User login
Prednisone, colchicine equivalent in efficacy for CPP crystal arthritis
PHILADELPHIA – Prednisone appears to have the edge over colchicine for control of pain in patients with acute calcium pyrophosphate (CPP) crystal arthritis, an intensely painful rheumatic disease primarily affecting older patients.
Among 111 patients with acute CPP crystal arthritis randomized to receive either prednisone or colchicine for control of acute pain in a multicenter study, 2 days of therapy with the oral agents provided equivalent pain relief on the second day, and patients generally tolerated each agent well, reported Tristan Pascart, MD, from the Groupement Hospitalier de l’Institut Catholique de Lille (France).
“Almost three-fourths of patients are considered to be good responders to both drugs on day 3, and, maybe, safety is the key issue distinguishing the two treatments: Colchicine was generally well tolerated, but even with this very short time frame of treatment, one patient out of five had diarrhea, which is more of a concern in this elderly population at risk of dehydration,” he said in an oral abstract session at the annual meeting of the American College of Rheumatology.
In contrast, only about 6% of patients assigned to prednisone had diarrhea, and other adverse events that occurred more frequently with the corticosteroid, including hypertension, hyperglycemia, and insomnia all resolved after the therapy was stopped.
Common and acutely painful
Acute CPP crystal arthritis is a common complication that often occurs during hospitalization for primarily nonrheumatologic causes, Dr. Pascart said, and “in the absence of clinical trials, the management relies on expert opinion, which stems from extrapolated data from gap studies” primarily with prednisone or colchicine, Dr. Pascart said.
To fill in the knowledge gap, Dr. Pascart and colleagues conducted the COLCHICORT study to evaluate whether the two drugs were comparable in efficacy and safety for control of acute pain in a vulnerable population.
The multicenter, open-label trial included patients older than age 65 years with an estimated glomerular filtration rate above 30 mL/min per 1.73 m2 who presented with acute CPP deposition arthritis with symptoms occurring within the previous 36 hours. CPP arthritis was defined by the identification of CPP crystals on synovial fluid analysis or typical clinical presentation with evidence of chondrocalcinosis on x-rays or ultrasound.
Patients with a history of gout, cognitive decline that could impair pain assessment, or contraindications to either of the study drugs were excluded.
The participants were randomized to receive either colchicine 1.5 mg (1 mg to start, then 0.5 mg one hour later) at baseline and then 1 mg on day 1, or oral prednisone 30 mg at baseline and on day 1. The patients also received 1 g of systemic acetaminophen, and three 50-mg doses of tramadol during the first 24 hours.
Of the 111 patients randomized, 54 were assigned to receive prednisone, and 57 were assigned to receive colchicine. Baseline characteristics were similar between the groups, with a mean age of about 86 years, body mass index of around 25 kg/m2, and blood pressure in the range of 130/69 mm Hg.
For nearly half of all patients in study each arm the most painful joint was the knee, followed by wrists and ankles.
There was no difference between the groups in the primary efficacy outcome of a change at 24 hours over baseline in visual analog scale (VAS) (0-100 mm) scores, either in a per-protocol analysis or modified intention-to-treat analysis. The mean change in VAS at 24 hours in the colchicine group was –36.6 mm, compared with –37.7 mm in the prednisone group. The investigators had previously determined that any difference between the two drugs of less than 13 mm on pain VAS at 24 hours would meet the definition for equivalent efficacy.
In both groups, a majority of patients had either an improvement greater than 50% in pain VAS scores and/or a pain VAS score less than 40 mm at both 24 and 48 hours.
At 7 days of follow-up, 21.8% of patients assigned to colchicine had diarrhea, compared with 5.6% of those assigned to prednisone. Adverse events occurring more frequently with prednisone included hyperglycemia, hypertension, and insomnia.
Patients who received colchicine and were also on statins had a trend toward a higher risk for diarrhea, but the study was not adequately powered to detect an association, and the trend was not statistically significant, Dr. Pascart said.
“Taken together, safety issues suggest that prednisone should be considered as the first-line therapy in acute CPP crystal arthritis. Future research is warranted to determine factors increasing the risk of colchicine-induced diarrhea,” he concluded.
Both drugs are used
Sara K. Tedeschi, MD, from Brigham & Women’s Hospital in Boston, who attended the session where the data were presented, has a special clinical interest in CPP deposition disease. She applauded Dr. Pascart and colleagues for conducting a rare clinical trial in CPP crystal arthritis.
In an interview, she said that the study suggests “we can keep in mind shorter courses of treatment for acute CPP crystal arthritis; I think that’s one big takeaway from this study.”
Asked whether she would change her practice based on the findings, Dr. Tedeschi replied: “I personally am not sure that I would be moved to use prednisone more than colchicine; I actually take away from this that colchicine is equivalent to prednisone for short-term use for CPP arthritis, but I think it’s also really important to note that this is in the context of quite a lot of acetaminophen and quite a lot of tramadol, and frankly I don’t usually use tramadol with my patients, but I might consider doing that, especially as there were no delirium events in this population.”
Dr. Tedeschi was not involved in the study.
Asked the same question, Michael Toprover, MD, from New York University Langone Medical Center, a moderator of the session who was not involved in the study, said: “I usually use a combination of medications. I generally, in someone who is hospitalized in particular and is in such severe pain, use a combination of colchicine and prednisone, unless I’m worried about infection, in which case I’ll start colchicine until we’ve proven that it’s CPPD, and then I’ll add prednisone.”
The study was funded by PHRC-1 GIRCI Nord Ouest, a clinical research program funded by the Ministry of Health in France. Dr. Pascart, Dr. Tedeschi, and Dr. Toprover all reported having no relevant conflicts of interest.
PHILADELPHIA – Prednisone appears to have the edge over colchicine for control of pain in patients with acute calcium pyrophosphate (CPP) crystal arthritis, an intensely painful rheumatic disease primarily affecting older patients.
Among 111 patients with acute CPP crystal arthritis randomized to receive either prednisone or colchicine for control of acute pain in a multicenter study, 2 days of therapy with the oral agents provided equivalent pain relief on the second day, and patients generally tolerated each agent well, reported Tristan Pascart, MD, from the Groupement Hospitalier de l’Institut Catholique de Lille (France).
“Almost three-fourths of patients are considered to be good responders to both drugs on day 3, and, maybe, safety is the key issue distinguishing the two treatments: Colchicine was generally well tolerated, but even with this very short time frame of treatment, one patient out of five had diarrhea, which is more of a concern in this elderly population at risk of dehydration,” he said in an oral abstract session at the annual meeting of the American College of Rheumatology.
In contrast, only about 6% of patients assigned to prednisone had diarrhea, and other adverse events that occurred more frequently with the corticosteroid, including hypertension, hyperglycemia, and insomnia all resolved after the therapy was stopped.
Common and acutely painful
Acute CPP crystal arthritis is a common complication that often occurs during hospitalization for primarily nonrheumatologic causes, Dr. Pascart said, and “in the absence of clinical trials, the management relies on expert opinion, which stems from extrapolated data from gap studies” primarily with prednisone or colchicine, Dr. Pascart said.
To fill in the knowledge gap, Dr. Pascart and colleagues conducted the COLCHICORT study to evaluate whether the two drugs were comparable in efficacy and safety for control of acute pain in a vulnerable population.
The multicenter, open-label trial included patients older than age 65 years with an estimated glomerular filtration rate above 30 mL/min per 1.73 m2 who presented with acute CPP deposition arthritis with symptoms occurring within the previous 36 hours. CPP arthritis was defined by the identification of CPP crystals on synovial fluid analysis or typical clinical presentation with evidence of chondrocalcinosis on x-rays or ultrasound.
Patients with a history of gout, cognitive decline that could impair pain assessment, or contraindications to either of the study drugs were excluded.
The participants were randomized to receive either colchicine 1.5 mg (1 mg to start, then 0.5 mg one hour later) at baseline and then 1 mg on day 1, or oral prednisone 30 mg at baseline and on day 1. The patients also received 1 g of systemic acetaminophen, and three 50-mg doses of tramadol during the first 24 hours.
Of the 111 patients randomized, 54 were assigned to receive prednisone, and 57 were assigned to receive colchicine. Baseline characteristics were similar between the groups, with a mean age of about 86 years, body mass index of around 25 kg/m2, and blood pressure in the range of 130/69 mm Hg.
For nearly half of all patients in study each arm the most painful joint was the knee, followed by wrists and ankles.
There was no difference between the groups in the primary efficacy outcome of a change at 24 hours over baseline in visual analog scale (VAS) (0-100 mm) scores, either in a per-protocol analysis or modified intention-to-treat analysis. The mean change in VAS at 24 hours in the colchicine group was –36.6 mm, compared with –37.7 mm in the prednisone group. The investigators had previously determined that any difference between the two drugs of less than 13 mm on pain VAS at 24 hours would meet the definition for equivalent efficacy.
In both groups, a majority of patients had either an improvement greater than 50% in pain VAS scores and/or a pain VAS score less than 40 mm at both 24 and 48 hours.
At 7 days of follow-up, 21.8% of patients assigned to colchicine had diarrhea, compared with 5.6% of those assigned to prednisone. Adverse events occurring more frequently with prednisone included hyperglycemia, hypertension, and insomnia.
Patients who received colchicine and were also on statins had a trend toward a higher risk for diarrhea, but the study was not adequately powered to detect an association, and the trend was not statistically significant, Dr. Pascart said.
“Taken together, safety issues suggest that prednisone should be considered as the first-line therapy in acute CPP crystal arthritis. Future research is warranted to determine factors increasing the risk of colchicine-induced diarrhea,” he concluded.
Both drugs are used
Sara K. Tedeschi, MD, from Brigham & Women’s Hospital in Boston, who attended the session where the data were presented, has a special clinical interest in CPP deposition disease. She applauded Dr. Pascart and colleagues for conducting a rare clinical trial in CPP crystal arthritis.
In an interview, she said that the study suggests “we can keep in mind shorter courses of treatment for acute CPP crystal arthritis; I think that’s one big takeaway from this study.”
Asked whether she would change her practice based on the findings, Dr. Tedeschi replied: “I personally am not sure that I would be moved to use prednisone more than colchicine; I actually take away from this that colchicine is equivalent to prednisone for short-term use for CPP arthritis, but I think it’s also really important to note that this is in the context of quite a lot of acetaminophen and quite a lot of tramadol, and frankly I don’t usually use tramadol with my patients, but I might consider doing that, especially as there were no delirium events in this population.”
Dr. Tedeschi was not involved in the study.
Asked the same question, Michael Toprover, MD, from New York University Langone Medical Center, a moderator of the session who was not involved in the study, said: “I usually use a combination of medications. I generally, in someone who is hospitalized in particular and is in such severe pain, use a combination of colchicine and prednisone, unless I’m worried about infection, in which case I’ll start colchicine until we’ve proven that it’s CPPD, and then I’ll add prednisone.”
The study was funded by PHRC-1 GIRCI Nord Ouest, a clinical research program funded by the Ministry of Health in France. Dr. Pascart, Dr. Tedeschi, and Dr. Toprover all reported having no relevant conflicts of interest.
PHILADELPHIA – Prednisone appears to have the edge over colchicine for control of pain in patients with acute calcium pyrophosphate (CPP) crystal arthritis, an intensely painful rheumatic disease primarily affecting older patients.
Among 111 patients with acute CPP crystal arthritis randomized to receive either prednisone or colchicine for control of acute pain in a multicenter study, 2 days of therapy with the oral agents provided equivalent pain relief on the second day, and patients generally tolerated each agent well, reported Tristan Pascart, MD, from the Groupement Hospitalier de l’Institut Catholique de Lille (France).
“Almost three-fourths of patients are considered to be good responders to both drugs on day 3, and, maybe, safety is the key issue distinguishing the two treatments: Colchicine was generally well tolerated, but even with this very short time frame of treatment, one patient out of five had diarrhea, which is more of a concern in this elderly population at risk of dehydration,” he said in an oral abstract session at the annual meeting of the American College of Rheumatology.
In contrast, only about 6% of patients assigned to prednisone had diarrhea, and other adverse events that occurred more frequently with the corticosteroid, including hypertension, hyperglycemia, and insomnia all resolved after the therapy was stopped.
Common and acutely painful
Acute CPP crystal arthritis is a common complication that often occurs during hospitalization for primarily nonrheumatologic causes, Dr. Pascart said, and “in the absence of clinical trials, the management relies on expert opinion, which stems from extrapolated data from gap studies” primarily with prednisone or colchicine, Dr. Pascart said.
To fill in the knowledge gap, Dr. Pascart and colleagues conducted the COLCHICORT study to evaluate whether the two drugs were comparable in efficacy and safety for control of acute pain in a vulnerable population.
The multicenter, open-label trial included patients older than age 65 years with an estimated glomerular filtration rate above 30 mL/min per 1.73 m2 who presented with acute CPP deposition arthritis with symptoms occurring within the previous 36 hours. CPP arthritis was defined by the identification of CPP crystals on synovial fluid analysis or typical clinical presentation with evidence of chondrocalcinosis on x-rays or ultrasound.
Patients with a history of gout, cognitive decline that could impair pain assessment, or contraindications to either of the study drugs were excluded.
The participants were randomized to receive either colchicine 1.5 mg (1 mg to start, then 0.5 mg one hour later) at baseline and then 1 mg on day 1, or oral prednisone 30 mg at baseline and on day 1. The patients also received 1 g of systemic acetaminophen, and three 50-mg doses of tramadol during the first 24 hours.
Of the 111 patients randomized, 54 were assigned to receive prednisone, and 57 were assigned to receive colchicine. Baseline characteristics were similar between the groups, with a mean age of about 86 years, body mass index of around 25 kg/m2, and blood pressure in the range of 130/69 mm Hg.
For nearly half of all patients in study each arm the most painful joint was the knee, followed by wrists and ankles.
There was no difference between the groups in the primary efficacy outcome of a change at 24 hours over baseline in visual analog scale (VAS) (0-100 mm) scores, either in a per-protocol analysis or modified intention-to-treat analysis. The mean change in VAS at 24 hours in the colchicine group was –36.6 mm, compared with –37.7 mm in the prednisone group. The investigators had previously determined that any difference between the two drugs of less than 13 mm on pain VAS at 24 hours would meet the definition for equivalent efficacy.
In both groups, a majority of patients had either an improvement greater than 50% in pain VAS scores and/or a pain VAS score less than 40 mm at both 24 and 48 hours.
At 7 days of follow-up, 21.8% of patients assigned to colchicine had diarrhea, compared with 5.6% of those assigned to prednisone. Adverse events occurring more frequently with prednisone included hyperglycemia, hypertension, and insomnia.
Patients who received colchicine and were also on statins had a trend toward a higher risk for diarrhea, but the study was not adequately powered to detect an association, and the trend was not statistically significant, Dr. Pascart said.
“Taken together, safety issues suggest that prednisone should be considered as the first-line therapy in acute CPP crystal arthritis. Future research is warranted to determine factors increasing the risk of colchicine-induced diarrhea,” he concluded.
Both drugs are used
Sara K. Tedeschi, MD, from Brigham & Women’s Hospital in Boston, who attended the session where the data were presented, has a special clinical interest in CPP deposition disease. She applauded Dr. Pascart and colleagues for conducting a rare clinical trial in CPP crystal arthritis.
In an interview, she said that the study suggests “we can keep in mind shorter courses of treatment for acute CPP crystal arthritis; I think that’s one big takeaway from this study.”
Asked whether she would change her practice based on the findings, Dr. Tedeschi replied: “I personally am not sure that I would be moved to use prednisone more than colchicine; I actually take away from this that colchicine is equivalent to prednisone for short-term use for CPP arthritis, but I think it’s also really important to note that this is in the context of quite a lot of acetaminophen and quite a lot of tramadol, and frankly I don’t usually use tramadol with my patients, but I might consider doing that, especially as there were no delirium events in this population.”
Dr. Tedeschi was not involved in the study.
Asked the same question, Michael Toprover, MD, from New York University Langone Medical Center, a moderator of the session who was not involved in the study, said: “I usually use a combination of medications. I generally, in someone who is hospitalized in particular and is in such severe pain, use a combination of colchicine and prednisone, unless I’m worried about infection, in which case I’ll start colchicine until we’ve proven that it’s CPPD, and then I’ll add prednisone.”
The study was funded by PHRC-1 GIRCI Nord Ouest, a clinical research program funded by the Ministry of Health in France. Dr. Pascart, Dr. Tedeschi, and Dr. Toprover all reported having no relevant conflicts of interest.
AT ACR 2022
Total replacement and fusion yield similar outcomes for ankle osteoarthritis
Ankle osteoarthritis remains a cause of severe pain and disability. Patients are treated nonoperatively if possible, but surgery is often needed for individuals with end-stage disease, wrote Andrew Goldberg, MBBS, of University College London and colleagues in the Annals of Internal Medicine.
“Most patients with ankle arthritis respond to nonoperative treatments, such as weight loss, activity modification, support braces, and analgesia, [but] once the disease has progressed to end-stage osteoarthritis, the main surgical treatments are total ankle re-placement or ankle arthrodesis,” Dr. Goldberg said, in an interview.
In the new study, patients were randomized to receive either a total ankle replacement (TAR) or ankle fusion (AF).
“We showed that, in both treatment groups the clinical scores improved hugely, by more than three times the minimal clinically important difference,” Dr. Goldberg said in an interview.
“Although the ankle replacement arm improved, on average, by more than an extra 4 points over ankle fusion, this was not considered clinically or statistically significant,” he said.
The study is the first randomized trial to show high-quality and robust results, he noted, and findings support data from previous studies.
“Although both TAR and ankle fusion have been shown to be effective, they are very different treatments, with one fusing the bones so that there is no ankle joint movement, and the other replacing the joint with the aim of retaining ankle joint movement. It is difficult for a patient to know which treatment is more suitable for them, with most seeking guidance from their surgeon,” he said.
Generating high-quality evidence
The study, a randomized, multicenter, open-label trial known as TARVA (Total Ankle Replacement Versus Ankle Arthrodesis), aimed to compare the clinical effectiveness of the two existing publicly funded U.K. treatment options, the authors wrote.
Patients were recruited at 17 U.K. centers between March 6, 2015, and Jan. 10, 2019. The study enrolled 303 adults aged 50-85 years with end-stage ankle osteoarthritis. The mean age of the participants was 68 years; 71% were men. A total of 137 TAR patients and 144 ankle fusion patients completed their surgeries with clinical scores available for analysis. Baseline characteristics were mainly similar between the groups.
Blinding was not possible because of the nature of the procedures, but the surgeons who screened the patients were not aware of the randomization allocations, the researchers noted. A total of 33 surgeons participated in the trial, with a median number of seven patients per surgeon during the study period.
For TAR, U.K. surgeons use both two-component, fixed-bearing and three-component, mobile-bearing implants, the authors write. Ankle fusion was done using the surgeon’s usual technique of either arthroscopic-assisted or open ankle fusion.
The primary outcome was the change in the Manchester–Oxford Foot Questionnaire walking/standing (MOXFQ-W/S) domain scores from baseline to 52 weeks after surgery. The MOXFQ-W/S uses a scale of 0-100, with lower scores representing better outcomes. Secondary outcomes included change in the MOXFQ-W/S scores at 26 weeks after surgery, as well as measures of patient quality of life.
No statistically significant difference
Overall, the mean MOXFQ-W/S scores improved significantly from baseline to 52 weeks for both groups, with average improvements of 49.9 in the TAR group and 44.4 points in the AF group. The average scores at 52 weeks were 31.4 in the TAR group and 36.8 in the AF group.
The adjusted difference in score change from baseline was –5.56, showing a slightly greater degree of improvement with TAR, but this difference was not clinically or statistically significant, the researchers noted.
Adverse event numbers were similar for both procedures, with 54% of TAR patients and 53% of AF patients experiencing at least 1 adverse event during the study period. Of those, 18% of TAR patients and 24% of AF patients experienced at least 1 serious adverse event.
However, the TAR patients experienced a higher rate of wound healing complications and nerve injuries, while thromboembolism was higher in the AF patients, the researchers noted.
A prespecified subgroup analysis of patients with osteoarthritis in adjacent joints suggested a greater improvement in TAR, compared with AF, a difference that increased when fixed-bearing TAR was compared with AF, the authors wrote.
“This reinforces previous reports that suggest that the presence of adjacent joint arthritis may be an indication for ankle replacement over AF,” the authors wrote in their discussion.
“Many of these patients did not have any symptoms in the adjacent joints,” they noted.
“The presence of adjacent joint arthritis, meaning the wear and tear of the joints around the ankle joint, seemed to favor ankle replacement,” Dr. Goldberg said. Approximately 30 joints in the foot continue to move after the ankle is fused, and if these adjacent joints are not healthy before surgery [as was the case in 42% of the study patients], the results of fusion were less successful, he explained.
A post hoc analysis between TAR subtypes showed that patients who had fixed-bearing TAR had significantly greater improvements, compared with AF patients, but this difference was not observed in patients who had mobile-bearing TAR, the researchers noted.
Dr. Goldberg said it was surprising “that, in a separate analysis, we found that the fixed-bearing ankle replacement patients [who accounted for half of the implants used] improved by a much greater difference when compared to ankle fusion.”
The study findings were limited by several factors including the short follow-up and study design that allowed surgeons to choose any implant and technique, the researchers noted.
Other limitations include a lack of data on cost-effectiveness and the impact of comorbidities on outcomes, they wrote. However, the study is the first completed multicenter randomized controlled trial to compare TAR and AF procedures for end-stage ankle osteoarthritis and shows that both yield similar clinical improvements, they concluded.
Data can inform treatment discussion
The take-home messages for clinicians are that both ankle replacement and ankle fusion are effective treatments that improve patients’ quality of life, and it is important to establish the health of adjacent joints before making treatment recommendations, Dr. Goldberg said.
“Careful counseling on the relative risks of each procedure should be part of the informed consent process,” he added. Ideally, all patients seeking surgical care for ankle arthritis should have a choice between ankle replacement and ankle fusion, but sometimes there is inequity of provision of the two treatments, he noted.
“We now encourage all surgeons to work in ankle arthritis networks so that every patient, no matter where they live, can have choice about the best treatment for them,” he said.
Researchers met the challenge of surgical RCT
Randomized trials of surgical interventions are challenging to conduct, and therefore limited, wrote Bruce Sangeorzan, MD, of the University of Washington, Seattle, and colleagues in an accompanying editorial. However, the new study was strengthened by the inclusion of 17 centers for heterogeneity of implant type and surgeon experience level, the editorialists said in the Annals of Internal Medicine.
The study is especially important, because ankle arthritis treatment is very understudied, compared with hip and knee arthritis, but it has a similar impact on activity, editorial coauthor Dr. Sangeorzan said in an interview.
“Randomized controlled trials are the gold standard for comparing medical therapies,” he said, “but they are very difficult to do in surgical treatments, particularly when the two treatments can be differentiated, in this case by movement of the ankle.”
In addition, there is a strong placebo effect attached to interventions, Dr. Sangeorzan noted. “Determining best-case treatment relies on prospective research, preferably randomized. Since both ankle fusion and ankle replacement are effective therapies, a prospective randomized trial is the best way to help make treatment decisions,” he said.
The current study findings are not surprising, but they are preliminary, and 1 year of follow-up is not enough to determine effectiveness, Dr. Sangeorzan emphasized. However, “the authors have done the hard work of randomizing the patients and collecting the data, and the patients can now be followed for a longer time,” he said.
“In addition, the trial was designed with multiple secondary outcome measures, so the data can be matched up with larger trials that were not randomized to identify key elements of success for each procedure,” he noted.
The key message for clinicians is that ankle arthritis has a significant impact on patients’ lives, but there are two effective treatments that can reduce the impact of the disease, said Dr. Sangeorzan. “The data suggest that there are differences in implant design and differences in comorbidities that should influence decision-making,” he added.
Additional research is needed in the form of a longer study duration with larger cohorts, said Dr. Sangeorzan. In particular, researchers need to determine what comorbidities might drive patients to one type of care vs. another, he said. “The suggestion that [patients receiving implants with two motion segments have better outcomes than those receiving implants with a one-motion segment] also deserves further study,” he added.
The research was supported by the UK National Institute for Health and Care Research Health Technology Assessment Programme. The trial was sponsored by University College London. Dr. Goldberg disclosed grant support from NIHR HTA, as well as financial relationships with companies including Stryker, Paragon 28, and stock options with Standing CT Company, Elstree Waterfront Outpatients, and X Bolt Orthopedics.
The editorialists had no financial conflicts to disclose.
Ankle osteoarthritis remains a cause of severe pain and disability. Patients are treated nonoperatively if possible, but surgery is often needed for individuals with end-stage disease, wrote Andrew Goldberg, MBBS, of University College London and colleagues in the Annals of Internal Medicine.
“Most patients with ankle arthritis respond to nonoperative treatments, such as weight loss, activity modification, support braces, and analgesia, [but] once the disease has progressed to end-stage osteoarthritis, the main surgical treatments are total ankle re-placement or ankle arthrodesis,” Dr. Goldberg said, in an interview.
In the new study, patients were randomized to receive either a total ankle replacement (TAR) or ankle fusion (AF).
“We showed that, in both treatment groups the clinical scores improved hugely, by more than three times the minimal clinically important difference,” Dr. Goldberg said in an interview.
“Although the ankle replacement arm improved, on average, by more than an extra 4 points over ankle fusion, this was not considered clinically or statistically significant,” he said.
The study is the first randomized trial to show high-quality and robust results, he noted, and findings support data from previous studies.
“Although both TAR and ankle fusion have been shown to be effective, they are very different treatments, with one fusing the bones so that there is no ankle joint movement, and the other replacing the joint with the aim of retaining ankle joint movement. It is difficult for a patient to know which treatment is more suitable for them, with most seeking guidance from their surgeon,” he said.
Generating high-quality evidence
The study, a randomized, multicenter, open-label trial known as TARVA (Total Ankle Replacement Versus Ankle Arthrodesis), aimed to compare the clinical effectiveness of the two existing publicly funded U.K. treatment options, the authors wrote.
Patients were recruited at 17 U.K. centers between March 6, 2015, and Jan. 10, 2019. The study enrolled 303 adults aged 50-85 years with end-stage ankle osteoarthritis. The mean age of the participants was 68 years; 71% were men. A total of 137 TAR patients and 144 ankle fusion patients completed their surgeries with clinical scores available for analysis. Baseline characteristics were mainly similar between the groups.
Blinding was not possible because of the nature of the procedures, but the surgeons who screened the patients were not aware of the randomization allocations, the researchers noted. A total of 33 surgeons participated in the trial, with a median number of seven patients per surgeon during the study period.
For TAR, U.K. surgeons use both two-component, fixed-bearing and three-component, mobile-bearing implants, the authors write. Ankle fusion was done using the surgeon’s usual technique of either arthroscopic-assisted or open ankle fusion.
The primary outcome was the change in the Manchester–Oxford Foot Questionnaire walking/standing (MOXFQ-W/S) domain scores from baseline to 52 weeks after surgery. The MOXFQ-W/S uses a scale of 0-100, with lower scores representing better outcomes. Secondary outcomes included change in the MOXFQ-W/S scores at 26 weeks after surgery, as well as measures of patient quality of life.
No statistically significant difference
Overall, the mean MOXFQ-W/S scores improved significantly from baseline to 52 weeks for both groups, with average improvements of 49.9 in the TAR group and 44.4 points in the AF group. The average scores at 52 weeks were 31.4 in the TAR group and 36.8 in the AF group.
The adjusted difference in score change from baseline was –5.56, showing a slightly greater degree of improvement with TAR, but this difference was not clinically or statistically significant, the researchers noted.
Adverse event numbers were similar for both procedures, with 54% of TAR patients and 53% of AF patients experiencing at least 1 adverse event during the study period. Of those, 18% of TAR patients and 24% of AF patients experienced at least 1 serious adverse event.
However, the TAR patients experienced a higher rate of wound healing complications and nerve injuries, while thromboembolism was higher in the AF patients, the researchers noted.
A prespecified subgroup analysis of patients with osteoarthritis in adjacent joints suggested a greater improvement in TAR, compared with AF, a difference that increased when fixed-bearing TAR was compared with AF, the authors wrote.
“This reinforces previous reports that suggest that the presence of adjacent joint arthritis may be an indication for ankle replacement over AF,” the authors wrote in their discussion.
“Many of these patients did not have any symptoms in the adjacent joints,” they noted.
“The presence of adjacent joint arthritis, meaning the wear and tear of the joints around the ankle joint, seemed to favor ankle replacement,” Dr. Goldberg said. Approximately 30 joints in the foot continue to move after the ankle is fused, and if these adjacent joints are not healthy before surgery [as was the case in 42% of the study patients], the results of fusion were less successful, he explained.
A post hoc analysis between TAR subtypes showed that patients who had fixed-bearing TAR had significantly greater improvements, compared with AF patients, but this difference was not observed in patients who had mobile-bearing TAR, the researchers noted.
Dr. Goldberg said it was surprising “that, in a separate analysis, we found that the fixed-bearing ankle replacement patients [who accounted for half of the implants used] improved by a much greater difference when compared to ankle fusion.”
The study findings were limited by several factors including the short follow-up and study design that allowed surgeons to choose any implant and technique, the researchers noted.
Other limitations include a lack of data on cost-effectiveness and the impact of comorbidities on outcomes, they wrote. However, the study is the first completed multicenter randomized controlled trial to compare TAR and AF procedures for end-stage ankle osteoarthritis and shows that both yield similar clinical improvements, they concluded.
Data can inform treatment discussion
The take-home messages for clinicians are that both ankle replacement and ankle fusion are effective treatments that improve patients’ quality of life, and it is important to establish the health of adjacent joints before making treatment recommendations, Dr. Goldberg said.
“Careful counseling on the relative risks of each procedure should be part of the informed consent process,” he added. Ideally, all patients seeking surgical care for ankle arthritis should have a choice between ankle replacement and ankle fusion, but sometimes there is inequity of provision of the two treatments, he noted.
“We now encourage all surgeons to work in ankle arthritis networks so that every patient, no matter where they live, can have choice about the best treatment for them,” he said.
Researchers met the challenge of surgical RCT
Randomized trials of surgical interventions are challenging to conduct, and therefore limited, wrote Bruce Sangeorzan, MD, of the University of Washington, Seattle, and colleagues in an accompanying editorial. However, the new study was strengthened by the inclusion of 17 centers for heterogeneity of implant type and surgeon experience level, the editorialists said in the Annals of Internal Medicine.
The study is especially important, because ankle arthritis treatment is very understudied, compared with hip and knee arthritis, but it has a similar impact on activity, editorial coauthor Dr. Sangeorzan said in an interview.
“Randomized controlled trials are the gold standard for comparing medical therapies,” he said, “but they are very difficult to do in surgical treatments, particularly when the two treatments can be differentiated, in this case by movement of the ankle.”
In addition, there is a strong placebo effect attached to interventions, Dr. Sangeorzan noted. “Determining best-case treatment relies on prospective research, preferably randomized. Since both ankle fusion and ankle replacement are effective therapies, a prospective randomized trial is the best way to help make treatment decisions,” he said.
The current study findings are not surprising, but they are preliminary, and 1 year of follow-up is not enough to determine effectiveness, Dr. Sangeorzan emphasized. However, “the authors have done the hard work of randomizing the patients and collecting the data, and the patients can now be followed for a longer time,” he said.
“In addition, the trial was designed with multiple secondary outcome measures, so the data can be matched up with larger trials that were not randomized to identify key elements of success for each procedure,” he noted.
The key message for clinicians is that ankle arthritis has a significant impact on patients’ lives, but there are two effective treatments that can reduce the impact of the disease, said Dr. Sangeorzan. “The data suggest that there are differences in implant design and differences in comorbidities that should influence decision-making,” he added.
Additional research is needed in the form of a longer study duration with larger cohorts, said Dr. Sangeorzan. In particular, researchers need to determine what comorbidities might drive patients to one type of care vs. another, he said. “The suggestion that [patients receiving implants with two motion segments have better outcomes than those receiving implants with a one-motion segment] also deserves further study,” he added.
The research was supported by the UK National Institute for Health and Care Research Health Technology Assessment Programme. The trial was sponsored by University College London. Dr. Goldberg disclosed grant support from NIHR HTA, as well as financial relationships with companies including Stryker, Paragon 28, and stock options with Standing CT Company, Elstree Waterfront Outpatients, and X Bolt Orthopedics.
The editorialists had no financial conflicts to disclose.
Ankle osteoarthritis remains a cause of severe pain and disability. Patients are treated nonoperatively if possible, but surgery is often needed for individuals with end-stage disease, wrote Andrew Goldberg, MBBS, of University College London and colleagues in the Annals of Internal Medicine.
“Most patients with ankle arthritis respond to nonoperative treatments, such as weight loss, activity modification, support braces, and analgesia, [but] once the disease has progressed to end-stage osteoarthritis, the main surgical treatments are total ankle re-placement or ankle arthrodesis,” Dr. Goldberg said, in an interview.
In the new study, patients were randomized to receive either a total ankle replacement (TAR) or ankle fusion (AF).
“We showed that, in both treatment groups the clinical scores improved hugely, by more than three times the minimal clinically important difference,” Dr. Goldberg said in an interview.
“Although the ankle replacement arm improved, on average, by more than an extra 4 points over ankle fusion, this was not considered clinically or statistically significant,” he said.
The study is the first randomized trial to show high-quality and robust results, he noted, and findings support data from previous studies.
“Although both TAR and ankle fusion have been shown to be effective, they are very different treatments, with one fusing the bones so that there is no ankle joint movement, and the other replacing the joint with the aim of retaining ankle joint movement. It is difficult for a patient to know which treatment is more suitable for them, with most seeking guidance from their surgeon,” he said.
Generating high-quality evidence
The study, a randomized, multicenter, open-label trial known as TARVA (Total Ankle Replacement Versus Ankle Arthrodesis), aimed to compare the clinical effectiveness of the two existing publicly funded U.K. treatment options, the authors wrote.
Patients were recruited at 17 U.K. centers between March 6, 2015, and Jan. 10, 2019. The study enrolled 303 adults aged 50-85 years with end-stage ankle osteoarthritis. The mean age of the participants was 68 years; 71% were men. A total of 137 TAR patients and 144 ankle fusion patients completed their surgeries with clinical scores available for analysis. Baseline characteristics were mainly similar between the groups.
Blinding was not possible because of the nature of the procedures, but the surgeons who screened the patients were not aware of the randomization allocations, the researchers noted. A total of 33 surgeons participated in the trial, with a median number of seven patients per surgeon during the study period.
For TAR, U.K. surgeons use both two-component, fixed-bearing and three-component, mobile-bearing implants, the authors write. Ankle fusion was done using the surgeon’s usual technique of either arthroscopic-assisted or open ankle fusion.
The primary outcome was the change in the Manchester–Oxford Foot Questionnaire walking/standing (MOXFQ-W/S) domain scores from baseline to 52 weeks after surgery. The MOXFQ-W/S uses a scale of 0-100, with lower scores representing better outcomes. Secondary outcomes included change in the MOXFQ-W/S scores at 26 weeks after surgery, as well as measures of patient quality of life.
No statistically significant difference
Overall, the mean MOXFQ-W/S scores improved significantly from baseline to 52 weeks for both groups, with average improvements of 49.9 in the TAR group and 44.4 points in the AF group. The average scores at 52 weeks were 31.4 in the TAR group and 36.8 in the AF group.
The adjusted difference in score change from baseline was –5.56, showing a slightly greater degree of improvement with TAR, but this difference was not clinically or statistically significant, the researchers noted.
Adverse event numbers were similar for both procedures, with 54% of TAR patients and 53% of AF patients experiencing at least 1 adverse event during the study period. Of those, 18% of TAR patients and 24% of AF patients experienced at least 1 serious adverse event.
However, the TAR patients experienced a higher rate of wound healing complications and nerve injuries, while thromboembolism was higher in the AF patients, the researchers noted.
A prespecified subgroup analysis of patients with osteoarthritis in adjacent joints suggested a greater improvement in TAR, compared with AF, a difference that increased when fixed-bearing TAR was compared with AF, the authors wrote.
“This reinforces previous reports that suggest that the presence of adjacent joint arthritis may be an indication for ankle replacement over AF,” the authors wrote in their discussion.
“Many of these patients did not have any symptoms in the adjacent joints,” they noted.
“The presence of adjacent joint arthritis, meaning the wear and tear of the joints around the ankle joint, seemed to favor ankle replacement,” Dr. Goldberg said. Approximately 30 joints in the foot continue to move after the ankle is fused, and if these adjacent joints are not healthy before surgery [as was the case in 42% of the study patients], the results of fusion were less successful, he explained.
A post hoc analysis between TAR subtypes showed that patients who had fixed-bearing TAR had significantly greater improvements, compared with AF patients, but this difference was not observed in patients who had mobile-bearing TAR, the researchers noted.
Dr. Goldberg said it was surprising “that, in a separate analysis, we found that the fixed-bearing ankle replacement patients [who accounted for half of the implants used] improved by a much greater difference when compared to ankle fusion.”
The study findings were limited by several factors including the short follow-up and study design that allowed surgeons to choose any implant and technique, the researchers noted.
Other limitations include a lack of data on cost-effectiveness and the impact of comorbidities on outcomes, they wrote. However, the study is the first completed multicenter randomized controlled trial to compare TAR and AF procedures for end-stage ankle osteoarthritis and shows that both yield similar clinical improvements, they concluded.
Data can inform treatment discussion
The take-home messages for clinicians are that both ankle replacement and ankle fusion are effective treatments that improve patients’ quality of life, and it is important to establish the health of adjacent joints before making treatment recommendations, Dr. Goldberg said.
“Careful counseling on the relative risks of each procedure should be part of the informed consent process,” he added. Ideally, all patients seeking surgical care for ankle arthritis should have a choice between ankle replacement and ankle fusion, but sometimes there is inequity of provision of the two treatments, he noted.
“We now encourage all surgeons to work in ankle arthritis networks so that every patient, no matter where they live, can have choice about the best treatment for them,” he said.
Researchers met the challenge of surgical RCT
Randomized trials of surgical interventions are challenging to conduct, and therefore limited, wrote Bruce Sangeorzan, MD, of the University of Washington, Seattle, and colleagues in an accompanying editorial. However, the new study was strengthened by the inclusion of 17 centers for heterogeneity of implant type and surgeon experience level, the editorialists said in the Annals of Internal Medicine.
The study is especially important, because ankle arthritis treatment is very understudied, compared with hip and knee arthritis, but it has a similar impact on activity, editorial coauthor Dr. Sangeorzan said in an interview.
“Randomized controlled trials are the gold standard for comparing medical therapies,” he said, “but they are very difficult to do in surgical treatments, particularly when the two treatments can be differentiated, in this case by movement of the ankle.”
In addition, there is a strong placebo effect attached to interventions, Dr. Sangeorzan noted. “Determining best-case treatment relies on prospective research, preferably randomized. Since both ankle fusion and ankle replacement are effective therapies, a prospective randomized trial is the best way to help make treatment decisions,” he said.
The current study findings are not surprising, but they are preliminary, and 1 year of follow-up is not enough to determine effectiveness, Dr. Sangeorzan emphasized. However, “the authors have done the hard work of randomizing the patients and collecting the data, and the patients can now be followed for a longer time,” he said.
“In addition, the trial was designed with multiple secondary outcome measures, so the data can be matched up with larger trials that were not randomized to identify key elements of success for each procedure,” he noted.
The key message for clinicians is that ankle arthritis has a significant impact on patients’ lives, but there are two effective treatments that can reduce the impact of the disease, said Dr. Sangeorzan. “The data suggest that there are differences in implant design and differences in comorbidities that should influence decision-making,” he added.
Additional research is needed in the form of a longer study duration with larger cohorts, said Dr. Sangeorzan. In particular, researchers need to determine what comorbidities might drive patients to one type of care vs. another, he said. “The suggestion that [patients receiving implants with two motion segments have better outcomes than those receiving implants with a one-motion segment] also deserves further study,” he added.
The research was supported by the UK National Institute for Health and Care Research Health Technology Assessment Programme. The trial was sponsored by University College London. Dr. Goldberg disclosed grant support from NIHR HTA, as well as financial relationships with companies including Stryker, Paragon 28, and stock options with Standing CT Company, Elstree Waterfront Outpatients, and X Bolt Orthopedics.
The editorialists had no financial conflicts to disclose.
New CDC guidance on prescribing opioids for pain
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.
The 2022 Clinical Practice Guideline provides guidance on determining whether to initiate opioids for pain; selecting opioids and determining opioid dosages; deciding duration of initial opioid prescription and conducting follow-up; and assessing risk and addressing potential harms of opioid use.
“Patients with pain should receive compassionate, safe, and effective pain care. We want clinicians and patients to have the information they need to weigh the benefits of different approaches to pain care, with the goal of helping people reduce their pain and improve their quality of life,” Christopher M. Jones, PharmD, DrPH, acting director for the CDC’s National Center for Injury Prevention and Control, said in a news release.
How to taper safely
The last guideline on the topic was released by CDC in 2016. Since then, new evidence has emerged regarding the benefits and risks of prescription opioids for acute and chronic pain, comparisons with nonopioid pain treatments, dosing strategies, opioid dose-dependent effects, risk mitigation strategies, and opioid tapering and discontinuation, the CDC says.
A “critical” addition to the 2022 guideline is advice on tapering opioids, Dr. Jones said during a press briefing.
“Practical tips on how to taper in an individualized patient-centered manner have been added to help clinicians if the decision is made to taper opioids, and the guideline explicitly advises against abrupt discontinuation or rapid dose reductions of opioids,” Dr. Jones said.
“That is based on lessons learned over the last several years as well as new science about how we approach tapering and the real harms that can result when patients are abruptly discontinued or rapidly tapered,” he added.
The updated guideline was published online Nov. 3 in the Morbidity and Mortality Weekly Report.
Key recommendations in the 100-page document include the following:
- In determining whether or not to initiate opioids, nonopioid therapies are at least as effective as opioids for many common types of acute pain. Use of nondrug and nonopioid drug therapies should be maximized as appropriate, and opioid therapy should only be considered for acute pain if it is anticipated that benefits outweigh risks to the patient.
- Before starting opioid therapy, providers should discuss with patients the realistic benefits and known risks of opioid therapy.
- Before starting ongoing opioid therapy for patients with subacute pain lasting 1 to 3 months or chronic pain lasting more than 3 months, providers should work with patients to establish treatment goals for pain and function, and consideration should be given as to how opioid therapy will be discontinued if benefits do not outweigh risks.
- Once opioids are started, the lowest effective dose of immediate-release opioids should be prescribed for no longer than needed for the expected duration of pain severe enough to require opioids.
- Within 1 to 4 weeks of starting opioid therapy for subacute or chronic pain, providers should work with patients to evaluate and carefully weigh benefits and risks of continuing opioid therapy; care should be exercised when increasing, continuing, or reducing opioid dosage.
- Before starting and periodically during ongoing opioid therapy, providers should evaluate risk for opioid-related harms and should work with patients to incorporate relevant strategies to mitigate risk, including offering naloxone and reviewing potential interactions with any other prescribed medications or substance used.
- Abrupt discontinuation of opioids should be avoided, especially for patients receiving high doses.
- For treating patients with opioid use disorder, treatment with evidence-based medications should be provided, or arrangements for such treatment should be made.
Dr. Jones emphasized that the guideline is “voluntary and meant to guide shared decision-making between a clinician and patient. It’s not meant to be implemented as absolute limits of policy or practice by clinicians, health systems, insurance companies, governmental entities.”
He also noted that the “current state of the overdose crisis, which is very much driven by illicit synthetic opioids, is not the aim of this guideline.
“The release of this guideline is really about advancing pain care and improving the lives of patients living with pain,” he said.
“We know that at least 1 in 5 people in the country have chronic pain. It’s one of the most common reasons why people present to their health care provider, and the goal here is to advance pain care, function, and quality of life for that patient population, while also reducing misuse, diversion, and consequences of prescription opioid misuse,” Dr. Jones added.
A version of this article first appeared on Medscape.com.
Gout too often treated only in emergency department
Only about one in three patients seen in the emergency department of an academic health system for acute gout had a follow-up visit that addressed this condition, Lesley Jackson, MD, of the University of Alabama at Birmingham, reported at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Dr. Jackson presented research done on patients seen within her university’s health system, looking at 72 patients seen in the ED between September 2021 and February 2022. Medications prescribed at discharge from the ED included corticosteroids (46 patients, or 64%), opioids (45 patients, 63%), NSAIDs (31 patients, 43%), and colchicine (23 patients, 32%).
Only 26 patients, or about 36%, had a subsequent outpatient visit in the UAB health system addressing gout, she said. Of 33 patients with any outpatient follow-up visit within the UAB system, 21 were within 1 month after the index ED visit, followed by 3 more prior to 3 months, and 9 more after 3 months.
The limitations of the study includes its collection of data from a single institution. But the results highlight the need for improved quality of care for gout, with too many people being treated for this condition primarily in the ED, she said.
In an email exchange arranged by the Arthritis Foundation, Herbert S. B. Baraf, MD, said he agreed that patients too often limit their treatment for gout to seeking care for acute attacks in the ED.
Because of competing demands, physicians working there are more to take a “Band-Aid” approach and not impress upon patients that gout is a lifelong condition that needs follow-up and monitoring, said Dr. Baraf, clinical professor of medicine at George Washington University, Washington, and an associate clinical professor at the University of Maryland, Baltimore. He retired from private practice in 2022.
“This problem is akin to the patient who has a hip fracture due to osteoporosis who gets a surgical repair but is never referred for osteoporotic management,” wrote Dr. Baraf, who is a former board member of the Arthritis Foundation.
He suggested viewing gout as a form of arthritis that has two components.
“The first, that which brings the patient to seek medical care, is the often exquisitely painful attack of pain and swelling in a joint or joints that comes on acutely,” he wrote. “Calming these attacks are the focus of the patient and the doctor, who does the evaluation as relief of pain and inflammation is the most pressing task at hand.”
But equally important is the second element, addressing the cause of these flare ups of arthritis, he wrote. Elevated uric acid leads to crystalline deposits of urate in the joints, particularly in the feet, ankles, knees, and hands. Over time, these deposits generate seemingly random flare ups of acute joint pain in one or more of these areas.
“Thus, when a patient presents to an emergency room with a first or second attack of gout, pain relief is the primary focus of the visit,” Dr. Baraf wrote. “But if over time that is the only focus, and the elevation of serum uric acid is not addressed, deposits will continue to mount and flare ups will occur with increasing frequency and severity.”
This study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Jackson has no relevant financial disclosures.
Only about one in three patients seen in the emergency department of an academic health system for acute gout had a follow-up visit that addressed this condition, Lesley Jackson, MD, of the University of Alabama at Birmingham, reported at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Dr. Jackson presented research done on patients seen within her university’s health system, looking at 72 patients seen in the ED between September 2021 and February 2022. Medications prescribed at discharge from the ED included corticosteroids (46 patients, or 64%), opioids (45 patients, 63%), NSAIDs (31 patients, 43%), and colchicine (23 patients, 32%).
Only 26 patients, or about 36%, had a subsequent outpatient visit in the UAB health system addressing gout, she said. Of 33 patients with any outpatient follow-up visit within the UAB system, 21 were within 1 month after the index ED visit, followed by 3 more prior to 3 months, and 9 more after 3 months.
The limitations of the study includes its collection of data from a single institution. But the results highlight the need for improved quality of care for gout, with too many people being treated for this condition primarily in the ED, she said.
In an email exchange arranged by the Arthritis Foundation, Herbert S. B. Baraf, MD, said he agreed that patients too often limit their treatment for gout to seeking care for acute attacks in the ED.
Because of competing demands, physicians working there are more to take a “Band-Aid” approach and not impress upon patients that gout is a lifelong condition that needs follow-up and monitoring, said Dr. Baraf, clinical professor of medicine at George Washington University, Washington, and an associate clinical professor at the University of Maryland, Baltimore. He retired from private practice in 2022.
“This problem is akin to the patient who has a hip fracture due to osteoporosis who gets a surgical repair but is never referred for osteoporotic management,” wrote Dr. Baraf, who is a former board member of the Arthritis Foundation.
He suggested viewing gout as a form of arthritis that has two components.
“The first, that which brings the patient to seek medical care, is the often exquisitely painful attack of pain and swelling in a joint or joints that comes on acutely,” he wrote. “Calming these attacks are the focus of the patient and the doctor, who does the evaluation as relief of pain and inflammation is the most pressing task at hand.”
But equally important is the second element, addressing the cause of these flare ups of arthritis, he wrote. Elevated uric acid leads to crystalline deposits of urate in the joints, particularly in the feet, ankles, knees, and hands. Over time, these deposits generate seemingly random flare ups of acute joint pain in one or more of these areas.
“Thus, when a patient presents to an emergency room with a first or second attack of gout, pain relief is the primary focus of the visit,” Dr. Baraf wrote. “But if over time that is the only focus, and the elevation of serum uric acid is not addressed, deposits will continue to mount and flare ups will occur with increasing frequency and severity.”
This study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Jackson has no relevant financial disclosures.
Only about one in three patients seen in the emergency department of an academic health system for acute gout had a follow-up visit that addressed this condition, Lesley Jackson, MD, of the University of Alabama at Birmingham, reported at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Dr. Jackson presented research done on patients seen within her university’s health system, looking at 72 patients seen in the ED between September 2021 and February 2022. Medications prescribed at discharge from the ED included corticosteroids (46 patients, or 64%), opioids (45 patients, 63%), NSAIDs (31 patients, 43%), and colchicine (23 patients, 32%).
Only 26 patients, or about 36%, had a subsequent outpatient visit in the UAB health system addressing gout, she said. Of 33 patients with any outpatient follow-up visit within the UAB system, 21 were within 1 month after the index ED visit, followed by 3 more prior to 3 months, and 9 more after 3 months.
The limitations of the study includes its collection of data from a single institution. But the results highlight the need for improved quality of care for gout, with too many people being treated for this condition primarily in the ED, she said.
In an email exchange arranged by the Arthritis Foundation, Herbert S. B. Baraf, MD, said he agreed that patients too often limit their treatment for gout to seeking care for acute attacks in the ED.
Because of competing demands, physicians working there are more to take a “Band-Aid” approach and not impress upon patients that gout is a lifelong condition that needs follow-up and monitoring, said Dr. Baraf, clinical professor of medicine at George Washington University, Washington, and an associate clinical professor at the University of Maryland, Baltimore. He retired from private practice in 2022.
“This problem is akin to the patient who has a hip fracture due to osteoporosis who gets a surgical repair but is never referred for osteoporotic management,” wrote Dr. Baraf, who is a former board member of the Arthritis Foundation.
He suggested viewing gout as a form of arthritis that has two components.
“The first, that which brings the patient to seek medical care, is the often exquisitely painful attack of pain and swelling in a joint or joints that comes on acutely,” he wrote. “Calming these attacks are the focus of the patient and the doctor, who does the evaluation as relief of pain and inflammation is the most pressing task at hand.”
But equally important is the second element, addressing the cause of these flare ups of arthritis, he wrote. Elevated uric acid leads to crystalline deposits of urate in the joints, particularly in the feet, ankles, knees, and hands. Over time, these deposits generate seemingly random flare ups of acute joint pain in one or more of these areas.
“Thus, when a patient presents to an emergency room with a first or second attack of gout, pain relief is the primary focus of the visit,” Dr. Baraf wrote. “But if over time that is the only focus, and the elevation of serum uric acid is not addressed, deposits will continue to mount and flare ups will occur with increasing frequency and severity.”
This study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Jackson has no relevant financial disclosures.
FROM G-CAN 2022
Pain in Cancer Survivors: Assess, Monitor, and Ask for Help
SAN DIEGO—As patients with cancer live longer, pain is going to become an even bigger challenge for clinicians, a palliative care specialist told cancer specialists in a presentation at the annual meeting of the Association of VA Hematology/Oncology (AVAHO) in September, and decisions about treatment are becoming more complicated amid the opioid epidemic.
Fortunately, guidelines and clinical experience offer helpful insight into the best practices, said hematologist/oncologist Andrea Ruskin, MD, medical director of palliative care at Veterans Administration (VA) Connecticut Healthcare System (VACHS).
As Ruskin pointed out, two-thirds of newly diagnosed cancer patients are living for at least 5 years, “but with this progress comes challenges.” More than one-third (37%) of cancer survivors report cancer pain, 21% have noncancer pain, and 45% have both. About 5% to 8% of VA cancer survivors use opioids for the long term, she said, although there have been few studies in this population.
Among patients with head and neck cancer, specifically, chronic pain affects 45%, and severe pain affects 11%. Subclinical PTSD, depression, anxiety, and low quality of life are common in this population. “We may cure them, but they have a lot of issues going forward.”
One key strategy is to perform a comprehensive pain assessment at the first visit, and then address pain at every subsequent visit. She recommended a physician resource from the American Society of Clinical Oncology, and a template may be useful to provide helpful questions, Ruskin said.
At VACHS certain questions are routine. “Is pain interfering with your function? Sometimes people say it’s always a 10, but it’s not affecting function at all. Ask if the medicine is working. And how are they taking it? Sometimes they say, ‘I’m taking that for sleep,’ and we say ‘No, Mr. Smith, that is not a sleep medication.’”
Be aware that some patients may use nonmedical opioids, she said. And set expectations early on. “Safe opioid use starts with the very first prescription,” she said. “If I have somebody with myeloma or head and neck cancer, I make it very clear that my goal is that we want you off the opioids after the radiation or once the disease is in remission. I really make an effort at the very beginning to make sure that we're all on the same page.”
As you continue to see a patient, consider ordering urine tests, she said, not as a punitive measure but to make sure you’re offering the safest and most effective treatment. “We don’t do it to say ‘no, no, no.’ We do it for safety and to make sure they’re not getting meds elsewhere.”
What are the best practices when pain doesn’t go away? Should they stay on opioids? According to Ruskin, few evidence-based guidelines address the “more nuanced care” that patients need when their pain lasts for months or years.
But there are useful resources. Ruskin highlighted the National Comprehensive Cancer Network’s survivorship guidelines, and she summarized a few of the available painkiller options. “Opioids are great, and adjuvants are so-so. They work in some people, but we definitely have room for improvement.”
What if patients have persistent opioid use after cancer recovery? “I try to taper if I can, and I try to explain why I’m tapering. It could take months or years to taper patients,” she said. And consider transitioning the patient to buprenorphine, a drug that treats both pain and opioid use disorder, if appropriate. “You don’t need a waiver if you use it for pain. It’s definitely something we’re using more of.”
One important step is to bring in colleagues to help. Psychologists, chiropractors, physical therapists, physiatrists, and pain pharmacists can all be helpful, she said. “Learn about your VA resources and who can partner with you to help these complicated patients. They’re all at your fingertips.”
SAN DIEGO—As patients with cancer live longer, pain is going to become an even bigger challenge for clinicians, a palliative care specialist told cancer specialists in a presentation at the annual meeting of the Association of VA Hematology/Oncology (AVAHO) in September, and decisions about treatment are becoming more complicated amid the opioid epidemic.
Fortunately, guidelines and clinical experience offer helpful insight into the best practices, said hematologist/oncologist Andrea Ruskin, MD, medical director of palliative care at Veterans Administration (VA) Connecticut Healthcare System (VACHS).
As Ruskin pointed out, two-thirds of newly diagnosed cancer patients are living for at least 5 years, “but with this progress comes challenges.” More than one-third (37%) of cancer survivors report cancer pain, 21% have noncancer pain, and 45% have both. About 5% to 8% of VA cancer survivors use opioids for the long term, she said, although there have been few studies in this population.
Among patients with head and neck cancer, specifically, chronic pain affects 45%, and severe pain affects 11%. Subclinical PTSD, depression, anxiety, and low quality of life are common in this population. “We may cure them, but they have a lot of issues going forward.”
One key strategy is to perform a comprehensive pain assessment at the first visit, and then address pain at every subsequent visit. She recommended a physician resource from the American Society of Clinical Oncology, and a template may be useful to provide helpful questions, Ruskin said.
At VACHS certain questions are routine. “Is pain interfering with your function? Sometimes people say it’s always a 10, but it’s not affecting function at all. Ask if the medicine is working. And how are they taking it? Sometimes they say, ‘I’m taking that for sleep,’ and we say ‘No, Mr. Smith, that is not a sleep medication.’”
Be aware that some patients may use nonmedical opioids, she said. And set expectations early on. “Safe opioid use starts with the very first prescription,” she said. “If I have somebody with myeloma or head and neck cancer, I make it very clear that my goal is that we want you off the opioids after the radiation or once the disease is in remission. I really make an effort at the very beginning to make sure that we're all on the same page.”
As you continue to see a patient, consider ordering urine tests, she said, not as a punitive measure but to make sure you’re offering the safest and most effective treatment. “We don’t do it to say ‘no, no, no.’ We do it for safety and to make sure they’re not getting meds elsewhere.”
What are the best practices when pain doesn’t go away? Should they stay on opioids? According to Ruskin, few evidence-based guidelines address the “more nuanced care” that patients need when their pain lasts for months or years.
But there are useful resources. Ruskin highlighted the National Comprehensive Cancer Network’s survivorship guidelines, and she summarized a few of the available painkiller options. “Opioids are great, and adjuvants are so-so. They work in some people, but we definitely have room for improvement.”
What if patients have persistent opioid use after cancer recovery? “I try to taper if I can, and I try to explain why I’m tapering. It could take months or years to taper patients,” she said. And consider transitioning the patient to buprenorphine, a drug that treats both pain and opioid use disorder, if appropriate. “You don’t need a waiver if you use it for pain. It’s definitely something we’re using more of.”
One important step is to bring in colleagues to help. Psychologists, chiropractors, physical therapists, physiatrists, and pain pharmacists can all be helpful, she said. “Learn about your VA resources and who can partner with you to help these complicated patients. They’re all at your fingertips.”
SAN DIEGO—As patients with cancer live longer, pain is going to become an even bigger challenge for clinicians, a palliative care specialist told cancer specialists in a presentation at the annual meeting of the Association of VA Hematology/Oncology (AVAHO) in September, and decisions about treatment are becoming more complicated amid the opioid epidemic.
Fortunately, guidelines and clinical experience offer helpful insight into the best practices, said hematologist/oncologist Andrea Ruskin, MD, medical director of palliative care at Veterans Administration (VA) Connecticut Healthcare System (VACHS).
As Ruskin pointed out, two-thirds of newly diagnosed cancer patients are living for at least 5 years, “but with this progress comes challenges.” More than one-third (37%) of cancer survivors report cancer pain, 21% have noncancer pain, and 45% have both. About 5% to 8% of VA cancer survivors use opioids for the long term, she said, although there have been few studies in this population.
Among patients with head and neck cancer, specifically, chronic pain affects 45%, and severe pain affects 11%. Subclinical PTSD, depression, anxiety, and low quality of life are common in this population. “We may cure them, but they have a lot of issues going forward.”
One key strategy is to perform a comprehensive pain assessment at the first visit, and then address pain at every subsequent visit. She recommended a physician resource from the American Society of Clinical Oncology, and a template may be useful to provide helpful questions, Ruskin said.
At VACHS certain questions are routine. “Is pain interfering with your function? Sometimes people say it’s always a 10, but it’s not affecting function at all. Ask if the medicine is working. And how are they taking it? Sometimes they say, ‘I’m taking that for sleep,’ and we say ‘No, Mr. Smith, that is not a sleep medication.’”
Be aware that some patients may use nonmedical opioids, she said. And set expectations early on. “Safe opioid use starts with the very first prescription,” she said. “If I have somebody with myeloma or head and neck cancer, I make it very clear that my goal is that we want you off the opioids after the radiation or once the disease is in remission. I really make an effort at the very beginning to make sure that we're all on the same page.”
As you continue to see a patient, consider ordering urine tests, she said, not as a punitive measure but to make sure you’re offering the safest and most effective treatment. “We don’t do it to say ‘no, no, no.’ We do it for safety and to make sure they’re not getting meds elsewhere.”
What are the best practices when pain doesn’t go away? Should they stay on opioids? According to Ruskin, few evidence-based guidelines address the “more nuanced care” that patients need when their pain lasts for months or years.
But there are useful resources. Ruskin highlighted the National Comprehensive Cancer Network’s survivorship guidelines, and she summarized a few of the available painkiller options. “Opioids are great, and adjuvants are so-so. They work in some people, but we definitely have room for improvement.”
What if patients have persistent opioid use after cancer recovery? “I try to taper if I can, and I try to explain why I’m tapering. It could take months or years to taper patients,” she said. And consider transitioning the patient to buprenorphine, a drug that treats both pain and opioid use disorder, if appropriate. “You don’t need a waiver if you use it for pain. It’s definitely something we’re using more of.”
One important step is to bring in colleagues to help. Psychologists, chiropractors, physical therapists, physiatrists, and pain pharmacists can all be helpful, she said. “Learn about your VA resources and who can partner with you to help these complicated patients. They’re all at your fingertips.”
Psychotropic medications for chronic pain
The opioid crisis presents a need to consider alternative options for treating chronic pain. There is significant overlap in neuroanatomical circuits that process pain, emotions, and motivation. Neurotransmitters modulated by psychotropic medications are also involved in regulating the pain pathways.1,2 In light of this, psychotropics can be considered for treating chronic pain in certain patients. The Table1-3 outlines various uses and adverse effects of select psychotropic medications used to treat pain, as well as their psychiatric uses.
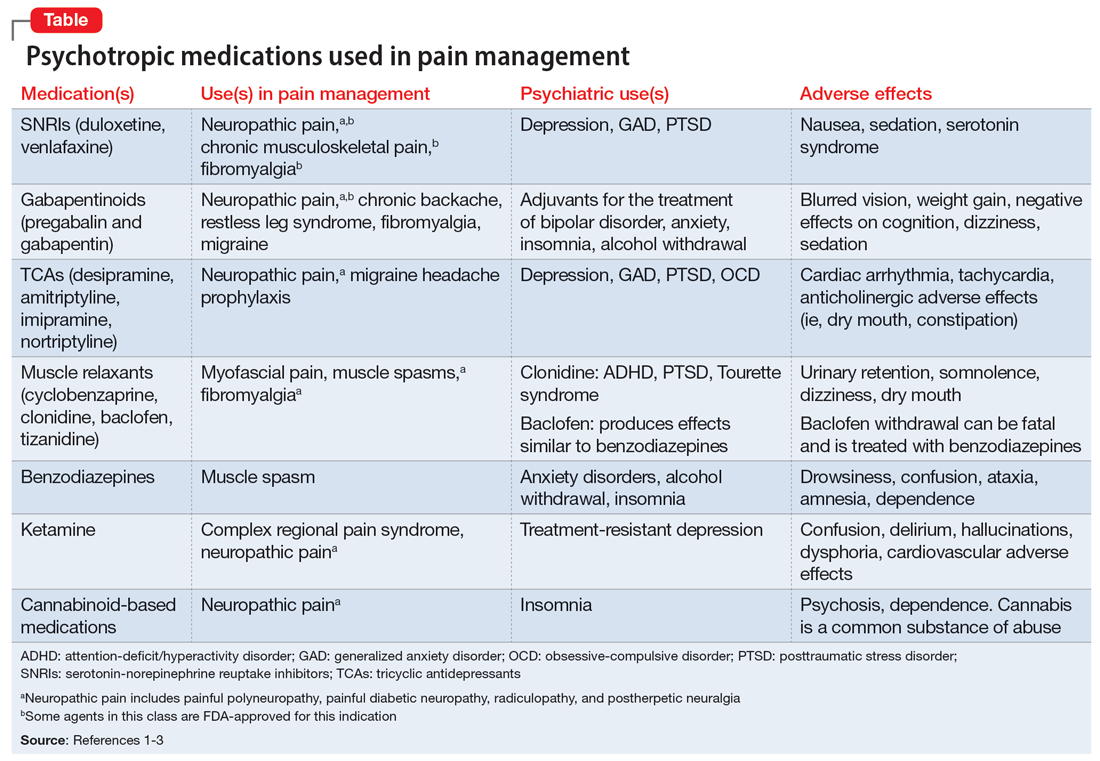
In addition to its psychiatric indications, the serotonin-norepinephrine reuptake inhibitor duloxetine is FDA-approved for treating fibromyalgia and diabetic neuropathic pain. It is often prescribed in the treatment of multiple pain disorders. Tricyclic antidepressants (TCAs) have the largest effect size in the treatment of neuropathic pain.2 Cyclobenzaprine is a TCA used to treat muscle spasms. Gabapentinoids (alpha-2 delta-1 calcium channel inhibition) are FDA-approved for treating postherpetic neuralgia, fibromyalgia, and diabetic neuropathy.1,2
Ketamine is an anesthetic with analgesic and antidepressant properties used as an IV infusion to manage several pain disorders.2 The alpha-2 adrenergic agonists tizanidine and clonidine are muscle relaxants2; the latter is used to treat attention-deficit/hyperactivity disorder and Tourette syndrome. Benzodiazepines (GABA-A agonists) are used for short-term treatment of anxiety disorders, insomnia, and muscle spasms.1,2 Baclofen (GABA-B receptor agonist) is used to treat spasticity.2 Medical cannabis (tetrahydrocannabinol/cannabidiol) is also gaining popularity for treating chronic pain and insomnia.1-3
1. Sutherland AM, Nicholls J, Bao J, et al. Overlaps in pharmacology for the treatment of chronic pain and mental health disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;87(Pt B):290-297.
2. Bajwa ZH, Wootton RJ, Warfield CA. Principles and Practice of Pain Medicine. 3rd ed. McGraw Hill; 2016.
3. McDonagh MS, Selph SS, Buckley DI, et al. Nonopioid Pharmacologic Treatments for Chronic Pain. Comparative Effectiveness Review No. 228. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER228
The opioid crisis presents a need to consider alternative options for treating chronic pain. There is significant overlap in neuroanatomical circuits that process pain, emotions, and motivation. Neurotransmitters modulated by psychotropic medications are also involved in regulating the pain pathways.1,2 In light of this, psychotropics can be considered for treating chronic pain in certain patients. The Table1-3 outlines various uses and adverse effects of select psychotropic medications used to treat pain, as well as their psychiatric uses.

In addition to its psychiatric indications, the serotonin-norepinephrine reuptake inhibitor duloxetine is FDA-approved for treating fibromyalgia and diabetic neuropathic pain. It is often prescribed in the treatment of multiple pain disorders. Tricyclic antidepressants (TCAs) have the largest effect size in the treatment of neuropathic pain.2 Cyclobenzaprine is a TCA used to treat muscle spasms. Gabapentinoids (alpha-2 delta-1 calcium channel inhibition) are FDA-approved for treating postherpetic neuralgia, fibromyalgia, and diabetic neuropathy.1,2
Ketamine is an anesthetic with analgesic and antidepressant properties used as an IV infusion to manage several pain disorders.2 The alpha-2 adrenergic agonists tizanidine and clonidine are muscle relaxants2; the latter is used to treat attention-deficit/hyperactivity disorder and Tourette syndrome. Benzodiazepines (GABA-A agonists) are used for short-term treatment of anxiety disorders, insomnia, and muscle spasms.1,2 Baclofen (GABA-B receptor agonist) is used to treat spasticity.2 Medical cannabis (tetrahydrocannabinol/cannabidiol) is also gaining popularity for treating chronic pain and insomnia.1-3
The opioid crisis presents a need to consider alternative options for treating chronic pain. There is significant overlap in neuroanatomical circuits that process pain, emotions, and motivation. Neurotransmitters modulated by psychotropic medications are also involved in regulating the pain pathways.1,2 In light of this, psychotropics can be considered for treating chronic pain in certain patients. The Table1-3 outlines various uses and adverse effects of select psychotropic medications used to treat pain, as well as their psychiatric uses.

In addition to its psychiatric indications, the serotonin-norepinephrine reuptake inhibitor duloxetine is FDA-approved for treating fibromyalgia and diabetic neuropathic pain. It is often prescribed in the treatment of multiple pain disorders. Tricyclic antidepressants (TCAs) have the largest effect size in the treatment of neuropathic pain.2 Cyclobenzaprine is a TCA used to treat muscle spasms. Gabapentinoids (alpha-2 delta-1 calcium channel inhibition) are FDA-approved for treating postherpetic neuralgia, fibromyalgia, and diabetic neuropathy.1,2
Ketamine is an anesthetic with analgesic and antidepressant properties used as an IV infusion to manage several pain disorders.2 The alpha-2 adrenergic agonists tizanidine and clonidine are muscle relaxants2; the latter is used to treat attention-deficit/hyperactivity disorder and Tourette syndrome. Benzodiazepines (GABA-A agonists) are used for short-term treatment of anxiety disorders, insomnia, and muscle spasms.1,2 Baclofen (GABA-B receptor agonist) is used to treat spasticity.2 Medical cannabis (tetrahydrocannabinol/cannabidiol) is also gaining popularity for treating chronic pain and insomnia.1-3
1. Sutherland AM, Nicholls J, Bao J, et al. Overlaps in pharmacology for the treatment of chronic pain and mental health disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;87(Pt B):290-297.
2. Bajwa ZH, Wootton RJ, Warfield CA. Principles and Practice of Pain Medicine. 3rd ed. McGraw Hill; 2016.
3. McDonagh MS, Selph SS, Buckley DI, et al. Nonopioid Pharmacologic Treatments for Chronic Pain. Comparative Effectiveness Review No. 228. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER228
1. Sutherland AM, Nicholls J, Bao J, et al. Overlaps in pharmacology for the treatment of chronic pain and mental health disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2018;87(Pt B):290-297.
2. Bajwa ZH, Wootton RJ, Warfield CA. Principles and Practice of Pain Medicine. 3rd ed. McGraw Hill; 2016.
3. McDonagh MS, Selph SS, Buckley DI, et al. Nonopioid Pharmacologic Treatments for Chronic Pain. Comparative Effectiveness Review No. 228. Agency for Healthcare Research and Quality; 2020. doi:10.23970/AHRQEPCCER228
The latest migraine therapies – some you might not know about
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr. Matthew Watto, here with my very good friend, Dr. Paul Williams. It’s time to talk about headaches. We did a great recent podcast on migraines, Headache Update: Making Migraines Less Painful with Dr. Kevin Weber. One of the quotes from that episode that stayed with me was when he said, “I tell my patients to think about migraine as an irritable old miser set in their ways, and your brain is set in its ways. It doesn’t like changes in routine. It doesn’t like lack of sleep, it doesn’t like being hungry, it doesn’t like being thirsty, and it doesn’t like changes in the weather.” That’s a reminder of the good, old-fashioned primary care tips for taking care of headache.
Paul N. Williams, MD: That’s right. Conservative supportive management goes by the wayside because we focus on the medications. But I thought that was a really nice way to start the episode.
Dr. Watto: I asked him about cervicogenic headaches, which I guess you have to diagnose by giving a cervical steroid injection and see if the patient feels better, but he said he doesn’t do this. This is expert opinion territory. He asks his patients with chronic headache about cervical neck pain, because if they have it, he goes after it with physical therapy, which can help with the headaches. I thought that was a great pearl that I hadn’t heard before.
Give the audience a pearl from this great episode.
Dr. Williams: We talked about foundational treatments. We reviewed some of the abortive therapies and over-the-counter products. Some patients do quite well with acetaminophen or NSAIDs. We also talked about triptans, which are the standard medicines that we all know about. You can use those in combination, by the way. Patients can take their triptan with the NSAID that works best for them. They don’t have to be used one at a time, trying one and then trying the other one if the first one doesn’t help. Dr. Weber gave us practical guides in terms of which triptans he favors. He mentioned rizatriptan and naratriptan, which is one that I had not used with any frequency. I’ve seen rizatriptan a fair amount and that one seems to be covered by most insurances. He favors those two triptans.
He also reminded us that even though there is theoretical concern for serotonin toxicity because these are serotonergic and you’ll see these scary pop-ups in your electronic health record, that concern is almost purely theoretical. It hasn’t been borne out. They are really safe medications to use. But do use caution if you have a patient with known cardiovascular disease or cerebrovascular disease. We spent a fair amount of time talking about chest pressure as a common side effect. We also talked about some of the newer agents.
Dr. Watto: I wanted to add something about the triptans. Part of the reason he favors rizatriptan and naratriptan is that they are newer. He thinks they tend to have fewer side effects. But he did mention sumatriptan because it comes in the most different formulations. If patients have severe nausea, there is a subcutaneous version of sumatriptan and also an intranasal version.
The new kids on the block are the CGRP receptor antagonists, and they are available for preventive and abortive therapy. The abortive therapies are probably what people will be seeing most often in primary care – ubrogepant and rimegepant. Patients can take ubrogepant for abortive therapy and then repeat it if necessary. That’s similar to what patients are used to with the triptans. Rimegepant is taken once daily for abortive therapy or every other day as a preventive agent. Those are two of the agents that you might see patients taking. I’ve certainly started to see them.
There are also a whole bunch of monoclonal antibodies that affect the CGRP pathway. Those are given either once a month by subcutaneous injection or once every 3 months, and one is an infusion. They are pretty safe, and the big appeal is that they can be used in patients with cardiovascular disease. He also said that he has some patients who take them because triptans can cause the medication overuse side effect, but the CGRP receptor antagonists don’t. It’s an option for some patients to take the CGRP receptor antagonists on certain days for abortive therapy and then they can take the triptans the rest of the month.
Dr. Weber said that in his practice, these new drugs have really been great, which I can imagine, if you’re a specialist, patients have exhausted many of the typical therapies we offer in primary care.
Paul, bring us home here. What else should we tell the audience about? In primary care, what can we offer these patients?
Dr. Williams: A lot of the stuff we can offer works, by the way. It’s exciting to have fancy new medications to use, but you don’t even necessarily need to get to that point. We have a lot of medications that we can use for migraine prophylaxis, such as the beta-blockers and antihypertensives. Candesartan was a new one to me, an angiotensin receptor blocker that apparently has good evidence for migraine prophylaxis and Dr. Weber swears by it. We talked about some of the antiseizure medications, such as topiramate, which is probably the one with the most comfort in primary care. Some older folks may be using valproic acid or the tricyclic antidepressants (amitriptyline and nortriptyline) because people with migraine often will have comorbid anxiety or trouble sleeping, so I find that can sometimes be an effective medication or if they have comorbid neuropathic pain.
Another one that was new to me was venlafaxine as migraine prophylaxis. It’s not something I’d heard about before this episode. Certainly, for someone with chronic pain or a mood disorder that’s comorbid with migraines, it may be worth a shot. So there are options that we can exhaust first, and we may actually be doing our specialist friends a favor by trying one or two of these in advance, because then by the time the patient gets to the neurologist, it makes the prior authorization process much easier for the newer, fancier-pants medications that we’re all very excited about.
Dr. Watto: Paul, we’ve teased this fantastic podcast episode filled with so much more great stuff, so people should check out Headache Update: Making Migraines Less Painful with Dr. Kevin Weber.
Until next time, this has been another episode of The Curbsiders, bringing you a little knowledge food for your brain hole.
The Curbsiders is an internal medicine podcast, in which three board-certified internists interview experts on clinically important topics. In a collaboration with Medscape, the Curbsiders share clinical pearls and practice-changing knowledge from selected podcasts.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr. Matthew Watto, here with my very good friend, Dr. Paul Williams. It’s time to talk about headaches. We did a great recent podcast on migraines, Headache Update: Making Migraines Less Painful with Dr. Kevin Weber. One of the quotes from that episode that stayed with me was when he said, “I tell my patients to think about migraine as an irritable old miser set in their ways, and your brain is set in its ways. It doesn’t like changes in routine. It doesn’t like lack of sleep, it doesn’t like being hungry, it doesn’t like being thirsty, and it doesn’t like changes in the weather.” That’s a reminder of the good, old-fashioned primary care tips for taking care of headache.
Paul N. Williams, MD: That’s right. Conservative supportive management goes by the wayside because we focus on the medications. But I thought that was a really nice way to start the episode.
Dr. Watto: I asked him about cervicogenic headaches, which I guess you have to diagnose by giving a cervical steroid injection and see if the patient feels better, but he said he doesn’t do this. This is expert opinion territory. He asks his patients with chronic headache about cervical neck pain, because if they have it, he goes after it with physical therapy, which can help with the headaches. I thought that was a great pearl that I hadn’t heard before.
Give the audience a pearl from this great episode.
Dr. Williams: We talked about foundational treatments. We reviewed some of the abortive therapies and over-the-counter products. Some patients do quite well with acetaminophen or NSAIDs. We also talked about triptans, which are the standard medicines that we all know about. You can use those in combination, by the way. Patients can take their triptan with the NSAID that works best for them. They don’t have to be used one at a time, trying one and then trying the other one if the first one doesn’t help. Dr. Weber gave us practical guides in terms of which triptans he favors. He mentioned rizatriptan and naratriptan, which is one that I had not used with any frequency. I’ve seen rizatriptan a fair amount and that one seems to be covered by most insurances. He favors those two triptans.
He also reminded us that even though there is theoretical concern for serotonin toxicity because these are serotonergic and you’ll see these scary pop-ups in your electronic health record, that concern is almost purely theoretical. It hasn’t been borne out. They are really safe medications to use. But do use caution if you have a patient with known cardiovascular disease or cerebrovascular disease. We spent a fair amount of time talking about chest pressure as a common side effect. We also talked about some of the newer agents.
Dr. Watto: I wanted to add something about the triptans. Part of the reason he favors rizatriptan and naratriptan is that they are newer. He thinks they tend to have fewer side effects. But he did mention sumatriptan because it comes in the most different formulations. If patients have severe nausea, there is a subcutaneous version of sumatriptan and also an intranasal version.
The new kids on the block are the CGRP receptor antagonists, and they are available for preventive and abortive therapy. The abortive therapies are probably what people will be seeing most often in primary care – ubrogepant and rimegepant. Patients can take ubrogepant for abortive therapy and then repeat it if necessary. That’s similar to what patients are used to with the triptans. Rimegepant is taken once daily for abortive therapy or every other day as a preventive agent. Those are two of the agents that you might see patients taking. I’ve certainly started to see them.
There are also a whole bunch of monoclonal antibodies that affect the CGRP pathway. Those are given either once a month by subcutaneous injection or once every 3 months, and one is an infusion. They are pretty safe, and the big appeal is that they can be used in patients with cardiovascular disease. He also said that he has some patients who take them because triptans can cause the medication overuse side effect, but the CGRP receptor antagonists don’t. It’s an option for some patients to take the CGRP receptor antagonists on certain days for abortive therapy and then they can take the triptans the rest of the month.
Dr. Weber said that in his practice, these new drugs have really been great, which I can imagine, if you’re a specialist, patients have exhausted many of the typical therapies we offer in primary care.
Paul, bring us home here. What else should we tell the audience about? In primary care, what can we offer these patients?
Dr. Williams: A lot of the stuff we can offer works, by the way. It’s exciting to have fancy new medications to use, but you don’t even necessarily need to get to that point. We have a lot of medications that we can use for migraine prophylaxis, such as the beta-blockers and antihypertensives. Candesartan was a new one to me, an angiotensin receptor blocker that apparently has good evidence for migraine prophylaxis and Dr. Weber swears by it. We talked about some of the antiseizure medications, such as topiramate, which is probably the one with the most comfort in primary care. Some older folks may be using valproic acid or the tricyclic antidepressants (amitriptyline and nortriptyline) because people with migraine often will have comorbid anxiety or trouble sleeping, so I find that can sometimes be an effective medication or if they have comorbid neuropathic pain.
Another one that was new to me was venlafaxine as migraine prophylaxis. It’s not something I’d heard about before this episode. Certainly, for someone with chronic pain or a mood disorder that’s comorbid with migraines, it may be worth a shot. So there are options that we can exhaust first, and we may actually be doing our specialist friends a favor by trying one or two of these in advance, because then by the time the patient gets to the neurologist, it makes the prior authorization process much easier for the newer, fancier-pants medications that we’re all very excited about.
Dr. Watto: Paul, we’ve teased this fantastic podcast episode filled with so much more great stuff, so people should check out Headache Update: Making Migraines Less Painful with Dr. Kevin Weber.
Until next time, this has been another episode of The Curbsiders, bringing you a little knowledge food for your brain hole.
The Curbsiders is an internal medicine podcast, in which three board-certified internists interview experts on clinically important topics. In a collaboration with Medscape, the Curbsiders share clinical pearls and practice-changing knowledge from selected podcasts.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr. Matthew Watto, here with my very good friend, Dr. Paul Williams. It’s time to talk about headaches. We did a great recent podcast on migraines, Headache Update: Making Migraines Less Painful with Dr. Kevin Weber. One of the quotes from that episode that stayed with me was when he said, “I tell my patients to think about migraine as an irritable old miser set in their ways, and your brain is set in its ways. It doesn’t like changes in routine. It doesn’t like lack of sleep, it doesn’t like being hungry, it doesn’t like being thirsty, and it doesn’t like changes in the weather.” That’s a reminder of the good, old-fashioned primary care tips for taking care of headache.
Paul N. Williams, MD: That’s right. Conservative supportive management goes by the wayside because we focus on the medications. But I thought that was a really nice way to start the episode.
Dr. Watto: I asked him about cervicogenic headaches, which I guess you have to diagnose by giving a cervical steroid injection and see if the patient feels better, but he said he doesn’t do this. This is expert opinion territory. He asks his patients with chronic headache about cervical neck pain, because if they have it, he goes after it with physical therapy, which can help with the headaches. I thought that was a great pearl that I hadn’t heard before.
Give the audience a pearl from this great episode.
Dr. Williams: We talked about foundational treatments. We reviewed some of the abortive therapies and over-the-counter products. Some patients do quite well with acetaminophen or NSAIDs. We also talked about triptans, which are the standard medicines that we all know about. You can use those in combination, by the way. Patients can take their triptan with the NSAID that works best for them. They don’t have to be used one at a time, trying one and then trying the other one if the first one doesn’t help. Dr. Weber gave us practical guides in terms of which triptans he favors. He mentioned rizatriptan and naratriptan, which is one that I had not used with any frequency. I’ve seen rizatriptan a fair amount and that one seems to be covered by most insurances. He favors those two triptans.
He also reminded us that even though there is theoretical concern for serotonin toxicity because these are serotonergic and you’ll see these scary pop-ups in your electronic health record, that concern is almost purely theoretical. It hasn’t been borne out. They are really safe medications to use. But do use caution if you have a patient with known cardiovascular disease or cerebrovascular disease. We spent a fair amount of time talking about chest pressure as a common side effect. We also talked about some of the newer agents.
Dr. Watto: I wanted to add something about the triptans. Part of the reason he favors rizatriptan and naratriptan is that they are newer. He thinks they tend to have fewer side effects. But he did mention sumatriptan because it comes in the most different formulations. If patients have severe nausea, there is a subcutaneous version of sumatriptan and also an intranasal version.
The new kids on the block are the CGRP receptor antagonists, and they are available for preventive and abortive therapy. The abortive therapies are probably what people will be seeing most often in primary care – ubrogepant and rimegepant. Patients can take ubrogepant for abortive therapy and then repeat it if necessary. That’s similar to what patients are used to with the triptans. Rimegepant is taken once daily for abortive therapy or every other day as a preventive agent. Those are two of the agents that you might see patients taking. I’ve certainly started to see them.
There are also a whole bunch of monoclonal antibodies that affect the CGRP pathway. Those are given either once a month by subcutaneous injection or once every 3 months, and one is an infusion. They are pretty safe, and the big appeal is that they can be used in patients with cardiovascular disease. He also said that he has some patients who take them because triptans can cause the medication overuse side effect, but the CGRP receptor antagonists don’t. It’s an option for some patients to take the CGRP receptor antagonists on certain days for abortive therapy and then they can take the triptans the rest of the month.
Dr. Weber said that in his practice, these new drugs have really been great, which I can imagine, if you’re a specialist, patients have exhausted many of the typical therapies we offer in primary care.
Paul, bring us home here. What else should we tell the audience about? In primary care, what can we offer these patients?
Dr. Williams: A lot of the stuff we can offer works, by the way. It’s exciting to have fancy new medications to use, but you don’t even necessarily need to get to that point. We have a lot of medications that we can use for migraine prophylaxis, such as the beta-blockers and antihypertensives. Candesartan was a new one to me, an angiotensin receptor blocker that apparently has good evidence for migraine prophylaxis and Dr. Weber swears by it. We talked about some of the antiseizure medications, such as topiramate, which is probably the one with the most comfort in primary care. Some older folks may be using valproic acid or the tricyclic antidepressants (amitriptyline and nortriptyline) because people with migraine often will have comorbid anxiety or trouble sleeping, so I find that can sometimes be an effective medication or if they have comorbid neuropathic pain.
Another one that was new to me was venlafaxine as migraine prophylaxis. It’s not something I’d heard about before this episode. Certainly, for someone with chronic pain or a mood disorder that’s comorbid with migraines, it may be worth a shot. So there are options that we can exhaust first, and we may actually be doing our specialist friends a favor by trying one or two of these in advance, because then by the time the patient gets to the neurologist, it makes the prior authorization process much easier for the newer, fancier-pants medications that we’re all very excited about.
Dr. Watto: Paul, we’ve teased this fantastic podcast episode filled with so much more great stuff, so people should check out Headache Update: Making Migraines Less Painful with Dr. Kevin Weber.
Until next time, this has been another episode of The Curbsiders, bringing you a little knowledge food for your brain hole.
The Curbsiders is an internal medicine podcast, in which three board-certified internists interview experts on clinically important topics. In a collaboration with Medscape, the Curbsiders share clinical pearls and practice-changing knowledge from selected podcasts.
A version of this article first appeared on Medscape.com.
Medications for Opioid Use Disorder Program in a VA Emergency Department
Opioid use disorder (OUD) is a public health crisis significantly affecting veterans. A substantial increase in veterans diagnosed with OUD has occurred, nearly tripling from 25,031 in 2003 to 69,142 in 2017
For patients with active OUD, medications for opioid use disorder (MOUD) reduce the risk of overdose and all-cause mortality.3 In 2009, the US Department of Veterans Affairs (VA) and Department of Defense (DoD) published clinical practice guidelines for substance use disorders that strongly recommended MOUD with either buprenorphine or methadone as a first-line treatment. In 2015 updated guidelines encouraged buprenorphine initiation in primary care settings.4,5 This was followed by an academic detailing campaign designed to encourage VA clinicians to initiate MOUD.1 Despite this institutional support, MOUD remains underutilized within the VA, with widely variable rates of prescribing among VA sites.1
Efforts to further expand MOUD cultivated interest in administering buprenorphine in VA emergency departments (EDs). Patients with OUD often use the ED for same-day care, providing opportunities to initiate buprenorphine in the ED 24 hours, 7 days per week. This has been especially true during the COVID-19 pandemic during which reliable access to usual recovery services has been disrupted and EDs have served as a safety net.6
Buprenorphine’s safety profile and prolonged effect duration make it superior to other MOUD options for ED administration. As a partial opioid agonist, buprenorphine is unlikely to cause significant sedation or respiratory depression compared with full agonists like methadone. This is known as the ceiling effect. Additionally, at higher doses, buprenorphine’s effects can last for about 3 days, potentially obviating the need for repeat dosing. D’Onofrio and colleagues seminal 2015 paper conceptually proved the feasibility and value of initiating buprenorphine in the ED; patients who received ED initiation therapy were more likely to be engaged in addiction treatment 30 days after their visit and have reduced rates of illicit opioid drug use.7 Such ED harm-reduction strategies are increasingly recognized as essential, given that 1 in 20 patients treated for a nonfatal opioid overdose in an ED will die within 1 year of their visit, many within 2 days.8 Finally, a significant barrier faced by physicians wanting to administer or prescribe buprenorphine for patients with OUD has been the special licensing required by the Drug Enforcement Administration Drug Addiction Treatment Act of 2000, also known as an X-waiver. A notable exception to this X-waiver requirement is the 72-hour rule, which allows nonwaivered practitioners to administer (but not prescribe for home use) buprenorphine to a patient to relieve acute withdrawal symptoms for up to 72 hours while arranging for specialist referral.Under the 72-hour rule, ED clinicians have a unique opportunity to treat patients experiencing acute withdrawal symptoms and bridge them to specialty care, without the burden of an X-waiver requirement.
The VA Greater Los Angeles Healthcare System (VAGLAHS), therefore, developed and implemented a program to administer buprenorphine in the ED to bridge patients with OUD to an appointment with substance use disorder (SUD) services. We describe our development, implementation and evaluation of this program protocol as a model for other VA EDs. This project was determined to be quality improvement (nonresearch) by the VAGLAHS Institutional Review Board.
ED MOUD Program
We engaged in a 2-month (January-March 2019) preimplementation process during which we (1) obtained stakeholder buy-in; (2) developed a protocol and supporting resources and tools; (3) worked with stakeholders to enact local organizational policy and process modifications; and (4) educated practitioners.
Appendix 1 provides an overview of MOUD terminology, pharmacology, and regulations. We developed an 8-step program implementation plan for the ED MOUD program (Figure 1).
Obtaining Stakeholder Buy-in
Two ED physician champions (MC, JH) organized all activities. Champions obtained stakeholder buy-in from clinical and administrative leaders as well as from frontline personnel in OUD specialty care, ED, and pharmacy services. ED social workers and clerks who schedule post-ED appointments also were engaged. These stakeholders emphasized the importance of fitting the developed protocol into the existing ED workflows as well as minimizing additional resources required to initiate and maintain the program.
We ascertained that in fiscal year 2018, VAGLAHS had 156 ED visits with International Statistical Classification of Diseases, Tenth Revision (ICD-10) codes related to OUD for 108 unique patients. Based on these data and in consultation with OUD specialty care, we determined that the potential number of referrals to the SUD clinic would be manageable with existing resources. Additionally, there was consensus that most opioid withdrawal patients could be treated in the urgent care portion of our ED since these patients generally do not require special monitoring. This consideration was important for obtaining ED stakeholder buy-in and for planning protocol logistics.
Developing the Protocol
We customized resources created by CalBridge Behavioral Health Navigator Program (CA Bridge), formerly called ED Bridge, a program of the Public Health Institute in Oakland, California, funded through California Department of Health Care Services. CA Bridge offers technical assistance and support for hospitals as well as guidance and tools for establishing processes for EDs providing buprenorphine prescriptions for the management of acute opioid withdrawal and serving as a bridge to follow-up care in SUD clinics.9 We also reviewed protocols described by D’Onofrio and colleagues. With iterative input from stakeholders, we created a protocol concretely delineating each process and corresponding responsible party with the overall aim of removing potential barriers to MOUD initiation and follow-up (Appendix 2).
Identifying Appropriate Follow-up
To operationalize protocol implementation, we built on VA’s Emergency Department Rapid Access Clinic (ED-RAC) process, a mechanism for scheduling appointments for post-ED specialty follow-up care. This process facilitated veterans’ access to urgent specialty care follow-up after ED visits by scheduling appointments prior to ED discharge.10 For the ED MOUD program, we adapted the ED-RAC process to schedule appointments in SUD clinic prior to ED discharge. These appointments allowed patients to be seen by an SUD clinician within 72 hours of ED discharge. This step was critical to working within the 72-hour rule without relying on X-waiver licensing of ED clinicians. Alternatively, as was previous practice, per patient preference, patients were also referred to non-VA residential rehabilitation services if the facility had capacity and patients met criteria for admission.
Identification of Eligible Veterans
Target patients were those primarily presenting with a request for treatment of opioid dependence or withdrawal. Patients were not actively screened for OUD. Clinicians diagnosed and assessed for OUD as per their usual practice. Patients with OUD who presented to the ED for other reasons were assessed, at clinician discretion, for their interest in receiving MOUD. If patients presented in moderate-to-severe withdrawal (eg, Clinical Opiate Withdrawal Scale [COWS] ≥ 8), buprenorphine was initiated in the ED. These patients were subsequently referred to either the local SUD clinic or to a residential treatment center. Patients presenting with a COWS score < 8 were referred to the outpatient SUD clinic or residential treatment centers without initiating buprenorphine from the ED. The SUD clinic or residential treatment centers could offer buprenorphine or other MOUD options. From the ED, prescribing buprenorphine for patients to self-initiate at home was not available as this required an X-waivered prescriber, which were limited during the program time frame.
Support Tools and Resources
To facilitate ED clinicians using the protocol, we worked with a programmer experienced with the Computerized Patient Record System, the VA electronic health record (EHR), to create electronic order menu sets that directed clinicians to the protocol and educational materials (Appendix 3). These menus are readily accessible and embedded into the ED clinician workflow. The menus highlight key elements of the protocol, including indications for initiation, contraindications, recommended dosing with quick orders, and how to obtain follow-up for the patient. Links also are provided to the protocol and patient discharge handouts, including the CA Bridge website.
Organizational Policy and Processes
Before implementing the developed protocol, we worked with stakeholders to modify organizational policies and processes. Our pharmacy agreed to stock buprenorphine in the ED to make it readily available. EHR restrictions that historically prohibited ordering buprenorphine for ED administration by nonwaivered clinicians were modified. Additionally, our chief of staff, pharmacy, and credentialing department agreed that physicians did not need to apply for additional delineated privileges.
Clinician Education
The final preparation step was educating clinicians and other protocol users. The VAGLAHS SUD chief presented a lecture and answered questions about MOUD to core ED faculty about the rising prevalence of OUD and use of buprenorphine as a recommended treatment.
Evaluation
To assess adherence to the developed protocol, we conducted a retrospective health record review of all ED visits March 1 to October 25, 2019, in which the patient had OUD and may have qualified for MOUD. To do this, we identified (1) ED visits with an OUD ICD-10 code as a primary or secondary diagnoses; (2) ED referrals to outpatient SUD treatment; and/or (3) ED visits in which buprenorphine was given or prescribed. We included the latter 2 criteria as application of ICD-10 codes for OUD care was inconsistent. Visits were excluded if patients did not have OUD, had OUD in remission, were already maintained on a stable MOUD regimen and no longer using illicit drugs or craving additional opioids, or were presenting solely for a refill or administration of a missed dose. Patients who relapsed were categorized as unstable. Visits were excluded if the patient was admitted to the hospital or left against medical advice. Patients on MOUD who had relapsed or requested a change in MOUD treatment were included. For all included visits, 2 ED physicians (MC, JH) reviewed the ED clinician and nursing notes, pharmacy and referral records, diagnostic codes, and veteran demographics.
In the evaluation, there were 130 visits with 92 unique veterans meeting inclusion criteria. The final sample included 70 visits with 47 unique veterans (Table 1). Of note, 24 (53%) patients self-identified as homeless or were engaged with VA housing services. Twelve veterans had multiple ED visits (7 patients with 2 visits; 5 patients with ≥ 3 visits). In 30 (43%) visits the veteran’s primary reason for seeking ED care was to obtain treatment for opioid withdrawal or receive MOUD. Type of opiate used was specified in 58% of visits; of these, 69% indicated heroin use and 17% prescription medications. Buprenorphine was initiated in the ED in 18 (26%) visits for 10 veterans. Appendix 4 outlines the clinical course and follow-up after these visits. Some veterans returned to the ED for buprenorphine redosing per the 72-hour rule. SUD clinic appointments were provided in 11 visits, and direct transfer to an inpatient rehabilitation center was arranged in 4 visits. In 42 (60%) visits, across 32 unique veterans, buprenorphine was not given in the ED, but patients were referred for SUD treat
A majority of veterans who received buprenorphine and a referral for an SUD appointment went to their initial SUD follow-up appointment and had ongoing engagement in addiction care 30 days after their index ED visit. Among veterans who did not receive buprenorphine but were referred for SUD treatment, about half went to their SUD appointments and about 1 in 5 had ongoing engagement in addiction care at 30 days after the index ED visit. Of note, 2 patients who received referrals died within 1 year of their index ED visit. The cause of death for one patient was an overdose; the other was unspecified.
DISCUSSION
We developed the ED MOUD program as a bridge to SUD specialty care. Our 8 implementation steps can serve as a model for implementing programs at other VA EDs. We demonstrated feasibility, high follow-up rates, and high retention in treatment.
Patients who received ED buprenorphine initiation were more likely to follow up and had higher rates of ongoing engagement at 30 days than did those who received only a clinic referral. In a similar Canadian study, buprenorphine was initiated in the ED, and patients followed up as a walk-in for addiction services; however, only 54% of patients presented to this initial follow-up.11 Our higher initial follow-up rate may be due to our ability to directly schedule clinic appointments. Our 70% 30-day follow-up rate is comparable, but slightly lower than the 2015 D’Onofrio and colleagues study in which 78% of patients remained engaged at 30 days.7 A possible reason is that in the D’Onofrio and colleagues study, all study physicians obtained X-waiver training and were able to prescribe buprenorphine after ED initiation or for self-initiation at home. X-waiver training was not required of our clinicians, and none of our patients were offered a prescription for self-initiation.
Our program demonstrates that it is feasible to develop a protocol without X-waiver licensing. This program provides a supportive framework for the use of MOUD and allows nonspecialists to gain experience and confidence in using buprenorphine. Any clinician could administer buprenorphine in the ED, and patients could be bridged at later ED visits until follow-up with a specialist. Of note, only a small percentage of the total visits for buprenorphine initiation required multiple daily visits for buprenorphine. Appointments with the specialist were assured to fall within a 72-hour window.
Our program has some limitations. First, the number of patients who were candidates for our ED MOUD program was small. In our 7-month review, only 47 patients were identified as potential candidates for MOUD treatment across 70 visits, and only 10 were initiated in the ED. Second, all patients were not actively screened for OUD. There was potential for missing eligible veterans as inclusion criteria relied on clinicians both recognizing OUD and manually entering a correct diagnostic code. We attempted to mitigate this by also reviewing all ED referrals to the SUD clinic and all patients who received buprenorphine in the ED. In addition, we do not have data on preimplementation rates of follow-up for comparison.
Future Directions
More than half of our patients did not receive ED buprenorphine initiation because they were not in moderate or severe withdrawal (COWS ≥ 8) similar to 57% of patients cited in the D’Onofrio and colleagues study.7 Teaching veterans how to start buprenorphine at home could greatly expand enrollment. However, this requires a prescription from an X-waiver licensed clinician. In 2021, the US Department of Health and Human Services removed the 8-hour training requirement for obtaining an X-waiver.12 However, clinicians are still required to apply for licensing. Eliminating the X-waiver requirement, as proposed by D’Onofrio and colleagues in a 2021 editorial, would have allowed all clinicians to offer home initiation.13
Previous studies suggest that despite the ability to provide a prescription, clinicians may be reluctant to offer home initiation.14–17 In a national VA 2019 survey, many emergency medicine physicians believe that SUD care is not in their scope of practice, as Dieujuste and colleagues described in Federal Practitioner.14 Although it is likely some attitudes have changed with the increased visibility of ED MOUD programs, there is still much work to be done to change perceptions.
Another area for improvement is screening for OUD in the ED to better reveal MOUD candidates. Missed opportunities (neither referral nor treatment offered) occurred in 21% of our visits. D’Onofrio and colleagues identified 66% of patients by screening all ED patients.7 Although universal screening for SUD in routine health care settings has been recommended, 2021 VA guidelines state that there is insufficient evidence to recommend universal screening.18-20 There are also limited data on the best screening tool for OUD in the ED.21 Further research on how to effectively and efficiently identify OUD patients in the ED is needed.
Conclusions
With minimal resource allocation, we started the program to offer MOUD with buprenorphine for patients with OUD at a VA ED and provided addiction treatment follow-up. This program, the first of its kind within VA, can be modeled and expanded to other VA facilities. Given increasing numbers of fatal opioid overdose, and significant adverse impacts of the COVID-19 pandemic on the OUD crisis, developing local and national strategies to treat OUD is essential. Future steps include improved screening and expanding capacity to offer home initiation by increasing the number of X-waiver ED clinicians.6
Acknowledgments
Thank you to Jeffrey Balsam, PharmD, BCPS, Veterans Affairs Greater Los Angeles Clinical Applications Coordinator for his contributions in creating a Computerized Patient Record System opioid use disorder screening tool. Thank you to Gracielle Tan, MD, Veterans Affairs Greater Los Angeles Health Science Specialist for her administrative assistance in manuscript preparation.
1. Wyse JJ, Gordon AJ, Dobscha SK, et al. Medications for opioid use disorder in the Department of Veterans Affairs (VA) health care system: historical perspective, lessons learned, and next steps. Subst Abuse. 2018;39(2):139-144. doi:10.1080/08897077.2018.1452327
2. Bohnert ASB, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs health system. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. Ma J, Bao Y-P, Wang R-J, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1968-1983. doi:10.1038/s41380-018-0094-5
4. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 2.0. US Department of Veterans Affairs; 2009.
5. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 3.0. US Department of Veterans Affairs. 2015. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPGRevised22216.pdf
6. Hulsey J, Mellis A, Kelly B. COVID-19 pandemic impact on patients, families and individuals in recovery from substance use disorder. Accessed July 7, 2021. https://www.addictionpolicy.org/covid19-report
7. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opiod dependence. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
8. Weiner SG, Baker O, Bernson D, Schuur JD. One-year mortality of patients after emergency department treatment for non-fatal opioid overdose. Ann Emerg Med. 2020;75(1):13-17. doi:10.1016/j.annemergmed.2019.04.020
9. CA Bridge. Updated 2021. Accessed July 1, 2022. https://cabridge.org
10. Penney L, Miake-Lye I, Lewis D, et al. Proceedings from the 11th annual conference on the science of dissemination and implementation: S72 spreading VA’s emergency department-rapid access clinics (ED-RAC) intervention: key factors for success. Implementation Sci. 2019;14(suppl 1). doi:10.1186/s13012-019-0878-2
11. Hu T, Snider-Alder M, Nijmeh L, Pyle A. Buprenorphine/naloxone induction in a Canadian emergency department with rapid access to community-based addictions providers. CJEM. 2019;21(4):492-498. doi:10.1017/cem.2019.24
12. US Department of Health and Human Services. Practice Guidelines for the Administration of Buprenorphine for Treating Opioid Use Disorder. Federal Register. Accessed July 1, 2022. https://www.federalregister.gov/documents/2021/04/28/2021-08961/practice-guidelines-for-the-administration-of-buprenorphine-for-treating-opioid-use-disorder
13. D’Onofrio G, Melnick ER, Hawk KF. Improve access to care for opioid use disorder: a call to eliminate the x-waiver requirement now. Ann Emerg Med. 2021;78(2):220-222. doi:10.1016/j.annemergmed.2021.03.023
14. Dieujuste N, Johnson-Koenke R, Celedon M, et al. Provider perceptions of opioid safety measures in VHA emergency department and urgent care centers. Fed Pract. 2021;38(9):412-419. doi:10.12788/fp.0179
15. Hawk KF, D’Onofrio G, Chawarski MC, et al. Barriers and faciliatators to clinician readiness to provide emergency department-initiated buprenorphine. JAMA Netw Open. 2020;3(5):e204561. doi:10.1001/jamanetworkopen.2020.4561
16. Lowenstein M, Kilaru A, Perrone J, et al. Barriers and facilitators for emergency department initiation of buprenorphine: a physician survey. Am J Emerg Med. 2019;37(9):1787-1790. doi:10.1016/j.ajem.2019.02.025
17. Srivastava A, Kahan M, Leece P, McAndrew A. Buprenorphine unobserved “home” induction: a survey of Ontario’s addiction physicians. Addic Sci Clin Pract. 2019;14(1):18. doi:10.1186/s13722-019-0146-4
18. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 4.0. US Department of Veterans Affairs. 2021. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPG.pdf
19. Patnode CD, Perdue LA, Rushkin M, et al. Screening for unhealthy drug use updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;323(22):2310-2338. doi:10.1001/jama.2019.21381
20. Coles S, Vosooney A. Evidence lacking to support universal unhealthy drug use screening. Am Fam Physician. 2021;103(2):72-73.
21. Sahota PK, Sharstry S, Mukamel DB, et al. Screening emergency department patients for opioid drug use: a qualitative systematic review. Addict Behav. 2018;85:139-146. doi:10.1016/j.addbeh.2018.05.022
Opioid use disorder (OUD) is a public health crisis significantly affecting veterans. A substantial increase in veterans diagnosed with OUD has occurred, nearly tripling from 25,031 in 2003 to 69,142 in 2017
For patients with active OUD, medications for opioid use disorder (MOUD) reduce the risk of overdose and all-cause mortality.3 In 2009, the US Department of Veterans Affairs (VA) and Department of Defense (DoD) published clinical practice guidelines for substance use disorders that strongly recommended MOUD with either buprenorphine or methadone as a first-line treatment. In 2015 updated guidelines encouraged buprenorphine initiation in primary care settings.4,5 This was followed by an academic detailing campaign designed to encourage VA clinicians to initiate MOUD.1 Despite this institutional support, MOUD remains underutilized within the VA, with widely variable rates of prescribing among VA sites.1
Efforts to further expand MOUD cultivated interest in administering buprenorphine in VA emergency departments (EDs). Patients with OUD often use the ED for same-day care, providing opportunities to initiate buprenorphine in the ED 24 hours, 7 days per week. This has been especially true during the COVID-19 pandemic during which reliable access to usual recovery services has been disrupted and EDs have served as a safety net.6
Buprenorphine’s safety profile and prolonged effect duration make it superior to other MOUD options for ED administration. As a partial opioid agonist, buprenorphine is unlikely to cause significant sedation or respiratory depression compared with full agonists like methadone. This is known as the ceiling effect. Additionally, at higher doses, buprenorphine’s effects can last for about 3 days, potentially obviating the need for repeat dosing. D’Onofrio and colleagues seminal 2015 paper conceptually proved the feasibility and value of initiating buprenorphine in the ED; patients who received ED initiation therapy were more likely to be engaged in addiction treatment 30 days after their visit and have reduced rates of illicit opioid drug use.7 Such ED harm-reduction strategies are increasingly recognized as essential, given that 1 in 20 patients treated for a nonfatal opioid overdose in an ED will die within 1 year of their visit, many within 2 days.8 Finally, a significant barrier faced by physicians wanting to administer or prescribe buprenorphine for patients with OUD has been the special licensing required by the Drug Enforcement Administration Drug Addiction Treatment Act of 2000, also known as an X-waiver. A notable exception to this X-waiver requirement is the 72-hour rule, which allows nonwaivered practitioners to administer (but not prescribe for home use) buprenorphine to a patient to relieve acute withdrawal symptoms for up to 72 hours while arranging for specialist referral.Under the 72-hour rule, ED clinicians have a unique opportunity to treat patients experiencing acute withdrawal symptoms and bridge them to specialty care, without the burden of an X-waiver requirement.
The VA Greater Los Angeles Healthcare System (VAGLAHS), therefore, developed and implemented a program to administer buprenorphine in the ED to bridge patients with OUD to an appointment with substance use disorder (SUD) services. We describe our development, implementation and evaluation of this program protocol as a model for other VA EDs. This project was determined to be quality improvement (nonresearch) by the VAGLAHS Institutional Review Board.
ED MOUD Program
We engaged in a 2-month (January-March 2019) preimplementation process during which we (1) obtained stakeholder buy-in; (2) developed a protocol and supporting resources and tools; (3) worked with stakeholders to enact local organizational policy and process modifications; and (4) educated practitioners.
Appendix 1 provides an overview of MOUD terminology, pharmacology, and regulations. We developed an 8-step program implementation plan for the ED MOUD program (Figure 1).
Obtaining Stakeholder Buy-in
Two ED physician champions (MC, JH) organized all activities. Champions obtained stakeholder buy-in from clinical and administrative leaders as well as from frontline personnel in OUD specialty care, ED, and pharmacy services. ED social workers and clerks who schedule post-ED appointments also were engaged. These stakeholders emphasized the importance of fitting the developed protocol into the existing ED workflows as well as minimizing additional resources required to initiate and maintain the program.
We ascertained that in fiscal year 2018, VAGLAHS had 156 ED visits with International Statistical Classification of Diseases, Tenth Revision (ICD-10) codes related to OUD for 108 unique patients. Based on these data and in consultation with OUD specialty care, we determined that the potential number of referrals to the SUD clinic would be manageable with existing resources. Additionally, there was consensus that most opioid withdrawal patients could be treated in the urgent care portion of our ED since these patients generally do not require special monitoring. This consideration was important for obtaining ED stakeholder buy-in and for planning protocol logistics.
Developing the Protocol
We customized resources created by CalBridge Behavioral Health Navigator Program (CA Bridge), formerly called ED Bridge, a program of the Public Health Institute in Oakland, California, funded through California Department of Health Care Services. CA Bridge offers technical assistance and support for hospitals as well as guidance and tools for establishing processes for EDs providing buprenorphine prescriptions for the management of acute opioid withdrawal and serving as a bridge to follow-up care in SUD clinics.9 We also reviewed protocols described by D’Onofrio and colleagues. With iterative input from stakeholders, we created a protocol concretely delineating each process and corresponding responsible party with the overall aim of removing potential barriers to MOUD initiation and follow-up (Appendix 2).
Identifying Appropriate Follow-up
To operationalize protocol implementation, we built on VA’s Emergency Department Rapid Access Clinic (ED-RAC) process, a mechanism for scheduling appointments for post-ED specialty follow-up care. This process facilitated veterans’ access to urgent specialty care follow-up after ED visits by scheduling appointments prior to ED discharge.10 For the ED MOUD program, we adapted the ED-RAC process to schedule appointments in SUD clinic prior to ED discharge. These appointments allowed patients to be seen by an SUD clinician within 72 hours of ED discharge. This step was critical to working within the 72-hour rule without relying on X-waiver licensing of ED clinicians. Alternatively, as was previous practice, per patient preference, patients were also referred to non-VA residential rehabilitation services if the facility had capacity and patients met criteria for admission.
Identification of Eligible Veterans
Target patients were those primarily presenting with a request for treatment of opioid dependence or withdrawal. Patients were not actively screened for OUD. Clinicians diagnosed and assessed for OUD as per their usual practice. Patients with OUD who presented to the ED for other reasons were assessed, at clinician discretion, for their interest in receiving MOUD. If patients presented in moderate-to-severe withdrawal (eg, Clinical Opiate Withdrawal Scale [COWS] ≥ 8), buprenorphine was initiated in the ED. These patients were subsequently referred to either the local SUD clinic or to a residential treatment center. Patients presenting with a COWS score < 8 were referred to the outpatient SUD clinic or residential treatment centers without initiating buprenorphine from the ED. The SUD clinic or residential treatment centers could offer buprenorphine or other MOUD options. From the ED, prescribing buprenorphine for patients to self-initiate at home was not available as this required an X-waivered prescriber, which were limited during the program time frame.
Support Tools and Resources
To facilitate ED clinicians using the protocol, we worked with a programmer experienced with the Computerized Patient Record System, the VA electronic health record (EHR), to create electronic order menu sets that directed clinicians to the protocol and educational materials (Appendix 3). These menus are readily accessible and embedded into the ED clinician workflow. The menus highlight key elements of the protocol, including indications for initiation, contraindications, recommended dosing with quick orders, and how to obtain follow-up for the patient. Links also are provided to the protocol and patient discharge handouts, including the CA Bridge website.
Organizational Policy and Processes
Before implementing the developed protocol, we worked with stakeholders to modify organizational policies and processes. Our pharmacy agreed to stock buprenorphine in the ED to make it readily available. EHR restrictions that historically prohibited ordering buprenorphine for ED administration by nonwaivered clinicians were modified. Additionally, our chief of staff, pharmacy, and credentialing department agreed that physicians did not need to apply for additional delineated privileges.
Clinician Education
The final preparation step was educating clinicians and other protocol users. The VAGLAHS SUD chief presented a lecture and answered questions about MOUD to core ED faculty about the rising prevalence of OUD and use of buprenorphine as a recommended treatment.
Evaluation
To assess adherence to the developed protocol, we conducted a retrospective health record review of all ED visits March 1 to October 25, 2019, in which the patient had OUD and may have qualified for MOUD. To do this, we identified (1) ED visits with an OUD ICD-10 code as a primary or secondary diagnoses; (2) ED referrals to outpatient SUD treatment; and/or (3) ED visits in which buprenorphine was given or prescribed. We included the latter 2 criteria as application of ICD-10 codes for OUD care was inconsistent. Visits were excluded if patients did not have OUD, had OUD in remission, were already maintained on a stable MOUD regimen and no longer using illicit drugs or craving additional opioids, or were presenting solely for a refill or administration of a missed dose. Patients who relapsed were categorized as unstable. Visits were excluded if the patient was admitted to the hospital or left against medical advice. Patients on MOUD who had relapsed or requested a change in MOUD treatment were included. For all included visits, 2 ED physicians (MC, JH) reviewed the ED clinician and nursing notes, pharmacy and referral records, diagnostic codes, and veteran demographics.
In the evaluation, there were 130 visits with 92 unique veterans meeting inclusion criteria. The final sample included 70 visits with 47 unique veterans (Table 1). Of note, 24 (53%) patients self-identified as homeless or were engaged with VA housing services. Twelve veterans had multiple ED visits (7 patients with 2 visits; 5 patients with ≥ 3 visits). In 30 (43%) visits the veteran’s primary reason for seeking ED care was to obtain treatment for opioid withdrawal or receive MOUD. Type of opiate used was specified in 58% of visits; of these, 69% indicated heroin use and 17% prescription medications. Buprenorphine was initiated in the ED in 18 (26%) visits for 10 veterans. Appendix 4 outlines the clinical course and follow-up after these visits. Some veterans returned to the ED for buprenorphine redosing per the 72-hour rule. SUD clinic appointments were provided in 11 visits, and direct transfer to an inpatient rehabilitation center was arranged in 4 visits. In 42 (60%) visits, across 32 unique veterans, buprenorphine was not given in the ED, but patients were referred for SUD treat
A majority of veterans who received buprenorphine and a referral for an SUD appointment went to their initial SUD follow-up appointment and had ongoing engagement in addiction care 30 days after their index ED visit. Among veterans who did not receive buprenorphine but were referred for SUD treatment, about half went to their SUD appointments and about 1 in 5 had ongoing engagement in addiction care at 30 days after the index ED visit. Of note, 2 patients who received referrals died within 1 year of their index ED visit. The cause of death for one patient was an overdose; the other was unspecified.
DISCUSSION
We developed the ED MOUD program as a bridge to SUD specialty care. Our 8 implementation steps can serve as a model for implementing programs at other VA EDs. We demonstrated feasibility, high follow-up rates, and high retention in treatment.
Patients who received ED buprenorphine initiation were more likely to follow up and had higher rates of ongoing engagement at 30 days than did those who received only a clinic referral. In a similar Canadian study, buprenorphine was initiated in the ED, and patients followed up as a walk-in for addiction services; however, only 54% of patients presented to this initial follow-up.11 Our higher initial follow-up rate may be due to our ability to directly schedule clinic appointments. Our 70% 30-day follow-up rate is comparable, but slightly lower than the 2015 D’Onofrio and colleagues study in which 78% of patients remained engaged at 30 days.7 A possible reason is that in the D’Onofrio and colleagues study, all study physicians obtained X-waiver training and were able to prescribe buprenorphine after ED initiation or for self-initiation at home. X-waiver training was not required of our clinicians, and none of our patients were offered a prescription for self-initiation.
Our program demonstrates that it is feasible to develop a protocol without X-waiver licensing. This program provides a supportive framework for the use of MOUD and allows nonspecialists to gain experience and confidence in using buprenorphine. Any clinician could administer buprenorphine in the ED, and patients could be bridged at later ED visits until follow-up with a specialist. Of note, only a small percentage of the total visits for buprenorphine initiation required multiple daily visits for buprenorphine. Appointments with the specialist were assured to fall within a 72-hour window.
Our program has some limitations. First, the number of patients who were candidates for our ED MOUD program was small. In our 7-month review, only 47 patients were identified as potential candidates for MOUD treatment across 70 visits, and only 10 were initiated in the ED. Second, all patients were not actively screened for OUD. There was potential for missing eligible veterans as inclusion criteria relied on clinicians both recognizing OUD and manually entering a correct diagnostic code. We attempted to mitigate this by also reviewing all ED referrals to the SUD clinic and all patients who received buprenorphine in the ED. In addition, we do not have data on preimplementation rates of follow-up for comparison.
Future Directions
More than half of our patients did not receive ED buprenorphine initiation because they were not in moderate or severe withdrawal (COWS ≥ 8) similar to 57% of patients cited in the D’Onofrio and colleagues study.7 Teaching veterans how to start buprenorphine at home could greatly expand enrollment. However, this requires a prescription from an X-waiver licensed clinician. In 2021, the US Department of Health and Human Services removed the 8-hour training requirement for obtaining an X-waiver.12 However, clinicians are still required to apply for licensing. Eliminating the X-waiver requirement, as proposed by D’Onofrio and colleagues in a 2021 editorial, would have allowed all clinicians to offer home initiation.13
Previous studies suggest that despite the ability to provide a prescription, clinicians may be reluctant to offer home initiation.14–17 In a national VA 2019 survey, many emergency medicine physicians believe that SUD care is not in their scope of practice, as Dieujuste and colleagues described in Federal Practitioner.14 Although it is likely some attitudes have changed with the increased visibility of ED MOUD programs, there is still much work to be done to change perceptions.
Another area for improvement is screening for OUD in the ED to better reveal MOUD candidates. Missed opportunities (neither referral nor treatment offered) occurred in 21% of our visits. D’Onofrio and colleagues identified 66% of patients by screening all ED patients.7 Although universal screening for SUD in routine health care settings has been recommended, 2021 VA guidelines state that there is insufficient evidence to recommend universal screening.18-20 There are also limited data on the best screening tool for OUD in the ED.21 Further research on how to effectively and efficiently identify OUD patients in the ED is needed.
Conclusions
With minimal resource allocation, we started the program to offer MOUD with buprenorphine for patients with OUD at a VA ED and provided addiction treatment follow-up. This program, the first of its kind within VA, can be modeled and expanded to other VA facilities. Given increasing numbers of fatal opioid overdose, and significant adverse impacts of the COVID-19 pandemic on the OUD crisis, developing local and national strategies to treat OUD is essential. Future steps include improved screening and expanding capacity to offer home initiation by increasing the number of X-waiver ED clinicians.6
Acknowledgments
Thank you to Jeffrey Balsam, PharmD, BCPS, Veterans Affairs Greater Los Angeles Clinical Applications Coordinator for his contributions in creating a Computerized Patient Record System opioid use disorder screening tool. Thank you to Gracielle Tan, MD, Veterans Affairs Greater Los Angeles Health Science Specialist for her administrative assistance in manuscript preparation.
Opioid use disorder (OUD) is a public health crisis significantly affecting veterans. A substantial increase in veterans diagnosed with OUD has occurred, nearly tripling from 25,031 in 2003 to 69,142 in 2017
For patients with active OUD, medications for opioid use disorder (MOUD) reduce the risk of overdose and all-cause mortality.3 In 2009, the US Department of Veterans Affairs (VA) and Department of Defense (DoD) published clinical practice guidelines for substance use disorders that strongly recommended MOUD with either buprenorphine or methadone as a first-line treatment. In 2015 updated guidelines encouraged buprenorphine initiation in primary care settings.4,5 This was followed by an academic detailing campaign designed to encourage VA clinicians to initiate MOUD.1 Despite this institutional support, MOUD remains underutilized within the VA, with widely variable rates of prescribing among VA sites.1
Efforts to further expand MOUD cultivated interest in administering buprenorphine in VA emergency departments (EDs). Patients with OUD often use the ED for same-day care, providing opportunities to initiate buprenorphine in the ED 24 hours, 7 days per week. This has been especially true during the COVID-19 pandemic during which reliable access to usual recovery services has been disrupted and EDs have served as a safety net.6
Buprenorphine’s safety profile and prolonged effect duration make it superior to other MOUD options for ED administration. As a partial opioid agonist, buprenorphine is unlikely to cause significant sedation or respiratory depression compared with full agonists like methadone. This is known as the ceiling effect. Additionally, at higher doses, buprenorphine’s effects can last for about 3 days, potentially obviating the need for repeat dosing. D’Onofrio and colleagues seminal 2015 paper conceptually proved the feasibility and value of initiating buprenorphine in the ED; patients who received ED initiation therapy were more likely to be engaged in addiction treatment 30 days after their visit and have reduced rates of illicit opioid drug use.7 Such ED harm-reduction strategies are increasingly recognized as essential, given that 1 in 20 patients treated for a nonfatal opioid overdose in an ED will die within 1 year of their visit, many within 2 days.8 Finally, a significant barrier faced by physicians wanting to administer or prescribe buprenorphine for patients with OUD has been the special licensing required by the Drug Enforcement Administration Drug Addiction Treatment Act of 2000, also known as an X-waiver. A notable exception to this X-waiver requirement is the 72-hour rule, which allows nonwaivered practitioners to administer (but not prescribe for home use) buprenorphine to a patient to relieve acute withdrawal symptoms for up to 72 hours while arranging for specialist referral.Under the 72-hour rule, ED clinicians have a unique opportunity to treat patients experiencing acute withdrawal symptoms and bridge them to specialty care, without the burden of an X-waiver requirement.
The VA Greater Los Angeles Healthcare System (VAGLAHS), therefore, developed and implemented a program to administer buprenorphine in the ED to bridge patients with OUD to an appointment with substance use disorder (SUD) services. We describe our development, implementation and evaluation of this program protocol as a model for other VA EDs. This project was determined to be quality improvement (nonresearch) by the VAGLAHS Institutional Review Board.
ED MOUD Program
We engaged in a 2-month (January-March 2019) preimplementation process during which we (1) obtained stakeholder buy-in; (2) developed a protocol and supporting resources and tools; (3) worked with stakeholders to enact local organizational policy and process modifications; and (4) educated practitioners.
Appendix 1 provides an overview of MOUD terminology, pharmacology, and regulations. We developed an 8-step program implementation plan for the ED MOUD program (Figure 1).
Obtaining Stakeholder Buy-in
Two ED physician champions (MC, JH) organized all activities. Champions obtained stakeholder buy-in from clinical and administrative leaders as well as from frontline personnel in OUD specialty care, ED, and pharmacy services. ED social workers and clerks who schedule post-ED appointments also were engaged. These stakeholders emphasized the importance of fitting the developed protocol into the existing ED workflows as well as minimizing additional resources required to initiate and maintain the program.
We ascertained that in fiscal year 2018, VAGLAHS had 156 ED visits with International Statistical Classification of Diseases, Tenth Revision (ICD-10) codes related to OUD for 108 unique patients. Based on these data and in consultation with OUD specialty care, we determined that the potential number of referrals to the SUD clinic would be manageable with existing resources. Additionally, there was consensus that most opioid withdrawal patients could be treated in the urgent care portion of our ED since these patients generally do not require special monitoring. This consideration was important for obtaining ED stakeholder buy-in and for planning protocol logistics.
Developing the Protocol
We customized resources created by CalBridge Behavioral Health Navigator Program (CA Bridge), formerly called ED Bridge, a program of the Public Health Institute in Oakland, California, funded through California Department of Health Care Services. CA Bridge offers technical assistance and support for hospitals as well as guidance and tools for establishing processes for EDs providing buprenorphine prescriptions for the management of acute opioid withdrawal and serving as a bridge to follow-up care in SUD clinics.9 We also reviewed protocols described by D’Onofrio and colleagues. With iterative input from stakeholders, we created a protocol concretely delineating each process and corresponding responsible party with the overall aim of removing potential barriers to MOUD initiation and follow-up (Appendix 2).
Identifying Appropriate Follow-up
To operationalize protocol implementation, we built on VA’s Emergency Department Rapid Access Clinic (ED-RAC) process, a mechanism for scheduling appointments for post-ED specialty follow-up care. This process facilitated veterans’ access to urgent specialty care follow-up after ED visits by scheduling appointments prior to ED discharge.10 For the ED MOUD program, we adapted the ED-RAC process to schedule appointments in SUD clinic prior to ED discharge. These appointments allowed patients to be seen by an SUD clinician within 72 hours of ED discharge. This step was critical to working within the 72-hour rule without relying on X-waiver licensing of ED clinicians. Alternatively, as was previous practice, per patient preference, patients were also referred to non-VA residential rehabilitation services if the facility had capacity and patients met criteria for admission.
Identification of Eligible Veterans
Target patients were those primarily presenting with a request for treatment of opioid dependence or withdrawal. Patients were not actively screened for OUD. Clinicians diagnosed and assessed for OUD as per their usual practice. Patients with OUD who presented to the ED for other reasons were assessed, at clinician discretion, for their interest in receiving MOUD. If patients presented in moderate-to-severe withdrawal (eg, Clinical Opiate Withdrawal Scale [COWS] ≥ 8), buprenorphine was initiated in the ED. These patients were subsequently referred to either the local SUD clinic or to a residential treatment center. Patients presenting with a COWS score < 8 were referred to the outpatient SUD clinic or residential treatment centers without initiating buprenorphine from the ED. The SUD clinic or residential treatment centers could offer buprenorphine or other MOUD options. From the ED, prescribing buprenorphine for patients to self-initiate at home was not available as this required an X-waivered prescriber, which were limited during the program time frame.
Support Tools and Resources
To facilitate ED clinicians using the protocol, we worked with a programmer experienced with the Computerized Patient Record System, the VA electronic health record (EHR), to create electronic order menu sets that directed clinicians to the protocol and educational materials (Appendix 3). These menus are readily accessible and embedded into the ED clinician workflow. The menus highlight key elements of the protocol, including indications for initiation, contraindications, recommended dosing with quick orders, and how to obtain follow-up for the patient. Links also are provided to the protocol and patient discharge handouts, including the CA Bridge website.
Organizational Policy and Processes
Before implementing the developed protocol, we worked with stakeholders to modify organizational policies and processes. Our pharmacy agreed to stock buprenorphine in the ED to make it readily available. EHR restrictions that historically prohibited ordering buprenorphine for ED administration by nonwaivered clinicians were modified. Additionally, our chief of staff, pharmacy, and credentialing department agreed that physicians did not need to apply for additional delineated privileges.
Clinician Education
The final preparation step was educating clinicians and other protocol users. The VAGLAHS SUD chief presented a lecture and answered questions about MOUD to core ED faculty about the rising prevalence of OUD and use of buprenorphine as a recommended treatment.
Evaluation
To assess adherence to the developed protocol, we conducted a retrospective health record review of all ED visits March 1 to October 25, 2019, in which the patient had OUD and may have qualified for MOUD. To do this, we identified (1) ED visits with an OUD ICD-10 code as a primary or secondary diagnoses; (2) ED referrals to outpatient SUD treatment; and/or (3) ED visits in which buprenorphine was given or prescribed. We included the latter 2 criteria as application of ICD-10 codes for OUD care was inconsistent. Visits were excluded if patients did not have OUD, had OUD in remission, were already maintained on a stable MOUD regimen and no longer using illicit drugs or craving additional opioids, or were presenting solely for a refill or administration of a missed dose. Patients who relapsed were categorized as unstable. Visits were excluded if the patient was admitted to the hospital or left against medical advice. Patients on MOUD who had relapsed or requested a change in MOUD treatment were included. For all included visits, 2 ED physicians (MC, JH) reviewed the ED clinician and nursing notes, pharmacy and referral records, diagnostic codes, and veteran demographics.
In the evaluation, there were 130 visits with 92 unique veterans meeting inclusion criteria. The final sample included 70 visits with 47 unique veterans (Table 1). Of note, 24 (53%) patients self-identified as homeless or were engaged with VA housing services. Twelve veterans had multiple ED visits (7 patients with 2 visits; 5 patients with ≥ 3 visits). In 30 (43%) visits the veteran’s primary reason for seeking ED care was to obtain treatment for opioid withdrawal or receive MOUD. Type of opiate used was specified in 58% of visits; of these, 69% indicated heroin use and 17% prescription medications. Buprenorphine was initiated in the ED in 18 (26%) visits for 10 veterans. Appendix 4 outlines the clinical course and follow-up after these visits. Some veterans returned to the ED for buprenorphine redosing per the 72-hour rule. SUD clinic appointments were provided in 11 visits, and direct transfer to an inpatient rehabilitation center was arranged in 4 visits. In 42 (60%) visits, across 32 unique veterans, buprenorphine was not given in the ED, but patients were referred for SUD treat
A majority of veterans who received buprenorphine and a referral for an SUD appointment went to their initial SUD follow-up appointment and had ongoing engagement in addiction care 30 days after their index ED visit. Among veterans who did not receive buprenorphine but were referred for SUD treatment, about half went to their SUD appointments and about 1 in 5 had ongoing engagement in addiction care at 30 days after the index ED visit. Of note, 2 patients who received referrals died within 1 year of their index ED visit. The cause of death for one patient was an overdose; the other was unspecified.
DISCUSSION
We developed the ED MOUD program as a bridge to SUD specialty care. Our 8 implementation steps can serve as a model for implementing programs at other VA EDs. We demonstrated feasibility, high follow-up rates, and high retention in treatment.
Patients who received ED buprenorphine initiation were more likely to follow up and had higher rates of ongoing engagement at 30 days than did those who received only a clinic referral. In a similar Canadian study, buprenorphine was initiated in the ED, and patients followed up as a walk-in for addiction services; however, only 54% of patients presented to this initial follow-up.11 Our higher initial follow-up rate may be due to our ability to directly schedule clinic appointments. Our 70% 30-day follow-up rate is comparable, but slightly lower than the 2015 D’Onofrio and colleagues study in which 78% of patients remained engaged at 30 days.7 A possible reason is that in the D’Onofrio and colleagues study, all study physicians obtained X-waiver training and were able to prescribe buprenorphine after ED initiation or for self-initiation at home. X-waiver training was not required of our clinicians, and none of our patients were offered a prescription for self-initiation.
Our program demonstrates that it is feasible to develop a protocol without X-waiver licensing. This program provides a supportive framework for the use of MOUD and allows nonspecialists to gain experience and confidence in using buprenorphine. Any clinician could administer buprenorphine in the ED, and patients could be bridged at later ED visits until follow-up with a specialist. Of note, only a small percentage of the total visits for buprenorphine initiation required multiple daily visits for buprenorphine. Appointments with the specialist were assured to fall within a 72-hour window.
Our program has some limitations. First, the number of patients who were candidates for our ED MOUD program was small. In our 7-month review, only 47 patients were identified as potential candidates for MOUD treatment across 70 visits, and only 10 were initiated in the ED. Second, all patients were not actively screened for OUD. There was potential for missing eligible veterans as inclusion criteria relied on clinicians both recognizing OUD and manually entering a correct diagnostic code. We attempted to mitigate this by also reviewing all ED referrals to the SUD clinic and all patients who received buprenorphine in the ED. In addition, we do not have data on preimplementation rates of follow-up for comparison.
Future Directions
More than half of our patients did not receive ED buprenorphine initiation because they were not in moderate or severe withdrawal (COWS ≥ 8) similar to 57% of patients cited in the D’Onofrio and colleagues study.7 Teaching veterans how to start buprenorphine at home could greatly expand enrollment. However, this requires a prescription from an X-waiver licensed clinician. In 2021, the US Department of Health and Human Services removed the 8-hour training requirement for obtaining an X-waiver.12 However, clinicians are still required to apply for licensing. Eliminating the X-waiver requirement, as proposed by D’Onofrio and colleagues in a 2021 editorial, would have allowed all clinicians to offer home initiation.13
Previous studies suggest that despite the ability to provide a prescription, clinicians may be reluctant to offer home initiation.14–17 In a national VA 2019 survey, many emergency medicine physicians believe that SUD care is not in their scope of practice, as Dieujuste and colleagues described in Federal Practitioner.14 Although it is likely some attitudes have changed with the increased visibility of ED MOUD programs, there is still much work to be done to change perceptions.
Another area for improvement is screening for OUD in the ED to better reveal MOUD candidates. Missed opportunities (neither referral nor treatment offered) occurred in 21% of our visits. D’Onofrio and colleagues identified 66% of patients by screening all ED patients.7 Although universal screening for SUD in routine health care settings has been recommended, 2021 VA guidelines state that there is insufficient evidence to recommend universal screening.18-20 There are also limited data on the best screening tool for OUD in the ED.21 Further research on how to effectively and efficiently identify OUD patients in the ED is needed.
Conclusions
With minimal resource allocation, we started the program to offer MOUD with buprenorphine for patients with OUD at a VA ED and provided addiction treatment follow-up. This program, the first of its kind within VA, can be modeled and expanded to other VA facilities. Given increasing numbers of fatal opioid overdose, and significant adverse impacts of the COVID-19 pandemic on the OUD crisis, developing local and national strategies to treat OUD is essential. Future steps include improved screening and expanding capacity to offer home initiation by increasing the number of X-waiver ED clinicians.6
Acknowledgments
Thank you to Jeffrey Balsam, PharmD, BCPS, Veterans Affairs Greater Los Angeles Clinical Applications Coordinator for his contributions in creating a Computerized Patient Record System opioid use disorder screening tool. Thank you to Gracielle Tan, MD, Veterans Affairs Greater Los Angeles Health Science Specialist for her administrative assistance in manuscript preparation.
1. Wyse JJ, Gordon AJ, Dobscha SK, et al. Medications for opioid use disorder in the Department of Veterans Affairs (VA) health care system: historical perspective, lessons learned, and next steps. Subst Abuse. 2018;39(2):139-144. doi:10.1080/08897077.2018.1452327
2. Bohnert ASB, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs health system. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. Ma J, Bao Y-P, Wang R-J, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1968-1983. doi:10.1038/s41380-018-0094-5
4. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 2.0. US Department of Veterans Affairs; 2009.
5. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 3.0. US Department of Veterans Affairs. 2015. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPGRevised22216.pdf
6. Hulsey J, Mellis A, Kelly B. COVID-19 pandemic impact on patients, families and individuals in recovery from substance use disorder. Accessed July 7, 2021. https://www.addictionpolicy.org/covid19-report
7. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opiod dependence. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
8. Weiner SG, Baker O, Bernson D, Schuur JD. One-year mortality of patients after emergency department treatment for non-fatal opioid overdose. Ann Emerg Med. 2020;75(1):13-17. doi:10.1016/j.annemergmed.2019.04.020
9. CA Bridge. Updated 2021. Accessed July 1, 2022. https://cabridge.org
10. Penney L, Miake-Lye I, Lewis D, et al. Proceedings from the 11th annual conference on the science of dissemination and implementation: S72 spreading VA’s emergency department-rapid access clinics (ED-RAC) intervention: key factors for success. Implementation Sci. 2019;14(suppl 1). doi:10.1186/s13012-019-0878-2
11. Hu T, Snider-Alder M, Nijmeh L, Pyle A. Buprenorphine/naloxone induction in a Canadian emergency department with rapid access to community-based addictions providers. CJEM. 2019;21(4):492-498. doi:10.1017/cem.2019.24
12. US Department of Health and Human Services. Practice Guidelines for the Administration of Buprenorphine for Treating Opioid Use Disorder. Federal Register. Accessed July 1, 2022. https://www.federalregister.gov/documents/2021/04/28/2021-08961/practice-guidelines-for-the-administration-of-buprenorphine-for-treating-opioid-use-disorder
13. D’Onofrio G, Melnick ER, Hawk KF. Improve access to care for opioid use disorder: a call to eliminate the x-waiver requirement now. Ann Emerg Med. 2021;78(2):220-222. doi:10.1016/j.annemergmed.2021.03.023
14. Dieujuste N, Johnson-Koenke R, Celedon M, et al. Provider perceptions of opioid safety measures in VHA emergency department and urgent care centers. Fed Pract. 2021;38(9):412-419. doi:10.12788/fp.0179
15. Hawk KF, D’Onofrio G, Chawarski MC, et al. Barriers and faciliatators to clinician readiness to provide emergency department-initiated buprenorphine. JAMA Netw Open. 2020;3(5):e204561. doi:10.1001/jamanetworkopen.2020.4561
16. Lowenstein M, Kilaru A, Perrone J, et al. Barriers and facilitators for emergency department initiation of buprenorphine: a physician survey. Am J Emerg Med. 2019;37(9):1787-1790. doi:10.1016/j.ajem.2019.02.025
17. Srivastava A, Kahan M, Leece P, McAndrew A. Buprenorphine unobserved “home” induction: a survey of Ontario’s addiction physicians. Addic Sci Clin Pract. 2019;14(1):18. doi:10.1186/s13722-019-0146-4
18. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 4.0. US Department of Veterans Affairs. 2021. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPG.pdf
19. Patnode CD, Perdue LA, Rushkin M, et al. Screening for unhealthy drug use updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;323(22):2310-2338. doi:10.1001/jama.2019.21381
20. Coles S, Vosooney A. Evidence lacking to support universal unhealthy drug use screening. Am Fam Physician. 2021;103(2):72-73.
21. Sahota PK, Sharstry S, Mukamel DB, et al. Screening emergency department patients for opioid drug use: a qualitative systematic review. Addict Behav. 2018;85:139-146. doi:10.1016/j.addbeh.2018.05.022
1. Wyse JJ, Gordon AJ, Dobscha SK, et al. Medications for opioid use disorder in the Department of Veterans Affairs (VA) health care system: historical perspective, lessons learned, and next steps. Subst Abuse. 2018;39(2):139-144. doi:10.1080/08897077.2018.1452327
2. Bohnert ASB, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs health system. Med Care. 2011;49(4):393-396. doi:10.1097/MLR.0b013e318202aa27
3. Ma J, Bao Y-P, Wang R-J, et al. Effects of medication-assisted treatment on mortality among opioids users: a systematic review and meta-analysis. Mol Psychiatry. 2019;24(12):1968-1983. doi:10.1038/s41380-018-0094-5
4. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 2.0. US Department of Veterans Affairs; 2009.
5. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 3.0. US Department of Veterans Affairs. 2015. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPGRevised22216.pdf
6. Hulsey J, Mellis A, Kelly B. COVID-19 pandemic impact on patients, families and individuals in recovery from substance use disorder. Accessed July 7, 2021. https://www.addictionpolicy.org/covid19-report
7. D’Onofrio G, O’Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opiod dependence. JAMA. 2015;313(16):1636-1644. doi:10.1001/jama.2015.3474
8. Weiner SG, Baker O, Bernson D, Schuur JD. One-year mortality of patients after emergency department treatment for non-fatal opioid overdose. Ann Emerg Med. 2020;75(1):13-17. doi:10.1016/j.annemergmed.2019.04.020
9. CA Bridge. Updated 2021. Accessed July 1, 2022. https://cabridge.org
10. Penney L, Miake-Lye I, Lewis D, et al. Proceedings from the 11th annual conference on the science of dissemination and implementation: S72 spreading VA’s emergency department-rapid access clinics (ED-RAC) intervention: key factors for success. Implementation Sci. 2019;14(suppl 1). doi:10.1186/s13012-019-0878-2
11. Hu T, Snider-Alder M, Nijmeh L, Pyle A. Buprenorphine/naloxone induction in a Canadian emergency department with rapid access to community-based addictions providers. CJEM. 2019;21(4):492-498. doi:10.1017/cem.2019.24
12. US Department of Health and Human Services. Practice Guidelines for the Administration of Buprenorphine for Treating Opioid Use Disorder. Federal Register. Accessed July 1, 2022. https://www.federalregister.gov/documents/2021/04/28/2021-08961/practice-guidelines-for-the-administration-of-buprenorphine-for-treating-opioid-use-disorder
13. D’Onofrio G, Melnick ER, Hawk KF. Improve access to care for opioid use disorder: a call to eliminate the x-waiver requirement now. Ann Emerg Med. 2021;78(2):220-222. doi:10.1016/j.annemergmed.2021.03.023
14. Dieujuste N, Johnson-Koenke R, Celedon M, et al. Provider perceptions of opioid safety measures in VHA emergency department and urgent care centers. Fed Pract. 2021;38(9):412-419. doi:10.12788/fp.0179
15. Hawk KF, D’Onofrio G, Chawarski MC, et al. Barriers and faciliatators to clinician readiness to provide emergency department-initiated buprenorphine. JAMA Netw Open. 2020;3(5):e204561. doi:10.1001/jamanetworkopen.2020.4561
16. Lowenstein M, Kilaru A, Perrone J, et al. Barriers and facilitators for emergency department initiation of buprenorphine: a physician survey. Am J Emerg Med. 2019;37(9):1787-1790. doi:10.1016/j.ajem.2019.02.025
17. Srivastava A, Kahan M, Leece P, McAndrew A. Buprenorphine unobserved “home” induction: a survey of Ontario’s addiction physicians. Addic Sci Clin Pract. 2019;14(1):18. doi:10.1186/s13722-019-0146-4
18. The Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Version 4.0. US Department of Veterans Affairs. 2021. Accessed July 1, 2022. https://www.healthquality.va.gov/guidelines/MH/sud/VADoDSUDCPG.pdf
19. Patnode CD, Perdue LA, Rushkin M, et al. Screening for unhealthy drug use updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;323(22):2310-2338. doi:10.1001/jama.2019.21381
20. Coles S, Vosooney A. Evidence lacking to support universal unhealthy drug use screening. Am Fam Physician. 2021;103(2):72-73.
21. Sahota PK, Sharstry S, Mukamel DB, et al. Screening emergency department patients for opioid drug use: a qualitative systematic review. Addict Behav. 2018;85:139-146. doi:10.1016/j.addbeh.2018.05.022
Soccer player with painful toe
A 13-YEAR-OLD GIRL presented to the clinic with a 1-year history of a slow-growing mass on the third toe of her right foot. As a soccer player, she experienced associated pain when kicking the ball or when wearing tight-fitting shoes. The lesion was otherwise asymptomatic. She denied any overt trauma to the area and indicated that the mass had enlarged over the previous year.
On exam, there was a nontender 8 × 8-mm firm nodule underneath the nail with associated nail dystrophy (FIGURE 1). The toe had full mobility, sensation was intact, and capillary refill time was < 2 seconds.
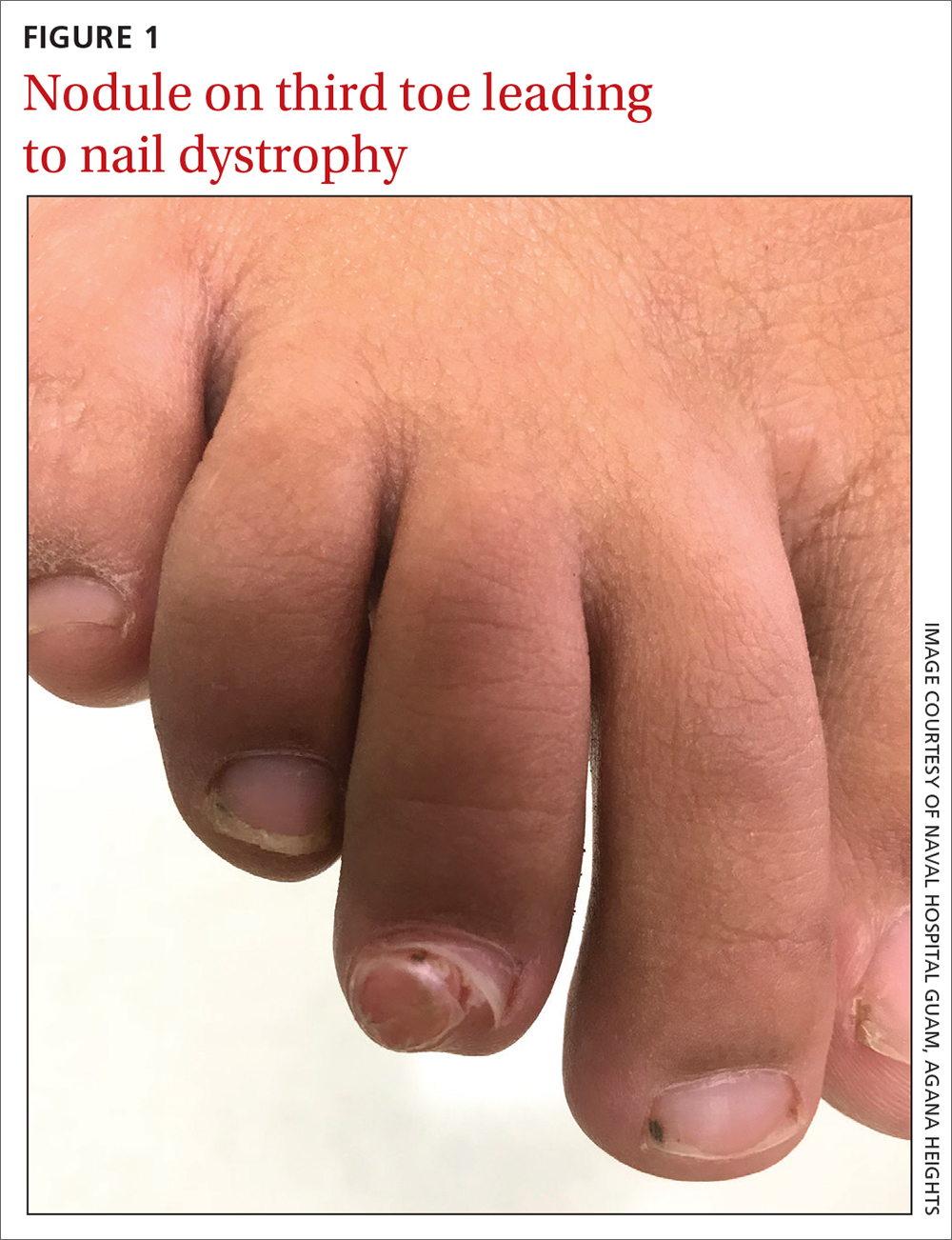
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Subungual exostosis
A plain radiograph of the patient’s foot showed continuity with the bony cortex and medullary space, confirming the diagnosis of subungual exostosis (FIGURE 2).1 An exostosis, or osteochondroma, is a form of benign bone tumor in which trabecular bone overgrows its normal border in a nodular pattern. When this occurs under the nail bed, it is called subungual exostosis.2 Exostosis represents 10% to 15% of all benign bone tumors, making it the most common benign bone tumor.3 Generally, the age of occurrence is 10 to 15 years.3
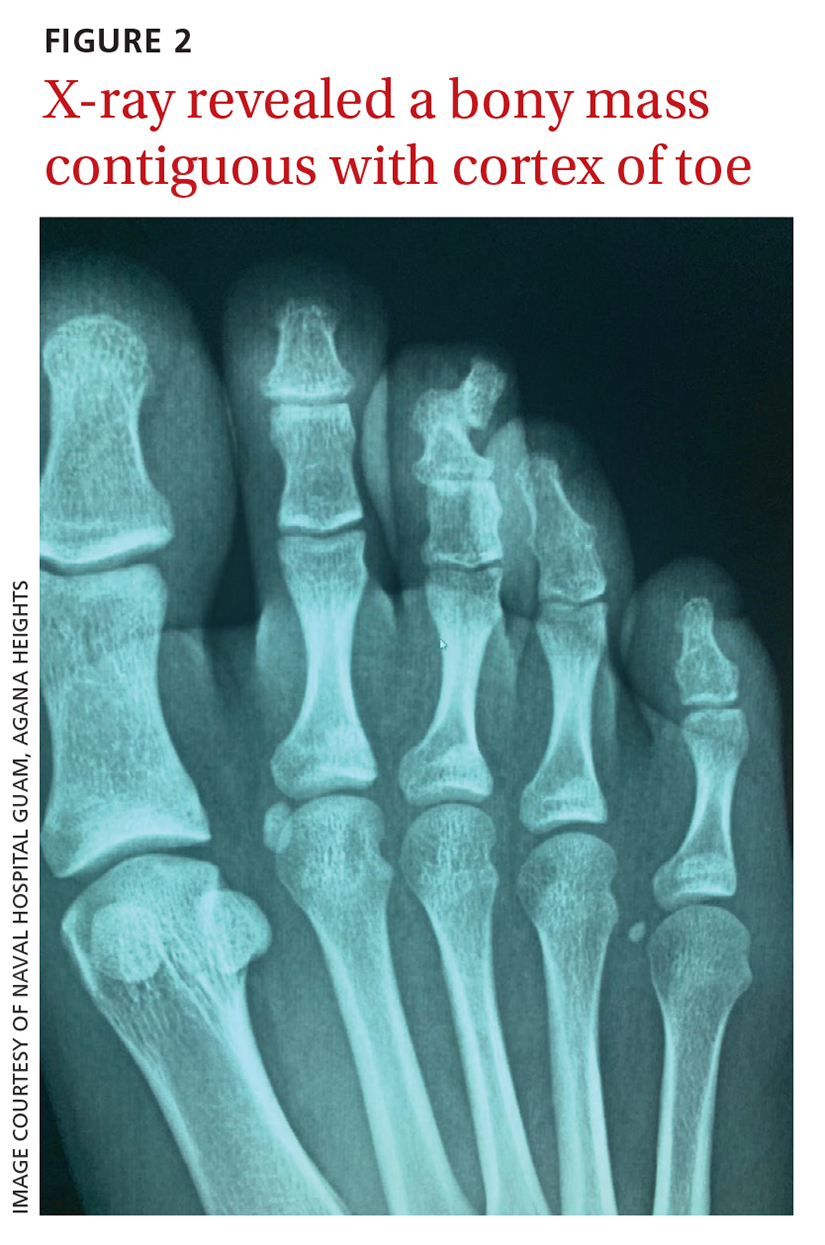
Repetitive trauma can be a culprit. Up to 8% of exostoses occur in the foot, with the most commonly affected area being the distal medial portion of the big toe.3,4 Repetitive trauma and infection are potential risk factors.3,4 The affected toe may be painful, but that is not always the case.4 Typically, lesions are solitary; however, multiple lesions can occur.4
Most pediatric foot lesions are benign and involve soft tissue
Benign soft-tissue masses make up the overwhelming majority of pediatric foot lesions, accounting for 61% to 87% of all foot lesions.3 Malignancies such as chondrosarcoma can occur and can be difficult to diagnose. Rapid growth, family history, size > 5 cm, heterogenous appearance on magnetic resonance imaging, and poorly defined margins are a few characteristics that should increase suspicion for possible malignancy.5
The differential diagnosis for a growth on the toe similar to the one our patient had would include pyogenic granuloma,
Pyogenic granulomas are benign vascular lesions that occur in patients of all ages. They tend to be dome-shaped and flesh-toned to violaceous red, and they are usually found on the head, neck, and extremities—especially fingers.6 They are associated with trauma and are classically tender with a propensity to bleed.6
Acral fibromyxoma is a benign, slow-growing, predominately painless, firm mass with an affinity for the great toe; the affected area includes the nail in 50% of cases.7 A radiograph may show bony erosion or scalloping due to mass effect; however, there will be no continuity with the bony matrix. (Such continuity would suggest exostosis.)
Periungual fibromas are benign soft-tissue masses, which are pink to red and firm, and emerge from underneath the nails, potentially resulting in dystrophy.8 They can bleed and cause pain, and are strongly associated with tuberous sclerosis.5
Continue to: Verruca vulgaris
Verruca vulgaris, the common wart, can also manifest in the subungual region as a firm, generally painless mass. It is the most common neoplasm of the hand and fingers.6 Tiny black dots that correspond to thrombosed capillaries are key to identifying this lesion.
Surgical excision when patient reaches maturity
The definitive treatment for subungual exostosis is surgical excision, preferably once the patient has reached skeletal maturity. Surgery at this point is associated with decreased recurrence rates.3,4 That said, excision may need to be performed sooner if the lesion is painful and leading to deformity.3
Our patient’s persistent pain prompted us to recommend surgical excision. She underwent a third digit exostectomy, which she tolerated without any issues. The patient was fitted with a postoperative shoe that she wore until her 2-week follow-up appointment, when her sutures were removed. The patient’s activity level progressed as tolerated. She regained full function and returned to playing soccer, without any pain, 3 months after her surgery.
1. Das PC, Hassan S, Kumar P. Subungual exostosis – clinical, radiological, and histological findings. Indian Dermatol Online J. 2019;10:202-203. doi: 10.4103/idoj.IDOJ_104_18
2. Yousefian F, Davis B, Browning JC. Pediatric subungual exostosis. Cutis. 2021;108:256-257. doi:10.12788/cutis.0380
3. Bouchard B, Bartlett M, Donnan L. Assessment of the pediatric foot mass. J Am Acad Orthop Surg. 2017;25:32-41. doi: 10.5435/JAAOS-D-15-00397
4. DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi: 10.1007/s11999-013-3345-4
5. Shah SH, Callahan MJ. Ultrasound evaluation of superficial lumps and bumps of the extremities in children: a 5-year retrospective review. Pediatr Radiol. 2013;43 suppl 1:S23-S40. doi: 10.1007/s00247-012-2590-0
6. Habif, Thomas P. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Mosby/Elsevier, 2016.
7. Ramya C, Nayak C, Tambe S. Superficial acral fibromyxoma. Indian J Dermatol. 2016;61:457-459. doi: 10.4103/0019-5154.185734
8. Ma D, Darling T, Moss J, et al. Histologic variants of periungual fibromas in tuberous sclerosis complex. J Am Acad Dermatol. 2011;64:442-444. doi: 10.1016/j.jaad.2010.03.002
A 13-YEAR-OLD GIRL presented to the clinic with a 1-year history of a slow-growing mass on the third toe of her right foot. As a soccer player, she experienced associated pain when kicking the ball or when wearing tight-fitting shoes. The lesion was otherwise asymptomatic. She denied any overt trauma to the area and indicated that the mass had enlarged over the previous year.
On exam, there was a nontender 8 × 8-mm firm nodule underneath the nail with associated nail dystrophy (FIGURE 1). The toe had full mobility, sensation was intact, and capillary refill time was < 2 seconds.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Subungual exostosis
A plain radiograph of the patient’s foot showed continuity with the bony cortex and medullary space, confirming the diagnosis of subungual exostosis (FIGURE 2).1 An exostosis, or osteochondroma, is a form of benign bone tumor in which trabecular bone overgrows its normal border in a nodular pattern. When this occurs under the nail bed, it is called subungual exostosis.2 Exostosis represents 10% to 15% of all benign bone tumors, making it the most common benign bone tumor.3 Generally, the age of occurrence is 10 to 15 years.3

Repetitive trauma can be a culprit. Up to 8% of exostoses occur in the foot, with the most commonly affected area being the distal medial portion of the big toe.3,4 Repetitive trauma and infection are potential risk factors.3,4 The affected toe may be painful, but that is not always the case.4 Typically, lesions are solitary; however, multiple lesions can occur.4
Most pediatric foot lesions are benign and involve soft tissue
Benign soft-tissue masses make up the overwhelming majority of pediatric foot lesions, accounting for 61% to 87% of all foot lesions.3 Malignancies such as chondrosarcoma can occur and can be difficult to diagnose. Rapid growth, family history, size > 5 cm, heterogenous appearance on magnetic resonance imaging, and poorly defined margins are a few characteristics that should increase suspicion for possible malignancy.5
The differential diagnosis for a growth on the toe similar to the one our patient had would include pyogenic granuloma,
Pyogenic granulomas are benign vascular lesions that occur in patients of all ages. They tend to be dome-shaped and flesh-toned to violaceous red, and they are usually found on the head, neck, and extremities—especially fingers.6 They are associated with trauma and are classically tender with a propensity to bleed.6
Acral fibromyxoma is a benign, slow-growing, predominately painless, firm mass with an affinity for the great toe; the affected area includes the nail in 50% of cases.7 A radiograph may show bony erosion or scalloping due to mass effect; however, there will be no continuity with the bony matrix. (Such continuity would suggest exostosis.)
Periungual fibromas are benign soft-tissue masses, which are pink to red and firm, and emerge from underneath the nails, potentially resulting in dystrophy.8 They can bleed and cause pain, and are strongly associated with tuberous sclerosis.5
Continue to: Verruca vulgaris
Verruca vulgaris, the common wart, can also manifest in the subungual region as a firm, generally painless mass. It is the most common neoplasm of the hand and fingers.6 Tiny black dots that correspond to thrombosed capillaries are key to identifying this lesion.
Surgical excision when patient reaches maturity
The definitive treatment for subungual exostosis is surgical excision, preferably once the patient has reached skeletal maturity. Surgery at this point is associated with decreased recurrence rates.3,4 That said, excision may need to be performed sooner if the lesion is painful and leading to deformity.3
Our patient’s persistent pain prompted us to recommend surgical excision. She underwent a third digit exostectomy, which she tolerated without any issues. The patient was fitted with a postoperative shoe that she wore until her 2-week follow-up appointment, when her sutures were removed. The patient’s activity level progressed as tolerated. She regained full function and returned to playing soccer, without any pain, 3 months after her surgery.
A 13-YEAR-OLD GIRL presented to the clinic with a 1-year history of a slow-growing mass on the third toe of her right foot. As a soccer player, she experienced associated pain when kicking the ball or when wearing tight-fitting shoes. The lesion was otherwise asymptomatic. She denied any overt trauma to the area and indicated that the mass had enlarged over the previous year.
On exam, there was a nontender 8 × 8-mm firm nodule underneath the nail with associated nail dystrophy (FIGURE 1). The toe had full mobility, sensation was intact, and capillary refill time was < 2 seconds.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Subungual exostosis
A plain radiograph of the patient’s foot showed continuity with the bony cortex and medullary space, confirming the diagnosis of subungual exostosis (FIGURE 2).1 An exostosis, or osteochondroma, is a form of benign bone tumor in which trabecular bone overgrows its normal border in a nodular pattern. When this occurs under the nail bed, it is called subungual exostosis.2 Exostosis represents 10% to 15% of all benign bone tumors, making it the most common benign bone tumor.3 Generally, the age of occurrence is 10 to 15 years.3

Repetitive trauma can be a culprit. Up to 8% of exostoses occur in the foot, with the most commonly affected area being the distal medial portion of the big toe.3,4 Repetitive trauma and infection are potential risk factors.3,4 The affected toe may be painful, but that is not always the case.4 Typically, lesions are solitary; however, multiple lesions can occur.4
Most pediatric foot lesions are benign and involve soft tissue
Benign soft-tissue masses make up the overwhelming majority of pediatric foot lesions, accounting for 61% to 87% of all foot lesions.3 Malignancies such as chondrosarcoma can occur and can be difficult to diagnose. Rapid growth, family history, size > 5 cm, heterogenous appearance on magnetic resonance imaging, and poorly defined margins are a few characteristics that should increase suspicion for possible malignancy.5
The differential diagnosis for a growth on the toe similar to the one our patient had would include pyogenic granuloma,
Pyogenic granulomas are benign vascular lesions that occur in patients of all ages. They tend to be dome-shaped and flesh-toned to violaceous red, and they are usually found on the head, neck, and extremities—especially fingers.6 They are associated with trauma and are classically tender with a propensity to bleed.6
Acral fibromyxoma is a benign, slow-growing, predominately painless, firm mass with an affinity for the great toe; the affected area includes the nail in 50% of cases.7 A radiograph may show bony erosion or scalloping due to mass effect; however, there will be no continuity with the bony matrix. (Such continuity would suggest exostosis.)
Periungual fibromas are benign soft-tissue masses, which are pink to red and firm, and emerge from underneath the nails, potentially resulting in dystrophy.8 They can bleed and cause pain, and are strongly associated with tuberous sclerosis.5
Continue to: Verruca vulgaris
Verruca vulgaris, the common wart, can also manifest in the subungual region as a firm, generally painless mass. It is the most common neoplasm of the hand and fingers.6 Tiny black dots that correspond to thrombosed capillaries are key to identifying this lesion.
Surgical excision when patient reaches maturity
The definitive treatment for subungual exostosis is surgical excision, preferably once the patient has reached skeletal maturity. Surgery at this point is associated with decreased recurrence rates.3,4 That said, excision may need to be performed sooner if the lesion is painful and leading to deformity.3
Our patient’s persistent pain prompted us to recommend surgical excision. She underwent a third digit exostectomy, which she tolerated without any issues. The patient was fitted with a postoperative shoe that she wore until her 2-week follow-up appointment, when her sutures were removed. The patient’s activity level progressed as tolerated. She regained full function and returned to playing soccer, without any pain, 3 months after her surgery.
1. Das PC, Hassan S, Kumar P. Subungual exostosis – clinical, radiological, and histological findings. Indian Dermatol Online J. 2019;10:202-203. doi: 10.4103/idoj.IDOJ_104_18
2. Yousefian F, Davis B, Browning JC. Pediatric subungual exostosis. Cutis. 2021;108:256-257. doi:10.12788/cutis.0380
3. Bouchard B, Bartlett M, Donnan L. Assessment of the pediatric foot mass. J Am Acad Orthop Surg. 2017;25:32-41. doi: 10.5435/JAAOS-D-15-00397
4. DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi: 10.1007/s11999-013-3345-4
5. Shah SH, Callahan MJ. Ultrasound evaluation of superficial lumps and bumps of the extremities in children: a 5-year retrospective review. Pediatr Radiol. 2013;43 suppl 1:S23-S40. doi: 10.1007/s00247-012-2590-0
6. Habif, Thomas P. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Mosby/Elsevier, 2016.
7. Ramya C, Nayak C, Tambe S. Superficial acral fibromyxoma. Indian J Dermatol. 2016;61:457-459. doi: 10.4103/0019-5154.185734
8. Ma D, Darling T, Moss J, et al. Histologic variants of periungual fibromas in tuberous sclerosis complex. J Am Acad Dermatol. 2011;64:442-444. doi: 10.1016/j.jaad.2010.03.002
1. Das PC, Hassan S, Kumar P. Subungual exostosis – clinical, radiological, and histological findings. Indian Dermatol Online J. 2019;10:202-203. doi: 10.4103/idoj.IDOJ_104_18
2. Yousefian F, Davis B, Browning JC. Pediatric subungual exostosis. Cutis. 2021;108:256-257. doi:10.12788/cutis.0380
3. Bouchard B, Bartlett M, Donnan L. Assessment of the pediatric foot mass. J Am Acad Orthop Surg. 2017;25:32-41. doi: 10.5435/JAAOS-D-15-00397
4. DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi: 10.1007/s11999-013-3345-4
5. Shah SH, Callahan MJ. Ultrasound evaluation of superficial lumps and bumps of the extremities in children: a 5-year retrospective review. Pediatr Radiol. 2013;43 suppl 1:S23-S40. doi: 10.1007/s00247-012-2590-0
6. Habif, Thomas P. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Mosby/Elsevier, 2016.
7. Ramya C, Nayak C, Tambe S. Superficial acral fibromyxoma. Indian J Dermatol. 2016;61:457-459. doi: 10.4103/0019-5154.185734
8. Ma D, Darling T, Moss J, et al. Histologic variants of periungual fibromas in tuberous sclerosis complex. J Am Acad Dermatol. 2011;64:442-444. doi: 10.1016/j.jaad.2010.03.002
Using SNRIs to prevent migraines in patients with depression
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.
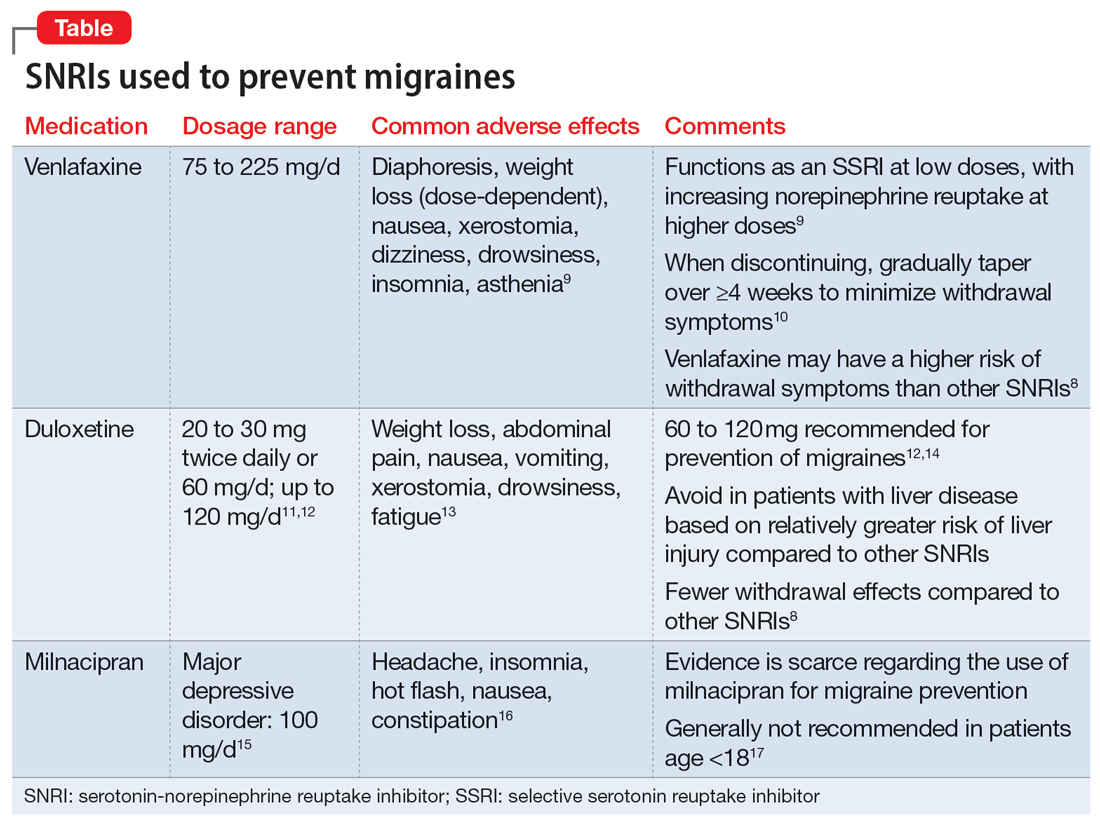
Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.

Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.

Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.