User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Vegetarian diets can improve high-risk cardiovascular disease
, a meta-analysis of randomized controlled trials shows.
“To the best of our knowledge, this meta-analysis is the first that generates evidence from randomized controlled trials to assess the association of vegetarian diets with outcomes in people affected by cardiovascular diseases,” report the authors. The study was published online in JAMA Network Open.
“The greatest improvements in hemoglobin A1c and low-density lipoprotein cholesterol (LDL-C) were observed in individuals with type 2 diabetes and people at high risk of cardiovascular disease, highlighting the potential protective and synergistic effects of vegetarian diets for the primary prevention of cardiovascular disease,” they say.
Poor diet is well-established as increasing the morbidity and mortality associated with cardiovascular disease; however, although data has linked vegetarian diets to cardiovascular disease prevention in the general population, research on the effectiveness of such diets in people at high risk of cardiovascular disease is lacking.
“To the best of our knowledge, no meta-analysis of randomized controlled trials has been conducted to investigate the association of vegetarian diets with outcomes among people with CVD – indeed, research here has primarily focused on observational studies,” writes Tian Wang, RD, and colleagues at the University of Sydney.
Greater decreases in LDL-C, A1c, and body weight with vegetarian diets
For the meta-analysis, researchers identified 20 randomized controlled trials involving vegetarian diets that included 1,878 adults with or at a high risk of cardiovascular disease and included measurements of LDL-C, A1c, or systolic blood pressure.
The studies were conducted in the United States, Asia, Europe, and New Zealand between 1990 and 2021. Sample sizes ranged from 12 to 291 participants.
The mean range age of participants was 28-64 years. Studies included patients with cardiovascular disease (four studies), diabetes (seven studies), and those with at least two cardiovascular risk factors (nine studies).
The mean duration of the dietary intervention was 25.4 weeks (range 2-24 months). The most commonly prescribed diets were vegan (plant-based foods only), lacto-ovo-vegetarian (excluded meat, poultry, seafood, and dairy products, but allowed eggs), and lacto-vegetarian (same as previous but allowed dairy products).
Overall, those who consumed a vegetarian diet for an average of 6 months, versus comparison diets, had significantly greater decreases in LDL-C (6.6 mg/dL beyond the reduction achieved with standard therapy); A1c (0.24%); and body weight (3.4 kg), but the reduction in systolic blood pressure (0.1 mmHg) was not significantly greater.
Assessment of the overall certainty of evidence evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) tool showed a moderate level of evidence for reductions in LDL-C and A1c with the vegetarian diet.
Lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (14.1 mg/dL); however, four out of the five trials restricted energy intake.
Of note, vegetarian diets were most effective for achieving glycemic control among people with type 2 diabetes and leading to improvements in weight among those at high risk of cardiovascular disease as well as those with type 2 diabetes.
The effects “suggest that vegetarian diets might have a synergistic [or at least nonantagonistic] use in potentiating the effects of optimal drug therapy in the prevention and treatment of a range of cardiometabolic diseases,” the authors write.
Although previous studies have shown similar improvements associated with a vegetarian diet, most studies did not stratify populations based on disease status, type of vegetarian diet, or comparison diet, the authors note.
The lack of improvement in systolic blood pressure is consistent with previous meta-analyses of vegetarian diets in general and suggests that salt intake may be the more important factor for those measures.
“[The meta-analysis] suggests that diet quality plays a major role in lowering blood pressure independent of animal food consumption, as the DASH [Dietary Approaches to Stop Hypertension] ... trial demonstrated,” the authors note.
Decreases in medication dose with vegetarian diet
Although most patients were taking medications to manage hypertension, hyperglycemia, and/or dyslipidemia at trial enrollment in as many as eight of the studies, the vegetarian diet intervention resulted in a decrease in medication dose.
In fact, medication use could obscure the favorable effects of vegetarian diets, which could have a larger effect size, the authors speculate.
“This hypothesis is supported by two randomized controlled trials in our meta-analysis that required patients not to take medication that could influence cardiometabolic outcomes, [and] these studies significantly improved systolic blood pressure and LDL-C,” they write.
Not all vegetarian diets are healthy
Although there are numerous variations in vegetarian diets, ranging from vegan diets that eliminate all animal food to pesco-vegetarian diets that allow fish or seafood, most that are well-balanced can provide health benefits including lower saturated fat, L-carnitine, and choline (precursors of the atherogenic TMAO), and other benefits that might explain the improvements seen in the meta-analysis.
The diets may also be high in dietary fiber, mono- and polyunsaturated fatty acids, potassium, magnesium, and phytochemical, and have lower glycemic index scores.
Of note, 12 studies in the meta-analysis emphasized low-fat content, which the authors speculate may have contributed to the improvements observed in LDC-C.
Specifically, lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (–14.1 mg/dL); however, four out of five of the trials restricted energy intake, which could have also played a role in improvements.
Importantly, not all vegetarian diets are healthy, and the authors caution about some that allow, for instance, deep-fried foods rich in trans-fatty acids and salt, such as tempura vegetables, potentially increasing the risk of type 2 diabetes and coronary heart disease.
They note that “more than one-third of the studies included in our meta-analysis did not emphasize the importance of consuming minimally processed plant-based whole foods.”
Overall, however, the fact that the greatest improvements in A1c and LDL-C were seen in patients with type 2 diabetes and those at high risk of CVD “highlight[s] the potential protective and synergistic effects of vegetarian diets for the primary prevention of CVD.”
A version of this article first appeared on Medscape.com.
, a meta-analysis of randomized controlled trials shows.
“To the best of our knowledge, this meta-analysis is the first that generates evidence from randomized controlled trials to assess the association of vegetarian diets with outcomes in people affected by cardiovascular diseases,” report the authors. The study was published online in JAMA Network Open.
“The greatest improvements in hemoglobin A1c and low-density lipoprotein cholesterol (LDL-C) were observed in individuals with type 2 diabetes and people at high risk of cardiovascular disease, highlighting the potential protective and synergistic effects of vegetarian diets for the primary prevention of cardiovascular disease,” they say.
Poor diet is well-established as increasing the morbidity and mortality associated with cardiovascular disease; however, although data has linked vegetarian diets to cardiovascular disease prevention in the general population, research on the effectiveness of such diets in people at high risk of cardiovascular disease is lacking.
“To the best of our knowledge, no meta-analysis of randomized controlled trials has been conducted to investigate the association of vegetarian diets with outcomes among people with CVD – indeed, research here has primarily focused on observational studies,” writes Tian Wang, RD, and colleagues at the University of Sydney.
Greater decreases in LDL-C, A1c, and body weight with vegetarian diets
For the meta-analysis, researchers identified 20 randomized controlled trials involving vegetarian diets that included 1,878 adults with or at a high risk of cardiovascular disease and included measurements of LDL-C, A1c, or systolic blood pressure.
The studies were conducted in the United States, Asia, Europe, and New Zealand between 1990 and 2021. Sample sizes ranged from 12 to 291 participants.
The mean range age of participants was 28-64 years. Studies included patients with cardiovascular disease (four studies), diabetes (seven studies), and those with at least two cardiovascular risk factors (nine studies).
The mean duration of the dietary intervention was 25.4 weeks (range 2-24 months). The most commonly prescribed diets were vegan (plant-based foods only), lacto-ovo-vegetarian (excluded meat, poultry, seafood, and dairy products, but allowed eggs), and lacto-vegetarian (same as previous but allowed dairy products).
Overall, those who consumed a vegetarian diet for an average of 6 months, versus comparison diets, had significantly greater decreases in LDL-C (6.6 mg/dL beyond the reduction achieved with standard therapy); A1c (0.24%); and body weight (3.4 kg), but the reduction in systolic blood pressure (0.1 mmHg) was not significantly greater.
Assessment of the overall certainty of evidence evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) tool showed a moderate level of evidence for reductions in LDL-C and A1c with the vegetarian diet.
Lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (14.1 mg/dL); however, four out of the five trials restricted energy intake.
Of note, vegetarian diets were most effective for achieving glycemic control among people with type 2 diabetes and leading to improvements in weight among those at high risk of cardiovascular disease as well as those with type 2 diabetes.
The effects “suggest that vegetarian diets might have a synergistic [or at least nonantagonistic] use in potentiating the effects of optimal drug therapy in the prevention and treatment of a range of cardiometabolic diseases,” the authors write.
Although previous studies have shown similar improvements associated with a vegetarian diet, most studies did not stratify populations based on disease status, type of vegetarian diet, or comparison diet, the authors note.
The lack of improvement in systolic blood pressure is consistent with previous meta-analyses of vegetarian diets in general and suggests that salt intake may be the more important factor for those measures.
“[The meta-analysis] suggests that diet quality plays a major role in lowering blood pressure independent of animal food consumption, as the DASH [Dietary Approaches to Stop Hypertension] ... trial demonstrated,” the authors note.
Decreases in medication dose with vegetarian diet
Although most patients were taking medications to manage hypertension, hyperglycemia, and/or dyslipidemia at trial enrollment in as many as eight of the studies, the vegetarian diet intervention resulted in a decrease in medication dose.
In fact, medication use could obscure the favorable effects of vegetarian diets, which could have a larger effect size, the authors speculate.
“This hypothesis is supported by two randomized controlled trials in our meta-analysis that required patients not to take medication that could influence cardiometabolic outcomes, [and] these studies significantly improved systolic blood pressure and LDL-C,” they write.
Not all vegetarian diets are healthy
Although there are numerous variations in vegetarian diets, ranging from vegan diets that eliminate all animal food to pesco-vegetarian diets that allow fish or seafood, most that are well-balanced can provide health benefits including lower saturated fat, L-carnitine, and choline (precursors of the atherogenic TMAO), and other benefits that might explain the improvements seen in the meta-analysis.
The diets may also be high in dietary fiber, mono- and polyunsaturated fatty acids, potassium, magnesium, and phytochemical, and have lower glycemic index scores.
Of note, 12 studies in the meta-analysis emphasized low-fat content, which the authors speculate may have contributed to the improvements observed in LDC-C.
Specifically, lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (–14.1 mg/dL); however, four out of five of the trials restricted energy intake, which could have also played a role in improvements.
Importantly, not all vegetarian diets are healthy, and the authors caution about some that allow, for instance, deep-fried foods rich in trans-fatty acids and salt, such as tempura vegetables, potentially increasing the risk of type 2 diabetes and coronary heart disease.
They note that “more than one-third of the studies included in our meta-analysis did not emphasize the importance of consuming minimally processed plant-based whole foods.”
Overall, however, the fact that the greatest improvements in A1c and LDL-C were seen in patients with type 2 diabetes and those at high risk of CVD “highlight[s] the potential protective and synergistic effects of vegetarian diets for the primary prevention of CVD.”
A version of this article first appeared on Medscape.com.
, a meta-analysis of randomized controlled trials shows.
“To the best of our knowledge, this meta-analysis is the first that generates evidence from randomized controlled trials to assess the association of vegetarian diets with outcomes in people affected by cardiovascular diseases,” report the authors. The study was published online in JAMA Network Open.
“The greatest improvements in hemoglobin A1c and low-density lipoprotein cholesterol (LDL-C) were observed in individuals with type 2 diabetes and people at high risk of cardiovascular disease, highlighting the potential protective and synergistic effects of vegetarian diets for the primary prevention of cardiovascular disease,” they say.
Poor diet is well-established as increasing the morbidity and mortality associated with cardiovascular disease; however, although data has linked vegetarian diets to cardiovascular disease prevention in the general population, research on the effectiveness of such diets in people at high risk of cardiovascular disease is lacking.
“To the best of our knowledge, no meta-analysis of randomized controlled trials has been conducted to investigate the association of vegetarian diets with outcomes among people with CVD – indeed, research here has primarily focused on observational studies,” writes Tian Wang, RD, and colleagues at the University of Sydney.
Greater decreases in LDL-C, A1c, and body weight with vegetarian diets
For the meta-analysis, researchers identified 20 randomized controlled trials involving vegetarian diets that included 1,878 adults with or at a high risk of cardiovascular disease and included measurements of LDL-C, A1c, or systolic blood pressure.
The studies were conducted in the United States, Asia, Europe, and New Zealand between 1990 and 2021. Sample sizes ranged from 12 to 291 participants.
The mean range age of participants was 28-64 years. Studies included patients with cardiovascular disease (four studies), diabetes (seven studies), and those with at least two cardiovascular risk factors (nine studies).
The mean duration of the dietary intervention was 25.4 weeks (range 2-24 months). The most commonly prescribed diets were vegan (plant-based foods only), lacto-ovo-vegetarian (excluded meat, poultry, seafood, and dairy products, but allowed eggs), and lacto-vegetarian (same as previous but allowed dairy products).
Overall, those who consumed a vegetarian diet for an average of 6 months, versus comparison diets, had significantly greater decreases in LDL-C (6.6 mg/dL beyond the reduction achieved with standard therapy); A1c (0.24%); and body weight (3.4 kg), but the reduction in systolic blood pressure (0.1 mmHg) was not significantly greater.
Assessment of the overall certainty of evidence evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) tool showed a moderate level of evidence for reductions in LDL-C and A1c with the vegetarian diet.
Lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (14.1 mg/dL); however, four out of the five trials restricted energy intake.
Of note, vegetarian diets were most effective for achieving glycemic control among people with type 2 diabetes and leading to improvements in weight among those at high risk of cardiovascular disease as well as those with type 2 diabetes.
The effects “suggest that vegetarian diets might have a synergistic [or at least nonantagonistic] use in potentiating the effects of optimal drug therapy in the prevention and treatment of a range of cardiometabolic diseases,” the authors write.
Although previous studies have shown similar improvements associated with a vegetarian diet, most studies did not stratify populations based on disease status, type of vegetarian diet, or comparison diet, the authors note.
The lack of improvement in systolic blood pressure is consistent with previous meta-analyses of vegetarian diets in general and suggests that salt intake may be the more important factor for those measures.
“[The meta-analysis] suggests that diet quality plays a major role in lowering blood pressure independent of animal food consumption, as the DASH [Dietary Approaches to Stop Hypertension] ... trial demonstrated,” the authors note.
Decreases in medication dose with vegetarian diet
Although most patients were taking medications to manage hypertension, hyperglycemia, and/or dyslipidemia at trial enrollment in as many as eight of the studies, the vegetarian diet intervention resulted in a decrease in medication dose.
In fact, medication use could obscure the favorable effects of vegetarian diets, which could have a larger effect size, the authors speculate.
“This hypothesis is supported by two randomized controlled trials in our meta-analysis that required patients not to take medication that could influence cardiometabolic outcomes, [and] these studies significantly improved systolic blood pressure and LDL-C,” they write.
Not all vegetarian diets are healthy
Although there are numerous variations in vegetarian diets, ranging from vegan diets that eliminate all animal food to pesco-vegetarian diets that allow fish or seafood, most that are well-balanced can provide health benefits including lower saturated fat, L-carnitine, and choline (precursors of the atherogenic TMAO), and other benefits that might explain the improvements seen in the meta-analysis.
The diets may also be high in dietary fiber, mono- and polyunsaturated fatty acids, potassium, magnesium, and phytochemical, and have lower glycemic index scores.
Of note, 12 studies in the meta-analysis emphasized low-fat content, which the authors speculate may have contributed to the improvements observed in LDC-C.
Specifically, lacto-ovo vegetarian diets were associated with the greatest reduction in LDL-C (–14.1 mg/dL); however, four out of five of the trials restricted energy intake, which could have also played a role in improvements.
Importantly, not all vegetarian diets are healthy, and the authors caution about some that allow, for instance, deep-fried foods rich in trans-fatty acids and salt, such as tempura vegetables, potentially increasing the risk of type 2 diabetes and coronary heart disease.
They note that “more than one-third of the studies included in our meta-analysis did not emphasize the importance of consuming minimally processed plant-based whole foods.”
Overall, however, the fact that the greatest improvements in A1c and LDL-C were seen in patients with type 2 diabetes and those at high risk of CVD “highlight[s] the potential protective and synergistic effects of vegetarian diets for the primary prevention of CVD.”
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Early or delayed AFib ablation after heart failure hospitalization?
TOPLINE:
Among patients with atrial fibrillation (AFib) hospitalized for worsening heart failure (HF), catheter (cath) ablation within 90 days of admission, compared with other times, is associated with reduced risk for all-cause mortality and HF-related mortality.
METHODOLOGY:
Cath ablation has become technically safer for patients with both AFib and HF, but the best timing for the ablation procedure after HF hospitalization has been unclear.
The study included 2,786 patients with HF who underwent cath ablation for AFib at 128 centers in the nationwide Japanese Registry of Acute Decompensated Heart Failure, were hospitalized with worsening HF, and survived at least 90 days after discharge.
The population included 103 individuals who underwent cath ablation within 90 days after admission; the remaining 2,683 participants served as the control group.
The researchers also looked at all-cause mortality 90 days after admission for HF in analysis of 83 early-ablation cases vs. 83 propensity-matched controls.
TAKEAWAY:
The early–cath ablation group was younger, predominantly male, had less history of prior HF hospitalizations, and greater incidence of paroxysmal AF, compared with the control group.
All-cause mortality was significantly lower in the early–cath ablation group than in the control group (hazard ratio, 0.38; 95% confidence interval, 0.24-0.60; P < .001) over a median of 4.1 years.
Risk reductions were similarly significant for secondary endpoints, including cardiovascular (CV) mortality and HF mortality.
In the matched cohort analysis (83 in both groups) all-cause mortality was significantly reduced for those in the early–cath ablation group, compared with the matched controls (HR, 0.47; 95% CI, 0.25-0.88; P = .014), with similarly significant risk reductions for CV mortality and HF mortality.
IN PRACTICE:
the report states. Early catheter ablation, as early as during the hospitalization for HF, “might be a way to stabilize HF and solve the problems associated with long hospitalization periods and polypharmacy.”
SOURCE:
The study was conducted by Kazuo Sakamoto, MD, PhD, Kyushu University, Fukuoka, Japan, and colleagues. It was published online July 19, 2023 in JACC: Clinical Electrophysiology.
LIMITATIONS:
The early-ablation cohort was much smaller than the control group, and the analysis could not adjust for any variation in institutional characteristics, such as location and available equipment. Other unmeasured potential confounders include duration of AFib and patient lifestyle characteristics and success or failure of ablation.
DISCLOSURES:
The study was funded by Johnson & Johnson, the Japan Agency for Medical Research and Development, and Ministry of Health and Labor. Dr. Sakamoto reports no relevant conflicts.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among patients with atrial fibrillation (AFib) hospitalized for worsening heart failure (HF), catheter (cath) ablation within 90 days of admission, compared with other times, is associated with reduced risk for all-cause mortality and HF-related mortality.
METHODOLOGY:
Cath ablation has become technically safer for patients with both AFib and HF, but the best timing for the ablation procedure after HF hospitalization has been unclear.
The study included 2,786 patients with HF who underwent cath ablation for AFib at 128 centers in the nationwide Japanese Registry of Acute Decompensated Heart Failure, were hospitalized with worsening HF, and survived at least 90 days after discharge.
The population included 103 individuals who underwent cath ablation within 90 days after admission; the remaining 2,683 participants served as the control group.
The researchers also looked at all-cause mortality 90 days after admission for HF in analysis of 83 early-ablation cases vs. 83 propensity-matched controls.
TAKEAWAY:
The early–cath ablation group was younger, predominantly male, had less history of prior HF hospitalizations, and greater incidence of paroxysmal AF, compared with the control group.
All-cause mortality was significantly lower in the early–cath ablation group than in the control group (hazard ratio, 0.38; 95% confidence interval, 0.24-0.60; P < .001) over a median of 4.1 years.
Risk reductions were similarly significant for secondary endpoints, including cardiovascular (CV) mortality and HF mortality.
In the matched cohort analysis (83 in both groups) all-cause mortality was significantly reduced for those in the early–cath ablation group, compared with the matched controls (HR, 0.47; 95% CI, 0.25-0.88; P = .014), with similarly significant risk reductions for CV mortality and HF mortality.
IN PRACTICE:
the report states. Early catheter ablation, as early as during the hospitalization for HF, “might be a way to stabilize HF and solve the problems associated with long hospitalization periods and polypharmacy.”
SOURCE:
The study was conducted by Kazuo Sakamoto, MD, PhD, Kyushu University, Fukuoka, Japan, and colleagues. It was published online July 19, 2023 in JACC: Clinical Electrophysiology.
LIMITATIONS:
The early-ablation cohort was much smaller than the control group, and the analysis could not adjust for any variation in institutional characteristics, such as location and available equipment. Other unmeasured potential confounders include duration of AFib and patient lifestyle characteristics and success or failure of ablation.
DISCLOSURES:
The study was funded by Johnson & Johnson, the Japan Agency for Medical Research and Development, and Ministry of Health and Labor. Dr. Sakamoto reports no relevant conflicts.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among patients with atrial fibrillation (AFib) hospitalized for worsening heart failure (HF), catheter (cath) ablation within 90 days of admission, compared with other times, is associated with reduced risk for all-cause mortality and HF-related mortality.
METHODOLOGY:
Cath ablation has become technically safer for patients with both AFib and HF, but the best timing for the ablation procedure after HF hospitalization has been unclear.
The study included 2,786 patients with HF who underwent cath ablation for AFib at 128 centers in the nationwide Japanese Registry of Acute Decompensated Heart Failure, were hospitalized with worsening HF, and survived at least 90 days after discharge.
The population included 103 individuals who underwent cath ablation within 90 days after admission; the remaining 2,683 participants served as the control group.
The researchers also looked at all-cause mortality 90 days after admission for HF in analysis of 83 early-ablation cases vs. 83 propensity-matched controls.
TAKEAWAY:
The early–cath ablation group was younger, predominantly male, had less history of prior HF hospitalizations, and greater incidence of paroxysmal AF, compared with the control group.
All-cause mortality was significantly lower in the early–cath ablation group than in the control group (hazard ratio, 0.38; 95% confidence interval, 0.24-0.60; P < .001) over a median of 4.1 years.
Risk reductions were similarly significant for secondary endpoints, including cardiovascular (CV) mortality and HF mortality.
In the matched cohort analysis (83 in both groups) all-cause mortality was significantly reduced for those in the early–cath ablation group, compared with the matched controls (HR, 0.47; 95% CI, 0.25-0.88; P = .014), with similarly significant risk reductions for CV mortality and HF mortality.
IN PRACTICE:
the report states. Early catheter ablation, as early as during the hospitalization for HF, “might be a way to stabilize HF and solve the problems associated with long hospitalization periods and polypharmacy.”
SOURCE:
The study was conducted by Kazuo Sakamoto, MD, PhD, Kyushu University, Fukuoka, Japan, and colleagues. It was published online July 19, 2023 in JACC: Clinical Electrophysiology.
LIMITATIONS:
The early-ablation cohort was much smaller than the control group, and the analysis could not adjust for any variation in institutional characteristics, such as location and available equipment. Other unmeasured potential confounders include duration of AFib and patient lifestyle characteristics and success or failure of ablation.
DISCLOSURES:
The study was funded by Johnson & Johnson, the Japan Agency for Medical Research and Development, and Ministry of Health and Labor. Dr. Sakamoto reports no relevant conflicts.
A version of this article first appeared on Medscape.com.
FROM JACC: CLINICAL ELECTROPHYSIOLOGY
Time to end direct-to-consumer ads, says physician
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Daily statin cuts cardiovascular risk in HIV
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BRISBANE, AUSTRALIA – that show pitavastatin therapy is associated with a significantly lower risk of cardiovascular events than placebo.
“There was a significant 35% lower risk of major adverse cardiovascular events after a median follow-up of 5.1 years “ said Steven Grinspoon, MD, from Massachusetts General Hospital and Harvard Medical School in Boston, who presented the final analysis of data from the REPRIEVE trial at the International AIDS Society Conference on HIV Science.
The results were simultaneously published in the New England Journal of Medicine. The primary endpoint of major adverse cardiovascular events included a composite of outcomes that included cardiovascular death, stroke, myocardial infarction, hospitalization for unstable angina, and transient ischemic attack among those treated with pitavastatin, compared with placebo (95% confidence interval, 0.48-0.90; P = .002).
The REPRIEVE trial was halted earlier this year for efficacy after an interim analysis pointed to a significantly lower rate of cardiovascular events in the treatment group.
The international double-blind, placebo-controlled trial randomly assigned 7,769 people with HIV infection, who were at low to moderate risk of cardiovascular disease, to either 4 mg daily of pitavastatin calcium or placebo.
The secondary outcome – a composite of major cardiovascular events and all-cause mortality – also showed a significant 21% reduction in risk with pitavastatin treatment, compared with placebo (95% CI, 0.65-0.96).
Cardiovascular events in HIV
HIV infection is an independent risk factor for cardiovascular disease, Dr. Grinspoon pointed out, and those living with HIV have about double the risk of myocardial infarction and stroke, compared with the general population.
“There’s an unmet need for people living with HIV who have low to moderate traditional risk, for whom HIV is even considered a risk equivalent but for whom no primary prevention strategy has been tested in a large trial,” Dr. Grinspoon said during an interview.
Those enrolled in the study had a 10-year Atherosclerotic Cardiovascular Disease risk score ranging from 2.1% to 7%, with a median of 4.5%. While LDL cholesterol levels at baseline ranged from 87 to 128 mg/dL, the study showed a similar reduction in cardiovascular risk regardless of LDL.
“These are types of people who, if they came to the doctor’s office right now before REPRIEVE, they would largely be told your risk score is not really making you eligible for a statin,” Dr. Grinspoon said.
He explained that what is most interesting about the reduction in risk is that it was nearly twice what would be expected with LDL lowering, based on what has previously been seen in statin trials in non–HIV-positive populations.
“I think the data are suggesting that it’s certainly in part due to the reduction in LDL – that is very important – but it’s also due to other factors beyond changes in LDL,” Dr. Grinspoon said. He speculated that the statin could be affecting anti-inflammatory and immune pathways, and that this could account for some of the reduction in cardiovascular risk, but “those data are cooking, and they’re being analyzed as we speak.”
In a substudy analysis of REPRIEVE, Markella Zanni, MD, associate professor of medicine at Harvard Medical School and Massachusetts General Hospital, focused on the women in the clinical trial.
Women’s risk
In REPRIEVE, 31.1% of the study population were women. Dr. Zanni and her team investigated whether there are differences in the way HIV affects the risk of developing atherosclerotic cardiovascular disease in women, compared with men.
They found that women have both higher levels of inflammatory markers, such as interleukin-6, C-reactive protein, and D-dimer, but a lower prevalence of coronary artery plaques than men.
“This finding represents an interesting paradox given that high levels of select inflammatory markers have been associated with coronary artery plaque, both among women living with HIV and among men living with HIV,” Dr. Zanni explained.
She says the researchers were hoping to further explore whether inflammation is fueling the increased risk for atherosclerotic disease, and particularly the higher risk evident in women living with HIV, compared with men.
“Women living with HIV should discuss with their treating clinicians heart risks and possible prevention strategies, including statin therapy coupled with healthy lifestyle changes addressing modifiable, traditional metabolic risk factors” she said.
Time for primary prevention?
All patients in the study were on antiretroviral therapy and investigators report that pitavastatin does not interact with these medications. The median CD4 cell count was 621 cells/mm3, and 87.5% of participants had an HIV viral load below the lower limit of quantification.
Participants were enrolled from 12 countries including the United States, Spain, Brazil, South Africa, and Thailand, and around two-thirds were non-White. Individuals of South Asian ethnicity showed the biggest reduction in cardiovascular risk with pitavastatin treatment.
There was a 74% higher rate of muscle pain and weakness in the pitavastatin group – affecting 91 people in the treatment arm and 53 in the placebo arm – but the majority were low grade. The rate of rhabdomyolysis of grade 3 or above was lower in the statin group, with three cases, compared with four cases in the placebo group.
Commenting on the findings, Laura Waters, MD, a genitourinary and HIV medicine consultant at Central and North West London NHS Foundation Trust’s Mortimer Market Centre, said that, while HIV infection was considered a risk factor for cardiovascular disease, risk calculators don’t specifically adjust for HIV infection.
“Now that we’ve got effective HIV drugs and people can enjoy normal life expectancy, cardiovascular disease is a particular issue for people with HIV,” she said.
Dr. Waters, who was not involved with the study, suggested that people living with HIV should discuss the use of statins with their doctor, but she acknowledged there are some barriers to treatment in people living with HIV. “It’s another pill, and when it’s a borderline [decision] it is easy to say, ‘I have to think about it,’ ” she said, with the result that statin treatment is often deferred.
The REPRIEVE study was supported by grants from the National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare. Dr. Grinspoon declared institutional grants from National Institutes of Health, Kowa Pharmaceuticals America, Gilead Sciences, and ViiV Healthcare and consultancies unrelated to the study. Dr. Zanni reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT IAS 2023
Same-session PCI rates ‘surprisingly high’
Stable patients who have a diagnostic cardiac catheterization for multivessel disease or two-vessel proximal left anterior descending disease often have percutaneous coronary intervention (PCI) in the same session, possibly without input from a multidisciplinary heart team, a new study suggests.
The study, a retrospective analysis of more than 8,000 catheterization procedures in New York State during 2018 and 2019, was published in JACC: Cardiovascular Interventions.
Among the stable patients with multivessel disease or left main (LM) disease who had PCI, 78.4% of PCIs were performed in the same session as their diagnostic catheterization procedure, known as ad hoc PCI, a “surprisingly high rate,” the authors wrote.
The 2011 clinical guidelines in place during the study period advised coronary artery bypass graft (CABG) surgery as a class 1 recommendation for LM disease, whereas PCI is a lower-class recommendation (J Am Coll Cardiol. 2011;58:e44-e-122), they noted.
“Ad hoc PCI can be inadvisable when guidelines indicate that patients can realize better outcomes with CABG surgery,” lead study author Edward L. Hannan, PhD, MS, said in an interview. “The issue is that ad hoc PCI eliminates the opportunity for a multidisciplinary heart team to evaluate the patient.”
Dr. Hannan is principal investigator for the cardiac services program at the New York State Department of Health in Albany and distinguished professor emeritus at the University of Albany School of Public Health.
The researchers analyzed data from two mandatory New York State PCI and cardiac surgery registries, the Percutaneous Coronary Interventions Reporting System and the Cardiac Surgery Reporting System. A total of 91,146 patients had an index PCI from Dec. 1, 2017, to Nov. 30, 2019.
The study included patients who had two-vessel disease with proximal left anterior descending (PLAD) disease, three-vessel disease or unprotected LM disease. Exclusion criteria included a previous revascularization, among a host of other factors. The analysis also identified 10,122 patients who had coronary artery bypass graft (CABG) surgery in addition to the 8,196 patients who had PCI.
The percentage for ad hoc PCI ranged from 58.7% for those with unprotected LM disease to 85.4% for patients with two-vessel PLAD. Among the patients who had PCI for three-vessel disease, 76.7% had an ad hoc PCI.
Selected subgroups had lower ad hoc PCI rates. When patients who had a myocardial infarction within 1-7 days were excluded, the ad hoc percentage decreased slightly to 77.2%. PCI patients with diabetes were also less likely to have ad hoc PCI (75.7% vs. 80.4%, P < .0001), as were patients with compromised left ventricular ejection fraction (< 35%; 64.6% vs. 80.5%, P < .0001).
When all revascularizations – PCI plus CABG – were taken into account, the rate of ad hoc PCIs was 35.1%. Rates were 63.9% for patients with two-vessel PLAD disease, 32.4% for those with three-vessel disease, and 11.5% for patients with unprotected LM disease.
One potential disadvantage of ad hoc PCI, the authors noted, is that it doesn’t allow time for a multidisciplinary heart team to evaluate the patient for a different treatment, such as CABG or medical therapy. “This multidisciplinary team can evaluate all the pros and cons of different approaches, such as PCI vs. CABG surgery in this case,” Dr. Hannan said.
The study findings imply a potential overutilization of PCI and a greater likelihood of forgoing a more appropriate intervention, he said, “given that we have chosen for the study groups of patients who in general benefit more with CABG surgery.”
The results also showed variability in ad hoc PCI rates among hospitals and physicians. “They are large enough to suggest that there is a fairly large variation across the state in the use of heart teams,” he said.
For unprotected LM disease, the risk adjusted rate for hospitals of ad hoc PCIs among all PCIs ranged from 25.6% in the lowest quartile to 93.7% in the highest. Physician rates of ad hoc PCIs for the same indication, which were ranked by tertile, ranged from 22% for the lowest to 84.3% for the highest (P < .001).
One strength of the study, Dr. Hannan said, is that it is a large population-based study that excluded groups for whom an ad hoc PCI would be appropriate, such as emergency patients. One limitation is that it did not account for legitimate reasons for ad hoc PCI, including contraindications for CABG surgery and patient refusal of CABG surgery.
In an invited editorial comment, James C. Blankenship, MD, and Krishna Patel, MD, wrote that this study shows that “past criticisms of ad hoc PCI have had seemingly little effect.”
“The article provides a striking example of a difference between guideline-directed practice and real-life practice,” Dr. Blankenship said in an interview. “Guideline recommendations for the heart team approach are well known by interventionalists, so the findings of this study do not reflect ignorance of cardiologists.” Dr. Blankenship, a cardiologist and professor of medicine at the University of New Mexico in Albuquerque, is a coauthor of the 2011 PCI guidelines.
It’s more likely the study findings “reflect unconscious biases and sincere beliefs of patients and interventionalists that PCI rather than CABG is in patients’ best interests,” Dr. Blankenship said.
He noted the variation in practice across hospitals and individuals suggests an opportunity for improvement. “If the guidelines are correct, then perhaps interventionalists should be held accountable for making sure the heart team approach is followed,” he said. “Alternatively, perhaps a modified approach that guarantees patient-centered decision making and is ethically acceptable could be identified.”
The study received funding from the New York State Department of Health. Dr. Hannan and Dr. Blankenship and Dr. Patel have no relevant disclosures.
Stable patients who have a diagnostic cardiac catheterization for multivessel disease or two-vessel proximal left anterior descending disease often have percutaneous coronary intervention (PCI) in the same session, possibly without input from a multidisciplinary heart team, a new study suggests.
The study, a retrospective analysis of more than 8,000 catheterization procedures in New York State during 2018 and 2019, was published in JACC: Cardiovascular Interventions.
Among the stable patients with multivessel disease or left main (LM) disease who had PCI, 78.4% of PCIs were performed in the same session as their diagnostic catheterization procedure, known as ad hoc PCI, a “surprisingly high rate,” the authors wrote.
The 2011 clinical guidelines in place during the study period advised coronary artery bypass graft (CABG) surgery as a class 1 recommendation for LM disease, whereas PCI is a lower-class recommendation (J Am Coll Cardiol. 2011;58:e44-e-122), they noted.
“Ad hoc PCI can be inadvisable when guidelines indicate that patients can realize better outcomes with CABG surgery,” lead study author Edward L. Hannan, PhD, MS, said in an interview. “The issue is that ad hoc PCI eliminates the opportunity for a multidisciplinary heart team to evaluate the patient.”
Dr. Hannan is principal investigator for the cardiac services program at the New York State Department of Health in Albany and distinguished professor emeritus at the University of Albany School of Public Health.
The researchers analyzed data from two mandatory New York State PCI and cardiac surgery registries, the Percutaneous Coronary Interventions Reporting System and the Cardiac Surgery Reporting System. A total of 91,146 patients had an index PCI from Dec. 1, 2017, to Nov. 30, 2019.
The study included patients who had two-vessel disease with proximal left anterior descending (PLAD) disease, three-vessel disease or unprotected LM disease. Exclusion criteria included a previous revascularization, among a host of other factors. The analysis also identified 10,122 patients who had coronary artery bypass graft (CABG) surgery in addition to the 8,196 patients who had PCI.
The percentage for ad hoc PCI ranged from 58.7% for those with unprotected LM disease to 85.4% for patients with two-vessel PLAD. Among the patients who had PCI for three-vessel disease, 76.7% had an ad hoc PCI.
Selected subgroups had lower ad hoc PCI rates. When patients who had a myocardial infarction within 1-7 days were excluded, the ad hoc percentage decreased slightly to 77.2%. PCI patients with diabetes were also less likely to have ad hoc PCI (75.7% vs. 80.4%, P < .0001), as were patients with compromised left ventricular ejection fraction (< 35%; 64.6% vs. 80.5%, P < .0001).
When all revascularizations – PCI plus CABG – were taken into account, the rate of ad hoc PCIs was 35.1%. Rates were 63.9% for patients with two-vessel PLAD disease, 32.4% for those with three-vessel disease, and 11.5% for patients with unprotected LM disease.
One potential disadvantage of ad hoc PCI, the authors noted, is that it doesn’t allow time for a multidisciplinary heart team to evaluate the patient for a different treatment, such as CABG or medical therapy. “This multidisciplinary team can evaluate all the pros and cons of different approaches, such as PCI vs. CABG surgery in this case,” Dr. Hannan said.
The study findings imply a potential overutilization of PCI and a greater likelihood of forgoing a more appropriate intervention, he said, “given that we have chosen for the study groups of patients who in general benefit more with CABG surgery.”
The results also showed variability in ad hoc PCI rates among hospitals and physicians. “They are large enough to suggest that there is a fairly large variation across the state in the use of heart teams,” he said.
For unprotected LM disease, the risk adjusted rate for hospitals of ad hoc PCIs among all PCIs ranged from 25.6% in the lowest quartile to 93.7% in the highest. Physician rates of ad hoc PCIs for the same indication, which were ranked by tertile, ranged from 22% for the lowest to 84.3% for the highest (P < .001).
One strength of the study, Dr. Hannan said, is that it is a large population-based study that excluded groups for whom an ad hoc PCI would be appropriate, such as emergency patients. One limitation is that it did not account for legitimate reasons for ad hoc PCI, including contraindications for CABG surgery and patient refusal of CABG surgery.
In an invited editorial comment, James C. Blankenship, MD, and Krishna Patel, MD, wrote that this study shows that “past criticisms of ad hoc PCI have had seemingly little effect.”
“The article provides a striking example of a difference between guideline-directed practice and real-life practice,” Dr. Blankenship said in an interview. “Guideline recommendations for the heart team approach are well known by interventionalists, so the findings of this study do not reflect ignorance of cardiologists.” Dr. Blankenship, a cardiologist and professor of medicine at the University of New Mexico in Albuquerque, is a coauthor of the 2011 PCI guidelines.
It’s more likely the study findings “reflect unconscious biases and sincere beliefs of patients and interventionalists that PCI rather than CABG is in patients’ best interests,” Dr. Blankenship said.
He noted the variation in practice across hospitals and individuals suggests an opportunity for improvement. “If the guidelines are correct, then perhaps interventionalists should be held accountable for making sure the heart team approach is followed,” he said. “Alternatively, perhaps a modified approach that guarantees patient-centered decision making and is ethically acceptable could be identified.”
The study received funding from the New York State Department of Health. Dr. Hannan and Dr. Blankenship and Dr. Patel have no relevant disclosures.
Stable patients who have a diagnostic cardiac catheterization for multivessel disease or two-vessel proximal left anterior descending disease often have percutaneous coronary intervention (PCI) in the same session, possibly without input from a multidisciplinary heart team, a new study suggests.
The study, a retrospective analysis of more than 8,000 catheterization procedures in New York State during 2018 and 2019, was published in JACC: Cardiovascular Interventions.
Among the stable patients with multivessel disease or left main (LM) disease who had PCI, 78.4% of PCIs were performed in the same session as their diagnostic catheterization procedure, known as ad hoc PCI, a “surprisingly high rate,” the authors wrote.
The 2011 clinical guidelines in place during the study period advised coronary artery bypass graft (CABG) surgery as a class 1 recommendation for LM disease, whereas PCI is a lower-class recommendation (J Am Coll Cardiol. 2011;58:e44-e-122), they noted.
“Ad hoc PCI can be inadvisable when guidelines indicate that patients can realize better outcomes with CABG surgery,” lead study author Edward L. Hannan, PhD, MS, said in an interview. “The issue is that ad hoc PCI eliminates the opportunity for a multidisciplinary heart team to evaluate the patient.”
Dr. Hannan is principal investigator for the cardiac services program at the New York State Department of Health in Albany and distinguished professor emeritus at the University of Albany School of Public Health.
The researchers analyzed data from two mandatory New York State PCI and cardiac surgery registries, the Percutaneous Coronary Interventions Reporting System and the Cardiac Surgery Reporting System. A total of 91,146 patients had an index PCI from Dec. 1, 2017, to Nov. 30, 2019.
The study included patients who had two-vessel disease with proximal left anterior descending (PLAD) disease, three-vessel disease or unprotected LM disease. Exclusion criteria included a previous revascularization, among a host of other factors. The analysis also identified 10,122 patients who had coronary artery bypass graft (CABG) surgery in addition to the 8,196 patients who had PCI.
The percentage for ad hoc PCI ranged from 58.7% for those with unprotected LM disease to 85.4% for patients with two-vessel PLAD. Among the patients who had PCI for three-vessel disease, 76.7% had an ad hoc PCI.
Selected subgroups had lower ad hoc PCI rates. When patients who had a myocardial infarction within 1-7 days were excluded, the ad hoc percentage decreased slightly to 77.2%. PCI patients with diabetes were also less likely to have ad hoc PCI (75.7% vs. 80.4%, P < .0001), as were patients with compromised left ventricular ejection fraction (< 35%; 64.6% vs. 80.5%, P < .0001).
When all revascularizations – PCI plus CABG – were taken into account, the rate of ad hoc PCIs was 35.1%. Rates were 63.9% for patients with two-vessel PLAD disease, 32.4% for those with three-vessel disease, and 11.5% for patients with unprotected LM disease.
One potential disadvantage of ad hoc PCI, the authors noted, is that it doesn’t allow time for a multidisciplinary heart team to evaluate the patient for a different treatment, such as CABG or medical therapy. “This multidisciplinary team can evaluate all the pros and cons of different approaches, such as PCI vs. CABG surgery in this case,” Dr. Hannan said.
The study findings imply a potential overutilization of PCI and a greater likelihood of forgoing a more appropriate intervention, he said, “given that we have chosen for the study groups of patients who in general benefit more with CABG surgery.”
The results also showed variability in ad hoc PCI rates among hospitals and physicians. “They are large enough to suggest that there is a fairly large variation across the state in the use of heart teams,” he said.
For unprotected LM disease, the risk adjusted rate for hospitals of ad hoc PCIs among all PCIs ranged from 25.6% in the lowest quartile to 93.7% in the highest. Physician rates of ad hoc PCIs for the same indication, which were ranked by tertile, ranged from 22% for the lowest to 84.3% for the highest (P < .001).
One strength of the study, Dr. Hannan said, is that it is a large population-based study that excluded groups for whom an ad hoc PCI would be appropriate, such as emergency patients. One limitation is that it did not account for legitimate reasons for ad hoc PCI, including contraindications for CABG surgery and patient refusal of CABG surgery.
In an invited editorial comment, James C. Blankenship, MD, and Krishna Patel, MD, wrote that this study shows that “past criticisms of ad hoc PCI have had seemingly little effect.”
“The article provides a striking example of a difference between guideline-directed practice and real-life practice,” Dr. Blankenship said in an interview. “Guideline recommendations for the heart team approach are well known by interventionalists, so the findings of this study do not reflect ignorance of cardiologists.” Dr. Blankenship, a cardiologist and professor of medicine at the University of New Mexico in Albuquerque, is a coauthor of the 2011 PCI guidelines.
It’s more likely the study findings “reflect unconscious biases and sincere beliefs of patients and interventionalists that PCI rather than CABG is in patients’ best interests,” Dr. Blankenship said.
He noted the variation in practice across hospitals and individuals suggests an opportunity for improvement. “If the guidelines are correct, then perhaps interventionalists should be held accountable for making sure the heart team approach is followed,” he said. “Alternatively, perhaps a modified approach that guarantees patient-centered decision making and is ethically acceptable could be identified.”
The study received funding from the New York State Department of Health. Dr. Hannan and Dr. Blankenship and Dr. Patel have no relevant disclosures.
FROM JACC; CARDIOVASCULAR INTERVENTIONS
Even exercise by ‘weekend warriors’ can cut CV risk
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.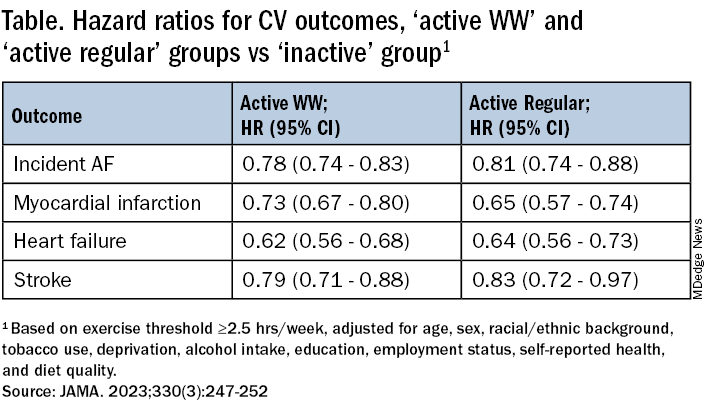
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
FROM JAMA
Rising patient costs tied to private equity ownership
The report was a collaboration of University of California, Berkeley, staff and researchers from two nonprofits, the American Antitrust Institute and the Washington Center for Equitable Growth. It provides “convincing evidence that incentives to put profits before patients have grown stronger with an increase in private equity ownership of physician practices,” lead author Richard Scheffler, PhD, of UC Berkeley said in a statement.
The report also noted that private equity acquisitions of physician groups have risen sixfold in just a decade, increasing from 75 deals in 2012 to 484 deals in 2021.
Separately, the American Medical Association earlier released a separate report on trends in physician practice arrangements, finding that the percentage of physicians working in private equity–owned groups was 4.5% in 2022, the same as in its previous 2020 report. The share of physicians working in private practices fell by 13 percentage points from 60.1% to 46.7% between 2012 and 2022, the AMA reported.
The Berkeley report and the AMA update come amid rising concerns about the effects of the decline of independent physician practices. The U.S. Senate Finance Committee, which oversees most federal health spending, held a June hearing examining the causes and consequences of increased corporate ownership in health care, including a look at physician practices.
“It’s increasingly clear that consolidation in health care is not lowering costs or increasing the quality of Americans’ health care,” Senate Finance Chairman Ron Wyden (D-Ore.) said in an email. “For private equity in health care in particular, there needs to be more transparency around ownership so the effect on these business relationships can be better understood.”
Federal and state agencies do not generally track acquisitions of physician practices.
The UC Berkeley report impressively documents the rising influence of private equity in health care, for which it’s tough to find good data, said Karen Joynt Maddox, MD, MPH, of Washington University in St. Louis. Dr. Maddox, a cardiologist and policy researcher who also has studied the effects of consolidation in health care, examined the new report at the request of this news organization.
“They did a great job with the data,” Dr. Maddox said. “One of the big issues around private equity, and in general, ‘corporatization’ and consolidation of health care, is that there’s not a great way to track ownership changes. It’s really difficult to study.”
Dr. Scheffler and colleagues used data from the commercial firm PitchBook to identify acquisitions of physician practices by private equity firms. They consulted IQVIA’s physician databases – OneKey and SK&A Office-Based Physicians Database – to learn about the location, size, and specialties of acquired practices. They also used data from the nonprofit Health Care Cost Institute, which tracks commercial health plan claims, to assess how private equity acquisitions affected prices.
The researchers then matched the findings for practices acquired by private equity firms from 2015 to 2021 against those for comparable physician practices that remained independent from 2012 to 2021.
The authors then tied private-equity ownership to the following price increases:
- Gastroenterology (14%; 95% confidence interval, 7.9%-20.4%
- Oncology (16.4%; 95% CI, 5.5%-28.4%)
- Dermatology (4.0%; 95% CI, 1%-7.1%)
- Ob.gyn. (8.8%; 95% CI, 3.8%-14%)
- Ophthalmology (8.7%; 95% CI, 5.1%-12.3%)
- Radiology (8.2%; 95% CI, 0.8%-16.1%)
- Orthopedics (7.1%; 95% CI, 2.2%-12.3%)
- Primary care (4.1%; 95% CI, 1.3%-7%)
The analysis also found higher prices for cardiology (8.7%; 95% CI, –6.4% to 26.1%) and urology (4.2%; 95% CI, –2.3% to 11.1%), but neither of these findings was statistically significant, one of the authors, Daniel R. Arnold, PhD, of UC Berkeley, said in an email. This was most likely caused by smaller sample sizes for these fields.
Factors driving consolidation
The two reports and the Senate Finance consolidation hearing raised similar issues, including calls to look at the factors driving more physicians out of independent practice, including Medicare reimbursement that may not keep up with rising inflation.
The Berkeley report authors called for Congress to add a broad inflation component to the Medicare physician fee schedule. It also called on Congress to add cases where Medicare, the biggest U.S. purchaser of health care, pays less for services when performed in independent practices than in hospital-affiliated ones.
Shawn Martin, executive vice president and CEO of the American Academy of Family Physicians, said his group appreciates how the report from UC Berkeley and nonprofit groups echoed recommendations many clinicians have made, including the call for a broad inflation adjustment for the fee schedule.
“To move the needle forward, Congress must advance site-neutral payment policies while also addressing the administrative requirements that take physicians away from the important work of caring for patients,” Mr. Martin said in an email.
Arnold Ventures provided funding for the report, which was a joint project of the American Antitrust Institute, the Nicholas C. Petris Center on Health Care Markets and Consumer Welfare, UC Berkeley, and the Washington Center for Equitable Growth.
A version of this article appeared on Medscape.com.
The report was a collaboration of University of California, Berkeley, staff and researchers from two nonprofits, the American Antitrust Institute and the Washington Center for Equitable Growth. It provides “convincing evidence that incentives to put profits before patients have grown stronger with an increase in private equity ownership of physician practices,” lead author Richard Scheffler, PhD, of UC Berkeley said in a statement.
The report also noted that private equity acquisitions of physician groups have risen sixfold in just a decade, increasing from 75 deals in 2012 to 484 deals in 2021.
Separately, the American Medical Association earlier released a separate report on trends in physician practice arrangements, finding that the percentage of physicians working in private equity–owned groups was 4.5% in 2022, the same as in its previous 2020 report. The share of physicians working in private practices fell by 13 percentage points from 60.1% to 46.7% between 2012 and 2022, the AMA reported.
The Berkeley report and the AMA update come amid rising concerns about the effects of the decline of independent physician practices. The U.S. Senate Finance Committee, which oversees most federal health spending, held a June hearing examining the causes and consequences of increased corporate ownership in health care, including a look at physician practices.
“It’s increasingly clear that consolidation in health care is not lowering costs or increasing the quality of Americans’ health care,” Senate Finance Chairman Ron Wyden (D-Ore.) said in an email. “For private equity in health care in particular, there needs to be more transparency around ownership so the effect on these business relationships can be better understood.”
Federal and state agencies do not generally track acquisitions of physician practices.
The UC Berkeley report impressively documents the rising influence of private equity in health care, for which it’s tough to find good data, said Karen Joynt Maddox, MD, MPH, of Washington University in St. Louis. Dr. Maddox, a cardiologist and policy researcher who also has studied the effects of consolidation in health care, examined the new report at the request of this news organization.
“They did a great job with the data,” Dr. Maddox said. “One of the big issues around private equity, and in general, ‘corporatization’ and consolidation of health care, is that there’s not a great way to track ownership changes. It’s really difficult to study.”
Dr. Scheffler and colleagues used data from the commercial firm PitchBook to identify acquisitions of physician practices by private equity firms. They consulted IQVIA’s physician databases – OneKey and SK&A Office-Based Physicians Database – to learn about the location, size, and specialties of acquired practices. They also used data from the nonprofit Health Care Cost Institute, which tracks commercial health plan claims, to assess how private equity acquisitions affected prices.
The researchers then matched the findings for practices acquired by private equity firms from 2015 to 2021 against those for comparable physician practices that remained independent from 2012 to 2021.
The authors then tied private-equity ownership to the following price increases:
- Gastroenterology (14%; 95% confidence interval, 7.9%-20.4%
- Oncology (16.4%; 95% CI, 5.5%-28.4%)
- Dermatology (4.0%; 95% CI, 1%-7.1%)
- Ob.gyn. (8.8%; 95% CI, 3.8%-14%)
- Ophthalmology (8.7%; 95% CI, 5.1%-12.3%)
- Radiology (8.2%; 95% CI, 0.8%-16.1%)
- Orthopedics (7.1%; 95% CI, 2.2%-12.3%)
- Primary care (4.1%; 95% CI, 1.3%-7%)
The analysis also found higher prices for cardiology (8.7%; 95% CI, –6.4% to 26.1%) and urology (4.2%; 95% CI, –2.3% to 11.1%), but neither of these findings was statistically significant, one of the authors, Daniel R. Arnold, PhD, of UC Berkeley, said in an email. This was most likely caused by smaller sample sizes for these fields.
Factors driving consolidation
The two reports and the Senate Finance consolidation hearing raised similar issues, including calls to look at the factors driving more physicians out of independent practice, including Medicare reimbursement that may not keep up with rising inflation.
The Berkeley report authors called for Congress to add a broad inflation component to the Medicare physician fee schedule. It also called on Congress to add cases where Medicare, the biggest U.S. purchaser of health care, pays less for services when performed in independent practices than in hospital-affiliated ones.
Shawn Martin, executive vice president and CEO of the American Academy of Family Physicians, said his group appreciates how the report from UC Berkeley and nonprofit groups echoed recommendations many clinicians have made, including the call for a broad inflation adjustment for the fee schedule.
“To move the needle forward, Congress must advance site-neutral payment policies while also addressing the administrative requirements that take physicians away from the important work of caring for patients,” Mr. Martin said in an email.
Arnold Ventures provided funding for the report, which was a joint project of the American Antitrust Institute, the Nicholas C. Petris Center on Health Care Markets and Consumer Welfare, UC Berkeley, and the Washington Center for Equitable Growth.
A version of this article appeared on Medscape.com.
The report was a collaboration of University of California, Berkeley, staff and researchers from two nonprofits, the American Antitrust Institute and the Washington Center for Equitable Growth. It provides “convincing evidence that incentives to put profits before patients have grown stronger with an increase in private equity ownership of physician practices,” lead author Richard Scheffler, PhD, of UC Berkeley said in a statement.
The report also noted that private equity acquisitions of physician groups have risen sixfold in just a decade, increasing from 75 deals in 2012 to 484 deals in 2021.
Separately, the American Medical Association earlier released a separate report on trends in physician practice arrangements, finding that the percentage of physicians working in private equity–owned groups was 4.5% in 2022, the same as in its previous 2020 report. The share of physicians working in private practices fell by 13 percentage points from 60.1% to 46.7% between 2012 and 2022, the AMA reported.
The Berkeley report and the AMA update come amid rising concerns about the effects of the decline of independent physician practices. The U.S. Senate Finance Committee, which oversees most federal health spending, held a June hearing examining the causes and consequences of increased corporate ownership in health care, including a look at physician practices.
“It’s increasingly clear that consolidation in health care is not lowering costs or increasing the quality of Americans’ health care,” Senate Finance Chairman Ron Wyden (D-Ore.) said in an email. “For private equity in health care in particular, there needs to be more transparency around ownership so the effect on these business relationships can be better understood.”
Federal and state agencies do not generally track acquisitions of physician practices.
The UC Berkeley report impressively documents the rising influence of private equity in health care, for which it’s tough to find good data, said Karen Joynt Maddox, MD, MPH, of Washington University in St. Louis. Dr. Maddox, a cardiologist and policy researcher who also has studied the effects of consolidation in health care, examined the new report at the request of this news organization.
“They did a great job with the data,” Dr. Maddox said. “One of the big issues around private equity, and in general, ‘corporatization’ and consolidation of health care, is that there’s not a great way to track ownership changes. It’s really difficult to study.”
Dr. Scheffler and colleagues used data from the commercial firm PitchBook to identify acquisitions of physician practices by private equity firms. They consulted IQVIA’s physician databases – OneKey and SK&A Office-Based Physicians Database – to learn about the location, size, and specialties of acquired practices. They also used data from the nonprofit Health Care Cost Institute, which tracks commercial health plan claims, to assess how private equity acquisitions affected prices.
The researchers then matched the findings for practices acquired by private equity firms from 2015 to 2021 against those for comparable physician practices that remained independent from 2012 to 2021.
The authors then tied private-equity ownership to the following price increases:
- Gastroenterology (14%; 95% confidence interval, 7.9%-20.4%
- Oncology (16.4%; 95% CI, 5.5%-28.4%)
- Dermatology (4.0%; 95% CI, 1%-7.1%)
- Ob.gyn. (8.8%; 95% CI, 3.8%-14%)
- Ophthalmology (8.7%; 95% CI, 5.1%-12.3%)
- Radiology (8.2%; 95% CI, 0.8%-16.1%)
- Orthopedics (7.1%; 95% CI, 2.2%-12.3%)
- Primary care (4.1%; 95% CI, 1.3%-7%)
The analysis also found higher prices for cardiology (8.7%; 95% CI, –6.4% to 26.1%) and urology (4.2%; 95% CI, –2.3% to 11.1%), but neither of these findings was statistically significant, one of the authors, Daniel R. Arnold, PhD, of UC Berkeley, said in an email. This was most likely caused by smaller sample sizes for these fields.
Factors driving consolidation
The two reports and the Senate Finance consolidation hearing raised similar issues, including calls to look at the factors driving more physicians out of independent practice, including Medicare reimbursement that may not keep up with rising inflation.
The Berkeley report authors called for Congress to add a broad inflation component to the Medicare physician fee schedule. It also called on Congress to add cases where Medicare, the biggest U.S. purchaser of health care, pays less for services when performed in independent practices than in hospital-affiliated ones.
Shawn Martin, executive vice president and CEO of the American Academy of Family Physicians, said his group appreciates how the report from UC Berkeley and nonprofit groups echoed recommendations many clinicians have made, including the call for a broad inflation adjustment for the fee schedule.
“To move the needle forward, Congress must advance site-neutral payment policies while also addressing the administrative requirements that take physicians away from the important work of caring for patients,” Mr. Martin said in an email.
Arnold Ventures provided funding for the report, which was a joint project of the American Antitrust Institute, the Nicholas C. Petris Center on Health Care Markets and Consumer Welfare, UC Berkeley, and the Washington Center for Equitable Growth.
A version of this article appeared on Medscape.com.
Omega-3s and AFib: No added risk from eating fish but high-dose supplement questions persist
Regular consumption of fish and other foods rich in omega-3 fatty acids (FA) won’t raise an individual’s risk for developing atrial fibrillation (AFib), suggests a meta-analysis of population-based studies.
The finding may alleviate recent concerns about higher-dose omega-3 FA supplement intake in clinical-trial patients at elevated cardiovascular (CV) risk, researchers say.
Indeed, across the 17 cohort studies in the meta-analysis, risk for incident AFib was unaffected by elevated circulating and adipose tissue levels of eicosapentaenoic acid (EPA) from dietary intake. Moreover, the risk appeared to drop significantly with such levels of docosahexaenoic acid (DHA), docosapentaenoic acid (DPA), and EPA plus DHA.
The signals of AFib risk associated with high-dose omega-3 FA in supplements or prescription form in some clinical trials “may not necessarily be generalizable to lower-dose habitual dietary omega-3 intakes,” concludes the study’s report published in the Journal of the American College of Cardiology.
Other recent research suggests that any elevated AFib risk from omega-3 FA intake is dose-related and may be associated with omega-3 FA supplement or medication intake in high doses, such as 4 g/day.
“Coupled with the more consistent benefits of these fatty acids in the prevention of adverse coronary events, our study suggests that current dietary guidelines recommending fish/omega-3 fatty acid consumption should be maintained,” conclude the authors of the report, led by Frank Qian, MD, MPH, Harvard T.H. Chan School of Public Health and Beth Israel Deaconess Medical Center, Boston.
The current study is “important in showing that physiologic levels of omega-3s that would accumulate through diet don’t seem to increase the risk of arrhythmia,” preventive cardiologist Sean Heffron, MD, NYU Langone Health and Grossman School of Medicine, told this news organization.
“It also lends credence to the fact that the increased risk is specific to the high dose supplementation, because that’s the only instance in which we’ve seen increased atrial fibrillation in association with omega-3s,” said Dr. Heffron, who wasn’t involved in the meta-analysis.
An accompanying editorial agrees. “Based on present evidence, moderate dietary intake of fish and seafood is unlikely to achieve sufficiently high levels of omega-3-FAs in blood or tissue that would result in increased AFib risk as observed in clinical trials of fish oil supplements and high-dose prescriptions,” write Christie Ballantyne, MD, and Xiaoming Jia, MD, Baylor College of Medicine, Houston.
Therefore, they conclude, “fish should continue to be an important part of the menu of a heart-healthy diet.”
The meta-analysis comprised 54,799 participants from 21 countries worldwide in 17 prospective cohort studies that yielded data on incident AFib, in which there were 7,720 cases of the arrhythmia over a median follow-up of 13 years. It looked at associations between such cases and levels of omega-3 FA in blood and adipose tissue samples.
In multivariable analysis, EPA levels were not associated with incident AFib, with a hazard ratio of 1.00 (95% confidence interval, 0.95-1.05) per interquintile range, which the report describes as the difference between the 90th and 10th percentiles.
In contrast, levels of DPA, DHA, and EPA plus DHA were all associated with reduced AFib incidence at interquintile-range HRs of 0.89 (95% CI, 0.83-0.95) for DPA, 0.90 (95% CI, 0.85-0.96) for DHA and 0.93 (95% CI, 0.87-0.99) for EPA and DHA combined.
“We found little evidence that the associations significantly varied by age, sex, or global region, or across the various lipid compartments,” the report states. “Moreover, the relationship between omega-3 fatty acids and AFib did not significantly differ among individuals at higher CV risk.”
The authors observe that the prevalence of omega-3 FA supplement use in the cohorts was very low, suggesting that the omega-3 FA biomarker levels largely reflected habitual dietary intake.
Most of the meta-analysis population were free of CV disease or at relatively low CV risk, they write, and “it is conceivable that the effects of omega-3 fatty acids on atrial arrhythmias may differ in those with existing CV disease versus without.”
However, they note, in a prespecified subgroup analysis of participants mirroring the REDUCE-IT cohort of people with established CV disease or at elevated CV risk, no association with incident AFib was observed for EPA and inverse associations emerged for DPA, DHA, and EPA plus DHA.
In their editorial, Dr. Ballantyne and Dr. Jia say the meta-analysis “represents the largest epidemiological study assessing laboratory-measured omega-3 fatty acid concentrations and AFib risk in the general population.”
But significant heterogeneity across the studies and their populations is a major limitation of the analysis, they write, and made for differences in protocols, sample preparation, outcomes ascertainment, follow-up time, and other variables.
“Despite a rigorous approach to harmonize the data across cohorts and adjusting for multiple confounders,” note Dr. Ballantyne and Dr. Jia, “observational studies always have potential for residual confounders.”
The findings support fish consumption as heart-healthy, they write, but “clinicians should be aware of and discuss with patients the risks versus benefits when prescribing high-dose omega-3 FA therapies.”
The Fatty Acid Research Institute retrospectively provided a small honorarium to a subset of the analysts who participated in this study, but it had no role in its design, analysis, manuscript writing, or decision to submit for publication, the report states. Dr. Ballantyne received grant/research support through his institution from Akcea, Amgen, Arrowhead, Esperion, Ionis, Merck, Novartis, and Regeneron, as well as consulting fees from Alnylam Pharmaceuticals, Althera, Amarin, Amgen, Arrowhead, AstraZeneca, Esperion, Genentech, Gilead, Matinas BioPharma, Merck, New Amsterdam, Novartis, Pfizer, and Regeneron. Dr. Heffron and Dr. Jia have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Regular consumption of fish and other foods rich in omega-3 fatty acids (FA) won’t raise an individual’s risk for developing atrial fibrillation (AFib), suggests a meta-analysis of population-based studies.
The finding may alleviate recent concerns about higher-dose omega-3 FA supplement intake in clinical-trial patients at elevated cardiovascular (CV) risk, researchers say.
Indeed, across the 17 cohort studies in the meta-analysis, risk for incident AFib was unaffected by elevated circulating and adipose tissue levels of eicosapentaenoic acid (EPA) from dietary intake. Moreover, the risk appeared to drop significantly with such levels of docosahexaenoic acid (DHA), docosapentaenoic acid (DPA), and EPA plus DHA.
The signals of AFib risk associated with high-dose omega-3 FA in supplements or prescription form in some clinical trials “may not necessarily be generalizable to lower-dose habitual dietary omega-3 intakes,” concludes the study’s report published in the Journal of the American College of Cardiology.
Other recent research suggests that any elevated AFib risk from omega-3 FA intake is dose-related and may be associated with omega-3 FA supplement or medication intake in high doses, such as 4 g/day.
“Coupled with the more consistent benefits of these fatty acids in the prevention of adverse coronary events, our study suggests that current dietary guidelines recommending fish/omega-3 fatty acid consumption should be maintained,” conclude the authors of the report, led by Frank Qian, MD, MPH, Harvard T.H. Chan School of Public Health and Beth Israel Deaconess Medical Center, Boston.
The current study is “important in showing that physiologic levels of omega-3s that would accumulate through diet don’t seem to increase the risk of arrhythmia,” preventive cardiologist Sean Heffron, MD, NYU Langone Health and Grossman School of Medicine, told this news organization.
“It also lends credence to the fact that the increased risk is specific to the high dose supplementation, because that’s the only instance in which we’ve seen increased atrial fibrillation in association with omega-3s,” said Dr. Heffron, who wasn’t involved in the meta-analysis.
An accompanying editorial agrees. “Based on present evidence, moderate dietary intake of fish and seafood is unlikely to achieve sufficiently high levels of omega-3-FAs in blood or tissue that would result in increased AFib risk as observed in clinical trials of fish oil supplements and high-dose prescriptions,” write Christie Ballantyne, MD, and Xiaoming Jia, MD, Baylor College of Medicine, Houston.
Therefore, they conclude, “fish should continue to be an important part of the menu of a heart-healthy diet.”
The meta-analysis comprised 54,799 participants from 21 countries worldwide in 17 prospective cohort studies that yielded data on incident AFib, in which there were 7,720 cases of the arrhythmia over a median follow-up of 13 years. It looked at associations between such cases and levels of omega-3 FA in blood and adipose tissue samples.
In multivariable analysis, EPA levels were not associated with incident AFib, with a hazard ratio of 1.00 (95% confidence interval, 0.95-1.05) per interquintile range, which the report describes as the difference between the 90th and 10th percentiles.
In contrast, levels of DPA, DHA, and EPA plus DHA were all associated with reduced AFib incidence at interquintile-range HRs of 0.89 (95% CI, 0.83-0.95) for DPA, 0.90 (95% CI, 0.85-0.96) for DHA and 0.93 (95% CI, 0.87-0.99) for EPA and DHA combined.
“We found little evidence that the associations significantly varied by age, sex, or global region, or across the various lipid compartments,” the report states. “Moreover, the relationship between omega-3 fatty acids and AFib did not significantly differ among individuals at higher CV risk.”
The authors observe that the prevalence of omega-3 FA supplement use in the cohorts was very low, suggesting that the omega-3 FA biomarker levels largely reflected habitual dietary intake.
Most of the meta-analysis population were free of CV disease or at relatively low CV risk, they write, and “it is conceivable that the effects of omega-3 fatty acids on atrial arrhythmias may differ in those with existing CV disease versus without.”
However, they note, in a prespecified subgroup analysis of participants mirroring the REDUCE-IT cohort of people with established CV disease or at elevated CV risk, no association with incident AFib was observed for EPA and inverse associations emerged for DPA, DHA, and EPA plus DHA.
In their editorial, Dr. Ballantyne and Dr. Jia say the meta-analysis “represents the largest epidemiological study assessing laboratory-measured omega-3 fatty acid concentrations and AFib risk in the general population.”
But significant heterogeneity across the studies and their populations is a major limitation of the analysis, they write, and made for differences in protocols, sample preparation, outcomes ascertainment, follow-up time, and other variables.
“Despite a rigorous approach to harmonize the data across cohorts and adjusting for multiple confounders,” note Dr. Ballantyne and Dr. Jia, “observational studies always have potential for residual confounders.”
The findings support fish consumption as heart-healthy, they write, but “clinicians should be aware of and discuss with patients the risks versus benefits when prescribing high-dose omega-3 FA therapies.”
The Fatty Acid Research Institute retrospectively provided a small honorarium to a subset of the analysts who participated in this study, but it had no role in its design, analysis, manuscript writing, or decision to submit for publication, the report states. Dr. Ballantyne received grant/research support through his institution from Akcea, Amgen, Arrowhead, Esperion, Ionis, Merck, Novartis, and Regeneron, as well as consulting fees from Alnylam Pharmaceuticals, Althera, Amarin, Amgen, Arrowhead, AstraZeneca, Esperion, Genentech, Gilead, Matinas BioPharma, Merck, New Amsterdam, Novartis, Pfizer, and Regeneron. Dr. Heffron and Dr. Jia have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Regular consumption of fish and other foods rich in omega-3 fatty acids (FA) won’t raise an individual’s risk for developing atrial fibrillation (AFib), suggests a meta-analysis of population-based studies.
The finding may alleviate recent concerns about higher-dose omega-3 FA supplement intake in clinical-trial patients at elevated cardiovascular (CV) risk, researchers say.
Indeed, across the 17 cohort studies in the meta-analysis, risk for incident AFib was unaffected by elevated circulating and adipose tissue levels of eicosapentaenoic acid (EPA) from dietary intake. Moreover, the risk appeared to drop significantly with such levels of docosahexaenoic acid (DHA), docosapentaenoic acid (DPA), and EPA plus DHA.
The signals of AFib risk associated with high-dose omega-3 FA in supplements or prescription form in some clinical trials “may not necessarily be generalizable to lower-dose habitual dietary omega-3 intakes,” concludes the study’s report published in the Journal of the American College of Cardiology.
Other recent research suggests that any elevated AFib risk from omega-3 FA intake is dose-related and may be associated with omega-3 FA supplement or medication intake in high doses, such as 4 g/day.
“Coupled with the more consistent benefits of these fatty acids in the prevention of adverse coronary events, our study suggests that current dietary guidelines recommending fish/omega-3 fatty acid consumption should be maintained,” conclude the authors of the report, led by Frank Qian, MD, MPH, Harvard T.H. Chan School of Public Health and Beth Israel Deaconess Medical Center, Boston.
The current study is “important in showing that physiologic levels of omega-3s that would accumulate through diet don’t seem to increase the risk of arrhythmia,” preventive cardiologist Sean Heffron, MD, NYU Langone Health and Grossman School of Medicine, told this news organization.
“It also lends credence to the fact that the increased risk is specific to the high dose supplementation, because that’s the only instance in which we’ve seen increased atrial fibrillation in association with omega-3s,” said Dr. Heffron, who wasn’t involved in the meta-analysis.
An accompanying editorial agrees. “Based on present evidence, moderate dietary intake of fish and seafood is unlikely to achieve sufficiently high levels of omega-3-FAs in blood or tissue that would result in increased AFib risk as observed in clinical trials of fish oil supplements and high-dose prescriptions,” write Christie Ballantyne, MD, and Xiaoming Jia, MD, Baylor College of Medicine, Houston.
Therefore, they conclude, “fish should continue to be an important part of the menu of a heart-healthy diet.”
The meta-analysis comprised 54,799 participants from 21 countries worldwide in 17 prospective cohort studies that yielded data on incident AFib, in which there were 7,720 cases of the arrhythmia over a median follow-up of 13 years. It looked at associations between such cases and levels of omega-3 FA in blood and adipose tissue samples.
In multivariable analysis, EPA levels were not associated with incident AFib, with a hazard ratio of 1.00 (95% confidence interval, 0.95-1.05) per interquintile range, which the report describes as the difference between the 90th and 10th percentiles.
In contrast, levels of DPA, DHA, and EPA plus DHA were all associated with reduced AFib incidence at interquintile-range HRs of 0.89 (95% CI, 0.83-0.95) for DPA, 0.90 (95% CI, 0.85-0.96) for DHA and 0.93 (95% CI, 0.87-0.99) for EPA and DHA combined.
“We found little evidence that the associations significantly varied by age, sex, or global region, or across the various lipid compartments,” the report states. “Moreover, the relationship between omega-3 fatty acids and AFib did not significantly differ among individuals at higher CV risk.”
The authors observe that the prevalence of omega-3 FA supplement use in the cohorts was very low, suggesting that the omega-3 FA biomarker levels largely reflected habitual dietary intake.
Most of the meta-analysis population were free of CV disease or at relatively low CV risk, they write, and “it is conceivable that the effects of omega-3 fatty acids on atrial arrhythmias may differ in those with existing CV disease versus without.”
However, they note, in a prespecified subgroup analysis of participants mirroring the REDUCE-IT cohort of people with established CV disease or at elevated CV risk, no association with incident AFib was observed for EPA and inverse associations emerged for DPA, DHA, and EPA plus DHA.
In their editorial, Dr. Ballantyne and Dr. Jia say the meta-analysis “represents the largest epidemiological study assessing laboratory-measured omega-3 fatty acid concentrations and AFib risk in the general population.”
But significant heterogeneity across the studies and their populations is a major limitation of the analysis, they write, and made for differences in protocols, sample preparation, outcomes ascertainment, follow-up time, and other variables.
“Despite a rigorous approach to harmonize the data across cohorts and adjusting for multiple confounders,” note Dr. Ballantyne and Dr. Jia, “observational studies always have potential for residual confounders.”
The findings support fish consumption as heart-healthy, they write, but “clinicians should be aware of and discuss with patients the risks versus benefits when prescribing high-dose omega-3 FA therapies.”
The Fatty Acid Research Institute retrospectively provided a small honorarium to a subset of the analysts who participated in this study, but it had no role in its design, analysis, manuscript writing, or decision to submit for publication, the report states. Dr. Ballantyne received grant/research support through his institution from Akcea, Amgen, Arrowhead, Esperion, Ionis, Merck, Novartis, and Regeneron, as well as consulting fees from Alnylam Pharmaceuticals, Althera, Amarin, Amgen, Arrowhead, AstraZeneca, Esperion, Genentech, Gilead, Matinas BioPharma, Merck, New Amsterdam, Novartis, Pfizer, and Regeneron. Dr. Heffron and Dr. Jia have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Nurse practitioners sue state over right to use ‘doctor’ title
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.
, saying it violates their first amendment right to use the honorific title without fear of regulatory repercussions.
The case highlights ongoing scope-creep battles as the American Medical Association tries to preserve the physician-led team model and nursing organizations and some lawmakers push for greater autonomy for allied professionals.
In the complaint filed in district court in June, plaintiffs Jacqueline Palmer, DNP, Heather Lewis, DNP, and Rodolfo Jaravata-Hanson, DNP, say they fear the state will sanction them. They note that “Doctor Sarah,” another DNP, was fined nearly $20,000 by the state last November for false advertising and fraud after using the moniker in her online advertising and social media accounts.
The fine was part of a settlement that the DNP, Sarah Erny, reached with the state to resolve allegations that she failed to identify her supervising physician and inform the public that she was not a medical doctor.
Under California’s Medical Practice Act, individuals cannot refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Instead, nurse practitioners certified by the California Board of Registered Nursing may use titles like “Certified Nurse Practitioner” and “Advanced Practice Registered Nurse,” corresponding letters such as APRN-CNP, RN, and NP, and phrases like pediatric nurse practitioner to identify specialization.
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
The nonprofit Pacific Legal Foundation represents the plaintiffs. In court records, its attorneys argue that after “years earning their advanced degrees and qualifications ... they should be able to speak truthfully about them in their workplaces, on their business cards, the Internet, and social media, so long as they clarify that they are nurse practitioners.”
State lawmakers’ attempts to clarify the roles of physicians and nurse practitioners have seen mixed results. Florida legislators recently passed a bill to prevent advanced practice nurses from using the honorific title, reserving it only for MDs and DOs. Gov. Ron DeSantis vetoed it last month.
In May, Georgia lawmakers passed the Health Care Practitioners Truth and Transparency Act. It requires advanced practice nurses and physician assistants with doctoral degrees who refer to themselves as doctors in a clinical setting to state they are not medical doctors or physicians.
Still, some health professionals say that the designation should only be used in academic settings or among peers, and that all doctoral degree holders should ditch the moniker at the bedside to ease patient communications.
Named as defendants in the suit are three state officials: California Attorney General Rob Bonta, state Medical Board President Kristina Lawson, and California Board of Registered Nursing Executive Officer Loretta Melby.
A version of this article first appeared on Medscape.com.



