User login
FDA approves higher dose brigatinib tablet for advanced ALK+ NSCLC
The Food and Drug Administration has approved 180 mg brigatinib (Alunbrig) tablets for treatment of anaplastic lymphoma kinase–positive (ALK+) metastatic non–small cell lung cancer (NSCLC), expanding on previously available 30-mg tablets.*
“With the approval of a 180-mg tablet, Alunbrig has become the only ALK inhibitor available as a one tablet per day dose that can be taken with or without food,” Ryan Cohlhepp, PharmD, vice president, U.S. Commercial, at Takeda Oncology said in a press release.
Approval of the regimen was based on objective response in the ongoing, two-arm, open-label, multicenter phase 2 ALTA trial, which enrolled 222 patients with metastatic or locally advanced ALK+ NSCLC who had progressed on crizotinib. Patients were randomized to brigatinib orally either 90 mg once daily or 180 mg once daily following a 7-day lead-in at 90 mg once daily. Of those in the 180-mg arm, 53% had an objective response, compared with 48% in the 90-mg arm.
Adverse reactions occurred in 40% of the patients in the 180-mg arm, compared with 38% in the 90-mg arm. The most common serious adverse reactions were pneumonia and interstitial lung disease/pneumonitis. Fatal adverse reactions occurred in 3.7% of the patients, caused by pneumonia (two patients), sudden death, dyspnea, respiratory failure, pulmonary embolism, bacterial meningitis and urosepsis (one patient each).
The ALTA trial is ongoing, and updated results will be presented at the World Conference on Lung Cancer in Yokohama, Japan, on Oct. 15-18, the company said in the press release.
*Correction, 10/4/17: An earlier version of this article misstated the previously available tablet sizes.
The Food and Drug Administration has approved 180 mg brigatinib (Alunbrig) tablets for treatment of anaplastic lymphoma kinase–positive (ALK+) metastatic non–small cell lung cancer (NSCLC), expanding on previously available 30-mg tablets.*
“With the approval of a 180-mg tablet, Alunbrig has become the only ALK inhibitor available as a one tablet per day dose that can be taken with or without food,” Ryan Cohlhepp, PharmD, vice president, U.S. Commercial, at Takeda Oncology said in a press release.
Approval of the regimen was based on objective response in the ongoing, two-arm, open-label, multicenter phase 2 ALTA trial, which enrolled 222 patients with metastatic or locally advanced ALK+ NSCLC who had progressed on crizotinib. Patients were randomized to brigatinib orally either 90 mg once daily or 180 mg once daily following a 7-day lead-in at 90 mg once daily. Of those in the 180-mg arm, 53% had an objective response, compared with 48% in the 90-mg arm.
Adverse reactions occurred in 40% of the patients in the 180-mg arm, compared with 38% in the 90-mg arm. The most common serious adverse reactions were pneumonia and interstitial lung disease/pneumonitis. Fatal adverse reactions occurred in 3.7% of the patients, caused by pneumonia (two patients), sudden death, dyspnea, respiratory failure, pulmonary embolism, bacterial meningitis and urosepsis (one patient each).
The ALTA trial is ongoing, and updated results will be presented at the World Conference on Lung Cancer in Yokohama, Japan, on Oct. 15-18, the company said in the press release.
*Correction, 10/4/17: An earlier version of this article misstated the previously available tablet sizes.
The Food and Drug Administration has approved 180 mg brigatinib (Alunbrig) tablets for treatment of anaplastic lymphoma kinase–positive (ALK+) metastatic non–small cell lung cancer (NSCLC), expanding on previously available 30-mg tablets.*
“With the approval of a 180-mg tablet, Alunbrig has become the only ALK inhibitor available as a one tablet per day dose that can be taken with or without food,” Ryan Cohlhepp, PharmD, vice president, U.S. Commercial, at Takeda Oncology said in a press release.
Approval of the regimen was based on objective response in the ongoing, two-arm, open-label, multicenter phase 2 ALTA trial, which enrolled 222 patients with metastatic or locally advanced ALK+ NSCLC who had progressed on crizotinib. Patients were randomized to brigatinib orally either 90 mg once daily or 180 mg once daily following a 7-day lead-in at 90 mg once daily. Of those in the 180-mg arm, 53% had an objective response, compared with 48% in the 90-mg arm.
Adverse reactions occurred in 40% of the patients in the 180-mg arm, compared with 38% in the 90-mg arm. The most common serious adverse reactions were pneumonia and interstitial lung disease/pneumonitis. Fatal adverse reactions occurred in 3.7% of the patients, caused by pneumonia (two patients), sudden death, dyspnea, respiratory failure, pulmonary embolism, bacterial meningitis and urosepsis (one patient each).
The ALTA trial is ongoing, and updated results will be presented at the World Conference on Lung Cancer in Yokohama, Japan, on Oct. 15-18, the company said in the press release.
*Correction, 10/4/17: An earlier version of this article misstated the previously available tablet sizes.
Liquid biopsy predicts checkpoint inhibitor response
The overall response rate to immune checkpoint inhibitors was 45% among cancer patients who had more than three variants of unknown significance in their circulating tumor DNA; among those with three or fewer, the response rate was 15%, according to a University of California, San Diego, investigation with 69 subjects.
Higher mutation burdens in circulating tumor DNA (ctDNA) also correlated with improved progression-free and overall survival across 20 cancer types, the investigators reported (Clin Cancer Res. 2017 Oct. 1. doi: 10.1158/1078-0432.CCR-17-1439).
Tumor mutation burdens can predict response to checkpoint inhibitors, but they are usually assessed by tissue biopsy, which is costly and invasive. The findings suggest that blood tests could replace tissue biopsies to green-light immune checkpoint inhibitor treatment.
“Our current results may be clinically exploitable. ... Liquid biopsies that assess blood-derived ctDNA are noninvasive, easily acquired, and inexpensive. The ctDNA derived from blood may also represent shed DNA from multiple metastatic sites, whereas tissue genomics reflects only the piece of tissue removed,” said investigators led by Yulian Khagi, MD, a hematology-oncology fellow at the university.
In a press statement, Dr. Khagi said “If verified by further studies, clinicians will be able to utilize the ... results of this simple blood test to make determinations about whether to use checkpoint inhibitor–based immune therapy in a variety of tumor types.”
The 69 patients were a median of 56 years old, and 43 (62.3%) were men. Melanoma, lung cancer, and head and neck cancer were the most common malignancies. The majority of patients had anti–PD-1 or PD-L1 monotherapy.
For most patients, blood samples were drawn a month or 2 before treatment. Next-generation sequencing (Guardant360) was done on ctDNA to detect alterations in cancer genes. Of the 69 patients, 20 (29%) had more than three variants of unknown significance (VUS); the rest had three or fewer.
The median overall survival was 15.3 months from the start of immunotherapy. For patients with three or fewer VUS, median overall survival was 10.72 months; for patients with more, median overall survival could not be calculated because more than half were alive at the study’s conclusion.
Median progression-fee survival was 2.07 months with three or fewer VUS, versus 3.84 months with more. The findings were statistically significant.
Similar results were found when all genomic alterations, not just VUS, were examined and dichotomized as six or more versus fewer than six.
“The number of genes assayed in our ctDNA analysis was only between 54 and 70. Unlike targeted NGS [next-generation sequencing] of tumor tissue, which often tests for hundreds of genes and allows a relatively accurate estimate of total mutational burden, targeted NGS of plasma ctDNA provides only a limited snapshot of the cancer genome. More extensive ctDNA gene panels merit investigation to determine if they increase the correlative value of our findings,” the investigators said.
The work was funded by the Joan and Irwin Jacobs Fund and the National Cancer Institute. Dr. Khagi had no industry disclosures. Three authors reported financial ties to a number of companies, including Boehringer, Merck, Guardant, and Pfizer. The senior author has ownership interests in CureMatch.
The overall response rate to immune checkpoint inhibitors was 45% among cancer patients who had more than three variants of unknown significance in their circulating tumor DNA; among those with three or fewer, the response rate was 15%, according to a University of California, San Diego, investigation with 69 subjects.
Higher mutation burdens in circulating tumor DNA (ctDNA) also correlated with improved progression-free and overall survival across 20 cancer types, the investigators reported (Clin Cancer Res. 2017 Oct. 1. doi: 10.1158/1078-0432.CCR-17-1439).
Tumor mutation burdens can predict response to checkpoint inhibitors, but they are usually assessed by tissue biopsy, which is costly and invasive. The findings suggest that blood tests could replace tissue biopsies to green-light immune checkpoint inhibitor treatment.
“Our current results may be clinically exploitable. ... Liquid biopsies that assess blood-derived ctDNA are noninvasive, easily acquired, and inexpensive. The ctDNA derived from blood may also represent shed DNA from multiple metastatic sites, whereas tissue genomics reflects only the piece of tissue removed,” said investigators led by Yulian Khagi, MD, a hematology-oncology fellow at the university.
In a press statement, Dr. Khagi said “If verified by further studies, clinicians will be able to utilize the ... results of this simple blood test to make determinations about whether to use checkpoint inhibitor–based immune therapy in a variety of tumor types.”
The 69 patients were a median of 56 years old, and 43 (62.3%) were men. Melanoma, lung cancer, and head and neck cancer were the most common malignancies. The majority of patients had anti–PD-1 or PD-L1 monotherapy.
For most patients, blood samples were drawn a month or 2 before treatment. Next-generation sequencing (Guardant360) was done on ctDNA to detect alterations in cancer genes. Of the 69 patients, 20 (29%) had more than three variants of unknown significance (VUS); the rest had three or fewer.
The median overall survival was 15.3 months from the start of immunotherapy. For patients with three or fewer VUS, median overall survival was 10.72 months; for patients with more, median overall survival could not be calculated because more than half were alive at the study’s conclusion.
Median progression-fee survival was 2.07 months with three or fewer VUS, versus 3.84 months with more. The findings were statistically significant.
Similar results were found when all genomic alterations, not just VUS, were examined and dichotomized as six or more versus fewer than six.
“The number of genes assayed in our ctDNA analysis was only between 54 and 70. Unlike targeted NGS [next-generation sequencing] of tumor tissue, which often tests for hundreds of genes and allows a relatively accurate estimate of total mutational burden, targeted NGS of plasma ctDNA provides only a limited snapshot of the cancer genome. More extensive ctDNA gene panels merit investigation to determine if they increase the correlative value of our findings,” the investigators said.
The work was funded by the Joan and Irwin Jacobs Fund and the National Cancer Institute. Dr. Khagi had no industry disclosures. Three authors reported financial ties to a number of companies, including Boehringer, Merck, Guardant, and Pfizer. The senior author has ownership interests in CureMatch.
The overall response rate to immune checkpoint inhibitors was 45% among cancer patients who had more than three variants of unknown significance in their circulating tumor DNA; among those with three or fewer, the response rate was 15%, according to a University of California, San Diego, investigation with 69 subjects.
Higher mutation burdens in circulating tumor DNA (ctDNA) also correlated with improved progression-free and overall survival across 20 cancer types, the investigators reported (Clin Cancer Res. 2017 Oct. 1. doi: 10.1158/1078-0432.CCR-17-1439).
Tumor mutation burdens can predict response to checkpoint inhibitors, but they are usually assessed by tissue biopsy, which is costly and invasive. The findings suggest that blood tests could replace tissue biopsies to green-light immune checkpoint inhibitor treatment.
“Our current results may be clinically exploitable. ... Liquid biopsies that assess blood-derived ctDNA are noninvasive, easily acquired, and inexpensive. The ctDNA derived from blood may also represent shed DNA from multiple metastatic sites, whereas tissue genomics reflects only the piece of tissue removed,” said investigators led by Yulian Khagi, MD, a hematology-oncology fellow at the university.
In a press statement, Dr. Khagi said “If verified by further studies, clinicians will be able to utilize the ... results of this simple blood test to make determinations about whether to use checkpoint inhibitor–based immune therapy in a variety of tumor types.”
The 69 patients were a median of 56 years old, and 43 (62.3%) were men. Melanoma, lung cancer, and head and neck cancer were the most common malignancies. The majority of patients had anti–PD-1 or PD-L1 monotherapy.
For most patients, blood samples were drawn a month or 2 before treatment. Next-generation sequencing (Guardant360) was done on ctDNA to detect alterations in cancer genes. Of the 69 patients, 20 (29%) had more than three variants of unknown significance (VUS); the rest had three or fewer.
The median overall survival was 15.3 months from the start of immunotherapy. For patients with three or fewer VUS, median overall survival was 10.72 months; for patients with more, median overall survival could not be calculated because more than half were alive at the study’s conclusion.
Median progression-fee survival was 2.07 months with three or fewer VUS, versus 3.84 months with more. The findings were statistically significant.
Similar results were found when all genomic alterations, not just VUS, were examined and dichotomized as six or more versus fewer than six.
“The number of genes assayed in our ctDNA analysis was only between 54 and 70. Unlike targeted NGS [next-generation sequencing] of tumor tissue, which often tests for hundreds of genes and allows a relatively accurate estimate of total mutational burden, targeted NGS of plasma ctDNA provides only a limited snapshot of the cancer genome. More extensive ctDNA gene panels merit investigation to determine if they increase the correlative value of our findings,” the investigators said.
The work was funded by the Joan and Irwin Jacobs Fund and the National Cancer Institute. Dr. Khagi had no industry disclosures. Three authors reported financial ties to a number of companies, including Boehringer, Merck, Guardant, and Pfizer. The senior author has ownership interests in CureMatch.
FROM CLINICAL CANCER RESEARCH
Key clinical point:
Major finding: The overall response rate to immune checkpoint inhibitors was 45% among cancer patients who had more than three variants of unknown significance in their circulating tumor DNA; among those with three or fewer, the response rate was 15%.
Data source: Review of 69 cancer patients.
Disclosures: The work was funded by the Joan and Irwin Jacobs Fund and the National Cancer Institute. Three investigators reported financial ties to a number of companies, including Boehringer, Merck, Guardant, and Pfizer. The senior author has ownership interests in CureMatch.
LUME-Mesa trial: Nintedanib improves PFS in mesothelioma
CHICAGO – Adding the oral kinase inhibitor nintedanib to pemetrexed/cisplatin resulted in substantial improvements in outcomes in patients with unresectable malignant pleural mesothelioma in phase 2 of the randomized, placebo-controlled phase 2/3 LUME-Meso study.
The effects were particularly pronounced among those with epithelioid histology, Jose Barrueco, PhD, of Boehringer Ingelheim Pharmaceuticals, Ridgefield, Conn., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Progression-free survival – the primary endpoint of the study – was improved in 44 patients randomized to receive up to six cycles of pemetrexed/cisplatin plus nintedanib, compared with 43 patients who received pemetrexed/cisplatin plus placebo (median 9.4 vs. 5.7 months; hazard ratio, 0.54), Dr. Barrueco said.
In the 89% of patients with epithelioid malignant pleural mesothelioma, progression-free survival was a median of 9.7 vs. 5.7 months with nintedanib vs. placebo (HR, 0.49).
There was a trend toward improved overall survival in the nintedanib group vs. the placebo group, (median 18.3 vs. 14.2 months; HR, 0.77; P = .319), and overall survival was slightly better in those with epithelioid histology (median 20.6 vs. 15.2 months ; HR, 0.70; P = .197).
Consistent with these results, the adjusted mean change in forced vital capacity at cycle eight also favored nintedanib over placebo (+10.0 vs. +2.8 for a mean treatment difference of 7.2 overall, and +14.1 vs. +4.2 for a mean treatment difference of 9.9 in those with epithelioid histology).
“Overall frequency of adverse events was consistent with the known safety profile of nintedanib,” Dr. Barrueco said, noting that most adverse events were reversible with dose reduction.
Study participants were chemotherapy-naive patients with a mean age of 67 years and Eastern Cooperative Oncology Group performance status of 0-1. They received pemetrexed at a dose of 500 mg/m2 and cisplatin at a dose of 75 mg/m2 on day 1 plus either nintedanib at a dose of 200 mg twice daily on days 2-21 or placebo, followed by monotherapy with nintedanib or placebo until progression or unacceptable toxicity.
“In conclusion, nintedanib plus pemetrexed/cisplatin demonstrated a signal for clinical benefit in the first-time treatment of patients with malignant pleural mesothelioma. This was evident in all endpoints of the trial, and consistently showed benefit for the nintedanib group,” Mr. Barrueco said, noting that phase 3 of the LUME-Meso study is now recruiting.
CHICAGO – Adding the oral kinase inhibitor nintedanib to pemetrexed/cisplatin resulted in substantial improvements in outcomes in patients with unresectable malignant pleural mesothelioma in phase 2 of the randomized, placebo-controlled phase 2/3 LUME-Meso study.
The effects were particularly pronounced among those with epithelioid histology, Jose Barrueco, PhD, of Boehringer Ingelheim Pharmaceuticals, Ridgefield, Conn., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Progression-free survival – the primary endpoint of the study – was improved in 44 patients randomized to receive up to six cycles of pemetrexed/cisplatin plus nintedanib, compared with 43 patients who received pemetrexed/cisplatin plus placebo (median 9.4 vs. 5.7 months; hazard ratio, 0.54), Dr. Barrueco said.
In the 89% of patients with epithelioid malignant pleural mesothelioma, progression-free survival was a median of 9.7 vs. 5.7 months with nintedanib vs. placebo (HR, 0.49).
There was a trend toward improved overall survival in the nintedanib group vs. the placebo group, (median 18.3 vs. 14.2 months; HR, 0.77; P = .319), and overall survival was slightly better in those with epithelioid histology (median 20.6 vs. 15.2 months ; HR, 0.70; P = .197).
Consistent with these results, the adjusted mean change in forced vital capacity at cycle eight also favored nintedanib over placebo (+10.0 vs. +2.8 for a mean treatment difference of 7.2 overall, and +14.1 vs. +4.2 for a mean treatment difference of 9.9 in those with epithelioid histology).
“Overall frequency of adverse events was consistent with the known safety profile of nintedanib,” Dr. Barrueco said, noting that most adverse events were reversible with dose reduction.
Study participants were chemotherapy-naive patients with a mean age of 67 years and Eastern Cooperative Oncology Group performance status of 0-1. They received pemetrexed at a dose of 500 mg/m2 and cisplatin at a dose of 75 mg/m2 on day 1 plus either nintedanib at a dose of 200 mg twice daily on days 2-21 or placebo, followed by monotherapy with nintedanib or placebo until progression or unacceptable toxicity.
“In conclusion, nintedanib plus pemetrexed/cisplatin demonstrated a signal for clinical benefit in the first-time treatment of patients with malignant pleural mesothelioma. This was evident in all endpoints of the trial, and consistently showed benefit for the nintedanib group,” Mr. Barrueco said, noting that phase 3 of the LUME-Meso study is now recruiting.
CHICAGO – Adding the oral kinase inhibitor nintedanib to pemetrexed/cisplatin resulted in substantial improvements in outcomes in patients with unresectable malignant pleural mesothelioma in phase 2 of the randomized, placebo-controlled phase 2/3 LUME-Meso study.
The effects were particularly pronounced among those with epithelioid histology, Jose Barrueco, PhD, of Boehringer Ingelheim Pharmaceuticals, Ridgefield, Conn., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Progression-free survival – the primary endpoint of the study – was improved in 44 patients randomized to receive up to six cycles of pemetrexed/cisplatin plus nintedanib, compared with 43 patients who received pemetrexed/cisplatin plus placebo (median 9.4 vs. 5.7 months; hazard ratio, 0.54), Dr. Barrueco said.
In the 89% of patients with epithelioid malignant pleural mesothelioma, progression-free survival was a median of 9.7 vs. 5.7 months with nintedanib vs. placebo (HR, 0.49).
There was a trend toward improved overall survival in the nintedanib group vs. the placebo group, (median 18.3 vs. 14.2 months; HR, 0.77; P = .319), and overall survival was slightly better in those with epithelioid histology (median 20.6 vs. 15.2 months ; HR, 0.70; P = .197).
Consistent with these results, the adjusted mean change in forced vital capacity at cycle eight also favored nintedanib over placebo (+10.0 vs. +2.8 for a mean treatment difference of 7.2 overall, and +14.1 vs. +4.2 for a mean treatment difference of 9.9 in those with epithelioid histology).
“Overall frequency of adverse events was consistent with the known safety profile of nintedanib,” Dr. Barrueco said, noting that most adverse events were reversible with dose reduction.
Study participants were chemotherapy-naive patients with a mean age of 67 years and Eastern Cooperative Oncology Group performance status of 0-1. They received pemetrexed at a dose of 500 mg/m2 and cisplatin at a dose of 75 mg/m2 on day 1 plus either nintedanib at a dose of 200 mg twice daily on days 2-21 or placebo, followed by monotherapy with nintedanib or placebo until progression or unacceptable toxicity.
“In conclusion, nintedanib plus pemetrexed/cisplatin demonstrated a signal for clinical benefit in the first-time treatment of patients with malignant pleural mesothelioma. This was evident in all endpoints of the trial, and consistently showed benefit for the nintedanib group,” Mr. Barrueco said, noting that phase 3 of the LUME-Meso study is now recruiting.
AT A SYMPOSIUM IN THORACIC ONCOLOGY
Key clinical point:
Major finding: Overall median PFS with nintedanib vs. placebo was 9.4 vs. 5.7 months (HR, 0.54).
Data source: Phase 2 of the LUME-Meso trial with 87 patients.
Disclosures: Dr. Barrueco is an employee of Boehringer Ingelheim.
Retrospective review: No difference in PFS, OS with radiation before PD-1/PD-L1 inhibition
CHICAGO – Exposure to radiation therapy prior to PD-1/PD-L1 therapy was not associated with improved outcomes in a retrospective review of 66 lung cancer patients.
The patients had stage IIIB or IV non–small cell lung cancer, median age of 64 years, received at least 6 weeks of single-agent anti-PD-1/PD-L1 therapy in the second-line setting or beyond, and had survived at least 8 weeks from immunotherapy initiation. Compared with 13 patients who received no radiation therapy, the 53 who received any prior radiation therapy – including 44 with extracranial radiation and 22 with intracranial radiation – did not differ significantly with respect to progression-free survival (median 4-5 months; hazard ratio, 0.83), or overall survival (median of about 12 months in both groups; HR, 0.96), Christopher Strouse, MD, of the University of Iowa, Iowa City, reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
There also were no significant differences in the outcomes between those who had extracranial radiation and those who had intracranial radiation (HRs for PFS and OS, respectively, 0.91 and 1.19), or (on univariate analysis), between those receiving any vs. no intracranial radiation therapy (HRs for PFS and OS, respectively, 0.92 and 0.98), Dr. Strouse said.
The patients who received extracranial radiation therapy had lower lymphocyte counts at the time of anti-PD-1/PD-L1 therapy initiation vs. those who received only radiation therapy (mean lymphocyte count, 809 vs. 1,519), and those who received intracranial radiation therapy were younger than those who did not (median age, 59 vs. 65 years), but the groups were similar with respect to other variables, including gender, histology, performance status, smoking history, KRAS mutation, and number of prior lines of systemic therapies. Anti-PD-1/PD-L1 therapies are promising treatment options for metastatic non–small cell lung cancer, and combining these agents with other immune-modulating therapies may enhance their efficacy and lead to a greater proportion of patients with responses to these treatments, Dr. Strouse noted.
“It’s known that immune response depends on a lot of steps, even beyond the PD-1/PD-L1 axis, and one possible explanation for some of these patients [not responding] may be that there is some failure along the way in some other step,” he said. “Our hypothesis was that radiation therapy would be helpful in overcoming some of these barriers.”
However, in this study, which is limited by small sample size and single-institution retrospective design, no such effect was identified.
The findings conflict with some larger studies, including the recently-reported PACIFIC study, which showed a significant PFS benefit in lung cancer patients who received chemoradiation therapy followed by treatment with the PD-L1 inhibitor durvalumab.
Dr. Strouse said he looks forward to seeing further reports looking into the effects of radiation therapy at different doses and timing.
Invited discussant Heather Wakelee, MD, of Stanford (Calif.) University, also stressed the limitations of the University of Iowa study, and noted that while there are many unanswered questions, findings such as those from the PACIFIC trial show that radiation and PD-L1 inhibition is here to stay.
“It appears safe; there will be more coming,” she said.
Dr. Strouse reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
CHICAGO – Exposure to radiation therapy prior to PD-1/PD-L1 therapy was not associated with improved outcomes in a retrospective review of 66 lung cancer patients.
The patients had stage IIIB or IV non–small cell lung cancer, median age of 64 years, received at least 6 weeks of single-agent anti-PD-1/PD-L1 therapy in the second-line setting or beyond, and had survived at least 8 weeks from immunotherapy initiation. Compared with 13 patients who received no radiation therapy, the 53 who received any prior radiation therapy – including 44 with extracranial radiation and 22 with intracranial radiation – did not differ significantly with respect to progression-free survival (median 4-5 months; hazard ratio, 0.83), or overall survival (median of about 12 months in both groups; HR, 0.96), Christopher Strouse, MD, of the University of Iowa, Iowa City, reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
There also were no significant differences in the outcomes between those who had extracranial radiation and those who had intracranial radiation (HRs for PFS and OS, respectively, 0.91 and 1.19), or (on univariate analysis), between those receiving any vs. no intracranial radiation therapy (HRs for PFS and OS, respectively, 0.92 and 0.98), Dr. Strouse said.
The patients who received extracranial radiation therapy had lower lymphocyte counts at the time of anti-PD-1/PD-L1 therapy initiation vs. those who received only radiation therapy (mean lymphocyte count, 809 vs. 1,519), and those who received intracranial radiation therapy were younger than those who did not (median age, 59 vs. 65 years), but the groups were similar with respect to other variables, including gender, histology, performance status, smoking history, KRAS mutation, and number of prior lines of systemic therapies. Anti-PD-1/PD-L1 therapies are promising treatment options for metastatic non–small cell lung cancer, and combining these agents with other immune-modulating therapies may enhance their efficacy and lead to a greater proportion of patients with responses to these treatments, Dr. Strouse noted.
“It’s known that immune response depends on a lot of steps, even beyond the PD-1/PD-L1 axis, and one possible explanation for some of these patients [not responding] may be that there is some failure along the way in some other step,” he said. “Our hypothesis was that radiation therapy would be helpful in overcoming some of these barriers.”
However, in this study, which is limited by small sample size and single-institution retrospective design, no such effect was identified.
The findings conflict with some larger studies, including the recently-reported PACIFIC study, which showed a significant PFS benefit in lung cancer patients who received chemoradiation therapy followed by treatment with the PD-L1 inhibitor durvalumab.
Dr. Strouse said he looks forward to seeing further reports looking into the effects of radiation therapy at different doses and timing.
Invited discussant Heather Wakelee, MD, of Stanford (Calif.) University, also stressed the limitations of the University of Iowa study, and noted that while there are many unanswered questions, findings such as those from the PACIFIC trial show that radiation and PD-L1 inhibition is here to stay.
“It appears safe; there will be more coming,” she said.
Dr. Strouse reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
CHICAGO – Exposure to radiation therapy prior to PD-1/PD-L1 therapy was not associated with improved outcomes in a retrospective review of 66 lung cancer patients.
The patients had stage IIIB or IV non–small cell lung cancer, median age of 64 years, received at least 6 weeks of single-agent anti-PD-1/PD-L1 therapy in the second-line setting or beyond, and had survived at least 8 weeks from immunotherapy initiation. Compared with 13 patients who received no radiation therapy, the 53 who received any prior radiation therapy – including 44 with extracranial radiation and 22 with intracranial radiation – did not differ significantly with respect to progression-free survival (median 4-5 months; hazard ratio, 0.83), or overall survival (median of about 12 months in both groups; HR, 0.96), Christopher Strouse, MD, of the University of Iowa, Iowa City, reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
There also were no significant differences in the outcomes between those who had extracranial radiation and those who had intracranial radiation (HRs for PFS and OS, respectively, 0.91 and 1.19), or (on univariate analysis), between those receiving any vs. no intracranial radiation therapy (HRs for PFS and OS, respectively, 0.92 and 0.98), Dr. Strouse said.
The patients who received extracranial radiation therapy had lower lymphocyte counts at the time of anti-PD-1/PD-L1 therapy initiation vs. those who received only radiation therapy (mean lymphocyte count, 809 vs. 1,519), and those who received intracranial radiation therapy were younger than those who did not (median age, 59 vs. 65 years), but the groups were similar with respect to other variables, including gender, histology, performance status, smoking history, KRAS mutation, and number of prior lines of systemic therapies. Anti-PD-1/PD-L1 therapies are promising treatment options for metastatic non–small cell lung cancer, and combining these agents with other immune-modulating therapies may enhance their efficacy and lead to a greater proportion of patients with responses to these treatments, Dr. Strouse noted.
“It’s known that immune response depends on a lot of steps, even beyond the PD-1/PD-L1 axis, and one possible explanation for some of these patients [not responding] may be that there is some failure along the way in some other step,” he said. “Our hypothesis was that radiation therapy would be helpful in overcoming some of these barriers.”
However, in this study, which is limited by small sample size and single-institution retrospective design, no such effect was identified.
The findings conflict with some larger studies, including the recently-reported PACIFIC study, which showed a significant PFS benefit in lung cancer patients who received chemoradiation therapy followed by treatment with the PD-L1 inhibitor durvalumab.
Dr. Strouse said he looks forward to seeing further reports looking into the effects of radiation therapy at different doses and timing.
Invited discussant Heather Wakelee, MD, of Stanford (Calif.) University, also stressed the limitations of the University of Iowa study, and noted that while there are many unanswered questions, findings such as those from the PACIFIC trial show that radiation and PD-L1 inhibition is here to stay.
“It appears safe; there will be more coming,” she said.
Dr. Strouse reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
AT A SYMPOSIUM IN THORACIC ONCOLOGY
Key clinical point:
Major finding: PFS and OS did not differ significantly between patients who did and did not receive prior radiation therapy (HRs for PFS and OS, respectively, 0.83 and 0.96).
Data source: A retrospective review of 66 patients.
Disclosures: Dr. Strouse reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
Motesanib flops again in lung cancer
Motesanib has flunked another phase III trial in advanced nonsquamous non–small-cell lung cancer, this time in East Asian patients.
Compared with placebo, the investigational oral vascular endothelial growth factor (VEGF) inhibitor did not significantly improve progression-free survival (PFS) or the secondary endpoint, overall survival (OS), when added to paclitaxel and carboplatin (P/C), reported Kaoru Kubota, MD, of Nippon Medical School, Tokyo, and his associates. “The findings are consistent with overall findings of the phase III MONET1 study but do not replicate those of the subgroup analysis of Asian patients,” they wrote in Journal of Clinical Oncology.
Motesanib is a small-molecular inhibitor of VEGF receptors 1, 2, and 3. In a phase II trial of patients with advanced nonsquamous non–small-cell lung cancer, motesanib resembled the anti-VEGF-A monoclonal antibody bevacizumab in terms of objective response rate, median PFS, and OS when added to paclitaxel and carboplatin. In the subsequent phase III MONET1 trial, however, motesanib plus P/C did not improve PFS over placebo plus P/C, except in a preplanned subgroup analysis of 227 East Asian patients, where it was associated with a 6.4-month greater median PFS (P = .02) and a 1.7-month greater OS (P = .001).
Based on those findings, Dr. Kubota and his associates randomly assigned 401 patients with advanced nonsquamous non–small-cell lung cancer to receive oral motesanib (125 mg) or placebo once daily plus paclitaxel (200 mg/m2 IV) and carboplatin (area under the concentration-time curve, 6 mg/mL per min IV) for up to six 3-week cycles. Patients were from Hong Kong, Korea, Japan, and Taiwan; averaged 65 years of age; and 72% were male (J Clin Oncol. 2017 Sep 13. doi: 10.1200/JCO.2017.72.7297).
After a median follow-up of 10 months, median PFS was 6.1 months in the motesanib plus P/C arm and 5.6 months in the placebo plus P/C arm (hazard ratio, 0.81; P = .08). Respective objective response rates were 60% and 42% (P less than .001), median times to tumor response were 1.4 and 1.6 months, and median durations of response were 5.3 and 4.1 months. Motesanib was associated with a higher rate of serious adverse events (87% versus 68%) and a higher rate of treatment discontinuation due to adverse events (33% versus 14%). Motesanib most often caused gastrointestinal disorders, hypertension, cholecystitis, gallbladder enlargement, and liver disorders.
Takeda Pharmaceuticals makes motesanib and sponsored the trial. Dr. Kubota disclosed honoraria and research funding from numerous pharmaceutical companies excluding Takeda. Four coinvestigators disclosed research funding from Takeda and two coinvestigators reported employment with the company. The remaining five researchers had no conflicts.
Motesanib has flunked another phase III trial in advanced nonsquamous non–small-cell lung cancer, this time in East Asian patients.
Compared with placebo, the investigational oral vascular endothelial growth factor (VEGF) inhibitor did not significantly improve progression-free survival (PFS) or the secondary endpoint, overall survival (OS), when added to paclitaxel and carboplatin (P/C), reported Kaoru Kubota, MD, of Nippon Medical School, Tokyo, and his associates. “The findings are consistent with overall findings of the phase III MONET1 study but do not replicate those of the subgroup analysis of Asian patients,” they wrote in Journal of Clinical Oncology.
Motesanib is a small-molecular inhibitor of VEGF receptors 1, 2, and 3. In a phase II trial of patients with advanced nonsquamous non–small-cell lung cancer, motesanib resembled the anti-VEGF-A monoclonal antibody bevacizumab in terms of objective response rate, median PFS, and OS when added to paclitaxel and carboplatin. In the subsequent phase III MONET1 trial, however, motesanib plus P/C did not improve PFS over placebo plus P/C, except in a preplanned subgroup analysis of 227 East Asian patients, where it was associated with a 6.4-month greater median PFS (P = .02) and a 1.7-month greater OS (P = .001).
Based on those findings, Dr. Kubota and his associates randomly assigned 401 patients with advanced nonsquamous non–small-cell lung cancer to receive oral motesanib (125 mg) or placebo once daily plus paclitaxel (200 mg/m2 IV) and carboplatin (area under the concentration-time curve, 6 mg/mL per min IV) for up to six 3-week cycles. Patients were from Hong Kong, Korea, Japan, and Taiwan; averaged 65 years of age; and 72% were male (J Clin Oncol. 2017 Sep 13. doi: 10.1200/JCO.2017.72.7297).
After a median follow-up of 10 months, median PFS was 6.1 months in the motesanib plus P/C arm and 5.6 months in the placebo plus P/C arm (hazard ratio, 0.81; P = .08). Respective objective response rates were 60% and 42% (P less than .001), median times to tumor response were 1.4 and 1.6 months, and median durations of response were 5.3 and 4.1 months. Motesanib was associated with a higher rate of serious adverse events (87% versus 68%) and a higher rate of treatment discontinuation due to adverse events (33% versus 14%). Motesanib most often caused gastrointestinal disorders, hypertension, cholecystitis, gallbladder enlargement, and liver disorders.
Takeda Pharmaceuticals makes motesanib and sponsored the trial. Dr. Kubota disclosed honoraria and research funding from numerous pharmaceutical companies excluding Takeda. Four coinvestigators disclosed research funding from Takeda and two coinvestigators reported employment with the company. The remaining five researchers had no conflicts.
Motesanib has flunked another phase III trial in advanced nonsquamous non–small-cell lung cancer, this time in East Asian patients.
Compared with placebo, the investigational oral vascular endothelial growth factor (VEGF) inhibitor did not significantly improve progression-free survival (PFS) or the secondary endpoint, overall survival (OS), when added to paclitaxel and carboplatin (P/C), reported Kaoru Kubota, MD, of Nippon Medical School, Tokyo, and his associates. “The findings are consistent with overall findings of the phase III MONET1 study but do not replicate those of the subgroup analysis of Asian patients,” they wrote in Journal of Clinical Oncology.
Motesanib is a small-molecular inhibitor of VEGF receptors 1, 2, and 3. In a phase II trial of patients with advanced nonsquamous non–small-cell lung cancer, motesanib resembled the anti-VEGF-A monoclonal antibody bevacizumab in terms of objective response rate, median PFS, and OS when added to paclitaxel and carboplatin. In the subsequent phase III MONET1 trial, however, motesanib plus P/C did not improve PFS over placebo plus P/C, except in a preplanned subgroup analysis of 227 East Asian patients, where it was associated with a 6.4-month greater median PFS (P = .02) and a 1.7-month greater OS (P = .001).
Based on those findings, Dr. Kubota and his associates randomly assigned 401 patients with advanced nonsquamous non–small-cell lung cancer to receive oral motesanib (125 mg) or placebo once daily plus paclitaxel (200 mg/m2 IV) and carboplatin (area under the concentration-time curve, 6 mg/mL per min IV) for up to six 3-week cycles. Patients were from Hong Kong, Korea, Japan, and Taiwan; averaged 65 years of age; and 72% were male (J Clin Oncol. 2017 Sep 13. doi: 10.1200/JCO.2017.72.7297).
After a median follow-up of 10 months, median PFS was 6.1 months in the motesanib plus P/C arm and 5.6 months in the placebo plus P/C arm (hazard ratio, 0.81; P = .08). Respective objective response rates were 60% and 42% (P less than .001), median times to tumor response were 1.4 and 1.6 months, and median durations of response were 5.3 and 4.1 months. Motesanib was associated with a higher rate of serious adverse events (87% versus 68%) and a higher rate of treatment discontinuation due to adverse events (33% versus 14%). Motesanib most often caused gastrointestinal disorders, hypertension, cholecystitis, gallbladder enlargement, and liver disorders.
Takeda Pharmaceuticals makes motesanib and sponsored the trial. Dr. Kubota disclosed honoraria and research funding from numerous pharmaceutical companies excluding Takeda. Four coinvestigators disclosed research funding from Takeda and two coinvestigators reported employment with the company. The remaining five researchers had no conflicts.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Belying a previous subgroup analysis, motesanib, an investigational oral vascular endothelial growth factor inhibitor, did not significantly improve progression-free survival when added to paclitaxel/carboplatin in East Asian patients with advanced nonsquamous non–small-cell lung cancer.
Major finding: After a median follow-up of 10 months, median PFS was 6.1 months among motesanib recipients and 5.6 months in the placebo group (hazard ratio, 0.81; P = .08).
Data source: A double-blind, phase III trial of 401 patients.
Disclosures: Takeda Pharmaceuticals makes motesanib and sponsored the trial. Dr. Kubota disclosed honoraria and research funding from numerous pharmaceutical companies excluding Takeda. Four coinvestigators disclosed research funding from Takeda and two coinvestigators reported employment with the company. The remaining five researchers had no conflicts.
No difference in survival between x-ray or CT NSCLC follow-up
Madrid – Computed tomography scans do not appear to be superior to plain old chest x-rays for follow-up of patients with completely resected non–small cell lung cancer (NSCLC), results of a randomized clinical trial suggest.
Among 1,775 patients followed out to 10 years with either a “minimal” protocol – consisting of history, physical exam, and periodic chest x-rays – or a “maximal” protocol – including CT scans of the thorax and upper abdomen, as well as bronchoscopy for squamous-cell carcinomas – there were no significant differences in overall survival at either 3, 5, or 8 years of follow-up, reported Virginie Westeel, MD, from the Centre Hospitalier Régional Universitaire of the Hôpital Jean Minjoz in Besançon, France.
In hopes of finding that answer, Dr. Westeel and colleagues in the French Cooperative Thoracic Oncology Group conducted a clinical trial comparing the standard follow-up approach recommended in most clinical guidelines, as described by Dr. Westeel, with an experimental protocol consisting of history and exam plus chest x-ray, CT scans, and fiber-optic bronchoscopy (mandatory for squamous- and large-cell carcinomas, optional for adenocarcinomas).
Patients with completely resected stage I, II, and IIIA tumors, and T4 tumors with pulmonary nodules in the same lobe, were randomly assigned to follow-up with one of the two protocols.
In each trial arm, the assigned procedures were repeated every 6 months after randomization for the first 2 years, then yearly until 5 years.
After a median follow-up of 8.7 years, there was no significant difference in the primary endpoint of overall survival. Median OS was 123.6 months in the maximal protocol group, compared with 99.7 months in the minimal protocol group (P = .037)
The 3-, 5-, and 8-year survival rates for the maximal and minimal protocols, respectively, were 76.1% vs. 77.3%, 65.8% vs. 66.7%, and 54.6% vs. 51.7%.
Because there appeared to be a separation of the survival curve beginning around 8 years, the investigators performed an exploratory 2-year landmark analysis.
They found that, among patients who had a recurrence within 24 months of randomization, there was no difference in OS between each follow-up protocol. However, among those patients with no recurrence within 24 months of resection, the median OS was not reached among patients assigned to the maximal protocol versus 129.3 months for those assigned to the minimal protocol (P = .04).
Patients without early recurrence had higher rates of secondary primary cancers, and for these patients, early detection with CT-based surveillance could explain the differences in overall survival, Dr. Westeel said.
“Our suggestion for practice is that, because there is no survival difference, both follow-up protocols are acceptable. However, a CT scan every 6 months is probably of no value in the first 2 years,” but yearly chest CTs to detect second primary cancers early may be of interest, she said.
Enriqueta Felip, MD, from Vall D’Hebron Institute of Oncology in Barcelona, who was not involved in the trial, commented that, while the study needed to be conducted, it was unlikely to change her clinical practice because of potential differences among patients with varying stages of NSCLC at the time of resection.
“I think it’s an important trial, [but] tomorrow I will follow my patients with a CT scan,” she said.
Dr. Felip was an invited expert at the briefing.
The study was supported by the French Ministry of Health, Fondation de France, and Laboratoire Lilly. Dr. Westeel and Dr. Felip reported no conflicts of interest relevant to the study.
Madrid – Computed tomography scans do not appear to be superior to plain old chest x-rays for follow-up of patients with completely resected non–small cell lung cancer (NSCLC), results of a randomized clinical trial suggest.
Among 1,775 patients followed out to 10 years with either a “minimal” protocol – consisting of history, physical exam, and periodic chest x-rays – or a “maximal” protocol – including CT scans of the thorax and upper abdomen, as well as bronchoscopy for squamous-cell carcinomas – there were no significant differences in overall survival at either 3, 5, or 8 years of follow-up, reported Virginie Westeel, MD, from the Centre Hospitalier Régional Universitaire of the Hôpital Jean Minjoz in Besançon, France.
In hopes of finding that answer, Dr. Westeel and colleagues in the French Cooperative Thoracic Oncology Group conducted a clinical trial comparing the standard follow-up approach recommended in most clinical guidelines, as described by Dr. Westeel, with an experimental protocol consisting of history and exam plus chest x-ray, CT scans, and fiber-optic bronchoscopy (mandatory for squamous- and large-cell carcinomas, optional for adenocarcinomas).
Patients with completely resected stage I, II, and IIIA tumors, and T4 tumors with pulmonary nodules in the same lobe, were randomly assigned to follow-up with one of the two protocols.
In each trial arm, the assigned procedures were repeated every 6 months after randomization for the first 2 years, then yearly until 5 years.
After a median follow-up of 8.7 years, there was no significant difference in the primary endpoint of overall survival. Median OS was 123.6 months in the maximal protocol group, compared with 99.7 months in the minimal protocol group (P = .037)
The 3-, 5-, and 8-year survival rates for the maximal and minimal protocols, respectively, were 76.1% vs. 77.3%, 65.8% vs. 66.7%, and 54.6% vs. 51.7%.
Because there appeared to be a separation of the survival curve beginning around 8 years, the investigators performed an exploratory 2-year landmark analysis.
They found that, among patients who had a recurrence within 24 months of randomization, there was no difference in OS between each follow-up protocol. However, among those patients with no recurrence within 24 months of resection, the median OS was not reached among patients assigned to the maximal protocol versus 129.3 months for those assigned to the minimal protocol (P = .04).
Patients without early recurrence had higher rates of secondary primary cancers, and for these patients, early detection with CT-based surveillance could explain the differences in overall survival, Dr. Westeel said.
“Our suggestion for practice is that, because there is no survival difference, both follow-up protocols are acceptable. However, a CT scan every 6 months is probably of no value in the first 2 years,” but yearly chest CTs to detect second primary cancers early may be of interest, she said.
Enriqueta Felip, MD, from Vall D’Hebron Institute of Oncology in Barcelona, who was not involved in the trial, commented that, while the study needed to be conducted, it was unlikely to change her clinical practice because of potential differences among patients with varying stages of NSCLC at the time of resection.
“I think it’s an important trial, [but] tomorrow I will follow my patients with a CT scan,” she said.
Dr. Felip was an invited expert at the briefing.
The study was supported by the French Ministry of Health, Fondation de France, and Laboratoire Lilly. Dr. Westeel and Dr. Felip reported no conflicts of interest relevant to the study.
Madrid – Computed tomography scans do not appear to be superior to plain old chest x-rays for follow-up of patients with completely resected non–small cell lung cancer (NSCLC), results of a randomized clinical trial suggest.
Among 1,775 patients followed out to 10 years with either a “minimal” protocol – consisting of history, physical exam, and periodic chest x-rays – or a “maximal” protocol – including CT scans of the thorax and upper abdomen, as well as bronchoscopy for squamous-cell carcinomas – there were no significant differences in overall survival at either 3, 5, or 8 years of follow-up, reported Virginie Westeel, MD, from the Centre Hospitalier Régional Universitaire of the Hôpital Jean Minjoz in Besançon, France.
In hopes of finding that answer, Dr. Westeel and colleagues in the French Cooperative Thoracic Oncology Group conducted a clinical trial comparing the standard follow-up approach recommended in most clinical guidelines, as described by Dr. Westeel, with an experimental protocol consisting of history and exam plus chest x-ray, CT scans, and fiber-optic bronchoscopy (mandatory for squamous- and large-cell carcinomas, optional for adenocarcinomas).
Patients with completely resected stage I, II, and IIIA tumors, and T4 tumors with pulmonary nodules in the same lobe, were randomly assigned to follow-up with one of the two protocols.
In each trial arm, the assigned procedures were repeated every 6 months after randomization for the first 2 years, then yearly until 5 years.
After a median follow-up of 8.7 years, there was no significant difference in the primary endpoint of overall survival. Median OS was 123.6 months in the maximal protocol group, compared with 99.7 months in the minimal protocol group (P = .037)
The 3-, 5-, and 8-year survival rates for the maximal and minimal protocols, respectively, were 76.1% vs. 77.3%, 65.8% vs. 66.7%, and 54.6% vs. 51.7%.
Because there appeared to be a separation of the survival curve beginning around 8 years, the investigators performed an exploratory 2-year landmark analysis.
They found that, among patients who had a recurrence within 24 months of randomization, there was no difference in OS between each follow-up protocol. However, among those patients with no recurrence within 24 months of resection, the median OS was not reached among patients assigned to the maximal protocol versus 129.3 months for those assigned to the minimal protocol (P = .04).
Patients without early recurrence had higher rates of secondary primary cancers, and for these patients, early detection with CT-based surveillance could explain the differences in overall survival, Dr. Westeel said.
“Our suggestion for practice is that, because there is no survival difference, both follow-up protocols are acceptable. However, a CT scan every 6 months is probably of no value in the first 2 years,” but yearly chest CTs to detect second primary cancers early may be of interest, she said.
Enriqueta Felip, MD, from Vall D’Hebron Institute of Oncology in Barcelona, who was not involved in the trial, commented that, while the study needed to be conducted, it was unlikely to change her clinical practice because of potential differences among patients with varying stages of NSCLC at the time of resection.
“I think it’s an important trial, [but] tomorrow I will follow my patients with a CT scan,” she said.
Dr. Felip was an invited expert at the briefing.
The study was supported by the French Ministry of Health, Fondation de France, and Laboratoire Lilly. Dr. Westeel and Dr. Felip reported no conflicts of interest relevant to the study.
AT ESMO 2017
Key clinical point: There were no differences in long-term overall survival among patients with resected NSCLC followed with x-rays and those followed with CT scans.
Major finding: Overall survival rates at 3, 5, and 8 years were similar among patients followed with minimal or maximal protocols.
Data source: Randomized controlled trial in 1,775 patients with completely resected non–small cell lung cancer.
Disclosures: The study was supported by the French Ministry of Health, Fondation de France, and Laboratoire Lilly. Dr. Westeel and Dr. Felip reported no conflicts of interest relevant to the study.
CRP may predict survival after immunotherapy for lung cancer
CHICAGO – A baseline C-reactive protein (CRP) level above 50 mg/L independently predicted worse overall survival after immunotherapy in patients with advanced non–small cell lung cancer and small cell lung cancer in a retrospective study.
In 99 patients treated with nivolumab after a first-line platinum doublet, the median baseline CRP level was 22 mg/L. After a median follow-up of 8.5 months, 50% of patients were alive, and, based on univariate and multivariate analysis, both liver involvement and having a CRP level greater than 50 mg/L were significantly associated with inferior overall survival after immunotherapy.
The median overall survival after immunotherapy was 9.3 months versus 2.7 months with a CRP level of 50 mg/L or less versus above 50 mg/L, Abdul Rafeh Naqash, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Notably, significant increases in CRP level, compared with baseline, were seen at the time of grade 2 to grade 4 immune-related adverse events, which occurred in 38.4% of patients. This is a hypothesis-generating finding in that it suggests there is dysregulation of the immune system, in the context of immune checkpoint blockade, that leads to a more proinflammatory state, which ultimately leads to immune-related adverse events, Dr. Naqash said.
Study subjects were adults with a median age of 65 years who were treated during April 2015-March 2017. Most were white (64.7%), were male (64.6%), and had non–small cell lung cancer (88%). Most had stage IV disease (70.7%), and the most common site for metastases was the bones (35.4%) and the liver (24.2%). Patients’ CRP levels were measured at anti-PD-1–treatment initiation and serially with subsequent doses.
The findings are important because the identification of predictive biomarkers in patients treated with anti-PD-1 therapy could provide valuable insights into underlying mechanisms regulating patient responses, elucidate resistance mechanisms, and help with optimal selection of patients for treatment with and development of patient-tailored treatment, Dr. Naqash said, noting that identifying such biomarkers has thus far been a challenge.
However, this study is limited by its retrospective design and limited follow-up; the findings require validation in prospective lung cancer trials, he concluded.
Dr. Naqash reported having no disclosures.
CHICAGO – A baseline C-reactive protein (CRP) level above 50 mg/L independently predicted worse overall survival after immunotherapy in patients with advanced non–small cell lung cancer and small cell lung cancer in a retrospective study.
In 99 patients treated with nivolumab after a first-line platinum doublet, the median baseline CRP level was 22 mg/L. After a median follow-up of 8.5 months, 50% of patients were alive, and, based on univariate and multivariate analysis, both liver involvement and having a CRP level greater than 50 mg/L were significantly associated with inferior overall survival after immunotherapy.
The median overall survival after immunotherapy was 9.3 months versus 2.7 months with a CRP level of 50 mg/L or less versus above 50 mg/L, Abdul Rafeh Naqash, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Notably, significant increases in CRP level, compared with baseline, were seen at the time of grade 2 to grade 4 immune-related adverse events, which occurred in 38.4% of patients. This is a hypothesis-generating finding in that it suggests there is dysregulation of the immune system, in the context of immune checkpoint blockade, that leads to a more proinflammatory state, which ultimately leads to immune-related adverse events, Dr. Naqash said.
Study subjects were adults with a median age of 65 years who were treated during April 2015-March 2017. Most were white (64.7%), were male (64.6%), and had non–small cell lung cancer (88%). Most had stage IV disease (70.7%), and the most common site for metastases was the bones (35.4%) and the liver (24.2%). Patients’ CRP levels were measured at anti-PD-1–treatment initiation and serially with subsequent doses.
The findings are important because the identification of predictive biomarkers in patients treated with anti-PD-1 therapy could provide valuable insights into underlying mechanisms regulating patient responses, elucidate resistance mechanisms, and help with optimal selection of patients for treatment with and development of patient-tailored treatment, Dr. Naqash said, noting that identifying such biomarkers has thus far been a challenge.
However, this study is limited by its retrospective design and limited follow-up; the findings require validation in prospective lung cancer trials, he concluded.
Dr. Naqash reported having no disclosures.
CHICAGO – A baseline C-reactive protein (CRP) level above 50 mg/L independently predicted worse overall survival after immunotherapy in patients with advanced non–small cell lung cancer and small cell lung cancer in a retrospective study.
In 99 patients treated with nivolumab after a first-line platinum doublet, the median baseline CRP level was 22 mg/L. After a median follow-up of 8.5 months, 50% of patients were alive, and, based on univariate and multivariate analysis, both liver involvement and having a CRP level greater than 50 mg/L were significantly associated with inferior overall survival after immunotherapy.
The median overall survival after immunotherapy was 9.3 months versus 2.7 months with a CRP level of 50 mg/L or less versus above 50 mg/L, Abdul Rafeh Naqash, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
Notably, significant increases in CRP level, compared with baseline, were seen at the time of grade 2 to grade 4 immune-related adverse events, which occurred in 38.4% of patients. This is a hypothesis-generating finding in that it suggests there is dysregulation of the immune system, in the context of immune checkpoint blockade, that leads to a more proinflammatory state, which ultimately leads to immune-related adverse events, Dr. Naqash said.
Study subjects were adults with a median age of 65 years who were treated during April 2015-March 2017. Most were white (64.7%), were male (64.6%), and had non–small cell lung cancer (88%). Most had stage IV disease (70.7%), and the most common site for metastases was the bones (35.4%) and the liver (24.2%). Patients’ CRP levels were measured at anti-PD-1–treatment initiation and serially with subsequent doses.
The findings are important because the identification of predictive biomarkers in patients treated with anti-PD-1 therapy could provide valuable insights into underlying mechanisms regulating patient responses, elucidate resistance mechanisms, and help with optimal selection of patients for treatment with and development of patient-tailored treatment, Dr. Naqash said, noting that identifying such biomarkers has thus far been a challenge.
However, this study is limited by its retrospective design and limited follow-up; the findings require validation in prospective lung cancer trials, he concluded.
Dr. Naqash reported having no disclosures.
AT A SYMPOSIUM IN THORACIC ONCOLOGY
Key clinical point:
Major finding: Median overall survival after immunotherapy: 9.3 months vs. 2.7 months with CRP of 50 mg/L or less vs. above 50 mg/L.
Data source: A retrospective study of 99 patients.
Disclosures: Dr. Naqash reported having no disclosures.
Tocilizumab looks promising for corticosteroid refractory anti-PD-1-related adverse events
CHICAGO – Tocilizumab may be a therapeutic option for steroid-refractory immune-related adverse events that are secondary to PD-1 blockade, according to findings from a review of patients treated with nivolumab for various malignancies.
Of 87 patients who were treated with the PD-1 inhibitor between April 2015 and October 2016, 34 received tocilizumab for high-grade immune-related adverse events (irAEs) that were refractory to corticosteroids. Of those, 27 experienced clinical improvement, which was defined as documentation of symptom resolution or hospital discharge within 7 days, Aparna Hegde, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
The median time to discharge was 4 days, and no adverse effect on median overall survival was seen between those who received tocilizumab and those who did not (6.1 vs, 6.7 months, respectively), Dr. Hegde said.
There was, however, a trend toward inferior overall survival in patients who required more than one dose of tocilizumab, but the difference was not statistically significant (hazard ratio, 1.72), she noted.
“Immune checkpoint inhibitors are associated with an unprecedented clinical benefit in patients with lung cancer. However, they are also associated with a unique spectrum of immune-mediated adverse events. While the standard of care for initial management of these adverse events is corticosteroids, the management of steroid-refractory events is poorly defined,” she said, adding that data from randomized trials on which consensus guidelines could be based are lacking.
At East Carolina University, a significant proportion of patients treated with PD-1 inhibitors were presenting with systemic inflammatory response syndrome (SIRS)-like symptoms and immune-related organ toxicities similar to what has been described in cytokine release syndrome, Dr. Hegde said.
Such symptoms have also been reported with other types of immune therapy, such as CAR T-cell therapy and bispecific T-cell receptor–engaging antibodies in hematologic malignancies, she noted.
“In our experience, when we treated these patients with tocilizumab, which is an [anti-interleukin-6] receptor monoclonal antibody, we saw dramatic and rapid responses, not only in the SIRS symptoms, but also in other immune-related organ toxicities. Therefore, we adopted the use of tocilizumab as our standard treatment for high-grade immune-related adverse events,” she said, explaining that it has been well documented that interleukin (IL)-6 levels increase during cytokine release syndrome and are mediators of inflammation, suggesting that blocking IL-6 may treat irAEs without compromising the efficacy of immune therapy.
The current study was undertaken to look more closely at the responses to tocilizumab and to assess overall survival in those who received tocilizumab.
Study participants were being prospectively followed as part of another ongoing study looking at the relationship between systemic inflammation and cancer-related symptom burden. Most (77) were being treated for lung cancer and 10 had other types of malignancy. They received nivolumab at a dose of 3 mg/kg (or a flat dose of 240 mg after September 2016) every 2 weeks, and received tocilizumab at a dose of 4 mg/kg given over 1 hour. Those with grade 3 or 4 irAEs also received supportive care and corticosteroids. Median follow-up was 10.6 months.
C-reactive protein (CRP), a reliable surrogate marker of IL-6, was drawn at the first nivolumab infusion and before each subsequent infusion as part of the study in which the patients were enrolled, and for the current analysis was measured in relation to irAEs.
Significant reductions were seen in CRP levels after tocilizumab treatment; similar responses have been described in cytokine release syndrome, Dr. Hegde noted.
“Tocilizumab is a therapeutic option for management of immune-related adverse events in patients who are already on corticosteroids. CRP may be of clinical utility in detecting immune-related adverse events as well as monitoring the response to tocilizumab,” she said, adding that the current analysis is limited by the small patient number, single-center setting, use of tocilizumab outside of a clinical trial setting, and short follow-up.
Therefore, the findings require confirmation in multicenter randomized trials to determine “the definitive utility of tocilizumab, as well as CRP as an accompanying biomarker in the management of high-grade steroid refractory immune-related adverse events.”
Heather Wakelee, MD, an invited discussant at the symposium, commended Dr. Hegde and her colleagues for “coming up with a novel idea about how to treat [irAEs],” but also stressed the need for further study.
“This is a novel agent that has the potential ability to manage toxicity, and that’s important, because when you get beyond the steroids that we use as a first-line approach ... there’s not a whole lot else. We definitely have a clear unmet need,” said Dr. Wakelee of Stanford (Calif.) University.
However, she stressed that the approach must be evaluated in multicenter randomized trials “before we can be widely discussing this as a good thing to be doing.”
Dr. Hegde reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
CHICAGO – Tocilizumab may be a therapeutic option for steroid-refractory immune-related adverse events that are secondary to PD-1 blockade, according to findings from a review of patients treated with nivolumab for various malignancies.
Of 87 patients who were treated with the PD-1 inhibitor between April 2015 and October 2016, 34 received tocilizumab for high-grade immune-related adverse events (irAEs) that were refractory to corticosteroids. Of those, 27 experienced clinical improvement, which was defined as documentation of symptom resolution or hospital discharge within 7 days, Aparna Hegde, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
The median time to discharge was 4 days, and no adverse effect on median overall survival was seen between those who received tocilizumab and those who did not (6.1 vs, 6.7 months, respectively), Dr. Hegde said.
There was, however, a trend toward inferior overall survival in patients who required more than one dose of tocilizumab, but the difference was not statistically significant (hazard ratio, 1.72), she noted.
“Immune checkpoint inhibitors are associated with an unprecedented clinical benefit in patients with lung cancer. However, they are also associated with a unique spectrum of immune-mediated adverse events. While the standard of care for initial management of these adverse events is corticosteroids, the management of steroid-refractory events is poorly defined,” she said, adding that data from randomized trials on which consensus guidelines could be based are lacking.
At East Carolina University, a significant proportion of patients treated with PD-1 inhibitors were presenting with systemic inflammatory response syndrome (SIRS)-like symptoms and immune-related organ toxicities similar to what has been described in cytokine release syndrome, Dr. Hegde said.
Such symptoms have also been reported with other types of immune therapy, such as CAR T-cell therapy and bispecific T-cell receptor–engaging antibodies in hematologic malignancies, she noted.
“In our experience, when we treated these patients with tocilizumab, which is an [anti-interleukin-6] receptor monoclonal antibody, we saw dramatic and rapid responses, not only in the SIRS symptoms, but also in other immune-related organ toxicities. Therefore, we adopted the use of tocilizumab as our standard treatment for high-grade immune-related adverse events,” she said, explaining that it has been well documented that interleukin (IL)-6 levels increase during cytokine release syndrome and are mediators of inflammation, suggesting that blocking IL-6 may treat irAEs without compromising the efficacy of immune therapy.
The current study was undertaken to look more closely at the responses to tocilizumab and to assess overall survival in those who received tocilizumab.
Study participants were being prospectively followed as part of another ongoing study looking at the relationship between systemic inflammation and cancer-related symptom burden. Most (77) were being treated for lung cancer and 10 had other types of malignancy. They received nivolumab at a dose of 3 mg/kg (or a flat dose of 240 mg after September 2016) every 2 weeks, and received tocilizumab at a dose of 4 mg/kg given over 1 hour. Those with grade 3 or 4 irAEs also received supportive care and corticosteroids. Median follow-up was 10.6 months.
C-reactive protein (CRP), a reliable surrogate marker of IL-6, was drawn at the first nivolumab infusion and before each subsequent infusion as part of the study in which the patients were enrolled, and for the current analysis was measured in relation to irAEs.
Significant reductions were seen in CRP levels after tocilizumab treatment; similar responses have been described in cytokine release syndrome, Dr. Hegde noted.
“Tocilizumab is a therapeutic option for management of immune-related adverse events in patients who are already on corticosteroids. CRP may be of clinical utility in detecting immune-related adverse events as well as monitoring the response to tocilizumab,” she said, adding that the current analysis is limited by the small patient number, single-center setting, use of tocilizumab outside of a clinical trial setting, and short follow-up.
Therefore, the findings require confirmation in multicenter randomized trials to determine “the definitive utility of tocilizumab, as well as CRP as an accompanying biomarker in the management of high-grade steroid refractory immune-related adverse events.”
Heather Wakelee, MD, an invited discussant at the symposium, commended Dr. Hegde and her colleagues for “coming up with a novel idea about how to treat [irAEs],” but also stressed the need for further study.
“This is a novel agent that has the potential ability to manage toxicity, and that’s important, because when you get beyond the steroids that we use as a first-line approach ... there’s not a whole lot else. We definitely have a clear unmet need,” said Dr. Wakelee of Stanford (Calif.) University.
However, she stressed that the approach must be evaluated in multicenter randomized trials “before we can be widely discussing this as a good thing to be doing.”
Dr. Hegde reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
CHICAGO – Tocilizumab may be a therapeutic option for steroid-refractory immune-related adverse events that are secondary to PD-1 blockade, according to findings from a review of patients treated with nivolumab for various malignancies.
Of 87 patients who were treated with the PD-1 inhibitor between April 2015 and October 2016, 34 received tocilizumab for high-grade immune-related adverse events (irAEs) that were refractory to corticosteroids. Of those, 27 experienced clinical improvement, which was defined as documentation of symptom resolution or hospital discharge within 7 days, Aparna Hegde, MD, of East Carolina University, Greenville, N.C., reported at the Chicago Multidisciplinary Symposium in Thoracic Oncology.
The median time to discharge was 4 days, and no adverse effect on median overall survival was seen between those who received tocilizumab and those who did not (6.1 vs, 6.7 months, respectively), Dr. Hegde said.
There was, however, a trend toward inferior overall survival in patients who required more than one dose of tocilizumab, but the difference was not statistically significant (hazard ratio, 1.72), she noted.
“Immune checkpoint inhibitors are associated with an unprecedented clinical benefit in patients with lung cancer. However, they are also associated with a unique spectrum of immune-mediated adverse events. While the standard of care for initial management of these adverse events is corticosteroids, the management of steroid-refractory events is poorly defined,” she said, adding that data from randomized trials on which consensus guidelines could be based are lacking.
At East Carolina University, a significant proportion of patients treated with PD-1 inhibitors were presenting with systemic inflammatory response syndrome (SIRS)-like symptoms and immune-related organ toxicities similar to what has been described in cytokine release syndrome, Dr. Hegde said.
Such symptoms have also been reported with other types of immune therapy, such as CAR T-cell therapy and bispecific T-cell receptor–engaging antibodies in hematologic malignancies, she noted.
“In our experience, when we treated these patients with tocilizumab, which is an [anti-interleukin-6] receptor monoclonal antibody, we saw dramatic and rapid responses, not only in the SIRS symptoms, but also in other immune-related organ toxicities. Therefore, we adopted the use of tocilizumab as our standard treatment for high-grade immune-related adverse events,” she said, explaining that it has been well documented that interleukin (IL)-6 levels increase during cytokine release syndrome and are mediators of inflammation, suggesting that blocking IL-6 may treat irAEs without compromising the efficacy of immune therapy.
The current study was undertaken to look more closely at the responses to tocilizumab and to assess overall survival in those who received tocilizumab.
Study participants were being prospectively followed as part of another ongoing study looking at the relationship between systemic inflammation and cancer-related symptom burden. Most (77) were being treated for lung cancer and 10 had other types of malignancy. They received nivolumab at a dose of 3 mg/kg (or a flat dose of 240 mg after September 2016) every 2 weeks, and received tocilizumab at a dose of 4 mg/kg given over 1 hour. Those with grade 3 or 4 irAEs also received supportive care and corticosteroids. Median follow-up was 10.6 months.
C-reactive protein (CRP), a reliable surrogate marker of IL-6, was drawn at the first nivolumab infusion and before each subsequent infusion as part of the study in which the patients were enrolled, and for the current analysis was measured in relation to irAEs.
Significant reductions were seen in CRP levels after tocilizumab treatment; similar responses have been described in cytokine release syndrome, Dr. Hegde noted.
“Tocilizumab is a therapeutic option for management of immune-related adverse events in patients who are already on corticosteroids. CRP may be of clinical utility in detecting immune-related adverse events as well as monitoring the response to tocilizumab,” she said, adding that the current analysis is limited by the small patient number, single-center setting, use of tocilizumab outside of a clinical trial setting, and short follow-up.
Therefore, the findings require confirmation in multicenter randomized trials to determine “the definitive utility of tocilizumab, as well as CRP as an accompanying biomarker in the management of high-grade steroid refractory immune-related adverse events.”
Heather Wakelee, MD, an invited discussant at the symposium, commended Dr. Hegde and her colleagues for “coming up with a novel idea about how to treat [irAEs],” but also stressed the need for further study.
“This is a novel agent that has the potential ability to manage toxicity, and that’s important, because when you get beyond the steroids that we use as a first-line approach ... there’s not a whole lot else. We definitely have a clear unmet need,” said Dr. Wakelee of Stanford (Calif.) University.
However, she stressed that the approach must be evaluated in multicenter randomized trials “before we can be widely discussing this as a good thing to be doing.”
Dr. Hegde reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
AT A SYMPOSIUM IN THORACIC ONCOLOGY
Key clinical point:
Major finding: Twenty-seven of 34 patients treated with tocilizumab experienced clinical improvement.
Data source: A review of 87 patients.
Disclosures: Dr. Hegde reported having no disclosures. Dr. Wakelee has been the institutional principal investigator for studies of nivolumab, tocilizumab, and other agents. She has consulted for Peregrine, ACEA, Pfizer, Helsinn, Genentech/Roche, Clovis, and Lilly, and received research/grant support from Clovis, Exelixis, AstraZeneca/Medimmune, Genentech/Roche, BMS, Gilead, Novartis, Xcovery, Pfizer, Celgene, Gilead, Pharmacyclics, and Lilly.
E-cigarettes most popular among youngest adults
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).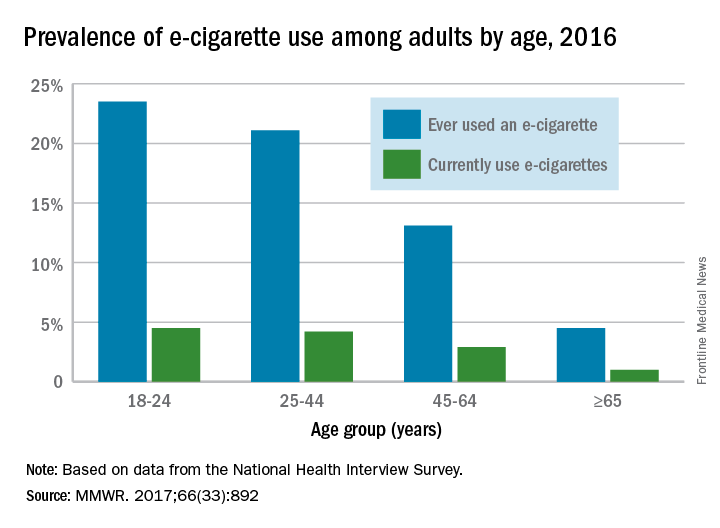
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).
FROM MMWR
FDA approves biosimilar to bevacizumab
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.

