User login
Evidence Grows for SGLT2 Inhibitors in Rheumatology
Over just a decade, sodium-glucose cotransporter-2 (SGLT2) inhibitors have revolutionized the second-line treatment of type 2 diabetes by improving the control of blood sugar, and they’re also being used to treat heart failure and chronic kidney disease. Now, there’s growing evidence that the medications have the potential to play a role in the treatment of a variety of rheumatologic diseases — gout, systemic lupus erythematosus (SLE), and lupus nephritis.
“I suspect that SGLT2 inhibitors may have a role in multiple rheumatic diseases,” said rheumatologist April Jorge, MD, of Harvard Medical School and Massachusetts General Hospital, Boston.
In gout, for example, “SGLT2 inhibitors hold great promise as a multipurpose treatment option,” said rheumatologist Chio Yokose, MD, MSc, also of Harvard Medical School and Massachusetts General Hospital. Both Dr. Jorge and Dr. Yokose spoke at recent medical conferences and in interviews about the potential value of the drugs in rheumatology.
There’s a big caveat. For the moment, SGLT2 inhibitors aren’t cleared for use in the treatment of rheumatologic conditions, and neither physician is ready to recommend prescribing them off-label outside of their FDA-approved indications.
But studies could pave the way toward more approved uses in rheumatology. And there’s good news for now: Many rheumatology patients may already be eligible to take the drugs because of other medical conditions. In gout, for example, “sizable proportions of patients have comorbidities for which they are already indicated,” Dr. Yokose said.
Research Hints at Gout-Busting Potential
The first SGLT2 inhibitor canagliflozin (Invokana), received FDA approval in 2013, followed by dapagliflozin (Farxiga), empagliflozin (Jardiance), ertugliflozin (Steglatro), and bexagliflozin (Brenzavvy). The drugs “lower blood sugar by causing the kidneys to remove sugar from the body through urine,” reports the National Kidney Foundation, and they “help to protect the kidneys and heart in people with CKD [chronic kidney disease].”
As Dr. Yokose noted in a presentation at the 2023 Gout Hyperuricemia and Crystal Associated Disease Network research symposium, SGLT2 inhibitors “have really become blockbuster drugs, and they’ve now been integrated into multiple professional society guidelines and recommendations.”
These drugs should not be confused with the wildly popular medications known as glucagon-like peptide-1 (GLP1) agonists, which include medications such as semaglutide (Ozempic and Wegovy). These drugs are generally administered via injection — unlike the oral SGLT2 inhibitors — and they’re variously indicated for type 2 diabetes and obesity.
Dr. Yokose highlighted research findings about the drugs in gout. A 2020 study, for example, tracked 295,907 US adults with type 2 diabetes who received a new prescription for an SGLT2 inhibitor or GLP1 agonist during 2013-2017. Those in the SGLT2 inhibitor group had a 36% lower risk of newly diagnosed gout (hazard ratio [HR], 0.64; 95% CI, 0.57-0.72), the researchers reported.
A similar study, a 2021 report from Taiwan, also linked SGLT2 inhibitors to improvement in gout incidence vs. dipeptidyl peptidase 4 (DPP4) inhibitors, diabetes drugs that are not linked to lower serum urate levels. In an adjusted analysis, the risk of gout was 11% lower in the SGLT2 inhibitor group (adjusted HR, 0.86; 95% CI, 0.78-0.95).
What about recurrent gout? In a 2023 study, Dr. Yokose and colleagues tracked patients with type 2 diabetes who began SGLT2 inhibitors or DPP4 inhibitors. Over the period from 2013 to 2017, those who took SGLT2 inhibitors were less likely to have gout flares (rate ratio [RR], 0.66; 95% CI, 0.57-0.75) and gout-primary emergency department visits/hospitalizations (RR, 0.52; 95% CI, 0.32-0.84).
“This finding requires further replication in other populations and compared to other drugs,” Dr. Yokose cautioned.
Another 2023 study analyzed UK data and reached similar results regarding risk of recurrent gout.
Lower Urate Levels and Less Inflammation Could Be Key
How might SGLT2 inhibitors reduce the risk of gout? Multiple studies have linked the drugs to lower serum urate levels, Dr. Yokose said, but researchers often excluded patients with gout.
For a small new study presented at the 2023 annual meeting of the American College of Rheumatology but not yet published, Dr. Yokose and colleagues reported that patients with gout who began SGLT2 inhibitors had lower urate levels than those who began a sulfonylurea, another second-line agent for type 2 diabetes. During the study period, up to 3 months before and after initiation, 43.5% of patients in the SGLT2 inhibitor group reached a target serum urate of < 6 mg/dL vs. 4.2% of sulfonylurea initiators.
“The magnitude of this reduction, while not as large as what can be achieved with appropriately titrated urate-lowering therapy such as allopurinol or febuxostat, is also not negligible. It’s believed to be between 1.5-2.0 mg/dL among patients with gout,” Dr. Yokose said. “Also, SGLT2 inhibitors are purported to have some anti-inflammatory effects that may target the same pathways responsible for the profound inflammation associated with acute gout flares. However, both the exact mechanisms underlying the serum urate-lowering and anti-inflammatory effects of SGLT2 [inhibitors] require further research and clarification.”
Moving forward, she said, “I would love to see some prospective studies of SGLT2 inhibitor use among patients with gout, looking at serum urate and clinical gout endpoints, as well as biomarkers to understand better the beneficial effects of SGLT2 inhibitors as it pertains to patients with gout.”
In Lupus, Findings Are More Mixed
Studies of SGLT2 inhibitors have excluded patients with lupus, limiting insight into their benefits in that specific population, said Dr. Jorge of Massachusetts General Hospital and Harvard Medical School. However, “one small phase I/II trial showed an acceptable safety profile of dapagliflozin add-on therapy in adult patients with SLE,” she said.
Her team is working to expand understanding about the drugs in people with lupus. At the 2023 ACR annual meeting, she presented the findings of a study that tracked patients with SLE who took SGLT2 inhibitors (n = 426, including 154 with lupus nephritis) or DPP4 inhibitors (n = 865, including 270 with lupus nephritis). Patients who took SGLT2 inhibitors had lower risks of major adverse cardiac events (HR, 0.69; 95% CI, 0.48-0.99) and renal progression (HR, 0.71; 95% CI, 0.51-0.98).
“Our results are promising, but the majority of patient with lupus who had received SGLT2 inhibitors also had the comorbidity of type 2 diabetes as a separate indication for SGLT2 inhibitor use,” Dr. Jorge said. “We still need to study the impact of SGLT2 inhibitors in patients with SLE and lupus nephritis who do not have a separate indication for the medication.”
Dr. Jorge added that “we do not yet know the ideal time to initiate SGLT2 inhibitors in the treatment of lupus nephritis. Specifically, it is not yet known whether these medications should be used in patients with persistent proteinuria due to damage from lupus nephritis or whether there is also a role to start these medications in patients with active lupus nephritis who are undergoing induction immunosuppression regimens.”
However, another study released at the 2023 ACR annual meeting suggested that SGLT2 inhibitors may not have a beneficial effect in lupus nephritis: “We observed a reduction in decline in eGFR [estimated glomerular filtration rate] after starting SGLT2 inhibitors; however, this reduction was not statistically significant … early experience suggested marginal benefit of SGLT2 inhibitors in SLE,” researchers from Johns Hopkins University, and the University of Maryland, Baltimore, reported.
“My cohort is not showing miracles from SGLT2 inhibitors,” study lead author Michelle Petri, MD, MPH, of Johns Hopkins, said in an interview.
Still, new European Alliance of Associations for Rheumatology recommendations for SLE now advise to consider the use of the drugs in patients with lupus nephritis who have reduced eGFR. Meanwhile, “the American College of Rheumatology is currently developing new treatment guidelines for SLE and for lupus nephritis, and SGLT2 inhibitors will likely be a topic of consideration,” Dr. Jorge added.
As for mechanism, Dr. Jorge said it’s not clear how the drugs may affect lupus. “It’s proposed that they have benefits in hemodynamic effects as well as potentially anti-inflammatory effects. The hemodynamic effects, including reducing intraglomerular hyperfiltration and reducing blood pressure, likely have similar benefits in patients with chronic kidney disease due to diabetic nephropathy or due to lupus nephritis with damage/scarring and persistent proteinuria. Patients with SLE and other chronic, systemic rheumatic diseases such as ANCA [antineutrophilic cytoplasmic antibody]-associated vasculitis also develop kidney disease and cardiovascular events mediated by inflammatory processes.”
Side Effects and Cost: Where Do They Fit In?
According to Dr. Yokose, SGLT2 inhibitors “are generally quite well-tolerated, and very serious adverse effects are rare.” Side effects include disrupted urination, increased thirst, genital infections, flu-like symptoms, and swelling.
Urinary-related problems are understandable “because these drugs cause the kidneys to pass more glucose into the urine,” University of Hong Kong cardiac specialist Bernard Cheung, MBBCh, PhD, who has studied SGLT2 inhibitors, said in an interview.
In Dr. Yokose’s 2023 study of SGLT2 inhibitors in recurrent gout, patients who took the drugs were 2.15 times more likely than the comparison group to have genital infections (hazard ratio, 2.15; 95% CI, 1.39-3.30). This finding “was what we’d expect,” she said.
She added that genital infection rates were higher among patients with diabetes, women, and uncircumcised men. “Fortunately, most experienced just a single mild episode that can readily be treated with topical therapy. There does not appear to be an increased risk of urinary tract infections.”
Dr. Cheung added that “doctors should be aware of a rare adverse effect called euglycemic ketoacidosis, in which the patient has increased ketones in the blood causing it to be more acidic than normal, but the blood glucose remains within the normal range.”
As for cost, goodrx.com reports that several SGLT2 inhibitors run about $550-$683 per month, making them expensive but still cheaper than GLP-1 agonists, which can cost $1,000 or more per month. Unlike the most popular GLP-1 agonists such as Ozempic, none of the SGLT2 inhibitors are in short supply, according to the American Society of Health-System Pharmacists.
“If someone with gout already has a cardiovascular-kidney-metabolic indication for SGLT2 inhibitors and also stands to benefit in terms of lowering serum urate and risk of recurrent gout flares, there is potential for high benefit relative to cost,” Dr. Yokose said.
She added: “It is well-documented that current gout care is suboptimal, and many patients end up in the emergency room or hospitalized for gout, which in and of itself is quite costly both for the patient and the health care system. Therefore, streamlining or integrating gout and comorbidity care with SGLT2 inhibitors could potentially be quite beneficial for patients with gout.”
In regard to lupus, “many patients with lupus undergo multiple hospitalizations related to their disease, which is a source of high health care costs,” Dr. Jorge said. “Additionally, chronic kidney disease and cardiovascular disease are major causes of disability and premature mortality. Further studies will be needed to better understand whether benefits of SGLT2 inhibitors may outweigh the costs of treatment.”
As for prescribing the drugs in lupus now, Dr. Jorge said they can be an option in lupus nephritis. “There is not a clear consensus of the ideal timing to initiate SGLT2 inhibitors — e.g., degree of proteinuria or eGFR range,” she said. “However, it is less controversial that SGLT2 inhibitors should be considered in particular for patients with lupus nephritis with ongoing proteinuria despite adequate treatment with conventional therapies.”
As for gout, Dr. Yokose isn’t ready to prescribe the drugs to patients who don’t have comorbidities that can be treated by the medications. However, she noted that those patients are rare.
“If I see a patient with gout with one or more of these comorbidities, and I see that they are not already on an SGLT2 inhibitor, I definitely take the time to talk to the patient about this exciting class of drugs and will consult with their other physicians about getting them started on an SGLT2 inhibitor.”
Dr. Yokose, Dr. Petri, and Dr. Cheung have no relevant disclosures. Dr. Jorge disclosed serving as a site investigator for SLE clinical trials funded by Bristol-Myers Squibb and Cabaletta Bio; the trials are not related to SGLT2 inhibitors.
Over just a decade, sodium-glucose cotransporter-2 (SGLT2) inhibitors have revolutionized the second-line treatment of type 2 diabetes by improving the control of blood sugar, and they’re also being used to treat heart failure and chronic kidney disease. Now, there’s growing evidence that the medications have the potential to play a role in the treatment of a variety of rheumatologic diseases — gout, systemic lupus erythematosus (SLE), and lupus nephritis.
“I suspect that SGLT2 inhibitors may have a role in multiple rheumatic diseases,” said rheumatologist April Jorge, MD, of Harvard Medical School and Massachusetts General Hospital, Boston.
In gout, for example, “SGLT2 inhibitors hold great promise as a multipurpose treatment option,” said rheumatologist Chio Yokose, MD, MSc, also of Harvard Medical School and Massachusetts General Hospital. Both Dr. Jorge and Dr. Yokose spoke at recent medical conferences and in interviews about the potential value of the drugs in rheumatology.
There’s a big caveat. For the moment, SGLT2 inhibitors aren’t cleared for use in the treatment of rheumatologic conditions, and neither physician is ready to recommend prescribing them off-label outside of their FDA-approved indications.
But studies could pave the way toward more approved uses in rheumatology. And there’s good news for now: Many rheumatology patients may already be eligible to take the drugs because of other medical conditions. In gout, for example, “sizable proportions of patients have comorbidities for which they are already indicated,” Dr. Yokose said.
Research Hints at Gout-Busting Potential
The first SGLT2 inhibitor canagliflozin (Invokana), received FDA approval in 2013, followed by dapagliflozin (Farxiga), empagliflozin (Jardiance), ertugliflozin (Steglatro), and bexagliflozin (Brenzavvy). The drugs “lower blood sugar by causing the kidneys to remove sugar from the body through urine,” reports the National Kidney Foundation, and they “help to protect the kidneys and heart in people with CKD [chronic kidney disease].”
As Dr. Yokose noted in a presentation at the 2023 Gout Hyperuricemia and Crystal Associated Disease Network research symposium, SGLT2 inhibitors “have really become blockbuster drugs, and they’ve now been integrated into multiple professional society guidelines and recommendations.”
These drugs should not be confused with the wildly popular medications known as glucagon-like peptide-1 (GLP1) agonists, which include medications such as semaglutide (Ozempic and Wegovy). These drugs are generally administered via injection — unlike the oral SGLT2 inhibitors — and they’re variously indicated for type 2 diabetes and obesity.
Dr. Yokose highlighted research findings about the drugs in gout. A 2020 study, for example, tracked 295,907 US adults with type 2 diabetes who received a new prescription for an SGLT2 inhibitor or GLP1 agonist during 2013-2017. Those in the SGLT2 inhibitor group had a 36% lower risk of newly diagnosed gout (hazard ratio [HR], 0.64; 95% CI, 0.57-0.72), the researchers reported.
A similar study, a 2021 report from Taiwan, also linked SGLT2 inhibitors to improvement in gout incidence vs. dipeptidyl peptidase 4 (DPP4) inhibitors, diabetes drugs that are not linked to lower serum urate levels. In an adjusted analysis, the risk of gout was 11% lower in the SGLT2 inhibitor group (adjusted HR, 0.86; 95% CI, 0.78-0.95).
What about recurrent gout? In a 2023 study, Dr. Yokose and colleagues tracked patients with type 2 diabetes who began SGLT2 inhibitors or DPP4 inhibitors. Over the period from 2013 to 2017, those who took SGLT2 inhibitors were less likely to have gout flares (rate ratio [RR], 0.66; 95% CI, 0.57-0.75) and gout-primary emergency department visits/hospitalizations (RR, 0.52; 95% CI, 0.32-0.84).
“This finding requires further replication in other populations and compared to other drugs,” Dr. Yokose cautioned.
Another 2023 study analyzed UK data and reached similar results regarding risk of recurrent gout.
Lower Urate Levels and Less Inflammation Could Be Key
How might SGLT2 inhibitors reduce the risk of gout? Multiple studies have linked the drugs to lower serum urate levels, Dr. Yokose said, but researchers often excluded patients with gout.
For a small new study presented at the 2023 annual meeting of the American College of Rheumatology but not yet published, Dr. Yokose and colleagues reported that patients with gout who began SGLT2 inhibitors had lower urate levels than those who began a sulfonylurea, another second-line agent for type 2 diabetes. During the study period, up to 3 months before and after initiation, 43.5% of patients in the SGLT2 inhibitor group reached a target serum urate of < 6 mg/dL vs. 4.2% of sulfonylurea initiators.
“The magnitude of this reduction, while not as large as what can be achieved with appropriately titrated urate-lowering therapy such as allopurinol or febuxostat, is also not negligible. It’s believed to be between 1.5-2.0 mg/dL among patients with gout,” Dr. Yokose said. “Also, SGLT2 inhibitors are purported to have some anti-inflammatory effects that may target the same pathways responsible for the profound inflammation associated with acute gout flares. However, both the exact mechanisms underlying the serum urate-lowering and anti-inflammatory effects of SGLT2 [inhibitors] require further research and clarification.”
Moving forward, she said, “I would love to see some prospective studies of SGLT2 inhibitor use among patients with gout, looking at serum urate and clinical gout endpoints, as well as biomarkers to understand better the beneficial effects of SGLT2 inhibitors as it pertains to patients with gout.”
In Lupus, Findings Are More Mixed
Studies of SGLT2 inhibitors have excluded patients with lupus, limiting insight into their benefits in that specific population, said Dr. Jorge of Massachusetts General Hospital and Harvard Medical School. However, “one small phase I/II trial showed an acceptable safety profile of dapagliflozin add-on therapy in adult patients with SLE,” she said.
Her team is working to expand understanding about the drugs in people with lupus. At the 2023 ACR annual meeting, she presented the findings of a study that tracked patients with SLE who took SGLT2 inhibitors (n = 426, including 154 with lupus nephritis) or DPP4 inhibitors (n = 865, including 270 with lupus nephritis). Patients who took SGLT2 inhibitors had lower risks of major adverse cardiac events (HR, 0.69; 95% CI, 0.48-0.99) and renal progression (HR, 0.71; 95% CI, 0.51-0.98).
“Our results are promising, but the majority of patient with lupus who had received SGLT2 inhibitors also had the comorbidity of type 2 diabetes as a separate indication for SGLT2 inhibitor use,” Dr. Jorge said. “We still need to study the impact of SGLT2 inhibitors in patients with SLE and lupus nephritis who do not have a separate indication for the medication.”
Dr. Jorge added that “we do not yet know the ideal time to initiate SGLT2 inhibitors in the treatment of lupus nephritis. Specifically, it is not yet known whether these medications should be used in patients with persistent proteinuria due to damage from lupus nephritis or whether there is also a role to start these medications in patients with active lupus nephritis who are undergoing induction immunosuppression regimens.”
However, another study released at the 2023 ACR annual meeting suggested that SGLT2 inhibitors may not have a beneficial effect in lupus nephritis: “We observed a reduction in decline in eGFR [estimated glomerular filtration rate] after starting SGLT2 inhibitors; however, this reduction was not statistically significant … early experience suggested marginal benefit of SGLT2 inhibitors in SLE,” researchers from Johns Hopkins University, and the University of Maryland, Baltimore, reported.
“My cohort is not showing miracles from SGLT2 inhibitors,” study lead author Michelle Petri, MD, MPH, of Johns Hopkins, said in an interview.
Still, new European Alliance of Associations for Rheumatology recommendations for SLE now advise to consider the use of the drugs in patients with lupus nephritis who have reduced eGFR. Meanwhile, “the American College of Rheumatology is currently developing new treatment guidelines for SLE and for lupus nephritis, and SGLT2 inhibitors will likely be a topic of consideration,” Dr. Jorge added.
As for mechanism, Dr. Jorge said it’s not clear how the drugs may affect lupus. “It’s proposed that they have benefits in hemodynamic effects as well as potentially anti-inflammatory effects. The hemodynamic effects, including reducing intraglomerular hyperfiltration and reducing blood pressure, likely have similar benefits in patients with chronic kidney disease due to diabetic nephropathy or due to lupus nephritis with damage/scarring and persistent proteinuria. Patients with SLE and other chronic, systemic rheumatic diseases such as ANCA [antineutrophilic cytoplasmic antibody]-associated vasculitis also develop kidney disease and cardiovascular events mediated by inflammatory processes.”
Side Effects and Cost: Where Do They Fit In?
According to Dr. Yokose, SGLT2 inhibitors “are generally quite well-tolerated, and very serious adverse effects are rare.” Side effects include disrupted urination, increased thirst, genital infections, flu-like symptoms, and swelling.
Urinary-related problems are understandable “because these drugs cause the kidneys to pass more glucose into the urine,” University of Hong Kong cardiac specialist Bernard Cheung, MBBCh, PhD, who has studied SGLT2 inhibitors, said in an interview.
In Dr. Yokose’s 2023 study of SGLT2 inhibitors in recurrent gout, patients who took the drugs were 2.15 times more likely than the comparison group to have genital infections (hazard ratio, 2.15; 95% CI, 1.39-3.30). This finding “was what we’d expect,” she said.
She added that genital infection rates were higher among patients with diabetes, women, and uncircumcised men. “Fortunately, most experienced just a single mild episode that can readily be treated with topical therapy. There does not appear to be an increased risk of urinary tract infections.”
Dr. Cheung added that “doctors should be aware of a rare adverse effect called euglycemic ketoacidosis, in which the patient has increased ketones in the blood causing it to be more acidic than normal, but the blood glucose remains within the normal range.”
As for cost, goodrx.com reports that several SGLT2 inhibitors run about $550-$683 per month, making them expensive but still cheaper than GLP-1 agonists, which can cost $1,000 or more per month. Unlike the most popular GLP-1 agonists such as Ozempic, none of the SGLT2 inhibitors are in short supply, according to the American Society of Health-System Pharmacists.
“If someone with gout already has a cardiovascular-kidney-metabolic indication for SGLT2 inhibitors and also stands to benefit in terms of lowering serum urate and risk of recurrent gout flares, there is potential for high benefit relative to cost,” Dr. Yokose said.
She added: “It is well-documented that current gout care is suboptimal, and many patients end up in the emergency room or hospitalized for gout, which in and of itself is quite costly both for the patient and the health care system. Therefore, streamlining or integrating gout and comorbidity care with SGLT2 inhibitors could potentially be quite beneficial for patients with gout.”
In regard to lupus, “many patients with lupus undergo multiple hospitalizations related to their disease, which is a source of high health care costs,” Dr. Jorge said. “Additionally, chronic kidney disease and cardiovascular disease are major causes of disability and premature mortality. Further studies will be needed to better understand whether benefits of SGLT2 inhibitors may outweigh the costs of treatment.”
As for prescribing the drugs in lupus now, Dr. Jorge said they can be an option in lupus nephritis. “There is not a clear consensus of the ideal timing to initiate SGLT2 inhibitors — e.g., degree of proteinuria or eGFR range,” she said. “However, it is less controversial that SGLT2 inhibitors should be considered in particular for patients with lupus nephritis with ongoing proteinuria despite adequate treatment with conventional therapies.”
As for gout, Dr. Yokose isn’t ready to prescribe the drugs to patients who don’t have comorbidities that can be treated by the medications. However, she noted that those patients are rare.
“If I see a patient with gout with one or more of these comorbidities, and I see that they are not already on an SGLT2 inhibitor, I definitely take the time to talk to the patient about this exciting class of drugs and will consult with their other physicians about getting them started on an SGLT2 inhibitor.”
Dr. Yokose, Dr. Petri, and Dr. Cheung have no relevant disclosures. Dr. Jorge disclosed serving as a site investigator for SLE clinical trials funded by Bristol-Myers Squibb and Cabaletta Bio; the trials are not related to SGLT2 inhibitors.
Over just a decade, sodium-glucose cotransporter-2 (SGLT2) inhibitors have revolutionized the second-line treatment of type 2 diabetes by improving the control of blood sugar, and they’re also being used to treat heart failure and chronic kidney disease. Now, there’s growing evidence that the medications have the potential to play a role in the treatment of a variety of rheumatologic diseases — gout, systemic lupus erythematosus (SLE), and lupus nephritis.
“I suspect that SGLT2 inhibitors may have a role in multiple rheumatic diseases,” said rheumatologist April Jorge, MD, of Harvard Medical School and Massachusetts General Hospital, Boston.
In gout, for example, “SGLT2 inhibitors hold great promise as a multipurpose treatment option,” said rheumatologist Chio Yokose, MD, MSc, also of Harvard Medical School and Massachusetts General Hospital. Both Dr. Jorge and Dr. Yokose spoke at recent medical conferences and in interviews about the potential value of the drugs in rheumatology.
There’s a big caveat. For the moment, SGLT2 inhibitors aren’t cleared for use in the treatment of rheumatologic conditions, and neither physician is ready to recommend prescribing them off-label outside of their FDA-approved indications.
But studies could pave the way toward more approved uses in rheumatology. And there’s good news for now: Many rheumatology patients may already be eligible to take the drugs because of other medical conditions. In gout, for example, “sizable proportions of patients have comorbidities for which they are already indicated,” Dr. Yokose said.
Research Hints at Gout-Busting Potential
The first SGLT2 inhibitor canagliflozin (Invokana), received FDA approval in 2013, followed by dapagliflozin (Farxiga), empagliflozin (Jardiance), ertugliflozin (Steglatro), and bexagliflozin (Brenzavvy). The drugs “lower blood sugar by causing the kidneys to remove sugar from the body through urine,” reports the National Kidney Foundation, and they “help to protect the kidneys and heart in people with CKD [chronic kidney disease].”
As Dr. Yokose noted in a presentation at the 2023 Gout Hyperuricemia and Crystal Associated Disease Network research symposium, SGLT2 inhibitors “have really become blockbuster drugs, and they’ve now been integrated into multiple professional society guidelines and recommendations.”
These drugs should not be confused with the wildly popular medications known as glucagon-like peptide-1 (GLP1) agonists, which include medications such as semaglutide (Ozempic and Wegovy). These drugs are generally administered via injection — unlike the oral SGLT2 inhibitors — and they’re variously indicated for type 2 diabetes and obesity.
Dr. Yokose highlighted research findings about the drugs in gout. A 2020 study, for example, tracked 295,907 US adults with type 2 diabetes who received a new prescription for an SGLT2 inhibitor or GLP1 agonist during 2013-2017. Those in the SGLT2 inhibitor group had a 36% lower risk of newly diagnosed gout (hazard ratio [HR], 0.64; 95% CI, 0.57-0.72), the researchers reported.
A similar study, a 2021 report from Taiwan, also linked SGLT2 inhibitors to improvement in gout incidence vs. dipeptidyl peptidase 4 (DPP4) inhibitors, diabetes drugs that are not linked to lower serum urate levels. In an adjusted analysis, the risk of gout was 11% lower in the SGLT2 inhibitor group (adjusted HR, 0.86; 95% CI, 0.78-0.95).
What about recurrent gout? In a 2023 study, Dr. Yokose and colleagues tracked patients with type 2 diabetes who began SGLT2 inhibitors or DPP4 inhibitors. Over the period from 2013 to 2017, those who took SGLT2 inhibitors were less likely to have gout flares (rate ratio [RR], 0.66; 95% CI, 0.57-0.75) and gout-primary emergency department visits/hospitalizations (RR, 0.52; 95% CI, 0.32-0.84).
“This finding requires further replication in other populations and compared to other drugs,” Dr. Yokose cautioned.
Another 2023 study analyzed UK data and reached similar results regarding risk of recurrent gout.
Lower Urate Levels and Less Inflammation Could Be Key
How might SGLT2 inhibitors reduce the risk of gout? Multiple studies have linked the drugs to lower serum urate levels, Dr. Yokose said, but researchers often excluded patients with gout.
For a small new study presented at the 2023 annual meeting of the American College of Rheumatology but not yet published, Dr. Yokose and colleagues reported that patients with gout who began SGLT2 inhibitors had lower urate levels than those who began a sulfonylurea, another second-line agent for type 2 diabetes. During the study period, up to 3 months before and after initiation, 43.5% of patients in the SGLT2 inhibitor group reached a target serum urate of < 6 mg/dL vs. 4.2% of sulfonylurea initiators.
“The magnitude of this reduction, while not as large as what can be achieved with appropriately titrated urate-lowering therapy such as allopurinol or febuxostat, is also not negligible. It’s believed to be between 1.5-2.0 mg/dL among patients with gout,” Dr. Yokose said. “Also, SGLT2 inhibitors are purported to have some anti-inflammatory effects that may target the same pathways responsible for the profound inflammation associated with acute gout flares. However, both the exact mechanisms underlying the serum urate-lowering and anti-inflammatory effects of SGLT2 [inhibitors] require further research and clarification.”
Moving forward, she said, “I would love to see some prospective studies of SGLT2 inhibitor use among patients with gout, looking at serum urate and clinical gout endpoints, as well as biomarkers to understand better the beneficial effects of SGLT2 inhibitors as it pertains to patients with gout.”
In Lupus, Findings Are More Mixed
Studies of SGLT2 inhibitors have excluded patients with lupus, limiting insight into their benefits in that specific population, said Dr. Jorge of Massachusetts General Hospital and Harvard Medical School. However, “one small phase I/II trial showed an acceptable safety profile of dapagliflozin add-on therapy in adult patients with SLE,” she said.
Her team is working to expand understanding about the drugs in people with lupus. At the 2023 ACR annual meeting, she presented the findings of a study that tracked patients with SLE who took SGLT2 inhibitors (n = 426, including 154 with lupus nephritis) or DPP4 inhibitors (n = 865, including 270 with lupus nephritis). Patients who took SGLT2 inhibitors had lower risks of major adverse cardiac events (HR, 0.69; 95% CI, 0.48-0.99) and renal progression (HR, 0.71; 95% CI, 0.51-0.98).
“Our results are promising, but the majority of patient with lupus who had received SGLT2 inhibitors also had the comorbidity of type 2 diabetes as a separate indication for SGLT2 inhibitor use,” Dr. Jorge said. “We still need to study the impact of SGLT2 inhibitors in patients with SLE and lupus nephritis who do not have a separate indication for the medication.”
Dr. Jorge added that “we do not yet know the ideal time to initiate SGLT2 inhibitors in the treatment of lupus nephritis. Specifically, it is not yet known whether these medications should be used in patients with persistent proteinuria due to damage from lupus nephritis or whether there is also a role to start these medications in patients with active lupus nephritis who are undergoing induction immunosuppression regimens.”
However, another study released at the 2023 ACR annual meeting suggested that SGLT2 inhibitors may not have a beneficial effect in lupus nephritis: “We observed a reduction in decline in eGFR [estimated glomerular filtration rate] after starting SGLT2 inhibitors; however, this reduction was not statistically significant … early experience suggested marginal benefit of SGLT2 inhibitors in SLE,” researchers from Johns Hopkins University, and the University of Maryland, Baltimore, reported.
“My cohort is not showing miracles from SGLT2 inhibitors,” study lead author Michelle Petri, MD, MPH, of Johns Hopkins, said in an interview.
Still, new European Alliance of Associations for Rheumatology recommendations for SLE now advise to consider the use of the drugs in patients with lupus nephritis who have reduced eGFR. Meanwhile, “the American College of Rheumatology is currently developing new treatment guidelines for SLE and for lupus nephritis, and SGLT2 inhibitors will likely be a topic of consideration,” Dr. Jorge added.
As for mechanism, Dr. Jorge said it’s not clear how the drugs may affect lupus. “It’s proposed that they have benefits in hemodynamic effects as well as potentially anti-inflammatory effects. The hemodynamic effects, including reducing intraglomerular hyperfiltration and reducing blood pressure, likely have similar benefits in patients with chronic kidney disease due to diabetic nephropathy or due to lupus nephritis with damage/scarring and persistent proteinuria. Patients with SLE and other chronic, systemic rheumatic diseases such as ANCA [antineutrophilic cytoplasmic antibody]-associated vasculitis also develop kidney disease and cardiovascular events mediated by inflammatory processes.”
Side Effects and Cost: Where Do They Fit In?
According to Dr. Yokose, SGLT2 inhibitors “are generally quite well-tolerated, and very serious adverse effects are rare.” Side effects include disrupted urination, increased thirst, genital infections, flu-like symptoms, and swelling.
Urinary-related problems are understandable “because these drugs cause the kidneys to pass more glucose into the urine,” University of Hong Kong cardiac specialist Bernard Cheung, MBBCh, PhD, who has studied SGLT2 inhibitors, said in an interview.
In Dr. Yokose’s 2023 study of SGLT2 inhibitors in recurrent gout, patients who took the drugs were 2.15 times more likely than the comparison group to have genital infections (hazard ratio, 2.15; 95% CI, 1.39-3.30). This finding “was what we’d expect,” she said.
She added that genital infection rates were higher among patients with diabetes, women, and uncircumcised men. “Fortunately, most experienced just a single mild episode that can readily be treated with topical therapy. There does not appear to be an increased risk of urinary tract infections.”
Dr. Cheung added that “doctors should be aware of a rare adverse effect called euglycemic ketoacidosis, in which the patient has increased ketones in the blood causing it to be more acidic than normal, but the blood glucose remains within the normal range.”
As for cost, goodrx.com reports that several SGLT2 inhibitors run about $550-$683 per month, making them expensive but still cheaper than GLP-1 agonists, which can cost $1,000 or more per month. Unlike the most popular GLP-1 agonists such as Ozempic, none of the SGLT2 inhibitors are in short supply, according to the American Society of Health-System Pharmacists.
“If someone with gout already has a cardiovascular-kidney-metabolic indication for SGLT2 inhibitors and also stands to benefit in terms of lowering serum urate and risk of recurrent gout flares, there is potential for high benefit relative to cost,” Dr. Yokose said.
She added: “It is well-documented that current gout care is suboptimal, and many patients end up in the emergency room or hospitalized for gout, which in and of itself is quite costly both for the patient and the health care system. Therefore, streamlining or integrating gout and comorbidity care with SGLT2 inhibitors could potentially be quite beneficial for patients with gout.”
In regard to lupus, “many patients with lupus undergo multiple hospitalizations related to their disease, which is a source of high health care costs,” Dr. Jorge said. “Additionally, chronic kidney disease and cardiovascular disease are major causes of disability and premature mortality. Further studies will be needed to better understand whether benefits of SGLT2 inhibitors may outweigh the costs of treatment.”
As for prescribing the drugs in lupus now, Dr. Jorge said they can be an option in lupus nephritis. “There is not a clear consensus of the ideal timing to initiate SGLT2 inhibitors — e.g., degree of proteinuria or eGFR range,” she said. “However, it is less controversial that SGLT2 inhibitors should be considered in particular for patients with lupus nephritis with ongoing proteinuria despite adequate treatment with conventional therapies.”
As for gout, Dr. Yokose isn’t ready to prescribe the drugs to patients who don’t have comorbidities that can be treated by the medications. However, she noted that those patients are rare.
“If I see a patient with gout with one or more of these comorbidities, and I see that they are not already on an SGLT2 inhibitor, I definitely take the time to talk to the patient about this exciting class of drugs and will consult with their other physicians about getting them started on an SGLT2 inhibitor.”
Dr. Yokose, Dr. Petri, and Dr. Cheung have no relevant disclosures. Dr. Jorge disclosed serving as a site investigator for SLE clinical trials funded by Bristol-Myers Squibb and Cabaletta Bio; the trials are not related to SGLT2 inhibitors.
Redosing Rituximab to Maintain ANCA Vasculitis Remission: When Is Best?
Maintaining remission in patients with antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis who have kept their autoantibodies in check after at least 2 years on rituximab therapy has proved challenging, but a team of nephrologists in Boston have reported that a longer-term strategy that uses a rise in B-cell levels as a threshold for rituximab infusions may be the better of two strategies at reducing relapse risks.
“The bottom line is with the B-cell strategy, which is that rituximab was redosed when the B cells recovered or started to recover, we only have a 6% rate in relapses by 3 years,” senior study author John L. Niles, MD, assistant professor of medicine at the Harvard Medical School and director of the Vasculitis and Glomerulonephritis Center at Massachusetts General Hospital in Boston, Massachusetts, told Medscape Medical News.
“Whereas in the other strategy, we were waiting for a serologic relapse and hoping we could prevent clinical relapses, but we still have about 30% rate of relapse by 3 years.”
Dr. Niles and his associates reported their findings from the MAINTANCVAS study (for MAINTenance of ANCA VASculitis) December 11, 2023, in Annals of the Rheumatic Diseases. Their single-center study compared two different treatment strategies in patients with ANCA-associated vasculitis in remission after completing at least 2 years of fixed-schedule rituximab therapy: an approach that reinfused rituximab upon B-cell repopulation, called the B-cell arm and a strategy that reinfused rituximab when serologic levels of ANCA increased significantly, which they called the ANCA arm. A total of 115 patients were randomly assigned to either arm.
Study Results
Median follow-up was 4.1 years from study entry. Throughout the study, 5 of 58 patients in the B-cell arm and 14 of 57 in the ANCA arm had relapses. According to Kaplan-Meier analysis, at 3 years after study entry, 4.1% of patients in the B-cell arm had a relapse vs 20.5% of patients in the ANCA arm. At 5 years, the respective relapse rates were 11.3% and 27.7%. Overall, four major relapses occurred in the B-cell arm and seven in the ANCA arm.
The COVID-19 pandemic caused the researchers to halt the study before it was fully enrolled, Dr. Niles said. The study also attributed high rates of serious adverse events (SAEs) in the B-cell arm to cases of COVID-19 in that study population. The overall number of SAEs was identical in both arms: 22 (P = .95). But the B-cell arm had six cases of COVID-19 vs one in the ANCA arm, including two deaths because of COVID-19.
The study findings provided insight into how to best individualize treatment in patients with ANCA-associated vasculitis, Dr. Niles said. “We will typically start with the B-cell strategy after 2 years, but to the extent that people have infections or hypogammaglobulinemia, we’ll start stretching a little longer on the B cells, and if the level is too high in terms of infection, we’ll stop and switch to the ANCA strategy,” he said.
He added, “Relapsers get a more strict B-cell strategy, and people with infections get much longer intervals or even switch entirely to the ANCA strategy.”
Because the study ended before it was fully enrolled, it was underpowered for subgroup analyses, Dr. Niles noted. One such potential subgroup was relapsing patients with interstitial lung disease as the primary clinical finding. “The interstitial lung disease doesn’t seem to respond as well to therapy as the other classic features of ANCA disease,” Dr. Niles said. “It’s the one part that’s the most problematic for the long run. It behaves differently, and there’s going to need to be more research on ILD. Fortunately, it’s a fairly small percentage of the total group, but it’s the most difficult part of this disease.”
Findings in Context
This study brings clarity on how to best manage patients with ANCA-associated vasculitis, Robert Hylland, MD, an assistant clinical professor of rheumatology at Michigan State University College of Osteopathic Medicine, told this news organization.
“Most of us have tried to discern from the literature that exists how to manage [ANCA-associated vasculitis]. There have been a number of different approaches, and they have changed over the course of time,” Dr. Hylland said. “But now this article helps us to understand how to proceed with this disease after we have induced remission. The ability to determine the validity of serology vs B-cell depletion was brought out very nicely in this article.”
The size of the study population was a strength of the study, Dr. Hylland said.
He credited the study authors for providing insight into using positive myeloperoxidase (MPO)- or proteinase 3 (PR3)-ANCA readings to guide treatment for relapses. The study defined a serologic ANCA flare in the ANCA arm as a fivefold increase in MPO and a fourfold rise in PR3.
“Many of us wouldn’t have recognized that a less than fivefold increase, for example, in the MPO could be watched for a while, where most of us would have been treating that serologic flare,” Hylland said.
The study also highlighted the difficulty of evaluating a patient who has neither a positive ANCA nor a significant increase in their B-cell counts and yet still has clinical signs and symptoms of a relapse, such as with granulomatosis with polyangiitis, also known as Wegener’s granulomatosis.
“A lot of physicians tend to feel a little more relaxed when they see their patient is serologically doing well and yet, when they come in, some of the subtle symptoms of Wegener’s could be ignored if you don’t recognize that there’s a considerable number who will come to you with having had treatment and still have negative serology,” Hylland said.
The study had no specific outside funding source. Dr. Niles and Dr. Hylland report no relevant financial relationships. Two co-authors report financial relationships with pharmaceutical companies.
A version of this article appeared on Medscape.com.
Maintaining remission in patients with antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis who have kept their autoantibodies in check after at least 2 years on rituximab therapy has proved challenging, but a team of nephrologists in Boston have reported that a longer-term strategy that uses a rise in B-cell levels as a threshold for rituximab infusions may be the better of two strategies at reducing relapse risks.
“The bottom line is with the B-cell strategy, which is that rituximab was redosed when the B cells recovered or started to recover, we only have a 6% rate in relapses by 3 years,” senior study author John L. Niles, MD, assistant professor of medicine at the Harvard Medical School and director of the Vasculitis and Glomerulonephritis Center at Massachusetts General Hospital in Boston, Massachusetts, told Medscape Medical News.
“Whereas in the other strategy, we were waiting for a serologic relapse and hoping we could prevent clinical relapses, but we still have about 30% rate of relapse by 3 years.”
Dr. Niles and his associates reported their findings from the MAINTANCVAS study (for MAINTenance of ANCA VASculitis) December 11, 2023, in Annals of the Rheumatic Diseases. Their single-center study compared two different treatment strategies in patients with ANCA-associated vasculitis in remission after completing at least 2 years of fixed-schedule rituximab therapy: an approach that reinfused rituximab upon B-cell repopulation, called the B-cell arm and a strategy that reinfused rituximab when serologic levels of ANCA increased significantly, which they called the ANCA arm. A total of 115 patients were randomly assigned to either arm.
Study Results
Median follow-up was 4.1 years from study entry. Throughout the study, 5 of 58 patients in the B-cell arm and 14 of 57 in the ANCA arm had relapses. According to Kaplan-Meier analysis, at 3 years after study entry, 4.1% of patients in the B-cell arm had a relapse vs 20.5% of patients in the ANCA arm. At 5 years, the respective relapse rates were 11.3% and 27.7%. Overall, four major relapses occurred in the B-cell arm and seven in the ANCA arm.
The COVID-19 pandemic caused the researchers to halt the study before it was fully enrolled, Dr. Niles said. The study also attributed high rates of serious adverse events (SAEs) in the B-cell arm to cases of COVID-19 in that study population. The overall number of SAEs was identical in both arms: 22 (P = .95). But the B-cell arm had six cases of COVID-19 vs one in the ANCA arm, including two deaths because of COVID-19.
The study findings provided insight into how to best individualize treatment in patients with ANCA-associated vasculitis, Dr. Niles said. “We will typically start with the B-cell strategy after 2 years, but to the extent that people have infections or hypogammaglobulinemia, we’ll start stretching a little longer on the B cells, and if the level is too high in terms of infection, we’ll stop and switch to the ANCA strategy,” he said.
He added, “Relapsers get a more strict B-cell strategy, and people with infections get much longer intervals or even switch entirely to the ANCA strategy.”
Because the study ended before it was fully enrolled, it was underpowered for subgroup analyses, Dr. Niles noted. One such potential subgroup was relapsing patients with interstitial lung disease as the primary clinical finding. “The interstitial lung disease doesn’t seem to respond as well to therapy as the other classic features of ANCA disease,” Dr. Niles said. “It’s the one part that’s the most problematic for the long run. It behaves differently, and there’s going to need to be more research on ILD. Fortunately, it’s a fairly small percentage of the total group, but it’s the most difficult part of this disease.”
Findings in Context
This study brings clarity on how to best manage patients with ANCA-associated vasculitis, Robert Hylland, MD, an assistant clinical professor of rheumatology at Michigan State University College of Osteopathic Medicine, told this news organization.
“Most of us have tried to discern from the literature that exists how to manage [ANCA-associated vasculitis]. There have been a number of different approaches, and they have changed over the course of time,” Dr. Hylland said. “But now this article helps us to understand how to proceed with this disease after we have induced remission. The ability to determine the validity of serology vs B-cell depletion was brought out very nicely in this article.”
The size of the study population was a strength of the study, Dr. Hylland said.
He credited the study authors for providing insight into using positive myeloperoxidase (MPO)- or proteinase 3 (PR3)-ANCA readings to guide treatment for relapses. The study defined a serologic ANCA flare in the ANCA arm as a fivefold increase in MPO and a fourfold rise in PR3.
“Many of us wouldn’t have recognized that a less than fivefold increase, for example, in the MPO could be watched for a while, where most of us would have been treating that serologic flare,” Hylland said.
The study also highlighted the difficulty of evaluating a patient who has neither a positive ANCA nor a significant increase in their B-cell counts and yet still has clinical signs and symptoms of a relapse, such as with granulomatosis with polyangiitis, also known as Wegener’s granulomatosis.
“A lot of physicians tend to feel a little more relaxed when they see their patient is serologically doing well and yet, when they come in, some of the subtle symptoms of Wegener’s could be ignored if you don’t recognize that there’s a considerable number who will come to you with having had treatment and still have negative serology,” Hylland said.
The study had no specific outside funding source. Dr. Niles and Dr. Hylland report no relevant financial relationships. Two co-authors report financial relationships with pharmaceutical companies.
A version of this article appeared on Medscape.com.
Maintaining remission in patients with antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis who have kept their autoantibodies in check after at least 2 years on rituximab therapy has proved challenging, but a team of nephrologists in Boston have reported that a longer-term strategy that uses a rise in B-cell levels as a threshold for rituximab infusions may be the better of two strategies at reducing relapse risks.
“The bottom line is with the B-cell strategy, which is that rituximab was redosed when the B cells recovered or started to recover, we only have a 6% rate in relapses by 3 years,” senior study author John L. Niles, MD, assistant professor of medicine at the Harvard Medical School and director of the Vasculitis and Glomerulonephritis Center at Massachusetts General Hospital in Boston, Massachusetts, told Medscape Medical News.
“Whereas in the other strategy, we were waiting for a serologic relapse and hoping we could prevent clinical relapses, but we still have about 30% rate of relapse by 3 years.”
Dr. Niles and his associates reported their findings from the MAINTANCVAS study (for MAINTenance of ANCA VASculitis) December 11, 2023, in Annals of the Rheumatic Diseases. Their single-center study compared two different treatment strategies in patients with ANCA-associated vasculitis in remission after completing at least 2 years of fixed-schedule rituximab therapy: an approach that reinfused rituximab upon B-cell repopulation, called the B-cell arm and a strategy that reinfused rituximab when serologic levels of ANCA increased significantly, which they called the ANCA arm. A total of 115 patients were randomly assigned to either arm.
Study Results
Median follow-up was 4.1 years from study entry. Throughout the study, 5 of 58 patients in the B-cell arm and 14 of 57 in the ANCA arm had relapses. According to Kaplan-Meier analysis, at 3 years after study entry, 4.1% of patients in the B-cell arm had a relapse vs 20.5% of patients in the ANCA arm. At 5 years, the respective relapse rates were 11.3% and 27.7%. Overall, four major relapses occurred in the B-cell arm and seven in the ANCA arm.
The COVID-19 pandemic caused the researchers to halt the study before it was fully enrolled, Dr. Niles said. The study also attributed high rates of serious adverse events (SAEs) in the B-cell arm to cases of COVID-19 in that study population. The overall number of SAEs was identical in both arms: 22 (P = .95). But the B-cell arm had six cases of COVID-19 vs one in the ANCA arm, including two deaths because of COVID-19.
The study findings provided insight into how to best individualize treatment in patients with ANCA-associated vasculitis, Dr. Niles said. “We will typically start with the B-cell strategy after 2 years, but to the extent that people have infections or hypogammaglobulinemia, we’ll start stretching a little longer on the B cells, and if the level is too high in terms of infection, we’ll stop and switch to the ANCA strategy,” he said.
He added, “Relapsers get a more strict B-cell strategy, and people with infections get much longer intervals or even switch entirely to the ANCA strategy.”
Because the study ended before it was fully enrolled, it was underpowered for subgroup analyses, Dr. Niles noted. One such potential subgroup was relapsing patients with interstitial lung disease as the primary clinical finding. “The interstitial lung disease doesn’t seem to respond as well to therapy as the other classic features of ANCA disease,” Dr. Niles said. “It’s the one part that’s the most problematic for the long run. It behaves differently, and there’s going to need to be more research on ILD. Fortunately, it’s a fairly small percentage of the total group, but it’s the most difficult part of this disease.”
Findings in Context
This study brings clarity on how to best manage patients with ANCA-associated vasculitis, Robert Hylland, MD, an assistant clinical professor of rheumatology at Michigan State University College of Osteopathic Medicine, told this news organization.
“Most of us have tried to discern from the literature that exists how to manage [ANCA-associated vasculitis]. There have been a number of different approaches, and they have changed over the course of time,” Dr. Hylland said. “But now this article helps us to understand how to proceed with this disease after we have induced remission. The ability to determine the validity of serology vs B-cell depletion was brought out very nicely in this article.”
The size of the study population was a strength of the study, Dr. Hylland said.
He credited the study authors for providing insight into using positive myeloperoxidase (MPO)- or proteinase 3 (PR3)-ANCA readings to guide treatment for relapses. The study defined a serologic ANCA flare in the ANCA arm as a fivefold increase in MPO and a fourfold rise in PR3.
“Many of us wouldn’t have recognized that a less than fivefold increase, for example, in the MPO could be watched for a while, where most of us would have been treating that serologic flare,” Hylland said.
The study also highlighted the difficulty of evaluating a patient who has neither a positive ANCA nor a significant increase in their B-cell counts and yet still has clinical signs and symptoms of a relapse, such as with granulomatosis with polyangiitis, also known as Wegener’s granulomatosis.
“A lot of physicians tend to feel a little more relaxed when they see their patient is serologically doing well and yet, when they come in, some of the subtle symptoms of Wegener’s could be ignored if you don’t recognize that there’s a considerable number who will come to you with having had treatment and still have negative serology,” Hylland said.
The study had no specific outside funding source. Dr. Niles and Dr. Hylland report no relevant financial relationships. Two co-authors report financial relationships with pharmaceutical companies.
A version of this article appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Conditional recommendations rule in new SARD-associated interstitial lung disease guidelines
SAN DIEGO – In the spring of 2024, the American College of Rheumatology is expected to release guidelines to help inform the screening, monitoring, and treatment of interstitial lung disease (ILD) in people with systemic autoimmune rheumatic diseases (SARDs).
The guidelines, which were previewed during a session at the ACR’s annual meeting, will include 50 recommendations, 3 of which met criteria for a strong rating:
- For people with SARDs at increased risk of developing ILD, the authors strongly recommend against screening with surgical lung biopsy.
- For people with systemic sclerosis (SSc)-related ILD, the authors strongly recommend against glucocorticoids as a first-line ILD treatment.
- For people with SSc-related ILD progression despite an initial ILD treatment, the authors strongly recommend against using long-term glucocorticoids.
Elana J. Bernstein, MD, MSc, a rheumatologist who directs the Columbia/New York-Presbyterian Scleroderma Center, and Sindhu R. Johnson, MD, a rheumatologist who directs the Toronto Scleroderma Program at the University of Toronto, provided a sneak peek of the recommendations to attendees before anticipated publication in Arthritis & Rheumatology and Arthritis Care & Research. For now, guideline summaries for screening and monitoring and treatment are currently available, and three manuscripts are under peer review: one about screening and monitoring, one about treatment, and one about the patient panel that participated in the effort.
“ILD is a significant cause of morbidity and mortality in people with SARDs,” said Dr. Bernstein, who is co-first author of the guidelines. “People with systemic sclerosis, rheumatoid arthritis, idiopathic inflammatory myopathies, mixed connective tissue disease, and Sjögren’s disease are at greatest risk of developing ILD.”
Pediatric patients with SARDs excluded
The guidelines’ population of interest was people 17 years of age and older who were diagnosed with SARDs with a high risk of ILD. Pediatric patients with SARDs were excluded from the endeavor, as were those with systemic lupus erythematosus, antineutrophil cytoplasmic antibody–associated vasculitis, sarcoidosis, ankylosing spondylitis, undifferentiated connective tissue disease, interstitial pneumonia with autoimmune features, and those with unclassifiable ILD.
In the realm of screening, the guideline authors conditionally recommend two screening tests for patients considered at increased risk of ILD: pulmonary function tests and high-resolution chest CT (HRCT). Pulmonary function tests should include spirometry, lung volumes, and diffusion capacity. “Office spirometry alone is insufficient,” said Dr. Johnson, who served as lead author of the guidelines. And while a HRCT scan is recommended, “some patients may present to the emergency room with acute onset shortness of breath, and they may receive a CT angiogram to screen for pulmonary embolism,” she said. “It’s important to note that CT angiograms are performed in incomplete inspiration to maximize pulmonary artery enhancement. This may produce atelectasis that may obscure or mimic ILD. As a result, CTA studies are often inadequate to screen for ILD.”
Once a patient is diagnosed with ILD, three tests are recommended for monitoring: pulmonary function testing (every 3-6 months the first year in patients with IIM and SSc, then less frequently once stable, and every 3-12 months in the first year in patients with RA, SjD, and MCTD, then less frequently once stable); ambulatory desaturation testing every 3-12 months; and HRCT as needed. Dr. Johnson noted that while that the screening of ILD lies within the realm of rheumatologists, “once a patient is diagnosed, we are encouraged to comanage these patients with pulmonologists,” she said. “Ambulatory desaturation testing is not an infrequent test in the hands of pulmonologists. This is where co-management can be helpful.” She characterized a 6-minute walk test with continuous oximetry as “insufficient and is not synonymous with ambulatory desaturation testing. Ambulatory desaturation testing includes up titration of oxygen if a patient desaturates.”
The guidelines conditionally recommend against using chest radiography, 6-minute walk test distance, ambulatory desaturation testing, and bronchoscopy for ILD screening, and there is a strong recommendation against surgical lung biopsy. “However, there are unique circumstances where these tests may be considered,” Dr. Johnson said. “For example, ambulatory desaturation testing may be helpful if a patient is unable to perform a pulmonary function test. Bronchoscopy may be used to rule out infection, sarcoidosis, lymphoma, or alveolar hemorrhage, and surgical lung biopsy may be considered if you’re trying to rule out a malignancy.”
Similarly, several tests are conditionally recommended against for the monitoring of ILD, including chest radiography, the 6-minute walk test distance, and bronchoscopy. “But there are unique circumstances where they may be considered,” she said. “The 6-minute walk test may be used if a patient is unable to perform a pulmonary function test or if they’re being assessed for lung transplantation. Bronchoscopy may be used to rule out infection or alveolar hemorrhage.”
Preferred treatment options described
First-line treatment recommendations for ILD were based on the best available published evidence, voting panel expertise, and patient preferences. For SSc, the preferred treatment options include mycophenolate (CellCept), tocilizumab (Actemra), or rituximab (Rituxan and biosimilars), while additional options include cyclophosphamide, nintedanib (Ofev), and azathioprine. For myositis, the preferred treatment options include mycophenolate, azathioprine, rituximab, or calcineurin inhibitors, while additional options include a Janus kinase (JAK) inhibitor or cyclophosphamide. For MCTD, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include tocilizumab or cyclophosphamide. For RA and Sjögren’s, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include cyclophosphamide. Dr. Johnson emphasized that there was low certainty evidence to recommend one treatment over another. “Many situations might lead a provider to choose a different option for ILD treatment, such as the presence of comorbidities or extra-pulmonary disease,” she said. “So, while our guidelines were focused on effectiveness for ILD, providers may choose therapies that will help ILD and other disease manifestations.”
The guidelines conditionally recommend a short course of glucocorticoids as a bridging therapy or for treatment of a flare of ILD in patients with myositis, MCTD, RA, and Sjögren’s. The panel strongly recommends against the use of glucocorticoids in patients with SSc due to the concern for inducing a scleroderma renal crisis. “While this may be common knowledge for rheumatologists, it may not be common knowledge for pulmonologists,” she said. “So here is an opportunity to educate our pulmonology colleagues in our consultation notes.”
The guidelines also include recommendations for progression of ILD, which was defined using the INBUILD trial criteria. Mycophenolate is conditionally recommended to be the first ILD treatment for all SARDs when progression occurs, if it wasn’t the first ILD treatment used. “If it was, then other medications that rheumatologists are used to can be considered as the next ILD treatment in the face of progression: rituximab, nintedanib, tocilizumab, and cyclophosphamide,” she said. The guidelines include a conditional recommendation against long-term glucocorticoid use in myositis, MCTD, RA, and Sjögren’s, plus a strong recommendation against long-term glucocorticoid use in SSc. Finally, there is a conditional recommendation of referral for lung transplant evaluation at the appropriate time at experienced centers.
Another group of recommendations has to do with cases of rapidly progressive ILD, which is characterized by rapid progression from no oxygen or a patient’s baseline oxygen requirement to a high oxygen requirement or intubation usually within days to weeks without a documented cause, such as infection or heart failure. “In cases of rapidly progressive ILD, which typically occurs in the setting of anti-MDA5 antibodies, there is a conditional recommendation for IV glucocorticoids plus two additional therapies: traditionally rituximab and mycophenolate,” Dr. Johnson said. “However, what may be new to some clinicians is combination IVIG [intravenous immunoglobulin] and a calcineurin inhibitor, notably tacrolimus,” she said. “This is the situation where experience at expert centers is influencing our guidelines in advance of data.”
A patient panel provided input
For the undertaking, a core team that included six rheumatologists; one pulmonologist; one thoracic radiologist; one expert on the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology; and two literature review experts developed clinically relevant population, intervention, comparator, and outcomes (PICO) questions. The literature review team included 13 rheumatologists, 8 pulmonologists, and 3 methodologists. Finally, a 21-member patient panel was convened to share their values and preferences regarding screening, monitoring, and treatment of SARD-related ILD. Of these, Dr. Bernstein said that 4 were at risk for ILD and 17 had been diagnosed with ILD. Next, the literature review team conducted a systematic review and used the GRADE methodology to rate the available evidence as high, moderate, low, or very low. Then, a voting panel comprising 13 rheumatologists, 10 pulmonologists, 1 radiologist, and 3 patients from the patient panel cast votes for each PICO question and made final recommendations.
The review of evidence left the guidelines authors with 241 PICO questions, “which is a lot,” Dr. Bernstein said. “To put this in perspective, some guidelines address only 10 or 15 PICO questions. Fortunately, we had a dedicated group of experts who were up to the challenge.” Dr. Johnson emphasized that the forthcoming guidelines should not be used by insurers to mandate a specific order of prescribing. “Clinicians must retain the latitude to prescribe medications based on individual patient factors and preferences,” she said.
Dr. Bernstein disclosed that she is an adviser to, a consultant for, and has received grant or research support from Boehringer Ingelheim and has also received grant or research support from Kadmon and Pfizer. Dr. Johnson disclosed that she has received research support from the American College of Rheumatology to develop these guidelines. She has also been an investigator for trials sponsored by Bristol-Myers Squibb, Roche, and Boehringer Ingelheim and has mitigated these relevant conflicts of interest 1 year prior to the development of these guidelines, and will continue to do so for the foreseeable future.
SAN DIEGO – In the spring of 2024, the American College of Rheumatology is expected to release guidelines to help inform the screening, monitoring, and treatment of interstitial lung disease (ILD) in people with systemic autoimmune rheumatic diseases (SARDs).
The guidelines, which were previewed during a session at the ACR’s annual meeting, will include 50 recommendations, 3 of which met criteria for a strong rating:
- For people with SARDs at increased risk of developing ILD, the authors strongly recommend against screening with surgical lung biopsy.
- For people with systemic sclerosis (SSc)-related ILD, the authors strongly recommend against glucocorticoids as a first-line ILD treatment.
- For people with SSc-related ILD progression despite an initial ILD treatment, the authors strongly recommend against using long-term glucocorticoids.
Elana J. Bernstein, MD, MSc, a rheumatologist who directs the Columbia/New York-Presbyterian Scleroderma Center, and Sindhu R. Johnson, MD, a rheumatologist who directs the Toronto Scleroderma Program at the University of Toronto, provided a sneak peek of the recommendations to attendees before anticipated publication in Arthritis & Rheumatology and Arthritis Care & Research. For now, guideline summaries for screening and monitoring and treatment are currently available, and three manuscripts are under peer review: one about screening and monitoring, one about treatment, and one about the patient panel that participated in the effort.
“ILD is a significant cause of morbidity and mortality in people with SARDs,” said Dr. Bernstein, who is co-first author of the guidelines. “People with systemic sclerosis, rheumatoid arthritis, idiopathic inflammatory myopathies, mixed connective tissue disease, and Sjögren’s disease are at greatest risk of developing ILD.”
Pediatric patients with SARDs excluded
The guidelines’ population of interest was people 17 years of age and older who were diagnosed with SARDs with a high risk of ILD. Pediatric patients with SARDs were excluded from the endeavor, as were those with systemic lupus erythematosus, antineutrophil cytoplasmic antibody–associated vasculitis, sarcoidosis, ankylosing spondylitis, undifferentiated connective tissue disease, interstitial pneumonia with autoimmune features, and those with unclassifiable ILD.
In the realm of screening, the guideline authors conditionally recommend two screening tests for patients considered at increased risk of ILD: pulmonary function tests and high-resolution chest CT (HRCT). Pulmonary function tests should include spirometry, lung volumes, and diffusion capacity. “Office spirometry alone is insufficient,” said Dr. Johnson, who served as lead author of the guidelines. And while a HRCT scan is recommended, “some patients may present to the emergency room with acute onset shortness of breath, and they may receive a CT angiogram to screen for pulmonary embolism,” she said. “It’s important to note that CT angiograms are performed in incomplete inspiration to maximize pulmonary artery enhancement. This may produce atelectasis that may obscure or mimic ILD. As a result, CTA studies are often inadequate to screen for ILD.”
Once a patient is diagnosed with ILD, three tests are recommended for monitoring: pulmonary function testing (every 3-6 months the first year in patients with IIM and SSc, then less frequently once stable, and every 3-12 months in the first year in patients with RA, SjD, and MCTD, then less frequently once stable); ambulatory desaturation testing every 3-12 months; and HRCT as needed. Dr. Johnson noted that while that the screening of ILD lies within the realm of rheumatologists, “once a patient is diagnosed, we are encouraged to comanage these patients with pulmonologists,” she said. “Ambulatory desaturation testing is not an infrequent test in the hands of pulmonologists. This is where co-management can be helpful.” She characterized a 6-minute walk test with continuous oximetry as “insufficient and is not synonymous with ambulatory desaturation testing. Ambulatory desaturation testing includes up titration of oxygen if a patient desaturates.”
The guidelines conditionally recommend against using chest radiography, 6-minute walk test distance, ambulatory desaturation testing, and bronchoscopy for ILD screening, and there is a strong recommendation against surgical lung biopsy. “However, there are unique circumstances where these tests may be considered,” Dr. Johnson said. “For example, ambulatory desaturation testing may be helpful if a patient is unable to perform a pulmonary function test. Bronchoscopy may be used to rule out infection, sarcoidosis, lymphoma, or alveolar hemorrhage, and surgical lung biopsy may be considered if you’re trying to rule out a malignancy.”
Similarly, several tests are conditionally recommended against for the monitoring of ILD, including chest radiography, the 6-minute walk test distance, and bronchoscopy. “But there are unique circumstances where they may be considered,” she said. “The 6-minute walk test may be used if a patient is unable to perform a pulmonary function test or if they’re being assessed for lung transplantation. Bronchoscopy may be used to rule out infection or alveolar hemorrhage.”
Preferred treatment options described
First-line treatment recommendations for ILD were based on the best available published evidence, voting panel expertise, and patient preferences. For SSc, the preferred treatment options include mycophenolate (CellCept), tocilizumab (Actemra), or rituximab (Rituxan and biosimilars), while additional options include cyclophosphamide, nintedanib (Ofev), and azathioprine. For myositis, the preferred treatment options include mycophenolate, azathioprine, rituximab, or calcineurin inhibitors, while additional options include a Janus kinase (JAK) inhibitor or cyclophosphamide. For MCTD, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include tocilizumab or cyclophosphamide. For RA and Sjögren’s, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include cyclophosphamide. Dr. Johnson emphasized that there was low certainty evidence to recommend one treatment over another. “Many situations might lead a provider to choose a different option for ILD treatment, such as the presence of comorbidities or extra-pulmonary disease,” she said. “So, while our guidelines were focused on effectiveness for ILD, providers may choose therapies that will help ILD and other disease manifestations.”
The guidelines conditionally recommend a short course of glucocorticoids as a bridging therapy or for treatment of a flare of ILD in patients with myositis, MCTD, RA, and Sjögren’s. The panel strongly recommends against the use of glucocorticoids in patients with SSc due to the concern for inducing a scleroderma renal crisis. “While this may be common knowledge for rheumatologists, it may not be common knowledge for pulmonologists,” she said. “So here is an opportunity to educate our pulmonology colleagues in our consultation notes.”
The guidelines also include recommendations for progression of ILD, which was defined using the INBUILD trial criteria. Mycophenolate is conditionally recommended to be the first ILD treatment for all SARDs when progression occurs, if it wasn’t the first ILD treatment used. “If it was, then other medications that rheumatologists are used to can be considered as the next ILD treatment in the face of progression: rituximab, nintedanib, tocilizumab, and cyclophosphamide,” she said. The guidelines include a conditional recommendation against long-term glucocorticoid use in myositis, MCTD, RA, and Sjögren’s, plus a strong recommendation against long-term glucocorticoid use in SSc. Finally, there is a conditional recommendation of referral for lung transplant evaluation at the appropriate time at experienced centers.
Another group of recommendations has to do with cases of rapidly progressive ILD, which is characterized by rapid progression from no oxygen or a patient’s baseline oxygen requirement to a high oxygen requirement or intubation usually within days to weeks without a documented cause, such as infection or heart failure. “In cases of rapidly progressive ILD, which typically occurs in the setting of anti-MDA5 antibodies, there is a conditional recommendation for IV glucocorticoids plus two additional therapies: traditionally rituximab and mycophenolate,” Dr. Johnson said. “However, what may be new to some clinicians is combination IVIG [intravenous immunoglobulin] and a calcineurin inhibitor, notably tacrolimus,” she said. “This is the situation where experience at expert centers is influencing our guidelines in advance of data.”
A patient panel provided input
For the undertaking, a core team that included six rheumatologists; one pulmonologist; one thoracic radiologist; one expert on the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology; and two literature review experts developed clinically relevant population, intervention, comparator, and outcomes (PICO) questions. The literature review team included 13 rheumatologists, 8 pulmonologists, and 3 methodologists. Finally, a 21-member patient panel was convened to share their values and preferences regarding screening, monitoring, and treatment of SARD-related ILD. Of these, Dr. Bernstein said that 4 were at risk for ILD and 17 had been diagnosed with ILD. Next, the literature review team conducted a systematic review and used the GRADE methodology to rate the available evidence as high, moderate, low, or very low. Then, a voting panel comprising 13 rheumatologists, 10 pulmonologists, 1 radiologist, and 3 patients from the patient panel cast votes for each PICO question and made final recommendations.
The review of evidence left the guidelines authors with 241 PICO questions, “which is a lot,” Dr. Bernstein said. “To put this in perspective, some guidelines address only 10 or 15 PICO questions. Fortunately, we had a dedicated group of experts who were up to the challenge.” Dr. Johnson emphasized that the forthcoming guidelines should not be used by insurers to mandate a specific order of prescribing. “Clinicians must retain the latitude to prescribe medications based on individual patient factors and preferences,” she said.
Dr. Bernstein disclosed that she is an adviser to, a consultant for, and has received grant or research support from Boehringer Ingelheim and has also received grant or research support from Kadmon and Pfizer. Dr. Johnson disclosed that she has received research support from the American College of Rheumatology to develop these guidelines. She has also been an investigator for trials sponsored by Bristol-Myers Squibb, Roche, and Boehringer Ingelheim and has mitigated these relevant conflicts of interest 1 year prior to the development of these guidelines, and will continue to do so for the foreseeable future.
SAN DIEGO – In the spring of 2024, the American College of Rheumatology is expected to release guidelines to help inform the screening, monitoring, and treatment of interstitial lung disease (ILD) in people with systemic autoimmune rheumatic diseases (SARDs).
The guidelines, which were previewed during a session at the ACR’s annual meeting, will include 50 recommendations, 3 of which met criteria for a strong rating:
- For people with SARDs at increased risk of developing ILD, the authors strongly recommend against screening with surgical lung biopsy.
- For people with systemic sclerosis (SSc)-related ILD, the authors strongly recommend against glucocorticoids as a first-line ILD treatment.
- For people with SSc-related ILD progression despite an initial ILD treatment, the authors strongly recommend against using long-term glucocorticoids.
Elana J. Bernstein, MD, MSc, a rheumatologist who directs the Columbia/New York-Presbyterian Scleroderma Center, and Sindhu R. Johnson, MD, a rheumatologist who directs the Toronto Scleroderma Program at the University of Toronto, provided a sneak peek of the recommendations to attendees before anticipated publication in Arthritis & Rheumatology and Arthritis Care & Research. For now, guideline summaries for screening and monitoring and treatment are currently available, and three manuscripts are under peer review: one about screening and monitoring, one about treatment, and one about the patient panel that participated in the effort.
“ILD is a significant cause of morbidity and mortality in people with SARDs,” said Dr. Bernstein, who is co-first author of the guidelines. “People with systemic sclerosis, rheumatoid arthritis, idiopathic inflammatory myopathies, mixed connective tissue disease, and Sjögren’s disease are at greatest risk of developing ILD.”
Pediatric patients with SARDs excluded
The guidelines’ population of interest was people 17 years of age and older who were diagnosed with SARDs with a high risk of ILD. Pediatric patients with SARDs were excluded from the endeavor, as were those with systemic lupus erythematosus, antineutrophil cytoplasmic antibody–associated vasculitis, sarcoidosis, ankylosing spondylitis, undifferentiated connective tissue disease, interstitial pneumonia with autoimmune features, and those with unclassifiable ILD.
In the realm of screening, the guideline authors conditionally recommend two screening tests for patients considered at increased risk of ILD: pulmonary function tests and high-resolution chest CT (HRCT). Pulmonary function tests should include spirometry, lung volumes, and diffusion capacity. “Office spirometry alone is insufficient,” said Dr. Johnson, who served as lead author of the guidelines. And while a HRCT scan is recommended, “some patients may present to the emergency room with acute onset shortness of breath, and they may receive a CT angiogram to screen for pulmonary embolism,” she said. “It’s important to note that CT angiograms are performed in incomplete inspiration to maximize pulmonary artery enhancement. This may produce atelectasis that may obscure or mimic ILD. As a result, CTA studies are often inadequate to screen for ILD.”
Once a patient is diagnosed with ILD, three tests are recommended for monitoring: pulmonary function testing (every 3-6 months the first year in patients with IIM and SSc, then less frequently once stable, and every 3-12 months in the first year in patients with RA, SjD, and MCTD, then less frequently once stable); ambulatory desaturation testing every 3-12 months; and HRCT as needed. Dr. Johnson noted that while that the screening of ILD lies within the realm of rheumatologists, “once a patient is diagnosed, we are encouraged to comanage these patients with pulmonologists,” she said. “Ambulatory desaturation testing is not an infrequent test in the hands of pulmonologists. This is where co-management can be helpful.” She characterized a 6-minute walk test with continuous oximetry as “insufficient and is not synonymous with ambulatory desaturation testing. Ambulatory desaturation testing includes up titration of oxygen if a patient desaturates.”
The guidelines conditionally recommend against using chest radiography, 6-minute walk test distance, ambulatory desaturation testing, and bronchoscopy for ILD screening, and there is a strong recommendation against surgical lung biopsy. “However, there are unique circumstances where these tests may be considered,” Dr. Johnson said. “For example, ambulatory desaturation testing may be helpful if a patient is unable to perform a pulmonary function test. Bronchoscopy may be used to rule out infection, sarcoidosis, lymphoma, or alveolar hemorrhage, and surgical lung biopsy may be considered if you’re trying to rule out a malignancy.”
Similarly, several tests are conditionally recommended against for the monitoring of ILD, including chest radiography, the 6-minute walk test distance, and bronchoscopy. “But there are unique circumstances where they may be considered,” she said. “The 6-minute walk test may be used if a patient is unable to perform a pulmonary function test or if they’re being assessed for lung transplantation. Bronchoscopy may be used to rule out infection or alveolar hemorrhage.”
Preferred treatment options described
First-line treatment recommendations for ILD were based on the best available published evidence, voting panel expertise, and patient preferences. For SSc, the preferred treatment options include mycophenolate (CellCept), tocilizumab (Actemra), or rituximab (Rituxan and biosimilars), while additional options include cyclophosphamide, nintedanib (Ofev), and azathioprine. For myositis, the preferred treatment options include mycophenolate, azathioprine, rituximab, or calcineurin inhibitors, while additional options include a Janus kinase (JAK) inhibitor or cyclophosphamide. For MCTD, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include tocilizumab or cyclophosphamide. For RA and Sjögren’s, the preferred treatment options include mycophenolate, azathioprine, or rituximab, while additional options include cyclophosphamide. Dr. Johnson emphasized that there was low certainty evidence to recommend one treatment over another. “Many situations might lead a provider to choose a different option for ILD treatment, such as the presence of comorbidities or extra-pulmonary disease,” she said. “So, while our guidelines were focused on effectiveness for ILD, providers may choose therapies that will help ILD and other disease manifestations.”
The guidelines conditionally recommend a short course of glucocorticoids as a bridging therapy or for treatment of a flare of ILD in patients with myositis, MCTD, RA, and Sjögren’s. The panel strongly recommends against the use of glucocorticoids in patients with SSc due to the concern for inducing a scleroderma renal crisis. “While this may be common knowledge for rheumatologists, it may not be common knowledge for pulmonologists,” she said. “So here is an opportunity to educate our pulmonology colleagues in our consultation notes.”
The guidelines also include recommendations for progression of ILD, which was defined using the INBUILD trial criteria. Mycophenolate is conditionally recommended to be the first ILD treatment for all SARDs when progression occurs, if it wasn’t the first ILD treatment used. “If it was, then other medications that rheumatologists are used to can be considered as the next ILD treatment in the face of progression: rituximab, nintedanib, tocilizumab, and cyclophosphamide,” she said. The guidelines include a conditional recommendation against long-term glucocorticoid use in myositis, MCTD, RA, and Sjögren’s, plus a strong recommendation against long-term glucocorticoid use in SSc. Finally, there is a conditional recommendation of referral for lung transplant evaluation at the appropriate time at experienced centers.
Another group of recommendations has to do with cases of rapidly progressive ILD, which is characterized by rapid progression from no oxygen or a patient’s baseline oxygen requirement to a high oxygen requirement or intubation usually within days to weeks without a documented cause, such as infection or heart failure. “In cases of rapidly progressive ILD, which typically occurs in the setting of anti-MDA5 antibodies, there is a conditional recommendation for IV glucocorticoids plus two additional therapies: traditionally rituximab and mycophenolate,” Dr. Johnson said. “However, what may be new to some clinicians is combination IVIG [intravenous immunoglobulin] and a calcineurin inhibitor, notably tacrolimus,” she said. “This is the situation where experience at expert centers is influencing our guidelines in advance of data.”
A patient panel provided input
For the undertaking, a core team that included six rheumatologists; one pulmonologist; one thoracic radiologist; one expert on the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology; and two literature review experts developed clinically relevant population, intervention, comparator, and outcomes (PICO) questions. The literature review team included 13 rheumatologists, 8 pulmonologists, and 3 methodologists. Finally, a 21-member patient panel was convened to share their values and preferences regarding screening, monitoring, and treatment of SARD-related ILD. Of these, Dr. Bernstein said that 4 were at risk for ILD and 17 had been diagnosed with ILD. Next, the literature review team conducted a systematic review and used the GRADE methodology to rate the available evidence as high, moderate, low, or very low. Then, a voting panel comprising 13 rheumatologists, 10 pulmonologists, 1 radiologist, and 3 patients from the patient panel cast votes for each PICO question and made final recommendations.
The review of evidence left the guidelines authors with 241 PICO questions, “which is a lot,” Dr. Bernstein said. “To put this in perspective, some guidelines address only 10 or 15 PICO questions. Fortunately, we had a dedicated group of experts who were up to the challenge.” Dr. Johnson emphasized that the forthcoming guidelines should not be used by insurers to mandate a specific order of prescribing. “Clinicians must retain the latitude to prescribe medications based on individual patient factors and preferences,” she said.
Dr. Bernstein disclosed that she is an adviser to, a consultant for, and has received grant or research support from Boehringer Ingelheim and has also received grant or research support from Kadmon and Pfizer. Dr. Johnson disclosed that she has received research support from the American College of Rheumatology to develop these guidelines. She has also been an investigator for trials sponsored by Bristol-Myers Squibb, Roche, and Boehringer Ingelheim and has mitigated these relevant conflicts of interest 1 year prior to the development of these guidelines, and will continue to do so for the foreseeable future.
AT ACR 2023
Benralizumab proves noninferior to mepolizumab for rare vasculitis, EGPA
SAN DIEGO – Treatment with benralizumab (Fasenra) achieved remission at 36 and 48 weeks at rates similar to those of mepolizumab (Nucala) in a head-to-head phase 3 trial of the two drugs for patients with a relapsing or refractory case of the rare vasculitis eosinophilic granulomatosis with polyangiitis (EGPA).
Benralizumab, a monoclonal antibody from AstraZeneca that binds to the alpha chain of the interleukin (IL)-5 receptor, is indicated as an add-on maintenance treatment for patients 12 years and older with severe eosinophilic asthma but is not currently approved for EGPA. Mepolizumab is a humanized monoclonal antibody targeting IL-5 and the only approved drug for EGPA.
Peter A. Merkel, MD, presented the trial, known as MANDARA, during a late-breaking poster session at the annual meeting of the American College of Rheumatology. A total of 140 patients with EGPA received either subcutaneous benralizumab 30 mg or mepolizumab 300 mg every 4 weeks for 52 weeks. The trial, which began recruitment in late 2019, was limited to patients at least 18 years of age with relapsing/refractory EGPA that required stable use of oral glucocorticoids (OGCs) and immunosuppressive therapy for at least 4 weeks prior to randomization, and the primary endpoint was the proportion of patients who achieved remission at weeks 36 and 48. Remission was defined as a Birmingham Vasculitis Activity Score (BVAS) of 0 plus an OGC dose of no more than 4 mg/day. Secondary endpoints included rates of accrued and maintained remission, OGC use, clinical benefit and complete response, blood eosinophil counts, total BVAS, and Vascular Damage Index scores. The mean age of the 140 patients was 52 years, and 60% were women.
Dr. Merkel and colleagues reported that the adjusted remission rate at both weeks 36 and 48 was 59.2% for those in the benralizumab arm and 56.5% for those in the mepolizumab arm (P = .7278). The percentage of patients who achieved a BVAS of 0 was similar between the two arms (83% in the benralizumab arm vs. 84.2% for those in the mepolizumab arm; P = .8502), as was the percentage of patients on an OGC dose of up to 4 mg/day (62.1% vs. 57.9%; P = .5942). At 48-52 weeks, 86.1% of patients in the benralizumab arm achieved up to a 50% reduction in OGC use, compared with 73.9% of those in the mepolizumab arm (P = .0611), and 41.4% of patients in the benralizumab arm achieved a 100% reduction in OGC use, compared with 25.8% of those in the mepolizumab arm (P = .0406).
In findings related to safety, the top three adverse events were COVID-19 (21.4% in the benralizumab arm vs. 27.1% in the mepolizumab arm, respectively), headache (17.1% vs. 15.7%), and arthralgia (17.1% vs. 11.4%).
“We were pleased with the findings because they met our expectations,” Dr. Merkel, chief of rheumatology and professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, said in an interview. “The hypothesis was that these two drugs would be equivalent and safe. The implication for patients is that they’ll have another treatment option for EGPA, which is an underrecognized disease with need for more effective treatments. I anticipate that the drug will be approved for use in EGPA, providing another option for treating this complicated multisystem eosinophilic-associated disease. Having more options for our biologic therapies is good.”
He characterized the retention of patients in MANDARA as “remarkable, despite the COVID-19 pandemic. Patients with rare diseases are quite dedicated to helping conduct research. They know that their disease is not common and that they could help others.”
The study was sponsored and funded by AstraZeneca. Dr. Merkel disclosed that he has received consulting fees and research support from many pharmaceutical companies, including AstraZeneca.
SAN DIEGO – Treatment with benralizumab (Fasenra) achieved remission at 36 and 48 weeks at rates similar to those of mepolizumab (Nucala) in a head-to-head phase 3 trial of the two drugs for patients with a relapsing or refractory case of the rare vasculitis eosinophilic granulomatosis with polyangiitis (EGPA).
Benralizumab, a monoclonal antibody from AstraZeneca that binds to the alpha chain of the interleukin (IL)-5 receptor, is indicated as an add-on maintenance treatment for patients 12 years and older with severe eosinophilic asthma but is not currently approved for EGPA. Mepolizumab is a humanized monoclonal antibody targeting IL-5 and the only approved drug for EGPA.
Peter A. Merkel, MD, presented the trial, known as MANDARA, during a late-breaking poster session at the annual meeting of the American College of Rheumatology. A total of 140 patients with EGPA received either subcutaneous benralizumab 30 mg or mepolizumab 300 mg every 4 weeks for 52 weeks. The trial, which began recruitment in late 2019, was limited to patients at least 18 years of age with relapsing/refractory EGPA that required stable use of oral glucocorticoids (OGCs) and immunosuppressive therapy for at least 4 weeks prior to randomization, and the primary endpoint was the proportion of patients who achieved remission at weeks 36 and 48. Remission was defined as a Birmingham Vasculitis Activity Score (BVAS) of 0 plus an OGC dose of no more than 4 mg/day. Secondary endpoints included rates of accrued and maintained remission, OGC use, clinical benefit and complete response, blood eosinophil counts, total BVAS, and Vascular Damage Index scores. The mean age of the 140 patients was 52 years, and 60% were women.
Dr. Merkel and colleagues reported that the adjusted remission rate at both weeks 36 and 48 was 59.2% for those in the benralizumab arm and 56.5% for those in the mepolizumab arm (P = .7278). The percentage of patients who achieved a BVAS of 0 was similar between the two arms (83% in the benralizumab arm vs. 84.2% for those in the mepolizumab arm; P = .8502), as was the percentage of patients on an OGC dose of up to 4 mg/day (62.1% vs. 57.9%; P = .5942). At 48-52 weeks, 86.1% of patients in the benralizumab arm achieved up to a 50% reduction in OGC use, compared with 73.9% of those in the mepolizumab arm (P = .0611), and 41.4% of patients in the benralizumab arm achieved a 100% reduction in OGC use, compared with 25.8% of those in the mepolizumab arm (P = .0406).
In findings related to safety, the top three adverse events were COVID-19 (21.4% in the benralizumab arm vs. 27.1% in the mepolizumab arm, respectively), headache (17.1% vs. 15.7%), and arthralgia (17.1% vs. 11.4%).
“We were pleased with the findings because they met our expectations,” Dr. Merkel, chief of rheumatology and professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, said in an interview. “The hypothesis was that these two drugs would be equivalent and safe. The implication for patients is that they’ll have another treatment option for EGPA, which is an underrecognized disease with need for more effective treatments. I anticipate that the drug will be approved for use in EGPA, providing another option for treating this complicated multisystem eosinophilic-associated disease. Having more options for our biologic therapies is good.”
He characterized the retention of patients in MANDARA as “remarkable, despite the COVID-19 pandemic. Patients with rare diseases are quite dedicated to helping conduct research. They know that their disease is not common and that they could help others.”
The study was sponsored and funded by AstraZeneca. Dr. Merkel disclosed that he has received consulting fees and research support from many pharmaceutical companies, including AstraZeneca.
SAN DIEGO – Treatment with benralizumab (Fasenra) achieved remission at 36 and 48 weeks at rates similar to those of mepolizumab (Nucala) in a head-to-head phase 3 trial of the two drugs for patients with a relapsing or refractory case of the rare vasculitis eosinophilic granulomatosis with polyangiitis (EGPA).
Benralizumab, a monoclonal antibody from AstraZeneca that binds to the alpha chain of the interleukin (IL)-5 receptor, is indicated as an add-on maintenance treatment for patients 12 years and older with severe eosinophilic asthma but is not currently approved for EGPA. Mepolizumab is a humanized monoclonal antibody targeting IL-5 and the only approved drug for EGPA.
Peter A. Merkel, MD, presented the trial, known as MANDARA, during a late-breaking poster session at the annual meeting of the American College of Rheumatology. A total of 140 patients with EGPA received either subcutaneous benralizumab 30 mg or mepolizumab 300 mg every 4 weeks for 52 weeks. The trial, which began recruitment in late 2019, was limited to patients at least 18 years of age with relapsing/refractory EGPA that required stable use of oral glucocorticoids (OGCs) and immunosuppressive therapy for at least 4 weeks prior to randomization, and the primary endpoint was the proportion of patients who achieved remission at weeks 36 and 48. Remission was defined as a Birmingham Vasculitis Activity Score (BVAS) of 0 plus an OGC dose of no more than 4 mg/day. Secondary endpoints included rates of accrued and maintained remission, OGC use, clinical benefit and complete response, blood eosinophil counts, total BVAS, and Vascular Damage Index scores. The mean age of the 140 patients was 52 years, and 60% were women.
Dr. Merkel and colleagues reported that the adjusted remission rate at both weeks 36 and 48 was 59.2% for those in the benralizumab arm and 56.5% for those in the mepolizumab arm (P = .7278). The percentage of patients who achieved a BVAS of 0 was similar between the two arms (83% in the benralizumab arm vs. 84.2% for those in the mepolizumab arm; P = .8502), as was the percentage of patients on an OGC dose of up to 4 mg/day (62.1% vs. 57.9%; P = .5942). At 48-52 weeks, 86.1% of patients in the benralizumab arm achieved up to a 50% reduction in OGC use, compared with 73.9% of those in the mepolizumab arm (P = .0611), and 41.4% of patients in the benralizumab arm achieved a 100% reduction in OGC use, compared with 25.8% of those in the mepolizumab arm (P = .0406).
In findings related to safety, the top three adverse events were COVID-19 (21.4% in the benralizumab arm vs. 27.1% in the mepolizumab arm, respectively), headache (17.1% vs. 15.7%), and arthralgia (17.1% vs. 11.4%).
“We were pleased with the findings because they met our expectations,” Dr. Merkel, chief of rheumatology and professor of medicine and epidemiology at the University of Pennsylvania, Philadelphia, said in an interview. “The hypothesis was that these two drugs would be equivalent and safe. The implication for patients is that they’ll have another treatment option for EGPA, which is an underrecognized disease with need for more effective treatments. I anticipate that the drug will be approved for use in EGPA, providing another option for treating this complicated multisystem eosinophilic-associated disease. Having more options for our biologic therapies is good.”
He characterized the retention of patients in MANDARA as “remarkable, despite the COVID-19 pandemic. Patients with rare diseases are quite dedicated to helping conduct research. They know that their disease is not common and that they could help others.”
The study was sponsored and funded by AstraZeneca. Dr. Merkel disclosed that he has received consulting fees and research support from many pharmaceutical companies, including AstraZeneca.
AT ACR 2023
Vasculitis confers higher risk for adverse pregnancy outcomes
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
AT ACR 2023
Caution raised on reduced-dose steroids in rare vasculitides GPA, MPA
SAN DIEGO – A real-world analysis linked the PEXIVAS reduced-dose glucocorticoid (GC) regimen to a higher likelihood of a group of poor outcomes such as death in patients with severe granulomatosis with polyangiitis (GPA) or microscopic polyangiitis (MPA).
The retrospective observational study, presented at the annual meeting of the American College of Rheumatology, aimed to see whether results from the landmark PEXIVAS trial would hold up in a real-world analysis given some important limitations of the PEXIVAS’s primary outcome and its relative lack of balance in choice of induction agents.
First author of the new study, Sophie Nagle, MD, of Cochin Hospital in Paris, and colleagues in the French Vasculitis Study Group noted that PEXIVAS “demonstrated noninferiority of reduced-dose GC regimen compared to standard dose for the incidence of death or end-stage kidney disease (ESKD), with a significant reduction in serious infections at 1 year. However, the primary endpoint did not include disease progression or relapse, the majority of patients received cyclophosphamide as induction therapy, and subgroup analysis showed a trend towards an increased risk of death or ESKD in [rituximab]-treated patients.”
The new findings “give us pause about using low-dose glucocorticoid regimens” and suggest that rheumatologists might be a little more conservative about their use than randomized controlled trials such as PEXIVAS might suggest, said Vanderbilt University vasculitis specialist Kevin W. Byram, MD, who’s familiar with the findings but did not take part in the study.
Dr. Nagle reported that among 234 patients with either GPA or MPA, 33.3% of 126 who received a reduced-dose GC regimen experienced one of the 12-month composite primary outcome’s events of death, disease relapse, ESKD, or disease progression before remission that required treatment modification, compared with 18.5% of 108 who received the standard GC regimen (P = .016).
In a propensity score analysis, the higher risk of poor outcomes in the reduced-dose group remained (hazard ratio, 1.57; 95% confidence interval, 0.93-2.64). A multivariate analysis also identified a higher risk for the composite primary outcome in the reduced-dose group (HR, 1.72; 95% CI, 1.08-2.74), although there was no association with an increased risk of death or ESKD.
The PEXIVAS study, published in 2020, supported lower GC doses in antineutrophil cytoplasmic antibody–associated vasculitis, potentially revolutionizing treatment. “Historically, we have used high doses of glucocorticoids on slower tapers to treat this disease, which itself is a strategy that leads to potential complications,” Dr. Byram said. “PEXIVAS suggested we could potentially use less glucocorticoids in these patients.”
For the retrospective, multicenter study, researchers tracked patients from 2018 to 2022, all aged 15 and above. They included 93 with MPA and 141 with GPA. Nearly half were female, and they had a mean age of 61 years. The patients had severe flare-ups treated with rituximab or cyclophosphamide induction and reduced-dose or standard GC regimen.
The standard care and reduced-dose groups were similar, Dr. Nagle said, although the standard group had significantly more patients with GPA (71% vs. 29% with MPA) than did the low-dose group (51% with GPA, 49% with MPA).
The researchers reported that in a reduced-dose subgroup, patients with creatinine levels above 300 micromol/L were more likely to meet the primary endpoint (relative risk, 2.14; 95% CI, 1.14-4.03). Those treated with the reduced-dose GC regimen were also more likely to reach the primary endpoint (HR, 1.61; 95% CI, 0.94-2.77) and die or develop ESKD (HR, 2.42; 95% CI, 1.04-5.66).
However, adverse events at 12 months were similar in both groups: The authors noted that those who received the reduced-dose GC regimen didn’t have higher risk of death, ESKD, or severe infections.
The authors highlighted that “increased vigilance is required when using the reduced-dose GC regimen especially in two subgroups of patients due to the risk of failure: Patients receiving rituximab as induction therapy [and] patients with severe initial kidney disease (serum creatinine > 300 micromol/L).”
The study authors note several limitations: The study is retrospective, and the standard dose group is heterogeneous.
“This study raises the idea that we need to be careful in using low-dose glucocorticoid regimens, but not avoid them all together,” Dr. Byram said. “The finding that those with worse kidney function fared worse lines up with my clinical experiences. There are clearly populations with this disease that could benefit from more steroid, and it tends to be the ones that are sicker at presentation, particularly those requiring ICU-level care.”
He advised colleagues to “not be dogmatic and use strict low-dose regimens ‘just because.’ ”
No study funding was reported. Dr. Nagle reported having no relevant financial relationships, and disclosures for other authors were not reported. Dr. Byram reports serving on the Vasculitis Foundation board of directors.
SAN DIEGO – A real-world analysis linked the PEXIVAS reduced-dose glucocorticoid (GC) regimen to a higher likelihood of a group of poor outcomes such as death in patients with severe granulomatosis with polyangiitis (GPA) or microscopic polyangiitis (MPA).
The retrospective observational study, presented at the annual meeting of the American College of Rheumatology, aimed to see whether results from the landmark PEXIVAS trial would hold up in a real-world analysis given some important limitations of the PEXIVAS’s primary outcome and its relative lack of balance in choice of induction agents.
First author of the new study, Sophie Nagle, MD, of Cochin Hospital in Paris, and colleagues in the French Vasculitis Study Group noted that PEXIVAS “demonstrated noninferiority of reduced-dose GC regimen compared to standard dose for the incidence of death or end-stage kidney disease (ESKD), with a significant reduction in serious infections at 1 year. However, the primary endpoint did not include disease progression or relapse, the majority of patients received cyclophosphamide as induction therapy, and subgroup analysis showed a trend towards an increased risk of death or ESKD in [rituximab]-treated patients.”
The new findings “give us pause about using low-dose glucocorticoid regimens” and suggest that rheumatologists might be a little more conservative about their use than randomized controlled trials such as PEXIVAS might suggest, said Vanderbilt University vasculitis specialist Kevin W. Byram, MD, who’s familiar with the findings but did not take part in the study.
Dr. Nagle reported that among 234 patients with either GPA or MPA, 33.3% of 126 who received a reduced-dose GC regimen experienced one of the 12-month composite primary outcome’s events of death, disease relapse, ESKD, or disease progression before remission that required treatment modification, compared with 18.5% of 108 who received the standard GC regimen (P = .016).
In a propensity score analysis, the higher risk of poor outcomes in the reduced-dose group remained (hazard ratio, 1.57; 95% confidence interval, 0.93-2.64). A multivariate analysis also identified a higher risk for the composite primary outcome in the reduced-dose group (HR, 1.72; 95% CI, 1.08-2.74), although there was no association with an increased risk of death or ESKD.
The PEXIVAS study, published in 2020, supported lower GC doses in antineutrophil cytoplasmic antibody–associated vasculitis, potentially revolutionizing treatment. “Historically, we have used high doses of glucocorticoids on slower tapers to treat this disease, which itself is a strategy that leads to potential complications,” Dr. Byram said. “PEXIVAS suggested we could potentially use less glucocorticoids in these patients.”
For the retrospective, multicenter study, researchers tracked patients from 2018 to 2022, all aged 15 and above. They included 93 with MPA and 141 with GPA. Nearly half were female, and they had a mean age of 61 years. The patients had severe flare-ups treated with rituximab or cyclophosphamide induction and reduced-dose or standard GC regimen.
The standard care and reduced-dose groups were similar, Dr. Nagle said, although the standard group had significantly more patients with GPA (71% vs. 29% with MPA) than did the low-dose group (51% with GPA, 49% with MPA).
The researchers reported that in a reduced-dose subgroup, patients with creatinine levels above 300 micromol/L were more likely to meet the primary endpoint (relative risk, 2.14; 95% CI, 1.14-4.03). Those treated with the reduced-dose GC regimen were also more likely to reach the primary endpoint (HR, 1.61; 95% CI, 0.94-2.77) and die or develop ESKD (HR, 2.42; 95% CI, 1.04-5.66).
However, adverse events at 12 months were similar in both groups: The authors noted that those who received the reduced-dose GC regimen didn’t have higher risk of death, ESKD, or severe infections.
The authors highlighted that “increased vigilance is required when using the reduced-dose GC regimen especially in two subgroups of patients due to the risk of failure: Patients receiving rituximab as induction therapy [and] patients with severe initial kidney disease (serum creatinine > 300 micromol/L).”
The study authors note several limitations: The study is retrospective, and the standard dose group is heterogeneous.
“This study raises the idea that we need to be careful in using low-dose glucocorticoid regimens, but not avoid them all together,” Dr. Byram said. “The finding that those with worse kidney function fared worse lines up with my clinical experiences. There are clearly populations with this disease that could benefit from more steroid, and it tends to be the ones that are sicker at presentation, particularly those requiring ICU-level care.”
He advised colleagues to “not be dogmatic and use strict low-dose regimens ‘just because.’ ”
No study funding was reported. Dr. Nagle reported having no relevant financial relationships, and disclosures for other authors were not reported. Dr. Byram reports serving on the Vasculitis Foundation board of directors.
SAN DIEGO – A real-world analysis linked the PEXIVAS reduced-dose glucocorticoid (GC) regimen to a higher likelihood of a group of poor outcomes such as death in patients with severe granulomatosis with polyangiitis (GPA) or microscopic polyangiitis (MPA).
The retrospective observational study, presented at the annual meeting of the American College of Rheumatology, aimed to see whether results from the landmark PEXIVAS trial would hold up in a real-world analysis given some important limitations of the PEXIVAS’s primary outcome and its relative lack of balance in choice of induction agents.
First author of the new study, Sophie Nagle, MD, of Cochin Hospital in Paris, and colleagues in the French Vasculitis Study Group noted that PEXIVAS “demonstrated noninferiority of reduced-dose GC regimen compared to standard dose for the incidence of death or end-stage kidney disease (ESKD), with a significant reduction in serious infections at 1 year. However, the primary endpoint did not include disease progression or relapse, the majority of patients received cyclophosphamide as induction therapy, and subgroup analysis showed a trend towards an increased risk of death or ESKD in [rituximab]-treated patients.”
The new findings “give us pause about using low-dose glucocorticoid regimens” and suggest that rheumatologists might be a little more conservative about their use than randomized controlled trials such as PEXIVAS might suggest, said Vanderbilt University vasculitis specialist Kevin W. Byram, MD, who’s familiar with the findings but did not take part in the study.
Dr. Nagle reported that among 234 patients with either GPA or MPA, 33.3% of 126 who received a reduced-dose GC regimen experienced one of the 12-month composite primary outcome’s events of death, disease relapse, ESKD, or disease progression before remission that required treatment modification, compared with 18.5% of 108 who received the standard GC regimen (P = .016).
In a propensity score analysis, the higher risk of poor outcomes in the reduced-dose group remained (hazard ratio, 1.57; 95% confidence interval, 0.93-2.64). A multivariate analysis also identified a higher risk for the composite primary outcome in the reduced-dose group (HR, 1.72; 95% CI, 1.08-2.74), although there was no association with an increased risk of death or ESKD.
The PEXIVAS study, published in 2020, supported lower GC doses in antineutrophil cytoplasmic antibody–associated vasculitis, potentially revolutionizing treatment. “Historically, we have used high doses of glucocorticoids on slower tapers to treat this disease, which itself is a strategy that leads to potential complications,” Dr. Byram said. “PEXIVAS suggested we could potentially use less glucocorticoids in these patients.”
For the retrospective, multicenter study, researchers tracked patients from 2018 to 2022, all aged 15 and above. They included 93 with MPA and 141 with GPA. Nearly half were female, and they had a mean age of 61 years. The patients had severe flare-ups treated with rituximab or cyclophosphamide induction and reduced-dose or standard GC regimen.
The standard care and reduced-dose groups were similar, Dr. Nagle said, although the standard group had significantly more patients with GPA (71% vs. 29% with MPA) than did the low-dose group (51% with GPA, 49% with MPA).
The researchers reported that in a reduced-dose subgroup, patients with creatinine levels above 300 micromol/L were more likely to meet the primary endpoint (relative risk, 2.14; 95% CI, 1.14-4.03). Those treated with the reduced-dose GC regimen were also more likely to reach the primary endpoint (HR, 1.61; 95% CI, 0.94-2.77) and die or develop ESKD (HR, 2.42; 95% CI, 1.04-5.66).
However, adverse events at 12 months were similar in both groups: The authors noted that those who received the reduced-dose GC regimen didn’t have higher risk of death, ESKD, or severe infections.
The authors highlighted that “increased vigilance is required when using the reduced-dose GC regimen especially in two subgroups of patients due to the risk of failure: Patients receiving rituximab as induction therapy [and] patients with severe initial kidney disease (serum creatinine > 300 micromol/L).”
The study authors note several limitations: The study is retrospective, and the standard dose group is heterogeneous.
“This study raises the idea that we need to be careful in using low-dose glucocorticoid regimens, but not avoid them all together,” Dr. Byram said. “The finding that those with worse kidney function fared worse lines up with my clinical experiences. There are clearly populations with this disease that could benefit from more steroid, and it tends to be the ones that are sicker at presentation, particularly those requiring ICU-level care.”
He advised colleagues to “not be dogmatic and use strict low-dose regimens ‘just because.’ ”
No study funding was reported. Dr. Nagle reported having no relevant financial relationships, and disclosures for other authors were not reported. Dr. Byram reports serving on the Vasculitis Foundation board of directors.
AT ACR 2023
Pregnancy in rheumatic disease quadruples risk of cardiovascular events
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.
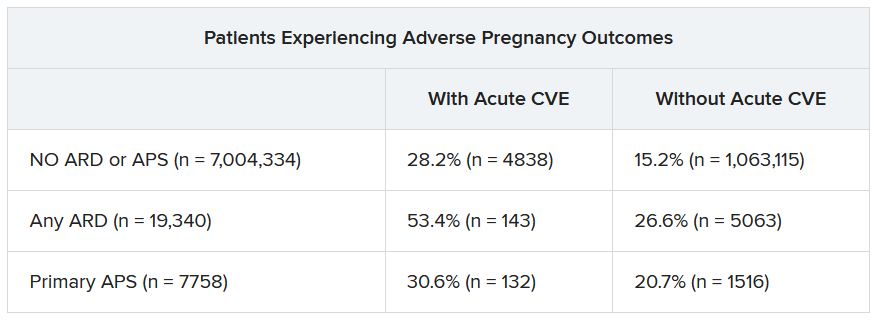
Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.

Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Pregnant individuals with autoimmune rheumatic diseases (ARDs) are at least four times more likely to experience an acute cardiovascular event (CVE) than are pregnant individuals without these conditions, according to new research presented at the annual meeting of the American College of Rheumatology. Pregnant individuals with primary antiphospholipid syndrome (APS) had a 15-fold increase in CVE risk.
Patients who experienced CVEs were also more likely to experience preterm birth and other adverse pregnancy outcomes (APOs).
Rashmi Dhital, MD, a rheumatology fellow at the University of California, San Diego, and colleagues examined the medical records of pregnant individuals in California who had delivered singleton live-born infants from 2005 to 2020. Using data from the Study of Outcomes in Mothers and Infants (SOMI) database, an administrative population-based birth cohort in California, they identified more than 7 million individuals, 19,340 with ARDs and 7,758 with APS.
They then analyzed how many patients experienced an acute CVE during pregnancy and up to 6 weeks after giving birth.
CVEs occurred in 2.0% of patients with ARDs, 6.9% of individuals with APS, and 0.4% of women without these conditions. CVE risk was four times higher in the ARDs group (adjusted relative risk, 4.1; 95% confidence interval, 3.7-4.5) and nearly 15 times higher in the APS group (aRR, 14.7; 95% CI, 13.5-16.0) than in the comparison group. Patients with systemic lupus erythematosus (SLE) had a sixfold higher risk of CVE, which was further exacerbated by concomitant APS (18-fold higher risk) or lupus nephritis (15-fold higher risk).
Dr. Dhital also classified CVEs as either venous thromboembolism and non-VTE events. Pregnant patients with APS had a high risk for VTE-only CVE (40-fold greater) and a 3.7-fold higher risk of non-VTE events, compared with pregnant patients without these conditions. Patients with SLE along with lupus nephritis had a 20-fold increased risk of VTE-only CVE and an 11-fold higher risk of non-VTE CVE.
Although the study grouped rheumatic diseases together, “lupus is generally driving these results,” Sharon Kolasinski, MD, of the University of Pennsylvania, Philadelphia, noted in an interview. She moderated the plenary session where the research was presented. “If you take out lupus, then what is the risk? That would be an interesting question.”
Between 25% and 30% of all CVEs occurred in the postpartum period, highlighting the importance of close monitoring of cardiovascular risks and events in women with ARDs or APS both during pregnancy and postpartum, Dr. Dhital noted.
Recognizing these risks “can sometimes be challenging due to a lower suspicion of CVE in younger patients, and also symptoms overlap with normal pregnancy,” Dr. Dhital said during her plenary presentation. Working with other clinical teams could help physicians detect these risks in patients.
“It’s important for us to remember that there’s increased risk of cardiovascular events in pregnancy in our patients. It’s uncommon, but it’s not zero,” added Dr. Kolasinski, and this study highlighted when physicians should be more focused about that risk.
Dr. Dhital noted there were some limitations to the study that are inherent in using administrative databases for research that relies on ICD codes, including “the availability of information on disease activity, medications, and labs, which may restrict clinical interpretation.”
SOMI data reinforced by National Inpatient Sample study
The findings were complemented by a study using the National Inpatient Sample database to explore CVE risk in pregnant individuals with various rheumatic diseases. Lead author Karun Shrestha, MD, a resident physician at St. Barnabas Hospital in New York, and colleagues identified delivery hospitalizations from 2016 to 2019 for individuals with SLE, RA, and systemic vasculitis and looked for CVEs including preeclampsia, peripartum cardiomyopathy (PPCM), heart failure, stroke, cardiac arrhythmias, and VTE.
Out of over 3.4 million delivery hospitalizations, researchers identified 5,900 individuals with SLE, 4,895 with RA, and 325 with vasculitis. After adjusting for confounding factors such as race, age, insurance, and other comorbidities, SLE was identified as an independent risk factor for preeclampsia (odds ratio, 1.5; 95% CI, 1.1-2.1), arrhythmia (OR, 3.17; 95% CI, 1.73-5.79), and venous thrombosis (OR, 8.4; 95% CI, 2.9-22.1). Vasculitis was tied to increased risk for preeclampsia (OR, 4.7; 95% CI, 2-11.3), stroke (OR, 513.3; 95% CI, 114-2,284), heart failure (OR, 24.17; 95% CI, 4.68-124.6), and PPCM (OR, 66.7; 95% CI, 8.7-509.4). RA was tied to an increased risk for preeclampsia (OR, 1.5; 95% CI, 1.05-2.1).
Patients with SLE or vasculitis had longer, more costly hospital stays, compared with those without these conditions, and they experienced higher rates of in-hospital mortality. While previous research has demonstrated that patients with SLE have higher risk of cardiac events, there is less literature on CVE risk in pregnancies for vasculitis, Dr. Shrestha said in an interview.
“It’s something to work on,” he said.
Adverse pregnancy outcomes higher with ARDs, APS
In a second abstract also led by Dr. Dhital using SOMI data, researchers found that pregnant individuals with ARDs or APS had a higher risk of experiencing an APO – preterm birth or small-for-gestational age – than individuals without these conditions. CVEs exacerbated that risk, regardless of underlying chronic health conditions.

Over half of patients with an ARD and a CVE during pregnancy experienced an APO – most commonly preterm birth. More than one in four pregnant individuals without ARD or APS who experienced a CVE also had an APO.
After differentiating CVEs as either VTE and non-VTE events, patients with ARD and a non-VTE CVE had a fivefold greater risk of early preterm birth (< 32 weeks) and a threefold higher risk of moderate preterm birth (32 to < 34 weeks).
“These findings highlight the need for close monitoring and management of pregnant women, not only for adverse outcomes, but also for cardiovascular risks and events, in order to identify those at the highest risk for adverse outcomes,” the authors wrote. “This need is particularly significant for individuals with ARDs, as 53.4% of our population with an ARD and CVE in pregnancy experienced an APO.”
Dr. Dhital, Dr. Kolasinski, and Dr. Shrestha disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACR 2023
Anemia, iron deficit common in rheumatic disease pregnancy
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
More evidence shows COVID-19’s link to risk for autoimmune disease
TOPLINE:
Research from South Korea provides additional evidence for the connection between COVID-19 and an increased risk for autoimmune conditions post infection.
METHODOLOGY:
- In this retrospective study, researchers identified 354,527 individuals diagnosed with COVID-19 via polymerase chain reaction (PCR) testing from Oct. 8, 2020, to Dec. 31, 2021.
- Researchers compared the COVID-19 group with 6,134,940 healthy individuals who had no evidence of COVID-19 to quantify the risk for autoimmune and autoinflammatory connective tissue disorders.
- Patients were followed until diagnosis, death, or end of study period (Dec. 31, 2021).
TAKEAWAY:
- Risks for alopecia areata, alopecia totalis, antineutrophil cytoplasmic antibody–associated vasculitis, Crohn’s disease, and sarcoidosis were higher in the COVID-19 group.
- Patients with more severe COVID-19 (admitted to the ICU) were at greater risk for many autoimmune conditions, including alopecia totalis, psoriasis, vitiligo, and vasculitis.
IN PRACTICE:
“Our results emphasize the need to focus on managing not only the acute stages of COVID-19 itself but also autoimmune diseases as complications of COVID-19,” the authors wrote.
SOURCE:
Sung Ha Lim, MD, of Yonsei University, Wonju, South Korea, was the first author of the study, published in JAMA Network Open.
LIMITATIONS:
The study was retrospective and was composed almost exclusively of individuals from a single ethnicity. The study could have included individuals with COVID-19 in the control group who did not undergo PCR testing. The analysis did not include detailed information on each patient, including genetic information, that could have contributed to autoimmune disease risk.
DISCLOSURES:
The study was supported by a fund from the research program of the Korea Medical Institute and by grants from the Korea Health Industry Development Institute, the Korean Ministry of Health & Welfare, and the National Research Foundation of Korea. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Research from South Korea provides additional evidence for the connection between COVID-19 and an increased risk for autoimmune conditions post infection.
METHODOLOGY:
- In this retrospective study, researchers identified 354,527 individuals diagnosed with COVID-19 via polymerase chain reaction (PCR) testing from Oct. 8, 2020, to Dec. 31, 2021.
- Researchers compared the COVID-19 group with 6,134,940 healthy individuals who had no evidence of COVID-19 to quantify the risk for autoimmune and autoinflammatory connective tissue disorders.
- Patients were followed until diagnosis, death, or end of study period (Dec. 31, 2021).
TAKEAWAY:
- Risks for alopecia areata, alopecia totalis, antineutrophil cytoplasmic antibody–associated vasculitis, Crohn’s disease, and sarcoidosis were higher in the COVID-19 group.
- Patients with more severe COVID-19 (admitted to the ICU) were at greater risk for many autoimmune conditions, including alopecia totalis, psoriasis, vitiligo, and vasculitis.
IN PRACTICE:
“Our results emphasize the need to focus on managing not only the acute stages of COVID-19 itself but also autoimmune diseases as complications of COVID-19,” the authors wrote.
SOURCE:
Sung Ha Lim, MD, of Yonsei University, Wonju, South Korea, was the first author of the study, published in JAMA Network Open.
LIMITATIONS:
The study was retrospective and was composed almost exclusively of individuals from a single ethnicity. The study could have included individuals with COVID-19 in the control group who did not undergo PCR testing. The analysis did not include detailed information on each patient, including genetic information, that could have contributed to autoimmune disease risk.
DISCLOSURES:
The study was supported by a fund from the research program of the Korea Medical Institute and by grants from the Korea Health Industry Development Institute, the Korean Ministry of Health & Welfare, and the National Research Foundation of Korea. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Research from South Korea provides additional evidence for the connection between COVID-19 and an increased risk for autoimmune conditions post infection.
METHODOLOGY:
- In this retrospective study, researchers identified 354,527 individuals diagnosed with COVID-19 via polymerase chain reaction (PCR) testing from Oct. 8, 2020, to Dec. 31, 2021.
- Researchers compared the COVID-19 group with 6,134,940 healthy individuals who had no evidence of COVID-19 to quantify the risk for autoimmune and autoinflammatory connective tissue disorders.
- Patients were followed until diagnosis, death, or end of study period (Dec. 31, 2021).
TAKEAWAY:
- Risks for alopecia areata, alopecia totalis, antineutrophil cytoplasmic antibody–associated vasculitis, Crohn’s disease, and sarcoidosis were higher in the COVID-19 group.
- Patients with more severe COVID-19 (admitted to the ICU) were at greater risk for many autoimmune conditions, including alopecia totalis, psoriasis, vitiligo, and vasculitis.
IN PRACTICE:
“Our results emphasize the need to focus on managing not only the acute stages of COVID-19 itself but also autoimmune diseases as complications of COVID-19,” the authors wrote.
SOURCE:
Sung Ha Lim, MD, of Yonsei University, Wonju, South Korea, was the first author of the study, published in JAMA Network Open.
LIMITATIONS:
The study was retrospective and was composed almost exclusively of individuals from a single ethnicity. The study could have included individuals with COVID-19 in the control group who did not undergo PCR testing. The analysis did not include detailed information on each patient, including genetic information, that could have contributed to autoimmune disease risk.
DISCLOSURES:
The study was supported by a fund from the research program of the Korea Medical Institute and by grants from the Korea Health Industry Development Institute, the Korean Ministry of Health & Welfare, and the National Research Foundation of Korea. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
PET scan at diagnosis may help to predict aneurysm risk in patients with giant cell arteritis
PET scans may serve as both a diagnostic and prognostic tool in giant cell arteritis (GCA), according to a new study.
In over 100 patients with GCA who underwent 18F-fluorodeoxyglucose PET imaging, those with elevated FDG uptake at diagnosis were more likely to develop thoracic aortic aneurysms.
“PET-CT has an excellent diagnostic accuracy for the diagnosis of GCA, certainly if both extracranial and intracranial vessels were assessed. This study shows that performing PET imaging at diagnosis in patients with GCA may also help estimate the future risk for aortic aneurysm formation,” lead author Lien Moreel, MD, of the department of internal medicine at University Hospitals Leuven (Belgium), wrote in an email. “PET imaging at diagnosis can provide both diagnostic and prognostic information in one imaging tool in patients with GCA.”
Previous retrospective studies have found an association between FDG uptake at diagnosis and risk for aortic complications, but “prospective studies confirming these findings are lacking,” the investigators wrote. The study was published online in Annals of Internal Medicine.
In the study, Dr. Moreel and colleagues prospectively followed 106 individuals diagnosed with GCA who received FDG-PET within 3 days after starting glucocorticoids. Patients also had CT imaging at diagnosis and then CT imaging annually for up to 10 years.
A PET scan was considered positive with an FDG uptake of grade 2 or higher in any of seven vascular regions (thoracic and abdominal aorta, subclavian, axillary, carotid, iliac, and femoral arteries). Researchers also used the results to quantify a total vascular score (TVS). Out of the entire cohort, 75 patients had a positive PET scan result.
These patients had a larger increase in the diameter of the ascending aorta and the descending aorta, as well the volume of thoracic aorta after 5 years, compared with those who had a negative PET scan result. These changes were also associated with higher TVS at diagnosis. Of the 23 patients who developed an aortic aneurysm, 18 had a positive PET scan at diagnosis.
The risk of incident thoracic aortic aneurysms was calculated to be 10 times higher in patients with positive PET scans. Fourteen of the 15 patients (93%) with an incident thoracic aortic aneurysm had positive PET results.
Up to now, “we’ve had no way of predicting which patients might be at risk of this potentially serious complication,” Kenneth Warrington, MD, chair of the department of rheumatology and director of the Vasculitis Clinic at the Mayo Clinic in Rochester, Minn., said in an interview. He was not involved with the research.
He hopes that the findings will help inform clinicians on how patients with GCA should be evaluated and monitored. Although the American College of Rheumatology conditionally recommends noninvasive imaging in patients newly diagnosed with GCA, guidance for follow-up on these patients is less clear.
“There are no clear guidelines, but most clinicians who take care of patients with GCA do obtain imaging periodically,” he said. “There is a lot of variability in the practice in terms of which type of scan is used and how often it’s done.”
Although this study did not specifically look at the benefit of screening patients, “we think that follow-up of aortic dimensions seems to be warranted in GCA patients with a positive PET scan result, especially in those with high intensity and broad extent of vascular inflammation,” Dr. Moreel said. “However, the added value of screening and the interval required should be addressed in future studies.”
Applying this study’s protocol in practice in the United States might be difficult, Dr. Warrington noted, as it can be challenging logistically to get imaging done within 3 days of starting steroids. However, Dr. Moreel said it is possible to delay the start of glucocorticoids until the PET scan is performed in patients without visual symptoms or jaw claudication.
PET scans are also expensive, and it can be difficult to get insurance coverage in the United States. However, other imaging modalities could potentially be used in similar ways, Dr. Warrington said. “One could potentially extrapolate to say that if there is difficulty with accessing PET scan, we could use other modalities like CT or MRI basically to see whether the aorta is inflamed or not.”
Dr. Moreel disclosed no relevant financial relationships. Dr. Warrington has received compensation for consulting activities with Sanofi. Eli Lilly, Kiniksa, and Bristol-Myers Squibb have provided support to the Mayo Clinic for clinical trials related to GCA, of which Dr. Warrington served as subinvestigator.
A version of this article appeared on Medscape.com.
PET scans may serve as both a diagnostic and prognostic tool in giant cell arteritis (GCA), according to a new study.
In over 100 patients with GCA who underwent 18F-fluorodeoxyglucose PET imaging, those with elevated FDG uptake at diagnosis were more likely to develop thoracic aortic aneurysms.
“PET-CT has an excellent diagnostic accuracy for the diagnosis of GCA, certainly if both extracranial and intracranial vessels were assessed. This study shows that performing PET imaging at diagnosis in patients with GCA may also help estimate the future risk for aortic aneurysm formation,” lead author Lien Moreel, MD, of the department of internal medicine at University Hospitals Leuven (Belgium), wrote in an email. “PET imaging at diagnosis can provide both diagnostic and prognostic information in one imaging tool in patients with GCA.”
Previous retrospective studies have found an association between FDG uptake at diagnosis and risk for aortic complications, but “prospective studies confirming these findings are lacking,” the investigators wrote. The study was published online in Annals of Internal Medicine.
In the study, Dr. Moreel and colleagues prospectively followed 106 individuals diagnosed with GCA who received FDG-PET within 3 days after starting glucocorticoids. Patients also had CT imaging at diagnosis and then CT imaging annually for up to 10 years.
A PET scan was considered positive with an FDG uptake of grade 2 or higher in any of seven vascular regions (thoracic and abdominal aorta, subclavian, axillary, carotid, iliac, and femoral arteries). Researchers also used the results to quantify a total vascular score (TVS). Out of the entire cohort, 75 patients had a positive PET scan result.
These patients had a larger increase in the diameter of the ascending aorta and the descending aorta, as well the volume of thoracic aorta after 5 years, compared with those who had a negative PET scan result. These changes were also associated with higher TVS at diagnosis. Of the 23 patients who developed an aortic aneurysm, 18 had a positive PET scan at diagnosis.
The risk of incident thoracic aortic aneurysms was calculated to be 10 times higher in patients with positive PET scans. Fourteen of the 15 patients (93%) with an incident thoracic aortic aneurysm had positive PET results.
Up to now, “we’ve had no way of predicting which patients might be at risk of this potentially serious complication,” Kenneth Warrington, MD, chair of the department of rheumatology and director of the Vasculitis Clinic at the Mayo Clinic in Rochester, Minn., said in an interview. He was not involved with the research.
He hopes that the findings will help inform clinicians on how patients with GCA should be evaluated and monitored. Although the American College of Rheumatology conditionally recommends noninvasive imaging in patients newly diagnosed with GCA, guidance for follow-up on these patients is less clear.
“There are no clear guidelines, but most clinicians who take care of patients with GCA do obtain imaging periodically,” he said. “There is a lot of variability in the practice in terms of which type of scan is used and how often it’s done.”
Although this study did not specifically look at the benefit of screening patients, “we think that follow-up of aortic dimensions seems to be warranted in GCA patients with a positive PET scan result, especially in those with high intensity and broad extent of vascular inflammation,” Dr. Moreel said. “However, the added value of screening and the interval required should be addressed in future studies.”
Applying this study’s protocol in practice in the United States might be difficult, Dr. Warrington noted, as it can be challenging logistically to get imaging done within 3 days of starting steroids. However, Dr. Moreel said it is possible to delay the start of glucocorticoids until the PET scan is performed in patients without visual symptoms or jaw claudication.
PET scans are also expensive, and it can be difficult to get insurance coverage in the United States. However, other imaging modalities could potentially be used in similar ways, Dr. Warrington said. “One could potentially extrapolate to say that if there is difficulty with accessing PET scan, we could use other modalities like CT or MRI basically to see whether the aorta is inflamed or not.”
Dr. Moreel disclosed no relevant financial relationships. Dr. Warrington has received compensation for consulting activities with Sanofi. Eli Lilly, Kiniksa, and Bristol-Myers Squibb have provided support to the Mayo Clinic for clinical trials related to GCA, of which Dr. Warrington served as subinvestigator.
A version of this article appeared on Medscape.com.
PET scans may serve as both a diagnostic and prognostic tool in giant cell arteritis (GCA), according to a new study.
In over 100 patients with GCA who underwent 18F-fluorodeoxyglucose PET imaging, those with elevated FDG uptake at diagnosis were more likely to develop thoracic aortic aneurysms.
“PET-CT has an excellent diagnostic accuracy for the diagnosis of GCA, certainly if both extracranial and intracranial vessels were assessed. This study shows that performing PET imaging at diagnosis in patients with GCA may also help estimate the future risk for aortic aneurysm formation,” lead author Lien Moreel, MD, of the department of internal medicine at University Hospitals Leuven (Belgium), wrote in an email. “PET imaging at diagnosis can provide both diagnostic and prognostic information in one imaging tool in patients with GCA.”
Previous retrospective studies have found an association between FDG uptake at diagnosis and risk for aortic complications, but “prospective studies confirming these findings are lacking,” the investigators wrote. The study was published online in Annals of Internal Medicine.
In the study, Dr. Moreel and colleagues prospectively followed 106 individuals diagnosed with GCA who received FDG-PET within 3 days after starting glucocorticoids. Patients also had CT imaging at diagnosis and then CT imaging annually for up to 10 years.
A PET scan was considered positive with an FDG uptake of grade 2 or higher in any of seven vascular regions (thoracic and abdominal aorta, subclavian, axillary, carotid, iliac, and femoral arteries). Researchers also used the results to quantify a total vascular score (TVS). Out of the entire cohort, 75 patients had a positive PET scan result.
These patients had a larger increase in the diameter of the ascending aorta and the descending aorta, as well the volume of thoracic aorta after 5 years, compared with those who had a negative PET scan result. These changes were also associated with higher TVS at diagnosis. Of the 23 patients who developed an aortic aneurysm, 18 had a positive PET scan at diagnosis.
The risk of incident thoracic aortic aneurysms was calculated to be 10 times higher in patients with positive PET scans. Fourteen of the 15 patients (93%) with an incident thoracic aortic aneurysm had positive PET results.
Up to now, “we’ve had no way of predicting which patients might be at risk of this potentially serious complication,” Kenneth Warrington, MD, chair of the department of rheumatology and director of the Vasculitis Clinic at the Mayo Clinic in Rochester, Minn., said in an interview. He was not involved with the research.
He hopes that the findings will help inform clinicians on how patients with GCA should be evaluated and monitored. Although the American College of Rheumatology conditionally recommends noninvasive imaging in patients newly diagnosed with GCA, guidance for follow-up on these patients is less clear.
“There are no clear guidelines, but most clinicians who take care of patients with GCA do obtain imaging periodically,” he said. “There is a lot of variability in the practice in terms of which type of scan is used and how often it’s done.”
Although this study did not specifically look at the benefit of screening patients, “we think that follow-up of aortic dimensions seems to be warranted in GCA patients with a positive PET scan result, especially in those with high intensity and broad extent of vascular inflammation,” Dr. Moreel said. “However, the added value of screening and the interval required should be addressed in future studies.”
Applying this study’s protocol in practice in the United States might be difficult, Dr. Warrington noted, as it can be challenging logistically to get imaging done within 3 days of starting steroids. However, Dr. Moreel said it is possible to delay the start of glucocorticoids until the PET scan is performed in patients without visual symptoms or jaw claudication.
PET scans are also expensive, and it can be difficult to get insurance coverage in the United States. However, other imaging modalities could potentially be used in similar ways, Dr. Warrington said. “One could potentially extrapolate to say that if there is difficulty with accessing PET scan, we could use other modalities like CT or MRI basically to see whether the aorta is inflamed or not.”
Dr. Moreel disclosed no relevant financial relationships. Dr. Warrington has received compensation for consulting activities with Sanofi. Eli Lilly, Kiniksa, and Bristol-Myers Squibb have provided support to the Mayo Clinic for clinical trials related to GCA, of which Dr. Warrington served as subinvestigator.
A version of this article appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE