User login
Using standardized patients to assess hospitalist communication skills
Hospitalists must create rapport and communicate large amounts of information in a short amount of time without having a prior relationship with the patient.1 High-quality communication can improve satisfaction and compliance, while poor communication leaves patients ill prepared to transition back to the community.2–10
Many medical schools use standardized patients (SPs) to both train and evaluate their students’ communication skills. To our knowledge, no published studies describe using SPs to assess or teach communication skills for hospitalists.
Our objective in this study was to use SPs to assess for deficits in our hospitalists’ communication skills and to determine whether feedback provided by SPs could improve hospitalist confidence in and performance of optimal communication behaviors.
METHODS
Setting and Participants
Scenario and Checklist Development
We developed 3 SP encounters around common hospitalist-patient interactions: daily rounding, discharge, and interacting with a difficult patient. In order to assess communication skills, we developed a checklist with 3 core domains: Courtesy and Respect, Listen, and Explain. Each domain corresponded to 1 of 3 questions on the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey that pertained to doctor’s communications skills: (1) How often did doctors treat you with courtesy and respect? (2) How often did doctors listen carefully to you? (3) How often did doctors explain things in a way you could understand? We then developed checklist items that corresponded to essential communication skills within each of the 3 domains. These communication skills were based on best practices and published literature.
Discharge Encounter (Table 2): Patient admitted the night before with abdominal pain by another hospitalist. The checklist was based on AIDET®, an effective communication skills training protocol that our hospitalist group had been trained on.11
Daily Rounding Encounter (Table 3): Patient being discharged after an admission for congestive heart failure. The checklist was developed from the Society of Hospital Medicine discharge toolkit.12
Difficult Patient Encounter (Table 4): A patient and his daughter who were unhappy because of a previously missed lung mass that was now found to be cancer. Our checklist was based on characteristics of therapeutic bedside manner.13
The checklist items were each scored using a 3-point scale of adequate, partial, or inadequate performance. A description of checklist items within each of the 3 domains is listed in Table 1. A postintervention survey was filled out by all hospitalists after the 3 encounters.
Simulated Encounters
All 3 encounters occurred on the same day and each one lasted 1 hour (20 minutes for the encounter, 10 minutes for a posttest survey, and 30 minutes of feedback from the SP). For each case, a task list was given to the hospitalist before walking into the room (Appendix 1). During the feedback session, the SP gave the hospitalist feedback using the case checklist items. They then watched a video of the encounter and the SP further emphasized areas for improvement.
SP Training
SP training consisted of three 3-hour training sessions, which included review of the case, script, guidance on scoring the checklist items, role plays with attending hospitalists, and feedback training. Each SP was assigned to only 1 case.
Seven of the 24 encounters for each SP were reviewed independently by 2 investigators who created a final score for each checklist item which was compared to the SP’s checklist item score. The kappa (k) statistic was used to evaluate inter-observer reliability using the SAS system software (SAS Institute Inc.).
Analysis
The percent of hospitalists who performed each checklist item adequately within in each of the 3 domains (Courtesy and Respect, Listen, and Explain) was calculated. To compare the 3 domains, t tests were used.
We calculated the percent that our hospitalist group received on the 3 HCAHPS doctor’s questions 1 year prior to our SP exercise and 1 year after the SP exercise.
RESULTS
Twenty-three hospitalists completed all 3 encounters. For the 3 domains (Courtesy and Respect, Listen, and Explain), hospitalists performed significantly better in the Listen domain compared to the other 2 domains, with a mean percent adequate score of 90.2 % (95% confidence interval [CI], 72.2%-100%; P < 0.05), and significantly worse in the Explain domain compared to the other 2 domains, with a mean percent adequate score of 65.0% (95% CI, 49.2%-83.6%; P < 0.05). The mean percent adequate score for the Courtesy and Respect domain was 81.6% (95% CI, 56%-100%). This was significantly higher than the Explain domain and significantly lower than the Listen domain.
Posttest survey results showed that hospitalists had an increased level of confidence in their bedside manner, patient satisfaction skills, and high-quality discharge discussion skills.
Inter-Rater Reliability
Inter-rater reliability for the discharge encounter, the daily rounding encounter, and the difficult patient encounter were 0.74 (95% CI, 0.64-0.84), 0.73 (95% CI, 0.63-0.82), and 0.73 (95% CI, 0.63-0.83), respectively.
HCAHPS
Four hundred sixteen HCAHPS surveys were returned in the year prior to our SP exercise, and the percent of patients who answered always to the questions on Courtesy and Respect, Listen, and Explain were 80.4%, 74.2 %, and 69.4 %, respectively. In the year after our SP exercise, 492 surveys were returned, and there was no significant change in HCAHP scores for the group (80.9% for Courtesy and Respect, 70.2% for the Listen question, and 70.5% for Explain).
DISCUSSION
We have shown that SPs can be used to assess deficits in hospitalist communication skills and provide feedback that can improve hospitalist confidence in performing optimal communication behaviors. We have also shown that hospitalists perceive the exercise as beneficial in improving their communication skills and perceive them as similar to their real patient encounters.
The Explain domain was significantly worse than the Courtesy and Respect and Listen domains for our hospitalists. Analysis of the checklist items within the Explain domain found that the items within this domain that were most problematic for hospitalists were summarizing information at the end of the encounter, using teach-back (a communication confirmation method where a healthcare provider asks a patient to repeat what was said to confirm understanding), encouraging additional questions by using open-ended statements (What questions do you have?) instead of close ended statements (Do you have any questions?), managing team and self-up, setting expectations on length of stay, and timing of tests. This correlated with our patient satisfaction HCAHPS data, which showed that patients consistently rated our hospitalists’ ability to explain things in a way they could understand lowest among the 3 questions. HCAHPS scores did not change after our SP exercise, and this lack of improvement may indicate that meaningful improvement in communication skills requires longitudinal interventions and real-time feedback rather than a single exercise, as was shown in a recent study looking at daily patient satisfaction score feedback given to internal medicine residents.14
Our study had several limitations. First, hospitalists knew they were being videotaped and observed, which may have altered their behaviors and may not reflect our hospitalists’ actual behaviors with patients. Furthermore, we did not examine whether the feedback given was incorporated into our hospitalists’ daily patient communications and whether this impacted our patients care other than examining HCAHPS scores.
CONCLUSION
SPs can be used to identify deficiencies in communication skills and provide specific guidance that improves hospitalist confidence in their communication skills.
Acknowledgment
This trial was funded by a grant from The Doctor’s Company Foundation.
Disclosure
None of the authors report any conflicts of interest.
1. Barnett PB. Rapport and the hospitalist. Am J Med. 2001;111(9B):31S-35S. PubMed
2. Kurtz S, Silverman J, Draper J. Teaching and learning communication skills in medicine.
2nd ed. London, UK: Radcliffe Publishing Ltd.; 2009.
3. Stewart MA. What is a successful doctor–patient interview? A study of interactions
and outcomes. Soc Sci Med. 1984;9:167-175. PubMed
4. Kaplan SH, Greenfield S, Ware JE. Assessing the effects of physician–patient interactions
on the outcomes of chronic disease. Med Care. 1989;27:S110-S127. PubMed
5. Levinson W, Lesser CS, Epstein RM. Developing physician communication skills for
patient-centered care. Health Aff (Millwood). 2010;29:1310-1318. PubMed
6. Griffin SJ, Kinmonth AL, Veltman MWM, Gillard S, Grant J, Stewart M. Effect
on health-related outcomes of interventions to alter the interaction between
patients and practitioners: a systematic review of trials. Ann Fam Med. 2004;2:
595-608. PubMed
7. Levinson W, Roter DL, Mullooly JP, Dull V, Frankel R. Physician-patient communication:
the relationship with malpractice claims among primary care physicians and
surgeons. JAMA. 1997;277:553-559. PubMed
8. Levinson W. Physician-patient communication: a key to malpractice prevention. [Editorial].
JAMA. 1994;272:1619-1620. PubMed
9. Beckman HB, Markakis KM, Suchman AL, Frankel RM. The doctor–patient relationship
and malpractice. Lessons from plaintiff depositions. Arch Intern Med.
1994;154:1365-1370. PubMed
10. Wofford MM, Wofford JL, Bothra J, Kendrick SB, Patient complaints about physician
behaviors: a qualitative study. Acad Med. 2004;79(2):134-138. PubMed
11. Studer Group. Acknowledge, Introduce, Duration, Explanation and Thank You.
http://www.studergroup.com/aidet. Accessed November 5, 2012.
12. SHM Discharge/Heart Failure Implementation Toolkit. https://www.hospitalmedicine.
org/Web/Quality_Innovation/Implementation_Toolkits/Congestive_Heart_
Failure/Web/Quality___Innovation/Implementation_Toolkit/CHF/CHF_overview.
aspx?hkey=f91120e3-6c8f-4a55-90e7-9b6a4b5472ef.
13. Carkhuff, RR. Helping and Human Relations: A Primer for Lay and Professional Helpers.
Volume I. New York, NY: Holt, Rinehart & Winston; 1969.
14. Banka G, Edgington S, Kyulo N, et al. Improving patient satisfaction through physician
education, feedback, and incentives. J Hosp Med. 2015;10:497-502. PubMed
Hospitalists must create rapport and communicate large amounts of information in a short amount of time without having a prior relationship with the patient.1 High-quality communication can improve satisfaction and compliance, while poor communication leaves patients ill prepared to transition back to the community.2–10
Many medical schools use standardized patients (SPs) to both train and evaluate their students’ communication skills. To our knowledge, no published studies describe using SPs to assess or teach communication skills for hospitalists.
Our objective in this study was to use SPs to assess for deficits in our hospitalists’ communication skills and to determine whether feedback provided by SPs could improve hospitalist confidence in and performance of optimal communication behaviors.
METHODS
Setting and Participants
Scenario and Checklist Development
We developed 3 SP encounters around common hospitalist-patient interactions: daily rounding, discharge, and interacting with a difficult patient. In order to assess communication skills, we developed a checklist with 3 core domains: Courtesy and Respect, Listen, and Explain. Each domain corresponded to 1 of 3 questions on the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey that pertained to doctor’s communications skills: (1) How often did doctors treat you with courtesy and respect? (2) How often did doctors listen carefully to you? (3) How often did doctors explain things in a way you could understand? We then developed checklist items that corresponded to essential communication skills within each of the 3 domains. These communication skills were based on best practices and published literature.
Discharge Encounter (Table 2): Patient admitted the night before with abdominal pain by another hospitalist. The checklist was based on AIDET®, an effective communication skills training protocol that our hospitalist group had been trained on.11
Daily Rounding Encounter (Table 3): Patient being discharged after an admission for congestive heart failure. The checklist was developed from the Society of Hospital Medicine discharge toolkit.12
Difficult Patient Encounter (Table 4): A patient and his daughter who were unhappy because of a previously missed lung mass that was now found to be cancer. Our checklist was based on characteristics of therapeutic bedside manner.13
The checklist items were each scored using a 3-point scale of adequate, partial, or inadequate performance. A description of checklist items within each of the 3 domains is listed in Table 1. A postintervention survey was filled out by all hospitalists after the 3 encounters.
Simulated Encounters
All 3 encounters occurred on the same day and each one lasted 1 hour (20 minutes for the encounter, 10 minutes for a posttest survey, and 30 minutes of feedback from the SP). For each case, a task list was given to the hospitalist before walking into the room (Appendix 1). During the feedback session, the SP gave the hospitalist feedback using the case checklist items. They then watched a video of the encounter and the SP further emphasized areas for improvement.
SP Training
SP training consisted of three 3-hour training sessions, which included review of the case, script, guidance on scoring the checklist items, role plays with attending hospitalists, and feedback training. Each SP was assigned to only 1 case.
Seven of the 24 encounters for each SP were reviewed independently by 2 investigators who created a final score for each checklist item which was compared to the SP’s checklist item score. The kappa (k) statistic was used to evaluate inter-observer reliability using the SAS system software (SAS Institute Inc.).
Analysis
The percent of hospitalists who performed each checklist item adequately within in each of the 3 domains (Courtesy and Respect, Listen, and Explain) was calculated. To compare the 3 domains, t tests were used.
We calculated the percent that our hospitalist group received on the 3 HCAHPS doctor’s questions 1 year prior to our SP exercise and 1 year after the SP exercise.
RESULTS
Twenty-three hospitalists completed all 3 encounters. For the 3 domains (Courtesy and Respect, Listen, and Explain), hospitalists performed significantly better in the Listen domain compared to the other 2 domains, with a mean percent adequate score of 90.2 % (95% confidence interval [CI], 72.2%-100%; P < 0.05), and significantly worse in the Explain domain compared to the other 2 domains, with a mean percent adequate score of 65.0% (95% CI, 49.2%-83.6%; P < 0.05). The mean percent adequate score for the Courtesy and Respect domain was 81.6% (95% CI, 56%-100%). This was significantly higher than the Explain domain and significantly lower than the Listen domain.
Posttest survey results showed that hospitalists had an increased level of confidence in their bedside manner, patient satisfaction skills, and high-quality discharge discussion skills.
Inter-Rater Reliability
Inter-rater reliability for the discharge encounter, the daily rounding encounter, and the difficult patient encounter were 0.74 (95% CI, 0.64-0.84), 0.73 (95% CI, 0.63-0.82), and 0.73 (95% CI, 0.63-0.83), respectively.
HCAHPS
Four hundred sixteen HCAHPS surveys were returned in the year prior to our SP exercise, and the percent of patients who answered always to the questions on Courtesy and Respect, Listen, and Explain were 80.4%, 74.2 %, and 69.4 %, respectively. In the year after our SP exercise, 492 surveys were returned, and there was no significant change in HCAHP scores for the group (80.9% for Courtesy and Respect, 70.2% for the Listen question, and 70.5% for Explain).
DISCUSSION
We have shown that SPs can be used to assess deficits in hospitalist communication skills and provide feedback that can improve hospitalist confidence in performing optimal communication behaviors. We have also shown that hospitalists perceive the exercise as beneficial in improving their communication skills and perceive them as similar to their real patient encounters.
The Explain domain was significantly worse than the Courtesy and Respect and Listen domains for our hospitalists. Analysis of the checklist items within the Explain domain found that the items within this domain that were most problematic for hospitalists were summarizing information at the end of the encounter, using teach-back (a communication confirmation method where a healthcare provider asks a patient to repeat what was said to confirm understanding), encouraging additional questions by using open-ended statements (What questions do you have?) instead of close ended statements (Do you have any questions?), managing team and self-up, setting expectations on length of stay, and timing of tests. This correlated with our patient satisfaction HCAHPS data, which showed that patients consistently rated our hospitalists’ ability to explain things in a way they could understand lowest among the 3 questions. HCAHPS scores did not change after our SP exercise, and this lack of improvement may indicate that meaningful improvement in communication skills requires longitudinal interventions and real-time feedback rather than a single exercise, as was shown in a recent study looking at daily patient satisfaction score feedback given to internal medicine residents.14
Our study had several limitations. First, hospitalists knew they were being videotaped and observed, which may have altered their behaviors and may not reflect our hospitalists’ actual behaviors with patients. Furthermore, we did not examine whether the feedback given was incorporated into our hospitalists’ daily patient communications and whether this impacted our patients care other than examining HCAHPS scores.
CONCLUSION
SPs can be used to identify deficiencies in communication skills and provide specific guidance that improves hospitalist confidence in their communication skills.
Acknowledgment
This trial was funded by a grant from The Doctor’s Company Foundation.
Disclosure
None of the authors report any conflicts of interest.
Hospitalists must create rapport and communicate large amounts of information in a short amount of time without having a prior relationship with the patient.1 High-quality communication can improve satisfaction and compliance, while poor communication leaves patients ill prepared to transition back to the community.2–10
Many medical schools use standardized patients (SPs) to both train and evaluate their students’ communication skills. To our knowledge, no published studies describe using SPs to assess or teach communication skills for hospitalists.
Our objective in this study was to use SPs to assess for deficits in our hospitalists’ communication skills and to determine whether feedback provided by SPs could improve hospitalist confidence in and performance of optimal communication behaviors.
METHODS
Setting and Participants
Scenario and Checklist Development
We developed 3 SP encounters around common hospitalist-patient interactions: daily rounding, discharge, and interacting with a difficult patient. In order to assess communication skills, we developed a checklist with 3 core domains: Courtesy and Respect, Listen, and Explain. Each domain corresponded to 1 of 3 questions on the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) survey that pertained to doctor’s communications skills: (1) How often did doctors treat you with courtesy and respect? (2) How often did doctors listen carefully to you? (3) How often did doctors explain things in a way you could understand? We then developed checklist items that corresponded to essential communication skills within each of the 3 domains. These communication skills were based on best practices and published literature.
Discharge Encounter (Table 2): Patient admitted the night before with abdominal pain by another hospitalist. The checklist was based on AIDET®, an effective communication skills training protocol that our hospitalist group had been trained on.11
Daily Rounding Encounter (Table 3): Patient being discharged after an admission for congestive heart failure. The checklist was developed from the Society of Hospital Medicine discharge toolkit.12
Difficult Patient Encounter (Table 4): A patient and his daughter who were unhappy because of a previously missed lung mass that was now found to be cancer. Our checklist was based on characteristics of therapeutic bedside manner.13
The checklist items were each scored using a 3-point scale of adequate, partial, or inadequate performance. A description of checklist items within each of the 3 domains is listed in Table 1. A postintervention survey was filled out by all hospitalists after the 3 encounters.
Simulated Encounters
All 3 encounters occurred on the same day and each one lasted 1 hour (20 minutes for the encounter, 10 minutes for a posttest survey, and 30 minutes of feedback from the SP). For each case, a task list was given to the hospitalist before walking into the room (Appendix 1). During the feedback session, the SP gave the hospitalist feedback using the case checklist items. They then watched a video of the encounter and the SP further emphasized areas for improvement.
SP Training
SP training consisted of three 3-hour training sessions, which included review of the case, script, guidance on scoring the checklist items, role plays with attending hospitalists, and feedback training. Each SP was assigned to only 1 case.
Seven of the 24 encounters for each SP were reviewed independently by 2 investigators who created a final score for each checklist item which was compared to the SP’s checklist item score. The kappa (k) statistic was used to evaluate inter-observer reliability using the SAS system software (SAS Institute Inc.).
Analysis
The percent of hospitalists who performed each checklist item adequately within in each of the 3 domains (Courtesy and Respect, Listen, and Explain) was calculated. To compare the 3 domains, t tests were used.
We calculated the percent that our hospitalist group received on the 3 HCAHPS doctor’s questions 1 year prior to our SP exercise and 1 year after the SP exercise.
RESULTS
Twenty-three hospitalists completed all 3 encounters. For the 3 domains (Courtesy and Respect, Listen, and Explain), hospitalists performed significantly better in the Listen domain compared to the other 2 domains, with a mean percent adequate score of 90.2 % (95% confidence interval [CI], 72.2%-100%; P < 0.05), and significantly worse in the Explain domain compared to the other 2 domains, with a mean percent adequate score of 65.0% (95% CI, 49.2%-83.6%; P < 0.05). The mean percent adequate score for the Courtesy and Respect domain was 81.6% (95% CI, 56%-100%). This was significantly higher than the Explain domain and significantly lower than the Listen domain.
Posttest survey results showed that hospitalists had an increased level of confidence in their bedside manner, patient satisfaction skills, and high-quality discharge discussion skills.
Inter-Rater Reliability
Inter-rater reliability for the discharge encounter, the daily rounding encounter, and the difficult patient encounter were 0.74 (95% CI, 0.64-0.84), 0.73 (95% CI, 0.63-0.82), and 0.73 (95% CI, 0.63-0.83), respectively.
HCAHPS
Four hundred sixteen HCAHPS surveys were returned in the year prior to our SP exercise, and the percent of patients who answered always to the questions on Courtesy and Respect, Listen, and Explain were 80.4%, 74.2 %, and 69.4 %, respectively. In the year after our SP exercise, 492 surveys were returned, and there was no significant change in HCAHP scores for the group (80.9% for Courtesy and Respect, 70.2% for the Listen question, and 70.5% for Explain).
DISCUSSION
We have shown that SPs can be used to assess deficits in hospitalist communication skills and provide feedback that can improve hospitalist confidence in performing optimal communication behaviors. We have also shown that hospitalists perceive the exercise as beneficial in improving their communication skills and perceive them as similar to their real patient encounters.
The Explain domain was significantly worse than the Courtesy and Respect and Listen domains for our hospitalists. Analysis of the checklist items within the Explain domain found that the items within this domain that were most problematic for hospitalists were summarizing information at the end of the encounter, using teach-back (a communication confirmation method where a healthcare provider asks a patient to repeat what was said to confirm understanding), encouraging additional questions by using open-ended statements (What questions do you have?) instead of close ended statements (Do you have any questions?), managing team and self-up, setting expectations on length of stay, and timing of tests. This correlated with our patient satisfaction HCAHPS data, which showed that patients consistently rated our hospitalists’ ability to explain things in a way they could understand lowest among the 3 questions. HCAHPS scores did not change after our SP exercise, and this lack of improvement may indicate that meaningful improvement in communication skills requires longitudinal interventions and real-time feedback rather than a single exercise, as was shown in a recent study looking at daily patient satisfaction score feedback given to internal medicine residents.14
Our study had several limitations. First, hospitalists knew they were being videotaped and observed, which may have altered their behaviors and may not reflect our hospitalists’ actual behaviors with patients. Furthermore, we did not examine whether the feedback given was incorporated into our hospitalists’ daily patient communications and whether this impacted our patients care other than examining HCAHPS scores.
CONCLUSION
SPs can be used to identify deficiencies in communication skills and provide specific guidance that improves hospitalist confidence in their communication skills.
Acknowledgment
This trial was funded by a grant from The Doctor’s Company Foundation.
Disclosure
None of the authors report any conflicts of interest.
1. Barnett PB. Rapport and the hospitalist. Am J Med. 2001;111(9B):31S-35S. PubMed
2. Kurtz S, Silverman J, Draper J. Teaching and learning communication skills in medicine.
2nd ed. London, UK: Radcliffe Publishing Ltd.; 2009.
3. Stewart MA. What is a successful doctor–patient interview? A study of interactions
and outcomes. Soc Sci Med. 1984;9:167-175. PubMed
4. Kaplan SH, Greenfield S, Ware JE. Assessing the effects of physician–patient interactions
on the outcomes of chronic disease. Med Care. 1989;27:S110-S127. PubMed
5. Levinson W, Lesser CS, Epstein RM. Developing physician communication skills for
patient-centered care. Health Aff (Millwood). 2010;29:1310-1318. PubMed
6. Griffin SJ, Kinmonth AL, Veltman MWM, Gillard S, Grant J, Stewart M. Effect
on health-related outcomes of interventions to alter the interaction between
patients and practitioners: a systematic review of trials. Ann Fam Med. 2004;2:
595-608. PubMed
7. Levinson W, Roter DL, Mullooly JP, Dull V, Frankel R. Physician-patient communication:
the relationship with malpractice claims among primary care physicians and
surgeons. JAMA. 1997;277:553-559. PubMed
8. Levinson W. Physician-patient communication: a key to malpractice prevention. [Editorial].
JAMA. 1994;272:1619-1620. PubMed
9. Beckman HB, Markakis KM, Suchman AL, Frankel RM. The doctor–patient relationship
and malpractice. Lessons from plaintiff depositions. Arch Intern Med.
1994;154:1365-1370. PubMed
10. Wofford MM, Wofford JL, Bothra J, Kendrick SB, Patient complaints about physician
behaviors: a qualitative study. Acad Med. 2004;79(2):134-138. PubMed
11. Studer Group. Acknowledge, Introduce, Duration, Explanation and Thank You.
http://www.studergroup.com/aidet. Accessed November 5, 2012.
12. SHM Discharge/Heart Failure Implementation Toolkit. https://www.hospitalmedicine.
org/Web/Quality_Innovation/Implementation_Toolkits/Congestive_Heart_
Failure/Web/Quality___Innovation/Implementation_Toolkit/CHF/CHF_overview.
aspx?hkey=f91120e3-6c8f-4a55-90e7-9b6a4b5472ef.
13. Carkhuff, RR. Helping and Human Relations: A Primer for Lay and Professional Helpers.
Volume I. New York, NY: Holt, Rinehart & Winston; 1969.
14. Banka G, Edgington S, Kyulo N, et al. Improving patient satisfaction through physician
education, feedback, and incentives. J Hosp Med. 2015;10:497-502. PubMed
1. Barnett PB. Rapport and the hospitalist. Am J Med. 2001;111(9B):31S-35S. PubMed
2. Kurtz S, Silverman J, Draper J. Teaching and learning communication skills in medicine.
2nd ed. London, UK: Radcliffe Publishing Ltd.; 2009.
3. Stewart MA. What is a successful doctor–patient interview? A study of interactions
and outcomes. Soc Sci Med. 1984;9:167-175. PubMed
4. Kaplan SH, Greenfield S, Ware JE. Assessing the effects of physician–patient interactions
on the outcomes of chronic disease. Med Care. 1989;27:S110-S127. PubMed
5. Levinson W, Lesser CS, Epstein RM. Developing physician communication skills for
patient-centered care. Health Aff (Millwood). 2010;29:1310-1318. PubMed
6. Griffin SJ, Kinmonth AL, Veltman MWM, Gillard S, Grant J, Stewart M. Effect
on health-related outcomes of interventions to alter the interaction between
patients and practitioners: a systematic review of trials. Ann Fam Med. 2004;2:
595-608. PubMed
7. Levinson W, Roter DL, Mullooly JP, Dull V, Frankel R. Physician-patient communication:
the relationship with malpractice claims among primary care physicians and
surgeons. JAMA. 1997;277:553-559. PubMed
8. Levinson W. Physician-patient communication: a key to malpractice prevention. [Editorial].
JAMA. 1994;272:1619-1620. PubMed
9. Beckman HB, Markakis KM, Suchman AL, Frankel RM. The doctor–patient relationship
and malpractice. Lessons from plaintiff depositions. Arch Intern Med.
1994;154:1365-1370. PubMed
10. Wofford MM, Wofford JL, Bothra J, Kendrick SB, Patient complaints about physician
behaviors: a qualitative study. Acad Med. 2004;79(2):134-138. PubMed
11. Studer Group. Acknowledge, Introduce, Duration, Explanation and Thank You.
http://www.studergroup.com/aidet. Accessed November 5, 2012.
12. SHM Discharge/Heart Failure Implementation Toolkit. https://www.hospitalmedicine.
org/Web/Quality_Innovation/Implementation_Toolkits/Congestive_Heart_
Failure/Web/Quality___Innovation/Implementation_Toolkit/CHF/CHF_overview.
aspx?hkey=f91120e3-6c8f-4a55-90e7-9b6a4b5472ef.
13. Carkhuff, RR. Helping and Human Relations: A Primer for Lay and Professional Helpers.
Volume I. New York, NY: Holt, Rinehart & Winston; 1969.
14. Banka G, Edgington S, Kyulo N, et al. Improving patient satisfaction through physician
education, feedback, and incentives. J Hosp Med. 2015;10:497-502. PubMed
© 2017 Society of Hospital Medicine
Fecal occult blood testing in hospitalized patients with upper gastrointestinal bleeding
The “Things We Do for No Reason” (TWDFNR) series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE REPORT
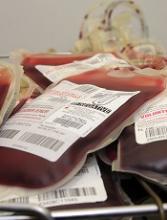
A 47-year-old man with a history of alcohol abuse, cirrhosis, and grade II esophageal varices is admitted for treatment of alcohol withdrawal. He reports having some dark-colored stools a week prior to admission, but his stools since then have been normal in color. A repeat hemoglobin is stable, but a fecal occult blood test is positive. What should be done next?
BACKGROUND
The US Preventive Services Task Force and the American College of Gastroenterology recommend fecal occult blood testing (FOBT) as one method for colorectal cancer (CRC) screening in average risk populations.1,2 FOBTs can be divided into guaiac-based tests (gFOBTs), which measure heme, and fecal immunochemical tests (FITs), which measure the globin portion of human hemoglobin (Hb). In gFOBTs, heme present in the sample reacts with a hydrogen peroxide developer to oxidize guaiac, producing a blue color.3 Screening gFOBT was shown to decrease mortality from CRC in several landmark studies in the 1990s, but its sensitivity is poor, ranging from 30% to 57%.4 Because the guaiac-induced color change is determined visually, interpretation of gFOBT results are subject to error. In a survey of 173 medical providers, 12% did not accurately interpret gFOBT results.5 In light of these limitations, recent guidelines support the use of newer FITs for CRC screening. FITs utilize antibodies directed against the human globin moiety and demonstrate an increased sensitivity when compared with gFOBTs (by 32% to 62%) for detecting neoplasm.6 While evidence supports the use of FOBTs in CRC screening, providers use these tests for nonvalidated purposes, including the evaluation of suspected acute upper gastrointestinal bleeding (UGIB).
WHY YOU MIGHT THINK FOBT is HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
Given the incidence (up to 100 per 100,000 persons per year) and high mortality of UGIB (up to 20,000 deaths annually in the United States),7 there would ideally be a noninvasive test available to help guide management. In evaluating a patient with possible acute UGIB, FOBT affords several theoretical benefits. FOBT is quick, inexpensive, and can be performed by any health professional. In contrast, the primary diagnostic procedure for UGIB, esophagogastroduodenoscopy (EGD), carries procedural and sedation-related risks, can be costly and time-consuming, and requires consultation from subspecialty providers.
WHY FOBT is NOT HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
While FOBTs are valuable as screening tests for CRC in the outpatient setting, their use has been extended to diagnose gastrointestinal (GI) bleeding in the inpatient setting without supporting data. As is true for many screening tests, FOBT is associated with a high incidence of false-positive results, or type I errors.8,9 False-positive FOBT results can occur from ingested blood via extra-intestinal sources (eg, epistaxis, gingival bleeding, pharyngitis, hemoptysis), or in medical conditions with intestinal mucosal inflammation (eg, esophagitis, gastritis, inflammatory bowel disease). False-positive results can also be due to clinically insignificant GI blood loss induced by medications (eg, aspirin, nonsteroidal anti-inflammatory drugs), alcohol,10 or by ingestion of meats, fruits, or vegetables containing peroxidase (eg, broccoli, cauliflower).11
Outpatients using FOBTs for cancer screening are advised to hold medications and avoid foods that may lead to false-positive results. Despite institution of these restrictions, false-positive rates are still high, as 37% to 53% of CRC screening patients with a positive FOBT have a subsequent negative colonoscopy, and only 11% to 21% of these patients have a source of bleeding identified on subsequent EGD.12 False-positive results might be even higher in the inpatient setting, where patients typically do not adhere to these restrictions. A review of FOBTs performed in 3 acute care hospitals revealed that 65% of patients tested were on at least one medication that impacted the validity of gFOBT results, and 98% had no evidence of dietary restriction prior to testing.13
The use of FOBTs (particularly FITs) is also subject to false-negative results, or type II errors. While FITs have increased specificity for lower GI bleeding, their ability to detect UGIB is limited, because most Hb is digested in the small intestine and not present in rectal stool.14 In a study of more than 2,700 patients, FIT results were not correlated with the presence of upper GI pathology.15 False-negative results are less common with gFOBTs, although these may occur with low volume, slow or intermittent bleeding,16 or with ingestion of substances that inhibit oxidation, such as vitamin C.17
Beyond these test limitations, studies suggest that the majority of inpatient FOBT results do not impact immediate medical decision-making or management. In one study, only 34% of hospitalized patients with a positive FOBT underwent further GI studies, with the majority of those patients (60%) receiving endoscopy before the results of the FOBT were known.18 In another study of 201 FOBTs performed on hospitalized patients, those with negative results underwent further GI evaluation at a higher rate than those with positive results (41% vs 38%).8 This aligns with a study that revealed the majority of patients suspected of having a GI bleed underwent endoscopic evaluation regardless of the FOBT result.9
WHEN MIGHT FOBT BE HELPFUL?
FOBT currently has a role in CRC screening and
WHAT WE SHOULD DO INSTEAD
A careful history, physical examination, and visual inspection of the stool remain the foundation of establishing UGIB as the etiology of anemia. Observed melena (either by passed stool or a rectal examination) has a likelihood ratio (LR) of 25 for UGIB; a patient’s self-report of stools that sounds melenic (black or tarry) has an LR of 5-6.19 An upper GI source may be further supported by an elevated blood urea nitrogen (BUN) to creatinine ratio, as blood is absorbed through the small bowel and patients may have concomitant decreased renal perfusion. A BUN to creatinine ratio of >30 is associated with a positive LR (LR+) of 7.5 for UGIB.19 Recall that the higher the LR+, and the lower the negative LR (LR-), the better the test is at ruling in and out the diagnosis, respectively. LR+ of 2–10 and LR– of 0.1–0.5 represent a modestly helpful diagnostic test, whereas LR+ >10 and LR- <0.1 are considered robust. These are generalizations only, as value of LR+/LR- depends on pretest probability.
Although Gastroccult23 may be considered for the detection of occult blood in gastric juice, its package insert states: “As with any occult blood test, results with the Gastroccult test cannot be considered conclusive evidence of the presence or absence of upper gastrointestinal bleeding or pathology.” As with any diagnostic evaluation, we would only recommend this test if it would change management.
- FOBT should not be performed to diagnose UGIB.
- When there is clinical suspicion of acute GI bleeding, the best diagnostic tools are a good history, physical examination, and visual inspection of the stool by the clinician to determine the presence of hematochezia or melena.
- Deferring FOBT to the ambulatory setting may improve test performance characteristics.
CONCLUSION
FOBT is validated as an outpatient colon cancer screening tool in asymptomatic patients, not for inpatient evaluation of acute GIB. Given the poor positive predictive value for a positive FOBT in an acute GIB scenario, the potential risk for unnecessary treatments or procedures is real. Conversely, a negative FOBT (particularly FIT) does not rule out GI bleeding and risks a false sense of security that may result in under-treatment. In most scenarios in which FOBT is performed, clinicians can make decisions based on a composite of history, physical exam, visual inspection of the stool, and laboratory investigation. Until further research substantiates the utility of FOBT for this purpose, we would recommend against the routine use of FOBT for evaluating UGIB in hospitalized patients.
Acknowledgment
Disclosure: The authors do not have any relevant financial disclosures to report. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason”? Let us know what you do in your practice and propose ideas for other “Things We Do for No Reason” topics. Please join in the conversation online at Twitter (#TWDFNR)/Facebook and don’t forget to “Like It” on Facebook or retweet it on Twitter. We invite you to propose ideas for other “Things We Do for No Reason” topics by e-mailingTWDFNR@hospitalmedicine.org.
1. U.S. Preventive Services Task Force. Screening for colorectal cancer: recommendation and rationale. Ann Intern Med. 2002;137:129-131. PubMed
2. Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143(3):844-857. PubMed
3. Carroll MRR, Seaman HE, Halloran HP. Tests and investigations for colorectal cancer screening. Clinical Biochemistry. 2014;47:921-939. PubMed
4. Tinmouth J, Lansdorp-Vogelaar I, Allison JE. Faecal immunochemical tests versus guaiac faecal occult blood tests: what clinicians and colorectal cancer screening programme organisers need to know. Gut. 2015;64(8):1327-1337. PubMed
5. Selinger RR, et al. Failure of health care professionals to interpret fecal occult blood tests accurately. Am J Med. 2003;114(1):64-67. PubMed
6. Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM. American College of Gastroenterology Guidelines for Colorectal Cancer Screening 2008. Am J Gastroenterol. 2009;104(3):739-750. PubMed
7. El-Tawil AM. Trends on gastrointestinal bleeding and mortality: Where are we standing? World J Gastroenterol. 2012;18(11):1154. PubMed
8. van Rijn AF, Stroobants AK, Deutekom M, et al. Inappropriate use of the faecal occult blood test in a university hospital in the Netherlands. Eur J Gastroenterol Hepatol. 2012;24(11):1266-1269. PubMed
9. Narula N, Ulic D, Al-Dabbagh R, et al. Fecal occult blood testing as a diagnostic test in symptomatic patients is not useful: a retrospective chart review. Can J Gastroenterol Hepatol. 2014;28(8):421-426. PubMed
10. Fleming, JL, Ahlquist DA, McGill DB, Zinsmeister AR, Ellefson RD, Schwartz S. Influence of aspirin and ethanol on fecal blood levels as determined by using the HemoQuant assay. Mayo Clin Proc. 1987;62(3):159-163. PubMed
11. Macrae FA, St John DJB. Relationship between patterns of bleeding and Hemoccult sensitivity in patients with colorectal cancers or adenomas. Gastroenterology. 1982;82:891-898. PubMed
12. Allard J, et al. Gastroscopy following a positive fecal occult blood test and negative colonoscopy: systematic review and guideline. Can J Gastroenterol. 2010;24(2):113-120. PubMed
13. Friedman A, Chan A, Chin LC, Deen A, Hammerschlag G, Lee M, et al. Use and abuse of faecal occult blood tests in an acute hospital inpatient setting. Intern Med J. 2010;40(2):107-111. PubMed
14. Allison JE, et al. Screening for colorectal neoplasms with new fecal occult blood tests: update on performance characteristics. J Natl Cancer Inst. 2007;99(19):1462-1470. PubMed
15. Chiang TH, Lee YC, Tu CH, Chiu HM, Wu MS. Performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract. CMAJ. 2011;183(13):1474-1481. PubMed
16. Bassett ML, Goulston KJ. False positive and negative hemoccult reactions on a normal diet and effect of diet restriction. Aust N Z J Med. 1980;10(1):1-4. PubMed
17. Jaffe, RM, Kasten B, Young DS, MacLowry JD. False-negative stool occult blood tests caused by ingestion of ascorbic acid (vitamin C). Ann Intern Med. 1975;83(6):824-826. PubMed
18. Ip S, Sokoro AAH, Kaita L, Ruiz C, McIntyre E, Singh H. Use of fecal occult blood testing in hospitalized patients: results of an audit. Can J Gastroenterol Hepatol. 2014;28(9):489-494. PubMed
19. Srygley FD, Gerardo CJ, Trun T, Fisher DA. Does this patient have a severe upper gastrointestinal bleed? JAMA. 2012;307(10):1072-1079. PubMed
20. Logue KA. Data Request - FOBT. June 2016. Regions Hospital, HealthPartners Laboratory, Saint Paul, Minnesota.
21. Population Clock. http://www.census.gov/popclock/. Accessed July 8, 2016.
22. Mosadeghi S, Ren H, Yen I, Bhuket T. Evaluation of fecal occult blood testing in the acute hospital setting. Gastrointestinal Endoscopy. 2015;81(5).
23. Gastroccult [package insert]. Beckman Coulter, Brea, CA. https://www.beckmancoulter.com/wsrportal/wsr/diagnostics/clinical-products/rapid-diagnostics/gas troccult/index.htm. Accessed March 18, 2008.
The “Things We Do for No Reason” (TWDFNR) series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE REPORT
A 47-year-old man with a history of alcohol abuse, cirrhosis, and grade II esophageal varices is admitted for treatment of alcohol withdrawal. He reports having some dark-colored stools a week prior to admission, but his stools since then have been normal in color. A repeat hemoglobin is stable, but a fecal occult blood test is positive. What should be done next?
BACKGROUND
The US Preventive Services Task Force and the American College of Gastroenterology recommend fecal occult blood testing (FOBT) as one method for colorectal cancer (CRC) screening in average risk populations.1,2 FOBTs can be divided into guaiac-based tests (gFOBTs), which measure heme, and fecal immunochemical tests (FITs), which measure the globin portion of human hemoglobin (Hb). In gFOBTs, heme present in the sample reacts with a hydrogen peroxide developer to oxidize guaiac, producing a blue color.3 Screening gFOBT was shown to decrease mortality from CRC in several landmark studies in the 1990s, but its sensitivity is poor, ranging from 30% to 57%.4 Because the guaiac-induced color change is determined visually, interpretation of gFOBT results are subject to error. In a survey of 173 medical providers, 12% did not accurately interpret gFOBT results.5 In light of these limitations, recent guidelines support the use of newer FITs for CRC screening. FITs utilize antibodies directed against the human globin moiety and demonstrate an increased sensitivity when compared with gFOBTs (by 32% to 62%) for detecting neoplasm.6 While evidence supports the use of FOBTs in CRC screening, providers use these tests for nonvalidated purposes, including the evaluation of suspected acute upper gastrointestinal bleeding (UGIB).
WHY YOU MIGHT THINK FOBT is HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
Given the incidence (up to 100 per 100,000 persons per year) and high mortality of UGIB (up to 20,000 deaths annually in the United States),7 there would ideally be a noninvasive test available to help guide management. In evaluating a patient with possible acute UGIB, FOBT affords several theoretical benefits. FOBT is quick, inexpensive, and can be performed by any health professional. In contrast, the primary diagnostic procedure for UGIB, esophagogastroduodenoscopy (EGD), carries procedural and sedation-related risks, can be costly and time-consuming, and requires consultation from subspecialty providers.
WHY FOBT is NOT HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
While FOBTs are valuable as screening tests for CRC in the outpatient setting, their use has been extended to diagnose gastrointestinal (GI) bleeding in the inpatient setting without supporting data. As is true for many screening tests, FOBT is associated with a high incidence of false-positive results, or type I errors.8,9 False-positive FOBT results can occur from ingested blood via extra-intestinal sources (eg, epistaxis, gingival bleeding, pharyngitis, hemoptysis), or in medical conditions with intestinal mucosal inflammation (eg, esophagitis, gastritis, inflammatory bowel disease). False-positive results can also be due to clinically insignificant GI blood loss induced by medications (eg, aspirin, nonsteroidal anti-inflammatory drugs), alcohol,10 or by ingestion of meats, fruits, or vegetables containing peroxidase (eg, broccoli, cauliflower).11
Outpatients using FOBTs for cancer screening are advised to hold medications and avoid foods that may lead to false-positive results. Despite institution of these restrictions, false-positive rates are still high, as 37% to 53% of CRC screening patients with a positive FOBT have a subsequent negative colonoscopy, and only 11% to 21% of these patients have a source of bleeding identified on subsequent EGD.12 False-positive results might be even higher in the inpatient setting, where patients typically do not adhere to these restrictions. A review of FOBTs performed in 3 acute care hospitals revealed that 65% of patients tested were on at least one medication that impacted the validity of gFOBT results, and 98% had no evidence of dietary restriction prior to testing.13
The use of FOBTs (particularly FITs) is also subject to false-negative results, or type II errors. While FITs have increased specificity for lower GI bleeding, their ability to detect UGIB is limited, because most Hb is digested in the small intestine and not present in rectal stool.14 In a study of more than 2,700 patients, FIT results were not correlated with the presence of upper GI pathology.15 False-negative results are less common with gFOBTs, although these may occur with low volume, slow or intermittent bleeding,16 or with ingestion of substances that inhibit oxidation, such as vitamin C.17
Beyond these test limitations, studies suggest that the majority of inpatient FOBT results do not impact immediate medical decision-making or management. In one study, only 34% of hospitalized patients with a positive FOBT underwent further GI studies, with the majority of those patients (60%) receiving endoscopy before the results of the FOBT were known.18 In another study of 201 FOBTs performed on hospitalized patients, those with negative results underwent further GI evaluation at a higher rate than those with positive results (41% vs 38%).8 This aligns with a study that revealed the majority of patients suspected of having a GI bleed underwent endoscopic evaluation regardless of the FOBT result.9
WHEN MIGHT FOBT BE HELPFUL?
FOBT currently has a role in CRC screening and
WHAT WE SHOULD DO INSTEAD
A careful history, physical examination, and visual inspection of the stool remain the foundation of establishing UGIB as the etiology of anemia. Observed melena (either by passed stool or a rectal examination) has a likelihood ratio (LR) of 25 for UGIB; a patient’s self-report of stools that sounds melenic (black or tarry) has an LR of 5-6.19 An upper GI source may be further supported by an elevated blood urea nitrogen (BUN) to creatinine ratio, as blood is absorbed through the small bowel and patients may have concomitant decreased renal perfusion. A BUN to creatinine ratio of >30 is associated with a positive LR (LR+) of 7.5 for UGIB.19 Recall that the higher the LR+, and the lower the negative LR (LR-), the better the test is at ruling in and out the diagnosis, respectively. LR+ of 2–10 and LR– of 0.1–0.5 represent a modestly helpful diagnostic test, whereas LR+ >10 and LR- <0.1 are considered robust. These are generalizations only, as value of LR+/LR- depends on pretest probability.
Although Gastroccult23 may be considered for the detection of occult blood in gastric juice, its package insert states: “As with any occult blood test, results with the Gastroccult test cannot be considered conclusive evidence of the presence or absence of upper gastrointestinal bleeding or pathology.” As with any diagnostic evaluation, we would only recommend this test if it would change management.
- FOBT should not be performed to diagnose UGIB.
- When there is clinical suspicion of acute GI bleeding, the best diagnostic tools are a good history, physical examination, and visual inspection of the stool by the clinician to determine the presence of hematochezia or melena.
- Deferring FOBT to the ambulatory setting may improve test performance characteristics.
CONCLUSION
FOBT is validated as an outpatient colon cancer screening tool in asymptomatic patients, not for inpatient evaluation of acute GIB. Given the poor positive predictive value for a positive FOBT in an acute GIB scenario, the potential risk for unnecessary treatments or procedures is real. Conversely, a negative FOBT (particularly FIT) does not rule out GI bleeding and risks a false sense of security that may result in under-treatment. In most scenarios in which FOBT is performed, clinicians can make decisions based on a composite of history, physical exam, visual inspection of the stool, and laboratory investigation. Until further research substantiates the utility of FOBT for this purpose, we would recommend against the routine use of FOBT for evaluating UGIB in hospitalized patients.
Acknowledgment
Disclosure: The authors do not have any relevant financial disclosures to report. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason”? Let us know what you do in your practice and propose ideas for other “Things We Do for No Reason” topics. Please join in the conversation online at Twitter (#TWDFNR)/Facebook and don’t forget to “Like It” on Facebook or retweet it on Twitter. We invite you to propose ideas for other “Things We Do for No Reason” topics by e-mailingTWDFNR@hospitalmedicine.org.
The “Things We Do for No Reason” (TWDFNR) series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE REPORT
A 47-year-old man with a history of alcohol abuse, cirrhosis, and grade II esophageal varices is admitted for treatment of alcohol withdrawal. He reports having some dark-colored stools a week prior to admission, but his stools since then have been normal in color. A repeat hemoglobin is stable, but a fecal occult blood test is positive. What should be done next?
BACKGROUND
The US Preventive Services Task Force and the American College of Gastroenterology recommend fecal occult blood testing (FOBT) as one method for colorectal cancer (CRC) screening in average risk populations.1,2 FOBTs can be divided into guaiac-based tests (gFOBTs), which measure heme, and fecal immunochemical tests (FITs), which measure the globin portion of human hemoglobin (Hb). In gFOBTs, heme present in the sample reacts with a hydrogen peroxide developer to oxidize guaiac, producing a blue color.3 Screening gFOBT was shown to decrease mortality from CRC in several landmark studies in the 1990s, but its sensitivity is poor, ranging from 30% to 57%.4 Because the guaiac-induced color change is determined visually, interpretation of gFOBT results are subject to error. In a survey of 173 medical providers, 12% did not accurately interpret gFOBT results.5 In light of these limitations, recent guidelines support the use of newer FITs for CRC screening. FITs utilize antibodies directed against the human globin moiety and demonstrate an increased sensitivity when compared with gFOBTs (by 32% to 62%) for detecting neoplasm.6 While evidence supports the use of FOBTs in CRC screening, providers use these tests for nonvalidated purposes, including the evaluation of suspected acute upper gastrointestinal bleeding (UGIB).
WHY YOU MIGHT THINK FOBT is HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
Given the incidence (up to 100 per 100,000 persons per year) and high mortality of UGIB (up to 20,000 deaths annually in the United States),7 there would ideally be a noninvasive test available to help guide management. In evaluating a patient with possible acute UGIB, FOBT affords several theoretical benefits. FOBT is quick, inexpensive, and can be performed by any health professional. In contrast, the primary diagnostic procedure for UGIB, esophagogastroduodenoscopy (EGD), carries procedural and sedation-related risks, can be costly and time-consuming, and requires consultation from subspecialty providers.
WHY FOBT is NOT HELPFUL FOR EVALUATION OF INPATIENTS WITH SUSPECTED ACUTE UGIB
While FOBTs are valuable as screening tests for CRC in the outpatient setting, their use has been extended to diagnose gastrointestinal (GI) bleeding in the inpatient setting without supporting data. As is true for many screening tests, FOBT is associated with a high incidence of false-positive results, or type I errors.8,9 False-positive FOBT results can occur from ingested blood via extra-intestinal sources (eg, epistaxis, gingival bleeding, pharyngitis, hemoptysis), or in medical conditions with intestinal mucosal inflammation (eg, esophagitis, gastritis, inflammatory bowel disease). False-positive results can also be due to clinically insignificant GI blood loss induced by medications (eg, aspirin, nonsteroidal anti-inflammatory drugs), alcohol,10 or by ingestion of meats, fruits, or vegetables containing peroxidase (eg, broccoli, cauliflower).11
Outpatients using FOBTs for cancer screening are advised to hold medications and avoid foods that may lead to false-positive results. Despite institution of these restrictions, false-positive rates are still high, as 37% to 53% of CRC screening patients with a positive FOBT have a subsequent negative colonoscopy, and only 11% to 21% of these patients have a source of bleeding identified on subsequent EGD.12 False-positive results might be even higher in the inpatient setting, where patients typically do not adhere to these restrictions. A review of FOBTs performed in 3 acute care hospitals revealed that 65% of patients tested were on at least one medication that impacted the validity of gFOBT results, and 98% had no evidence of dietary restriction prior to testing.13
The use of FOBTs (particularly FITs) is also subject to false-negative results, or type II errors. While FITs have increased specificity for lower GI bleeding, their ability to detect UGIB is limited, because most Hb is digested in the small intestine and not present in rectal stool.14 In a study of more than 2,700 patients, FIT results were not correlated with the presence of upper GI pathology.15 False-negative results are less common with gFOBTs, although these may occur with low volume, slow or intermittent bleeding,16 or with ingestion of substances that inhibit oxidation, such as vitamin C.17
Beyond these test limitations, studies suggest that the majority of inpatient FOBT results do not impact immediate medical decision-making or management. In one study, only 34% of hospitalized patients with a positive FOBT underwent further GI studies, with the majority of those patients (60%) receiving endoscopy before the results of the FOBT were known.18 In another study of 201 FOBTs performed on hospitalized patients, those with negative results underwent further GI evaluation at a higher rate than those with positive results (41% vs 38%).8 This aligns with a study that revealed the majority of patients suspected of having a GI bleed underwent endoscopic evaluation regardless of the FOBT result.9
WHEN MIGHT FOBT BE HELPFUL?
FOBT currently has a role in CRC screening and
WHAT WE SHOULD DO INSTEAD
A careful history, physical examination, and visual inspection of the stool remain the foundation of establishing UGIB as the etiology of anemia. Observed melena (either by passed stool or a rectal examination) has a likelihood ratio (LR) of 25 for UGIB; a patient’s self-report of stools that sounds melenic (black or tarry) has an LR of 5-6.19 An upper GI source may be further supported by an elevated blood urea nitrogen (BUN) to creatinine ratio, as blood is absorbed through the small bowel and patients may have concomitant decreased renal perfusion. A BUN to creatinine ratio of >30 is associated with a positive LR (LR+) of 7.5 for UGIB.19 Recall that the higher the LR+, and the lower the negative LR (LR-), the better the test is at ruling in and out the diagnosis, respectively. LR+ of 2–10 and LR– of 0.1–0.5 represent a modestly helpful diagnostic test, whereas LR+ >10 and LR- <0.1 are considered robust. These are generalizations only, as value of LR+/LR- depends on pretest probability.
Although Gastroccult23 may be considered for the detection of occult blood in gastric juice, its package insert states: “As with any occult blood test, results with the Gastroccult test cannot be considered conclusive evidence of the presence or absence of upper gastrointestinal bleeding or pathology.” As with any diagnostic evaluation, we would only recommend this test if it would change management.
- FOBT should not be performed to diagnose UGIB.
- When there is clinical suspicion of acute GI bleeding, the best diagnostic tools are a good history, physical examination, and visual inspection of the stool by the clinician to determine the presence of hematochezia or melena.
- Deferring FOBT to the ambulatory setting may improve test performance characteristics.
CONCLUSION
FOBT is validated as an outpatient colon cancer screening tool in asymptomatic patients, not for inpatient evaluation of acute GIB. Given the poor positive predictive value for a positive FOBT in an acute GIB scenario, the potential risk for unnecessary treatments or procedures is real. Conversely, a negative FOBT (particularly FIT) does not rule out GI bleeding and risks a false sense of security that may result in under-treatment. In most scenarios in which FOBT is performed, clinicians can make decisions based on a composite of history, physical exam, visual inspection of the stool, and laboratory investigation. Until further research substantiates the utility of FOBT for this purpose, we would recommend against the routine use of FOBT for evaluating UGIB in hospitalized patients.
Acknowledgment
Disclosure: The authors do not have any relevant financial disclosures to report. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason”? Let us know what you do in your practice and propose ideas for other “Things We Do for No Reason” topics. Please join in the conversation online at Twitter (#TWDFNR)/Facebook and don’t forget to “Like It” on Facebook or retweet it on Twitter. We invite you to propose ideas for other “Things We Do for No Reason” topics by e-mailingTWDFNR@hospitalmedicine.org.
1. U.S. Preventive Services Task Force. Screening for colorectal cancer: recommendation and rationale. Ann Intern Med. 2002;137:129-131. PubMed
2. Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143(3):844-857. PubMed
3. Carroll MRR, Seaman HE, Halloran HP. Tests and investigations for colorectal cancer screening. Clinical Biochemistry. 2014;47:921-939. PubMed
4. Tinmouth J, Lansdorp-Vogelaar I, Allison JE. Faecal immunochemical tests versus guaiac faecal occult blood tests: what clinicians and colorectal cancer screening programme organisers need to know. Gut. 2015;64(8):1327-1337. PubMed
5. Selinger RR, et al. Failure of health care professionals to interpret fecal occult blood tests accurately. Am J Med. 2003;114(1):64-67. PubMed
6. Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM. American College of Gastroenterology Guidelines for Colorectal Cancer Screening 2008. Am J Gastroenterol. 2009;104(3):739-750. PubMed
7. El-Tawil AM. Trends on gastrointestinal bleeding and mortality: Where are we standing? World J Gastroenterol. 2012;18(11):1154. PubMed
8. van Rijn AF, Stroobants AK, Deutekom M, et al. Inappropriate use of the faecal occult blood test in a university hospital in the Netherlands. Eur J Gastroenterol Hepatol. 2012;24(11):1266-1269. PubMed
9. Narula N, Ulic D, Al-Dabbagh R, et al. Fecal occult blood testing as a diagnostic test in symptomatic patients is not useful: a retrospective chart review. Can J Gastroenterol Hepatol. 2014;28(8):421-426. PubMed
10. Fleming, JL, Ahlquist DA, McGill DB, Zinsmeister AR, Ellefson RD, Schwartz S. Influence of aspirin and ethanol on fecal blood levels as determined by using the HemoQuant assay. Mayo Clin Proc. 1987;62(3):159-163. PubMed
11. Macrae FA, St John DJB. Relationship between patterns of bleeding and Hemoccult sensitivity in patients with colorectal cancers or adenomas. Gastroenterology. 1982;82:891-898. PubMed
12. Allard J, et al. Gastroscopy following a positive fecal occult blood test and negative colonoscopy: systematic review and guideline. Can J Gastroenterol. 2010;24(2):113-120. PubMed
13. Friedman A, Chan A, Chin LC, Deen A, Hammerschlag G, Lee M, et al. Use and abuse of faecal occult blood tests in an acute hospital inpatient setting. Intern Med J. 2010;40(2):107-111. PubMed
14. Allison JE, et al. Screening for colorectal neoplasms with new fecal occult blood tests: update on performance characteristics. J Natl Cancer Inst. 2007;99(19):1462-1470. PubMed
15. Chiang TH, Lee YC, Tu CH, Chiu HM, Wu MS. Performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract. CMAJ. 2011;183(13):1474-1481. PubMed
16. Bassett ML, Goulston KJ. False positive and negative hemoccult reactions on a normal diet and effect of diet restriction. Aust N Z J Med. 1980;10(1):1-4. PubMed
17. Jaffe, RM, Kasten B, Young DS, MacLowry JD. False-negative stool occult blood tests caused by ingestion of ascorbic acid (vitamin C). Ann Intern Med. 1975;83(6):824-826. PubMed
18. Ip S, Sokoro AAH, Kaita L, Ruiz C, McIntyre E, Singh H. Use of fecal occult blood testing in hospitalized patients: results of an audit. Can J Gastroenterol Hepatol. 2014;28(9):489-494. PubMed
19. Srygley FD, Gerardo CJ, Trun T, Fisher DA. Does this patient have a severe upper gastrointestinal bleed? JAMA. 2012;307(10):1072-1079. PubMed
20. Logue KA. Data Request - FOBT. June 2016. Regions Hospital, HealthPartners Laboratory, Saint Paul, Minnesota.
21. Population Clock. http://www.census.gov/popclock/. Accessed July 8, 2016.
22. Mosadeghi S, Ren H, Yen I, Bhuket T. Evaluation of fecal occult blood testing in the acute hospital setting. Gastrointestinal Endoscopy. 2015;81(5).
23. Gastroccult [package insert]. Beckman Coulter, Brea, CA. https://www.beckmancoulter.com/wsrportal/wsr/diagnostics/clinical-products/rapid-diagnostics/gas troccult/index.htm. Accessed March 18, 2008.
1. U.S. Preventive Services Task Force. Screening for colorectal cancer: recommendation and rationale. Ann Intern Med. 2002;137:129-131. PubMed
2. Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143(3):844-857. PubMed
3. Carroll MRR, Seaman HE, Halloran HP. Tests and investigations for colorectal cancer screening. Clinical Biochemistry. 2014;47:921-939. PubMed
4. Tinmouth J, Lansdorp-Vogelaar I, Allison JE. Faecal immunochemical tests versus guaiac faecal occult blood tests: what clinicians and colorectal cancer screening programme organisers need to know. Gut. 2015;64(8):1327-1337. PubMed
5. Selinger RR, et al. Failure of health care professionals to interpret fecal occult blood tests accurately. Am J Med. 2003;114(1):64-67. PubMed
6. Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM. American College of Gastroenterology Guidelines for Colorectal Cancer Screening 2008. Am J Gastroenterol. 2009;104(3):739-750. PubMed
7. El-Tawil AM. Trends on gastrointestinal bleeding and mortality: Where are we standing? World J Gastroenterol. 2012;18(11):1154. PubMed
8. van Rijn AF, Stroobants AK, Deutekom M, et al. Inappropriate use of the faecal occult blood test in a university hospital in the Netherlands. Eur J Gastroenterol Hepatol. 2012;24(11):1266-1269. PubMed
9. Narula N, Ulic D, Al-Dabbagh R, et al. Fecal occult blood testing as a diagnostic test in symptomatic patients is not useful: a retrospective chart review. Can J Gastroenterol Hepatol. 2014;28(8):421-426. PubMed
10. Fleming, JL, Ahlquist DA, McGill DB, Zinsmeister AR, Ellefson RD, Schwartz S. Influence of aspirin and ethanol on fecal blood levels as determined by using the HemoQuant assay. Mayo Clin Proc. 1987;62(3):159-163. PubMed
11. Macrae FA, St John DJB. Relationship between patterns of bleeding and Hemoccult sensitivity in patients with colorectal cancers or adenomas. Gastroenterology. 1982;82:891-898. PubMed
12. Allard J, et al. Gastroscopy following a positive fecal occult blood test and negative colonoscopy: systematic review and guideline. Can J Gastroenterol. 2010;24(2):113-120. PubMed
13. Friedman A, Chan A, Chin LC, Deen A, Hammerschlag G, Lee M, et al. Use and abuse of faecal occult blood tests in an acute hospital inpatient setting. Intern Med J. 2010;40(2):107-111. PubMed
14. Allison JE, et al. Screening for colorectal neoplasms with new fecal occult blood tests: update on performance characteristics. J Natl Cancer Inst. 2007;99(19):1462-1470. PubMed
15. Chiang TH, Lee YC, Tu CH, Chiu HM, Wu MS. Performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract. CMAJ. 2011;183(13):1474-1481. PubMed
16. Bassett ML, Goulston KJ. False positive and negative hemoccult reactions on a normal diet and effect of diet restriction. Aust N Z J Med. 1980;10(1):1-4. PubMed
17. Jaffe, RM, Kasten B, Young DS, MacLowry JD. False-negative stool occult blood tests caused by ingestion of ascorbic acid (vitamin C). Ann Intern Med. 1975;83(6):824-826. PubMed
18. Ip S, Sokoro AAH, Kaita L, Ruiz C, McIntyre E, Singh H. Use of fecal occult blood testing in hospitalized patients: results of an audit. Can J Gastroenterol Hepatol. 2014;28(9):489-494. PubMed
19. Srygley FD, Gerardo CJ, Trun T, Fisher DA. Does this patient have a severe upper gastrointestinal bleed? JAMA. 2012;307(10):1072-1079. PubMed
20. Logue KA. Data Request - FOBT. June 2016. Regions Hospital, HealthPartners Laboratory, Saint Paul, Minnesota.
21. Population Clock. http://www.census.gov/popclock/. Accessed July 8, 2016.
22. Mosadeghi S, Ren H, Yen I, Bhuket T. Evaluation of fecal occult blood testing in the acute hospital setting. Gastrointestinal Endoscopy. 2015;81(5).
23. Gastroccult [package insert]. Beckman Coulter, Brea, CA. https://www.beckmancoulter.com/wsrportal/wsr/diagnostics/clinical-products/rapid-diagnostics/gas troccult/index.htm. Accessed March 18, 2008.
© 2017 Society of Hospital Medicine
TNFi treatment halves ankylosing spondylitis progression
MADRID – At least 2 years of tumor necrosis factor–inhibitor treatment of patients with ankylosing spondylitis nearly halved the rate of spinal radiographic progression in a study involving 432 Swiss patients.
In addition, patients on a tumor necrosis factor inhibitor (TNFi) who achieved low disease activity, reflected in an Ankylosing Spondylitis (AS) Disease Activity Score of 1.3 or less, showed virtually no spinal radiographic progression during a 2-year follow-up, Adrian Ciurea, MD, reported at the European Congress of Rheumatology.
He cautioned, however, that the evidence only shows correlation and can’t prove a causal relationship between TNFi treatment and slowed spinal radiographic progression because of potential residual confounding.
Dr. Ciurea and his associates analyzed records for AS patients enrolled in the Swiss Clinical Quality Management in Rheumatic Diseases cohort who underwent at least two spinal radiographs separated by a 2-year gap. They assessed the radiographs using the modified Stoke AS Spinal Score (mSASSS), and they defined progression as a gain of at least two units on the mSASSS during a 2-year period between radiographs.
The 432 AS patients in the study averaged 40 years old, two-thirds were men, and they had AS symptoms for an average of nearly 14 years. Their average AS Disease Activity Score (ASDAS) at entry was 2.8.
A multivariate analysis that controlled for several variables, including sex, smoking history, baseline mSASSS, and exercise, identified three parameters that had significant correlations with radiographic progression: Men had more than double the rate of progression, compared with women; higher baseline mSASSS was linked with a higher rate of progression; and a greater-than-2-year history of treatment with a TNFi was linked with a 48% reduced rate of progression, reported Dr. Ciurea, a rheumatologist at the Zürich University Hospital.
The duration of treatment also mattered. Patients who received at least 4 years of TNFi treatment had a statistically significant 68% reduced rate of radiographic spinal progression. In contrast, patients who received a TNFi for fewer than 4 years but more than 2 years had a 42% lower rate of progression that was of borderline statistical significance. TNFi treatment that started during the 2 years immediately preceding the radiograph failed to show a significant link with reduced progression.
Further analysis also showed a tight correlation between patients’ disease activity while on TNFi treatment and radiographic progression. Patients who maintained an average ASDAS of 2.1 or less during the 2 years prior to radiographic assessment showed an average mSASSS gain of 0.31 units over that 2-year period, compared with an average 1.45-unit mSASSS gain among patients whose average ASDAS remained above 2.1, a statistically significant difference between these two groups. Patients with even more inactive disease on TNFi treatment – those who maintained an average ASDAS of 1.3 or less – had an average 0.01-unit rise in their mSASSS after 2 years of treatment, compared with an average 0.52-unit mSASSS rise after 2 years in patients with an ASDAS of more than 1.3 but less than 2.1, he said.
The cohort study received partial support from Merck Sharpe & Dohme. Dr. Ciurea has been a consultant to or speaker for Abbvie, Celgene, Eli Lilly, Janssen-Cilag, Merck Sharp & Dohme, Novartis, Pfizer, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – At least 2 years of tumor necrosis factor–inhibitor treatment of patients with ankylosing spondylitis nearly halved the rate of spinal radiographic progression in a study involving 432 Swiss patients.
In addition, patients on a tumor necrosis factor inhibitor (TNFi) who achieved low disease activity, reflected in an Ankylosing Spondylitis (AS) Disease Activity Score of 1.3 or less, showed virtually no spinal radiographic progression during a 2-year follow-up, Adrian Ciurea, MD, reported at the European Congress of Rheumatology.
He cautioned, however, that the evidence only shows correlation and can’t prove a causal relationship between TNFi treatment and slowed spinal radiographic progression because of potential residual confounding.
Dr. Ciurea and his associates analyzed records for AS patients enrolled in the Swiss Clinical Quality Management in Rheumatic Diseases cohort who underwent at least two spinal radiographs separated by a 2-year gap. They assessed the radiographs using the modified Stoke AS Spinal Score (mSASSS), and they defined progression as a gain of at least two units on the mSASSS during a 2-year period between radiographs.
The 432 AS patients in the study averaged 40 years old, two-thirds were men, and they had AS symptoms for an average of nearly 14 years. Their average AS Disease Activity Score (ASDAS) at entry was 2.8.
A multivariate analysis that controlled for several variables, including sex, smoking history, baseline mSASSS, and exercise, identified three parameters that had significant correlations with radiographic progression: Men had more than double the rate of progression, compared with women; higher baseline mSASSS was linked with a higher rate of progression; and a greater-than-2-year history of treatment with a TNFi was linked with a 48% reduced rate of progression, reported Dr. Ciurea, a rheumatologist at the Zürich University Hospital.
The duration of treatment also mattered. Patients who received at least 4 years of TNFi treatment had a statistically significant 68% reduced rate of radiographic spinal progression. In contrast, patients who received a TNFi for fewer than 4 years but more than 2 years had a 42% lower rate of progression that was of borderline statistical significance. TNFi treatment that started during the 2 years immediately preceding the radiograph failed to show a significant link with reduced progression.
Further analysis also showed a tight correlation between patients’ disease activity while on TNFi treatment and radiographic progression. Patients who maintained an average ASDAS of 2.1 or less during the 2 years prior to radiographic assessment showed an average mSASSS gain of 0.31 units over that 2-year period, compared with an average 1.45-unit mSASSS gain among patients whose average ASDAS remained above 2.1, a statistically significant difference between these two groups. Patients with even more inactive disease on TNFi treatment – those who maintained an average ASDAS of 1.3 or less – had an average 0.01-unit rise in their mSASSS after 2 years of treatment, compared with an average 0.52-unit mSASSS rise after 2 years in patients with an ASDAS of more than 1.3 but less than 2.1, he said.
The cohort study received partial support from Merck Sharpe & Dohme. Dr. Ciurea has been a consultant to or speaker for Abbvie, Celgene, Eli Lilly, Janssen-Cilag, Merck Sharp & Dohme, Novartis, Pfizer, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – At least 2 years of tumor necrosis factor–inhibitor treatment of patients with ankylosing spondylitis nearly halved the rate of spinal radiographic progression in a study involving 432 Swiss patients.
In addition, patients on a tumor necrosis factor inhibitor (TNFi) who achieved low disease activity, reflected in an Ankylosing Spondylitis (AS) Disease Activity Score of 1.3 or less, showed virtually no spinal radiographic progression during a 2-year follow-up, Adrian Ciurea, MD, reported at the European Congress of Rheumatology.
He cautioned, however, that the evidence only shows correlation and can’t prove a causal relationship between TNFi treatment and slowed spinal radiographic progression because of potential residual confounding.
Dr. Ciurea and his associates analyzed records for AS patients enrolled in the Swiss Clinical Quality Management in Rheumatic Diseases cohort who underwent at least two spinal radiographs separated by a 2-year gap. They assessed the radiographs using the modified Stoke AS Spinal Score (mSASSS), and they defined progression as a gain of at least two units on the mSASSS during a 2-year period between radiographs.
The 432 AS patients in the study averaged 40 years old, two-thirds were men, and they had AS symptoms for an average of nearly 14 years. Their average AS Disease Activity Score (ASDAS) at entry was 2.8.
A multivariate analysis that controlled for several variables, including sex, smoking history, baseline mSASSS, and exercise, identified three parameters that had significant correlations with radiographic progression: Men had more than double the rate of progression, compared with women; higher baseline mSASSS was linked with a higher rate of progression; and a greater-than-2-year history of treatment with a TNFi was linked with a 48% reduced rate of progression, reported Dr. Ciurea, a rheumatologist at the Zürich University Hospital.
The duration of treatment also mattered. Patients who received at least 4 years of TNFi treatment had a statistically significant 68% reduced rate of radiographic spinal progression. In contrast, patients who received a TNFi for fewer than 4 years but more than 2 years had a 42% lower rate of progression that was of borderline statistical significance. TNFi treatment that started during the 2 years immediately preceding the radiograph failed to show a significant link with reduced progression.
Further analysis also showed a tight correlation between patients’ disease activity while on TNFi treatment and radiographic progression. Patients who maintained an average ASDAS of 2.1 or less during the 2 years prior to radiographic assessment showed an average mSASSS gain of 0.31 units over that 2-year period, compared with an average 1.45-unit mSASSS gain among patients whose average ASDAS remained above 2.1, a statistically significant difference between these two groups. Patients with even more inactive disease on TNFi treatment – those who maintained an average ASDAS of 1.3 or less – had an average 0.01-unit rise in their mSASSS after 2 years of treatment, compared with an average 0.52-unit mSASSS rise after 2 years in patients with an ASDAS of more than 1.3 but less than 2.1, he said.
The cohort study received partial support from Merck Sharpe & Dohme. Dr. Ciurea has been a consultant to or speaker for Abbvie, Celgene, Eli Lilly, Janssen-Cilag, Merck Sharp & Dohme, Novartis, Pfizer, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE EULAR 2017 CONGRESS
Key clinical point:
Major finding: Prolonged TNFi treatment was linked with a 48% lower rate of spinal radiographic progression, compared with shorter treatment.
Data source: Review of 432 patients in the Swiss Clinical Quality Management in Rheumatic Diseases cohort.
Disclosures: The cohort study received partial support from Merck Sharpe & Dohme. Dr. Ciurea has been a consultant to or speaker for Abbvie, Celgene, Eli Lilly, Janssen-Cilag, Merck Sharp & Dohme, Novartis, Pfizer, and UCB.
Pediatrics Committee’s role amplified with subspecialty’s evolution
Editor’s note: Each month, SHM puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. For more information on how you can lend your expertise to help SHM improve the care of hospitalized patients, log on to www.hospitalmedicine.org/getinvolved.
This month, The Hospitalist spotlights Sandra Gage, MD, PhD, SFHM, associate professor of pediatrics in the section of hospital medicine at the Medical College of Wisconsin, newly appointed chair of SHM’s Pediatrics Committee, and SHM member of almost 20 years.
Why did you choose a career in pediatric hospital medicine, and how did you become an SHM member?
I would say that pediatric hospital medicine chose me. After obtaining a degree in physical therapy and spending five years treating children with a variety of neurological and neurodevelopmental disorders, I went back to school to get my MD and a PhD in neurobiology, thinking that I would specialize in either pediatric neurology or pediatric physical medicine and rehabilitation.
I always had an interest in treating children but never considered general pediatrics because spending my time in the outpatient clinic setting had little appeal for me. This was before the concept of being a “hospitalist” was widespread – and even before the phrase was coined – but there were a few providers in my academic pediatric group who focused on inpatient care. The pace, variety and challenge of treating hospitalized children was exactly what I was looking for, and, following completion of my pediatric residency, I slowly became a full-time hospitalist.
What is the Pediatrics Committee currently working on, and what do you hope to accomplish during your term as Committee Chair?
With subspecialty status coming soon, rapidly expanding interest in the profession and the introduction of hospitalists into more areas of care, the landscape of pediatric hospital medicine is ever-changing. This amplifies the importance of the Pediatrics Committee’s role. The overall goals of the committee are to promote the growth and development of pediatric hospital medicine as a field and to provide educational and practical resources for individual practitioners.
The 2017-2018 committee comprises enthusiastic members from a wide variety of practice settings. At our first meeting in May, we formulated many exciting and innovative ideas to achieve our goals. As we continue to narrow down our approach and finalize our tasks for the year, we are also beginning to determine the content for the pediatric track at HM18. An example of a project the committee has executed in the past is the development of hospitalist-specific American Board of Pediatrics Maintenance of Certification modules for the SHM Learning Portal. In addition, the 2017 Pediatric Hospital Medicine (PHM) meeting is hosted by SHM this July in Nashville, and many Pediatrics Committee members are hard at work on finalizing those plans.
How has the PHM meeting evolved since its inception, and what value do you find in attending?
I have been an attendee of PHM many times over the years. The meeting has grown from a small group of no more than 100 individuals in a few hotel meeting rooms to more than 1,000 attendees and a wide variety of tracks and offerings. The growth of this meeting is truly reflective of the growth of our subspecialty, and the meeting brings together practitioners, both old and new, in an atmosphere full of innovations and ideas. Like SHM’s annual meeting, the PHM meeting is a great place for learning, sharing, and networking.
What advice do you have for fellow pediatric hospitalists during this transformational time in health care?
The direction of health care has provided fodder for lively discussion since I started my career 20 years ago. The nature of the practice of medicine is evolving, and, as physicians, we must be adept at navigating the changing climate while maintaining our goal of providing excellent care for our patients. As hospitalists, we have the opportunity to be in the forefront of the changes that will impact hospital care and utilization.
Whether our work is done at a local or a national level, as a group or as individuals, I believe that hospitalists will have an active role in directing the course of the future of medicine. We spend much of our clinical time advocating for our patients, but your experience is important and your voice can make an important contribution to the direction of health care for one child or for all children. Whether it is in the hospital hallway or on the Hill, continue to strive to do what you already do best.
Felicia Steele is SHM’s communications coordinator.
Editor’s note: Each month, SHM puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. For more information on how you can lend your expertise to help SHM improve the care of hospitalized patients, log on to www.hospitalmedicine.org/getinvolved.
This month, The Hospitalist spotlights Sandra Gage, MD, PhD, SFHM, associate professor of pediatrics in the section of hospital medicine at the Medical College of Wisconsin, newly appointed chair of SHM’s Pediatrics Committee, and SHM member of almost 20 years.
Why did you choose a career in pediatric hospital medicine, and how did you become an SHM member?
I would say that pediatric hospital medicine chose me. After obtaining a degree in physical therapy and spending five years treating children with a variety of neurological and neurodevelopmental disorders, I went back to school to get my MD and a PhD in neurobiology, thinking that I would specialize in either pediatric neurology or pediatric physical medicine and rehabilitation.
I always had an interest in treating children but never considered general pediatrics because spending my time in the outpatient clinic setting had little appeal for me. This was before the concept of being a “hospitalist” was widespread – and even before the phrase was coined – but there were a few providers in my academic pediatric group who focused on inpatient care. The pace, variety and challenge of treating hospitalized children was exactly what I was looking for, and, following completion of my pediatric residency, I slowly became a full-time hospitalist.
What is the Pediatrics Committee currently working on, and what do you hope to accomplish during your term as Committee Chair?
With subspecialty status coming soon, rapidly expanding interest in the profession and the introduction of hospitalists into more areas of care, the landscape of pediatric hospital medicine is ever-changing. This amplifies the importance of the Pediatrics Committee’s role. The overall goals of the committee are to promote the growth and development of pediatric hospital medicine as a field and to provide educational and practical resources for individual practitioners.
The 2017-2018 committee comprises enthusiastic members from a wide variety of practice settings. At our first meeting in May, we formulated many exciting and innovative ideas to achieve our goals. As we continue to narrow down our approach and finalize our tasks for the year, we are also beginning to determine the content for the pediatric track at HM18. An example of a project the committee has executed in the past is the development of hospitalist-specific American Board of Pediatrics Maintenance of Certification modules for the SHM Learning Portal. In addition, the 2017 Pediatric Hospital Medicine (PHM) meeting is hosted by SHM this July in Nashville, and many Pediatrics Committee members are hard at work on finalizing those plans.
How has the PHM meeting evolved since its inception, and what value do you find in attending?
I have been an attendee of PHM many times over the years. The meeting has grown from a small group of no more than 100 individuals in a few hotel meeting rooms to more than 1,000 attendees and a wide variety of tracks and offerings. The growth of this meeting is truly reflective of the growth of our subspecialty, and the meeting brings together practitioners, both old and new, in an atmosphere full of innovations and ideas. Like SHM’s annual meeting, the PHM meeting is a great place for learning, sharing, and networking.
What advice do you have for fellow pediatric hospitalists during this transformational time in health care?
The direction of health care has provided fodder for lively discussion since I started my career 20 years ago. The nature of the practice of medicine is evolving, and, as physicians, we must be adept at navigating the changing climate while maintaining our goal of providing excellent care for our patients. As hospitalists, we have the opportunity to be in the forefront of the changes that will impact hospital care and utilization.
Whether our work is done at a local or a national level, as a group or as individuals, I believe that hospitalists will have an active role in directing the course of the future of medicine. We spend much of our clinical time advocating for our patients, but your experience is important and your voice can make an important contribution to the direction of health care for one child or for all children. Whether it is in the hospital hallway or on the Hill, continue to strive to do what you already do best.
Felicia Steele is SHM’s communications coordinator.
Editor’s note: Each month, SHM puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. For more information on how you can lend your expertise to help SHM improve the care of hospitalized patients, log on to www.hospitalmedicine.org/getinvolved.
This month, The Hospitalist spotlights Sandra Gage, MD, PhD, SFHM, associate professor of pediatrics in the section of hospital medicine at the Medical College of Wisconsin, newly appointed chair of SHM’s Pediatrics Committee, and SHM member of almost 20 years.
Why did you choose a career in pediatric hospital medicine, and how did you become an SHM member?
I would say that pediatric hospital medicine chose me. After obtaining a degree in physical therapy and spending five years treating children with a variety of neurological and neurodevelopmental disorders, I went back to school to get my MD and a PhD in neurobiology, thinking that I would specialize in either pediatric neurology or pediatric physical medicine and rehabilitation.
I always had an interest in treating children but never considered general pediatrics because spending my time in the outpatient clinic setting had little appeal for me. This was before the concept of being a “hospitalist” was widespread – and even before the phrase was coined – but there were a few providers in my academic pediatric group who focused on inpatient care. The pace, variety and challenge of treating hospitalized children was exactly what I was looking for, and, following completion of my pediatric residency, I slowly became a full-time hospitalist.
What is the Pediatrics Committee currently working on, and what do you hope to accomplish during your term as Committee Chair?
With subspecialty status coming soon, rapidly expanding interest in the profession and the introduction of hospitalists into more areas of care, the landscape of pediatric hospital medicine is ever-changing. This amplifies the importance of the Pediatrics Committee’s role. The overall goals of the committee are to promote the growth and development of pediatric hospital medicine as a field and to provide educational and practical resources for individual practitioners.
The 2017-2018 committee comprises enthusiastic members from a wide variety of practice settings. At our first meeting in May, we formulated many exciting and innovative ideas to achieve our goals. As we continue to narrow down our approach and finalize our tasks for the year, we are also beginning to determine the content for the pediatric track at HM18. An example of a project the committee has executed in the past is the development of hospitalist-specific American Board of Pediatrics Maintenance of Certification modules for the SHM Learning Portal. In addition, the 2017 Pediatric Hospital Medicine (PHM) meeting is hosted by SHM this July in Nashville, and many Pediatrics Committee members are hard at work on finalizing those plans.
How has the PHM meeting evolved since its inception, and what value do you find in attending?
I have been an attendee of PHM many times over the years. The meeting has grown from a small group of no more than 100 individuals in a few hotel meeting rooms to more than 1,000 attendees and a wide variety of tracks and offerings. The growth of this meeting is truly reflective of the growth of our subspecialty, and the meeting brings together practitioners, both old and new, in an atmosphere full of innovations and ideas. Like SHM’s annual meeting, the PHM meeting is a great place for learning, sharing, and networking.
What advice do you have for fellow pediatric hospitalists during this transformational time in health care?
The direction of health care has provided fodder for lively discussion since I started my career 20 years ago. The nature of the practice of medicine is evolving, and, as physicians, we must be adept at navigating the changing climate while maintaining our goal of providing excellent care for our patients. As hospitalists, we have the opportunity to be in the forefront of the changes that will impact hospital care and utilization.
Whether our work is done at a local or a national level, as a group or as individuals, I believe that hospitalists will have an active role in directing the course of the future of medicine. We spend much of our clinical time advocating for our patients, but your experience is important and your voice can make an important contribution to the direction of health care for one child or for all children. Whether it is in the hospital hallway or on the Hill, continue to strive to do what you already do best.
Felicia Steele is SHM’s communications coordinator.
Nonpathologic Postdeployment Transition Symptoms in Combat National Guard Members and Reservists
The rigid dichotomy between combat deployment and postdeployment environments necessitates a multitude of cognitive, behavioral, and emotional adjustments for National Guard members and reservists to resume postdeployment civilian lifestyles successfully. Reacclimating to the postdeployment world is not a quick process for these veterans because of the time required to adjust from a deeply ingrained military combat mentality to civilian life. The process of this reintegration into the civilian world is known as postdeployment transition.
More than half of post-9/11 combat veterans report at least some difficulty with postdeployment transition.1,2 Frequently encountered symptoms of this period include impaired sleep, low frustration tolerance, decreased attention, poor concentration, short-term memory deficits, and difficulty with emotional regulation.1,3,4 Veterans will have difficulty reintegrating into the family unit and society without successful coping strategies to address these symptoms. If transition symptoms are prolonged, veterans are at risk for developing chronic adjustment difficulty or mental health issues.
Although there is significant attention paid to postdeployment adjustment by military family advocacy groups, there is little information in the medical literature on what constitutes common, nonpathologic postdeployment reactions among combat veterans. Frequently, when postdeployment transition symptoms are discussed, the medical literature tends to explain these in the context of a mental health disorder, such posttraumatic stress disorder (PTSD) or a cognitive injury, such as traumatic brain injury.5-8 Without a balanced understanding of normal postdeployment transitions, a health care provider (HCP) inappropriately may equate transition symptoms with the presence of mental health disorders or cognitive injury and medicalize the coping strategies needed to promote healthy adjustment.
The purpose of this article is to promote HCP awareness of common, nonpathologic postdeployment transition symptoms in combat veterans who are National Guard members or reservists. Such knowledge will enable HCPs to evaluate transition symptoms among these combat veterans reentering the civilian world, normalize common transition reactions, and recognize when further intervention is needed. This article reflects the author’s experience as a medical director working in a VA postdeployment clinic combined with data available in the medical literature and lay press.
Postdeployment Transition Symptoms
Dysregulation of emotional expression in returning combat veterans potentially can be present throughout the postdeployment period of adjustment. Although individual experiences vary widely in intensity and frequency, during postdeployment transition veterans often note difficulty in adjusting emotional expression to match that of nonmilitary counterparts.1,9-11 These difficulties usually fall into 2 broad categories: (1) relative emotional neutrality to major life events that cause nonmilitary civilians great joy or sadness; and (2) overreaction to trivial events, causing significant irritation, anger, or sadness that normally would not produce such emotional reactions in nonmilitary civilians. The former is largely overlooked in medical literature to date except in relation to the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) categories, and the latter is often described in limited terms as increased irritability, restlessness, and low frustration tolerance. This emotional dysregulation creates confusing paradoxes for veterans. For example, a veteran might feel no strong emotion when notified of the death of a close relative and yet cry uncontrollably while watching a sad scene in a fictional movie.
Sleep difficulties are intrinsic to the postdeployment period.9-12 Sleep-wake cycles often are altered, reflecting residual effects of the rigid schedules required by military duties and poor sleep hygiene in the combat theater. Inadequate, nonrestful sleep is frequently reported on return to the civilian world. Difficulty falling asleep or difficulty staying asleep also commonly occurs. Nightmares may be present.
Transient difficulty with concentration and attention is often prominent within the postdeployment transition period.9-11,13 Manifestations are variable, but problems with focusing on minor tasks are commonly reported. A more intense effort to master new concepts may be required. Learning styles developed during predeployment phases may be altered so that more conscious effort is required to comprehend and retain new information.
Short-term memory frequently may be affected during postdeployment transition.9-11,13 Veterans often report postdeployment difficulty in recalling appointments or tasks that must be completed even if they had a keen sense of memory during predeployment or deployment. Veterans also may have difficulty recalling the details of specific routines that were done without hesitation during deployment. Compared with predeployment time frames, veterans may exert greater effort to recall newly learned material.
Automatic behaviors necessary for survival in a combat theater still may be prominent in the postdeployment period.10,11,14 Aggressive driving required to avoid deployment ambush may be problematic during the postdeployment transition. Steering clear of any roadside trash may be a residual instinctive drive postdeployment because of the risk of improvised explosive devices concealed by debris in the combat theater. Veterans may avoid sitting with their back to the exit as the result of military safety training. Carrying weapons to ensure safety may be a compelling urge, because being armed and ready at all times was necessary for survival during deployment. Avoiding large crowds may be another strong tendency, because throngs of people were associated with potential danger in the combat theater.
Decision making may be challenging to resume in the postdeployment phase.9-11,15 In the deployment theater, time is relativel structured with rules in place, whereas at home veterans face a myriad of choices and decisions that must be made in order to complete the responsibilities of everyday living. As a result, making decisions about what item to buy, which clothes to wear, or what activities to prioritize, though relatively minor, can be a source of significant frustration. It may be difficult to independently navigate a realm of options available for new employment, schooling, or benefits, especially when there is little or no prior experience with these issues.
Relationship of Symptoms to Mental Health Diagnoses
Postdeployment transition symptoms do not automatically indicate the presence of an underlying mental health diagnosis. However, persistent and/or severe symptoms of postdeployment transition can overlap with or contribute to the development of mental health concerns (Table 1).14 The effects of the emotional disconnect also can exacerbate underlying mental health diagnoses.
While postdeployment emotional numbness to major life events, irritability, sleep disturbances, and impaired concentration can be associated with acute stress disorder (ASD) or PTSD, there is a constellation of other symptoms that must be present to diagnose these psychiatric conditions.16 Diagnostic criteria include persistent intrusive symptoms associated with the trauma, persistent avoidance of triggers/reminders associated with the trauma, significant changes in physiologic and cognitive arousal states, and negative changes in mood or cognition related to the trauma.16 The symptoms must cause significant impairment in some aspect of functioning on an individual, social, or occupational level. Acute stress disorder occurs when the symptoms last 30 days or less, whereas PTSD is diagnosed if the symptoms persist longer than a month.
Impaired emotional regulation, sleep disturbances, and decreased concentration also can be associated with depression or anxiety but are insufficient in themselves to make the diagnosis of those disorders.16 At least a 2-week history of depressed mood or inability to experience interest or pleasure in activities must be present as one of the criteria for depression as well as 4 or more other symptoms affecting sleep, appetite, energy, movement, self-esteem, or suicidal thoughts. Anxiety disorders have varying specific diagnostic criteria, but recurrent excessive worrying is a hallmark. Just like ASD or PTSD, the diagnostic symptoms of either depression or anxiety disorders must be causing significant impairment in functioning on an individual, social, or occupational level.
Irritability, sleep disturbances, agitation, memory impairment, and difficulty with concentration and attention can mimic the symptoms associated with mild-to-moderate traumatic brain injury (TBI).17,18 However, symptom onset must have a temporal relationship with a TBI. The presence of other TBI symptoms not associated with normal postdeployment transition usually can be used to differentiate between the diagnoses. Those TBI symptoms include recurrent headaches, poor balance, dizziness, tinnitus, and/or light sensitivity. In the majority of mild TBI cases, the symptoms resolve spontaneously within 3 months of TBI symptom manifestation.16,19 For those with persistent postconcussive syndrome, symptoms usually stabilize or improve over time.18,19 If symptoms worsen, there is often a confounding diagnosis such as PTSD or depression.17,20,21
Some returning combat veterans mistakenly believe postdeployment emotional transition symptoms are always a sign of a mental health disorder. Because there is a significant stigma associated with mental health disorders as well as potential repercussions on their service record if they use mental health resources, many reservists and National Guard members avoid accessing health care services if they are experiencing postdeployment adjustment issues, especially if those symptoms are related to emotional transitions.22-24 Unfortunately, such avoidance carries the risk that stress-inducing symptoms will persist and potentiate adjustment problems.
Course of Symptoms
The range for the postdeployment adjustment period generally falls within 3 to 12 months but can extend longer, depending on individual factors.10,11,25 Factors include presence of significant physical injury or illness, co-occurrence of mental health issues, underlying communication styles, and efficacy of coping strategies chosen. Although there is no clear-cut time frame for transition, ideally transition is complete when the returning veteran successfully enters his or her civilian lifestyle roles and feels a sense of purpose and belonging in society.
Postdeployment transition symptoms occur on a continuum in terms of duration and intensity for reservists and National Guard members. It is difficult to predict how specific transition symptoms will affect a particular veteran. The degree to which those symptoms will complicate reintegration depends on the individual veteran’s ability to adapt within the psychosocial context in which the symptoms occur. For example, minor irritation may be short-lived if a veteran can employ techniques to diffuse that feeling. Alternatively, minor irritation also suddenly may explode into a powerful wave of anger if the veteran has significant underlying emotional tension. Similarly, impaired short-term memory may be limited to forgetting a few appointments or may be so common that the veteran is at risk of losing track of his or her day. The level of memory impairment depends on emotional functioning, co-occurring stressors, and use of adaptive strategies.
In general, as these veterans successfully take on civilian routines, postdeployment transition symptoms will improve. Although such symptom improvement may be a passive process for some veterans, others will need to actively employ strategies to help change the military combat mind-set. The goal is to initiate useful interventions early in transition before symptoms become problematic.14
There are numerous self-help techniques and mobile apps that can be applied to a wide number of symptoms. Viable strategies include exercise, yoga, meditation, mindfulness training, and cognitive reframing. Reaching out for early assistance from various military assistance organizations that are well versed in dealing with postdeployment transition challenges often is helpful for reducing stress and navigating postdeployment obstacles (Table 2).
Symptom Strain and Exacerbation
Whenever stumbling blocks are encountered during the postdeployment period, any transition symptom can persist and/or worsen.10,11,14 Emotional disconnect and other transition symptoms can be exacerbated by physical, psychological, and social stressors common in the postdeployment period. Insomnia, poor quality sleep, or other sleep impairments that frequently occur as part of postdeployment transition can negatively impact the veteran’s ability to psychologically cope with daytime stressors. Poor concentration and short-term memory impairment noted by many reservists and National Guard members in the postdeployment phase can cause increased difficulty in attention to the moment and complicate completion of routine tasks. These difficulties can compound frustration and irritation to minor events and make it hard to emotionally connect with more serious issues.
Concentration and attention to mundane activities may be further reduced if the veteran feels no connection to the civilian world and/or experiences the surreal sensation that he or she should be attending to more serious life and death matters, such as those experienced in the combat theater. Ongoing psychological adjustment to physical injuries sustained during deployment can limit emotional flexibility when adapting to either minor or major stressors. Insufficient financial resources, work issues, or school problems can potentiate irritation, anger, and sadness and create an overwhelming emotional overload, leading to helplessness and hopelessness.
Perceived irregularities in emotional connection to the civilian world can significantly strain interpersonal relationships and be powerful impediments to successful reintegration.9,11,14 Failure to express emotions to major life events in the civilian world can result in combat veterans being viewed as not empathetic to others’ feelings. Overreaction to trivial events during postdeployment can lead to the veteran being labeled as unreasonable, controlling, and/or unpredictable. Persistent emotional disconnect with civilians engenders a growing sense of emotional isolation from family and friends when there is either incorrect interpretation of emotional transitions or failure to adapt healthy coping strategies. This isolation further enlarges the emotional chasm and may greatly diminish the veteran’s ability to seek assistance and appropriately address stressors in the civilian world.
Transition and the Family
Emotional disconnection may be more acutely felt within the immediate family unit.26 Redistribution of family unit responsibilities during deployment may mean that roles the veteran played during predeployment now may be handled by a partner. On the veteran’s return to the civilian world, such circumstances require active renegotiation of duties. Interactions with loved ones, especially children, may be colored by the family members’ individual perspectives on deployment as well as by the veteran’s transition symptoms. When there is disagreement about role responsibilities and/or underlying family resentment about deployment, conditions are ripe for significant discord between the veteran and family members, vital loss of partner intimacy, and notable loss of psychological safety to express feelings within the family unit. If there are concerns about infidelity by the veteran or significant other during the period of deployment, postdeployment tensions can further escalate. If unaddressed in the presence of emotional disconnect, any of these situations can raise the risk of domestic violence and destruction of relationships.
Without adequate knowledge of common postdeployment transitions and coping strategies, the postdeployment transition period is often bewildering to returning veterans and their families. They are taken aback by postdeployment behaviors that do not conform to the veteran’s predeployment personality or mannerisms. Families may feel they have “lost” the veteran and view the emotionally distant postdeployment veteran as a stranger. Veterans mistakenly may view the postdeployment emotional disconnect as evidence that they were permanently altered by deployment and no longer can assimilate into the civilian world. Unless veterans and families develop an awareness of the postdeployment transition symptoms and healthy coping strategies, these perspectives can contribute to a veteran’s persistent feelings of alienation, significant sense of personal failure, and loss of vital social supports.
When transition symptoms are or have the potential to become significant stressors, veterans would benefit from mental health counseling either individually or with family members. Overcoming the stigma of seeking mental health services can prove challenging. Explaining that these postdeployment symptoms occur commonly, stem from military combat training, can be reversed, and when reversed will empower the individual to control his or her life may help veterans overcome the stigma and seek mental health services.
The fear of future career impairment with the military reserve or National Guard is another real concern among this cohort who might consider accessing behavioral health care, especially since VA mental health medical records can be accessed by DoD officials through links with the VHA. Fortunately, this concern can be alleviated through the use of Vet Centers, free-standing counseling centers nationwide that offer no-cost individual and family counseling to veterans with combat exposure. Vet Center counseling records are completely confidential, never shared, and are not linked to the VHA electronic health record, the DoD, or any other entity. Although Vet Center providers don’t prescribe medications, the counselors can actively address many issues for veterans and their families. For individuals who do not live near a Vet Center or for those who require psychiatric medications, a frank discussion on the benefits of treatment vs the risk of treatment avoidance must be held.
Assessing Symptoms and Coping Mechanisms
Postdeployment transition symptoms vary, depending on the nature and context of the symptom. Not only must the returning reservist and National Guard member be screened for symptoms, but HCPs also should assess the impact of those symptoms on the veteran and his or her interpersonal relationships. Some veterans will feel that the symptoms have relatively minor impact in their lives, because the veteran can easily compensate for the transient effects. Others may feel that the symptoms are somewhat burdensome because the issues are complicating the smooth transition to civilian roles. Still others will judge the symptoms to be devastating because of the negative effects on personal control, selfesteem, and emotional connection with family and friends.
In addition to screening for symptoms, HCPs should assess these veterans’ current coping adaptations to various transition symptoms. Whereas some activities may be functional and promote reintegration, other short-term coping solutions may cripple the veteran’s ability to successfully resume civilian life. Global avoidance of communication with others and/or retreating from all social situations is a destructive coping pattern that can further alienate veterans from their families and the civilian world. Reacting with anger to all stressful issues is another maladaptive pattern of coping with life’s frustrations. Because of the potential to self-medicate when dealing with social difficulties, depression, anxiety, or other mental health diagnoses, veterans may develop an inappropriate reliance on drugs or alcohol to handle postdeployment stressors.27 Therefore, HCP screening for substance use disorders (SUD) is important so that interventions can be initiated early.
Because of the overlap of postdeployment transition symptoms with mental health disorders and the relative frequency of those mental health disorders among combat veterans, HCPs should have a heightened awareness of the potential for co-occurring mental health difficulties in the postdeployment reservist and National Guard cohort. Health care providers should screen for depression, anxiety, and PTSD. Even if initial screening is done early within the transition period, repeat screening would be of benefit 6 months into the postdeployment period because of the tendency of mental health issues to develop during that time.28,29
By evaluating the impact of the transition symptom and coping strategies on these veterans’ lives, HCPs can better determine which strategies might adequately compensate for symptom effects. In general, informal counseling, even if just to help veterans normalize postdeployment transition symptoms and develop a plan to address such symptoms, can significantly minimize the negative impact of transition symptoms.14,26 Specific symptoms should be targeted by interventions that match the degree of symptom impact.
Symptoms to be aggressively addressed are those that significantly interfere with successful reintegration into the civilian world. For example, persistent sleep difficulties should be dealt with because they can worsen all other transition symptoms. However, the majority of strategies to address sleep do not require medication unless there are confounding factors such as severe nightmares. Minor memory issues attributed to the transition phase can be mitigated by several strategies to improve recall, including use of task lists, digital calendars, or other memory-prodding techniques. However, severe memory issues related to depression or anxiety likely would require pharmaceutical assistance and formal counseling in addition to other nonpharmacologic approaches.
Intermittent irritation or restlessness may be amenable to selfhelp strategies, but significant anger outbursts or aggression will require additional support, such as formal behavioral interventions to help identify the triggers and develop strategic plans to reduce emotional tension. A mild sense of not belonging may resolve without intervention, but a stronger sense of alienation will require further evaluation.
Conclusion
Civilian reintegration after combat deployment is a gradual process rather than a discrete event for reservists and National Guard members. There are common, nonpathologic postdeployment transition symptoms that, if misunderstood or inappropriately addressed, can complicate civilian reintegration. Health care providers are in the unique position to promote a healthy postdeployment transition by assisting veterans to recognize nonpathologic transition symptoms, select appropriate coping strategies, and seek further assistance for more complex problems.
1. Pew Research Center. War and sacrifice in the post 9/11 era: executive summary. http://www
.pewsocialtrends.org/2011/10/05/war-and-sacrifice-in-the-post-911-era. Published October 5, 2011. Accessed June 12, 2017.
2. Interian A, Kline A, Callahan L, Losonczy M. Readjustment stressors and early mental health treatment seeking by returning National Guard soldiers with PTSD. Psychiatr Serv. 2012;63(9):855-861.
3. Spelman JF, Hunt SC, Seal KH, Burgo-Black AL. Post deployment care for returning combat veterans. J Gen Intern Med. 2012;27(9):1200-1209.
4. Vasterling JJ, Daily ES, Friedman MJ. Posttraumatic stress reactions over time: the battlefield, homecoming, and long-term course. In: Ruzek JI, Schnurr PP, Vasterling JJ, Friedman MJ, eds. Caring for Veterans With Deployment-Related Stress Disorders: Iraq, Afghanistan, and Beyond. Washington,DC: American Psychological Association;2011:chap 2.
5. Wilcox SL, Oh H, Redmon SA, Chicas J, Hassan AM, Lee PJ, Ell K. A scope of the problem: Postdeployment reintegration challenges in a National Guard Unit. Work. 2015;50(1):73-83.
6. Griffith J. Homecoming of citizen soldiers: Postdeployment problems and service use among Army National Guard soldiers. Community Ment Health J. 2017. doi:10.1007/s10597-017-0132-9. (Epub ahead of print)
7. Schultz M, Glickman ME, Eisen SV. Predictors of decline in overall mental health, PTSD and alcohol use in OEF/OIF veterans. Comprehensive Psychiatry. 2014;55(7):1654-1664.
8. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in National Guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011:68(1):79-89.
9. U.S. Department of Veterans Affairs, National Center for PTSD. Returning from the war zone: a guide for military personnel. http://www.ptsd.va.gov/public/reintegration/guide-pdf/SMGuide.pdf. Updated January 2014. Accessed June 12, 2017.
10. Slone LB, Friedman MJ. After the War Zone: A Practical Guide for Returning Troops and their Families. Philadelphia, PA: Da Capo Press; 2008.
11. Ainspan ND, Penk WE, eds. When the Warrior Returns: Making the Transition at Home. Annapolis, MD: Naval Institute Press; 2012.
12. Yosick T, Bates M, Moore M, Crowe C, Phillips J, Davison J. A review of post-deployment reintegration: evidence, challenges, and strategies for program development. http://www.dcoe.mil/files/Review_of_Post-Deployment_Reintegration.pdf. Published February 10, 2012. Accessed June 12, 2017.
13. Vasterling JJ, Proctor SP, Amoroso P, Kane R, Heeren T, White RF. Neuropsychological outcomes of army personnel following deployment to the Iraq war. JAMA. 2006;296(5):519-529.
14. Castro CA, Kintzle S, Hassan AM. The combat veteran paradox: paradoxes and dilemmas encountered with reintegrating combat veterans and the agencies that support them. Traumatology. 2015;21(4):299-310.
15. Rivers FM, Gordon S, Speraw S, Reese S. U.S. Army nurses’ reintegration and homecoming experiences after Iraq and Afghanistan. Mil Med. 2013;178(2):166-173.
16. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington,VA: American Psychiatric Association;2013.
17. Tanielian T, Jaycox LH, eds. Invisible Wounds of War: Psychological and Cognitive Injuries, Their Consequences, and Services to Assist Recovery. Santa Monica, CA: Rand Corporation, 2008.
18. Orff HJ, Hays CC, Heldreth AA, Stein MB, Twamley EW. Clinical considerations in the evaluation and management of patients following traumatic brain injury. Focus. 2013;11(3):328-340.
19. Morissette SB, Woodward M, Kimbrel NA, et al. Deployment-related TBI, persistent postconcussive symptoms, PTSD, and depression in OEF/OIF veterans. Rehabil Psychol. 2011;56(4):340-350.
20. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in national guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011;68(1):79-89.
21. Wilk JE, Herrell RK, Wynn GH, Riviere LA, Hoge CW. Mild traumatic brain injury (concussion), posttraumatic stress disorder, and depression in U.S. soldiers involved in combat deployments: association with postdeployment symptoms. Psychosom Med. 2012;74(3):249-257.
22. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
23. Hines LA, Goodwin L, Jones M, et al. Factors affecting help seeking for mental health problems after deployment to Iraq and Afghanistan. Psychiatr Serv. 2014;65(1):98-105.
24. Gorman LA, Blow AJ, Ames BD, Read PL. National Guard families after combat: mental health, use of mental health services, and perceived treatment barriers. Psychiatr Serv. 2011;62(1):28-34.
25. Marek LI, Hollingsworth WG, D’Aniello C, et al. Returning home: what we know about the reintegration of deployed service members into their families and communities. https://www.ncfr.org/ncfr-report/focus/military-families/returninghome. Published March 1, 2012. Accessed June 13, 2017.
26. Bowling UB, Sherman MD. Welcoming them home: supporting service members and their families in navigating the tasks of reintegration. Prof Psychol Res Pr. 2008;39(4):451-458.
27. Jacobson IG, Ryan MA, Hooper TI, et al. Alcohol use and alcohol-related problems before
and after military combat deployment. JAMA. 2008;300(6):663-675.
28. Seal KH, Metzler TH, Gima KS, Bertenthal D, Maguen S, Marmar CR. Trends and risk factors for mental health diagnoses among Iraq and Afghanistan veterans Department of Veterans Affairs health care, 2002-2008. Am J Public Health. 2009;99(9):1651-1658.
29. Milliken CS, Auchterlonie JL, Hoge CW. Longitudinal assessment of mental health problems among active and reserve component soldiers returning from the Iraq war. JAMA. 2007;298(18):2141-2148.
The rigid dichotomy between combat deployment and postdeployment environments necessitates a multitude of cognitive, behavioral, and emotional adjustments for National Guard members and reservists to resume postdeployment civilian lifestyles successfully. Reacclimating to the postdeployment world is not a quick process for these veterans because of the time required to adjust from a deeply ingrained military combat mentality to civilian life. The process of this reintegration into the civilian world is known as postdeployment transition.
More than half of post-9/11 combat veterans report at least some difficulty with postdeployment transition.1,2 Frequently encountered symptoms of this period include impaired sleep, low frustration tolerance, decreased attention, poor concentration, short-term memory deficits, and difficulty with emotional regulation.1,3,4 Veterans will have difficulty reintegrating into the family unit and society without successful coping strategies to address these symptoms. If transition symptoms are prolonged, veterans are at risk for developing chronic adjustment difficulty or mental health issues.
Although there is significant attention paid to postdeployment adjustment by military family advocacy groups, there is little information in the medical literature on what constitutes common, nonpathologic postdeployment reactions among combat veterans. Frequently, when postdeployment transition symptoms are discussed, the medical literature tends to explain these in the context of a mental health disorder, such posttraumatic stress disorder (PTSD) or a cognitive injury, such as traumatic brain injury.5-8 Without a balanced understanding of normal postdeployment transitions, a health care provider (HCP) inappropriately may equate transition symptoms with the presence of mental health disorders or cognitive injury and medicalize the coping strategies needed to promote healthy adjustment.
The purpose of this article is to promote HCP awareness of common, nonpathologic postdeployment transition symptoms in combat veterans who are National Guard members or reservists. Such knowledge will enable HCPs to evaluate transition symptoms among these combat veterans reentering the civilian world, normalize common transition reactions, and recognize when further intervention is needed. This article reflects the author’s experience as a medical director working in a VA postdeployment clinic combined with data available in the medical literature and lay press.
Postdeployment Transition Symptoms
Dysregulation of emotional expression in returning combat veterans potentially can be present throughout the postdeployment period of adjustment. Although individual experiences vary widely in intensity and frequency, during postdeployment transition veterans often note difficulty in adjusting emotional expression to match that of nonmilitary counterparts.1,9-11 These difficulties usually fall into 2 broad categories: (1) relative emotional neutrality to major life events that cause nonmilitary civilians great joy or sadness; and (2) overreaction to trivial events, causing significant irritation, anger, or sadness that normally would not produce such emotional reactions in nonmilitary civilians. The former is largely overlooked in medical literature to date except in relation to the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) categories, and the latter is often described in limited terms as increased irritability, restlessness, and low frustration tolerance. This emotional dysregulation creates confusing paradoxes for veterans. For example, a veteran might feel no strong emotion when notified of the death of a close relative and yet cry uncontrollably while watching a sad scene in a fictional movie.
Sleep difficulties are intrinsic to the postdeployment period.9-12 Sleep-wake cycles often are altered, reflecting residual effects of the rigid schedules required by military duties and poor sleep hygiene in the combat theater. Inadequate, nonrestful sleep is frequently reported on return to the civilian world. Difficulty falling asleep or difficulty staying asleep also commonly occurs. Nightmares may be present.
Transient difficulty with concentration and attention is often prominent within the postdeployment transition period.9-11,13 Manifestations are variable, but problems with focusing on minor tasks are commonly reported. A more intense effort to master new concepts may be required. Learning styles developed during predeployment phases may be altered so that more conscious effort is required to comprehend and retain new information.
Short-term memory frequently may be affected during postdeployment transition.9-11,13 Veterans often report postdeployment difficulty in recalling appointments or tasks that must be completed even if they had a keen sense of memory during predeployment or deployment. Veterans also may have difficulty recalling the details of specific routines that were done without hesitation during deployment. Compared with predeployment time frames, veterans may exert greater effort to recall newly learned material.
Automatic behaviors necessary for survival in a combat theater still may be prominent in the postdeployment period.10,11,14 Aggressive driving required to avoid deployment ambush may be problematic during the postdeployment transition. Steering clear of any roadside trash may be a residual instinctive drive postdeployment because of the risk of improvised explosive devices concealed by debris in the combat theater. Veterans may avoid sitting with their back to the exit as the result of military safety training. Carrying weapons to ensure safety may be a compelling urge, because being armed and ready at all times was necessary for survival during deployment. Avoiding large crowds may be another strong tendency, because throngs of people were associated with potential danger in the combat theater.
Decision making may be challenging to resume in the postdeployment phase.9-11,15 In the deployment theater, time is relativel structured with rules in place, whereas at home veterans face a myriad of choices and decisions that must be made in order to complete the responsibilities of everyday living. As a result, making decisions about what item to buy, which clothes to wear, or what activities to prioritize, though relatively minor, can be a source of significant frustration. It may be difficult to independently navigate a realm of options available for new employment, schooling, or benefits, especially when there is little or no prior experience with these issues.
Relationship of Symptoms to Mental Health Diagnoses
Postdeployment transition symptoms do not automatically indicate the presence of an underlying mental health diagnosis. However, persistent and/or severe symptoms of postdeployment transition can overlap with or contribute to the development of mental health concerns (Table 1).14 The effects of the emotional disconnect also can exacerbate underlying mental health diagnoses.
While postdeployment emotional numbness to major life events, irritability, sleep disturbances, and impaired concentration can be associated with acute stress disorder (ASD) or PTSD, there is a constellation of other symptoms that must be present to diagnose these psychiatric conditions.16 Diagnostic criteria include persistent intrusive symptoms associated with the trauma, persistent avoidance of triggers/reminders associated with the trauma, significant changes in physiologic and cognitive arousal states, and negative changes in mood or cognition related to the trauma.16 The symptoms must cause significant impairment in some aspect of functioning on an individual, social, or occupational level. Acute stress disorder occurs when the symptoms last 30 days or less, whereas PTSD is diagnosed if the symptoms persist longer than a month.
Impaired emotional regulation, sleep disturbances, and decreased concentration also can be associated with depression or anxiety but are insufficient in themselves to make the diagnosis of those disorders.16 At least a 2-week history of depressed mood or inability to experience interest or pleasure in activities must be present as one of the criteria for depression as well as 4 or more other symptoms affecting sleep, appetite, energy, movement, self-esteem, or suicidal thoughts. Anxiety disorders have varying specific diagnostic criteria, but recurrent excessive worrying is a hallmark. Just like ASD or PTSD, the diagnostic symptoms of either depression or anxiety disorders must be causing significant impairment in functioning on an individual, social, or occupational level.
Irritability, sleep disturbances, agitation, memory impairment, and difficulty with concentration and attention can mimic the symptoms associated with mild-to-moderate traumatic brain injury (TBI).17,18 However, symptom onset must have a temporal relationship with a TBI. The presence of other TBI symptoms not associated with normal postdeployment transition usually can be used to differentiate between the diagnoses. Those TBI symptoms include recurrent headaches, poor balance, dizziness, tinnitus, and/or light sensitivity. In the majority of mild TBI cases, the symptoms resolve spontaneously within 3 months of TBI symptom manifestation.16,19 For those with persistent postconcussive syndrome, symptoms usually stabilize or improve over time.18,19 If symptoms worsen, there is often a confounding diagnosis such as PTSD or depression.17,20,21
Some returning combat veterans mistakenly believe postdeployment emotional transition symptoms are always a sign of a mental health disorder. Because there is a significant stigma associated with mental health disorders as well as potential repercussions on their service record if they use mental health resources, many reservists and National Guard members avoid accessing health care services if they are experiencing postdeployment adjustment issues, especially if those symptoms are related to emotional transitions.22-24 Unfortunately, such avoidance carries the risk that stress-inducing symptoms will persist and potentiate adjustment problems.
Course of Symptoms
The range for the postdeployment adjustment period generally falls within 3 to 12 months but can extend longer, depending on individual factors.10,11,25 Factors include presence of significant physical injury or illness, co-occurrence of mental health issues, underlying communication styles, and efficacy of coping strategies chosen. Although there is no clear-cut time frame for transition, ideally transition is complete when the returning veteran successfully enters his or her civilian lifestyle roles and feels a sense of purpose and belonging in society.
Postdeployment transition symptoms occur on a continuum in terms of duration and intensity for reservists and National Guard members. It is difficult to predict how specific transition symptoms will affect a particular veteran. The degree to which those symptoms will complicate reintegration depends on the individual veteran’s ability to adapt within the psychosocial context in which the symptoms occur. For example, minor irritation may be short-lived if a veteran can employ techniques to diffuse that feeling. Alternatively, minor irritation also suddenly may explode into a powerful wave of anger if the veteran has significant underlying emotional tension. Similarly, impaired short-term memory may be limited to forgetting a few appointments or may be so common that the veteran is at risk of losing track of his or her day. The level of memory impairment depends on emotional functioning, co-occurring stressors, and use of adaptive strategies.
In general, as these veterans successfully take on civilian routines, postdeployment transition symptoms will improve. Although such symptom improvement may be a passive process for some veterans, others will need to actively employ strategies to help change the military combat mind-set. The goal is to initiate useful interventions early in transition before symptoms become problematic.14
There are numerous self-help techniques and mobile apps that can be applied to a wide number of symptoms. Viable strategies include exercise, yoga, meditation, mindfulness training, and cognitive reframing. Reaching out for early assistance from various military assistance organizations that are well versed in dealing with postdeployment transition challenges often is helpful for reducing stress and navigating postdeployment obstacles (Table 2).
Symptom Strain and Exacerbation
Whenever stumbling blocks are encountered during the postdeployment period, any transition symptom can persist and/or worsen.10,11,14 Emotional disconnect and other transition symptoms can be exacerbated by physical, psychological, and social stressors common in the postdeployment period. Insomnia, poor quality sleep, or other sleep impairments that frequently occur as part of postdeployment transition can negatively impact the veteran’s ability to psychologically cope with daytime stressors. Poor concentration and short-term memory impairment noted by many reservists and National Guard members in the postdeployment phase can cause increased difficulty in attention to the moment and complicate completion of routine tasks. These difficulties can compound frustration and irritation to minor events and make it hard to emotionally connect with more serious issues.
Concentration and attention to mundane activities may be further reduced if the veteran feels no connection to the civilian world and/or experiences the surreal sensation that he or she should be attending to more serious life and death matters, such as those experienced in the combat theater. Ongoing psychological adjustment to physical injuries sustained during deployment can limit emotional flexibility when adapting to either minor or major stressors. Insufficient financial resources, work issues, or school problems can potentiate irritation, anger, and sadness and create an overwhelming emotional overload, leading to helplessness and hopelessness.
Perceived irregularities in emotional connection to the civilian world can significantly strain interpersonal relationships and be powerful impediments to successful reintegration.9,11,14 Failure to express emotions to major life events in the civilian world can result in combat veterans being viewed as not empathetic to others’ feelings. Overreaction to trivial events during postdeployment can lead to the veteran being labeled as unreasonable, controlling, and/or unpredictable. Persistent emotional disconnect with civilians engenders a growing sense of emotional isolation from family and friends when there is either incorrect interpretation of emotional transitions or failure to adapt healthy coping strategies. This isolation further enlarges the emotional chasm and may greatly diminish the veteran’s ability to seek assistance and appropriately address stressors in the civilian world.
Transition and the Family
Emotional disconnection may be more acutely felt within the immediate family unit.26 Redistribution of family unit responsibilities during deployment may mean that roles the veteran played during predeployment now may be handled by a partner. On the veteran’s return to the civilian world, such circumstances require active renegotiation of duties. Interactions with loved ones, especially children, may be colored by the family members’ individual perspectives on deployment as well as by the veteran’s transition symptoms. When there is disagreement about role responsibilities and/or underlying family resentment about deployment, conditions are ripe for significant discord between the veteran and family members, vital loss of partner intimacy, and notable loss of psychological safety to express feelings within the family unit. If there are concerns about infidelity by the veteran or significant other during the period of deployment, postdeployment tensions can further escalate. If unaddressed in the presence of emotional disconnect, any of these situations can raise the risk of domestic violence and destruction of relationships.
Without adequate knowledge of common postdeployment transitions and coping strategies, the postdeployment transition period is often bewildering to returning veterans and their families. They are taken aback by postdeployment behaviors that do not conform to the veteran’s predeployment personality or mannerisms. Families may feel they have “lost” the veteran and view the emotionally distant postdeployment veteran as a stranger. Veterans mistakenly may view the postdeployment emotional disconnect as evidence that they were permanently altered by deployment and no longer can assimilate into the civilian world. Unless veterans and families develop an awareness of the postdeployment transition symptoms and healthy coping strategies, these perspectives can contribute to a veteran’s persistent feelings of alienation, significant sense of personal failure, and loss of vital social supports.
When transition symptoms are or have the potential to become significant stressors, veterans would benefit from mental health counseling either individually or with family members. Overcoming the stigma of seeking mental health services can prove challenging. Explaining that these postdeployment symptoms occur commonly, stem from military combat training, can be reversed, and when reversed will empower the individual to control his or her life may help veterans overcome the stigma and seek mental health services.
The fear of future career impairment with the military reserve or National Guard is another real concern among this cohort who might consider accessing behavioral health care, especially since VA mental health medical records can be accessed by DoD officials through links with the VHA. Fortunately, this concern can be alleviated through the use of Vet Centers, free-standing counseling centers nationwide that offer no-cost individual and family counseling to veterans with combat exposure. Vet Center counseling records are completely confidential, never shared, and are not linked to the VHA electronic health record, the DoD, or any other entity. Although Vet Center providers don’t prescribe medications, the counselors can actively address many issues for veterans and their families. For individuals who do not live near a Vet Center or for those who require psychiatric medications, a frank discussion on the benefits of treatment vs the risk of treatment avoidance must be held.
Assessing Symptoms and Coping Mechanisms
Postdeployment transition symptoms vary, depending on the nature and context of the symptom. Not only must the returning reservist and National Guard member be screened for symptoms, but HCPs also should assess the impact of those symptoms on the veteran and his or her interpersonal relationships. Some veterans will feel that the symptoms have relatively minor impact in their lives, because the veteran can easily compensate for the transient effects. Others may feel that the symptoms are somewhat burdensome because the issues are complicating the smooth transition to civilian roles. Still others will judge the symptoms to be devastating because of the negative effects on personal control, selfesteem, and emotional connection with family and friends.
In addition to screening for symptoms, HCPs should assess these veterans’ current coping adaptations to various transition symptoms. Whereas some activities may be functional and promote reintegration, other short-term coping solutions may cripple the veteran’s ability to successfully resume civilian life. Global avoidance of communication with others and/or retreating from all social situations is a destructive coping pattern that can further alienate veterans from their families and the civilian world. Reacting with anger to all stressful issues is another maladaptive pattern of coping with life’s frustrations. Because of the potential to self-medicate when dealing with social difficulties, depression, anxiety, or other mental health diagnoses, veterans may develop an inappropriate reliance on drugs or alcohol to handle postdeployment stressors.27 Therefore, HCP screening for substance use disorders (SUD) is important so that interventions can be initiated early.
Because of the overlap of postdeployment transition symptoms with mental health disorders and the relative frequency of those mental health disorders among combat veterans, HCPs should have a heightened awareness of the potential for co-occurring mental health difficulties in the postdeployment reservist and National Guard cohort. Health care providers should screen for depression, anxiety, and PTSD. Even if initial screening is done early within the transition period, repeat screening would be of benefit 6 months into the postdeployment period because of the tendency of mental health issues to develop during that time.28,29
By evaluating the impact of the transition symptom and coping strategies on these veterans’ lives, HCPs can better determine which strategies might adequately compensate for symptom effects. In general, informal counseling, even if just to help veterans normalize postdeployment transition symptoms and develop a plan to address such symptoms, can significantly minimize the negative impact of transition symptoms.14,26 Specific symptoms should be targeted by interventions that match the degree of symptom impact.
Symptoms to be aggressively addressed are those that significantly interfere with successful reintegration into the civilian world. For example, persistent sleep difficulties should be dealt with because they can worsen all other transition symptoms. However, the majority of strategies to address sleep do not require medication unless there are confounding factors such as severe nightmares. Minor memory issues attributed to the transition phase can be mitigated by several strategies to improve recall, including use of task lists, digital calendars, or other memory-prodding techniques. However, severe memory issues related to depression or anxiety likely would require pharmaceutical assistance and formal counseling in addition to other nonpharmacologic approaches.
Intermittent irritation or restlessness may be amenable to selfhelp strategies, but significant anger outbursts or aggression will require additional support, such as formal behavioral interventions to help identify the triggers and develop strategic plans to reduce emotional tension. A mild sense of not belonging may resolve without intervention, but a stronger sense of alienation will require further evaluation.
Conclusion
Civilian reintegration after combat deployment is a gradual process rather than a discrete event for reservists and National Guard members. There are common, nonpathologic postdeployment transition symptoms that, if misunderstood or inappropriately addressed, can complicate civilian reintegration. Health care providers are in the unique position to promote a healthy postdeployment transition by assisting veterans to recognize nonpathologic transition symptoms, select appropriate coping strategies, and seek further assistance for more complex problems.
The rigid dichotomy between combat deployment and postdeployment environments necessitates a multitude of cognitive, behavioral, and emotional adjustments for National Guard members and reservists to resume postdeployment civilian lifestyles successfully. Reacclimating to the postdeployment world is not a quick process for these veterans because of the time required to adjust from a deeply ingrained military combat mentality to civilian life. The process of this reintegration into the civilian world is known as postdeployment transition.
More than half of post-9/11 combat veterans report at least some difficulty with postdeployment transition.1,2 Frequently encountered symptoms of this period include impaired sleep, low frustration tolerance, decreased attention, poor concentration, short-term memory deficits, and difficulty with emotional regulation.1,3,4 Veterans will have difficulty reintegrating into the family unit and society without successful coping strategies to address these symptoms. If transition symptoms are prolonged, veterans are at risk for developing chronic adjustment difficulty or mental health issues.
Although there is significant attention paid to postdeployment adjustment by military family advocacy groups, there is little information in the medical literature on what constitutes common, nonpathologic postdeployment reactions among combat veterans. Frequently, when postdeployment transition symptoms are discussed, the medical literature tends to explain these in the context of a mental health disorder, such posttraumatic stress disorder (PTSD) or a cognitive injury, such as traumatic brain injury.5-8 Without a balanced understanding of normal postdeployment transitions, a health care provider (HCP) inappropriately may equate transition symptoms with the presence of mental health disorders or cognitive injury and medicalize the coping strategies needed to promote healthy adjustment.
The purpose of this article is to promote HCP awareness of common, nonpathologic postdeployment transition symptoms in combat veterans who are National Guard members or reservists. Such knowledge will enable HCPs to evaluate transition symptoms among these combat veterans reentering the civilian world, normalize common transition reactions, and recognize when further intervention is needed. This article reflects the author’s experience as a medical director working in a VA postdeployment clinic combined with data available in the medical literature and lay press.
Postdeployment Transition Symptoms
Dysregulation of emotional expression in returning combat veterans potentially can be present throughout the postdeployment period of adjustment. Although individual experiences vary widely in intensity and frequency, during postdeployment transition veterans often note difficulty in adjusting emotional expression to match that of nonmilitary counterparts.1,9-11 These difficulties usually fall into 2 broad categories: (1) relative emotional neutrality to major life events that cause nonmilitary civilians great joy or sadness; and (2) overreaction to trivial events, causing significant irritation, anger, or sadness that normally would not produce such emotional reactions in nonmilitary civilians. The former is largely overlooked in medical literature to date except in relation to the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) categories, and the latter is often described in limited terms as increased irritability, restlessness, and low frustration tolerance. This emotional dysregulation creates confusing paradoxes for veterans. For example, a veteran might feel no strong emotion when notified of the death of a close relative and yet cry uncontrollably while watching a sad scene in a fictional movie.
Sleep difficulties are intrinsic to the postdeployment period.9-12 Sleep-wake cycles often are altered, reflecting residual effects of the rigid schedules required by military duties and poor sleep hygiene in the combat theater. Inadequate, nonrestful sleep is frequently reported on return to the civilian world. Difficulty falling asleep or difficulty staying asleep also commonly occurs. Nightmares may be present.
Transient difficulty with concentration and attention is often prominent within the postdeployment transition period.9-11,13 Manifestations are variable, but problems with focusing on minor tasks are commonly reported. A more intense effort to master new concepts may be required. Learning styles developed during predeployment phases may be altered so that more conscious effort is required to comprehend and retain new information.
Short-term memory frequently may be affected during postdeployment transition.9-11,13 Veterans often report postdeployment difficulty in recalling appointments or tasks that must be completed even if they had a keen sense of memory during predeployment or deployment. Veterans also may have difficulty recalling the details of specific routines that were done without hesitation during deployment. Compared with predeployment time frames, veterans may exert greater effort to recall newly learned material.
Automatic behaviors necessary for survival in a combat theater still may be prominent in the postdeployment period.10,11,14 Aggressive driving required to avoid deployment ambush may be problematic during the postdeployment transition. Steering clear of any roadside trash may be a residual instinctive drive postdeployment because of the risk of improvised explosive devices concealed by debris in the combat theater. Veterans may avoid sitting with their back to the exit as the result of military safety training. Carrying weapons to ensure safety may be a compelling urge, because being armed and ready at all times was necessary for survival during deployment. Avoiding large crowds may be another strong tendency, because throngs of people were associated with potential danger in the combat theater.
Decision making may be challenging to resume in the postdeployment phase.9-11,15 In the deployment theater, time is relativel structured with rules in place, whereas at home veterans face a myriad of choices and decisions that must be made in order to complete the responsibilities of everyday living. As a result, making decisions about what item to buy, which clothes to wear, or what activities to prioritize, though relatively minor, can be a source of significant frustration. It may be difficult to independently navigate a realm of options available for new employment, schooling, or benefits, especially when there is little or no prior experience with these issues.
Relationship of Symptoms to Mental Health Diagnoses
Postdeployment transition symptoms do not automatically indicate the presence of an underlying mental health diagnosis. However, persistent and/or severe symptoms of postdeployment transition can overlap with or contribute to the development of mental health concerns (Table 1).14 The effects of the emotional disconnect also can exacerbate underlying mental health diagnoses.
While postdeployment emotional numbness to major life events, irritability, sleep disturbances, and impaired concentration can be associated with acute stress disorder (ASD) or PTSD, there is a constellation of other symptoms that must be present to diagnose these psychiatric conditions.16 Diagnostic criteria include persistent intrusive symptoms associated with the trauma, persistent avoidance of triggers/reminders associated with the trauma, significant changes in physiologic and cognitive arousal states, and negative changes in mood or cognition related to the trauma.16 The symptoms must cause significant impairment in some aspect of functioning on an individual, social, or occupational level. Acute stress disorder occurs when the symptoms last 30 days or less, whereas PTSD is diagnosed if the symptoms persist longer than a month.
Impaired emotional regulation, sleep disturbances, and decreased concentration also can be associated with depression or anxiety but are insufficient in themselves to make the diagnosis of those disorders.16 At least a 2-week history of depressed mood or inability to experience interest or pleasure in activities must be present as one of the criteria for depression as well as 4 or more other symptoms affecting sleep, appetite, energy, movement, self-esteem, or suicidal thoughts. Anxiety disorders have varying specific diagnostic criteria, but recurrent excessive worrying is a hallmark. Just like ASD or PTSD, the diagnostic symptoms of either depression or anxiety disorders must be causing significant impairment in functioning on an individual, social, or occupational level.
Irritability, sleep disturbances, agitation, memory impairment, and difficulty with concentration and attention can mimic the symptoms associated with mild-to-moderate traumatic brain injury (TBI).17,18 However, symptom onset must have a temporal relationship with a TBI. The presence of other TBI symptoms not associated with normal postdeployment transition usually can be used to differentiate between the diagnoses. Those TBI symptoms include recurrent headaches, poor balance, dizziness, tinnitus, and/or light sensitivity. In the majority of mild TBI cases, the symptoms resolve spontaneously within 3 months of TBI symptom manifestation.16,19 For those with persistent postconcussive syndrome, symptoms usually stabilize or improve over time.18,19 If symptoms worsen, there is often a confounding diagnosis such as PTSD or depression.17,20,21
Some returning combat veterans mistakenly believe postdeployment emotional transition symptoms are always a sign of a mental health disorder. Because there is a significant stigma associated with mental health disorders as well as potential repercussions on their service record if they use mental health resources, many reservists and National Guard members avoid accessing health care services if they are experiencing postdeployment adjustment issues, especially if those symptoms are related to emotional transitions.22-24 Unfortunately, such avoidance carries the risk that stress-inducing symptoms will persist and potentiate adjustment problems.
Course of Symptoms
The range for the postdeployment adjustment period generally falls within 3 to 12 months but can extend longer, depending on individual factors.10,11,25 Factors include presence of significant physical injury or illness, co-occurrence of mental health issues, underlying communication styles, and efficacy of coping strategies chosen. Although there is no clear-cut time frame for transition, ideally transition is complete when the returning veteran successfully enters his or her civilian lifestyle roles and feels a sense of purpose and belonging in society.
Postdeployment transition symptoms occur on a continuum in terms of duration and intensity for reservists and National Guard members. It is difficult to predict how specific transition symptoms will affect a particular veteran. The degree to which those symptoms will complicate reintegration depends on the individual veteran’s ability to adapt within the psychosocial context in which the symptoms occur. For example, minor irritation may be short-lived if a veteran can employ techniques to diffuse that feeling. Alternatively, minor irritation also suddenly may explode into a powerful wave of anger if the veteran has significant underlying emotional tension. Similarly, impaired short-term memory may be limited to forgetting a few appointments or may be so common that the veteran is at risk of losing track of his or her day. The level of memory impairment depends on emotional functioning, co-occurring stressors, and use of adaptive strategies.
In general, as these veterans successfully take on civilian routines, postdeployment transition symptoms will improve. Although such symptom improvement may be a passive process for some veterans, others will need to actively employ strategies to help change the military combat mind-set. The goal is to initiate useful interventions early in transition before symptoms become problematic.14
There are numerous self-help techniques and mobile apps that can be applied to a wide number of symptoms. Viable strategies include exercise, yoga, meditation, mindfulness training, and cognitive reframing. Reaching out for early assistance from various military assistance organizations that are well versed in dealing with postdeployment transition challenges often is helpful for reducing stress and navigating postdeployment obstacles (Table 2).
Symptom Strain and Exacerbation
Whenever stumbling blocks are encountered during the postdeployment period, any transition symptom can persist and/or worsen.10,11,14 Emotional disconnect and other transition symptoms can be exacerbated by physical, psychological, and social stressors common in the postdeployment period. Insomnia, poor quality sleep, or other sleep impairments that frequently occur as part of postdeployment transition can negatively impact the veteran’s ability to psychologically cope with daytime stressors. Poor concentration and short-term memory impairment noted by many reservists and National Guard members in the postdeployment phase can cause increased difficulty in attention to the moment and complicate completion of routine tasks. These difficulties can compound frustration and irritation to minor events and make it hard to emotionally connect with more serious issues.
Concentration and attention to mundane activities may be further reduced if the veteran feels no connection to the civilian world and/or experiences the surreal sensation that he or she should be attending to more serious life and death matters, such as those experienced in the combat theater. Ongoing psychological adjustment to physical injuries sustained during deployment can limit emotional flexibility when adapting to either minor or major stressors. Insufficient financial resources, work issues, or school problems can potentiate irritation, anger, and sadness and create an overwhelming emotional overload, leading to helplessness and hopelessness.
Perceived irregularities in emotional connection to the civilian world can significantly strain interpersonal relationships and be powerful impediments to successful reintegration.9,11,14 Failure to express emotions to major life events in the civilian world can result in combat veterans being viewed as not empathetic to others’ feelings. Overreaction to trivial events during postdeployment can lead to the veteran being labeled as unreasonable, controlling, and/or unpredictable. Persistent emotional disconnect with civilians engenders a growing sense of emotional isolation from family and friends when there is either incorrect interpretation of emotional transitions or failure to adapt healthy coping strategies. This isolation further enlarges the emotional chasm and may greatly diminish the veteran’s ability to seek assistance and appropriately address stressors in the civilian world.
Transition and the Family
Emotional disconnection may be more acutely felt within the immediate family unit.26 Redistribution of family unit responsibilities during deployment may mean that roles the veteran played during predeployment now may be handled by a partner. On the veteran’s return to the civilian world, such circumstances require active renegotiation of duties. Interactions with loved ones, especially children, may be colored by the family members’ individual perspectives on deployment as well as by the veteran’s transition symptoms. When there is disagreement about role responsibilities and/or underlying family resentment about deployment, conditions are ripe for significant discord between the veteran and family members, vital loss of partner intimacy, and notable loss of psychological safety to express feelings within the family unit. If there are concerns about infidelity by the veteran or significant other during the period of deployment, postdeployment tensions can further escalate. If unaddressed in the presence of emotional disconnect, any of these situations can raise the risk of domestic violence and destruction of relationships.
Without adequate knowledge of common postdeployment transitions and coping strategies, the postdeployment transition period is often bewildering to returning veterans and their families. They are taken aback by postdeployment behaviors that do not conform to the veteran’s predeployment personality or mannerisms. Families may feel they have “lost” the veteran and view the emotionally distant postdeployment veteran as a stranger. Veterans mistakenly may view the postdeployment emotional disconnect as evidence that they were permanently altered by deployment and no longer can assimilate into the civilian world. Unless veterans and families develop an awareness of the postdeployment transition symptoms and healthy coping strategies, these perspectives can contribute to a veteran’s persistent feelings of alienation, significant sense of personal failure, and loss of vital social supports.
When transition symptoms are or have the potential to become significant stressors, veterans would benefit from mental health counseling either individually or with family members. Overcoming the stigma of seeking mental health services can prove challenging. Explaining that these postdeployment symptoms occur commonly, stem from military combat training, can be reversed, and when reversed will empower the individual to control his or her life may help veterans overcome the stigma and seek mental health services.
The fear of future career impairment with the military reserve or National Guard is another real concern among this cohort who might consider accessing behavioral health care, especially since VA mental health medical records can be accessed by DoD officials through links with the VHA. Fortunately, this concern can be alleviated through the use of Vet Centers, free-standing counseling centers nationwide that offer no-cost individual and family counseling to veterans with combat exposure. Vet Center counseling records are completely confidential, never shared, and are not linked to the VHA electronic health record, the DoD, or any other entity. Although Vet Center providers don’t prescribe medications, the counselors can actively address many issues for veterans and their families. For individuals who do not live near a Vet Center or for those who require psychiatric medications, a frank discussion on the benefits of treatment vs the risk of treatment avoidance must be held.
Assessing Symptoms and Coping Mechanisms
Postdeployment transition symptoms vary, depending on the nature and context of the symptom. Not only must the returning reservist and National Guard member be screened for symptoms, but HCPs also should assess the impact of those symptoms on the veteran and his or her interpersonal relationships. Some veterans will feel that the symptoms have relatively minor impact in their lives, because the veteran can easily compensate for the transient effects. Others may feel that the symptoms are somewhat burdensome because the issues are complicating the smooth transition to civilian roles. Still others will judge the symptoms to be devastating because of the negative effects on personal control, selfesteem, and emotional connection with family and friends.
In addition to screening for symptoms, HCPs should assess these veterans’ current coping adaptations to various transition symptoms. Whereas some activities may be functional and promote reintegration, other short-term coping solutions may cripple the veteran’s ability to successfully resume civilian life. Global avoidance of communication with others and/or retreating from all social situations is a destructive coping pattern that can further alienate veterans from their families and the civilian world. Reacting with anger to all stressful issues is another maladaptive pattern of coping with life’s frustrations. Because of the potential to self-medicate when dealing with social difficulties, depression, anxiety, or other mental health diagnoses, veterans may develop an inappropriate reliance on drugs or alcohol to handle postdeployment stressors.27 Therefore, HCP screening for substance use disorders (SUD) is important so that interventions can be initiated early.
Because of the overlap of postdeployment transition symptoms with mental health disorders and the relative frequency of those mental health disorders among combat veterans, HCPs should have a heightened awareness of the potential for co-occurring mental health difficulties in the postdeployment reservist and National Guard cohort. Health care providers should screen for depression, anxiety, and PTSD. Even if initial screening is done early within the transition period, repeat screening would be of benefit 6 months into the postdeployment period because of the tendency of mental health issues to develop during that time.28,29
By evaluating the impact of the transition symptom and coping strategies on these veterans’ lives, HCPs can better determine which strategies might adequately compensate for symptom effects. In general, informal counseling, even if just to help veterans normalize postdeployment transition symptoms and develop a plan to address such symptoms, can significantly minimize the negative impact of transition symptoms.14,26 Specific symptoms should be targeted by interventions that match the degree of symptom impact.
Symptoms to be aggressively addressed are those that significantly interfere with successful reintegration into the civilian world. For example, persistent sleep difficulties should be dealt with because they can worsen all other transition symptoms. However, the majority of strategies to address sleep do not require medication unless there are confounding factors such as severe nightmares. Minor memory issues attributed to the transition phase can be mitigated by several strategies to improve recall, including use of task lists, digital calendars, or other memory-prodding techniques. However, severe memory issues related to depression or anxiety likely would require pharmaceutical assistance and formal counseling in addition to other nonpharmacologic approaches.
Intermittent irritation or restlessness may be amenable to selfhelp strategies, but significant anger outbursts or aggression will require additional support, such as formal behavioral interventions to help identify the triggers and develop strategic plans to reduce emotional tension. A mild sense of not belonging may resolve without intervention, but a stronger sense of alienation will require further evaluation.
Conclusion
Civilian reintegration after combat deployment is a gradual process rather than a discrete event for reservists and National Guard members. There are common, nonpathologic postdeployment transition symptoms that, if misunderstood or inappropriately addressed, can complicate civilian reintegration. Health care providers are in the unique position to promote a healthy postdeployment transition by assisting veterans to recognize nonpathologic transition symptoms, select appropriate coping strategies, and seek further assistance for more complex problems.
1. Pew Research Center. War and sacrifice in the post 9/11 era: executive summary. http://www
.pewsocialtrends.org/2011/10/05/war-and-sacrifice-in-the-post-911-era. Published October 5, 2011. Accessed June 12, 2017.
2. Interian A, Kline A, Callahan L, Losonczy M. Readjustment stressors and early mental health treatment seeking by returning National Guard soldiers with PTSD. Psychiatr Serv. 2012;63(9):855-861.
3. Spelman JF, Hunt SC, Seal KH, Burgo-Black AL. Post deployment care for returning combat veterans. J Gen Intern Med. 2012;27(9):1200-1209.
4. Vasterling JJ, Daily ES, Friedman MJ. Posttraumatic stress reactions over time: the battlefield, homecoming, and long-term course. In: Ruzek JI, Schnurr PP, Vasterling JJ, Friedman MJ, eds. Caring for Veterans With Deployment-Related Stress Disorders: Iraq, Afghanistan, and Beyond. Washington,DC: American Psychological Association;2011:chap 2.
5. Wilcox SL, Oh H, Redmon SA, Chicas J, Hassan AM, Lee PJ, Ell K. A scope of the problem: Postdeployment reintegration challenges in a National Guard Unit. Work. 2015;50(1):73-83.
6. Griffith J. Homecoming of citizen soldiers: Postdeployment problems and service use among Army National Guard soldiers. Community Ment Health J. 2017. doi:10.1007/s10597-017-0132-9. (Epub ahead of print)
7. Schultz M, Glickman ME, Eisen SV. Predictors of decline in overall mental health, PTSD and alcohol use in OEF/OIF veterans. Comprehensive Psychiatry. 2014;55(7):1654-1664.
8. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in National Guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011:68(1):79-89.
9. U.S. Department of Veterans Affairs, National Center for PTSD. Returning from the war zone: a guide for military personnel. http://www.ptsd.va.gov/public/reintegration/guide-pdf/SMGuide.pdf. Updated January 2014. Accessed June 12, 2017.
10. Slone LB, Friedman MJ. After the War Zone: A Practical Guide for Returning Troops and their Families. Philadelphia, PA: Da Capo Press; 2008.
11. Ainspan ND, Penk WE, eds. When the Warrior Returns: Making the Transition at Home. Annapolis, MD: Naval Institute Press; 2012.
12. Yosick T, Bates M, Moore M, Crowe C, Phillips J, Davison J. A review of post-deployment reintegration: evidence, challenges, and strategies for program development. http://www.dcoe.mil/files/Review_of_Post-Deployment_Reintegration.pdf. Published February 10, 2012. Accessed June 12, 2017.
13. Vasterling JJ, Proctor SP, Amoroso P, Kane R, Heeren T, White RF. Neuropsychological outcomes of army personnel following deployment to the Iraq war. JAMA. 2006;296(5):519-529.
14. Castro CA, Kintzle S, Hassan AM. The combat veteran paradox: paradoxes and dilemmas encountered with reintegrating combat veterans and the agencies that support them. Traumatology. 2015;21(4):299-310.
15. Rivers FM, Gordon S, Speraw S, Reese S. U.S. Army nurses’ reintegration and homecoming experiences after Iraq and Afghanistan. Mil Med. 2013;178(2):166-173.
16. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington,VA: American Psychiatric Association;2013.
17. Tanielian T, Jaycox LH, eds. Invisible Wounds of War: Psychological and Cognitive Injuries, Their Consequences, and Services to Assist Recovery. Santa Monica, CA: Rand Corporation, 2008.
18. Orff HJ, Hays CC, Heldreth AA, Stein MB, Twamley EW. Clinical considerations in the evaluation and management of patients following traumatic brain injury. Focus. 2013;11(3):328-340.
19. Morissette SB, Woodward M, Kimbrel NA, et al. Deployment-related TBI, persistent postconcussive symptoms, PTSD, and depression in OEF/OIF veterans. Rehabil Psychol. 2011;56(4):340-350.
20. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in national guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011;68(1):79-89.
21. Wilk JE, Herrell RK, Wynn GH, Riviere LA, Hoge CW. Mild traumatic brain injury (concussion), posttraumatic stress disorder, and depression in U.S. soldiers involved in combat deployments: association with postdeployment symptoms. Psychosom Med. 2012;74(3):249-257.
22. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
23. Hines LA, Goodwin L, Jones M, et al. Factors affecting help seeking for mental health problems after deployment to Iraq and Afghanistan. Psychiatr Serv. 2014;65(1):98-105.
24. Gorman LA, Blow AJ, Ames BD, Read PL. National Guard families after combat: mental health, use of mental health services, and perceived treatment barriers. Psychiatr Serv. 2011;62(1):28-34.
25. Marek LI, Hollingsworth WG, D’Aniello C, et al. Returning home: what we know about the reintegration of deployed service members into their families and communities. https://www.ncfr.org/ncfr-report/focus/military-families/returninghome. Published March 1, 2012. Accessed June 13, 2017.
26. Bowling UB, Sherman MD. Welcoming them home: supporting service members and their families in navigating the tasks of reintegration. Prof Psychol Res Pr. 2008;39(4):451-458.
27. Jacobson IG, Ryan MA, Hooper TI, et al. Alcohol use and alcohol-related problems before
and after military combat deployment. JAMA. 2008;300(6):663-675.
28. Seal KH, Metzler TH, Gima KS, Bertenthal D, Maguen S, Marmar CR. Trends and risk factors for mental health diagnoses among Iraq and Afghanistan veterans Department of Veterans Affairs health care, 2002-2008. Am J Public Health. 2009;99(9):1651-1658.
29. Milliken CS, Auchterlonie JL, Hoge CW. Longitudinal assessment of mental health problems among active and reserve component soldiers returning from the Iraq war. JAMA. 2007;298(18):2141-2148.
1. Pew Research Center. War and sacrifice in the post 9/11 era: executive summary. http://www
.pewsocialtrends.org/2011/10/05/war-and-sacrifice-in-the-post-911-era. Published October 5, 2011. Accessed June 12, 2017.
2. Interian A, Kline A, Callahan L, Losonczy M. Readjustment stressors and early mental health treatment seeking by returning National Guard soldiers with PTSD. Psychiatr Serv. 2012;63(9):855-861.
3. Spelman JF, Hunt SC, Seal KH, Burgo-Black AL. Post deployment care for returning combat veterans. J Gen Intern Med. 2012;27(9):1200-1209.
4. Vasterling JJ, Daily ES, Friedman MJ. Posttraumatic stress reactions over time: the battlefield, homecoming, and long-term course. In: Ruzek JI, Schnurr PP, Vasterling JJ, Friedman MJ, eds. Caring for Veterans With Deployment-Related Stress Disorders: Iraq, Afghanistan, and Beyond. Washington,DC: American Psychological Association;2011:chap 2.
5. Wilcox SL, Oh H, Redmon SA, Chicas J, Hassan AM, Lee PJ, Ell K. A scope of the problem: Postdeployment reintegration challenges in a National Guard Unit. Work. 2015;50(1):73-83.
6. Griffith J. Homecoming of citizen soldiers: Postdeployment problems and service use among Army National Guard soldiers. Community Ment Health J. 2017. doi:10.1007/s10597-017-0132-9. (Epub ahead of print)
7. Schultz M, Glickman ME, Eisen SV. Predictors of decline in overall mental health, PTSD and alcohol use in OEF/OIF veterans. Comprehensive Psychiatry. 2014;55(7):1654-1664.
8. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in National Guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011:68(1):79-89.
9. U.S. Department of Veterans Affairs, National Center for PTSD. Returning from the war zone: a guide for military personnel. http://www.ptsd.va.gov/public/reintegration/guide-pdf/SMGuide.pdf. Updated January 2014. Accessed June 12, 2017.
10. Slone LB, Friedman MJ. After the War Zone: A Practical Guide for Returning Troops and their Families. Philadelphia, PA: Da Capo Press; 2008.
11. Ainspan ND, Penk WE, eds. When the Warrior Returns: Making the Transition at Home. Annapolis, MD: Naval Institute Press; 2012.
12. Yosick T, Bates M, Moore M, Crowe C, Phillips J, Davison J. A review of post-deployment reintegration: evidence, challenges, and strategies for program development. http://www.dcoe.mil/files/Review_of_Post-Deployment_Reintegration.pdf. Published February 10, 2012. Accessed June 12, 2017.
13. Vasterling JJ, Proctor SP, Amoroso P, Kane R, Heeren T, White RF. Neuropsychological outcomes of army personnel following deployment to the Iraq war. JAMA. 2006;296(5):519-529.
14. Castro CA, Kintzle S, Hassan AM. The combat veteran paradox: paradoxes and dilemmas encountered with reintegrating combat veterans and the agencies that support them. Traumatology. 2015;21(4):299-310.
15. Rivers FM, Gordon S, Speraw S, Reese S. U.S. Army nurses’ reintegration and homecoming experiences after Iraq and Afghanistan. Mil Med. 2013;178(2):166-173.
16. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington,VA: American Psychiatric Association;2013.
17. Tanielian T, Jaycox LH, eds. Invisible Wounds of War: Psychological and Cognitive Injuries, Their Consequences, and Services to Assist Recovery. Santa Monica, CA: Rand Corporation, 2008.
18. Orff HJ, Hays CC, Heldreth AA, Stein MB, Twamley EW. Clinical considerations in the evaluation and management of patients following traumatic brain injury. Focus. 2013;11(3):328-340.
19. Morissette SB, Woodward M, Kimbrel NA, et al. Deployment-related TBI, persistent postconcussive symptoms, PTSD, and depression in OEF/OIF veterans. Rehabil Psychol. 2011;56(4):340-350.
20. Polusny MA, Kehle SM, Nelson NW, Erbes CR, Arbisi PA, Thuras P. Longitudinal effects of mild traumatic brain injury and posttraumatic stress disorder comorbidity on postdeployment outcomes in national guard soldiers deployed to Iraq. Arch Gen Psychiatry. 2011;68(1):79-89.
21. Wilk JE, Herrell RK, Wynn GH, Riviere LA, Hoge CW. Mild traumatic brain injury (concussion), posttraumatic stress disorder, and depression in U.S. soldiers involved in combat deployments: association with postdeployment symptoms. Psychosom Med. 2012;74(3):249-257.
22. Hoge CW, Grossman SH, Auchterlonie JL, Riviere LA, Milliken CS, Wilk JE. PTSD treatment for soldiers after combat deployment: low utilization of mental health care and reasons for dropout. Psychiatr Serv. 2014;65(8):997-1004.
23. Hines LA, Goodwin L, Jones M, et al. Factors affecting help seeking for mental health problems after deployment to Iraq and Afghanistan. Psychiatr Serv. 2014;65(1):98-105.
24. Gorman LA, Blow AJ, Ames BD, Read PL. National Guard families after combat: mental health, use of mental health services, and perceived treatment barriers. Psychiatr Serv. 2011;62(1):28-34.
25. Marek LI, Hollingsworth WG, D’Aniello C, et al. Returning home: what we know about the reintegration of deployed service members into their families and communities. https://www.ncfr.org/ncfr-report/focus/military-families/returninghome. Published March 1, 2012. Accessed June 13, 2017.
26. Bowling UB, Sherman MD. Welcoming them home: supporting service members and their families in navigating the tasks of reintegration. Prof Psychol Res Pr. 2008;39(4):451-458.
27. Jacobson IG, Ryan MA, Hooper TI, et al. Alcohol use and alcohol-related problems before
and after military combat deployment. JAMA. 2008;300(6):663-675.
28. Seal KH, Metzler TH, Gima KS, Bertenthal D, Maguen S, Marmar CR. Trends and risk factors for mental health diagnoses among Iraq and Afghanistan veterans Department of Veterans Affairs health care, 2002-2008. Am J Public Health. 2009;99(9):1651-1658.
29. Milliken CS, Auchterlonie JL, Hoge CW. Longitudinal assessment of mental health problems among active and reserve component soldiers returning from the Iraq war. JAMA. 2007;298(18):2141-2148.
Luspatercept appears safe, effective in β-thalassemia
MADRID—Results of a phase 2 study have shown that luspatercept can produce sustained increases in hemoglobin and reductions in transfusion burden in adults with β-thalassemia.
Some patients are still receiving the drug and experiencing clinical benefits beyond 24 months.
Luspatercept has been well-tolerated in these patients, producing no serious adverse events (AEs).
“Luspatercept has many characteristics that are promising . . .,” said Antonio G. Piga, MD, of Turin University in Turin, Italy.
He presented results of the phase 2 study at the 22nd Congress of the European Hematology Association (EHA) as abstract S129.
The study was sponsored by Celgene in collaboration with Acceleron Pharma.
Dr Piga presented data on 63 patients—32 of whom were transfusion-dependent (TD) and 31 of whom were non-transfusion-dependent (NTD).
For the entire study cohort, the median age was 38 (range, 20-62), 52% of patients were male, and 67% had undergone a splenectomy.
In the NTD patients, the median hemoglobin at baseline was 8.5 g/dL (range, 6.5-9.8).
The TD patients received a median of 8 (range, 4-18) red blood cell (RBC) units every 12 weeks.
For the 3-month base study, patients received luspatercept at 0.2 mg/kg to 1.25 mg/kg every 3 weeks.
In the ongoing extension study, patients can receive luspatercept at 0.8 mg/kg to 1.25 mg/kg every 3 weeks for up to 5 years.
Efficacy in NTD patients
The median duration of treatment for NTD patients (n=31) was 18.6 months (range, 1.3-29.4 months; ongoing).
Over a 12-week period, 71% (22/31) of NTD patients saw at least a 1.0 g/dL increase in mean hemoglobin from baseline. Fifty-two percent (16/31) saw an increase of 1.5 g/dL or greater.
To assess quality of life in NTD patients, the researchers used FACIT-F, a 13-item questionnaire used to assess anemia-related symptoms such as fatigue and weakness.
Fifty-eight percent (7/12) of patients with a baseline FACIT-F deficit (<44 points) had improved by at least 3 points at 48 weeks.
And 86% (6/7) of patients with at least a 3-point increase in FACIT-F score had at least a 1.0 g/dL improvement in mean hemoglobin over a 12-week period.
Efficacy in TD patients
The median duration of treatment for TD patients was 14.2 months (range, 0.7-27.2 months, ongoing).
Seventy-eight percent (25/32) of these patients had at least a 20% reduction in RBC units transfused from 12 weeks pre-treatment to any 12-week interval on treatment. Sixty-nine percent (22/32) had at least a 33% reduction at any 12-week interval.
Fifty percent of patients (12/24) who received an estimated 6 to 20 RBC units every 24 weeks achieved a reduction in transfusion burden from baseline of at least 33% in the fixed 12-week interval from weeks 13 to 24.
Forty-six percent (11/24) achieved a reduction in transfusion burden from baseline of at least 33% in the interval from weeks 37 to 48.
Safety
The most common AEs possibly or probably related to luspatercept were bone pain (38%), headache (28%), myalgia (22%), arthralgia (19%), musculoskeletal pain (17%), asthenia (14%), injection site pain (13%), and back pain (11%).
Most AEs were grade 1 or 2 in severity. Treatment-related grade 3 AEs included bone pain (n=3), asthenia (n=2), and headache (n=1).
Dr Piga said these results support an ongoing phase 3 study of luspatercept in regularly transfused patients with β-thalassemia (BELIEVE, NCT02604433), which recently completed enrollment. ![]()
MADRID—Results of a phase 2 study have shown that luspatercept can produce sustained increases in hemoglobin and reductions in transfusion burden in adults with β-thalassemia.
Some patients are still receiving the drug and experiencing clinical benefits beyond 24 months.
Luspatercept has been well-tolerated in these patients, producing no serious adverse events (AEs).
“Luspatercept has many characteristics that are promising . . .,” said Antonio G. Piga, MD, of Turin University in Turin, Italy.
He presented results of the phase 2 study at the 22nd Congress of the European Hematology Association (EHA) as abstract S129.
The study was sponsored by Celgene in collaboration with Acceleron Pharma.
Dr Piga presented data on 63 patients—32 of whom were transfusion-dependent (TD) and 31 of whom were non-transfusion-dependent (NTD).
For the entire study cohort, the median age was 38 (range, 20-62), 52% of patients were male, and 67% had undergone a splenectomy.
In the NTD patients, the median hemoglobin at baseline was 8.5 g/dL (range, 6.5-9.8).
The TD patients received a median of 8 (range, 4-18) red blood cell (RBC) units every 12 weeks.
For the 3-month base study, patients received luspatercept at 0.2 mg/kg to 1.25 mg/kg every 3 weeks.
In the ongoing extension study, patients can receive luspatercept at 0.8 mg/kg to 1.25 mg/kg every 3 weeks for up to 5 years.
Efficacy in NTD patients
The median duration of treatment for NTD patients (n=31) was 18.6 months (range, 1.3-29.4 months; ongoing).
Over a 12-week period, 71% (22/31) of NTD patients saw at least a 1.0 g/dL increase in mean hemoglobin from baseline. Fifty-two percent (16/31) saw an increase of 1.5 g/dL or greater.
To assess quality of life in NTD patients, the researchers used FACIT-F, a 13-item questionnaire used to assess anemia-related symptoms such as fatigue and weakness.
Fifty-eight percent (7/12) of patients with a baseline FACIT-F deficit (<44 points) had improved by at least 3 points at 48 weeks.
And 86% (6/7) of patients with at least a 3-point increase in FACIT-F score had at least a 1.0 g/dL improvement in mean hemoglobin over a 12-week period.
Efficacy in TD patients
The median duration of treatment for TD patients was 14.2 months (range, 0.7-27.2 months, ongoing).
Seventy-eight percent (25/32) of these patients had at least a 20% reduction in RBC units transfused from 12 weeks pre-treatment to any 12-week interval on treatment. Sixty-nine percent (22/32) had at least a 33% reduction at any 12-week interval.
Fifty percent of patients (12/24) who received an estimated 6 to 20 RBC units every 24 weeks achieved a reduction in transfusion burden from baseline of at least 33% in the fixed 12-week interval from weeks 13 to 24.
Forty-six percent (11/24) achieved a reduction in transfusion burden from baseline of at least 33% in the interval from weeks 37 to 48.
Safety
The most common AEs possibly or probably related to luspatercept were bone pain (38%), headache (28%), myalgia (22%), arthralgia (19%), musculoskeletal pain (17%), asthenia (14%), injection site pain (13%), and back pain (11%).
Most AEs were grade 1 or 2 in severity. Treatment-related grade 3 AEs included bone pain (n=3), asthenia (n=2), and headache (n=1).
Dr Piga said these results support an ongoing phase 3 study of luspatercept in regularly transfused patients with β-thalassemia (BELIEVE, NCT02604433), which recently completed enrollment. ![]()
MADRID—Results of a phase 2 study have shown that luspatercept can produce sustained increases in hemoglobin and reductions in transfusion burden in adults with β-thalassemia.
Some patients are still receiving the drug and experiencing clinical benefits beyond 24 months.
Luspatercept has been well-tolerated in these patients, producing no serious adverse events (AEs).
“Luspatercept has many characteristics that are promising . . .,” said Antonio G. Piga, MD, of Turin University in Turin, Italy.
He presented results of the phase 2 study at the 22nd Congress of the European Hematology Association (EHA) as abstract S129.
The study was sponsored by Celgene in collaboration with Acceleron Pharma.
Dr Piga presented data on 63 patients—32 of whom were transfusion-dependent (TD) and 31 of whom were non-transfusion-dependent (NTD).
For the entire study cohort, the median age was 38 (range, 20-62), 52% of patients were male, and 67% had undergone a splenectomy.
In the NTD patients, the median hemoglobin at baseline was 8.5 g/dL (range, 6.5-9.8).
The TD patients received a median of 8 (range, 4-18) red blood cell (RBC) units every 12 weeks.
For the 3-month base study, patients received luspatercept at 0.2 mg/kg to 1.25 mg/kg every 3 weeks.
In the ongoing extension study, patients can receive luspatercept at 0.8 mg/kg to 1.25 mg/kg every 3 weeks for up to 5 years.
Efficacy in NTD patients
The median duration of treatment for NTD patients (n=31) was 18.6 months (range, 1.3-29.4 months; ongoing).
Over a 12-week period, 71% (22/31) of NTD patients saw at least a 1.0 g/dL increase in mean hemoglobin from baseline. Fifty-two percent (16/31) saw an increase of 1.5 g/dL or greater.
To assess quality of life in NTD patients, the researchers used FACIT-F, a 13-item questionnaire used to assess anemia-related symptoms such as fatigue and weakness.
Fifty-eight percent (7/12) of patients with a baseline FACIT-F deficit (<44 points) had improved by at least 3 points at 48 weeks.
And 86% (6/7) of patients with at least a 3-point increase in FACIT-F score had at least a 1.0 g/dL improvement in mean hemoglobin over a 12-week period.
Efficacy in TD patients
The median duration of treatment for TD patients was 14.2 months (range, 0.7-27.2 months, ongoing).
Seventy-eight percent (25/32) of these patients had at least a 20% reduction in RBC units transfused from 12 weeks pre-treatment to any 12-week interval on treatment. Sixty-nine percent (22/32) had at least a 33% reduction at any 12-week interval.
Fifty percent of patients (12/24) who received an estimated 6 to 20 RBC units every 24 weeks achieved a reduction in transfusion burden from baseline of at least 33% in the fixed 12-week interval from weeks 13 to 24.
Forty-six percent (11/24) achieved a reduction in transfusion burden from baseline of at least 33% in the interval from weeks 37 to 48.
Safety
The most common AEs possibly or probably related to luspatercept were bone pain (38%), headache (28%), myalgia (22%), arthralgia (19%), musculoskeletal pain (17%), asthenia (14%), injection site pain (13%), and back pain (11%).
Most AEs were grade 1 or 2 in severity. Treatment-related grade 3 AEs included bone pain (n=3), asthenia (n=2), and headache (n=1).
Dr Piga said these results support an ongoing phase 3 study of luspatercept in regularly transfused patients with β-thalassemia (BELIEVE, NCT02604433), which recently completed enrollment. ![]()
Nivolumab for long-term treatment of cHL after auto-HSCT
LUGANO, SWITZERLAND—Nivolumab can provide long-term treatment for a broad range of adults who have relapsed or refractory classical Hodgkin lymphoma (cHL) after autologous hematopoietic stem cell transplant (auto-HSCT), according to a presentation at the 14th International Conference on Malignant Lymphoma (ICML).
In the phase 2 CheckMate-205 study, cHL patients achieved durable responses regardless of the depth of response, previous exposure to brentuximab vedotin (BV), and refractoriness to prior therapies.
Researchers observed sustained progression-free survival (PFS) in patients with stable disease (SD) or better, and the safety profile of nivolumab was considered acceptable.
“Nivolumab offers a favorable treatment outcome for patients who have relapsed disease after autologous stem cell transplant,” said Michelle Fanale, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fanale presented results from CheckMate-205 at 14-ICML. The study was sponsored by Bristol-Myers Squibb Company.
CheckMate-205 enrolled 243 adults with relapsed or refractory cHL who had undergone auto-HSCT. Patients were divided into 3 cohorts:
- Cohort A included patients who were naïve to BV (n=63)
- Cohort B included patients who received BV only after auto-HSCT (n=80)
- Cohort C included patients who received BV before and/or after auto-HSCT (n=100).
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year were to discontinue nivolumab, but they could resume treatment with the drug if they relapsed within 2 years.
Patient characteristics
The median age was 33 (range, 18-65) in cohort A, 37 (range, 18-72) in cohort B, and 32 (range, 19-69) in cohort C.
ECOG performance status was 0 for 62% of patients in cohort A, 53% in cohort B, and 50% in cohort C. The remaining patients had a performance status of 1.
The percentage of patients with stage IV disease was 38% in cohort A, 68% in cohort B, and 61% in cohort C.
The median number of prior therapies was 2 (range, 2-8) in cohort A, 4 (range, 3-15) in cohort B, and 4 (range, 2-9) in cohort C. Fifty-nine percent, 74%, and 69% of patients, respectively, had received prior radiotherapy.
The median time from diagnosis to the first dose of nivolumab was 3.1 years (range, 1.0-30.6) in cohort A, 6.2 years (range, 1.3-25.1) in cohort B, and 3.5 years (range, 1.0-24.9) in cohort C.
The median time from auto-HSCT to the first dose of nivolumab was 1.0 years (range, 0.3-18.2) in cohort A, 3.4 years (range, 0.2-19.0) in cohort B, and 1.7 years (range, 0.2-17.0) in cohort C.
Safety
The most common drug-related adverse events (AEs) were fatigue (23% any grade, 1% grade 3/4), diarrhea (15% any grade, 1% grade 3/4), infusion-related reactions (14% any grade, <1% grade 3/4), rash (12% any grade, 1% grade 3/4), nausea (10% grade 1/2), and pruritus (10% grade 1/2).
The most common drug-related serious AEs were infusion-related reactions (2% any grade, <1% grade 3/4) and pneumonitis (1% grade 1/2).
Drug-related AEs leading to treatment discontinuation were pneumonitis (2% grade 1/2) and autoimmune hepatitis (1% grade 3/4).
There were no deaths due to drug-related AEs.
Response
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
Partial response (PR) was the best response for 53% of all patients, 37% of patients in cohort A, 55% in cohort B, and 61% in cohort C.
SD was the best response for 19% of all patients, 24% of patients in cohort A, 21% in cohort B, and 15% in cohort C.
In post-hoc analyses, responses were similar irrespective of BV treatment sequence.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, and 15 months for cohort C.
The median duration of response in patients with a CR was 20 months overall and for cohorts A and B, but it was 15 months for cohort C.
The median duration of response in patients with a PR was 13 months overall, 17 months for cohort A, 11 months for cohort B, and 13 months for cohort C.
Survival
The median PFS for all patients was 15 months (range, 11-19). The median PFS was 22 months (range, 19-not reached) for patients who achieved a CR, 15 months (range, 11-19) for those who achieved a PR, and 11 months (range, 6-18) for those who had SD.
The median PFS was 18 months (range, 11-22) for patients in cohort A, 15 months (range, 11-20) for cohort B, and 12 months (range, 11-18) for cohort C.
The median overall survival (OS) has not been reached in any of the cohorts. The 12-months OS is 92% overall, 93% in cohort A, 95% in cohort B, and 90% in cohort C.
Patient status after extended follow-up
Forty percent of all patients were still on treatment after extended follow-up, as were 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
The most common reason for stopping treatment was disease progression—25% of cohort A, 28% of cohort B, and 24% of cohort C.
Patients also stopped treatment due to nivolumab-related toxicity—5% in cohort A, 11% in cohort B, and 7% in cohort C. Three percent, 1%, and 1%, respectively, stopped due to AEs unrelated to nivolumab.
Three percent of patients in cohort C stopped because they had attained the maximum clinical benefit, and 8% in cohort C completed treatment. This includes 7 patients who discontinued treatment because they were in CR for 1 year.
None of the patients in cohort A or B discontinued because they attained the maximum clinical benefit or because they completed treatment.
Eight percent of patients in cohort A, 10% in cohort B, and 17% in cohort C discontinued so they could proceed to HSCT.
Outcomes after allo-HSCT
Forty-four patients received allogeneic (allo-) HSCT after nivolumab. The median post-HSCT follow-up was 5.5 months (range, 0-19), and the median time from last dose of nivolumab to allo-HSCT was 1.6 months (range, 0.5-13.5).
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 27%. The rate of grade 3-4 acute GVHD was 17%, and the rate of chronic GVHD was 10%. At 6 months, the rates were 30%, 20%, and 15%, respectively.
The incidence of transplant-related mortality was 13% at 100 days and at 6 months.
“While there are risks, potentially, for acute GVHD and transplant-related mortality, these aren’t necessarily significantly different from what we’ve seen from other historical publications,” Dr Fanale said.
She cited data showing that the 100-day incidence of acute GVHD in cHL patients who underwent allo-HSCT ranges from 26% to 60%, and the incidence of transplant-related mortality in these patients ranges from 6% to 28%. ![]()
LUGANO, SWITZERLAND—Nivolumab can provide long-term treatment for a broad range of adults who have relapsed or refractory classical Hodgkin lymphoma (cHL) after autologous hematopoietic stem cell transplant (auto-HSCT), according to a presentation at the 14th International Conference on Malignant Lymphoma (ICML).
In the phase 2 CheckMate-205 study, cHL patients achieved durable responses regardless of the depth of response, previous exposure to brentuximab vedotin (BV), and refractoriness to prior therapies.
Researchers observed sustained progression-free survival (PFS) in patients with stable disease (SD) or better, and the safety profile of nivolumab was considered acceptable.
“Nivolumab offers a favorable treatment outcome for patients who have relapsed disease after autologous stem cell transplant,” said Michelle Fanale, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fanale presented results from CheckMate-205 at 14-ICML. The study was sponsored by Bristol-Myers Squibb Company.
CheckMate-205 enrolled 243 adults with relapsed or refractory cHL who had undergone auto-HSCT. Patients were divided into 3 cohorts:
- Cohort A included patients who were naïve to BV (n=63)
- Cohort B included patients who received BV only after auto-HSCT (n=80)
- Cohort C included patients who received BV before and/or after auto-HSCT (n=100).
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year were to discontinue nivolumab, but they could resume treatment with the drug if they relapsed within 2 years.
Patient characteristics
The median age was 33 (range, 18-65) in cohort A, 37 (range, 18-72) in cohort B, and 32 (range, 19-69) in cohort C.
ECOG performance status was 0 for 62% of patients in cohort A, 53% in cohort B, and 50% in cohort C. The remaining patients had a performance status of 1.
The percentage of patients with stage IV disease was 38% in cohort A, 68% in cohort B, and 61% in cohort C.
The median number of prior therapies was 2 (range, 2-8) in cohort A, 4 (range, 3-15) in cohort B, and 4 (range, 2-9) in cohort C. Fifty-nine percent, 74%, and 69% of patients, respectively, had received prior radiotherapy.
The median time from diagnosis to the first dose of nivolumab was 3.1 years (range, 1.0-30.6) in cohort A, 6.2 years (range, 1.3-25.1) in cohort B, and 3.5 years (range, 1.0-24.9) in cohort C.
The median time from auto-HSCT to the first dose of nivolumab was 1.0 years (range, 0.3-18.2) in cohort A, 3.4 years (range, 0.2-19.0) in cohort B, and 1.7 years (range, 0.2-17.0) in cohort C.
Safety
The most common drug-related adverse events (AEs) were fatigue (23% any grade, 1% grade 3/4), diarrhea (15% any grade, 1% grade 3/4), infusion-related reactions (14% any grade, <1% grade 3/4), rash (12% any grade, 1% grade 3/4), nausea (10% grade 1/2), and pruritus (10% grade 1/2).
The most common drug-related serious AEs were infusion-related reactions (2% any grade, <1% grade 3/4) and pneumonitis (1% grade 1/2).
Drug-related AEs leading to treatment discontinuation were pneumonitis (2% grade 1/2) and autoimmune hepatitis (1% grade 3/4).
There were no deaths due to drug-related AEs.
Response
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
Partial response (PR) was the best response for 53% of all patients, 37% of patients in cohort A, 55% in cohort B, and 61% in cohort C.
SD was the best response for 19% of all patients, 24% of patients in cohort A, 21% in cohort B, and 15% in cohort C.
In post-hoc analyses, responses were similar irrespective of BV treatment sequence.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, and 15 months for cohort C.
The median duration of response in patients with a CR was 20 months overall and for cohorts A and B, but it was 15 months for cohort C.
The median duration of response in patients with a PR was 13 months overall, 17 months for cohort A, 11 months for cohort B, and 13 months for cohort C.
Survival
The median PFS for all patients was 15 months (range, 11-19). The median PFS was 22 months (range, 19-not reached) for patients who achieved a CR, 15 months (range, 11-19) for those who achieved a PR, and 11 months (range, 6-18) for those who had SD.
The median PFS was 18 months (range, 11-22) for patients in cohort A, 15 months (range, 11-20) for cohort B, and 12 months (range, 11-18) for cohort C.
The median overall survival (OS) has not been reached in any of the cohorts. The 12-months OS is 92% overall, 93% in cohort A, 95% in cohort B, and 90% in cohort C.
Patient status after extended follow-up
Forty percent of all patients were still on treatment after extended follow-up, as were 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
The most common reason for stopping treatment was disease progression—25% of cohort A, 28% of cohort B, and 24% of cohort C.
Patients also stopped treatment due to nivolumab-related toxicity—5% in cohort A, 11% in cohort B, and 7% in cohort C. Three percent, 1%, and 1%, respectively, stopped due to AEs unrelated to nivolumab.
Three percent of patients in cohort C stopped because they had attained the maximum clinical benefit, and 8% in cohort C completed treatment. This includes 7 patients who discontinued treatment because they were in CR for 1 year.
None of the patients in cohort A or B discontinued because they attained the maximum clinical benefit or because they completed treatment.
Eight percent of patients in cohort A, 10% in cohort B, and 17% in cohort C discontinued so they could proceed to HSCT.
Outcomes after allo-HSCT
Forty-four patients received allogeneic (allo-) HSCT after nivolumab. The median post-HSCT follow-up was 5.5 months (range, 0-19), and the median time from last dose of nivolumab to allo-HSCT was 1.6 months (range, 0.5-13.5).
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 27%. The rate of grade 3-4 acute GVHD was 17%, and the rate of chronic GVHD was 10%. At 6 months, the rates were 30%, 20%, and 15%, respectively.
The incidence of transplant-related mortality was 13% at 100 days and at 6 months.
“While there are risks, potentially, for acute GVHD and transplant-related mortality, these aren’t necessarily significantly different from what we’ve seen from other historical publications,” Dr Fanale said.
She cited data showing that the 100-day incidence of acute GVHD in cHL patients who underwent allo-HSCT ranges from 26% to 60%, and the incidence of transplant-related mortality in these patients ranges from 6% to 28%. ![]()
LUGANO, SWITZERLAND—Nivolumab can provide long-term treatment for a broad range of adults who have relapsed or refractory classical Hodgkin lymphoma (cHL) after autologous hematopoietic stem cell transplant (auto-HSCT), according to a presentation at the 14th International Conference on Malignant Lymphoma (ICML).
In the phase 2 CheckMate-205 study, cHL patients achieved durable responses regardless of the depth of response, previous exposure to brentuximab vedotin (BV), and refractoriness to prior therapies.
Researchers observed sustained progression-free survival (PFS) in patients with stable disease (SD) or better, and the safety profile of nivolumab was considered acceptable.
“Nivolumab offers a favorable treatment outcome for patients who have relapsed disease after autologous stem cell transplant,” said Michelle Fanale, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fanale presented results from CheckMate-205 at 14-ICML. The study was sponsored by Bristol-Myers Squibb Company.
CheckMate-205 enrolled 243 adults with relapsed or refractory cHL who had undergone auto-HSCT. Patients were divided into 3 cohorts:
- Cohort A included patients who were naïve to BV (n=63)
- Cohort B included patients who received BV only after auto-HSCT (n=80)
- Cohort C included patients who received BV before and/or after auto-HSCT (n=100).
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year were to discontinue nivolumab, but they could resume treatment with the drug if they relapsed within 2 years.
Patient characteristics
The median age was 33 (range, 18-65) in cohort A, 37 (range, 18-72) in cohort B, and 32 (range, 19-69) in cohort C.
ECOG performance status was 0 for 62% of patients in cohort A, 53% in cohort B, and 50% in cohort C. The remaining patients had a performance status of 1.
The percentage of patients with stage IV disease was 38% in cohort A, 68% in cohort B, and 61% in cohort C.
The median number of prior therapies was 2 (range, 2-8) in cohort A, 4 (range, 3-15) in cohort B, and 4 (range, 2-9) in cohort C. Fifty-nine percent, 74%, and 69% of patients, respectively, had received prior radiotherapy.
The median time from diagnosis to the first dose of nivolumab was 3.1 years (range, 1.0-30.6) in cohort A, 6.2 years (range, 1.3-25.1) in cohort B, and 3.5 years (range, 1.0-24.9) in cohort C.
The median time from auto-HSCT to the first dose of nivolumab was 1.0 years (range, 0.3-18.2) in cohort A, 3.4 years (range, 0.2-19.0) in cohort B, and 1.7 years (range, 0.2-17.0) in cohort C.
Safety
The most common drug-related adverse events (AEs) were fatigue (23% any grade, 1% grade 3/4), diarrhea (15% any grade, 1% grade 3/4), infusion-related reactions (14% any grade, <1% grade 3/4), rash (12% any grade, 1% grade 3/4), nausea (10% grade 1/2), and pruritus (10% grade 1/2).
The most common drug-related serious AEs were infusion-related reactions (2% any grade, <1% grade 3/4) and pneumonitis (1% grade 1/2).
Drug-related AEs leading to treatment discontinuation were pneumonitis (2% grade 1/2) and autoimmune hepatitis (1% grade 3/4).
There were no deaths due to drug-related AEs.
Response
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
Partial response (PR) was the best response for 53% of all patients, 37% of patients in cohort A, 55% in cohort B, and 61% in cohort C.
SD was the best response for 19% of all patients, 24% of patients in cohort A, 21% in cohort B, and 15% in cohort C.
In post-hoc analyses, responses were similar irrespective of BV treatment sequence.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, and 15 months for cohort C.
The median duration of response in patients with a CR was 20 months overall and for cohorts A and B, but it was 15 months for cohort C.
The median duration of response in patients with a PR was 13 months overall, 17 months for cohort A, 11 months for cohort B, and 13 months for cohort C.
Survival
The median PFS for all patients was 15 months (range, 11-19). The median PFS was 22 months (range, 19-not reached) for patients who achieved a CR, 15 months (range, 11-19) for those who achieved a PR, and 11 months (range, 6-18) for those who had SD.
The median PFS was 18 months (range, 11-22) for patients in cohort A, 15 months (range, 11-20) for cohort B, and 12 months (range, 11-18) for cohort C.
The median overall survival (OS) has not been reached in any of the cohorts. The 12-months OS is 92% overall, 93% in cohort A, 95% in cohort B, and 90% in cohort C.
Patient status after extended follow-up
Forty percent of all patients were still on treatment after extended follow-up, as were 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
The most common reason for stopping treatment was disease progression—25% of cohort A, 28% of cohort B, and 24% of cohort C.
Patients also stopped treatment due to nivolumab-related toxicity—5% in cohort A, 11% in cohort B, and 7% in cohort C. Three percent, 1%, and 1%, respectively, stopped due to AEs unrelated to nivolumab.
Three percent of patients in cohort C stopped because they had attained the maximum clinical benefit, and 8% in cohort C completed treatment. This includes 7 patients who discontinued treatment because they were in CR for 1 year.
None of the patients in cohort A or B discontinued because they attained the maximum clinical benefit or because they completed treatment.
Eight percent of patients in cohort A, 10% in cohort B, and 17% in cohort C discontinued so they could proceed to HSCT.
Outcomes after allo-HSCT
Forty-four patients received allogeneic (allo-) HSCT after nivolumab. The median post-HSCT follow-up was 5.5 months (range, 0-19), and the median time from last dose of nivolumab to allo-HSCT was 1.6 months (range, 0.5-13.5).
At 100 days, the rate of grade 2-4 acute graft-vs-host disease (GVHD) was 27%. The rate of grade 3-4 acute GVHD was 17%, and the rate of chronic GVHD was 10%. At 6 months, the rates were 30%, 20%, and 15%, respectively.
The incidence of transplant-related mortality was 13% at 100 days and at 6 months.
“While there are risks, potentially, for acute GVHD and transplant-related mortality, these aren’t necessarily significantly different from what we’ve seen from other historical publications,” Dr Fanale said.
She cited data showing that the 100-day incidence of acute GVHD in cHL patients who underwent allo-HSCT ranges from 26% to 60%, and the incidence of transplant-related mortality in these patients ranges from 6% to 28%. ![]()
Countries with high malaria burden don’t receive research funding
A new study has revealed inequalities in malaria research funding in sub-Saharan Africa.
The study showed that some countries with a high malaria burden—such as Sierra Leone, Congo, Central African Republic, and Guinea—received little to no funding for malaria research in recent years.
However, other countries—such as Tanzania, Uganda, and Kenya—received close to $100 million in funding for malaria research.
Michael Head, PhD, of the University of Southampton in the UK, and his colleagues reported these findings in The Lancet Global Health.
“We have been able to provide a comprehensive overview of the landscape of funding for malaria in sub-Saharan Africa, a massive area where around 90% of worldwide malaria cases occur,” Dr Head said.
“We’ve shown that there are countries that are being neglected, and the global health community should reconsider strategies around resource allocation to reduce inequities and improve equality.”
For this study, Dr Head and his colleagues analyzed funding data spanning the period from 1997 to 2013. The data were sourced from 13 major public and philanthropic global health funders, as well as from funding databases.
The researchers ranked 45 countries according to the level of malaria research funding they received.
All of the countries studied received funding for malaria control, which includes investment for bed nets, public health schemes, and antimalarial drugs.
However, 8 of the 45 countries did not receive any funding related to malaria research. This included Central African Republic, Sierra Leone, and Congo—countries with a “reasonably high” malaria burden/mortality rate, according to Dr Head and his colleagues.
In all, there were 333 research awards, totaling $814.4 million. The countries that received the most research funding were Tanzania ($107.8 million), Uganda ($97.9 million), and Kenya ($92.9 million).
The 8 countries that received no research funding were Cape Verde, Botswana, Djibouti, Central African Republic, Mauritania, Congo, Chad, and Sierra Leone.
Dr Head and his colleagues suggested that the reason for the disparity in funding allocation could be, in part, due to the presence of established high-quality research infrastructure in countries such as Tanzania and Kenya, and political instability and poor healthcare infrastructures in lower-ranked nations such as Central African Republic or Sierra Leone.
“[N]ew investment in malaria research and development in these areas can encourage the development of improved health systems,” Dr Head said. “Many countries in sub-Saharan Africa simply do not have an established research infrastructure, and it is difficult for research funders to make investments in these settings.”
“Ultimately, however, there are neglected populations in these countries who suffer greatly from malaria and other diseases. Investments in health improve the wealth of a nation, and we need to be smarter with allocating limited resources to best help to reduce clear health inequalities.” ![]()
A new study has revealed inequalities in malaria research funding in sub-Saharan Africa.
The study showed that some countries with a high malaria burden—such as Sierra Leone, Congo, Central African Republic, and Guinea—received little to no funding for malaria research in recent years.
However, other countries—such as Tanzania, Uganda, and Kenya—received close to $100 million in funding for malaria research.
Michael Head, PhD, of the University of Southampton in the UK, and his colleagues reported these findings in The Lancet Global Health.
“We have been able to provide a comprehensive overview of the landscape of funding for malaria in sub-Saharan Africa, a massive area where around 90% of worldwide malaria cases occur,” Dr Head said.
“We’ve shown that there are countries that are being neglected, and the global health community should reconsider strategies around resource allocation to reduce inequities and improve equality.”
For this study, Dr Head and his colleagues analyzed funding data spanning the period from 1997 to 2013. The data were sourced from 13 major public and philanthropic global health funders, as well as from funding databases.
The researchers ranked 45 countries according to the level of malaria research funding they received.
All of the countries studied received funding for malaria control, which includes investment for bed nets, public health schemes, and antimalarial drugs.
However, 8 of the 45 countries did not receive any funding related to malaria research. This included Central African Republic, Sierra Leone, and Congo—countries with a “reasonably high” malaria burden/mortality rate, according to Dr Head and his colleagues.
In all, there were 333 research awards, totaling $814.4 million. The countries that received the most research funding were Tanzania ($107.8 million), Uganda ($97.9 million), and Kenya ($92.9 million).
The 8 countries that received no research funding were Cape Verde, Botswana, Djibouti, Central African Republic, Mauritania, Congo, Chad, and Sierra Leone.
Dr Head and his colleagues suggested that the reason for the disparity in funding allocation could be, in part, due to the presence of established high-quality research infrastructure in countries such as Tanzania and Kenya, and political instability and poor healthcare infrastructures in lower-ranked nations such as Central African Republic or Sierra Leone.
“[N]ew investment in malaria research and development in these areas can encourage the development of improved health systems,” Dr Head said. “Many countries in sub-Saharan Africa simply do not have an established research infrastructure, and it is difficult for research funders to make investments in these settings.”
“Ultimately, however, there are neglected populations in these countries who suffer greatly from malaria and other diseases. Investments in health improve the wealth of a nation, and we need to be smarter with allocating limited resources to best help to reduce clear health inequalities.” ![]()
A new study has revealed inequalities in malaria research funding in sub-Saharan Africa.
The study showed that some countries with a high malaria burden—such as Sierra Leone, Congo, Central African Republic, and Guinea—received little to no funding for malaria research in recent years.
However, other countries—such as Tanzania, Uganda, and Kenya—received close to $100 million in funding for malaria research.
Michael Head, PhD, of the University of Southampton in the UK, and his colleagues reported these findings in The Lancet Global Health.
“We have been able to provide a comprehensive overview of the landscape of funding for malaria in sub-Saharan Africa, a massive area where around 90% of worldwide malaria cases occur,” Dr Head said.
“We’ve shown that there are countries that are being neglected, and the global health community should reconsider strategies around resource allocation to reduce inequities and improve equality.”
For this study, Dr Head and his colleagues analyzed funding data spanning the period from 1997 to 2013. The data were sourced from 13 major public and philanthropic global health funders, as well as from funding databases.
The researchers ranked 45 countries according to the level of malaria research funding they received.
All of the countries studied received funding for malaria control, which includes investment for bed nets, public health schemes, and antimalarial drugs.
However, 8 of the 45 countries did not receive any funding related to malaria research. This included Central African Republic, Sierra Leone, and Congo—countries with a “reasonably high” malaria burden/mortality rate, according to Dr Head and his colleagues.
In all, there were 333 research awards, totaling $814.4 million. The countries that received the most research funding were Tanzania ($107.8 million), Uganda ($97.9 million), and Kenya ($92.9 million).
The 8 countries that received no research funding were Cape Verde, Botswana, Djibouti, Central African Republic, Mauritania, Congo, Chad, and Sierra Leone.
Dr Head and his colleagues suggested that the reason for the disparity in funding allocation could be, in part, due to the presence of established high-quality research infrastructure in countries such as Tanzania and Kenya, and political instability and poor healthcare infrastructures in lower-ranked nations such as Central African Republic or Sierra Leone.
“[N]ew investment in malaria research and development in these areas can encourage the development of improved health systems,” Dr Head said. “Many countries in sub-Saharan Africa simply do not have an established research infrastructure, and it is difficult for research funders to make investments in these settings.”
“Ultimately, however, there are neglected populations in these countries who suffer greatly from malaria and other diseases. Investments in health improve the wealth of a nation, and we need to be smarter with allocating limited resources to best help to reduce clear health inequalities.” ![]()
July 2017: Click for Credit
Here are 6 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. High-dose Oral Vitamin D3 Significantly Reduced Effects of Sunburn
To take the posttest, go to: http://bit.ly/2tmDiKc
Expires May 23, 2018
2. Women Less Likely to Be Diagnosed With Sleep Disorders
To take the posttest, go to: http://bit.ly/2rgLdne
Expires May 30, 2018
3. RA Treatment Delays Raise Risk for Long-term Disability
To take the posttest, go to: http://bit.ly/2tC0IGF
Expires May 30, 2018
4. Target Self-medication of Mood and Anxiety Symptoms
To take the posttest, go to: http://bit.ly/2vy5jel
Expires May 2, 2018
5. Two New Biomarkers for Breast Cancer Show Validity
To take the posttest, go to: http://bit.ly/2ve9H2L
Expires May 2, 2018
6. Time to Therapy for Gram-positive Bacteremia Reduced From 60 Hours to 4 Hours
To take the posttest, go to: http://bit.ly/2ssacIf
Expires May 25, 2018
Here are 6 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. High-dose Oral Vitamin D3 Significantly Reduced Effects of Sunburn
To take the posttest, go to: http://bit.ly/2tmDiKc
Expires May 23, 2018
2. Women Less Likely to Be Diagnosed With Sleep Disorders
To take the posttest, go to: http://bit.ly/2rgLdne
Expires May 30, 2018
3. RA Treatment Delays Raise Risk for Long-term Disability
To take the posttest, go to: http://bit.ly/2tC0IGF
Expires May 30, 2018
4. Target Self-medication of Mood and Anxiety Symptoms
To take the posttest, go to: http://bit.ly/2vy5jel
Expires May 2, 2018
5. Two New Biomarkers for Breast Cancer Show Validity
To take the posttest, go to: http://bit.ly/2ve9H2L
Expires May 2, 2018
6. Time to Therapy for Gram-positive Bacteremia Reduced From 60 Hours to 4 Hours
To take the posttest, go to: http://bit.ly/2ssacIf
Expires May 25, 2018
Here are 6 articles in the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. High-dose Oral Vitamin D3 Significantly Reduced Effects of Sunburn
To take the posttest, go to: http://bit.ly/2tmDiKc
Expires May 23, 2018
2. Women Less Likely to Be Diagnosed With Sleep Disorders
To take the posttest, go to: http://bit.ly/2rgLdne
Expires May 30, 2018
3. RA Treatment Delays Raise Risk for Long-term Disability
To take the posttest, go to: http://bit.ly/2tC0IGF
Expires May 30, 2018
4. Target Self-medication of Mood and Anxiety Symptoms
To take the posttest, go to: http://bit.ly/2vy5jel
Expires May 2, 2018
5. Two New Biomarkers for Breast Cancer Show Validity
To take the posttest, go to: http://bit.ly/2ve9H2L
Expires May 2, 2018
6. Time to Therapy for Gram-positive Bacteremia Reduced From 60 Hours to 4 Hours
To take the posttest, go to: http://bit.ly/2ssacIf
Expires May 25, 2018
Endo removes Opana ER from market
Even as it defended the product’s safety when appropriately used, Endo International withdrew from the market its long-acting opioid agonist Opana ER, in compliance with a June 8 Food and Drug Administration request. The company “continues to believe in the safety, efficacy, and favorable benefit-risk profile of Opana ER (oxymorphone hydrochloride extended release) when used as intended, and notes that the company has taken significant steps over the years to combat misuse and abuse,” according to a news release posted on Endo’s website. “Nevertheless, after careful consideration and consultation with the FDA following [its] June 2017 withdrawal request, the company has decided to voluntarily remove Opana ER from the market.”
In fact, the data showed a significant shift in the route of abuse of Opana ER from nasal to injection following the product’s reformulation. Injection abuse of reformulated Opana ER has been associated with a serious outbreak of HIV and hepatitis C, as well as cases of a thrombotic microangiopathy.
Endo said it will work with FDA to coordinate a smooth removal of the product, and insisted that the drug is safe and effective.
“Endo reiterates that neither the FDA’s withdrawal request nor Endo’s decision to voluntarily remove Opana ER from the market reflect a finding that the product is not safe or effective when taken as prescribed. To the contrary, Endo remains confident in the clinical research and other data demonstrating Opana ER’s safety and efficacy, as well as its favorable risk-benefit profile when used as intended in appropriate patients.”
Opana ER was first approved in 2006 for the management of moderate to severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time. It was reformulated in 2012, with the intent of making it “resistant to physical and chemical manipulation for abuse by snorting or injecting,” according to the FDA release.
On Twitter @Alz_gal
Even as it defended the product’s safety when appropriately used, Endo International withdrew from the market its long-acting opioid agonist Opana ER, in compliance with a June 8 Food and Drug Administration request. The company “continues to believe in the safety, efficacy, and favorable benefit-risk profile of Opana ER (oxymorphone hydrochloride extended release) when used as intended, and notes that the company has taken significant steps over the years to combat misuse and abuse,” according to a news release posted on Endo’s website. “Nevertheless, after careful consideration and consultation with the FDA following [its] June 2017 withdrawal request, the company has decided to voluntarily remove Opana ER from the market.”
In fact, the data showed a significant shift in the route of abuse of Opana ER from nasal to injection following the product’s reformulation. Injection abuse of reformulated Opana ER has been associated with a serious outbreak of HIV and hepatitis C, as well as cases of a thrombotic microangiopathy.
Endo said it will work with FDA to coordinate a smooth removal of the product, and insisted that the drug is safe and effective.
“Endo reiterates that neither the FDA’s withdrawal request nor Endo’s decision to voluntarily remove Opana ER from the market reflect a finding that the product is not safe or effective when taken as prescribed. To the contrary, Endo remains confident in the clinical research and other data demonstrating Opana ER’s safety and efficacy, as well as its favorable risk-benefit profile when used as intended in appropriate patients.”
Opana ER was first approved in 2006 for the management of moderate to severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time. It was reformulated in 2012, with the intent of making it “resistant to physical and chemical manipulation for abuse by snorting or injecting,” according to the FDA release.
On Twitter @Alz_gal
Even as it defended the product’s safety when appropriately used, Endo International withdrew from the market its long-acting opioid agonist Opana ER, in compliance with a June 8 Food and Drug Administration request. The company “continues to believe in the safety, efficacy, and favorable benefit-risk profile of Opana ER (oxymorphone hydrochloride extended release) when used as intended, and notes that the company has taken significant steps over the years to combat misuse and abuse,” according to a news release posted on Endo’s website. “Nevertheless, after careful consideration and consultation with the FDA following [its] June 2017 withdrawal request, the company has decided to voluntarily remove Opana ER from the market.”
In fact, the data showed a significant shift in the route of abuse of Opana ER from nasal to injection following the product’s reformulation. Injection abuse of reformulated Opana ER has been associated with a serious outbreak of HIV and hepatitis C, as well as cases of a thrombotic microangiopathy.
Endo said it will work with FDA to coordinate a smooth removal of the product, and insisted that the drug is safe and effective.
“Endo reiterates that neither the FDA’s withdrawal request nor Endo’s decision to voluntarily remove Opana ER from the market reflect a finding that the product is not safe or effective when taken as prescribed. To the contrary, Endo remains confident in the clinical research and other data demonstrating Opana ER’s safety and efficacy, as well as its favorable risk-benefit profile when used as intended in appropriate patients.”
Opana ER was first approved in 2006 for the management of moderate to severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time. It was reformulated in 2012, with the intent of making it “resistant to physical and chemical manipulation for abuse by snorting or injecting,” according to the FDA release.
On Twitter @Alz_gal