User login
Cold viruses thrived in kids as other viruses faded in 2020
The common-cold viruses rhinovirus (RV) and enterovirus (EV) continued to circulate among children during the COVID-19 pandemic while there were sharp declines in influenza, respiratory syncytial virus (RSV), and other respiratory viruses, new data indicate.
Researchers used data from the Centers for Disease Control and Prevention’s New Vaccine Surveillance Network. The cases involved 37,676 children in seven geographically diverse U.S. medical centers between December 2016 and January 2021. Patients presented to emergency departments or were hospitalized with RV, EV, and other acute respiratory viruses.
The investigators found that the percentage of children in whom RV/EV was detected from March 2020 to January 2021 was similar to the percentage during the same months in 2017-2018 and 2019-2020. However, the proportion of children infected with influenza, RSV, and other respiratory viruses combined dropped significantly in comparison to the three prior seasons.
Danielle Rankin, MPH, lead author of the study and a doctoral candidate in pediatric infectious disease at Vanderbilt University, in Nashville, Tenn., presented the study on Sept. 30 during a press conference at IDWeek 2021, an annual scientific meeting on infectious diseases.
“Reasoning for rhinovirus and enterovirus circulation is unknown but may be attributed to a number of factors, such as different transmission routes or the prolonged survival of the virus on surfaces,” Ms. Rankin said. “Improved understanding of these persistent factors of RV/EV and the role of nonpharmaceutical interventions on transmission dynamics can further guide future prevention recommendations and guidelines.”
Coauthor Claire Midgley, PhD, an epidemiologist in the Division of Viral Diseases at the CDC, told reporters that further studies will assess why RV and EV remained during the pandemic and which virus types within the RV/EV group persisted.
“We do know that the virus can spread through secretions on people’s hands,” she said. “Washing kids’ hands regularly and trying not to touch your face where possible is a really effective way to prevent transmission,” Dr. Midgley said.
“The more we understand about all of these factors, the better we can inform prevention measures.”
Andrew T. Pavia, MD, chief, division of pediatric infectious diseases, University of Utah, Salt Lake City, who was not involved in the study, told this news organization that rhinoviruses can persist in the nose for a very long time, especially in younger children, which increases the opportunities for transmission.
“Very young children who are unable to wear masks or are unlikely to wear them well may be acting as the reservoir, allowing transmission in households,” he said. “There is also an enormous pool of diverse rhinoviruses, so past colds provide limited immunity, as everyone has found out from experience.”
Martha Perry, MD, associate professor at the University of North Carolina at Chapel Hill and chief of adolescent medicine, told this news organization that some of the differences in the prevalence of viruses may be because of their seasonality.
“Times when there were more mask mandates were times when RSV and influenza are more prevalent,” said Dr. Perry, who was not involved with the study. “We were masking more intently during those times, and there was loosening of restrictions when we see more enterovirus, particularly because that tends to be more of a summer/fall virus.”
She agreed that the differences may result from the way the viruses are transmitted.
“Perhaps masks were helping with RSV and influenza, but perhaps there was not as much hand washing or cleansing as needed to prevent the spread of rhinovirus and enterovirus, because those are viruses that require a bit more hand washing,” Dr. Perry said. “They are less aerosolized and better spread with hand-to-hand contact.”
Dr. Perry added that on the flip side, “it’s really exciting that there are ways we can prevent RSV and influenza, which tend to cause more severe infection.”
Ms. Rankin said limitations of the study include the fact that from March 2020 to January 2021, health care–seeking behaviors may have changed because of the pandemic and that the study does not include the frequency of respiratory viruses in the outpatient setting.
The sharp 2020-2021 decline in RSV reported in the study may have reversed after many of the COVID-19 restrictions were lifted this summer.
This news organization reported in June of this year that the CDC has issued a health advisory to notify clinicians and caregivers about an increase in cases of interseasonal RSV in parts of the southern United States.
The CDC has urged broader testing for RSV among patients presenting with acute respiratory illness who test negative for SARS-CoV-2.
The study’s authors, Ms. Pavia, and Dr. Perry have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The common-cold viruses rhinovirus (RV) and enterovirus (EV) continued to circulate among children during the COVID-19 pandemic while there were sharp declines in influenza, respiratory syncytial virus (RSV), and other respiratory viruses, new data indicate.
Researchers used data from the Centers for Disease Control and Prevention’s New Vaccine Surveillance Network. The cases involved 37,676 children in seven geographically diverse U.S. medical centers between December 2016 and January 2021. Patients presented to emergency departments or were hospitalized with RV, EV, and other acute respiratory viruses.
The investigators found that the percentage of children in whom RV/EV was detected from March 2020 to January 2021 was similar to the percentage during the same months in 2017-2018 and 2019-2020. However, the proportion of children infected with influenza, RSV, and other respiratory viruses combined dropped significantly in comparison to the three prior seasons.
Danielle Rankin, MPH, lead author of the study and a doctoral candidate in pediatric infectious disease at Vanderbilt University, in Nashville, Tenn., presented the study on Sept. 30 during a press conference at IDWeek 2021, an annual scientific meeting on infectious diseases.
“Reasoning for rhinovirus and enterovirus circulation is unknown but may be attributed to a number of factors, such as different transmission routes or the prolonged survival of the virus on surfaces,” Ms. Rankin said. “Improved understanding of these persistent factors of RV/EV and the role of nonpharmaceutical interventions on transmission dynamics can further guide future prevention recommendations and guidelines.”
Coauthor Claire Midgley, PhD, an epidemiologist in the Division of Viral Diseases at the CDC, told reporters that further studies will assess why RV and EV remained during the pandemic and which virus types within the RV/EV group persisted.
“We do know that the virus can spread through secretions on people’s hands,” she said. “Washing kids’ hands regularly and trying not to touch your face where possible is a really effective way to prevent transmission,” Dr. Midgley said.
“The more we understand about all of these factors, the better we can inform prevention measures.”
Andrew T. Pavia, MD, chief, division of pediatric infectious diseases, University of Utah, Salt Lake City, who was not involved in the study, told this news organization that rhinoviruses can persist in the nose for a very long time, especially in younger children, which increases the opportunities for transmission.
“Very young children who are unable to wear masks or are unlikely to wear them well may be acting as the reservoir, allowing transmission in households,” he said. “There is also an enormous pool of diverse rhinoviruses, so past colds provide limited immunity, as everyone has found out from experience.”
Martha Perry, MD, associate professor at the University of North Carolina at Chapel Hill and chief of adolescent medicine, told this news organization that some of the differences in the prevalence of viruses may be because of their seasonality.
“Times when there were more mask mandates were times when RSV and influenza are more prevalent,” said Dr. Perry, who was not involved with the study. “We were masking more intently during those times, and there was loosening of restrictions when we see more enterovirus, particularly because that tends to be more of a summer/fall virus.”
She agreed that the differences may result from the way the viruses are transmitted.
“Perhaps masks were helping with RSV and influenza, but perhaps there was not as much hand washing or cleansing as needed to prevent the spread of rhinovirus and enterovirus, because those are viruses that require a bit more hand washing,” Dr. Perry said. “They are less aerosolized and better spread with hand-to-hand contact.”
Dr. Perry added that on the flip side, “it’s really exciting that there are ways we can prevent RSV and influenza, which tend to cause more severe infection.”
Ms. Rankin said limitations of the study include the fact that from March 2020 to January 2021, health care–seeking behaviors may have changed because of the pandemic and that the study does not include the frequency of respiratory viruses in the outpatient setting.
The sharp 2020-2021 decline in RSV reported in the study may have reversed after many of the COVID-19 restrictions were lifted this summer.
This news organization reported in June of this year that the CDC has issued a health advisory to notify clinicians and caregivers about an increase in cases of interseasonal RSV in parts of the southern United States.
The CDC has urged broader testing for RSV among patients presenting with acute respiratory illness who test negative for SARS-CoV-2.
The study’s authors, Ms. Pavia, and Dr. Perry have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The common-cold viruses rhinovirus (RV) and enterovirus (EV) continued to circulate among children during the COVID-19 pandemic while there were sharp declines in influenza, respiratory syncytial virus (RSV), and other respiratory viruses, new data indicate.
Researchers used data from the Centers for Disease Control and Prevention’s New Vaccine Surveillance Network. The cases involved 37,676 children in seven geographically diverse U.S. medical centers between December 2016 and January 2021. Patients presented to emergency departments or were hospitalized with RV, EV, and other acute respiratory viruses.
The investigators found that the percentage of children in whom RV/EV was detected from March 2020 to January 2021 was similar to the percentage during the same months in 2017-2018 and 2019-2020. However, the proportion of children infected with influenza, RSV, and other respiratory viruses combined dropped significantly in comparison to the three prior seasons.
Danielle Rankin, MPH, lead author of the study and a doctoral candidate in pediatric infectious disease at Vanderbilt University, in Nashville, Tenn., presented the study on Sept. 30 during a press conference at IDWeek 2021, an annual scientific meeting on infectious diseases.
“Reasoning for rhinovirus and enterovirus circulation is unknown but may be attributed to a number of factors, such as different transmission routes or the prolonged survival of the virus on surfaces,” Ms. Rankin said. “Improved understanding of these persistent factors of RV/EV and the role of nonpharmaceutical interventions on transmission dynamics can further guide future prevention recommendations and guidelines.”
Coauthor Claire Midgley, PhD, an epidemiologist in the Division of Viral Diseases at the CDC, told reporters that further studies will assess why RV and EV remained during the pandemic and which virus types within the RV/EV group persisted.
“We do know that the virus can spread through secretions on people’s hands,” she said. “Washing kids’ hands regularly and trying not to touch your face where possible is a really effective way to prevent transmission,” Dr. Midgley said.
“The more we understand about all of these factors, the better we can inform prevention measures.”
Andrew T. Pavia, MD, chief, division of pediatric infectious diseases, University of Utah, Salt Lake City, who was not involved in the study, told this news organization that rhinoviruses can persist in the nose for a very long time, especially in younger children, which increases the opportunities for transmission.
“Very young children who are unable to wear masks or are unlikely to wear them well may be acting as the reservoir, allowing transmission in households,” he said. “There is also an enormous pool of diverse rhinoviruses, so past colds provide limited immunity, as everyone has found out from experience.”
Martha Perry, MD, associate professor at the University of North Carolina at Chapel Hill and chief of adolescent medicine, told this news organization that some of the differences in the prevalence of viruses may be because of their seasonality.
“Times when there were more mask mandates were times when RSV and influenza are more prevalent,” said Dr. Perry, who was not involved with the study. “We were masking more intently during those times, and there was loosening of restrictions when we see more enterovirus, particularly because that tends to be more of a summer/fall virus.”
She agreed that the differences may result from the way the viruses are transmitted.
“Perhaps masks were helping with RSV and influenza, but perhaps there was not as much hand washing or cleansing as needed to prevent the spread of rhinovirus and enterovirus, because those are viruses that require a bit more hand washing,” Dr. Perry said. “They are less aerosolized and better spread with hand-to-hand contact.”
Dr. Perry added that on the flip side, “it’s really exciting that there are ways we can prevent RSV and influenza, which tend to cause more severe infection.”
Ms. Rankin said limitations of the study include the fact that from March 2020 to January 2021, health care–seeking behaviors may have changed because of the pandemic and that the study does not include the frequency of respiratory viruses in the outpatient setting.
The sharp 2020-2021 decline in RSV reported in the study may have reversed after many of the COVID-19 restrictions were lifted this summer.
This news organization reported in June of this year that the CDC has issued a health advisory to notify clinicians and caregivers about an increase in cases of interseasonal RSV in parts of the southern United States.
The CDC has urged broader testing for RSV among patients presenting with acute respiratory illness who test negative for SARS-CoV-2.
The study’s authors, Ms. Pavia, and Dr. Perry have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Role of Inpatient Dermatology Consultations
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
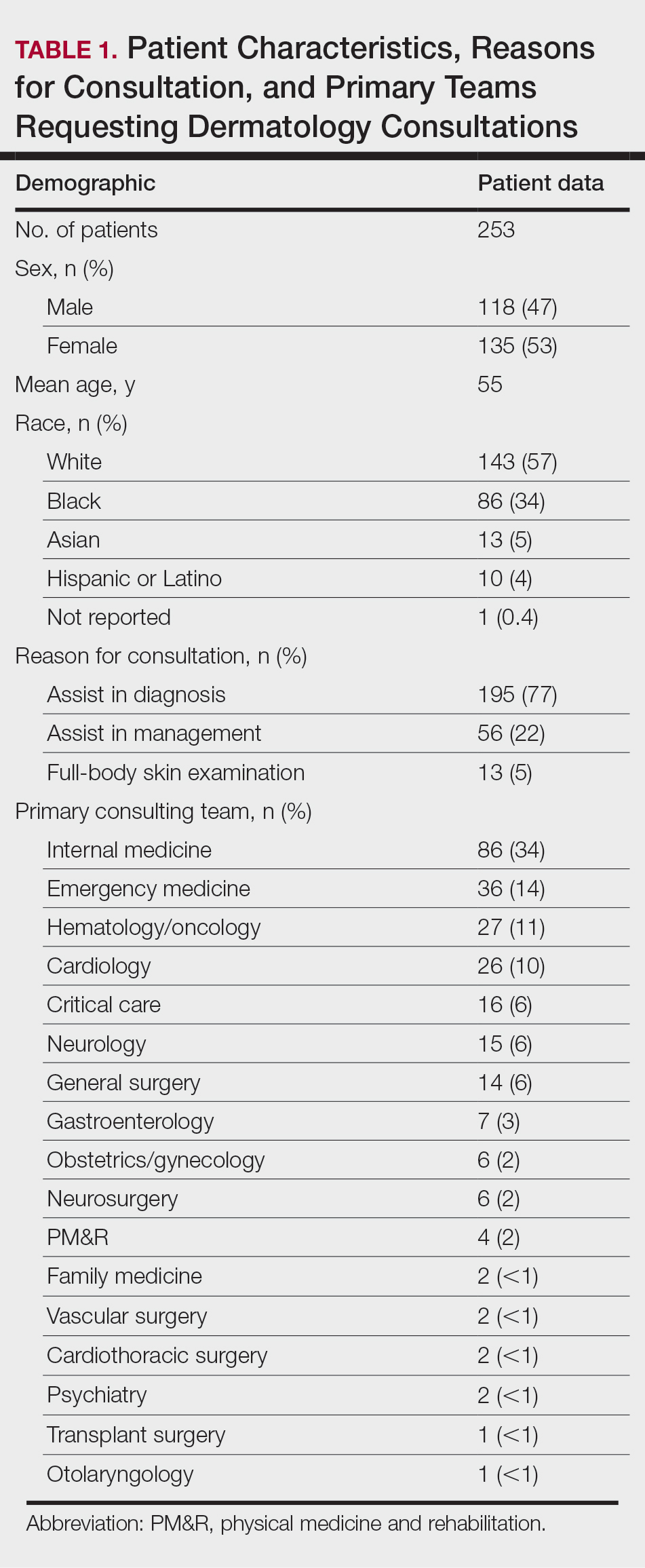
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.
Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
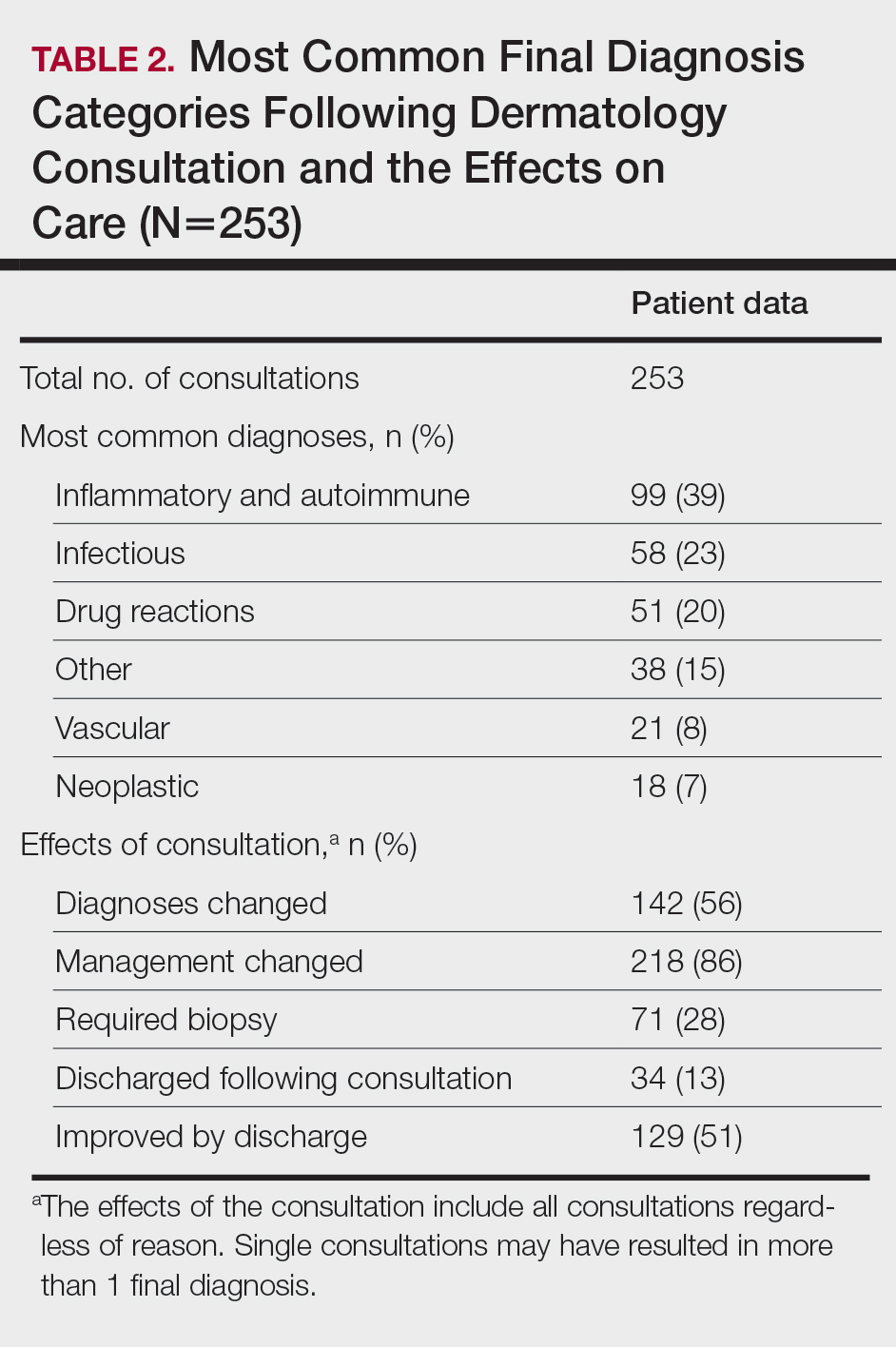
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.
Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.
Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.
Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.
Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.
Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
Practice Points
- Inpatient dermatologists fill knowledge gaps that often alter the diagnosis, management, and hospital course of hospitalized patients.
- Several medical specialties benefit from niche expertise of inpatient dermatologists specific to their patient population.
- Integration of inpatient dermatology consultations can prevent unnecessary hospital admissions and medication administration.
Children and COVID: Decline of summer surge continues
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
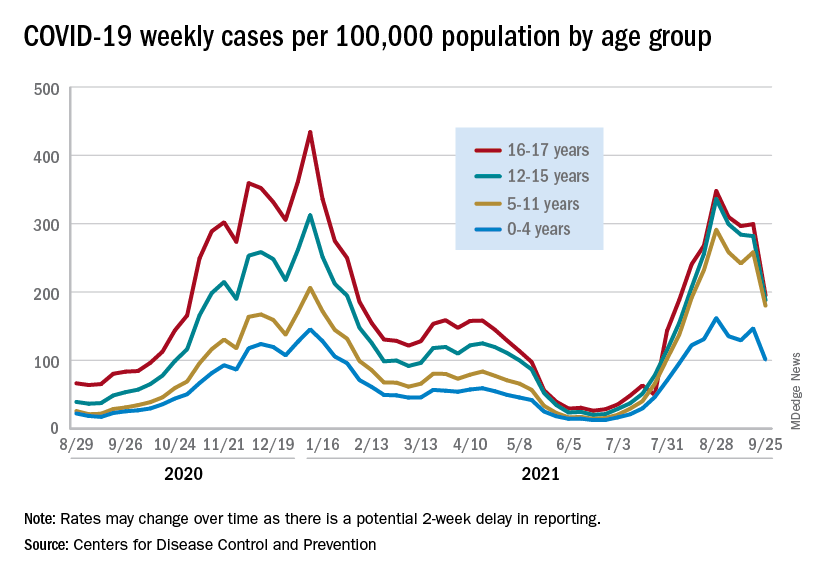
Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
The continuing decline in COVID-19 incidence suggests the latest surge has peaked as new cases in children dropped for the 4th consecutive week, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

Preliminary data from the Centers for Disease Control and Prevention, however, show an uptick in new cases in late September, largely among younger children, that may indicate otherwise. Those data have a potential 2-week reporting delay, the CDC said on its COVID Data Tracker, so the most recent points on the graph (see above) could still go up.
. Those new cases made up almost 27% of all cases for the week, and the nearly 5.9 million child cases that have been reported since the start of the pandemic represent 16.2% of cases among Americans of all ages, the two groups said in their weekly COVID-19 report.
The CDC data on new cases by age group suggest that younger children have borne a heavier burden in the summer surge of COVID than they did last winter. The rate of new cases was not as high for 16- and 17-year-olds in the summer, but the other age groups all reached higher peaks than in the winter, including the 12- to 15-year-olds, who have been getting vaccinated since May, according to the COVID Data Tracker.
With vaccination approval getting closer for children under age 12 years, initiation in those already eligible continues to slide. Those aged 12-15 made up just 6.9% of new vaccinations during the 2 weeks from Sept. 21 to Oct. 4, and that figure has been dropping since July 13-26, when it was 14.1%. Vaccine initiation among 16- and 17-year-olds over that time has dropped by almost half, from 5.4% to 2.9%, the CDC data show.
All the vaccinations so far add up to this: Almost 55% of those aged 12-15 have gotten at least one dose of COVID vaccine, as have over 62% of those aged 16-17, and 52% of the older group is fully vaccinated, as is 44% of the younger group. Altogether, 10.8 million children were fully vaccinated as of Oct. 4, including those under 12 who may be participating in clinical trials or had a birth date entered incorrectly, the CDC said.
COVID-19: Two more cases of mucosal skin ulcers reported in male teens
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Irish A similar case in an adolescent, also with ulcers affecting the mouth and penis, was reported earlier in 2021 in the United States.
“Our cases show that a swab for COVID-19 can be added to the list of investigations for mucosal and cutaneous rashes in children and probably adults,” said dermatologist Stephanie Bowe, MD, of South Infirmary-Victoria University Hospital in Cork, Ireland, in an interview. “Our patients seemed to improve with IV steroids, but there is not enough data to recommend them to all patients or for use in the different cutaneous presentations associated with COVID-19.”
The new case reports were presented at the 2021 meeting of the World Congress of Pediatric Dermatology and published in Pediatric Dermatology.
Researchers have noted that skin disorders linked to COVID-19 infection are different than those in adults. In children, the conditions include morbilliform rash, pernio-like acral lesions, urticaria, macular erythema, vesicular eruption, papulosquamous eruption, and retiform purpura. “The pathogenesis of each is not fully understood but likely related to the inflammatory response to COVID-19 and the various pathways within the body, which become activated,” Dr. Bowe said.
The first patient, a 17-year-old boy, presented at clinic 6 days after he’d been confirmed to be infected with COVID-19 and 8 days after developing fever and cough. “He had a 2-day history of conjunctivitis and ulceration of his oral mucosa, erythematous circumferential erosions of the glans penis with no other cutaneous findings,” the authors write in the report.
The boy “was distressed and embarrassed about his genital ulceration and also found eating very painful due to his oral ulceration,” Dr. Bowe said.
The second patient, a 14-year-old boy, was hospitalized 7 days after a positive COVID-19 test and 9 days after developing cough and fever. “He had a 5-day history of ulceration of the oral mucosa with mild conjunctivitis,” the authors wrote. “Ulceration of the glans penis developed on day 2 of admission.”
The 14-year-old was sicker than the 17-year-old boy, Dr. Bowe said. “He was unable to tolerate an oral diet for several days and had exquisite pain and vomiting with his coughing fits.”
This patient had a history of recurrent herpes labialis, but it’s unclear whether herpes simplex virus (HSV) played a role in the COVID-19–related case. “There is a possibility that the patient was more susceptible to viral cutaneous reactions during COVID-19 infection, but we didn’t have any definite history of HSV infection at the time of mucositis,” Dr. Bowe said. “We also didn’t have any swabs positive for HSV even though several were done at the time.”
Both patients received IV steroids – hydrocortisone at 100 mg 3 times daily for 3 days. This treatment was used “because of deterioration in symptoms and COVID-19 infection,” Dr. Bowe said. “IV steroids were used for respiratory symptoms of COVID-19, so we felt these cutaneous symptoms may have also been caused by an inflammatory response and might benefit from steroids. There was very little literature about this specific situation, though.”
She added that intravenous steroids wouldn’t be appropriate for most pediatric patients, and noted that “their use is controversial in the literature for erythema multiforme and RIME.”
In addition, the patients received betamethasone valerate 0.1% ointment once daily, hydrocortisone 2.5 mg buccal tablets 4 times daily, analgesia with acetaminophen and ibuprofen, and intravenous hydration. The first patient also received prednisolone 1% eye drops, while the second patient was given lidocaine hydrochloride mouthwash and total parenteral nutrition for 5 days.
The patients were discharged after 4 and 14 days, respectively.
Dermatologists in Massachusetts reported a similar case earlier in 2021 in a 17-year-old boy who was positive for COVID-19 and presented with “shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities.”
The patient received betamethasone valerate 0.1% ointment for the lips and penis, intraoral dexamethasone solution, viscous lidocaine, acetaminophen, and ibuprofen. He also received oral prednisone at approximately 1 mg/kg daily for 4 consecutive days after worsening oral pain. A recurrence of oral pain 3 months later was resolved with a higher and longer treatment with oral prednisone.
Dermatologists have also reported cases of erythema multiforme lesions of the mucosa in adults with COVID-19. One case was reported in Iran, and the other in France.
The authors report no study funding and disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Oteseconazole promising for recurrent yeast infections
A phase 3, randomized, double-blind, controlled trial has shown that oteseconazole (Mycovia Pharmaceuticals), an oral antifungal agent, is safe and effective in treating acute and recurrent yeast infections (vulvovaginal candidiasis [VVC]) and in preventing recurrence of acute VVC episodes.
Findings of the ultraVIOLET trial, which compared oteseconazole with the standard fluconazole, were presented at IDWeek 2021, an annual scientific meeting on infectious diseases, by lead author Mark G. Martens, MD, a professor in the department of obstetrics and gynecology at Drexel University College of Medicine in Philadelphia.
About 75% of all women will have a yeast infection in their lifetime, Dr. Martens noted. About 138 million women worldwide have recurring episodes (at least three acute episodes in the last year) of the debilitating condition.
“Recurrent vulvovaginal candidiasis typically requires treatment of the acute episode followed by long-term suppressive therapy with either weekly or biweekly fluconazole,” Dr. Martens said. However, when therapy stops, more than 50% of patients with recurrent VVC experience an infection within the next 6 months, which takes a significant toll on daily life.
Additionally, fluconazole has been linked with safety issues concerning chronic dosing, he said, citing liver toxicity, drug-drug interactions and “increased risk of miscarriage and birth defects when used during pregnancy.”
Topical treatments have been associated with messy application and burning, he noted.
For this study, researchers enrolled 219 women with a history of recurrent VVC at 51 U.S. sites. Participants were randomized either to 600 mg oteseconazole on day 1, 450 mg oteseconazole on day 2 or placebo capsules; or three sequential 150 mg doses (every 72 hours) of fluconazole together with matching placebo capsules.
In the maintenance phase, 185 women with resolved acute VVC (clinical signs and symptoms were scored below 3) on day 14 received 150 mg oteseconazole or placebo weekly for 11 weeks.
Oteseconazole was superior to fluconazole/placebo in the proportion of subjects with at least one culture-verified acute VVC episode through week 50 in the intent-to-treat population (P < .001) which included subjects who failed to clear their infection in the induction phase.
The average percentage of participants with at least one culture-verified acute VVC episode through week 50 was lower in the oteseconazole group (5.1%), compared with the fluconazole/placebo group (42.2%).
Oteseconazole was noninferior to fluconazole in the proportion of subjects with resolved acute VVC infections at day 14 – 93.2% for the oteseconazole group vs. 95.8% for the fluconazole/placebo group.
The percentages of women who had at least one treatment-emergent adverse event (TEAE) were similar – 54% in the oteseconazole group and 64% in the fluconazole/placebo group. Most TEAEs were mild or moderate and there were no drug-related SAEs or adverse effects on liver function.
“There was no difference in the two groups in he baseline characteristics of age, race, and history of diabetes,” he said.
Oluwatosin Goje, MD, an ob.gyn. with the Cleveland Clinic told this news organization that the drug may offer another option for women who don’t respond to azoles.
“The CDC guidelines say, and I agree, that most episodes of recurrent VVC that are caused by Candida albicans will respond to topical azoles, to oral azoles, to the known drugs that are available. You just may have to use them for a prolonged period of time,” Dr. Goje said. But some patients won’t respond to azoles, the currently available drugs, and topical treatments – so new options are welcome for them, she noted.
She pointed out that the U.S. Food and Drug Administration in June approved ibrexafungerp (Brexafemme), the first oral nonazole treatment for vaginal yeast infections. It was the first approved medicine in a novel antifungal class in more than 2 decades.
Dr. Goje, who runs a large clinic with substantial numbers of women with recurrent yeast infections, said the psychosocial problems women with recurrent yeast infections face – and the time off work and money spent trying to get temporary relief from over-the-counter medications – is underestimated.
“Women have long suffered vaginitis. It can be a lot of social and economic burden. So anything in the toolbox to help women is welcome,” Dr. Goje said.
The study was sponsored by Mycovia Pharmaceuticals. Dr. Martens reports no relevant financial relationships. Several coauthors are either employees of Mycovia or receive support from the company. Dr. Goje has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A phase 3, randomized, double-blind, controlled trial has shown that oteseconazole (Mycovia Pharmaceuticals), an oral antifungal agent, is safe and effective in treating acute and recurrent yeast infections (vulvovaginal candidiasis [VVC]) and in preventing recurrence of acute VVC episodes.
Findings of the ultraVIOLET trial, which compared oteseconazole with the standard fluconazole, were presented at IDWeek 2021, an annual scientific meeting on infectious diseases, by lead author Mark G. Martens, MD, a professor in the department of obstetrics and gynecology at Drexel University College of Medicine in Philadelphia.
About 75% of all women will have a yeast infection in their lifetime, Dr. Martens noted. About 138 million women worldwide have recurring episodes (at least three acute episodes in the last year) of the debilitating condition.
“Recurrent vulvovaginal candidiasis typically requires treatment of the acute episode followed by long-term suppressive therapy with either weekly or biweekly fluconazole,” Dr. Martens said. However, when therapy stops, more than 50% of patients with recurrent VVC experience an infection within the next 6 months, which takes a significant toll on daily life.
Additionally, fluconazole has been linked with safety issues concerning chronic dosing, he said, citing liver toxicity, drug-drug interactions and “increased risk of miscarriage and birth defects when used during pregnancy.”
Topical treatments have been associated with messy application and burning, he noted.
For this study, researchers enrolled 219 women with a history of recurrent VVC at 51 U.S. sites. Participants were randomized either to 600 mg oteseconazole on day 1, 450 mg oteseconazole on day 2 or placebo capsules; or three sequential 150 mg doses (every 72 hours) of fluconazole together with matching placebo capsules.
In the maintenance phase, 185 women with resolved acute VVC (clinical signs and symptoms were scored below 3) on day 14 received 150 mg oteseconazole or placebo weekly for 11 weeks.
Oteseconazole was superior to fluconazole/placebo in the proportion of subjects with at least one culture-verified acute VVC episode through week 50 in the intent-to-treat population (P < .001) which included subjects who failed to clear their infection in the induction phase.
The average percentage of participants with at least one culture-verified acute VVC episode through week 50 was lower in the oteseconazole group (5.1%), compared with the fluconazole/placebo group (42.2%).
Oteseconazole was noninferior to fluconazole in the proportion of subjects with resolved acute VVC infections at day 14 – 93.2% for the oteseconazole group vs. 95.8% for the fluconazole/placebo group.
The percentages of women who had at least one treatment-emergent adverse event (TEAE) were similar – 54% in the oteseconazole group and 64% in the fluconazole/placebo group. Most TEAEs were mild or moderate and there were no drug-related SAEs or adverse effects on liver function.
“There was no difference in the two groups in he baseline characteristics of age, race, and history of diabetes,” he said.
Oluwatosin Goje, MD, an ob.gyn. with the Cleveland Clinic told this news organization that the drug may offer another option for women who don’t respond to azoles.
“The CDC guidelines say, and I agree, that most episodes of recurrent VVC that are caused by Candida albicans will respond to topical azoles, to oral azoles, to the known drugs that are available. You just may have to use them for a prolonged period of time,” Dr. Goje said. But some patients won’t respond to azoles, the currently available drugs, and topical treatments – so new options are welcome for them, she noted.
She pointed out that the U.S. Food and Drug Administration in June approved ibrexafungerp (Brexafemme), the first oral nonazole treatment for vaginal yeast infections. It was the first approved medicine in a novel antifungal class in more than 2 decades.
Dr. Goje, who runs a large clinic with substantial numbers of women with recurrent yeast infections, said the psychosocial problems women with recurrent yeast infections face – and the time off work and money spent trying to get temporary relief from over-the-counter medications – is underestimated.
“Women have long suffered vaginitis. It can be a lot of social and economic burden. So anything in the toolbox to help women is welcome,” Dr. Goje said.
The study was sponsored by Mycovia Pharmaceuticals. Dr. Martens reports no relevant financial relationships. Several coauthors are either employees of Mycovia or receive support from the company. Dr. Goje has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A phase 3, randomized, double-blind, controlled trial has shown that oteseconazole (Mycovia Pharmaceuticals), an oral antifungal agent, is safe and effective in treating acute and recurrent yeast infections (vulvovaginal candidiasis [VVC]) and in preventing recurrence of acute VVC episodes.
Findings of the ultraVIOLET trial, which compared oteseconazole with the standard fluconazole, were presented at IDWeek 2021, an annual scientific meeting on infectious diseases, by lead author Mark G. Martens, MD, a professor in the department of obstetrics and gynecology at Drexel University College of Medicine in Philadelphia.
About 75% of all women will have a yeast infection in their lifetime, Dr. Martens noted. About 138 million women worldwide have recurring episodes (at least three acute episodes in the last year) of the debilitating condition.
“Recurrent vulvovaginal candidiasis typically requires treatment of the acute episode followed by long-term suppressive therapy with either weekly or biweekly fluconazole,” Dr. Martens said. However, when therapy stops, more than 50% of patients with recurrent VVC experience an infection within the next 6 months, which takes a significant toll on daily life.
Additionally, fluconazole has been linked with safety issues concerning chronic dosing, he said, citing liver toxicity, drug-drug interactions and “increased risk of miscarriage and birth defects when used during pregnancy.”
Topical treatments have been associated with messy application and burning, he noted.
For this study, researchers enrolled 219 women with a history of recurrent VVC at 51 U.S. sites. Participants were randomized either to 600 mg oteseconazole on day 1, 450 mg oteseconazole on day 2 or placebo capsules; or three sequential 150 mg doses (every 72 hours) of fluconazole together with matching placebo capsules.
In the maintenance phase, 185 women with resolved acute VVC (clinical signs and symptoms were scored below 3) on day 14 received 150 mg oteseconazole or placebo weekly for 11 weeks.
Oteseconazole was superior to fluconazole/placebo in the proportion of subjects with at least one culture-verified acute VVC episode through week 50 in the intent-to-treat population (P < .001) which included subjects who failed to clear their infection in the induction phase.
The average percentage of participants with at least one culture-verified acute VVC episode through week 50 was lower in the oteseconazole group (5.1%), compared with the fluconazole/placebo group (42.2%).
Oteseconazole was noninferior to fluconazole in the proportion of subjects with resolved acute VVC infections at day 14 – 93.2% for the oteseconazole group vs. 95.8% for the fluconazole/placebo group.
The percentages of women who had at least one treatment-emergent adverse event (TEAE) were similar – 54% in the oteseconazole group and 64% in the fluconazole/placebo group. Most TEAEs were mild or moderate and there were no drug-related SAEs or adverse effects on liver function.
“There was no difference in the two groups in he baseline characteristics of age, race, and history of diabetes,” he said.
Oluwatosin Goje, MD, an ob.gyn. with the Cleveland Clinic told this news organization that the drug may offer another option for women who don’t respond to azoles.
“The CDC guidelines say, and I agree, that most episodes of recurrent VVC that are caused by Candida albicans will respond to topical azoles, to oral azoles, to the known drugs that are available. You just may have to use them for a prolonged period of time,” Dr. Goje said. But some patients won’t respond to azoles, the currently available drugs, and topical treatments – so new options are welcome for them, she noted.
She pointed out that the U.S. Food and Drug Administration in June approved ibrexafungerp (Brexafemme), the first oral nonazole treatment for vaginal yeast infections. It was the first approved medicine in a novel antifungal class in more than 2 decades.
Dr. Goje, who runs a large clinic with substantial numbers of women with recurrent yeast infections, said the psychosocial problems women with recurrent yeast infections face – and the time off work and money spent trying to get temporary relief from over-the-counter medications – is underestimated.
“Women have long suffered vaginitis. It can be a lot of social and economic burden. So anything in the toolbox to help women is welcome,” Dr. Goje said.
The study was sponsored by Mycovia Pharmaceuticals. Dr. Martens reports no relevant financial relationships. Several coauthors are either employees of Mycovia or receive support from the company. Dr. Goje has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID vaccine controversies: How can hospitalists help?
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
On April 1, Houston Methodist Hospital in Houston, Texas, announced a new policy that all of its staff would need to be vaccinated against COVID-19 by June 7 in order to hold onto their jobs. Most responded positively but an estimated 150 staff members who did not comply either resigned or were terminated. A lawsuit by employees opposed to the vaccine mandate was dismissed by Federal District Court Judge Lynn Hughes in June, although a subsequent lawsuit was filed Aug. 16.
Vaccines have been shown to dramatically reduce both the incidence and the severity of COVID infections. Vaccinations of health care workers, especially those who have direct contact with patients, are demonstrated to be effective strategies to significantly reduce, although not eliminate, the possibility of viral transmissions to patients – or to health care workers themselves – thus saving lives.
Hospitalists, in their central role in the care of hospitalized patients, and often with primary responsibility for managing their hospital’s COVID-19 caseloads, may find themselves encountering conversations about the vaccine, its safety, effectiveness, and mandates with their peers, other hospital staff, patients, and families, and their communities. They can play key roles in advocating for the vaccine, answering questions, clarifying the science, and dispelling misinformation – for those who are willing to listen.
Becker’s Hospital Review, which has kept an ongoing tally of announced vaccine mandate policies in hospitals, health systems, and health departments nationwide, reported on Aug. 13 that 1,850 or 30% of U.S. hospitals, had announced vaccine mandates.1 Often exceptions can be made, such as for medical or religious reasons, or with other declarations or opt-out provisions. But in many settings, mandating COVID vaccinations won’t be easy.
Amith Skandhan, MD, SFHM, FACP, a hospitalist at Southeast Health Medical Center in Dothan, Ala., and a core faculty member in the internal medicine residency program at Alabama College of Osteopathic Medicine, said that implementing vaccine mandates will be more difficult in smaller health systems, in rural communities, and in states with lower vaccination rates and greater vaccine controversy.
Alabama has the lowest vaccination rates in the country, reflected in the recent rise in COVID cases and hospitalizations, even higher than during the surge of late 2020, Dr. Skandhan said. “In June we had one COVID patient in this hospital.” By late August the number was 119 COVID patients and climbing.
But where he works, in a health system where staffing is already spread thin, a vaccine mandate would be challenging. “What if our staff started leaving? It’s only 10 minutes from here to the Florida or Georgia border,” Dr. Skandhan said. Health care workers opposed to vaccinations would have the option of easily seeking work elsewhere.
When contacted for this article, he had been off work for several days but was mentally preparing himself to go back. “I’m not even following the [COVID-19] numbers but I am prepared for the worst. I know it will be mostly COVID. People just don’t realize what goes into this work.”
Dr. Skandhan, who said he was the third or fourth person in Alabama to receive the COVID vaccine, often finds himself feeling frustrated and angry – in the midst of a surge in cases that could have been prevented – that such a beneficial medical advance for bringing the pandemic under control became so politicized. “It is imperative that we find out why this mistrust exists and work to address it. It has to be done.”
Protecting health care professionals
On July 26, the Society of Hospital Medicine joined 50 other health care organizations including the American Medical Association, American Nurses Association, and American Academy of Pediatrics in advocating for all health care employers to require their employees to be vaccinated against COVID, in order to protect the safety of all patients and residents of health care facilities.2
“As an organization, we support vaccinating health care workers, including hospitalists, to help stop the spread of COVID-19 and the increasingly dominant Delta variant,” said SHM’s chief executive officer Eric E. Howell, MD, MHM, in a prepared statement. “We aim to uphold the highest standards among hospitalists and other health care providers to help protect our fellow health care professionals, our patients, and our communities.”
To that end, Dr. Skandhan has started conversations with hospital staff who he knows are not vaccinated. “For some, we’re not able to have a civil conversation, but in most cases I can help to persuade people.” The reasons people give for not getting vaccinated are not based in science, he said. “I am worried about the safety of our hospitalists and staff nurses.” But unvaccinated frontline workers are also putting their patients at risk. “Can we say why they’re hesitating? Can we have an honest discourse? If we can’t do that with our colleagues, how can we blame the patients?”
Dr. Skandhan encourages hospitalists to start simply in their own hospitals, trying to influence their own departments and colleagues. “If you can convince one or two more every week, you can start a chain reaction. Have that conversation. Use your trust.” For some hospitalized patients, the vaccination conversation comes too late, after their infection, but even some of them might consider obtaining it down the road or trying to persuade family members to get vaccinated.
Adult hospitalists, however, may not have received training in how to effectively address vaccine fears and misconceptions among their patients, he said. Because the patients they see in the hospital are already very sick, they don’t get a lot of practice talking about vaccines except, perhaps, for the influenza vaccine.
Pediatric hospitalists have more experience with such conversations involving their patients’ parents, Dr. Skandhan said. “It comes more naturally to them. We need to learn quickly from them about how to talk about vaccines with our patients.”
Pediatric training and experience
Anika Kumar, MD, FHM, FAAP, a pediatric hospitalist at the Cleveland Clinic and the pediatric editor of The Hospitalist, agrees that pediatricians and pediatric hospitalists often have received more training in how to lead vaccination conversations. She often talks about vaccines with the parents of hospitalized children relative to chicken pox, measles, and other diseases of childhood.
Pediatric hospitalists may also ask to administer the hepatitis B vaccine to newborn babies, along with other preventive treatments such as eye drops and vitamin K shots. “I often encourage the influenza vaccine prior to the patient’s hospital discharge, especially for kids with chronic conditions, asthma, diabetes, or premature birth. We talk about how the influenza vaccine isn’t perfect, but it helps to prevent more serious disease,” she said.
“A lot of vaccine hesitancy comes from misunderstandings about the role of vaccines,” she said. People forget that for years children have been getting vaccines before starting school. “Misinformation and opinions about vaccines have existed for decades. What’s new today is the abundance of sources for obtaining these opinions. My job is to inform families of scientific facts and to address their concerns.”
It has become more common recently for parents to say they don’t want their kids to get vaccinated, Dr. Kumar said. Another group is better described as vaccine hesitant and just needs more information. “I may not, by the time they leave the hospital, convince them to allow me to administer the vaccine. But in the discharge summary, I document that I had this conversation. I’ve done my due diligence and tried to start a larger dialogue. I say: ‘I encourage you to continue this discussion with the pediatrician you trust.’ I also communicate with the outpatient team,” she said.
“But it’s our responsibility, because we’re the ones seeing these patients, to do whatever we can to keep our patients from getting sick. A lot of challenging conversations we have with families are just trying to find out where they’re at with the issue – which can lead to productive dialogue.”
Ariel Carpenter, MD, a 4th-year resident in internal medicine and pediatrics at the University of Louisville (Ky.), and a future pediatric hospitalist, agreed that her combined training in med-peds has been helpful preparation for the vaccine conversation. That training has included techniques of motivational interviewing. In pediatrics, she explained, the communication is a little softer. “I try to approach my patients in a family-centered way.”
Dr. Carpenter recently wrote a personal essay for Louisville Medicine magazine from the perspective of growing up homeschooled by a mother who didn’t believe in vaccines.3 As a teenager, she independently obtained the complete childhood vaccine series so that she could do medical shadowing and volunteering. In medical school she became a passionate vaccine advocate, eventually persuading her mother to change her mind on the subject in time for the COVID vaccine.
“There’s not one answer to the vaccination dilemma,” she said. “Different approaches are required because there are so many different reasons for it. Based on my own life experience, I try to approach patients where they are – not from a place of data and science. What worked in my own family, and works with my patients, is first to establish trust. If they trust you, they’re more likely to listen. Simply ask their worries and concerns,” Dr. Carpenter said.
“A lot of them haven’t had the opportunity before to sit down with a physician they trust and have their worries listened to. They don’t feel heard in our medical system. So I remind myself that I need to understand my patients first – before inserting myself into the conversation.”
Many patients she sees are in an information bubble, with a very different understanding of the issue than their doctors. “A lot of well-meaning people feel they are making the safer choice. Very few truly don’t care about protecting others. But they don’t feel the urgency about that and see the vaccine as the scarier option right now.”
Frontline vaccine advocates
Hospitalists are the frontline advocates within their hospital system, in a position to lead, so they need to make vaccines a priority, Dr. Carpenter said. They should also make sure that their hospitals have ready access to the vaccine, so patients who agree to receive it are able to get it quickly. “In our hospital they can get the shot within a few hours if the opportunity arises. We stocked the Johnson & Johnson vaccine so that they wouldn’t have to connect with another health care provider in order to get a second dose.”
Hospitals should also invest in access to vaccine counseling training and personnel. “Fund a nurse clinician who can screen and counsel hospitalized patients for vaccination. If they meet resistance, they can then refer to the dedicated physician of the day to have the conversation,” she said. “But if we don’t mention it, patients will assume we don’t feel strongly about it.”
Because hospitalists are front and center in treating COVID, they need to be the experts and the people offering guidance, said Shyam Odeti, MD, SFHM, FAAFP, section chief for hospital medicine at the Carilion Clinic in Roanoke, Va. “What we’re trying to do is spread awareness. We educated physician groups, learners, and clinical teams during the initial phase, and now mostly patients and their families.” COVID vaccine reluctance is hard to overcome, Dr. Odeti said. People feel the vaccine was developed very quickly. But there are different ways to present it.
“Like most doctors, I thought people would jump on a vaccine to get past the pandemic. I was surprised and then disappointed. Right now, the pandemic is among the unvaccinated. So we face these encounters, and we’re doing our best to overcome the misinformation. My organization is 100% supportive. We talk about these issues every day.”
Carilion, effective Oct. 1, has required unvaccinated employees to get weekly COVID tests and wear an N95 mask while working, and has developed Facebook pages, other social media, and an Internet presence to address these issues. “We’ve gone to the local African-American community with physician leaders active in that community. We had a Spanish language roundtable,” Dr. Odeti said.
Dr. Skandhan reported that the Wiregrass regional chapter of SHM recently organized a successful statewide community educational event aimed at empowering community leaders to address vaccine misinformation and mistrust. “We surveyed religious leaders and pastors regarding the causes of vaccine hesitancy and reached out to physicians active in community awareness.” Based on that input, a presentation by the faith leaders was developed. Legislators from the Alabama State Senate’s Healthcare Policy Committee were also invited to the presentation and discussion.
Trying to stay positive
It’s important to try to stay positive, Dr. Odeti said. “We have to be empathetic with every patient. We have to keep working at this, since there’s no way out of the pandemic except through vaccinations. But it all creates stress for hospitalists. Our job is made significantly more difficult by the vaccine controversy.”
Jennifer Cowart, MD, a hospitalist at Mayo Clinic in Jacksonville, Fla., has been outspoken in her community about vaccination and masking issues, talking to reporters, attending rallies and press conferences, posting on social media, and speaking in favor of mask policies at a local school board meeting. She is part of an informal local group called Doctors Fighting COVID, which meets online to strategize how to share its expertise, including writing a recent letter about masks to Jacksonville’s mayor.
“In July, when we saw the Delta variant surging locally, we held a webinar via local media, taking calls about the vaccine from the community. I’m trying not to make this a political issue, but we are health officials.” Dr. Cowart said she also tries not to raise her voice when speaking with vaccine opponents and tries to remain empathetic. “Even though inwardly I’m screaming, I try to stay calm. The misinformation is real. People are afraid and feeling pressure. I do my best, but I’m human, too.”
Hospitalists need to pull whatever levers they can to help advance understanding of vaccines, Dr. Cowart said. “In the hospital, our biggest issue is time. We often don’t have it, with a long list of patients to see. But every patient encounter is an opportunity to talk to patients, whether they have COVID or something else.” Sometimes, she might go back to a patient’s room after rounds to resume the conversation.
Hospital nurses have been trained and entrusted to do tobacco abatement counseling, she said, so why not mobilize them for vaccine education? “Or respiratory therapists, who do inhaler training, could talk about what it’s like to care for COVID patients. There’s a whole bunch of staff in the hospital who could be mobilized,” she said.
“I feel passionate about vaccines, as a hospitalist, as a medical educator, as a daughter, as a responsible member of society,” said Eileen Barrett, MD, MPH, SFHM, MACP, director of continuing medical education at the University of New Mexico, Albuquerque. “I see this as a personal and societal responsibility. When I speak about the vaccine among groups of doctors, I say we need to stay in our lane regarding our skills at interpreting the science and not undermining it.”
Some health care worker hesitancy is from distrust of pharmaceutical companies, or of federal agencies, she said. “Our research has highlighted to me the widespread inequity issues in our health care system. We should also take a long, hard look at how we teach the scientific method to health professionals. That will be part of a pandemic retrospective.”
Sometimes with people who are vaccine deliberative, whether health care workers or patients, there is a small window of opportunity. “We need to hear people and respond to them as people. Then, if they are willing to get vaccinated, we need to accomplish that as quickly and easily as possible,” Dr. Barrett said. “I see them make a face and say, ‘Well, okay, I’ll do it.’ We need to get the vaccine to them that same day. We should be able to accomplish that.”
References
1. Gamble M. 30% of US hospitals mandate vaccination for employment. Becker’s Hospital Review. 2021 Aug 13. www.beckershospitalreview.com/workforce/covid-19-vaccination-needed-to-work-at-30-of-us-hospitals.html .
2. Society of Hospital Medicine signs on to joint statement in support of health worker COVID-19 vaccine mandates. Press release. 2021 Jul 26. www.hospitalmedicine.org/news-publications/press-releases/society-of-hospital-medicine-signs-on-to-joint-statement-of-support-of-health-worker-covid-19-vaccine-mandates/.
3. Carpenter A. A physician’s lessons from an unvaccinated childhood. Louisville Medicine. 2021 July;69(2):26-7. https://viewer.joomag.com/louisville-medicine-volume-69-issue-2/0045988001624974172?short&.
Lessons for hospitalists from the vaccination controversy
1. Remain up-to-date on information about the COVID infection, its treatment, and vaccination efficacy data.
2. Hospitalists should take advantage of their positions to lead conversations in their facilities about the importance of COVID vaccinations.
3. Other professionals in the hospital, with some additional training and support, could take on the role of providing vaccine education and support – with a physician to back them up on difficult cases.
4. It’s important to listen to people’s concerns, try to build trust, and establish dialogue before starting to convey a lot of information. People need to feel heard.
5. If you are successful in persuading someone to take the vaccine, a shot should be promptly and easily accessible to them.
6. Pediatric hospitalists may have more experience and skill with vaccine discussions, which they should share with their peers who treat adults.
Flu shot highly recommended this year
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
With the Delta variant of COVID-19 still raging in the United States and ICUs in parts of the country filled with patients with the coronavirus, experts are voicing concern about the added risk of a difficult flu season.
Two mathematical models are predicting a big rebound in the number and severity of flu cases in the 2021-22 season after 2020-2021’s flu season failed to show up when public health measures brought in to control COVID-19 seemed to have the added benefit of stopping the flu.
But both analyses, posted to the medRxiv preprint server and not yet peer reviewed by other experts, have come to the same conclusion: The flu could make a comeback this year.
In the worst-case scenario, the United States could see an extra 300,000-400,000 hospitalizations from the flu – almost double the usual number – according to senior study author Mark Roberts, MD, director of the Public Health Dynamics Laboratory at the University of Pittsburgh. These numbers could be a disaster in areas where hospitals are already filled with COVID-19 patients.
Waning natural immunity in the public because of 2020-2021’s missing flu season could make people, especially young children, more likely to get the virus.
“Usually, a combination of natural immunity and vaccination helps tamp down seasonal influenza,” said Dr. Roberts. “If we don’t have the first part, we’ll have to rely more on the vaccine.”
In a typical year, about half of Americans get the flu shot. The new mathematical models predict that the vaccination rate would need to rise to about 75% to avoid the extra hospitalizations. But even a 10% increase in vaccination rates could reduce hospitalizations by 6%-46%, depending on what strains are dominant.
Usually, the Southern Hemisphere flu season, from February to August, helps show what the Northern Hemisphere can expect over the coming winter. But with strict COVID-19 measures and limits on international travel still in place in countries like Australia and New Zealand and much of South America, it has been another record-low year for flu infections, said Ian Barr, PhD, deputy director of the World Health Organization’s Collaborating Center for Reference and Research on Influenza in Melbourne.
Australia detected only around 500 cases in 2021, compared with about 300,000 in a normal year, and recorded no hospitalizations or deaths from the flu. New Zealand recorded just two cases.
“I’ve never seen anything like this,” Dr. Barr said.
In Australia, the mild flu season led to fewer people getting their flu shot than usual. The rate fell from around 50% to just 33%, said Dr. Barr. “If that happens in the U.S., the population will be even more vulnerable because there has been almost no flu for more than 12 months,” he said.
Both Dr. Roberts and Dr. Barr say it is vital that as many people as possible get vaccinated during the upcoming flu season, especially children who will have almost no natural immunity to the virus.
“The vaccine is our best weapon against the flu, especially for the most at-risk groups,” said Dr. Barr.
Other parts of the world had mixed results. India saw a high number of flu cases, while neighboring Sri Lanka had very few. West Africa also saw quite a high level of circulating virus. Overall, the flu was detected in 45 countries during the Southern Hemisphere season, less than half of what might be expected in a normal year, said Dr. Barr.
Despite the overall low numbers, the WHO saw enough in the data to make two changes to 2022’s Southern Hemisphere vaccine formulation at its meeting on Sept. 24, after changing just one of the strains for the Northern Hemisphere vaccine at its meeting in February.
The CDC recommends that everyone 6 months or older get the flu shot, with few exceptions.
A version of this article first appeared on WebMD.com.
Worried parents scramble to vaccinate kids despite FDA guidance
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
One week after reporting promising results from the trial of their COVID-19 vaccine in children ages 5-11, Pfizer and BioNTech announced they’d submitted the data to the Food and Drug Administration. But that hasn’t stopped some parents from discreetly getting their children under age 12 vaccinated.
“The FDA, you never want to get ahead of their judgment,” Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, told MSNBC on Sept. 28. “But I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the okay so that we can start vaccinating children, hopefully before the end of October.”
Lying to vaccinate now
More than half of all parents with children under 12 say they plan to get their kids vaccinated, according to a Gallup poll.
And although the FDA and the American Academy of Pediatrics have warned against it, some parents whose children can pass for 12 have lied to get them vaccinated already.
Dawn G. is a mom of two in southwest Missouri, where less than 45% of the population has been fully vaccinated. Her son turns 12 in early October, but in-person school started in mid-August.
“It was scary, thinking of him going to school for even 2 months,” she said. “Some parents thought their kid had a low chance of getting COVID, and their kid died. Nobody expects it to be them.”
In July, she and her husband took their son to a walk-in clinic and lied about his age.
“So many things can happen, from bullying to school shootings, and now this added pandemic risk,” she said. “I’ll do anything I can to protect my child, and a birthdate seems so arbitrary. He’ll be 12 in a matter of weeks. It seems ridiculous that that date would stop me from protecting him.”
In northern California, Carrie S. had a similar thought. When the vaccine was authorized for children ages 12-15 in May, the older of her two children got the shot right away. But her youngest doesn’t turn 12 until November.
“We were tempted to get the younger one vaccinated in May, but it didn’t seem like a rush. We were willing to wait to get the dosage right,” she ssaid. “But as Delta came through, there were no options for online school, the CDC was dropping mask expectations –it seemed like the world was ready to forget the pandemic was happening. It seemed like the least-bad option to get her vaccinated so she could go back to school, and we could find some balance of risk in our lives.”
Adult vs. pediatric doses
For now, experts advise against getting younger children vaccinated, even those who are the size of an adult, because of the way the human immune system develops.
“It’s not really about size,” said Anne Liu, MD, an immunologist and pediatrics professor at Stanford (Calif.) University. “The immune system behaves differently at different ages. Younger kids tend to have a more exuberant innate immune system, which is the part of the immune system that senses danger, even before it has developed a memory response.”
The adult Pfizer-BioNTech vaccine contains 30 mcg of mRNA, while the pediatric dose is just 10 mcg. That smaller dose produces an immune response similar to what’s seen in adults who receive 30 mcg, according to Pfizer.
“We were one of the sites that was involved in the phase 1 trial, a lot of times that’s called a dose-finding trial,” said Michael Smith, MD, a coinvestigator for the COVID vaccine trials done at Duke University. “And basically, if younger kids got a higher dose, they had more of a reaction, so it hurt more. They had fever, they had more redness and swelling at the site of the injection, and they just felt lousy, more than at the lower doses.”
At this point, with Pfizer’s data showing that younger children need a smaller dose, it doesn’t make sense to lie about your child’s age, said Dr. Smith.
“If my two options were having my child get the infection versus getting the vaccine, I’d get the vaccine. But we’re a few weeks away from getting the lower dose approved in kids,” he said. “It’s certainly safer. I don’t expect major, lifelong side effects from the higher dose, but it’s going to hurt, your kid’s going to have a fever, they’re going to feel lousy for a couple days, and they just don’t need that much antigen.”
A version of this article first appeared on WebMD.com.
Children and COVID: New cases topped 200,000 after 3 weeks of declines
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.
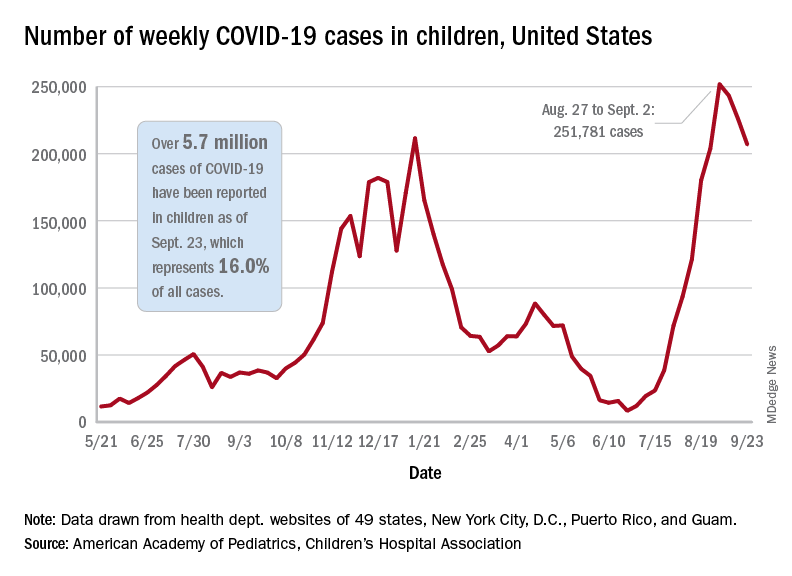
based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.

based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Weekly COVID-19 cases in children dropped again, but the count remained above 200,000 for the fifth consecutive week, according to the American Academy of Pediatrics and the Children’s Hospital Association.

based on the data in the AAP/CHA joint weekly report on COVID in children.
In the most recent week, Sept. 17-23, there were almost 207,000 new cases of COVID-19 in children, which represented 26.7% of all cases reported in the 46 states that are currently posting data by age on their COVID dashboards, the AAP and CHA said. (New York has never reported such data by age, and Alabama, Nebraska, and Texas have not updated their websites since July 29, June 24, and Aug. 26, respectively.)
The decline in new vaccinations among children, however, began before the summer surge in new cases hit its peak – 251,781 during the week of Aug. 27 to Sept. 2 – and has continued for 7 straight weeks in children aged 12-17 years, based on data from the Centers for Disease Control and Prevention.
There were about 172,000 COVID vaccine initiations in children aged 12-17 for the week of Sept. 21-27, the lowest number since April, before it was approved for use in 12- to 15-year-olds. That figure is down by almost a third from the previous week and by more than two-thirds since early August, just before the decline in vaccinations began, according to the CDC’s COVID Data Tracker.
The cumulative vaccine situation looks like this: Just over 13 million children under age 18 years have received at least one dose as of Sept. 27, and almost 10.6 million are fully vaccinated. By age group, 53.9% of 12- to 15-year-olds and 61.6% of 16- to 17-year-olds have received at least one dose, with corresponding figures of 43.3% and 51.3% for full vaccination, the CDC said.
COVID-related hospital admissions also continue to fall after peaking at 0.51 children aged 0-17 per 100,000 population on Sept. 4. The admission rate was down to 0.45 per 100,000 as of Sept. 17, and the latest 7-day average (Sept. 19-25) was 258 admissions, compared with a peak of 371 for the week of Aug. 29 to Sept. 4, the CDC reported.
“Although we have seen slight improvements in COVID-19 volumes in the past week, we are at the beginning of an anticipated increase in” multi-inflammatory syndrome in children, Margaret Rush, MD, president of Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn., said at a recent hearing of the House Committee on Energy and Commerce’s Oversight subcommittee. That increase would be expected to produce “a secondary wave of seriously ill children 3-6 weeks after acute infection peaks in the community,” the American Hospital Association said.
Meanwhile, Dr. Rush noted, there are signs that seasonal viruses are coming into play. “With the emergence of the Delta variant, we’ve experienced a steep increase in COVID-19 hospitalizations among children on top of an early surge of [respiratory syncytial virus], a serious respiratory illness we usually see in the winter months,” she said in a prepared statement before her testimony.
Polyethylene glycol linked to rare allergic reactions seen with mRNA COVID-19 vaccines
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A common inert ingredient may be the culprit behind the rare allergic reactions reported among individuals who have received mRNA COVID-19 vaccines, according to investigators at a large regional health center that was among the first to administer the shots.
Blood samples from 10 of 11 individuals with suspected allergic reactions reacted to polyethylene glycol (PEG), a component of both the Pfizer and Moderna mRNA vaccines, according to a report in JAMA Network Open.
In total, only 22 individuals had suspected allergic reactions out of nearly 39,000 mRNA COVID-19 vaccine doses administered, the investigators reported, noting that the reactions were generally mild and all fully resolved.
Those findings should be reassuring to individuals who are reticent to sign up for a COVID-19 vaccine because of fear of an allergic reaction, said study senior author Kari Nadeau, MD, PhD, director of the Parker Center for Allergy and Asthma Research at Stanford (Calif.) University.
“We’re hoping that this word will get out and then that the companies could also think about making vaccines that have other products in them that don’t include polyethylene glycol,” Dr. Nadeau said in an interview.
PEG is a compound used in many products, including pharmaceuticals, cosmetics, and food. In the mRNA COVID-19 vaccines, PEG serves to stabilize the lipid nanoparticles that help protect and transport mRNA. However, its use in this setting has been linked to allergic reactions in this and previous studies.
No immunoglobulin E (IgE) antibodies to PEG were detected among the 22 individuals with suspected allergic reactions to mRNA COVID-19 vaccine, but PEG immunoglobulin G (IgG) was present. That suggests non-IgE mediated allergic reactions to PEG may be implicated for the majority of cases, Dr. Nadeau said.
This case series provides interesting new evidence to confirm previous reports that a mechanism other than the classic IgE-mediated allergic response is behind the suspected allergic reactions that are occurring after mRNA COVID-19 vaccine, said Aleena Banerji, MD, associate professor at Harvard Medical School, Boston, and clinical director of the Drug Allergy Program at Massachusetts General Hospital.
“We need to further understand the mechanism of these reactions, but what we know is that IGE mediated allergy to excipients like PEG is probably not the main cause,” Dr. Banerji, who was not involved in the study, said in an interview.
In a recent research letter published in JAMA Internal Medicine, Dr. Banerji and coauthors reported that all individuals with immediate suspected allergic reactions to mRNA COVID-19 vaccine went on to tolerate the second dose, with mild symptoms reported in the minority of patients (32 out of 159, or about 20%).
“Again, that is very consistent with not having an IgE-mediated allergy, so it seems to all be fitting with that picture,” Dr. Banerji said.
The case series by Dr. Nadeau and coauthors was based on review of nearly 39,000 mRNA COVID-19 vaccine doses administered between December 18, 2020 and January 26, 2021. Most mRNA vaccine recipients were Stanford-affiliated health care workers, according to the report.
Among recipients of those doses, they identified 148 individuals who had anaphylaxis-related ICD-10 codes recorded over the same time period. In a review of medical records, investigators pinpointed 22 individuals as having suspected allergy and invited them to participate in follow-up allergy testing.
A total of 11 individuals underwent skin prick testing, but none of them tested positive to PEG or to polysorbate 80, another excipient that has been linked to vaccine-related allergic reactions. One of the patients tested positive to the same mRNA vaccine they had previously received, according to the report.
Those same 11 individuals also underwent basophil activation testing (BAT). In contrast to the skin testing results, BAT results were positive for PEG in 10 of 11 cases (or 91%) and positive for their administered vaccine in all 11 cases, the report shows.
High levels of IgG to PEG were identified in blood samples of individuals with an allergy to the vaccine. Investigators said it’s possible that the BAT results were activated due to IgG via complement activation–related pseudoallergy, or CARPA, as has been hypothesized by some other investigators.
The negative skin prick testing results for PEG, which contrast with the positive BAT results to PEG, suggest that the former may not be appropriate for use as a predictive marker of potential vaccine allergy, according to Dr. Nadeau.
“The take-home message for doctors is to be careful,” she said. “Don’t assume that just because the person skin-tests negative to PEG or to the vaccine itself that you’re out of the woods, because the skin test would be often negative in those scenarios.”
The study was supported by a grants from the Asthma and Allergic Diseases Cooperative Research Centers, a grant from the National Institutes of Health, the National Institute of Allergy and Infectious Disease SARS Vaccine study, the Parker Foundation, the Crown Foundation, and the Sunshine Foundation. Dr. Nadeau reports numerous conflicts with various sources in the industry. Dr. Banerji has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.