmieri PA, et al. The better weight-better sleep study: a pilot intervention in primary care. Am J Health Behav. 2012;36:319-334. doi: 10.5993/AJHB.36.3.4
53. Leach MJ, Page AT. Herbal medicine for insomnia: a systematic review and meta-analysis. Sleep Med Rev. 2015;24:1-12. doi: 10.1016/j.smrv.2014.12.003
54. Sateia MJ, Buysse DJ, Krystal AD, et al. Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017;13:307-349. doi: 10.5664/jcsm.6470
PRACTICE RECOMMENDATIONS
› Consider cognitive behaviorial therapy for insomnia (CBT-I) first-line treatment for insomnia. A
› Carefully review patients’ medication lists, as many pharmaceuticals can affect weight and sleep. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Semaglutide win in HFpEF with obesity regardless of ejection fraction: STEP-HFpEF
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.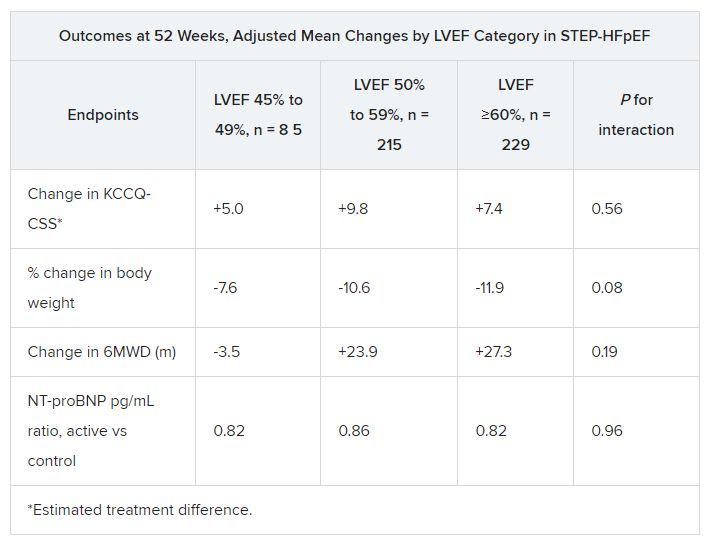
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
CLEVELAND – independently of baseline left-ventricular ejection fraction (LVEF).
The finding comes from a prespecified secondary analysis of the STEP-HFpEF trial of more than 500 nondiabetic patients with obesity and HF with an initial LVEF of 45% or greater.
They suggest that for patients with the obesity phenotype of HFpEF, semaglutide (Wegovy) could potentially join SGLT2 inhibitors on the short list of meds with consistent treatment effects whether LVEF is mildly reduced, preserved, or in the normal range.
That would distinguish the drug, a glucagon-like peptide-1 (GLP-1) receptor agonist, from mineralocorticoid receptor antagonists (MRA), sacubitril-valsartan (Entresto), and other renin-angiotensin-system inhibitors (RASi), whose benefits tend to taper off with rising LVEF.
The patients assigned to semaglutide showed significant improvement in both primary endpoints – change in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and change in body weight at 52 weeks – whether their baseline LVEF was 45%-49%, 50%-59%, or 60% or greater.
Results were similar for improvements in 6-minute walk distance (6MWD) and levels of NT-terminal pro–brain natriuretic peptide (NT-proBNP) and C-reactive protein, observed Javed Butler, MD, when presenting the analysis at the annual meeting of the Heart Failure Society of America, Cleveland.
Dr. Butler, of Baylor Scott and White Research Institute, Dallas, and the University of Mississippi, Jackson, is also lead author of the study, which was published on the same day in the Journal of the American College of Cardiology.
In his presentation, Dr. Butler singled out the NT-proBNP finding as “very meaningful” with respect to understanding potential mechanisms of the drug effects observed in the trial.
For example, people with obesity tend to have lower than average natriuretic peptide levels that “actually go up a bit” when they lose weight, he observed. But in the trial, “we saw a reduction in NT-proBNP in spite of the weight loss,” regardless of LVEF category.
John McMurray, MD, University of Glasgow, the invited discussant for Dr. Butler’s presentation, agreed that it raises the question whether weight loss was the sole semaglutide effect responsible for the improvement in heart failure status and biomarkers. The accompanying NT-proBNP reductions – when the opposite might otherwise have been expected – may point to a possible mechanism of action that is “something more than just weight loss,” he said. “If that were the case, it becomes very important, because it means that this treatment might do good things in non-obese patients or might do good things in patients with other types of heart failure.”
‘Vital reassurance’
More definitive trials are needed “to clarify safety and efficacy of obesity-targeted therapeutics in HF across the ejection fraction spectrum,” according to an accompanying editorial).
Still, the STEP-HFpEF analysis “strengthens the role of GLP-1 [receptor agonists] to ameliorate health status” for patients with obesity and HF with mildly reduced or preserved ejection fraction, write Muthiah Vaduganathan, MD, MPH, and John W. Ostrominski, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Its findings “provide vital reassurance” on semaglutide safety and efficacy in HF with below-normal LVEF and “tentatively support the existence of a more general, LVEF-independent, obesity-related HF phenotype capable of favorable modification with incretin-based therapies.”
The lack of heterogeneity in treatment effects across LVEF subgroups “is not surprising,” but “the findings reinforce that the benefits of this therapy in those meeting trial criteria do not vary by left ventricular ejection fraction,” Gregg C. Fonarow, MD, University of California, Los Angeles, Medical Center, said in an interview.
It remains unknown, however, “whether the improvement in health status, functional status, and reduced inflammation” will translate to reduced risk of cardiovascular death or HF hospitalization, said Dr. Fonarow, who isn’t connected to STEP-HFpEF.
It’s a question for future studies, he agreed, whether semaglutide would confer similar benefits for patients with obesity and HF with LVEF less than 45% or in non-obese HF patients.
Dr. McMurray proposed that future GLP-1 receptor agonist heart-failure trials should include non-obese patients to determine whether the effects seen in STEP-HFpEF were due to something more than weight loss. Trials in patients with obesity and HF with reduced LVEF would also be important.
“If it turns out just to be about weight loss, then we need to think about the alternatives,” including diet, exercise, and bariatric surgery but also, potentially, weight-loss drugs other than semaglutide, he said.
No heterogeneity by LVEF
STEP-HFpEF randomly assigned 529 patients free of diabetes with an LVEF greater than or equal to 45%, a body mass index (BMI) of at least 30 kg/m2, and NYHA functional status of 2-4 to either a placebo injection or 2.4-mg semaglutide subcutaneously once a week (the dose used for weight reduction) atop standard care.
As previously reported, those assigned to semaglutide showed significant improvements at 1 year in symptoms and in physical limitation, per changes in KCCQ-CSS, and weight loss, compared with the control group. Their exercise capacity, as measured by 6MWD, also improved.
The more weight patients lost while taking semaglutide, the better their KCCQ-CSS and 6MWD outcomes, a prior secondary analysis suggested. But the STEP-HFpEF researchers said weight loss did not appear to explain all of their gains, compared with usual care.
For the current analysis, the 263 patients assigned to receive semaglutide and 266 control patients were divided into three groups by baseline LVEF and compared for the same outcomes.
The semaglutide group, compared with control patients, also showed a significantly increased hierarchical composite win ratio, 1.72 (95% CI, 1.37-2.15; P < .001), that was consistent across LVEF categories and that accounted for all-cause mortality, HF events, KCCQ-CSS and 6MWD changes, and change in CRP.
Limitations make it hard to generalize the results, the authors caution. Well over 90% of the participants were White patients, for example, and the overall trial was not powered to show subgroup differences.
Given the many patients with HFpEF who have a cardiometabolic phenotype and are with overweight or obesity, write Dr. Butler and colleagues, their treatment approach “may ultimately include combination therapy with SGLT2 inhibitors and GLP-1 receptor agonists, given their non-overlapping and complementary mechanisms of action.”
Dr. Fonarow noted that both MRAs and sacubitril-valsartan offer clinical benefits for patients with HF and LVEF “in the 41%-60% range” that are evident “across BMI categories.”
So it’s likely, he said, that those medications as well as SGLT2 inhibitors will be used along with GLP-1 receptor agonists for patients with HFpEF and obesity.
STEP-HFpEF was funded by Novo Nordisk. Dr. Butler and the other authors disclose consulting for many companies, a list of which can be found in the report. Dr. Fonarow reports consulting for multiple companies. Dr. McMurray discloses consulting for AstraZeneca. Dr. Ostrominski reports no relevant disclosures. Dr. Vaduganathan discloses receiving grant support, serving on advisory boards, or speaking for multiple companies and serving on committees for studies sponsored by AstraZeneca, Galmed, Novartis, Bayer AG, Occlutech, and Impulse Dynamics.
A version of this article appeared on Medscape.com.
AT HFSA 2023
