User login
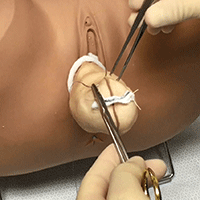
Complete colpectomy & colpocleisis: Model for simulation

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist
This video is brought to you by
View more videos from SGS here
Atrial Fibrillation
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Primary Prevention of Coronary Artery Disease
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Asthma in 2016
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: PBC patients with compensated cirrhosis fare well on obeticholic acid
BOSTON – Patients with primary biliary cholangitis (PBC) who have compensated cirrhosis fared just as well on obeticholic acid (OCA) as did PBC patients without cirrhosis, according to an analysis of data from POISE, the pivotal clinical trial for approval of OCA for PBC.
The POISE trial included 36 individuals with PBC and compensated cirrhosis, since cirrhosis “is an endpoint for virtually all liver diseases,” John Vierling, MD, said in a video interview at the meeting.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
To see how this group fared, Dr. Vierling and his coinvestigators performed a post hoc analysis of the POISE data to examine OCA’s safety and efficacy for patients with compensated cirrhosis. Patients with decompensated cirrhosis were not included in the trial.
Dr. Vierling, chief of hepatology at Baylor College of Medicine, Houston, noted that investigators worked hard to set the bar high for inclusion in the group with cirrhosis, to achieve very high specificity. “We did this by using very stringent criteria of liver biopsy, or transient elastography adjusted for a very high range of kilopascals required to diagnose cirrhosis in cholestatic patients,” he said. To be included, patients also had to have elevated total bilirubin levels and a baseline alkaline phosphatase level greater than five times the upper limit of normal.
Statistically, the patients were evenly distributed across the placebo arm and the two treatment arms, one of which dosed OCA at 10 mg/day; the other treatment arm had flexible dosing at 5-10 mg/day.
The POISE trial used a composite primary efficacy endpoint of achieving an alkaline phosphatase (ALP) less than 1.67 times the upper limit of normal, with total bilirubin within normal limits, and at least a 15% reduction in ALP.
“Significantly more OCA-treated patients with cirrhosis achieved the primary composite endpoint compared to placebo,” Dr. Vierling and his coauthors wrote in a poster presented at the annual meeting for the American Association for the Study of Liver Diseases. The difference was individually significant for all three values that made up the composite primary endpoint as well.
Secondary endpoints included gamma-glutamyltransferase, alanine aminotrasferase, and aspartate aminotransferase, all of which were significantly reduced among patients taking OCA. Patients on placebo saw these values rise over the time period of the study.
There were no new safety signals seen in the post hoc analysis of the group with cirrhosis that were not seen in the trial at large, said Dr. Vierling. Two individuals in the subgroup dropped out of the trial because of pruritis, a similar proportion to that seen in the full trial population.
The drug’s manufacturer, Intercept Pharmaceuticals, is working with the Food and Drug Administration to establish appropriate doses and intervals for obeticholic acid so it may be used safely in individuals with decompensated cirrhosis, said Dr. Vierling.
Obeticholic acid, a farnesoid-X receptor agonist, is an approved agent to use as add-on therapy to ursodeoxycholic acid (UDCA), or as monotherapy for patients who can’t tolerate UDCA.
Dr. Vierling disclosed financial relationships with Intercept Pharmaceuticals and with several other pharmaceutical companies. The study was funded by Intercept Pharmaceuticals.
koakes@frontlinemedcom.com
On Twitter @karioakes
BOSTON – Patients with primary biliary cholangitis (PBC) who have compensated cirrhosis fared just as well on obeticholic acid (OCA) as did PBC patients without cirrhosis, according to an analysis of data from POISE, the pivotal clinical trial for approval of OCA for PBC.
The POISE trial included 36 individuals with PBC and compensated cirrhosis, since cirrhosis “is an endpoint for virtually all liver diseases,” John Vierling, MD, said in a video interview at the meeting.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
To see how this group fared, Dr. Vierling and his coinvestigators performed a post hoc analysis of the POISE data to examine OCA’s safety and efficacy for patients with compensated cirrhosis. Patients with decompensated cirrhosis were not included in the trial.
Dr. Vierling, chief of hepatology at Baylor College of Medicine, Houston, noted that investigators worked hard to set the bar high for inclusion in the group with cirrhosis, to achieve very high specificity. “We did this by using very stringent criteria of liver biopsy, or transient elastography adjusted for a very high range of kilopascals required to diagnose cirrhosis in cholestatic patients,” he said. To be included, patients also had to have elevated total bilirubin levels and a baseline alkaline phosphatase level greater than five times the upper limit of normal.
Statistically, the patients were evenly distributed across the placebo arm and the two treatment arms, one of which dosed OCA at 10 mg/day; the other treatment arm had flexible dosing at 5-10 mg/day.
The POISE trial used a composite primary efficacy endpoint of achieving an alkaline phosphatase (ALP) less than 1.67 times the upper limit of normal, with total bilirubin within normal limits, and at least a 15% reduction in ALP.
“Significantly more OCA-treated patients with cirrhosis achieved the primary composite endpoint compared to placebo,” Dr. Vierling and his coauthors wrote in a poster presented at the annual meeting for the American Association for the Study of Liver Diseases. The difference was individually significant for all three values that made up the composite primary endpoint as well.
Secondary endpoints included gamma-glutamyltransferase, alanine aminotrasferase, and aspartate aminotransferase, all of which were significantly reduced among patients taking OCA. Patients on placebo saw these values rise over the time period of the study.
There were no new safety signals seen in the post hoc analysis of the group with cirrhosis that were not seen in the trial at large, said Dr. Vierling. Two individuals in the subgroup dropped out of the trial because of pruritis, a similar proportion to that seen in the full trial population.
The drug’s manufacturer, Intercept Pharmaceuticals, is working with the Food and Drug Administration to establish appropriate doses and intervals for obeticholic acid so it may be used safely in individuals with decompensated cirrhosis, said Dr. Vierling.
Obeticholic acid, a farnesoid-X receptor agonist, is an approved agent to use as add-on therapy to ursodeoxycholic acid (UDCA), or as monotherapy for patients who can’t tolerate UDCA.
Dr. Vierling disclosed financial relationships with Intercept Pharmaceuticals and with several other pharmaceutical companies. The study was funded by Intercept Pharmaceuticals.
koakes@frontlinemedcom.com
On Twitter @karioakes
BOSTON – Patients with primary biliary cholangitis (PBC) who have compensated cirrhosis fared just as well on obeticholic acid (OCA) as did PBC patients without cirrhosis, according to an analysis of data from POISE, the pivotal clinical trial for approval of OCA for PBC.
The POISE trial included 36 individuals with PBC and compensated cirrhosis, since cirrhosis “is an endpoint for virtually all liver diseases,” John Vierling, MD, said in a video interview at the meeting.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
To see how this group fared, Dr. Vierling and his coinvestigators performed a post hoc analysis of the POISE data to examine OCA’s safety and efficacy for patients with compensated cirrhosis. Patients with decompensated cirrhosis were not included in the trial.
Dr. Vierling, chief of hepatology at Baylor College of Medicine, Houston, noted that investigators worked hard to set the bar high for inclusion in the group with cirrhosis, to achieve very high specificity. “We did this by using very stringent criteria of liver biopsy, or transient elastography adjusted for a very high range of kilopascals required to diagnose cirrhosis in cholestatic patients,” he said. To be included, patients also had to have elevated total bilirubin levels and a baseline alkaline phosphatase level greater than five times the upper limit of normal.
Statistically, the patients were evenly distributed across the placebo arm and the two treatment arms, one of which dosed OCA at 10 mg/day; the other treatment arm had flexible dosing at 5-10 mg/day.
The POISE trial used a composite primary efficacy endpoint of achieving an alkaline phosphatase (ALP) less than 1.67 times the upper limit of normal, with total bilirubin within normal limits, and at least a 15% reduction in ALP.
“Significantly more OCA-treated patients with cirrhosis achieved the primary composite endpoint compared to placebo,” Dr. Vierling and his coauthors wrote in a poster presented at the annual meeting for the American Association for the Study of Liver Diseases. The difference was individually significant for all three values that made up the composite primary endpoint as well.
Secondary endpoints included gamma-glutamyltransferase, alanine aminotrasferase, and aspartate aminotransferase, all of which were significantly reduced among patients taking OCA. Patients on placebo saw these values rise over the time period of the study.
There were no new safety signals seen in the post hoc analysis of the group with cirrhosis that were not seen in the trial at large, said Dr. Vierling. Two individuals in the subgroup dropped out of the trial because of pruritis, a similar proportion to that seen in the full trial population.
The drug’s manufacturer, Intercept Pharmaceuticals, is working with the Food and Drug Administration to establish appropriate doses and intervals for obeticholic acid so it may be used safely in individuals with decompensated cirrhosis, said Dr. Vierling.
Obeticholic acid, a farnesoid-X receptor agonist, is an approved agent to use as add-on therapy to ursodeoxycholic acid (UDCA), or as monotherapy for patients who can’t tolerate UDCA.
Dr. Vierling disclosed financial relationships with Intercept Pharmaceuticals and with several other pharmaceutical companies. The study was funded by Intercept Pharmaceuticals.
koakes@frontlinemedcom.com
On Twitter @karioakes
EXPERT ANALYSIS FROM THE LIVER MEETING
VIDEO: Biologics: Proposed guideline addresses perioperative management
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ACR ANNUAL MEETING
VIDEO: Bariatric surgery may protect against heart failure
NEW ORLEANS – Results of a new 40,000-patient Swedish observational study provide the strongest evidence to date suggesting a causal relationship between bariatric surgery and reduced risk of heart failure, according to Johan Sundström, MD.
The study, which included patients drawn from two large Swedish national registries, demonstrated that bariatric surgery was associated with a 46% reduction in the incidence of heart failure during a median 4.1 years of follow-up, compared with an intensive lifestyle modification program for weight loss.
“These are observational data, but it’s a very large study population – and probably there will never be a large randomized trial of bariatric surgery versus weight loss through intensive lifestyle modification as a means of reducing the risk of heart failure,” Dr. Sundström, professor of epidemiology and a cardiologist at Uppsala (Sweden) University, said at the American Heart Association scientific sessions.
The study included 25,804 bariatric surgery patients in SOReg, the Scandinavian Obesity Surgery Registry, and a matched comparator group of 13,701 participants in a Swedish national registry of obese participants in a commercial Sweden-based intensive structural lifestyle modification program for weight loss called Itrim. The two groups were matched for baseline body mass index, which was a mean of 41.5 kg, and numerous other demographic factors and comorbid conditions. Participants weighed an average of 119 kg at baseline. None of the subjects had a history of heart failure.
The bariatric surgery group lost substantially more weight than did the lifestyle modification group: an average loss of about 35 kg after 1 year, which was 18.8 kg more than in the lifestyle modification group. After 2 years, the bariatric surgery group had an average of 22.6 kg more weight loss than did the comparison group.
The primary outcome was hospitalization for new-onset heart failure during a median 4.1 years of follow-up. Subjects were well below the age range when the incidence of heart failure accelerates – they averaged 41 years of age – but 73 of them did develop heart failure during follow-up. The incidence was 46% lower in the bariatric surgery patients. This supports the study hypothesis that bariatric surgery leads to a low incidence of new-onset heart failure, compared with intensive lifestyle modification because of its larger weight loss effect.
When Dr. Sundström and his coinvestigators combined the two study groups, they found that a 10-kg weight loss at 1 year was associated with a 23% reduction in the risk of heart failure during follow-up, irrespective of whether the weight loss was achieved surgically or through the lifestyle program.
“A great way of studying causality is to take away the exposure and note what happens to the outcome. If there’s a causal link, then if you take away the risk factor – in this case, obesity – the disease should go away,” he explained in a video interview.
The reduced risk of heart failure in the bariatric surgery patients wasn’t because of fewer acute MIs. Indeed, their acute MI rate during follow-up was similar to that of the lifestyle modification group. But bariatric surgery was associated with relative risk reductions of 35%-37% for atrial fibrillation or need for diabetes or blood pressure–lowering medications at 1 year – and atrial fibrillation, diabetes, and hypertension are all established risk factors for heart failure, Dr. Sundström noted.
The Itrim intensive lifestyle modification program entailed an initial very-low-energy diet for the first 3 months in order to achieve massive weight loss, followed by a 9-month maintenance program involving motivational counseling, exercise, behavioral therapy, and a restricted diet.
Dr. Sundström said he and his coinvestigators plan to continue the study and expand it to look at differences in additional cardiovascular endpoints as patients age.
The study was funded by the U.S. National Institute of Diabetes and Digestive and Kidney Diseases, Uppsala University, the Karolinska Institute, and the Swedish Research Council. Dr. Sundström reported serving as a scientific advisor to Itrim.
NEW ORLEANS – Results of a new 40,000-patient Swedish observational study provide the strongest evidence to date suggesting a causal relationship between bariatric surgery and reduced risk of heart failure, according to Johan Sundström, MD.
The study, which included patients drawn from two large Swedish national registries, demonstrated that bariatric surgery was associated with a 46% reduction in the incidence of heart failure during a median 4.1 years of follow-up, compared with an intensive lifestyle modification program for weight loss.
“These are observational data, but it’s a very large study population – and probably there will never be a large randomized trial of bariatric surgery versus weight loss through intensive lifestyle modification as a means of reducing the risk of heart failure,” Dr. Sundström, professor of epidemiology and a cardiologist at Uppsala (Sweden) University, said at the American Heart Association scientific sessions.
The study included 25,804 bariatric surgery patients in SOReg, the Scandinavian Obesity Surgery Registry, and a matched comparator group of 13,701 participants in a Swedish national registry of obese participants in a commercial Sweden-based intensive structural lifestyle modification program for weight loss called Itrim. The two groups were matched for baseline body mass index, which was a mean of 41.5 kg, and numerous other demographic factors and comorbid conditions. Participants weighed an average of 119 kg at baseline. None of the subjects had a history of heart failure.
The bariatric surgery group lost substantially more weight than did the lifestyle modification group: an average loss of about 35 kg after 1 year, which was 18.8 kg more than in the lifestyle modification group. After 2 years, the bariatric surgery group had an average of 22.6 kg more weight loss than did the comparison group.
The primary outcome was hospitalization for new-onset heart failure during a median 4.1 years of follow-up. Subjects were well below the age range when the incidence of heart failure accelerates – they averaged 41 years of age – but 73 of them did develop heart failure during follow-up. The incidence was 46% lower in the bariatric surgery patients. This supports the study hypothesis that bariatric surgery leads to a low incidence of new-onset heart failure, compared with intensive lifestyle modification because of its larger weight loss effect.
When Dr. Sundström and his coinvestigators combined the two study groups, they found that a 10-kg weight loss at 1 year was associated with a 23% reduction in the risk of heart failure during follow-up, irrespective of whether the weight loss was achieved surgically or through the lifestyle program.
“A great way of studying causality is to take away the exposure and note what happens to the outcome. If there’s a causal link, then if you take away the risk factor – in this case, obesity – the disease should go away,” he explained in a video interview.
The reduced risk of heart failure in the bariatric surgery patients wasn’t because of fewer acute MIs. Indeed, their acute MI rate during follow-up was similar to that of the lifestyle modification group. But bariatric surgery was associated with relative risk reductions of 35%-37% for atrial fibrillation or need for diabetes or blood pressure–lowering medications at 1 year – and atrial fibrillation, diabetes, and hypertension are all established risk factors for heart failure, Dr. Sundström noted.
The Itrim intensive lifestyle modification program entailed an initial very-low-energy diet for the first 3 months in order to achieve massive weight loss, followed by a 9-month maintenance program involving motivational counseling, exercise, behavioral therapy, and a restricted diet.
Dr. Sundström said he and his coinvestigators plan to continue the study and expand it to look at differences in additional cardiovascular endpoints as patients age.
The study was funded by the U.S. National Institute of Diabetes and Digestive and Kidney Diseases, Uppsala University, the Karolinska Institute, and the Swedish Research Council. Dr. Sundström reported serving as a scientific advisor to Itrim.
NEW ORLEANS – Results of a new 40,000-patient Swedish observational study provide the strongest evidence to date suggesting a causal relationship between bariatric surgery and reduced risk of heart failure, according to Johan Sundström, MD.
The study, which included patients drawn from two large Swedish national registries, demonstrated that bariatric surgery was associated with a 46% reduction in the incidence of heart failure during a median 4.1 years of follow-up, compared with an intensive lifestyle modification program for weight loss.
“These are observational data, but it’s a very large study population – and probably there will never be a large randomized trial of bariatric surgery versus weight loss through intensive lifestyle modification as a means of reducing the risk of heart failure,” Dr. Sundström, professor of epidemiology and a cardiologist at Uppsala (Sweden) University, said at the American Heart Association scientific sessions.
The study included 25,804 bariatric surgery patients in SOReg, the Scandinavian Obesity Surgery Registry, and a matched comparator group of 13,701 participants in a Swedish national registry of obese participants in a commercial Sweden-based intensive structural lifestyle modification program for weight loss called Itrim. The two groups were matched for baseline body mass index, which was a mean of 41.5 kg, and numerous other demographic factors and comorbid conditions. Participants weighed an average of 119 kg at baseline. None of the subjects had a history of heart failure.
The bariatric surgery group lost substantially more weight than did the lifestyle modification group: an average loss of about 35 kg after 1 year, which was 18.8 kg more than in the lifestyle modification group. After 2 years, the bariatric surgery group had an average of 22.6 kg more weight loss than did the comparison group.
The primary outcome was hospitalization for new-onset heart failure during a median 4.1 years of follow-up. Subjects were well below the age range when the incidence of heart failure accelerates – they averaged 41 years of age – but 73 of them did develop heart failure during follow-up. The incidence was 46% lower in the bariatric surgery patients. This supports the study hypothesis that bariatric surgery leads to a low incidence of new-onset heart failure, compared with intensive lifestyle modification because of its larger weight loss effect.
When Dr. Sundström and his coinvestigators combined the two study groups, they found that a 10-kg weight loss at 1 year was associated with a 23% reduction in the risk of heart failure during follow-up, irrespective of whether the weight loss was achieved surgically or through the lifestyle program.
“A great way of studying causality is to take away the exposure and note what happens to the outcome. If there’s a causal link, then if you take away the risk factor – in this case, obesity – the disease should go away,” he explained in a video interview.
The reduced risk of heart failure in the bariatric surgery patients wasn’t because of fewer acute MIs. Indeed, their acute MI rate during follow-up was similar to that of the lifestyle modification group. But bariatric surgery was associated with relative risk reductions of 35%-37% for atrial fibrillation or need for diabetes or blood pressure–lowering medications at 1 year – and atrial fibrillation, diabetes, and hypertension are all established risk factors for heart failure, Dr. Sundström noted.
The Itrim intensive lifestyle modification program entailed an initial very-low-energy diet for the first 3 months in order to achieve massive weight loss, followed by a 9-month maintenance program involving motivational counseling, exercise, behavioral therapy, and a restricted diet.
Dr. Sundström said he and his coinvestigators plan to continue the study and expand it to look at differences in additional cardiovascular endpoints as patients age.
The study was funded by the U.S. National Institute of Diabetes and Digestive and Kidney Diseases, Uppsala University, the Karolinska Institute, and the Swedish Research Council. Dr. Sundström reported serving as a scientific advisor to Itrim.
AT THE AHA SCIENTIFIC SESSIONS 2016
Key clinical point:
Major finding: The incidence of new-onset heart failure was 46% lower during follow-up after bariatric surgery than among participants in an intensive lifestyle modification program for weight loss.
Data source: This observational registry study followed nearly 26,000 Swedish bariatric surgery patients and 14,000 matched participants in a commercial intensive lifestyle modification program for a median of 4.1 years.
Disclosures: The study was funded by the U.S. National Institutes of Diabetes and Digestive and Kidney Diseases, Uppsala University, the Karolinska Institute, and the Swedish Research Council. The presenter reported serving as a scientific advisor to Itrim.
VIDEO: Don’t be surprised by weight gain in men after HCV cure
BOSTON – In the new era of direct-acting antiviral (DAA) therapy, physicians will be seeing more and more patients who have achieved a cure of their hepatitis C virus (HCV). Once freed from the burden of a chronic illness, patients feel better and may eat better. Unexpected weight gain and potential associated health effects may be the next set of challenges patients and their physicians will face.
A single-center retrospective study of patients who had achieved sustained virologic response (SVR) after treatment for HCV found a small but significant weight gain in men, but not women. Additionally, according to noninvasive assessments, liver fat increased significantly in men, but not women, after SVR was achieved.
In a study of 63 patients (42 male, 67%) who received DAA treatment for HCV, mean weight gain for men after SVR was 2.8 pounds (range, –26 to +17; P = .0459), and body mass index (BMI) increased by a mean 0.50 kg/m2 (range, –3.6 to +3.33; P = .0176). No significant change was seen for women when pre- and posttreatment measures were compared.
Isaac Wasserman, a medical student at Mount Sinai Medical Center, New York, presented the results of the single-center retrospective study in a poster presentation at the annual meeting of the American Association for the Study of Liver Diseases.
To assess changes in liver fat, Mr. Wasserman and his coinvestigator used results of pre- and posttreatment transient elastography with controlled attenuation parameter (CAP). CAP measures the degree to which the ultrasound signal is attenuated by liver fat, he explained in a video interview.
For men, hepatic steatosis increased by this measure, with CAP measurements up by a mean 18 dB/m (range, –106 to +128, P = .0314). Mr. Wasserman and his colleagues wrote, “The change in liver fat was large enough to push 11% of the cohort (n = 7 of 63) into advanced steatosis (CAP greater than 300 dB/m).” Again, the women studied had no significant posttreatment change in liver fat.
Post-SVR weight gain appeared to be the culprit in the increased fat seen in the posttreatment livers. Mr. Wasserman and his colleagues in the abstract accompanying the presentation, “Changes in weight were positively correlated with changes in liver fat (P = .006).”
Mr. Wasserman said that he and his coinvestigators believe that social, and not biochemical or mechanistic, reasons underlie the weight gain and increased hepatic steatosis. They are planning further investigation of social and economic factors that may underlie the difference seen in this study, and hope to continue and expand data acquisition to validate their findings.
Mr. Wasserman reported no conflicts of interest or outside sources of funding for the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @karioakes
BOSTON – In the new era of direct-acting antiviral (DAA) therapy, physicians will be seeing more and more patients who have achieved a cure of their hepatitis C virus (HCV). Once freed from the burden of a chronic illness, patients feel better and may eat better. Unexpected weight gain and potential associated health effects may be the next set of challenges patients and their physicians will face.
A single-center retrospective study of patients who had achieved sustained virologic response (SVR) after treatment for HCV found a small but significant weight gain in men, but not women. Additionally, according to noninvasive assessments, liver fat increased significantly in men, but not women, after SVR was achieved.
In a study of 63 patients (42 male, 67%) who received DAA treatment for HCV, mean weight gain for men after SVR was 2.8 pounds (range, –26 to +17; P = .0459), and body mass index (BMI) increased by a mean 0.50 kg/m2 (range, –3.6 to +3.33; P = .0176). No significant change was seen for women when pre- and posttreatment measures were compared.
Isaac Wasserman, a medical student at Mount Sinai Medical Center, New York, presented the results of the single-center retrospective study in a poster presentation at the annual meeting of the American Association for the Study of Liver Diseases.
To assess changes in liver fat, Mr. Wasserman and his coinvestigator used results of pre- and posttreatment transient elastography with controlled attenuation parameter (CAP). CAP measures the degree to which the ultrasound signal is attenuated by liver fat, he explained in a video interview.
For men, hepatic steatosis increased by this measure, with CAP measurements up by a mean 18 dB/m (range, –106 to +128, P = .0314). Mr. Wasserman and his colleagues wrote, “The change in liver fat was large enough to push 11% of the cohort (n = 7 of 63) into advanced steatosis (CAP greater than 300 dB/m).” Again, the women studied had no significant posttreatment change in liver fat.
Post-SVR weight gain appeared to be the culprit in the increased fat seen in the posttreatment livers. Mr. Wasserman and his colleagues in the abstract accompanying the presentation, “Changes in weight were positively correlated with changes in liver fat (P = .006).”
Mr. Wasserman said that he and his coinvestigators believe that social, and not biochemical or mechanistic, reasons underlie the weight gain and increased hepatic steatosis. They are planning further investigation of social and economic factors that may underlie the difference seen in this study, and hope to continue and expand data acquisition to validate their findings.
Mr. Wasserman reported no conflicts of interest or outside sources of funding for the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @karioakes
BOSTON – In the new era of direct-acting antiviral (DAA) therapy, physicians will be seeing more and more patients who have achieved a cure of their hepatitis C virus (HCV). Once freed from the burden of a chronic illness, patients feel better and may eat better. Unexpected weight gain and potential associated health effects may be the next set of challenges patients and their physicians will face.
A single-center retrospective study of patients who had achieved sustained virologic response (SVR) after treatment for HCV found a small but significant weight gain in men, but not women. Additionally, according to noninvasive assessments, liver fat increased significantly in men, but not women, after SVR was achieved.
In a study of 63 patients (42 male, 67%) who received DAA treatment for HCV, mean weight gain for men after SVR was 2.8 pounds (range, –26 to +17; P = .0459), and body mass index (BMI) increased by a mean 0.50 kg/m2 (range, –3.6 to +3.33; P = .0176). No significant change was seen for women when pre- and posttreatment measures were compared.
Isaac Wasserman, a medical student at Mount Sinai Medical Center, New York, presented the results of the single-center retrospective study in a poster presentation at the annual meeting of the American Association for the Study of Liver Diseases.
To assess changes in liver fat, Mr. Wasserman and his coinvestigator used results of pre- and posttreatment transient elastography with controlled attenuation parameter (CAP). CAP measures the degree to which the ultrasound signal is attenuated by liver fat, he explained in a video interview.
For men, hepatic steatosis increased by this measure, with CAP measurements up by a mean 18 dB/m (range, –106 to +128, P = .0314). Mr. Wasserman and his colleagues wrote, “The change in liver fat was large enough to push 11% of the cohort (n = 7 of 63) into advanced steatosis (CAP greater than 300 dB/m).” Again, the women studied had no significant posttreatment change in liver fat.
Post-SVR weight gain appeared to be the culprit in the increased fat seen in the posttreatment livers. Mr. Wasserman and his colleagues in the abstract accompanying the presentation, “Changes in weight were positively correlated with changes in liver fat (P = .006).”
Mr. Wasserman said that he and his coinvestigators believe that social, and not biochemical or mechanistic, reasons underlie the weight gain and increased hepatic steatosis. They are planning further investigation of social and economic factors that may underlie the difference seen in this study, and hope to continue and expand data acquisition to validate their findings.
Mr. Wasserman reported no conflicts of interest or outside sources of funding for the study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @karioakes
AT THE LIVER MEETING
VIDEO: For CABG, double arterial grafts found no better than single
NEW ORLEANS – Patients undergoing coronary artery bypass graft (CABG) surgery do not see any 5-year survival advantage when their surgeon uses both internal mammary (thoracic) arteries for grafting rather than just one of them along with a vein, finds an interim analysis from the randomized Arterial Revascularization Trial (ART).
Overall, about 8.5% of the 3,102 patients randomized had died 5 years after surgery, with no significant difference between the bilateral graft and single graft groups, according to data reported at the American Heart Association scientific sessions and simultaneously published (N Engl J Med. 2016 Nov 14. doi: 10.1056/NEJMoa1610021). The former had roughly triple the rate of sternal reconstruction, mainly driven by complications in insulin-dependent diabetes patients having high body mass index.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
There is strong angiographic evidence that vein grafts have a high rate of failure over time because of atherosclerosis, but internal mammary artery grafts retain excellent patency, he elaborated. “People have speculated that this superior patency of internal mammary arteries will translate into a clinical survival benefit,” and observational data indeed suggest that the bilateral artery strategy reduces mortality by about one-fifth relative to the single artery strategy.
Yet uptake of the bilateral procedure has been low. It is used in fewer than 5% of patients undergoing CABG in the United States and fewer than 10% of those in Europe, reflecting concerns about its greater technical complexity, potentially increased mortality and morbidity, and – until now – lack of evidence from randomized trials.
“What I think we can conclude today is that there are excellent 5-year outcomes of CABG in both groups. This study confirms that it’s at least safe to use bilateral grafts over the medium term,” Dr. Taggart commented. He discussed the results in a video interview conducted at the meeting.
These interim ART data probably won’t sway practice one way or the other, he said. “People who believe in arterial grafts will continue to do them, and those who are not enthusiastic about the prospect of a slightly technically more difficult operation [can now] remain comfortable as to why they are not using both internal mammary arteries.”
Pointed questions
The lack of difference was possibly due to a very high level of guideline-based medical therapy in the trial (which may have especially protected the vein grafts) or to the fact that the annual failure rate of vein grafts is modest and steady up to 5 years but accelerates thereafter, Dr. Taggart proposed. The trial’s primary outcome of 10-year survival, expected in 2018, will likely differ, speculated Dr. Sellke, who is also program chair for the AHA scientific sessions.
“Do you think multiple arterial grafting is superior to single internal mammary artery grafting considering the lack of improvement in survival and other outcomes in the study, with the increase in sternal wound infections?” he asked.
“I personally, if I needed the operation, would insist on having bilateral internal mammary artery grafts done by an experienced operator because it is totally counterintuitive to believe that having more patent grafts in your heart at 10 to 20 years of follow-up is of no benefit,” Dr. Taggart maintained.
When data meet clinical practice
It may require time for the benefit of the bilateral artery graft to emerge, he agreed. “I’m undeterred from my belief that ... in patients who are getting CABG done in their 40s or 50s or early 60s, betting on a graft that’s going to outperform vein grafts is the better strategy.”
Until the trial’s 10-year results become available, physicians may wish to put these interim results in the context when counseling patients, according to Dr. Gardner.
“We have indisputable evidence that arterial grafts have better long-term patency than vein grafts,” he elaborated. “If we had a very sophisticated patient, we might tell them that we were a little surprised that this head-to-head trial of single versus double didn’t show any survival benefit at 5 years, but we still are persuaded by the data that shows the better patency, and we think in the situation that the patient’s in, that we would still recommend a double mammary, assuming that the patient doesn’t have comorbidities that would make that more dangerous.”
Trial details
ART enrolled patients from 28 cardiac surgical centers in seven countries. The patients, all of whom had multivessel coronary disease and were scheduled to undergo CABG, were randomized evenly to single or bilateral internal thoracic artery grafts.
The interim results showed differences in nonadherence to the randomized operation: 2.4% of patients in the single graft group ultimately underwent got bilateral grafts, whereas 14% of patients in the bilateral graft group ultimately got a single graft.
“This raises questions about how experienced some surgeons were with the use of bilateral internal mammary artery grafts,” Dr. Taggart commented.
At 5 years of follow-up, 8.7% of patients in the bilateral graft group and 8.4% of patients in the single graft group had died, a nonsignificant difference. “Those mortalities are similar to what has been observed in other contemporary trials of CABG,” he noted. There was no difference between diabetic and nondiabetic patients with respect to this outcome.
The rate of the composite outcome of death, myocardial infarction, or stroke was 12.2% in the bilateral graft group and 12.7% in the single graft group, also a nonsignificant difference.
On the other hand, patients in the bilateral graft group had higher rates of sternal wound complications (3.5% vs. 1.9%; P = .005) and sternal reconstruction (1.9% vs. 0.6%; P = .002).
The groups were statistically indistinguishable with respect to rates of mortality, major bleeding, or need for repeat revascularization, as well as angina status and quality of life measures, according to Dr. Taggart, who disclosed that he had no relevant conflicts of interest.
NEW ORLEANS – Patients undergoing coronary artery bypass graft (CABG) surgery do not see any 5-year survival advantage when their surgeon uses both internal mammary (thoracic) arteries for grafting rather than just one of them along with a vein, finds an interim analysis from the randomized Arterial Revascularization Trial (ART).
Overall, about 8.5% of the 3,102 patients randomized had died 5 years after surgery, with no significant difference between the bilateral graft and single graft groups, according to data reported at the American Heart Association scientific sessions and simultaneously published (N Engl J Med. 2016 Nov 14. doi: 10.1056/NEJMoa1610021). The former had roughly triple the rate of sternal reconstruction, mainly driven by complications in insulin-dependent diabetes patients having high body mass index.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
There is strong angiographic evidence that vein grafts have a high rate of failure over time because of atherosclerosis, but internal mammary artery grafts retain excellent patency, he elaborated. “People have speculated that this superior patency of internal mammary arteries will translate into a clinical survival benefit,” and observational data indeed suggest that the bilateral artery strategy reduces mortality by about one-fifth relative to the single artery strategy.
Yet uptake of the bilateral procedure has been low. It is used in fewer than 5% of patients undergoing CABG in the United States and fewer than 10% of those in Europe, reflecting concerns about its greater technical complexity, potentially increased mortality and morbidity, and – until now – lack of evidence from randomized trials.
“What I think we can conclude today is that there are excellent 5-year outcomes of CABG in both groups. This study confirms that it’s at least safe to use bilateral grafts over the medium term,” Dr. Taggart commented. He discussed the results in a video interview conducted at the meeting.
These interim ART data probably won’t sway practice one way or the other, he said. “People who believe in arterial grafts will continue to do them, and those who are not enthusiastic about the prospect of a slightly technically more difficult operation [can now] remain comfortable as to why they are not using both internal mammary arteries.”
Pointed questions
The lack of difference was possibly due to a very high level of guideline-based medical therapy in the trial (which may have especially protected the vein grafts) or to the fact that the annual failure rate of vein grafts is modest and steady up to 5 years but accelerates thereafter, Dr. Taggart proposed. The trial’s primary outcome of 10-year survival, expected in 2018, will likely differ, speculated Dr. Sellke, who is also program chair for the AHA scientific sessions.
“Do you think multiple arterial grafting is superior to single internal mammary artery grafting considering the lack of improvement in survival and other outcomes in the study, with the increase in sternal wound infections?” he asked.
“I personally, if I needed the operation, would insist on having bilateral internal mammary artery grafts done by an experienced operator because it is totally counterintuitive to believe that having more patent grafts in your heart at 10 to 20 years of follow-up is of no benefit,” Dr. Taggart maintained.
When data meet clinical practice
It may require time for the benefit of the bilateral artery graft to emerge, he agreed. “I’m undeterred from my belief that ... in patients who are getting CABG done in their 40s or 50s or early 60s, betting on a graft that’s going to outperform vein grafts is the better strategy.”
Until the trial’s 10-year results become available, physicians may wish to put these interim results in the context when counseling patients, according to Dr. Gardner.
“We have indisputable evidence that arterial grafts have better long-term patency than vein grafts,” he elaborated. “If we had a very sophisticated patient, we might tell them that we were a little surprised that this head-to-head trial of single versus double didn’t show any survival benefit at 5 years, but we still are persuaded by the data that shows the better patency, and we think in the situation that the patient’s in, that we would still recommend a double mammary, assuming that the patient doesn’t have comorbidities that would make that more dangerous.”
Trial details
ART enrolled patients from 28 cardiac surgical centers in seven countries. The patients, all of whom had multivessel coronary disease and were scheduled to undergo CABG, were randomized evenly to single or bilateral internal thoracic artery grafts.
The interim results showed differences in nonadherence to the randomized operation: 2.4% of patients in the single graft group ultimately underwent got bilateral grafts, whereas 14% of patients in the bilateral graft group ultimately got a single graft.
“This raises questions about how experienced some surgeons were with the use of bilateral internal mammary artery grafts,” Dr. Taggart commented.
At 5 years of follow-up, 8.7% of patients in the bilateral graft group and 8.4% of patients in the single graft group had died, a nonsignificant difference. “Those mortalities are similar to what has been observed in other contemporary trials of CABG,” he noted. There was no difference between diabetic and nondiabetic patients with respect to this outcome.
The rate of the composite outcome of death, myocardial infarction, or stroke was 12.2% in the bilateral graft group and 12.7% in the single graft group, also a nonsignificant difference.
On the other hand, patients in the bilateral graft group had higher rates of sternal wound complications (3.5% vs. 1.9%; P = .005) and sternal reconstruction (1.9% vs. 0.6%; P = .002).
The groups were statistically indistinguishable with respect to rates of mortality, major bleeding, or need for repeat revascularization, as well as angina status and quality of life measures, according to Dr. Taggart, who disclosed that he had no relevant conflicts of interest.
NEW ORLEANS – Patients undergoing coronary artery bypass graft (CABG) surgery do not see any 5-year survival advantage when their surgeon uses both internal mammary (thoracic) arteries for grafting rather than just one of them along with a vein, finds an interim analysis from the randomized Arterial Revascularization Trial (ART).
Overall, about 8.5% of the 3,102 patients randomized had died 5 years after surgery, with no significant difference between the bilateral graft and single graft groups, according to data reported at the American Heart Association scientific sessions and simultaneously published (N Engl J Med. 2016 Nov 14. doi: 10.1056/NEJMoa1610021). The former had roughly triple the rate of sternal reconstruction, mainly driven by complications in insulin-dependent diabetes patients having high body mass index.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
There is strong angiographic evidence that vein grafts have a high rate of failure over time because of atherosclerosis, but internal mammary artery grafts retain excellent patency, he elaborated. “People have speculated that this superior patency of internal mammary arteries will translate into a clinical survival benefit,” and observational data indeed suggest that the bilateral artery strategy reduces mortality by about one-fifth relative to the single artery strategy.
Yet uptake of the bilateral procedure has been low. It is used in fewer than 5% of patients undergoing CABG in the United States and fewer than 10% of those in Europe, reflecting concerns about its greater technical complexity, potentially increased mortality and morbidity, and – until now – lack of evidence from randomized trials.
“What I think we can conclude today is that there are excellent 5-year outcomes of CABG in both groups. This study confirms that it’s at least safe to use bilateral grafts over the medium term,” Dr. Taggart commented. He discussed the results in a video interview conducted at the meeting.
These interim ART data probably won’t sway practice one way or the other, he said. “People who believe in arterial grafts will continue to do them, and those who are not enthusiastic about the prospect of a slightly technically more difficult operation [can now] remain comfortable as to why they are not using both internal mammary arteries.”
Pointed questions
The lack of difference was possibly due to a very high level of guideline-based medical therapy in the trial (which may have especially protected the vein grafts) or to the fact that the annual failure rate of vein grafts is modest and steady up to 5 years but accelerates thereafter, Dr. Taggart proposed. The trial’s primary outcome of 10-year survival, expected in 2018, will likely differ, speculated Dr. Sellke, who is also program chair for the AHA scientific sessions.
“Do you think multiple arterial grafting is superior to single internal mammary artery grafting considering the lack of improvement in survival and other outcomes in the study, with the increase in sternal wound infections?” he asked.
“I personally, if I needed the operation, would insist on having bilateral internal mammary artery grafts done by an experienced operator because it is totally counterintuitive to believe that having more patent grafts in your heart at 10 to 20 years of follow-up is of no benefit,” Dr. Taggart maintained.
When data meet clinical practice
It may require time for the benefit of the bilateral artery graft to emerge, he agreed. “I’m undeterred from my belief that ... in patients who are getting CABG done in their 40s or 50s or early 60s, betting on a graft that’s going to outperform vein grafts is the better strategy.”
Until the trial’s 10-year results become available, physicians may wish to put these interim results in the context when counseling patients, according to Dr. Gardner.
“We have indisputable evidence that arterial grafts have better long-term patency than vein grafts,” he elaborated. “If we had a very sophisticated patient, we might tell them that we were a little surprised that this head-to-head trial of single versus double didn’t show any survival benefit at 5 years, but we still are persuaded by the data that shows the better patency, and we think in the situation that the patient’s in, that we would still recommend a double mammary, assuming that the patient doesn’t have comorbidities that would make that more dangerous.”
Trial details
ART enrolled patients from 28 cardiac surgical centers in seven countries. The patients, all of whom had multivessel coronary disease and were scheduled to undergo CABG, were randomized evenly to single or bilateral internal thoracic artery grafts.
The interim results showed differences in nonadherence to the randomized operation: 2.4% of patients in the single graft group ultimately underwent got bilateral grafts, whereas 14% of patients in the bilateral graft group ultimately got a single graft.
“This raises questions about how experienced some surgeons were with the use of bilateral internal mammary artery grafts,” Dr. Taggart commented.
At 5 years of follow-up, 8.7% of patients in the bilateral graft group and 8.4% of patients in the single graft group had died, a nonsignificant difference. “Those mortalities are similar to what has been observed in other contemporary trials of CABG,” he noted. There was no difference between diabetic and nondiabetic patients with respect to this outcome.
The rate of the composite outcome of death, myocardial infarction, or stroke was 12.2% in the bilateral graft group and 12.7% in the single graft group, also a nonsignificant difference.
On the other hand, patients in the bilateral graft group had higher rates of sternal wound complications (3.5% vs. 1.9%; P = .005) and sternal reconstruction (1.9% vs. 0.6%; P = .002).
The groups were statistically indistinguishable with respect to rates of mortality, major bleeding, or need for repeat revascularization, as well as angina status and quality of life measures, according to Dr. Taggart, who disclosed that he had no relevant conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: At 5 years, the rate of all-cause mortality was 8.7% in the bilateral graft group and 8.4% in the single graft group, a nonsignificant difference.
Data source: ART, a randomized trial among 3,102 patients with multivessel coronary disease undergoing CABG.
Disclosures: Dr. Taggart had no relevant conflicts of interest. The trial was funded by the U.K. Medical Research Council, the British Heart Foundation, and the U.K. National Institute of Health Research Efficacy and Mechanistic Evaluation.
VIDEO: Choosing Your Path: Academic or Community Hospital Medicine?
Choosing hospital medicine as a specialty means choosing between practicing community HM or academic HM. Or does it? Elizabeth Cook, MD, of Hospital Medicine Associates in Lynchburg VA; Stella Fitzgibbon, MD, FACP, FHM, with Memorial Hermann Hospital in The Woodlands, TX; and Chris Moriates, MD, of Dell Medical School at UT Austin, talk about the options available in community and academic HM, and moving between them during an HM career.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Choosing hospital medicine as a specialty means choosing between practicing community HM or academic HM. Or does it? Elizabeth Cook, MD, of Hospital Medicine Associates in Lynchburg VA; Stella Fitzgibbon, MD, FACP, FHM, with Memorial Hermann Hospital in The Woodlands, TX; and Chris Moriates, MD, of Dell Medical School at UT Austin, talk about the options available in community and academic HM, and moving between them during an HM career.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Choosing hospital medicine as a specialty means choosing between practicing community HM or academic HM. Or does it? Elizabeth Cook, MD, of Hospital Medicine Associates in Lynchburg VA; Stella Fitzgibbon, MD, FACP, FHM, with Memorial Hermann Hospital in The Woodlands, TX; and Chris Moriates, MD, of Dell Medical School at UT Austin, talk about the options available in community and academic HM, and moving between them during an HM career.