User login
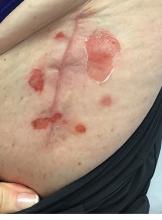
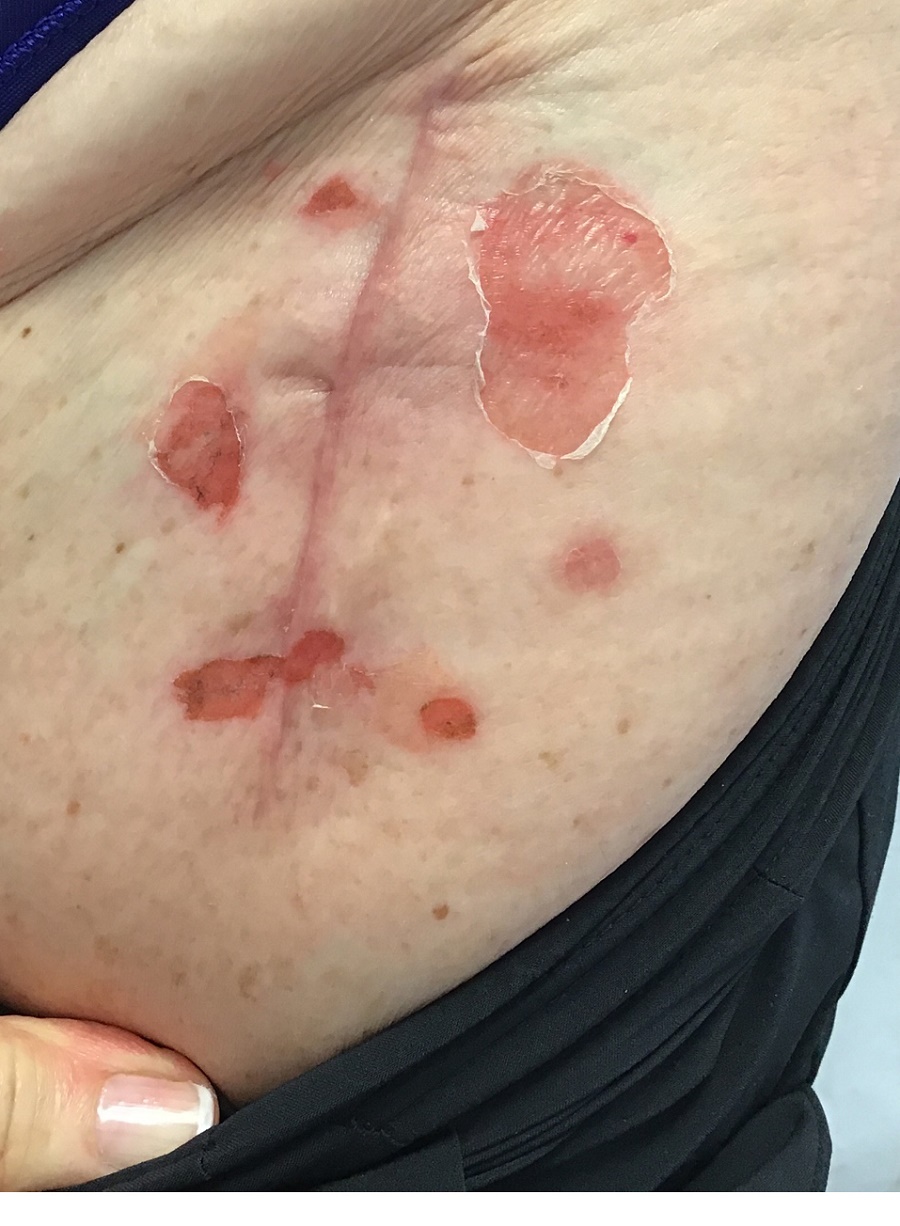
A 71-year-old White female developed erosions after hip replacement surgery 2 months prior to presentation
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.
The patient had been diagnosed with pemphigus vulgaris (PV) 1 year prior to presentation with erosions on the axilla. Biopsy at that time revealed intraepithelial acantholytic blistering with areas of suprabasilar and subcorneal clefting. Direct immunofluorescence was positive for linear/granular IgG deposition throughout the epithelial cell surfaces, as well as linear/granular C3 deposits of the lower two thirds of the epithelial strata, consistent for pemphigus vulgaris.
There is likely a genetic predisposition. Medications that may induce pemphigus include penicillamine, nifedipine, or captopril.
Clinically, PV presents with flaccid blistering lesions that may be cutaneous and/or mucosal. Bullae can progress to erosions and crusting, which then heal with pigment alteration but not scarring. The most commonly affected sites are the mouth, intertriginous areas, face, and neck. Mucosal lesions can involve the lips, esophagus, conjunctiva, and genitals.
Biopsy for histology and direct immunofluorescence is important in distinguishing between PV and other blistering disorders. Up to 75% of patients with active disease also have a positive indirect immunofluorescence with circulating IgG.
There are numerous reports in the literature of PV occurring in previous surgical scars, and areas of friction or trauma. This so-called Koebner’s phenomenon is seen more commonly in several dermatologic conditions, such as psoriasis, lichen planus, verruca vulgaris, and vitiligo.
Treatment for PV is generally immunosuppressive. Systemic therapy usually begins with prednisone and then is transitioned to a steroid sparing agent such as mycophenolate mofetil. Other steroid sparing agents include azathioprine, methotrexate, cyclophosphamide, and intravenous immunoglobulin. Secondary infections are possible and should be treated. Topical therapies aimed at reducing pain, especially in mucosal lesions, can be beneficial.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Cerottini JP et al. Eur J Dermatol. 2000 Oct-Nov;10(7):546-7.
Reichert-Penetrat S et al. Eur J Dermatol. 1998 Jan-Feb;8(1):60-2.
Saini P et al. Skinmed. 2020 Aug 1;18(4):252-253.
Hypnosis May Offer Relief During Sharp Debridement of Skin Ulcers
TOPLINE:
Hypnosis reduces pain during sharp debridement of skin ulcers in patients with immune-mediated inflammatory diseases, with most patients reporting decreased pain awareness and lasting pain relief for 2-3 days after the procedure.
METHODOLOGY:
- Researchers reported their experience with the anecdotal use of hypnosis for pain management in debridement of skin ulcers in immune-mediated inflammatory diseases.
- They studied 16 participants (14 women; mean age, 56 years; 14 with systemic sclerosis or morphea) with recurrent skin ulcerations requiring sharp debridement, who presented to a wound care clinic at the Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom. The participants had negative experiences with pharmacologic pain management.
- Participants consented to hypnosis during debridement as the only mode of analgesia, conducted by the same hypnosis-trained, experienced healthcare professional in charge of their ulcer care.
- Ulcer pain scores were recorded using a numerical rating pain scale before and immediately after debridement, with a score of 0 indicating no pain and 10 indicating worst pain.
TAKEAWAY:
- Hypnosis reduced the median pre-debridement ulcer pain score from 8 (interquartile range [IQR], 7-10) to 0.5 (IQR, 0-2) immediately after the procedure.
- Of 16 participants, 14 reported being aware of the procedure but not feeling the pain, with only two participants experiencing a brief spike in pain.
- The other two participants reported experiencing reduced awareness and being pain-free during the procedure.
- Five participants reported a lasting decrease in pain perception for 2-3 days after the procedure.
IN PRACTICE:
“These preliminary data underscore the potential for the integration of hypnosis into the management of intervention-related pain in clinical care,” the authors wrote.
SOURCE:
The study was led by Begonya Alcacer-Pitarch, PhD, Leeds Institute of Rheumatic and Musculoskeletal Medicine, the University of Leeds, and Chapel Allerton Hospital in Leeds, United Kingdom. It was published as a correspondence on September 10, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The small sample size may limit the generalizability of the findings. The methods used for data collection were not standardized, and the individuals included in the study may have introduced selection bias.
DISCLOSURES:
The study did not have a funding source. The authors declared no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Hypnosis reduces pain during sharp debridement of skin ulcers in patients with immune-mediated inflammatory diseases, with most patients reporting decreased pain awareness and lasting pain relief for 2-3 days after the procedure.
METHODOLOGY:
- Researchers reported their experience with the anecdotal use of hypnosis for pain management in debridement of skin ulcers in immune-mediated inflammatory diseases.
- They studied 16 participants (14 women; mean age, 56 years; 14 with systemic sclerosis or morphea) with recurrent skin ulcerations requiring sharp debridement, who presented to a wound care clinic at the Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom. The participants had negative experiences with pharmacologic pain management.
- Participants consented to hypnosis during debridement as the only mode of analgesia, conducted by the same hypnosis-trained, experienced healthcare professional in charge of their ulcer care.
- Ulcer pain scores were recorded using a numerical rating pain scale before and immediately after debridement, with a score of 0 indicating no pain and 10 indicating worst pain.
TAKEAWAY:
- Hypnosis reduced the median pre-debridement ulcer pain score from 8 (interquartile range [IQR], 7-10) to 0.5 (IQR, 0-2) immediately after the procedure.
- Of 16 participants, 14 reported being aware of the procedure but not feeling the pain, with only two participants experiencing a brief spike in pain.
- The other two participants reported experiencing reduced awareness and being pain-free during the procedure.
- Five participants reported a lasting decrease in pain perception for 2-3 days after the procedure.
IN PRACTICE:
“These preliminary data underscore the potential for the integration of hypnosis into the management of intervention-related pain in clinical care,” the authors wrote.
SOURCE:
The study was led by Begonya Alcacer-Pitarch, PhD, Leeds Institute of Rheumatic and Musculoskeletal Medicine, the University of Leeds, and Chapel Allerton Hospital in Leeds, United Kingdom. It was published as a correspondence on September 10, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The small sample size may limit the generalizability of the findings. The methods used for data collection were not standardized, and the individuals included in the study may have introduced selection bias.
DISCLOSURES:
The study did not have a funding source. The authors declared no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Hypnosis reduces pain during sharp debridement of skin ulcers in patients with immune-mediated inflammatory diseases, with most patients reporting decreased pain awareness and lasting pain relief for 2-3 days after the procedure.
METHODOLOGY:
- Researchers reported their experience with the anecdotal use of hypnosis for pain management in debridement of skin ulcers in immune-mediated inflammatory diseases.
- They studied 16 participants (14 women; mean age, 56 years; 14 with systemic sclerosis or morphea) with recurrent skin ulcerations requiring sharp debridement, who presented to a wound care clinic at the Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom. The participants had negative experiences with pharmacologic pain management.
- Participants consented to hypnosis during debridement as the only mode of analgesia, conducted by the same hypnosis-trained, experienced healthcare professional in charge of their ulcer care.
- Ulcer pain scores were recorded using a numerical rating pain scale before and immediately after debridement, with a score of 0 indicating no pain and 10 indicating worst pain.
TAKEAWAY:
- Hypnosis reduced the median pre-debridement ulcer pain score from 8 (interquartile range [IQR], 7-10) to 0.5 (IQR, 0-2) immediately after the procedure.
- Of 16 participants, 14 reported being aware of the procedure but not feeling the pain, with only two participants experiencing a brief spike in pain.
- The other two participants reported experiencing reduced awareness and being pain-free during the procedure.
- Five participants reported a lasting decrease in pain perception for 2-3 days after the procedure.
IN PRACTICE:
“These preliminary data underscore the potential for the integration of hypnosis into the management of intervention-related pain in clinical care,” the authors wrote.
SOURCE:
The study was led by Begonya Alcacer-Pitarch, PhD, Leeds Institute of Rheumatic and Musculoskeletal Medicine, the University of Leeds, and Chapel Allerton Hospital in Leeds, United Kingdom. It was published as a correspondence on September 10, 2024, in The Lancet Rheumatology.
LIMITATIONS:
The small sample size may limit the generalizability of the findings. The methods used for data collection were not standardized, and the individuals included in the study may have introduced selection bias.
DISCLOSURES:
The study did not have a funding source. The authors declared no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Seborrheic Dermatitis in Black Patients: New Therapies Offer Hope
NEW YORK — not only in this group but also overall, now that there is an approved therapy with an array of alternatives and adjunctive medications, according to Shawn Kwatra, MD.
The list of therapies effective against SD, often employed in combination, is lengthy, but topical 0.3% roflumilast foam (Zoryve), approved by the Food and Drug Administration (FDA) late last year for treating SD, has a high rate of efficacy and should now be considered a first-line treatment option, according to Dr. Kwatra, professor and chair of the Department of Dermatology, University of Maryland School of Medicine, Baltimore.
New Approved Therapy Draws Attention to SD
Emphasizing that topical roflumilast does not necessarily replace the use of over-the-counter shampoos and emollients or a list of prescription drugs used off-label to control this condition, he said it is also important for another reason.
“It shines a light on this disease,” said Dr. Kwatra, speaking at the 2024 Skin of Color Update. While his comments were focused primarily on individuals with darker skin, his major take home messages were broadly relevant across skin types.
He acknowledged that for years he “had not given seborrheic dermatitis the respect that it deserves” even though this condition comes after only acne and eczema as chief complaints among Black individuals seeing a dermatologist. The estimated global incidence is 5%, according to Dr. Kwatra, but he considers this estimate of an often “forgotten disease” too low.
One reason is that many individuals self-treat with over-the-counter solutions and never bring the complaint to a clinician. Dr. Kwatra said that he now looks for it routinely and points it out to patients who have come to him for another reason.
In patients with darker skin, the signs of SD can differ. While scalp involvement is generally easy to identify across skin types, the inflammation and erythema, sebum production, scaling and itch, and Malassezia that accompanies and drives SD might be missed in a patient with darker skin without specifically looking for these signs.
Skin and Gut Microbiome Involvement Suspected
The underlying causes of SD are understood as an inflammatory process involving keratinocyte disruption and proliferation that ultimately impairs skin barrier function, causes water loss, and produces scale stemming from stratum corneum, but Dr. Kwatra said that there is increasing evidence of a major role for both the skin and gut microbiome.
In regard to the skin microbiome, Malassezia has long been recognized as linked to SD and is a target of treatment, but evidence that the gut microbiome might be participating is relatively new. One clue comes from the fact that oral antifungal therapies, such as itraconazole, are known to reduce risk for SD relapse, an effect that might be a function of their ability to modulate the gut microbiome, according to Dr. Kwatra.
Topical roflumilast, a phosphodiesterase-4 inhibitor, was effective for SD in a vehicle-controlled phase 3 trial published in 2023. He characterized the adverse event profile as “pretty clean,” but he emphasized that a role for many other strategies remains. This is particularly true for challenging forms of SD. For example, topical tacrolimus provided meaningful protection against relapse over a period of more than 6 months in a 2021 trial that enrolled patients with severe facial SD.
The topical Janus kinase inhibitor ruxolitinib, 1.5%, (approved for atopic dermatitis and vitiligo) has also been reported to be effective for refractory facial SD. It is being evaluated in a phase 2 study of SD, according to Dr. Kwatra. A topical PDE4 inhibitor is also being evaluated for SD in a phase 2 study, he said.
Given the heterogeneity of the presentation of SD and the value of combining different mechanisms of action, Dr. Kwatra does not think any drug by itself will be a cure for SD. However, the chances of success with current drug combinations are high.
It is for this reason that Dr. Kwatra encourages clinicians to look for this disease routinely, including among patients who have a different presenting complaint. “Patients do not always bring it up, so bring it up,” he said.
This is good advice, according to Andrew F. Alexis, MD, MPH, professor of clinical dermatology and Vice-chair for Diversity and Inclusion of the Department of Dermatology, Weill Cornell Medicine, New York City. He agreed that the recent introduction of a therapy approved by the FDA is an impetus to look for SD and to talk with patients about treatment options.
In addition, while he also considers roflumilast foam to be a first-line drug, he agreed that combination therapies might be needed to increase the likely of rapid control of scalp and skin involvement. “SD is probably underestimated as a clinical problem, and we do have good treatments to offer for the patients who are affected,” he said at the meeting.
Dr. Kwatra reported no relevant disclosures. Dr. Alexis reported financial relationships with more than 25 pharmaceutical companies.
A version of this article appeared on Medscape.com.
NEW YORK — not only in this group but also overall, now that there is an approved therapy with an array of alternatives and adjunctive medications, according to Shawn Kwatra, MD.
The list of therapies effective against SD, often employed in combination, is lengthy, but topical 0.3% roflumilast foam (Zoryve), approved by the Food and Drug Administration (FDA) late last year for treating SD, has a high rate of efficacy and should now be considered a first-line treatment option, according to Dr. Kwatra, professor and chair of the Department of Dermatology, University of Maryland School of Medicine, Baltimore.
New Approved Therapy Draws Attention to SD
Emphasizing that topical roflumilast does not necessarily replace the use of over-the-counter shampoos and emollients or a list of prescription drugs used off-label to control this condition, he said it is also important for another reason.
“It shines a light on this disease,” said Dr. Kwatra, speaking at the 2024 Skin of Color Update. While his comments were focused primarily on individuals with darker skin, his major take home messages were broadly relevant across skin types.
He acknowledged that for years he “had not given seborrheic dermatitis the respect that it deserves” even though this condition comes after only acne and eczema as chief complaints among Black individuals seeing a dermatologist. The estimated global incidence is 5%, according to Dr. Kwatra, but he considers this estimate of an often “forgotten disease” too low.
One reason is that many individuals self-treat with over-the-counter solutions and never bring the complaint to a clinician. Dr. Kwatra said that he now looks for it routinely and points it out to patients who have come to him for another reason.
In patients with darker skin, the signs of SD can differ. While scalp involvement is generally easy to identify across skin types, the inflammation and erythema, sebum production, scaling and itch, and Malassezia that accompanies and drives SD might be missed in a patient with darker skin without specifically looking for these signs.
Skin and Gut Microbiome Involvement Suspected
The underlying causes of SD are understood as an inflammatory process involving keratinocyte disruption and proliferation that ultimately impairs skin barrier function, causes water loss, and produces scale stemming from stratum corneum, but Dr. Kwatra said that there is increasing evidence of a major role for both the skin and gut microbiome.
In regard to the skin microbiome, Malassezia has long been recognized as linked to SD and is a target of treatment, but evidence that the gut microbiome might be participating is relatively new. One clue comes from the fact that oral antifungal therapies, such as itraconazole, are known to reduce risk for SD relapse, an effect that might be a function of their ability to modulate the gut microbiome, according to Dr. Kwatra.
Topical roflumilast, a phosphodiesterase-4 inhibitor, was effective for SD in a vehicle-controlled phase 3 trial published in 2023. He characterized the adverse event profile as “pretty clean,” but he emphasized that a role for many other strategies remains. This is particularly true for challenging forms of SD. For example, topical tacrolimus provided meaningful protection against relapse over a period of more than 6 months in a 2021 trial that enrolled patients with severe facial SD.
The topical Janus kinase inhibitor ruxolitinib, 1.5%, (approved for atopic dermatitis and vitiligo) has also been reported to be effective for refractory facial SD. It is being evaluated in a phase 2 study of SD, according to Dr. Kwatra. A topical PDE4 inhibitor is also being evaluated for SD in a phase 2 study, he said.
Given the heterogeneity of the presentation of SD and the value of combining different mechanisms of action, Dr. Kwatra does not think any drug by itself will be a cure for SD. However, the chances of success with current drug combinations are high.
It is for this reason that Dr. Kwatra encourages clinicians to look for this disease routinely, including among patients who have a different presenting complaint. “Patients do not always bring it up, so bring it up,” he said.
This is good advice, according to Andrew F. Alexis, MD, MPH, professor of clinical dermatology and Vice-chair for Diversity and Inclusion of the Department of Dermatology, Weill Cornell Medicine, New York City. He agreed that the recent introduction of a therapy approved by the FDA is an impetus to look for SD and to talk with patients about treatment options.
In addition, while he also considers roflumilast foam to be a first-line drug, he agreed that combination therapies might be needed to increase the likely of rapid control of scalp and skin involvement. “SD is probably underestimated as a clinical problem, and we do have good treatments to offer for the patients who are affected,” he said at the meeting.
Dr. Kwatra reported no relevant disclosures. Dr. Alexis reported financial relationships with more than 25 pharmaceutical companies.
A version of this article appeared on Medscape.com.
NEW YORK — not only in this group but also overall, now that there is an approved therapy with an array of alternatives and adjunctive medications, according to Shawn Kwatra, MD.
The list of therapies effective against SD, often employed in combination, is lengthy, but topical 0.3% roflumilast foam (Zoryve), approved by the Food and Drug Administration (FDA) late last year for treating SD, has a high rate of efficacy and should now be considered a first-line treatment option, according to Dr. Kwatra, professor and chair of the Department of Dermatology, University of Maryland School of Medicine, Baltimore.
New Approved Therapy Draws Attention to SD
Emphasizing that topical roflumilast does not necessarily replace the use of over-the-counter shampoos and emollients or a list of prescription drugs used off-label to control this condition, he said it is also important for another reason.
“It shines a light on this disease,” said Dr. Kwatra, speaking at the 2024 Skin of Color Update. While his comments were focused primarily on individuals with darker skin, his major take home messages were broadly relevant across skin types.
He acknowledged that for years he “had not given seborrheic dermatitis the respect that it deserves” even though this condition comes after only acne and eczema as chief complaints among Black individuals seeing a dermatologist. The estimated global incidence is 5%, according to Dr. Kwatra, but he considers this estimate of an often “forgotten disease” too low.
One reason is that many individuals self-treat with over-the-counter solutions and never bring the complaint to a clinician. Dr. Kwatra said that he now looks for it routinely and points it out to patients who have come to him for another reason.
In patients with darker skin, the signs of SD can differ. While scalp involvement is generally easy to identify across skin types, the inflammation and erythema, sebum production, scaling and itch, and Malassezia that accompanies and drives SD might be missed in a patient with darker skin without specifically looking for these signs.
Skin and Gut Microbiome Involvement Suspected
The underlying causes of SD are understood as an inflammatory process involving keratinocyte disruption and proliferation that ultimately impairs skin barrier function, causes water loss, and produces scale stemming from stratum corneum, but Dr. Kwatra said that there is increasing evidence of a major role for both the skin and gut microbiome.
In regard to the skin microbiome, Malassezia has long been recognized as linked to SD and is a target of treatment, but evidence that the gut microbiome might be participating is relatively new. One clue comes from the fact that oral antifungal therapies, such as itraconazole, are known to reduce risk for SD relapse, an effect that might be a function of their ability to modulate the gut microbiome, according to Dr. Kwatra.
Topical roflumilast, a phosphodiesterase-4 inhibitor, was effective for SD in a vehicle-controlled phase 3 trial published in 2023. He characterized the adverse event profile as “pretty clean,” but he emphasized that a role for many other strategies remains. This is particularly true for challenging forms of SD. For example, topical tacrolimus provided meaningful protection against relapse over a period of more than 6 months in a 2021 trial that enrolled patients with severe facial SD.
The topical Janus kinase inhibitor ruxolitinib, 1.5%, (approved for atopic dermatitis and vitiligo) has also been reported to be effective for refractory facial SD. It is being evaluated in a phase 2 study of SD, according to Dr. Kwatra. A topical PDE4 inhibitor is also being evaluated for SD in a phase 2 study, he said.
Given the heterogeneity of the presentation of SD and the value of combining different mechanisms of action, Dr. Kwatra does not think any drug by itself will be a cure for SD. However, the chances of success with current drug combinations are high.
It is for this reason that Dr. Kwatra encourages clinicians to look for this disease routinely, including among patients who have a different presenting complaint. “Patients do not always bring it up, so bring it up,” he said.
This is good advice, according to Andrew F. Alexis, MD, MPH, professor of clinical dermatology and Vice-chair for Diversity and Inclusion of the Department of Dermatology, Weill Cornell Medicine, New York City. He agreed that the recent introduction of a therapy approved by the FDA is an impetus to look for SD and to talk with patients about treatment options.
In addition, while he also considers roflumilast foam to be a first-line drug, he agreed that combination therapies might be needed to increase the likely of rapid control of scalp and skin involvement. “SD is probably underestimated as a clinical problem, and we do have good treatments to offer for the patients who are affected,” he said at the meeting.
Dr. Kwatra reported no relevant disclosures. Dr. Alexis reported financial relationships with more than 25 pharmaceutical companies.
A version of this article appeared on Medscape.com.
FROM SOC 2024
Identifying Drug-Induced Rashes in Skin of Color: Heightened Awareness Can Accelerate Diagnosis
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
NEW YORK — Because of their heterogeneity in appearance, to speed the diagnosis.
This risk for a delayed or missed diagnosis in patients with darker skin is shared across skin rashes, but drug-induced hypersensitivity syndrome (DIHS) is a telling example, according to Joanna Harp, MD, director of the Inpatient Dermatology Consult Service, NewYork–Presbyterian Hospital, New York City.
DIHS, also known as a drug reaction with eosinophilia and systemic symptoms, is a type IV hypersensitivity reaction, Dr. Harp explained. While the fact that this disorder does not always include eosinophilia prompted the DIHS acronym, the maculopapular rash often serves as a critical clue of the underlying etiology.
In patients with darker skin, DIHS skin manifestations “can look different, can be more severe, and can have worse outcomes,” Dr. Harp said. As with other skin rashes that are primarily erythematous, the DIHS rash is often more subtle in Black-skinned patients, typically appearing gray or violaceous rather than red.
“The high amount of scale can be a clue,” said Dr. Harp, speaking at the 2024 Skin of Color Update. Scale is particularly prominent among Black patients, she said, because of the greater relative transepidermal water loss than lighter skin, increasing dryness and susceptibility to scale.
The maculopapular rash is “similar to a simple drug eruption, although it is usually more impressive,” she said. Emphasizing that DIHS is a systemic disease, she noted that the characteristic rash is typically accompanied by inflammation in multiple organs that not only includes the mucous membranes but can include major organs such as the lungs, kidneys, and heart.
In patients with DIHS and many of the even more serious types of rashes traced to drug exposures, such as Stevens-Johnson syndrome (SJS) or erythema multiforme, the delay to appearance of the rash from the time of exposure can be the most confusing element.
“It can be months for some drugs such as allopurinol,” said Dr. Harp, pointing out that Black and Asian patients are more likely to carry the HLA-B*5801 genotype, a known risk factor for allopurinol hypersensitivity.
Signs of AGEP Can Be Subtle in Black Patients
Some of the same principles for diagnosing drug-induced rash in darker skin can also be applied to acute generalized exanthematous pustulosis (AGEP), another type IV hypersensitivity reaction. Like all drug-induced rashes, the earlier AGEP is recognized and treated, the better the outcome, but in Black patients, the signs can be subtle.
“The onset is usually fast and occurs in 1-2 days after [the causative drug] exposure,” said Dr. Harp, adding that antibiotics, such as cephalosporins or penicillin, and calcium channel blockers are among the prominent causes of AGEP.
One of the hallmark signs of early-onset AGEP are tiny erythematous pustules in flexural areas, such as the neck or the armpits. The issue of detecting erythema in darker skin is also relevant to this area, but there is an additional problem, according to Dr. Harp. The pustules often dry up quickly, leaving a neutrophilic scale that further complicates the effort to see the characteristic erythema.
“If you see a lot of scale, look for erythema underneath. Think of inflammation,” Dr. Harp said, explaining that the clinical appearance evolves quickly. “If you do not see the pustules, it does not mean they were not there; you just missed them.”
In addition to the flexural areas, “AGEP loves the ears, the face, and the geographic tongue,” she said, offering several pearls to help with the diagnosis. These include side lighting to make papules easier to see, pressing on the skin to highlight the difference between erythematous skin and blanched skin, and checking less pigmented skin, such as on the hands and feet, which makes erythema easier to see.
Steroids are often the first-line treatment for drug-induced skin rashes, but Dr. Harp moves to etanercept or cyclosporine for the most serious drug reactions, such as SJS and toxic epidermal necrolysis.
Etanercept is typically her first choice because patients with systemic hypersensitivity reactions with major organ involvement are often quite ill, making cyclosporine harder to use. In her experience, etanercept has been well tolerated.
Conversely, she cautioned against the use of intravenous immunoglobulin (IVIG). Although this has been used traditionally for severe drug hypersensitivity reactions, “the data are not there,” she said. The data are stronger for a combination of high-dose steroids and IVIG, but she thinks even these data are inconsistent and not as strong as the data supporting etanercept or cyclosporine. She encouraged centers still using IVIG to consider alternatives.
After drug sensitivity reactions are controlled, follow-up care is particularly important for Black patients who face greater risks for sequelae, such as hypopigmentation, hyperpigmentation, or keloids. She recommended aggressive use of emollients and sunscreens for an extended period after lesions resolve to lessen these risks.
Differences in the manifestations of drug-induced skin rashes by race and ethnicity are important and perhaps underappreciated, agreed Shawn Kwatra, MD, professor and chairman of the Department of Dermatology, University of Maryland, Baltimore.
Asked to comment at the meeting, Dr. Kwatra said that he appreciated Dr. Harp’s effort to translate published data and her experience into an overview that increases awareness of the risk for missed or delayed diagnoses of drug-induced rashes in skin of color. He noted that the strategies to identify erythema and pustules, such as increased suspicion in skin of color and the extra steps to rule them out, such as the use of side lighting in the case of pustules for AGEP, are simple and practical.
Dr. Harp and Dr. Kwatra had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM SOC 2024
FDA Initiative Aims to Improve Diversity in Clinical Trials
NEW YORK — Underrepresentation by gender and race in major clinical trials has been a cause for complaint for decades, but the Food and Drug Administration (FDA) has drafted a regulatory solution to this issue expected to be implemented sometime in 2025.
This initiative, known as the according to Valerie M. Harvey, MD, MPH, associate clinical professor, Edward Via College of Osteopathic Medicine, Blacksburg, Virginia. These rules will be codified, she said at the 2024 Skin of Color Update.
Once the DAP is enacted, “the sponsor must specify the rationale and goals for study enrollment by age, ethnicity, sex, and race,” she said. Furthermore, the submission to the FDA must “describe the methods to meet the diversity benchmarks.”
Lack of Trial Diversity Is Common Across Medicine
Although she focused on the relevance of this initiative to dermatology, Dr. Harvey said the lack of diversity in clinical trials is pervasive throughout medicine. In one survey of randomized controlled trials, less than 60% of trials even specified the race and ethnicity of the participants. In recent psoriasis trials, only 30% met a diversity definition of ≥ 20% of patients identifying as minority (Black, Hispanic, Asian, or other non-White group), said Dr. Harvey, who practices dermatology in Newport News, Virginia.
The FDA draft guidance for the DAP was released in June 2024 and is now available for submitting comments (until September 26). The plan is expected to be published in June 2025, according to Dr. Harvey. It will pertain to all pivotal and phase 3 trials enrolling 180 days after the publication date and will be relevant to all drugs and biologics as well as certain devices.
This initiative could be a critical step toward ensuring diversity in major clinical trials after years of stagnation, Dr. Harvey said, noting that despite repeated calls for more diversity in clinical trials, the literature suggests “little progress.”
However, she said that increasing diversity in clinical trials is just one step toward gathering data about the generalizability of efficacy and safety across racial and ethnic groups. A much more complex issue involves how race and ethnicity are defined in order to understand differences, if any, for efficacy and risk.
“Race is a dynamic social construct and a poor measure for biologic variation and skin color,” Dr. Harvey said. This means that work is needed to address the more complex issue of race and ethnicity stratification that will help clinicians understand the relative benefits and risks for the drugs in these trials.
Rather than differences based on genetic or other sources of biologic differences, she said, outcomes by race alone are often suspected of reflecting disparities in access to healthcare rather than a difference in therapeutic response.
Skin Color Is Inadequate to Define Race
When stratifying patients by race or ethnicity, Dr. Harvey said that “we have to be very, very careful in considering the study purpose and what the study question is.” A study attempting to compare benefits and risks among subgroups by race or ethnicity will require descriptors beyond skin color.
The recognized limitations of measuring skin tone as a surrogate of race are one reason for widespread interest in moving away from the Fitzpatrick skin type (FST) rating that has been widely considered a standard, according to Dr. Harvey. Several alternatives have been proposed, including the Monk Skin Tone Scale, the Individual Typology Angle, and the Eumelanin Human Skin Color Scale, but she cautioned that these are less well validated and generally have the limitations of the FST.
If skin color was ever useful for grouping individuals on the basis of shared physiology, growing rates of intermarriage and immigration have made skin color increasingly irrelevant to racial identity. If the goal is to evaluate the safety and efficacy of drugs across racial groups and ethnicities, the characterization of populations will almost certainly require multiple descriptors and biomarkers, she said.
“It is very important to have many tools for characterizing patients by skin type,” Susan Taylor, MD, professor of dermatology and vice chair for diversity, equity, and inclusion for the Department of Dermatology, University of Pennsylvania, Philadelphia, said in an interview at the meeting.
The reason is “there are limitations to all of them,” she said, noting also that the questions being asked about how and if skin color and race are relevant to therapeutic options differ by the question, such as innate response or access to care.
Dr. Taylor is part of a workshop that she said is evaluating a combination of instruments for characterizing skin color and race in ways relevant to the specific question being asked.
The solutions might differ. While simple clinical assessments involving skin color might be made with methods captured on a smartphone app, Dr. Taylor acknowledged that far more complex tools might be required to document the effect of racial or ethnic differences in drug efficacy and safety in a research setting.
Outside of a research setting, any tools that might be useful for assessing race as a variable must be practical, according to Dr. Harvey. She suggested that these must be time efficient, of reasonable cost, and most importantly, reliable.
Tools meeting these criteria do not currently exist, but Dr. Harvey said the work is underway. She expects a “top-down” collaborative approach to validate alternatives to the FST. If such tools can be developed with buy-in from the FDA, they might be particularly useful for translating trial data to patient care, she added.
Dr. Harvey reported financial relationships with AbbVie, Bristol-Myers Squibb, Janssen, Johnson & Johnson, L’Oréal, and SkinCeuticals. Dr. Taylor, president-elect of the American Academy of Dermatology, reported financial relationships with more than 25 pharmaceutical and cosmetic companies.
A version of this article appeared on Medscape.com.
NEW YORK — Underrepresentation by gender and race in major clinical trials has been a cause for complaint for decades, but the Food and Drug Administration (FDA) has drafted a regulatory solution to this issue expected to be implemented sometime in 2025.
This initiative, known as the according to Valerie M. Harvey, MD, MPH, associate clinical professor, Edward Via College of Osteopathic Medicine, Blacksburg, Virginia. These rules will be codified, she said at the 2024 Skin of Color Update.
Once the DAP is enacted, “the sponsor must specify the rationale and goals for study enrollment by age, ethnicity, sex, and race,” she said. Furthermore, the submission to the FDA must “describe the methods to meet the diversity benchmarks.”
Lack of Trial Diversity Is Common Across Medicine
Although she focused on the relevance of this initiative to dermatology, Dr. Harvey said the lack of diversity in clinical trials is pervasive throughout medicine. In one survey of randomized controlled trials, less than 60% of trials even specified the race and ethnicity of the participants. In recent psoriasis trials, only 30% met a diversity definition of ≥ 20% of patients identifying as minority (Black, Hispanic, Asian, or other non-White group), said Dr. Harvey, who practices dermatology in Newport News, Virginia.
The FDA draft guidance for the DAP was released in June 2024 and is now available for submitting comments (until September 26). The plan is expected to be published in June 2025, according to Dr. Harvey. It will pertain to all pivotal and phase 3 trials enrolling 180 days after the publication date and will be relevant to all drugs and biologics as well as certain devices.
This initiative could be a critical step toward ensuring diversity in major clinical trials after years of stagnation, Dr. Harvey said, noting that despite repeated calls for more diversity in clinical trials, the literature suggests “little progress.”
However, she said that increasing diversity in clinical trials is just one step toward gathering data about the generalizability of efficacy and safety across racial and ethnic groups. A much more complex issue involves how race and ethnicity are defined in order to understand differences, if any, for efficacy and risk.
“Race is a dynamic social construct and a poor measure for biologic variation and skin color,” Dr. Harvey said. This means that work is needed to address the more complex issue of race and ethnicity stratification that will help clinicians understand the relative benefits and risks for the drugs in these trials.
Rather than differences based on genetic or other sources of biologic differences, she said, outcomes by race alone are often suspected of reflecting disparities in access to healthcare rather than a difference in therapeutic response.
Skin Color Is Inadequate to Define Race
When stratifying patients by race or ethnicity, Dr. Harvey said that “we have to be very, very careful in considering the study purpose and what the study question is.” A study attempting to compare benefits and risks among subgroups by race or ethnicity will require descriptors beyond skin color.
The recognized limitations of measuring skin tone as a surrogate of race are one reason for widespread interest in moving away from the Fitzpatrick skin type (FST) rating that has been widely considered a standard, according to Dr. Harvey. Several alternatives have been proposed, including the Monk Skin Tone Scale, the Individual Typology Angle, and the Eumelanin Human Skin Color Scale, but she cautioned that these are less well validated and generally have the limitations of the FST.
If skin color was ever useful for grouping individuals on the basis of shared physiology, growing rates of intermarriage and immigration have made skin color increasingly irrelevant to racial identity. If the goal is to evaluate the safety and efficacy of drugs across racial groups and ethnicities, the characterization of populations will almost certainly require multiple descriptors and biomarkers, she said.
“It is very important to have many tools for characterizing patients by skin type,” Susan Taylor, MD, professor of dermatology and vice chair for diversity, equity, and inclusion for the Department of Dermatology, University of Pennsylvania, Philadelphia, said in an interview at the meeting.
The reason is “there are limitations to all of them,” she said, noting also that the questions being asked about how and if skin color and race are relevant to therapeutic options differ by the question, such as innate response or access to care.
Dr. Taylor is part of a workshop that she said is evaluating a combination of instruments for characterizing skin color and race in ways relevant to the specific question being asked.
The solutions might differ. While simple clinical assessments involving skin color might be made with methods captured on a smartphone app, Dr. Taylor acknowledged that far more complex tools might be required to document the effect of racial or ethnic differences in drug efficacy and safety in a research setting.
Outside of a research setting, any tools that might be useful for assessing race as a variable must be practical, according to Dr. Harvey. She suggested that these must be time efficient, of reasonable cost, and most importantly, reliable.
Tools meeting these criteria do not currently exist, but Dr. Harvey said the work is underway. She expects a “top-down” collaborative approach to validate alternatives to the FST. If such tools can be developed with buy-in from the FDA, they might be particularly useful for translating trial data to patient care, she added.
Dr. Harvey reported financial relationships with AbbVie, Bristol-Myers Squibb, Janssen, Johnson & Johnson, L’Oréal, and SkinCeuticals. Dr. Taylor, president-elect of the American Academy of Dermatology, reported financial relationships with more than 25 pharmaceutical and cosmetic companies.
A version of this article appeared on Medscape.com.
NEW YORK — Underrepresentation by gender and race in major clinical trials has been a cause for complaint for decades, but the Food and Drug Administration (FDA) has drafted a regulatory solution to this issue expected to be implemented sometime in 2025.
This initiative, known as the according to Valerie M. Harvey, MD, MPH, associate clinical professor, Edward Via College of Osteopathic Medicine, Blacksburg, Virginia. These rules will be codified, she said at the 2024 Skin of Color Update.
Once the DAP is enacted, “the sponsor must specify the rationale and goals for study enrollment by age, ethnicity, sex, and race,” she said. Furthermore, the submission to the FDA must “describe the methods to meet the diversity benchmarks.”
Lack of Trial Diversity Is Common Across Medicine
Although she focused on the relevance of this initiative to dermatology, Dr. Harvey said the lack of diversity in clinical trials is pervasive throughout medicine. In one survey of randomized controlled trials, less than 60% of trials even specified the race and ethnicity of the participants. In recent psoriasis trials, only 30% met a diversity definition of ≥ 20% of patients identifying as minority (Black, Hispanic, Asian, or other non-White group), said Dr. Harvey, who practices dermatology in Newport News, Virginia.
The FDA draft guidance for the DAP was released in June 2024 and is now available for submitting comments (until September 26). The plan is expected to be published in June 2025, according to Dr. Harvey. It will pertain to all pivotal and phase 3 trials enrolling 180 days after the publication date and will be relevant to all drugs and biologics as well as certain devices.
This initiative could be a critical step toward ensuring diversity in major clinical trials after years of stagnation, Dr. Harvey said, noting that despite repeated calls for more diversity in clinical trials, the literature suggests “little progress.”
However, she said that increasing diversity in clinical trials is just one step toward gathering data about the generalizability of efficacy and safety across racial and ethnic groups. A much more complex issue involves how race and ethnicity are defined in order to understand differences, if any, for efficacy and risk.
“Race is a dynamic social construct and a poor measure for biologic variation and skin color,” Dr. Harvey said. This means that work is needed to address the more complex issue of race and ethnicity stratification that will help clinicians understand the relative benefits and risks for the drugs in these trials.
Rather than differences based on genetic or other sources of biologic differences, she said, outcomes by race alone are often suspected of reflecting disparities in access to healthcare rather than a difference in therapeutic response.
Skin Color Is Inadequate to Define Race
When stratifying patients by race or ethnicity, Dr. Harvey said that “we have to be very, very careful in considering the study purpose and what the study question is.” A study attempting to compare benefits and risks among subgroups by race or ethnicity will require descriptors beyond skin color.
The recognized limitations of measuring skin tone as a surrogate of race are one reason for widespread interest in moving away from the Fitzpatrick skin type (FST) rating that has been widely considered a standard, according to Dr. Harvey. Several alternatives have been proposed, including the Monk Skin Tone Scale, the Individual Typology Angle, and the Eumelanin Human Skin Color Scale, but she cautioned that these are less well validated and generally have the limitations of the FST.
If skin color was ever useful for grouping individuals on the basis of shared physiology, growing rates of intermarriage and immigration have made skin color increasingly irrelevant to racial identity. If the goal is to evaluate the safety and efficacy of drugs across racial groups and ethnicities, the characterization of populations will almost certainly require multiple descriptors and biomarkers, she said.
“It is very important to have many tools for characterizing patients by skin type,” Susan Taylor, MD, professor of dermatology and vice chair for diversity, equity, and inclusion for the Department of Dermatology, University of Pennsylvania, Philadelphia, said in an interview at the meeting.
The reason is “there are limitations to all of them,” she said, noting also that the questions being asked about how and if skin color and race are relevant to therapeutic options differ by the question, such as innate response or access to care.
Dr. Taylor is part of a workshop that she said is evaluating a combination of instruments for characterizing skin color and race in ways relevant to the specific question being asked.
The solutions might differ. While simple clinical assessments involving skin color might be made with methods captured on a smartphone app, Dr. Taylor acknowledged that far more complex tools might be required to document the effect of racial or ethnic differences in drug efficacy and safety in a research setting.
Outside of a research setting, any tools that might be useful for assessing race as a variable must be practical, according to Dr. Harvey. She suggested that these must be time efficient, of reasonable cost, and most importantly, reliable.
Tools meeting these criteria do not currently exist, but Dr. Harvey said the work is underway. She expects a “top-down” collaborative approach to validate alternatives to the FST. If such tools can be developed with buy-in from the FDA, they might be particularly useful for translating trial data to patient care, she added.
Dr. Harvey reported financial relationships with AbbVie, Bristol-Myers Squibb, Janssen, Johnson & Johnson, L’Oréal, and SkinCeuticals. Dr. Taylor, president-elect of the American Academy of Dermatology, reported financial relationships with more than 25 pharmaceutical and cosmetic companies.
A version of this article appeared on Medscape.com.
FROM SOC 2024
Expert Warns of Problems with Large Language Models in Dermatology
HUNTINGTON BEACH, CALIF. — and alarmed.
Of the 134 respondents who completed the survey, 87 (65%) reported using LLMs in a clinical setting. Of those 87 respondents, 17 (20%) used LMMs daily, 28 (32%) weekly, 5 (6%) monthly, and 37 (43%) rarely. That represents “pretty significant usage,” Dr. Daneshjou, assistant professor of biomedical data science and dermatology at Stanford University, Palo Alto, California, said at the annual meeting of the Pacific Dermatologic Association.
Most of the respondents reported using LLMs for patient care (79%), followed by administrative tasks (74%), medical records (43%), and education (18%), “which can be problematic,” she said. “These models are not appropriate to use for patient care.”
When asked about their thoughts on the accuracy of LLMs, 58% of respondents deemed them to be “somewhat accurate” and 7% viewed them as “extremely accurate.”
The overall survey responses raise concern because LLMs “are not trained for accuracy; they are trained initially as a next-word predictor on large bodies of tech data,” Dr. Daneshjou said. “LLMs are already being implemented but have the potential to cause harm and bias, and I believe they will if we implement them the way things are rolling out right now. I don’t understand why we’re implementing something without any clinical trial or showing that it improves care before we throw untested technology into our healthcare system.”
Meanwhile, Epic and Microsoft are collaborating to bring AI technology to electronic health records, she said, and Epic is building more than 100 new AI features for physicians and patients. “I think it’s important for every physician and trainee to understand what is going on in the realm of AI,” said Dr. Daneshjou, who is an associate editor for the monthly journal NEJM AI. “Be involved in the conversation because we are the clinical experts, and a lot of people making decisions and building tools do not have the clinical expertise.”
To further illustrate her concerns, Dr. Daneshjou referenced a red teaming event she and her colleagues held with computer scientists, biomedical data scientists, engineers, and physicians across multiple specialties to identify issues related to safety, bias, factual errors, and/or security issues in GPT-3.5, GPT-4, and GPT-4 with internet. The goal was to mimic clinical health scenarios, ask the LLM to respond, and have the team members review the accuracy of LLM responses.
The participants found that nearly 20% of LLM responses were inappropriate. For example, in one task, an LLM was asked to calculate a RegiSCAR score for Drug Reaction With Eosinophilia and Systemic Symptoms for a patient, but the response included an incorrect score for eosinophilia. “That’s why these tools can be so dangerous because you’re reading along and everything seems right, but there might be something so minor that can impact patient care and you might miss it,” Dr. Daneshjou said.
On a related note, she advised against dermatologists uploading images into GPT-4 Vision, an LLM that can analyze images and provide textual responses to questions about them, and she recommends not using GPT-4 Vision for any diagnostic support. At this time, “GPT-4 Vision overcalls malignancies, and the specificity and sensitivity are not very good,” she explained.
Dr. Daneshjou disclosed that she has served as an adviser to MDalgorithms and Revea and has received consulting fees from Pfizer, L’Oréal, Frazier Healthcare Partners, and DWA and research funding from UCB.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIF. — and alarmed.
Of the 134 respondents who completed the survey, 87 (65%) reported using LLMs in a clinical setting. Of those 87 respondents, 17 (20%) used LMMs daily, 28 (32%) weekly, 5 (6%) monthly, and 37 (43%) rarely. That represents “pretty significant usage,” Dr. Daneshjou, assistant professor of biomedical data science and dermatology at Stanford University, Palo Alto, California, said at the annual meeting of the Pacific Dermatologic Association.
Most of the respondents reported using LLMs for patient care (79%), followed by administrative tasks (74%), medical records (43%), and education (18%), “which can be problematic,” she said. “These models are not appropriate to use for patient care.”
When asked about their thoughts on the accuracy of LLMs, 58% of respondents deemed them to be “somewhat accurate” and 7% viewed them as “extremely accurate.”
The overall survey responses raise concern because LLMs “are not trained for accuracy; they are trained initially as a next-word predictor on large bodies of tech data,” Dr. Daneshjou said. “LLMs are already being implemented but have the potential to cause harm and bias, and I believe they will if we implement them the way things are rolling out right now. I don’t understand why we’re implementing something without any clinical trial or showing that it improves care before we throw untested technology into our healthcare system.”
Meanwhile, Epic and Microsoft are collaborating to bring AI technology to electronic health records, she said, and Epic is building more than 100 new AI features for physicians and patients. “I think it’s important for every physician and trainee to understand what is going on in the realm of AI,” said Dr. Daneshjou, who is an associate editor for the monthly journal NEJM AI. “Be involved in the conversation because we are the clinical experts, and a lot of people making decisions and building tools do not have the clinical expertise.”
To further illustrate her concerns, Dr. Daneshjou referenced a red teaming event she and her colleagues held with computer scientists, biomedical data scientists, engineers, and physicians across multiple specialties to identify issues related to safety, bias, factual errors, and/or security issues in GPT-3.5, GPT-4, and GPT-4 with internet. The goal was to mimic clinical health scenarios, ask the LLM to respond, and have the team members review the accuracy of LLM responses.
The participants found that nearly 20% of LLM responses were inappropriate. For example, in one task, an LLM was asked to calculate a RegiSCAR score for Drug Reaction With Eosinophilia and Systemic Symptoms for a patient, but the response included an incorrect score for eosinophilia. “That’s why these tools can be so dangerous because you’re reading along and everything seems right, but there might be something so minor that can impact patient care and you might miss it,” Dr. Daneshjou said.
On a related note, she advised against dermatologists uploading images into GPT-4 Vision, an LLM that can analyze images and provide textual responses to questions about them, and she recommends not using GPT-4 Vision for any diagnostic support. At this time, “GPT-4 Vision overcalls malignancies, and the specificity and sensitivity are not very good,” she explained.
Dr. Daneshjou disclosed that she has served as an adviser to MDalgorithms and Revea and has received consulting fees from Pfizer, L’Oréal, Frazier Healthcare Partners, and DWA and research funding from UCB.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIF. — and alarmed.
Of the 134 respondents who completed the survey, 87 (65%) reported using LLMs in a clinical setting. Of those 87 respondents, 17 (20%) used LMMs daily, 28 (32%) weekly, 5 (6%) monthly, and 37 (43%) rarely. That represents “pretty significant usage,” Dr. Daneshjou, assistant professor of biomedical data science and dermatology at Stanford University, Palo Alto, California, said at the annual meeting of the Pacific Dermatologic Association.
Most of the respondents reported using LLMs for patient care (79%), followed by administrative tasks (74%), medical records (43%), and education (18%), “which can be problematic,” she said. “These models are not appropriate to use for patient care.”
When asked about their thoughts on the accuracy of LLMs, 58% of respondents deemed them to be “somewhat accurate” and 7% viewed them as “extremely accurate.”
The overall survey responses raise concern because LLMs “are not trained for accuracy; they are trained initially as a next-word predictor on large bodies of tech data,” Dr. Daneshjou said. “LLMs are already being implemented but have the potential to cause harm and bias, and I believe they will if we implement them the way things are rolling out right now. I don’t understand why we’re implementing something without any clinical trial or showing that it improves care before we throw untested technology into our healthcare system.”
Meanwhile, Epic and Microsoft are collaborating to bring AI technology to electronic health records, she said, and Epic is building more than 100 new AI features for physicians and patients. “I think it’s important for every physician and trainee to understand what is going on in the realm of AI,” said Dr. Daneshjou, who is an associate editor for the monthly journal NEJM AI. “Be involved in the conversation because we are the clinical experts, and a lot of people making decisions and building tools do not have the clinical expertise.”
To further illustrate her concerns, Dr. Daneshjou referenced a red teaming event she and her colleagues held with computer scientists, biomedical data scientists, engineers, and physicians across multiple specialties to identify issues related to safety, bias, factual errors, and/or security issues in GPT-3.5, GPT-4, and GPT-4 with internet. The goal was to mimic clinical health scenarios, ask the LLM to respond, and have the team members review the accuracy of LLM responses.
The participants found that nearly 20% of LLM responses were inappropriate. For example, in one task, an LLM was asked to calculate a RegiSCAR score for Drug Reaction With Eosinophilia and Systemic Symptoms for a patient, but the response included an incorrect score for eosinophilia. “That’s why these tools can be so dangerous because you’re reading along and everything seems right, but there might be something so minor that can impact patient care and you might miss it,” Dr. Daneshjou said.
On a related note, she advised against dermatologists uploading images into GPT-4 Vision, an LLM that can analyze images and provide textual responses to questions about them, and she recommends not using GPT-4 Vision for any diagnostic support. At this time, “GPT-4 Vision overcalls malignancies, and the specificity and sensitivity are not very good,” she explained.
Dr. Daneshjou disclosed that she has served as an adviser to MDalgorithms and Revea and has received consulting fees from Pfizer, L’Oréal, Frazier Healthcare Partners, and DWA and research funding from UCB.
A version of this article first appeared on Medscape.com.
FROM PDA 2024
Laser, Radiofrequency Therapies Offer Little Benefit for Genitourinary Syndrome of Menopause
CHICAGO — Use of CO2 lasers and similar “energy-based” treatments result in little to no benefit for genitourinary syndrome of menopause (GSM) symptoms, according to research presented at the The Menopause Society 2024 Annual Meeting in Chicago on September 12.
“There was a concern that menopausal women are being targeted for treatments that may not have a lot of benefit and might have significant harms,” Elisheva Danan, MD, MPH, a physician at the Minneapolis VA Health Care System and an assistant professor of medicine at the University of Minnesota Medical School in Minneapolis, told this news organization. While she was not surprised to find little evidence of benefit, “we were a little bit surprised that we also didn’t find significant evidence of harms.”
The study was unable to evaluate the potential for financial harms, but Dr. Danan noted that these therapies are often expensive and not typically covered by insurance. The treatments appear to be used primarily in private practice, she said, while “most academic clinicians were not familiar with these and do not use these lasers.”
The American Urological Association had requested the review, Dr. Danan said, “to inform clinical guidelines that they could put out for practitioners about treating genital urinary syndrome from menopause.” Yet the evidence available remains slim. “There’s a lot of outcomes that were not looked at by most of these [trials], or they were looked at in a way that we couldn’t separate out,” she said.
Kamalini Das, MD, a professor of ob.gyn. at the University of Minnesota who was not involved in the research, was surprised by the findings because studies to date have been variable, “but since this looks at multiple studies and they find no benefits, I would take these results as more significant than any of the small studies,” she told this news organization.
Dr. Das said she has patients who ask about using these therapies and have had them done. “So far, I’ve told them the jury is out on whether it will help or not, that there are some studies that say they’re beneficial and some studies that they’re not,” Dr. Das said.
But this new review changes what she will tell patients going forward, she said. “This is a good study because it consolidates lots of little studies, so I think I would use this to say, looking at all the studies together, this treatment is not beneficial.”
GSM occurs due to the body’s reduced production of estrogen and affects anywhere from 27% to 84% of postmenopausal women. It can involve a constellation of symptoms ranging from vaginal discomfort and irritation to painful urination or intercourse. Typical recommended treatments for GSM include systemic hormone therapy, localized hormonal treatments such as vaginal estrogen or dehydroepiandrosterone, nonhormonal creams and moisturizers, and the prescription drug ospemifene.
Most of these have been found effective, according to a recent systematic review Dr. Danan published in the Annals of Internal Medicine that this news organization covered. But recent years have also seen a rapid increase in interest and the availability of energy-based treatments for GSM, such as CO2 laser and radiofrequency interventions, particularly for those who cannot or do not want to use hormonal treatments. The idea behind these newer therapies is that they “heat tissue to cause a denaturation of collagen fibers and induce a wound-healing response,” with the aim of “enhancement of vaginal elasticity, restoration of premenopausal epithelial function, and symptom improvement,” the authors wrote.
Evidence has been scant and uneven for the safety and effectiveness of these treatments, and they have not been evaluated by the US Food and Drug Administration. The agency issued a warning in 2018 with remarks from then Commissioner Scott Gottlieb that the “products have serious risks and don’t have adequate evidence to support their use for these purposes.”
Much of the evidence has focused on CO2 lasers instead of other energy-based treatments, however, and a raft of new studies have been published on these interventions in the past 2 years. Dr. Danan and colleagues, therefore, assessed the most current state of the research with a systematic review of randomized controlled trials (RCTs) and prospective observational studies with control groups published through December 11, 2023.
Included studies needed to evaluate an energy-based treatment for at least 8 weeks in a minimum of 40 postmenopausal women (20 in each group) who had one or more GSM symptoms. The authors also included nonrandomized and uncontrolled studies with a follow-up of a year or more to assess possible adverse events. The studies also needed to assess at least one of eight core outcomes: Dyspareunia; vulvovaginal dryness; vulvovaginal discomfort/irritation; dysuria; change in most bothersome symptom; treatment satisfaction; adverse events; and distress, bother, or interference associated with genitourinary symptoms.
The authors identified 32 studies, including 16 RCTs, one quasi-RCT, and 15 nonrandomized studies. The researchers extracted and analyzed data from the 10 RCTs and one quasi-RCT that were rated as having low to moderate risk for bias.
Most of these studies assessed CO2 lasers alone, while three assessed erbium:yttrium-aluminum-garnet (Er:YAG) laser, and one looked at CO2 lasers vs radiofrequency treatments.
The average age of participants ranged from 56 to 64 years, and most trials were in the United States. Results showed that CO2 lasers led to little or no difference in dysuria, dyspareunia, or quality of life when compared with sham lasers. The CO2 laser therapy also showed little to no difference compared with vaginal estrogen creams for dyspareunia, dryness, discomfort/irritation, dysuria, or quality of life.
Most CO2 laser studies reported on most outcomes, but the Er:YAG studies tended to report only on quality of life and/or one or two other outcomes. The radiofrequency study only assessed dyspareunia and quality of life.
“Treatment effects on other outcomes and effects of Er:YAG laser or radiofrequency on any outcomes are very uncertain,” the authors reported. Few adverse events and no serious adverse events were reported based on 15 studies, including the additional non-RCTs that had follow-up for at least a year.
“There are case reports and other types of studies that have shown some bad outcomes using laser therapies, and we really wanted to be expansive and include anything, especially because this is such a new treatment and all these trials were in the last couple of years,” Dr. Danan said.
The review was limited by inconsistent or nonvalidated outcome reporting in the studies as well as small populations and short follow-up, typically less than 3 months.
The research was funded by the Agency for Healthcare Research and Quality and Patient-Centered Outcomes Research Institute. Dr. Danan and Dr. Das had no disclosures.
A version of this article first appeared on Medscape.com.
CHICAGO — Use of CO2 lasers and similar “energy-based” treatments result in little to no benefit for genitourinary syndrome of menopause (GSM) symptoms, according to research presented at the The Menopause Society 2024 Annual Meeting in Chicago on September 12.
“There was a concern that menopausal women are being targeted for treatments that may not have a lot of benefit and might have significant harms,” Elisheva Danan, MD, MPH, a physician at the Minneapolis VA Health Care System and an assistant professor of medicine at the University of Minnesota Medical School in Minneapolis, told this news organization. While she was not surprised to find little evidence of benefit, “we were a little bit surprised that we also didn’t find significant evidence of harms.”
The study was unable to evaluate the potential for financial harms, but Dr. Danan noted that these therapies are often expensive and not typically covered by insurance. The treatments appear to be used primarily in private practice, she said, while “most academic clinicians were not familiar with these and do not use these lasers.”
The American Urological Association had requested the review, Dr. Danan said, “to inform clinical guidelines that they could put out for practitioners about treating genital urinary syndrome from menopause.” Yet the evidence available remains slim. “There’s a lot of outcomes that were not looked at by most of these [trials], or they were looked at in a way that we couldn’t separate out,” she said.
Kamalini Das, MD, a professor of ob.gyn. at the University of Minnesota who was not involved in the research, was surprised by the findings because studies to date have been variable, “but since this looks at multiple studies and they find no benefits, I would take these results as more significant than any of the small studies,” she told this news organization.
Dr. Das said she has patients who ask about using these therapies and have had them done. “So far, I’ve told them the jury is out on whether it will help or not, that there are some studies that say they’re beneficial and some studies that they’re not,” Dr. Das said.
But this new review changes what she will tell patients going forward, she said. “This is a good study because it consolidates lots of little studies, so I think I would use this to say, looking at all the studies together, this treatment is not beneficial.”
GSM occurs due to the body’s reduced production of estrogen and affects anywhere from 27% to 84% of postmenopausal women. It can involve a constellation of symptoms ranging from vaginal discomfort and irritation to painful urination or intercourse. Typical recommended treatments for GSM include systemic hormone therapy, localized hormonal treatments such as vaginal estrogen or dehydroepiandrosterone, nonhormonal creams and moisturizers, and the prescription drug ospemifene.
Most of these have been found effective, according to a recent systematic review Dr. Danan published in the Annals of Internal Medicine that this news organization covered. But recent years have also seen a rapid increase in interest and the availability of energy-based treatments for GSM, such as CO2 laser and radiofrequency interventions, particularly for those who cannot or do not want to use hormonal treatments. The idea behind these newer therapies is that they “heat tissue to cause a denaturation of collagen fibers and induce a wound-healing response,” with the aim of “enhancement of vaginal elasticity, restoration of premenopausal epithelial function, and symptom improvement,” the authors wrote.
Evidence has been scant and uneven for the safety and effectiveness of these treatments, and they have not been evaluated by the US Food and Drug Administration. The agency issued a warning in 2018 with remarks from then Commissioner Scott Gottlieb that the “products have serious risks and don’t have adequate evidence to support their use for these purposes.”
Much of the evidence has focused on CO2 lasers instead of other energy-based treatments, however, and a raft of new studies have been published on these interventions in the past 2 years. Dr. Danan and colleagues, therefore, assessed the most current state of the research with a systematic review of randomized controlled trials (RCTs) and prospective observational studies with control groups published through December 11, 2023.
Included studies needed to evaluate an energy-based treatment for at least 8 weeks in a minimum of 40 postmenopausal women (20 in each group) who had one or more GSM symptoms. The authors also included nonrandomized and uncontrolled studies with a follow-up of a year or more to assess possible adverse events. The studies also needed to assess at least one of eight core outcomes: Dyspareunia; vulvovaginal dryness; vulvovaginal discomfort/irritation; dysuria; change in most bothersome symptom; treatment satisfaction; adverse events; and distress, bother, or interference associated with genitourinary symptoms.
The authors identified 32 studies, including 16 RCTs, one quasi-RCT, and 15 nonrandomized studies. The researchers extracted and analyzed data from the 10 RCTs and one quasi-RCT that were rated as having low to moderate risk for bias.
Most of these studies assessed CO2 lasers alone, while three assessed erbium:yttrium-aluminum-garnet (Er:YAG) laser, and one looked at CO2 lasers vs radiofrequency treatments.
The average age of participants ranged from 56 to 64 years, and most trials were in the United States. Results showed that CO2 lasers led to little or no difference in dysuria, dyspareunia, or quality of life when compared with sham lasers. The CO2 laser therapy also showed little to no difference compared with vaginal estrogen creams for dyspareunia, dryness, discomfort/irritation, dysuria, or quality of life.
Most CO2 laser studies reported on most outcomes, but the Er:YAG studies tended to report only on quality of life and/or one or two other outcomes. The radiofrequency study only assessed dyspareunia and quality of life.
“Treatment effects on other outcomes and effects of Er:YAG laser or radiofrequency on any outcomes are very uncertain,” the authors reported. Few adverse events and no serious adverse events were reported based on 15 studies, including the additional non-RCTs that had follow-up for at least a year.
“There are case reports and other types of studies that have shown some bad outcomes using laser therapies, and we really wanted to be expansive and include anything, especially because this is such a new treatment and all these trials were in the last couple of years,” Dr. Danan said.
The review was limited by inconsistent or nonvalidated outcome reporting in the studies as well as small populations and short follow-up, typically less than 3 months.
The research was funded by the Agency for Healthcare Research and Quality and Patient-Centered Outcomes Research Institute. Dr. Danan and Dr. Das had no disclosures.
A version of this article first appeared on Medscape.com.
CHICAGO — Use of CO2 lasers and similar “energy-based” treatments result in little to no benefit for genitourinary syndrome of menopause (GSM) symptoms, according to research presented at the The Menopause Society 2024 Annual Meeting in Chicago on September 12.
“There was a concern that menopausal women are being targeted for treatments that may not have a lot of benefit and might have significant harms,” Elisheva Danan, MD, MPH, a physician at the Minneapolis VA Health Care System and an assistant professor of medicine at the University of Minnesota Medical School in Minneapolis, told this news organization. While she was not surprised to find little evidence of benefit, “we were a little bit surprised that we also didn’t find significant evidence of harms.”
The study was unable to evaluate the potential for financial harms, but Dr. Danan noted that these therapies are often expensive and not typically covered by insurance. The treatments appear to be used primarily in private practice, she said, while “most academic clinicians were not familiar with these and do not use these lasers.”
The American Urological Association had requested the review, Dr. Danan said, “to inform clinical guidelines that they could put out for practitioners about treating genital urinary syndrome from menopause.” Yet the evidence available remains slim. “There’s a lot of outcomes that were not looked at by most of these [trials], or they were looked at in a way that we couldn’t separate out,” she said.
Kamalini Das, MD, a professor of ob.gyn. at the University of Minnesota who was not involved in the research, was surprised by the findings because studies to date have been variable, “but since this looks at multiple studies and they find no benefits, I would take these results as more significant than any of the small studies,” she told this news organization.
Dr. Das said she has patients who ask about using these therapies and have had them done. “So far, I’ve told them the jury is out on whether it will help or not, that there are some studies that say they’re beneficial and some studies that they’re not,” Dr. Das said.
But this new review changes what she will tell patients going forward, she said. “This is a good study because it consolidates lots of little studies, so I think I would use this to say, looking at all the studies together, this treatment is not beneficial.”
GSM occurs due to the body’s reduced production of estrogen and affects anywhere from 27% to 84% of postmenopausal women. It can involve a constellation of symptoms ranging from vaginal discomfort and irritation to painful urination or intercourse. Typical recommended treatments for GSM include systemic hormone therapy, localized hormonal treatments such as vaginal estrogen or dehydroepiandrosterone, nonhormonal creams and moisturizers, and the prescription drug ospemifene.
Most of these have been found effective, according to a recent systematic review Dr. Danan published in the Annals of Internal Medicine that this news organization covered. But recent years have also seen a rapid increase in interest and the availability of energy-based treatments for GSM, such as CO2 laser and radiofrequency interventions, particularly for those who cannot or do not want to use hormonal treatments. The idea behind these newer therapies is that they “heat tissue to cause a denaturation of collagen fibers and induce a wound-healing response,” with the aim of “enhancement of vaginal elasticity, restoration of premenopausal epithelial function, and symptom improvement,” the authors wrote.
Evidence has been scant and uneven for the safety and effectiveness of these treatments, and they have not been evaluated by the US Food and Drug Administration. The agency issued a warning in 2018 with remarks from then Commissioner Scott Gottlieb that the “products have serious risks and don’t have adequate evidence to support their use for these purposes.”
Much of the evidence has focused on CO2 lasers instead of other energy-based treatments, however, and a raft of new studies have been published on these interventions in the past 2 years. Dr. Danan and colleagues, therefore, assessed the most current state of the research with a systematic review of randomized controlled trials (RCTs) and prospective observational studies with control groups published through December 11, 2023.
Included studies needed to evaluate an energy-based treatment for at least 8 weeks in a minimum of 40 postmenopausal women (20 in each group) who had one or more GSM symptoms. The authors also included nonrandomized and uncontrolled studies with a follow-up of a year or more to assess possible adverse events. The studies also needed to assess at least one of eight core outcomes: Dyspareunia; vulvovaginal dryness; vulvovaginal discomfort/irritation; dysuria; change in most bothersome symptom; treatment satisfaction; adverse events; and distress, bother, or interference associated with genitourinary symptoms.
The authors identified 32 studies, including 16 RCTs, one quasi-RCT, and 15 nonrandomized studies. The researchers extracted and analyzed data from the 10 RCTs and one quasi-RCT that were rated as having low to moderate risk for bias.
Most of these studies assessed CO2 lasers alone, while three assessed erbium:yttrium-aluminum-garnet (Er:YAG) laser, and one looked at CO2 lasers vs radiofrequency treatments.
The average age of participants ranged from 56 to 64 years, and most trials were in the United States. Results showed that CO2 lasers led to little or no difference in dysuria, dyspareunia, or quality of life when compared with sham lasers. The CO2 laser therapy also showed little to no difference compared with vaginal estrogen creams for dyspareunia, dryness, discomfort/irritation, dysuria, or quality of life.
Most CO2 laser studies reported on most outcomes, but the Er:YAG studies tended to report only on quality of life and/or one or two other outcomes. The radiofrequency study only assessed dyspareunia and quality of life.
“Treatment effects on other outcomes and effects of Er:YAG laser or radiofrequency on any outcomes are very uncertain,” the authors reported. Few adverse events and no serious adverse events were reported based on 15 studies, including the additional non-RCTs that had follow-up for at least a year.
“There are case reports and other types of studies that have shown some bad outcomes using laser therapies, and we really wanted to be expansive and include anything, especially because this is such a new treatment and all these trials were in the last couple of years,” Dr. Danan said.
The review was limited by inconsistent or nonvalidated outcome reporting in the studies as well as small populations and short follow-up, typically less than 3 months.
The research was funded by the Agency for Healthcare Research and Quality and Patient-Centered Outcomes Research Institute. Dr. Danan and Dr. Das had no disclosures.
A version of this article first appeared on Medscape.com.
FROM THE MENOPAUSE SOCIETY 2024
Analysis of Colchicine’s Drug-Drug Interactions Finds Little Risk
TOPLINE:
The presence of an operational classification of drug interactions (ORCA) class 3 or 4 drug-drug interactions (DDIs) did not increase the risk for colchicine-related gastrointestinal adverse events or modify the effect of colchicine on death or hospitalization caused by COVID-19 infection in ambulatory patients.
METHODOLOGY:
- This secondary analysis of the COLCORONA trial aimed to evaluate if a potential DDI of colchicine was associated with changes in its pharmacokinetics or modified its clinical safety and efficacy in patients with COVID-19.
- Overall, 4432 ambulatory patients with COVID-19 (median age, 54 years; 54% women) were randomly assigned to receive colchicine 0.5 mg twice daily for 3 days and then 0.5 mg once daily for 27 days (n = 2205) or a placebo (n = 2227).
- All the participants had at least one high-risk criterion such as age ≥ 70 years, diabetes, heart failure, systolic blood pressure ≥ 150 mm Hg, respiratory disease, coronary disease, body temperature ≥ 38.4 °C within the last 48 hours, dyspnea, bicytopenia, pancytopenia, or high neutrophil count with low lymphocyte count.
- The medications that could interact with colchicine were determined and categorized under ORCA classes 1 (contraindicated), 2 (provisionally contraindicated), 3 (conditional use), or 4 (minimal risk).
- The primary outcome was any gastrointestinal adverse event assessed over a 30-day follow-up period.
TAKEAWAY:
- Among all the participants, 1% received medications with an ORCA class 2 interaction, 14% with a class 3 interaction, and 13% with a class 4 interaction; rosuvastatin (12%) and atorvastatin (10%) were the most common interacting medications.
- The odds of any gastrointestinal adverse event were 1.80 times and 1.68 times higher in the colchicine arm than in the placebo arm among those without and with a DDI, respectively, with the effect of colchicine being consistent regardless of the presence of drug interactions (P = .69 for interaction).
- Similarly, DDIs did not influence the effect of colchicine on combined risk for COVID-19 hospitalization or mortality (P = .80 for interaction).
IN PRACTICE:
“Once potential DDIs have been identified through screening, they must be tested,” Hemalkumar B. Mehta, PhD, and G. Caleb Alexander, MD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, wrote in an invited commentary published online in JAMA Network Open. “Theoretical DDIs may not translate into real-world harms,” they added.
SOURCE:
The study was led by Lama S. Alfehaid, PharmD, of Brigham and Women’s Hospital, Boston. It was published online in JAMA Network Open.
LIMITATIONS:
This study focused on the medications used by participants at baseline, which may not have captured all potential DDIs. The findings did not provide information on rare adverse events, such as rhabdomyolysis, which usually occur months after initiating drug therapy. Furthermore, all the study participants had confirmed SARS-CoV-2 infection, which may have increased their susceptibility to adverse reactions associated with the use of colchicine.
DISCLOSURES:
Some authors were supported by grants from the National Institutes of Health/National Heart, Lung, and Blood Institute, American Heart Association, and other sources. The authors also declared serving on advisory boards or on the board of directors; receiving personal fees, grants, research support, or speaking fees; or having other ties with many pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The presence of an operational classification of drug interactions (ORCA) class 3 or 4 drug-drug interactions (DDIs) did not increase the risk for colchicine-related gastrointestinal adverse events or modify the effect of colchicine on death or hospitalization caused by COVID-19 infection in ambulatory patients.
METHODOLOGY:
- This secondary analysis of the COLCORONA trial aimed to evaluate if a potential DDI of colchicine was associated with changes in its pharmacokinetics or modified its clinical safety and efficacy in patients with COVID-19.
- Overall, 4432 ambulatory patients with COVID-19 (median age, 54 years; 54% women) were randomly assigned to receive colchicine 0.5 mg twice daily for 3 days and then 0.5 mg once daily for 27 days (n = 2205) or a placebo (n = 2227).
- All the participants had at least one high-risk criterion such as age ≥ 70 years, diabetes, heart failure, systolic blood pressure ≥ 150 mm Hg, respiratory disease, coronary disease, body temperature ≥ 38.4 °C within the last 48 hours, dyspnea, bicytopenia, pancytopenia, or high neutrophil count with low lymphocyte count.
- The medications that could interact with colchicine were determined and categorized under ORCA classes 1 (contraindicated), 2 (provisionally contraindicated), 3 (conditional use), or 4 (minimal risk).
- The primary outcome was any gastrointestinal adverse event assessed over a 30-day follow-up period.
TAKEAWAY:
- Among all the participants, 1% received medications with an ORCA class 2 interaction, 14% with a class 3 interaction, and 13% with a class 4 interaction; rosuvastatin (12%) and atorvastatin (10%) were the most common interacting medications.
- The odds of any gastrointestinal adverse event were 1.80 times and 1.68 times higher in the colchicine arm than in the placebo arm among those without and with a DDI, respectively, with the effect of colchicine being consistent regardless of the presence of drug interactions (P = .69 for interaction).
- Similarly, DDIs did not influence the effect of colchicine on combined risk for COVID-19 hospitalization or mortality (P = .80 for interaction).
IN PRACTICE:
“Once potential DDIs have been identified through screening, they must be tested,” Hemalkumar B. Mehta, PhD, and G. Caleb Alexander, MD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, wrote in an invited commentary published online in JAMA Network Open. “Theoretical DDIs may not translate into real-world harms,” they added.
SOURCE:
The study was led by Lama S. Alfehaid, PharmD, of Brigham and Women’s Hospital, Boston. It was published online in JAMA Network Open.
LIMITATIONS:
This study focused on the medications used by participants at baseline, which may not have captured all potential DDIs. The findings did not provide information on rare adverse events, such as rhabdomyolysis, which usually occur months after initiating drug therapy. Furthermore, all the study participants had confirmed SARS-CoV-2 infection, which may have increased their susceptibility to adverse reactions associated with the use of colchicine.
DISCLOSURES:
Some authors were supported by grants from the National Institutes of Health/National Heart, Lung, and Blood Institute, American Heart Association, and other sources. The authors also declared serving on advisory boards or on the board of directors; receiving personal fees, grants, research support, or speaking fees; or having other ties with many pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The presence of an operational classification of drug interactions (ORCA) class 3 or 4 drug-drug interactions (DDIs) did not increase the risk for colchicine-related gastrointestinal adverse events or modify the effect of colchicine on death or hospitalization caused by COVID-19 infection in ambulatory patients.
METHODOLOGY:
- This secondary analysis of the COLCORONA trial aimed to evaluate if a potential DDI of colchicine was associated with changes in its pharmacokinetics or modified its clinical safety and efficacy in patients with COVID-19.
- Overall, 4432 ambulatory patients with COVID-19 (median age, 54 years; 54% women) were randomly assigned to receive colchicine 0.5 mg twice daily for 3 days and then 0.5 mg once daily for 27 days (n = 2205) or a placebo (n = 2227).
- All the participants had at least one high-risk criterion such as age ≥ 70 years, diabetes, heart failure, systolic blood pressure ≥ 150 mm Hg, respiratory disease, coronary disease, body temperature ≥ 38.4 °C within the last 48 hours, dyspnea, bicytopenia, pancytopenia, or high neutrophil count with low lymphocyte count.
- The medications that could interact with colchicine were determined and categorized under ORCA classes 1 (contraindicated), 2 (provisionally contraindicated), 3 (conditional use), or 4 (minimal risk).
- The primary outcome was any gastrointestinal adverse event assessed over a 30-day follow-up period.
TAKEAWAY:
- Among all the participants, 1% received medications with an ORCA class 2 interaction, 14% with a class 3 interaction, and 13% with a class 4 interaction; rosuvastatin (12%) and atorvastatin (10%) were the most common interacting medications.
- The odds of any gastrointestinal adverse event were 1.80 times and 1.68 times higher in the colchicine arm than in the placebo arm among those without and with a DDI, respectively, with the effect of colchicine being consistent regardless of the presence of drug interactions (P = .69 for interaction).
- Similarly, DDIs did not influence the effect of colchicine on combined risk for COVID-19 hospitalization or mortality (P = .80 for interaction).
IN PRACTICE:
“Once potential DDIs have been identified through screening, they must be tested,” Hemalkumar B. Mehta, PhD, and G. Caleb Alexander, MD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, wrote in an invited commentary published online in JAMA Network Open. “Theoretical DDIs may not translate into real-world harms,” they added.
SOURCE:
The study was led by Lama S. Alfehaid, PharmD, of Brigham and Women’s Hospital, Boston. It was published online in JAMA Network Open.
LIMITATIONS:
This study focused on the medications used by participants at baseline, which may not have captured all potential DDIs. The findings did not provide information on rare adverse events, such as rhabdomyolysis, which usually occur months after initiating drug therapy. Furthermore, all the study participants had confirmed SARS-CoV-2 infection, which may have increased their susceptibility to adverse reactions associated with the use of colchicine.
DISCLOSURES:
Some authors were supported by grants from the National Institutes of Health/National Heart, Lung, and Blood Institute, American Heart Association, and other sources. The authors also declared serving on advisory boards or on the board of directors; receiving personal fees, grants, research support, or speaking fees; or having other ties with many pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Neurofibromatosis: What Affects Quality of Life Most?
TOPLINE:
Mobile images may be reliable for assessing cutaneous neurofibroma (cNF) features in patients with neurofibromatosis type 1 (NF1), according to a crowd-sourced .
METHODOLOGY:
- To learn more about the association of cNFs with QoL, pain, and itch in patients with this rare disease, researchers enrolled 1016 individuals aged 40 years and older with NF1 who had at least one cNF, from May 2021 to December 2023, after reaching out to patient-led or NF1 advocacy organizations in 13 countries, including the United States.
- Participants provided demographic data, detailed photographs, and saliva samples for genetic sequencing, with 583 participants (mean age, 51.7 years; 65.9% women) submitting high-quality photographs from seven body regions at the time of the study analysis.
- A subset of 50 participants also underwent whole-body imaging.
- Four researchers independently rated the photographs for various cNF features, including general severity, number, size, facial severity, and subtypes.
TAKEAWAY:
- Based on evaluations by NF1 specialists, the agreement between mobile and whole-body images was “substantial” (74%-88% agreement) for the number of cNFs, general severity, and facial severity. Agreement between self-reported numbers of cNFs and investigator-rated numbers based on photographs was “minimal to fair.”
- Female sex, the number of cNFs, severity of cNFs on the face, and globular cNFs were associated with worse QoL (based on Skindex scores); severity of cNFs on the face had the strongest impact on overall QoL (P < .001).
- An increasing number of cNFs and worsening facial severity were strongly correlated with higher emotion subdomain scores.
- A higher number of cNFs, more severe cNFs on the face, and larger cNFs were all slightly associated with increased itch and pain (P < .01).
IN PRACTICE:
“To develop effective therapeutics, meaningful clinical outcomes that are tied with improvement in QoL for persons with NF1 must be clearly defined,” the authors wrote. The results of this study, they added, “suggested the benefit of this crowd-sourced resource by identifying the features of cNFs with the greatest association with QoL and symptoms of pain and itch in persons with NF1, highlighting new intervention strategies and features to target to most improve QoL in NF1.”
SOURCE:
The study was led by Michelle Jade Lin, BS, Stanford University School of Medicine, Redwood City, California, and was published online in JAMA Dermatology.
LIMITATIONS:
The study included only a small number of individuals from racial and ethnic minority groups and did not capture ethnicity information, which could have provided further insights into disease impact across different demographics.
DISCLOSURES:
This study was supported by Johns Hopkins University, Baltimore, and the Bloomberg Family Foundation. Ms. Lin reported support from the Stanford Medical Scholars Research Program. Three authors reported personal fees or grants outside this work. Other authors reported no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Mobile images may be reliable for assessing cutaneous neurofibroma (cNF) features in patients with neurofibromatosis type 1 (NF1), according to a crowd-sourced .
METHODOLOGY:
- To learn more about the association of cNFs with QoL, pain, and itch in patients with this rare disease, researchers enrolled 1016 individuals aged 40 years and older with NF1 who had at least one cNF, from May 2021 to December 2023, after reaching out to patient-led or NF1 advocacy organizations in 13 countries, including the United States.
- Participants provided demographic data, detailed photographs, and saliva samples for genetic sequencing, with 583 participants (mean age, 51.7 years; 65.9% women) submitting high-quality photographs from seven body regions at the time of the study analysis.
- A subset of 50 participants also underwent whole-body imaging.
- Four researchers independently rated the photographs for various cNF features, including general severity, number, size, facial severity, and subtypes.
TAKEAWAY:
- Based on evaluations by NF1 specialists, the agreement between mobile and whole-body images was “substantial” (74%-88% agreement) for the number of cNFs, general severity, and facial severity. Agreement between self-reported numbers of cNFs and investigator-rated numbers based on photographs was “minimal to fair.”
- Female sex, the number of cNFs, severity of cNFs on the face, and globular cNFs were associated with worse QoL (based on Skindex scores); severity of cNFs on the face had the strongest impact on overall QoL (P < .001).
- An increasing number of cNFs and worsening facial severity were strongly correlated with higher emotion subdomain scores.
- A higher number of cNFs, more severe cNFs on the face, and larger cNFs were all slightly associated with increased itch and pain (P < .01).
IN PRACTICE:
“To develop effective therapeutics, meaningful clinical outcomes that are tied with improvement in QoL for persons with NF1 must be clearly defined,” the authors wrote. The results of this study, they added, “suggested the benefit of this crowd-sourced resource by identifying the features of cNFs with the greatest association with QoL and symptoms of pain and itch in persons with NF1, highlighting new intervention strategies and features to target to most improve QoL in NF1.”
SOURCE:
The study was led by Michelle Jade Lin, BS, Stanford University School of Medicine, Redwood City, California, and was published online in JAMA Dermatology.
LIMITATIONS:
The study included only a small number of individuals from racial and ethnic minority groups and did not capture ethnicity information, which could have provided further insights into disease impact across different demographics.
DISCLOSURES:
This study was supported by Johns Hopkins University, Baltimore, and the Bloomberg Family Foundation. Ms. Lin reported support from the Stanford Medical Scholars Research Program. Three authors reported personal fees or grants outside this work. Other authors reported no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Mobile images may be reliable for assessing cutaneous neurofibroma (cNF) features in patients with neurofibromatosis type 1 (NF1), according to a crowd-sourced .
METHODOLOGY:
- To learn more about the association of cNFs with QoL, pain, and itch in patients with this rare disease, researchers enrolled 1016 individuals aged 40 years and older with NF1 who had at least one cNF, from May 2021 to December 2023, after reaching out to patient-led or NF1 advocacy organizations in 13 countries, including the United States.
- Participants provided demographic data, detailed photographs, and saliva samples for genetic sequencing, with 583 participants (mean age, 51.7 years; 65.9% women) submitting high-quality photographs from seven body regions at the time of the study analysis.
- A subset of 50 participants also underwent whole-body imaging.
- Four researchers independently rated the photographs for various cNF features, including general severity, number, size, facial severity, and subtypes.
TAKEAWAY:
- Based on evaluations by NF1 specialists, the agreement between mobile and whole-body images was “substantial” (74%-88% agreement) for the number of cNFs, general severity, and facial severity. Agreement between self-reported numbers of cNFs and investigator-rated numbers based on photographs was “minimal to fair.”
- Female sex, the number of cNFs, severity of cNFs on the face, and globular cNFs were associated with worse QoL (based on Skindex scores); severity of cNFs on the face had the strongest impact on overall QoL (P < .001).
- An increasing number of cNFs and worsening facial severity were strongly correlated with higher emotion subdomain scores.
- A higher number of cNFs, more severe cNFs on the face, and larger cNFs were all slightly associated with increased itch and pain (P < .01).
IN PRACTICE:
“To develop effective therapeutics, meaningful clinical outcomes that are tied with improvement in QoL for persons with NF1 must be clearly defined,” the authors wrote. The results of this study, they added, “suggested the benefit of this crowd-sourced resource by identifying the features of cNFs with the greatest association with QoL and symptoms of pain and itch in persons with NF1, highlighting new intervention strategies and features to target to most improve QoL in NF1.”
SOURCE:
The study was led by Michelle Jade Lin, BS, Stanford University School of Medicine, Redwood City, California, and was published online in JAMA Dermatology.
LIMITATIONS:
The study included only a small number of individuals from racial and ethnic minority groups and did not capture ethnicity information, which could have provided further insights into disease impact across different demographics.
DISCLOSURES:
This study was supported by Johns Hopkins University, Baltimore, and the Bloomberg Family Foundation. Ms. Lin reported support from the Stanford Medical Scholars Research Program. Three authors reported personal fees or grants outside this work. Other authors reported no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa Risk Reduced After Patients Quit Smoking
TOPLINE:
, with this reduction becoming evident 3-4 years after cessation, in a cohort study from Korea.
METHODOLOGY:
- Researchers conducted a population-based cohort study using the Korean National Health Insurance Service database.
- A total of 6,230,189 participants in South Korea who underwent two consecutive biennial health examinations from 2004 to 2005 and 2006 to 2007 were included.
- Participants were categorized into six groups on the basis of their smoking status at both checkups: Sustained smokers, relapsed smokers, new smokers, smoking quitters, sustained ex-smokers, and never smokers.
- The primary outcome was the development of HS.
TAKEAWAY:
- A total of 3761 HS cases were detected during the 84,457,025 person-years of observation.
- Smoking quitters (adjusted hazard ratio [AHR], 0.68; 95% CI, 0.56-0.83), sustained ex-smokers (AHR, 0.67; 95% CI, 0.57-0.77), and never smokers (AHR, 0.57; 95% CI, 0.52-0.63) exhibited a reduced risk of developing HS compared with sustained smokers.
- The risk of developing HS varied over time, with smoking quitters showing no significant risk reduction compared with sustained smokers in the first 3 years. After 3 years, a statistically significant decrease in HS risk was observed among quitters, which persisted over time.
- At 3-6 years, the risk reduction in sustained quitters was comparable with that of never smokers (AHR, 0.58 and 0.63, respectively).
IN PRACTICE:
“Smoking cessation and maintaining a smoke-free lifestyle may be important preventive measures against the development of HS,” the authors concluded. In an accompanying editorial, Alexandra Charrow, MD, and Leandra A. Barnes, MD, of the departments of dermatology at Brigham and Women’s Hospital, Boston, and Stanford University, Palo Alto, California, respectively, wrote that while the study “importantly contributes to the understanding of the association of smoking tobacco and HS onset, prospective cohort studies in large, diverse cohorts of patients with HS may help dermatologists better understand the causal relationship between smoking and the onset or exacerbation of HS.” For now, they added, “dermatologists must continue to use comprehensive HS treatment strategies, including lifestyle modifications that promote overall health like smoking cessation, to improve the lives of those enduring HS.”
SOURCE:
The study was led by Seong Rae Kim, MD, Department of Dermatology, Seoul National University College of Medicine, Seoul, Republic of Korea, and was published online, along with the editorial, on August 21 in JAMA Dermatology.
LIMITATIONS:
The study limitations include the potential for unexamined confounding factors like hereditary background, reliance on self-reported smoking status, and the exclusion of electronic cigarette use and nicotine replacement therapy. The predominantly male smoker population may limit generalizability, and delayed diagnosis of HS may not reflect the actual time of onset.
DISCLOSURES:
The study funding source was not disclosed. One study author reported various financial ties with pharmaceutical companies outside this work; other authors had no disclosures. Dr. Charrow’s disclosures included receiving personal fees from several pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, with this reduction becoming evident 3-4 years after cessation, in a cohort study from Korea.
METHODOLOGY:
- Researchers conducted a population-based cohort study using the Korean National Health Insurance Service database.
- A total of 6,230,189 participants in South Korea who underwent two consecutive biennial health examinations from 2004 to 2005 and 2006 to 2007 were included.
- Participants were categorized into six groups on the basis of their smoking status at both checkups: Sustained smokers, relapsed smokers, new smokers, smoking quitters, sustained ex-smokers, and never smokers.
- The primary outcome was the development of HS.
TAKEAWAY:
- A total of 3761 HS cases were detected during the 84,457,025 person-years of observation.
- Smoking quitters (adjusted hazard ratio [AHR], 0.68; 95% CI, 0.56-0.83), sustained ex-smokers (AHR, 0.67; 95% CI, 0.57-0.77), and never smokers (AHR, 0.57; 95% CI, 0.52-0.63) exhibited a reduced risk of developing HS compared with sustained smokers.
- The risk of developing HS varied over time, with smoking quitters showing no significant risk reduction compared with sustained smokers in the first 3 years. After 3 years, a statistically significant decrease in HS risk was observed among quitters, which persisted over time.
- At 3-6 years, the risk reduction in sustained quitters was comparable with that of never smokers (AHR, 0.58 and 0.63, respectively).
IN PRACTICE:
“Smoking cessation and maintaining a smoke-free lifestyle may be important preventive measures against the development of HS,” the authors concluded. In an accompanying editorial, Alexandra Charrow, MD, and Leandra A. Barnes, MD, of the departments of dermatology at Brigham and Women’s Hospital, Boston, and Stanford University, Palo Alto, California, respectively, wrote that while the study “importantly contributes to the understanding of the association of smoking tobacco and HS onset, prospective cohort studies in large, diverse cohorts of patients with HS may help dermatologists better understand the causal relationship between smoking and the onset or exacerbation of HS.” For now, they added, “dermatologists must continue to use comprehensive HS treatment strategies, including lifestyle modifications that promote overall health like smoking cessation, to improve the lives of those enduring HS.”
SOURCE:
The study was led by Seong Rae Kim, MD, Department of Dermatology, Seoul National University College of Medicine, Seoul, Republic of Korea, and was published online, along with the editorial, on August 21 in JAMA Dermatology.
LIMITATIONS:
The study limitations include the potential for unexamined confounding factors like hereditary background, reliance on self-reported smoking status, and the exclusion of electronic cigarette use and nicotine replacement therapy. The predominantly male smoker population may limit generalizability, and delayed diagnosis of HS may not reflect the actual time of onset.
DISCLOSURES:
The study funding source was not disclosed. One study author reported various financial ties with pharmaceutical companies outside this work; other authors had no disclosures. Dr. Charrow’s disclosures included receiving personal fees from several pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
, with this reduction becoming evident 3-4 years after cessation, in a cohort study from Korea.
METHODOLOGY:
- Researchers conducted a population-based cohort study using the Korean National Health Insurance Service database.
- A total of 6,230,189 participants in South Korea who underwent two consecutive biennial health examinations from 2004 to 2005 and 2006 to 2007 were included.
- Participants were categorized into six groups on the basis of their smoking status at both checkups: Sustained smokers, relapsed smokers, new smokers, smoking quitters, sustained ex-smokers, and never smokers.
- The primary outcome was the development of HS.
TAKEAWAY:
- A total of 3761 HS cases were detected during the 84,457,025 person-years of observation.
- Smoking quitters (adjusted hazard ratio [AHR], 0.68; 95% CI, 0.56-0.83), sustained ex-smokers (AHR, 0.67; 95% CI, 0.57-0.77), and never smokers (AHR, 0.57; 95% CI, 0.52-0.63) exhibited a reduced risk of developing HS compared with sustained smokers.
- The risk of developing HS varied over time, with smoking quitters showing no significant risk reduction compared with sustained smokers in the first 3 years. After 3 years, a statistically significant decrease in HS risk was observed among quitters, which persisted over time.
- At 3-6 years, the risk reduction in sustained quitters was comparable with that of never smokers (AHR, 0.58 and 0.63, respectively).
IN PRACTICE:
“Smoking cessation and maintaining a smoke-free lifestyle may be important preventive measures against the development of HS,” the authors concluded. In an accompanying editorial, Alexandra Charrow, MD, and Leandra A. Barnes, MD, of the departments of dermatology at Brigham and Women’s Hospital, Boston, and Stanford University, Palo Alto, California, respectively, wrote that while the study “importantly contributes to the understanding of the association of smoking tobacco and HS onset, prospective cohort studies in large, diverse cohorts of patients with HS may help dermatologists better understand the causal relationship between smoking and the onset or exacerbation of HS.” For now, they added, “dermatologists must continue to use comprehensive HS treatment strategies, including lifestyle modifications that promote overall health like smoking cessation, to improve the lives of those enduring HS.”
SOURCE:
The study was led by Seong Rae Kim, MD, Department of Dermatology, Seoul National University College of Medicine, Seoul, Republic of Korea, and was published online, along with the editorial, on August 21 in JAMA Dermatology.
LIMITATIONS:
The study limitations include the potential for unexamined confounding factors like hereditary background, reliance on self-reported smoking status, and the exclusion of electronic cigarette use and nicotine replacement therapy. The predominantly male smoker population may limit generalizability, and delayed diagnosis of HS may not reflect the actual time of onset.
DISCLOSURES:
The study funding source was not disclosed. One study author reported various financial ties with pharmaceutical companies outside this work; other authors had no disclosures. Dr. Charrow’s disclosures included receiving personal fees from several pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.