User login
ADHD in preschool kids: Adrenergic agonists may be a better fit
A new study finds that alpha2-adrenergic agonists may be of benefit and have fewer side effects than stimulant medications for the treatment of attention-deficit/hyperactivity disorder in preschool-age children.
The study was published online May 4 in JAMA.
As part of a retrospective analysis, Elizabeth Harstad, MD, MPH, of Boston Children’s Hospital and colleagues evaluated health record data from 497 preschool-age children with ADHD across seven developmental-behavioral pediatric practices in the United States. Children included in the evaluation were younger than 6 years and were treated for ADHD between Jan. 1, 2013, and July 1, 2017, with either an alpha2-adrenergic agonist or a stimulant.
Overall, 175 children (35%) were prescribed an alpha2-adrenergic agonist (most often guanfacine) as first-line ADHD medication, and 322 children (65%) were prescribed a stimulant (most often a methylphenidate-based preparation). Before any medication regimens were initiated, 62% of children received behavioral therapy.
“These findings suggest that for some children there may be a concern about either how well a stimulant will work or how well a stimulant will be tolerated that is leading clinicians to instead prescribe an alpha2-adrenergic agonist as the first medication tried,” Dr. Harstad said in an interview.
Clinical improvement was noted in 66% of children treated with alpha2-adrenergic agonists (95% confidence interval, 57.5%-73.9%) and in 78% of children treated with stimulants (95% CI, 72.4%-83.4%).
Most adverse effects were more common among children who received stimulants than among those who received alpha2-adrenergic agonists. These adverse effects included difficulty falling asleep (21% vs. 11%), decreased appetite (38% vs. 7%), increased stomachaches (13% vs. 5%), and increased skin picking/repetitive behaviors (11% vs. 5%). Only daytime sleepiness was more frequent among children who received an alpha2-adrenergic agonist rather than a stimulant (38% vs. 3%).
“We also found that for the youngest children (<4 years old), those initiated on alpha2-adrenergic agonists stayed on these medications longer than those initiated on stimulants, which may indicate that they are better tolerated, although more research is needed to confirm this,” Dr. Harstad said.
“While our study focused on how well medications work and how well they are tolerated when used to treat preschool-age children with ADHD, it is important to remember that behavioral therapy is recommended as first-line treatment for ADHD in preschool-age children, not medication,” Dr. Harstad added.
Mark Wolraich, MD, of the University of Oklahoma, echoed that sentiment. “The article mentions that behavioral interventions, in the form of parent training in behavior management, is an effective first-line treatment” and, per the American Academy of Pediatrics guidelines, “is the first line of treatment recommended for preschool-age children before medication should be considered.”
Dr. Wolraich also noted that “neither drug has official FDA [U.S. Food and Drug Administration] approval in this age group” but that “methylphenidate comes the closest to having met the FDA requirements for approval in this age group, which is why the AAP guidelines recommended its use if parent training in behavior management is not sufficient.”
Although Dr. Harstad and colleagues note that the study included a large and diverse sample size from across the United States, they acknowledge that “further research, including from randomized clinical trials, is needed to assess comparative effectiveness of alpha2-adrenergic agonists versus stimulants.”
Funding for the study was provided through a cooperative agreement with the Maternal and Child Health Bureau, the Health Resources and Services Administration, and the U.S. Department of Health & Human Services. Dr. Harstad has reported receiving reported receiving compensation for serving as a medical reviewer for Understood.org and grant funding from the Palmer Family Fund for Autism Research to conduct research related to autism spectrum disorder at Boston Children’s Hospital. Disclosures for the other authors are listed in the original article. Dr. Wolraich has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study finds that alpha2-adrenergic agonists may be of benefit and have fewer side effects than stimulant medications for the treatment of attention-deficit/hyperactivity disorder in preschool-age children.
The study was published online May 4 in JAMA.
As part of a retrospective analysis, Elizabeth Harstad, MD, MPH, of Boston Children’s Hospital and colleagues evaluated health record data from 497 preschool-age children with ADHD across seven developmental-behavioral pediatric practices in the United States. Children included in the evaluation were younger than 6 years and were treated for ADHD between Jan. 1, 2013, and July 1, 2017, with either an alpha2-adrenergic agonist or a stimulant.
Overall, 175 children (35%) were prescribed an alpha2-adrenergic agonist (most often guanfacine) as first-line ADHD medication, and 322 children (65%) were prescribed a stimulant (most often a methylphenidate-based preparation). Before any medication regimens were initiated, 62% of children received behavioral therapy.
“These findings suggest that for some children there may be a concern about either how well a stimulant will work or how well a stimulant will be tolerated that is leading clinicians to instead prescribe an alpha2-adrenergic agonist as the first medication tried,” Dr. Harstad said in an interview.
Clinical improvement was noted in 66% of children treated with alpha2-adrenergic agonists (95% confidence interval, 57.5%-73.9%) and in 78% of children treated with stimulants (95% CI, 72.4%-83.4%).
Most adverse effects were more common among children who received stimulants than among those who received alpha2-adrenergic agonists. These adverse effects included difficulty falling asleep (21% vs. 11%), decreased appetite (38% vs. 7%), increased stomachaches (13% vs. 5%), and increased skin picking/repetitive behaviors (11% vs. 5%). Only daytime sleepiness was more frequent among children who received an alpha2-adrenergic agonist rather than a stimulant (38% vs. 3%).
“We also found that for the youngest children (<4 years old), those initiated on alpha2-adrenergic agonists stayed on these medications longer than those initiated on stimulants, which may indicate that they are better tolerated, although more research is needed to confirm this,” Dr. Harstad said.
“While our study focused on how well medications work and how well they are tolerated when used to treat preschool-age children with ADHD, it is important to remember that behavioral therapy is recommended as first-line treatment for ADHD in preschool-age children, not medication,” Dr. Harstad added.
Mark Wolraich, MD, of the University of Oklahoma, echoed that sentiment. “The article mentions that behavioral interventions, in the form of parent training in behavior management, is an effective first-line treatment” and, per the American Academy of Pediatrics guidelines, “is the first line of treatment recommended for preschool-age children before medication should be considered.”
Dr. Wolraich also noted that “neither drug has official FDA [U.S. Food and Drug Administration] approval in this age group” but that “methylphenidate comes the closest to having met the FDA requirements for approval in this age group, which is why the AAP guidelines recommended its use if parent training in behavior management is not sufficient.”
Although Dr. Harstad and colleagues note that the study included a large and diverse sample size from across the United States, they acknowledge that “further research, including from randomized clinical trials, is needed to assess comparative effectiveness of alpha2-adrenergic agonists versus stimulants.”
Funding for the study was provided through a cooperative agreement with the Maternal and Child Health Bureau, the Health Resources and Services Administration, and the U.S. Department of Health & Human Services. Dr. Harstad has reported receiving reported receiving compensation for serving as a medical reviewer for Understood.org and grant funding from the Palmer Family Fund for Autism Research to conduct research related to autism spectrum disorder at Boston Children’s Hospital. Disclosures for the other authors are listed in the original article. Dr. Wolraich has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study finds that alpha2-adrenergic agonists may be of benefit and have fewer side effects than stimulant medications for the treatment of attention-deficit/hyperactivity disorder in preschool-age children.
The study was published online May 4 in JAMA.
As part of a retrospective analysis, Elizabeth Harstad, MD, MPH, of Boston Children’s Hospital and colleagues evaluated health record data from 497 preschool-age children with ADHD across seven developmental-behavioral pediatric practices in the United States. Children included in the evaluation were younger than 6 years and were treated for ADHD between Jan. 1, 2013, and July 1, 2017, with either an alpha2-adrenergic agonist or a stimulant.
Overall, 175 children (35%) were prescribed an alpha2-adrenergic agonist (most often guanfacine) as first-line ADHD medication, and 322 children (65%) were prescribed a stimulant (most often a methylphenidate-based preparation). Before any medication regimens were initiated, 62% of children received behavioral therapy.
“These findings suggest that for some children there may be a concern about either how well a stimulant will work or how well a stimulant will be tolerated that is leading clinicians to instead prescribe an alpha2-adrenergic agonist as the first medication tried,” Dr. Harstad said in an interview.
Clinical improvement was noted in 66% of children treated with alpha2-adrenergic agonists (95% confidence interval, 57.5%-73.9%) and in 78% of children treated with stimulants (95% CI, 72.4%-83.4%).
Most adverse effects were more common among children who received stimulants than among those who received alpha2-adrenergic agonists. These adverse effects included difficulty falling asleep (21% vs. 11%), decreased appetite (38% vs. 7%), increased stomachaches (13% vs. 5%), and increased skin picking/repetitive behaviors (11% vs. 5%). Only daytime sleepiness was more frequent among children who received an alpha2-adrenergic agonist rather than a stimulant (38% vs. 3%).
“We also found that for the youngest children (<4 years old), those initiated on alpha2-adrenergic agonists stayed on these medications longer than those initiated on stimulants, which may indicate that they are better tolerated, although more research is needed to confirm this,” Dr. Harstad said.
“While our study focused on how well medications work and how well they are tolerated when used to treat preschool-age children with ADHD, it is important to remember that behavioral therapy is recommended as first-line treatment for ADHD in preschool-age children, not medication,” Dr. Harstad added.
Mark Wolraich, MD, of the University of Oklahoma, echoed that sentiment. “The article mentions that behavioral interventions, in the form of parent training in behavior management, is an effective first-line treatment” and, per the American Academy of Pediatrics guidelines, “is the first line of treatment recommended for preschool-age children before medication should be considered.”
Dr. Wolraich also noted that “neither drug has official FDA [U.S. Food and Drug Administration] approval in this age group” but that “methylphenidate comes the closest to having met the FDA requirements for approval in this age group, which is why the AAP guidelines recommended its use if parent training in behavior management is not sufficient.”
Although Dr. Harstad and colleagues note that the study included a large and diverse sample size from across the United States, they acknowledge that “further research, including from randomized clinical trials, is needed to assess comparative effectiveness of alpha2-adrenergic agonists versus stimulants.”
Funding for the study was provided through a cooperative agreement with the Maternal and Child Health Bureau, the Health Resources and Services Administration, and the U.S. Department of Health & Human Services. Dr. Harstad has reported receiving reported receiving compensation for serving as a medical reviewer for Understood.org and grant funding from the Palmer Family Fund for Autism Research to conduct research related to autism spectrum disorder at Boston Children’s Hospital. Disclosures for the other authors are listed in the original article. Dr. Wolraich has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Keep antibiotics unchanged in breakthrough UTIs
Changing the continuous antibiotic prophylactic agent had no significant effect on the risk of a second infection in children with breakthrough urinary tract infections (UTIs), based on data from 62 children treated at a single center.
Continuous antibiotic prophylaxis (CAP) is often used for UTI prevention in children with febrile UTIs or anomalies that predispose them to UTIs, such as vesicoureteral reflux (VUR) or bladder and bowel dysfunction, said Lane M. Shish, MPH, of the University of Washington, Bothell, and colleagues in a poster (#1245) presented at the Pediatric Academic Societies annual meeting.
CAP, once initiated, is used until a planned endpoint or a breakthrough UTI, at which point alternative treatments usually include surgical intervention or a CAP agent change, the researchers said. However, changing the CAP agent is based on consensus without evidence of benefit, they noted.
To evaluate the potential effect of switching or maintaining CAP in cases of breakthrough UTIs, the researchers conducted a retrospective cohort study of all patients younger than 18 years on CAP for UTI prevention enrolled in a pediatric urology registry between January 2013 and August 2020.
All patients experienced a breakthrough UTI while on CAP; CAP was changed for 24 patients and left unchanged for 38 patients.
The primary outcome of second-breakthrough infections occurred in 12 of the changed CAP group and 22 of the unchanged group, with a relative risk of 0.86. The percentage of second breakthrough UTIs resistant to the current CAP was not significantly different between the changed and unchanged CAP groups (75% vs. 77%; P = 0.88).
The researchers also identified a rate ratio of 0.67 for a second breakthrough UTI in the changed CAP group, and found that approximately one-third of these patients (33.3%) developed antibiotic resistance to their initial antibiotic agent and the changed antibiotic agent.
The study findings were limited by several factors, including the retrospective design and small sample size, the researchers noted.
However, the results suggest that changing the CAP after an initial breakthrough UTI in children did not increase the risk of a second breakthrough UTI, and that CAP changing did introduce a risk of developing a second UTI with increased CAP resistance, the researchers noted. The results support leaving a child’s CAP unchanged after an initial breakthrough UTI, although additional research is needed to verify the findings, including studies involving a larger cohort with a multi-institutional prospective evaluation, they concluded.
Manage UTIs to reduce recurrence and resistance
“As we know, avoiding recurrent UTIs is important in preserving renal function in pediatric patients,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
“Avoiding recurrent UTIs is also important to avoid the development and spread of multidrug-resistant organisms,” he said.
Dr. Joos said he was surprised by some of the study findings. “I was surprised that, over the course of this 7-year retrospective review, overall only approximately 50% of patients with a first breakthrough UTI on CAP developed a second breakthrough UTI,” he noted. “Also, the relative risk of a second UTI was not significantly affected by whether the CAP antibiotic was changed after the first infection,” he said. “It would be interesting to see whether these results hold up in a randomized, prospective study,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Joos had no financial conflicts to disclose, but serves as a member of the Pediatric News Editorial Advisory Board.
Changing the continuous antibiotic prophylactic agent had no significant effect on the risk of a second infection in children with breakthrough urinary tract infections (UTIs), based on data from 62 children treated at a single center.
Continuous antibiotic prophylaxis (CAP) is often used for UTI prevention in children with febrile UTIs or anomalies that predispose them to UTIs, such as vesicoureteral reflux (VUR) or bladder and bowel dysfunction, said Lane M. Shish, MPH, of the University of Washington, Bothell, and colleagues in a poster (#1245) presented at the Pediatric Academic Societies annual meeting.
CAP, once initiated, is used until a planned endpoint or a breakthrough UTI, at which point alternative treatments usually include surgical intervention or a CAP agent change, the researchers said. However, changing the CAP agent is based on consensus without evidence of benefit, they noted.
To evaluate the potential effect of switching or maintaining CAP in cases of breakthrough UTIs, the researchers conducted a retrospective cohort study of all patients younger than 18 years on CAP for UTI prevention enrolled in a pediatric urology registry between January 2013 and August 2020.
All patients experienced a breakthrough UTI while on CAP; CAP was changed for 24 patients and left unchanged for 38 patients.
The primary outcome of second-breakthrough infections occurred in 12 of the changed CAP group and 22 of the unchanged group, with a relative risk of 0.86. The percentage of second breakthrough UTIs resistant to the current CAP was not significantly different between the changed and unchanged CAP groups (75% vs. 77%; P = 0.88).
The researchers also identified a rate ratio of 0.67 for a second breakthrough UTI in the changed CAP group, and found that approximately one-third of these patients (33.3%) developed antibiotic resistance to their initial antibiotic agent and the changed antibiotic agent.
The study findings were limited by several factors, including the retrospective design and small sample size, the researchers noted.
However, the results suggest that changing the CAP after an initial breakthrough UTI in children did not increase the risk of a second breakthrough UTI, and that CAP changing did introduce a risk of developing a second UTI with increased CAP resistance, the researchers noted. The results support leaving a child’s CAP unchanged after an initial breakthrough UTI, although additional research is needed to verify the findings, including studies involving a larger cohort with a multi-institutional prospective evaluation, they concluded.
Manage UTIs to reduce recurrence and resistance
“As we know, avoiding recurrent UTIs is important in preserving renal function in pediatric patients,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
“Avoiding recurrent UTIs is also important to avoid the development and spread of multidrug-resistant organisms,” he said.
Dr. Joos said he was surprised by some of the study findings. “I was surprised that, over the course of this 7-year retrospective review, overall only approximately 50% of patients with a first breakthrough UTI on CAP developed a second breakthrough UTI,” he noted. “Also, the relative risk of a second UTI was not significantly affected by whether the CAP antibiotic was changed after the first infection,” he said. “It would be interesting to see whether these results hold up in a randomized, prospective study,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Joos had no financial conflicts to disclose, but serves as a member of the Pediatric News Editorial Advisory Board.
Changing the continuous antibiotic prophylactic agent had no significant effect on the risk of a second infection in children with breakthrough urinary tract infections (UTIs), based on data from 62 children treated at a single center.
Continuous antibiotic prophylaxis (CAP) is often used for UTI prevention in children with febrile UTIs or anomalies that predispose them to UTIs, such as vesicoureteral reflux (VUR) or bladder and bowel dysfunction, said Lane M. Shish, MPH, of the University of Washington, Bothell, and colleagues in a poster (#1245) presented at the Pediatric Academic Societies annual meeting.
CAP, once initiated, is used until a planned endpoint or a breakthrough UTI, at which point alternative treatments usually include surgical intervention or a CAP agent change, the researchers said. However, changing the CAP agent is based on consensus without evidence of benefit, they noted.
To evaluate the potential effect of switching or maintaining CAP in cases of breakthrough UTIs, the researchers conducted a retrospective cohort study of all patients younger than 18 years on CAP for UTI prevention enrolled in a pediatric urology registry between January 2013 and August 2020.
All patients experienced a breakthrough UTI while on CAP; CAP was changed for 24 patients and left unchanged for 38 patients.
The primary outcome of second-breakthrough infections occurred in 12 of the changed CAP group and 22 of the unchanged group, with a relative risk of 0.86. The percentage of second breakthrough UTIs resistant to the current CAP was not significantly different between the changed and unchanged CAP groups (75% vs. 77%; P = 0.88).
The researchers also identified a rate ratio of 0.67 for a second breakthrough UTI in the changed CAP group, and found that approximately one-third of these patients (33.3%) developed antibiotic resistance to their initial antibiotic agent and the changed antibiotic agent.
The study findings were limited by several factors, including the retrospective design and small sample size, the researchers noted.
However, the results suggest that changing the CAP after an initial breakthrough UTI in children did not increase the risk of a second breakthrough UTI, and that CAP changing did introduce a risk of developing a second UTI with increased CAP resistance, the researchers noted. The results support leaving a child’s CAP unchanged after an initial breakthrough UTI, although additional research is needed to verify the findings, including studies involving a larger cohort with a multi-institutional prospective evaluation, they concluded.
Manage UTIs to reduce recurrence and resistance
“As we know, avoiding recurrent UTIs is important in preserving renal function in pediatric patients,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
“Avoiding recurrent UTIs is also important to avoid the development and spread of multidrug-resistant organisms,” he said.
Dr. Joos said he was surprised by some of the study findings. “I was surprised that, over the course of this 7-year retrospective review, overall only approximately 50% of patients with a first breakthrough UTI on CAP developed a second breakthrough UTI,” he noted. “Also, the relative risk of a second UTI was not significantly affected by whether the CAP antibiotic was changed after the first infection,” he said. “It would be interesting to see whether these results hold up in a randomized, prospective study,” he added.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Joos had no financial conflicts to disclose, but serves as a member of the Pediatric News Editorial Advisory Board.
FROM PAS 2021
Attending a patient’s funeral: How psychiatrists decide
Psychiatrists often develop long-term relationships with their patients, but what happens when a patient dies? Should the psychiatrist attend the patient’s funeral?
It’s a question Ashley Pettaway, MD, faced as a medical resident at the University of Alabama School of Medicine.
For 2 months, Dr. Pettaway was involved in the day-to-day care of a woman in her 40s who ultimately died. As part of that care, Dr. Pettaway had regular meetings with the patient’s husband and family members.
“The patient was about my mother’s age, so I naturally was kind of attached to her,” Dr. Pettaway told this news organization. After she died, her family invited Dr. Pettaway to the funeral.
“While I couldn’t make it to the funeral, it got me thinking. Should I go? If I go, what do I say? Who do I sit with? How do I introduce myself?” wondered Dr. Pettaway, now a resident in the department of psychiatry and neurobehavioral sciences, University of Virginia, Charlottesville.
She turned to the literature but found very little regarding psychiatrists attending their patients’ funerals. “This was surprising to me because in psychiatry, you can get so engrossed in patients’ lives,” Dr. Pettaway said.
Given the lack of rules or formal guidance on psychiatrists attending patients’ funerals, Dr. Pettaway and her mentor, Gabrielle Marzani, MD, conducted an informal survey of 12 supervising psychiatrists at the University of Virginia.
The survey results were presented at the virtual American Psychiatric Association 2021 Annual Meeting.
Ten of the 12 psychiatrists who were surveyed were caring for a patient who died while under their care. Five of those psychiatrists reported going to at least one patient’s funeral over the course of their career.
Among the psychiatrists who attended a patient’s funeral, their attendance was often based on their clinical intuition, their relationship with the family, or whether the patient was an established presence in the community. In the latter case, the psychiatrist attended as a community member.
The number of years in practice also mattered. Fewer senior faculty reported that they would be hesitant to attend and that they would not attend without a formal invitation from the family. Senior career psychiatrists were more likely to attend and felt that an invitation was not required.
None of the psychiatrists surveyed had received training or guidance on attending patients’ funerals at any point in their career.
Given the absence of formal recommendations, Dr. Pettaway believes increased conversation on this topic as part of residency training programs would help psychiatrists navigate these complex situations.
A complex issue
Commenting on the topic for an interview, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, said this is an “interesting and important topic that is underdiscussed.”
“I don’t think there’s a right answer that applies to every situation,” said Dr. Appelbaum, a past president of the APA.
There will be times, he said, when psychiatrists or other mental health professionals have worked closely with a patient for many years and may have interacted with the family over that period.
“When that patient passes away, they may feel, and the family may feel, that it would be comforting and appropriate for them to be at the funeral,” said Dr. Appelbaum.
However, he added,
“There are obviously a number of complexities involved. One is how the family feels about the relationship with the psychiatrist – whether they were accepting of the reality that the patient had a mental disorder and was in treatment,” he said.
There is also the question of confidentiality, said Dr. Appelbaum.
“If it’s a large funeral and the psychiatrist is just one face in the crowd, that’s not likely to be an issue. But if it’s a relatively small group of mourners, all of whom know each other, and an unknown figure pops up, that could raise questions and perhaps inadvertently reveal to family members or friends that the deceased had a psychiatric condition and was in treatment. That needs to be taken into account as well,” he added.
In cases in which the family invites the psychiatrist, confidentiality is not a concern, and attendance by the psychiatrist is something the patient would have wanted, said Dr. Appelbaum.
How the patient died may also be factor. When a patient dies by suicide, it’s an “emotionally charged situation for both sides,” said Dr. Appelbaum.
In the case of a suicide, he noted, the deceased was often an active patient, and both the psychiatrist and the family are dealing with strong emotions – the psychiatrist with regret over loss of the patient and perhaps with questions as to what could have been done differently, and the family with sorrow but “also sometimes with suspicion or anger in that the psychiatrist somehow failed to keep the patient alive,” Dr. Appelbaum noted.
“In this situation, it’s even more crucial for the psychiatrist or other mental health professionals to take the lead from the family – perhaps to initiate contact to express condolences and inquire delicately about the funeral arrangements and whether their presence would be welcomed,” he said.
The research had no specific funding. Dr. Pettaway and Dr. Appelbaum have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psychiatrists often develop long-term relationships with their patients, but what happens when a patient dies? Should the psychiatrist attend the patient’s funeral?
It’s a question Ashley Pettaway, MD, faced as a medical resident at the University of Alabama School of Medicine.
For 2 months, Dr. Pettaway was involved in the day-to-day care of a woman in her 40s who ultimately died. As part of that care, Dr. Pettaway had regular meetings with the patient’s husband and family members.
“The patient was about my mother’s age, so I naturally was kind of attached to her,” Dr. Pettaway told this news organization. After she died, her family invited Dr. Pettaway to the funeral.
“While I couldn’t make it to the funeral, it got me thinking. Should I go? If I go, what do I say? Who do I sit with? How do I introduce myself?” wondered Dr. Pettaway, now a resident in the department of psychiatry and neurobehavioral sciences, University of Virginia, Charlottesville.
She turned to the literature but found very little regarding psychiatrists attending their patients’ funerals. “This was surprising to me because in psychiatry, you can get so engrossed in patients’ lives,” Dr. Pettaway said.
Given the lack of rules or formal guidance on psychiatrists attending patients’ funerals, Dr. Pettaway and her mentor, Gabrielle Marzani, MD, conducted an informal survey of 12 supervising psychiatrists at the University of Virginia.
The survey results were presented at the virtual American Psychiatric Association 2021 Annual Meeting.
Ten of the 12 psychiatrists who were surveyed were caring for a patient who died while under their care. Five of those psychiatrists reported going to at least one patient’s funeral over the course of their career.
Among the psychiatrists who attended a patient’s funeral, their attendance was often based on their clinical intuition, their relationship with the family, or whether the patient was an established presence in the community. In the latter case, the psychiatrist attended as a community member.
The number of years in practice also mattered. Fewer senior faculty reported that they would be hesitant to attend and that they would not attend without a formal invitation from the family. Senior career psychiatrists were more likely to attend and felt that an invitation was not required.
None of the psychiatrists surveyed had received training or guidance on attending patients’ funerals at any point in their career.
Given the absence of formal recommendations, Dr. Pettaway believes increased conversation on this topic as part of residency training programs would help psychiatrists navigate these complex situations.
A complex issue
Commenting on the topic for an interview, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, said this is an “interesting and important topic that is underdiscussed.”
“I don’t think there’s a right answer that applies to every situation,” said Dr. Appelbaum, a past president of the APA.
There will be times, he said, when psychiatrists or other mental health professionals have worked closely with a patient for many years and may have interacted with the family over that period.
“When that patient passes away, they may feel, and the family may feel, that it would be comforting and appropriate for them to be at the funeral,” said Dr. Appelbaum.
However, he added,
“There are obviously a number of complexities involved. One is how the family feels about the relationship with the psychiatrist – whether they were accepting of the reality that the patient had a mental disorder and was in treatment,” he said.
There is also the question of confidentiality, said Dr. Appelbaum.
“If it’s a large funeral and the psychiatrist is just one face in the crowd, that’s not likely to be an issue. But if it’s a relatively small group of mourners, all of whom know each other, and an unknown figure pops up, that could raise questions and perhaps inadvertently reveal to family members or friends that the deceased had a psychiatric condition and was in treatment. That needs to be taken into account as well,” he added.
In cases in which the family invites the psychiatrist, confidentiality is not a concern, and attendance by the psychiatrist is something the patient would have wanted, said Dr. Appelbaum.
How the patient died may also be factor. When a patient dies by suicide, it’s an “emotionally charged situation for both sides,” said Dr. Appelbaum.
In the case of a suicide, he noted, the deceased was often an active patient, and both the psychiatrist and the family are dealing with strong emotions – the psychiatrist with regret over loss of the patient and perhaps with questions as to what could have been done differently, and the family with sorrow but “also sometimes with suspicion or anger in that the psychiatrist somehow failed to keep the patient alive,” Dr. Appelbaum noted.
“In this situation, it’s even more crucial for the psychiatrist or other mental health professionals to take the lead from the family – perhaps to initiate contact to express condolences and inquire delicately about the funeral arrangements and whether their presence would be welcomed,” he said.
The research had no specific funding. Dr. Pettaway and Dr. Appelbaum have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Psychiatrists often develop long-term relationships with their patients, but what happens when a patient dies? Should the psychiatrist attend the patient’s funeral?
It’s a question Ashley Pettaway, MD, faced as a medical resident at the University of Alabama School of Medicine.
For 2 months, Dr. Pettaway was involved in the day-to-day care of a woman in her 40s who ultimately died. As part of that care, Dr. Pettaway had regular meetings with the patient’s husband and family members.
“The patient was about my mother’s age, so I naturally was kind of attached to her,” Dr. Pettaway told this news organization. After she died, her family invited Dr. Pettaway to the funeral.
“While I couldn’t make it to the funeral, it got me thinking. Should I go? If I go, what do I say? Who do I sit with? How do I introduce myself?” wondered Dr. Pettaway, now a resident in the department of psychiatry and neurobehavioral sciences, University of Virginia, Charlottesville.
She turned to the literature but found very little regarding psychiatrists attending their patients’ funerals. “This was surprising to me because in psychiatry, you can get so engrossed in patients’ lives,” Dr. Pettaway said.
Given the lack of rules or formal guidance on psychiatrists attending patients’ funerals, Dr. Pettaway and her mentor, Gabrielle Marzani, MD, conducted an informal survey of 12 supervising psychiatrists at the University of Virginia.
The survey results were presented at the virtual American Psychiatric Association 2021 Annual Meeting.
Ten of the 12 psychiatrists who were surveyed were caring for a patient who died while under their care. Five of those psychiatrists reported going to at least one patient’s funeral over the course of their career.
Among the psychiatrists who attended a patient’s funeral, their attendance was often based on their clinical intuition, their relationship with the family, or whether the patient was an established presence in the community. In the latter case, the psychiatrist attended as a community member.
The number of years in practice also mattered. Fewer senior faculty reported that they would be hesitant to attend and that they would not attend without a formal invitation from the family. Senior career psychiatrists were more likely to attend and felt that an invitation was not required.
None of the psychiatrists surveyed had received training or guidance on attending patients’ funerals at any point in their career.
Given the absence of formal recommendations, Dr. Pettaway believes increased conversation on this topic as part of residency training programs would help psychiatrists navigate these complex situations.
A complex issue
Commenting on the topic for an interview, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University, New York, said this is an “interesting and important topic that is underdiscussed.”
“I don’t think there’s a right answer that applies to every situation,” said Dr. Appelbaum, a past president of the APA.
There will be times, he said, when psychiatrists or other mental health professionals have worked closely with a patient for many years and may have interacted with the family over that period.
“When that patient passes away, they may feel, and the family may feel, that it would be comforting and appropriate for them to be at the funeral,” said Dr. Appelbaum.
However, he added,
“There are obviously a number of complexities involved. One is how the family feels about the relationship with the psychiatrist – whether they were accepting of the reality that the patient had a mental disorder and was in treatment,” he said.
There is also the question of confidentiality, said Dr. Appelbaum.
“If it’s a large funeral and the psychiatrist is just one face in the crowd, that’s not likely to be an issue. But if it’s a relatively small group of mourners, all of whom know each other, and an unknown figure pops up, that could raise questions and perhaps inadvertently reveal to family members or friends that the deceased had a psychiatric condition and was in treatment. That needs to be taken into account as well,” he added.
In cases in which the family invites the psychiatrist, confidentiality is not a concern, and attendance by the psychiatrist is something the patient would have wanted, said Dr. Appelbaum.
How the patient died may also be factor. When a patient dies by suicide, it’s an “emotionally charged situation for both sides,” said Dr. Appelbaum.
In the case of a suicide, he noted, the deceased was often an active patient, and both the psychiatrist and the family are dealing with strong emotions – the psychiatrist with regret over loss of the patient and perhaps with questions as to what could have been done differently, and the family with sorrow but “also sometimes with suspicion or anger in that the psychiatrist somehow failed to keep the patient alive,” Dr. Appelbaum noted.
“In this situation, it’s even more crucial for the psychiatrist or other mental health professionals to take the lead from the family – perhaps to initiate contact to express condolences and inquire delicately about the funeral arrangements and whether their presence would be welcomed,” he said.
The research had no specific funding. Dr. Pettaway and Dr. Appelbaum have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA clears cap device for ‘smart’ insulin pens for diabetes
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
Support group for Asian Americans uses theater to cope with COVID
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Palliative care in the pandemic: How one hospital met the challenge
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
FROM SGIM 2021
FDA blazes path for ‘real-world’ evidence as proof of efficacy
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
Impact of the COVID-19 Pandemic on Multiple Sclerosis Care for Veterans
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
Fresh look at ISCHEMIA bolsters conservative message in stable CAD
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.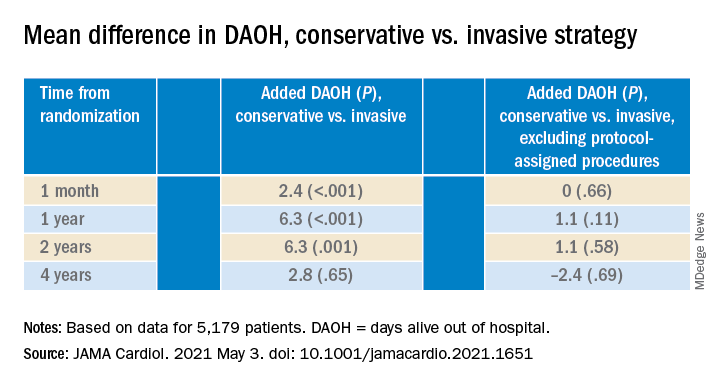
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
Screening High-Risk Women Veterans for Breast Cancer
The number of women seeking care from the Veterans Health Administration (VHA) is increasing.1 In 2015, there were 2 million women veterans in the United States, which is 9.4% of the total veteran population. This group is expected to increase at an average of about 18,000 women per year for the next 10 years.2 The percentage of women veterans who are US Department of Veterans Affairs (VA) users aged 45 to 64 years rose 46% from 2000 to 2015.1,3-4 It is estimated that 15% of veterans who used VA services in 2020 were women.1 Nineteen percent of women veterans are Black.1 The median age of women veterans in 2015 was 50 years.5 Breast cancer is the leading cancer affecting female veterans, and data suggest they have an increased risk of breast cancer based on unique service-related exposures.1,6-9
In the US, about 10 million women are eligible for breast cancer preventive therapy, including, but not limited to, medications, surgery, or lifestyle changes.10 Secondary prevention options include change in surveillance that can reduce their risk or identify cancer at an earlier stage when treatment is more effective. The United States Preventive Services Task Force, the National Comprehensive Cancer Network, the American Society for Clinical Oncology, the National Institute for Health and Care Excellence, and the Oncology Nursing Society recommend screening women aged ≥ 35 years to assess breast cancer risk.11-18 If a woman is at increased risk, she may be a candidate for chemoprevention, prozphylactic surgery, and possibly an enhanced screening regimen.
Urban and minority women are an understudied population. Most veterans (75%) live in urban or suburban settings.19,20 Urban veteran women constitute an important potential study population.
Chemoprevention measures have been underused because of factors involving both women and their health care providers. A large proportion of women are unaware of their higher risk status due to lack of adequate screening and risk assessment.21,22 In addition to patient lack of awareness of their high-risk status, primary care physicians are also reluctant to prescribe chemopreventive agents due to a lack of comfort or familiarity with the risks and benefits.23-26 The STAR2015, BCPT2005, IBIS2014, MAP3 2011, IBIS-I 2014, and IBIS II 2014 studies clearly demonstrate a 49 to 62% reduction in risk for women using chemoprevention such as selective estrogen receptor modulators or aromatase inhibitors, respectively.27-32 Yet only 4 to 9% of high-risk women not enrolled in a clinical trial are using chemoprevention.33-39
The possibility of developing breast cancer also may be increased because of a positive family history or being a member of a family in which there is a known susceptibility gene mutation.40 Based on these risk factors, women may be eligible for tailored follow-up and genetic counseling.41-44
Nationally, 7 to 10% of the civilian US population will experience posttraumatic stress disorder (PTSD).45 The rates are remarkably higher for women veterans, with roughly 20% diagnosed with PTSD.46,47 Anxiety and PTSD have been implicated in poor adherence to medical advice.48,49
In 2014, a national VA multidisciplinary group focused on breast cancer prevention, detection, treatment, and research to address breast health in the growing population of women veterans. High-risk breast cancer screenings are not routinely carried out by the VA in primary care, women’s health, or oncology services. Furthermore, the recording of screening questionnaire results was not synchronized until a standard questionnaire was created and approved as a template by this group in the VA electronic medical record (EMR) in 2015.
Several prediction models can identify which women are at an increased risk of developing breast cancer. The most commonly used risk assessment model, the Gail breast cancer risk assessment tool (BCRAT), has been refined to include women of additional ethnicities (https://www.cancer.gov/bcrisktool).
This pilot project was launched to identify an effective manner to screen women veterans regarding their risk of developing breast cancer and refer them for chemoprevention education or genetic counseling as appropriate.
Methods
A high-risk breast cancer screening questionnaire based on the Gail BCRAT and including lifestyle questions was developed and included as a note template in the VA EMR. The James J. Peters VA Medical Center, Bronx, NY (JJPVAMC) and the Washington DC VA Medical Center (DCVAMC) ran a pilot study between 2015 and 2018 using this breast cancer screening questionnaire to collect data from women veterans. Quality Executive Committee and institutional review board approvals were granted respectively.
Eligibility criteria included women aged ≥ 35 years with no personal history of breast cancer. Most patients were self-referred, but participants also were recruited during VA Breast Cancer Awareness month events, health fairs, or at informational tables in the hospital lobbies. After completing the 20 multiple choice questionnaire with a study team member, either in person or over the phone, a 5-year and lifetime risk of invasive breast cancer was calculated using the Gail BCRAT. A woman is considered high risk and eligible for chemoprevention if her 5-year risk is > 1.66% or her lifetime risk is ≥ 20%. Eligibility for genetic counseling is based on the Breast Cancer Referral Screening Tool, which includes a personal or family history of breast or ovarian cancer and Jewish ancestry.
All patients were notified of their average or high risk status by a clinician. Those who were deemed to be average risk received a follow-up letter in the mail with instructions (eg, to follow-up with a yearly mammogram). Those who were deemed to be high risk for developing breast cancer were asked to come in for an appointment with the study principal investigator (a VA oncologist/breast cancer specialist) to discuss prevention options, further screening, or referrals to genetic counseling. Depending on a patient’s other health factors, a woman at high risk for developing breast cancer also may be a candidate for chemoprevention with tamoxifen, raloxifene, exemestane, anastrozole, or letrozole.
Data on the participant’s lifestyle, including exercise, diet, and smoking, were evaluated to determine whether these factors had an impact on risk status.
Results
The JJP and DC VAMCs screened 103 women veterans between 2015 and 2018. Four patients were excluded for nonveteran (spousal) status, leaving 99 women veterans with a mean age of 54 years. The most common self-reported races were Black (60%), non-Hispanic White (14%), and Hispanic or Latino (13%) (Table 1).
Women veterans in our study were nearly 3-times more likely than the general population were to receive a high-risk Gail Score/BCRAT (35% vs 13%, respectively).50,51 Of this subset, 46% had breast biopsies, and 86% had a positive family history. Thirty-one percent of Black women in our study were high risk, while nationally, 8.2 to 13.3% of Black women aged 50 to 59 years are considered high risk.50,51 Of the Black high-risk group with a high Gail/BCRAT score, 94% had a positive family history, and 33% had a history of breast biopsy (Table 2).
Of the 35 high-risk patients 26 (74%) patients accepted consultations for chemoprevention and 5 (19%) started chemoprevention. Of this high-risk group, 13 (37%) patients were referred for genetic counseling (Table 3).44 The prevalence of PTSD was present in 31% of high-risk women and 29% of the cohort (Figure).The lifestyle questions indicated that, among all participants, 79% had an overweight or obese body mass index; 58% exercised weekly; 51% consumed alcohol; 14% were smokers; and 21% consumed 3 to 4 servings of fruits/vegetables daily.
Discussion
Breast cancer is the most common cancer in women.52 The number of women with breast cancer in the VHA has more than tripled from 1995 to 2012.1 The lifetime risk of developing breast cancer in the general population is about 13%.50 This rate can be affected by risk factors including age, hormone exposure, family history, radiation exposure, and lifestyle factors, such as weight and alcohol use.6,52-56 In the United States, invasive breast cancer affects 1 in 8 women.50,52,57
Our screened population showed nearly 3 times as many women veterans were at an increased risk for breast cancer when compared with historical averages in US women. This difference may be based on a high rate of prior breast biopsies or positive family history, although a provocative study using the Surveillance, Epidemiology, and End Results database showed military women to have higher rates of breast cancer as well.9 Historically, Blacks are vastly understudied in clinical research with only 5% representation on a national level.5,58 The urban locations of both pilot sites (Washington, DC and Bronx, NY) allowed for the inclusion of minority patients in our study. We found that the rates of breast cancer in Black women veterans to be higher than seen nationally, possibly prompting further screening initiatives for this understudied population.
Our pilot study’s chemoprevention utilization (19%) was double the < 10% seen in the national population.33-35 The presence of a knowledgeable breast health practitioner to recruit study participants and offer personalized counseling to women veterans is a likely factor in overcoming barriers to chemopreventive acceptance. These participants may have been motivated to seek care for their high-risk status given a strong family history and prior breast biopsies.
Interestingly, a 3-fold higher PTSD rate was seen in this pilot population (29%) when compared with PTSD rates in the general female population (7-10%) and still one-third higher than the general population of women veterans (20%).45-47 Mental health, anxiety, and PTSD have been barriers to patients who sought treatment and have been implicated in poor adherence to medical advice.48,49 Cancer screening can induce anxiety in patients, and it may be amplified in patients with PTSD. It was remarkable that although adherence with screening recommendations is decreased when PTSD is present, our patient population demonstrated a higher rate of screening adherence.
Women who are seen at the VA often use multiple clinical specialties, and their EMR can be accessed across VA medical centers nationwide. Therefore, identifying women veterans who meet screening criteria is easily attainable within the VA.
When comparing high-risk with average risk women, the lifestyle results (BMI, smoking history, exercise and consumption of fruits, vegetables and alcohol) were essentially the same. Lifestyle factors were similar to national population rates and were unlikely to impact risk levels.
Limitations
Study limitations included a high number of self-referrals and the large percentage of patients with a family history of breast cancer, making them more likely to seek screening. The higher-than-average risk of breast cancer may be driven by a high rate of breast biopsies and a strong family history. Lifestyle metrics could not be accurately compared to other national assessments of lifestyle factors due to the difference in data points that we used or the format of our questions.
Conclusions
As the number of women veterans increases and the incidence of breast cancer in women veterans rise, chemoprevention options should follow national guidelines. To our knowledge, this is the only oncology study with 60% Black women veterans. This study had a higher participation rate for Black women veterans than is typically seen in national research studies and shows the VA to be a germane source for further understanding of an understudied population that may benefit from increased screening for breast cancer.
A team-based, multidisciplinary model that meets the unique healthcare needs of women veterans results in a patient-centric delivery of care for assessing breast cancer risk status and prevention options. This model can be replicated nationally by directing primary care physicians and women’s health practitioners to a risk-assessment questionnaire and referring high-risk women for appropriate preventative care. Given that these results show chemoprevention adherence rates doubled those seen nationally, perhaps techniques used within this VA pilot study may be adapted to decrease breast cancer incidence nationally.
Since the rate of PTSD among women veterans is triple the national average, we would expect adherence rates to be lower in our patient cohort. However, the multidisciplinary approach we used in this study (eg, 1:1 consultation with oncologist; genetic counseling referrals; mental health support available), may have improved adherence rates. Perhaps the high rates of PTSD seen in the VA patient population can be a useful way to explore patient adherence rates in those with mental illness and medical conditions.
Future research with a larger cohort may lead to greater insight into the correlation between PTSD and adherence to treatment. Exploring the connection between breast cancer, epigenetics, and specific military service-related exposures could be an area of analysis among this veteran population exhibiting increased breast cancer rates. VAMCs are situated in rural, suburban, and urban locations across the United States and offers a diverse socioeconomic and ethnic patient population for inclusion in clinical investigations. Women veterans make up a small subpopulation of women in the United States, but it is worth considering VA patients as an untapped resource for research collaboration.
Acknowledgements
The authors thank Steven Sanchez and Marissa Vallette, PhD, Breast Health Research Group. This research project was approved by the James J. Peters VA Medical Center Quality Executive Committee and the Washington, DC VA Medical Center Institutional Review Board. This work was supported by the US Department of Veterans Affairs. This work did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the US Government, or any of its agencies.
1. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. The past, present and future of women veterans. Published February 2017. Accessed April 28, 2021. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf.
2. Frayne SM, Carney DV, Bastian L, et al. The VA Women’s Health Practice-Based Research Network: amplifying women veterans’ voices in VA research. J Gen Intern Med. 2013;28 Suppl 2(Suppl 2):S504-S509. doi:10.1007/s11606-013-2476-3
3. US Department of Veterans Affairs, Veterans Health Administration, Women’s Health Evaluation Initiative, Women Veterans Health Strategic Health Care Group. Sourcebook: women veterans in the Veterans Health Administration. Volume 1: Sociodemographic characteristics and use of VHA care. Published December 2010. Accessed April 12, 2021. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2455
4. Bean-Mayberry B, Yano EM, Bayliss N, Navratil J, Weisman CS, Scholle SH. Federally funded comprehensive women’s health centers: leading innovation in women’s healthcare delivery. J Womens Health (Larchmt). 2007;16(9):1281-1290. doi:10.1089/jwh.2006.0284
5. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics.VA utilization profile FY 2016. Published November 2017. Accessed April 12, 2021. https://www.va.gov/vetdata/docs/QuickFacts/VA_Utilization_Profile.PDF
6. Ekenga CC, Parks CG, Sandler DP. Chemical exposures in the workplace and breast cancer risk: a prospective cohort study. Int J Cancer. 2015;137(7):1765-1774. doi:10.1002/ijc.29545
7. Rennix CP, Quinn MM, Amoroso PJ, Eisen EA, Wegman DH. Risk of breast cancer among enlisted Army women occupationally exposed to volatile organic compounds. Am J Ind Med. 2005;48(3):157-167. doi:10.1002/ajim.20201
8. Ritz B. Cancer mortality among workers exposed to chemicals during uranium processing. J Occup Environ Med. 1999;41(7):556-566. doi:10.1097/00043764-199907000-00004
9. Zhu K, Devesa SS, Wu H, et al. Cancer incidence in the U.S. military population: comparison with rates from the SEER program. Cancer Epidemiol Biomarkers Prev. 2009;18(6):1740-1745. doi:10.1158/1055-9965.EPI-09-0041
10. Freedman AN, Yu B, Gail MH, et al. Benefit/risk assessment for breast cancer chemoprevention with raloxifene or tamoxifen for women age 50 years or older [published correction appears in J Clin Oncol. 2013 Nov 10;31(32):4167]. J Clin Oncol. 2011;29(17):2327-2333. doi:10.1200/JCO.2010.33.0258
11. Greene, H. Cancer prevention, screening and early detection. In: Gobel BH, Triest-Robertson S, Vogel WH, eds. Advanced Oncology Nursing Certification Review and Resource Manual. 3rd ed. Oncology Nursing Society; 2016:1-34. https://www.ons.org/sites/default/files/publication_pdfs/2%20ADVPrac%20chapter%201.pdf
12. National Comprehensive Cancer Network. NCCN Breast Cancer Risk Reduction. Version 1.2021 NCCN Clinical Practice Guidelines in Oncology. Updated March 24, 2021 Accessed April 12, 2021. https://www.nccn.org/professionals/physician_gls/pdf/breast_risk.pdf
13. US Preventive Services Task Force. Breast cancer: Medications use to reduce risk. Updated September 3, 2019. Accessed April 12, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/breast-cancer-medications-for-risk-reduction
14. Moyer VA; U.S. Preventive Services Task Force. Medications to decrease the risk for breast cancer in women: recommendations from the U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;159(10):698-708. doi:10.7326/0003-4819-159-10-201311190-00717
15. Boucher JE. Chemoprevention: an overview of pharmacologic agents and nursing considerations. Clin J Oncol Nurs. 2018;22(3):350-353. doi:10.1188/18.CJON.350-353
16. Nichols HB, Stürmer T, Lee VS, et al. Breast cancer chemoprevention in an integrated health care setting. JCO Clin Cancer Inform. 2017;1:1-12. doi:10.1200/CCI.16.00059
17. Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16(11):1362-1389. doi:10.6004/jnccn.2018.0083
18. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline [published correction appears in J Clin Oncol. 2013 Dec 1;31(34):4383]. J Clin Oncol. 2013;31(23):2942-2962. doi:10.1200/JCO.2013.49.3122
19. Sealy-Jefferson S, Roseland ME, Cote ML, et al. rural-urban residence and stage at breast cancer diagnosis among postmenopausal women: The Women’s Health Initiative. J Womens Health (Larchmt). 2019;28(2):276-283. doi:10.1089/jwh.2017.6884
20. Holder KA. Veterans in rural America: 2011-2015. Published January 25, 2017. Accessed April 12, 2021. https://www.census.gov/library/publications/2017/acs/acs-36.html
21. Owens WL, Gallagher TJ, Kincheloe MJ, Ruetten VL. Implementation in a large health system of a program to identify women at high risk for breast cancer. J Oncol Pract. 2011;7(2):85-88. doi:10.1200/JOP.2010.000107
2. Pivot X, Viguier J, Touboul C, et al. Breast cancer screening controversy: too much or not enough?. Eur J Cancer Prev. 2015;24 Suppl:S73-S76. doi:10.1097/CEJ.0000000000000145
23. Bidassie B, Kovach A, Vallette MA, et al. Breast Cancer risk assessment and chemoprevention use among veterans affairs primary care providers: a national online survey. Mil Med. 2020;185(3-4):512-518. doi:10.1093/milmed/usz291
24. Brewster AM, Davidson NE, McCaskill-Stevens W. Chemoprevention for breast cancer: overcoming barriers to treatment. Am Soc Clin Oncol Educ Book. 2012;85-90. doi:10.14694/EdBook_AM.2012.32.152
25. Meyskens FL Jr, Curt GA, Brenner DE, et al. Regulatory approval of cancer risk-reducing (chemopreventive) drugs: moving what we have learned into the clinic. Cancer Prev Res (Phila). 2011;4(3):311-323. doi:10.1158/1940-6207.CAPR-09-0014
26. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36(3):533-558. doi:10.1016/j.pop.2009.04.003
27. Vogel VG. Selective estrogen receptor modulators and aromatase inhibitors for breast cancer chemoprevention. Curr Drug Targets. 2011;12(13):1874-1887. doi:10.2174/138945011798184164
28. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial [published correction appears in JAMA. 2006 Dec 27;296(24):2926] [published correction appears in JAMA. 2007 Sep 5;298(9):973]. JAMA. 2006;295(23):2727-2741. doi:10.1001/jama.295.23.joc60074
29. Pruthi S, Heisey RE, Bevers TB. Chemoprevention for breast cancer. Ann Surg Oncol. 2015;22(10):3230-3235. doi:10.1245/s10434-015-4715-9
30. Cuzick J, Sestak I, Forbes JF, et al. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial [published correction appears in Lancet. 2014 Mar 22;383(9922):1040] [published correction appears in Lancet. 2017 Mar 11;389(10073):1010]. Lancet. 2014;383(9922):1041-1048. doi:10.1016/S0140-6736(13)62292-8
31. Bozovic-Spasojevic I, Azambuja E, McCaskill-Stevens W, Dinh P, Cardoso F. Chemoprevention for breast cancer. Cancer Treat Rev. 2012;38(5):329-339. doi:10.1016/j.ctrv.2011.07.005
32. Gabriel EM, Jatoi I. Breast cancer chemoprevention. Expert Rev Anticancer Ther. 2012;12(2):223-228. doi:10.1586/era.11.206

33. Crew KD, Albain KS, Hershman DL, Unger JM, Lo SS. How do we increase uptake of tamoxifen and other anti-estrogens for breast cancer prevention?. NPJ Breast Cancer. 2017;3:20. Published 2017 May 19. doi:10.1038/s41523-017-0021-y
34. Ropka ME, Keim J, Philbrick JT. Patient decisions about breast cancer chemoprevention: a systematic review and meta-analysis. J Clin Oncol. 2010;28(18):3090-3095. doi:10.1200/JCO.2009.27.8077
35. Smith SG, Sestak I, Forster A, et al. Factors affecting uptake and adherence to breast cancer chemoprevention: a systematic review and meta-analysis. Ann Oncol. 2016;27(4):575-590. doi:10.1093/annonc/mdv590
36. Grann VR, Patel PR, Jacobson JS, et al. Comparative effectiveness of screening and prevention strategies among BRCA1/2-affected mutation carriers. Breast Cancer Res Treat. 2011 Feb;125(3):837-847. doi:10.1007/s10549-010-1043-4
37. Goss PE, Ingle JN, Alés-Martínez JE, et al. Exemestane for breast-cancer prevention in postmenopausal women [published correction appears in N Engl J Med. 2011 Oct 6;365(14):1361]. N Engl J Med. 2011;364(25):2381-2391. doi:10.1056/NEJMoa1103507
38. Kmietowicz Z. Five in six women reject drugs that could reduce their risk of breast cancer. BMJ. 2015;351:h6650. Published 2015 Dec 8. doi:10.1136/bmj.h6650
39. Nelson HD, Fu R, Griffin JC, Nygren P, Smith ME, Humphrey L. Systematic review: comparative effectiveness of medications to reduce risk for primary breast cancer. Ann Intern Med. 2009;151(10):703-235. doi:10.7326/0003-4819-151-10-200911170-00147
40. Dahabreh IJ, Wieland LS, Adam GP, Halladay C, Lau J, Trikalinos TA. Core needle and open surgery biopsy for diagnosis of breast lesions: an update to the 2009 report. Published September 2014. Accessed April 12, 2021. https://www.ncbi.nlm.nih.gov/books/NBK246878
41. National Cancer Institute. Genetics of breast and ovarian cancer (PDQ)—health profession version. Updated February 12, 2021. Accessed April 12, 2021. http://www.cancer.gov/cancertopics/pdq/genetics/breast-and-ovarian/HealthProfessional
42. US Department of Health and Human Services. National Institutes of Health, National Institute of Environmental Health Sciences The sister study. Accessed April 12, 2021. https://sisterstudy.niehs.nih.gov/english/NIEHS.htm
43. Tutt A, Ashworth A. Can genetic testing guide treatment in breast cancer?. Eur J Cancer. 2008;44(18):2774-2780. doi:10.1016/j.ejca.2008.10.009
44. Katz SJ, Ward KC, Hamilton AS, et al. Gaps in receipt of clinically indicated genetic counseling after diagnosis of breast cancer. J Clin Oncol. 2018;36(12):1218-1224. doi:10.1200/JCO.2017.76.2369
45. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in adults? Updated October 17, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_adults.asp
46. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in women? Updated October 16, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_women.asp
47. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in veterans? Updated September 24, 2018. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_veterans.asp
48. Lindberg NM, Wellisch D. Anxiety and compliance among women at high risk for breast cancer. Ann Behav Med. 2001;23(4):298-303. doi:10.1207/S15324796ABM2304_9
49. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101-2107. doi:10.1001/archinte.160.14.2101
50. Centers for Disease Control and Prevention. MMWR appendix: breast cancer rates among black women and white women. Updated October 13, 2016. Accessed April 12, 2021. https://www.cdc.gov/cancer/breast/statistics/trends_invasive.htm
51. Richardson LC, Henley SJ, Miller JW, Massetti G, Thomas CC. Patterns and trends in age-specific black-white differences in breast cancer incidence and mortality - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2016;65(40):1093-1098. Published 2016 Oct 14. doi:10.15585/mmwr.mm6540a1
52. Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA. Environmental pollutants and breast cancer: epidemiologic studies. Cancer. 2007;109(12 Suppl):2667-2711. doi:10.1002/cncr.22655
53. Brophy JT, Keith MM, Watterson A, et al. Breast cancer risk in relation to occupations with exposure to carcinogens and endocrine disruptors: a Canadian case-control study. Environ Health. 2012;11:87. Published 2012 Nov 19. doi:10.1186/1476-069X-11-87
54. Labrèche F, Goldberg MS, Valois MF, Nadon L. Postmenopausal breast cancer and occupational exposures. Occup Environ Med. 2010;67(4):263-269. doi:10.1136/oem.2009.049817
55. National Institute of Environmental Health Sciences, Interagency Breast Cancer & Environmental Research Coordinating Committee. Breast cancer and the environment: prioritizing prevention. Updated March 8, 2013. Accessed April 12, 2021. https://www.niehs.nih.gov/about/boards/ibcercc/index.cfm
56. Gail MH, Costantino JP, Pee D, et al. Projecting individualized absolute invasive breast cancer risk in African American women [published correction appears in J Natl Cancer Inst. 2008 Aug 6;100(15):1118] [published correction appears in J Natl Cancer Inst. 2008 Mar 5;100(5):373]. J Natl Cancer Inst. 2007;99(23):1782-1792. doi:10.1093/jnci/djm223
57. Corbie-Smith G, Thomas SB, Williams MV, Moody-Ayers S. Attitudes and beliefs of African Americans toward participation in medical research. J Gen Intern Med. 1999;14(9):537-546. doi:10.1046/j.1525-1497.1999.07048.x
58. Braunstein JB, Sherber NS, Schulman SP, Ding EL, Powe NR. Race, medical researcher distrust, perceived harm, and willingness to participate in cardiovascular prevention trials. Medicine (Baltimore). 2008;87(1):1-9. doi:10.1097/MD.0b013e3181625d78
The number of women seeking care from the Veterans Health Administration (VHA) is increasing.1 In 2015, there were 2 million women veterans in the United States, which is 9.4% of the total veteran population. This group is expected to increase at an average of about 18,000 women per year for the next 10 years.2 The percentage of women veterans who are US Department of Veterans Affairs (VA) users aged 45 to 64 years rose 46% from 2000 to 2015.1,3-4 It is estimated that 15% of veterans who used VA services in 2020 were women.1 Nineteen percent of women veterans are Black.1 The median age of women veterans in 2015 was 50 years.5 Breast cancer is the leading cancer affecting female veterans, and data suggest they have an increased risk of breast cancer based on unique service-related exposures.1,6-9
In the US, about 10 million women are eligible for breast cancer preventive therapy, including, but not limited to, medications, surgery, or lifestyle changes.10 Secondary prevention options include change in surveillance that can reduce their risk or identify cancer at an earlier stage when treatment is more effective. The United States Preventive Services Task Force, the National Comprehensive Cancer Network, the American Society for Clinical Oncology, the National Institute for Health and Care Excellence, and the Oncology Nursing Society recommend screening women aged ≥ 35 years to assess breast cancer risk.11-18 If a woman is at increased risk, she may be a candidate for chemoprevention, prozphylactic surgery, and possibly an enhanced screening regimen.
Urban and minority women are an understudied population. Most veterans (75%) live in urban or suburban settings.19,20 Urban veteran women constitute an important potential study population.
Chemoprevention measures have been underused because of factors involving both women and their health care providers. A large proportion of women are unaware of their higher risk status due to lack of adequate screening and risk assessment.21,22 In addition to patient lack of awareness of their high-risk status, primary care physicians are also reluctant to prescribe chemopreventive agents due to a lack of comfort or familiarity with the risks and benefits.23-26 The STAR2015, BCPT2005, IBIS2014, MAP3 2011, IBIS-I 2014, and IBIS II 2014 studies clearly demonstrate a 49 to 62% reduction in risk for women using chemoprevention such as selective estrogen receptor modulators or aromatase inhibitors, respectively.27-32 Yet only 4 to 9% of high-risk women not enrolled in a clinical trial are using chemoprevention.33-39
The possibility of developing breast cancer also may be increased because of a positive family history or being a member of a family in which there is a known susceptibility gene mutation.40 Based on these risk factors, women may be eligible for tailored follow-up and genetic counseling.41-44
Nationally, 7 to 10% of the civilian US population will experience posttraumatic stress disorder (PTSD).45 The rates are remarkably higher for women veterans, with roughly 20% diagnosed with PTSD.46,47 Anxiety and PTSD have been implicated in poor adherence to medical advice.48,49
In 2014, a national VA multidisciplinary group focused on breast cancer prevention, detection, treatment, and research to address breast health in the growing population of women veterans. High-risk breast cancer screenings are not routinely carried out by the VA in primary care, women’s health, or oncology services. Furthermore, the recording of screening questionnaire results was not synchronized until a standard questionnaire was created and approved as a template by this group in the VA electronic medical record (EMR) in 2015.
Several prediction models can identify which women are at an increased risk of developing breast cancer. The most commonly used risk assessment model, the Gail breast cancer risk assessment tool (BCRAT), has been refined to include women of additional ethnicities (https://www.cancer.gov/bcrisktool).
This pilot project was launched to identify an effective manner to screen women veterans regarding their risk of developing breast cancer and refer them for chemoprevention education or genetic counseling as appropriate.
Methods
A high-risk breast cancer screening questionnaire based on the Gail BCRAT and including lifestyle questions was developed and included as a note template in the VA EMR. The James J. Peters VA Medical Center, Bronx, NY (JJPVAMC) and the Washington DC VA Medical Center (DCVAMC) ran a pilot study between 2015 and 2018 using this breast cancer screening questionnaire to collect data from women veterans. Quality Executive Committee and institutional review board approvals were granted respectively.
Eligibility criteria included women aged ≥ 35 years with no personal history of breast cancer. Most patients were self-referred, but participants also were recruited during VA Breast Cancer Awareness month events, health fairs, or at informational tables in the hospital lobbies. After completing the 20 multiple choice questionnaire with a study team member, either in person or over the phone, a 5-year and lifetime risk of invasive breast cancer was calculated using the Gail BCRAT. A woman is considered high risk and eligible for chemoprevention if her 5-year risk is > 1.66% or her lifetime risk is ≥ 20%. Eligibility for genetic counseling is based on the Breast Cancer Referral Screening Tool, which includes a personal or family history of breast or ovarian cancer and Jewish ancestry.
All patients were notified of their average or high risk status by a clinician. Those who were deemed to be average risk received a follow-up letter in the mail with instructions (eg, to follow-up with a yearly mammogram). Those who were deemed to be high risk for developing breast cancer were asked to come in for an appointment with the study principal investigator (a VA oncologist/breast cancer specialist) to discuss prevention options, further screening, or referrals to genetic counseling. Depending on a patient’s other health factors, a woman at high risk for developing breast cancer also may be a candidate for chemoprevention with tamoxifen, raloxifene, exemestane, anastrozole, or letrozole.
Data on the participant’s lifestyle, including exercise, diet, and smoking, were evaluated to determine whether these factors had an impact on risk status.
Results
The JJP and DC VAMCs screened 103 women veterans between 2015 and 2018. Four patients were excluded for nonveteran (spousal) status, leaving 99 women veterans with a mean age of 54 years. The most common self-reported races were Black (60%), non-Hispanic White (14%), and Hispanic or Latino (13%) (Table 1).
Women veterans in our study were nearly 3-times more likely than the general population were to receive a high-risk Gail Score/BCRAT (35% vs 13%, respectively).50,51 Of this subset, 46% had breast biopsies, and 86% had a positive family history. Thirty-one percent of Black women in our study were high risk, while nationally, 8.2 to 13.3% of Black women aged 50 to 59 years are considered high risk.50,51 Of the Black high-risk group with a high Gail/BCRAT score, 94% had a positive family history, and 33% had a history of breast biopsy (Table 2).
Of the 35 high-risk patients 26 (74%) patients accepted consultations for chemoprevention and 5 (19%) started chemoprevention. Of this high-risk group, 13 (37%) patients were referred for genetic counseling (Table 3).44 The prevalence of PTSD was present in 31% of high-risk women and 29% of the cohort (Figure).The lifestyle questions indicated that, among all participants, 79% had an overweight or obese body mass index; 58% exercised weekly; 51% consumed alcohol; 14% were smokers; and 21% consumed 3 to 4 servings of fruits/vegetables daily.
Discussion
Breast cancer is the most common cancer in women.52 The number of women with breast cancer in the VHA has more than tripled from 1995 to 2012.1 The lifetime risk of developing breast cancer in the general population is about 13%.50 This rate can be affected by risk factors including age, hormone exposure, family history, radiation exposure, and lifestyle factors, such as weight and alcohol use.6,52-56 In the United States, invasive breast cancer affects 1 in 8 women.50,52,57
Our screened population showed nearly 3 times as many women veterans were at an increased risk for breast cancer when compared with historical averages in US women. This difference may be based on a high rate of prior breast biopsies or positive family history, although a provocative study using the Surveillance, Epidemiology, and End Results database showed military women to have higher rates of breast cancer as well.9 Historically, Blacks are vastly understudied in clinical research with only 5% representation on a national level.5,58 The urban locations of both pilot sites (Washington, DC and Bronx, NY) allowed for the inclusion of minority patients in our study. We found that the rates of breast cancer in Black women veterans to be higher than seen nationally, possibly prompting further screening initiatives for this understudied population.
Our pilot study’s chemoprevention utilization (19%) was double the < 10% seen in the national population.33-35 The presence of a knowledgeable breast health practitioner to recruit study participants and offer personalized counseling to women veterans is a likely factor in overcoming barriers to chemopreventive acceptance. These participants may have been motivated to seek care for their high-risk status given a strong family history and prior breast biopsies.
Interestingly, a 3-fold higher PTSD rate was seen in this pilot population (29%) when compared with PTSD rates in the general female population (7-10%) and still one-third higher than the general population of women veterans (20%).45-47 Mental health, anxiety, and PTSD have been barriers to patients who sought treatment and have been implicated in poor adherence to medical advice.48,49 Cancer screening can induce anxiety in patients, and it may be amplified in patients with PTSD. It was remarkable that although adherence with screening recommendations is decreased when PTSD is present, our patient population demonstrated a higher rate of screening adherence.
Women who are seen at the VA often use multiple clinical specialties, and their EMR can be accessed across VA medical centers nationwide. Therefore, identifying women veterans who meet screening criteria is easily attainable within the VA.
When comparing high-risk with average risk women, the lifestyle results (BMI, smoking history, exercise and consumption of fruits, vegetables and alcohol) were essentially the same. Lifestyle factors were similar to national population rates and were unlikely to impact risk levels.
Limitations
Study limitations included a high number of self-referrals and the large percentage of patients with a family history of breast cancer, making them more likely to seek screening. The higher-than-average risk of breast cancer may be driven by a high rate of breast biopsies and a strong family history. Lifestyle metrics could not be accurately compared to other national assessments of lifestyle factors due to the difference in data points that we used or the format of our questions.
Conclusions
As the number of women veterans increases and the incidence of breast cancer in women veterans rise, chemoprevention options should follow national guidelines. To our knowledge, this is the only oncology study with 60% Black women veterans. This study had a higher participation rate for Black women veterans than is typically seen in national research studies and shows the VA to be a germane source for further understanding of an understudied population that may benefit from increased screening for breast cancer.
A team-based, multidisciplinary model that meets the unique healthcare needs of women veterans results in a patient-centric delivery of care for assessing breast cancer risk status and prevention options. This model can be replicated nationally by directing primary care physicians and women’s health practitioners to a risk-assessment questionnaire and referring high-risk women for appropriate preventative care. Given that these results show chemoprevention adherence rates doubled those seen nationally, perhaps techniques used within this VA pilot study may be adapted to decrease breast cancer incidence nationally.
Since the rate of PTSD among women veterans is triple the national average, we would expect adherence rates to be lower in our patient cohort. However, the multidisciplinary approach we used in this study (eg, 1:1 consultation with oncologist; genetic counseling referrals; mental health support available), may have improved adherence rates. Perhaps the high rates of PTSD seen in the VA patient population can be a useful way to explore patient adherence rates in those with mental illness and medical conditions.
Future research with a larger cohort may lead to greater insight into the correlation between PTSD and adherence to treatment. Exploring the connection between breast cancer, epigenetics, and specific military service-related exposures could be an area of analysis among this veteran population exhibiting increased breast cancer rates. VAMCs are situated in rural, suburban, and urban locations across the United States and offers a diverse socioeconomic and ethnic patient population for inclusion in clinical investigations. Women veterans make up a small subpopulation of women in the United States, but it is worth considering VA patients as an untapped resource for research collaboration.
Acknowledgements
The authors thank Steven Sanchez and Marissa Vallette, PhD, Breast Health Research Group. This research project was approved by the James J. Peters VA Medical Center Quality Executive Committee and the Washington, DC VA Medical Center Institutional Review Board. This work was supported by the US Department of Veterans Affairs. This work did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the US Government, or any of its agencies.
The number of women seeking care from the Veterans Health Administration (VHA) is increasing.1 In 2015, there were 2 million women veterans in the United States, which is 9.4% of the total veteran population. This group is expected to increase at an average of about 18,000 women per year for the next 10 years.2 The percentage of women veterans who are US Department of Veterans Affairs (VA) users aged 45 to 64 years rose 46% from 2000 to 2015.1,3-4 It is estimated that 15% of veterans who used VA services in 2020 were women.1 Nineteen percent of women veterans are Black.1 The median age of women veterans in 2015 was 50 years.5 Breast cancer is the leading cancer affecting female veterans, and data suggest they have an increased risk of breast cancer based on unique service-related exposures.1,6-9
In the US, about 10 million women are eligible for breast cancer preventive therapy, including, but not limited to, medications, surgery, or lifestyle changes.10 Secondary prevention options include change in surveillance that can reduce their risk or identify cancer at an earlier stage when treatment is more effective. The United States Preventive Services Task Force, the National Comprehensive Cancer Network, the American Society for Clinical Oncology, the National Institute for Health and Care Excellence, and the Oncology Nursing Society recommend screening women aged ≥ 35 years to assess breast cancer risk.11-18 If a woman is at increased risk, she may be a candidate for chemoprevention, prozphylactic surgery, and possibly an enhanced screening regimen.
Urban and minority women are an understudied population. Most veterans (75%) live in urban or suburban settings.19,20 Urban veteran women constitute an important potential study population.
Chemoprevention measures have been underused because of factors involving both women and their health care providers. A large proportion of women are unaware of their higher risk status due to lack of adequate screening and risk assessment.21,22 In addition to patient lack of awareness of their high-risk status, primary care physicians are also reluctant to prescribe chemopreventive agents due to a lack of comfort or familiarity with the risks and benefits.23-26 The STAR2015, BCPT2005, IBIS2014, MAP3 2011, IBIS-I 2014, and IBIS II 2014 studies clearly demonstrate a 49 to 62% reduction in risk for women using chemoprevention such as selective estrogen receptor modulators or aromatase inhibitors, respectively.27-32 Yet only 4 to 9% of high-risk women not enrolled in a clinical trial are using chemoprevention.33-39
The possibility of developing breast cancer also may be increased because of a positive family history or being a member of a family in which there is a known susceptibility gene mutation.40 Based on these risk factors, women may be eligible for tailored follow-up and genetic counseling.41-44
Nationally, 7 to 10% of the civilian US population will experience posttraumatic stress disorder (PTSD).45 The rates are remarkably higher for women veterans, with roughly 20% diagnosed with PTSD.46,47 Anxiety and PTSD have been implicated in poor adherence to medical advice.48,49
In 2014, a national VA multidisciplinary group focused on breast cancer prevention, detection, treatment, and research to address breast health in the growing population of women veterans. High-risk breast cancer screenings are not routinely carried out by the VA in primary care, women’s health, or oncology services. Furthermore, the recording of screening questionnaire results was not synchronized until a standard questionnaire was created and approved as a template by this group in the VA electronic medical record (EMR) in 2015.
Several prediction models can identify which women are at an increased risk of developing breast cancer. The most commonly used risk assessment model, the Gail breast cancer risk assessment tool (BCRAT), has been refined to include women of additional ethnicities (https://www.cancer.gov/bcrisktool).
This pilot project was launched to identify an effective manner to screen women veterans regarding their risk of developing breast cancer and refer them for chemoprevention education or genetic counseling as appropriate.
Methods
A high-risk breast cancer screening questionnaire based on the Gail BCRAT and including lifestyle questions was developed and included as a note template in the VA EMR. The James J. Peters VA Medical Center, Bronx, NY (JJPVAMC) and the Washington DC VA Medical Center (DCVAMC) ran a pilot study between 2015 and 2018 using this breast cancer screening questionnaire to collect data from women veterans. Quality Executive Committee and institutional review board approvals were granted respectively.
Eligibility criteria included women aged ≥ 35 years with no personal history of breast cancer. Most patients were self-referred, but participants also were recruited during VA Breast Cancer Awareness month events, health fairs, or at informational tables in the hospital lobbies. After completing the 20 multiple choice questionnaire with a study team member, either in person or over the phone, a 5-year and lifetime risk of invasive breast cancer was calculated using the Gail BCRAT. A woman is considered high risk and eligible for chemoprevention if her 5-year risk is > 1.66% or her lifetime risk is ≥ 20%. Eligibility for genetic counseling is based on the Breast Cancer Referral Screening Tool, which includes a personal or family history of breast or ovarian cancer and Jewish ancestry.
All patients were notified of their average or high risk status by a clinician. Those who were deemed to be average risk received a follow-up letter in the mail with instructions (eg, to follow-up with a yearly mammogram). Those who were deemed to be high risk for developing breast cancer were asked to come in for an appointment with the study principal investigator (a VA oncologist/breast cancer specialist) to discuss prevention options, further screening, or referrals to genetic counseling. Depending on a patient’s other health factors, a woman at high risk for developing breast cancer also may be a candidate for chemoprevention with tamoxifen, raloxifene, exemestane, anastrozole, or letrozole.
Data on the participant’s lifestyle, including exercise, diet, and smoking, were evaluated to determine whether these factors had an impact on risk status.
Results
The JJP and DC VAMCs screened 103 women veterans between 2015 and 2018. Four patients were excluded for nonveteran (spousal) status, leaving 99 women veterans with a mean age of 54 years. The most common self-reported races were Black (60%), non-Hispanic White (14%), and Hispanic or Latino (13%) (Table 1).
Women veterans in our study were nearly 3-times more likely than the general population were to receive a high-risk Gail Score/BCRAT (35% vs 13%, respectively).50,51 Of this subset, 46% had breast biopsies, and 86% had a positive family history. Thirty-one percent of Black women in our study were high risk, while nationally, 8.2 to 13.3% of Black women aged 50 to 59 years are considered high risk.50,51 Of the Black high-risk group with a high Gail/BCRAT score, 94% had a positive family history, and 33% had a history of breast biopsy (Table 2).
Of the 35 high-risk patients 26 (74%) patients accepted consultations for chemoprevention and 5 (19%) started chemoprevention. Of this high-risk group, 13 (37%) patients were referred for genetic counseling (Table 3).44 The prevalence of PTSD was present in 31% of high-risk women and 29% of the cohort (Figure).The lifestyle questions indicated that, among all participants, 79% had an overweight or obese body mass index; 58% exercised weekly; 51% consumed alcohol; 14% were smokers; and 21% consumed 3 to 4 servings of fruits/vegetables daily.
Discussion
Breast cancer is the most common cancer in women.52 The number of women with breast cancer in the VHA has more than tripled from 1995 to 2012.1 The lifetime risk of developing breast cancer in the general population is about 13%.50 This rate can be affected by risk factors including age, hormone exposure, family history, radiation exposure, and lifestyle factors, such as weight and alcohol use.6,52-56 In the United States, invasive breast cancer affects 1 in 8 women.50,52,57
Our screened population showed nearly 3 times as many women veterans were at an increased risk for breast cancer when compared with historical averages in US women. This difference may be based on a high rate of prior breast biopsies or positive family history, although a provocative study using the Surveillance, Epidemiology, and End Results database showed military women to have higher rates of breast cancer as well.9 Historically, Blacks are vastly understudied in clinical research with only 5% representation on a national level.5,58 The urban locations of both pilot sites (Washington, DC and Bronx, NY) allowed for the inclusion of minority patients in our study. We found that the rates of breast cancer in Black women veterans to be higher than seen nationally, possibly prompting further screening initiatives for this understudied population.
Our pilot study’s chemoprevention utilization (19%) was double the < 10% seen in the national population.33-35 The presence of a knowledgeable breast health practitioner to recruit study participants and offer personalized counseling to women veterans is a likely factor in overcoming barriers to chemopreventive acceptance. These participants may have been motivated to seek care for their high-risk status given a strong family history and prior breast biopsies.
Interestingly, a 3-fold higher PTSD rate was seen in this pilot population (29%) when compared with PTSD rates in the general female population (7-10%) and still one-third higher than the general population of women veterans (20%).45-47 Mental health, anxiety, and PTSD have been barriers to patients who sought treatment and have been implicated in poor adherence to medical advice.48,49 Cancer screening can induce anxiety in patients, and it may be amplified in patients with PTSD. It was remarkable that although adherence with screening recommendations is decreased when PTSD is present, our patient population demonstrated a higher rate of screening adherence.
Women who are seen at the VA often use multiple clinical specialties, and their EMR can be accessed across VA medical centers nationwide. Therefore, identifying women veterans who meet screening criteria is easily attainable within the VA.
When comparing high-risk with average risk women, the lifestyle results (BMI, smoking history, exercise and consumption of fruits, vegetables and alcohol) were essentially the same. Lifestyle factors were similar to national population rates and were unlikely to impact risk levels.
Limitations
Study limitations included a high number of self-referrals and the large percentage of patients with a family history of breast cancer, making them more likely to seek screening. The higher-than-average risk of breast cancer may be driven by a high rate of breast biopsies and a strong family history. Lifestyle metrics could not be accurately compared to other national assessments of lifestyle factors due to the difference in data points that we used or the format of our questions.
Conclusions
As the number of women veterans increases and the incidence of breast cancer in women veterans rise, chemoprevention options should follow national guidelines. To our knowledge, this is the only oncology study with 60% Black women veterans. This study had a higher participation rate for Black women veterans than is typically seen in national research studies and shows the VA to be a germane source for further understanding of an understudied population that may benefit from increased screening for breast cancer.
A team-based, multidisciplinary model that meets the unique healthcare needs of women veterans results in a patient-centric delivery of care for assessing breast cancer risk status and prevention options. This model can be replicated nationally by directing primary care physicians and women’s health practitioners to a risk-assessment questionnaire and referring high-risk women for appropriate preventative care. Given that these results show chemoprevention adherence rates doubled those seen nationally, perhaps techniques used within this VA pilot study may be adapted to decrease breast cancer incidence nationally.
Since the rate of PTSD among women veterans is triple the national average, we would expect adherence rates to be lower in our patient cohort. However, the multidisciplinary approach we used in this study (eg, 1:1 consultation with oncologist; genetic counseling referrals; mental health support available), may have improved adherence rates. Perhaps the high rates of PTSD seen in the VA patient population can be a useful way to explore patient adherence rates in those with mental illness and medical conditions.
Future research with a larger cohort may lead to greater insight into the correlation between PTSD and adherence to treatment. Exploring the connection between breast cancer, epigenetics, and specific military service-related exposures could be an area of analysis among this veteran population exhibiting increased breast cancer rates. VAMCs are situated in rural, suburban, and urban locations across the United States and offers a diverse socioeconomic and ethnic patient population for inclusion in clinical investigations. Women veterans make up a small subpopulation of women in the United States, but it is worth considering VA patients as an untapped resource for research collaboration.
Acknowledgements
The authors thank Steven Sanchez and Marissa Vallette, PhD, Breast Health Research Group. This research project was approved by the James J. Peters VA Medical Center Quality Executive Committee and the Washington, DC VA Medical Center Institutional Review Board. This work was supported by the US Department of Veterans Affairs. This work did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the US Government, or any of its agencies.
1. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. The past, present and future of women veterans. Published February 2017. Accessed April 28, 2021. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf.
2. Frayne SM, Carney DV, Bastian L, et al. The VA Women’s Health Practice-Based Research Network: amplifying women veterans’ voices in VA research. J Gen Intern Med. 2013;28 Suppl 2(Suppl 2):S504-S509. doi:10.1007/s11606-013-2476-3
3. US Department of Veterans Affairs, Veterans Health Administration, Women’s Health Evaluation Initiative, Women Veterans Health Strategic Health Care Group. Sourcebook: women veterans in the Veterans Health Administration. Volume 1: Sociodemographic characteristics and use of VHA care. Published December 2010. Accessed April 12, 2021. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2455
4. Bean-Mayberry B, Yano EM, Bayliss N, Navratil J, Weisman CS, Scholle SH. Federally funded comprehensive women’s health centers: leading innovation in women’s healthcare delivery. J Womens Health (Larchmt). 2007;16(9):1281-1290. doi:10.1089/jwh.2006.0284
5. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics.VA utilization profile FY 2016. Published November 2017. Accessed April 12, 2021. https://www.va.gov/vetdata/docs/QuickFacts/VA_Utilization_Profile.PDF
6. Ekenga CC, Parks CG, Sandler DP. Chemical exposures in the workplace and breast cancer risk: a prospective cohort study. Int J Cancer. 2015;137(7):1765-1774. doi:10.1002/ijc.29545
7. Rennix CP, Quinn MM, Amoroso PJ, Eisen EA, Wegman DH. Risk of breast cancer among enlisted Army women occupationally exposed to volatile organic compounds. Am J Ind Med. 2005;48(3):157-167. doi:10.1002/ajim.20201
8. Ritz B. Cancer mortality among workers exposed to chemicals during uranium processing. J Occup Environ Med. 1999;41(7):556-566. doi:10.1097/00043764-199907000-00004
9. Zhu K, Devesa SS, Wu H, et al. Cancer incidence in the U.S. military population: comparison with rates from the SEER program. Cancer Epidemiol Biomarkers Prev. 2009;18(6):1740-1745. doi:10.1158/1055-9965.EPI-09-0041
10. Freedman AN, Yu B, Gail MH, et al. Benefit/risk assessment for breast cancer chemoprevention with raloxifene or tamoxifen for women age 50 years or older [published correction appears in J Clin Oncol. 2013 Nov 10;31(32):4167]. J Clin Oncol. 2011;29(17):2327-2333. doi:10.1200/JCO.2010.33.0258
11. Greene, H. Cancer prevention, screening and early detection. In: Gobel BH, Triest-Robertson S, Vogel WH, eds. Advanced Oncology Nursing Certification Review and Resource Manual. 3rd ed. Oncology Nursing Society; 2016:1-34. https://www.ons.org/sites/default/files/publication_pdfs/2%20ADVPrac%20chapter%201.pdf
12. National Comprehensive Cancer Network. NCCN Breast Cancer Risk Reduction. Version 1.2021 NCCN Clinical Practice Guidelines in Oncology. Updated March 24, 2021 Accessed April 12, 2021. https://www.nccn.org/professionals/physician_gls/pdf/breast_risk.pdf
13. US Preventive Services Task Force. Breast cancer: Medications use to reduce risk. Updated September 3, 2019. Accessed April 12, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/breast-cancer-medications-for-risk-reduction
14. Moyer VA; U.S. Preventive Services Task Force. Medications to decrease the risk for breast cancer in women: recommendations from the U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;159(10):698-708. doi:10.7326/0003-4819-159-10-201311190-00717
15. Boucher JE. Chemoprevention: an overview of pharmacologic agents and nursing considerations. Clin J Oncol Nurs. 2018;22(3):350-353. doi:10.1188/18.CJON.350-353
16. Nichols HB, Stürmer T, Lee VS, et al. Breast cancer chemoprevention in an integrated health care setting. JCO Clin Cancer Inform. 2017;1:1-12. doi:10.1200/CCI.16.00059
17. Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16(11):1362-1389. doi:10.6004/jnccn.2018.0083
18. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline [published correction appears in J Clin Oncol. 2013 Dec 1;31(34):4383]. J Clin Oncol. 2013;31(23):2942-2962. doi:10.1200/JCO.2013.49.3122
19. Sealy-Jefferson S, Roseland ME, Cote ML, et al. rural-urban residence and stage at breast cancer diagnosis among postmenopausal women: The Women’s Health Initiative. J Womens Health (Larchmt). 2019;28(2):276-283. doi:10.1089/jwh.2017.6884
20. Holder KA. Veterans in rural America: 2011-2015. Published January 25, 2017. Accessed April 12, 2021. https://www.census.gov/library/publications/2017/acs/acs-36.html
21. Owens WL, Gallagher TJ, Kincheloe MJ, Ruetten VL. Implementation in a large health system of a program to identify women at high risk for breast cancer. J Oncol Pract. 2011;7(2):85-88. doi:10.1200/JOP.2010.000107
2. Pivot X, Viguier J, Touboul C, et al. Breast cancer screening controversy: too much or not enough?. Eur J Cancer Prev. 2015;24 Suppl:S73-S76. doi:10.1097/CEJ.0000000000000145
23. Bidassie B, Kovach A, Vallette MA, et al. Breast Cancer risk assessment and chemoprevention use among veterans affairs primary care providers: a national online survey. Mil Med. 2020;185(3-4):512-518. doi:10.1093/milmed/usz291
24. Brewster AM, Davidson NE, McCaskill-Stevens W. Chemoprevention for breast cancer: overcoming barriers to treatment. Am Soc Clin Oncol Educ Book. 2012;85-90. doi:10.14694/EdBook_AM.2012.32.152
25. Meyskens FL Jr, Curt GA, Brenner DE, et al. Regulatory approval of cancer risk-reducing (chemopreventive) drugs: moving what we have learned into the clinic. Cancer Prev Res (Phila). 2011;4(3):311-323. doi:10.1158/1940-6207.CAPR-09-0014
26. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36(3):533-558. doi:10.1016/j.pop.2009.04.003
27. Vogel VG. Selective estrogen receptor modulators and aromatase inhibitors for breast cancer chemoprevention. Curr Drug Targets. 2011;12(13):1874-1887. doi:10.2174/138945011798184164
28. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial [published correction appears in JAMA. 2006 Dec 27;296(24):2926] [published correction appears in JAMA. 2007 Sep 5;298(9):973]. JAMA. 2006;295(23):2727-2741. doi:10.1001/jama.295.23.joc60074
29. Pruthi S, Heisey RE, Bevers TB. Chemoprevention for breast cancer. Ann Surg Oncol. 2015;22(10):3230-3235. doi:10.1245/s10434-015-4715-9
30. Cuzick J, Sestak I, Forbes JF, et al. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial [published correction appears in Lancet. 2014 Mar 22;383(9922):1040] [published correction appears in Lancet. 2017 Mar 11;389(10073):1010]. Lancet. 2014;383(9922):1041-1048. doi:10.1016/S0140-6736(13)62292-8
31. Bozovic-Spasojevic I, Azambuja E, McCaskill-Stevens W, Dinh P, Cardoso F. Chemoprevention for breast cancer. Cancer Treat Rev. 2012;38(5):329-339. doi:10.1016/j.ctrv.2011.07.005
32. Gabriel EM, Jatoi I. Breast cancer chemoprevention. Expert Rev Anticancer Ther. 2012;12(2):223-228. doi:10.1586/era.11.206

33. Crew KD, Albain KS, Hershman DL, Unger JM, Lo SS. How do we increase uptake of tamoxifen and other anti-estrogens for breast cancer prevention?. NPJ Breast Cancer. 2017;3:20. Published 2017 May 19. doi:10.1038/s41523-017-0021-y
34. Ropka ME, Keim J, Philbrick JT. Patient decisions about breast cancer chemoprevention: a systematic review and meta-analysis. J Clin Oncol. 2010;28(18):3090-3095. doi:10.1200/JCO.2009.27.8077
35. Smith SG, Sestak I, Forster A, et al. Factors affecting uptake and adherence to breast cancer chemoprevention: a systematic review and meta-analysis. Ann Oncol. 2016;27(4):575-590. doi:10.1093/annonc/mdv590
36. Grann VR, Patel PR, Jacobson JS, et al. Comparative effectiveness of screening and prevention strategies among BRCA1/2-affected mutation carriers. Breast Cancer Res Treat. 2011 Feb;125(3):837-847. doi:10.1007/s10549-010-1043-4
37. Goss PE, Ingle JN, Alés-Martínez JE, et al. Exemestane for breast-cancer prevention in postmenopausal women [published correction appears in N Engl J Med. 2011 Oct 6;365(14):1361]. N Engl J Med. 2011;364(25):2381-2391. doi:10.1056/NEJMoa1103507
38. Kmietowicz Z. Five in six women reject drugs that could reduce their risk of breast cancer. BMJ. 2015;351:h6650. Published 2015 Dec 8. doi:10.1136/bmj.h6650
39. Nelson HD, Fu R, Griffin JC, Nygren P, Smith ME, Humphrey L. Systematic review: comparative effectiveness of medications to reduce risk for primary breast cancer. Ann Intern Med. 2009;151(10):703-235. doi:10.7326/0003-4819-151-10-200911170-00147
40. Dahabreh IJ, Wieland LS, Adam GP, Halladay C, Lau J, Trikalinos TA. Core needle and open surgery biopsy for diagnosis of breast lesions: an update to the 2009 report. Published September 2014. Accessed April 12, 2021. https://www.ncbi.nlm.nih.gov/books/NBK246878
41. National Cancer Institute. Genetics of breast and ovarian cancer (PDQ)—health profession version. Updated February 12, 2021. Accessed April 12, 2021. http://www.cancer.gov/cancertopics/pdq/genetics/breast-and-ovarian/HealthProfessional
42. US Department of Health and Human Services. National Institutes of Health, National Institute of Environmental Health Sciences The sister study. Accessed April 12, 2021. https://sisterstudy.niehs.nih.gov/english/NIEHS.htm
43. Tutt A, Ashworth A. Can genetic testing guide treatment in breast cancer?. Eur J Cancer. 2008;44(18):2774-2780. doi:10.1016/j.ejca.2008.10.009
44. Katz SJ, Ward KC, Hamilton AS, et al. Gaps in receipt of clinically indicated genetic counseling after diagnosis of breast cancer. J Clin Oncol. 2018;36(12):1218-1224. doi:10.1200/JCO.2017.76.2369
45. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in adults? Updated October 17, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_adults.asp
46. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in women? Updated October 16, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_women.asp
47. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in veterans? Updated September 24, 2018. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_veterans.asp
48. Lindberg NM, Wellisch D. Anxiety and compliance among women at high risk for breast cancer. Ann Behav Med. 2001;23(4):298-303. doi:10.1207/S15324796ABM2304_9
49. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101-2107. doi:10.1001/archinte.160.14.2101
50. Centers for Disease Control and Prevention. MMWR appendix: breast cancer rates among black women and white women. Updated October 13, 2016. Accessed April 12, 2021. https://www.cdc.gov/cancer/breast/statistics/trends_invasive.htm
51. Richardson LC, Henley SJ, Miller JW, Massetti G, Thomas CC. Patterns and trends in age-specific black-white differences in breast cancer incidence and mortality - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2016;65(40):1093-1098. Published 2016 Oct 14. doi:10.15585/mmwr.mm6540a1
52. Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA. Environmental pollutants and breast cancer: epidemiologic studies. Cancer. 2007;109(12 Suppl):2667-2711. doi:10.1002/cncr.22655
53. Brophy JT, Keith MM, Watterson A, et al. Breast cancer risk in relation to occupations with exposure to carcinogens and endocrine disruptors: a Canadian case-control study. Environ Health. 2012;11:87. Published 2012 Nov 19. doi:10.1186/1476-069X-11-87
54. Labrèche F, Goldberg MS, Valois MF, Nadon L. Postmenopausal breast cancer and occupational exposures. Occup Environ Med. 2010;67(4):263-269. doi:10.1136/oem.2009.049817
55. National Institute of Environmental Health Sciences, Interagency Breast Cancer & Environmental Research Coordinating Committee. Breast cancer and the environment: prioritizing prevention. Updated March 8, 2013. Accessed April 12, 2021. https://www.niehs.nih.gov/about/boards/ibcercc/index.cfm
56. Gail MH, Costantino JP, Pee D, et al. Projecting individualized absolute invasive breast cancer risk in African American women [published correction appears in J Natl Cancer Inst. 2008 Aug 6;100(15):1118] [published correction appears in J Natl Cancer Inst. 2008 Mar 5;100(5):373]. J Natl Cancer Inst. 2007;99(23):1782-1792. doi:10.1093/jnci/djm223
57. Corbie-Smith G, Thomas SB, Williams MV, Moody-Ayers S. Attitudes and beliefs of African Americans toward participation in medical research. J Gen Intern Med. 1999;14(9):537-546. doi:10.1046/j.1525-1497.1999.07048.x
58. Braunstein JB, Sherber NS, Schulman SP, Ding EL, Powe NR. Race, medical researcher distrust, perceived harm, and willingness to participate in cardiovascular prevention trials. Medicine (Baltimore). 2008;87(1):1-9. doi:10.1097/MD.0b013e3181625d78
1. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics. The past, present and future of women veterans. Published February 2017. Accessed April 28, 2021. https://www.va.gov/vetdata/docs/specialreports/women_veterans_2015_final.pdf.
2. Frayne SM, Carney DV, Bastian L, et al. The VA Women’s Health Practice-Based Research Network: amplifying women veterans’ voices in VA research. J Gen Intern Med. 2013;28 Suppl 2(Suppl 2):S504-S509. doi:10.1007/s11606-013-2476-3
3. US Department of Veterans Affairs, Veterans Health Administration, Women’s Health Evaluation Initiative, Women Veterans Health Strategic Health Care Group. Sourcebook: women veterans in the Veterans Health Administration. Volume 1: Sociodemographic characteristics and use of VHA care. Published December 2010. Accessed April 12, 2021. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=2455
4. Bean-Mayberry B, Yano EM, Bayliss N, Navratil J, Weisman CS, Scholle SH. Federally funded comprehensive women’s health centers: leading innovation in women’s healthcare delivery. J Womens Health (Larchmt). 2007;16(9):1281-1290. doi:10.1089/jwh.2006.0284
5. US Department of Veterans Affairs. National Center for Veterans Analysis and Statistics.VA utilization profile FY 2016. Published November 2017. Accessed April 12, 2021. https://www.va.gov/vetdata/docs/QuickFacts/VA_Utilization_Profile.PDF
6. Ekenga CC, Parks CG, Sandler DP. Chemical exposures in the workplace and breast cancer risk: a prospective cohort study. Int J Cancer. 2015;137(7):1765-1774. doi:10.1002/ijc.29545
7. Rennix CP, Quinn MM, Amoroso PJ, Eisen EA, Wegman DH. Risk of breast cancer among enlisted Army women occupationally exposed to volatile organic compounds. Am J Ind Med. 2005;48(3):157-167. doi:10.1002/ajim.20201
8. Ritz B. Cancer mortality among workers exposed to chemicals during uranium processing. J Occup Environ Med. 1999;41(7):556-566. doi:10.1097/00043764-199907000-00004
9. Zhu K, Devesa SS, Wu H, et al. Cancer incidence in the U.S. military population: comparison with rates from the SEER program. Cancer Epidemiol Biomarkers Prev. 2009;18(6):1740-1745. doi:10.1158/1055-9965.EPI-09-0041
10. Freedman AN, Yu B, Gail MH, et al. Benefit/risk assessment for breast cancer chemoprevention with raloxifene or tamoxifen for women age 50 years or older [published correction appears in J Clin Oncol. 2013 Nov 10;31(32):4167]. J Clin Oncol. 2011;29(17):2327-2333. doi:10.1200/JCO.2010.33.0258
11. Greene, H. Cancer prevention, screening and early detection. In: Gobel BH, Triest-Robertson S, Vogel WH, eds. Advanced Oncology Nursing Certification Review and Resource Manual. 3rd ed. Oncology Nursing Society; 2016:1-34. https://www.ons.org/sites/default/files/publication_pdfs/2%20ADVPrac%20chapter%201.pdf
12. National Comprehensive Cancer Network. NCCN Breast Cancer Risk Reduction. Version 1.2021 NCCN Clinical Practice Guidelines in Oncology. Updated March 24, 2021 Accessed April 12, 2021. https://www.nccn.org/professionals/physician_gls/pdf/breast_risk.pdf
13. US Preventive Services Task Force. Breast cancer: Medications use to reduce risk. Updated September 3, 2019. Accessed April 12, 2021. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/breast-cancer-medications-for-risk-reduction
14. Moyer VA; U.S. Preventive Services Task Force. Medications to decrease the risk for breast cancer in women: recommendations from the U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;159(10):698-708. doi:10.7326/0003-4819-159-10-201311190-00717
15. Boucher JE. Chemoprevention: an overview of pharmacologic agents and nursing considerations. Clin J Oncol Nurs. 2018;22(3):350-353. doi:10.1188/18.CJON.350-353
16. Nichols HB, Stürmer T, Lee VS, et al. Breast cancer chemoprevention in an integrated health care setting. JCO Clin Cancer Inform. 2017;1:1-12. doi:10.1200/CCI.16.00059
17. Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16(11):1362-1389. doi:10.6004/jnccn.2018.0083
18. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline [published correction appears in J Clin Oncol. 2013 Dec 1;31(34):4383]. J Clin Oncol. 2013;31(23):2942-2962. doi:10.1200/JCO.2013.49.3122
19. Sealy-Jefferson S, Roseland ME, Cote ML, et al. rural-urban residence and stage at breast cancer diagnosis among postmenopausal women: The Women’s Health Initiative. J Womens Health (Larchmt). 2019;28(2):276-283. doi:10.1089/jwh.2017.6884
20. Holder KA. Veterans in rural America: 2011-2015. Published January 25, 2017. Accessed April 12, 2021. https://www.census.gov/library/publications/2017/acs/acs-36.html
21. Owens WL, Gallagher TJ, Kincheloe MJ, Ruetten VL. Implementation in a large health system of a program to identify women at high risk for breast cancer. J Oncol Pract. 2011;7(2):85-88. doi:10.1200/JOP.2010.000107
2. Pivot X, Viguier J, Touboul C, et al. Breast cancer screening controversy: too much or not enough?. Eur J Cancer Prev. 2015;24 Suppl:S73-S76. doi:10.1097/CEJ.0000000000000145
23. Bidassie B, Kovach A, Vallette MA, et al. Breast Cancer risk assessment and chemoprevention use among veterans affairs primary care providers: a national online survey. Mil Med. 2020;185(3-4):512-518. doi:10.1093/milmed/usz291
24. Brewster AM, Davidson NE, McCaskill-Stevens W. Chemoprevention for breast cancer: overcoming barriers to treatment. Am Soc Clin Oncol Educ Book. 2012;85-90. doi:10.14694/EdBook_AM.2012.32.152
25. Meyskens FL Jr, Curt GA, Brenner DE, et al. Regulatory approval of cancer risk-reducing (chemopreventive) drugs: moving what we have learned into the clinic. Cancer Prev Res (Phila). 2011;4(3):311-323. doi:10.1158/1940-6207.CAPR-09-0014
26. Tice JA, Kerlikowske K. Screening and prevention of breast cancer in primary care. Prim Care. 2009;36(3):533-558. doi:10.1016/j.pop.2009.04.003
27. Vogel VG. Selective estrogen receptor modulators and aromatase inhibitors for breast cancer chemoprevention. Curr Drug Targets. 2011;12(13):1874-1887. doi:10.2174/138945011798184164
28. Vogel VG, Costantino JP, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial [published correction appears in JAMA. 2006 Dec 27;296(24):2926] [published correction appears in JAMA. 2007 Sep 5;298(9):973]. JAMA. 2006;295(23):2727-2741. doi:10.1001/jama.295.23.joc60074
29. Pruthi S, Heisey RE, Bevers TB. Chemoprevention for breast cancer. Ann Surg Oncol. 2015;22(10):3230-3235. doi:10.1245/s10434-015-4715-9
30. Cuzick J, Sestak I, Forbes JF, et al. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial [published correction appears in Lancet. 2014 Mar 22;383(9922):1040] [published correction appears in Lancet. 2017 Mar 11;389(10073):1010]. Lancet. 2014;383(9922):1041-1048. doi:10.1016/S0140-6736(13)62292-8
31. Bozovic-Spasojevic I, Azambuja E, McCaskill-Stevens W, Dinh P, Cardoso F. Chemoprevention for breast cancer. Cancer Treat Rev. 2012;38(5):329-339. doi:10.1016/j.ctrv.2011.07.005
32. Gabriel EM, Jatoi I. Breast cancer chemoprevention. Expert Rev Anticancer Ther. 2012;12(2):223-228. doi:10.1586/era.11.206

33. Crew KD, Albain KS, Hershman DL, Unger JM, Lo SS. How do we increase uptake of tamoxifen and other anti-estrogens for breast cancer prevention?. NPJ Breast Cancer. 2017;3:20. Published 2017 May 19. doi:10.1038/s41523-017-0021-y
34. Ropka ME, Keim J, Philbrick JT. Patient decisions about breast cancer chemoprevention: a systematic review and meta-analysis. J Clin Oncol. 2010;28(18):3090-3095. doi:10.1200/JCO.2009.27.8077
35. Smith SG, Sestak I, Forster A, et al. Factors affecting uptake and adherence to breast cancer chemoprevention: a systematic review and meta-analysis. Ann Oncol. 2016;27(4):575-590. doi:10.1093/annonc/mdv590
36. Grann VR, Patel PR, Jacobson JS, et al. Comparative effectiveness of screening and prevention strategies among BRCA1/2-affected mutation carriers. Breast Cancer Res Treat. 2011 Feb;125(3):837-847. doi:10.1007/s10549-010-1043-4
37. Goss PE, Ingle JN, Alés-Martínez JE, et al. Exemestane for breast-cancer prevention in postmenopausal women [published correction appears in N Engl J Med. 2011 Oct 6;365(14):1361]. N Engl J Med. 2011;364(25):2381-2391. doi:10.1056/NEJMoa1103507
38. Kmietowicz Z. Five in six women reject drugs that could reduce their risk of breast cancer. BMJ. 2015;351:h6650. Published 2015 Dec 8. doi:10.1136/bmj.h6650
39. Nelson HD, Fu R, Griffin JC, Nygren P, Smith ME, Humphrey L. Systematic review: comparative effectiveness of medications to reduce risk for primary breast cancer. Ann Intern Med. 2009;151(10):703-235. doi:10.7326/0003-4819-151-10-200911170-00147
40. Dahabreh IJ, Wieland LS, Adam GP, Halladay C, Lau J, Trikalinos TA. Core needle and open surgery biopsy for diagnosis of breast lesions: an update to the 2009 report. Published September 2014. Accessed April 12, 2021. https://www.ncbi.nlm.nih.gov/books/NBK246878
41. National Cancer Institute. Genetics of breast and ovarian cancer (PDQ)—health profession version. Updated February 12, 2021. Accessed April 12, 2021. http://www.cancer.gov/cancertopics/pdq/genetics/breast-and-ovarian/HealthProfessional
42. US Department of Health and Human Services. National Institutes of Health, National Institute of Environmental Health Sciences The sister study. Accessed April 12, 2021. https://sisterstudy.niehs.nih.gov/english/NIEHS.htm
43. Tutt A, Ashworth A. Can genetic testing guide treatment in breast cancer?. Eur J Cancer. 2008;44(18):2774-2780. doi:10.1016/j.ejca.2008.10.009
44. Katz SJ, Ward KC, Hamilton AS, et al. Gaps in receipt of clinically indicated genetic counseling after diagnosis of breast cancer. J Clin Oncol. 2018;36(12):1218-1224. doi:10.1200/JCO.2017.76.2369
45. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in adults? Updated October 17, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_adults.asp
46. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in women? Updated October 16, 2019. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_women.asp
47. US Department of Veterans Affairs. PTSD: National Center for PTSD. How common is PTSD in veterans? Updated September 24, 2018. Accessed April 12, 2021. https://www.ptsd.va.gov/understand/common/common_veterans.asp
48. Lindberg NM, Wellisch D. Anxiety and compliance among women at high risk for breast cancer. Ann Behav Med. 2001;23(4):298-303. doi:10.1207/S15324796ABM2304_9
49. DiMatteo MR, Lepper HS, Croghan TW. Depression is a risk factor for noncompliance with medical treatment: meta-analysis of the effects of anxiety and depression on patient adherence. Arch Intern Med. 2000;160(14):2101-2107. doi:10.1001/archinte.160.14.2101
50. Centers for Disease Control and Prevention. MMWR appendix: breast cancer rates among black women and white women. Updated October 13, 2016. Accessed April 12, 2021. https://www.cdc.gov/cancer/breast/statistics/trends_invasive.htm
51. Richardson LC, Henley SJ, Miller JW, Massetti G, Thomas CC. Patterns and trends in age-specific black-white differences in breast cancer incidence and mortality - United States, 1999-2014. MMWR Morb Mortal Wkly Rep. 2016;65(40):1093-1098. Published 2016 Oct 14. doi:10.15585/mmwr.mm6540a1
52. Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA. Environmental pollutants and breast cancer: epidemiologic studies. Cancer. 2007;109(12 Suppl):2667-2711. doi:10.1002/cncr.22655
53. Brophy JT, Keith MM, Watterson A, et al. Breast cancer risk in relation to occupations with exposure to carcinogens and endocrine disruptors: a Canadian case-control study. Environ Health. 2012;11:87. Published 2012 Nov 19. doi:10.1186/1476-069X-11-87
54. Labrèche F, Goldberg MS, Valois MF, Nadon L. Postmenopausal breast cancer and occupational exposures. Occup Environ Med. 2010;67(4):263-269. doi:10.1136/oem.2009.049817
55. National Institute of Environmental Health Sciences, Interagency Breast Cancer & Environmental Research Coordinating Committee. Breast cancer and the environment: prioritizing prevention. Updated March 8, 2013. Accessed April 12, 2021. https://www.niehs.nih.gov/about/boards/ibcercc/index.cfm
56. Gail MH, Costantino JP, Pee D, et al. Projecting individualized absolute invasive breast cancer risk in African American women [published correction appears in J Natl Cancer Inst. 2008 Aug 6;100(15):1118] [published correction appears in J Natl Cancer Inst. 2008 Mar 5;100(5):373]. J Natl Cancer Inst. 2007;99(23):1782-1792. doi:10.1093/jnci/djm223
57. Corbie-Smith G, Thomas SB, Williams MV, Moody-Ayers S. Attitudes and beliefs of African Americans toward participation in medical research. J Gen Intern Med. 1999;14(9):537-546. doi:10.1046/j.1525-1497.1999.07048.x
58. Braunstein JB, Sherber NS, Schulman SP, Ding EL, Powe NR. Race, medical researcher distrust, perceived harm, and willingness to participate in cardiovascular prevention trials. Medicine (Baltimore). 2008;87(1):1-9. doi:10.1097/MD.0b013e3181625d78