User login
New understanding of suicide attempts emerges
even in the absence of a psychiatric disorder.
This finding suggests the genetic underpinnings of suicide attempts are partially shared and partially distinct from those of related psychiatric disorders, the investigators note.
“This study brings us a step closer to understanding the neurobiology of suicidality, with the ultimate goal of developing new treatments and prevention strategies,” Niamh Mullins, PhD, department of psychiatry, department of genetics and genomic sciences, Icahn School of Medicine at Mount Sinai in New York, said in an interview.
The study was published online in Biological Psychiatry.
Largest study to date
In the largest genetic association study of suicide attempt published to date, the researchers conducted a genome-wide association study (GWAS) of 29,782 suicide attempt cases and 519,961 controls in the International Suicide Genetics Consortium (ISGC).
Two loci reached genome-wide significance for suicide attempt – the major histocompatibility complex and an intergenic locus on chromosome 7, the latter of which remained associated with suicide attempt after conditioning on psychiatric disorders and was replicated in an independent cohort of over 14,000 veterans in the Million Veteran Program.
“This is the first replicated genetic locus that contributes more to suicide attempt than related psychiatric disorders,” Dr. Mullins said.
“The study found overlap in the genetic basis of suicide attempt and that of related psychiatric disorders, particularly major depression, but also with that of nonpsychiatric risk factors such as smoking, pain, risk-taking behavior, sleep disturbances, and poorer general health,” Dr. Mullins said.
“These genetic relationships between suicide attempt and nonpsychiatric risk factors were not a by-product of comorbid psychiatric illness, suggesting that there is some shared biological basis between suicide attempt and nonpsychiatric risk factors,” she added.
Dr. Mullins cautioned that the findings do not have any immediate impact on patient care.
“The ultimate goal of this research is to gain insight into the underlying biological pathways involved in suicide attempts or suicidal thoughts, providing potential avenues to treatments and prevention strategies,” she said.
“The study findings also point to the importance of studying the potential direct causal paths between these risk factors and suicide attempt in patients with and without psychiatric illness,” Douglas Ruderfer, PhD, of Vanderbilt University Medical Center, Nashville, Tenn., cofounder and cochair of the consortium and senior author of the paper, added in a news release.
A version of this article first appeared on Medscape.com.
even in the absence of a psychiatric disorder.
This finding suggests the genetic underpinnings of suicide attempts are partially shared and partially distinct from those of related psychiatric disorders, the investigators note.
“This study brings us a step closer to understanding the neurobiology of suicidality, with the ultimate goal of developing new treatments and prevention strategies,” Niamh Mullins, PhD, department of psychiatry, department of genetics and genomic sciences, Icahn School of Medicine at Mount Sinai in New York, said in an interview.
The study was published online in Biological Psychiatry.
Largest study to date
In the largest genetic association study of suicide attempt published to date, the researchers conducted a genome-wide association study (GWAS) of 29,782 suicide attempt cases and 519,961 controls in the International Suicide Genetics Consortium (ISGC).
Two loci reached genome-wide significance for suicide attempt – the major histocompatibility complex and an intergenic locus on chromosome 7, the latter of which remained associated with suicide attempt after conditioning on psychiatric disorders and was replicated in an independent cohort of over 14,000 veterans in the Million Veteran Program.
“This is the first replicated genetic locus that contributes more to suicide attempt than related psychiatric disorders,” Dr. Mullins said.
“The study found overlap in the genetic basis of suicide attempt and that of related psychiatric disorders, particularly major depression, but also with that of nonpsychiatric risk factors such as smoking, pain, risk-taking behavior, sleep disturbances, and poorer general health,” Dr. Mullins said.
“These genetic relationships between suicide attempt and nonpsychiatric risk factors were not a by-product of comorbid psychiatric illness, suggesting that there is some shared biological basis between suicide attempt and nonpsychiatric risk factors,” she added.
Dr. Mullins cautioned that the findings do not have any immediate impact on patient care.
“The ultimate goal of this research is to gain insight into the underlying biological pathways involved in suicide attempts or suicidal thoughts, providing potential avenues to treatments and prevention strategies,” she said.
“The study findings also point to the importance of studying the potential direct causal paths between these risk factors and suicide attempt in patients with and without psychiatric illness,” Douglas Ruderfer, PhD, of Vanderbilt University Medical Center, Nashville, Tenn., cofounder and cochair of the consortium and senior author of the paper, added in a news release.
A version of this article first appeared on Medscape.com.
even in the absence of a psychiatric disorder.
This finding suggests the genetic underpinnings of suicide attempts are partially shared and partially distinct from those of related psychiatric disorders, the investigators note.
“This study brings us a step closer to understanding the neurobiology of suicidality, with the ultimate goal of developing new treatments and prevention strategies,” Niamh Mullins, PhD, department of psychiatry, department of genetics and genomic sciences, Icahn School of Medicine at Mount Sinai in New York, said in an interview.
The study was published online in Biological Psychiatry.
Largest study to date
In the largest genetic association study of suicide attempt published to date, the researchers conducted a genome-wide association study (GWAS) of 29,782 suicide attempt cases and 519,961 controls in the International Suicide Genetics Consortium (ISGC).
Two loci reached genome-wide significance for suicide attempt – the major histocompatibility complex and an intergenic locus on chromosome 7, the latter of which remained associated with suicide attempt after conditioning on psychiatric disorders and was replicated in an independent cohort of over 14,000 veterans in the Million Veteran Program.
“This is the first replicated genetic locus that contributes more to suicide attempt than related psychiatric disorders,” Dr. Mullins said.
“The study found overlap in the genetic basis of suicide attempt and that of related psychiatric disorders, particularly major depression, but also with that of nonpsychiatric risk factors such as smoking, pain, risk-taking behavior, sleep disturbances, and poorer general health,” Dr. Mullins said.
“These genetic relationships between suicide attempt and nonpsychiatric risk factors were not a by-product of comorbid psychiatric illness, suggesting that there is some shared biological basis between suicide attempt and nonpsychiatric risk factors,” she added.
Dr. Mullins cautioned that the findings do not have any immediate impact on patient care.
“The ultimate goal of this research is to gain insight into the underlying biological pathways involved in suicide attempts or suicidal thoughts, providing potential avenues to treatments and prevention strategies,” she said.
“The study findings also point to the importance of studying the potential direct causal paths between these risk factors and suicide attempt in patients with and without psychiatric illness,” Douglas Ruderfer, PhD, of Vanderbilt University Medical Center, Nashville, Tenn., cofounder and cochair of the consortium and senior author of the paper, added in a news release.
A version of this article first appeared on Medscape.com.
FROM BIOLOGICAL PSYCHIATRY
Treating homeless patients: Book offers key insights
As a psychiatrist dedicated to working with people who are experiencing homelessness, I was very impressed with the new book edited by Col. (Ret.) Elspeth Cameron Ritchie, MD, MPH, and Maria D. Llorente, MD, about treating and providing services to this vulnerable population.
The book, “Clinical Management of the Homeless Patient: Social, Psychiatric, and Medical Issues” (Cham, Switzerland: Springer Nature Switzerland, 2021), offers an in-depth review and analysis of the biopsychosocial complexities that affect how medical and behavioral health conditions present in those who are unhoused. Notably, the book recommends with great sensitivity best practices to address these conditions with care, understanding, and love.
This text, invaluable in particular for those of us clinicians who work with people experiencing homelessness (PEH), provides a historical context of homelessness in the United States, an evaluation of the current state, and indispensable guidance for medical and behavioral health practitioners, case managers, housing navigators, and policy makers alike. It also serves as an inspiring source for those who are considering work in the public sector while reminding those of us in the field why we continue to do this challenging and rewarding work.
Tips can provide hope to clinicians
The volume is divided into four clear sections that are easy to navigate depending on your area of expertise and interest. Each chapter consolidates an extensive literature review into an intriguing and thought-provoking analysis. Part I, “The Big Picture – Social and Medical Issues,” focuses on conditions that disproportionately affect those who are unhoused. The authors offer a glimpse into the unique challenges of managing routine health conditions. They also detail the practical knowledge that’s needed to best care for our most vulnerable neighbors; for example, promoting a shared decision-making model; simplifying treatment plans; prescribing, when possible, medications that are dosed daily – instead of multiple times per day; allowing for walk-in appointments; and addressing cultural, linguistic, and educational barriers.
Most chapters highlight informative case examples that bring the text to life. It can be heartbreaking to recognize and witness the inhumane conditions in which PEH live, and these practical tips and suggestions for future policies based on best practices can help prevent burnout and provide hope for those who care for this community.
Part II, “Psychiatric Issues and Treatments,” presents a brief yet comprehensive history on homelessness, beginning with the deep shame that PEH experienced in Colonial times as the result of cultural and religious influences. Sadly, that negative judgment continues to this day.
The authors also explain how deinstitutionalization and transinstitutionalization have shaped the current state of homelessness, including why many PEH receive their care in emergency departments while incarcerated. This section highlights the barriers of care that are created not just by the patient, but also by the clinicians and systems of care – and what’s needed practically to overcome those challenges.
I appreciate the chapter on substance use disorders. It reminds us that the most commonly used substance among PEH is tobacco, which has serious health effects and for which we have treatment; nevertheless, . This section also provides examples of the trauma-informed language to use when addressing difficult and sometimes stigmatizing topics, such as survival sex and trauma history.
The evidence-based discussion continues in Part III with a focus on topics that everyone working with PEH should understand, including food insecurity, the criminal justice system, and sex trafficking. Part IV highlights best practices that should be replicated in every community, including Housing First approaches, medical respite care, and multiple Veterans Administration programs.
Throughout the text, major themes reverberate across the chapters, beginning with empathy. All who work with PEH must understand the conditions and challenges PEH face every day that affect their physical and mental health. The authors offer a stark and pointed reminder that being unhoused amounts to a full-time job just to meet basic needs. In addition, the devastating role of trauma and structural racism in creating and promoting the conditions that lead someone to be unhoused cannot be underestimated.
Fortunately, the primary aim of the book is to highlight solutions, and it’s here that the book shines. While some interventions are well-known, such as the importance of working in multidisciplinary teams, building trust and rapport with our patients, and urging clinicians and institutions to examine their own judgments and biases that might interfere with humane treatment, other suggestions will lead some readers into new territory. The authors, for example, maintain that we need more data and evidence-based research that include PEH. They also make a case for more preventive care and enhanced professional education for all health care workers that centers on trauma-informed care, social determinants of health, and the unique needs of especially vulnerable communities, such as the unhoused LBGTQ+ community and policies that promote best practices, such as Housing First. The book is a stirring read. It offers both inspiration and practical guidance for all who are currently working with or interested in caring for people experiencing homelessness.
Dr. Bird is a psychiatrist with Alameda County Health Care for the Homeless and the TRUST Clinic in Oakland, Calif. She also is a cofounder of StreetHealth, a backpack street medicine team that provides psychiatric and substance use disorder treatment to people experiencing homelessness in downtown Oakland.
Dr. Bird has no disclosures.
As a psychiatrist dedicated to working with people who are experiencing homelessness, I was very impressed with the new book edited by Col. (Ret.) Elspeth Cameron Ritchie, MD, MPH, and Maria D. Llorente, MD, about treating and providing services to this vulnerable population.
The book, “Clinical Management of the Homeless Patient: Social, Psychiatric, and Medical Issues” (Cham, Switzerland: Springer Nature Switzerland, 2021), offers an in-depth review and analysis of the biopsychosocial complexities that affect how medical and behavioral health conditions present in those who are unhoused. Notably, the book recommends with great sensitivity best practices to address these conditions with care, understanding, and love.
This text, invaluable in particular for those of us clinicians who work with people experiencing homelessness (PEH), provides a historical context of homelessness in the United States, an evaluation of the current state, and indispensable guidance for medical and behavioral health practitioners, case managers, housing navigators, and policy makers alike. It also serves as an inspiring source for those who are considering work in the public sector while reminding those of us in the field why we continue to do this challenging and rewarding work.
Tips can provide hope to clinicians
The volume is divided into four clear sections that are easy to navigate depending on your area of expertise and interest. Each chapter consolidates an extensive literature review into an intriguing and thought-provoking analysis. Part I, “The Big Picture – Social and Medical Issues,” focuses on conditions that disproportionately affect those who are unhoused. The authors offer a glimpse into the unique challenges of managing routine health conditions. They also detail the practical knowledge that’s needed to best care for our most vulnerable neighbors; for example, promoting a shared decision-making model; simplifying treatment plans; prescribing, when possible, medications that are dosed daily – instead of multiple times per day; allowing for walk-in appointments; and addressing cultural, linguistic, and educational barriers.
Most chapters highlight informative case examples that bring the text to life. It can be heartbreaking to recognize and witness the inhumane conditions in which PEH live, and these practical tips and suggestions for future policies based on best practices can help prevent burnout and provide hope for those who care for this community.
Part II, “Psychiatric Issues and Treatments,” presents a brief yet comprehensive history on homelessness, beginning with the deep shame that PEH experienced in Colonial times as the result of cultural and religious influences. Sadly, that negative judgment continues to this day.
The authors also explain how deinstitutionalization and transinstitutionalization have shaped the current state of homelessness, including why many PEH receive their care in emergency departments while incarcerated. This section highlights the barriers of care that are created not just by the patient, but also by the clinicians and systems of care – and what’s needed practically to overcome those challenges.
I appreciate the chapter on substance use disorders. It reminds us that the most commonly used substance among PEH is tobacco, which has serious health effects and for which we have treatment; nevertheless, . This section also provides examples of the trauma-informed language to use when addressing difficult and sometimes stigmatizing topics, such as survival sex and trauma history.
The evidence-based discussion continues in Part III with a focus on topics that everyone working with PEH should understand, including food insecurity, the criminal justice system, and sex trafficking. Part IV highlights best practices that should be replicated in every community, including Housing First approaches, medical respite care, and multiple Veterans Administration programs.
Throughout the text, major themes reverberate across the chapters, beginning with empathy. All who work with PEH must understand the conditions and challenges PEH face every day that affect their physical and mental health. The authors offer a stark and pointed reminder that being unhoused amounts to a full-time job just to meet basic needs. In addition, the devastating role of trauma and structural racism in creating and promoting the conditions that lead someone to be unhoused cannot be underestimated.
Fortunately, the primary aim of the book is to highlight solutions, and it’s here that the book shines. While some interventions are well-known, such as the importance of working in multidisciplinary teams, building trust and rapport with our patients, and urging clinicians and institutions to examine their own judgments and biases that might interfere with humane treatment, other suggestions will lead some readers into new territory. The authors, for example, maintain that we need more data and evidence-based research that include PEH. They also make a case for more preventive care and enhanced professional education for all health care workers that centers on trauma-informed care, social determinants of health, and the unique needs of especially vulnerable communities, such as the unhoused LBGTQ+ community and policies that promote best practices, such as Housing First. The book is a stirring read. It offers both inspiration and practical guidance for all who are currently working with or interested in caring for people experiencing homelessness.
Dr. Bird is a psychiatrist with Alameda County Health Care for the Homeless and the TRUST Clinic in Oakland, Calif. She also is a cofounder of StreetHealth, a backpack street medicine team that provides psychiatric and substance use disorder treatment to people experiencing homelessness in downtown Oakland.
Dr. Bird has no disclosures.
As a psychiatrist dedicated to working with people who are experiencing homelessness, I was very impressed with the new book edited by Col. (Ret.) Elspeth Cameron Ritchie, MD, MPH, and Maria D. Llorente, MD, about treating and providing services to this vulnerable population.
The book, “Clinical Management of the Homeless Patient: Social, Psychiatric, and Medical Issues” (Cham, Switzerland: Springer Nature Switzerland, 2021), offers an in-depth review and analysis of the biopsychosocial complexities that affect how medical and behavioral health conditions present in those who are unhoused. Notably, the book recommends with great sensitivity best practices to address these conditions with care, understanding, and love.
This text, invaluable in particular for those of us clinicians who work with people experiencing homelessness (PEH), provides a historical context of homelessness in the United States, an evaluation of the current state, and indispensable guidance for medical and behavioral health practitioners, case managers, housing navigators, and policy makers alike. It also serves as an inspiring source for those who are considering work in the public sector while reminding those of us in the field why we continue to do this challenging and rewarding work.
Tips can provide hope to clinicians
The volume is divided into four clear sections that are easy to navigate depending on your area of expertise and interest. Each chapter consolidates an extensive literature review into an intriguing and thought-provoking analysis. Part I, “The Big Picture – Social and Medical Issues,” focuses on conditions that disproportionately affect those who are unhoused. The authors offer a glimpse into the unique challenges of managing routine health conditions. They also detail the practical knowledge that’s needed to best care for our most vulnerable neighbors; for example, promoting a shared decision-making model; simplifying treatment plans; prescribing, when possible, medications that are dosed daily – instead of multiple times per day; allowing for walk-in appointments; and addressing cultural, linguistic, and educational barriers.
Most chapters highlight informative case examples that bring the text to life. It can be heartbreaking to recognize and witness the inhumane conditions in which PEH live, and these practical tips and suggestions for future policies based on best practices can help prevent burnout and provide hope for those who care for this community.
Part II, “Psychiatric Issues and Treatments,” presents a brief yet comprehensive history on homelessness, beginning with the deep shame that PEH experienced in Colonial times as the result of cultural and religious influences. Sadly, that negative judgment continues to this day.
The authors also explain how deinstitutionalization and transinstitutionalization have shaped the current state of homelessness, including why many PEH receive their care in emergency departments while incarcerated. This section highlights the barriers of care that are created not just by the patient, but also by the clinicians and systems of care – and what’s needed practically to overcome those challenges.
I appreciate the chapter on substance use disorders. It reminds us that the most commonly used substance among PEH is tobacco, which has serious health effects and for which we have treatment; nevertheless, . This section also provides examples of the trauma-informed language to use when addressing difficult and sometimes stigmatizing topics, such as survival sex and trauma history.
The evidence-based discussion continues in Part III with a focus on topics that everyone working with PEH should understand, including food insecurity, the criminal justice system, and sex trafficking. Part IV highlights best practices that should be replicated in every community, including Housing First approaches, medical respite care, and multiple Veterans Administration programs.
Throughout the text, major themes reverberate across the chapters, beginning with empathy. All who work with PEH must understand the conditions and challenges PEH face every day that affect their physical and mental health. The authors offer a stark and pointed reminder that being unhoused amounts to a full-time job just to meet basic needs. In addition, the devastating role of trauma and structural racism in creating and promoting the conditions that lead someone to be unhoused cannot be underestimated.
Fortunately, the primary aim of the book is to highlight solutions, and it’s here that the book shines. While some interventions are well-known, such as the importance of working in multidisciplinary teams, building trust and rapport with our patients, and urging clinicians and institutions to examine their own judgments and biases that might interfere with humane treatment, other suggestions will lead some readers into new territory. The authors, for example, maintain that we need more data and evidence-based research that include PEH. They also make a case for more preventive care and enhanced professional education for all health care workers that centers on trauma-informed care, social determinants of health, and the unique needs of especially vulnerable communities, such as the unhoused LBGTQ+ community and policies that promote best practices, such as Housing First. The book is a stirring read. It offers both inspiration and practical guidance for all who are currently working with or interested in caring for people experiencing homelessness.
Dr. Bird is a psychiatrist with Alameda County Health Care for the Homeless and the TRUST Clinic in Oakland, Calif. She also is a cofounder of StreetHealth, a backpack street medicine team that provides psychiatric and substance use disorder treatment to people experiencing homelessness in downtown Oakland.
Dr. Bird has no disclosures.
Last call? Moderate alcohol’s health benefits look increasingly doubtful
When holiday shoppers recently went to their local liquor stores in search of some liquid spirit, many were instead greeted by the sight of increasingly barren shelves.
Although partly a result of global supply chain issues, this was also yet more evidence of the rising demand for alcohol among adults during these difficult COVID years. It’s a trend that has led to concerns of an echo pandemic of alcohol-related morbidity, which has begun to play out in the form of rising rates of gastrointestinal and liver disease, hospital admissions for alcoholic hepatitis, and alcohol-related incidents of domestic violence.
Those who imbibe alcohol in low to moderate levels may not see themselves reflected in such stories of drinking’s hefty tolls. They’re instead following established health guidance that a little bit of alcohol now and then actually has robust health benefits. Yet the past few of years have seen a notable fraying of this idea, as emerging data calls into question whether alcohol in moderation should really continue to be just what the doctor ordered.
Behind the curve: Alcohol’s diminishing cardioprotective value
Perhaps the most resonant argument for the benefits of light to moderate alcohol consumption – usually defined as between one to two drinks a day – has been its proposed cardioprotective value. In this way, alcohol differs from tobacco, which is unsafe at any level. Alcohol’s proposed cardioprotective effects are often represented as a J-shaped curve, with moderate drinking occupying the sweet spot between teetotaling and heavy/binge drinking when it comes to reduced mortality.
In reality, this association is more likely “a statistical artifact” largely derived from low-quality observational studies, according to Christopher Labos, MD, CM, MSc, an epidemiologist and cardiologist at the Queen Elizabeth Health Complex in Montreal.
“When you look at studies that correct for things like reverse causation, or the fact that some people who drink zero alcohol are former drinkers who used to drink alcohol, then you realize that the protective benefit of alcohol is either minimal or nonexistent and that alcohol does more harm than good to our society,” said Dr. Labos, who detailed the reasons underpinning alcohol’s unearned cardioprotective reputation in a 2020 Medscape commentary.
This statistical limitation was on display in July 2021 when BMC Medicine published results from meta-analyses suggesting that current drinkers need not stop consuming small amounts of alcohol for the secondary prevention of cardiovascular disease (CVD). The study’s own investigators noted that it likely overestimated the reduced risk of CVD by including former heavy drinkers as nondrinkers.
Even if the J-shaped curve exists, its simplicity is deceiving. CVD risk increases alongside alcohol consumption owning to a complicated array of genetic and lifestyle factors. The curve also presents something of a catch-22. If you like alcohol enough to drink it every day, staying at the nadir of the curve where you’d gain the most benefits may prove challenging.
Another factor dimming alcohol’s cardioprotective reputation came via recent data that atrial fibrillation episodes can be triggered by acute alcohol use. A randomized, controlled trial published in the New England Journal of Medicine concluded that abstinence reduced arrhythmia recurrences in regular drinkers with atrial fibrillation.
“If we can replicate that, I think we’ll find that reducing alcohol consumption might be a very effective way to prevent and treat atrial fibrillation,” said Dr. Labos.
However, J-curve proponents will note the publication of study data from the UK Biobank indicating that low levels of alcohol consumption confers the greatest reduction in atrial fibrillation risk.
An overlooked carcinogen no longer
Surveys indicate that less than half of Americans realize alcohol increases cancer risk. That might have changed just a bit this year. In early 2021, an epidemiological analysis estimated that alcohol contributed to 4.8% of cancer cases and 3.2% of cancer deaths in the United States. Then the Lancet Oncology published the results of a high-profile, population-based study on the global burden of cancer as a result of alcoho. Although the main takeaway message was that 4% of new cancer cases worldwide in 2020 were attributable to alcohol, it was also noteworthy that moderate drinking accounted for 103,100 out of 741,300 of these projected annual cases.
“The risk of cancer increases even with low or moderate levels of drinking,” said the study’s lead author Harriet Rumgay, BSc, from the International Agency for Research on Cancer in Lyon, France. “Drinking less means you’ll have a lower risk of cancer than if you drink heavily, but there is no safe limit of alcohol consumption.”
The study linked alcohol consumption with an increased risk of at least seven different cancer types, including cancers of the oral cavity, pharynx, larynx, esophagus, colon, rectum, liver, and breast.
Although in North America men represented about two-thirds of the burden of cancer caused by alcohol, Ms. Rumgay added that “low and moderate levels of drinking [one or two alcoholic drinks per day] contributed relatively more cancer cases among women than among men.”
Yet more negative news for moderate alcohol drinkers arrived in August 2021, when a team of South Korean researchers published data in JAMA Network Open showing that, when it came to the risk of developing gastrointestinal cancers, even binge drinking may be preferable to continuous but moderate consumption.
who in updating its guidelines in 2020 after an 8-year interim offered this succinct piece of advice: “It is best not to drink alcohol.”
Neurotoxic implications
There has similarly been a reconsideration of the effects of moderate alcohol consumption on brain health.
A recent report of multimodal MRI brain and cognitive testing data from over 25,000 participants in the UK Biobank study indicate that alcohol may have no safe dosage . Even moderate consumption reduced gray matter volume and functional connectivity, negative associations that were increased in those with higher blood pressure and body mass index.
Speaking with this news organization in May 2021, an investigator said: “The size of the effect is small, albeit greater than any other modifiable risk factor,” noting that the changes have been linked to decreased memory and dementia.
Louise Mewton, PhD, from the Center for Healthy Brain Aging at the University of New South Wales, Sydney, said that these results provide an interesting comparison with others into the association between alcohol and dementia.
“A recent study of over 1 million dementia cases in France indicated that problematic alcohol use (alcohol use disorders) were one of the strongest risk factors for dementia – even more so than things like high blood pressure and diabetes,” Dr. Mewton said in an interview. In comparison, “the most-recent reviews indicate that 4 drinks/week is associated with the lowest risk for dementia – so we’re talking about very low levels of alcohol use in terms of maintaining brain health.
“Understanding why very small amounts of alcohol appear to be protective in terms of dementia but damaging when we look at brain scans is something that would be really interesting to unpack.”
Dr. Mewton and colleagues recently published data suggesting that there are three periods when the brain might be particularly susceptible to alcohol’s neurotoxic effects: gestation (from conception to birth), later adolescence (15-19 years), and older adulthood (over 65 years). Directing behavioral interventions to patients in these stages may therefore be beneficial.
And there’s no time too soon to promote abstinence among those with alcohol use disorder, as brain damage is shown to still occur even in the immediate period after people cease drinking.
Although in one more argument for the J-shaped curve’s relevance, data from the Massachusetts General Brigham Biobank recently indicated that moderate alcohol use, unlike low and heavy use, lowered both stress-related neurobiological activity and major adverse cardiovascular events.
Getting patients to reconsider alcohol’s ‘benefits’
These new findings mean physicians will find themselves imparting a more nuanced message about the health impacts of moderate alcohol consumption than in prior years. To aid those efforts, Ms. Rumgay advised clinicians to consult a special issue of the journal Nutrients that features review articles of alcohol›s impact on various health outcomes.
Ms. Rumgay also supports broader policy changes.
“There is some evidence that adding cancer warnings to alcohol labels, similar to those used on cigarette packages, might deter people from purchasing alcohol products and increase awareness of the causal link with cancer,” she said. “But the most effective ways of reducing alcohol use in the population are through increasing the price of alcohol through higher taxes, limiting purchasing availability, and reducing marketing of alcohol brands to the public.”
Dr. Mewton recommended various interventions for patients who still find it difficult to curtail their drinking.
“For less severe, problematic use, things like cognitive-behavioral therapy and motivational therapy are very effective in reducing alcohol consumption,” she said in an interview.
For all the discussion about how the COVID-19 pandemic has exacerbated problematic drinking, it has also provided an opportunity for getting patients to reexamine their relationship to alcohol. And as Dr. Labos noted, emerging data on alcohol’s negative effects probably won’t be considered earth-shattering to most patients.
“Deep down, I think most people know that alcohol is not healthy, but it is part of our social culture and so we find ways to justify our own behavior,” he said in an interview.
Dr. Labos suggested that clinicians reframe alcohol in their patients’ minds for what it really is – “an indulgence that we shouldn’t have too much of very often.
“Just like junk food, that doesn’t mean we can’t enjoy small amounts occasionally, but we have to stop presenting that it is good for us, because it isn’t.”
A version of this article first appeared on Medscape.com.
When holiday shoppers recently went to their local liquor stores in search of some liquid spirit, many were instead greeted by the sight of increasingly barren shelves.
Although partly a result of global supply chain issues, this was also yet more evidence of the rising demand for alcohol among adults during these difficult COVID years. It’s a trend that has led to concerns of an echo pandemic of alcohol-related morbidity, which has begun to play out in the form of rising rates of gastrointestinal and liver disease, hospital admissions for alcoholic hepatitis, and alcohol-related incidents of domestic violence.
Those who imbibe alcohol in low to moderate levels may not see themselves reflected in such stories of drinking’s hefty tolls. They’re instead following established health guidance that a little bit of alcohol now and then actually has robust health benefits. Yet the past few of years have seen a notable fraying of this idea, as emerging data calls into question whether alcohol in moderation should really continue to be just what the doctor ordered.
Behind the curve: Alcohol’s diminishing cardioprotective value
Perhaps the most resonant argument for the benefits of light to moderate alcohol consumption – usually defined as between one to two drinks a day – has been its proposed cardioprotective value. In this way, alcohol differs from tobacco, which is unsafe at any level. Alcohol’s proposed cardioprotective effects are often represented as a J-shaped curve, with moderate drinking occupying the sweet spot between teetotaling and heavy/binge drinking when it comes to reduced mortality.
In reality, this association is more likely “a statistical artifact” largely derived from low-quality observational studies, according to Christopher Labos, MD, CM, MSc, an epidemiologist and cardiologist at the Queen Elizabeth Health Complex in Montreal.
“When you look at studies that correct for things like reverse causation, or the fact that some people who drink zero alcohol are former drinkers who used to drink alcohol, then you realize that the protective benefit of alcohol is either minimal or nonexistent and that alcohol does more harm than good to our society,” said Dr. Labos, who detailed the reasons underpinning alcohol’s unearned cardioprotective reputation in a 2020 Medscape commentary.
This statistical limitation was on display in July 2021 when BMC Medicine published results from meta-analyses suggesting that current drinkers need not stop consuming small amounts of alcohol for the secondary prevention of cardiovascular disease (CVD). The study’s own investigators noted that it likely overestimated the reduced risk of CVD by including former heavy drinkers as nondrinkers.
Even if the J-shaped curve exists, its simplicity is deceiving. CVD risk increases alongside alcohol consumption owning to a complicated array of genetic and lifestyle factors. The curve also presents something of a catch-22. If you like alcohol enough to drink it every day, staying at the nadir of the curve where you’d gain the most benefits may prove challenging.
Another factor dimming alcohol’s cardioprotective reputation came via recent data that atrial fibrillation episodes can be triggered by acute alcohol use. A randomized, controlled trial published in the New England Journal of Medicine concluded that abstinence reduced arrhythmia recurrences in regular drinkers with atrial fibrillation.
“If we can replicate that, I think we’ll find that reducing alcohol consumption might be a very effective way to prevent and treat atrial fibrillation,” said Dr. Labos.
However, J-curve proponents will note the publication of study data from the UK Biobank indicating that low levels of alcohol consumption confers the greatest reduction in atrial fibrillation risk.
An overlooked carcinogen no longer
Surveys indicate that less than half of Americans realize alcohol increases cancer risk. That might have changed just a bit this year. In early 2021, an epidemiological analysis estimated that alcohol contributed to 4.8% of cancer cases and 3.2% of cancer deaths in the United States. Then the Lancet Oncology published the results of a high-profile, population-based study on the global burden of cancer as a result of alcoho. Although the main takeaway message was that 4% of new cancer cases worldwide in 2020 were attributable to alcohol, it was also noteworthy that moderate drinking accounted for 103,100 out of 741,300 of these projected annual cases.
“The risk of cancer increases even with low or moderate levels of drinking,” said the study’s lead author Harriet Rumgay, BSc, from the International Agency for Research on Cancer in Lyon, France. “Drinking less means you’ll have a lower risk of cancer than if you drink heavily, but there is no safe limit of alcohol consumption.”
The study linked alcohol consumption with an increased risk of at least seven different cancer types, including cancers of the oral cavity, pharynx, larynx, esophagus, colon, rectum, liver, and breast.
Although in North America men represented about two-thirds of the burden of cancer caused by alcohol, Ms. Rumgay added that “low and moderate levels of drinking [one or two alcoholic drinks per day] contributed relatively more cancer cases among women than among men.”
Yet more negative news for moderate alcohol drinkers arrived in August 2021, when a team of South Korean researchers published data in JAMA Network Open showing that, when it came to the risk of developing gastrointestinal cancers, even binge drinking may be preferable to continuous but moderate consumption.
who in updating its guidelines in 2020 after an 8-year interim offered this succinct piece of advice: “It is best not to drink alcohol.”
Neurotoxic implications
There has similarly been a reconsideration of the effects of moderate alcohol consumption on brain health.
A recent report of multimodal MRI brain and cognitive testing data from over 25,000 participants in the UK Biobank study indicate that alcohol may have no safe dosage . Even moderate consumption reduced gray matter volume and functional connectivity, negative associations that were increased in those with higher blood pressure and body mass index.
Speaking with this news organization in May 2021, an investigator said: “The size of the effect is small, albeit greater than any other modifiable risk factor,” noting that the changes have been linked to decreased memory and dementia.
Louise Mewton, PhD, from the Center for Healthy Brain Aging at the University of New South Wales, Sydney, said that these results provide an interesting comparison with others into the association between alcohol and dementia.
“A recent study of over 1 million dementia cases in France indicated that problematic alcohol use (alcohol use disorders) were one of the strongest risk factors for dementia – even more so than things like high blood pressure and diabetes,” Dr. Mewton said in an interview. In comparison, “the most-recent reviews indicate that 4 drinks/week is associated with the lowest risk for dementia – so we’re talking about very low levels of alcohol use in terms of maintaining brain health.
“Understanding why very small amounts of alcohol appear to be protective in terms of dementia but damaging when we look at brain scans is something that would be really interesting to unpack.”
Dr. Mewton and colleagues recently published data suggesting that there are three periods when the brain might be particularly susceptible to alcohol’s neurotoxic effects: gestation (from conception to birth), later adolescence (15-19 years), and older adulthood (over 65 years). Directing behavioral interventions to patients in these stages may therefore be beneficial.
And there’s no time too soon to promote abstinence among those with alcohol use disorder, as brain damage is shown to still occur even in the immediate period after people cease drinking.
Although in one more argument for the J-shaped curve’s relevance, data from the Massachusetts General Brigham Biobank recently indicated that moderate alcohol use, unlike low and heavy use, lowered both stress-related neurobiological activity and major adverse cardiovascular events.
Getting patients to reconsider alcohol’s ‘benefits’
These new findings mean physicians will find themselves imparting a more nuanced message about the health impacts of moderate alcohol consumption than in prior years. To aid those efforts, Ms. Rumgay advised clinicians to consult a special issue of the journal Nutrients that features review articles of alcohol›s impact on various health outcomes.
Ms. Rumgay also supports broader policy changes.
“There is some evidence that adding cancer warnings to alcohol labels, similar to those used on cigarette packages, might deter people from purchasing alcohol products and increase awareness of the causal link with cancer,” she said. “But the most effective ways of reducing alcohol use in the population are through increasing the price of alcohol through higher taxes, limiting purchasing availability, and reducing marketing of alcohol brands to the public.”
Dr. Mewton recommended various interventions for patients who still find it difficult to curtail their drinking.
“For less severe, problematic use, things like cognitive-behavioral therapy and motivational therapy are very effective in reducing alcohol consumption,” she said in an interview.
For all the discussion about how the COVID-19 pandemic has exacerbated problematic drinking, it has also provided an opportunity for getting patients to reexamine their relationship to alcohol. And as Dr. Labos noted, emerging data on alcohol’s negative effects probably won’t be considered earth-shattering to most patients.
“Deep down, I think most people know that alcohol is not healthy, but it is part of our social culture and so we find ways to justify our own behavior,” he said in an interview.
Dr. Labos suggested that clinicians reframe alcohol in their patients’ minds for what it really is – “an indulgence that we shouldn’t have too much of very often.
“Just like junk food, that doesn’t mean we can’t enjoy small amounts occasionally, but we have to stop presenting that it is good for us, because it isn’t.”
A version of this article first appeared on Medscape.com.
When holiday shoppers recently went to their local liquor stores in search of some liquid spirit, many were instead greeted by the sight of increasingly barren shelves.
Although partly a result of global supply chain issues, this was also yet more evidence of the rising demand for alcohol among adults during these difficult COVID years. It’s a trend that has led to concerns of an echo pandemic of alcohol-related morbidity, which has begun to play out in the form of rising rates of gastrointestinal and liver disease, hospital admissions for alcoholic hepatitis, and alcohol-related incidents of domestic violence.
Those who imbibe alcohol in low to moderate levels may not see themselves reflected in such stories of drinking’s hefty tolls. They’re instead following established health guidance that a little bit of alcohol now and then actually has robust health benefits. Yet the past few of years have seen a notable fraying of this idea, as emerging data calls into question whether alcohol in moderation should really continue to be just what the doctor ordered.
Behind the curve: Alcohol’s diminishing cardioprotective value
Perhaps the most resonant argument for the benefits of light to moderate alcohol consumption – usually defined as between one to two drinks a day – has been its proposed cardioprotective value. In this way, alcohol differs from tobacco, which is unsafe at any level. Alcohol’s proposed cardioprotective effects are often represented as a J-shaped curve, with moderate drinking occupying the sweet spot between teetotaling and heavy/binge drinking when it comes to reduced mortality.
In reality, this association is more likely “a statistical artifact” largely derived from low-quality observational studies, according to Christopher Labos, MD, CM, MSc, an epidemiologist and cardiologist at the Queen Elizabeth Health Complex in Montreal.
“When you look at studies that correct for things like reverse causation, or the fact that some people who drink zero alcohol are former drinkers who used to drink alcohol, then you realize that the protective benefit of alcohol is either minimal or nonexistent and that alcohol does more harm than good to our society,” said Dr. Labos, who detailed the reasons underpinning alcohol’s unearned cardioprotective reputation in a 2020 Medscape commentary.
This statistical limitation was on display in July 2021 when BMC Medicine published results from meta-analyses suggesting that current drinkers need not stop consuming small amounts of alcohol for the secondary prevention of cardiovascular disease (CVD). The study’s own investigators noted that it likely overestimated the reduced risk of CVD by including former heavy drinkers as nondrinkers.
Even if the J-shaped curve exists, its simplicity is deceiving. CVD risk increases alongside alcohol consumption owning to a complicated array of genetic and lifestyle factors. The curve also presents something of a catch-22. If you like alcohol enough to drink it every day, staying at the nadir of the curve where you’d gain the most benefits may prove challenging.
Another factor dimming alcohol’s cardioprotective reputation came via recent data that atrial fibrillation episodes can be triggered by acute alcohol use. A randomized, controlled trial published in the New England Journal of Medicine concluded that abstinence reduced arrhythmia recurrences in regular drinkers with atrial fibrillation.
“If we can replicate that, I think we’ll find that reducing alcohol consumption might be a very effective way to prevent and treat atrial fibrillation,” said Dr. Labos.
However, J-curve proponents will note the publication of study data from the UK Biobank indicating that low levels of alcohol consumption confers the greatest reduction in atrial fibrillation risk.
An overlooked carcinogen no longer
Surveys indicate that less than half of Americans realize alcohol increases cancer risk. That might have changed just a bit this year. In early 2021, an epidemiological analysis estimated that alcohol contributed to 4.8% of cancer cases and 3.2% of cancer deaths in the United States. Then the Lancet Oncology published the results of a high-profile, population-based study on the global burden of cancer as a result of alcoho. Although the main takeaway message was that 4% of new cancer cases worldwide in 2020 were attributable to alcohol, it was also noteworthy that moderate drinking accounted for 103,100 out of 741,300 of these projected annual cases.
“The risk of cancer increases even with low or moderate levels of drinking,” said the study’s lead author Harriet Rumgay, BSc, from the International Agency for Research on Cancer in Lyon, France. “Drinking less means you’ll have a lower risk of cancer than if you drink heavily, but there is no safe limit of alcohol consumption.”
The study linked alcohol consumption with an increased risk of at least seven different cancer types, including cancers of the oral cavity, pharynx, larynx, esophagus, colon, rectum, liver, and breast.
Although in North America men represented about two-thirds of the burden of cancer caused by alcohol, Ms. Rumgay added that “low and moderate levels of drinking [one or two alcoholic drinks per day] contributed relatively more cancer cases among women than among men.”
Yet more negative news for moderate alcohol drinkers arrived in August 2021, when a team of South Korean researchers published data in JAMA Network Open showing that, when it came to the risk of developing gastrointestinal cancers, even binge drinking may be preferable to continuous but moderate consumption.
who in updating its guidelines in 2020 after an 8-year interim offered this succinct piece of advice: “It is best not to drink alcohol.”
Neurotoxic implications
There has similarly been a reconsideration of the effects of moderate alcohol consumption on brain health.
A recent report of multimodal MRI brain and cognitive testing data from over 25,000 participants in the UK Biobank study indicate that alcohol may have no safe dosage . Even moderate consumption reduced gray matter volume and functional connectivity, negative associations that were increased in those with higher blood pressure and body mass index.
Speaking with this news organization in May 2021, an investigator said: “The size of the effect is small, albeit greater than any other modifiable risk factor,” noting that the changes have been linked to decreased memory and dementia.
Louise Mewton, PhD, from the Center for Healthy Brain Aging at the University of New South Wales, Sydney, said that these results provide an interesting comparison with others into the association between alcohol and dementia.
“A recent study of over 1 million dementia cases in France indicated that problematic alcohol use (alcohol use disorders) were one of the strongest risk factors for dementia – even more so than things like high blood pressure and diabetes,” Dr. Mewton said in an interview. In comparison, “the most-recent reviews indicate that 4 drinks/week is associated with the lowest risk for dementia – so we’re talking about very low levels of alcohol use in terms of maintaining brain health.
“Understanding why very small amounts of alcohol appear to be protective in terms of dementia but damaging when we look at brain scans is something that would be really interesting to unpack.”
Dr. Mewton and colleagues recently published data suggesting that there are three periods when the brain might be particularly susceptible to alcohol’s neurotoxic effects: gestation (from conception to birth), later adolescence (15-19 years), and older adulthood (over 65 years). Directing behavioral interventions to patients in these stages may therefore be beneficial.
And there’s no time too soon to promote abstinence among those with alcohol use disorder, as brain damage is shown to still occur even in the immediate period after people cease drinking.
Although in one more argument for the J-shaped curve’s relevance, data from the Massachusetts General Brigham Biobank recently indicated that moderate alcohol use, unlike low and heavy use, lowered both stress-related neurobiological activity and major adverse cardiovascular events.
Getting patients to reconsider alcohol’s ‘benefits’
These new findings mean physicians will find themselves imparting a more nuanced message about the health impacts of moderate alcohol consumption than in prior years. To aid those efforts, Ms. Rumgay advised clinicians to consult a special issue of the journal Nutrients that features review articles of alcohol›s impact on various health outcomes.
Ms. Rumgay also supports broader policy changes.
“There is some evidence that adding cancer warnings to alcohol labels, similar to those used on cigarette packages, might deter people from purchasing alcohol products and increase awareness of the causal link with cancer,” she said. “But the most effective ways of reducing alcohol use in the population are through increasing the price of alcohol through higher taxes, limiting purchasing availability, and reducing marketing of alcohol brands to the public.”
Dr. Mewton recommended various interventions for patients who still find it difficult to curtail their drinking.
“For less severe, problematic use, things like cognitive-behavioral therapy and motivational therapy are very effective in reducing alcohol consumption,” she said in an interview.
For all the discussion about how the COVID-19 pandemic has exacerbated problematic drinking, it has also provided an opportunity for getting patients to reexamine their relationship to alcohol. And as Dr. Labos noted, emerging data on alcohol’s negative effects probably won’t be considered earth-shattering to most patients.
“Deep down, I think most people know that alcohol is not healthy, but it is part of our social culture and so we find ways to justify our own behavior,” he said in an interview.
Dr. Labos suggested that clinicians reframe alcohol in their patients’ minds for what it really is – “an indulgence that we shouldn’t have too much of very often.
“Just like junk food, that doesn’t mean we can’t enjoy small amounts occasionally, but we have to stop presenting that it is good for us, because it isn’t.”
A version of this article first appeared on Medscape.com.
Fish oil: ‘No net benefit’ for depression prevention?
Fish oil supplementation does not help prevent depression or boost mood, new research suggests.
The VITAL-DEP study included more than 18,000 participants. Among adults aged 50 years or older free of clinically relevant depressive symptoms at baseline, long-term use of marine omega-3 fatty acid (omega-3) supplements did not reduce risk for depression or clinically relevant depressive symptoms — or make a difference in the quality of mood.
“While a small increase in risk of depression was inside the statistical margin of significance, there was no harmful or beneficial effect of omega-3 on the overall course of mood during the roughly 5 to 7 years of follow-up,” lead author Olivia I. Okereke, MD, Massachusetts General Hospital and Harvard Medical School, Boston, told Medscape Medical News.
“The takeaway from our study is that there is no net benefit of long-term use of daily omega-3 fish oil supplements for preventing depression or boosting mood,” Okereke said.
The findings were published online Dec. 21 in JAMA.
Assessing general population risk
For many years, experts have recommended omega-3 supplements for reduction in depression recurrence in some high-risk patients, Okereke noted.
“However, there are no guidelines related to the use of omega-3 supplements for preventing depression in the general population. Therefore, we undertook this study to provide clarity in the issue,” she said.
The VITAL-DEP study enrolled 18,353 older adults (mean age, 67.5 years; 49% women). Of these, 16,657 were at risk for incident depression, defined as having no previous history of depression; and 1696 were at risk for recurrent depression, defined as having a history of depression but not having undergone treatment for depression within the past 2 years.
Roughly half the participants were randomly assigned to receive marine omega-3 fatty acids (1 g/d of fish oil, including 465 mg of eicosapentaenoic acid [EPA] and 375 mg of docosahexaenoic acid [DHA]) and the other half to matching placebo for an average of 5.3 years.
“Because of the large sample size and long follow-up, we were able to test the effects of daily omega-3 fish oil supplements on universal prevention of depression in the adult population,” Okereke said.
No significant benefit
Results showed risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the omega-3 group and the placebo group.
The omega-3 group had 651 depression or clinically relevant depressive symptom events (13.9 per 1000 person-years), and the placebo group had 583 depression or clinically relevant depressive symptom events (12.3 per 1000 person-years). The hazard ratio was 1.13 (95% CI, 1.01 - 1.26; P = .03).
There were also no significant between-group differences in longitudinal mood scores. The mean difference in change in 8-item Patient Health Questionnaire (PHQ-8) score was 0.03 points (95% CI, −0.01 to 0.07; P = .19).
“Patients, physicians, and other clinicians should understand that there are still many reasons for some people, under the guidance of their health care providers, to take omega-3 fish oil supplements,” Okereke noted.
“These supplements increasingly have been found to have benefits for cardiac disease prevention and treatment of inflammatory conditions, in addition to being used for management of existing depressive disorders in some high-risk patients,” she said.
“However, the results of our study indicate there is no reason for adults in the general population to be taking daily omega-3 fish oil supplements solely for the purpose of preventing depression or for maintaining a positive mood,” she added.
Okereke noted, however, that the VITAL-DEP study used 1 g/day of omega-3 fatty acids and there may be a greater benefit from taking higher doses, such as 4 g/day.
Cautionary notes
Commenting on the study for Medscape Medical News, Kuan-Pin Su, MD, PhD, chief of the Department of General Psychiatry, China Medical University, Taichung, Taiwan, highlighted some of the limitations cited by the investigators.
First, depression or depressive symptoms were defined using self-rating scales, which are “convenient to screen for depressive disorders, but a high score obtained on a self-rating scale does not necessarily indicate the presence of depressive psychopathology,” said Su, who was not involved with the research.
He also noted that use of 465 mg of EPA and 375 mg of DHA in VITAL-DEP “might be too low” to have an impact.
Finally, Su said it is “very important to also address the potential for type I error, which makes the secondary and subgroup analyses less reliable.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pronova BioPharma donated the fish oil and matching placebo. Okereke reported receiving royalties from Springer Publishing. Su is a founding committee member of the International Society for Nutritional Psychiatry Research, the board director of the International Society for the Study of Fatty Acids, and an associate editor of the journal Brain, Behavior, and Immunity.
A version of this article first appeared on Medscape.com.
Fish oil supplementation does not help prevent depression or boost mood, new research suggests.
The VITAL-DEP study included more than 18,000 participants. Among adults aged 50 years or older free of clinically relevant depressive symptoms at baseline, long-term use of marine omega-3 fatty acid (omega-3) supplements did not reduce risk for depression or clinically relevant depressive symptoms — or make a difference in the quality of mood.
“While a small increase in risk of depression was inside the statistical margin of significance, there was no harmful or beneficial effect of omega-3 on the overall course of mood during the roughly 5 to 7 years of follow-up,” lead author Olivia I. Okereke, MD, Massachusetts General Hospital and Harvard Medical School, Boston, told Medscape Medical News.
“The takeaway from our study is that there is no net benefit of long-term use of daily omega-3 fish oil supplements for preventing depression or boosting mood,” Okereke said.
The findings were published online Dec. 21 in JAMA.
Assessing general population risk
For many years, experts have recommended omega-3 supplements for reduction in depression recurrence in some high-risk patients, Okereke noted.
“However, there are no guidelines related to the use of omega-3 supplements for preventing depression in the general population. Therefore, we undertook this study to provide clarity in the issue,” she said.
The VITAL-DEP study enrolled 18,353 older adults (mean age, 67.5 years; 49% women). Of these, 16,657 were at risk for incident depression, defined as having no previous history of depression; and 1696 were at risk for recurrent depression, defined as having a history of depression but not having undergone treatment for depression within the past 2 years.
Roughly half the participants were randomly assigned to receive marine omega-3 fatty acids (1 g/d of fish oil, including 465 mg of eicosapentaenoic acid [EPA] and 375 mg of docosahexaenoic acid [DHA]) and the other half to matching placebo for an average of 5.3 years.
“Because of the large sample size and long follow-up, we were able to test the effects of daily omega-3 fish oil supplements on universal prevention of depression in the adult population,” Okereke said.
No significant benefit
Results showed risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the omega-3 group and the placebo group.
The omega-3 group had 651 depression or clinically relevant depressive symptom events (13.9 per 1000 person-years), and the placebo group had 583 depression or clinically relevant depressive symptom events (12.3 per 1000 person-years). The hazard ratio was 1.13 (95% CI, 1.01 - 1.26; P = .03).
There were also no significant between-group differences in longitudinal mood scores. The mean difference in change in 8-item Patient Health Questionnaire (PHQ-8) score was 0.03 points (95% CI, −0.01 to 0.07; P = .19).
“Patients, physicians, and other clinicians should understand that there are still many reasons for some people, under the guidance of their health care providers, to take omega-3 fish oil supplements,” Okereke noted.
“These supplements increasingly have been found to have benefits for cardiac disease prevention and treatment of inflammatory conditions, in addition to being used for management of existing depressive disorders in some high-risk patients,” she said.
“However, the results of our study indicate there is no reason for adults in the general population to be taking daily omega-3 fish oil supplements solely for the purpose of preventing depression or for maintaining a positive mood,” she added.
Okereke noted, however, that the VITAL-DEP study used 1 g/day of omega-3 fatty acids and there may be a greater benefit from taking higher doses, such as 4 g/day.
Cautionary notes
Commenting on the study for Medscape Medical News, Kuan-Pin Su, MD, PhD, chief of the Department of General Psychiatry, China Medical University, Taichung, Taiwan, highlighted some of the limitations cited by the investigators.
First, depression or depressive symptoms were defined using self-rating scales, which are “convenient to screen for depressive disorders, but a high score obtained on a self-rating scale does not necessarily indicate the presence of depressive psychopathology,” said Su, who was not involved with the research.
He also noted that use of 465 mg of EPA and 375 mg of DHA in VITAL-DEP “might be too low” to have an impact.
Finally, Su said it is “very important to also address the potential for type I error, which makes the secondary and subgroup analyses less reliable.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pronova BioPharma donated the fish oil and matching placebo. Okereke reported receiving royalties from Springer Publishing. Su is a founding committee member of the International Society for Nutritional Psychiatry Research, the board director of the International Society for the Study of Fatty Acids, and an associate editor of the journal Brain, Behavior, and Immunity.
A version of this article first appeared on Medscape.com.
Fish oil supplementation does not help prevent depression or boost mood, new research suggests.
The VITAL-DEP study included more than 18,000 participants. Among adults aged 50 years or older free of clinically relevant depressive symptoms at baseline, long-term use of marine omega-3 fatty acid (omega-3) supplements did not reduce risk for depression or clinically relevant depressive symptoms — or make a difference in the quality of mood.
“While a small increase in risk of depression was inside the statistical margin of significance, there was no harmful or beneficial effect of omega-3 on the overall course of mood during the roughly 5 to 7 years of follow-up,” lead author Olivia I. Okereke, MD, Massachusetts General Hospital and Harvard Medical School, Boston, told Medscape Medical News.
“The takeaway from our study is that there is no net benefit of long-term use of daily omega-3 fish oil supplements for preventing depression or boosting mood,” Okereke said.
The findings were published online Dec. 21 in JAMA.
Assessing general population risk
For many years, experts have recommended omega-3 supplements for reduction in depression recurrence in some high-risk patients, Okereke noted.
“However, there are no guidelines related to the use of omega-3 supplements for preventing depression in the general population. Therefore, we undertook this study to provide clarity in the issue,” she said.
The VITAL-DEP study enrolled 18,353 older adults (mean age, 67.5 years; 49% women). Of these, 16,657 were at risk for incident depression, defined as having no previous history of depression; and 1696 were at risk for recurrent depression, defined as having a history of depression but not having undergone treatment for depression within the past 2 years.
Roughly half the participants were randomly assigned to receive marine omega-3 fatty acids (1 g/d of fish oil, including 465 mg of eicosapentaenoic acid [EPA] and 375 mg of docosahexaenoic acid [DHA]) and the other half to matching placebo for an average of 5.3 years.
“Because of the large sample size and long follow-up, we were able to test the effects of daily omega-3 fish oil supplements on universal prevention of depression in the adult population,” Okereke said.
No significant benefit
Results showed risk for depression or clinically relevant depressive symptoms (total of incident and recurrent cases) was not significantly different between the omega-3 group and the placebo group.
The omega-3 group had 651 depression or clinically relevant depressive symptom events (13.9 per 1000 person-years), and the placebo group had 583 depression or clinically relevant depressive symptom events (12.3 per 1000 person-years). The hazard ratio was 1.13 (95% CI, 1.01 - 1.26; P = .03).
There were also no significant between-group differences in longitudinal mood scores. The mean difference in change in 8-item Patient Health Questionnaire (PHQ-8) score was 0.03 points (95% CI, −0.01 to 0.07; P = .19).
“Patients, physicians, and other clinicians should understand that there are still many reasons for some people, under the guidance of their health care providers, to take omega-3 fish oil supplements,” Okereke noted.
“These supplements increasingly have been found to have benefits for cardiac disease prevention and treatment of inflammatory conditions, in addition to being used for management of existing depressive disorders in some high-risk patients,” she said.
“However, the results of our study indicate there is no reason for adults in the general population to be taking daily omega-3 fish oil supplements solely for the purpose of preventing depression or for maintaining a positive mood,” she added.
Okereke noted, however, that the VITAL-DEP study used 1 g/day of omega-3 fatty acids and there may be a greater benefit from taking higher doses, such as 4 g/day.
Cautionary notes
Commenting on the study for Medscape Medical News, Kuan-Pin Su, MD, PhD, chief of the Department of General Psychiatry, China Medical University, Taichung, Taiwan, highlighted some of the limitations cited by the investigators.
First, depression or depressive symptoms were defined using self-rating scales, which are “convenient to screen for depressive disorders, but a high score obtained on a self-rating scale does not necessarily indicate the presence of depressive psychopathology,” said Su, who was not involved with the research.
He also noted that use of 465 mg of EPA and 375 mg of DHA in VITAL-DEP “might be too low” to have an impact.
Finally, Su said it is “very important to also address the potential for type I error, which makes the secondary and subgroup analyses less reliable.”
VITAL-DEP was supported by a grant from the National Institute of Mental Health. Pronova BioPharma donated the fish oil and matching placebo. Okereke reported receiving royalties from Springer Publishing. Su is a founding committee member of the International Society for Nutritional Psychiatry Research, the board director of the International Society for the Study of Fatty Acids, and an associate editor of the journal Brain, Behavior, and Immunity.
A version of this article first appeared on Medscape.com.
COVID-19 and coping with superimposed traumas
While 2022 is lurking around the corner, many of us still have 2020 on our minds. Social media posts are already emerging: “No new years resolutions. It is the circumstances turn to improve [sic],” one post declares. Others proclaim that it is difficult coming to terms with the idea that 2022 is actually pronounced “2020 too.” A critical difference exists between then and now – we have experienced months of living in limbo and rolling with the punches of pandemic life.
In some ways, it has become easy to think of the early pandemic days as a distant memory, yet respect that the impact of 2020 has been indelible for virtually all of us and feels palpable as if it were yesterday.
The year 2020 was marked by the beginning of the COVID-19 pandemic, which was accompanied by extreme uncertainty, loss of all kinds, and emotional turmoil. The early pandemic had a profound economic and social impact, with added stress tethered to political and race-related division in America that created divides among families and friends, and yielded ceaseless discourse related to divergent perspectives. This only exacerbated the stress that came with the pandemic, given that providing support and leaning on one another was more important than ever. All of this was compounded by natural disasters that have plagued the country.
So much was unprecedented. There was a collective sense of feeling “worn down,” and the burnout that was felt was quite profound. Enormous amounts of mental and physical effort were allocated to simply surviving, getting basic needs met, having enough food and supplies, and completing basic tasks. Ordinary relating felt taxing. At this stage of the pandemic, the COVID-19 experience can be conceived of as a traumatic stressor capable of eliciting a traumatic response and exacerbating other mental health symptoms. Our capacity to cope has been diminished. Anxiety rates have soared, as have rates of clinical depression. Those most affected have had lower household incomes, are unmarried, and have experienced pandemic-related stressors. The links between the impact of the pandemic on mental health have been clear.
The pandemic has forced the landscape of social support to dramatically change. Initially, we felt pulled to connect and we leaned into the use of virtual platforms to connect for all matters (simple social gatherings, big birthday events, family reunions, celebration of holidays, work duties, and academic work). However, “Zoom fatigue” began to set in, and our screen time was maxed out. There has been the added dynamic of frontline workers who did not have the option to work virtually or from home. This group largely has felt disconnected from others who didn’t understand the depth of their anxiety and loneliness of their experience. Health care workers have had to make challenging, life-and-death, patient-related decisions that called into question personal morals and ethics all while their own lives were at risk.
Fast-forward to the present, and support systems have either strengthened or worn down – which has yielded a unique dichotomy. Maintaining friendships has either felt of utmost importance given the impact of the disconnect and physical distance or has felt challenging given the mental energy expended from working and connecting virtually. Empathy burnout is also a real and important facet in the equation. We begin to ask the question: Are we checking in with others in the spirit of authentic relating, to cultivate real connection, or to check a box?
Impact of layered traumas
It is interesting to think about the pandemic’s traumatic impact being “superimposed” on top of the “ordinary traumas” experienced outside of the pandemic. We are essentially at the 2-year mark, in some ways have cultivated a sense of resilience and found ways to adapt, and in other ways at times feel right back where we were in early 2020. There were moments that felt hopeful, glimmers of normalcy, and setbacks that all ebbed and flowed – but even so, there have not been many “mental breaks,” only temporary and transient reprieves. Some got sick and died; some recovered; and others are still experiencing long-hauler syndrome and have lingering sequelae. Despite adaptation and resilience, one can’t help but wonder the impact of superimposed traumas on top of this collective trauma. Many of us have not even rebounded from the pandemic, and then are faced with loss, grief, challenges, illness, hard and big life decisions. We are challenged to answer the question: How do we endure in the face of this trauma inception?
It has been a challenging time for all, including those who are ordinarily happy-go-lucky, resilient, and see the glass half-full and are struggling with the idea of struggling. I am no “resilience expert” but gleaned much wisdom from responding to the Surfside, Fla., building collapse. This was a collective trauma that took place in the summer of 2021, and the wisdom of this event highlighted the value of collective healing and unification even in spite of the times. What happened in Surfside was a shock, and the loss was felt by those directly affected, the surrounding community, and those who were part of the disaster response efforts. All of those parties had been processing losses prior to this – loss of normalcy because of the pandemic, loss of people we loved as a result, other personal losses – and this community tragedy was yet another loss to disentangle on top of a period in U.S. history demarcated by a great lack of unity, divisiveness, anger, and hatred. The collapse highlighted the small size yet interconnectedness of the community and the power of connection and authentic relating. It was overwhelming in the moment but extremely heartening and beautiful to see the amount of willingness to drop everything and help. Despite feeling worn down from the pandemic, people drew upon their internal resources, natural goodness, and kindness “reserves” to provide support.
Responding to the collapse highlighted that resilience in the context of collective trauma requires flexibility, embracing uncertainty, cultivating unity, and paying attention to meeting basic needs/self-care. The role of kindness cannot be overemphasized. In the realm of reflecting on the notion of kindness, it is worth noting how much power there is to bearing witness to someone’s experience, especially when they are in pain. People often diminish the role or at the very least do not recognize the power of showing up for someone and just listening. Pandemic resilience, and coping with coalescing traumas, is likely composed of these same facets that were essential in the context of coping with the collapse.
It is not only the immediate impact of a trauma as much as the aftermath that needs to processed and worked through. In one sense, people feel that they should be adjusted to and accustomed to this new reality, and at the same time, one has to remember and reflect on how unnatural this experience has been. There is an impact of a cumulative onslaught of negative events, and it is hard to imagine not being phased, remaining unchanged, or not feeling affected. We may feel hardened and that there are limits to the compassion we have to offer others. We may be feel empathic. There can be desensitization and an apathy to others’ suffering when our patience is worn down and we have limited bandwidth. There are data to support the idea that a level of habituation occurs to individuals who experience multiple traumas, which yields a level of “sensitization” to the negative impact of subsequent events. It becomes easy to make comparisons of suffering. The challenge will be to rise above these and make a conscious effort to connect with who and how we were before we were worn down.
I am still in awe about how much I learned from the victims’ families, survivors, and my colleagues at Surfside – about pain, suffering, loss, resilience, coping, fortitude, and meaning making. We were all forced to think beyond ourselves, show up for others, and unify in a way that remedied this period of fragmentation. With respect to the pandemic and “where we are at now,” some elements of our lives are stabilizing; other aspects feel volatile from the fatigue of what we have been experiencing. This pandemic has not fully abated, but we can find some clarity in the value of setting boundaries and knowing our limits – but not overlooking the power of unity and kindness and the value of the reciprocating those qualities.
Dr. Feldman is a licensed clinical psychologist in private practice in Miami. She is an adjunct professor in the college of psychology at Nova Southeastern University, Fort Lauderdale, Fla., where she teaches clinical psychology doctoral students. She also serves on the board of directors of the Southeast Florida Association for Psychoanalytic Psychology. Dr. Feldman has no disclosures.
While 2022 is lurking around the corner, many of us still have 2020 on our minds. Social media posts are already emerging: “No new years resolutions. It is the circumstances turn to improve [sic],” one post declares. Others proclaim that it is difficult coming to terms with the idea that 2022 is actually pronounced “2020 too.” A critical difference exists between then and now – we have experienced months of living in limbo and rolling with the punches of pandemic life.
In some ways, it has become easy to think of the early pandemic days as a distant memory, yet respect that the impact of 2020 has been indelible for virtually all of us and feels palpable as if it were yesterday.
The year 2020 was marked by the beginning of the COVID-19 pandemic, which was accompanied by extreme uncertainty, loss of all kinds, and emotional turmoil. The early pandemic had a profound economic and social impact, with added stress tethered to political and race-related division in America that created divides among families and friends, and yielded ceaseless discourse related to divergent perspectives. This only exacerbated the stress that came with the pandemic, given that providing support and leaning on one another was more important than ever. All of this was compounded by natural disasters that have plagued the country.
So much was unprecedented. There was a collective sense of feeling “worn down,” and the burnout that was felt was quite profound. Enormous amounts of mental and physical effort were allocated to simply surviving, getting basic needs met, having enough food and supplies, and completing basic tasks. Ordinary relating felt taxing. At this stage of the pandemic, the COVID-19 experience can be conceived of as a traumatic stressor capable of eliciting a traumatic response and exacerbating other mental health symptoms. Our capacity to cope has been diminished. Anxiety rates have soared, as have rates of clinical depression. Those most affected have had lower household incomes, are unmarried, and have experienced pandemic-related stressors. The links between the impact of the pandemic on mental health have been clear.
The pandemic has forced the landscape of social support to dramatically change. Initially, we felt pulled to connect and we leaned into the use of virtual platforms to connect for all matters (simple social gatherings, big birthday events, family reunions, celebration of holidays, work duties, and academic work). However, “Zoom fatigue” began to set in, and our screen time was maxed out. There has been the added dynamic of frontline workers who did not have the option to work virtually or from home. This group largely has felt disconnected from others who didn’t understand the depth of their anxiety and loneliness of their experience. Health care workers have had to make challenging, life-and-death, patient-related decisions that called into question personal morals and ethics all while their own lives were at risk.
Fast-forward to the present, and support systems have either strengthened or worn down – which has yielded a unique dichotomy. Maintaining friendships has either felt of utmost importance given the impact of the disconnect and physical distance or has felt challenging given the mental energy expended from working and connecting virtually. Empathy burnout is also a real and important facet in the equation. We begin to ask the question: Are we checking in with others in the spirit of authentic relating, to cultivate real connection, or to check a box?
Impact of layered traumas
It is interesting to think about the pandemic’s traumatic impact being “superimposed” on top of the “ordinary traumas” experienced outside of the pandemic. We are essentially at the 2-year mark, in some ways have cultivated a sense of resilience and found ways to adapt, and in other ways at times feel right back where we were in early 2020. There were moments that felt hopeful, glimmers of normalcy, and setbacks that all ebbed and flowed – but even so, there have not been many “mental breaks,” only temporary and transient reprieves. Some got sick and died; some recovered; and others are still experiencing long-hauler syndrome and have lingering sequelae. Despite adaptation and resilience, one can’t help but wonder the impact of superimposed traumas on top of this collective trauma. Many of us have not even rebounded from the pandemic, and then are faced with loss, grief, challenges, illness, hard and big life decisions. We are challenged to answer the question: How do we endure in the face of this trauma inception?
It has been a challenging time for all, including those who are ordinarily happy-go-lucky, resilient, and see the glass half-full and are struggling with the idea of struggling. I am no “resilience expert” but gleaned much wisdom from responding to the Surfside, Fla., building collapse. This was a collective trauma that took place in the summer of 2021, and the wisdom of this event highlighted the value of collective healing and unification even in spite of the times. What happened in Surfside was a shock, and the loss was felt by those directly affected, the surrounding community, and those who were part of the disaster response efforts. All of those parties had been processing losses prior to this – loss of normalcy because of the pandemic, loss of people we loved as a result, other personal losses – and this community tragedy was yet another loss to disentangle on top of a period in U.S. history demarcated by a great lack of unity, divisiveness, anger, and hatred. The collapse highlighted the small size yet interconnectedness of the community and the power of connection and authentic relating. It was overwhelming in the moment but extremely heartening and beautiful to see the amount of willingness to drop everything and help. Despite feeling worn down from the pandemic, people drew upon their internal resources, natural goodness, and kindness “reserves” to provide support.
Responding to the collapse highlighted that resilience in the context of collective trauma requires flexibility, embracing uncertainty, cultivating unity, and paying attention to meeting basic needs/self-care. The role of kindness cannot be overemphasized. In the realm of reflecting on the notion of kindness, it is worth noting how much power there is to bearing witness to someone’s experience, especially when they are in pain. People often diminish the role or at the very least do not recognize the power of showing up for someone and just listening. Pandemic resilience, and coping with coalescing traumas, is likely composed of these same facets that were essential in the context of coping with the collapse.
It is not only the immediate impact of a trauma as much as the aftermath that needs to processed and worked through. In one sense, people feel that they should be adjusted to and accustomed to this new reality, and at the same time, one has to remember and reflect on how unnatural this experience has been. There is an impact of a cumulative onslaught of negative events, and it is hard to imagine not being phased, remaining unchanged, or not feeling affected. We may feel hardened and that there are limits to the compassion we have to offer others. We may be feel empathic. There can be desensitization and an apathy to others’ suffering when our patience is worn down and we have limited bandwidth. There are data to support the idea that a level of habituation occurs to individuals who experience multiple traumas, which yields a level of “sensitization” to the negative impact of subsequent events. It becomes easy to make comparisons of suffering. The challenge will be to rise above these and make a conscious effort to connect with who and how we were before we were worn down.
I am still in awe about how much I learned from the victims’ families, survivors, and my colleagues at Surfside – about pain, suffering, loss, resilience, coping, fortitude, and meaning making. We were all forced to think beyond ourselves, show up for others, and unify in a way that remedied this period of fragmentation. With respect to the pandemic and “where we are at now,” some elements of our lives are stabilizing; other aspects feel volatile from the fatigue of what we have been experiencing. This pandemic has not fully abated, but we can find some clarity in the value of setting boundaries and knowing our limits – but not overlooking the power of unity and kindness and the value of the reciprocating those qualities.
Dr. Feldman is a licensed clinical psychologist in private practice in Miami. She is an adjunct professor in the college of psychology at Nova Southeastern University, Fort Lauderdale, Fla., where she teaches clinical psychology doctoral students. She also serves on the board of directors of the Southeast Florida Association for Psychoanalytic Psychology. Dr. Feldman has no disclosures.
While 2022 is lurking around the corner, many of us still have 2020 on our minds. Social media posts are already emerging: “No new years resolutions. It is the circumstances turn to improve [sic],” one post declares. Others proclaim that it is difficult coming to terms with the idea that 2022 is actually pronounced “2020 too.” A critical difference exists between then and now – we have experienced months of living in limbo and rolling with the punches of pandemic life.
In some ways, it has become easy to think of the early pandemic days as a distant memory, yet respect that the impact of 2020 has been indelible for virtually all of us and feels palpable as if it were yesterday.
The year 2020 was marked by the beginning of the COVID-19 pandemic, which was accompanied by extreme uncertainty, loss of all kinds, and emotional turmoil. The early pandemic had a profound economic and social impact, with added stress tethered to political and race-related division in America that created divides among families and friends, and yielded ceaseless discourse related to divergent perspectives. This only exacerbated the stress that came with the pandemic, given that providing support and leaning on one another was more important than ever. All of this was compounded by natural disasters that have plagued the country.
So much was unprecedented. There was a collective sense of feeling “worn down,” and the burnout that was felt was quite profound. Enormous amounts of mental and physical effort were allocated to simply surviving, getting basic needs met, having enough food and supplies, and completing basic tasks. Ordinary relating felt taxing. At this stage of the pandemic, the COVID-19 experience can be conceived of as a traumatic stressor capable of eliciting a traumatic response and exacerbating other mental health symptoms. Our capacity to cope has been diminished. Anxiety rates have soared, as have rates of clinical depression. Those most affected have had lower household incomes, are unmarried, and have experienced pandemic-related stressors. The links between the impact of the pandemic on mental health have been clear.
The pandemic has forced the landscape of social support to dramatically change. Initially, we felt pulled to connect and we leaned into the use of virtual platforms to connect for all matters (simple social gatherings, big birthday events, family reunions, celebration of holidays, work duties, and academic work). However, “Zoom fatigue” began to set in, and our screen time was maxed out. There has been the added dynamic of frontline workers who did not have the option to work virtually or from home. This group largely has felt disconnected from others who didn’t understand the depth of their anxiety and loneliness of their experience. Health care workers have had to make challenging, life-and-death, patient-related decisions that called into question personal morals and ethics all while their own lives were at risk.
Fast-forward to the present, and support systems have either strengthened or worn down – which has yielded a unique dichotomy. Maintaining friendships has either felt of utmost importance given the impact of the disconnect and physical distance or has felt challenging given the mental energy expended from working and connecting virtually. Empathy burnout is also a real and important facet in the equation. We begin to ask the question: Are we checking in with others in the spirit of authentic relating, to cultivate real connection, or to check a box?
Impact of layered traumas
It is interesting to think about the pandemic’s traumatic impact being “superimposed” on top of the “ordinary traumas” experienced outside of the pandemic. We are essentially at the 2-year mark, in some ways have cultivated a sense of resilience and found ways to adapt, and in other ways at times feel right back where we were in early 2020. There were moments that felt hopeful, glimmers of normalcy, and setbacks that all ebbed and flowed – but even so, there have not been many “mental breaks,” only temporary and transient reprieves. Some got sick and died; some recovered; and others are still experiencing long-hauler syndrome and have lingering sequelae. Despite adaptation and resilience, one can’t help but wonder the impact of superimposed traumas on top of this collective trauma. Many of us have not even rebounded from the pandemic, and then are faced with loss, grief, challenges, illness, hard and big life decisions. We are challenged to answer the question: How do we endure in the face of this trauma inception?
It has been a challenging time for all, including those who are ordinarily happy-go-lucky, resilient, and see the glass half-full and are struggling with the idea of struggling. I am no “resilience expert” but gleaned much wisdom from responding to the Surfside, Fla., building collapse. This was a collective trauma that took place in the summer of 2021, and the wisdom of this event highlighted the value of collective healing and unification even in spite of the times. What happened in Surfside was a shock, and the loss was felt by those directly affected, the surrounding community, and those who were part of the disaster response efforts. All of those parties had been processing losses prior to this – loss of normalcy because of the pandemic, loss of people we loved as a result, other personal losses – and this community tragedy was yet another loss to disentangle on top of a period in U.S. history demarcated by a great lack of unity, divisiveness, anger, and hatred. The collapse highlighted the small size yet interconnectedness of the community and the power of connection and authentic relating. It was overwhelming in the moment but extremely heartening and beautiful to see the amount of willingness to drop everything and help. Despite feeling worn down from the pandemic, people drew upon their internal resources, natural goodness, and kindness “reserves” to provide support.
Responding to the collapse highlighted that resilience in the context of collective trauma requires flexibility, embracing uncertainty, cultivating unity, and paying attention to meeting basic needs/self-care. The role of kindness cannot be overemphasized. In the realm of reflecting on the notion of kindness, it is worth noting how much power there is to bearing witness to someone’s experience, especially when they are in pain. People often diminish the role or at the very least do not recognize the power of showing up for someone and just listening. Pandemic resilience, and coping with coalescing traumas, is likely composed of these same facets that were essential in the context of coping with the collapse.
It is not only the immediate impact of a trauma as much as the aftermath that needs to processed and worked through. In one sense, people feel that they should be adjusted to and accustomed to this new reality, and at the same time, one has to remember and reflect on how unnatural this experience has been. There is an impact of a cumulative onslaught of negative events, and it is hard to imagine not being phased, remaining unchanged, or not feeling affected. We may feel hardened and that there are limits to the compassion we have to offer others. We may be feel empathic. There can be desensitization and an apathy to others’ suffering when our patience is worn down and we have limited bandwidth. There are data to support the idea that a level of habituation occurs to individuals who experience multiple traumas, which yields a level of “sensitization” to the negative impact of subsequent events. It becomes easy to make comparisons of suffering. The challenge will be to rise above these and make a conscious effort to connect with who and how we were before we were worn down.
I am still in awe about how much I learned from the victims’ families, survivors, and my colleagues at Surfside – about pain, suffering, loss, resilience, coping, fortitude, and meaning making. We were all forced to think beyond ourselves, show up for others, and unify in a way that remedied this period of fragmentation. With respect to the pandemic and “where we are at now,” some elements of our lives are stabilizing; other aspects feel volatile from the fatigue of what we have been experiencing. This pandemic has not fully abated, but we can find some clarity in the value of setting boundaries and knowing our limits – but not overlooking the power of unity and kindness and the value of the reciprocating those qualities.
Dr. Feldman is a licensed clinical psychologist in private practice in Miami. She is an adjunct professor in the college of psychology at Nova Southeastern University, Fort Lauderdale, Fla., where she teaches clinical psychology doctoral students. She also serves on the board of directors of the Southeast Florida Association for Psychoanalytic Psychology. Dr. Feldman has no disclosures.
FDA grants new indication to lumateperone (Caplyta) for bipolar depression
The Food and Drug Administration has expanded approval of lumateperone (Caplyta) to include treatment of adults with depressive episodes associated with bipolar I and II disorder, as monotherapy or adjunctive therapy with lithium or valproate.
This makes lumateperone the only FDA-approved drug for this indication.
“The efficacy, and favorable safety and tolerability profile, make Caplyta an important treatment option for the millions of patients living with bipolar I or II depression and represents a major development for these patients,” Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said in a company news release.
Lumateperone was first approved by the FDA in 2019 for the treatment of adults with schizophrenia.
‘Positioned to launch immediately’
that showed treatment with lumateperone, alone or with lithium or valproate, significantly improved depressive symptoms for patients with major depressive episodes associated with bipolar I and bipolar II disorders.
In these studies, treatment with a 42-mg once-daily dose was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale score versus placebo.
Lumateperone also showed a statistically significant improvement in the key secondary endpoint relating to clinical global impression of bipolar disorder.
Somnolence/sedation, dizziness, nausea, and dry mouth were the most commonly reported adverse events associated with the medication. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. Incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
Sharon Mates, PhD, chairman and CEO of Intra-Cellular Therapies, noted in the same press release that the company is “positioned to launch immediately and are excited to offer Caplyta to the millions of patients living with bipolar depression.”
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded approval of lumateperone (Caplyta) to include treatment of adults with depressive episodes associated with bipolar I and II disorder, as monotherapy or adjunctive therapy with lithium or valproate.
This makes lumateperone the only FDA-approved drug for this indication.
“The efficacy, and favorable safety and tolerability profile, make Caplyta an important treatment option for the millions of patients living with bipolar I or II depression and represents a major development for these patients,” Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said in a company news release.
Lumateperone was first approved by the FDA in 2019 for the treatment of adults with schizophrenia.
‘Positioned to launch immediately’
that showed treatment with lumateperone, alone or with lithium or valproate, significantly improved depressive symptoms for patients with major depressive episodes associated with bipolar I and bipolar II disorders.
In these studies, treatment with a 42-mg once-daily dose was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale score versus placebo.
Lumateperone also showed a statistically significant improvement in the key secondary endpoint relating to clinical global impression of bipolar disorder.
Somnolence/sedation, dizziness, nausea, and dry mouth were the most commonly reported adverse events associated with the medication. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. Incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
Sharon Mates, PhD, chairman and CEO of Intra-Cellular Therapies, noted in the same press release that the company is “positioned to launch immediately and are excited to offer Caplyta to the millions of patients living with bipolar depression.”
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded approval of lumateperone (Caplyta) to include treatment of adults with depressive episodes associated with bipolar I and II disorder, as monotherapy or adjunctive therapy with lithium or valproate.
This makes lumateperone the only FDA-approved drug for this indication.
“The efficacy, and favorable safety and tolerability profile, make Caplyta an important treatment option for the millions of patients living with bipolar I or II depression and represents a major development for these patients,” Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said in a company news release.
Lumateperone was first approved by the FDA in 2019 for the treatment of adults with schizophrenia.
‘Positioned to launch immediately’
that showed treatment with lumateperone, alone or with lithium or valproate, significantly improved depressive symptoms for patients with major depressive episodes associated with bipolar I and bipolar II disorders.
In these studies, treatment with a 42-mg once-daily dose was associated with significantly greater improvement from baseline in Montgomery-Åsberg Depression Rating Scale score versus placebo.
Lumateperone also showed a statistically significant improvement in the key secondary endpoint relating to clinical global impression of bipolar disorder.
Somnolence/sedation, dizziness, nausea, and dry mouth were the most commonly reported adverse events associated with the medication. Minimal changes were observed in weight and vital signs and in results of metabolic or endocrine assessments. Incidence of extrapyramidal symptom–related events was low and was similar to those with placebo.
Sharon Mates, PhD, chairman and CEO of Intra-Cellular Therapies, noted in the same press release that the company is “positioned to launch immediately and are excited to offer Caplyta to the millions of patients living with bipolar depression.”
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
Advisory on youth mental health crisis gets mixed reviews
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
A pandemic silver lining? Dramatic drop in teen drug use
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.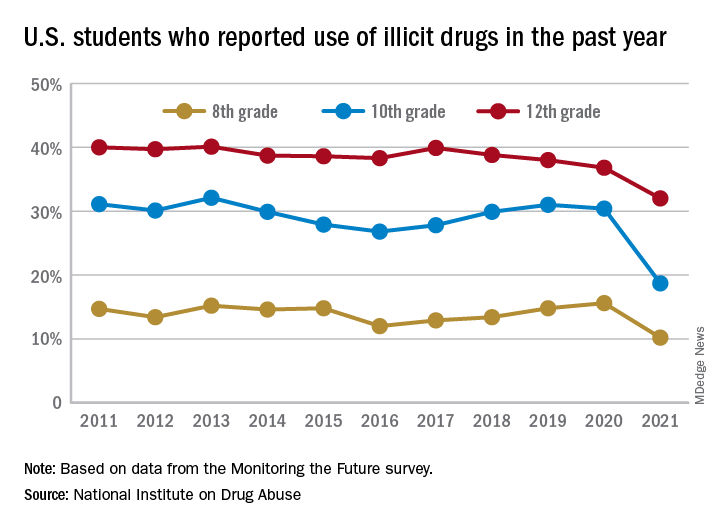
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
More evidence ties some antipsychotics to increased breast cancer risk
New research provides more evidence that antipsychotics that raise prolactin levels are tied to a significantly increased risk for breast cancer.
The relative risk for breast cancer was 62% higher in women who took category 1 antipsychotic medications associated with high prolactin levels. These include haloperidol (Haldol), paliperidone (Invega), and risperidone (Risperdal). Additionally, the risk was 54% higher in those taking category 2 antipsychotics that have mid-range effects on prolactin. These include iloperidone (Fanapt), lurasidone (Latuda), and olanzapine (Zyprexa).
In contrast, category 3 antipsychotics which have a lesser effect on prolactin levels were not associated with any increase in breast cancer risk. These drugs include aripiprazole (Abilify), asenapine (Saphris), brexpiprazole (Rexulti), cariprazine (Vraylar), clozapine (multiple brands), quetiapine (Seroquel), and ziprasidone (Geodon).
While the “absolute” breast cancer risk for these drugs is unclear, “we can make the case that high circulating prolactin levels are associated with breast cancer risk. This follows what is already known about prolactin from prior studies, notably the nurses’ health studies,” Tahir Rahman, MD, associate professor of psychiatry, Washington University School of Medicine, St. Louis, told this news organization.
“We don’t want to alarm patients taking antipsychotic drugs for life-threatening mental health problems, but we also think it is time for doctors to track prolactin levels and vigilantly monitor their patients who are being treated with antipsychotics,” Dr. Rahman added in a news release.
The study was published online Dec. 3 in the Journal of Clinical Psychopharmacology.
Test prolactin levels
Using administrative claims data, the researchers evaluated breast cancer risk in women aged 18-64 exposed to antipsychotic medications compared with anticonvulsants and/or lithium.
They identified 914 cases of invasive breast cancer among 540,737 women.
Roughly 52% of the study population filled at least one prescription for a category 3 antipsychotic agent, whereas 15% filled at least one prescription for a category 1 agent; 49% of women filled at least one prescription for an anticonvulsant medication during the study period.
Exposure to all antipsychotics was independently associated with a 35% increased risk for breast cancer (adjusted hazard ratio, 1.35; 95% CI, 1.14-1.61), the study team found.
Compared with anticonvulsants or lithium, the risk for breast cancer was significantly increased for high prolactin (category 1) antipsychotics (adjusted hazard ratio, 1.62; 95% CI, 1.30-2.03) and for mid-prolactin (category 2) drugs (aHR 1.54; 95% CI, 1.19-1.99), with no increased risk for category 3 antipsychotics.
“Our research is obviously of interest for preventing breast cancer in antipsychotic-treated patients. Checking a blood prolactin level is cheap and easy [and a high level is] fairly simple to mitigate,” said Dr. Rahman.
A matter of debate
Reached for comment, Christoph Correll, MD, professor of psychiatry and molecular medicine, Zucker School of Medicine at Hofstra/Northwell, Hempstead, New York, said, “The potential elevation of breast cancer risk depending on the dose and time of treatment with antipsychotic medications with varying degrees of prolactin-raising properties has been a topic of research and matter of debate.”
This new study “adds another data point indicating that antipsychotics that are associated on average with a higher prolactin-raising effect than other antipsychotics may increase the risk of breast cancer in women to some degree,” said Dr. Correll, who was not involved with the study.
However, he cautioned that “naturalistic data are always vulnerable to residual confounding, for example, unmeasured effects that could also at least partially explain the results, and the follow-up time of only 4 years (maximum 6 years) in this study was relatively short.
“Nevertheless, given availability of many different antipsychotics with varying degrees of prolactin-raising potential, in women requiring antipsychotic treatment, less prolactin-raising antipsychotics may be preferable,” Dr. Correll said.
“In women receiving prolactin-raising antipsychotics for medium- and longer-term maintenance therapy, prolactin levels should be monitored,” he added.
When an elevated prolactin level is detected, this should be addressed “either via dose reduction, a switch to an alternative antipsychotic that does not raise prolactin levels significantly, or the addition of a partial or full D2 agonist when the prolactin-raising antipsychotic should be continued based on individualized risk assessment,” Dr. Correll advised.
This work was supported by an award from the Alvin J. Siteman Cancer Center; the National Cancer Institute and the National Center for Advancing Translational Sciences of the National Institutes of Health; the Taylor Family Institute for Innovative Psychiatric Research; and the Center for Brain Research in Mood Disorders. The authors have disclosed no relevant financial relationships. Dr. Correll has received royalties from UpToDate and is a stock option holder of LB Pharma.
A version of this article first appeared on Medscape.com.
New research provides more evidence that antipsychotics that raise prolactin levels are tied to a significantly increased risk for breast cancer.
The relative risk for breast cancer was 62% higher in women who took category 1 antipsychotic medications associated with high prolactin levels. These include haloperidol (Haldol), paliperidone (Invega), and risperidone (Risperdal). Additionally, the risk was 54% higher in those taking category 2 antipsychotics that have mid-range effects on prolactin. These include iloperidone (Fanapt), lurasidone (Latuda), and olanzapine (Zyprexa).
In contrast, category 3 antipsychotics which have a lesser effect on prolactin levels were not associated with any increase in breast cancer risk. These drugs include aripiprazole (Abilify), asenapine (Saphris), brexpiprazole (Rexulti), cariprazine (Vraylar), clozapine (multiple brands), quetiapine (Seroquel), and ziprasidone (Geodon).
While the “absolute” breast cancer risk for these drugs is unclear, “we can make the case that high circulating prolactin levels are associated with breast cancer risk. This follows what is already known about prolactin from prior studies, notably the nurses’ health studies,” Tahir Rahman, MD, associate professor of psychiatry, Washington University School of Medicine, St. Louis, told this news organization.
“We don’t want to alarm patients taking antipsychotic drugs for life-threatening mental health problems, but we also think it is time for doctors to track prolactin levels and vigilantly monitor their patients who are being treated with antipsychotics,” Dr. Rahman added in a news release.
The study was published online Dec. 3 in the Journal of Clinical Psychopharmacology.
Test prolactin levels
Using administrative claims data, the researchers evaluated breast cancer risk in women aged 18-64 exposed to antipsychotic medications compared with anticonvulsants and/or lithium.
They identified 914 cases of invasive breast cancer among 540,737 women.
Roughly 52% of the study population filled at least one prescription for a category 3 antipsychotic agent, whereas 15% filled at least one prescription for a category 1 agent; 49% of women filled at least one prescription for an anticonvulsant medication during the study period.
Exposure to all antipsychotics was independently associated with a 35% increased risk for breast cancer (adjusted hazard ratio, 1.35; 95% CI, 1.14-1.61), the study team found.
Compared with anticonvulsants or lithium, the risk for breast cancer was significantly increased for high prolactin (category 1) antipsychotics (adjusted hazard ratio, 1.62; 95% CI, 1.30-2.03) and for mid-prolactin (category 2) drugs (aHR 1.54; 95% CI, 1.19-1.99), with no increased risk for category 3 antipsychotics.
“Our research is obviously of interest for preventing breast cancer in antipsychotic-treated patients. Checking a blood prolactin level is cheap and easy [and a high level is] fairly simple to mitigate,” said Dr. Rahman.
A matter of debate
Reached for comment, Christoph Correll, MD, professor of psychiatry and molecular medicine, Zucker School of Medicine at Hofstra/Northwell, Hempstead, New York, said, “The potential elevation of breast cancer risk depending on the dose and time of treatment with antipsychotic medications with varying degrees of prolactin-raising properties has been a topic of research and matter of debate.”
This new study “adds another data point indicating that antipsychotics that are associated on average with a higher prolactin-raising effect than other antipsychotics may increase the risk of breast cancer in women to some degree,” said Dr. Correll, who was not involved with the study.
However, he cautioned that “naturalistic data are always vulnerable to residual confounding, for example, unmeasured effects that could also at least partially explain the results, and the follow-up time of only 4 years (maximum 6 years) in this study was relatively short.
“Nevertheless, given availability of many different antipsychotics with varying degrees of prolactin-raising potential, in women requiring antipsychotic treatment, less prolactin-raising antipsychotics may be preferable,” Dr. Correll said.
“In women receiving prolactin-raising antipsychotics for medium- and longer-term maintenance therapy, prolactin levels should be monitored,” he added.
When an elevated prolactin level is detected, this should be addressed “either via dose reduction, a switch to an alternative antipsychotic that does not raise prolactin levels significantly, or the addition of a partial or full D2 agonist when the prolactin-raising antipsychotic should be continued based on individualized risk assessment,” Dr. Correll advised.
This work was supported by an award from the Alvin J. Siteman Cancer Center; the National Cancer Institute and the National Center for Advancing Translational Sciences of the National Institutes of Health; the Taylor Family Institute for Innovative Psychiatric Research; and the Center for Brain Research in Mood Disorders. The authors have disclosed no relevant financial relationships. Dr. Correll has received royalties from UpToDate and is a stock option holder of LB Pharma.
A version of this article first appeared on Medscape.com.
New research provides more evidence that antipsychotics that raise prolactin levels are tied to a significantly increased risk for breast cancer.
The relative risk for breast cancer was 62% higher in women who took category 1 antipsychotic medications associated with high prolactin levels. These include haloperidol (Haldol), paliperidone (Invega), and risperidone (Risperdal). Additionally, the risk was 54% higher in those taking category 2 antipsychotics that have mid-range effects on prolactin. These include iloperidone (Fanapt), lurasidone (Latuda), and olanzapine (Zyprexa).
In contrast, category 3 antipsychotics which have a lesser effect on prolactin levels were not associated with any increase in breast cancer risk. These drugs include aripiprazole (Abilify), asenapine (Saphris), brexpiprazole (Rexulti), cariprazine (Vraylar), clozapine (multiple brands), quetiapine (Seroquel), and ziprasidone (Geodon).
While the “absolute” breast cancer risk for these drugs is unclear, “we can make the case that high circulating prolactin levels are associated with breast cancer risk. This follows what is already known about prolactin from prior studies, notably the nurses’ health studies,” Tahir Rahman, MD, associate professor of psychiatry, Washington University School of Medicine, St. Louis, told this news organization.
“We don’t want to alarm patients taking antipsychotic drugs for life-threatening mental health problems, but we also think it is time for doctors to track prolactin levels and vigilantly monitor their patients who are being treated with antipsychotics,” Dr. Rahman added in a news release.
The study was published online Dec. 3 in the Journal of Clinical Psychopharmacology.
Test prolactin levels
Using administrative claims data, the researchers evaluated breast cancer risk in women aged 18-64 exposed to antipsychotic medications compared with anticonvulsants and/or lithium.
They identified 914 cases of invasive breast cancer among 540,737 women.
Roughly 52% of the study population filled at least one prescription for a category 3 antipsychotic agent, whereas 15% filled at least one prescription for a category 1 agent; 49% of women filled at least one prescription for an anticonvulsant medication during the study period.
Exposure to all antipsychotics was independently associated with a 35% increased risk for breast cancer (adjusted hazard ratio, 1.35; 95% CI, 1.14-1.61), the study team found.
Compared with anticonvulsants or lithium, the risk for breast cancer was significantly increased for high prolactin (category 1) antipsychotics (adjusted hazard ratio, 1.62; 95% CI, 1.30-2.03) and for mid-prolactin (category 2) drugs (aHR 1.54; 95% CI, 1.19-1.99), with no increased risk for category 3 antipsychotics.
“Our research is obviously of interest for preventing breast cancer in antipsychotic-treated patients. Checking a blood prolactin level is cheap and easy [and a high level is] fairly simple to mitigate,” said Dr. Rahman.
A matter of debate
Reached for comment, Christoph Correll, MD, professor of psychiatry and molecular medicine, Zucker School of Medicine at Hofstra/Northwell, Hempstead, New York, said, “The potential elevation of breast cancer risk depending on the dose and time of treatment with antipsychotic medications with varying degrees of prolactin-raising properties has been a topic of research and matter of debate.”
This new study “adds another data point indicating that antipsychotics that are associated on average with a higher prolactin-raising effect than other antipsychotics may increase the risk of breast cancer in women to some degree,” said Dr. Correll, who was not involved with the study.
However, he cautioned that “naturalistic data are always vulnerable to residual confounding, for example, unmeasured effects that could also at least partially explain the results, and the follow-up time of only 4 years (maximum 6 years) in this study was relatively short.
“Nevertheless, given availability of many different antipsychotics with varying degrees of prolactin-raising potential, in women requiring antipsychotic treatment, less prolactin-raising antipsychotics may be preferable,” Dr. Correll said.
“In women receiving prolactin-raising antipsychotics for medium- and longer-term maintenance therapy, prolactin levels should be monitored,” he added.
When an elevated prolactin level is detected, this should be addressed “either via dose reduction, a switch to an alternative antipsychotic that does not raise prolactin levels significantly, or the addition of a partial or full D2 agonist when the prolactin-raising antipsychotic should be continued based on individualized risk assessment,” Dr. Correll advised.
This work was supported by an award from the Alvin J. Siteman Cancer Center; the National Cancer Institute and the National Center for Advancing Translational Sciences of the National Institutes of Health; the Taylor Family Institute for Innovative Psychiatric Research; and the Center for Brain Research in Mood Disorders. The authors have disclosed no relevant financial relationships. Dr. Correll has received royalties from UpToDate and is a stock option holder of LB Pharma.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHOPHARMACOLOGY
Assessing Outcomes Between Risperidone Microspheres and Paliperidone Palmitate Long-Acting Injectable Antipsychotics Among Veterans
Medication nonadherence is common with oral antipsychotic formulations, resulting in relapse, increased morbidity, and more frequent psychiatric hospitalization.1-7 Psychiatric hospitalization and illness decompensation is costly to health care systems and leads to reduced quality of life for veterans and families.6,7 Long-acting injectable antipsychotics (LAIAs) were developed to enhance antipsychotic adherence and improve patient outcomes, including reduced psychiatric hospitalization.8-12
Little outcomes data exist comparing LAIAs, including biweekly risperidone microspheres and monthly paliperidone palmitate.10-13 Risperidone microspheres require a 3-week oral crossover and are administered every 2 weeks, whereas paliperidone palmitate does not require an oral crossover and is administered every 4 weeks. The paliperidone palmitate loading regimen replaces an oral crossover.
The primary objective of this study was to compare the number of psychiatric hospitalizations between veterans administered risperidone microspheres and those on paliperidone palmitate pre- and post-LAIA initiation. Secondary objectives were to assess rehospitalization rates between patients taking risperidone microspheres and paliperidone palmitate, reduction in pre- and posthospitalization rates with LAIAs, and medication adherence.
Methods
This observational study with a retrospective cohort design was conducted at the Veterans Affairs Loma Linda Healthcare System (VALLHS) in California. We examined veterans who were initiated on LAIAs risperidone microspheres or paliperidone palmitate from January 01, 2016 through December 31, 2018. Veterans who were aged ≥ 18 years and received ≥ 2 injections of either risperidone microspheres or paliperidone palmitate during the study period were included. Veterans were excluded if they had received < 2 doses of either LAIA, received the LAIA outside of the review period, were nonadherent to risperidone crossover if they received risperidone microspheres, or transferred their care to another facility. At VALLHS, LAIA injections are administered by a nurse, and veterans must travel to the facility to receive the injections.
Extracted patient chart elements included participant demographics; diagnoses; comorbid alcohol, nicotine, opioid, or other substance use; duration on LAIA; psychiatric hospitalizations pre- and postinitiation of the LAIA; medication adherence; and medication discontinuation based on clinician documentation and clinic orders (Table 1).
Nonadherence to LAIA was defined as missing an injection by > 3 days for risperidone microspheres and > 7 days for paliperidone palmitate. This time frame was based on pharmacokinetic information listed in the products’ package inserts.14,15 Nonadherence to oral risperidone crossover with risperidone microspheres was defined as ≤ 80% of days covered.
Data Analysis
Patient demographics were analyzed using descriptive statistics and experimental comparisons between the risperidone microspheres and paliperidone palmitate groups to assess baseline differences between groups. Psychiatric hospitalizations pre- and post-LAIA were analyzed with parallel group (between veterans–independent groups) and pre-post (within veterans–dependent groups) designs. Index hospitalizations were examined for a period equivalent to the length of time veterans were on the LAIA. Psychiatric rehospitalization rates were analyzed for patients who had index hospitalizations and were rehospitalized for any period when they were receiving the LAIA. Incidences of pre- and post-LAIA hospitalizations were calculated in 100 person-years.
Parallel-group analysis was analyzed using the χ2 and Mann-Whitney U tests. Pre-post analyses were analyzed using the Wilcoxon rank sum test. P was set at < .05 for statistical significance.
Results
We screened 111 veterans, and 97 were included in this study (risperidone microspheres, 44; paliperidone palmitate, 53). Mean (SD) age was 46 (13.8) years, 92% were male, 38% were White, 94% were diagnosed with schizophrenia or schizoaffective disorder, and 11% were homeless. Substance use was documented as 52% for nicotine products, 40% for alcohol, 31% for cannabis, 27% for methamphetamine, 7% for cocaine, and 3% for opioids. Cannabis, methamphetamine, cocaine, and opioid use were based on clinician documentation and listed as active diagnoses at the time of LAIA initiation. Statistical significance was found in index hospitalizations P = .009) and history of cocaine use disorder (6.8% vs 7.5%, P < .001).
Veterans administered risperidone microspheres had fewer mean (SD) post-LAIA hospitalizations (0.4 [1.0] vs 0.9 [1.5]; P = .02) and were less likely to be rehospitalized (22.7% vs 47.2%, P = .01) compared with paliperidone palmitate. However, veterans taking risperidone microspheres had a shorter mean (SD) treatment duration (41.6 [40.2] vs 58.2 [45.7] weeks, P = .04) compared with paliperidone palmitate, mainly because patients switched to a different LAIA or oral antipsychotic. No differences were detected in nonadherence and discontinuation between risperidone microspheres and paliperidone palmitate. All veterans in the risperidone microspheres group adhered to oral risperidone crossover with an average 87.8% days covered (Table 2).
The average maintenance dose of risperidone microspheres was 42 mg every 2 weeks and 153 mg every 4 weeks for paliperidone palmitate.
Across the sample, 84% of veterans had a previous psychiatric hospitalization, although veterans initiated on risperidone microspheres had significantly higher mean (SD) index hospitalizations than those started on paliperidone palmitate (3.2 [2.6] risperidone microspheres vs 2.1 [1.9] paliperidone palmitate, P = .009). Both groups had significant decreases in mean (SD) hospitalizations (3.2 [2.6] to 0.4 [1.0], risperidone microspheres vs 2.1 [1.9] to 0.9 [1.5] paliperidone palmitate). The risperidone microspheres group had a larger decrease in mean (SD) hospitalizations post-LAIA (2.8 [2.9] risperidone microspheres vs 1.3 [1.7] paliperidone palmitate, P = .001) (Table 3).
Differences in incidence per 100 person-years between pre- and post-LAIA hospitalizations were larger in risperidone microspheres users than in paliperidone palmitate (73.8 vs 33.7, P = .01) (Figure). No differences between risperidone microspheres and paliperidone palmitate were detected when looking at incidence pre-LAIA (102.2 vs 75.8, P = .22) and post-LAIA (28.4 vs 42.1, P = .38) separately.
Thirty veterans in the risperidone microspheres group discontinued LAIA: 11 were nonadherent, 5 experienced adverse effects (AEs), and 14 discontinued due to inconvenience. Among 33 veterans in the paliperidone palmitate group who discontinued the LAIA, 15 were nonadherent, 11 experienced AEs, 4 stopped due to of inconvenience, and 3 switched to a less frequently administered LAIA. The most common AEs reported were injection site reactions, cholinergic AEs (salivation, lacrimation, urination), orthostasis, and weight gain.
Discussion
The main finding of this study was that initiation of LAIAs significantly reduced hospitalizations. Veterans taking risperidone microspheres had higher index hospitalizations and lower posttreatment hospitalizations compared with paliperidone palmitate. We found that patients initiated on risperidone microspheres had more hospitalizations before use of a LAIA than those initiated on paliperidone palmitate. Risperidone microspheres reduced the number of hospitalization post-LAIA significantly more than paliperidone palmitate. We also found that veterans taking risperidone microspheres were on the medication for less mean (SD) time than those on paliperidone palmitate (41.6 [40.2] vs 58.2 [45.7] weeks; P = .04).
To our knowledge, this is one of the few studies that compared outcomes of psychiatric hospitalizations, medication adherence, and treatment discontinuation between risperidone microspheres and paliperidone palmitate, specifically in a veteran population.16-19 Limosin and colleagues aimed to compare length of stay during the initial hospitalization, rehospitalization risk, and treatment duration between risperidone microspheres and paliperidone palmitate in patients with schizophrenia.16 These researchers detected no differences in initial hospitalization duration and time to rehospitalization between risperidone microspheres and paliperidone palmitate.16 The study revealed a more favorable trend in time to discontinuation for paliperidone palmitate, but switching between LAIAs might have confounded the data.16 The authors note that their study lacked power, and patients on paliperidone palmitate had significantly more nonpsychiatric comorbidities.16 Joshi and colleagues looked at adherence, medication discontinuation, hospitalization rates, emergency department visits, and hospitalization costs between risperidone microspheres and paliperidone palmitate in patients identified in Truven MarketScan Commercial, Medicare Supplemental, and Medicaid Multi-State insurance databases.17 The authors found paliperidone palmitate to be superior in all objectives with better adherence, lower discontinuation rates, less likelihood of hospitalization, fewer emergency department visits, and lower hospitalization costs compared with risperidone microspheres.17 Korell and colleagues aimed to establish reference ranges for plasma concentrations of risperidone and paliperidone among adherent patients.18
The researchers established reference ranges for risperidone and paliperidone plasma concentrations that represented expected variability within a population and were derived from population pharmacokinetic models.18 Gopal and colleagues conducted a post hoc comparison between paliperidone palmitate and oral risperidone during initiation of long-acting injectable risperidone in patients with acute schizophrenia.19 The researchers found that during the first month after initiating long-acting injectable risperidone, paliperidone palmitate without oral supplementation had similar efficacy and safety to oral risperidone among these patients.19
LAIAs can create a steadier drug plasma concentration compared with oral antipsychotics and do not need to be taken daily. These agents improve adherence by reducing the frequency of medication administrations.20-24 Assessing nonadherence is easier with LAIAs by counting missed injections compared with oral antipsychotics that require calculation of percentage of days covered.25
The results in our study are somewhat unexpected in part because of the close relationship between risperidone and paliperidone. Risperidone is converted to paliperidone (9-OH-risperidone) via hepatic cytochrome P450 2D6. Although the molecules do not have identical pharmacologic profiles, it is accepted that they are similar enough that risperidone can establish oral tolerability when transitioning therapy to paliperidone palmitate and vice versa.24 Although the active moiety in risperidone microspheres and paliperidone palmitate is similar, the dosing interval for risperidone microspheres is 2 weeks compared with 4 weeks with paliperidone palmitate. One potential explanation as to why veterans started on risperidone microspheres experienced better outcomes is because they had twice as many office visits with the health care team. Facility procedures dictate veterans receive the LAIA at an on-site clinic. During the visits, a licensed vocational nurse administers the injection and monitors the patient for 15 to 30 minutes afterward.
Despite new LAIAs coming to market, high-quality data examining potential differences in treatment outcomes among agents are limited. This is problematic for clinicians who want to optimize care by understanding how administration schedules or other aspects of LAIA use could modify treatment outcomes. Our results suggest that an advantage might exist in selecting an agent with a more frequent administration schedule, at least initially. This could allow for close monitoring and regular therapeutic contact, which could improve short-term outcomes. This conclusion is supported by meta-analyses, randomized controlled trials, and conceptual articles conducted by Wehring and colleagues, Berwaerts and colleagues, and Parellada and colleagues, respectively, who examined patients on different LAIAs and contact with health care professionals as part of their research.26-28 These researchers concluded that patients who had regular contact with a health care professional had better outcomes when initiated on a LAIA.26-28
Limitations
There are several limitations in this study. Retrospective and observational methods introduce risks of bias and confounding variables. Sample size might have limited statistical power to detect differences. Veterans might have had undocumented pre- or posthospitalizations at other institutions, which was not accounted for and lack of rehospitalization is not conclusive of a positive outcome. Institutions could improve on our study and help to fill gaps in comparative data by conducting larger analyses over longer periods and including more LAIA agents.
Conclusions
Although veterans that were administered risperidone microspheres had a shorter treatment duration, they were less likely to be rehospitalized, had a fewer mean number of post-LAIA hospitalizations, and had a larger difference in incidence in 100 person-years compared with veterans on paliperidone palmitate. Nonadherence and discontinuation rates were comparable between risperidone microspheres and paliperidone palmitate. Future studies could aim to further clarify differences in outcomes among agents or administration schedules.
1. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
2. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223. doi:10.1056/NEJMoa051688
3. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500
4. Haywood TW, Kravitz HM, Grossman LS, Cavanaugh JL Jr, Davis JM, Lewis DA. Predicting the “revolving door” phenomenon among patients with schizophrenic, schizoaffective, and affective disorders. Am J Psychiatry. 1995;152(6):856-561. doi:10.1176/ajp.152.6.856
5. Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8:32. doi:10.1186/1471-244X-8-32
6. Weiden PJ, Kozma C, Grogg A, Locklear J. Partial compliance and risk of rehospitalization among California Medicaid patients with schizophrenia. Psychiatr Serv. 2004;55(8):886-891. doi:10.1176/appi.ps.55.8.886
7. Gilmer TP, Dolder CR, Lacro JP, et al. Adherence to treatment with antipsychotic medication and health care costs among Medicaid beneficiaries with schizophrenia. Am J Psychiatry. 2004;161(4):692-699. doi:10.1176/appi.ajp.161.4.692
8. Lafeuille MH, Dean J, Carter V, et al. Systematic review of long-acting injectables versus oral atypical antipsychotics on hospitalization in schizophrenia. Curr Med Res Opin. 2014;30(8):1643-1655. doi:10.1185/03007995.2014.915211
9. Yu W, Wagner TH, Chen S, Barnett PG. Average cost of VA rehabilitation, mental health, and long-term hospital stays. Med Care Res Rev. 2003;60(3 suppl):40S-53S. doi:10.1177/1077558703256724
10. Duncan EJ, Woolson SL, Hamer RM. Treatment compliance in veterans administration schizophrenia spectrum patients treated with risperidone long-acting injectable. Int Clin Psychopharmacol. 2012;27(5):283-290. doi:10.1097/YIC.0b013e328354b534
11. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
12. Dimitropoulos E, Drogemuller L, Wong K. Evaluation of concurrent oral and long-acting injectable antipsychotic prescribing at the Minneapolis Veterans Affairs Health Care System. J Clin Psychopharmacol. 2017;37(5):605-608. doi:10.1097/JCP.0000000000000755
13. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
14. Risperdal Consta. Package insert. Janssen Pharmaceutical; 2007.
15. Invega Sustenna. Package insert. Janssen Pharmaceutical; 2009.
16. Limosin F, Belhadi D, Comet D, et al. Comparison of paliperidone palmitate and risperidone long-acting injection in schizophrenic patients: results from a multicenter retrospective cohort study in France. J Clin Psychopharmacol. 2018;38(1):19-26. doi:10.1097/JCP.0000000000000827
17. Joshi K, Pan X, Wang R, Yang E, Benson C. Healthcare resource utilization of second-generation long-acting injectable antipsychotics in schizophrenia: risperidone versus paliperidone palmitate. Curr Med Res Opin. 2016;32(11):1873-1881. doi: 10.1080/03007995.2016.1219706
18. Korell J, Green B, Remmerie B, Vermeulen A. Determination of plasma concentration reference ranges for risperidone and paliperidone. CPT Pharmacometrics Syst Pharmacol. 2017;6(9):589-595. doi:10.1002/psp4.12217
19. Gopal S, Pandina G, Lane R, et al. A post-hoc comparison of paliperidone palmitate to oral risperidone during initiation of long-acting risperidone injection in patients with acute schizophrenia. Innov Clin Neurosci. 2011;8(8):26-33.
20. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
21. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
22. Green AI, Brunette MF, Dawson R, et al. Long-acting injectable vs oral risperidone for schizophrenia and co-occurring alcohol use disorder: a randomized trial. J Clin Psychiatry. 2015;76(10):1359-1365. doi:10.4088/JCP.13m08838
23. Rezansoff SN, Moniruzzaman A, Fazel S, Procyshyn R, Somers JM. Adherence to antipsychotic medication among homeless adults in Vancouver, Canada: a 15-year retrospective cohort study. Soc Psychiatry Psychiatr Epidemiol. 2016;51(12):1623-1632. doi:10.1007/s00127-016-1259-7
24. Castillo EG, Stroup TS. Effectiveness of long-acting injectable antipsychotics: a clinical perspective. Evid Based Ment Health. 2015;18(2):36-39. doi:10.1136/eb-2015-102086
25. Marder SR. Overview of partial compliance. J Clin Psychiatry. 2003;64 (suppl 16):3-9.
26. Wehring HJ, Thedford S, Koola M, Kelly DL. Patient and health care provider perspectives on long acting injectable antipsychotics in schizophrenia and the introduction of olanzapine long-acting injection. J Cent Nerv Syst Dis. 2011;2011(3):107-123. doi:10.4137/JCNSD.S4091
27. Berwaerts J, Liu Y, Gopal S, et al. Efficacy and safety of the 3-month formulation of paliperidone palmitate vs placebo for relapse prevention of schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2015;72(8):830-839. doi:10.1001/jamapsychiatry.2015.0241
28. Parellada E, Bioque M. Barriers to the use of long-acting injectable antipsychotics in the management of schizophrenia. CNS Drugs. 2016;30(8):689-701. doi:10.1007/s40263-016-0350-7
Medication nonadherence is common with oral antipsychotic formulations, resulting in relapse, increased morbidity, and more frequent psychiatric hospitalization.1-7 Psychiatric hospitalization and illness decompensation is costly to health care systems and leads to reduced quality of life for veterans and families.6,7 Long-acting injectable antipsychotics (LAIAs) were developed to enhance antipsychotic adherence and improve patient outcomes, including reduced psychiatric hospitalization.8-12
Little outcomes data exist comparing LAIAs, including biweekly risperidone microspheres and monthly paliperidone palmitate.10-13 Risperidone microspheres require a 3-week oral crossover and are administered every 2 weeks, whereas paliperidone palmitate does not require an oral crossover and is administered every 4 weeks. The paliperidone palmitate loading regimen replaces an oral crossover.
The primary objective of this study was to compare the number of psychiatric hospitalizations between veterans administered risperidone microspheres and those on paliperidone palmitate pre- and post-LAIA initiation. Secondary objectives were to assess rehospitalization rates between patients taking risperidone microspheres and paliperidone palmitate, reduction in pre- and posthospitalization rates with LAIAs, and medication adherence.
Methods
This observational study with a retrospective cohort design was conducted at the Veterans Affairs Loma Linda Healthcare System (VALLHS) in California. We examined veterans who were initiated on LAIAs risperidone microspheres or paliperidone palmitate from January 01, 2016 through December 31, 2018. Veterans who were aged ≥ 18 years and received ≥ 2 injections of either risperidone microspheres or paliperidone palmitate during the study period were included. Veterans were excluded if they had received < 2 doses of either LAIA, received the LAIA outside of the review period, were nonadherent to risperidone crossover if they received risperidone microspheres, or transferred their care to another facility. At VALLHS, LAIA injections are administered by a nurse, and veterans must travel to the facility to receive the injections.
Extracted patient chart elements included participant demographics; diagnoses; comorbid alcohol, nicotine, opioid, or other substance use; duration on LAIA; psychiatric hospitalizations pre- and postinitiation of the LAIA; medication adherence; and medication discontinuation based on clinician documentation and clinic orders (Table 1).
Nonadherence to LAIA was defined as missing an injection by > 3 days for risperidone microspheres and > 7 days for paliperidone palmitate. This time frame was based on pharmacokinetic information listed in the products’ package inserts.14,15 Nonadherence to oral risperidone crossover with risperidone microspheres was defined as ≤ 80% of days covered.
Data Analysis
Patient demographics were analyzed using descriptive statistics and experimental comparisons between the risperidone microspheres and paliperidone palmitate groups to assess baseline differences between groups. Psychiatric hospitalizations pre- and post-LAIA were analyzed with parallel group (between veterans–independent groups) and pre-post (within veterans–dependent groups) designs. Index hospitalizations were examined for a period equivalent to the length of time veterans were on the LAIA. Psychiatric rehospitalization rates were analyzed for patients who had index hospitalizations and were rehospitalized for any period when they were receiving the LAIA. Incidences of pre- and post-LAIA hospitalizations were calculated in 100 person-years.
Parallel-group analysis was analyzed using the χ2 and Mann-Whitney U tests. Pre-post analyses were analyzed using the Wilcoxon rank sum test. P was set at < .05 for statistical significance.
Results
We screened 111 veterans, and 97 were included in this study (risperidone microspheres, 44; paliperidone palmitate, 53). Mean (SD) age was 46 (13.8) years, 92% were male, 38% were White, 94% were diagnosed with schizophrenia or schizoaffective disorder, and 11% were homeless. Substance use was documented as 52% for nicotine products, 40% for alcohol, 31% for cannabis, 27% for methamphetamine, 7% for cocaine, and 3% for opioids. Cannabis, methamphetamine, cocaine, and opioid use were based on clinician documentation and listed as active diagnoses at the time of LAIA initiation. Statistical significance was found in index hospitalizations P = .009) and history of cocaine use disorder (6.8% vs 7.5%, P < .001).
Veterans administered risperidone microspheres had fewer mean (SD) post-LAIA hospitalizations (0.4 [1.0] vs 0.9 [1.5]; P = .02) and were less likely to be rehospitalized (22.7% vs 47.2%, P = .01) compared with paliperidone palmitate. However, veterans taking risperidone microspheres had a shorter mean (SD) treatment duration (41.6 [40.2] vs 58.2 [45.7] weeks, P = .04) compared with paliperidone palmitate, mainly because patients switched to a different LAIA or oral antipsychotic. No differences were detected in nonadherence and discontinuation between risperidone microspheres and paliperidone palmitate. All veterans in the risperidone microspheres group adhered to oral risperidone crossover with an average 87.8% days covered (Table 2).
The average maintenance dose of risperidone microspheres was 42 mg every 2 weeks and 153 mg every 4 weeks for paliperidone palmitate.
Across the sample, 84% of veterans had a previous psychiatric hospitalization, although veterans initiated on risperidone microspheres had significantly higher mean (SD) index hospitalizations than those started on paliperidone palmitate (3.2 [2.6] risperidone microspheres vs 2.1 [1.9] paliperidone palmitate, P = .009). Both groups had significant decreases in mean (SD) hospitalizations (3.2 [2.6] to 0.4 [1.0], risperidone microspheres vs 2.1 [1.9] to 0.9 [1.5] paliperidone palmitate). The risperidone microspheres group had a larger decrease in mean (SD) hospitalizations post-LAIA (2.8 [2.9] risperidone microspheres vs 1.3 [1.7] paliperidone palmitate, P = .001) (Table 3).
Differences in incidence per 100 person-years between pre- and post-LAIA hospitalizations were larger in risperidone microspheres users than in paliperidone palmitate (73.8 vs 33.7, P = .01) (Figure). No differences between risperidone microspheres and paliperidone palmitate were detected when looking at incidence pre-LAIA (102.2 vs 75.8, P = .22) and post-LAIA (28.4 vs 42.1, P = .38) separately.
Thirty veterans in the risperidone microspheres group discontinued LAIA: 11 were nonadherent, 5 experienced adverse effects (AEs), and 14 discontinued due to inconvenience. Among 33 veterans in the paliperidone palmitate group who discontinued the LAIA, 15 were nonadherent, 11 experienced AEs, 4 stopped due to of inconvenience, and 3 switched to a less frequently administered LAIA. The most common AEs reported were injection site reactions, cholinergic AEs (salivation, lacrimation, urination), orthostasis, and weight gain.
Discussion
The main finding of this study was that initiation of LAIAs significantly reduced hospitalizations. Veterans taking risperidone microspheres had higher index hospitalizations and lower posttreatment hospitalizations compared with paliperidone palmitate. We found that patients initiated on risperidone microspheres had more hospitalizations before use of a LAIA than those initiated on paliperidone palmitate. Risperidone microspheres reduced the number of hospitalization post-LAIA significantly more than paliperidone palmitate. We also found that veterans taking risperidone microspheres were on the medication for less mean (SD) time than those on paliperidone palmitate (41.6 [40.2] vs 58.2 [45.7] weeks; P = .04).
To our knowledge, this is one of the few studies that compared outcomes of psychiatric hospitalizations, medication adherence, and treatment discontinuation between risperidone microspheres and paliperidone palmitate, specifically in a veteran population.16-19 Limosin and colleagues aimed to compare length of stay during the initial hospitalization, rehospitalization risk, and treatment duration between risperidone microspheres and paliperidone palmitate in patients with schizophrenia.16 These researchers detected no differences in initial hospitalization duration and time to rehospitalization between risperidone microspheres and paliperidone palmitate.16 The study revealed a more favorable trend in time to discontinuation for paliperidone palmitate, but switching between LAIAs might have confounded the data.16 The authors note that their study lacked power, and patients on paliperidone palmitate had significantly more nonpsychiatric comorbidities.16 Joshi and colleagues looked at adherence, medication discontinuation, hospitalization rates, emergency department visits, and hospitalization costs between risperidone microspheres and paliperidone palmitate in patients identified in Truven MarketScan Commercial, Medicare Supplemental, and Medicaid Multi-State insurance databases.17 The authors found paliperidone palmitate to be superior in all objectives with better adherence, lower discontinuation rates, less likelihood of hospitalization, fewer emergency department visits, and lower hospitalization costs compared with risperidone microspheres.17 Korell and colleagues aimed to establish reference ranges for plasma concentrations of risperidone and paliperidone among adherent patients.18
The researchers established reference ranges for risperidone and paliperidone plasma concentrations that represented expected variability within a population and were derived from population pharmacokinetic models.18 Gopal and colleagues conducted a post hoc comparison between paliperidone palmitate and oral risperidone during initiation of long-acting injectable risperidone in patients with acute schizophrenia.19 The researchers found that during the first month after initiating long-acting injectable risperidone, paliperidone palmitate without oral supplementation had similar efficacy and safety to oral risperidone among these patients.19
LAIAs can create a steadier drug plasma concentration compared with oral antipsychotics and do not need to be taken daily. These agents improve adherence by reducing the frequency of medication administrations.20-24 Assessing nonadherence is easier with LAIAs by counting missed injections compared with oral antipsychotics that require calculation of percentage of days covered.25
The results in our study are somewhat unexpected in part because of the close relationship between risperidone and paliperidone. Risperidone is converted to paliperidone (9-OH-risperidone) via hepatic cytochrome P450 2D6. Although the molecules do not have identical pharmacologic profiles, it is accepted that they are similar enough that risperidone can establish oral tolerability when transitioning therapy to paliperidone palmitate and vice versa.24 Although the active moiety in risperidone microspheres and paliperidone palmitate is similar, the dosing interval for risperidone microspheres is 2 weeks compared with 4 weeks with paliperidone palmitate. One potential explanation as to why veterans started on risperidone microspheres experienced better outcomes is because they had twice as many office visits with the health care team. Facility procedures dictate veterans receive the LAIA at an on-site clinic. During the visits, a licensed vocational nurse administers the injection and monitors the patient for 15 to 30 minutes afterward.
Despite new LAIAs coming to market, high-quality data examining potential differences in treatment outcomes among agents are limited. This is problematic for clinicians who want to optimize care by understanding how administration schedules or other aspects of LAIA use could modify treatment outcomes. Our results suggest that an advantage might exist in selecting an agent with a more frequent administration schedule, at least initially. This could allow for close monitoring and regular therapeutic contact, which could improve short-term outcomes. This conclusion is supported by meta-analyses, randomized controlled trials, and conceptual articles conducted by Wehring and colleagues, Berwaerts and colleagues, and Parellada and colleagues, respectively, who examined patients on different LAIAs and contact with health care professionals as part of their research.26-28 These researchers concluded that patients who had regular contact with a health care professional had better outcomes when initiated on a LAIA.26-28
Limitations
There are several limitations in this study. Retrospective and observational methods introduce risks of bias and confounding variables. Sample size might have limited statistical power to detect differences. Veterans might have had undocumented pre- or posthospitalizations at other institutions, which was not accounted for and lack of rehospitalization is not conclusive of a positive outcome. Institutions could improve on our study and help to fill gaps in comparative data by conducting larger analyses over longer periods and including more LAIA agents.
Conclusions
Although veterans that were administered risperidone microspheres had a shorter treatment duration, they were less likely to be rehospitalized, had a fewer mean number of post-LAIA hospitalizations, and had a larger difference in incidence in 100 person-years compared with veterans on paliperidone palmitate. Nonadherence and discontinuation rates were comparable between risperidone microspheres and paliperidone palmitate. Future studies could aim to further clarify differences in outcomes among agents or administration schedules.
Medication nonadherence is common with oral antipsychotic formulations, resulting in relapse, increased morbidity, and more frequent psychiatric hospitalization.1-7 Psychiatric hospitalization and illness decompensation is costly to health care systems and leads to reduced quality of life for veterans and families.6,7 Long-acting injectable antipsychotics (LAIAs) were developed to enhance antipsychotic adherence and improve patient outcomes, including reduced psychiatric hospitalization.8-12
Little outcomes data exist comparing LAIAs, including biweekly risperidone microspheres and monthly paliperidone palmitate.10-13 Risperidone microspheres require a 3-week oral crossover and are administered every 2 weeks, whereas paliperidone palmitate does not require an oral crossover and is administered every 4 weeks. The paliperidone palmitate loading regimen replaces an oral crossover.
The primary objective of this study was to compare the number of psychiatric hospitalizations between veterans administered risperidone microspheres and those on paliperidone palmitate pre- and post-LAIA initiation. Secondary objectives were to assess rehospitalization rates between patients taking risperidone microspheres and paliperidone palmitate, reduction in pre- and posthospitalization rates with LAIAs, and medication adherence.
Methods
This observational study with a retrospective cohort design was conducted at the Veterans Affairs Loma Linda Healthcare System (VALLHS) in California. We examined veterans who were initiated on LAIAs risperidone microspheres or paliperidone palmitate from January 01, 2016 through December 31, 2018. Veterans who were aged ≥ 18 years and received ≥ 2 injections of either risperidone microspheres or paliperidone palmitate during the study period were included. Veterans were excluded if they had received < 2 doses of either LAIA, received the LAIA outside of the review period, were nonadherent to risperidone crossover if they received risperidone microspheres, or transferred their care to another facility. At VALLHS, LAIA injections are administered by a nurse, and veterans must travel to the facility to receive the injections.
Extracted patient chart elements included participant demographics; diagnoses; comorbid alcohol, nicotine, opioid, or other substance use; duration on LAIA; psychiatric hospitalizations pre- and postinitiation of the LAIA; medication adherence; and medication discontinuation based on clinician documentation and clinic orders (Table 1).
Nonadherence to LAIA was defined as missing an injection by > 3 days for risperidone microspheres and > 7 days for paliperidone palmitate. This time frame was based on pharmacokinetic information listed in the products’ package inserts.14,15 Nonadherence to oral risperidone crossover with risperidone microspheres was defined as ≤ 80% of days covered.
Data Analysis
Patient demographics were analyzed using descriptive statistics and experimental comparisons between the risperidone microspheres and paliperidone palmitate groups to assess baseline differences between groups. Psychiatric hospitalizations pre- and post-LAIA were analyzed with parallel group (between veterans–independent groups) and pre-post (within veterans–dependent groups) designs. Index hospitalizations were examined for a period equivalent to the length of time veterans were on the LAIA. Psychiatric rehospitalization rates were analyzed for patients who had index hospitalizations and were rehospitalized for any period when they were receiving the LAIA. Incidences of pre- and post-LAIA hospitalizations were calculated in 100 person-years.
Parallel-group analysis was analyzed using the χ2 and Mann-Whitney U tests. Pre-post analyses were analyzed using the Wilcoxon rank sum test. P was set at < .05 for statistical significance.
Results
We screened 111 veterans, and 97 were included in this study (risperidone microspheres, 44; paliperidone palmitate, 53). Mean (SD) age was 46 (13.8) years, 92% were male, 38% were White, 94% were diagnosed with schizophrenia or schizoaffective disorder, and 11% were homeless. Substance use was documented as 52% for nicotine products, 40% for alcohol, 31% for cannabis, 27% for methamphetamine, 7% for cocaine, and 3% for opioids. Cannabis, methamphetamine, cocaine, and opioid use were based on clinician documentation and listed as active diagnoses at the time of LAIA initiation. Statistical significance was found in index hospitalizations P = .009) and history of cocaine use disorder (6.8% vs 7.5%, P < .001).
Veterans administered risperidone microspheres had fewer mean (SD) post-LAIA hospitalizations (0.4 [1.0] vs 0.9 [1.5]; P = .02) and were less likely to be rehospitalized (22.7% vs 47.2%, P = .01) compared with paliperidone palmitate. However, veterans taking risperidone microspheres had a shorter mean (SD) treatment duration (41.6 [40.2] vs 58.2 [45.7] weeks, P = .04) compared with paliperidone palmitate, mainly because patients switched to a different LAIA or oral antipsychotic. No differences were detected in nonadherence and discontinuation between risperidone microspheres and paliperidone palmitate. All veterans in the risperidone microspheres group adhered to oral risperidone crossover with an average 87.8% days covered (Table 2).
The average maintenance dose of risperidone microspheres was 42 mg every 2 weeks and 153 mg every 4 weeks for paliperidone palmitate.
Across the sample, 84% of veterans had a previous psychiatric hospitalization, although veterans initiated on risperidone microspheres had significantly higher mean (SD) index hospitalizations than those started on paliperidone palmitate (3.2 [2.6] risperidone microspheres vs 2.1 [1.9] paliperidone palmitate, P = .009). Both groups had significant decreases in mean (SD) hospitalizations (3.2 [2.6] to 0.4 [1.0], risperidone microspheres vs 2.1 [1.9] to 0.9 [1.5] paliperidone palmitate). The risperidone microspheres group had a larger decrease in mean (SD) hospitalizations post-LAIA (2.8 [2.9] risperidone microspheres vs 1.3 [1.7] paliperidone palmitate, P = .001) (Table 3).
Differences in incidence per 100 person-years between pre- and post-LAIA hospitalizations were larger in risperidone microspheres users than in paliperidone palmitate (73.8 vs 33.7, P = .01) (Figure). No differences between risperidone microspheres and paliperidone palmitate were detected when looking at incidence pre-LAIA (102.2 vs 75.8, P = .22) and post-LAIA (28.4 vs 42.1, P = .38) separately.
Thirty veterans in the risperidone microspheres group discontinued LAIA: 11 were nonadherent, 5 experienced adverse effects (AEs), and 14 discontinued due to inconvenience. Among 33 veterans in the paliperidone palmitate group who discontinued the LAIA, 15 were nonadherent, 11 experienced AEs, 4 stopped due to of inconvenience, and 3 switched to a less frequently administered LAIA. The most common AEs reported were injection site reactions, cholinergic AEs (salivation, lacrimation, urination), orthostasis, and weight gain.
Discussion
The main finding of this study was that initiation of LAIAs significantly reduced hospitalizations. Veterans taking risperidone microspheres had higher index hospitalizations and lower posttreatment hospitalizations compared with paliperidone palmitate. We found that patients initiated on risperidone microspheres had more hospitalizations before use of a LAIA than those initiated on paliperidone palmitate. Risperidone microspheres reduced the number of hospitalization post-LAIA significantly more than paliperidone palmitate. We also found that veterans taking risperidone microspheres were on the medication for less mean (SD) time than those on paliperidone palmitate (41.6 [40.2] vs 58.2 [45.7] weeks; P = .04).
To our knowledge, this is one of the few studies that compared outcomes of psychiatric hospitalizations, medication adherence, and treatment discontinuation between risperidone microspheres and paliperidone palmitate, specifically in a veteran population.16-19 Limosin and colleagues aimed to compare length of stay during the initial hospitalization, rehospitalization risk, and treatment duration between risperidone microspheres and paliperidone palmitate in patients with schizophrenia.16 These researchers detected no differences in initial hospitalization duration and time to rehospitalization between risperidone microspheres and paliperidone palmitate.16 The study revealed a more favorable trend in time to discontinuation for paliperidone palmitate, but switching between LAIAs might have confounded the data.16 The authors note that their study lacked power, and patients on paliperidone palmitate had significantly more nonpsychiatric comorbidities.16 Joshi and colleagues looked at adherence, medication discontinuation, hospitalization rates, emergency department visits, and hospitalization costs between risperidone microspheres and paliperidone palmitate in patients identified in Truven MarketScan Commercial, Medicare Supplemental, and Medicaid Multi-State insurance databases.17 The authors found paliperidone palmitate to be superior in all objectives with better adherence, lower discontinuation rates, less likelihood of hospitalization, fewer emergency department visits, and lower hospitalization costs compared with risperidone microspheres.17 Korell and colleagues aimed to establish reference ranges for plasma concentrations of risperidone and paliperidone among adherent patients.18
The researchers established reference ranges for risperidone and paliperidone plasma concentrations that represented expected variability within a population and were derived from population pharmacokinetic models.18 Gopal and colleagues conducted a post hoc comparison between paliperidone palmitate and oral risperidone during initiation of long-acting injectable risperidone in patients with acute schizophrenia.19 The researchers found that during the first month after initiating long-acting injectable risperidone, paliperidone palmitate without oral supplementation had similar efficacy and safety to oral risperidone among these patients.19
LAIAs can create a steadier drug plasma concentration compared with oral antipsychotics and do not need to be taken daily. These agents improve adherence by reducing the frequency of medication administrations.20-24 Assessing nonadherence is easier with LAIAs by counting missed injections compared with oral antipsychotics that require calculation of percentage of days covered.25
The results in our study are somewhat unexpected in part because of the close relationship between risperidone and paliperidone. Risperidone is converted to paliperidone (9-OH-risperidone) via hepatic cytochrome P450 2D6. Although the molecules do not have identical pharmacologic profiles, it is accepted that they are similar enough that risperidone can establish oral tolerability when transitioning therapy to paliperidone palmitate and vice versa.24 Although the active moiety in risperidone microspheres and paliperidone palmitate is similar, the dosing interval for risperidone microspheres is 2 weeks compared with 4 weeks with paliperidone palmitate. One potential explanation as to why veterans started on risperidone microspheres experienced better outcomes is because they had twice as many office visits with the health care team. Facility procedures dictate veterans receive the LAIA at an on-site clinic. During the visits, a licensed vocational nurse administers the injection and monitors the patient for 15 to 30 minutes afterward.
Despite new LAIAs coming to market, high-quality data examining potential differences in treatment outcomes among agents are limited. This is problematic for clinicians who want to optimize care by understanding how administration schedules or other aspects of LAIA use could modify treatment outcomes. Our results suggest that an advantage might exist in selecting an agent with a more frequent administration schedule, at least initially. This could allow for close monitoring and regular therapeutic contact, which could improve short-term outcomes. This conclusion is supported by meta-analyses, randomized controlled trials, and conceptual articles conducted by Wehring and colleagues, Berwaerts and colleagues, and Parellada and colleagues, respectively, who examined patients on different LAIAs and contact with health care professionals as part of their research.26-28 These researchers concluded that patients who had regular contact with a health care professional had better outcomes when initiated on a LAIA.26-28
Limitations
There are several limitations in this study. Retrospective and observational methods introduce risks of bias and confounding variables. Sample size might have limited statistical power to detect differences. Veterans might have had undocumented pre- or posthospitalizations at other institutions, which was not accounted for and lack of rehospitalization is not conclusive of a positive outcome. Institutions could improve on our study and help to fill gaps in comparative data by conducting larger analyses over longer periods and including more LAIA agents.
Conclusions
Although veterans that were administered risperidone microspheres had a shorter treatment duration, they were less likely to be rehospitalized, had a fewer mean number of post-LAIA hospitalizations, and had a larger difference in incidence in 100 person-years compared with veterans on paliperidone palmitate. Nonadherence and discontinuation rates were comparable between risperidone microspheres and paliperidone palmitate. Future studies could aim to further clarify differences in outcomes among agents or administration schedules.
1. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
2. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223. doi:10.1056/NEJMoa051688
3. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500
4. Haywood TW, Kravitz HM, Grossman LS, Cavanaugh JL Jr, Davis JM, Lewis DA. Predicting the “revolving door” phenomenon among patients with schizophrenic, schizoaffective, and affective disorders. Am J Psychiatry. 1995;152(6):856-561. doi:10.1176/ajp.152.6.856
5. Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8:32. doi:10.1186/1471-244X-8-32
6. Weiden PJ, Kozma C, Grogg A, Locklear J. Partial compliance and risk of rehospitalization among California Medicaid patients with schizophrenia. Psychiatr Serv. 2004;55(8):886-891. doi:10.1176/appi.ps.55.8.886
7. Gilmer TP, Dolder CR, Lacro JP, et al. Adherence to treatment with antipsychotic medication and health care costs among Medicaid beneficiaries with schizophrenia. Am J Psychiatry. 2004;161(4):692-699. doi:10.1176/appi.ajp.161.4.692
8. Lafeuille MH, Dean J, Carter V, et al. Systematic review of long-acting injectables versus oral atypical antipsychotics on hospitalization in schizophrenia. Curr Med Res Opin. 2014;30(8):1643-1655. doi:10.1185/03007995.2014.915211
9. Yu W, Wagner TH, Chen S, Barnett PG. Average cost of VA rehabilitation, mental health, and long-term hospital stays. Med Care Res Rev. 2003;60(3 suppl):40S-53S. doi:10.1177/1077558703256724
10. Duncan EJ, Woolson SL, Hamer RM. Treatment compliance in veterans administration schizophrenia spectrum patients treated with risperidone long-acting injectable. Int Clin Psychopharmacol. 2012;27(5):283-290. doi:10.1097/YIC.0b013e328354b534
11. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
12. Dimitropoulos E, Drogemuller L, Wong K. Evaluation of concurrent oral and long-acting injectable antipsychotic prescribing at the Minneapolis Veterans Affairs Health Care System. J Clin Psychopharmacol. 2017;37(5):605-608. doi:10.1097/JCP.0000000000000755
13. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
14. Risperdal Consta. Package insert. Janssen Pharmaceutical; 2007.
15. Invega Sustenna. Package insert. Janssen Pharmaceutical; 2009.
16. Limosin F, Belhadi D, Comet D, et al. Comparison of paliperidone palmitate and risperidone long-acting injection in schizophrenic patients: results from a multicenter retrospective cohort study in France. J Clin Psychopharmacol. 2018;38(1):19-26. doi:10.1097/JCP.0000000000000827
17. Joshi K, Pan X, Wang R, Yang E, Benson C. Healthcare resource utilization of second-generation long-acting injectable antipsychotics in schizophrenia: risperidone versus paliperidone palmitate. Curr Med Res Opin. 2016;32(11):1873-1881. doi: 10.1080/03007995.2016.1219706
18. Korell J, Green B, Remmerie B, Vermeulen A. Determination of plasma concentration reference ranges for risperidone and paliperidone. CPT Pharmacometrics Syst Pharmacol. 2017;6(9):589-595. doi:10.1002/psp4.12217
19. Gopal S, Pandina G, Lane R, et al. A post-hoc comparison of paliperidone palmitate to oral risperidone during initiation of long-acting risperidone injection in patients with acute schizophrenia. Innov Clin Neurosci. 2011;8(8):26-33.
20. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
21. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
22. Green AI, Brunette MF, Dawson R, et al. Long-acting injectable vs oral risperidone for schizophrenia and co-occurring alcohol use disorder: a randomized trial. J Clin Psychiatry. 2015;76(10):1359-1365. doi:10.4088/JCP.13m08838
23. Rezansoff SN, Moniruzzaman A, Fazel S, Procyshyn R, Somers JM. Adherence to antipsychotic medication among homeless adults in Vancouver, Canada: a 15-year retrospective cohort study. Soc Psychiatry Psychiatr Epidemiol. 2016;51(12):1623-1632. doi:10.1007/s00127-016-1259-7
24. Castillo EG, Stroup TS. Effectiveness of long-acting injectable antipsychotics: a clinical perspective. Evid Based Ment Health. 2015;18(2):36-39. doi:10.1136/eb-2015-102086
25. Marder SR. Overview of partial compliance. J Clin Psychiatry. 2003;64 (suppl 16):3-9.
26. Wehring HJ, Thedford S, Koola M, Kelly DL. Patient and health care provider perspectives on long acting injectable antipsychotics in schizophrenia and the introduction of olanzapine long-acting injection. J Cent Nerv Syst Dis. 2011;2011(3):107-123. doi:10.4137/JCNSD.S4091
27. Berwaerts J, Liu Y, Gopal S, et al. Efficacy and safety of the 3-month formulation of paliperidone palmitate vs placebo for relapse prevention of schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2015;72(8):830-839. doi:10.1001/jamapsychiatry.2015.0241
28. Parellada E, Bioque M. Barriers to the use of long-acting injectable antipsychotics in the management of schizophrenia. CNS Drugs. 2016;30(8):689-701. doi:10.1007/s40263-016-0350-7
1. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl 2):1-56.
2. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223. doi:10.1056/NEJMoa051688
3. Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv. 2008;59(5):500-506. doi:10.1176/ps.2008.59.5.500
4. Haywood TW, Kravitz HM, Grossman LS, Cavanaugh JL Jr, Davis JM, Lewis DA. Predicting the “revolving door” phenomenon among patients with schizophrenic, schizoaffective, and affective disorders. Am J Psychiatry. 1995;152(6):856-561. doi:10.1176/ajp.152.6.856
5. Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8:32. doi:10.1186/1471-244X-8-32
6. Weiden PJ, Kozma C, Grogg A, Locklear J. Partial compliance and risk of rehospitalization among California Medicaid patients with schizophrenia. Psychiatr Serv. 2004;55(8):886-891. doi:10.1176/appi.ps.55.8.886
7. Gilmer TP, Dolder CR, Lacro JP, et al. Adherence to treatment with antipsychotic medication and health care costs among Medicaid beneficiaries with schizophrenia. Am J Psychiatry. 2004;161(4):692-699. doi:10.1176/appi.ajp.161.4.692
8. Lafeuille MH, Dean J, Carter V, et al. Systematic review of long-acting injectables versus oral atypical antipsychotics on hospitalization in schizophrenia. Curr Med Res Opin. 2014;30(8):1643-1655. doi:10.1185/03007995.2014.915211
9. Yu W, Wagner TH, Chen S, Barnett PG. Average cost of VA rehabilitation, mental health, and long-term hospital stays. Med Care Res Rev. 2003;60(3 suppl):40S-53S. doi:10.1177/1077558703256724
10. Duncan EJ, Woolson SL, Hamer RM. Treatment compliance in veterans administration schizophrenia spectrum patients treated with risperidone long-acting injectable. Int Clin Psychopharmacol. 2012;27(5):283-290. doi:10.1097/YIC.0b013e328354b534
11. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
12. Dimitropoulos E, Drogemuller L, Wong K. Evaluation of concurrent oral and long-acting injectable antipsychotic prescribing at the Minneapolis Veterans Affairs Health Care System. J Clin Psychopharmacol. 2017;37(5):605-608. doi:10.1097/JCP.0000000000000755
13. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
14. Risperdal Consta. Package insert. Janssen Pharmaceutical; 2007.
15. Invega Sustenna. Package insert. Janssen Pharmaceutical; 2009.
16. Limosin F, Belhadi D, Comet D, et al. Comparison of paliperidone palmitate and risperidone long-acting injection in schizophrenic patients: results from a multicenter retrospective cohort study in France. J Clin Psychopharmacol. 2018;38(1):19-26. doi:10.1097/JCP.0000000000000827
17. Joshi K, Pan X, Wang R, Yang E, Benson C. Healthcare resource utilization of second-generation long-acting injectable antipsychotics in schizophrenia: risperidone versus paliperidone palmitate. Curr Med Res Opin. 2016;32(11):1873-1881. doi: 10.1080/03007995.2016.1219706
18. Korell J, Green B, Remmerie B, Vermeulen A. Determination of plasma concentration reference ranges for risperidone and paliperidone. CPT Pharmacometrics Syst Pharmacol. 2017;6(9):589-595. doi:10.1002/psp4.12217
19. Gopal S, Pandina G, Lane R, et al. A post-hoc comparison of paliperidone palmitate to oral risperidone during initiation of long-acting risperidone injection in patients with acute schizophrenia. Innov Clin Neurosci. 2011;8(8):26-33.
20. Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754-768. doi:10.18553/jmcp.2015.21.9.754
21. Romstadt N, Wonson E. Outcomes comparison of long-acting injectable antipsychotic initiation in treatment-naïve veterans in the inpatient versus outpatient setting. Ment Health Clin. 2018;8(1):24-27. doi:10.9740/mhc.2018.01.024
22. Green AI, Brunette MF, Dawson R, et al. Long-acting injectable vs oral risperidone for schizophrenia and co-occurring alcohol use disorder: a randomized trial. J Clin Psychiatry. 2015;76(10):1359-1365. doi:10.4088/JCP.13m08838
23. Rezansoff SN, Moniruzzaman A, Fazel S, Procyshyn R, Somers JM. Adherence to antipsychotic medication among homeless adults in Vancouver, Canada: a 15-year retrospective cohort study. Soc Psychiatry Psychiatr Epidemiol. 2016;51(12):1623-1632. doi:10.1007/s00127-016-1259-7
24. Castillo EG, Stroup TS. Effectiveness of long-acting injectable antipsychotics: a clinical perspective. Evid Based Ment Health. 2015;18(2):36-39. doi:10.1136/eb-2015-102086
25. Marder SR. Overview of partial compliance. J Clin Psychiatry. 2003;64 (suppl 16):3-9.
26. Wehring HJ, Thedford S, Koola M, Kelly DL. Patient and health care provider perspectives on long acting injectable antipsychotics in schizophrenia and the introduction of olanzapine long-acting injection. J Cent Nerv Syst Dis. 2011;2011(3):107-123. doi:10.4137/JCNSD.S4091
27. Berwaerts J, Liu Y, Gopal S, et al. Efficacy and safety of the 3-month formulation of paliperidone palmitate vs placebo for relapse prevention of schizophrenia: a randomized clinical trial. JAMA Psychiatry. 2015;72(8):830-839. doi:10.1001/jamapsychiatry.2015.0241
28. Parellada E, Bioque M. Barriers to the use of long-acting injectable antipsychotics in the management of schizophrenia. CNS Drugs. 2016;30(8):689-701. doi:10.1007/s40263-016-0350-7