User login
COVID-19 vaccination rate rising quickly among adolescents
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.
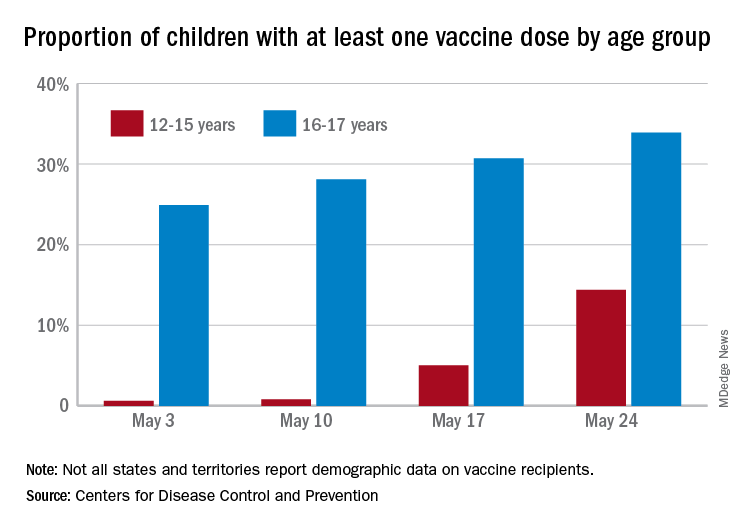
As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
Study identifies strong association between use of rotavirus vaccines, 60% reduction in infection
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
FROM JAMA PEDIATRICS
CDC recommends use of Pfizer’s COVID vaccine in 12- to 15-year-olds
The Centers for Disease Control and Prevention’s director Rochelle Walensky, MD, signed off on an advisory panel’s recommendation May 12 endorsing the use of the Pfizer-BioNTech COVID-19 vaccine in adolescents aged 12-15 years.
Earlier in the day the CDC’s Advisory Committee on Immunization Practices voted 14-0 in favor of the safety and effectiveness of the vaccine in younger teens.
Dr. Walensky said in an official statement.
The Food and Drug Administration on May 10 issued an emergency use authorization (EUA) for the Pfizer-BioNTech COVID-19 vaccine for the prevention of COVID-19 in individuals 12-15 years old. The FDA first cleared the Pfizer-BioNTech vaccine through an EUA in December 2020 for those ages 16 and older. Pfizer this month also initiated steps with the FDA toward a full approval of its vaccine.
Dr. Walenksy urged parents to seriously consider vaccinating their children.
“Understandably, some parents want more information before their children receive a vaccine,” she said. “I encourage parents with questions to talk to your child’s healthcare provider or your family doctor to learn more about the vaccine.”
Vaccine “safe and effective”
Separately, the American Academy of Pediatrics issued a statement May 12 in support of vaccinating all children ages 12 and older who are eligible for the federally authorized COVID-19 vaccine.
“As a pediatrician and a parent, I have looked forward to getting my own children and patients vaccinated, and I am thrilled that those ages 12 and older can now be protected,” said AAP President Lee Savio Beers, MD, in a statement. “The data continue to show that this vaccine is safe and effective. I urge all parents to call their pediatrician to learn more about how to get their children and teens vaccinated.”
The expanded clearance for the Pfizer vaccine is seen as a critical step for allowing teens to resume activities on which they missed out during the pandemic.
“We’ve seen the harm done to children’s mental and emotional health as they’ve missed out on so many experiences during the pandemic,” Dr. Beers said. “Vaccinating children will protect them and allow them to fully engage in all of the activities – school, sports, socializing with friends and family – that are so important to their health and development.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s director Rochelle Walensky, MD, signed off on an advisory panel’s recommendation May 12 endorsing the use of the Pfizer-BioNTech COVID-19 vaccine in adolescents aged 12-15 years.
Earlier in the day the CDC’s Advisory Committee on Immunization Practices voted 14-0 in favor of the safety and effectiveness of the vaccine in younger teens.
Dr. Walensky said in an official statement.
The Food and Drug Administration on May 10 issued an emergency use authorization (EUA) for the Pfizer-BioNTech COVID-19 vaccine for the prevention of COVID-19 in individuals 12-15 years old. The FDA first cleared the Pfizer-BioNTech vaccine through an EUA in December 2020 for those ages 16 and older. Pfizer this month also initiated steps with the FDA toward a full approval of its vaccine.
Dr. Walenksy urged parents to seriously consider vaccinating their children.
“Understandably, some parents want more information before their children receive a vaccine,” she said. “I encourage parents with questions to talk to your child’s healthcare provider or your family doctor to learn more about the vaccine.”
Vaccine “safe and effective”
Separately, the American Academy of Pediatrics issued a statement May 12 in support of vaccinating all children ages 12 and older who are eligible for the federally authorized COVID-19 vaccine.
“As a pediatrician and a parent, I have looked forward to getting my own children and patients vaccinated, and I am thrilled that those ages 12 and older can now be protected,” said AAP President Lee Savio Beers, MD, in a statement. “The data continue to show that this vaccine is safe and effective. I urge all parents to call their pediatrician to learn more about how to get their children and teens vaccinated.”
The expanded clearance for the Pfizer vaccine is seen as a critical step for allowing teens to resume activities on which they missed out during the pandemic.
“We’ve seen the harm done to children’s mental and emotional health as they’ve missed out on so many experiences during the pandemic,” Dr. Beers said. “Vaccinating children will protect them and allow them to fully engage in all of the activities – school, sports, socializing with friends and family – that are so important to their health and development.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s director Rochelle Walensky, MD, signed off on an advisory panel’s recommendation May 12 endorsing the use of the Pfizer-BioNTech COVID-19 vaccine in adolescents aged 12-15 years.
Earlier in the day the CDC’s Advisory Committee on Immunization Practices voted 14-0 in favor of the safety and effectiveness of the vaccine in younger teens.
Dr. Walensky said in an official statement.
The Food and Drug Administration on May 10 issued an emergency use authorization (EUA) for the Pfizer-BioNTech COVID-19 vaccine for the prevention of COVID-19 in individuals 12-15 years old. The FDA first cleared the Pfizer-BioNTech vaccine through an EUA in December 2020 for those ages 16 and older. Pfizer this month also initiated steps with the FDA toward a full approval of its vaccine.
Dr. Walenksy urged parents to seriously consider vaccinating their children.
“Understandably, some parents want more information before their children receive a vaccine,” she said. “I encourage parents with questions to talk to your child’s healthcare provider or your family doctor to learn more about the vaccine.”
Vaccine “safe and effective”
Separately, the American Academy of Pediatrics issued a statement May 12 in support of vaccinating all children ages 12 and older who are eligible for the federally authorized COVID-19 vaccine.
“As a pediatrician and a parent, I have looked forward to getting my own children and patients vaccinated, and I am thrilled that those ages 12 and older can now be protected,” said AAP President Lee Savio Beers, MD, in a statement. “The data continue to show that this vaccine is safe and effective. I urge all parents to call their pediatrician to learn more about how to get their children and teens vaccinated.”
The expanded clearance for the Pfizer vaccine is seen as a critical step for allowing teens to resume activities on which they missed out during the pandemic.
“We’ve seen the harm done to children’s mental and emotional health as they’ve missed out on so many experiences during the pandemic,” Dr. Beers said. “Vaccinating children will protect them and allow them to fully engage in all of the activities – school, sports, socializing with friends and family – that are so important to their health and development.”
A version of this article first appeared on Medscape.com.
States ready plans to get Pfizer COVID vaccine to younger teens
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
FDA set to okay Pfizer vaccine in younger teens
The Food and Drug Administration could expand the use of the Pfizer COVID-19 vaccine to teens early next week, The New York Times and CNN reported, both citing unnamed officials familiar with the agency’s plans.
In late March, Pfizer submitted data to the FDA showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12 to 15. Their vaccine is already authorized for use teens and adults ages 16 and older.
The move would make about 17 million more Americans eligible for vaccination and would be a major step toward getting both adolescents and teens back into classrooms full time by next fall.
“Across the globe, we are longing for a normal life. This is especially true for our children. The initial results we have seen in the adolescent studies suggest that children are particularly well protected by vaccination, which is very encouraging given the trends we have seen in recent weeks regarding the spread of the B.1.1.7 U.K. variant,” Ugur Sahin, CEO and co-founder of Pfizer partner BioNTech, said in a March 31 press release.
Getting schools fully reopened for in-person learning has been a goal of both the Trump and Biden administrations, but it has been tricky to pull off, as some parents and teachers have been reluctant to return to classrooms with so much uncertainty about the risk and the role of children in spreading the virus.
A recent study of roughly 150,000 school-aged children in Israel found that while kids under age 10 were unlikely to catch or spread the virus as they reentered classrooms. Older children, though, were a different story. The study found that children ages 10-19 had risks of catching the virus that were as high as adults ages 20-60.
The risk for severe illness and death from COVID-19 rises with age.
Children and teens are at relatively low risk from severe outcomes after a COVID-19 infection compared to adults, but they can catch it and some will get really sick with it, especially if they have an underlying health condition, like obesity or asthma that makes them more vulnerable.
Beyond the initial infection, children can get a rare late complication called MIS-C, that while treatable, can be severe and requires hospitalization. Emerging reports also suggest there are some kids that become long haulers in much the same way adults do, dealing with lingering problems for months after they first get sick.
As new variants of the coronavirus circulate in the United States, some states have seen big increases in the number of children and teens with COVID. In Michigan, for example, which recently dealt with a spring surge of cases dominated by the B.1.1.7 variant, cases in children and teens quadrupled in April compared to February.
Beyond individual protection, vaccinating children and teens has been seen as important to achieving strong community protection, or herd immunity, against the new coronavirus.
If the FDA expands the authorization for the Pfizer vaccine, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will likely meet to review data on the safety and efficacy of the vaccine. The committee may then vote on new recommendations for use of the vaccine in the United States.
Not everyone agrees with the idea that American adolescents, who are at relatively low risk of bad outcomes, could get access to COVID vaccines ahead of vulnerable essential workers and seniors in other parts of the world that are still fighting the pandemic with little access to vaccines.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration could expand the use of the Pfizer COVID-19 vaccine to teens early next week, The New York Times and CNN reported, both citing unnamed officials familiar with the agency’s plans.
In late March, Pfizer submitted data to the FDA showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12 to 15. Their vaccine is already authorized for use teens and adults ages 16 and older.
The move would make about 17 million more Americans eligible for vaccination and would be a major step toward getting both adolescents and teens back into classrooms full time by next fall.
“Across the globe, we are longing for a normal life. This is especially true for our children. The initial results we have seen in the adolescent studies suggest that children are particularly well protected by vaccination, which is very encouraging given the trends we have seen in recent weeks regarding the spread of the B.1.1.7 U.K. variant,” Ugur Sahin, CEO and co-founder of Pfizer partner BioNTech, said in a March 31 press release.
Getting schools fully reopened for in-person learning has been a goal of both the Trump and Biden administrations, but it has been tricky to pull off, as some parents and teachers have been reluctant to return to classrooms with so much uncertainty about the risk and the role of children in spreading the virus.
A recent study of roughly 150,000 school-aged children in Israel found that while kids under age 10 were unlikely to catch or spread the virus as they reentered classrooms. Older children, though, were a different story. The study found that children ages 10-19 had risks of catching the virus that were as high as adults ages 20-60.
The risk for severe illness and death from COVID-19 rises with age.
Children and teens are at relatively low risk from severe outcomes after a COVID-19 infection compared to adults, but they can catch it and some will get really sick with it, especially if they have an underlying health condition, like obesity or asthma that makes them more vulnerable.
Beyond the initial infection, children can get a rare late complication called MIS-C, that while treatable, can be severe and requires hospitalization. Emerging reports also suggest there are some kids that become long haulers in much the same way adults do, dealing with lingering problems for months after they first get sick.
As new variants of the coronavirus circulate in the United States, some states have seen big increases in the number of children and teens with COVID. In Michigan, for example, which recently dealt with a spring surge of cases dominated by the B.1.1.7 variant, cases in children and teens quadrupled in April compared to February.
Beyond individual protection, vaccinating children and teens has been seen as important to achieving strong community protection, or herd immunity, against the new coronavirus.
If the FDA expands the authorization for the Pfizer vaccine, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will likely meet to review data on the safety and efficacy of the vaccine. The committee may then vote on new recommendations for use of the vaccine in the United States.
Not everyone agrees with the idea that American adolescents, who are at relatively low risk of bad outcomes, could get access to COVID vaccines ahead of vulnerable essential workers and seniors in other parts of the world that are still fighting the pandemic with little access to vaccines.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration could expand the use of the Pfizer COVID-19 vaccine to teens early next week, The New York Times and CNN reported, both citing unnamed officials familiar with the agency’s plans.
In late March, Pfizer submitted data to the FDA showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12 to 15. Their vaccine is already authorized for use teens and adults ages 16 and older.
The move would make about 17 million more Americans eligible for vaccination and would be a major step toward getting both adolescents and teens back into classrooms full time by next fall.
“Across the globe, we are longing for a normal life. This is especially true for our children. The initial results we have seen in the adolescent studies suggest that children are particularly well protected by vaccination, which is very encouraging given the trends we have seen in recent weeks regarding the spread of the B.1.1.7 U.K. variant,” Ugur Sahin, CEO and co-founder of Pfizer partner BioNTech, said in a March 31 press release.
Getting schools fully reopened for in-person learning has been a goal of both the Trump and Biden administrations, but it has been tricky to pull off, as some parents and teachers have been reluctant to return to classrooms with so much uncertainty about the risk and the role of children in spreading the virus.
A recent study of roughly 150,000 school-aged children in Israel found that while kids under age 10 were unlikely to catch or spread the virus as they reentered classrooms. Older children, though, were a different story. The study found that children ages 10-19 had risks of catching the virus that were as high as adults ages 20-60.
The risk for severe illness and death from COVID-19 rises with age.
Children and teens are at relatively low risk from severe outcomes after a COVID-19 infection compared to adults, but they can catch it and some will get really sick with it, especially if they have an underlying health condition, like obesity or asthma that makes them more vulnerable.
Beyond the initial infection, children can get a rare late complication called MIS-C, that while treatable, can be severe and requires hospitalization. Emerging reports also suggest there are some kids that become long haulers in much the same way adults do, dealing with lingering problems for months after they first get sick.
As new variants of the coronavirus circulate in the United States, some states have seen big increases in the number of children and teens with COVID. In Michigan, for example, which recently dealt with a spring surge of cases dominated by the B.1.1.7 variant, cases in children and teens quadrupled in April compared to February.
Beyond individual protection, vaccinating children and teens has been seen as important to achieving strong community protection, or herd immunity, against the new coronavirus.
If the FDA expands the authorization for the Pfizer vaccine, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will likely meet to review data on the safety and efficacy of the vaccine. The committee may then vote on new recommendations for use of the vaccine in the United States.
Not everyone agrees with the idea that American adolescents, who are at relatively low risk of bad outcomes, could get access to COVID vaccines ahead of vulnerable essential workers and seniors in other parts of the world that are still fighting the pandemic with little access to vaccines.
A version of this article first appeared on WebMD.com.
HPV vaccination rates continue to climb among young adults in U.S.
Although vaccination rates against the human papillomavirus remain low for young adults across the United States, the number of self-reported HPV vaccinations among women and men aged between 18 and 21 years has markedly increased since 2010, according to new research findings.
The findings were published online April 27, 2021, as a research letter in JAMA.
In 2006, the Food and Drug Administration approved the HPV vaccine for the prevention of cervical cancer and genital warts in female patients. Three years later, the FDA approved the vaccine for the prevention of anogenital cancer and warts in male patients.
The Advisory Committee on Immunization Practices and the Centers for Disease Control and Prevention recommend two doses of the HPV vaccine for children aged 11-12 years. Adolescents and young adults may need three doses over the course of 6 months if they start their vaccine series on or following their 15th birthday.
For persons who have not previously received the HPV vaccine or who did not receive adequate doses, the HPV vaccine is recommended through age 26. Data on the rates of vaccination among young adults between 18 and 21 years of age in the United States are sparse, and it is not known how well vaccination programs are progressing in the country.
In the recently published JAMA research letter, investigators from the University of Michigan, Ann Arbor, examined data for the period 2010-2018 from the cross-sectional National Health Interview Survey. Respondents included in the analysis were aged 18-21 years. They were asked whether they had received the HPV vaccine before age 18 and at what age they had been vaccinated against the virus.
The researchers also assessed whether the respondents had received any HPV vaccine dose between the ages of 18 and 21 years. The findings were limited to self-reported vaccination status.
In total, 6,606 women and 6,038 men were included in the analysis. Approximately 42% of women and 16% of men said they had received at least one HPV vaccine dose at any age. The proportion of female patients who reported receiving an HPV vaccine dose significantly increased from 32% in 2010 to 55% in 2018 (P =.001). Similarly, among men, the percentage significantly increased from 2% in 2010 to 34% in 2018 (P <.001).
Approximately 4% of the female respondents and 3% of the male respondents reported that they had received an HPV vaccine between the ages of 18 and 21 years; 46% of women and 29% of men who received the vaccine between these ages completed the recommended vaccination series.
Findings from the study highlight the continual need for improving vaccination rates among vulnerable populations. Lead study author Michelle Chen, MD, MHS, a professor in the department of otolaryngology–head and neck surgery at the University of Michigan, explained in an interview that there are multiple barriers to HPV vaccination among young adults. “These barriers to vaccination among young adults primarily include cost, lack of knowledge and awareness, missed opportunities for vaccination, rapidly changing guidelines, and initial gender-based guidelines,” said Dr. Chen.
Clinicians play a large role in improving vaccination rates among young adults, who may lack awareness of the overall importance of inoculation against the potentially debilitating and deadly virus. Dr. Chen noted that clinicians can lead the way by increasing gender-inclusive awareness of HPV-associated diseases and HPV vaccination, by performing routine vaccine eligibility assessments for young adults regardless of sex, by developing robust reminder and recall strategies to improve series completion rates, and by offering patients resources regarding assistance programs to address cost barriers for uninsured patients.
“Young adult men are particularly vulnerable [to HPV], because they start to age out of pediatric health practices,” added Dr. Chen. “Thus, a multilevel gender-inclusive approach is needed to target clinicians, patients, parents, and community-based organizations.”
Gypsyamber D’Souza, PhD, professor of epidemiology at Johns Hopkins University, Baltimore, said in an interview that the initial uptake of HPV vaccination was slow in the United States but that progress has been made in recent years among persons in the targeted age range of 11-12 years. “However, catch-up vaccination has lagged behind, and sadly, we’re still seeing low uptake in those older ages that are still eligible and where we know there still is tremendous benefit,” she said.
Dr. D’Souza is a lead investigator in the MOUTH trial, which is currently enrolling patients. That trial will examine potential biomarkers for oropharyngeal cancer risk among people with known risk factors for HPV who came of age prior to the rollout of the vaccine.
She explained that many parents want their children to be vaccinated for HPV after they hear about the vaccine, but because the health care system in the United States is an “opt-in” system, rather than an “opt-out” one, parents need to actively seek out vaccination. Children then move toward adulthood without having received the recommended vaccine course. “There are individuals who did not get vaccinated at the ages of 11 and 12 and then forget to ask about it later, or the provider asks about it and the patients don’t have enough information,” Dr. D’Souza said.
She noted that one reason why HPV vaccination rates remain low among young adults is that the vaccine is not often kept in stock other than in pediatric clinics. “Because vaccines expire and clinics don’t have a lot of people in that age group getting vaccinated, they may not have it regularly in stock, making this one reason it might be hard for someone to get vaccinated.”
The HPV vaccine is not effective for clearing HPV once a patient acquires the infection, she added. “So young adulthood is a critical time where we have individuals who still can benefit from being vaccinated, but if we wait too long, they’ll age out of those ages where we see the highest efficacy.”
Ultimately, said Dr. D’Souza, clinicians need to catch people at multiple time points and work to remove barriers to vaccination, including letting patients know that HPV vaccination is covered by insurance. “There’s a lot of opportunity to prevent future cancers in young adults by having care providers for that age group talk about the vaccine and remember to offer it.”
A version of this article first appeared on Medscape.com.
Although vaccination rates against the human papillomavirus remain low for young adults across the United States, the number of self-reported HPV vaccinations among women and men aged between 18 and 21 years has markedly increased since 2010, according to new research findings.
The findings were published online April 27, 2021, as a research letter in JAMA.
In 2006, the Food and Drug Administration approved the HPV vaccine for the prevention of cervical cancer and genital warts in female patients. Three years later, the FDA approved the vaccine for the prevention of anogenital cancer and warts in male patients.
The Advisory Committee on Immunization Practices and the Centers for Disease Control and Prevention recommend two doses of the HPV vaccine for children aged 11-12 years. Adolescents and young adults may need three doses over the course of 6 months if they start their vaccine series on or following their 15th birthday.
For persons who have not previously received the HPV vaccine or who did not receive adequate doses, the HPV vaccine is recommended through age 26. Data on the rates of vaccination among young adults between 18 and 21 years of age in the United States are sparse, and it is not known how well vaccination programs are progressing in the country.
In the recently published JAMA research letter, investigators from the University of Michigan, Ann Arbor, examined data for the period 2010-2018 from the cross-sectional National Health Interview Survey. Respondents included in the analysis were aged 18-21 years. They were asked whether they had received the HPV vaccine before age 18 and at what age they had been vaccinated against the virus.
The researchers also assessed whether the respondents had received any HPV vaccine dose between the ages of 18 and 21 years. The findings were limited to self-reported vaccination status.
In total, 6,606 women and 6,038 men were included in the analysis. Approximately 42% of women and 16% of men said they had received at least one HPV vaccine dose at any age. The proportion of female patients who reported receiving an HPV vaccine dose significantly increased from 32% in 2010 to 55% in 2018 (P =.001). Similarly, among men, the percentage significantly increased from 2% in 2010 to 34% in 2018 (P <.001).
Approximately 4% of the female respondents and 3% of the male respondents reported that they had received an HPV vaccine between the ages of 18 and 21 years; 46% of women and 29% of men who received the vaccine between these ages completed the recommended vaccination series.
Findings from the study highlight the continual need for improving vaccination rates among vulnerable populations. Lead study author Michelle Chen, MD, MHS, a professor in the department of otolaryngology–head and neck surgery at the University of Michigan, explained in an interview that there are multiple barriers to HPV vaccination among young adults. “These barriers to vaccination among young adults primarily include cost, lack of knowledge and awareness, missed opportunities for vaccination, rapidly changing guidelines, and initial gender-based guidelines,” said Dr. Chen.
Clinicians play a large role in improving vaccination rates among young adults, who may lack awareness of the overall importance of inoculation against the potentially debilitating and deadly virus. Dr. Chen noted that clinicians can lead the way by increasing gender-inclusive awareness of HPV-associated diseases and HPV vaccination, by performing routine vaccine eligibility assessments for young adults regardless of sex, by developing robust reminder and recall strategies to improve series completion rates, and by offering patients resources regarding assistance programs to address cost barriers for uninsured patients.
“Young adult men are particularly vulnerable [to HPV], because they start to age out of pediatric health practices,” added Dr. Chen. “Thus, a multilevel gender-inclusive approach is needed to target clinicians, patients, parents, and community-based organizations.”
Gypsyamber D’Souza, PhD, professor of epidemiology at Johns Hopkins University, Baltimore, said in an interview that the initial uptake of HPV vaccination was slow in the United States but that progress has been made in recent years among persons in the targeted age range of 11-12 years. “However, catch-up vaccination has lagged behind, and sadly, we’re still seeing low uptake in those older ages that are still eligible and where we know there still is tremendous benefit,” she said.
Dr. D’Souza is a lead investigator in the MOUTH trial, which is currently enrolling patients. That trial will examine potential biomarkers for oropharyngeal cancer risk among people with known risk factors for HPV who came of age prior to the rollout of the vaccine.
She explained that many parents want their children to be vaccinated for HPV after they hear about the vaccine, but because the health care system in the United States is an “opt-in” system, rather than an “opt-out” one, parents need to actively seek out vaccination. Children then move toward adulthood without having received the recommended vaccine course. “There are individuals who did not get vaccinated at the ages of 11 and 12 and then forget to ask about it later, or the provider asks about it and the patients don’t have enough information,” Dr. D’Souza said.
She noted that one reason why HPV vaccination rates remain low among young adults is that the vaccine is not often kept in stock other than in pediatric clinics. “Because vaccines expire and clinics don’t have a lot of people in that age group getting vaccinated, they may not have it regularly in stock, making this one reason it might be hard for someone to get vaccinated.”
The HPV vaccine is not effective for clearing HPV once a patient acquires the infection, she added. “So young adulthood is a critical time where we have individuals who still can benefit from being vaccinated, but if we wait too long, they’ll age out of those ages where we see the highest efficacy.”
Ultimately, said Dr. D’Souza, clinicians need to catch people at multiple time points and work to remove barriers to vaccination, including letting patients know that HPV vaccination is covered by insurance. “There’s a lot of opportunity to prevent future cancers in young adults by having care providers for that age group talk about the vaccine and remember to offer it.”
A version of this article first appeared on Medscape.com.
Although vaccination rates against the human papillomavirus remain low for young adults across the United States, the number of self-reported HPV vaccinations among women and men aged between 18 and 21 years has markedly increased since 2010, according to new research findings.
The findings were published online April 27, 2021, as a research letter in JAMA.
In 2006, the Food and Drug Administration approved the HPV vaccine for the prevention of cervical cancer and genital warts in female patients. Three years later, the FDA approved the vaccine for the prevention of anogenital cancer and warts in male patients.
The Advisory Committee on Immunization Practices and the Centers for Disease Control and Prevention recommend two doses of the HPV vaccine for children aged 11-12 years. Adolescents and young adults may need three doses over the course of 6 months if they start their vaccine series on or following their 15th birthday.
For persons who have not previously received the HPV vaccine or who did not receive adequate doses, the HPV vaccine is recommended through age 26. Data on the rates of vaccination among young adults between 18 and 21 years of age in the United States are sparse, and it is not known how well vaccination programs are progressing in the country.
In the recently published JAMA research letter, investigators from the University of Michigan, Ann Arbor, examined data for the period 2010-2018 from the cross-sectional National Health Interview Survey. Respondents included in the analysis were aged 18-21 years. They were asked whether they had received the HPV vaccine before age 18 and at what age they had been vaccinated against the virus.
The researchers also assessed whether the respondents had received any HPV vaccine dose between the ages of 18 and 21 years. The findings were limited to self-reported vaccination status.
In total, 6,606 women and 6,038 men were included in the analysis. Approximately 42% of women and 16% of men said they had received at least one HPV vaccine dose at any age. The proportion of female patients who reported receiving an HPV vaccine dose significantly increased from 32% in 2010 to 55% in 2018 (P =.001). Similarly, among men, the percentage significantly increased from 2% in 2010 to 34% in 2018 (P <.001).
Approximately 4% of the female respondents and 3% of the male respondents reported that they had received an HPV vaccine between the ages of 18 and 21 years; 46% of women and 29% of men who received the vaccine between these ages completed the recommended vaccination series.
Findings from the study highlight the continual need for improving vaccination rates among vulnerable populations. Lead study author Michelle Chen, MD, MHS, a professor in the department of otolaryngology–head and neck surgery at the University of Michigan, explained in an interview that there are multiple barriers to HPV vaccination among young adults. “These barriers to vaccination among young adults primarily include cost, lack of knowledge and awareness, missed opportunities for vaccination, rapidly changing guidelines, and initial gender-based guidelines,” said Dr. Chen.
Clinicians play a large role in improving vaccination rates among young adults, who may lack awareness of the overall importance of inoculation against the potentially debilitating and deadly virus. Dr. Chen noted that clinicians can lead the way by increasing gender-inclusive awareness of HPV-associated diseases and HPV vaccination, by performing routine vaccine eligibility assessments for young adults regardless of sex, by developing robust reminder and recall strategies to improve series completion rates, and by offering patients resources regarding assistance programs to address cost barriers for uninsured patients.
“Young adult men are particularly vulnerable [to HPV], because they start to age out of pediatric health practices,” added Dr. Chen. “Thus, a multilevel gender-inclusive approach is needed to target clinicians, patients, parents, and community-based organizations.”
Gypsyamber D’Souza, PhD, professor of epidemiology at Johns Hopkins University, Baltimore, said in an interview that the initial uptake of HPV vaccination was slow in the United States but that progress has been made in recent years among persons in the targeted age range of 11-12 years. “However, catch-up vaccination has lagged behind, and sadly, we’re still seeing low uptake in those older ages that are still eligible and where we know there still is tremendous benefit,” she said.
Dr. D’Souza is a lead investigator in the MOUTH trial, which is currently enrolling patients. That trial will examine potential biomarkers for oropharyngeal cancer risk among people with known risk factors for HPV who came of age prior to the rollout of the vaccine.
She explained that many parents want their children to be vaccinated for HPV after they hear about the vaccine, but because the health care system in the United States is an “opt-in” system, rather than an “opt-out” one, parents need to actively seek out vaccination. Children then move toward adulthood without having received the recommended vaccine course. “There are individuals who did not get vaccinated at the ages of 11 and 12 and then forget to ask about it later, or the provider asks about it and the patients don’t have enough information,” Dr. D’Souza said.
She noted that one reason why HPV vaccination rates remain low among young adults is that the vaccine is not often kept in stock other than in pediatric clinics. “Because vaccines expire and clinics don’t have a lot of people in that age group getting vaccinated, they may not have it regularly in stock, making this one reason it might be hard for someone to get vaccinated.”
The HPV vaccine is not effective for clearing HPV once a patient acquires the infection, she added. “So young adulthood is a critical time where we have individuals who still can benefit from being vaccinated, but if we wait too long, they’ll age out of those ages where we see the highest efficacy.”
Ultimately, said Dr. D’Souza, clinicians need to catch people at multiple time points and work to remove barriers to vaccination, including letting patients know that HPV vaccination is covered by insurance. “There’s a lot of opportunity to prevent future cancers in young adults by having care providers for that age group talk about the vaccine and remember to offer it.”
A version of this article first appeared on Medscape.com.
Pfizer and Moderna vaccines appear safe, effective during pregnancy
The Pfizer and Moderna COVID-19 vaccines appear to be safe in pregnant patients, according to preliminary findings published in the New England Journal of Medicine.
The Centers for Disease Control and Prevention have said pregnant people have an increased risk of being severely ill from COVID-19; however, this group was excluded from major clinical trials that led up to the current vaccine approvals.
But based on the new findings, Rochelle Walensky, MD, director of the CDC, announced during a White House COVID-19 briefing that the CDC recommends that pregnant people receive the COVID-19 vaccine.
The new study, which analyzed data between Dec. 14, 2020, and Feb. 28, 2021, from three federal databases, adds to a pool of limited data about the safety and efficacy of the vaccine in pregnant persons. Researchers did not include people who received the Johnson & Johnson vaccine because it received emergency use authorization on Feb. 27, just 1 day before they study’s cutoff.
“Our hope is that these initial data will be reassuring to pregnant people and their health care providers as well as the public, and contribute to increasing vaccination rates,” study author Christine Olson, MD, said in an interview. “While the data are preliminary and will continue to be analyzed as more reports become available, our findings are reassuring.”
For the study, Dr. Olson and colleagues analyzed v-safe survey data, data from those enrolled in the v-safe pregnancy registry, and Vaccine Adverse Event Reporting System (VAERS) reports.
Researchers found that 86% of pregnancies resulted in a live birth, 12.6 % resulted in spontaneous abortions, and 0.1% resulted in stillbirth. They also found that, among the live births, 9.4% were preterm, 3.2% of babies were small for their gestational age, and 2.2% had congenital anomalies.
Researchers also found that injection-site pain, fatigue, and headaches were reported more frequently in pregnant patients than among those who were not pregnant. Among VAERS reports, they found that 70% of adverse events were nonpregnancy specific. Nearly 30% involved pregnancy- or neonatal-specific adverse events. The most frequently reported pregnancy-related events were spontaneous abortions, followed by stillbirths, premature rupture of membranes and vaginal bleeding.
“I think the results are actually quite reassuring as the proportion of the pregnancy outcomes, such as pregnancy loss and health effects to the newborns, are really quite consistent with what we’d expect in the background rate of the population,” Dr. Walensky said in a podcast accompanying the study. “So this study adds to growing evidence confirming that pregnant people develop a robust immune response to COVID-19 vaccination without so far seeing any adverse events to the mom or the fetus.”
Researchers said limitations of the study include the accuracy of self-reported data, and there being limited information on other potential risk factors for adverse pregnancies and neonatal outcomes. They acknowledged that continuous monitoring is needed to look at maternal safety and pregnancy outcomes in earlier stages of pregnancy and during the preconception period.
David Jaspan, DO, chair of the department of obstetrics and gynecology at Einstein Medical Center, Philadelphia, who was not involved with the study, said in an interview that, despite the limitations, the study provides much-needed insight on the vaccine’s safety and efficacy in pregnant patients.
“In December we had no data for any pregnant patient,” Dr. Jaspan said. “And now just 4 short months later, this paper [has data from] at least had 35,000 people. We can’t answer every question, but we have more answers today than we had just 4 months ago.”
Dr. Olson hopes the present data is enough to help inform decision-making of pregnant patients and their health care providers when it comes to deciding to get the COVID-19 vaccination.
The study author and experts interviewed disclosed no relevant financial relationships.
The Pfizer and Moderna COVID-19 vaccines appear to be safe in pregnant patients, according to preliminary findings published in the New England Journal of Medicine.
The Centers for Disease Control and Prevention have said pregnant people have an increased risk of being severely ill from COVID-19; however, this group was excluded from major clinical trials that led up to the current vaccine approvals.
But based on the new findings, Rochelle Walensky, MD, director of the CDC, announced during a White House COVID-19 briefing that the CDC recommends that pregnant people receive the COVID-19 vaccine.
The new study, which analyzed data between Dec. 14, 2020, and Feb. 28, 2021, from three federal databases, adds to a pool of limited data about the safety and efficacy of the vaccine in pregnant persons. Researchers did not include people who received the Johnson & Johnson vaccine because it received emergency use authorization on Feb. 27, just 1 day before they study’s cutoff.
“Our hope is that these initial data will be reassuring to pregnant people and their health care providers as well as the public, and contribute to increasing vaccination rates,” study author Christine Olson, MD, said in an interview. “While the data are preliminary and will continue to be analyzed as more reports become available, our findings are reassuring.”
For the study, Dr. Olson and colleagues analyzed v-safe survey data, data from those enrolled in the v-safe pregnancy registry, and Vaccine Adverse Event Reporting System (VAERS) reports.
Researchers found that 86% of pregnancies resulted in a live birth, 12.6 % resulted in spontaneous abortions, and 0.1% resulted in stillbirth. They also found that, among the live births, 9.4% were preterm, 3.2% of babies were small for their gestational age, and 2.2% had congenital anomalies.
Researchers also found that injection-site pain, fatigue, and headaches were reported more frequently in pregnant patients than among those who were not pregnant. Among VAERS reports, they found that 70% of adverse events were nonpregnancy specific. Nearly 30% involved pregnancy- or neonatal-specific adverse events. The most frequently reported pregnancy-related events were spontaneous abortions, followed by stillbirths, premature rupture of membranes and vaginal bleeding.
“I think the results are actually quite reassuring as the proportion of the pregnancy outcomes, such as pregnancy loss and health effects to the newborns, are really quite consistent with what we’d expect in the background rate of the population,” Dr. Walensky said in a podcast accompanying the study. “So this study adds to growing evidence confirming that pregnant people develop a robust immune response to COVID-19 vaccination without so far seeing any adverse events to the mom or the fetus.”
Researchers said limitations of the study include the accuracy of self-reported data, and there being limited information on other potential risk factors for adverse pregnancies and neonatal outcomes. They acknowledged that continuous monitoring is needed to look at maternal safety and pregnancy outcomes in earlier stages of pregnancy and during the preconception period.
David Jaspan, DO, chair of the department of obstetrics and gynecology at Einstein Medical Center, Philadelphia, who was not involved with the study, said in an interview that, despite the limitations, the study provides much-needed insight on the vaccine’s safety and efficacy in pregnant patients.
“In December we had no data for any pregnant patient,” Dr. Jaspan said. “And now just 4 short months later, this paper [has data from] at least had 35,000 people. We can’t answer every question, but we have more answers today than we had just 4 months ago.”
Dr. Olson hopes the present data is enough to help inform decision-making of pregnant patients and their health care providers when it comes to deciding to get the COVID-19 vaccination.
The study author and experts interviewed disclosed no relevant financial relationships.
The Pfizer and Moderna COVID-19 vaccines appear to be safe in pregnant patients, according to preliminary findings published in the New England Journal of Medicine.
The Centers for Disease Control and Prevention have said pregnant people have an increased risk of being severely ill from COVID-19; however, this group was excluded from major clinical trials that led up to the current vaccine approvals.
But based on the new findings, Rochelle Walensky, MD, director of the CDC, announced during a White House COVID-19 briefing that the CDC recommends that pregnant people receive the COVID-19 vaccine.
The new study, which analyzed data between Dec. 14, 2020, and Feb. 28, 2021, from three federal databases, adds to a pool of limited data about the safety and efficacy of the vaccine in pregnant persons. Researchers did not include people who received the Johnson & Johnson vaccine because it received emergency use authorization on Feb. 27, just 1 day before they study’s cutoff.
“Our hope is that these initial data will be reassuring to pregnant people and their health care providers as well as the public, and contribute to increasing vaccination rates,” study author Christine Olson, MD, said in an interview. “While the data are preliminary and will continue to be analyzed as more reports become available, our findings are reassuring.”
For the study, Dr. Olson and colleagues analyzed v-safe survey data, data from those enrolled in the v-safe pregnancy registry, and Vaccine Adverse Event Reporting System (VAERS) reports.
Researchers found that 86% of pregnancies resulted in a live birth, 12.6 % resulted in spontaneous abortions, and 0.1% resulted in stillbirth. They also found that, among the live births, 9.4% were preterm, 3.2% of babies were small for their gestational age, and 2.2% had congenital anomalies.
Researchers also found that injection-site pain, fatigue, and headaches were reported more frequently in pregnant patients than among those who were not pregnant. Among VAERS reports, they found that 70% of adverse events were nonpregnancy specific. Nearly 30% involved pregnancy- or neonatal-specific adverse events. The most frequently reported pregnancy-related events were spontaneous abortions, followed by stillbirths, premature rupture of membranes and vaginal bleeding.
“I think the results are actually quite reassuring as the proportion of the pregnancy outcomes, such as pregnancy loss and health effects to the newborns, are really quite consistent with what we’d expect in the background rate of the population,” Dr. Walensky said in a podcast accompanying the study. “So this study adds to growing evidence confirming that pregnant people develop a robust immune response to COVID-19 vaccination without so far seeing any adverse events to the mom or the fetus.”
Researchers said limitations of the study include the accuracy of self-reported data, and there being limited information on other potential risk factors for adverse pregnancies and neonatal outcomes. They acknowledged that continuous monitoring is needed to look at maternal safety and pregnancy outcomes in earlier stages of pregnancy and during the preconception period.
David Jaspan, DO, chair of the department of obstetrics and gynecology at Einstein Medical Center, Philadelphia, who was not involved with the study, said in an interview that, despite the limitations, the study provides much-needed insight on the vaccine’s safety and efficacy in pregnant patients.
“In December we had no data for any pregnant patient,” Dr. Jaspan said. “And now just 4 short months later, this paper [has data from] at least had 35,000 people. We can’t answer every question, but we have more answers today than we had just 4 months ago.”
Dr. Olson hopes the present data is enough to help inform decision-making of pregnant patients and their health care providers when it comes to deciding to get the COVID-19 vaccination.
The study author and experts interviewed disclosed no relevant financial relationships.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Feds lift pause of J&J COVID vaccine, add new warning
Use of the Johnson & Johnson COVID-19 vaccine should resume in the United States for all adults, the Food and Drug Administration and Centers for Disease Contol and Prevention said April 23, although health care providers should warn patients of the risk of developing the rare and serious blood clots that caused the agencies to pause the vaccine’s distribution earlier this month.
“What we are seeing is the overall rate of events was 1.9 cases per million people. In women 18 to 29 years there was an approximate 7 cases per million. The risk is even lower in women over the age of 50 at .9 cases per million,” CDC Director Rochelle Walensky, MD, said in a news briefing the same day.
In the end, the potential benefits of the vaccine far outweighed its risks.
“In terms of benefits, we found that for every 1 million doses of this vaccine, the J&J vaccine could prevent over 650 hospitalizations and 12 deaths among women ages 18-49,” Dr. Walensky said. The potential benefits to women over 50 were even greater: It could prevent 4,700 hospitalizations and 650 deaths.
“In the end, this vaccine was shown to be safe and effective for the vast majority of people,” Dr. Walensky said.
The recommendation to continue the vaccine’s rollout came barely 2 hours after a CDC Advisory Committee on Immunization Practices voted to recommend the pause be lifted. The vote was 10-4 with one abstention.
The decision also includes instructions for the warning directed at women under 50 who have an increased risk of a rare but serious blood clot disorder called thrombosis with thrombocytopenia syndrome (TTS).
As of April 21, 15 cases of TTS, all in women and 13 of them in women under 50, have been confirmed among 7.98 million doses of the J&J vaccine administered in the United States. Three women have died.
The FDA and CDC recommended the pause on April 13 after reports that 6 women developed a blood clotting disorder 6 to 13 days after they received the J&J vaccine.
William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville, and a non-voting ACIP member, said in an interview the panel made the right recommendation.
He applauded both the decision to restart the vaccine and the updated warning information that “will explain [TTS] more fully to people, particularly women, who are coming to be vaccinated.”
As to women in the risk group needing to have a choice of vaccines, Dr. Schaffner said that will be addressed differently across the country.
“Every provider will not have alternative vaccines in their location so there will be many different ways to do this. You may have to get this information and select which site you’re going to depending on which vaccine is available if this matter is important to you,” he noted.
ACIP made the decision after a 6-hour emergency meeting to hear evidence on the Johnson & Johnson vaccine's protective benefits against COVID-19 vs. risk of TTS.
In the CDC-FDA press briefing, Dr. Walensky pointed out that over the past few days, as regulators have reviewed the rare events, newly identified patients had been treated appropriately, without the use of heparin, which is not advised for treating TTS.
As a result, regulators felt as if their messages had gotten out to doctors who now knew how to take special precautions when treating patients with the disorder.
She said the Johnson & Johnson shot remained an important option because it was convenient to give and easier to store than the other vaccines currently authorized in the United States.
Peter Marks, MD, the director of FDA’s Center for Biologics Evaluation and Research, said the agency had already added information describing the risk of the rare clotting disorder to its fact sheets for patients and doctors.
Janet Woodcock, MD, acting commissioner of the FDA, said vaccination centers could resume giving the “one and done” shots as early as April 24.
This article was updated April 24, 2021, and first appeared on WebMD.com.
Use of the Johnson & Johnson COVID-19 vaccine should resume in the United States for all adults, the Food and Drug Administration and Centers for Disease Contol and Prevention said April 23, although health care providers should warn patients of the risk of developing the rare and serious blood clots that caused the agencies to pause the vaccine’s distribution earlier this month.
“What we are seeing is the overall rate of events was 1.9 cases per million people. In women 18 to 29 years there was an approximate 7 cases per million. The risk is even lower in women over the age of 50 at .9 cases per million,” CDC Director Rochelle Walensky, MD, said in a news briefing the same day.
In the end, the potential benefits of the vaccine far outweighed its risks.
“In terms of benefits, we found that for every 1 million doses of this vaccine, the J&J vaccine could prevent over 650 hospitalizations and 12 deaths among women ages 18-49,” Dr. Walensky said. The potential benefits to women over 50 were even greater: It could prevent 4,700 hospitalizations and 650 deaths.
“In the end, this vaccine was shown to be safe and effective for the vast majority of people,” Dr. Walensky said.
The recommendation to continue the vaccine’s rollout came barely 2 hours after a CDC Advisory Committee on Immunization Practices voted to recommend the pause be lifted. The vote was 10-4 with one abstention.
The decision also includes instructions for the warning directed at women under 50 who have an increased risk of a rare but serious blood clot disorder called thrombosis with thrombocytopenia syndrome (TTS).
As of April 21, 15 cases of TTS, all in women and 13 of them in women under 50, have been confirmed among 7.98 million doses of the J&J vaccine administered in the United States. Three women have died.
The FDA and CDC recommended the pause on April 13 after reports that 6 women developed a blood clotting disorder 6 to 13 days after they received the J&J vaccine.
William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville, and a non-voting ACIP member, said in an interview the panel made the right recommendation.
He applauded both the decision to restart the vaccine and the updated warning information that “will explain [TTS] more fully to people, particularly women, who are coming to be vaccinated.”
As to women in the risk group needing to have a choice of vaccines, Dr. Schaffner said that will be addressed differently across the country.
“Every provider will not have alternative vaccines in their location so there will be many different ways to do this. You may have to get this information and select which site you’re going to depending on which vaccine is available if this matter is important to you,” he noted.
ACIP made the decision after a 6-hour emergency meeting to hear evidence on the Johnson & Johnson vaccine's protective benefits against COVID-19 vs. risk of TTS.
In the CDC-FDA press briefing, Dr. Walensky pointed out that over the past few days, as regulators have reviewed the rare events, newly identified patients had been treated appropriately, without the use of heparin, which is not advised for treating TTS.
As a result, regulators felt as if their messages had gotten out to doctors who now knew how to take special precautions when treating patients with the disorder.
She said the Johnson & Johnson shot remained an important option because it was convenient to give and easier to store than the other vaccines currently authorized in the United States.
Peter Marks, MD, the director of FDA’s Center for Biologics Evaluation and Research, said the agency had already added information describing the risk of the rare clotting disorder to its fact sheets for patients and doctors.
Janet Woodcock, MD, acting commissioner of the FDA, said vaccination centers could resume giving the “one and done” shots as early as April 24.
This article was updated April 24, 2021, and first appeared on WebMD.com.
Use of the Johnson & Johnson COVID-19 vaccine should resume in the United States for all adults, the Food and Drug Administration and Centers for Disease Contol and Prevention said April 23, although health care providers should warn patients of the risk of developing the rare and serious blood clots that caused the agencies to pause the vaccine’s distribution earlier this month.
“What we are seeing is the overall rate of events was 1.9 cases per million people. In women 18 to 29 years there was an approximate 7 cases per million. The risk is even lower in women over the age of 50 at .9 cases per million,” CDC Director Rochelle Walensky, MD, said in a news briefing the same day.
In the end, the potential benefits of the vaccine far outweighed its risks.
“In terms of benefits, we found that for every 1 million doses of this vaccine, the J&J vaccine could prevent over 650 hospitalizations and 12 deaths among women ages 18-49,” Dr. Walensky said. The potential benefits to women over 50 were even greater: It could prevent 4,700 hospitalizations and 650 deaths.
“In the end, this vaccine was shown to be safe and effective for the vast majority of people,” Dr. Walensky said.
The recommendation to continue the vaccine’s rollout came barely 2 hours after a CDC Advisory Committee on Immunization Practices voted to recommend the pause be lifted. The vote was 10-4 with one abstention.
The decision also includes instructions for the warning directed at women under 50 who have an increased risk of a rare but serious blood clot disorder called thrombosis with thrombocytopenia syndrome (TTS).
As of April 21, 15 cases of TTS, all in women and 13 of them in women under 50, have been confirmed among 7.98 million doses of the J&J vaccine administered in the United States. Three women have died.
The FDA and CDC recommended the pause on April 13 after reports that 6 women developed a blood clotting disorder 6 to 13 days after they received the J&J vaccine.
William Schaffner, MD, an infectious disease expert at Vanderbilt University in Nashville, and a non-voting ACIP member, said in an interview the panel made the right recommendation.
He applauded both the decision to restart the vaccine and the updated warning information that “will explain [TTS] more fully to people, particularly women, who are coming to be vaccinated.”
As to women in the risk group needing to have a choice of vaccines, Dr. Schaffner said that will be addressed differently across the country.
“Every provider will not have alternative vaccines in their location so there will be many different ways to do this. You may have to get this information and select which site you’re going to depending on which vaccine is available if this matter is important to you,” he noted.
ACIP made the decision after a 6-hour emergency meeting to hear evidence on the Johnson & Johnson vaccine's protective benefits against COVID-19 vs. risk of TTS.
In the CDC-FDA press briefing, Dr. Walensky pointed out that over the past few days, as regulators have reviewed the rare events, newly identified patients had been treated appropriately, without the use of heparin, which is not advised for treating TTS.
As a result, regulators felt as if their messages had gotten out to doctors who now knew how to take special precautions when treating patients with the disorder.
She said the Johnson & Johnson shot remained an important option because it was convenient to give and easier to store than the other vaccines currently authorized in the United States.
Peter Marks, MD, the director of FDA’s Center for Biologics Evaluation and Research, said the agency had already added information describing the risk of the rare clotting disorder to its fact sheets for patients and doctors.
Janet Woodcock, MD, acting commissioner of the FDA, said vaccination centers could resume giving the “one and done” shots as early as April 24.
This article was updated April 24, 2021, and first appeared on WebMD.com.
Vaccinating homebound patients is an uphill battle

There are about 2 million to 4 million homebound patients in the United States, according to a webinar from The Trust for America’s Health, which was broadcast in March. But many of these individuals have not been vaccinated yet because of logistical challenges.
Some homebound COVID-19 immunization programs are administering Moderna and Pfizer vaccines to their patients, but many state, city, and local programs administered the Johnson & Johnson vaccine after it was cleared for use by the Food and Drug Administration in February 2021. The efficacy of the one-shot vaccine, as well as it being easier to store and ship than the Moderna and Pfizer vaccines, makes getting it to homebound patients less challenging.
“With Pfizer and Moderna, transportation is a challenge because the temperature demands and the fragility of [messenger] RNA–based vaccines,” Brent Feorene, executive director of the American Academy of Home Care Medicine, said in an interview. That’s why [the Johnson & Johnson] vaccine held such promise – it’s less fragile, [can be stored in] higher temperatures, and was a one shot.”
Other hurdles to getting homebound patients vaccinated had already been in place prior to the 10-day-pause on using the J&J vaccine that occurred for federal agencies to consider possible serious side effects linked to it.
Many roadblocks to vaccination
Although many homebound patients can’t readily go out into the community and be exposed to the COVID-19 virus themselves, they are dependent on caregivers and family members who do go out into the community.
“Their friends, family, neighbors, home health aides, and other kinds of health care workers come into the home,” said Shawn Amer, clinical program director at Central Ohio Primary Care in Columbus.
Nurses from Ms. Amer’s practice vaccinated approximately ten homebound patients with the J&J vaccine through a pilot program in March. Then on April 24, nurses from Central Ohio Primary Care vaccinated just under 40 homebound patients and about a handful of their caregivers who were not able to get their vaccines elsewhere, according to Ms. Amer. This time they used the Pfizer vaccine and will be returning to these patients’ homes on May 15 to administer the second dose.
“Any time you are getting in the car and adding miles, it adds complexity,” Ms. Amer said.
“We called patients 24 to 36 hours before coming to their homes to make sure they were ready, but we learned that just because the healthcare power of attorney agrees to a patient getting vaccinated does not mean that patient will be willing to get the vaccine when the nurse shows up," she noted.
Ms. Amer elaborated that three patients with dementia refused the vaccine when nurses arrived at their home on April 24.
“We had to pivot and find other people,” Ms. Amer. Her practice ended up having to waste one shot.
Expenses are greater
The higher costs of getting homebound patients vaccinated is an additional hurdle to getting these vulnerable individuals protected by COVID-19 shots.
Vaccinating patients in their homes “doesn’t require a lot of technology, but it does require a lot of time” and the staffing expense becomes part of the challenge, Ms. Amer noted.
For each of the two days that Central Ohio Primary Care provides the Pfizer vaccine to homebound patients, the practice needs to pay seven nurses to administer the vaccine, Ms. Amer explained.
There have also been reports of organizations that administer the vaccines – which are free for patients because the federal government is paying for them – not being paid enough by Medicare to cover staff time and efforts to vaccinate patients in their homes, Kaiser Health News reported. According to the Centers for Medicare & Medicaid Services, they pay $40 for the administration of a single-dose COVID-19 vaccine and, for COVID-19 vaccines requiring multiple doses, Medicare pays approximately $40 for each dose in the series. These rates were implemented after March 15. Before that date, the rates were even lower, with the Medicare reimbursement rates for initial doses of COVID-19 vaccines being $16.94 and final doses being $28.39.
William Dombi, president of the National Association for Home Care & Hospice, told Kaiser Health News that the actual cost of these homebound visits are closer to $150 or $160.
“The reimbursement for the injection is pretty minimal,” Mr. Feorene said. “So unless you’re a larger organization and able to have staff to deploy some of your smaller practices, just couldn’t afford to do it.”
Many homebound patients have also been unable to get the lifesaving shots because of logistical roadblocks and many practices not being able to do home visits.
“I think that initially when the [Centers for Disease Control and Prevention] came out with vaccine guidance for medical providers, they offered no guidance for in-home medical providers and we had to go back and ask for that, which they did produce,” Mr. Feorene said. “And we’re grateful for that. But I think just this general understanding that there is a population of folks that are [limited to their home], that they do receive medical care and other care in the home, and that we have to remember that the medical providers who provide care in the home are also primary care providers.”
Furthermore, trying to navigate or find programs delivering vaccines to the homebound can be difficult depending on where a patient lives.
While some programs have been launched on the country or city level – the New York Fire Department launched a pilot program to bring the Johnson & Johnson vaccine to homebound seniors – other programs have been spearheaded by hospital networks like Northwell and Mount Sinai. However, many of these hospital networks only reach out to people who already have a relationship with the hospital.
Ms Amer said identifying homebound patients and reaching out to them can be tough and can contribute to the logistics and time involved in setting patients up for the vaccine.
“Reaching some of these patients is difficult,” Ms. Amer noted. “Sometimes the best way to reach them or get a hold of them is through their caregiver. And so do you have the right phone number? Do you have the right name?”
Overcoming the challenges
With the absence of a national plan targeting homebound patients, many local initiatives were launched to help these individuals get vaccinated. Local fire department paramedics have gone door to door to administer the COVID-19 vaccine in cities like Chicago, New York, and Miami. The suspension of the Johnson & Johnson vaccine resulted in the suspension of in-home vaccinations for some people in New York City. However, the program resumed after the FDA and CDC lifted the pause on April 24. 
Health systems like Mount Sinai vaccinated approximately 530 people through the Mount Sinai Visiting Doctors Program, including patients and their caregivers, according to Peter Gliatto, MD, associate director of the Mount Sinai Visiting Doctors Program.
“In different cities, townships, and jurisdictions, different health departments and different provider groups are approaching [the distribution of the COVID-19 vaccine] slightly differently,” Ms. Amer said. So a lot of the decisions surrounding the distribution of shots are local or dependent on local resourcing.
People who live in rural areas present a unique challenge, but Mr. Feorene said reaching out to local emergency medical services or the local health departments can provide some insight on what their town is doing to vaccinate homebound patients.
“I think understanding what a [public health department] is doing would be the very first place to start,” Mr. Feorene said in an interview.
If a patient is bedridden and is mobile enough to sit in a car, Mr. Feorene also recommends finding out if there are vaccine fairs “within a reasonable driving distance.”
Ms. Amer said continuing this mission of getting homebound patients vaccinated is necessary for public health.
“Even if it’s going to take longer to vaccinate these homebound patients, we still have to make an effort. So much of the country’s vaccine efforts have been focused on getting as many shots in as many arms as quickly as possible. And that is definitely super important,” she said.
Ms. Amer is working with her practice’s primary care physicians to try to identify all of those patients who are functionally debilitated or unable to leave their home to get vaccinated and that Central Ohio Primary Care will vaccinate more homebound patients, she added.
The experts interviewed in this article have no conflicts.
Katie Lennon contributed to this report.
This article was updated 4/29/21.

There are about 2 million to 4 million homebound patients in the United States, according to a webinar from The Trust for America’s Health, which was broadcast in March. But many of these individuals have not been vaccinated yet because of logistical challenges.
Some homebound COVID-19 immunization programs are administering Moderna and Pfizer vaccines to their patients, but many state, city, and local programs administered the Johnson & Johnson vaccine after it was cleared for use by the Food and Drug Administration in February 2021. The efficacy of the one-shot vaccine, as well as it being easier to store and ship than the Moderna and Pfizer vaccines, makes getting it to homebound patients less challenging.
“With Pfizer and Moderna, transportation is a challenge because the temperature demands and the fragility of [messenger] RNA–based vaccines,” Brent Feorene, executive director of the American Academy of Home Care Medicine, said in an interview. That’s why [the Johnson & Johnson] vaccine held such promise – it’s less fragile, [can be stored in] higher temperatures, and was a one shot.”
Other hurdles to getting homebound patients vaccinated had already been in place prior to the 10-day-pause on using the J&J vaccine that occurred for federal agencies to consider possible serious side effects linked to it.
Many roadblocks to vaccination
Although many homebound patients can’t readily go out into the community and be exposed to the COVID-19 virus themselves, they are dependent on caregivers and family members who do go out into the community.
“Their friends, family, neighbors, home health aides, and other kinds of health care workers come into the home,” said Shawn Amer, clinical program director at Central Ohio Primary Care in Columbus.
Nurses from Ms. Amer’s practice vaccinated approximately ten homebound patients with the J&J vaccine through a pilot program in March. Then on April 24, nurses from Central Ohio Primary Care vaccinated just under 40 homebound patients and about a handful of their caregivers who were not able to get their vaccines elsewhere, according to Ms. Amer. This time they used the Pfizer vaccine and will be returning to these patients’ homes on May 15 to administer the second dose.
“Any time you are getting in the car and adding miles, it adds complexity,” Ms. Amer said.
“We called patients 24 to 36 hours before coming to their homes to make sure they were ready, but we learned that just because the healthcare power of attorney agrees to a patient getting vaccinated does not mean that patient will be willing to get the vaccine when the nurse shows up," she noted.
Ms. Amer elaborated that three patients with dementia refused the vaccine when nurses arrived at their home on April 24.
“We had to pivot and find other people,” Ms. Amer. Her practice ended up having to waste one shot.
Expenses are greater
The higher costs of getting homebound patients vaccinated is an additional hurdle to getting these vulnerable individuals protected by COVID-19 shots.
Vaccinating patients in their homes “doesn’t require a lot of technology, but it does require a lot of time” and the staffing expense becomes part of the challenge, Ms. Amer noted.
For each of the two days that Central Ohio Primary Care provides the Pfizer vaccine to homebound patients, the practice needs to pay seven nurses to administer the vaccine, Ms. Amer explained.
There have also been reports of organizations that administer the vaccines – which are free for patients because the federal government is paying for them – not being paid enough by Medicare to cover staff time and efforts to vaccinate patients in their homes, Kaiser Health News reported. According to the Centers for Medicare & Medicaid Services, they pay $40 for the administration of a single-dose COVID-19 vaccine and, for COVID-19 vaccines requiring multiple doses, Medicare pays approximately $40 for each dose in the series. These rates were implemented after March 15. Before that date, the rates were even lower, with the Medicare reimbursement rates for initial doses of COVID-19 vaccines being $16.94 and final doses being $28.39.
William Dombi, president of the National Association for Home Care & Hospice, told Kaiser Health News that the actual cost of these homebound visits are closer to $150 or $160.
“The reimbursement for the injection is pretty minimal,” Mr. Feorene said. “So unless you’re a larger organization and able to have staff to deploy some of your smaller practices, just couldn’t afford to do it.”
Many homebound patients have also been unable to get the lifesaving shots because of logistical roadblocks and many practices not being able to do home visits.
“I think that initially when the [Centers for Disease Control and Prevention] came out with vaccine guidance for medical providers, they offered no guidance for in-home medical providers and we had to go back and ask for that, which they did produce,” Mr. Feorene said. “And we’re grateful for that. But I think just this general understanding that there is a population of folks that are [limited to their home], that they do receive medical care and other care in the home, and that we have to remember that the medical providers who provide care in the home are also primary care providers.”
Furthermore, trying to navigate or find programs delivering vaccines to the homebound can be difficult depending on where a patient lives.
While some programs have been launched on the country or city level – the New York Fire Department launched a pilot program to bring the Johnson & Johnson vaccine to homebound seniors – other programs have been spearheaded by hospital networks like Northwell and Mount Sinai. However, many of these hospital networks only reach out to people who already have a relationship with the hospital.
Ms Amer said identifying homebound patients and reaching out to them can be tough and can contribute to the logistics and time involved in setting patients up for the vaccine.
“Reaching some of these patients is difficult,” Ms. Amer noted. “Sometimes the best way to reach them or get a hold of them is through their caregiver. And so do you have the right phone number? Do you have the right name?”
Overcoming the challenges
With the absence of a national plan targeting homebound patients, many local initiatives were launched to help these individuals get vaccinated. Local fire department paramedics have gone door to door to administer the COVID-19 vaccine in cities like Chicago, New York, and Miami. The suspension of the Johnson & Johnson vaccine resulted in the suspension of in-home vaccinations for some people in New York City. However, the program resumed after the FDA and CDC lifted the pause on April 24. 
Health systems like Mount Sinai vaccinated approximately 530 people through the Mount Sinai Visiting Doctors Program, including patients and their caregivers, according to Peter Gliatto, MD, associate director of the Mount Sinai Visiting Doctors Program.
“In different cities, townships, and jurisdictions, different health departments and different provider groups are approaching [the distribution of the COVID-19 vaccine] slightly differently,” Ms. Amer said. So a lot of the decisions surrounding the distribution of shots are local or dependent on local resourcing.
People who live in rural areas present a unique challenge, but Mr. Feorene said reaching out to local emergency medical services or the local health departments can provide some insight on what their town is doing to vaccinate homebound patients.
“I think understanding what a [public health department] is doing would be the very first place to start,” Mr. Feorene said in an interview.
If a patient is bedridden and is mobile enough to sit in a car, Mr. Feorene also recommends finding out if there are vaccine fairs “within a reasonable driving distance.”
Ms. Amer said continuing this mission of getting homebound patients vaccinated is necessary for public health.
“Even if it’s going to take longer to vaccinate these homebound patients, we still have to make an effort. So much of the country’s vaccine efforts have been focused on getting as many shots in as many arms as quickly as possible. And that is definitely super important,” she said.
Ms. Amer is working with her practice’s primary care physicians to try to identify all of those patients who are functionally debilitated or unable to leave their home to get vaccinated and that Central Ohio Primary Care will vaccinate more homebound patients, she added.
The experts interviewed in this article have no conflicts.
Katie Lennon contributed to this report.
This article was updated 4/29/21.

There are about 2 million to 4 million homebound patients in the United States, according to a webinar from The Trust for America’s Health, which was broadcast in March. But many of these individuals have not been vaccinated yet because of logistical challenges.
Some homebound COVID-19 immunization programs are administering Moderna and Pfizer vaccines to their patients, but many state, city, and local programs administered the Johnson & Johnson vaccine after it was cleared for use by the Food and Drug Administration in February 2021. The efficacy of the one-shot vaccine, as well as it being easier to store and ship than the Moderna and Pfizer vaccines, makes getting it to homebound patients less challenging.
“With Pfizer and Moderna, transportation is a challenge because the temperature demands and the fragility of [messenger] RNA–based vaccines,” Brent Feorene, executive director of the American Academy of Home Care Medicine, said in an interview. That’s why [the Johnson & Johnson] vaccine held such promise – it’s less fragile, [can be stored in] higher temperatures, and was a one shot.”
Other hurdles to getting homebound patients vaccinated had already been in place prior to the 10-day-pause on using the J&J vaccine that occurred for federal agencies to consider possible serious side effects linked to it.
Many roadblocks to vaccination
Although many homebound patients can’t readily go out into the community and be exposed to the COVID-19 virus themselves, they are dependent on caregivers and family members who do go out into the community.
“Their friends, family, neighbors, home health aides, and other kinds of health care workers come into the home,” said Shawn Amer, clinical program director at Central Ohio Primary Care in Columbus.
Nurses from Ms. Amer’s practice vaccinated approximately ten homebound patients with the J&J vaccine through a pilot program in March. Then on April 24, nurses from Central Ohio Primary Care vaccinated just under 40 homebound patients and about a handful of their caregivers who were not able to get their vaccines elsewhere, according to Ms. Amer. This time they used the Pfizer vaccine and will be returning to these patients’ homes on May 15 to administer the second dose.
“Any time you are getting in the car and adding miles, it adds complexity,” Ms. Amer said.
“We called patients 24 to 36 hours before coming to their homes to make sure they were ready, but we learned that just because the healthcare power of attorney agrees to a patient getting vaccinated does not mean that patient will be willing to get the vaccine when the nurse shows up," she noted.
Ms. Amer elaborated that three patients with dementia refused the vaccine when nurses arrived at their home on April 24.
“We had to pivot and find other people,” Ms. Amer. Her practice ended up having to waste one shot.
Expenses are greater
The higher costs of getting homebound patients vaccinated is an additional hurdle to getting these vulnerable individuals protected by COVID-19 shots.
Vaccinating patients in their homes “doesn’t require a lot of technology, but it does require a lot of time” and the staffing expense becomes part of the challenge, Ms. Amer noted.
For each of the two days that Central Ohio Primary Care provides the Pfizer vaccine to homebound patients, the practice needs to pay seven nurses to administer the vaccine, Ms. Amer explained.
There have also been reports of organizations that administer the vaccines – which are free for patients because the federal government is paying for them – not being paid enough by Medicare to cover staff time and efforts to vaccinate patients in their homes, Kaiser Health News reported. According to the Centers for Medicare & Medicaid Services, they pay $40 for the administration of a single-dose COVID-19 vaccine and, for COVID-19 vaccines requiring multiple doses, Medicare pays approximately $40 for each dose in the series. These rates were implemented after March 15. Before that date, the rates were even lower, with the Medicare reimbursement rates for initial doses of COVID-19 vaccines being $16.94 and final doses being $28.39.
William Dombi, president of the National Association for Home Care & Hospice, told Kaiser Health News that the actual cost of these homebound visits are closer to $150 or $160.
“The reimbursement for the injection is pretty minimal,” Mr. Feorene said. “So unless you’re a larger organization and able to have staff to deploy some of your smaller practices, just couldn’t afford to do it.”
Many homebound patients have also been unable to get the lifesaving shots because of logistical roadblocks and many practices not being able to do home visits.
“I think that initially when the [Centers for Disease Control and Prevention] came out with vaccine guidance for medical providers, they offered no guidance for in-home medical providers and we had to go back and ask for that, which they did produce,” Mr. Feorene said. “And we’re grateful for that. But I think just this general understanding that there is a population of folks that are [limited to their home], that they do receive medical care and other care in the home, and that we have to remember that the medical providers who provide care in the home are also primary care providers.”
Furthermore, trying to navigate or find programs delivering vaccines to the homebound can be difficult depending on where a patient lives.
While some programs have been launched on the country or city level – the New York Fire Department launched a pilot program to bring the Johnson & Johnson vaccine to homebound seniors – other programs have been spearheaded by hospital networks like Northwell and Mount Sinai. However, many of these hospital networks only reach out to people who already have a relationship with the hospital.
Ms Amer said identifying homebound patients and reaching out to them can be tough and can contribute to the logistics and time involved in setting patients up for the vaccine.
“Reaching some of these patients is difficult,” Ms. Amer noted. “Sometimes the best way to reach them or get a hold of them is through their caregiver. And so do you have the right phone number? Do you have the right name?”
Overcoming the challenges
With the absence of a national plan targeting homebound patients, many local initiatives were launched to help these individuals get vaccinated. Local fire department paramedics have gone door to door to administer the COVID-19 vaccine in cities like Chicago, New York, and Miami. The suspension of the Johnson & Johnson vaccine resulted in the suspension of in-home vaccinations for some people in New York City. However, the program resumed after the FDA and CDC lifted the pause on April 24. 
Health systems like Mount Sinai vaccinated approximately 530 people through the Mount Sinai Visiting Doctors Program, including patients and their caregivers, according to Peter Gliatto, MD, associate director of the Mount Sinai Visiting Doctors Program.
“In different cities, townships, and jurisdictions, different health departments and different provider groups are approaching [the distribution of the COVID-19 vaccine] slightly differently,” Ms. Amer said. So a lot of the decisions surrounding the distribution of shots are local or dependent on local resourcing.
People who live in rural areas present a unique challenge, but Mr. Feorene said reaching out to local emergency medical services or the local health departments can provide some insight on what their town is doing to vaccinate homebound patients.
“I think understanding what a [public health department] is doing would be the very first place to start,” Mr. Feorene said in an interview.
If a patient is bedridden and is mobile enough to sit in a car, Mr. Feorene also recommends finding out if there are vaccine fairs “within a reasonable driving distance.”
Ms. Amer said continuing this mission of getting homebound patients vaccinated is necessary for public health.
“Even if it’s going to take longer to vaccinate these homebound patients, we still have to make an effort. So much of the country’s vaccine efforts have been focused on getting as many shots in as many arms as quickly as possible. And that is definitely super important,” she said.
Ms. Amer is working with her practice’s primary care physicians to try to identify all of those patients who are functionally debilitated or unable to leave their home to get vaccinated and that Central Ohio Primary Care will vaccinate more homebound patients, she added.
The experts interviewed in this article have no conflicts.
Katie Lennon contributed to this report.
This article was updated 4/29/21.
More signs COVID shots are safe for pregnant women
As the U.S. races to vaccinate millions of people against the coronavirus, pregnant women face the extra challenge of not knowing whether the vaccines are safe for them or their unborn babies.
None of the recent COVID-19 vaccine trials, including those for Pfizer, Moderna, and Johnson & Johnson, enrolled pregnant or breastfeeding women because they consider them a high-risk group.
That was despite the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists asking that pregnant and breastfeeding women be included in trials. The Food and Drug Administration even included pregnant women in the COVID-19 vaccine emergency use authorization (EUA) because of their higher risk of having a more severe disease.
Despite that lack of clinical trial data, more and more smaller studies are suggesting that the vaccines are safe for both mother and child.
Pfizer is now studying its two-dose vaccine in 4,000 pregnant and breastfeeding women to see how safe, tolerated, and robust their immune response is. Researchers will also look at how safe the vaccine is for infants and whether mothers pass along antibodies to children. But the preliminary results won’t be available until the end of the year, a Pfizer spokesperson says.
Without that information, pregnant women are less likely to get vaccinated, according to a large international survey. Less than 45% of pregnant women in the United States said they intended to get vaccinated even when they were told the vaccine was safe and 90% effective. That figure rises to 52% of pregnant women in 16 countries, including the United States, compared with 74% of nonpregnant women willing to be vaccinated. The findings were published online March 1, 2021, in the European Journal of Epidemiology.
The vaccine-hesitant pregnant women in the international study were most concerned that the COVID-19 vaccine could harm their developing fetuses, a worry related to the lack of clinical evidence in pregnant women, said lead researcher Julia Wu, ScD, an epidemiologist at the Harvard School of Public Health’s Human Immunomics Initiative in Boston.
The information vacuum also increases the chances that “people will fall victim to misinformation campaigns like the one on social media that claims that the COVID-19 vaccine causes infertility,” Dr. Wu said. This unfounded claim has deterred some women of childbearing age from getting the vaccine.
Deciding to get vaccinated
Frontline health care professionals were in the first group eligible to receive the vaccine in December 2020. “All of us who were pregnant ... had to decide whether to wait for the data, because we don’t know what the risks are, or go ahead and get it [the vaccine]. We had been dealing with the pandemic for months and were afraid of being exposed to the virus and infecting family members,” said Jacqueline Parchem, MD, a maternal-fetal medicine specialist at the University of Texas Health Science Center, Houston.
Given the lack of safety data, the CDC guidance to pregnant women has been to consult with their doctors and that it’s a personal choice. The Center for Disease Control and Prevention’s latest vaccine guidance said that “there is no evidence that antibodies formed from COVID-19 vaccination cause any problem with pregnancy, including the development of the placenta.”
The CDC is monitoring vaccinated people through its v-safe program and reported on April 12 that more than 86,000 v-safe participants said they were pregnant when they were vaccinated.
Health care workers who were nursing their infants when they were eligible for the vaccine faced a similar dilemma as pregnant women – they lacked the data on them to make a truly informed decision.
“I was nervous about the vaccine side effects for myself and whether my son Bennett, who was about a year old, would experience any of these himself,” said Christa Carrig, a labor and delivery nurse at Massachusetts General Hospital in Boston, who was breastfeeding at the time.
She and Dr. Parchem know that pregnant women with COVID-19 are more likely to have severe illness and complications such as high blood pressure and preterm delivery. “Pregnancy takes a toll on the body. When a woman gets COVID-19 and that insult is added, women who were otherwise young and healthy get much sicker than you would expect,” said Ms. Carrig.
“As a high-risk pregnancy specialist, I know that, with COVID, that babies don’t do well when moms are sick,” said Dr. Parchem.
Pregnant women accounted for more than 84,629 cases of COVID-19 and 95 deaths in the United States between Jan. 22 last year and April 12 this year, according to the CDC COVID data tracker.
Dr. Parchem and Ms. Carrig decided to get vaccinated because of their high risk of exposure to COVID-19 at work. After the second dose, Ms. Carrig reported chills but Bennett had no side effects from breastfeeding. Dr. Parchem, who delivered a healthy baby boy in February, reported no side effects other than a sore arm.
“There’s also a psychological benefit to returning to some sense of normalcy,” said Dr. Parchem. “My mother was finally able to visit us to see the new baby after we were all vaccinated. This was the first visit in more than a year.”
New study results
Ms. Carrig was one of 131 vaccinated hospital workers in the Boston area who took part in the first study to profile the immune response in pregnant and breastfeeding women and compare it with both nonpregnant and pregnant women who had COVID-19.
The study was not designed to evaluate the safety of the vaccines or whether they prevent COVID-19 illness and hospitalizations. That is the role of the large vaccine trials, the authors said.
The participants were aged 18-45 years and received both doses of either Pfizer or Moderna vaccines during one of their trimesters. They provided blood and/or breast milk samples after each vaccine dose, 2-6 weeks after the last dose, and at delivery for the 10 who gave birth during the study.
The vaccines produced a similar strong antibody response among the pregnant/breastfeeding women and nonpregnant women. Their antibody levels were much higher than those found in the pregnant women who had COVID-19, the researchers reported on March 25, 2021, in the American Journal of Obstetrics and Gynecology.
“This is important because a lot of people tend to think once they’ve had COVID-19, they are protected from the virus. This finding suggests that the vaccines produce a stronger antibody response than the infection itself, and this might be important for long-lasting protection against COVID-19,” said Dr. Parchem.
The study also addressed whether newborns benefit from the antibodies produced by their mothers. “In the 10 women who delivered, we detected antibodies in their umbilical cords and breast milk,” says Andrea Edlow, MD, lead researcher and a maternal-fetal medicine specialist at Massachusetts General Hospital.
Newborns are particularly vulnerable to respiratory infections because they have small airways and their immune systems are underdeveloped. These infections can be lethal early in life.
“The public health strategy is to vaccinate mothers against respiratory viruses, bacteria, and parasites that neonates up to 6 months are exposed to. Influenza and pertussis (whooping cough) are two examples of vaccines that we give mothers that we know transfer [antibodies] across the umbilical cord,” said Dr. Edlow.
But this “passive transfer immunity” is different from active immunity, when the body produces its own antibody immune response, she explains.
A different study, also published in March, confirmed that antibodies were transferred from 27 vaccinated pregnant mothers to their infants when they delivered. A new finding was that the women who were vaccinated with both doses and earlier in their third semester passed on more antibodies than the women who were vaccinated later or with only one dose.
Impact of the studies
The Society for Maternal-Fetal Medicine updated its guidance on counseling pregnant and lactating patients about the COVID-19 vaccines to include Dr. Edlow’s study.
“We were struck by how much pregnant and breastfeeding women want to participate in research and to help others in the same situation make decisions. I hope this will be an example to drug companies doing research on new vaccines in the future – that they should not be left behind and can make decisions themselves whether to participate after weighing the risks and benefits,” said Dr. Edlow.
She continues to enroll more vaccinated women in her study in the Boston area, including non–health care workers who have asked to take part.
“It was worth getting vaccinated and participating in the study. I know that I have antibodies and it worked and that I passed them on to Bennett. Also, I know that all the information is available for other women who are questioning whether to get vaccinated or not,” said Ms. Carrig.
Dr. Parchem is also taking part in the CDC’s v-safe pregnancy registry, which is collecting health and safety data on vaccinated pregnant women.
Before she was vaccinated, Dr. Parchem said, “my advice was very measured because we lacked data either saying that it definitely works or showing that it was unsafe. Now that we have this data supporting the benefits, I feel more confident in recommending the vaccines.”
A version of this article first appeared on Medscape.com.
As the U.S. races to vaccinate millions of people against the coronavirus, pregnant women face the extra challenge of not knowing whether the vaccines are safe for them or their unborn babies.
None of the recent COVID-19 vaccine trials, including those for Pfizer, Moderna, and Johnson & Johnson, enrolled pregnant or breastfeeding women because they consider them a high-risk group.
That was despite the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists asking that pregnant and breastfeeding women be included in trials. The Food and Drug Administration even included pregnant women in the COVID-19 vaccine emergency use authorization (EUA) because of their higher risk of having a more severe disease.
Despite that lack of clinical trial data, more and more smaller studies are suggesting that the vaccines are safe for both mother and child.
Pfizer is now studying its two-dose vaccine in 4,000 pregnant and breastfeeding women to see how safe, tolerated, and robust their immune response is. Researchers will also look at how safe the vaccine is for infants and whether mothers pass along antibodies to children. But the preliminary results won’t be available until the end of the year, a Pfizer spokesperson says.
Without that information, pregnant women are less likely to get vaccinated, according to a large international survey. Less than 45% of pregnant women in the United States said they intended to get vaccinated even when they were told the vaccine was safe and 90% effective. That figure rises to 52% of pregnant women in 16 countries, including the United States, compared with 74% of nonpregnant women willing to be vaccinated. The findings were published online March 1, 2021, in the European Journal of Epidemiology.
The vaccine-hesitant pregnant women in the international study were most concerned that the COVID-19 vaccine could harm their developing fetuses, a worry related to the lack of clinical evidence in pregnant women, said lead researcher Julia Wu, ScD, an epidemiologist at the Harvard School of Public Health’s Human Immunomics Initiative in Boston.
The information vacuum also increases the chances that “people will fall victim to misinformation campaigns like the one on social media that claims that the COVID-19 vaccine causes infertility,” Dr. Wu said. This unfounded claim has deterred some women of childbearing age from getting the vaccine.
Deciding to get vaccinated
Frontline health care professionals were in the first group eligible to receive the vaccine in December 2020. “All of us who were pregnant ... had to decide whether to wait for the data, because we don’t know what the risks are, or go ahead and get it [the vaccine]. We had been dealing with the pandemic for months and were afraid of being exposed to the virus and infecting family members,” said Jacqueline Parchem, MD, a maternal-fetal medicine specialist at the University of Texas Health Science Center, Houston.
Given the lack of safety data, the CDC guidance to pregnant women has been to consult with their doctors and that it’s a personal choice. The Center for Disease Control and Prevention’s latest vaccine guidance said that “there is no evidence that antibodies formed from COVID-19 vaccination cause any problem with pregnancy, including the development of the placenta.”
The CDC is monitoring vaccinated people through its v-safe program and reported on April 12 that more than 86,000 v-safe participants said they were pregnant when they were vaccinated.
Health care workers who were nursing their infants when they were eligible for the vaccine faced a similar dilemma as pregnant women – they lacked the data on them to make a truly informed decision.
“I was nervous about the vaccine side effects for myself and whether my son Bennett, who was about a year old, would experience any of these himself,” said Christa Carrig, a labor and delivery nurse at Massachusetts General Hospital in Boston, who was breastfeeding at the time.
She and Dr. Parchem know that pregnant women with COVID-19 are more likely to have severe illness and complications such as high blood pressure and preterm delivery. “Pregnancy takes a toll on the body. When a woman gets COVID-19 and that insult is added, women who were otherwise young and healthy get much sicker than you would expect,” said Ms. Carrig.
“As a high-risk pregnancy specialist, I know that, with COVID, that babies don’t do well when moms are sick,” said Dr. Parchem.
Pregnant women accounted for more than 84,629 cases of COVID-19 and 95 deaths in the United States between Jan. 22 last year and April 12 this year, according to the CDC COVID data tracker.
Dr. Parchem and Ms. Carrig decided to get vaccinated because of their high risk of exposure to COVID-19 at work. After the second dose, Ms. Carrig reported chills but Bennett had no side effects from breastfeeding. Dr. Parchem, who delivered a healthy baby boy in February, reported no side effects other than a sore arm.
“There’s also a psychological benefit to returning to some sense of normalcy,” said Dr. Parchem. “My mother was finally able to visit us to see the new baby after we were all vaccinated. This was the first visit in more than a year.”
New study results
Ms. Carrig was one of 131 vaccinated hospital workers in the Boston area who took part in the first study to profile the immune response in pregnant and breastfeeding women and compare it with both nonpregnant and pregnant women who had COVID-19.
The study was not designed to evaluate the safety of the vaccines or whether they prevent COVID-19 illness and hospitalizations. That is the role of the large vaccine trials, the authors said.
The participants were aged 18-45 years and received both doses of either Pfizer or Moderna vaccines during one of their trimesters. They provided blood and/or breast milk samples after each vaccine dose, 2-6 weeks after the last dose, and at delivery for the 10 who gave birth during the study.
The vaccines produced a similar strong antibody response among the pregnant/breastfeeding women and nonpregnant women. Their antibody levels were much higher than those found in the pregnant women who had COVID-19, the researchers reported on March 25, 2021, in the American Journal of Obstetrics and Gynecology.
“This is important because a lot of people tend to think once they’ve had COVID-19, they are protected from the virus. This finding suggests that the vaccines produce a stronger antibody response than the infection itself, and this might be important for long-lasting protection against COVID-19,” said Dr. Parchem.
The study also addressed whether newborns benefit from the antibodies produced by their mothers. “In the 10 women who delivered, we detected antibodies in their umbilical cords and breast milk,” says Andrea Edlow, MD, lead researcher and a maternal-fetal medicine specialist at Massachusetts General Hospital.
Newborns are particularly vulnerable to respiratory infections because they have small airways and their immune systems are underdeveloped. These infections can be lethal early in life.
“The public health strategy is to vaccinate mothers against respiratory viruses, bacteria, and parasites that neonates up to 6 months are exposed to. Influenza and pertussis (whooping cough) are two examples of vaccines that we give mothers that we know transfer [antibodies] across the umbilical cord,” said Dr. Edlow.
But this “passive transfer immunity” is different from active immunity, when the body produces its own antibody immune response, she explains.
A different study, also published in March, confirmed that antibodies were transferred from 27 vaccinated pregnant mothers to their infants when they delivered. A new finding was that the women who were vaccinated with both doses and earlier in their third semester passed on more antibodies than the women who were vaccinated later or with only one dose.
Impact of the studies
The Society for Maternal-Fetal Medicine updated its guidance on counseling pregnant and lactating patients about the COVID-19 vaccines to include Dr. Edlow’s study.
“We were struck by how much pregnant and breastfeeding women want to participate in research and to help others in the same situation make decisions. I hope this will be an example to drug companies doing research on new vaccines in the future – that they should not be left behind and can make decisions themselves whether to participate after weighing the risks and benefits,” said Dr. Edlow.
She continues to enroll more vaccinated women in her study in the Boston area, including non–health care workers who have asked to take part.
“It was worth getting vaccinated and participating in the study. I know that I have antibodies and it worked and that I passed them on to Bennett. Also, I know that all the information is available for other women who are questioning whether to get vaccinated or not,” said Ms. Carrig.
Dr. Parchem is also taking part in the CDC’s v-safe pregnancy registry, which is collecting health and safety data on vaccinated pregnant women.
Before she was vaccinated, Dr. Parchem said, “my advice was very measured because we lacked data either saying that it definitely works or showing that it was unsafe. Now that we have this data supporting the benefits, I feel more confident in recommending the vaccines.”
A version of this article first appeared on Medscape.com.
As the U.S. races to vaccinate millions of people against the coronavirus, pregnant women face the extra challenge of not knowing whether the vaccines are safe for them or their unborn babies.
None of the recent COVID-19 vaccine trials, including those for Pfizer, Moderna, and Johnson & Johnson, enrolled pregnant or breastfeeding women because they consider them a high-risk group.
That was despite the Society for Maternal-Fetal Medicine and the American College of Obstetricians and Gynecologists asking that pregnant and breastfeeding women be included in trials. The Food and Drug Administration even included pregnant women in the COVID-19 vaccine emergency use authorization (EUA) because of their higher risk of having a more severe disease.
Despite that lack of clinical trial data, more and more smaller studies are suggesting that the vaccines are safe for both mother and child.
Pfizer is now studying its two-dose vaccine in 4,000 pregnant and breastfeeding women to see how safe, tolerated, and robust their immune response is. Researchers will also look at how safe the vaccine is for infants and whether mothers pass along antibodies to children. But the preliminary results won’t be available until the end of the year, a Pfizer spokesperson says.
Without that information, pregnant women are less likely to get vaccinated, according to a large international survey. Less than 45% of pregnant women in the United States said they intended to get vaccinated even when they were told the vaccine was safe and 90% effective. That figure rises to 52% of pregnant women in 16 countries, including the United States, compared with 74% of nonpregnant women willing to be vaccinated. The findings were published online March 1, 2021, in the European Journal of Epidemiology.
The vaccine-hesitant pregnant women in the international study were most concerned that the COVID-19 vaccine could harm their developing fetuses, a worry related to the lack of clinical evidence in pregnant women, said lead researcher Julia Wu, ScD, an epidemiologist at the Harvard School of Public Health’s Human Immunomics Initiative in Boston.
The information vacuum also increases the chances that “people will fall victim to misinformation campaigns like the one on social media that claims that the COVID-19 vaccine causes infertility,” Dr. Wu said. This unfounded claim has deterred some women of childbearing age from getting the vaccine.
Deciding to get vaccinated
Frontline health care professionals were in the first group eligible to receive the vaccine in December 2020. “All of us who were pregnant ... had to decide whether to wait for the data, because we don’t know what the risks are, or go ahead and get it [the vaccine]. We had been dealing with the pandemic for months and were afraid of being exposed to the virus and infecting family members,” said Jacqueline Parchem, MD, a maternal-fetal medicine specialist at the University of Texas Health Science Center, Houston.
Given the lack of safety data, the CDC guidance to pregnant women has been to consult with their doctors and that it’s a personal choice. The Center for Disease Control and Prevention’s latest vaccine guidance said that “there is no evidence that antibodies formed from COVID-19 vaccination cause any problem with pregnancy, including the development of the placenta.”
The CDC is monitoring vaccinated people through its v-safe program and reported on April 12 that more than 86,000 v-safe participants said they were pregnant when they were vaccinated.
Health care workers who were nursing their infants when they were eligible for the vaccine faced a similar dilemma as pregnant women – they lacked the data on them to make a truly informed decision.
“I was nervous about the vaccine side effects for myself and whether my son Bennett, who was about a year old, would experience any of these himself,” said Christa Carrig, a labor and delivery nurse at Massachusetts General Hospital in Boston, who was breastfeeding at the time.
She and Dr. Parchem know that pregnant women with COVID-19 are more likely to have severe illness and complications such as high blood pressure and preterm delivery. “Pregnancy takes a toll on the body. When a woman gets COVID-19 and that insult is added, women who were otherwise young and healthy get much sicker than you would expect,” said Ms. Carrig.
“As a high-risk pregnancy specialist, I know that, with COVID, that babies don’t do well when moms are sick,” said Dr. Parchem.
Pregnant women accounted for more than 84,629 cases of COVID-19 and 95 deaths in the United States between Jan. 22 last year and April 12 this year, according to the CDC COVID data tracker.
Dr. Parchem and Ms. Carrig decided to get vaccinated because of their high risk of exposure to COVID-19 at work. After the second dose, Ms. Carrig reported chills but Bennett had no side effects from breastfeeding. Dr. Parchem, who delivered a healthy baby boy in February, reported no side effects other than a sore arm.
“There’s also a psychological benefit to returning to some sense of normalcy,” said Dr. Parchem. “My mother was finally able to visit us to see the new baby after we were all vaccinated. This was the first visit in more than a year.”
New study results
Ms. Carrig was one of 131 vaccinated hospital workers in the Boston area who took part in the first study to profile the immune response in pregnant and breastfeeding women and compare it with both nonpregnant and pregnant women who had COVID-19.
The study was not designed to evaluate the safety of the vaccines or whether they prevent COVID-19 illness and hospitalizations. That is the role of the large vaccine trials, the authors said.
The participants were aged 18-45 years and received both doses of either Pfizer or Moderna vaccines during one of their trimesters. They provided blood and/or breast milk samples after each vaccine dose, 2-6 weeks after the last dose, and at delivery for the 10 who gave birth during the study.
The vaccines produced a similar strong antibody response among the pregnant/breastfeeding women and nonpregnant women. Their antibody levels were much higher than those found in the pregnant women who had COVID-19, the researchers reported on March 25, 2021, in the American Journal of Obstetrics and Gynecology.
“This is important because a lot of people tend to think once they’ve had COVID-19, they are protected from the virus. This finding suggests that the vaccines produce a stronger antibody response than the infection itself, and this might be important for long-lasting protection against COVID-19,” said Dr. Parchem.
The study also addressed whether newborns benefit from the antibodies produced by their mothers. “In the 10 women who delivered, we detected antibodies in their umbilical cords and breast milk,” says Andrea Edlow, MD, lead researcher and a maternal-fetal medicine specialist at Massachusetts General Hospital.
Newborns are particularly vulnerable to respiratory infections because they have small airways and their immune systems are underdeveloped. These infections can be lethal early in life.
“The public health strategy is to vaccinate mothers against respiratory viruses, bacteria, and parasites that neonates up to 6 months are exposed to. Influenza and pertussis (whooping cough) are two examples of vaccines that we give mothers that we know transfer [antibodies] across the umbilical cord,” said Dr. Edlow.
But this “passive transfer immunity” is different from active immunity, when the body produces its own antibody immune response, she explains.
A different study, also published in March, confirmed that antibodies were transferred from 27 vaccinated pregnant mothers to their infants when they delivered. A new finding was that the women who were vaccinated with both doses and earlier in their third semester passed on more antibodies than the women who were vaccinated later or with only one dose.
Impact of the studies
The Society for Maternal-Fetal Medicine updated its guidance on counseling pregnant and lactating patients about the COVID-19 vaccines to include Dr. Edlow’s study.
“We were struck by how much pregnant and breastfeeding women want to participate in research and to help others in the same situation make decisions. I hope this will be an example to drug companies doing research on new vaccines in the future – that they should not be left behind and can make decisions themselves whether to participate after weighing the risks and benefits,” said Dr. Edlow.
She continues to enroll more vaccinated women in her study in the Boston area, including non–health care workers who have asked to take part.
“It was worth getting vaccinated and participating in the study. I know that I have antibodies and it worked and that I passed them on to Bennett. Also, I know that all the information is available for other women who are questioning whether to get vaccinated or not,” said Ms. Carrig.
Dr. Parchem is also taking part in the CDC’s v-safe pregnancy registry, which is collecting health and safety data on vaccinated pregnant women.
Before she was vaccinated, Dr. Parchem said, “my advice was very measured because we lacked data either saying that it definitely works or showing that it was unsafe. Now that we have this data supporting the benefits, I feel more confident in recommending the vaccines.”
A version of this article first appeared on Medscape.com.