User login
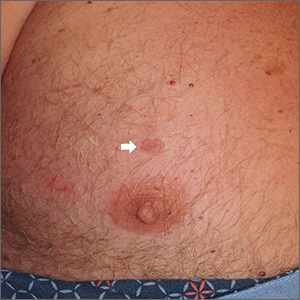
Chest lesion
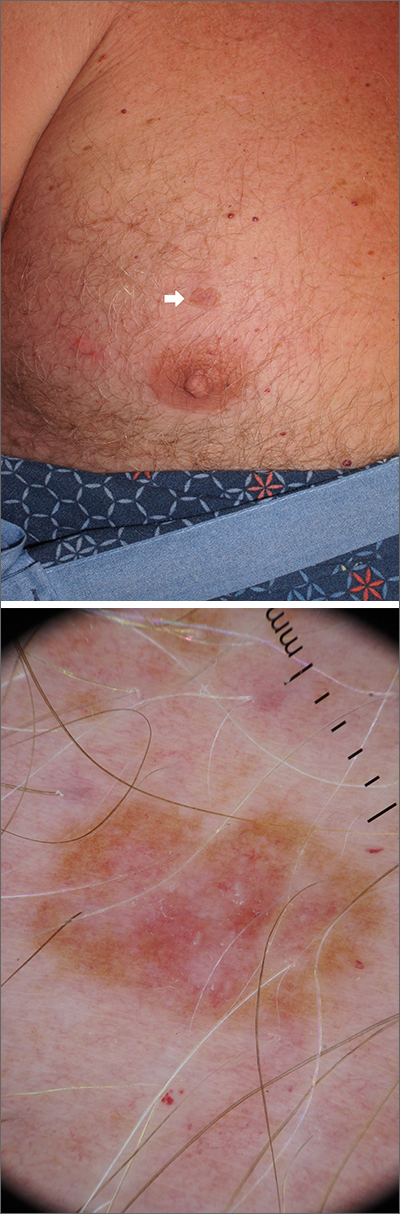
A scoop shave biopsy was performed, including at least a 1-mm margin of normal-looking skin. Pathology was consistent with melanoma in situ.
Melanoma in situ, also called Stage 0 melanoma, is defined by atypical melanocytes that have not begun to invade the dermis and, therefore, have a Breslow thickness of 0 mm. While invasive melanoma is responsible for the largest number of skin cancer deaths in the United States (estimated to be 7990 in 2023), melanoma in situ maintains a very high cure rate when treated appropriately.1
Dermoscopy can help differentiate melanoma from benign nevi or other benign skin lesions. In this case, dermoscopy revealed a fine pigment network at the periphery that indicated this lesion was made up of melanocytes. There were also atypical vascular markings (the milky red color) in the center. Taken together, these findings were strongly indicative of melanoma.
Standard of care for melanoma in situ is a wide local excision with a margin of 5 to 10 mm. Melanoma in situ does not require sentinel lymph node biopsy. However, a lymph node biopsy would have been necessary if the melanoma had been ≥ 1 mm in thickness or if it had been ≥ 0.8 mm in thickness with higher-risk features, such as an increased number of mitoses per high-power field on pathology. Mohs micrographic surgery (MMS) is emerging as an alternative method to wide local excision to treat melanoma in situ. However, it can only be done in specialized centers that can do rapid immunohistochemical staining on frozen sections. MMS is especially useful in cosmetically sensitive areas of the body and in areas where the true size of the melanoma in situ is unclear.
This patient subsequently underwent a wide local excision in the office with a margin of 6 mm. A sentinel lymph node biopsy was not performed. The patient will continue with skin surveillance consisting of full skin exams 3 to 4 times in the first year of diagnosis, then twice annually for Years 2 to 5. He will then come in for annual skin exams after that.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. American Cancer Society. Cancer Facts & Figures 2023. Atlanta: American Cancer Society; 2023. Accessed February 20, 2023. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf

A scoop shave biopsy was performed, including at least a 1-mm margin of normal-looking skin. Pathology was consistent with melanoma in situ.
Melanoma in situ, also called Stage 0 melanoma, is defined by atypical melanocytes that have not begun to invade the dermis and, therefore, have a Breslow thickness of 0 mm. While invasive melanoma is responsible for the largest number of skin cancer deaths in the United States (estimated to be 7990 in 2023), melanoma in situ maintains a very high cure rate when treated appropriately.1
Dermoscopy can help differentiate melanoma from benign nevi or other benign skin lesions. In this case, dermoscopy revealed a fine pigment network at the periphery that indicated this lesion was made up of melanocytes. There were also atypical vascular markings (the milky red color) in the center. Taken together, these findings were strongly indicative of melanoma.
Standard of care for melanoma in situ is a wide local excision with a margin of 5 to 10 mm. Melanoma in situ does not require sentinel lymph node biopsy. However, a lymph node biopsy would have been necessary if the melanoma had been ≥ 1 mm in thickness or if it had been ≥ 0.8 mm in thickness with higher-risk features, such as an increased number of mitoses per high-power field on pathology. Mohs micrographic surgery (MMS) is emerging as an alternative method to wide local excision to treat melanoma in situ. However, it can only be done in specialized centers that can do rapid immunohistochemical staining on frozen sections. MMS is especially useful in cosmetically sensitive areas of the body and in areas where the true size of the melanoma in situ is unclear.
This patient subsequently underwent a wide local excision in the office with a margin of 6 mm. A sentinel lymph node biopsy was not performed. The patient will continue with skin surveillance consisting of full skin exams 3 to 4 times in the first year of diagnosis, then twice annually for Years 2 to 5. He will then come in for annual skin exams after that.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

A scoop shave biopsy was performed, including at least a 1-mm margin of normal-looking skin. Pathology was consistent with melanoma in situ.
Melanoma in situ, also called Stage 0 melanoma, is defined by atypical melanocytes that have not begun to invade the dermis and, therefore, have a Breslow thickness of 0 mm. While invasive melanoma is responsible for the largest number of skin cancer deaths in the United States (estimated to be 7990 in 2023), melanoma in situ maintains a very high cure rate when treated appropriately.1
Dermoscopy can help differentiate melanoma from benign nevi or other benign skin lesions. In this case, dermoscopy revealed a fine pigment network at the periphery that indicated this lesion was made up of melanocytes. There were also atypical vascular markings (the milky red color) in the center. Taken together, these findings were strongly indicative of melanoma.
Standard of care for melanoma in situ is a wide local excision with a margin of 5 to 10 mm. Melanoma in situ does not require sentinel lymph node biopsy. However, a lymph node biopsy would have been necessary if the melanoma had been ≥ 1 mm in thickness or if it had been ≥ 0.8 mm in thickness with higher-risk features, such as an increased number of mitoses per high-power field on pathology. Mohs micrographic surgery (MMS) is emerging as an alternative method to wide local excision to treat melanoma in situ. However, it can only be done in specialized centers that can do rapid immunohistochemical staining on frozen sections. MMS is especially useful in cosmetically sensitive areas of the body and in areas where the true size of the melanoma in situ is unclear.
This patient subsequently underwent a wide local excision in the office with a margin of 6 mm. A sentinel lymph node biopsy was not performed. The patient will continue with skin surveillance consisting of full skin exams 3 to 4 times in the first year of diagnosis, then twice annually for Years 2 to 5. He will then come in for annual skin exams after that.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. American Cancer Society. Cancer Facts & Figures 2023. Atlanta: American Cancer Society; 2023. Accessed February 20, 2023. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
1. American Cancer Society. Cancer Facts & Figures 2023. Atlanta: American Cancer Society; 2023. Accessed February 20, 2023. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
Topical or intralesional cidofovir an option for recalcitrant warts
HONOLULU – Combining or those located in areas that are challenging to treat, according to John S. Barbieri, MD, MBA.
“There are 5 million office visits per year in the United States for warts and molluscum, and they’re most common in pediatrics,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “In fact, some studies have suggested that one in three children in primary school suffers from warts.”
According to a 2012 Cochrane review of topical therapies for warts, first-line treatments such as salicylic acid, cryotherapy, 5-FU, or Candida antigen injection often have modest efficacy when used alone. For example, the authors found that salicylic acid and cryotherapy cleared warts in about 60%-70% of cases, respectively, but clearance rates were improved by combining the two therapies.
In an earlier literature review and meta-analysis, investigators evaluated the effect of 5-FU plus salicylic acid or salicylic acid alone. The therapeutic effect for common warts across all studies was a 63.4% response rate (complete healing) for 5-FU/SA vversus 23.1% for the 5-FU–free controls, respectively. For plantar warts, the response rate was 63% versus 11%, respectively.
“But what about the person with multiple warts or those in challenging locations where you might worry about destructive treatments hurting the nail fold or causing nail dystrophy?” Dr. Barbieri asked. “Maybe they’ve used salicylic acid or intralesional Candida and they’re still not getting better. What can we do for these patients?”
Emerging research suggests that topical cidofovir can be a valuable option for recalcitrant warts or those in sensitive locations. In a case report of a 10-year-old boy with more than 50 severe verrucous papules on his hands and face that were recalcitrant to multiple conventional therapies, topical 1% cidofovir applied daily for 8 weeks was effective, with no adverse side effects. A young female patient who presented to Dr. Barbieri with multiple warts around the nail matrix of several fingers experienced complete clearance after treatment with topical cidofovir, he said. Other researchers found this approach to be effective for plantar warts as well, in a report of two brothers with severe combined immunodeficiency after hematopoietic stem cell transplantation with persistent warts that did not respond to traditional topical treatments.
“Topical cidofovir is typically a painless treatment, which is nice, especially for our pediatric patients who might be afraid of other therapies like or cryotherapy or intralesional injections,” One limitation is that it is “a bit expensive,” Dr. Barbieri said. “To have topical cidofovir compounded is typically $100-$300, depending on the quantity and strength that you ask for.”
Intralesional cidofovir is another treatment option. In a retrospective study of 58 patients, Dr. Barbieri and colleagues evaluated the outcome of intralesional cidofovir treatment of warts in immunocompromised and nonimmunocompromised patients. Rates of improvement ranged from 98.3% to 100%, while resolution rates ranged from 75.9% to 97.6%.
“Most of the patients had warts for more than 5 years and almost half of them had recalcitrant warts,” Dr. Barbieri said. “These were mostly adult patients, but I think this is a treatment that can work in younger populations as well. About 10%-15% had HIV or cancer or diabetes or were transplant recipients, but despite these challenges and despite these recalcitrant warts, about 100% had improvement.”
He pointed out that cidofovir is available as a 75 mg/mL vial that comes with a 5 mL single-use vial. He dilutes this with normal saline to create a 15 mg/mL solution.
“If you want to be efficient you can try to schedule multiple patients together on the same day as a single vial is sufficient to treat about 25 patients,” assuming about 1 mL is injected per patient, he said. “The challenge with intralesional cidofovir is that it’s painful beyond just the needle part of the injection. Sometimes a nerve block can be helpful. But this can be an effective treatment for patients with recalcitrant warts or those with comorbidities.”
Other intralesional therapies to try for recalcitrant warts, he said, include bleomycin (1 U/mL solution, 1-2 mL per treatment, spaced every 2-4 weeks), and 5-FU (a 4:1 mixture of 5-FU [50 mg/mL] and 2% lidocaine).
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Medscape and this news organization are owned by the same parent company.
HONOLULU – Combining or those located in areas that are challenging to treat, according to John S. Barbieri, MD, MBA.
“There are 5 million office visits per year in the United States for warts and molluscum, and they’re most common in pediatrics,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “In fact, some studies have suggested that one in three children in primary school suffers from warts.”
According to a 2012 Cochrane review of topical therapies for warts, first-line treatments such as salicylic acid, cryotherapy, 5-FU, or Candida antigen injection often have modest efficacy when used alone. For example, the authors found that salicylic acid and cryotherapy cleared warts in about 60%-70% of cases, respectively, but clearance rates were improved by combining the two therapies.
In an earlier literature review and meta-analysis, investigators evaluated the effect of 5-FU plus salicylic acid or salicylic acid alone. The therapeutic effect for common warts across all studies was a 63.4% response rate (complete healing) for 5-FU/SA vversus 23.1% for the 5-FU–free controls, respectively. For plantar warts, the response rate was 63% versus 11%, respectively.
“But what about the person with multiple warts or those in challenging locations where you might worry about destructive treatments hurting the nail fold or causing nail dystrophy?” Dr. Barbieri asked. “Maybe they’ve used salicylic acid or intralesional Candida and they’re still not getting better. What can we do for these patients?”
Emerging research suggests that topical cidofovir can be a valuable option for recalcitrant warts or those in sensitive locations. In a case report of a 10-year-old boy with more than 50 severe verrucous papules on his hands and face that were recalcitrant to multiple conventional therapies, topical 1% cidofovir applied daily for 8 weeks was effective, with no adverse side effects. A young female patient who presented to Dr. Barbieri with multiple warts around the nail matrix of several fingers experienced complete clearance after treatment with topical cidofovir, he said. Other researchers found this approach to be effective for plantar warts as well, in a report of two brothers with severe combined immunodeficiency after hematopoietic stem cell transplantation with persistent warts that did not respond to traditional topical treatments.
“Topical cidofovir is typically a painless treatment, which is nice, especially for our pediatric patients who might be afraid of other therapies like or cryotherapy or intralesional injections,” One limitation is that it is “a bit expensive,” Dr. Barbieri said. “To have topical cidofovir compounded is typically $100-$300, depending on the quantity and strength that you ask for.”
Intralesional cidofovir is another treatment option. In a retrospective study of 58 patients, Dr. Barbieri and colleagues evaluated the outcome of intralesional cidofovir treatment of warts in immunocompromised and nonimmunocompromised patients. Rates of improvement ranged from 98.3% to 100%, while resolution rates ranged from 75.9% to 97.6%.
“Most of the patients had warts for more than 5 years and almost half of them had recalcitrant warts,” Dr. Barbieri said. “These were mostly adult patients, but I think this is a treatment that can work in younger populations as well. About 10%-15% had HIV or cancer or diabetes or were transplant recipients, but despite these challenges and despite these recalcitrant warts, about 100% had improvement.”
He pointed out that cidofovir is available as a 75 mg/mL vial that comes with a 5 mL single-use vial. He dilutes this with normal saline to create a 15 mg/mL solution.
“If you want to be efficient you can try to schedule multiple patients together on the same day as a single vial is sufficient to treat about 25 patients,” assuming about 1 mL is injected per patient, he said. “The challenge with intralesional cidofovir is that it’s painful beyond just the needle part of the injection. Sometimes a nerve block can be helpful. But this can be an effective treatment for patients with recalcitrant warts or those with comorbidities.”
Other intralesional therapies to try for recalcitrant warts, he said, include bleomycin (1 U/mL solution, 1-2 mL per treatment, spaced every 2-4 weeks), and 5-FU (a 4:1 mixture of 5-FU [50 mg/mL] and 2% lidocaine).
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Medscape and this news organization are owned by the same parent company.
HONOLULU – Combining or those located in areas that are challenging to treat, according to John S. Barbieri, MD, MBA.
“There are 5 million office visits per year in the United States for warts and molluscum, and they’re most common in pediatrics,” Dr. Barbieri, of the department of dermatology at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “In fact, some studies have suggested that one in three children in primary school suffers from warts.”
According to a 2012 Cochrane review of topical therapies for warts, first-line treatments such as salicylic acid, cryotherapy, 5-FU, or Candida antigen injection often have modest efficacy when used alone. For example, the authors found that salicylic acid and cryotherapy cleared warts in about 60%-70% of cases, respectively, but clearance rates were improved by combining the two therapies.
In an earlier literature review and meta-analysis, investigators evaluated the effect of 5-FU plus salicylic acid or salicylic acid alone. The therapeutic effect for common warts across all studies was a 63.4% response rate (complete healing) for 5-FU/SA vversus 23.1% for the 5-FU–free controls, respectively. For plantar warts, the response rate was 63% versus 11%, respectively.
“But what about the person with multiple warts or those in challenging locations where you might worry about destructive treatments hurting the nail fold or causing nail dystrophy?” Dr. Barbieri asked. “Maybe they’ve used salicylic acid or intralesional Candida and they’re still not getting better. What can we do for these patients?”
Emerging research suggests that topical cidofovir can be a valuable option for recalcitrant warts or those in sensitive locations. In a case report of a 10-year-old boy with more than 50 severe verrucous papules on his hands and face that were recalcitrant to multiple conventional therapies, topical 1% cidofovir applied daily for 8 weeks was effective, with no adverse side effects. A young female patient who presented to Dr. Barbieri with multiple warts around the nail matrix of several fingers experienced complete clearance after treatment with topical cidofovir, he said. Other researchers found this approach to be effective for plantar warts as well, in a report of two brothers with severe combined immunodeficiency after hematopoietic stem cell transplantation with persistent warts that did not respond to traditional topical treatments.
“Topical cidofovir is typically a painless treatment, which is nice, especially for our pediatric patients who might be afraid of other therapies like or cryotherapy or intralesional injections,” One limitation is that it is “a bit expensive,” Dr. Barbieri said. “To have topical cidofovir compounded is typically $100-$300, depending on the quantity and strength that you ask for.”
Intralesional cidofovir is another treatment option. In a retrospective study of 58 patients, Dr. Barbieri and colleagues evaluated the outcome of intralesional cidofovir treatment of warts in immunocompromised and nonimmunocompromised patients. Rates of improvement ranged from 98.3% to 100%, while resolution rates ranged from 75.9% to 97.6%.
“Most of the patients had warts for more than 5 years and almost half of them had recalcitrant warts,” Dr. Barbieri said. “These were mostly adult patients, but I think this is a treatment that can work in younger populations as well. About 10%-15% had HIV or cancer or diabetes or were transplant recipients, but despite these challenges and despite these recalcitrant warts, about 100% had improvement.”
He pointed out that cidofovir is available as a 75 mg/mL vial that comes with a 5 mL single-use vial. He dilutes this with normal saline to create a 15 mg/mL solution.
“If you want to be efficient you can try to schedule multiple patients together on the same day as a single vial is sufficient to treat about 25 patients,” assuming about 1 mL is injected per patient, he said. “The challenge with intralesional cidofovir is that it’s painful beyond just the needle part of the injection. Sometimes a nerve block can be helpful. But this can be an effective treatment for patients with recalcitrant warts or those with comorbidities.”
Other intralesional therapies to try for recalcitrant warts, he said, include bleomycin (1 U/mL solution, 1-2 mL per treatment, spaced every 2-4 weeks), and 5-FU (a 4:1 mixture of 5-FU [50 mg/mL] and 2% lidocaine).
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPELIVE! HAWAII DERMATOLOGY SEMINAR
Treating nail psoriasis: Intralesional injections and biologics
HONOLULU – combined with systemic therapy.
One might think of intralesional injections “as a torture method from the medieval days,” she said at the Hawaii Dermatology Seminar provided by MedscapeLIVE!, but intramatricial corticosteroid injections have been performed for many years as a treatment for nail psoriasis, typically with triamcinolone acetonide.
According to Dr. Armstrong, professor of dermatology and associate dean of clinical research at the University of Southern California, Los Angeles, nail matrix psoriasis can present as pitting, leukonychia, red macules in the lunula, crumbling, or trachyonychia. Nail bed psoriasis can present as splinter hemorrhages and onycholysis, hyperkeratosis and splinter hemorrhages, salmon patch or oil spot dyschromia, or onycholysis and salmon patch dyschromia.
In a German cross-sectional study of patients with psoriasis, nails were one of the body sites that have the greatest impact on quality of life – especially those in younger age groups.
While topical treatments are generally considered first for limited disease involving special areas such as the nails, systemic therapy is warranted in patients with moderate-to-severe involvement of specific sites or in those refractory to topical therapy, Dr. Armstrong said.
In 2018, Indian researchers published results from an open-label study of 17 patients, with nail psoriasis, comparing three treatments . Patients were assigned to three groups of 30 nails each and treated with intramatricial injections of triamcinolone acetonide (10 mg/mL), methotrexate (25 mg/mL), and cyclosporine (50 mg/mL), respectively. Each nail was treated with two injections at 6-week intervals and graded at 24 weeks using the Nail Psoriasis Severity Index (NAPSI). In the triamcinolone acetonide and methotrexate groups, 50% of treated nails showed a greater than 75% improvement at 24 weeks, compared with 33% of those in the cyclosporine group. The most side effects occurred in the nails treated with cyclosporine.
When Dr. Armstrong performs intramatricial injections, she uses triamcinolone acetonide at 10 mg/mL. However, she said, “my favorite way of treating severe nail psoriasis is with biologics.”
In an early study of patients with moderate to severe psoriasis treated with the tumor necrosis factor blocker adalimumab 80 mg subcutaneously at week 0, followed by 40 mg subcutaneously every other week from weeks 1 to 15, a post hoc analysis on the effects on nail psoriasis showed a 10-point decrease in the median NAPSI score through week 16 – from 21 to 11 .
In VOYAGE 2, which compared the interleukin-23 blocker guselkumab and adalimumab in patients with moderate to severe psoriasis, the mean percent improvement from baseline in the NAPSI score was similar in patients treated with adalimumab or guselkumab at week 16 (39.6% vs. 46.9%, respectively) and at week 24 (55% vs. 53.7%).
In another study of patients with nail psoriasis, researchers evaluated the efficacy of the IL-17A antagonist secukinumab 150 mg, 300 mg, or placebo at weeks 0, 1, 2, 3, and 4, and every 4 weeks thereafter for 2.5 years. At 2.5 years, the mean reduction in NAPSI score was 63.6% in the secukinumab 150 mg group and 73.3% in the secukinumab 300 mg group.
“I do have to tell my patients what to expect, because the nails grow out slowly, but over time we do see this increase in efficacy,” Dr. Armstrong said.
Studies of another IL-17A antagonist, ixekizumab, have yielded positive results as well, she noted. In 2021, Taiwanese researchers published a systematic review and network meta-analysis to evaluate the efficacy of small molecule inhibitors and biologics in treating nail psoriasis. They drew from 39 studies involving 15,673 patients with nail psoriasis and found that the oral Janus kinase inhibitor tofacitinib and ixekizumab had the best efficacy for treating nail psoriasis in 10-16 weeks and 24-26 weeks, respectively.
“They found that overall, the biologics have a good effect on nail psoriasis and that the treatment effects are overall quite similar,” Dr. Armstrong said.
Dr. Armstrong disclosed that she is a consultant or adviser for numerous pharmaceutical companies. She has also received research funding from Bristol-Myers Squibb, Dermavant, Dermira, Leo, Lilly, Pfizer, and UCB Pharma.
HONOLULU – combined with systemic therapy.
One might think of intralesional injections “as a torture method from the medieval days,” she said at the Hawaii Dermatology Seminar provided by MedscapeLIVE!, but intramatricial corticosteroid injections have been performed for many years as a treatment for nail psoriasis, typically with triamcinolone acetonide.
According to Dr. Armstrong, professor of dermatology and associate dean of clinical research at the University of Southern California, Los Angeles, nail matrix psoriasis can present as pitting, leukonychia, red macules in the lunula, crumbling, or trachyonychia. Nail bed psoriasis can present as splinter hemorrhages and onycholysis, hyperkeratosis and splinter hemorrhages, salmon patch or oil spot dyschromia, or onycholysis and salmon patch dyschromia.
In a German cross-sectional study of patients with psoriasis, nails were one of the body sites that have the greatest impact on quality of life – especially those in younger age groups.
While topical treatments are generally considered first for limited disease involving special areas such as the nails, systemic therapy is warranted in patients with moderate-to-severe involvement of specific sites or in those refractory to topical therapy, Dr. Armstrong said.
In 2018, Indian researchers published results from an open-label study of 17 patients, with nail psoriasis, comparing three treatments . Patients were assigned to three groups of 30 nails each and treated with intramatricial injections of triamcinolone acetonide (10 mg/mL), methotrexate (25 mg/mL), and cyclosporine (50 mg/mL), respectively. Each nail was treated with two injections at 6-week intervals and graded at 24 weeks using the Nail Psoriasis Severity Index (NAPSI). In the triamcinolone acetonide and methotrexate groups, 50% of treated nails showed a greater than 75% improvement at 24 weeks, compared with 33% of those in the cyclosporine group. The most side effects occurred in the nails treated with cyclosporine.
When Dr. Armstrong performs intramatricial injections, she uses triamcinolone acetonide at 10 mg/mL. However, she said, “my favorite way of treating severe nail psoriasis is with biologics.”
In an early study of patients with moderate to severe psoriasis treated with the tumor necrosis factor blocker adalimumab 80 mg subcutaneously at week 0, followed by 40 mg subcutaneously every other week from weeks 1 to 15, a post hoc analysis on the effects on nail psoriasis showed a 10-point decrease in the median NAPSI score through week 16 – from 21 to 11 .
In VOYAGE 2, which compared the interleukin-23 blocker guselkumab and adalimumab in patients with moderate to severe psoriasis, the mean percent improvement from baseline in the NAPSI score was similar in patients treated with adalimumab or guselkumab at week 16 (39.6% vs. 46.9%, respectively) and at week 24 (55% vs. 53.7%).
In another study of patients with nail psoriasis, researchers evaluated the efficacy of the IL-17A antagonist secukinumab 150 mg, 300 mg, or placebo at weeks 0, 1, 2, 3, and 4, and every 4 weeks thereafter for 2.5 years. At 2.5 years, the mean reduction in NAPSI score was 63.6% in the secukinumab 150 mg group and 73.3% in the secukinumab 300 mg group.
“I do have to tell my patients what to expect, because the nails grow out slowly, but over time we do see this increase in efficacy,” Dr. Armstrong said.
Studies of another IL-17A antagonist, ixekizumab, have yielded positive results as well, she noted. In 2021, Taiwanese researchers published a systematic review and network meta-analysis to evaluate the efficacy of small molecule inhibitors and biologics in treating nail psoriasis. They drew from 39 studies involving 15,673 patients with nail psoriasis and found that the oral Janus kinase inhibitor tofacitinib and ixekizumab had the best efficacy for treating nail psoriasis in 10-16 weeks and 24-26 weeks, respectively.
“They found that overall, the biologics have a good effect on nail psoriasis and that the treatment effects are overall quite similar,” Dr. Armstrong said.
Dr. Armstrong disclosed that she is a consultant or adviser for numerous pharmaceutical companies. She has also received research funding from Bristol-Myers Squibb, Dermavant, Dermira, Leo, Lilly, Pfizer, and UCB Pharma.
HONOLULU – combined with systemic therapy.
One might think of intralesional injections “as a torture method from the medieval days,” she said at the Hawaii Dermatology Seminar provided by MedscapeLIVE!, but intramatricial corticosteroid injections have been performed for many years as a treatment for nail psoriasis, typically with triamcinolone acetonide.
According to Dr. Armstrong, professor of dermatology and associate dean of clinical research at the University of Southern California, Los Angeles, nail matrix psoriasis can present as pitting, leukonychia, red macules in the lunula, crumbling, or trachyonychia. Nail bed psoriasis can present as splinter hemorrhages and onycholysis, hyperkeratosis and splinter hemorrhages, salmon patch or oil spot dyschromia, or onycholysis and salmon patch dyschromia.
In a German cross-sectional study of patients with psoriasis, nails were one of the body sites that have the greatest impact on quality of life – especially those in younger age groups.
While topical treatments are generally considered first for limited disease involving special areas such as the nails, systemic therapy is warranted in patients with moderate-to-severe involvement of specific sites or in those refractory to topical therapy, Dr. Armstrong said.
In 2018, Indian researchers published results from an open-label study of 17 patients, with nail psoriasis, comparing three treatments . Patients were assigned to three groups of 30 nails each and treated with intramatricial injections of triamcinolone acetonide (10 mg/mL), methotrexate (25 mg/mL), and cyclosporine (50 mg/mL), respectively. Each nail was treated with two injections at 6-week intervals and graded at 24 weeks using the Nail Psoriasis Severity Index (NAPSI). In the triamcinolone acetonide and methotrexate groups, 50% of treated nails showed a greater than 75% improvement at 24 weeks, compared with 33% of those in the cyclosporine group. The most side effects occurred in the nails treated with cyclosporine.
When Dr. Armstrong performs intramatricial injections, she uses triamcinolone acetonide at 10 mg/mL. However, she said, “my favorite way of treating severe nail psoriasis is with biologics.”
In an early study of patients with moderate to severe psoriasis treated with the tumor necrosis factor blocker adalimumab 80 mg subcutaneously at week 0, followed by 40 mg subcutaneously every other week from weeks 1 to 15, a post hoc analysis on the effects on nail psoriasis showed a 10-point decrease in the median NAPSI score through week 16 – from 21 to 11 .
In VOYAGE 2, which compared the interleukin-23 blocker guselkumab and adalimumab in patients with moderate to severe psoriasis, the mean percent improvement from baseline in the NAPSI score was similar in patients treated with adalimumab or guselkumab at week 16 (39.6% vs. 46.9%, respectively) and at week 24 (55% vs. 53.7%).
In another study of patients with nail psoriasis, researchers evaluated the efficacy of the IL-17A antagonist secukinumab 150 mg, 300 mg, or placebo at weeks 0, 1, 2, 3, and 4, and every 4 weeks thereafter for 2.5 years. At 2.5 years, the mean reduction in NAPSI score was 63.6% in the secukinumab 150 mg group and 73.3% in the secukinumab 300 mg group.
“I do have to tell my patients what to expect, because the nails grow out slowly, but over time we do see this increase in efficacy,” Dr. Armstrong said.
Studies of another IL-17A antagonist, ixekizumab, have yielded positive results as well, she noted. In 2021, Taiwanese researchers published a systematic review and network meta-analysis to evaluate the efficacy of small molecule inhibitors and biologics in treating nail psoriasis. They drew from 39 studies involving 15,673 patients with nail psoriasis and found that the oral Janus kinase inhibitor tofacitinib and ixekizumab had the best efficacy for treating nail psoriasis in 10-16 weeks and 24-26 weeks, respectively.
“They found that overall, the biologics have a good effect on nail psoriasis and that the treatment effects are overall quite similar,” Dr. Armstrong said.
Dr. Armstrong disclosed that she is a consultant or adviser for numerous pharmaceutical companies. She has also received research funding from Bristol-Myers Squibb, Dermavant, Dermira, Leo, Lilly, Pfizer, and UCB Pharma.
AT THE MEDSCAPELIVE! HAWAII DERMATOLOGY SEMINAR
What’s holding back physicians from prescribing biosimilars? Four specialties weigh in
While most providers think that biosimilars will positively impact care, few feel that the economic benefits of biosimilars to date are enough to motivate switching.
In a new survey of over 350 dermatologists, gastroenterologists, ophthalmologists, and rheumatologists, clinicians shared their opinions on the rapidly evolving landscape of biosimilars, detailing top concerns about prescribing these medications and how they presently use biosimilars in clinical practice. Across all specialties, providers said they would be most likely to prescribe biosimilars to new patients or if a patient’s health plan mandated the switch. Most providers listed concerns about biosimilar efficacy and lack of economic benefit as the main barriers to adoption of biosimilars in clinical practice.
Cardinal Health, a health care services company based in Dublin, Ohio, conducted the surveys from July through October 2022.
Rheumatologists want cost-savings for patients
2023 is gearing up to be a big year for biosimilars for inflammatory diseases, with at least eight adalimumab biosimilars entering the market in the United States. Amjevita, manufactured by Amgen, was the first to become commercially available on Jan. 31. Out of 103 surveyed rheumatologists, 62% said they were very comfortable prescribing biosimilars to patients, and 32% said they were somewhat comfortable. Providers said they would be most likely to prescribe a biosimilar to new patients (40%) or if biosimilars were mandated by a patient’s health plan (41%). Nearly one-third (31%) of rheumatologists said that a discount of 21%-30% from a reference product would be necessary to consider switching a patient to a biosimilar.
There are several reasons why a rheumatologist might be wary of switching patients to biosimilars, said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “Rheumatologists will always express concern about changing medications that work well for their patients. It is not ideal to ‘force switch’ to a different product, even if it is almost identical,” he told this news organization in an email. “Also, we must remember that a patient on a biologic has failed traditional medications, which speaks to the struggle a patient must endure to get their disease under control. Fail-first situations can cause a rheumatologist to be initially resistant or hesitant to any changes.”
The top concerns among rheumatologists about prescribing biosimilars were medication efficacy (36%), lack of economic benefit (24%), and evaluating when to prescribe a biosimilar versus a reference product (17%). For adalimumab biosimilars, rheumatologists said that interchangeability – a regulatory designation where a biosimilar can be automatically substituted for its reference product at the pharmacy – and citrate-free formulation were the most important product attributes. Sixty-four percent of providers also noted that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
“There needs to be a true reduction in price, to change providers’ opinions on the economic benefits of biosimilars – in the system generally and for the patient,” Dr. Snow said. “Things will get there eventually, but it is not there yet, based on the list prices we see for some biosimilars.”
Gastroenterologists emphasize patient education
Gastroenterology is another specialty to be affected by the influx of adalimumab biosimilars. Out of 72 surveyed gastroenterologists, 86% said they were very comfortable prescribing biosimilars. About half (49%) said they would be most likely to prescribe a biosimilar to patients with health plans mandating a biosimilar. More than 60% of surveyed gastroenterologists said that biosimilars would positively impact care; providers were divided on the current economic benefits of biosimilars, with 36% saying that the current discounts on biosimilars versus reference products were not favorable enough to motivate switching, and 35% stating that they were. A total of 40% of surveyed providers said that savings of 21%-30%, compared with savings of a reference product, would motivate them to switch patients to a biosimilar, with all other clinical factors being equal.
Gastroenterologists said that, along with the efficacy and cost savings of biosimilars, providing patient education (18%) was a top concern when prescribing biosimilars. Eighty-four percent of respondents said that educating patients about biosimilars as safe and effective treatment options was at least somewhat important. Nearly all participants (99%) cited device ease-of-use as at least somewhat important when considering prescribing adalimumab biosimilars, in addition to interchangeability (97%) and citrate-free formulation (93%).
“Despite general acceptance of biosimilars, there remains some uncertainty regarding their place in the current gastroenterology landscape,” wrote Vivek Kaul, MD, a professor of medicine at the University of Rochester (N.Y.) Medical Center, in the report. “This is likely because only half of the survey respondents believed that biosimilars will positively impact gastroenterology care, further highlighting the ongoing need for real-world data and incorporation of biosimilar use and interchangeability into clinical guidelines.”
Few dermatologists currently prescribe biosimilars
Eight out of ten dermatologists reported being at least somewhat comfortable prescribing biosimilars to patients, though fewer than 20% said they had prescribed a biosimilar in the past year. This indicates limited adoption of infliximab biosimilars, which were the only biosimilars with a dermatologic indication available in 2022, Alex Gross, MD, a dermatologist in Cumming, Ga., noted in his featured commentary in the report. Just 15% of respondents disagreed that biosimilars would have a positive impact on care, and 41% said they were excited about new biosimilars becoming available.
About half (47%) of dermatologists thought the economic benefits of biosimilars were not strong enough to motivate switching patients from reference products. Twenty-nine percent of respondents said that discounts of 21%-30% from a reference product would motivate them to switch patients to a biosimilar, with all other clinical factors being equal, while 20% said they were not likely to prescribe a biosimilar regardless of savings.
Dermatologists may be concerned that these cost savings may not be passed onto patients, said Alison Ehrlich, MD, a dermatologist in Washington, in an email to this news organization. Patient out-of-pocket cost savings would need to be “both significant and transparent” to begin to change providers’ minds, she noted.
Biosimilar efficacy was a top concern for 48% of dermatologists, while 13% said their main concern around prescribing biosimilars was lack of payer adoption. At least 95% of providers said that device ease-of-use and interchangeability were the most important attributes when considering adalimumab biosimilars. Nearly two-thirds (65%) reported that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
If both patients and providers are informed on biosimilar use and there are cost benefits, dermatologists’ opinions may become more favorable toward biosimilars, but that will take time, Dr. Ehrlich said. “We are very early in the game for biosimilar use in dermatology,” she added.
Ophthalmologists remain wary
Biosimilars have been relatively new to ophthalmology, with the first ranibizumab biosimilar becoming commercially available in July 2022. In the survey, 64 retina specialists were asked different questions than participants from other specialties to gauge ophthalmologists› familiarity with the biosimilars approval process and their overall comfort prescribing these medications. The primary concerns with prescribing biosimilars among respondents was payer coverage (52%), being uncomfortable with biosimilars from a clinical standpoint (48%), and administrative barriers (45%), such as prior authorization. Despite this lack of comfort with biosimilars, two-thirds of participants thought the U.S. Food and Drug Administration approval process for these medications was sufficient to evaluate their efficacy and safety. Still, fewer than half (48%) of providers said they do or would prescribe biosimilars.
George Williams, MD, a spokesperson for the American Academy of Ophthalmology, noted that the FDA approval process for biosimilars was not as rigorous as for the respective reference product, and fewer patients are followed over a shorter time period. “Since anti–[vascular endothelial growth factor (VEGF)] therapy for indications such as neovascular age-related macular degeneration continues indefinitely over years, ophthalmologists may have concerns about the long-term efficacy and safety when applied to larger real-world populations. Ophthalmologists are well aware of safety issues with VEGF inhibitors arising after FDA approval,” he told this news organization in an email.
When asked about the likelihood of using either aflibercept or ranibizumab biosimilars in their clinical practice once commercially available, 70% of ophthalmologists said they would be at least somewhat likely to prescribe aflibercept biosimilars, and 64% said they would be at least somewhat likely to prescribe ranibizumab biosimilars. About half of respondents said they would not likely switch a currently stable patient on either aflibercept or ranibizumab to the corresponding biosimilar. More than half of ophthalmologists (56%) said they would prescribe a biosimilar only if it had an interchangeability designation.
Out of all four specialties, ophthalmologists more frequently reported that higher discounts from a reference product would be necessary to consider switching a patient to a biosimilar. Currently, many ophthalmologists are comfortable with the off-label use of bevacizumab (Avastin) for treating wet age-related macular degeneration, which also offers more cost savings than any currently available biosimilar on the market, Dr. Williams said.
While the limited number of respondents makes it difficult to draw concrete conclusions, Dr. Williams emphasized that the AAO supported the use of biosimilars. “We believe that with clinical experience ophthalmic biosimilars will become useful therapeutic agents,” he noted.
A version of this article first appeared on Medscape.com.
While most providers think that biosimilars will positively impact care, few feel that the economic benefits of biosimilars to date are enough to motivate switching.
In a new survey of over 350 dermatologists, gastroenterologists, ophthalmologists, and rheumatologists, clinicians shared their opinions on the rapidly evolving landscape of biosimilars, detailing top concerns about prescribing these medications and how they presently use biosimilars in clinical practice. Across all specialties, providers said they would be most likely to prescribe biosimilars to new patients or if a patient’s health plan mandated the switch. Most providers listed concerns about biosimilar efficacy and lack of economic benefit as the main barriers to adoption of biosimilars in clinical practice.
Cardinal Health, a health care services company based in Dublin, Ohio, conducted the surveys from July through October 2022.
Rheumatologists want cost-savings for patients
2023 is gearing up to be a big year for biosimilars for inflammatory diseases, with at least eight adalimumab biosimilars entering the market in the United States. Amjevita, manufactured by Amgen, was the first to become commercially available on Jan. 31. Out of 103 surveyed rheumatologists, 62% said they were very comfortable prescribing biosimilars to patients, and 32% said they were somewhat comfortable. Providers said they would be most likely to prescribe a biosimilar to new patients (40%) or if biosimilars were mandated by a patient’s health plan (41%). Nearly one-third (31%) of rheumatologists said that a discount of 21%-30% from a reference product would be necessary to consider switching a patient to a biosimilar.
There are several reasons why a rheumatologist might be wary of switching patients to biosimilars, said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “Rheumatologists will always express concern about changing medications that work well for their patients. It is not ideal to ‘force switch’ to a different product, even if it is almost identical,” he told this news organization in an email. “Also, we must remember that a patient on a biologic has failed traditional medications, which speaks to the struggle a patient must endure to get their disease under control. Fail-first situations can cause a rheumatologist to be initially resistant or hesitant to any changes.”
The top concerns among rheumatologists about prescribing biosimilars were medication efficacy (36%), lack of economic benefit (24%), and evaluating when to prescribe a biosimilar versus a reference product (17%). For adalimumab biosimilars, rheumatologists said that interchangeability – a regulatory designation where a biosimilar can be automatically substituted for its reference product at the pharmacy – and citrate-free formulation were the most important product attributes. Sixty-four percent of providers also noted that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
“There needs to be a true reduction in price, to change providers’ opinions on the economic benefits of biosimilars – in the system generally and for the patient,” Dr. Snow said. “Things will get there eventually, but it is not there yet, based on the list prices we see for some biosimilars.”
Gastroenterologists emphasize patient education
Gastroenterology is another specialty to be affected by the influx of adalimumab biosimilars. Out of 72 surveyed gastroenterologists, 86% said they were very comfortable prescribing biosimilars. About half (49%) said they would be most likely to prescribe a biosimilar to patients with health plans mandating a biosimilar. More than 60% of surveyed gastroenterologists said that biosimilars would positively impact care; providers were divided on the current economic benefits of biosimilars, with 36% saying that the current discounts on biosimilars versus reference products were not favorable enough to motivate switching, and 35% stating that they were. A total of 40% of surveyed providers said that savings of 21%-30%, compared with savings of a reference product, would motivate them to switch patients to a biosimilar, with all other clinical factors being equal.
Gastroenterologists said that, along with the efficacy and cost savings of biosimilars, providing patient education (18%) was a top concern when prescribing biosimilars. Eighty-four percent of respondents said that educating patients about biosimilars as safe and effective treatment options was at least somewhat important. Nearly all participants (99%) cited device ease-of-use as at least somewhat important when considering prescribing adalimumab biosimilars, in addition to interchangeability (97%) and citrate-free formulation (93%).
“Despite general acceptance of biosimilars, there remains some uncertainty regarding their place in the current gastroenterology landscape,” wrote Vivek Kaul, MD, a professor of medicine at the University of Rochester (N.Y.) Medical Center, in the report. “This is likely because only half of the survey respondents believed that biosimilars will positively impact gastroenterology care, further highlighting the ongoing need for real-world data and incorporation of biosimilar use and interchangeability into clinical guidelines.”
Few dermatologists currently prescribe biosimilars
Eight out of ten dermatologists reported being at least somewhat comfortable prescribing biosimilars to patients, though fewer than 20% said they had prescribed a biosimilar in the past year. This indicates limited adoption of infliximab biosimilars, which were the only biosimilars with a dermatologic indication available in 2022, Alex Gross, MD, a dermatologist in Cumming, Ga., noted in his featured commentary in the report. Just 15% of respondents disagreed that biosimilars would have a positive impact on care, and 41% said they were excited about new biosimilars becoming available.
About half (47%) of dermatologists thought the economic benefits of biosimilars were not strong enough to motivate switching patients from reference products. Twenty-nine percent of respondents said that discounts of 21%-30% from a reference product would motivate them to switch patients to a biosimilar, with all other clinical factors being equal, while 20% said they were not likely to prescribe a biosimilar regardless of savings.
Dermatologists may be concerned that these cost savings may not be passed onto patients, said Alison Ehrlich, MD, a dermatologist in Washington, in an email to this news organization. Patient out-of-pocket cost savings would need to be “both significant and transparent” to begin to change providers’ minds, she noted.
Biosimilar efficacy was a top concern for 48% of dermatologists, while 13% said their main concern around prescribing biosimilars was lack of payer adoption. At least 95% of providers said that device ease-of-use and interchangeability were the most important attributes when considering adalimumab biosimilars. Nearly two-thirds (65%) reported that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
If both patients and providers are informed on biosimilar use and there are cost benefits, dermatologists’ opinions may become more favorable toward biosimilars, but that will take time, Dr. Ehrlich said. “We are very early in the game for biosimilar use in dermatology,” she added.
Ophthalmologists remain wary
Biosimilars have been relatively new to ophthalmology, with the first ranibizumab biosimilar becoming commercially available in July 2022. In the survey, 64 retina specialists were asked different questions than participants from other specialties to gauge ophthalmologists› familiarity with the biosimilars approval process and their overall comfort prescribing these medications. The primary concerns with prescribing biosimilars among respondents was payer coverage (52%), being uncomfortable with biosimilars from a clinical standpoint (48%), and administrative barriers (45%), such as prior authorization. Despite this lack of comfort with biosimilars, two-thirds of participants thought the U.S. Food and Drug Administration approval process for these medications was sufficient to evaluate their efficacy and safety. Still, fewer than half (48%) of providers said they do or would prescribe biosimilars.
George Williams, MD, a spokesperson for the American Academy of Ophthalmology, noted that the FDA approval process for biosimilars was not as rigorous as for the respective reference product, and fewer patients are followed over a shorter time period. “Since anti–[vascular endothelial growth factor (VEGF)] therapy for indications such as neovascular age-related macular degeneration continues indefinitely over years, ophthalmologists may have concerns about the long-term efficacy and safety when applied to larger real-world populations. Ophthalmologists are well aware of safety issues with VEGF inhibitors arising after FDA approval,” he told this news organization in an email.
When asked about the likelihood of using either aflibercept or ranibizumab biosimilars in their clinical practice once commercially available, 70% of ophthalmologists said they would be at least somewhat likely to prescribe aflibercept biosimilars, and 64% said they would be at least somewhat likely to prescribe ranibizumab biosimilars. About half of respondents said they would not likely switch a currently stable patient on either aflibercept or ranibizumab to the corresponding biosimilar. More than half of ophthalmologists (56%) said they would prescribe a biosimilar only if it had an interchangeability designation.
Out of all four specialties, ophthalmologists more frequently reported that higher discounts from a reference product would be necessary to consider switching a patient to a biosimilar. Currently, many ophthalmologists are comfortable with the off-label use of bevacizumab (Avastin) for treating wet age-related macular degeneration, which also offers more cost savings than any currently available biosimilar on the market, Dr. Williams said.
While the limited number of respondents makes it difficult to draw concrete conclusions, Dr. Williams emphasized that the AAO supported the use of biosimilars. “We believe that with clinical experience ophthalmic biosimilars will become useful therapeutic agents,” he noted.
A version of this article first appeared on Medscape.com.
While most providers think that biosimilars will positively impact care, few feel that the economic benefits of biosimilars to date are enough to motivate switching.
In a new survey of over 350 dermatologists, gastroenterologists, ophthalmologists, and rheumatologists, clinicians shared their opinions on the rapidly evolving landscape of biosimilars, detailing top concerns about prescribing these medications and how they presently use biosimilars in clinical practice. Across all specialties, providers said they would be most likely to prescribe biosimilars to new patients or if a patient’s health plan mandated the switch. Most providers listed concerns about biosimilar efficacy and lack of economic benefit as the main barriers to adoption of biosimilars in clinical practice.
Cardinal Health, a health care services company based in Dublin, Ohio, conducted the surveys from July through October 2022.
Rheumatologists want cost-savings for patients
2023 is gearing up to be a big year for biosimilars for inflammatory diseases, with at least eight adalimumab biosimilars entering the market in the United States. Amjevita, manufactured by Amgen, was the first to become commercially available on Jan. 31. Out of 103 surveyed rheumatologists, 62% said they were very comfortable prescribing biosimilars to patients, and 32% said they were somewhat comfortable. Providers said they would be most likely to prescribe a biosimilar to new patients (40%) or if biosimilars were mandated by a patient’s health plan (41%). Nearly one-third (31%) of rheumatologists said that a discount of 21%-30% from a reference product would be necessary to consider switching a patient to a biosimilar.
There are several reasons why a rheumatologist might be wary of switching patients to biosimilars, said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “Rheumatologists will always express concern about changing medications that work well for their patients. It is not ideal to ‘force switch’ to a different product, even if it is almost identical,” he told this news organization in an email. “Also, we must remember that a patient on a biologic has failed traditional medications, which speaks to the struggle a patient must endure to get their disease under control. Fail-first situations can cause a rheumatologist to be initially resistant or hesitant to any changes.”
The top concerns among rheumatologists about prescribing biosimilars were medication efficacy (36%), lack of economic benefit (24%), and evaluating when to prescribe a biosimilar versus a reference product (17%). For adalimumab biosimilars, rheumatologists said that interchangeability – a regulatory designation where a biosimilar can be automatically substituted for its reference product at the pharmacy – and citrate-free formulation were the most important product attributes. Sixty-four percent of providers also noted that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
“There needs to be a true reduction in price, to change providers’ opinions on the economic benefits of biosimilars – in the system generally and for the patient,” Dr. Snow said. “Things will get there eventually, but it is not there yet, based on the list prices we see for some biosimilars.”
Gastroenterologists emphasize patient education
Gastroenterology is another specialty to be affected by the influx of adalimumab biosimilars. Out of 72 surveyed gastroenterologists, 86% said they were very comfortable prescribing biosimilars. About half (49%) said they would be most likely to prescribe a biosimilar to patients with health plans mandating a biosimilar. More than 60% of surveyed gastroenterologists said that biosimilars would positively impact care; providers were divided on the current economic benefits of biosimilars, with 36% saying that the current discounts on biosimilars versus reference products were not favorable enough to motivate switching, and 35% stating that they were. A total of 40% of surveyed providers said that savings of 21%-30%, compared with savings of a reference product, would motivate them to switch patients to a biosimilar, with all other clinical factors being equal.
Gastroenterologists said that, along with the efficacy and cost savings of biosimilars, providing patient education (18%) was a top concern when prescribing biosimilars. Eighty-four percent of respondents said that educating patients about biosimilars as safe and effective treatment options was at least somewhat important. Nearly all participants (99%) cited device ease-of-use as at least somewhat important when considering prescribing adalimumab biosimilars, in addition to interchangeability (97%) and citrate-free formulation (93%).
“Despite general acceptance of biosimilars, there remains some uncertainty regarding their place in the current gastroenterology landscape,” wrote Vivek Kaul, MD, a professor of medicine at the University of Rochester (N.Y.) Medical Center, in the report. “This is likely because only half of the survey respondents believed that biosimilars will positively impact gastroenterology care, further highlighting the ongoing need for real-world data and incorporation of biosimilar use and interchangeability into clinical guidelines.”
Few dermatologists currently prescribe biosimilars
Eight out of ten dermatologists reported being at least somewhat comfortable prescribing biosimilars to patients, though fewer than 20% said they had prescribed a biosimilar in the past year. This indicates limited adoption of infliximab biosimilars, which were the only biosimilars with a dermatologic indication available in 2022, Alex Gross, MD, a dermatologist in Cumming, Ga., noted in his featured commentary in the report. Just 15% of respondents disagreed that biosimilars would have a positive impact on care, and 41% said they were excited about new biosimilars becoming available.
About half (47%) of dermatologists thought the economic benefits of biosimilars were not strong enough to motivate switching patients from reference products. Twenty-nine percent of respondents said that discounts of 21%-30% from a reference product would motivate them to switch patients to a biosimilar, with all other clinical factors being equal, while 20% said they were not likely to prescribe a biosimilar regardless of savings.
Dermatologists may be concerned that these cost savings may not be passed onto patients, said Alison Ehrlich, MD, a dermatologist in Washington, in an email to this news organization. Patient out-of-pocket cost savings would need to be “both significant and transparent” to begin to change providers’ minds, she noted.
Biosimilar efficacy was a top concern for 48% of dermatologists, while 13% said their main concern around prescribing biosimilars was lack of payer adoption. At least 95% of providers said that device ease-of-use and interchangeability were the most important attributes when considering adalimumab biosimilars. Nearly two-thirds (65%) reported that patient out-of-pocket cost would be key when deciding to prescribe an adalimumab biosimilar.
If both patients and providers are informed on biosimilar use and there are cost benefits, dermatologists’ opinions may become more favorable toward biosimilars, but that will take time, Dr. Ehrlich said. “We are very early in the game for biosimilar use in dermatology,” she added.
Ophthalmologists remain wary
Biosimilars have been relatively new to ophthalmology, with the first ranibizumab biosimilar becoming commercially available in July 2022. In the survey, 64 retina specialists were asked different questions than participants from other specialties to gauge ophthalmologists› familiarity with the biosimilars approval process and their overall comfort prescribing these medications. The primary concerns with prescribing biosimilars among respondents was payer coverage (52%), being uncomfortable with biosimilars from a clinical standpoint (48%), and administrative barriers (45%), such as prior authorization. Despite this lack of comfort with biosimilars, two-thirds of participants thought the U.S. Food and Drug Administration approval process for these medications was sufficient to evaluate their efficacy and safety. Still, fewer than half (48%) of providers said they do or would prescribe biosimilars.
George Williams, MD, a spokesperson for the American Academy of Ophthalmology, noted that the FDA approval process for biosimilars was not as rigorous as for the respective reference product, and fewer patients are followed over a shorter time period. “Since anti–[vascular endothelial growth factor (VEGF)] therapy for indications such as neovascular age-related macular degeneration continues indefinitely over years, ophthalmologists may have concerns about the long-term efficacy and safety when applied to larger real-world populations. Ophthalmologists are well aware of safety issues with VEGF inhibitors arising after FDA approval,” he told this news organization in an email.
When asked about the likelihood of using either aflibercept or ranibizumab biosimilars in their clinical practice once commercially available, 70% of ophthalmologists said they would be at least somewhat likely to prescribe aflibercept biosimilars, and 64% said they would be at least somewhat likely to prescribe ranibizumab biosimilars. About half of respondents said they would not likely switch a currently stable patient on either aflibercept or ranibizumab to the corresponding biosimilar. More than half of ophthalmologists (56%) said they would prescribe a biosimilar only if it had an interchangeability designation.
Out of all four specialties, ophthalmologists more frequently reported that higher discounts from a reference product would be necessary to consider switching a patient to a biosimilar. Currently, many ophthalmologists are comfortable with the off-label use of bevacizumab (Avastin) for treating wet age-related macular degeneration, which also offers more cost savings than any currently available biosimilar on the market, Dr. Williams said.
While the limited number of respondents makes it difficult to draw concrete conclusions, Dr. Williams emphasized that the AAO supported the use of biosimilars. “We believe that with clinical experience ophthalmic biosimilars will become useful therapeutic agents,” he noted.
A version of this article first appeared on Medscape.com.
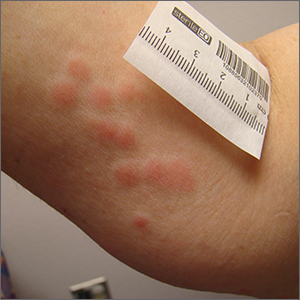
Clustered erythematous limb lesions
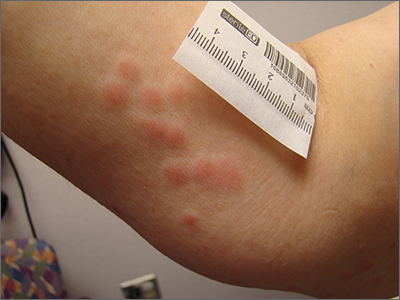
Clustered erythematous macular to papular lesions, especially those that stop at clothing lines, are a frequent manifestation of insect bites. In this case, the lesions lacked a central punctum that is common in many insect bites, so the most likely culprit was bed bugs. It is likely that this patient’s friend inadvertently brought the bed bugs into the home in her luggage or packed belongings. Over time, they spread around the home, causing the patient’s bites and inflammation. When questioned, the patient noted that she could actually see bugs around the couch in her home.
The scientific name for bed bugs is Cimex lectularis. Bed bugs require a blood meal from a host to survive, but they do not remain attached to the human body. Instead, they live in nearby fabrics. Bed bugs are visible to the naked eye when they are in the open, although they usually remain along the seams of fabric, edges of bedding, or in cracks and crevices. Often the feces of bed bugs will collect and be seen as dark spots or streaks on bedding.1
Treatment hinges on the eradication of the bed bugs. The erythematous itching lesions will resolve spontaneously over 1 to 2 weeks. Topical corticosteroids, including hydrocortisone, can be used as necessary to control the itching. Oral antihistamines can also help with itching.
Eradication of all the bed bugs in the home can be difficult and warrant professional extermination services. Washing clothing in hot water of at least 140 °F will kill the insects. Freezing items below –4 °F for at least 2 hours is also effective but may not be possible with home freezers.
It’s worth noting that resistance to insecticides has developed, making chemical eradication difficult. An alternative extermination protocol involves heating an entire home to the required temperatures to eradicate the infestation.1
This patient noted that she had already thrown away the couch, clothes, and bedding where she had seen the insects and had sprayed her apartment with insecticide. She was counseled to contact a professional exterminator to further evaluate the home for any additional areas of infestation and treat if any bed bugs were still in the home. She was also counseled to use loratadine 10 mg/d orally and topical 1% hydrocortisone ointment, as needed, for the itching and inflammation.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Parola P, Izri A. Bedbugs. N Engl J Med. 2020;382:2230-2237. doi: 10.1056/NEJMcp1905840

Clustered erythematous macular to papular lesions, especially those that stop at clothing lines, are a frequent manifestation of insect bites. In this case, the lesions lacked a central punctum that is common in many insect bites, so the most likely culprit was bed bugs. It is likely that this patient’s friend inadvertently brought the bed bugs into the home in her luggage or packed belongings. Over time, they spread around the home, causing the patient’s bites and inflammation. When questioned, the patient noted that she could actually see bugs around the couch in her home.
The scientific name for bed bugs is Cimex lectularis. Bed bugs require a blood meal from a host to survive, but they do not remain attached to the human body. Instead, they live in nearby fabrics. Bed bugs are visible to the naked eye when they are in the open, although they usually remain along the seams of fabric, edges of bedding, or in cracks and crevices. Often the feces of bed bugs will collect and be seen as dark spots or streaks on bedding.1
Treatment hinges on the eradication of the bed bugs. The erythematous itching lesions will resolve spontaneously over 1 to 2 weeks. Topical corticosteroids, including hydrocortisone, can be used as necessary to control the itching. Oral antihistamines can also help with itching.
Eradication of all the bed bugs in the home can be difficult and warrant professional extermination services. Washing clothing in hot water of at least 140 °F will kill the insects. Freezing items below –4 °F for at least 2 hours is also effective but may not be possible with home freezers.
It’s worth noting that resistance to insecticides has developed, making chemical eradication difficult. An alternative extermination protocol involves heating an entire home to the required temperatures to eradicate the infestation.1
This patient noted that she had already thrown away the couch, clothes, and bedding where she had seen the insects and had sprayed her apartment with insecticide. She was counseled to contact a professional exterminator to further evaluate the home for any additional areas of infestation and treat if any bed bugs were still in the home. She was also counseled to use loratadine 10 mg/d orally and topical 1% hydrocortisone ointment, as needed, for the itching and inflammation.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

Clustered erythematous macular to papular lesions, especially those that stop at clothing lines, are a frequent manifestation of insect bites. In this case, the lesions lacked a central punctum that is common in many insect bites, so the most likely culprit was bed bugs. It is likely that this patient’s friend inadvertently brought the bed bugs into the home in her luggage or packed belongings. Over time, they spread around the home, causing the patient’s bites and inflammation. When questioned, the patient noted that she could actually see bugs around the couch in her home.
The scientific name for bed bugs is Cimex lectularis. Bed bugs require a blood meal from a host to survive, but they do not remain attached to the human body. Instead, they live in nearby fabrics. Bed bugs are visible to the naked eye when they are in the open, although they usually remain along the seams of fabric, edges of bedding, or in cracks and crevices. Often the feces of bed bugs will collect and be seen as dark spots or streaks on bedding.1
Treatment hinges on the eradication of the bed bugs. The erythematous itching lesions will resolve spontaneously over 1 to 2 weeks. Topical corticosteroids, including hydrocortisone, can be used as necessary to control the itching. Oral antihistamines can also help with itching.
Eradication of all the bed bugs in the home can be difficult and warrant professional extermination services. Washing clothing in hot water of at least 140 °F will kill the insects. Freezing items below –4 °F for at least 2 hours is also effective but may not be possible with home freezers.
It’s worth noting that resistance to insecticides has developed, making chemical eradication difficult. An alternative extermination protocol involves heating an entire home to the required temperatures to eradicate the infestation.1
This patient noted that she had already thrown away the couch, clothes, and bedding where she had seen the insects and had sprayed her apartment with insecticide. She was counseled to contact a professional exterminator to further evaluate the home for any additional areas of infestation and treat if any bed bugs were still in the home. She was also counseled to use loratadine 10 mg/d orally and topical 1% hydrocortisone ointment, as needed, for the itching and inflammation.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Parola P, Izri A. Bedbugs. N Engl J Med. 2020;382:2230-2237. doi: 10.1056/NEJMcp1905840
1. Parola P, Izri A. Bedbugs. N Engl J Med. 2020;382:2230-2237. doi: 10.1056/NEJMcp1905840
Treating rosacea: Combination therapy, benzoyl peroxide, and the ‘STOP’ mnemonic
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
HONOLULU – More often than not, patients with rosacea require a combination of treatments to optimize the management of the disease, according to Julie C. Harper, MD.
“We’ve been more comfortable with the idea of combination therapy for acne than we have been for rosacea,” Dr. Harper, who practices in Birmingham, Ala., said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “If patients are doing great on one treatment, then don’t change it. But if there’s room for improvement, think about combinations.”
Treatment options for papules and pustules include ivermectin, metronidazole, azelaic acid, sodium sulfacetamide/sulfur, modified release doxycycline, minocycline foam, and microencapsulated benzoyl peroxide, 5%. Options for persistent background erythema include brimonidine and oxymetazoline, as well as device-based treatments, which include the pulsed dye laser, the KTP laser, intense pulsed light, and electrosurgery.
Dr. Harper said that she has been especially surprised by the effectiveness of one of these options, microencapsulated benzoyl peroxide cream, 5% (Epsolay), which is approved by the Food and Drug Administration for treating inflammatory lesions of rosacea in adults. In two identical, phase 3 randomized clinical trials of patients with inflammatory rosacea lesions, those treated with microencapsulated benzoyl peroxide achieved a 68.8% reduction in inflammatory lesions at 12 weeks (including 42.5% at week 2), compared with 38%-46% of those on the vehicle, according to the April 2022 announcement of the approval from the manufacturers, Sol-Gel Technologies and Galderma.
“A common drug is playing a key role,” Dr. Harper said. “What’s the mechanism of action? I have no idea. I wonder if there may be a bacterial pathogen after all,” possibly Staphylococcus epidermidis, she added. However, she noted, “it does appear that benzoyl peroxide has an impact on Demodex, so maybe that’s the primary way it’s working.”
In her opinion, a key standout from the clinical trial data is the drug’s rapid onset of action, with a 42.5% reduction of lesions at week 2. “What makes this different is that the 5% microencapsulated benzoyl peroxide cream is wrapped up in a silica shell,” said Dr. Harper, a past president of the American Acne and Rosacea Society. “The silica shell kind of acts like a speed bump that slows the release of drug onto the skin. We think that’s what may be giving us this better tolerability.”
In an interview at the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that prior to the approval of Epsolay, benzoyl peroxide was never considered a first-line treatment for rosacea. “The problem is, the conventional formulation is irritating to the skin,” said Dr. Stein Gold, who was involved in clinical trials of Epsolay.
“The benzoyl peroxide encapsulated in the silica shell allows for a slow and steady delivery of medication to the skin in a very controlled manner. It is exceptionally good at getting rosacea under control. In the clinical trials, when we looked at the baseline irritation of the skin and followed those patients when they used the benzoyl 5% microencapsulated benzoyl peroxide cream, the irritation improved.”
‘STOP’ mnemonic
When treating her patients with rosacea, Dr. Harper incorporates the mnemonic “STOP” to these patient visits:
S: Identify signs and symptoms of rosacea.
T: Discuss triggers. “We cannot make this disease triggerless, so when you’re talking to your patients, you need to find out what’s triggering their rosacea,” she said.
O: Agree on a treatment outcome. “What is it that’s important to the patient?” she said. “They may tell you, ‘I want to be able to not be so red,’ or ‘I want to get rid of the bumps,’ or ‘I want my eyes to not feel so dry.’ ”
P: Develop a plan that helps achieve that desired outcome with patients.
Dr. Harper disclosed ties with Almirall, Cassiopeia, Cutera, Galderma, EPI, L’Oréal, Ortho Dermatologics, Sol Gel, Sun Pharmaceutical Industries, and Vyne.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPE LIVE! HAWAII DERMATOLOGY SEMINAR
What does the future of psoriasis treatment look like?
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
HONOLULU – During office visits with Andrew Blauvelt, MD, MBA, many patients well controlled on biologic therapy for their moderate to severe psoriasis often ask him when their scheduled injections can stop.
The most common question he hears is, “ ‘Why do I have to keep doing this? I’ve been clear for 2 or 3 years,’ ” Dr. Blauvelt, president of Oregon Medical Research Center, Portland, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “We have terrific drugs for psoriasis, but how can we do better?”
Development of oral biologics. At least two companies are developing a peptide-type small molecule that blocks interleukin (IL)-17 or IL-23 signaling, but would be given as a pill, he said. Another concept in the works is a robotic pill for drug delivery. The pill, which is being developed by Rani Therapeutics, protects the biotherapeutic drug payload from digestion in the GI tract and auto-injects it into the wall of the small intestine, according to a report of two studies that demonstrated the safety and tolerability of the robotic pill in healthy humans.
In an animal study, the same researchers showed that delivering monoclonal antibodies with the robotic pill achieved bioavailability on par with that obtained by standard subcutaneous injections.
Identifying “super responders” who require less frequent dosing of medication. “There’s data to suggest that we can kind of back off treatment in these patients,” Dr. Blauvelt said.
Hitting treatment hard and early. “There’s a concept in medicine of hitting disease hard and hitting it early, before the disease can establish itself and cause damage,” he said.
Targeting tissue resident memory T cells. In psoriasis, the idea is that if you treat earlier, when patients are just diagnosed, “perhaps you might be able to decrease resident memory T cells that set up shop in the skin and are responsible for disease recurrences,” Dr. Blauvelt said. “Research has shown that IL-23 blockers decrease tissue resident memory T cells, and IL-17 blockers don’t. This could explain why we see long remissions in this class of drug because we’re getting at these resident memory T cells and knocking them down,” he explained. “Our hypothesis is that hitting hard and early in the treatment course with high-dose IL-23 blockade may be an effective strategy to induce long-term remissions and possible cure, what we call ‘knock-out therapy.’ ”
In a pilot study of 20 patients, Dr. Blauvelt and colleagues are evaluating whether higher initial doses of the IL-23 antagonist risankizumab (300 mg and 600 mg, 2 times and 4 times the standard initial doses for plaque psoriasis) can more effectively target resident memory T cells. “This involves dosing at weeks 0, 4, and 16, then stopping and measuring resident T cells in the tissue to see how long we can induce psoriasis remissions,” Dr. Blauvelt said.
“I have no data to share, but I think we have the potential for unprecedented PASI-100 numbers with no added safety concerns, and the potential to break away from established regular dosing patterns,” such as the possibility of yearly dosing, the possibility of long-term remissions, and the possibility of cure in some patients, he noted.
Inducing tolerance. This refers to efforts aimed at increasing regulatory T cells, which are natural T cells that calm inflammation. He described it as “revving up our natural anti-inflammatory T cells to help balance the immune system.”
Gene editing. This involves using CRISPR gene editing technology to cut genes as a way to cure disease. “What if we cut the IL-23 receptor?” Dr. Blauvelt asked. “You would get rid of that whole signaling pathway. Would the patient be fine?”
In an interview a the meeting, Linda Stein Gold, MD, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit, said that Dr. Blauvelt “has a very exciting view” of the future of psoriasis treatments. “I think that some of it will come true; we’ll have to see which,” Dr. Stein Gold said. “The idea that we might be able to change the trajectory of disease by being aggressive upfront, and possibly modify the course, is exciting. That would be a wonderful new treatment approach.”
Dr. Blauvelt disclosed ties with AbbVie, Abcentra, Affibody, Aligos, Almirall, Alumis, Amgen, AnaptysBio, Arcutis, Arena, ASLAN Pharma, Athenex, Bluefin, Boehringer Ingelheim, Bristol Myers Squibb, Cara, Dermavant, EcoR1, Escient, Evelo, Evommune, Forte, Galderma, Highlightll, Incyte, Innovent Bio, Janssen, Landos, Leo, Lilly, Merck, Novartis, Pfizer, Rapt, Regeneron, Sanofi-Genzyme, Spherix, Sun Pharmaceuticals Industries, TLL Pharmaceutical, TrialSpark, UCB Pharma, Vibliome, and Xencor.
Dr. Stein Gold disclosed ties with Almirall, Cutera, Dermata, Galderma, Novartis, Ortho Dermatologics, and Sun Pharmaceutical Industries, Ltd.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPE LIVE! HAWAII DERMATOLOGY SEMINAR
How to manage isotretinoin’s bothersome mucocutaneous side effects
HONOLULU –
“If they don’t have dry lips, you have to wonder if they’re even absorbing isotretinoin,” Dr. Barbieri, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “Everyone is going to get dry lips.”
According to a retrospective review of 1,743 patients started on isotretinoin, other common mucocutaneous side effects include eczema, nose bleeds, and eye problems. Emerging research suggests that there may be a role for oral omega-3 in decreasing such side effects of the drug. In a case control study, 118 patients were randomized to isotretinoin alone or isotretinoin plus 1 g/day of oral omega-3 for 16 weeks. At week 16, the rate of dry lips was 26% in the isoretinoin only group compared with 14% in the combination group; similar trends were seen with dry nose (11% vs. 0 %, respectively) and dry skin (11% vs. 2%).
“Omega-3 is a simple thing that we can think about recommending for patients,” Dr. Barbieri said. “It’s very safe, inexpensive, and it may help us manage these common sides effect we run into.”
Another potential side effect of isotretinoin that he characterized as underappreciated is chronic dry eye and other ocular changes. One retrospective cohort study of 14,682 adolescents and young adults in Israel found that use of the drug resulted in reduced tear production and reduced tear quality. In another study, a review and meta-analysis of 21 publications involving 1,105 eyes of 842 patients, isotretinoin use was associated with increased conjunctival fluorescein staining, decreased corneal thickness, and worse patient-reported ocular surface disease index scores.
“These changes may be mediated by meibomian gland dysfunction and atrophy,” Dr. Barbieri said. “Fortunately, many of these tear film changes appear to resolve after treatment. Those changes in corneal thickness do seem to get better. That’s reassuring.”
In a study of 54 patients treated with isotretinoin, tear production and quality returned to baseline within 6 months of treatment completion. “But some changes in the meibomian gland may be persistent,” Dr. Barbieri said. “At 6 and 12 months after the end of treatment, you can still see changes in the meibomian glands of patients who were treated with a standard course of 120 to 150 mg/kg isotretinoin,” he said, referring to the results of a study of 88 patients .
One study investigated the effects of omega-3 fatty acids and punctal plugs on tear film and ocular surface parameters in 90 patients receiving systemic isotretinoin therapy. They were divided into three groups: Those who received a soft preloaded silicone plug that was inserted in the inferior punctum of both eyes and received oral omega-3 fatty acid capsules twice daily for a total dose of 1,040 mg/day for 6 months; those who received a soft preloaded silicone plug and oral placebo, and those who received isotretinoin alone. At 6 months’ follow-up, those who were treated with omega-3 combined with the preloaded silicone plug had better meibomian gland function than did those who received isotretinoin alone or isotretinoin with the preloaded silicone plug.
Dr. Barbieri also noted that antihistamines may play a role in enhancing the effect of isotretinoin. In one study, 20 patients were treated with isotretinoin 0.4 mg/kg per day and 20 patients were also treated with an antihistamine, desloratadine 5 mg/day for 12 weeks. At week 12, patients in the group treated with isotretinoin and the antihistamine showed a more statistically significant decrease in acne lesion counts, compared with the isotretinoin-only group (reductions of 44.8% vs. 17.8%, respectively, in noninflammatory lesions; 55.8% vs. 22.9% in inflammatory lesions, and 45.6% vs. 18.7% in total lesions (P < .05 for all associations).
A subsequent larger study yielded similar findings. There were also lower rates of initial flaring and higher rates of patient satisfaction in the antihistamine groups in both studies.
In an interview at the meeting, Lawrence F. Eichenfield, MD, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, described Dr. Barbieri as “a leader in taking a comprehensive view on what the history and latest information is on isotretinoin. His fresh approach is something everyone should consider and figure out what they can use in their practice.”
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics. Medscape and this news organization are owned by the same parent company.
HONOLULU –
“If they don’t have dry lips, you have to wonder if they’re even absorbing isotretinoin,” Dr. Barbieri, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “Everyone is going to get dry lips.”
According to a retrospective review of 1,743 patients started on isotretinoin, other common mucocutaneous side effects include eczema, nose bleeds, and eye problems. Emerging research suggests that there may be a role for oral omega-3 in decreasing such side effects of the drug. In a case control study, 118 patients were randomized to isotretinoin alone or isotretinoin plus 1 g/day of oral omega-3 for 16 weeks. At week 16, the rate of dry lips was 26% in the isoretinoin only group compared with 14% in the combination group; similar trends were seen with dry nose (11% vs. 0 %, respectively) and dry skin (11% vs. 2%).
“Omega-3 is a simple thing that we can think about recommending for patients,” Dr. Barbieri said. “It’s very safe, inexpensive, and it may help us manage these common sides effect we run into.”
Another potential side effect of isotretinoin that he characterized as underappreciated is chronic dry eye and other ocular changes. One retrospective cohort study of 14,682 adolescents and young adults in Israel found that use of the drug resulted in reduced tear production and reduced tear quality. In another study, a review and meta-analysis of 21 publications involving 1,105 eyes of 842 patients, isotretinoin use was associated with increased conjunctival fluorescein staining, decreased corneal thickness, and worse patient-reported ocular surface disease index scores.
“These changes may be mediated by meibomian gland dysfunction and atrophy,” Dr. Barbieri said. “Fortunately, many of these tear film changes appear to resolve after treatment. Those changes in corneal thickness do seem to get better. That’s reassuring.”
In a study of 54 patients treated with isotretinoin, tear production and quality returned to baseline within 6 months of treatment completion. “But some changes in the meibomian gland may be persistent,” Dr. Barbieri said. “At 6 and 12 months after the end of treatment, you can still see changes in the meibomian glands of patients who were treated with a standard course of 120 to 150 mg/kg isotretinoin,” he said, referring to the results of a study of 88 patients .
One study investigated the effects of omega-3 fatty acids and punctal plugs on tear film and ocular surface parameters in 90 patients receiving systemic isotretinoin therapy. They were divided into three groups: Those who received a soft preloaded silicone plug that was inserted in the inferior punctum of both eyes and received oral omega-3 fatty acid capsules twice daily for a total dose of 1,040 mg/day for 6 months; those who received a soft preloaded silicone plug and oral placebo, and those who received isotretinoin alone. At 6 months’ follow-up, those who were treated with omega-3 combined with the preloaded silicone plug had better meibomian gland function than did those who received isotretinoin alone or isotretinoin with the preloaded silicone plug.
Dr. Barbieri also noted that antihistamines may play a role in enhancing the effect of isotretinoin. In one study, 20 patients were treated with isotretinoin 0.4 mg/kg per day and 20 patients were also treated with an antihistamine, desloratadine 5 mg/day for 12 weeks. At week 12, patients in the group treated with isotretinoin and the antihistamine showed a more statistically significant decrease in acne lesion counts, compared with the isotretinoin-only group (reductions of 44.8% vs. 17.8%, respectively, in noninflammatory lesions; 55.8% vs. 22.9% in inflammatory lesions, and 45.6% vs. 18.7% in total lesions (P < .05 for all associations).
A subsequent larger study yielded similar findings. There were also lower rates of initial flaring and higher rates of patient satisfaction in the antihistamine groups in both studies.
In an interview at the meeting, Lawrence F. Eichenfield, MD, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, described Dr. Barbieri as “a leader in taking a comprehensive view on what the history and latest information is on isotretinoin. His fresh approach is something everyone should consider and figure out what they can use in their practice.”
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics. Medscape and this news organization are owned by the same parent company.
HONOLULU –
“If they don’t have dry lips, you have to wonder if they’re even absorbing isotretinoin,” Dr. Barbieri, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said at the Hawaii Dermatology Seminar provided by MedscapeLIVE! “Everyone is going to get dry lips.”
According to a retrospective review of 1,743 patients started on isotretinoin, other common mucocutaneous side effects include eczema, nose bleeds, and eye problems. Emerging research suggests that there may be a role for oral omega-3 in decreasing such side effects of the drug. In a case control study, 118 patients were randomized to isotretinoin alone or isotretinoin plus 1 g/day of oral omega-3 for 16 weeks. At week 16, the rate of dry lips was 26% in the isoretinoin only group compared with 14% in the combination group; similar trends were seen with dry nose (11% vs. 0 %, respectively) and dry skin (11% vs. 2%).
“Omega-3 is a simple thing that we can think about recommending for patients,” Dr. Barbieri said. “It’s very safe, inexpensive, and it may help us manage these common sides effect we run into.”
Another potential side effect of isotretinoin that he characterized as underappreciated is chronic dry eye and other ocular changes. One retrospective cohort study of 14,682 adolescents and young adults in Israel found that use of the drug resulted in reduced tear production and reduced tear quality. In another study, a review and meta-analysis of 21 publications involving 1,105 eyes of 842 patients, isotretinoin use was associated with increased conjunctival fluorescein staining, decreased corneal thickness, and worse patient-reported ocular surface disease index scores.
“These changes may be mediated by meibomian gland dysfunction and atrophy,” Dr. Barbieri said. “Fortunately, many of these tear film changes appear to resolve after treatment. Those changes in corneal thickness do seem to get better. That’s reassuring.”
In a study of 54 patients treated with isotretinoin, tear production and quality returned to baseline within 6 months of treatment completion. “But some changes in the meibomian gland may be persistent,” Dr. Barbieri said. “At 6 and 12 months after the end of treatment, you can still see changes in the meibomian glands of patients who were treated with a standard course of 120 to 150 mg/kg isotretinoin,” he said, referring to the results of a study of 88 patients .
One study investigated the effects of omega-3 fatty acids and punctal plugs on tear film and ocular surface parameters in 90 patients receiving systemic isotretinoin therapy. They were divided into three groups: Those who received a soft preloaded silicone plug that was inserted in the inferior punctum of both eyes and received oral omega-3 fatty acid capsules twice daily for a total dose of 1,040 mg/day for 6 months; those who received a soft preloaded silicone plug and oral placebo, and those who received isotretinoin alone. At 6 months’ follow-up, those who were treated with omega-3 combined with the preloaded silicone plug had better meibomian gland function than did those who received isotretinoin alone or isotretinoin with the preloaded silicone plug.
Dr. Barbieri also noted that antihistamines may play a role in enhancing the effect of isotretinoin. In one study, 20 patients were treated with isotretinoin 0.4 mg/kg per day and 20 patients were also treated with an antihistamine, desloratadine 5 mg/day for 12 weeks. At week 12, patients in the group treated with isotretinoin and the antihistamine showed a more statistically significant decrease in acne lesion counts, compared with the isotretinoin-only group (reductions of 44.8% vs. 17.8%, respectively, in noninflammatory lesions; 55.8% vs. 22.9% in inflammatory lesions, and 45.6% vs. 18.7% in total lesions (P < .05 for all associations).
A subsequent larger study yielded similar findings. There were also lower rates of initial flaring and higher rates of patient satisfaction in the antihistamine groups in both studies.
In an interview at the meeting, Lawrence F. Eichenfield, MD, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, described Dr. Barbieri as “a leader in taking a comprehensive view on what the history and latest information is on isotretinoin. His fresh approach is something everyone should consider and figure out what they can use in their practice.”
Dr. Barbieri disclosed that he receives consulting fees from Dexcel for work unrelated to his presentation. Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics. Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPE LIVE! HAWAII DERMATOLOGY SEMINAR
Expert discusses pros, cons of molecular tests for melanoma
SAN DIEGO – , according to Gregory A. Hosler, MD, PhD.
At the annual Cutaneous Malignancy Update, Dr. Hosler, director of dermatopathology for ProPath, highlighted the following molecular tests currently used for the diagnosis of challenging melanocytic lesions:
Comparative genomic hybridization (CGH). This technique allows for the detection of chromosomal copy number changes throughout the tumor genome. “With CGH, test (tumor) DNA and normal DNA are differentially labeled and compared to a reference library. Gains and losses of portions of the tumor genome are determined by comparing the relative signals from these two groups,” said Dr. Hosler, clinical professor of pathology and dermatology at the University of Texas Southwestern Medical Center, Dallas.
“In the past, your library was a metaphase of spread of chromosomes, which introduced technical challenges and made performance of the assay labor intensive. Because of this, CGH is not routinely performed by clinical laboratories and is used more as an exploratory/research technique.”
Array CGH (also known as SNP array). Newer versions of CGH use short DNA sequences that are tiled onto a chip. “The interesting thing about these chips is that you can purchase them or design them on your own,” Dr. Hosler said. “The chips may cover the entire genome or cover specific areas of the genome at higher resolution.” One upside of array CGH, he continued, is that it allows one to detect essentially all gains or losses of chromosomal material in a single reaction. “It is not subject to the artifacts associated with cutting thin sections like with fluorescence in situ hybridization (FISH); it can detect copy number neutral loss of heterozygosity, and it is more scalable,” Dr. Hosler said at the meeting, which was hosted by Scripps MD Anderson Cancer Center.
One downside of array CGH is that does not allow one to analyze specific cells, “so if you have a tumor that’s heterogeneous, the assay is agnostic to this and spits out a result based on all the material provided,” he said. “You can’t parse out different areas of the lesion. It also does not track balanced translocations.” In addition, he said, “there are also questions about reimbursement and these are lab-developed tests, so each lab’s assay is different. Finally, it requires specialized equipment and expertise for interpretation.”
FISH. First-generation melanoma FISH assays, which became available in 2009, used six probes and four colors and had a sensitivity of about 87% and specificity of about 95%, Dr. Hosler said, but there were problems with those assays, particularly related to Spitz nevi. Spitz nevi often duplicate their chromosomes, “so instead of being diploid they’re tetraploid,” he said.
“The second-generation melanoma FISH assays addressed this by adding centromeres to the assay, and targeted probes could be compared to the centromeres on the same chromosome to determine if these were true copy number gains, due to genetic instability, or gains or losses of entire arms or whole chromosomes. This modification and the addition of new targets really improved upon the sensitivity and specificity (94% and 98%, respectively),” he said, noting that this assay is widely used.
Upsides of melanoma FISH assays are that they are a “fairly routine methodology” in large clinical laboratories, he said, and that many labs are familiar with interpretation. “I would say the biggest advantage to FISH is its ability to analyze specific cells, which is useful with small or heterogeneous tumors,” Dr. Hosler said. “Also, there is a genetic reimbursement code for it, and it yields diagnostic and potentially prognostic information.” For example, certain copy number changes have shown to portend a worse prognosis if they’re present in a melanocytic tumor, including alterations in CDKN2A, CCND1, MYC, topoisomerase, and BAP1.
Downsides of melanoma FISH assays are that they are expensive, labor-intensive, and require experts to interpret the results. “The stacking and truncation of cell nuclei innate to paraffin-embedded FISH make interpretation difficult,” he said. “Also, all colors cannot be viewed simultaneously, and each lab’s assay potentially is different, requiring validation. These are not [Food and Drug Administration]-approved tests.”
Next generation sequencing (NGS). Also known as high-throughput sequencing, this technique allows for the generation of millions of sequencing reads that are aligned to a standard human genome, and likely represents the wave of the future. “With NGS you can increase breadth, so you can sequence the entire genome if you want, but you can also increase depth, meaning increasing the number of reads over a single target of the genome,” Dr. Hosler said. “That’s useful if you’re looking for a low frequency mutation.”
For example, NGS allows one to detect alterations of BRAF and KIT and other potentially actionable alterations. It can also be used to detect mutations in benign and malignant melanocytic lesions, including historically diagnostically challenging Spitz and desmoplastic subgroups. Several different NGS technologies exist, and there are different strategies behind each assay, including whole genome sequencing, whole exome sequencing, transcriptome sequencing, and targeted panels. “I’ve seen panels of 10 and I’ve seen panels of 1,500; there’s a wide range,” Dr. Hosler said. “The biggest challenge with NGS, currently, is that it’s difficult to interpret. Trying to figure out what’s important and what’s not important can be challenging. Often you need a team of people who are experts in bioinformatics to interpret these results.”
Slow turnaround time is another downside. “It can take a month to get results, and sometimes clinicians don’t want to wait that long, especially if they think a lesion is melanoma, so that’s an area of focus for NGS laboratories,” he said. “And there are questions on reimbursement. If you run NGS on every unusual melanocytic lesion, that’s not a good use of health care dollars. Who’s paying for it? I don’t have an answer for you. It’s all over the map right now. Each lab’s test and billing practice is different.”
Dr. Hosler reported having no relevant financial disclosures. ProPath is a nationwide pathology practice.
SAN DIEGO – , according to Gregory A. Hosler, MD, PhD.
At the annual Cutaneous Malignancy Update, Dr. Hosler, director of dermatopathology for ProPath, highlighted the following molecular tests currently used for the diagnosis of challenging melanocytic lesions:
Comparative genomic hybridization (CGH). This technique allows for the detection of chromosomal copy number changes throughout the tumor genome. “With CGH, test (tumor) DNA and normal DNA are differentially labeled and compared to a reference library. Gains and losses of portions of the tumor genome are determined by comparing the relative signals from these two groups,” said Dr. Hosler, clinical professor of pathology and dermatology at the University of Texas Southwestern Medical Center, Dallas.
“In the past, your library was a metaphase of spread of chromosomes, which introduced technical challenges and made performance of the assay labor intensive. Because of this, CGH is not routinely performed by clinical laboratories and is used more as an exploratory/research technique.”
Array CGH (also known as SNP array). Newer versions of CGH use short DNA sequences that are tiled onto a chip. “The interesting thing about these chips is that you can purchase them or design them on your own,” Dr. Hosler said. “The chips may cover the entire genome or cover specific areas of the genome at higher resolution.” One upside of array CGH, he continued, is that it allows one to detect essentially all gains or losses of chromosomal material in a single reaction. “It is not subject to the artifacts associated with cutting thin sections like with fluorescence in situ hybridization (FISH); it can detect copy number neutral loss of heterozygosity, and it is more scalable,” Dr. Hosler said at the meeting, which was hosted by Scripps MD Anderson Cancer Center.
One downside of array CGH is that does not allow one to analyze specific cells, “so if you have a tumor that’s heterogeneous, the assay is agnostic to this and spits out a result based on all the material provided,” he said. “You can’t parse out different areas of the lesion. It also does not track balanced translocations.” In addition, he said, “there are also questions about reimbursement and these are lab-developed tests, so each lab’s assay is different. Finally, it requires specialized equipment and expertise for interpretation.”
FISH. First-generation melanoma FISH assays, which became available in 2009, used six probes and four colors and had a sensitivity of about 87% and specificity of about 95%, Dr. Hosler said, but there were problems with those assays, particularly related to Spitz nevi. Spitz nevi often duplicate their chromosomes, “so instead of being diploid they’re tetraploid,” he said.
“The second-generation melanoma FISH assays addressed this by adding centromeres to the assay, and targeted probes could be compared to the centromeres on the same chromosome to determine if these were true copy number gains, due to genetic instability, or gains or losses of entire arms or whole chromosomes. This modification and the addition of new targets really improved upon the sensitivity and specificity (94% and 98%, respectively),” he said, noting that this assay is widely used.
Upsides of melanoma FISH assays are that they are a “fairly routine methodology” in large clinical laboratories, he said, and that many labs are familiar with interpretation. “I would say the biggest advantage to FISH is its ability to analyze specific cells, which is useful with small or heterogeneous tumors,” Dr. Hosler said. “Also, there is a genetic reimbursement code for it, and it yields diagnostic and potentially prognostic information.” For example, certain copy number changes have shown to portend a worse prognosis if they’re present in a melanocytic tumor, including alterations in CDKN2A, CCND1, MYC, topoisomerase, and BAP1.
Downsides of melanoma FISH assays are that they are expensive, labor-intensive, and require experts to interpret the results. “The stacking and truncation of cell nuclei innate to paraffin-embedded FISH make interpretation difficult,” he said. “Also, all colors cannot be viewed simultaneously, and each lab’s assay potentially is different, requiring validation. These are not [Food and Drug Administration]-approved tests.”
Next generation sequencing (NGS). Also known as high-throughput sequencing, this technique allows for the generation of millions of sequencing reads that are aligned to a standard human genome, and likely represents the wave of the future. “With NGS you can increase breadth, so you can sequence the entire genome if you want, but you can also increase depth, meaning increasing the number of reads over a single target of the genome,” Dr. Hosler said. “That’s useful if you’re looking for a low frequency mutation.”
For example, NGS allows one to detect alterations of BRAF and KIT and other potentially actionable alterations. It can also be used to detect mutations in benign and malignant melanocytic lesions, including historically diagnostically challenging Spitz and desmoplastic subgroups. Several different NGS technologies exist, and there are different strategies behind each assay, including whole genome sequencing, whole exome sequencing, transcriptome sequencing, and targeted panels. “I’ve seen panels of 10 and I’ve seen panels of 1,500; there’s a wide range,” Dr. Hosler said. “The biggest challenge with NGS, currently, is that it’s difficult to interpret. Trying to figure out what’s important and what’s not important can be challenging. Often you need a team of people who are experts in bioinformatics to interpret these results.”
Slow turnaround time is another downside. “It can take a month to get results, and sometimes clinicians don’t want to wait that long, especially if they think a lesion is melanoma, so that’s an area of focus for NGS laboratories,” he said. “And there are questions on reimbursement. If you run NGS on every unusual melanocytic lesion, that’s not a good use of health care dollars. Who’s paying for it? I don’t have an answer for you. It’s all over the map right now. Each lab’s test and billing practice is different.”
Dr. Hosler reported having no relevant financial disclosures. ProPath is a nationwide pathology practice.
SAN DIEGO – , according to Gregory A. Hosler, MD, PhD.
At the annual Cutaneous Malignancy Update, Dr. Hosler, director of dermatopathology for ProPath, highlighted the following molecular tests currently used for the diagnosis of challenging melanocytic lesions:
Comparative genomic hybridization (CGH). This technique allows for the detection of chromosomal copy number changes throughout the tumor genome. “With CGH, test (tumor) DNA and normal DNA are differentially labeled and compared to a reference library. Gains and losses of portions of the tumor genome are determined by comparing the relative signals from these two groups,” said Dr. Hosler, clinical professor of pathology and dermatology at the University of Texas Southwestern Medical Center, Dallas.
“In the past, your library was a metaphase of spread of chromosomes, which introduced technical challenges and made performance of the assay labor intensive. Because of this, CGH is not routinely performed by clinical laboratories and is used more as an exploratory/research technique.”
Array CGH (also known as SNP array). Newer versions of CGH use short DNA sequences that are tiled onto a chip. “The interesting thing about these chips is that you can purchase them or design them on your own,” Dr. Hosler said. “The chips may cover the entire genome or cover specific areas of the genome at higher resolution.” One upside of array CGH, he continued, is that it allows one to detect essentially all gains or losses of chromosomal material in a single reaction. “It is not subject to the artifacts associated with cutting thin sections like with fluorescence in situ hybridization (FISH); it can detect copy number neutral loss of heterozygosity, and it is more scalable,” Dr. Hosler said at the meeting, which was hosted by Scripps MD Anderson Cancer Center.
One downside of array CGH is that does not allow one to analyze specific cells, “so if you have a tumor that’s heterogeneous, the assay is agnostic to this and spits out a result based on all the material provided,” he said. “You can’t parse out different areas of the lesion. It also does not track balanced translocations.” In addition, he said, “there are also questions about reimbursement and these are lab-developed tests, so each lab’s assay is different. Finally, it requires specialized equipment and expertise for interpretation.”
FISH. First-generation melanoma FISH assays, which became available in 2009, used six probes and four colors and had a sensitivity of about 87% and specificity of about 95%, Dr. Hosler said, but there were problems with those assays, particularly related to Spitz nevi. Spitz nevi often duplicate their chromosomes, “so instead of being diploid they’re tetraploid,” he said.
“The second-generation melanoma FISH assays addressed this by adding centromeres to the assay, and targeted probes could be compared to the centromeres on the same chromosome to determine if these were true copy number gains, due to genetic instability, or gains or losses of entire arms or whole chromosomes. This modification and the addition of new targets really improved upon the sensitivity and specificity (94% and 98%, respectively),” he said, noting that this assay is widely used.
Upsides of melanoma FISH assays are that they are a “fairly routine methodology” in large clinical laboratories, he said, and that many labs are familiar with interpretation. “I would say the biggest advantage to FISH is its ability to analyze specific cells, which is useful with small or heterogeneous tumors,” Dr. Hosler said. “Also, there is a genetic reimbursement code for it, and it yields diagnostic and potentially prognostic information.” For example, certain copy number changes have shown to portend a worse prognosis if they’re present in a melanocytic tumor, including alterations in CDKN2A, CCND1, MYC, topoisomerase, and BAP1.
Downsides of melanoma FISH assays are that they are expensive, labor-intensive, and require experts to interpret the results. “The stacking and truncation of cell nuclei innate to paraffin-embedded FISH make interpretation difficult,” he said. “Also, all colors cannot be viewed simultaneously, and each lab’s assay potentially is different, requiring validation. These are not [Food and Drug Administration]-approved tests.”
Next generation sequencing (NGS). Also known as high-throughput sequencing, this technique allows for the generation of millions of sequencing reads that are aligned to a standard human genome, and likely represents the wave of the future. “With NGS you can increase breadth, so you can sequence the entire genome if you want, but you can also increase depth, meaning increasing the number of reads over a single target of the genome,” Dr. Hosler said. “That’s useful if you’re looking for a low frequency mutation.”
For example, NGS allows one to detect alterations of BRAF and KIT and other potentially actionable alterations. It can also be used to detect mutations in benign and malignant melanocytic lesions, including historically diagnostically challenging Spitz and desmoplastic subgroups. Several different NGS technologies exist, and there are different strategies behind each assay, including whole genome sequencing, whole exome sequencing, transcriptome sequencing, and targeted panels. “I’ve seen panels of 10 and I’ve seen panels of 1,500; there’s a wide range,” Dr. Hosler said. “The biggest challenge with NGS, currently, is that it’s difficult to interpret. Trying to figure out what’s important and what’s not important can be challenging. Often you need a team of people who are experts in bioinformatics to interpret these results.”
Slow turnaround time is another downside. “It can take a month to get results, and sometimes clinicians don’t want to wait that long, especially if they think a lesion is melanoma, so that’s an area of focus for NGS laboratories,” he said. “And there are questions on reimbursement. If you run NGS on every unusual melanocytic lesion, that’s not a good use of health care dollars. Who’s paying for it? I don’t have an answer for you. It’s all over the map right now. Each lab’s test and billing practice is different.”
Dr. Hosler reported having no relevant financial disclosures. ProPath is a nationwide pathology practice.
AT MELANOMA 2023
Antibiotics and SJS/TEN: Study provides global prevalence
of SJS/TEN in connection with antibiotics.
“SJS/TEN is considered the most severe form of drug hypersensitivity reaction, and antibiotics are an important risk,” Erika Yue Lee, MD, and associates wrote in JAMA Dermatology.
Their analysis, which involved 38 studies published since 1987 with 2,917 patients from more than 20 countries, showed that 86% of all SJS/TEN cases were associated with a single drug, with the rest involving multiple drug triggers, infections, or other causes. More than a quarter (28%) of those patients had used an antibiotic, and the sulfonamides were the class most often triggering SJS/TEN, said Dr. Lee of the University of Toronto and associates.
Sulfonamides were responsible for 32% of the antibiotic-associated cases, which works out to 11% of all SJS/TEN cases included in the analysis. Penicillins were next with 22% of all antibiotic-associated cases, followed by the cephalosporins (11%), fluoroquinolones (4%), and macrolides (2%), the investigators reported.
A subgroup analysis conducted by age indicated that “there was no difference in the proportion of antibiotics associated with SJS/TEN between adult and pediatric groups,” they noted.
There were differences, however, among the various antibiotic classes. Sulfonamides represented 54% of antibiotic-triggered reactions in children, compared with 25% in adults, but adults were significantly more likely to have cephalosporin (23%) and fluoroquinolone (5%) involvement than were children (2% and 0, respectively). Macrolide-induced SJS/TEN was more common in children (18% vs. 1%), while the penicillin rate was 18% for both age groups, Dr. Lee and associates said.
A second subgroup analysis establishing the proportion of antibiotic-induced SJS/TEN by continent ranked Australia highest with 43%, but that was based on only one study of 42 patients. North America was slightly lower at 37%, but the analysis included 14 studies and 932 patients. Asia’s 16 studies and 1,298 patients were divided into three regions, with the lowest being the southeast at 16%, according to the researchers.
“Global sulfonamide antibiotic use has been decreasing since 2000 despite an ongoing upward trend of use in other antibiotic classes,” they wrote, but “antibiotics remain one of the most common culprit drugs for SJS/TEN in both adults and children worldwide.”
One of Dr. Lee’s associates has received personal fees from Janssen, AstraZeneca, UpToDate, Verve, BioCryst, Regeneron Pharmaceuticals, and Novavax and has served as codirector of IIID Pty Ltd, which holds a patent for HLA-B*57:01 testing and has a patent pending for detection of HLA-A*32:01 in connection with diagnosing drug reaction without any financial remuneration outside this study.
of SJS/TEN in connection with antibiotics.
“SJS/TEN is considered the most severe form of drug hypersensitivity reaction, and antibiotics are an important risk,” Erika Yue Lee, MD, and associates wrote in JAMA Dermatology.
Their analysis, which involved 38 studies published since 1987 with 2,917 patients from more than 20 countries, showed that 86% of all SJS/TEN cases were associated with a single drug, with the rest involving multiple drug triggers, infections, or other causes. More than a quarter (28%) of those patients had used an antibiotic, and the sulfonamides were the class most often triggering SJS/TEN, said Dr. Lee of the University of Toronto and associates.
Sulfonamides were responsible for 32% of the antibiotic-associated cases, which works out to 11% of all SJS/TEN cases included in the analysis. Penicillins were next with 22% of all antibiotic-associated cases, followed by the cephalosporins (11%), fluoroquinolones (4%), and macrolides (2%), the investigators reported.
A subgroup analysis conducted by age indicated that “there was no difference in the proportion of antibiotics associated with SJS/TEN between adult and pediatric groups,” they noted.
There were differences, however, among the various antibiotic classes. Sulfonamides represented 54% of antibiotic-triggered reactions in children, compared with 25% in adults, but adults were significantly more likely to have cephalosporin (23%) and fluoroquinolone (5%) involvement than were children (2% and 0, respectively). Macrolide-induced SJS/TEN was more common in children (18% vs. 1%), while the penicillin rate was 18% for both age groups, Dr. Lee and associates said.
A second subgroup analysis establishing the proportion of antibiotic-induced SJS/TEN by continent ranked Australia highest with 43%, but that was based on only one study of 42 patients. North America was slightly lower at 37%, but the analysis included 14 studies and 932 patients. Asia’s 16 studies and 1,298 patients were divided into three regions, with the lowest being the southeast at 16%, according to the researchers.
“Global sulfonamide antibiotic use has been decreasing since 2000 despite an ongoing upward trend of use in other antibiotic classes,” they wrote, but “antibiotics remain one of the most common culprit drugs for SJS/TEN in both adults and children worldwide.”
One of Dr. Lee’s associates has received personal fees from Janssen, AstraZeneca, UpToDate, Verve, BioCryst, Regeneron Pharmaceuticals, and Novavax and has served as codirector of IIID Pty Ltd, which holds a patent for HLA-B*57:01 testing and has a patent pending for detection of HLA-A*32:01 in connection with diagnosing drug reaction without any financial remuneration outside this study.
of SJS/TEN in connection with antibiotics.
“SJS/TEN is considered the most severe form of drug hypersensitivity reaction, and antibiotics are an important risk,” Erika Yue Lee, MD, and associates wrote in JAMA Dermatology.
Their analysis, which involved 38 studies published since 1987 with 2,917 patients from more than 20 countries, showed that 86% of all SJS/TEN cases were associated with a single drug, with the rest involving multiple drug triggers, infections, or other causes. More than a quarter (28%) of those patients had used an antibiotic, and the sulfonamides were the class most often triggering SJS/TEN, said Dr. Lee of the University of Toronto and associates.
Sulfonamides were responsible for 32% of the antibiotic-associated cases, which works out to 11% of all SJS/TEN cases included in the analysis. Penicillins were next with 22% of all antibiotic-associated cases, followed by the cephalosporins (11%), fluoroquinolones (4%), and macrolides (2%), the investigators reported.
A subgroup analysis conducted by age indicated that “there was no difference in the proportion of antibiotics associated with SJS/TEN between adult and pediatric groups,” they noted.
There were differences, however, among the various antibiotic classes. Sulfonamides represented 54% of antibiotic-triggered reactions in children, compared with 25% in adults, but adults were significantly more likely to have cephalosporin (23%) and fluoroquinolone (5%) involvement than were children (2% and 0, respectively). Macrolide-induced SJS/TEN was more common in children (18% vs. 1%), while the penicillin rate was 18% for both age groups, Dr. Lee and associates said.
A second subgroup analysis establishing the proportion of antibiotic-induced SJS/TEN by continent ranked Australia highest with 43%, but that was based on only one study of 42 patients. North America was slightly lower at 37%, but the analysis included 14 studies and 932 patients. Asia’s 16 studies and 1,298 patients were divided into three regions, with the lowest being the southeast at 16%, according to the researchers.
“Global sulfonamide antibiotic use has been decreasing since 2000 despite an ongoing upward trend of use in other antibiotic classes,” they wrote, but “antibiotics remain one of the most common culprit drugs for SJS/TEN in both adults and children worldwide.”
One of Dr. Lee’s associates has received personal fees from Janssen, AstraZeneca, UpToDate, Verve, BioCryst, Regeneron Pharmaceuticals, and Novavax and has served as codirector of IIID Pty Ltd, which holds a patent for HLA-B*57:01 testing and has a patent pending for detection of HLA-A*32:01 in connection with diagnosing drug reaction without any financial remuneration outside this study.
FROM JAMA DERMATOLOGY