User login
Study Evaluates Safety of Benzoyl Peroxide Products for Acne
according to results from an analysis that used gas chromatography–mass spectrometry and other methods.
The analysis, which was published in the Journal of Investigative Dermatology and expands on a similar study released more than 6 months ago, also found that encapsulated BPO products break down into benzene at room temperature but that refrigerating them may mitigate this effect.
“Our research provides the first experimental evidence that cold storage can help reduce the rate of benzoyl peroxide breakdown into benzene,” said one of the study authors, Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Connecticut. “Therefore, cold storage throughout the entire supply chain — from manufacturing to patient use — is a reasonable and proportional measure at this time for those continuing to use benzoyl peroxide medicine.” One acne product, the newer prescription triple-combination therapy (adapalene-clindamycin-BPO) “already has a cold shipping process in place; the patient just needs to continue that at home,” he noted.
For the study — which was funded by an independent lab, Valisure — researchers led by Valisure CEO and founder David Light, used gas chromatography-mass spectrometry to detect benzene levels in 111 BPO drug products from major US retailers and selected ion flow tube mass-spectrometry to quantify the release of benzene in real time. Benzene levels ranged from 0.16 ppm to 35.30 ppm, and 38 of the products (34%) had levels above the FDA limit of 2 ppm for drug products. “The results of the products sampled in this study suggest that formulation is likely the strongest contributor to benzene concentrations in BPO drug products that are commercially available, since the magnitude of benzene detected correlates most closely with specific brands or product types within certain brands,” the study authors wrote.
When the researchers tested the stability of a prescription encapsulated BPO drug product at cold (2 °C) and elevated temperature (50 °C), no apparent benzene formation was observed at 2 °C, whereas high levels of benzene formed at 50 °C, “suggesting that encapsulation technology may not stabilize BPO drug products, but cold storage may greatly reduce benzene formation,” they wrote.
In another component of the study, researchers exposed a BP drug product to a UVA/UVB lamp for 2 hours and found detectable benzene through evaporation and substantial benzene formation when exposed to UV light at levels below peak sunlight. The experiment “strongly justifies the package label warnings to avoid sun exposure when using BPO drug products,” the authors wrote. “Further evaluation to determine the influence of sun exposure on BPO drug product degradation and benzene formation is warranted.”
In an interview, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, Massachusetts characterized the findings as “an important issue that we should take seriously.” However, “we also must not overreact.”
BPO is a foundational acne treatment without any clear alternative, he said, pointing out that no evidence currently exists “to support that routine use of benzoyl peroxide–containing products for acne is associated with a meaningful risk of benzene in the blood or an increased risk of cancer.”
And although it is prudent to minimize benzene exposure as much as possible, Barbieri continued, “it is not clear that these levels are a clinically meaningful incremental risk in the setting of an acne cream or wash. There is minimal cutaneous absorption of benzene, and it is uncertain how much benzene aerosolizes with routine use, particularly for washes which are not left on the skin.”
Bunick said that the combined data from this and the study published in March 2024 affected which BPO products he recommends for patients with acne. “I am using exclusively the triple combination therapy (adapalene-clindamycin-benzoyl peroxide) because I know it has the necessary cold supply chain in place to protect the product’s stability. I further encourage patients to place all their benzoyl peroxide–containing products in the refrigerator at home to reduce benzene formation and exposure.”
Bunick reported having served as an investigator and/or a consultant/speaker for many pharmaceutical companies, including as a consultant for Ortho-Dermatologics; but none related to this study. Barbieri reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to results from an analysis that used gas chromatography–mass spectrometry and other methods.
The analysis, which was published in the Journal of Investigative Dermatology and expands on a similar study released more than 6 months ago, also found that encapsulated BPO products break down into benzene at room temperature but that refrigerating them may mitigate this effect.
“Our research provides the first experimental evidence that cold storage can help reduce the rate of benzoyl peroxide breakdown into benzene,” said one of the study authors, Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Connecticut. “Therefore, cold storage throughout the entire supply chain — from manufacturing to patient use — is a reasonable and proportional measure at this time for those continuing to use benzoyl peroxide medicine.” One acne product, the newer prescription triple-combination therapy (adapalene-clindamycin-BPO) “already has a cold shipping process in place; the patient just needs to continue that at home,” he noted.
For the study — which was funded by an independent lab, Valisure — researchers led by Valisure CEO and founder David Light, used gas chromatography-mass spectrometry to detect benzene levels in 111 BPO drug products from major US retailers and selected ion flow tube mass-spectrometry to quantify the release of benzene in real time. Benzene levels ranged from 0.16 ppm to 35.30 ppm, and 38 of the products (34%) had levels above the FDA limit of 2 ppm for drug products. “The results of the products sampled in this study suggest that formulation is likely the strongest contributor to benzene concentrations in BPO drug products that are commercially available, since the magnitude of benzene detected correlates most closely with specific brands or product types within certain brands,” the study authors wrote.
When the researchers tested the stability of a prescription encapsulated BPO drug product at cold (2 °C) and elevated temperature (50 °C), no apparent benzene formation was observed at 2 °C, whereas high levels of benzene formed at 50 °C, “suggesting that encapsulation technology may not stabilize BPO drug products, but cold storage may greatly reduce benzene formation,” they wrote.
In another component of the study, researchers exposed a BP drug product to a UVA/UVB lamp for 2 hours and found detectable benzene through evaporation and substantial benzene formation when exposed to UV light at levels below peak sunlight. The experiment “strongly justifies the package label warnings to avoid sun exposure when using BPO drug products,” the authors wrote. “Further evaluation to determine the influence of sun exposure on BPO drug product degradation and benzene formation is warranted.”
In an interview, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, Massachusetts characterized the findings as “an important issue that we should take seriously.” However, “we also must not overreact.”
BPO is a foundational acne treatment without any clear alternative, he said, pointing out that no evidence currently exists “to support that routine use of benzoyl peroxide–containing products for acne is associated with a meaningful risk of benzene in the blood or an increased risk of cancer.”
And although it is prudent to minimize benzene exposure as much as possible, Barbieri continued, “it is not clear that these levels are a clinically meaningful incremental risk in the setting of an acne cream or wash. There is minimal cutaneous absorption of benzene, and it is uncertain how much benzene aerosolizes with routine use, particularly for washes which are not left on the skin.”
Bunick said that the combined data from this and the study published in March 2024 affected which BPO products he recommends for patients with acne. “I am using exclusively the triple combination therapy (adapalene-clindamycin-benzoyl peroxide) because I know it has the necessary cold supply chain in place to protect the product’s stability. I further encourage patients to place all their benzoyl peroxide–containing products in the refrigerator at home to reduce benzene formation and exposure.”
Bunick reported having served as an investigator and/or a consultant/speaker for many pharmaceutical companies, including as a consultant for Ortho-Dermatologics; but none related to this study. Barbieri reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
according to results from an analysis that used gas chromatography–mass spectrometry and other methods.
The analysis, which was published in the Journal of Investigative Dermatology and expands on a similar study released more than 6 months ago, also found that encapsulated BPO products break down into benzene at room temperature but that refrigerating them may mitigate this effect.
“Our research provides the first experimental evidence that cold storage can help reduce the rate of benzoyl peroxide breakdown into benzene,” said one of the study authors, Christopher G. Bunick, MD, PhD, associate professor of dermatology at Yale University, New Haven, Connecticut. “Therefore, cold storage throughout the entire supply chain — from manufacturing to patient use — is a reasonable and proportional measure at this time for those continuing to use benzoyl peroxide medicine.” One acne product, the newer prescription triple-combination therapy (adapalene-clindamycin-BPO) “already has a cold shipping process in place; the patient just needs to continue that at home,” he noted.
For the study — which was funded by an independent lab, Valisure — researchers led by Valisure CEO and founder David Light, used gas chromatography-mass spectrometry to detect benzene levels in 111 BPO drug products from major US retailers and selected ion flow tube mass-spectrometry to quantify the release of benzene in real time. Benzene levels ranged from 0.16 ppm to 35.30 ppm, and 38 of the products (34%) had levels above the FDA limit of 2 ppm for drug products. “The results of the products sampled in this study suggest that formulation is likely the strongest contributor to benzene concentrations in BPO drug products that are commercially available, since the magnitude of benzene detected correlates most closely with specific brands or product types within certain brands,” the study authors wrote.
When the researchers tested the stability of a prescription encapsulated BPO drug product at cold (2 °C) and elevated temperature (50 °C), no apparent benzene formation was observed at 2 °C, whereas high levels of benzene formed at 50 °C, “suggesting that encapsulation technology may not stabilize BPO drug products, but cold storage may greatly reduce benzene formation,” they wrote.
In another component of the study, researchers exposed a BP drug product to a UVA/UVB lamp for 2 hours and found detectable benzene through evaporation and substantial benzene formation when exposed to UV light at levels below peak sunlight. The experiment “strongly justifies the package label warnings to avoid sun exposure when using BPO drug products,” the authors wrote. “Further evaluation to determine the influence of sun exposure on BPO drug product degradation and benzene formation is warranted.”
In an interview, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, Massachusetts characterized the findings as “an important issue that we should take seriously.” However, “we also must not overreact.”
BPO is a foundational acne treatment without any clear alternative, he said, pointing out that no evidence currently exists “to support that routine use of benzoyl peroxide–containing products for acne is associated with a meaningful risk of benzene in the blood or an increased risk of cancer.”
And although it is prudent to minimize benzene exposure as much as possible, Barbieri continued, “it is not clear that these levels are a clinically meaningful incremental risk in the setting of an acne cream or wash. There is minimal cutaneous absorption of benzene, and it is uncertain how much benzene aerosolizes with routine use, particularly for washes which are not left on the skin.”
Bunick said that the combined data from this and the study published in March 2024 affected which BPO products he recommends for patients with acne. “I am using exclusively the triple combination therapy (adapalene-clindamycin-benzoyl peroxide) because I know it has the necessary cold supply chain in place to protect the product’s stability. I further encourage patients to place all their benzoyl peroxide–containing products in the refrigerator at home to reduce benzene formation and exposure.”
Bunick reported having served as an investigator and/or a consultant/speaker for many pharmaceutical companies, including as a consultant for Ortho-Dermatologics; but none related to this study. Barbieri reported having no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
A 7-Year-Old Boy Presents With Dark Spots on His Scalp and Areas of Poor Hair Growth
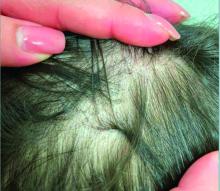
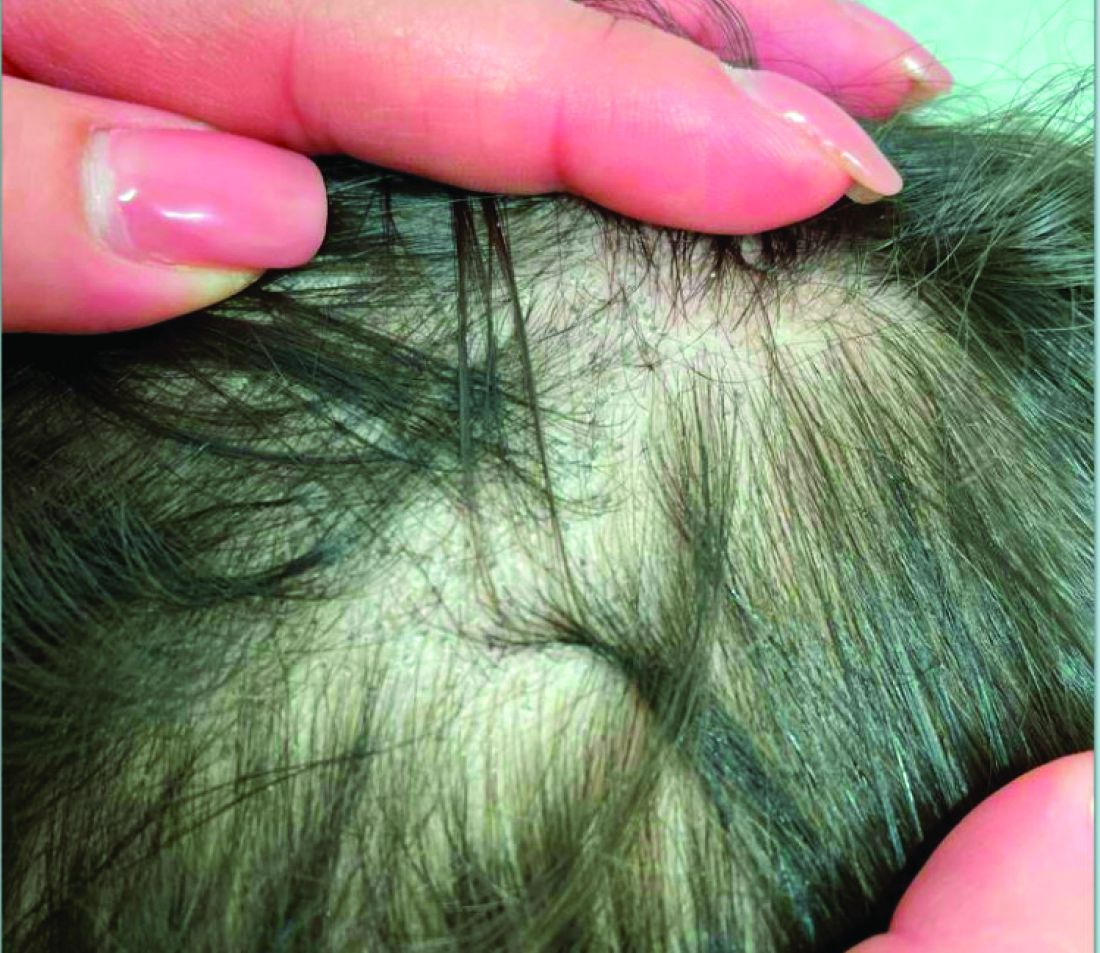
Given the trichoscopic findings, scrapings from the scaly areas were taken and revealed hyphae, confirming the diagnosis of tinea capitis. A fungal culture identified Trichophyton tonsurans as the causative organism.
Tinea capitis is the most common dermatophyte infection in children. Risk factors include participation in close-contact sports like wrestling or jiu-jitsu, attendance at daycare for younger children, African American hair care practices, pet ownership (particularly cats and rodents), and living in overcrowded conditions.
Diagnosis of tinea capitis requires a thorough clinical history to identify potential risk factors. On physical examination, patchy hair loss with associated scaling should raise suspicion for tinea capitis. Inflammatory signs, such as pustules and swelling, may suggest the presence of a kerion, further supporting the diagnosis. Although some practitioners use Wood’s lamp to help with diagnosis, its utility is limited. It detects fluorescence in Microsporum species (exothrix infections) but not in Trichophyton species (endothrix infections).
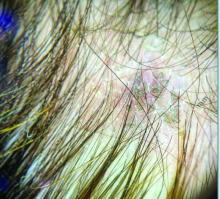
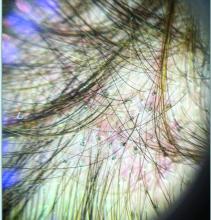
Trichoscopy can be a valuable tool when inflammation is minimal, and only hair loss and scaling are observed. Trichoscopic findings suggestive of tinea capitis include comma hairs, corkscrew hairs (as seen in this patient), Morse code-like hairs, zigzag hairs, bent hairs, block hairs, and i-hairs. Other common, though not characteristic, findings include broken hairs, black dots, perifollicular scaling, and diffuse scaling.
KOH (potassium hydroxide) analysis is another useful method for detecting fungal elements, though it does not identify the specific fungus and may not be available in all clinical settings. Mycologic culture remains the gold standard for diagnosing tinea capitis, though results can take 3-4 weeks. Newer diagnostic techniques, such as PCR analysis and MALDI-TOF/MS, offer more rapid identification of the causative organism.
The differential diagnosis includes:
- Seborrheic dermatitis, which presents with greasy, yellowish scales and itching, with trichoscopy showing twisted, coiled hairs and yellowish scaling.
- Psoriasis, which can mimic tinea capitis but presents with well-demarcated red plaques and silvery-white scales. Trichoscopy shows red dots and uniform scaling.
- Alopecia areata, which causes patchy hair loss without inflammation or scaling, with trichoscopic findings of exclamation mark hairs, black dots, and yellow dots.
- Trichotillomania, a hair-pulling disorder, which results in irregular patches of hair loss. Trichoscopy shows broken hairs of varying lengths, V-sign hairs, and flame-shaped residues at follicular openings.
Treatment of tinea capitis requires systemic antifungals and topical agents to prevent fungal spore spread. Several treatment guidelines are available from different institutions. Griseofulvin (FDA-approved for patients > 2 years of age) has been widely used, particularly for Microsporum canis infections. However, due to limited availability in many countries, terbinafine (FDA-approved for patients > 4 years of age) is now commonly used as first-line therapy, especially for Trichophyton species. Treatment typically lasts 4-6 weeks, and post-treatment cultures may be recommended to confirm mycologic cure.
Concerns about drug resistance have emerged, particularly for terbinafine-resistant dermatophytes linked to mutations in the squalene epoxidase enzyme. Resistance may be driven by limited antifungal availability and poor adherence to prolonged treatment regimens. While fluconazole and itraconazole are used off-label, growing evidence supports their effectiveness, although one large trial showed suboptimal cure rates with fluconazole.
Though systemic antifungals are generally safe, hepatotoxicity remains a concern, especially in patients with hepatic conditions or other comorbidities. Lab monitoring is advised for patients on prolonged or multiple therapies, or for those with coexisting conditions. The decision to conduct lab monitoring should be discussed with parents, balancing the very low risk of hepatotoxicity in healthy children against their comfort level.
An alternative to systemic therapy is photodynamic therapy (PDT), which has been reported as successful in treating tinea capitis infections, particularly in cases of T. mentagrophytes and M. canis. However, large-scale trials are needed to confirm PDT’s efficacy and safety.
In conclusion, children presenting with hair loss, scaling, and associated dark spots on the scalp should be evaluated for fungal infection. While trichoscopy can aid in diagnosis, fungal culture remains the gold standard for confirmation.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Rudnicka L et al. Hair shafts in trichoscopy: clues for diagnosis of hair and scalp diseases. Dermatol Clin. 2013 Oct;31(4):695-708, x. doi: 10.1016/j.det.2013.06.007.
Gupta AK et al. An update on tinea capitis in children. Pediatr Dermatol. 2024 Aug 7. doi: 10.1111/pde.15708.
Anna Waskiel-Burnat et al. Trichoscopy of tinea capitis: A systematic review. Dermatol Ther (Heidelb). 2020 Feb;10(1):43-52. doi: 10.1007/s13555-019-00350-1.
Given the trichoscopic findings, scrapings from the scaly areas were taken and revealed hyphae, confirming the diagnosis of tinea capitis. A fungal culture identified Trichophyton tonsurans as the causative organism.
Tinea capitis is the most common dermatophyte infection in children. Risk factors include participation in close-contact sports like wrestling or jiu-jitsu, attendance at daycare for younger children, African American hair care practices, pet ownership (particularly cats and rodents), and living in overcrowded conditions.
Diagnosis of tinea capitis requires a thorough clinical history to identify potential risk factors. On physical examination, patchy hair loss with associated scaling should raise suspicion for tinea capitis. Inflammatory signs, such as pustules and swelling, may suggest the presence of a kerion, further supporting the diagnosis. Although some practitioners use Wood’s lamp to help with diagnosis, its utility is limited. It detects fluorescence in Microsporum species (exothrix infections) but not in Trichophyton species (endothrix infections).
Trichoscopy can be a valuable tool when inflammation is minimal, and only hair loss and scaling are observed. Trichoscopic findings suggestive of tinea capitis include comma hairs, corkscrew hairs (as seen in this patient), Morse code-like hairs, zigzag hairs, bent hairs, block hairs, and i-hairs. Other common, though not characteristic, findings include broken hairs, black dots, perifollicular scaling, and diffuse scaling.
KOH (potassium hydroxide) analysis is another useful method for detecting fungal elements, though it does not identify the specific fungus and may not be available in all clinical settings. Mycologic culture remains the gold standard for diagnosing tinea capitis, though results can take 3-4 weeks. Newer diagnostic techniques, such as PCR analysis and MALDI-TOF/MS, offer more rapid identification of the causative organism.
The differential diagnosis includes:
- Seborrheic dermatitis, which presents with greasy, yellowish scales and itching, with trichoscopy showing twisted, coiled hairs and yellowish scaling.
- Psoriasis, which can mimic tinea capitis but presents with well-demarcated red plaques and silvery-white scales. Trichoscopy shows red dots and uniform scaling.
- Alopecia areata, which causes patchy hair loss without inflammation or scaling, with trichoscopic findings of exclamation mark hairs, black dots, and yellow dots.
- Trichotillomania, a hair-pulling disorder, which results in irregular patches of hair loss. Trichoscopy shows broken hairs of varying lengths, V-sign hairs, and flame-shaped residues at follicular openings.
Treatment of tinea capitis requires systemic antifungals and topical agents to prevent fungal spore spread. Several treatment guidelines are available from different institutions. Griseofulvin (FDA-approved for patients > 2 years of age) has been widely used, particularly for Microsporum canis infections. However, due to limited availability in many countries, terbinafine (FDA-approved for patients > 4 years of age) is now commonly used as first-line therapy, especially for Trichophyton species. Treatment typically lasts 4-6 weeks, and post-treatment cultures may be recommended to confirm mycologic cure.
Concerns about drug resistance have emerged, particularly for terbinafine-resistant dermatophytes linked to mutations in the squalene epoxidase enzyme. Resistance may be driven by limited antifungal availability and poor adherence to prolonged treatment regimens. While fluconazole and itraconazole are used off-label, growing evidence supports their effectiveness, although one large trial showed suboptimal cure rates with fluconazole.
Though systemic antifungals are generally safe, hepatotoxicity remains a concern, especially in patients with hepatic conditions or other comorbidities. Lab monitoring is advised for patients on prolonged or multiple therapies, or for those with coexisting conditions. The decision to conduct lab monitoring should be discussed with parents, balancing the very low risk of hepatotoxicity in healthy children against their comfort level.
An alternative to systemic therapy is photodynamic therapy (PDT), which has been reported as successful in treating tinea capitis infections, particularly in cases of T. mentagrophytes and M. canis. However, large-scale trials are needed to confirm PDT’s efficacy and safety.
In conclusion, children presenting with hair loss, scaling, and associated dark spots on the scalp should be evaluated for fungal infection. While trichoscopy can aid in diagnosis, fungal culture remains the gold standard for confirmation.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Rudnicka L et al. Hair shafts in trichoscopy: clues for diagnosis of hair and scalp diseases. Dermatol Clin. 2013 Oct;31(4):695-708, x. doi: 10.1016/j.det.2013.06.007.
Gupta AK et al. An update on tinea capitis in children. Pediatr Dermatol. 2024 Aug 7. doi: 10.1111/pde.15708.
Anna Waskiel-Burnat et al. Trichoscopy of tinea capitis: A systematic review. Dermatol Ther (Heidelb). 2020 Feb;10(1):43-52. doi: 10.1007/s13555-019-00350-1.
Given the trichoscopic findings, scrapings from the scaly areas were taken and revealed hyphae, confirming the diagnosis of tinea capitis. A fungal culture identified Trichophyton tonsurans as the causative organism.
Tinea capitis is the most common dermatophyte infection in children. Risk factors include participation in close-contact sports like wrestling or jiu-jitsu, attendance at daycare for younger children, African American hair care practices, pet ownership (particularly cats and rodents), and living in overcrowded conditions.
Diagnosis of tinea capitis requires a thorough clinical history to identify potential risk factors. On physical examination, patchy hair loss with associated scaling should raise suspicion for tinea capitis. Inflammatory signs, such as pustules and swelling, may suggest the presence of a kerion, further supporting the diagnosis. Although some practitioners use Wood’s lamp to help with diagnosis, its utility is limited. It detects fluorescence in Microsporum species (exothrix infections) but not in Trichophyton species (endothrix infections).
Trichoscopy can be a valuable tool when inflammation is minimal, and only hair loss and scaling are observed. Trichoscopic findings suggestive of tinea capitis include comma hairs, corkscrew hairs (as seen in this patient), Morse code-like hairs, zigzag hairs, bent hairs, block hairs, and i-hairs. Other common, though not characteristic, findings include broken hairs, black dots, perifollicular scaling, and diffuse scaling.
KOH (potassium hydroxide) analysis is another useful method for detecting fungal elements, though it does not identify the specific fungus and may not be available in all clinical settings. Mycologic culture remains the gold standard for diagnosing tinea capitis, though results can take 3-4 weeks. Newer diagnostic techniques, such as PCR analysis and MALDI-TOF/MS, offer more rapid identification of the causative organism.
The differential diagnosis includes:
- Seborrheic dermatitis, which presents with greasy, yellowish scales and itching, with trichoscopy showing twisted, coiled hairs and yellowish scaling.
- Psoriasis, which can mimic tinea capitis but presents with well-demarcated red plaques and silvery-white scales. Trichoscopy shows red dots and uniform scaling.
- Alopecia areata, which causes patchy hair loss without inflammation or scaling, with trichoscopic findings of exclamation mark hairs, black dots, and yellow dots.
- Trichotillomania, a hair-pulling disorder, which results in irregular patches of hair loss. Trichoscopy shows broken hairs of varying lengths, V-sign hairs, and flame-shaped residues at follicular openings.
Treatment of tinea capitis requires systemic antifungals and topical agents to prevent fungal spore spread. Several treatment guidelines are available from different institutions. Griseofulvin (FDA-approved for patients > 2 years of age) has been widely used, particularly for Microsporum canis infections. However, due to limited availability in many countries, terbinafine (FDA-approved for patients > 4 years of age) is now commonly used as first-line therapy, especially for Trichophyton species. Treatment typically lasts 4-6 weeks, and post-treatment cultures may be recommended to confirm mycologic cure.
Concerns about drug resistance have emerged, particularly for terbinafine-resistant dermatophytes linked to mutations in the squalene epoxidase enzyme. Resistance may be driven by limited antifungal availability and poor adherence to prolonged treatment regimens. While fluconazole and itraconazole are used off-label, growing evidence supports their effectiveness, although one large trial showed suboptimal cure rates with fluconazole.
Though systemic antifungals are generally safe, hepatotoxicity remains a concern, especially in patients with hepatic conditions or other comorbidities. Lab monitoring is advised for patients on prolonged or multiple therapies, or for those with coexisting conditions. The decision to conduct lab monitoring should be discussed with parents, balancing the very low risk of hepatotoxicity in healthy children against their comfort level.
An alternative to systemic therapy is photodynamic therapy (PDT), which has been reported as successful in treating tinea capitis infections, particularly in cases of T. mentagrophytes and M. canis. However, large-scale trials are needed to confirm PDT’s efficacy and safety.
In conclusion, children presenting with hair loss, scaling, and associated dark spots on the scalp should be evaluated for fungal infection. While trichoscopy can aid in diagnosis, fungal culture remains the gold standard for confirmation.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Rudnicka L et al. Hair shafts in trichoscopy: clues for diagnosis of hair and scalp diseases. Dermatol Clin. 2013 Oct;31(4):695-708, x. doi: 10.1016/j.det.2013.06.007.
Gupta AK et al. An update on tinea capitis in children. Pediatr Dermatol. 2024 Aug 7. doi: 10.1111/pde.15708.
Anna Waskiel-Burnat et al. Trichoscopy of tinea capitis: A systematic review. Dermatol Ther (Heidelb). 2020 Feb;10(1):43-52. doi: 10.1007/s13555-019-00350-1.
Pulsed Dye Laser a “Go-To Device” Option for Acne Treatment When Access to 1726-nm Lasers Is Limited
CARLSBAD, CALIF. — Lasers and energy-based treatments alone or in combination with medical therapy may improve outcomes for patients with moderate to severe acne, according to Arielle Kauvar, MD.
At the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium, Kauvar, director of New York Laser & Skin Care, New York City, highlighted several reasons why using lasers for acne is beneficial. “First, we know that topical therapy alone is often ineffective, and antibiotic treatment does not address the cause of acne and can alter the skin and gut microbiome,” she said. “Isotretinoin is highly effective, but there’s an increasing reluctance to use it. Lasers and energy devices are effective in treating acne and may also treat the post-inflammatory hyperpigmentation and scarring associated with it.”
The pathogenesis of acne is multifactorial, she continued, including a disruption of sebaceous gland activity, with overproduction and alteration of sebum and abnormal follicular keratinization. Acne also causes an imbalance of the skin microbiome, local inflammation, and activation of both innate and adaptive immunity.
“Many studies point to the fact that inflammation and immune system activation may actually be the primary event” of acne formation, said Kauvar, who is also a clinical professor of dermatology at New York University, New York City. “This persistent immune activation is also associated with scarring,” she noted. “So, are we off the mark in terms of trying to kill sebaceous glands? Should we be concentrating on anti-inflammatory approaches?”
AviClear became the first 1726-nm laser cleared by the US Food and Drug Administration (FDA) for the treatment of mild to severe acne vulgaris in 2022, followed a few months later with the FDA clearance of another 1726-nm laser, the Accure Acne Laser System in November 2022. These lasers cause selective photothermolysis of sebaceous glands, but according to Kauvar, “access to these devices is somewhat limited at this time.”
What is available includes her go-to device, the pulsed dye laser (PDL), which has been widely studied and shown in a systematic review and meta-analysis of studies to be effective for acne. The PDL “targets dermal blood vessels facilitating inflammation, upregulates TGF-beta, and inhibits CD4+ T cell-mediated inflammation,” she said. “It can also treat PIH [post-inflammatory hyperpigmentation] and may be helpful in scar prevention.”
In an abstract presented at The American Society for Laser Medicine and Surgery (ASLMS) 2024 annual meeting, Kauvar and colleagues conducted a real-world study of PDL therapy in 15 adult women with recalcitrant acne who were maintained on their medical treatment regimen. Their mean age was 27 years, and they had skin types II-IV; they underwent four monthly PDL treatments with follow-up at 1 and 3 months. At each visit, the researchers took digital photographs and counted inflammatory acne lesions, non-inflammatory acne lesions, and post-inflammatory pigment alteration (PIPA) lesions.
The main outcomes of interest were the investigator global assessment (IGA) scores at the 1- and 3-month follow-up visits. Kauvar and colleagues observed a significant improvement in IGA scores at the 1- and 3-month follow-up visits (P < .05), with an average decrease of 1.8 and 1.6 points in the acne severity scale, respectively, from a baseline score of 3.4. By the 3-month follow-up visits, counts of inflammatory and non-inflammatory lesions decreased significantly (P < .05), and 61% of study participants showed a decrease in the PIPA count. No adverse events occurred.
Kauvar disclosed that she has conducted research for Candela, Lumenis, and Sofwave, and is an adviser to Acclaro.
A version of this article first appeared on Medscape.com.
CARLSBAD, CALIF. — Lasers and energy-based treatments alone or in combination with medical therapy may improve outcomes for patients with moderate to severe acne, according to Arielle Kauvar, MD.
At the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium, Kauvar, director of New York Laser & Skin Care, New York City, highlighted several reasons why using lasers for acne is beneficial. “First, we know that topical therapy alone is often ineffective, and antibiotic treatment does not address the cause of acne and can alter the skin and gut microbiome,” she said. “Isotretinoin is highly effective, but there’s an increasing reluctance to use it. Lasers and energy devices are effective in treating acne and may also treat the post-inflammatory hyperpigmentation and scarring associated with it.”
The pathogenesis of acne is multifactorial, she continued, including a disruption of sebaceous gland activity, with overproduction and alteration of sebum and abnormal follicular keratinization. Acne also causes an imbalance of the skin microbiome, local inflammation, and activation of both innate and adaptive immunity.
“Many studies point to the fact that inflammation and immune system activation may actually be the primary event” of acne formation, said Kauvar, who is also a clinical professor of dermatology at New York University, New York City. “This persistent immune activation is also associated with scarring,” she noted. “So, are we off the mark in terms of trying to kill sebaceous glands? Should we be concentrating on anti-inflammatory approaches?”
AviClear became the first 1726-nm laser cleared by the US Food and Drug Administration (FDA) for the treatment of mild to severe acne vulgaris in 2022, followed a few months later with the FDA clearance of another 1726-nm laser, the Accure Acne Laser System in November 2022. These lasers cause selective photothermolysis of sebaceous glands, but according to Kauvar, “access to these devices is somewhat limited at this time.”
What is available includes her go-to device, the pulsed dye laser (PDL), which has been widely studied and shown in a systematic review and meta-analysis of studies to be effective for acne. The PDL “targets dermal blood vessels facilitating inflammation, upregulates TGF-beta, and inhibits CD4+ T cell-mediated inflammation,” she said. “It can also treat PIH [post-inflammatory hyperpigmentation] and may be helpful in scar prevention.”
In an abstract presented at The American Society for Laser Medicine and Surgery (ASLMS) 2024 annual meeting, Kauvar and colleagues conducted a real-world study of PDL therapy in 15 adult women with recalcitrant acne who were maintained on their medical treatment regimen. Their mean age was 27 years, and they had skin types II-IV; they underwent four monthly PDL treatments with follow-up at 1 and 3 months. At each visit, the researchers took digital photographs and counted inflammatory acne lesions, non-inflammatory acne lesions, and post-inflammatory pigment alteration (PIPA) lesions.
The main outcomes of interest were the investigator global assessment (IGA) scores at the 1- and 3-month follow-up visits. Kauvar and colleagues observed a significant improvement in IGA scores at the 1- and 3-month follow-up visits (P < .05), with an average decrease of 1.8 and 1.6 points in the acne severity scale, respectively, from a baseline score of 3.4. By the 3-month follow-up visits, counts of inflammatory and non-inflammatory lesions decreased significantly (P < .05), and 61% of study participants showed a decrease in the PIPA count. No adverse events occurred.
Kauvar disclosed that she has conducted research for Candela, Lumenis, and Sofwave, and is an adviser to Acclaro.
A version of this article first appeared on Medscape.com.
CARLSBAD, CALIF. — Lasers and energy-based treatments alone or in combination with medical therapy may improve outcomes for patients with moderate to severe acne, according to Arielle Kauvar, MD.
At the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium, Kauvar, director of New York Laser & Skin Care, New York City, highlighted several reasons why using lasers for acne is beneficial. “First, we know that topical therapy alone is often ineffective, and antibiotic treatment does not address the cause of acne and can alter the skin and gut microbiome,” she said. “Isotretinoin is highly effective, but there’s an increasing reluctance to use it. Lasers and energy devices are effective in treating acne and may also treat the post-inflammatory hyperpigmentation and scarring associated with it.”
The pathogenesis of acne is multifactorial, she continued, including a disruption of sebaceous gland activity, with overproduction and alteration of sebum and abnormal follicular keratinization. Acne also causes an imbalance of the skin microbiome, local inflammation, and activation of both innate and adaptive immunity.
“Many studies point to the fact that inflammation and immune system activation may actually be the primary event” of acne formation, said Kauvar, who is also a clinical professor of dermatology at New York University, New York City. “This persistent immune activation is also associated with scarring,” she noted. “So, are we off the mark in terms of trying to kill sebaceous glands? Should we be concentrating on anti-inflammatory approaches?”
AviClear became the first 1726-nm laser cleared by the US Food and Drug Administration (FDA) for the treatment of mild to severe acne vulgaris in 2022, followed a few months later with the FDA clearance of another 1726-nm laser, the Accure Acne Laser System in November 2022. These lasers cause selective photothermolysis of sebaceous glands, but according to Kauvar, “access to these devices is somewhat limited at this time.”
What is available includes her go-to device, the pulsed dye laser (PDL), which has been widely studied and shown in a systematic review and meta-analysis of studies to be effective for acne. The PDL “targets dermal blood vessels facilitating inflammation, upregulates TGF-beta, and inhibits CD4+ T cell-mediated inflammation,” she said. “It can also treat PIH [post-inflammatory hyperpigmentation] and may be helpful in scar prevention.”
In an abstract presented at The American Society for Laser Medicine and Surgery (ASLMS) 2024 annual meeting, Kauvar and colleagues conducted a real-world study of PDL therapy in 15 adult women with recalcitrant acne who were maintained on their medical treatment regimen. Their mean age was 27 years, and they had skin types II-IV; they underwent four monthly PDL treatments with follow-up at 1 and 3 months. At each visit, the researchers took digital photographs and counted inflammatory acne lesions, non-inflammatory acne lesions, and post-inflammatory pigment alteration (PIPA) lesions.
The main outcomes of interest were the investigator global assessment (IGA) scores at the 1- and 3-month follow-up visits. Kauvar and colleagues observed a significant improvement in IGA scores at the 1- and 3-month follow-up visits (P < .05), with an average decrease of 1.8 and 1.6 points in the acne severity scale, respectively, from a baseline score of 3.4. By the 3-month follow-up visits, counts of inflammatory and non-inflammatory lesions decreased significantly (P < .05), and 61% of study participants showed a decrease in the PIPA count. No adverse events occurred.
Kauvar disclosed that she has conducted research for Candela, Lumenis, and Sofwave, and is an adviser to Acclaro.
A version of this article first appeared on Medscape.com.
Mycosis Fungoides: Measured Approach Key to Treatment
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
FROM PDA 2024
Down Syndrome: Several Cutaneous Conditions Common, Study Finds
TOPLINE:
(DS) in a 10-year retrospective study.
METHODOLOGY:
- Researchers conducted a multicenter retrospective study of 1529 patients with DS from eight outpatient dermatology clinics in the United States and Canada between 2011 and 2021.
- In total, 50.8% of patients were children (0-12 years), 25.2% were adolescents (13-17 years), and 24% were adults (≥ 18 years).
- The researchers evaluated skin conditions in the patients.
TAKEAWAY:
- Eczematous dermatitis was the most common diagnosis, affecting 26% of patients, followed by folliculitis (19.3%) and seborrheic dermatitis (15.6%). Dermatophyte infections were diagnosed in 13%.
- Alopecia areata was the most common autoimmune skin condition, diagnosed in 178 patients (11.6%); 135 (75.8%) were children. Vitiligo was diagnosed in 66 patients (4.3%).
- The most common cutaneous infections were onychomycosis (5.9%), tinea pedis (5%), and verruca vulgaris/other viral warts (5%).
- High-risk medication use was reported in 4.3% of patients; acne vulgaris, hidradenitis suppurativa, and eczematous dermatitis were the most common associated conditions with such medications.
IN PRACTICE:
“Children, adolescents, and adults with DS are most often found to have eczematous, adnexal, and autoimmune skin conditions at outpatient dermatology visits,” the authors wrote. Their findings, they added, “offer valuable insights for clinicians and researchers, aiding in the improved prioritization of screening, diagnosis, and management, as well as facilitating both basic science and clinical research into prevalent skin conditions in individuals with DS.”
SOURCE:
The study was led by Tasya Rakasiwi, of the Department of Dermatology, Dartmouth Health, Manchester, New Hampshire, and was published online in Pediatric Dermatology.
LIMITATIONS:
Over 50% of the patients were children, potentially resulting in bias toward pediatric diagnoses and younger ages of presentation. Race, ethnicity, and sociodemographic factors were not captured, limiting the generalizability of the findings. Medical codes often do not capture disease phenotype or severity, and the manual conversion of International Classification of Diseases (ICD) 9 to ICD-10 codes may introduce potential conversion errors.
DISCLOSURES:
The study was supported by the Pediatric Dermatology Research Alliance. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
(DS) in a 10-year retrospective study.
METHODOLOGY:
- Researchers conducted a multicenter retrospective study of 1529 patients with DS from eight outpatient dermatology clinics in the United States and Canada between 2011 and 2021.
- In total, 50.8% of patients were children (0-12 years), 25.2% were adolescents (13-17 years), and 24% were adults (≥ 18 years).
- The researchers evaluated skin conditions in the patients.
TAKEAWAY:
- Eczematous dermatitis was the most common diagnosis, affecting 26% of patients, followed by folliculitis (19.3%) and seborrheic dermatitis (15.6%). Dermatophyte infections were diagnosed in 13%.
- Alopecia areata was the most common autoimmune skin condition, diagnosed in 178 patients (11.6%); 135 (75.8%) were children. Vitiligo was diagnosed in 66 patients (4.3%).
- The most common cutaneous infections were onychomycosis (5.9%), tinea pedis (5%), and verruca vulgaris/other viral warts (5%).
- High-risk medication use was reported in 4.3% of patients; acne vulgaris, hidradenitis suppurativa, and eczematous dermatitis were the most common associated conditions with such medications.
IN PRACTICE:
“Children, adolescents, and adults with DS are most often found to have eczematous, adnexal, and autoimmune skin conditions at outpatient dermatology visits,” the authors wrote. Their findings, they added, “offer valuable insights for clinicians and researchers, aiding in the improved prioritization of screening, diagnosis, and management, as well as facilitating both basic science and clinical research into prevalent skin conditions in individuals with DS.”
SOURCE:
The study was led by Tasya Rakasiwi, of the Department of Dermatology, Dartmouth Health, Manchester, New Hampshire, and was published online in Pediatric Dermatology.
LIMITATIONS:
Over 50% of the patients were children, potentially resulting in bias toward pediatric diagnoses and younger ages of presentation. Race, ethnicity, and sociodemographic factors were not captured, limiting the generalizability of the findings. Medical codes often do not capture disease phenotype or severity, and the manual conversion of International Classification of Diseases (ICD) 9 to ICD-10 codes may introduce potential conversion errors.
DISCLOSURES:
The study was supported by the Pediatric Dermatology Research Alliance. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
(DS) in a 10-year retrospective study.
METHODOLOGY:
- Researchers conducted a multicenter retrospective study of 1529 patients with DS from eight outpatient dermatology clinics in the United States and Canada between 2011 and 2021.
- In total, 50.8% of patients were children (0-12 years), 25.2% were adolescents (13-17 years), and 24% were adults (≥ 18 years).
- The researchers evaluated skin conditions in the patients.
TAKEAWAY:
- Eczematous dermatitis was the most common diagnosis, affecting 26% of patients, followed by folliculitis (19.3%) and seborrheic dermatitis (15.6%). Dermatophyte infections were diagnosed in 13%.
- Alopecia areata was the most common autoimmune skin condition, diagnosed in 178 patients (11.6%); 135 (75.8%) were children. Vitiligo was diagnosed in 66 patients (4.3%).
- The most common cutaneous infections were onychomycosis (5.9%), tinea pedis (5%), and verruca vulgaris/other viral warts (5%).
- High-risk medication use was reported in 4.3% of patients; acne vulgaris, hidradenitis suppurativa, and eczematous dermatitis were the most common associated conditions with such medications.
IN PRACTICE:
“Children, adolescents, and adults with DS are most often found to have eczematous, adnexal, and autoimmune skin conditions at outpatient dermatology visits,” the authors wrote. Their findings, they added, “offer valuable insights for clinicians and researchers, aiding in the improved prioritization of screening, diagnosis, and management, as well as facilitating both basic science and clinical research into prevalent skin conditions in individuals with DS.”
SOURCE:
The study was led by Tasya Rakasiwi, of the Department of Dermatology, Dartmouth Health, Manchester, New Hampshire, and was published online in Pediatric Dermatology.
LIMITATIONS:
Over 50% of the patients were children, potentially resulting in bias toward pediatric diagnoses and younger ages of presentation. Race, ethnicity, and sociodemographic factors were not captured, limiting the generalizability of the findings. Medical codes often do not capture disease phenotype or severity, and the manual conversion of International Classification of Diseases (ICD) 9 to ICD-10 codes may introduce potential conversion errors.
DISCLOSURES:
The study was supported by the Pediatric Dermatology Research Alliance. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Study Supports Efficacy of Home-Based Phototherapy for Psoriasis
TOPLINE:
study.
METHODOLOGY:
- The pragmatic, investigator-initiated, open-label, noninferiority, randomized trial compared the effectiveness of 12 weeks of treatment with narrow-band ultraviolet B phototherapy administered at home (n = 393) vs at the doctor’s office (n = 390).
- Overall, 783 patients with plaque or guttate psoriasis (mean age, 48 years; 48% women) were enrolled at 42 academic and private clinical dermatology practices in the United States from March 1, 2019, to December 4, 2023, and were followed up through June 2024. At baseline, the mean Physician Global Assessment (PGA) and the mean Dermatology Life Quality Index (DLQI) scores were 2.7 and 12.2, respectively.
- The two co-primary endpoints were a PGA score ≤ 1 indicating clear or almost clear skin and a DLQI score ≤ 5.
TAKEAWAY:
- At 12 weeks, a PGA score ≤ 1 was achieved in 32.8% of patients using home-based phototherapy and in 25.6% of those who received office-based phototherapy (P < .001).
- At 12 weeks, a DLQI score ≤ 5 was achieved in 52.4% and 33.6% of home- and office-treated patients, respectively (P < .001).
- Similar benefits were seen across all Fitzpatrick skin types.
- A higher percentage of patients were adherent to home-based (51.4%) vs office-based (15.9%) phototherapy (P < .001).
IN PRACTICE:
“These data support the use of home phototherapy as a first-line treatment option for psoriasis,” and “efforts are needed to make home and office phototherapy more available to patients,” said the study’s lead author.
SOURCE:
Joel M. Gelfand, MD, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania, Philadelphia, presented the findings at the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis meeting during the annual meeting of the European Academy of Dermatology and Venereology, with simultaneous publication in JAMA Dermatology.
LIMITATIONS:
This was an open-label trial and because of its pragmatic design, outcome data were missing. The cost of the home-based phototherapy equipment used in the study was $6040.88, which was mostly covered by Medicare, but direct costs to patients may have varied depending on their insurance plan.
DISCLOSURES:
The Patient-Centered Outcomes Research Institute funded the study. Daavlin provided and shipped machines for home-based phototherapy to patients at no cost. Dr. Gelfand disclosed serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, and other companies. The full list of author disclosures can be found in the published study.
A version of this article first appeared on Medscape.com.
TOPLINE:
study.
METHODOLOGY:
- The pragmatic, investigator-initiated, open-label, noninferiority, randomized trial compared the effectiveness of 12 weeks of treatment with narrow-band ultraviolet B phototherapy administered at home (n = 393) vs at the doctor’s office (n = 390).
- Overall, 783 patients with plaque or guttate psoriasis (mean age, 48 years; 48% women) were enrolled at 42 academic and private clinical dermatology practices in the United States from March 1, 2019, to December 4, 2023, and were followed up through June 2024. At baseline, the mean Physician Global Assessment (PGA) and the mean Dermatology Life Quality Index (DLQI) scores were 2.7 and 12.2, respectively.
- The two co-primary endpoints were a PGA score ≤ 1 indicating clear or almost clear skin and a DLQI score ≤ 5.
TAKEAWAY:
- At 12 weeks, a PGA score ≤ 1 was achieved in 32.8% of patients using home-based phototherapy and in 25.6% of those who received office-based phototherapy (P < .001).
- At 12 weeks, a DLQI score ≤ 5 was achieved in 52.4% and 33.6% of home- and office-treated patients, respectively (P < .001).
- Similar benefits were seen across all Fitzpatrick skin types.
- A higher percentage of patients were adherent to home-based (51.4%) vs office-based (15.9%) phototherapy (P < .001).
IN PRACTICE:
“These data support the use of home phototherapy as a first-line treatment option for psoriasis,” and “efforts are needed to make home and office phototherapy more available to patients,” said the study’s lead author.
SOURCE:
Joel M. Gelfand, MD, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania, Philadelphia, presented the findings at the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis meeting during the annual meeting of the European Academy of Dermatology and Venereology, with simultaneous publication in JAMA Dermatology.
LIMITATIONS:
This was an open-label trial and because of its pragmatic design, outcome data were missing. The cost of the home-based phototherapy equipment used in the study was $6040.88, which was mostly covered by Medicare, but direct costs to patients may have varied depending on their insurance plan.
DISCLOSURES:
The Patient-Centered Outcomes Research Institute funded the study. Daavlin provided and shipped machines for home-based phototherapy to patients at no cost. Dr. Gelfand disclosed serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, and other companies. The full list of author disclosures can be found in the published study.
A version of this article first appeared on Medscape.com.
TOPLINE:
study.
METHODOLOGY:
- The pragmatic, investigator-initiated, open-label, noninferiority, randomized trial compared the effectiveness of 12 weeks of treatment with narrow-band ultraviolet B phototherapy administered at home (n = 393) vs at the doctor’s office (n = 390).
- Overall, 783 patients with plaque or guttate psoriasis (mean age, 48 years; 48% women) were enrolled at 42 academic and private clinical dermatology practices in the United States from March 1, 2019, to December 4, 2023, and were followed up through June 2024. At baseline, the mean Physician Global Assessment (PGA) and the mean Dermatology Life Quality Index (DLQI) scores were 2.7 and 12.2, respectively.
- The two co-primary endpoints were a PGA score ≤ 1 indicating clear or almost clear skin and a DLQI score ≤ 5.
TAKEAWAY:
- At 12 weeks, a PGA score ≤ 1 was achieved in 32.8% of patients using home-based phototherapy and in 25.6% of those who received office-based phototherapy (P < .001).
- At 12 weeks, a DLQI score ≤ 5 was achieved in 52.4% and 33.6% of home- and office-treated patients, respectively (P < .001).
- Similar benefits were seen across all Fitzpatrick skin types.
- A higher percentage of patients were adherent to home-based (51.4%) vs office-based (15.9%) phototherapy (P < .001).
IN PRACTICE:
“These data support the use of home phototherapy as a first-line treatment option for psoriasis,” and “efforts are needed to make home and office phototherapy more available to patients,” said the study’s lead author.
SOURCE:
Joel M. Gelfand, MD, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania, Philadelphia, presented the findings at the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis meeting during the annual meeting of the European Academy of Dermatology and Venereology, with simultaneous publication in JAMA Dermatology.
LIMITATIONS:
This was an open-label trial and because of its pragmatic design, outcome data were missing. The cost of the home-based phototherapy equipment used in the study was $6040.88, which was mostly covered by Medicare, but direct costs to patients may have varied depending on their insurance plan.
DISCLOSURES:
The Patient-Centered Outcomes Research Institute funded the study. Daavlin provided and shipped machines for home-based phototherapy to patients at no cost. Dr. Gelfand disclosed serving as a consultant for AbbVie, Artax, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, and other companies. The full list of author disclosures can be found in the published study.
A version of this article first appeared on Medscape.com.
Aspects of the Skin Microbiome Remain Elusive
SAN DIEGO — Although it has been known for several years that
In one review of the topic, researchers from the National Institutes of Health wrote that the skin is composed of 1.8 million diverse habitats with an abundance of folds, invaginations, and specialized niches that support a wide range of microorganisms. “Many of these microorganisms are harmless and, in some cases, provide vital functions for us to live and they have not evolved over time,” Jill S. Waibel, MD, medical director of the Miami Dermatology and Laser Institute, said at the annual Masters of Aesthetics Symposium.
“This is complex ecosystem that we don’t really talk about,” she said. “There is wide topographical distribution of bacteria on skin sites. The bacteria we have on our head and neck area is different from that on our feet. There is also a lot of interpersonal variation of the skin microbiome, so one person may have a lot of one type of bacteria and not as much of another.”
A Shield From Foreign Pathogens
At its core, Dr. Waibel continued, the skin microbiome functions as an interface between the human body and the environment, a physical barrier that prevents the invasion of foreign pathogens. The skin also provides a home to commensal microbiota. She likened the skin’s landscape to that of the tundra: “It’s desiccated, has poor nutrients, and it’s very acidic, thus pathogens have a hard time living on it,” she said. “However, our skin microorganisms have adapted to utilize the sparse nutrients available on the skin. That’s why I tell my patients, ‘don’t use a sugar scrub because you’re potentially feeding these bad bacteria.’ ”
According to more recent research, the skin microbiota in healthy adults remains stable over time, despite environmental perturbations, and they have important roles in educating the innate and adaptive arms of the cutaneous immune system. “Some skin diseases are associated with an altered microbial state: dysbiosis,” said Dr. Waibel, subsection chief of dermatology at Baptist Health South Florida, Miami Beach. “Reversion of this may help prevent or treat the disease.”
She cited the following factors that influence the skin microbiome:
- Genetics affects the skin microbiome considerably. Individuals with autoimmune predispositions have different microbiota compared with those who don’t.
- Climate, pollution, and hygiene practices the other influencing factors. “Even clothing can impact the microbiome, by causing the transfer of microorganisms,” she said.
- Age and hormonal changes (particularly during puberty) and senescence alter the microbial landscape.
- Systemic health conditions such as diabetes mellitus and irritable bowel disease, as well as cutaneous conditions like psoriasis and atopic dermatitis can also disrupt the skin microbiome.
Ingredients contained in soaps, antibiotics, and cosmetics can also cause skin dysbiosis, Dr. Waibel said. However, the integrity of the skin’s microbiome following dermatological procedures such as excisions, dermabrasion, laser therapy, and other physical procedures is less understood, according to a recent review of the topic. Phototherapy appears to be the most extensively studied, “and shows an increase in microbial diversity post-treatment,” she said. “Light treatments have been found to kill bacteria by inducing DNA damage. More studies need to be performed on specific wavelengths of light used, conditions being treated and individual patient differences.”
According to the review’s authors, no change in the microbiome was observed in studies of debridement. “That was surprising, as it is a method to remove unhealthy tissue that often contains pathogenic bacteria,” Dr. Waibel said. “The big take-home message is that we need more research.”
Dr. Waibel disclosed that she has conducted clinical trials for several device and pharmaceutical companies.
A version of this article first appeared on Medscape.com.
SAN DIEGO — Although it has been known for several years that
In one review of the topic, researchers from the National Institutes of Health wrote that the skin is composed of 1.8 million diverse habitats with an abundance of folds, invaginations, and specialized niches that support a wide range of microorganisms. “Many of these microorganisms are harmless and, in some cases, provide vital functions for us to live and they have not evolved over time,” Jill S. Waibel, MD, medical director of the Miami Dermatology and Laser Institute, said at the annual Masters of Aesthetics Symposium.
“This is complex ecosystem that we don’t really talk about,” she said. “There is wide topographical distribution of bacteria on skin sites. The bacteria we have on our head and neck area is different from that on our feet. There is also a lot of interpersonal variation of the skin microbiome, so one person may have a lot of one type of bacteria and not as much of another.”
A Shield From Foreign Pathogens
At its core, Dr. Waibel continued, the skin microbiome functions as an interface between the human body and the environment, a physical barrier that prevents the invasion of foreign pathogens. The skin also provides a home to commensal microbiota. She likened the skin’s landscape to that of the tundra: “It’s desiccated, has poor nutrients, and it’s very acidic, thus pathogens have a hard time living on it,” she said. “However, our skin microorganisms have adapted to utilize the sparse nutrients available on the skin. That’s why I tell my patients, ‘don’t use a sugar scrub because you’re potentially feeding these bad bacteria.’ ”
According to more recent research, the skin microbiota in healthy adults remains stable over time, despite environmental perturbations, and they have important roles in educating the innate and adaptive arms of the cutaneous immune system. “Some skin diseases are associated with an altered microbial state: dysbiosis,” said Dr. Waibel, subsection chief of dermatology at Baptist Health South Florida, Miami Beach. “Reversion of this may help prevent or treat the disease.”
She cited the following factors that influence the skin microbiome:
- Genetics affects the skin microbiome considerably. Individuals with autoimmune predispositions have different microbiota compared with those who don’t.
- Climate, pollution, and hygiene practices the other influencing factors. “Even clothing can impact the microbiome, by causing the transfer of microorganisms,” she said.
- Age and hormonal changes (particularly during puberty) and senescence alter the microbial landscape.
- Systemic health conditions such as diabetes mellitus and irritable bowel disease, as well as cutaneous conditions like psoriasis and atopic dermatitis can also disrupt the skin microbiome.
Ingredients contained in soaps, antibiotics, and cosmetics can also cause skin dysbiosis, Dr. Waibel said. However, the integrity of the skin’s microbiome following dermatological procedures such as excisions, dermabrasion, laser therapy, and other physical procedures is less understood, according to a recent review of the topic. Phototherapy appears to be the most extensively studied, “and shows an increase in microbial diversity post-treatment,” she said. “Light treatments have been found to kill bacteria by inducing DNA damage. More studies need to be performed on specific wavelengths of light used, conditions being treated and individual patient differences.”
According to the review’s authors, no change in the microbiome was observed in studies of debridement. “That was surprising, as it is a method to remove unhealthy tissue that often contains pathogenic bacteria,” Dr. Waibel said. “The big take-home message is that we need more research.”
Dr. Waibel disclosed that she has conducted clinical trials for several device and pharmaceutical companies.
A version of this article first appeared on Medscape.com.
SAN DIEGO — Although it has been known for several years that
In one review of the topic, researchers from the National Institutes of Health wrote that the skin is composed of 1.8 million diverse habitats with an abundance of folds, invaginations, and specialized niches that support a wide range of microorganisms. “Many of these microorganisms are harmless and, in some cases, provide vital functions for us to live and they have not evolved over time,” Jill S. Waibel, MD, medical director of the Miami Dermatology and Laser Institute, said at the annual Masters of Aesthetics Symposium.
“This is complex ecosystem that we don’t really talk about,” she said. “There is wide topographical distribution of bacteria on skin sites. The bacteria we have on our head and neck area is different from that on our feet. There is also a lot of interpersonal variation of the skin microbiome, so one person may have a lot of one type of bacteria and not as much of another.”
A Shield From Foreign Pathogens
At its core, Dr. Waibel continued, the skin microbiome functions as an interface between the human body and the environment, a physical barrier that prevents the invasion of foreign pathogens. The skin also provides a home to commensal microbiota. She likened the skin’s landscape to that of the tundra: “It’s desiccated, has poor nutrients, and it’s very acidic, thus pathogens have a hard time living on it,” she said. “However, our skin microorganisms have adapted to utilize the sparse nutrients available on the skin. That’s why I tell my patients, ‘don’t use a sugar scrub because you’re potentially feeding these bad bacteria.’ ”
According to more recent research, the skin microbiota in healthy adults remains stable over time, despite environmental perturbations, and they have important roles in educating the innate and adaptive arms of the cutaneous immune system. “Some skin diseases are associated with an altered microbial state: dysbiosis,” said Dr. Waibel, subsection chief of dermatology at Baptist Health South Florida, Miami Beach. “Reversion of this may help prevent or treat the disease.”
She cited the following factors that influence the skin microbiome:
- Genetics affects the skin microbiome considerably. Individuals with autoimmune predispositions have different microbiota compared with those who don’t.
- Climate, pollution, and hygiene practices the other influencing factors. “Even clothing can impact the microbiome, by causing the transfer of microorganisms,” she said.
- Age and hormonal changes (particularly during puberty) and senescence alter the microbial landscape.
- Systemic health conditions such as diabetes mellitus and irritable bowel disease, as well as cutaneous conditions like psoriasis and atopic dermatitis can also disrupt the skin microbiome.
Ingredients contained in soaps, antibiotics, and cosmetics can also cause skin dysbiosis, Dr. Waibel said. However, the integrity of the skin’s microbiome following dermatological procedures such as excisions, dermabrasion, laser therapy, and other physical procedures is less understood, according to a recent review of the topic. Phototherapy appears to be the most extensively studied, “and shows an increase in microbial diversity post-treatment,” she said. “Light treatments have been found to kill bacteria by inducing DNA damage. More studies need to be performed on specific wavelengths of light used, conditions being treated and individual patient differences.”
According to the review’s authors, no change in the microbiome was observed in studies of debridement. “That was surprising, as it is a method to remove unhealthy tissue that often contains pathogenic bacteria,” Dr. Waibel said. “The big take-home message is that we need more research.”
Dr. Waibel disclosed that she has conducted clinical trials for several device and pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE 2024 MASTERS OF AESTHETICS SYMPOSIUM
Childhood-Onset Atopic Dermatitis Adds Burden in Adulthood
AMSTERDAM — There is a mountain of evidence that atopic dermatitis (AD) exerts a large negative impact on quality of life, but a unique study with
These data, drawn from the ambitious Scars of Life (SOL) project, “suggest that childhood AD persisting into adulthood is its own phenotype,” reported Jonathan I. Silverberg, MD, PhD, director of clinical research, Department of Dermatology, George Washington University, Washington, DC.
One reasonable message from these data is that the failure to achieve adequate control of AD in children, whether by a late start of systemic agents or other reasons, results in a greater lifetime burden of disease when the burden beyond physical symptoms is measured, according to Dr. Silverberg.
More Than 30,000 From Five Continents Participated
In the SOL project, which was designed to analyze how the age of AD onset affects the severity of symptoms and quality of life, completed questionnaires were collected from 30,801 individuals in 27 countries on five continents. The questions, which elicited data to measure the burden of AD, were developed in association with several professional and patient associations with an interest in AD, including the National Eczema Association.
The SOL project has produced an enormous amount of data in four distinct groups, but Dr. Silverberg, speaking in a late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology, focused on a comparison between the 2875 participants who had AD in childhood that has persisted into adulthood and the 7383 adults with adult-onset AD. Data from the other two subsets in SOL — AD in childhood but not in adulthood and no AD in either phase of life — are expected to fuel an extended series of publications.
In the two groups, baseline characteristics were similar with about 60% reporting moderate to severe symptoms and a median age of about 37 years. The proportion of women was 61% in both groups.
Using the PUSH-D questionnaire, which Dr. Silverberg described as a validated tool for gauging a sense of stigmatization, the greater burden of AD was remarkably consistent for those with childhood-onset AD vs adult-onset AD. With higher scores representing a greater sense of stigmatization, the differences in the overall score (23.0 vs 18.1; P < .0001) were highly significant as was every other domain evaluated.
For all five social behavior domains, such as avoiding contact in public and wariness of approaching people spontaneously, having AD onset in childhood persisting into adulthood produced significantly higher scores than having AD onset in adulthood, with no exceptions (P < .001 for all).
AD From Childhood Consistently Results in Worse Outcomes
Providing examples for some of the other 12 domains, Dr. Silverberg maintained that feelings of shame and psychological discomfort were always greater in adults with AD persistent since childhood vs AD starting in adulthood. The P values for these outcomes, such as experiencing bias at work or reporting a sense that others avoided them, were typically highly significant (P < .001).
Compared with those whose AD started in adulthood, “adults with atopic eczema that started during childhood have significantly more difficulties in their life, including occupational relationships, daily life, personal life, and partner or family relationships,” Dr. Silverberg reported.
He said that the data were controlled for multiple confounders, particularly greater severity of AD. He acknowledged that childhood onset might be considered a surrogate for more severe disease, but the data were controlled for this possibility.
Despite the fact that there are “thousands of studies across all age groups showing the burden of AD,” Dr. Silverberg considers these data to be unique by emphasizing the burden of chronicity rather than the impact of AD in any single moment in time.
For those with chronic AD from childhood, “the effect is not just on physical health but a deep negative influence on psychological and social aspects of life,” Dr. Silverberg said, suggesting that the independent effects of chronicity might be worth studying across other dermatologic diseases.
“Regulatory agencies focus on what you can do in that moment of time, losing the bigger picture of how patients are affected chronically,” he said, adding that this is an area of clinical research that should be further explored.
What the data further suggest “is that the earlier we intervene, the more likely patients will do better long term,” he said.
Data Provide Evidence of Systemic Therapy in Kids
For Gudrun Ratzinger, MD, of the Department of Dermatology and Venerology at the Medical University of Innsbruck in Austria, these are valuable data.
“When I prescribe systemic therapies to children, I often get resistance from the healthcare system and even other colleagues,” said Dr. Ratzinger, who was asked to comment on the results. “We are at a teaching hospital, but I often find that when patients return to their home physician, the systemic therapies are stopped.”
In her own practice, she believes the most effective therapies should be introduced in children and adults when complete control is not achieved on first-line drugs. “These data are very helpful for me in explaining to others the importance of effective treatment of atopic dermatitis in children,” she said.
Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies, including those that make drugs for AD. Dr. Ratzinger reported financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Eli Lilly, Janssen, Leo Pharma, Novartis, Pelpharma, Pfizer, and UCB.
A version of this article first appeared on Medscape.com.
AMSTERDAM — There is a mountain of evidence that atopic dermatitis (AD) exerts a large negative impact on quality of life, but a unique study with
These data, drawn from the ambitious Scars of Life (SOL) project, “suggest that childhood AD persisting into adulthood is its own phenotype,” reported Jonathan I. Silverberg, MD, PhD, director of clinical research, Department of Dermatology, George Washington University, Washington, DC.
One reasonable message from these data is that the failure to achieve adequate control of AD in children, whether by a late start of systemic agents or other reasons, results in a greater lifetime burden of disease when the burden beyond physical symptoms is measured, according to Dr. Silverberg.
More Than 30,000 From Five Continents Participated
In the SOL project, which was designed to analyze how the age of AD onset affects the severity of symptoms and quality of life, completed questionnaires were collected from 30,801 individuals in 27 countries on five continents. The questions, which elicited data to measure the burden of AD, were developed in association with several professional and patient associations with an interest in AD, including the National Eczema Association.
The SOL project has produced an enormous amount of data in four distinct groups, but Dr. Silverberg, speaking in a late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology, focused on a comparison between the 2875 participants who had AD in childhood that has persisted into adulthood and the 7383 adults with adult-onset AD. Data from the other two subsets in SOL — AD in childhood but not in adulthood and no AD in either phase of life — are expected to fuel an extended series of publications.
In the two groups, baseline characteristics were similar with about 60% reporting moderate to severe symptoms and a median age of about 37 years. The proportion of women was 61% in both groups.
Using the PUSH-D questionnaire, which Dr. Silverberg described as a validated tool for gauging a sense of stigmatization, the greater burden of AD was remarkably consistent for those with childhood-onset AD vs adult-onset AD. With higher scores representing a greater sense of stigmatization, the differences in the overall score (23.0 vs 18.1; P < .0001) were highly significant as was every other domain evaluated.
For all five social behavior domains, such as avoiding contact in public and wariness of approaching people spontaneously, having AD onset in childhood persisting into adulthood produced significantly higher scores than having AD onset in adulthood, with no exceptions (P < .001 for all).
AD From Childhood Consistently Results in Worse Outcomes
Providing examples for some of the other 12 domains, Dr. Silverberg maintained that feelings of shame and psychological discomfort were always greater in adults with AD persistent since childhood vs AD starting in adulthood. The P values for these outcomes, such as experiencing bias at work or reporting a sense that others avoided them, were typically highly significant (P < .001).
Compared with those whose AD started in adulthood, “adults with atopic eczema that started during childhood have significantly more difficulties in their life, including occupational relationships, daily life, personal life, and partner or family relationships,” Dr. Silverberg reported.
He said that the data were controlled for multiple confounders, particularly greater severity of AD. He acknowledged that childhood onset might be considered a surrogate for more severe disease, but the data were controlled for this possibility.
Despite the fact that there are “thousands of studies across all age groups showing the burden of AD,” Dr. Silverberg considers these data to be unique by emphasizing the burden of chronicity rather than the impact of AD in any single moment in time.
For those with chronic AD from childhood, “the effect is not just on physical health but a deep negative influence on psychological and social aspects of life,” Dr. Silverberg said, suggesting that the independent effects of chronicity might be worth studying across other dermatologic diseases.
“Regulatory agencies focus on what you can do in that moment of time, losing the bigger picture of how patients are affected chronically,” he said, adding that this is an area of clinical research that should be further explored.
What the data further suggest “is that the earlier we intervene, the more likely patients will do better long term,” he said.
Data Provide Evidence of Systemic Therapy in Kids
For Gudrun Ratzinger, MD, of the Department of Dermatology and Venerology at the Medical University of Innsbruck in Austria, these are valuable data.
“When I prescribe systemic therapies to children, I often get resistance from the healthcare system and even other colleagues,” said Dr. Ratzinger, who was asked to comment on the results. “We are at a teaching hospital, but I often find that when patients return to their home physician, the systemic therapies are stopped.”
In her own practice, she believes the most effective therapies should be introduced in children and adults when complete control is not achieved on first-line drugs. “These data are very helpful for me in explaining to others the importance of effective treatment of atopic dermatitis in children,” she said.
Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies, including those that make drugs for AD. Dr. Ratzinger reported financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Eli Lilly, Janssen, Leo Pharma, Novartis, Pelpharma, Pfizer, and UCB.
A version of this article first appeared on Medscape.com.
AMSTERDAM — There is a mountain of evidence that atopic dermatitis (AD) exerts a large negative impact on quality of life, but a unique study with
These data, drawn from the ambitious Scars of Life (SOL) project, “suggest that childhood AD persisting into adulthood is its own phenotype,” reported Jonathan I. Silverberg, MD, PhD, director of clinical research, Department of Dermatology, George Washington University, Washington, DC.
One reasonable message from these data is that the failure to achieve adequate control of AD in children, whether by a late start of systemic agents or other reasons, results in a greater lifetime burden of disease when the burden beyond physical symptoms is measured, according to Dr. Silverberg.
More Than 30,000 From Five Continents Participated
In the SOL project, which was designed to analyze how the age of AD onset affects the severity of symptoms and quality of life, completed questionnaires were collected from 30,801 individuals in 27 countries on five continents. The questions, which elicited data to measure the burden of AD, were developed in association with several professional and patient associations with an interest in AD, including the National Eczema Association.
The SOL project has produced an enormous amount of data in four distinct groups, but Dr. Silverberg, speaking in a late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology, focused on a comparison between the 2875 participants who had AD in childhood that has persisted into adulthood and the 7383 adults with adult-onset AD. Data from the other two subsets in SOL — AD in childhood but not in adulthood and no AD in either phase of life — are expected to fuel an extended series of publications.
In the two groups, baseline characteristics were similar with about 60% reporting moderate to severe symptoms and a median age of about 37 years. The proportion of women was 61% in both groups.
Using the PUSH-D questionnaire, which Dr. Silverberg described as a validated tool for gauging a sense of stigmatization, the greater burden of AD was remarkably consistent for those with childhood-onset AD vs adult-onset AD. With higher scores representing a greater sense of stigmatization, the differences in the overall score (23.0 vs 18.1; P < .0001) were highly significant as was every other domain evaluated.
For all five social behavior domains, such as avoiding contact in public and wariness of approaching people spontaneously, having AD onset in childhood persisting into adulthood produced significantly higher scores than having AD onset in adulthood, with no exceptions (P < .001 for all).
AD From Childhood Consistently Results in Worse Outcomes
Providing examples for some of the other 12 domains, Dr. Silverberg maintained that feelings of shame and psychological discomfort were always greater in adults with AD persistent since childhood vs AD starting in adulthood. The P values for these outcomes, such as experiencing bias at work or reporting a sense that others avoided them, were typically highly significant (P < .001).
Compared with those whose AD started in adulthood, “adults with atopic eczema that started during childhood have significantly more difficulties in their life, including occupational relationships, daily life, personal life, and partner or family relationships,” Dr. Silverberg reported.
He said that the data were controlled for multiple confounders, particularly greater severity of AD. He acknowledged that childhood onset might be considered a surrogate for more severe disease, but the data were controlled for this possibility.
Despite the fact that there are “thousands of studies across all age groups showing the burden of AD,” Dr. Silverberg considers these data to be unique by emphasizing the burden of chronicity rather than the impact of AD in any single moment in time.
For those with chronic AD from childhood, “the effect is not just on physical health but a deep negative influence on psychological and social aspects of life,” Dr. Silverberg said, suggesting that the independent effects of chronicity might be worth studying across other dermatologic diseases.
“Regulatory agencies focus on what you can do in that moment of time, losing the bigger picture of how patients are affected chronically,” he said, adding that this is an area of clinical research that should be further explored.
What the data further suggest “is that the earlier we intervene, the more likely patients will do better long term,” he said.
Data Provide Evidence of Systemic Therapy in Kids
For Gudrun Ratzinger, MD, of the Department of Dermatology and Venerology at the Medical University of Innsbruck in Austria, these are valuable data.
“When I prescribe systemic therapies to children, I often get resistance from the healthcare system and even other colleagues,” said Dr. Ratzinger, who was asked to comment on the results. “We are at a teaching hospital, but I often find that when patients return to their home physician, the systemic therapies are stopped.”
In her own practice, she believes the most effective therapies should be introduced in children and adults when complete control is not achieved on first-line drugs. “These data are very helpful for me in explaining to others the importance of effective treatment of atopic dermatitis in children,” she said.
Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies, including those that make drugs for AD. Dr. Ratzinger reported financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Eli Lilly, Janssen, Leo Pharma, Novartis, Pelpharma, Pfizer, and UCB.
A version of this article first appeared on Medscape.com.
FROM EADV 2024
Phase3 Data: Atopic Dermatitis Symptoms Improved with Topical Roflumilast
TOPLINE:
(AD) in two phase 3 trials.
METHODOLOGY:
- Two randomized, parallel-group, double-blind, vehicle-controlled phase 3 trials, INTEGUMENT-1 (n = 654) and INTEGUMENT-2 (n = 683), enrolled patients aged ≥ 6 years with mild to moderate AD who were randomly assigned in a 2:1 ratio to roflumilast cream 0.15%, a phosphodiesterase 4 inhibitor, or vehicle cream once daily for 4 weeks.
- The primary efficacy endpoint was the Validated Investigator Global Assessment for AD (vIGA-AD) success at week 4, defined as a score of 0 (clear) or 1 (almost clear) plus improvement of at least two grades from baseline.
- Secondary endpoints included vIGA-AD success at week 4 in patients with a baseline score of 3, at least a four-point reduction in the Worst Itch Numeric Rating Scale (WI-NRS), and at least a 75% reduction in the Eczema Area and Severity Index (EASI-75) at weeks 1, 2, and 4.
TAKEAWAY:
- Significantly more patients receiving roflumilast achieved vIGA-AD success at week 4 vs those in the vehicle group in INTEGUMENT-1 (32.0% vs 15.2%; P < .001) and INTEGUMENT-2 (28.9% vs 12.0%; P < .001), which was consistent across all age groups and in those with a baseline score of 3.
- Similarly, a greater proportion of patients treated with roflumilast vs vehicle achieved at least a four-point reduction in WI-NRS at weeks 1, 2, and 4, with improvements noted as early as 24 hours after the first application (P < .05 at all subsequent timepoints).
- The number of patients achieving EASI-75 and vIGA-AD scores of 0 or 1 was significantly higher with roflumilast than with vehicle at weeks 1, 2, and 4.
- Most treatment-emergent adverse events (TEAEs) were mild to moderate, with only 0.9% of the patients experiencing serious TEAEs in each trial. More than 95% of the patients showed no signs of irritation, and over 90% reported no or mild sensation at the application site.
IN PRACTICE:
The two phase 3 randomized clinical trials of patients with AD treated with roflumilast cream 0.15% “demonstrated improvement across multiple efficacy endpoints, including reducing pruritus within 24 hours after application, with favorable safety and tolerability,” the authors wrote. “Additional research, including subgroup analyses, will provide more data regarding the efficacy and safety of roflumilast cream 0.15%, in patients with AD,” they added.
SOURCE:
The study was led by Eric L. Simpson, MD, of the Department of Dermatology, Oregon Health & Science University, Portland, Oregon, and was published online on September 18 in JAMA Dermatology.
LIMITATIONS:
A short duration, a minimum age limit of 6 years, and the lack of an active comparator may influence the interpretation and generalizability of the results.
DISCLOSURES:
The study was sponsored by Arcutis Biotherapeutics. Simpson received grants and personal fees from Arcutis during this study. Three authors reported being employees and/or stockholders of Arcutis, two other authors reported patents for Arcutis, and several authors declared having various ties with various sources, including Arcutis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AD) in two phase 3 trials.
METHODOLOGY:
- Two randomized, parallel-group, double-blind, vehicle-controlled phase 3 trials, INTEGUMENT-1 (n = 654) and INTEGUMENT-2 (n = 683), enrolled patients aged ≥ 6 years with mild to moderate AD who were randomly assigned in a 2:1 ratio to roflumilast cream 0.15%, a phosphodiesterase 4 inhibitor, or vehicle cream once daily for 4 weeks.
- The primary efficacy endpoint was the Validated Investigator Global Assessment for AD (vIGA-AD) success at week 4, defined as a score of 0 (clear) or 1 (almost clear) plus improvement of at least two grades from baseline.
- Secondary endpoints included vIGA-AD success at week 4 in patients with a baseline score of 3, at least a four-point reduction in the Worst Itch Numeric Rating Scale (WI-NRS), and at least a 75% reduction in the Eczema Area and Severity Index (EASI-75) at weeks 1, 2, and 4.
TAKEAWAY:
- Significantly more patients receiving roflumilast achieved vIGA-AD success at week 4 vs those in the vehicle group in INTEGUMENT-1 (32.0% vs 15.2%; P < .001) and INTEGUMENT-2 (28.9% vs 12.0%; P < .001), which was consistent across all age groups and in those with a baseline score of 3.
- Similarly, a greater proportion of patients treated with roflumilast vs vehicle achieved at least a four-point reduction in WI-NRS at weeks 1, 2, and 4, with improvements noted as early as 24 hours after the first application (P < .05 at all subsequent timepoints).
- The number of patients achieving EASI-75 and vIGA-AD scores of 0 or 1 was significantly higher with roflumilast than with vehicle at weeks 1, 2, and 4.
- Most treatment-emergent adverse events (TEAEs) were mild to moderate, with only 0.9% of the patients experiencing serious TEAEs in each trial. More than 95% of the patients showed no signs of irritation, and over 90% reported no or mild sensation at the application site.
IN PRACTICE:
The two phase 3 randomized clinical trials of patients with AD treated with roflumilast cream 0.15% “demonstrated improvement across multiple efficacy endpoints, including reducing pruritus within 24 hours after application, with favorable safety and tolerability,” the authors wrote. “Additional research, including subgroup analyses, will provide more data regarding the efficacy and safety of roflumilast cream 0.15%, in patients with AD,” they added.
SOURCE:
The study was led by Eric L. Simpson, MD, of the Department of Dermatology, Oregon Health & Science University, Portland, Oregon, and was published online on September 18 in JAMA Dermatology.
LIMITATIONS:
A short duration, a minimum age limit of 6 years, and the lack of an active comparator may influence the interpretation and generalizability of the results.
DISCLOSURES:
The study was sponsored by Arcutis Biotherapeutics. Simpson received grants and personal fees from Arcutis during this study. Three authors reported being employees and/or stockholders of Arcutis, two other authors reported patents for Arcutis, and several authors declared having various ties with various sources, including Arcutis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
(AD) in two phase 3 trials.
METHODOLOGY:
- Two randomized, parallel-group, double-blind, vehicle-controlled phase 3 trials, INTEGUMENT-1 (n = 654) and INTEGUMENT-2 (n = 683), enrolled patients aged ≥ 6 years with mild to moderate AD who were randomly assigned in a 2:1 ratio to roflumilast cream 0.15%, a phosphodiesterase 4 inhibitor, or vehicle cream once daily for 4 weeks.
- The primary efficacy endpoint was the Validated Investigator Global Assessment for AD (vIGA-AD) success at week 4, defined as a score of 0 (clear) or 1 (almost clear) plus improvement of at least two grades from baseline.
- Secondary endpoints included vIGA-AD success at week 4 in patients with a baseline score of 3, at least a four-point reduction in the Worst Itch Numeric Rating Scale (WI-NRS), and at least a 75% reduction in the Eczema Area and Severity Index (EASI-75) at weeks 1, 2, and 4.
TAKEAWAY:
- Significantly more patients receiving roflumilast achieved vIGA-AD success at week 4 vs those in the vehicle group in INTEGUMENT-1 (32.0% vs 15.2%; P < .001) and INTEGUMENT-2 (28.9% vs 12.0%; P < .001), which was consistent across all age groups and in those with a baseline score of 3.
- Similarly, a greater proportion of patients treated with roflumilast vs vehicle achieved at least a four-point reduction in WI-NRS at weeks 1, 2, and 4, with improvements noted as early as 24 hours after the first application (P < .05 at all subsequent timepoints).
- The number of patients achieving EASI-75 and vIGA-AD scores of 0 or 1 was significantly higher with roflumilast than with vehicle at weeks 1, 2, and 4.
- Most treatment-emergent adverse events (TEAEs) were mild to moderate, with only 0.9% of the patients experiencing serious TEAEs in each trial. More than 95% of the patients showed no signs of irritation, and over 90% reported no or mild sensation at the application site.
IN PRACTICE:
The two phase 3 randomized clinical trials of patients with AD treated with roflumilast cream 0.15% “demonstrated improvement across multiple efficacy endpoints, including reducing pruritus within 24 hours after application, with favorable safety and tolerability,” the authors wrote. “Additional research, including subgroup analyses, will provide more data regarding the efficacy and safety of roflumilast cream 0.15%, in patients with AD,” they added.
SOURCE:
The study was led by Eric L. Simpson, MD, of the Department of Dermatology, Oregon Health & Science University, Portland, Oregon, and was published online on September 18 in JAMA Dermatology.
LIMITATIONS:
A short duration, a minimum age limit of 6 years, and the lack of an active comparator may influence the interpretation and generalizability of the results.
DISCLOSURES:
The study was sponsored by Arcutis Biotherapeutics. Simpson received grants and personal fees from Arcutis during this study. Three authors reported being employees and/or stockholders of Arcutis, two other authors reported patents for Arcutis, and several authors declared having various ties with various sources, including Arcutis.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
A 71-year-old White female developed erosions after hip replacement surgery 2 months prior to presentation
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.