User login
Smoking-cessation attempts changed little over 7-year span
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
according to the Centers for Disease Control and Prevention.

The median percentage of adult smokers who tried to quit cigarettes over the past year went from 64.9% in 2011 to 65.4% in 2017, CDC investigators reported in the Morbidity and Mortality Weekly Report, but the rate has gone down since 2014, when it reached 66.9%.
“The limited progress in increasing quit attempts … together with the variation in quit attempt prevalence among states, underscores the importance of enhanced efforts to motivate and help smokers to quit,” wrote Kimp Walton, MS, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and associates.
State-specific trends in quit-attempt rates reflected the national situation. The prevalence of past-year cessation attempts went up significantly in four states (Kansas, Louisiana, Virginia, and West Virginia) from 2011 to 2017, went down significantly in two states (New York and Tennessee), and did not change significantly in the other 44 states and the District of Columbia, they wrote.
In 2017, cigarette smokers in Connecticut were the most likely to have tried to quit in the past year, with a rate of 71.6%. The only other places with rates greater than 70% were Delaware, D.C., New Jersey, and Texas. The lowest quit-attempt rate that year, 58.6%, belonged to Wisconsin, with Iowa and Missouri the only other states under 60%, the investigators reported based on data from annual Behavioral Risk Factor Surveillance System surveys.
“Because most smokers make multiple quit attempts before succeeding, as many as 30 on average, tobacco dependence is viewed as a chronic, relapsing condition that requires repeated intervention. Smokers should be encouraged to keep trying to quit until they succeed, and health care providers should be encouraged to keep supporting smokers until they quit,” investigators wrote.
SOURCE: Walton K et al. MMWR. 2019 Jul 19;68(28):621-6.
FROM MMWR
New analysis challenges fluid resuscitation guidelines for patients in shock
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
Although guideline recommended, according to a detailed analysis of available data, including a randomized trial.
Several sets of guidelines for resuscitation of patients in shock have advocated volume expansion with bolus intravenous fluid, but that recommendation was based on expected physiologic benefits not a randomized trial. The only randomized trial associated this approach showed increased mortality, and a new analysis of these and other data appears to explain why.
According to the findings of a study lead by Michael Levin, MD, of the department of medicine at Imperial College London and colleagues, “volume resuscitation is associated with deterioration of respiratory function and neurological function in some patients.” Their study was published in Lancet Respiratory Medicine. The authors stated that saline-induced hyperchloremic acidosis appears to have been “a major contributor” to the observed increase in adverse outcomes.
The key take home message is that “normal saline and other unbuffered crystalloid solutions should be avoided in resuscitating seriously ill patients,” according to the authors, who believe the findings might be relevant to adults as well as children.
The controversy about the role of volume expansion for management of shock was ignited by a 2011 trial called FEAST (N Engl J Med. 2011;364:2483-95). That trial, which randomized African children with severe febrile illness to a bolus of 20-40 mg of 5% albumin solution, a bolus of 0.9% saline solution, or no bolus, was halted early when 48-hour mortality data showed a lower death rate in the no bolus group (7.3%) than either the albumin (10.6%) or saline (10.5%) bolus groups.
The FEAST result was unexpected and so contrary to accepted thinking that it prompted widespread debate, including whether findings in the resource-poor area of the world where the FEAST trial was conducted could be extrapolated to centers elsewhere in the world. Arguing for benefit, fluid resuscitation is known to increase pulse pressure and urinary output. Arguing against benefit, pulmonary edema is a known complication of bolus fluid replacement.
In an attempt to address and potentially resolve this controversy, data collected in the FEAST trial along with four other sets of data involving volume expansion in critically ill children were evaluated with a focus on changes in cardiovascular, neurological, and respiratory function. Analysis of blood biochemistry and blood oxygen transport were also conducted.
The cardiovascular, respiratory, and neurologic functions were scored on the basis of objective measurements, such as heart rate, respiratory rate, and blood pressure. These measures were evaluated prior to fluid administration and at 1 hour, 4 hours, 8 hours, 24 hours, and 48 hours after fluid administration. Odds ratio (OR) of an adverse outcome were evaluated in the context of each 10-unit change in these scores.
Relative to baseline, there was worsening respiratory and neurological function after fluid administration. Although cardiovascular function improved, hemoglobin concentrations were lower in those who received fluid than in those who did not. Fluid resuscitation was also associated with lower bicarbonate and increased base deficit and chloride at 24 hours.
Regression modeling with physiological variables suggests “that the increased mortality in FEAST can be explained by bolus-induced worsening in respiratory and neurological function, hemodilution, and hyperchloremic acidosis,” according to the authors.
Analyses of the four other sets of data, which included children treated for meningococcal sepsis in the United Kingdom, acutely ill with malaria treated in Malawi, and cohorts of children in South Africa and a London hospital for acute illnesses, provided supportive data.
Although this analysis does not address the value of administering buffered solutions in low volumes, the authors concluded that the data from the FEAST trial are generalizable. They challenge the routine use of bolus infusions of saline or albumin in the initial management of shock, which has been guideline recommended. The risks of fluid resuscitation might be particularly high among children who already have compromised respiratory or neurologic function.
SOURCE: Levin M et al. Lancet Respir Med. 2019;7:581-93.
FROM THE LANCET RESPIRATORY MEDICINE
Little association found between in utero H1N1 vaccine and 5-year health outcomes
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
according to Laura K. Walsh of the University of Ottawa and associates.
The investigators conducted a population-based retrospective cohort study from November 2009 to October 2010 of all live births within the province of Ontario. Of the 104,249 eligible live births reported to the Ontario birth registry, 31,295 were exposed to the H1N1 vaccine in utero. After adjustment, there were no significant differences in the women who did and did not receive vaccines during pregnancy, according to the study, published in the BMJ.
After a median follow-up of 5 years, 14% of children received an asthma diagnosis, with a median age at diagnosis of 1.8 years. Children were more likely to receive an asthma diagnosis if their mothers had a preexisting condition or if they were born preterm. At follow-up, 34% of children had at least one upper respiratory tract infection. Sensory disorder, neoplasm, and pediatric complex chronic condition were rare, each occurring in less than 1% of the study cohort (BMJ. 2019 Jul 10. doi: 10.1136/bmj.l4151).
No significant association was found between prenatal exposure to the H1N1 vaccine and upper or lower respiratory infections, otitis media, all infections, neoplasms, sensory disorders, rates of urgent and inpatient health services use, pediatric complex chronic conditions, or mortality. A weak but significant association was observed for asthma (adjusted hazard ratio, 1.05; 95% confidence interval, 1.02-1.09), and a weak inverse association was found for gastrointestinal infections (adjusted incidence rate ratio, 0.94; 95% CI, 0.91-0.98).
“Although we observed a small, but statistically significant, increase in pediatric asthma and a reduction in gastrointestinal infections, we are not aware of any biologic mechanisms to explain these findings. Future studies in different settings and with different influenza vaccine formulations will be important for developing the evidence base on longer-term pediatric outcomes following influenza vaccination during pregnancy,” the investigators concluded.
The study was funded by grants from the Canadian Institutes of Health Research and the Institute for Clinical Evaluative Sciences.
FROM THE BMJ
Vaping device marketers take aim at youth through social media
with targeted messages and images, a study of e-cigarette promotion has found.
In 2018, the JUUL company declared a commitment to support efforts to raise the age of legal purchase of tobacco to age 21 years in all U.S. states. In addition, JUUL deleted its official Facebook and Instagram accounts in November 2018, but the promotion of these products has continued through affiliated marketing campaigns from other online vendors.
Vaping among teens has shot up in popularity in recent years. The prevalence of vaping among young people aged 16-19 years has been estimated at 16% in 2018, up from 11% in 2017 (BMJ. 2019 Jun 19. doi: 10.1136/bmj.12219. A study published in JAMA Pediatrics (2019;173[7]:690-92) found that an estimated 81% of users following a popular Twitter account (@JUULvapor) were aged 13-20 years, with 45% in the 13-17 year age range.
Elizabeth C. Hair, PhD, senior vice president of the Truth Initiative Schroeder Institute, and a team of investigators conducted a study of the “proliferation of JUUL-related content across four themes over a 3-month period: overt promotional content, nicotine and addiction-related content, lifestyle content, and content related to youth culture.” The study appeared online in Tobacco Control (2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824).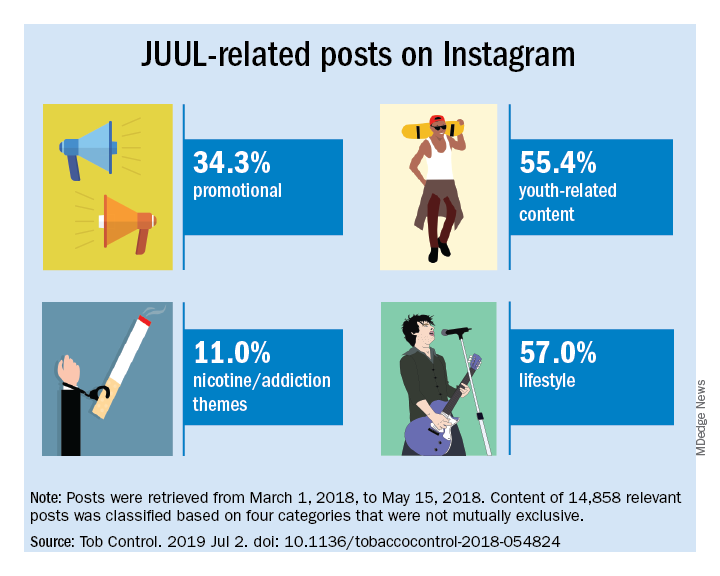
The investigators did a content analysis of social media posts on Instagram related to JUUL and JUUL-like products from March 1 to May 15, 2018. Hash-tag keyword queries of JUUL-related posts on Instagram were collected from the Instagram application programming interface through NUVI, a licensed syndicator of the Instagram firehose. The researchers used 50 hashtags to capture and enumerate individual posts. Examples of the hashtags used are #juul, #juuling, #juulvapor, #juulpod, #switchtojuul, and #juulgang. All posts were included from the official JUUL account and JUUL-related accounts with the highest number of followers at the time of data collection (e.g., @juulcentral, @juulnation, @juul_university, @juul.girls).
The search identified 14,838 posts by 5,201 unique users that featured content relating to product promotion, nicotine and addiction messages, youth culture, and lifestyle themes. Posts were rated promotional incluced branded content, URLs linking to commercial websites, and hashtags indicating affiliations with commercial sites.
Nicotine/addiction posts contained “references to nicotine, including compatible pod-related brand names and nicotine content, as well as any references to addiction or nicotine dependence (e.g., daily use, being an addict, junkie, “nichead,” fiend, maniac), or effects of nicotine use (e.g., “buzz”).
Youth-themed posts included stylistic features such as jargon or slang, acronyms common among youth (e.g., di4j, doit4juul), youth-oriented cartoons, JUUL wrap imagery, youth entertainment, and music. Posts with references to school, the classroom, and other places frequented by youth and youth social networks, family, and peers were included in the youth-themed category.
Lifestyle content referenced "social norms and acceptability-related messages contained any mentions of online or offline communitiesand peer groups (eg, collegelife, juulgirls, juulgang, vapeusa, collegedaily, vapelyfe hashtags) as well as JUUL use during social activities, events, social acceptance of JUULing and any mentions of JUULing as a characteristic of cultural or social identity."
Content analysis of the posts found that 34.3% were promotional, 11% referenced nicotine and addiction themes, and 55.4% featured youth-oriented cultural themes, and 57% featured lifestyle themes. There was overlap among the categories, for example, the 71.9% of the promotional posts had lifestyle messages included and 86.3% of the nicotine/addiction posts contained lifestyle elements. The promotional posts also contained some hashtags referencing cannabis (#420, #710).
An additional feature of the promotional posts is the incentivizing messages. “More than more than a third of JUUL-related posts containing overt promotional content that highlights ways to obtain products at reduced cost, such as giveaways and incentivized friend-tagging. This finding is consistent with previous research which found that Twitter users employed person-tagging (e.g., @username) when purchasing JUUL, suggesting friend-tagging plays an important role in motivating product use,” the researchers wrote.
The study was limited by the short time frame, the analysis of Instagram postings only, and the limitation of only 50 hashtags. These limitations may result in underreporting of the amount of JUUL-related social media messaging that targets youth. In addition, the investigators did not analyze the origin of accounts or the identity of the individuals creating the content.
“The results of this study demonstrate the reach of organic posts that contain JUUL-related content, and posts by third-party vendors of vaping products, who continue to push explicitly youth-targeted advertisements for JUUL and similar e-cigarette products under JUUL-related hashtags,” Dr. Hair wrote. “Our research and studies done by others in the field are one way to build the evidence base to advocate for stricter social media marketing restrictions on tobacco products that are applicable to all players in the field.”
She added that the Food and Drug Administration should use its power to restrict e-cigarette manufacturers from using social media to market to young people. “We also think that social media platforms should do more to adopt and enforce strong and well-enforced policies against the promotion of any tobacco products to young adults,” she concluded.
The study was sponsored by the Truth Initiative. The Truth Initiative was created as a part of the Master Settlement Agreement (MSA) that was negotiated between the tobacco industry and 46 states and the District of Columbia in 1998. The MSA created the American Legacy Foundation (now known as the Truth Initiative), a nonprofit research and educational organization that focuses its efforts on preventing teen smoking and encouraging smokers to quit.
SOURCE: Czaplicki L et al. Tob Control. 2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824.
This article was updated 7/17/2019.
with targeted messages and images, a study of e-cigarette promotion has found.
In 2018, the JUUL company declared a commitment to support efforts to raise the age of legal purchase of tobacco to age 21 years in all U.S. states. In addition, JUUL deleted its official Facebook and Instagram accounts in November 2018, but the promotion of these products has continued through affiliated marketing campaigns from other online vendors.
Vaping among teens has shot up in popularity in recent years. The prevalence of vaping among young people aged 16-19 years has been estimated at 16% in 2018, up from 11% in 2017 (BMJ. 2019 Jun 19. doi: 10.1136/bmj.12219. A study published in JAMA Pediatrics (2019;173[7]:690-92) found that an estimated 81% of users following a popular Twitter account (@JUULvapor) were aged 13-20 years, with 45% in the 13-17 year age range.
Elizabeth C. Hair, PhD, senior vice president of the Truth Initiative Schroeder Institute, and a team of investigators conducted a study of the “proliferation of JUUL-related content across four themes over a 3-month period: overt promotional content, nicotine and addiction-related content, lifestyle content, and content related to youth culture.” The study appeared online in Tobacco Control (2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824).
The investigators did a content analysis of social media posts on Instagram related to JUUL and JUUL-like products from March 1 to May 15, 2018. Hash-tag keyword queries of JUUL-related posts on Instagram were collected from the Instagram application programming interface through NUVI, a licensed syndicator of the Instagram firehose. The researchers used 50 hashtags to capture and enumerate individual posts. Examples of the hashtags used are #juul, #juuling, #juulvapor, #juulpod, #switchtojuul, and #juulgang. All posts were included from the official JUUL account and JUUL-related accounts with the highest number of followers at the time of data collection (e.g., @juulcentral, @juulnation, @juul_university, @juul.girls).
The search identified 14,838 posts by 5,201 unique users that featured content relating to product promotion, nicotine and addiction messages, youth culture, and lifestyle themes. Posts were rated promotional incluced branded content, URLs linking to commercial websites, and hashtags indicating affiliations with commercial sites.
Nicotine/addiction posts contained “references to nicotine, including compatible pod-related brand names and nicotine content, as well as any references to addiction or nicotine dependence (e.g., daily use, being an addict, junkie, “nichead,” fiend, maniac), or effects of nicotine use (e.g., “buzz”).
Youth-themed posts included stylistic features such as jargon or slang, acronyms common among youth (e.g., di4j, doit4juul), youth-oriented cartoons, JUUL wrap imagery, youth entertainment, and music. Posts with references to school, the classroom, and other places frequented by youth and youth social networks, family, and peers were included in the youth-themed category.
Lifestyle content referenced "social norms and acceptability-related messages contained any mentions of online or offline communitiesand peer groups (eg, collegelife, juulgirls, juulgang, vapeusa, collegedaily, vapelyfe hashtags) as well as JUUL use during social activities, events, social acceptance of JUULing and any mentions of JUULing as a characteristic of cultural or social identity."
Content analysis of the posts found that 34.3% were promotional, 11% referenced nicotine and addiction themes, and 55.4% featured youth-oriented cultural themes, and 57% featured lifestyle themes. There was overlap among the categories, for example, the 71.9% of the promotional posts had lifestyle messages included and 86.3% of the nicotine/addiction posts contained lifestyle elements. The promotional posts also contained some hashtags referencing cannabis (#420, #710).
An additional feature of the promotional posts is the incentivizing messages. “More than more than a third of JUUL-related posts containing overt promotional content that highlights ways to obtain products at reduced cost, such as giveaways and incentivized friend-tagging. This finding is consistent with previous research which found that Twitter users employed person-tagging (e.g., @username) when purchasing JUUL, suggesting friend-tagging plays an important role in motivating product use,” the researchers wrote.
The study was limited by the short time frame, the analysis of Instagram postings only, and the limitation of only 50 hashtags. These limitations may result in underreporting of the amount of JUUL-related social media messaging that targets youth. In addition, the investigators did not analyze the origin of accounts or the identity of the individuals creating the content.
“The results of this study demonstrate the reach of organic posts that contain JUUL-related content, and posts by third-party vendors of vaping products, who continue to push explicitly youth-targeted advertisements for JUUL and similar e-cigarette products under JUUL-related hashtags,” Dr. Hair wrote. “Our research and studies done by others in the field are one way to build the evidence base to advocate for stricter social media marketing restrictions on tobacco products that are applicable to all players in the field.”
She added that the Food and Drug Administration should use its power to restrict e-cigarette manufacturers from using social media to market to young people. “We also think that social media platforms should do more to adopt and enforce strong and well-enforced policies against the promotion of any tobacco products to young adults,” she concluded.
The study was sponsored by the Truth Initiative. The Truth Initiative was created as a part of the Master Settlement Agreement (MSA) that was negotiated between the tobacco industry and 46 states and the District of Columbia in 1998. The MSA created the American Legacy Foundation (now known as the Truth Initiative), a nonprofit research and educational organization that focuses its efforts on preventing teen smoking and encouraging smokers to quit.
SOURCE: Czaplicki L et al. Tob Control. 2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824.
This article was updated 7/17/2019.
with targeted messages and images, a study of e-cigarette promotion has found.
In 2018, the JUUL company declared a commitment to support efforts to raise the age of legal purchase of tobacco to age 21 years in all U.S. states. In addition, JUUL deleted its official Facebook and Instagram accounts in November 2018, but the promotion of these products has continued through affiliated marketing campaigns from other online vendors.
Vaping among teens has shot up in popularity in recent years. The prevalence of vaping among young people aged 16-19 years has been estimated at 16% in 2018, up from 11% in 2017 (BMJ. 2019 Jun 19. doi: 10.1136/bmj.12219. A study published in JAMA Pediatrics (2019;173[7]:690-92) found that an estimated 81% of users following a popular Twitter account (@JUULvapor) were aged 13-20 years, with 45% in the 13-17 year age range.
Elizabeth C. Hair, PhD, senior vice president of the Truth Initiative Schroeder Institute, and a team of investigators conducted a study of the “proliferation of JUUL-related content across four themes over a 3-month period: overt promotional content, nicotine and addiction-related content, lifestyle content, and content related to youth culture.” The study appeared online in Tobacco Control (2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824).
The investigators did a content analysis of social media posts on Instagram related to JUUL and JUUL-like products from March 1 to May 15, 2018. Hash-tag keyword queries of JUUL-related posts on Instagram were collected from the Instagram application programming interface through NUVI, a licensed syndicator of the Instagram firehose. The researchers used 50 hashtags to capture and enumerate individual posts. Examples of the hashtags used are #juul, #juuling, #juulvapor, #juulpod, #switchtojuul, and #juulgang. All posts were included from the official JUUL account and JUUL-related accounts with the highest number of followers at the time of data collection (e.g., @juulcentral, @juulnation, @juul_university, @juul.girls).
The search identified 14,838 posts by 5,201 unique users that featured content relating to product promotion, nicotine and addiction messages, youth culture, and lifestyle themes. Posts were rated promotional incluced branded content, URLs linking to commercial websites, and hashtags indicating affiliations with commercial sites.
Nicotine/addiction posts contained “references to nicotine, including compatible pod-related brand names and nicotine content, as well as any references to addiction or nicotine dependence (e.g., daily use, being an addict, junkie, “nichead,” fiend, maniac), or effects of nicotine use (e.g., “buzz”).
Youth-themed posts included stylistic features such as jargon or slang, acronyms common among youth (e.g., di4j, doit4juul), youth-oriented cartoons, JUUL wrap imagery, youth entertainment, and music. Posts with references to school, the classroom, and other places frequented by youth and youth social networks, family, and peers were included in the youth-themed category.
Lifestyle content referenced "social norms and acceptability-related messages contained any mentions of online or offline communitiesand peer groups (eg, collegelife, juulgirls, juulgang, vapeusa, collegedaily, vapelyfe hashtags) as well as JUUL use during social activities, events, social acceptance of JUULing and any mentions of JUULing as a characteristic of cultural or social identity."
Content analysis of the posts found that 34.3% were promotional, 11% referenced nicotine and addiction themes, and 55.4% featured youth-oriented cultural themes, and 57% featured lifestyle themes. There was overlap among the categories, for example, the 71.9% of the promotional posts had lifestyle messages included and 86.3% of the nicotine/addiction posts contained lifestyle elements. The promotional posts also contained some hashtags referencing cannabis (#420, #710).
An additional feature of the promotional posts is the incentivizing messages. “More than more than a third of JUUL-related posts containing overt promotional content that highlights ways to obtain products at reduced cost, such as giveaways and incentivized friend-tagging. This finding is consistent with previous research which found that Twitter users employed person-tagging (e.g., @username) when purchasing JUUL, suggesting friend-tagging plays an important role in motivating product use,” the researchers wrote.
The study was limited by the short time frame, the analysis of Instagram postings only, and the limitation of only 50 hashtags. These limitations may result in underreporting of the amount of JUUL-related social media messaging that targets youth. In addition, the investigators did not analyze the origin of accounts or the identity of the individuals creating the content.
“The results of this study demonstrate the reach of organic posts that contain JUUL-related content, and posts by third-party vendors of vaping products, who continue to push explicitly youth-targeted advertisements for JUUL and similar e-cigarette products under JUUL-related hashtags,” Dr. Hair wrote. “Our research and studies done by others in the field are one way to build the evidence base to advocate for stricter social media marketing restrictions on tobacco products that are applicable to all players in the field.”
She added that the Food and Drug Administration should use its power to restrict e-cigarette manufacturers from using social media to market to young people. “We also think that social media platforms should do more to adopt and enforce strong and well-enforced policies against the promotion of any tobacco products to young adults,” she concluded.
The study was sponsored by the Truth Initiative. The Truth Initiative was created as a part of the Master Settlement Agreement (MSA) that was negotiated between the tobacco industry and 46 states and the District of Columbia in 1998. The MSA created the American Legacy Foundation (now known as the Truth Initiative), a nonprofit research and educational organization that focuses its efforts on preventing teen smoking and encouraging smokers to quit.
SOURCE: Czaplicki L et al. Tob Control. 2019 Jul 2; doi: 10.1136/tobaccocontrol-2018-054824.
This article was updated 7/17/2019.
FROM TOBACCO CONTROL
Statin use linked to less depression, anxiety in ACOS patients
based on data from approximately 9,000 patients.
Although asthma–COPD overlap syndrome (ACOS) has been associated with depression, the effects of oral and inhaled corticosteroids on anxiety and depression in these patients have not been well investigated, wrote Jun-Jun Yeh, MD, of Ditmanson Medical Foundation Chia-Yi (Taiwan) Christian Hospital, and colleagues.
In a study published in the Journal of Affective Disorders, the researchers analyzed 9,139 ACOS patients including 1,252 statin users and 7,887 nonstatin users; 62% were male.
The statin users had significantly lower risk of both anxiety and depression than did the nonstatin users (adjusted hazard ratio 0.34 for anxiety and 0.36 for depression) after researchers controlled for factors including age, sex, comorbidities, and medications. Statin users experienced a total of 109 anxiety or depression events over an average of 8 years’ follow-up, while nonstatin users experienced a total of 1,333 anxiety or depression events over an average of 5 years’ follow-up.
The incidence density rate of anxiety was 11/1,000 person-years for statin users and 33/1,000 person-years for nonstatin users. The incidence density rate of depression was 3/1,000 person-years for statin users and 9/1,000 person-years for nonstatin users.
Significantly lower risk of anxiety and depression also were observed in statin users, compared with nonstatin users, in subgroups of men, women, patients younger than 50 years, and patients aged 50 years and older. The risks of anxiety and depression were lower in statin users versus nonstatin users across all subgroups with or without inhaled or oral corticosteroids.
Overall, the statin users were significantly younger, had more comorbidities, and were more likely to use inhaled or oral corticosteroids than were the nonstatin users.
The findings were limited by several factors including the retrospective nature of the study and a lack of information on prescribed daily doses of medication, the researchers noted. However, the results support those from previous studies and suggest that “the anti-inflammatory effect of statins may attenuate anxiety and depression in ACOS patients, even in the late stages of the disease,” although the exact mechanism of action remains unknown and larger, randomized, controlled trials are needed, they said.
The study was supported by grants from a variety of organizations in Taiwan, China, and Japan. The researchers had no financial conflicts to disclose.
SOURCE: Yeh JJ et al. J Affect Disord. 2019 Jun 15; 253:277-84.
based on data from approximately 9,000 patients.
Although asthma–COPD overlap syndrome (ACOS) has been associated with depression, the effects of oral and inhaled corticosteroids on anxiety and depression in these patients have not been well investigated, wrote Jun-Jun Yeh, MD, of Ditmanson Medical Foundation Chia-Yi (Taiwan) Christian Hospital, and colleagues.
In a study published in the Journal of Affective Disorders, the researchers analyzed 9,139 ACOS patients including 1,252 statin users and 7,887 nonstatin users; 62% were male.
The statin users had significantly lower risk of both anxiety and depression than did the nonstatin users (adjusted hazard ratio 0.34 for anxiety and 0.36 for depression) after researchers controlled for factors including age, sex, comorbidities, and medications. Statin users experienced a total of 109 anxiety or depression events over an average of 8 years’ follow-up, while nonstatin users experienced a total of 1,333 anxiety or depression events over an average of 5 years’ follow-up.
The incidence density rate of anxiety was 11/1,000 person-years for statin users and 33/1,000 person-years for nonstatin users. The incidence density rate of depression was 3/1,000 person-years for statin users and 9/1,000 person-years for nonstatin users.
Significantly lower risk of anxiety and depression also were observed in statin users, compared with nonstatin users, in subgroups of men, women, patients younger than 50 years, and patients aged 50 years and older. The risks of anxiety and depression were lower in statin users versus nonstatin users across all subgroups with or without inhaled or oral corticosteroids.
Overall, the statin users were significantly younger, had more comorbidities, and were more likely to use inhaled or oral corticosteroids than were the nonstatin users.
The findings were limited by several factors including the retrospective nature of the study and a lack of information on prescribed daily doses of medication, the researchers noted. However, the results support those from previous studies and suggest that “the anti-inflammatory effect of statins may attenuate anxiety and depression in ACOS patients, even in the late stages of the disease,” although the exact mechanism of action remains unknown and larger, randomized, controlled trials are needed, they said.
The study was supported by grants from a variety of organizations in Taiwan, China, and Japan. The researchers had no financial conflicts to disclose.
SOURCE: Yeh JJ et al. J Affect Disord. 2019 Jun 15; 253:277-84.
based on data from approximately 9,000 patients.
Although asthma–COPD overlap syndrome (ACOS) has been associated with depression, the effects of oral and inhaled corticosteroids on anxiety and depression in these patients have not been well investigated, wrote Jun-Jun Yeh, MD, of Ditmanson Medical Foundation Chia-Yi (Taiwan) Christian Hospital, and colleagues.
In a study published in the Journal of Affective Disorders, the researchers analyzed 9,139 ACOS patients including 1,252 statin users and 7,887 nonstatin users; 62% were male.
The statin users had significantly lower risk of both anxiety and depression than did the nonstatin users (adjusted hazard ratio 0.34 for anxiety and 0.36 for depression) after researchers controlled for factors including age, sex, comorbidities, and medications. Statin users experienced a total of 109 anxiety or depression events over an average of 8 years’ follow-up, while nonstatin users experienced a total of 1,333 anxiety or depression events over an average of 5 years’ follow-up.
The incidence density rate of anxiety was 11/1,000 person-years for statin users and 33/1,000 person-years for nonstatin users. The incidence density rate of depression was 3/1,000 person-years for statin users and 9/1,000 person-years for nonstatin users.
Significantly lower risk of anxiety and depression also were observed in statin users, compared with nonstatin users, in subgroups of men, women, patients younger than 50 years, and patients aged 50 years and older. The risks of anxiety and depression were lower in statin users versus nonstatin users across all subgroups with or without inhaled or oral corticosteroids.
Overall, the statin users were significantly younger, had more comorbidities, and were more likely to use inhaled or oral corticosteroids than were the nonstatin users.
The findings were limited by several factors including the retrospective nature of the study and a lack of information on prescribed daily doses of medication, the researchers noted. However, the results support those from previous studies and suggest that “the anti-inflammatory effect of statins may attenuate anxiety and depression in ACOS patients, even in the late stages of the disease,” although the exact mechanism of action remains unknown and larger, randomized, controlled trials are needed, they said.
The study was supported by grants from a variety of organizations in Taiwan, China, and Japan. The researchers had no financial conflicts to disclose.
SOURCE: Yeh JJ et al. J Affect Disord. 2019 Jun 15; 253:277-84.
FROM THE JOURNAL OF AFFECTIVE DISORDERS
COPD eosinophil counts predict steroid responders
Triple therapy with an inhaled corticosteroid is particularly helpful for patients with chronic obstructive pulmonary disease (COPD) who have high baseline eosinophil counts, a trial involving more than 10,000 patients found.
Former smokers received greater benefit from inhaled corticosteroids (ICS) than did current smokers, reported lead author Steven Pascoe, MBBS, of GlaxoSmithKline and colleagues. The investigators noted that these findings can help personalize therapy for patients with COPD, which can be challenging to treat because of its heterogeneity. The study was published in Lancet Respiratory Medicine.
The phase 3 IMPACT trial compared single-inhaler fluticasone furoate–umeclidinium–vilanterol with umeclidinium-vilanterol and fluticasone furoate–vilanterol in patients with moderate to very severe COPD at high risk of exacerbation. Of the 10,333 patients involved, approximately one-quarter (26%) had one or more severe exacerbations in the previous year and half (47%) had two or more moderate exacerbations in the same time period. All patients were symptomatic and were aged 40 years or older. A variety of baseline and demographic patient characteristics were recorded, including blood eosinophil count, smoking status, and others. Responses to therapy were measured with trough forced expiratory volume in 1 second (FEV1), symptom scoring, and a quality of life questionnaire.
After 52 weeks, results showed that higher baseline eosinophil counts were associated with progressively greater benefits in favor of triple therapy. For patients with baseline blood eosinophil counts of at least 310 cells per mcL, triple therapy was associated with about half as many moderate and severe exacerbations as treatment with umeclidinium-vilanterol (rate ratio = 0.56; 95% confidence interval, 0.47-0.66). For patients with less than 90 cells per mcL at baseline, the rate ratio for the same two regimens was 0.88, but with a confidence interval crossing 1 (0.74-1.04). For fluticasone furoate–vilanterol vs. umeclidinium-vilanterol, high baseline eosinophil count again demonstrated its predictive power for ICS efficacy, again with an associated rate ratio of 0.56 (0.47-0.66), compared with 1.09 (0.91-1.29) for patients below the lower threshold. Symptom scoring, quality of life, and FEV1 followed a similar trend, although the investigators noted that this was “less marked” for FEV1. Although the trend held regardless of smoking status, benefits were more pronounced among former smokers than current smokers.
“In former smokers, ICS benefits were observed at all blood eosinophil counts when comparing triple therapy with umeclidinium-vilanterol, whereas in current smokers no ICS benefit was observed at lower eosinophil counts, less than approximately 200 eosinophils per [mcL],” the investigators wrote.
“Overall, these results show the potential use of blood eosinophil counts in conjunction with smoking status to predict the magnitude of ICS response within a dual or triple-combination therapy,” the investigators concluded. “Future approaches to the pharmacological management of COPD should move beyond the simple dichotomization of each clinical or biomarker variable, toward more complex algorithms that integrate the interactions between important variables including exacerbation history, smoking status, and blood eosinophil counts.”
The study was funded by GlaxoSmithKline. The investigators disclosed additional relationships with AstraZeneca, Boehringer Ingelheim, Chiesi, CSA Medical, and others.
SOURCE: Pascoe S et al. Lancet Resp Med. 2019 Jul 4. doi: 10.1016/S2213-2600(19)30190-0.
Triple therapy with an inhaled corticosteroid is particularly helpful for patients with chronic obstructive pulmonary disease (COPD) who have high baseline eosinophil counts, a trial involving more than 10,000 patients found.
Former smokers received greater benefit from inhaled corticosteroids (ICS) than did current smokers, reported lead author Steven Pascoe, MBBS, of GlaxoSmithKline and colleagues. The investigators noted that these findings can help personalize therapy for patients with COPD, which can be challenging to treat because of its heterogeneity. The study was published in Lancet Respiratory Medicine.
The phase 3 IMPACT trial compared single-inhaler fluticasone furoate–umeclidinium–vilanterol with umeclidinium-vilanterol and fluticasone furoate–vilanterol in patients with moderate to very severe COPD at high risk of exacerbation. Of the 10,333 patients involved, approximately one-quarter (26%) had one or more severe exacerbations in the previous year and half (47%) had two or more moderate exacerbations in the same time period. All patients were symptomatic and were aged 40 years or older. A variety of baseline and demographic patient characteristics were recorded, including blood eosinophil count, smoking status, and others. Responses to therapy were measured with trough forced expiratory volume in 1 second (FEV1), symptom scoring, and a quality of life questionnaire.
After 52 weeks, results showed that higher baseline eosinophil counts were associated with progressively greater benefits in favor of triple therapy. For patients with baseline blood eosinophil counts of at least 310 cells per mcL, triple therapy was associated with about half as many moderate and severe exacerbations as treatment with umeclidinium-vilanterol (rate ratio = 0.56; 95% confidence interval, 0.47-0.66). For patients with less than 90 cells per mcL at baseline, the rate ratio for the same two regimens was 0.88, but with a confidence interval crossing 1 (0.74-1.04). For fluticasone furoate–vilanterol vs. umeclidinium-vilanterol, high baseline eosinophil count again demonstrated its predictive power for ICS efficacy, again with an associated rate ratio of 0.56 (0.47-0.66), compared with 1.09 (0.91-1.29) for patients below the lower threshold. Symptom scoring, quality of life, and FEV1 followed a similar trend, although the investigators noted that this was “less marked” for FEV1. Although the trend held regardless of smoking status, benefits were more pronounced among former smokers than current smokers.
“In former smokers, ICS benefits were observed at all blood eosinophil counts when comparing triple therapy with umeclidinium-vilanterol, whereas in current smokers no ICS benefit was observed at lower eosinophil counts, less than approximately 200 eosinophils per [mcL],” the investigators wrote.
“Overall, these results show the potential use of blood eosinophil counts in conjunction with smoking status to predict the magnitude of ICS response within a dual or triple-combination therapy,” the investigators concluded. “Future approaches to the pharmacological management of COPD should move beyond the simple dichotomization of each clinical or biomarker variable, toward more complex algorithms that integrate the interactions between important variables including exacerbation history, smoking status, and blood eosinophil counts.”
The study was funded by GlaxoSmithKline. The investigators disclosed additional relationships with AstraZeneca, Boehringer Ingelheim, Chiesi, CSA Medical, and others.
SOURCE: Pascoe S et al. Lancet Resp Med. 2019 Jul 4. doi: 10.1016/S2213-2600(19)30190-0.
Triple therapy with an inhaled corticosteroid is particularly helpful for patients with chronic obstructive pulmonary disease (COPD) who have high baseline eosinophil counts, a trial involving more than 10,000 patients found.
Former smokers received greater benefit from inhaled corticosteroids (ICS) than did current smokers, reported lead author Steven Pascoe, MBBS, of GlaxoSmithKline and colleagues. The investigators noted that these findings can help personalize therapy for patients with COPD, which can be challenging to treat because of its heterogeneity. The study was published in Lancet Respiratory Medicine.
The phase 3 IMPACT trial compared single-inhaler fluticasone furoate–umeclidinium–vilanterol with umeclidinium-vilanterol and fluticasone furoate–vilanterol in patients with moderate to very severe COPD at high risk of exacerbation. Of the 10,333 patients involved, approximately one-quarter (26%) had one or more severe exacerbations in the previous year and half (47%) had two or more moderate exacerbations in the same time period. All patients were symptomatic and were aged 40 years or older. A variety of baseline and demographic patient characteristics were recorded, including blood eosinophil count, smoking status, and others. Responses to therapy were measured with trough forced expiratory volume in 1 second (FEV1), symptom scoring, and a quality of life questionnaire.
After 52 weeks, results showed that higher baseline eosinophil counts were associated with progressively greater benefits in favor of triple therapy. For patients with baseline blood eosinophil counts of at least 310 cells per mcL, triple therapy was associated with about half as many moderate and severe exacerbations as treatment with umeclidinium-vilanterol (rate ratio = 0.56; 95% confidence interval, 0.47-0.66). For patients with less than 90 cells per mcL at baseline, the rate ratio for the same two regimens was 0.88, but with a confidence interval crossing 1 (0.74-1.04). For fluticasone furoate–vilanterol vs. umeclidinium-vilanterol, high baseline eosinophil count again demonstrated its predictive power for ICS efficacy, again with an associated rate ratio of 0.56 (0.47-0.66), compared with 1.09 (0.91-1.29) for patients below the lower threshold. Symptom scoring, quality of life, and FEV1 followed a similar trend, although the investigators noted that this was “less marked” for FEV1. Although the trend held regardless of smoking status, benefits were more pronounced among former smokers than current smokers.
“In former smokers, ICS benefits were observed at all blood eosinophil counts when comparing triple therapy with umeclidinium-vilanterol, whereas in current smokers no ICS benefit was observed at lower eosinophil counts, less than approximately 200 eosinophils per [mcL],” the investigators wrote.
“Overall, these results show the potential use of blood eosinophil counts in conjunction with smoking status to predict the magnitude of ICS response within a dual or triple-combination therapy,” the investigators concluded. “Future approaches to the pharmacological management of COPD should move beyond the simple dichotomization of each clinical or biomarker variable, toward more complex algorithms that integrate the interactions between important variables including exacerbation history, smoking status, and blood eosinophil counts.”
The study was funded by GlaxoSmithKline. The investigators disclosed additional relationships with AstraZeneca, Boehringer Ingelheim, Chiesi, CSA Medical, and others.
SOURCE: Pascoe S et al. Lancet Resp Med. 2019 Jul 4. doi: 10.1016/S2213-2600(19)30190-0.
FROM LANCET RESPIRATORY MEDICINE
Measles cases have slowed but not stopped
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.
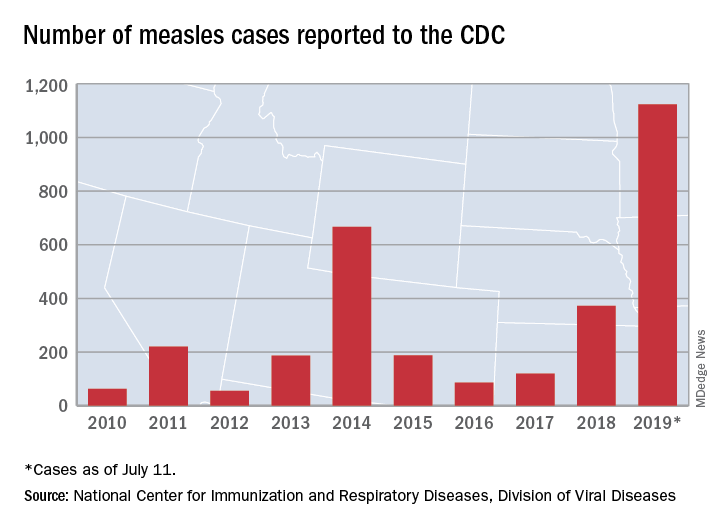
That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.

That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.
The United States continues to slowly add new cases of measles to 2019’s postelimination-record total, but California was officially removed from the outbreak list this week, according to the Centers for Disease Control and Prevention.

That is the highest number of cases reported since measles was declared eliminated in 2000 and the most in a single year since 1992.
The end of outbreak-related activity in California leaves three locations still dealing with ongoing cases: Rockland County, N.Y.; New York City; and King, Pierce, and Snohomish Counties in Washington, the CDC said.
Those three jurisdictions currently report the following:
- reported four new cases from July 3 to July 11 and is up to 175 cases for the year.
- had one new case from July 1 to July 8 and is now at 564 for the year.
- reported two cases from July 1 to July 10 and is now at 10 for the year (the other two counties have a total of three cases). Clark County in Washington reported 71 cases in an earlier, unrelated outbreak.