User login
Not COVID Toes: Pool Palms and Feet in Pediatric Patients
Practice Gap
Frictional, symmetric, asymptomatic, erythematous macules of the hands and feet can be mistaken for perniolike lesions associated with COVID-19, commonly known as COVID toes. However, in a low-risk setting without other associated symptoms or concerning findings on examination, consider and inquire about frequent use of a swimming pool. This activity can lead to localized pressure- and friction-induced erythema on palmar and plantar surfaces, called “pool palms and feet,” expanding on the already-named lesion “pool palms”—an entity that is distinct from COVID toes.
Technique for Diagnosis
We evaluated 4 patients in the outpatient setting who presented with localized, patterned, erythematous lesions of the hands or feet, or both, during the COVID-19 pandemic. The parents of our patients were concerned that the rash represented “COVID fingers and toes,” which are perniolike lesions seen in patients with suspected or confirmed current or prior COVID-19.1
Pernio, also known as chilblains, is a superficial inflammatory vascular response, usually in the setting of exposure to cold.2 This phenomenon usually appears as erythematous or violaceous macules and papules on acral skin, particularly on the dorsum and sides of the fingers and toes, with edema, vesiculation, and ulceration in more severe cases. Initially, it is pruritic and painful at times.
With COVID toes, there often is a delayed presentation of perniolike lesions after the onset of other COVID-19 symptoms, such as fever, cough, headache, and sore throat.2,3 It has been described more often in younger patients and those with milder disease. However, because our patients had no known exposure to SARS-CoV-2 or other associated symptoms, our suspicion was low.
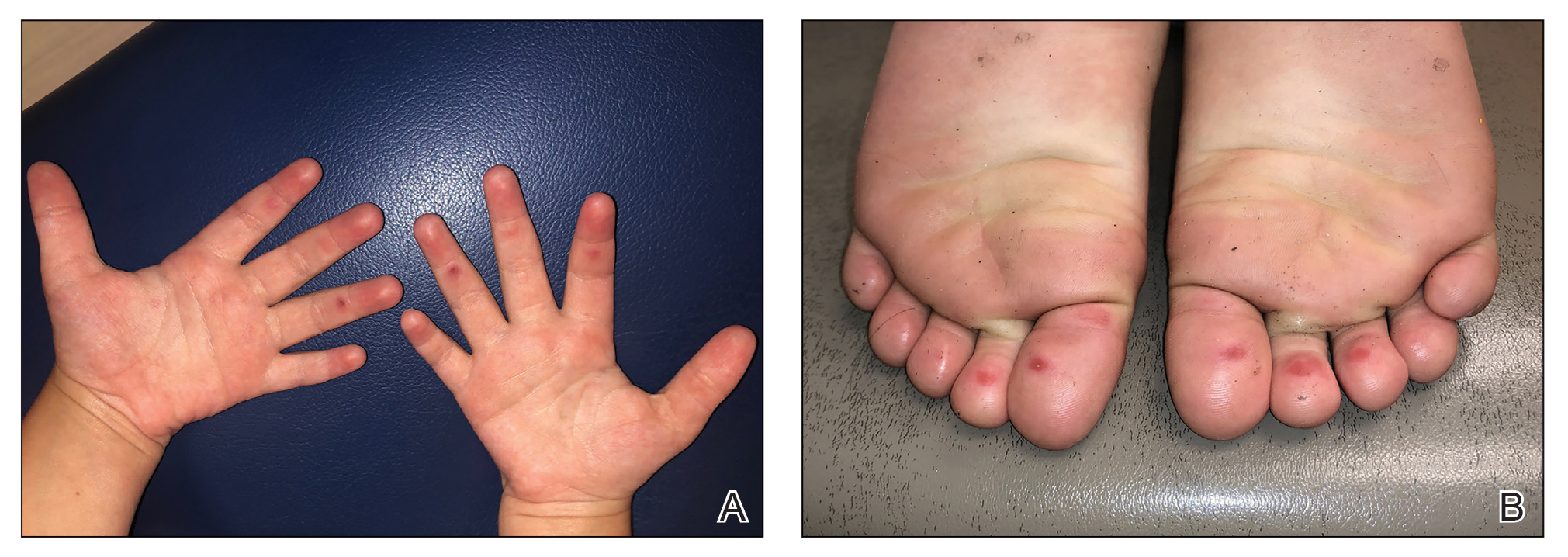
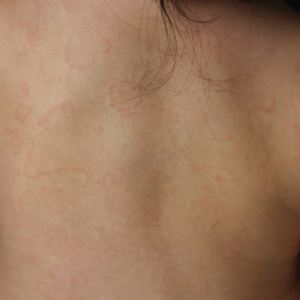
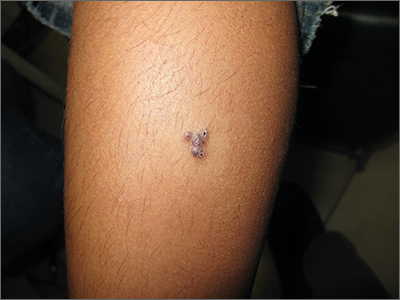
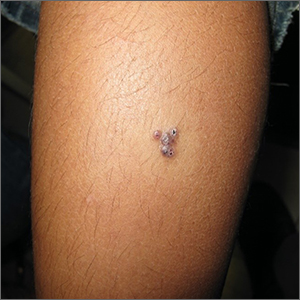
The 4 patients we evaluated—aged 4 to 12 years and in their usual good health—had blanchable erythema of the palmar fingers, palmar eminences of both hands, and plantar surfaces of both feet (Figure). There was no swelling or tenderness, and the lesions had no violaceous coloration, vesiculation, or ulceration. There was no associated pruritus or pain. One patient reported rough texture and mild peeling of the hands.
Upon further inquiry, the patients reported a history of extended time spent in home swimming pools, including holding on to the edge of the pool, due to limitation of activities because of COVID restrictions. One parent noted that the pool that caused the rash had a rough nonslip surface, whereas other pools that the children used, which had a smoother surface, caused no problems.
The morphology of symmetric blanching erythema in areas of pressure and friction, in the absence of a notable medical history, signs, or symptoms, was consistent with a diagnosis of pool palms, which has been described in the medical literature.4-9 Pool palms can affect the palms and soles, which are subject to substantial friction, especially when a person is getting in and out of the pool. There is a general consensus that pool palms is a frictional dermatitis affecting children because the greater fragility of their skin is exacerbated by immersion in water.4-9
Pool palms and feet is benign. Only supportive care, with cessation of swimming and application of emollients, is necessary.
Apart from COVID-19, other conditions to consider in a patient with erythematous lesions of the palms and soles include eczematous dermatitis; neutrophilic eccrine hidradenitis; and, if lesions are vesicular, hand-foot-and-mouth disease. Juvenile plantar dermatosis, which is thought to be due to moisture with occlusion in shoes, also might be considered but is distinguished by more scales and fissures that can be painful.
Location of the lesions is a critical variable. The patients we evaluated had lesions primarily on palmar and plantar surfaces where contact with pool surfaces was greatest, such as at bony prominences, which supported a diagnosis of frictional dermatitis, such as pool palms and feet. A thorough history and physical examination are helpful in determining the diagnosis.
Practical Implications
It is important to consider and recognize this localized pressure phenomenon of pool palms and feet, thus obviating an unnecessary workup or therapeutic interventions. Specifically, a finding of erythematous asymptomatic macules, with or without scaling, on bony prominences of the palms and soles is more consistent with pool palms and feet.
Pernio and COVID toes both present as erythematous to violaceous papules and macules, with edema, vesiculation, and ulceration in severe cases, often on the dorsum and sides of fingers and toes; typically the conditions are pruritic and painful at times.
Explaining the diagnosis of pool palms and feet and sharing one’s experience with similar cases might help alleviate parental fear and anxiety during the COVID-19 pandemic.
- de Masson A, Bouaziz J-D, Sulimovic L, et al; SNDV (French National Union of Dermatologists–Venereologists). Chilblains is a common cutaneous finding during the COVID-19 pandemic: a retrospective nationwide study from France. J Am Acad Dermatol. 2020;83:667-670. doi:10.1016/j.jaad.2020.04.161
- Freeman EE, McMahon DE, Lipoff JB, et al; American Academy of Dermatology Ad Hoc Task Force on COVID-19. Pernio-like skin lesions associated with COVID-19: a case series of 318 patients from 8 countries. J Am Acad Dermatol. 2020;83:486-492. doi:10.1016/j.jaad.2020.05.109
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129. doi:10.1016/j.jaad.2020.06.1016
- Blauvelt A, Duarte AM, Schachner LA. Pool palms. J Am Acad Dermatol. 1992;27:111. doi:10.1016/s0190-9622(08)80819-5
- Wong L-C, Rogers M. Pool palms. Pediatr Dermatol. 2007;24:95. doi:10.1111/j.1525-1470.2007.00347.x
- Novoa A, Klear S. Pool palms. Arch Dis Child. 2016;101:41. doi:10.1136/archdischild-2015-309633
- Morgado-Carasco D, Feola H, Vargas-Mora P. Pool palms. Dermatol Pract Concept. 2020;10:e2020009. doi:10.5826/dpc.1001a09
- Cutrone M, Valerio E, Grimalt R. Pool palms: a case report. Dermatol Case Rep. 2019;4:1000154.
- Martína JM, Ricart JM. Erythematous–violaceous lesions on the palms. Actas Dermosifiliogr. 2009;100:507-508.
Practice Gap
Frictional, symmetric, asymptomatic, erythematous macules of the hands and feet can be mistaken for perniolike lesions associated with COVID-19, commonly known as COVID toes. However, in a low-risk setting without other associated symptoms or concerning findings on examination, consider and inquire about frequent use of a swimming pool. This activity can lead to localized pressure- and friction-induced erythema on palmar and plantar surfaces, called “pool palms and feet,” expanding on the already-named lesion “pool palms”—an entity that is distinct from COVID toes.
Technique for Diagnosis
We evaluated 4 patients in the outpatient setting who presented with localized, patterned, erythematous lesions of the hands or feet, or both, during the COVID-19 pandemic. The parents of our patients were concerned that the rash represented “COVID fingers and toes,” which are perniolike lesions seen in patients with suspected or confirmed current or prior COVID-19.1
Pernio, also known as chilblains, is a superficial inflammatory vascular response, usually in the setting of exposure to cold.2 This phenomenon usually appears as erythematous or violaceous macules and papules on acral skin, particularly on the dorsum and sides of the fingers and toes, with edema, vesiculation, and ulceration in more severe cases. Initially, it is pruritic and painful at times.
With COVID toes, there often is a delayed presentation of perniolike lesions after the onset of other COVID-19 symptoms, such as fever, cough, headache, and sore throat.2,3 It has been described more often in younger patients and those with milder disease. However, because our patients had no known exposure to SARS-CoV-2 or other associated symptoms, our suspicion was low.
The 4 patients we evaluated—aged 4 to 12 years and in their usual good health—had blanchable erythema of the palmar fingers, palmar eminences of both hands, and plantar surfaces of both feet (Figure). There was no swelling or tenderness, and the lesions had no violaceous coloration, vesiculation, or ulceration. There was no associated pruritus or pain. One patient reported rough texture and mild peeling of the hands.
Upon further inquiry, the patients reported a history of extended time spent in home swimming pools, including holding on to the edge of the pool, due to limitation of activities because of COVID restrictions. One parent noted that the pool that caused the rash had a rough nonslip surface, whereas other pools that the children used, which had a smoother surface, caused no problems.
The morphology of symmetric blanching erythema in areas of pressure and friction, in the absence of a notable medical history, signs, or symptoms, was consistent with a diagnosis of pool palms, which has been described in the medical literature.4-9 Pool palms can affect the palms and soles, which are subject to substantial friction, especially when a person is getting in and out of the pool. There is a general consensus that pool palms is a frictional dermatitis affecting children because the greater fragility of their skin is exacerbated by immersion in water.4-9
Pool palms and feet is benign. Only supportive care, with cessation of swimming and application of emollients, is necessary.
Apart from COVID-19, other conditions to consider in a patient with erythematous lesions of the palms and soles include eczematous dermatitis; neutrophilic eccrine hidradenitis; and, if lesions are vesicular, hand-foot-and-mouth disease. Juvenile plantar dermatosis, which is thought to be due to moisture with occlusion in shoes, also might be considered but is distinguished by more scales and fissures that can be painful.
Location of the lesions is a critical variable. The patients we evaluated had lesions primarily on palmar and plantar surfaces where contact with pool surfaces was greatest, such as at bony prominences, which supported a diagnosis of frictional dermatitis, such as pool palms and feet. A thorough history and physical examination are helpful in determining the diagnosis.
Practical Implications
It is important to consider and recognize this localized pressure phenomenon of pool palms and feet, thus obviating an unnecessary workup or therapeutic interventions. Specifically, a finding of erythematous asymptomatic macules, with or without scaling, on bony prominences of the palms and soles is more consistent with pool palms and feet.
Pernio and COVID toes both present as erythematous to violaceous papules and macules, with edema, vesiculation, and ulceration in severe cases, often on the dorsum and sides of fingers and toes; typically the conditions are pruritic and painful at times.
Explaining the diagnosis of pool palms and feet and sharing one’s experience with similar cases might help alleviate parental fear and anxiety during the COVID-19 pandemic.
Practice Gap
Frictional, symmetric, asymptomatic, erythematous macules of the hands and feet can be mistaken for perniolike lesions associated with COVID-19, commonly known as COVID toes. However, in a low-risk setting without other associated symptoms or concerning findings on examination, consider and inquire about frequent use of a swimming pool. This activity can lead to localized pressure- and friction-induced erythema on palmar and plantar surfaces, called “pool palms and feet,” expanding on the already-named lesion “pool palms”—an entity that is distinct from COVID toes.
Technique for Diagnosis
We evaluated 4 patients in the outpatient setting who presented with localized, patterned, erythematous lesions of the hands or feet, or both, during the COVID-19 pandemic. The parents of our patients were concerned that the rash represented “COVID fingers and toes,” which are perniolike lesions seen in patients with suspected or confirmed current or prior COVID-19.1
Pernio, also known as chilblains, is a superficial inflammatory vascular response, usually in the setting of exposure to cold.2 This phenomenon usually appears as erythematous or violaceous macules and papules on acral skin, particularly on the dorsum and sides of the fingers and toes, with edema, vesiculation, and ulceration in more severe cases. Initially, it is pruritic and painful at times.
With COVID toes, there often is a delayed presentation of perniolike lesions after the onset of other COVID-19 symptoms, such as fever, cough, headache, and sore throat.2,3 It has been described more often in younger patients and those with milder disease. However, because our patients had no known exposure to SARS-CoV-2 or other associated symptoms, our suspicion was low.
The 4 patients we evaluated—aged 4 to 12 years and in their usual good health—had blanchable erythema of the palmar fingers, palmar eminences of both hands, and plantar surfaces of both feet (Figure). There was no swelling or tenderness, and the lesions had no violaceous coloration, vesiculation, or ulceration. There was no associated pruritus or pain. One patient reported rough texture and mild peeling of the hands.
Upon further inquiry, the patients reported a history of extended time spent in home swimming pools, including holding on to the edge of the pool, due to limitation of activities because of COVID restrictions. One parent noted that the pool that caused the rash had a rough nonslip surface, whereas other pools that the children used, which had a smoother surface, caused no problems.
The morphology of symmetric blanching erythema in areas of pressure and friction, in the absence of a notable medical history, signs, or symptoms, was consistent with a diagnosis of pool palms, which has been described in the medical literature.4-9 Pool palms can affect the palms and soles, which are subject to substantial friction, especially when a person is getting in and out of the pool. There is a general consensus that pool palms is a frictional dermatitis affecting children because the greater fragility of their skin is exacerbated by immersion in water.4-9
Pool palms and feet is benign. Only supportive care, with cessation of swimming and application of emollients, is necessary.
Apart from COVID-19, other conditions to consider in a patient with erythematous lesions of the palms and soles include eczematous dermatitis; neutrophilic eccrine hidradenitis; and, if lesions are vesicular, hand-foot-and-mouth disease. Juvenile plantar dermatosis, which is thought to be due to moisture with occlusion in shoes, also might be considered but is distinguished by more scales and fissures that can be painful.
Location of the lesions is a critical variable. The patients we evaluated had lesions primarily on palmar and plantar surfaces where contact with pool surfaces was greatest, such as at bony prominences, which supported a diagnosis of frictional dermatitis, such as pool palms and feet. A thorough history and physical examination are helpful in determining the diagnosis.
Practical Implications
It is important to consider and recognize this localized pressure phenomenon of pool palms and feet, thus obviating an unnecessary workup or therapeutic interventions. Specifically, a finding of erythematous asymptomatic macules, with or without scaling, on bony prominences of the palms and soles is more consistent with pool palms and feet.
Pernio and COVID toes both present as erythematous to violaceous papules and macules, with edema, vesiculation, and ulceration in severe cases, often on the dorsum and sides of fingers and toes; typically the conditions are pruritic and painful at times.
Explaining the diagnosis of pool palms and feet and sharing one’s experience with similar cases might help alleviate parental fear and anxiety during the COVID-19 pandemic.
- de Masson A, Bouaziz J-D, Sulimovic L, et al; SNDV (French National Union of Dermatologists–Venereologists). Chilblains is a common cutaneous finding during the COVID-19 pandemic: a retrospective nationwide study from France. J Am Acad Dermatol. 2020;83:667-670. doi:10.1016/j.jaad.2020.04.161
- Freeman EE, McMahon DE, Lipoff JB, et al; American Academy of Dermatology Ad Hoc Task Force on COVID-19. Pernio-like skin lesions associated with COVID-19: a case series of 318 patients from 8 countries. J Am Acad Dermatol. 2020;83:486-492. doi:10.1016/j.jaad.2020.05.109
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129. doi:10.1016/j.jaad.2020.06.1016
- Blauvelt A, Duarte AM, Schachner LA. Pool palms. J Am Acad Dermatol. 1992;27:111. doi:10.1016/s0190-9622(08)80819-5
- Wong L-C, Rogers M. Pool palms. Pediatr Dermatol. 2007;24:95. doi:10.1111/j.1525-1470.2007.00347.x
- Novoa A, Klear S. Pool palms. Arch Dis Child. 2016;101:41. doi:10.1136/archdischild-2015-309633
- Morgado-Carasco D, Feola H, Vargas-Mora P. Pool palms. Dermatol Pract Concept. 2020;10:e2020009. doi:10.5826/dpc.1001a09
- Cutrone M, Valerio E, Grimalt R. Pool palms: a case report. Dermatol Case Rep. 2019;4:1000154.
- Martína JM, Ricart JM. Erythematous–violaceous lesions on the palms. Actas Dermosifiliogr. 2009;100:507-508.
- de Masson A, Bouaziz J-D, Sulimovic L, et al; SNDV (French National Union of Dermatologists–Venereologists). Chilblains is a common cutaneous finding during the COVID-19 pandemic: a retrospective nationwide study from France. J Am Acad Dermatol. 2020;83:667-670. doi:10.1016/j.jaad.2020.04.161
- Freeman EE, McMahon DE, Lipoff JB, et al; American Academy of Dermatology Ad Hoc Task Force on COVID-19. Pernio-like skin lesions associated with COVID-19: a case series of 318 patients from 8 countries. J Am Acad Dermatol. 2020;83:486-492. doi:10.1016/j.jaad.2020.05.109
- Freeman EE, McMahon DE, Lipoff JB, et al. The spectrum of COVID-19-associated dermatologic manifestations: an international registry of 716 patients from 31 countries. J Am Acad Dermatol. 2020;83:1118-1129. doi:10.1016/j.jaad.2020.06.1016
- Blauvelt A, Duarte AM, Schachner LA. Pool palms. J Am Acad Dermatol. 1992;27:111. doi:10.1016/s0190-9622(08)80819-5
- Wong L-C, Rogers M. Pool palms. Pediatr Dermatol. 2007;24:95. doi:10.1111/j.1525-1470.2007.00347.x
- Novoa A, Klear S. Pool palms. Arch Dis Child. 2016;101:41. doi:10.1136/archdischild-2015-309633
- Morgado-Carasco D, Feola H, Vargas-Mora P. Pool palms. Dermatol Pract Concept. 2020;10:e2020009. doi:10.5826/dpc.1001a09
- Cutrone M, Valerio E, Grimalt R. Pool palms: a case report. Dermatol Case Rep. 2019;4:1000154.
- Martína JM, Ricart JM. Erythematous–violaceous lesions on the palms. Actas Dermosifiliogr. 2009;100:507-508.
Management of Pediatric Nail Psoriasis
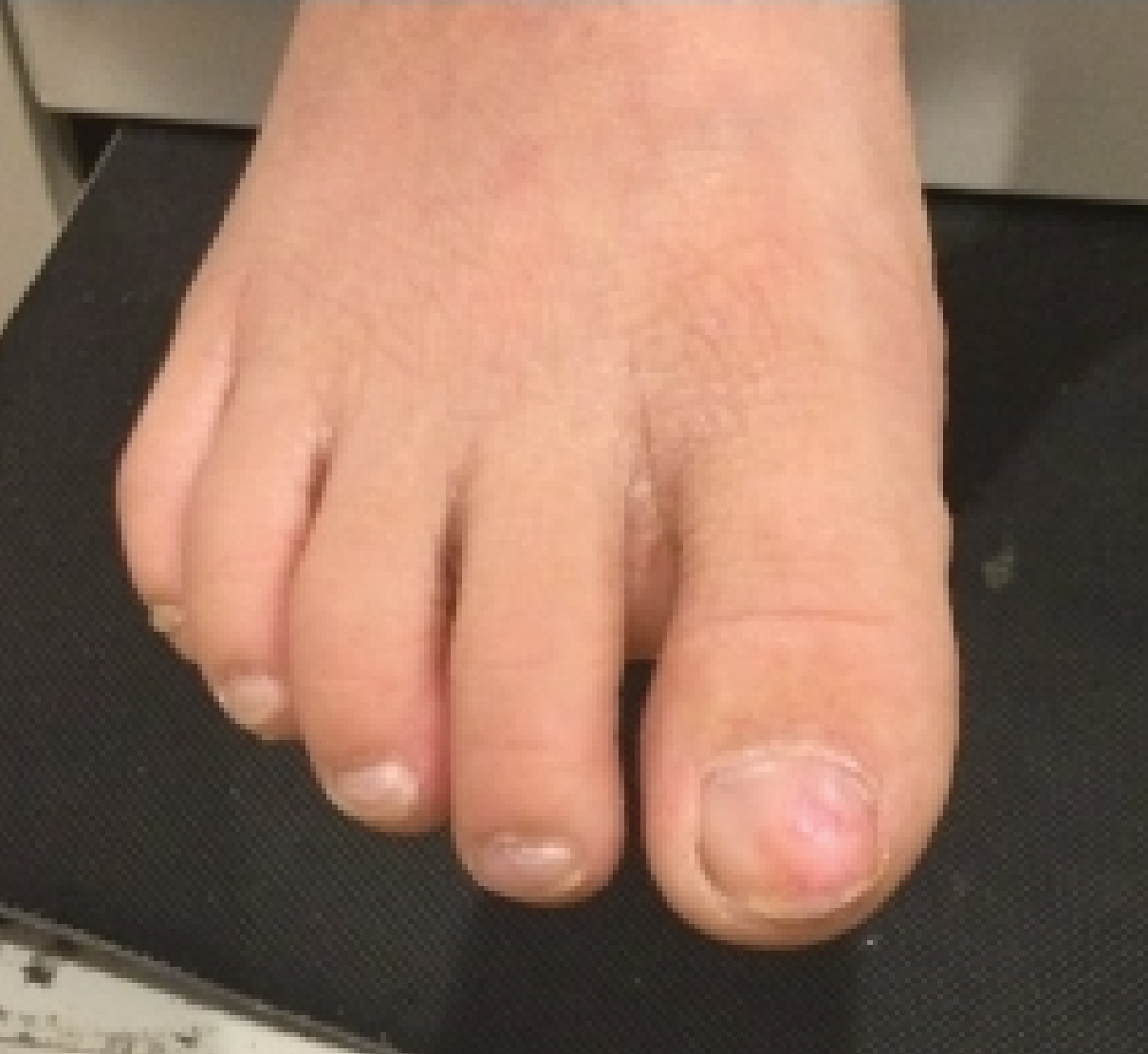
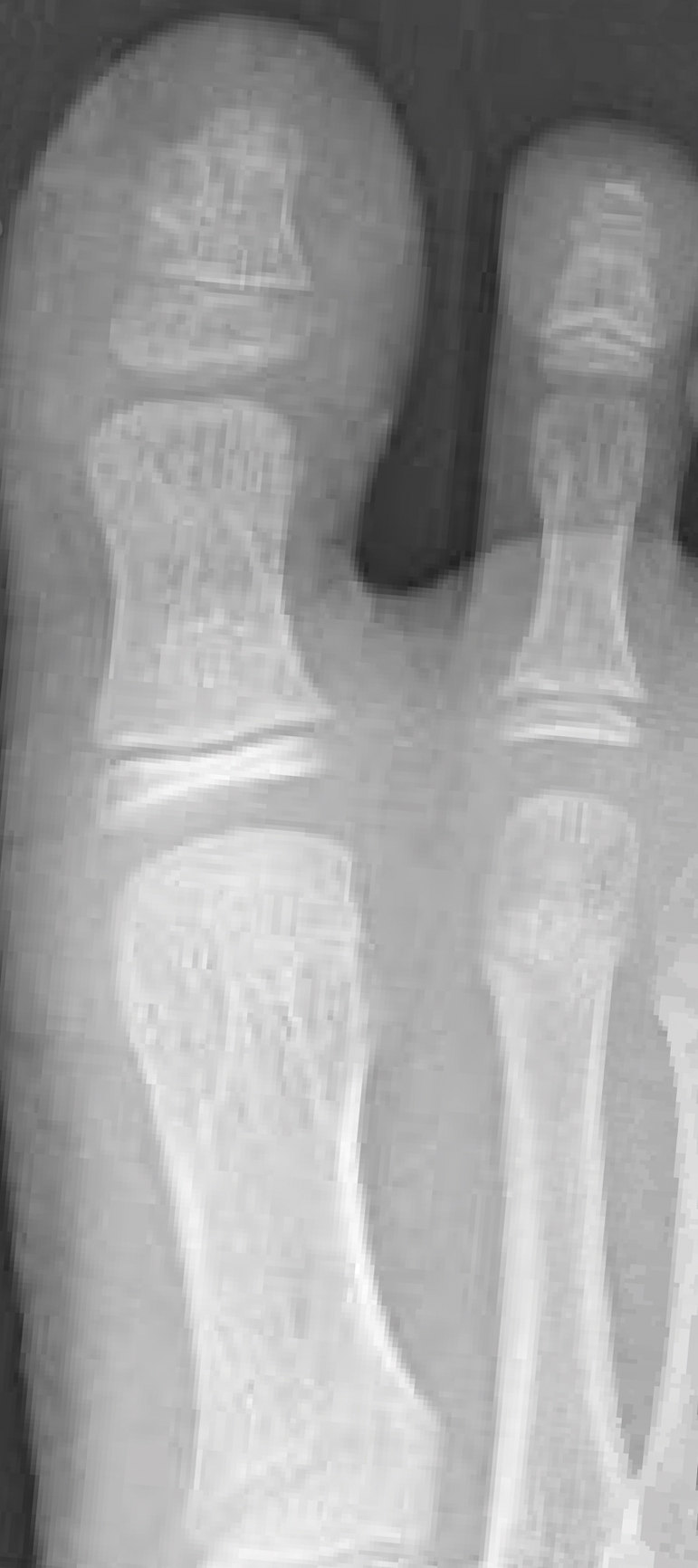
Pediatric nail psoriasis is a condition that has not been extensively studied. The prevalence of nail alterations in pediatric patients with psoriasis varies among different studies, ranging from 17% to 39.2%.1 Nail pitting, onycholysis associated with subungual hyperkeratosis, paronychia, and pachyonychia are the most frequent features of psoriatic nail involvement in children.2,3 The management of nail psoriasis in children and adolescents is critical due to the quality-of-life impact, from potential functional impairment issues to the obvious cosmetic problems, which can aggravate the psychologic distress and social embarrassment of patients with psoriasis. Despite the emergence of modern potent systemic agents to treat chronic plaque psoriasis, nail psoriasis often is refractory to treatment.4 Coupled with the limited on-label options for psoriasis treatment in children, the management of nail psoriasis in this special patient group constitutes an even greater therapeutic challenge. This report aims to summarize the limited existing data on the successful management of nail psoriasis in the pediatric population.
Reviewing the Literature on Nail Psoriasis
We conducted a search of PubMed articles indexed for MEDLINE, Embase, and Scopus using the following Medical Subject Headings key terms: nail psoriasis and children, juvenile, pediatric. Additional articles were identified from the reference lists of the retrieved articles and citations. Our search included reports in the English language published from 2000 to 2019. The selection process included the following 2 steps: screening of the titles and abstracts, followed by evaluation of the selected full-text articles.
Topical Treatments for Nail Psoriasis
Because most systemic antipsoriatic treatments that can be administered in adult patients have not yet been granted an official license for administration in children, topical treatments are considered by many physicians as the preferred first-line therapy for psoriatic nail involvement in pediatric patients.5,6 However, only scarce data are available in the literature concerning the successful use of local agents in pediatric patients with psoriasis.
The main limitation of local treatments relates mostly to their impaired penetration into the affected area (nails). To optimize drug penetration, some authors suggest the use of potent keratolytic topical preparations to reduce the nail volume and facilitate drug absorption.7 A popular suggestion is trimming the onycholytic nail plate followed by 40% urea avulsion to treat subungual hyperkeratosis8 or simply the use of occlusive 40% urea in petroleum jelly.9 Other approaches include clipping the onycholytic nail over the diseased nail bed or processing the nail plate through grinding or even drilling holes with the use of mechanical burrs or ablative lasers to enhance the penetration of the topical agent.7
A frequent approach in pediatric patients is clipping the detached nails combined with daily application of calcipotriene (calcipotriol) and steroids, such as betamethasone dipropionate.5,8 Reports on the use of regimens with clobetasol propionate ointment 0.05% under occlusion, with or without the concomitant use of calcipotriol solution 0.005%, also are present in the literature but not always with satisfactory results.10,11 Another successfully administered topical steroid is mometasone furoate cream 0.1%.12 Although the use of intralesional triamcinolone acetonide also has demonstrated encouraging outcomes in isolated reports,13 associated adverse events, such as pain and hematomas, can result in tolerability issues for pediatric patients.7
Piraccini et al14 described the case of an 8-year-old patient with pustular nail psoriasis who showed improvement within 3 to 6 months of treatment with topical calcipotriol 5 μg/g as monotherapy applied to the nail and periungual tissues twice daily. Another approach, described by Diluvio et al,15 is the use of tazarotene gel 0.05% applied once daily to the affected nail plates, nail folds, and periungual skin without occlusion. In a 6-year-old patient with isolated nail psoriasis, this treatment regimen demonstrated notable improvement within 8 weeks.15
Systemic Treatments for Nail Psoriasis
Data on the successful administration of systemic agents in pediatric patients also are extremely scarce. Due to the lack of clinical trials, everyday practice is mostly based on isolated case series and case reports.
Methotrexate—Lee11 described the case of an 11-year-old girl with severe, symptomatic, 20-nail psoriatic onychodystrophy who showed a complete response to oral methotrexate 5 mg/wk after topical clobetasol propionate and calcipotriol failed. Improvement was seen as early as 4 weeks after therapy initiation, and complete resolution of the lesions was documented after 9 and 13 months of methotrexate therapy for the fingers and toes, respectively.11 The successful use of methotrexate in the improvement of psoriatic nail dystrophy in a pediatric patient also was documented by Teran et al.16 In this case, a 9-year-old girl with erythrodermic psoriasis, psoriatic arthritis, and severe onychodystrophy showed notable amelioration of all psoriatic manifestations, including the nail findings, with systemic methotrexate therapy (dose not specified).16 Notably, the authors reported that the improvement of onychodystrophy occurred with considerable delay compared to the other psoriatic lesions,16 indicating the already-known refractoriness of nail psoriasis to the various therapeutic attempts.9-15
Acitretin—Another agent that has been linked with partial improvement of acrodermatitis continua of Hallopeau (ACH)–associated onychodystrophy is acitretin. In a case series of 15 pediatric patients with pustular psoriasis, a 5-year-old boy with severe nail involvement presented with partial amelioration of nail changes with acitretin within the first 6 weeks of treatment using the following regimen: initial dosage of 0.8 mg/kg/d for 6 weeks, followed by 0.3 mg/kg/d for 4 weeks.17
Biologics—The emerging use of biologics in pediatric psoriasis also has brought important advances in the successful management of nail psoriasis in children and adolescents.18-21 Wells et al18 presented the case of an 8-year-old girl with nail psoriasis, psoriatic arthritis, and plaque psoriasis who showed complete resolution of all psoriatic manifestations, including nail involvement, within 3 months of treatment with secukinumab 150 mg subcutaneously every 4 weeks. Prior failed treatments included various systemic agents (ie, subcutaneous methotrexate 20 mg/m2, etanercept 0.8 mg/kg weekly, adalimumab 40 mg every 2 weeks) as well as topical agents (ie, urea, tazarotene, corticosteroids) and intralesional triamcinolone.18
Infliximab also has been successfully used for pediatric nail psoriasis. Watabe et al19 presented the case of an 8-year-old girl with psoriatic onychodystrophy in addition to psoriatic onycho-pachydermo-periostitis. Prior therapy with adalimumab 20 mg every other week combined with methotrexate 10 mg weekly failed. She experienced notable amelioration of the nail dystrophy within 3 months of using a combination of infliximab and methotrexate (infliximab 5 mg/kg intravenously on weeks 0, 2, and 6, and every 8 weeks thereafter; methotrexate 10 mg/wk).19
Cases in which infliximab has resulted in rapid yet only transient restoration of psoriatic onychodystrophy also are present in the literature. Pereira et al20 reported that a 3-year-old patient with severe 20-digit onychodystrophy in addition to pustular psoriasis had complete resolution of nail lesions within 2 weeks of treatment with infliximab (5 mg/kg at weeks 0, 2, and 6, and then every 7 weeks thereafter), which was sustained over the course of 1 year. The therapy had to be discontinued because of exacerbation of the cutaneous symptoms; thereafter, etanercept was initiated. Although the patient noted major improvement of all skin lesions under etanercept, only moderate amelioration of the psoriatic nail lesions was demonstrated.20
Dini et al21 described a 9-year-old girl with severe ACH-associated psoriatic onychodystrophy who showed complete clearance of all lesions within 8 weeks of treatment with adalimumab (initially 80 mg, followed by 40 mg after 1 week and then 40 mg every other week). Prior treatment with potent topical corticosteroids, cyclosporine (3 mg/kg/d for 6 months), and etanercept (0.4 mg/kg twice weekly for 3 months) was ineffective.21
Phototherapy—Other systemic agents with reported satisfactory outcomes in the treatment of psoriatic onychodystrophy include thalidomide combined with UVB phototherapy. Kiszewski et al22 described a 2-year-old patient with ACH and severe 19-digit onychodystrophy. Prior failed therapies included occluded clobetasol ointment 0.05%, occluded pimecrolimus 0.1%, and systemic methotrexate, while systemic acitretin (0.8 mg⁄kg⁄d) resulted in elevated cholesterol levels and therefore had to be interrupted. Improvement was seen 2 months after the initiation of a combined broadband UVB and thalidomide (50 mg⁄d) treatment, with no documented relapses after discontinuation of therapy.22
Narrowband UVB (311 nm) also has been used as monotherapy for ACH-associated onychodystrophy, as demonstrated by Bordignon et al.23 They reported a 9-year-old patient who showed partial improvement of isolated onychodystrophy of the fourth nail plate of the left hand after 36 sessions of narrowband UVB using a 311-nm filtering handpiece with a square spot size of 19×19 mm.23
Conclusion
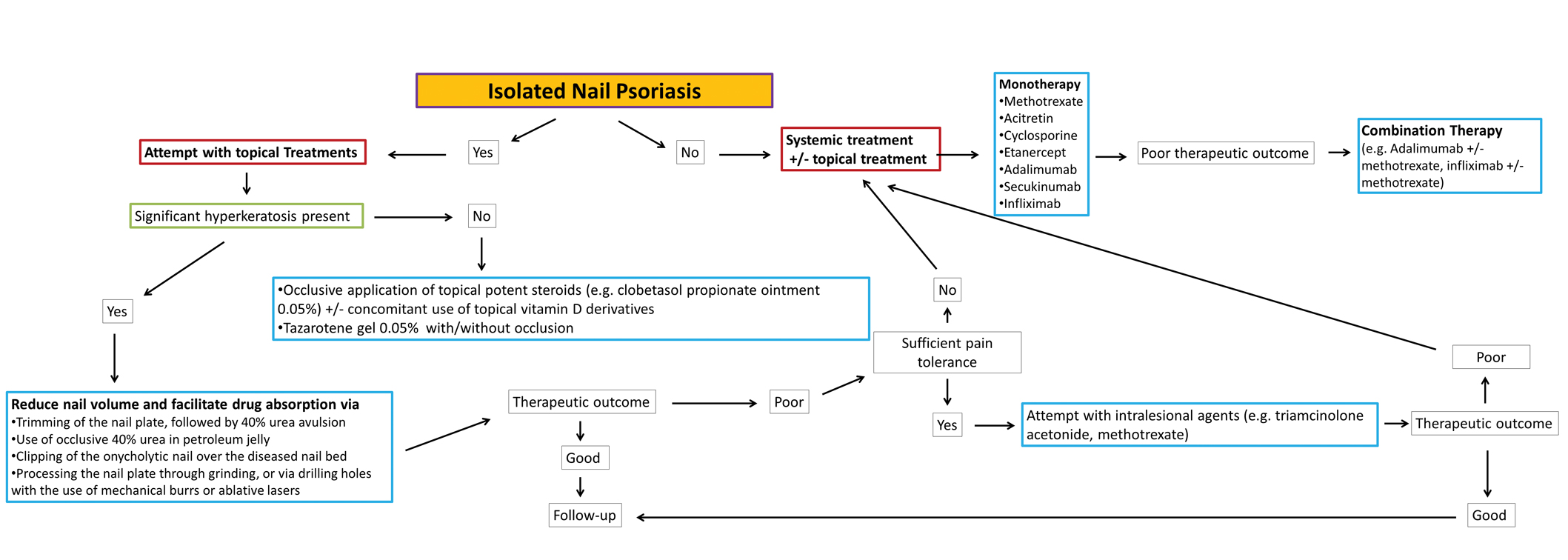
Nail psoriasis constitutes a type of psoriasis that is not only refractory to most treatments but is accompanied by substantial psychological and occasionally functional burden for the affected individuals.24 Data concerning therapeutic options in the pediatric population are extremely limited, and therefore the everyday practice often involves administration of off-label medications, which can constitute a dilemma for many physicians, especially for safety.10 We suggest a simple therapeutic algorithm for the management of pediatric nail psoriasis based on the summarized data that are currently available in the literature. This algorithm is shown in the eFigure.
As progressively more agents—especially biologics—receive approval for use in plaque psoriasis in pediatric patients,25 it is expected that gradually more real-life data on their side efficacy for plaque psoriasis of the nails in children also will come to light. Furthermore, their on-label use in pediatric psoriasis patients will facilitate further relevant clinical trials to this target group so that the potential of these medications in the management of nail psoriasis can be fully explored.
- Uber M, Carvalho VO, Abagge KT, et al. Clinical features and nail clippings in 52 children with psoriasis. Pediatr Dermatol. 2018;35:202-207.
- Pourchot D, Bodemer C, Phan A, et al. Nail psoriasis: a systematic evaluation in 313 children with psoriasis. Pediatr Dermatol. 2017;34:58-63.
- Piraccini BM, Triantafyllopoulou I, Prevezas C, et al. Nail psoriasis in children: common or uncommon? results from a 10-year double-center study. Skin Appendage Disord. 2015;1:43-48.
- Baran R. The burden of nail psoriasis: an introduction. Dermatology. 2010;221(suppl 1):1-5.
- Richert B, André J. Nail disorders in children: diagnosis and management. Am J Clin Dermatol. 2011;12:101-112.
- Trüeb RM. Therapies for childhood psoriasis. Curr Probl Dermatol. 2009;38:137-159.
- Haneke E. Nail psoriasis: clinical features, pathogenesis, differential diagnoses, and management. Psoriasis (Auckl). 2017;7:51-63.
- Piraccini BM, Starace M. Nail disorders in infants and children. Curr Opin Pediatr. 2014;26:440-445.
- Duran-McKinster C, Ortiz-Solis D, Granados J, et al. Juvenile psoriatic arthritis with nail psoriasis in the absence of cutaneous lesions. Int J Dermatol. 2000;39:32-35.
- Holzberg M, Ruben BS, Baran R. Psoriasis restricted to the nail in a 7-year-old child. should biologics be an appropriate treatment modality when considering quality of life? J Eur Acad Dermatol Venereol. 2014;28:668-670.
- Lee JY. Severe 20-nail psoriasis successfully treated by low dose methotrexate. Dermatol Online J. 2009;15:8.
- Liao YC, Lee JY. Psoriasis in a 3-month-old infant with Kawasaki disease. Dermatol Online J. 2009;15:10.
- Khoo BP, Giam YC. A pilot study on the role of intralesional triamcinolone acetonide in the treatment of pitted nails in children. Singapore Med J. 2000;41:66-68.
- Piraccini BM, Tosti A, Iorizzo M, et al. Pustular psoriasis of the nails: treatment and long-term follow-up of 46 patients. Br J Dermatol. 2001;144:1000-1005.
- Diluvio L, Campione E, Paternò EJ, et al. Childhood nail psoriasis: a useful treatment with tazarotene 0.05%. Pediatr Dermatol. 2007;24:332-333.
- Teran CG, Teran-Escalera CN, Balderrama C. A severe case of erythrodermic psoriasis associated with advanced nail and joint manifestations: a case report. J Med Case Rep. 2010;4:179.
- Chen P, Li C, Xue R, et al. Efficacy and safety of acitretin monotherapy in children with pustular psoriasis: results from 15 cases and a literature review. J Dermatolog Treat. 2018;29:353-363.
- Wells LE, Evans T, Hilton R, et al. Use of secukinumab in a pediatric patient leads to significant improvement in nail psoriasis and psoriatic arthritis. Pediatr Dermatol. 2019;36:384-385.
- Watabe D, Endoh K, Maeda F, et al. Childhood-onset psoriatic onycho-pachydermo-periostitis treated successfully with infliximab. Eur J Dermatol. 2015;25:506-508.
- Pereira TM, Vieira AP, Fernandes JC, et al. Anti-TNF-alpha therapy in childhood pustular psoriasis. Dermatology. 2006;213:350-352.
- Dini V, Barbanera S, Romanelli M. Efficacy of adalimumab for the treatment of refractory paediatric acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2013;93:588-589.
- Kiszewski AE, De Villa D, Scheibel I, et al. An infant with acrodermatitis continua of Hallopeau: successful treatment with thalidomide and UVB therapy. Pediatr Dermatol. 2009;26:105-106.
- Bordignon M, Zattra E, Albertin C, et al. Successful treatment of a 9-year-old boy affected by acrodermatitis continua of Hallopeau with targeted ultraviolet B narrow-band phototherapy. Photodermatol Photoimmunol Photomed. 2010;26:41-43.
- Fabroni C, Gori A, Troiano M, et al. Infliximab efficacy in nail psoriasis. a retrospective study in 48 patients. J Eur Acad Dermatol Venereol. 2011;25:549-553.
- Lilly’s Taltz® (ixekizumab) receives U.S. FDA approval for the treatment of pediatric patients with moderate to severe plaque psoriasis. Eli Lilly and Company. March 30, 2020. Accessed September 24, 2021. https://investor.lilly.com/news-releases/news-release-details/lillys-taltzr-ixekizumab-receives-us-fda-approval-treatment-1
Pediatric nail psoriasis is a condition that has not been extensively studied. The prevalence of nail alterations in pediatric patients with psoriasis varies among different studies, ranging from 17% to 39.2%.1 Nail pitting, onycholysis associated with subungual hyperkeratosis, paronychia, and pachyonychia are the most frequent features of psoriatic nail involvement in children.2,3 The management of nail psoriasis in children and adolescents is critical due to the quality-of-life impact, from potential functional impairment issues to the obvious cosmetic problems, which can aggravate the psychologic distress and social embarrassment of patients with psoriasis. Despite the emergence of modern potent systemic agents to treat chronic plaque psoriasis, nail psoriasis often is refractory to treatment.4 Coupled with the limited on-label options for psoriasis treatment in children, the management of nail psoriasis in this special patient group constitutes an even greater therapeutic challenge. This report aims to summarize the limited existing data on the successful management of nail psoriasis in the pediatric population.
Reviewing the Literature on Nail Psoriasis
We conducted a search of PubMed articles indexed for MEDLINE, Embase, and Scopus using the following Medical Subject Headings key terms: nail psoriasis and children, juvenile, pediatric. Additional articles were identified from the reference lists of the retrieved articles and citations. Our search included reports in the English language published from 2000 to 2019. The selection process included the following 2 steps: screening of the titles and abstracts, followed by evaluation of the selected full-text articles.
Topical Treatments for Nail Psoriasis
Because most systemic antipsoriatic treatments that can be administered in adult patients have not yet been granted an official license for administration in children, topical treatments are considered by many physicians as the preferred first-line therapy for psoriatic nail involvement in pediatric patients.5,6 However, only scarce data are available in the literature concerning the successful use of local agents in pediatric patients with psoriasis.
The main limitation of local treatments relates mostly to their impaired penetration into the affected area (nails). To optimize drug penetration, some authors suggest the use of potent keratolytic topical preparations to reduce the nail volume and facilitate drug absorption.7 A popular suggestion is trimming the onycholytic nail plate followed by 40% urea avulsion to treat subungual hyperkeratosis8 or simply the use of occlusive 40% urea in petroleum jelly.9 Other approaches include clipping the onycholytic nail over the diseased nail bed or processing the nail plate through grinding or even drilling holes with the use of mechanical burrs or ablative lasers to enhance the penetration of the topical agent.7
A frequent approach in pediatric patients is clipping the detached nails combined with daily application of calcipotriene (calcipotriol) and steroids, such as betamethasone dipropionate.5,8 Reports on the use of regimens with clobetasol propionate ointment 0.05% under occlusion, with or without the concomitant use of calcipotriol solution 0.005%, also are present in the literature but not always with satisfactory results.10,11 Another successfully administered topical steroid is mometasone furoate cream 0.1%.12 Although the use of intralesional triamcinolone acetonide also has demonstrated encouraging outcomes in isolated reports,13 associated adverse events, such as pain and hematomas, can result in tolerability issues for pediatric patients.7
Piraccini et al14 described the case of an 8-year-old patient with pustular nail psoriasis who showed improvement within 3 to 6 months of treatment with topical calcipotriol 5 μg/g as monotherapy applied to the nail and periungual tissues twice daily. Another approach, described by Diluvio et al,15 is the use of tazarotene gel 0.05% applied once daily to the affected nail plates, nail folds, and periungual skin without occlusion. In a 6-year-old patient with isolated nail psoriasis, this treatment regimen demonstrated notable improvement within 8 weeks.15
Systemic Treatments for Nail Psoriasis
Data on the successful administration of systemic agents in pediatric patients also are extremely scarce. Due to the lack of clinical trials, everyday practice is mostly based on isolated case series and case reports.
Methotrexate—Lee11 described the case of an 11-year-old girl with severe, symptomatic, 20-nail psoriatic onychodystrophy who showed a complete response to oral methotrexate 5 mg/wk after topical clobetasol propionate and calcipotriol failed. Improvement was seen as early as 4 weeks after therapy initiation, and complete resolution of the lesions was documented after 9 and 13 months of methotrexate therapy for the fingers and toes, respectively.11 The successful use of methotrexate in the improvement of psoriatic nail dystrophy in a pediatric patient also was documented by Teran et al.16 In this case, a 9-year-old girl with erythrodermic psoriasis, psoriatic arthritis, and severe onychodystrophy showed notable amelioration of all psoriatic manifestations, including the nail findings, with systemic methotrexate therapy (dose not specified).16 Notably, the authors reported that the improvement of onychodystrophy occurred with considerable delay compared to the other psoriatic lesions,16 indicating the already-known refractoriness of nail psoriasis to the various therapeutic attempts.9-15
Acitretin—Another agent that has been linked with partial improvement of acrodermatitis continua of Hallopeau (ACH)–associated onychodystrophy is acitretin. In a case series of 15 pediatric patients with pustular psoriasis, a 5-year-old boy with severe nail involvement presented with partial amelioration of nail changes with acitretin within the first 6 weeks of treatment using the following regimen: initial dosage of 0.8 mg/kg/d for 6 weeks, followed by 0.3 mg/kg/d for 4 weeks.17
Biologics—The emerging use of biologics in pediatric psoriasis also has brought important advances in the successful management of nail psoriasis in children and adolescents.18-21 Wells et al18 presented the case of an 8-year-old girl with nail psoriasis, psoriatic arthritis, and plaque psoriasis who showed complete resolution of all psoriatic manifestations, including nail involvement, within 3 months of treatment with secukinumab 150 mg subcutaneously every 4 weeks. Prior failed treatments included various systemic agents (ie, subcutaneous methotrexate 20 mg/m2, etanercept 0.8 mg/kg weekly, adalimumab 40 mg every 2 weeks) as well as topical agents (ie, urea, tazarotene, corticosteroids) and intralesional triamcinolone.18
Infliximab also has been successfully used for pediatric nail psoriasis. Watabe et al19 presented the case of an 8-year-old girl with psoriatic onychodystrophy in addition to psoriatic onycho-pachydermo-periostitis. Prior therapy with adalimumab 20 mg every other week combined with methotrexate 10 mg weekly failed. She experienced notable amelioration of the nail dystrophy within 3 months of using a combination of infliximab and methotrexate (infliximab 5 mg/kg intravenously on weeks 0, 2, and 6, and every 8 weeks thereafter; methotrexate 10 mg/wk).19
Cases in which infliximab has resulted in rapid yet only transient restoration of psoriatic onychodystrophy also are present in the literature. Pereira et al20 reported that a 3-year-old patient with severe 20-digit onychodystrophy in addition to pustular psoriasis had complete resolution of nail lesions within 2 weeks of treatment with infliximab (5 mg/kg at weeks 0, 2, and 6, and then every 7 weeks thereafter), which was sustained over the course of 1 year. The therapy had to be discontinued because of exacerbation of the cutaneous symptoms; thereafter, etanercept was initiated. Although the patient noted major improvement of all skin lesions under etanercept, only moderate amelioration of the psoriatic nail lesions was demonstrated.20
Dini et al21 described a 9-year-old girl with severe ACH-associated psoriatic onychodystrophy who showed complete clearance of all lesions within 8 weeks of treatment with adalimumab (initially 80 mg, followed by 40 mg after 1 week and then 40 mg every other week). Prior treatment with potent topical corticosteroids, cyclosporine (3 mg/kg/d for 6 months), and etanercept (0.4 mg/kg twice weekly for 3 months) was ineffective.21
Phototherapy—Other systemic agents with reported satisfactory outcomes in the treatment of psoriatic onychodystrophy include thalidomide combined with UVB phototherapy. Kiszewski et al22 described a 2-year-old patient with ACH and severe 19-digit onychodystrophy. Prior failed therapies included occluded clobetasol ointment 0.05%, occluded pimecrolimus 0.1%, and systemic methotrexate, while systemic acitretin (0.8 mg⁄kg⁄d) resulted in elevated cholesterol levels and therefore had to be interrupted. Improvement was seen 2 months after the initiation of a combined broadband UVB and thalidomide (50 mg⁄d) treatment, with no documented relapses after discontinuation of therapy.22
Narrowband UVB (311 nm) also has been used as monotherapy for ACH-associated onychodystrophy, as demonstrated by Bordignon et al.23 They reported a 9-year-old patient who showed partial improvement of isolated onychodystrophy of the fourth nail plate of the left hand after 36 sessions of narrowband UVB using a 311-nm filtering handpiece with a square spot size of 19×19 mm.23
Conclusion
Nail psoriasis constitutes a type of psoriasis that is not only refractory to most treatments but is accompanied by substantial psychological and occasionally functional burden for the affected individuals.24 Data concerning therapeutic options in the pediatric population are extremely limited, and therefore the everyday practice often involves administration of off-label medications, which can constitute a dilemma for many physicians, especially for safety.10 We suggest a simple therapeutic algorithm for the management of pediatric nail psoriasis based on the summarized data that are currently available in the literature. This algorithm is shown in the eFigure.
As progressively more agents—especially biologics—receive approval for use in plaque psoriasis in pediatric patients,25 it is expected that gradually more real-life data on their side efficacy for plaque psoriasis of the nails in children also will come to light. Furthermore, their on-label use in pediatric psoriasis patients will facilitate further relevant clinical trials to this target group so that the potential of these medications in the management of nail psoriasis can be fully explored.
Pediatric nail psoriasis is a condition that has not been extensively studied. The prevalence of nail alterations in pediatric patients with psoriasis varies among different studies, ranging from 17% to 39.2%.1 Nail pitting, onycholysis associated with subungual hyperkeratosis, paronychia, and pachyonychia are the most frequent features of psoriatic nail involvement in children.2,3 The management of nail psoriasis in children and adolescents is critical due to the quality-of-life impact, from potential functional impairment issues to the obvious cosmetic problems, which can aggravate the psychologic distress and social embarrassment of patients with psoriasis. Despite the emergence of modern potent systemic agents to treat chronic plaque psoriasis, nail psoriasis often is refractory to treatment.4 Coupled with the limited on-label options for psoriasis treatment in children, the management of nail psoriasis in this special patient group constitutes an even greater therapeutic challenge. This report aims to summarize the limited existing data on the successful management of nail psoriasis in the pediatric population.
Reviewing the Literature on Nail Psoriasis
We conducted a search of PubMed articles indexed for MEDLINE, Embase, and Scopus using the following Medical Subject Headings key terms: nail psoriasis and children, juvenile, pediatric. Additional articles were identified from the reference lists of the retrieved articles and citations. Our search included reports in the English language published from 2000 to 2019. The selection process included the following 2 steps: screening of the titles and abstracts, followed by evaluation of the selected full-text articles.
Topical Treatments for Nail Psoriasis
Because most systemic antipsoriatic treatments that can be administered in adult patients have not yet been granted an official license for administration in children, topical treatments are considered by many physicians as the preferred first-line therapy for psoriatic nail involvement in pediatric patients.5,6 However, only scarce data are available in the literature concerning the successful use of local agents in pediatric patients with psoriasis.
The main limitation of local treatments relates mostly to their impaired penetration into the affected area (nails). To optimize drug penetration, some authors suggest the use of potent keratolytic topical preparations to reduce the nail volume and facilitate drug absorption.7 A popular suggestion is trimming the onycholytic nail plate followed by 40% urea avulsion to treat subungual hyperkeratosis8 or simply the use of occlusive 40% urea in petroleum jelly.9 Other approaches include clipping the onycholytic nail over the diseased nail bed or processing the nail plate through grinding or even drilling holes with the use of mechanical burrs or ablative lasers to enhance the penetration of the topical agent.7
A frequent approach in pediatric patients is clipping the detached nails combined with daily application of calcipotriene (calcipotriol) and steroids, such as betamethasone dipropionate.5,8 Reports on the use of regimens with clobetasol propionate ointment 0.05% under occlusion, with or without the concomitant use of calcipotriol solution 0.005%, also are present in the literature but not always with satisfactory results.10,11 Another successfully administered topical steroid is mometasone furoate cream 0.1%.12 Although the use of intralesional triamcinolone acetonide also has demonstrated encouraging outcomes in isolated reports,13 associated adverse events, such as pain and hematomas, can result in tolerability issues for pediatric patients.7
Piraccini et al14 described the case of an 8-year-old patient with pustular nail psoriasis who showed improvement within 3 to 6 months of treatment with topical calcipotriol 5 μg/g as monotherapy applied to the nail and periungual tissues twice daily. Another approach, described by Diluvio et al,15 is the use of tazarotene gel 0.05% applied once daily to the affected nail plates, nail folds, and periungual skin without occlusion. In a 6-year-old patient with isolated nail psoriasis, this treatment regimen demonstrated notable improvement within 8 weeks.15
Systemic Treatments for Nail Psoriasis
Data on the successful administration of systemic agents in pediatric patients also are extremely scarce. Due to the lack of clinical trials, everyday practice is mostly based on isolated case series and case reports.
Methotrexate—Lee11 described the case of an 11-year-old girl with severe, symptomatic, 20-nail psoriatic onychodystrophy who showed a complete response to oral methotrexate 5 mg/wk after topical clobetasol propionate and calcipotriol failed. Improvement was seen as early as 4 weeks after therapy initiation, and complete resolution of the lesions was documented after 9 and 13 months of methotrexate therapy for the fingers and toes, respectively.11 The successful use of methotrexate in the improvement of psoriatic nail dystrophy in a pediatric patient also was documented by Teran et al.16 In this case, a 9-year-old girl with erythrodermic psoriasis, psoriatic arthritis, and severe onychodystrophy showed notable amelioration of all psoriatic manifestations, including the nail findings, with systemic methotrexate therapy (dose not specified).16 Notably, the authors reported that the improvement of onychodystrophy occurred with considerable delay compared to the other psoriatic lesions,16 indicating the already-known refractoriness of nail psoriasis to the various therapeutic attempts.9-15
Acitretin—Another agent that has been linked with partial improvement of acrodermatitis continua of Hallopeau (ACH)–associated onychodystrophy is acitretin. In a case series of 15 pediatric patients with pustular psoriasis, a 5-year-old boy with severe nail involvement presented with partial amelioration of nail changes with acitretin within the first 6 weeks of treatment using the following regimen: initial dosage of 0.8 mg/kg/d for 6 weeks, followed by 0.3 mg/kg/d for 4 weeks.17
Biologics—The emerging use of biologics in pediatric psoriasis also has brought important advances in the successful management of nail psoriasis in children and adolescents.18-21 Wells et al18 presented the case of an 8-year-old girl with nail psoriasis, psoriatic arthritis, and plaque psoriasis who showed complete resolution of all psoriatic manifestations, including nail involvement, within 3 months of treatment with secukinumab 150 mg subcutaneously every 4 weeks. Prior failed treatments included various systemic agents (ie, subcutaneous methotrexate 20 mg/m2, etanercept 0.8 mg/kg weekly, adalimumab 40 mg every 2 weeks) as well as topical agents (ie, urea, tazarotene, corticosteroids) and intralesional triamcinolone.18
Infliximab also has been successfully used for pediatric nail psoriasis. Watabe et al19 presented the case of an 8-year-old girl with psoriatic onychodystrophy in addition to psoriatic onycho-pachydermo-periostitis. Prior therapy with adalimumab 20 mg every other week combined with methotrexate 10 mg weekly failed. She experienced notable amelioration of the nail dystrophy within 3 months of using a combination of infliximab and methotrexate (infliximab 5 mg/kg intravenously on weeks 0, 2, and 6, and every 8 weeks thereafter; methotrexate 10 mg/wk).19
Cases in which infliximab has resulted in rapid yet only transient restoration of psoriatic onychodystrophy also are present in the literature. Pereira et al20 reported that a 3-year-old patient with severe 20-digit onychodystrophy in addition to pustular psoriasis had complete resolution of nail lesions within 2 weeks of treatment with infliximab (5 mg/kg at weeks 0, 2, and 6, and then every 7 weeks thereafter), which was sustained over the course of 1 year. The therapy had to be discontinued because of exacerbation of the cutaneous symptoms; thereafter, etanercept was initiated. Although the patient noted major improvement of all skin lesions under etanercept, only moderate amelioration of the psoriatic nail lesions was demonstrated.20
Dini et al21 described a 9-year-old girl with severe ACH-associated psoriatic onychodystrophy who showed complete clearance of all lesions within 8 weeks of treatment with adalimumab (initially 80 mg, followed by 40 mg after 1 week and then 40 mg every other week). Prior treatment with potent topical corticosteroids, cyclosporine (3 mg/kg/d for 6 months), and etanercept (0.4 mg/kg twice weekly for 3 months) was ineffective.21
Phototherapy—Other systemic agents with reported satisfactory outcomes in the treatment of psoriatic onychodystrophy include thalidomide combined with UVB phototherapy. Kiszewski et al22 described a 2-year-old patient with ACH and severe 19-digit onychodystrophy. Prior failed therapies included occluded clobetasol ointment 0.05%, occluded pimecrolimus 0.1%, and systemic methotrexate, while systemic acitretin (0.8 mg⁄kg⁄d) resulted in elevated cholesterol levels and therefore had to be interrupted. Improvement was seen 2 months after the initiation of a combined broadband UVB and thalidomide (50 mg⁄d) treatment, with no documented relapses after discontinuation of therapy.22
Narrowband UVB (311 nm) also has been used as monotherapy for ACH-associated onychodystrophy, as demonstrated by Bordignon et al.23 They reported a 9-year-old patient who showed partial improvement of isolated onychodystrophy of the fourth nail plate of the left hand after 36 sessions of narrowband UVB using a 311-nm filtering handpiece with a square spot size of 19×19 mm.23
Conclusion
Nail psoriasis constitutes a type of psoriasis that is not only refractory to most treatments but is accompanied by substantial psychological and occasionally functional burden for the affected individuals.24 Data concerning therapeutic options in the pediatric population are extremely limited, and therefore the everyday practice often involves administration of off-label medications, which can constitute a dilemma for many physicians, especially for safety.10 We suggest a simple therapeutic algorithm for the management of pediatric nail psoriasis based on the summarized data that are currently available in the literature. This algorithm is shown in the eFigure.
As progressively more agents—especially biologics—receive approval for use in plaque psoriasis in pediatric patients,25 it is expected that gradually more real-life data on their side efficacy for plaque psoriasis of the nails in children also will come to light. Furthermore, their on-label use in pediatric psoriasis patients will facilitate further relevant clinical trials to this target group so that the potential of these medications in the management of nail psoriasis can be fully explored.
- Uber M, Carvalho VO, Abagge KT, et al. Clinical features and nail clippings in 52 children with psoriasis. Pediatr Dermatol. 2018;35:202-207.
- Pourchot D, Bodemer C, Phan A, et al. Nail psoriasis: a systematic evaluation in 313 children with psoriasis. Pediatr Dermatol. 2017;34:58-63.
- Piraccini BM, Triantafyllopoulou I, Prevezas C, et al. Nail psoriasis in children: common or uncommon? results from a 10-year double-center study. Skin Appendage Disord. 2015;1:43-48.
- Baran R. The burden of nail psoriasis: an introduction. Dermatology. 2010;221(suppl 1):1-5.
- Richert B, André J. Nail disorders in children: diagnosis and management. Am J Clin Dermatol. 2011;12:101-112.
- Trüeb RM. Therapies for childhood psoriasis. Curr Probl Dermatol. 2009;38:137-159.
- Haneke E. Nail psoriasis: clinical features, pathogenesis, differential diagnoses, and management. Psoriasis (Auckl). 2017;7:51-63.
- Piraccini BM, Starace M. Nail disorders in infants and children. Curr Opin Pediatr. 2014;26:440-445.
- Duran-McKinster C, Ortiz-Solis D, Granados J, et al. Juvenile psoriatic arthritis with nail psoriasis in the absence of cutaneous lesions. Int J Dermatol. 2000;39:32-35.
- Holzberg M, Ruben BS, Baran R. Psoriasis restricted to the nail in a 7-year-old child. should biologics be an appropriate treatment modality when considering quality of life? J Eur Acad Dermatol Venereol. 2014;28:668-670.
- Lee JY. Severe 20-nail psoriasis successfully treated by low dose methotrexate. Dermatol Online J. 2009;15:8.
- Liao YC, Lee JY. Psoriasis in a 3-month-old infant with Kawasaki disease. Dermatol Online J. 2009;15:10.
- Khoo BP, Giam YC. A pilot study on the role of intralesional triamcinolone acetonide in the treatment of pitted nails in children. Singapore Med J. 2000;41:66-68.
- Piraccini BM, Tosti A, Iorizzo M, et al. Pustular psoriasis of the nails: treatment and long-term follow-up of 46 patients. Br J Dermatol. 2001;144:1000-1005.
- Diluvio L, Campione E, Paternò EJ, et al. Childhood nail psoriasis: a useful treatment with tazarotene 0.05%. Pediatr Dermatol. 2007;24:332-333.
- Teran CG, Teran-Escalera CN, Balderrama C. A severe case of erythrodermic psoriasis associated with advanced nail and joint manifestations: a case report. J Med Case Rep. 2010;4:179.
- Chen P, Li C, Xue R, et al. Efficacy and safety of acitretin monotherapy in children with pustular psoriasis: results from 15 cases and a literature review. J Dermatolog Treat. 2018;29:353-363.
- Wells LE, Evans T, Hilton R, et al. Use of secukinumab in a pediatric patient leads to significant improvement in nail psoriasis and psoriatic arthritis. Pediatr Dermatol. 2019;36:384-385.
- Watabe D, Endoh K, Maeda F, et al. Childhood-onset psoriatic onycho-pachydermo-periostitis treated successfully with infliximab. Eur J Dermatol. 2015;25:506-508.
- Pereira TM, Vieira AP, Fernandes JC, et al. Anti-TNF-alpha therapy in childhood pustular psoriasis. Dermatology. 2006;213:350-352.
- Dini V, Barbanera S, Romanelli M. Efficacy of adalimumab for the treatment of refractory paediatric acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2013;93:588-589.
- Kiszewski AE, De Villa D, Scheibel I, et al. An infant with acrodermatitis continua of Hallopeau: successful treatment with thalidomide and UVB therapy. Pediatr Dermatol. 2009;26:105-106.
- Bordignon M, Zattra E, Albertin C, et al. Successful treatment of a 9-year-old boy affected by acrodermatitis continua of Hallopeau with targeted ultraviolet B narrow-band phototherapy. Photodermatol Photoimmunol Photomed. 2010;26:41-43.
- Fabroni C, Gori A, Troiano M, et al. Infliximab efficacy in nail psoriasis. a retrospective study in 48 patients. J Eur Acad Dermatol Venereol. 2011;25:549-553.
- Lilly’s Taltz® (ixekizumab) receives U.S. FDA approval for the treatment of pediatric patients with moderate to severe plaque psoriasis. Eli Lilly and Company. March 30, 2020. Accessed September 24, 2021. https://investor.lilly.com/news-releases/news-release-details/lillys-taltzr-ixekizumab-receives-us-fda-approval-treatment-1
- Uber M, Carvalho VO, Abagge KT, et al. Clinical features and nail clippings in 52 children with psoriasis. Pediatr Dermatol. 2018;35:202-207.
- Pourchot D, Bodemer C, Phan A, et al. Nail psoriasis: a systematic evaluation in 313 children with psoriasis. Pediatr Dermatol. 2017;34:58-63.
- Piraccini BM, Triantafyllopoulou I, Prevezas C, et al. Nail psoriasis in children: common or uncommon? results from a 10-year double-center study. Skin Appendage Disord. 2015;1:43-48.
- Baran R. The burden of nail psoriasis: an introduction. Dermatology. 2010;221(suppl 1):1-5.
- Richert B, André J. Nail disorders in children: diagnosis and management. Am J Clin Dermatol. 2011;12:101-112.
- Trüeb RM. Therapies for childhood psoriasis. Curr Probl Dermatol. 2009;38:137-159.
- Haneke E. Nail psoriasis: clinical features, pathogenesis, differential diagnoses, and management. Psoriasis (Auckl). 2017;7:51-63.
- Piraccini BM, Starace M. Nail disorders in infants and children. Curr Opin Pediatr. 2014;26:440-445.
- Duran-McKinster C, Ortiz-Solis D, Granados J, et al. Juvenile psoriatic arthritis with nail psoriasis in the absence of cutaneous lesions. Int J Dermatol. 2000;39:32-35.
- Holzberg M, Ruben BS, Baran R. Psoriasis restricted to the nail in a 7-year-old child. should biologics be an appropriate treatment modality when considering quality of life? J Eur Acad Dermatol Venereol. 2014;28:668-670.
- Lee JY. Severe 20-nail psoriasis successfully treated by low dose methotrexate. Dermatol Online J. 2009;15:8.
- Liao YC, Lee JY. Psoriasis in a 3-month-old infant with Kawasaki disease. Dermatol Online J. 2009;15:10.
- Khoo BP, Giam YC. A pilot study on the role of intralesional triamcinolone acetonide in the treatment of pitted nails in children. Singapore Med J. 2000;41:66-68.
- Piraccini BM, Tosti A, Iorizzo M, et al. Pustular psoriasis of the nails: treatment and long-term follow-up of 46 patients. Br J Dermatol. 2001;144:1000-1005.
- Diluvio L, Campione E, Paternò EJ, et al. Childhood nail psoriasis: a useful treatment with tazarotene 0.05%. Pediatr Dermatol. 2007;24:332-333.
- Teran CG, Teran-Escalera CN, Balderrama C. A severe case of erythrodermic psoriasis associated with advanced nail and joint manifestations: a case report. J Med Case Rep. 2010;4:179.
- Chen P, Li C, Xue R, et al. Efficacy and safety of acitretin monotherapy in children with pustular psoriasis: results from 15 cases and a literature review. J Dermatolog Treat. 2018;29:353-363.
- Wells LE, Evans T, Hilton R, et al. Use of secukinumab in a pediatric patient leads to significant improvement in nail psoriasis and psoriatic arthritis. Pediatr Dermatol. 2019;36:384-385.
- Watabe D, Endoh K, Maeda F, et al. Childhood-onset psoriatic onycho-pachydermo-periostitis treated successfully with infliximab. Eur J Dermatol. 2015;25:506-508.
- Pereira TM, Vieira AP, Fernandes JC, et al. Anti-TNF-alpha therapy in childhood pustular psoriasis. Dermatology. 2006;213:350-352.
- Dini V, Barbanera S, Romanelli M. Efficacy of adalimumab for the treatment of refractory paediatric acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2013;93:588-589.
- Kiszewski AE, De Villa D, Scheibel I, et al. An infant with acrodermatitis continua of Hallopeau: successful treatment with thalidomide and UVB therapy. Pediatr Dermatol. 2009;26:105-106.
- Bordignon M, Zattra E, Albertin C, et al. Successful treatment of a 9-year-old boy affected by acrodermatitis continua of Hallopeau with targeted ultraviolet B narrow-band phototherapy. Photodermatol Photoimmunol Photomed. 2010;26:41-43.
- Fabroni C, Gori A, Troiano M, et al. Infliximab efficacy in nail psoriasis. a retrospective study in 48 patients. J Eur Acad Dermatol Venereol. 2011;25:549-553.
- Lilly’s Taltz® (ixekizumab) receives U.S. FDA approval for the treatment of pediatric patients with moderate to severe plaque psoriasis. Eli Lilly and Company. March 30, 2020. Accessed September 24, 2021. https://investor.lilly.com/news-releases/news-release-details/lillys-taltzr-ixekizumab-receives-us-fda-approval-treatment-1
Practice Points
- No clinical trials assessing the management of pediatric nail psoriasis currently are present in the literature. Limited information on the treatment of pediatric nail psoriasis exists, mostly in the form of small case series and case reports.
- As more agents are approved for on-label use in plaque psoriasis in pediatric patients, gradually more real-life data on their efficacy for nail psoriasis in children are expected to come to light.
Annular Erythema of Infancy With Reactive Helper T Lymphocytes
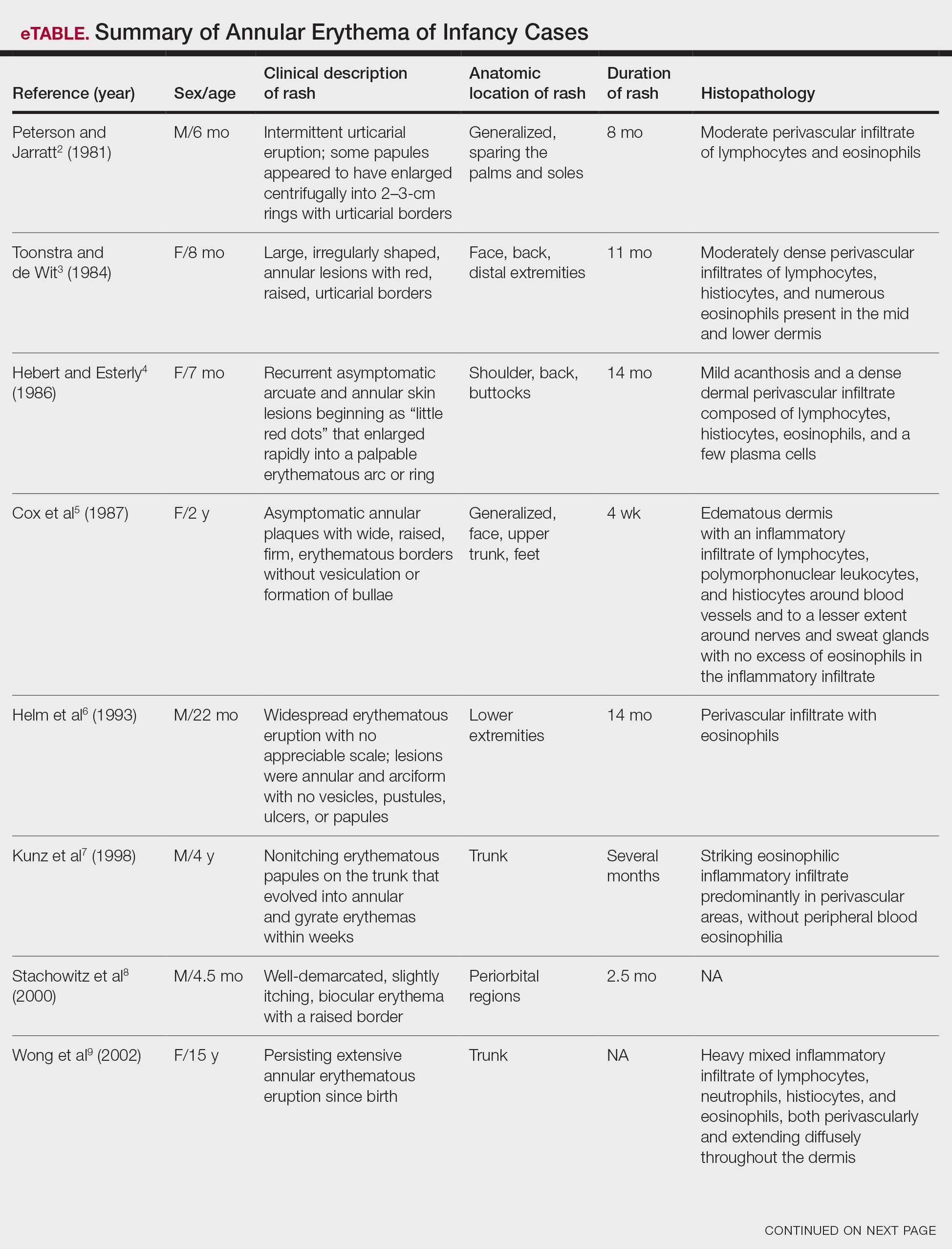
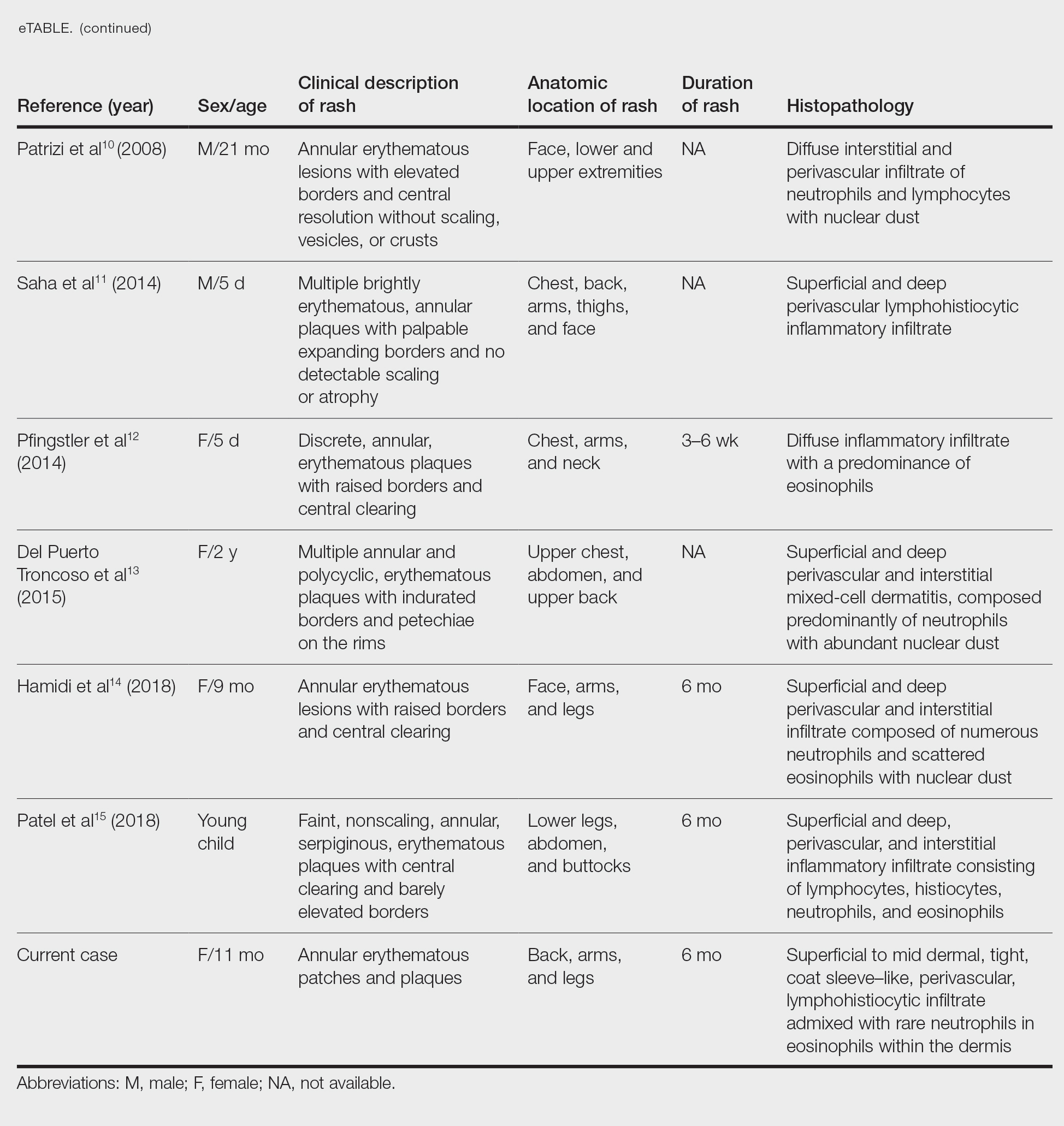
Annular erythemas of infancy (AEIs) are rare benign skin eruptions characterized by annular or circinate, erythematous patches and plaques that arise in patients younger than 1 year.1 Annular erythemas of infancy originally were described by Peterson and Jarratt2 in 1981. Relatively few cases of AEIs have been reported in the literature (eTable).2-15
Case Report
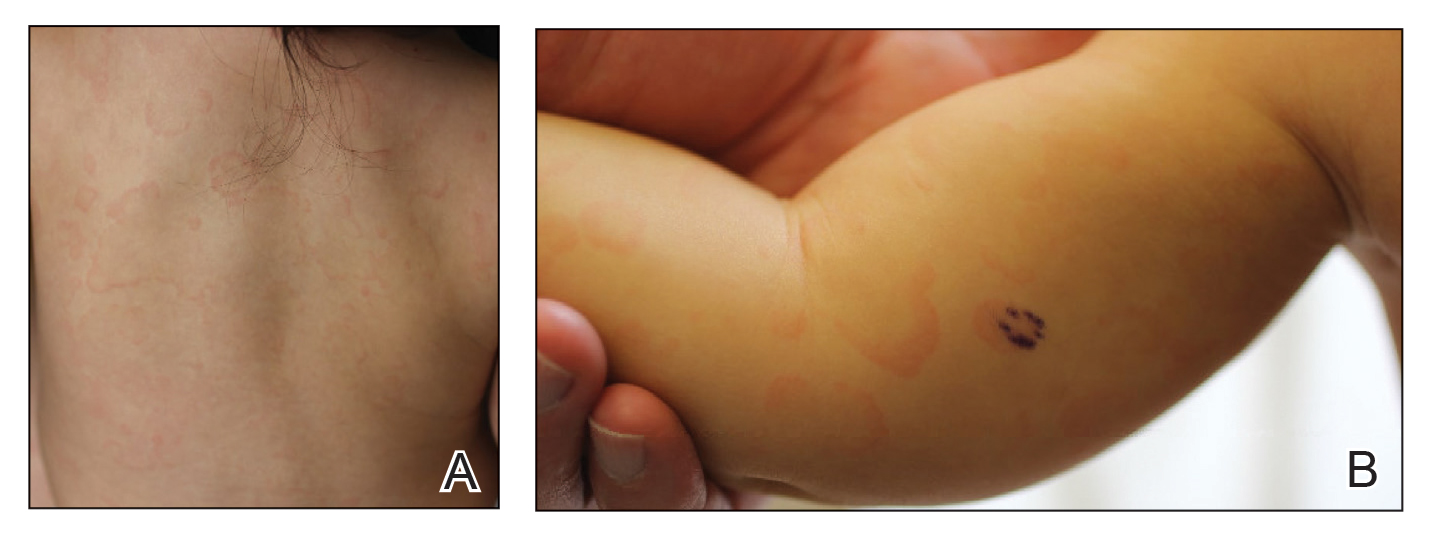
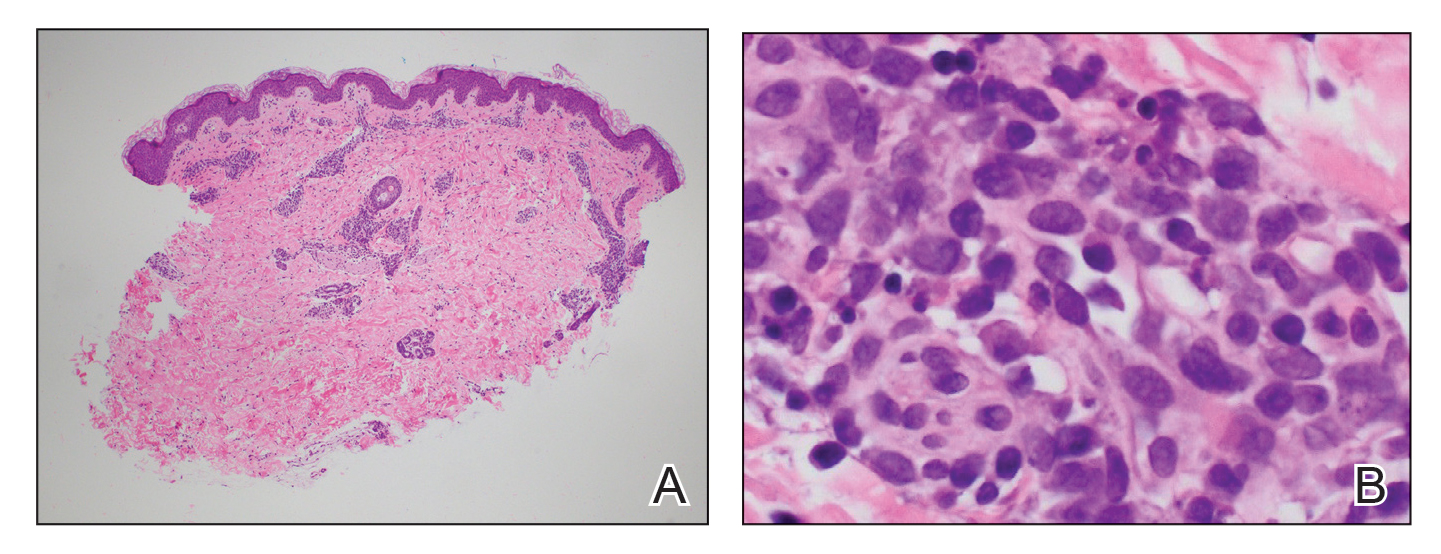
An 11-month-old girl presented to dermatology for a rash characterized by annular erythematous patches and plaques on the back, arms, and legs (Figure 1). Three months prior, the rash was more diffuse, monomorphic, and papular. Based on physical examination, the differential diagnosis included a gyrate erythema such as erythema annulare centrifugum (EAC), neonatal lupus, a viral exanthem, leukemia cutis, and AEI. A skin punch biopsy was performed.
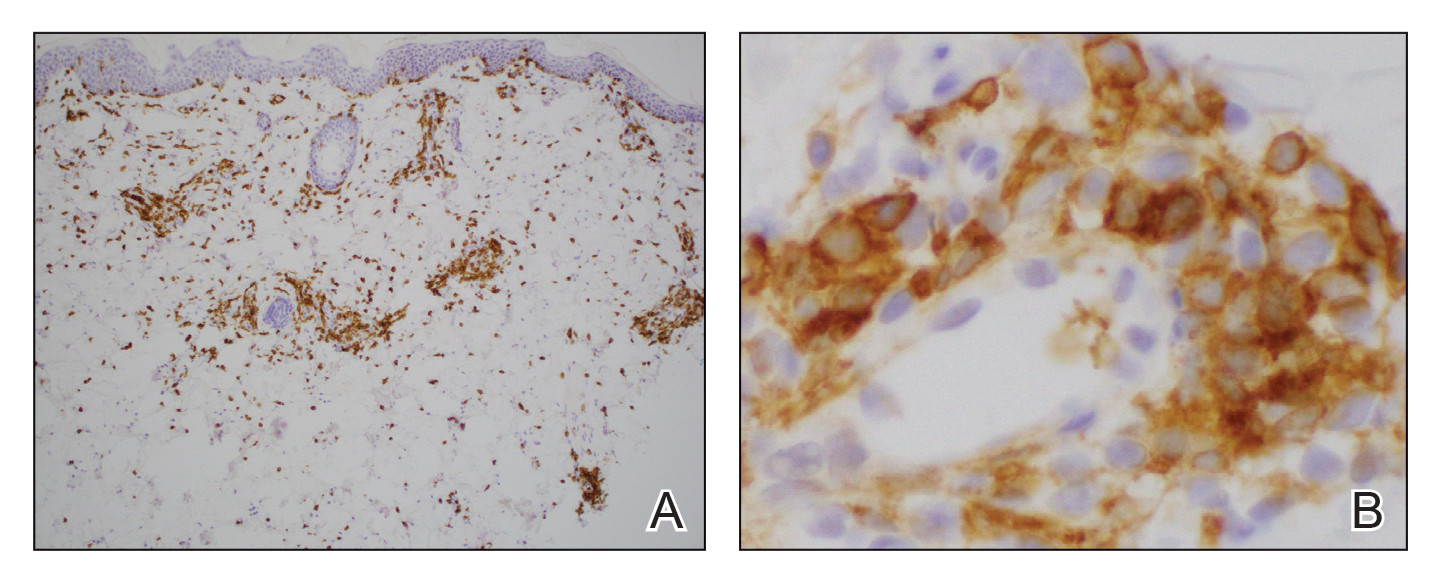
Histologically, the biopsy revealed a superficial to mid dermal, tight, coat sleeve–like, perivascular lymphohistiocytic infiltrate admixed with rare neutrophils in eosinophils within the dermis (Figure 2A). The infiltrate also contained numerous large mononuclear cells with enlarged nuclei, fine loose chromatin, rare nucleoli, and a thin rim of cytoplasm (Figure 2B). There were associated apoptotic bodies with karyorrhectic debris. Immunohistochemistry exhibited enlarged cells that were strong staining with CD3 and CD4, which was consistent with reactive helper T cells (Figure 3). A myeloperoxidase stain highlighted few neutrophils. Stains for terminal deoxynucleotidyl transferase, CD1a, CD117, and CD34 were negative. These findings along with the clinical presentation yielded a diagnosis of AEI with reactive helper T cells.
Comment
Clinical Presentation of AEIs—Annular erythemas of infancy are rare benign skin eruptions that develop in the first few months of life.1,16 Few cases have been reported (eTable). Clinically, AEIs are characterized by annular or circinate, erythematous patches and plaques. They can occur on the face, trunk, and extremities, and they completely resolve by 1 year of age in most cases. One case was reported to persist in a patient from birth until 15 years of age.9 It is thought that AEIs may occur as a hypersensitivity reaction to an unrecognized antigen.
Histopathology—Histologically, AEIs demonstrate a superficial and deep, perivascular, inflammatory infiltrate in the dermis composed of small lymphocytes, some neutrophils, and eosinophils.16 Less common variants of AEI include eosinophilic annular erythema, characterized by a diffuse dermal infiltrate of eosinophils and some lymphocytes, and neutrophilic figurate erythema of infancy, characterized by a dermal infiltrate with neutrophils and leukocytoclasis without vasculitis.1
Our patient’s skin rash was unusual in that the biopsy demonstrated few neutrophils, rare eosinophils, and larger mononuclear cells consistent with reactive helper T lymphocytes. Although these cells may raise concern for an atypical lymphoid infiltrate, recognition of areas with more conventional histopathology of AEIs can facilitate the correct diagnosis.
Differential Diagnosis—The main considerations in the differential diagnosis for AEIs include the following: EAC, familial annular erythema, erythema gyratum atrophicans transiens neonatale, erythema chronicum migrans, urticaria, tinea corporis, neonatal lupus erythematosus, viral exanthems, and leukemia cutis.16
Erythema annulare centrifugum typically begins in middle age and follows a course of 2 or more years.2 It occurs in association with an underlying infection or neoplasm, and it can develop on the trunk and proximal extremities. Morphologically, EAC can present with arcuate or polycyclic lesions with trailing scale. Histologically, a skin biopsy shows a tight, coat sleeve–like, perivascular, lymphohistiocytic infiltrate in the dermis, with variable epidermal spongiosis and parakeratosis.16 Our patient’s biopsy did show a tight perivascular infiltrate, raising suspicion for EAC. However, the eruption occurred in infancy, and she had no clinical evidence of infection or neoplasm.
Familial annular erythemas can arise within a few days after birth and can present on any part of the body, including the tongue.2 Individual lesions can persist for 4 to 5 days and can accompany congenital malformations. Morphologically, they can present as papules that slowly enlarge to form arcuate lesions with central hyperpigmentation. Histologically, there can be a mild, perivascular, lymphocytic infiltrate in the dermis.16 Our patient’s lesions showed no scale or pigmentation and occurred without a family history or associated malformations.
Erythema gyratum atrophicans transiens neonatale also can arise in the first few days of life and can affect the trunk, neck, and lips.16 Morphologically, the skin lesions can present as arcuate erythematous patches (3–20 mm) with raised borders and central atrophy. Histologically, there is epidermal atrophy with a dermal perivascular mononuclear cell infiltrate with edema. Our patient’s clinical presentation was not classic for this condition, and the lesions showed no atrophy.
Erythema chronicum migrans can arise in children, often with a history of an arthropod bite.13 Morphologically, lesions can evolve over weeks to months and rarely are multiple. Erythema chronicum migrans most commonly occurs in the United States in association with Lyme disease from infection with Borrelia burgdorferi. Histologically, erythema chronicum migrans shows a superficial and deep, perivascular lymphocytic infiltrate in the dermis with plasma cells and eosinophils. A silver stain can demonstrate dermal spirochetes. Our patient had no history of an arthropod bite. A Warthin-Starry stain performed on the biopsy was negative for spirochetes, and serologies for Lyme disease were negative.
Urticaria is rare in neonates and can occur on any part of the body.2 Morphologically, the skin lesions can present as arcuate, erythematous, and polycyclic plaques that wax and wane. Histologically, there is dermal edema with a mild, perivascular and interstitial, mixed inflammatory infiltrate.16 Our patient’s biopsy did not reveal notable edema, and the perivascular infiltrate was coat sleeve–like with few neutrophils and eosinophils. The patient did not respond to initial treatment with antihistamines, making urticaria less likely.
Tinea corporis is rare in neonates and can occur on any part of the body.13 Morphologically, it can present as scaly annular lesions that are fixed and more persistent. Histologically, there are fungal hyphae and/or yeast in the stratum corneum with spongiotic dermatitis and parakeratosis. Our patient’s lesions were not scaly, and the biopsy demonstrated minimal spongiosis. A periodic acid–Schiff special stain was negative for fungal microorganisms.
Neonatal lupus erythematosus can arise at birth or during the first few weeks of life.16 Morphologically, the skin lesions occur on the scalp, forehead, or neck in a periorbital or malar distribution. They can present as erythematous, annular, scaly patches and plaques. Transplacental transmission of material autoantibodies has been implicated in the etiology, and a complication is infantile heart block. Histologically, a skin biopsy typically shows interface/lichenoid dermatitis. However, our patient’s biopsy did not demonstrate interface changes, and serologically she was negative for autoantibodies.
Viral exanthems are skin eruptions that accompany underlying viral infections.17 Morphologically, patients can present with an erythematous maculopapular rash, sometimes with vesicular, petechial, and urticarial lesions. Laboratory confirmation is made by virus-specific serologies. Histologically, viral exanthems can show a superficial, perivascular, lymphocytic infiltrate in the dermis, with reactive T cells and epidermal spongiosis. Our patient was afebrile and had no known sick contacts. A cytomegalovirus immunohistochemical study on the biopsy was negative, and an Epstein-Barr encoding region in situ hybridization study was negative.
Leukemia cutis is the infiltration of the skin by leukemic cells, most often in conjunction with systemic leukemia.18 In infants and children, the most common leukemia is B-cell acute lymphoblastic leukemia. Morphologically, the skin lesions are characterized by single or multiple violaceous papules, nodules, and plaques. Histologically, there is a perivascular to interstitial infiltrate of atypical mononuclear cells in the dermis and sometimes subcutis. The leukemic cells demonstrate enlarged nuclei with coarse chromatin and prominent nucleoli. Increased mitotic activity may be seen with karyorrhectic debris. Immunohistochemically, the tumor cells can be positive for myeloperoxidase, CD43, CD68, CD34, and CD117.18 Although our patient’s biopsy demonstrated mononuclear cells with karyorrhexis, the cells did not have striking atypia and were negative for blast markers. A recent complete blood cell count on the patient was normal.
Conclusion
We report an unusual case of AEI with mononuclear cells consistent with helper T cells. One must keep these cells in mind when evaluating a biopsy of AEI, as they are benign and not suggestive of an atypical lymphoid infiltrate or leukemia cutis. This will prevent misdiagnosis and ensure that the patient receives appropriate management.
- Ríos-Martín JJ, Ferrándiz-Pulido L, Moreno-Ramírez D. Approaches to the dermatopathologic diagnosis of figurate lesions [in Spanish]. Actas Dermosifiliogr. 2011;102:316-324. doi:10.1016/j.ad.2010.12.009
- Peterson AO, Jarratt M. Annular erythema of infancy. Arch Dermatol. 1981;117:145-148.
- Toonstra J, de Wit RF. “Persistent” annular erythema of infancy. Arch Dermatol.1984;120:1069-1072.
- Hebert AA, Esterly NB. Annular erythema of infancy. J Am Acad Dermatol. 1986;14:339-343.
- Cox NH, McQueen A, Evans TJ, et al. An annular erythema of infancy. Arch Dermatol. 1987;123:510-513.
- Helm TN, Bass J, Chang LW, et al. Persistent annular erythema of infancy. Pediatr Dermatol. 1993;10:46-48.
- Kunz M, Hamm K, Bröcker EB, et al. Annular erythema in childhood—a new eosinophilic dermatosis [in German]. Hautarzt. 1998;49:131-134.
- Stachowitz S, Abeck D, Schmidt T, et al. Persistent annular erythema of infancy associated with intestinal Candida colonization. Clin Exp Dermatol. 2000;25:404-405.
- Wong L-C, Kakakios A, Rogers M. Congenital annular erythema persisting in a 15-year-old girl. Australas J Dermatol. 2002;43:55-61.
- Patrizi A, Savoia F, Varotti E, et al. Neutrophilic figurate erythema of infancy. Pediatr Dermatol. 2008;25:255-260. doi:10.1111/j.1525-1470.2008.00646.x
- Saha A, Seth J, Mukherjee S, et al. Annular erythema of infancy: a diagnostic challenge. Indian J Paediatr Dermatol. 2014;15:147-149. doi:10.4103/2319-7250.143678
- Pfingstler LF, Miller KP, Pride H. Recurring diffuse annular erythematous plaques in a newborn. JAMA Dermatol. 2014;150:565-566. doi:10.1001/jamadermatol.2013.8059
- Del Puerto Troncoso C, Curi Tuma M, González Bombardiere S, et al. Neutrophilic figurate erythema of infancy associated with juvenile myelomonocytic leukemia. Actas Dermosifiliogr. 2015;106:431-433. doi:10.1016/j.ad.2014.09.013
- Hamidi S, Prose NS, Selim MA. Neutrophilic figurate erythema of infancy: a diagnostic challenge [published online December 26, 2018]. J Cutan Pathol. 2019;46:216-220. doi:10.1111/cup.13394
- Patel N, Goldbach H, Hogeling M. An annular eruption in a young child. JAMA Dermatol. 2018;154:1213-1214. doi:10.1001/jamadermatol.2018.1174
- Palit A, Inamadar AC. Annular, erythematous skin lesions in a neonate. Indian Dermatol Online J. 2012;3:45-47. doi:10.4103/2229-5178.93504
- Keighley CL, Saunderson RB, Kok J, et al. Viral exanthems. Curr Opin Infect Dis. 2015;28:139-150. doi:10.1097/QCO.0000000000000145
- Cronin DMP, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110. doi:10.1309/AJCP6GR8BDEXPKHR
Annular erythemas of infancy (AEIs) are rare benign skin eruptions characterized by annular or circinate, erythematous patches and plaques that arise in patients younger than 1 year.1 Annular erythemas of infancy originally were described by Peterson and Jarratt2 in 1981. Relatively few cases of AEIs have been reported in the literature (eTable).2-15
Case Report
An 11-month-old girl presented to dermatology for a rash characterized by annular erythematous patches and plaques on the back, arms, and legs (Figure 1). Three months prior, the rash was more diffuse, monomorphic, and papular. Based on physical examination, the differential diagnosis included a gyrate erythema such as erythema annulare centrifugum (EAC), neonatal lupus, a viral exanthem, leukemia cutis, and AEI. A skin punch biopsy was performed.
Histologically, the biopsy revealed a superficial to mid dermal, tight, coat sleeve–like, perivascular lymphohistiocytic infiltrate admixed with rare neutrophils in eosinophils within the dermis (Figure 2A). The infiltrate also contained numerous large mononuclear cells with enlarged nuclei, fine loose chromatin, rare nucleoli, and a thin rim of cytoplasm (Figure 2B). There were associated apoptotic bodies with karyorrhectic debris. Immunohistochemistry exhibited enlarged cells that were strong staining with CD3 and CD4, which was consistent with reactive helper T cells (Figure 3). A myeloperoxidase stain highlighted few neutrophils. Stains for terminal deoxynucleotidyl transferase, CD1a, CD117, and CD34 were negative. These findings along with the clinical presentation yielded a diagnosis of AEI with reactive helper T cells.
Comment
Clinical Presentation of AEIs—Annular erythemas of infancy are rare benign skin eruptions that develop in the first few months of life.1,16 Few cases have been reported (eTable). Clinically, AEIs are characterized by annular or circinate, erythematous patches and plaques. They can occur on the face, trunk, and extremities, and they completely resolve by 1 year of age in most cases. One case was reported to persist in a patient from birth until 15 years of age.9 It is thought that AEIs may occur as a hypersensitivity reaction to an unrecognized antigen.
Histopathology—Histologically, AEIs demonstrate a superficial and deep, perivascular, inflammatory infiltrate in the dermis composed of small lymphocytes, some neutrophils, and eosinophils.16 Less common variants of AEI include eosinophilic annular erythema, characterized by a diffuse dermal infiltrate of eosinophils and some lymphocytes, and neutrophilic figurate erythema of infancy, characterized by a dermal infiltrate with neutrophils and leukocytoclasis without vasculitis.1
Our patient’s skin rash was unusual in that the biopsy demonstrated few neutrophils, rare eosinophils, and larger mononuclear cells consistent with reactive helper T lymphocytes. Although these cells may raise concern for an atypical lymphoid infiltrate, recognition of areas with more conventional histopathology of AEIs can facilitate the correct diagnosis.
Differential Diagnosis—The main considerations in the differential diagnosis for AEIs include the following: EAC, familial annular erythema, erythema gyratum atrophicans transiens neonatale, erythema chronicum migrans, urticaria, tinea corporis, neonatal lupus erythematosus, viral exanthems, and leukemia cutis.16
Erythema annulare centrifugum typically begins in middle age and follows a course of 2 or more years.2 It occurs in association with an underlying infection or neoplasm, and it can develop on the trunk and proximal extremities. Morphologically, EAC can present with arcuate or polycyclic lesions with trailing scale. Histologically, a skin biopsy shows a tight, coat sleeve–like, perivascular, lymphohistiocytic infiltrate in the dermis, with variable epidermal spongiosis and parakeratosis.16 Our patient’s biopsy did show a tight perivascular infiltrate, raising suspicion for EAC. However, the eruption occurred in infancy, and she had no clinical evidence of infection or neoplasm.
Familial annular erythemas can arise within a few days after birth and can present on any part of the body, including the tongue.2 Individual lesions can persist for 4 to 5 days and can accompany congenital malformations. Morphologically, they can present as papules that slowly enlarge to form arcuate lesions with central hyperpigmentation. Histologically, there can be a mild, perivascular, lymphocytic infiltrate in the dermis.16 Our patient’s lesions showed no scale or pigmentation and occurred without a family history or associated malformations.
Erythema gyratum atrophicans transiens neonatale also can arise in the first few days of life and can affect the trunk, neck, and lips.16 Morphologically, the skin lesions can present as arcuate erythematous patches (3–20 mm) with raised borders and central atrophy. Histologically, there is epidermal atrophy with a dermal perivascular mononuclear cell infiltrate with edema. Our patient’s clinical presentation was not classic for this condition, and the lesions showed no atrophy.
Erythema chronicum migrans can arise in children, often with a history of an arthropod bite.13 Morphologically, lesions can evolve over weeks to months and rarely are multiple. Erythema chronicum migrans most commonly occurs in the United States in association with Lyme disease from infection with Borrelia burgdorferi. Histologically, erythema chronicum migrans shows a superficial and deep, perivascular lymphocytic infiltrate in the dermis with plasma cells and eosinophils. A silver stain can demonstrate dermal spirochetes. Our patient had no history of an arthropod bite. A Warthin-Starry stain performed on the biopsy was negative for spirochetes, and serologies for Lyme disease were negative.
Urticaria is rare in neonates and can occur on any part of the body.2 Morphologically, the skin lesions can present as arcuate, erythematous, and polycyclic plaques that wax and wane. Histologically, there is dermal edema with a mild, perivascular and interstitial, mixed inflammatory infiltrate.16 Our patient’s biopsy did not reveal notable edema, and the perivascular infiltrate was coat sleeve–like with few neutrophils and eosinophils. The patient did not respond to initial treatment with antihistamines, making urticaria less likely.
Tinea corporis is rare in neonates and can occur on any part of the body.13 Morphologically, it can present as scaly annular lesions that are fixed and more persistent. Histologically, there are fungal hyphae and/or yeast in the stratum corneum with spongiotic dermatitis and parakeratosis. Our patient’s lesions were not scaly, and the biopsy demonstrated minimal spongiosis. A periodic acid–Schiff special stain was negative for fungal microorganisms.
Neonatal lupus erythematosus can arise at birth or during the first few weeks of life.16 Morphologically, the skin lesions occur on the scalp, forehead, or neck in a periorbital or malar distribution. They can present as erythematous, annular, scaly patches and plaques. Transplacental transmission of material autoantibodies has been implicated in the etiology, and a complication is infantile heart block. Histologically, a skin biopsy typically shows interface/lichenoid dermatitis. However, our patient’s biopsy did not demonstrate interface changes, and serologically she was negative for autoantibodies.
Viral exanthems are skin eruptions that accompany underlying viral infections.17 Morphologically, patients can present with an erythematous maculopapular rash, sometimes with vesicular, petechial, and urticarial lesions. Laboratory confirmation is made by virus-specific serologies. Histologically, viral exanthems can show a superficial, perivascular, lymphocytic infiltrate in the dermis, with reactive T cells and epidermal spongiosis. Our patient was afebrile and had no known sick contacts. A cytomegalovirus immunohistochemical study on the biopsy was negative, and an Epstein-Barr encoding region in situ hybridization study was negative.
Leukemia cutis is the infiltration of the skin by leukemic cells, most often in conjunction with systemic leukemia.18 In infants and children, the most common leukemia is B-cell acute lymphoblastic leukemia. Morphologically, the skin lesions are characterized by single or multiple violaceous papules, nodules, and plaques. Histologically, there is a perivascular to interstitial infiltrate of atypical mononuclear cells in the dermis and sometimes subcutis. The leukemic cells demonstrate enlarged nuclei with coarse chromatin and prominent nucleoli. Increased mitotic activity may be seen with karyorrhectic debris. Immunohistochemically, the tumor cells can be positive for myeloperoxidase, CD43, CD68, CD34, and CD117.18 Although our patient’s biopsy demonstrated mononuclear cells with karyorrhexis, the cells did not have striking atypia and were negative for blast markers. A recent complete blood cell count on the patient was normal.
Conclusion
We report an unusual case of AEI with mononuclear cells consistent with helper T cells. One must keep these cells in mind when evaluating a biopsy of AEI, as they are benign and not suggestive of an atypical lymphoid infiltrate or leukemia cutis. This will prevent misdiagnosis and ensure that the patient receives appropriate management.
Annular erythemas of infancy (AEIs) are rare benign skin eruptions characterized by annular or circinate, erythematous patches and plaques that arise in patients younger than 1 year.1 Annular erythemas of infancy originally were described by Peterson and Jarratt2 in 1981. Relatively few cases of AEIs have been reported in the literature (eTable).2-15
Case Report
An 11-month-old girl presented to dermatology for a rash characterized by annular erythematous patches and plaques on the back, arms, and legs (Figure 1). Three months prior, the rash was more diffuse, monomorphic, and papular. Based on physical examination, the differential diagnosis included a gyrate erythema such as erythema annulare centrifugum (EAC), neonatal lupus, a viral exanthem, leukemia cutis, and AEI. A skin punch biopsy was performed.
Histologically, the biopsy revealed a superficial to mid dermal, tight, coat sleeve–like, perivascular lymphohistiocytic infiltrate admixed with rare neutrophils in eosinophils within the dermis (Figure 2A). The infiltrate also contained numerous large mononuclear cells with enlarged nuclei, fine loose chromatin, rare nucleoli, and a thin rim of cytoplasm (Figure 2B). There were associated apoptotic bodies with karyorrhectic debris. Immunohistochemistry exhibited enlarged cells that were strong staining with CD3 and CD4, which was consistent with reactive helper T cells (Figure 3). A myeloperoxidase stain highlighted few neutrophils. Stains for terminal deoxynucleotidyl transferase, CD1a, CD117, and CD34 were negative. These findings along with the clinical presentation yielded a diagnosis of AEI with reactive helper T cells.
Comment
Clinical Presentation of AEIs—Annular erythemas of infancy are rare benign skin eruptions that develop in the first few months of life.1,16 Few cases have been reported (eTable). Clinically, AEIs are characterized by annular or circinate, erythematous patches and plaques. They can occur on the face, trunk, and extremities, and they completely resolve by 1 year of age in most cases. One case was reported to persist in a patient from birth until 15 years of age.9 It is thought that AEIs may occur as a hypersensitivity reaction to an unrecognized antigen.
Histopathology—Histologically, AEIs demonstrate a superficial and deep, perivascular, inflammatory infiltrate in the dermis composed of small lymphocytes, some neutrophils, and eosinophils.16 Less common variants of AEI include eosinophilic annular erythema, characterized by a diffuse dermal infiltrate of eosinophils and some lymphocytes, and neutrophilic figurate erythema of infancy, characterized by a dermal infiltrate with neutrophils and leukocytoclasis without vasculitis.1
Our patient’s skin rash was unusual in that the biopsy demonstrated few neutrophils, rare eosinophils, and larger mononuclear cells consistent with reactive helper T lymphocytes. Although these cells may raise concern for an atypical lymphoid infiltrate, recognition of areas with more conventional histopathology of AEIs can facilitate the correct diagnosis.
Differential Diagnosis—The main considerations in the differential diagnosis for AEIs include the following: EAC, familial annular erythema, erythema gyratum atrophicans transiens neonatale, erythema chronicum migrans, urticaria, tinea corporis, neonatal lupus erythematosus, viral exanthems, and leukemia cutis.16
Erythema annulare centrifugum typically begins in middle age and follows a course of 2 or more years.2 It occurs in association with an underlying infection or neoplasm, and it can develop on the trunk and proximal extremities. Morphologically, EAC can present with arcuate or polycyclic lesions with trailing scale. Histologically, a skin biopsy shows a tight, coat sleeve–like, perivascular, lymphohistiocytic infiltrate in the dermis, with variable epidermal spongiosis and parakeratosis.16 Our patient’s biopsy did show a tight perivascular infiltrate, raising suspicion for EAC. However, the eruption occurred in infancy, and she had no clinical evidence of infection or neoplasm.
Familial annular erythemas can arise within a few days after birth and can present on any part of the body, including the tongue.2 Individual lesions can persist for 4 to 5 days and can accompany congenital malformations. Morphologically, they can present as papules that slowly enlarge to form arcuate lesions with central hyperpigmentation. Histologically, there can be a mild, perivascular, lymphocytic infiltrate in the dermis.16 Our patient’s lesions showed no scale or pigmentation and occurred without a family history or associated malformations.
Erythema gyratum atrophicans transiens neonatale also can arise in the first few days of life and can affect the trunk, neck, and lips.16 Morphologically, the skin lesions can present as arcuate erythematous patches (3–20 mm) with raised borders and central atrophy. Histologically, there is epidermal atrophy with a dermal perivascular mononuclear cell infiltrate with edema. Our patient’s clinical presentation was not classic for this condition, and the lesions showed no atrophy.
Erythema chronicum migrans can arise in children, often with a history of an arthropod bite.13 Morphologically, lesions can evolve over weeks to months and rarely are multiple. Erythema chronicum migrans most commonly occurs in the United States in association with Lyme disease from infection with Borrelia burgdorferi. Histologically, erythema chronicum migrans shows a superficial and deep, perivascular lymphocytic infiltrate in the dermis with plasma cells and eosinophils. A silver stain can demonstrate dermal spirochetes. Our patient had no history of an arthropod bite. A Warthin-Starry stain performed on the biopsy was negative for spirochetes, and serologies for Lyme disease were negative.
Urticaria is rare in neonates and can occur on any part of the body.2 Morphologically, the skin lesions can present as arcuate, erythematous, and polycyclic plaques that wax and wane. Histologically, there is dermal edema with a mild, perivascular and interstitial, mixed inflammatory infiltrate.16 Our patient’s biopsy did not reveal notable edema, and the perivascular infiltrate was coat sleeve–like with few neutrophils and eosinophils. The patient did not respond to initial treatment with antihistamines, making urticaria less likely.
Tinea corporis is rare in neonates and can occur on any part of the body.13 Morphologically, it can present as scaly annular lesions that are fixed and more persistent. Histologically, there are fungal hyphae and/or yeast in the stratum corneum with spongiotic dermatitis and parakeratosis. Our patient’s lesions were not scaly, and the biopsy demonstrated minimal spongiosis. A periodic acid–Schiff special stain was negative for fungal microorganisms.
Neonatal lupus erythematosus can arise at birth or during the first few weeks of life.16 Morphologically, the skin lesions occur on the scalp, forehead, or neck in a periorbital or malar distribution. They can present as erythematous, annular, scaly patches and plaques. Transplacental transmission of material autoantibodies has been implicated in the etiology, and a complication is infantile heart block. Histologically, a skin biopsy typically shows interface/lichenoid dermatitis. However, our patient’s biopsy did not demonstrate interface changes, and serologically she was negative for autoantibodies.
Viral exanthems are skin eruptions that accompany underlying viral infections.17 Morphologically, patients can present with an erythematous maculopapular rash, sometimes with vesicular, petechial, and urticarial lesions. Laboratory confirmation is made by virus-specific serologies. Histologically, viral exanthems can show a superficial, perivascular, lymphocytic infiltrate in the dermis, with reactive T cells and epidermal spongiosis. Our patient was afebrile and had no known sick contacts. A cytomegalovirus immunohistochemical study on the biopsy was negative, and an Epstein-Barr encoding region in situ hybridization study was negative.
Leukemia cutis is the infiltration of the skin by leukemic cells, most often in conjunction with systemic leukemia.18 In infants and children, the most common leukemia is B-cell acute lymphoblastic leukemia. Morphologically, the skin lesions are characterized by single or multiple violaceous papules, nodules, and plaques. Histologically, there is a perivascular to interstitial infiltrate of atypical mononuclear cells in the dermis and sometimes subcutis. The leukemic cells demonstrate enlarged nuclei with coarse chromatin and prominent nucleoli. Increased mitotic activity may be seen with karyorrhectic debris. Immunohistochemically, the tumor cells can be positive for myeloperoxidase, CD43, CD68, CD34, and CD117.18 Although our patient’s biopsy demonstrated mononuclear cells with karyorrhexis, the cells did not have striking atypia and were negative for blast markers. A recent complete blood cell count on the patient was normal.
Conclusion
We report an unusual case of AEI with mononuclear cells consistent with helper T cells. One must keep these cells in mind when evaluating a biopsy of AEI, as they are benign and not suggestive of an atypical lymphoid infiltrate or leukemia cutis. This will prevent misdiagnosis and ensure that the patient receives appropriate management.
- Ríos-Martín JJ, Ferrándiz-Pulido L, Moreno-Ramírez D. Approaches to the dermatopathologic diagnosis of figurate lesions [in Spanish]. Actas Dermosifiliogr. 2011;102:316-324. doi:10.1016/j.ad.2010.12.009
- Peterson AO, Jarratt M. Annular erythema of infancy. Arch Dermatol. 1981;117:145-148.
- Toonstra J, de Wit RF. “Persistent” annular erythema of infancy. Arch Dermatol.1984;120:1069-1072.
- Hebert AA, Esterly NB. Annular erythema of infancy. J Am Acad Dermatol. 1986;14:339-343.
- Cox NH, McQueen A, Evans TJ, et al. An annular erythema of infancy. Arch Dermatol. 1987;123:510-513.
- Helm TN, Bass J, Chang LW, et al. Persistent annular erythema of infancy. Pediatr Dermatol. 1993;10:46-48.
- Kunz M, Hamm K, Bröcker EB, et al. Annular erythema in childhood—a new eosinophilic dermatosis [in German]. Hautarzt. 1998;49:131-134.
- Stachowitz S, Abeck D, Schmidt T, et al. Persistent annular erythema of infancy associated with intestinal Candida colonization. Clin Exp Dermatol. 2000;25:404-405.
- Wong L-C, Kakakios A, Rogers M. Congenital annular erythema persisting in a 15-year-old girl. Australas J Dermatol. 2002;43:55-61.
- Patrizi A, Savoia F, Varotti E, et al. Neutrophilic figurate erythema of infancy. Pediatr Dermatol. 2008;25:255-260. doi:10.1111/j.1525-1470.2008.00646.x
- Saha A, Seth J, Mukherjee S, et al. Annular erythema of infancy: a diagnostic challenge. Indian J Paediatr Dermatol. 2014;15:147-149. doi:10.4103/2319-7250.143678
- Pfingstler LF, Miller KP, Pride H. Recurring diffuse annular erythematous plaques in a newborn. JAMA Dermatol. 2014;150:565-566. doi:10.1001/jamadermatol.2013.8059
- Del Puerto Troncoso C, Curi Tuma M, González Bombardiere S, et al. Neutrophilic figurate erythema of infancy associated with juvenile myelomonocytic leukemia. Actas Dermosifiliogr. 2015;106:431-433. doi:10.1016/j.ad.2014.09.013
- Hamidi S, Prose NS, Selim MA. Neutrophilic figurate erythema of infancy: a diagnostic challenge [published online December 26, 2018]. J Cutan Pathol. 2019;46:216-220. doi:10.1111/cup.13394
- Patel N, Goldbach H, Hogeling M. An annular eruption in a young child. JAMA Dermatol. 2018;154:1213-1214. doi:10.1001/jamadermatol.2018.1174
- Palit A, Inamadar AC. Annular, erythematous skin lesions in a neonate. Indian Dermatol Online J. 2012;3:45-47. doi:10.4103/2229-5178.93504
- Keighley CL, Saunderson RB, Kok J, et al. Viral exanthems. Curr Opin Infect Dis. 2015;28:139-150. doi:10.1097/QCO.0000000000000145
- Cronin DMP, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110. doi:10.1309/AJCP6GR8BDEXPKHR
- Ríos-Martín JJ, Ferrándiz-Pulido L, Moreno-Ramírez D. Approaches to the dermatopathologic diagnosis of figurate lesions [in Spanish]. Actas Dermosifiliogr. 2011;102:316-324. doi:10.1016/j.ad.2010.12.009
- Peterson AO, Jarratt M. Annular erythema of infancy. Arch Dermatol. 1981;117:145-148.
- Toonstra J, de Wit RF. “Persistent” annular erythema of infancy. Arch Dermatol.1984;120:1069-1072.
- Hebert AA, Esterly NB. Annular erythema of infancy. J Am Acad Dermatol. 1986;14:339-343.
- Cox NH, McQueen A, Evans TJ, et al. An annular erythema of infancy. Arch Dermatol. 1987;123:510-513.
- Helm TN, Bass J, Chang LW, et al. Persistent annular erythema of infancy. Pediatr Dermatol. 1993;10:46-48.
- Kunz M, Hamm K, Bröcker EB, et al. Annular erythema in childhood—a new eosinophilic dermatosis [in German]. Hautarzt. 1998;49:131-134.
- Stachowitz S, Abeck D, Schmidt T, et al. Persistent annular erythema of infancy associated with intestinal Candida colonization. Clin Exp Dermatol. 2000;25:404-405.
- Wong L-C, Kakakios A, Rogers M. Congenital annular erythema persisting in a 15-year-old girl. Australas J Dermatol. 2002;43:55-61.
- Patrizi A, Savoia F, Varotti E, et al. Neutrophilic figurate erythema of infancy. Pediatr Dermatol. 2008;25:255-260. doi:10.1111/j.1525-1470.2008.00646.x
- Saha A, Seth J, Mukherjee S, et al. Annular erythema of infancy: a diagnostic challenge. Indian J Paediatr Dermatol. 2014;15:147-149. doi:10.4103/2319-7250.143678
- Pfingstler LF, Miller KP, Pride H. Recurring diffuse annular erythematous plaques in a newborn. JAMA Dermatol. 2014;150:565-566. doi:10.1001/jamadermatol.2013.8059
- Del Puerto Troncoso C, Curi Tuma M, González Bombardiere S, et al. Neutrophilic figurate erythema of infancy associated with juvenile myelomonocytic leukemia. Actas Dermosifiliogr. 2015;106:431-433. doi:10.1016/j.ad.2014.09.013
- Hamidi S, Prose NS, Selim MA. Neutrophilic figurate erythema of infancy: a diagnostic challenge [published online December 26, 2018]. J Cutan Pathol. 2019;46:216-220. doi:10.1111/cup.13394
- Patel N, Goldbach H, Hogeling M. An annular eruption in a young child. JAMA Dermatol. 2018;154:1213-1214. doi:10.1001/jamadermatol.2018.1174
- Palit A, Inamadar AC. Annular, erythematous skin lesions in a neonate. Indian Dermatol Online J. 2012;3:45-47. doi:10.4103/2229-5178.93504
- Keighley CL, Saunderson RB, Kok J, et al. Viral exanthems. Curr Opin Infect Dis. 2015;28:139-150. doi:10.1097/QCO.0000000000000145
- Cronin DMP, George TI, Sundram UN. An updated approach to the diagnosis of myeloid leukemia cutis. Am J Clin Pathol. 2009;132:101-110. doi:10.1309/AJCP6GR8BDEXPKHR
Practice Points
- Annular erythemas of infancy (AEIs) are rare benign skin eruptions characterized by persistent, annular, urticarial, nonpruritic patches and plaques that develop in patients younger than 1 year.
- Although AEIs are benign, lesions with uncommon histologic features such as large mononuclear cells consistent with reactive helper T lymphocytes may pose diagnostic challenges.
Pediatric Subungual Exostosis
Exostosis is a type of benign bone tumor in which trabecular (spongy) bone overgrows its normal border in a nodular pattern. 1,2 Histologically, it usually is surrounded by a fibrocartilaginous cap. 3 It is most commonly found on the lateral or medial aspect of the foot and is thought to be caused by trauma, either physical pressure or infection. 4 When this lesion is found under the nail bed, it is termed subungual exostosis ( Dupuytren exostosis ) . 3 Sequelae of a subungual exostosis include nail dystrophy and lifting of the nail away from the toe, in addition to infection and possible loss of the toenail (onycholysis). There are only 2 genetic conditions related to exostosis: hereditary multiple exostosis and multiple exostoses-mental retardation syndrome.
An exostosis may appear to be a wart on first inspection. It may present similar to osteochondromas, and the only way to get a true diagnosis is by biopsy of the lesion. The treatment for an exostosis is surgery. The surgeon must remove the lesion at the base of the bone from which it grows to prevent recurrence of the lesion.5
Because exostosis may cause nail bed disruption, the differential diagnosis may include nail deformities, such as traumatic onycholysis, onychogryphosis, verrucae, subungual infection, or nail trauma.6,7
Case Report
A 7-year-old boy presented with changes of the right great toenail over the last 4 months. The patient noted that the affected nail was discolored, dystrophic, painful, and thickened. He did not recall prior trauma to the affected nail, and his mother stated that the lesion was growing and becoming more painful with a throbbing sensation at times. He described the pain as stabbing, which was exacerbated while walking and playing sports. Neither the patient nor his family had ever had any similar condition. He was not taking any medications, only a daily multivitamin. He had a history of eczematous dermatitis and keratosis pilaris without any other medical illnesses. He had a family history of psoriasis; however, no prior instances of exostosis had been reported. He had no medication allergies.
A full-body cutaneous and nail examination showed a well-developed, well-nourished boy who was in no acute distress. A firm, subungual, pink, pearly,hyperkeratotic nodule was appreciated on the right great toe (Figure 1). The lesion was tender to palpation. The rest of the examination and review of systems were normal.
From the clinical findings, a differential diagnosis of glomus tumor, hemangioma, and infection was considered. Periodic acid–Schiff stain was negative, which ruled out fungal infection. Nail avulsion and a shave biopsy were performed under general anesthesia. There was an exostosis arising from the dorsal aspect of the great toe measuring approximately 5 mm in width at the base and approximately 1 mm in height, which endorsed a diagnosis of distal phalanx subungual exostosis. A postsurgery radiograph (Figure 2) showed residual bone below the level of shave removal at the nail bed.
Comment
Exostosis is most commonly found on the lateral or medial aspect of the hallux (great toe) in patients younger than 18 years.8 Diagnosis often is obvious, even without a radiograph or biopsy, because the exostosis comes out from under the tip of the nail. Our case was interesting because the patient was a child, and the exostosis did not lift the nail or extrude from the distal tip of the nail bed. Evidence suggests that a greater-than-expected genetic influence contributes to an exostosis, though further investigation is needed to determine all of the causes and risk factors for subungual bony exostosis. Timely diagnosis and treatment are essential to the prevention of sequelae of the disease, such as toe infection or chronic pain.
- de Palma L, Gigante A, Specchia N. Subungual exostosis of the foot. Foot Ankle Int. 1996;17:758-763. doi:10.1177/107110079601701208
- Multhopp-Stephens H, Walling AK. Subungual (Dupuytren’s) exostosis. J Pediatr Orthop. 1995;15:582-584. doi:10.1097/01241398-199509000-00006
- Davis DA, Cohen PR. Subungual exostosis: case report and review of the literature. Pediatr Dermatol. 1996;13:212-218.
- Guarneri C, Guarneri F, Risitano G, et al. Solitary asymptomatic nodule of the great toe. Int J Dermatol. 2005;44:245-247.
- Letts M, Davidson D, Nizalik E. Subungual exostosis: diagnosis and treatment in children. J Trauma. 1998;44:346-349.
- Hoy NY, Leung AKC, Metelitsa AI, et al. New concepts in median nail dystrophy, onychomycosis, and hand, foot, and mouth disease nail pathology. ISRN Dermatol. 2012;2012:680163.
- Rich P, Scher RK. Examination of the nail and work-up of nail conditions. In: Rich P, Scher RK, eds. An Atlas of Diseases of the Nail. Parthenon Publishing; 2003.
- DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi:10.1007/s11999-013-3345-4
Exostosis is a type of benign bone tumor in which trabecular (spongy) bone overgrows its normal border in a nodular pattern. 1,2 Histologically, it usually is surrounded by a fibrocartilaginous cap. 3 It is most commonly found on the lateral or medial aspect of the foot and is thought to be caused by trauma, either physical pressure or infection. 4 When this lesion is found under the nail bed, it is termed subungual exostosis ( Dupuytren exostosis ) . 3 Sequelae of a subungual exostosis include nail dystrophy and lifting of the nail away from the toe, in addition to infection and possible loss of the toenail (onycholysis). There are only 2 genetic conditions related to exostosis: hereditary multiple exostosis and multiple exostoses-mental retardation syndrome.
An exostosis may appear to be a wart on first inspection. It may present similar to osteochondromas, and the only way to get a true diagnosis is by biopsy of the lesion. The treatment for an exostosis is surgery. The surgeon must remove the lesion at the base of the bone from which it grows to prevent recurrence of the lesion.5
Because exostosis may cause nail bed disruption, the differential diagnosis may include nail deformities, such as traumatic onycholysis, onychogryphosis, verrucae, subungual infection, or nail trauma.6,7
Case Report
A 7-year-old boy presented with changes of the right great toenail over the last 4 months. The patient noted that the affected nail was discolored, dystrophic, painful, and thickened. He did not recall prior trauma to the affected nail, and his mother stated that the lesion was growing and becoming more painful with a throbbing sensation at times. He described the pain as stabbing, which was exacerbated while walking and playing sports. Neither the patient nor his family had ever had any similar condition. He was not taking any medications, only a daily multivitamin. He had a history of eczematous dermatitis and keratosis pilaris without any other medical illnesses. He had a family history of psoriasis; however, no prior instances of exostosis had been reported. He had no medication allergies.
A full-body cutaneous and nail examination showed a well-developed, well-nourished boy who was in no acute distress. A firm, subungual, pink, pearly,hyperkeratotic nodule was appreciated on the right great toe (Figure 1). The lesion was tender to palpation. The rest of the examination and review of systems were normal.
From the clinical findings, a differential diagnosis of glomus tumor, hemangioma, and infection was considered. Periodic acid–Schiff stain was negative, which ruled out fungal infection. Nail avulsion and a shave biopsy were performed under general anesthesia. There was an exostosis arising from the dorsal aspect of the great toe measuring approximately 5 mm in width at the base and approximately 1 mm in height, which endorsed a diagnosis of distal phalanx subungual exostosis. A postsurgery radiograph (Figure 2) showed residual bone below the level of shave removal at the nail bed.
Comment
Exostosis is most commonly found on the lateral or medial aspect of the hallux (great toe) in patients younger than 18 years.8 Diagnosis often is obvious, even without a radiograph or biopsy, because the exostosis comes out from under the tip of the nail. Our case was interesting because the patient was a child, and the exostosis did not lift the nail or extrude from the distal tip of the nail bed. Evidence suggests that a greater-than-expected genetic influence contributes to an exostosis, though further investigation is needed to determine all of the causes and risk factors for subungual bony exostosis. Timely diagnosis and treatment are essential to the prevention of sequelae of the disease, such as toe infection or chronic pain.
Exostosis is a type of benign bone tumor in which trabecular (spongy) bone overgrows its normal border in a nodular pattern. 1,2 Histologically, it usually is surrounded by a fibrocartilaginous cap. 3 It is most commonly found on the lateral or medial aspect of the foot and is thought to be caused by trauma, either physical pressure or infection. 4 When this lesion is found under the nail bed, it is termed subungual exostosis ( Dupuytren exostosis ) . 3 Sequelae of a subungual exostosis include nail dystrophy and lifting of the nail away from the toe, in addition to infection and possible loss of the toenail (onycholysis). There are only 2 genetic conditions related to exostosis: hereditary multiple exostosis and multiple exostoses-mental retardation syndrome.
An exostosis may appear to be a wart on first inspection. It may present similar to osteochondromas, and the only way to get a true diagnosis is by biopsy of the lesion. The treatment for an exostosis is surgery. The surgeon must remove the lesion at the base of the bone from which it grows to prevent recurrence of the lesion.5
Because exostosis may cause nail bed disruption, the differential diagnosis may include nail deformities, such as traumatic onycholysis, onychogryphosis, verrucae, subungual infection, or nail trauma.6,7
Case Report
A 7-year-old boy presented with changes of the right great toenail over the last 4 months. The patient noted that the affected nail was discolored, dystrophic, painful, and thickened. He did not recall prior trauma to the affected nail, and his mother stated that the lesion was growing and becoming more painful with a throbbing sensation at times. He described the pain as stabbing, which was exacerbated while walking and playing sports. Neither the patient nor his family had ever had any similar condition. He was not taking any medications, only a daily multivitamin. He had a history of eczematous dermatitis and keratosis pilaris without any other medical illnesses. He had a family history of psoriasis; however, no prior instances of exostosis had been reported. He had no medication allergies.
A full-body cutaneous and nail examination showed a well-developed, well-nourished boy who was in no acute distress. A firm, subungual, pink, pearly,hyperkeratotic nodule was appreciated on the right great toe (Figure 1). The lesion was tender to palpation. The rest of the examination and review of systems were normal.
From the clinical findings, a differential diagnosis of glomus tumor, hemangioma, and infection was considered. Periodic acid–Schiff stain was negative, which ruled out fungal infection. Nail avulsion and a shave biopsy were performed under general anesthesia. There was an exostosis arising from the dorsal aspect of the great toe measuring approximately 5 mm in width at the base and approximately 1 mm in height, which endorsed a diagnosis of distal phalanx subungual exostosis. A postsurgery radiograph (Figure 2) showed residual bone below the level of shave removal at the nail bed.
Comment
Exostosis is most commonly found on the lateral or medial aspect of the hallux (great toe) in patients younger than 18 years.8 Diagnosis often is obvious, even without a radiograph or biopsy, because the exostosis comes out from under the tip of the nail. Our case was interesting because the patient was a child, and the exostosis did not lift the nail or extrude from the distal tip of the nail bed. Evidence suggests that a greater-than-expected genetic influence contributes to an exostosis, though further investigation is needed to determine all of the causes and risk factors for subungual bony exostosis. Timely diagnosis and treatment are essential to the prevention of sequelae of the disease, such as toe infection or chronic pain.
- de Palma L, Gigante A, Specchia N. Subungual exostosis of the foot. Foot Ankle Int. 1996;17:758-763. doi:10.1177/107110079601701208
- Multhopp-Stephens H, Walling AK. Subungual (Dupuytren’s) exostosis. J Pediatr Orthop. 1995;15:582-584. doi:10.1097/01241398-199509000-00006
- Davis DA, Cohen PR. Subungual exostosis: case report and review of the literature. Pediatr Dermatol. 1996;13:212-218.
- Guarneri C, Guarneri F, Risitano G, et al. Solitary asymptomatic nodule of the great toe. Int J Dermatol. 2005;44:245-247.
- Letts M, Davidson D, Nizalik E. Subungual exostosis: diagnosis and treatment in children. J Trauma. 1998;44:346-349.
- Hoy NY, Leung AKC, Metelitsa AI, et al. New concepts in median nail dystrophy, onychomycosis, and hand, foot, and mouth disease nail pathology. ISRN Dermatol. 2012;2012:680163.
- Rich P, Scher RK. Examination of the nail and work-up of nail conditions. In: Rich P, Scher RK, eds. An Atlas of Diseases of the Nail. Parthenon Publishing; 2003.
- DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi:10.1007/s11999-013-3345-4
- de Palma L, Gigante A, Specchia N. Subungual exostosis of the foot. Foot Ankle Int. 1996;17:758-763. doi:10.1177/107110079601701208
- Multhopp-Stephens H, Walling AK. Subungual (Dupuytren’s) exostosis. J Pediatr Orthop. 1995;15:582-584. doi:10.1097/01241398-199509000-00006
- Davis DA, Cohen PR. Subungual exostosis: case report and review of the literature. Pediatr Dermatol. 1996;13:212-218.
- Guarneri C, Guarneri F, Risitano G, et al. Solitary asymptomatic nodule of the great toe. Int J Dermatol. 2005;44:245-247.
- Letts M, Davidson D, Nizalik E. Subungual exostosis: diagnosis and treatment in children. J Trauma. 1998;44:346-349.
- Hoy NY, Leung AKC, Metelitsa AI, et al. New concepts in median nail dystrophy, onychomycosis, and hand, foot, and mouth disease nail pathology. ISRN Dermatol. 2012;2012:680163.
- Rich P, Scher RK. Examination of the nail and work-up of nail conditions. In: Rich P, Scher RK, eds. An Atlas of Diseases of the Nail. Parthenon Publishing; 2003.
- DaCambra MP, Gupta SK, Ferri-de-Barros F. Subungual exostosis of the toes: a systematic review. Clin Orthop Relat Res. 2014;472:1251-1259. doi:10.1007/s11999-013-3345-4
Practice Points
- Nail dystrophy can have a variety of causes, most commonly trauma, onychomycosis, verrucae, or subungual exostosis.
- Exostosis is a benign osteochondral tumor commonly found on the lateral or medial aspect of the hallux (great toe) in pediatric and young adult patients.
- A radiograph can be used as a preliminary tool for diagnosis, but subungual exostosis must be confirmed by biopsy or tissue histology at the time of excision.
Enlarging purple plaque on leg
Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318
Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Clinical and dermoscopic features were consistent with a sporadic angiokeratoma, a benign ectasia of vessels associated with keratinization. Diagnosis was confirmed with shave biopsy.
Sporadic angiokeratomas are common and increase with age. They may occur anywhere on the skin, including mucous membranes. Dermoscopy of an angiokeratoma will reveal vascular lacunae—small pools of red or near black blood—as well as keratin scale, which appears as a white veil or shiny white lines.1
Other subtypes of angiokeratomas may be more widespread, including angiokeratoma of Fordyce, which manifests on the vulva and scrotum (usually in adulthood), or angiokeratoma circumscriptum, which occurs congenitally. There are some rare syndromic conditions associated with widespread angiokeratomas, such as Fabry disease.
The differential diagnosis for this solitary, vascular-appearing papule or plaque includes cherry angioma, pyogenic granuloma, and melanoma. Over time, the keratinization may increase, and lesions may look like a wart. A punch or shave biopsy will distinguish an angiokeratoma from other types of lesions. When a lesion is small enough, it may also be curative.
Angiokeratomas do not require treatment. However, because they may occur in cosmetically sensitive areas or bleed when traumatized, remedies are often sought. Excision, cryotherapy, electrocautery, and vascular laser are all possible treatments.
In this case, the patient was treated with curettage and light electrocautery, which left a small scar.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318
1. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318-325. doi: 10.1001/archderm.143.3.318
Babies are dying of syphilis. It’s 100% preventable.
This story was originally published on ProPublica and was co-published with NPR.
When Mai Yang is looking for a patient, she travels light. She dresses deliberately — not too formal, so she won’t be mistaken for a police officer; not too casual, so people will look past her tiny 4-foot-10 stature and youthful face and trust her with sensitive health information. Always, she wears closed-toed shoes, “just in case I need to run.”
Yang carries a stack of cards issued by the Centers for Disease Control and Prevention that show what happens when the Treponema pallidum bacteria invades a patient’s body. There’s a photo of an angry red sore on a penis. There’s one of a tongue, marred by mucus-lined lesions. And there’s one of a newborn baby, its belly, torso and thighs dotted in a rash, its mouth open, as if caught midcry.
It was because of the prospect of one such baby that Yang found herself walking through a homeless encampment on a blazing July day in Huron, Calif., an hour’s drive southwest of her office at the Fresno County Department of Public Health. She was looking for a pregnant woman named Angelica, whose visit to a community clinic had triggered a report to the health department’s sexually transmitted disease program. Angelica had tested positive for syphilis. If she was not treated, her baby could end up like the one in the picture or worse — there was a 40% chance the baby would die.
Ms. Yang knew, though, that if she helped Angelica get treated with three weekly shots of penicillin at least 30 days before she gave birth, it was likely that the infection would be wiped out and her baby would be born without any symptoms at all. Every case of congenital syphilis, when a baby is born with the disease, is avoidable. Each is considered a “sentinel event,” a warning that the public health system is failing.
The alarms are now clamoring. In the United States, more than 129,800 syphilis cases were recorded in 2019, double the case count of five years prior. In the same time period, cases of congenital syphilis quadrupled: 1,870 babies were born with the disease; 128 died. Case counts from 2020 are still being finalized, but the CDC has said that reported cases of congenital syphilis have already exceeded the prior year. Black, Hispanic, and Native American babies are disproportionately at risk.
There was a time, not too long ago, when CDC officials thought they could eliminate the centuries-old scourge from the United States, for adults and babies. But the effort lost steam and cases soon crept up again. Syphilis is not an outlier. The United States goes through what former CDC director Dr. Tom Frieden calls “a deadly cycle of panic and neglect” in which emergencies propel officials to scramble and throw money at a problem — whether that’s Ebola, Zika, or COVID-19. Then, as fear ebbs, so does the attention and motivation to finish the task.
The last fraction of cases can be the hardest to solve, whether that’s eradicating a bug or getting vaccines into arms, yet too often, that’s exactly when political attention gets diverted to the next alarm. The result: The hardest to reach and most vulnerable populations are the ones left suffering, after everyone else looks away.
Ms. Yang first received Angelica’s lab report on June 17. The address listed was a P.O. box, and the phone number belonged to her sister, who said Angelica was living in Huron. That was a piece of luck: Huron is tiny; the city spans just 1.6 square miles. On her first visit, a worker at the Alamo Motel said she knew Angelica and directed Ms. Yang to a nearby homeless encampment. Angelica wasn’t there, so Ms. Yang returned a second time, bringing one of the health department nurses who could serve as an interpreter.
They made their way to the barren patch of land behind Huron Valley Foods, the local grocery store, where people took shelter in makeshift lean-tos composed of cardboard boxes, scrap wood ,and scavenged furniture, draped with sheets that served as ceilings and curtains. Yang stopped outside one of the structures, calling a greeting.
“Hi, I’m from the health department, I’m looking for Angelica.”
The nurse echoed her in Spanish.
Angelica emerged, squinting in the sunlight. Ms. Yang couldn’t tell if she was visibly pregnant yet, as her body was obscured by an oversized shirt. The two women were about the same age: Ms. Yang 26 and Angelica 27. Ms. Yang led her away from the tent, so they could speak privately. Angelica seemed reticent, surprised by the sudden appearance of the two health officers. “You’re not in trouble,” Ms. Yang said, before revealing the results of her blood test.
Angelica had never heard of syphilis.
“Have you been to prenatal care?”
Angelica shook her head. The local clinic had referred her to an obstetrician in Hanford, a 30-minute drive away. She had no car. She also mentioned that she didn’t intend to raise her baby; her two oldest children lived with her mother, and this one likely would, too.
Ms. Yang pulled out the CDC cards, showing them to Angelica and asking if she had experienced any of the symptoms illustrated. No, Angelica said, her lips pursed with disgust.
“Right now you still feel healthy, but this bacteria is still in your body,” Ms. Yang pressed. “You need to get the infection treated to prevent further health complications to yourself and your baby.”
The community clinic was just across the street. “Can we walk you over to the clinic and make sure you get seen so we can get this taken care of?”
Angelica demurred. She said she hadn’t showered for a week and wanted to wash up first. She said she’d go later.
Ms. Yang tried once more to extract a promise: “What time do you think you’ll go?”
“Today, for sure.”
Syphilis is called The Great Imitator: It can look like any number of diseases. In its first stage, the only evidence of infection is a painless sore at the bacteria’s point of entry. Weeks later, as the bacteria multiplies, skin rashes bloom on the palms of the hands and bottoms of the feet. Other traits of this stage include fever, headaches, muscle aches, sore throat, and fatigue. These symptoms eventually disappear and the patient progresses into the latent phase, which betrays no external signs. But if left untreated, after a decade or more, syphilis will reemerge in up to 30% of patients, capable of wreaking horror on a wide range of organ systems. Dr. Marion Sims, president of the American Medical Association in 1876, called it a “terrible scourge, which begins with lamb-like mildness and ends with lion-like rage that ruthlessly destroys everything in its way.”
The corkscrew-shaped bacteria can infiltrate the nervous system at any stage of the infection. Ms. Yang is haunted by her memory of interviewing a young man whose dementia was so severe that he didn’t know why he was in the hospital or how old he was. And regardless of symptoms or stage, the bacteria can penetrate the placenta to infect a fetus. Even in these cases the infection is unpredictable: Many babies are born with normal physical features, but others can have deformed bones or damaged brains, and they can struggle to hear, see, or breathe.
From its earliest days, syphilis has been shrouded in stigma. The first recorded outbreak was in the late 15th century, when Charles VIII led the French army to invade Naples. Italian physicians described French soldiers covered with pustules, dying from a sexually transmitted disease. As the affliction spread, Italians called it the French Disease. The French blamed the Neopolitans. It was also called the German, Polish, or Spanish disease, depending on which neighbor one wanted to blame. Even its name bears the taint of divine judgement: It comes from a 16th-century poem that tells of a shepherd, Syphilus, who offended the god Apollo and was punished with a hideous disease.
By 1937 in America, when former Surgeon General Thomas Parran wrote the book “Shadow on the Land,” he estimated some 680,000 people were under treatment for syphilis; about 60,000 babies were being born annually with congenital syphilis. There was no cure, and the stigma was so strong that public health officials feared even properly documenting cases.
Thanks to Dr. Parran’s ardent advocacy, Congress in 1938 passed the National Venereal Disease Control Act, which created grants for states to set up clinics and support testing and treatment. Other than a short-lived funding effort during World War I, this was the first coordinated federal push to respond to the disease.
Around the same time, the Public Health Service launched an effort to record the natural history of syphilis. Situated in Tuskegee, Ala., the infamous study recruited 600 black men. By the early 1940s, penicillin became widely available and was found to be a reliable cure, but the treatment was withheld from the study participants. Outrage over the ethical violations would cast a stain across syphilis research for decades to come and fuel generations of mistrust in the medical system among Black Americans that continues to this day.
With the introduction of penicillin, cases began to plummet. Twice, the CDC has announced efforts to wipe out the disease — once in the 1960s and again in 1999.
In the latest effort, the CDC announced that the United States had “a unique opportunity to eliminate syphilis within its borders,” thanks to historically low rates, with 80% of counties reporting zero cases. The concentration of cases in the South “identifies communities in which there is a fundamental failure of public health capacity,” the agency noted, adding that elimination — which it defined as fewer than 1,000 cases a year — would “decrease one of our most glaring racial disparities in health.”
Two years after the campaign began, cases started climbing, first among gay men and later, heterosexuals. Cases in women started accelerating in 2013, followed shortly by increasing numbers of babies born with syphilis.The reasons for failure are complex; people relaxed safer sex practices after the advent of potent HIV combination therapies, increased methamphetamine use drove riskier behavior and an explosion of online dating made it hard to track and test sexual partners, according to Dr. Ina Park, medical director of the California Prevention Training Center at the University of California San Francisco.
But federal and state public health efforts were hamstrung from the get-go. In 1999, the CDC said it would need about $35 million to $39 million in new federal funds annually for at least five years to eliminate syphilis. The agency got less than half of what it asked for, according to Jo Valentine, former program coordinator of the CDC’s Syphilis Elimination Effort. As cases rose, the CDC modified its goals in 2006 from 0.4 primary and secondary syphilis cases per 100,000 in population to 2.2 cases per 100,000. By 2013, as elimination seemed less and less viable, the CDC changed its focus to ending congenital syphilis only.
Since then, funding has remained anemic. From 2015 to 2020, the CDC’s budget for preventing sexually transmitted infections grew by 2.2%. Taking inflation into account, that’s a 7.4% reduction in purchasing power. In the same period, cases of syphilis, gonorrhea and chlamydia — the three STDs that have federally funded control programs — increased by nearly 30%.
“We have a long history of nearly eradicating something, then changing our attention, and seeing a resurgence in numbers,” said David Harvey, executive director of the National Coalition of STD Directors. “We have more congenital syphilis cases today in America than we ever had pediatric AIDS at the height of the AIDS epidemic. It’s heartbreaking.”
Adriane Casalotti, chief of government and public affairs at the National Association of County and City Health Officials, warns that the United States should not be surprised to see case counts continue to climb. “The bugs don’t go away,” she said. “They’re just waiting for the next opportunity, when you’re not paying attention.”
Ms. Yang waited until the end of the day, then called the clinic to see if Angelica had gone for her shot. She had not. Ms. Yang would have to block off another half day to visit Huron again, but she had three dozen other cases to deal with.
States in the South and West have seen the highest syphilis rates in recent years. In 2017, 64 babies in Fresno County were born with syphilis at a rate of 440 babies per 100,000 live births — about 19 times the national rate. While the county had managed to lower case counts in the two years that followed, the pandemic threatened to unravel that progress, forcing STD staffers to do COVID-19 contact tracing, pausing field visits to find infected people, and scaring patients from seeking care. Ms. Yang’s colleague handled three cases of stillbirth in 2020; in each, the woman was never diagnosed with syphilis because she feared catching the coronavirus and skipped prenatal care.
Ms. Yang, whose caseload peaked at 70 during a COVID-19 surge, knew she would not be able handle them all as thoroughly as she’d like to. “When I was being mentored by another investigator, he said: ‘You’re not a superhero. You can’t save everybody,’” she said. She prioritizes men who have sex with men, because there’s a higher prevalence of syphilis in that population, and pregnant people, because of the horrific consequences for babies.
The job of a disease intervention specialist isn’t for everyone: It means meeting patients whenever and wherever they are available — in the mop closet of a bus station, in a quiet parking lot — to inform them about the disease, to extract names of sex partners and to encourage treatment. Patients are often reluctant to talk. They can get belligerent, upset that “the government” has their personal information or shattered at the thought that a partner is likely cheating on them. Salaries typically start in the low $40,000s.
Jena Adams, Ms. Yang’s supervisor, has eight investigators working on HIV and syphilis. In the middle of 2020, she lost two and replaced them only recently. “It’s been exhausting,” Ms. Adams said. She has only one specialist who is trained to take blood samples in the field, crucial for guaranteeing that the partners of those who test positive for syphilis also get tested. Ms. Adams wants to get phlebotomy training for the rest of her staff, but it’s $2,000 per person. The department also doesn’t have anyone who can administer penicillin injections in the field; that would have been key when Ms. Yang met Angelica. For a while, a nurse who worked in the tuberculosis program would ride along to give penicillin shots on a volunteer basis. Then he, too, left the health department.
Much of the resources in public health trickle down from the CDC, which distributes money to states, which then parcel it out to counties. The CDC gets its budget from Congress, which tells the agency, by line item, exactly how much money it can spend to fight a disease or virus, in an uncommonly specific manner not seen in many other agencies. The decisions are often politically driven and can be detached from actual health needs.
When the House and Senate appropriations committees meet to decide how much the CDC will get for each line item, they are barraged by lobbyists for individual disease interests. Stephanie Arnold Pang, senior director of policy and government relations at the National Coalition of STD Directors, can pick out the groups by sight: breast cancer wears pink, Alzheimer’s goes in purple, multiple sclerosis comes in orange, HIV in red. STD prevention advocates, like herself, don a green ribbon, but they’re far outnumbered.
And unlike diseases that might already be familiar to lawmakers, or have patient and family spokespeople who can tell their own powerful stories, syphilis doesn’t have many willing poster children. “Congressmen don’t wake up one day and say, ‘Oh hey, there’s congenital syphilis in my jurisdiction.’ You have to raise awareness,” Arnold Pang said. It can be hard jockeying for a meeting. “Some offices might say, ‘I don’t have time for you because we’ve just seen HIV.’ ... Sometimes, it feels like you’re talking into a void.”
The consequences of the political nature of public health funding have become more obvious during the coronavirus pandemic. The 2014 Ebola epidemic was seen as a “global wakeup call” that the world wasn’t prepared for a major pandemic, yet in 2018, the CDC scaled back its epidemic prevention work as money ran out. “If you’ve got to choose between Alzheimer’s research and stopping an outbreak that may not happen? Stopping an outbreak that might not happen doesn’t do well,” said Dr. Frieden, the former CDC director. “The CDC needs to have more money and more flexible money. Otherwise, we’re going to be in this situation long term.”
In May 2021, President Joe Biden’s administration announced it would set aside $7.4 billion over the next five years to hire and train public health workers, including $1.1 billion for more disease intervention specialists like Ms. Yang. Public health officials are thrilled to have the chance to expand their workforce, but some worry the time horizon may be too short. “We’ve seen this movie before, right?” Dr. Frieden said. “Everyone gets concerned when there’s an outbreak, and when that outbreak stops, the headlines stop, and an economic downturn happens, the budget gets cut.”
Fresno’s STD clinic was shuttered in 2010 amid the Great Recession. Many others have vanished since the passage of the Affordable Care Act. Health leaders thought “by magically beefing up the primary care system, that we would do a better job of catching STIs and treating them,” said Mr. Harvey, the executive director of the National Coalition of STD Directors. That hasn’t worked out; people want access to anonymous services, and primary care doctors often don’t have STDs top of mind. The coalition is lobbying Congress for funding to support STD clinical services, proposing a three-year demonstration project funded at $600 million.
It’s one of Ms. Adams’ dreams to see Fresno’s STD clinic restored as it was. “You could come in for an HIV test and get other STDs checked,” she said. “And if a patient is positive, you can give a first injection on the spot.”
On Aug. 12, Ms. Yang set out for Huron again, speeding past groves of almond trees and fields of grapes in the department’s white Chevy Cruze. She brought along a colleague, Jorge Sevilla, who had recently transferred to the STD program from COVID-19 contact tracing. Ms. Yang was anxious to find Angelica again. “She’s probably in her second trimester now,” she said.
They found her outside of a pale yellow house a few blocks from the homeless encampment; the owner was letting her stay in a shed tucked in the corner of the dirt yard. This time, it was evident that she was pregnant. Ms. Yang noted that Angelica was wearing a wig; hair loss is a symptom of syphilis.
“Do you remember me?” Ms. Yang asked.
Angelica nodded. She didn’t seem surprised to see Ms. Yang again. (I came along, and Mr. Sevilla explained who I was and that I was writing about syphilis and the people affected by it. Angelica signed a release for me to report about her case, and she said she had no problem with me writing about her or even using her full name. ProPublica chose to only print her first name.)
“How are you doing? How’s the baby?”
“Bien.”
“So the last time we talked, we were going to have you go to United Healthcare Center to get treatment. Have you gone since?”
Angelica shook her head.
“We brought some gift cards...” Mr. Sevilla started in Spanish. The department uses them as incentives for completing injections. But Angelica was already shaking her head. The nearest Walmart was the next town over.
Ms. Yang turned to her partner. “Tell her: So the reason why we’re coming out here again is because we really need her to go in for treatment. ... We really are concerned for the baby’s health especially since she’s had the infection for quite a while.”
Angelica listened while Mr. Sevilla interpreted, her eyes on the ground. Then she looked up. “Orita?” she asked. Right now?
“I’ll walk with you,” Ms. Yang offered. Angelica shook her head. “She said she wants to shower first before she goes over there,” Mr. Sevilla said.
Ms. Yang made a face. “She said that to me last time.” Ms. Yang offered to wait, but Angelica didn’t want the health officers to linger by the house. She said she would meet them by the clinic in 15 minutes.
Ms. Yang was reluctant to let her go but again had no other option. She and Mr. Sevilla drove to the clinic, then stood on the corner of the parking lot, staring down the road.
Talk to the pediatricians, obstetricians, and families on the front lines of the congenital syphilis surge and it becomes clear why Ms. Yang and others are trying so desperately to prevent cases. Dr. J. B. Cantey, associate professor in pediatrics at UT Health San Antonio, remembers a baby girl born at 25 weeks gestation who weighed a pound and a half. Syphilis had spread through her bones and lungs. She spent five months in the neonatal intensive care unit, breathing through a ventilator, and was still eating through a tube when she was discharged.
Then, there are the miscarriages, the stillbirths and the inconsolable parents. Dr. Irene Stafford, an associate professor and maternal-fetal medicine specialist at UT Health in Houston, cannot forget a patient who came in at 36 weeks for a routine checkup, pregnant with her first child. Dr. Stafford realized that there was no heartbeat. “She could see on my face that something was really wrong,” Dr. Stafford recalled. She had to let the patient know that syphilis had killed her baby. “She was hysterical, just bawling,” Dr. Stafford said. “I’ve seen people’s families ripped apart and I’ve seen beautiful babies die.” Fewer than 10% of patients who experience a stillbirth are tested for syphilis, suggesting that cases are underdiagnosed.
A Texas grandmother named Solidad Odunuga offers a glimpse into what the future could hold for Angelica’s mother, who may wind up raising her baby.
In February of last year, Ms. Odunuga got a call from the Lyndon B. Johnson Hospital in Houston. A nurse told her that her daughter was about to give birth and that child protective services had been called. Ms. Odunuga had lost contact with her daughter, who struggled with homelessness and substance abuse. She arrived in time to see her grandson delivered, premature at 30 weeks old, weighing 2.7 pounds. He tested positive for syphilis.
When a child protective worker asked Ms. Odunuga to take custody of the infant, she felt a wave of dread. “I was in denial,” she recalled. “I did not plan to be a mom again.” The baby’s medical problems were daunting: “Global developmental delays ... concerns for visual impairments ... high risk of cerebral palsy,” read a note from the doctor at the time.
Still, Ms. Odunuga visited her grandson every day for three months, driving to the NICU from her job at the University of Houston. “I’d put him in my shirt to keep him warm and hold him there.” She fell in love. She named him Emmanuel.
Once Emmanuel was discharged, Ms. Odunuga realized she had no choice but to quit her job. While Medicaid covered the costs of Emmanuel’s treatment, it was on her to care for him. From infancy, Emmanuel’s life has been a whirlwind of constant therapy. Today, at 20 months old, Odunuga brings him to physical, occupational, speech, and developmental therapy, each a different appointment on a different day of the week.
Emmanuel has thrived beyond what his doctors predicted, toddling so fast that Ms. Odunuga can’t look away for a minute and beaming as he waves his favorite toy phone. Yet he still suffers from gagging issues, which means Ms. Odunuga can’t feed him any solid foods. Liquid gets into his lungs when he aspirates; it has led to pneumonia three times. Emmanuel has a special stroller that helps keep his head in a position that won’t aggravate his persistent reflux, but Odunuga said she still has to pull over on the side of the road sometimes when she hears him projectile vomiting from the backseat.
The days are endless. Once she puts Emmanuel to bed, Ms. Odunuga starts planning the next day’s appointments. “I’ve had to cry alone, scream out alone,” she said. “Sometimes I wake up and think, Is this real? And then I hear him in the next room.”
Putting aside the challenge of eliminating syphilis entirely, everyone agrees it’s both doable and necessary to prevent newborn cases. “There was a crisis in perinatal HIV almost 30 years ago and people stood up and said this is not OK — it’s not acceptable for babies to be born in that condition. ... [We] brought it down from 1,700 babies born each year with perinatal HIV to less than 40 per year today,” said Virginia Bowen, an epidemiologist at the CDC. “Now here we are with a slightly different condition. We can also stand up and say, ‘This is not acceptable.’” Belarus, Bermuda, Cuba, Malaysia, Thailand, and Sri Lanka are among countries recognized by the World Health Organization for eliminating congenital syphilis.
Success starts with filling gaps across the health care system.
For almost a century, public health experts have advocated for testing pregnant patients more than once for syphilis in order to catch the infection. But policies nationwide still don’t reflect this best practice. Six states have no prenatal screening requirement at all. Even in states that require three tests, public health officials say that many physicians aren’t aware of the requirements. Dr. Stafford, the maternal-fetal medicine specialist in Houston, says she’s tired of hearing her own peers in medicine tell her, “Oh, syphilis is a problem?”
It costs public health departments less than 25 cents a dose to buy penicillin, but for a private practice, it’s more than $1,000, according to Dr. Park of the University of California San Francisco. “There’s no incentive for a private physician to stock a dose that could expire before it’s used, so they often don’t have it. So a woman comes in, they say, ‘We’ll send you to the emergency department or health department to get it,’ then [the patients] don’t show up.”
A vaccine would be invaluable for preventing spread among people at high risk for reinfection. But there is none. Scientists only recently figured out how to grow the bacteria in the lab, prompting grants from the National Institutes of Health to fund research into a vaccine. Dr. Justin Radolf, a researcher at the University of Connecticut School of Medicine, said he hopes his team will have a vaccine candidate by the end of its five-year grant. But it’ll likely take years more to find a manufacturer and run human trials.
Public health agencies also need to recognize that many of the hurdles to getting pregnant people treated involve access to care, economic stability, safe housing and transportation. In Fresno, Ms. Adams has been working on ways her department can collaborate with mental health services. Recently, one of her disease intervention specialists managed to get a pregnant woman treated with penicillin shots and, at the patient’s request, connected her with an addiction treatment center.
Gaining a patient’s cooperation means seeing them as complex humans instead of just a case to solve. “There may be past traumas with the health care system,” said Cynthia Deverson, project manager of the Houston Fetal Infant Morbidity Review. “There’s the fear of being discovered if she’s doing something illegal to survive. ... She may need to be in a certain place at a certain time so she can get something to eat, or maybe it’s the only time of the day that’s safe for her to sleep. They’re not going to tell you that. Yes, they understand there’s a problem, but it’s not an immediate threat, maybe they don’t feel bad yet, so obviously this is not urgent. ...
“What helps to gain trust is consistency,” she said. “Literally, it’s seeing that [disease specialist] constantly, daily. ... The woman can see that you’re not going to harm her, you’re saying, ‘I’m here at this time if you need me.’”
Ms. Yang stood outside the clinic, waiting for Angelica to show up, baking in the 90-degree heat. Her feelings ranged from irritation — Why didn’t she just go? I’d have more energy for other cases — to an appreciation for the parts of Angelica’s story that she didn’t know — She’s in survival mode. I need to be more patient.
Fifteen minutes ticked by, then 20.
“OK,” Ms. Yang announced. “We’re going back.”
She asked Sevilla if he would be OK if they drove Angelica to the clinic; they technically weren’t supposed to because of coronavirus precautions, but Ms. Yang wasn’t sure she could convince Angelica to walk. Mr. Sevilla gave her the thumbs up.
When they pulled up, they saw Angelica sitting in the backyard, chatting with a friend. She now wore a fresh T-shirt and had shoes on her feet. Angelica sat silently in the back seat as Ms. Yang drove to the clinic. A few minutes later, they pulled up to the parking lot.
Finally, Ms. Yang thought. We got her here.
The clinic was packed with people waiting for COVID-19 tests and vaccinations. A worker there had previously told Ms. Yang that a walk-in would be fine, but a receptionist now said they were too busy to treat Angelica. She would have to return.
Ms. Yang felt a surge of frustration, sensing that her hard-fought opportunity was slipping away. She tried to talk to the nurse supervisor, but he wasn’t available. She tried to leave the gift cards at the office to reward Angelica if she came, but the receptionist said she couldn’t hold them. While Ms. Yang negotiated, Mr. Sevilla sat with Angelica in the car, waiting.
Finally, Ms. Yang accepted this was yet another thing she couldn’t control.
She drove Angelica back to the yellow house. As they arrived, she tried once more to impress on her just how important it was to get treated, asking Mr. Sevilla to interpret. “We don’t want it to get any more serious, because she can go blind, she could go deaf, she could lose her baby.”
Angelica already had the door halfway open.
“So on a scale from one to 10, how important is this to get treated?” Ms. Yang asked.
“Ten,” Angelica said. Ms. Yang reminded her of the appointment that afternoon. Then Angelica stepped out and returned to the dusty yard.
Ms. Yang lingered for a moment, watching Angelica go. Then she turned the car back onto the highway and set off toward Fresno, knowing, already, that she’d be back.
Postscript: A reporter visited Huron twice more in the months that followed, including once independently to try to interview Angelica, but she wasn’t in town. Ms. Yang has visited Huron twice more as well — six times in total thus far. In October, a couple of men at the yellow house said Angelica was still in town, still pregnant. Ms. Yang and Mr. Sevilla spent an hour driving around, talking to residents, hoping to catch Angelica. But she was nowhere to be found.
ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
This story was originally published on ProPublica and was co-published with NPR.
When Mai Yang is looking for a patient, she travels light. She dresses deliberately — not too formal, so she won’t be mistaken for a police officer; not too casual, so people will look past her tiny 4-foot-10 stature and youthful face and trust her with sensitive health information. Always, she wears closed-toed shoes, “just in case I need to run.”
Yang carries a stack of cards issued by the Centers for Disease Control and Prevention that show what happens when the Treponema pallidum bacteria invades a patient’s body. There’s a photo of an angry red sore on a penis. There’s one of a tongue, marred by mucus-lined lesions. And there’s one of a newborn baby, its belly, torso and thighs dotted in a rash, its mouth open, as if caught midcry.
It was because of the prospect of one such baby that Yang found herself walking through a homeless encampment on a blazing July day in Huron, Calif., an hour’s drive southwest of her office at the Fresno County Department of Public Health. She was looking for a pregnant woman named Angelica, whose visit to a community clinic had triggered a report to the health department’s sexually transmitted disease program. Angelica had tested positive for syphilis. If she was not treated, her baby could end up like the one in the picture or worse — there was a 40% chance the baby would die.
Ms. Yang knew, though, that if she helped Angelica get treated with three weekly shots of penicillin at least 30 days before she gave birth, it was likely that the infection would be wiped out and her baby would be born without any symptoms at all. Every case of congenital syphilis, when a baby is born with the disease, is avoidable. Each is considered a “sentinel event,” a warning that the public health system is failing.
The alarms are now clamoring. In the United States, more than 129,800 syphilis cases were recorded in 2019, double the case count of five years prior. In the same time period, cases of congenital syphilis quadrupled: 1,870 babies were born with the disease; 128 died. Case counts from 2020 are still being finalized, but the CDC has said that reported cases of congenital syphilis have already exceeded the prior year. Black, Hispanic, and Native American babies are disproportionately at risk.
There was a time, not too long ago, when CDC officials thought they could eliminate the centuries-old scourge from the United States, for adults and babies. But the effort lost steam and cases soon crept up again. Syphilis is not an outlier. The United States goes through what former CDC director Dr. Tom Frieden calls “a deadly cycle of panic and neglect” in which emergencies propel officials to scramble and throw money at a problem — whether that’s Ebola, Zika, or COVID-19. Then, as fear ebbs, so does the attention and motivation to finish the task.
The last fraction of cases can be the hardest to solve, whether that’s eradicating a bug or getting vaccines into arms, yet too often, that’s exactly when political attention gets diverted to the next alarm. The result: The hardest to reach and most vulnerable populations are the ones left suffering, after everyone else looks away.
Ms. Yang first received Angelica’s lab report on June 17. The address listed was a P.O. box, and the phone number belonged to her sister, who said Angelica was living in Huron. That was a piece of luck: Huron is tiny; the city spans just 1.6 square miles. On her first visit, a worker at the Alamo Motel said she knew Angelica and directed Ms. Yang to a nearby homeless encampment. Angelica wasn’t there, so Ms. Yang returned a second time, bringing one of the health department nurses who could serve as an interpreter.
They made their way to the barren patch of land behind Huron Valley Foods, the local grocery store, where people took shelter in makeshift lean-tos composed of cardboard boxes, scrap wood ,and scavenged furniture, draped with sheets that served as ceilings and curtains. Yang stopped outside one of the structures, calling a greeting.
“Hi, I’m from the health department, I’m looking for Angelica.”
The nurse echoed her in Spanish.
Angelica emerged, squinting in the sunlight. Ms. Yang couldn’t tell if she was visibly pregnant yet, as her body was obscured by an oversized shirt. The two women were about the same age: Ms. Yang 26 and Angelica 27. Ms. Yang led her away from the tent, so they could speak privately. Angelica seemed reticent, surprised by the sudden appearance of the two health officers. “You’re not in trouble,” Ms. Yang said, before revealing the results of her blood test.
Angelica had never heard of syphilis.
“Have you been to prenatal care?”
Angelica shook her head. The local clinic had referred her to an obstetrician in Hanford, a 30-minute drive away. She had no car. She also mentioned that she didn’t intend to raise her baby; her two oldest children lived with her mother, and this one likely would, too.
Ms. Yang pulled out the CDC cards, showing them to Angelica and asking if she had experienced any of the symptoms illustrated. No, Angelica said, her lips pursed with disgust.
“Right now you still feel healthy, but this bacteria is still in your body,” Ms. Yang pressed. “You need to get the infection treated to prevent further health complications to yourself and your baby.”
The community clinic was just across the street. “Can we walk you over to the clinic and make sure you get seen so we can get this taken care of?”
Angelica demurred. She said she hadn’t showered for a week and wanted to wash up first. She said she’d go later.
Ms. Yang tried once more to extract a promise: “What time do you think you’ll go?”
“Today, for sure.”
Syphilis is called The Great Imitator: It can look like any number of diseases. In its first stage, the only evidence of infection is a painless sore at the bacteria’s point of entry. Weeks later, as the bacteria multiplies, skin rashes bloom on the palms of the hands and bottoms of the feet. Other traits of this stage include fever, headaches, muscle aches, sore throat, and fatigue. These symptoms eventually disappear and the patient progresses into the latent phase, which betrays no external signs. But if left untreated, after a decade or more, syphilis will reemerge in up to 30% of patients, capable of wreaking horror on a wide range of organ systems. Dr. Marion Sims, president of the American Medical Association in 1876, called it a “terrible scourge, which begins with lamb-like mildness and ends with lion-like rage that ruthlessly destroys everything in its way.”
The corkscrew-shaped bacteria can infiltrate the nervous system at any stage of the infection. Ms. Yang is haunted by her memory of interviewing a young man whose dementia was so severe that he didn’t know why he was in the hospital or how old he was. And regardless of symptoms or stage, the bacteria can penetrate the placenta to infect a fetus. Even in these cases the infection is unpredictable: Many babies are born with normal physical features, but others can have deformed bones or damaged brains, and they can struggle to hear, see, or breathe.
From its earliest days, syphilis has been shrouded in stigma. The first recorded outbreak was in the late 15th century, when Charles VIII led the French army to invade Naples. Italian physicians described French soldiers covered with pustules, dying from a sexually transmitted disease. As the affliction spread, Italians called it the French Disease. The French blamed the Neopolitans. It was also called the German, Polish, or Spanish disease, depending on which neighbor one wanted to blame. Even its name bears the taint of divine judgement: It comes from a 16th-century poem that tells of a shepherd, Syphilus, who offended the god Apollo and was punished with a hideous disease.
By 1937 in America, when former Surgeon General Thomas Parran wrote the book “Shadow on the Land,” he estimated some 680,000 people were under treatment for syphilis; about 60,000 babies were being born annually with congenital syphilis. There was no cure, and the stigma was so strong that public health officials feared even properly documenting cases.
Thanks to Dr. Parran’s ardent advocacy, Congress in 1938 passed the National Venereal Disease Control Act, which created grants for states to set up clinics and support testing and treatment. Other than a short-lived funding effort during World War I, this was the first coordinated federal push to respond to the disease.
Around the same time, the Public Health Service launched an effort to record the natural history of syphilis. Situated in Tuskegee, Ala., the infamous study recruited 600 black men. By the early 1940s, penicillin became widely available and was found to be a reliable cure, but the treatment was withheld from the study participants. Outrage over the ethical violations would cast a stain across syphilis research for decades to come and fuel generations of mistrust in the medical system among Black Americans that continues to this day.
With the introduction of penicillin, cases began to plummet. Twice, the CDC has announced efforts to wipe out the disease — once in the 1960s and again in 1999.
In the latest effort, the CDC announced that the United States had “a unique opportunity to eliminate syphilis within its borders,” thanks to historically low rates, with 80% of counties reporting zero cases. The concentration of cases in the South “identifies communities in which there is a fundamental failure of public health capacity,” the agency noted, adding that elimination — which it defined as fewer than 1,000 cases a year — would “decrease one of our most glaring racial disparities in health.”
Two years after the campaign began, cases started climbing, first among gay men and later, heterosexuals. Cases in women started accelerating in 2013, followed shortly by increasing numbers of babies born with syphilis.The reasons for failure are complex; people relaxed safer sex practices after the advent of potent HIV combination therapies, increased methamphetamine use drove riskier behavior and an explosion of online dating made it hard to track and test sexual partners, according to Dr. Ina Park, medical director of the California Prevention Training Center at the University of California San Francisco.
But federal and state public health efforts were hamstrung from the get-go. In 1999, the CDC said it would need about $35 million to $39 million in new federal funds annually for at least five years to eliminate syphilis. The agency got less than half of what it asked for, according to Jo Valentine, former program coordinator of the CDC’s Syphilis Elimination Effort. As cases rose, the CDC modified its goals in 2006 from 0.4 primary and secondary syphilis cases per 100,000 in population to 2.2 cases per 100,000. By 2013, as elimination seemed less and less viable, the CDC changed its focus to ending congenital syphilis only.
Since then, funding has remained anemic. From 2015 to 2020, the CDC’s budget for preventing sexually transmitted infections grew by 2.2%. Taking inflation into account, that’s a 7.4% reduction in purchasing power. In the same period, cases of syphilis, gonorrhea and chlamydia — the three STDs that have federally funded control programs — increased by nearly 30%.
“We have a long history of nearly eradicating something, then changing our attention, and seeing a resurgence in numbers,” said David Harvey, executive director of the National Coalition of STD Directors. “We have more congenital syphilis cases today in America than we ever had pediatric AIDS at the height of the AIDS epidemic. It’s heartbreaking.”
Adriane Casalotti, chief of government and public affairs at the National Association of County and City Health Officials, warns that the United States should not be surprised to see case counts continue to climb. “The bugs don’t go away,” she said. “They’re just waiting for the next opportunity, when you’re not paying attention.”
Ms. Yang waited until the end of the day, then called the clinic to see if Angelica had gone for her shot. She had not. Ms. Yang would have to block off another half day to visit Huron again, but she had three dozen other cases to deal with.
States in the South and West have seen the highest syphilis rates in recent years. In 2017, 64 babies in Fresno County were born with syphilis at a rate of 440 babies per 100,000 live births — about 19 times the national rate. While the county had managed to lower case counts in the two years that followed, the pandemic threatened to unravel that progress, forcing STD staffers to do COVID-19 contact tracing, pausing field visits to find infected people, and scaring patients from seeking care. Ms. Yang’s colleague handled three cases of stillbirth in 2020; in each, the woman was never diagnosed with syphilis because she feared catching the coronavirus and skipped prenatal care.
Ms. Yang, whose caseload peaked at 70 during a COVID-19 surge, knew she would not be able handle them all as thoroughly as she’d like to. “When I was being mentored by another investigator, he said: ‘You’re not a superhero. You can’t save everybody,’” she said. She prioritizes men who have sex with men, because there’s a higher prevalence of syphilis in that population, and pregnant people, because of the horrific consequences for babies.
The job of a disease intervention specialist isn’t for everyone: It means meeting patients whenever and wherever they are available — in the mop closet of a bus station, in a quiet parking lot — to inform them about the disease, to extract names of sex partners and to encourage treatment. Patients are often reluctant to talk. They can get belligerent, upset that “the government” has their personal information or shattered at the thought that a partner is likely cheating on them. Salaries typically start in the low $40,000s.
Jena Adams, Ms. Yang’s supervisor, has eight investigators working on HIV and syphilis. In the middle of 2020, she lost two and replaced them only recently. “It’s been exhausting,” Ms. Adams said. She has only one specialist who is trained to take blood samples in the field, crucial for guaranteeing that the partners of those who test positive for syphilis also get tested. Ms. Adams wants to get phlebotomy training for the rest of her staff, but it’s $2,000 per person. The department also doesn’t have anyone who can administer penicillin injections in the field; that would have been key when Ms. Yang met Angelica. For a while, a nurse who worked in the tuberculosis program would ride along to give penicillin shots on a volunteer basis. Then he, too, left the health department.
Much of the resources in public health trickle down from the CDC, which distributes money to states, which then parcel it out to counties. The CDC gets its budget from Congress, which tells the agency, by line item, exactly how much money it can spend to fight a disease or virus, in an uncommonly specific manner not seen in many other agencies. The decisions are often politically driven and can be detached from actual health needs.
When the House and Senate appropriations committees meet to decide how much the CDC will get for each line item, they are barraged by lobbyists for individual disease interests. Stephanie Arnold Pang, senior director of policy and government relations at the National Coalition of STD Directors, can pick out the groups by sight: breast cancer wears pink, Alzheimer’s goes in purple, multiple sclerosis comes in orange, HIV in red. STD prevention advocates, like herself, don a green ribbon, but they’re far outnumbered.
And unlike diseases that might already be familiar to lawmakers, or have patient and family spokespeople who can tell their own powerful stories, syphilis doesn’t have many willing poster children. “Congressmen don’t wake up one day and say, ‘Oh hey, there’s congenital syphilis in my jurisdiction.’ You have to raise awareness,” Arnold Pang said. It can be hard jockeying for a meeting. “Some offices might say, ‘I don’t have time for you because we’ve just seen HIV.’ ... Sometimes, it feels like you’re talking into a void.”
The consequences of the political nature of public health funding have become more obvious during the coronavirus pandemic. The 2014 Ebola epidemic was seen as a “global wakeup call” that the world wasn’t prepared for a major pandemic, yet in 2018, the CDC scaled back its epidemic prevention work as money ran out. “If you’ve got to choose between Alzheimer’s research and stopping an outbreak that may not happen? Stopping an outbreak that might not happen doesn’t do well,” said Dr. Frieden, the former CDC director. “The CDC needs to have more money and more flexible money. Otherwise, we’re going to be in this situation long term.”
In May 2021, President Joe Biden’s administration announced it would set aside $7.4 billion over the next five years to hire and train public health workers, including $1.1 billion for more disease intervention specialists like Ms. Yang. Public health officials are thrilled to have the chance to expand their workforce, but some worry the time horizon may be too short. “We’ve seen this movie before, right?” Dr. Frieden said. “Everyone gets concerned when there’s an outbreak, and when that outbreak stops, the headlines stop, and an economic downturn happens, the budget gets cut.”
Fresno’s STD clinic was shuttered in 2010 amid the Great Recession. Many others have vanished since the passage of the Affordable Care Act. Health leaders thought “by magically beefing up the primary care system, that we would do a better job of catching STIs and treating them,” said Mr. Harvey, the executive director of the National Coalition of STD Directors. That hasn’t worked out; people want access to anonymous services, and primary care doctors often don’t have STDs top of mind. The coalition is lobbying Congress for funding to support STD clinical services, proposing a three-year demonstration project funded at $600 million.
It’s one of Ms. Adams’ dreams to see Fresno’s STD clinic restored as it was. “You could come in for an HIV test and get other STDs checked,” she said. “And if a patient is positive, you can give a first injection on the spot.”
On Aug. 12, Ms. Yang set out for Huron again, speeding past groves of almond trees and fields of grapes in the department’s white Chevy Cruze. She brought along a colleague, Jorge Sevilla, who had recently transferred to the STD program from COVID-19 contact tracing. Ms. Yang was anxious to find Angelica again. “She’s probably in her second trimester now,” she said.
They found her outside of a pale yellow house a few blocks from the homeless encampment; the owner was letting her stay in a shed tucked in the corner of the dirt yard. This time, it was evident that she was pregnant. Ms. Yang noted that Angelica was wearing a wig; hair loss is a symptom of syphilis.
“Do you remember me?” Ms. Yang asked.
Angelica nodded. She didn’t seem surprised to see Ms. Yang again. (I came along, and Mr. Sevilla explained who I was and that I was writing about syphilis and the people affected by it. Angelica signed a release for me to report about her case, and she said she had no problem with me writing about her or even using her full name. ProPublica chose to only print her first name.)
“How are you doing? How’s the baby?”
“Bien.”
“So the last time we talked, we were going to have you go to United Healthcare Center to get treatment. Have you gone since?”
Angelica shook her head.
“We brought some gift cards...” Mr. Sevilla started in Spanish. The department uses them as incentives for completing injections. But Angelica was already shaking her head. The nearest Walmart was the next town over.
Ms. Yang turned to her partner. “Tell her: So the reason why we’re coming out here again is because we really need her to go in for treatment. ... We really are concerned for the baby’s health especially since she’s had the infection for quite a while.”
Angelica listened while Mr. Sevilla interpreted, her eyes on the ground. Then she looked up. “Orita?” she asked. Right now?
“I’ll walk with you,” Ms. Yang offered. Angelica shook her head. “She said she wants to shower first before she goes over there,” Mr. Sevilla said.
Ms. Yang made a face. “She said that to me last time.” Ms. Yang offered to wait, but Angelica didn’t want the health officers to linger by the house. She said she would meet them by the clinic in 15 minutes.
Ms. Yang was reluctant to let her go but again had no other option. She and Mr. Sevilla drove to the clinic, then stood on the corner of the parking lot, staring down the road.
Talk to the pediatricians, obstetricians, and families on the front lines of the congenital syphilis surge and it becomes clear why Ms. Yang and others are trying so desperately to prevent cases. Dr. J. B. Cantey, associate professor in pediatrics at UT Health San Antonio, remembers a baby girl born at 25 weeks gestation who weighed a pound and a half. Syphilis had spread through her bones and lungs. She spent five months in the neonatal intensive care unit, breathing through a ventilator, and was still eating through a tube when she was discharged.
Then, there are the miscarriages, the stillbirths and the inconsolable parents. Dr. Irene Stafford, an associate professor and maternal-fetal medicine specialist at UT Health in Houston, cannot forget a patient who came in at 36 weeks for a routine checkup, pregnant with her first child. Dr. Stafford realized that there was no heartbeat. “She could see on my face that something was really wrong,” Dr. Stafford recalled. She had to let the patient know that syphilis had killed her baby. “She was hysterical, just bawling,” Dr. Stafford said. “I’ve seen people’s families ripped apart and I’ve seen beautiful babies die.” Fewer than 10% of patients who experience a stillbirth are tested for syphilis, suggesting that cases are underdiagnosed.
A Texas grandmother named Solidad Odunuga offers a glimpse into what the future could hold for Angelica’s mother, who may wind up raising her baby.
In February of last year, Ms. Odunuga got a call from the Lyndon B. Johnson Hospital in Houston. A nurse told her that her daughter was about to give birth and that child protective services had been called. Ms. Odunuga had lost contact with her daughter, who struggled with homelessness and substance abuse. She arrived in time to see her grandson delivered, premature at 30 weeks old, weighing 2.7 pounds. He tested positive for syphilis.
When a child protective worker asked Ms. Odunuga to take custody of the infant, she felt a wave of dread. “I was in denial,” she recalled. “I did not plan to be a mom again.” The baby’s medical problems were daunting: “Global developmental delays ... concerns for visual impairments ... high risk of cerebral palsy,” read a note from the doctor at the time.
Still, Ms. Odunuga visited her grandson every day for three months, driving to the NICU from her job at the University of Houston. “I’d put him in my shirt to keep him warm and hold him there.” She fell in love. She named him Emmanuel.
Once Emmanuel was discharged, Ms. Odunuga realized she had no choice but to quit her job. While Medicaid covered the costs of Emmanuel’s treatment, it was on her to care for him. From infancy, Emmanuel’s life has been a whirlwind of constant therapy. Today, at 20 months old, Odunuga brings him to physical, occupational, speech, and developmental therapy, each a different appointment on a different day of the week.
Emmanuel has thrived beyond what his doctors predicted, toddling so fast that Ms. Odunuga can’t look away for a minute and beaming as he waves his favorite toy phone. Yet he still suffers from gagging issues, which means Ms. Odunuga can’t feed him any solid foods. Liquid gets into his lungs when he aspirates; it has led to pneumonia three times. Emmanuel has a special stroller that helps keep his head in a position that won’t aggravate his persistent reflux, but Odunuga said she still has to pull over on the side of the road sometimes when she hears him projectile vomiting from the backseat.
The days are endless. Once she puts Emmanuel to bed, Ms. Odunuga starts planning the next day’s appointments. “I’ve had to cry alone, scream out alone,” she said. “Sometimes I wake up and think, Is this real? And then I hear him in the next room.”
Putting aside the challenge of eliminating syphilis entirely, everyone agrees it’s both doable and necessary to prevent newborn cases. “There was a crisis in perinatal HIV almost 30 years ago and people stood up and said this is not OK — it’s not acceptable for babies to be born in that condition. ... [We] brought it down from 1,700 babies born each year with perinatal HIV to less than 40 per year today,” said Virginia Bowen, an epidemiologist at the CDC. “Now here we are with a slightly different condition. We can also stand up and say, ‘This is not acceptable.’” Belarus, Bermuda, Cuba, Malaysia, Thailand, and Sri Lanka are among countries recognized by the World Health Organization for eliminating congenital syphilis.
Success starts with filling gaps across the health care system.
For almost a century, public health experts have advocated for testing pregnant patients more than once for syphilis in order to catch the infection. But policies nationwide still don’t reflect this best practice. Six states have no prenatal screening requirement at all. Even in states that require three tests, public health officials say that many physicians aren’t aware of the requirements. Dr. Stafford, the maternal-fetal medicine specialist in Houston, says she’s tired of hearing her own peers in medicine tell her, “Oh, syphilis is a problem?”
It costs public health departments less than 25 cents a dose to buy penicillin, but for a private practice, it’s more than $1,000, according to Dr. Park of the University of California San Francisco. “There’s no incentive for a private physician to stock a dose that could expire before it’s used, so they often don’t have it. So a woman comes in, they say, ‘We’ll send you to the emergency department or health department to get it,’ then [the patients] don’t show up.”
A vaccine would be invaluable for preventing spread among people at high risk for reinfection. But there is none. Scientists only recently figured out how to grow the bacteria in the lab, prompting grants from the National Institutes of Health to fund research into a vaccine. Dr. Justin Radolf, a researcher at the University of Connecticut School of Medicine, said he hopes his team will have a vaccine candidate by the end of its five-year grant. But it’ll likely take years more to find a manufacturer and run human trials.
Public health agencies also need to recognize that many of the hurdles to getting pregnant people treated involve access to care, economic stability, safe housing and transportation. In Fresno, Ms. Adams has been working on ways her department can collaborate with mental health services. Recently, one of her disease intervention specialists managed to get a pregnant woman treated with penicillin shots and, at the patient’s request, connected her with an addiction treatment center.
Gaining a patient’s cooperation means seeing them as complex humans instead of just a case to solve. “There may be past traumas with the health care system,” said Cynthia Deverson, project manager of the Houston Fetal Infant Morbidity Review. “There’s the fear of being discovered if she’s doing something illegal to survive. ... She may need to be in a certain place at a certain time so she can get something to eat, or maybe it’s the only time of the day that’s safe for her to sleep. They’re not going to tell you that. Yes, they understand there’s a problem, but it’s not an immediate threat, maybe they don’t feel bad yet, so obviously this is not urgent. ...
“What helps to gain trust is consistency,” she said. “Literally, it’s seeing that [disease specialist] constantly, daily. ... The woman can see that you’re not going to harm her, you’re saying, ‘I’m here at this time if you need me.’”
Ms. Yang stood outside the clinic, waiting for Angelica to show up, baking in the 90-degree heat. Her feelings ranged from irritation — Why didn’t she just go? I’d have more energy for other cases — to an appreciation for the parts of Angelica’s story that she didn’t know — She’s in survival mode. I need to be more patient.
Fifteen minutes ticked by, then 20.
“OK,” Ms. Yang announced. “We’re going back.”
She asked Sevilla if he would be OK if they drove Angelica to the clinic; they technically weren’t supposed to because of coronavirus precautions, but Ms. Yang wasn’t sure she could convince Angelica to walk. Mr. Sevilla gave her the thumbs up.
When they pulled up, they saw Angelica sitting in the backyard, chatting with a friend. She now wore a fresh T-shirt and had shoes on her feet. Angelica sat silently in the back seat as Ms. Yang drove to the clinic. A few minutes later, they pulled up to the parking lot.
Finally, Ms. Yang thought. We got her here.
The clinic was packed with people waiting for COVID-19 tests and vaccinations. A worker there had previously told Ms. Yang that a walk-in would be fine, but a receptionist now said they were too busy to treat Angelica. She would have to return.
Ms. Yang felt a surge of frustration, sensing that her hard-fought opportunity was slipping away. She tried to talk to the nurse supervisor, but he wasn’t available. She tried to leave the gift cards at the office to reward Angelica if she came, but the receptionist said she couldn’t hold them. While Ms. Yang negotiated, Mr. Sevilla sat with Angelica in the car, waiting.
Finally, Ms. Yang accepted this was yet another thing she couldn’t control.
She drove Angelica back to the yellow house. As they arrived, she tried once more to impress on her just how important it was to get treated, asking Mr. Sevilla to interpret. “We don’t want it to get any more serious, because she can go blind, she could go deaf, she could lose her baby.”
Angelica already had the door halfway open.
“So on a scale from one to 10, how important is this to get treated?” Ms. Yang asked.
“Ten,” Angelica said. Ms. Yang reminded her of the appointment that afternoon. Then Angelica stepped out and returned to the dusty yard.
Ms. Yang lingered for a moment, watching Angelica go. Then she turned the car back onto the highway and set off toward Fresno, knowing, already, that she’d be back.
Postscript: A reporter visited Huron twice more in the months that followed, including once independently to try to interview Angelica, but she wasn’t in town. Ms. Yang has visited Huron twice more as well — six times in total thus far. In October, a couple of men at the yellow house said Angelica was still in town, still pregnant. Ms. Yang and Mr. Sevilla spent an hour driving around, talking to residents, hoping to catch Angelica. But she was nowhere to be found.
ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
This story was originally published on ProPublica and was co-published with NPR.
When Mai Yang is looking for a patient, she travels light. She dresses deliberately — not too formal, so she won’t be mistaken for a police officer; not too casual, so people will look past her tiny 4-foot-10 stature and youthful face and trust her with sensitive health information. Always, she wears closed-toed shoes, “just in case I need to run.”
Yang carries a stack of cards issued by the Centers for Disease Control and Prevention that show what happens when the Treponema pallidum bacteria invades a patient’s body. There’s a photo of an angry red sore on a penis. There’s one of a tongue, marred by mucus-lined lesions. And there’s one of a newborn baby, its belly, torso and thighs dotted in a rash, its mouth open, as if caught midcry.
It was because of the prospect of one such baby that Yang found herself walking through a homeless encampment on a blazing July day in Huron, Calif., an hour’s drive southwest of her office at the Fresno County Department of Public Health. She was looking for a pregnant woman named Angelica, whose visit to a community clinic had triggered a report to the health department’s sexually transmitted disease program. Angelica had tested positive for syphilis. If she was not treated, her baby could end up like the one in the picture or worse — there was a 40% chance the baby would die.
Ms. Yang knew, though, that if she helped Angelica get treated with three weekly shots of penicillin at least 30 days before she gave birth, it was likely that the infection would be wiped out and her baby would be born without any symptoms at all. Every case of congenital syphilis, when a baby is born with the disease, is avoidable. Each is considered a “sentinel event,” a warning that the public health system is failing.
The alarms are now clamoring. In the United States, more than 129,800 syphilis cases were recorded in 2019, double the case count of five years prior. In the same time period, cases of congenital syphilis quadrupled: 1,870 babies were born with the disease; 128 died. Case counts from 2020 are still being finalized, but the CDC has said that reported cases of congenital syphilis have already exceeded the prior year. Black, Hispanic, and Native American babies are disproportionately at risk.
There was a time, not too long ago, when CDC officials thought they could eliminate the centuries-old scourge from the United States, for adults and babies. But the effort lost steam and cases soon crept up again. Syphilis is not an outlier. The United States goes through what former CDC director Dr. Tom Frieden calls “a deadly cycle of panic and neglect” in which emergencies propel officials to scramble and throw money at a problem — whether that’s Ebola, Zika, or COVID-19. Then, as fear ebbs, so does the attention and motivation to finish the task.
The last fraction of cases can be the hardest to solve, whether that’s eradicating a bug or getting vaccines into arms, yet too often, that’s exactly when political attention gets diverted to the next alarm. The result: The hardest to reach and most vulnerable populations are the ones left suffering, after everyone else looks away.
Ms. Yang first received Angelica’s lab report on June 17. The address listed was a P.O. box, and the phone number belonged to her sister, who said Angelica was living in Huron. That was a piece of luck: Huron is tiny; the city spans just 1.6 square miles. On her first visit, a worker at the Alamo Motel said she knew Angelica and directed Ms. Yang to a nearby homeless encampment. Angelica wasn’t there, so Ms. Yang returned a second time, bringing one of the health department nurses who could serve as an interpreter.
They made their way to the barren patch of land behind Huron Valley Foods, the local grocery store, where people took shelter in makeshift lean-tos composed of cardboard boxes, scrap wood ,and scavenged furniture, draped with sheets that served as ceilings and curtains. Yang stopped outside one of the structures, calling a greeting.
“Hi, I’m from the health department, I’m looking for Angelica.”
The nurse echoed her in Spanish.
Angelica emerged, squinting in the sunlight. Ms. Yang couldn’t tell if she was visibly pregnant yet, as her body was obscured by an oversized shirt. The two women were about the same age: Ms. Yang 26 and Angelica 27. Ms. Yang led her away from the tent, so they could speak privately. Angelica seemed reticent, surprised by the sudden appearance of the two health officers. “You’re not in trouble,” Ms. Yang said, before revealing the results of her blood test.
Angelica had never heard of syphilis.
“Have you been to prenatal care?”
Angelica shook her head. The local clinic had referred her to an obstetrician in Hanford, a 30-minute drive away. She had no car. She also mentioned that she didn’t intend to raise her baby; her two oldest children lived with her mother, and this one likely would, too.
Ms. Yang pulled out the CDC cards, showing them to Angelica and asking if she had experienced any of the symptoms illustrated. No, Angelica said, her lips pursed with disgust.
“Right now you still feel healthy, but this bacteria is still in your body,” Ms. Yang pressed. “You need to get the infection treated to prevent further health complications to yourself and your baby.”
The community clinic was just across the street. “Can we walk you over to the clinic and make sure you get seen so we can get this taken care of?”
Angelica demurred. She said she hadn’t showered for a week and wanted to wash up first. She said she’d go later.
Ms. Yang tried once more to extract a promise: “What time do you think you’ll go?”
“Today, for sure.”
Syphilis is called The Great Imitator: It can look like any number of diseases. In its first stage, the only evidence of infection is a painless sore at the bacteria’s point of entry. Weeks later, as the bacteria multiplies, skin rashes bloom on the palms of the hands and bottoms of the feet. Other traits of this stage include fever, headaches, muscle aches, sore throat, and fatigue. These symptoms eventually disappear and the patient progresses into the latent phase, which betrays no external signs. But if left untreated, after a decade or more, syphilis will reemerge in up to 30% of patients, capable of wreaking horror on a wide range of organ systems. Dr. Marion Sims, president of the American Medical Association in 1876, called it a “terrible scourge, which begins with lamb-like mildness and ends with lion-like rage that ruthlessly destroys everything in its way.”
The corkscrew-shaped bacteria can infiltrate the nervous system at any stage of the infection. Ms. Yang is haunted by her memory of interviewing a young man whose dementia was so severe that he didn’t know why he was in the hospital or how old he was. And regardless of symptoms or stage, the bacteria can penetrate the placenta to infect a fetus. Even in these cases the infection is unpredictable: Many babies are born with normal physical features, but others can have deformed bones or damaged brains, and they can struggle to hear, see, or breathe.
From its earliest days, syphilis has been shrouded in stigma. The first recorded outbreak was in the late 15th century, when Charles VIII led the French army to invade Naples. Italian physicians described French soldiers covered with pustules, dying from a sexually transmitted disease. As the affliction spread, Italians called it the French Disease. The French blamed the Neopolitans. It was also called the German, Polish, or Spanish disease, depending on which neighbor one wanted to blame. Even its name bears the taint of divine judgement: It comes from a 16th-century poem that tells of a shepherd, Syphilus, who offended the god Apollo and was punished with a hideous disease.
By 1937 in America, when former Surgeon General Thomas Parran wrote the book “Shadow on the Land,” he estimated some 680,000 people were under treatment for syphilis; about 60,000 babies were being born annually with congenital syphilis. There was no cure, and the stigma was so strong that public health officials feared even properly documenting cases.
Thanks to Dr. Parran’s ardent advocacy, Congress in 1938 passed the National Venereal Disease Control Act, which created grants for states to set up clinics and support testing and treatment. Other than a short-lived funding effort during World War I, this was the first coordinated federal push to respond to the disease.
Around the same time, the Public Health Service launched an effort to record the natural history of syphilis. Situated in Tuskegee, Ala., the infamous study recruited 600 black men. By the early 1940s, penicillin became widely available and was found to be a reliable cure, but the treatment was withheld from the study participants. Outrage over the ethical violations would cast a stain across syphilis research for decades to come and fuel generations of mistrust in the medical system among Black Americans that continues to this day.
With the introduction of penicillin, cases began to plummet. Twice, the CDC has announced efforts to wipe out the disease — once in the 1960s and again in 1999.
In the latest effort, the CDC announced that the United States had “a unique opportunity to eliminate syphilis within its borders,” thanks to historically low rates, with 80% of counties reporting zero cases. The concentration of cases in the South “identifies communities in which there is a fundamental failure of public health capacity,” the agency noted, adding that elimination — which it defined as fewer than 1,000 cases a year — would “decrease one of our most glaring racial disparities in health.”
Two years after the campaign began, cases started climbing, first among gay men and later, heterosexuals. Cases in women started accelerating in 2013, followed shortly by increasing numbers of babies born with syphilis.The reasons for failure are complex; people relaxed safer sex practices after the advent of potent HIV combination therapies, increased methamphetamine use drove riskier behavior and an explosion of online dating made it hard to track and test sexual partners, according to Dr. Ina Park, medical director of the California Prevention Training Center at the University of California San Francisco.
But federal and state public health efforts were hamstrung from the get-go. In 1999, the CDC said it would need about $35 million to $39 million in new federal funds annually for at least five years to eliminate syphilis. The agency got less than half of what it asked for, according to Jo Valentine, former program coordinator of the CDC’s Syphilis Elimination Effort. As cases rose, the CDC modified its goals in 2006 from 0.4 primary and secondary syphilis cases per 100,000 in population to 2.2 cases per 100,000. By 2013, as elimination seemed less and less viable, the CDC changed its focus to ending congenital syphilis only.
Since then, funding has remained anemic. From 2015 to 2020, the CDC’s budget for preventing sexually transmitted infections grew by 2.2%. Taking inflation into account, that’s a 7.4% reduction in purchasing power. In the same period, cases of syphilis, gonorrhea and chlamydia — the three STDs that have federally funded control programs — increased by nearly 30%.
“We have a long history of nearly eradicating something, then changing our attention, and seeing a resurgence in numbers,” said David Harvey, executive director of the National Coalition of STD Directors. “We have more congenital syphilis cases today in America than we ever had pediatric AIDS at the height of the AIDS epidemic. It’s heartbreaking.”
Adriane Casalotti, chief of government and public affairs at the National Association of County and City Health Officials, warns that the United States should not be surprised to see case counts continue to climb. “The bugs don’t go away,” she said. “They’re just waiting for the next opportunity, when you’re not paying attention.”
Ms. Yang waited until the end of the day, then called the clinic to see if Angelica had gone for her shot. She had not. Ms. Yang would have to block off another half day to visit Huron again, but she had three dozen other cases to deal with.
States in the South and West have seen the highest syphilis rates in recent years. In 2017, 64 babies in Fresno County were born with syphilis at a rate of 440 babies per 100,000 live births — about 19 times the national rate. While the county had managed to lower case counts in the two years that followed, the pandemic threatened to unravel that progress, forcing STD staffers to do COVID-19 contact tracing, pausing field visits to find infected people, and scaring patients from seeking care. Ms. Yang’s colleague handled three cases of stillbirth in 2020; in each, the woman was never diagnosed with syphilis because she feared catching the coronavirus and skipped prenatal care.
Ms. Yang, whose caseload peaked at 70 during a COVID-19 surge, knew she would not be able handle them all as thoroughly as she’d like to. “When I was being mentored by another investigator, he said: ‘You’re not a superhero. You can’t save everybody,’” she said. She prioritizes men who have sex with men, because there’s a higher prevalence of syphilis in that population, and pregnant people, because of the horrific consequences for babies.
The job of a disease intervention specialist isn’t for everyone: It means meeting patients whenever and wherever they are available — in the mop closet of a bus station, in a quiet parking lot — to inform them about the disease, to extract names of sex partners and to encourage treatment. Patients are often reluctant to talk. They can get belligerent, upset that “the government” has their personal information or shattered at the thought that a partner is likely cheating on them. Salaries typically start in the low $40,000s.
Jena Adams, Ms. Yang’s supervisor, has eight investigators working on HIV and syphilis. In the middle of 2020, she lost two and replaced them only recently. “It’s been exhausting,” Ms. Adams said. She has only one specialist who is trained to take blood samples in the field, crucial for guaranteeing that the partners of those who test positive for syphilis also get tested. Ms. Adams wants to get phlebotomy training for the rest of her staff, but it’s $2,000 per person. The department also doesn’t have anyone who can administer penicillin injections in the field; that would have been key when Ms. Yang met Angelica. For a while, a nurse who worked in the tuberculosis program would ride along to give penicillin shots on a volunteer basis. Then he, too, left the health department.
Much of the resources in public health trickle down from the CDC, which distributes money to states, which then parcel it out to counties. The CDC gets its budget from Congress, which tells the agency, by line item, exactly how much money it can spend to fight a disease or virus, in an uncommonly specific manner not seen in many other agencies. The decisions are often politically driven and can be detached from actual health needs.
When the House and Senate appropriations committees meet to decide how much the CDC will get for each line item, they are barraged by lobbyists for individual disease interests. Stephanie Arnold Pang, senior director of policy and government relations at the National Coalition of STD Directors, can pick out the groups by sight: breast cancer wears pink, Alzheimer’s goes in purple, multiple sclerosis comes in orange, HIV in red. STD prevention advocates, like herself, don a green ribbon, but they’re far outnumbered.
And unlike diseases that might already be familiar to lawmakers, or have patient and family spokespeople who can tell their own powerful stories, syphilis doesn’t have many willing poster children. “Congressmen don’t wake up one day and say, ‘Oh hey, there’s congenital syphilis in my jurisdiction.’ You have to raise awareness,” Arnold Pang said. It can be hard jockeying for a meeting. “Some offices might say, ‘I don’t have time for you because we’ve just seen HIV.’ ... Sometimes, it feels like you’re talking into a void.”
The consequences of the political nature of public health funding have become more obvious during the coronavirus pandemic. The 2014 Ebola epidemic was seen as a “global wakeup call” that the world wasn’t prepared for a major pandemic, yet in 2018, the CDC scaled back its epidemic prevention work as money ran out. “If you’ve got to choose between Alzheimer’s research and stopping an outbreak that may not happen? Stopping an outbreak that might not happen doesn’t do well,” said Dr. Frieden, the former CDC director. “The CDC needs to have more money and more flexible money. Otherwise, we’re going to be in this situation long term.”
In May 2021, President Joe Biden’s administration announced it would set aside $7.4 billion over the next five years to hire and train public health workers, including $1.1 billion for more disease intervention specialists like Ms. Yang. Public health officials are thrilled to have the chance to expand their workforce, but some worry the time horizon may be too short. “We’ve seen this movie before, right?” Dr. Frieden said. “Everyone gets concerned when there’s an outbreak, and when that outbreak stops, the headlines stop, and an economic downturn happens, the budget gets cut.”
Fresno’s STD clinic was shuttered in 2010 amid the Great Recession. Many others have vanished since the passage of the Affordable Care Act. Health leaders thought “by magically beefing up the primary care system, that we would do a better job of catching STIs and treating them,” said Mr. Harvey, the executive director of the National Coalition of STD Directors. That hasn’t worked out; people want access to anonymous services, and primary care doctors often don’t have STDs top of mind. The coalition is lobbying Congress for funding to support STD clinical services, proposing a three-year demonstration project funded at $600 million.
It’s one of Ms. Adams’ dreams to see Fresno’s STD clinic restored as it was. “You could come in for an HIV test and get other STDs checked,” she said. “And if a patient is positive, you can give a first injection on the spot.”
On Aug. 12, Ms. Yang set out for Huron again, speeding past groves of almond trees and fields of grapes in the department’s white Chevy Cruze. She brought along a colleague, Jorge Sevilla, who had recently transferred to the STD program from COVID-19 contact tracing. Ms. Yang was anxious to find Angelica again. “She’s probably in her second trimester now,” she said.
They found her outside of a pale yellow house a few blocks from the homeless encampment; the owner was letting her stay in a shed tucked in the corner of the dirt yard. This time, it was evident that she was pregnant. Ms. Yang noted that Angelica was wearing a wig; hair loss is a symptom of syphilis.
“Do you remember me?” Ms. Yang asked.
Angelica nodded. She didn’t seem surprised to see Ms. Yang again. (I came along, and Mr. Sevilla explained who I was and that I was writing about syphilis and the people affected by it. Angelica signed a release for me to report about her case, and she said she had no problem with me writing about her or even using her full name. ProPublica chose to only print her first name.)
“How are you doing? How’s the baby?”
“Bien.”
“So the last time we talked, we were going to have you go to United Healthcare Center to get treatment. Have you gone since?”
Angelica shook her head.
“We brought some gift cards...” Mr. Sevilla started in Spanish. The department uses them as incentives for completing injections. But Angelica was already shaking her head. The nearest Walmart was the next town over.
Ms. Yang turned to her partner. “Tell her: So the reason why we’re coming out here again is because we really need her to go in for treatment. ... We really are concerned for the baby’s health especially since she’s had the infection for quite a while.”
Angelica listened while Mr. Sevilla interpreted, her eyes on the ground. Then she looked up. “Orita?” she asked. Right now?
“I’ll walk with you,” Ms. Yang offered. Angelica shook her head. “She said she wants to shower first before she goes over there,” Mr. Sevilla said.
Ms. Yang made a face. “She said that to me last time.” Ms. Yang offered to wait, but Angelica didn’t want the health officers to linger by the house. She said she would meet them by the clinic in 15 minutes.
Ms. Yang was reluctant to let her go but again had no other option. She and Mr. Sevilla drove to the clinic, then stood on the corner of the parking lot, staring down the road.
Talk to the pediatricians, obstetricians, and families on the front lines of the congenital syphilis surge and it becomes clear why Ms. Yang and others are trying so desperately to prevent cases. Dr. J. B. Cantey, associate professor in pediatrics at UT Health San Antonio, remembers a baby girl born at 25 weeks gestation who weighed a pound and a half. Syphilis had spread through her bones and lungs. She spent five months in the neonatal intensive care unit, breathing through a ventilator, and was still eating through a tube when she was discharged.
Then, there are the miscarriages, the stillbirths and the inconsolable parents. Dr. Irene Stafford, an associate professor and maternal-fetal medicine specialist at UT Health in Houston, cannot forget a patient who came in at 36 weeks for a routine checkup, pregnant with her first child. Dr. Stafford realized that there was no heartbeat. “She could see on my face that something was really wrong,” Dr. Stafford recalled. She had to let the patient know that syphilis had killed her baby. “She was hysterical, just bawling,” Dr. Stafford said. “I’ve seen people’s families ripped apart and I’ve seen beautiful babies die.” Fewer than 10% of patients who experience a stillbirth are tested for syphilis, suggesting that cases are underdiagnosed.
A Texas grandmother named Solidad Odunuga offers a glimpse into what the future could hold for Angelica’s mother, who may wind up raising her baby.
In February of last year, Ms. Odunuga got a call from the Lyndon B. Johnson Hospital in Houston. A nurse told her that her daughter was about to give birth and that child protective services had been called. Ms. Odunuga had lost contact with her daughter, who struggled with homelessness and substance abuse. She arrived in time to see her grandson delivered, premature at 30 weeks old, weighing 2.7 pounds. He tested positive for syphilis.
When a child protective worker asked Ms. Odunuga to take custody of the infant, she felt a wave of dread. “I was in denial,” she recalled. “I did not plan to be a mom again.” The baby’s medical problems were daunting: “Global developmental delays ... concerns for visual impairments ... high risk of cerebral palsy,” read a note from the doctor at the time.
Still, Ms. Odunuga visited her grandson every day for three months, driving to the NICU from her job at the University of Houston. “I’d put him in my shirt to keep him warm and hold him there.” She fell in love. She named him Emmanuel.
Once Emmanuel was discharged, Ms. Odunuga realized she had no choice but to quit her job. While Medicaid covered the costs of Emmanuel’s treatment, it was on her to care for him. From infancy, Emmanuel’s life has been a whirlwind of constant therapy. Today, at 20 months old, Odunuga brings him to physical, occupational, speech, and developmental therapy, each a different appointment on a different day of the week.
Emmanuel has thrived beyond what his doctors predicted, toddling so fast that Ms. Odunuga can’t look away for a minute and beaming as he waves his favorite toy phone. Yet he still suffers from gagging issues, which means Ms. Odunuga can’t feed him any solid foods. Liquid gets into his lungs when he aspirates; it has led to pneumonia three times. Emmanuel has a special stroller that helps keep his head in a position that won’t aggravate his persistent reflux, but Odunuga said she still has to pull over on the side of the road sometimes when she hears him projectile vomiting from the backseat.
The days are endless. Once she puts Emmanuel to bed, Ms. Odunuga starts planning the next day’s appointments. “I’ve had to cry alone, scream out alone,” she said. “Sometimes I wake up and think, Is this real? And then I hear him in the next room.”
Putting aside the challenge of eliminating syphilis entirely, everyone agrees it’s both doable and necessary to prevent newborn cases. “There was a crisis in perinatal HIV almost 30 years ago and people stood up and said this is not OK — it’s not acceptable for babies to be born in that condition. ... [We] brought it down from 1,700 babies born each year with perinatal HIV to less than 40 per year today,” said Virginia Bowen, an epidemiologist at the CDC. “Now here we are with a slightly different condition. We can also stand up and say, ‘This is not acceptable.’” Belarus, Bermuda, Cuba, Malaysia, Thailand, and Sri Lanka are among countries recognized by the World Health Organization for eliminating congenital syphilis.
Success starts with filling gaps across the health care system.
For almost a century, public health experts have advocated for testing pregnant patients more than once for syphilis in order to catch the infection. But policies nationwide still don’t reflect this best practice. Six states have no prenatal screening requirement at all. Even in states that require three tests, public health officials say that many physicians aren’t aware of the requirements. Dr. Stafford, the maternal-fetal medicine specialist in Houston, says she’s tired of hearing her own peers in medicine tell her, “Oh, syphilis is a problem?”
It costs public health departments less than 25 cents a dose to buy penicillin, but for a private practice, it’s more than $1,000, according to Dr. Park of the University of California San Francisco. “There’s no incentive for a private physician to stock a dose that could expire before it’s used, so they often don’t have it. So a woman comes in, they say, ‘We’ll send you to the emergency department or health department to get it,’ then [the patients] don’t show up.”
A vaccine would be invaluable for preventing spread among people at high risk for reinfection. But there is none. Scientists only recently figured out how to grow the bacteria in the lab, prompting grants from the National Institutes of Health to fund research into a vaccine. Dr. Justin Radolf, a researcher at the University of Connecticut School of Medicine, said he hopes his team will have a vaccine candidate by the end of its five-year grant. But it’ll likely take years more to find a manufacturer and run human trials.
Public health agencies also need to recognize that many of the hurdles to getting pregnant people treated involve access to care, economic stability, safe housing and transportation. In Fresno, Ms. Adams has been working on ways her department can collaborate with mental health services. Recently, one of her disease intervention specialists managed to get a pregnant woman treated with penicillin shots and, at the patient’s request, connected her with an addiction treatment center.
Gaining a patient’s cooperation means seeing them as complex humans instead of just a case to solve. “There may be past traumas with the health care system,” said Cynthia Deverson, project manager of the Houston Fetal Infant Morbidity Review. “There’s the fear of being discovered if she’s doing something illegal to survive. ... She may need to be in a certain place at a certain time so she can get something to eat, or maybe it’s the only time of the day that’s safe for her to sleep. They’re not going to tell you that. Yes, they understand there’s a problem, but it’s not an immediate threat, maybe they don’t feel bad yet, so obviously this is not urgent. ...
“What helps to gain trust is consistency,” she said. “Literally, it’s seeing that [disease specialist] constantly, daily. ... The woman can see that you’re not going to harm her, you’re saying, ‘I’m here at this time if you need me.’”
Ms. Yang stood outside the clinic, waiting for Angelica to show up, baking in the 90-degree heat. Her feelings ranged from irritation — Why didn’t she just go? I’d have more energy for other cases — to an appreciation for the parts of Angelica’s story that she didn’t know — She’s in survival mode. I need to be more patient.
Fifteen minutes ticked by, then 20.
“OK,” Ms. Yang announced. “We’re going back.”
She asked Sevilla if he would be OK if they drove Angelica to the clinic; they technically weren’t supposed to because of coronavirus precautions, but Ms. Yang wasn’t sure she could convince Angelica to walk. Mr. Sevilla gave her the thumbs up.
When they pulled up, they saw Angelica sitting in the backyard, chatting with a friend. She now wore a fresh T-shirt and had shoes on her feet. Angelica sat silently in the back seat as Ms. Yang drove to the clinic. A few minutes later, they pulled up to the parking lot.
Finally, Ms. Yang thought. We got her here.
The clinic was packed with people waiting for COVID-19 tests and vaccinations. A worker there had previously told Ms. Yang that a walk-in would be fine, but a receptionist now said they were too busy to treat Angelica. She would have to return.
Ms. Yang felt a surge of frustration, sensing that her hard-fought opportunity was slipping away. She tried to talk to the nurse supervisor, but he wasn’t available. She tried to leave the gift cards at the office to reward Angelica if she came, but the receptionist said she couldn’t hold them. While Ms. Yang negotiated, Mr. Sevilla sat with Angelica in the car, waiting.
Finally, Ms. Yang accepted this was yet another thing she couldn’t control.
She drove Angelica back to the yellow house. As they arrived, she tried once more to impress on her just how important it was to get treated, asking Mr. Sevilla to interpret. “We don’t want it to get any more serious, because she can go blind, she could go deaf, she could lose her baby.”
Angelica already had the door halfway open.
“So on a scale from one to 10, how important is this to get treated?” Ms. Yang asked.
“Ten,” Angelica said. Ms. Yang reminded her of the appointment that afternoon. Then Angelica stepped out and returned to the dusty yard.
Ms. Yang lingered for a moment, watching Angelica go. Then she turned the car back onto the highway and set off toward Fresno, knowing, already, that she’d be back.
Postscript: A reporter visited Huron twice more in the months that followed, including once independently to try to interview Angelica, but she wasn’t in town. Ms. Yang has visited Huron twice more as well — six times in total thus far. In October, a couple of men at the yellow house said Angelica was still in town, still pregnant. Ms. Yang and Mr. Sevilla spent an hour driving around, talking to residents, hoping to catch Angelica. But she was nowhere to be found.
ProPublica is a nonprofit newsroom that investigates abuses of power. Sign up to receive their biggest stories as soon as they’re published.
Q&A: Meeting the challenge of giving COVID vaccines to younger kids
This news organization spoke to several pediatric experts to get answers.
More than 6 million children and adolescents (up to age 18 years) in the United States have been infected with SARS-CoV-2. Children represent about 17% of all cases, and an estimated 0.1%-2% of infected children end up hospitalized, according to Oct. 28 data from the American Academy of Pediatrics.
Physicians and other health care practitioners are gearing up for what could be an influx of patients. “Pediatricians are standing by to talk with families about the vaccine and to administer the vaccine to children as soon as possible,” Lee Savio Beers, MD, FAAP, president of the AAP, said in a statement.
In this Q&A, this news organization asked for additional advice from Sara “Sally” Goza, MD, a pediatrician in Fayetteville, Georgia, and immediate past president of the AAP; Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine and codirector of the Texas Children’s Hospital Center for Vaccine Development, both in Houston; and Danielle M. Zerr, MD, professor and chief of the division of pediatric infectious disease at the University of Washington, Seattle, and medical director of infection prevention at Seattle Children’s Hospital.
Q: How are smaller pediatric practices and solo practitioners going to handle the additional vaccinations?
Dr. Goza: It’s a scheduling challenge with this rollout and all the people who want it and want it right now. They’re going to want it this week.
I’ve actually had some children asking their moms: “When can I get it? When can I get it?” It’s been very interesting – they are chomping at the bit.
If I give the vaccine to a patient this week, in 3 weeks the second dose will be right around Thanksgiving. No one in my office is going to want to be here to give the shot on Thanksgiving, and no patient is going to want to come in on Thanksgiving weekend. So I’m trying to delay those parents – saying, let’s do it next week. That way we’re not messing up a holiday.
Children are going to need two doses, and they won’t be fully protected until 2 weeks after their second dose. So they won’t get full protection for Thanksgiving, but they will have full protection for Christmas.
I know there are a lot of pediatricians who have preordered the vaccine. I know in our office they sent us an email ... to let us know our vaccines are being shipped. So I think a lot of pediatricians are going to have the vaccine.
Q: How should pediatricians counsel parents who are fearful or hesitant?
Dr. Hotez: It’s important to emphasize the severity of the 2021 summer Delta epidemic in children. We need to get beyond this false narrative that COVID only produces a mild disease in children. It’s caused thousands of pediatric hospitalizations, not to mention long COVID.
Dr. Zerr: It is key to find out what concerns parents have and then focus on answering their specific questions. It is helpful to emphasize the safety and efficacy of the vaccine and to explain the rigorous processes that the vaccine went through to receive Food and Drug Administration approval.
Q: How should pediatricians counter any misinformation/disinformation out there about the COVID-19 vaccines?
Dr. Goza: The most important thing is not to discount what they are saying. Don’t say: “That’s crazy” or “That’s not true.” Don’t roll your eyes and say: “Really, you’re going to believe all that?”
Instead, have a conversation with them about why we think that is not true, or why we know that’s not true. We really have to have that relationship and ask: “Well, what are your concerns?” And then really counter (any misinformation) with facts, with science, and based on your experience.
Q: Do the data presented to the FDA and the CDC about the safety and effectiveness of the COVID-19 vaccine for 5- to 11-year-olds seem robust to you?
Dr. Zerr: Yes, and data collection will be ongoing.
Dr. Hotez: I’ve only seen what’s publicly available so far, and it seems to support moving forward with emergency use authorization. The only shortfall is the size, roughly 2,200 children, which would not be of sufficient size to detect a rare safety signal.
Q: Do previous controversies around pediatric vaccines (for example, the MMR vaccine and autism) give pediatricians some background and experience so they can address any pushback on the COVID-19 vaccines?
Dr. Goza: Pediatricians have been dealing with vaccine hesitancy for a while now, ever since the MMR and autism controversy started. Even before then, there were certain groups of people who didn’t want vaccines.
We’ve really worked hard at helping teach pediatricians how to deal with the misinformation, how to counter it, and how to help parents understand the vaccines are safe and effective – and that they save lives.
That (experience) will help us in some ways. Unfortunately, there is more misinformation out there – there is almost a concerted effort on misinformation. It’s big.
Pediatricians will do everything we can, but we need help countering it. We need the misinformation to quit getting spread on social media. We can talk one on one with patients and families, but if all they are hearing on social media is the misinformation, it’s really hard.
Q: Are pediatricians, especially solo practitioners or pediatricians at smaller practices, going to face challenges with multidose vials and not wasting vaccine product?
Dr. Goza: I’m at a small practice. We have 3.5 FTEs (full-time equivalents) of MDs and three FTEs of nurse practitioners. So we’re not that big – about six providers.
You know, it is a challenge. We’re not going to buy the super-duper freezer, and we’re not going to be able to store these vaccines for a long period of time.
So when we order, we need smaller amounts. For the 12- to 18-year-olds, [maximum storage] was 45 days. Now for the 5- to 11-year-olds, we’re going to be able to store the vaccine in the refrigerator for 10 weeks, which gives us more leeway there.
We try to do all of vaccinations on 1 day, so we know how many people are coming in, and we are not going to waste too many doses.
Our Department of Public Health in Georgia has said: “We want these vaccines in the arms of kids, and if you have to waste some doses, don’t worry about it.” But it’s a 10-dose vial. It’s going to be hard for me to open it up for one child. I just don’t like wasting anything like this.
Our main goal is to get this vaccine in to the arms of children whose parents want it.
Q: What are some additional sources of information for pediatricians?
Dr. Zerr: There are a lot of great resources on vaccine hesitancy from reputable sources, including these from the CDC and from the National Academies of Sciences, Engineering, and Medicine:
- Building Confidence With OVID-19 Vaccines
- How to Talk With Parents About COVID-19 Vaccination
- Strategies for Building Confidence in the COVID-19 Vaccines
- Communication Strategies for Building Confidence in COVID-19 Vaccines: Addressing Variants and Childhood Vaccinations
A version of this article first appeared on Medscape.com.
This news organization spoke to several pediatric experts to get answers.
More than 6 million children and adolescents (up to age 18 years) in the United States have been infected with SARS-CoV-2. Children represent about 17% of all cases, and an estimated 0.1%-2% of infected children end up hospitalized, according to Oct. 28 data from the American Academy of Pediatrics.
Physicians and other health care practitioners are gearing up for what could be an influx of patients. “Pediatricians are standing by to talk with families about the vaccine and to administer the vaccine to children as soon as possible,” Lee Savio Beers, MD, FAAP, president of the AAP, said in a statement.
In this Q&A, this news organization asked for additional advice from Sara “Sally” Goza, MD, a pediatrician in Fayetteville, Georgia, and immediate past president of the AAP; Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine and codirector of the Texas Children’s Hospital Center for Vaccine Development, both in Houston; and Danielle M. Zerr, MD, professor and chief of the division of pediatric infectious disease at the University of Washington, Seattle, and medical director of infection prevention at Seattle Children’s Hospital.
Q: How are smaller pediatric practices and solo practitioners going to handle the additional vaccinations?
Dr. Goza: It’s a scheduling challenge with this rollout and all the people who want it and want it right now. They’re going to want it this week.
I’ve actually had some children asking their moms: “When can I get it? When can I get it?” It’s been very interesting – they are chomping at the bit.
If I give the vaccine to a patient this week, in 3 weeks the second dose will be right around Thanksgiving. No one in my office is going to want to be here to give the shot on Thanksgiving, and no patient is going to want to come in on Thanksgiving weekend. So I’m trying to delay those parents – saying, let’s do it next week. That way we’re not messing up a holiday.
Children are going to need two doses, and they won’t be fully protected until 2 weeks after their second dose. So they won’t get full protection for Thanksgiving, but they will have full protection for Christmas.
I know there are a lot of pediatricians who have preordered the vaccine. I know in our office they sent us an email ... to let us know our vaccines are being shipped. So I think a lot of pediatricians are going to have the vaccine.
Q: How should pediatricians counsel parents who are fearful or hesitant?
Dr. Hotez: It’s important to emphasize the severity of the 2021 summer Delta epidemic in children. We need to get beyond this false narrative that COVID only produces a mild disease in children. It’s caused thousands of pediatric hospitalizations, not to mention long COVID.
Dr. Zerr: It is key to find out what concerns parents have and then focus on answering their specific questions. It is helpful to emphasize the safety and efficacy of the vaccine and to explain the rigorous processes that the vaccine went through to receive Food and Drug Administration approval.
Q: How should pediatricians counter any misinformation/disinformation out there about the COVID-19 vaccines?
Dr. Goza: The most important thing is not to discount what they are saying. Don’t say: “That’s crazy” or “That’s not true.” Don’t roll your eyes and say: “Really, you’re going to believe all that?”
Instead, have a conversation with them about why we think that is not true, or why we know that’s not true. We really have to have that relationship and ask: “Well, what are your concerns?” And then really counter (any misinformation) with facts, with science, and based on your experience.
Q: Do the data presented to the FDA and the CDC about the safety and effectiveness of the COVID-19 vaccine for 5- to 11-year-olds seem robust to you?
Dr. Zerr: Yes, and data collection will be ongoing.
Dr. Hotez: I’ve only seen what’s publicly available so far, and it seems to support moving forward with emergency use authorization. The only shortfall is the size, roughly 2,200 children, which would not be of sufficient size to detect a rare safety signal.
Q: Do previous controversies around pediatric vaccines (for example, the MMR vaccine and autism) give pediatricians some background and experience so they can address any pushback on the COVID-19 vaccines?
Dr. Goza: Pediatricians have been dealing with vaccine hesitancy for a while now, ever since the MMR and autism controversy started. Even before then, there were certain groups of people who didn’t want vaccines.
We’ve really worked hard at helping teach pediatricians how to deal with the misinformation, how to counter it, and how to help parents understand the vaccines are safe and effective – and that they save lives.
That (experience) will help us in some ways. Unfortunately, there is more misinformation out there – there is almost a concerted effort on misinformation. It’s big.
Pediatricians will do everything we can, but we need help countering it. We need the misinformation to quit getting spread on social media. We can talk one on one with patients and families, but if all they are hearing on social media is the misinformation, it’s really hard.
Q: Are pediatricians, especially solo practitioners or pediatricians at smaller practices, going to face challenges with multidose vials and not wasting vaccine product?
Dr. Goza: I’m at a small practice. We have 3.5 FTEs (full-time equivalents) of MDs and three FTEs of nurse practitioners. So we’re not that big – about six providers.
You know, it is a challenge. We’re not going to buy the super-duper freezer, and we’re not going to be able to store these vaccines for a long period of time.
So when we order, we need smaller amounts. For the 12- to 18-year-olds, [maximum storage] was 45 days. Now for the 5- to 11-year-olds, we’re going to be able to store the vaccine in the refrigerator for 10 weeks, which gives us more leeway there.
We try to do all of vaccinations on 1 day, so we know how many people are coming in, and we are not going to waste too many doses.
Our Department of Public Health in Georgia has said: “We want these vaccines in the arms of kids, and if you have to waste some doses, don’t worry about it.” But it’s a 10-dose vial. It’s going to be hard for me to open it up for one child. I just don’t like wasting anything like this.
Our main goal is to get this vaccine in to the arms of children whose parents want it.
Q: What are some additional sources of information for pediatricians?
Dr. Zerr: There are a lot of great resources on vaccine hesitancy from reputable sources, including these from the CDC and from the National Academies of Sciences, Engineering, and Medicine:
- Building Confidence With OVID-19 Vaccines
- How to Talk With Parents About COVID-19 Vaccination
- Strategies for Building Confidence in the COVID-19 Vaccines
- Communication Strategies for Building Confidence in COVID-19 Vaccines: Addressing Variants and Childhood Vaccinations
A version of this article first appeared on Medscape.com.
This news organization spoke to several pediatric experts to get answers.
More than 6 million children and adolescents (up to age 18 years) in the United States have been infected with SARS-CoV-2. Children represent about 17% of all cases, and an estimated 0.1%-2% of infected children end up hospitalized, according to Oct. 28 data from the American Academy of Pediatrics.
Physicians and other health care practitioners are gearing up for what could be an influx of patients. “Pediatricians are standing by to talk with families about the vaccine and to administer the vaccine to children as soon as possible,” Lee Savio Beers, MD, FAAP, president of the AAP, said in a statement.
In this Q&A, this news organization asked for additional advice from Sara “Sally” Goza, MD, a pediatrician in Fayetteville, Georgia, and immediate past president of the AAP; Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine and codirector of the Texas Children’s Hospital Center for Vaccine Development, both in Houston; and Danielle M. Zerr, MD, professor and chief of the division of pediatric infectious disease at the University of Washington, Seattle, and medical director of infection prevention at Seattle Children’s Hospital.
Q: How are smaller pediatric practices and solo practitioners going to handle the additional vaccinations?
Dr. Goza: It’s a scheduling challenge with this rollout and all the people who want it and want it right now. They’re going to want it this week.
I’ve actually had some children asking their moms: “When can I get it? When can I get it?” It’s been very interesting – they are chomping at the bit.
If I give the vaccine to a patient this week, in 3 weeks the second dose will be right around Thanksgiving. No one in my office is going to want to be here to give the shot on Thanksgiving, and no patient is going to want to come in on Thanksgiving weekend. So I’m trying to delay those parents – saying, let’s do it next week. That way we’re not messing up a holiday.
Children are going to need two doses, and they won’t be fully protected until 2 weeks after their second dose. So they won’t get full protection for Thanksgiving, but they will have full protection for Christmas.
I know there are a lot of pediatricians who have preordered the vaccine. I know in our office they sent us an email ... to let us know our vaccines are being shipped. So I think a lot of pediatricians are going to have the vaccine.
Q: How should pediatricians counsel parents who are fearful or hesitant?
Dr. Hotez: It’s important to emphasize the severity of the 2021 summer Delta epidemic in children. We need to get beyond this false narrative that COVID only produces a mild disease in children. It’s caused thousands of pediatric hospitalizations, not to mention long COVID.
Dr. Zerr: It is key to find out what concerns parents have and then focus on answering their specific questions. It is helpful to emphasize the safety and efficacy of the vaccine and to explain the rigorous processes that the vaccine went through to receive Food and Drug Administration approval.
Q: How should pediatricians counter any misinformation/disinformation out there about the COVID-19 vaccines?
Dr. Goza: The most important thing is not to discount what they are saying. Don’t say: “That’s crazy” or “That’s not true.” Don’t roll your eyes and say: “Really, you’re going to believe all that?”
Instead, have a conversation with them about why we think that is not true, or why we know that’s not true. We really have to have that relationship and ask: “Well, what are your concerns?” And then really counter (any misinformation) with facts, with science, and based on your experience.
Q: Do the data presented to the FDA and the CDC about the safety and effectiveness of the COVID-19 vaccine for 5- to 11-year-olds seem robust to you?
Dr. Zerr: Yes, and data collection will be ongoing.
Dr. Hotez: I’ve only seen what’s publicly available so far, and it seems to support moving forward with emergency use authorization. The only shortfall is the size, roughly 2,200 children, which would not be of sufficient size to detect a rare safety signal.
Q: Do previous controversies around pediatric vaccines (for example, the MMR vaccine and autism) give pediatricians some background and experience so they can address any pushback on the COVID-19 vaccines?
Dr. Goza: Pediatricians have been dealing with vaccine hesitancy for a while now, ever since the MMR and autism controversy started. Even before then, there were certain groups of people who didn’t want vaccines.
We’ve really worked hard at helping teach pediatricians how to deal with the misinformation, how to counter it, and how to help parents understand the vaccines are safe and effective – and that they save lives.
That (experience) will help us in some ways. Unfortunately, there is more misinformation out there – there is almost a concerted effort on misinformation. It’s big.
Pediatricians will do everything we can, but we need help countering it. We need the misinformation to quit getting spread on social media. We can talk one on one with patients and families, but if all they are hearing on social media is the misinformation, it’s really hard.
Q: Are pediatricians, especially solo practitioners or pediatricians at smaller practices, going to face challenges with multidose vials and not wasting vaccine product?
Dr. Goza: I’m at a small practice. We have 3.5 FTEs (full-time equivalents) of MDs and three FTEs of nurse practitioners. So we’re not that big – about six providers.
You know, it is a challenge. We’re not going to buy the super-duper freezer, and we’re not going to be able to store these vaccines for a long period of time.
So when we order, we need smaller amounts. For the 12- to 18-year-olds, [maximum storage] was 45 days. Now for the 5- to 11-year-olds, we’re going to be able to store the vaccine in the refrigerator for 10 weeks, which gives us more leeway there.
We try to do all of vaccinations on 1 day, so we know how many people are coming in, and we are not going to waste too many doses.
Our Department of Public Health in Georgia has said: “We want these vaccines in the arms of kids, and if you have to waste some doses, don’t worry about it.” But it’s a 10-dose vial. It’s going to be hard for me to open it up for one child. I just don’t like wasting anything like this.
Our main goal is to get this vaccine in to the arms of children whose parents want it.
Q: What are some additional sources of information for pediatricians?
Dr. Zerr: There are a lot of great resources on vaccine hesitancy from reputable sources, including these from the CDC and from the National Academies of Sciences, Engineering, and Medicine:
- Building Confidence With OVID-19 Vaccines
- How to Talk With Parents About COVID-19 Vaccination
- Strategies for Building Confidence in the COVID-19 Vaccines
- Communication Strategies for Building Confidence in COVID-19 Vaccines: Addressing Variants and Childhood Vaccinations
A version of this article first appeared on Medscape.com.
Can adjunct corticosteroids help in childrens’ eye and throat infections?
Adding anti-inflammatory corticosteroids to antibiotics for certain pediatric throat and ocular infections may have some benefit, according to results from two recent database studies, but their benefit remains unclear.
Using steroids in this setting is a practice many pediatricians consider, although no clear guidance exists.
Drawing on data from a registry of 51 free-standing children’s hospitals in the Pediatric Health Information System (PHIS), and published online in Pediatrics, the analyses looked, respectively, at retro- and parapharyngeal abscesses (RPAs/PPAs) and acute orbital cellulitis.
Throat abscesses
In the first study, pediatrician Pratichi K. Goenka, MD, of Cohen Children’s Medical Center–Northwell Health and an assistant professor at Hofstra University, both in New Hyde Park, N.Y., and colleagues reported on the effect of systemic corticosteroids on several outcomes in RPAs/PPAs in 2,259 well-matched patients. The patients, aged 2 months to 8 years, were treated at 46 hospitals during the period from January 2016 to December 2019.
The data revealed that the 582 (25.8%) who received steroids had a significantly lower rate of surgical drainage, the study’s primary endpoint (odds ratio, 0.28; 95% confidence interval, 0.22-0.36). There was no difference, however, in length of hospital stay (rate ratio, 0.97; 95% CI, 0.92-1.02).
Those in the steroid group also had lower overall hospital costs and were less likely to be given opioid medications for pain. They were, however, more likely to undergo repeat CT imaging and also had a higher 7-day ED revisit rate but no increase in readmission 30 days after discharge: 4% versus 3% in the nonsteroid group (P = .29).
“As hospitalists, we share the care of young children with RPAs and PPAs with our otolaryngology colleagues. The primary therapy is antibiotics but there was no clear guidance on the next step, and the current literature had no answers as to how systemic corticosteroids might impact the care of these children,” Dr. Goenka said in an interview. “So we wanted to leverage the PHIS data to better understand the association with the need for surgery and length of stay. Surgery is painful and often involves IV administration of opioid painkillers. It’s something we may be able to avoid if we can optimize medical treatment.”
Pending results from randomized trials, what immediate impact could these registry findings have? “We hope that physicians will think about the best initial medical treatment plan for these children,” Dr. Goenka said. ”Given these data, I would be more likely to incorporate steroids early on in medical treatment.”
She emphasized, however, that before routine adoption prospective studies are needed to clearly identify which patients will have a strong benefit and which will not benefit. “That is the nuanced discussion that will happen with more prospective work.”
Dr. Goenka and associates explained that the rising incidence of RPAs and PPAs over the past 20 years has been attributed to more cases of tonsillitis because of a shift away from tonsillectomies, as well as the changing epidemiology of methicillin-resistant Staphylococcus aureus.
In an accompanying editorial, Ellen R. Wald, MD, and Jens C. Eickhoff, PhD, of the University of Wisconsin–Madison, stated that the use of corticosteroids in bacterial meningitis is often cited as an example of the benefits of steroids in infection. “The specific rationale for use of corticosteroids is [their] anti-inflammatory effects, which may result in decreases of swelling and/ or edema to facilitate drainage, perfusion, reduction in pain, and healing.”
They cautioned, however, that the pharmacologic effects of steroids are myriad and complicated, and include potential masking of the clinical course of disease, thereby delaying appropriate therapy for unrecognized deterioration, as well potential immunosuppression.
Acute orbital cellulitis
In the second retrospective analysis, a group led by pediatrician Maria Anna Leszczynska, MD, of Johns Hopkins All Children’s Hospital in Baltimore, analyzed a retrospective PHIS cohort of 5,645 children younger than 18 years with a primary diagnosis of orbital cellulitis treated at 51 hospitals from January 2007 to December 2018.
Of these, 1,347 (24%) received steroids, but, contrary to earlier reports, the data showed no reduction in length of stay associated with these drugs after adjustment for age, meningitis, abscess, or vision issues (ebeta, 1.01; 95% CI, 0.97-1.06). Corticosteroid exposure was, however, associated with operative episodes after 2 days’ hospitalization (OR, 2.05; 95% CI, 1.29-3.27) and 30-day readmission (OR, 2.40; 95% CI, 1.52-3.78).
“Among children hospitalized for orbital cellulitis, we did not observe the reduction in LOS [length of stay] for patients prescribed systemic corticosteroids as described previously in the literature,” the authors wrote.
In terms of surgical procedures, 52.0% of corticosteroid recipients versus 14.0% of nonrecipients underwent surgery (P < .001), and more were hospitalized in the pediatric ICU (4.4% vs 2.6%; P < .001).
According to the editorialists: “Both observations suggest that children who received steroids may have been a sicker group of patients.”
Dr. Wald and Dr. Eickhoff pointed out that the effect of steroids is ultimately unclear because of the retrospective study’s inherent potential for bias because of unobserved confounders. Were steroids prescribed more often when children were perceived to be sicker with more severe disease, or did these medications cause worse outcomes?
The authors agreed that the study could not determine causality. “Although we used all available markers of disease severity, there does not exist a validated disease severity clinical score for pediatric orbital cellulitis,” they wrote.
According to Ricardo A. Quinonez, MD, an associate professor of pediatrics and division and service chief of pediatric hospital medicine at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, “orbital cellulitis is a not very common thing in children so we don’t treat many patients with this. But having said that, there is usually some debate among providers about whether to use steroids.”
Some centers use them routinely for central nervous system and eye infections or extensions of sinusitis, he said, but there is variability in the prescribing of corticosteroids. “There’s ongoing discussion as to whether they‘re as helpful in orbital cellulitis as they are in similar conditions,” Dr. Quinonez said in an interview. “At our institution we don’t typically prescribe them – not never but not routinely. Children who are sicker tend to get steroids, as they do in other conditions.”
In the context of PPA as in the first study, he added, “I think the evidence favoring the use of steroids in infections that affect the airway is stronger, and their use is definitely more prevalent in those instances.”
While both PHIS analyses suggested some benefit from steroids, he continued, some children may not benefit and there may be harms. “The evidence is still mostly retrospective and observational with no multicenter randomized controlled data. Without those data the evidence is difficult to interpret and subject to all the biases that observational and retrospective data is subject to and the current evidence should not lead physicians to change their practice until controlled, randomized evidence is available.”
The editorialists concurred with the study authors and Dr. Quinonez that large, controlled, prospective clinical trials are needed to ascertain the effect of steroids and to standardize the approach to diagnosis and management. “Use of administrative databases are not optimal to answer questions related to outcome,” they wrote.
The study by Dr. Goenka and associates received no external funding; the study by Dr. Leszczynska and associates also received no external funding. None of the authors declared potential competing interests. Dr. Quinonez had no competing interests to declare. Dr. Wald and Dr. Eickhoff disclosed no competing interests with regard to their editorial.
Adding anti-inflammatory corticosteroids to antibiotics for certain pediatric throat and ocular infections may have some benefit, according to results from two recent database studies, but their benefit remains unclear.
Using steroids in this setting is a practice many pediatricians consider, although no clear guidance exists.
Drawing on data from a registry of 51 free-standing children’s hospitals in the Pediatric Health Information System (PHIS), and published online in Pediatrics, the analyses looked, respectively, at retro- and parapharyngeal abscesses (RPAs/PPAs) and acute orbital cellulitis.
Throat abscesses
In the first study, pediatrician Pratichi K. Goenka, MD, of Cohen Children’s Medical Center–Northwell Health and an assistant professor at Hofstra University, both in New Hyde Park, N.Y., and colleagues reported on the effect of systemic corticosteroids on several outcomes in RPAs/PPAs in 2,259 well-matched patients. The patients, aged 2 months to 8 years, were treated at 46 hospitals during the period from January 2016 to December 2019.
The data revealed that the 582 (25.8%) who received steroids had a significantly lower rate of surgical drainage, the study’s primary endpoint (odds ratio, 0.28; 95% confidence interval, 0.22-0.36). There was no difference, however, in length of hospital stay (rate ratio, 0.97; 95% CI, 0.92-1.02).
Those in the steroid group also had lower overall hospital costs and were less likely to be given opioid medications for pain. They were, however, more likely to undergo repeat CT imaging and also had a higher 7-day ED revisit rate but no increase in readmission 30 days after discharge: 4% versus 3% in the nonsteroid group (P = .29).
“As hospitalists, we share the care of young children with RPAs and PPAs with our otolaryngology colleagues. The primary therapy is antibiotics but there was no clear guidance on the next step, and the current literature had no answers as to how systemic corticosteroids might impact the care of these children,” Dr. Goenka said in an interview. “So we wanted to leverage the PHIS data to better understand the association with the need for surgery and length of stay. Surgery is painful and often involves IV administration of opioid painkillers. It’s something we may be able to avoid if we can optimize medical treatment.”
Pending results from randomized trials, what immediate impact could these registry findings have? “We hope that physicians will think about the best initial medical treatment plan for these children,” Dr. Goenka said. ”Given these data, I would be more likely to incorporate steroids early on in medical treatment.”
She emphasized, however, that before routine adoption prospective studies are needed to clearly identify which patients will have a strong benefit and which will not benefit. “That is the nuanced discussion that will happen with more prospective work.”
Dr. Goenka and associates explained that the rising incidence of RPAs and PPAs over the past 20 years has been attributed to more cases of tonsillitis because of a shift away from tonsillectomies, as well as the changing epidemiology of methicillin-resistant Staphylococcus aureus.
In an accompanying editorial, Ellen R. Wald, MD, and Jens C. Eickhoff, PhD, of the University of Wisconsin–Madison, stated that the use of corticosteroids in bacterial meningitis is often cited as an example of the benefits of steroids in infection. “The specific rationale for use of corticosteroids is [their] anti-inflammatory effects, which may result in decreases of swelling and/ or edema to facilitate drainage, perfusion, reduction in pain, and healing.”
They cautioned, however, that the pharmacologic effects of steroids are myriad and complicated, and include potential masking of the clinical course of disease, thereby delaying appropriate therapy for unrecognized deterioration, as well potential immunosuppression.
Acute orbital cellulitis
In the second retrospective analysis, a group led by pediatrician Maria Anna Leszczynska, MD, of Johns Hopkins All Children’s Hospital in Baltimore, analyzed a retrospective PHIS cohort of 5,645 children younger than 18 years with a primary diagnosis of orbital cellulitis treated at 51 hospitals from January 2007 to December 2018.
Of these, 1,347 (24%) received steroids, but, contrary to earlier reports, the data showed no reduction in length of stay associated with these drugs after adjustment for age, meningitis, abscess, or vision issues (ebeta, 1.01; 95% CI, 0.97-1.06). Corticosteroid exposure was, however, associated with operative episodes after 2 days’ hospitalization (OR, 2.05; 95% CI, 1.29-3.27) and 30-day readmission (OR, 2.40; 95% CI, 1.52-3.78).
“Among children hospitalized for orbital cellulitis, we did not observe the reduction in LOS [length of stay] for patients prescribed systemic corticosteroids as described previously in the literature,” the authors wrote.
In terms of surgical procedures, 52.0% of corticosteroid recipients versus 14.0% of nonrecipients underwent surgery (P < .001), and more were hospitalized in the pediatric ICU (4.4% vs 2.6%; P < .001).
According to the editorialists: “Both observations suggest that children who received steroids may have been a sicker group of patients.”
Dr. Wald and Dr. Eickhoff pointed out that the effect of steroids is ultimately unclear because of the retrospective study’s inherent potential for bias because of unobserved confounders. Were steroids prescribed more often when children were perceived to be sicker with more severe disease, or did these medications cause worse outcomes?
The authors agreed that the study could not determine causality. “Although we used all available markers of disease severity, there does not exist a validated disease severity clinical score for pediatric orbital cellulitis,” they wrote.
According to Ricardo A. Quinonez, MD, an associate professor of pediatrics and division and service chief of pediatric hospital medicine at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, “orbital cellulitis is a not very common thing in children so we don’t treat many patients with this. But having said that, there is usually some debate among providers about whether to use steroids.”
Some centers use them routinely for central nervous system and eye infections or extensions of sinusitis, he said, but there is variability in the prescribing of corticosteroids. “There’s ongoing discussion as to whether they‘re as helpful in orbital cellulitis as they are in similar conditions,” Dr. Quinonez said in an interview. “At our institution we don’t typically prescribe them – not never but not routinely. Children who are sicker tend to get steroids, as they do in other conditions.”
In the context of PPA as in the first study, he added, “I think the evidence favoring the use of steroids in infections that affect the airway is stronger, and their use is definitely more prevalent in those instances.”
While both PHIS analyses suggested some benefit from steroids, he continued, some children may not benefit and there may be harms. “The evidence is still mostly retrospective and observational with no multicenter randomized controlled data. Without those data the evidence is difficult to interpret and subject to all the biases that observational and retrospective data is subject to and the current evidence should not lead physicians to change their practice until controlled, randomized evidence is available.”
The editorialists concurred with the study authors and Dr. Quinonez that large, controlled, prospective clinical trials are needed to ascertain the effect of steroids and to standardize the approach to diagnosis and management. “Use of administrative databases are not optimal to answer questions related to outcome,” they wrote.
The study by Dr. Goenka and associates received no external funding; the study by Dr. Leszczynska and associates also received no external funding. None of the authors declared potential competing interests. Dr. Quinonez had no competing interests to declare. Dr. Wald and Dr. Eickhoff disclosed no competing interests with regard to their editorial.
Adding anti-inflammatory corticosteroids to antibiotics for certain pediatric throat and ocular infections may have some benefit, according to results from two recent database studies, but their benefit remains unclear.
Using steroids in this setting is a practice many pediatricians consider, although no clear guidance exists.
Drawing on data from a registry of 51 free-standing children’s hospitals in the Pediatric Health Information System (PHIS), and published online in Pediatrics, the analyses looked, respectively, at retro- and parapharyngeal abscesses (RPAs/PPAs) and acute orbital cellulitis.
Throat abscesses
In the first study, pediatrician Pratichi K. Goenka, MD, of Cohen Children’s Medical Center–Northwell Health and an assistant professor at Hofstra University, both in New Hyde Park, N.Y., and colleagues reported on the effect of systemic corticosteroids on several outcomes in RPAs/PPAs in 2,259 well-matched patients. The patients, aged 2 months to 8 years, were treated at 46 hospitals during the period from January 2016 to December 2019.
The data revealed that the 582 (25.8%) who received steroids had a significantly lower rate of surgical drainage, the study’s primary endpoint (odds ratio, 0.28; 95% confidence interval, 0.22-0.36). There was no difference, however, in length of hospital stay (rate ratio, 0.97; 95% CI, 0.92-1.02).
Those in the steroid group also had lower overall hospital costs and were less likely to be given opioid medications for pain. They were, however, more likely to undergo repeat CT imaging and also had a higher 7-day ED revisit rate but no increase in readmission 30 days after discharge: 4% versus 3% in the nonsteroid group (P = .29).
“As hospitalists, we share the care of young children with RPAs and PPAs with our otolaryngology colleagues. The primary therapy is antibiotics but there was no clear guidance on the next step, and the current literature had no answers as to how systemic corticosteroids might impact the care of these children,” Dr. Goenka said in an interview. “So we wanted to leverage the PHIS data to better understand the association with the need for surgery and length of stay. Surgery is painful and often involves IV administration of opioid painkillers. It’s something we may be able to avoid if we can optimize medical treatment.”
Pending results from randomized trials, what immediate impact could these registry findings have? “We hope that physicians will think about the best initial medical treatment plan for these children,” Dr. Goenka said. ”Given these data, I would be more likely to incorporate steroids early on in medical treatment.”
She emphasized, however, that before routine adoption prospective studies are needed to clearly identify which patients will have a strong benefit and which will not benefit. “That is the nuanced discussion that will happen with more prospective work.”
Dr. Goenka and associates explained that the rising incidence of RPAs and PPAs over the past 20 years has been attributed to more cases of tonsillitis because of a shift away from tonsillectomies, as well as the changing epidemiology of methicillin-resistant Staphylococcus aureus.
In an accompanying editorial, Ellen R. Wald, MD, and Jens C. Eickhoff, PhD, of the University of Wisconsin–Madison, stated that the use of corticosteroids in bacterial meningitis is often cited as an example of the benefits of steroids in infection. “The specific rationale for use of corticosteroids is [their] anti-inflammatory effects, which may result in decreases of swelling and/ or edema to facilitate drainage, perfusion, reduction in pain, and healing.”
They cautioned, however, that the pharmacologic effects of steroids are myriad and complicated, and include potential masking of the clinical course of disease, thereby delaying appropriate therapy for unrecognized deterioration, as well potential immunosuppression.
Acute orbital cellulitis
In the second retrospective analysis, a group led by pediatrician Maria Anna Leszczynska, MD, of Johns Hopkins All Children’s Hospital in Baltimore, analyzed a retrospective PHIS cohort of 5,645 children younger than 18 years with a primary diagnosis of orbital cellulitis treated at 51 hospitals from January 2007 to December 2018.
Of these, 1,347 (24%) received steroids, but, contrary to earlier reports, the data showed no reduction in length of stay associated with these drugs after adjustment for age, meningitis, abscess, or vision issues (ebeta, 1.01; 95% CI, 0.97-1.06). Corticosteroid exposure was, however, associated with operative episodes after 2 days’ hospitalization (OR, 2.05; 95% CI, 1.29-3.27) and 30-day readmission (OR, 2.40; 95% CI, 1.52-3.78).
“Among children hospitalized for orbital cellulitis, we did not observe the reduction in LOS [length of stay] for patients prescribed systemic corticosteroids as described previously in the literature,” the authors wrote.
In terms of surgical procedures, 52.0% of corticosteroid recipients versus 14.0% of nonrecipients underwent surgery (P < .001), and more were hospitalized in the pediatric ICU (4.4% vs 2.6%; P < .001).
According to the editorialists: “Both observations suggest that children who received steroids may have been a sicker group of patients.”
Dr. Wald and Dr. Eickhoff pointed out that the effect of steroids is ultimately unclear because of the retrospective study’s inherent potential for bias because of unobserved confounders. Were steroids prescribed more often when children were perceived to be sicker with more severe disease, or did these medications cause worse outcomes?
The authors agreed that the study could not determine causality. “Although we used all available markers of disease severity, there does not exist a validated disease severity clinical score for pediatric orbital cellulitis,” they wrote.
According to Ricardo A. Quinonez, MD, an associate professor of pediatrics and division and service chief of pediatric hospital medicine at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, “orbital cellulitis is a not very common thing in children so we don’t treat many patients with this. But having said that, there is usually some debate among providers about whether to use steroids.”
Some centers use them routinely for central nervous system and eye infections or extensions of sinusitis, he said, but there is variability in the prescribing of corticosteroids. “There’s ongoing discussion as to whether they‘re as helpful in orbital cellulitis as they are in similar conditions,” Dr. Quinonez said in an interview. “At our institution we don’t typically prescribe them – not never but not routinely. Children who are sicker tend to get steroids, as they do in other conditions.”
In the context of PPA as in the first study, he added, “I think the evidence favoring the use of steroids in infections that affect the airway is stronger, and their use is definitely more prevalent in those instances.”
While both PHIS analyses suggested some benefit from steroids, he continued, some children may not benefit and there may be harms. “The evidence is still mostly retrospective and observational with no multicenter randomized controlled data. Without those data the evidence is difficult to interpret and subject to all the biases that observational and retrospective data is subject to and the current evidence should not lead physicians to change their practice until controlled, randomized evidence is available.”
The editorialists concurred with the study authors and Dr. Quinonez that large, controlled, prospective clinical trials are needed to ascertain the effect of steroids and to standardize the approach to diagnosis and management. “Use of administrative databases are not optimal to answer questions related to outcome,” they wrote.
The study by Dr. Goenka and associates received no external funding; the study by Dr. Leszczynska and associates also received no external funding. None of the authors declared potential competing interests. Dr. Quinonez had no competing interests to declare. Dr. Wald and Dr. Eickhoff disclosed no competing interests with regard to their editorial.
FROM PEDIATRICS
Health care unaffordability common for pregnant/postpartum women
Financial hardship remains prevalent among pregnant and postpartum women, despite the implementation of the Affordable Care Act (ACA), according to new findings published in JAMA Network Open.
Nearly a quarter (24%) of pregnant and postpartum women reported having unmet health care needs, 60% had health care unaffordability, and 54% reported general financial stress. Notably, the type of insurance was associated with the ability to afford health care.
Those with private insurance, along with women with lower incomes, were more likely to experience unaffordable health care, compared to those covered by public insurance or who had higher incomes.
Senior study author Michelle H. Moniz, MD, assistant professor in the department of obstetrics and gynecology at the University of Michigan, Ann Arbor, was surprised by multiple study findings. “The prevalence of financial hardship overall, and the three individual indicators of hardship, did not change over time from 2013 to 2018,” she said. “The ACA was enacted just prior to the study period, and while this policy had many benefits for women – especially around increasing insurance coverage – it does not seem to have improved financial hardship among pregnant and postpartum women.”
She emphasized that two groups were at the highest risk of health care unaffordability: those with private insurance and those living on low incomes. “This is notable, as we often think of private insurance as offering ‘Cadillac coverage,’ but our prior work suggests that privately insured women have strikingly high out-of-pocket costs for pregnancy and childbirth-related care,” Dr. Moniz said.
These expenses include deductibles, copays, and coinsurance payments, which come to about $4,500 on average. Medicaid plans, in contrast, have exceedingly low out-of-pocket costs for pregnant and postpartum women. “Findings from the current study call for targeted policy interventions to alleviate financial strain and remove financial barriers to health care access for privately insured families,” she said. “Similarly, families living on lower incomes were also at high risk of health care unaffordability. This may be because even small out-of-pocket costs, or health care–associated costs, account for a larger share of the family’s income.”
This finding for lower-income women calls for targeted policy interventions. “Sliding-scale deductibles, for example, are one solution that might mitigate economic hardship and remove cost-related barriers to health care for pregnant and postpartum women,” Dr. Moniz added.
Health care unaffordability high
In this study, Dr. Moniz and colleagues evaluated the prevalence of financial hardship among peripartum women over time, and how it was affected by their income level and the type of insurance coverage.
They conducted a cross-sectional study that included peripartum women between the ages of 18 and 45 years who reported being currently pregnant or pregnant in the past 12 months. The women were all participants in the National Health Interview Survey, which covers the period from 2013 to 2018, and the data were analyzed from January to May 2021.
The cohort included 3,509 peripartum women, and was weighted to represent 1,050,789 women, with a mean age of 29 years. In 2018, an estimated 39,017 of 184,018 (21.2%) were Black; 36,045 (19.6%) were Hispanic; and 97,366 (52.9%) were White. In the latter years of the study period, the participants tended to be older, more highly educated, and less likely to lack insurance.
When the authors compared the unadjusted reported financial hardship outcome by each study year, unmet health care need (2013: 27.9% [95% confidence interval, 24.4%-31.7%]; 2018: 23.7% [95% CI, 19.5%-28.6%]), health care unaffordability (2013: 65.7% [95% CI, 61.1%-70.0%]; 2018: 58.8% [95% CI, 53.4%-64.0%]), and general financial stress (2013: 60.6% [95% CI, 55.2%-65.8%]; 2018: 53.8% [95% CI, 47.8%-59.8%]) remained largely unchanged between 2013 and 2018.
When they looked at the relationship between insurance type, income, and financial difficulties, some degree of financial hardship was common across all groups; private insurance: 63.8% [95% CI, 61.1%-66.6%]; with public insurance: 49.9% [95% CI, 46.4%-53.4%]; with no insurance: 81.8% [95% CI, 76.4%-87.3%]; with income < 400% of the federal poverty level (FPL): 65.5% [95% CI, 62.1%-66.9%]; with income at least 400% of the FPL: 49.3% [95% CI,44.7%-53.9%]).
Those without any insurance had the highest odds of reporting unmet health care needs (adjusted OR [aOR], 4.40; 95% CI, 3.23-6.00) and health care unaffordability (aOR, 5.18; 95% CI, 3.49-7.70) compared with women who received public insurance.
But while women with private insurance had lower odds of reporting unmet health care needs (aOR, 0.67; 95% CI, 0.52-0.87), they faced higher odds of reporting health care unaffordability (aOR, 1.88; 95% CI, 1.49-2.36) compared to women who had public insurance.
Those with household incomes of less than 400% of the FPL had higher odds of reporting unmet health care need (aOR,1.50; 95% CI, 1.08-2.08) and health care unaffordability (aOR, 1.98; 95% CI, 1.54-2.55) versus women whose household incomes were at least 400% of FPL. The odds of general financial stress did not significantly differ by insurance status/type or income level.
Weighing in on the data
Jamie Daw, PhD, assistant professor of health policy and management, Columbia University Mailman School of Public Health, New York, noted that many people think of private insurance as “good coverage.”
“But the portion of medical costs that patients are required to pay under private plans has risen dramatically over the past decade,” she said. “Over half of the U.S. workforce is now enrolled in high-deductible plans, where the average deductible was $4,500 in 2020. The private insurance of today does not provide sufficient financial protection for most families, who would need to have the liquid assets to cover childbirth.”
Another expert agreed that the high out-of-pocket costs for women with private health insurance were probably responsible for making peripartum health care more unaffordable. These included costs for pregnancy care as well as for maternal and infant care during and after childbirth.
“This study reporting the high unmet medical needs and unaffordability of health care for peripartum women further underscores that the U.S. health care system is not meeting the needs of pregnant women, mothers, and their newborn infants,” said Lois K. Lee, MD, associate professor of pediatrics and emergency medicine at Harvard Medical School and associate director for public policy at the Sandra L. Fenwick Institute for Pediatric Health Equity and Inclusion, Boston.
“It is imperative to optimize the health of pregnant mothers to optimize the health of infants, who are our future society,” she said. “Policies which would expand Medicaid coverage to a full 1-year postpartum across all states is one important strategy to improve health care access and affordability to peripartum women. However, this must be part of a multipronged approach addressing the social determinants of health, as insurance coverage alone will not fully address this important health issue of peripartum women, and their children.”
Dr Moniz reported receiving personal fees from the RAND Corporation, the Society of Family Planning outside the submitted work and grant K08 HS025465 from the Agency for Healthcare Research and Quality. Dr. Daw has no disclosures. Dr. Lee reports speaker fees from the American Academy of Pediatrics and SUNY Upstate Medical University. Coauthor Dr. Taylor was supported by the National Clinician Scholars Program at the University of Michigan. Dr Dalton was supported by grant R01 HS023784 from the Agency for Healthcare Research and Quality.
Financial hardship remains prevalent among pregnant and postpartum women, despite the implementation of the Affordable Care Act (ACA), according to new findings published in JAMA Network Open.
Nearly a quarter (24%) of pregnant and postpartum women reported having unmet health care needs, 60% had health care unaffordability, and 54% reported general financial stress. Notably, the type of insurance was associated with the ability to afford health care.
Those with private insurance, along with women with lower incomes, were more likely to experience unaffordable health care, compared to those covered by public insurance or who had higher incomes.
Senior study author Michelle H. Moniz, MD, assistant professor in the department of obstetrics and gynecology at the University of Michigan, Ann Arbor, was surprised by multiple study findings. “The prevalence of financial hardship overall, and the three individual indicators of hardship, did not change over time from 2013 to 2018,” she said. “The ACA was enacted just prior to the study period, and while this policy had many benefits for women – especially around increasing insurance coverage – it does not seem to have improved financial hardship among pregnant and postpartum women.”
She emphasized that two groups were at the highest risk of health care unaffordability: those with private insurance and those living on low incomes. “This is notable, as we often think of private insurance as offering ‘Cadillac coverage,’ but our prior work suggests that privately insured women have strikingly high out-of-pocket costs for pregnancy and childbirth-related care,” Dr. Moniz said.
These expenses include deductibles, copays, and coinsurance payments, which come to about $4,500 on average. Medicaid plans, in contrast, have exceedingly low out-of-pocket costs for pregnant and postpartum women. “Findings from the current study call for targeted policy interventions to alleviate financial strain and remove financial barriers to health care access for privately insured families,” she said. “Similarly, families living on lower incomes were also at high risk of health care unaffordability. This may be because even small out-of-pocket costs, or health care–associated costs, account for a larger share of the family’s income.”
This finding for lower-income women calls for targeted policy interventions. “Sliding-scale deductibles, for example, are one solution that might mitigate economic hardship and remove cost-related barriers to health care for pregnant and postpartum women,” Dr. Moniz added.
Health care unaffordability high
In this study, Dr. Moniz and colleagues evaluated the prevalence of financial hardship among peripartum women over time, and how it was affected by their income level and the type of insurance coverage.
They conducted a cross-sectional study that included peripartum women between the ages of 18 and 45 years who reported being currently pregnant or pregnant in the past 12 months. The women were all participants in the National Health Interview Survey, which covers the period from 2013 to 2018, and the data were analyzed from January to May 2021.
The cohort included 3,509 peripartum women, and was weighted to represent 1,050,789 women, with a mean age of 29 years. In 2018, an estimated 39,017 of 184,018 (21.2%) were Black; 36,045 (19.6%) were Hispanic; and 97,366 (52.9%) were White. In the latter years of the study period, the participants tended to be older, more highly educated, and less likely to lack insurance.
When the authors compared the unadjusted reported financial hardship outcome by each study year, unmet health care need (2013: 27.9% [95% confidence interval, 24.4%-31.7%]; 2018: 23.7% [95% CI, 19.5%-28.6%]), health care unaffordability (2013: 65.7% [95% CI, 61.1%-70.0%]; 2018: 58.8% [95% CI, 53.4%-64.0%]), and general financial stress (2013: 60.6% [95% CI, 55.2%-65.8%]; 2018: 53.8% [95% CI, 47.8%-59.8%]) remained largely unchanged between 2013 and 2018.
When they looked at the relationship between insurance type, income, and financial difficulties, some degree of financial hardship was common across all groups; private insurance: 63.8% [95% CI, 61.1%-66.6%]; with public insurance: 49.9% [95% CI, 46.4%-53.4%]; with no insurance: 81.8% [95% CI, 76.4%-87.3%]; with income < 400% of the federal poverty level (FPL): 65.5% [95% CI, 62.1%-66.9%]; with income at least 400% of the FPL: 49.3% [95% CI,44.7%-53.9%]).
Those without any insurance had the highest odds of reporting unmet health care needs (adjusted OR [aOR], 4.40; 95% CI, 3.23-6.00) and health care unaffordability (aOR, 5.18; 95% CI, 3.49-7.70) compared with women who received public insurance.
But while women with private insurance had lower odds of reporting unmet health care needs (aOR, 0.67; 95% CI, 0.52-0.87), they faced higher odds of reporting health care unaffordability (aOR, 1.88; 95% CI, 1.49-2.36) compared to women who had public insurance.
Those with household incomes of less than 400% of the FPL had higher odds of reporting unmet health care need (aOR,1.50; 95% CI, 1.08-2.08) and health care unaffordability (aOR, 1.98; 95% CI, 1.54-2.55) versus women whose household incomes were at least 400% of FPL. The odds of general financial stress did not significantly differ by insurance status/type or income level.
Weighing in on the data
Jamie Daw, PhD, assistant professor of health policy and management, Columbia University Mailman School of Public Health, New York, noted that many people think of private insurance as “good coverage.”
“But the portion of medical costs that patients are required to pay under private plans has risen dramatically over the past decade,” she said. “Over half of the U.S. workforce is now enrolled in high-deductible plans, where the average deductible was $4,500 in 2020. The private insurance of today does not provide sufficient financial protection for most families, who would need to have the liquid assets to cover childbirth.”
Another expert agreed that the high out-of-pocket costs for women with private health insurance were probably responsible for making peripartum health care more unaffordable. These included costs for pregnancy care as well as for maternal and infant care during and after childbirth.
“This study reporting the high unmet medical needs and unaffordability of health care for peripartum women further underscores that the U.S. health care system is not meeting the needs of pregnant women, mothers, and their newborn infants,” said Lois K. Lee, MD, associate professor of pediatrics and emergency medicine at Harvard Medical School and associate director for public policy at the Sandra L. Fenwick Institute for Pediatric Health Equity and Inclusion, Boston.
“It is imperative to optimize the health of pregnant mothers to optimize the health of infants, who are our future society,” she said. “Policies which would expand Medicaid coverage to a full 1-year postpartum across all states is one important strategy to improve health care access and affordability to peripartum women. However, this must be part of a multipronged approach addressing the social determinants of health, as insurance coverage alone will not fully address this important health issue of peripartum women, and their children.”
Dr Moniz reported receiving personal fees from the RAND Corporation, the Society of Family Planning outside the submitted work and grant K08 HS025465 from the Agency for Healthcare Research and Quality. Dr. Daw has no disclosures. Dr. Lee reports speaker fees from the American Academy of Pediatrics and SUNY Upstate Medical University. Coauthor Dr. Taylor was supported by the National Clinician Scholars Program at the University of Michigan. Dr Dalton was supported by grant R01 HS023784 from the Agency for Healthcare Research and Quality.
Financial hardship remains prevalent among pregnant and postpartum women, despite the implementation of the Affordable Care Act (ACA), according to new findings published in JAMA Network Open.
Nearly a quarter (24%) of pregnant and postpartum women reported having unmet health care needs, 60% had health care unaffordability, and 54% reported general financial stress. Notably, the type of insurance was associated with the ability to afford health care.
Those with private insurance, along with women with lower incomes, were more likely to experience unaffordable health care, compared to those covered by public insurance or who had higher incomes.
Senior study author Michelle H. Moniz, MD, assistant professor in the department of obstetrics and gynecology at the University of Michigan, Ann Arbor, was surprised by multiple study findings. “The prevalence of financial hardship overall, and the three individual indicators of hardship, did not change over time from 2013 to 2018,” she said. “The ACA was enacted just prior to the study period, and while this policy had many benefits for women – especially around increasing insurance coverage – it does not seem to have improved financial hardship among pregnant and postpartum women.”
She emphasized that two groups were at the highest risk of health care unaffordability: those with private insurance and those living on low incomes. “This is notable, as we often think of private insurance as offering ‘Cadillac coverage,’ but our prior work suggests that privately insured women have strikingly high out-of-pocket costs for pregnancy and childbirth-related care,” Dr. Moniz said.
These expenses include deductibles, copays, and coinsurance payments, which come to about $4,500 on average. Medicaid plans, in contrast, have exceedingly low out-of-pocket costs for pregnant and postpartum women. “Findings from the current study call for targeted policy interventions to alleviate financial strain and remove financial barriers to health care access for privately insured families,” she said. “Similarly, families living on lower incomes were also at high risk of health care unaffordability. This may be because even small out-of-pocket costs, or health care–associated costs, account for a larger share of the family’s income.”
This finding for lower-income women calls for targeted policy interventions. “Sliding-scale deductibles, for example, are one solution that might mitigate economic hardship and remove cost-related barriers to health care for pregnant and postpartum women,” Dr. Moniz added.
Health care unaffordability high
In this study, Dr. Moniz and colleagues evaluated the prevalence of financial hardship among peripartum women over time, and how it was affected by their income level and the type of insurance coverage.
They conducted a cross-sectional study that included peripartum women between the ages of 18 and 45 years who reported being currently pregnant or pregnant in the past 12 months. The women were all participants in the National Health Interview Survey, which covers the period from 2013 to 2018, and the data were analyzed from January to May 2021.
The cohort included 3,509 peripartum women, and was weighted to represent 1,050,789 women, with a mean age of 29 years. In 2018, an estimated 39,017 of 184,018 (21.2%) were Black; 36,045 (19.6%) were Hispanic; and 97,366 (52.9%) were White. In the latter years of the study period, the participants tended to be older, more highly educated, and less likely to lack insurance.
When the authors compared the unadjusted reported financial hardship outcome by each study year, unmet health care need (2013: 27.9% [95% confidence interval, 24.4%-31.7%]; 2018: 23.7% [95% CI, 19.5%-28.6%]), health care unaffordability (2013: 65.7% [95% CI, 61.1%-70.0%]; 2018: 58.8% [95% CI, 53.4%-64.0%]), and general financial stress (2013: 60.6% [95% CI, 55.2%-65.8%]; 2018: 53.8% [95% CI, 47.8%-59.8%]) remained largely unchanged between 2013 and 2018.
When they looked at the relationship between insurance type, income, and financial difficulties, some degree of financial hardship was common across all groups; private insurance: 63.8% [95% CI, 61.1%-66.6%]; with public insurance: 49.9% [95% CI, 46.4%-53.4%]; with no insurance: 81.8% [95% CI, 76.4%-87.3%]; with income < 400% of the federal poverty level (FPL): 65.5% [95% CI, 62.1%-66.9%]; with income at least 400% of the FPL: 49.3% [95% CI,44.7%-53.9%]).
Those without any insurance had the highest odds of reporting unmet health care needs (adjusted OR [aOR], 4.40; 95% CI, 3.23-6.00) and health care unaffordability (aOR, 5.18; 95% CI, 3.49-7.70) compared with women who received public insurance.
But while women with private insurance had lower odds of reporting unmet health care needs (aOR, 0.67; 95% CI, 0.52-0.87), they faced higher odds of reporting health care unaffordability (aOR, 1.88; 95% CI, 1.49-2.36) compared to women who had public insurance.
Those with household incomes of less than 400% of the FPL had higher odds of reporting unmet health care need (aOR,1.50; 95% CI, 1.08-2.08) and health care unaffordability (aOR, 1.98; 95% CI, 1.54-2.55) versus women whose household incomes were at least 400% of FPL. The odds of general financial stress did not significantly differ by insurance status/type or income level.
Weighing in on the data
Jamie Daw, PhD, assistant professor of health policy and management, Columbia University Mailman School of Public Health, New York, noted that many people think of private insurance as “good coverage.”
“But the portion of medical costs that patients are required to pay under private plans has risen dramatically over the past decade,” she said. “Over half of the U.S. workforce is now enrolled in high-deductible plans, where the average deductible was $4,500 in 2020. The private insurance of today does not provide sufficient financial protection for most families, who would need to have the liquid assets to cover childbirth.”
Another expert agreed that the high out-of-pocket costs for women with private health insurance were probably responsible for making peripartum health care more unaffordable. These included costs for pregnancy care as well as for maternal and infant care during and after childbirth.
“This study reporting the high unmet medical needs and unaffordability of health care for peripartum women further underscores that the U.S. health care system is not meeting the needs of pregnant women, mothers, and their newborn infants,” said Lois K. Lee, MD, associate professor of pediatrics and emergency medicine at Harvard Medical School and associate director for public policy at the Sandra L. Fenwick Institute for Pediatric Health Equity and Inclusion, Boston.
“It is imperative to optimize the health of pregnant mothers to optimize the health of infants, who are our future society,” she said. “Policies which would expand Medicaid coverage to a full 1-year postpartum across all states is one important strategy to improve health care access and affordability to peripartum women. However, this must be part of a multipronged approach addressing the social determinants of health, as insurance coverage alone will not fully address this important health issue of peripartum women, and their children.”
Dr Moniz reported receiving personal fees from the RAND Corporation, the Society of Family Planning outside the submitted work and grant K08 HS025465 from the Agency for Healthcare Research and Quality. Dr. Daw has no disclosures. Dr. Lee reports speaker fees from the American Academy of Pediatrics and SUNY Upstate Medical University. Coauthor Dr. Taylor was supported by the National Clinician Scholars Program at the University of Michigan. Dr Dalton was supported by grant R01 HS023784 from the Agency for Healthcare Research and Quality.
FROM JAMA NETWORK OPEN
Time to attack hypoactivity in our children
My 50th medical school reunion has come and gone. This milestone offered me another opportunity to look back over the last 5 decades of pediatrics that I have watched pass under the bridge. Triggered by the discovery of two recently published studies, this particular view back over my shoulder induced a wave of sadness, anger, and frustration that I have had trouble shaking.
The first study demonstrated a strong positive effect of exercise on academic achievement, the other found that children who were more physically active have weathered the pandemic with fewer mental health problems.
These studies are just two pieces of a growing body of evidence that our sedentary lifestyles are shortening our lives and launching our children into adulthood burdened with a raft of health risks they could possibly have avoided by being more physically active. Encountering these two papers just as the alumni office was inviting me to engage in an orgy of retrospection and introspection made me consider how little I and others in my profession have done to substantially address this scourge on our young people.
Yes, I have tried to encourage my patients to be less sedentary and more active. Yes, I have tried to set a very visible example by bicycling and walking around town. Yes, I have coached youth sports teams. All of my children and grandchildren are leading active lives and appear to be reaping the benefits. But in the grander scheme of things I feel that neither I nor the American Academy of Pediatrics has made a difference.
In March of 2020 the AAP published a clinical report that lists the numerous positive associations between activity and health that includes a comprehensive collection of suggestions for providers on how we might assess the problem of inactivity and then play a role in addressing it with our patients and our communities. Unfortunately, the message’s importance was lost in the glut of pandemic news.
While the AAP’s report should have been published many decades ago, I doubt the delay lessened its impact significantly because the report is primarily a compendium of recommendations that in the long run will be seen as just another example of us believers preaching to the choir.
Making lifestyle changes on the order of magnitude necessary to convert an increasingly sedentary population into one that unconsciously becomes physically active requires more than recommendations. It is only natural that folks have trouble saying “No.”
No to the entertainment of electronic devices. No to the comforts of all-weather enclosed transportation. No to hours on the couch. Overcoming the inertia built into our society is going to require more than encouragement, recommendations, and professional sports–sponsored presidential initiatives.
Mandate has become a politically charged dirty word. But our current experience with the COVID-19 vaccines should help us realize that there is a significant segment of the population that doesn’t like being told what to do even if the outcome is in their best interest. Education and rewards have fallen short, but the evidence is mounting that mandates can work.
There was a time when physical activity was built into every child’s school day. For a variety of bad reasons, vigorous physical education classes and once- or twice-daily outdoor recesses have disappeared from the educational landscape. It is time to return to them in a robust form. Unfortunately, because activity isn’t happening at home it will take a government mandate.
There will be pushback. Even from some educators whose observations should have shown them the critical role of physical activity in health and academic success. We must move the distraction of the phenomenon once known simply as hyperactivity to the back burner and tackle the real epidemic of hypoactivity that is destroying our children.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
My 50th medical school reunion has come and gone. This milestone offered me another opportunity to look back over the last 5 decades of pediatrics that I have watched pass under the bridge. Triggered by the discovery of two recently published studies, this particular view back over my shoulder induced a wave of sadness, anger, and frustration that I have had trouble shaking.
The first study demonstrated a strong positive effect of exercise on academic achievement, the other found that children who were more physically active have weathered the pandemic with fewer mental health problems.
These studies are just two pieces of a growing body of evidence that our sedentary lifestyles are shortening our lives and launching our children into adulthood burdened with a raft of health risks they could possibly have avoided by being more physically active. Encountering these two papers just as the alumni office was inviting me to engage in an orgy of retrospection and introspection made me consider how little I and others in my profession have done to substantially address this scourge on our young people.
Yes, I have tried to encourage my patients to be less sedentary and more active. Yes, I have tried to set a very visible example by bicycling and walking around town. Yes, I have coached youth sports teams. All of my children and grandchildren are leading active lives and appear to be reaping the benefits. But in the grander scheme of things I feel that neither I nor the American Academy of Pediatrics has made a difference.
In March of 2020 the AAP published a clinical report that lists the numerous positive associations between activity and health that includes a comprehensive collection of suggestions for providers on how we might assess the problem of inactivity and then play a role in addressing it with our patients and our communities. Unfortunately, the message’s importance was lost in the glut of pandemic news.
While the AAP’s report should have been published many decades ago, I doubt the delay lessened its impact significantly because the report is primarily a compendium of recommendations that in the long run will be seen as just another example of us believers preaching to the choir.
Making lifestyle changes on the order of magnitude necessary to convert an increasingly sedentary population into one that unconsciously becomes physically active requires more than recommendations. It is only natural that folks have trouble saying “No.”
No to the entertainment of electronic devices. No to the comforts of all-weather enclosed transportation. No to hours on the couch. Overcoming the inertia built into our society is going to require more than encouragement, recommendations, and professional sports–sponsored presidential initiatives.
Mandate has become a politically charged dirty word. But our current experience with the COVID-19 vaccines should help us realize that there is a significant segment of the population that doesn’t like being told what to do even if the outcome is in their best interest. Education and rewards have fallen short, but the evidence is mounting that mandates can work.
There was a time when physical activity was built into every child’s school day. For a variety of bad reasons, vigorous physical education classes and once- or twice-daily outdoor recesses have disappeared from the educational landscape. It is time to return to them in a robust form. Unfortunately, because activity isn’t happening at home it will take a government mandate.
There will be pushback. Even from some educators whose observations should have shown them the critical role of physical activity in health and academic success. We must move the distraction of the phenomenon once known simply as hyperactivity to the back burner and tackle the real epidemic of hypoactivity that is destroying our children.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
My 50th medical school reunion has come and gone. This milestone offered me another opportunity to look back over the last 5 decades of pediatrics that I have watched pass under the bridge. Triggered by the discovery of two recently published studies, this particular view back over my shoulder induced a wave of sadness, anger, and frustration that I have had trouble shaking.
The first study demonstrated a strong positive effect of exercise on academic achievement, the other found that children who were more physically active have weathered the pandemic with fewer mental health problems.
These studies are just two pieces of a growing body of evidence that our sedentary lifestyles are shortening our lives and launching our children into adulthood burdened with a raft of health risks they could possibly have avoided by being more physically active. Encountering these two papers just as the alumni office was inviting me to engage in an orgy of retrospection and introspection made me consider how little I and others in my profession have done to substantially address this scourge on our young people.
Yes, I have tried to encourage my patients to be less sedentary and more active. Yes, I have tried to set a very visible example by bicycling and walking around town. Yes, I have coached youth sports teams. All of my children and grandchildren are leading active lives and appear to be reaping the benefits. But in the grander scheme of things I feel that neither I nor the American Academy of Pediatrics has made a difference.
In March of 2020 the AAP published a clinical report that lists the numerous positive associations between activity and health that includes a comprehensive collection of suggestions for providers on how we might assess the problem of inactivity and then play a role in addressing it with our patients and our communities. Unfortunately, the message’s importance was lost in the glut of pandemic news.
While the AAP’s report should have been published many decades ago, I doubt the delay lessened its impact significantly because the report is primarily a compendium of recommendations that in the long run will be seen as just another example of us believers preaching to the choir.
Making lifestyle changes on the order of magnitude necessary to convert an increasingly sedentary population into one that unconsciously becomes physically active requires more than recommendations. It is only natural that folks have trouble saying “No.”
No to the entertainment of electronic devices. No to the comforts of all-weather enclosed transportation. No to hours on the couch. Overcoming the inertia built into our society is going to require more than encouragement, recommendations, and professional sports–sponsored presidential initiatives.
Mandate has become a politically charged dirty word. But our current experience with the COVID-19 vaccines should help us realize that there is a significant segment of the population that doesn’t like being told what to do even if the outcome is in their best interest. Education and rewards have fallen short, but the evidence is mounting that mandates can work.
There was a time when physical activity was built into every child’s school day. For a variety of bad reasons, vigorous physical education classes and once- or twice-daily outdoor recesses have disappeared from the educational landscape. It is time to return to them in a robust form. Unfortunately, because activity isn’t happening at home it will take a government mandate.
There will be pushback. Even from some educators whose observations should have shown them the critical role of physical activity in health and academic success. We must move the distraction of the phenomenon once known simply as hyperactivity to the back burner and tackle the real epidemic of hypoactivity that is destroying our children.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.