User login
Are Beta-Blockers Safe for COPD?
Everyone takes a pharmacology class in medical school that includes a lecture on beta receptors. They’re in the heart (beta-1) and lungs (beta-2), and drug compounds agonize or antagonize one or both. The professor will caution against using antagonists (beta blockade) for patients with chronic obstructive pulmonary disease (COPD) lest they further impair the patient’s irreversibly narrowed airways. Obsequious students mature into obsequious doctors, intent on “doing no harm.” For better or worse, you withhold beta-blockers from your patient with COPD and comorbid cardiac disease.
Perhaps because the pulmonologist isn’t usually the one who decides whether a beta-blocker is prescribed, I’ve been napping on this topic since training. Early in fellowship, I read an ACP Journal Club article about a Cochrane systematic review (yes, I read a review of a review) that concluded that beta-blockers are fine in patients with COPD. The summary appealed to my bias towards evidence-based medicine (EBM) supplanting physiology, medical school, and everything else. I was more apt to believe my stodgy residency attendings than the stodgy pharmacology professor. Even though COPD and cardiovascular disease share multiple risk factors, I had never reinvestigated the relationship between beta-blockers and COPD.
Turns out that while I was sleeping, the debate continued. Go figure. Just last month a prospective, observational study published in JAMA Network Open found that beta-blockers did not increase the risk for cardiovascular or respiratory events among patients with COPD being discharged after hospitalization for acute myocardial infarction. Although this could be viewed as a triumph for EBM over physiology and a validation of my decade-plus of intellectual laziness, the results are actually pretty thin. These studies, in which patients with an indication for a therapy (a beta-blocker in this case) are analyzed by whether or not they received it, are problematic. The fanciest statistics — in this case, they used propensity scores — can’t control for residual confounding. What drove the physicians to prescribe in some cases but not others? We can only guess.
This might be okay if there hadn’t been a randomized controlled trial (RCT) published in 2019 in The New England Journal of Medicine that found that beta-blockers increase the risk for severe COPD exacerbations. In EBM, the RCT trumps all. Ironically, this trial was designed to test whether beta-blockers reduce severe COPD exacerbations. Yes, we’d come full circle. There was enough biologic plausibility to support a positive effect, or so thought the study authors and the Department of Defense (DOD) — for reasons I can’t possibly guess, the DOD funded this RCT. My pharmacology professor must be rolling over in his tenure.
The RCT did leave beta-blockers some wiggle room. The authors purposely excluded anyone with a cardiovascular indication for a beta-blocker. The intent was to ensure beneficial effects were isolated to respiratory and not cardiovascular outcomes. Of course, the reason I’m writing and you’re reading this is that COPD and cardiovascular disease co-occur at a high rate. The RCT notwithstanding, we prescribe beta-blockers to patients with COPD because they have a cardiac indication, not to reduce acute COPD exacerbations. So, it’s possible there’d be a net beta-blocker benefit in patients with COPD and comorbid heart disease.
That’s where the JAMA Network Open study comes in, but as discussed, methodologic weaknesses preclude its being the final word. That said, I think it’s unlikely we’ll see a COPD with comorbid cardiac disease RCT performed to assess whether beta-blockers provide a net benefit, unless maybe the DOD wants to fund another one of these. In the meantime, I’m calling clinical equipoise and punting. Fortunately for me, I don’t have to prescribe beta-blockers.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
Everyone takes a pharmacology class in medical school that includes a lecture on beta receptors. They’re in the heart (beta-1) and lungs (beta-2), and drug compounds agonize or antagonize one or both. The professor will caution against using antagonists (beta blockade) for patients with chronic obstructive pulmonary disease (COPD) lest they further impair the patient’s irreversibly narrowed airways. Obsequious students mature into obsequious doctors, intent on “doing no harm.” For better or worse, you withhold beta-blockers from your patient with COPD and comorbid cardiac disease.
Perhaps because the pulmonologist isn’t usually the one who decides whether a beta-blocker is prescribed, I’ve been napping on this topic since training. Early in fellowship, I read an ACP Journal Club article about a Cochrane systematic review (yes, I read a review of a review) that concluded that beta-blockers are fine in patients with COPD. The summary appealed to my bias towards evidence-based medicine (EBM) supplanting physiology, medical school, and everything else. I was more apt to believe my stodgy residency attendings than the stodgy pharmacology professor. Even though COPD and cardiovascular disease share multiple risk factors, I had never reinvestigated the relationship between beta-blockers and COPD.
Turns out that while I was sleeping, the debate continued. Go figure. Just last month a prospective, observational study published in JAMA Network Open found that beta-blockers did not increase the risk for cardiovascular or respiratory events among patients with COPD being discharged after hospitalization for acute myocardial infarction. Although this could be viewed as a triumph for EBM over physiology and a validation of my decade-plus of intellectual laziness, the results are actually pretty thin. These studies, in which patients with an indication for a therapy (a beta-blocker in this case) are analyzed by whether or not they received it, are problematic. The fanciest statistics — in this case, they used propensity scores — can’t control for residual confounding. What drove the physicians to prescribe in some cases but not others? We can only guess.
This might be okay if there hadn’t been a randomized controlled trial (RCT) published in 2019 in The New England Journal of Medicine that found that beta-blockers increase the risk for severe COPD exacerbations. In EBM, the RCT trumps all. Ironically, this trial was designed to test whether beta-blockers reduce severe COPD exacerbations. Yes, we’d come full circle. There was enough biologic plausibility to support a positive effect, or so thought the study authors and the Department of Defense (DOD) — for reasons I can’t possibly guess, the DOD funded this RCT. My pharmacology professor must be rolling over in his tenure.
The RCT did leave beta-blockers some wiggle room. The authors purposely excluded anyone with a cardiovascular indication for a beta-blocker. The intent was to ensure beneficial effects were isolated to respiratory and not cardiovascular outcomes. Of course, the reason I’m writing and you’re reading this is that COPD and cardiovascular disease co-occur at a high rate. The RCT notwithstanding, we prescribe beta-blockers to patients with COPD because they have a cardiac indication, not to reduce acute COPD exacerbations. So, it’s possible there’d be a net beta-blocker benefit in patients with COPD and comorbid heart disease.
That’s where the JAMA Network Open study comes in, but as discussed, methodologic weaknesses preclude its being the final word. That said, I think it’s unlikely we’ll see a COPD with comorbid cardiac disease RCT performed to assess whether beta-blockers provide a net benefit, unless maybe the DOD wants to fund another one of these. In the meantime, I’m calling clinical equipoise and punting. Fortunately for me, I don’t have to prescribe beta-blockers.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
Everyone takes a pharmacology class in medical school that includes a lecture on beta receptors. They’re in the heart (beta-1) and lungs (beta-2), and drug compounds agonize or antagonize one or both. The professor will caution against using antagonists (beta blockade) for patients with chronic obstructive pulmonary disease (COPD) lest they further impair the patient’s irreversibly narrowed airways. Obsequious students mature into obsequious doctors, intent on “doing no harm.” For better or worse, you withhold beta-blockers from your patient with COPD and comorbid cardiac disease.
Perhaps because the pulmonologist isn’t usually the one who decides whether a beta-blocker is prescribed, I’ve been napping on this topic since training. Early in fellowship, I read an ACP Journal Club article about a Cochrane systematic review (yes, I read a review of a review) that concluded that beta-blockers are fine in patients with COPD. The summary appealed to my bias towards evidence-based medicine (EBM) supplanting physiology, medical school, and everything else. I was more apt to believe my stodgy residency attendings than the stodgy pharmacology professor. Even though COPD and cardiovascular disease share multiple risk factors, I had never reinvestigated the relationship between beta-blockers and COPD.
Turns out that while I was sleeping, the debate continued. Go figure. Just last month a prospective, observational study published in JAMA Network Open found that beta-blockers did not increase the risk for cardiovascular or respiratory events among patients with COPD being discharged after hospitalization for acute myocardial infarction. Although this could be viewed as a triumph for EBM over physiology and a validation of my decade-plus of intellectual laziness, the results are actually pretty thin. These studies, in which patients with an indication for a therapy (a beta-blocker in this case) are analyzed by whether or not they received it, are problematic. The fanciest statistics — in this case, they used propensity scores — can’t control for residual confounding. What drove the physicians to prescribe in some cases but not others? We can only guess.
This might be okay if there hadn’t been a randomized controlled trial (RCT) published in 2019 in The New England Journal of Medicine that found that beta-blockers increase the risk for severe COPD exacerbations. In EBM, the RCT trumps all. Ironically, this trial was designed to test whether beta-blockers reduce severe COPD exacerbations. Yes, we’d come full circle. There was enough biologic plausibility to support a positive effect, or so thought the study authors and the Department of Defense (DOD) — for reasons I can’t possibly guess, the DOD funded this RCT. My pharmacology professor must be rolling over in his tenure.
The RCT did leave beta-blockers some wiggle room. The authors purposely excluded anyone with a cardiovascular indication for a beta-blocker. The intent was to ensure beneficial effects were isolated to respiratory and not cardiovascular outcomes. Of course, the reason I’m writing and you’re reading this is that COPD and cardiovascular disease co-occur at a high rate. The RCT notwithstanding, we prescribe beta-blockers to patients with COPD because they have a cardiac indication, not to reduce acute COPD exacerbations. So, it’s possible there’d be a net beta-blocker benefit in patients with COPD and comorbid heart disease.
That’s where the JAMA Network Open study comes in, but as discussed, methodologic weaknesses preclude its being the final word. That said, I think it’s unlikely we’ll see a COPD with comorbid cardiac disease RCT performed to assess whether beta-blockers provide a net benefit, unless maybe the DOD wants to fund another one of these. In the meantime, I’m calling clinical equipoise and punting. Fortunately for me, I don’t have to prescribe beta-blockers.
Dr. Holley is professor of medicine at Uniformed Services University in Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center in Washington, DC. He reported conflicts of interest with Metapharm, CHEST College, and WebMD.
A version of this article first appeared on Medscape.com.
More Illnesses Possible Related Linked to Counterfeit Botulinum Toxin Reported
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
— two in the intensive care unit. None of the cases required intubation, according to an announcement of an investigation into these reports in by the Centers for Disease Control and Prevention (CDC).
The report, published online in the Morbidity and Mortality Weekly Report, notes that the four patients in Tennessee received counterfeit BoNT, while product information was not available for the three cases in New York City. “However, one person reported paying less than US wholesale acquisition cost for the administered product, and another reported that the product had been purchased overseas,” the authors of the report wrote. The development underscores that BoNT injections “should be administered only by licensed and trained providers using recommended doses of FDA [Food and Drug Admininstration]-approved products.”
This report follows a CDC advisory published in April 2024 of at least 22 people from 11 states who reported serious reactions after receiving botulinum toxin injections from unlicensed or untrained individuals or in nonhealthcare settings, such as homes and spas.
The median age of the women in the July report was 48 years, and signs and symptoms included ptosis, dry mouth, dysphagia, shortness of breath, and weakness. Onset occurred between February 23 and March 7, 2024.
“This investigation did not determine why these illnesses occurred after cosmetic BoNT injections; potential reasons might include use of counterfeit BoNT, which might be more potent or contain harmful additional ingredients or higher susceptibility to BoNT effects among some persons,” the investigators wrote. They recommended further studies to describe the clinical spectrum of cosmetic BoNT injection effects such as severity of signs and symptoms.
For cases of suspected systemic botulism, the CDC recommends calling the local or state health department for consultation and antitoxin release (as well as information on reporting adverse events). Alternatively, the 24/7 phone number for the CDC clinical botulism service is 770-488-7100.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Cost of Drugs Can Be Breathtaking for COPD Patients
For patients with chronic obstructive pulmonary disease (COPD), the Global Initiative on Obstructive Lung Disease recommends long-term term pharmacologic and nonpharmacologic therapies based on each patient’s symptoms and disease severity.
Yet even the most effective drugs work only when patients take them as directed, and according to the World Health Organization, fewer than half of all patients worldwide are fully compliant with long-term COPD drug regimens.
And as a recent cross-sectional study showed, nearly one in six patients in the United States reported missing a COPD drug dose, lowering the dose, or delaying filling a prescription for financial reasons.
“I care for patients with COPD as their pulmonologist, and this is a very common problem that we see in clinical practice,” said Meredith McCormack, MD, a pulmonary and critical care medicine physician and associate director of the Pulmonary & Critical Care Medicine Division at Johns Hopkins University in Baltimore.
Dr. McCormack, a national spokesperson for The American Lung Association, said that she shows new patients the photos of all available inhalers and asks which ones they have and how they take them.
“I would say that a majority of the time people are taking their medicines slightly differently than prescribed, and often, this is due to cost,” she said.
Serious Consequences
Cost-related medication nonadherence (CRN), as investigators term it, can have major health effects and can be significantly more costly in the long run due to increased hospitalization rates, higher morbidity, and greater risk for COPD-related death associated with suboptimal care.
“For some patients even a month or two of being off medications increases the risk of having exacerbations, having more symptoms, [and] having a decline in their lung function,” said Douglas M. Beach, MD, a pulmonologist at Beth Israel Deaconess Medical Center in Boston.
In the aforementioned cross-sectional study, published in the open access journal BMC Public Health, Xin Wen, MD, from the Jiamusi (China) University School of Public Health, and colleagues looked at data on a representative sample of US adults who participated in the US National Health Interview Survey from 2013 through 2020.
The sample included 15,928 persons aged 18 years or older with a self-reported history of COPD who completed a CRN survey including the following questions:
During the past 12 months, have you
- Skipped medication doses to save money?
- Taken less medicine than prescribed to save money?
- Delayed filling a prescription to save money?
The investigators found that a weighted 18.56% of participants representing 2.39 million persons with COPD answered “yes” to one of the questions.
Translated into representative population numbers, that works out to an estimated 1.61 million persons with COPD missing doses, 1.72 million taking lower doses than those prescribed, and 2.03 million delaying filling prescriptions to save money.
A multivariable logistic regression analysis showed that those who were most likely to be nonadherent for financial reasons were patients younger than 65 years, women, persons with low family income, those who lacked health insurance, and patients with multiple comorbidities, the authors found.
Financial Barriers
One of the biggest barriers to COPD medication adherence is, somewhat paradoxically, insurance status, particularly Medicare, said Corinne Young, MSN, FNP-C, FCCP, from Colorado Springs Pulmonary Consultants.
“What’s so unfair about Medicare is that patients have to buy a drug plan, so they have to already pay for an extra plan to have access to drugs, and the plans vary because there are so many choices,” she said in an interview.
Elderly patients may be confused about the available options and may choose the Medicare Advantage plan with the lowest monthly premiums, which have the highest annual deductibles, usually in the $5000-and-up range, she said.
In addition, the Medicare Part D prescription coverage gap, commonly known as the “donut hole,” requires patients to pay a percentage of drug costs above a certain limit ($5030 in 2024) until a yearly out-of-pocket limit (currently $8000) is reached, after which the plan will again pick up most of the costs.
Although makers of inhalers have voluntarily agreed to limit monthly co-pays to $35 for uninsured patients, Medicare plans require insured patients to shell out considerably more, with 30 days of Trelegy Ellipta (fluticasone furoate, umeclidinium, and vilanterol) setting patients back nearly $350 per month, according to a recent search of costs for a United Healthcare Medicare Advantage plan.
Chasing Lower-Cost Options
“I have a lot of patients who use Canadian pharmacies to try to get around it, and I have a lot of patients who make a trip to Mexico every year and load up. I have patients who don’t take their inhalers as they are supposed to in order to make them last longer, and I have patients who take the inhalers of other family members,” Ms. Young said.
Humayun Anjum, MD, FCCP, from Dallas Pulmonary and Critical Care in Dallas, Texas, said in an interview that when patients claim that a prescribed drug isn’t working as expected for them, financial pressures may be partly to blame.
“When you investigate a little bit more, that’s where things become a bit more clear, and the patient may say ‘yeah, I stopped using this inhaler because it was costing me 200 bucks a month and I’m already on other medications,’ ” he said.
He noted that, when possible, he will steer patients toward discount prescription services such as GoodRx, which offers discounts at local pharmacies, or Mark Cuban Cost Plus Drugs, an online pharmacy that offers generic versions of Advair Diskus (fluticasone propionate/salmeterol) at a 100-50 mcg dose for $94.70, a savings of $307.30 over retail pharmacies, according to the company’s website.
Dr. Beach noted that Beth Israel Deaconess has a pharmacist embedded in the pulmonary clinic who can help eligible patients get financial assistance to pay for their medications.
“The influencing factors of CRN are multifaceted and necessitating more rigorous research. Health policy interventions focusing on reducing drug costs, delaying disease progression, preventing exacerbations, and reducing the risk of comorbidities may improve the economic burden of COPD and its outcomes,” Dr. Wen and colleagues wrote.
The study by Dr. Wen and colleagues was funded by grants from Chinese national and academic sources. Dr. McCormack has served as a consultant to Aridis, Boehringer Ingelheim, GlaxoSmithKline, MCG Diagnostics, ndd Medical Technologies, and UpToDate. Ms. Young, Dr. Anjum, and Dr. Beach reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
For patients with chronic obstructive pulmonary disease (COPD), the Global Initiative on Obstructive Lung Disease recommends long-term term pharmacologic and nonpharmacologic therapies based on each patient’s symptoms and disease severity.
Yet even the most effective drugs work only when patients take them as directed, and according to the World Health Organization, fewer than half of all patients worldwide are fully compliant with long-term COPD drug regimens.
And as a recent cross-sectional study showed, nearly one in six patients in the United States reported missing a COPD drug dose, lowering the dose, or delaying filling a prescription for financial reasons.
“I care for patients with COPD as their pulmonologist, and this is a very common problem that we see in clinical practice,” said Meredith McCormack, MD, a pulmonary and critical care medicine physician and associate director of the Pulmonary & Critical Care Medicine Division at Johns Hopkins University in Baltimore.
Dr. McCormack, a national spokesperson for The American Lung Association, said that she shows new patients the photos of all available inhalers and asks which ones they have and how they take them.
“I would say that a majority of the time people are taking their medicines slightly differently than prescribed, and often, this is due to cost,” she said.
Serious Consequences
Cost-related medication nonadherence (CRN), as investigators term it, can have major health effects and can be significantly more costly in the long run due to increased hospitalization rates, higher morbidity, and greater risk for COPD-related death associated with suboptimal care.
“For some patients even a month or two of being off medications increases the risk of having exacerbations, having more symptoms, [and] having a decline in their lung function,” said Douglas M. Beach, MD, a pulmonologist at Beth Israel Deaconess Medical Center in Boston.
In the aforementioned cross-sectional study, published in the open access journal BMC Public Health, Xin Wen, MD, from the Jiamusi (China) University School of Public Health, and colleagues looked at data on a representative sample of US adults who participated in the US National Health Interview Survey from 2013 through 2020.
The sample included 15,928 persons aged 18 years or older with a self-reported history of COPD who completed a CRN survey including the following questions:
During the past 12 months, have you
- Skipped medication doses to save money?
- Taken less medicine than prescribed to save money?
- Delayed filling a prescription to save money?
The investigators found that a weighted 18.56% of participants representing 2.39 million persons with COPD answered “yes” to one of the questions.
Translated into representative population numbers, that works out to an estimated 1.61 million persons with COPD missing doses, 1.72 million taking lower doses than those prescribed, and 2.03 million delaying filling prescriptions to save money.
A multivariable logistic regression analysis showed that those who were most likely to be nonadherent for financial reasons were patients younger than 65 years, women, persons with low family income, those who lacked health insurance, and patients with multiple comorbidities, the authors found.
Financial Barriers
One of the biggest barriers to COPD medication adherence is, somewhat paradoxically, insurance status, particularly Medicare, said Corinne Young, MSN, FNP-C, FCCP, from Colorado Springs Pulmonary Consultants.
“What’s so unfair about Medicare is that patients have to buy a drug plan, so they have to already pay for an extra plan to have access to drugs, and the plans vary because there are so many choices,” she said in an interview.
Elderly patients may be confused about the available options and may choose the Medicare Advantage plan with the lowest monthly premiums, which have the highest annual deductibles, usually in the $5000-and-up range, she said.
In addition, the Medicare Part D prescription coverage gap, commonly known as the “donut hole,” requires patients to pay a percentage of drug costs above a certain limit ($5030 in 2024) until a yearly out-of-pocket limit (currently $8000) is reached, after which the plan will again pick up most of the costs.
Although makers of inhalers have voluntarily agreed to limit monthly co-pays to $35 for uninsured patients, Medicare plans require insured patients to shell out considerably more, with 30 days of Trelegy Ellipta (fluticasone furoate, umeclidinium, and vilanterol) setting patients back nearly $350 per month, according to a recent search of costs for a United Healthcare Medicare Advantage plan.
Chasing Lower-Cost Options
“I have a lot of patients who use Canadian pharmacies to try to get around it, and I have a lot of patients who make a trip to Mexico every year and load up. I have patients who don’t take their inhalers as they are supposed to in order to make them last longer, and I have patients who take the inhalers of other family members,” Ms. Young said.
Humayun Anjum, MD, FCCP, from Dallas Pulmonary and Critical Care in Dallas, Texas, said in an interview that when patients claim that a prescribed drug isn’t working as expected for them, financial pressures may be partly to blame.
“When you investigate a little bit more, that’s where things become a bit more clear, and the patient may say ‘yeah, I stopped using this inhaler because it was costing me 200 bucks a month and I’m already on other medications,’ ” he said.
He noted that, when possible, he will steer patients toward discount prescription services such as GoodRx, which offers discounts at local pharmacies, or Mark Cuban Cost Plus Drugs, an online pharmacy that offers generic versions of Advair Diskus (fluticasone propionate/salmeterol) at a 100-50 mcg dose for $94.70, a savings of $307.30 over retail pharmacies, according to the company’s website.
Dr. Beach noted that Beth Israel Deaconess has a pharmacist embedded in the pulmonary clinic who can help eligible patients get financial assistance to pay for their medications.
“The influencing factors of CRN are multifaceted and necessitating more rigorous research. Health policy interventions focusing on reducing drug costs, delaying disease progression, preventing exacerbations, and reducing the risk of comorbidities may improve the economic burden of COPD and its outcomes,” Dr. Wen and colleagues wrote.
The study by Dr. Wen and colleagues was funded by grants from Chinese national and academic sources. Dr. McCormack has served as a consultant to Aridis, Boehringer Ingelheim, GlaxoSmithKline, MCG Diagnostics, ndd Medical Technologies, and UpToDate. Ms. Young, Dr. Anjum, and Dr. Beach reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
For patients with chronic obstructive pulmonary disease (COPD), the Global Initiative on Obstructive Lung Disease recommends long-term term pharmacologic and nonpharmacologic therapies based on each patient’s symptoms and disease severity.
Yet even the most effective drugs work only when patients take them as directed, and according to the World Health Organization, fewer than half of all patients worldwide are fully compliant with long-term COPD drug regimens.
And as a recent cross-sectional study showed, nearly one in six patients in the United States reported missing a COPD drug dose, lowering the dose, or delaying filling a prescription for financial reasons.
“I care for patients with COPD as their pulmonologist, and this is a very common problem that we see in clinical practice,” said Meredith McCormack, MD, a pulmonary and critical care medicine physician and associate director of the Pulmonary & Critical Care Medicine Division at Johns Hopkins University in Baltimore.
Dr. McCormack, a national spokesperson for The American Lung Association, said that she shows new patients the photos of all available inhalers and asks which ones they have and how they take them.
“I would say that a majority of the time people are taking their medicines slightly differently than prescribed, and often, this is due to cost,” she said.
Serious Consequences
Cost-related medication nonadherence (CRN), as investigators term it, can have major health effects and can be significantly more costly in the long run due to increased hospitalization rates, higher morbidity, and greater risk for COPD-related death associated with suboptimal care.
“For some patients even a month or two of being off medications increases the risk of having exacerbations, having more symptoms, [and] having a decline in their lung function,” said Douglas M. Beach, MD, a pulmonologist at Beth Israel Deaconess Medical Center in Boston.
In the aforementioned cross-sectional study, published in the open access journal BMC Public Health, Xin Wen, MD, from the Jiamusi (China) University School of Public Health, and colleagues looked at data on a representative sample of US adults who participated in the US National Health Interview Survey from 2013 through 2020.
The sample included 15,928 persons aged 18 years or older with a self-reported history of COPD who completed a CRN survey including the following questions:
During the past 12 months, have you
- Skipped medication doses to save money?
- Taken less medicine than prescribed to save money?
- Delayed filling a prescription to save money?
The investigators found that a weighted 18.56% of participants representing 2.39 million persons with COPD answered “yes” to one of the questions.
Translated into representative population numbers, that works out to an estimated 1.61 million persons with COPD missing doses, 1.72 million taking lower doses than those prescribed, and 2.03 million delaying filling prescriptions to save money.
A multivariable logistic regression analysis showed that those who were most likely to be nonadherent for financial reasons were patients younger than 65 years, women, persons with low family income, those who lacked health insurance, and patients with multiple comorbidities, the authors found.
Financial Barriers
One of the biggest barriers to COPD medication adherence is, somewhat paradoxically, insurance status, particularly Medicare, said Corinne Young, MSN, FNP-C, FCCP, from Colorado Springs Pulmonary Consultants.
“What’s so unfair about Medicare is that patients have to buy a drug plan, so they have to already pay for an extra plan to have access to drugs, and the plans vary because there are so many choices,” she said in an interview.
Elderly patients may be confused about the available options and may choose the Medicare Advantage plan with the lowest monthly premiums, which have the highest annual deductibles, usually in the $5000-and-up range, she said.
In addition, the Medicare Part D prescription coverage gap, commonly known as the “donut hole,” requires patients to pay a percentage of drug costs above a certain limit ($5030 in 2024) until a yearly out-of-pocket limit (currently $8000) is reached, after which the plan will again pick up most of the costs.
Although makers of inhalers have voluntarily agreed to limit monthly co-pays to $35 for uninsured patients, Medicare plans require insured patients to shell out considerably more, with 30 days of Trelegy Ellipta (fluticasone furoate, umeclidinium, and vilanterol) setting patients back nearly $350 per month, according to a recent search of costs for a United Healthcare Medicare Advantage plan.
Chasing Lower-Cost Options
“I have a lot of patients who use Canadian pharmacies to try to get around it, and I have a lot of patients who make a trip to Mexico every year and load up. I have patients who don’t take their inhalers as they are supposed to in order to make them last longer, and I have patients who take the inhalers of other family members,” Ms. Young said.
Humayun Anjum, MD, FCCP, from Dallas Pulmonary and Critical Care in Dallas, Texas, said in an interview that when patients claim that a prescribed drug isn’t working as expected for them, financial pressures may be partly to blame.
“When you investigate a little bit more, that’s where things become a bit more clear, and the patient may say ‘yeah, I stopped using this inhaler because it was costing me 200 bucks a month and I’m already on other medications,’ ” he said.
He noted that, when possible, he will steer patients toward discount prescription services such as GoodRx, which offers discounts at local pharmacies, or Mark Cuban Cost Plus Drugs, an online pharmacy that offers generic versions of Advair Diskus (fluticasone propionate/salmeterol) at a 100-50 mcg dose for $94.70, a savings of $307.30 over retail pharmacies, according to the company’s website.
Dr. Beach noted that Beth Israel Deaconess has a pharmacist embedded in the pulmonary clinic who can help eligible patients get financial assistance to pay for their medications.
“The influencing factors of CRN are multifaceted and necessitating more rigorous research. Health policy interventions focusing on reducing drug costs, delaying disease progression, preventing exacerbations, and reducing the risk of comorbidities may improve the economic burden of COPD and its outcomes,” Dr. Wen and colleagues wrote.
The study by Dr. Wen and colleagues was funded by grants from Chinese national and academic sources. Dr. McCormack has served as a consultant to Aridis, Boehringer Ingelheim, GlaxoSmithKline, MCG Diagnostics, ndd Medical Technologies, and UpToDate. Ms. Young, Dr. Anjum, and Dr. Beach reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Measuring Restrictive Lung Disease Severity Using FEV1 vs TLC
Respiratory diseases have varied clinical presentations and are classified as restrictive, obstructive, mixed, or normal. Restrictive lung diseases have reduced lung volumes, either due to an alteration in lung parenchyma or a disease of the pleura, chest wall, or neuromuscular apparatus. If caused by parenchymal lung disease, restrictive lung disorders are accompanied by reduced gas transfer, which may be portrayed clinically by desaturation after exercise. Based on anatomical structures, the causes of lung volume reduction may be intrinsic or extrinsic. Intrinsic causes correspond to diseases of the lung parenchyma, such as idiopathic fibrotic diseases, connective-tissue diseases, drug-induced lung diseases, and other primary diseases of the lungs. Extrinsic causes refer to disorders outside the lungs or extra-pulmonary diseases such as neuromuscular and nonmuscular diseases of the chest wall.1 For example, obesity and myasthenia gravis can cause restrictive lung diseases, one through mechanical interference of lung expansion and the other through neuromuscular impedance of thoracic cage expansion. All these diseases eventually result in lung restriction, impaired lung function, and respiratory failure. This heterogenicity of disease makes establishing a single severity criterion difficult.
Laboratory testing, imaging studies, and examinations are important for determining the pulmonary disease and its course and progression. The pulmonary function test (PFT), which consists of multiple procedures that are performed depending on the information needed, has been an essential tool in practice for the pulmonologist. The PFT includes spirometry, lung volume measurement, respiratory muscle strength, diffusion capacity, and a broncho-provocation test. Each test has a particular role in assisting the diagnosis and/or follow-up of the patient. Spirometry is frequently used due to its range of dynamic physiological parameters, ease of use, and accessibility. It is used for the diagnosis of pulmonary symptoms, in the assessment of disability, and preoperatory evaluation, including lung resection surgery, assisting in the diagnosis, monitoring, and therapy response of pulmonary diseases.
A systematic approach to PFT interpretation is recommended by several societies, such as the American Thoracic Society (ATS) and the European Respiratory Society (ERS).2 The pulmonary function test results must be reproducible and meet established standards to ensure reliable and consistent clinical outcomes. A restrictive respiratory disease is defined by a decrease in total lung capacity (TLC) (< 5% of predicted value) and a normal forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio.2 Although other findings—such as a decrease in vital capacity—should prompt an investigation into whether the patient has a possible restrictive respiratory disease, the sole presence of this parameter is not definitive or diagnostic of a restrictive impairment.2-4 The assessment of severity is typically determined by TLC. Unfortunately, the severity of a restrictive respiratory disease and the degree of patient discomfort do not always correlate when utilizing just TLC. Pulmonary sarcoidosis, for example, is a granulomatous lung disease with a restrictive PFT pattern and a disease burden that may vary over time. Having a more consistent method of grading the severity of the restrictive lung disease may help guide treatment. The modified Medical Research Council (mMRC) scale, a 5-point dyspnea scale, is widely used in assessing the severity of dyspnea in various respiratory conditions, including chronic obstructive pulmonary disease (COPD), where its scores have been associated with patient mortality.1,5 The goal of this study was to document the associations between objective parameters obtained through PFT and other variables, with an established measurement of dyspnea to assess the severity grade of restrictive lung diseases.
Methods
This retrospective record review at the Veterans Affairs Caribbean Healthcare System (VACHS) in San Juan, Puerto Rico, wasconducted using the Veterans Health Information Systems and Technology Architecture to identify patients with a PFT, including spirometry, that indicated a restrictive ventilator pattern based on the current ATS/ERS Task Force on Lung Function Testing.2 Patients were included if they were aged ≥ 21 years, PFT with TLC ≤ 80% predicted, mMRC score documented on PFT, and documented diffusing capacity of the lung for carbon monoxide (DLCO). Patients were excluded if their FEV1/vital capacity (VC) was < 70% predicted using the largest VC, or no mMRC score was available. All patients meeting the inclusion criteria were considered regardless of comorbidities.
The PFT results of all adult patients, including those performed between June 1, 2013, and January 6, 2016, were submitted to spirometry, and lung volume measurements were analyzed. Sociodemographic information was collected, including sex, ethnicity, age, height, weight, and basal metabolic index. Other data found in PFTs, such as smoking status, smoking in packs/year, mMRC score, predicted TLC value, imaging present (chest X-ray, computed tomography), and hospitalizations and exacerbations within 1 year were collected. In addition, we examined the predicted values for FEV1, DLCO, and DLCO/VA (calculated using the Ayer equation), FVC (calculated using the Knudson equation), expiratory reserve volume, inspiratory VC, and slow VC. PaO2, PaCO2, and Alveolar-arterial gradients also were collected.6-9 Information about heart failure status was gathered through medical evaluation of notes and cardiac studies. All categorical variables were correlated with Spearman analysis and quantitative variables with average percentages. P values were calculated with analysis of variance.
Results
Of 6461 VACHS patient records reviewed, 415 met the inclusion criteria. Patients were divided according to their mMRC score: 65 had mMRC score of 0, 87 had an mMRC score of 1, 2 had an mMRC score of 2, 146 had an mMRC of 3, and 115 had an mMRC score of 4. The population was primarily male (98.6%) and of Hispanic ethnicity (96.4%), with a mean age of 72 years (Table 1). Most patients (n = 269, 64.0%) were prior smokers, while 135 patients (32.5%) had never smoked, and 11 (2.7%) were current smokers. At baseline, 169 patients (41.4%) had interstitial lung disease, 39 (9.6%) had chest wall disorders, 29 (7.1%) had occupational exposure, 25 (6.1%) had pneumonitis, and 14 (3.4%) had neuromuscular disorders.
There was a statistically significant relationship between mMRC score and hospitalization and FEV1 but not TLC (Table 2). As mMRC increased, so did hospitalizations: a total of 168 patients (40.5%) were hospitalized; 24 patients (36.9%) had an mMRC score of 0, 30 patients (34.0%) had an mMRC score of 1, 2 patients (100%) had an mMRC score of 2, 54 patients (37.0%) had an mMRC score of 3, and 58 patients (50.0%) had an mMRC score of 4 (P = .04). Mean (SD) TLC values increased as mMRC scores increased. Mean (SD) TLC was 70.5% (33.0) for the entire population; 68.8% (7.2) for patients with an mMRC score of 0, 70.8% (5.8) for patients with an mMRC score of 1, 75.0% (1.4) for patients with an mMRC score of 2, 70.1% (7.2) for patients with an mMRC score of 3, and 71.5% (62.1) for patients with an mMRC score of 4 (P = .10) (Figure 1). There was an associated decrease in mean (SD) FEV1 with mMRC. Mean (SD) FEV1 was 76.2% (18.9) for the entire population; 81.7% (19.3) for patients with an mMRC score of 0, 80.9% (18) for patients with an mMRC score of 1, 93.5% (34.6) for patients with an mMRC score of 2, 76.2% (17.1) for patients with an mMRC score of 3, and 69.2% (19.4) for patients with an mMRC score of 4; (P < .001) (Figure 2).
The correlation between mMRC and FEV1 (r = 0.25, P < .001) was stronger than the correlation between mMRC and TLC (r = 0.15, P < .001). The correlations for DLCO (P < .001), DLCO/VA (P < .001), hemoglobin (P < .02), and PaO2 (P < .001) were all statistically significant (P < .005), but with no strong identifiable trend.
Discussion
The patient population of this study was primarily older males of Hispanic ethnicity with a history of smoking. There was no association between body mass index or smoking status with worsening dyspnea as measured with mMRC scores. We observed no significant correlation between mMRC scores and various factors such as comorbidities including heart conditions, and epidemiological factors like the etiology of lung disease, including both intrinsic and extrinsic causes. This lack of association was anticipated, as restrictive lung diseases in our study predominantly arose from intrinsic pulmonary etiologies, such as interstitial lung disease. A difference between more hospitalizations and worsening dyspnea was identified. There was a slightly higher correlation between FEV1 and mMRC scores when compared with TLC and mMRC scores concerning worsening dyspnea, which could indicate that the use of FEV1 should be preferred over previous recommendations to use TLC.10 Other guidelines have utilized exercise capacity via the 6-minute walk test as a marker of severity with spirometry values and found that DLCO was correlated with severity.11
The latest ERS/ATS guidelines recommend z scores for grading the severity of obstructive lung diseases but do not recommend them for the diagnosis of restrictive lung diseases.12 A z score encompasses diverse variables (eg, age, sex, and ethnicity) to provide more uniform and consistent results. Other studies have been done to relate z scores to other spirometry variables with restrictive lung disease. One such study indicates the potential benefit of using FVC alone to grade restrictive lung diseases.13 There continues to be great diversity in the interpretation of pulmonary function tests, and we believe the information gathered can provide valuable insight for managing patients with restrictive lung diseases.
Limitations
Only 2 patients reported an mMRC score of 2 in our study. This may have affected statistical outcomes. It also may reveal possible deficits in the efficacy of patient education on the mMRC scale. This study was also limited by its small sample size, single center location, and the distribution of patients that reported an mMRC favored either low or high values. The patients in this study, who were all veterans, may not be representative of other patient populations.
Conclusions
There continue to be few factors associated with the physiological severity of the defective oxygen delivery and reported dyspnea of a patient with restrictive lung disease that allows for an accurate, repeatable grading of severity. Using FEV1 instead of TLC to determine the severity of a restrictive lung disease should be reconsidered. We could not find any other strong correlation among other factors studied. Further research should be conducted to continue looking for variables that more accurately depict patient dyspnea in restrictive lung disease.
Acknowledgments
This study is based upon work supported by the Veterans Affairs Caribbean Healthcare System in San Juan, Puerto Rico, and is the result of work supported by Pulmonary & Critical Care Medicine service, with resources and the use of its facilities.
1. Hegewald MJ, Crapo RO. Pulmonary function testing. In: Broaddus VC, Ernst JD, King Jr TE, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 5th ed. Saunders; 2010:522-553.
2. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J. 2005;26(5):948-968. doi:10.1183/09031936.05.00035205
3. Rabe KF, Beghé B, Luppi F, Fabbri LM. Update in chronic obstructive pulmonary disease 2006. Am J Respir Crit Care Med. 2007;175(12):1222-1232. doi:10.1164/rccm.200704-586UP
4. Global Initiative for Chronic Obstructive Lung Disease (GOLD). Spirometry for health care providers Accessed April 30, 2024. https://goldcopd.org/wp-content/uploads/2016/04/GOLD_Spirometry_2010.pdf
5. Mannino DM, Holguin F, Pavlin BI, Ferdinands JM. Risk factors for prevalence of and mortality related to restriction on spirometry: findings from the First National Health and Nutrition Examination Survey and follow-up. Int J Tuberc Lung Dis. 2005;9(6):613-621.
6. Knudson RJ, Lebowitz MD, Holberg CJ, Burrows B. Changes in the normal maximal expiratory flow-volume curve with growth and aging. Am Rev Respir Dis. 1983;127(6):725-734. doi:10.1164/arrd.1983.127.6.725
7. Knudson RJ, Burrows B, Lebowitz MD. The maximal expiratory flow-volume curve: its use in the detection of ventilatory abnormalities in a population study. Am Rev Respir Dis. 1976;114(5):871-879. doi:10.1164/arrd.1976.114.5.871
8. Knudson RJ, Lebowitz MD, Burton AP, Knudson DE. The closing volume test: evaluation of nitrogen and bolus methods in a random population. Am Rev Respir Dis. 1977;115(3):423-434. doi:10.1164/arrd.1977.115.3.423
9. Ayers LN, Ginsberg ML, Fein J, Wasserman K. Diffusing capacity, specific diffusing capacity and interpretation of diffusion defects. West J Med. 1975;123(4):255-264.
10. Lung function testing: selection of reference values and interpretative strategies. American Thoracic Society. Am Rev Respir Dis. 1991;144(5):1202-1218. doi:10.1164/ajrccm/144.5.1202
11. Larson J, Wrzos K, Corazalla E, Wang Q, Kim HJ, Cho RJ. Should FEV1 be used to grade restrictive impairment? A single-center comparison of lung function parameters to 6-minute walk test in patients with restrictive lung disease. HSOA J Pulm Med Respir Res. 2023;9:082. doi:10.24966/PMRR-0177/100082
12. Stanojevic S, Kaminsky DA, Miller MR, et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J. 2022;60(1):2101499. Published 2022 Jul 13. doi:10.1183/13993003.01499-2021
13. Myrberg T, Lindberg A, Eriksson B, et al. Restrictive spirometry versus restrictive lung function using the GLI reference values. Clin Physiol Funct Imaging. 2022;42(3):181-189. doi:10.1111/cpf.12745
Respiratory diseases have varied clinical presentations and are classified as restrictive, obstructive, mixed, or normal. Restrictive lung diseases have reduced lung volumes, either due to an alteration in lung parenchyma or a disease of the pleura, chest wall, or neuromuscular apparatus. If caused by parenchymal lung disease, restrictive lung disorders are accompanied by reduced gas transfer, which may be portrayed clinically by desaturation after exercise. Based on anatomical structures, the causes of lung volume reduction may be intrinsic or extrinsic. Intrinsic causes correspond to diseases of the lung parenchyma, such as idiopathic fibrotic diseases, connective-tissue diseases, drug-induced lung diseases, and other primary diseases of the lungs. Extrinsic causes refer to disorders outside the lungs or extra-pulmonary diseases such as neuromuscular and nonmuscular diseases of the chest wall.1 For example, obesity and myasthenia gravis can cause restrictive lung diseases, one through mechanical interference of lung expansion and the other through neuromuscular impedance of thoracic cage expansion. All these diseases eventually result in lung restriction, impaired lung function, and respiratory failure. This heterogenicity of disease makes establishing a single severity criterion difficult.
Laboratory testing, imaging studies, and examinations are important for determining the pulmonary disease and its course and progression. The pulmonary function test (PFT), which consists of multiple procedures that are performed depending on the information needed, has been an essential tool in practice for the pulmonologist. The PFT includes spirometry, lung volume measurement, respiratory muscle strength, diffusion capacity, and a broncho-provocation test. Each test has a particular role in assisting the diagnosis and/or follow-up of the patient. Spirometry is frequently used due to its range of dynamic physiological parameters, ease of use, and accessibility. It is used for the diagnosis of pulmonary symptoms, in the assessment of disability, and preoperatory evaluation, including lung resection surgery, assisting in the diagnosis, monitoring, and therapy response of pulmonary diseases.
A systematic approach to PFT interpretation is recommended by several societies, such as the American Thoracic Society (ATS) and the European Respiratory Society (ERS).2 The pulmonary function test results must be reproducible and meet established standards to ensure reliable and consistent clinical outcomes. A restrictive respiratory disease is defined by a decrease in total lung capacity (TLC) (< 5% of predicted value) and a normal forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio.2 Although other findings—such as a decrease in vital capacity—should prompt an investigation into whether the patient has a possible restrictive respiratory disease, the sole presence of this parameter is not definitive or diagnostic of a restrictive impairment.2-4 The assessment of severity is typically determined by TLC. Unfortunately, the severity of a restrictive respiratory disease and the degree of patient discomfort do not always correlate when utilizing just TLC. Pulmonary sarcoidosis, for example, is a granulomatous lung disease with a restrictive PFT pattern and a disease burden that may vary over time. Having a more consistent method of grading the severity of the restrictive lung disease may help guide treatment. The modified Medical Research Council (mMRC) scale, a 5-point dyspnea scale, is widely used in assessing the severity of dyspnea in various respiratory conditions, including chronic obstructive pulmonary disease (COPD), where its scores have been associated with patient mortality.1,5 The goal of this study was to document the associations between objective parameters obtained through PFT and other variables, with an established measurement of dyspnea to assess the severity grade of restrictive lung diseases.
Methods
This retrospective record review at the Veterans Affairs Caribbean Healthcare System (VACHS) in San Juan, Puerto Rico, wasconducted using the Veterans Health Information Systems and Technology Architecture to identify patients with a PFT, including spirometry, that indicated a restrictive ventilator pattern based on the current ATS/ERS Task Force on Lung Function Testing.2 Patients were included if they were aged ≥ 21 years, PFT with TLC ≤ 80% predicted, mMRC score documented on PFT, and documented diffusing capacity of the lung for carbon monoxide (DLCO). Patients were excluded if their FEV1/vital capacity (VC) was < 70% predicted using the largest VC, or no mMRC score was available. All patients meeting the inclusion criteria were considered regardless of comorbidities.
The PFT results of all adult patients, including those performed between June 1, 2013, and January 6, 2016, were submitted to spirometry, and lung volume measurements were analyzed. Sociodemographic information was collected, including sex, ethnicity, age, height, weight, and basal metabolic index. Other data found in PFTs, such as smoking status, smoking in packs/year, mMRC score, predicted TLC value, imaging present (chest X-ray, computed tomography), and hospitalizations and exacerbations within 1 year were collected. In addition, we examined the predicted values for FEV1, DLCO, and DLCO/VA (calculated using the Ayer equation), FVC (calculated using the Knudson equation), expiratory reserve volume, inspiratory VC, and slow VC. PaO2, PaCO2, and Alveolar-arterial gradients also were collected.6-9 Information about heart failure status was gathered through medical evaluation of notes and cardiac studies. All categorical variables were correlated with Spearman analysis and quantitative variables with average percentages. P values were calculated with analysis of variance.
Results
Of 6461 VACHS patient records reviewed, 415 met the inclusion criteria. Patients were divided according to their mMRC score: 65 had mMRC score of 0, 87 had an mMRC score of 1, 2 had an mMRC score of 2, 146 had an mMRC of 3, and 115 had an mMRC score of 4. The population was primarily male (98.6%) and of Hispanic ethnicity (96.4%), with a mean age of 72 years (Table 1). Most patients (n = 269, 64.0%) were prior smokers, while 135 patients (32.5%) had never smoked, and 11 (2.7%) were current smokers. At baseline, 169 patients (41.4%) had interstitial lung disease, 39 (9.6%) had chest wall disorders, 29 (7.1%) had occupational exposure, 25 (6.1%) had pneumonitis, and 14 (3.4%) had neuromuscular disorders.
There was a statistically significant relationship between mMRC score and hospitalization and FEV1 but not TLC (Table 2). As mMRC increased, so did hospitalizations: a total of 168 patients (40.5%) were hospitalized; 24 patients (36.9%) had an mMRC score of 0, 30 patients (34.0%) had an mMRC score of 1, 2 patients (100%) had an mMRC score of 2, 54 patients (37.0%) had an mMRC score of 3, and 58 patients (50.0%) had an mMRC score of 4 (P = .04). Mean (SD) TLC values increased as mMRC scores increased. Mean (SD) TLC was 70.5% (33.0) for the entire population; 68.8% (7.2) for patients with an mMRC score of 0, 70.8% (5.8) for patients with an mMRC score of 1, 75.0% (1.4) for patients with an mMRC score of 2, 70.1% (7.2) for patients with an mMRC score of 3, and 71.5% (62.1) for patients with an mMRC score of 4 (P = .10) (Figure 1). There was an associated decrease in mean (SD) FEV1 with mMRC. Mean (SD) FEV1 was 76.2% (18.9) for the entire population; 81.7% (19.3) for patients with an mMRC score of 0, 80.9% (18) for patients with an mMRC score of 1, 93.5% (34.6) for patients with an mMRC score of 2, 76.2% (17.1) for patients with an mMRC score of 3, and 69.2% (19.4) for patients with an mMRC score of 4; (P < .001) (Figure 2).
The correlation between mMRC and FEV1 (r = 0.25, P < .001) was stronger than the correlation between mMRC and TLC (r = 0.15, P < .001). The correlations for DLCO (P < .001), DLCO/VA (P < .001), hemoglobin (P < .02), and PaO2 (P < .001) were all statistically significant (P < .005), but with no strong identifiable trend.
Discussion
The patient population of this study was primarily older males of Hispanic ethnicity with a history of smoking. There was no association between body mass index or smoking status with worsening dyspnea as measured with mMRC scores. We observed no significant correlation between mMRC scores and various factors such as comorbidities including heart conditions, and epidemiological factors like the etiology of lung disease, including both intrinsic and extrinsic causes. This lack of association was anticipated, as restrictive lung diseases in our study predominantly arose from intrinsic pulmonary etiologies, such as interstitial lung disease. A difference between more hospitalizations and worsening dyspnea was identified. There was a slightly higher correlation between FEV1 and mMRC scores when compared with TLC and mMRC scores concerning worsening dyspnea, which could indicate that the use of FEV1 should be preferred over previous recommendations to use TLC.10 Other guidelines have utilized exercise capacity via the 6-minute walk test as a marker of severity with spirometry values and found that DLCO was correlated with severity.11
The latest ERS/ATS guidelines recommend z scores for grading the severity of obstructive lung diseases but do not recommend them for the diagnosis of restrictive lung diseases.12 A z score encompasses diverse variables (eg, age, sex, and ethnicity) to provide more uniform and consistent results. Other studies have been done to relate z scores to other spirometry variables with restrictive lung disease. One such study indicates the potential benefit of using FVC alone to grade restrictive lung diseases.13 There continues to be great diversity in the interpretation of pulmonary function tests, and we believe the information gathered can provide valuable insight for managing patients with restrictive lung diseases.
Limitations
Only 2 patients reported an mMRC score of 2 in our study. This may have affected statistical outcomes. It also may reveal possible deficits in the efficacy of patient education on the mMRC scale. This study was also limited by its small sample size, single center location, and the distribution of patients that reported an mMRC favored either low or high values. The patients in this study, who were all veterans, may not be representative of other patient populations.
Conclusions
There continue to be few factors associated with the physiological severity of the defective oxygen delivery and reported dyspnea of a patient with restrictive lung disease that allows for an accurate, repeatable grading of severity. Using FEV1 instead of TLC to determine the severity of a restrictive lung disease should be reconsidered. We could not find any other strong correlation among other factors studied. Further research should be conducted to continue looking for variables that more accurately depict patient dyspnea in restrictive lung disease.
Acknowledgments
This study is based upon work supported by the Veterans Affairs Caribbean Healthcare System in San Juan, Puerto Rico, and is the result of work supported by Pulmonary & Critical Care Medicine service, with resources and the use of its facilities.
Respiratory diseases have varied clinical presentations and are classified as restrictive, obstructive, mixed, or normal. Restrictive lung diseases have reduced lung volumes, either due to an alteration in lung parenchyma or a disease of the pleura, chest wall, or neuromuscular apparatus. If caused by parenchymal lung disease, restrictive lung disorders are accompanied by reduced gas transfer, which may be portrayed clinically by desaturation after exercise. Based on anatomical structures, the causes of lung volume reduction may be intrinsic or extrinsic. Intrinsic causes correspond to diseases of the lung parenchyma, such as idiopathic fibrotic diseases, connective-tissue diseases, drug-induced lung diseases, and other primary diseases of the lungs. Extrinsic causes refer to disorders outside the lungs or extra-pulmonary diseases such as neuromuscular and nonmuscular diseases of the chest wall.1 For example, obesity and myasthenia gravis can cause restrictive lung diseases, one through mechanical interference of lung expansion and the other through neuromuscular impedance of thoracic cage expansion. All these diseases eventually result in lung restriction, impaired lung function, and respiratory failure. This heterogenicity of disease makes establishing a single severity criterion difficult.
Laboratory testing, imaging studies, and examinations are important for determining the pulmonary disease and its course and progression. The pulmonary function test (PFT), which consists of multiple procedures that are performed depending on the information needed, has been an essential tool in practice for the pulmonologist. The PFT includes spirometry, lung volume measurement, respiratory muscle strength, diffusion capacity, and a broncho-provocation test. Each test has a particular role in assisting the diagnosis and/or follow-up of the patient. Spirometry is frequently used due to its range of dynamic physiological parameters, ease of use, and accessibility. It is used for the diagnosis of pulmonary symptoms, in the assessment of disability, and preoperatory evaluation, including lung resection surgery, assisting in the diagnosis, monitoring, and therapy response of pulmonary diseases.
A systematic approach to PFT interpretation is recommended by several societies, such as the American Thoracic Society (ATS) and the European Respiratory Society (ERS).2 The pulmonary function test results must be reproducible and meet established standards to ensure reliable and consistent clinical outcomes. A restrictive respiratory disease is defined by a decrease in total lung capacity (TLC) (< 5% of predicted value) and a normal forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio.2 Although other findings—such as a decrease in vital capacity—should prompt an investigation into whether the patient has a possible restrictive respiratory disease, the sole presence of this parameter is not definitive or diagnostic of a restrictive impairment.2-4 The assessment of severity is typically determined by TLC. Unfortunately, the severity of a restrictive respiratory disease and the degree of patient discomfort do not always correlate when utilizing just TLC. Pulmonary sarcoidosis, for example, is a granulomatous lung disease with a restrictive PFT pattern and a disease burden that may vary over time. Having a more consistent method of grading the severity of the restrictive lung disease may help guide treatment. The modified Medical Research Council (mMRC) scale, a 5-point dyspnea scale, is widely used in assessing the severity of dyspnea in various respiratory conditions, including chronic obstructive pulmonary disease (COPD), where its scores have been associated with patient mortality.1,5 The goal of this study was to document the associations between objective parameters obtained through PFT and other variables, with an established measurement of dyspnea to assess the severity grade of restrictive lung diseases.
Methods
This retrospective record review at the Veterans Affairs Caribbean Healthcare System (VACHS) in San Juan, Puerto Rico, wasconducted using the Veterans Health Information Systems and Technology Architecture to identify patients with a PFT, including spirometry, that indicated a restrictive ventilator pattern based on the current ATS/ERS Task Force on Lung Function Testing.2 Patients were included if they were aged ≥ 21 years, PFT with TLC ≤ 80% predicted, mMRC score documented on PFT, and documented diffusing capacity of the lung for carbon monoxide (DLCO). Patients were excluded if their FEV1/vital capacity (VC) was < 70% predicted using the largest VC, or no mMRC score was available. All patients meeting the inclusion criteria were considered regardless of comorbidities.
The PFT results of all adult patients, including those performed between June 1, 2013, and January 6, 2016, were submitted to spirometry, and lung volume measurements were analyzed. Sociodemographic information was collected, including sex, ethnicity, age, height, weight, and basal metabolic index. Other data found in PFTs, such as smoking status, smoking in packs/year, mMRC score, predicted TLC value, imaging present (chest X-ray, computed tomography), and hospitalizations and exacerbations within 1 year were collected. In addition, we examined the predicted values for FEV1, DLCO, and DLCO/VA (calculated using the Ayer equation), FVC (calculated using the Knudson equation), expiratory reserve volume, inspiratory VC, and slow VC. PaO2, PaCO2, and Alveolar-arterial gradients also were collected.6-9 Information about heart failure status was gathered through medical evaluation of notes and cardiac studies. All categorical variables were correlated with Spearman analysis and quantitative variables with average percentages. P values were calculated with analysis of variance.
Results
Of 6461 VACHS patient records reviewed, 415 met the inclusion criteria. Patients were divided according to their mMRC score: 65 had mMRC score of 0, 87 had an mMRC score of 1, 2 had an mMRC score of 2, 146 had an mMRC of 3, and 115 had an mMRC score of 4. The population was primarily male (98.6%) and of Hispanic ethnicity (96.4%), with a mean age of 72 years (Table 1). Most patients (n = 269, 64.0%) were prior smokers, while 135 patients (32.5%) had never smoked, and 11 (2.7%) were current smokers. At baseline, 169 patients (41.4%) had interstitial lung disease, 39 (9.6%) had chest wall disorders, 29 (7.1%) had occupational exposure, 25 (6.1%) had pneumonitis, and 14 (3.4%) had neuromuscular disorders.
There was a statistically significant relationship between mMRC score and hospitalization and FEV1 but not TLC (Table 2). As mMRC increased, so did hospitalizations: a total of 168 patients (40.5%) were hospitalized; 24 patients (36.9%) had an mMRC score of 0, 30 patients (34.0%) had an mMRC score of 1, 2 patients (100%) had an mMRC score of 2, 54 patients (37.0%) had an mMRC score of 3, and 58 patients (50.0%) had an mMRC score of 4 (P = .04). Mean (SD) TLC values increased as mMRC scores increased. Mean (SD) TLC was 70.5% (33.0) for the entire population; 68.8% (7.2) for patients with an mMRC score of 0, 70.8% (5.8) for patients with an mMRC score of 1, 75.0% (1.4) for patients with an mMRC score of 2, 70.1% (7.2) for patients with an mMRC score of 3, and 71.5% (62.1) for patients with an mMRC score of 4 (P = .10) (Figure 1). There was an associated decrease in mean (SD) FEV1 with mMRC. Mean (SD) FEV1 was 76.2% (18.9) for the entire population; 81.7% (19.3) for patients with an mMRC score of 0, 80.9% (18) for patients with an mMRC score of 1, 93.5% (34.6) for patients with an mMRC score of 2, 76.2% (17.1) for patients with an mMRC score of 3, and 69.2% (19.4) for patients with an mMRC score of 4; (P < .001) (Figure 2).
The correlation between mMRC and FEV1 (r = 0.25, P < .001) was stronger than the correlation between mMRC and TLC (r = 0.15, P < .001). The correlations for DLCO (P < .001), DLCO/VA (P < .001), hemoglobin (P < .02), and PaO2 (P < .001) were all statistically significant (P < .005), but with no strong identifiable trend.
Discussion
The patient population of this study was primarily older males of Hispanic ethnicity with a history of smoking. There was no association between body mass index or smoking status with worsening dyspnea as measured with mMRC scores. We observed no significant correlation between mMRC scores and various factors such as comorbidities including heart conditions, and epidemiological factors like the etiology of lung disease, including both intrinsic and extrinsic causes. This lack of association was anticipated, as restrictive lung diseases in our study predominantly arose from intrinsic pulmonary etiologies, such as interstitial lung disease. A difference between more hospitalizations and worsening dyspnea was identified. There was a slightly higher correlation between FEV1 and mMRC scores when compared with TLC and mMRC scores concerning worsening dyspnea, which could indicate that the use of FEV1 should be preferred over previous recommendations to use TLC.10 Other guidelines have utilized exercise capacity via the 6-minute walk test as a marker of severity with spirometry values and found that DLCO was correlated with severity.11
The latest ERS/ATS guidelines recommend z scores for grading the severity of obstructive lung diseases but do not recommend them for the diagnosis of restrictive lung diseases.12 A z score encompasses diverse variables (eg, age, sex, and ethnicity) to provide more uniform and consistent results. Other studies have been done to relate z scores to other spirometry variables with restrictive lung disease. One such study indicates the potential benefit of using FVC alone to grade restrictive lung diseases.13 There continues to be great diversity in the interpretation of pulmonary function tests, and we believe the information gathered can provide valuable insight for managing patients with restrictive lung diseases.
Limitations
Only 2 patients reported an mMRC score of 2 in our study. This may have affected statistical outcomes. It also may reveal possible deficits in the efficacy of patient education on the mMRC scale. This study was also limited by its small sample size, single center location, and the distribution of patients that reported an mMRC favored either low or high values. The patients in this study, who were all veterans, may not be representative of other patient populations.
Conclusions
There continue to be few factors associated with the physiological severity of the defective oxygen delivery and reported dyspnea of a patient with restrictive lung disease that allows for an accurate, repeatable grading of severity. Using FEV1 instead of TLC to determine the severity of a restrictive lung disease should be reconsidered. We could not find any other strong correlation among other factors studied. Further research should be conducted to continue looking for variables that more accurately depict patient dyspnea in restrictive lung disease.
Acknowledgments
This study is based upon work supported by the Veterans Affairs Caribbean Healthcare System in San Juan, Puerto Rico, and is the result of work supported by Pulmonary & Critical Care Medicine service, with resources and the use of its facilities.
1. Hegewald MJ, Crapo RO. Pulmonary function testing. In: Broaddus VC, Ernst JD, King Jr TE, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 5th ed. Saunders; 2010:522-553.
2. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J. 2005;26(5):948-968. doi:10.1183/09031936.05.00035205
3. Rabe KF, Beghé B, Luppi F, Fabbri LM. Update in chronic obstructive pulmonary disease 2006. Am J Respir Crit Care Med. 2007;175(12):1222-1232. doi:10.1164/rccm.200704-586UP
4. Global Initiative for Chronic Obstructive Lung Disease (GOLD). Spirometry for health care providers Accessed April 30, 2024. https://goldcopd.org/wp-content/uploads/2016/04/GOLD_Spirometry_2010.pdf
5. Mannino DM, Holguin F, Pavlin BI, Ferdinands JM. Risk factors for prevalence of and mortality related to restriction on spirometry: findings from the First National Health and Nutrition Examination Survey and follow-up. Int J Tuberc Lung Dis. 2005;9(6):613-621.
6. Knudson RJ, Lebowitz MD, Holberg CJ, Burrows B. Changes in the normal maximal expiratory flow-volume curve with growth and aging. Am Rev Respir Dis. 1983;127(6):725-734. doi:10.1164/arrd.1983.127.6.725
7. Knudson RJ, Burrows B, Lebowitz MD. The maximal expiratory flow-volume curve: its use in the detection of ventilatory abnormalities in a population study. Am Rev Respir Dis. 1976;114(5):871-879. doi:10.1164/arrd.1976.114.5.871
8. Knudson RJ, Lebowitz MD, Burton AP, Knudson DE. The closing volume test: evaluation of nitrogen and bolus methods in a random population. Am Rev Respir Dis. 1977;115(3):423-434. doi:10.1164/arrd.1977.115.3.423
9. Ayers LN, Ginsberg ML, Fein J, Wasserman K. Diffusing capacity, specific diffusing capacity and interpretation of diffusion defects. West J Med. 1975;123(4):255-264.
10. Lung function testing: selection of reference values and interpretative strategies. American Thoracic Society. Am Rev Respir Dis. 1991;144(5):1202-1218. doi:10.1164/ajrccm/144.5.1202
11. Larson J, Wrzos K, Corazalla E, Wang Q, Kim HJ, Cho RJ. Should FEV1 be used to grade restrictive impairment? A single-center comparison of lung function parameters to 6-minute walk test in patients with restrictive lung disease. HSOA J Pulm Med Respir Res. 2023;9:082. doi:10.24966/PMRR-0177/100082
12. Stanojevic S, Kaminsky DA, Miller MR, et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J. 2022;60(1):2101499. Published 2022 Jul 13. doi:10.1183/13993003.01499-2021
13. Myrberg T, Lindberg A, Eriksson B, et al. Restrictive spirometry versus restrictive lung function using the GLI reference values. Clin Physiol Funct Imaging. 2022;42(3):181-189. doi:10.1111/cpf.12745
1. Hegewald MJ, Crapo RO. Pulmonary function testing. In: Broaddus VC, Ernst JD, King Jr TE, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 5th ed. Saunders; 2010:522-553.
2. Pellegrino R, Viegi G, Brusasco V, et al. Interpretative strategies for lung function tests. Eur Respir J. 2005;26(5):948-968. doi:10.1183/09031936.05.00035205
3. Rabe KF, Beghé B, Luppi F, Fabbri LM. Update in chronic obstructive pulmonary disease 2006. Am J Respir Crit Care Med. 2007;175(12):1222-1232. doi:10.1164/rccm.200704-586UP
4. Global Initiative for Chronic Obstructive Lung Disease (GOLD). Spirometry for health care providers Accessed April 30, 2024. https://goldcopd.org/wp-content/uploads/2016/04/GOLD_Spirometry_2010.pdf
5. Mannino DM, Holguin F, Pavlin BI, Ferdinands JM. Risk factors for prevalence of and mortality related to restriction on spirometry: findings from the First National Health and Nutrition Examination Survey and follow-up. Int J Tuberc Lung Dis. 2005;9(6):613-621.
6. Knudson RJ, Lebowitz MD, Holberg CJ, Burrows B. Changes in the normal maximal expiratory flow-volume curve with growth and aging. Am Rev Respir Dis. 1983;127(6):725-734. doi:10.1164/arrd.1983.127.6.725
7. Knudson RJ, Burrows B, Lebowitz MD. The maximal expiratory flow-volume curve: its use in the detection of ventilatory abnormalities in a population study. Am Rev Respir Dis. 1976;114(5):871-879. doi:10.1164/arrd.1976.114.5.871
8. Knudson RJ, Lebowitz MD, Burton AP, Knudson DE. The closing volume test: evaluation of nitrogen and bolus methods in a random population. Am Rev Respir Dis. 1977;115(3):423-434. doi:10.1164/arrd.1977.115.3.423
9. Ayers LN, Ginsberg ML, Fein J, Wasserman K. Diffusing capacity, specific diffusing capacity and interpretation of diffusion defects. West J Med. 1975;123(4):255-264.
10. Lung function testing: selection of reference values and interpretative strategies. American Thoracic Society. Am Rev Respir Dis. 1991;144(5):1202-1218. doi:10.1164/ajrccm/144.5.1202
11. Larson J, Wrzos K, Corazalla E, Wang Q, Kim HJ, Cho RJ. Should FEV1 be used to grade restrictive impairment? A single-center comparison of lung function parameters to 6-minute walk test in patients with restrictive lung disease. HSOA J Pulm Med Respir Res. 2023;9:082. doi:10.24966/PMRR-0177/100082
12. Stanojevic S, Kaminsky DA, Miller MR, et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J. 2022;60(1):2101499. Published 2022 Jul 13. doi:10.1183/13993003.01499-2021
13. Myrberg T, Lindberg A, Eriksson B, et al. Restrictive spirometry versus restrictive lung function using the GLI reference values. Clin Physiol Funct Imaging. 2022;42(3):181-189. doi:10.1111/cpf.12745
Quitting Smoking Boosts Life Expectancy at Any Age
TOPLINE:
Quitting smoking at any age increases life expectancy, with the most significant increases observed in younger individuals. But people who quit over age 65 can extend life expectancy.
METHODOLOGY:
- Researchers analyzed the detrimental effects of smoking and the positive impacts of cessation on life expectancy in individuals aged 35-75 years.
- Age-specific death rates by smoking status were calculated using the relative risks for all-cause mortality derived from the Cancer Prevention Study II data, 2018 National Health Interview Survey smoking prevalence data, and 2018 all-cause mortality rates.
- Life tables were constructed to obtain information on the life expectancies of people who never smoked, those who currently smoked, and those who previously smoked but quit at various ages.
- Estimates of years lost due to smoking and years gained by quitting smoking were calculated for people starting at age 35 and over 10-year increments.
TAKEAWAY:
- Compared with people who never smoked, those who smoked at ages 35, 45, 55, 65, and 75 years and continued smoking throughout their lives would lose 9.1, 8.3, 7.3, 5.9, and 4.4 years, respectively.
- People who quit smoking at ages 35, 45, 55, 65, and 75 years would have life expectancies that are shorter by 1.2, 2.7, 3.9, 4.2, and 3.7 years, respectively, than those of same-age individuals who never smoked.
- Individuals who quit smoking at ages 35, 45, 55, 65, and 75 years would experience an additional 8.0, 5.6, 3.4, 1.7, and 0.7 years of life expectancy compared with those who continued smoking.
- People who quit at ages 65 and 75 years would have a 23.4% and 14.2% chance of gaining at least 1 additional year of life.
IN PRACTICE:
“This cessation benefit is not limited to young- and middle-aged adults who smoke; this study demonstrates its applicability to seniors as well. These findings may be valuable for clinicians seeking scientific evidence to motivate their patients who smoke to quit,” the authors wrote.
SOURCE:
The study was led by Thuy T.T. Le, PhD, from the Department of Health Management and Policy at the University of Michigan School of Public Health in Ann Arbor and published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s estimates were according to data from 2018 and may not reflect current trends. The estimates also did not account for variability in smoking intensity among individuals.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute of the US National Institutes of Health and the US Food and Drug Administration Center for Tobacco Products. The authors declared that they had no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Quitting smoking at any age increases life expectancy, with the most significant increases observed in younger individuals. But people who quit over age 65 can extend life expectancy.
METHODOLOGY:
- Researchers analyzed the detrimental effects of smoking and the positive impacts of cessation on life expectancy in individuals aged 35-75 years.
- Age-specific death rates by smoking status were calculated using the relative risks for all-cause mortality derived from the Cancer Prevention Study II data, 2018 National Health Interview Survey smoking prevalence data, and 2018 all-cause mortality rates.
- Life tables were constructed to obtain information on the life expectancies of people who never smoked, those who currently smoked, and those who previously smoked but quit at various ages.
- Estimates of years lost due to smoking and years gained by quitting smoking were calculated for people starting at age 35 and over 10-year increments.
TAKEAWAY:
- Compared with people who never smoked, those who smoked at ages 35, 45, 55, 65, and 75 years and continued smoking throughout their lives would lose 9.1, 8.3, 7.3, 5.9, and 4.4 years, respectively.
- People who quit smoking at ages 35, 45, 55, 65, and 75 years would have life expectancies that are shorter by 1.2, 2.7, 3.9, 4.2, and 3.7 years, respectively, than those of same-age individuals who never smoked.
- Individuals who quit smoking at ages 35, 45, 55, 65, and 75 years would experience an additional 8.0, 5.6, 3.4, 1.7, and 0.7 years of life expectancy compared with those who continued smoking.
- People who quit at ages 65 and 75 years would have a 23.4% and 14.2% chance of gaining at least 1 additional year of life.
IN PRACTICE:
“This cessation benefit is not limited to young- and middle-aged adults who smoke; this study demonstrates its applicability to seniors as well. These findings may be valuable for clinicians seeking scientific evidence to motivate their patients who smoke to quit,” the authors wrote.
SOURCE:
The study was led by Thuy T.T. Le, PhD, from the Department of Health Management and Policy at the University of Michigan School of Public Health in Ann Arbor and published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s estimates were according to data from 2018 and may not reflect current trends. The estimates also did not account for variability in smoking intensity among individuals.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute of the US National Institutes of Health and the US Food and Drug Administration Center for Tobacco Products. The authors declared that they had no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Quitting smoking at any age increases life expectancy, with the most significant increases observed in younger individuals. But people who quit over age 65 can extend life expectancy.
METHODOLOGY:
- Researchers analyzed the detrimental effects of smoking and the positive impacts of cessation on life expectancy in individuals aged 35-75 years.
- Age-specific death rates by smoking status were calculated using the relative risks for all-cause mortality derived from the Cancer Prevention Study II data, 2018 National Health Interview Survey smoking prevalence data, and 2018 all-cause mortality rates.
- Life tables were constructed to obtain information on the life expectancies of people who never smoked, those who currently smoked, and those who previously smoked but quit at various ages.
- Estimates of years lost due to smoking and years gained by quitting smoking were calculated for people starting at age 35 and over 10-year increments.
TAKEAWAY:
- Compared with people who never smoked, those who smoked at ages 35, 45, 55, 65, and 75 years and continued smoking throughout their lives would lose 9.1, 8.3, 7.3, 5.9, and 4.4 years, respectively.
- People who quit smoking at ages 35, 45, 55, 65, and 75 years would have life expectancies that are shorter by 1.2, 2.7, 3.9, 4.2, and 3.7 years, respectively, than those of same-age individuals who never smoked.
- Individuals who quit smoking at ages 35, 45, 55, 65, and 75 years would experience an additional 8.0, 5.6, 3.4, 1.7, and 0.7 years of life expectancy compared with those who continued smoking.
- People who quit at ages 65 and 75 years would have a 23.4% and 14.2% chance of gaining at least 1 additional year of life.
IN PRACTICE:
“This cessation benefit is not limited to young- and middle-aged adults who smoke; this study demonstrates its applicability to seniors as well. These findings may be valuable for clinicians seeking scientific evidence to motivate their patients who smoke to quit,” the authors wrote.
SOURCE:
The study was led by Thuy T.T. Le, PhD, from the Department of Health Management and Policy at the University of Michigan School of Public Health in Ann Arbor and published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study’s estimates were according to data from 2018 and may not reflect current trends. The estimates also did not account for variability in smoking intensity among individuals.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute of the US National Institutes of Health and the US Food and Drug Administration Center for Tobacco Products. The authors declared that they had no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Specific Antipsychotics Linked to Increased Pneumonia Risk
TOPLINE:
High-dose antipsychotics, particularly quetiapine, clozapine, and olanzapine, are linked to increased pneumonia risk in patients with schizophrenia, new data show. Monotherapy with high anticholinergic burden also raises pneumonia risk.
METHODOLOGY:
- Using several nationwide data registers, investigators pulled data on individuals who received inpatient care for schizophrenia or schizoaffective disorder (n = 61,889) between 1972 and 2014.
- Data on drug use were gathered from a prescription register and included dispensing dates, cost, dose, package size, and drug formulation. Data on dates and causes of death were obtained from the Causes of Death register.
- After entering the cohort, follow-up started in January 1996 or after the first diagnosis of schizophrenia for those diagnosed between 1996 and 2014.
- The primary outcome was hospitalization caused by pneumonia as the main diagnosis for hospital admission.
TAKEAWAY:
- During 22 years of follow-up, 8917 patients (14.4%) had one or more hospitalizations for pneumonia, and 1137 (12.8%) died within 30 days of admission.
- Pneumonia risk was the highest with the use of high-dose (> 440 mg/d) quetiapine (P = .003), followed by high- (≥ 330 mg/d) and medium-dose (180 to < 330 mg/d) clozapine (both P < .001) and high-dose (≥ 11 mg/d) olanzapine (P = .02).
- Compared with no antipsychotic use, antipsychotic monotherapy was associated with an increased pneumonia risk (P = .03), whereas antipsychotic polytherapy was not.
- Only the use of antipsychotics with high anticholinergic potency was associated with pneumonia risk (P < .001).
IN PRACTICE:
“Identification of antipsychotic drugs that are associated with pneumonia risk may better inform prevention programs (eg, vaccinations),” the researchers noted. “Second, the availability of pneumonia risk estimates for individual antipsychotics and for groups of antipsychotics may foster personalized prescribing guidelines.”
SOURCE:
The study was led by Jurjen Luykx, MD, Amsterdam University Medical Center, Amsterdam, the Netherlands. It was published online in JAMA Psychiatry.
LIMITATIONS:
The investigators could not correct for all possible risk factors that may increase pneumonia risk in individuals with schizophrenia, such as smoking and lifestyle habits. Also, cases of pneumonia that didn’t require hospital admission couldn’t be included in the analysis, so the findings may generalize only to cases of severe pneumonia.
DISCLOSURES:
The study was funded by the Finnish Ministry of Social Affairs and Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
High-dose antipsychotics, particularly quetiapine, clozapine, and olanzapine, are linked to increased pneumonia risk in patients with schizophrenia, new data show. Monotherapy with high anticholinergic burden also raises pneumonia risk.
METHODOLOGY:
- Using several nationwide data registers, investigators pulled data on individuals who received inpatient care for schizophrenia or schizoaffective disorder (n = 61,889) between 1972 and 2014.
- Data on drug use were gathered from a prescription register and included dispensing dates, cost, dose, package size, and drug formulation. Data on dates and causes of death were obtained from the Causes of Death register.
- After entering the cohort, follow-up started in January 1996 or after the first diagnosis of schizophrenia for those diagnosed between 1996 and 2014.
- The primary outcome was hospitalization caused by pneumonia as the main diagnosis for hospital admission.
TAKEAWAY:
- During 22 years of follow-up, 8917 patients (14.4%) had one or more hospitalizations for pneumonia, and 1137 (12.8%) died within 30 days of admission.
- Pneumonia risk was the highest with the use of high-dose (> 440 mg/d) quetiapine (P = .003), followed by high- (≥ 330 mg/d) and medium-dose (180 to < 330 mg/d) clozapine (both P < .001) and high-dose (≥ 11 mg/d) olanzapine (P = .02).
- Compared with no antipsychotic use, antipsychotic monotherapy was associated with an increased pneumonia risk (P = .03), whereas antipsychotic polytherapy was not.
- Only the use of antipsychotics with high anticholinergic potency was associated with pneumonia risk (P < .001).
IN PRACTICE:
“Identification of antipsychotic drugs that are associated with pneumonia risk may better inform prevention programs (eg, vaccinations),” the researchers noted. “Second, the availability of pneumonia risk estimates for individual antipsychotics and for groups of antipsychotics may foster personalized prescribing guidelines.”
SOURCE:
The study was led by Jurjen Luykx, MD, Amsterdam University Medical Center, Amsterdam, the Netherlands. It was published online in JAMA Psychiatry.
LIMITATIONS:
The investigators could not correct for all possible risk factors that may increase pneumonia risk in individuals with schizophrenia, such as smoking and lifestyle habits. Also, cases of pneumonia that didn’t require hospital admission couldn’t be included in the analysis, so the findings may generalize only to cases of severe pneumonia.
DISCLOSURES:
The study was funded by the Finnish Ministry of Social Affairs and Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
High-dose antipsychotics, particularly quetiapine, clozapine, and olanzapine, are linked to increased pneumonia risk in patients with schizophrenia, new data show. Monotherapy with high anticholinergic burden also raises pneumonia risk.
METHODOLOGY:
- Using several nationwide data registers, investigators pulled data on individuals who received inpatient care for schizophrenia or schizoaffective disorder (n = 61,889) between 1972 and 2014.
- Data on drug use were gathered from a prescription register and included dispensing dates, cost, dose, package size, and drug formulation. Data on dates and causes of death were obtained from the Causes of Death register.
- After entering the cohort, follow-up started in January 1996 or after the first diagnosis of schizophrenia for those diagnosed between 1996 and 2014.
- The primary outcome was hospitalization caused by pneumonia as the main diagnosis for hospital admission.
TAKEAWAY:
- During 22 years of follow-up, 8917 patients (14.4%) had one or more hospitalizations for pneumonia, and 1137 (12.8%) died within 30 days of admission.
- Pneumonia risk was the highest with the use of high-dose (> 440 mg/d) quetiapine (P = .003), followed by high- (≥ 330 mg/d) and medium-dose (180 to < 330 mg/d) clozapine (both P < .001) and high-dose (≥ 11 mg/d) olanzapine (P = .02).
- Compared with no antipsychotic use, antipsychotic monotherapy was associated with an increased pneumonia risk (P = .03), whereas antipsychotic polytherapy was not.
- Only the use of antipsychotics with high anticholinergic potency was associated with pneumonia risk (P < .001).
IN PRACTICE:
“Identification of antipsychotic drugs that are associated with pneumonia risk may better inform prevention programs (eg, vaccinations),” the researchers noted. “Second, the availability of pneumonia risk estimates for individual antipsychotics and for groups of antipsychotics may foster personalized prescribing guidelines.”
SOURCE:
The study was led by Jurjen Luykx, MD, Amsterdam University Medical Center, Amsterdam, the Netherlands. It was published online in JAMA Psychiatry.
LIMITATIONS:
The investigators could not correct for all possible risk factors that may increase pneumonia risk in individuals with schizophrenia, such as smoking and lifestyle habits. Also, cases of pneumonia that didn’t require hospital admission couldn’t be included in the analysis, so the findings may generalize only to cases of severe pneumonia.
DISCLOSURES:
The study was funded by the Finnish Ministry of Social Affairs and Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Top reads from the CHEST journal portfolio
Understanding RA with COPD, lung cancer prediction models, and chronic cardiac dysfunction
Journal CHEST®
Does Rheumatoid Arthritis Increase the Risk of COPD?
By: Chiwook Chung, MD, and colleagues
Notably, individuals with seropositive RA exhibit a greater risk of COPD onset than those with seronegative RA. Although smoking history didn’t affect the relationship between RA and COPD, monitoring respiratory symptoms and pulmonary function in patients with RA, especially patients who are seropositive, is crucial. These findings underscore the importance of interdisciplinary collaboration between rheumatologists and pulmonologists to enhance early detection and management strategies for pulmonary complications in patients with RA.
– Commentary by Corinne Young, MSN, FNP-C, FCCP, Member of the CHEST Physician® Editorial Board
CHEST Pulmonary®
The Lung Cancer Prediction Model “Stress Test”
By: Brent E. Heideman, MD, and colleagues
Current lung cancer prediction models have limited utility in high-risk patients referred for diagnostic biopsy. In a study of 322 indeterminate pulmonary nodules, the Brock, Mayo Clinic, Herder, and Veterans Affairs models showed modest discrimination between benign and malignant nodules (AUCs 0.67-0.77). The models performed poorly for low-risk patients (negative predictive values 63%-71%) and suboptimally for high-risk patients (positive predictive values 73%-87%), suggesting referring physicians use additional clinical information not captured in these models to identify high-risk patients needing biopsy. New prediction models and biomarkers specifically developed and calibrated for high-risk populations are needed to better inform clinical decision-making. Incorporating interval imaging to assess changes in nodule characteristics could potentially improve model performance. Tailored risk assessment tools are crucial for optimizing management and reducing unnecessary invasive procedures in this challenging patient population.
– Commentary by Russell Miller, MD, Member of the CHEST Physician Editorial Board
CHEST Critical Care ®
Characterizing Cardiac Function in ICU Survivors of Sepsis
By: Kevin Garrity, MBChB, and colleagues
While chronic cardiac dysfunction is one of the proposed mechanisms of long-term impairment post critical illness, its prevalence, mechanisms, and associations with disability following admission for sepsis are not well understood. Garrity and colleagues describe the Characterization of Cardiovascular Function in ICU Survivors of Sepsis (CONDUCT-ICU) protocol, a prospective study including two ICUs in Scotland aimed to better define cardiovascular dysfunction in survivors of sepsis. Designed to enroll 69 patients, demographics, cardiac and inflammatory biomarkers, and echocardiograms will be obtained on ICU discharge with additional laboratory data, cardiac magnetic resonance imaging, and patient-reported outcome measures to be obtained at 6 to 10 weeks. This novel multimodal approach will provide understanding into the role of cardiovascular dysfunction following critical illness as well as offer mechanistic insights. The investigators hope to obtain operational and pilot data for larger future studies.
– Commentary by Eugene Yuriditsky, MD, FCCP, Member of the CHEST Physician Editorial Board
Understanding RA with COPD, lung cancer prediction models, and chronic cardiac dysfunction
Understanding RA with COPD, lung cancer prediction models, and chronic cardiac dysfunction
Journal CHEST®
Does Rheumatoid Arthritis Increase the Risk of COPD?
By: Chiwook Chung, MD, and colleagues
Notably, individuals with seropositive RA exhibit a greater risk of COPD onset than those with seronegative RA. Although smoking history didn’t affect the relationship between RA and COPD, monitoring respiratory symptoms and pulmonary function in patients with RA, especially patients who are seropositive, is crucial. These findings underscore the importance of interdisciplinary collaboration between rheumatologists and pulmonologists to enhance early detection and management strategies for pulmonary complications in patients with RA.
– Commentary by Corinne Young, MSN, FNP-C, FCCP, Member of the CHEST Physician® Editorial Board
CHEST Pulmonary®
The Lung Cancer Prediction Model “Stress Test”
By: Brent E. Heideman, MD, and colleagues
Current lung cancer prediction models have limited utility in high-risk patients referred for diagnostic biopsy. In a study of 322 indeterminate pulmonary nodules, the Brock, Mayo Clinic, Herder, and Veterans Affairs models showed modest discrimination between benign and malignant nodules (AUCs 0.67-0.77). The models performed poorly for low-risk patients (negative predictive values 63%-71%) and suboptimally for high-risk patients (positive predictive values 73%-87%), suggesting referring physicians use additional clinical information not captured in these models to identify high-risk patients needing biopsy. New prediction models and biomarkers specifically developed and calibrated for high-risk populations are needed to better inform clinical decision-making. Incorporating interval imaging to assess changes in nodule characteristics could potentially improve model performance. Tailored risk assessment tools are crucial for optimizing management and reducing unnecessary invasive procedures in this challenging patient population.
– Commentary by Russell Miller, MD, Member of the CHEST Physician Editorial Board
CHEST Critical Care ®
Characterizing Cardiac Function in ICU Survivors of Sepsis
By: Kevin Garrity, MBChB, and colleagues
While chronic cardiac dysfunction is one of the proposed mechanisms of long-term impairment post critical illness, its prevalence, mechanisms, and associations with disability following admission for sepsis are not well understood. Garrity and colleagues describe the Characterization of Cardiovascular Function in ICU Survivors of Sepsis (CONDUCT-ICU) protocol, a prospective study including two ICUs in Scotland aimed to better define cardiovascular dysfunction in survivors of sepsis. Designed to enroll 69 patients, demographics, cardiac and inflammatory biomarkers, and echocardiograms will be obtained on ICU discharge with additional laboratory data, cardiac magnetic resonance imaging, and patient-reported outcome measures to be obtained at 6 to 10 weeks. This novel multimodal approach will provide understanding into the role of cardiovascular dysfunction following critical illness as well as offer mechanistic insights. The investigators hope to obtain operational and pilot data for larger future studies.
– Commentary by Eugene Yuriditsky, MD, FCCP, Member of the CHEST Physician Editorial Board
Journal CHEST®
Does Rheumatoid Arthritis Increase the Risk of COPD?
By: Chiwook Chung, MD, and colleagues
Notably, individuals with seropositive RA exhibit a greater risk of COPD onset than those with seronegative RA. Although smoking history didn’t affect the relationship between RA and COPD, monitoring respiratory symptoms and pulmonary function in patients with RA, especially patients who are seropositive, is crucial. These findings underscore the importance of interdisciplinary collaboration between rheumatologists and pulmonologists to enhance early detection and management strategies for pulmonary complications in patients with RA.
– Commentary by Corinne Young, MSN, FNP-C, FCCP, Member of the CHEST Physician® Editorial Board
CHEST Pulmonary®
The Lung Cancer Prediction Model “Stress Test”
By: Brent E. Heideman, MD, and colleagues
Current lung cancer prediction models have limited utility in high-risk patients referred for diagnostic biopsy. In a study of 322 indeterminate pulmonary nodules, the Brock, Mayo Clinic, Herder, and Veterans Affairs models showed modest discrimination between benign and malignant nodules (AUCs 0.67-0.77). The models performed poorly for low-risk patients (negative predictive values 63%-71%) and suboptimally for high-risk patients (positive predictive values 73%-87%), suggesting referring physicians use additional clinical information not captured in these models to identify high-risk patients needing biopsy. New prediction models and biomarkers specifically developed and calibrated for high-risk populations are needed to better inform clinical decision-making. Incorporating interval imaging to assess changes in nodule characteristics could potentially improve model performance. Tailored risk assessment tools are crucial for optimizing management and reducing unnecessary invasive procedures in this challenging patient population.
– Commentary by Russell Miller, MD, Member of the CHEST Physician Editorial Board
CHEST Critical Care ®
Characterizing Cardiac Function in ICU Survivors of Sepsis
By: Kevin Garrity, MBChB, and colleagues
While chronic cardiac dysfunction is one of the proposed mechanisms of long-term impairment post critical illness, its prevalence, mechanisms, and associations with disability following admission for sepsis are not well understood. Garrity and colleagues describe the Characterization of Cardiovascular Function in ICU Survivors of Sepsis (CONDUCT-ICU) protocol, a prospective study including two ICUs in Scotland aimed to better define cardiovascular dysfunction in survivors of sepsis. Designed to enroll 69 patients, demographics, cardiac and inflammatory biomarkers, and echocardiograms will be obtained on ICU discharge with additional laboratory data, cardiac magnetic resonance imaging, and patient-reported outcome measures to be obtained at 6 to 10 weeks. This novel multimodal approach will provide understanding into the role of cardiovascular dysfunction following critical illness as well as offer mechanistic insights. The investigators hope to obtain operational and pilot data for larger future studies.
– Commentary by Eugene Yuriditsky, MD, FCCP, Member of the CHEST Physician Editorial Board
Sex-Related Differences Found in IgG4-Related Disease Epidemiology
TOPLINE:
Men with immunoglobulin G4 (IgG4)-related disease exhibit significantly lower serum lipase levels and a greater likelihood of organ involvement than women, highlighting significant sex-dependent differences in disease manifestations.
METHODOLOGY:
- Researchers conducted a retrospective study of 328 patients (69% men) diagnosed with IgG4-related disease at the Massachusetts General Hospital – Rheumatology Clinic, Boston, who met the American College of Rheumatology–European Alliance of Associations for Rheumatology (ACR-EULAR) classification criteria between January 2008 and May 2023.
- Among the 328 patients, 69% were men and 31% were women, with a significant male-to-female ratio of 2.2:1.0. Men were typically older at diagnosis (median age, 63.7 vs 58.2 years).
- Data on serum lipase levels, renal involvement, and other clinical and laboratory parameters were collected.
TAKEAWAY:
- Men had higher baseline ACR-EULAR scores, indicating more severe disease (median score of 35.0 vs 29.5; P = .0010).
- Male patients demonstrated a median baseline serum lipase concentration of 24.5 U/L, significantly lower than the 33.5 U/L observed in women.
- Pancreatic (50% vs 26%) or renal (36% vs 18%) involvement was more common in men.
- Men exhibited higher IgG4 levels (P = .0050) and active B-cell responses in the blood (P = .0095).
IN PRACTICE:
According to the authors, this work confirms “the impression of an important sex disparity among patients with IgG4-related disease, with most patients being male, and male patients demonstrating strong tendencies toward more severe disease than female patients.”
SOURCE:
The study was led by Isha Jha, MD, Massachusetts General Hospital, Boston. It was published online on May 30, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The study’s retrospective design may limit the ability to establish causality between sex differences and IgG4-related disease manifestations. A relatively small percentage of patients were assessed before receiving any immunosuppressive treatment, potentially influencing the observed clinical parameters.
DISCLOSURES:
This work was supported by the National Institutes of Health/National Institute of Allergy and Infectious Diseases, the Rheumatology Research Foundation, and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some authors declared financial ties outside this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Men with immunoglobulin G4 (IgG4)-related disease exhibit significantly lower serum lipase levels and a greater likelihood of organ involvement than women, highlighting significant sex-dependent differences in disease manifestations.
METHODOLOGY:
- Researchers conducted a retrospective study of 328 patients (69% men) diagnosed with IgG4-related disease at the Massachusetts General Hospital – Rheumatology Clinic, Boston, who met the American College of Rheumatology–European Alliance of Associations for Rheumatology (ACR-EULAR) classification criteria between January 2008 and May 2023.
- Among the 328 patients, 69% were men and 31% were women, with a significant male-to-female ratio of 2.2:1.0. Men were typically older at diagnosis (median age, 63.7 vs 58.2 years).
- Data on serum lipase levels, renal involvement, and other clinical and laboratory parameters were collected.
TAKEAWAY:
- Men had higher baseline ACR-EULAR scores, indicating more severe disease (median score of 35.0 vs 29.5; P = .0010).
- Male patients demonstrated a median baseline serum lipase concentration of 24.5 U/L, significantly lower than the 33.5 U/L observed in women.
- Pancreatic (50% vs 26%) or renal (36% vs 18%) involvement was more common in men.
- Men exhibited higher IgG4 levels (P = .0050) and active B-cell responses in the blood (P = .0095).
IN PRACTICE:
According to the authors, this work confirms “the impression of an important sex disparity among patients with IgG4-related disease, with most patients being male, and male patients demonstrating strong tendencies toward more severe disease than female patients.”
SOURCE:
The study was led by Isha Jha, MD, Massachusetts General Hospital, Boston. It was published online on May 30, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The study’s retrospective design may limit the ability to establish causality between sex differences and IgG4-related disease manifestations. A relatively small percentage of patients were assessed before receiving any immunosuppressive treatment, potentially influencing the observed clinical parameters.
DISCLOSURES:
This work was supported by the National Institutes of Health/National Institute of Allergy and Infectious Diseases, the Rheumatology Research Foundation, and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some authors declared financial ties outside this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Men with immunoglobulin G4 (IgG4)-related disease exhibit significantly lower serum lipase levels and a greater likelihood of organ involvement than women, highlighting significant sex-dependent differences in disease manifestations.
METHODOLOGY:
- Researchers conducted a retrospective study of 328 patients (69% men) diagnosed with IgG4-related disease at the Massachusetts General Hospital – Rheumatology Clinic, Boston, who met the American College of Rheumatology–European Alliance of Associations for Rheumatology (ACR-EULAR) classification criteria between January 2008 and May 2023.
- Among the 328 patients, 69% were men and 31% were women, with a significant male-to-female ratio of 2.2:1.0. Men were typically older at diagnosis (median age, 63.7 vs 58.2 years).
- Data on serum lipase levels, renal involvement, and other clinical and laboratory parameters were collected.
TAKEAWAY:
- Men had higher baseline ACR-EULAR scores, indicating more severe disease (median score of 35.0 vs 29.5; P = .0010).
- Male patients demonstrated a median baseline serum lipase concentration of 24.5 U/L, significantly lower than the 33.5 U/L observed in women.
- Pancreatic (50% vs 26%) or renal (36% vs 18%) involvement was more common in men.
- Men exhibited higher IgG4 levels (P = .0050) and active B-cell responses in the blood (P = .0095).
IN PRACTICE:
According to the authors, this work confirms “the impression of an important sex disparity among patients with IgG4-related disease, with most patients being male, and male patients demonstrating strong tendencies toward more severe disease than female patients.”
SOURCE:
The study was led by Isha Jha, MD, Massachusetts General Hospital, Boston. It was published online on May 30, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The study’s retrospective design may limit the ability to establish causality between sex differences and IgG4-related disease manifestations. A relatively small percentage of patients were assessed before receiving any immunosuppressive treatment, potentially influencing the observed clinical parameters.
DISCLOSURES:
This work was supported by the National Institutes of Health/National Institute of Allergy and Infectious Diseases, the Rheumatology Research Foundation, and the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Some authors declared financial ties outside this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Autoantibodies Nonspecific to Systemic Sclerosis May Play Role in ILD Prediction
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
FROM EULAR 2024
The Tyranny of Beta-Blockers
Beta-blockers are excellent drugs. They’re cheap and effective; feature prominently in hypertension guidelines; and remain a sine qua non for coronary artery disease, myocardial infarction, and heart failure treatment. They’ve been around forever, and we know they work. Good luck finding an adult medicine patient who isn’t on one.
Beta-blockers act by slowing resting heart rate (and blunting the heart rate response to exercise. The latter is a pernicious cause of activity intolerance that often goes unchecked. Even when the adverse effects of beta-blockers are appreciated, providers are loath to alter dosing, much less stop the drug. After all, beta-blockers are an integral part of guideline-directed medical therapy (GDMT), and GDMT saves lives.
Balancing Heart Rate and Stroke Volume Effects
To augment cardiac output and optimize oxygen uptake (VO2) during exercise, we need the heart rate response. In fact, the heart rate response contributes more to cardiac output than augmenting stroke volume (SV) and more to VO2 than the increase in arteriovenous (AV) oxygen difference. An inability to increase the heart rate commensurate with physiologic work is called chronotropic incompetence (CI). That’s what beta-blockers do ─ they cause CI.
Physiology dictates that CI will cause activity intolerance. That said, it’s hard to quantify the impact from beta-blockers at the individual patient level. Data suggest the heart rate effect is profound. A study in patients without heart failure found that 22% of participants on beta-blockers had CI, and the investigators used a conservative CI definition (≤ 62% of heart rate reserve used). A recent report published in JAMA Cardiology found that stopping beta-blockers in patients with heart failure allowed for an extra 30 beats/min at max exercise.
Wasserman and Whipp’s textbook, the last word on all things exercise, presents a sample subject who undergoes two separate cardiopulmonary exercise tests (CPETs). Before the first, he’s given a placebo, and before the second, he gets an intravenous beta-blocker. He’s a 23-year-old otherwise healthy male — the perfect test case for isolating beta-blocker impact without confounding by comorbid diseases, other medications, or deconditioning. His max heart rate dropped by 30 beats/min after the beta-blocker, identical to what we saw in the JAMA Cardiology study (with the heart rate increasing by 30 beats/min following withdrawal). Case closed. Stop the beta-blockers on your patients so they can meet their exercise goals and get healthy!
Such pithy enthusiasm discounts physiology’s complexities. When blunting our patient’s heart rate response with beta-blockers, we also increase diastolic filling time, which increases SV. For the 23-year-old in Wasserman and Whipp’s physiology textbook, the beta-blocker increased O2 pulse (the product of SV and AV difference). Presumably, this is mediated by the increased SV. There was a net reduction in VO2 peak, but it was nominal, suggesting that the drop in heart rate was largely offset by the increase in O2 pulse. For the patients in the JAMA Cardiology study, the entire group had a small increase in VO2 peak with beta-blocker withdrawal, but the effect differed by left ventricular function. Across different studies, the beta-blocker effect on heart rate is consistent but the change in overall exercise capacity is not.
Patient Variability in Beta-Blocker Response
In addition to left ventricular function, there are other factors likely to drive variability at the patient level. We’ve treated the response to beta-blockers as a class effect — an obvious oversimplification. The impact on exercise and the heart will vary by dose and drug (eg, atenolol vs metoprolol vs carvedilol, and so on). Beta-blockers can also affect the lungs, and we’re still debating how cautious to be in the presence of asthma or chronic obstructive pulmonary disease.
In a world of infinite time, resources, and expertise, we’d CPET everyone before and after beta-blocker use. Our current reality requires the unthinkable: We’ll have to talk to each other and our patients. For example, heart failure guidelines recommend titrating drugs to match the dose from trials that proved efficacy. These doses are quite high. Simple discussion with the cardiologist and the patient may allow for an adjustment back down with careful monitoring and close attention to activity tolerance. With any luck, you’ll preserve the benefits from GDMT while optimizing your patient›s ability to meet their exercise goals.
Dr. Holley, professor in the department of medicine, Uniformed Services University, Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center, Washington, disclosed ties with Metapharm, CHEST College, and WebMD.
A version of this article appeared on Medscape.com.
Beta-blockers are excellent drugs. They’re cheap and effective; feature prominently in hypertension guidelines; and remain a sine qua non for coronary artery disease, myocardial infarction, and heart failure treatment. They’ve been around forever, and we know they work. Good luck finding an adult medicine patient who isn’t on one.
Beta-blockers act by slowing resting heart rate (and blunting the heart rate response to exercise. The latter is a pernicious cause of activity intolerance that often goes unchecked. Even when the adverse effects of beta-blockers are appreciated, providers are loath to alter dosing, much less stop the drug. After all, beta-blockers are an integral part of guideline-directed medical therapy (GDMT), and GDMT saves lives.
Balancing Heart Rate and Stroke Volume Effects
To augment cardiac output and optimize oxygen uptake (VO2) during exercise, we need the heart rate response. In fact, the heart rate response contributes more to cardiac output than augmenting stroke volume (SV) and more to VO2 than the increase in arteriovenous (AV) oxygen difference. An inability to increase the heart rate commensurate with physiologic work is called chronotropic incompetence (CI). That’s what beta-blockers do ─ they cause CI.
Physiology dictates that CI will cause activity intolerance. That said, it’s hard to quantify the impact from beta-blockers at the individual patient level. Data suggest the heart rate effect is profound. A study in patients without heart failure found that 22% of participants on beta-blockers had CI, and the investigators used a conservative CI definition (≤ 62% of heart rate reserve used). A recent report published in JAMA Cardiology found that stopping beta-blockers in patients with heart failure allowed for an extra 30 beats/min at max exercise.
Wasserman and Whipp’s textbook, the last word on all things exercise, presents a sample subject who undergoes two separate cardiopulmonary exercise tests (CPETs). Before the first, he’s given a placebo, and before the second, he gets an intravenous beta-blocker. He’s a 23-year-old otherwise healthy male — the perfect test case for isolating beta-blocker impact without confounding by comorbid diseases, other medications, or deconditioning. His max heart rate dropped by 30 beats/min after the beta-blocker, identical to what we saw in the JAMA Cardiology study (with the heart rate increasing by 30 beats/min following withdrawal). Case closed. Stop the beta-blockers on your patients so they can meet their exercise goals and get healthy!
Such pithy enthusiasm discounts physiology’s complexities. When blunting our patient’s heart rate response with beta-blockers, we also increase diastolic filling time, which increases SV. For the 23-year-old in Wasserman and Whipp’s physiology textbook, the beta-blocker increased O2 pulse (the product of SV and AV difference). Presumably, this is mediated by the increased SV. There was a net reduction in VO2 peak, but it was nominal, suggesting that the drop in heart rate was largely offset by the increase in O2 pulse. For the patients in the JAMA Cardiology study, the entire group had a small increase in VO2 peak with beta-blocker withdrawal, but the effect differed by left ventricular function. Across different studies, the beta-blocker effect on heart rate is consistent but the change in overall exercise capacity is not.
Patient Variability in Beta-Blocker Response
In addition to left ventricular function, there are other factors likely to drive variability at the patient level. We’ve treated the response to beta-blockers as a class effect — an obvious oversimplification. The impact on exercise and the heart will vary by dose and drug (eg, atenolol vs metoprolol vs carvedilol, and so on). Beta-blockers can also affect the lungs, and we’re still debating how cautious to be in the presence of asthma or chronic obstructive pulmonary disease.
In a world of infinite time, resources, and expertise, we’d CPET everyone before and after beta-blocker use. Our current reality requires the unthinkable: We’ll have to talk to each other and our patients. For example, heart failure guidelines recommend titrating drugs to match the dose from trials that proved efficacy. These doses are quite high. Simple discussion with the cardiologist and the patient may allow for an adjustment back down with careful monitoring and close attention to activity tolerance. With any luck, you’ll preserve the benefits from GDMT while optimizing your patient›s ability to meet their exercise goals.
Dr. Holley, professor in the department of medicine, Uniformed Services University, Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center, Washington, disclosed ties with Metapharm, CHEST College, and WebMD.
A version of this article appeared on Medscape.com.
Beta-blockers are excellent drugs. They’re cheap and effective; feature prominently in hypertension guidelines; and remain a sine qua non for coronary artery disease, myocardial infarction, and heart failure treatment. They’ve been around forever, and we know they work. Good luck finding an adult medicine patient who isn’t on one.
Beta-blockers act by slowing resting heart rate (and blunting the heart rate response to exercise. The latter is a pernicious cause of activity intolerance that often goes unchecked. Even when the adverse effects of beta-blockers are appreciated, providers are loath to alter dosing, much less stop the drug. After all, beta-blockers are an integral part of guideline-directed medical therapy (GDMT), and GDMT saves lives.
Balancing Heart Rate and Stroke Volume Effects
To augment cardiac output and optimize oxygen uptake (VO2) during exercise, we need the heart rate response. In fact, the heart rate response contributes more to cardiac output than augmenting stroke volume (SV) and more to VO2 than the increase in arteriovenous (AV) oxygen difference. An inability to increase the heart rate commensurate with physiologic work is called chronotropic incompetence (CI). That’s what beta-blockers do ─ they cause CI.
Physiology dictates that CI will cause activity intolerance. That said, it’s hard to quantify the impact from beta-blockers at the individual patient level. Data suggest the heart rate effect is profound. A study in patients without heart failure found that 22% of participants on beta-blockers had CI, and the investigators used a conservative CI definition (≤ 62% of heart rate reserve used). A recent report published in JAMA Cardiology found that stopping beta-blockers in patients with heart failure allowed for an extra 30 beats/min at max exercise.
Wasserman and Whipp’s textbook, the last word on all things exercise, presents a sample subject who undergoes two separate cardiopulmonary exercise tests (CPETs). Before the first, he’s given a placebo, and before the second, he gets an intravenous beta-blocker. He’s a 23-year-old otherwise healthy male — the perfect test case for isolating beta-blocker impact without confounding by comorbid diseases, other medications, or deconditioning. His max heart rate dropped by 30 beats/min after the beta-blocker, identical to what we saw in the JAMA Cardiology study (with the heart rate increasing by 30 beats/min following withdrawal). Case closed. Stop the beta-blockers on your patients so they can meet their exercise goals and get healthy!
Such pithy enthusiasm discounts physiology’s complexities. When blunting our patient’s heart rate response with beta-blockers, we also increase diastolic filling time, which increases SV. For the 23-year-old in Wasserman and Whipp’s physiology textbook, the beta-blocker increased O2 pulse (the product of SV and AV difference). Presumably, this is mediated by the increased SV. There was a net reduction in VO2 peak, but it was nominal, suggesting that the drop in heart rate was largely offset by the increase in O2 pulse. For the patients in the JAMA Cardiology study, the entire group had a small increase in VO2 peak with beta-blocker withdrawal, but the effect differed by left ventricular function. Across different studies, the beta-blocker effect on heart rate is consistent but the change in overall exercise capacity is not.
Patient Variability in Beta-Blocker Response
In addition to left ventricular function, there are other factors likely to drive variability at the patient level. We’ve treated the response to beta-blockers as a class effect — an obvious oversimplification. The impact on exercise and the heart will vary by dose and drug (eg, atenolol vs metoprolol vs carvedilol, and so on). Beta-blockers can also affect the lungs, and we’re still debating how cautious to be in the presence of asthma or chronic obstructive pulmonary disease.
In a world of infinite time, resources, and expertise, we’d CPET everyone before and after beta-blocker use. Our current reality requires the unthinkable: We’ll have to talk to each other and our patients. For example, heart failure guidelines recommend titrating drugs to match the dose from trials that proved efficacy. These doses are quite high. Simple discussion with the cardiologist and the patient may allow for an adjustment back down with careful monitoring and close attention to activity tolerance. With any luck, you’ll preserve the benefits from GDMT while optimizing your patient›s ability to meet their exercise goals.
Dr. Holley, professor in the department of medicine, Uniformed Services University, Bethesda, Maryland, and a pulmonary/sleep and critical care medicine physician at MedStar Washington Hospital Center, Washington, disclosed ties with Metapharm, CHEST College, and WebMD.
A version of this article appeared on Medscape.com.


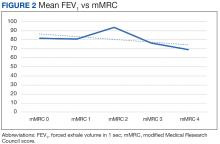
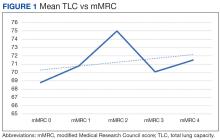
 Author disclosures
Author disclosures


