User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
How well do JAK inhibitors work for atopic dermatitis?
largely because of the heterogeneous nature of the disease.
“Atopic dermatitis patients have different complaints,” Jacob P. Thyssen, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Some of them have repeated infections. Some have psychiatric symptoms. Others have widespread eczema. When you talk about how well they work, it really depends on what aspects of AD, what subgroups of AD, and how well they work with comorbidities of AD.”
Baricitinib, a JAK1/JAK2 inhibitor in 2-mg and 4-mg tablets, is available in the European Union, and is under Food and Drug Administration review for AD in the United States. Two JAK1 inhibitors continue to be evaluated in AD clinical trials and are also under FDA review for AD: abrocitinib (100 mg and 200 mg) and upadacitinib (15 mg and 30 mg). None of these agents have been tested in head-to-head trials and only one (abrocitinib) has been compared with the interleukin-4 receptor–alpha antagonist dupilumab, which makes meaningful direct comparisons impossible. (Baricitinib and upadacitinib are approved for treating RA in the United States.)
In his informal assessment from clinical trial data of how these three JAK inhibitors compare with the biologic agents dupilumab and tralokinumab, with potency as an indication, Dr. Thyssen, professor of dermatology at the University of Copenhagen, observed that abrocitinib and dupilumab “are somewhere in the middle,” tralokinumab and baricitinib are “slightly weaker,” while upadacitinib is “very potent.” (Dupilumab is approved by the FDA for treating AD ages 6 and older, and tralokinumab, a fully human monoclonal antibody that binds to IL-13, is under FDA review for AD.)
However, he cautioned that making direct comparisons of these drugs is limited by differences in clinical trial designs, trial length, severity of disease at baseline, and demographics. “Placebo effects also differ between trials, and the speed of onset is different between JAK inhibitors and biologic agents. Because of this, efficacy can be difficult to assess over 12-16 weeks. That’s why long-term studies are necessary.”
It’s also tricky to compare safety signals with baricitinib, abrocitinib, and upadacitinib, “because some of them are JAK1 inhibitors; others are JAK1/JAK2 inhibitors,” he continued. “Even the molecules that inhibit JAK1 are different, so making a comparison between abrocitinib and upadacitinib requires studies that do this is in the best way and over a long period of time.”
Safety signals
Common safety signals in this drug class include nasopharyngitis, nausea, and headache. “Many of these are short lasting, meaning that patients will perhaps have a headache for a day or two and then it will be over,” said Dr. Thyssen, who is also a consultant dermatologist at Bispebjerg Hospital in Copenhagen. “This means that even though we see high proportions of safety signals, this is probably not going to limit the use of JAK inhibitors in most of our patients. Then we have an acne signal in higher proportions for abrocitinib and upadacitinib than for baricitinib, so perhaps this is related to the potency.”
There is also an increased risk for infections, including herpes zoster. “Is this a class effect?” he asked. “We see quite a bit for baricitinib, particularly when it’s used for rheumatoid arthritis. We also see it in AD patients, but we don’t know to what degree yet. We need the real-world evidence before we can make any conclusions.” Routine blood monitoring tests are also required in patients taking JAK inhibitors, because of the risk for leukopenia and effects on liver enzymes.
Then there’s the risk of deep vein thrombosis/pulmonary embolism. “This is mostly linked to baricitinib use, but is this a class effect or is it specific to baricitinib?” he asked. “We’ll have to wait and see, but I think overall, this is not something I have great fear of because we see that AD patients are young, usually with a normal [body mass index], at least in Europe. But we have to study this closely.”
From a clinical standpoint, JAK1/2 inhibitors work well on every measurable aspect of AD, he said, including eczema severity, itch, skin pain, sleep, and quality of life. “Based on conference abstracts and publications, they seem to work equally well independent of race, BMI, atopy status, age, and whether their AD is extrinsic or intrinsic,” Dr. Thyssen added. “One thing we haven’t learned from the companies is, what patients have the highest likelihood of getting a good treatment response? We don’t have good biomarkers yet, but anything the companies can do to help us identify the patients with the greatest chance of success would be so welcome.”
The best available data suggest that JAK inhibitors benefit AD patients with certain comorbidities, including inflammatory bowel disease (with upadacitinib), RA (with both baricitinib and upadacitinib), and alopecia areata (with baricitinib). “These drugs also have been shown to work well for the psychiatric symptoms of disease,” he said.
“As for patients with type 2 inflammation in the airways such as asthma and rhinitis, dupilumab works, but do the JAK inhibitors work? It’s possible from a mode of action standpoint, but we don’t know.” It also remains unclear how JAK inhibitors will fare in the treatment of chronic hand eczema and ocular surface disease, like allergic conjunctivitis, he said.
Despite the unknowns, Dr. Thyssen emphasized the promise that JAK inhibitors hold for AD patients. “We know they provide good AD control,” he said. “For some, like baricitinib, you may need to instruct the patient to use topical corticosteroids as well, but this does not seem to be necessary for upadacitinib and abrocitinib. You really have a single bullet here that will take away most of the problems for many patients, with very fast onset of action, which is important for our patients.”
Dr. Thyssen disclosed that he is a speaker, advisory board member, and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
largely because of the heterogeneous nature of the disease.
“Atopic dermatitis patients have different complaints,” Jacob P. Thyssen, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Some of them have repeated infections. Some have psychiatric symptoms. Others have widespread eczema. When you talk about how well they work, it really depends on what aspects of AD, what subgroups of AD, and how well they work with comorbidities of AD.”
Baricitinib, a JAK1/JAK2 inhibitor in 2-mg and 4-mg tablets, is available in the European Union, and is under Food and Drug Administration review for AD in the United States. Two JAK1 inhibitors continue to be evaluated in AD clinical trials and are also under FDA review for AD: abrocitinib (100 mg and 200 mg) and upadacitinib (15 mg and 30 mg). None of these agents have been tested in head-to-head trials and only one (abrocitinib) has been compared with the interleukin-4 receptor–alpha antagonist dupilumab, which makes meaningful direct comparisons impossible. (Baricitinib and upadacitinib are approved for treating RA in the United States.)
In his informal assessment from clinical trial data of how these three JAK inhibitors compare with the biologic agents dupilumab and tralokinumab, with potency as an indication, Dr. Thyssen, professor of dermatology at the University of Copenhagen, observed that abrocitinib and dupilumab “are somewhere in the middle,” tralokinumab and baricitinib are “slightly weaker,” while upadacitinib is “very potent.” (Dupilumab is approved by the FDA for treating AD ages 6 and older, and tralokinumab, a fully human monoclonal antibody that binds to IL-13, is under FDA review for AD.)
However, he cautioned that making direct comparisons of these drugs is limited by differences in clinical trial designs, trial length, severity of disease at baseline, and demographics. “Placebo effects also differ between trials, and the speed of onset is different between JAK inhibitors and biologic agents. Because of this, efficacy can be difficult to assess over 12-16 weeks. That’s why long-term studies are necessary.”
It’s also tricky to compare safety signals with baricitinib, abrocitinib, and upadacitinib, “because some of them are JAK1 inhibitors; others are JAK1/JAK2 inhibitors,” he continued. “Even the molecules that inhibit JAK1 are different, so making a comparison between abrocitinib and upadacitinib requires studies that do this is in the best way and over a long period of time.”
Safety signals
Common safety signals in this drug class include nasopharyngitis, nausea, and headache. “Many of these are short lasting, meaning that patients will perhaps have a headache for a day or two and then it will be over,” said Dr. Thyssen, who is also a consultant dermatologist at Bispebjerg Hospital in Copenhagen. “This means that even though we see high proportions of safety signals, this is probably not going to limit the use of JAK inhibitors in most of our patients. Then we have an acne signal in higher proportions for abrocitinib and upadacitinib than for baricitinib, so perhaps this is related to the potency.”
There is also an increased risk for infections, including herpes zoster. “Is this a class effect?” he asked. “We see quite a bit for baricitinib, particularly when it’s used for rheumatoid arthritis. We also see it in AD patients, but we don’t know to what degree yet. We need the real-world evidence before we can make any conclusions.” Routine blood monitoring tests are also required in patients taking JAK inhibitors, because of the risk for leukopenia and effects on liver enzymes.
Then there’s the risk of deep vein thrombosis/pulmonary embolism. “This is mostly linked to baricitinib use, but is this a class effect or is it specific to baricitinib?” he asked. “We’ll have to wait and see, but I think overall, this is not something I have great fear of because we see that AD patients are young, usually with a normal [body mass index], at least in Europe. But we have to study this closely.”
From a clinical standpoint, JAK1/2 inhibitors work well on every measurable aspect of AD, he said, including eczema severity, itch, skin pain, sleep, and quality of life. “Based on conference abstracts and publications, they seem to work equally well independent of race, BMI, atopy status, age, and whether their AD is extrinsic or intrinsic,” Dr. Thyssen added. “One thing we haven’t learned from the companies is, what patients have the highest likelihood of getting a good treatment response? We don’t have good biomarkers yet, but anything the companies can do to help us identify the patients with the greatest chance of success would be so welcome.”
The best available data suggest that JAK inhibitors benefit AD patients with certain comorbidities, including inflammatory bowel disease (with upadacitinib), RA (with both baricitinib and upadacitinib), and alopecia areata (with baricitinib). “These drugs also have been shown to work well for the psychiatric symptoms of disease,” he said.
“As for patients with type 2 inflammation in the airways such as asthma and rhinitis, dupilumab works, but do the JAK inhibitors work? It’s possible from a mode of action standpoint, but we don’t know.” It also remains unclear how JAK inhibitors will fare in the treatment of chronic hand eczema and ocular surface disease, like allergic conjunctivitis, he said.
Despite the unknowns, Dr. Thyssen emphasized the promise that JAK inhibitors hold for AD patients. “We know they provide good AD control,” he said. “For some, like baricitinib, you may need to instruct the patient to use topical corticosteroids as well, but this does not seem to be necessary for upadacitinib and abrocitinib. You really have a single bullet here that will take away most of the problems for many patients, with very fast onset of action, which is important for our patients.”
Dr. Thyssen disclosed that he is a speaker, advisory board member, and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
largely because of the heterogeneous nature of the disease.
“Atopic dermatitis patients have different complaints,” Jacob P. Thyssen, MD, PhD, said during the Revolutionizing Atopic Dermatitis symposium. “Some of them have repeated infections. Some have psychiatric symptoms. Others have widespread eczema. When you talk about how well they work, it really depends on what aspects of AD, what subgroups of AD, and how well they work with comorbidities of AD.”
Baricitinib, a JAK1/JAK2 inhibitor in 2-mg and 4-mg tablets, is available in the European Union, and is under Food and Drug Administration review for AD in the United States. Two JAK1 inhibitors continue to be evaluated in AD clinical trials and are also under FDA review for AD: abrocitinib (100 mg and 200 mg) and upadacitinib (15 mg and 30 mg). None of these agents have been tested in head-to-head trials and only one (abrocitinib) has been compared with the interleukin-4 receptor–alpha antagonist dupilumab, which makes meaningful direct comparisons impossible. (Baricitinib and upadacitinib are approved for treating RA in the United States.)
In his informal assessment from clinical trial data of how these three JAK inhibitors compare with the biologic agents dupilumab and tralokinumab, with potency as an indication, Dr. Thyssen, professor of dermatology at the University of Copenhagen, observed that abrocitinib and dupilumab “are somewhere in the middle,” tralokinumab and baricitinib are “slightly weaker,” while upadacitinib is “very potent.” (Dupilumab is approved by the FDA for treating AD ages 6 and older, and tralokinumab, a fully human monoclonal antibody that binds to IL-13, is under FDA review for AD.)
However, he cautioned that making direct comparisons of these drugs is limited by differences in clinical trial designs, trial length, severity of disease at baseline, and demographics. “Placebo effects also differ between trials, and the speed of onset is different between JAK inhibitors and biologic agents. Because of this, efficacy can be difficult to assess over 12-16 weeks. That’s why long-term studies are necessary.”
It’s also tricky to compare safety signals with baricitinib, abrocitinib, and upadacitinib, “because some of them are JAK1 inhibitors; others are JAK1/JAK2 inhibitors,” he continued. “Even the molecules that inhibit JAK1 are different, so making a comparison between abrocitinib and upadacitinib requires studies that do this is in the best way and over a long period of time.”
Safety signals
Common safety signals in this drug class include nasopharyngitis, nausea, and headache. “Many of these are short lasting, meaning that patients will perhaps have a headache for a day or two and then it will be over,” said Dr. Thyssen, who is also a consultant dermatologist at Bispebjerg Hospital in Copenhagen. “This means that even though we see high proportions of safety signals, this is probably not going to limit the use of JAK inhibitors in most of our patients. Then we have an acne signal in higher proportions for abrocitinib and upadacitinib than for baricitinib, so perhaps this is related to the potency.”
There is also an increased risk for infections, including herpes zoster. “Is this a class effect?” he asked. “We see quite a bit for baricitinib, particularly when it’s used for rheumatoid arthritis. We also see it in AD patients, but we don’t know to what degree yet. We need the real-world evidence before we can make any conclusions.” Routine blood monitoring tests are also required in patients taking JAK inhibitors, because of the risk for leukopenia and effects on liver enzymes.
Then there’s the risk of deep vein thrombosis/pulmonary embolism. “This is mostly linked to baricitinib use, but is this a class effect or is it specific to baricitinib?” he asked. “We’ll have to wait and see, but I think overall, this is not something I have great fear of because we see that AD patients are young, usually with a normal [body mass index], at least in Europe. But we have to study this closely.”
From a clinical standpoint, JAK1/2 inhibitors work well on every measurable aspect of AD, he said, including eczema severity, itch, skin pain, sleep, and quality of life. “Based on conference abstracts and publications, they seem to work equally well independent of race, BMI, atopy status, age, and whether their AD is extrinsic or intrinsic,” Dr. Thyssen added. “One thing we haven’t learned from the companies is, what patients have the highest likelihood of getting a good treatment response? We don’t have good biomarkers yet, but anything the companies can do to help us identify the patients with the greatest chance of success would be so welcome.”
The best available data suggest that JAK inhibitors benefit AD patients with certain comorbidities, including inflammatory bowel disease (with upadacitinib), RA (with both baricitinib and upadacitinib), and alopecia areata (with baricitinib). “These drugs also have been shown to work well for the psychiatric symptoms of disease,” he said.
“As for patients with type 2 inflammation in the airways such as asthma and rhinitis, dupilumab works, but do the JAK inhibitors work? It’s possible from a mode of action standpoint, but we don’t know.” It also remains unclear how JAK inhibitors will fare in the treatment of chronic hand eczema and ocular surface disease, like allergic conjunctivitis, he said.
Despite the unknowns, Dr. Thyssen emphasized the promise that JAK inhibitors hold for AD patients. “We know they provide good AD control,” he said. “For some, like baricitinib, you may need to instruct the patient to use topical corticosteroids as well, but this does not seem to be necessary for upadacitinib and abrocitinib. You really have a single bullet here that will take away most of the problems for many patients, with very fast onset of action, which is important for our patients.”
Dr. Thyssen disclosed that he is a speaker, advisory board member, and/or investigator for Regeneron, Sanofi-Genzyme, Eli Lilly, Pfizer, LEO Pharma, AbbVie, and Almirall.
FROM REVOLUTIONIZING AD 2021
Indoor tanning ICD-10 codes may be underused, study finds
according to a study presented at the annual meeting of the Society for Investigative Dermatology.
“Since indoor tanning ICD-10 codes were only recently universally implemented in 2015, and providers may still be using other codes that cover similar services, we think our data likely underestimate the number of encounters and sequelae associated with indoor tanning,” Alexandria M. Brown, BSA, of Baylor College of Medicine, Houston, said in her presentation. “We think increased usage of these indoor tanning exposure codes in coming years will strengthen this body of indoor tanning literature and data.”
Using insurance claims data on about 43 million patients from Truven Health MarketScan, Ms. Brown and colleagues analyzed patient encounters with ICD-10 indoor tanning codes W89.1, W89.1XXA, W89.1XXD, and W89.1XXS between 2016 and 2018 for about 43 million patients. Overall, there were 4,550 patient encounters where these codes had been recorded, with most (99%) occurring in an outpatient setting. The majority of providers at these encounters were dermatologists (72%). Patients were mostly women (85%); and most were ages 25-34 years (19.4%), 35-44 years (20.6%), 45-54 years (22.7%), and 55-64 years (19%). Almost 5% were 65 and over, 11.7% were ages 18-24, and 1.6% were under age 18.
The use of indoor tanning codes were most common in the Midwest (55 per 100,000 encounters with dermatologists), compared with 16 per 100,000 in the Northeast, 21 per 100,000 in the West, and 28 per 100,000 in the South. CPT codes for “destruction of a premalignant lesion” and “biopsy” were the most frequently used codes entered at visits where indoor tanning codes were also entered, and were present in 15.1% of encounters and 18.4% of encounters, respectively.
“This suggests that many of these encounters may have been for skin cancer surveillance and that indoor tanning exposure may have been coded as part of a patient’s skin cancer risk profile,” Ms. Brown noted.
The study shows how these codes are being used and could help determine health care use patterns for these patients as well as their comorbidities, behaviors, and risk factors, according to the authors, who believe this is the first study to look at the use of ICD-10 indoor tanning codes.
“Any effort to reduce indoor tanning requires knowledge of the population at risk. It has been shown that the ability to recognize and provide counseling to at-risk patients can improve sun protective behaviors and reduce indoor tanning,” Ms. Brown said. Claims databases can be a “valuable tool to better understand patients who have been exposed to indoor tanning and their associated risk factors, comorbidities, behaviors, and health care utilization.”
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said the study was interesting and “provides some guidance with respect to who, when, and where in the U.S. to target educational initiatives on the harms of tanning beds.”
Dr. Friedman, who was not involved with the research, agreed with the authors’ assertion that their study was underestimating the use of indoor tanning beds. “Using a large database provides the means to better generalize one’s dataset; however in this case, it relies on proper coding by the practitioner,” or even using the code for tanning bed use at all.
“There also could be some inherent bias given most of the cases for which the code was used was for skin cancer surveillance, and therefore tanning bed use was top of mind,” he said.
While he believes this study may not be most efficient way of determining demographics of at-risk individuals using tanning beds, Dr. Friedman said the results “should serve as the impetus to develop public health campaigns around this information, following which research can be conducted to evaluate if the intervention had an impact.”
Ms. Brown and Dr. Friedman reported no relevant financial disclosures.
according to a study presented at the annual meeting of the Society for Investigative Dermatology.
“Since indoor tanning ICD-10 codes were only recently universally implemented in 2015, and providers may still be using other codes that cover similar services, we think our data likely underestimate the number of encounters and sequelae associated with indoor tanning,” Alexandria M. Brown, BSA, of Baylor College of Medicine, Houston, said in her presentation. “We think increased usage of these indoor tanning exposure codes in coming years will strengthen this body of indoor tanning literature and data.”
Using insurance claims data on about 43 million patients from Truven Health MarketScan, Ms. Brown and colleagues analyzed patient encounters with ICD-10 indoor tanning codes W89.1, W89.1XXA, W89.1XXD, and W89.1XXS between 2016 and 2018 for about 43 million patients. Overall, there were 4,550 patient encounters where these codes had been recorded, with most (99%) occurring in an outpatient setting. The majority of providers at these encounters were dermatologists (72%). Patients were mostly women (85%); and most were ages 25-34 years (19.4%), 35-44 years (20.6%), 45-54 years (22.7%), and 55-64 years (19%). Almost 5% were 65 and over, 11.7% were ages 18-24, and 1.6% were under age 18.
The use of indoor tanning codes were most common in the Midwest (55 per 100,000 encounters with dermatologists), compared with 16 per 100,000 in the Northeast, 21 per 100,000 in the West, and 28 per 100,000 in the South. CPT codes for “destruction of a premalignant lesion” and “biopsy” were the most frequently used codes entered at visits where indoor tanning codes were also entered, and were present in 15.1% of encounters and 18.4% of encounters, respectively.
“This suggests that many of these encounters may have been for skin cancer surveillance and that indoor tanning exposure may have been coded as part of a patient’s skin cancer risk profile,” Ms. Brown noted.
The study shows how these codes are being used and could help determine health care use patterns for these patients as well as their comorbidities, behaviors, and risk factors, according to the authors, who believe this is the first study to look at the use of ICD-10 indoor tanning codes.
“Any effort to reduce indoor tanning requires knowledge of the population at risk. It has been shown that the ability to recognize and provide counseling to at-risk patients can improve sun protective behaviors and reduce indoor tanning,” Ms. Brown said. Claims databases can be a “valuable tool to better understand patients who have been exposed to indoor tanning and their associated risk factors, comorbidities, behaviors, and health care utilization.”
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said the study was interesting and “provides some guidance with respect to who, when, and where in the U.S. to target educational initiatives on the harms of tanning beds.”
Dr. Friedman, who was not involved with the research, agreed with the authors’ assertion that their study was underestimating the use of indoor tanning beds. “Using a large database provides the means to better generalize one’s dataset; however in this case, it relies on proper coding by the practitioner,” or even using the code for tanning bed use at all.
“There also could be some inherent bias given most of the cases for which the code was used was for skin cancer surveillance, and therefore tanning bed use was top of mind,” he said.
While he believes this study may not be most efficient way of determining demographics of at-risk individuals using tanning beds, Dr. Friedman said the results “should serve as the impetus to develop public health campaigns around this information, following which research can be conducted to evaluate if the intervention had an impact.”
Ms. Brown and Dr. Friedman reported no relevant financial disclosures.
according to a study presented at the annual meeting of the Society for Investigative Dermatology.
“Since indoor tanning ICD-10 codes were only recently universally implemented in 2015, and providers may still be using other codes that cover similar services, we think our data likely underestimate the number of encounters and sequelae associated with indoor tanning,” Alexandria M. Brown, BSA, of Baylor College of Medicine, Houston, said in her presentation. “We think increased usage of these indoor tanning exposure codes in coming years will strengthen this body of indoor tanning literature and data.”
Using insurance claims data on about 43 million patients from Truven Health MarketScan, Ms. Brown and colleagues analyzed patient encounters with ICD-10 indoor tanning codes W89.1, W89.1XXA, W89.1XXD, and W89.1XXS between 2016 and 2018 for about 43 million patients. Overall, there were 4,550 patient encounters where these codes had been recorded, with most (99%) occurring in an outpatient setting. The majority of providers at these encounters were dermatologists (72%). Patients were mostly women (85%); and most were ages 25-34 years (19.4%), 35-44 years (20.6%), 45-54 years (22.7%), and 55-64 years (19%). Almost 5% were 65 and over, 11.7% were ages 18-24, and 1.6% were under age 18.
The use of indoor tanning codes were most common in the Midwest (55 per 100,000 encounters with dermatologists), compared with 16 per 100,000 in the Northeast, 21 per 100,000 in the West, and 28 per 100,000 in the South. CPT codes for “destruction of a premalignant lesion” and “biopsy” were the most frequently used codes entered at visits where indoor tanning codes were also entered, and were present in 15.1% of encounters and 18.4% of encounters, respectively.
“This suggests that many of these encounters may have been for skin cancer surveillance and that indoor tanning exposure may have been coded as part of a patient’s skin cancer risk profile,” Ms. Brown noted.
The study shows how these codes are being used and could help determine health care use patterns for these patients as well as their comorbidities, behaviors, and risk factors, according to the authors, who believe this is the first study to look at the use of ICD-10 indoor tanning codes.
“Any effort to reduce indoor tanning requires knowledge of the population at risk. It has been shown that the ability to recognize and provide counseling to at-risk patients can improve sun protective behaviors and reduce indoor tanning,” Ms. Brown said. Claims databases can be a “valuable tool to better understand patients who have been exposed to indoor tanning and their associated risk factors, comorbidities, behaviors, and health care utilization.”
In an interview, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, said the study was interesting and “provides some guidance with respect to who, when, and where in the U.S. to target educational initiatives on the harms of tanning beds.”
Dr. Friedman, who was not involved with the research, agreed with the authors’ assertion that their study was underestimating the use of indoor tanning beds. “Using a large database provides the means to better generalize one’s dataset; however in this case, it relies on proper coding by the practitioner,” or even using the code for tanning bed use at all.
“There also could be some inherent bias given most of the cases for which the code was used was for skin cancer surveillance, and therefore tanning bed use was top of mind,” he said.
While he believes this study may not be most efficient way of determining demographics of at-risk individuals using tanning beds, Dr. Friedman said the results “should serve as the impetus to develop public health campaigns around this information, following which research can be conducted to evaluate if the intervention had an impact.”
Ms. Brown and Dr. Friedman reported no relevant financial disclosures.
FROM SID 2021
Female doctors of color say they feel pressure to change their look
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
It started when a Latina doctor tweeted that she lost points on a practical exam in medical school because of her hoop earrings, with the evaluator writing “earrings, unprofessional.”
That led other female doctors to cite their own experiences, reported The Lily, a Washington Post publication aimed at millennial women. Many women posted photos of themselves wearing hoops, which have long been associated with Latina and African American women, the outlet said.
“There’s a big movement to police women of color and how they present themselves in medical spaces,” said Briana Christophers, an MD-PhD student at the Tri-Institutional MD-PhD Program in New York. “I think in part it’s a way of trying to make people who don’t usually fit the mold, fit the mold.”
Ms. Christophers, who identifies as Latina, said she was urged to wear a black or navy suit when interviewing for doctorate programs. She wore a black suit with a lavender blouse and received comments about that – some positive, some not, she said.
“Sometimes you don’t know how to interpret those sorts of comments,” Ms. Christophers said. “Do you remember because you like the shirt, or because you don’t think I should have done that?”
Doctors of color still stand out in American medicine. The Lily cited the Association of American Medical Colleges as saying that in 2018, Hispanics made up 5.8% of active American doctors and African Americans made up 5%.
Studies show that medical professionals of color often don’t receive the same respect as their White counterparts, with some people questioning whether they’re actually doctors.
“At work, wearing my white coat that has my name pretty big on it with a badge that says doctor on it, I still get asked if I’m the environmental services staff,” Alexandra Sims, MD, a pediatrician in Cincinnati, told The Lily. “I think it just demonstrates how deeply ingrained bias, racism, and sexism are in society and that we have a lot of work to do to disrupt that.”
Dr. Sims said the tweet about hoop earrings led her to wonder about daily decisions she makes about dress.
“Am I too much? Is this too much? Is this earring too big? Is this nail polish color too loud? And how will that be received at work?” she said, noting that she may opt not to wear hoops in certain situations, such as when she’s dealing with a grabby baby.
Monica Verduzco-Gutierrez, MD, professor and chair of the department of rehabilitation medicine at University of Texas Health, San Antonio, said doctors should be judged on the care they provide, not their appearance.
“Judging someone based on their earrings or their jumpsuit or whatever else that they’re noticing about the student is not an appropriate way to judge the student’s ability to take care of a patient,” Dr. Verduzco-Gutierrez said, noting that she was not speaking on behalf of the school.
A version of this article was first published on WebMD.com .
New analysis puts U.S. psoriasis prevalence at 3%
, according to an analysis of national survey data from 2011 to 2014.
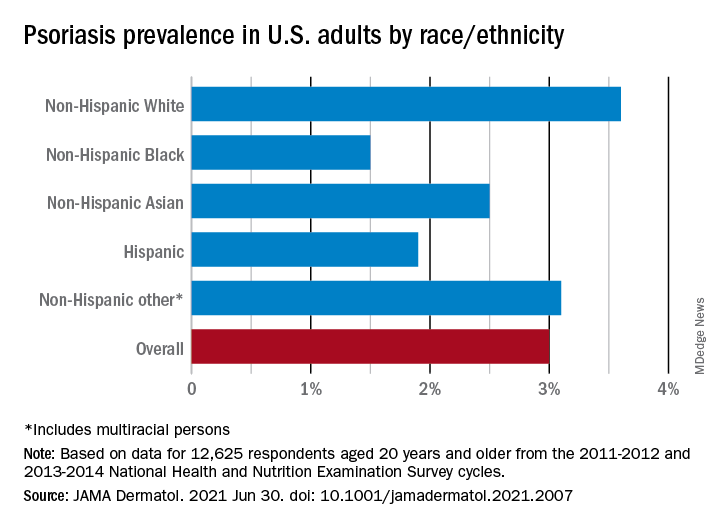
“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
, according to an analysis of national survey data from 2011 to 2014.

“The adult prevalence rate of 3.0% continues to place psoriasis as one of the most common immune-mediated diseases affecting adults” in the United States, April W. Armstrong, MD, MPH, and associates said in a report published in JAMA Dermatology. At that rate, approximately 7,560,000 Americans aged 20 years or older have psoriasis.
That overall rate among adults aged 20 years and older, based on data from the 2011-2012 and 2013-2014 cycles of the National Health and Nutrition Examination Survey (NHANES), did not change significantly when compared with the 2003-2004 NHANES, when it was 3.15% among those aged 20-59, said Dr. Armstrong, professor of dermatology, University of Southern California, Los Angeles, and associates.
For the 2011-2014 period, psoriasis prevalence was similar between women (3.2%) and men (2.8%) but was significantly associated with older age and White/non-White status. Those aged 50-59 years had the highest prevalence of any age group at 4.3% and those aged 70 and older had a rate of 3.9%, while those aged 20-29 were the lowest at 1.6%, the investigators reported.
The prevalence in non-Hispanic Whites in the United States was 3.6% over the study period, and their odds ratio for having psoriasis was 1.92, compared with non-White individuals. Asian respondents had a prevalence of 2.5%, with the Hispanic population at 1.9%, non-Hispanic Black respondents at 1.5%, and those identifying as other (including multiracial persons) at 3.1%, they said.
The NHANES sample consisted of 12,638 people who had participated in the question that asked if they had ever been diagnosed with psoriasis by a physician or other health care professional, of whom 12,625 gave a definitive yes or no answer, the investigators noted.
A much smaller number, 329, also answered a question about the severity of their disease: Fifty-six percent had little or no psoriasis, almost 22% reported 1-2 palms of involvement, 16% had 3-10 palms of involvement, and 5.5% said the coverage was more than 10 palms. Since the survey did not distinguish between treated and untreated patients, however, some “of those reporting low body surface area involvement may be receiving treatments that are controlling their otherwise more extensive disease,” they wrote.
Dr. Armstrong and another investigator said that they have received grants, personal fees, and honoraria from a number of pharmaceutical companies; two other investigators are employees of the National Psoriasis Foundation.
FROM JAMA DERMATOLOGY
New details of myocarditis linked to COVID vaccines
Further details from multiple cases of myocarditis linked to the Pfizer and Moderna mRNA COVID vaccines have been described in recent papers in the medical literature.
The cases appear to occur almost exclusively in males and most often in younger age groups. While symptoms and signs of myocarditis mostly resolved with a few days of supportive care, long-term effects are unknown at present.
The authors of all the reports and of two accompanying editorials in JAMA Cardiology are unanimous in their opinion that the benefits of vaccination still outweigh the risks.
The Centers for Disease Control and Prevention’s but committee members delivered a strong endorsement for continuing to vaccinate young people with the mRNA vaccines.
The current case reports are published in two papers in JAMA Cardiology and in three in Circulation.
U.S. military reports 23 cases
In one report in JAMA Cardiology, authors led by Jay Montgomery, MD, from Walter Reed National Military Medical Center in Bethesda, Md., described 23 cases from the U.S. Military Health System of individuals with acute myocarditis who presented within 4 days after mRNA-based COVID-19 vaccination (7 Pfizer and 16 Moderna).
All patients were male, 22 of 23 were on active duty, and the median age was 25 years (range, 20-51); 20 of the 23 cases occurred after receipt of a second dose of an mRNA COVID-19 vaccine.
The patients all presented with acute onset of marked chest pain. All patients had significantly elevated cardiac troponin levels. Among eight patients who underwent cardiac MRI (cMRI), all had findings consistent with the clinical diagnosis of myocarditis.
Additional testing did not identify other possible causes of myocarditis. All patients received brief supportive care and were recovered or recovering.
The authors reported that the military administered more than 2.8 million doses of mRNA COVID-19 vaccine in this period, and while the observed number of myocarditis cases was small, the number was “substantially higher” than expected among male military members after a second vaccine dose.
They noted that, based on historical data, among the 544,000 second doses to military members there may have been 0-10 expected myocarditis cases, but they observed 19 cases.
“All patients in this series reflect substantial similarities in demographic characteristics, proximate vaccine dose, onset interval, and character of vaccine-associated myocarditis. The consistent pattern of clinical presentation, rapid recovery, and absence of evidence of other causes support the diagnosis of hypersensitivity myocarditis,” they stated.
They added that presentation after a second vaccine dose or, in three patients, when vaccination followed SARS-CoV-2 infection, suggests that prior exposure was relevant in the hypersensitivity response.
“The spectrum of clinical presentation and reliance on patients seeking health care and on health care professionals recognizing a rare vaccine-associated adverse event limits determination of the true incidence of this condition,” the authors wrote.
They stressed that recognition of vaccine-associated myocarditis is clinically important because diagnosis impacts management, recommendations for exercise, and monitoring for cardiomyopathy.
But the authors also acknowledged that it is important to frame concerns about potential vaccine-associated myocarditis within the context of the current pandemic.
“Infection with SARS-CoV-2 is a clear cause of serious cardiac injury in many patients. ... Prevalence of cardiac injury may be as high as 60% in seriously ill patients. Notably, nearly 1% of highly fit athletes with mild COVID-19 infection have evidence of myocarditis on cMRI,” they wrote.
“Given that COVID-19 vaccines are remarkably effective at preventing infection, any risk of rare adverse events following immunization must be carefully weighed against the very substantial benefit of vaccination,” they concluded.
Four cases at Duke
In the second paper in JAMA Cardiology, a group led by Han W. Kim, MD, reported four patients with acute myocarditis occurring within days of mRNA COVID-19 vaccination (two Pfizer and two Moderna) in patients treated at Duke University Medical Center, Durham, N.C. The hospital courses of the four patients with myocarditis following COVID-19 vaccination were uneventful, and they were discharged within 2-4 days.
The authors said that, although a causal relationship cannot be established, none of the patients had a viral prodrome or had coincident testing that revealed an alternative explanation.
They stated that these four patients represent the majority of patients with acute myocarditis identified in the past 3 months at their institution, and this led to the highest total number of patients with acute myocarditis, compared with the same 3-month period for the past 5 years.
“Additionally, we identified only those patients with severe unremitting chest pain who sought medical attention. Those with mild or moderate chest pain might not seek medical attention, and it is possible that subclinical myocarditis may occur and could be detected by active surveillance, as has been described with smallpox vaccination,” they wrote.
Further case reports
In one of the papers in Circulation, a group led by Kathryn F. Larson, MD, from the Mayo Clinic in Rochester, Minn., described eight patients hospitalized with chest pain who were diagnosed with myocarditis within 2-4 days of receiving either the Pfizer or Moderna vaccine.
Two of the patients had previously been infected by SARS-CoV-2 without need for hospitalization. All individuals were otherwise healthy males between the ages of 21 and 56 years. All but one patient developed symptoms after their second dose, and the one patient who developed myocarditis after the first vaccine dose had previously been infected with SARS-CoV-2.
Systemic symptoms began within 24 hours after vaccine administration in five of eight patients, with chest pain presenting between 48 and 96 hours later. Troponin values were elevated in all individuals and appeared to peak the day after admission, whereas none had eosinophilia.
Cardiac MRI revealed findings consistent with myocarditis in all patients. All patients had resolution of their chest pain and were discharged from the hospital in stable condition.
“The patients presented here demonstrated typical signs, symptoms, and diagnostic features of acute myocarditis. The temporal association between receiving an mRNA-based COVID-19 vaccine and the development of myocarditis is notable,” the authors said.
They added that they would consider the use of corticosteroids in these patients but cautioned that this could reduce the specific immune response against SARS-COV-2 triggered by the vaccine. “Thus, the duration of corticosteroid administration should be limited to the resolution of the symptoms or ventricular arrhythmias or the recovery of the left ventricular ejection fraction.”
Pending publication of long-term outcome data after SARS-CoV-2 vaccine–related myocarditis, they suggest adherence to the current consensus recommendation to abstain from competitive sports for a period of 3-6 months with reevaluation prior to sports participation.
In another of the Circulation papers, a group led by Carolyn M. Rosner, MSN, presented a case series of seven patients hospitalized for acute myocarditis-like illness following COVID-19 vaccination, from two U.S. medical centers, in Falls Church, Va., and Dallas. All patients were males below the age of 40 years and of White or Hispanic race/ethnicity. Only one patient reported prior history of COVID-19 infection. Six patients received mRNA (Moderna or Pfizer) and one received the adenovirus (Johnson & Johnson) vaccine. All patients presented 3-7 days post vaccination with acute onset chest pain and biochemical evidence of myocardial injury.
Hospital length of stay was 3 days, and all patients’ symptoms resolved by hospital discharge.
And finally, the third paper in Circulation reported a detailed description of one patient – a 52-year-old, previously healthy male who presented with acute myocarditis 3 days after the administration of the second dose of Moderna’s COVID-19 vaccine. The symptoms resolved, and there was a gradual improvement in cMRI findings. Ischemic injury and other potential causes of acute myocardial injury were excluded, as were other potential infectious causes of myocarditis, and there was no evidence of systemic autoimmune disease.
“Clinicians should be aware that myocarditis may be present in patients exhibiting cardiac signs and symptoms 2-4 days after COVID-19 vaccination,” the authors said.
They added that additional surveillance of such adverse events post–COVID-19 vaccination will help identify subgroups at higher risk for this vaccine-related effect, and whether additional precautions are necessary.
‘Benefits outweigh risk’
In an accompanying editorial in JAMA Cardiology, three doctors from the CDC cite several other reports of myocarditis after mRNA COVID vaccination. These include a case report published in Pediatrics of seven male adolescents aged 14-19 years who presented with myocarditis or myopericarditis within 4 days after receipt of a second dose of the Pfizer vaccine.
But the editorialists noted that the most comprehensive data about the risk for myocarditis following immunization with mRNA vaccines comes from Israel.
The Israeli Ministry of Health recently posted data describing 121 myocarditis cases occurring within 30 days of a second dose of mRNA vaccine among 5,049,424 persons, suggesting a crude incidence rate of approximately 24 cases per million.
On the current case reports, the CDC doctors wrote: “The striking clinical similarities in the presentations of these patients, their recent vaccination with an mRNA-based COVID-19 vaccine, and the lack of any alternative etiologies for acute myocarditis suggest an association with immunization.”
They said that acute onset of chest pain 3-5 days after vaccine administration, usually after a second dose, is a typical feature of reported cases and suggests an immune-mediated mechanism.
But SARS-CoV-2 infection also causes cardiac injury which may result in severe outcomes, and based on currently available data, myocarditis following immunization with current mRNA-based vaccines is rare.
“At present, the benefits of immunization in preventing severe morbidity favors continued COVID-19 vaccination, particularly considering the increasing COVID-19 hospitalization rates among adolescents reported during spring 2021,” the editorialists stated.
But they added that many questions remain. These include whether modifications are needed to the vaccine schedule among persons with a history of possible or confirmed myocarditis after COVID vaccine, how should postvaccine myocarditis be managed, how often should follow-up assessments be performed, how might follow-up assessments affect recommendations to avoid vigorous physical activity following the diagnosis of myocarditis, and do all likely cases of acute myocarditis that appear to be uncomplicated require cardiac MRI for more definitive diagnosis?
“While the data needed to answer such questions are being collected, there is an opportunity for researchers with expertise in myocarditis to develop a comprehensive, national assessment of the natural history, pathogenesis, and treatment of acute myocarditis associated with receipt of mRNA-based COVID-19 vaccines,” they concluded.
In a second editorial in JAMA Cardiology, a group of editors from the journal acknowledged that publication of the current case reports may contribute to additional public concern regarding immunization. But they added that clinicians discussing immunization with patients should recognize that these case series suggest that the symptomatic events consistent with myocarditis are still very rare and appear to be self-limiting.
“Given the risks of COVID-19, including the risk of myocarditis from COVID-19 infection, the editors do not believe these case reports are sufficient to interrupt the march toward maximal vaccination against SARS-CoV-2 as expeditiously as possible,” they said.
A version of this article first appeared on Medscape.com.
Further details from multiple cases of myocarditis linked to the Pfizer and Moderna mRNA COVID vaccines have been described in recent papers in the medical literature.
The cases appear to occur almost exclusively in males and most often in younger age groups. While symptoms and signs of myocarditis mostly resolved with a few days of supportive care, long-term effects are unknown at present.
The authors of all the reports and of two accompanying editorials in JAMA Cardiology are unanimous in their opinion that the benefits of vaccination still outweigh the risks.
The Centers for Disease Control and Prevention’s but committee members delivered a strong endorsement for continuing to vaccinate young people with the mRNA vaccines.
The current case reports are published in two papers in JAMA Cardiology and in three in Circulation.
U.S. military reports 23 cases
In one report in JAMA Cardiology, authors led by Jay Montgomery, MD, from Walter Reed National Military Medical Center in Bethesda, Md., described 23 cases from the U.S. Military Health System of individuals with acute myocarditis who presented within 4 days after mRNA-based COVID-19 vaccination (7 Pfizer and 16 Moderna).
All patients were male, 22 of 23 were on active duty, and the median age was 25 years (range, 20-51); 20 of the 23 cases occurred after receipt of a second dose of an mRNA COVID-19 vaccine.
The patients all presented with acute onset of marked chest pain. All patients had significantly elevated cardiac troponin levels. Among eight patients who underwent cardiac MRI (cMRI), all had findings consistent with the clinical diagnosis of myocarditis.
Additional testing did not identify other possible causes of myocarditis. All patients received brief supportive care and were recovered or recovering.
The authors reported that the military administered more than 2.8 million doses of mRNA COVID-19 vaccine in this period, and while the observed number of myocarditis cases was small, the number was “substantially higher” than expected among male military members after a second vaccine dose.
They noted that, based on historical data, among the 544,000 second doses to military members there may have been 0-10 expected myocarditis cases, but they observed 19 cases.
“All patients in this series reflect substantial similarities in demographic characteristics, proximate vaccine dose, onset interval, and character of vaccine-associated myocarditis. The consistent pattern of clinical presentation, rapid recovery, and absence of evidence of other causes support the diagnosis of hypersensitivity myocarditis,” they stated.
They added that presentation after a second vaccine dose or, in three patients, when vaccination followed SARS-CoV-2 infection, suggests that prior exposure was relevant in the hypersensitivity response.
“The spectrum of clinical presentation and reliance on patients seeking health care and on health care professionals recognizing a rare vaccine-associated adverse event limits determination of the true incidence of this condition,” the authors wrote.
They stressed that recognition of vaccine-associated myocarditis is clinically important because diagnosis impacts management, recommendations for exercise, and monitoring for cardiomyopathy.
But the authors also acknowledged that it is important to frame concerns about potential vaccine-associated myocarditis within the context of the current pandemic.
“Infection with SARS-CoV-2 is a clear cause of serious cardiac injury in many patients. ... Prevalence of cardiac injury may be as high as 60% in seriously ill patients. Notably, nearly 1% of highly fit athletes with mild COVID-19 infection have evidence of myocarditis on cMRI,” they wrote.
“Given that COVID-19 vaccines are remarkably effective at preventing infection, any risk of rare adverse events following immunization must be carefully weighed against the very substantial benefit of vaccination,” they concluded.
Four cases at Duke
In the second paper in JAMA Cardiology, a group led by Han W. Kim, MD, reported four patients with acute myocarditis occurring within days of mRNA COVID-19 vaccination (two Pfizer and two Moderna) in patients treated at Duke University Medical Center, Durham, N.C. The hospital courses of the four patients with myocarditis following COVID-19 vaccination were uneventful, and they were discharged within 2-4 days.
The authors said that, although a causal relationship cannot be established, none of the patients had a viral prodrome or had coincident testing that revealed an alternative explanation.
They stated that these four patients represent the majority of patients with acute myocarditis identified in the past 3 months at their institution, and this led to the highest total number of patients with acute myocarditis, compared with the same 3-month period for the past 5 years.
“Additionally, we identified only those patients with severe unremitting chest pain who sought medical attention. Those with mild or moderate chest pain might not seek medical attention, and it is possible that subclinical myocarditis may occur and could be detected by active surveillance, as has been described with smallpox vaccination,” they wrote.
Further case reports
In one of the papers in Circulation, a group led by Kathryn F. Larson, MD, from the Mayo Clinic in Rochester, Minn., described eight patients hospitalized with chest pain who were diagnosed with myocarditis within 2-4 days of receiving either the Pfizer or Moderna vaccine.
Two of the patients had previously been infected by SARS-CoV-2 without need for hospitalization. All individuals were otherwise healthy males between the ages of 21 and 56 years. All but one patient developed symptoms after their second dose, and the one patient who developed myocarditis after the first vaccine dose had previously been infected with SARS-CoV-2.
Systemic symptoms began within 24 hours after vaccine administration in five of eight patients, with chest pain presenting between 48 and 96 hours later. Troponin values were elevated in all individuals and appeared to peak the day after admission, whereas none had eosinophilia.
Cardiac MRI revealed findings consistent with myocarditis in all patients. All patients had resolution of their chest pain and were discharged from the hospital in stable condition.
“The patients presented here demonstrated typical signs, symptoms, and diagnostic features of acute myocarditis. The temporal association between receiving an mRNA-based COVID-19 vaccine and the development of myocarditis is notable,” the authors said.
They added that they would consider the use of corticosteroids in these patients but cautioned that this could reduce the specific immune response against SARS-COV-2 triggered by the vaccine. “Thus, the duration of corticosteroid administration should be limited to the resolution of the symptoms or ventricular arrhythmias or the recovery of the left ventricular ejection fraction.”
Pending publication of long-term outcome data after SARS-CoV-2 vaccine–related myocarditis, they suggest adherence to the current consensus recommendation to abstain from competitive sports for a period of 3-6 months with reevaluation prior to sports participation.
In another of the Circulation papers, a group led by Carolyn M. Rosner, MSN, presented a case series of seven patients hospitalized for acute myocarditis-like illness following COVID-19 vaccination, from two U.S. medical centers, in Falls Church, Va., and Dallas. All patients were males below the age of 40 years and of White or Hispanic race/ethnicity. Only one patient reported prior history of COVID-19 infection. Six patients received mRNA (Moderna or Pfizer) and one received the adenovirus (Johnson & Johnson) vaccine. All patients presented 3-7 days post vaccination with acute onset chest pain and biochemical evidence of myocardial injury.
Hospital length of stay was 3 days, and all patients’ symptoms resolved by hospital discharge.
And finally, the third paper in Circulation reported a detailed description of one patient – a 52-year-old, previously healthy male who presented with acute myocarditis 3 days after the administration of the second dose of Moderna’s COVID-19 vaccine. The symptoms resolved, and there was a gradual improvement in cMRI findings. Ischemic injury and other potential causes of acute myocardial injury were excluded, as were other potential infectious causes of myocarditis, and there was no evidence of systemic autoimmune disease.
“Clinicians should be aware that myocarditis may be present in patients exhibiting cardiac signs and symptoms 2-4 days after COVID-19 vaccination,” the authors said.
They added that additional surveillance of such adverse events post–COVID-19 vaccination will help identify subgroups at higher risk for this vaccine-related effect, and whether additional precautions are necessary.
‘Benefits outweigh risk’
In an accompanying editorial in JAMA Cardiology, three doctors from the CDC cite several other reports of myocarditis after mRNA COVID vaccination. These include a case report published in Pediatrics of seven male adolescents aged 14-19 years who presented with myocarditis or myopericarditis within 4 days after receipt of a second dose of the Pfizer vaccine.
But the editorialists noted that the most comprehensive data about the risk for myocarditis following immunization with mRNA vaccines comes from Israel.
The Israeli Ministry of Health recently posted data describing 121 myocarditis cases occurring within 30 days of a second dose of mRNA vaccine among 5,049,424 persons, suggesting a crude incidence rate of approximately 24 cases per million.
On the current case reports, the CDC doctors wrote: “The striking clinical similarities in the presentations of these patients, their recent vaccination with an mRNA-based COVID-19 vaccine, and the lack of any alternative etiologies for acute myocarditis suggest an association with immunization.”
They said that acute onset of chest pain 3-5 days after vaccine administration, usually after a second dose, is a typical feature of reported cases and suggests an immune-mediated mechanism.
But SARS-CoV-2 infection also causes cardiac injury which may result in severe outcomes, and based on currently available data, myocarditis following immunization with current mRNA-based vaccines is rare.
“At present, the benefits of immunization in preventing severe morbidity favors continued COVID-19 vaccination, particularly considering the increasing COVID-19 hospitalization rates among adolescents reported during spring 2021,” the editorialists stated.
But they added that many questions remain. These include whether modifications are needed to the vaccine schedule among persons with a history of possible or confirmed myocarditis after COVID vaccine, how should postvaccine myocarditis be managed, how often should follow-up assessments be performed, how might follow-up assessments affect recommendations to avoid vigorous physical activity following the diagnosis of myocarditis, and do all likely cases of acute myocarditis that appear to be uncomplicated require cardiac MRI for more definitive diagnosis?
“While the data needed to answer such questions are being collected, there is an opportunity for researchers with expertise in myocarditis to develop a comprehensive, national assessment of the natural history, pathogenesis, and treatment of acute myocarditis associated with receipt of mRNA-based COVID-19 vaccines,” they concluded.
In a second editorial in JAMA Cardiology, a group of editors from the journal acknowledged that publication of the current case reports may contribute to additional public concern regarding immunization. But they added that clinicians discussing immunization with patients should recognize that these case series suggest that the symptomatic events consistent with myocarditis are still very rare and appear to be self-limiting.
“Given the risks of COVID-19, including the risk of myocarditis from COVID-19 infection, the editors do not believe these case reports are sufficient to interrupt the march toward maximal vaccination against SARS-CoV-2 as expeditiously as possible,” they said.
A version of this article first appeared on Medscape.com.
Further details from multiple cases of myocarditis linked to the Pfizer and Moderna mRNA COVID vaccines have been described in recent papers in the medical literature.
The cases appear to occur almost exclusively in males and most often in younger age groups. While symptoms and signs of myocarditis mostly resolved with a few days of supportive care, long-term effects are unknown at present.
The authors of all the reports and of two accompanying editorials in JAMA Cardiology are unanimous in their opinion that the benefits of vaccination still outweigh the risks.
The Centers for Disease Control and Prevention’s but committee members delivered a strong endorsement for continuing to vaccinate young people with the mRNA vaccines.
The current case reports are published in two papers in JAMA Cardiology and in three in Circulation.
U.S. military reports 23 cases
In one report in JAMA Cardiology, authors led by Jay Montgomery, MD, from Walter Reed National Military Medical Center in Bethesda, Md., described 23 cases from the U.S. Military Health System of individuals with acute myocarditis who presented within 4 days after mRNA-based COVID-19 vaccination (7 Pfizer and 16 Moderna).
All patients were male, 22 of 23 were on active duty, and the median age was 25 years (range, 20-51); 20 of the 23 cases occurred after receipt of a second dose of an mRNA COVID-19 vaccine.
The patients all presented with acute onset of marked chest pain. All patients had significantly elevated cardiac troponin levels. Among eight patients who underwent cardiac MRI (cMRI), all had findings consistent with the clinical diagnosis of myocarditis.
Additional testing did not identify other possible causes of myocarditis. All patients received brief supportive care and were recovered or recovering.
The authors reported that the military administered more than 2.8 million doses of mRNA COVID-19 vaccine in this period, and while the observed number of myocarditis cases was small, the number was “substantially higher” than expected among male military members after a second vaccine dose.
They noted that, based on historical data, among the 544,000 second doses to military members there may have been 0-10 expected myocarditis cases, but they observed 19 cases.
“All patients in this series reflect substantial similarities in demographic characteristics, proximate vaccine dose, onset interval, and character of vaccine-associated myocarditis. The consistent pattern of clinical presentation, rapid recovery, and absence of evidence of other causes support the diagnosis of hypersensitivity myocarditis,” they stated.
They added that presentation after a second vaccine dose or, in three patients, when vaccination followed SARS-CoV-2 infection, suggests that prior exposure was relevant in the hypersensitivity response.
“The spectrum of clinical presentation and reliance on patients seeking health care and on health care professionals recognizing a rare vaccine-associated adverse event limits determination of the true incidence of this condition,” the authors wrote.
They stressed that recognition of vaccine-associated myocarditis is clinically important because diagnosis impacts management, recommendations for exercise, and monitoring for cardiomyopathy.
But the authors also acknowledged that it is important to frame concerns about potential vaccine-associated myocarditis within the context of the current pandemic.
“Infection with SARS-CoV-2 is a clear cause of serious cardiac injury in many patients. ... Prevalence of cardiac injury may be as high as 60% in seriously ill patients. Notably, nearly 1% of highly fit athletes with mild COVID-19 infection have evidence of myocarditis on cMRI,” they wrote.
“Given that COVID-19 vaccines are remarkably effective at preventing infection, any risk of rare adverse events following immunization must be carefully weighed against the very substantial benefit of vaccination,” they concluded.
Four cases at Duke
In the second paper in JAMA Cardiology, a group led by Han W. Kim, MD, reported four patients with acute myocarditis occurring within days of mRNA COVID-19 vaccination (two Pfizer and two Moderna) in patients treated at Duke University Medical Center, Durham, N.C. The hospital courses of the four patients with myocarditis following COVID-19 vaccination were uneventful, and they were discharged within 2-4 days.
The authors said that, although a causal relationship cannot be established, none of the patients had a viral prodrome or had coincident testing that revealed an alternative explanation.
They stated that these four patients represent the majority of patients with acute myocarditis identified in the past 3 months at their institution, and this led to the highest total number of patients with acute myocarditis, compared with the same 3-month period for the past 5 years.
“Additionally, we identified only those patients with severe unremitting chest pain who sought medical attention. Those with mild or moderate chest pain might not seek medical attention, and it is possible that subclinical myocarditis may occur and could be detected by active surveillance, as has been described with smallpox vaccination,” they wrote.
Further case reports
In one of the papers in Circulation, a group led by Kathryn F. Larson, MD, from the Mayo Clinic in Rochester, Minn., described eight patients hospitalized with chest pain who were diagnosed with myocarditis within 2-4 days of receiving either the Pfizer or Moderna vaccine.
Two of the patients had previously been infected by SARS-CoV-2 without need for hospitalization. All individuals were otherwise healthy males between the ages of 21 and 56 years. All but one patient developed symptoms after their second dose, and the one patient who developed myocarditis after the first vaccine dose had previously been infected with SARS-CoV-2.
Systemic symptoms began within 24 hours after vaccine administration in five of eight patients, with chest pain presenting between 48 and 96 hours later. Troponin values were elevated in all individuals and appeared to peak the day after admission, whereas none had eosinophilia.
Cardiac MRI revealed findings consistent with myocarditis in all patients. All patients had resolution of their chest pain and were discharged from the hospital in stable condition.
“The patients presented here demonstrated typical signs, symptoms, and diagnostic features of acute myocarditis. The temporal association between receiving an mRNA-based COVID-19 vaccine and the development of myocarditis is notable,” the authors said.
They added that they would consider the use of corticosteroids in these patients but cautioned that this could reduce the specific immune response against SARS-COV-2 triggered by the vaccine. “Thus, the duration of corticosteroid administration should be limited to the resolution of the symptoms or ventricular arrhythmias or the recovery of the left ventricular ejection fraction.”
Pending publication of long-term outcome data after SARS-CoV-2 vaccine–related myocarditis, they suggest adherence to the current consensus recommendation to abstain from competitive sports for a period of 3-6 months with reevaluation prior to sports participation.
In another of the Circulation papers, a group led by Carolyn M. Rosner, MSN, presented a case series of seven patients hospitalized for acute myocarditis-like illness following COVID-19 vaccination, from two U.S. medical centers, in Falls Church, Va., and Dallas. All patients were males below the age of 40 years and of White or Hispanic race/ethnicity. Only one patient reported prior history of COVID-19 infection. Six patients received mRNA (Moderna or Pfizer) and one received the adenovirus (Johnson & Johnson) vaccine. All patients presented 3-7 days post vaccination with acute onset chest pain and biochemical evidence of myocardial injury.
Hospital length of stay was 3 days, and all patients’ symptoms resolved by hospital discharge.
And finally, the third paper in Circulation reported a detailed description of one patient – a 52-year-old, previously healthy male who presented with acute myocarditis 3 days after the administration of the second dose of Moderna’s COVID-19 vaccine. The symptoms resolved, and there was a gradual improvement in cMRI findings. Ischemic injury and other potential causes of acute myocardial injury were excluded, as were other potential infectious causes of myocarditis, and there was no evidence of systemic autoimmune disease.
“Clinicians should be aware that myocarditis may be present in patients exhibiting cardiac signs and symptoms 2-4 days after COVID-19 vaccination,” the authors said.
They added that additional surveillance of such adverse events post–COVID-19 vaccination will help identify subgroups at higher risk for this vaccine-related effect, and whether additional precautions are necessary.
‘Benefits outweigh risk’
In an accompanying editorial in JAMA Cardiology, three doctors from the CDC cite several other reports of myocarditis after mRNA COVID vaccination. These include a case report published in Pediatrics of seven male adolescents aged 14-19 years who presented with myocarditis or myopericarditis within 4 days after receipt of a second dose of the Pfizer vaccine.
But the editorialists noted that the most comprehensive data about the risk for myocarditis following immunization with mRNA vaccines comes from Israel.
The Israeli Ministry of Health recently posted data describing 121 myocarditis cases occurring within 30 days of a second dose of mRNA vaccine among 5,049,424 persons, suggesting a crude incidence rate of approximately 24 cases per million.
On the current case reports, the CDC doctors wrote: “The striking clinical similarities in the presentations of these patients, their recent vaccination with an mRNA-based COVID-19 vaccine, and the lack of any alternative etiologies for acute myocarditis suggest an association with immunization.”
They said that acute onset of chest pain 3-5 days after vaccine administration, usually after a second dose, is a typical feature of reported cases and suggests an immune-mediated mechanism.
But SARS-CoV-2 infection also causes cardiac injury which may result in severe outcomes, and based on currently available data, myocarditis following immunization with current mRNA-based vaccines is rare.
“At present, the benefits of immunization in preventing severe morbidity favors continued COVID-19 vaccination, particularly considering the increasing COVID-19 hospitalization rates among adolescents reported during spring 2021,” the editorialists stated.
But they added that many questions remain. These include whether modifications are needed to the vaccine schedule among persons with a history of possible or confirmed myocarditis after COVID vaccine, how should postvaccine myocarditis be managed, how often should follow-up assessments be performed, how might follow-up assessments affect recommendations to avoid vigorous physical activity following the diagnosis of myocarditis, and do all likely cases of acute myocarditis that appear to be uncomplicated require cardiac MRI for more definitive diagnosis?
“While the data needed to answer such questions are being collected, there is an opportunity for researchers with expertise in myocarditis to develop a comprehensive, national assessment of the natural history, pathogenesis, and treatment of acute myocarditis associated with receipt of mRNA-based COVID-19 vaccines,” they concluded.
In a second editorial in JAMA Cardiology, a group of editors from the journal acknowledged that publication of the current case reports may contribute to additional public concern regarding immunization. But they added that clinicians discussing immunization with patients should recognize that these case series suggest that the symptomatic events consistent with myocarditis are still very rare and appear to be self-limiting.
“Given the risks of COVID-19, including the risk of myocarditis from COVID-19 infection, the editors do not believe these case reports are sufficient to interrupt the march toward maximal vaccination against SARS-CoV-2 as expeditiously as possible,” they said.
A version of this article first appeared on Medscape.com.
Expert shares practical considerations when prescribing dupilumab
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
.
This scenario was illustrated in a 2020 retrospective study of 179 adults with AD who were cared for at the University of Pittsburgh Medical Center, which found that 37% did not start dupilumab, mainly due to insurance denial (19%) and high copay (11%).
“We’ve all seen this in our practice,” Amy S. Paller, MD, said during the Revolutionizing Atopic Dermatitis symposium. “We’ve also seen the denials until we get step therapy in there, so if I have a child whom I want to treat with dupilumab for safety reasons, I don’t like being told that I’m going to have to use cyclosporine or methotrexate or a medication that I think may have higher risks and certainly [would] require blood monitoring–yet that’s the state for some patients.”
Dupilumab, an interleukin-4 receptor alpha antagonist, is approved for treatment of moderate to severe AD in patients ages 6 and older.
When working to obtain insurance approval of dupilumab, Dr. Paller reminded dermatologists to document that the patient has moderate to severe AD “and document the negative effect on quality of life in order to try to help make it easier to get these medications for our patients.”
Starting patients on dupilumab
Dr. Paller, the Walter J. Hamlin Chair and Professor of Dermatology at Northwestern University, Chicago, said that if patients are on another systemic medication prior to starting dupilumab, she allows a transition period of 1-2 months. “Don’t just stop that drug because it’s ‘not working,’ ” she said. “I usually do a full dose for the first month, and a half dose for the next month before starting dupilumab. Also, don’t stop the use of topical corticosteroids. They can increase treatment response by 10%-20%, even when patients are on dupilumab.”
She recommends a 3- to 4-month trial of dupilumab while monitoring changes in disease severity, itch, and quality of life. “Usually there’s evidence of early improvement by 2 months in those who are going to do well enough to stay on the drug by about 4 months out,” she said. “In my experience, most pediatric patients do very well. In those with an inadequate response, about 50% will do better if you can increase the dose or frequency. Flares can still occur in those who do well. I usually push topicals when that happens.”
If patients respond well after starting dupilumab, Dr. Paller recommends that they continue on the drug for at least a year before considering a taper with the hope of “resetting” the immune system and having sustained improvement off drug. “Some parents and patients don’t want to stop the drug,” but for those who do, she tells them that she does not want to abruptly stop treatment, but to “space out the dosing” instead. “If someone is pretty much clear with the medication and is able to continue with topicals as you dial down, that’s great. But don’t even think about taking them off if somebody’s not clear or virtually clear, particularly if they start to flare with lower frequency.”
Data on effectiveness
Real-world data suggest that the effectiveness of dupilumab is similar to the efficacy seen in clinical trials. For example, a recently published systematic review and meta-analysis of 3,303 AD patients on dupilumab found that after 16 weeks of therapy, 60% achieved a 75% improvement in the Eczema Area and Severity (EASI75) score, and 27% achieved an EASI90. In a Dutch study of 210 adults treated with dupilumab for 52 weeks, enrolled in a Dutch registry, the mean percent reduction in EASI score was 70% at 16 weeks and 76.6% by 52 weeks.
In addition, there was at least a 4-point improvement in the Patient-Oriented Eczema Measure (POEM) score and at least a 4-point improvement in the Itch Numeric Rating Scale (NRS), said Dr. Paller, who was not involved in the study. “These patient-reported improvements were seen very early on,” she noted.
What about drug survival at 1 year? In a retrospective cohort study that drew from insurance databases, 1,963 adults given dupilumab were studied for a mean of 315 days. The rate of persistence was 92% at 6 months and 77% at 12 months. “That means that it’s still effective,” Dr. Paller said.
While that is a short period of time, she compared these results with long-term survival of nonsteroid systemic immunosuppressants such as cyclosporine, referring to a study of adults with AD treated with systemic immunosuppressants, which found “a 32% persistence rate at 12 months in drugs that require more monitoring, so more burden.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for dupilumab (Dupixent) manufacturers Regeneron and Sanofi, AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, and RAPT Therapeutics.
FROM REVOLUTIONIZING AD 2021
Malignancy risk: Secukinumab shows long-term safety for psoriasis, PsA, ankylosing spondylitis
that included 49 clinical trials.
Secukinumab (Cosentyx), an interleukin-17A antagonist, is approved for several conditions: moderate to severe psoriasis in children and adults, PsA, ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis.
Although secukinumab has demonstrated safety and tolerability, data on long-term malignancy rates are limited, wrote Mark Lebwohl, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors.
In a study published in the British Journal of Dermatology, they analyzed the combined safety data from clinical trials and postmarketing surveillance. The study population included 10,685 patients with psoriasis, 2,523 patients with PsA, and 1,311 patients with ankylosing spondylitis who received at least one approved dose of secukinumab (300 mg or 150 mg). The maximum follow-up was 5 years. The exposure-adjusted incidence rate was defined as the incidence rates per 100 patient treatment-years (PTY). The cumulative exposure for patients with psoriasis, PsA, and AS was 16,482, 4,944, and 2,668 PTY, respectively, with average follow-up times of 1.54, 1.96, and 2.03 years, respectively.
The observed and the expected number of malignancies were comparable, with a standardized incidence ratio (SIR) for malignancy of 0.99 across all treatment indications, the researchers said. In further analysis of malignancy by indication, the SIR was 0.87, 1.16, and 1.61 for psoriasis, PsA, and AS, respectively.
Data from postmarketing surveillance showed similar results: The estimated crude cumulative incidence reporting rate per 100 PTY was 0.27 for malignancy across all indications. The cumulative exposure was 285,811 PTY.
The study findings were limited by several factors including the post hoc design, differences in clinical trial methodologies, and lack of controlling for confounding variables, such as smoking status and previous exposure to systemic and biologic treatments, the researchers noted. In addition, the analysis did not include postexposure follow-up data, or data on patients who discontinued clinical trials, they said.
Overall, the analysis is the largest to date and supports the low risk of malignancy in patients with psoriasis, PsA, and AS treated with secukinumab, the researchers noted.
However, “while this assessment provides a broader understanding of the safety of secukinumab and supports its long-term use in these chronic systemic inflammatory conditions, registry data are further warranted to fully understand the real-world effect of biologics on malignancy risk,” they concluded.
“Secukinumab is a relatively newer biologic, approved in 2015, and there is currently a lack of longer-term data on the incidence of malignancy in secukinumab-treated patients, so it’s important to look at the data we have so far on this topic so we can better understand the long-term risks and counsel our psoriasis and psoriatic arthritis patients,” Flavia Fedeles, MD, of the department of dermatology at Massachusetts General Hospital, Boston, said in an interview.
Dr. Fedeles, who was not involved with the study, said that she was not surprised by the study results. “Data reported in the past from phase 3 clinical trials of secukinumab compared with placebo did not show an increase in risk of malignancy, though at that time no long-term safety data or data from patients with history of malignancy was available,” she said. “This study is reassuring in that there wasn’t a signal of increased malignancy events up to 5 years of secukinumab treatment,” said Dr. Fedeles.
However, she noted that the study has a number of limitations, including the use of clinical trials data, which have stringent inclusion/exclusion criteria that can lead to selection bias, the use of postmarketing surveillance data, the post hoc nature of the analysis, and the fact that the sponsor of the trial was the manufacturer of secukinumab, which “potentially can lead to bias to this study.”
She added that “registry data are needed to fully understand the real-world long-term effect of secukinumab on malignancy risk.”
The study was funded by Novartis. Lead author Dr. Lebwohl disclosed participating in advisory boards and/or as an investigator and/or speaker and receiving grants and/or honoraria from multiple companies including Novartis. Several study coauthors are employees of Novartis.
Dr. Fedeles had no financial conflicts to disclose.
that included 49 clinical trials.
Secukinumab (Cosentyx), an interleukin-17A antagonist, is approved for several conditions: moderate to severe psoriasis in children and adults, PsA, ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis.
Although secukinumab has demonstrated safety and tolerability, data on long-term malignancy rates are limited, wrote Mark Lebwohl, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors.
In a study published in the British Journal of Dermatology, they analyzed the combined safety data from clinical trials and postmarketing surveillance. The study population included 10,685 patients with psoriasis, 2,523 patients with PsA, and 1,311 patients with ankylosing spondylitis who received at least one approved dose of secukinumab (300 mg or 150 mg). The maximum follow-up was 5 years. The exposure-adjusted incidence rate was defined as the incidence rates per 100 patient treatment-years (PTY). The cumulative exposure for patients with psoriasis, PsA, and AS was 16,482, 4,944, and 2,668 PTY, respectively, with average follow-up times of 1.54, 1.96, and 2.03 years, respectively.
The observed and the expected number of malignancies were comparable, with a standardized incidence ratio (SIR) for malignancy of 0.99 across all treatment indications, the researchers said. In further analysis of malignancy by indication, the SIR was 0.87, 1.16, and 1.61 for psoriasis, PsA, and AS, respectively.
Data from postmarketing surveillance showed similar results: The estimated crude cumulative incidence reporting rate per 100 PTY was 0.27 for malignancy across all indications. The cumulative exposure was 285,811 PTY.
The study findings were limited by several factors including the post hoc design, differences in clinical trial methodologies, and lack of controlling for confounding variables, such as smoking status and previous exposure to systemic and biologic treatments, the researchers noted. In addition, the analysis did not include postexposure follow-up data, or data on patients who discontinued clinical trials, they said.
Overall, the analysis is the largest to date and supports the low risk of malignancy in patients with psoriasis, PsA, and AS treated with secukinumab, the researchers noted.
However, “while this assessment provides a broader understanding of the safety of secukinumab and supports its long-term use in these chronic systemic inflammatory conditions, registry data are further warranted to fully understand the real-world effect of biologics on malignancy risk,” they concluded.
“Secukinumab is a relatively newer biologic, approved in 2015, and there is currently a lack of longer-term data on the incidence of malignancy in secukinumab-treated patients, so it’s important to look at the data we have so far on this topic so we can better understand the long-term risks and counsel our psoriasis and psoriatic arthritis patients,” Flavia Fedeles, MD, of the department of dermatology at Massachusetts General Hospital, Boston, said in an interview.
Dr. Fedeles, who was not involved with the study, said that she was not surprised by the study results. “Data reported in the past from phase 3 clinical trials of secukinumab compared with placebo did not show an increase in risk of malignancy, though at that time no long-term safety data or data from patients with history of malignancy was available,” she said. “This study is reassuring in that there wasn’t a signal of increased malignancy events up to 5 years of secukinumab treatment,” said Dr. Fedeles.
However, she noted that the study has a number of limitations, including the use of clinical trials data, which have stringent inclusion/exclusion criteria that can lead to selection bias, the use of postmarketing surveillance data, the post hoc nature of the analysis, and the fact that the sponsor of the trial was the manufacturer of secukinumab, which “potentially can lead to bias to this study.”
She added that “registry data are needed to fully understand the real-world long-term effect of secukinumab on malignancy risk.”
The study was funded by Novartis. Lead author Dr. Lebwohl disclosed participating in advisory boards and/or as an investigator and/or speaker and receiving grants and/or honoraria from multiple companies including Novartis. Several study coauthors are employees of Novartis.
Dr. Fedeles had no financial conflicts to disclose.
that included 49 clinical trials.
Secukinumab (Cosentyx), an interleukin-17A antagonist, is approved for several conditions: moderate to severe psoriasis in children and adults, PsA, ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis.
Although secukinumab has demonstrated safety and tolerability, data on long-term malignancy rates are limited, wrote Mark Lebwohl, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors.
In a study published in the British Journal of Dermatology, they analyzed the combined safety data from clinical trials and postmarketing surveillance. The study population included 10,685 patients with psoriasis, 2,523 patients with PsA, and 1,311 patients with ankylosing spondylitis who received at least one approved dose of secukinumab (300 mg or 150 mg). The maximum follow-up was 5 years. The exposure-adjusted incidence rate was defined as the incidence rates per 100 patient treatment-years (PTY). The cumulative exposure for patients with psoriasis, PsA, and AS was 16,482, 4,944, and 2,668 PTY, respectively, with average follow-up times of 1.54, 1.96, and 2.03 years, respectively.
The observed and the expected number of malignancies were comparable, with a standardized incidence ratio (SIR) for malignancy of 0.99 across all treatment indications, the researchers said. In further analysis of malignancy by indication, the SIR was 0.87, 1.16, and 1.61 for psoriasis, PsA, and AS, respectively.
Data from postmarketing surveillance showed similar results: The estimated crude cumulative incidence reporting rate per 100 PTY was 0.27 for malignancy across all indications. The cumulative exposure was 285,811 PTY.
The study findings were limited by several factors including the post hoc design, differences in clinical trial methodologies, and lack of controlling for confounding variables, such as smoking status and previous exposure to systemic and biologic treatments, the researchers noted. In addition, the analysis did not include postexposure follow-up data, or data on patients who discontinued clinical trials, they said.
Overall, the analysis is the largest to date and supports the low risk of malignancy in patients with psoriasis, PsA, and AS treated with secukinumab, the researchers noted.
However, “while this assessment provides a broader understanding of the safety of secukinumab and supports its long-term use in these chronic systemic inflammatory conditions, registry data are further warranted to fully understand the real-world effect of biologics on malignancy risk,” they concluded.
“Secukinumab is a relatively newer biologic, approved in 2015, and there is currently a lack of longer-term data on the incidence of malignancy in secukinumab-treated patients, so it’s important to look at the data we have so far on this topic so we can better understand the long-term risks and counsel our psoriasis and psoriatic arthritis patients,” Flavia Fedeles, MD, of the department of dermatology at Massachusetts General Hospital, Boston, said in an interview.
Dr. Fedeles, who was not involved with the study, said that she was not surprised by the study results. “Data reported in the past from phase 3 clinical trials of secukinumab compared with placebo did not show an increase in risk of malignancy, though at that time no long-term safety data or data from patients with history of malignancy was available,” she said. “This study is reassuring in that there wasn’t a signal of increased malignancy events up to 5 years of secukinumab treatment,” said Dr. Fedeles.
However, she noted that the study has a number of limitations, including the use of clinical trials data, which have stringent inclusion/exclusion criteria that can lead to selection bias, the use of postmarketing surveillance data, the post hoc nature of the analysis, and the fact that the sponsor of the trial was the manufacturer of secukinumab, which “potentially can lead to bias to this study.”
She added that “registry data are needed to fully understand the real-world long-term effect of secukinumab on malignancy risk.”
The study was funded by Novartis. Lead author Dr. Lebwohl disclosed participating in advisory boards and/or as an investigator and/or speaker and receiving grants and/or honoraria from multiple companies including Novartis. Several study coauthors are employees of Novartis.
Dr. Fedeles had no financial conflicts to disclose.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
A pacemaker that 'just disappears' and a magnetic diet device
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Ignore this pacemaker and it will go away
At some point – and now seems to be that point – we have to say enough is enough. The throwaway culture that produces phones, TVs, and computers that get tossed in the trash because they can’t be repaired has gone too far. That’s right, we’re looking at you, medical science!
This time, it’s a pacemaker that just disappears when it’s no longer needed. Some lazy heart surgeon decided that it was way too much trouble to do another surgery to remove the leads when a temporary pacemaker was no longer needed. You know the type: “It sure would be nice if the pacemaker components were biocompatible and were naturally absorbed by the body over the course of a few weeks and wouldn’t need to be surgically extracted.” Slacker.
Well, get a load of this. Researchers at Northwestern and George Washington universities say that they have come up with a transient pacemaker that “harvests energy from an external, remote antenna using near-field communication protocols – the same technology used in smartphones for electronic payments and in RFID tags.”
That means no batteries and no wires that have to be removed and can cause infections. Because the infectious disease docs also are too lazy to do their jobs, apparently.
The lack of onboard infrastructure means that the device can be very small – it weighs less than half a gram and is only 250 microns thick. And yes, it is bioresorbable and completely harmless. It fully degrades and disappears in 5-7 weeks through the body’s natural biologic processes, “thereby avoiding the need for physical removal of the pacemaker electrodes. This is potentially a major victory for postoperative patients,” said Dr. Rishi Arora, one of the investigators.
A victory for patients, he says. Not a word about the time and effort saved by the surgeons. Typical.
It’s a mask! No, it’s a COVID-19 test!
Mask wearing has gotten more lax as people get vaccinated for COVID-19, but as wearing masks for virus prevention is becoming more normalized in western society, some saw an opportunity to make them work for diagnosis.
Researchers from the Massachusetts Institute of Technology and the Wyss Institute for Biologically Inspired Engineering at Harvard University have found a way to do just that with their wearable freeze-dried cell-free (wFDCF) technology. A single push of a button releases water from a reservoir in the mask that sequentially activates three different freeze-dried biological reactions, which detect the SARS-CoV-2 virus in the wearer’s breath.
Initially meant as a tool for the Zika outbreak in 2015, the team made a quick pivot in May 2020. But this isn’t just some run-of-the-mill, at-home test. The data prove that the wFDCF mask is comparable to polymerase chain reactions tests, the standard in COVID-19 detection. Plus there aren’t any extra factors to deal with, like room or instrument temperature to ensure accuracy. In just 90 minutes, the mask gives results on a readout in a way similar to that of a pregnancy test. Voilà! To have COVID-19 or not to have COVID-19 is an easily answered question.
At LOTME, we think this is a big improvement from having dogs, or even three-foot rats, sniffing out coronavirus.
But wait, there’s more. “In addition to face masks, our programmable biosensors can be integrated into other garments to provide on-the-go detection of dangerous substances including viruses, bacteria, toxins, and chemical agents,” said Peter Nguyen, PhD, study coauthor and research scientist at the Wyss Institute. The technology can be used on lab coats, scrubs, military uniforms, and uniforms of first responders who may come in contact with hazardous pathogens and toxins. Think of all the lives saved and possible avoidances.
If only it could diagnose bad breath.
Finally, an excuse for the all-beer diet
Weight loss is hard work. Extremely hard work, and, as evidenced by the constant inundation and advertisement of quick fixes, crash diets, and expensive gym memberships, there’s not really a solid, 100% solution to the issue. Until now, thanks to a team of doctors from New Zealand, who’ve decided that the best way to combat obesity is to leave you in constant agony.
The DentalSlim Diet Control device is certainly a radical yet comically logical attempt to combat obesity. The creators say that the biggest problem with dieting is compliance, and, well, it’s difficult to eat too much if you can’t actually open your mouth. The metal contraption is mounted onto your teeth and uses magnetic locks to prevent the user from opening their mouths more than 2 mm. That’s less than a tenth of an inch. Which is not a lot. So not a lot that essentially all you can consume is liquid.
Oh, and they’ve got results to back up their madness. In a small study, seven otherwise healthy obese women lost an average of 5.1% of their body weight after using the DentalSlim for 2 weeks, though they did complain that the device was difficult to use, caused discomfort and difficulty speaking, made them more tense, and in general made life “less satisfying.” And one participant was able to cheat the system and consume nonhealthy food like chocolate by melting it.
So, there you are, if you want a weight-loss solution that tortures you and has far bigger holes than the one it leaves for your mouth, try the DentalSlim. Or, you know, don’t eat that eighth slice of pizza and maybe go for a walk later. Your choice.
Almost all U.S. COVID-19 deaths now in the unvaccinated
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
If you, a friend, or a loved one remain unvaccinated against COVID-19 at this point – for whatever reason – you are at higher risk of dying if you become infected.
That’s the conclusion of a new report released by the Associated Press looking at COVID-19 deaths during May 2021.
Of more than 18,000 people who died from COVID-19, for example, only about 150 were fully vaccinated. That’s less than 1%.
“Recently, I was working in the emergency room [and] I saw a 21-year-old African American who came in with shortness of breath,” said Vino K. Palli, MD, MPH, a physician specializing in emergency medicine, internal medicine, and urgent care.
The patient rapidly deteriorated and required intubation and ventilation. She was transferred to a specialized hospital for possible extracorporeal membrane oxygenation (ECMO) treatment.
“This patient was unvaccinated, along with her entire family. This would have been easily preventable,” added Dr. Palli, who is also founder and CEO of MiDoctor Urgent Care in New York City.
“Vaccine misinformation, compounded with vaccine inertia and vaccine access, have contributed to this,” he added. “Even though we have a surplus amount of vaccines at this time, we are only seeing 50% to 55% of completely vaccinated patients.”
Authors of the Associated Press report also acknowledge that some people who are fully vaccinated can get a breakthrough infection. These occurred in fewer than 1,200 of more than 853,000 people hospitalized for COVID-19 in May, or about 0.1%.
The Associated Press came up with these numbers using data from the Centers for Disease Control and Prevention. The CDC tracks the numbers of cases, hospitalizations, and deaths but does not breakdown rates by vaccination status.
Stronger argument for vaccination?
“The fact that only 0.8% of COVID-19 deaths are in the fully vaccinated should persuade those people still hesitant about vaccination,” said Hugh Cassiere, MD, medical director of Respiratory Therapy Services at North Shore University Hospital in Manhasset, New York.
Stuart C. Ray, MD, professor of medicine and oncology in the Division of Infectious Diseases at Johns Hopkins University, Baltimore, agreed. “It seems compelling, even for skeptics, that unvaccinated people represent 99% of those now dying from COVID-19 when they represent less than 50% of the adult population in the United States.”
The findings from the study could be more persuasive than previous arguments made in favor of immunization, Dr. Ray said. “These recent findings of striking reductions in risk of death in the vaccinated are more directly attributable and harder to ignore or dismiss.”
Brian Labus, PhD, MPH, of the University of Nevada Las Vegas (UNLV) is less convinced. “While this might change some peoples’ minds, it probably won’t make a major difference. People have many different reasons for not getting vaccinated, and this is only one of the things they consider.”
The study adds information that was not available before, said Dr. Labus, assistant professor in the Department of Epidemiology and Biostatistics at the UNLV School of Public Health. “We study the vaccine under tightly controlled, ideal conditions. This is the evidence that it works as well in the real world as it did in the trials, and that is what is most important in implementing a vaccination program,” added Dr. Labus.
“The scientific data has honed in on one thing: Vaccines are effective in preventing hospitalizations, ICU admissions, ventilations, and deaths,” agreed Dr. Palli.
“We now know that almost all deaths occurred in patients who were not vaccinated. We also know that all vaccines are effective against various strains that are in circulation right now, including the Delta variant, which is rapidly spreading,” Dr. Palli said.
Dr. Cassiere pointed out that the unvaccinated are not only at higher risk of developing COVID-19 but also of spreading, being hospitalized for, and dying from the infection. Avoiding “long hauler” symptoms is another argument in favor of immunization, he added.
As of June 28, the CDC reports that 63% of Americans 12 years and older have received at least one dose of a COVID-19 vaccine, and 54% are fully vaccinated.
Worldwide worry?
Although overall rates of U.S. COVID-19 hospitalizations and deaths are down, the outlook may not remain as encouraging. “I hope I’m wrong about this, but I anticipate that the coming fall and winter will bring increasingly localized versions of similar findings – severe disease and death due to SARS-CoV-2 infection in regions or groups with lower vaccination rates,” Dr. Ray said.
There could be a silver lining, he added: “If this unfortunate surge occurs, the health and economic consequences seem likely to erode much of the remaining hesitancy regarding vaccination.”
The rise of more infectious SARS-CoV-2 variants, such as the Delta variant, could also throw a wrench in controlling COVID-19. “This isn’t just a domestic issue,” Dr. Ray said. “We have learned that the world is a small place in pandemic times.”
The Associated Press investigators state that their findings support the high efficacy of the vaccine. Also, given the current widespread availability of COVID-19 vaccines in the United States, they believe many of the COVID-19 deaths now occurring are preventable.
Public health measures should have continued longer to protect unvaccinated individuals, especially Black Americans, Hispanic Americans, and other minorities, Dr. Palli said. “Only time will tell if re-opening and abandoning all public health measures by the CDC was premature.”
A version of this article first appeared on Medscape.com.
Physician fired after slurs, including ‘cannibalism,’ against Israel
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.
Fidaa Wishah, MD, a pediatric radiologist at Phoenix Children’s Hospital in Arizona, has been fired after the hospital reviewed evidence that included her anti-Israel comments on social media, according to the hospital’s statement.
On May 26, Dr. Wishah posted, “We will uncover your thirst to kill our Palestinian children. … We sense your fear. The fear of your collapse. A state based on atrocity, inhumanity, racism and cannibalism never last long! Hey #israel … your end is coming sooner than you think.”
Phoenix Children’s Hospital did not respond to this news organization’s request for comment but said in a statement to the Jewish News Syndicate : “After a thorough review of the facts related to this matter, this individual is no longer providing care at Phoenix Children’s. All children in the care of Phoenix Children’s receive hope, healing and the best possible health care, regardless of race, color, disability, religion, gender, gender identity, sexual orientation or national origin.”
Dr. Wishah’s profile has been removed from the hospital website. Her LinkedIn profile indicates she had been a pediatric radiology fellow at Stanford (Calif.) University, specializing in advanced magnetic resonance imaging and fetal imaging and had been a senior staff pediatric radiologist at Henry Ford Health System in Detroit.
It wasn’t the first time antisemitic comments have led to the firing of a physician. Last year, this news organization wrote about Lara Kollab, DO, a first-year resident fired for her antisemitic tweets. She was subsequently barred from medicine.
In the same post from May 26, Dr. Wishah also wrote: “We will not be #censored anymore! Bomb our media buildings and we have the phones[.] Bribe the mainstream media and we have our small #socialmedia platforms[.] From our windows ... from our streets ... next the rubble we will expose you to the world[.] We will expose the #massacre and #genocide you #zionists are proud of[.]”
Today, CAIR-AZ, a group whose mission is to “enhance understanding of Islam, protect civil rights, promote justice, and empower American Muslims,” according to its website, announced that it, along with three private law firms, will represent Dr. Wishah in what they referred to as “her wrongful termination case against Phoenix Children’s Hospital.”
The announcement, which mentions that Dr. Wishah was born and raised in Gaza, said, “Dr. Wishah has been a medical doctor since 2010 and has spent the vast majority of her career as a pediatric physician. Despite caring for thousands of children, many of whom are Jewish, she has never been accused of discriminating against any of her patients or colleagues.”
The statement added, “PCH’s decision to terminate Dr. Wishah is shameful and an attack on freedom of speech.”
A version of this article first appeared on Medscape.com.