User login
Does stenting of severe renal artery stenosis improve outomes compared with medical therapy alone?
No. In patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting offers no additional benefit when added to comprehensive medical therapy.
In these patients, renal artery stenting in addition to antihypertensive drug therapy can improve blood pressure control modestly but has no significant effect on outcomes such as adverse cardiovascular events and death. And because renal artery stenting carries a risk of complications, medical management should continue to be the first-line therapy.
RENAL ARTERY STENOSIS
Renal artery stenosis is a common form of peripheral artery disease. Atherosclerosis is the most common cause, but it can also be caused by fibromuscular dysplasia or vasculitis (eg, Takayasu arteritis). It is most often unilateral, but bilateral disease has also been reported.
The prevalence of atherosclerotic renal vascular disease in the US Medicare population is 0.5%, and 5.5% in those with chronic kidney disease.1 Furthermore, renal artery stenosis is found in 6.8% of adults over age 65.2 The prevalence increases with age and is higher in patients with hyperlipidemia, peripheral arterial disease, and hypertension. The prevalence of renal artery stenosis in patients with atherosclerotic disease and renal dysfunction is as high as 50%.3
Patients with peripheral artery disease may be five times more likely to develop renal artery stenosis than people without peripheral artery disease.4 Significant stenosis can result in resistant arterial hypertension, renal insufficiency, left ventricular hypertrophy, and congestive heart failure.5
Nephropathy due to renal artery stenosis is complex and is caused by hypoperfusion and chronic microatheroembolism. Renal artery stenosis leads to oxidative stress, inflammation, fibrosis in the stenotic kidney, and, over time, loss of kidney function. Hypoperfusion also leads to activation of the renin-angiotensin-aldosterone system, which plays a role in development of left ventricular hypertrophy.5,6
Adequate blood pressure control, goal-directed lipid-lowering therapy, smoking cessation, and other preventive measures are the foundation of management.
RENAL ARTERY STENOSIS AND HYPERTENSION
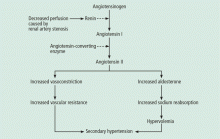
Renal artery stenosis is a cause of secondary hypertension. The stenosis decreases renal perfusion pressure, activating the release of renin and the production of angiotensin II, which in turn raises the blood pressure by two mechanisms (Figure 1): directly, by causing generalized vasoconstriction, and indirectly, by stimulating the release of aldosterone, which in turn increases the reabsorption of sodium and causes hypervolemia. These two mechanisms play a major role in renal vascular hypertension when renal artery stenosis is bilateral. In unilateral renal artery stenosis, pressure diuresis in the unaffected kidney compensates for the reabsorption of sodium in the affected kidney, keeping the blood pressure down. However, with time, the unaffected kidney will develop hypertensive nephropathy, and pressure diuresis will be lost.7,8 In addition, the activation of the renin-angiotensin-aldosterone system results in structural heart disease, such as left ventricular hypertrophy,5 and may shorten survival.
STENTING PLUS ANTIHYPERTENSIVE DRUG THERAPY
Because observational studies showed improvement in blood pressure control after endovascular stenting of atherosclerotic renal artery stenosis,9,10 this approach became a treatment option for uncontrolled hypertension in these patients. The 2005 joint guidelines of the American College of Cardiology and the American Heart Association11 considered percutaneous revascularization a reasonable option (level of evidence B) for patients who meet one of the following criteria:
- Hemodynamically significant stenosis and accelerated, resistant, or malignant hypertension, hypertension with an unexplained unilateral small kidney, or hypertension with intolerance to medication
- Renal artery stenosis and progressive chronic kidney disease with bilateral stenosis or stenosis in a solitary functioning kidney
- Hemodynamically significant stenosis and recurrent, unexplained congestive heart failure or sudden, unexplained pulmonary edema or unstable angina.11
However, no randomized study has shown a direct benefit of renal artery stenting on rates of cardiovascular events or renal function compared with drug therapy alone.
TRIALS OF STENTING VS MEDICAL THERAPY ALONE
Technical improvements have led to more widespread use of diagnostic and interventional endovascular tools for renal artery revascularization. Studies over the past 10 years examined the impact of stenting in patients with uncontrolled hypertension.
The STAR trial
In the Stent Placement and Blood Pressure and Lipid-lowering for the Prevention of Progression of Renal Dysfunction Caused by Atherosclerotic Ostial Stenosis of the Renal Artery (STAR) trial,9 patients with creatinine clearance less than 80 mL/min/1.73 m2, renal artery stenosis greater than 50%, and well-controlled blood pressure were randomized to either renal artery stenting plus medical therapy or medical therapy alone. The authors concluded that stenting had no effect on the progression of renal dysfunction but led to a small number of significant, procedure-related complications. The study was criticized for including patients with mild stenosis (< 50% stenosis) and for being underpowered for the primary end point.
The ASTRAL study
The Angioplasty and Stenting for Renal Artery Lesions (ASTRAL) study10 was a similar comparison with similar results, showing no benefit from stenting with respect to renal function, systolic blood pressure control, cardiovascular events, or death.
HERCULES
The Herculink Elite Cobalt Chromium Renal Stent Trial to Demonstrate Efficacy and Safety (HERCULES)12 was a prospective multicenter study of the effects of renal artery stenting in 202 patients with significant renal artery stenosis and uncontrolled hypertension. It showed a reduction in systolic blood pressure from baseline (P < .0001). However, follow-up was only 9 months, which was insufficient to show a significant effect on long-term cardiovascular and cerebrovascular outcomes.
The CORAL trial
The Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL) trial13 used more stringent definitions and longer follow-up. It randomized 947 patients to either stenting plus medical therapy or medical therapy alone. Patients had atherosclerotic renal artery stenosis, defined as stenosis of at least 80% or stenosis of 60% to 80% with a gradient of at least 20 mm Hg in the systolic pressure), and either systolic hypertension while taking two or more antihypertensive drugs or stage 3 or higher chronic kidney disease (glomerular filtration rate < 60 mL/min/1.73 m2 as calculated by the Modification of Diet in Renal Disease formula).
Participants were followed for 43 months to detect the occurrence of adverse cardiovascular and renal events. There was no significant difference in primary outcome between stenting plus drug therapy and drug therapy alone (35.1% and 35.8%, respectively; P = .58). However, stenting plus drug therapy was associated with modestly lower systolic pressures compared with drug therapy alone (−2.3 mm Hg, 95% confidence interval −4.4 to −0.2 mm Hg, P = .03).13 This study provided strong evidence that renal artery stenting offers no significant benefit to patients with moderately severe atherosclerotic renal artery stenosis, and that stenting may actually pose an unnecessary risk.
COMPLICATIONS OF RENAL ARTERY STENTING
Complications of renal artery stenting are a limiting factor compared with drug therapy alone, especially since the procedure offers no significant benefit in outcome. Procedural complication rates of 10% to 15% have been reported.9,10,12 The CORAL trial reported arterial dissection in 2.2%, branch-vessel occlusion in 1.2%, and distal embolization in 1.2% of patients undergoing stenting.13 Other reported complications have included stent misplacement requiring an additional stent, access-vessel damage, stent embolization, renal artery thrombosis or occlusion, and death.10,12
- Kalra PA, Guo H, Kausz AT, et al. Atherosclerotic renovascular disease in United States patients aged 67 years or older: risk factors, revascularization, and prognosis. Kidney Int 2005; 68:293–301.
- Hansen KJ, Edwards MS, Craven TE, et al. Prevalence of renovascular disease in the elderly: a population-based study. J Vasc Surg 2002; 36:443–451.
- Uzu T, Takeji M, Yamada N, et al. Prevalence and outcome of renal artery stenosis in atherosclerotic patients with renal dysfunction. Hypertens Res 2002; 25:537–542.
- Benjamin MM, Fazel P, Filardo G, Choi JW, Stoler RC. Prevalence of and risk factors of renal artery stenosis in patients with resistant hypertension. Am J Cardiol 2014; 113:687–690.
- Wu S, Polavarapu N, Stouffer GA. Left ventricular hypertrophy in patients with renal artery stenosis. Am J Med Sci 2006; 332:334–338.
- Lerman LO, Textor SC, Grande JP. Mechanisms of tissue injury in renal artery stenosis: ischemia and beyond. Prog Cardiovasc Dis 2009; 52:196–203.
- Black HR, Glickman MG, Schiff M Jr, Pingoud EG. Renovascular hypertension: pathophysiology, diagnosis, and treatment. Yale J Biol Med 1978; 51:635–654.
- Tobe SW, Burgess E, Lebel M. Atherosclerotic renovascular disease. Can J Cardiol 2006; 22:623–628.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of stent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by atherosclerotic ostial stenosis of the renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
No. In patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting offers no additional benefit when added to comprehensive medical therapy.
In these patients, renal artery stenting in addition to antihypertensive drug therapy can improve blood pressure control modestly but has no significant effect on outcomes such as adverse cardiovascular events and death. And because renal artery stenting carries a risk of complications, medical management should continue to be the first-line therapy.
RENAL ARTERY STENOSIS
Renal artery stenosis is a common form of peripheral artery disease. Atherosclerosis is the most common cause, but it can also be caused by fibromuscular dysplasia or vasculitis (eg, Takayasu arteritis). It is most often unilateral, but bilateral disease has also been reported.
The prevalence of atherosclerotic renal vascular disease in the US Medicare population is 0.5%, and 5.5% in those with chronic kidney disease.1 Furthermore, renal artery stenosis is found in 6.8% of adults over age 65.2 The prevalence increases with age and is higher in patients with hyperlipidemia, peripheral arterial disease, and hypertension. The prevalence of renal artery stenosis in patients with atherosclerotic disease and renal dysfunction is as high as 50%.3
Patients with peripheral artery disease may be five times more likely to develop renal artery stenosis than people without peripheral artery disease.4 Significant stenosis can result in resistant arterial hypertension, renal insufficiency, left ventricular hypertrophy, and congestive heart failure.5
Nephropathy due to renal artery stenosis is complex and is caused by hypoperfusion and chronic microatheroembolism. Renal artery stenosis leads to oxidative stress, inflammation, fibrosis in the stenotic kidney, and, over time, loss of kidney function. Hypoperfusion also leads to activation of the renin-angiotensin-aldosterone system, which plays a role in development of left ventricular hypertrophy.5,6
Adequate blood pressure control, goal-directed lipid-lowering therapy, smoking cessation, and other preventive measures are the foundation of management.
RENAL ARTERY STENOSIS AND HYPERTENSION
Renal artery stenosis is a cause of secondary hypertension. The stenosis decreases renal perfusion pressure, activating the release of renin and the production of angiotensin II, which in turn raises the blood pressure by two mechanisms (Figure 1): directly, by causing generalized vasoconstriction, and indirectly, by stimulating the release of aldosterone, which in turn increases the reabsorption of sodium and causes hypervolemia. These two mechanisms play a major role in renal vascular hypertension when renal artery stenosis is bilateral. In unilateral renal artery stenosis, pressure diuresis in the unaffected kidney compensates for the reabsorption of sodium in the affected kidney, keeping the blood pressure down. However, with time, the unaffected kidney will develop hypertensive nephropathy, and pressure diuresis will be lost.7,8 In addition, the activation of the renin-angiotensin-aldosterone system results in structural heart disease, such as left ventricular hypertrophy,5 and may shorten survival.
STENTING PLUS ANTIHYPERTENSIVE DRUG THERAPY
Because observational studies showed improvement in blood pressure control after endovascular stenting of atherosclerotic renal artery stenosis,9,10 this approach became a treatment option for uncontrolled hypertension in these patients. The 2005 joint guidelines of the American College of Cardiology and the American Heart Association11 considered percutaneous revascularization a reasonable option (level of evidence B) for patients who meet one of the following criteria:
- Hemodynamically significant stenosis and accelerated, resistant, or malignant hypertension, hypertension with an unexplained unilateral small kidney, or hypertension with intolerance to medication
- Renal artery stenosis and progressive chronic kidney disease with bilateral stenosis or stenosis in a solitary functioning kidney
- Hemodynamically significant stenosis and recurrent, unexplained congestive heart failure or sudden, unexplained pulmonary edema or unstable angina.11
However, no randomized study has shown a direct benefit of renal artery stenting on rates of cardiovascular events or renal function compared with drug therapy alone.
TRIALS OF STENTING VS MEDICAL THERAPY ALONE
Technical improvements have led to more widespread use of diagnostic and interventional endovascular tools for renal artery revascularization. Studies over the past 10 years examined the impact of stenting in patients with uncontrolled hypertension.
The STAR trial
In the Stent Placement and Blood Pressure and Lipid-lowering for the Prevention of Progression of Renal Dysfunction Caused by Atherosclerotic Ostial Stenosis of the Renal Artery (STAR) trial,9 patients with creatinine clearance less than 80 mL/min/1.73 m2, renal artery stenosis greater than 50%, and well-controlled blood pressure were randomized to either renal artery stenting plus medical therapy or medical therapy alone. The authors concluded that stenting had no effect on the progression of renal dysfunction but led to a small number of significant, procedure-related complications. The study was criticized for including patients with mild stenosis (< 50% stenosis) and for being underpowered for the primary end point.
The ASTRAL study
The Angioplasty and Stenting for Renal Artery Lesions (ASTRAL) study10 was a similar comparison with similar results, showing no benefit from stenting with respect to renal function, systolic blood pressure control, cardiovascular events, or death.
HERCULES
The Herculink Elite Cobalt Chromium Renal Stent Trial to Demonstrate Efficacy and Safety (HERCULES)12 was a prospective multicenter study of the effects of renal artery stenting in 202 patients with significant renal artery stenosis and uncontrolled hypertension. It showed a reduction in systolic blood pressure from baseline (P < .0001). However, follow-up was only 9 months, which was insufficient to show a significant effect on long-term cardiovascular and cerebrovascular outcomes.
The CORAL trial
The Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL) trial13 used more stringent definitions and longer follow-up. It randomized 947 patients to either stenting plus medical therapy or medical therapy alone. Patients had atherosclerotic renal artery stenosis, defined as stenosis of at least 80% or stenosis of 60% to 80% with a gradient of at least 20 mm Hg in the systolic pressure), and either systolic hypertension while taking two or more antihypertensive drugs or stage 3 or higher chronic kidney disease (glomerular filtration rate < 60 mL/min/1.73 m2 as calculated by the Modification of Diet in Renal Disease formula).
Participants were followed for 43 months to detect the occurrence of adverse cardiovascular and renal events. There was no significant difference in primary outcome between stenting plus drug therapy and drug therapy alone (35.1% and 35.8%, respectively; P = .58). However, stenting plus drug therapy was associated with modestly lower systolic pressures compared with drug therapy alone (−2.3 mm Hg, 95% confidence interval −4.4 to −0.2 mm Hg, P = .03).13 This study provided strong evidence that renal artery stenting offers no significant benefit to patients with moderately severe atherosclerotic renal artery stenosis, and that stenting may actually pose an unnecessary risk.
COMPLICATIONS OF RENAL ARTERY STENTING
Complications of renal artery stenting are a limiting factor compared with drug therapy alone, especially since the procedure offers no significant benefit in outcome. Procedural complication rates of 10% to 15% have been reported.9,10,12 The CORAL trial reported arterial dissection in 2.2%, branch-vessel occlusion in 1.2%, and distal embolization in 1.2% of patients undergoing stenting.13 Other reported complications have included stent misplacement requiring an additional stent, access-vessel damage, stent embolization, renal artery thrombosis or occlusion, and death.10,12
No. In patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting offers no additional benefit when added to comprehensive medical therapy.
In these patients, renal artery stenting in addition to antihypertensive drug therapy can improve blood pressure control modestly but has no significant effect on outcomes such as adverse cardiovascular events and death. And because renal artery stenting carries a risk of complications, medical management should continue to be the first-line therapy.
RENAL ARTERY STENOSIS
Renal artery stenosis is a common form of peripheral artery disease. Atherosclerosis is the most common cause, but it can also be caused by fibromuscular dysplasia or vasculitis (eg, Takayasu arteritis). It is most often unilateral, but bilateral disease has also been reported.
The prevalence of atherosclerotic renal vascular disease in the US Medicare population is 0.5%, and 5.5% in those with chronic kidney disease.1 Furthermore, renal artery stenosis is found in 6.8% of adults over age 65.2 The prevalence increases with age and is higher in patients with hyperlipidemia, peripheral arterial disease, and hypertension. The prevalence of renal artery stenosis in patients with atherosclerotic disease and renal dysfunction is as high as 50%.3
Patients with peripheral artery disease may be five times more likely to develop renal artery stenosis than people without peripheral artery disease.4 Significant stenosis can result in resistant arterial hypertension, renal insufficiency, left ventricular hypertrophy, and congestive heart failure.5
Nephropathy due to renal artery stenosis is complex and is caused by hypoperfusion and chronic microatheroembolism. Renal artery stenosis leads to oxidative stress, inflammation, fibrosis in the stenotic kidney, and, over time, loss of kidney function. Hypoperfusion also leads to activation of the renin-angiotensin-aldosterone system, which plays a role in development of left ventricular hypertrophy.5,6
Adequate blood pressure control, goal-directed lipid-lowering therapy, smoking cessation, and other preventive measures are the foundation of management.
RENAL ARTERY STENOSIS AND HYPERTENSION
Renal artery stenosis is a cause of secondary hypertension. The stenosis decreases renal perfusion pressure, activating the release of renin and the production of angiotensin II, which in turn raises the blood pressure by two mechanisms (Figure 1): directly, by causing generalized vasoconstriction, and indirectly, by stimulating the release of aldosterone, which in turn increases the reabsorption of sodium and causes hypervolemia. These two mechanisms play a major role in renal vascular hypertension when renal artery stenosis is bilateral. In unilateral renal artery stenosis, pressure diuresis in the unaffected kidney compensates for the reabsorption of sodium in the affected kidney, keeping the blood pressure down. However, with time, the unaffected kidney will develop hypertensive nephropathy, and pressure diuresis will be lost.7,8 In addition, the activation of the renin-angiotensin-aldosterone system results in structural heart disease, such as left ventricular hypertrophy,5 and may shorten survival.
STENTING PLUS ANTIHYPERTENSIVE DRUG THERAPY
Because observational studies showed improvement in blood pressure control after endovascular stenting of atherosclerotic renal artery stenosis,9,10 this approach became a treatment option for uncontrolled hypertension in these patients. The 2005 joint guidelines of the American College of Cardiology and the American Heart Association11 considered percutaneous revascularization a reasonable option (level of evidence B) for patients who meet one of the following criteria:
- Hemodynamically significant stenosis and accelerated, resistant, or malignant hypertension, hypertension with an unexplained unilateral small kidney, or hypertension with intolerance to medication
- Renal artery stenosis and progressive chronic kidney disease with bilateral stenosis or stenosis in a solitary functioning kidney
- Hemodynamically significant stenosis and recurrent, unexplained congestive heart failure or sudden, unexplained pulmonary edema or unstable angina.11
However, no randomized study has shown a direct benefit of renal artery stenting on rates of cardiovascular events or renal function compared with drug therapy alone.
TRIALS OF STENTING VS MEDICAL THERAPY ALONE
Technical improvements have led to more widespread use of diagnostic and interventional endovascular tools for renal artery revascularization. Studies over the past 10 years examined the impact of stenting in patients with uncontrolled hypertension.
The STAR trial
In the Stent Placement and Blood Pressure and Lipid-lowering for the Prevention of Progression of Renal Dysfunction Caused by Atherosclerotic Ostial Stenosis of the Renal Artery (STAR) trial,9 patients with creatinine clearance less than 80 mL/min/1.73 m2, renal artery stenosis greater than 50%, and well-controlled blood pressure were randomized to either renal artery stenting plus medical therapy or medical therapy alone. The authors concluded that stenting had no effect on the progression of renal dysfunction but led to a small number of significant, procedure-related complications. The study was criticized for including patients with mild stenosis (< 50% stenosis) and for being underpowered for the primary end point.
The ASTRAL study
The Angioplasty and Stenting for Renal Artery Lesions (ASTRAL) study10 was a similar comparison with similar results, showing no benefit from stenting with respect to renal function, systolic blood pressure control, cardiovascular events, or death.
HERCULES
The Herculink Elite Cobalt Chromium Renal Stent Trial to Demonstrate Efficacy and Safety (HERCULES)12 was a prospective multicenter study of the effects of renal artery stenting in 202 patients with significant renal artery stenosis and uncontrolled hypertension. It showed a reduction in systolic blood pressure from baseline (P < .0001). However, follow-up was only 9 months, which was insufficient to show a significant effect on long-term cardiovascular and cerebrovascular outcomes.
The CORAL trial
The Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL) trial13 used more stringent definitions and longer follow-up. It randomized 947 patients to either stenting plus medical therapy or medical therapy alone. Patients had atherosclerotic renal artery stenosis, defined as stenosis of at least 80% or stenosis of 60% to 80% with a gradient of at least 20 mm Hg in the systolic pressure), and either systolic hypertension while taking two or more antihypertensive drugs or stage 3 or higher chronic kidney disease (glomerular filtration rate < 60 mL/min/1.73 m2 as calculated by the Modification of Diet in Renal Disease formula).
Participants were followed for 43 months to detect the occurrence of adverse cardiovascular and renal events. There was no significant difference in primary outcome between stenting plus drug therapy and drug therapy alone (35.1% and 35.8%, respectively; P = .58). However, stenting plus drug therapy was associated with modestly lower systolic pressures compared with drug therapy alone (−2.3 mm Hg, 95% confidence interval −4.4 to −0.2 mm Hg, P = .03).13 This study provided strong evidence that renal artery stenting offers no significant benefit to patients with moderately severe atherosclerotic renal artery stenosis, and that stenting may actually pose an unnecessary risk.
COMPLICATIONS OF RENAL ARTERY STENTING
Complications of renal artery stenting are a limiting factor compared with drug therapy alone, especially since the procedure offers no significant benefit in outcome. Procedural complication rates of 10% to 15% have been reported.9,10,12 The CORAL trial reported arterial dissection in 2.2%, branch-vessel occlusion in 1.2%, and distal embolization in 1.2% of patients undergoing stenting.13 Other reported complications have included stent misplacement requiring an additional stent, access-vessel damage, stent embolization, renal artery thrombosis or occlusion, and death.10,12
- Kalra PA, Guo H, Kausz AT, et al. Atherosclerotic renovascular disease in United States patients aged 67 years or older: risk factors, revascularization, and prognosis. Kidney Int 2005; 68:293–301.
- Hansen KJ, Edwards MS, Craven TE, et al. Prevalence of renovascular disease in the elderly: a population-based study. J Vasc Surg 2002; 36:443–451.
- Uzu T, Takeji M, Yamada N, et al. Prevalence and outcome of renal artery stenosis in atherosclerotic patients with renal dysfunction. Hypertens Res 2002; 25:537–542.
- Benjamin MM, Fazel P, Filardo G, Choi JW, Stoler RC. Prevalence of and risk factors of renal artery stenosis in patients with resistant hypertension. Am J Cardiol 2014; 113:687–690.
- Wu S, Polavarapu N, Stouffer GA. Left ventricular hypertrophy in patients with renal artery stenosis. Am J Med Sci 2006; 332:334–338.
- Lerman LO, Textor SC, Grande JP. Mechanisms of tissue injury in renal artery stenosis: ischemia and beyond. Prog Cardiovasc Dis 2009; 52:196–203.
- Black HR, Glickman MG, Schiff M Jr, Pingoud EG. Renovascular hypertension: pathophysiology, diagnosis, and treatment. Yale J Biol Med 1978; 51:635–654.
- Tobe SW, Burgess E, Lebel M. Atherosclerotic renovascular disease. Can J Cardiol 2006; 22:623–628.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of stent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by atherosclerotic ostial stenosis of the renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
- Kalra PA, Guo H, Kausz AT, et al. Atherosclerotic renovascular disease in United States patients aged 67 years or older: risk factors, revascularization, and prognosis. Kidney Int 2005; 68:293–301.
- Hansen KJ, Edwards MS, Craven TE, et al. Prevalence of renovascular disease in the elderly: a population-based study. J Vasc Surg 2002; 36:443–451.
- Uzu T, Takeji M, Yamada N, et al. Prevalence and outcome of renal artery stenosis in atherosclerotic patients with renal dysfunction. Hypertens Res 2002; 25:537–542.
- Benjamin MM, Fazel P, Filardo G, Choi JW, Stoler RC. Prevalence of and risk factors of renal artery stenosis in patients with resistant hypertension. Am J Cardiol 2014; 113:687–690.
- Wu S, Polavarapu N, Stouffer GA. Left ventricular hypertrophy in patients with renal artery stenosis. Am J Med Sci 2006; 332:334–338.
- Lerman LO, Textor SC, Grande JP. Mechanisms of tissue injury in renal artery stenosis: ischemia and beyond. Prog Cardiovasc Dis 2009; 52:196–203.
- Black HR, Glickman MG, Schiff M Jr, Pingoud EG. Renovascular hypertension: pathophysiology, diagnosis, and treatment. Yale J Biol Med 1978; 51:635–654.
- Tobe SW, Burgess E, Lebel M. Atherosclerotic renovascular disease. Can J Cardiol 2006; 22:623–628.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of stent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by atherosclerotic ostial stenosis of the renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
Stenting may benefit select patients with severe renal artery stenosis
In their article in this issue of the Cleveland Clinic Journal of Medicine, Kabach et al answer no to the question of whether stenting of severe renal artery stenosis improves outcomes compared with medical therapy alone.1 They review the findings of four key studies2–5 published between 2003 and 2014 and conclude that, in patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting with medical therapy can improve blood pressure control but has no significant impact on cardiovascular or mortality outcomes.1
Furthermore, the authors state that in view of the risk of complications associated with stenting, medical management should continue to be the first-line therapy.1 Indeed, the ASTRAL study (Angioplasty and Stenting for Renal Artery Lesions) investigators found substantial risks without evidence of a worthwhile clinical benefit from revascularization in patients with atherosclerotic renovascular disease.3
Nevertheless, I believe that this procedure may benefit certain patients.
MAYO CLINIC COHORT STUDY
In 2008, our group at Mayo Clinic Health system in Eau Claire, Wisconsin, published the results of a prospective cohort study in 26 patients with renal artery stenosis and chronic kidney disease who presented with rapidly worsening renal failure (defined as an increase in serum creatinine of ≥ 25%) while receiving an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB).6,7
These drugs—inhibitors of the renin-angiotensin-aldosterone system—slow the progression of chronic kidney disease but can acutely worsen renal function, especially in patients with renal artery stenosis, and withdrawing them in this situation was the focus of our study.
The patients (10 men and 16 women) ranged in age from 63 to 87 (mean age 75.3).
At enrollment, the ACE inhibitors and ARBs were discontinued, standard nephrologic care was applied, and the glomerular filtration rate (estimated by the Modification of Diet in Renal Disease Study equation) was monitored. After at least 2 weeks, percutaneous renal angioplasty with stent placement was considered if the patient met any of the following criteria:
- Persistence of renal failure
- Flash pulmonary edema
- Uncontrolled hypertension despite the use of at least three antihypertensive medications.
Nine patients underwent percutaneous angioplasty and stenting and 17 did not. The procedure was done on one renal artery in 8 patients and both renal arteries in 1. Indications for the procedure were recent worsening of renal failure in 8 patients and recent worsening renal failure together with symptomatic flash pulmonary edema in 1 patient. (Flash pulmonary edema is the only class I recommendation for percutaneous renal angioplasty in the 2006 joint guidelines of the American College of Cardiology and the American Heart Association.8) As noted above, all the patients were experiencing acute exacerbation of chronic kidney disease at the time.
We found clear evidence of additional improved and sustained renal function in the patients who underwent percutaneous renal angioplasty and stenting compared with the patients who did not (Figures 1 and 2).6,7
In an editorial9 following publication of the ASTRAL study, our group reported a subsequent 82-month analysis of our 26-patient cohort completed in June 2009. In the 7 surviving patients who had undergone percutaneous renal angioplasty and stenting, the estimated glomerular filtration rate had increased from 27.4 mL/min/1.73 m2 (± 12.7, range 11–47) at baseline to 50.3 (± 21.7, range 23–68) (P = .018) after 46.9 months.
PATIENT NUMBER 13
To illustrate how percutaneous renal angioplasty and stenting can reverse recently worsening renal failure in renal artery stenosis, I would like to discuss in greater detail a patient from our two previous reports.6,7
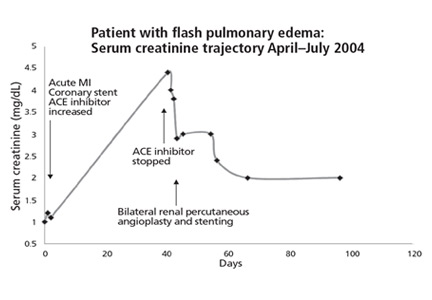
Patient 13, a 67-year-old woman with hypertension, was referred to our nephrology service in September 2004 to consider starting hemodialysis for symptomatic renal failure. Her serum creatinine had increased to 3.4 mg/dL from a previous baseline level of 2.0 mg/dL, and she also had worsening anemia and hyperkalemia. She had been taking lisinopril 10 mg/day for the last 12 months. Magnetic resonance angiography revealed high-grade (> 95%) bilateral renal artery stenosis with an atrophic left kidney.
Lisinopril was promptly discontinued, and within 2 weeks her serum creatinine level had decreased by more than 0.5 mg/dL. In mid-November 2004, she underwent right renal artery angioplasty with stent placement. After that, her serum creatinine decreased further, and 3 months later it had dropped to 1.6 mg/dL. The value continued to improve, with the lowest measurement of 1.1 mg/dL, equivalent to an estimated glomerular filtration rate of 51 mL/min/1.73 m2. This was in May 2006, 19 months after stopping lisinopril and 17 months after angioplasty and stenting. The last available serum creatinine level (August 2006) was 1.2 mg/dL, equivalent to an estimated glomerular filtration rate of 45 mL/min/1.73 m2 (Figure 1). Unfortunately, the patient died of metastatic lung cancer in December of that year.
Also of note, the patient who underwent angioplasty because of recurrent flash pulmonary edema had no recurrences of it afterward.
We concluded that, in patients with hemodynamically significant renal artery stenosis presenting with acutely worsening renal failure, renal angioplasty with stenting has the potential to reverse renal failure, improve blood pressure control, and stop flash pulmonary edema.6–8
Notably, all patients in our study who underwent renal angioplasty with stenting had new-onset acute renal injury as defined by an increase in the baseline serum creatinine of more than 25% during the 3 months before stent placement.6–8 Patients in the STAR,2 HERCULES,4 and CORAL5 studies had renal artery stenosis but otherwise stable chronic kidney disease at the time of enrollment. In the ASTRAL study,3 12% of the patients had acute kidney injury on study enrollment, defined as an increase in the serum creatinine level of more than 20% or of more than 1.13 mg/dL.3
While we strongly agree with aggressive yet monitored medical therapy for patients with hemodynamically significant renal artery stenosis, I posit that selected patients do indeed derive significant clinical benefits from renal angioplasty and stenting. Our group’s prospective individual-patient-level data support the paradigm that angioplasty with stenting is useful in patients with renal artery stenosis who experience “acute-on-chronic” kidney disease.
The pathophysiology of renal artery stenosis and the progression of chronic kidney disease are complex, and many factors affect patient outcomes and response to treatment. Thus, the message is that treatment of severe renal artery stenosis must be individualized.9–11 No one treatment fits all.10,11
- Kabach A, Agha OQ, Baibars M, Alraiyes AH, Alraies MC. Does stenting of severe renal artery stenosis improve outcomes compared with medical therapy alone? Cleve Clin J Med 2015; 82:491–494.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of STent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by Atherosclerotic ostial stenosis of the Renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Jaff MR, Bates M, Sullivan T, et al; HERCULES Investigators. Significant reduction in systolic blood pressure following renal artery stenting in patients with uncontrolled hypertension: results from the HERCULES trial. Catheter Cardiovasc Interv 2012; 80:343–350.
- Cooper CJ, Murphy TP, Cutlip DE, et al; CORAL Investigators. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N Engl J Med 2014; 370:13–22.
- Onuigbo MAC, Onuigbo NTC. Worsening renal failure in older chronic kidney disease patients with renal artery stenosis concurrently on renin angiotensin aldosterone system blockade: a prospective 50-month Mayo Health System clinic analysis. QJM 2008; 101:519–527.
- Onuigbo MA, Onuigbo NT. Renal failure and concurrent RAAS blockade in older CKD patients with renal artery stenosis: an extended Mayo Clinic prospective 63-month experience. Ren Fail 2008; 30:363–371.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
- Onuigbo M, Frenandes R, Nijhawan V. The ASTRAL trial results revisited—to stent or not to stent in renal artery stenosis? QJM 2010; 103:357.
- Singh M, Kovacs DF, Singh A, Dhaliwal P, Khosla S. ACE inhibition and renal artery stenosis: what lessons have we learnt? A 21st century perspective. In: Onuigbo MAC, ed. ACE inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 2. New York, NY: NOVA Publishers; 2013:203–218.
- Onuigbo MA, Onuigbo C, Onuigbo V, et al. The CKD enigma, reengineering CKD care, narrowing asymmetric information and confronting ethicomedicinomics in CKD care: the introduction of the new 'CKD express©' IT software program. In: Onuigbo MAC, ed. ACE Inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 1. New York, NY: NOVA Publishers; 2013: 41–56.
In their article in this issue of the Cleveland Clinic Journal of Medicine, Kabach et al answer no to the question of whether stenting of severe renal artery stenosis improves outcomes compared with medical therapy alone.1 They review the findings of four key studies2–5 published between 2003 and 2014 and conclude that, in patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting with medical therapy can improve blood pressure control but has no significant impact on cardiovascular or mortality outcomes.1
Furthermore, the authors state that in view of the risk of complications associated with stenting, medical management should continue to be the first-line therapy.1 Indeed, the ASTRAL study (Angioplasty and Stenting for Renal Artery Lesions) investigators found substantial risks without evidence of a worthwhile clinical benefit from revascularization in patients with atherosclerotic renovascular disease.3
Nevertheless, I believe that this procedure may benefit certain patients.
MAYO CLINIC COHORT STUDY
In 2008, our group at Mayo Clinic Health system in Eau Claire, Wisconsin, published the results of a prospective cohort study in 26 patients with renal artery stenosis and chronic kidney disease who presented with rapidly worsening renal failure (defined as an increase in serum creatinine of ≥ 25%) while receiving an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB).6,7
These drugs—inhibitors of the renin-angiotensin-aldosterone system—slow the progression of chronic kidney disease but can acutely worsen renal function, especially in patients with renal artery stenosis, and withdrawing them in this situation was the focus of our study.
The patients (10 men and 16 women) ranged in age from 63 to 87 (mean age 75.3).
At enrollment, the ACE inhibitors and ARBs were discontinued, standard nephrologic care was applied, and the glomerular filtration rate (estimated by the Modification of Diet in Renal Disease Study equation) was monitored. After at least 2 weeks, percutaneous renal angioplasty with stent placement was considered if the patient met any of the following criteria:
- Persistence of renal failure
- Flash pulmonary edema
- Uncontrolled hypertension despite the use of at least three antihypertensive medications.
Nine patients underwent percutaneous angioplasty and stenting and 17 did not. The procedure was done on one renal artery in 8 patients and both renal arteries in 1. Indications for the procedure were recent worsening of renal failure in 8 patients and recent worsening renal failure together with symptomatic flash pulmonary edema in 1 patient. (Flash pulmonary edema is the only class I recommendation for percutaneous renal angioplasty in the 2006 joint guidelines of the American College of Cardiology and the American Heart Association.8) As noted above, all the patients were experiencing acute exacerbation of chronic kidney disease at the time.
We found clear evidence of additional improved and sustained renal function in the patients who underwent percutaneous renal angioplasty and stenting compared with the patients who did not (Figures 1 and 2).6,7
In an editorial9 following publication of the ASTRAL study, our group reported a subsequent 82-month analysis of our 26-patient cohort completed in June 2009. In the 7 surviving patients who had undergone percutaneous renal angioplasty and stenting, the estimated glomerular filtration rate had increased from 27.4 mL/min/1.73 m2 (± 12.7, range 11–47) at baseline to 50.3 (± 21.7, range 23–68) (P = .018) after 46.9 months.
PATIENT NUMBER 13
To illustrate how percutaneous renal angioplasty and stenting can reverse recently worsening renal failure in renal artery stenosis, I would like to discuss in greater detail a patient from our two previous reports.6,7
Patient 13, a 67-year-old woman with hypertension, was referred to our nephrology service in September 2004 to consider starting hemodialysis for symptomatic renal failure. Her serum creatinine had increased to 3.4 mg/dL from a previous baseline level of 2.0 mg/dL, and she also had worsening anemia and hyperkalemia. She had been taking lisinopril 10 mg/day for the last 12 months. Magnetic resonance angiography revealed high-grade (> 95%) bilateral renal artery stenosis with an atrophic left kidney.
Lisinopril was promptly discontinued, and within 2 weeks her serum creatinine level had decreased by more than 0.5 mg/dL. In mid-November 2004, she underwent right renal artery angioplasty with stent placement. After that, her serum creatinine decreased further, and 3 months later it had dropped to 1.6 mg/dL. The value continued to improve, with the lowest measurement of 1.1 mg/dL, equivalent to an estimated glomerular filtration rate of 51 mL/min/1.73 m2. This was in May 2006, 19 months after stopping lisinopril and 17 months after angioplasty and stenting. The last available serum creatinine level (August 2006) was 1.2 mg/dL, equivalent to an estimated glomerular filtration rate of 45 mL/min/1.73 m2 (Figure 1). Unfortunately, the patient died of metastatic lung cancer in December of that year.
Also of note, the patient who underwent angioplasty because of recurrent flash pulmonary edema had no recurrences of it afterward.
We concluded that, in patients with hemodynamically significant renal artery stenosis presenting with acutely worsening renal failure, renal angioplasty with stenting has the potential to reverse renal failure, improve blood pressure control, and stop flash pulmonary edema.6–8
Notably, all patients in our study who underwent renal angioplasty with stenting had new-onset acute renal injury as defined by an increase in the baseline serum creatinine of more than 25% during the 3 months before stent placement.6–8 Patients in the STAR,2 HERCULES,4 and CORAL5 studies had renal artery stenosis but otherwise stable chronic kidney disease at the time of enrollment. In the ASTRAL study,3 12% of the patients had acute kidney injury on study enrollment, defined as an increase in the serum creatinine level of more than 20% or of more than 1.13 mg/dL.3
While we strongly agree with aggressive yet monitored medical therapy for patients with hemodynamically significant renal artery stenosis, I posit that selected patients do indeed derive significant clinical benefits from renal angioplasty and stenting. Our group’s prospective individual-patient-level data support the paradigm that angioplasty with stenting is useful in patients with renal artery stenosis who experience “acute-on-chronic” kidney disease.
The pathophysiology of renal artery stenosis and the progression of chronic kidney disease are complex, and many factors affect patient outcomes and response to treatment. Thus, the message is that treatment of severe renal artery stenosis must be individualized.9–11 No one treatment fits all.10,11
In their article in this issue of the Cleveland Clinic Journal of Medicine, Kabach et al answer no to the question of whether stenting of severe renal artery stenosis improves outcomes compared with medical therapy alone.1 They review the findings of four key studies2–5 published between 2003 and 2014 and conclude that, in patients with severe atherosclerotic renal artery stenosis and hypertension or chronic kidney disease, renal artery stenting with medical therapy can improve blood pressure control but has no significant impact on cardiovascular or mortality outcomes.1
Furthermore, the authors state that in view of the risk of complications associated with stenting, medical management should continue to be the first-line therapy.1 Indeed, the ASTRAL study (Angioplasty and Stenting for Renal Artery Lesions) investigators found substantial risks without evidence of a worthwhile clinical benefit from revascularization in patients with atherosclerotic renovascular disease.3
Nevertheless, I believe that this procedure may benefit certain patients.
MAYO CLINIC COHORT STUDY
In 2008, our group at Mayo Clinic Health system in Eau Claire, Wisconsin, published the results of a prospective cohort study in 26 patients with renal artery stenosis and chronic kidney disease who presented with rapidly worsening renal failure (defined as an increase in serum creatinine of ≥ 25%) while receiving an angiotensin-converting enzyme (ACE) inhibitor or an angiotensin II receptor blocker (ARB).6,7
These drugs—inhibitors of the renin-angiotensin-aldosterone system—slow the progression of chronic kidney disease but can acutely worsen renal function, especially in patients with renal artery stenosis, and withdrawing them in this situation was the focus of our study.
The patients (10 men and 16 women) ranged in age from 63 to 87 (mean age 75.3).
At enrollment, the ACE inhibitors and ARBs were discontinued, standard nephrologic care was applied, and the glomerular filtration rate (estimated by the Modification of Diet in Renal Disease Study equation) was monitored. After at least 2 weeks, percutaneous renal angioplasty with stent placement was considered if the patient met any of the following criteria:
- Persistence of renal failure
- Flash pulmonary edema
- Uncontrolled hypertension despite the use of at least three antihypertensive medications.
Nine patients underwent percutaneous angioplasty and stenting and 17 did not. The procedure was done on one renal artery in 8 patients and both renal arteries in 1. Indications for the procedure were recent worsening of renal failure in 8 patients and recent worsening renal failure together with symptomatic flash pulmonary edema in 1 patient. (Flash pulmonary edema is the only class I recommendation for percutaneous renal angioplasty in the 2006 joint guidelines of the American College of Cardiology and the American Heart Association.8) As noted above, all the patients were experiencing acute exacerbation of chronic kidney disease at the time.
We found clear evidence of additional improved and sustained renal function in the patients who underwent percutaneous renal angioplasty and stenting compared with the patients who did not (Figures 1 and 2).6,7
In an editorial9 following publication of the ASTRAL study, our group reported a subsequent 82-month analysis of our 26-patient cohort completed in June 2009. In the 7 surviving patients who had undergone percutaneous renal angioplasty and stenting, the estimated glomerular filtration rate had increased from 27.4 mL/min/1.73 m2 (± 12.7, range 11–47) at baseline to 50.3 (± 21.7, range 23–68) (P = .018) after 46.9 months.
PATIENT NUMBER 13
To illustrate how percutaneous renal angioplasty and stenting can reverse recently worsening renal failure in renal artery stenosis, I would like to discuss in greater detail a patient from our two previous reports.6,7
Patient 13, a 67-year-old woman with hypertension, was referred to our nephrology service in September 2004 to consider starting hemodialysis for symptomatic renal failure. Her serum creatinine had increased to 3.4 mg/dL from a previous baseline level of 2.0 mg/dL, and she also had worsening anemia and hyperkalemia. She had been taking lisinopril 10 mg/day for the last 12 months. Magnetic resonance angiography revealed high-grade (> 95%) bilateral renal artery stenosis with an atrophic left kidney.
Lisinopril was promptly discontinued, and within 2 weeks her serum creatinine level had decreased by more than 0.5 mg/dL. In mid-November 2004, she underwent right renal artery angioplasty with stent placement. After that, her serum creatinine decreased further, and 3 months later it had dropped to 1.6 mg/dL. The value continued to improve, with the lowest measurement of 1.1 mg/dL, equivalent to an estimated glomerular filtration rate of 51 mL/min/1.73 m2. This was in May 2006, 19 months after stopping lisinopril and 17 months after angioplasty and stenting. The last available serum creatinine level (August 2006) was 1.2 mg/dL, equivalent to an estimated glomerular filtration rate of 45 mL/min/1.73 m2 (Figure 1). Unfortunately, the patient died of metastatic lung cancer in December of that year.
Also of note, the patient who underwent angioplasty because of recurrent flash pulmonary edema had no recurrences of it afterward.
We concluded that, in patients with hemodynamically significant renal artery stenosis presenting with acutely worsening renal failure, renal angioplasty with stenting has the potential to reverse renal failure, improve blood pressure control, and stop flash pulmonary edema.6–8
Notably, all patients in our study who underwent renal angioplasty with stenting had new-onset acute renal injury as defined by an increase in the baseline serum creatinine of more than 25% during the 3 months before stent placement.6–8 Patients in the STAR,2 HERCULES,4 and CORAL5 studies had renal artery stenosis but otherwise stable chronic kidney disease at the time of enrollment. In the ASTRAL study,3 12% of the patients had acute kidney injury on study enrollment, defined as an increase in the serum creatinine level of more than 20% or of more than 1.13 mg/dL.3
While we strongly agree with aggressive yet monitored medical therapy for patients with hemodynamically significant renal artery stenosis, I posit that selected patients do indeed derive significant clinical benefits from renal angioplasty and stenting. Our group’s prospective individual-patient-level data support the paradigm that angioplasty with stenting is useful in patients with renal artery stenosis who experience “acute-on-chronic” kidney disease.
The pathophysiology of renal artery stenosis and the progression of chronic kidney disease are complex, and many factors affect patient outcomes and response to treatment. Thus, the message is that treatment of severe renal artery stenosis must be individualized.9–11 No one treatment fits all.10,11
- Kabach A, Agha OQ, Baibars M, Alraiyes AH, Alraies MC. Does stenting of severe renal artery stenosis improve outcomes compared with medical therapy alone? Cleve Clin J Med 2015; 82:491–494.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of STent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by Atherosclerotic ostial stenosis of the Renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Jaff MR, Bates M, Sullivan T, et al; HERCULES Investigators. Significant reduction in systolic blood pressure following renal artery stenting in patients with uncontrolled hypertension: results from the HERCULES trial. Catheter Cardiovasc Interv 2012; 80:343–350.
- Cooper CJ, Murphy TP, Cutlip DE, et al; CORAL Investigators. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N Engl J Med 2014; 370:13–22.
- Onuigbo MAC, Onuigbo NTC. Worsening renal failure in older chronic kidney disease patients with renal artery stenosis concurrently on renin angiotensin aldosterone system blockade: a prospective 50-month Mayo Health System clinic analysis. QJM 2008; 101:519–527.
- Onuigbo MA, Onuigbo NT. Renal failure and concurrent RAAS blockade in older CKD patients with renal artery stenosis: an extended Mayo Clinic prospective 63-month experience. Ren Fail 2008; 30:363–371.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
- Onuigbo M, Frenandes R, Nijhawan V. The ASTRAL trial results revisited—to stent or not to stent in renal artery stenosis? QJM 2010; 103:357.
- Singh M, Kovacs DF, Singh A, Dhaliwal P, Khosla S. ACE inhibition and renal artery stenosis: what lessons have we learnt? A 21st century perspective. In: Onuigbo MAC, ed. ACE inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 2. New York, NY: NOVA Publishers; 2013:203–218.
- Onuigbo MA, Onuigbo C, Onuigbo V, et al. The CKD enigma, reengineering CKD care, narrowing asymmetric information and confronting ethicomedicinomics in CKD care: the introduction of the new 'CKD express©' IT software program. In: Onuigbo MAC, ed. ACE Inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 1. New York, NY: NOVA Publishers; 2013: 41–56.
- Kabach A, Agha OQ, Baibars M, Alraiyes AH, Alraies MC. Does stenting of severe renal artery stenosis improve outcomes compared with medical therapy alone? Cleve Clin J Med 2015; 82:491–494.
- Bax L, Mali WP, Buskens E, et al; STAR Study Group. The benefit of STent placement and blood pressure and lipid-lowering for the prevention of progression of renal dysfunction caused by Atherosclerotic ostial stenosis of the Renal artery. The STAR-study: rationale and study design. J Nephrol 2003; 16:807–812.
- ASTRAL Investigators; Wheatley K, Ives N, Gray R, et al. Revascularization versus medical therapy for renal-artery stenosis. N Engl J Med 2009; 361:1953–1962.
- Jaff MR, Bates M, Sullivan T, et al; HERCULES Investigators. Significant reduction in systolic blood pressure following renal artery stenting in patients with uncontrolled hypertension: results from the HERCULES trial. Catheter Cardiovasc Interv 2012; 80:343–350.
- Cooper CJ, Murphy TP, Cutlip DE, et al; CORAL Investigators. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N Engl J Med 2014; 370:13–22.
- Onuigbo MAC, Onuigbo NTC. Worsening renal failure in older chronic kidney disease patients with renal artery stenosis concurrently on renin angiotensin aldosterone system blockade: a prospective 50-month Mayo Health System clinic analysis. QJM 2008; 101:519–527.
- Onuigbo MA, Onuigbo NT. Renal failure and concurrent RAAS blockade in older CKD patients with renal artery stenosis: an extended Mayo Clinic prospective 63-month experience. Ren Fail 2008; 30:363–371.
- Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): executive summary. J Am Coll Cardiol 2006; 47:1239–1312.
- Onuigbo M, Frenandes R, Nijhawan V. The ASTRAL trial results revisited—to stent or not to stent in renal artery stenosis? QJM 2010; 103:357.
- Singh M, Kovacs DF, Singh A, Dhaliwal P, Khosla S. ACE inhibition and renal artery stenosis: what lessons have we learnt? A 21st century perspective. In: Onuigbo MAC, ed. ACE inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 2. New York, NY: NOVA Publishers; 2013:203–218.
- Onuigbo MA, Onuigbo C, Onuigbo V, et al. The CKD enigma, reengineering CKD care, narrowing asymmetric information and confronting ethicomedicinomics in CKD care: the introduction of the new 'CKD express©' IT software program. In: Onuigbo MAC, ed. ACE Inhibitors: Medical Uses, Mechanisms of Action, Potential Adverse Effects and Related Topics. Volume 1. New York, NY: NOVA Publishers; 2013: 41–56.
Parvovirus mimicking acute HIV infection
A 25-year-old Jamaican man presented to the emergency department for evaluation of a rash on his face, back, and hands (Figure 1). He recalled a puncture injury after handling garbage at work. He denied recent travel, blood transfusions, or sick contacts. He was not aware of any recent arthropod bites. All standard vaccines including a tetanus booster were up to date.
Examination revealed edema of the hands and uvula. He was discharged with diphenhydramine and a short course of a systemic corticosteroid for presumed contact dermatitis.
He returned 4 days later with new symptoms, including sore throat, fever with a temperature of 103°F (39.4°C), oropharyngeal pain, dysphagia, dysuria, and purpura on the hands, abdomen, and legs. He was admitted to the hospital.
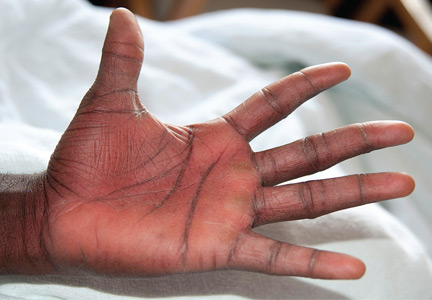
Examination revealed bright red confluent erythema of both legs extending to the lower abdomen, with petechiae on the palms (Figure 2), soles, toes, and fingers. Several small scrotal ulcers with well-defined borders were noted. Oral examination revealed white-yellow adherent plaques on the tongue and similar small ulcers on the lower lip and soft and hard palates.
A complete blood cell count revealed absolute lymphopenia, with a white blood cell count of 0.64 × 109/L (reference range 1.0–4.8) and a neutrophil percentage of 78.9% (39%–68%). Other values were within normal limits, with a red blood cell count of 5.3 × 1012/L (3.9–5.5) and a platelet count of 161 × 109/L (150–350). Serum liver enzymes were also within normal limits.
Treatment with intravenous fluids and intramuscular penicillin G was started empirically, pending a workup for infectious disease. Tests for syphilis immunoglobulin G (IgG), streptococci, anti-streptolysin O, Epstein-Barr virus, and human immunodeficiency virus (HIV) 1 and 2 were negative. The scrotal ulcers were swabbed, and culture and direct fluorescent antibody testing for cytomegalovirus and herpes simplex virus were negative. Urine testing for gonococcal and chlamydial infection was negative.
On the fifth day of hospitalization, the patient’s condition was improving, but there was still no definitive diagnosis. Consultation with the inpatient dermatology team prompted testing for parvovirus B19 infection, based on the gloves-and-socks distribution of the purpura. Testing revealed a slightly elevated parvovirus B19 IgG titer (2.61) and a significantly elevated parvovirus B19 IgM titer (12.74), which confirmed acute parvovirus infection.
The patient’s condition improved over several days with fluid administration, and he was discharged in good condition. He returned 1 week later for a follow-up appointment, at which time only superficial desquamation was noted in the areas previously affected by purpura.
PARVOVIRUS B19: NOT ONLY IN CHILDREN
Parvovirus B19 is responsible for the common childhood viral exanthem known as fifth disease.1 However, although much less common, the virus can also affect young adults, precipitating a dermatosis referred to as gloves-and-socks syndrome characterized by purpura on the hands and feet,2,3 and with a higher incidence in the spring and summer.1
Although papular-purpuric gloves-and- socks syndrome is characterized by purpura on the hands and feet, the cheeks, oral mucosa, inner thighs, buttocks, and genitalia are affected in about 50% of patients.2 In one report, in two-thirds of adult patients the presentation was caused by parvovirus B19 infection,4 but the syndrome has also been associated with Epstein-Barr virus, cytomegalovirus, human herpesvirus types 6 and 7, hepatitis B virus, rubella virus, and varicella zoster virus.4
Parvovirus B19 infection is commonly associated with systemic manifestations such as fever, fatigue, and lymphadenopathy, as well as swelling of the lips, cutaneous and mucosal ulcerations, polyarthritis, and petechiae involving the hard palate, the soft palate, or both.1
The syndrome is self-limited and resolves within 1 to 2 weeks.1
THE DIAGNOSTIC CHALLENGE
The differential diagnosis of the syndrome’s gloves-and-socks presentation includes hand-foot-mouth disease, erythema multiforme, Henoch-Schönlein purpura, and Kawasaki disease,4 in addition to viral exanthems and sexually transmitted diseases. Our patient’s fever, rash, and absolute lymphopenia focused attention on possible HIV infection, which caused the patient significant anxiety while awaiting the results of HIV testing. Heightened awareness of the cutaneous presentation of parvovirus B19 infection can help avoid unnecessary hospitalization and patient anxiety.
- Smith PT, Landry ML, Carey H, Krasnoff J, Cooney E. Papular-purpuric gloves and socks syndrome associated with acute parvovirus B19 infection: case report and review. Clin Infect Dis 1998; 27:164–168.
- Harms M, Feldmann R, Saurat JH. Papular-purpuric “gloves and socks” syndrome. J Am Acad Dermatol 1990; 23:850–854.
- Bagot M, Revuz J. Papular-purpuric “gloves and socks” syndrome: primary infection with parvovirus B19? J Am Acad Dermatol 1991; 25:341–342.
- Gutermuth J, Nadas K, Zirbs M, et al. Papular-purpuric gloves and socks syndrome. Lancet 2011; 378:198.
A 25-year-old Jamaican man presented to the emergency department for evaluation of a rash on his face, back, and hands (Figure 1). He recalled a puncture injury after handling garbage at work. He denied recent travel, blood transfusions, or sick contacts. He was not aware of any recent arthropod bites. All standard vaccines including a tetanus booster were up to date.
Examination revealed edema of the hands and uvula. He was discharged with diphenhydramine and a short course of a systemic corticosteroid for presumed contact dermatitis.
He returned 4 days later with new symptoms, including sore throat, fever with a temperature of 103°F (39.4°C), oropharyngeal pain, dysphagia, dysuria, and purpura on the hands, abdomen, and legs. He was admitted to the hospital.
Examination revealed bright red confluent erythema of both legs extending to the lower abdomen, with petechiae on the palms (Figure 2), soles, toes, and fingers. Several small scrotal ulcers with well-defined borders were noted. Oral examination revealed white-yellow adherent plaques on the tongue and similar small ulcers on the lower lip and soft and hard palates.
A complete blood cell count revealed absolute lymphopenia, with a white blood cell count of 0.64 × 109/L (reference range 1.0–4.8) and a neutrophil percentage of 78.9% (39%–68%). Other values were within normal limits, with a red blood cell count of 5.3 × 1012/L (3.9–5.5) and a platelet count of 161 × 109/L (150–350). Serum liver enzymes were also within normal limits.
Treatment with intravenous fluids and intramuscular penicillin G was started empirically, pending a workup for infectious disease. Tests for syphilis immunoglobulin G (IgG), streptococci, anti-streptolysin O, Epstein-Barr virus, and human immunodeficiency virus (HIV) 1 and 2 were negative. The scrotal ulcers were swabbed, and culture and direct fluorescent antibody testing for cytomegalovirus and herpes simplex virus were negative. Urine testing for gonococcal and chlamydial infection was negative.
On the fifth day of hospitalization, the patient’s condition was improving, but there was still no definitive diagnosis. Consultation with the inpatient dermatology team prompted testing for parvovirus B19 infection, based on the gloves-and-socks distribution of the purpura. Testing revealed a slightly elevated parvovirus B19 IgG titer (2.61) and a significantly elevated parvovirus B19 IgM titer (12.74), which confirmed acute parvovirus infection.
The patient’s condition improved over several days with fluid administration, and he was discharged in good condition. He returned 1 week later for a follow-up appointment, at which time only superficial desquamation was noted in the areas previously affected by purpura.
PARVOVIRUS B19: NOT ONLY IN CHILDREN
Parvovirus B19 is responsible for the common childhood viral exanthem known as fifth disease.1 However, although much less common, the virus can also affect young adults, precipitating a dermatosis referred to as gloves-and-socks syndrome characterized by purpura on the hands and feet,2,3 and with a higher incidence in the spring and summer.1
Although papular-purpuric gloves-and- socks syndrome is characterized by purpura on the hands and feet, the cheeks, oral mucosa, inner thighs, buttocks, and genitalia are affected in about 50% of patients.2 In one report, in two-thirds of adult patients the presentation was caused by parvovirus B19 infection,4 but the syndrome has also been associated with Epstein-Barr virus, cytomegalovirus, human herpesvirus types 6 and 7, hepatitis B virus, rubella virus, and varicella zoster virus.4
Parvovirus B19 infection is commonly associated with systemic manifestations such as fever, fatigue, and lymphadenopathy, as well as swelling of the lips, cutaneous and mucosal ulcerations, polyarthritis, and petechiae involving the hard palate, the soft palate, or both.1
The syndrome is self-limited and resolves within 1 to 2 weeks.1
THE DIAGNOSTIC CHALLENGE
The differential diagnosis of the syndrome’s gloves-and-socks presentation includes hand-foot-mouth disease, erythema multiforme, Henoch-Schönlein purpura, and Kawasaki disease,4 in addition to viral exanthems and sexually transmitted diseases. Our patient’s fever, rash, and absolute lymphopenia focused attention on possible HIV infection, which caused the patient significant anxiety while awaiting the results of HIV testing. Heightened awareness of the cutaneous presentation of parvovirus B19 infection can help avoid unnecessary hospitalization and patient anxiety.
A 25-year-old Jamaican man presented to the emergency department for evaluation of a rash on his face, back, and hands (Figure 1). He recalled a puncture injury after handling garbage at work. He denied recent travel, blood transfusions, or sick contacts. He was not aware of any recent arthropod bites. All standard vaccines including a tetanus booster were up to date.
Examination revealed edema of the hands and uvula. He was discharged with diphenhydramine and a short course of a systemic corticosteroid for presumed contact dermatitis.
He returned 4 days later with new symptoms, including sore throat, fever with a temperature of 103°F (39.4°C), oropharyngeal pain, dysphagia, dysuria, and purpura on the hands, abdomen, and legs. He was admitted to the hospital.
Examination revealed bright red confluent erythema of both legs extending to the lower abdomen, with petechiae on the palms (Figure 2), soles, toes, and fingers. Several small scrotal ulcers with well-defined borders were noted. Oral examination revealed white-yellow adherent plaques on the tongue and similar small ulcers on the lower lip and soft and hard palates.
A complete blood cell count revealed absolute lymphopenia, with a white blood cell count of 0.64 × 109/L (reference range 1.0–4.8) and a neutrophil percentage of 78.9% (39%–68%). Other values were within normal limits, with a red blood cell count of 5.3 × 1012/L (3.9–5.5) and a platelet count of 161 × 109/L (150–350). Serum liver enzymes were also within normal limits.
Treatment with intravenous fluids and intramuscular penicillin G was started empirically, pending a workup for infectious disease. Tests for syphilis immunoglobulin G (IgG), streptococci, anti-streptolysin O, Epstein-Barr virus, and human immunodeficiency virus (HIV) 1 and 2 were negative. The scrotal ulcers were swabbed, and culture and direct fluorescent antibody testing for cytomegalovirus and herpes simplex virus were negative. Urine testing for gonococcal and chlamydial infection was negative.
On the fifth day of hospitalization, the patient’s condition was improving, but there was still no definitive diagnosis. Consultation with the inpatient dermatology team prompted testing for parvovirus B19 infection, based on the gloves-and-socks distribution of the purpura. Testing revealed a slightly elevated parvovirus B19 IgG titer (2.61) and a significantly elevated parvovirus B19 IgM titer (12.74), which confirmed acute parvovirus infection.
The patient’s condition improved over several days with fluid administration, and he was discharged in good condition. He returned 1 week later for a follow-up appointment, at which time only superficial desquamation was noted in the areas previously affected by purpura.
PARVOVIRUS B19: NOT ONLY IN CHILDREN
Parvovirus B19 is responsible for the common childhood viral exanthem known as fifth disease.1 However, although much less common, the virus can also affect young adults, precipitating a dermatosis referred to as gloves-and-socks syndrome characterized by purpura on the hands and feet,2,3 and with a higher incidence in the spring and summer.1
Although papular-purpuric gloves-and- socks syndrome is characterized by purpura on the hands and feet, the cheeks, oral mucosa, inner thighs, buttocks, and genitalia are affected in about 50% of patients.2 In one report, in two-thirds of adult patients the presentation was caused by parvovirus B19 infection,4 but the syndrome has also been associated with Epstein-Barr virus, cytomegalovirus, human herpesvirus types 6 and 7, hepatitis B virus, rubella virus, and varicella zoster virus.4
Parvovirus B19 infection is commonly associated with systemic manifestations such as fever, fatigue, and lymphadenopathy, as well as swelling of the lips, cutaneous and mucosal ulcerations, polyarthritis, and petechiae involving the hard palate, the soft palate, or both.1
The syndrome is self-limited and resolves within 1 to 2 weeks.1
THE DIAGNOSTIC CHALLENGE
The differential diagnosis of the syndrome’s gloves-and-socks presentation includes hand-foot-mouth disease, erythema multiforme, Henoch-Schönlein purpura, and Kawasaki disease,4 in addition to viral exanthems and sexually transmitted diseases. Our patient’s fever, rash, and absolute lymphopenia focused attention on possible HIV infection, which caused the patient significant anxiety while awaiting the results of HIV testing. Heightened awareness of the cutaneous presentation of parvovirus B19 infection can help avoid unnecessary hospitalization and patient anxiety.
- Smith PT, Landry ML, Carey H, Krasnoff J, Cooney E. Papular-purpuric gloves and socks syndrome associated with acute parvovirus B19 infection: case report and review. Clin Infect Dis 1998; 27:164–168.
- Harms M, Feldmann R, Saurat JH. Papular-purpuric “gloves and socks” syndrome. J Am Acad Dermatol 1990; 23:850–854.
- Bagot M, Revuz J. Papular-purpuric “gloves and socks” syndrome: primary infection with parvovirus B19? J Am Acad Dermatol 1991; 25:341–342.
- Gutermuth J, Nadas K, Zirbs M, et al. Papular-purpuric gloves and socks syndrome. Lancet 2011; 378:198.
- Smith PT, Landry ML, Carey H, Krasnoff J, Cooney E. Papular-purpuric gloves and socks syndrome associated with acute parvovirus B19 infection: case report and review. Clin Infect Dis 1998; 27:164–168.
- Harms M, Feldmann R, Saurat JH. Papular-purpuric “gloves and socks” syndrome. J Am Acad Dermatol 1990; 23:850–854.
- Bagot M, Revuz J. Papular-purpuric “gloves and socks” syndrome: primary infection with parvovirus B19? J Am Acad Dermatol 1991; 25:341–342.
- Gutermuth J, Nadas K, Zirbs M, et al. Papular-purpuric gloves and socks syndrome. Lancet 2011; 378:198.
Remembering that old dogs can still do tricks
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
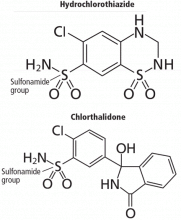
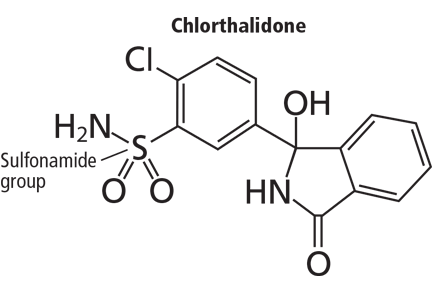
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.
Diuretics for hypertension: Hydrochlorothiazide or chlorthalidone?
The thiazide diuretic hydrochlorothiazide and the thiazidelike diuretic chlorthalidone are two old drugs that are still useful. Although similar, they differ in important ways still not fully appreciated more than a half century after they were introduced.
Most hypertension guidelines recommend thiazide diuretics as one of the classes of agents that can be used either as initial antihypertensive drug therapy or as part of combination therapy.1–3
In the United States, hydrochlorothiazide is used more often than chlorthalidone, but many clinical trials of antihypertensive therapy have used chlorthalidone.4,5 In recent years, particularly after the publication of the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), interest in chlorthalidone has been increasing, and new data are now available comparing these two diuretics.6 While current US guidelines do not recommend one over the other, British guidelines prefer chlorthalidone.7
This review summarizes the data comparing the two drugs’ pharmacology, antihypertensive effect, and impact on clinical outcomes to help guide clinicians in choosing antihypertensive drug therapy.
PHARMACOLOGY AND MECHANISM OF ACTION
Many of the differences in effectiveness and adverse effects of hydrochlorothiazide and chlorthalidone are thought to be due to their different pharmacodynamic and pharmacokinetic effects.
Pharmacodynamic effects
Hydrochlorothiazide and chlorthalidone differ significantly in chemical structure (Figure 1), but both contain a sulfonamide group that inhibits carbonic anhydrase activity, which may be associated with lower vascular contractility. Both drugs are concentrated in the kidney and secreted into the tubular lumen8; therefore, their therapeutic diuretic effects are often achieved with relatively low plasma concentrations.
Both drugs inhibit the sodium-chloride cotransporter in the luminal membrane of the distal convoluted tubule of the ascending loop of Henle, leading to a modest natriuresis and diuresis. The exact mechanism by which they lower blood pressure is not known: while the initial response is from diuresis and volume changes, long-term reduction in blood pressure is through uncertain mechanisms. In addition, chlorthalidone may have beneficial effects on endothelial function and oxidative stress.9,10
Both drugs also increase secretion of potassium and hydrogen ions and promote increased reabsorption of calcium through increased expression of a sodium-calcium exchange channel.8 Chlorthalidone may cause more inhibition of carbonic anhydrase than hydrochlorothiazide, which can lead to lower intracellular pH and cell volume. This effect may in part explain a pleiotropic effect of chlorthalidone, ie, inhibition of platelet function, which in turn may contribute to this drug’s beneficial effect on cardiovascular outcomes.9
Pharmacokinetic differences
Hydrochlorothiazide and chlorthalidone have important differences in their pharmacokinetic properties (Table 1).11
Hydrochlorothiazide has its onset of action in about 2 hours, and it reaches its peak in 4 to 6 hours. Though its duration of action is short—up to 12 hours—its pharmacodynamic response can be much longer than predicted by its kinetics, allowing once-daily dosing.8
Chlorthalidone has a longer duration of action than hydrochlorothiazide. This may be because it has a very high volume of distribution, since it is taken up into red blood cells and is bound to carbonic anhydrase.12 This may result in a “drug reservoir” that keeps drug levels higher for a longer time.13 Its long duration of action makes it a favorable choice for patients who have difficulty adhering to medication instructions. In addition, a missed dose is unlikely to have a “rebound” effect like that seen with some other antihypertensive agents. However, both chlorthalidone and hydrochlorothiazide are effective if taken once daily.
BLOOD PRESSURE-LOWERING
Both hydrochlorothiazide and chlorthalidone are effective antihypertensive agents. Table 2 summarizes findings from studies that evaluated their blood pressure-lowering effect at various doses.14–33 However, relatively few studies have directly compared these two agents’ effects on blood pressure.
Ernst et al,34 in a small study (but probably the best one to address this issue), compared chlorthalidone 12.5 mg/day (force-titrated to 25 mg/day) and hydrochlorothiazide 25 mg/day (force-titrated to 50 mg/day) in untreated hypertensive patients. After 8 weeks, ambulatory blood pressure monitoring indicated a greater reduction from baseline in systolic blood pressure with chlorthalidone 25 mg/day than with hydrochlorothiazide 50 mg/day (24-hour mean –12.4 vs –7.4 mm Hg, P = .05). Interestingly, the change in nighttime blood pressure was greater in the chlorthalidone group (–13.5 mm Hg) than in the hydrochlorothiazide group (–6.4 mm Hg; P = .009). These data suggest that at the doses studied, chlorthalidone is more effective than hydrochlorothiazide in lowering systolic blood pressure.
Bakris et al,35 using a different study design, compared the single-pill combination of azilsartan medoxomil and chlorthalidone vs coadministration of azilsartan medoxomil and hydrochlorothiazide in participants with stage 2 primary hypertension (≥ 160/100 mm Hg). Systolic blood pressure, as measured in the clinic, declined more with the chlorthalidone combination (–35.1 mm Hg) than with the hydrochlorothiazide combination (–29.5 mm Hg, mean difference –5.6 mm Hg, P < .001).
Meta-analyses also support the conclusion that chlorthalidone is more potent than hydrochlorothiazide in lowering blood pressure.35,36 Several studies have shown that chlorthalidone at the same dose is 1.5 to 2 times as potent as hydrochlorothiazide.33,36,37 Therefore, for clinical purposes, it is reasonable to consider chlorthalidone 12.5 mg daily as similar to 25 mg of hydrochlorothiazide daily.
ADVERSE EFFECTS
Electrolyte disturbances are a common adverse effect of thiazide diuretics.
Hypokalemia. All thiazide diuretics cause potassium wasting. The frequency of hypokalemia depends on the dose, frequency of administration, diet, and other pharmacologic agents used.
Two large clinical trials, the Systolic Hypertension in the Elderly Program and ALLHAT, found that chlorthalidone caused hypokalemia requiring therapy in about 6% to 8% of patients.38,39 Chlorthalidone therapy was associated with a lowering of serum potassium levels of 0.2 to 0.5 mmol/L.36 In ALLHAT, chlorthalidone was associated with a reduction in potassium levels of approximately 0.2 mmol/L after 4 years.38
All diuretics require monitoring of electrolytes, especially during the first 2 weeks of therapy. Once a steady state is reached, patients are not usually at risk of hypokalemia unless the dose is increased, extrarenal losses of potassium increase, or dietary potassium is reduced.
Other electrolyte changes. Thiazide and thiazide-like diuretics can cause other metabolic and endocrine abnormalities such as hypochloremic alkalosis, hyponatremia, and hypercalcemia.40,41 They can also cause photosensitivity and can precipitate gout.42
Observational studies have suggested that metabolic adverse effects such as hypokalemia and hyperuricemia are more common with chlorthalidone than with hydrochlorothiazide.43 It is prudent to monitor laboratory values periodically in patients on diuretic therapy.
DRUG INTERACTIONS
The drug interaction profiles of hydrochlorothiazide and chlorthalidone are also similar. The most common interactions are pharmacodynamic interactions resulting from potassium depletion caused by the diuretics.
Antiarrythymic drugs. Hypokalemia is a risk factor for arrhythmias such as torsades de pointes, and the risk is magnified with concomitant therapy with antiarrhythmic agents that prolong the QT interval independently of serum potassium concentration (eg, sotalol, dronedarone, ibutilide, propafenone). Therefore, combinations of drugs that can cause hypokalemia (eg, diuretics) and antiarrhythmic agents require vigilant monitoring of potassium and appropriate replenishment.44
Dofetilide is a class III antiarrhythmic agent and, like other antiarrhythmic drugs, carries a risk of QT prolongation and torsades de pointes, which is magnified by hypokalemia.45 In addition, dofetilide undergoes active tubular secretion in the kidney via the cation transport system, which is inhibited by hydrochlorothiazide.45 The resulting increase in plasma concentrations of dofetilide may magnify the risk of arrhythmias. Chlorthalidone has not been specifically studied in combination with dofetilide, but thiazide diuretics in general are thought to have a similar effect on tubular secretion and, therefore, should be considered similar to hydrochlorothiazide in this regard.
Digoxin. Similarly, digoxin toxicity may be enhanced in hypokalemia. As with antiarrhythmic agents, serum potassium should be carefully monitored and replenished appropriately when diuretics are used in combination with digoxin.
Lithium is reabsorbed in the proximal tubule along with sodium. Diuretics including hydrochlorothiazide and chlorthalidone that alter sodium reabsorption can also alter lithium absorption.46 When sodium reabsorption is decreased, lithium ion reabsorption is increased and may result in lithium toxicity. Although this combination is not contraindicated, monitoring of serum lithium concentrations and clinical signs and symptoms of lithium toxicity is recommended during initiation and dose adjustments of thiazide diuretics.
Nonsteroidal anti-inflammatory drugs can decrease the natriuretic, diuretic, and antihypertensive effects of both hydrochlorothiazide and chlorthalidone.47
Renin-angiotensin-aldosterone system antagonists, ie, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and the renin inhibitor aliskiren, have potentially beneficial interactions with hydrochlorothiazide and chlorthalidone, producing additive decreases in blood pressure when coadministered with these diuretics. These effects may be particularly potent early in concomitant therapy and allow use of lower doses of diuretics, typically 12.5 mg of hydrochlorothiazide in combination therapy.
LONG-TERM EFFECTS ON CARDIOVASCULAR EVENTS
The long-term goal in treating hypertension is to lower the risk of cardiovascular disease. Therefore, the clinician needs to consider the effect of antihypertensive drug therapy on long-term clinical outcomes.
Antihypertensive drug therapy based on thiazide diuretics has been shown to lower cardiovascular risk when compared with placebo.48 In addition, the effect of chlorthalidone-based antihypertensive therapy was similar to that of other antihypertensive drug classes in preventing most cardiovascular outcomes in ALLHAT.4
However, no study has directly compared hydrochlorothiazide and chlorthalidone with the primary outcome of reduction in long-term cardiovascular events. The data to date come from observational studies and meta-analyses. For example, in a retrospective analysis of the Multiple Risk Factor Intervention Trial, cardiovascular events were significantly fewer in those receiving chlorthalidone vs hydrochlorothiazide (P = .0016).43
In a systematic review and meta-analysis, chlorthalidone was associated with a 23% lower risk of heart failure and a 21% lower risk of all cardiovascular events.49
However, a Canadian observational study of 29,873 patients found no difference in the composite outcome of death or hospitalization for heart failure, stroke, or myocardial infarction between chlorthalidone recipients (3.2 events per 100 person-years) and hydrochlorothiazide recipients (3.4 events per 100 person-years; adjusted hazard ratio 0.93, 95% confidence interval 0.81–1.06).50
In summary, it is unclear whether chlorthalidone or hydrochlorothiazide is superior in preventing cardiovascular events.
SUMMARY
Thiazide and thiazidelike diuretics play an important role in managing hypertension in most patients. The eighth Joint National Committee guidelines do not recommend either hydrochlorothiazide or chlorthalidone over the other. The target dose recommendations are hydrochlorothiazide 25 to 50 mg or chlorthalidone 12.5 to 25 mg daily, with lower doses considered for the elderly.
There are important differences between hydrochlorothiazide and chlorthalidone in pharmacology, potency, and frequency of metabolic side effects. Clinicians should consider these factors to tailor the choice of thiazide diuretic therapy in hypertensive patients.
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014; 311:507–520.
- Dasgupta K, Quinn RR, Zarnke KB, et al; Canadian Hypertension Education Program. The 2014 Canadian Hypertension Education Program recommendations for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol 2014; 30:485–501.
- Mancia G, Fagard R, Narkiewicz K, et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 2013; 34:2159–2219.
- ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group; The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002; 288:2981–2997.
- Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA 1991; 265:3255–3264.
- Roush GC, Kaur R, Ernst ME. Diuretics: a review and update. J Cardiovasc Pharmacol Ther 2014; 19:5–13.
- McCormack T, Krause T, O’Flynn N. Management of hypertension in adults in primary care: NICE guideline. Br J Gen Pract 2012; 62:163–164.
- Bhattacharaya M, Alper SL. Pharmacology of volume regulation. In: Golan DE, Tashjian AH Jr, Armstrong EJ, Armstrong AW, editors. Principles of Pharmacology: The pathophysiologic Basis of Drug Therapy. 3rd ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2012:332–352.
- Woodman R, Brown C, Lockette W. Chlorthalidone decreases platelet aggregation and vascular permeability and promotes angiogenesis. Hypertension 2010; 56:463–470.
- Sato K, Dohi Y, Kojima M, Takase H, Suzuki S, Ito S. Antioxidative effects of thiazide diuretics in refractory hypertensive patients. A randomized crossover trial of chlortalidone and trichlormethiazide. Arzneimittelforschung 2010; 60:612–616.
- US National Library of Medicine. Dailymed. dailymed.nlm.nih.gov. Accessed May 14, 2015.
- Collste P, Garle M, Rawlins MD, Sjöqvist F. Interindividual differences in chlorthalidone concentration in plasma and red cells of man after single and multiple doses. Eur J Clin Pharmacol 1976; 9:319–325.
- Roush GC, Buddharaju V, Ernst ME, Holford TR. Chlorthalidone: mechanisms of action and effect on cardiovascular events. Curr Hypertens Rep 2013; 15:514–521.
- Pool JL, Cushman WC, Saini RK, Nwachuku CE, Battikha JP. Use of the factorial design and quadratic response surface models to evaluate the fosinopril and hydrochlorothiazide combination therapy in hypertension. Am J Hypertens 1997; 10:117–123.
- Pool JL, Glazer R, Weinberger M, Alvarado R, Huang J, Graff A. Comparison of valsartan/hydrochlorothiazide combination therapy at doses up to 320/25 mg versus monotherapy: a double-blind, placebo-controlled study followed by long-term combination therapy in hypertensive adults. Clin Ther 2007; 29:61–73.
- Horie Y, Higaki J, Takeuchi M. Design, statistical analysis and sample size calculation of dose response study of telmisartan and hydrochlorothiazide. Contemp Clin Trials 2007; 28:647–653.
- Chrysant SG. Antihypertensive effectiveness of low-dose lisinopril-hydrochlorothiazide combination. A large multicenter study. Lisinopril-Hydrochlorothiazide Group. Arch Intern Med 1994; 154:737–743.
- Lacourcière Y, Arnott W. Placebo-controlled comparison of the effects of nebivolol and low-dose hydrochlorothiazide as monotherapies and in combination on blood pressure and lipid profile in hypertensive patients. J Hum Hypertens 1994; 8:283–288.
- Villamil A, Chrysant SG, Calhoun D, et al. Renin inhibition with aliskiren provides additive antihypertensive efficacy when used in combination with hydrochlorothiazide. J Hypertens 2007; 25:217–226.
- McGill JB, Reilly PA. Telmisartan plus hydrochlorothiazide versus telmisartan or hydrochlorothiazide monotherapy in patients with mild to moderate hypertension: a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial. Clin Ther 2001; 23:833–850.
- Weir MR, Weber MA, Punzi HA, Serfer HM, Rosenblatt S, Cady WJ. A dose escalation trial comparing the combination of diltiazem SR and hydrochlorothiazide with the monotherapies in patients with essential hypertension. J Hum Hypertens 1992; 6:133–138.
- Goldberg MR, Rockhold FW, Offen WW, Dornseif BE. Dose-effect and concentration-effect relationships of pinacidil and hydrochlorothiazide in hypertension. Clin Pharmacol Ther 1989; 46:208–218.
- Papademetriou V, Hainer JW, Sugg J, Munzer D; ATTACH Study Group. Factorial antihypertensive study of an extended-release metoprolol and hydrochlorothiazide combination. Am J Hypertens 2006; 19:1217–1225.
- Chrysant SG, Chrysant GS. Antihypertensive efficacy of olmesartan medoxomil alone and in combination with hydrochlorothiazide. Expert Opin Pharmacother 2004; 5:657–667.
- Kochar M, Guthrie R, Triscari J, Kassler-Taub K, Reeves RA. Matrix study of irbesartan with hydrochlorothiazide in mild-to-moderate hypertension. Am J Hypertens 1999; 12:797–805.
- Benz JR, Black HR, Graff A, Reed A, Fitzsimmons S, Shi Y. Valsartan and hydrochlorothiazide in patients with essential hypertension. A multiple dose, double-blind, placebo controlled trial comparing combination therapy with monotherapy. J Hum Hypertens 1998; 12:861–866.
- Jounela AJ, Lilja M, Lumme J, et al. Relation between low dose of hydrochlorothiazide, antihypertensive effect and adverse effects. Blood Press 1994; 3:231–235.
- Scholze J, Breitstadt A, Cairns V, et al. Short report: ramipril and hydrochlorothiazide combination therapy in hypertension: a clinical trial of factorial design. East Germany Collaborative Trial Group. J Hypertens 1993; 11:217–221.
- Canter D, Frank GJ, Knapp LE, Phelps M, Quade M, Texter M. Quinapril and hydrochlorothiazide combination for control of hypertension: assessment by factorial design. Quinapril Investigator Group. J Hum Hypertens 1994; 8:155–162.
- Vardan S, Mehrotra KG, Mookherjee S, Willsey GA, Gens JD, Green DE. Efficacy and reduced metabolic side effects of a 15-mg chlorthalidone formulation in the treatment of mild hypertension. A multicenter study. JAMA 1987; 258:484–488.
- Materson BJ, Oster JR, Michael UF, et al. Dose response to chlorthalidone in patients with mild hypertension. Efficacy of a lower dose. Clin Pharmacol Ther 1978; 24:192–198.
- Morledge JH, Ettinger B, Aranda J, et al. Isolated systolic hypertension in the elderly. A placebo-controlled, dose-response evaluation of chlorthalidone. J Am Geriatr Soc 1986; 34:199–206.
- Peterzan MA, Hardy R, Chaturvedi N, Hughes AD. Meta-analysis of dose-response relationships for hydrochlorothiazide, chlorthalidone, and bendroflumethiazide on blood pressure, serum potassium, and urate. Hypertension 2012; 59:1104–1109.
- Ernst ME, Carter BL, Goerdt CJ, et al. Comparative antihypertensive effects of hydrochlorothiazide and chlorthalidone on ambulatory and office blood pressure. Hypertension 2006; 47:352–358.
- Bakris GL, Sica D, White WB, et al. Antihypertensive efficacy of hydrochlorothiazide vs chlorthalidone combined with azilsartan medoxomil. Am J Med 2012; 25:1229.e1–1229.e10.
- Ernst ME, Carter BL, Zheng S, Grimm RH Jr. Meta-analysis of dose-response characteristics of hydrochlorothiazide and chlorthalidone: effects on systolic blood pressure and potassium. Am J Hypertens 2010; 23:440–446.
- Carter BL, Ernst ME, Cohen JD. Hydrochlorothiazide versus chlorthalidone: evidence supporting their interchangeability. Hypertension 2004; 43:4–9.
- Alderman MH, Piller LB, Ford CE, et al; Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial Collaborative Research Group. Clinical significance of incident hypokalemia and hyperkalemia in treated hypertensive patients in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Hypertension 2012; 59:926–933.
- Franse LV, Pahor M, Di Bari M, Somes GW, Cushman WC, Applegate WB. Hypokalemia associated with diuretic use and cardiovascular events in the Systolic Hypertension in the Elderly Program. Hypertension 2000; 35:1025–1030.
- Egom EE, Chirico D, Clark AL. A review of thiazide-induced hyponatraemia. Clin Med 2011; 11:448–451.
- Palmer BF. Metabolic complications associated with use of diuretics. Semin Nephrol 2011; 31:542–552.
- Hueskes BA, Roovers EA, Mantel-Teeuwisse AK, Janssens HJ, van de Lisdonk EH, Janssen M. Use of diuretics and the risk of gouty arthritis: a systematic review. Semin Arthritis Rheum 2012; 41:879–889.
- Dorsch MP, Gillespie BW, Erickson SR, Bleske BE, Weder AB. Chlorthalidone reduces cardiovascular events compared with hydrochlorothiazide: a retrospective cohort analysis. Hypertension 2011; 57:689–694.
- Trinkley KE, Page RL 2nd, Lien H, Yamanouye K, Tisdale JE. QT interval prolongation and the risk of torsades de pointes: essentials for clinicians. Curr Med Res Opin 2013; 29:1719–1726.
- Crist LW, Dixon DL. Considerations for dofetilide use in the elderly. Consult Pharm 2014; 29:270–274.
- Handler J. Lithium and antihypertensive medication: a potentially dangerous interaction. J Clin Hypertens (Greenwich) 2009; 11:738–742.
- Pavlicević I, Kuzmanić M, Rumboldt M, Rumboldt Z. Interaction between antihypertensives and NSAIDs in primary care: a controlled trial. Can J Clin Pharmacol 2008; 15:e372–e382.
- Psaty BM, Smith NL, Siscovick DS, et al. Health outcomes associated with antihypertensive therapies used as first-line agents. A systematic review and meta-analysis. JAMA 1997; 277:739–745.
- Roush GC, Holford TR, Guddati AK. Chlorthalidone compared with hydrochlorothiazide in reducing cardiovascular events: systematic review and network meta-analyses. Hypertension 2012; 59:1110–1117.
- Dhalla IA, Gomes T, Yao Z, et al. Chlorthalidone versus hydrochlorothiazide for the treatment of hypertension in older adults: a population-based cohort study. Ann Intern Med 2013; 158:447–455.
The thiazide diuretic hydrochlorothiazide and the thiazidelike diuretic chlorthalidone are two old drugs that are still useful. Although similar, they differ in important ways still not fully appreciated more than a half century after they were introduced.
Most hypertension guidelines recommend thiazide diuretics as one of the classes of agents that can be used either as initial antihypertensive drug therapy or as part of combination therapy.1–3
In the United States, hydrochlorothiazide is used more often than chlorthalidone, but many clinical trials of antihypertensive therapy have used chlorthalidone.4,5 In recent years, particularly after the publication of the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), interest in chlorthalidone has been increasing, and new data are now available comparing these two diuretics.6 While current US guidelines do not recommend one over the other, British guidelines prefer chlorthalidone.7
This review summarizes the data comparing the two drugs’ pharmacology, antihypertensive effect, and impact on clinical outcomes to help guide clinicians in choosing antihypertensive drug therapy.
PHARMACOLOGY AND MECHANISM OF ACTION
Many of the differences in effectiveness and adverse effects of hydrochlorothiazide and chlorthalidone are thought to be due to their different pharmacodynamic and pharmacokinetic effects.
Pharmacodynamic effects
Hydrochlorothiazide and chlorthalidone differ significantly in chemical structure (Figure 1), but both contain a sulfonamide group that inhibits carbonic anhydrase activity, which may be associated with lower vascular contractility. Both drugs are concentrated in the kidney and secreted into the tubular lumen8; therefore, their therapeutic diuretic effects are often achieved with relatively low plasma concentrations.
Both drugs inhibit the sodium-chloride cotransporter in the luminal membrane of the distal convoluted tubule of the ascending loop of Henle, leading to a modest natriuresis and diuresis. The exact mechanism by which they lower blood pressure is not known: while the initial response is from diuresis and volume changes, long-term reduction in blood pressure is through uncertain mechanisms. In addition, chlorthalidone may have beneficial effects on endothelial function and oxidative stress.9,10
Both drugs also increase secretion of potassium and hydrogen ions and promote increased reabsorption of calcium through increased expression of a sodium-calcium exchange channel.8 Chlorthalidone may cause more inhibition of carbonic anhydrase than hydrochlorothiazide, which can lead to lower intracellular pH and cell volume. This effect may in part explain a pleiotropic effect of chlorthalidone, ie, inhibition of platelet function, which in turn may contribute to this drug’s beneficial effect on cardiovascular outcomes.9
Pharmacokinetic differences
Hydrochlorothiazide and chlorthalidone have important differences in their pharmacokinetic properties (Table 1).11
Hydrochlorothiazide has its onset of action in about 2 hours, and it reaches its peak in 4 to 6 hours. Though its duration of action is short—up to 12 hours—its pharmacodynamic response can be much longer than predicted by its kinetics, allowing once-daily dosing.8
Chlorthalidone has a longer duration of action than hydrochlorothiazide. This may be because it has a very high volume of distribution, since it is taken up into red blood cells and is bound to carbonic anhydrase.12 This may result in a “drug reservoir” that keeps drug levels higher for a longer time.13 Its long duration of action makes it a favorable choice for patients who have difficulty adhering to medication instructions. In addition, a missed dose is unlikely to have a “rebound” effect like that seen with some other antihypertensive agents. However, both chlorthalidone and hydrochlorothiazide are effective if taken once daily.
BLOOD PRESSURE-LOWERING
Both hydrochlorothiazide and chlorthalidone are effective antihypertensive agents. Table 2 summarizes findings from studies that evaluated their blood pressure-lowering effect at various doses.14–33 However, relatively few studies have directly compared these two agents’ effects on blood pressure.
Ernst et al,34 in a small study (but probably the best one to address this issue), compared chlorthalidone 12.5 mg/day (force-titrated to 25 mg/day) and hydrochlorothiazide 25 mg/day (force-titrated to 50 mg/day) in untreated hypertensive patients. After 8 weeks, ambulatory blood pressure monitoring indicated a greater reduction from baseline in systolic blood pressure with chlorthalidone 25 mg/day than with hydrochlorothiazide 50 mg/day (24-hour mean –12.4 vs –7.4 mm Hg, P = .05). Interestingly, the change in nighttime blood pressure was greater in the chlorthalidone group (–13.5 mm Hg) than in the hydrochlorothiazide group (–6.4 mm Hg; P = .009). These data suggest that at the doses studied, chlorthalidone is more effective than hydrochlorothiazide in lowering systolic blood pressure.
Bakris et al,35 using a different study design, compared the single-pill combination of azilsartan medoxomil and chlorthalidone vs coadministration of azilsartan medoxomil and hydrochlorothiazide in participants with stage 2 primary hypertension (≥ 160/100 mm Hg). Systolic blood pressure, as measured in the clinic, declined more with the chlorthalidone combination (–35.1 mm Hg) than with the hydrochlorothiazide combination (–29.5 mm Hg, mean difference –5.6 mm Hg, P < .001).
Meta-analyses also support the conclusion that chlorthalidone is more potent than hydrochlorothiazide in lowering blood pressure.35,36 Several studies have shown that chlorthalidone at the same dose is 1.5 to 2 times as potent as hydrochlorothiazide.33,36,37 Therefore, for clinical purposes, it is reasonable to consider chlorthalidone 12.5 mg daily as similar to 25 mg of hydrochlorothiazide daily.
ADVERSE EFFECTS
Electrolyte disturbances are a common adverse effect of thiazide diuretics.
Hypokalemia. All thiazide diuretics cause potassium wasting. The frequency of hypokalemia depends on the dose, frequency of administration, diet, and other pharmacologic agents used.
Two large clinical trials, the Systolic Hypertension in the Elderly Program and ALLHAT, found that chlorthalidone caused hypokalemia requiring therapy in about 6% to 8% of patients.38,39 Chlorthalidone therapy was associated with a lowering of serum potassium levels of 0.2 to 0.5 mmol/L.36 In ALLHAT, chlorthalidone was associated with a reduction in potassium levels of approximately 0.2 mmol/L after 4 years.38
All diuretics require monitoring of electrolytes, especially during the first 2 weeks of therapy. Once a steady state is reached, patients are not usually at risk of hypokalemia unless the dose is increased, extrarenal losses of potassium increase, or dietary potassium is reduced.
Other electrolyte changes. Thiazide and thiazide-like diuretics can cause other metabolic and endocrine abnormalities such as hypochloremic alkalosis, hyponatremia, and hypercalcemia.40,41 They can also cause photosensitivity and can precipitate gout.42
Observational studies have suggested that metabolic adverse effects such as hypokalemia and hyperuricemia are more common with chlorthalidone than with hydrochlorothiazide.43 It is prudent to monitor laboratory values periodically in patients on diuretic therapy.
DRUG INTERACTIONS
The drug interaction profiles of hydrochlorothiazide and chlorthalidone are also similar. The most common interactions are pharmacodynamic interactions resulting from potassium depletion caused by the diuretics.
Antiarrythymic drugs. Hypokalemia is a risk factor for arrhythmias such as torsades de pointes, and the risk is magnified with concomitant therapy with antiarrhythmic agents that prolong the QT interval independently of serum potassium concentration (eg, sotalol, dronedarone, ibutilide, propafenone). Therefore, combinations of drugs that can cause hypokalemia (eg, diuretics) and antiarrhythmic agents require vigilant monitoring of potassium and appropriate replenishment.44
Dofetilide is a class III antiarrhythmic agent and, like other antiarrhythmic drugs, carries a risk of QT prolongation and torsades de pointes, which is magnified by hypokalemia.45 In addition, dofetilide undergoes active tubular secretion in the kidney via the cation transport system, which is inhibited by hydrochlorothiazide.45 The resulting increase in plasma concentrations of dofetilide may magnify the risk of arrhythmias. Chlorthalidone has not been specifically studied in combination with dofetilide, but thiazide diuretics in general are thought to have a similar effect on tubular secretion and, therefore, should be considered similar to hydrochlorothiazide in this regard.
Digoxin. Similarly, digoxin toxicity may be enhanced in hypokalemia. As with antiarrhythmic agents, serum potassium should be carefully monitored and replenished appropriately when diuretics are used in combination with digoxin.
Lithium is reabsorbed in the proximal tubule along with sodium. Diuretics including hydrochlorothiazide and chlorthalidone that alter sodium reabsorption can also alter lithium absorption.46 When sodium reabsorption is decreased, lithium ion reabsorption is increased and may result in lithium toxicity. Although this combination is not contraindicated, monitoring of serum lithium concentrations and clinical signs and symptoms of lithium toxicity is recommended during initiation and dose adjustments of thiazide diuretics.
Nonsteroidal anti-inflammatory drugs can decrease the natriuretic, diuretic, and antihypertensive effects of both hydrochlorothiazide and chlorthalidone.47
Renin-angiotensin-aldosterone system antagonists, ie, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and the renin inhibitor aliskiren, have potentially beneficial interactions with hydrochlorothiazide and chlorthalidone, producing additive decreases in blood pressure when coadministered with these diuretics. These effects may be particularly potent early in concomitant therapy and allow use of lower doses of diuretics, typically 12.5 mg of hydrochlorothiazide in combination therapy.
LONG-TERM EFFECTS ON CARDIOVASCULAR EVENTS
The long-term goal in treating hypertension is to lower the risk of cardiovascular disease. Therefore, the clinician needs to consider the effect of antihypertensive drug therapy on long-term clinical outcomes.
Antihypertensive drug therapy based on thiazide diuretics has been shown to lower cardiovascular risk when compared with placebo.48 In addition, the effect of chlorthalidone-based antihypertensive therapy was similar to that of other antihypertensive drug classes in preventing most cardiovascular outcomes in ALLHAT.4
However, no study has directly compared hydrochlorothiazide and chlorthalidone with the primary outcome of reduction in long-term cardiovascular events. The data to date come from observational studies and meta-analyses. For example, in a retrospective analysis of the Multiple Risk Factor Intervention Trial, cardiovascular events were significantly fewer in those receiving chlorthalidone vs hydrochlorothiazide (P = .0016).43
In a systematic review and meta-analysis, chlorthalidone was associated with a 23% lower risk of heart failure and a 21% lower risk of all cardiovascular events.49
However, a Canadian observational study of 29,873 patients found no difference in the composite outcome of death or hospitalization for heart failure, stroke, or myocardial infarction between chlorthalidone recipients (3.2 events per 100 person-years) and hydrochlorothiazide recipients (3.4 events per 100 person-years; adjusted hazard ratio 0.93, 95% confidence interval 0.81–1.06).50
In summary, it is unclear whether chlorthalidone or hydrochlorothiazide is superior in preventing cardiovascular events.
SUMMARY
Thiazide and thiazidelike diuretics play an important role in managing hypertension in most patients. The eighth Joint National Committee guidelines do not recommend either hydrochlorothiazide or chlorthalidone over the other. The target dose recommendations are hydrochlorothiazide 25 to 50 mg or chlorthalidone 12.5 to 25 mg daily, with lower doses considered for the elderly.
There are important differences between hydrochlorothiazide and chlorthalidone in pharmacology, potency, and frequency of metabolic side effects. Clinicians should consider these factors to tailor the choice of thiazide diuretic therapy in hypertensive patients.
The thiazide diuretic hydrochlorothiazide and the thiazidelike diuretic chlorthalidone are two old drugs that are still useful. Although similar, they differ in important ways still not fully appreciated more than a half century after they were introduced.
Most hypertension guidelines recommend thiazide diuretics as one of the classes of agents that can be used either as initial antihypertensive drug therapy or as part of combination therapy.1–3
In the United States, hydrochlorothiazide is used more often than chlorthalidone, but many clinical trials of antihypertensive therapy have used chlorthalidone.4,5 In recent years, particularly after the publication of the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), interest in chlorthalidone has been increasing, and new data are now available comparing these two diuretics.6 While current US guidelines do not recommend one over the other, British guidelines prefer chlorthalidone.7
This review summarizes the data comparing the two drugs’ pharmacology, antihypertensive effect, and impact on clinical outcomes to help guide clinicians in choosing antihypertensive drug therapy.
PHARMACOLOGY AND MECHANISM OF ACTION
Many of the differences in effectiveness and adverse effects of hydrochlorothiazide and chlorthalidone are thought to be due to their different pharmacodynamic and pharmacokinetic effects.
Pharmacodynamic effects
Hydrochlorothiazide and chlorthalidone differ significantly in chemical structure (Figure 1), but both contain a sulfonamide group that inhibits carbonic anhydrase activity, which may be associated with lower vascular contractility. Both drugs are concentrated in the kidney and secreted into the tubular lumen8; therefore, their therapeutic diuretic effects are often achieved with relatively low plasma concentrations.
Both drugs inhibit the sodium-chloride cotransporter in the luminal membrane of the distal convoluted tubule of the ascending loop of Henle, leading to a modest natriuresis and diuresis. The exact mechanism by which they lower blood pressure is not known: while the initial response is from diuresis and volume changes, long-term reduction in blood pressure is through uncertain mechanisms. In addition, chlorthalidone may have beneficial effects on endothelial function and oxidative stress.9,10
Both drugs also increase secretion of potassium and hydrogen ions and promote increased reabsorption of calcium through increased expression of a sodium-calcium exchange channel.8 Chlorthalidone may cause more inhibition of carbonic anhydrase than hydrochlorothiazide, which can lead to lower intracellular pH and cell volume. This effect may in part explain a pleiotropic effect of chlorthalidone, ie, inhibition of platelet function, which in turn may contribute to this drug’s beneficial effect on cardiovascular outcomes.9
Pharmacokinetic differences
Hydrochlorothiazide and chlorthalidone have important differences in their pharmacokinetic properties (Table 1).11
Hydrochlorothiazide has its onset of action in about 2 hours, and it reaches its peak in 4 to 6 hours. Though its duration of action is short—up to 12 hours—its pharmacodynamic response can be much longer than predicted by its kinetics, allowing once-daily dosing.8
Chlorthalidone has a longer duration of action than hydrochlorothiazide. This may be because it has a very high volume of distribution, since it is taken up into red blood cells and is bound to carbonic anhydrase.12 This may result in a “drug reservoir” that keeps drug levels higher for a longer time.13 Its long duration of action makes it a favorable choice for patients who have difficulty adhering to medication instructions. In addition, a missed dose is unlikely to have a “rebound” effect like that seen with some other antihypertensive agents. However, both chlorthalidone and hydrochlorothiazide are effective if taken once daily.
BLOOD PRESSURE-LOWERING
Both hydrochlorothiazide and chlorthalidone are effective antihypertensive agents. Table 2 summarizes findings from studies that evaluated their blood pressure-lowering effect at various doses.14–33 However, relatively few studies have directly compared these two agents’ effects on blood pressure.
Ernst et al,34 in a small study (but probably the best one to address this issue), compared chlorthalidone 12.5 mg/day (force-titrated to 25 mg/day) and hydrochlorothiazide 25 mg/day (force-titrated to 50 mg/day) in untreated hypertensive patients. After 8 weeks, ambulatory blood pressure monitoring indicated a greater reduction from baseline in systolic blood pressure with chlorthalidone 25 mg/day than with hydrochlorothiazide 50 mg/day (24-hour mean –12.4 vs –7.4 mm Hg, P = .05). Interestingly, the change in nighttime blood pressure was greater in the chlorthalidone group (–13.5 mm Hg) than in the hydrochlorothiazide group (–6.4 mm Hg; P = .009). These data suggest that at the doses studied, chlorthalidone is more effective than hydrochlorothiazide in lowering systolic blood pressure.
Bakris et al,35 using a different study design, compared the single-pill combination of azilsartan medoxomil and chlorthalidone vs coadministration of azilsartan medoxomil and hydrochlorothiazide in participants with stage 2 primary hypertension (≥ 160/100 mm Hg). Systolic blood pressure, as measured in the clinic, declined more with the chlorthalidone combination (–35.1 mm Hg) than with the hydrochlorothiazide combination (–29.5 mm Hg, mean difference –5.6 mm Hg, P < .001).
Meta-analyses also support the conclusion that chlorthalidone is more potent than hydrochlorothiazide in lowering blood pressure.35,36 Several studies have shown that chlorthalidone at the same dose is 1.5 to 2 times as potent as hydrochlorothiazide.33,36,37 Therefore, for clinical purposes, it is reasonable to consider chlorthalidone 12.5 mg daily as similar to 25 mg of hydrochlorothiazide daily.
ADVERSE EFFECTS
Electrolyte disturbances are a common adverse effect of thiazide diuretics.
Hypokalemia. All thiazide diuretics cause potassium wasting. The frequency of hypokalemia depends on the dose, frequency of administration, diet, and other pharmacologic agents used.
Two large clinical trials, the Systolic Hypertension in the Elderly Program and ALLHAT, found that chlorthalidone caused hypokalemia requiring therapy in about 6% to 8% of patients.38,39 Chlorthalidone therapy was associated with a lowering of serum potassium levels of 0.2 to 0.5 mmol/L.36 In ALLHAT, chlorthalidone was associated with a reduction in potassium levels of approximately 0.2 mmol/L after 4 years.38
All diuretics require monitoring of electrolytes, especially during the first 2 weeks of therapy. Once a steady state is reached, patients are not usually at risk of hypokalemia unless the dose is increased, extrarenal losses of potassium increase, or dietary potassium is reduced.
Other electrolyte changes. Thiazide and thiazide-like diuretics can cause other metabolic and endocrine abnormalities such as hypochloremic alkalosis, hyponatremia, and hypercalcemia.40,41 They can also cause photosensitivity and can precipitate gout.42
Observational studies have suggested that metabolic adverse effects such as hypokalemia and hyperuricemia are more common with chlorthalidone than with hydrochlorothiazide.43 It is prudent to monitor laboratory values periodically in patients on diuretic therapy.
DRUG INTERACTIONS
The drug interaction profiles of hydrochlorothiazide and chlorthalidone are also similar. The most common interactions are pharmacodynamic interactions resulting from potassium depletion caused by the diuretics.
Antiarrythymic drugs. Hypokalemia is a risk factor for arrhythmias such as torsades de pointes, and the risk is magnified with concomitant therapy with antiarrhythmic agents that prolong the QT interval independently of serum potassium concentration (eg, sotalol, dronedarone, ibutilide, propafenone). Therefore, combinations of drugs that can cause hypokalemia (eg, diuretics) and antiarrhythmic agents require vigilant monitoring of potassium and appropriate replenishment.44
Dofetilide is a class III antiarrhythmic agent and, like other antiarrhythmic drugs, carries a risk of QT prolongation and torsades de pointes, which is magnified by hypokalemia.45 In addition, dofetilide undergoes active tubular secretion in the kidney via the cation transport system, which is inhibited by hydrochlorothiazide.45 The resulting increase in plasma concentrations of dofetilide may magnify the risk of arrhythmias. Chlorthalidone has not been specifically studied in combination with dofetilide, but thiazide diuretics in general are thought to have a similar effect on tubular secretion and, therefore, should be considered similar to hydrochlorothiazide in this regard.
Digoxin. Similarly, digoxin toxicity may be enhanced in hypokalemia. As with antiarrhythmic agents, serum potassium should be carefully monitored and replenished appropriately when diuretics are used in combination with digoxin.
Lithium is reabsorbed in the proximal tubule along with sodium. Diuretics including hydrochlorothiazide and chlorthalidone that alter sodium reabsorption can also alter lithium absorption.46 When sodium reabsorption is decreased, lithium ion reabsorption is increased and may result in lithium toxicity. Although this combination is not contraindicated, monitoring of serum lithium concentrations and clinical signs and symptoms of lithium toxicity is recommended during initiation and dose adjustments of thiazide diuretics.
Nonsteroidal anti-inflammatory drugs can decrease the natriuretic, diuretic, and antihypertensive effects of both hydrochlorothiazide and chlorthalidone.47
Renin-angiotensin-aldosterone system antagonists, ie, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and the renin inhibitor aliskiren, have potentially beneficial interactions with hydrochlorothiazide and chlorthalidone, producing additive decreases in blood pressure when coadministered with these diuretics. These effects may be particularly potent early in concomitant therapy and allow use of lower doses of diuretics, typically 12.5 mg of hydrochlorothiazide in combination therapy.
LONG-TERM EFFECTS ON CARDIOVASCULAR EVENTS
The long-term goal in treating hypertension is to lower the risk of cardiovascular disease. Therefore, the clinician needs to consider the effect of antihypertensive drug therapy on long-term clinical outcomes.
Antihypertensive drug therapy based on thiazide diuretics has been shown to lower cardiovascular risk when compared with placebo.48 In addition, the effect of chlorthalidone-based antihypertensive therapy was similar to that of other antihypertensive drug classes in preventing most cardiovascular outcomes in ALLHAT.4
However, no study has directly compared hydrochlorothiazide and chlorthalidone with the primary outcome of reduction in long-term cardiovascular events. The data to date come from observational studies and meta-analyses. For example, in a retrospective analysis of the Multiple Risk Factor Intervention Trial, cardiovascular events were significantly fewer in those receiving chlorthalidone vs hydrochlorothiazide (P = .0016).43
In a systematic review and meta-analysis, chlorthalidone was associated with a 23% lower risk of heart failure and a 21% lower risk of all cardiovascular events.49
However, a Canadian observational study of 29,873 patients found no difference in the composite outcome of death or hospitalization for heart failure, stroke, or myocardial infarction between chlorthalidone recipients (3.2 events per 100 person-years) and hydrochlorothiazide recipients (3.4 events per 100 person-years; adjusted hazard ratio 0.93, 95% confidence interval 0.81–1.06).50
In summary, it is unclear whether chlorthalidone or hydrochlorothiazide is superior in preventing cardiovascular events.
SUMMARY
Thiazide and thiazidelike diuretics play an important role in managing hypertension in most patients. The eighth Joint National Committee guidelines do not recommend either hydrochlorothiazide or chlorthalidone over the other. The target dose recommendations are hydrochlorothiazide 25 to 50 mg or chlorthalidone 12.5 to 25 mg daily, with lower doses considered for the elderly.
There are important differences between hydrochlorothiazide and chlorthalidone in pharmacology, potency, and frequency of metabolic side effects. Clinicians should consider these factors to tailor the choice of thiazide diuretic therapy in hypertensive patients.
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014; 311:507–520.
- Dasgupta K, Quinn RR, Zarnke KB, et al; Canadian Hypertension Education Program. The 2014 Canadian Hypertension Education Program recommendations for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol 2014; 30:485–501.
- Mancia G, Fagard R, Narkiewicz K, et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 2013; 34:2159–2219.
- ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group; The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002; 288:2981–2997.
- Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA 1991; 265:3255–3264.
- Roush GC, Kaur R, Ernst ME. Diuretics: a review and update. J Cardiovasc Pharmacol Ther 2014; 19:5–13.
- McCormack T, Krause T, O’Flynn N. Management of hypertension in adults in primary care: NICE guideline. Br J Gen Pract 2012; 62:163–164.
- Bhattacharaya M, Alper SL. Pharmacology of volume regulation. In: Golan DE, Tashjian AH Jr, Armstrong EJ, Armstrong AW, editors. Principles of Pharmacology: The pathophysiologic Basis of Drug Therapy. 3rd ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2012:332–352.
- Woodman R, Brown C, Lockette W. Chlorthalidone decreases platelet aggregation and vascular permeability and promotes angiogenesis. Hypertension 2010; 56:463–470.
- Sato K, Dohi Y, Kojima M, Takase H, Suzuki S, Ito S. Antioxidative effects of thiazide diuretics in refractory hypertensive patients. A randomized crossover trial of chlortalidone and trichlormethiazide. Arzneimittelforschung 2010; 60:612–616.
- US National Library of Medicine. Dailymed. dailymed.nlm.nih.gov. Accessed May 14, 2015.
- Collste P, Garle M, Rawlins MD, Sjöqvist F. Interindividual differences in chlorthalidone concentration in plasma and red cells of man after single and multiple doses. Eur J Clin Pharmacol 1976; 9:319–325.
- Roush GC, Buddharaju V, Ernst ME, Holford TR. Chlorthalidone: mechanisms of action and effect on cardiovascular events. Curr Hypertens Rep 2013; 15:514–521.
- Pool JL, Cushman WC, Saini RK, Nwachuku CE, Battikha JP. Use of the factorial design and quadratic response surface models to evaluate the fosinopril and hydrochlorothiazide combination therapy in hypertension. Am J Hypertens 1997; 10:117–123.
- Pool JL, Glazer R, Weinberger M, Alvarado R, Huang J, Graff A. Comparison of valsartan/hydrochlorothiazide combination therapy at doses up to 320/25 mg versus monotherapy: a double-blind, placebo-controlled study followed by long-term combination therapy in hypertensive adults. Clin Ther 2007; 29:61–73.
- Horie Y, Higaki J, Takeuchi M. Design, statistical analysis and sample size calculation of dose response study of telmisartan and hydrochlorothiazide. Contemp Clin Trials 2007; 28:647–653.
- Chrysant SG. Antihypertensive effectiveness of low-dose lisinopril-hydrochlorothiazide combination. A large multicenter study. Lisinopril-Hydrochlorothiazide Group. Arch Intern Med 1994; 154:737–743.
- Lacourcière Y, Arnott W. Placebo-controlled comparison of the effects of nebivolol and low-dose hydrochlorothiazide as monotherapies and in combination on blood pressure and lipid profile in hypertensive patients. J Hum Hypertens 1994; 8:283–288.
- Villamil A, Chrysant SG, Calhoun D, et al. Renin inhibition with aliskiren provides additive antihypertensive efficacy when used in combination with hydrochlorothiazide. J Hypertens 2007; 25:217–226.
- McGill JB, Reilly PA. Telmisartan plus hydrochlorothiazide versus telmisartan or hydrochlorothiazide monotherapy in patients with mild to moderate hypertension: a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial. Clin Ther 2001; 23:833–850.
- Weir MR, Weber MA, Punzi HA, Serfer HM, Rosenblatt S, Cady WJ. A dose escalation trial comparing the combination of diltiazem SR and hydrochlorothiazide with the monotherapies in patients with essential hypertension. J Hum Hypertens 1992; 6:133–138.
- Goldberg MR, Rockhold FW, Offen WW, Dornseif BE. Dose-effect and concentration-effect relationships of pinacidil and hydrochlorothiazide in hypertension. Clin Pharmacol Ther 1989; 46:208–218.
- Papademetriou V, Hainer JW, Sugg J, Munzer D; ATTACH Study Group. Factorial antihypertensive study of an extended-release metoprolol and hydrochlorothiazide combination. Am J Hypertens 2006; 19:1217–1225.
- Chrysant SG, Chrysant GS. Antihypertensive efficacy of olmesartan medoxomil alone and in combination with hydrochlorothiazide. Expert Opin Pharmacother 2004; 5:657–667.
- Kochar M, Guthrie R, Triscari J, Kassler-Taub K, Reeves RA. Matrix study of irbesartan with hydrochlorothiazide in mild-to-moderate hypertension. Am J Hypertens 1999; 12:797–805.
- Benz JR, Black HR, Graff A, Reed A, Fitzsimmons S, Shi Y. Valsartan and hydrochlorothiazide in patients with essential hypertension. A multiple dose, double-blind, placebo controlled trial comparing combination therapy with monotherapy. J Hum Hypertens 1998; 12:861–866.
- Jounela AJ, Lilja M, Lumme J, et al. Relation between low dose of hydrochlorothiazide, antihypertensive effect and adverse effects. Blood Press 1994; 3:231–235.
- Scholze J, Breitstadt A, Cairns V, et al. Short report: ramipril and hydrochlorothiazide combination therapy in hypertension: a clinical trial of factorial design. East Germany Collaborative Trial Group. J Hypertens 1993; 11:217–221.
- Canter D, Frank GJ, Knapp LE, Phelps M, Quade M, Texter M. Quinapril and hydrochlorothiazide combination for control of hypertension: assessment by factorial design. Quinapril Investigator Group. J Hum Hypertens 1994; 8:155–162.
- Vardan S, Mehrotra KG, Mookherjee S, Willsey GA, Gens JD, Green DE. Efficacy and reduced metabolic side effects of a 15-mg chlorthalidone formulation in the treatment of mild hypertension. A multicenter study. JAMA 1987; 258:484–488.
- Materson BJ, Oster JR, Michael UF, et al. Dose response to chlorthalidone in patients with mild hypertension. Efficacy of a lower dose. Clin Pharmacol Ther 1978; 24:192–198.
- Morledge JH, Ettinger B, Aranda J, et al. Isolated systolic hypertension in the elderly. A placebo-controlled, dose-response evaluation of chlorthalidone. J Am Geriatr Soc 1986; 34:199–206.
- Peterzan MA, Hardy R, Chaturvedi N, Hughes AD. Meta-analysis of dose-response relationships for hydrochlorothiazide, chlorthalidone, and bendroflumethiazide on blood pressure, serum potassium, and urate. Hypertension 2012; 59:1104–1109.
- Ernst ME, Carter BL, Goerdt CJ, et al. Comparative antihypertensive effects of hydrochlorothiazide and chlorthalidone on ambulatory and office blood pressure. Hypertension 2006; 47:352–358.
- Bakris GL, Sica D, White WB, et al. Antihypertensive efficacy of hydrochlorothiazide vs chlorthalidone combined with azilsartan medoxomil. Am J Med 2012; 25:1229.e1–1229.e10.
- Ernst ME, Carter BL, Zheng S, Grimm RH Jr. Meta-analysis of dose-response characteristics of hydrochlorothiazide and chlorthalidone: effects on systolic blood pressure and potassium. Am J Hypertens 2010; 23:440–446.
- Carter BL, Ernst ME, Cohen JD. Hydrochlorothiazide versus chlorthalidone: evidence supporting their interchangeability. Hypertension 2004; 43:4–9.
- Alderman MH, Piller LB, Ford CE, et al; Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial Collaborative Research Group. Clinical significance of incident hypokalemia and hyperkalemia in treated hypertensive patients in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Hypertension 2012; 59:926–933.
- Franse LV, Pahor M, Di Bari M, Somes GW, Cushman WC, Applegate WB. Hypokalemia associated with diuretic use and cardiovascular events in the Systolic Hypertension in the Elderly Program. Hypertension 2000; 35:1025–1030.
- Egom EE, Chirico D, Clark AL. A review of thiazide-induced hyponatraemia. Clin Med 2011; 11:448–451.
- Palmer BF. Metabolic complications associated with use of diuretics. Semin Nephrol 2011; 31:542–552.
- Hueskes BA, Roovers EA, Mantel-Teeuwisse AK, Janssens HJ, van de Lisdonk EH, Janssen M. Use of diuretics and the risk of gouty arthritis: a systematic review. Semin Arthritis Rheum 2012; 41:879–889.
- Dorsch MP, Gillespie BW, Erickson SR, Bleske BE, Weder AB. Chlorthalidone reduces cardiovascular events compared with hydrochlorothiazide: a retrospective cohort analysis. Hypertension 2011; 57:689–694.
- Trinkley KE, Page RL 2nd, Lien H, Yamanouye K, Tisdale JE. QT interval prolongation and the risk of torsades de pointes: essentials for clinicians. Curr Med Res Opin 2013; 29:1719–1726.
- Crist LW, Dixon DL. Considerations for dofetilide use in the elderly. Consult Pharm 2014; 29:270–274.
- Handler J. Lithium and antihypertensive medication: a potentially dangerous interaction. J Clin Hypertens (Greenwich) 2009; 11:738–742.
- Pavlicević I, Kuzmanić M, Rumboldt M, Rumboldt Z. Interaction between antihypertensives and NSAIDs in primary care: a controlled trial. Can J Clin Pharmacol 2008; 15:e372–e382.
- Psaty BM, Smith NL, Siscovick DS, et al. Health outcomes associated with antihypertensive therapies used as first-line agents. A systematic review and meta-analysis. JAMA 1997; 277:739–745.
- Roush GC, Holford TR, Guddati AK. Chlorthalidone compared with hydrochlorothiazide in reducing cardiovascular events: systematic review and network meta-analyses. Hypertension 2012; 59:1110–1117.
- Dhalla IA, Gomes T, Yao Z, et al. Chlorthalidone versus hydrochlorothiazide for the treatment of hypertension in older adults: a population-based cohort study. Ann Intern Med 2013; 158:447–455.
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 2014; 311:507–520.
- Dasgupta K, Quinn RR, Zarnke KB, et al; Canadian Hypertension Education Program. The 2014 Canadian Hypertension Education Program recommendations for blood pressure measurement, diagnosis, assessment of risk, prevention, and treatment of hypertension. Can J Cardiol 2014; 30:485–501.
- Mancia G, Fagard R, Narkiewicz K, et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 2013; 34:2159–2219.
- ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group; The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002; 288:2981–2997.
- Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA 1991; 265:3255–3264.
- Roush GC, Kaur R, Ernst ME. Diuretics: a review and update. J Cardiovasc Pharmacol Ther 2014; 19:5–13.
- McCormack T, Krause T, O’Flynn N. Management of hypertension in adults in primary care: NICE guideline. Br J Gen Pract 2012; 62:163–164.
- Bhattacharaya M, Alper SL. Pharmacology of volume regulation. In: Golan DE, Tashjian AH Jr, Armstrong EJ, Armstrong AW, editors. Principles of Pharmacology: The pathophysiologic Basis of Drug Therapy. 3rd ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2012:332–352.
- Woodman R, Brown C, Lockette W. Chlorthalidone decreases platelet aggregation and vascular permeability and promotes angiogenesis. Hypertension 2010; 56:463–470.
- Sato K, Dohi Y, Kojima M, Takase H, Suzuki S, Ito S. Antioxidative effects of thiazide diuretics in refractory hypertensive patients. A randomized crossover trial of chlortalidone and trichlormethiazide. Arzneimittelforschung 2010; 60:612–616.
- US National Library of Medicine. Dailymed. dailymed.nlm.nih.gov. Accessed May 14, 2015.
- Collste P, Garle M, Rawlins MD, Sjöqvist F. Interindividual differences in chlorthalidone concentration in plasma and red cells of man after single and multiple doses. Eur J Clin Pharmacol 1976; 9:319–325.
- Roush GC, Buddharaju V, Ernst ME, Holford TR. Chlorthalidone: mechanisms of action and effect on cardiovascular events. Curr Hypertens Rep 2013; 15:514–521.
- Pool JL, Cushman WC, Saini RK, Nwachuku CE, Battikha JP. Use of the factorial design and quadratic response surface models to evaluate the fosinopril and hydrochlorothiazide combination therapy in hypertension. Am J Hypertens 1997; 10:117–123.
- Pool JL, Glazer R, Weinberger M, Alvarado R, Huang J, Graff A. Comparison of valsartan/hydrochlorothiazide combination therapy at doses up to 320/25 mg versus monotherapy: a double-blind, placebo-controlled study followed by long-term combination therapy in hypertensive adults. Clin Ther 2007; 29:61–73.
- Horie Y, Higaki J, Takeuchi M. Design, statistical analysis and sample size calculation of dose response study of telmisartan and hydrochlorothiazide. Contemp Clin Trials 2007; 28:647–653.
- Chrysant SG. Antihypertensive effectiveness of low-dose lisinopril-hydrochlorothiazide combination. A large multicenter study. Lisinopril-Hydrochlorothiazide Group. Arch Intern Med 1994; 154:737–743.
- Lacourcière Y, Arnott W. Placebo-controlled comparison of the effects of nebivolol and low-dose hydrochlorothiazide as monotherapies and in combination on blood pressure and lipid profile in hypertensive patients. J Hum Hypertens 1994; 8:283–288.
- Villamil A, Chrysant SG, Calhoun D, et al. Renin inhibition with aliskiren provides additive antihypertensive efficacy when used in combination with hydrochlorothiazide. J Hypertens 2007; 25:217–226.
- McGill JB, Reilly PA. Telmisartan plus hydrochlorothiazide versus telmisartan or hydrochlorothiazide monotherapy in patients with mild to moderate hypertension: a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial. Clin Ther 2001; 23:833–850.
- Weir MR, Weber MA, Punzi HA, Serfer HM, Rosenblatt S, Cady WJ. A dose escalation trial comparing the combination of diltiazem SR and hydrochlorothiazide with the monotherapies in patients with essential hypertension. J Hum Hypertens 1992; 6:133–138.
- Goldberg MR, Rockhold FW, Offen WW, Dornseif BE. Dose-effect and concentration-effect relationships of pinacidil and hydrochlorothiazide in hypertension. Clin Pharmacol Ther 1989; 46:208–218.
- Papademetriou V, Hainer JW, Sugg J, Munzer D; ATTACH Study Group. Factorial antihypertensive study of an extended-release metoprolol and hydrochlorothiazide combination. Am J Hypertens 2006; 19:1217–1225.
- Chrysant SG, Chrysant GS. Antihypertensive efficacy of olmesartan medoxomil alone and in combination with hydrochlorothiazide. Expert Opin Pharmacother 2004; 5:657–667.
- Kochar M, Guthrie R, Triscari J, Kassler-Taub K, Reeves RA. Matrix study of irbesartan with hydrochlorothiazide in mild-to-moderate hypertension. Am J Hypertens 1999; 12:797–805.
- Benz JR, Black HR, Graff A, Reed A, Fitzsimmons S, Shi Y. Valsartan and hydrochlorothiazide in patients with essential hypertension. A multiple dose, double-blind, placebo controlled trial comparing combination therapy with monotherapy. J Hum Hypertens 1998; 12:861–866.
- Jounela AJ, Lilja M, Lumme J, et al. Relation between low dose of hydrochlorothiazide, antihypertensive effect and adverse effects. Blood Press 1994; 3:231–235.
- Scholze J, Breitstadt A, Cairns V, et al. Short report: ramipril and hydrochlorothiazide combination therapy in hypertension: a clinical trial of factorial design. East Germany Collaborative Trial Group. J Hypertens 1993; 11:217–221.
- Canter D, Frank GJ, Knapp LE, Phelps M, Quade M, Texter M. Quinapril and hydrochlorothiazide combination for control of hypertension: assessment by factorial design. Quinapril Investigator Group. J Hum Hypertens 1994; 8:155–162.
- Vardan S, Mehrotra KG, Mookherjee S, Willsey GA, Gens JD, Green DE. Efficacy and reduced metabolic side effects of a 15-mg chlorthalidone formulation in the treatment of mild hypertension. A multicenter study. JAMA 1987; 258:484–488.
- Materson BJ, Oster JR, Michael UF, et al. Dose response to chlorthalidone in patients with mild hypertension. Efficacy of a lower dose. Clin Pharmacol Ther 1978; 24:192–198.
- Morledge JH, Ettinger B, Aranda J, et al. Isolated systolic hypertension in the elderly. A placebo-controlled, dose-response evaluation of chlorthalidone. J Am Geriatr Soc 1986; 34:199–206.
- Peterzan MA, Hardy R, Chaturvedi N, Hughes AD. Meta-analysis of dose-response relationships for hydrochlorothiazide, chlorthalidone, and bendroflumethiazide on blood pressure, serum potassium, and urate. Hypertension 2012; 59:1104–1109.
- Ernst ME, Carter BL, Goerdt CJ, et al. Comparative antihypertensive effects of hydrochlorothiazide and chlorthalidone on ambulatory and office blood pressure. Hypertension 2006; 47:352–358.
- Bakris GL, Sica D, White WB, et al. Antihypertensive efficacy of hydrochlorothiazide vs chlorthalidone combined with azilsartan medoxomil. Am J Med 2012; 25:1229.e1–1229.e10.
- Ernst ME, Carter BL, Zheng S, Grimm RH Jr. Meta-analysis of dose-response characteristics of hydrochlorothiazide and chlorthalidone: effects on systolic blood pressure and potassium. Am J Hypertens 2010; 23:440–446.
- Carter BL, Ernst ME, Cohen JD. Hydrochlorothiazide versus chlorthalidone: evidence supporting their interchangeability. Hypertension 2004; 43:4–9.
- Alderman MH, Piller LB, Ford CE, et al; Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial Collaborative Research Group. Clinical significance of incident hypokalemia and hyperkalemia in treated hypertensive patients in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Hypertension 2012; 59:926–933.
- Franse LV, Pahor M, Di Bari M, Somes GW, Cushman WC, Applegate WB. Hypokalemia associated with diuretic use and cardiovascular events in the Systolic Hypertension in the Elderly Program. Hypertension 2000; 35:1025–1030.
- Egom EE, Chirico D, Clark AL. A review of thiazide-induced hyponatraemia. Clin Med 2011; 11:448–451.
- Palmer BF. Metabolic complications associated with use of diuretics. Semin Nephrol 2011; 31:542–552.
- Hueskes BA, Roovers EA, Mantel-Teeuwisse AK, Janssens HJ, van de Lisdonk EH, Janssen M. Use of diuretics and the risk of gouty arthritis: a systematic review. Semin Arthritis Rheum 2012; 41:879–889.
- Dorsch MP, Gillespie BW, Erickson SR, Bleske BE, Weder AB. Chlorthalidone reduces cardiovascular events compared with hydrochlorothiazide: a retrospective cohort analysis. Hypertension 2011; 57:689–694.
- Trinkley KE, Page RL 2nd, Lien H, Yamanouye K, Tisdale JE. QT interval prolongation and the risk of torsades de pointes: essentials for clinicians. Curr Med Res Opin 2013; 29:1719–1726.
- Crist LW, Dixon DL. Considerations for dofetilide use in the elderly. Consult Pharm 2014; 29:270–274.
- Handler J. Lithium and antihypertensive medication: a potentially dangerous interaction. J Clin Hypertens (Greenwich) 2009; 11:738–742.
- Pavlicević I, Kuzmanić M, Rumboldt M, Rumboldt Z. Interaction between antihypertensives and NSAIDs in primary care: a controlled trial. Can J Clin Pharmacol 2008; 15:e372–e382.
- Psaty BM, Smith NL, Siscovick DS, et al. Health outcomes associated with antihypertensive therapies used as first-line agents. A systematic review and meta-analysis. JAMA 1997; 277:739–745.
- Roush GC, Holford TR, Guddati AK. Chlorthalidone compared with hydrochlorothiazide in reducing cardiovascular events: systematic review and network meta-analyses. Hypertension 2012; 59:1110–1117.
- Dhalla IA, Gomes T, Yao Z, et al. Chlorthalidone versus hydrochlorothiazide for the treatment of hypertension in older adults: a population-based cohort study. Ann Intern Med 2013; 158:447–455.
KEY POINTS
- Chlorthalidone has a longer duration of action and a longer half-life than hydrochlorothiazide.
- Chlorthalidone may be more potent than hydrochlorothiazide in lowering blood pressure, but it also may be associated with more metabolic adverse effects, such as hypokalemia.
- No study has conclusively shown either drug to be better in preventing adverse clinical outcomes.
- These differences should be considered when making choices about thiazide diuretic therapy for hypertension.
Lifestyle changes, surgical weight loss benefit NAFLD
Losing weight through lifestyle changes can significantly improve several measures of nonalcoholic fatty liver disease, particularly if patients lose at least 10% of their body weight, and bariatric surgery is a valid alternative if diet and exercise fail, according to two studies reported in the August issue of Gastroenterology (2015 Apr. 9 [doi:10.1053/j.gastro.2015.04.005]).
Global rates of nonalcoholic fatty liver disease (NAFLD) are up because of the “parallel epidemics” of obesity and type 2 diabetes mellitus, said Dr. Eduardo Vilar-Gomez of the National Institute of Hepatology in Havana, Cuba, who authored the study of lifestyle changes. There are no approved therapies for the more aggressive form of NAFLD, steatohepatitis, he and his associates said. In past studies, patients who lost about 7%-10% of their body weight substantially improved their NAFLD activity score and had reductions in steatosis, lobular inflammation, and ballooning, but the results did not extend to fibrosis, and prospective studies of the effect of lifestyle changes on histology are lacking, the researchers added .
To fill the gap, they followed 293 adults with histologically confirmed nonalcoholic steatohepatitis who completed a 52-week lifestyle intervention program that included keeping a food diary, restricting saturated fats to less than 10% of total intake, and walking at least 200 minutes a week. Patients had not received hypolipidemic treatment in the preceding 3 months and were not allowed to take insulin sensitizers or vitamin E, both of which are potentially beneficial for nonalcoholic steatohepatitis, the investigators said.
At the end of the yearlong program, steatohepatitis had resolved in 25% of patients, 47% had lower NAFLD activity scores, and 19% had regression of fibrosis, the researchers reported. Although only 30% of participants lost at least 5% of their body weight, weight loss correlated positively with resolution of steatohepatitis and with 2-point reductions in histologic activity scores (P <.001). Among patients who lost at least 10% of their body weight, 90% had resolution of steatohepatitis, 45% had regression of fibrosis, and all had improved histologic activity scores, even if they had negative risk factors such as female sex, a baseline body mass index of at least 35 kg/m2, and a baseline fasting glucose level of at least 5.5 mmol/L, the investigators added. “Our findings support the current recommendation for weight loss using lifestyle modification as the first step in the management of patients with nonalcoholic steatohepatitis,” they wrote.
But what if lifestyle changes fail? The impact of bariatric surgery on nonalcoholic steatohepatitis has not been well studied, said Dr. Guillaume Lassailly at CHRU Lille (France). He and his associates therefore followed 109 morbidly obese patients (BMI ≥40 kg/m2) with biopsy-confirmed nonalcoholic steatohepatitis who underwent bariatric surgery at a single tertiary hospital (Gastroenterology 2015 Apr. 25 [doi:10.1053/j.gastro.2015.04.014]) .
One year after surgery, 85% of patients had achieved disease resolution (95% confidence interval, 75.8%-92.2%), the investigators reported. Stratifying patients by baseline Brunt scores showed that those with milder presurgical disease were more likely to have complete resolution than were patients whose disease was severe (94% vs. 70%; P <.05), the researchers added. Histologic analyses supported the findings, revealing steatosis in 60% of presurgical tissue samples, compared with 10% of samples taken a year after surgery. In addition, average NAFLD disease scores dropped from 5 to 1 (P <.001), hepatocyte ballooning decreased in 84% of samples, lobular inflammation decreased in 67%, and Metavir fibrosis scores dropped in one-third of specimens.
Notably, BMI scores for patients with persistent postsurgical disease dropped by an average of only 9.1, compared with 12.3 for patients whose disease resolved (P = .005), said the researchers. Gastric bypass surgery achieved greater weight loss and improvements in disease status, compared with laparoscopic banding, they added. “The encouraging results of the present study suggest that bariatric surgery should be tested in multicenter, randomized controlled trials in morbidly or severely obese patients with nonalcoholic steatohepatitis who did not respond to lifestyle therapy,” they wrote.
The Cuban National Institute of Gastroenterology and Ministry of Health partially funded the study by Dr. Vilar-Gomez and his associates. The French Ministry of Health and the Conseil Regional Nord-Pas de Calais supported the work by Dr. Lassailly and his colleagues. All investigators declared having no relevant financial conflicts of interest.
Losing weight through lifestyle changes can significantly improve several measures of nonalcoholic fatty liver disease, particularly if patients lose at least 10% of their body weight, and bariatric surgery is a valid alternative if diet and exercise fail, according to two studies reported in the August issue of Gastroenterology (2015 Apr. 9 [doi:10.1053/j.gastro.2015.04.005]).
Global rates of nonalcoholic fatty liver disease (NAFLD) are up because of the “parallel epidemics” of obesity and type 2 diabetes mellitus, said Dr. Eduardo Vilar-Gomez of the National Institute of Hepatology in Havana, Cuba, who authored the study of lifestyle changes. There are no approved therapies for the more aggressive form of NAFLD, steatohepatitis, he and his associates said. In past studies, patients who lost about 7%-10% of their body weight substantially improved their NAFLD activity score and had reductions in steatosis, lobular inflammation, and ballooning, but the results did not extend to fibrosis, and prospective studies of the effect of lifestyle changes on histology are lacking, the researchers added .
To fill the gap, they followed 293 adults with histologically confirmed nonalcoholic steatohepatitis who completed a 52-week lifestyle intervention program that included keeping a food diary, restricting saturated fats to less than 10% of total intake, and walking at least 200 minutes a week. Patients had not received hypolipidemic treatment in the preceding 3 months and were not allowed to take insulin sensitizers or vitamin E, both of which are potentially beneficial for nonalcoholic steatohepatitis, the investigators said.
At the end of the yearlong program, steatohepatitis had resolved in 25% of patients, 47% had lower NAFLD activity scores, and 19% had regression of fibrosis, the researchers reported. Although only 30% of participants lost at least 5% of their body weight, weight loss correlated positively with resolution of steatohepatitis and with 2-point reductions in histologic activity scores (P <.001). Among patients who lost at least 10% of their body weight, 90% had resolution of steatohepatitis, 45% had regression of fibrosis, and all had improved histologic activity scores, even if they had negative risk factors such as female sex, a baseline body mass index of at least 35 kg/m2, and a baseline fasting glucose level of at least 5.5 mmol/L, the investigators added. “Our findings support the current recommendation for weight loss using lifestyle modification as the first step in the management of patients with nonalcoholic steatohepatitis,” they wrote.
But what if lifestyle changes fail? The impact of bariatric surgery on nonalcoholic steatohepatitis has not been well studied, said Dr. Guillaume Lassailly at CHRU Lille (France). He and his associates therefore followed 109 morbidly obese patients (BMI ≥40 kg/m2) with biopsy-confirmed nonalcoholic steatohepatitis who underwent bariatric surgery at a single tertiary hospital (Gastroenterology 2015 Apr. 25 [doi:10.1053/j.gastro.2015.04.014]) .
One year after surgery, 85% of patients had achieved disease resolution (95% confidence interval, 75.8%-92.2%), the investigators reported. Stratifying patients by baseline Brunt scores showed that those with milder presurgical disease were more likely to have complete resolution than were patients whose disease was severe (94% vs. 70%; P <.05), the researchers added. Histologic analyses supported the findings, revealing steatosis in 60% of presurgical tissue samples, compared with 10% of samples taken a year after surgery. In addition, average NAFLD disease scores dropped from 5 to 1 (P <.001), hepatocyte ballooning decreased in 84% of samples, lobular inflammation decreased in 67%, and Metavir fibrosis scores dropped in one-third of specimens.
Notably, BMI scores for patients with persistent postsurgical disease dropped by an average of only 9.1, compared with 12.3 for patients whose disease resolved (P = .005), said the researchers. Gastric bypass surgery achieved greater weight loss and improvements in disease status, compared with laparoscopic banding, they added. “The encouraging results of the present study suggest that bariatric surgery should be tested in multicenter, randomized controlled trials in morbidly or severely obese patients with nonalcoholic steatohepatitis who did not respond to lifestyle therapy,” they wrote.
The Cuban National Institute of Gastroenterology and Ministry of Health partially funded the study by Dr. Vilar-Gomez and his associates. The French Ministry of Health and the Conseil Regional Nord-Pas de Calais supported the work by Dr. Lassailly and his colleagues. All investigators declared having no relevant financial conflicts of interest.
Losing weight through lifestyle changes can significantly improve several measures of nonalcoholic fatty liver disease, particularly if patients lose at least 10% of their body weight, and bariatric surgery is a valid alternative if diet and exercise fail, according to two studies reported in the August issue of Gastroenterology (2015 Apr. 9 [doi:10.1053/j.gastro.2015.04.005]).
Global rates of nonalcoholic fatty liver disease (NAFLD) are up because of the “parallel epidemics” of obesity and type 2 diabetes mellitus, said Dr. Eduardo Vilar-Gomez of the National Institute of Hepatology in Havana, Cuba, who authored the study of lifestyle changes. There are no approved therapies for the more aggressive form of NAFLD, steatohepatitis, he and his associates said. In past studies, patients who lost about 7%-10% of their body weight substantially improved their NAFLD activity score and had reductions in steatosis, lobular inflammation, and ballooning, but the results did not extend to fibrosis, and prospective studies of the effect of lifestyle changes on histology are lacking, the researchers added .
To fill the gap, they followed 293 adults with histologically confirmed nonalcoholic steatohepatitis who completed a 52-week lifestyle intervention program that included keeping a food diary, restricting saturated fats to less than 10% of total intake, and walking at least 200 minutes a week. Patients had not received hypolipidemic treatment in the preceding 3 months and were not allowed to take insulin sensitizers or vitamin E, both of which are potentially beneficial for nonalcoholic steatohepatitis, the investigators said.
At the end of the yearlong program, steatohepatitis had resolved in 25% of patients, 47% had lower NAFLD activity scores, and 19% had regression of fibrosis, the researchers reported. Although only 30% of participants lost at least 5% of their body weight, weight loss correlated positively with resolution of steatohepatitis and with 2-point reductions in histologic activity scores (P <.001). Among patients who lost at least 10% of their body weight, 90% had resolution of steatohepatitis, 45% had regression of fibrosis, and all had improved histologic activity scores, even if they had negative risk factors such as female sex, a baseline body mass index of at least 35 kg/m2, and a baseline fasting glucose level of at least 5.5 mmol/L, the investigators added. “Our findings support the current recommendation for weight loss using lifestyle modification as the first step in the management of patients with nonalcoholic steatohepatitis,” they wrote.
But what if lifestyle changes fail? The impact of bariatric surgery on nonalcoholic steatohepatitis has not been well studied, said Dr. Guillaume Lassailly at CHRU Lille (France). He and his associates therefore followed 109 morbidly obese patients (BMI ≥40 kg/m2) with biopsy-confirmed nonalcoholic steatohepatitis who underwent bariatric surgery at a single tertiary hospital (Gastroenterology 2015 Apr. 25 [doi:10.1053/j.gastro.2015.04.014]) .
One year after surgery, 85% of patients had achieved disease resolution (95% confidence interval, 75.8%-92.2%), the investigators reported. Stratifying patients by baseline Brunt scores showed that those with milder presurgical disease were more likely to have complete resolution than were patients whose disease was severe (94% vs. 70%; P <.05), the researchers added. Histologic analyses supported the findings, revealing steatosis in 60% of presurgical tissue samples, compared with 10% of samples taken a year after surgery. In addition, average NAFLD disease scores dropped from 5 to 1 (P <.001), hepatocyte ballooning decreased in 84% of samples, lobular inflammation decreased in 67%, and Metavir fibrosis scores dropped in one-third of specimens.
Notably, BMI scores for patients with persistent postsurgical disease dropped by an average of only 9.1, compared with 12.3 for patients whose disease resolved (P = .005), said the researchers. Gastric bypass surgery achieved greater weight loss and improvements in disease status, compared with laparoscopic banding, they added. “The encouraging results of the present study suggest that bariatric surgery should be tested in multicenter, randomized controlled trials in morbidly or severely obese patients with nonalcoholic steatohepatitis who did not respond to lifestyle therapy,” they wrote.
The Cuban National Institute of Gastroenterology and Ministry of Health partially funded the study by Dr. Vilar-Gomez and his associates. The French Ministry of Health and the Conseil Regional Nord-Pas de Calais supported the work by Dr. Lassailly and his colleagues. All investigators declared having no relevant financial conflicts of interest.
FROM GASTROENTEROLOGY
Key clinical point: Losing weight through lifestyle changes or bariatric surgery can significantly improve several measures of nonalcoholic steatohepatitis.
Major finding: A yearlong diet and exercise program led to resolution of nonalcoholic steatohepatitis in 25% of patients, while bariatric surgery achieved that outcome for 85% of patients in a separate study.
Data source: Two prospective uncontrolled cohort studies of 402 total adults with nonalcoholic steatohepatitis (the more severe form of nonalcoholic fatty liver disease).
Disclosures: The Cuban National Institute of Gastroenterology and Ministry of Health partially funded the study by Dr. Vilar-Gomez and his associates. The French Ministry of Health and the Conseil Regional Nord-Pas de Calais supported the work by Dr. Lassailly and his colleagues. All investigators declared having no relevant financial conflicts of interest.
FDA approves azelaic acid foam for rosacea
A foam formulation of azelaic acid has been approved for the topical treatment of the inflammatory papules and pustules of mild to moderate rosacea, the manufacturer announced July 31.
Azelaic acid foam, 15%, was compared with a vehicle foam, applied twice a day for 12 weeks, in two studies with a total of 1,362 people with papulopustular rosacea (mean age 50.6 years) and a mean of about 21 inflammatory papules and pustules at baseline. The success rate was defined as a score of clear or minimal with at least a two-step reduction from baseline on the Investigator’s Global Assessment scale.
At 12 weeks, the success rate in the two studies, respectively, was 32% and 43% among those treated with azelaic foam vs. 23% and 32% among controls, according to the Bayer HealthCare press release announcing the approval.
The most common adverse events associated with treatment were pain at the application site in about 6%, pruritus (2.5%), dryness (0.7%), and erythema (0.7%). Warnings in the prescribing information note that treatment has been associated with “isolated” cases of hypopigmentation and can cause eye irritation, and that the contents are flammable. Patients are instructed to avoid “fire, flame, and smoking during and immediately following application.”
Azelaic acid foam is marketed as Finacea Foam by Bayer; it is the first foam formulation of azelaic acid to be approved, and it will be available in September, according to the company. A gel formulation of azelaic acid, 15%, was approved by the FDA in 2002 for topical treatment of inflammatory papules and pustules of mild to moderate rosacea.
Serious adverse events associated with azelaic acid should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch/default.htm.
A foam formulation of azelaic acid has been approved for the topical treatment of the inflammatory papules and pustules of mild to moderate rosacea, the manufacturer announced July 31.
Azelaic acid foam, 15%, was compared with a vehicle foam, applied twice a day for 12 weeks, in two studies with a total of 1,362 people with papulopustular rosacea (mean age 50.6 years) and a mean of about 21 inflammatory papules and pustules at baseline. The success rate was defined as a score of clear or minimal with at least a two-step reduction from baseline on the Investigator’s Global Assessment scale.
At 12 weeks, the success rate in the two studies, respectively, was 32% and 43% among those treated with azelaic foam vs. 23% and 32% among controls, according to the Bayer HealthCare press release announcing the approval.
The most common adverse events associated with treatment were pain at the application site in about 6%, pruritus (2.5%), dryness (0.7%), and erythema (0.7%). Warnings in the prescribing information note that treatment has been associated with “isolated” cases of hypopigmentation and can cause eye irritation, and that the contents are flammable. Patients are instructed to avoid “fire, flame, and smoking during and immediately following application.”
Azelaic acid foam is marketed as Finacea Foam by Bayer; it is the first foam formulation of azelaic acid to be approved, and it will be available in September, according to the company. A gel formulation of azelaic acid, 15%, was approved by the FDA in 2002 for topical treatment of inflammatory papules and pustules of mild to moderate rosacea.
Serious adverse events associated with azelaic acid should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch/default.htm.
A foam formulation of azelaic acid has been approved for the topical treatment of the inflammatory papules and pustules of mild to moderate rosacea, the manufacturer announced July 31.
Azelaic acid foam, 15%, was compared with a vehicle foam, applied twice a day for 12 weeks, in two studies with a total of 1,362 people with papulopustular rosacea (mean age 50.6 years) and a mean of about 21 inflammatory papules and pustules at baseline. The success rate was defined as a score of clear or minimal with at least a two-step reduction from baseline on the Investigator’s Global Assessment scale.
At 12 weeks, the success rate in the two studies, respectively, was 32% and 43% among those treated with azelaic foam vs. 23% and 32% among controls, according to the Bayer HealthCare press release announcing the approval.
The most common adverse events associated with treatment were pain at the application site in about 6%, pruritus (2.5%), dryness (0.7%), and erythema (0.7%). Warnings in the prescribing information note that treatment has been associated with “isolated” cases of hypopigmentation and can cause eye irritation, and that the contents are flammable. Patients are instructed to avoid “fire, flame, and smoking during and immediately following application.”
Azelaic acid foam is marketed as Finacea Foam by Bayer; it is the first foam formulation of azelaic acid to be approved, and it will be available in September, according to the company. A gel formulation of azelaic acid, 15%, was approved by the FDA in 2002 for topical treatment of inflammatory papules and pustules of mild to moderate rosacea.
Serious adverse events associated with azelaic acid should be reported to the FDA’s MedWatch program at 800-332-1088 or www.fda.gov/Safety/MedWatch/default.htm.
New guidelines stress identifying Lynch syndrome
Clinicians and researchers must get better at identifying Lynch syndrome because the diagnosis is so often missed, according to new AGA clinical guidelines for diagnosing and managing the disorder, published in the September issue of Gastroenterology.
Lynch syndrome, previously known as hereditary nonpolyposis colorectal cancer syndrome (HNPCC), is the most common heritable cause of colorectal cancer and also is associated with cancers of the endometrium, stomach, small intestine, pancreas, biliary tract, ovary, urinary tract, and brain. Lynch syndrome accounts for 2%-3% of all colorectal cancers in the United States, and the estimated prevalence is 1 in 440 in the general population. People with the syndrome are estimated to have a lifetime cumulative incidence of colorectal cancer approaching 80%, and affected women have an estimated 60% lifetime cumulative incidence of endometrial cancer.
The new AGA guidelines focus on identifying Lynch syndrome, both in patients without cancer who have a family history suggestive of the disorder and in all patients who develop colorectal cancer, said Dr. Joel H. Rubenstein and his associates on the clinical guidelines committee (Gastroenterology 2015 Jul. 28 [doi: 10.1053/j.gastro.2015.07.036]).
The guidelines strongly recommend that all colorectal cancers now be tested using immunohistochemistry or assessment of microsatellite instability to identify potential cases of Lynch syndrome. Given the high incidence of colorectal cancer in the United States, this recommendation in particular “may be ripe for consideration as a process measure of quality of care,” they noted.
Until now, older patients with colorectal cancer have not undergone such testing because the yield of positive results was lower than in younger patients, but now there is new appreciation that these results have a significant impact on younger family members, not just the patients themselves. From the perspective of preventing cancer in these relatives, such testing is actually cost effective, said Dr. Rubenstein of the Veterans Affairs Center for Clinical Management Research and the gastroenterology division at the University of Michigan, both in Ann Arbor, and his associates.
At present, the evidence is insufficient to recommend either of these tests above the other for identifying Lynch syndrome. The two have comparable sensitivities and specificities.
The guidelines also recommend that, in people who have no personal history of colorectal or other cancer but who have a family history that suggests Lynch syndrome, risk prediction should be performed, “rather than doing nothing.” If a first-degree relative is known to have a Lynch mutation, people should be offered germline genetic testing for that mutation. Alternatively, “if tumor tissue from an affected relative is available, the screening process should begin with testing on that tumor.”
If none of this information is available, online risk prediction models or free downloadable software incorporating such models can be used to quickly and easily estimate the probability of carrying a Lynch syndrome mutation. This approach is “imperative” to improve case finding, since it is likely that most Lynch syndrome kindreds are undiagnosed, Dr. Rubenstein and his associates said.
Such patients should be offered risk-prediction models rather than proceeding directly to germline genetic testing because of the currently high costs of genetic testing. People without cancer who have a family history suggestive of Lynch syndrome should proceed straight to germline testing if they are considered to be at high risk – for example, if they meet the highly specific Amsterdam criteria.
The AGA guidelines strongly recommend that patients identified as having Lynch syndrome undergo surveillance colonoscopy, as opposed to no surveillance. Good-quality evidence shows that this strategy decreases the overall burden of colorectal cancer and reduces disease-specific mortality. People who carry Lynch syndrome genetic mutations increase their life expectancy by 7 years if they undergo surveillance colonoscopy, and cost-effectiveness analyses indicate that the expense of such screening is lower than the expense of no screening.
The optimal screening interval for such patients has not been determined, but low-quality evidence suggests that undergoing colonoscopy every 1-2 years is the “most prudent” course and is better than doing so at longer intervals.
The guidelines also include a conditional recommendation that people found to have Lynch syndrome should be offered aspirin therapy as cancer prophylaxis. The optimal dose and frequency of aspirin use is not yet known, and there is no evidence that the treatment improves mortality, but some low-quality evidence suggests that aspirin therapy reduces the risk of colorectal and other cancers, and the risk of adverse events is quite low.
Clinicians and researchers must get better at identifying Lynch syndrome because the diagnosis is so often missed, according to new AGA clinical guidelines for diagnosing and managing the disorder, published in the September issue of Gastroenterology.
Lynch syndrome, previously known as hereditary nonpolyposis colorectal cancer syndrome (HNPCC), is the most common heritable cause of colorectal cancer and also is associated with cancers of the endometrium, stomach, small intestine, pancreas, biliary tract, ovary, urinary tract, and brain. Lynch syndrome accounts for 2%-3% of all colorectal cancers in the United States, and the estimated prevalence is 1 in 440 in the general population. People with the syndrome are estimated to have a lifetime cumulative incidence of colorectal cancer approaching 80%, and affected women have an estimated 60% lifetime cumulative incidence of endometrial cancer.
The new AGA guidelines focus on identifying Lynch syndrome, both in patients without cancer who have a family history suggestive of the disorder and in all patients who develop colorectal cancer, said Dr. Joel H. Rubenstein and his associates on the clinical guidelines committee (Gastroenterology 2015 Jul. 28 [doi: 10.1053/j.gastro.2015.07.036]).
The guidelines strongly recommend that all colorectal cancers now be tested using immunohistochemistry or assessment of microsatellite instability to identify potential cases of Lynch syndrome. Given the high incidence of colorectal cancer in the United States, this recommendation in particular “may be ripe for consideration as a process measure of quality of care,” they noted.
Until now, older patients with colorectal cancer have not undergone such testing because the yield of positive results was lower than in younger patients, but now there is new appreciation that these results have a significant impact on younger family members, not just the patients themselves. From the perspective of preventing cancer in these relatives, such testing is actually cost effective, said Dr. Rubenstein of the Veterans Affairs Center for Clinical Management Research and the gastroenterology division at the University of Michigan, both in Ann Arbor, and his associates.
At present, the evidence is insufficient to recommend either of these tests above the other for identifying Lynch syndrome. The two have comparable sensitivities and specificities.
The guidelines also recommend that, in people who have no personal history of colorectal or other cancer but who have a family history that suggests Lynch syndrome, risk prediction should be performed, “rather than doing nothing.” If a first-degree relative is known to have a Lynch mutation, people should be offered germline genetic testing for that mutation. Alternatively, “if tumor tissue from an affected relative is available, the screening process should begin with testing on that tumor.”
If none of this information is available, online risk prediction models or free downloadable software incorporating such models can be used to quickly and easily estimate the probability of carrying a Lynch syndrome mutation. This approach is “imperative” to improve case finding, since it is likely that most Lynch syndrome kindreds are undiagnosed, Dr. Rubenstein and his associates said.
Such patients should be offered risk-prediction models rather than proceeding directly to germline genetic testing because of the currently high costs of genetic testing. People without cancer who have a family history suggestive of Lynch syndrome should proceed straight to germline testing if they are considered to be at high risk – for example, if they meet the highly specific Amsterdam criteria.
The AGA guidelines strongly recommend that patients identified as having Lynch syndrome undergo surveillance colonoscopy, as opposed to no surveillance. Good-quality evidence shows that this strategy decreases the overall burden of colorectal cancer and reduces disease-specific mortality. People who carry Lynch syndrome genetic mutations increase their life expectancy by 7 years if they undergo surveillance colonoscopy, and cost-effectiveness analyses indicate that the expense of such screening is lower than the expense of no screening.
The optimal screening interval for such patients has not been determined, but low-quality evidence suggests that undergoing colonoscopy every 1-2 years is the “most prudent” course and is better than doing so at longer intervals.
The guidelines also include a conditional recommendation that people found to have Lynch syndrome should be offered aspirin therapy as cancer prophylaxis. The optimal dose and frequency of aspirin use is not yet known, and there is no evidence that the treatment improves mortality, but some low-quality evidence suggests that aspirin therapy reduces the risk of colorectal and other cancers, and the risk of adverse events is quite low.
Clinicians and researchers must get better at identifying Lynch syndrome because the diagnosis is so often missed, according to new AGA clinical guidelines for diagnosing and managing the disorder, published in the September issue of Gastroenterology.
Lynch syndrome, previously known as hereditary nonpolyposis colorectal cancer syndrome (HNPCC), is the most common heritable cause of colorectal cancer and also is associated with cancers of the endometrium, stomach, small intestine, pancreas, biliary tract, ovary, urinary tract, and brain. Lynch syndrome accounts for 2%-3% of all colorectal cancers in the United States, and the estimated prevalence is 1 in 440 in the general population. People with the syndrome are estimated to have a lifetime cumulative incidence of colorectal cancer approaching 80%, and affected women have an estimated 60% lifetime cumulative incidence of endometrial cancer.
The new AGA guidelines focus on identifying Lynch syndrome, both in patients without cancer who have a family history suggestive of the disorder and in all patients who develop colorectal cancer, said Dr. Joel H. Rubenstein and his associates on the clinical guidelines committee (Gastroenterology 2015 Jul. 28 [doi: 10.1053/j.gastro.2015.07.036]).
The guidelines strongly recommend that all colorectal cancers now be tested using immunohistochemistry or assessment of microsatellite instability to identify potential cases of Lynch syndrome. Given the high incidence of colorectal cancer in the United States, this recommendation in particular “may be ripe for consideration as a process measure of quality of care,” they noted.
Until now, older patients with colorectal cancer have not undergone such testing because the yield of positive results was lower than in younger patients, but now there is new appreciation that these results have a significant impact on younger family members, not just the patients themselves. From the perspective of preventing cancer in these relatives, such testing is actually cost effective, said Dr. Rubenstein of the Veterans Affairs Center for Clinical Management Research and the gastroenterology division at the University of Michigan, both in Ann Arbor, and his associates.
At present, the evidence is insufficient to recommend either of these tests above the other for identifying Lynch syndrome. The two have comparable sensitivities and specificities.
The guidelines also recommend that, in people who have no personal history of colorectal or other cancer but who have a family history that suggests Lynch syndrome, risk prediction should be performed, “rather than doing nothing.” If a first-degree relative is known to have a Lynch mutation, people should be offered germline genetic testing for that mutation. Alternatively, “if tumor tissue from an affected relative is available, the screening process should begin with testing on that tumor.”
If none of this information is available, online risk prediction models or free downloadable software incorporating such models can be used to quickly and easily estimate the probability of carrying a Lynch syndrome mutation. This approach is “imperative” to improve case finding, since it is likely that most Lynch syndrome kindreds are undiagnosed, Dr. Rubenstein and his associates said.
Such patients should be offered risk-prediction models rather than proceeding directly to germline genetic testing because of the currently high costs of genetic testing. People without cancer who have a family history suggestive of Lynch syndrome should proceed straight to germline testing if they are considered to be at high risk – for example, if they meet the highly specific Amsterdam criteria.
The AGA guidelines strongly recommend that patients identified as having Lynch syndrome undergo surveillance colonoscopy, as opposed to no surveillance. Good-quality evidence shows that this strategy decreases the overall burden of colorectal cancer and reduces disease-specific mortality. People who carry Lynch syndrome genetic mutations increase their life expectancy by 7 years if they undergo surveillance colonoscopy, and cost-effectiveness analyses indicate that the expense of such screening is lower than the expense of no screening.
The optimal screening interval for such patients has not been determined, but low-quality evidence suggests that undergoing colonoscopy every 1-2 years is the “most prudent” course and is better than doing so at longer intervals.
The guidelines also include a conditional recommendation that people found to have Lynch syndrome should be offered aspirin therapy as cancer prophylaxis. The optimal dose and frequency of aspirin use is not yet known, and there is no evidence that the treatment improves mortality, but some low-quality evidence suggests that aspirin therapy reduces the risk of colorectal and other cancers, and the risk of adverse events is quite low.
FROM GASTROENTEROLOGY
Key clinical point: New AGA guidelines stress that identification of Lynch syndrome must improve because it is so underdiagnosed.
Major finding: Lynch syndrome accounts for 2%-3% of all colorectal cancers in the United States, and the estimated prevalence is 1 in 440 members of the general population.
Data source: A review of the literature and compilation of six key recommendations for diagnosing and managing Lynch syndrome.
Disclosures: This work was supported by the AGA Institute. Dr. Rubenstein and his associates on the clinical guidelines committee reported having no relevant financial or professional conflicts of interest.
Update on informed consent
Question: Which of the following statements regarding the doctrine of informed consent is best?
A. All jurisdictions now require disclosure geared toward the reasonable patient rather than the reasonable doctor.
B. Disclosures are less important and sometimes unnecessary in global clinical research protocols.
C. The trend is toward the use of simplified and personalized consent forms.
D. A and C.
Answer: C. An important development in the doctrine of informed consent deals with the typical consent form, which is lengthy (going on 14 pages in one report), largely incomprehensible, and densely legalistic. Worse, the physician may misconstrue the form as a proxy for risk disclosure, whereas in fact the duly signed form merely purports to indicate that meaningful discussion of risks, benefits, and alternatives had taken place – if indeed that has been the case.
A review of more than 500 separate informed consent forms revealed these documents have limited educational value, go mostly unread, and are frequently misunderstood by patients (Arch. Surg. 2000;135:26-33).Thus, there is much to recommend the recent move toward shorter forms that use simpler language, bigger type fonts, and even graphics to improve patient and family understanding.
For example, a novel personalized approach currently being tested offers evidence-based benefits and risks to patients undergoing elective percutaneous coronary intervention. The consent document draws on the American College of Cardiology’s National Cardiovascular Data Registry to offer individualized risk estimates, and lists the experience of the health care team and the anticipated financial costs (JAMA 2010;303:1190-1).
Such approaches are consonant with the notion of patient-centered care, which the Institute of Medicine has identified as a core attribute of high-quality health care systems.
One of the continuing controversies in informed consent is the requisite standard of disclosure.
Historically, what needed to be disclosed was judged by what was ordinarily expected of the reasonable physician in the community. However, in 1972, Canterbury v. Spence (464 F.2d 772 [D.C. Cir. 1972]) persuasively argued for replacing such a professional standard (what doctors customarily would disclose) with a patient-oriented standard, i.e., what a reasonable patient would want to know.
California quickly followed, and an increasing number of jurisdictions such as Alaska, Hawaii, Massachusetts, Minnesota, Texas, West Virginia, and Wisconsin have since embraced this patient-centered standard. However, a number of other jurisdictions, including Arkansas, Indiana, Michigan, Montana, Nebraska, Nevada, and Wyoming, continue to adhere to the professional or physician-centered standard.
The momentum toward patient-centered disclosure appears to be growing, and has spread to other common-law jurisdictions abroad such as Canada and Australia. The United Kingdom was a steadfast opponent of patient-centric consent since Sidaway v. Board of Governors of the Bethlem Royal Hospital and the Maudsley Hospital ([1985] AC 871), its seminal House of Lords decision in the 1980s.
However, it finally abandoned its opposition on March 11, 2015, when its Supreme Court decided the case of a baby boy who sustained brain injury and arm paralysis at birth as a result of shoulder dystocia (Montgomery v. Lanarkshire Health Board [2015] UKSC 11).
The plaintiff alleged that being a diabetic mother, she should have been warned of the risks of shoulder dystocia and offered the alternative of a cesarean section. The obstetrician said she did not warn because the risks of a serious problem were very small. The court sided with the plaintiff, overturned Sidaway’s professional standard, and adopted the patient standard of disclosure as the new law.
What needs to be disclosed continues to plague the practitioner. The term “material risks” merely begs the question of the definition of “material.” It has been said that the crucial factor is the patient’s need.
The vexing issue surrounding the scope of risk disclosure is compounded by court decisions allowing inquiry outside the medical condition itself, such as a physician’s alcoholism or HIV status, and financial incentives amounting to a breach of fiduciary responsibility. However, courts have tended to reject requiring disclosure of practitioner experience or patient prognosis.
Novel issues continue to surface. For example, a recent Wisconsin Supreme Court case dealt with whether it was necessary for a physician to disclose the availability of a collateral test not directly linked to a patient’s diagnosis.
In Jandre v. Wis. Injured Patients & Families Comp. Fund (813 N.W.2d 627 [Wis. 2012]), a doctor diagnosed Bell’s palsy as the cause of a patient’s neurologic symptoms, because she heard no carotid bruits and the head CT scan was normal. Shortly thereafter, the patient developed a stroke. Although the doctor was found not negligent for the misdiagnosis, the plaintiff then proceeded on a lack of informed consent theory, asserting that the doctor should have advised him of the availability of a carotid ultrasound test to rule out a TIA.
In a momentous decision, the Wisconsin Supreme Court affirmed the lower court’s judgment in finding the physician liable for failing to disclose the availability of the ultrasound procedure.
Another area where informed consent is under renewed scrutiny is in research. Doctors in practice are increasingly participating as coinvestigators in industry-sponsored drug trials, and should be familiar with what is expected in the research context.
Augmented rules regarding informed consent govern all clinical research, with patient safety and free meaningful choice being paramount considerations. Federal and state regulations, implemented and monitored by institutional review boards, serve to enforce compliance and safeguard the safety of experimental subjects.
Still, as predicted nearly 50 years ago by Dr. Henry K. Beecher, “In any precise sense, statements regarding consent are meaningless unless one knows how fully the patient was informed of all risks, and if these are not known, that fact should also be made clear. A far more dependable safeguard than consent is the presence of a truly responsible investigator” (N. Engl. J. Med. 1966;274:1354-60).
Scandals in human research led by or in collaboration with U.S. investigators in foreign countries have been in the news. Global clinical trials are supposed to be carried out in compliance with the Nuremberg Code and the Declaration of Helsinki, but there may be occasional lapses or disregard for such international laws. A recent example is the trial using the antibiotic trovafloxacin, administered orally, versus FDA-approved parenteral ceftriaxone, during a meningitis outbreak in Nigeria (N. Engl. J. Med. 2009;360:2050-3).
Finally, the politics of abortion and first amendment rights have managed to insinuate themselves into the informed consent debate. Can states mandate disclosure in the name of securing meaningful informed consent, e.g., requiring physicians to provide information that might encourage a woman to reconsider her decision to have an abortion?
In 1992, the U.S. Supreme Court decided that this passes constitutional muster, because it did not place an undue burden on the woman. However, the Fourth Circuit Appellate Court recently ruled unconstitutional a North Carolina statute called Display of Real-Time View Requirement. The statute requires physicians to display ultrasound images of the unborn child to a mother contemplating an abortion, which the court ruled violated the First Amendment’s prohibition on state-compelled speech (N. Engl. J. Med. 2015;372:1285-7).
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at siang@hawaii.edu.
Question: Which of the following statements regarding the doctrine of informed consent is best?
A. All jurisdictions now require disclosure geared toward the reasonable patient rather than the reasonable doctor.
B. Disclosures are less important and sometimes unnecessary in global clinical research protocols.
C. The trend is toward the use of simplified and personalized consent forms.
D. A and C.
Answer: C. An important development in the doctrine of informed consent deals with the typical consent form, which is lengthy (going on 14 pages in one report), largely incomprehensible, and densely legalistic. Worse, the physician may misconstrue the form as a proxy for risk disclosure, whereas in fact the duly signed form merely purports to indicate that meaningful discussion of risks, benefits, and alternatives had taken place – if indeed that has been the case.
A review of more than 500 separate informed consent forms revealed these documents have limited educational value, go mostly unread, and are frequently misunderstood by patients (Arch. Surg. 2000;135:26-33).Thus, there is much to recommend the recent move toward shorter forms that use simpler language, bigger type fonts, and even graphics to improve patient and family understanding.
For example, a novel personalized approach currently being tested offers evidence-based benefits and risks to patients undergoing elective percutaneous coronary intervention. The consent document draws on the American College of Cardiology’s National Cardiovascular Data Registry to offer individualized risk estimates, and lists the experience of the health care team and the anticipated financial costs (JAMA 2010;303:1190-1).
Such approaches are consonant with the notion of patient-centered care, which the Institute of Medicine has identified as a core attribute of high-quality health care systems.
One of the continuing controversies in informed consent is the requisite standard of disclosure.
Historically, what needed to be disclosed was judged by what was ordinarily expected of the reasonable physician in the community. However, in 1972, Canterbury v. Spence (464 F.2d 772 [D.C. Cir. 1972]) persuasively argued for replacing such a professional standard (what doctors customarily would disclose) with a patient-oriented standard, i.e., what a reasonable patient would want to know.
California quickly followed, and an increasing number of jurisdictions such as Alaska, Hawaii, Massachusetts, Minnesota, Texas, West Virginia, and Wisconsin have since embraced this patient-centered standard. However, a number of other jurisdictions, including Arkansas, Indiana, Michigan, Montana, Nebraska, Nevada, and Wyoming, continue to adhere to the professional or physician-centered standard.
The momentum toward patient-centered disclosure appears to be growing, and has spread to other common-law jurisdictions abroad such as Canada and Australia. The United Kingdom was a steadfast opponent of patient-centric consent since Sidaway v. Board of Governors of the Bethlem Royal Hospital and the Maudsley Hospital ([1985] AC 871), its seminal House of Lords decision in the 1980s.
However, it finally abandoned its opposition on March 11, 2015, when its Supreme Court decided the case of a baby boy who sustained brain injury and arm paralysis at birth as a result of shoulder dystocia (Montgomery v. Lanarkshire Health Board [2015] UKSC 11).
The plaintiff alleged that being a diabetic mother, she should have been warned of the risks of shoulder dystocia and offered the alternative of a cesarean section. The obstetrician said she did not warn because the risks of a serious problem were very small. The court sided with the plaintiff, overturned Sidaway’s professional standard, and adopted the patient standard of disclosure as the new law.
What needs to be disclosed continues to plague the practitioner. The term “material risks” merely begs the question of the definition of “material.” It has been said that the crucial factor is the patient’s need.
The vexing issue surrounding the scope of risk disclosure is compounded by court decisions allowing inquiry outside the medical condition itself, such as a physician’s alcoholism or HIV status, and financial incentives amounting to a breach of fiduciary responsibility. However, courts have tended to reject requiring disclosure of practitioner experience or patient prognosis.
Novel issues continue to surface. For example, a recent Wisconsin Supreme Court case dealt with whether it was necessary for a physician to disclose the availability of a collateral test not directly linked to a patient’s diagnosis.
In Jandre v. Wis. Injured Patients & Families Comp. Fund (813 N.W.2d 627 [Wis. 2012]), a doctor diagnosed Bell’s palsy as the cause of a patient’s neurologic symptoms, because she heard no carotid bruits and the head CT scan was normal. Shortly thereafter, the patient developed a stroke. Although the doctor was found not negligent for the misdiagnosis, the plaintiff then proceeded on a lack of informed consent theory, asserting that the doctor should have advised him of the availability of a carotid ultrasound test to rule out a TIA.
In a momentous decision, the Wisconsin Supreme Court affirmed the lower court’s judgment in finding the physician liable for failing to disclose the availability of the ultrasound procedure.
Another area where informed consent is under renewed scrutiny is in research. Doctors in practice are increasingly participating as coinvestigators in industry-sponsored drug trials, and should be familiar with what is expected in the research context.
Augmented rules regarding informed consent govern all clinical research, with patient safety and free meaningful choice being paramount considerations. Federal and state regulations, implemented and monitored by institutional review boards, serve to enforce compliance and safeguard the safety of experimental subjects.
Still, as predicted nearly 50 years ago by Dr. Henry K. Beecher, “In any precise sense, statements regarding consent are meaningless unless one knows how fully the patient was informed of all risks, and if these are not known, that fact should also be made clear. A far more dependable safeguard than consent is the presence of a truly responsible investigator” (N. Engl. J. Med. 1966;274:1354-60).
Scandals in human research led by or in collaboration with U.S. investigators in foreign countries have been in the news. Global clinical trials are supposed to be carried out in compliance with the Nuremberg Code and the Declaration of Helsinki, but there may be occasional lapses or disregard for such international laws. A recent example is the trial using the antibiotic trovafloxacin, administered orally, versus FDA-approved parenteral ceftriaxone, during a meningitis outbreak in Nigeria (N. Engl. J. Med. 2009;360:2050-3).
Finally, the politics of abortion and first amendment rights have managed to insinuate themselves into the informed consent debate. Can states mandate disclosure in the name of securing meaningful informed consent, e.g., requiring physicians to provide information that might encourage a woman to reconsider her decision to have an abortion?
In 1992, the U.S. Supreme Court decided that this passes constitutional muster, because it did not place an undue burden on the woman. However, the Fourth Circuit Appellate Court recently ruled unconstitutional a North Carolina statute called Display of Real-Time View Requirement. The statute requires physicians to display ultrasound images of the unborn child to a mother contemplating an abortion, which the court ruled violated the First Amendment’s prohibition on state-compelled speech (N. Engl. J. Med. 2015;372:1285-7).
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at siang@hawaii.edu.
Question: Which of the following statements regarding the doctrine of informed consent is best?
A. All jurisdictions now require disclosure geared toward the reasonable patient rather than the reasonable doctor.
B. Disclosures are less important and sometimes unnecessary in global clinical research protocols.
C. The trend is toward the use of simplified and personalized consent forms.
D. A and C.
Answer: C. An important development in the doctrine of informed consent deals with the typical consent form, which is lengthy (going on 14 pages in one report), largely incomprehensible, and densely legalistic. Worse, the physician may misconstrue the form as a proxy for risk disclosure, whereas in fact the duly signed form merely purports to indicate that meaningful discussion of risks, benefits, and alternatives had taken place – if indeed that has been the case.
A review of more than 500 separate informed consent forms revealed these documents have limited educational value, go mostly unread, and are frequently misunderstood by patients (Arch. Surg. 2000;135:26-33).Thus, there is much to recommend the recent move toward shorter forms that use simpler language, bigger type fonts, and even graphics to improve patient and family understanding.
For example, a novel personalized approach currently being tested offers evidence-based benefits and risks to patients undergoing elective percutaneous coronary intervention. The consent document draws on the American College of Cardiology’s National Cardiovascular Data Registry to offer individualized risk estimates, and lists the experience of the health care team and the anticipated financial costs (JAMA 2010;303:1190-1).
Such approaches are consonant with the notion of patient-centered care, which the Institute of Medicine has identified as a core attribute of high-quality health care systems.
One of the continuing controversies in informed consent is the requisite standard of disclosure.
Historically, what needed to be disclosed was judged by what was ordinarily expected of the reasonable physician in the community. However, in 1972, Canterbury v. Spence (464 F.2d 772 [D.C. Cir. 1972]) persuasively argued for replacing such a professional standard (what doctors customarily would disclose) with a patient-oriented standard, i.e., what a reasonable patient would want to know.
California quickly followed, and an increasing number of jurisdictions such as Alaska, Hawaii, Massachusetts, Minnesota, Texas, West Virginia, and Wisconsin have since embraced this patient-centered standard. However, a number of other jurisdictions, including Arkansas, Indiana, Michigan, Montana, Nebraska, Nevada, and Wyoming, continue to adhere to the professional or physician-centered standard.
The momentum toward patient-centered disclosure appears to be growing, and has spread to other common-law jurisdictions abroad such as Canada and Australia. The United Kingdom was a steadfast opponent of patient-centric consent since Sidaway v. Board of Governors of the Bethlem Royal Hospital and the Maudsley Hospital ([1985] AC 871), its seminal House of Lords decision in the 1980s.
However, it finally abandoned its opposition on March 11, 2015, when its Supreme Court decided the case of a baby boy who sustained brain injury and arm paralysis at birth as a result of shoulder dystocia (Montgomery v. Lanarkshire Health Board [2015] UKSC 11).
The plaintiff alleged that being a diabetic mother, she should have been warned of the risks of shoulder dystocia and offered the alternative of a cesarean section. The obstetrician said she did not warn because the risks of a serious problem were very small. The court sided with the plaintiff, overturned Sidaway’s professional standard, and adopted the patient standard of disclosure as the new law.
What needs to be disclosed continues to plague the practitioner. The term “material risks” merely begs the question of the definition of “material.” It has been said that the crucial factor is the patient’s need.
The vexing issue surrounding the scope of risk disclosure is compounded by court decisions allowing inquiry outside the medical condition itself, such as a physician’s alcoholism or HIV status, and financial incentives amounting to a breach of fiduciary responsibility. However, courts have tended to reject requiring disclosure of practitioner experience or patient prognosis.
Novel issues continue to surface. For example, a recent Wisconsin Supreme Court case dealt with whether it was necessary for a physician to disclose the availability of a collateral test not directly linked to a patient’s diagnosis.
In Jandre v. Wis. Injured Patients & Families Comp. Fund (813 N.W.2d 627 [Wis. 2012]), a doctor diagnosed Bell’s palsy as the cause of a patient’s neurologic symptoms, because she heard no carotid bruits and the head CT scan was normal. Shortly thereafter, the patient developed a stroke. Although the doctor was found not negligent for the misdiagnosis, the plaintiff then proceeded on a lack of informed consent theory, asserting that the doctor should have advised him of the availability of a carotid ultrasound test to rule out a TIA.
In a momentous decision, the Wisconsin Supreme Court affirmed the lower court’s judgment in finding the physician liable for failing to disclose the availability of the ultrasound procedure.
Another area where informed consent is under renewed scrutiny is in research. Doctors in practice are increasingly participating as coinvestigators in industry-sponsored drug trials, and should be familiar with what is expected in the research context.
Augmented rules regarding informed consent govern all clinical research, with patient safety and free meaningful choice being paramount considerations. Federal and state regulations, implemented and monitored by institutional review boards, serve to enforce compliance and safeguard the safety of experimental subjects.
Still, as predicted nearly 50 years ago by Dr. Henry K. Beecher, “In any precise sense, statements regarding consent are meaningless unless one knows how fully the patient was informed of all risks, and if these are not known, that fact should also be made clear. A far more dependable safeguard than consent is the presence of a truly responsible investigator” (N. Engl. J. Med. 1966;274:1354-60).
Scandals in human research led by or in collaboration with U.S. investigators in foreign countries have been in the news. Global clinical trials are supposed to be carried out in compliance with the Nuremberg Code and the Declaration of Helsinki, but there may be occasional lapses or disregard for such international laws. A recent example is the trial using the antibiotic trovafloxacin, administered orally, versus FDA-approved parenteral ceftriaxone, during a meningitis outbreak in Nigeria (N. Engl. J. Med. 2009;360:2050-3).
Finally, the politics of abortion and first amendment rights have managed to insinuate themselves into the informed consent debate. Can states mandate disclosure in the name of securing meaningful informed consent, e.g., requiring physicians to provide information that might encourage a woman to reconsider her decision to have an abortion?
In 1992, the U.S. Supreme Court decided that this passes constitutional muster, because it did not place an undue burden on the woman. However, the Fourth Circuit Appellate Court recently ruled unconstitutional a North Carolina statute called Display of Real-Time View Requirement. The statute requires physicians to display ultrasound images of the unborn child to a mother contemplating an abortion, which the court ruled violated the First Amendment’s prohibition on state-compelled speech (N. Engl. J. Med. 2015;372:1285-7).
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at siang@hawaii.edu.
Witch hunts and hidden agendas
Witch hunt: An intensive effort to discover and expose disloyalty, subversion, dishonesty or the like, usually based on slight, doubtful or irrelevant evidence, and to punish accordingly. - Merriam-Webster Dictionary
Over the years, several excellent vascular surgeons have either lost their jobs or been pressured to resign for reasons that are unclear – although in some instances a few particular cases with bad outcomes have been cited as a reason.
Vascular Surgery is a difficult specialty. If one takes on challenging cases that need treatment – patients with large complex AAAs, extensive foot gangrene, or high-risk symptomatic carotid disease, an occasional poor outcome can be expected even if everything the vascular surgeon does is correct. One, two, or three bad outcomes per year in a busy vascular practice are inevitable, and should not be a reason for a vascular surgeon’s termination. Something more must be involved. Likely it is a hidden agenda.
That something can best be summarized by the term “medical politics.” This term may sound benign. In fact, medical politics are anything but benign. In the case of vascular surgeons, they are often subject to the governance and direction of administrators or senior surgical leaders who have little understanding of the risks of complex vascular surgery, and are interested only in the money generated by vascular admissions and procedures. Quantity and dollars trumps quality most times in the eyes of these administrative leaders.
Moreover, these leaders are not immune from the frailties of bias and jealousy which motivate much of human behavior. Throughout my career I have seen many instances when such administrative leaders resented a vascular surgeon because he or she used many hospital resources for a large practice, was successful academically, but behaved “too independently” from the larger department or its leader. It was one reason, vascular surgery needed to be its own independent specialty rather than a part of general surgery.
In the case of the vascular surgeons facing termination for reasons that they cannot understand, there is usually an administrative superior who is involved. Perhaps the vascular surgeon has questioned the authority or judgment of the superior on the grounds that they do not understand a particular vascular surgery issue. The resulting anger of the superior prompts him to want to eliminate the vascular surgeon who is under his administrative control. The witch hunt begins and may be inflamed by other unjustified motives such as jealousy or other hidden agendas which may never be known.
Once the witch hunt has begun, if the vascular surgeon has one or a few cases with bad outcomes, they make him or her vulnerable to the unjustified attack leading to termination by those who do not understand the nature of vascular surgery. Other factors, e.g., a disgruntled or jealous competitor in vascular surgery, can exacerbate the situation and accelerate the termination. The few bad-outcome cases, however, are the excuse or smoking gun which is used as the reason for termination. What goes unrecognized is the fact that every competent vascular surgeon who treats sick patients has such cases which can be used if uninformed enemies wish to focus on them.
How can a vascular surgeon protect him or herself from such witch hunts? First, they must be aware that the nature of vascular surgery and its requirement for treating challenging patients makes them vulnerable if one or another superior dislikes them or is “out to get them.” Second, if one is dealing with a case likely to have a bad outcome, it should be discussed beforehand with superiors, and it is wise to get help from a colleague who can share responsibility if things go poorly.
Third, try not to make enemies of important administrative leaders by challenging their authority or judgment.
Share successes with them, rather than taking all the glory. It will help to neutralize their jealousy. If possible, be humble. Make as many friends in other specialties as possible. They can sometimes diffuse the intensity of a witch hunt. And lastly be aware that any vascular surgeon, particularly a good one, can be the subject of a damaging witch hunt in the imperfect world in which we function.
Dr. Veith is professor of surgery at New York University Medical Center and the Cleveland Clinic and an associate medical editor for Vascular Specialist.
The ideas and opinions expressed in Vascsular Specialist do not necessarily reflect those of the Society or publisher.
Witch hunt: An intensive effort to discover and expose disloyalty, subversion, dishonesty or the like, usually based on slight, doubtful or irrelevant evidence, and to punish accordingly. - Merriam-Webster Dictionary
Over the years, several excellent vascular surgeons have either lost their jobs or been pressured to resign for reasons that are unclear – although in some instances a few particular cases with bad outcomes have been cited as a reason.
Vascular Surgery is a difficult specialty. If one takes on challenging cases that need treatment – patients with large complex AAAs, extensive foot gangrene, or high-risk symptomatic carotid disease, an occasional poor outcome can be expected even if everything the vascular surgeon does is correct. One, two, or three bad outcomes per year in a busy vascular practice are inevitable, and should not be a reason for a vascular surgeon’s termination. Something more must be involved. Likely it is a hidden agenda.
That something can best be summarized by the term “medical politics.” This term may sound benign. In fact, medical politics are anything but benign. In the case of vascular surgeons, they are often subject to the governance and direction of administrators or senior surgical leaders who have little understanding of the risks of complex vascular surgery, and are interested only in the money generated by vascular admissions and procedures. Quantity and dollars trumps quality most times in the eyes of these administrative leaders.
Moreover, these leaders are not immune from the frailties of bias and jealousy which motivate much of human behavior. Throughout my career I have seen many instances when such administrative leaders resented a vascular surgeon because he or she used many hospital resources for a large practice, was successful academically, but behaved “too independently” from the larger department or its leader. It was one reason, vascular surgery needed to be its own independent specialty rather than a part of general surgery.
In the case of the vascular surgeons facing termination for reasons that they cannot understand, there is usually an administrative superior who is involved. Perhaps the vascular surgeon has questioned the authority or judgment of the superior on the grounds that they do not understand a particular vascular surgery issue. The resulting anger of the superior prompts him to want to eliminate the vascular surgeon who is under his administrative control. The witch hunt begins and may be inflamed by other unjustified motives such as jealousy or other hidden agendas which may never be known.
Once the witch hunt has begun, if the vascular surgeon has one or a few cases with bad outcomes, they make him or her vulnerable to the unjustified attack leading to termination by those who do not understand the nature of vascular surgery. Other factors, e.g., a disgruntled or jealous competitor in vascular surgery, can exacerbate the situation and accelerate the termination. The few bad-outcome cases, however, are the excuse or smoking gun which is used as the reason for termination. What goes unrecognized is the fact that every competent vascular surgeon who treats sick patients has such cases which can be used if uninformed enemies wish to focus on them.
How can a vascular surgeon protect him or herself from such witch hunts? First, they must be aware that the nature of vascular surgery and its requirement for treating challenging patients makes them vulnerable if one or another superior dislikes them or is “out to get them.” Second, if one is dealing with a case likely to have a bad outcome, it should be discussed beforehand with superiors, and it is wise to get help from a colleague who can share responsibility if things go poorly.
Third, try not to make enemies of important administrative leaders by challenging their authority or judgment.
Share successes with them, rather than taking all the glory. It will help to neutralize their jealousy. If possible, be humble. Make as many friends in other specialties as possible. They can sometimes diffuse the intensity of a witch hunt. And lastly be aware that any vascular surgeon, particularly a good one, can be the subject of a damaging witch hunt in the imperfect world in which we function.
Dr. Veith is professor of surgery at New York University Medical Center and the Cleveland Clinic and an associate medical editor for Vascular Specialist.
The ideas and opinions expressed in Vascsular Specialist do not necessarily reflect those of the Society or publisher.
Witch hunt: An intensive effort to discover and expose disloyalty, subversion, dishonesty or the like, usually based on slight, doubtful or irrelevant evidence, and to punish accordingly. - Merriam-Webster Dictionary
Over the years, several excellent vascular surgeons have either lost their jobs or been pressured to resign for reasons that are unclear – although in some instances a few particular cases with bad outcomes have been cited as a reason.
Vascular Surgery is a difficult specialty. If one takes on challenging cases that need treatment – patients with large complex AAAs, extensive foot gangrene, or high-risk symptomatic carotid disease, an occasional poor outcome can be expected even if everything the vascular surgeon does is correct. One, two, or three bad outcomes per year in a busy vascular practice are inevitable, and should not be a reason for a vascular surgeon’s termination. Something more must be involved. Likely it is a hidden agenda.
That something can best be summarized by the term “medical politics.” This term may sound benign. In fact, medical politics are anything but benign. In the case of vascular surgeons, they are often subject to the governance and direction of administrators or senior surgical leaders who have little understanding of the risks of complex vascular surgery, and are interested only in the money generated by vascular admissions and procedures. Quantity and dollars trumps quality most times in the eyes of these administrative leaders.
Moreover, these leaders are not immune from the frailties of bias and jealousy which motivate much of human behavior. Throughout my career I have seen many instances when such administrative leaders resented a vascular surgeon because he or she used many hospital resources for a large practice, was successful academically, but behaved “too independently” from the larger department or its leader. It was one reason, vascular surgery needed to be its own independent specialty rather than a part of general surgery.
In the case of the vascular surgeons facing termination for reasons that they cannot understand, there is usually an administrative superior who is involved. Perhaps the vascular surgeon has questioned the authority or judgment of the superior on the grounds that they do not understand a particular vascular surgery issue. The resulting anger of the superior prompts him to want to eliminate the vascular surgeon who is under his administrative control. The witch hunt begins and may be inflamed by other unjustified motives such as jealousy or other hidden agendas which may never be known.
Once the witch hunt has begun, if the vascular surgeon has one or a few cases with bad outcomes, they make him or her vulnerable to the unjustified attack leading to termination by those who do not understand the nature of vascular surgery. Other factors, e.g., a disgruntled or jealous competitor in vascular surgery, can exacerbate the situation and accelerate the termination. The few bad-outcome cases, however, are the excuse or smoking gun which is used as the reason for termination. What goes unrecognized is the fact that every competent vascular surgeon who treats sick patients has such cases which can be used if uninformed enemies wish to focus on them.
How can a vascular surgeon protect him or herself from such witch hunts? First, they must be aware that the nature of vascular surgery and its requirement for treating challenging patients makes them vulnerable if one or another superior dislikes them or is “out to get them.” Second, if one is dealing with a case likely to have a bad outcome, it should be discussed beforehand with superiors, and it is wise to get help from a colleague who can share responsibility if things go poorly.
Third, try not to make enemies of important administrative leaders by challenging their authority or judgment.
Share successes with them, rather than taking all the glory. It will help to neutralize their jealousy. If possible, be humble. Make as many friends in other specialties as possible. They can sometimes diffuse the intensity of a witch hunt. And lastly be aware that any vascular surgeon, particularly a good one, can be the subject of a damaging witch hunt in the imperfect world in which we function.
Dr. Veith is professor of surgery at New York University Medical Center and the Cleveland Clinic and an associate medical editor for Vascular Specialist.
The ideas and opinions expressed in Vascsular Specialist do not necessarily reflect those of the Society or publisher.