User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Patient contact time vs. admin: Is your contract fair?
What’s in a day’s work? For doctors, it’s typically a mix of seeing patients and completing paperwork and follow-up. Often it extends well past the standard workday.
Dennis Hursh, JD, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, describes one overwhelmed ob.gyn. who recently consulted him for this problem.
“My client had accepted a position in a group practice where his contract stated he would be working during normal office hours, Monday through Friday, from 8 a.m. to 5 p.m. – in other words, a 40-hour workweek,” Mr. Hursh said.
But the distressed physician discovered that actually, he was working almost twice as many hours. “He’d get to work early to do charting, then see patients during the 40 hours, perhaps grabbing a quick sandwich for a few minutes – and then stay after 5 [p.m.] for a few more hours when he’d work on charts or other administrative tasks. Then he’d get something to eat, work on more charts, then go to bed, get up in the morning, and repeat.”
Mr. Hursh summarized the client’s life: “Eating, sleeping, practicing clinical medicine, and doing nonclinical tasks.”
It turned out that the 40-hour workweek included in the contract referred to patient-facing hours, not to all of the ancillary tasks that are part of practicing medicine in this day and age. “Unfortunately, this is far from an isolated story,” said Mr. Hursh.
Be aware of what’s in the contract
“The first draft of many standard physician employment contracts often omits mention of patient contact hour requirements and rather uses vague verbiage such as ‘full-time’ employment or ‘1.0 FTE’ – or full-time equivalent – without defining that term,” said Mr. Hursh. Typically, the 40 hours exclude call coverage, but most physicians understand that and, at least at first glance, it all sounds very reasonable.
But once charting, hours on the phone, arguing with managed care companies, sending in prescriptions, administrative meetings, and other tasks are thrown in, the work hours expand dramatically. Moreover, if your employer doesn’t utilize hospitalists, you may be expected to “round” outside of the 40 hours, which can be particularly burdensome if the employer admits patients to multiple hospitals.
Amanda Hill, JD, owner of Hill Health Law based in Austin, Texas, told this news organization that this predicament isn’t unique to physicians. Exempt employees who don’t clock in and out are often expected to work overtime – that is, to “work as long as it takes to get the job done.” It can affect NPs, PAs, and many others in the health care space. But the number of tasks that fall upon a doctor’s shoulders and the fact that patients’ health and lives are at stake up the ante and make the situation far more difficult for doctors than for employees in other industries.
So it’s important to nail down precise terms in the contract and, if possible, negotiate for a more humane schedule by specifying how the working hours will be used.
“It’s true that a 1.0 FTE definition is too vague,” Ms. Hill said. “I’ve negotiated a lot of contracts where we nail down in writing that the in-office schedule equals 34 hours per week, so the physician is guaranteed an additional 6 hours for administrative time.”
Mr. Hursh usually asks for 32 hours of patient contact per week, which leaves 1 full day per week to catch up on basic administrative tasks. “It’s important for employers to recognize that seeing patients isn’t the only thing a doctor does and there’s a lot of work in addition to face-to-face time,” he said.
But he hasn’t always been successful. One physician client was seeking a workweek consisting of 36 patient contact hours, “which is 90% of the usual FTE of a 40-hour week,” said Mr. Hursh. “But the employer called it ‘part-time,’ as if the doctor were planning to be lying in the sun for the other 4 hours.”
The client decided to accept a 10% pay cut and 10% less vacation to guarantee that she had those extra hours for administrative tasks. “She’s probably working way more than 36 hours a week, but maybe closer to 50 or 60 instead of 70 or more,” he said.
Clarify call coverage
Call coverage is typically not included in the hours a physician is contracted to work on a weekly basis. “Most contracts have call, and it’s usually evenly distributed among parties in a practice, but call can expand if another doctor is out sick, for example,” said Ms. Hill.
Sometimes the language in the contract is vague regarding call coverage. “I ask, how many shifts per year is the doctor is expected to work? Then, I try to negotiate extra pay if more shifts arise,” she said. “The hospital or practice may not demand extra call because they don’t want to pay extra money to the physician.”
On the other hand, some physicians may be eager to take extra call if it means extra income.
Ms. Hill stated that one of her clients was being paid as a “part-time, 2-day-a-week provider” but was asked to be on call and take night and weekend work. When you added it all up, she was putting in almost 30 hours a week.
“This is abusive to a provider that works so hard for patients,” Ms. Hill said. “We have to protect them through the contract language, so they have something hard and fast to point to when their administrator pushes them too hard. Doctors should get value for their time.”
Ms. Hill and her client pushed for more money, and the employer gave in. “All we had to do was to point out how many hours she was actually working. She didn’t mind all the extra call, but she wanted to be compensated.” The doctor’s salary was hiked by $25,000.
Differences in specialties and settings
There are some specialties where it might be easier to have more defined hours, while other specialties are more challenging. Anu Murthy, Esq., an attorney and associate contract review specialist at Contract Diagnostics (a national firm that reviews physician contracts) told this news organization that the work of hospitalists, intensivists, and emergency department physicians, for example, is done in shifts, which tend to be fixed hours.
“They need to get their charting completed so that whoever takes over on the next shift has access to the most recent notes about the patient,” she said. By contrast, surgeons can’t always account for how long a given surgery will take. “It could be as long as 9 hours,” she said. Notes need to be written immediately for the sake of the patient’s postsurgical care.
Dermatologists tend to deal with fewer emergencies, compared with other specialists, and it’s easier for their patients to be slotted into an organized schedule. On the other hand, primary care doctors – internists, family practice physicians, and pediatricians – may be seeing 40-50 patients a day, one every 15 minutes.
Practice setting also makes a difference, said Ms. Murthy. Veterans Administration (VA) hospitals or government-run clinics tend to have more rigidly defined hours, compared with other settings, so if you’re in a VA hospital or government-run clinic, work-life balance tends to be better.
Physicians who work remotely via telehealth also tend to have a better work-life balance, compared with those who see patients in person, Ms. Murthy said. But the difference may be in not having to spend extra time commuting to work or interacting with others in the work environment, since some research has suggested that telehealth physicians may actually spend more time engaged in charting after hours, compared with their in-person counterparts.
Using scribes to maximize your time
Elliott Trotter, MD, is an emergency medicine physician, associate clinical professor of emergency medicine at Texas Christian University Medical Schools, and founder of the ScribeNest, a Texas-based company that trains health care scribes. He told this news organization that there are ways to maximize one’s time during shifts so that much of the charting can be accomplished during working hours.
“About 28 years ago, I realized that the documentation load for physicians was enormous and at that time I developed the Modern Scribe, using premed students for ‘elbow support’ to help with the workload by documenting the ED encounters in real time during the encounter so I wouldn’t have to do so later.”
Over the years, as EHRs have become more ubiquitous and onerous, the role of the scribe has “evolved from a luxury to a necessity,” said Dr. Trotter. The scribes can actually record the encounter directly into the EHR so that the physician doesn’t have to do so later and doesn’t have to look at a computer screen but can look at the patient during the encounter.
“This enhances communication and has been shown to improve patient care,” he said.
Dr. Trotter said he rarely, if ever, needs to do documentation after hours. “But one of my physician colleagues had over 500 charts in his in-basket on a regular basis, which was overwhelming and untenable.”
The use of AI in health care is rapidly growing. Tools to help hasten the process of taking notes through use of AI-generated summaries is something appealing to many doctors. Ms. Hill warned physicians to “be careful not to rely so heavily on AI that you trust it over your own words.” She noted that it can make mistakes, and the liability always remains with the clinician.
Creating time-efficient strategies
Wilfrid Noel Raby, PhD, MD, a psychiatrist in private practice in Teaneck, N.J., was formerly a psychiatrist in the substance abuse unit at Montefiore Hospital, New York. He told this news organization that he developed a system whereby he rarely had to take work home with him. “I was working only 20 hours a week, but I was usually able to do my charting during those hours, as well as seeing patients,” he said. “I scheduled my appointments and structured a little ‘buffer time’ between them so that I had time to document the first appointment before moving on to the next one.”
There were days when this wasn’t possible because there were too many patients who needed to be seen back-to-back. “So I developed my own template where I could take rapid, very standardized notes that fit into the format of the EHR and met those expectations.” Then, when he had finished seeing patients, he could quickly enter the content of his notes into the EHR. If necessary, he completed his charting on a different day.
Viwek Bisen, DO, assistant professor of psychiatry, Hackensack (N.J.) University Medical Center, is a psychiatrist in the emergency department. “My contract is based on a traditional 40-hour workweek, with 80% of my time allotted to seeing patients and 20% of my time allotted to administration.”
But the way his time actually plays out is that he’s seeing patients during about half of the 32 hours. “The rest of the time, I’m charting, speaking to family members of patients, writing notes, engaging in team meetings, and dealing with insurance companies.” Dr. Bisen has developed his own system of completing his notes while still in the hospital. “I’ve learned to be efficient and manage my time better, so I no longer have to take work home with me.”
“At the end of the day, doctors are people,” Ms. Hill said. “They are not machines. Maybe in residency and fellowship they may grind out impossible shifts with little sleep, but this pace isn’t tenable for an entire career.”
A version of this article first appeared on Medscape.com.
What’s in a day’s work? For doctors, it’s typically a mix of seeing patients and completing paperwork and follow-up. Often it extends well past the standard workday.
Dennis Hursh, JD, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, describes one overwhelmed ob.gyn. who recently consulted him for this problem.
“My client had accepted a position in a group practice where his contract stated he would be working during normal office hours, Monday through Friday, from 8 a.m. to 5 p.m. – in other words, a 40-hour workweek,” Mr. Hursh said.
But the distressed physician discovered that actually, he was working almost twice as many hours. “He’d get to work early to do charting, then see patients during the 40 hours, perhaps grabbing a quick sandwich for a few minutes – and then stay after 5 [p.m.] for a few more hours when he’d work on charts or other administrative tasks. Then he’d get something to eat, work on more charts, then go to bed, get up in the morning, and repeat.”
Mr. Hursh summarized the client’s life: “Eating, sleeping, practicing clinical medicine, and doing nonclinical tasks.”
It turned out that the 40-hour workweek included in the contract referred to patient-facing hours, not to all of the ancillary tasks that are part of practicing medicine in this day and age. “Unfortunately, this is far from an isolated story,” said Mr. Hursh.
Be aware of what’s in the contract
“The first draft of many standard physician employment contracts often omits mention of patient contact hour requirements and rather uses vague verbiage such as ‘full-time’ employment or ‘1.0 FTE’ – or full-time equivalent – without defining that term,” said Mr. Hursh. Typically, the 40 hours exclude call coverage, but most physicians understand that and, at least at first glance, it all sounds very reasonable.
But once charting, hours on the phone, arguing with managed care companies, sending in prescriptions, administrative meetings, and other tasks are thrown in, the work hours expand dramatically. Moreover, if your employer doesn’t utilize hospitalists, you may be expected to “round” outside of the 40 hours, which can be particularly burdensome if the employer admits patients to multiple hospitals.
Amanda Hill, JD, owner of Hill Health Law based in Austin, Texas, told this news organization that this predicament isn’t unique to physicians. Exempt employees who don’t clock in and out are often expected to work overtime – that is, to “work as long as it takes to get the job done.” It can affect NPs, PAs, and many others in the health care space. But the number of tasks that fall upon a doctor’s shoulders and the fact that patients’ health and lives are at stake up the ante and make the situation far more difficult for doctors than for employees in other industries.
So it’s important to nail down precise terms in the contract and, if possible, negotiate for a more humane schedule by specifying how the working hours will be used.
“It’s true that a 1.0 FTE definition is too vague,” Ms. Hill said. “I’ve negotiated a lot of contracts where we nail down in writing that the in-office schedule equals 34 hours per week, so the physician is guaranteed an additional 6 hours for administrative time.”
Mr. Hursh usually asks for 32 hours of patient contact per week, which leaves 1 full day per week to catch up on basic administrative tasks. “It’s important for employers to recognize that seeing patients isn’t the only thing a doctor does and there’s a lot of work in addition to face-to-face time,” he said.
But he hasn’t always been successful. One physician client was seeking a workweek consisting of 36 patient contact hours, “which is 90% of the usual FTE of a 40-hour week,” said Mr. Hursh. “But the employer called it ‘part-time,’ as if the doctor were planning to be lying in the sun for the other 4 hours.”
The client decided to accept a 10% pay cut and 10% less vacation to guarantee that she had those extra hours for administrative tasks. “She’s probably working way more than 36 hours a week, but maybe closer to 50 or 60 instead of 70 or more,” he said.
Clarify call coverage
Call coverage is typically not included in the hours a physician is contracted to work on a weekly basis. “Most contracts have call, and it’s usually evenly distributed among parties in a practice, but call can expand if another doctor is out sick, for example,” said Ms. Hill.
Sometimes the language in the contract is vague regarding call coverage. “I ask, how many shifts per year is the doctor is expected to work? Then, I try to negotiate extra pay if more shifts arise,” she said. “The hospital or practice may not demand extra call because they don’t want to pay extra money to the physician.”
On the other hand, some physicians may be eager to take extra call if it means extra income.
Ms. Hill stated that one of her clients was being paid as a “part-time, 2-day-a-week provider” but was asked to be on call and take night and weekend work. When you added it all up, she was putting in almost 30 hours a week.
“This is abusive to a provider that works so hard for patients,” Ms. Hill said. “We have to protect them through the contract language, so they have something hard and fast to point to when their administrator pushes them too hard. Doctors should get value for their time.”
Ms. Hill and her client pushed for more money, and the employer gave in. “All we had to do was to point out how many hours she was actually working. She didn’t mind all the extra call, but she wanted to be compensated.” The doctor’s salary was hiked by $25,000.
Differences in specialties and settings
There are some specialties where it might be easier to have more defined hours, while other specialties are more challenging. Anu Murthy, Esq., an attorney and associate contract review specialist at Contract Diagnostics (a national firm that reviews physician contracts) told this news organization that the work of hospitalists, intensivists, and emergency department physicians, for example, is done in shifts, which tend to be fixed hours.
“They need to get their charting completed so that whoever takes over on the next shift has access to the most recent notes about the patient,” she said. By contrast, surgeons can’t always account for how long a given surgery will take. “It could be as long as 9 hours,” she said. Notes need to be written immediately for the sake of the patient’s postsurgical care.
Dermatologists tend to deal with fewer emergencies, compared with other specialists, and it’s easier for their patients to be slotted into an organized schedule. On the other hand, primary care doctors – internists, family practice physicians, and pediatricians – may be seeing 40-50 patients a day, one every 15 minutes.
Practice setting also makes a difference, said Ms. Murthy. Veterans Administration (VA) hospitals or government-run clinics tend to have more rigidly defined hours, compared with other settings, so if you’re in a VA hospital or government-run clinic, work-life balance tends to be better.
Physicians who work remotely via telehealth also tend to have a better work-life balance, compared with those who see patients in person, Ms. Murthy said. But the difference may be in not having to spend extra time commuting to work or interacting with others in the work environment, since some research has suggested that telehealth physicians may actually spend more time engaged in charting after hours, compared with their in-person counterparts.
Using scribes to maximize your time
Elliott Trotter, MD, is an emergency medicine physician, associate clinical professor of emergency medicine at Texas Christian University Medical Schools, and founder of the ScribeNest, a Texas-based company that trains health care scribes. He told this news organization that there are ways to maximize one’s time during shifts so that much of the charting can be accomplished during working hours.
“About 28 years ago, I realized that the documentation load for physicians was enormous and at that time I developed the Modern Scribe, using premed students for ‘elbow support’ to help with the workload by documenting the ED encounters in real time during the encounter so I wouldn’t have to do so later.”
Over the years, as EHRs have become more ubiquitous and onerous, the role of the scribe has “evolved from a luxury to a necessity,” said Dr. Trotter. The scribes can actually record the encounter directly into the EHR so that the physician doesn’t have to do so later and doesn’t have to look at a computer screen but can look at the patient during the encounter.
“This enhances communication and has been shown to improve patient care,” he said.
Dr. Trotter said he rarely, if ever, needs to do documentation after hours. “But one of my physician colleagues had over 500 charts in his in-basket on a regular basis, which was overwhelming and untenable.”
The use of AI in health care is rapidly growing. Tools to help hasten the process of taking notes through use of AI-generated summaries is something appealing to many doctors. Ms. Hill warned physicians to “be careful not to rely so heavily on AI that you trust it over your own words.” She noted that it can make mistakes, and the liability always remains with the clinician.
Creating time-efficient strategies
Wilfrid Noel Raby, PhD, MD, a psychiatrist in private practice in Teaneck, N.J., was formerly a psychiatrist in the substance abuse unit at Montefiore Hospital, New York. He told this news organization that he developed a system whereby he rarely had to take work home with him. “I was working only 20 hours a week, but I was usually able to do my charting during those hours, as well as seeing patients,” he said. “I scheduled my appointments and structured a little ‘buffer time’ between them so that I had time to document the first appointment before moving on to the next one.”
There were days when this wasn’t possible because there were too many patients who needed to be seen back-to-back. “So I developed my own template where I could take rapid, very standardized notes that fit into the format of the EHR and met those expectations.” Then, when he had finished seeing patients, he could quickly enter the content of his notes into the EHR. If necessary, he completed his charting on a different day.
Viwek Bisen, DO, assistant professor of psychiatry, Hackensack (N.J.) University Medical Center, is a psychiatrist in the emergency department. “My contract is based on a traditional 40-hour workweek, with 80% of my time allotted to seeing patients and 20% of my time allotted to administration.”
But the way his time actually plays out is that he’s seeing patients during about half of the 32 hours. “The rest of the time, I’m charting, speaking to family members of patients, writing notes, engaging in team meetings, and dealing with insurance companies.” Dr. Bisen has developed his own system of completing his notes while still in the hospital. “I’ve learned to be efficient and manage my time better, so I no longer have to take work home with me.”
“At the end of the day, doctors are people,” Ms. Hill said. “They are not machines. Maybe in residency and fellowship they may grind out impossible shifts with little sleep, but this pace isn’t tenable for an entire career.”
A version of this article first appeared on Medscape.com.
What’s in a day’s work? For doctors, it’s typically a mix of seeing patients and completing paperwork and follow-up. Often it extends well past the standard workday.
Dennis Hursh, JD, managing partner of Physician Agreements Health Law, a Pennsylvania-based law firm that represents physicians, describes one overwhelmed ob.gyn. who recently consulted him for this problem.
“My client had accepted a position in a group practice where his contract stated he would be working during normal office hours, Monday through Friday, from 8 a.m. to 5 p.m. – in other words, a 40-hour workweek,” Mr. Hursh said.
But the distressed physician discovered that actually, he was working almost twice as many hours. “He’d get to work early to do charting, then see patients during the 40 hours, perhaps grabbing a quick sandwich for a few minutes – and then stay after 5 [p.m.] for a few more hours when he’d work on charts or other administrative tasks. Then he’d get something to eat, work on more charts, then go to bed, get up in the morning, and repeat.”
Mr. Hursh summarized the client’s life: “Eating, sleeping, practicing clinical medicine, and doing nonclinical tasks.”
It turned out that the 40-hour workweek included in the contract referred to patient-facing hours, not to all of the ancillary tasks that are part of practicing medicine in this day and age. “Unfortunately, this is far from an isolated story,” said Mr. Hursh.
Be aware of what’s in the contract
“The first draft of many standard physician employment contracts often omits mention of patient contact hour requirements and rather uses vague verbiage such as ‘full-time’ employment or ‘1.0 FTE’ – or full-time equivalent – without defining that term,” said Mr. Hursh. Typically, the 40 hours exclude call coverage, but most physicians understand that and, at least at first glance, it all sounds very reasonable.
But once charting, hours on the phone, arguing with managed care companies, sending in prescriptions, administrative meetings, and other tasks are thrown in, the work hours expand dramatically. Moreover, if your employer doesn’t utilize hospitalists, you may be expected to “round” outside of the 40 hours, which can be particularly burdensome if the employer admits patients to multiple hospitals.
Amanda Hill, JD, owner of Hill Health Law based in Austin, Texas, told this news organization that this predicament isn’t unique to physicians. Exempt employees who don’t clock in and out are often expected to work overtime – that is, to “work as long as it takes to get the job done.” It can affect NPs, PAs, and many others in the health care space. But the number of tasks that fall upon a doctor’s shoulders and the fact that patients’ health and lives are at stake up the ante and make the situation far more difficult for doctors than for employees in other industries.
So it’s important to nail down precise terms in the contract and, if possible, negotiate for a more humane schedule by specifying how the working hours will be used.
“It’s true that a 1.0 FTE definition is too vague,” Ms. Hill said. “I’ve negotiated a lot of contracts where we nail down in writing that the in-office schedule equals 34 hours per week, so the physician is guaranteed an additional 6 hours for administrative time.”
Mr. Hursh usually asks for 32 hours of patient contact per week, which leaves 1 full day per week to catch up on basic administrative tasks. “It’s important for employers to recognize that seeing patients isn’t the only thing a doctor does and there’s a lot of work in addition to face-to-face time,” he said.
But he hasn’t always been successful. One physician client was seeking a workweek consisting of 36 patient contact hours, “which is 90% of the usual FTE of a 40-hour week,” said Mr. Hursh. “But the employer called it ‘part-time,’ as if the doctor were planning to be lying in the sun for the other 4 hours.”
The client decided to accept a 10% pay cut and 10% less vacation to guarantee that she had those extra hours for administrative tasks. “She’s probably working way more than 36 hours a week, but maybe closer to 50 or 60 instead of 70 or more,” he said.
Clarify call coverage
Call coverage is typically not included in the hours a physician is contracted to work on a weekly basis. “Most contracts have call, and it’s usually evenly distributed among parties in a practice, but call can expand if another doctor is out sick, for example,” said Ms. Hill.
Sometimes the language in the contract is vague regarding call coverage. “I ask, how many shifts per year is the doctor is expected to work? Then, I try to negotiate extra pay if more shifts arise,” she said. “The hospital or practice may not demand extra call because they don’t want to pay extra money to the physician.”
On the other hand, some physicians may be eager to take extra call if it means extra income.
Ms. Hill stated that one of her clients was being paid as a “part-time, 2-day-a-week provider” but was asked to be on call and take night and weekend work. When you added it all up, she was putting in almost 30 hours a week.
“This is abusive to a provider that works so hard for patients,” Ms. Hill said. “We have to protect them through the contract language, so they have something hard and fast to point to when their administrator pushes them too hard. Doctors should get value for their time.”
Ms. Hill and her client pushed for more money, and the employer gave in. “All we had to do was to point out how many hours she was actually working. She didn’t mind all the extra call, but she wanted to be compensated.” The doctor’s salary was hiked by $25,000.
Differences in specialties and settings
There are some specialties where it might be easier to have more defined hours, while other specialties are more challenging. Anu Murthy, Esq., an attorney and associate contract review specialist at Contract Diagnostics (a national firm that reviews physician contracts) told this news organization that the work of hospitalists, intensivists, and emergency department physicians, for example, is done in shifts, which tend to be fixed hours.
“They need to get their charting completed so that whoever takes over on the next shift has access to the most recent notes about the patient,” she said. By contrast, surgeons can’t always account for how long a given surgery will take. “It could be as long as 9 hours,” she said. Notes need to be written immediately for the sake of the patient’s postsurgical care.
Dermatologists tend to deal with fewer emergencies, compared with other specialists, and it’s easier for their patients to be slotted into an organized schedule. On the other hand, primary care doctors – internists, family practice physicians, and pediatricians – may be seeing 40-50 patients a day, one every 15 minutes.
Practice setting also makes a difference, said Ms. Murthy. Veterans Administration (VA) hospitals or government-run clinics tend to have more rigidly defined hours, compared with other settings, so if you’re in a VA hospital or government-run clinic, work-life balance tends to be better.
Physicians who work remotely via telehealth also tend to have a better work-life balance, compared with those who see patients in person, Ms. Murthy said. But the difference may be in not having to spend extra time commuting to work or interacting with others in the work environment, since some research has suggested that telehealth physicians may actually spend more time engaged in charting after hours, compared with their in-person counterparts.
Using scribes to maximize your time
Elliott Trotter, MD, is an emergency medicine physician, associate clinical professor of emergency medicine at Texas Christian University Medical Schools, and founder of the ScribeNest, a Texas-based company that trains health care scribes. He told this news organization that there are ways to maximize one’s time during shifts so that much of the charting can be accomplished during working hours.
“About 28 years ago, I realized that the documentation load for physicians was enormous and at that time I developed the Modern Scribe, using premed students for ‘elbow support’ to help with the workload by documenting the ED encounters in real time during the encounter so I wouldn’t have to do so later.”
Over the years, as EHRs have become more ubiquitous and onerous, the role of the scribe has “evolved from a luxury to a necessity,” said Dr. Trotter. The scribes can actually record the encounter directly into the EHR so that the physician doesn’t have to do so later and doesn’t have to look at a computer screen but can look at the patient during the encounter.
“This enhances communication and has been shown to improve patient care,” he said.
Dr. Trotter said he rarely, if ever, needs to do documentation after hours. “But one of my physician colleagues had over 500 charts in his in-basket on a regular basis, which was overwhelming and untenable.”
The use of AI in health care is rapidly growing. Tools to help hasten the process of taking notes through use of AI-generated summaries is something appealing to many doctors. Ms. Hill warned physicians to “be careful not to rely so heavily on AI that you trust it over your own words.” She noted that it can make mistakes, and the liability always remains with the clinician.
Creating time-efficient strategies
Wilfrid Noel Raby, PhD, MD, a psychiatrist in private practice in Teaneck, N.J., was formerly a psychiatrist in the substance abuse unit at Montefiore Hospital, New York. He told this news organization that he developed a system whereby he rarely had to take work home with him. “I was working only 20 hours a week, but I was usually able to do my charting during those hours, as well as seeing patients,” he said. “I scheduled my appointments and structured a little ‘buffer time’ between them so that I had time to document the first appointment before moving on to the next one.”
There were days when this wasn’t possible because there were too many patients who needed to be seen back-to-back. “So I developed my own template where I could take rapid, very standardized notes that fit into the format of the EHR and met those expectations.” Then, when he had finished seeing patients, he could quickly enter the content of his notes into the EHR. If necessary, he completed his charting on a different day.
Viwek Bisen, DO, assistant professor of psychiatry, Hackensack (N.J.) University Medical Center, is a psychiatrist in the emergency department. “My contract is based on a traditional 40-hour workweek, with 80% of my time allotted to seeing patients and 20% of my time allotted to administration.”
But the way his time actually plays out is that he’s seeing patients during about half of the 32 hours. “The rest of the time, I’m charting, speaking to family members of patients, writing notes, engaging in team meetings, and dealing with insurance companies.” Dr. Bisen has developed his own system of completing his notes while still in the hospital. “I’ve learned to be efficient and manage my time better, so I no longer have to take work home with me.”
“At the end of the day, doctors are people,” Ms. Hill said. “They are not machines. Maybe in residency and fellowship they may grind out impossible shifts with little sleep, but this pace isn’t tenable for an entire career.”
A version of this article first appeared on Medscape.com.
Gaps persist in awareness, treatment of high LDL cholesterol
TOPLINE:
The prevalence of elevated LDL cholesterol (LDL-C) has declined over the past 2 decades, but 1 in 17 Americans still have a level of 160-189 mg/dL, and 1 in 48 have a level of at least 190 mg/dL, new research shows. Among people with the higher LDL-C level, one in four are both unaware and untreated, the authors report.
METHODOLOGY:
- Using data on 23,667 adult participants in the National Health and Nutrition Examination Survey conducted from 1999 to 2020, researchers identified 1,851 (7.8%) with an LDL-C level of 160-189 mg/dL and 669 (2.8%) with an LDL-C level of at least 190 mg/dL.
- Individuals were classified as “unaware” if they had never had their LDL-C measured or had never been informed of having elevated LDL-C and as “untreated” if their medications didn’t include a statin, ezetimibe, a bile acid sequestrant, or a proprotein convertase subtilisin/kexin type 9 inhibitor.
- The authors compared the prevalence of “unaware” and “untreated” by age, sex, race and ethnicity, educational attainment, poverty index, and insurance status.
TAKEAWAY:
- During the study period, the age-adjusted prevalence of an LDL-C level of 160-189 mg/dL declined from 12.4% (95% confidence interval, 10.0%-15.3%), representing 21.5 million U.S. adults, to 6.1% (95% CI, 4.8%-7.6%), representing 14.0 million adults (P < .001).
- The age-adjusted prevalence of an LDL-C level of at least 190 mg/dL declined from 3.8% (95% CI, 2.8%-5.2%), representing 6.6 million adults, to 2.1% (95% CI, 1.4%-3.0%), representing 4.8 million adults (P = .001).
- Among those with an LDL-C level of 160-189 mg/dL, the proportion of who were unaware and untreated declined from 52.1% to 42.7%, and among those with an LDL-C level of at least 190 mg/dL, it declined from 40.8% to 26.8%.
- Being unaware and untreated was more common in younger adults, men, racial and ethnic minority groups, those with lower educational attainment, those with lower income, and those without health insurance.
IN PRACTICE:
The lack of awareness and treatment of high LDL-C uncovered by the study “may be due to difficulties accessing primary care, low rates of screening in primary care, lack of consensus on screening recommendations, insufficient emphasis on LDL-C as a quality measure, and hesitance to treat asymptomatic individuals,” the authors concluded.
SOURCE:
The research was led by Ahmed Sayed, MBBS, faculty of medicine, Ain Shams University, Cairo, Egypt. It was published online in JAMA Cardiology.
LIMITATIONS:
The analysis was limited by a small number of participants with LDL-C levels of at least 190 mg/dL, possible nonresponse bias, and dependency on participant recall of whether LDL-C was previously measured. The inclusion of pregnant women may have influenced LDL-C levels.
DISCLOSURES:
Dr. Sayed has no relevant conflict of interest. The disclosures of the other authors are listed in the original publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
The prevalence of elevated LDL cholesterol (LDL-C) has declined over the past 2 decades, but 1 in 17 Americans still have a level of 160-189 mg/dL, and 1 in 48 have a level of at least 190 mg/dL, new research shows. Among people with the higher LDL-C level, one in four are both unaware and untreated, the authors report.
METHODOLOGY:
- Using data on 23,667 adult participants in the National Health and Nutrition Examination Survey conducted from 1999 to 2020, researchers identified 1,851 (7.8%) with an LDL-C level of 160-189 mg/dL and 669 (2.8%) with an LDL-C level of at least 190 mg/dL.
- Individuals were classified as “unaware” if they had never had their LDL-C measured or had never been informed of having elevated LDL-C and as “untreated” if their medications didn’t include a statin, ezetimibe, a bile acid sequestrant, or a proprotein convertase subtilisin/kexin type 9 inhibitor.
- The authors compared the prevalence of “unaware” and “untreated” by age, sex, race and ethnicity, educational attainment, poverty index, and insurance status.
TAKEAWAY:
- During the study period, the age-adjusted prevalence of an LDL-C level of 160-189 mg/dL declined from 12.4% (95% confidence interval, 10.0%-15.3%), representing 21.5 million U.S. adults, to 6.1% (95% CI, 4.8%-7.6%), representing 14.0 million adults (P < .001).
- The age-adjusted prevalence of an LDL-C level of at least 190 mg/dL declined from 3.8% (95% CI, 2.8%-5.2%), representing 6.6 million adults, to 2.1% (95% CI, 1.4%-3.0%), representing 4.8 million adults (P = .001).
- Among those with an LDL-C level of 160-189 mg/dL, the proportion of who were unaware and untreated declined from 52.1% to 42.7%, and among those with an LDL-C level of at least 190 mg/dL, it declined from 40.8% to 26.8%.
- Being unaware and untreated was more common in younger adults, men, racial and ethnic minority groups, those with lower educational attainment, those with lower income, and those without health insurance.
IN PRACTICE:
The lack of awareness and treatment of high LDL-C uncovered by the study “may be due to difficulties accessing primary care, low rates of screening in primary care, lack of consensus on screening recommendations, insufficient emphasis on LDL-C as a quality measure, and hesitance to treat asymptomatic individuals,” the authors concluded.
SOURCE:
The research was led by Ahmed Sayed, MBBS, faculty of medicine, Ain Shams University, Cairo, Egypt. It was published online in JAMA Cardiology.
LIMITATIONS:
The analysis was limited by a small number of participants with LDL-C levels of at least 190 mg/dL, possible nonresponse bias, and dependency on participant recall of whether LDL-C was previously measured. The inclusion of pregnant women may have influenced LDL-C levels.
DISCLOSURES:
Dr. Sayed has no relevant conflict of interest. The disclosures of the other authors are listed in the original publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
The prevalence of elevated LDL cholesterol (LDL-C) has declined over the past 2 decades, but 1 in 17 Americans still have a level of 160-189 mg/dL, and 1 in 48 have a level of at least 190 mg/dL, new research shows. Among people with the higher LDL-C level, one in four are both unaware and untreated, the authors report.
METHODOLOGY:
- Using data on 23,667 adult participants in the National Health and Nutrition Examination Survey conducted from 1999 to 2020, researchers identified 1,851 (7.8%) with an LDL-C level of 160-189 mg/dL and 669 (2.8%) with an LDL-C level of at least 190 mg/dL.
- Individuals were classified as “unaware” if they had never had their LDL-C measured or had never been informed of having elevated LDL-C and as “untreated” if their medications didn’t include a statin, ezetimibe, a bile acid sequestrant, or a proprotein convertase subtilisin/kexin type 9 inhibitor.
- The authors compared the prevalence of “unaware” and “untreated” by age, sex, race and ethnicity, educational attainment, poverty index, and insurance status.
TAKEAWAY:
- During the study period, the age-adjusted prevalence of an LDL-C level of 160-189 mg/dL declined from 12.4% (95% confidence interval, 10.0%-15.3%), representing 21.5 million U.S. adults, to 6.1% (95% CI, 4.8%-7.6%), representing 14.0 million adults (P < .001).
- The age-adjusted prevalence of an LDL-C level of at least 190 mg/dL declined from 3.8% (95% CI, 2.8%-5.2%), representing 6.6 million adults, to 2.1% (95% CI, 1.4%-3.0%), representing 4.8 million adults (P = .001).
- Among those with an LDL-C level of 160-189 mg/dL, the proportion of who were unaware and untreated declined from 52.1% to 42.7%, and among those with an LDL-C level of at least 190 mg/dL, it declined from 40.8% to 26.8%.
- Being unaware and untreated was more common in younger adults, men, racial and ethnic minority groups, those with lower educational attainment, those with lower income, and those without health insurance.
IN PRACTICE:
The lack of awareness and treatment of high LDL-C uncovered by the study “may be due to difficulties accessing primary care, low rates of screening in primary care, lack of consensus on screening recommendations, insufficient emphasis on LDL-C as a quality measure, and hesitance to treat asymptomatic individuals,” the authors concluded.
SOURCE:
The research was led by Ahmed Sayed, MBBS, faculty of medicine, Ain Shams University, Cairo, Egypt. It was published online in JAMA Cardiology.
LIMITATIONS:
The analysis was limited by a small number of participants with LDL-C levels of at least 190 mg/dL, possible nonresponse bias, and dependency on participant recall of whether LDL-C was previously measured. The inclusion of pregnant women may have influenced LDL-C levels.
DISCLOSURES:
Dr. Sayed has no relevant conflict of interest. The disclosures of the other authors are listed in the original publication.
A version of this article first appeared on Medscape.com.
Drug-coated balloon beats conventional angioplasty for high-risk patients with in-stent restenosis
SAN FRANCISCO – For the treatment of coronary artery in-stent restenosis, angioplasty with a drug-coated balloon (AGENT DCB; Boston Scientific) was superior to conventional balloon angioplasty in preventing target lesion failure at 1 year in a high-risk patient population.
Approximate 50% reductions in the rates of target lesion restenosis and target vessel myocardial infarction (MI) accounted for the superior findings with the AGENT DCB over conventional balloon angioplasty.
Robert Yeh, MD, of Beth Israel Deaconess Medical Center in Boston reported at the annual Transcatheter Cardiovascular Therapeutics congress. “This represented a 38% relative risk reduction as well as a 10% absolute risk reduction in the endpoint. The P value for superiority was 0.0063, highly statistically significant.”
In-stent restenosis is clinically challenging and accounts for about 10% of all percutaneous coronary interventions. “Sometimes these patients have multiple layers, and that could be a third or fourth layer of stent, something that we try to avoid,” he said.
Drug-coated balloons, which are not currently approved in the United States, can deliver drugs that inhibit blockages from reforming, “without leaving additional layers of metal behind,” he added. Such devices are already available in Europe and Japan.
AGENT IDE was a prospective, multicenter, superiority trial that randomly assigned 480 patients 2:1 to the AGENT DCB (n = 321) or to conventional balloon angioplasty (n = 159). Randomization occurred after successful pre-dilation of the target vessel.
The trial included patients with in-stent restenosis previously treated with a bare metal or a drug-eluting stent with lesion lengths < 26 mm (reference vessel diameter: > 2 mm to ≤ 4), and percent diameter stenosis of more than 70% if they were asymptomatic or of more than 50% if they were symptomatic. Patients were excluded if they had a recent ST-elevation MI, bifurcation, saphenous vein or arterial graft, or thrombus in the target vessel.
All received dual antiplatelet therapy for at least 1 month and then antiplatelet monotherapy for the duration of the trial. The primary endpoint was target lesion failure at 1 year, a composite of target lesion restenosis, target vessel-related MI, or cardiac death. More than 93% of patients in each arm were available for evaluation of the primary endpoint.
The two groups were well balanced at baseline: Approximate age was 68 years, 27% were women, and three quarters were White. Approximately 28%-32% had had a prior coronary artery bypass graft, 20%-22% had previous heart failure, and about 22% had a history of left main coronary artery disease. Half had diabetes, and about half had stable angina.
Multiple stent layers were common in 43% of each group. Stenosis diameter was about 65% at baseline for the two groups and was reduced to 22% post procedure.
Outcomes all favored AGENT DCB
In the AGENT DCB group, the technical success rate was 92.9% vs 89.3% for balloon angioplasty. Intravascular imaging was used during the procedure in 72.3% of DCB cases and in 76.7% of balloon cases.
Besides demonstrating a nearly 38% reduction in the primary endpoint of target lesion failure at 1 year for the DCB over conventional balloon angioplasty, DCB nearly halved the rate of target lesion revascularization and target vessel MI and was superior on other measures of clinical outcome.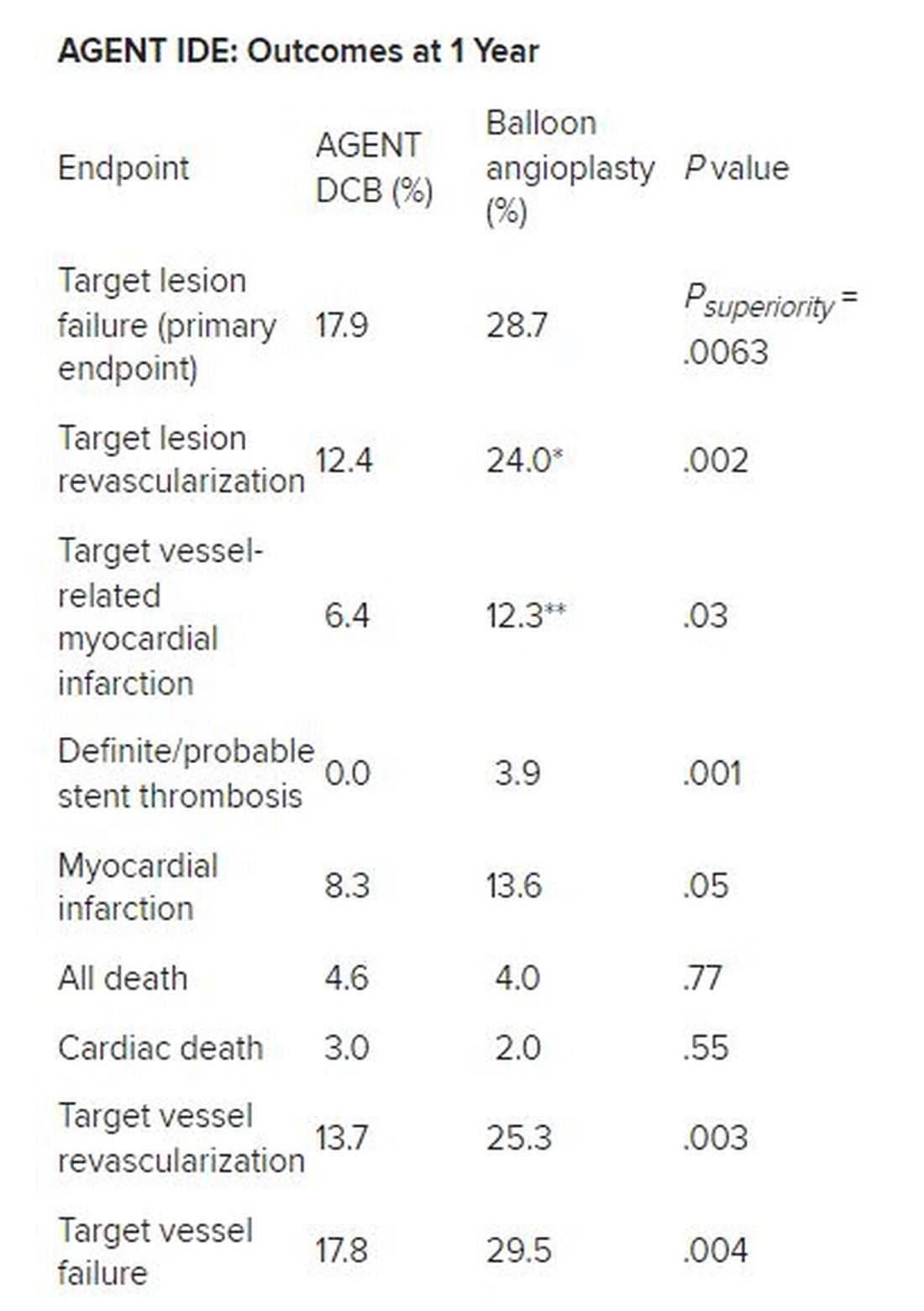
*Hazard ratio, 0.49; 95% CI, 0.31-0.79; ** HR, 0.51; 95% CI, 0.27-0.95
There was no stent rethrombosis with the DCB vs 3.9% with the conventional balloon angioplasty. Of note, there were no differences between the groups in terms of cardiac or noncardiac death.
Subgroup analyses of the primary outcome in terms of sex, age, diabetes, vessel size, or single or multiple stent layers all trended in favor of AGENT DCB but were not statistically significant for interaction.
The study is being expanded to include 600 patients. This device is a US Food and Drug Administration–designated breakthrough device, “and this pivotal trial will be the primary evidence used to support FDA approval,” Dr. Yeh said. “And given the marked superiority over conventional balloon angioplasty, I believe that the AGENT DCB is likely to become an important new treatment option for patients with coronary stenosis in the United States.”
Long overdue
Róisín Colleran, MBBCh, of the Cardiovascular Research Institute Dublin at Mater Private Hospital in Ireland, the designated discussant, first congratulated Dr. Yeh and his coinvestigators on the study’s conduct and findings.
“This study is long overdue,” she said. As Dr. Yeh noted, about 10% of PCI procedures are done for in-stent restenosis, Dr. Colleran said, but in 2023, there is still no coronary drug eluting balloon approved for this indication in the US, despite the class 1 recommendation in the 2014 European guidelines.
She pointed to the trial results, saying they are “clear...a significant reduction in target lesion failure driven by halving in rates of both target lesion revascularization and target vessel MI.”
Strengths of the study are it is the largest of its kind to date, with 480 patients, conducted at 40 US centers, using device-specific endpoints. There was a “very high” intravascular imaging rate of 75% in a cohort with a high risk for in-stent restenosis, consisting of 50% of patients with diabetes and more than 40% with multiple stents.
“The main limitation is the choice of comparator,” Dr. Colleran said. Balloon angioplasty is inferior to both stenting and drug coated balloon therapy for treatment of in-stent restenosis but is the standard of care in the United States, she noted. “I think...for regulatory reasons this was the comparator chosen,” she said.
“I think the implications are clear,” Dr. Colleran added. “This trial should provide a basis for regulatory approval of the drug coated balloon treatment of in-stent restenosis in the U.S. and finally provide this as an available treatment option for such patients.”
Dr. Yeh reported receiving grant/research support from Abbott Vascular, BD Bard, Boston Scientific, Cook Medical, Philips Medical, and Medtronic, and consulting for Abbott Vascular, Boston Scientific, CathWorks, Elixir Medical, Infraredx, Medtronic, Shockwave Medical, and Zol. Dr. Colleran had no disclosures. The trial was supported by Boston Scientific.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – For the treatment of coronary artery in-stent restenosis, angioplasty with a drug-coated balloon (AGENT DCB; Boston Scientific) was superior to conventional balloon angioplasty in preventing target lesion failure at 1 year in a high-risk patient population.
Approximate 50% reductions in the rates of target lesion restenosis and target vessel myocardial infarction (MI) accounted for the superior findings with the AGENT DCB over conventional balloon angioplasty.
Robert Yeh, MD, of Beth Israel Deaconess Medical Center in Boston reported at the annual Transcatheter Cardiovascular Therapeutics congress. “This represented a 38% relative risk reduction as well as a 10% absolute risk reduction in the endpoint. The P value for superiority was 0.0063, highly statistically significant.”
In-stent restenosis is clinically challenging and accounts for about 10% of all percutaneous coronary interventions. “Sometimes these patients have multiple layers, and that could be a third or fourth layer of stent, something that we try to avoid,” he said.
Drug-coated balloons, which are not currently approved in the United States, can deliver drugs that inhibit blockages from reforming, “without leaving additional layers of metal behind,” he added. Such devices are already available in Europe and Japan.
AGENT IDE was a prospective, multicenter, superiority trial that randomly assigned 480 patients 2:1 to the AGENT DCB (n = 321) or to conventional balloon angioplasty (n = 159). Randomization occurred after successful pre-dilation of the target vessel.
The trial included patients with in-stent restenosis previously treated with a bare metal or a drug-eluting stent with lesion lengths < 26 mm (reference vessel diameter: > 2 mm to ≤ 4), and percent diameter stenosis of more than 70% if they were asymptomatic or of more than 50% if they were symptomatic. Patients were excluded if they had a recent ST-elevation MI, bifurcation, saphenous vein or arterial graft, or thrombus in the target vessel.
All received dual antiplatelet therapy for at least 1 month and then antiplatelet monotherapy for the duration of the trial. The primary endpoint was target lesion failure at 1 year, a composite of target lesion restenosis, target vessel-related MI, or cardiac death. More than 93% of patients in each arm were available for evaluation of the primary endpoint.
The two groups were well balanced at baseline: Approximate age was 68 years, 27% were women, and three quarters were White. Approximately 28%-32% had had a prior coronary artery bypass graft, 20%-22% had previous heart failure, and about 22% had a history of left main coronary artery disease. Half had diabetes, and about half had stable angina.
Multiple stent layers were common in 43% of each group. Stenosis diameter was about 65% at baseline for the two groups and was reduced to 22% post procedure.
Outcomes all favored AGENT DCB
In the AGENT DCB group, the technical success rate was 92.9% vs 89.3% for balloon angioplasty. Intravascular imaging was used during the procedure in 72.3% of DCB cases and in 76.7% of balloon cases.
Besides demonstrating a nearly 38% reduction in the primary endpoint of target lesion failure at 1 year for the DCB over conventional balloon angioplasty, DCB nearly halved the rate of target lesion revascularization and target vessel MI and was superior on other measures of clinical outcome.
*Hazard ratio, 0.49; 95% CI, 0.31-0.79; ** HR, 0.51; 95% CI, 0.27-0.95
There was no stent rethrombosis with the DCB vs 3.9% with the conventional balloon angioplasty. Of note, there were no differences between the groups in terms of cardiac or noncardiac death.
Subgroup analyses of the primary outcome in terms of sex, age, diabetes, vessel size, or single or multiple stent layers all trended in favor of AGENT DCB but were not statistically significant for interaction.
The study is being expanded to include 600 patients. This device is a US Food and Drug Administration–designated breakthrough device, “and this pivotal trial will be the primary evidence used to support FDA approval,” Dr. Yeh said. “And given the marked superiority over conventional balloon angioplasty, I believe that the AGENT DCB is likely to become an important new treatment option for patients with coronary stenosis in the United States.”
Long overdue
Róisín Colleran, MBBCh, of the Cardiovascular Research Institute Dublin at Mater Private Hospital in Ireland, the designated discussant, first congratulated Dr. Yeh and his coinvestigators on the study’s conduct and findings.
“This study is long overdue,” she said. As Dr. Yeh noted, about 10% of PCI procedures are done for in-stent restenosis, Dr. Colleran said, but in 2023, there is still no coronary drug eluting balloon approved for this indication in the US, despite the class 1 recommendation in the 2014 European guidelines.
She pointed to the trial results, saying they are “clear...a significant reduction in target lesion failure driven by halving in rates of both target lesion revascularization and target vessel MI.”
Strengths of the study are it is the largest of its kind to date, with 480 patients, conducted at 40 US centers, using device-specific endpoints. There was a “very high” intravascular imaging rate of 75% in a cohort with a high risk for in-stent restenosis, consisting of 50% of patients with diabetes and more than 40% with multiple stents.
“The main limitation is the choice of comparator,” Dr. Colleran said. Balloon angioplasty is inferior to both stenting and drug coated balloon therapy for treatment of in-stent restenosis but is the standard of care in the United States, she noted. “I think...for regulatory reasons this was the comparator chosen,” she said.
“I think the implications are clear,” Dr. Colleran added. “This trial should provide a basis for regulatory approval of the drug coated balloon treatment of in-stent restenosis in the U.S. and finally provide this as an available treatment option for such patients.”
Dr. Yeh reported receiving grant/research support from Abbott Vascular, BD Bard, Boston Scientific, Cook Medical, Philips Medical, and Medtronic, and consulting for Abbott Vascular, Boston Scientific, CathWorks, Elixir Medical, Infraredx, Medtronic, Shockwave Medical, and Zol. Dr. Colleran had no disclosures. The trial was supported by Boston Scientific.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – For the treatment of coronary artery in-stent restenosis, angioplasty with a drug-coated balloon (AGENT DCB; Boston Scientific) was superior to conventional balloon angioplasty in preventing target lesion failure at 1 year in a high-risk patient population.
Approximate 50% reductions in the rates of target lesion restenosis and target vessel myocardial infarction (MI) accounted for the superior findings with the AGENT DCB over conventional balloon angioplasty.
Robert Yeh, MD, of Beth Israel Deaconess Medical Center in Boston reported at the annual Transcatheter Cardiovascular Therapeutics congress. “This represented a 38% relative risk reduction as well as a 10% absolute risk reduction in the endpoint. The P value for superiority was 0.0063, highly statistically significant.”
In-stent restenosis is clinically challenging and accounts for about 10% of all percutaneous coronary interventions. “Sometimes these patients have multiple layers, and that could be a third or fourth layer of stent, something that we try to avoid,” he said.
Drug-coated balloons, which are not currently approved in the United States, can deliver drugs that inhibit blockages from reforming, “without leaving additional layers of metal behind,” he added. Such devices are already available in Europe and Japan.
AGENT IDE was a prospective, multicenter, superiority trial that randomly assigned 480 patients 2:1 to the AGENT DCB (n = 321) or to conventional balloon angioplasty (n = 159). Randomization occurred after successful pre-dilation of the target vessel.
The trial included patients with in-stent restenosis previously treated with a bare metal or a drug-eluting stent with lesion lengths < 26 mm (reference vessel diameter: > 2 mm to ≤ 4), and percent diameter stenosis of more than 70% if they were asymptomatic or of more than 50% if they were symptomatic. Patients were excluded if they had a recent ST-elevation MI, bifurcation, saphenous vein or arterial graft, or thrombus in the target vessel.
All received dual antiplatelet therapy for at least 1 month and then antiplatelet monotherapy for the duration of the trial. The primary endpoint was target lesion failure at 1 year, a composite of target lesion restenosis, target vessel-related MI, or cardiac death. More than 93% of patients in each arm were available for evaluation of the primary endpoint.
The two groups were well balanced at baseline: Approximate age was 68 years, 27% were women, and three quarters were White. Approximately 28%-32% had had a prior coronary artery bypass graft, 20%-22% had previous heart failure, and about 22% had a history of left main coronary artery disease. Half had diabetes, and about half had stable angina.
Multiple stent layers were common in 43% of each group. Stenosis diameter was about 65% at baseline for the two groups and was reduced to 22% post procedure.
Outcomes all favored AGENT DCB
In the AGENT DCB group, the technical success rate was 92.9% vs 89.3% for balloon angioplasty. Intravascular imaging was used during the procedure in 72.3% of DCB cases and in 76.7% of balloon cases.
Besides demonstrating a nearly 38% reduction in the primary endpoint of target lesion failure at 1 year for the DCB over conventional balloon angioplasty, DCB nearly halved the rate of target lesion revascularization and target vessel MI and was superior on other measures of clinical outcome.
*Hazard ratio, 0.49; 95% CI, 0.31-0.79; ** HR, 0.51; 95% CI, 0.27-0.95
There was no stent rethrombosis with the DCB vs 3.9% with the conventional balloon angioplasty. Of note, there were no differences between the groups in terms of cardiac or noncardiac death.
Subgroup analyses of the primary outcome in terms of sex, age, diabetes, vessel size, or single or multiple stent layers all trended in favor of AGENT DCB but were not statistically significant for interaction.
The study is being expanded to include 600 patients. This device is a US Food and Drug Administration–designated breakthrough device, “and this pivotal trial will be the primary evidence used to support FDA approval,” Dr. Yeh said. “And given the marked superiority over conventional balloon angioplasty, I believe that the AGENT DCB is likely to become an important new treatment option for patients with coronary stenosis in the United States.”
Long overdue
Róisín Colleran, MBBCh, of the Cardiovascular Research Institute Dublin at Mater Private Hospital in Ireland, the designated discussant, first congratulated Dr. Yeh and his coinvestigators on the study’s conduct and findings.
“This study is long overdue,” she said. As Dr. Yeh noted, about 10% of PCI procedures are done for in-stent restenosis, Dr. Colleran said, but in 2023, there is still no coronary drug eluting balloon approved for this indication in the US, despite the class 1 recommendation in the 2014 European guidelines.
She pointed to the trial results, saying they are “clear...a significant reduction in target lesion failure driven by halving in rates of both target lesion revascularization and target vessel MI.”
Strengths of the study are it is the largest of its kind to date, with 480 patients, conducted at 40 US centers, using device-specific endpoints. There was a “very high” intravascular imaging rate of 75% in a cohort with a high risk for in-stent restenosis, consisting of 50% of patients with diabetes and more than 40% with multiple stents.
“The main limitation is the choice of comparator,” Dr. Colleran said. Balloon angioplasty is inferior to both stenting and drug coated balloon therapy for treatment of in-stent restenosis but is the standard of care in the United States, she noted. “I think...for regulatory reasons this was the comparator chosen,” she said.
“I think the implications are clear,” Dr. Colleran added. “This trial should provide a basis for regulatory approval of the drug coated balloon treatment of in-stent restenosis in the U.S. and finally provide this as an available treatment option for such patients.”
Dr. Yeh reported receiving grant/research support from Abbott Vascular, BD Bard, Boston Scientific, Cook Medical, Philips Medical, and Medtronic, and consulting for Abbott Vascular, Boston Scientific, CathWorks, Elixir Medical, Infraredx, Medtronic, Shockwave Medical, and Zol. Dr. Colleran had no disclosures. The trial was supported by Boston Scientific.
A version of this article first appeared on Medscape.com.
AT TCT 2023
AF tied to 45% increase in mild cognitive impairment
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- From over 4.3 million people in the UK primary electronic health record (EHR) database, researchers identified 233,833 (5.4%) with AF (mean age, 74.2 years) and randomly selected one age- and sex-matched control person without AF for each AF case patient.
- The primary outcome was incidence of mild cognitive impairment (MCI).
- The authors adjusted for age, sex, year at study entry, socioeconomic status, smoking, and a number of comorbid conditions.
- During a median of 5.3 years of follow-up, there were 4,269 incident MCI cases among both AF and non-AF patients.
TAKEAWAY:
- Individuals with AF had a higher risk of MCI than that of those without AF (adjusted hazard ratio [aHR], 1.45; 95% confidence interval [CI], 1.35-1.56).
- Besides AF, older age (risk ratio [RR], 1.08) and history of depression (RR, 1.44) were associated with greater risk of MCI, as were female sex, greater socioeconomic deprivation, stroke, and multimorbidity, including, for example, diabetes, hypercholesterolemia, and peripheral artery disease (all P < .001).
- Individuals with AF who received oral anticoagulants or amiodarone were not at increased risk of MCI, as was the case for those treated with digoxin.
- Individuals with AF and MCI were at greater risk of dementia (aHR, 1.25; 95% CI, 1.09-1.42). Sex, smoking, chronic kidney disease, and multi-comorbidity were among factors linked to elevated dementia risk.
IN PRACTICE:
The findings emphasize the association of multi-comorbidity and cardiovascular risk factors with development of MCI and progression to dementia in AF patients, the authors wrote. They noted that the data suggest combining anticoagulation and symptom and comorbidity management may prevent cognitive deterioration.
SOURCE:
The study was conducted by Sheng-Chia Chung, PhD, Institute of Health informatics Research, University College London, and colleagues. It was published online Oct. 25, 2023, as a research letter in the Journal of the American College of Cardiology (JACC): Advances.
LIMITATIONS:
The EHR dataset may have lacked granularity and detail, and some risk factors or comorbidities may not have been measured. While those with AF receiving digoxin or amiodarone treatment had no higher risk of MCI than their non-AF peers, the study’s observational design and very wide confidence intervals for these subgroups prevent making solid inferences about causality or a potential protective role of these drugs.
DISCLOSURES:
Dr. Chung is supported by the National Institute of Health and Care Research (NIHR) Author Rui Providencia, MD, PhD, of the Institute of Health informatics Research, University College London, is supported by the University College London British Heart Foundation and NIHR. All other authors report no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- From over 4.3 million people in the UK primary electronic health record (EHR) database, researchers identified 233,833 (5.4%) with AF (mean age, 74.2 years) and randomly selected one age- and sex-matched control person without AF for each AF case patient.
- The primary outcome was incidence of mild cognitive impairment (MCI).
- The authors adjusted for age, sex, year at study entry, socioeconomic status, smoking, and a number of comorbid conditions.
- During a median of 5.3 years of follow-up, there were 4,269 incident MCI cases among both AF and non-AF patients.
TAKEAWAY:
- Individuals with AF had a higher risk of MCI than that of those without AF (adjusted hazard ratio [aHR], 1.45; 95% confidence interval [CI], 1.35-1.56).
- Besides AF, older age (risk ratio [RR], 1.08) and history of depression (RR, 1.44) were associated with greater risk of MCI, as were female sex, greater socioeconomic deprivation, stroke, and multimorbidity, including, for example, diabetes, hypercholesterolemia, and peripheral artery disease (all P < .001).
- Individuals with AF who received oral anticoagulants or amiodarone were not at increased risk of MCI, as was the case for those treated with digoxin.
- Individuals with AF and MCI were at greater risk of dementia (aHR, 1.25; 95% CI, 1.09-1.42). Sex, smoking, chronic kidney disease, and multi-comorbidity were among factors linked to elevated dementia risk.
IN PRACTICE:
The findings emphasize the association of multi-comorbidity and cardiovascular risk factors with development of MCI and progression to dementia in AF patients, the authors wrote. They noted that the data suggest combining anticoagulation and symptom and comorbidity management may prevent cognitive deterioration.
SOURCE:
The study was conducted by Sheng-Chia Chung, PhD, Institute of Health informatics Research, University College London, and colleagues. It was published online Oct. 25, 2023, as a research letter in the Journal of the American College of Cardiology (JACC): Advances.
LIMITATIONS:
The EHR dataset may have lacked granularity and detail, and some risk factors or comorbidities may not have been measured. While those with AF receiving digoxin or amiodarone treatment had no higher risk of MCI than their non-AF peers, the study’s observational design and very wide confidence intervals for these subgroups prevent making solid inferences about causality or a potential protective role of these drugs.
DISCLOSURES:
Dr. Chung is supported by the National Institute of Health and Care Research (NIHR) Author Rui Providencia, MD, PhD, of the Institute of Health informatics Research, University College London, is supported by the University College London British Heart Foundation and NIHR. All other authors report no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- From over 4.3 million people in the UK primary electronic health record (EHR) database, researchers identified 233,833 (5.4%) with AF (mean age, 74.2 years) and randomly selected one age- and sex-matched control person without AF for each AF case patient.
- The primary outcome was incidence of mild cognitive impairment (MCI).
- The authors adjusted for age, sex, year at study entry, socioeconomic status, smoking, and a number of comorbid conditions.
- During a median of 5.3 years of follow-up, there were 4,269 incident MCI cases among both AF and non-AF patients.
TAKEAWAY:
- Individuals with AF had a higher risk of MCI than that of those without AF (adjusted hazard ratio [aHR], 1.45; 95% confidence interval [CI], 1.35-1.56).
- Besides AF, older age (risk ratio [RR], 1.08) and history of depression (RR, 1.44) were associated with greater risk of MCI, as were female sex, greater socioeconomic deprivation, stroke, and multimorbidity, including, for example, diabetes, hypercholesterolemia, and peripheral artery disease (all P < .001).
- Individuals with AF who received oral anticoagulants or amiodarone were not at increased risk of MCI, as was the case for those treated with digoxin.
- Individuals with AF and MCI were at greater risk of dementia (aHR, 1.25; 95% CI, 1.09-1.42). Sex, smoking, chronic kidney disease, and multi-comorbidity were among factors linked to elevated dementia risk.
IN PRACTICE:
The findings emphasize the association of multi-comorbidity and cardiovascular risk factors with development of MCI and progression to dementia in AF patients, the authors wrote. They noted that the data suggest combining anticoagulation and symptom and comorbidity management may prevent cognitive deterioration.
SOURCE:
The study was conducted by Sheng-Chia Chung, PhD, Institute of Health informatics Research, University College London, and colleagues. It was published online Oct. 25, 2023, as a research letter in the Journal of the American College of Cardiology (JACC): Advances.
LIMITATIONS:
The EHR dataset may have lacked granularity and detail, and some risk factors or comorbidities may not have been measured. While those with AF receiving digoxin or amiodarone treatment had no higher risk of MCI than their non-AF peers, the study’s observational design and very wide confidence intervals for these subgroups prevent making solid inferences about causality or a potential protective role of these drugs.
DISCLOSURES:
Dr. Chung is supported by the National Institute of Health and Care Research (NIHR) Author Rui Providencia, MD, PhD, of the Institute of Health informatics Research, University College London, is supported by the University College London British Heart Foundation and NIHR. All other authors report no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Second pig heart recipient dies
the University of Maryland Medical Center (UMMC), Baltimore, reported in a statement.
Mr. Faucette, a former lab tech who was turned down repeatedly for a standard allograft transplantation because of his various medical conditions, received the pig heart transplant on Sept. 20, 2023.
He first came to UMMC as a patient on Sept. 14. When he was admitted, he was in end-stage heart failure. Shortly before the surgery, his heart stopped, and he required resuscitation.
On Sept. 15, the Food and Drug Administration granted an emergency authorization for the surgery through its single-patient investigational new drug compassionate use pathway.
“My only real hope left is to go with the pig heart, the xenotransplant,” Mr. Faucette said in an interview from his hospital room a few days before his surgery. “At least now I have hope, and I have a chance.” He made “significant progress” in the month after the surgery, participating in physical therapy and spending time with family, according to the university. But in the days before his death, the heart showed signs of rejection.
“Mr. Faucette’s last wish was for us to make the most of what we have learned from our experience, so others may be guaranteed a chance for a new heart when a human organ is unavailable,” said Bartley P. Griffith, MD, who transplanted the pig heart into Mr. Faucette at UMMC. “He then told the team of doctors and nurses who gathered around him that he loved us. We will miss him tremendously.”
Muhammad M. Mohiuddin, MD, professor of surgery and scientific/program director of the Cardiac Xenotransplantation Program at the University of Maryland School of Medicine, said that “Mr. Faucette was a scientist who not only read and interpreted his own biopsies, but who understood the important contribution he was making in advancing the field.
“As with the first patient, David Bennett Sr., we intend to conduct an extensive analysis to identify factors that can be prevented in future transplants; this will allow us to continue to move forward and educate our colleagues in the field on our experience,” Dr. Mohiuddin added.
The researchers don’t plan to make further comments until their investigation is complete, a university spokesperson said in an interview.
UMMC performed the first transplant of a genetically modified pig heart in January 2022. Mr. Bennett, the recipient of that heart, survived for 60 days. The researchers published their initial findings in The New England Journal of Medicine, and then the results of their follow-up investigation in The Lancet.
A version of this article first appeared on Medscape.com.
the University of Maryland Medical Center (UMMC), Baltimore, reported in a statement.
Mr. Faucette, a former lab tech who was turned down repeatedly for a standard allograft transplantation because of his various medical conditions, received the pig heart transplant on Sept. 20, 2023.
He first came to UMMC as a patient on Sept. 14. When he was admitted, he was in end-stage heart failure. Shortly before the surgery, his heart stopped, and he required resuscitation.
On Sept. 15, the Food and Drug Administration granted an emergency authorization for the surgery through its single-patient investigational new drug compassionate use pathway.
“My only real hope left is to go with the pig heart, the xenotransplant,” Mr. Faucette said in an interview from his hospital room a few days before his surgery. “At least now I have hope, and I have a chance.” He made “significant progress” in the month after the surgery, participating in physical therapy and spending time with family, according to the university. But in the days before his death, the heart showed signs of rejection.
“Mr. Faucette’s last wish was for us to make the most of what we have learned from our experience, so others may be guaranteed a chance for a new heart when a human organ is unavailable,” said Bartley P. Griffith, MD, who transplanted the pig heart into Mr. Faucette at UMMC. “He then told the team of doctors and nurses who gathered around him that he loved us. We will miss him tremendously.”
Muhammad M. Mohiuddin, MD, professor of surgery and scientific/program director of the Cardiac Xenotransplantation Program at the University of Maryland School of Medicine, said that “Mr. Faucette was a scientist who not only read and interpreted his own biopsies, but who understood the important contribution he was making in advancing the field.
“As with the first patient, David Bennett Sr., we intend to conduct an extensive analysis to identify factors that can be prevented in future transplants; this will allow us to continue to move forward and educate our colleagues in the field on our experience,” Dr. Mohiuddin added.
The researchers don’t plan to make further comments until their investigation is complete, a university spokesperson said in an interview.
UMMC performed the first transplant of a genetically modified pig heart in January 2022. Mr. Bennett, the recipient of that heart, survived for 60 days. The researchers published their initial findings in The New England Journal of Medicine, and then the results of their follow-up investigation in The Lancet.
A version of this article first appeared on Medscape.com.
the University of Maryland Medical Center (UMMC), Baltimore, reported in a statement.
Mr. Faucette, a former lab tech who was turned down repeatedly for a standard allograft transplantation because of his various medical conditions, received the pig heart transplant on Sept. 20, 2023.
He first came to UMMC as a patient on Sept. 14. When he was admitted, he was in end-stage heart failure. Shortly before the surgery, his heart stopped, and he required resuscitation.
On Sept. 15, the Food and Drug Administration granted an emergency authorization for the surgery through its single-patient investigational new drug compassionate use pathway.
“My only real hope left is to go with the pig heart, the xenotransplant,” Mr. Faucette said in an interview from his hospital room a few days before his surgery. “At least now I have hope, and I have a chance.” He made “significant progress” in the month after the surgery, participating in physical therapy and spending time with family, according to the university. But in the days before his death, the heart showed signs of rejection.
“Mr. Faucette’s last wish was for us to make the most of what we have learned from our experience, so others may be guaranteed a chance for a new heart when a human organ is unavailable,” said Bartley P. Griffith, MD, who transplanted the pig heart into Mr. Faucette at UMMC. “He then told the team of doctors and nurses who gathered around him that he loved us. We will miss him tremendously.”
Muhammad M. Mohiuddin, MD, professor of surgery and scientific/program director of the Cardiac Xenotransplantation Program at the University of Maryland School of Medicine, said that “Mr. Faucette was a scientist who not only read and interpreted his own biopsies, but who understood the important contribution he was making in advancing the field.
“As with the first patient, David Bennett Sr., we intend to conduct an extensive analysis to identify factors that can be prevented in future transplants; this will allow us to continue to move forward and educate our colleagues in the field on our experience,” Dr. Mohiuddin added.
The researchers don’t plan to make further comments until their investigation is complete, a university spokesperson said in an interview.
UMMC performed the first transplant of a genetically modified pig heart in January 2022. Mr. Bennett, the recipient of that heart, survived for 60 days. The researchers published their initial findings in The New England Journal of Medicine, and then the results of their follow-up investigation in The Lancet.
A version of this article first appeared on Medscape.com.
Older adults at risk from inappropriate prescribing
Roughly 2% of prescriptions to older patients appear to be inappropriate – but the figure does not appear to differ between physicians and nurse practitioners, according to a study published in Annals of Internal Medicine.
Older adults are “especially vulnerable to adverse drug events from inappropriate prescribing due to comorbidities and aging-related physiological changes,” said Johnny Huynh, MA, doctoral candidate in economics at UCLA and lead author of the study. “Considering the volume of prescriptions for older adults, even a small percentage can translate to a big impact on adverse drug events and spending.”
In recent years, more states have granted prescriptive authority to NPs, while professional medical organizations have opposed the reforms and made claims about differences in quality of care.
The medical community must focus on the prescribing performance of individual clinicians rather than whether an NP has prescriptive authority, said David Studdert, LLB, ScD, MPH, professor of health policy at Stanford (Calif.) University and a co-author of the study.
“Don’t fixate on whether nurse practitioners have prescriptive authority or don’t,” said Mr. Studdert. “Just try to identify those practitioners who need to boost their performance.”
The investigators found that rates of potentially inappropriate prescribing were “virtually identical.” Adjusted rates were 1.66 per 100 prescriptions for NPs versus 1.68 per 100 prescriptions for physicians (adjusted odds ratio, 0.99; 95% confidence interval, 0.97-1.01).
“Older adults often have more than one chronic condition and are prescribed multiple medications to manage these conditions, putting them at risk for adverse events,” said Paula Rochon, MD, MPH, founding director of the Women’s Age Lab and professor in the Division of Geriatric Medicine at Dalla Lana School of Public Health in Toronto. “Furthermore, older women are more likely than men to have multiple medical problems and experience adverse drug events.”
Dr. Rochon led a 2021 research review on polypharmacy and inappropriate prescribing among older adults in both the United States and abroad. She and her team noted that while women are physiologically more susceptible to drug-related harm, rates of inappropriate prescribing also tend to be higher for women, such as in the case of senior U.S. veterans and older adults in Canada.
The researchers analyzed data over a 7-year period starting in 2013 from 23,669 primary care NPs and 50,060 physicians who wrote prescriptions for at least 100 patients with Medicare Part D coverage. Data from 29 states, which had all expanded prescriptive authority to NPs, was included.
Prescriptive quality was defined by the American Geriatrics Society’s Beers Criteria, a list of potentially inappropriate medications (PIMs) for adults ages 65 and over. Mr. Studdert said it’s important to note the nuance in the Beers Criteria.
“It’s not to say that there may not be certain clinical circumstances where it’s appropriate to” prescribe these drugs, Mr. Studdert said, “But generally, it’s not appropriate.”
Ten medications accounted for 99.5% of the PIMs prescribed, including drugs that were antidepressants, muscle relaxants, hypnotics, antihistamines (generation 1), antispasmodics, sulfonylureas, barbiturates, antineoplastics, thyroid medications, and nonsteroidal anti-inflammatory drugs.
The top three most frequently potentially inappropriately prescribed were antidepressants (0.393 NPs vs. 0.481 PCPs per 100 prescriptions), muscle relaxants (0.372 NPs vs. 0.305 PCPs per 100), and hypnotics (0.364 NPs vs. 0.440 PCPs per 100). Both antidepressants and hypnotics are associated with an increased risk for falls and fractures among older adults, while muscle relaxants have been shown to increase the risk for hospitalization in this population.
Despite the overall similar PIM rates, NPs were more present in the “tails,” or highest and lowest end of the quality bell curve. The higher variation among NPs means these patients are at a higher risk of receiving a prescription for an inappropriate medication, said David Chan, MD, PhD, associate professor of health policy at Stanford (Calif.) School of Medicine, and a co-author of the study.
Other studies have shown “high-intensity prescribers” were more likely to dispense drugs like benzodiazepines and opioids, which can be harmful to older patients.
According to Dr. Rochon, clinicians should use the Beers Criteria and STOPP/START Criteria to guide decision-making, along with the DRUGS framework, which follows a geriatric medicine approach that advises clinicians to discuss goals of care with their patients and conduct routine reviews of medications.
Prescribers should also avoid prescribing cascades, which “occur when a drug is prescribed, an adverse event occurs that is misinterpreted as a new medical condition, and a further drug is prescribed to treat that medical condition,” Dr. Rochon said.
To reduce cascades, “it’s important to document when a medication was started, why it was started, and who started it so that this information is available when evaluating if a medication continues to be needed,” she said.
The study was funded by grants from Robert Wood Johnson Foundation and National Science Foundation. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Roughly 2% of prescriptions to older patients appear to be inappropriate – but the figure does not appear to differ between physicians and nurse practitioners, according to a study published in Annals of Internal Medicine.
Older adults are “especially vulnerable to adverse drug events from inappropriate prescribing due to comorbidities and aging-related physiological changes,” said Johnny Huynh, MA, doctoral candidate in economics at UCLA and lead author of the study. “Considering the volume of prescriptions for older adults, even a small percentage can translate to a big impact on adverse drug events and spending.”
In recent years, more states have granted prescriptive authority to NPs, while professional medical organizations have opposed the reforms and made claims about differences in quality of care.
The medical community must focus on the prescribing performance of individual clinicians rather than whether an NP has prescriptive authority, said David Studdert, LLB, ScD, MPH, professor of health policy at Stanford (Calif.) University and a co-author of the study.
“Don’t fixate on whether nurse practitioners have prescriptive authority or don’t,” said Mr. Studdert. “Just try to identify those practitioners who need to boost their performance.”
The investigators found that rates of potentially inappropriate prescribing were “virtually identical.” Adjusted rates were 1.66 per 100 prescriptions for NPs versus 1.68 per 100 prescriptions for physicians (adjusted odds ratio, 0.99; 95% confidence interval, 0.97-1.01).
“Older adults often have more than one chronic condition and are prescribed multiple medications to manage these conditions, putting them at risk for adverse events,” said Paula Rochon, MD, MPH, founding director of the Women’s Age Lab and professor in the Division of Geriatric Medicine at Dalla Lana School of Public Health in Toronto. “Furthermore, older women are more likely than men to have multiple medical problems and experience adverse drug events.”
Dr. Rochon led a 2021 research review on polypharmacy and inappropriate prescribing among older adults in both the United States and abroad. She and her team noted that while women are physiologically more susceptible to drug-related harm, rates of inappropriate prescribing also tend to be higher for women, such as in the case of senior U.S. veterans and older adults in Canada.
The researchers analyzed data over a 7-year period starting in 2013 from 23,669 primary care NPs and 50,060 physicians who wrote prescriptions for at least 100 patients with Medicare Part D coverage. Data from 29 states, which had all expanded prescriptive authority to NPs, was included.
Prescriptive quality was defined by the American Geriatrics Society’s Beers Criteria, a list of potentially inappropriate medications (PIMs) for adults ages 65 and over. Mr. Studdert said it’s important to note the nuance in the Beers Criteria.
“It’s not to say that there may not be certain clinical circumstances where it’s appropriate to” prescribe these drugs, Mr. Studdert said, “But generally, it’s not appropriate.”
Ten medications accounted for 99.5% of the PIMs prescribed, including drugs that were antidepressants, muscle relaxants, hypnotics, antihistamines (generation 1), antispasmodics, sulfonylureas, barbiturates, antineoplastics, thyroid medications, and nonsteroidal anti-inflammatory drugs.
The top three most frequently potentially inappropriately prescribed were antidepressants (0.393 NPs vs. 0.481 PCPs per 100 prescriptions), muscle relaxants (0.372 NPs vs. 0.305 PCPs per 100), and hypnotics (0.364 NPs vs. 0.440 PCPs per 100). Both antidepressants and hypnotics are associated with an increased risk for falls and fractures among older adults, while muscle relaxants have been shown to increase the risk for hospitalization in this population.
Despite the overall similar PIM rates, NPs were more present in the “tails,” or highest and lowest end of the quality bell curve. The higher variation among NPs means these patients are at a higher risk of receiving a prescription for an inappropriate medication, said David Chan, MD, PhD, associate professor of health policy at Stanford (Calif.) School of Medicine, and a co-author of the study.
Other studies have shown “high-intensity prescribers” were more likely to dispense drugs like benzodiazepines and opioids, which can be harmful to older patients.
According to Dr. Rochon, clinicians should use the Beers Criteria and STOPP/START Criteria to guide decision-making, along with the DRUGS framework, which follows a geriatric medicine approach that advises clinicians to discuss goals of care with their patients and conduct routine reviews of medications.
Prescribers should also avoid prescribing cascades, which “occur when a drug is prescribed, an adverse event occurs that is misinterpreted as a new medical condition, and a further drug is prescribed to treat that medical condition,” Dr. Rochon said.
To reduce cascades, “it’s important to document when a medication was started, why it was started, and who started it so that this information is available when evaluating if a medication continues to be needed,” she said.
The study was funded by grants from Robert Wood Johnson Foundation and National Science Foundation. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Roughly 2% of prescriptions to older patients appear to be inappropriate – but the figure does not appear to differ between physicians and nurse practitioners, according to a study published in Annals of Internal Medicine.
Older adults are “especially vulnerable to adverse drug events from inappropriate prescribing due to comorbidities and aging-related physiological changes,” said Johnny Huynh, MA, doctoral candidate in economics at UCLA and lead author of the study. “Considering the volume of prescriptions for older adults, even a small percentage can translate to a big impact on adverse drug events and spending.”
In recent years, more states have granted prescriptive authority to NPs, while professional medical organizations have opposed the reforms and made claims about differences in quality of care.
The medical community must focus on the prescribing performance of individual clinicians rather than whether an NP has prescriptive authority, said David Studdert, LLB, ScD, MPH, professor of health policy at Stanford (Calif.) University and a co-author of the study.
“Don’t fixate on whether nurse practitioners have prescriptive authority or don’t,” said Mr. Studdert. “Just try to identify those practitioners who need to boost their performance.”
The investigators found that rates of potentially inappropriate prescribing were “virtually identical.” Adjusted rates were 1.66 per 100 prescriptions for NPs versus 1.68 per 100 prescriptions for physicians (adjusted odds ratio, 0.99; 95% confidence interval, 0.97-1.01).
“Older adults often have more than one chronic condition and are prescribed multiple medications to manage these conditions, putting them at risk for adverse events,” said Paula Rochon, MD, MPH, founding director of the Women’s Age Lab and professor in the Division of Geriatric Medicine at Dalla Lana School of Public Health in Toronto. “Furthermore, older women are more likely than men to have multiple medical problems and experience adverse drug events.”
Dr. Rochon led a 2021 research review on polypharmacy and inappropriate prescribing among older adults in both the United States and abroad. She and her team noted that while women are physiologically more susceptible to drug-related harm, rates of inappropriate prescribing also tend to be higher for women, such as in the case of senior U.S. veterans and older adults in Canada.
The researchers analyzed data over a 7-year period starting in 2013 from 23,669 primary care NPs and 50,060 physicians who wrote prescriptions for at least 100 patients with Medicare Part D coverage. Data from 29 states, which had all expanded prescriptive authority to NPs, was included.
Prescriptive quality was defined by the American Geriatrics Society’s Beers Criteria, a list of potentially inappropriate medications (PIMs) for adults ages 65 and over. Mr. Studdert said it’s important to note the nuance in the Beers Criteria.
“It’s not to say that there may not be certain clinical circumstances where it’s appropriate to” prescribe these drugs, Mr. Studdert said, “But generally, it’s not appropriate.”
Ten medications accounted for 99.5% of the PIMs prescribed, including drugs that were antidepressants, muscle relaxants, hypnotics, antihistamines (generation 1), antispasmodics, sulfonylureas, barbiturates, antineoplastics, thyroid medications, and nonsteroidal anti-inflammatory drugs.
The top three most frequently potentially inappropriately prescribed were antidepressants (0.393 NPs vs. 0.481 PCPs per 100 prescriptions), muscle relaxants (0.372 NPs vs. 0.305 PCPs per 100), and hypnotics (0.364 NPs vs. 0.440 PCPs per 100). Both antidepressants and hypnotics are associated with an increased risk for falls and fractures among older adults, while muscle relaxants have been shown to increase the risk for hospitalization in this population.
Despite the overall similar PIM rates, NPs were more present in the “tails,” or highest and lowest end of the quality bell curve. The higher variation among NPs means these patients are at a higher risk of receiving a prescription for an inappropriate medication, said David Chan, MD, PhD, associate professor of health policy at Stanford (Calif.) School of Medicine, and a co-author of the study.
Other studies have shown “high-intensity prescribers” were more likely to dispense drugs like benzodiazepines and opioids, which can be harmful to older patients.
According to Dr. Rochon, clinicians should use the Beers Criteria and STOPP/START Criteria to guide decision-making, along with the DRUGS framework, which follows a geriatric medicine approach that advises clinicians to discuss goals of care with their patients and conduct routine reviews of medications.
Prescribers should also avoid prescribing cascades, which “occur when a drug is prescribed, an adverse event occurs that is misinterpreted as a new medical condition, and a further drug is prescribed to treat that medical condition,” Dr. Rochon said.
To reduce cascades, “it’s important to document when a medication was started, why it was started, and who started it so that this information is available when evaluating if a medication continues to be needed,” she said.
The study was funded by grants from Robert Wood Johnson Foundation and National Science Foundation. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Survey finds oral minoxidil shortage in Washington-area pharmacies
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
A .
Patients are not finding out until they go to pick up their prescription, which can result in an interruption of treatment – and, potentially a loss of hard-earned hair gain, said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was the lead author of the survey, published online on Oct. 26 as a research letter in the Journal of Drugs in Dermatology.
Going off low-dose oral minoxidil may spark a telogen effluvium event, and that is very disappointing to patients, Dr. Friedman told this news organization.
“There needs to be some system that alerts us,” he said. “Even if it’s a minor shortage, just so we’re aware. We can then prepare patients,” he added, noting that it would be better for someone to be taking a lower-than-normal dose rather than no medication at all while they wait for a refill.
Minoxidil has long been approved in a topical formulation to treat androgenetic alopecia, but a low-dose oral form has gained currency in the wake of findings that it might more effectively treat hair loss, and is without side effects. A New York Times article in August 2022 touting low-dose oral minoxidil as a cheap and effective hair loss drug appeared to ignite interest in this option. In May, 2023, researchers reporting in JAMA Network Open demonstrated a significant uptick in prescriptions for oral minoxidil in the wake of the article’s publication.
Oral minoxidil is approved by the Food and Drug Administration only for hypertension, but dermatologists are prescribing it off-label at a lower dose for hair loss. Dr. Friedman said it’s not clear whether the shortages his team found are national in scope, or whether they are a result of increased demand, or other factors.
After several patients told him they were having trouble filling minoxidil prescriptions, and colleagues said they’d had patients with similar experiences, Dr. Friedman and his colleagues undertook the survey. In the first week of October 2023, they contacted 277 pharmacies by phone in Washington and surrounding Virginia and Maryland counties. The pharmacies were CVS, Giant, Walgreens, and Harris Teeter.
Of the 277 pharmacies they contacted, 40% (111) reported availability of 2.5-mg tablets for a 30-day supply, and just under 30% (82) reported having 10-mg tablets for a 30-day supply.
For treating hair loss, most patients are prescribed 2.5-mg pills, with starting doses ranging from 0.625 mg to 5 mg twice a day, Dr. Friedman said. The 10-mg dose is more frequently prescribed for hypertension.
Only 28% (19 of 67) of the Maryland pharmacies had 30-day supplies of 2.5-mg tablets on hand, and just 22% (15) of the Maryland pharmacies had 30-day supplies of 10-mg tablets. In Northern Virginia, 44% (63 of 143) of the pharmacies had 30-day supplies of the 2.5 mg tablets, as did just 43% (29 of 67) of the Washington pharmacies.
Dr. Friedman said he has started giving patients paper prescriptions they can use to shop around, rather than electronically sending a prescription to a particular pharmacy.
Neither the Food and Drug Administration nor the American Society of Health System Pharmacists lists oral minoxidil as a drug in shortage.
Michael Ganio, PharmD, senior director of pharmacy practice and quality for ASHP, said the organization received a report from wholesalers in mid-September showing spotty oral minoxidil availability, with the drug on backorder with some manufacturers. ASHP's shortages list is compiled from reports from physicians, manufacturers and wholesalers, he said.
Under what he calls "blue sky conditions," pharmacies using a just-in-time inventory model should be able to fill prescriptions within hours or days, which might explain why some pharmacies in the Washington, DC area survey did not have a 30-day supply on hand, he said. However, Dr. Ganio noted that the causes of drug shortages are complex and multi-factorial. For now, he said there have been no oral minoxidil shortage reports since mid-September.
But Dr. Friedman said some of his patients have waited weeks for a new supply – and that no one is aware of the problem until the last moment.
The lack of alerts or transparency “also erodes the physician-patient relationship because there’s this expectation of the patient that we should have known this,” said Dr. Friedman.
Dr. Friedman reports no relevant financial relationships.
This story was updated on 11/2/2023.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Semaglutide prescribing surged in the past year
Among more than 350,000 prescribers in the nationwide DrFirst network between December 2022 and June 2023, prescriptions for the weight loss formulation Wegovy rose sixfold while those for Ozempic, the lower-dose version for treating type 2 diabetes, increased by 65%.
Before December 2022, prescribing for both semaglutide drug formulations had been relatively flat. Ozempic was approved in the United States for treating type 2 diabetes in 2017, and Wegovy for weight loss in 2021. Prescribing of oral type 2 diabetes drugs also rose during the study period but to a lesser degree.
General and family practice providers were the most frequent semaglutide providers, accounting for 30% of the total, followed by internists at 15%, endocrinologists at 4%, ob.gyns. at 2%, and pediatricians at 1%. Other specialists writing less than 1% of the prescriptions included cardiologists, emergency medicine physicians, hospitalists, psychiatrists, and surgeons.
“What I think is interesting is that in a relatively short period of time, primary care providers got comfortable with writing [prescriptions] for a drug that’s relatively new ... That isn’t always the case ... To me, it’s actually pretty telling that within a year or year and a half, the primary care field got very comfortable writing [prescriptions] for these [glucagon-like peptide 1 receptor agonists],” DrFirst chief medical officer Colin Banas, MD, said in an interview.
Asked to comment, S. Sethu K. Reddy, MD, president of the American Association of Clinical Endocrinologists, noted, “It is to be expected when there is an agent that not only lowers blood sugar levels but also may result in weight loss. These medications are packaged conveniently for a primary care physician to prescribe. There is enough awareness amongst the public in that the patients themselves often ask their physician about the medication.”
Moreover, Dr. Reddy noted, “there is clinical evidence that these medications not only improve diabetes control but also reduce the risk of cardiovascular events. The lack of cardiovascular safety data was a missing piece of the puzzle in the past. So, currently, if someone has type 2 diabetes and is at greater risk of cardiovascular disease, there is little controversy for the patient to receive GLP-1 analogs.”
Are patients actually getting the prescribed medications?
However, Sharon W. Lahiri, MD, of Wayne State University School of Medicine and Henry Ford Hospital, Detroit, pointed out that prescription data don’t equate to actual drug use. “It depends what type of insurance a person has. ... We write prescriptions on a daily basis for semaglutide. At least five or more come into our inbox every day saying it’s denied.”
Earlier this year, Dr. Lahiri co-authored results from a survey of 125 health care providers between February 9 and March 14, 2022, seeking to identify factors influencing medication choices and barriers to prescribing both GLP-1 agonists and sodium-glucose cotransporter 2 inhibitors. High cost and the need for prior authorizations were reported as the main barriers to prescribing drugs in these two classes, along with a lack of experience among some specialists.
Dr. Lahiri told this news organization that many insurers don’t cover Wegovy at all, or they mandate stepped-care paradigms in which the patient must enroll in behavior modification programs for a period of time or first try older, less expensive weight loss drugs such as phentermine, topiramate, or orlistat before they authorize coverage for Wegovy or even for the older weight-loss GLP-1 agonist drug Saxenda. “And then, they require you to document why the prior drugs didn’t work or couldn’t be tolerated.”
Moreover, Wegovy coverage is often time-limited, varying anywhere from 3 months to 2 years, and some insurers require a visit where the patient must have lost at least 5% of their body weight for coverage to continue.
Dr. Lahiri said recently she’s also encountered such “step” requirements when she’s tried to prescribe the “twincretin” Mounjaro for treating type 2 diabetes, where insurers will require trials of other GLP-1 agonists first. “So, it’s very complicated. I would say the barriers are definitely worse now. I don’t think the number of written prescriptions reflects that at all.”
Indeed, Dr. Banas noted, “more patients are going to pay out of pocket for Wegovy than for Ozempic if they have a diabetes indication.” And he added, “In my clinical observation, insurance coverage for obesity medication appears to be holding steady. I haven’t seen a massive increase in these drugs being covered for obesity per se, but I definitely see more coverage for diabetes use cases.”
The study was funded by DrFirst. Dr. Banas is an employee of DrFirst. Dr. Reddy and Dr. Lahiri have no disclosures.
A version of this article appeared on Medscape.com.
Among more than 350,000 prescribers in the nationwide DrFirst network between December 2022 and June 2023, prescriptions for the weight loss formulation Wegovy rose sixfold while those for Ozempic, the lower-dose version for treating type 2 diabetes, increased by 65%.
Before December 2022, prescribing for both semaglutide drug formulations had been relatively flat. Ozempic was approved in the United States for treating type 2 diabetes in 2017, and Wegovy for weight loss in 2021. Prescribing of oral type 2 diabetes drugs also rose during the study period but to a lesser degree.
General and family practice providers were the most frequent semaglutide providers, accounting for 30% of the total, followed by internists at 15%, endocrinologists at 4%, ob.gyns. at 2%, and pediatricians at 1%. Other specialists writing less than 1% of the prescriptions included cardiologists, emergency medicine physicians, hospitalists, psychiatrists, and surgeons.
“What I think is interesting is that in a relatively short period of time, primary care providers got comfortable with writing [prescriptions] for a drug that’s relatively new ... That isn’t always the case ... To me, it’s actually pretty telling that within a year or year and a half, the primary care field got very comfortable writing [prescriptions] for these [glucagon-like peptide 1 receptor agonists],” DrFirst chief medical officer Colin Banas, MD, said in an interview.
Asked to comment, S. Sethu K. Reddy, MD, president of the American Association of Clinical Endocrinologists, noted, “It is to be expected when there is an agent that not only lowers blood sugar levels but also may result in weight loss. These medications are packaged conveniently for a primary care physician to prescribe. There is enough awareness amongst the public in that the patients themselves often ask their physician about the medication.”
Moreover, Dr. Reddy noted, “there is clinical evidence that these medications not only improve diabetes control but also reduce the risk of cardiovascular events. The lack of cardiovascular safety data was a missing piece of the puzzle in the past. So, currently, if someone has type 2 diabetes and is at greater risk of cardiovascular disease, there is little controversy for the patient to receive GLP-1 analogs.”
Are patients actually getting the prescribed medications?
However, Sharon W. Lahiri, MD, of Wayne State University School of Medicine and Henry Ford Hospital, Detroit, pointed out that prescription data don’t equate to actual drug use. “It depends what type of insurance a person has. ... We write prescriptions on a daily basis for semaglutide. At least five or more come into our inbox every day saying it’s denied.”
Earlier this year, Dr. Lahiri co-authored results from a survey of 125 health care providers between February 9 and March 14, 2022, seeking to identify factors influencing medication choices and barriers to prescribing both GLP-1 agonists and sodium-glucose cotransporter 2 inhibitors. High cost and the need for prior authorizations were reported as the main barriers to prescribing drugs in these two classes, along with a lack of experience among some specialists.
Dr. Lahiri told this news organization that many insurers don’t cover Wegovy at all, or they mandate stepped-care paradigms in which the patient must enroll in behavior modification programs for a period of time or first try older, less expensive weight loss drugs such as phentermine, topiramate, or orlistat before they authorize coverage for Wegovy or even for the older weight-loss GLP-1 agonist drug Saxenda. “And then, they require you to document why the prior drugs didn’t work or couldn’t be tolerated.”
Moreover, Wegovy coverage is often time-limited, varying anywhere from 3 months to 2 years, and some insurers require a visit where the patient must have lost at least 5% of their body weight for coverage to continue.
Dr. Lahiri said recently she’s also encountered such “step” requirements when she’s tried to prescribe the “twincretin” Mounjaro for treating type 2 diabetes, where insurers will require trials of other GLP-1 agonists first. “So, it’s very complicated. I would say the barriers are definitely worse now. I don’t think the number of written prescriptions reflects that at all.”
Indeed, Dr. Banas noted, “more patients are going to pay out of pocket for Wegovy than for Ozempic if they have a diabetes indication.” And he added, “In my clinical observation, insurance coverage for obesity medication appears to be holding steady. I haven’t seen a massive increase in these drugs being covered for obesity per se, but I definitely see more coverage for diabetes use cases.”
The study was funded by DrFirst. Dr. Banas is an employee of DrFirst. Dr. Reddy and Dr. Lahiri have no disclosures.
A version of this article appeared on Medscape.com.
Among more than 350,000 prescribers in the nationwide DrFirst network between December 2022 and June 2023, prescriptions for the weight loss formulation Wegovy rose sixfold while those for Ozempic, the lower-dose version for treating type 2 diabetes, increased by 65%.
Before December 2022, prescribing for both semaglutide drug formulations had been relatively flat. Ozempic was approved in the United States for treating type 2 diabetes in 2017, and Wegovy for weight loss in 2021. Prescribing of oral type 2 diabetes drugs also rose during the study period but to a lesser degree.
General and family practice providers were the most frequent semaglutide providers, accounting for 30% of the total, followed by internists at 15%, endocrinologists at 4%, ob.gyns. at 2%, and pediatricians at 1%. Other specialists writing less than 1% of the prescriptions included cardiologists, emergency medicine physicians, hospitalists, psychiatrists, and surgeons.
“What I think is interesting is that in a relatively short period of time, primary care providers got comfortable with writing [prescriptions] for a drug that’s relatively new ... That isn’t always the case ... To me, it’s actually pretty telling that within a year or year and a half, the primary care field got very comfortable writing [prescriptions] for these [glucagon-like peptide 1 receptor agonists],” DrFirst chief medical officer Colin Banas, MD, said in an interview.
Asked to comment, S. Sethu K. Reddy, MD, president of the American Association of Clinical Endocrinologists, noted, “It is to be expected when there is an agent that not only lowers blood sugar levels but also may result in weight loss. These medications are packaged conveniently for a primary care physician to prescribe. There is enough awareness amongst the public in that the patients themselves often ask their physician about the medication.”
Moreover, Dr. Reddy noted, “there is clinical evidence that these medications not only improve diabetes control but also reduce the risk of cardiovascular events. The lack of cardiovascular safety data was a missing piece of the puzzle in the past. So, currently, if someone has type 2 diabetes and is at greater risk of cardiovascular disease, there is little controversy for the patient to receive GLP-1 analogs.”
Are patients actually getting the prescribed medications?
However, Sharon W. Lahiri, MD, of Wayne State University School of Medicine and Henry Ford Hospital, Detroit, pointed out that prescription data don’t equate to actual drug use. “It depends what type of insurance a person has. ... We write prescriptions on a daily basis for semaglutide. At least five or more come into our inbox every day saying it’s denied.”
Earlier this year, Dr. Lahiri co-authored results from a survey of 125 health care providers between February 9 and March 14, 2022, seeking to identify factors influencing medication choices and barriers to prescribing both GLP-1 agonists and sodium-glucose cotransporter 2 inhibitors. High cost and the need for prior authorizations were reported as the main barriers to prescribing drugs in these two classes, along with a lack of experience among some specialists.
Dr. Lahiri told this news organization that many insurers don’t cover Wegovy at all, or they mandate stepped-care paradigms in which the patient must enroll in behavior modification programs for a period of time or first try older, less expensive weight loss drugs such as phentermine, topiramate, or orlistat before they authorize coverage for Wegovy or even for the older weight-loss GLP-1 agonist drug Saxenda. “And then, they require you to document why the prior drugs didn’t work or couldn’t be tolerated.”
Moreover, Wegovy coverage is often time-limited, varying anywhere from 3 months to 2 years, and some insurers require a visit where the patient must have lost at least 5% of their body weight for coverage to continue.
Dr. Lahiri said recently she’s also encountered such “step” requirements when she’s tried to prescribe the “twincretin” Mounjaro for treating type 2 diabetes, where insurers will require trials of other GLP-1 agonists first. “So, it’s very complicated. I would say the barriers are definitely worse now. I don’t think the number of written prescriptions reflects that at all.”
Indeed, Dr. Banas noted, “more patients are going to pay out of pocket for Wegovy than for Ozempic if they have a diabetes indication.” And he added, “In my clinical observation, insurance coverage for obesity medication appears to be holding steady. I haven’t seen a massive increase in these drugs being covered for obesity per se, but I definitely see more coverage for diabetes use cases.”
The study was funded by DrFirst. Dr. Banas is an employee of DrFirst. Dr. Reddy and Dr. Lahiri have no disclosures.
A version of this article appeared on Medscape.com.
84-year-old MD contests employer’s mandatory cognitive tests for older docs
Lylas G. Mogk, MD, recently sued Henry Ford Health and Henry Ford Medical Group in federal court, alleging that the mandatory cognitive test violates the Americans with Disabilities Act, the Age Discrimination in Employment Act, and two Michigan laws.
Dr. Mogk’s lawsuit follows a widely watched 2020 case in which the U.S. Equal Employment Opportunity Commission sued Yale New Haven Hospital, the teaching hospital of Yale University, for age discrimination. According to the lawsuit, the hospital illegally required neuropsychological and eye examinations of physicians aged 70 or older who sought to gain or renew staff privileges.
According to the lawsuit, Dr. Mogk is a member of Henry Ford Medical Group, which in 2017 required all members aged 70 and older to undergo cognitive screening tests. The tests would be repeated every 5 years thereafter, the lawsuit said, and anyone who refused would have to resign or be fired.
Dr. Mogk completed the screening, although no information about the results or outcome was mentioned in the lawsuit. It’s not clear whether Henry Ford’s cognitive test mandate remains in place; a spokesperson for Henry Ford Health and attorneys for Dr. Mogk declined to comment.
The number of practicing physicians in their 70s and beyond is rising. A 2021 report found that 12% of U.S. licensed physicians in 2020 were least 70 years old, up from 9% in 2010 and an increase from 75,627 to 120,510. The percentage of doctors aged 60-69 grew to 19% from 16% in 2010.
The number of health systems requiring testing of older physicians isn’t known, although various reports suggest at least a dozen have had mandates.
The University of California, San Diego, offers a physical and mental screening program that health organizations can use to evaluate “late-career physicians,” and a 2021 report noted that “Nebraska’s Children’s Hospital requires physicians aged 70 years and older to undergo an assessment by several peers, a complete physical, and unspecified cognitive screening.” Another system, Hartford HealthCare, mandated an annual reappointment process for clinicians aged 70 or older, requiring them to undergo various exams.
There’s evidence that physician performance declines with age. However, age-based cognitive testing can run afoul of federal and state laws against age discrimination, said Sharona Hoffman, JD, professor of law and bioethics at Case Western Reserve University, Cleveland, in an interview.
Federal law prohibits age-related restrictions on employment but allows exceptions in areas like public safety, said Ms. Hoffman, who’s written about age discrimination and testing requirements. Pilots, law enforcement officers, firefighters, and air controllers, for example, can be forced to retire at specific ages.
It’s not clear how many physicians took the cognitive tests required by Henry Ford Medical Group.
However, details are available about the policy at Yale New Haven Hospital: According to the EEOC lawsuit, from 2016 to 2019, 145 physicians aged 70 or older took the mandatory test. Of those, seven individuals failed either or both of the exams, 14 were listed as “borderline deficient,” and one was listed as “deficient.” Another five refused testing and either resigned or changed their status. The EEOC case against the hospital is still pending.
“You can make an argument that health care is like a public safety job because people put their lives in the hands of doctors,” Ms. Hoffman said.
In defending mandatory cognitive tests, she said, health care systems could say, “it’s not really discrimination; we’re not forcing them to retire, we’re not limiting their work in any way. We’re just doing testing to make sure they perform competently, and the ADA allows us to conduct testing that is job-related.”
Indeed, a Yale New Haven Hospital spokesman made an argument along these lines in a statement regarding the 2020 lawsuit: The “policy is designed to protect our patients from potential harm while including safeguards to ensure that our physicians are treated fairly. The policy is modeled on similar standards in other industries, and we are confident that no discrimination has occurred and will vigorously defend ourselves in this matter.”
However, Ms. Hoffman herself doesn’t buy these arguments. Requiring tests only for older physicians does appear to be discrimination based on age, she said. As an alternative, “employers can do close supervision of people. As soon as there are performance problems or patient complaints, you need to see a doctor or get testing done.”
Another option is to mandate tests at specific ages via licensing boards. “I don’t think that would be legally problematic,” Ms. Hoffman said.
What else can be done to protect patients from clinicians whose skills have significantly declined as they’ve aged? The 2021 report in Neurology Clinical Practice notes that there are disadvantages to several strategies.
One common approach, waiting to evaluate a clinician until an error occurs, can lead to patient harm, the report’s authors wrote. Relying on reporting by peers is problematic because “physicians have been very resistant to reporting colleagues who are impaired” and the “medical apprenticeship model discourages physicians from reporting on senior colleagues.”
Physician self-assessment is yet another option, but “loss of insight may be a component of an individual’s impairment,” the authors wrote.
So what’s the best solution? The authors recommended “a relatively brief cognitive screening followed by more extensive testing for the most impaired individuals.” This approach “appears most reliable in confidentially identifying truly impaired physicians while minimizing the chance of a falsely flagging unimpaired individuals. This strategy allows aging physicians to continue working while safeguarding both their reputations and their patients’ health.”
Ms. Hoffman has no disclosures.
A version of this article first appeared on Medscape.com.
Lylas G. Mogk, MD, recently sued Henry Ford Health and Henry Ford Medical Group in federal court, alleging that the mandatory cognitive test violates the Americans with Disabilities Act, the Age Discrimination in Employment Act, and two Michigan laws.
Dr. Mogk’s lawsuit follows a widely watched 2020 case in which the U.S. Equal Employment Opportunity Commission sued Yale New Haven Hospital, the teaching hospital of Yale University, for age discrimination. According to the lawsuit, the hospital illegally required neuropsychological and eye examinations of physicians aged 70 or older who sought to gain or renew staff privileges.
According to the lawsuit, Dr. Mogk is a member of Henry Ford Medical Group, which in 2017 required all members aged 70 and older to undergo cognitive screening tests. The tests would be repeated every 5 years thereafter, the lawsuit said, and anyone who refused would have to resign or be fired.
Dr. Mogk completed the screening, although no information about the results or outcome was mentioned in the lawsuit. It’s not clear whether Henry Ford’s cognitive test mandate remains in place; a spokesperson for Henry Ford Health and attorneys for Dr. Mogk declined to comment.
The number of practicing physicians in their 70s and beyond is rising. A 2021 report found that 12% of U.S. licensed physicians in 2020 were least 70 years old, up from 9% in 2010 and an increase from 75,627 to 120,510. The percentage of doctors aged 60-69 grew to 19% from 16% in 2010.
The number of health systems requiring testing of older physicians isn’t known, although various reports suggest at least a dozen have had mandates.
The University of California, San Diego, offers a physical and mental screening program that health organizations can use to evaluate “late-career physicians,” and a 2021 report noted that “Nebraska’s Children’s Hospital requires physicians aged 70 years and older to undergo an assessment by several peers, a complete physical, and unspecified cognitive screening.” Another system, Hartford HealthCare, mandated an annual reappointment process for clinicians aged 70 or older, requiring them to undergo various exams.
There’s evidence that physician performance declines with age. However, age-based cognitive testing can run afoul of federal and state laws against age discrimination, said Sharona Hoffman, JD, professor of law and bioethics at Case Western Reserve University, Cleveland, in an interview.
Federal law prohibits age-related restrictions on employment but allows exceptions in areas like public safety, said Ms. Hoffman, who’s written about age discrimination and testing requirements. Pilots, law enforcement officers, firefighters, and air controllers, for example, can be forced to retire at specific ages.
It’s not clear how many physicians took the cognitive tests required by Henry Ford Medical Group.
However, details are available about the policy at Yale New Haven Hospital: According to the EEOC lawsuit, from 2016 to 2019, 145 physicians aged 70 or older took the mandatory test. Of those, seven individuals failed either or both of the exams, 14 were listed as “borderline deficient,” and one was listed as “deficient.” Another five refused testing and either resigned or changed their status. The EEOC case against the hospital is still pending.
“You can make an argument that health care is like a public safety job because people put their lives in the hands of doctors,” Ms. Hoffman said.
In defending mandatory cognitive tests, she said, health care systems could say, “it’s not really discrimination; we’re not forcing them to retire, we’re not limiting their work in any way. We’re just doing testing to make sure they perform competently, and the ADA allows us to conduct testing that is job-related.”
Indeed, a Yale New Haven Hospital spokesman made an argument along these lines in a statement regarding the 2020 lawsuit: The “policy is designed to protect our patients from potential harm while including safeguards to ensure that our physicians are treated fairly. The policy is modeled on similar standards in other industries, and we are confident that no discrimination has occurred and will vigorously defend ourselves in this matter.”
However, Ms. Hoffman herself doesn’t buy these arguments. Requiring tests only for older physicians does appear to be discrimination based on age, she said. As an alternative, “employers can do close supervision of people. As soon as there are performance problems or patient complaints, you need to see a doctor or get testing done.”
Another option is to mandate tests at specific ages via licensing boards. “I don’t think that would be legally problematic,” Ms. Hoffman said.
What else can be done to protect patients from clinicians whose skills have significantly declined as they’ve aged? The 2021 report in Neurology Clinical Practice notes that there are disadvantages to several strategies.
One common approach, waiting to evaluate a clinician until an error occurs, can lead to patient harm, the report’s authors wrote. Relying on reporting by peers is problematic because “physicians have been very resistant to reporting colleagues who are impaired” and the “medical apprenticeship model discourages physicians from reporting on senior colleagues.”
Physician self-assessment is yet another option, but “loss of insight may be a component of an individual’s impairment,” the authors wrote.
So what’s the best solution? The authors recommended “a relatively brief cognitive screening followed by more extensive testing for the most impaired individuals.” This approach “appears most reliable in confidentially identifying truly impaired physicians while minimizing the chance of a falsely flagging unimpaired individuals. This strategy allows aging physicians to continue working while safeguarding both their reputations and their patients’ health.”
Ms. Hoffman has no disclosures.
A version of this article first appeared on Medscape.com.
Lylas G. Mogk, MD, recently sued Henry Ford Health and Henry Ford Medical Group in federal court, alleging that the mandatory cognitive test violates the Americans with Disabilities Act, the Age Discrimination in Employment Act, and two Michigan laws.
Dr. Mogk’s lawsuit follows a widely watched 2020 case in which the U.S. Equal Employment Opportunity Commission sued Yale New Haven Hospital, the teaching hospital of Yale University, for age discrimination. According to the lawsuit, the hospital illegally required neuropsychological and eye examinations of physicians aged 70 or older who sought to gain or renew staff privileges.
According to the lawsuit, Dr. Mogk is a member of Henry Ford Medical Group, which in 2017 required all members aged 70 and older to undergo cognitive screening tests. The tests would be repeated every 5 years thereafter, the lawsuit said, and anyone who refused would have to resign or be fired.
Dr. Mogk completed the screening, although no information about the results or outcome was mentioned in the lawsuit. It’s not clear whether Henry Ford’s cognitive test mandate remains in place; a spokesperson for Henry Ford Health and attorneys for Dr. Mogk declined to comment.
The number of practicing physicians in their 70s and beyond is rising. A 2021 report found that 12% of U.S. licensed physicians in 2020 were least 70 years old, up from 9% in 2010 and an increase from 75,627 to 120,510. The percentage of doctors aged 60-69 grew to 19% from 16% in 2010.
The number of health systems requiring testing of older physicians isn’t known, although various reports suggest at least a dozen have had mandates.
The University of California, San Diego, offers a physical and mental screening program that health organizations can use to evaluate “late-career physicians,” and a 2021 report noted that “Nebraska’s Children’s Hospital requires physicians aged 70 years and older to undergo an assessment by several peers, a complete physical, and unspecified cognitive screening.” Another system, Hartford HealthCare, mandated an annual reappointment process for clinicians aged 70 or older, requiring them to undergo various exams.
There’s evidence that physician performance declines with age. However, age-based cognitive testing can run afoul of federal and state laws against age discrimination, said Sharona Hoffman, JD, professor of law and bioethics at Case Western Reserve University, Cleveland, in an interview.
Federal law prohibits age-related restrictions on employment but allows exceptions in areas like public safety, said Ms. Hoffman, who’s written about age discrimination and testing requirements. Pilots, law enforcement officers, firefighters, and air controllers, for example, can be forced to retire at specific ages.
It’s not clear how many physicians took the cognitive tests required by Henry Ford Medical Group.
However, details are available about the policy at Yale New Haven Hospital: According to the EEOC lawsuit, from 2016 to 2019, 145 physicians aged 70 or older took the mandatory test. Of those, seven individuals failed either or both of the exams, 14 were listed as “borderline deficient,” and one was listed as “deficient.” Another five refused testing and either resigned or changed their status. The EEOC case against the hospital is still pending.
“You can make an argument that health care is like a public safety job because people put their lives in the hands of doctors,” Ms. Hoffman said.
In defending mandatory cognitive tests, she said, health care systems could say, “it’s not really discrimination; we’re not forcing them to retire, we’re not limiting their work in any way. We’re just doing testing to make sure they perform competently, and the ADA allows us to conduct testing that is job-related.”
Indeed, a Yale New Haven Hospital spokesman made an argument along these lines in a statement regarding the 2020 lawsuit: The “policy is designed to protect our patients from potential harm while including safeguards to ensure that our physicians are treated fairly. The policy is modeled on similar standards in other industries, and we are confident that no discrimination has occurred and will vigorously defend ourselves in this matter.”
However, Ms. Hoffman herself doesn’t buy these arguments. Requiring tests only for older physicians does appear to be discrimination based on age, she said. As an alternative, “employers can do close supervision of people. As soon as there are performance problems or patient complaints, you need to see a doctor or get testing done.”
Another option is to mandate tests at specific ages via licensing boards. “I don’t think that would be legally problematic,” Ms. Hoffman said.
What else can be done to protect patients from clinicians whose skills have significantly declined as they’ve aged? The 2021 report in Neurology Clinical Practice notes that there are disadvantages to several strategies.
One common approach, waiting to evaluate a clinician until an error occurs, can lead to patient harm, the report’s authors wrote. Relying on reporting by peers is problematic because “physicians have been very resistant to reporting colleagues who are impaired” and the “medical apprenticeship model discourages physicians from reporting on senior colleagues.”
Physician self-assessment is yet another option, but “loss of insight may be a component of an individual’s impairment,” the authors wrote.
So what’s the best solution? The authors recommended “a relatively brief cognitive screening followed by more extensive testing for the most impaired individuals.” This approach “appears most reliable in confidentially identifying truly impaired physicians while minimizing the chance of a falsely flagging unimpaired individuals. This strategy allows aging physicians to continue working while safeguarding both their reputations and their patients’ health.”
Ms. Hoffman has no disclosures.
A version of this article first appeared on Medscape.com.
Review finds no CV or VTE risk signal with use of JAK inhibitors for skin indications
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
, results from a systematic literature review, and meta-analysis showed.
“There remains a knowledge gap regarding the risk of JAK inhibitor use and VTE and/or MACE in the dermatologic population,” researchers led by Michael S. Garshick, MD, a cardiologist at New York University Langone Health, wrote in their study, which was published online in JAMA Dermatology . “Pooled safety studies suggest that the risk of MACE and VTE may be lower in patients treated with JAK inhibitors for a dermatologic indication than the risk observed in the ORAL Surveillance study, which may be related to the younger age and better health status of those enrolled in trials for dermatologic indications.” The results of that study, which included patients with rheumatoid arthritis only, resulted in the addition of a boxed warning in the labels for topical and oral JAK inhibitors regarding the increased risk of MACE, VTE, serious infections, malignancies, and death .
For the review – thought to be the first to specifically evaluate these risks for dermatologic indications – the researchers searched PubMed and ClinicalTrials.gov from inception through April 1, 2023, for phase 3 dermatology randomized clinical trials (RCTs) to evaluate the risk of MACE, VTE, and all-cause mortality with JAK inhibitors, compared with placebo or an active comparator in the treatment of immune-mediated inflammatory skin diseases. They followed Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and used a random-effects model and the DerSimonian-Laird method to calculate adverse events with odds ratios.
The database search yielded 35 RCTs with a total of 20,651 patients. Their mean age was 38.5 years, 54% were male, and the mean follow-up time was 4.9 months. Of the 35 trials, most (21) involved patients with atopic dermatitis, followed by psoriasis/psoriatic arthritis (9 trials), alopecia areata (3 trials) and vitiligo (2 trials).
The researchers found no significant difference between JAK inhibitors and placebo/active comparator in composite MACE and all-cause mortality (odds ratio, 0.83; 95% confidence interval, 0.44-1.57) or in VTE (OR, 0.52; 95% CI, 0.26-1.04).
In a secondary analysis, which included additional psoriatic arthritis RCTs, no significant differences between the treatment and placebo/active comparator groups were observed. Similarly, subgroup analyses of oral versus topical JAK inhibitors and a sensitivity analysis that excluded pediatric trials showed no significant differences between patients exposed to JAK inhibitors and those not exposed.
The researchers acknowledged certain limitations of the review, including the lack of access to patient-level data, the fact that most trials only included short-term follow-up, and that the findings have limited generalizability to an older patient population. “It remains unclear if the cardiovascular risks of JAK inhibitors are primarily due to patient level cardiovascular risk factors or are drug mediated,” they concluded. “Dermatologists should carefully select patients and assess baseline cardiovascular risk factors when considering JAK therapy. Cardiovascular risk assessment should continue for the duration of treatment.”
Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, who was asked to comment on the study results, characterized the findings as reassuring to dermatologists who may be reluctant to initiate therapy with JAK inhibitors based on concerns about safety signals for MACE, VTE, and all-cause mortality.
“These data systematically show that across medications and across conditions, there doesn’t appear to be an increased signal for these events during the short-term, placebo-controlled period which generally spans a few months in most studies,” he told this news organization. The findings, he added, “align well with our clinical experience to date for JAK inhibitor use in inflammatory skin disease. Short-term safety, particularly in relation to boxed warning events such MACE, VTE, and all-cause mortality, have generally been favorable with real-world use. It’s good to have a rigorous statistical analysis to refer to when setting patient expectations.”
However, he noted that these data only examined short-term safety during the placebo or active comparator-controlled periods. “Considering that events like MACE or VTE may take many months or years to manifest, continued long-term data generation is needed to fully answer the question of risk,” he said.
Dr. Garshick disclosed that he received grants from Pfizer and personal fees from Bristol Myers Squibb during the conduct of the study and personal fees from Kiniksa Pharmaceuticals outside the submitted work. Several other coauthors reported having advisory board roles and/or having received funding or support from several pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including those that develop JAK inhibitors.
FROM JAMA DERMATOLOGY