User login
Evaluating Ankylosing Spondylitis
Shingrix effective in older adults with preexisting immune-mediated disorders
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
FROM RHEUMATOLOGY
Strategic approach mitigates impact of antidrug antibodies in patients with rheumatic diseases
Monitor antidrug antibodies for their impact on drug clearance to maximize treatment outcomes in rheumatology patients treated with biologics, Niels Vande Casteele, PharmD, PhD, said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Antidrug antibodies (ADAb) are associated with impaired drug efficacy and safety, he noted. Furthermore, he indicated that when ADAb bind to the drug, they can either block the activity of the drug directly and/or may cause the formation of complexes, leading to accelerated drug clearance and reduced drug exposure.
Dr. Vande Casteele, assistant professor in the department of medicine at the University of California, San Diego, outlined factors that contribute to immunogenicity, which occurs when the body reacts to neoantigens, or when there is a breakdown in immune tolerance, he said.
Genetics can play a key role in the risk for immunogenicity, as can the route of administration, dose, treatment duration, and concomitant diseases or medications, he explained.
In addition, product-related factors including sequence variation, glycosylation, host cells, contaminants and processing impurities, formulation, and handling and storage issues can impact immunogenicity, he noted.
For example, Dr. Vande Casteele cited a study in which the proportion of infliximab-treated patients with positive ADAbs was substantially higher among those receiving the drug intravenously, compared with those receiving it subcutaneously. As for treatment dosing, data on patients treated with infliximab have shown that maintenance therapy is associated with lower rates of immunogenicity, compared to episodic therapy, he said.
In terms of genetics, Dr. Vande Casteele cited a study published in January in Gastroenterology showing the presence of the HLA-DQA1*05 allele, carried by approximately 40% of the European population, significantly increased the rate of immunogenicity to infliximab and adalimumab in patients with Crohn’s disease (hazard ratio, 1.90).
Therapeutic drug monitoring and overcoming immunogenicity
Dr. Vande Casteele also reviewed how to measure ADAbs. “Antidrug antibody units and concentrations can differ across assays,” he said.
In clinical practice, “the majority of patients at the time of secondary loss of response will present with low drug exposure, and that is when you measure antidrug antibodies,” he said.
In rheumatology patients, the presence of ADAbs against anti–tumor necrosis factor monoclonal antibodies conveys a risk for treatment discontinuation, as well as “a risk of development of hypersensitivity reactions in all immune-mediated inflammatory diseases,” Dr. Vande Casteele said.
However, “the combined use of anti–tumor necrosis factor monoclonal antibodies and disease-modifying antirheumatic drugs reduces the development of antibodies and subsequent risks,” he noted.
For therapeutic drug monitoring in patients with a secondary loss of response, “first, look at the trough concentration,” Dr. Vande Casteele said. “If it is optimal, then ADAbs are probably inconsequential.” If the trough is low or undetectable, examine ADAbs, he added. To manage ADAbs, data support the use of drug dose escalation in some cases. However, “you may be able to overcome the antidrug antibodies in some patients with dose escalation, but this is not always a cost-effective strategy in the long term,” and some patients fail a drug despite adequate drug concentration, which may mean they are failing the mechanism, and not because of pharmacokinetic-related issues, he said.
Dr. Vande Casteele cited a post hoc analysis of the TAXIT trial, published in Gastroenterology. It was the first prospective study to look at proactive therapeutic drug monitoring in patients with inflammatory bowel diseases treated with maintenance infliximab. This post hoc analysis showed that ADAbs were overcome with dose escalation in nearly 50% of patients in the lowest two ADAb quartiles at the start of the trial, and although ADAb were masked by dose escalation in the highest two quartiles, measurement with a drug-sensitive assay showed that ADAb never disappeared, he said.
Another strategy to try to overcome immunogenicity is to add an immunomodulator, Dr. Vande Casteele said. He cited a recent study published in Rheumatology showing that the effect of methotrexate was mediated through immunogenicity for immunogenic compounds such as adalimumab.
Importantly, there is a risk for immunogenicity across agents, he noted. “Patients who are antibody positive to the prior anti-TNF are at a higher risk of developing antibodies to subsequent anti-TNFs.”
Dr. Vande Casteele reported receiving research grants from R-Biopharm; grants and personal fees from Takeda and UCB; and personal fees from Alimentiv (formerly Robarts Clinical Trials), Celltrion, and Prometheus. Global Academy for Medical Education and this news organization are owned by the same parent company.
Monitor antidrug antibodies for their impact on drug clearance to maximize treatment outcomes in rheumatology patients treated with biologics, Niels Vande Casteele, PharmD, PhD, said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Antidrug antibodies (ADAb) are associated with impaired drug efficacy and safety, he noted. Furthermore, he indicated that when ADAb bind to the drug, they can either block the activity of the drug directly and/or may cause the formation of complexes, leading to accelerated drug clearance and reduced drug exposure.
Dr. Vande Casteele, assistant professor in the department of medicine at the University of California, San Diego, outlined factors that contribute to immunogenicity, which occurs when the body reacts to neoantigens, or when there is a breakdown in immune tolerance, he said.
Genetics can play a key role in the risk for immunogenicity, as can the route of administration, dose, treatment duration, and concomitant diseases or medications, he explained.
In addition, product-related factors including sequence variation, glycosylation, host cells, contaminants and processing impurities, formulation, and handling and storage issues can impact immunogenicity, he noted.
For example, Dr. Vande Casteele cited a study in which the proportion of infliximab-treated patients with positive ADAbs was substantially higher among those receiving the drug intravenously, compared with those receiving it subcutaneously. As for treatment dosing, data on patients treated with infliximab have shown that maintenance therapy is associated with lower rates of immunogenicity, compared to episodic therapy, he said.
In terms of genetics, Dr. Vande Casteele cited a study published in January in Gastroenterology showing the presence of the HLA-DQA1*05 allele, carried by approximately 40% of the European population, significantly increased the rate of immunogenicity to infliximab and adalimumab in patients with Crohn’s disease (hazard ratio, 1.90).
Therapeutic drug monitoring and overcoming immunogenicity
Dr. Vande Casteele also reviewed how to measure ADAbs. “Antidrug antibody units and concentrations can differ across assays,” he said.
In clinical practice, “the majority of patients at the time of secondary loss of response will present with low drug exposure, and that is when you measure antidrug antibodies,” he said.
In rheumatology patients, the presence of ADAbs against anti–tumor necrosis factor monoclonal antibodies conveys a risk for treatment discontinuation, as well as “a risk of development of hypersensitivity reactions in all immune-mediated inflammatory diseases,” Dr. Vande Casteele said.
However, “the combined use of anti–tumor necrosis factor monoclonal antibodies and disease-modifying antirheumatic drugs reduces the development of antibodies and subsequent risks,” he noted.
For therapeutic drug monitoring in patients with a secondary loss of response, “first, look at the trough concentration,” Dr. Vande Casteele said. “If it is optimal, then ADAbs are probably inconsequential.” If the trough is low or undetectable, examine ADAbs, he added. To manage ADAbs, data support the use of drug dose escalation in some cases. However, “you may be able to overcome the antidrug antibodies in some patients with dose escalation, but this is not always a cost-effective strategy in the long term,” and some patients fail a drug despite adequate drug concentration, which may mean they are failing the mechanism, and not because of pharmacokinetic-related issues, he said.
Dr. Vande Casteele cited a post hoc analysis of the TAXIT trial, published in Gastroenterology. It was the first prospective study to look at proactive therapeutic drug monitoring in patients with inflammatory bowel diseases treated with maintenance infliximab. This post hoc analysis showed that ADAbs were overcome with dose escalation in nearly 50% of patients in the lowest two ADAb quartiles at the start of the trial, and although ADAb were masked by dose escalation in the highest two quartiles, measurement with a drug-sensitive assay showed that ADAb never disappeared, he said.
Another strategy to try to overcome immunogenicity is to add an immunomodulator, Dr. Vande Casteele said. He cited a recent study published in Rheumatology showing that the effect of methotrexate was mediated through immunogenicity for immunogenic compounds such as adalimumab.
Importantly, there is a risk for immunogenicity across agents, he noted. “Patients who are antibody positive to the prior anti-TNF are at a higher risk of developing antibodies to subsequent anti-TNFs.”
Dr. Vande Casteele reported receiving research grants from R-Biopharm; grants and personal fees from Takeda and UCB; and personal fees from Alimentiv (formerly Robarts Clinical Trials), Celltrion, and Prometheus. Global Academy for Medical Education and this news organization are owned by the same parent company.
Monitor antidrug antibodies for their impact on drug clearance to maximize treatment outcomes in rheumatology patients treated with biologics, Niels Vande Casteele, PharmD, PhD, said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Antidrug antibodies (ADAb) are associated with impaired drug efficacy and safety, he noted. Furthermore, he indicated that when ADAb bind to the drug, they can either block the activity of the drug directly and/or may cause the formation of complexes, leading to accelerated drug clearance and reduced drug exposure.
Dr. Vande Casteele, assistant professor in the department of medicine at the University of California, San Diego, outlined factors that contribute to immunogenicity, which occurs when the body reacts to neoantigens, or when there is a breakdown in immune tolerance, he said.
Genetics can play a key role in the risk for immunogenicity, as can the route of administration, dose, treatment duration, and concomitant diseases or medications, he explained.
In addition, product-related factors including sequence variation, glycosylation, host cells, contaminants and processing impurities, formulation, and handling and storage issues can impact immunogenicity, he noted.
For example, Dr. Vande Casteele cited a study in which the proportion of infliximab-treated patients with positive ADAbs was substantially higher among those receiving the drug intravenously, compared with those receiving it subcutaneously. As for treatment dosing, data on patients treated with infliximab have shown that maintenance therapy is associated with lower rates of immunogenicity, compared to episodic therapy, he said.
In terms of genetics, Dr. Vande Casteele cited a study published in January in Gastroenterology showing the presence of the HLA-DQA1*05 allele, carried by approximately 40% of the European population, significantly increased the rate of immunogenicity to infliximab and adalimumab in patients with Crohn’s disease (hazard ratio, 1.90).
Therapeutic drug monitoring and overcoming immunogenicity
Dr. Vande Casteele also reviewed how to measure ADAbs. “Antidrug antibody units and concentrations can differ across assays,” he said.
In clinical practice, “the majority of patients at the time of secondary loss of response will present with low drug exposure, and that is when you measure antidrug antibodies,” he said.
In rheumatology patients, the presence of ADAbs against anti–tumor necrosis factor monoclonal antibodies conveys a risk for treatment discontinuation, as well as “a risk of development of hypersensitivity reactions in all immune-mediated inflammatory diseases,” Dr. Vande Casteele said.
However, “the combined use of anti–tumor necrosis factor monoclonal antibodies and disease-modifying antirheumatic drugs reduces the development of antibodies and subsequent risks,” he noted.
For therapeutic drug monitoring in patients with a secondary loss of response, “first, look at the trough concentration,” Dr. Vande Casteele said. “If it is optimal, then ADAbs are probably inconsequential.” If the trough is low or undetectable, examine ADAbs, he added. To manage ADAbs, data support the use of drug dose escalation in some cases. However, “you may be able to overcome the antidrug antibodies in some patients with dose escalation, but this is not always a cost-effective strategy in the long term,” and some patients fail a drug despite adequate drug concentration, which may mean they are failing the mechanism, and not because of pharmacokinetic-related issues, he said.
Dr. Vande Casteele cited a post hoc analysis of the TAXIT trial, published in Gastroenterology. It was the first prospective study to look at proactive therapeutic drug monitoring in patients with inflammatory bowel diseases treated with maintenance infliximab. This post hoc analysis showed that ADAbs were overcome with dose escalation in nearly 50% of patients in the lowest two ADAb quartiles at the start of the trial, and although ADAb were masked by dose escalation in the highest two quartiles, measurement with a drug-sensitive assay showed that ADAb never disappeared, he said.
Another strategy to try to overcome immunogenicity is to add an immunomodulator, Dr. Vande Casteele said. He cited a recent study published in Rheumatology showing that the effect of methotrexate was mediated through immunogenicity for immunogenic compounds such as adalimumab.
Importantly, there is a risk for immunogenicity across agents, he noted. “Patients who are antibody positive to the prior anti-TNF are at a higher risk of developing antibodies to subsequent anti-TNFs.”
Dr. Vande Casteele reported receiving research grants from R-Biopharm; grants and personal fees from Takeda and UCB; and personal fees from Alimentiv (formerly Robarts Clinical Trials), Celltrion, and Prometheus. Global Academy for Medical Education and this news organization are owned by the same parent company.
FROM PRD 2020
Dr. Len Calabrese gives advice on vaccinating adult patients with rheumatic disease
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
When it comes to preventing infection in rheumatology patients, “vaccination is the best mode of infection protection” and works synergistically with masks and hand washing, according to Leonard H. Calabrese, DO.
“Patients with rheumatic diseases have increased morbidity and mortality [from infection] and a lot of risk factors, including age, comorbidities, cytopenias, and extra-articular disease immunosuppression,” he said in a virtual presentation at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
Unfortunately, vaccination uptake remains “much lower than we would like in this country,” he said. Notably, influenza vaccination remains well below the World Health Organization target of 75%, he said.
Influenza vaccination
Flu vaccination will be even more important this year in the context of the COVID-19 pandemic, said Dr. Calabrese, professor of medicine and the RJ Fasenmyer Chair of Clinical Immunology at the Cleveland Clinic in Ohio. “For everyone who comes in with a respiratory illness, we will have to figure out whether it is flu or COVID,” he emphasized.
The Centers for Disease Control and Prevention recommendations include a detailed special considerations section for patients with immunocompromising conditions; “the notes have everything you need to know” about advising rheumatology patients, most of whom can safely receive a flu vaccine, he said.
One concern that always comes up is whether an antibody response will be suppressed based on therapy, Dr. Calabrese noted. Two major drugs with the greatest ability to reduce response are methotrexate and rituximab, he said. His tip: “Withhold methotrexate for two doses following seasonal flu vaccination.” This advice stems from a series of “practice-changing” studies by Park et al. published in 2017, 2018, and 2019 that showed benefit in withholding methotrexate for two doses following vaccination.
In the past, high-dose trivalent flu vaccines have been more expensive, and not necessarily practice changing, with studies showing varying clinical effectiveness and cost-effectiveness, Dr. Calabrese said. This year, a high-dose quadrivalent vaccine should be available that showed a 24% improvement in protection from all strains of influenza, compared with the standard vaccine in a head-to-head, randomized, controlled trial, he noted.
“All patients in rheumatology practices should get a flu vaccine,” with a 2-week hold on methotrexate following vaccination, he advised, and those aged 65 years and older should receive the high-dose quadrivalent. Younger patients on immunosuppressive therapy also might be considered for the high-dose vaccine, he said.
Pneumococcal vaccination
Dr. Calabrese also emphasized the value of pneumococcal vaccines for rheumatology patients. “The mortality for invasive disease ranges from 5% to 32%, but patients with immunocompromising conditions are at increased risk.”
Dr. Calabrese added a note on safety: Patients with cryopyrin-associated periodic syndrome (CAPS), a rare hereditary inflammatory disorder with cutaneous, neurologic, ophthalmologic, and rheumatologic manifestations, may have severe local and systemic reactions to the 23-valent polysaccharide vaccine (PPSV23), he said.
However, immunization against pneumococcal disease is safe and effective for most patients with autoimmune and inflammatory disorders regardless of their current therapy, he said. As with influenza, the CDC’s vaccination recommendations provide details for special situations, including immunocompromised individuals, he noted.
Dr. Calabrese recommended the 13-valent pneumococcal conjugate vaccine (PCV13) as soon as possible for rheumatology patients who have never been vaccinated, with follow-up doses of the 23-valent polysaccharide vaccine (PPSV23) at least 8 weeks later, and a PPSV23 booster 5 years after the first PPSV23 dose.
Protecting against shingles
When it comes to managing the varicella zoster virus (VZV) in immunocompromised patients, “prevention is preferable to treatment, as our patients are particularly vulnerable because of age and declining immunity,” Dr. Calabrese said.
Prevention is important because “once herpes zoster develops, the available treatments, including antiviral therapy, do not prevent postherpetic neuralgia in all patients,” he emphasized. “The treatments are complicated and not always effective,” he added.
The complications of zoster are well known, but recent data show an increased risk of cardiovascular disease as well, Dr. Calabrese said. “All the more reason to protect rheumatology patients from incident zoster,” he said.
Currently, the nonlive recombinant subunit zoster vaccine (Shingrix) is the preferred option for VZV vaccination according to the CDC’s Advisory Committee on Immunization Practices, Dr. Calabrese said. The CDC initially recommended its use to prevent herpes zoster and related complications in all immunocompetent adults aged 50 years and older; in an update, a C-level recommendation extends to “all patients aged 50 with or without immunosuppressive illnesses regardless of previous Zostavax exposure,” Dr. Calabrese said. “All patients on or starting [Janus] kinase inhibitors, regardless of age, should be considered” to receive the herpes zoster vaccine, he noted.
In general, promoting vaccination for rheumatology patients and for all patients is a multipronged effort that might include reminders, rewards, education, and standing orders, Dr. Calabrese said. Clinicians must continue to educate patients not only by strongly recommending the appropriate vaccines, but dispelling myths about vaccination, addressing fears, and providing current and accurate information, he said.
Dr. Calabrese disclosed relationships with AbbVie, Bristol-Myers Squibb, Crescendo, Genentech, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, Sanofi-Regeneron, and UCB.
Global Academy for Medical Education and this news organization are owned by the same parent company.
FROM PRD 2020
Trabecular bone loss may contribute to axial spondyloarthritis progression
A paper published in Seminars in Arthritis and Rheumatism reports the outcomes of a prospective observational cohort study in 245 patients with axial spondyloarthritis that sought to identify possible predictors of spinal radiographic progression of the disease.
Joon-Yong Jung, MD, PhD, and colleagues from the Catholic University of Korea, Seoul, South Korea, wrote that inflammation of the vertebrae is the first stage of progression of axial spondyloarthritis. One hypothesis is that this inflammation is associated with trabecular bone loss, which then leads to spinal instability, which in turns causes biomechanical stress on the area and the formation of new bone as syndesmophytes.
To evaluate the possible relationship between trabecular bone loss and syndesmophytes, researchers used dual-energy x-ray absorptiometry imaging of the lumbar spine, which can assess the microarchitecture of trabecular bone, as well as radiographs of the cervical and lumbar spine at 2 and 4 years of follow-up to assess the presence of syndesmophytes.
At baseline, 40% of patients had syndesmophytes, and the mean number of syndesmophytes was 3.3. A total of 11% of patients at baseline had mild trabecular bone loss, defined as trabecular bone score values between 1.230 and 1.310, and 10% had severe trabecular bone loss, with a score of 1.230 or less.
While on average patients had an increase of 1.41 syndesmophytes every 2 years during the study, patients with severe trabecular bone loss at baseline formed 1.26 more syndesmophytes every 2 years than did patients with normal trabecular bone loss score. After adjusting for variables such as disease activity and clinical factors, the authors found that both mild and severe trabecular bone loss were independently associated with progression of structural damage in the cervical and lumbar spine.
Patients with mild trabecular bone loss had a 120% greater odds of new syndesmophyte formation over the next 2 years, compared with those with normal trabecular bone loss scores, while those with severe loss had a 280% greater odds.
“The more severe the trabecular bone loss, the stronger the effect on the progression of the spine,” the authors wrote. Other factors associated with new syndesmophyte formation included higher C-reactive protein levels, longer symptom duration, smoking, and high NSAID index.
The study also pointed to an association between trabecular bone loss and modified Stoke Ankylosing Spondylitis Spinal Score. Patients with severe trabecular bone loss showed an average increase of 0.37 in their modified Stoke Ankylosing Spondylitis Spinal Score over 2 years, compared with patients with normal trabecular bone loss score at baseline, even after adjusting for confounders.
The authors commented that inflammation is hypothesized to lead to structural damage in two ways. “Inflammation-induced bone loss in the spine results in instability, another type of biomechanical stress, which then triggers a biomechanical response in an attempt to increase stability,” they wrote. Or inflammation leads to the formation of granulated repair tissue which then triggers new bone formation.
Whatever the mechanism, the authors said finding that trabecular bone loss is associated with disease progression suggests a possible use for the trabecular bone score as a practical and noninvasive means to predict spinal progression in patients with axial spondyloarthritis.
The study received no funding, and the authors said they had no conflicts of interest to declare.
SOURCE: Jung J-Y et al. Semin Arthritis Rheum. 2020;50(5):827-33.
A paper published in Seminars in Arthritis and Rheumatism reports the outcomes of a prospective observational cohort study in 245 patients with axial spondyloarthritis that sought to identify possible predictors of spinal radiographic progression of the disease.
Joon-Yong Jung, MD, PhD, and colleagues from the Catholic University of Korea, Seoul, South Korea, wrote that inflammation of the vertebrae is the first stage of progression of axial spondyloarthritis. One hypothesis is that this inflammation is associated with trabecular bone loss, which then leads to spinal instability, which in turns causes biomechanical stress on the area and the formation of new bone as syndesmophytes.
To evaluate the possible relationship between trabecular bone loss and syndesmophytes, researchers used dual-energy x-ray absorptiometry imaging of the lumbar spine, which can assess the microarchitecture of trabecular bone, as well as radiographs of the cervical and lumbar spine at 2 and 4 years of follow-up to assess the presence of syndesmophytes.
At baseline, 40% of patients had syndesmophytes, and the mean number of syndesmophytes was 3.3. A total of 11% of patients at baseline had mild trabecular bone loss, defined as trabecular bone score values between 1.230 and 1.310, and 10% had severe trabecular bone loss, with a score of 1.230 or less.
While on average patients had an increase of 1.41 syndesmophytes every 2 years during the study, patients with severe trabecular bone loss at baseline formed 1.26 more syndesmophytes every 2 years than did patients with normal trabecular bone loss score. After adjusting for variables such as disease activity and clinical factors, the authors found that both mild and severe trabecular bone loss were independently associated with progression of structural damage in the cervical and lumbar spine.
Patients with mild trabecular bone loss had a 120% greater odds of new syndesmophyte formation over the next 2 years, compared with those with normal trabecular bone loss scores, while those with severe loss had a 280% greater odds.
“The more severe the trabecular bone loss, the stronger the effect on the progression of the spine,” the authors wrote. Other factors associated with new syndesmophyte formation included higher C-reactive protein levels, longer symptom duration, smoking, and high NSAID index.
The study also pointed to an association between trabecular bone loss and modified Stoke Ankylosing Spondylitis Spinal Score. Patients with severe trabecular bone loss showed an average increase of 0.37 in their modified Stoke Ankylosing Spondylitis Spinal Score over 2 years, compared with patients with normal trabecular bone loss score at baseline, even after adjusting for confounders.
The authors commented that inflammation is hypothesized to lead to structural damage in two ways. “Inflammation-induced bone loss in the spine results in instability, another type of biomechanical stress, which then triggers a biomechanical response in an attempt to increase stability,” they wrote. Or inflammation leads to the formation of granulated repair tissue which then triggers new bone formation.
Whatever the mechanism, the authors said finding that trabecular bone loss is associated with disease progression suggests a possible use for the trabecular bone score as a practical and noninvasive means to predict spinal progression in patients with axial spondyloarthritis.
The study received no funding, and the authors said they had no conflicts of interest to declare.
SOURCE: Jung J-Y et al. Semin Arthritis Rheum. 2020;50(5):827-33.
A paper published in Seminars in Arthritis and Rheumatism reports the outcomes of a prospective observational cohort study in 245 patients with axial spondyloarthritis that sought to identify possible predictors of spinal radiographic progression of the disease.
Joon-Yong Jung, MD, PhD, and colleagues from the Catholic University of Korea, Seoul, South Korea, wrote that inflammation of the vertebrae is the first stage of progression of axial spondyloarthritis. One hypothesis is that this inflammation is associated with trabecular bone loss, which then leads to spinal instability, which in turns causes biomechanical stress on the area and the formation of new bone as syndesmophytes.
To evaluate the possible relationship between trabecular bone loss and syndesmophytes, researchers used dual-energy x-ray absorptiometry imaging of the lumbar spine, which can assess the microarchitecture of trabecular bone, as well as radiographs of the cervical and lumbar spine at 2 and 4 years of follow-up to assess the presence of syndesmophytes.
At baseline, 40% of patients had syndesmophytes, and the mean number of syndesmophytes was 3.3. A total of 11% of patients at baseline had mild trabecular bone loss, defined as trabecular bone score values between 1.230 and 1.310, and 10% had severe trabecular bone loss, with a score of 1.230 or less.
While on average patients had an increase of 1.41 syndesmophytes every 2 years during the study, patients with severe trabecular bone loss at baseline formed 1.26 more syndesmophytes every 2 years than did patients with normal trabecular bone loss score. After adjusting for variables such as disease activity and clinical factors, the authors found that both mild and severe trabecular bone loss were independently associated with progression of structural damage in the cervical and lumbar spine.
Patients with mild trabecular bone loss had a 120% greater odds of new syndesmophyte formation over the next 2 years, compared with those with normal trabecular bone loss scores, while those with severe loss had a 280% greater odds.
“The more severe the trabecular bone loss, the stronger the effect on the progression of the spine,” the authors wrote. Other factors associated with new syndesmophyte formation included higher C-reactive protein levels, longer symptom duration, smoking, and high NSAID index.
The study also pointed to an association between trabecular bone loss and modified Stoke Ankylosing Spondylitis Spinal Score. Patients with severe trabecular bone loss showed an average increase of 0.37 in their modified Stoke Ankylosing Spondylitis Spinal Score over 2 years, compared with patients with normal trabecular bone loss score at baseline, even after adjusting for confounders.
The authors commented that inflammation is hypothesized to lead to structural damage in two ways. “Inflammation-induced bone loss in the spine results in instability, another type of biomechanical stress, which then triggers a biomechanical response in an attempt to increase stability,” they wrote. Or inflammation leads to the formation of granulated repair tissue which then triggers new bone formation.
Whatever the mechanism, the authors said finding that trabecular bone loss is associated with disease progression suggests a possible use for the trabecular bone score as a practical and noninvasive means to predict spinal progression in patients with axial spondyloarthritis.
The study received no funding, and the authors said they had no conflicts of interest to declare.
SOURCE: Jung J-Y et al. Semin Arthritis Rheum. 2020;50(5):827-33.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
Ankylosing Spondylitis: Clinical Presentation
TNF inhibitors linked to inflammatory CNS events
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
Humira topped drug-revenue list for 2019
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.
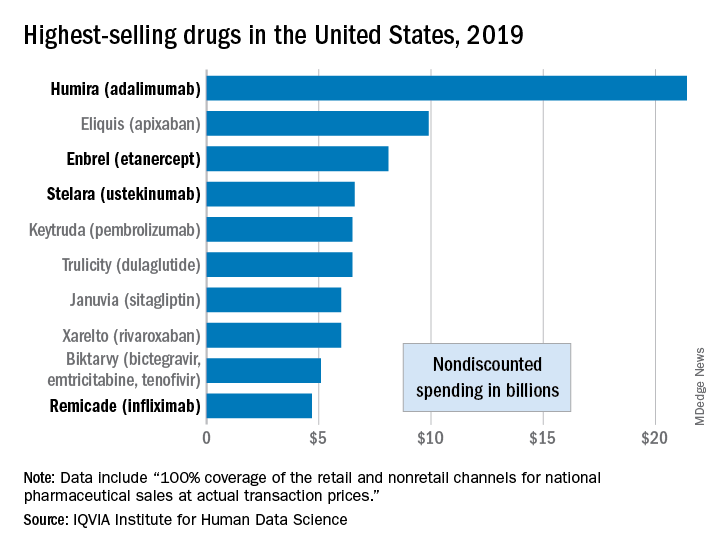
Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Obesity negatively affects axial spondyloarthritis disease activity
Obesity in adults with axial spondyloarthritis is associated with significantly worse scores on measures of disease activity when compared with normal-weight patients, according to findings from a meta-analysis of 10 studies.
Obesity or overweight could influence axial spondyloarthritis (axSpA) disease activity in several ways, including production of inflammatory mediators by adipose tissue, joint pain caused by excess weight, loss of muscle mass, and increased atherosclerosis, wrote Augusta Ortolan, MD, of the University of Padova (Italy), and colleagues.
However, “it is less clear whether overweight or obesity per se may be a cause of higher disease activity scores,” they added.
In a systematic review published in Arthritis Care & Research, the investigators identified 10 studies to use in the meta-analysis that involved associations between body mass index (BMI) and disease activity in adults with axSpA. The review included three cohort studies and seven cross-sectional studies of patients aged 18 years and older. Disease activity was assessed using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
Overall, the mean difference in BASDAI between patients with normal BMI and overweight or obese patients was –0.38 (P < .0001), and the mean difference in ASDAS between the same groups was –0.19 (P < .0001). When separated into overweight and obese categories, only the difference between normal weight and obesity remained significantly associated with differences in scores on both measures (–0.78 and –0.42, respectively; P < .0001 for both measures). The difference in scores between normal-weight and overweight/obese patients increased with BMI categories, which suggests a possible “dose-effect” relationship between fat mass and disease activity, the researchers wrote.
The study findings were limited by several factors, including the lack of randomized, controlled trials, and potential bias and inconsistency involving BMI measurements, the researchers noted.
However, “we were able to mitigate such limitations via a strict methodology and consistency in the outcomes, thus allowing us to use mean differences – instead of standardized mean differences – as outcomes, which are much easier to interpret and can be directly related to the measurement unit of the outcome,” they wrote.
The results extend data from previous studies, which “could help interpret disease activity measures and understand the difference we could expect between an axSpA patient with normal BMI and increased BMI,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Ortolan A et al. Arthritis Care Res. 2020 Aug 16. doi: 10.1002/acr.24416.
Obesity in adults with axial spondyloarthritis is associated with significantly worse scores on measures of disease activity when compared with normal-weight patients, according to findings from a meta-analysis of 10 studies.
Obesity or overweight could influence axial spondyloarthritis (axSpA) disease activity in several ways, including production of inflammatory mediators by adipose tissue, joint pain caused by excess weight, loss of muscle mass, and increased atherosclerosis, wrote Augusta Ortolan, MD, of the University of Padova (Italy), and colleagues.
However, “it is less clear whether overweight or obesity per se may be a cause of higher disease activity scores,” they added.
In a systematic review published in Arthritis Care & Research, the investigators identified 10 studies to use in the meta-analysis that involved associations between body mass index (BMI) and disease activity in adults with axSpA. The review included three cohort studies and seven cross-sectional studies of patients aged 18 years and older. Disease activity was assessed using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
Overall, the mean difference in BASDAI between patients with normal BMI and overweight or obese patients was –0.38 (P < .0001), and the mean difference in ASDAS between the same groups was –0.19 (P < .0001). When separated into overweight and obese categories, only the difference between normal weight and obesity remained significantly associated with differences in scores on both measures (–0.78 and –0.42, respectively; P < .0001 for both measures). The difference in scores between normal-weight and overweight/obese patients increased with BMI categories, which suggests a possible “dose-effect” relationship between fat mass and disease activity, the researchers wrote.
The study findings were limited by several factors, including the lack of randomized, controlled trials, and potential bias and inconsistency involving BMI measurements, the researchers noted.
However, “we were able to mitigate such limitations via a strict methodology and consistency in the outcomes, thus allowing us to use mean differences – instead of standardized mean differences – as outcomes, which are much easier to interpret and can be directly related to the measurement unit of the outcome,” they wrote.
The results extend data from previous studies, which “could help interpret disease activity measures and understand the difference we could expect between an axSpA patient with normal BMI and increased BMI,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Ortolan A et al. Arthritis Care Res. 2020 Aug 16. doi: 10.1002/acr.24416.
Obesity in adults with axial spondyloarthritis is associated with significantly worse scores on measures of disease activity when compared with normal-weight patients, according to findings from a meta-analysis of 10 studies.
Obesity or overweight could influence axial spondyloarthritis (axSpA) disease activity in several ways, including production of inflammatory mediators by adipose tissue, joint pain caused by excess weight, loss of muscle mass, and increased atherosclerosis, wrote Augusta Ortolan, MD, of the University of Padova (Italy), and colleagues.
However, “it is less clear whether overweight or obesity per se may be a cause of higher disease activity scores,” they added.
In a systematic review published in Arthritis Care & Research, the investigators identified 10 studies to use in the meta-analysis that involved associations between body mass index (BMI) and disease activity in adults with axSpA. The review included three cohort studies and seven cross-sectional studies of patients aged 18 years and older. Disease activity was assessed using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
Overall, the mean difference in BASDAI between patients with normal BMI and overweight or obese patients was –0.38 (P < .0001), and the mean difference in ASDAS between the same groups was –0.19 (P < .0001). When separated into overweight and obese categories, only the difference between normal weight and obesity remained significantly associated with differences in scores on both measures (–0.78 and –0.42, respectively; P < .0001 for both measures). The difference in scores between normal-weight and overweight/obese patients increased with BMI categories, which suggests a possible “dose-effect” relationship between fat mass and disease activity, the researchers wrote.
The study findings were limited by several factors, including the lack of randomized, controlled trials, and potential bias and inconsistency involving BMI measurements, the researchers noted.
However, “we were able to mitigate such limitations via a strict methodology and consistency in the outcomes, thus allowing us to use mean differences – instead of standardized mean differences – as outcomes, which are much easier to interpret and can be directly related to the measurement unit of the outcome,” they wrote.
The results extend data from previous studies, which “could help interpret disease activity measures and understand the difference we could expect between an axSpA patient with normal BMI and increased BMI,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Ortolan A et al. Arthritis Care Res. 2020 Aug 16. doi: 10.1002/acr.24416.
FROM ARTHRITIS CARE & RESEARCH