User login
Psoriatic Arthritis Overview
Prior authorizations for infusibles cause delays, toxicities
Rheumatologist Zachary S. Wallace, MD, knew just how prior authorization requirements were impacting his staff time and work flow when he embarked on a study several years ago. Managing authorizations for infusible medications alone was about to become a full-time job for one of the administrative assistants in the rheumatology unit at Massachusetts General Hospital in Boston.
His research questions concerned patients. “There’s a lot of talk about how much onus prior authorization requirements put on providers and the practice,” Dr. Wallace said. ”I was interested in understanding what impact [these requirements] have on patients themselves.”
Dr. Wallace led a review of the EHRs of 225 patients for whom an infusible medication such as rituximab and infliximab was ordered by 1 of the 16 physicians in the rheumatology unit between July 2016 and June 2018. The findings – that patients who needed prior authorizations for infusible medications had a significantly longer time to treatment initiation and higher prednisone-equivalent glucocorticoid exposure – were reported online in Arthritis Care & Research.
Among patients whose authorizations were initially denied, these differences were “pretty drastic,” Dr. Wallace said. The median time to receiving a first infusion was 50 days, compared with 27 days when permission was not required, and glucocorticoid exposure during the 3 months following the request was 605 mg versus 160 mg.
Among patients whose authorizations were not denied, the median time to first infusion was 31 days, compared with 27 days when authorization was not required, and the mean glucocorticoid exposure over 3 months was 364 mg versus 160 mg.
“I hope that our findings will help facilitate discussions with insurance providers, pharmacy benefit managers, and state and federal legislators about the need to address the impact that prior authorization requirements have on patients and providers,” said Dr. Wallace, also of the clinical epidemiology program in the division of rheumatology, allergy and immunology at Massachusetts General, and an assistant professor of medicine at Harvard Medical School, Boston.
Of the 225 patients for whom an infusible medication was ordered, 71% required preauthorization. Of these, 79% were approved and 21% were denied after the first request. And in a finding that Dr. Wallace called “somewhat surprising,” 82% of the authorizations originally denied were approved after appeal.
All told, prior authorizations for infusible medications were eventually approved in all but a small number of cases. “We go through all this effort to get these prior authorizations approved, and 96% of the time, they were ultimately approved,” he said in an interview.
Christopher Phillips, MD, a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the American College of Rheumatology’s committee on rheumatologic care, said the findings “give further credence” to rheumatologists’ concerns. “We know [from our own experiences] that prior authorizations delay care, and we know that delays can cause harm to patients. We now have hard data backing up this assertion.”
Regarding the high number of authorization approvals, “there’s an argument to be made that for certain treatments and certain conditions where the success rate of appeals is high enough, you shouldn’t be subjecting these treatments to these [preauthorization] policies,” he said.
Calls for prior authorization reform
Most patients in the study (71%) had private insurance. But the findings also have implications for Medicare, Dr. Wallace said, as recent federal policies have expanded Medicare Advantage plans’ authority to use prior authorization in conjunction with step therapy for medications administered under Part B. Step therapy favors primary use of what insurers deem the most cost-effective therapies.
The ACR is one of almost 370 physician, patient, and health care organizations that are urging Congress to pass a bipartisan bill aimed at streamlining and standardizing prior authorization under the Medicare Advantage program. The legislation – Improving Seniors’ Timely Access to Care Act of 2019 (H.R. 3107) – was introduced by Reps. Suzan DelBene (D-Wash.), Mike Kelly (R-Pa.), Roger Marshall, MD (R-Kan.), and Ami Bera, MD (D-Calif.).
The bill calls for the creation of an electronic prior authorization program and a “real-time process for items and services that are routinely approved,” as well as greater Centers for Medicare & Medicaid Services oversight on how Medicare Advantage plans use prior authorization. Plans would be required to report to the CMS on the extent of their use of prior authorization and the rate of approvals or denials. They would also be held accountable for making timely prior authorization determinations and providing rationales for denials, according to a letter to Congress cosigned by the ACR.
In a press release about the legislation, Paula Marchetta, MD, president of the ACR, said that “the unregulated use of prior authorization has devolved into a time-consuming and obstructive process that often stalls or outright revokes patient access to medically necessary therapies.” She added that “many health care plans now use prior authorization indiscriminately.”
Cathryn Donaldson, director of communications for America’s Health Insurance Plans (AHIP), said in an email that prior authorization is used for less than 15% of covered services, and that, along with step therapy, it “helps ensure that patients receive care that is safe, effective, and necessary.” AHIP “knows that prior authorization can be improved,” she said, and is committed to streamlining the process.
A demonstration project on the automation of various parts of prior authorization is being coordinated with health information technology companies, plans, and providers, she noted.
The federal legislation is based at least partly on a consensus statement drafted by AHIP, the American Medical Association, and four other organizations representing hospitals, medical groups, and health plans on ways to improve the prior authorization process. Among the items mentioned in the statement is that “regular review” of services subject to prior authorization could help identify therapies that “no longer warrant” prior authorization because of low denial rates.
Outside of Medicare Advantage, the AMA is aware of at least 85 bills being introduced in states this year that address utilization management in commercial plans. Nearly all these bills attempt to reform prior authorization programs in some way, according to R. J. Mills, media relations coordinator for the AMA.
Rheumatologic patients hard hit
Off-label medication use was the most common reason (82%) for a prior authorization denial in the Massachusetts General study, even though 78% of the patients for whom infusible medications were prescribed had a condition with no Food and Drug Administration–approved treatment. Having such a condition was associated with 120% or 190% higher odds of having a denial in unadjusted and adjusted (for age and sex) analyses, Dr. Wallace and colleagues reported.
Moreover, nearly half (48%) of the patients with denials had already tried or were currently taking an oral disease-modifying antirheumatic drug, such as methotrexate.
The majority of denials were for the use of rituximab (70%), followed by infliximab (12%) and tocilizumab (12%). Most of the denials (79%) were appealed successfully through a peer-to-peer discussion. In five cases, the insurer’s preferred drug (for example, adalimumab) had to be used rather than the requested infusion (for example, infliximab).
Infused medications, many of which are biologics, are among the most expensive drugs prescribed for patients with rheumatic diseases. They were easiest for Dr. Wallace to study because of the way prior authorizations are handled in his unit, but prior authorization requirements are “widespread” in rheumatology practices across treatment types, he and Dr. Phillips said.
“Some of our relatively inexpensive treatments are subject to prior authorization requirements,” Dr. Phillips said. “We hear stories about prednisone needing a prior authorization sometimes.”
With respect to infusible medications, the insurance subcommittee is hearing from ACR members about seemingly increasing numbers of both clinical coverage reviews – for example, reviews of prior treatments – and site-of-care restrictions, Dr. Phillips noted. “Some carriers are insisting on infusions in non-hospital-based settings, for cost savings, or on home infusions, which are concerning because of [possible] infusion reactions and medical service availability.”
The application of step therapy to rheumatologic patients is troubling because of the “often unique medical circumstances of the patient,” Dr. Phillips said. “There are enough differences among the [tumor necrosis factor] antagonists, for instance, that make one more appropriate for a certain patient than another. Those differences are not brought into consideration with these policies.”
There are other ways in which prior authorization processes “are not well informed medically,” he said, recalling a case brought to the attention of the subcommittee in which a patient prescribed a biologic drug for psoriatic arthritis was denied authorization because “the documentation did not include a [disease activity measure] that is specific to RA and not used for psoriatic arthritis.”
It is not uncommon for authorizations for infusible medications to take 2 weeks or longer to secure – even when initially approved. In the AMA’s 2018 Prior Authorization Physician Survey, 65% reported waiting at least 1 business day for a decision and 26% reported waiting at least 3 business days for responses. “With infusibles, we’re absolutely dealing with a much longer time,” Dr. Phillips said.
In Dr. Wallace’s study, the finding that prior authorizations facilitated greater prednisone-equivalent glucocorticoid exposure is important, he and his colleagues wrote, because these medications may put patients at higher risk of infection, cardiovascular disease, and diabetes – even in low doses and with short-term use. Notably, the median delay to the initiation of treatment was 29 days, regardless of prior authorization requirements. Dr. Wallace said the delays “likely reflect a combination of factors” – including infusion center waiting lists and patient-level factors – and that his team is “thinking about how to facilitate better access [to their practice’s infusion center] for those who are approved for treatment.”
The most common conditions for which infused medication was ordered were inflammatory arthritis (32%), vasculitis (23%), and IgG4-related disease (17%). The 225 patients in the study had an average age of 53 years.
Dr. Wallace reported that he has no relevant financial disclosures.
SOURCE: Wallace ZS et al. Arthritis Care Res. 2019 Sep 10. doi: 10.1002/acr.24062.
Rheumatologist Zachary S. Wallace, MD, knew just how prior authorization requirements were impacting his staff time and work flow when he embarked on a study several years ago. Managing authorizations for infusible medications alone was about to become a full-time job for one of the administrative assistants in the rheumatology unit at Massachusetts General Hospital in Boston.
His research questions concerned patients. “There’s a lot of talk about how much onus prior authorization requirements put on providers and the practice,” Dr. Wallace said. ”I was interested in understanding what impact [these requirements] have on patients themselves.”
Dr. Wallace led a review of the EHRs of 225 patients for whom an infusible medication such as rituximab and infliximab was ordered by 1 of the 16 physicians in the rheumatology unit between July 2016 and June 2018. The findings – that patients who needed prior authorizations for infusible medications had a significantly longer time to treatment initiation and higher prednisone-equivalent glucocorticoid exposure – were reported online in Arthritis Care & Research.
Among patients whose authorizations were initially denied, these differences were “pretty drastic,” Dr. Wallace said. The median time to receiving a first infusion was 50 days, compared with 27 days when permission was not required, and glucocorticoid exposure during the 3 months following the request was 605 mg versus 160 mg.
Among patients whose authorizations were not denied, the median time to first infusion was 31 days, compared with 27 days when authorization was not required, and the mean glucocorticoid exposure over 3 months was 364 mg versus 160 mg.
“I hope that our findings will help facilitate discussions with insurance providers, pharmacy benefit managers, and state and federal legislators about the need to address the impact that prior authorization requirements have on patients and providers,” said Dr. Wallace, also of the clinical epidemiology program in the division of rheumatology, allergy and immunology at Massachusetts General, and an assistant professor of medicine at Harvard Medical School, Boston.
Of the 225 patients for whom an infusible medication was ordered, 71% required preauthorization. Of these, 79% were approved and 21% were denied after the first request. And in a finding that Dr. Wallace called “somewhat surprising,” 82% of the authorizations originally denied were approved after appeal.
All told, prior authorizations for infusible medications were eventually approved in all but a small number of cases. “We go through all this effort to get these prior authorizations approved, and 96% of the time, they were ultimately approved,” he said in an interview.
Christopher Phillips, MD, a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the American College of Rheumatology’s committee on rheumatologic care, said the findings “give further credence” to rheumatologists’ concerns. “We know [from our own experiences] that prior authorizations delay care, and we know that delays can cause harm to patients. We now have hard data backing up this assertion.”
Regarding the high number of authorization approvals, “there’s an argument to be made that for certain treatments and certain conditions where the success rate of appeals is high enough, you shouldn’t be subjecting these treatments to these [preauthorization] policies,” he said.
Calls for prior authorization reform
Most patients in the study (71%) had private insurance. But the findings also have implications for Medicare, Dr. Wallace said, as recent federal policies have expanded Medicare Advantage plans’ authority to use prior authorization in conjunction with step therapy for medications administered under Part B. Step therapy favors primary use of what insurers deem the most cost-effective therapies.
The ACR is one of almost 370 physician, patient, and health care organizations that are urging Congress to pass a bipartisan bill aimed at streamlining and standardizing prior authorization under the Medicare Advantage program. The legislation – Improving Seniors’ Timely Access to Care Act of 2019 (H.R. 3107) – was introduced by Reps. Suzan DelBene (D-Wash.), Mike Kelly (R-Pa.), Roger Marshall, MD (R-Kan.), and Ami Bera, MD (D-Calif.).
The bill calls for the creation of an electronic prior authorization program and a “real-time process for items and services that are routinely approved,” as well as greater Centers for Medicare & Medicaid Services oversight on how Medicare Advantage plans use prior authorization. Plans would be required to report to the CMS on the extent of their use of prior authorization and the rate of approvals or denials. They would also be held accountable for making timely prior authorization determinations and providing rationales for denials, according to a letter to Congress cosigned by the ACR.
In a press release about the legislation, Paula Marchetta, MD, president of the ACR, said that “the unregulated use of prior authorization has devolved into a time-consuming and obstructive process that often stalls or outright revokes patient access to medically necessary therapies.” She added that “many health care plans now use prior authorization indiscriminately.”
Cathryn Donaldson, director of communications for America’s Health Insurance Plans (AHIP), said in an email that prior authorization is used for less than 15% of covered services, and that, along with step therapy, it “helps ensure that patients receive care that is safe, effective, and necessary.” AHIP “knows that prior authorization can be improved,” she said, and is committed to streamlining the process.
A demonstration project on the automation of various parts of prior authorization is being coordinated with health information technology companies, plans, and providers, she noted.
The federal legislation is based at least partly on a consensus statement drafted by AHIP, the American Medical Association, and four other organizations representing hospitals, medical groups, and health plans on ways to improve the prior authorization process. Among the items mentioned in the statement is that “regular review” of services subject to prior authorization could help identify therapies that “no longer warrant” prior authorization because of low denial rates.
Outside of Medicare Advantage, the AMA is aware of at least 85 bills being introduced in states this year that address utilization management in commercial plans. Nearly all these bills attempt to reform prior authorization programs in some way, according to R. J. Mills, media relations coordinator for the AMA.
Rheumatologic patients hard hit
Off-label medication use was the most common reason (82%) for a prior authorization denial in the Massachusetts General study, even though 78% of the patients for whom infusible medications were prescribed had a condition with no Food and Drug Administration–approved treatment. Having such a condition was associated with 120% or 190% higher odds of having a denial in unadjusted and adjusted (for age and sex) analyses, Dr. Wallace and colleagues reported.
Moreover, nearly half (48%) of the patients with denials had already tried or were currently taking an oral disease-modifying antirheumatic drug, such as methotrexate.
The majority of denials were for the use of rituximab (70%), followed by infliximab (12%) and tocilizumab (12%). Most of the denials (79%) were appealed successfully through a peer-to-peer discussion. In five cases, the insurer’s preferred drug (for example, adalimumab) had to be used rather than the requested infusion (for example, infliximab).
Infused medications, many of which are biologics, are among the most expensive drugs prescribed for patients with rheumatic diseases. They were easiest for Dr. Wallace to study because of the way prior authorizations are handled in his unit, but prior authorization requirements are “widespread” in rheumatology practices across treatment types, he and Dr. Phillips said.
“Some of our relatively inexpensive treatments are subject to prior authorization requirements,” Dr. Phillips said. “We hear stories about prednisone needing a prior authorization sometimes.”
With respect to infusible medications, the insurance subcommittee is hearing from ACR members about seemingly increasing numbers of both clinical coverage reviews – for example, reviews of prior treatments – and site-of-care restrictions, Dr. Phillips noted. “Some carriers are insisting on infusions in non-hospital-based settings, for cost savings, or on home infusions, which are concerning because of [possible] infusion reactions and medical service availability.”
The application of step therapy to rheumatologic patients is troubling because of the “often unique medical circumstances of the patient,” Dr. Phillips said. “There are enough differences among the [tumor necrosis factor] antagonists, for instance, that make one more appropriate for a certain patient than another. Those differences are not brought into consideration with these policies.”
There are other ways in which prior authorization processes “are not well informed medically,” he said, recalling a case brought to the attention of the subcommittee in which a patient prescribed a biologic drug for psoriatic arthritis was denied authorization because “the documentation did not include a [disease activity measure] that is specific to RA and not used for psoriatic arthritis.”
It is not uncommon for authorizations for infusible medications to take 2 weeks or longer to secure – even when initially approved. In the AMA’s 2018 Prior Authorization Physician Survey, 65% reported waiting at least 1 business day for a decision and 26% reported waiting at least 3 business days for responses. “With infusibles, we’re absolutely dealing with a much longer time,” Dr. Phillips said.
In Dr. Wallace’s study, the finding that prior authorizations facilitated greater prednisone-equivalent glucocorticoid exposure is important, he and his colleagues wrote, because these medications may put patients at higher risk of infection, cardiovascular disease, and diabetes – even in low doses and with short-term use. Notably, the median delay to the initiation of treatment was 29 days, regardless of prior authorization requirements. Dr. Wallace said the delays “likely reflect a combination of factors” – including infusion center waiting lists and patient-level factors – and that his team is “thinking about how to facilitate better access [to their practice’s infusion center] for those who are approved for treatment.”
The most common conditions for which infused medication was ordered were inflammatory arthritis (32%), vasculitis (23%), and IgG4-related disease (17%). The 225 patients in the study had an average age of 53 years.
Dr. Wallace reported that he has no relevant financial disclosures.
SOURCE: Wallace ZS et al. Arthritis Care Res. 2019 Sep 10. doi: 10.1002/acr.24062.
Rheumatologist Zachary S. Wallace, MD, knew just how prior authorization requirements were impacting his staff time and work flow when he embarked on a study several years ago. Managing authorizations for infusible medications alone was about to become a full-time job for one of the administrative assistants in the rheumatology unit at Massachusetts General Hospital in Boston.
His research questions concerned patients. “There’s a lot of talk about how much onus prior authorization requirements put on providers and the practice,” Dr. Wallace said. ”I was interested in understanding what impact [these requirements] have on patients themselves.”
Dr. Wallace led a review of the EHRs of 225 patients for whom an infusible medication such as rituximab and infliximab was ordered by 1 of the 16 physicians in the rheumatology unit between July 2016 and June 2018. The findings – that patients who needed prior authorizations for infusible medications had a significantly longer time to treatment initiation and higher prednisone-equivalent glucocorticoid exposure – were reported online in Arthritis Care & Research.
Among patients whose authorizations were initially denied, these differences were “pretty drastic,” Dr. Wallace said. The median time to receiving a first infusion was 50 days, compared with 27 days when permission was not required, and glucocorticoid exposure during the 3 months following the request was 605 mg versus 160 mg.
Among patients whose authorizations were not denied, the median time to first infusion was 31 days, compared with 27 days when authorization was not required, and the mean glucocorticoid exposure over 3 months was 364 mg versus 160 mg.
“I hope that our findings will help facilitate discussions with insurance providers, pharmacy benefit managers, and state and federal legislators about the need to address the impact that prior authorization requirements have on patients and providers,” said Dr. Wallace, also of the clinical epidemiology program in the division of rheumatology, allergy and immunology at Massachusetts General, and an assistant professor of medicine at Harvard Medical School, Boston.
Of the 225 patients for whom an infusible medication was ordered, 71% required preauthorization. Of these, 79% were approved and 21% were denied after the first request. And in a finding that Dr. Wallace called “somewhat surprising,” 82% of the authorizations originally denied were approved after appeal.
All told, prior authorizations for infusible medications were eventually approved in all but a small number of cases. “We go through all this effort to get these prior authorizations approved, and 96% of the time, they were ultimately approved,” he said in an interview.
Christopher Phillips, MD, a community rheumatologist in Paducah, Ky., who serves as chair of the insurance subcommittee of the American College of Rheumatology’s committee on rheumatologic care, said the findings “give further credence” to rheumatologists’ concerns. “We know [from our own experiences] that prior authorizations delay care, and we know that delays can cause harm to patients. We now have hard data backing up this assertion.”
Regarding the high number of authorization approvals, “there’s an argument to be made that for certain treatments and certain conditions where the success rate of appeals is high enough, you shouldn’t be subjecting these treatments to these [preauthorization] policies,” he said.
Calls for prior authorization reform
Most patients in the study (71%) had private insurance. But the findings also have implications for Medicare, Dr. Wallace said, as recent federal policies have expanded Medicare Advantage plans’ authority to use prior authorization in conjunction with step therapy for medications administered under Part B. Step therapy favors primary use of what insurers deem the most cost-effective therapies.
The ACR is one of almost 370 physician, patient, and health care organizations that are urging Congress to pass a bipartisan bill aimed at streamlining and standardizing prior authorization under the Medicare Advantage program. The legislation – Improving Seniors’ Timely Access to Care Act of 2019 (H.R. 3107) – was introduced by Reps. Suzan DelBene (D-Wash.), Mike Kelly (R-Pa.), Roger Marshall, MD (R-Kan.), and Ami Bera, MD (D-Calif.).
The bill calls for the creation of an electronic prior authorization program and a “real-time process for items and services that are routinely approved,” as well as greater Centers for Medicare & Medicaid Services oversight on how Medicare Advantage plans use prior authorization. Plans would be required to report to the CMS on the extent of their use of prior authorization and the rate of approvals or denials. They would also be held accountable for making timely prior authorization determinations and providing rationales for denials, according to a letter to Congress cosigned by the ACR.
In a press release about the legislation, Paula Marchetta, MD, president of the ACR, said that “the unregulated use of prior authorization has devolved into a time-consuming and obstructive process that often stalls or outright revokes patient access to medically necessary therapies.” She added that “many health care plans now use prior authorization indiscriminately.”
Cathryn Donaldson, director of communications for America’s Health Insurance Plans (AHIP), said in an email that prior authorization is used for less than 15% of covered services, and that, along with step therapy, it “helps ensure that patients receive care that is safe, effective, and necessary.” AHIP “knows that prior authorization can be improved,” she said, and is committed to streamlining the process.
A demonstration project on the automation of various parts of prior authorization is being coordinated with health information technology companies, plans, and providers, she noted.
The federal legislation is based at least partly on a consensus statement drafted by AHIP, the American Medical Association, and four other organizations representing hospitals, medical groups, and health plans on ways to improve the prior authorization process. Among the items mentioned in the statement is that “regular review” of services subject to prior authorization could help identify therapies that “no longer warrant” prior authorization because of low denial rates.
Outside of Medicare Advantage, the AMA is aware of at least 85 bills being introduced in states this year that address utilization management in commercial plans. Nearly all these bills attempt to reform prior authorization programs in some way, according to R. J. Mills, media relations coordinator for the AMA.
Rheumatologic patients hard hit
Off-label medication use was the most common reason (82%) for a prior authorization denial in the Massachusetts General study, even though 78% of the patients for whom infusible medications were prescribed had a condition with no Food and Drug Administration–approved treatment. Having such a condition was associated with 120% or 190% higher odds of having a denial in unadjusted and adjusted (for age and sex) analyses, Dr. Wallace and colleagues reported.
Moreover, nearly half (48%) of the patients with denials had already tried or were currently taking an oral disease-modifying antirheumatic drug, such as methotrexate.
The majority of denials were for the use of rituximab (70%), followed by infliximab (12%) and tocilizumab (12%). Most of the denials (79%) were appealed successfully through a peer-to-peer discussion. In five cases, the insurer’s preferred drug (for example, adalimumab) had to be used rather than the requested infusion (for example, infliximab).
Infused medications, many of which are biologics, are among the most expensive drugs prescribed for patients with rheumatic diseases. They were easiest for Dr. Wallace to study because of the way prior authorizations are handled in his unit, but prior authorization requirements are “widespread” in rheumatology practices across treatment types, he and Dr. Phillips said.
“Some of our relatively inexpensive treatments are subject to prior authorization requirements,” Dr. Phillips said. “We hear stories about prednisone needing a prior authorization sometimes.”
With respect to infusible medications, the insurance subcommittee is hearing from ACR members about seemingly increasing numbers of both clinical coverage reviews – for example, reviews of prior treatments – and site-of-care restrictions, Dr. Phillips noted. “Some carriers are insisting on infusions in non-hospital-based settings, for cost savings, or on home infusions, which are concerning because of [possible] infusion reactions and medical service availability.”
The application of step therapy to rheumatologic patients is troubling because of the “often unique medical circumstances of the patient,” Dr. Phillips said. “There are enough differences among the [tumor necrosis factor] antagonists, for instance, that make one more appropriate for a certain patient than another. Those differences are not brought into consideration with these policies.”
There are other ways in which prior authorization processes “are not well informed medically,” he said, recalling a case brought to the attention of the subcommittee in which a patient prescribed a biologic drug for psoriatic arthritis was denied authorization because “the documentation did not include a [disease activity measure] that is specific to RA and not used for psoriatic arthritis.”
It is not uncommon for authorizations for infusible medications to take 2 weeks or longer to secure – even when initially approved. In the AMA’s 2018 Prior Authorization Physician Survey, 65% reported waiting at least 1 business day for a decision and 26% reported waiting at least 3 business days for responses. “With infusibles, we’re absolutely dealing with a much longer time,” Dr. Phillips said.
In Dr. Wallace’s study, the finding that prior authorizations facilitated greater prednisone-equivalent glucocorticoid exposure is important, he and his colleagues wrote, because these medications may put patients at higher risk of infection, cardiovascular disease, and diabetes – even in low doses and with short-term use. Notably, the median delay to the initiation of treatment was 29 days, regardless of prior authorization requirements. Dr. Wallace said the delays “likely reflect a combination of factors” – including infusion center waiting lists and patient-level factors – and that his team is “thinking about how to facilitate better access [to their practice’s infusion center] for those who are approved for treatment.”
The most common conditions for which infused medication was ordered were inflammatory arthritis (32%), vasculitis (23%), and IgG4-related disease (17%). The 225 patients in the study had an average age of 53 years.
Dr. Wallace reported that he has no relevant financial disclosures.
SOURCE: Wallace ZS et al. Arthritis Care Res. 2019 Sep 10. doi: 10.1002/acr.24062.
FROM ARTHRITIS CARE & RESEARCH
PsA Fast Facts: Comorbidities



Obesity, moderate drinking linked to psoriatic arthritis
a study has found.
Around one in five people with psoriasis will develop psoriatic arthritis (PsA), wrote Amelia Green of the University of Bath (England) and coauthors in the British Journal of Dermatology.
Previous studies have explored possible links between obesity, alcohol consumption, or smoking, and an increased risk of developing psoriatic arthritis. However, some of these studies found conflicting results or had limitations such as measuring only a single exposure.
In a cohort study, the Ms. Green and her colleagues examined data from the U.K. Clinical Practice Research Datalink for 90,189 individuals with psoriasis, 1,409 of whom were subsequently also diagnosed with psoriatic arthritis.
The analysis showed a significant association between increasing body mass index (BMI) and increasing odds of developing psoriatic arthritis. Compared with individuals with a BMI below 25 kg/m2, those with a BMI of 25.0-29.9 had a 79% greater odds of psoriatic arthritis, those with a BMI of 30.0-34.9 had a 2.10-fold greater odds, and those with a BMI at or above 35 had a 2.68-fold greater odds of developing psoriatic arthritis (P for trend less than .001). Adjustment for potential confounders such as sex, age, duration and severity of psoriasis, diabetes, smoking, and alcohol use slightly attenuated the association, but it remained statistically significant.
Researchers also examined the cumulative effect of lower BMIs over time, and found that over a 10-year period, reductions in BMI were associated with reductions in the risk of developing PsA, compared with remaining at the same BMI over that time.
“Here we have shown for the first time that losing weight over time could reduce the risk of developing PsA in a population with documented psoriasis,” the authors wrote. “As the effect of obesity on the risk of developing PsA may in fact occur with some delay and change over time, our analysis took into account both updated BMI measurements over time and the possible nonlinear and cumulative effects of BMI, which have not previously been investigated.”
Commenting on the mechanisms underlying the association between obesity and the development of PsA, the authors noted that adipose tissue is a source of inflammatory mediators such as adipokines and proinflammatory cytokines, which could lead to the development of PsA. Increasing body weight also could cause microtraumas of the connective tissue between tendon and bone, which may act as an initiating pathogenic event for PsA.
Moderate drinkers – defined as 0.1–3.0 drinks per day – had 57% higher odds of developing PsA when compared with nondrinkers, but former drinkers or heavy drinkers did not have an increased risk.
The study also didn’t see any effect of either past or current smoking on the risk of PsA, although there was a nonsignificant interaction with obesity that hinted at increased odds.
“While we found no association between smoking status and the development of PsA in people with psoriasis, further analysis revealed that the effect of smoking on the risk of PsA was possibly mediated through the effect of BMI on PsA; in other words, the protective effect of smoking may be associated with lower BMI among smokers,” the authors wrote.
Patients who developed PsA were also more likely to be younger (mean age of 44.7 years vs. 48.5 years), have severe psoriasis, and have had the disease for a shorter duration.
The study was funded by the National Institute for Health Research, and the authors declared grants from the funder during the conduct of the study. No other conflicts of interest were declared.
SOURCE: Green A et al. Br J Dermatol. 2019 Jun 18. doi: 10.1111/bjd.18227
a study has found.
Around one in five people with psoriasis will develop psoriatic arthritis (PsA), wrote Amelia Green of the University of Bath (England) and coauthors in the British Journal of Dermatology.
Previous studies have explored possible links between obesity, alcohol consumption, or smoking, and an increased risk of developing psoriatic arthritis. However, some of these studies found conflicting results or had limitations such as measuring only a single exposure.
In a cohort study, the Ms. Green and her colleagues examined data from the U.K. Clinical Practice Research Datalink for 90,189 individuals with psoriasis, 1,409 of whom were subsequently also diagnosed with psoriatic arthritis.
The analysis showed a significant association between increasing body mass index (BMI) and increasing odds of developing psoriatic arthritis. Compared with individuals with a BMI below 25 kg/m2, those with a BMI of 25.0-29.9 had a 79% greater odds of psoriatic arthritis, those with a BMI of 30.0-34.9 had a 2.10-fold greater odds, and those with a BMI at or above 35 had a 2.68-fold greater odds of developing psoriatic arthritis (P for trend less than .001). Adjustment for potential confounders such as sex, age, duration and severity of psoriasis, diabetes, smoking, and alcohol use slightly attenuated the association, but it remained statistically significant.
Researchers also examined the cumulative effect of lower BMIs over time, and found that over a 10-year period, reductions in BMI were associated with reductions in the risk of developing PsA, compared with remaining at the same BMI over that time.
“Here we have shown for the first time that losing weight over time could reduce the risk of developing PsA in a population with documented psoriasis,” the authors wrote. “As the effect of obesity on the risk of developing PsA may in fact occur with some delay and change over time, our analysis took into account both updated BMI measurements over time and the possible nonlinear and cumulative effects of BMI, which have not previously been investigated.”
Commenting on the mechanisms underlying the association between obesity and the development of PsA, the authors noted that adipose tissue is a source of inflammatory mediators such as adipokines and proinflammatory cytokines, which could lead to the development of PsA. Increasing body weight also could cause microtraumas of the connective tissue between tendon and bone, which may act as an initiating pathogenic event for PsA.
Moderate drinkers – defined as 0.1–3.0 drinks per day – had 57% higher odds of developing PsA when compared with nondrinkers, but former drinkers or heavy drinkers did not have an increased risk.
The study also didn’t see any effect of either past or current smoking on the risk of PsA, although there was a nonsignificant interaction with obesity that hinted at increased odds.
“While we found no association between smoking status and the development of PsA in people with psoriasis, further analysis revealed that the effect of smoking on the risk of PsA was possibly mediated through the effect of BMI on PsA; in other words, the protective effect of smoking may be associated with lower BMI among smokers,” the authors wrote.
Patients who developed PsA were also more likely to be younger (mean age of 44.7 years vs. 48.5 years), have severe psoriasis, and have had the disease for a shorter duration.
The study was funded by the National Institute for Health Research, and the authors declared grants from the funder during the conduct of the study. No other conflicts of interest were declared.
SOURCE: Green A et al. Br J Dermatol. 2019 Jun 18. doi: 10.1111/bjd.18227
a study has found.
Around one in five people with psoriasis will develop psoriatic arthritis (PsA), wrote Amelia Green of the University of Bath (England) and coauthors in the British Journal of Dermatology.
Previous studies have explored possible links between obesity, alcohol consumption, or smoking, and an increased risk of developing psoriatic arthritis. However, some of these studies found conflicting results or had limitations such as measuring only a single exposure.
In a cohort study, the Ms. Green and her colleagues examined data from the U.K. Clinical Practice Research Datalink for 90,189 individuals with psoriasis, 1,409 of whom were subsequently also diagnosed with psoriatic arthritis.
The analysis showed a significant association between increasing body mass index (BMI) and increasing odds of developing psoriatic arthritis. Compared with individuals with a BMI below 25 kg/m2, those with a BMI of 25.0-29.9 had a 79% greater odds of psoriatic arthritis, those with a BMI of 30.0-34.9 had a 2.10-fold greater odds, and those with a BMI at or above 35 had a 2.68-fold greater odds of developing psoriatic arthritis (P for trend less than .001). Adjustment for potential confounders such as sex, age, duration and severity of psoriasis, diabetes, smoking, and alcohol use slightly attenuated the association, but it remained statistically significant.
Researchers also examined the cumulative effect of lower BMIs over time, and found that over a 10-year period, reductions in BMI were associated with reductions in the risk of developing PsA, compared with remaining at the same BMI over that time.
“Here we have shown for the first time that losing weight over time could reduce the risk of developing PsA in a population with documented psoriasis,” the authors wrote. “As the effect of obesity on the risk of developing PsA may in fact occur with some delay and change over time, our analysis took into account both updated BMI measurements over time and the possible nonlinear and cumulative effects of BMI, which have not previously been investigated.”
Commenting on the mechanisms underlying the association between obesity and the development of PsA, the authors noted that adipose tissue is a source of inflammatory mediators such as adipokines and proinflammatory cytokines, which could lead to the development of PsA. Increasing body weight also could cause microtraumas of the connective tissue between tendon and bone, which may act as an initiating pathogenic event for PsA.
Moderate drinkers – defined as 0.1–3.0 drinks per day – had 57% higher odds of developing PsA when compared with nondrinkers, but former drinkers or heavy drinkers did not have an increased risk.
The study also didn’t see any effect of either past or current smoking on the risk of PsA, although there was a nonsignificant interaction with obesity that hinted at increased odds.
“While we found no association between smoking status and the development of PsA in people with psoriasis, further analysis revealed that the effect of smoking on the risk of PsA was possibly mediated through the effect of BMI on PsA; in other words, the protective effect of smoking may be associated with lower BMI among smokers,” the authors wrote.
Patients who developed PsA were also more likely to be younger (mean age of 44.7 years vs. 48.5 years), have severe psoriasis, and have had the disease for a shorter duration.
The study was funded by the National Institute for Health Research, and the authors declared grants from the funder during the conduct of the study. No other conflicts of interest were declared.
SOURCE: Green A et al. Br J Dermatol. 2019 Jun 18. doi: 10.1111/bjd.18227
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Long-term advantages may not endure after early tight control in psoriatic arthritis
, based on data from 110 patients in the TIght COntrol of inflammation in early Psoriatic Arthritis (TICOPA) study.
In the original study, significantly more adults with psoriatic arthritis (PsA) who were randomized to a tight control, treat-to-target group achieved minimal disease activity criteria after 48 weeks, compared with a standard care group (40% vs. 25%).
“Following exit from this study, we hypothesized that this advantage would translate to a clinical advantage in the medium term,” wrote Laura C. Coates, MBChB, PhD, of the University of Oxford (England) and colleagues.
In a study published in Rheumatology, the researchers examined data from 54 patients in the tight control arm of TICOPA and 56 patients in the standard care arm.
At 5 years after the completion of the TICOPA study, 69% of patients in the tight control group and 76% of patients in the standard care group were considered to be in low disease activity. In addition, methotrexate use after 5 years was similar between the tight control and standard care groups (44% and 54%, respectively) and both groups had reduced methotrexate use since the study’s end.
Overall use of biologic drugs was similar between the tight control and standard care groups after 5 years (54% and 52%, respectively), although overall use of biologics was higher in the tight control group at the end of the original study, compared with the standard care group (33% vs. 9%).
The findings were limited by several factors, notably the lack of intention to continue treatment or observations beyond the end of the original TICOPA study, and the patients’ status at 5 years was based on routine clinician notes with no formal assessment of minimal disease activity or objective measure of disease status, the researchers noted.
However, “this result reflects clinical practice in routine rheumatology care,” and any benefit of early tight control on later disease activity could not be determined, they added.
The current study was funded by the National Institute for Health Research infrastructure at Leeds and Oxford (England). The original TICOPA study was funded by Arthritis Research UK (now called Versus Arthritis) and Pfizer. Some of the investigators disclosed financial relationships with companies that market drugs for PsA.
SOURCE: Coates LC et al. Rheumatology. 2019 Aug 31. doi: 10.1093/rheumatology/kez369.
, based on data from 110 patients in the TIght COntrol of inflammation in early Psoriatic Arthritis (TICOPA) study.
In the original study, significantly more adults with psoriatic arthritis (PsA) who were randomized to a tight control, treat-to-target group achieved minimal disease activity criteria after 48 weeks, compared with a standard care group (40% vs. 25%).
“Following exit from this study, we hypothesized that this advantage would translate to a clinical advantage in the medium term,” wrote Laura C. Coates, MBChB, PhD, of the University of Oxford (England) and colleagues.
In a study published in Rheumatology, the researchers examined data from 54 patients in the tight control arm of TICOPA and 56 patients in the standard care arm.
At 5 years after the completion of the TICOPA study, 69% of patients in the tight control group and 76% of patients in the standard care group were considered to be in low disease activity. In addition, methotrexate use after 5 years was similar between the tight control and standard care groups (44% and 54%, respectively) and both groups had reduced methotrexate use since the study’s end.
Overall use of biologic drugs was similar between the tight control and standard care groups after 5 years (54% and 52%, respectively), although overall use of biologics was higher in the tight control group at the end of the original study, compared with the standard care group (33% vs. 9%).
The findings were limited by several factors, notably the lack of intention to continue treatment or observations beyond the end of the original TICOPA study, and the patients’ status at 5 years was based on routine clinician notes with no formal assessment of minimal disease activity or objective measure of disease status, the researchers noted.
However, “this result reflects clinical practice in routine rheumatology care,” and any benefit of early tight control on later disease activity could not be determined, they added.
The current study was funded by the National Institute for Health Research infrastructure at Leeds and Oxford (England). The original TICOPA study was funded by Arthritis Research UK (now called Versus Arthritis) and Pfizer. Some of the investigators disclosed financial relationships with companies that market drugs for PsA.
SOURCE: Coates LC et al. Rheumatology. 2019 Aug 31. doi: 10.1093/rheumatology/kez369.
, based on data from 110 patients in the TIght COntrol of inflammation in early Psoriatic Arthritis (TICOPA) study.
In the original study, significantly more adults with psoriatic arthritis (PsA) who were randomized to a tight control, treat-to-target group achieved minimal disease activity criteria after 48 weeks, compared with a standard care group (40% vs. 25%).
“Following exit from this study, we hypothesized that this advantage would translate to a clinical advantage in the medium term,” wrote Laura C. Coates, MBChB, PhD, of the University of Oxford (England) and colleagues.
In a study published in Rheumatology, the researchers examined data from 54 patients in the tight control arm of TICOPA and 56 patients in the standard care arm.
At 5 years after the completion of the TICOPA study, 69% of patients in the tight control group and 76% of patients in the standard care group were considered to be in low disease activity. In addition, methotrexate use after 5 years was similar between the tight control and standard care groups (44% and 54%, respectively) and both groups had reduced methotrexate use since the study’s end.
Overall use of biologic drugs was similar between the tight control and standard care groups after 5 years (54% and 52%, respectively), although overall use of biologics was higher in the tight control group at the end of the original study, compared with the standard care group (33% vs. 9%).
The findings were limited by several factors, notably the lack of intention to continue treatment or observations beyond the end of the original TICOPA study, and the patients’ status at 5 years was based on routine clinician notes with no formal assessment of minimal disease activity or objective measure of disease status, the researchers noted.
However, “this result reflects clinical practice in routine rheumatology care,” and any benefit of early tight control on later disease activity could not be determined, they added.
The current study was funded by the National Institute for Health Research infrastructure at Leeds and Oxford (England). The original TICOPA study was funded by Arthritis Research UK (now called Versus Arthritis) and Pfizer. Some of the investigators disclosed financial relationships with companies that market drugs for PsA.
SOURCE: Coates LC et al. Rheumatology. 2019 Aug 31. doi: 10.1093/rheumatology/kez369.
FROM RHEUMATOLOGY
Key clinical point: Patients in a psoriatic arthritis study comparing tight control and standard care showed no significant difference in disease activity 5 years later.
Major finding: At 5 years after the end of the TICOPA trial, 69% in the tight control group vs. 76% in the standard care group were considered to be in low disease activity.
Study details: The data come from a follow-up of 110 patients from the TIght COntrol of inflammation in early Psoriatic Arthritis (TICOPA) study.
Disclosures: The current study was funded by the National Institute for Health Research infrastructure at Leeds and Oxford (England). The original TICOPA study was funded by Arthritis Research UK (now called Versus Arthritis) and Pfizer. Some of the investigators disclosed financial relationships with companies that market drugs for PsA.
Source: Coates LC et al. Rheumatology. 2019 Aug 31. doi: 10.1093/rheumatology/kez369.
Cannabidiol may interact with rheumatologic drugs
A number of medications commonly prescribed by rheumatologists may interact with cannabidiol oil, investigators at the Imperial College Healthcare NHS Trust, London, reported.
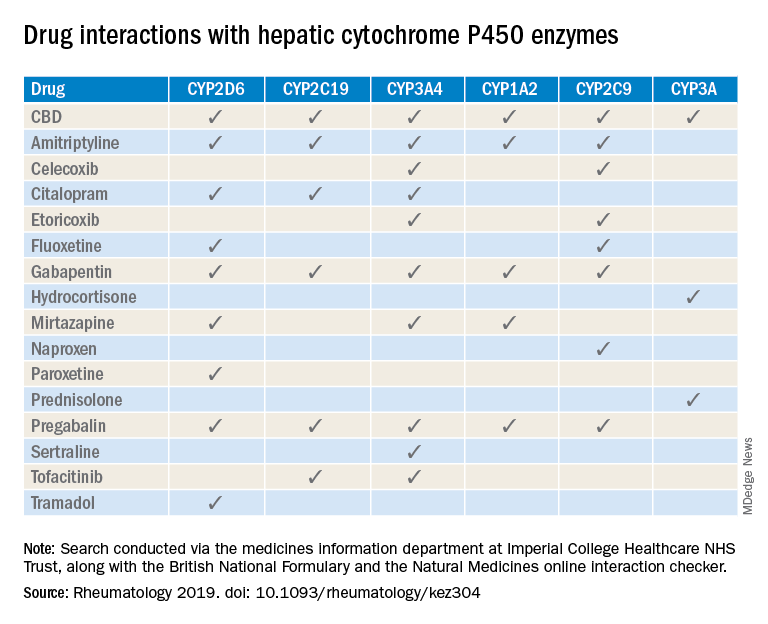
“Patients are increasingly requesting information concerning the safety of CBD oil,” Taryn Youngstein, MD, and associates said in letter to the editor in Rheumatology, but current guidelines on the use of medical cannabis do “not address the potential interactions between CBD oil and medicines frequently used in the rheumatology clinic.”
The most important potential CBD interaction, they suggested, may be with corticosteroids. Hydrocortisone and prednisolone both inhibit the cytochrome P450 enzyme CYP3A, but CBD is a potent inhibitor of CYP3A, so “concomitant use may decrease glucocorticoid clearance and increase risk of systemic [corticosteroid] side effects,” the investigators wrote.
CBD also is known to inhibit the cytochrome P450 isozymes CYP2C9, CYP2D6, CYP2C19, CYP3A4, and CYP1A2, which, alone or in combination, are involved in the metabolization of naproxen, tramadol, amitriptyline, and tofacitinib (Xeljanz), according to a literature search done via the college’s medicine information department that also used the British National Formulary and the Natural Medicines online interaction checker.
The Janus kinase inhibitor tofacitinib is included among the possible interactions, but the other Food and Drug Administration–approved JAK inhibitor, baricitinib (Olumiant), is primarily metabolized by the kidneys and should not have significant interaction with CBD, Dr. Youngstein and associates said. Most of the conventional synthetic and biologic disease-modifying antirheumatic drugs, including methotrexate, hydroxychloroquine, adalimumab (Humira), and abatacept (Orencia), also are expected to be relatively free from CBD interactions.
This first published report on interactions between CBD oil and common rheumatology medications “highlights the importance of taking comprehensive drug histories, by asking directly about drugs considered alternative medicines and food supplements,” they said.
The investigators declared no conflicts of interest, and there was no specific funding for the study.
SOURCE: Wilson-Morkeh H et al. Rheumatology. 2019 July 29. doi: 10.1093/rheumatology/kez304.
A number of medications commonly prescribed by rheumatologists may interact with cannabidiol oil, investigators at the Imperial College Healthcare NHS Trust, London, reported.

“Patients are increasingly requesting information concerning the safety of CBD oil,” Taryn Youngstein, MD, and associates said in letter to the editor in Rheumatology, but current guidelines on the use of medical cannabis do “not address the potential interactions between CBD oil and medicines frequently used in the rheumatology clinic.”
The most important potential CBD interaction, they suggested, may be with corticosteroids. Hydrocortisone and prednisolone both inhibit the cytochrome P450 enzyme CYP3A, but CBD is a potent inhibitor of CYP3A, so “concomitant use may decrease glucocorticoid clearance and increase risk of systemic [corticosteroid] side effects,” the investigators wrote.
CBD also is known to inhibit the cytochrome P450 isozymes CYP2C9, CYP2D6, CYP2C19, CYP3A4, and CYP1A2, which, alone or in combination, are involved in the metabolization of naproxen, tramadol, amitriptyline, and tofacitinib (Xeljanz), according to a literature search done via the college’s medicine information department that also used the British National Formulary and the Natural Medicines online interaction checker.
The Janus kinase inhibitor tofacitinib is included among the possible interactions, but the other Food and Drug Administration–approved JAK inhibitor, baricitinib (Olumiant), is primarily metabolized by the kidneys and should not have significant interaction with CBD, Dr. Youngstein and associates said. Most of the conventional synthetic and biologic disease-modifying antirheumatic drugs, including methotrexate, hydroxychloroquine, adalimumab (Humira), and abatacept (Orencia), also are expected to be relatively free from CBD interactions.
This first published report on interactions between CBD oil and common rheumatology medications “highlights the importance of taking comprehensive drug histories, by asking directly about drugs considered alternative medicines and food supplements,” they said.
The investigators declared no conflicts of interest, and there was no specific funding for the study.
SOURCE: Wilson-Morkeh H et al. Rheumatology. 2019 July 29. doi: 10.1093/rheumatology/kez304.
A number of medications commonly prescribed by rheumatologists may interact with cannabidiol oil, investigators at the Imperial College Healthcare NHS Trust, London, reported.

“Patients are increasingly requesting information concerning the safety of CBD oil,” Taryn Youngstein, MD, and associates said in letter to the editor in Rheumatology, but current guidelines on the use of medical cannabis do “not address the potential interactions between CBD oil and medicines frequently used in the rheumatology clinic.”
The most important potential CBD interaction, they suggested, may be with corticosteroids. Hydrocortisone and prednisolone both inhibit the cytochrome P450 enzyme CYP3A, but CBD is a potent inhibitor of CYP3A, so “concomitant use may decrease glucocorticoid clearance and increase risk of systemic [corticosteroid] side effects,” the investigators wrote.
CBD also is known to inhibit the cytochrome P450 isozymes CYP2C9, CYP2D6, CYP2C19, CYP3A4, and CYP1A2, which, alone or in combination, are involved in the metabolization of naproxen, tramadol, amitriptyline, and tofacitinib (Xeljanz), according to a literature search done via the college’s medicine information department that also used the British National Formulary and the Natural Medicines online interaction checker.
The Janus kinase inhibitor tofacitinib is included among the possible interactions, but the other Food and Drug Administration–approved JAK inhibitor, baricitinib (Olumiant), is primarily metabolized by the kidneys and should not have significant interaction with CBD, Dr. Youngstein and associates said. Most of the conventional synthetic and biologic disease-modifying antirheumatic drugs, including methotrexate, hydroxychloroquine, adalimumab (Humira), and abatacept (Orencia), also are expected to be relatively free from CBD interactions.
This first published report on interactions between CBD oil and common rheumatology medications “highlights the importance of taking comprehensive drug histories, by asking directly about drugs considered alternative medicines and food supplements,” they said.
The investigators declared no conflicts of interest, and there was no specific funding for the study.
SOURCE: Wilson-Morkeh H et al. Rheumatology. 2019 July 29. doi: 10.1093/rheumatology/kez304.
FROM RHEUMATOLOGY
EULAR updates vaccination recommendations for autoimmune inflammatory rheumatic disease patients
Vaccination status should be reviewed annually for patients with autoimmune inflammatory rheumatic diseases, according to updated recommendations from the European League Against Rheumatism.
Patients with autoimmune inflammatory rheumatic diseases (AIIRD) are at increased risk for infections, and vaccination has been shown to reduce risk by “potentially translating into a lower rate of hospital admissions due to infections, emergency room visits, and the rate of invasive infectious diseases,” wrote Victoria Furer, MD, of Tel Aviv Sourasky Medical Center, and members of the task force that updated the recommendations, which were published in Annals of the Rheumatic Diseases.
However, AIIRD patients often go unvaccinated because of a lack of awareness or concerns about vaccine safety and efficacy, they said (Ann Rheum Dis. 2019 Aug 14. doi: 10.1136/annrheumdis-2019-215882).
The task force consisted of 21 experts, including patients, rheumatologists, immunologists, an infectious disease specialist, and health professionals in rheumatology representing eight countries. They evaluated data from four systematic literature reviews and developed nine recommendations based on six key principles.
“For each recommendation, the level of evidence for the incidence/prevalence of vaccine preventable infection in AIIRD, and efficacy/immunogenicity/safety of vaccination were stated, when available, followed by the strength of recommendation and the level of agreement,” the task force wrote.
These overarching principles start with an annual assessment of vaccination status by the AIIRD patient’s rheumatology team. Other principles include explanation of an individualized vaccination program to the patient as a foundation for joint decision-making, vaccinating patients during quiescent disease periods, vaccinating in advance of planned immunosuppression when possible, considering non-live vaccines for AIIRD patients also treated with systemic glucocorticoids and DMARDs, and considering live-attenuated vaccines with caution.
Several of the nine recommendations developed by the task force are modified from the previous recommendations issued in 2011. The task force made its recommendations with an eye toward optimizing individual risk stratification and avoiding “unnecessary” vaccination in AIIRD patients with low risk of infection as part of the update process. A notable change from the 2011 guidelines is the recommendation of both influenza and pneumococcal vaccinations for the majority of patients with AIIRD as opposed to all patients to emphasize the importance of individualized risk assessment, the task force noted.
The recommendations state that influenza vaccination and pneumococcal vaccination should be “strongly considered” for patients with AIIRD, and patients also should receive tetanus toxoid vaccination according to recommendations for the general population. However, clinicians should consider passive immunization for patients treated with B-cell depleting therapy, the task force wrote.
AIIRD patients at risk for hepatitis A and B should receive vaccinations for those diseases, with boosters or passive immunization if indicated, and high-risk patients may consider herpes zoster vaccination, according to the recommendations.
In addition, AIIRD patients – especially patients with systemic lupus erythematosus – should receive human papilloma virus vaccination according to recommendations for the general population, but AIIRD patients should avoid yellow fever vaccination, the task force stated. However, for AIIRD patients traveling to areas of yellow fever risk, “withholding immunosuppressive therapy to allow a safe vaccination or measuring serology in previously exposed patients may be considered.”
Finally, mothers treated with biologics during the second half of pregnancy should avoid live-attenuated vaccines for their newborns, and immunocompetent household members of AIIRD patients should be encouraged to follow national guidelines for routine vaccination with the exception of the oral polio vaccine, the task force concluded.
Vaccination status should be reviewed annually for patients with autoimmune inflammatory rheumatic diseases, according to updated recommendations from the European League Against Rheumatism.
Patients with autoimmune inflammatory rheumatic diseases (AIIRD) are at increased risk for infections, and vaccination has been shown to reduce risk by “potentially translating into a lower rate of hospital admissions due to infections, emergency room visits, and the rate of invasive infectious diseases,” wrote Victoria Furer, MD, of Tel Aviv Sourasky Medical Center, and members of the task force that updated the recommendations, which were published in Annals of the Rheumatic Diseases.
However, AIIRD patients often go unvaccinated because of a lack of awareness or concerns about vaccine safety and efficacy, they said (Ann Rheum Dis. 2019 Aug 14. doi: 10.1136/annrheumdis-2019-215882).
The task force consisted of 21 experts, including patients, rheumatologists, immunologists, an infectious disease specialist, and health professionals in rheumatology representing eight countries. They evaluated data from four systematic literature reviews and developed nine recommendations based on six key principles.
“For each recommendation, the level of evidence for the incidence/prevalence of vaccine preventable infection in AIIRD, and efficacy/immunogenicity/safety of vaccination were stated, when available, followed by the strength of recommendation and the level of agreement,” the task force wrote.
These overarching principles start with an annual assessment of vaccination status by the AIIRD patient’s rheumatology team. Other principles include explanation of an individualized vaccination program to the patient as a foundation for joint decision-making, vaccinating patients during quiescent disease periods, vaccinating in advance of planned immunosuppression when possible, considering non-live vaccines for AIIRD patients also treated with systemic glucocorticoids and DMARDs, and considering live-attenuated vaccines with caution.
Several of the nine recommendations developed by the task force are modified from the previous recommendations issued in 2011. The task force made its recommendations with an eye toward optimizing individual risk stratification and avoiding “unnecessary” vaccination in AIIRD patients with low risk of infection as part of the update process. A notable change from the 2011 guidelines is the recommendation of both influenza and pneumococcal vaccinations for the majority of patients with AIIRD as opposed to all patients to emphasize the importance of individualized risk assessment, the task force noted.
The recommendations state that influenza vaccination and pneumococcal vaccination should be “strongly considered” for patients with AIIRD, and patients also should receive tetanus toxoid vaccination according to recommendations for the general population. However, clinicians should consider passive immunization for patients treated with B-cell depleting therapy, the task force wrote.
AIIRD patients at risk for hepatitis A and B should receive vaccinations for those diseases, with boosters or passive immunization if indicated, and high-risk patients may consider herpes zoster vaccination, according to the recommendations.
In addition, AIIRD patients – especially patients with systemic lupus erythematosus – should receive human papilloma virus vaccination according to recommendations for the general population, but AIIRD patients should avoid yellow fever vaccination, the task force stated. However, for AIIRD patients traveling to areas of yellow fever risk, “withholding immunosuppressive therapy to allow a safe vaccination or measuring serology in previously exposed patients may be considered.”
Finally, mothers treated with biologics during the second half of pregnancy should avoid live-attenuated vaccines for their newborns, and immunocompetent household members of AIIRD patients should be encouraged to follow national guidelines for routine vaccination with the exception of the oral polio vaccine, the task force concluded.
Vaccination status should be reviewed annually for patients with autoimmune inflammatory rheumatic diseases, according to updated recommendations from the European League Against Rheumatism.
Patients with autoimmune inflammatory rheumatic diseases (AIIRD) are at increased risk for infections, and vaccination has been shown to reduce risk by “potentially translating into a lower rate of hospital admissions due to infections, emergency room visits, and the rate of invasive infectious diseases,” wrote Victoria Furer, MD, of Tel Aviv Sourasky Medical Center, and members of the task force that updated the recommendations, which were published in Annals of the Rheumatic Diseases.
However, AIIRD patients often go unvaccinated because of a lack of awareness or concerns about vaccine safety and efficacy, they said (Ann Rheum Dis. 2019 Aug 14. doi: 10.1136/annrheumdis-2019-215882).
The task force consisted of 21 experts, including patients, rheumatologists, immunologists, an infectious disease specialist, and health professionals in rheumatology representing eight countries. They evaluated data from four systematic literature reviews and developed nine recommendations based on six key principles.
“For each recommendation, the level of evidence for the incidence/prevalence of vaccine preventable infection in AIIRD, and efficacy/immunogenicity/safety of vaccination were stated, when available, followed by the strength of recommendation and the level of agreement,” the task force wrote.
These overarching principles start with an annual assessment of vaccination status by the AIIRD patient’s rheumatology team. Other principles include explanation of an individualized vaccination program to the patient as a foundation for joint decision-making, vaccinating patients during quiescent disease periods, vaccinating in advance of planned immunosuppression when possible, considering non-live vaccines for AIIRD patients also treated with systemic glucocorticoids and DMARDs, and considering live-attenuated vaccines with caution.
Several of the nine recommendations developed by the task force are modified from the previous recommendations issued in 2011. The task force made its recommendations with an eye toward optimizing individual risk stratification and avoiding “unnecessary” vaccination in AIIRD patients with low risk of infection as part of the update process. A notable change from the 2011 guidelines is the recommendation of both influenza and pneumococcal vaccinations for the majority of patients with AIIRD as opposed to all patients to emphasize the importance of individualized risk assessment, the task force noted.
The recommendations state that influenza vaccination and pneumococcal vaccination should be “strongly considered” for patients with AIIRD, and patients also should receive tetanus toxoid vaccination according to recommendations for the general population. However, clinicians should consider passive immunization for patients treated with B-cell depleting therapy, the task force wrote.
AIIRD patients at risk for hepatitis A and B should receive vaccinations for those diseases, with boosters or passive immunization if indicated, and high-risk patients may consider herpes zoster vaccination, according to the recommendations.
In addition, AIIRD patients – especially patients with systemic lupus erythematosus – should receive human papilloma virus vaccination according to recommendations for the general population, but AIIRD patients should avoid yellow fever vaccination, the task force stated. However, for AIIRD patients traveling to areas of yellow fever risk, “withholding immunosuppressive therapy to allow a safe vaccination or measuring serology in previously exposed patients may be considered.”
Finally, mothers treated with biologics during the second half of pregnancy should avoid live-attenuated vaccines for their newborns, and immunocompetent household members of AIIRD patients should be encouraged to follow national guidelines for routine vaccination with the exception of the oral polio vaccine, the task force concluded.
FROM ANNALS OF THE RHEUMATIC DISEASES
PROMIS tools provide useful data for managing rheumatology patients
LAKE BUENA VISTA, FLA. –
The PROMIS tools – which like most patient-reported outcome (PRO) measurement tools are designed to evaluate and monitor physical, mental, and social health – can be used both for the general population and for individuals living with chronic conditions, Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham (UAB), said at the annual meeting of the Florida Society of Rheumatology.
The tools take a deeper dive into various symptoms and their effects; for instance, with respect to physical health, they measure fatigue, physical function, sleep disturbance, pain intensity, and pain interference – the extent to which pain “messes your patient’s life up,” explained Dr. Curtis, who also is codirector of the UAB Pharmacoepidemiology and Pharmacoeconomics Unit.
Additional physical health domains that PROs measure include dyspnea, gastrointestinal symptoms, pain behavior, pain quality, sexual function, and sleep-related impairment.
These are “things that, honestly, we don’t talk about much as a field, but absolutely affect patients with autoimmune diseases,” he said. “You know, sexual function – that doesn’t come up in my practice spontaneously very often, but there are ways you can quantify that, and for many patients that’s actually a big deal.”
The domains measured by PROMIS tools for mental health look at anxiety and depression, but also delve into alcohol use, anger, cognitive function, life satisfaction, self-efficacy for managing chronic conditions, substance use, and more. The domains for social health address ability to participate in social roles and activities, as well as companionship, satisfaction with social roles and activity, social isolation, and social support.
“You can’t go on a hike with friends [and] be far from a bathroom, because you have bad arthritis and you have Crohn’s disease. Well, that’s kind of an important thing that may or may not come up in your discussions about inflammatory arthritis associated with [inflammatory bowel disease],” he said.
Another example is a patient who is embarrassed attending social functions or wearing a swimsuit because of really bad psoriasis.
“These are the kinds of things that I’m suggesting you and I probably want to measure if we’re providing holistic care to rheumatology patients,” Dr. Curtis said.
The PROMIS tools provide a simple, user-friendly means for doing so in English, Spanish, and many other languages, he noted.
All the scales use the same 1-100 scoring range, which simplifies measurements. They are available for free by download and can be printed or used electronically for use in the office, at home, on the web, and via smartphone.
The NIH developed the PROMIS tools several years ago and validated them for multiple chronic disease populations, Dr. Curtis said, adding that the tools include multiple individual domains and overall “profiles” of varying lengths.
Most are fixed-length scales that are between 4 and 10 questions and can be completed within 30-60 seconds per scale, so several scales can be completed within 5-10 minutes.
However, some scales are longer and provide greater detail.
“The nice thing is that if you ask a few more questions you can get more precise information – there’s more of a floor and ceiling. You can detect people who do really well. You can distinguish between the marathon runners and the 5K-ers and the people who can walk 2 miles but aren’t going to run a race,” he explained.
Further, the PROMIS tools, like the 36-item Short Form Health Survey (SF-36), are benchmarked against the U.S. adult population, allowing for assessment of how a specific drug or treatment “impacts your arthritis patient on a scale that would also be relevant for somebody who doesn’t have arthritis, they have diabetes.”
The metrics and scales are the same, and that can be helpful when trying to get a payer to pay for a particular drug, he said.
“None of these are rheumatology specific; this puts PROs into a language that can help rheumatology contend for the value of the care that we provide on a scale that would be relevant for any other chronic illness, even for nonrheumatology patients,” he explained.
In addition, minimally important differences (group mean change of about 2-3 units) and minimally clinical important differences for individuals (5 units) have been established.
“So we know what the numbers mean, and this is true for all of the scales,” he said.
PROMIS tools also include computer-adaptive testing (CAT) versions, which helps to personalize the scales to provide more precise information for a given patient and eliminate irrelevant information.
Of note, PROMIS health measures are among the data that can be tracked on a smartphone using Arthritis Power, an arthritis research registry developed with the help of a recent infrastructure grant awarded to the Center for Education and Research and Therapeutics of Musculoskeletal Disorders at UAB, Dr. Curtis said.
The measures were also shown in the AWARE study to track closely with other measures, including the Clinical Disease Activity Index (CDAI), and with patient improvement on therapy.
“So these PROMIS scores are tracking with things that you and I are familiar with ... and it looks like these scores are faithfully tracking, over time, patients getting better on therapies that we would expect them to,” he said. “I think this is additional validation – not just from the National Institutes of Health and a decade of research by lots of different groups, but in our own field – that these actually correlate with disease activity ... and that when you start an effective therapy like a [tumor necrosis factor inhibitor] they’re going to improve as you would anticipate.”
Dr. Curtis reported funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Patient-Centered Outcomes Research Institute. He has also consulted for or received research grants from Amgen, AbbVie, Bristol-Myers Squibb, CORRONA, Lilly, Janssen, Myriad, Novartis, Roche, Pfizer, and Sanofi/Regeneron.
LAKE BUENA VISTA, FLA. –
The PROMIS tools – which like most patient-reported outcome (PRO) measurement tools are designed to evaluate and monitor physical, mental, and social health – can be used both for the general population and for individuals living with chronic conditions, Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham (UAB), said at the annual meeting of the Florida Society of Rheumatology.
The tools take a deeper dive into various symptoms and their effects; for instance, with respect to physical health, they measure fatigue, physical function, sleep disturbance, pain intensity, and pain interference – the extent to which pain “messes your patient’s life up,” explained Dr. Curtis, who also is codirector of the UAB Pharmacoepidemiology and Pharmacoeconomics Unit.
Additional physical health domains that PROs measure include dyspnea, gastrointestinal symptoms, pain behavior, pain quality, sexual function, and sleep-related impairment.
These are “things that, honestly, we don’t talk about much as a field, but absolutely affect patients with autoimmune diseases,” he said. “You know, sexual function – that doesn’t come up in my practice spontaneously very often, but there are ways you can quantify that, and for many patients that’s actually a big deal.”
The domains measured by PROMIS tools for mental health look at anxiety and depression, but also delve into alcohol use, anger, cognitive function, life satisfaction, self-efficacy for managing chronic conditions, substance use, and more. The domains for social health address ability to participate in social roles and activities, as well as companionship, satisfaction with social roles and activity, social isolation, and social support.
“You can’t go on a hike with friends [and] be far from a bathroom, because you have bad arthritis and you have Crohn’s disease. Well, that’s kind of an important thing that may or may not come up in your discussions about inflammatory arthritis associated with [inflammatory bowel disease],” he said.
Another example is a patient who is embarrassed attending social functions or wearing a swimsuit because of really bad psoriasis.
“These are the kinds of things that I’m suggesting you and I probably want to measure if we’re providing holistic care to rheumatology patients,” Dr. Curtis said.
The PROMIS tools provide a simple, user-friendly means for doing so in English, Spanish, and many other languages, he noted.
All the scales use the same 1-100 scoring range, which simplifies measurements. They are available for free by download and can be printed or used electronically for use in the office, at home, on the web, and via smartphone.
The NIH developed the PROMIS tools several years ago and validated them for multiple chronic disease populations, Dr. Curtis said, adding that the tools include multiple individual domains and overall “profiles” of varying lengths.
Most are fixed-length scales that are between 4 and 10 questions and can be completed within 30-60 seconds per scale, so several scales can be completed within 5-10 minutes.
However, some scales are longer and provide greater detail.
“The nice thing is that if you ask a few more questions you can get more precise information – there’s more of a floor and ceiling. You can detect people who do really well. You can distinguish between the marathon runners and the 5K-ers and the people who can walk 2 miles but aren’t going to run a race,” he explained.
Further, the PROMIS tools, like the 36-item Short Form Health Survey (SF-36), are benchmarked against the U.S. adult population, allowing for assessment of how a specific drug or treatment “impacts your arthritis patient on a scale that would also be relevant for somebody who doesn’t have arthritis, they have diabetes.”
The metrics and scales are the same, and that can be helpful when trying to get a payer to pay for a particular drug, he said.
“None of these are rheumatology specific; this puts PROs into a language that can help rheumatology contend for the value of the care that we provide on a scale that would be relevant for any other chronic illness, even for nonrheumatology patients,” he explained.
In addition, minimally important differences (group mean change of about 2-3 units) and minimally clinical important differences for individuals (5 units) have been established.
“So we know what the numbers mean, and this is true for all of the scales,” he said.
PROMIS tools also include computer-adaptive testing (CAT) versions, which helps to personalize the scales to provide more precise information for a given patient and eliminate irrelevant information.
Of note, PROMIS health measures are among the data that can be tracked on a smartphone using Arthritis Power, an arthritis research registry developed with the help of a recent infrastructure grant awarded to the Center for Education and Research and Therapeutics of Musculoskeletal Disorders at UAB, Dr. Curtis said.
The measures were also shown in the AWARE study to track closely with other measures, including the Clinical Disease Activity Index (CDAI), and with patient improvement on therapy.
“So these PROMIS scores are tracking with things that you and I are familiar with ... and it looks like these scores are faithfully tracking, over time, patients getting better on therapies that we would expect them to,” he said. “I think this is additional validation – not just from the National Institutes of Health and a decade of research by lots of different groups, but in our own field – that these actually correlate with disease activity ... and that when you start an effective therapy like a [tumor necrosis factor inhibitor] they’re going to improve as you would anticipate.”
Dr. Curtis reported funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Patient-Centered Outcomes Research Institute. He has also consulted for or received research grants from Amgen, AbbVie, Bristol-Myers Squibb, CORRONA, Lilly, Janssen, Myriad, Novartis, Roche, Pfizer, and Sanofi/Regeneron.
LAKE BUENA VISTA, FLA. –
The PROMIS tools – which like most patient-reported outcome (PRO) measurement tools are designed to evaluate and monitor physical, mental, and social health – can be used both for the general population and for individuals living with chronic conditions, Dr. Curtis, professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham (UAB), said at the annual meeting of the Florida Society of Rheumatology.
The tools take a deeper dive into various symptoms and their effects; for instance, with respect to physical health, they measure fatigue, physical function, sleep disturbance, pain intensity, and pain interference – the extent to which pain “messes your patient’s life up,” explained Dr. Curtis, who also is codirector of the UAB Pharmacoepidemiology and Pharmacoeconomics Unit.
Additional physical health domains that PROs measure include dyspnea, gastrointestinal symptoms, pain behavior, pain quality, sexual function, and sleep-related impairment.
These are “things that, honestly, we don’t talk about much as a field, but absolutely affect patients with autoimmune diseases,” he said. “You know, sexual function – that doesn’t come up in my practice spontaneously very often, but there are ways you can quantify that, and for many patients that’s actually a big deal.”
The domains measured by PROMIS tools for mental health look at anxiety and depression, but also delve into alcohol use, anger, cognitive function, life satisfaction, self-efficacy for managing chronic conditions, substance use, and more. The domains for social health address ability to participate in social roles and activities, as well as companionship, satisfaction with social roles and activity, social isolation, and social support.
“You can’t go on a hike with friends [and] be far from a bathroom, because you have bad arthritis and you have Crohn’s disease. Well, that’s kind of an important thing that may or may not come up in your discussions about inflammatory arthritis associated with [inflammatory bowel disease],” he said.
Another example is a patient who is embarrassed attending social functions or wearing a swimsuit because of really bad psoriasis.
“These are the kinds of things that I’m suggesting you and I probably want to measure if we’re providing holistic care to rheumatology patients,” Dr. Curtis said.
The PROMIS tools provide a simple, user-friendly means for doing so in English, Spanish, and many other languages, he noted.
All the scales use the same 1-100 scoring range, which simplifies measurements. They are available for free by download and can be printed or used electronically for use in the office, at home, on the web, and via smartphone.
The NIH developed the PROMIS tools several years ago and validated them for multiple chronic disease populations, Dr. Curtis said, adding that the tools include multiple individual domains and overall “profiles” of varying lengths.
Most are fixed-length scales that are between 4 and 10 questions and can be completed within 30-60 seconds per scale, so several scales can be completed within 5-10 minutes.
However, some scales are longer and provide greater detail.
“The nice thing is that if you ask a few more questions you can get more precise information – there’s more of a floor and ceiling. You can detect people who do really well. You can distinguish between the marathon runners and the 5K-ers and the people who can walk 2 miles but aren’t going to run a race,” he explained.
Further, the PROMIS tools, like the 36-item Short Form Health Survey (SF-36), are benchmarked against the U.S. adult population, allowing for assessment of how a specific drug or treatment “impacts your arthritis patient on a scale that would also be relevant for somebody who doesn’t have arthritis, they have diabetes.”
The metrics and scales are the same, and that can be helpful when trying to get a payer to pay for a particular drug, he said.
“None of these are rheumatology specific; this puts PROs into a language that can help rheumatology contend for the value of the care that we provide on a scale that would be relevant for any other chronic illness, even for nonrheumatology patients,” he explained.
In addition, minimally important differences (group mean change of about 2-3 units) and minimally clinical important differences for individuals (5 units) have been established.
“So we know what the numbers mean, and this is true for all of the scales,” he said.
PROMIS tools also include computer-adaptive testing (CAT) versions, which helps to personalize the scales to provide more precise information for a given patient and eliminate irrelevant information.
Of note, PROMIS health measures are among the data that can be tracked on a smartphone using Arthritis Power, an arthritis research registry developed with the help of a recent infrastructure grant awarded to the Center for Education and Research and Therapeutics of Musculoskeletal Disorders at UAB, Dr. Curtis said.
The measures were also shown in the AWARE study to track closely with other measures, including the Clinical Disease Activity Index (CDAI), and with patient improvement on therapy.
“So these PROMIS scores are tracking with things that you and I are familiar with ... and it looks like these scores are faithfully tracking, over time, patients getting better on therapies that we would expect them to,” he said. “I think this is additional validation – not just from the National Institutes of Health and a decade of research by lots of different groups, but in our own field – that these actually correlate with disease activity ... and that when you start an effective therapy like a [tumor necrosis factor inhibitor] they’re going to improve as you would anticipate.”
Dr. Curtis reported funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Patient-Centered Outcomes Research Institute. He has also consulted for or received research grants from Amgen, AbbVie, Bristol-Myers Squibb, CORRONA, Lilly, Janssen, Myriad, Novartis, Roche, Pfizer, and Sanofi/Regeneron.
EXPERT ANALYSIS FROM FSR 2019
Psoriasis patients on biologics show improved heart health
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
FROM JAMA CARDIOLOGY
FDA finds increased blood clot, death risk associated with Xeljanz
The Food and Drug Administration has issued a safety alert approving new boxed warnings about increased blood clot and mortality risk associated with the 10-mg, twice-daily dose of tofacitinib (Xeljanz), as well as a new limitation for patients with ulcerative colitis receiving the medication.
Tofacitinib, a Janus kinase inhibitor, was first approved by the FDA in 2012 for the treatment of rheumatoid arthritis (RA). An indication for psoriatic arthritis was added in 2017, and one for ulcerative colitis was added in 2018.
After the 2012 approval, the FDA commissioned a postmarketing trial in patients with RA on background methotrexate to evaluate safety and the risk of cancer, heart-related events, and infection. The 5- and 10-mg tofacitinib twice daily doses are being analyzed in an ongoing study in comparison with a tumor necrosis factor (TNF) inhibitor.
An interim analysis of the trial’s data, as of January 2019, found an increased risk of blood clots and death in patients receiving 10-mg tofacitinib twice daily, compared with the TNF inhibitor and the twice-daily, 5-mg dose. Overall, there were 19 cases of blood clots in the lung out of 3,884 patient-years of follow-up in patients who received tofacitinib 10 mg twice daily, compared with 3 cases out of 3,982 patient-years in patients who received TNF inhibitors. There were also 45 cases of death from all causes during follow-up for tofacitinib 10 mg twice daily, compared with 25 cases in patients who received TNF inhibitors.
Patients with symptoms of thrombosis also receiving tofacitinib should immediately discontinue the medication. Tofacitinib should not be given to patients with ulcerative colitis unless they are not treated effectively with a TNF inhibitor or do not tolerate TNF inhibitors; ulcerative colitis patients should receive the lowest effective dosage, and if the higher dosage is necessary, it should be limited to the shortest amount of time possible, the FDA noted.
The Food and Drug Administration has issued a safety alert approving new boxed warnings about increased blood clot and mortality risk associated with the 10-mg, twice-daily dose of tofacitinib (Xeljanz), as well as a new limitation for patients with ulcerative colitis receiving the medication.
Tofacitinib, a Janus kinase inhibitor, was first approved by the FDA in 2012 for the treatment of rheumatoid arthritis (RA). An indication for psoriatic arthritis was added in 2017, and one for ulcerative colitis was added in 2018.
After the 2012 approval, the FDA commissioned a postmarketing trial in patients with RA on background methotrexate to evaluate safety and the risk of cancer, heart-related events, and infection. The 5- and 10-mg tofacitinib twice daily doses are being analyzed in an ongoing study in comparison with a tumor necrosis factor (TNF) inhibitor.
An interim analysis of the trial’s data, as of January 2019, found an increased risk of blood clots and death in patients receiving 10-mg tofacitinib twice daily, compared with the TNF inhibitor and the twice-daily, 5-mg dose. Overall, there were 19 cases of blood clots in the lung out of 3,884 patient-years of follow-up in patients who received tofacitinib 10 mg twice daily, compared with 3 cases out of 3,982 patient-years in patients who received TNF inhibitors. There were also 45 cases of death from all causes during follow-up for tofacitinib 10 mg twice daily, compared with 25 cases in patients who received TNF inhibitors.
Patients with symptoms of thrombosis also receiving tofacitinib should immediately discontinue the medication. Tofacitinib should not be given to patients with ulcerative colitis unless they are not treated effectively with a TNF inhibitor or do not tolerate TNF inhibitors; ulcerative colitis patients should receive the lowest effective dosage, and if the higher dosage is necessary, it should be limited to the shortest amount of time possible, the FDA noted.
The Food and Drug Administration has issued a safety alert approving new boxed warnings about increased blood clot and mortality risk associated with the 10-mg, twice-daily dose of tofacitinib (Xeljanz), as well as a new limitation for patients with ulcerative colitis receiving the medication.
Tofacitinib, a Janus kinase inhibitor, was first approved by the FDA in 2012 for the treatment of rheumatoid arthritis (RA). An indication for psoriatic arthritis was added in 2017, and one for ulcerative colitis was added in 2018.
After the 2012 approval, the FDA commissioned a postmarketing trial in patients with RA on background methotrexate to evaluate safety and the risk of cancer, heart-related events, and infection. The 5- and 10-mg tofacitinib twice daily doses are being analyzed in an ongoing study in comparison with a tumor necrosis factor (TNF) inhibitor.
An interim analysis of the trial’s data, as of January 2019, found an increased risk of blood clots and death in patients receiving 10-mg tofacitinib twice daily, compared with the TNF inhibitor and the twice-daily, 5-mg dose. Overall, there were 19 cases of blood clots in the lung out of 3,884 patient-years of follow-up in patients who received tofacitinib 10 mg twice daily, compared with 3 cases out of 3,982 patient-years in patients who received TNF inhibitors. There were also 45 cases of death from all causes during follow-up for tofacitinib 10 mg twice daily, compared with 25 cases in patients who received TNF inhibitors.
Patients with symptoms of thrombosis also receiving tofacitinib should immediately discontinue the medication. Tofacitinib should not be given to patients with ulcerative colitis unless they are not treated effectively with a TNF inhibitor or do not tolerate TNF inhibitors; ulcerative colitis patients should receive the lowest effective dosage, and if the higher dosage is necessary, it should be limited to the shortest amount of time possible, the FDA noted.