User login
Guselkumab’s efficacy, safety confirmed in patients with psoriatic arthritis and prior TNFi exposure
A new study has established guselkumab (Tremfya) as both a safe and effective treatment option for psoriatic arthritis (PsA) in patients who had previously responded poorly to tumor necrosis factor inhibitors (TNFis).
“While the positive guselkumab benefit-risk profile observed through week 24 was maintained through 1 year, real-world evidence will further inform long-term guselkumab persistence in TNFi-inadequate response patients,” writes Laura C. Coates, MBChB, PhD, of the University of Oxford (England), and her coauthors. The study was published in the Annals of the Rheumatic Diseases.
Previous studies indicated that the anti–interleukin-23p19 monoclonal antibody improved outcomes in patients with PsA, even after 1 year, but some uncertainty remained regarding the surprisingly similar level of effectiveness in biologic-naive and TNFi-treated patients. Guselkumab is approved for treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy and adults with active psoriatic arthritis.
Clarity on guselkumab’s effectiveness in certain patients
“In previous studies that cemented guselkumab as a treatment option for PsA, what was odd was that the results were pretty comparable,” Eric M. Ruderman, MD, professor of medicine and associate chief for clinical affairs in the division of rheumatology at Northwestern University in Chicago, Illinois, said in an interview. “We didn’t really have a sense of how well it worked in patients who had failed other biologics, which is where you might expect a drug with a new mechanism to be used when it comes into a particular disease category.
“Not surprisingly, in this study, the overall response rate was a little less than the response rate in the other two trials,” said Dr. Ruderman, who was not involved in the study. “You can’t really compare across studies, but it does fit with what we might expect: People who’ve previously failed a TNF inhibitor might be a little less likely to respond to guselkumab, compared to someone who hasn’t seen a TNF inhibitor.”
When asked about potential follow-up studies, Dr. Ruderman noted that “the missing piece of the puzzle is that we still really have no way to compare this to other biologics. The next step would be to ask, in a single trial, what happens if you give some people TNF inhibitors and some people guselkumab? Just to try to give us context. Is this equivalent? Is it less effective? More effective? Where does it fit? Without that information, rheumatologists may struggle to figure out who is the right person for this drug and how often should they use it.”
Study details
To assess the efficacy and safety of guselkumab in patients who had previously taken TNFis but stopped because of inefficacy or intolerance, the researchers launched a randomized, double-blind study called COSMOS at 84 European sites from March 2019 to November 2020. The study’s 285 patients – 52% of whom were women, with an average overall age of 49 – were assigned to two groups: guselkumab (n = 189) or placebo (n = 96). A total of 88% of all patients had used one TNFi prior; 12% had used two.
The guselkumab group received 100-mg injections at week 0, week 4, and then every 8 weeks through week 44; the placebo group received injections at weeks 0, 4, 12, and 20, followed by 100 mg of guselkumab at weeks 24, 28, 36, and 44. Patients with less than 5% improvement from baseline in both tender and swollen joint counts at week 16 qualified for early escape to “initiate or increase the dose of one permitted concomitant medication up to the maximum allowed dose at the physician’s discretion.” Ultimately, 88% of patients in the guselkumab arm and 83% of the placebo arm completed the study.
At 24 weeks, more than 44% of the guselkumab group achieved a 20% or greater improvement in American College of Rheumatology criteria (ACR20), compared with just under 20% of the placebo group, a difference of nearly 25% (95% confidence interval, 14.1%-35.2%; multiplicity-adjusted P < .001). At 48 weeks, nearly 58% of the guselkumab group had achieved ACR20; of the 51 patients in the placebo arm who started taking guselkumab at week 24, 55% achieved ACR20 by week 48.
Through 24 weeks, 80 patients in the guselkumab group (42%) and 46 patients in the placebo group (48%) experienced adverse events; only 3.7% and 3.1% developed serious adverse events, respectively. The most common adverse events in the guselkumab group at that point included nasopharyngitis (5%) and upper respiratory tract infection (4%), which occurred at a similar frequency (5% and 3%) in the placebo group.
The authors acknowledge their study’s limitations, including imbalances in baseline characteristics such as gender and weight, as well as the COSMOS study being restricted to European patients and thus potentially limiting diversity. In addition, while the COVID-19 pandemic may have increased major protocol deviations near the end of the study, the authors note that “most were related to timing of study visits and did not impact efficacy.”
The study was funded by Janssen, and six authors reported being employees of the company. The authors also acknowledge numerous potential conflicts of interest, including receiving consulting fees and research grants from various pharmaceutical companies, including Janssen. Dr. Ruderman is a consultant for AbbVie, Bristol-Myers Squibb, Eli Lilly, Novartis, Pfizer, and Janssen and served on the data safety monitoring committee for two other phase 3 guselkumab trials.
A version of this article first appeared on Medscape.com.
A new study has established guselkumab (Tremfya) as both a safe and effective treatment option for psoriatic arthritis (PsA) in patients who had previously responded poorly to tumor necrosis factor inhibitors (TNFis).
“While the positive guselkumab benefit-risk profile observed through week 24 was maintained through 1 year, real-world evidence will further inform long-term guselkumab persistence in TNFi-inadequate response patients,” writes Laura C. Coates, MBChB, PhD, of the University of Oxford (England), and her coauthors. The study was published in the Annals of the Rheumatic Diseases.
Previous studies indicated that the anti–interleukin-23p19 monoclonal antibody improved outcomes in patients with PsA, even after 1 year, but some uncertainty remained regarding the surprisingly similar level of effectiveness in biologic-naive and TNFi-treated patients. Guselkumab is approved for treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy and adults with active psoriatic arthritis.
Clarity on guselkumab’s effectiveness in certain patients
“In previous studies that cemented guselkumab as a treatment option for PsA, what was odd was that the results were pretty comparable,” Eric M. Ruderman, MD, professor of medicine and associate chief for clinical affairs in the division of rheumatology at Northwestern University in Chicago, Illinois, said in an interview. “We didn’t really have a sense of how well it worked in patients who had failed other biologics, which is where you might expect a drug with a new mechanism to be used when it comes into a particular disease category.
“Not surprisingly, in this study, the overall response rate was a little less than the response rate in the other two trials,” said Dr. Ruderman, who was not involved in the study. “You can’t really compare across studies, but it does fit with what we might expect: People who’ve previously failed a TNF inhibitor might be a little less likely to respond to guselkumab, compared to someone who hasn’t seen a TNF inhibitor.”
When asked about potential follow-up studies, Dr. Ruderman noted that “the missing piece of the puzzle is that we still really have no way to compare this to other biologics. The next step would be to ask, in a single trial, what happens if you give some people TNF inhibitors and some people guselkumab? Just to try to give us context. Is this equivalent? Is it less effective? More effective? Where does it fit? Without that information, rheumatologists may struggle to figure out who is the right person for this drug and how often should they use it.”
Study details
To assess the efficacy and safety of guselkumab in patients who had previously taken TNFis but stopped because of inefficacy or intolerance, the researchers launched a randomized, double-blind study called COSMOS at 84 European sites from March 2019 to November 2020. The study’s 285 patients – 52% of whom were women, with an average overall age of 49 – were assigned to two groups: guselkumab (n = 189) or placebo (n = 96). A total of 88% of all patients had used one TNFi prior; 12% had used two.
The guselkumab group received 100-mg injections at week 0, week 4, and then every 8 weeks through week 44; the placebo group received injections at weeks 0, 4, 12, and 20, followed by 100 mg of guselkumab at weeks 24, 28, 36, and 44. Patients with less than 5% improvement from baseline in both tender and swollen joint counts at week 16 qualified for early escape to “initiate or increase the dose of one permitted concomitant medication up to the maximum allowed dose at the physician’s discretion.” Ultimately, 88% of patients in the guselkumab arm and 83% of the placebo arm completed the study.
At 24 weeks, more than 44% of the guselkumab group achieved a 20% or greater improvement in American College of Rheumatology criteria (ACR20), compared with just under 20% of the placebo group, a difference of nearly 25% (95% confidence interval, 14.1%-35.2%; multiplicity-adjusted P < .001). At 48 weeks, nearly 58% of the guselkumab group had achieved ACR20; of the 51 patients in the placebo arm who started taking guselkumab at week 24, 55% achieved ACR20 by week 48.
Through 24 weeks, 80 patients in the guselkumab group (42%) and 46 patients in the placebo group (48%) experienced adverse events; only 3.7% and 3.1% developed serious adverse events, respectively. The most common adverse events in the guselkumab group at that point included nasopharyngitis (5%) and upper respiratory tract infection (4%), which occurred at a similar frequency (5% and 3%) in the placebo group.
The authors acknowledge their study’s limitations, including imbalances in baseline characteristics such as gender and weight, as well as the COSMOS study being restricted to European patients and thus potentially limiting diversity. In addition, while the COVID-19 pandemic may have increased major protocol deviations near the end of the study, the authors note that “most were related to timing of study visits and did not impact efficacy.”
The study was funded by Janssen, and six authors reported being employees of the company. The authors also acknowledge numerous potential conflicts of interest, including receiving consulting fees and research grants from various pharmaceutical companies, including Janssen. Dr. Ruderman is a consultant for AbbVie, Bristol-Myers Squibb, Eli Lilly, Novartis, Pfizer, and Janssen and served on the data safety monitoring committee for two other phase 3 guselkumab trials.
A version of this article first appeared on Medscape.com.
A new study has established guselkumab (Tremfya) as both a safe and effective treatment option for psoriatic arthritis (PsA) in patients who had previously responded poorly to tumor necrosis factor inhibitors (TNFis).
“While the positive guselkumab benefit-risk profile observed through week 24 was maintained through 1 year, real-world evidence will further inform long-term guselkumab persistence in TNFi-inadequate response patients,” writes Laura C. Coates, MBChB, PhD, of the University of Oxford (England), and her coauthors. The study was published in the Annals of the Rheumatic Diseases.
Previous studies indicated that the anti–interleukin-23p19 monoclonal antibody improved outcomes in patients with PsA, even after 1 year, but some uncertainty remained regarding the surprisingly similar level of effectiveness in biologic-naive and TNFi-treated patients. Guselkumab is approved for treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy and adults with active psoriatic arthritis.
Clarity on guselkumab’s effectiveness in certain patients
“In previous studies that cemented guselkumab as a treatment option for PsA, what was odd was that the results were pretty comparable,” Eric M. Ruderman, MD, professor of medicine and associate chief for clinical affairs in the division of rheumatology at Northwestern University in Chicago, Illinois, said in an interview. “We didn’t really have a sense of how well it worked in patients who had failed other biologics, which is where you might expect a drug with a new mechanism to be used when it comes into a particular disease category.
“Not surprisingly, in this study, the overall response rate was a little less than the response rate in the other two trials,” said Dr. Ruderman, who was not involved in the study. “You can’t really compare across studies, but it does fit with what we might expect: People who’ve previously failed a TNF inhibitor might be a little less likely to respond to guselkumab, compared to someone who hasn’t seen a TNF inhibitor.”
When asked about potential follow-up studies, Dr. Ruderman noted that “the missing piece of the puzzle is that we still really have no way to compare this to other biologics. The next step would be to ask, in a single trial, what happens if you give some people TNF inhibitors and some people guselkumab? Just to try to give us context. Is this equivalent? Is it less effective? More effective? Where does it fit? Without that information, rheumatologists may struggle to figure out who is the right person for this drug and how often should they use it.”
Study details
To assess the efficacy and safety of guselkumab in patients who had previously taken TNFis but stopped because of inefficacy or intolerance, the researchers launched a randomized, double-blind study called COSMOS at 84 European sites from March 2019 to November 2020. The study’s 285 patients – 52% of whom were women, with an average overall age of 49 – were assigned to two groups: guselkumab (n = 189) or placebo (n = 96). A total of 88% of all patients had used one TNFi prior; 12% had used two.
The guselkumab group received 100-mg injections at week 0, week 4, and then every 8 weeks through week 44; the placebo group received injections at weeks 0, 4, 12, and 20, followed by 100 mg of guselkumab at weeks 24, 28, 36, and 44. Patients with less than 5% improvement from baseline in both tender and swollen joint counts at week 16 qualified for early escape to “initiate or increase the dose of one permitted concomitant medication up to the maximum allowed dose at the physician’s discretion.” Ultimately, 88% of patients in the guselkumab arm and 83% of the placebo arm completed the study.
At 24 weeks, more than 44% of the guselkumab group achieved a 20% or greater improvement in American College of Rheumatology criteria (ACR20), compared with just under 20% of the placebo group, a difference of nearly 25% (95% confidence interval, 14.1%-35.2%; multiplicity-adjusted P < .001). At 48 weeks, nearly 58% of the guselkumab group had achieved ACR20; of the 51 patients in the placebo arm who started taking guselkumab at week 24, 55% achieved ACR20 by week 48.
Through 24 weeks, 80 patients in the guselkumab group (42%) and 46 patients in the placebo group (48%) experienced adverse events; only 3.7% and 3.1% developed serious adverse events, respectively. The most common adverse events in the guselkumab group at that point included nasopharyngitis (5%) and upper respiratory tract infection (4%), which occurred at a similar frequency (5% and 3%) in the placebo group.
The authors acknowledge their study’s limitations, including imbalances in baseline characteristics such as gender and weight, as well as the COSMOS study being restricted to European patients and thus potentially limiting diversity. In addition, while the COVID-19 pandemic may have increased major protocol deviations near the end of the study, the authors note that “most were related to timing of study visits and did not impact efficacy.”
The study was funded by Janssen, and six authors reported being employees of the company. The authors also acknowledge numerous potential conflicts of interest, including receiving consulting fees and research grants from various pharmaceutical companies, including Janssen. Dr. Ruderman is a consultant for AbbVie, Bristol-Myers Squibb, Eli Lilly, Novartis, Pfizer, and Janssen and served on the data safety monitoring committee for two other phase 3 guselkumab trials.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Expert shares top five atopic dermatitis–related questions he fields
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
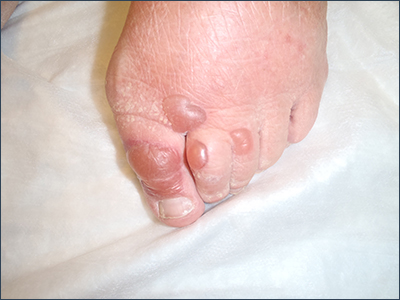
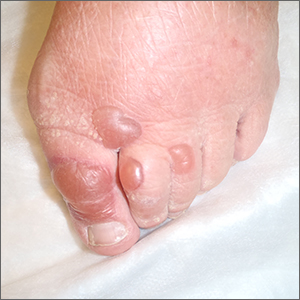
Bullae on elderly woman’s toes
A biopsy was performed and sent for immunofluorescence; the results were negative. This, along with the patient’s history of diabetes, led us to the diagnosis of bullosis diabeticorum (BD). This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes.
The etiology of BD is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites.
A diagnosis of BD can be made when biopsy with immunofluorescence excludes other histologically similar diagnoses such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And, while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported.1,2 Cultures are warranted only if a secondary infection is suspected.
The distribution of lesions and the presence—or absence—of systemic symptoms go a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy.3 Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.3
The blisters associated with BD spontaneously resolve over several weeks without treatment but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection. For this patient, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
This case was adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman’s toes. J Fam Pract. 2014;63:273-274.
1. James WD, Odom RB, Goette DK. Bullous eruption of diabetes. A case with positive immunofluorescence microscopy findings. Arch Dermatol. 1980;116:1191-1192.
2. Basarab T, Munn SE, McGrath J, et al. Bullous diabeticorum. A case report and literature review. Clin Exp Dermatol. 1995;20:218-220. doi: 10.1111/j.1365-2230.1995.tb01305.x
3. Hull C, Zone JJ. Approach to the patient with cutaneous blisters. UpToDate. Updated July 30, 2019. Accessed September 14, 2021. www.uptodate.com/contents/approach-to-the-patient-with-cutaneous-blisters
A biopsy was performed and sent for immunofluorescence; the results were negative. This, along with the patient’s history of diabetes, led us to the diagnosis of bullosis diabeticorum (BD). This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes.
The etiology of BD is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites.
A diagnosis of BD can be made when biopsy with immunofluorescence excludes other histologically similar diagnoses such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And, while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported.1,2 Cultures are warranted only if a secondary infection is suspected.
The distribution of lesions and the presence—or absence—of systemic symptoms go a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy.3 Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.3
The blisters associated with BD spontaneously resolve over several weeks without treatment but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection. For this patient, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
This case was adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman’s toes. J Fam Pract. 2014;63:273-274.
A biopsy was performed and sent for immunofluorescence; the results were negative. This, along with the patient’s history of diabetes, led us to the diagnosis of bullosis diabeticorum (BD). This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes.
The etiology of BD is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites.
A diagnosis of BD can be made when biopsy with immunofluorescence excludes other histologically similar diagnoses such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And, while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported.1,2 Cultures are warranted only if a secondary infection is suspected.
The distribution of lesions and the presence—or absence—of systemic symptoms go a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy.3 Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.3
The blisters associated with BD spontaneously resolve over several weeks without treatment but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection. For this patient, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
This case was adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman’s toes. J Fam Pract. 2014;63:273-274.
1. James WD, Odom RB, Goette DK. Bullous eruption of diabetes. A case with positive immunofluorescence microscopy findings. Arch Dermatol. 1980;116:1191-1192.
2. Basarab T, Munn SE, McGrath J, et al. Bullous diabeticorum. A case report and literature review. Clin Exp Dermatol. 1995;20:218-220. doi: 10.1111/j.1365-2230.1995.tb01305.x
3. Hull C, Zone JJ. Approach to the patient with cutaneous blisters. UpToDate. Updated July 30, 2019. Accessed September 14, 2021. www.uptodate.com/contents/approach-to-the-patient-with-cutaneous-blisters
1. James WD, Odom RB, Goette DK. Bullous eruption of diabetes. A case with positive immunofluorescence microscopy findings. Arch Dermatol. 1980;116:1191-1192.
2. Basarab T, Munn SE, McGrath J, et al. Bullous diabeticorum. A case report and literature review. Clin Exp Dermatol. 1995;20:218-220. doi: 10.1111/j.1365-2230.1995.tb01305.x
3. Hull C, Zone JJ. Approach to the patient with cutaneous blisters. UpToDate. Updated July 30, 2019. Accessed September 14, 2021. www.uptodate.com/contents/approach-to-the-patient-with-cutaneous-blisters
Association of height, BMI, and AD in young children may be transient
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM JAMA DERMATOLOGY
Two questions can help establish a diagnosis of hidradenitis suppurativa
According to Iltefat H. Hamzavi, MD,
If the answer to the first question is “yes” and the patient has had at least two boils in intertriginous areas, that person likely has HS, a disease of apocrine gland–bearing skin that occurs in 1%-4% of people, has a higher prevalence in Blacks, compared with Whites, and affects more women than men by a 3:1 ratio.
“Current treatments offer limited efficacy, and the disease is chronic and recurrent,” Dr. Hamzavi, of the department of dermatology at Henry Ford Health System, Detroit, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “You often see nodules, abscesses, fistulae, and scarring,” with all different skin types represented in the majority of patients.
Typical HS lesions appear as inflamed nodules, abscesses, draining fistulas, and scars as well as double-headed “tombstone” comedones, he said. These are typically located in the axilla, intermammary folds, in the groin, around the genitals, and on the buttocks. Atypical lesions can also occur – often folliculitis and open comedones in locations such as the waistline, the neck, and behind the ears.
The differential diagnosis is wide-ranging and includes bacterial abscess, inflamed cyst, folliculitis, pilonidal sinus, cellulitis, and cutaneous Crohn’s disease. Pain may appear out of proportion to the physical examination.
“There is a window of opportunity to treat HS, early in the disease process,” Dr. Hamzavi said. “There are no definitive cures for HS but lots of treatment options.”
According to clinical management guidelines published by the United States and Canadian Hidradenitis Suppurativa Foundations, options for moderate stage disease include antibiotics, antiandrogens, retinoids, immunosuppression/biologics, deroofing, and limited excision with primary closure. Options for severe disease include radical excision.
“HS requires a mix of medical and procedural treatments based on the number of nodules,” Dr. Hamzavi said. “Because the disease has so many different phases, there is no perfect outcome measure yet, but progress is being made.”
In 2018, an effort to develop a consensus core outcome set of domains regarding what to measure in clinical trials of HS was launched; it is known as the Hidradenitis Suppurativa Core Outcomes Set International Collaboration (HISTORIC). It was formed as a collaboration between the International Dermatology Outcome Measures (IDEOM) initiative, the Cochrane Skin Group – Core Outcome Set Initiative (CSG-COUSIN), and Zealand University Hospital, Roskilde.
HISTORIC is now part of the partnership with CSG-COUSIN and this work continues onward. Core domains as defined by the group include pain, physical signs, HS-specific quality of life, global assessment, and disease progression. “For now, we are mostly using some objective measures and some patient-reported outcomes with the addition of ultrasound in some centers,” Dr. Hamzavi said.
He underscored the importance of lifestyle modifications in patients with HS, including smoking cessation and weight loss, as well as decreasing pressure/friction on lesions, using warm compresses, and modifying diet. “This generally involves a low-inflammatory diet: Low carbohydrate, low dairy, and higher protein content, but there is much work needed to understand the role of diet in HS,” he said.
“This is a tough disease, but the compassion you offer these patients will be paid back to you a thousandfold. They tend to be some of the happiest and most appreciative patients you will ever have in your practice.”
Dr. Hamzavi disclosed that he has been a clinical investigator for Clinuvel, Incyte, Pfizer, Avita, and Ferndale Labs. He has also been a consultant for Pfizer, AbbVie, Novartis, and Aclaris, and has received a grant from Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
According to Iltefat H. Hamzavi, MD,
If the answer to the first question is “yes” and the patient has had at least two boils in intertriginous areas, that person likely has HS, a disease of apocrine gland–bearing skin that occurs in 1%-4% of people, has a higher prevalence in Blacks, compared with Whites, and affects more women than men by a 3:1 ratio.
“Current treatments offer limited efficacy, and the disease is chronic and recurrent,” Dr. Hamzavi, of the department of dermatology at Henry Ford Health System, Detroit, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “You often see nodules, abscesses, fistulae, and scarring,” with all different skin types represented in the majority of patients.
Typical HS lesions appear as inflamed nodules, abscesses, draining fistulas, and scars as well as double-headed “tombstone” comedones, he said. These are typically located in the axilla, intermammary folds, in the groin, around the genitals, and on the buttocks. Atypical lesions can also occur – often folliculitis and open comedones in locations such as the waistline, the neck, and behind the ears.
The differential diagnosis is wide-ranging and includes bacterial abscess, inflamed cyst, folliculitis, pilonidal sinus, cellulitis, and cutaneous Crohn’s disease. Pain may appear out of proportion to the physical examination.
“There is a window of opportunity to treat HS, early in the disease process,” Dr. Hamzavi said. “There are no definitive cures for HS but lots of treatment options.”
According to clinical management guidelines published by the United States and Canadian Hidradenitis Suppurativa Foundations, options for moderate stage disease include antibiotics, antiandrogens, retinoids, immunosuppression/biologics, deroofing, and limited excision with primary closure. Options for severe disease include radical excision.
“HS requires a mix of medical and procedural treatments based on the number of nodules,” Dr. Hamzavi said. “Because the disease has so many different phases, there is no perfect outcome measure yet, but progress is being made.”
In 2018, an effort to develop a consensus core outcome set of domains regarding what to measure in clinical trials of HS was launched; it is known as the Hidradenitis Suppurativa Core Outcomes Set International Collaboration (HISTORIC). It was formed as a collaboration between the International Dermatology Outcome Measures (IDEOM) initiative, the Cochrane Skin Group – Core Outcome Set Initiative (CSG-COUSIN), and Zealand University Hospital, Roskilde.
HISTORIC is now part of the partnership with CSG-COUSIN and this work continues onward. Core domains as defined by the group include pain, physical signs, HS-specific quality of life, global assessment, and disease progression. “For now, we are mostly using some objective measures and some patient-reported outcomes with the addition of ultrasound in some centers,” Dr. Hamzavi said.
He underscored the importance of lifestyle modifications in patients with HS, including smoking cessation and weight loss, as well as decreasing pressure/friction on lesions, using warm compresses, and modifying diet. “This generally involves a low-inflammatory diet: Low carbohydrate, low dairy, and higher protein content, but there is much work needed to understand the role of diet in HS,” he said.
“This is a tough disease, but the compassion you offer these patients will be paid back to you a thousandfold. They tend to be some of the happiest and most appreciative patients you will ever have in your practice.”
Dr. Hamzavi disclosed that he has been a clinical investigator for Clinuvel, Incyte, Pfizer, Avita, and Ferndale Labs. He has also been a consultant for Pfizer, AbbVie, Novartis, and Aclaris, and has received a grant from Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
According to Iltefat H. Hamzavi, MD,
If the answer to the first question is “yes” and the patient has had at least two boils in intertriginous areas, that person likely has HS, a disease of apocrine gland–bearing skin that occurs in 1%-4% of people, has a higher prevalence in Blacks, compared with Whites, and affects more women than men by a 3:1 ratio.
“Current treatments offer limited efficacy, and the disease is chronic and recurrent,” Dr. Hamzavi, of the department of dermatology at Henry Ford Health System, Detroit, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “You often see nodules, abscesses, fistulae, and scarring,” with all different skin types represented in the majority of patients.
Typical HS lesions appear as inflamed nodules, abscesses, draining fistulas, and scars as well as double-headed “tombstone” comedones, he said. These are typically located in the axilla, intermammary folds, in the groin, around the genitals, and on the buttocks. Atypical lesions can also occur – often folliculitis and open comedones in locations such as the waistline, the neck, and behind the ears.
The differential diagnosis is wide-ranging and includes bacterial abscess, inflamed cyst, folliculitis, pilonidal sinus, cellulitis, and cutaneous Crohn’s disease. Pain may appear out of proportion to the physical examination.
“There is a window of opportunity to treat HS, early in the disease process,” Dr. Hamzavi said. “There are no definitive cures for HS but lots of treatment options.”
According to clinical management guidelines published by the United States and Canadian Hidradenitis Suppurativa Foundations, options for moderate stage disease include antibiotics, antiandrogens, retinoids, immunosuppression/biologics, deroofing, and limited excision with primary closure. Options for severe disease include radical excision.
“HS requires a mix of medical and procedural treatments based on the number of nodules,” Dr. Hamzavi said. “Because the disease has so many different phases, there is no perfect outcome measure yet, but progress is being made.”
In 2018, an effort to develop a consensus core outcome set of domains regarding what to measure in clinical trials of HS was launched; it is known as the Hidradenitis Suppurativa Core Outcomes Set International Collaboration (HISTORIC). It was formed as a collaboration between the International Dermatology Outcome Measures (IDEOM) initiative, the Cochrane Skin Group – Core Outcome Set Initiative (CSG-COUSIN), and Zealand University Hospital, Roskilde.
HISTORIC is now part of the partnership with CSG-COUSIN and this work continues onward. Core domains as defined by the group include pain, physical signs, HS-specific quality of life, global assessment, and disease progression. “For now, we are mostly using some objective measures and some patient-reported outcomes with the addition of ultrasound in some centers,” Dr. Hamzavi said.
He underscored the importance of lifestyle modifications in patients with HS, including smoking cessation and weight loss, as well as decreasing pressure/friction on lesions, using warm compresses, and modifying diet. “This generally involves a low-inflammatory diet: Low carbohydrate, low dairy, and higher protein content, but there is much work needed to understand the role of diet in HS,” he said.
“This is a tough disease, but the compassion you offer these patients will be paid back to you a thousandfold. They tend to be some of the happiest and most appreciative patients you will ever have in your practice.”
Dr. Hamzavi disclosed that he has been a clinical investigator for Clinuvel, Incyte, Pfizer, Avita, and Ferndale Labs. He has also been a consultant for Pfizer, AbbVie, Novartis, and Aclaris, and has received a grant from Estee Lauder.
MedscapeLive and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Study finds nadolol noninferior to propranolol for infantile hemangiomas
according to a study published in JAMA Pediatrics.
“In our experience, nadolol is preferable to propranolol given its observed efficacy and similar safety profile [and] its more predictable metabolism that does not involve the liver,” lead author Elena Pope, MD, told this news organization. “In addition, the fact that nadolol is less lipophilic than propranolol makes it less likely to cross the blood-brain barrier and potentially affect the central nervous system,” added Dr. Pope, who is head of the division of pediatric dermatology at the Hospital for Sick Children, Toronto, and professor of pediatric medicine at the University of Toronto.
The prospective double-blind, randomized noninferiority study was conducted between 2016 and 2020 at two tertiary academic pediatric dermatology clinics in Ontario, Canada. It included 71 infants with a corrected gestational age of 1-6 months whose hemangiomas were greater than 1.5 cm on the face or 3 cm or greater on another body part and had the potential to cause functional impairment or cosmetic disfigurement.
Patients were randomized to either nadolol (oral suspension, 10 mg/mL) or propranolol (oral suspension, 5 mg/mL) beginning at a dose of 0.5 mg/kg per day twice a day and titrated weekly by 0.5 mg/kg per day until the maximum dose of 2 mg/kg per day. The dose was then adjusted until week 24, based on patient weight and clinical response, after which parents could choose to continue the infant on the assigned medication or switch to the other one. Follow-up visits occurred every 2 months after that until week 52.
For the main study outcome, measured by visual analog scale (VAS) scores at week 24, the between-group differences of IH size and color from baseline were 8.8 and 17.1, respectively, in favor of the nadolol group, the researchers report, with similar results seen at week 52. Safety data were similar for both treatments, “demonstrating that nadolol was noninferior to propranolol,” they write.
Additionally, the mean size involution, compared with baseline was 97.9% in the nadolol group and 89.1% in the propranolol group, and the mean color fading was 94.5% in the nadolol group, compared with 80.5% in the propranolol group. During the study, nadolol was also “59% faster in achieving 75% shrinkage of IH, compared with propranolol (P = .02) and 105% faster in achieving 100% shrinkage (P = .07),” they add.
“A considerable portion of patients experienced at least one mild adverse event (77.1% vs. 94.4% at 0-24 weeks and 84.2% vs. 74.2% at 24-52 weeks in the nadolol group vs. the propranolol group, respectively), with a median of two in each intervention group,” they noted, adding that while these numbers are high, they are similar to those in previous clinical trials.
“The efficacy data coupled with a more predictable pharmacokinetic profile and lower chance of crossing the blood-brain barrier may make nadolol a favorable alternative intervention in patients with IHs,” the authors conclude. However, they add that “further studies are needed to prove superiority over propranolol.”
Asked to comment on the results, Ilona J. Frieden, MD, director of the Birthmarks & Vascular Anomalies Center at the University of California, San Francisco, said that while this is a “very interesting study and deserves further consideration,” the findings do not reach the level at which they would change guidelines. “The vast majority of patients being treated with a systemic medication for IH are in fact getting propranolol,” said Dr. Frieden, coauthor of the American Academy of Pediatrics Clinical Practice Guideline for the Management of Infantile Hemangiomas.
“Though this study – designed as a noninferiority study – does seem to show slightly better outcomes from nadolol versus propranolol … it is a relatively small study,” she told this news organization. “Infantile hemangiomas are a very heterogeneous group, and larger studies and longer-term outcome data would be needed to truly compare the two modalities of treatment.”
Concern over the safety of nadolol was raised in a case report published in Pediatrics, which described the death of a 10-week-old girl 7 weeks after starting nadolol for IH. The infant was found to have an elevated postmortem cardiac blood nadolol level of 0.94 mg/L. “Although we debated the conclusion of that report in terms of death attribution to nadolol, one practical pearl is to instruct the parents to discontinue nadolol if the baby has no bowel movements for more than 3 days,” Dr. Pope advised.
The author of that case report, Eric McGillis, MD, program director of clinical pharmacology and toxicology and an emergency physician at Alberta Health Services, in Calgary, Alt., said the conclusion of his report has been taken out of context. “We acknowledge that our case report, like any case report, cannot prove causation,” he told this news organization. “We hypothesized that nadolol may have contributed to the death of the infant based on the limited pharmacokinetic data currently available for nadolol in infants. Nadolol is largely eliminated in the feces and infants may have infrequent stooling based on diet and other factors; therefore, nadolol may accumulate,” he noted.
The infant in the case report did not have a bowel movement for 10 days “and had an elevated postmortem cardiac nadolol concentration in the absence of another obvious cause of death. More pharmacokinetic studies on nadolol in this population are needed to substantiate our hypothesis. However, in the meantime, we agree that having parents monitor stool output for dose adjustments makes practical sense and can potentially reduce harm.”
Dr. Pope presented the results of the study earlier this year at the annual meeting of the Society for Pediatric Dermatology.
The study was supported by Physician Services, Ont. Dr. Pope has reported serving as an advisory board member for Boehringer Ingelheim, Novartis, Sanofi Genzyme, and Timber. Other authors have reported receiving personal fees from Pierre Fabre during the conduct of the study, as well as personal fees from Amgen, Ipsen, Novartis, Pfizer, and Sanofi Genzyme; grants from AbbVie, Clementia, Mayne Pharma, and Sanofi Genzyme; and grants and personal fees from Venthera. One author has a patent for a new topical treatment of IH. Dr. Frieden has reported being a consultant for Pfizer (data safety board), Novartis, and Venthera. Dr. McGillis has reported no relevant financial relationships.
Commentary by Lawrence W. Eichenfield, MD
The treatment of functionally significant and deforming hemangiomas has been revolutionized by propranolol, developed after the observation by Christine Léauté-Labrèze, MD, that a child who developed hypertension as a side effect of systemic steroids for a nasal hemangioma and was prescribed propranolol for the hypertension had rapid shrinkage of the hemangioma. The study by Pope and colleagues assesses nadolol as an alternative to propranolol, showing noninferiority and in some parameters improved outcomes and speed of response. The drug appeared to be fairly well tolerated in the study, though there is a prior published case report of a death from nadolol use for hemangioma treatment from a different Canadian center. Nadolol may be an important alternative to propranolol; however, propranolol remains the only FDA-approved medication for infantile hemangiomas and the generally recommended medication in the American Academy of Pediatrics guidelines for management of infantile hemangiomas.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
according to a study published in JAMA Pediatrics.
“In our experience, nadolol is preferable to propranolol given its observed efficacy and similar safety profile [and] its more predictable metabolism that does not involve the liver,” lead author Elena Pope, MD, told this news organization. “In addition, the fact that nadolol is less lipophilic than propranolol makes it less likely to cross the blood-brain barrier and potentially affect the central nervous system,” added Dr. Pope, who is head of the division of pediatric dermatology at the Hospital for Sick Children, Toronto, and professor of pediatric medicine at the University of Toronto.
The prospective double-blind, randomized noninferiority study was conducted between 2016 and 2020 at two tertiary academic pediatric dermatology clinics in Ontario, Canada. It included 71 infants with a corrected gestational age of 1-6 months whose hemangiomas were greater than 1.5 cm on the face or 3 cm or greater on another body part and had the potential to cause functional impairment or cosmetic disfigurement.
Patients were randomized to either nadolol (oral suspension, 10 mg/mL) or propranolol (oral suspension, 5 mg/mL) beginning at a dose of 0.5 mg/kg per day twice a day and titrated weekly by 0.5 mg/kg per day until the maximum dose of 2 mg/kg per day. The dose was then adjusted until week 24, based on patient weight and clinical response, after which parents could choose to continue the infant on the assigned medication or switch to the other one. Follow-up visits occurred every 2 months after that until week 52.
For the main study outcome, measured by visual analog scale (VAS) scores at week 24, the between-group differences of IH size and color from baseline were 8.8 and 17.1, respectively, in favor of the nadolol group, the researchers report, with similar results seen at week 52. Safety data were similar for both treatments, “demonstrating that nadolol was noninferior to propranolol,” they write.
Additionally, the mean size involution, compared with baseline was 97.9% in the nadolol group and 89.1% in the propranolol group, and the mean color fading was 94.5% in the nadolol group, compared with 80.5% in the propranolol group. During the study, nadolol was also “59% faster in achieving 75% shrinkage of IH, compared with propranolol (P = .02) and 105% faster in achieving 100% shrinkage (P = .07),” they add.
“A considerable portion of patients experienced at least one mild adverse event (77.1% vs. 94.4% at 0-24 weeks and 84.2% vs. 74.2% at 24-52 weeks in the nadolol group vs. the propranolol group, respectively), with a median of two in each intervention group,” they noted, adding that while these numbers are high, they are similar to those in previous clinical trials.
“The efficacy data coupled with a more predictable pharmacokinetic profile and lower chance of crossing the blood-brain barrier may make nadolol a favorable alternative intervention in patients with IHs,” the authors conclude. However, they add that “further studies are needed to prove superiority over propranolol.”
Asked to comment on the results, Ilona J. Frieden, MD, director of the Birthmarks & Vascular Anomalies Center at the University of California, San Francisco, said that while this is a “very interesting study and deserves further consideration,” the findings do not reach the level at which they would change guidelines. “The vast majority of patients being treated with a systemic medication for IH are in fact getting propranolol,” said Dr. Frieden, coauthor of the American Academy of Pediatrics Clinical Practice Guideline for the Management of Infantile Hemangiomas.
“Though this study – designed as a noninferiority study – does seem to show slightly better outcomes from nadolol versus propranolol … it is a relatively small study,” she told this news organization. “Infantile hemangiomas are a very heterogeneous group, and larger studies and longer-term outcome data would be needed to truly compare the two modalities of treatment.”
Concern over the safety of nadolol was raised in a case report published in Pediatrics, which described the death of a 10-week-old girl 7 weeks after starting nadolol for IH. The infant was found to have an elevated postmortem cardiac blood nadolol level of 0.94 mg/L. “Although we debated the conclusion of that report in terms of death attribution to nadolol, one practical pearl is to instruct the parents to discontinue nadolol if the baby has no bowel movements for more than 3 days,” Dr. Pope advised.
The author of that case report, Eric McGillis, MD, program director of clinical pharmacology and toxicology and an emergency physician at Alberta Health Services, in Calgary, Alt., said the conclusion of his report has been taken out of context. “We acknowledge that our case report, like any case report, cannot prove causation,” he told this news organization. “We hypothesized that nadolol may have contributed to the death of the infant based on the limited pharmacokinetic data currently available for nadolol in infants. Nadolol is largely eliminated in the feces and infants may have infrequent stooling based on diet and other factors; therefore, nadolol may accumulate,” he noted.
The infant in the case report did not have a bowel movement for 10 days “and had an elevated postmortem cardiac nadolol concentration in the absence of another obvious cause of death. More pharmacokinetic studies on nadolol in this population are needed to substantiate our hypothesis. However, in the meantime, we agree that having parents monitor stool output for dose adjustments makes practical sense and can potentially reduce harm.”
Dr. Pope presented the results of the study earlier this year at the annual meeting of the Society for Pediatric Dermatology.
The study was supported by Physician Services, Ont. Dr. Pope has reported serving as an advisory board member for Boehringer Ingelheim, Novartis, Sanofi Genzyme, and Timber. Other authors have reported receiving personal fees from Pierre Fabre during the conduct of the study, as well as personal fees from Amgen, Ipsen, Novartis, Pfizer, and Sanofi Genzyme; grants from AbbVie, Clementia, Mayne Pharma, and Sanofi Genzyme; and grants and personal fees from Venthera. One author has a patent for a new topical treatment of IH. Dr. Frieden has reported being a consultant for Pfizer (data safety board), Novartis, and Venthera. Dr. McGillis has reported no relevant financial relationships.
Commentary by Lawrence W. Eichenfield, MD
The treatment of functionally significant and deforming hemangiomas has been revolutionized by propranolol, developed after the observation by Christine Léauté-Labrèze, MD, that a child who developed hypertension as a side effect of systemic steroids for a nasal hemangioma and was prescribed propranolol for the hypertension had rapid shrinkage of the hemangioma. The study by Pope and colleagues assesses nadolol as an alternative to propranolol, showing noninferiority and in some parameters improved outcomes and speed of response. The drug appeared to be fairly well tolerated in the study, though there is a prior published case report of a death from nadolol use for hemangioma treatment from a different Canadian center. Nadolol may be an important alternative to propranolol; however, propranolol remains the only FDA-approved medication for infantile hemangiomas and the generally recommended medication in the American Academy of Pediatrics guidelines for management of infantile hemangiomas.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
according to a study published in JAMA Pediatrics.
“In our experience, nadolol is preferable to propranolol given its observed efficacy and similar safety profile [and] its more predictable metabolism that does not involve the liver,” lead author Elena Pope, MD, told this news organization. “In addition, the fact that nadolol is less lipophilic than propranolol makes it less likely to cross the blood-brain barrier and potentially affect the central nervous system,” added Dr. Pope, who is head of the division of pediatric dermatology at the Hospital for Sick Children, Toronto, and professor of pediatric medicine at the University of Toronto.
The prospective double-blind, randomized noninferiority study was conducted between 2016 and 2020 at two tertiary academic pediatric dermatology clinics in Ontario, Canada. It included 71 infants with a corrected gestational age of 1-6 months whose hemangiomas were greater than 1.5 cm on the face or 3 cm or greater on another body part and had the potential to cause functional impairment or cosmetic disfigurement.
Patients were randomized to either nadolol (oral suspension, 10 mg/mL) or propranolol (oral suspension, 5 mg/mL) beginning at a dose of 0.5 mg/kg per day twice a day and titrated weekly by 0.5 mg/kg per day until the maximum dose of 2 mg/kg per day. The dose was then adjusted until week 24, based on patient weight and clinical response, after which parents could choose to continue the infant on the assigned medication or switch to the other one. Follow-up visits occurred every 2 months after that until week 52.
For the main study outcome, measured by visual analog scale (VAS) scores at week 24, the between-group differences of IH size and color from baseline were 8.8 and 17.1, respectively, in favor of the nadolol group, the researchers report, with similar results seen at week 52. Safety data were similar for both treatments, “demonstrating that nadolol was noninferior to propranolol,” they write.
Additionally, the mean size involution, compared with baseline was 97.9% in the nadolol group and 89.1% in the propranolol group, and the mean color fading was 94.5% in the nadolol group, compared with 80.5% in the propranolol group. During the study, nadolol was also “59% faster in achieving 75% shrinkage of IH, compared with propranolol (P = .02) and 105% faster in achieving 100% shrinkage (P = .07),” they add.
“A considerable portion of patients experienced at least one mild adverse event (77.1% vs. 94.4% at 0-24 weeks and 84.2% vs. 74.2% at 24-52 weeks in the nadolol group vs. the propranolol group, respectively), with a median of two in each intervention group,” they noted, adding that while these numbers are high, they are similar to those in previous clinical trials.
“The efficacy data coupled with a more predictable pharmacokinetic profile and lower chance of crossing the blood-brain barrier may make nadolol a favorable alternative intervention in patients with IHs,” the authors conclude. However, they add that “further studies are needed to prove superiority over propranolol.”
Asked to comment on the results, Ilona J. Frieden, MD, director of the Birthmarks & Vascular Anomalies Center at the University of California, San Francisco, said that while this is a “very interesting study and deserves further consideration,” the findings do not reach the level at which they would change guidelines. “The vast majority of patients being treated with a systemic medication for IH are in fact getting propranolol,” said Dr. Frieden, coauthor of the American Academy of Pediatrics Clinical Practice Guideline for the Management of Infantile Hemangiomas.
“Though this study – designed as a noninferiority study – does seem to show slightly better outcomes from nadolol versus propranolol … it is a relatively small study,” she told this news organization. “Infantile hemangiomas are a very heterogeneous group, and larger studies and longer-term outcome data would be needed to truly compare the two modalities of treatment.”
Concern over the safety of nadolol was raised in a case report published in Pediatrics, which described the death of a 10-week-old girl 7 weeks after starting nadolol for IH. The infant was found to have an elevated postmortem cardiac blood nadolol level of 0.94 mg/L. “Although we debated the conclusion of that report in terms of death attribution to nadolol, one practical pearl is to instruct the parents to discontinue nadolol if the baby has no bowel movements for more than 3 days,” Dr. Pope advised.
The author of that case report, Eric McGillis, MD, program director of clinical pharmacology and toxicology and an emergency physician at Alberta Health Services, in Calgary, Alt., said the conclusion of his report has been taken out of context. “We acknowledge that our case report, like any case report, cannot prove causation,” he told this news organization. “We hypothesized that nadolol may have contributed to the death of the infant based on the limited pharmacokinetic data currently available for nadolol in infants. Nadolol is largely eliminated in the feces and infants may have infrequent stooling based on diet and other factors; therefore, nadolol may accumulate,” he noted.
The infant in the case report did not have a bowel movement for 10 days “and had an elevated postmortem cardiac nadolol concentration in the absence of another obvious cause of death. More pharmacokinetic studies on nadolol in this population are needed to substantiate our hypothesis. However, in the meantime, we agree that having parents monitor stool output for dose adjustments makes practical sense and can potentially reduce harm.”
Dr. Pope presented the results of the study earlier this year at the annual meeting of the Society for Pediatric Dermatology.
The study was supported by Physician Services, Ont. Dr. Pope has reported serving as an advisory board member for Boehringer Ingelheim, Novartis, Sanofi Genzyme, and Timber. Other authors have reported receiving personal fees from Pierre Fabre during the conduct of the study, as well as personal fees from Amgen, Ipsen, Novartis, Pfizer, and Sanofi Genzyme; grants from AbbVie, Clementia, Mayne Pharma, and Sanofi Genzyme; and grants and personal fees from Venthera. One author has a patent for a new topical treatment of IH. Dr. Frieden has reported being a consultant for Pfizer (data safety board), Novartis, and Venthera. Dr. McGillis has reported no relevant financial relationships.
Commentary by Lawrence W. Eichenfield, MD
The treatment of functionally significant and deforming hemangiomas has been revolutionized by propranolol, developed after the observation by Christine Léauté-Labrèze, MD, that a child who developed hypertension as a side effect of systemic steroids for a nasal hemangioma and was prescribed propranolol for the hypertension had rapid shrinkage of the hemangioma. The study by Pope and colleagues assesses nadolol as an alternative to propranolol, showing noninferiority and in some parameters improved outcomes and speed of response. The drug appeared to be fairly well tolerated in the study, though there is a prior published case report of a death from nadolol use for hemangioma treatment from a different Canadian center. Nadolol may be an important alternative to propranolol; however, propranolol remains the only FDA-approved medication for infantile hemangiomas and the generally recommended medication in the American Academy of Pediatrics guidelines for management of infantile hemangiomas.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM JAMA PEDIATRICS
Certain opioids hold promise for treating itch
Certain opioids are proving to be effective in treating a variety of itch conditions, according to Brian S. Kim, MD.
“We know that opioids or opiates do cause itch in a significant number of patients,” Dr. Kim, a dermatologist who is codirector of the Center for the Study of Itch & Sensory Disorders at Washington University, St. Louis, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It’s thought to do this by way of acting as a pruritogen at times and stimulating sensory neurons [that] then activate the itch cascade. But it’s also been well known that endogenous kappa opioids can activate sensory neurons that can then suppress itch and gate out signals from these opiates, but perhaps other pruritogens as well.”
Multiple drugs differentially target kappa-opioid receptor (KOR) and mu-opioid receptor (MOR) pathways, he continued. For example, oral naltrexone is a MOR antagonist, oral nalfurafine and intravenous difelikefalin are KOR agonists, while intranasal butorphanol and oral nalbuphine have a dual mechanism.
Difelikefalin is the first Food and Drug Administration–approved treatment for uremic pruritus associated with dialysis, approved in August 2021 for moderate-to-severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis; it is administered intravenously. During the 2021 annual congress of the European Academy of Dermatology and Venereology, Dr. Kim and colleagues presented findings from a phase 2 trial of 401 people with atopic dermatitis (AD) and moderate to severe pruritus, who were randomized to receive oral difelikefalin at a dose of 0.25 mg, 0.5 mg, or 1.0 mg, or placebo over a 12-week treatment period. The primary endpoint, change from baseline in Itch Numerical Rating Scale score, was not met in any of the difelikefalin dose groups in the overall study population, but patients with a body surface area of less than 10% experienced a significant improvement in itch at week 12 in the combined difelikefalin dose group in (P = .039). A significant reduction in itch with difelikefalin was seen in this group of patients with itch-dominant AD, as early as the second day of treatment.
In another trial, 373 hemodialysis patients with moderate or severe uremic pruritus were randomized in a 1: 1:1 ratio to nalbuphine extended-release tablets 120 mg, 60 mg, or placebo and treated for 8 weeks. The researchers found that nalbuphine 120 mg significantly reduced the itching intensity. Specifically, from a baseline numerical rate scale (NRS) of 6.9, the mean NRS declined by 3.5 and by 2.8 in the nalbuphine 120-mg and the placebo groups, respectively (P = .017).
In a separate, unpublished multicenter, randomized, phase 2/3 trial, researchers evaluated the safety and antipruritic efficacy of nalbuphine extended-release tablets dosed twice daily at 90 mg and 180 mg in 62 patients in the United States and Europe. The proportion of patients in the nalbuphine 180-mg arm who met 50% responder criteria at week 10 or last observed visit approached statistical significance (P = .083), and this arm met statistical significance for patients who completed treatment (P = .028).
Dr. Kim disclosed that he has served as a consultant for AbbVie, AstraZeneca, Cara Therapeutics, Galderma, GlaxoSmithKline, LEO Pharma, Lilly, Pfizer, Regeneron, Sanofi, Trevi Therapeutics. He also has conducted contracted research for Cara Therapeutics and LEO Pharma.
MedscapeLive and this news organization are owned by the same parent company.
Certain opioids are proving to be effective in treating a variety of itch conditions, according to Brian S. Kim, MD.
“We know that opioids or opiates do cause itch in a significant number of patients,” Dr. Kim, a dermatologist who is codirector of the Center for the Study of Itch & Sensory Disorders at Washington University, St. Louis, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It’s thought to do this by way of acting as a pruritogen at times and stimulating sensory neurons [that] then activate the itch cascade. But it’s also been well known that endogenous kappa opioids can activate sensory neurons that can then suppress itch and gate out signals from these opiates, but perhaps other pruritogens as well.”
Multiple drugs differentially target kappa-opioid receptor (KOR) and mu-opioid receptor (MOR) pathways, he continued. For example, oral naltrexone is a MOR antagonist, oral nalfurafine and intravenous difelikefalin are KOR agonists, while intranasal butorphanol and oral nalbuphine have a dual mechanism.
Difelikefalin is the first Food and Drug Administration–approved treatment for uremic pruritus associated with dialysis, approved in August 2021 for moderate-to-severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis; it is administered intravenously. During the 2021 annual congress of the European Academy of Dermatology and Venereology, Dr. Kim and colleagues presented findings from a phase 2 trial of 401 people with atopic dermatitis (AD) and moderate to severe pruritus, who were randomized to receive oral difelikefalin at a dose of 0.25 mg, 0.5 mg, or 1.0 mg, or placebo over a 12-week treatment period. The primary endpoint, change from baseline in Itch Numerical Rating Scale score, was not met in any of the difelikefalin dose groups in the overall study population, but patients with a body surface area of less than 10% experienced a significant improvement in itch at week 12 in the combined difelikefalin dose group in (P = .039). A significant reduction in itch with difelikefalin was seen in this group of patients with itch-dominant AD, as early as the second day of treatment.
In another trial, 373 hemodialysis patients with moderate or severe uremic pruritus were randomized in a 1: 1:1 ratio to nalbuphine extended-release tablets 120 mg, 60 mg, or placebo and treated for 8 weeks. The researchers found that nalbuphine 120 mg significantly reduced the itching intensity. Specifically, from a baseline numerical rate scale (NRS) of 6.9, the mean NRS declined by 3.5 and by 2.8 in the nalbuphine 120-mg and the placebo groups, respectively (P = .017).
In a separate, unpublished multicenter, randomized, phase 2/3 trial, researchers evaluated the safety and antipruritic efficacy of nalbuphine extended-release tablets dosed twice daily at 90 mg and 180 mg in 62 patients in the United States and Europe. The proportion of patients in the nalbuphine 180-mg arm who met 50% responder criteria at week 10 or last observed visit approached statistical significance (P = .083), and this arm met statistical significance for patients who completed treatment (P = .028).
Dr. Kim disclosed that he has served as a consultant for AbbVie, AstraZeneca, Cara Therapeutics, Galderma, GlaxoSmithKline, LEO Pharma, Lilly, Pfizer, Regeneron, Sanofi, Trevi Therapeutics. He also has conducted contracted research for Cara Therapeutics and LEO Pharma.
MedscapeLive and this news organization are owned by the same parent company.
Certain opioids are proving to be effective in treating a variety of itch conditions, according to Brian S. Kim, MD.
“We know that opioids or opiates do cause itch in a significant number of patients,” Dr. Kim, a dermatologist who is codirector of the Center for the Study of Itch & Sensory Disorders at Washington University, St. Louis, said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It’s thought to do this by way of acting as a pruritogen at times and stimulating sensory neurons [that] then activate the itch cascade. But it’s also been well known that endogenous kappa opioids can activate sensory neurons that can then suppress itch and gate out signals from these opiates, but perhaps other pruritogens as well.”
Multiple drugs differentially target kappa-opioid receptor (KOR) and mu-opioid receptor (MOR) pathways, he continued. For example, oral naltrexone is a MOR antagonist, oral nalfurafine and intravenous difelikefalin are KOR agonists, while intranasal butorphanol and oral nalbuphine have a dual mechanism.
Difelikefalin is the first Food and Drug Administration–approved treatment for uremic pruritus associated with dialysis, approved in August 2021 for moderate-to-severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis; it is administered intravenously. During the 2021 annual congress of the European Academy of Dermatology and Venereology, Dr. Kim and colleagues presented findings from a phase 2 trial of 401 people with atopic dermatitis (AD) and moderate to severe pruritus, who were randomized to receive oral difelikefalin at a dose of 0.25 mg, 0.5 mg, or 1.0 mg, or placebo over a 12-week treatment period. The primary endpoint, change from baseline in Itch Numerical Rating Scale score, was not met in any of the difelikefalin dose groups in the overall study population, but patients with a body surface area of less than 10% experienced a significant improvement in itch at week 12 in the combined difelikefalin dose group in (P = .039). A significant reduction in itch with difelikefalin was seen in this group of patients with itch-dominant AD, as early as the second day of treatment.
In another trial, 373 hemodialysis patients with moderate or severe uremic pruritus were randomized in a 1: 1:1 ratio to nalbuphine extended-release tablets 120 mg, 60 mg, or placebo and treated for 8 weeks. The researchers found that nalbuphine 120 mg significantly reduced the itching intensity. Specifically, from a baseline numerical rate scale (NRS) of 6.9, the mean NRS declined by 3.5 and by 2.8 in the nalbuphine 120-mg and the placebo groups, respectively (P = .017).
In a separate, unpublished multicenter, randomized, phase 2/3 trial, researchers evaluated the safety and antipruritic efficacy of nalbuphine extended-release tablets dosed twice daily at 90 mg and 180 mg in 62 patients in the United States and Europe. The proportion of patients in the nalbuphine 180-mg arm who met 50% responder criteria at week 10 or last observed visit approached statistical significance (P = .083), and this arm met statistical significance for patients who completed treatment (P = .028).
Dr. Kim disclosed that he has served as a consultant for AbbVie, AstraZeneca, Cara Therapeutics, Galderma, GlaxoSmithKline, LEO Pharma, Lilly, Pfizer, Regeneron, Sanofi, Trevi Therapeutics. He also has conducted contracted research for Cara Therapeutics and LEO Pharma.
MedscapeLive and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Validity of commercial serologic tests for dermatomyositis still questionable
, according to Jeffrey P. Callen, MD.
That’s because the validity and reproducibility of testing in commercial laboratories remain questionable, Dr. Callen, professor of medicine and chief of the division of dermatology at the University of Louisville, Ky., said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “The testing in research laboratories is not widely available and the results are often delayed by weeks to months,” he said.
In addition, while the associations between antibody results and risks of malignancy or pulmonary disease are “statistically valid,” he said, “there are patients with disease in whom antibodies are not present and those without associated disease in whom the testing was positive.” For example, there are patients positive for anti–transition initiation factor (TIF)-1gamma but don’t have a malignancy, “and the ones with anti-MDA-5 tend to have pulmonary disease, but there are patients with anti-MDA-5 who don’t have pulmonary disease.”
Compared with patients with systemic lupus erythematosus, patients with dermatomyositis tend to have more itching and they tend of have fewer serologic abnormalities, such as anti-Ro/SS-A antibody, “but there is overlap,” Dr. Callen said. “The reason to differentiate cutaneous lupus erythematosus from dermatomyositis is because we think that patients who have amyopathic dermatomyositis still have an increased risk of having or developing an internal malignancy,” he added. Another differentiating point that is substantive is the presence of Gottron papules.
In a recent development related to antibody testing, researchers demonstrated that the IgG2 isotype of anti-TIF-1gamma antibodies is a biomarker of cancer and mortality in adult dermatomyositis.
According to population-based studies, about 20%-25% of dermatomyositis patients have had, have, or will develop a cancer (Lancet 2001;357: 96-100). Amyopathic dermatomyositis patients may also have cancer. Polymyositis patients generally have lower rates and their risk of subsequent malignancy is much closer to that of the general population, suggesting that the presence of the association is due to a “diagnostic suspicion bias,” Dr. Callen said.
A large-scale multicenter cohort study that set out to identify the risk factors and prognosis of patients with cancer-associated myositis found that ovarian cancer seems to be overrepresented. The only serologic abnormality that was statistically significant was anti-TIF-1gamma antibody (P less than .001). Patients with cancer-associated myositis also have less overall survival compared with those with non–cancer-associated myositis (P = .004), with malignancy being the primary cause of death (P less than .001).
In what is believed to be the largest study of its kind, Dr. Callen and colleagues retrospectively examined the prevalence of malignancy and screening practices in 400 dermatomyositis patients. Of the 400 patients, 48 (12%) had malignancies, and 21 cancers (40%) were diagnosed within 1 year of the dermatomyositis diagnosis. Both classic dermatomyositis and amyopathic dermatomyositis were associated with cancer, and 27 patients (6.8%) had a cancer at the time of diagnosis. Of those, 59% were asymptomatic; their cancers were discovered with CT scans, suggesting that “blind” screening is effective in identifying cancers in DM patients.
Dr. Callen’s malignancy evaluation includes chest x-ray, CT of the chest and abdomen, stool Hematest in all dermatomyositis patients; a mammogram, pelvic ultrasound and/or CT of the pelvis in women; and age, race or ethnicity-related testing. “I generally reevaluate patients annually for 3 years, because data from epidemiologic studies suggest that after 3 years [from the initial diagnosis], the rates of malignancy return toward normal,” he said. “I also evaluate any new symptom that might be suggestive of malignancy. The remaining issue is how to handle a patient in remission for several years, but who develops a relapse. What I do is perform another malignancy assessment.”
According to results from a meta-analysis of risk factors and systematic review of screening approaches, factors that increase malignancy risk include dermatomyositis subtype (risk ratio, 2.21), older age (weighted mean difference 11.19), male gender (RR, 1.53), dysphagia (RR, 2.09), cutaneous necrosis (RR, 2.73), and positive anti-TIF-1gamma (RR, 4.41).
Factors associated with a decreased risk of malignancy include polymyositis (RR, 0.49), clinically amyopathic dermatomyositis subtypes (RR, 0.44), Raynaud’s phenomenon (RR, 0.61), interstitial lung disease (RR, 0.49), very high serum creatine kinase (WMD –1189.96) or lactate dehydrogenase levels (WMD –336.53), and anti-Jo1 (RR, 0.45) or anti-EJ (RR, 0.17) positivity.
The analysis also found that CT scanning of the thorax, abdomen and pelvis appeared to yield a high proportion of underlying asymptomatic cancers. Limited evidence relating to the utility of tumor markers and 18F-FDG PET/CT was available.
As for treatment, the use of tofacitinib for cutaneous lesions of dermatomyositis has been suggested in various studies. In a recent open-label study of 10 patients with dermatomyositis who took extended release the JAK inhibitor tofacitinib 11 mg daily for 12 weeks, half experienced moderate improvement in disease activity, and the other half experienced minimal improvement. JAK inhibitors have been used in patients with juvenile dermatomyositis.
Dr. Callen’s treatment approach with dermatomyositis patients includes recommendations for sunscreens and protective clothing, plus assessment of vitamin D levels. “I will use topical emollients, corticosteroids, and calcineurin inhibitors,” he said. “Antimalarials might be used. I generally reach for methotrexate or mycophenolate mofetil relatively early. IVIG has also been studied.” Off-label therapies that have been used include dapsone, thalidomide, leflunomide, sirolimus, chlorambucil, etanercept, infliximab, rituximab, apremilast, tofacitinib, lenabasum, and low-dose naltrexone.
Dr. Callen disclosed that he is a consultant to Genentech and is a member of the safety monitoring committee for Principia Biopharma. He holds equity in Celgene, Pfizer, 3M, Johnson & Johnson, Merck, Abbott Laboratories, AbbVie, Procter & Gamble, Gilead, Allergen, and Amgen.
MedscapeLive and this news organization are owned by the same parent company.
, according to Jeffrey P. Callen, MD.
That’s because the validity and reproducibility of testing in commercial laboratories remain questionable, Dr. Callen, professor of medicine and chief of the division of dermatology at the University of Louisville, Ky., said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “The testing in research laboratories is not widely available and the results are often delayed by weeks to months,” he said.
In addition, while the associations between antibody results and risks of malignancy or pulmonary disease are “statistically valid,” he said, “there are patients with disease in whom antibodies are not present and those without associated disease in whom the testing was positive.” For example, there are patients positive for anti–transition initiation factor (TIF)-1gamma but don’t have a malignancy, “and the ones with anti-MDA-5 tend to have pulmonary disease, but there are patients with anti-MDA-5 who don’t have pulmonary disease.”
Compared with patients with systemic lupus erythematosus, patients with dermatomyositis tend to have more itching and they tend of have fewer serologic abnormalities, such as anti-Ro/SS-A antibody, “but there is overlap,” Dr. Callen said. “The reason to differentiate cutaneous lupus erythematosus from dermatomyositis is because we think that patients who have amyopathic dermatomyositis still have an increased risk of having or developing an internal malignancy,” he added. Another differentiating point that is substantive is the presence of Gottron papules.
In a recent development related to antibody testing, researchers demonstrated that the IgG2 isotype of anti-TIF-1gamma antibodies is a biomarker of cancer and mortality in adult dermatomyositis.
According to population-based studies, about 20%-25% of dermatomyositis patients have had, have, or will develop a cancer (Lancet 2001;357: 96-100). Amyopathic dermatomyositis patients may also have cancer. Polymyositis patients generally have lower rates and their risk of subsequent malignancy is much closer to that of the general population, suggesting that the presence of the association is due to a “diagnostic suspicion bias,” Dr. Callen said.
A large-scale multicenter cohort study that set out to identify the risk factors and prognosis of patients with cancer-associated myositis found that ovarian cancer seems to be overrepresented. The only serologic abnormality that was statistically significant was anti-TIF-1gamma antibody (P less than .001). Patients with cancer-associated myositis also have less overall survival compared with those with non–cancer-associated myositis (P = .004), with malignancy being the primary cause of death (P less than .001).
In what is believed to be the largest study of its kind, Dr. Callen and colleagues retrospectively examined the prevalence of malignancy and screening practices in 400 dermatomyositis patients. Of the 400 patients, 48 (12%) had malignancies, and 21 cancers (40%) were diagnosed within 1 year of the dermatomyositis diagnosis. Both classic dermatomyositis and amyopathic dermatomyositis were associated with cancer, and 27 patients (6.8%) had a cancer at the time of diagnosis. Of those, 59% were asymptomatic; their cancers were discovered with CT scans, suggesting that “blind” screening is effective in identifying cancers in DM patients.
Dr. Callen’s malignancy evaluation includes chest x-ray, CT of the chest and abdomen, stool Hematest in all dermatomyositis patients; a mammogram, pelvic ultrasound and/or CT of the pelvis in women; and age, race or ethnicity-related testing. “I generally reevaluate patients annually for 3 years, because data from epidemiologic studies suggest that after 3 years [from the initial diagnosis], the rates of malignancy return toward normal,” he said. “I also evaluate any new symptom that might be suggestive of malignancy. The remaining issue is how to handle a patient in remission for several years, but who develops a relapse. What I do is perform another malignancy assessment.”
According to results from a meta-analysis of risk factors and systematic review of screening approaches, factors that increase malignancy risk include dermatomyositis subtype (risk ratio, 2.21), older age (weighted mean difference 11.19), male gender (RR, 1.53), dysphagia (RR, 2.09), cutaneous necrosis (RR, 2.73), and positive anti-TIF-1gamma (RR, 4.41).
Factors associated with a decreased risk of malignancy include polymyositis (RR, 0.49), clinically amyopathic dermatomyositis subtypes (RR, 0.44), Raynaud’s phenomenon (RR, 0.61), interstitial lung disease (RR, 0.49), very high serum creatine kinase (WMD –1189.96) or lactate dehydrogenase levels (WMD –336.53), and anti-Jo1 (RR, 0.45) or anti-EJ (RR, 0.17) positivity.
The analysis also found that CT scanning of the thorax, abdomen and pelvis appeared to yield a high proportion of underlying asymptomatic cancers. Limited evidence relating to the utility of tumor markers and 18F-FDG PET/CT was available.
As for treatment, the use of tofacitinib for cutaneous lesions of dermatomyositis has been suggested in various studies. In a recent open-label study of 10 patients with dermatomyositis who took extended release the JAK inhibitor tofacitinib 11 mg daily for 12 weeks, half experienced moderate improvement in disease activity, and the other half experienced minimal improvement. JAK inhibitors have been used in patients with juvenile dermatomyositis.
Dr. Callen’s treatment approach with dermatomyositis patients includes recommendations for sunscreens and protective clothing, plus assessment of vitamin D levels. “I will use topical emollients, corticosteroids, and calcineurin inhibitors,” he said. “Antimalarials might be used. I generally reach for methotrexate or mycophenolate mofetil relatively early. IVIG has also been studied.” Off-label therapies that have been used include dapsone, thalidomide, leflunomide, sirolimus, chlorambucil, etanercept, infliximab, rituximab, apremilast, tofacitinib, lenabasum, and low-dose naltrexone.
Dr. Callen disclosed that he is a consultant to Genentech and is a member of the safety monitoring committee for Principia Biopharma. He holds equity in Celgene, Pfizer, 3M, Johnson & Johnson, Merck, Abbott Laboratories, AbbVie, Procter & Gamble, Gilead, Allergen, and Amgen.
MedscapeLive and this news organization are owned by the same parent company.
, according to Jeffrey P. Callen, MD.
That’s because the validity and reproducibility of testing in commercial laboratories remain questionable, Dr. Callen, professor of medicine and chief of the division of dermatology at the University of Louisville, Ky., said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “The testing in research laboratories is not widely available and the results are often delayed by weeks to months,” he said.
In addition, while the associations between antibody results and risks of malignancy or pulmonary disease are “statistically valid,” he said, “there are patients with disease in whom antibodies are not present and those without associated disease in whom the testing was positive.” For example, there are patients positive for anti–transition initiation factor (TIF)-1gamma but don’t have a malignancy, “and the ones with anti-MDA-5 tend to have pulmonary disease, but there are patients with anti-MDA-5 who don’t have pulmonary disease.”
Compared with patients with systemic lupus erythematosus, patients with dermatomyositis tend to have more itching and they tend of have fewer serologic abnormalities, such as anti-Ro/SS-A antibody, “but there is overlap,” Dr. Callen said. “The reason to differentiate cutaneous lupus erythematosus from dermatomyositis is because we think that patients who have amyopathic dermatomyositis still have an increased risk of having or developing an internal malignancy,” he added. Another differentiating point that is substantive is the presence of Gottron papules.
In a recent development related to antibody testing, researchers demonstrated that the IgG2 isotype of anti-TIF-1gamma antibodies is a biomarker of cancer and mortality in adult dermatomyositis.
According to population-based studies, about 20%-25% of dermatomyositis patients have had, have, or will develop a cancer (Lancet 2001;357: 96-100). Amyopathic dermatomyositis patients may also have cancer. Polymyositis patients generally have lower rates and their risk of subsequent malignancy is much closer to that of the general population, suggesting that the presence of the association is due to a “diagnostic suspicion bias,” Dr. Callen said.
A large-scale multicenter cohort study that set out to identify the risk factors and prognosis of patients with cancer-associated myositis found that ovarian cancer seems to be overrepresented. The only serologic abnormality that was statistically significant was anti-TIF-1gamma antibody (P less than .001). Patients with cancer-associated myositis also have less overall survival compared with those with non–cancer-associated myositis (P = .004), with malignancy being the primary cause of death (P less than .001).
In what is believed to be the largest study of its kind, Dr. Callen and colleagues retrospectively examined the prevalence of malignancy and screening practices in 400 dermatomyositis patients. Of the 400 patients, 48 (12%) had malignancies, and 21 cancers (40%) were diagnosed within 1 year of the dermatomyositis diagnosis. Both classic dermatomyositis and amyopathic dermatomyositis were associated with cancer, and 27 patients (6.8%) had a cancer at the time of diagnosis. Of those, 59% were asymptomatic; their cancers were discovered with CT scans, suggesting that “blind” screening is effective in identifying cancers in DM patients.
Dr. Callen’s malignancy evaluation includes chest x-ray, CT of the chest and abdomen, stool Hematest in all dermatomyositis patients; a mammogram, pelvic ultrasound and/or CT of the pelvis in women; and age, race or ethnicity-related testing. “I generally reevaluate patients annually for 3 years, because data from epidemiologic studies suggest that after 3 years [from the initial diagnosis], the rates of malignancy return toward normal,” he said. “I also evaluate any new symptom that might be suggestive of malignancy. The remaining issue is how to handle a patient in remission for several years, but who develops a relapse. What I do is perform another malignancy assessment.”
According to results from a meta-analysis of risk factors and systematic review of screening approaches, factors that increase malignancy risk include dermatomyositis subtype (risk ratio, 2.21), older age (weighted mean difference 11.19), male gender (RR, 1.53), dysphagia (RR, 2.09), cutaneous necrosis (RR, 2.73), and positive anti-TIF-1gamma (RR, 4.41).
Factors associated with a decreased risk of malignancy include polymyositis (RR, 0.49), clinically amyopathic dermatomyositis subtypes (RR, 0.44), Raynaud’s phenomenon (RR, 0.61), interstitial lung disease (RR, 0.49), very high serum creatine kinase (WMD –1189.96) or lactate dehydrogenase levels (WMD –336.53), and anti-Jo1 (RR, 0.45) or anti-EJ (RR, 0.17) positivity.
The analysis also found that CT scanning of the thorax, abdomen and pelvis appeared to yield a high proportion of underlying asymptomatic cancers. Limited evidence relating to the utility of tumor markers and 18F-FDG PET/CT was available.
As for treatment, the use of tofacitinib for cutaneous lesions of dermatomyositis has been suggested in various studies. In a recent open-label study of 10 patients with dermatomyositis who took extended release the JAK inhibitor tofacitinib 11 mg daily for 12 weeks, half experienced moderate improvement in disease activity, and the other half experienced minimal improvement. JAK inhibitors have been used in patients with juvenile dermatomyositis.
Dr. Callen’s treatment approach with dermatomyositis patients includes recommendations for sunscreens and protective clothing, plus assessment of vitamin D levels. “I will use topical emollients, corticosteroids, and calcineurin inhibitors,” he said. “Antimalarials might be used. I generally reach for methotrexate or mycophenolate mofetil relatively early. IVIG has also been studied.” Off-label therapies that have been used include dapsone, thalidomide, leflunomide, sirolimus, chlorambucil, etanercept, infliximab, rituximab, apremilast, tofacitinib, lenabasum, and low-dose naltrexone.
Dr. Callen disclosed that he is a consultant to Genentech and is a member of the safety monitoring committee for Principia Biopharma. He holds equity in Celgene, Pfizer, 3M, Johnson & Johnson, Merck, Abbott Laboratories, AbbVie, Procter & Gamble, Gilead, Allergen, and Amgen.
MedscapeLive and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Prevalence of undiagnosed vitiligo is ‘remarkably high’
A new

“The remarkably high number of participants with undiagnosed vitiligo” indicates a need for “the development and validation of teledermatology apps that allow for potential diagnosis,” Kavita Gandhi, MS, of the patient and health impact group at Pfizer in Collegeville, Pa., and associates said in JAMA Dermatology.
The estimated range of 0.76%-1.11% prevalence represents 1.9 million to 2.8 million adults with vitiligo in the general population, based on responses from 40,888 participants surveyed between Dec. 30, 2019, and March 11, 2020, and further physician evaluation of photos uploaded by 113 respondents, they explained. The investigators used a representative sample of the U.S. population, of people ages 18-85 years.
A prior vitiligo diagnosis was reported by 314 participants, and another 249 screened positive through the survey, for a self-reported overall prevalence of 1.38% in the adult population and a previously undiagnosed prevalence of 0.61%. The physician adjudication brought the overall prevalence down to 0.76% and the undiagnosed prevalence to 0.29%. “These findings suggest that up to 40% of adults with vitiligo in the U.S. may be undiagnosed,” the investigators wrote.
Survey questions covering the laterality of lesions broke the 1.38% overall prevalence down to 0.77% nonsegmental vitiligo (self-reported as bilateral) and 0.61% segmental (unilateral). The 0.76% overall prevalence provided by the three dermatologist reviewers worked out to 0.58% classified as nonsegmental and 0.18% as segmental, Ms. Gandhi and associates said.
“The distinction between segmental and nonsegmental vitiligo is of prime importance [since] patients are usually concerned by the spreading of the disease and its unpredictable course, which is the hallmark of nonsegmental vitiligo,” the researchers noted.
The analysis was the first, to the authors’ knowledge, to identify several trends among the undiagnosed population. The proportion of nonwhite adults was higher in the undiagnosed group (40.2%) than among those with a diagnosis (31.5%), as was Hispanic, Latino, or Spanish origin (21.3% vs. 15.3%). Unilateral presentation was seen in 54.2% of the undiagnosed adults and 37.3% of those with diagnosed vitiligo, they reported.
The study was sponsored by Pfizer, which employs several of the investigators. Two of the investigators disclosed multiple conflicts of interest involving other companies.
A new

“The remarkably high number of participants with undiagnosed vitiligo” indicates a need for “the development and validation of teledermatology apps that allow for potential diagnosis,” Kavita Gandhi, MS, of the patient and health impact group at Pfizer in Collegeville, Pa., and associates said in JAMA Dermatology.
The estimated range of 0.76%-1.11% prevalence represents 1.9 million to 2.8 million adults with vitiligo in the general population, based on responses from 40,888 participants surveyed between Dec. 30, 2019, and March 11, 2020, and further physician evaluation of photos uploaded by 113 respondents, they explained. The investigators used a representative sample of the U.S. population, of people ages 18-85 years.
A prior vitiligo diagnosis was reported by 314 participants, and another 249 screened positive through the survey, for a self-reported overall prevalence of 1.38% in the adult population and a previously undiagnosed prevalence of 0.61%. The physician adjudication brought the overall prevalence down to 0.76% and the undiagnosed prevalence to 0.29%. “These findings suggest that up to 40% of adults with vitiligo in the U.S. may be undiagnosed,” the investigators wrote.
Survey questions covering the laterality of lesions broke the 1.38% overall prevalence down to 0.77% nonsegmental vitiligo (self-reported as bilateral) and 0.61% segmental (unilateral). The 0.76% overall prevalence provided by the three dermatologist reviewers worked out to 0.58% classified as nonsegmental and 0.18% as segmental, Ms. Gandhi and associates said.
“The distinction between segmental and nonsegmental vitiligo is of prime importance [since] patients are usually concerned by the spreading of the disease and its unpredictable course, which is the hallmark of nonsegmental vitiligo,” the researchers noted.
The analysis was the first, to the authors’ knowledge, to identify several trends among the undiagnosed population. The proportion of nonwhite adults was higher in the undiagnosed group (40.2%) than among those with a diagnosis (31.5%), as was Hispanic, Latino, or Spanish origin (21.3% vs. 15.3%). Unilateral presentation was seen in 54.2% of the undiagnosed adults and 37.3% of those with diagnosed vitiligo, they reported.
The study was sponsored by Pfizer, which employs several of the investigators. Two of the investigators disclosed multiple conflicts of interest involving other companies.
A new

“The remarkably high number of participants with undiagnosed vitiligo” indicates a need for “the development and validation of teledermatology apps that allow for potential diagnosis,” Kavita Gandhi, MS, of the patient and health impact group at Pfizer in Collegeville, Pa., and associates said in JAMA Dermatology.
The estimated range of 0.76%-1.11% prevalence represents 1.9 million to 2.8 million adults with vitiligo in the general population, based on responses from 40,888 participants surveyed between Dec. 30, 2019, and March 11, 2020, and further physician evaluation of photos uploaded by 113 respondents, they explained. The investigators used a representative sample of the U.S. population, of people ages 18-85 years.
A prior vitiligo diagnosis was reported by 314 participants, and another 249 screened positive through the survey, for a self-reported overall prevalence of 1.38% in the adult population and a previously undiagnosed prevalence of 0.61%. The physician adjudication brought the overall prevalence down to 0.76% and the undiagnosed prevalence to 0.29%. “These findings suggest that up to 40% of adults with vitiligo in the U.S. may be undiagnosed,” the investigators wrote.
Survey questions covering the laterality of lesions broke the 1.38% overall prevalence down to 0.77% nonsegmental vitiligo (self-reported as bilateral) and 0.61% segmental (unilateral). The 0.76% overall prevalence provided by the three dermatologist reviewers worked out to 0.58% classified as nonsegmental and 0.18% as segmental, Ms. Gandhi and associates said.
“The distinction between segmental and nonsegmental vitiligo is of prime importance [since] patients are usually concerned by the spreading of the disease and its unpredictable course, which is the hallmark of nonsegmental vitiligo,” the researchers noted.
The analysis was the first, to the authors’ knowledge, to identify several trends among the undiagnosed population. The proportion of nonwhite adults was higher in the undiagnosed group (40.2%) than among those with a diagnosis (31.5%), as was Hispanic, Latino, or Spanish origin (21.3% vs. 15.3%). Unilateral presentation was seen in 54.2% of the undiagnosed adults and 37.3% of those with diagnosed vitiligo, they reported.
The study was sponsored by Pfizer, which employs several of the investigators. Two of the investigators disclosed multiple conflicts of interest involving other companies.
FROM JAMA DERMATOLOGY
Rosacea is in the eye of the beholder, expert says
In the clinical experience of Emmy Graber, MD, MBA, rosacea is in the eye of the beholder.
“It’s not really up to us as the providers as to what’s important to the patient or how bad their rosacea is,” she said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It really is up to the patient,” added Dr. Graber, president of The Dermatology Institute of Boston, who recommends asking patients about how severe they consider their rosacea to be, and what about rosacea bothers them most. Their responses may be surprising.
A study published in 2017 showed that complete resolution of even mild rosacea prolongs remission of rosacea, and most importantly, improves the quality of life for patients. “So, don’t discount what you consider to be mild rosacea in patients,” she said.
Skin care recommendations
“And don’t forget about basic skin care,” she advised. A recently published Chinese study of 999 rosacea patients and 1,010 controls with healthy skin found that a high frequency of cleansing and expansive use of cleansers were positively correlated with rosacea occurrence, suggesting that overcleansing can be a risk factor for rosacea. “Ask your patient, ‘how often are you cleaning your face?’ ” Dr. Graber suggested. “You might find that they’re overdoing it by washing three or four times a day. Several studies have shown that basic skin care alone improves rosacea.”
Skin care recommendations for patients with rosacea include avoiding chemical or physical exfoliants and alcohol-based topical products, and moisturizing and washing their faces with mild, synthetic detergent-based products rather than traditional soaps, which may further alkalinize and irritate the skin. “Patients should also be counseled to use physical-based sunscreens rather than chemical-based sunscreens,” she said.
Treating erythema
For treating erythema with topicals, a systematic review published in 2019 found the most evidence for brimonidine 0.33% gel, an alpha2-adrenergic agonist, and oxymetazoline 1% cream, an alpha1-adrenergic agonist. “Both of these products functionally constrict facial blood vessels,” and are Food and Drug Administration approved for treating persistent erythema, Dr. Graber said. “These products improve erythema within 3 hours of and up to 12 hours after application and overall, they are well tolerated.”
Based on clinical trial results, about 15% of patients on brimonidine report adverse reactions such as dermatitis, burning, pruritus, and erythema, compared with 8% of patients on oxymetazoline. At the same time, up to 20% of individuals on brimonidine report rebound erythema, compared with fewer than 1% of those using oxymetazoline. Laser and light therapies such as pulse-dye lasers, potassium-titanyl-phosphate lasers, and intense-pulse light devices are also effective in treating persistent erythema but are less effective for transient flushing.
Treatment of papules and pustules
For treating papules and pustules, the 2019 systemic review also found high-certainty evidence for using azelaic acid and topical ivermectin, and moderate-certainty evidence for using topical metronidazole and topical minocycline. “Topical ivermectin was demonstrated to be the most effective topical treatment for papulopustular rosacea and to provide the greatest psychological benefit to these patients,” Dr. Graber said.
In a double-blind, multicenter 15-week trial comparing azelaic acid 15% gel with metronidazole 0.75% gel in patients with papulopustular rosacea, both agents were found to be effective. But those treated with azelaic acid 15% gel had a greater reduction in lesion counts and erythema, and improvement in global assessments, compared with metronidazole 0.75% gel. However, the azelaic acid 15% gel was associated with more stinging compared with metronidazole 0.75% gel, although it was usually transient.
Another study, a double-blind, single-center, 15-week trial, compared the efficacy of azelaic acid 20% cream with metronidazole 0.75% cream. Both agents were found to be effective and had similar levels of reductions in papules and pustules. However, patients in the azelaic acid 20% cream arm had significantly higher physician ratings of global improvement, as well as overall higher patient satisfaction.
More recently, a phase 3 study of 962 patients found that ivermectin 1% cream once daily improved quality of life slightly more than metronidazole 0.75% cream twice daily. No difference in adverse events were noted between the two agents.
Other options for treating papules and pustules include topical minocycline 1.5% foam, which is FDA approved for rosacea, as well as second-line agents topical sodium sulfacetamide with sulfur cleanser (cream or lotion), and permethrin, Dr. Graber said.
As for treating papules and pustules with oral agents, the strongest evidence favors oral tetracyclines and isotretinoin, she noted.
Doxycycline, minocycline, tetracycline, and sarecycline can be used as monotherapy or coadministered with topical agents. “The addition of topical agents may also help to shorten the duration of antibiotic use, which is very important,” Dr. Graber said.
She noted that oral beta-blockers might be useful to treat persistent erythema and flushing because they antagonize the effects of sympathetic nerve stimulation and circulating catecholamines at b-adrenoceptors. Carvedilol and propranolol have been the most studied. The most common potential side effects are hypotension and bradycardia.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
In the clinical experience of Emmy Graber, MD, MBA, rosacea is in the eye of the beholder.
“It’s not really up to us as the providers as to what’s important to the patient or how bad their rosacea is,” she said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It really is up to the patient,” added Dr. Graber, president of The Dermatology Institute of Boston, who recommends asking patients about how severe they consider their rosacea to be, and what about rosacea bothers them most. Their responses may be surprising.
A study published in 2017 showed that complete resolution of even mild rosacea prolongs remission of rosacea, and most importantly, improves the quality of life for patients. “So, don’t discount what you consider to be mild rosacea in patients,” she said.
Skin care recommendations
“And don’t forget about basic skin care,” she advised. A recently published Chinese study of 999 rosacea patients and 1,010 controls with healthy skin found that a high frequency of cleansing and expansive use of cleansers were positively correlated with rosacea occurrence, suggesting that overcleansing can be a risk factor for rosacea. “Ask your patient, ‘how often are you cleaning your face?’ ” Dr. Graber suggested. “You might find that they’re overdoing it by washing three or four times a day. Several studies have shown that basic skin care alone improves rosacea.”
Skin care recommendations for patients with rosacea include avoiding chemical or physical exfoliants and alcohol-based topical products, and moisturizing and washing their faces with mild, synthetic detergent-based products rather than traditional soaps, which may further alkalinize and irritate the skin. “Patients should also be counseled to use physical-based sunscreens rather than chemical-based sunscreens,” she said.
Treating erythema
For treating erythema with topicals, a systematic review published in 2019 found the most evidence for brimonidine 0.33% gel, an alpha2-adrenergic agonist, and oxymetazoline 1% cream, an alpha1-adrenergic agonist. “Both of these products functionally constrict facial blood vessels,” and are Food and Drug Administration approved for treating persistent erythema, Dr. Graber said. “These products improve erythema within 3 hours of and up to 12 hours after application and overall, they are well tolerated.”
Based on clinical trial results, about 15% of patients on brimonidine report adverse reactions such as dermatitis, burning, pruritus, and erythema, compared with 8% of patients on oxymetazoline. At the same time, up to 20% of individuals on brimonidine report rebound erythema, compared with fewer than 1% of those using oxymetazoline. Laser and light therapies such as pulse-dye lasers, potassium-titanyl-phosphate lasers, and intense-pulse light devices are also effective in treating persistent erythema but are less effective for transient flushing.
Treatment of papules and pustules
For treating papules and pustules, the 2019 systemic review also found high-certainty evidence for using azelaic acid and topical ivermectin, and moderate-certainty evidence for using topical metronidazole and topical minocycline. “Topical ivermectin was demonstrated to be the most effective topical treatment for papulopustular rosacea and to provide the greatest psychological benefit to these patients,” Dr. Graber said.
In a double-blind, multicenter 15-week trial comparing azelaic acid 15% gel with metronidazole 0.75% gel in patients with papulopustular rosacea, both agents were found to be effective. But those treated with azelaic acid 15% gel had a greater reduction in lesion counts and erythema, and improvement in global assessments, compared with metronidazole 0.75% gel. However, the azelaic acid 15% gel was associated with more stinging compared with metronidazole 0.75% gel, although it was usually transient.
Another study, a double-blind, single-center, 15-week trial, compared the efficacy of azelaic acid 20% cream with metronidazole 0.75% cream. Both agents were found to be effective and had similar levels of reductions in papules and pustules. However, patients in the azelaic acid 20% cream arm had significantly higher physician ratings of global improvement, as well as overall higher patient satisfaction.
More recently, a phase 3 study of 962 patients found that ivermectin 1% cream once daily improved quality of life slightly more than metronidazole 0.75% cream twice daily. No difference in adverse events were noted between the two agents.
Other options for treating papules and pustules include topical minocycline 1.5% foam, which is FDA approved for rosacea, as well as second-line agents topical sodium sulfacetamide with sulfur cleanser (cream or lotion), and permethrin, Dr. Graber said.
As for treating papules and pustules with oral agents, the strongest evidence favors oral tetracyclines and isotretinoin, she noted.
Doxycycline, minocycline, tetracycline, and sarecycline can be used as monotherapy or coadministered with topical agents. “The addition of topical agents may also help to shorten the duration of antibiotic use, which is very important,” Dr. Graber said.
She noted that oral beta-blockers might be useful to treat persistent erythema and flushing because they antagonize the effects of sympathetic nerve stimulation and circulating catecholamines at b-adrenoceptors. Carvedilol and propranolol have been the most studied. The most common potential side effects are hypotension and bradycardia.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
In the clinical experience of Emmy Graber, MD, MBA, rosacea is in the eye of the beholder.
“It’s not really up to us as the providers as to what’s important to the patient or how bad their rosacea is,” she said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “It really is up to the patient,” added Dr. Graber, president of The Dermatology Institute of Boston, who recommends asking patients about how severe they consider their rosacea to be, and what about rosacea bothers them most. Their responses may be surprising.
A study published in 2017 showed that complete resolution of even mild rosacea prolongs remission of rosacea, and most importantly, improves the quality of life for patients. “So, don’t discount what you consider to be mild rosacea in patients,” she said.
Skin care recommendations
“And don’t forget about basic skin care,” she advised. A recently published Chinese study of 999 rosacea patients and 1,010 controls with healthy skin found that a high frequency of cleansing and expansive use of cleansers were positively correlated with rosacea occurrence, suggesting that overcleansing can be a risk factor for rosacea. “Ask your patient, ‘how often are you cleaning your face?’ ” Dr. Graber suggested. “You might find that they’re overdoing it by washing three or four times a day. Several studies have shown that basic skin care alone improves rosacea.”
Skin care recommendations for patients with rosacea include avoiding chemical or physical exfoliants and alcohol-based topical products, and moisturizing and washing their faces with mild, synthetic detergent-based products rather than traditional soaps, which may further alkalinize and irritate the skin. “Patients should also be counseled to use physical-based sunscreens rather than chemical-based sunscreens,” she said.
Treating erythema
For treating erythema with topicals, a systematic review published in 2019 found the most evidence for brimonidine 0.33% gel, an alpha2-adrenergic agonist, and oxymetazoline 1% cream, an alpha1-adrenergic agonist. “Both of these products functionally constrict facial blood vessels,” and are Food and Drug Administration approved for treating persistent erythema, Dr. Graber said. “These products improve erythema within 3 hours of and up to 12 hours after application and overall, they are well tolerated.”
Based on clinical trial results, about 15% of patients on brimonidine report adverse reactions such as dermatitis, burning, pruritus, and erythema, compared with 8% of patients on oxymetazoline. At the same time, up to 20% of individuals on brimonidine report rebound erythema, compared with fewer than 1% of those using oxymetazoline. Laser and light therapies such as pulse-dye lasers, potassium-titanyl-phosphate lasers, and intense-pulse light devices are also effective in treating persistent erythema but are less effective for transient flushing.
Treatment of papules and pustules
For treating papules and pustules, the 2019 systemic review also found high-certainty evidence for using azelaic acid and topical ivermectin, and moderate-certainty evidence for using topical metronidazole and topical minocycline. “Topical ivermectin was demonstrated to be the most effective topical treatment for papulopustular rosacea and to provide the greatest psychological benefit to these patients,” Dr. Graber said.
In a double-blind, multicenter 15-week trial comparing azelaic acid 15% gel with metronidazole 0.75% gel in patients with papulopustular rosacea, both agents were found to be effective. But those treated with azelaic acid 15% gel had a greater reduction in lesion counts and erythema, and improvement in global assessments, compared with metronidazole 0.75% gel. However, the azelaic acid 15% gel was associated with more stinging compared with metronidazole 0.75% gel, although it was usually transient.
Another study, a double-blind, single-center, 15-week trial, compared the efficacy of azelaic acid 20% cream with metronidazole 0.75% cream. Both agents were found to be effective and had similar levels of reductions in papules and pustules. However, patients in the azelaic acid 20% cream arm had significantly higher physician ratings of global improvement, as well as overall higher patient satisfaction.
More recently, a phase 3 study of 962 patients found that ivermectin 1% cream once daily improved quality of life slightly more than metronidazole 0.75% cream twice daily. No difference in adverse events were noted between the two agents.
Other options for treating papules and pustules include topical minocycline 1.5% foam, which is FDA approved for rosacea, as well as second-line agents topical sodium sulfacetamide with sulfur cleanser (cream or lotion), and permethrin, Dr. Graber said.
As for treating papules and pustules with oral agents, the strongest evidence favors oral tetracyclines and isotretinoin, she noted.
Doxycycline, minocycline, tetracycline, and sarecycline can be used as monotherapy or coadministered with topical agents. “The addition of topical agents may also help to shorten the duration of antibiotic use, which is very important,” Dr. Graber said.
She noted that oral beta-blockers might be useful to treat persistent erythema and flushing because they antagonize the effects of sympathetic nerve stimulation and circulating catecholamines at b-adrenoceptors. Carvedilol and propranolol have been the most studied. The most common potential side effects are hypotension and bradycardia.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
FROM MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR