User login
Clinical Psychiatry News is the online destination and multimedia properties of Clinica Psychiatry News, the independent news publication for psychiatrists. Since 1971, Clinical Psychiatry News has been the leading source of news and commentary about clinical developments in psychiatry as well as health care policy and regulations that affect the physician's practice.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
ketamine
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
suicide
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-cpn')]
div[contains(@class, 'pane-pub-home-cpn')]
div[contains(@class, 'pane-pub-topic-cpn')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Top questions answered about COVID-19 boosters for your patients
Confusion continues to circulate in the wake of decisions on booster doses of the Pfizer/BioNTech COVID-19 vaccine, all announced within 1 week. Many people – including those now eligible and those who officially have to wait for their shot at a third dose – have questions.
Multiple agencies are involved in the booster decisions, and they have put out multiple – and sometimes conflicting – messages about booster doses, leaving more questions than answers for many people.
On Sept. 22, the Food and Drug Administration granted an emergency use authorization (EUA) for a booster dose of the Pfizer mRNA COVID-19 vaccine for those 65 and older and those at high risk for severe illness from the coronavirus, including essential workers whose jobs increase their risk for infection – such as frontline health care workers.
The Centers for Disease Control and Prevention Director Rochelle Walensky, MD, then overruled advice from the agency’s Advisory Committee on Immunization Practices (ACIP) to recommend boosters for essential workers such as those working on the front lines during the pandemic.
As it stands now, the CDC recommends that the following groups should get a third dose of the Pfizer vaccine:
- People aged 65 years and older.
- People aged 18 years and older in long-term care settings.
- People aged 50-64 years with underlying medical conditions.
The CDC also recommends that the following groups may receive a booster shot of the Pfizer vaccine, based on their individual benefits and risks:
- People aged 18-49 years with underlying medical conditions.
- People aged 18-64 years at increased risk for COVID-19 exposure and transmission because of occupational or institutional setting.
The CDC currently considers the following groups at increased risk for COVID-19:
- First responders (health care workers, firefighters, police, congregate care staff).
- Education staff (teachers, support staff, day care workers).
- Food and agriculture workers.
- Manufacturing workers.
- Corrections workers.
- U.S. Postal Service workers.
- Public transit workers.
- Grocery store workers.
Health care professionals, among the most trusted sources of COVID-19 information, are likely to encounter a number of patients wondering how all this will work.
“It’s fantastic that boosters will be available for those who the data supports need [them],” Rachael Piltch-Loeb, PhD, said during a media briefing on Sept. 23, held between the FDA and CDC decisions.
“But we’re really in a place where we have a lot more questions and answers about what the next phase of the vaccine availability and updates are going to be in the United States,” added Dr. Piltch-Loeb, preparedness fellow in the division of policy translation and leadership development and a research associate in the department of biostatistics at the Harvard T. H. Chan School of Public Health in Boston.
1. What is the biggest concern you are hearing from patients about getting a booster?
“The biggest concerns are that everyone wants it and they don’t know where to get it. In health care’s defense, the CDC just figured out what to do,” said Janet Englund, MD, professor of pediatric infectious diseases and an infectious disease and virology expert at Seattle Children’s Hospital in Washington.
“Everyone thinks they should be eligible for a booster ... people in their 50s who are not yet 65+, people with young grandchildren, etc.,” she added. “I’m at Seattle Children’s Hospital, so people are asking about booster shots and about getting their children vaccinated.”
Boosters for all COVID-19 vaccines are completely free.
“All COVID-19 vaccines, including booster doses, will be provided free of charge to the U.S. population,” the CDC has said.
2. Will patients need to prove they meet eligibility criteria for a booster shot or will it be the honor system?
“No, patients will only need to attest that they fall into one of the high-risk groups for whom a booster vaccine is authorized,” said Robert Atmar, MD, professor of infectious diseases at Baylor College of Medicine in Houston.
Dr. Piltch-Loeb agreed. “It is likely to be an honor system. It is very unlikely that there will be punishments or other ramifications ... if doses are administered, beyond the approved usage.”
3. If a patient who had the Moderna or the Johnson and Johnson vaccination requests a booster, can health care workers give them Pfizer?
The short answer is no. “This only applies to individuals who have received the Pfizer vaccine,” Dr. Piltch-Loeb said.
More data will be needed before other vaccine boosters are authorized, she added.
“My understanding is the Moderna people have just recently submitted their information, all of their data to the FDA and J&J is in line to do that very shortly,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University in Nashville, Tenn. “I would hope that within the next month to 6 weeks, we will get information about both of those vaccines,” Dr. Schaffner said.
4. When are the “mix-and-match” vaccine study results expected to come out?
“We expect that data from the study will be available in the coming weeks,” said Dr. Atmar, who is the national co-principal investigator of a mix-and-match booster trial launched in June 2021.
5. Are side effects of a booster vaccine expected to be about the same as what people experienced during their first or second immunization?
“I’m expecting the side effects will be similar to the second dose,” Dr. Englund said.
“The data presented ... at ACIP suggests that the side effects from the third shot are either the same or actually less than the first two shots,” said Carlos del Rio, MD, distinguished professor of medicine, epidemiology, and global health, and executive associate dean of Emory University School of Medicine at Grady Health System in Atlanta.
”Everyone reacts very differently to vaccines, regardless of vaccine type,” said Eric Ascher, MD, a family medicine physician at Lenox Hill Hospital in New York City. “I have had patients (as well as personal experience) where there were none to minimal symptoms, and others who felt they had a mild flu for 24 hours.”
“I expect no side effects greater than what was felt with you prior doses,” he said. “The vaccine is very safe and the benefit of vaccination outweighs the risks of any mild side effects.”
6. Is it unethical to give a booster to someone outside the approved groups if there are doses remaining at the end of the day in an open vial?
“Offering a booster shot to someone outside of approved groups if remaining doses will go to waste at the end of the day seems like a prudent decision, and relatively harmless action,” said Faith Fletcher, PhD, assistant professor at the Center for Medical Ethics and Health Policy at Baylor College of Medicine.
“However, if doses continue to fall in the laps of unapproved groups, we must evaluate the vaccine systems and structures that advantage some groups and disadvantage others,” she added. “We know that the distribution of COVID-19 vaccines has not been equitable – and some groups have been left behind.”
“I am not an ethicist and there are many competing concerns that this question addresses,” Dr. Atmar said. For example, “there is not a limitation of vaccine supply in the U.S., so that using leftover vaccine to prevent waste is no longer a major concern in the U.S.”
It could be more of a legal than ethical question, Dr. Atmar said. For an individual outside the authorized groups, legally, the FDA’s EUA for boosting does not allow the vaccine to be administered to this person, he said.
“The rationale for the restricted use in the EUA is that at this time the safety and risks associated with such administration are not known, and the benefits also have not been determined,” Dr. Atmar said. “Members of the ACIP raised concerns about other individuals who may potentially benefit from a booster but are not eligible and the importance of making boosters available to them, but from a legal standpoint – I am also not a lawyer, so this is my understanding – administration of the vaccine is limited to those identified in the EUA.”
7. What is the likelihood that one shot will combine COVID and flu protection in the near future?
It is not likely, Dr. Englund said. “The reason is that the flu vaccine changes so much, and it already has four different antigens. This is assuming we keep the same method of making the flu vaccine – the answer could be different if the flu vaccine becomes an mRNA vaccine in the future.”
Companies such as Moderna and Novavax are testing single-dose shots for COVID-19 and influenza, but they are still far from having anything ready for this flu season in the United States.
8. Is there any chance a booster shot distributed now will need to be redesigned for a future variant?
“Absolutely,” Dr. Englund said. “And a booster dose is the time we may want to consider re-engineering a vaccine.”
9. Do you think the FDA/CDC limitations on who is eligible for a booster was in any way influenced by the World Health Organization call for prioritizing shots for the unvaccinated in lower-resource countries?
“This is absolutely still a global problem,” Dr. Piltch-Loeb said. “We need to get more vaccine to more countries and more people as soon as possible, because if there’s anything we’ve seen about the variants it is that ... they can come from all different places.”
“That being said, I think that it is unlikely to change the course of action in the U.S.,” she added, when it comes to comparing the global need with the domestic policy priorities of the administration.
Dr. Atmar was more direct. “No,” he said. “The WHO recommends against boosting of anyone. The U.S. decisions about boosting those in this country who are eligible are aimed toward addressing perceived needs domestically at the same time that vaccines are being provided to other countries.
“The philosophy is to address both ‘needs’ at the same time,” Dr. Atmar said.
10. What does the future hold for booster shots?
“Predicting the future is really hard, especially when it involves COVID,” Dr. del Rio said.
“Having said that, COVID is not the flu, so I doubt there will be need for annual boosters. I think the population eligible for boosters will be expanded ... and the major population not addressed at this point is the people that received either Moderna or J&J [vaccines].”
Kelly Davis contributed to this feature. A version of this article first appeared on Medscape.com.
Confusion continues to circulate in the wake of decisions on booster doses of the Pfizer/BioNTech COVID-19 vaccine, all announced within 1 week. Many people – including those now eligible and those who officially have to wait for their shot at a third dose – have questions.
Multiple agencies are involved in the booster decisions, and they have put out multiple – and sometimes conflicting – messages about booster doses, leaving more questions than answers for many people.
On Sept. 22, the Food and Drug Administration granted an emergency use authorization (EUA) for a booster dose of the Pfizer mRNA COVID-19 vaccine for those 65 and older and those at high risk for severe illness from the coronavirus, including essential workers whose jobs increase their risk for infection – such as frontline health care workers.
The Centers for Disease Control and Prevention Director Rochelle Walensky, MD, then overruled advice from the agency’s Advisory Committee on Immunization Practices (ACIP) to recommend boosters for essential workers such as those working on the front lines during the pandemic.
As it stands now, the CDC recommends that the following groups should get a third dose of the Pfizer vaccine:
- People aged 65 years and older.
- People aged 18 years and older in long-term care settings.
- People aged 50-64 years with underlying medical conditions.
The CDC also recommends that the following groups may receive a booster shot of the Pfizer vaccine, based on their individual benefits and risks:
- People aged 18-49 years with underlying medical conditions.
- People aged 18-64 years at increased risk for COVID-19 exposure and transmission because of occupational or institutional setting.
The CDC currently considers the following groups at increased risk for COVID-19:
- First responders (health care workers, firefighters, police, congregate care staff).
- Education staff (teachers, support staff, day care workers).
- Food and agriculture workers.
- Manufacturing workers.
- Corrections workers.
- U.S. Postal Service workers.
- Public transit workers.
- Grocery store workers.
Health care professionals, among the most trusted sources of COVID-19 information, are likely to encounter a number of patients wondering how all this will work.
“It’s fantastic that boosters will be available for those who the data supports need [them],” Rachael Piltch-Loeb, PhD, said during a media briefing on Sept. 23, held between the FDA and CDC decisions.
“But we’re really in a place where we have a lot more questions and answers about what the next phase of the vaccine availability and updates are going to be in the United States,” added Dr. Piltch-Loeb, preparedness fellow in the division of policy translation and leadership development and a research associate in the department of biostatistics at the Harvard T. H. Chan School of Public Health in Boston.
1. What is the biggest concern you are hearing from patients about getting a booster?
“The biggest concerns are that everyone wants it and they don’t know where to get it. In health care’s defense, the CDC just figured out what to do,” said Janet Englund, MD, professor of pediatric infectious diseases and an infectious disease and virology expert at Seattle Children’s Hospital in Washington.
“Everyone thinks they should be eligible for a booster ... people in their 50s who are not yet 65+, people with young grandchildren, etc.,” she added. “I’m at Seattle Children’s Hospital, so people are asking about booster shots and about getting their children vaccinated.”
Boosters for all COVID-19 vaccines are completely free.
“All COVID-19 vaccines, including booster doses, will be provided free of charge to the U.S. population,” the CDC has said.
2. Will patients need to prove they meet eligibility criteria for a booster shot or will it be the honor system?
“No, patients will only need to attest that they fall into one of the high-risk groups for whom a booster vaccine is authorized,” said Robert Atmar, MD, professor of infectious diseases at Baylor College of Medicine in Houston.
Dr. Piltch-Loeb agreed. “It is likely to be an honor system. It is very unlikely that there will be punishments or other ramifications ... if doses are administered, beyond the approved usage.”
3. If a patient who had the Moderna or the Johnson and Johnson vaccination requests a booster, can health care workers give them Pfizer?
The short answer is no. “This only applies to individuals who have received the Pfizer vaccine,” Dr. Piltch-Loeb said.
More data will be needed before other vaccine boosters are authorized, she added.
“My understanding is the Moderna people have just recently submitted their information, all of their data to the FDA and J&J is in line to do that very shortly,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University in Nashville, Tenn. “I would hope that within the next month to 6 weeks, we will get information about both of those vaccines,” Dr. Schaffner said.
4. When are the “mix-and-match” vaccine study results expected to come out?
“We expect that data from the study will be available in the coming weeks,” said Dr. Atmar, who is the national co-principal investigator of a mix-and-match booster trial launched in June 2021.
5. Are side effects of a booster vaccine expected to be about the same as what people experienced during their first or second immunization?
“I’m expecting the side effects will be similar to the second dose,” Dr. Englund said.
“The data presented ... at ACIP suggests that the side effects from the third shot are either the same or actually less than the first two shots,” said Carlos del Rio, MD, distinguished professor of medicine, epidemiology, and global health, and executive associate dean of Emory University School of Medicine at Grady Health System in Atlanta.
”Everyone reacts very differently to vaccines, regardless of vaccine type,” said Eric Ascher, MD, a family medicine physician at Lenox Hill Hospital in New York City. “I have had patients (as well as personal experience) where there were none to minimal symptoms, and others who felt they had a mild flu for 24 hours.”
“I expect no side effects greater than what was felt with you prior doses,” he said. “The vaccine is very safe and the benefit of vaccination outweighs the risks of any mild side effects.”
6. Is it unethical to give a booster to someone outside the approved groups if there are doses remaining at the end of the day in an open vial?
“Offering a booster shot to someone outside of approved groups if remaining doses will go to waste at the end of the day seems like a prudent decision, and relatively harmless action,” said Faith Fletcher, PhD, assistant professor at the Center for Medical Ethics and Health Policy at Baylor College of Medicine.
“However, if doses continue to fall in the laps of unapproved groups, we must evaluate the vaccine systems and structures that advantage some groups and disadvantage others,” she added. “We know that the distribution of COVID-19 vaccines has not been equitable – and some groups have been left behind.”
“I am not an ethicist and there are many competing concerns that this question addresses,” Dr. Atmar said. For example, “there is not a limitation of vaccine supply in the U.S., so that using leftover vaccine to prevent waste is no longer a major concern in the U.S.”
It could be more of a legal than ethical question, Dr. Atmar said. For an individual outside the authorized groups, legally, the FDA’s EUA for boosting does not allow the vaccine to be administered to this person, he said.
“The rationale for the restricted use in the EUA is that at this time the safety and risks associated with such administration are not known, and the benefits also have not been determined,” Dr. Atmar said. “Members of the ACIP raised concerns about other individuals who may potentially benefit from a booster but are not eligible and the importance of making boosters available to them, but from a legal standpoint – I am also not a lawyer, so this is my understanding – administration of the vaccine is limited to those identified in the EUA.”
7. What is the likelihood that one shot will combine COVID and flu protection in the near future?
It is not likely, Dr. Englund said. “The reason is that the flu vaccine changes so much, and it already has four different antigens. This is assuming we keep the same method of making the flu vaccine – the answer could be different if the flu vaccine becomes an mRNA vaccine in the future.”
Companies such as Moderna and Novavax are testing single-dose shots for COVID-19 and influenza, but they are still far from having anything ready for this flu season in the United States.
8. Is there any chance a booster shot distributed now will need to be redesigned for a future variant?
“Absolutely,” Dr. Englund said. “And a booster dose is the time we may want to consider re-engineering a vaccine.”
9. Do you think the FDA/CDC limitations on who is eligible for a booster was in any way influenced by the World Health Organization call for prioritizing shots for the unvaccinated in lower-resource countries?
“This is absolutely still a global problem,” Dr. Piltch-Loeb said. “We need to get more vaccine to more countries and more people as soon as possible, because if there’s anything we’ve seen about the variants it is that ... they can come from all different places.”
“That being said, I think that it is unlikely to change the course of action in the U.S.,” she added, when it comes to comparing the global need with the domestic policy priorities of the administration.
Dr. Atmar was more direct. “No,” he said. “The WHO recommends against boosting of anyone. The U.S. decisions about boosting those in this country who are eligible are aimed toward addressing perceived needs domestically at the same time that vaccines are being provided to other countries.
“The philosophy is to address both ‘needs’ at the same time,” Dr. Atmar said.
10. What does the future hold for booster shots?
“Predicting the future is really hard, especially when it involves COVID,” Dr. del Rio said.
“Having said that, COVID is not the flu, so I doubt there will be need for annual boosters. I think the population eligible for boosters will be expanded ... and the major population not addressed at this point is the people that received either Moderna or J&J [vaccines].”
Kelly Davis contributed to this feature. A version of this article first appeared on Medscape.com.
Confusion continues to circulate in the wake of decisions on booster doses of the Pfizer/BioNTech COVID-19 vaccine, all announced within 1 week. Many people – including those now eligible and those who officially have to wait for their shot at a third dose – have questions.
Multiple agencies are involved in the booster decisions, and they have put out multiple – and sometimes conflicting – messages about booster doses, leaving more questions than answers for many people.
On Sept. 22, the Food and Drug Administration granted an emergency use authorization (EUA) for a booster dose of the Pfizer mRNA COVID-19 vaccine for those 65 and older and those at high risk for severe illness from the coronavirus, including essential workers whose jobs increase their risk for infection – such as frontline health care workers.
The Centers for Disease Control and Prevention Director Rochelle Walensky, MD, then overruled advice from the agency’s Advisory Committee on Immunization Practices (ACIP) to recommend boosters for essential workers such as those working on the front lines during the pandemic.
As it stands now, the CDC recommends that the following groups should get a third dose of the Pfizer vaccine:
- People aged 65 years and older.
- People aged 18 years and older in long-term care settings.
- People aged 50-64 years with underlying medical conditions.
The CDC also recommends that the following groups may receive a booster shot of the Pfizer vaccine, based on their individual benefits and risks:
- People aged 18-49 years with underlying medical conditions.
- People aged 18-64 years at increased risk for COVID-19 exposure and transmission because of occupational or institutional setting.
The CDC currently considers the following groups at increased risk for COVID-19:
- First responders (health care workers, firefighters, police, congregate care staff).
- Education staff (teachers, support staff, day care workers).
- Food and agriculture workers.
- Manufacturing workers.
- Corrections workers.
- U.S. Postal Service workers.
- Public transit workers.
- Grocery store workers.
Health care professionals, among the most trusted sources of COVID-19 information, are likely to encounter a number of patients wondering how all this will work.
“It’s fantastic that boosters will be available for those who the data supports need [them],” Rachael Piltch-Loeb, PhD, said during a media briefing on Sept. 23, held between the FDA and CDC decisions.
“But we’re really in a place where we have a lot more questions and answers about what the next phase of the vaccine availability and updates are going to be in the United States,” added Dr. Piltch-Loeb, preparedness fellow in the division of policy translation and leadership development and a research associate in the department of biostatistics at the Harvard T. H. Chan School of Public Health in Boston.
1. What is the biggest concern you are hearing from patients about getting a booster?
“The biggest concerns are that everyone wants it and they don’t know where to get it. In health care’s defense, the CDC just figured out what to do,” said Janet Englund, MD, professor of pediatric infectious diseases and an infectious disease and virology expert at Seattle Children’s Hospital in Washington.
“Everyone thinks they should be eligible for a booster ... people in their 50s who are not yet 65+, people with young grandchildren, etc.,” she added. “I’m at Seattle Children’s Hospital, so people are asking about booster shots and about getting their children vaccinated.”
Boosters for all COVID-19 vaccines are completely free.
“All COVID-19 vaccines, including booster doses, will be provided free of charge to the U.S. population,” the CDC has said.
2. Will patients need to prove they meet eligibility criteria for a booster shot or will it be the honor system?
“No, patients will only need to attest that they fall into one of the high-risk groups for whom a booster vaccine is authorized,” said Robert Atmar, MD, professor of infectious diseases at Baylor College of Medicine in Houston.
Dr. Piltch-Loeb agreed. “It is likely to be an honor system. It is very unlikely that there will be punishments or other ramifications ... if doses are administered, beyond the approved usage.”
3. If a patient who had the Moderna or the Johnson and Johnson vaccination requests a booster, can health care workers give them Pfizer?
The short answer is no. “This only applies to individuals who have received the Pfizer vaccine,” Dr. Piltch-Loeb said.
More data will be needed before other vaccine boosters are authorized, she added.
“My understanding is the Moderna people have just recently submitted their information, all of their data to the FDA and J&J is in line to do that very shortly,” said William Schaffner, MD, professor of preventive medicine and infectious diseases at Vanderbilt University in Nashville, Tenn. “I would hope that within the next month to 6 weeks, we will get information about both of those vaccines,” Dr. Schaffner said.
4. When are the “mix-and-match” vaccine study results expected to come out?
“We expect that data from the study will be available in the coming weeks,” said Dr. Atmar, who is the national co-principal investigator of a mix-and-match booster trial launched in June 2021.
5. Are side effects of a booster vaccine expected to be about the same as what people experienced during their first or second immunization?
“I’m expecting the side effects will be similar to the second dose,” Dr. Englund said.
“The data presented ... at ACIP suggests that the side effects from the third shot are either the same or actually less than the first two shots,” said Carlos del Rio, MD, distinguished professor of medicine, epidemiology, and global health, and executive associate dean of Emory University School of Medicine at Grady Health System in Atlanta.
”Everyone reacts very differently to vaccines, regardless of vaccine type,” said Eric Ascher, MD, a family medicine physician at Lenox Hill Hospital in New York City. “I have had patients (as well as personal experience) where there were none to minimal symptoms, and others who felt they had a mild flu for 24 hours.”
“I expect no side effects greater than what was felt with you prior doses,” he said. “The vaccine is very safe and the benefit of vaccination outweighs the risks of any mild side effects.”
6. Is it unethical to give a booster to someone outside the approved groups if there are doses remaining at the end of the day in an open vial?
“Offering a booster shot to someone outside of approved groups if remaining doses will go to waste at the end of the day seems like a prudent decision, and relatively harmless action,” said Faith Fletcher, PhD, assistant professor at the Center for Medical Ethics and Health Policy at Baylor College of Medicine.
“However, if doses continue to fall in the laps of unapproved groups, we must evaluate the vaccine systems and structures that advantage some groups and disadvantage others,” she added. “We know that the distribution of COVID-19 vaccines has not been equitable – and some groups have been left behind.”
“I am not an ethicist and there are many competing concerns that this question addresses,” Dr. Atmar said. For example, “there is not a limitation of vaccine supply in the U.S., so that using leftover vaccine to prevent waste is no longer a major concern in the U.S.”
It could be more of a legal than ethical question, Dr. Atmar said. For an individual outside the authorized groups, legally, the FDA’s EUA for boosting does not allow the vaccine to be administered to this person, he said.
“The rationale for the restricted use in the EUA is that at this time the safety and risks associated with such administration are not known, and the benefits also have not been determined,” Dr. Atmar said. “Members of the ACIP raised concerns about other individuals who may potentially benefit from a booster but are not eligible and the importance of making boosters available to them, but from a legal standpoint – I am also not a lawyer, so this is my understanding – administration of the vaccine is limited to those identified in the EUA.”
7. What is the likelihood that one shot will combine COVID and flu protection in the near future?
It is not likely, Dr. Englund said. “The reason is that the flu vaccine changes so much, and it already has four different antigens. This is assuming we keep the same method of making the flu vaccine – the answer could be different if the flu vaccine becomes an mRNA vaccine in the future.”
Companies such as Moderna and Novavax are testing single-dose shots for COVID-19 and influenza, but they are still far from having anything ready for this flu season in the United States.
8. Is there any chance a booster shot distributed now will need to be redesigned for a future variant?
“Absolutely,” Dr. Englund said. “And a booster dose is the time we may want to consider re-engineering a vaccine.”
9. Do you think the FDA/CDC limitations on who is eligible for a booster was in any way influenced by the World Health Organization call for prioritizing shots for the unvaccinated in lower-resource countries?
“This is absolutely still a global problem,” Dr. Piltch-Loeb said. “We need to get more vaccine to more countries and more people as soon as possible, because if there’s anything we’ve seen about the variants it is that ... they can come from all different places.”
“That being said, I think that it is unlikely to change the course of action in the U.S.,” she added, when it comes to comparing the global need with the domestic policy priorities of the administration.
Dr. Atmar was more direct. “No,” he said. “The WHO recommends against boosting of anyone. The U.S. decisions about boosting those in this country who are eligible are aimed toward addressing perceived needs domestically at the same time that vaccines are being provided to other countries.
“The philosophy is to address both ‘needs’ at the same time,” Dr. Atmar said.
10. What does the future hold for booster shots?
“Predicting the future is really hard, especially when it involves COVID,” Dr. del Rio said.
“Having said that, COVID is not the flu, so I doubt there will be need for annual boosters. I think the population eligible for boosters will be expanded ... and the major population not addressed at this point is the people that received either Moderna or J&J [vaccines].”
Kelly Davis contributed to this feature. A version of this article first appeared on Medscape.com.
One in three children fall short of sleep recommendations
Just over one-third of children in the United States get less sleep than recommended, with higher rates occurring among several racial/ethnic and socioeconomic groups, according to a report from the Centers for Disease Control and Prevention.
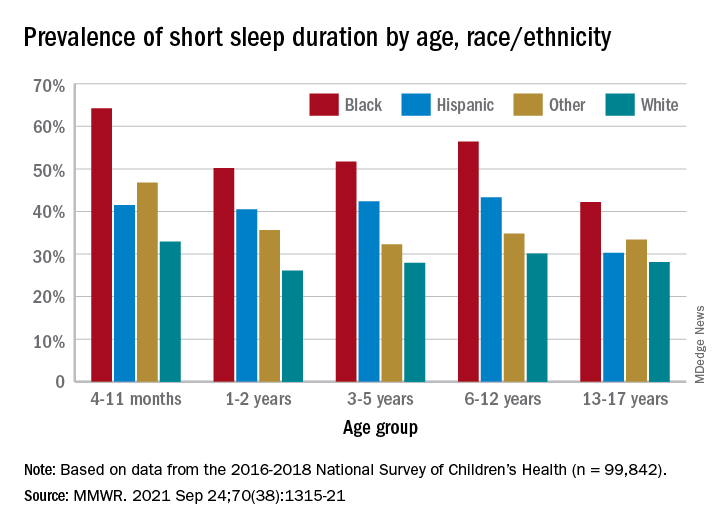
, Anne G. Wheaton, PhD, and Angelika H. Claussen, PhD, said in the Morbidity and Mortality Weekly Report.
Unlike previous reports, this analysis showed that adolescents were less likely than infants to have short sleep duration, 31.2% vs. 40.3%. These latest data are based on the 2016-2018 editions of the National Survey of Children’s Health, and the “difference might be explained by NSCH’s reliance on parent report rather than self-report with Youth Risk Behavior Surveys,” they suggested.
Black children had the highest prevalence of any group included in the study, as parents reported that 50.8% of all ages were not getting the recommended amount of sleep, compared with 39.1% among Hispanics, 34.6% for other races, and 28.8% for Whites. The figure for Black infants was 64.2%, almost double the prevalence for White infants (32.9%), said Dr. Wheaton and Dr. Claussen of the CDC.
Short sleep duration also was more common in children from lower-income families and among those with less educated parents. Geography had an effect as well, with prevalence “highest in the Southeast, similar to geographic variation in adequate sleep observed for adults,” they noted.
Previous research has shown that “sleep disparity was associated with various social determinants of health (e.g., poverty, food insecurity, and perceived racism), which can increase chronic and acute stress and result in environmental and psychological factors that negatively affect sleep duration and can compound long-term health risks,” the investigators wrote.
Short sleep duration by age group was defined as less the following amounts: Twelve hours for infants (4-11 months), 11 hours for children aged 1-2 years, 10 hours for children aged 3-5 years, 9 hours for children aged 6-12, and 8 hours for adolescents (13-17 years), they explained. Responses for the survey’s sleep-duration question totaled 99,842 for the 3 years included.
Just over one-third of children in the United States get less sleep than recommended, with higher rates occurring among several racial/ethnic and socioeconomic groups, according to a report from the Centers for Disease Control and Prevention.

, Anne G. Wheaton, PhD, and Angelika H. Claussen, PhD, said in the Morbidity and Mortality Weekly Report.
Unlike previous reports, this analysis showed that adolescents were less likely than infants to have short sleep duration, 31.2% vs. 40.3%. These latest data are based on the 2016-2018 editions of the National Survey of Children’s Health, and the “difference might be explained by NSCH’s reliance on parent report rather than self-report with Youth Risk Behavior Surveys,” they suggested.
Black children had the highest prevalence of any group included in the study, as parents reported that 50.8% of all ages were not getting the recommended amount of sleep, compared with 39.1% among Hispanics, 34.6% for other races, and 28.8% for Whites. The figure for Black infants was 64.2%, almost double the prevalence for White infants (32.9%), said Dr. Wheaton and Dr. Claussen of the CDC.
Short sleep duration also was more common in children from lower-income families and among those with less educated parents. Geography had an effect as well, with prevalence “highest in the Southeast, similar to geographic variation in adequate sleep observed for adults,” they noted.
Previous research has shown that “sleep disparity was associated with various social determinants of health (e.g., poverty, food insecurity, and perceived racism), which can increase chronic and acute stress and result in environmental and psychological factors that negatively affect sleep duration and can compound long-term health risks,” the investigators wrote.
Short sleep duration by age group was defined as less the following amounts: Twelve hours for infants (4-11 months), 11 hours for children aged 1-2 years, 10 hours for children aged 3-5 years, 9 hours for children aged 6-12, and 8 hours for adolescents (13-17 years), they explained. Responses for the survey’s sleep-duration question totaled 99,842 for the 3 years included.
Just over one-third of children in the United States get less sleep than recommended, with higher rates occurring among several racial/ethnic and socioeconomic groups, according to a report from the Centers for Disease Control and Prevention.

, Anne G. Wheaton, PhD, and Angelika H. Claussen, PhD, said in the Morbidity and Mortality Weekly Report.
Unlike previous reports, this analysis showed that adolescents were less likely than infants to have short sleep duration, 31.2% vs. 40.3%. These latest data are based on the 2016-2018 editions of the National Survey of Children’s Health, and the “difference might be explained by NSCH’s reliance on parent report rather than self-report with Youth Risk Behavior Surveys,” they suggested.
Black children had the highest prevalence of any group included in the study, as parents reported that 50.8% of all ages were not getting the recommended amount of sleep, compared with 39.1% among Hispanics, 34.6% for other races, and 28.8% for Whites. The figure for Black infants was 64.2%, almost double the prevalence for White infants (32.9%), said Dr. Wheaton and Dr. Claussen of the CDC.
Short sleep duration also was more common in children from lower-income families and among those with less educated parents. Geography had an effect as well, with prevalence “highest in the Southeast, similar to geographic variation in adequate sleep observed for adults,” they noted.
Previous research has shown that “sleep disparity was associated with various social determinants of health (e.g., poverty, food insecurity, and perceived racism), which can increase chronic and acute stress and result in environmental and psychological factors that negatively affect sleep duration and can compound long-term health risks,” the investigators wrote.
Short sleep duration by age group was defined as less the following amounts: Twelve hours for infants (4-11 months), 11 hours for children aged 1-2 years, 10 hours for children aged 3-5 years, 9 hours for children aged 6-12, and 8 hours for adolescents (13-17 years), they explained. Responses for the survey’s sleep-duration question totaled 99,842 for the 3 years included.
FROM MMWR
CDC chief overrules panel, OKs boosters for health care workers
The CDC’s Advisory Committee on Immunization Practices earlier Thursday voted to allow several groups of Americans to get a booster shot, but voted not to recommend it for adults age 18 to 64 who live or work in a place where the risk of COVID-19 is high. That would have included health care workers and other frontline employees.
But CDC Director Rochelle Walensky, MD, decided to reverse that recommendation and include the 18-to-64-year-olds in her final decision.
“As CDC Director, it is my job to recognize where our actions can have the greatest impact,” Dr. Walensky said in a statement late Thursday night, according to published reports. “At CDC, we are tasked with analyzing complex, often imperfect data to make concrete recommendations that optimize health. In a pandemic, even with uncertainty, we must take actions that we anticipate will do the greatest good.”
Dr. Walensky agreed with the rest of the advisory committee's decisions, which included recommendations that the following groups also be eligible for a booster shot:
- Adults ages 65 and up and residents of long-term care facilities
- Adults ages 50 to 64 who have an underlying medical condition that may increase their risk from a COVID infection
- Adults ages 18 to 49 who may be at increased risk from a COVID-19 infection because of an underlying medical condition, if a person feels like they need one based on a consideration of their individual benefit and risks.
About 26 million Americans are at least 6 months past the last dose of the Pfizer vaccines, making them eligible to receive a third dose. About 13.6 million of them are over the age of 65. Another 5.3 million are ages 50 to 64.
In making the recommendations, the committee left out healthcare workers. This was a departure from the Food and Drug Administration’s authorization which included boosters for those 65 and over, and for people 18 through 64 years of age who are at high risk for severe illness from the coronavirus, including essential workers – such as those in healthcare -- whose jobs increase their risk for infection.
This is the group Dr. Walensky added to the eligible list on her own.
Committee members “did not buy the need in occupational or institutional settings,” said William Schaffner, MD, an infectious disease specialist at Vanderbilt University in Nashville. Dr. Schaffner sits on the ACIP workgroup that considered the evidence behind boosters. He said that he would have voted yes to offer boosters to healthcare and other essential workers.
“There was a real split in the committee,” he said.
The vote on boosters for healthcare and other high-risk workers was rejected 9 to 6.
“I think that there is ample evidence that people such as healthcare workers do not have repeated exposure in the workplace,” said Beth Bell, MD, a clinical professor at the University of Washington. “They’re using PPE as they should and they’re following the other policies within the healthcare setting. There’s lots of evidence that suggest that health care workers who become infected become infected because of exposures in the community.”
She was not alone in feeling cautious.
“I think this is an extremely slippery slope,” said Sarah Long, MD, a pediatric infectious disease specialist at Drexel University in Philadelphia, before her vote to reject boosters for healthcare and other high-risk workers.
“We might as well just say, ‘Give it to everybody 18 and over.’ We have an extremely effective vaccine. It’s like saying it’s not working, and it is working.”
The committee saw data showing that all of the vaccines remain highly protective against hospitalization and death for all age groups, though protection against getting sick with COVID has waned slightly over time and with the dominance of the more contagious Delta variant. Those at highest risk for a severe breakthrough infection — those that cause hospitalization or death — are older adults.
How much will the U.S. benefit from boosters?
Some felt squeamish about broadly recommending boosters at all.
“We have too much hope on the line with these boosters,” said James Loehr, MD, who is a family physician in Ithaca, N.Y. Dr. Loehr said he felt the goal of giving boosters in the United States should be to decrease hospitalizations, and he felt they would, but that the impact would likely be smaller than appreciated.
Based on his calculations of the benefits of boosters for each age group, Dr. Loehr said if boosters were given to all 13 million seniors previously vaccinated with the Pfizer vaccine, we might prevent 200 hospitalizations a day, “which would be a lot,” he noted. But, he said, “considering that we have 10,000 hospitalizations a day now, it’s probably not that much.”
Others agreed.
“I really think this is a solution looking for a problem,” said Jason Goldman, MD, an associate professor at Florida Atlantic University who was representing the American College of Physicians. “You know, I don’t think it’s going to address the issue of the pandemic. I really think it’s just going to create more confusion on the provider from the position of implementation, and I really think it’s going really far afield of the data.”
ACIP Chair Grace Lee, MD, a pediatric infectious disease specialist at Stanford, said she had cared for children who had died of COVID.
“I can tell you that their family members really wished they had extra protection for their kids, because they weren’t symptomatic. Nobody else was sick at home,” she said.
Dr. Lee said for her, access was paramount, and she was in favor of expanding access to boosters for as many people as possible.
Next steps
People who were initially vaccinated with either Moderna or Johnson & Johnson vaccines are excluded from booster recommendations, something many on the committee were uncomfortable with.
The FDA is still considering Moderna’s application to market booster doses. Johnson & Johnson hasn’t yet applied to the FDA for permission to offer second doses in the United States.
While the ACIP’s recommendations are important, in this case, they may not have a huge practical effect, said Schaffner. The CDC has already approved third shots for people who are immunocompromised, and no proof of a medical condition is required to get one.
More than 2 million people have already gotten a third dose, he noted, and not all of them are immunocompromised.
“They have heard the president say that, you know, everybody should get a booster, and they’ve taken that at face value,” he said.
A version of this article first appeared on WebMD.com.
The CDC’s Advisory Committee on Immunization Practices earlier Thursday voted to allow several groups of Americans to get a booster shot, but voted not to recommend it for adults age 18 to 64 who live or work in a place where the risk of COVID-19 is high. That would have included health care workers and other frontline employees.
But CDC Director Rochelle Walensky, MD, decided to reverse that recommendation and include the 18-to-64-year-olds in her final decision.
“As CDC Director, it is my job to recognize where our actions can have the greatest impact,” Dr. Walensky said in a statement late Thursday night, according to published reports. “At CDC, we are tasked with analyzing complex, often imperfect data to make concrete recommendations that optimize health. In a pandemic, even with uncertainty, we must take actions that we anticipate will do the greatest good.”
Dr. Walensky agreed with the rest of the advisory committee's decisions, which included recommendations that the following groups also be eligible for a booster shot:
- Adults ages 65 and up and residents of long-term care facilities
- Adults ages 50 to 64 who have an underlying medical condition that may increase their risk from a COVID infection
- Adults ages 18 to 49 who may be at increased risk from a COVID-19 infection because of an underlying medical condition, if a person feels like they need one based on a consideration of their individual benefit and risks.
About 26 million Americans are at least 6 months past the last dose of the Pfizer vaccines, making them eligible to receive a third dose. About 13.6 million of them are over the age of 65. Another 5.3 million are ages 50 to 64.
In making the recommendations, the committee left out healthcare workers. This was a departure from the Food and Drug Administration’s authorization which included boosters for those 65 and over, and for people 18 through 64 years of age who are at high risk for severe illness from the coronavirus, including essential workers – such as those in healthcare -- whose jobs increase their risk for infection.
This is the group Dr. Walensky added to the eligible list on her own.
Committee members “did not buy the need in occupational or institutional settings,” said William Schaffner, MD, an infectious disease specialist at Vanderbilt University in Nashville. Dr. Schaffner sits on the ACIP workgroup that considered the evidence behind boosters. He said that he would have voted yes to offer boosters to healthcare and other essential workers.
“There was a real split in the committee,” he said.
The vote on boosters for healthcare and other high-risk workers was rejected 9 to 6.
“I think that there is ample evidence that people such as healthcare workers do not have repeated exposure in the workplace,” said Beth Bell, MD, a clinical professor at the University of Washington. “They’re using PPE as they should and they’re following the other policies within the healthcare setting. There’s lots of evidence that suggest that health care workers who become infected become infected because of exposures in the community.”
She was not alone in feeling cautious.
“I think this is an extremely slippery slope,” said Sarah Long, MD, a pediatric infectious disease specialist at Drexel University in Philadelphia, before her vote to reject boosters for healthcare and other high-risk workers.
“We might as well just say, ‘Give it to everybody 18 and over.’ We have an extremely effective vaccine. It’s like saying it’s not working, and it is working.”
The committee saw data showing that all of the vaccines remain highly protective against hospitalization and death for all age groups, though protection against getting sick with COVID has waned slightly over time and with the dominance of the more contagious Delta variant. Those at highest risk for a severe breakthrough infection — those that cause hospitalization or death — are older adults.
How much will the U.S. benefit from boosters?
Some felt squeamish about broadly recommending boosters at all.
“We have too much hope on the line with these boosters,” said James Loehr, MD, who is a family physician in Ithaca, N.Y. Dr. Loehr said he felt the goal of giving boosters in the United States should be to decrease hospitalizations, and he felt they would, but that the impact would likely be smaller than appreciated.
Based on his calculations of the benefits of boosters for each age group, Dr. Loehr said if boosters were given to all 13 million seniors previously vaccinated with the Pfizer vaccine, we might prevent 200 hospitalizations a day, “which would be a lot,” he noted. But, he said, “considering that we have 10,000 hospitalizations a day now, it’s probably not that much.”
Others agreed.
“I really think this is a solution looking for a problem,” said Jason Goldman, MD, an associate professor at Florida Atlantic University who was representing the American College of Physicians. “You know, I don’t think it’s going to address the issue of the pandemic. I really think it’s just going to create more confusion on the provider from the position of implementation, and I really think it’s going really far afield of the data.”
ACIP Chair Grace Lee, MD, a pediatric infectious disease specialist at Stanford, said she had cared for children who had died of COVID.
“I can tell you that their family members really wished they had extra protection for their kids, because they weren’t symptomatic. Nobody else was sick at home,” she said.
Dr. Lee said for her, access was paramount, and she was in favor of expanding access to boosters for as many people as possible.
Next steps
People who were initially vaccinated with either Moderna or Johnson & Johnson vaccines are excluded from booster recommendations, something many on the committee were uncomfortable with.
The FDA is still considering Moderna’s application to market booster doses. Johnson & Johnson hasn’t yet applied to the FDA for permission to offer second doses in the United States.
While the ACIP’s recommendations are important, in this case, they may not have a huge practical effect, said Schaffner. The CDC has already approved third shots for people who are immunocompromised, and no proof of a medical condition is required to get one.
More than 2 million people have already gotten a third dose, he noted, and not all of them are immunocompromised.
“They have heard the president say that, you know, everybody should get a booster, and they’ve taken that at face value,” he said.
A version of this article first appeared on WebMD.com.
The CDC’s Advisory Committee on Immunization Practices earlier Thursday voted to allow several groups of Americans to get a booster shot, but voted not to recommend it for adults age 18 to 64 who live or work in a place where the risk of COVID-19 is high. That would have included health care workers and other frontline employees.
But CDC Director Rochelle Walensky, MD, decided to reverse that recommendation and include the 18-to-64-year-olds in her final decision.
“As CDC Director, it is my job to recognize where our actions can have the greatest impact,” Dr. Walensky said in a statement late Thursday night, according to published reports. “At CDC, we are tasked with analyzing complex, often imperfect data to make concrete recommendations that optimize health. In a pandemic, even with uncertainty, we must take actions that we anticipate will do the greatest good.”
Dr. Walensky agreed with the rest of the advisory committee's decisions, which included recommendations that the following groups also be eligible for a booster shot:
- Adults ages 65 and up and residents of long-term care facilities
- Adults ages 50 to 64 who have an underlying medical condition that may increase their risk from a COVID infection
- Adults ages 18 to 49 who may be at increased risk from a COVID-19 infection because of an underlying medical condition, if a person feels like they need one based on a consideration of their individual benefit and risks.
About 26 million Americans are at least 6 months past the last dose of the Pfizer vaccines, making them eligible to receive a third dose. About 13.6 million of them are over the age of 65. Another 5.3 million are ages 50 to 64.
In making the recommendations, the committee left out healthcare workers. This was a departure from the Food and Drug Administration’s authorization which included boosters for those 65 and over, and for people 18 through 64 years of age who are at high risk for severe illness from the coronavirus, including essential workers – such as those in healthcare -- whose jobs increase their risk for infection.
This is the group Dr. Walensky added to the eligible list on her own.
Committee members “did not buy the need in occupational or institutional settings,” said William Schaffner, MD, an infectious disease specialist at Vanderbilt University in Nashville. Dr. Schaffner sits on the ACIP workgroup that considered the evidence behind boosters. He said that he would have voted yes to offer boosters to healthcare and other essential workers.
“There was a real split in the committee,” he said.
The vote on boosters for healthcare and other high-risk workers was rejected 9 to 6.
“I think that there is ample evidence that people such as healthcare workers do not have repeated exposure in the workplace,” said Beth Bell, MD, a clinical professor at the University of Washington. “They’re using PPE as they should and they’re following the other policies within the healthcare setting. There’s lots of evidence that suggest that health care workers who become infected become infected because of exposures in the community.”
She was not alone in feeling cautious.
“I think this is an extremely slippery slope,” said Sarah Long, MD, a pediatric infectious disease specialist at Drexel University in Philadelphia, before her vote to reject boosters for healthcare and other high-risk workers.
“We might as well just say, ‘Give it to everybody 18 and over.’ We have an extremely effective vaccine. It’s like saying it’s not working, and it is working.”
The committee saw data showing that all of the vaccines remain highly protective against hospitalization and death for all age groups, though protection against getting sick with COVID has waned slightly over time and with the dominance of the more contagious Delta variant. Those at highest risk for a severe breakthrough infection — those that cause hospitalization or death — are older adults.
How much will the U.S. benefit from boosters?
Some felt squeamish about broadly recommending boosters at all.
“We have too much hope on the line with these boosters,” said James Loehr, MD, who is a family physician in Ithaca, N.Y. Dr. Loehr said he felt the goal of giving boosters in the United States should be to decrease hospitalizations, and he felt they would, but that the impact would likely be smaller than appreciated.
Based on his calculations of the benefits of boosters for each age group, Dr. Loehr said if boosters were given to all 13 million seniors previously vaccinated with the Pfizer vaccine, we might prevent 200 hospitalizations a day, “which would be a lot,” he noted. But, he said, “considering that we have 10,000 hospitalizations a day now, it’s probably not that much.”
Others agreed.
“I really think this is a solution looking for a problem,” said Jason Goldman, MD, an associate professor at Florida Atlantic University who was representing the American College of Physicians. “You know, I don’t think it’s going to address the issue of the pandemic. I really think it’s just going to create more confusion on the provider from the position of implementation, and I really think it’s going really far afield of the data.”
ACIP Chair Grace Lee, MD, a pediatric infectious disease specialist at Stanford, said she had cared for children who had died of COVID.
“I can tell you that their family members really wished they had extra protection for their kids, because they weren’t symptomatic. Nobody else was sick at home,” she said.
Dr. Lee said for her, access was paramount, and she was in favor of expanding access to boosters for as many people as possible.
Next steps
People who were initially vaccinated with either Moderna or Johnson & Johnson vaccines are excluded from booster recommendations, something many on the committee were uncomfortable with.
The FDA is still considering Moderna’s application to market booster doses. Johnson & Johnson hasn’t yet applied to the FDA for permission to offer second doses in the United States.
While the ACIP’s recommendations are important, in this case, they may not have a huge practical effect, said Schaffner. The CDC has already approved third shots for people who are immunocompromised, and no proof of a medical condition is required to get one.
More than 2 million people have already gotten a third dose, he noted, and not all of them are immunocompromised.
“They have heard the president say that, you know, everybody should get a booster, and they’ve taken that at face value,” he said.
A version of this article first appeared on WebMD.com.
Dopamine and reward: The story of social media
How often do you find yourself on social media? The first thing I do when I wake up is check my email and text messages, as well as my Facebook, Snapchat, and Instagram notifications.
Some 150,000 messages are shared on Facebook each minute; 293 million daily active users worldwide were recorded on Snapchat during the second quarter of 2021; 127.2 million monthly active users in the United States are projected to be on Instagram by 2023.
Social media has gained the hearts and wonder of many around the world. It’s absolutely incredible how ingrained it has become in our lives as a medium for creativity, outlet for communication, and platform for information. In fact, these online network tools have now become essential during COVID-19 to ensure productive workflow, keep in touch with our loved ones, and, overall, maintain social capital. Social media has truly emerged as a powerful form of living beyond our physical selves.
Yet, increased (and addictive) social media use is associated with negative health outcomes, especially among adolescents. For example, in a study reporting parent and adolescent accounts of social media use, it was reported that social media use was associated with hyperactivity/impulsivity, depression, anxiety, loneliness, and a fear of missing out. Furthermore, a meta-analysis investigating the relationship between social media use and depressive symptoms among adolescents found a small but significant and positive relationship between the two. However, additional research is required to elucidate this association.
Notwithstanding, the addictive nature of social media has previously been called out as analogous to the addictive nature of gambling. Let’s think about it. Whether you’re on Instagram, TikTok, or a similar platform, you can’t help but scroll from one video to the next. It’s one 5- to 10-second video after the next, and before you know it, you’ve spent the past hour going through random videos – but you can’t stop. Why is that so?
Social media actually “rewires” our brain such that we expect instant gratification. In other words, when we get a notification, message, like, or share, we expect fast and short-term pleasure/reward because the brain will produce a “hit of dopamine.” However, it is important to note that the reward system is not delimited to the dopaminergic pathway and, in fact, should be understood as a complex network system (i.e., governed by changes in brain morphology through addiction and excessive behavior). Given the quick pace of the social media world, the reward pathways in our brain change and there’s an increasing demand for attention, perpetuating an addictive mindset.
When we refresh our page, we expect instant gratification. But what happens when we don’t get a like, or a message, or some sort of “reward”? Recounts of social media use by adolescents have likened online attention to popularity. Accordingly, a lack of constant attention on social media has created a vicious cycle of anxiety, loneliness, and depression because of a failure to receive “virtual” reward. Taken together, social media may be harmful because it distorts our self-image, and while social media platforms help connect us, they can also ironically make us feel isolated, lower our self-confidence, and diminish our overall sense of well-being.
As the platforms for communication and information have evolved so rapidly over the past decade, there is a need to establish boundaries between what is beneficial and what is potentially detrimental to our mental health. While social media companies should play a role in mitigating addictive social network behavior, it would also seem counterintuitive to the general business model. In that case, who takes charge? This multifaceted problem requires a multidisciplinary approach.
Leanna M.W. Lui is an MSc candidate at the University of Toronto.
A version of this article first appeared on Medscape.com.
How often do you find yourself on social media? The first thing I do when I wake up is check my email and text messages, as well as my Facebook, Snapchat, and Instagram notifications.
Some 150,000 messages are shared on Facebook each minute; 293 million daily active users worldwide were recorded on Snapchat during the second quarter of 2021; 127.2 million monthly active users in the United States are projected to be on Instagram by 2023.
Social media has gained the hearts and wonder of many around the world. It’s absolutely incredible how ingrained it has become in our lives as a medium for creativity, outlet for communication, and platform for information. In fact, these online network tools have now become essential during COVID-19 to ensure productive workflow, keep in touch with our loved ones, and, overall, maintain social capital. Social media has truly emerged as a powerful form of living beyond our physical selves.
Yet, increased (and addictive) social media use is associated with negative health outcomes, especially among adolescents. For example, in a study reporting parent and adolescent accounts of social media use, it was reported that social media use was associated with hyperactivity/impulsivity, depression, anxiety, loneliness, and a fear of missing out. Furthermore, a meta-analysis investigating the relationship between social media use and depressive symptoms among adolescents found a small but significant and positive relationship between the two. However, additional research is required to elucidate this association.
Notwithstanding, the addictive nature of social media has previously been called out as analogous to the addictive nature of gambling. Let’s think about it. Whether you’re on Instagram, TikTok, or a similar platform, you can’t help but scroll from one video to the next. It’s one 5- to 10-second video after the next, and before you know it, you’ve spent the past hour going through random videos – but you can’t stop. Why is that so?
Social media actually “rewires” our brain such that we expect instant gratification. In other words, when we get a notification, message, like, or share, we expect fast and short-term pleasure/reward because the brain will produce a “hit of dopamine.” However, it is important to note that the reward system is not delimited to the dopaminergic pathway and, in fact, should be understood as a complex network system (i.e., governed by changes in brain morphology through addiction and excessive behavior). Given the quick pace of the social media world, the reward pathways in our brain change and there’s an increasing demand for attention, perpetuating an addictive mindset.
When we refresh our page, we expect instant gratification. But what happens when we don’t get a like, or a message, or some sort of “reward”? Recounts of social media use by adolescents have likened online attention to popularity. Accordingly, a lack of constant attention on social media has created a vicious cycle of anxiety, loneliness, and depression because of a failure to receive “virtual” reward. Taken together, social media may be harmful because it distorts our self-image, and while social media platforms help connect us, they can also ironically make us feel isolated, lower our self-confidence, and diminish our overall sense of well-being.
As the platforms for communication and information have evolved so rapidly over the past decade, there is a need to establish boundaries between what is beneficial and what is potentially detrimental to our mental health. While social media companies should play a role in mitigating addictive social network behavior, it would also seem counterintuitive to the general business model. In that case, who takes charge? This multifaceted problem requires a multidisciplinary approach.
Leanna M.W. Lui is an MSc candidate at the University of Toronto.
A version of this article first appeared on Medscape.com.
How often do you find yourself on social media? The first thing I do when I wake up is check my email and text messages, as well as my Facebook, Snapchat, and Instagram notifications.
Some 150,000 messages are shared on Facebook each minute; 293 million daily active users worldwide were recorded on Snapchat during the second quarter of 2021; 127.2 million monthly active users in the United States are projected to be on Instagram by 2023.
Social media has gained the hearts and wonder of many around the world. It’s absolutely incredible how ingrained it has become in our lives as a medium for creativity, outlet for communication, and platform for information. In fact, these online network tools have now become essential during COVID-19 to ensure productive workflow, keep in touch with our loved ones, and, overall, maintain social capital. Social media has truly emerged as a powerful form of living beyond our physical selves.
Yet, increased (and addictive) social media use is associated with negative health outcomes, especially among adolescents. For example, in a study reporting parent and adolescent accounts of social media use, it was reported that social media use was associated with hyperactivity/impulsivity, depression, anxiety, loneliness, and a fear of missing out. Furthermore, a meta-analysis investigating the relationship between social media use and depressive symptoms among adolescents found a small but significant and positive relationship between the two. However, additional research is required to elucidate this association.
Notwithstanding, the addictive nature of social media has previously been called out as analogous to the addictive nature of gambling. Let’s think about it. Whether you’re on Instagram, TikTok, or a similar platform, you can’t help but scroll from one video to the next. It’s one 5- to 10-second video after the next, and before you know it, you’ve spent the past hour going through random videos – but you can’t stop. Why is that so?
Social media actually “rewires” our brain such that we expect instant gratification. In other words, when we get a notification, message, like, or share, we expect fast and short-term pleasure/reward because the brain will produce a “hit of dopamine.” However, it is important to note that the reward system is not delimited to the dopaminergic pathway and, in fact, should be understood as a complex network system (i.e., governed by changes in brain morphology through addiction and excessive behavior). Given the quick pace of the social media world, the reward pathways in our brain change and there’s an increasing demand for attention, perpetuating an addictive mindset.
When we refresh our page, we expect instant gratification. But what happens when we don’t get a like, or a message, or some sort of “reward”? Recounts of social media use by adolescents have likened online attention to popularity. Accordingly, a lack of constant attention on social media has created a vicious cycle of anxiety, loneliness, and depression because of a failure to receive “virtual” reward. Taken together, social media may be harmful because it distorts our self-image, and while social media platforms help connect us, they can also ironically make us feel isolated, lower our self-confidence, and diminish our overall sense of well-being.
As the platforms for communication and information have evolved so rapidly over the past decade, there is a need to establish boundaries between what is beneficial and what is potentially detrimental to our mental health. While social media companies should play a role in mitigating addictive social network behavior, it would also seem counterintuitive to the general business model. In that case, who takes charge? This multifaceted problem requires a multidisciplinary approach.
Leanna M.W. Lui is an MSc candidate at the University of Toronto.
A version of this article first appeared on Medscape.com.
Study identifies pandemic-related stressor in Parkinson’s disease
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
a team of researchers in the Netherlands reported, but they also identified meaningful targets for intervention.
Lisanne Dommershuijsen, MSc, a PhD candidate and researcher in epidemiology at the Erasmus University Medical Center in Rotterdam, the Netherlands, reported on a cross-sectional study of 833 participants with Parkinson’s disease in the PRIME-NL study at the International Congress of Parkinson’s Disease and Movement Disorders. The average age of participants was 70.2 and 38% were women.
“We studied targeted hypothetical interventions on COVID-19 stressors in people with Parkinson’s disease,” Ms. Dommershuijsen said. “This disruption in normal life caused considerable psychological stress in community-dwelling individuals. People with Parkinson’s disease might be especially vulnerable to this stress.
“For instance, because reduced levels of physical activity have worsened symptoms or because people with Parkinson’s often have difficulty with flexible [adaptations] to drastic and rapid changes in daily routines, such as those introduced by the COVID-19 pandemic, previous studies found that COVID-19 worsened depression and anxiety symptoms and reduced quality of life (QOL) in people with Parkinson’s disease,” Ms. Dommershuijsen said.
Hence, the goal of the study was to identify the most vulnerable subgroups in the Parkinson’s population and to suggest potential interventions to ameliorate these impacts, she said.
The study focused on eight different stressors that emerged in the pandemic: access to care, medicine and nursing services; loss of social contact; canceled social events; tension or conflict in the home; inability to perform physical activity or relax; and COVID-19 symptoms. The outcomes of interest were depression, as measured with the Beck Depression Inventory (BDI); anxiety, as measured with the Spielberger State-Trait Anxiety Inventory (STAI); and QOL, with the Parkinson’s Disease Quality of Life Questionnaire. The aggregate resulted in a scale of 0-40, with the mean stressor score in the study being 9.6, Ms. Dommershuijsen said.
The BDI and STAI scores for social stressors – loss of social contacts, social events canceled and tension or conflict at home – exceeded those for the so-called care stressors – problems accessing care, medication or nursing – she said, although all eight stressors yielded higher BDI and STAI scores across the board.
Vulnerable subgroups
“When we looked at vulnerable subgroups of people with Parkinson’s disease, we found more pronounced associations between the COVID-19 stress and mental health in women, in highly educated participants, and in participants with advanced Parkinson’s disease,” Ms. Dommershuijsen said. The impact on women and people with advanced disease is explainable, Ms. Dommershuijsen added in an interview; the former because depressive symptoms are more common in women, and the latter because loss of access to care impacts mental wellness.
“The finding that social stressors were more related to anxiety in highly educated people was surprising to us, given that depression in general is more common in people with a lower education,” she said in an interview. “One previous study of the general population suggested this might be related to expectations about available resources, but this findings and the possible explanation warrants further investigation.”
When the study stratified for coping strategies, the COVID-19 stressors had a smaller effect on depressive and anxiety symptoms in Parkinson’s disease patients prone to confrontive coping and planful problem solving, she said. “Whereas, we observed a larger effect of these stressors in people who are prone to using distancing or seeking social support as coping mechanisms,” Ms. Dommershuijsen said.
The researchers also created a model of a hypothetical 50% reduction in COVID-19 stressors among all study participants, but the effect wasn’t clinically relevant, Ms. Dommershuijsen said. However, in people with advanced Parkinson’s disease – that is, with an Movement Disorder Society–Unified Parkinson Disease Rating Scale score above median – the effect was clinically relevant in all outcomes.
The potential interventions the study identified were telemedicine via virtual consultations to alleviate care stressors, and virtual support groups and online classes to address social stressors. “However, a more personalized approach is needed to target tension or conflict at home, which was the most important social stressor influencing depression and anxiety symptoms in our study,” she said. “Social work can play an important role here.”
Asked to comment on the study, Roy Alcalay, MD, professor of neurology at Columbia University Irving Medical Center in New York, said in an interview that the findings align with his research on the impact of COVID-19 and related restrictions on people with Parkinson’s disease.
“The pandemic has affected people in different ways,” he said. “Initially very acutely, people just didn’t have access to doctors. There was also the acute question in movement disorders, but also in other diseases where the people with Parkinson’s disease are going to have the worse outcome when they have COVID-19.” Dr. Alcalay authored two recent papers on the impact of COVID-19 in people with Parkinson’s disease.
“Then we see that, in addition to that question, there’s the question of even if they don’t have COVID-19, just the social distancing and the lack of access to health care, and specifically to physical and occupational therapy and other services, can be quite damaging,” he said.
What’s commendable about the study, he said, was that it just doesn’t highlight the problem. “They’re also highlighting potential solutions, that planful problem solving and coping strategies can be helpful to people.”
Neither Ms. Dommershuijsen nor Dr. Alcalay have any relevant relationships to disclose.
FROM MDS VIRTUAL CONGRESS 2021
New COVID-19 strain has reached the U.S.
Deadline, citing a Centers for Disease Control and Prevention report, said 26 residents and 20 workers tested positive for COVID-19 at a skilled care nursing home. The facility has 83 residents and 116 employees.
On March 1, 28 specimens that had been subjected to whole genome sequencing were found to have “mutations aligning with the R.1 lineage,” Deadline said.
About 90% of the facility’s residents and 52% of the staff had received two COVID vaccine doses, the CDC said. Because of the high vaccination rate, the finding raises concerns about “reduced protective immunity” in relation to the R.1 variant, the CDC said.
However, the nursing home case appears to show that the vaccine keeps most people from getting extremely sick, the CDC said. The vaccine was 86.5% protective against symptomatic illness among residents and 87.1% protective for employees.
“Compared with unvaccinated persons, vaccinated persons had reduced risk for SARS-CoV-2 infection and symptomatic COVID-19,” the CDC said. The vaccination of nursing home residents and health care workers “is essential to reduce the risk for symptomatic COVID-19, as is continued focus on infection prevention and control practices,” the CDC said.
Since being reported in Kentucky, R.1 has been detected more than 10,000 times in the United States, Forbes reported, basing that number on entries in the GISAID SARS-CoV-2 database.
Overall, more than 42 million cases of COVID have been reported since the start of the pandemic.
Deadline reported that the R.1 strain was first detected in Japan in January among three members of one family. The family members had no history of traveling abroad, Deadline said, citing an National Institutes of Health report.
The CDC has not classified R.1 as a variant of concern yet but noted it has “several mutations of importance” and “demonstrates evidence of increasing virus transmissibility.”
A version of this article first appeared on WebMD.com.
Deadline, citing a Centers for Disease Control and Prevention report, said 26 residents and 20 workers tested positive for COVID-19 at a skilled care nursing home. The facility has 83 residents and 116 employees.
On March 1, 28 specimens that had been subjected to whole genome sequencing were found to have “mutations aligning with the R.1 lineage,” Deadline said.
About 90% of the facility’s residents and 52% of the staff had received two COVID vaccine doses, the CDC said. Because of the high vaccination rate, the finding raises concerns about “reduced protective immunity” in relation to the R.1 variant, the CDC said.
However, the nursing home case appears to show that the vaccine keeps most people from getting extremely sick, the CDC said. The vaccine was 86.5% protective against symptomatic illness among residents and 87.1% protective for employees.
“Compared with unvaccinated persons, vaccinated persons had reduced risk for SARS-CoV-2 infection and symptomatic COVID-19,” the CDC said. The vaccination of nursing home residents and health care workers “is essential to reduce the risk for symptomatic COVID-19, as is continued focus on infection prevention and control practices,” the CDC said.
Since being reported in Kentucky, R.1 has been detected more than 10,000 times in the United States, Forbes reported, basing that number on entries in the GISAID SARS-CoV-2 database.
Overall, more than 42 million cases of COVID have been reported since the start of the pandemic.
Deadline reported that the R.1 strain was first detected in Japan in January among three members of one family. The family members had no history of traveling abroad, Deadline said, citing an National Institutes of Health report.
The CDC has not classified R.1 as a variant of concern yet but noted it has “several mutations of importance” and “demonstrates evidence of increasing virus transmissibility.”
A version of this article first appeared on WebMD.com.
Deadline, citing a Centers for Disease Control and Prevention report, said 26 residents and 20 workers tested positive for COVID-19 at a skilled care nursing home. The facility has 83 residents and 116 employees.
On March 1, 28 specimens that had been subjected to whole genome sequencing were found to have “mutations aligning with the R.1 lineage,” Deadline said.
About 90% of the facility’s residents and 52% of the staff had received two COVID vaccine doses, the CDC said. Because of the high vaccination rate, the finding raises concerns about “reduced protective immunity” in relation to the R.1 variant, the CDC said.
However, the nursing home case appears to show that the vaccine keeps most people from getting extremely sick, the CDC said. The vaccine was 86.5% protective against symptomatic illness among residents and 87.1% protective for employees.
“Compared with unvaccinated persons, vaccinated persons had reduced risk for SARS-CoV-2 infection and symptomatic COVID-19,” the CDC said. The vaccination of nursing home residents and health care workers “is essential to reduce the risk for symptomatic COVID-19, as is continued focus on infection prevention and control practices,” the CDC said.
Since being reported in Kentucky, R.1 has been detected more than 10,000 times in the United States, Forbes reported, basing that number on entries in the GISAID SARS-CoV-2 database.
Overall, more than 42 million cases of COVID have been reported since the start of the pandemic.
Deadline reported that the R.1 strain was first detected in Japan in January among three members of one family. The family members had no history of traveling abroad, Deadline said, citing an National Institutes of Health report.
The CDC has not classified R.1 as a variant of concern yet but noted it has “several mutations of importance” and “demonstrates evidence of increasing virus transmissibility.”
A version of this article first appeared on WebMD.com.
Sexual assault in women tied to increased stroke, dementia risk
Traumatic experiences, especially sexual assault, may put women at greater risk for poor brain health.
In the Ms Brain study, middle-aged women with trauma exposure had a greater volume of white matter hyperintensities (WMHs) than those without trauma. In addition, the differences persisted even after adjusting for depressive or post-traumatic stress symptoms.
WMHs are “an important indicator of small vessel disease in the brain and have been linked to future stroke risk, dementia risk, and mortality,” lead investigator Rebecca Thurston, PhD, from the University of Pittsburgh, told this news organization.
“What I take from this is, really, that sexual assault has implications for women’s health, far beyond exclusively mental health outcomes, but also for their cardiovascular health, as we have shown in other work and for their stroke and dementia risk as we are seeing in the present work,” Dr. Thurston added.
The study was presented at the North American Menopause Society (NAMS) Annual Meeting in Washington, D.C., and has been accepted for publication in the journal Brain Imaging and Behavior.
Beyond the usual suspects
As part of the study, 145 women (mean age, 59 years) free of clinical cardiovascular disease, stroke, or dementia provided their medical history, including history of traumatic experiences, depression, and post-traumatic stress disorder and underwent magnetic resonance brain imaging for WMHs.
More than two-thirds (68%) of the women reported at least one trauma, most commonly sexual assault (23%).
In multivariate analysis, women with trauma exposure had greater WMH volume than women without trauma (P = .01), with sexual assault most strongly associated with greater WMH volume (P = .02).
The associations persisted after adjusting for depressive or post-traumatic stress symptoms.
“A history of sexual assault was particularly related to white matter hyperintensities in the parietal lobe, and these kinds of white matter hyperintensities have been linked to Alzheimer’s disease in a fairly pronounced way,” Dr. Thurston said.
“When we think about risk factors for stroke, dementia, we need to think beyond exclusively our usual suspects and also think about women [who experienced] psychological trauma and experienced sexual assault in particular. So ask about it and consider it part of your screening regimen,” she added.
‘Burgeoning’ literature
Commenting on the findings, Charles Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, and director of its Institute for Early Life Adversity Research, said the research adds to the “burgeoning literature on the long term neurobiological consequences of trauma and more specifically, sexual abuse, on brain imaging measures.”
“Our group and others reported several years ago that patients with mood disorders, more specifically bipolar disorder and major depression, had higher rates of WMH than matched controls. Those older studies did not control for a history of early life adversity such as childhood maltreatment,” Dr. Nemeroff said.
“In addition to this finding of increased WMH in subjects exposed to trauma is a very large literature documenting other central nervous system (CNS) changes in this population, including cortical thinning in certain brain areas and clearly an emerging finding that different forms of childhood maltreatment are associated with quite distinct structural brain alterations in adulthood,” he noted.
The study was supported by grants from the National Institutes of Health. Dr. Thurston and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Traumatic experiences, especially sexual assault, may put women at greater risk for poor brain health.
In the Ms Brain study, middle-aged women with trauma exposure had a greater volume of white matter hyperintensities (WMHs) than those without trauma. In addition, the differences persisted even after adjusting for depressive or post-traumatic stress symptoms.
WMHs are “an important indicator of small vessel disease in the brain and have been linked to future stroke risk, dementia risk, and mortality,” lead investigator Rebecca Thurston, PhD, from the University of Pittsburgh, told this news organization.
“What I take from this is, really, that sexual assault has implications for women’s health, far beyond exclusively mental health outcomes, but also for their cardiovascular health, as we have shown in other work and for their stroke and dementia risk as we are seeing in the present work,” Dr. Thurston added.
The study was presented at the North American Menopause Society (NAMS) Annual Meeting in Washington, D.C., and has been accepted for publication in the journal Brain Imaging and Behavior.
Beyond the usual suspects
As part of the study, 145 women (mean age, 59 years) free of clinical cardiovascular disease, stroke, or dementia provided their medical history, including history of traumatic experiences, depression, and post-traumatic stress disorder and underwent magnetic resonance brain imaging for WMHs.
More than two-thirds (68%) of the women reported at least one trauma, most commonly sexual assault (23%).
In multivariate analysis, women with trauma exposure had greater WMH volume than women without trauma (P = .01), with sexual assault most strongly associated with greater WMH volume (P = .02).
The associations persisted after adjusting for depressive or post-traumatic stress symptoms.
“A history of sexual assault was particularly related to white matter hyperintensities in the parietal lobe, and these kinds of white matter hyperintensities have been linked to Alzheimer’s disease in a fairly pronounced way,” Dr. Thurston said.
“When we think about risk factors for stroke, dementia, we need to think beyond exclusively our usual suspects and also think about women [who experienced] psychological trauma and experienced sexual assault in particular. So ask about it and consider it part of your screening regimen,” she added.
‘Burgeoning’ literature
Commenting on the findings, Charles Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, and director of its Institute for Early Life Adversity Research, said the research adds to the “burgeoning literature on the long term neurobiological consequences of trauma and more specifically, sexual abuse, on brain imaging measures.”
“Our group and others reported several years ago that patients with mood disorders, more specifically bipolar disorder and major depression, had higher rates of WMH than matched controls. Those older studies did not control for a history of early life adversity such as childhood maltreatment,” Dr. Nemeroff said.
“In addition to this finding of increased WMH in subjects exposed to trauma is a very large literature documenting other central nervous system (CNS) changes in this population, including cortical thinning in certain brain areas and clearly an emerging finding that different forms of childhood maltreatment are associated with quite distinct structural brain alterations in adulthood,” he noted.
The study was supported by grants from the National Institutes of Health. Dr. Thurston and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Traumatic experiences, especially sexual assault, may put women at greater risk for poor brain health.
In the Ms Brain study, middle-aged women with trauma exposure had a greater volume of white matter hyperintensities (WMHs) than those without trauma. In addition, the differences persisted even after adjusting for depressive or post-traumatic stress symptoms.
WMHs are “an important indicator of small vessel disease in the brain and have been linked to future stroke risk, dementia risk, and mortality,” lead investigator Rebecca Thurston, PhD, from the University of Pittsburgh, told this news organization.
“What I take from this is, really, that sexual assault has implications for women’s health, far beyond exclusively mental health outcomes, but also for their cardiovascular health, as we have shown in other work and for their stroke and dementia risk as we are seeing in the present work,” Dr. Thurston added.
The study was presented at the North American Menopause Society (NAMS) Annual Meeting in Washington, D.C., and has been accepted for publication in the journal Brain Imaging and Behavior.
Beyond the usual suspects
As part of the study, 145 women (mean age, 59 years) free of clinical cardiovascular disease, stroke, or dementia provided their medical history, including history of traumatic experiences, depression, and post-traumatic stress disorder and underwent magnetic resonance brain imaging for WMHs.
More than two-thirds (68%) of the women reported at least one trauma, most commonly sexual assault (23%).
In multivariate analysis, women with trauma exposure had greater WMH volume than women without trauma (P = .01), with sexual assault most strongly associated with greater WMH volume (P = .02).
The associations persisted after adjusting for depressive or post-traumatic stress symptoms.
“A history of sexual assault was particularly related to white matter hyperintensities in the parietal lobe, and these kinds of white matter hyperintensities have been linked to Alzheimer’s disease in a fairly pronounced way,” Dr. Thurston said.
“When we think about risk factors for stroke, dementia, we need to think beyond exclusively our usual suspects and also think about women [who experienced] psychological trauma and experienced sexual assault in particular. So ask about it and consider it part of your screening regimen,” she added.
‘Burgeoning’ literature
Commenting on the findings, Charles Nemeroff, MD, PhD, professor and chair, department of psychiatry and behavioral sciences, Dell Medical School, University of Texas at Austin, and director of its Institute for Early Life Adversity Research, said the research adds to the “burgeoning literature on the long term neurobiological consequences of trauma and more specifically, sexual abuse, on brain imaging measures.”
“Our group and others reported several years ago that patients with mood disorders, more specifically bipolar disorder and major depression, had higher rates of WMH than matched controls. Those older studies did not control for a history of early life adversity such as childhood maltreatment,” Dr. Nemeroff said.
“In addition to this finding of increased WMH in subjects exposed to trauma is a very large literature documenting other central nervous system (CNS) changes in this population, including cortical thinning in certain brain areas and clearly an emerging finding that different forms of childhood maltreatment are associated with quite distinct structural brain alterations in adulthood,” he noted.
The study was supported by grants from the National Institutes of Health. Dr. Thurston and Dr. Nemeroff have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pandemic restrictions ignite innovative pivot for psychiatry
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
As medical school faculty members – and our students – know well, the COVID-19 pandemic forced us to become creative and shift much of our curricula online. Many hospitals chose to limit medical student rotations because of safety concerns. Students fell victim to canceled psychiatry rotations and electives during the pandemic’s early days. Privacy issues, combined with stigma tied to mental illness, made this shift to virtual instruction particularly challenging. But as a field, we persevered! And, as we learned during our shift toward telemedicine, many of the changes we made in medical education are probably here to stay.
Our team at the New York Institute of Technology College of Osteopathic Medicine (NYITCOM) was able to implement a novel curriculum that allowed our students to learn psychiatry and maintain high-quality medical school education.
We developed an online course for third-year students’ rotation in psychiatry, with several modules that focused on a variety of psychiatric topics and disorders, including the basic classifications and categories of depression, anxiety, personality disorders, and psychotic disorders. There were also video encounters available showing actual patient encounters. On completion of the online module, a faculty session was held to discuss topics of concern/confusion to the students, areas of interest, and a variety of related topics, such as professionalism in psychiatry, essentials of the mental status exam, management of diverse populations, and COVID repercussions in psychiatry.
For fourth-year students, we developed a telemedicine psychiatry elective, which allowed the students to observe psychiatric evaluations, psychiatric medication review visits, and even follow-up psychotherapy sessions, with the school’s clinical psychologists. The new method was minimally invasive, and it was accepted by patients and welcomed by the students.
During a time when hospitals were limiting onsite student rotations and discouraging patient contact, medical students still needed to experience patient interactions. As the director of the school’s Center for Behavioral Health, I designed an additional program that allowed students to participate in observing patients who presented with psychiatric complaints and symptoms. It had to be confidential in nature, accessible, and safe.
I recalled my own training in a hospital setting, where students and residents were allowed to observe a patient being evaluated by an attending, through a one-way mirror. It was a method that was acceptable at the time in a hospital, but unfortunately, not in a private office setting. As such, students and residents experienced such an interaction in acute inpatient and/or outpatient clinics of a hospital. The experience was invaluable.
The concept was simple, yet very efficient. The clinicians in the Center for Behavioral Health were seeing all patients with psychiatric needs via a HIPAA-compliant telemedicine platform. Access was granted for students – with the patient’s consent – and they “entered the session” without being seen or heard. This presented little to no distraction to the patient, and the student was able to observe a range of clinical sessions.
The course also provided online supplemental modules, including essential psychiatric topics, psychopharmacology, and a psychotherapeutic module that discussed a myriad of therapeutic interventions. In addition, the student was supervised weekly by the course director, the psychopharmacologist, and the clinical psychologist. The course director provided daily wrap-up reviews as well.
Originally, this new approach was envisioned as a temporary solution for use during the pandemic. But it has become clear that this approach would be beneficial post pandemic as well. Most of the students who participated in the course were actually interested in pursuing psychiatry as their future specialty. It allowed them to observe a population of patients firsthand that they might encounter in private practice, as opposed to only hospital settings.
Being present in a session with a patient with psychiatric symptoms and diagnoses has always been a challenge. Many patients refuse to have another medical professional in the room because of the intimate details being discussed and their associated stigma. The patients’ inability to see or hear the student during the sessions allows them to ignore the students’ presence – or at least not be intimidated by it. This, therefore, allows the students access and affords them a unique and memorable educational experience.
The pandemic curtailed and altered medical students’ traditional exposure to patients, but we found innovative ways to redefine it. As difficult as COVID-19 has been for the health care community, we have been able to use the restrictions forced by the pandemic to identify innovative ways to improve the education of our medical students.
In addition to serving as director of the Center for Behavioral Health at NYITCOM in Old Westbury, N.Y., Dr. Jarkon is assistant professor in the department of family medicine. She has no disclosures.
Your bathroom towel rack has a dirty little secret
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
Bacteria get the rack ... the towel rack
Obviously, bathrooms have germs. Some people are cleaner about their bathrooms than others, but in general most people just try not to think about the microscopic critters crawling about.
Now you would probably think that the toilet is the dirtiest part of the bathroom because that’s where ... you know, most of the business takes place. Or maybe you’d guess the floor. Truth be told, though, the dirtiest part of the bathroom is where the towels are hung.
According to research conducted by electric heating company Rointe in the United Kingdom, bathroom radiators and towel racks/bars are the most germy and dirty parts of the bathroom.
Company investigators examined five bathrooms using swabs that changed color on contact with bacteria and found that 60% of towel racks and radiators were “really dirty,” compared with 50% of sink drains and just 10% of toilets.
Most people probably pay more attention to the sink, floors, and toilets while cleaning, the company suggested, and dampness is a factor in bacteria growth, so it’s no surprise that towels that stay wet on a rack are prime spots for dust, mildew, and mold.
The toilet may be busier, but you don’t put your face in it.
Anti-vaxxers would like to be called ‘purebloods’
COVID-19 anti-vaxxers are an interesting bunch, to be kind. And TikTok is a wacky place. So you can just imagine that anti-vaxxer TikTok is a very strange place. The citizens of anti-vax TikTok have decided that the real reason so many people dislike them is branding. They consider anti-vaccination to be a negative word (duh), so they now want to be referred to as “purebloods.”
Harry Potter doesn’t quite occupy the zeitgeist as it once did, so let’s give you a reminder: In the books, purebloods came from old wizarding families and claimed not to have any Muggle, or nonmagic, blood. While having pure wizard blood was no guarantee of being a villain, most of them were. In addition, it is made quite clear throughout the novels that having supposedly pure blood had no relevance on one’s wizarding ability. Pureblood was a meaningless title, and only the characters with small, cruel minds concerned themselves over it.
Perhaps the anti-vaxxers have decided that they want to be called the same thing. Maybe they just like the name. It does sound impressive and vaguely regal: Pureblood. Like something the nobles of medieval Europe might have used.
Critical-thinking skills may be in short supply here, or maybe the anti-vaxxers know exactly what they’re doing.
Hated broccoli? Blame your DNA
Were you that kid who would rather sit at the table for hours than eat your broccoli? Well, as much as your parents might have pushed you, new research suggests that it might be their fault you didn’t like it to begin with.
Investigators at Australia’s national science agency, CSIRO, recently reported that distaste for Brassica vegetables – broccoli, Brussels sprouts, cabbage, and cauliflower – can be traced to the oral microbiome.
These vegetables have a compound called S-methyl-L-cysteine sulfoxide that gives off sulfurous odors ... mmm, sulfurous ... when mixed with an enzyme in the plant, and that enzyme is also produced by bacteria in some people’s oral microbiomes. So why do adults tolerate these Brassica veggies more than children? It’s all about levels.
The researchers tested the idea by asking 98 child/parent pairs to rate the odors and by using gas chromatography-olfactometry-mass spectrometry to identify the odor-active compounds in both raw and steamed cauliflower and broccoli. The children whose saliva produced high levels of sulfur volatiles disliked Brassica vegetables the most, they reported, and the children with high levels of sulfur volatiles usually had parents who produced high levels.
Despite that connection, however, the distaste for raw Brassica seen in children wasn’t seen in adults.
Maybe it’s not that taste buds change as we age, maybe we just learn to tolerate the sulfurousness.
New finasteride lawsuit brings renewed attention to psychiatric, ED adverse event reports
A new merit a closer look and, potentially, better counseling and monitoring from clinicians.
The nonprofit advocacy group Public Citizen filed the suit on behalf of the Post-Finasteride Syndrome Foundation (PFSF) in the U.S. District Court for the District of Columbia. The PFSF had filed a citizen’s petition in 2017 that requested that the FDA either take the 1-mg formulation off the market, or add warnings about the potential for erectile dysfunction, depression, and suicidal ideation, among other adverse reactions.
The PFSF has alleged that long-term use of Propecia (and its generic equivalents) can lead to postfinasteride syndrome (PFS), characterized by sexual dysfunction and psycho-neurocognitive symptoms. The symptoms may continue long after men stop taking the drug, according to PFSF.
Public Citizen said the FDA needs to take action in part because U.S. prescriptions of the hair loss formulation “more than doubled from 2015 to 2020,” and online and telemedicine companies such as Hims, Roman, and Keeps “aggressively market and sell generic finasteride for hair loss.” According to GoodRx, a 1-month supply of generic 1-mg tablets costs as little as $8-$10.
Both Canadian and British regulatory authorities have added warnings about depression and suicide to the Propecia label but the FDA has not changed its labeling. An agency spokesperson told this news organization that the “FDA does not comment on the status of pending citizen petitions or on pending litigation.”
Propecia’s developer, Merck, has not responded to several requests for comment from this news organization.
Why some patients develop PFS and others do not is still not understood, but some clinicians said they counsel all patients on the risks of severe and persistent side effects that have been associated with Propecia.
Robert M. Bernstein, MD, of the department of dermatology at Columbia University, New York, and a fellow of the International Society of Hair Restoration Surgery, said that 2%-4% of his patients have some side effects, similar to the original reported incidence, with sexual dysfunction being the most common.
If a man experiences an adverse effect, the drug should be stopped, Dr. Bernstein said in an interview. He noted that “there seems to be a significant increased risk of persistent side effects in people with certain psychiatric conditions, and those people should be counseled carefully before considering the medication.”
“Everybody should be warned that the risk of persistent side effects is real but in the average person it is quite uncommon,” added Dr. Bernstein, founder of Bernstein Medical, a division of Schweiger Dermatology Group focusing on the diagnosis and treatment of hair loss. “I don’t think it should be withdrawn from the market,” he said.
Alan Jacobs, MD, a Manhattan-based neuroendocrinologist and behavioral neurologist in private practice who said he has treated hundreds of men for PFS, and who is an expert witness for the plaintiff in a suit alleging that finasteride led to a man’s suicide, said that taking the drug off the market would be unfortunate because it helps so many men. “I don’t think you need to get rid of the drug per se,” he said in an interview. “But very rapidly, people need to do clinical research to find out how to predict who’s more at risk,” he added.
Michael S. Irwig, MD, associate professor of medicine at Harvard Medical School, Boston, who has studied the persistent sexual and nonsexual side effects of finasteride, said he believes there should be a boxed warning on the finasteride label to let the men who take it “know that they can have permanent persistent sexual dysfunction, and/or depression and suicide have been noted with this medicine.
“Those who prescribe it should be having a conversation with patients about the potential risks and benefits so that everybody knows about the potential before they get on the medicine,” said Dr. Irwig, who also is an endocrinologist at Beth Israel Deaconess Medical Center in Boston.
Other countries warn of psychiatric effects
The FDA approved the 1-mg form of finasteride for male pattern hair loss in 1997.
In 2012, the label and the patient insert were updated to state that side effects included less desire for sex, erectile dysfunction, and a decrease in the amount of semen produced, but that those adverse events occurred in less than 2% of men and generally went away in most men who stopped taking the drug.
That label change unleashed a flood of more than 1,000 lawsuits against Merck. The company reportedly settled at least half of them for $4.3 million in 2018. The Superior Court of New Jersey closed out the consolidated class action against Merck in May 2021, noting that all of the cases had been settled or dismissed.
The suits generally accused Merck of not giving adequate warning about sexual side effects, according to an investigation by Reuters. That 2019 special report found that Merck had understated the number of men who experienced sexual side effects and the duration of those symptoms. The news organization also reported that from 2009 to 2018, the FDA received 5,000 reports of sexual or mental health side effects – and sometimes both – in men who took finasteride. Some 350 of the men reported suicidal thoughts, and there were 50 reports of suicide.
Public Citizen’s lawsuit alleges that VigiBase, which is managed by the World Health Organization Collaborating Centre for International Drug Monitoring, lists 378 cases of suicidal ideation, 39 cases of suicide attempt, and 88 cases of completed suicide associated with finasteride use. VigiBase collects data from 153 countries on adverse reactions to medications.
In February 2021, more documents from the class action lawsuits were unsealed in response to a Reuters request. According to the news organization, the documents showed that Merck knew of reports of depression, including suicidal thoughts, as early as 2009.
However, according to Reuters, the FDA in 2011 granted Merck’s request to only note depression as a potential side effect, without including the risk of suicidal ideation.
The current FDA label notes a small incidence of sexual dysfunction, including decreased libido (1.8% in trials) and erectile dysfunction (1.3%) and mentions depression as a side effect observed during the postmarketing period.
The Canadian label has the same statistics on sexual side effects but is much stronger on mental adverse effects: “Psychiatric disorders: mood alterations and depression, decreased libido that continued after discontinuation of treatment. Mood alterations including depressed mood and, less frequently, suicidal ideation have been reported in patients treated with finasteride 1 mg. Patients should be monitored for psychiatric symptoms, and if these occur, the patient should be advised to seek medical advice.”
In the United Kingdom, patients prescribed the drug are given a leaflet, which notes that “Mood alterations such as depressed mood, depression and, less frequently, suicidal thoughts have been reported in patients treated with Propecia,” and advises patients to stop taking the drug if they experience any of those symptoms and to discuss it with their physician.
Public Citizen noted in its lawsuit that French and German drug regulators have sent letters to clinicians advising them to inform patients of the risk of suicidal thoughts and anxiety.
Is there biological plausibility?
To bolster its argument that finasteride has dangerous psychiatric side effects, the advocacy organization cited a study first published in JAMA Dermatology in late 2020 that investigated suicidality and psychological adverse events in patients taking finasteride.
David-Dan Nguyen, MPH, and his colleagues at Brigham and Women’s Hospital in Boston, McGill University, Montreal, and the University of Montreal, examined the VigiBase database and found 356 cases of suicidality and 2,926 psychological adverse events; cases were highest from 2015 to 2019.
They documented what they called a “significant disproportionality signal for suicidality (reporting odds ratio, 1.63; 95% confidence interval, 2.90-4.15) and psychological adverse events (ROR, 4.33; 95% CI, 4.17-4.49) with finasteride, especially in younger men and those with alopecia, but not in older men or those with benign prostatic hyperplasia.
The study authors noted that some studies have suggested that men with depression have low levels of the neurosteroid allopregnanolone, which is produced by the 5-alpha reductase enzyme. Finasteride is a 5-alpha reductase inhibitor.
According to Public Citizen’s lawsuit, “The product labeling does not disclose important information about finasteride’s mechanism of action,” and “the drug inhibits multiple steroid hormone pathways that are responsible for the formation of brain neurosteroids that regulate many critical functions in the central nervous system, like sexual function, mood, sleep, cognitive function, the stress response, and motivation.”
Dr. Jacobs said that “there’s a lot of good solid high-quality research, mostly in animals, but also some on humans, showing a plausible link between blocking 5-alpha reductase in the brain, deficiency of neuroactive steroids, and depression.”
The author of an accompanying editorial, Roger S. Ho, MD, MPH, an associate professor in the department of dermatology, New York University, was skeptical. “Without a plausible biological hypothesis pharmacodynamically linking the drug and the reported adverse event, this kind of analysis may lead to false findings,” Dr. Ho said in the editorial about the Nguyen study.
Dr. Ho also wrote that he believed that the lack of a suicidality signal for dutasteride, a drug with a similar mechanism of action, but without as much media attention, “hints at a potential reporting bias unique to finasteride.”
He recommended that clinicians “conduct a full evaluation and a detailed, personalized risk-benefit assessment for patients before each prescription of finasteride.”
Important medicine, important caveats
Dr. Jacobs said that many of the men who come to him with side effects after taking finasteride have “been blown off by most of the doctors they go to see.”
Urologists dismiss them because their sexual dysfunction is not a gonad issue. They are told that it’s in their head, said Dr. Jacobs, adding that, “it is in their head, but it’s biological.”
The drug’s label advises that sexual side effects disappear when the drug is stopped. “That’s only true most of the time, not all of the time,” said Dr. Jacobs, adding that the persistence of any side effects impacts what he calls a “small subset” of men who take the drug.
“We have treated tens of thousands of patients who have benefited from the medicine and had no side effects,” said Dr. Bernstein. “But there is a lot that’s still not known about it.”
Even so, “baldness in young people is not a benign condition,” he said, adding that it can be socially debilitating. “An 18-year-old with a full head of thick hair who’s totally bald in 3 or 4 years – that can totally change his psyche,” Dr. Bernstein said. Finasteride may be the best option for those young men, and it is an important medication, he said. Does it need to be used more carefully? “Certainly you can’t argue with that,” he commented.
Dr. Bernstein and Dr. Irwig reported no conflicts. Dr. Jacobs disclosed that he is an expert witness for the plaintiffs in a suit against Propecia maker Merck.
A new merit a closer look and, potentially, better counseling and monitoring from clinicians.
The nonprofit advocacy group Public Citizen filed the suit on behalf of the Post-Finasteride Syndrome Foundation (PFSF) in the U.S. District Court for the District of Columbia. The PFSF had filed a citizen’s petition in 2017 that requested that the FDA either take the 1-mg formulation off the market, or add warnings about the potential for erectile dysfunction, depression, and suicidal ideation, among other adverse reactions.
The PFSF has alleged that long-term use of Propecia (and its generic equivalents) can lead to postfinasteride syndrome (PFS), characterized by sexual dysfunction and psycho-neurocognitive symptoms. The symptoms may continue long after men stop taking the drug, according to PFSF.
Public Citizen said the FDA needs to take action in part because U.S. prescriptions of the hair loss formulation “more than doubled from 2015 to 2020,” and online and telemedicine companies such as Hims, Roman, and Keeps “aggressively market and sell generic finasteride for hair loss.” According to GoodRx, a 1-month supply of generic 1-mg tablets costs as little as $8-$10.
Both Canadian and British regulatory authorities have added warnings about depression and suicide to the Propecia label but the FDA has not changed its labeling. An agency spokesperson told this news organization that the “FDA does not comment on the status of pending citizen petitions or on pending litigation.”
Propecia’s developer, Merck, has not responded to several requests for comment from this news organization.
Why some patients develop PFS and others do not is still not understood, but some clinicians said they counsel all patients on the risks of severe and persistent side effects that have been associated with Propecia.
Robert M. Bernstein, MD, of the department of dermatology at Columbia University, New York, and a fellow of the International Society of Hair Restoration Surgery, said that 2%-4% of his patients have some side effects, similar to the original reported incidence, with sexual dysfunction being the most common.
If a man experiences an adverse effect, the drug should be stopped, Dr. Bernstein said in an interview. He noted that “there seems to be a significant increased risk of persistent side effects in people with certain psychiatric conditions, and those people should be counseled carefully before considering the medication.”
“Everybody should be warned that the risk of persistent side effects is real but in the average person it is quite uncommon,” added Dr. Bernstein, founder of Bernstein Medical, a division of Schweiger Dermatology Group focusing on the diagnosis and treatment of hair loss. “I don’t think it should be withdrawn from the market,” he said.
Alan Jacobs, MD, a Manhattan-based neuroendocrinologist and behavioral neurologist in private practice who said he has treated hundreds of men for PFS, and who is an expert witness for the plaintiff in a suit alleging that finasteride led to a man’s suicide, said that taking the drug off the market would be unfortunate because it helps so many men. “I don’t think you need to get rid of the drug per se,” he said in an interview. “But very rapidly, people need to do clinical research to find out how to predict who’s more at risk,” he added.
Michael S. Irwig, MD, associate professor of medicine at Harvard Medical School, Boston, who has studied the persistent sexual and nonsexual side effects of finasteride, said he believes there should be a boxed warning on the finasteride label to let the men who take it “know that they can have permanent persistent sexual dysfunction, and/or depression and suicide have been noted with this medicine.
“Those who prescribe it should be having a conversation with patients about the potential risks and benefits so that everybody knows about the potential before they get on the medicine,” said Dr. Irwig, who also is an endocrinologist at Beth Israel Deaconess Medical Center in Boston.
Other countries warn of psychiatric effects
The FDA approved the 1-mg form of finasteride for male pattern hair loss in 1997.
In 2012, the label and the patient insert were updated to state that side effects included less desire for sex, erectile dysfunction, and a decrease in the amount of semen produced, but that those adverse events occurred in less than 2% of men and generally went away in most men who stopped taking the drug.
That label change unleashed a flood of more than 1,000 lawsuits against Merck. The company reportedly settled at least half of them for $4.3 million in 2018. The Superior Court of New Jersey closed out the consolidated class action against Merck in May 2021, noting that all of the cases had been settled or dismissed.
The suits generally accused Merck of not giving adequate warning about sexual side effects, according to an investigation by Reuters. That 2019 special report found that Merck had understated the number of men who experienced sexual side effects and the duration of those symptoms. The news organization also reported that from 2009 to 2018, the FDA received 5,000 reports of sexual or mental health side effects – and sometimes both – in men who took finasteride. Some 350 of the men reported suicidal thoughts, and there were 50 reports of suicide.
Public Citizen’s lawsuit alleges that VigiBase, which is managed by the World Health Organization Collaborating Centre for International Drug Monitoring, lists 378 cases of suicidal ideation, 39 cases of suicide attempt, and 88 cases of completed suicide associated with finasteride use. VigiBase collects data from 153 countries on adverse reactions to medications.
In February 2021, more documents from the class action lawsuits were unsealed in response to a Reuters request. According to the news organization, the documents showed that Merck knew of reports of depression, including suicidal thoughts, as early as 2009.
However, according to Reuters, the FDA in 2011 granted Merck’s request to only note depression as a potential side effect, without including the risk of suicidal ideation.
The current FDA label notes a small incidence of sexual dysfunction, including decreased libido (1.8% in trials) and erectile dysfunction (1.3%) and mentions depression as a side effect observed during the postmarketing period.
The Canadian label has the same statistics on sexual side effects but is much stronger on mental adverse effects: “Psychiatric disorders: mood alterations and depression, decreased libido that continued after discontinuation of treatment. Mood alterations including depressed mood and, less frequently, suicidal ideation have been reported in patients treated with finasteride 1 mg. Patients should be monitored for psychiatric symptoms, and if these occur, the patient should be advised to seek medical advice.”
In the United Kingdom, patients prescribed the drug are given a leaflet, which notes that “Mood alterations such as depressed mood, depression and, less frequently, suicidal thoughts have been reported in patients treated with Propecia,” and advises patients to stop taking the drug if they experience any of those symptoms and to discuss it with their physician.
Public Citizen noted in its lawsuit that French and German drug regulators have sent letters to clinicians advising them to inform patients of the risk of suicidal thoughts and anxiety.
Is there biological plausibility?
To bolster its argument that finasteride has dangerous psychiatric side effects, the advocacy organization cited a study first published in JAMA Dermatology in late 2020 that investigated suicidality and psychological adverse events in patients taking finasteride.
David-Dan Nguyen, MPH, and his colleagues at Brigham and Women’s Hospital in Boston, McGill University, Montreal, and the University of Montreal, examined the VigiBase database and found 356 cases of suicidality and 2,926 psychological adverse events; cases were highest from 2015 to 2019.
They documented what they called a “significant disproportionality signal for suicidality (reporting odds ratio, 1.63; 95% confidence interval, 2.90-4.15) and psychological adverse events (ROR, 4.33; 95% CI, 4.17-4.49) with finasteride, especially in younger men and those with alopecia, but not in older men or those with benign prostatic hyperplasia.
The study authors noted that some studies have suggested that men with depression have low levels of the neurosteroid allopregnanolone, which is produced by the 5-alpha reductase enzyme. Finasteride is a 5-alpha reductase inhibitor.
According to Public Citizen’s lawsuit, “The product labeling does not disclose important information about finasteride’s mechanism of action,” and “the drug inhibits multiple steroid hormone pathways that are responsible for the formation of brain neurosteroids that regulate many critical functions in the central nervous system, like sexual function, mood, sleep, cognitive function, the stress response, and motivation.”
Dr. Jacobs said that “there’s a lot of good solid high-quality research, mostly in animals, but also some on humans, showing a plausible link between blocking 5-alpha reductase in the brain, deficiency of neuroactive steroids, and depression.”
The author of an accompanying editorial, Roger S. Ho, MD, MPH, an associate professor in the department of dermatology, New York University, was skeptical. “Without a plausible biological hypothesis pharmacodynamically linking the drug and the reported adverse event, this kind of analysis may lead to false findings,” Dr. Ho said in the editorial about the Nguyen study.
Dr. Ho also wrote that he believed that the lack of a suicidality signal for dutasteride, a drug with a similar mechanism of action, but without as much media attention, “hints at a potential reporting bias unique to finasteride.”
He recommended that clinicians “conduct a full evaluation and a detailed, personalized risk-benefit assessment for patients before each prescription of finasteride.”
Important medicine, important caveats
Dr. Jacobs said that many of the men who come to him with side effects after taking finasteride have “been blown off by most of the doctors they go to see.”
Urologists dismiss them because their sexual dysfunction is not a gonad issue. They are told that it’s in their head, said Dr. Jacobs, adding that, “it is in their head, but it’s biological.”
The drug’s label advises that sexual side effects disappear when the drug is stopped. “That’s only true most of the time, not all of the time,” said Dr. Jacobs, adding that the persistence of any side effects impacts what he calls a “small subset” of men who take the drug.
“We have treated tens of thousands of patients who have benefited from the medicine and had no side effects,” said Dr. Bernstein. “But there is a lot that’s still not known about it.”
Even so, “baldness in young people is not a benign condition,” he said, adding that it can be socially debilitating. “An 18-year-old with a full head of thick hair who’s totally bald in 3 or 4 years – that can totally change his psyche,” Dr. Bernstein said. Finasteride may be the best option for those young men, and it is an important medication, he said. Does it need to be used more carefully? “Certainly you can’t argue with that,” he commented.
Dr. Bernstein and Dr. Irwig reported no conflicts. Dr. Jacobs disclosed that he is an expert witness for the plaintiffs in a suit against Propecia maker Merck.
A new merit a closer look and, potentially, better counseling and monitoring from clinicians.
The nonprofit advocacy group Public Citizen filed the suit on behalf of the Post-Finasteride Syndrome Foundation (PFSF) in the U.S. District Court for the District of Columbia. The PFSF had filed a citizen’s petition in 2017 that requested that the FDA either take the 1-mg formulation off the market, or add warnings about the potential for erectile dysfunction, depression, and suicidal ideation, among other adverse reactions.
The PFSF has alleged that long-term use of Propecia (and its generic equivalents) can lead to postfinasteride syndrome (PFS), characterized by sexual dysfunction and psycho-neurocognitive symptoms. The symptoms may continue long after men stop taking the drug, according to PFSF.
Public Citizen said the FDA needs to take action in part because U.S. prescriptions of the hair loss formulation “more than doubled from 2015 to 2020,” and online and telemedicine companies such as Hims, Roman, and Keeps “aggressively market and sell generic finasteride for hair loss.” According to GoodRx, a 1-month supply of generic 1-mg tablets costs as little as $8-$10.
Both Canadian and British regulatory authorities have added warnings about depression and suicide to the Propecia label but the FDA has not changed its labeling. An agency spokesperson told this news organization that the “FDA does not comment on the status of pending citizen petitions or on pending litigation.”
Propecia’s developer, Merck, has not responded to several requests for comment from this news organization.
Why some patients develop PFS and others do not is still not understood, but some clinicians said they counsel all patients on the risks of severe and persistent side effects that have been associated with Propecia.
Robert M. Bernstein, MD, of the department of dermatology at Columbia University, New York, and a fellow of the International Society of Hair Restoration Surgery, said that 2%-4% of his patients have some side effects, similar to the original reported incidence, with sexual dysfunction being the most common.
If a man experiences an adverse effect, the drug should be stopped, Dr. Bernstein said in an interview. He noted that “there seems to be a significant increased risk of persistent side effects in people with certain psychiatric conditions, and those people should be counseled carefully before considering the medication.”
“Everybody should be warned that the risk of persistent side effects is real but in the average person it is quite uncommon,” added Dr. Bernstein, founder of Bernstein Medical, a division of Schweiger Dermatology Group focusing on the diagnosis and treatment of hair loss. “I don’t think it should be withdrawn from the market,” he said.
Alan Jacobs, MD, a Manhattan-based neuroendocrinologist and behavioral neurologist in private practice who said he has treated hundreds of men for PFS, and who is an expert witness for the plaintiff in a suit alleging that finasteride led to a man’s suicide, said that taking the drug off the market would be unfortunate because it helps so many men. “I don’t think you need to get rid of the drug per se,” he said in an interview. “But very rapidly, people need to do clinical research to find out how to predict who’s more at risk,” he added.
Michael S. Irwig, MD, associate professor of medicine at Harvard Medical School, Boston, who has studied the persistent sexual and nonsexual side effects of finasteride, said he believes there should be a boxed warning on the finasteride label to let the men who take it “know that they can have permanent persistent sexual dysfunction, and/or depression and suicide have been noted with this medicine.
“Those who prescribe it should be having a conversation with patients about the potential risks and benefits so that everybody knows about the potential before they get on the medicine,” said Dr. Irwig, who also is an endocrinologist at Beth Israel Deaconess Medical Center in Boston.
Other countries warn of psychiatric effects
The FDA approved the 1-mg form of finasteride for male pattern hair loss in 1997.
In 2012, the label and the patient insert were updated to state that side effects included less desire for sex, erectile dysfunction, and a decrease in the amount of semen produced, but that those adverse events occurred in less than 2% of men and generally went away in most men who stopped taking the drug.
That label change unleashed a flood of more than 1,000 lawsuits against Merck. The company reportedly settled at least half of them for $4.3 million in 2018. The Superior Court of New Jersey closed out the consolidated class action against Merck in May 2021, noting that all of the cases had been settled or dismissed.
The suits generally accused Merck of not giving adequate warning about sexual side effects, according to an investigation by Reuters. That 2019 special report found that Merck had understated the number of men who experienced sexual side effects and the duration of those symptoms. The news organization also reported that from 2009 to 2018, the FDA received 5,000 reports of sexual or mental health side effects – and sometimes both – in men who took finasteride. Some 350 of the men reported suicidal thoughts, and there were 50 reports of suicide.
Public Citizen’s lawsuit alleges that VigiBase, which is managed by the World Health Organization Collaborating Centre for International Drug Monitoring, lists 378 cases of suicidal ideation, 39 cases of suicide attempt, and 88 cases of completed suicide associated with finasteride use. VigiBase collects data from 153 countries on adverse reactions to medications.
In February 2021, more documents from the class action lawsuits were unsealed in response to a Reuters request. According to the news organization, the documents showed that Merck knew of reports of depression, including suicidal thoughts, as early as 2009.
However, according to Reuters, the FDA in 2011 granted Merck’s request to only note depression as a potential side effect, without including the risk of suicidal ideation.
The current FDA label notes a small incidence of sexual dysfunction, including decreased libido (1.8% in trials) and erectile dysfunction (1.3%) and mentions depression as a side effect observed during the postmarketing period.
The Canadian label has the same statistics on sexual side effects but is much stronger on mental adverse effects: “Psychiatric disorders: mood alterations and depression, decreased libido that continued after discontinuation of treatment. Mood alterations including depressed mood and, less frequently, suicidal ideation have been reported in patients treated with finasteride 1 mg. Patients should be monitored for psychiatric symptoms, and if these occur, the patient should be advised to seek medical advice.”
In the United Kingdom, patients prescribed the drug are given a leaflet, which notes that “Mood alterations such as depressed mood, depression and, less frequently, suicidal thoughts have been reported in patients treated with Propecia,” and advises patients to stop taking the drug if they experience any of those symptoms and to discuss it with their physician.
Public Citizen noted in its lawsuit that French and German drug regulators have sent letters to clinicians advising them to inform patients of the risk of suicidal thoughts and anxiety.
Is there biological plausibility?
To bolster its argument that finasteride has dangerous psychiatric side effects, the advocacy organization cited a study first published in JAMA Dermatology in late 2020 that investigated suicidality and psychological adverse events in patients taking finasteride.
David-Dan Nguyen, MPH, and his colleagues at Brigham and Women’s Hospital in Boston, McGill University, Montreal, and the University of Montreal, examined the VigiBase database and found 356 cases of suicidality and 2,926 psychological adverse events; cases were highest from 2015 to 2019.
They documented what they called a “significant disproportionality signal for suicidality (reporting odds ratio, 1.63; 95% confidence interval, 2.90-4.15) and psychological adverse events (ROR, 4.33; 95% CI, 4.17-4.49) with finasteride, especially in younger men and those with alopecia, but not in older men or those with benign prostatic hyperplasia.
The study authors noted that some studies have suggested that men with depression have low levels of the neurosteroid allopregnanolone, which is produced by the 5-alpha reductase enzyme. Finasteride is a 5-alpha reductase inhibitor.
According to Public Citizen’s lawsuit, “The product labeling does not disclose important information about finasteride’s mechanism of action,” and “the drug inhibits multiple steroid hormone pathways that are responsible for the formation of brain neurosteroids that regulate many critical functions in the central nervous system, like sexual function, mood, sleep, cognitive function, the stress response, and motivation.”
Dr. Jacobs said that “there’s a lot of good solid high-quality research, mostly in animals, but also some on humans, showing a plausible link between blocking 5-alpha reductase in the brain, deficiency of neuroactive steroids, and depression.”
The author of an accompanying editorial, Roger S. Ho, MD, MPH, an associate professor in the department of dermatology, New York University, was skeptical. “Without a plausible biological hypothesis pharmacodynamically linking the drug and the reported adverse event, this kind of analysis may lead to false findings,” Dr. Ho said in the editorial about the Nguyen study.
Dr. Ho also wrote that he believed that the lack of a suicidality signal for dutasteride, a drug with a similar mechanism of action, but without as much media attention, “hints at a potential reporting bias unique to finasteride.”
He recommended that clinicians “conduct a full evaluation and a detailed, personalized risk-benefit assessment for patients before each prescription of finasteride.”
Important medicine, important caveats
Dr. Jacobs said that many of the men who come to him with side effects after taking finasteride have “been blown off by most of the doctors they go to see.”
Urologists dismiss them because their sexual dysfunction is not a gonad issue. They are told that it’s in their head, said Dr. Jacobs, adding that, “it is in their head, but it’s biological.”
The drug’s label advises that sexual side effects disappear when the drug is stopped. “That’s only true most of the time, not all of the time,” said Dr. Jacobs, adding that the persistence of any side effects impacts what he calls a “small subset” of men who take the drug.
“We have treated tens of thousands of patients who have benefited from the medicine and had no side effects,” said Dr. Bernstein. “But there is a lot that’s still not known about it.”
Even so, “baldness in young people is not a benign condition,” he said, adding that it can be socially debilitating. “An 18-year-old with a full head of thick hair who’s totally bald in 3 or 4 years – that can totally change his psyche,” Dr. Bernstein said. Finasteride may be the best option for those young men, and it is an important medication, he said. Does it need to be used more carefully? “Certainly you can’t argue with that,” he commented.
Dr. Bernstein and Dr. Irwig reported no conflicts. Dr. Jacobs disclosed that he is an expert witness for the plaintiffs in a suit against Propecia maker Merck.
















