User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
FDA cancels or postpones meetings amid COVID-19 concerns
Officials at the Food and Drug Administration’s Center for Drug Evaluation and Research are taking the precautionary step of canceling or postponing advisory committee meetings and limiting staff travel in an effort to help curb the spread of the COVID-19.
“The outbreak of respiratory illness caused by a novel coronavirus, COVID-19, that started in China is spreading to other countries, including the United States,” CDER Director Janet Woodcock, MD, said in a memo to CDER staff. “As a precaution, FDA is canceling foreign official agency travel and limiting domestic travel to mission critical only, effective immediately and through April.”
Additionally, the memo notes that “CDER-organized external meetings, conferences, and workshops will be postponed or canceled from March 10 through April.”
“To mitigate the impact on our work, I encourage you to hold meetings with external stakeholders through teleconference, when possible,” she wrote.
Thus far, only a few CDER events on the FDA’s meeting webpage are listed as being canceled or postponed. Some of the affected meetings include a March 10 public meeting on patient-focused drug development for stimulant-use disorder, a March 11 meeting of the Nonprescription Drug Advisory Committee, and a March 30 public meeting on patient-focused drug development for vitiligo, all of which are postponed until further notice. The Center for Biologics Evaluation and Research also has postponed until further notice its U.S.–Japan Cellular and Gene Therapy Conference, originally scheduled for March 12.
Dr. Woodcock also noted in the memo that in relation to inspections, “we plan to use technology and established agreements with our foreign counterparts to minimize disruptions to the drug supply chain and to applications under review, so that Americans can continue to get their medications.”
Officials at the Food and Drug Administration’s Center for Drug Evaluation and Research are taking the precautionary step of canceling or postponing advisory committee meetings and limiting staff travel in an effort to help curb the spread of the COVID-19.
“The outbreak of respiratory illness caused by a novel coronavirus, COVID-19, that started in China is spreading to other countries, including the United States,” CDER Director Janet Woodcock, MD, said in a memo to CDER staff. “As a precaution, FDA is canceling foreign official agency travel and limiting domestic travel to mission critical only, effective immediately and through April.”
Additionally, the memo notes that “CDER-organized external meetings, conferences, and workshops will be postponed or canceled from March 10 through April.”
“To mitigate the impact on our work, I encourage you to hold meetings with external stakeholders through teleconference, when possible,” she wrote.
Thus far, only a few CDER events on the FDA’s meeting webpage are listed as being canceled or postponed. Some of the affected meetings include a March 10 public meeting on patient-focused drug development for stimulant-use disorder, a March 11 meeting of the Nonprescription Drug Advisory Committee, and a March 30 public meeting on patient-focused drug development for vitiligo, all of which are postponed until further notice. The Center for Biologics Evaluation and Research also has postponed until further notice its U.S.–Japan Cellular and Gene Therapy Conference, originally scheduled for March 12.
Dr. Woodcock also noted in the memo that in relation to inspections, “we plan to use technology and established agreements with our foreign counterparts to minimize disruptions to the drug supply chain and to applications under review, so that Americans can continue to get their medications.”
Officials at the Food and Drug Administration’s Center for Drug Evaluation and Research are taking the precautionary step of canceling or postponing advisory committee meetings and limiting staff travel in an effort to help curb the spread of the COVID-19.
“The outbreak of respiratory illness caused by a novel coronavirus, COVID-19, that started in China is spreading to other countries, including the United States,” CDER Director Janet Woodcock, MD, said in a memo to CDER staff. “As a precaution, FDA is canceling foreign official agency travel and limiting domestic travel to mission critical only, effective immediately and through April.”
Additionally, the memo notes that “CDER-organized external meetings, conferences, and workshops will be postponed or canceled from March 10 through April.”
“To mitigate the impact on our work, I encourage you to hold meetings with external stakeholders through teleconference, when possible,” she wrote.
Thus far, only a few CDER events on the FDA’s meeting webpage are listed as being canceled or postponed. Some of the affected meetings include a March 10 public meeting on patient-focused drug development for stimulant-use disorder, a March 11 meeting of the Nonprescription Drug Advisory Committee, and a March 30 public meeting on patient-focused drug development for vitiligo, all of which are postponed until further notice. The Center for Biologics Evaluation and Research also has postponed until further notice its U.S.–Japan Cellular and Gene Therapy Conference, originally scheduled for March 12.
Dr. Woodcock also noted in the memo that in relation to inspections, “we plan to use technology and established agreements with our foreign counterparts to minimize disruptions to the drug supply chain and to applications under review, so that Americans can continue to get their medications.”
Age does not appear to affect efficacy of siponimod in secondary progressive MS
WEST PALM BEACH, FLA. – (MS), according to data presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. Siponimod’s tolerability also does not vary with age, the researchers said.
Although many effective treatments for relapsing-remitting MS are available, patients still have a high risk of developing secondary progressive MS. Increasing age is associated with disability accumulation, regardless of disease duration. This accumulation of disability could affect treatment outcomes.
Siponimod is an approved treatment for adults with relapsing forms of MS, including clinically isolated syndrome and active secondary progressive MS. Compared with placebo, siponimod significantly reduced the risk of confirmed disability progression in the phase 3 EXPAND trial.
Efficacy was similar between age groups
Le Hua, MD, a neurologist at the Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas, and colleagues sought to evaluate the safety and efficacy of siponimod by age in patients with active secondary progressive MS who participated in the EXPAND study. The investigators defined active disease as a relapse in the 2 years before screening or one or more T1 gadolinium–enhancing lesions at baseline. Time to 3- and 6-month confirmed disability progression, defined using Expanded Disability Status Scale (EDSS) scores, were the efficacy endpoints of this analysis. Dr. Hua and colleagues also examined adverse events, serious adverse events, and adverse events leading to treatment discontinuation.
Of the 1,651 patients included in EXPAND, 779 had active secondary progressive MS. Dr. Hua and colleagues categorized this group as younger than 45 years (306 patients) or 45 years or older (473 patients). In the overall EXPAND population, siponimod reduced the risk of 3-month confirmed disability progression by 21% and the risk of 6-month confirmed disability progression by 26%, compared with placebo. Among patients younger than 45 years, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 32% and 40%, respectively. Among patients aged 45 years or older, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 31% and 33%, respectively.
Siponimod’s safety profile was similar between age groups. Among participants younger than 45 years, the rate of any adverse event was 82.6% for siponimod and 82.8% for placebo. In patients age 45 years or older, the rate of any adverse event was 89.8% for siponimod and 75.9% for placebo. The rate of serious adverse events was lower for siponimod than for placebo in both age groups. Among patients younger than 45 years, the rate was 12.7% for siponimod and 15.1% for placebo. Among patients age 45 years or older, the rate was 18.2% for siponimod and 19.4% for placebo. The rate of adverse events leading to discontinuation, however, was slightly higher among older patients. For younger patients, the rate was 3.3% for siponimod and 4.3% for placebo. For older patients, the rate was 7.6% for siponimod and 7.1% for placebo.
Real-world data will provide clearer answers
Rates of adverse events, serious adverse events, and adverse events leading to discontinuation were all higher in patients aged 45 or older. “This was not a surprising finding,” said Dr. Hua. “The differences appeared small, and the study was not powered to detect if these differences were significant. ... In general, older patients are more likely to have more cardiac events and infections than younger patients.”
Few studies have analyzed the efficacy and safety of MS therapy by age, said Dr. Hua. In 2015, Matell et al. evaluated the effectiveness of natalizumab in patients over age 50 years. In that study, a greater number of older patients, compared with younger patients, stopped treatment because of lack of effectiveness. Studies of other disease-modifying therapies (DMTs) have included subgroup analyses based on age, “but subgroup analyses are limited in the ability to make any strong conclusions regarding significant differences in safety,” said Dr. Hua. “A recent meta-analysis of published clinical trials of all DMTs performed by Weideman et al. indicated that the efficacy of DMTs appears to wane after age 53 years. However, there are limitations in interpreting this data, as they weren’t able to evaluate the raw data. And in most clinical trials, the age cutoff is usually 55 years, which limits generalizations. Real-world data analyses are needed to truly understand efficacy and safety of DMT in older patients.”
Novartis funded the study. Dr. Hua received fees from Novartis, Biogen, Celgene, EMD Serono, Genentech, and Genzyme.
SOURCE: Hua L et al. ACTRIMS FORUM 2020, Abstract P029.
WEST PALM BEACH, FLA. – (MS), according to data presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. Siponimod’s tolerability also does not vary with age, the researchers said.
Although many effective treatments for relapsing-remitting MS are available, patients still have a high risk of developing secondary progressive MS. Increasing age is associated with disability accumulation, regardless of disease duration. This accumulation of disability could affect treatment outcomes.
Siponimod is an approved treatment for adults with relapsing forms of MS, including clinically isolated syndrome and active secondary progressive MS. Compared with placebo, siponimod significantly reduced the risk of confirmed disability progression in the phase 3 EXPAND trial.
Efficacy was similar between age groups
Le Hua, MD, a neurologist at the Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas, and colleagues sought to evaluate the safety and efficacy of siponimod by age in patients with active secondary progressive MS who participated in the EXPAND study. The investigators defined active disease as a relapse in the 2 years before screening or one or more T1 gadolinium–enhancing lesions at baseline. Time to 3- and 6-month confirmed disability progression, defined using Expanded Disability Status Scale (EDSS) scores, were the efficacy endpoints of this analysis. Dr. Hua and colleagues also examined adverse events, serious adverse events, and adverse events leading to treatment discontinuation.
Of the 1,651 patients included in EXPAND, 779 had active secondary progressive MS. Dr. Hua and colleagues categorized this group as younger than 45 years (306 patients) or 45 years or older (473 patients). In the overall EXPAND population, siponimod reduced the risk of 3-month confirmed disability progression by 21% and the risk of 6-month confirmed disability progression by 26%, compared with placebo. Among patients younger than 45 years, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 32% and 40%, respectively. Among patients aged 45 years or older, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 31% and 33%, respectively.
Siponimod’s safety profile was similar between age groups. Among participants younger than 45 years, the rate of any adverse event was 82.6% for siponimod and 82.8% for placebo. In patients age 45 years or older, the rate of any adverse event was 89.8% for siponimod and 75.9% for placebo. The rate of serious adverse events was lower for siponimod than for placebo in both age groups. Among patients younger than 45 years, the rate was 12.7% for siponimod and 15.1% for placebo. Among patients age 45 years or older, the rate was 18.2% for siponimod and 19.4% for placebo. The rate of adverse events leading to discontinuation, however, was slightly higher among older patients. For younger patients, the rate was 3.3% for siponimod and 4.3% for placebo. For older patients, the rate was 7.6% for siponimod and 7.1% for placebo.
Real-world data will provide clearer answers
Rates of adverse events, serious adverse events, and adverse events leading to discontinuation were all higher in patients aged 45 or older. “This was not a surprising finding,” said Dr. Hua. “The differences appeared small, and the study was not powered to detect if these differences were significant. ... In general, older patients are more likely to have more cardiac events and infections than younger patients.”
Few studies have analyzed the efficacy and safety of MS therapy by age, said Dr. Hua. In 2015, Matell et al. evaluated the effectiveness of natalizumab in patients over age 50 years. In that study, a greater number of older patients, compared with younger patients, stopped treatment because of lack of effectiveness. Studies of other disease-modifying therapies (DMTs) have included subgroup analyses based on age, “but subgroup analyses are limited in the ability to make any strong conclusions regarding significant differences in safety,” said Dr. Hua. “A recent meta-analysis of published clinical trials of all DMTs performed by Weideman et al. indicated that the efficacy of DMTs appears to wane after age 53 years. However, there are limitations in interpreting this data, as they weren’t able to evaluate the raw data. And in most clinical trials, the age cutoff is usually 55 years, which limits generalizations. Real-world data analyses are needed to truly understand efficacy and safety of DMT in older patients.”
Novartis funded the study. Dr. Hua received fees from Novartis, Biogen, Celgene, EMD Serono, Genentech, and Genzyme.
SOURCE: Hua L et al. ACTRIMS FORUM 2020, Abstract P029.
WEST PALM BEACH, FLA. – (MS), according to data presented at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. Siponimod’s tolerability also does not vary with age, the researchers said.
Although many effective treatments for relapsing-remitting MS are available, patients still have a high risk of developing secondary progressive MS. Increasing age is associated with disability accumulation, regardless of disease duration. This accumulation of disability could affect treatment outcomes.
Siponimod is an approved treatment for adults with relapsing forms of MS, including clinically isolated syndrome and active secondary progressive MS. Compared with placebo, siponimod significantly reduced the risk of confirmed disability progression in the phase 3 EXPAND trial.
Efficacy was similar between age groups
Le Hua, MD, a neurologist at the Cleveland Clinic Lou Ruvo Center for Brain Health in Las Vegas, and colleagues sought to evaluate the safety and efficacy of siponimod by age in patients with active secondary progressive MS who participated in the EXPAND study. The investigators defined active disease as a relapse in the 2 years before screening or one or more T1 gadolinium–enhancing lesions at baseline. Time to 3- and 6-month confirmed disability progression, defined using Expanded Disability Status Scale (EDSS) scores, were the efficacy endpoints of this analysis. Dr. Hua and colleagues also examined adverse events, serious adverse events, and adverse events leading to treatment discontinuation.
Of the 1,651 patients included in EXPAND, 779 had active secondary progressive MS. Dr. Hua and colleagues categorized this group as younger than 45 years (306 patients) or 45 years or older (473 patients). In the overall EXPAND population, siponimod reduced the risk of 3-month confirmed disability progression by 21% and the risk of 6-month confirmed disability progression by 26%, compared with placebo. Among patients younger than 45 years, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 32% and 40%, respectively. Among patients aged 45 years or older, siponimod reduced the risks of 3- and 6-month confirmed disability progression by 31% and 33%, respectively.
Siponimod’s safety profile was similar between age groups. Among participants younger than 45 years, the rate of any adverse event was 82.6% for siponimod and 82.8% for placebo. In patients age 45 years or older, the rate of any adverse event was 89.8% for siponimod and 75.9% for placebo. The rate of serious adverse events was lower for siponimod than for placebo in both age groups. Among patients younger than 45 years, the rate was 12.7% for siponimod and 15.1% for placebo. Among patients age 45 years or older, the rate was 18.2% for siponimod and 19.4% for placebo. The rate of adverse events leading to discontinuation, however, was slightly higher among older patients. For younger patients, the rate was 3.3% for siponimod and 4.3% for placebo. For older patients, the rate was 7.6% for siponimod and 7.1% for placebo.
Real-world data will provide clearer answers
Rates of adverse events, serious adverse events, and adverse events leading to discontinuation were all higher in patients aged 45 or older. “This was not a surprising finding,” said Dr. Hua. “The differences appeared small, and the study was not powered to detect if these differences were significant. ... In general, older patients are more likely to have more cardiac events and infections than younger patients.”
Few studies have analyzed the efficacy and safety of MS therapy by age, said Dr. Hua. In 2015, Matell et al. evaluated the effectiveness of natalizumab in patients over age 50 years. In that study, a greater number of older patients, compared with younger patients, stopped treatment because of lack of effectiveness. Studies of other disease-modifying therapies (DMTs) have included subgroup analyses based on age, “but subgroup analyses are limited in the ability to make any strong conclusions regarding significant differences in safety,” said Dr. Hua. “A recent meta-analysis of published clinical trials of all DMTs performed by Weideman et al. indicated that the efficacy of DMTs appears to wane after age 53 years. However, there are limitations in interpreting this data, as they weren’t able to evaluate the raw data. And in most clinical trials, the age cutoff is usually 55 years, which limits generalizations. Real-world data analyses are needed to truly understand efficacy and safety of DMT in older patients.”
Novartis funded the study. Dr. Hua received fees from Novartis, Biogen, Celgene, EMD Serono, Genentech, and Genzyme.
SOURCE: Hua L et al. ACTRIMS FORUM 2020, Abstract P029.
REPORTING FROM ACTRIMS FORUM 2020
American Headache Society updates guideline on neuroimaging for migraine
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
Migraine with atypical features may require neuroimaging, according to the guideline. These include an unusual aura; change in clinical features; a first or worst migraine; a migraine that presents with brainstem aura, confusion, or motor manifestation; migraine accompaniments in later life; headaches that are side-locked or posttraumatic; and aura that presents without headache.
Assessing the evidence
The recommendation to avoid MRI or CT in otherwise neurologically normal patients with migraine carried a grade A recommendation from the American Headache Society, while the specific considerations for neuroimaging was based on consensus and carried a grade C recommendation, according to lead author Randolph W. Evans, MD, of the department of neurology at Baylor College of Medicine in Houston, and colleagues.
The recommendations, published in the journal Headache (2020 Feb;60(2):318-36), came from a systematic review of 23 studies of adults at least 18 years old who underwent MRI or CT during outpatient treatment for migraine between 1973 and 2018. Ten studies looked at CT neuroimaging in patients with migraine, nine studies examined MRI neuroimaging alone in patients with migraine, and four studies contained adults with headache or migraine who underwent either MRI or CT. The majority of studies analyzed were retrospective or cross-sectional in nature, while four studies were prospective observational studies.
Dr. Evans and colleagues noted that neuroimaging for patients with suspected migraine is ordered for a variety of reasons, such as excluding conditions that aren’t migraine, diagnostic certainty, cognitive bias, practice workflow, medicolegal concerns, addressing patient and family anxiety, and addressing clinician anxiety. Neuroimaging also can be costly, they said, adding up to an estimated $1 billion annually according to one study, and can lead to additional testing from findings that may not be clinically significant.
Good advice, with caveats
In an interview, Alan M. Rapoport, MD, editor-in-chief of Neurology Reviews, said that while he generally does not like broad guideline recommendations, the recommendation made by the American Headache Society to avoid neuroimaging in patients with a normal neurological examination without any atypical features and red flags “takes most of the important factors into consideration and will work almost all the time.” The recommendation made by consensus for specific considerations of neuroimaging was issued by top headache specialists in the United States who reviewed the data, and it is unlikely a patient with a migraine as diagnosed by the International Classification of Headache Disorders with a normal neurological examination would have a significant abnormality that would appear with imaging, Dr. Rapoport said.
“If everyone caring for migraine patients knew these recommendations, and used them unless the patients fit the exclusions mentioned, we would have more efficient clinical practice and save lots of money on unnecessary scanning,” he said.
However, Dr. Rapoport, clinical professor of neurology at the University of California, Los Angeles, founder of the New England Center for Headache, and past president of The International Headache Society, said that not all clinicians will be convinced by the American Headache Society’s recommendations.
“Various third parties often jump on society recommendations or guidelines and prevent smart clinicians from doing what they need to do when they want to disregard the recommendation or guideline,” he explained. “More importantly, if a physician feels the need to think out of the box and image a patient without a clear reason, and the patient cannot pay for the scan when a medical insurance company refuses to authorize it, there can be a bad result if the patient does not get the study.”
Dr. Rapoport noted that the guideline does not address situations where neuroimaging may not pick up conditions that lead to migraine, such as a subarachnoid or subdural hemorrhage, reversible cerebral vasoconstriction syndrome, or early aspects of low cerebrospinal fluid pressure syndrome. Anxiety on the part of the patient or the clinician is another area that can be addressed by future research, he said.
“If the clinician does a good job of explaining the odds of anything significant being found with a typical migraine history and normal examination, and the patient says [they] need an MRI with contrast to be sure, it will be difficult to dissuade them,” said Dr. Rapoport. “If you don’t order one, they will find a way to get one. If it is abnormal, you could be in trouble. Also, if the clinician has no good reason to do a scan but has anxiety about what is being missed, it will probably get done.”
There was no funding source for the guidelines. The authors reported personal and institutional relationships in the form of advisory board memberships, investigator appointments, speakers bureau positions, research support, and consultancies for a variety of pharmaceutical companies, agencies, institutions, publishers, and other organizations.
FROM HEADACHE
Hyperlipidemia occurs earlier in patients with MS
WEST PALM BEACH, FLA. – Onset of hyperlipidemia tends to occur earlier in patients with multiple sclerosis (MS), compared with matched controls, according to new research. Among females and African Americans, the effect of MS on age of hyperlipidemia onset may be especially pronounced, said Diane Krill, PhD, professor of biological sciences at Point Park University, Pittsburgh, and colleagues.
Many patients with MS have hyperlipidemia, and adverse lipid profiles correlate with physical and cognitive impairment in this population. “There is evidence of endothelial dysfunction and inflammation in both MS and hyperlipidemia, but the timing of onset of hyperlipidemia is not known,” the researchers said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To assess whether patients with MS have hyperlipidemia diagnosed at an earlier age, relative to matched controls, Dr. Krill and colleagues analyzed data from the Cleveland Clinic health system. They included in their analyses patients with at least two hyperlipidemia diagnoses and at least five encounters with a primary care physician. They matched each patient with MS to four patients without MS using variables such as birth year, sex, race, and year of first encounter. In all, the study included 669 patients with MS and 2,676 controls. The investigators examined age of hyperlipidemia onset using multivariable Cox proportional hazard models that adjusted for sex, race, smoking, and body mass index.
Patients with MS had a 20% increased risk for earlier onset of hyperlipidemia, relative to matched controls. The effect was greater among females (hazard ratio, 1.22) and African Americans (HR, 1.42). Patients with MS have earlier onset of hyperlipidemia “irrespective of the relationship with age of MS onset,” Dr. Krill and colleagues noted. “Additional research is warranted to further characterize the temporal relationships between MS and hyperlipidemia, as well as considerations for timing of disease-modifying therapies.”
The researchers had no relevant disclosures.
SOURCE: Krill D et al. ACTRIMS Forum 2020, Abstract P085.
WEST PALM BEACH, FLA. – Onset of hyperlipidemia tends to occur earlier in patients with multiple sclerosis (MS), compared with matched controls, according to new research. Among females and African Americans, the effect of MS on age of hyperlipidemia onset may be especially pronounced, said Diane Krill, PhD, professor of biological sciences at Point Park University, Pittsburgh, and colleagues.
Many patients with MS have hyperlipidemia, and adverse lipid profiles correlate with physical and cognitive impairment in this population. “There is evidence of endothelial dysfunction and inflammation in both MS and hyperlipidemia, but the timing of onset of hyperlipidemia is not known,” the researchers said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To assess whether patients with MS have hyperlipidemia diagnosed at an earlier age, relative to matched controls, Dr. Krill and colleagues analyzed data from the Cleveland Clinic health system. They included in their analyses patients with at least two hyperlipidemia diagnoses and at least five encounters with a primary care physician. They matched each patient with MS to four patients without MS using variables such as birth year, sex, race, and year of first encounter. In all, the study included 669 patients with MS and 2,676 controls. The investigators examined age of hyperlipidemia onset using multivariable Cox proportional hazard models that adjusted for sex, race, smoking, and body mass index.
Patients with MS had a 20% increased risk for earlier onset of hyperlipidemia, relative to matched controls. The effect was greater among females (hazard ratio, 1.22) and African Americans (HR, 1.42). Patients with MS have earlier onset of hyperlipidemia “irrespective of the relationship with age of MS onset,” Dr. Krill and colleagues noted. “Additional research is warranted to further characterize the temporal relationships between MS and hyperlipidemia, as well as considerations for timing of disease-modifying therapies.”
The researchers had no relevant disclosures.
SOURCE: Krill D et al. ACTRIMS Forum 2020, Abstract P085.
WEST PALM BEACH, FLA. – Onset of hyperlipidemia tends to occur earlier in patients with multiple sclerosis (MS), compared with matched controls, according to new research. Among females and African Americans, the effect of MS on age of hyperlipidemia onset may be especially pronounced, said Diane Krill, PhD, professor of biological sciences at Point Park University, Pittsburgh, and colleagues.
Many patients with MS have hyperlipidemia, and adverse lipid profiles correlate with physical and cognitive impairment in this population. “There is evidence of endothelial dysfunction and inflammation in both MS and hyperlipidemia, but the timing of onset of hyperlipidemia is not known,” the researchers said at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To assess whether patients with MS have hyperlipidemia diagnosed at an earlier age, relative to matched controls, Dr. Krill and colleagues analyzed data from the Cleveland Clinic health system. They included in their analyses patients with at least two hyperlipidemia diagnoses and at least five encounters with a primary care physician. They matched each patient with MS to four patients without MS using variables such as birth year, sex, race, and year of first encounter. In all, the study included 669 patients with MS and 2,676 controls. The investigators examined age of hyperlipidemia onset using multivariable Cox proportional hazard models that adjusted for sex, race, smoking, and body mass index.
Patients with MS had a 20% increased risk for earlier onset of hyperlipidemia, relative to matched controls. The effect was greater among females (hazard ratio, 1.22) and African Americans (HR, 1.42). Patients with MS have earlier onset of hyperlipidemia “irrespective of the relationship with age of MS onset,” Dr. Krill and colleagues noted. “Additional research is warranted to further characterize the temporal relationships between MS and hyperlipidemia, as well as considerations for timing of disease-modifying therapies.”
The researchers had no relevant disclosures.
SOURCE: Krill D et al. ACTRIMS Forum 2020, Abstract P085.
REPORTING FROM ACTRIMS FORUM 2020
Some infected patients could show COVID-19 symptoms after quarantine
Although a 14-day quarantine after exposure to novel coronavirus is “well supported” by evidence, some infected individuals will not become symptomatic until after that period, according to authors of a recent analysis published in Annals of Internal Medicine.
Most individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) will develop symptoms by day 12 of the infection, which is within the 14-day period of active monitoring currently recommended by the Centers for Disease Control and Prevention, the authors wrote.
However, an estimated 101 out of 10,000 cases could become symptomatic after the end of that 14-day monitoring period, they cautioned.
“Our analyses do not preclude that estimate from being higher,” said the investigators, led by Stephen A. Lauer, PhD, MD, of Johns Hopkins Bloomberg School of Public Health, Baltimore.
The analysis, based on 181 confirmed cases of coronavirus disease 2019 (COVID-19) that were documented outside of the outbreak epicenter, Wuhan, China, makes “more conservative assumptions” about the window of symptom onset and potential for continued exposure, compared with analyses in previous studies, the researchers wrote.
The estimated incubation period for SARS-CoV-2 in the 181-patient study was a median of 5.1 days, which is comparable with previous estimates based on COVID-19 cases outside of Wuhan and consistent with other known human coronavirus diseases, such as SARS, which had a reported mean incubation period of 5 days, Dr. Lauer and colleagues noted.
Symptoms developed within 11.5 days for 97.5% of patients in the study.
Whether it’s acceptable to have 101 out of 10,000 cases becoming symptomatic beyond the recommended quarantine window depends on two factors, according to the authors. The first is the expected infection risk in the population that is being monitored, and the second is “judgment about the cost of missing cases,” wrote the authors.
In an interview, Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau, Oceanside, N.Y., said that in practical terms, the results suggest that the majority of patients with COVID-19 will be identified within 14 days, with an “outside chance” of an infected individual leaving quarantine and transmitting virus for a short period of time before becoming symptomatic.
“I think the proper message to give those patients [who are asymptomatic upon leaving quarantine] is, ‘after 14 days, we’re pretty sure you’re out of the woods, but should you get any symptoms, immediately requarantine yourself and seek medical care,” he said.
Study coauthor Kyra H. Grantz, a doctoral graduate student at the Johns Hopkins Bloomberg School of Public Health, said that extending a quarantine beyond 14 days might be considered in the highest-risk scenarios, though the benefits of doing so would have to be weighed against the costs to public health and to the individuals under quarantine.
“Our estimate of the incubation period definitely supports the 14-day recommendation that the CDC has been using,” she said in an interview.
Dr. Grantz emphasized that the estimate of 101 out of 10,000 cases developing symptoms after day 14 of active monitoring – representing the 99th percentile of cases – assumes the “most conservative, worst-case scenario” in a population that is fully infected.
“If you’re looking at a following a cohort of 1,000 people whom you think may have been exposed, only a certain percentage will be infected, and only a certain percentage of those will even develop symptoms – before we get to this idea of how many people would we miss,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Institute of Allergy and Infectious Diseases, the National Institute of General Medical Sciences, and the Alexander von Humboldt Foundation. Four authors reported disclosures related to those entities, and the remaining five reported no conflicts of interest.
SOURCE: Lauer SA et al. Ann Intern Med. 2020 Mar 9. doi:10.1101/2020.02.02.20020016.
Although a 14-day quarantine after exposure to novel coronavirus is “well supported” by evidence, some infected individuals will not become symptomatic until after that period, according to authors of a recent analysis published in Annals of Internal Medicine.
Most individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) will develop symptoms by day 12 of the infection, which is within the 14-day period of active monitoring currently recommended by the Centers for Disease Control and Prevention, the authors wrote.
However, an estimated 101 out of 10,000 cases could become symptomatic after the end of that 14-day monitoring period, they cautioned.
“Our analyses do not preclude that estimate from being higher,” said the investigators, led by Stephen A. Lauer, PhD, MD, of Johns Hopkins Bloomberg School of Public Health, Baltimore.
The analysis, based on 181 confirmed cases of coronavirus disease 2019 (COVID-19) that were documented outside of the outbreak epicenter, Wuhan, China, makes “more conservative assumptions” about the window of symptom onset and potential for continued exposure, compared with analyses in previous studies, the researchers wrote.
The estimated incubation period for SARS-CoV-2 in the 181-patient study was a median of 5.1 days, which is comparable with previous estimates based on COVID-19 cases outside of Wuhan and consistent with other known human coronavirus diseases, such as SARS, which had a reported mean incubation period of 5 days, Dr. Lauer and colleagues noted.
Symptoms developed within 11.5 days for 97.5% of patients in the study.
Whether it’s acceptable to have 101 out of 10,000 cases becoming symptomatic beyond the recommended quarantine window depends on two factors, according to the authors. The first is the expected infection risk in the population that is being monitored, and the second is “judgment about the cost of missing cases,” wrote the authors.
In an interview, Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau, Oceanside, N.Y., said that in practical terms, the results suggest that the majority of patients with COVID-19 will be identified within 14 days, with an “outside chance” of an infected individual leaving quarantine and transmitting virus for a short period of time before becoming symptomatic.
“I think the proper message to give those patients [who are asymptomatic upon leaving quarantine] is, ‘after 14 days, we’re pretty sure you’re out of the woods, but should you get any symptoms, immediately requarantine yourself and seek medical care,” he said.
Study coauthor Kyra H. Grantz, a doctoral graduate student at the Johns Hopkins Bloomberg School of Public Health, said that extending a quarantine beyond 14 days might be considered in the highest-risk scenarios, though the benefits of doing so would have to be weighed against the costs to public health and to the individuals under quarantine.
“Our estimate of the incubation period definitely supports the 14-day recommendation that the CDC has been using,” she said in an interview.
Dr. Grantz emphasized that the estimate of 101 out of 10,000 cases developing symptoms after day 14 of active monitoring – representing the 99th percentile of cases – assumes the “most conservative, worst-case scenario” in a population that is fully infected.
“If you’re looking at a following a cohort of 1,000 people whom you think may have been exposed, only a certain percentage will be infected, and only a certain percentage of those will even develop symptoms – before we get to this idea of how many people would we miss,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Institute of Allergy and Infectious Diseases, the National Institute of General Medical Sciences, and the Alexander von Humboldt Foundation. Four authors reported disclosures related to those entities, and the remaining five reported no conflicts of interest.
SOURCE: Lauer SA et al. Ann Intern Med. 2020 Mar 9. doi:10.1101/2020.02.02.20020016.
Although a 14-day quarantine after exposure to novel coronavirus is “well supported” by evidence, some infected individuals will not become symptomatic until after that period, according to authors of a recent analysis published in Annals of Internal Medicine.
Most individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) will develop symptoms by day 12 of the infection, which is within the 14-day period of active monitoring currently recommended by the Centers for Disease Control and Prevention, the authors wrote.
However, an estimated 101 out of 10,000 cases could become symptomatic after the end of that 14-day monitoring period, they cautioned.
“Our analyses do not preclude that estimate from being higher,” said the investigators, led by Stephen A. Lauer, PhD, MD, of Johns Hopkins Bloomberg School of Public Health, Baltimore.
The analysis, based on 181 confirmed cases of coronavirus disease 2019 (COVID-19) that were documented outside of the outbreak epicenter, Wuhan, China, makes “more conservative assumptions” about the window of symptom onset and potential for continued exposure, compared with analyses in previous studies, the researchers wrote.
The estimated incubation period for SARS-CoV-2 in the 181-patient study was a median of 5.1 days, which is comparable with previous estimates based on COVID-19 cases outside of Wuhan and consistent with other known human coronavirus diseases, such as SARS, which had a reported mean incubation period of 5 days, Dr. Lauer and colleagues noted.
Symptoms developed within 11.5 days for 97.5% of patients in the study.
Whether it’s acceptable to have 101 out of 10,000 cases becoming symptomatic beyond the recommended quarantine window depends on two factors, according to the authors. The first is the expected infection risk in the population that is being monitored, and the second is “judgment about the cost of missing cases,” wrote the authors.
In an interview, Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau, Oceanside, N.Y., said that in practical terms, the results suggest that the majority of patients with COVID-19 will be identified within 14 days, with an “outside chance” of an infected individual leaving quarantine and transmitting virus for a short period of time before becoming symptomatic.
“I think the proper message to give those patients [who are asymptomatic upon leaving quarantine] is, ‘after 14 days, we’re pretty sure you’re out of the woods, but should you get any symptoms, immediately requarantine yourself and seek medical care,” he said.
Study coauthor Kyra H. Grantz, a doctoral graduate student at the Johns Hopkins Bloomberg School of Public Health, said that extending a quarantine beyond 14 days might be considered in the highest-risk scenarios, though the benefits of doing so would have to be weighed against the costs to public health and to the individuals under quarantine.
“Our estimate of the incubation period definitely supports the 14-day recommendation that the CDC has been using,” she said in an interview.
Dr. Grantz emphasized that the estimate of 101 out of 10,000 cases developing symptoms after day 14 of active monitoring – representing the 99th percentile of cases – assumes the “most conservative, worst-case scenario” in a population that is fully infected.
“If you’re looking at a following a cohort of 1,000 people whom you think may have been exposed, only a certain percentage will be infected, and only a certain percentage of those will even develop symptoms – before we get to this idea of how many people would we miss,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Institute of Allergy and Infectious Diseases, the National Institute of General Medical Sciences, and the Alexander von Humboldt Foundation. Four authors reported disclosures related to those entities, and the remaining five reported no conflicts of interest.
SOURCE: Lauer SA et al. Ann Intern Med. 2020 Mar 9. doi:10.1101/2020.02.02.20020016.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Some individuals who are infected with the novel coronavirus could become symptomatic after the active 14-day quarantine period.
Major finding: The median incubation period was 5.1 days, with 97.5% of patients developing symptoms within 11.5 days, implying that 101 of every 10,000 cases (99th percentile) would develop symptoms beyond the quarantine period.
Study details: Analysis of 181 confirmed COVID-19 cases identified outside of the outbreak epicenter, Wuhan, China.
Disclosures: The study was supported by the U.S. Centers for Disease Control and Prevention, the National Institute of Allergy and Infectious Diseases, the National Institute of General Medical Sciences, and the Alexander von Humboldt Foundation. Four authors reported disclosures related to those entities, and the remaining five reported no conflicts of interest.
Source: Lauer SA et al. Ann Intern Med. 2020 Mar 9. doi: 10.1101/2020.02.02.20020016.
Is there empathy erosion?
You learned a lot of things in medical school. But there must have been some things that you unlearned on the way to your degree. For instance, you unlearned that you could catch a cold by playing outside on a cold damp day without your jacket. You unlearned that handling a toad would give you warts.
The authors of a recent study suggest that over your 4 years in medical school you also unlearned how to be empathetic (“Does Empathy Decline in the Clinical Phase of Medical Education? A Nationwide, Multi-institutional, Cross-Sectional Study of Students at DO-Granting Medical Schools,” Acad Med. 2020 Jan 21. doi: 10.1097/ACM.0000000000003175). The researchers surveyed more than 10,000 medical students at nearly 50 DO-granting medical schools using standardized questionnaire called the Jefferson Scale of Empathy. They discovered that the students in the clinical phase (years 3 and 4) had lower “empathy scores” than the students in the preclinical phase of their education (years 1 and 2). This decline was statistically significant but “negligible” in magnitude. One wonders why they even chose to publish their results, particularly when the number of respondents to the web-based survey declined with each successive year in medical school. Having looked at the a sample of some of the questions being asked, I can understand why third- and fourth-year students couldn’t be bothered to respond. They were too busy to answer a few dozen “lame” questions.
There may be a decline in empathy over the course our medical training, but I’m not sure that this study can speak to it. An older study found that although medical students scores on a self-administered scale declined between the second and third year, the observed empathetic behavior actually increased. If I had to choose, I would lean more heavily on the results of the behavioral observations.
Certainly, we all changed over the course of our medical education. Including postgraduate training, it may have lasted a decade or more. We saw hundreds of patients, observed life and death on a scale and with an intensity that most of us previously had never experienced. Our perspective changed from being a naive observer to playing the role of an active participant. Did that change include a decline in our capacity for empathy?
Something had to change. We found quickly that we didn’t have the time or emotional energy to learn as much about the person hiding behind every complaint as we once thought we should. We had to cut corners. Sometimes we cut too many. On the other hand, as we saw more patients we may have learned more efficient ways of discovering what we needed to know about them to become an effective and caring physician. If we found ourselves in a specialty in which patients have a high mortality, we were forced to learn ways of protecting ourselves from the emotional damage.
What would you call this process? Was it empathy erosion? Was it a hardening or toughening? Or was it simply maturation? Whatever term you use, it was an obligatory process if we hoped to survive. However, not all of us have done it well. Some of us have narrowed our focus to see only the complaint and the diagnosis, and we too often fail to see the human hiding in plain sight.
For those of us who completed our training with our empathy intact, was this the result of a genetic gift or the atmosphere our parents had created at home? I suspect that in most cases our capacity for empathy as physicians was nurtured and enhanced by the role models we encountered during our training. The mentors we most revered were those who had already been through the annealing process of medical school and specialty training and become even more skilled at caring than when they left college. It is an intangible that can’t be taught. Sadly, there is no way of guaranteeing that everyone who enters medical school will be exposed to or benefit from even one of these master physicians.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
You learned a lot of things in medical school. But there must have been some things that you unlearned on the way to your degree. For instance, you unlearned that you could catch a cold by playing outside on a cold damp day without your jacket. You unlearned that handling a toad would give you warts.
The authors of a recent study suggest that over your 4 years in medical school you also unlearned how to be empathetic (“Does Empathy Decline in the Clinical Phase of Medical Education? A Nationwide, Multi-institutional, Cross-Sectional Study of Students at DO-Granting Medical Schools,” Acad Med. 2020 Jan 21. doi: 10.1097/ACM.0000000000003175). The researchers surveyed more than 10,000 medical students at nearly 50 DO-granting medical schools using standardized questionnaire called the Jefferson Scale of Empathy. They discovered that the students in the clinical phase (years 3 and 4) had lower “empathy scores” than the students in the preclinical phase of their education (years 1 and 2). This decline was statistically significant but “negligible” in magnitude. One wonders why they even chose to publish their results, particularly when the number of respondents to the web-based survey declined with each successive year in medical school. Having looked at the a sample of some of the questions being asked, I can understand why third- and fourth-year students couldn’t be bothered to respond. They were too busy to answer a few dozen “lame” questions.
There may be a decline in empathy over the course our medical training, but I’m not sure that this study can speak to it. An older study found that although medical students scores on a self-administered scale declined between the second and third year, the observed empathetic behavior actually increased. If I had to choose, I would lean more heavily on the results of the behavioral observations.
Certainly, we all changed over the course of our medical education. Including postgraduate training, it may have lasted a decade or more. We saw hundreds of patients, observed life and death on a scale and with an intensity that most of us previously had never experienced. Our perspective changed from being a naive observer to playing the role of an active participant. Did that change include a decline in our capacity for empathy?
Something had to change. We found quickly that we didn’t have the time or emotional energy to learn as much about the person hiding behind every complaint as we once thought we should. We had to cut corners. Sometimes we cut too many. On the other hand, as we saw more patients we may have learned more efficient ways of discovering what we needed to know about them to become an effective and caring physician. If we found ourselves in a specialty in which patients have a high mortality, we were forced to learn ways of protecting ourselves from the emotional damage.
What would you call this process? Was it empathy erosion? Was it a hardening or toughening? Or was it simply maturation? Whatever term you use, it was an obligatory process if we hoped to survive. However, not all of us have done it well. Some of us have narrowed our focus to see only the complaint and the diagnosis, and we too often fail to see the human hiding in plain sight.
For those of us who completed our training with our empathy intact, was this the result of a genetic gift or the atmosphere our parents had created at home? I suspect that in most cases our capacity for empathy as physicians was nurtured and enhanced by the role models we encountered during our training. The mentors we most revered were those who had already been through the annealing process of medical school and specialty training and become even more skilled at caring than when they left college. It is an intangible that can’t be taught. Sadly, there is no way of guaranteeing that everyone who enters medical school will be exposed to or benefit from even one of these master physicians.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
You learned a lot of things in medical school. But there must have been some things that you unlearned on the way to your degree. For instance, you unlearned that you could catch a cold by playing outside on a cold damp day without your jacket. You unlearned that handling a toad would give you warts.
The authors of a recent study suggest that over your 4 years in medical school you also unlearned how to be empathetic (“Does Empathy Decline in the Clinical Phase of Medical Education? A Nationwide, Multi-institutional, Cross-Sectional Study of Students at DO-Granting Medical Schools,” Acad Med. 2020 Jan 21. doi: 10.1097/ACM.0000000000003175). The researchers surveyed more than 10,000 medical students at nearly 50 DO-granting medical schools using standardized questionnaire called the Jefferson Scale of Empathy. They discovered that the students in the clinical phase (years 3 and 4) had lower “empathy scores” than the students in the preclinical phase of their education (years 1 and 2). This decline was statistically significant but “negligible” in magnitude. One wonders why they even chose to publish their results, particularly when the number of respondents to the web-based survey declined with each successive year in medical school. Having looked at the a sample of some of the questions being asked, I can understand why third- and fourth-year students couldn’t be bothered to respond. They were too busy to answer a few dozen “lame” questions.
There may be a decline in empathy over the course our medical training, but I’m not sure that this study can speak to it. An older study found that although medical students scores on a self-administered scale declined between the second and third year, the observed empathetic behavior actually increased. If I had to choose, I would lean more heavily on the results of the behavioral observations.
Certainly, we all changed over the course of our medical education. Including postgraduate training, it may have lasted a decade or more. We saw hundreds of patients, observed life and death on a scale and with an intensity that most of us previously had never experienced. Our perspective changed from being a naive observer to playing the role of an active participant. Did that change include a decline in our capacity for empathy?
Something had to change. We found quickly that we didn’t have the time or emotional energy to learn as much about the person hiding behind every complaint as we once thought we should. We had to cut corners. Sometimes we cut too many. On the other hand, as we saw more patients we may have learned more efficient ways of discovering what we needed to know about them to become an effective and caring physician. If we found ourselves in a specialty in which patients have a high mortality, we were forced to learn ways of protecting ourselves from the emotional damage.
What would you call this process? Was it empathy erosion? Was it a hardening or toughening? Or was it simply maturation? Whatever term you use, it was an obligatory process if we hoped to survive. However, not all of us have done it well. Some of us have narrowed our focus to see only the complaint and the diagnosis, and we too often fail to see the human hiding in plain sight.
For those of us who completed our training with our empathy intact, was this the result of a genetic gift or the atmosphere our parents had created at home? I suspect that in most cases our capacity for empathy as physicians was nurtured and enhanced by the role models we encountered during our training. The mentors we most revered were those who had already been through the annealing process of medical school and specialty training and become even more skilled at caring than when they left college. It is an intangible that can’t be taught. Sadly, there is no way of guaranteeing that everyone who enters medical school will be exposed to or benefit from even one of these master physicians.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
TBI deaths from falls on the rise
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.
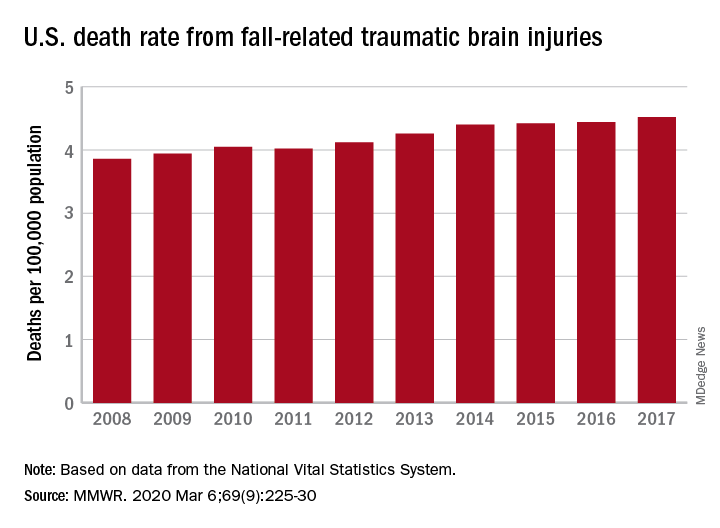
Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.

Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
A 17% surge in mortality from fall-related traumatic brain injuries from 2008 to 2017 was driven largely by increases among those aged 75 years and older, according to investigators from the Centers for Disease Control and Prevention.

Nationally, the rate of deaths from traumatic brain injuries (TBIs) caused by unintentional falls rose from 3.86 per 100,000 population in 2008 to 4.52 per 100,000 in 2017, as the number of deaths went from 12,311 to 17,408, said Alexis B. Peterson, PhD, and Scott R. Kegler, PhD, of the CDC’s National Center for Injury Prevention and Control in Atlanta.
“This increase might be explained by longer survival following the onset of common diseases such as stroke, cancer, and heart disease or be attributable to the increasing population of older adults in the United States,” they suggested in the Mortality and Morbidity Weekly Report.
The rate of fall-related TBI among Americans aged 75 years and older increased by an average of 2.6% per year from 2008 to 2017, compared with 1.8% in those aged 55-74. Over that same time, death rates dropped for those aged 35-44 (–0.3%), 18-34 (–1.1%), and 0-17 (–4.3%), they said, based on data from the National Vital Statistics System’s multiple cause-of-death database.
The death rate increased fastest in residents of rural areas (2.9% per year), but deaths from fall-related TBI were up at all levels of urbanization. The largest central cities and fringe metro areas were up by 1.4% a year, with larger annual increases seen in medium-size cities (2.1%), small cities (2.2%), and small towns (2.1%), Dr. Peterson and Dr. Kegler said.
Rates of TBI-related mortality in general are higher in rural areas, they noted, and “heterogeneity in the availability and accessibility of resources (e.g., access to high-level trauma centers and rehabilitative services) can result in disparities in postinjury outcomes.”
State-specific rates increased in 45 states, although Alaska was excluded from the analysis because of its small number of cases (less than 20). Increases were significant in 29 states, but none of the changes were significant in the 4 states with lower rates at the end of the study period, the investigators reported.
“In older adults, evidence-based fall prevention strategies can prevent falls and avert costly medical expenditures,” Dr. Peterson and Dr. Kegler said, suggesting that health care providers “consider prescribing exercises that incorporate balance, strength and gait activities, such as tai chi, and reviewing and managing medications linked to falls.”
SOURCE: Peterson AB, Kegler SR. MMWR. 2019 Mar 6;69(9):225-30.
FROM MMWR
Stress-related disorders linked to later neurodegenerative diseases
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
Individuals with posttraumatic stress disorder (PTSD), acute stress reaction, adjustment disorder, or other stress reactions had an 80% increased risk of vascular neurodegenerative diseases, according to results of the study, which was based on Swedish population registry data.
Risk of primary neurodegenerative diseases was increased as well in people with those conditions, but only by 31%, according to lead author Huan Song, MD, PhD, of Sichuan University in Chengdu, China.
“The stronger association observed for neurodegenerative diseases with a vascular component, compared with primary neurodegenerative diseases, suggested a considerable role of a possible cerebrovascular pathway,” Dr. Song and coauthors said in a report on the study appearing in JAMA Neurology.
While some previous studies have linked stress-related disorders to neurodegenerative diseases – particularly PTSD and dementia – this is believed to be the first, according to the investigators, to comprehensively evaluate all stress-related disorders in relation to the most common neurodegenerative conditions.
When considering neurodegenerative conditions separately, they found a statistically significant association between stress-related disorders and Alzheimer’s disease, while linkages with Parkinson’s disease and amyotrophic lateral sclerosis (ALS) were “comparable” but associations did not reach statistical significance, according to investigators.
Based on these findings, stress reduction should be recommended in addition to daily physical activity, mental activity, and a heart-healthy diet to potentially reduce risk of onset or worsening of cognitive decline, according to Chun Lim, MD, PhD, medical director of the cognitive neurology unit at Beth Israel Deaconess Medical Center in Boston.
“We don’t really have great evidence that anything slows down the progression of Alzheimer’s disease, but there are some suggestions that for people who lead heart-healthy lifestyles or adhere to a Mediterranean diet, fewer develop cognitive issues over 5-10 years,” Dr. Lim said in an interview. “Because of this paper, stress reduction may be one additional way to hopefully help these patients these patients that have or are concerned about cognitive issues.”
The population-matched cohort of the study included 61,748 individuals with stress-related disorders and 595,335 matched individuals without those disorders, while the sibling-matched cohort included 44,839 individuals with those disorders and 78,482 without. The median age at the start of follow-up was 47 years and 39.4% of those with stress-related disorders were male.
During follow-up, the incidence of neurodegenerative diseases per 1,000 person-years was 1.50 for individuals with stress-related disorders, versus 0.82 for those without stress-related disorders, according to the report. Risk of primary neurodegenerative diseases was increased among those with stress-related disorders, compared with those without, with a hazard ratio of 1.31 (95% confidence interval, 1.15-1.48). However, the risk of vascular neurodegenerative diseases was significantly higher, with an HR of 1.80 (95% CI, 1.40-2.31; P = .03 for the difference between hazard ratios).
Results of the matched sibling cohort supported results of the population-matched cohort, though the elevated risk of vascular neurodegenerative diseases among those with stress-related disorders was “slightly lower” than in the population-based cohort, Dr. Song and coauthors wrote in their report.
Beyond causing a host of hormonal and medical issues, stress can lead to sleep issues that may have long-term consequences, Dr. Lim noted in the interview.
“There’s some thought that quality sleep is important for memory formation, and if people are under a fair amount of stress and they have really poor sleep, that can also lead to cognitive issues including memory impairment,” he said.
“There are these multiple avenues that may be contributing to the accelerated development of these kinds of issues,” he added, “so I think this paper suggests more ways to counsel the patients about using lifestyle modifications to slow down the development of these cognitive impairments.”
Funding for the study came from the Swedish Research Council, Icelandic Research Fund; ,European Research Council the Karolinska Institutet, Swedish Research Council, and West China Hospital. Authors of the study provided disclosures related to those organizations as well as Shire/Takeda and Evolan.
SOURCE: Song H et al. JAMA Neurol. 2020 Mar 9. doi: 10.1001/jamaneurol.2020.0117.
FROM JAMA NEUROLOGY
AUGUSTUS: Apixaban surpassed warfarin despite prior stroke or thromboembolism
LOS ANGELES – The edge that the direct-acting oral anticoagulant apixaban (Eliquis) has over warfarin for safely preventing ischemic events in patients with atrial fibrillation and either a recent acute coronary syndrome event or a recent percutaneous coronary intervention held up even in patients with a history of stroke, transient ischemic attack, or thromboembolic event, according to a prespecified secondary analysis of data collected in the AUGUSTUS trial.
The treatment advantages of apixaban, compared with warfarin, seen in the overall AUGUSTUS results, first reported in March 2019, “were consistent” with the benefits seen in the subgroup of enrolled patients with a prior stroke, transient ischemic attack (TIA), or thromboembolic (TE) event, M. Cecilia Bahit, MD, said at the International Stroke Conference sponsored by the American Heart Association.
All patients in AUGUSTUS received a P2Y12 inhibitor antiplatelet drug, which was clopidogrel for more than 90% of patients. The two-by-two factorial design of AUGUSTUS also assessed the safety and efficacy of either adding or withholding aspirin from the two-drug regimen that all patients in the study received with a P2Y12 inhibitor plus an anticoagulant (apixaban or warfarin). The most notable finding of the aspirin versus placebo analysis was that patients without a prior stroke, TIA, or TE event had a “more profound” increase in their rate of major or clinically relevant minor bleeds when also treated with aspirin, compared with patients who received aspirin and had a history of stroke, TIA, or TE event, reported Dr. Bahit, a chief of cardiology and director of clinical research at the INECO Foundation in Rosario, Argentina.
In general, the findings of the secondary analysis that took into account stroke, TIA, or TE history “confirmed” the main AUGUSTUS findings, Dr. Bahit said; an antithrombotic regimen of apixaban plus clopidogrel (or other P2Y12 inhibitor) without aspirin was superior for both efficacy and safety, compared with the alternative regimens that either substituted warfarin for apixaban or that added aspirin.
AUGUSTUS enrolled 4,614 atrial fibrillation (AFib) patients who either had a recent acute coronary syndrome (ACS) event or had recently undergone percutaneous coronary intervention (PCI) at any of 492 sites in 33 countries during 2015-2018. The study’s primary endpoint was the incidence of major or clinically relevant minor bleeds after 6 months, which was significantly lower in the subgroups that received apixaban instead of warfarin and in patients who received placebo instead of aspirin. The secondary endpoint of death or hospitalization after 6 months was also significantly lower in the apixaban-treated patients, compared with those on warfarin, while the aspirin and placebo subgroups showed no difference in the incidence of these events (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
The results reported by Dr. Bahit also highlighted both the high risk faced by patients with AFib who also have had an ACS event or PCI, as well as a prior stroke, TIA, or TE event, noted Larry B. Goldstein, MD, professor and chairman of neurology at the University of Kentucky, Lexington. “It’s difficult, because these patients had an ACS event or PCI, and you don’t want a coronary too close up, but do these patients really need a P2Y12 inhibitor plus an anticoagulant? Could these patients do as well on apixaban only? I would have liked to see that treatment arm in the study,” Dr. Goldstein commented in an interview.
“These are challenging patients because they often require anticoagulation for the AFib as well as antiplatelet agents” for the recent PCI or ACS event, commented Mitchell S.V. Elkind, MD, professor of neurology at Columbia University, New York. “The question has always been: How many blood thinners should these patients be on? Potentially they could be on three different agents [an anticoagulant and two antiplatelet drugs], and we know that all of those drugs together pretty dramatically increase the risk of bleeding. About 15% of the patients in the overall AUGUSTUS trial had either cerebrovascular disease or systemic thromboembolism, so this was a small subgroup of the overall trial, but the overall trial was large so it’s a significant number of patients who met this criteria. The results confirmed that even in a group of patients who may be considered at high risk because they have a prior history of cerebrovascular disease use of apixaban instead of warfarin seemed safer, and that those patients did not need to be on aspirin as well as their other antiplatelet agent. Patients with a history of stroke, in fact, had a lower risk of bleeding than the other patients in this trial, so one could argue that they should be on an agent like apixaban as well as an antiplatelet agent like clopidogrel without addition of aspirin,” he said in a recorded statement.
In addition to implications for using prescription drugs like apixaban and clopidogrel, the findings also send a message about the need for very aggressive implementation of lifestyle measures that can reduce cardiovascular disease risk in these patients, added Dr. Goldstein. The AUGUSTUS outcome analyses that subdivided the study population into those with a prior stroke, TIA, or TE event – 633 patients or about 14% of the 4,581 patients eligible for this analysis – and those who did not have this history showed the extremely high, incrementally elevated risk faced by patients with these prior events.
A history of stroke, TIA, or TE event linked with a jump in the 90-day rate of major or clinically relevant minor bleeds from 13% without this history to 17%, which is a 31% relative increase; it boosted the 90-day rate of death or hospitalization from 25% to 31%, a 24% relative increase; and it jacked up the rate of death or ischemic events from 6% to 9%, a 50% relative increase, Dr. Bahit reported.
These substantial increases “suggest we need to be very aggressive” in managing these high-risk patients who combine a background of AFib, a prior stroke, TIA, or TE events, and a recent ACS event or PCI, Dr. Goldstein observed. In these patients, he suggested that clinicians make sure to address smoking cessation, obesity, exercise, diet, and statin use, and get each of these to an optimal level to further cut risk. If all five of these basic interventions were successfully administered to a patient they could collectively cut the patient’s event risk by about 80%, he added.
AUGUSTUS was funded by Bristol-Myers Squibb and Pfizer, the companies that jointly market apixaban. Dr. Bahit has received honoraria from Pfizer, and from CSL Behring and Merck. Dr. Elkind and Dr. Goldstein had no relevant disclosures.
SOURCE: Bahit MC et al. ISC 2020, Abstract LB22.
LOS ANGELES – The edge that the direct-acting oral anticoagulant apixaban (Eliquis) has over warfarin for safely preventing ischemic events in patients with atrial fibrillation and either a recent acute coronary syndrome event or a recent percutaneous coronary intervention held up even in patients with a history of stroke, transient ischemic attack, or thromboembolic event, according to a prespecified secondary analysis of data collected in the AUGUSTUS trial.
The treatment advantages of apixaban, compared with warfarin, seen in the overall AUGUSTUS results, first reported in March 2019, “were consistent” with the benefits seen in the subgroup of enrolled patients with a prior stroke, transient ischemic attack (TIA), or thromboembolic (TE) event, M. Cecilia Bahit, MD, said at the International Stroke Conference sponsored by the American Heart Association.
All patients in AUGUSTUS received a P2Y12 inhibitor antiplatelet drug, which was clopidogrel for more than 90% of patients. The two-by-two factorial design of AUGUSTUS also assessed the safety and efficacy of either adding or withholding aspirin from the two-drug regimen that all patients in the study received with a P2Y12 inhibitor plus an anticoagulant (apixaban or warfarin). The most notable finding of the aspirin versus placebo analysis was that patients without a prior stroke, TIA, or TE event had a “more profound” increase in their rate of major or clinically relevant minor bleeds when also treated with aspirin, compared with patients who received aspirin and had a history of stroke, TIA, or TE event, reported Dr. Bahit, a chief of cardiology and director of clinical research at the INECO Foundation in Rosario, Argentina.
In general, the findings of the secondary analysis that took into account stroke, TIA, or TE history “confirmed” the main AUGUSTUS findings, Dr. Bahit said; an antithrombotic regimen of apixaban plus clopidogrel (or other P2Y12 inhibitor) without aspirin was superior for both efficacy and safety, compared with the alternative regimens that either substituted warfarin for apixaban or that added aspirin.
AUGUSTUS enrolled 4,614 atrial fibrillation (AFib) patients who either had a recent acute coronary syndrome (ACS) event or had recently undergone percutaneous coronary intervention (PCI) at any of 492 sites in 33 countries during 2015-2018. The study’s primary endpoint was the incidence of major or clinically relevant minor bleeds after 6 months, which was significantly lower in the subgroups that received apixaban instead of warfarin and in patients who received placebo instead of aspirin. The secondary endpoint of death or hospitalization after 6 months was also significantly lower in the apixaban-treated patients, compared with those on warfarin, while the aspirin and placebo subgroups showed no difference in the incidence of these events (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
The results reported by Dr. Bahit also highlighted both the high risk faced by patients with AFib who also have had an ACS event or PCI, as well as a prior stroke, TIA, or TE event, noted Larry B. Goldstein, MD, professor and chairman of neurology at the University of Kentucky, Lexington. “It’s difficult, because these patients had an ACS event or PCI, and you don’t want a coronary too close up, but do these patients really need a P2Y12 inhibitor plus an anticoagulant? Could these patients do as well on apixaban only? I would have liked to see that treatment arm in the study,” Dr. Goldstein commented in an interview.
“These are challenging patients because they often require anticoagulation for the AFib as well as antiplatelet agents” for the recent PCI or ACS event, commented Mitchell S.V. Elkind, MD, professor of neurology at Columbia University, New York. “The question has always been: How many blood thinners should these patients be on? Potentially they could be on three different agents [an anticoagulant and two antiplatelet drugs], and we know that all of those drugs together pretty dramatically increase the risk of bleeding. About 15% of the patients in the overall AUGUSTUS trial had either cerebrovascular disease or systemic thromboembolism, so this was a small subgroup of the overall trial, but the overall trial was large so it’s a significant number of patients who met this criteria. The results confirmed that even in a group of patients who may be considered at high risk because they have a prior history of cerebrovascular disease use of apixaban instead of warfarin seemed safer, and that those patients did not need to be on aspirin as well as their other antiplatelet agent. Patients with a history of stroke, in fact, had a lower risk of bleeding than the other patients in this trial, so one could argue that they should be on an agent like apixaban as well as an antiplatelet agent like clopidogrel without addition of aspirin,” he said in a recorded statement.
In addition to implications for using prescription drugs like apixaban and clopidogrel, the findings also send a message about the need for very aggressive implementation of lifestyle measures that can reduce cardiovascular disease risk in these patients, added Dr. Goldstein. The AUGUSTUS outcome analyses that subdivided the study population into those with a prior stroke, TIA, or TE event – 633 patients or about 14% of the 4,581 patients eligible for this analysis – and those who did not have this history showed the extremely high, incrementally elevated risk faced by patients with these prior events.
A history of stroke, TIA, or TE event linked with a jump in the 90-day rate of major or clinically relevant minor bleeds from 13% without this history to 17%, which is a 31% relative increase; it boosted the 90-day rate of death or hospitalization from 25% to 31%, a 24% relative increase; and it jacked up the rate of death or ischemic events from 6% to 9%, a 50% relative increase, Dr. Bahit reported.
These substantial increases “suggest we need to be very aggressive” in managing these high-risk patients who combine a background of AFib, a prior stroke, TIA, or TE events, and a recent ACS event or PCI, Dr. Goldstein observed. In these patients, he suggested that clinicians make sure to address smoking cessation, obesity, exercise, diet, and statin use, and get each of these to an optimal level to further cut risk. If all five of these basic interventions were successfully administered to a patient they could collectively cut the patient’s event risk by about 80%, he added.
AUGUSTUS was funded by Bristol-Myers Squibb and Pfizer, the companies that jointly market apixaban. Dr. Bahit has received honoraria from Pfizer, and from CSL Behring and Merck. Dr. Elkind and Dr. Goldstein had no relevant disclosures.
SOURCE: Bahit MC et al. ISC 2020, Abstract LB22.
LOS ANGELES – The edge that the direct-acting oral anticoagulant apixaban (Eliquis) has over warfarin for safely preventing ischemic events in patients with atrial fibrillation and either a recent acute coronary syndrome event or a recent percutaneous coronary intervention held up even in patients with a history of stroke, transient ischemic attack, or thromboembolic event, according to a prespecified secondary analysis of data collected in the AUGUSTUS trial.
The treatment advantages of apixaban, compared with warfarin, seen in the overall AUGUSTUS results, first reported in March 2019, “were consistent” with the benefits seen in the subgroup of enrolled patients with a prior stroke, transient ischemic attack (TIA), or thromboembolic (TE) event, M. Cecilia Bahit, MD, said at the International Stroke Conference sponsored by the American Heart Association.
All patients in AUGUSTUS received a P2Y12 inhibitor antiplatelet drug, which was clopidogrel for more than 90% of patients. The two-by-two factorial design of AUGUSTUS also assessed the safety and efficacy of either adding or withholding aspirin from the two-drug regimen that all patients in the study received with a P2Y12 inhibitor plus an anticoagulant (apixaban or warfarin). The most notable finding of the aspirin versus placebo analysis was that patients without a prior stroke, TIA, or TE event had a “more profound” increase in their rate of major or clinically relevant minor bleeds when also treated with aspirin, compared with patients who received aspirin and had a history of stroke, TIA, or TE event, reported Dr. Bahit, a chief of cardiology and director of clinical research at the INECO Foundation in Rosario, Argentina.
In general, the findings of the secondary analysis that took into account stroke, TIA, or TE history “confirmed” the main AUGUSTUS findings, Dr. Bahit said; an antithrombotic regimen of apixaban plus clopidogrel (or other P2Y12 inhibitor) without aspirin was superior for both efficacy and safety, compared with the alternative regimens that either substituted warfarin for apixaban or that added aspirin.
AUGUSTUS enrolled 4,614 atrial fibrillation (AFib) patients who either had a recent acute coronary syndrome (ACS) event or had recently undergone percutaneous coronary intervention (PCI) at any of 492 sites in 33 countries during 2015-2018. The study’s primary endpoint was the incidence of major or clinically relevant minor bleeds after 6 months, which was significantly lower in the subgroups that received apixaban instead of warfarin and in patients who received placebo instead of aspirin. The secondary endpoint of death or hospitalization after 6 months was also significantly lower in the apixaban-treated patients, compared with those on warfarin, while the aspirin and placebo subgroups showed no difference in the incidence of these events (N Engl J Med. 2019 Apr 18;380[16]:1509-24).
The results reported by Dr. Bahit also highlighted both the high risk faced by patients with AFib who also have had an ACS event or PCI, as well as a prior stroke, TIA, or TE event, noted Larry B. Goldstein, MD, professor and chairman of neurology at the University of Kentucky, Lexington. “It’s difficult, because these patients had an ACS event or PCI, and you don’t want a coronary too close up, but do these patients really need a P2Y12 inhibitor plus an anticoagulant? Could these patients do as well on apixaban only? I would have liked to see that treatment arm in the study,” Dr. Goldstein commented in an interview.
“These are challenging patients because they often require anticoagulation for the AFib as well as antiplatelet agents” for the recent PCI or ACS event, commented Mitchell S.V. Elkind, MD, professor of neurology at Columbia University, New York. “The question has always been: How many blood thinners should these patients be on? Potentially they could be on three different agents [an anticoagulant and two antiplatelet drugs], and we know that all of those drugs together pretty dramatically increase the risk of bleeding. About 15% of the patients in the overall AUGUSTUS trial had either cerebrovascular disease or systemic thromboembolism, so this was a small subgroup of the overall trial, but the overall trial was large so it’s a significant number of patients who met this criteria. The results confirmed that even in a group of patients who may be considered at high risk because they have a prior history of cerebrovascular disease use of apixaban instead of warfarin seemed safer, and that those patients did not need to be on aspirin as well as their other antiplatelet agent. Patients with a history of stroke, in fact, had a lower risk of bleeding than the other patients in this trial, so one could argue that they should be on an agent like apixaban as well as an antiplatelet agent like clopidogrel without addition of aspirin,” he said in a recorded statement.
In addition to implications for using prescription drugs like apixaban and clopidogrel, the findings also send a message about the need for very aggressive implementation of lifestyle measures that can reduce cardiovascular disease risk in these patients, added Dr. Goldstein. The AUGUSTUS outcome analyses that subdivided the study population into those with a prior stroke, TIA, or TE event – 633 patients or about 14% of the 4,581 patients eligible for this analysis – and those who did not have this history showed the extremely high, incrementally elevated risk faced by patients with these prior events.
A history of stroke, TIA, or TE event linked with a jump in the 90-day rate of major or clinically relevant minor bleeds from 13% without this history to 17%, which is a 31% relative increase; it boosted the 90-day rate of death or hospitalization from 25% to 31%, a 24% relative increase; and it jacked up the rate of death or ischemic events from 6% to 9%, a 50% relative increase, Dr. Bahit reported.
These substantial increases “suggest we need to be very aggressive” in managing these high-risk patients who combine a background of AFib, a prior stroke, TIA, or TE events, and a recent ACS event or PCI, Dr. Goldstein observed. In these patients, he suggested that clinicians make sure to address smoking cessation, obesity, exercise, diet, and statin use, and get each of these to an optimal level to further cut risk. If all five of these basic interventions were successfully administered to a patient they could collectively cut the patient’s event risk by about 80%, he added.
AUGUSTUS was funded by Bristol-Myers Squibb and Pfizer, the companies that jointly market apixaban. Dr. Bahit has received honoraria from Pfizer, and from CSL Behring and Merck. Dr. Elkind and Dr. Goldstein had no relevant disclosures.
SOURCE: Bahit MC et al. ISC 2020, Abstract LB22.
REPORTING FROM ISC 2020
Novel coronavirus may cause environmental contamination through fecal shedding
The toilet bowl, sink, and bathroom door handle of an isolation room housing a patient with the novel coronavirus tested positive for the virus, raising the possibility that viral shedding in the stool could represent another route of transmission, investigators reported.
Air outlet fans and other room sites also tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), though an anteroom, a corridor, and most personal protective equipment (PPE) worn by health care providers tested negative, according to the researchers, led by Sean Wei Xiang Ong, MBBS, of the National Centre for Infectious Diseases, Singapore.
Taken together, these findings suggest a “need for strict adherence to environmental and hand hygiene” to combat significant environmental contamination through respiratory droplets and fecal shedding, Dr. Ong and colleagues wrote in JAMA.
Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau in New York, said these results demonstrate that SARS-CoV-2 is “clearly capable” of contaminating bathroom sinks and toilets.
“That wouldn’t have been the first place I would have thought of, before this study,” he said in an interview. “You need to pay attention to cleaning the bathrooms, which we obviously do, but that’s an important reminder.”
The report by Dr. Ong and coauthors included a total of three patients housed in airborne infection isolation rooms in a dedicated SARS-CoV-2 outbreak center in Singapore. For each patient, surface samples were taken from 26 sites in the isolation room, an anteroom, and a bathroom. Samples were also taken from PPE on physicians as they left the patient rooms.
Samples for the first patient, taken right after routine cleaning, were all negative, according to researchers. That room was sampled twice, on days 4 and 10 of the illness, while the patient was still symptomatic. Likewise, for the second patient, postcleaning samples were negative; those samples were taken 2 days after cleaning.
However, for the third patient, samples were taken before routine cleaning. In this case, Dr. Ong and colleagues said 13 of 15 room sites (87%) were positive, including air outlet fans, while 3 of 5 toilet sites (60%) were positive as well, though no contamination was found in the anteroom, corridor, or in air samples.
That patient had two stool samples that were positive for SARS-CoV-2, but no diarrhea, authors said, and had upper respiratory tract involvement without pneumonia.
The fact that swabs of the air exhaust outlets tested positive suggests that virus-laden droplets could be “displaced by airflows” and end up on vents or other equipment, Dr. Ong and coauthors reported.
All PPE samples tested negative, except for the front of one shoe.
“The risk of transmission from contaminated footwear is likely low, as evidenced by negative results in the anteroom and corridor,” they wrote.
While this study included only a small number of patients, Dr. Glatt said the findings represent an important and useful contribution to the literature on coronavirus disease 2019 (COVID-19).
“Every day we’re getting more information, and each little piece of the puzzle helps us in the overall management of individuals with COVID-19,” he said in the interview. “They’re adding to our ability to manage, control, and mitigate further spread of the disease.”
Funding for the study came from the National Medical Research Council in Singapore and DSO National Laboratories. Dr. Ong and colleagues reported no conflicts of interest.
SOURCE: Ong SWX et al. JAMA. 2020 Mar 4. doi: 10.1001/jama.2020.3227.
The toilet bowl, sink, and bathroom door handle of an isolation room housing a patient with the novel coronavirus tested positive for the virus, raising the possibility that viral shedding in the stool could represent another route of transmission, investigators reported.
Air outlet fans and other room sites also tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), though an anteroom, a corridor, and most personal protective equipment (PPE) worn by health care providers tested negative, according to the researchers, led by Sean Wei Xiang Ong, MBBS, of the National Centre for Infectious Diseases, Singapore.
Taken together, these findings suggest a “need for strict adherence to environmental and hand hygiene” to combat significant environmental contamination through respiratory droplets and fecal shedding, Dr. Ong and colleagues wrote in JAMA.
Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau in New York, said these results demonstrate that SARS-CoV-2 is “clearly capable” of contaminating bathroom sinks and toilets.
“That wouldn’t have been the first place I would have thought of, before this study,” he said in an interview. “You need to pay attention to cleaning the bathrooms, which we obviously do, but that’s an important reminder.”
The report by Dr. Ong and coauthors included a total of three patients housed in airborne infection isolation rooms in a dedicated SARS-CoV-2 outbreak center in Singapore. For each patient, surface samples were taken from 26 sites in the isolation room, an anteroom, and a bathroom. Samples were also taken from PPE on physicians as they left the patient rooms.
Samples for the first patient, taken right after routine cleaning, were all negative, according to researchers. That room was sampled twice, on days 4 and 10 of the illness, while the patient was still symptomatic. Likewise, for the second patient, postcleaning samples were negative; those samples were taken 2 days after cleaning.
However, for the third patient, samples were taken before routine cleaning. In this case, Dr. Ong and colleagues said 13 of 15 room sites (87%) were positive, including air outlet fans, while 3 of 5 toilet sites (60%) were positive as well, though no contamination was found in the anteroom, corridor, or in air samples.
That patient had two stool samples that were positive for SARS-CoV-2, but no diarrhea, authors said, and had upper respiratory tract involvement without pneumonia.
The fact that swabs of the air exhaust outlets tested positive suggests that virus-laden droplets could be “displaced by airflows” and end up on vents or other equipment, Dr. Ong and coauthors reported.
All PPE samples tested negative, except for the front of one shoe.
“The risk of transmission from contaminated footwear is likely low, as evidenced by negative results in the anteroom and corridor,” they wrote.
While this study included only a small number of patients, Dr. Glatt said the findings represent an important and useful contribution to the literature on coronavirus disease 2019 (COVID-19).
“Every day we’re getting more information, and each little piece of the puzzle helps us in the overall management of individuals with COVID-19,” he said in the interview. “They’re adding to our ability to manage, control, and mitigate further spread of the disease.”
Funding for the study came from the National Medical Research Council in Singapore and DSO National Laboratories. Dr. Ong and colleagues reported no conflicts of interest.
SOURCE: Ong SWX et al. JAMA. 2020 Mar 4. doi: 10.1001/jama.2020.3227.
The toilet bowl, sink, and bathroom door handle of an isolation room housing a patient with the novel coronavirus tested positive for the virus, raising the possibility that viral shedding in the stool could represent another route of transmission, investigators reported.
Air outlet fans and other room sites also tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), though an anteroom, a corridor, and most personal protective equipment (PPE) worn by health care providers tested negative, according to the researchers, led by Sean Wei Xiang Ong, MBBS, of the National Centre for Infectious Diseases, Singapore.
Taken together, these findings suggest a “need for strict adherence to environmental and hand hygiene” to combat significant environmental contamination through respiratory droplets and fecal shedding, Dr. Ong and colleagues wrote in JAMA.
Aaron Eli Glatt, MD, chair of medicine at Mount Sinai South Nassau in New York, said these results demonstrate that SARS-CoV-2 is “clearly capable” of contaminating bathroom sinks and toilets.
“That wouldn’t have been the first place I would have thought of, before this study,” he said in an interview. “You need to pay attention to cleaning the bathrooms, which we obviously do, but that’s an important reminder.”
The report by Dr. Ong and coauthors included a total of three patients housed in airborne infection isolation rooms in a dedicated SARS-CoV-2 outbreak center in Singapore. For each patient, surface samples were taken from 26 sites in the isolation room, an anteroom, and a bathroom. Samples were also taken from PPE on physicians as they left the patient rooms.
Samples for the first patient, taken right after routine cleaning, were all negative, according to researchers. That room was sampled twice, on days 4 and 10 of the illness, while the patient was still symptomatic. Likewise, for the second patient, postcleaning samples were negative; those samples were taken 2 days after cleaning.
However, for the third patient, samples were taken before routine cleaning. In this case, Dr. Ong and colleagues said 13 of 15 room sites (87%) were positive, including air outlet fans, while 3 of 5 toilet sites (60%) were positive as well, though no contamination was found in the anteroom, corridor, or in air samples.
That patient had two stool samples that were positive for SARS-CoV-2, but no diarrhea, authors said, and had upper respiratory tract involvement without pneumonia.
The fact that swabs of the air exhaust outlets tested positive suggests that virus-laden droplets could be “displaced by airflows” and end up on vents or other equipment, Dr. Ong and coauthors reported.
All PPE samples tested negative, except for the front of one shoe.
“The risk of transmission from contaminated footwear is likely low, as evidenced by negative results in the anteroom and corridor,” they wrote.
While this study included only a small number of patients, Dr. Glatt said the findings represent an important and useful contribution to the literature on coronavirus disease 2019 (COVID-19).
“Every day we’re getting more information, and each little piece of the puzzle helps us in the overall management of individuals with COVID-19,” he said in the interview. “They’re adding to our ability to manage, control, and mitigate further spread of the disease.”
Funding for the study came from the National Medical Research Council in Singapore and DSO National Laboratories. Dr. Ong and colleagues reported no conflicts of interest.
SOURCE: Ong SWX et al. JAMA. 2020 Mar 4. doi: 10.1001/jama.2020.3227.
FROM JAMA