User login
Children and COVID: Weekly cases can’t sustain downward trend
New COVID-19 cases in children inched up in late October, just 1 week after dipping to their lowest level in more than a year, and some measures of pediatric emergency visits and hospital admissions rose as well.
There was an 8% increase in the number of cases for the week of Oct. 21-27, compared with the previous week, but this week’s total was still below 25,000, and the overall trend since the beginning of September is still one of decline, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.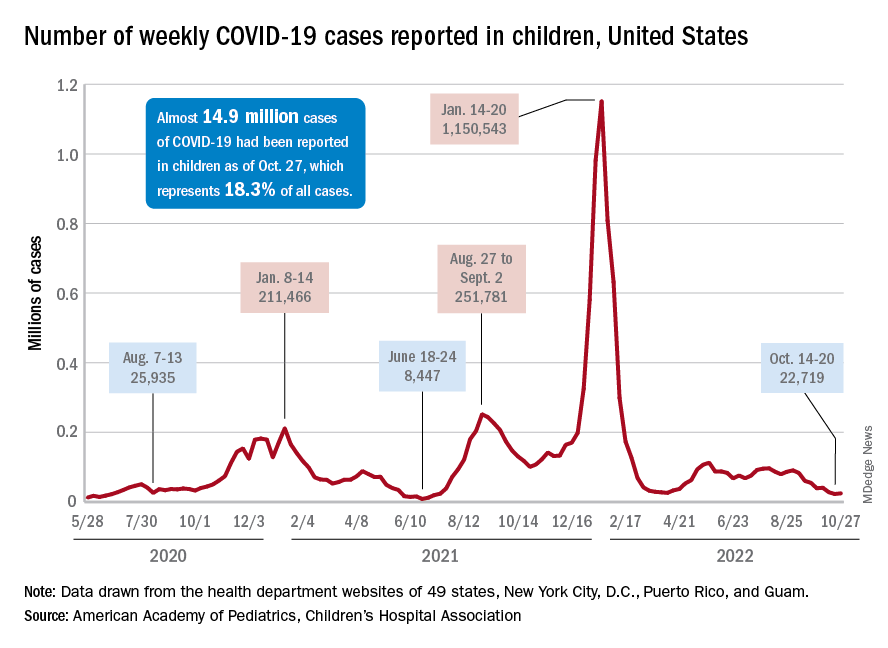
A similar increase can be seen for hospitalizations with confirmed COVID. The rate for children aged 0-17 years fell from 0.44 admissions per 100,000 population at the end of August to 0.16 per 100,000 on Oct. 23. Hospitalizations have since ticked up to 0.17 per 100,000, according to the Centers for Disease Control and Prevention.
Emergency department visits with diagnosed COVID among children aged 16-17 years, as a percentage of all ED visits, rose from 0.6% on Oct. 21 to 0.8% on Oct. 26. ED visits for 12- to 15-year-olds rose from 0.6% to 0.7% at about the same time, with both increases coming after declines that started in late August. No such increase has occurred yet among children aged 0-11 years, the CDC reported on its COVID Data Tracker.
One small milestone reached in the past week involved the proportion of all COVID cases that have occurred in children. The total number of child cases as of Oct. 27 was almost 14.9 million, which represents 18.3% of cases in all Americans, according to the AAP and CHA. That figure had been sitting at 18.4% since mid-August after reaching as high as 19.0% during the spring.
The CDC puts total COVID-related hospital admissions for children aged 0-17 at 163,588 since Aug. 1, 2020, which is 3.0% of all U.S. admissions. Total pediatric deaths number 1,843, or just about 0.2% of all COVID-related fatalities since the start of the pandemic, the CDC data show.
The latest vaccination figures show that 71.3% of children aged 12-17 years have received at least one dose, as have 38.8% of 5- to 11-year-olds, 8.4% of 2- to 4-year-olds, and 5.5% of those under age 2. Full vaccination by age group looks like this: 60.9% (12-17 years), 31.7% (5-11 years), 3.7% (2-4 years), and 2.1% (<2 years), the CDC reported. Almost 30% of children aged 12-17 have gotten a first booster dose, as have 16% of 5- to 11-year-olds.
New COVID-19 cases in children inched up in late October, just 1 week after dipping to their lowest level in more than a year, and some measures of pediatric emergency visits and hospital admissions rose as well.
There was an 8% increase in the number of cases for the week of Oct. 21-27, compared with the previous week, but this week’s total was still below 25,000, and the overall trend since the beginning of September is still one of decline, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
A similar increase can be seen for hospitalizations with confirmed COVID. The rate for children aged 0-17 years fell from 0.44 admissions per 100,000 population at the end of August to 0.16 per 100,000 on Oct. 23. Hospitalizations have since ticked up to 0.17 per 100,000, according to the Centers for Disease Control and Prevention.
Emergency department visits with diagnosed COVID among children aged 16-17 years, as a percentage of all ED visits, rose from 0.6% on Oct. 21 to 0.8% on Oct. 26. ED visits for 12- to 15-year-olds rose from 0.6% to 0.7% at about the same time, with both increases coming after declines that started in late August. No such increase has occurred yet among children aged 0-11 years, the CDC reported on its COVID Data Tracker.
One small milestone reached in the past week involved the proportion of all COVID cases that have occurred in children. The total number of child cases as of Oct. 27 was almost 14.9 million, which represents 18.3% of cases in all Americans, according to the AAP and CHA. That figure had been sitting at 18.4% since mid-August after reaching as high as 19.0% during the spring.
The CDC puts total COVID-related hospital admissions for children aged 0-17 at 163,588 since Aug. 1, 2020, which is 3.0% of all U.S. admissions. Total pediatric deaths number 1,843, or just about 0.2% of all COVID-related fatalities since the start of the pandemic, the CDC data show.
The latest vaccination figures show that 71.3% of children aged 12-17 years have received at least one dose, as have 38.8% of 5- to 11-year-olds, 8.4% of 2- to 4-year-olds, and 5.5% of those under age 2. Full vaccination by age group looks like this: 60.9% (12-17 years), 31.7% (5-11 years), 3.7% (2-4 years), and 2.1% (<2 years), the CDC reported. Almost 30% of children aged 12-17 have gotten a first booster dose, as have 16% of 5- to 11-year-olds.
New COVID-19 cases in children inched up in late October, just 1 week after dipping to their lowest level in more than a year, and some measures of pediatric emergency visits and hospital admissions rose as well.
There was an 8% increase in the number of cases for the week of Oct. 21-27, compared with the previous week, but this week’s total was still below 25,000, and the overall trend since the beginning of September is still one of decline, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
A similar increase can be seen for hospitalizations with confirmed COVID. The rate for children aged 0-17 years fell from 0.44 admissions per 100,000 population at the end of August to 0.16 per 100,000 on Oct. 23. Hospitalizations have since ticked up to 0.17 per 100,000, according to the Centers for Disease Control and Prevention.
Emergency department visits with diagnosed COVID among children aged 16-17 years, as a percentage of all ED visits, rose from 0.6% on Oct. 21 to 0.8% on Oct. 26. ED visits for 12- to 15-year-olds rose from 0.6% to 0.7% at about the same time, with both increases coming after declines that started in late August. No such increase has occurred yet among children aged 0-11 years, the CDC reported on its COVID Data Tracker.
One small milestone reached in the past week involved the proportion of all COVID cases that have occurred in children. The total number of child cases as of Oct. 27 was almost 14.9 million, which represents 18.3% of cases in all Americans, according to the AAP and CHA. That figure had been sitting at 18.4% since mid-August after reaching as high as 19.0% during the spring.
The CDC puts total COVID-related hospital admissions for children aged 0-17 at 163,588 since Aug. 1, 2020, which is 3.0% of all U.S. admissions. Total pediatric deaths number 1,843, or just about 0.2% of all COVID-related fatalities since the start of the pandemic, the CDC data show.
The latest vaccination figures show that 71.3% of children aged 12-17 years have received at least one dose, as have 38.8% of 5- to 11-year-olds, 8.4% of 2- to 4-year-olds, and 5.5% of those under age 2. Full vaccination by age group looks like this: 60.9% (12-17 years), 31.7% (5-11 years), 3.7% (2-4 years), and 2.1% (<2 years), the CDC reported. Almost 30% of children aged 12-17 have gotten a first booster dose, as have 16% of 5- to 11-year-olds.
Oral FMT on par with colonic FMT for recurrent C. difficile
A real-world analysis confirms that fecal microbiota transplantation (FMT) is highly effective for recurrent Clostridioides difficile infection (rCDI) – and there is no difference between delivery by capsule (cap-FMT) and colonoscopy (colo-FMT).
“We present one of the largest cohorts involving people who received capsule FMT. Byron Vaughn, MD, with the division of gastroenterology, hepatology, and nutrition, University of Minnesota, Minneapolis, said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
The Food and Drug Administration allows FMT to be used for patients who have failed standard treatment for rCDI under a policy of enforcement discretion.
The past decade has seen an increase in the use of FMT in clinical practice, owing to an increase in cases of rCDI after failure of standard antibiotic therapy.
Unlike antibiotics, which perpetuate and worsen intestinal dysbiosis, FMT restores the diversity and function of host microbiota, effectively breaking the cycle of rCDI, the authors of the study noted. But it’s been unclear whether the efficacy and safety of FMT vary by route of administration.
Effective without procedural risks
To investigate, Dr. Vaughn and colleagues evaluated clinical outcomes and adverse events in 170 patients with rCDI who underwent cap-FMT and 96 peers who underwent colo-FMT.
FMT was performed using one of two standardized formulations of microbiota manufactured by the University of Minnesota microbiota therapeutics program: freeze-dried/encapsulated or frozen-thawed/liquid.
Overall, the cure rates of CDI were 86% at 1 month and 81% at 2 months. There was no statistically significant difference at either time between cap-FMT and colo-FMT.
The 1-month cure rate was 84% with cap-FMT and 91% with colo-FMT; at 2 months, the cure rates were 81% and 83%, respectively.
Cap-FMT has a safety and effectiveness profile similar to that of colo-FMT, without the procedural risks of colonoscopy, the researchers concluded.
They cautioned that, although FMT is highly effective overall, patient selection is a key factor to optimizing FMT success.
Older age and hemodialysis were associated with FMT failure by 2 months on multivariate logistic regression.
“These risk factors can help determine if a patient should receive FMT or an alternative therapy for rCDI. This is not to say FMT should be avoided in older patients or those on dialysis, but clinicians should be aware of these associations in light of other options for rCDI,” Dr. Vaughn said.
Confirming prior studies, antibiotic use after FMT was a major factor in its failure. Patient selection for FMT should include an assessment of the potential need for antibiotics after transplant, the researchers noted.
One serious adverse event (aspiration pneumonia) was related to colonoscopy; otherwise, no new safety signals were identified.
As reported in other studies, changes in bowel function, including diarrhea, constipation, gas, and bloating were common, although it’s tough to disentangle gastrointestinal symptoms related to FMT from those after CDI, the researchers said. Importantly, no transmission of an infectious agent related to FMT was identified.
Two good options
The researchers said their findings are “highly generalizable” because the population reflects all FMT use by participating institutions and contains a mix of academic centers and private practices.
Many patients included in the study would not have been eligible for a clinical trial, owing to their having many comorbid conditions, including immune compromise and inflammatory bowel disease, the authors noted.
“FMT is recommended by major gastroenterology and infectious disease society guidelines,” Dr. Vaughn said. “Our group, and others, have consistently found strategies that incorporate FMT as cost-effective strategies for treating rCDI.”
However, lack of access to FMT products often is a barrier to treatment, he said.
“A stool banking model, similar to the nonprofit blood banking model, may be a useful solution to ensure equitable access to FMT to all who need it,” Dr. Vaughn added.
Reached for comment, Majdi Osman, MD, MPH, told this news organization that the study is valuable, “as it nicely shows in a real-world setting that capsules and colonoscopy are good options for patients who need this.”
Dr. Osman is chief medical officer of OpenBiome, a nonprofit organization that operates a public stool bank and is the major FMT source in the United States. The organization has provided over 63,000 FMT treatments to over 1,200 hospitals in the United States.
“FMT has become standard of care for patients who failed antibiotic therapy, and certainly is being used widely as a treatment option for these patients who have often run out of existing options,” Dr. Osman said.
Support for the study was provided by a donation from Achieving Cures Together, a nonprofit organization dedicated to advancing microbiome-based research. Dr. Vaughn receives grant support from Takeda, Roche, Celgene, and Diasorin and has received consulting fees from Prometheus and AbbVie. Dr. Osman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A real-world analysis confirms that fecal microbiota transplantation (FMT) is highly effective for recurrent Clostridioides difficile infection (rCDI) – and there is no difference between delivery by capsule (cap-FMT) and colonoscopy (colo-FMT).
“We present one of the largest cohorts involving people who received capsule FMT. Byron Vaughn, MD, with the division of gastroenterology, hepatology, and nutrition, University of Minnesota, Minneapolis, said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
The Food and Drug Administration allows FMT to be used for patients who have failed standard treatment for rCDI under a policy of enforcement discretion.
The past decade has seen an increase in the use of FMT in clinical practice, owing to an increase in cases of rCDI after failure of standard antibiotic therapy.
Unlike antibiotics, which perpetuate and worsen intestinal dysbiosis, FMT restores the diversity and function of host microbiota, effectively breaking the cycle of rCDI, the authors of the study noted. But it’s been unclear whether the efficacy and safety of FMT vary by route of administration.
Effective without procedural risks
To investigate, Dr. Vaughn and colleagues evaluated clinical outcomes and adverse events in 170 patients with rCDI who underwent cap-FMT and 96 peers who underwent colo-FMT.
FMT was performed using one of two standardized formulations of microbiota manufactured by the University of Minnesota microbiota therapeutics program: freeze-dried/encapsulated or frozen-thawed/liquid.
Overall, the cure rates of CDI were 86% at 1 month and 81% at 2 months. There was no statistically significant difference at either time between cap-FMT and colo-FMT.
The 1-month cure rate was 84% with cap-FMT and 91% with colo-FMT; at 2 months, the cure rates were 81% and 83%, respectively.
Cap-FMT has a safety and effectiveness profile similar to that of colo-FMT, without the procedural risks of colonoscopy, the researchers concluded.
They cautioned that, although FMT is highly effective overall, patient selection is a key factor to optimizing FMT success.
Older age and hemodialysis were associated with FMT failure by 2 months on multivariate logistic regression.
“These risk factors can help determine if a patient should receive FMT or an alternative therapy for rCDI. This is not to say FMT should be avoided in older patients or those on dialysis, but clinicians should be aware of these associations in light of other options for rCDI,” Dr. Vaughn said.
Confirming prior studies, antibiotic use after FMT was a major factor in its failure. Patient selection for FMT should include an assessment of the potential need for antibiotics after transplant, the researchers noted.
One serious adverse event (aspiration pneumonia) was related to colonoscopy; otherwise, no new safety signals were identified.
As reported in other studies, changes in bowel function, including diarrhea, constipation, gas, and bloating were common, although it’s tough to disentangle gastrointestinal symptoms related to FMT from those after CDI, the researchers said. Importantly, no transmission of an infectious agent related to FMT was identified.
Two good options
The researchers said their findings are “highly generalizable” because the population reflects all FMT use by participating institutions and contains a mix of academic centers and private practices.
Many patients included in the study would not have been eligible for a clinical trial, owing to their having many comorbid conditions, including immune compromise and inflammatory bowel disease, the authors noted.
“FMT is recommended by major gastroenterology and infectious disease society guidelines,” Dr. Vaughn said. “Our group, and others, have consistently found strategies that incorporate FMT as cost-effective strategies for treating rCDI.”
However, lack of access to FMT products often is a barrier to treatment, he said.
“A stool banking model, similar to the nonprofit blood banking model, may be a useful solution to ensure equitable access to FMT to all who need it,” Dr. Vaughn added.
Reached for comment, Majdi Osman, MD, MPH, told this news organization that the study is valuable, “as it nicely shows in a real-world setting that capsules and colonoscopy are good options for patients who need this.”
Dr. Osman is chief medical officer of OpenBiome, a nonprofit organization that operates a public stool bank and is the major FMT source in the United States. The organization has provided over 63,000 FMT treatments to over 1,200 hospitals in the United States.
“FMT has become standard of care for patients who failed antibiotic therapy, and certainly is being used widely as a treatment option for these patients who have often run out of existing options,” Dr. Osman said.
Support for the study was provided by a donation from Achieving Cures Together, a nonprofit organization dedicated to advancing microbiome-based research. Dr. Vaughn receives grant support from Takeda, Roche, Celgene, and Diasorin and has received consulting fees from Prometheus and AbbVie. Dr. Osman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A real-world analysis confirms that fecal microbiota transplantation (FMT) is highly effective for recurrent Clostridioides difficile infection (rCDI) – and there is no difference between delivery by capsule (cap-FMT) and colonoscopy (colo-FMT).
“We present one of the largest cohorts involving people who received capsule FMT. Byron Vaughn, MD, with the division of gastroenterology, hepatology, and nutrition, University of Minnesota, Minneapolis, said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
The Food and Drug Administration allows FMT to be used for patients who have failed standard treatment for rCDI under a policy of enforcement discretion.
The past decade has seen an increase in the use of FMT in clinical practice, owing to an increase in cases of rCDI after failure of standard antibiotic therapy.
Unlike antibiotics, which perpetuate and worsen intestinal dysbiosis, FMT restores the diversity and function of host microbiota, effectively breaking the cycle of rCDI, the authors of the study noted. But it’s been unclear whether the efficacy and safety of FMT vary by route of administration.
Effective without procedural risks
To investigate, Dr. Vaughn and colleagues evaluated clinical outcomes and adverse events in 170 patients with rCDI who underwent cap-FMT and 96 peers who underwent colo-FMT.
FMT was performed using one of two standardized formulations of microbiota manufactured by the University of Minnesota microbiota therapeutics program: freeze-dried/encapsulated or frozen-thawed/liquid.
Overall, the cure rates of CDI were 86% at 1 month and 81% at 2 months. There was no statistically significant difference at either time between cap-FMT and colo-FMT.
The 1-month cure rate was 84% with cap-FMT and 91% with colo-FMT; at 2 months, the cure rates were 81% and 83%, respectively.
Cap-FMT has a safety and effectiveness profile similar to that of colo-FMT, without the procedural risks of colonoscopy, the researchers concluded.
They cautioned that, although FMT is highly effective overall, patient selection is a key factor to optimizing FMT success.
Older age and hemodialysis were associated with FMT failure by 2 months on multivariate logistic regression.
“These risk factors can help determine if a patient should receive FMT or an alternative therapy for rCDI. This is not to say FMT should be avoided in older patients or those on dialysis, but clinicians should be aware of these associations in light of other options for rCDI,” Dr. Vaughn said.
Confirming prior studies, antibiotic use after FMT was a major factor in its failure. Patient selection for FMT should include an assessment of the potential need for antibiotics after transplant, the researchers noted.
One serious adverse event (aspiration pneumonia) was related to colonoscopy; otherwise, no new safety signals were identified.
As reported in other studies, changes in bowel function, including diarrhea, constipation, gas, and bloating were common, although it’s tough to disentangle gastrointestinal symptoms related to FMT from those after CDI, the researchers said. Importantly, no transmission of an infectious agent related to FMT was identified.
Two good options
The researchers said their findings are “highly generalizable” because the population reflects all FMT use by participating institutions and contains a mix of academic centers and private practices.
Many patients included in the study would not have been eligible for a clinical trial, owing to their having many comorbid conditions, including immune compromise and inflammatory bowel disease, the authors noted.
“FMT is recommended by major gastroenterology and infectious disease society guidelines,” Dr. Vaughn said. “Our group, and others, have consistently found strategies that incorporate FMT as cost-effective strategies for treating rCDI.”
However, lack of access to FMT products often is a barrier to treatment, he said.
“A stool banking model, similar to the nonprofit blood banking model, may be a useful solution to ensure equitable access to FMT to all who need it,” Dr. Vaughn added.
Reached for comment, Majdi Osman, MD, MPH, told this news organization that the study is valuable, “as it nicely shows in a real-world setting that capsules and colonoscopy are good options for patients who need this.”
Dr. Osman is chief medical officer of OpenBiome, a nonprofit organization that operates a public stool bank and is the major FMT source in the United States. The organization has provided over 63,000 FMT treatments to over 1,200 hospitals in the United States.
“FMT has become standard of care for patients who failed antibiotic therapy, and certainly is being used widely as a treatment option for these patients who have often run out of existing options,” Dr. Osman said.
Support for the study was provided by a donation from Achieving Cures Together, a nonprofit organization dedicated to advancing microbiome-based research. Dr. Vaughn receives grant support from Takeda, Roche, Celgene, and Diasorin and has received consulting fees from Prometheus and AbbVie. Dr. Osman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Recurrent urinary tract infections: What’s good prophylaxis?
For those affected, recurrent urinary tract infections (UTIs) are sometimes stressful. However, even an informative discussion about risk factors and the imparting of behavioral recommendations can be very helpful for many women. Antibiotic prophylaxis should only be considered once all nonantibiotic therapy options have been exhausted.
One in seven women suffers at least once a year from cystitis. Around a third of those women develop a further urinary tract infection 6-12 months after the first infection. A urinary tract infection is classified as recurrent if two symptomatic episodes have occurred within the last 6 months or if three episodes have occurred within the last 12 months.
There are many different approaches to reducing the recurrence rate of urinary tract infections, Daniel Klussmann and Florian Wagenlehner, MD, of the department and outpatient clinic for urology at the University of Giessen (Germany) wrote in DMW Klinischer Fortschritt. Aside from general information and advice,
Fluids and D-mannose
An individual consultation discussion is the most important nonantibiotic strategy. Studies have shown that this strategy alone can lower the frequency of recurrent UTIs. According to the authors, special education programs on the causes and behavioral measures are especially helpful. Included in these programs is the recommendation to drink a sufficient, but not excessive, amount of fluids: approximately 1.5 liters per day. In one randomized study, this level of consumption halved UTI frequency. However, drinking an excessive amount of fluids should also be avoided, otherwise the antimicrobial peptides present in the urine become overly diluted.
The regular consumption of fruit juice, especially of that from berries, is also beneficial, according to the authors. However, study results on long-term prevention using cranberry products are inconsistent, and they are not recommended in the updated guideline. Like cranberries, D-mannose also inhibits the fimbriae of the Escherichia coli bacteria and therefore the bacteria’s ability to bind to the bladder epithelium. The authors cite a study in which, following the intake of 2 g of D-mannose dissolved in a glass of water every day, the rate of urinary tract infections dropped significantly, compared with consumption of placebo.
Additional recommendations in the S3 guideline include various phytotherapeutic products such as bearberry leaves, nasturtium herb, or horseradish root, although studies on the comparability of phytotherapeutic agents are very difficult to execute, the authors conceded.
It is already known that there is a positive correlation (by a factor of 60) between the recurrence rate of UTIs and the frequency of sexual intercourse. Even with contraceptive methods (such as vaginal suppositories, diaphragms or condoms coated with spermicide, and intrauterine devices), the risk of urinary tract infections increases by a factor of 2-14. Sexual abstinence, even if temporary, can be a remedy. Evidence for the recommendation to urinate immediately after coitus is contradictory in the literature, however. Excessive intimate hygiene clearly damages the local protective environment.
Estrogen substitution beneficial
For postmenopausal women, there is also the option of local estriol substitution (0.5 mg/day) as another nonantibiotic method of prophylaxis. This treatment serves as therapy for vaginal atrophy and reduces both vaginal colonization with uropathogens and the vaginal pH level. The authors cite Scandinavian studies that detected no increase in the risk of breast cancer from the local application of estriol.
Furthermore, the current guidelines recommend oral immunostimulation with bacterial cell wall components from uropathogenic strains of E. coli (OM-89, Uro-Vaxom). The authors reported on two meta-studies in which the average recurrence rate was reduced by 39%, compared with placebo. In addition, the treatment time for breakthrough infections decreased significantly, and prevention with OM-89 could even be started during acute therapy. Also recommended is parenteral immunostimulation with inactivated pathogens (StroVac). Acupuncture as cutaneous immunostimulation has also displayed a positive protective effect.
Only when nonantibiotic therapy fails and the patient is under a high amount of psychological strain should antibiotic prophylaxis be initiated, according to the authors. A period of 3-6 months should be the target here. When choosing an antibiotic and before starting therapy, the corresponding pathogen should be confirmed through a urine culture, and resistance testing should be performed. On the other hand, single-use, postcoital antibiotic prevention could be an alternative, particularly for women in whom a correlation between recurrent UTIs and sexual intercourse has been suspected, the authors wrote.
This article was translated from Univadis Germany. A version appeared on Medscape.com.
For those affected, recurrent urinary tract infections (UTIs) are sometimes stressful. However, even an informative discussion about risk factors and the imparting of behavioral recommendations can be very helpful for many women. Antibiotic prophylaxis should only be considered once all nonantibiotic therapy options have been exhausted.
One in seven women suffers at least once a year from cystitis. Around a third of those women develop a further urinary tract infection 6-12 months after the first infection. A urinary tract infection is classified as recurrent if two symptomatic episodes have occurred within the last 6 months or if three episodes have occurred within the last 12 months.
There are many different approaches to reducing the recurrence rate of urinary tract infections, Daniel Klussmann and Florian Wagenlehner, MD, of the department and outpatient clinic for urology at the University of Giessen (Germany) wrote in DMW Klinischer Fortschritt. Aside from general information and advice,
Fluids and D-mannose
An individual consultation discussion is the most important nonantibiotic strategy. Studies have shown that this strategy alone can lower the frequency of recurrent UTIs. According to the authors, special education programs on the causes and behavioral measures are especially helpful. Included in these programs is the recommendation to drink a sufficient, but not excessive, amount of fluids: approximately 1.5 liters per day. In one randomized study, this level of consumption halved UTI frequency. However, drinking an excessive amount of fluids should also be avoided, otherwise the antimicrobial peptides present in the urine become overly diluted.
The regular consumption of fruit juice, especially of that from berries, is also beneficial, according to the authors. However, study results on long-term prevention using cranberry products are inconsistent, and they are not recommended in the updated guideline. Like cranberries, D-mannose also inhibits the fimbriae of the Escherichia coli bacteria and therefore the bacteria’s ability to bind to the bladder epithelium. The authors cite a study in which, following the intake of 2 g of D-mannose dissolved in a glass of water every day, the rate of urinary tract infections dropped significantly, compared with consumption of placebo.
Additional recommendations in the S3 guideline include various phytotherapeutic products such as bearberry leaves, nasturtium herb, or horseradish root, although studies on the comparability of phytotherapeutic agents are very difficult to execute, the authors conceded.
It is already known that there is a positive correlation (by a factor of 60) between the recurrence rate of UTIs and the frequency of sexual intercourse. Even with contraceptive methods (such as vaginal suppositories, diaphragms or condoms coated with spermicide, and intrauterine devices), the risk of urinary tract infections increases by a factor of 2-14. Sexual abstinence, even if temporary, can be a remedy. Evidence for the recommendation to urinate immediately after coitus is contradictory in the literature, however. Excessive intimate hygiene clearly damages the local protective environment.
Estrogen substitution beneficial
For postmenopausal women, there is also the option of local estriol substitution (0.5 mg/day) as another nonantibiotic method of prophylaxis. This treatment serves as therapy for vaginal atrophy and reduces both vaginal colonization with uropathogens and the vaginal pH level. The authors cite Scandinavian studies that detected no increase in the risk of breast cancer from the local application of estriol.
Furthermore, the current guidelines recommend oral immunostimulation with bacterial cell wall components from uropathogenic strains of E. coli (OM-89, Uro-Vaxom). The authors reported on two meta-studies in which the average recurrence rate was reduced by 39%, compared with placebo. In addition, the treatment time for breakthrough infections decreased significantly, and prevention with OM-89 could even be started during acute therapy. Also recommended is parenteral immunostimulation with inactivated pathogens (StroVac). Acupuncture as cutaneous immunostimulation has also displayed a positive protective effect.
Only when nonantibiotic therapy fails and the patient is under a high amount of psychological strain should antibiotic prophylaxis be initiated, according to the authors. A period of 3-6 months should be the target here. When choosing an antibiotic and before starting therapy, the corresponding pathogen should be confirmed through a urine culture, and resistance testing should be performed. On the other hand, single-use, postcoital antibiotic prevention could be an alternative, particularly for women in whom a correlation between recurrent UTIs and sexual intercourse has been suspected, the authors wrote.
This article was translated from Univadis Germany. A version appeared on Medscape.com.
For those affected, recurrent urinary tract infections (UTIs) are sometimes stressful. However, even an informative discussion about risk factors and the imparting of behavioral recommendations can be very helpful for many women. Antibiotic prophylaxis should only be considered once all nonantibiotic therapy options have been exhausted.
One in seven women suffers at least once a year from cystitis. Around a third of those women develop a further urinary tract infection 6-12 months after the first infection. A urinary tract infection is classified as recurrent if two symptomatic episodes have occurred within the last 6 months or if three episodes have occurred within the last 12 months.
There are many different approaches to reducing the recurrence rate of urinary tract infections, Daniel Klussmann and Florian Wagenlehner, MD, of the department and outpatient clinic for urology at the University of Giessen (Germany) wrote in DMW Klinischer Fortschritt. Aside from general information and advice,
Fluids and D-mannose
An individual consultation discussion is the most important nonantibiotic strategy. Studies have shown that this strategy alone can lower the frequency of recurrent UTIs. According to the authors, special education programs on the causes and behavioral measures are especially helpful. Included in these programs is the recommendation to drink a sufficient, but not excessive, amount of fluids: approximately 1.5 liters per day. In one randomized study, this level of consumption halved UTI frequency. However, drinking an excessive amount of fluids should also be avoided, otherwise the antimicrobial peptides present in the urine become overly diluted.
The regular consumption of fruit juice, especially of that from berries, is also beneficial, according to the authors. However, study results on long-term prevention using cranberry products are inconsistent, and they are not recommended in the updated guideline. Like cranberries, D-mannose also inhibits the fimbriae of the Escherichia coli bacteria and therefore the bacteria’s ability to bind to the bladder epithelium. The authors cite a study in which, following the intake of 2 g of D-mannose dissolved in a glass of water every day, the rate of urinary tract infections dropped significantly, compared with consumption of placebo.
Additional recommendations in the S3 guideline include various phytotherapeutic products such as bearberry leaves, nasturtium herb, or horseradish root, although studies on the comparability of phytotherapeutic agents are very difficult to execute, the authors conceded.
It is already known that there is a positive correlation (by a factor of 60) between the recurrence rate of UTIs and the frequency of sexual intercourse. Even with contraceptive methods (such as vaginal suppositories, diaphragms or condoms coated with spermicide, and intrauterine devices), the risk of urinary tract infections increases by a factor of 2-14. Sexual abstinence, even if temporary, can be a remedy. Evidence for the recommendation to urinate immediately after coitus is contradictory in the literature, however. Excessive intimate hygiene clearly damages the local protective environment.
Estrogen substitution beneficial
For postmenopausal women, there is also the option of local estriol substitution (0.5 mg/day) as another nonantibiotic method of prophylaxis. This treatment serves as therapy for vaginal atrophy and reduces both vaginal colonization with uropathogens and the vaginal pH level. The authors cite Scandinavian studies that detected no increase in the risk of breast cancer from the local application of estriol.
Furthermore, the current guidelines recommend oral immunostimulation with bacterial cell wall components from uropathogenic strains of E. coli (OM-89, Uro-Vaxom). The authors reported on two meta-studies in which the average recurrence rate was reduced by 39%, compared with placebo. In addition, the treatment time for breakthrough infections decreased significantly, and prevention with OM-89 could even be started during acute therapy. Also recommended is parenteral immunostimulation with inactivated pathogens (StroVac). Acupuncture as cutaneous immunostimulation has also displayed a positive protective effect.
Only when nonantibiotic therapy fails and the patient is under a high amount of psychological strain should antibiotic prophylaxis be initiated, according to the authors. A period of 3-6 months should be the target here. When choosing an antibiotic and before starting therapy, the corresponding pathogen should be confirmed through a urine culture, and resistance testing should be performed. On the other hand, single-use, postcoital antibiotic prevention could be an alternative, particularly for women in whom a correlation between recurrent UTIs and sexual intercourse has been suspected, the authors wrote.
This article was translated from Univadis Germany. A version appeared on Medscape.com.
FROM DMW KLINISCHER FORTSCHRITT
Original COVID-19 vaccines fall short against Omicron subvariants for the immunocompromised
The effectiveness of up to three doses of COVID-19 vaccine was moderate overall and significantly lower among individuals with immunocompromising conditions, compared with the general population during the period of Omicron dominance, according to an analysis of data from more than 34,000 hospitalizations.
Previous studies have suggested lower COVID-19 vaccine effectiveness among immunocompromised individuals, compared with healthy individuals from the general population, but data from the period in which Omicron subvariants have been dominant are limited, wrote Amadea Britton, MD, of the Centers for Disease Control and Prevention’s COVID-19 Emergency Response Team, and colleagues.
The CDC currently recommends an expanded primary vaccine series of three doses of an mRNA vaccine, and the Advisory Committee on Immunization Practices has recommended a fourth dose with the new bivalent booster that contains elements of the Omicron variant, the researchers noted.
In a study published in the CDC’s Morbidity and Mortality Weekly Report, the researchers identified 34,220 adults with immunocompromising conditions who were hospitalized for COVID-19–like illness between Dec. 16, 2021, and Aug. 20, 2022. These conditions included solid malignancy (40.5%), hematologic malignancy (14.6%), rheumatologic or inflammatory disorder (24.4%), other intrinsic immune condition or immunodeficiency (38.5%), or organ or stem cell transplant (8.6%). They used data from the CDC’s VISION Network, a multistate database. The data include spring and summer 2022, when the BA.4 and BA.5 Omicron subvariants dominated other strains, and adults with immunocompromising conditions were eligible for a total of four vaccine doses (two primary doses and two boosters). The median age of the study population was 69 years, and 25.7%, 41.7%, and 7.0% had received two, three, and four doses, respectively, of COVID-19 vaccine.
Overall, vaccine effectiveness (VE) among immunocompromised patients was 34% after two vaccine doses, increasing to 71% during days 7-89 after a third dose, then declining to 41% 90 days or more after that dose.
During the full Omicron period, VE was 36% for 14 or more days after dose two, 69% for 7-89 days after dose three, and 44% for 90 or more days after dose three.
When VE was stratified by sublineage period, VE was higher 7 or more days after dose three during the predominance of BA.1 (67%), compared with VE during the dominant periods of BA.2/BA.2.12.1 (32%) and BA.4/BA.5 (35%).
In the later periods when Omicron BA.2/BA.2.12.1 and BA.4/BA.5 variants dominated, and individuals who had received three doses of vaccine were eligible for a fourth, VE against these variants was 32% 90 or more days after dose three and 43% 7 or more days after dose four.
VE was lowest among individuals with potentially more severe immunocompromising conditions, notably solid organ or stem cell transplants, the researchers wrote in their discussion.
The study findings were limited by several factors including the use of ICD-9 and -10 discharge diagnosis codes for immunocompromising conditions, potential confounding in VE models, lack of data on outpatient treatments such as nirmatelvir/ritonavir (Paxlovid), and lack of COVID-19 genomic sequencing data that may have affected which sublineage was identified, the researchers noted.
However, “this study confirms that even with boosters, immunocompromised adults, because of their weakened immune systems, are still at high risk of moderate to severe COVID,” said coauthor Brian Dixon, PhD, of the Regenstrief Institute and Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, in a press release about the study.
“Given the incomplete protection against hospitalization afforded by monovalent COVID-19 vaccines, persons with immunocompromising conditions might benefit from updated bivalent vaccine booster doses that target recently circulating Omicron sublineages, in line with ACIP [Advisory Committee on Immunization Practices] recommendations,” the researchers concluded in the study.
The study was funded by the CDC. The researchers had no financial conflicts to disclose. The VISION Network is a collaboration between the CDC, the Regenstrief Institute, and seven health care systems across the United States: Columbia University Irving Medical Center (New York), HealthPartners (Wisconsin), Intermountain Healthcare (Utah), Kaiser Permanente Northern California, Kaiser Permanente Northwest (Washington State), the University of Colorado, and Paso Del Norte Health Information Exchange (Texas).
The effectiveness of up to three doses of COVID-19 vaccine was moderate overall and significantly lower among individuals with immunocompromising conditions, compared with the general population during the period of Omicron dominance, according to an analysis of data from more than 34,000 hospitalizations.
Previous studies have suggested lower COVID-19 vaccine effectiveness among immunocompromised individuals, compared with healthy individuals from the general population, but data from the period in which Omicron subvariants have been dominant are limited, wrote Amadea Britton, MD, of the Centers for Disease Control and Prevention’s COVID-19 Emergency Response Team, and colleagues.
The CDC currently recommends an expanded primary vaccine series of three doses of an mRNA vaccine, and the Advisory Committee on Immunization Practices has recommended a fourth dose with the new bivalent booster that contains elements of the Omicron variant, the researchers noted.
In a study published in the CDC’s Morbidity and Mortality Weekly Report, the researchers identified 34,220 adults with immunocompromising conditions who were hospitalized for COVID-19–like illness between Dec. 16, 2021, and Aug. 20, 2022. These conditions included solid malignancy (40.5%), hematologic malignancy (14.6%), rheumatologic or inflammatory disorder (24.4%), other intrinsic immune condition or immunodeficiency (38.5%), or organ or stem cell transplant (8.6%). They used data from the CDC’s VISION Network, a multistate database. The data include spring and summer 2022, when the BA.4 and BA.5 Omicron subvariants dominated other strains, and adults with immunocompromising conditions were eligible for a total of four vaccine doses (two primary doses and two boosters). The median age of the study population was 69 years, and 25.7%, 41.7%, and 7.0% had received two, three, and four doses, respectively, of COVID-19 vaccine.
Overall, vaccine effectiveness (VE) among immunocompromised patients was 34% after two vaccine doses, increasing to 71% during days 7-89 after a third dose, then declining to 41% 90 days or more after that dose.
During the full Omicron period, VE was 36% for 14 or more days after dose two, 69% for 7-89 days after dose three, and 44% for 90 or more days after dose three.
When VE was stratified by sublineage period, VE was higher 7 or more days after dose three during the predominance of BA.1 (67%), compared with VE during the dominant periods of BA.2/BA.2.12.1 (32%) and BA.4/BA.5 (35%).
In the later periods when Omicron BA.2/BA.2.12.1 and BA.4/BA.5 variants dominated, and individuals who had received three doses of vaccine were eligible for a fourth, VE against these variants was 32% 90 or more days after dose three and 43% 7 or more days after dose four.
VE was lowest among individuals with potentially more severe immunocompromising conditions, notably solid organ or stem cell transplants, the researchers wrote in their discussion.
The study findings were limited by several factors including the use of ICD-9 and -10 discharge diagnosis codes for immunocompromising conditions, potential confounding in VE models, lack of data on outpatient treatments such as nirmatelvir/ritonavir (Paxlovid), and lack of COVID-19 genomic sequencing data that may have affected which sublineage was identified, the researchers noted.
However, “this study confirms that even with boosters, immunocompromised adults, because of their weakened immune systems, are still at high risk of moderate to severe COVID,” said coauthor Brian Dixon, PhD, of the Regenstrief Institute and Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, in a press release about the study.
“Given the incomplete protection against hospitalization afforded by monovalent COVID-19 vaccines, persons with immunocompromising conditions might benefit from updated bivalent vaccine booster doses that target recently circulating Omicron sublineages, in line with ACIP [Advisory Committee on Immunization Practices] recommendations,” the researchers concluded in the study.
The study was funded by the CDC. The researchers had no financial conflicts to disclose. The VISION Network is a collaboration between the CDC, the Regenstrief Institute, and seven health care systems across the United States: Columbia University Irving Medical Center (New York), HealthPartners (Wisconsin), Intermountain Healthcare (Utah), Kaiser Permanente Northern California, Kaiser Permanente Northwest (Washington State), the University of Colorado, and Paso Del Norte Health Information Exchange (Texas).
The effectiveness of up to three doses of COVID-19 vaccine was moderate overall and significantly lower among individuals with immunocompromising conditions, compared with the general population during the period of Omicron dominance, according to an analysis of data from more than 34,000 hospitalizations.
Previous studies have suggested lower COVID-19 vaccine effectiveness among immunocompromised individuals, compared with healthy individuals from the general population, but data from the period in which Omicron subvariants have been dominant are limited, wrote Amadea Britton, MD, of the Centers for Disease Control and Prevention’s COVID-19 Emergency Response Team, and colleagues.
The CDC currently recommends an expanded primary vaccine series of three doses of an mRNA vaccine, and the Advisory Committee on Immunization Practices has recommended a fourth dose with the new bivalent booster that contains elements of the Omicron variant, the researchers noted.
In a study published in the CDC’s Morbidity and Mortality Weekly Report, the researchers identified 34,220 adults with immunocompromising conditions who were hospitalized for COVID-19–like illness between Dec. 16, 2021, and Aug. 20, 2022. These conditions included solid malignancy (40.5%), hematologic malignancy (14.6%), rheumatologic or inflammatory disorder (24.4%), other intrinsic immune condition or immunodeficiency (38.5%), or organ or stem cell transplant (8.6%). They used data from the CDC’s VISION Network, a multistate database. The data include spring and summer 2022, when the BA.4 and BA.5 Omicron subvariants dominated other strains, and adults with immunocompromising conditions were eligible for a total of four vaccine doses (two primary doses and two boosters). The median age of the study population was 69 years, and 25.7%, 41.7%, and 7.0% had received two, three, and four doses, respectively, of COVID-19 vaccine.
Overall, vaccine effectiveness (VE) among immunocompromised patients was 34% after two vaccine doses, increasing to 71% during days 7-89 after a third dose, then declining to 41% 90 days or more after that dose.
During the full Omicron period, VE was 36% for 14 or more days after dose two, 69% for 7-89 days after dose three, and 44% for 90 or more days after dose three.
When VE was stratified by sublineage period, VE was higher 7 or more days after dose three during the predominance of BA.1 (67%), compared with VE during the dominant periods of BA.2/BA.2.12.1 (32%) and BA.4/BA.5 (35%).
In the later periods when Omicron BA.2/BA.2.12.1 and BA.4/BA.5 variants dominated, and individuals who had received three doses of vaccine were eligible for a fourth, VE against these variants was 32% 90 or more days after dose three and 43% 7 or more days after dose four.
VE was lowest among individuals with potentially more severe immunocompromising conditions, notably solid organ or stem cell transplants, the researchers wrote in their discussion.
The study findings were limited by several factors including the use of ICD-9 and -10 discharge diagnosis codes for immunocompromising conditions, potential confounding in VE models, lack of data on outpatient treatments such as nirmatelvir/ritonavir (Paxlovid), and lack of COVID-19 genomic sequencing data that may have affected which sublineage was identified, the researchers noted.
However, “this study confirms that even with boosters, immunocompromised adults, because of their weakened immune systems, are still at high risk of moderate to severe COVID,” said coauthor Brian Dixon, PhD, of the Regenstrief Institute and Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, in a press release about the study.
“Given the incomplete protection against hospitalization afforded by monovalent COVID-19 vaccines, persons with immunocompromising conditions might benefit from updated bivalent vaccine booster doses that target recently circulating Omicron sublineages, in line with ACIP [Advisory Committee on Immunization Practices] recommendations,” the researchers concluded in the study.
The study was funded by the CDC. The researchers had no financial conflicts to disclose. The VISION Network is a collaboration between the CDC, the Regenstrief Institute, and seven health care systems across the United States: Columbia University Irving Medical Center (New York), HealthPartners (Wisconsin), Intermountain Healthcare (Utah), Kaiser Permanente Northern California, Kaiser Permanente Northwest (Washington State), the University of Colorado, and Paso Del Norte Health Information Exchange (Texas).
FROM MMWR
Syphilis screening: Who and when
The US Preventive Services Task Force (USPSTF) published updated recommendations on screening for syphilis on September 27.1 The Task Force continues to recommend screening for all adolescents and adults who are at increased risk for infection. (As part of previous recommendations, the USPSTF also advocates screening all pregnant women for syphilis early in their pregnancy to prevent congenital syphilis.2)
Who is at increased risk? Men who have sex with men (MSM), those with HIV or other sexually transmitted infections (STIs), those who use illicit drugs, and those with a history of incarceration, sex work, or military service are considered to be at increased risk for syphilis. Additionally, since state and local health departments collect and publish STI incidence data, it’s important to stay up to date on how common syphilis is in one’s community and tailor screening practices accordingly.
Men account for more than 80% of all primary and secondary syphilis infections, and MSM account for 53% of cases in men.3 The highest rates of syphilis are in men ages 25-29 years and 30-34 years (58.1 and 55.7 cases per 100,000, respectively).3
Why screening is important. Primary and secondary syphilis rates have increased steadily from an all-time low of 2.1 per 100,000 in 2000 to 12.7 per 100,000 in 2020.4 There were 171,074 cases reported in 2021.5
If not detected and treated, syphilis will progress from the primary and secondary stages to a latent form. About one-third of those with latent syphilis will develop tertiary syphilis, which can affect every organ system and cause multiple neurologic disorders.
How to screen. Syphilis screening typically involves a 2-step process. The first test that should be performed is a Venereal Disease Research Laboratory (VDRL) or rapid plasma reagin (RPR) test. This is followed by a treponemal antibody test if the initial test is positive. While the VDRL and RPR tests have high sensitivity, many other conditions can cause a false-positive result, necessitating confirmation with the more specific antibody test.
As far as frequency, the Task Force suggests screening annually for those at continued risk and more frequently (every 3 or 6 months) for those at highest risk.
Treatment for primary, secondary, and early latent syphilis (< 1 year’s duration) is a single intramuscular (IM) injection of benzathine penicillin, 2.4 million units. For late latent syphilis or syphilis of unknown duration, treatment is benzathine penicillin, 2.4 million units, administered in 3 weekly IM doses.
Treatment for those with penicillin allergies depends on the stage of syphilis and whether or not the patient is pregnant. Refer to the STD treatment guidelines for guidance.6
The CDC recommends presumptive treatment for anyone who has had sexual contact in the past 90 days with a person who’s been given a diagnosis of primary, secondary, or early latent syphilis.6
And finally, remember that all STIs are reportable to your local health department, which can assist with contract tracing and treatment follow-up.
1. USPSTF. Syphilis infection in nonpregnant adolescents and adults: Screening. Final recommendation statement. September 27, 2022. Accessed October 25, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-nonpregnant-adults-adolescents-screening
2. USPSTF. Syphilis infection in pregnant women: screening. Final recommendation statement. September 4, 2018. Accessed October 25, 2022. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-in-pregnancy-screening
3. CDC. Sexually transmitted disease surveillance 2020: syphilis. Updated August 22, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/figures/2020-STD-Surveillance-Syphilis.pptx
4. CDC. Sexually transmitted disease surveillance 2020. Table 1: Sexually transmitted diseases—reported cases and rates of reported cases, United States, 1941-2020. Updated April 12, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/tables/1.htm
5. CDC. Preliminary 2021 STD surveillance data. Updated September 1, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2021/default.htm
6. Workowski KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recommend Rep. 2021;70:1-187.
The US Preventive Services Task Force (USPSTF) published updated recommendations on screening for syphilis on September 27.1 The Task Force continues to recommend screening for all adolescents and adults who are at increased risk for infection. (As part of previous recommendations, the USPSTF also advocates screening all pregnant women for syphilis early in their pregnancy to prevent congenital syphilis.2)
Who is at increased risk? Men who have sex with men (MSM), those with HIV or other sexually transmitted infections (STIs), those who use illicit drugs, and those with a history of incarceration, sex work, or military service are considered to be at increased risk for syphilis. Additionally, since state and local health departments collect and publish STI incidence data, it’s important to stay up to date on how common syphilis is in one’s community and tailor screening practices accordingly.
Men account for more than 80% of all primary and secondary syphilis infections, and MSM account for 53% of cases in men.3 The highest rates of syphilis are in men ages 25-29 years and 30-34 years (58.1 and 55.7 cases per 100,000, respectively).3
Why screening is important. Primary and secondary syphilis rates have increased steadily from an all-time low of 2.1 per 100,000 in 2000 to 12.7 per 100,000 in 2020.4 There were 171,074 cases reported in 2021.5
If not detected and treated, syphilis will progress from the primary and secondary stages to a latent form. About one-third of those with latent syphilis will develop tertiary syphilis, which can affect every organ system and cause multiple neurologic disorders.
How to screen. Syphilis screening typically involves a 2-step process. The first test that should be performed is a Venereal Disease Research Laboratory (VDRL) or rapid plasma reagin (RPR) test. This is followed by a treponemal antibody test if the initial test is positive. While the VDRL and RPR tests have high sensitivity, many other conditions can cause a false-positive result, necessitating confirmation with the more specific antibody test.
As far as frequency, the Task Force suggests screening annually for those at continued risk and more frequently (every 3 or 6 months) for those at highest risk.
Treatment for primary, secondary, and early latent syphilis (< 1 year’s duration) is a single intramuscular (IM) injection of benzathine penicillin, 2.4 million units. For late latent syphilis or syphilis of unknown duration, treatment is benzathine penicillin, 2.4 million units, administered in 3 weekly IM doses.
Treatment for those with penicillin allergies depends on the stage of syphilis and whether or not the patient is pregnant. Refer to the STD treatment guidelines for guidance.6
The CDC recommends presumptive treatment for anyone who has had sexual contact in the past 90 days with a person who’s been given a diagnosis of primary, secondary, or early latent syphilis.6
And finally, remember that all STIs are reportable to your local health department, which can assist with contract tracing and treatment follow-up.
The US Preventive Services Task Force (USPSTF) published updated recommendations on screening for syphilis on September 27.1 The Task Force continues to recommend screening for all adolescents and adults who are at increased risk for infection. (As part of previous recommendations, the USPSTF also advocates screening all pregnant women for syphilis early in their pregnancy to prevent congenital syphilis.2)
Who is at increased risk? Men who have sex with men (MSM), those with HIV or other sexually transmitted infections (STIs), those who use illicit drugs, and those with a history of incarceration, sex work, or military service are considered to be at increased risk for syphilis. Additionally, since state and local health departments collect and publish STI incidence data, it’s important to stay up to date on how common syphilis is in one’s community and tailor screening practices accordingly.
Men account for more than 80% of all primary and secondary syphilis infections, and MSM account for 53% of cases in men.3 The highest rates of syphilis are in men ages 25-29 years and 30-34 years (58.1 and 55.7 cases per 100,000, respectively).3
Why screening is important. Primary and secondary syphilis rates have increased steadily from an all-time low of 2.1 per 100,000 in 2000 to 12.7 per 100,000 in 2020.4 There were 171,074 cases reported in 2021.5
If not detected and treated, syphilis will progress from the primary and secondary stages to a latent form. About one-third of those with latent syphilis will develop tertiary syphilis, which can affect every organ system and cause multiple neurologic disorders.
How to screen. Syphilis screening typically involves a 2-step process. The first test that should be performed is a Venereal Disease Research Laboratory (VDRL) or rapid plasma reagin (RPR) test. This is followed by a treponemal antibody test if the initial test is positive. While the VDRL and RPR tests have high sensitivity, many other conditions can cause a false-positive result, necessitating confirmation with the more specific antibody test.
As far as frequency, the Task Force suggests screening annually for those at continued risk and more frequently (every 3 or 6 months) for those at highest risk.
Treatment for primary, secondary, and early latent syphilis (< 1 year’s duration) is a single intramuscular (IM) injection of benzathine penicillin, 2.4 million units. For late latent syphilis or syphilis of unknown duration, treatment is benzathine penicillin, 2.4 million units, administered in 3 weekly IM doses.
Treatment for those with penicillin allergies depends on the stage of syphilis and whether or not the patient is pregnant. Refer to the STD treatment guidelines for guidance.6
The CDC recommends presumptive treatment for anyone who has had sexual contact in the past 90 days with a person who’s been given a diagnosis of primary, secondary, or early latent syphilis.6
And finally, remember that all STIs are reportable to your local health department, which can assist with contract tracing and treatment follow-up.
1. USPSTF. Syphilis infection in nonpregnant adolescents and adults: Screening. Final recommendation statement. September 27, 2022. Accessed October 25, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-nonpregnant-adults-adolescents-screening
2. USPSTF. Syphilis infection in pregnant women: screening. Final recommendation statement. September 4, 2018. Accessed October 25, 2022. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-in-pregnancy-screening
3. CDC. Sexually transmitted disease surveillance 2020: syphilis. Updated August 22, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/figures/2020-STD-Surveillance-Syphilis.pptx
4. CDC. Sexually transmitted disease surveillance 2020. Table 1: Sexually transmitted diseases—reported cases and rates of reported cases, United States, 1941-2020. Updated April 12, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/tables/1.htm
5. CDC. Preliminary 2021 STD surveillance data. Updated September 1, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2021/default.htm
6. Workowski KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recommend Rep. 2021;70:1-187.
1. USPSTF. Syphilis infection in nonpregnant adolescents and adults: Screening. Final recommendation statement. September 27, 2022. Accessed October 25, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-nonpregnant-adults-adolescents-screening
2. USPSTF. Syphilis infection in pregnant women: screening. Final recommendation statement. September 4, 2018. Accessed October 25, 2022. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/syphilis-infection-in-pregnancy-screening
3. CDC. Sexually transmitted disease surveillance 2020: syphilis. Updated August 22, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/figures/2020-STD-Surveillance-Syphilis.pptx
4. CDC. Sexually transmitted disease surveillance 2020. Table 1: Sexually transmitted diseases—reported cases and rates of reported cases, United States, 1941-2020. Updated April 12, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2020/tables/1.htm
5. CDC. Preliminary 2021 STD surveillance data. Updated September 1, 2022. Accessed October 25, 2022. www.cdc.gov/std/statistics/2021/default.htm
6. Workowski KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recommend Rep. 2021;70:1-187.
RBX2660 shows promise in breaking the cycle of recurrent C. difficile
CHARLOTTE, N.C. –
Following a standard course of antibiotics, a one-time treatment with RBX2660 was successful for three quarters of participants at 8 weeks, according to a new study. It also prevented additional bouts, with 84% of these initial responders remaining free of C. difficile infection at 6 months.
The ongoing phase 3, open-label PUNCH CD3-OLS study expands on clinical trial experience by treating more “real-world” patients. People who might have been excluded from previous research because of comorbidities, such as irritable bowel syndrome, inflammatory bowel disease, and immunosuppression, were included.
The study also placed no limit on the number of previous rounds of C. difficile infections.
“Even when you expand the patient population to make it more generalizable, we’re still seeing both a high cure rate and a high success rate,” Sahil Khanna, MBBS, a gastroenterologist and hepatologist at the Mayo Clinic in Rochester, Minn., said in an interview.
“We also are not seeing any kind of safety signals that can be attributed to this particular product,” he said.
Dr. Khanna presented the findings during the annual meeting of the American College of Gastroenterology, which were also published simultaneously in the journal Drugs. The research by Dr. Khanna and associates received an ACG Outstanding Research Award in the colon category.
Study design and results
RBX2660 (Rebyota) is a microbiota-based live biotherapeutic in development from Ferring Pharmaceuticals. The treatment contains human stool collected from prescreened, qualified donors and is prepared according to good manufacturing standards.
After standard-of-care antibiotics and a 72-hour washout period, participants received a single 150-mL dose rectally by enema. RBX2660 is administered by a health care professional.
The median age of study participants was 63 years, with 45% aged 65 years or older, and 70% were women. Overall, 37% of participants had Crohn’s disease and 4% had ulcerative colitis.
At the time of screening, about half of participants had a history of one or two infections with C. difficile, and the remaining half reported three or more episodes.
Of the 402 participants whose outcomes could be analyzed, 75% reported treatment success, meaning no further C. difficile infections at 8 weeks. This was consistent with the 75% of 60 participants free of C. difficile in the interim analysis reported in 2021. Efficacy results were based on a modified intent-to-treat analysis.
Of the 300 participants who responded to RBX2660 at 8 weeks, 262 were followed up to 6 months, with 84% of these reporting no C. difficile recurrence.
“If you succeeded to 8 weeks, there was a high likelihood that you would succeed up to 6 months,” Dr. Khanna said.
For the subset of participants with inflammatory bowel disease, Dr. Khanna noted that the success rates were in the 80% range, which is higher than what is seen in clinic fecal microbiota transplantation programs.
Adverse events
Of the participants, 63% reported treatment-emergent adverse events. Most events were mild to moderate in severity, the researchers reported, with diarrhea and abdominal pain being the most common.
“When you look at the treatment-emergent adverse events, it’s important to put them into context in terms of this patient population,” Dr. Khanna said. “This recurrent population has developed underlying gastrointestinal symptoms like abdominal pain, diarrhea, nausea, vomiting, and weight loss.”
Some of these adverse events persist beyond resolution of the C. difficile infection, and the adverse-event profile with RBX2660 is consistent with what is seen following fecal microbiota transplantation, he added.
The serious adverse events “were very, very few,” Dr. Khanna said.
Overall, 11% of participants reported a serious adverse event. The majority were related to the C. difficile infection or an underlying comorbidity, he noted.
“Excruciating for patients to deal with”
Traditionally, there could be “some hesitation on the patient’s part [to undergo therapy] just because it’s delivered rectally,” session comoderator Lisa Malter, MD, said in an interview.
However, C. difficile can be “excruciating for patients to deal with,” said Dr. Malter, a gastroenterologist and professor of medicine at New York University Langone Health. They “may be more than willing to take [this agent] because it gets them feeling better.”
“This is a positive adjunct to our current therapies for C. diff in terms of trying to knock it out once a standard course of antibiotics has been administered,” she added.
Currently, people with recurrent C. difficile seek fecal microbiota material from a biobank or from a close friend or loved one.
But Dr. Malter noted that asking someone you know to donate fecal matter for transplantation requires several steps. Donors are screened to make sure they are free of gastrointestinal illness, are not taking any contraindicated medications, and do not have active infection.
Fecal microbiota samples from a biobank are more standardized, but there have been intermittent shutdowns and availability has been limited during the pandemic, she said.
Dr. Malter added that one unanswered question is how much of the colon is covered by therapy delivery via enema compared with colonoscope delivery during fecal microbiota transplantation.
“If it’s delivered colonoscopically, you get the entire colon. In contrast with an enema, you really only hit the left side of the colon,” she said.
FDA advisory committee nod
On Sept. 26, the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee reviewed evidence for RBX2660. The committee voted 13 to 4 that data were adequate to support the effectiveness of RBX2660 to reduce the recurrence of C. difficile infection in adults following antibiotic treatment for recurrent infections.
Members also voted 12 to 4, with one abstention, that the data were adequate to support the product’s safety.
The FDA often follows its advisory committee recommendations but is not required to do so.
“The hope would be that this would get through the usual FDA pipeline of an approval in the near future,” Dr. Khanna said.
The study was funded by Ferring Pharmaceuticals. Dr. Khanna reported receiving grant and research funding from Ferring. Dr. Malter reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHARLOTTE, N.C. –
Following a standard course of antibiotics, a one-time treatment with RBX2660 was successful for three quarters of participants at 8 weeks, according to a new study. It also prevented additional bouts, with 84% of these initial responders remaining free of C. difficile infection at 6 months.
The ongoing phase 3, open-label PUNCH CD3-OLS study expands on clinical trial experience by treating more “real-world” patients. People who might have been excluded from previous research because of comorbidities, such as irritable bowel syndrome, inflammatory bowel disease, and immunosuppression, were included.
The study also placed no limit on the number of previous rounds of C. difficile infections.
“Even when you expand the patient population to make it more generalizable, we’re still seeing both a high cure rate and a high success rate,” Sahil Khanna, MBBS, a gastroenterologist and hepatologist at the Mayo Clinic in Rochester, Minn., said in an interview.
“We also are not seeing any kind of safety signals that can be attributed to this particular product,” he said.
Dr. Khanna presented the findings during the annual meeting of the American College of Gastroenterology, which were also published simultaneously in the journal Drugs. The research by Dr. Khanna and associates received an ACG Outstanding Research Award in the colon category.
Study design and results
RBX2660 (Rebyota) is a microbiota-based live biotherapeutic in development from Ferring Pharmaceuticals. The treatment contains human stool collected from prescreened, qualified donors and is prepared according to good manufacturing standards.
After standard-of-care antibiotics and a 72-hour washout period, participants received a single 150-mL dose rectally by enema. RBX2660 is administered by a health care professional.
The median age of study participants was 63 years, with 45% aged 65 years or older, and 70% were women. Overall, 37% of participants had Crohn’s disease and 4% had ulcerative colitis.
At the time of screening, about half of participants had a history of one or two infections with C. difficile, and the remaining half reported three or more episodes.
Of the 402 participants whose outcomes could be analyzed, 75% reported treatment success, meaning no further C. difficile infections at 8 weeks. This was consistent with the 75% of 60 participants free of C. difficile in the interim analysis reported in 2021. Efficacy results were based on a modified intent-to-treat analysis.
Of the 300 participants who responded to RBX2660 at 8 weeks, 262 were followed up to 6 months, with 84% of these reporting no C. difficile recurrence.
“If you succeeded to 8 weeks, there was a high likelihood that you would succeed up to 6 months,” Dr. Khanna said.
For the subset of participants with inflammatory bowel disease, Dr. Khanna noted that the success rates were in the 80% range, which is higher than what is seen in clinic fecal microbiota transplantation programs.
Adverse events
Of the participants, 63% reported treatment-emergent adverse events. Most events were mild to moderate in severity, the researchers reported, with diarrhea and abdominal pain being the most common.
“When you look at the treatment-emergent adverse events, it’s important to put them into context in terms of this patient population,” Dr. Khanna said. “This recurrent population has developed underlying gastrointestinal symptoms like abdominal pain, diarrhea, nausea, vomiting, and weight loss.”
Some of these adverse events persist beyond resolution of the C. difficile infection, and the adverse-event profile with RBX2660 is consistent with what is seen following fecal microbiota transplantation, he added.
The serious adverse events “were very, very few,” Dr. Khanna said.
Overall, 11% of participants reported a serious adverse event. The majority were related to the C. difficile infection or an underlying comorbidity, he noted.
“Excruciating for patients to deal with”
Traditionally, there could be “some hesitation on the patient’s part [to undergo therapy] just because it’s delivered rectally,” session comoderator Lisa Malter, MD, said in an interview.
However, C. difficile can be “excruciating for patients to deal with,” said Dr. Malter, a gastroenterologist and professor of medicine at New York University Langone Health. They “may be more than willing to take [this agent] because it gets them feeling better.”
“This is a positive adjunct to our current therapies for C. diff in terms of trying to knock it out once a standard course of antibiotics has been administered,” she added.
Currently, people with recurrent C. difficile seek fecal microbiota material from a biobank or from a close friend or loved one.
But Dr. Malter noted that asking someone you know to donate fecal matter for transplantation requires several steps. Donors are screened to make sure they are free of gastrointestinal illness, are not taking any contraindicated medications, and do not have active infection.
Fecal microbiota samples from a biobank are more standardized, but there have been intermittent shutdowns and availability has been limited during the pandemic, she said.
Dr. Malter added that one unanswered question is how much of the colon is covered by therapy delivery via enema compared with colonoscope delivery during fecal microbiota transplantation.
“If it’s delivered colonoscopically, you get the entire colon. In contrast with an enema, you really only hit the left side of the colon,” she said.
FDA advisory committee nod
On Sept. 26, the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee reviewed evidence for RBX2660. The committee voted 13 to 4 that data were adequate to support the effectiveness of RBX2660 to reduce the recurrence of C. difficile infection in adults following antibiotic treatment for recurrent infections.
Members also voted 12 to 4, with one abstention, that the data were adequate to support the product’s safety.
The FDA often follows its advisory committee recommendations but is not required to do so.
“The hope would be that this would get through the usual FDA pipeline of an approval in the near future,” Dr. Khanna said.
The study was funded by Ferring Pharmaceuticals. Dr. Khanna reported receiving grant and research funding from Ferring. Dr. Malter reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHARLOTTE, N.C. –
Following a standard course of antibiotics, a one-time treatment with RBX2660 was successful for three quarters of participants at 8 weeks, according to a new study. It also prevented additional bouts, with 84% of these initial responders remaining free of C. difficile infection at 6 months.
The ongoing phase 3, open-label PUNCH CD3-OLS study expands on clinical trial experience by treating more “real-world” patients. People who might have been excluded from previous research because of comorbidities, such as irritable bowel syndrome, inflammatory bowel disease, and immunosuppression, were included.
The study also placed no limit on the number of previous rounds of C. difficile infections.
“Even when you expand the patient population to make it more generalizable, we’re still seeing both a high cure rate and a high success rate,” Sahil Khanna, MBBS, a gastroenterologist and hepatologist at the Mayo Clinic in Rochester, Minn., said in an interview.
“We also are not seeing any kind of safety signals that can be attributed to this particular product,” he said.
Dr. Khanna presented the findings during the annual meeting of the American College of Gastroenterology, which were also published simultaneously in the journal Drugs. The research by Dr. Khanna and associates received an ACG Outstanding Research Award in the colon category.
Study design and results
RBX2660 (Rebyota) is a microbiota-based live biotherapeutic in development from Ferring Pharmaceuticals. The treatment contains human stool collected from prescreened, qualified donors and is prepared according to good manufacturing standards.
After standard-of-care antibiotics and a 72-hour washout period, participants received a single 150-mL dose rectally by enema. RBX2660 is administered by a health care professional.
The median age of study participants was 63 years, with 45% aged 65 years or older, and 70% were women. Overall, 37% of participants had Crohn’s disease and 4% had ulcerative colitis.
At the time of screening, about half of participants had a history of one or two infections with C. difficile, and the remaining half reported three or more episodes.
Of the 402 participants whose outcomes could be analyzed, 75% reported treatment success, meaning no further C. difficile infections at 8 weeks. This was consistent with the 75% of 60 participants free of C. difficile in the interim analysis reported in 2021. Efficacy results were based on a modified intent-to-treat analysis.
Of the 300 participants who responded to RBX2660 at 8 weeks, 262 were followed up to 6 months, with 84% of these reporting no C. difficile recurrence.
“If you succeeded to 8 weeks, there was a high likelihood that you would succeed up to 6 months,” Dr. Khanna said.
For the subset of participants with inflammatory bowel disease, Dr. Khanna noted that the success rates were in the 80% range, which is higher than what is seen in clinic fecal microbiota transplantation programs.
Adverse events
Of the participants, 63% reported treatment-emergent adverse events. Most events were mild to moderate in severity, the researchers reported, with diarrhea and abdominal pain being the most common.
“When you look at the treatment-emergent adverse events, it’s important to put them into context in terms of this patient population,” Dr. Khanna said. “This recurrent population has developed underlying gastrointestinal symptoms like abdominal pain, diarrhea, nausea, vomiting, and weight loss.”
Some of these adverse events persist beyond resolution of the C. difficile infection, and the adverse-event profile with RBX2660 is consistent with what is seen following fecal microbiota transplantation, he added.
The serious adverse events “were very, very few,” Dr. Khanna said.
Overall, 11% of participants reported a serious adverse event. The majority were related to the C. difficile infection or an underlying comorbidity, he noted.
“Excruciating for patients to deal with”
Traditionally, there could be “some hesitation on the patient’s part [to undergo therapy] just because it’s delivered rectally,” session comoderator Lisa Malter, MD, said in an interview.
However, C. difficile can be “excruciating for patients to deal with,” said Dr. Malter, a gastroenterologist and professor of medicine at New York University Langone Health. They “may be more than willing to take [this agent] because it gets them feeling better.”
“This is a positive adjunct to our current therapies for C. diff in terms of trying to knock it out once a standard course of antibiotics has been administered,” she added.
Currently, people with recurrent C. difficile seek fecal microbiota material from a biobank or from a close friend or loved one.
But Dr. Malter noted that asking someone you know to donate fecal matter for transplantation requires several steps. Donors are screened to make sure they are free of gastrointestinal illness, are not taking any contraindicated medications, and do not have active infection.
Fecal microbiota samples from a biobank are more standardized, but there have been intermittent shutdowns and availability has been limited during the pandemic, she said.
Dr. Malter added that one unanswered question is how much of the colon is covered by therapy delivery via enema compared with colonoscope delivery during fecal microbiota transplantation.
“If it’s delivered colonoscopically, you get the entire colon. In contrast with an enema, you really only hit the left side of the colon,” she said.
FDA advisory committee nod
On Sept. 26, the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee reviewed evidence for RBX2660. The committee voted 13 to 4 that data were adequate to support the effectiveness of RBX2660 to reduce the recurrence of C. difficile infection in adults following antibiotic treatment for recurrent infections.
Members also voted 12 to 4, with one abstention, that the data were adequate to support the product’s safety.
The FDA often follows its advisory committee recommendations but is not required to do so.
“The hope would be that this would get through the usual FDA pipeline of an approval in the near future,” Dr. Khanna said.
The study was funded by Ferring Pharmaceuticals. Dr. Khanna reported receiving grant and research funding from Ferring. Dr. Malter reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACG 2022
Droplet dispersal in sterile processing units far exceeds guideline limit
In the era of Ebola, COVID-19, and even Legionnaires, technicians and other staff working behind the scenes to ensure provider and patient safety continue to face a long-recognized but under addressed challenge: splashes and airborne droplets.
Granted, National Institute for Occupational Safety and Health (NIOSH) standards, industry standards, and professional guidelines are all in place to prevent unintentional exposure to pathogens. However, findings from a newly published study in the American Journal of Infection Control suggest they fall short.
In the study, researchers found that simulated manual cleaning of medical devices generated a drenching splash throughout the process with droplet dispersal exceeding 7 feet (2.1 meters).
Cori L. Ofstead, MSPH, lead author and president/CEO of Ofstead & Associates, Bloomington, Minn., told this news organization. “That’s the problem with having standards and guidelines that are not based on relevant evidence, [which] in this case, is a single study that was done in an intensive care area where they had an infection outbreak.”
Ms. Ofstead was referring to a report in the journal Infection Control and Hospital Epidemiology, detailing a Canadian investigation involving a multidrug-resistant Pseudomonas aeruginosa outbreak in an ICU. The report implicated the faucets over the hand hygiene sinks, with fluorescent dye showing droplet dispersal roughly 3 feet away from the sinks.
“Somehow it [the 3-feet rule] got implemented in guidelines in sterile processing decontamination areas, which are not the same as hand hygiene,’’ Ms. Ofstead explained.
With a goal of providing more current evidence on droplet generation and dispersal, as well as personal protection equipment (PPE) exposure/effectiveness, she and her colleagues simulated manual cleaning of a decommissioned colonoscope and transvaginal ultrasound probe, using for the study location a new academic sterile processing unit.
To detect droplet generation and dispersal as well as splash following common technician activities (for example, colonoscope brushing, scrubbing, rinsing and transport to an automated endoscope reprocessor [AER] for sterilization), the researchers affixed blue moisture-detection paper to environmental surfaces, on carts positioned 4 feet (1.2 meters) from the sink (to simulate observers), and along a 15-foot pathway between the sink and AER.
They observed droplets everywhere.
Technician activities such as running the faucet and rinsing the probe under running water generated substantial splashing overall. Instrument rinsing in particular produced small and large droplets and confluent puddles of water around the sink and in the broad area surrounding the workspace. Droplets were also dispersed on the floor 7.25 feet (2.2 meters) away and along the entire 15-foot path from the sink to the AER.
At the sink, the technician risked drenching exposure from head to toe during most activities, and even observers positioned 3-4 feet away were found to have droplets on their gowns. In addition, saturated shoe covers reportedly tracked moisture away from the sink to the unit door – a distance of 13 feet (4 meters) – and 2 feet (0.6 meters) farther out into the PPE foyer for donning and doffing.
Although PPE gowns effectively repelled moisture during cleaning of a single device, Ms. Ofstead emphasized that technicians typically handle up to 10 instruments during a normal, 2-hour shift, further increasing exposure risk with each subsequent cleaning.
However, perhaps one of the most surprising findings was that despite an optimal unit design, including physical separation of clean and dirty activities and pressurized air flow to protect workers, droplets were still broadly dispersed.
Current efforts, however well-intentioned, might not be offering the degree of protection (and consideration) that sterile processing technicians need.
“The study was conducted in a new sterile processing area that had an extra excellent kind of distancing and three separate rooms, something that I think most of our hospitals are working toward,” Stella Hines, MD, associate professor at the University of Maryland School of Medicine, Baltimore, explained. Dr. Hines was not directly involved in the study.
“But it also really kind of highlighted what’s happening to workers potentially,” she added. “For example, we want to know if that spray or splatter has a live microbe it in that could cause a problem or ... in a highly wet environment, if that water has some kind of chemical in it that could pose an occupational hazard to the worker based on skin or mucous membrane exposure.”
Ms. Ofstead agreed. “We need to be thinking about the exposure of critically important workers and the environment in an era where we are worried about aerosol-generating procedures and superbugs,” she explained.
Dr. Hines and Ms. Ofstead also noted that the majority of staff involved in front-line patient care have never actually ventured into the sterile processing units nor do they recognize the risks that technicians working in these units face on a daily, or even hourly, basis.
“The people who run these operations are very well trained and knowledgeable. I think that it would be helpful for them to know that they’re appreciated and for the people upstairs on the front lines using the equipment to see what goes on downstairs and all of the painstaking steps that need to be in place for the equipment to come out of sterile processing and be ready to go,” said Dr. Hines.
In the meantime, hospital leaders need to address the challenges and danger posed by migrating infectious droplets, especially for workers involved in processes that stir them up in the first place – workers who by the end of their shifts are unavoidably drenched with infectious blood and tissue secretions.
“I think that it’s going to take a much bigger kind of worldview from hospital leadership,” Dr. Hines said.
The study was supported in part by a grant from Healthmark Industries. Ms. Ofstead reports research grants or consulting fees through her organization with 3M Company, Ambu, Boston Scientific, Cleanis, Fortive/Advanced Sterilization Products, Healthmark Industries, Pentax, and Steris/Cantel/Medviators. Dr. Hines reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the era of Ebola, COVID-19, and even Legionnaires, technicians and other staff working behind the scenes to ensure provider and patient safety continue to face a long-recognized but under addressed challenge: splashes and airborne droplets.
Granted, National Institute for Occupational Safety and Health (NIOSH) standards, industry standards, and professional guidelines are all in place to prevent unintentional exposure to pathogens. However, findings from a newly published study in the American Journal of Infection Control suggest they fall short.
In the study, researchers found that simulated manual cleaning of medical devices generated a drenching splash throughout the process with droplet dispersal exceeding 7 feet (2.1 meters).
Cori L. Ofstead, MSPH, lead author and president/CEO of Ofstead & Associates, Bloomington, Minn., told this news organization. “That’s the problem with having standards and guidelines that are not based on relevant evidence, [which] in this case, is a single study that was done in an intensive care area where they had an infection outbreak.”
Ms. Ofstead was referring to a report in the journal Infection Control and Hospital Epidemiology, detailing a Canadian investigation involving a multidrug-resistant Pseudomonas aeruginosa outbreak in an ICU. The report implicated the faucets over the hand hygiene sinks, with fluorescent dye showing droplet dispersal roughly 3 feet away from the sinks.
“Somehow it [the 3-feet rule] got implemented in guidelines in sterile processing decontamination areas, which are not the same as hand hygiene,’’ Ms. Ofstead explained.
With a goal of providing more current evidence on droplet generation and dispersal, as well as personal protection equipment (PPE) exposure/effectiveness, she and her colleagues simulated manual cleaning of a decommissioned colonoscope and transvaginal ultrasound probe, using for the study location a new academic sterile processing unit.
To detect droplet generation and dispersal as well as splash following common technician activities (for example, colonoscope brushing, scrubbing, rinsing and transport to an automated endoscope reprocessor [AER] for sterilization), the researchers affixed blue moisture-detection paper to environmental surfaces, on carts positioned 4 feet (1.2 meters) from the sink (to simulate observers), and along a 15-foot pathway between the sink and AER.
They observed droplets everywhere.
Technician activities such as running the faucet and rinsing the probe under running water generated substantial splashing overall. Instrument rinsing in particular produced small and large droplets and confluent puddles of water around the sink and in the broad area surrounding the workspace. Droplets were also dispersed on the floor 7.25 feet (2.2 meters) away and along the entire 15-foot path from the sink to the AER.
At the sink, the technician risked drenching exposure from head to toe during most activities, and even observers positioned 3-4 feet away were found to have droplets on their gowns. In addition, saturated shoe covers reportedly tracked moisture away from the sink to the unit door – a distance of 13 feet (4 meters) – and 2 feet (0.6 meters) farther out into the PPE foyer for donning and doffing.
Although PPE gowns effectively repelled moisture during cleaning of a single device, Ms. Ofstead emphasized that technicians typically handle up to 10 instruments during a normal, 2-hour shift, further increasing exposure risk with each subsequent cleaning.
However, perhaps one of the most surprising findings was that despite an optimal unit design, including physical separation of clean and dirty activities and pressurized air flow to protect workers, droplets were still broadly dispersed.
Current efforts, however well-intentioned, might not be offering the degree of protection (and consideration) that sterile processing technicians need.
“The study was conducted in a new sterile processing area that had an extra excellent kind of distancing and three separate rooms, something that I think most of our hospitals are working toward,” Stella Hines, MD, associate professor at the University of Maryland School of Medicine, Baltimore, explained. Dr. Hines was not directly involved in the study.
“But it also really kind of highlighted what’s happening to workers potentially,” she added. “For example, we want to know if that spray or splatter has a live microbe it in that could cause a problem or ... in a highly wet environment, if that water has some kind of chemical in it that could pose an occupational hazard to the worker based on skin or mucous membrane exposure.”
Ms. Ofstead agreed. “We need to be thinking about the exposure of critically important workers and the environment in an era where we are worried about aerosol-generating procedures and superbugs,” she explained.
Dr. Hines and Ms. Ofstead also noted that the majority of staff involved in front-line patient care have never actually ventured into the sterile processing units nor do they recognize the risks that technicians working in these units face on a daily, or even hourly, basis.
“The people who run these operations are very well trained and knowledgeable. I think that it would be helpful for them to know that they’re appreciated and for the people upstairs on the front lines using the equipment to see what goes on downstairs and all of the painstaking steps that need to be in place for the equipment to come out of sterile processing and be ready to go,” said Dr. Hines.
In the meantime, hospital leaders need to address the challenges and danger posed by migrating infectious droplets, especially for workers involved in processes that stir them up in the first place – workers who by the end of their shifts are unavoidably drenched with infectious blood and tissue secretions.
“I think that it’s going to take a much bigger kind of worldview from hospital leadership,” Dr. Hines said.
The study was supported in part by a grant from Healthmark Industries. Ms. Ofstead reports research grants or consulting fees through her organization with 3M Company, Ambu, Boston Scientific, Cleanis, Fortive/Advanced Sterilization Products, Healthmark Industries, Pentax, and Steris/Cantel/Medviators. Dr. Hines reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the era of Ebola, COVID-19, and even Legionnaires, technicians and other staff working behind the scenes to ensure provider and patient safety continue to face a long-recognized but under addressed challenge: splashes and airborne droplets.
Granted, National Institute for Occupational Safety and Health (NIOSH) standards, industry standards, and professional guidelines are all in place to prevent unintentional exposure to pathogens. However, findings from a newly published study in the American Journal of Infection Control suggest they fall short.
In the study, researchers found that simulated manual cleaning of medical devices generated a drenching splash throughout the process with droplet dispersal exceeding 7 feet (2.1 meters).
Cori L. Ofstead, MSPH, lead author and president/CEO of Ofstead & Associates, Bloomington, Minn., told this news organization. “That’s the problem with having standards and guidelines that are not based on relevant evidence, [which] in this case, is a single study that was done in an intensive care area where they had an infection outbreak.”
Ms. Ofstead was referring to a report in the journal Infection Control and Hospital Epidemiology, detailing a Canadian investigation involving a multidrug-resistant Pseudomonas aeruginosa outbreak in an ICU. The report implicated the faucets over the hand hygiene sinks, with fluorescent dye showing droplet dispersal roughly 3 feet away from the sinks.
“Somehow it [the 3-feet rule] got implemented in guidelines in sterile processing decontamination areas, which are not the same as hand hygiene,’’ Ms. Ofstead explained.
With a goal of providing more current evidence on droplet generation and dispersal, as well as personal protection equipment (PPE) exposure/effectiveness, she and her colleagues simulated manual cleaning of a decommissioned colonoscope and transvaginal ultrasound probe, using for the study location a new academic sterile processing unit.
To detect droplet generation and dispersal as well as splash following common technician activities (for example, colonoscope brushing, scrubbing, rinsing and transport to an automated endoscope reprocessor [AER] for sterilization), the researchers affixed blue moisture-detection paper to environmental surfaces, on carts positioned 4 feet (1.2 meters) from the sink (to simulate observers), and along a 15-foot pathway between the sink and AER.
They observed droplets everywhere.
Technician activities such as running the faucet and rinsing the probe under running water generated substantial splashing overall. Instrument rinsing in particular produced small and large droplets and confluent puddles of water around the sink and in the broad area surrounding the workspace. Droplets were also dispersed on the floor 7.25 feet (2.2 meters) away and along the entire 15-foot path from the sink to the AER.
At the sink, the technician risked drenching exposure from head to toe during most activities, and even observers positioned 3-4 feet away were found to have droplets on their gowns. In addition, saturated shoe covers reportedly tracked moisture away from the sink to the unit door – a distance of 13 feet (4 meters) – and 2 feet (0.6 meters) farther out into the PPE foyer for donning and doffing.
Although PPE gowns effectively repelled moisture during cleaning of a single device, Ms. Ofstead emphasized that technicians typically handle up to 10 instruments during a normal, 2-hour shift, further increasing exposure risk with each subsequent cleaning.
However, perhaps one of the most surprising findings was that despite an optimal unit design, including physical separation of clean and dirty activities and pressurized air flow to protect workers, droplets were still broadly dispersed.
Current efforts, however well-intentioned, might not be offering the degree of protection (and consideration) that sterile processing technicians need.
“The study was conducted in a new sterile processing area that had an extra excellent kind of distancing and three separate rooms, something that I think most of our hospitals are working toward,” Stella Hines, MD, associate professor at the University of Maryland School of Medicine, Baltimore, explained. Dr. Hines was not directly involved in the study.
“But it also really kind of highlighted what’s happening to workers potentially,” she added. “For example, we want to know if that spray or splatter has a live microbe it in that could cause a problem or ... in a highly wet environment, if that water has some kind of chemical in it that could pose an occupational hazard to the worker based on skin or mucous membrane exposure.”
Ms. Ofstead agreed. “We need to be thinking about the exposure of critically important workers and the environment in an era where we are worried about aerosol-generating procedures and superbugs,” she explained.
Dr. Hines and Ms. Ofstead also noted that the majority of staff involved in front-line patient care have never actually ventured into the sterile processing units nor do they recognize the risks that technicians working in these units face on a daily, or even hourly, basis.
“The people who run these operations are very well trained and knowledgeable. I think that it would be helpful for them to know that they’re appreciated and for the people upstairs on the front lines using the equipment to see what goes on downstairs and all of the painstaking steps that need to be in place for the equipment to come out of sterile processing and be ready to go,” said Dr. Hines.
In the meantime, hospital leaders need to address the challenges and danger posed by migrating infectious droplets, especially for workers involved in processes that stir them up in the first place – workers who by the end of their shifts are unavoidably drenched with infectious blood and tissue secretions.
“I think that it’s going to take a much bigger kind of worldview from hospital leadership,” Dr. Hines said.
The study was supported in part by a grant from Healthmark Industries. Ms. Ofstead reports research grants or consulting fees through her organization with 3M Company, Ambu, Boston Scientific, Cleanis, Fortive/Advanced Sterilization Products, Healthmark Industries, Pentax, and Steris/Cantel/Medviators. Dr. Hines reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF INFECTION CONTROL
Is it flu, RSV, or COVID? Experts fear the ‘tripledemic’
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
Just when we thought this holiday season, finally, would be the back-to-normal one, some infectious disease experts are warning that a so-called “tripledemic” – influenza, COVID-19, and RSV – may be in the forecast.
The warning isn’t without basis.
The flu season has gotten an early start. As of Oct. 21, early increases in seasonal flu activity have been reported in most of the country, the Centers for Disease Control and Prevention said, with the southeast and south-central areas having the highest activity levels.
Children’s hospitals and EDs are seeing a surge in children with RSV.
COVID-19 cases are trending down, according to the CDC, but epidemiologists – scientists who study disease outbreaks – always have their eyes on emerging variants.
said Justin Lessler, PhD, a professor of epidemiology at the University of North Carolina at Chapel Hill. Dr. Lessler is on the coordinating team for the COVID-19 Scenario Modeling Hub, which aims to predict the course COVID-19, and the Flu Scenario Modeling Hub, which does the same for influenza.
For COVID-19, some models are predicting some spikes before Christmas, he said, and others see a new wave in 2023. For the flu, the model is predicting an earlier-than-usual start, as the CDC has reported.
While flu activity is relatively low, the CDC said, the season is off to an early start. For the week ending Oct. 21, 1,674 patients were hospitalized for flu, higher than in the summer months but fewer than the 2,675 hospitalizations for the week of May 15, 2022.
As of Oct. 20, COVID-19 cases have declined 12% over the last 2 weeks, nationwide. But hospitalizations are up 10% in much of the Northeast, The New York Times reports, and the improvement in cases and deaths has been slowing down.
As of Oct. 15, 15% of RSV tests reported nationwide were positive, compared with about 11% at that time in 2021, the CDC said. The surveillance collects information from 75 counties in 12 states.
Experts point out that the viruses – all three are respiratory viruses – are simply playing catchup.
“They spread the same way and along with lots of other viruses, and you tend to see an increase in them during the cold months,” said Timothy Brewer, MD, professor of medicine and epidemiology at UCLA.
The increase in all three viruses “is almost predictable at this point in the pandemic,” said Dean Blumberg, MD, a professor and chief of pediatric infectious diseases at the University of California Davis Health. “All the respiratory viruses are out of whack.”
Last year, RSV cases were up, too, and began to appear very early, he said, in the summer instead of in the cooler months. Flu also appeared early in 2021, as it has in 2022.
That contrasts with the flu season of 2020-2021, when COVID precautions were nearly universal, and cases were down. At UC Davis, “we didn’t have one pediatric admission due to influenza in the 2020-2021 [flu] season,” Dr. Blumberg said.
The number of pediatric flu deaths usually range from 37 to 199 per year, according to CDC records. But in the 2020-2021 season, the CDC recorded one pediatric flu death in the U.S.
Both children and adults have had less contact with others the past two seasons, Dr. Blumberg said, “and they don’t get the immunity they got with those infections [previously]. That’s why we are seeing out-of-season, early season [viruses].”
Eventually, he said, the cases of flu and RSV will return to previous levels. “It could be as soon as next year,” Dr. Blumberg said. And COVID-19, hopefully, will become like influenza, he said.
“RSV has always come around in the fall and winter,” said Elizabeth Murray, DO, a pediatric emergency medicine doctor at the University of Rochester (N.Y.) Medical Center and a spokesperson for the American Academy of Pediatrics. In 2022, children are back in school and for the most part not masking. “It’s a perfect storm for all the germs to spread now. They’ve just been waiting for their opportunity to come back.”
Self-care vs. not
RSV can pose a risk for anyone, but most at risk are children under age 5, especially infants under age 1, and adults over age 65. There is no vaccine for it. Symptoms include a runny nose, decreased appetite, coughing, sneezing, fever, and wheezing. But in young infants, there may only be decreased activity, crankiness, and breathing issues, the CDC said.
Keep an eye on the breathing if RSV is suspected, Dr. Murray tells parents. If your child can’t breathe easily, is unable to lie down comfortably, can’t speak clearly, or is sucking in the chest muscles to breathe, get medical help. Most kids with RSV can stay home and recover, she said, but often will need to be checked by a medical professional.
She advises against getting an oximeter to measure oxygen levels for home use. “They are often not accurate,” she said. If in doubt about how serious your child’s symptoms are, “don’t wait it out,” and don’t hesitate to call 911.
Symptoms of flu, COVID, and RSV can overlap. But each can involve breathing problems, which can be an emergency.
“It’s important to seek medical attention for any concerning symptoms, but especially severe shortness of breath or difficulty breathing, as these could signal the need for supplemental oxygen or other emergency interventions,” said Mandy De Vries, a respiratory therapist and director of education at the American Association for Respiratory Care. Inhalation treatment or mechanical ventilation may be needed for severe respiratory issues.
Precautions
To avoid the tripledemic – or any single infection – Timothy Brewer, MD, a professor of medicine and epidemiology at the University of California, Los Angeles, suggests some familiar measures: “Stay home if you’re feeling sick. Make sure you are up to date on your vaccinations. Wear a mask indoors.”
A version of this article first appeared on Medscape.com.
HPV-positive women who undergo IVF don’t have worse outcomes
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
FROM ASRM 2022
IgA Vasculitis in the Setting of Biologic Therapy for Psoriasis and Recurrent Cutaneous Methicillin-Resistant Staphylococcus aureus Colonization
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.
Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.
Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.
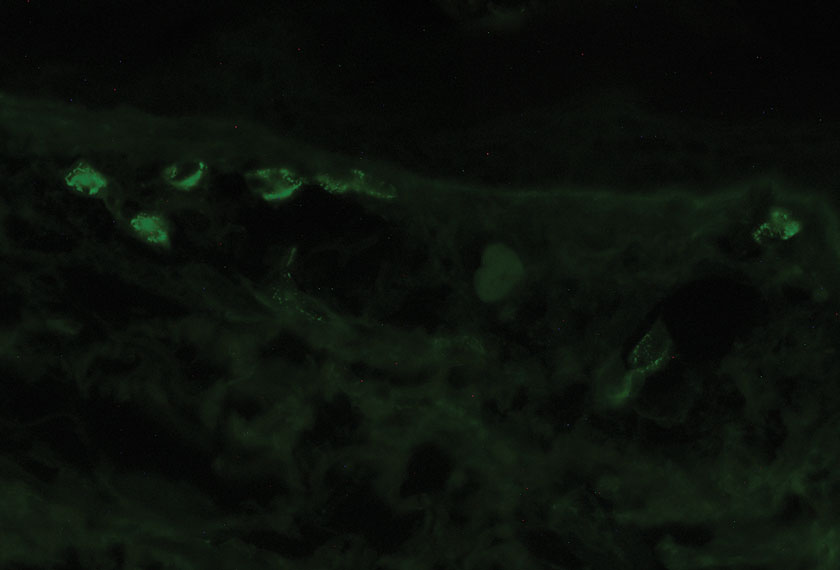
Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.
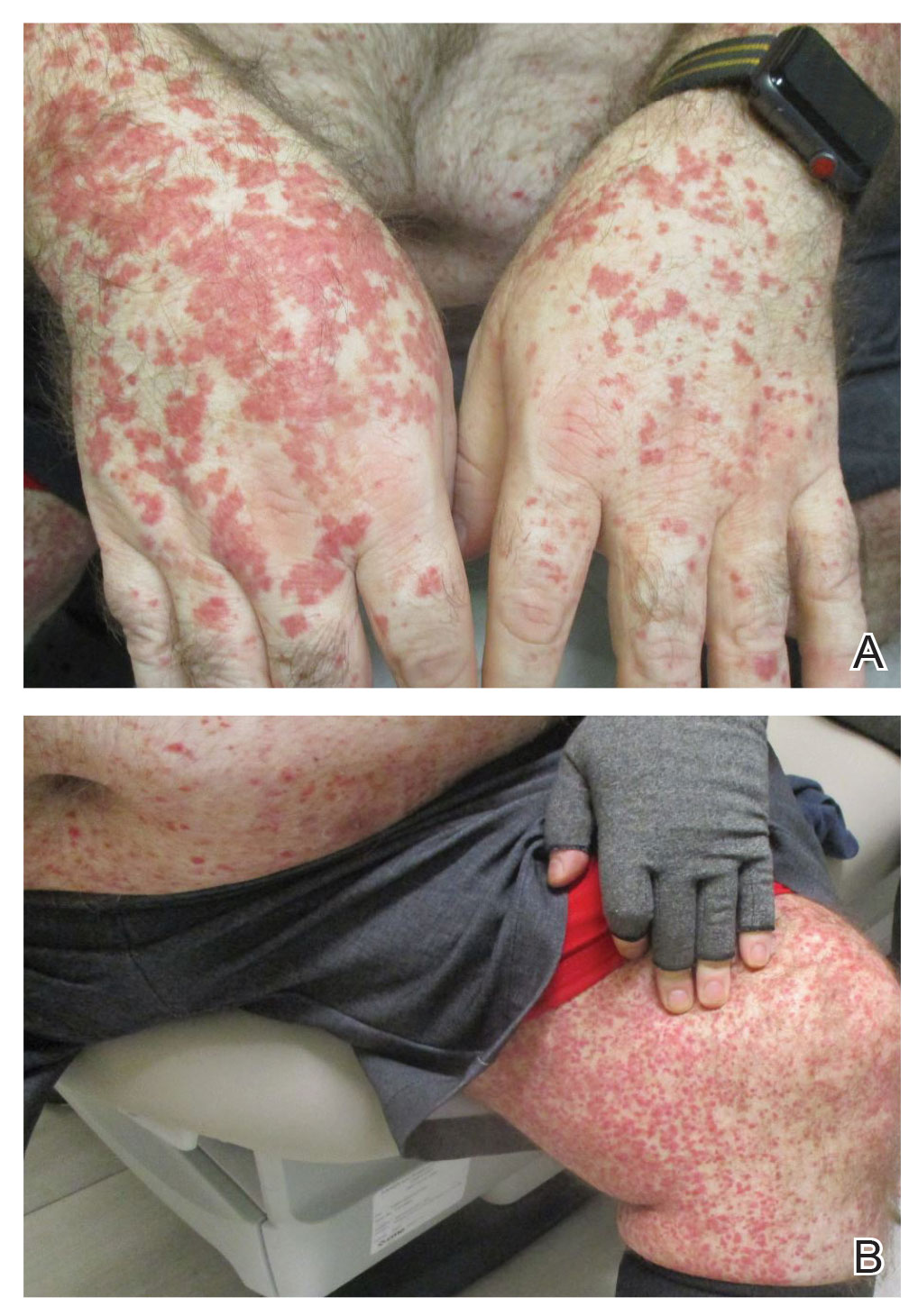
Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7
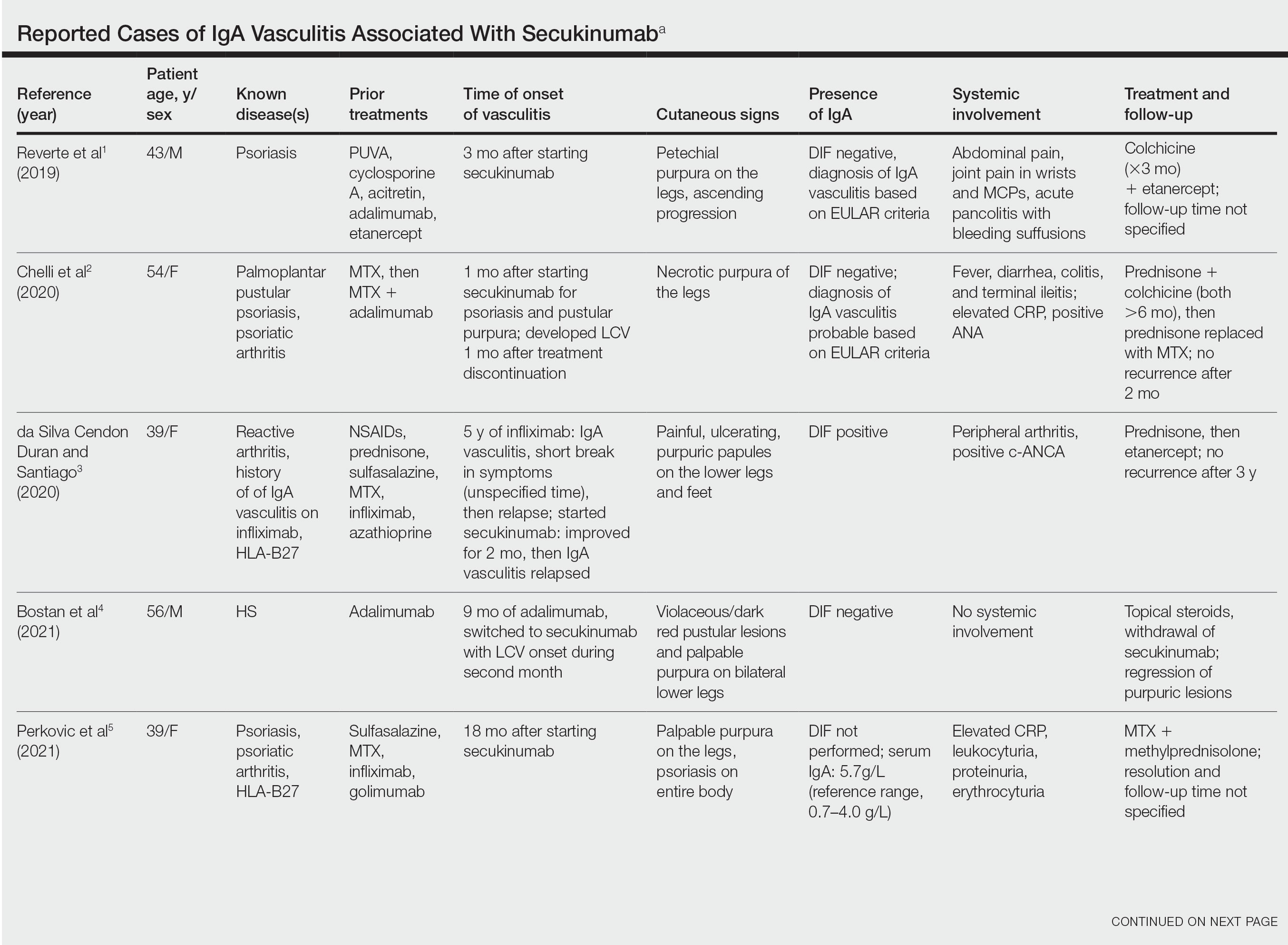
The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10
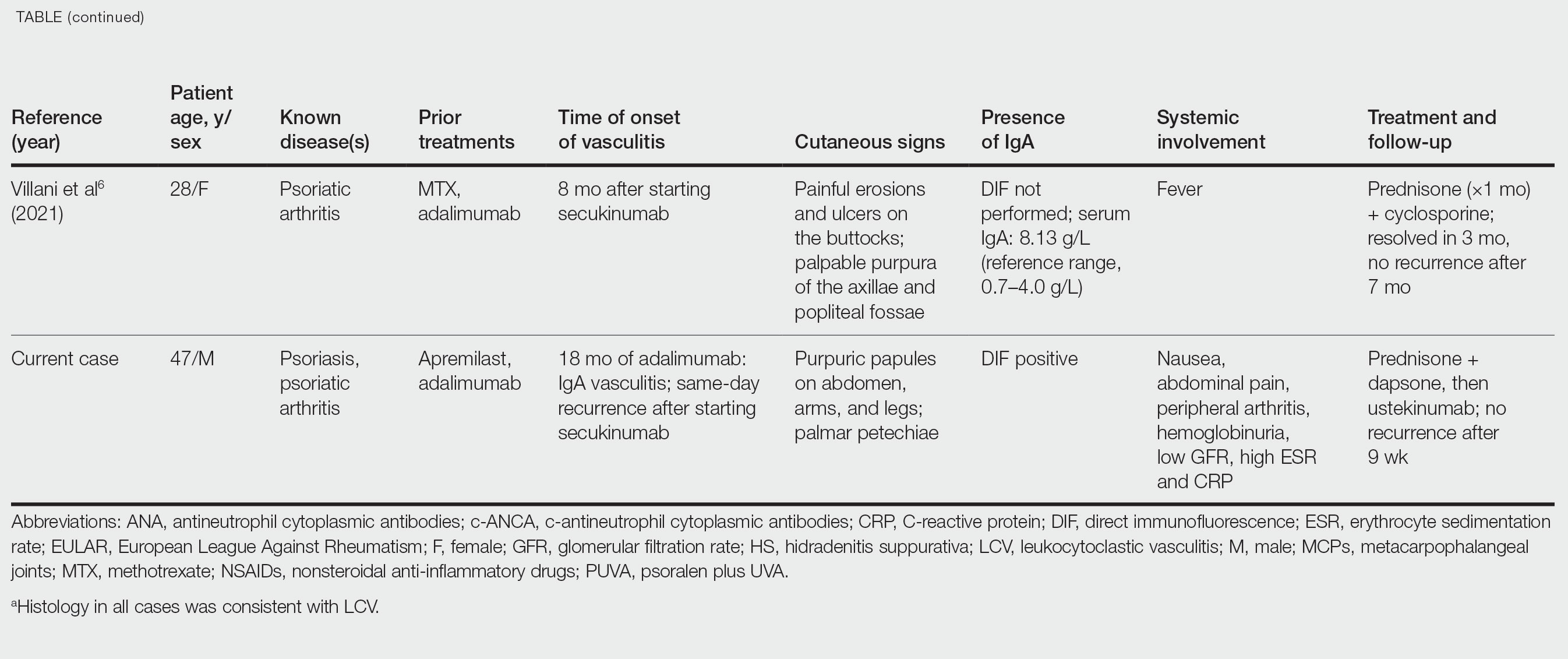
There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Practice Points
- Biologic medications including adalimumab and more rarely secukinumab may be associated with leukocytoclastic vasculitis; a smaller subset of patients may experience IgA vasculitis.
- The IL-23 blocker ustekinumab may represent an ideal therapeutic agent when secukinumabassociated vasculitis is suspected. Because IL-23 is the main driver and sustainer of TH17 cell differentiation, it may cease the main causative cytokine cascades “upstream.”