User login
Microbiome studies among those awarded National Rosacea Society grants
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
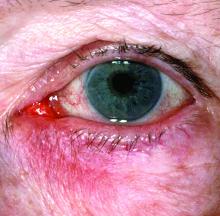
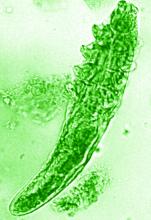
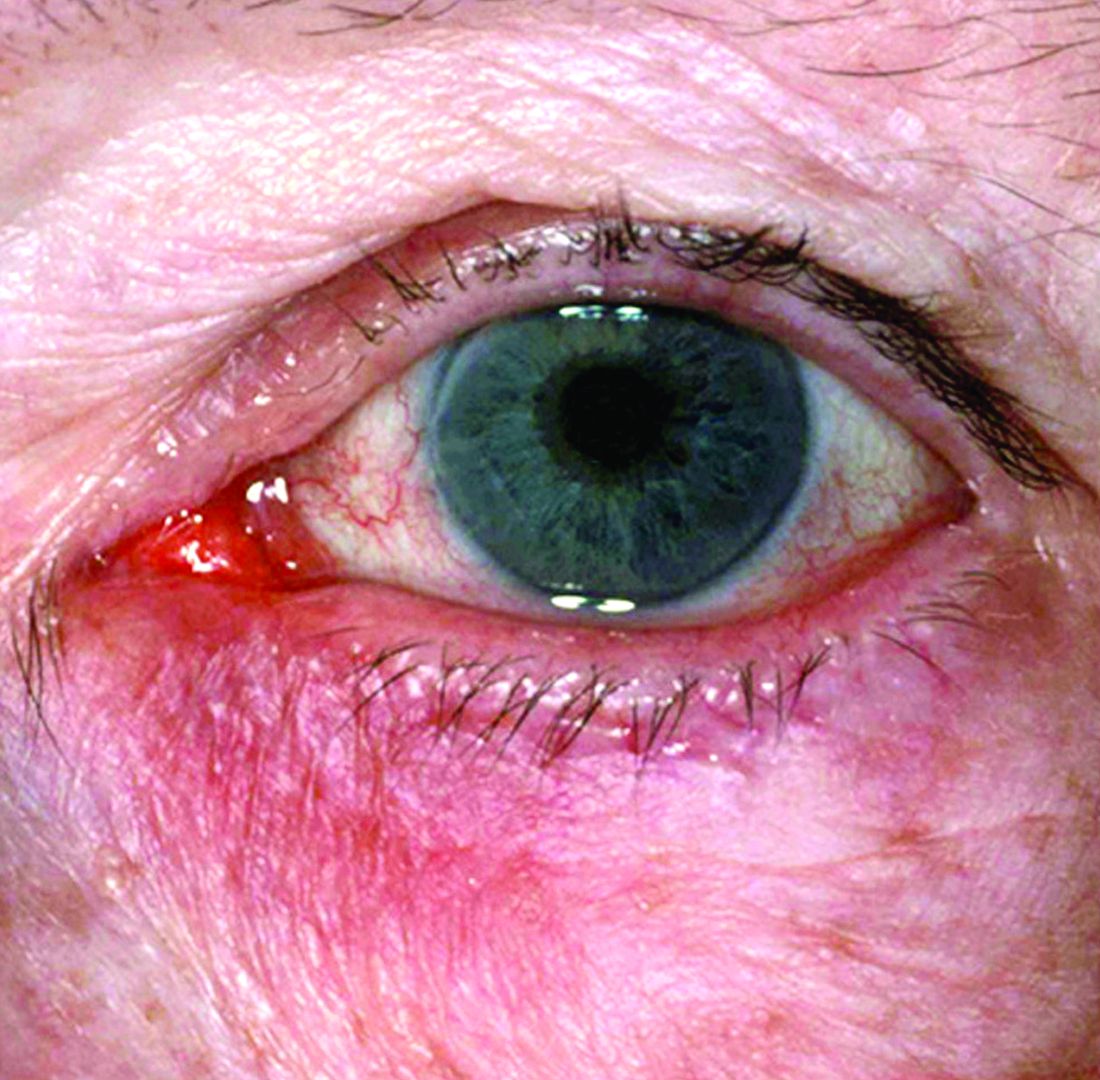
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at info@rosacea.org.
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at info@rosacea.org.
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at info@rosacea.org.
Topical options for acne patients continue to expand
yet they are likely underused in today’s clinical practice.
A study of prescribing practices from 2012 to 2014 indicated that dermatologists prescribed retinoids for just 58.8% of acne cases, while nondermatologists prescribed them for only 32.4% of cases. “If the guidelines are telling us that we should use topical retinoids for almost all of our acne patients, why are we using them for half of the patients?” Emmy Graber, MD, MBA, asked during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We have a lot of options today for topical retinoids,” she added, noting that, in the past few years, trifarotene cream 0.005% and new formulations of tazarotene lotion (0.045%) and tretinoin lotion (0.05%) have become available.
According to Dr. Graber, president of The Dermatology Institute of Boston, tazarotene has been considered the most efficacious topical retinoid but is generally the least well tolerated, while adapalene has often been considered to be one of the better-tolerated topical retinoids. “This is a broad generalization,” she said. “One should also take into account the concentration and formulation of the retinoid. Cutaneous adverse events increase in severity as the concentration increases regardless of the vehicle.” There are no studies comparing trifarotene with other topical retinoids, she added.
In two phase 2, double-blind, vehicle-controlled studies (PERFECT 1 and PERFECT 2), researchers randomized more than 2,400 patients with moderate facial or truncal acne to receive trifarotene cream or a vehicle for 12 weeks. The mean percent change from baseline in facial inflammatory lesions in the trifarotene-treated group was –54.4% and –66.2% in PERFECT 1, and PERFECT 2, respectively, while the mean percent change from baseline in facial noninflammatory lesions was –49.7% and –57.7%, respectively.
In addition, the mean percent change from baseline in truncal inflammatory lesions in the trifarotene-treated groups was –57.4% and –65.4%, respectively, while the mean percent change from baseline in truncal noninflammatory lesions was –49.1% and –55.2%, respectively.
The choice of vehicle may affect absorption of topical retinoids, and some formulations may increase skin hydration and decrease transepidermal water loss, “which is a good thing,” Dr. Graber said. “Also, vehicles aim to slow drug delivery over time while also making sure that the drug penetrates into the pilosebaceous unit.”
One recent advance is the honeycomb-like polymeric emulsion technology found in tretinoin 0.05% lotion and tazarotene 0.045% lotion. These formulations contain droplets of the tretinoin and tazarotene embedded in a honeycomb matrix with hydrating agents. “I think this is exciting and could enhance our patient compliance and tolerability,” she said. Another unique feature about these two products, especially the tretinoin product, is the very small particle size with this new formulation. “It’s small enough that it can penetrate down into the pilosebaceous unit,” which is different than with older formulations, in which the tretinoin “largely just sat on the surface of the skin and didn’t penetrate into the pilosebaceous unit.” In addition, she said, “there’s only 9% degradation of the tretinoin in UV light, compared to 72% degradation of standard tretinoin 0.025% gel, and with the new tretinoin formulation, there’s no degradation when used with benzoyl peroxide.”
Another new topical retinoid to consider is a fixed-dose combination of encapsulated benzoyl peroxide 3% and encapsulated tretinoin 0.1% cream (Twyneo), which was approved by the Food and Drug Administration in July 2021 for the treatment of acne in adults and children aged 9 years and older. “Typically, benzoyl peroxide and tretinoin cannot be mixed in the same tube to stability issues,” she said. “Here, each product is individually encapsulated in a silica shell so that they can be applied together.”
The approval was supported by positive results from two phase 3, randomized, double-blind, vehicle-controlled, multicenter studies (NCT03761784 and NCT03761810), in which Twyneo demonstrated efficacy and a favorable tolerability profile in patients aged 9 years and older with facial acne.
Another topical treatment option, dapsone, is now FDA approved for ages 9 and up, expanded from its initial indication for ages 12 and up. The new indication is based on a phase 4, multicenter, open-label study in which acne patients aged 9-11 years applied dapsone 7.5% gel once daily to the face and acne-affected areas on the upper chest, upper back, and shoulders for 12 weeks. After 12 weeks, facial acne was clear or almost clear in about 47% of patients. “Inflammatory, noninflammatory, and total lesions decreased from baseline, but there was a greater reduction in noninflammatory lesions, so if you have a very young patient with acne, now you can consider dapsone gel,” Dr. Graber said.
In August 2020, clascoterone cream became the first topical androgen receptor inhibitor approved for the treatment of acne in patients 12 years of age and older. It is a drug believed to address sebum and inflammation directly in the sebaceous gland and is structurally similar to dihydrotestosterone and spironolactone.
“This is a completely new drug category in acne,” she said. “Unlike all oral antiandrogen therapies, clascoterone cream can be used in both males and females with acne. It’s the first acne drug to have a new mechanism of action in almost 40 years, since isotretinoin was approved in 1982.”
In vitro, she continued, clascoterone competes with dihydrotestosterone for binding to the androgen receptor, inhibiting downstream signaling and leading to inhibited sebum production, reduced secretion of inflammatory cytokines, and inhibition of inflammatory pathways. Two phase 3 studies that led to its approval involved 1,440 patients with moderate to severe facial acne aged 9-58 years. The cream was applied twice a day for 12 weeks and treatment adherence was approximately 90%. The researchers found that clascoterone cream was significantly more effective than vehicle cream at achieving Investigator’s Global Assessment scores of 0 (clear) or 1 (almost clear), the definition of treatment success in the study, and reducing noninflammatory lesion and inflammatory lesion counts at week 12. “There were no safety issues noted during these studies, and clascoterone cream was well tolerated,” Dr. Graber said.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
Commentary by Lawrence W. Eichenfield, MD
Acne vulgaris remains an issue of tremendous importance to preteens, teens, and young adults, with approximately 85% of individuals aged 12-24 being affected. Expanding options for topical treatments may help bring effective disease control. Dr. Graber pointed out that historically, pediatricians and other primary care practitioners utilize topical retinoids less often for acne care as compared with dermatologists or guidelines recommendations (either the AAP’s or AAD’s). There are now expanded options, including over-the-counter retinoids (adapalene 0.1% gel), generic and trade brand topical tretinoin products, prescription adapalene medications, older and recently approved tazarotene products, and a newer type of topical retinoid, trifarotene. Novel formulations and emulsion technology, as well as retinoid developed in combination products, give more options in patients down to 9 years of age. A novel topical anti-androgen, clascoterone, is in its own category, as the first topical “hormonal agent,” allowing hormonal therapy to be used for males as well as females (aged 12 years and up). A recent review in JAMA (2021 Nov 23;326[20]:2055-67) incorporates many of these newer medications into management suggestions, emphasizing that first-line therapies are topical retinoids, benzoyl peroxide, azelaic acid, or combinations of topicals, whereas in more severe disease, oral antibiotics such as doxycycline or minocycline, hormonal therapies such as combination oral conceptive agents or spironolactone, or isotretinoin are most effective.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
yet they are likely underused in today’s clinical practice.
A study of prescribing practices from 2012 to 2014 indicated that dermatologists prescribed retinoids for just 58.8% of acne cases, while nondermatologists prescribed them for only 32.4% of cases. “If the guidelines are telling us that we should use topical retinoids for almost all of our acne patients, why are we using them for half of the patients?” Emmy Graber, MD, MBA, asked during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We have a lot of options today for topical retinoids,” she added, noting that, in the past few years, trifarotene cream 0.005% and new formulations of tazarotene lotion (0.045%) and tretinoin lotion (0.05%) have become available.
According to Dr. Graber, president of The Dermatology Institute of Boston, tazarotene has been considered the most efficacious topical retinoid but is generally the least well tolerated, while adapalene has often been considered to be one of the better-tolerated topical retinoids. “This is a broad generalization,” she said. “One should also take into account the concentration and formulation of the retinoid. Cutaneous adverse events increase in severity as the concentration increases regardless of the vehicle.” There are no studies comparing trifarotene with other topical retinoids, she added.
In two phase 2, double-blind, vehicle-controlled studies (PERFECT 1 and PERFECT 2), researchers randomized more than 2,400 patients with moderate facial or truncal acne to receive trifarotene cream or a vehicle for 12 weeks. The mean percent change from baseline in facial inflammatory lesions in the trifarotene-treated group was –54.4% and –66.2% in PERFECT 1, and PERFECT 2, respectively, while the mean percent change from baseline in facial noninflammatory lesions was –49.7% and –57.7%, respectively.
In addition, the mean percent change from baseline in truncal inflammatory lesions in the trifarotene-treated groups was –57.4% and –65.4%, respectively, while the mean percent change from baseline in truncal noninflammatory lesions was –49.1% and –55.2%, respectively.
The choice of vehicle may affect absorption of topical retinoids, and some formulations may increase skin hydration and decrease transepidermal water loss, “which is a good thing,” Dr. Graber said. “Also, vehicles aim to slow drug delivery over time while also making sure that the drug penetrates into the pilosebaceous unit.”
One recent advance is the honeycomb-like polymeric emulsion technology found in tretinoin 0.05% lotion and tazarotene 0.045% lotion. These formulations contain droplets of the tretinoin and tazarotene embedded in a honeycomb matrix with hydrating agents. “I think this is exciting and could enhance our patient compliance and tolerability,” she said. Another unique feature about these two products, especially the tretinoin product, is the very small particle size with this new formulation. “It’s small enough that it can penetrate down into the pilosebaceous unit,” which is different than with older formulations, in which the tretinoin “largely just sat on the surface of the skin and didn’t penetrate into the pilosebaceous unit.” In addition, she said, “there’s only 9% degradation of the tretinoin in UV light, compared to 72% degradation of standard tretinoin 0.025% gel, and with the new tretinoin formulation, there’s no degradation when used with benzoyl peroxide.”
Another new topical retinoid to consider is a fixed-dose combination of encapsulated benzoyl peroxide 3% and encapsulated tretinoin 0.1% cream (Twyneo), which was approved by the Food and Drug Administration in July 2021 for the treatment of acne in adults and children aged 9 years and older. “Typically, benzoyl peroxide and tretinoin cannot be mixed in the same tube to stability issues,” she said. “Here, each product is individually encapsulated in a silica shell so that they can be applied together.”
The approval was supported by positive results from two phase 3, randomized, double-blind, vehicle-controlled, multicenter studies (NCT03761784 and NCT03761810), in which Twyneo demonstrated efficacy and a favorable tolerability profile in patients aged 9 years and older with facial acne.
Another topical treatment option, dapsone, is now FDA approved for ages 9 and up, expanded from its initial indication for ages 12 and up. The new indication is based on a phase 4, multicenter, open-label study in which acne patients aged 9-11 years applied dapsone 7.5% gel once daily to the face and acne-affected areas on the upper chest, upper back, and shoulders for 12 weeks. After 12 weeks, facial acne was clear or almost clear in about 47% of patients. “Inflammatory, noninflammatory, and total lesions decreased from baseline, but there was a greater reduction in noninflammatory lesions, so if you have a very young patient with acne, now you can consider dapsone gel,” Dr. Graber said.
In August 2020, clascoterone cream became the first topical androgen receptor inhibitor approved for the treatment of acne in patients 12 years of age and older. It is a drug believed to address sebum and inflammation directly in the sebaceous gland and is structurally similar to dihydrotestosterone and spironolactone.
“This is a completely new drug category in acne,” she said. “Unlike all oral antiandrogen therapies, clascoterone cream can be used in both males and females with acne. It’s the first acne drug to have a new mechanism of action in almost 40 years, since isotretinoin was approved in 1982.”
In vitro, she continued, clascoterone competes with dihydrotestosterone for binding to the androgen receptor, inhibiting downstream signaling and leading to inhibited sebum production, reduced secretion of inflammatory cytokines, and inhibition of inflammatory pathways. Two phase 3 studies that led to its approval involved 1,440 patients with moderate to severe facial acne aged 9-58 years. The cream was applied twice a day for 12 weeks and treatment adherence was approximately 90%. The researchers found that clascoterone cream was significantly more effective than vehicle cream at achieving Investigator’s Global Assessment scores of 0 (clear) or 1 (almost clear), the definition of treatment success in the study, and reducing noninflammatory lesion and inflammatory lesion counts at week 12. “There were no safety issues noted during these studies, and clascoterone cream was well tolerated,” Dr. Graber said.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
Commentary by Lawrence W. Eichenfield, MD
Acne vulgaris remains an issue of tremendous importance to preteens, teens, and young adults, with approximately 85% of individuals aged 12-24 being affected. Expanding options for topical treatments may help bring effective disease control. Dr. Graber pointed out that historically, pediatricians and other primary care practitioners utilize topical retinoids less often for acne care as compared with dermatologists or guidelines recommendations (either the AAP’s or AAD’s). There are now expanded options, including over-the-counter retinoids (adapalene 0.1% gel), generic and trade brand topical tretinoin products, prescription adapalene medications, older and recently approved tazarotene products, and a newer type of topical retinoid, trifarotene. Novel formulations and emulsion technology, as well as retinoid developed in combination products, give more options in patients down to 9 years of age. A novel topical anti-androgen, clascoterone, is in its own category, as the first topical “hormonal agent,” allowing hormonal therapy to be used for males as well as females (aged 12 years and up). A recent review in JAMA (2021 Nov 23;326[20]:2055-67) incorporates many of these newer medications into management suggestions, emphasizing that first-line therapies are topical retinoids, benzoyl peroxide, azelaic acid, or combinations of topicals, whereas in more severe disease, oral antibiotics such as doxycycline or minocycline, hormonal therapies such as combination oral conceptive agents or spironolactone, or isotretinoin are most effective.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
yet they are likely underused in today’s clinical practice.
A study of prescribing practices from 2012 to 2014 indicated that dermatologists prescribed retinoids for just 58.8% of acne cases, while nondermatologists prescribed them for only 32.4% of cases. “If the guidelines are telling us that we should use topical retinoids for almost all of our acne patients, why are we using them for half of the patients?” Emmy Graber, MD, MBA, asked during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We have a lot of options today for topical retinoids,” she added, noting that, in the past few years, trifarotene cream 0.005% and new formulations of tazarotene lotion (0.045%) and tretinoin lotion (0.05%) have become available.
According to Dr. Graber, president of The Dermatology Institute of Boston, tazarotene has been considered the most efficacious topical retinoid but is generally the least well tolerated, while adapalene has often been considered to be one of the better-tolerated topical retinoids. “This is a broad generalization,” she said. “One should also take into account the concentration and formulation of the retinoid. Cutaneous adverse events increase in severity as the concentration increases regardless of the vehicle.” There are no studies comparing trifarotene with other topical retinoids, she added.
In two phase 2, double-blind, vehicle-controlled studies (PERFECT 1 and PERFECT 2), researchers randomized more than 2,400 patients with moderate facial or truncal acne to receive trifarotene cream or a vehicle for 12 weeks. The mean percent change from baseline in facial inflammatory lesions in the trifarotene-treated group was –54.4% and –66.2% in PERFECT 1, and PERFECT 2, respectively, while the mean percent change from baseline in facial noninflammatory lesions was –49.7% and –57.7%, respectively.
In addition, the mean percent change from baseline in truncal inflammatory lesions in the trifarotene-treated groups was –57.4% and –65.4%, respectively, while the mean percent change from baseline in truncal noninflammatory lesions was –49.1% and –55.2%, respectively.
The choice of vehicle may affect absorption of topical retinoids, and some formulations may increase skin hydration and decrease transepidermal water loss, “which is a good thing,” Dr. Graber said. “Also, vehicles aim to slow drug delivery over time while also making sure that the drug penetrates into the pilosebaceous unit.”
One recent advance is the honeycomb-like polymeric emulsion technology found in tretinoin 0.05% lotion and tazarotene 0.045% lotion. These formulations contain droplets of the tretinoin and tazarotene embedded in a honeycomb matrix with hydrating agents. “I think this is exciting and could enhance our patient compliance and tolerability,” she said. Another unique feature about these two products, especially the tretinoin product, is the very small particle size with this new formulation. “It’s small enough that it can penetrate down into the pilosebaceous unit,” which is different than with older formulations, in which the tretinoin “largely just sat on the surface of the skin and didn’t penetrate into the pilosebaceous unit.” In addition, she said, “there’s only 9% degradation of the tretinoin in UV light, compared to 72% degradation of standard tretinoin 0.025% gel, and with the new tretinoin formulation, there’s no degradation when used with benzoyl peroxide.”
Another new topical retinoid to consider is a fixed-dose combination of encapsulated benzoyl peroxide 3% and encapsulated tretinoin 0.1% cream (Twyneo), which was approved by the Food and Drug Administration in July 2021 for the treatment of acne in adults and children aged 9 years and older. “Typically, benzoyl peroxide and tretinoin cannot be mixed in the same tube to stability issues,” she said. “Here, each product is individually encapsulated in a silica shell so that they can be applied together.”
The approval was supported by positive results from two phase 3, randomized, double-blind, vehicle-controlled, multicenter studies (NCT03761784 and NCT03761810), in which Twyneo demonstrated efficacy and a favorable tolerability profile in patients aged 9 years and older with facial acne.
Another topical treatment option, dapsone, is now FDA approved for ages 9 and up, expanded from its initial indication for ages 12 and up. The new indication is based on a phase 4, multicenter, open-label study in which acne patients aged 9-11 years applied dapsone 7.5% gel once daily to the face and acne-affected areas on the upper chest, upper back, and shoulders for 12 weeks. After 12 weeks, facial acne was clear or almost clear in about 47% of patients. “Inflammatory, noninflammatory, and total lesions decreased from baseline, but there was a greater reduction in noninflammatory lesions, so if you have a very young patient with acne, now you can consider dapsone gel,” Dr. Graber said.
In August 2020, clascoterone cream became the first topical androgen receptor inhibitor approved for the treatment of acne in patients 12 years of age and older. It is a drug believed to address sebum and inflammation directly in the sebaceous gland and is structurally similar to dihydrotestosterone and spironolactone.
“This is a completely new drug category in acne,” she said. “Unlike all oral antiandrogen therapies, clascoterone cream can be used in both males and females with acne. It’s the first acne drug to have a new mechanism of action in almost 40 years, since isotretinoin was approved in 1982.”
In vitro, she continued, clascoterone competes with dihydrotestosterone for binding to the androgen receptor, inhibiting downstream signaling and leading to inhibited sebum production, reduced secretion of inflammatory cytokines, and inhibition of inflammatory pathways. Two phase 3 studies that led to its approval involved 1,440 patients with moderate to severe facial acne aged 9-58 years. The cream was applied twice a day for 12 weeks and treatment adherence was approximately 90%. The researchers found that clascoterone cream was significantly more effective than vehicle cream at achieving Investigator’s Global Assessment scores of 0 (clear) or 1 (almost clear), the definition of treatment success in the study, and reducing noninflammatory lesion and inflammatory lesion counts at week 12. “There were no safety issues noted during these studies, and clascoterone cream was well tolerated,” Dr. Graber said.
Dr. Graber disclosed that she is a consultant/adviser for Digital Diagnostics, Almirall, Hovione, Keratin Biosciences, La Roche Posay, Ortho Dermatologics, Sebacia, Sol-Gel, Verrica, and WebMD. She is also a research investigator for Hovione, Ortho Dermatologics, Sebacia, and she receives royalties from Wolters Kluwer Health.
MedscapeLive and this news organization are owned by the same parent company.
Commentary by Lawrence W. Eichenfield, MD
Acne vulgaris remains an issue of tremendous importance to preteens, teens, and young adults, with approximately 85% of individuals aged 12-24 being affected. Expanding options for topical treatments may help bring effective disease control. Dr. Graber pointed out that historically, pediatricians and other primary care practitioners utilize topical retinoids less often for acne care as compared with dermatologists or guidelines recommendations (either the AAP’s or AAD’s). There are now expanded options, including over-the-counter retinoids (adapalene 0.1% gel), generic and trade brand topical tretinoin products, prescription adapalene medications, older and recently approved tazarotene products, and a newer type of topical retinoid, trifarotene. Novel formulations and emulsion technology, as well as retinoid developed in combination products, give more options in patients down to 9 years of age. A novel topical anti-androgen, clascoterone, is in its own category, as the first topical “hormonal agent,” allowing hormonal therapy to be used for males as well as females (aged 12 years and up). A recent review in JAMA (2021 Nov 23;326[20]:2055-67) incorporates many of these newer medications into management suggestions, emphasizing that first-line therapies are topical retinoids, benzoyl peroxide, azelaic acid, or combinations of topicals, whereas in more severe disease, oral antibiotics such as doxycycline or minocycline, hormonal therapies such as combination oral conceptive agents or spironolactone, or isotretinoin are most effective.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
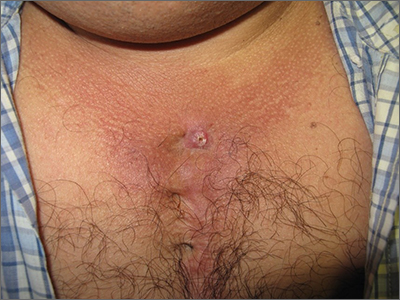
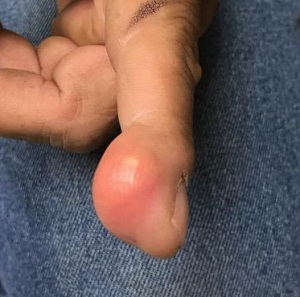
New wound over an old scar
While a squamous cell carcinoma presenting this way is perhaps more common, this nonhealing draining papule over a sternal scar was actually a sternocutaneous fistula (SCF). Examination revealed multiple bound down pits along the sternotomy scar. Very gentle probing of the papule in consideration of biopsy revealed a wire foreign body—the end of a sternotomy wire. A culture of yellow discharge ultimately grew Staphylococcus aureus.
SCF is a rare, and sometimes devastating, complication of cardiac surgery that occurred in 0.23% of cases at 1-year in a single center study of 12,297 patients over 9 years.1 As in this case, it may also present distantly from the time of surgery. The risk of SCF increases with smoking, previous sternal wound infection, renal failure, and use of bone wax during surgery.1
As soon as there was concern for SCF as a possible diagnosis, the patient was referred to, and quickly evaluated by, Cardiothoracic Surgery. Ultrasound and computed tomography imaging did not reveal any osteomyelitis or deep mediastinal disease. He was treated with debridement and removal of the sternotomy wire. At the 1-year follow-up, he had no further episodes of skin infection in the area.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Steingrímsson S, Gustafsson R, Gudbjartsson T, et al. Sternocutaneous fistulas after cardiac surgery: incidence and late outcome during a ten-year follow-up. Ann Thorac Surg. 2009;88:1910-1915. doi: 10.1016/j.athoracsur.2009.07.012
While a squamous cell carcinoma presenting this way is perhaps more common, this nonhealing draining papule over a sternal scar was actually a sternocutaneous fistula (SCF). Examination revealed multiple bound down pits along the sternotomy scar. Very gentle probing of the papule in consideration of biopsy revealed a wire foreign body—the end of a sternotomy wire. A culture of yellow discharge ultimately grew Staphylococcus aureus.
SCF is a rare, and sometimes devastating, complication of cardiac surgery that occurred in 0.23% of cases at 1-year in a single center study of 12,297 patients over 9 years.1 As in this case, it may also present distantly from the time of surgery. The risk of SCF increases with smoking, previous sternal wound infection, renal failure, and use of bone wax during surgery.1
As soon as there was concern for SCF as a possible diagnosis, the patient was referred to, and quickly evaluated by, Cardiothoracic Surgery. Ultrasound and computed tomography imaging did not reveal any osteomyelitis or deep mediastinal disease. He was treated with debridement and removal of the sternotomy wire. At the 1-year follow-up, he had no further episodes of skin infection in the area.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
While a squamous cell carcinoma presenting this way is perhaps more common, this nonhealing draining papule over a sternal scar was actually a sternocutaneous fistula (SCF). Examination revealed multiple bound down pits along the sternotomy scar. Very gentle probing of the papule in consideration of biopsy revealed a wire foreign body—the end of a sternotomy wire. A culture of yellow discharge ultimately grew Staphylococcus aureus.
SCF is a rare, and sometimes devastating, complication of cardiac surgery that occurred in 0.23% of cases at 1-year in a single center study of 12,297 patients over 9 years.1 As in this case, it may also present distantly from the time of surgery. The risk of SCF increases with smoking, previous sternal wound infection, renal failure, and use of bone wax during surgery.1
As soon as there was concern for SCF as a possible diagnosis, the patient was referred to, and quickly evaluated by, Cardiothoracic Surgery. Ultrasound and computed tomography imaging did not reveal any osteomyelitis or deep mediastinal disease. He was treated with debridement and removal of the sternotomy wire. At the 1-year follow-up, he had no further episodes of skin infection in the area.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Steingrímsson S, Gustafsson R, Gudbjartsson T, et al. Sternocutaneous fistulas after cardiac surgery: incidence and late outcome during a ten-year follow-up. Ann Thorac Surg. 2009;88:1910-1915. doi: 10.1016/j.athoracsur.2009.07.012
1. Steingrímsson S, Gustafsson R, Gudbjartsson T, et al. Sternocutaneous fistulas after cardiac surgery: incidence and late outcome during a ten-year follow-up. Ann Thorac Surg. 2009;88:1910-1915. doi: 10.1016/j.athoracsur.2009.07.012
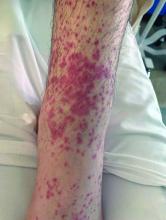
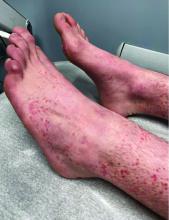
A 73-year-old White male presented with 2 days of a very pruritic rash
Reactions can occur anytime from within the first 2 weeks of treatment up to 10 days after the treatment has been discontinued. If a drug is rechallenged, eruptions may occur sooner. Pruritus is commonly seen. Clinically, erythematous papules and macules present symmetrically on the trunk and upper extremities and then become more generalized. A low-grade fever may be present.
Antibiotics are the most common causes of exanthematous drug eruptions. Penicillins and trimethoprim-sulfamethoxazole are common offenders. Cephalosporins, anticonvulsants, and allopurinol may also induce a reaction. As this condition is diagnosed clinically, skin biopsy is often not necessary. Histology is nonspecific and shows a mild perivascular lymphocytic infiltrate and few epidermal necrotic keratinocytes.
In drug reaction with eosinophilia and systemic symptoms (DRESS), symptoms present 2-6 weeks after the offending medication has been started. The cutaneous rash appears similar to an exanthematous drug eruption; however, lesions will also present on the face, and facial edema may occur. Fever is often present. Laboratory findings include a marked peripheral blood hypereosinophilia. Elevated liver function tests may be seen. Viruses such as Epstein-Barr virus, enteroviruses, adenovirus, early HIV, human herpesvirus 6, and parvovirus B19 have a similar clinical appearance to an exanthematous drug eruption. A mild eosinophilia, as seen in a drug eruption, helps to distinguish between a drug eruption and viral exanthem. In Stevens-Johnson Syndrome, mucosal membranes are involved and skin is often painful or appears dusky.
Treatment of exanthematous drug eruptions is largely supportive. Discontinuing the drug will help speed resolution and topical steroids may alleviate pruritus.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Bolognia J et al. “Dermatology” (St. Louis: Mosby/Elsevier, 2008).
2. James W et al. “Andrews’ Diseases of the Skin,” 13th ed. (Philadelphia: Saunders Elsevier, 2006).
Reactions can occur anytime from within the first 2 weeks of treatment up to 10 days after the treatment has been discontinued. If a drug is rechallenged, eruptions may occur sooner. Pruritus is commonly seen. Clinically, erythematous papules and macules present symmetrically on the trunk and upper extremities and then become more generalized. A low-grade fever may be present.
Antibiotics are the most common causes of exanthematous drug eruptions. Penicillins and trimethoprim-sulfamethoxazole are common offenders. Cephalosporins, anticonvulsants, and allopurinol may also induce a reaction. As this condition is diagnosed clinically, skin biopsy is often not necessary. Histology is nonspecific and shows a mild perivascular lymphocytic infiltrate and few epidermal necrotic keratinocytes.
In drug reaction with eosinophilia and systemic symptoms (DRESS), symptoms present 2-6 weeks after the offending medication has been started. The cutaneous rash appears similar to an exanthematous drug eruption; however, lesions will also present on the face, and facial edema may occur. Fever is often present. Laboratory findings include a marked peripheral blood hypereosinophilia. Elevated liver function tests may be seen. Viruses such as Epstein-Barr virus, enteroviruses, adenovirus, early HIV, human herpesvirus 6, and parvovirus B19 have a similar clinical appearance to an exanthematous drug eruption. A mild eosinophilia, as seen in a drug eruption, helps to distinguish between a drug eruption and viral exanthem. In Stevens-Johnson Syndrome, mucosal membranes are involved and skin is often painful or appears dusky.
Treatment of exanthematous drug eruptions is largely supportive. Discontinuing the drug will help speed resolution and topical steroids may alleviate pruritus.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Bolognia J et al. “Dermatology” (St. Louis: Mosby/Elsevier, 2008).
2. James W et al. “Andrews’ Diseases of the Skin,” 13th ed. (Philadelphia: Saunders Elsevier, 2006).
Reactions can occur anytime from within the first 2 weeks of treatment up to 10 days after the treatment has been discontinued. If a drug is rechallenged, eruptions may occur sooner. Pruritus is commonly seen. Clinically, erythematous papules and macules present symmetrically on the trunk and upper extremities and then become more generalized. A low-grade fever may be present.
Antibiotics are the most common causes of exanthematous drug eruptions. Penicillins and trimethoprim-sulfamethoxazole are common offenders. Cephalosporins, anticonvulsants, and allopurinol may also induce a reaction. As this condition is diagnosed clinically, skin biopsy is often not necessary. Histology is nonspecific and shows a mild perivascular lymphocytic infiltrate and few epidermal necrotic keratinocytes.
In drug reaction with eosinophilia and systemic symptoms (DRESS), symptoms present 2-6 weeks after the offending medication has been started. The cutaneous rash appears similar to an exanthematous drug eruption; however, lesions will also present on the face, and facial edema may occur. Fever is often present. Laboratory findings include a marked peripheral blood hypereosinophilia. Elevated liver function tests may be seen. Viruses such as Epstein-Barr virus, enteroviruses, adenovirus, early HIV, human herpesvirus 6, and parvovirus B19 have a similar clinical appearance to an exanthematous drug eruption. A mild eosinophilia, as seen in a drug eruption, helps to distinguish between a drug eruption and viral exanthem. In Stevens-Johnson Syndrome, mucosal membranes are involved and skin is often painful or appears dusky.
Treatment of exanthematous drug eruptions is largely supportive. Discontinuing the drug will help speed resolution and topical steroids may alleviate pruritus.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Bolognia J et al. “Dermatology” (St. Louis: Mosby/Elsevier, 2008).
2. James W et al. “Andrews’ Diseases of the Skin,” 13th ed. (Philadelphia: Saunders Elsevier, 2006).
Seborrheic dermatitis
THE COMPARISON
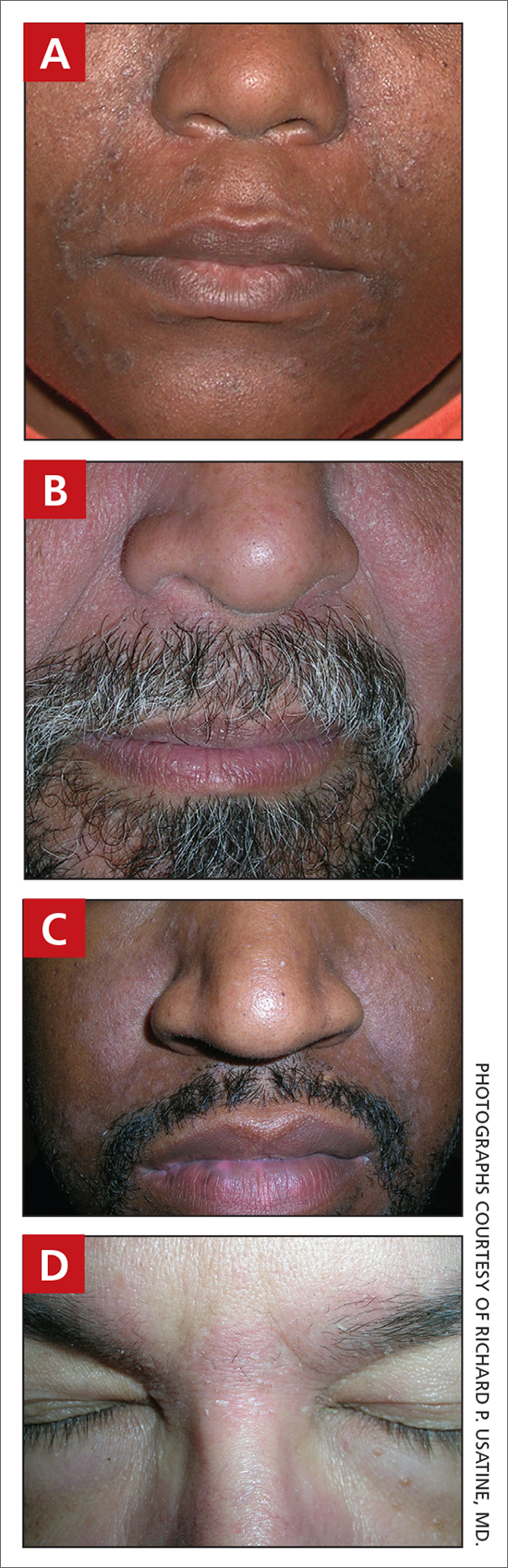
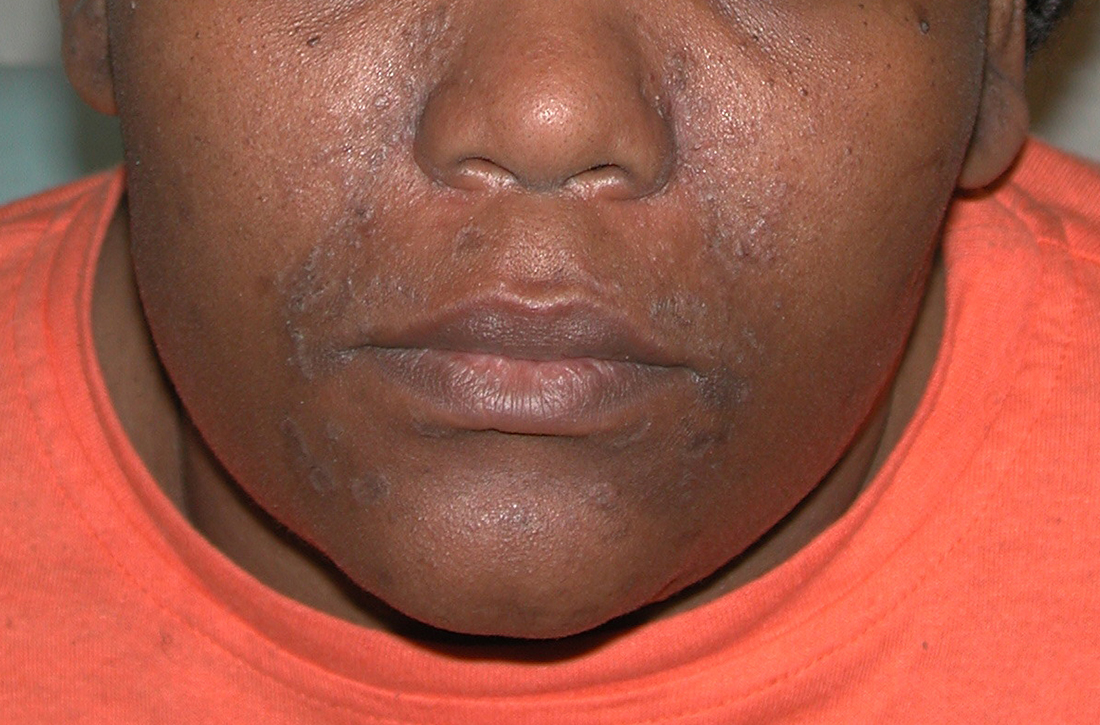
A Seborrheic dermatitis in a woman with brown-gray greasy scale, as well as petaloid papules and plaques that are especially prominent in the nasolabial folds.
B Seborrheic dermatitis in a man with erythema, scale, and mild postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
C Seborrheic dermatitis in a man with erythema, faint scale, and postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
D Seborrheic dermatitis in a man with erythema and scale of the eyebrows and glabellar region.
Seborrheic dermatitis (SD) is an inflammatory condition that is thought to be part of a response to Malassezia yeast. The scalp and face are most commonly affected, particularly the nasolabial folds, eyebrows, ears, postauricular areas, and beard area. Men also may have SD on the mid upper chest in association with chest hair. In infants, the scalp and body skin folds often are affected.
Epidemiology
SD affects patients of all ages: infants, adolescents, and adults. It is among the most common dermatologic diagnoses reported in Black patients in the United States.1
Key clinical features in darker skin tones
- In those with darker skin tones, arcuate, polycyclic, or petaloid (flower petallike) plaques may be present (FIGURE A). Also, hypopigmented patches and plaques may be prominent (FIGURES B AND C). The classic description includes thin pink patches and plaques with white greasy scale on the face (FIGURE D).
- The scalp may have diffuse scale or isolated scaly plaques.
Worth noting
- In those with tightly coiled hair, there is a predisposition for dry hair and increased risk for breakage.
- Treatment plans for patients with SD often include frequent hair washing. However, in those with tightly coiled hair, the treatment plan may need to be modified due to hair texture, tendency for dryness, and washing frequency preferences. Washing the scalp at least every 1 to 2 weeks may be a preferred approach for those with tightly coiled hair at increased risk for dryness/breakage vs washing daily.2 In a sample of 201 caregivers of Black girls, Rucker Wright et al3 found that washing the hair more than once per week was not correlated with a lower prevalence of SD.
- If tightly coiled hair is temporarily straightened with heat (eg, blow-dryer, flat iron), adding a liquid-based treatment such as clobetasol solution or fluocinonide solution will cause the hair to revert to its normal curl pattern.
- It is appropriate to ask patients for their vehicle preference for medications.2 For example, if clobetasol is the treatment selected for the patient, the vehicle can reflect patient preference for a liquid, foam, cream, or ointment.
- Some antifungal/antiyeast shampoos may cause further hair dryness and breakage.
- Treatment may be delayed because patients often use various topical pomades and ointments to cover up the scale and help with pruritus.
- Diffuse scale of tinea capitis in school- aged children can be mistaken for SD, which leads to delayed diagnosis and treatment.
- Clinicians should become comfortable with scalp examinations in patients with tightly coiled hair. Patients with chief concerns related to their hair and scalp expect their clinicians to touch these areas. Avoid leaning in to examine the patient without touching the patient’s hair and scalp.2,4
Health disparity highlight
SD is among the most common cutaneous disorders diagnosed in patients with skin of color.1,5 Delay in recognition of SD in those with darker skin tones leads to delayed treatment. SD of the face can cause notable postinflammatory pigmentation alteration. Pigmentation changes in the skin further impact quality of life.
1. Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
2. Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. 2021;10.1111/ pde.14525
3. Rucker Wright D, Gathers R, Kapke A, et al. Hair care practices and their association with scalp and hair disorders in African American girls. J Am Acad Dermatol. 2011;64: 253-262. doi:10.1016/j.jaad.2010.05.037
4. Grayson C, Heath C. An approach to examining tightly coiled hair among patients with hair loss in race-discordant patientphysician interactions. JAMA Dermatol. 2021;157:505-506. doi:10.1001/jamadermatol.2021.0338
5. Gaulding JV, Gutierrez D, Bhatia BK, et al. Epidemiology of skin diseases in a diverse patient population. J Drugs Dermatol. 2018;17:1032-1036.
THE COMPARISON
A Seborrheic dermatitis in a woman with brown-gray greasy scale, as well as petaloid papules and plaques that are especially prominent in the nasolabial folds.
B Seborrheic dermatitis in a man with erythema, scale, and mild postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
C Seborrheic dermatitis in a man with erythema, faint scale, and postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
D Seborrheic dermatitis in a man with erythema and scale of the eyebrows and glabellar region.
Seborrheic dermatitis (SD) is an inflammatory condition that is thought to be part of a response to Malassezia yeast. The scalp and face are most commonly affected, particularly the nasolabial folds, eyebrows, ears, postauricular areas, and beard area. Men also may have SD on the mid upper chest in association with chest hair. In infants, the scalp and body skin folds often are affected.
Epidemiology
SD affects patients of all ages: infants, adolescents, and adults. It is among the most common dermatologic diagnoses reported in Black patients in the United States.1
Key clinical features in darker skin tones
- In those with darker skin tones, arcuate, polycyclic, or petaloid (flower petallike) plaques may be present (FIGURE A). Also, hypopigmented patches and plaques may be prominent (FIGURES B AND C). The classic description includes thin pink patches and plaques with white greasy scale on the face (FIGURE D).
- The scalp may have diffuse scale or isolated scaly plaques.
Worth noting
- In those with tightly coiled hair, there is a predisposition for dry hair and increased risk for breakage.
- Treatment plans for patients with SD often include frequent hair washing. However, in those with tightly coiled hair, the treatment plan may need to be modified due to hair texture, tendency for dryness, and washing frequency preferences. Washing the scalp at least every 1 to 2 weeks may be a preferred approach for those with tightly coiled hair at increased risk for dryness/breakage vs washing daily.2 In a sample of 201 caregivers of Black girls, Rucker Wright et al3 found that washing the hair more than once per week was not correlated with a lower prevalence of SD.
- If tightly coiled hair is temporarily straightened with heat (eg, blow-dryer, flat iron), adding a liquid-based treatment such as clobetasol solution or fluocinonide solution will cause the hair to revert to its normal curl pattern.
- It is appropriate to ask patients for their vehicle preference for medications.2 For example, if clobetasol is the treatment selected for the patient, the vehicle can reflect patient preference for a liquid, foam, cream, or ointment.
- Some antifungal/antiyeast shampoos may cause further hair dryness and breakage.
- Treatment may be delayed because patients often use various topical pomades and ointments to cover up the scale and help with pruritus.
- Diffuse scale of tinea capitis in school- aged children can be mistaken for SD, which leads to delayed diagnosis and treatment.
- Clinicians should become comfortable with scalp examinations in patients with tightly coiled hair. Patients with chief concerns related to their hair and scalp expect their clinicians to touch these areas. Avoid leaning in to examine the patient without touching the patient’s hair and scalp.2,4
Health disparity highlight
SD is among the most common cutaneous disorders diagnosed in patients with skin of color.1,5 Delay in recognition of SD in those with darker skin tones leads to delayed treatment. SD of the face can cause notable postinflammatory pigmentation alteration. Pigmentation changes in the skin further impact quality of life.
THE COMPARISON
A Seborrheic dermatitis in a woman with brown-gray greasy scale, as well as petaloid papules and plaques that are especially prominent in the nasolabial folds.
B Seborrheic dermatitis in a man with erythema, scale, and mild postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
C Seborrheic dermatitis in a man with erythema, faint scale, and postinflammatory hypopigmentation that are especially prominent in the nasolabial folds.
D Seborrheic dermatitis in a man with erythema and scale of the eyebrows and glabellar region.
Seborrheic dermatitis (SD) is an inflammatory condition that is thought to be part of a response to Malassezia yeast. The scalp and face are most commonly affected, particularly the nasolabial folds, eyebrows, ears, postauricular areas, and beard area. Men also may have SD on the mid upper chest in association with chest hair. In infants, the scalp and body skin folds often are affected.
Epidemiology
SD affects patients of all ages: infants, adolescents, and adults. It is among the most common dermatologic diagnoses reported in Black patients in the United States.1
Key clinical features in darker skin tones
- In those with darker skin tones, arcuate, polycyclic, or petaloid (flower petallike) plaques may be present (FIGURE A). Also, hypopigmented patches and plaques may be prominent (FIGURES B AND C). The classic description includes thin pink patches and plaques with white greasy scale on the face (FIGURE D).
- The scalp may have diffuse scale or isolated scaly plaques.
Worth noting
- In those with tightly coiled hair, there is a predisposition for dry hair and increased risk for breakage.
- Treatment plans for patients with SD often include frequent hair washing. However, in those with tightly coiled hair, the treatment plan may need to be modified due to hair texture, tendency for dryness, and washing frequency preferences. Washing the scalp at least every 1 to 2 weeks may be a preferred approach for those with tightly coiled hair at increased risk for dryness/breakage vs washing daily.2 In a sample of 201 caregivers of Black girls, Rucker Wright et al3 found that washing the hair more than once per week was not correlated with a lower prevalence of SD.
- If tightly coiled hair is temporarily straightened with heat (eg, blow-dryer, flat iron), adding a liquid-based treatment such as clobetasol solution or fluocinonide solution will cause the hair to revert to its normal curl pattern.
- It is appropriate to ask patients for their vehicle preference for medications.2 For example, if clobetasol is the treatment selected for the patient, the vehicle can reflect patient preference for a liquid, foam, cream, or ointment.
- Some antifungal/antiyeast shampoos may cause further hair dryness and breakage.
- Treatment may be delayed because patients often use various topical pomades and ointments to cover up the scale and help with pruritus.
- Diffuse scale of tinea capitis in school- aged children can be mistaken for SD, which leads to delayed diagnosis and treatment.
- Clinicians should become comfortable with scalp examinations in patients with tightly coiled hair. Patients with chief concerns related to their hair and scalp expect their clinicians to touch these areas. Avoid leaning in to examine the patient without touching the patient’s hair and scalp.2,4
Health disparity highlight
SD is among the most common cutaneous disorders diagnosed in patients with skin of color.1,5 Delay in recognition of SD in those with darker skin tones leads to delayed treatment. SD of the face can cause notable postinflammatory pigmentation alteration. Pigmentation changes in the skin further impact quality of life.
1. Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
2. Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. 2021;10.1111/ pde.14525
3. Rucker Wright D, Gathers R, Kapke A, et al. Hair care practices and their association with scalp and hair disorders in African American girls. J Am Acad Dermatol. 2011;64: 253-262. doi:10.1016/j.jaad.2010.05.037
4. Grayson C, Heath C. An approach to examining tightly coiled hair among patients with hair loss in race-discordant patientphysician interactions. JAMA Dermatol. 2021;157:505-506. doi:10.1001/jamadermatol.2021.0338
5. Gaulding JV, Gutierrez D, Bhatia BK, et al. Epidemiology of skin diseases in a diverse patient population. J Drugs Dermatol. 2018;17:1032-1036.
1. Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
2. Grayson C, Heath C. Tips for addressing common conditions affecting pediatric and adolescent patients with skin of color [published online March 2, 2021]. Pediatr Dermatol. 2021;10.1111/ pde.14525
3. Rucker Wright D, Gathers R, Kapke A, et al. Hair care practices and their association with scalp and hair disorders in African American girls. J Am Acad Dermatol. 2011;64: 253-262. doi:10.1016/j.jaad.2010.05.037
4. Grayson C, Heath C. An approach to examining tightly coiled hair among patients with hair loss in race-discordant patientphysician interactions. JAMA Dermatol. 2021;157:505-506. doi:10.1001/jamadermatol.2021.0338
5. Gaulding JV, Gutierrez D, Bhatia BK, et al. Epidemiology of skin diseases in a diverse patient population. J Drugs Dermatol. 2018;17:1032-1036.
AI: Skin of color underrepresented in datasets used to identify skin cancer
An in the databases, researchers in the United Kingdom report.
Out of 106,950 skin lesions documented in 21 open-access databases and 17 open-access atlases identified by David Wen, BMBCh, from the University of Oxford (England), and colleagues, 2,436 images contained information on Fitzpatrick skin type. Of these, “only 10 images were from individuals with Fitzpatrick skin type V, and only a single image was from an individual with Fitzpatrick skin type VI,” the researchers said. “The ethnicity of these individuals was either Brazilian or unknown.”
In two datasets containing 1,585 images with ethnicity data, “no images were from individuals with an African, Afro-Caribbean, or South Asian background,” Dr. Wen and colleagues noted. “Coupled with the geographical origins of datasets, there was massive under-representation of skin lesion images from darker-skinned populations.”
The results of their systematic review were presented at the National Cancer Research Institute Festival and published on Nov. 9, 2021, in The Lancet Digital Health. To the best of their knowledge, they wrote, this is “the first systematic review of publicly available skin lesion images comprising predominantly dermoscopic and macroscopic images available through open access datasets and atlases.”
Overall, 11 of 14 datasets (79%) were from North America, Europe, or Oceania among datasets with information on country of origin, the researchers said. Either dermoscopic images or macroscopic photographs were the only types of images available in 19 of 21 (91%) datasets. There was some variation in the clinical information available, with 81,662 images (76.4%) containing information on age, 82,848 images (77.5%) having information on gender, and 79,561 images having information about body site (74.4%).
The researchers explained that these datasets might be of limited use in a real-world setting where the images aren’t representative of the population. Artificial intelligence (AI) programs that train using images of patients with one skin type, for example, can potentially misdiagnose patients of another skin type, they said.
“AI programs hold a lot of potential for diagnosing skin cancer because it can look at pictures and quickly and cost-effectively evaluate any worrying spots on the skin,” Dr. Wen said in a press release from the NCRI Festival. “However, it’s important to know about the images and patients used to develop programs, as these influence which groups of people the programs will be most effective for in real-life settings. Research has shown that programs trained on images taken from people with lighter skin types only might not be as accurate for people with darker skin, and vice versa.”
There was also “limited information on who, how and why the images were taken,” Dr. Wen said in the release. “This has implications for the programs developed from these images, due to uncertainty around how they may perform in different groups of people, especially in those who aren’t well represented in datasets, such as those with darker skin. This can potentially lead to the exclusion or even harm of these groups from AI technologies.”
While there are no current guidelines for developing skin image datasets, quality standards are needed, according to the researchers.
“Ensuring equitable digital health includes building unbiased, representative datasets to ensure that the algorithms that are created benefit people of all backgrounds and skin types,” they concluded in the study.
Neil Steven, MBBS, MA, PhD, FRCP, an NCRI Skin Group member who was not involved with the research, stated in the press release that the results from the study by Dr. Wen and colleagues “raise concerns about the ability of AI to assist in skin cancer diagnosis, especially in a global context.”
“I hope this work will continue and help ensure that the progress we make in using AI in medicine will benefit all patients, recognizing that human skin color is highly diverse,” said Dr. Steven, honorary consultant in medical oncology at University Hospitals Birmingham (England) NHS Foundation Trust.
‘We need more images of everybody’
Dermatologist Adewole Adamson, MD, MPP, assistant professor in the department of internal medicine (division of dermatology) at the University of Texas at Austin, said in an interview that a “major potential downside” of algorithms not trained on diverse datasets is the potential for incorrect diagnoses.
“The harms of algorithms used for diagnostic purposes in the skin can be particularly significant because of the scalability of this technology. A lot of thought needs to be put into how these algorithms are developed and tested,” said Dr. Adamson, who reviewed the manuscript of The Lancet Digital Health study but was not involved with the research.
He referred to the results of a recently published study in JAMA Dermatology, which found that only 10% of studies used to develop or test deep-learning algorithms contained metadata on skin tone. “Furthermore, most datasets are from countries where darker skin types are not represented. [These] algorithms therefore likely underperform on people of darker skin types and thus, users should be wary,” Dr. Adamson said.
A consensus guideline should be developed for public AI algorithms, he said, which should have metadata containing information on sex, race/ethnicity, geographic location, skin type, and part of the body. “This distribution should also be reported in any publication of an algorithm so that users can see if the distribution of the population in the training data mirrors that of the population in which it is intended to be used,” he added.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the research, said that, while this issue of underrepresentation has been known in dermatology for some time, the strength of the Lancet study is that it is a large study, with a message of “we need more images of everybody.”
“This is probably the broadest study looking at every possible accessible resource and taking an organized approach,” Dr. Friedman said in an interview. “But I think it also raises some important points about how we think about skin tones and how we refer to them as well with respect to misusing classification schemes that we currently have.”
While using ethnicity data and certain Fitzpatrick skin types as a proxy for darker skin is a limitation of the metadata the study authors had available, it also highlights “a broader problem with respect to lexicon regarding skin tone,” he explained.
“Skin does not have a race, it doesn’t have an ethnicity,” Dr. Friedman said.
A dataset that contains not only different skin tones but how different dermatologic conditions look across skin tones is important. “If you just look at one photo of one skin tone, you missed the fact that clinical presentations can be so polymorphic, especially because of different skin tones,” Dr. Friedman said.
“We need to keep pushing this message to ensure that images keep getting collected. We [need to] ensure that there’s quality control with these images and that we’re disseminating them in a way that everyone has access, both from self-learning, but also to teach others,” said Dr. Friedman, coeditor of a recently introduced dermatology atlas showing skin conditions in different skin tones.
Adamson reports no relevant financial relationships. Dr. Friedman is a coeditor of a dermatology atlas supported by Allergan Aesthetics and SkinBetter Science. This study was funded by NHSX and the Health Foundation. Three authors reported being paid employees of Databiology at the time of the study. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An in the databases, researchers in the United Kingdom report.
Out of 106,950 skin lesions documented in 21 open-access databases and 17 open-access atlases identified by David Wen, BMBCh, from the University of Oxford (England), and colleagues, 2,436 images contained information on Fitzpatrick skin type. Of these, “only 10 images were from individuals with Fitzpatrick skin type V, and only a single image was from an individual with Fitzpatrick skin type VI,” the researchers said. “The ethnicity of these individuals was either Brazilian or unknown.”
In two datasets containing 1,585 images with ethnicity data, “no images were from individuals with an African, Afro-Caribbean, or South Asian background,” Dr. Wen and colleagues noted. “Coupled with the geographical origins of datasets, there was massive under-representation of skin lesion images from darker-skinned populations.”
The results of their systematic review were presented at the National Cancer Research Institute Festival and published on Nov. 9, 2021, in The Lancet Digital Health. To the best of their knowledge, they wrote, this is “the first systematic review of publicly available skin lesion images comprising predominantly dermoscopic and macroscopic images available through open access datasets and atlases.”
Overall, 11 of 14 datasets (79%) were from North America, Europe, or Oceania among datasets with information on country of origin, the researchers said. Either dermoscopic images or macroscopic photographs were the only types of images available in 19 of 21 (91%) datasets. There was some variation in the clinical information available, with 81,662 images (76.4%) containing information on age, 82,848 images (77.5%) having information on gender, and 79,561 images having information about body site (74.4%).
The researchers explained that these datasets might be of limited use in a real-world setting where the images aren’t representative of the population. Artificial intelligence (AI) programs that train using images of patients with one skin type, for example, can potentially misdiagnose patients of another skin type, they said.
“AI programs hold a lot of potential for diagnosing skin cancer because it can look at pictures and quickly and cost-effectively evaluate any worrying spots on the skin,” Dr. Wen said in a press release from the NCRI Festival. “However, it’s important to know about the images and patients used to develop programs, as these influence which groups of people the programs will be most effective for in real-life settings. Research has shown that programs trained on images taken from people with lighter skin types only might not be as accurate for people with darker skin, and vice versa.”
There was also “limited information on who, how and why the images were taken,” Dr. Wen said in the release. “This has implications for the programs developed from these images, due to uncertainty around how they may perform in different groups of people, especially in those who aren’t well represented in datasets, such as those with darker skin. This can potentially lead to the exclusion or even harm of these groups from AI technologies.”
While there are no current guidelines for developing skin image datasets, quality standards are needed, according to the researchers.
“Ensuring equitable digital health includes building unbiased, representative datasets to ensure that the algorithms that are created benefit people of all backgrounds and skin types,” they concluded in the study.
Neil Steven, MBBS, MA, PhD, FRCP, an NCRI Skin Group member who was not involved with the research, stated in the press release that the results from the study by Dr. Wen and colleagues “raise concerns about the ability of AI to assist in skin cancer diagnosis, especially in a global context.”
“I hope this work will continue and help ensure that the progress we make in using AI in medicine will benefit all patients, recognizing that human skin color is highly diverse,” said Dr. Steven, honorary consultant in medical oncology at University Hospitals Birmingham (England) NHS Foundation Trust.
‘We need more images of everybody’
Dermatologist Adewole Adamson, MD, MPP, assistant professor in the department of internal medicine (division of dermatology) at the University of Texas at Austin, said in an interview that a “major potential downside” of algorithms not trained on diverse datasets is the potential for incorrect diagnoses.
“The harms of algorithms used for diagnostic purposes in the skin can be particularly significant because of the scalability of this technology. A lot of thought needs to be put into how these algorithms are developed and tested,” said Dr. Adamson, who reviewed the manuscript of The Lancet Digital Health study but was not involved with the research.
He referred to the results of a recently published study in JAMA Dermatology, which found that only 10% of studies used to develop or test deep-learning algorithms contained metadata on skin tone. “Furthermore, most datasets are from countries where darker skin types are not represented. [These] algorithms therefore likely underperform on people of darker skin types and thus, users should be wary,” Dr. Adamson said.
A consensus guideline should be developed for public AI algorithms, he said, which should have metadata containing information on sex, race/ethnicity, geographic location, skin type, and part of the body. “This distribution should also be reported in any publication of an algorithm so that users can see if the distribution of the population in the training data mirrors that of the population in which it is intended to be used,” he added.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the research, said that, while this issue of underrepresentation has been known in dermatology for some time, the strength of the Lancet study is that it is a large study, with a message of “we need more images of everybody.”
“This is probably the broadest study looking at every possible accessible resource and taking an organized approach,” Dr. Friedman said in an interview. “But I think it also raises some important points about how we think about skin tones and how we refer to them as well with respect to misusing classification schemes that we currently have.”
While using ethnicity data and certain Fitzpatrick skin types as a proxy for darker skin is a limitation of the metadata the study authors had available, it also highlights “a broader problem with respect to lexicon regarding skin tone,” he explained.
“Skin does not have a race, it doesn’t have an ethnicity,” Dr. Friedman said.
A dataset that contains not only different skin tones but how different dermatologic conditions look across skin tones is important. “If you just look at one photo of one skin tone, you missed the fact that clinical presentations can be so polymorphic, especially because of different skin tones,” Dr. Friedman said.
“We need to keep pushing this message to ensure that images keep getting collected. We [need to] ensure that there’s quality control with these images and that we’re disseminating them in a way that everyone has access, both from self-learning, but also to teach others,” said Dr. Friedman, coeditor of a recently introduced dermatology atlas showing skin conditions in different skin tones.
Adamson reports no relevant financial relationships. Dr. Friedman is a coeditor of a dermatology atlas supported by Allergan Aesthetics and SkinBetter Science. This study was funded by NHSX and the Health Foundation. Three authors reported being paid employees of Databiology at the time of the study. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An in the databases, researchers in the United Kingdom report.
Out of 106,950 skin lesions documented in 21 open-access databases and 17 open-access atlases identified by David Wen, BMBCh, from the University of Oxford (England), and colleagues, 2,436 images contained information on Fitzpatrick skin type. Of these, “only 10 images were from individuals with Fitzpatrick skin type V, and only a single image was from an individual with Fitzpatrick skin type VI,” the researchers said. “The ethnicity of these individuals was either Brazilian or unknown.”
In two datasets containing 1,585 images with ethnicity data, “no images were from individuals with an African, Afro-Caribbean, or South Asian background,” Dr. Wen and colleagues noted. “Coupled with the geographical origins of datasets, there was massive under-representation of skin lesion images from darker-skinned populations.”
The results of their systematic review were presented at the National Cancer Research Institute Festival and published on Nov. 9, 2021, in The Lancet Digital Health. To the best of their knowledge, they wrote, this is “the first systematic review of publicly available skin lesion images comprising predominantly dermoscopic and macroscopic images available through open access datasets and atlases.”
Overall, 11 of 14 datasets (79%) were from North America, Europe, or Oceania among datasets with information on country of origin, the researchers said. Either dermoscopic images or macroscopic photographs were the only types of images available in 19 of 21 (91%) datasets. There was some variation in the clinical information available, with 81,662 images (76.4%) containing information on age, 82,848 images (77.5%) having information on gender, and 79,561 images having information about body site (74.4%).
The researchers explained that these datasets might be of limited use in a real-world setting where the images aren’t representative of the population. Artificial intelligence (AI) programs that train using images of patients with one skin type, for example, can potentially misdiagnose patients of another skin type, they said.
“AI programs hold a lot of potential for diagnosing skin cancer because it can look at pictures and quickly and cost-effectively evaluate any worrying spots on the skin,” Dr. Wen said in a press release from the NCRI Festival. “However, it’s important to know about the images and patients used to develop programs, as these influence which groups of people the programs will be most effective for in real-life settings. Research has shown that programs trained on images taken from people with lighter skin types only might not be as accurate for people with darker skin, and vice versa.”
There was also “limited information on who, how and why the images were taken,” Dr. Wen said in the release. “This has implications for the programs developed from these images, due to uncertainty around how they may perform in different groups of people, especially in those who aren’t well represented in datasets, such as those with darker skin. This can potentially lead to the exclusion or even harm of these groups from AI technologies.”
While there are no current guidelines for developing skin image datasets, quality standards are needed, according to the researchers.
“Ensuring equitable digital health includes building unbiased, representative datasets to ensure that the algorithms that are created benefit people of all backgrounds and skin types,” they concluded in the study.
Neil Steven, MBBS, MA, PhD, FRCP, an NCRI Skin Group member who was not involved with the research, stated in the press release that the results from the study by Dr. Wen and colleagues “raise concerns about the ability of AI to assist in skin cancer diagnosis, especially in a global context.”
“I hope this work will continue and help ensure that the progress we make in using AI in medicine will benefit all patients, recognizing that human skin color is highly diverse,” said Dr. Steven, honorary consultant in medical oncology at University Hospitals Birmingham (England) NHS Foundation Trust.
‘We need more images of everybody’
Dermatologist Adewole Adamson, MD, MPP, assistant professor in the department of internal medicine (division of dermatology) at the University of Texas at Austin, said in an interview that a “major potential downside” of algorithms not trained on diverse datasets is the potential for incorrect diagnoses.
“The harms of algorithms used for diagnostic purposes in the skin can be particularly significant because of the scalability of this technology. A lot of thought needs to be put into how these algorithms are developed and tested,” said Dr. Adamson, who reviewed the manuscript of The Lancet Digital Health study but was not involved with the research.
He referred to the results of a recently published study in JAMA Dermatology, which found that only 10% of studies used to develop or test deep-learning algorithms contained metadata on skin tone. “Furthermore, most datasets are from countries where darker skin types are not represented. [These] algorithms therefore likely underperform on people of darker skin types and thus, users should be wary,” Dr. Adamson said.
A consensus guideline should be developed for public AI algorithms, he said, which should have metadata containing information on sex, race/ethnicity, geographic location, skin type, and part of the body. “This distribution should also be reported in any publication of an algorithm so that users can see if the distribution of the population in the training data mirrors that of the population in which it is intended to be used,” he added.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the research, said that, while this issue of underrepresentation has been known in dermatology for some time, the strength of the Lancet study is that it is a large study, with a message of “we need more images of everybody.”
“This is probably the broadest study looking at every possible accessible resource and taking an organized approach,” Dr. Friedman said in an interview. “But I think it also raises some important points about how we think about skin tones and how we refer to them as well with respect to misusing classification schemes that we currently have.”
While using ethnicity data and certain Fitzpatrick skin types as a proxy for darker skin is a limitation of the metadata the study authors had available, it also highlights “a broader problem with respect to lexicon regarding skin tone,” he explained.
“Skin does not have a race, it doesn’t have an ethnicity,” Dr. Friedman said.
A dataset that contains not only different skin tones but how different dermatologic conditions look across skin tones is important. “If you just look at one photo of one skin tone, you missed the fact that clinical presentations can be so polymorphic, especially because of different skin tones,” Dr. Friedman said.
“We need to keep pushing this message to ensure that images keep getting collected. We [need to] ensure that there’s quality control with these images and that we’re disseminating them in a way that everyone has access, both from self-learning, but also to teach others,” said Dr. Friedman, coeditor of a recently introduced dermatology atlas showing skin conditions in different skin tones.
Adamson reports no relevant financial relationships. Dr. Friedman is a coeditor of a dermatology atlas supported by Allergan Aesthetics and SkinBetter Science. This study was funded by NHSX and the Health Foundation. Three authors reported being paid employees of Databiology at the time of the study. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding Insult to Injury
ANSWER
The correct answer is inclusion cyst (choice “c”).
DISCUSSION
Inclusion cysts are also called traumatic inclusion cysts or implantation cysts and are quite distinct from “sebaceous,” epidermal, or epidermoid cysts. An inclusion cyst results from traumatic implantation of surface adnexal structures (eg, sebaceous glands) that continue to function, eventuating in the formation of an organized sac whose wall is composed of stratified squamous epithelium with a granular layer, no significant atypia, and surrounding pasty lamellated acellular keratin.
Hands are the most commonly affected area, although the precipitating puncture wound doesn’t have to be as impressive as this patient’s was. Nails and sewing needles can produce the same result.
The patient’s lesion was removed, at which point its pasty contents (a diagnostic clue) were revealed, and the wound closed. Although the absence of redness or tenderness helped to rule out some items in the differential (eg, felon, abscess), and the lesion demonstrated clear cystic features, the specimen was sent for pathologic examination for confirmation, since cancer would also belong in the differential for such a lesion.
ANSWER
The correct answer is inclusion cyst (choice “c”).
DISCUSSION
Inclusion cysts are also called traumatic inclusion cysts or implantation cysts and are quite distinct from “sebaceous,” epidermal, or epidermoid cysts. An inclusion cyst results from traumatic implantation of surface adnexal structures (eg, sebaceous glands) that continue to function, eventuating in the formation of an organized sac whose wall is composed of stratified squamous epithelium with a granular layer, no significant atypia, and surrounding pasty lamellated acellular keratin.
Hands are the most commonly affected area, although the precipitating puncture wound doesn’t have to be as impressive as this patient’s was. Nails and sewing needles can produce the same result.
The patient’s lesion was removed, at which point its pasty contents (a diagnostic clue) were revealed, and the wound closed. Although the absence of redness or tenderness helped to rule out some items in the differential (eg, felon, abscess), and the lesion demonstrated clear cystic features, the specimen was sent for pathologic examination for confirmation, since cancer would also belong in the differential for such a lesion.
ANSWER
The correct answer is inclusion cyst (choice “c”).
DISCUSSION
Inclusion cysts are also called traumatic inclusion cysts or implantation cysts and are quite distinct from “sebaceous,” epidermal, or epidermoid cysts. An inclusion cyst results from traumatic implantation of surface adnexal structures (eg, sebaceous glands) that continue to function, eventuating in the formation of an organized sac whose wall is composed of stratified squamous epithelium with a granular layer, no significant atypia, and surrounding pasty lamellated acellular keratin.
Hands are the most commonly affected area, although the precipitating puncture wound doesn’t have to be as impressive as this patient’s was. Nails and sewing needles can produce the same result.
The patient’s lesion was removed, at which point its pasty contents (a diagnostic clue) were revealed, and the wound closed. Although the absence of redness or tenderness helped to rule out some items in the differential (eg, felon, abscess), and the lesion demonstrated clear cystic features, the specimen was sent for pathologic examination for confirmation, since cancer would also belong in the differential for such a lesion.
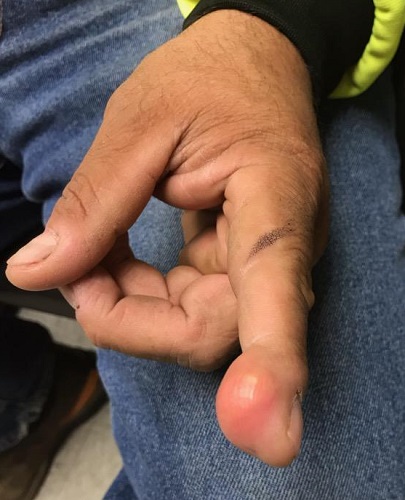
A 38-year-old man sustained an on-the-job injury to his finger, which was punctured by a tiny drill bit. He was promptly seen by a clinician, who administered a tetanus injection and prescribed a 2-week course of an antibiotic. However, within a few weeks, the patient noticed a lesion on the affected finger. It caused little to no pain but grew steadily, until it interfered with his functioning and prompted a return to the clinic.
The patient was in good health, despite being a long-term smoker. X-rays of the finger showed no bony changes or evidence of a foreign body. A referral to dermatology was provided.
Examination revealed a firm, painless, 2-cm subcutaneous mass in the pad of his left index finger. The surface felt quite tense, but there was no disruption nor any redness. Palpation of relevant nodal locations failed to reveal any palpable masses.
What is the diagnosis?
Numerous morphologies of skin rashes have been described in the setting of COVID-19, including pernio, livedoid rash, exanthem, and vasculitis. This classic constellation of symptoms (palpable purpura on buttocks/legs, abdominal pain, arthralgia, hematuria) is highly consistent with Henoch-Schonlein purpura (HSP). There are now multiple case reports of COVID-19–associated HSP.
HSP is the most common type of childhood systemic vasculitis. It is mediated by immunoglobulin A (IgA) immune complex deposition and has been associated with respiratory tract infections, streptococcal species, parainfluenza virus, and human parvovirus B19, medications, vaccinations, and malignancies. HSP is usually a self-limiting disease, with a course over 4-6 weeks, and can affect multiple organs, including the skin, gastrointestinal tract, joints, and the kidneys. The diagnostic criteria include palpable purpura in the presence of one or more of the following: diffuse abdominal pain, arthritis or arthralgia, any biopsy showing predominant IgA deposition, and renal involvement in the form of hematuria or proteinuria. Renal disease is variable and is the most significant indicator of long-term prognosis. This teenager was treated with oral corticosteroids because of the severe periarticular edema and responded rapidly. His subsequent urine analyses normalized.
What is on the differential?
Multisystem inflammatory syndrome in children (MIS-C) is a rare, potentially fatal, complication of COVID-19 infection that causes inflammation of multiple organs, including the heart, lungs, kidneys, brain, skin, eyes, or the gastrointestinal tract. It commonly affects children around ages 8-9 years. Initial symptoms include fever, rash, red eyes, diarrhea, and vomiting that appear 2-6 weeks post COVID-19 infection. Like HSP, MIS-C can present with edema of the extremities, worsening hand/foot pain, and hematuria; however, the absence of both fever and the pattern of system involvement seen with MIS-C and classic findings in this patient are more consistent with HSP.
Reactive infectious mucocutaneous eruption (RIME) was recently coined to encompass both infection-associated Stevens-Johnson eruptions including Mycoplasma pneumoniae-induced rash and mucositis (MIRM) and mucocutaneous eruptions caused by nonmycoplasma pathogens (including Chlamydia pneumoniae, human parainfluenza virus 2, rhinovirus, adenovirus, enterovirus, human metapneumovirus, influenza B virus, and COVID-19). It is usually seen in male children and adolescents. Prodromal symptoms include cough, fever, and malaise and they precede the prominent feature of mucositis. Our patient’s lack of mucosal involvement is not consistent with RIME.
Perniosis (chilblains) is characterized by localized edematous patches of erythema or cyanosis on exposed extremities, that may be associated with cold exposure. Lesions are usually symmetric and self-limiting, and symptoms can include numbness, tingling, pruritus, burning, or pain. Pernio-like skin lesions have been seen during the COVID-19 pandemic, though many patients have negative testing for infection by PCR and serology. Pernio may also be seen with autoimmune diseases or malignancy.
Meningococcemia is a rare disease caused by infection with gram-negative diplococci bacteria Neisseria meningitidis and spreads through saliva or respiratory secretions. Its clinical presentation can vary widely, from transient fever to fulminant disease. It is characterized by upper respiratory tract infection, fever, and petechial lesions associated with thrombocytopenia and coagulopathy.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Laborada is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Laborada have no relevant financial disclosures.
References
AlGhoozi DA, AlKhayyat HM. BMJ Case Reports CP 2021;14:e239910.
Jacobi M et al. Pediatr Infect Dis J. 2021;40(2):e93-4.
Paller A, Mancini AJ. Hurwitz clinical pediatric dermatology: A textbook of skin disorders of childhood and adolescence. 4th ed. Philadelphia (PA): Elsevier Saunders; 2011.
Radia T et al. Paediatr Respir Rev. 2021;38:51-7.
Ramien ML. Clin Exp Dermatol. 2021;46(3):420-9.
Numerous morphologies of skin rashes have been described in the setting of COVID-19, including pernio, livedoid rash, exanthem, and vasculitis. This classic constellation of symptoms (palpable purpura on buttocks/legs, abdominal pain, arthralgia, hematuria) is highly consistent with Henoch-Schonlein purpura (HSP). There are now multiple case reports of COVID-19–associated HSP.
HSP is the most common type of childhood systemic vasculitis. It is mediated by immunoglobulin A (IgA) immune complex deposition and has been associated with respiratory tract infections, streptococcal species, parainfluenza virus, and human parvovirus B19, medications, vaccinations, and malignancies. HSP is usually a self-limiting disease, with a course over 4-6 weeks, and can affect multiple organs, including the skin, gastrointestinal tract, joints, and the kidneys. The diagnostic criteria include palpable purpura in the presence of one or more of the following: diffuse abdominal pain, arthritis or arthralgia, any biopsy showing predominant IgA deposition, and renal involvement in the form of hematuria or proteinuria. Renal disease is variable and is the most significant indicator of long-term prognosis. This teenager was treated with oral corticosteroids because of the severe periarticular edema and responded rapidly. His subsequent urine analyses normalized.
What is on the differential?
Multisystem inflammatory syndrome in children (MIS-C) is a rare, potentially fatal, complication of COVID-19 infection that causes inflammation of multiple organs, including the heart, lungs, kidneys, brain, skin, eyes, or the gastrointestinal tract. It commonly affects children around ages 8-9 years. Initial symptoms include fever, rash, red eyes, diarrhea, and vomiting that appear 2-6 weeks post COVID-19 infection. Like HSP, MIS-C can present with edema of the extremities, worsening hand/foot pain, and hematuria; however, the absence of both fever and the pattern of system involvement seen with MIS-C and classic findings in this patient are more consistent with HSP.
Reactive infectious mucocutaneous eruption (RIME) was recently coined to encompass both infection-associated Stevens-Johnson eruptions including Mycoplasma pneumoniae-induced rash and mucositis (MIRM) and mucocutaneous eruptions caused by nonmycoplasma pathogens (including Chlamydia pneumoniae, human parainfluenza virus 2, rhinovirus, adenovirus, enterovirus, human metapneumovirus, influenza B virus, and COVID-19). It is usually seen in male children and adolescents. Prodromal symptoms include cough, fever, and malaise and they precede the prominent feature of mucositis. Our patient’s lack of mucosal involvement is not consistent with RIME.
Perniosis (chilblains) is characterized by localized edematous patches of erythema or cyanosis on exposed extremities, that may be associated with cold exposure. Lesions are usually symmetric and self-limiting, and symptoms can include numbness, tingling, pruritus, burning, or pain. Pernio-like skin lesions have been seen during the COVID-19 pandemic, though many patients have negative testing for infection by PCR and serology. Pernio may also be seen with autoimmune diseases or malignancy.
Meningococcemia is a rare disease caused by infection with gram-negative diplococci bacteria Neisseria meningitidis and spreads through saliva or respiratory secretions. Its clinical presentation can vary widely, from transient fever to fulminant disease. It is characterized by upper respiratory tract infection, fever, and petechial lesions associated with thrombocytopenia and coagulopathy.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Laborada is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Laborada have no relevant financial disclosures.
References
AlGhoozi DA, AlKhayyat HM. BMJ Case Reports CP 2021;14:e239910.
Jacobi M et al. Pediatr Infect Dis J. 2021;40(2):e93-4.
Paller A, Mancini AJ. Hurwitz clinical pediatric dermatology: A textbook of skin disorders of childhood and adolescence. 4th ed. Philadelphia (PA): Elsevier Saunders; 2011.
Radia T et al. Paediatr Respir Rev. 2021;38:51-7.
Ramien ML. Clin Exp Dermatol. 2021;46(3):420-9.
Numerous morphologies of skin rashes have been described in the setting of COVID-19, including pernio, livedoid rash, exanthem, and vasculitis. This classic constellation of symptoms (palpable purpura on buttocks/legs, abdominal pain, arthralgia, hematuria) is highly consistent with Henoch-Schonlein purpura (HSP). There are now multiple case reports of COVID-19–associated HSP.
HSP is the most common type of childhood systemic vasculitis. It is mediated by immunoglobulin A (IgA) immune complex deposition and has been associated with respiratory tract infections, streptococcal species, parainfluenza virus, and human parvovirus B19, medications, vaccinations, and malignancies. HSP is usually a self-limiting disease, with a course over 4-6 weeks, and can affect multiple organs, including the skin, gastrointestinal tract, joints, and the kidneys. The diagnostic criteria include palpable purpura in the presence of one or more of the following: diffuse abdominal pain, arthritis or arthralgia, any biopsy showing predominant IgA deposition, and renal involvement in the form of hematuria or proteinuria. Renal disease is variable and is the most significant indicator of long-term prognosis. This teenager was treated with oral corticosteroids because of the severe periarticular edema and responded rapidly. His subsequent urine analyses normalized.
What is on the differential?
Multisystem inflammatory syndrome in children (MIS-C) is a rare, potentially fatal, complication of COVID-19 infection that causes inflammation of multiple organs, including the heart, lungs, kidneys, brain, skin, eyes, or the gastrointestinal tract. It commonly affects children around ages 8-9 years. Initial symptoms include fever, rash, red eyes, diarrhea, and vomiting that appear 2-6 weeks post COVID-19 infection. Like HSP, MIS-C can present with edema of the extremities, worsening hand/foot pain, and hematuria; however, the absence of both fever and the pattern of system involvement seen with MIS-C and classic findings in this patient are more consistent with HSP.
Reactive infectious mucocutaneous eruption (RIME) was recently coined to encompass both infection-associated Stevens-Johnson eruptions including Mycoplasma pneumoniae-induced rash and mucositis (MIRM) and mucocutaneous eruptions caused by nonmycoplasma pathogens (including Chlamydia pneumoniae, human parainfluenza virus 2, rhinovirus, adenovirus, enterovirus, human metapneumovirus, influenza B virus, and COVID-19). It is usually seen in male children and adolescents. Prodromal symptoms include cough, fever, and malaise and they precede the prominent feature of mucositis. Our patient’s lack of mucosal involvement is not consistent with RIME.
Perniosis (chilblains) is characterized by localized edematous patches of erythema or cyanosis on exposed extremities, that may be associated with cold exposure. Lesions are usually symmetric and self-limiting, and symptoms can include numbness, tingling, pruritus, burning, or pain. Pernio-like skin lesions have been seen during the COVID-19 pandemic, though many patients have negative testing for infection by PCR and serology. Pernio may also be seen with autoimmune diseases or malignancy.
Meningococcemia is a rare disease caused by infection with gram-negative diplococci bacteria Neisseria meningitidis and spreads through saliva or respiratory secretions. Its clinical presentation can vary widely, from transient fever to fulminant disease. It is characterized by upper respiratory tract infection, fever, and petechial lesions associated with thrombocytopenia and coagulopathy.
Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Ms. Laborada is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital. Dr. Eichenfield and Ms. Laborada have no relevant financial disclosures.
References
AlGhoozi DA, AlKhayyat HM. BMJ Case Reports CP 2021;14:e239910.
Jacobi M et al. Pediatr Infect Dis J. 2021;40(2):e93-4.
Paller A, Mancini AJ. Hurwitz clinical pediatric dermatology: A textbook of skin disorders of childhood and adolescence. 4th ed. Philadelphia (PA): Elsevier Saunders; 2011.
Radia T et al. Paediatr Respir Rev. 2021;38:51-7.
Ramien ML. Clin Exp Dermatol. 2021;46(3):420-9.
Striae gravidarum: More than a ‘nuisance,’ say researchers
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Contact allergens in medical devices: A cause for concern?
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.