User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Stress, insomnia tied to increased AFib risk for older women
TOPLINE:
Eight psychosocial factors, grouped into two distinct clusters, are significantly associated with risk for atrial fibrillation in postmenopausal women, with insomnia and stressful life events (SLEs) being the most strongly associated with AFib, a large new study has found.
METHODOLOGY:
- In addition to traditional risk factors such as obesity, advanced age, ethnicity, smoking, alcohol, hypertension, diabetes, coronary artery disease, heart failure, and emotional and psychological distress may also affect AFib.
- The study included 83,736 postmenopausal women in the Women’s Health Initiative (mean age, 63.9 years; 88.1% White) who did not have AFib at baseline.
- From questionnaires, researchers collected information on psychosocial stressors and used hierarchical cluster analysis to identify patterns of psychosocial predictors.
- They separated these clusters into quartiles, identified associations between psychosocial exposure variables, and adjusted for traditional risk factors.
- Over an average follow-up of 10.5 years, 23,954 participants (28.6%) developed incident AFib.
TAKEAWAY:
- The analysis generated two clusters of distinct psychosocial variables that were significantly associated with AFib: the Stress Cluster, including SLEs, depressive symptoms, and insomnia; and the Strain Cluster, including three personality traits: optimism, cynical hostility, and emotional expressiveness; and two social measures: social support, and social strain.
- Those in the highest quartiles of both the Stress Cluster and the Strain Cluster had greater rates of AFib, compared with those in the lowest quartiles.
- In a final model, the association between SLEs (hazard ratio, 1.02; 95% confidence interval, 1.01-1.04) and insomnia (HR, 1.04; 95% CI, 1.03-1.06) were most strongly linked to increased incidence of AFib, and a sensitivity analysis using snoring as a surrogate marker for sleep apnea didn’t change this outcome, supporting the independent effect of insomnia on AFib.
- In subgroup analyses, the Stress Cluster had a stronger association with AFib incidence in younger (50-69 years) versus older women (70-79 years), and in non-Hispanic White and Asian women versus Hispanic and non-Hispanic Black women.
IN PRACTICE:
The results support the hypothesis that psychosocial predictors account for additional risk for AFib “above and beyond” traditional risk factors, the authors wrote. Identifying and addressing sex-specific, modifiable risk factors, including insomnia, “may help reduce the burden of AF[ib] in aging women.”
SOURCE:
The study was conducted by Susan X. Zhao, MD, division of cardiology, department of medicine, Santa Clara Valley Medical Center, San Jose, Calif., and colleagues. It was published online in the Journal of the American Heart Association.
LIMITATIONS:
The psychometric questionnaires were administered only at study entry, but psychosocial variables may change over time. Data on sleep apnea and other sleep disorders, which may confound the relationship between insomnia and AFib, were not available, and although the study included a sensitivity analysis controlling for snoring, this is an imperfect surrogate for sleep apnea. Generalizability to other demographic, racial, and ethnic groups is limited.
DISCLOSURES:
The Women’s Health Initiative program is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Eight psychosocial factors, grouped into two distinct clusters, are significantly associated with risk for atrial fibrillation in postmenopausal women, with insomnia and stressful life events (SLEs) being the most strongly associated with AFib, a large new study has found.
METHODOLOGY:
- In addition to traditional risk factors such as obesity, advanced age, ethnicity, smoking, alcohol, hypertension, diabetes, coronary artery disease, heart failure, and emotional and psychological distress may also affect AFib.
- The study included 83,736 postmenopausal women in the Women’s Health Initiative (mean age, 63.9 years; 88.1% White) who did not have AFib at baseline.
- From questionnaires, researchers collected information on psychosocial stressors and used hierarchical cluster analysis to identify patterns of psychosocial predictors.
- They separated these clusters into quartiles, identified associations between psychosocial exposure variables, and adjusted for traditional risk factors.
- Over an average follow-up of 10.5 years, 23,954 participants (28.6%) developed incident AFib.
TAKEAWAY:
- The analysis generated two clusters of distinct psychosocial variables that were significantly associated with AFib: the Stress Cluster, including SLEs, depressive symptoms, and insomnia; and the Strain Cluster, including three personality traits: optimism, cynical hostility, and emotional expressiveness; and two social measures: social support, and social strain.
- Those in the highest quartiles of both the Stress Cluster and the Strain Cluster had greater rates of AFib, compared with those in the lowest quartiles.
- In a final model, the association between SLEs (hazard ratio, 1.02; 95% confidence interval, 1.01-1.04) and insomnia (HR, 1.04; 95% CI, 1.03-1.06) were most strongly linked to increased incidence of AFib, and a sensitivity analysis using snoring as a surrogate marker for sleep apnea didn’t change this outcome, supporting the independent effect of insomnia on AFib.
- In subgroup analyses, the Stress Cluster had a stronger association with AFib incidence in younger (50-69 years) versus older women (70-79 years), and in non-Hispanic White and Asian women versus Hispanic and non-Hispanic Black women.
IN PRACTICE:
The results support the hypothesis that psychosocial predictors account for additional risk for AFib “above and beyond” traditional risk factors, the authors wrote. Identifying and addressing sex-specific, modifiable risk factors, including insomnia, “may help reduce the burden of AF[ib] in aging women.”
SOURCE:
The study was conducted by Susan X. Zhao, MD, division of cardiology, department of medicine, Santa Clara Valley Medical Center, San Jose, Calif., and colleagues. It was published online in the Journal of the American Heart Association.
LIMITATIONS:
The psychometric questionnaires were administered only at study entry, but psychosocial variables may change over time. Data on sleep apnea and other sleep disorders, which may confound the relationship between insomnia and AFib, were not available, and although the study included a sensitivity analysis controlling for snoring, this is an imperfect surrogate for sleep apnea. Generalizability to other demographic, racial, and ethnic groups is limited.
DISCLOSURES:
The Women’s Health Initiative program is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Eight psychosocial factors, grouped into two distinct clusters, are significantly associated with risk for atrial fibrillation in postmenopausal women, with insomnia and stressful life events (SLEs) being the most strongly associated with AFib, a large new study has found.
METHODOLOGY:
- In addition to traditional risk factors such as obesity, advanced age, ethnicity, smoking, alcohol, hypertension, diabetes, coronary artery disease, heart failure, and emotional and psychological distress may also affect AFib.
- The study included 83,736 postmenopausal women in the Women’s Health Initiative (mean age, 63.9 years; 88.1% White) who did not have AFib at baseline.
- From questionnaires, researchers collected information on psychosocial stressors and used hierarchical cluster analysis to identify patterns of psychosocial predictors.
- They separated these clusters into quartiles, identified associations between psychosocial exposure variables, and adjusted for traditional risk factors.
- Over an average follow-up of 10.5 years, 23,954 participants (28.6%) developed incident AFib.
TAKEAWAY:
- The analysis generated two clusters of distinct psychosocial variables that were significantly associated with AFib: the Stress Cluster, including SLEs, depressive symptoms, and insomnia; and the Strain Cluster, including three personality traits: optimism, cynical hostility, and emotional expressiveness; and two social measures: social support, and social strain.
- Those in the highest quartiles of both the Stress Cluster and the Strain Cluster had greater rates of AFib, compared with those in the lowest quartiles.
- In a final model, the association between SLEs (hazard ratio, 1.02; 95% confidence interval, 1.01-1.04) and insomnia (HR, 1.04; 95% CI, 1.03-1.06) were most strongly linked to increased incidence of AFib, and a sensitivity analysis using snoring as a surrogate marker for sleep apnea didn’t change this outcome, supporting the independent effect of insomnia on AFib.
- In subgroup analyses, the Stress Cluster had a stronger association with AFib incidence in younger (50-69 years) versus older women (70-79 years), and in non-Hispanic White and Asian women versus Hispanic and non-Hispanic Black women.
IN PRACTICE:
The results support the hypothesis that psychosocial predictors account for additional risk for AFib “above and beyond” traditional risk factors, the authors wrote. Identifying and addressing sex-specific, modifiable risk factors, including insomnia, “may help reduce the burden of AF[ib] in aging women.”
SOURCE:
The study was conducted by Susan X. Zhao, MD, division of cardiology, department of medicine, Santa Clara Valley Medical Center, San Jose, Calif., and colleagues. It was published online in the Journal of the American Heart Association.
LIMITATIONS:
The psychometric questionnaires were administered only at study entry, but psychosocial variables may change over time. Data on sleep apnea and other sleep disorders, which may confound the relationship between insomnia and AFib, were not available, and although the study included a sensitivity analysis controlling for snoring, this is an imperfect surrogate for sleep apnea. Generalizability to other demographic, racial, and ethnic groups is limited.
DISCLOSURES:
The Women’s Health Initiative program is funded by the National Heart, Lung, and Blood Institute; National Institutes of Health; and the Department of Health & Human Services. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM JOURNAL OF THE AMERICAN HEART ASSOCIATION
Disenfranchised grief: What it looks like, where it goes
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
What happens to grief when those around you don’t understand it? Where does it go? How do you process it?
Disenfranchised grief, when someone or society more generally doesn’t see a loss as worthy of mourning, can deprive people of experiencing or processing their sadness. This grief, which may be triggered by the death of an ex-spouse, a pet, a failed adoption, can be painful and long-lasting.
Suzanne Cole, MD: ‘I didn’t feel the right to grieve’
During the COVID-19 pandemic, my little sister unexpectedly died. Though she was not one of the nearly 7 million people who died of the virus, in 2021 she became another type of statistic: one of the 109,699 people in the United State who died from a drug overdose. Hers was from fentanyl laced with methamphetamines.
Her death unraveled me. I felt deep guilt that I could not pull her from the sweeping current that had wrenched her from mainstream society into the underbelly of sex work and toward the solace of mind-altering drugs.
But I did not feel the right to grieve for her as I have grieved for other loved ones who were not blamed for their exit from this world. My sister was living a sordid life on the fringes of society. My grief felt invalid, undeserved. Yet, in the eyes of other “upstanding citizens,” her life was not as worth grieving – or so I thought. I tucked my sorrow into a small corner of my soul so no one would see, and I carried on.
To this day, the shame I feel robbed me of the ability to freely talk about her or share the searing pain I feel. Tears still prick my eyes when I think of her, but I have become adept at swallowing them, shaking off the waves of grief as though nothing happened. Even now, I cannot shake the pervasive feeling that my silent tears don’t deserve to be wept.
Don S. Dizon, MD: Working through tragedy
As a medical student, I worked with an outpatient physician as part of a third-year rotation. When we met, the first thing that struck me was how disheveled he looked. His clothes were wrinkled, and his pants were baggy. He took cigarette breaks, which I found disturbing.
But I quickly came to admire him. Despite my first impression, he was the type of doctor I aspired to be. He didn’t need to look at a patient’s chart to recall who they were. He just knew them. He greeted patients warmly, asked about their family. He even remembered the special occasions his patients had mentioned since their past visit. He epitomized empathy and connectedness.
Spending one day in clinic brought to light the challenges of forming such bonds with patients. A man came into the cancer clinic reporting chest pain and was triaged to an exam room. Soon after, the patient was found unresponsive on the floor. Nurses were yelling for help, and the doctor ran in and started CPR while minutes ticked by waiting for an ambulance that could take him to the ED.
By the time help arrived, the patient was blue.
He had died in the clinic in the middle of the day, as the waiting room filled. After the body was taken away, the doctor went into the bathroom. About 20 minutes later, he came out, eyes bloodshot, and continued with the rest of his day, ensuring each patient was seen and cared for.
As a medical student, it hit me how hard it must be to see something so tragic like the end of a life and then continue with your day as if nothing had happened. This is an experience of grief I later came to know well after nearly 30 years treating patients with advanced cancers: compartmentalizing it and carrying on.
A space for grieving: The Schwartz Center Rounds
Disenfranchised grief, the grief that is hard to share and often seems wrong to feel in the first place, can be triggered in many situations. Losing a person others don’t believe deserve to be grieved, such as an abusive partner or someone who committed a crime; losing someone you cared for in a professional role; a loss experienced in a breakup or same-sex partnership, if that relationship was not accepted by one’s family; loss from infertility, miscarriage, stillbirth, or failed adoption; loss that may be taboo or stigmatized, such as deaths via suicide or abortion; and loss of a job, home, or possession that you treasure.
Many of us have had similar situations or will, and the feeling that no one understands the need to mourn can be paralyzing and alienating. In the early days, intense, crushing feelings can cause intrusive, distracting thoughts, and over time, that grief can linger and find a permanent place in our minds.
More and more, though, we are being given opportunities to reflect on these sad moments.
The Schwartz Rounds are an example of such an opportunity. In these rounds, we gather to talk about the experience of caring for people, not the science of medicine.
During one particularly powerful rounds, I spoke to my colleagues about my initial meeting with a patient who was very sick. I detailed the experience of telling her children and her at that initial consult how I thought she was dying and that I did not recommend therapy. I remember how they cried. And I remembered how powerless I felt.
As I recalled that memory during Schwartz Rounds, I could not stop from crying. The unfairness of being a physician meeting someone for the first time and having to tell them such bad news overwhelmed me.
Even more poignant, I had the chance to reconnect with this woman’s children, who were present that day, not as audience members but as participants. Their presence may have brought my emotions to the surface more strongly. In that moment, I could show them the feelings I had bottled up for the sake of professionalism. Ultimately, I felt relieved, freer somehow, as if this burden my soul was carrying had been lifted.
Although we are both grateful for forums like this, these opportunities to share and express the grief we may have hidden away are not as common as they should be.
As physicians, we may express grief by shedding tears at the bedside of a patient nearing the end of life or through the anxiety we feel when our patient suffers a severe reaction to treatment. But we tend to put it away, to go on with our day, because there are others to be seen and cared for and more work to be done. Somehow, we move forward, shedding tears in one room and celebrating victories in another.
We need to create more spaces to express and feel grief, so we don’t get lost in it. Because understanding how grief impacts us, as people and as providers, is one of the most important realizations we can make as we go about our time-honored profession as healers.
Dr. Dizon is the director of women’s cancers at Lifespan Cancer Institute, director of medical oncology at Rhode Island Hospital, and a professor of medicine at Brown University, all in Providence. He reported conflicts of interest with Regeneron, AstraZeneca, Clovis, Bristol-Myers Squibb, and Kazia.
A version of this article first appeared on Medscape.com.
Steady VKA therapy beats switch to NOAC in frail AFib patients: FRAIL-AF
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
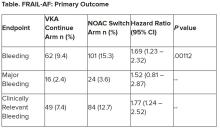
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
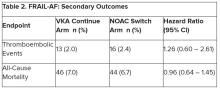
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Switching frail patients with atrial fibrillation (AFib) from anticoagulation therapy with vitamin K antagonists (VKAs) to a novel oral anticoagulant (NOAC) resulted in more bleeding without any reduction in thromboembolic complications or all-cause mortality, randomized trial results show.
The study, FRAIL-AF, is the first randomized NOAC trial to exclusively include frail older patients, said lead author Linda P.T. Joosten, MD, Julius Center for Health Sciences and Primary Care in Utrecht, the Netherlands, and these unexpected findings provide evidence that goes beyond what is currently available.
“Data from the FRAIL-AF trial showed that switching from a VKA to a NOAC should not be considered without a clear indication in frail older patients with AF[ib], as switching to a NOAC leads to 69% more bleeding,” she concluded, without any benefit on secondary clinical endpoints, including thromboembolic events and all-cause mortality.
“The results turned out different than we expected,” Dr. Joosten said. “The hypothesis of this superiority trial was that switching from VKA therapy to a NOAC would result in less bleeding. However, we observed the opposite. After the interim analysis, the data and safety monitoring board advised to stop inclusion because switching from a VKA to a NOAC was clearly contraindicated with a hazard ratio of 1.69 and a highly significant P value of .001.”
Results of FRAIL-AF were presented at the annual congress of the European Society of Cardiology and published online in the journal Circulation.
Session moderator Renate B. Schnabel, MD, interventional cardiologist with University Heart & Vascular Center Hamburg (Germany), congratulated the researchers on these “astonishing” data.
“The thing I want to emphasize here is that, in the absence of randomized controlled trial data, we should be very cautious in extrapolating data from the landmark trials to populations not enrolled in those, and to rely on observational data only,” Dr. Schnabel told Dr. Joosten. “We need randomized controlled trials that sometimes give astonishing results.”
Frailty a clinical syndrome
Frailty is “a lot more than just aging, multiple comorbidities and polypharmacy,” Dr. Joosten explained. “It’s really a clinical syndrome, with people with a high biological vulnerability, dependency on significant others, and a reduced capacity to resist stressors, all leading to a reduced homeostatic reserve.”
Frailty is common in the community, with a prevalence of about 12%, she noted, “and even more important, AF[ib] in frail older people is very common, with a prevalence of 18%. And “without any doubt, we have to adequately anticoagulate frail AF[ib] patients, as they have a high stroke risk, with an incidence of 12.4% per year,” Dr. Joosten noted, compared with 3.9% per year among nonfrail AFib patients.
NOACs are preferred over VKAs in nonfrail AFib patients, after four major trials, RE-LY with dabigatran, ROCKET-AF with rivaroxaban, ARISTOTLE with apixaban, and ENGAGE-AF with edoxaban, showed that NOAC treatment resulted in less major bleeding while stroke risk was comparable with treatment with warfarin, she noted.
The 2023 European Heart Rhythm Association consensus document on management of arrhythmias in frailty syndrome concludes that the advantages of NOACs relative to VKAs are “likely consistent” in frail and nonfrail AFib patients, but the level of evidence is low.
So it’s unknown if NOACs are preferred over VKAs in frail AFib patients, “and it’s even more questionable whether patients on VKAs should switch to NOAC therapy,” Dr. Joosten said.
This new trial aimed to answer the question of whether switching frail AFib patients currently managed on a VKA to a NOAC would reduce bleeding. FRAIL-AF was a pragmatic, multicenter, open-label, randomized, controlled superiority trial.
Older AFib patients were deemed frail if they were aged 75 years or older and had a score of 3 or more on the validated Groningen Frailty Indicator (GFI). Patients with a glomerular filtration rate of less than 30 mL/min per 1.73 m2 or with valvular AFib were excluded.
Eligible patients were then assigned randomly to switch from their international normalized ratio (INR)–guided VKA treatment with either 1 mg acenocoumarol or 3 mg phenprocoumon, to a NOAC, or to continue VKA treatment. They were followed for 12 months for the primary outcome – major bleeding or clinically relevant nonmajor bleeding complication, whichever came first – accounting for death as a competing risk.
A total of 1,330 patients were randomly assigned between January 2018 and June 2022. Their mean age was 83 years, and they had a median GFI of 4. After randomization, 6 patients in the switch-to-NOAC arm, and 1 in the continue-VKA arm were found to have exclusion criteria, so in the end, 662 patients were switched from a VKA to NOAC, while 661 continued on VKA therapy. The choice of NOAC was made by the treating physician.
Major bleeding was defined as a fatal bleeding; bleeding in a critical area or organ; bleeding leading to transfusion; and/or bleeding leading to a fall in hemoglobin level of 2 g/dL (1.24 mmol/L) or more. Nonmajor bleeding was bleeding not considered major but requiring face-to-face consultation, hospitalization or increased level of care, or medical intervention.
After a prespecified futility analysis planned after 163 primary outcome events, the trial was halted when it was seen that there were 101 primary outcome events in the switch arm compared to 62 in the continue arm, Dr. Joosten said. The difference appeared to be driven by clinically relevant nonmajor bleeding.
Secondary outcomes of thromboembolic events and all-cause mortality were similar between the groups.
Completely different patients
Discussant at the meeting for the presentation was Isabelle C. Van Gelder, MD, University Medical Centre Groningen (the Netherlands). She said the results are important and relevant because it “provides data on an important gap of knowledge in our AF[ib] guidelines, and a note for all the cardiologists – this study was not done in the hospital. This trial was done in general practitioner practices, so that’s important to consider.”
Comparing FRAIL-AF patients with those of the four previous NOAC trials, “you see that enormous difference in age,” with an average age of 83 years versus 70-73 years in those trials. “These are completely different patients than have been included previously,” she said.
That GFI score of 4 or more includes patients on four or more different types of medication, as well as memory complaints, an inability to walk around the house, and problems with vision or hearing.
The finding of a 69% increase in bleeding with NOACs in FRAIL-AF was “completely unexpected, and I think that we as cardiologists and as NOAC believers did not expect it at all, but it is as clear as it is.” The curves don’t diverge immediately, but rather after 3 months or thereafter, “so it has nothing to do with the switching process. So why did it occur?”
The Netherlands has dedicated thrombosis services that might improve time in therapeutic range for VKA patients, but there is no real difference in TTRs in FRAIL-AF versus the other NOAC trials, Dr. Van Gelder noted.
The most likely suspect in her view is frailty itself, in particular the tendency for patients to be on a high number of medications. A previous study showed, for example, that polypharmacy could be used as a proxy for the effect of frailty on bleeding risk; patients on 10 or more medications had a higher risk for bleeding on treatment with rivaroxaban versus those on 4 or fewer medications.
“Therefore, in my view, why was there such a high risk of bleeding? It’s because these are other patients than we are normally used to treat, we as cardiologists,” although general practitioners see these patients all the time. “It’s all about frailty.”
NOACs are still relatively new drugs, with possible unknown interactions, she added. Because of their frailty and polypharmacy, these patients may benefit from INR control, Dr. Van Gelder speculated. “Therefore, I agree with them that we should be careful; if such old, frail patients survive on VKA, do not change medications and do not switch!”
The study was supported by the Dutch government with additional and unrestricted educational grants from Boehringer Ingelheim, BMS-Pfizer, Bayer, and Daiichi Sankyo. Dr. Joosten reported no relevant financial relationships. Dr. Van Gelder reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE ESC CONGRESS 2023
‘New dawn’ for aldosterone as drug target in hypertension?
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
Once-daily treatment with the selective aldosterone synthase inhibitor lorundrostat (Mineralys Therapeutics) safely and significantly reduced blood pressure in adults with uncontrolled hypertension in a phase 2, randomized, controlled trial.
Eight weeks after adding lorundrostat (50 mg or 100 mg once daily) or placebo to background therapy, the medication lowered seated automated office systolic BP significantly more than placebo (−9.6 mm Hg with 50 mg; −7.8 mm Hg with 100 mg), with the greatest effects seen in adults with obesity.
“We need new drugs for treatment-resistant hypertension,” study investigator Steven Nissen, MD, chief academic officer at the Heart Vascular & Thoracic Institute at the Cleveland Clinic, said in an interview. Lorundrostat represents a “new class” of antihypertensive that “looks to be safe and we’re seeing very large reductions in blood pressure.”
Results of the Target-HTN trial were published online in JAMA to coincide with presentation at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Aldosterone’s contribution ‘vastly underappreciated’
Excess aldosterone production contributes to uncontrolled BP in patients with obesity and other associated diseases, such as obstructive sleep apnea and metabolic syndrome.
“Aldosterone’s contribution to uncontrolled hypertension is vastly underappreciated,” first author and study presenter Luke Laffin, MD, also with the Cleveland Clinic, said in an interview.
Aldosterone synthase inhibitors are a novel class of BP-lowering medications that decrease aldosterone production. Lorundrostat is one of two such agents in advanced clinical development. The other is baxdrostat (CinCor Pharma/AstraZeneca).
The Target-HTN randomized, placebo-controlled, dose-ranging trial enrolled 200 adults (mean age, 66 years; 60% women) with uncontrolled hypertension while taking two or more antihypertensive medications; 42% of participants were taking three or more antihypertensive medications, 48% were obese and 40% had diabetes.
The study population was divided into two cohorts: an initial cohort of 163 adults with suppressed plasma renin activity at baseline (PRA ≤ 1.0 ng/mL per hour) and elevated plasma aldosterone (≥ 1.0 ng/dL) and a second cohort of 37 adults with PRA greater than 1.0 ng/mL per hour.
Participants were randomly assigned to placebo or one of five doses of lorundrostat in the initial cohort (12.5 mg, 50 mg, or 100 mg once daily or 12.5 mg or 25 mg twice daily).
In the second cohort, participants were randomly assigned (1:6) to placebo or lorundrostat 100 mg once daily. The primary endpoint was change in automated office systolic BP from baseline to week 8.
Among participants with suppressed PRA, following 8 weeks of treatment, changes in office systolic BP of −14.1, −13.2, and −6.9 mm Hg were observed with 100 mg, 50 mg, and 12.5 mg once-daily lorundrostat, respectively, compared with a change of −4.1 mm Hg with placebo.
Reductions in systolic BP in individuals receiving twice-daily doses of 25 mg and 12.5 mg of lorundrostat were −10.1 and −13.8 mm Hg, respectively.
Among participants without suppressed PRA, lorundrostat 100 mg once daily decreased systolic BP by 11.4 mm Hg, similar to BP reduction in those with suppressed PRA receiving the same dose.
A prespecified subgroup analysis showed that participants with obesity demonstrated greater BP lowering in response to lorundrostat.
No instances of cortisol insufficiency occurred. Six participants had increases in serum potassium above 6.0 mEq/L (6.0 mmol/L) that corrected with dose reduction or drug discontinuation.
The increase in serum potassium is “expected and manageable,” Dr. Laffin said in an interview. “Anytime you disrupt aldosterone production, you’re going to have to have an increase in serum potassium, but it’s very manageable and not something that is worrisome.”
A phase 2 trial in 300 adults with uncontrolled hypertension is currently underway. The trial will evaluate the BP-lowering effects of lorundrostat, administered on a background of a standardized antihypertensive medication regimen. A larger phase 3 study will start before the end of the year.
‘New dawn’ for therapies targeting aldosterone
The author of an editorial in JAMA noted that more 70 years after the first isolation of aldosterone, then called electrocortin, “there is a new dawn for therapies targeting aldosterone.”
“There is now real potential to provide better-targeted treatment for patients in whom aldosterone excess is known to contribute to their clinical condition and influence their clinical outcome, notably those with difficult-to-control hypertension, obesity, heart failure, chronic kidney disease, and the many with yet-to-be-diagnosed primary aldosteronism,” said Bryan Williams, MD, University College London.
The trial was funded by Mineralys Therapeutics, which is developing lorundrostat. Dr. Laffin reported that the Cleveland Clinic, his employer, was a study site for the Target-HTN trial and that C5Research, the academic research organization of the Cleveland Clinic, receives payment for services related to other Mineralys clinical trials. Dr. Laffin also reported receipt of personal fees from Medtronic, Lilly, and Crispr Therapeutics, grants from AstraZeneca, and stock options for LucidAct Health and Gordy Health. Dr. Nissen reported receipt of grants from Mineralys during the conduct of the study and grants from AbbVie, AstraZeneca, Amgen, Bristol-Myers Squibb, Lilly, Esperion Therapeutics, Medtronic, grants from MyoKardia, New Amsterdam Pharmaceuticals, Novartis, and Silence Therapeutics. Dr. Williams reported being the unremunerated chair of the steering committee designing a phase 3 trial of the aldosterone synthase inhibitor baxdrostat for AstraZeneca.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION 2023
Is AFib ablation the fifth pillar in heart failure care? CASTLE-HTx
Recorded Aug. 28, 2023. This transcript has been edited for clarity.
John M. Mandrola, MD: I’m here at the European Society of Cardiology meeting, and I’m very excited to have two colleagues whom I met at the Western Atrial Fibrillation Symposium (Western AFib) and who presented the CASTLE-HTx study. This is Christian Sohns and Philipp Sommer, and the CASTLE-HTx study is very exciting.
Before I get into that, I really want to introduce the concept of atrial fibrillation in heart failure. I like to say that there are two big populations of patients with atrial fibrillation, and the vast majority can be treated slowly with reassurance and education. There is a group of patients who have heart failure who, when they develop atrial fibrillation, can degenerate rapidly. The CASTLE-HTx study looked at catheter ablation versus medical therapy in patients with advanced heart failure.
Christian, why don’t you tell us the top-line results and what you found.
CASTLE-HTx key findings
Christian Sohns, MD, PhD: Thanks, first of all, for mentioning this special cohort of patients in end-stage heart failure, which is very important. The endpoint of the study was a composite of death from any cause or left ventricular assist device (LVAD) implantation and heart transplantation. These are very hard, strong clinical endpoints, not the rate of rehospitalization or something like that.
Catheter ablation was superior to medical therapy alone in terms of this composite endpoint. That was driven by cardiovascular death and all-cause mortality, which highlights the fact that you should always consider atrial fibrillation ablation in the end-stage heart failure cohort. The findings were driven by the fact that we saw left ventricular reverse remodeling and the reduction of atrial fibrillation in these patients.
Dr. Mandrola: Tell me about how it came about. It was conducted at your center. Who were these patients?
Philipp Sommer, MD: As one of the biggest centers for heart transplantations all over Europe, with roughly 100 transplants per year, we had many patients being referred to our center with the questions of whether those patients are eligible for a heart transplantation. Not all of the patients in our study were listed for a transplant, but all of them were admitted in that end-stage heart failure status to evaluate their eligibility for transplant.
If we look at the baseline data of those patients, they had an ejection fraction of 29%. They had a 6-minute walk test as a functional capacity parameter of around 300 m. Approximately two thirds of them were New York Heart Association class III and IV, which is significantly worse than what we saw in the previous studies dealing with heart failure patients.
I think overall, if you also look at NT-proBNP levels, this is a really sick patient population where some people might doubt if they should admit and refer those patients for an ablation procedure. Therefore, it’s really interesting and fascinating to see the results.
Dr. Mandrola: I did read in the manuscript, and I heard from you, that these were recruited as outpatients. So they were stable outpatients who were referred to the center for consideration of an LVAD or transplant?
Dr. Sohns: The definition of stability is very difficult in these patients because they have hospital stays, they have a history of drug therapy, and they have a history of interventions also behind them – not atrial fibrillation ablation, but others. I think these patients are referred because the referring physicians are done with the case. They can no longer offer any option to the patients other than surgical treatment, assist device, pump implantation, or transplantation.
If you look at the guidelines, they do not comment on atrial fibrillation ablation in this cohort of patients. Also, they have different recommendations between the American societies and the European societies regarding what is end-stage heart failure and how to treat these patients. Therefore, it was a big benefit of CASTLE-HTx that we randomized a cohort of patients with advanced end-stage heart failure.
How can AFib ablation have such big, early effects?
Dr. Mandrola: These are very clinically significant findings, with large effect sizes and very early separation of the Kaplan-Meier curves. How do you explain how dramatic an effect that is, and how early of an effect?
Dr. Sommer: That’s one of the key questions at the end of the day. I think our job basically was to provide the data and to ensure that the data are clean and that it’s all perfectly done. The interpretation of these data is really kind of difficult, although we do not have the 100% perfect and obvious explanation why the curves separated so early. Our view on that is that we are talking about a pretty fragile patient population, so little differences like having a tachyarrhythmia of 110 day in, day out or being in sinus rhythm of 60 can make a huge difference. That’s obviously pretty early.
The one that remains in tachyarrhythmia will deteriorate and will require an LVAD after a couple of months, and the one that you may keep in sinus rhythm, even with reduced atrial fibrillation burden – not zero, but reduced atrial fibrillation burden – and improved LV function, all of a sudden this patient will still remain on a low level of being stable, but he or she will remain stable and will not require any surgical interventions for the next 1.5-2 years. If we can manage to do this, just postponing the natural cause of the disease, I think that is a great benefit for the patient.
Dr. Mandrola: One of the things that comes up in our center is that I look at some of these patients and think, there’s no way I can put this patient under general anesthetic and do all of this. Your ablation procedure wasn’t that extensive, was it?
Dr. Sohns: On the one hand, no. On the other hand, yes. You need to take into consideration that it has been performed by experienced physicians with experience in heart failure treatment and atrial fibrillation in heart transplantation centers, though it›s not sure that we can transfer these results one-to-one to all other centers in the world.
It is very clear that we have almost no major complications in these patients. We were able to do these ablation procedures without general anesthesia. We have 60% of patients who had pulmonary vein isolation only and 40% of patients who have PVI and additional therapy. We have a procedure duration of almost 90 minutes during radiofrequency ablation.
We have different categories. When you talk about the different patient cohorts, we also see different stages of myocardial tissue damage, which will be part of another publication for sure. It is, in part, surprising how normal some of the atria were despite having a volume of 180 mL, but they had no fibrosis. That was very interesting.
Dr. Mandrola: How did the persistent vs paroxysmal atrial fibrillation sort out? Were these mostly patients with persistent atrial fibrillation?
Dr. Sommer: Two-thirds were persistent. It would be expected in this patient population that you would not find so many paroxysmal cases. I think it›s very important what Christian was just mentioning that when we discussed the trial design, we were anticipating problems with the sedation, for example. With the follow-up of those procedures, would they decompensate because of the fluid that you have to deliver during such a procedure.
We were quite surprised at the end of the day that the procedures were quite straightforward. Fortunately, we had no major complications. I think there were four complications in the 100 ablated patients. I think we were really positive about how the procedures turned out.
I should mention that one of the exclusion criteria was a left atrial diameter of about 60 mm. The huge ones may be very diseased, and maybe the hopeless ones were excluded from the study. Below 60 mm, we did the ablation.
Rhythm control
Dr. Mandrola: One of my colleagues, who is even more skeptical than me, wanted me to ask you, why wouldn’t you take a patient with persistent atrial fibrillation who had heart failure and just cardiovert and use amiodarone and try and maintain sinus rhythm that way?
Dr. Sohns: It is important to mention that 50% of the patients have already had amiodarone before they were randomized and enrolled for the trial. It might bring you a couple of minutes or a couple of hours [of relief], but the patients would get recurrence.
It was very interesting also, and this is in line with the data from Jason Andrade, who demonstrated that we were able to reduce the percentage of patients with persistent atrial fibrillation to paroxysmal. We did a down-staging of the underlying disease. This is not possible with cardioversion or drugs, for example.
Dr. Sommer: What I really like about that question and that comment is the idea that rhythm control in this subset of patients obviously has a role and an importance. It may be a cardioversion initially, giving amiodarone if they didn’t have that before, and you can keep the patient in sinus rhythm with this therapy, I think we’re reaching the same goal.
I think the critical point to get into the mind of physicians who treat heart failure is that sinus rhythm is beneficial, however you get there. Ablation, of course, as in other studies, is the most powerful tool to get there. Cardioversion can be a really good thing to do; you just have to think about it and consider it.
Dr. Mandrola: I do want to say to everybody that there is a tension sometimes between the heart failure community and the electrophysiology community. I think the ideal situation is that we work together, because I think that we can help with the maintenance of sinus rhythm. The control group mortality at 1 year was 20%, and I’ve heard people say that that’s not advanced heart failure. Advanced heart failure patients have much higher mortality than that. My colleague who is a heart failure specialist was criticizing a selection bias in picking the best patients. How would you answer that?
Dr. Sohns: There are data available from Eurotransplant, for example, that the waiting list mortality is 18%, so I think we are almost in line with this 20% mortality in this conservative group. You cannot generalize it. All these patients have different histories. We have 60% dilated cardiomyopathy and 40% ischemic cardiomyopathy. I think it is a very representative group in contrast to your friend who suggests that it is not.
Dr. Sommer: What I like about the discussion is that some approach us to say that the mortality in the control group is much too high – like, what are you doing with those patients that you create so many endpoints? Then others say that it’s not high enough because that is not end-stage heart failure. Come on! We have a patient cohort that is very well described and very well characterized.
If the label is end-stage heart failure, advanced heart failure, or whatever, they are sicker than the patients that we had in earlier trials. The patients that we treated were mostly excluded from all other trials. We opened the door. We found a clear result. I think everyone can see whatever you like to see.
Dr. Mandrola: What would your take-home message be after having done this trial design, the trial was conducted in your single center, and you come up with these amazing results? What would your message be to the whole community?
Dr. Sohns: Taking into consideration how severely sick these patients are, I can just repeat it: They are one step away from death, more or less, or from surgical intervention that can prolong their life. You should also consider that there are options like atrial fibrillation ablation that can buy time, postpone the natural course, or even in some patients replace the destination therapy. Therefore, in my opinion the next guidelines should recommend that every patient should carefully be checked for sinus rhythm before bringing these patients into the environment of transplantation.
Dr. Sommer: My interpretation is that we have to try to bring into physicians’ minds that besides a well-established and well-documented effect of drug therapy with the fabulous four, we may now have the fabulous five, including an ablation option for patients with atrial fibrillation.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. Dr. Sohns is deputy director of the Heart and Diabetes Center NRW, Ruhr University Bochum, Bad Oeynhausen, Germany. Dr. Sommer is professor of cardiology at the Heart and Diabetes Center NRW. Dr. Mandrola reported no conflicts of interest. Dr. Sohns reported receiving research funding from Else Kröner–Fresenius–Stiftung. Dr. Sommer reported consulting with Abbott, Biosense Webster, Boston Scientific, and Medtronic USA.
A version of this article first appeared on Medscape.com.
Recorded Aug. 28, 2023. This transcript has been edited for clarity.
John M. Mandrola, MD: I’m here at the European Society of Cardiology meeting, and I’m very excited to have two colleagues whom I met at the Western Atrial Fibrillation Symposium (Western AFib) and who presented the CASTLE-HTx study. This is Christian Sohns and Philipp Sommer, and the CASTLE-HTx study is very exciting.
Before I get into that, I really want to introduce the concept of atrial fibrillation in heart failure. I like to say that there are two big populations of patients with atrial fibrillation, and the vast majority can be treated slowly with reassurance and education. There is a group of patients who have heart failure who, when they develop atrial fibrillation, can degenerate rapidly. The CASTLE-HTx study looked at catheter ablation versus medical therapy in patients with advanced heart failure.
Christian, why don’t you tell us the top-line results and what you found.
CASTLE-HTx key findings
Christian Sohns, MD, PhD: Thanks, first of all, for mentioning this special cohort of patients in end-stage heart failure, which is very important. The endpoint of the study was a composite of death from any cause or left ventricular assist device (LVAD) implantation and heart transplantation. These are very hard, strong clinical endpoints, not the rate of rehospitalization or something like that.
Catheter ablation was superior to medical therapy alone in terms of this composite endpoint. That was driven by cardiovascular death and all-cause mortality, which highlights the fact that you should always consider atrial fibrillation ablation in the end-stage heart failure cohort. The findings were driven by the fact that we saw left ventricular reverse remodeling and the reduction of atrial fibrillation in these patients.
Dr. Mandrola: Tell me about how it came about. It was conducted at your center. Who were these patients?
Philipp Sommer, MD: As one of the biggest centers for heart transplantations all over Europe, with roughly 100 transplants per year, we had many patients being referred to our center with the questions of whether those patients are eligible for a heart transplantation. Not all of the patients in our study were listed for a transplant, but all of them were admitted in that end-stage heart failure status to evaluate their eligibility for transplant.
If we look at the baseline data of those patients, they had an ejection fraction of 29%. They had a 6-minute walk test as a functional capacity parameter of around 300 m. Approximately two thirds of them were New York Heart Association class III and IV, which is significantly worse than what we saw in the previous studies dealing with heart failure patients.
I think overall, if you also look at NT-proBNP levels, this is a really sick patient population where some people might doubt if they should admit and refer those patients for an ablation procedure. Therefore, it’s really interesting and fascinating to see the results.
Dr. Mandrola: I did read in the manuscript, and I heard from you, that these were recruited as outpatients. So they were stable outpatients who were referred to the center for consideration of an LVAD or transplant?
Dr. Sohns: The definition of stability is very difficult in these patients because they have hospital stays, they have a history of drug therapy, and they have a history of interventions also behind them – not atrial fibrillation ablation, but others. I think these patients are referred because the referring physicians are done with the case. They can no longer offer any option to the patients other than surgical treatment, assist device, pump implantation, or transplantation.
If you look at the guidelines, they do not comment on atrial fibrillation ablation in this cohort of patients. Also, they have different recommendations between the American societies and the European societies regarding what is end-stage heart failure and how to treat these patients. Therefore, it was a big benefit of CASTLE-HTx that we randomized a cohort of patients with advanced end-stage heart failure.
How can AFib ablation have such big, early effects?
Dr. Mandrola: These are very clinically significant findings, with large effect sizes and very early separation of the Kaplan-Meier curves. How do you explain how dramatic an effect that is, and how early of an effect?
Dr. Sommer: That’s one of the key questions at the end of the day. I think our job basically was to provide the data and to ensure that the data are clean and that it’s all perfectly done. The interpretation of these data is really kind of difficult, although we do not have the 100% perfect and obvious explanation why the curves separated so early. Our view on that is that we are talking about a pretty fragile patient population, so little differences like having a tachyarrhythmia of 110 day in, day out or being in sinus rhythm of 60 can make a huge difference. That’s obviously pretty early.
The one that remains in tachyarrhythmia will deteriorate and will require an LVAD after a couple of months, and the one that you may keep in sinus rhythm, even with reduced atrial fibrillation burden – not zero, but reduced atrial fibrillation burden – and improved LV function, all of a sudden this patient will still remain on a low level of being stable, but he or she will remain stable and will not require any surgical interventions for the next 1.5-2 years. If we can manage to do this, just postponing the natural cause of the disease, I think that is a great benefit for the patient.
Dr. Mandrola: One of the things that comes up in our center is that I look at some of these patients and think, there’s no way I can put this patient under general anesthetic and do all of this. Your ablation procedure wasn’t that extensive, was it?
Dr. Sohns: On the one hand, no. On the other hand, yes. You need to take into consideration that it has been performed by experienced physicians with experience in heart failure treatment and atrial fibrillation in heart transplantation centers, though it›s not sure that we can transfer these results one-to-one to all other centers in the world.
It is very clear that we have almost no major complications in these patients. We were able to do these ablation procedures without general anesthesia. We have 60% of patients who had pulmonary vein isolation only and 40% of patients who have PVI and additional therapy. We have a procedure duration of almost 90 minutes during radiofrequency ablation.
We have different categories. When you talk about the different patient cohorts, we also see different stages of myocardial tissue damage, which will be part of another publication for sure. It is, in part, surprising how normal some of the atria were despite having a volume of 180 mL, but they had no fibrosis. That was very interesting.
Dr. Mandrola: How did the persistent vs paroxysmal atrial fibrillation sort out? Were these mostly patients with persistent atrial fibrillation?
Dr. Sommer: Two-thirds were persistent. It would be expected in this patient population that you would not find so many paroxysmal cases. I think it›s very important what Christian was just mentioning that when we discussed the trial design, we were anticipating problems with the sedation, for example. With the follow-up of those procedures, would they decompensate because of the fluid that you have to deliver during such a procedure.
We were quite surprised at the end of the day that the procedures were quite straightforward. Fortunately, we had no major complications. I think there were four complications in the 100 ablated patients. I think we were really positive about how the procedures turned out.
I should mention that one of the exclusion criteria was a left atrial diameter of about 60 mm. The huge ones may be very diseased, and maybe the hopeless ones were excluded from the study. Below 60 mm, we did the ablation.
Rhythm control
Dr. Mandrola: One of my colleagues, who is even more skeptical than me, wanted me to ask you, why wouldn’t you take a patient with persistent atrial fibrillation who had heart failure and just cardiovert and use amiodarone and try and maintain sinus rhythm that way?
Dr. Sohns: It is important to mention that 50% of the patients have already had amiodarone before they were randomized and enrolled for the trial. It might bring you a couple of minutes or a couple of hours [of relief], but the patients would get recurrence.
It was very interesting also, and this is in line with the data from Jason Andrade, who demonstrated that we were able to reduce the percentage of patients with persistent atrial fibrillation to paroxysmal. We did a down-staging of the underlying disease. This is not possible with cardioversion or drugs, for example.
Dr. Sommer: What I really like about that question and that comment is the idea that rhythm control in this subset of patients obviously has a role and an importance. It may be a cardioversion initially, giving amiodarone if they didn’t have that before, and you can keep the patient in sinus rhythm with this therapy, I think we’re reaching the same goal.
I think the critical point to get into the mind of physicians who treat heart failure is that sinus rhythm is beneficial, however you get there. Ablation, of course, as in other studies, is the most powerful tool to get there. Cardioversion can be a really good thing to do; you just have to think about it and consider it.
Dr. Mandrola: I do want to say to everybody that there is a tension sometimes between the heart failure community and the electrophysiology community. I think the ideal situation is that we work together, because I think that we can help with the maintenance of sinus rhythm. The control group mortality at 1 year was 20%, and I’ve heard people say that that’s not advanced heart failure. Advanced heart failure patients have much higher mortality than that. My colleague who is a heart failure specialist was criticizing a selection bias in picking the best patients. How would you answer that?
Dr. Sohns: There are data available from Eurotransplant, for example, that the waiting list mortality is 18%, so I think we are almost in line with this 20% mortality in this conservative group. You cannot generalize it. All these patients have different histories. We have 60% dilated cardiomyopathy and 40% ischemic cardiomyopathy. I think it is a very representative group in contrast to your friend who suggests that it is not.
Dr. Sommer: What I like about the discussion is that some approach us to say that the mortality in the control group is much too high – like, what are you doing with those patients that you create so many endpoints? Then others say that it’s not high enough because that is not end-stage heart failure. Come on! We have a patient cohort that is very well described and very well characterized.
If the label is end-stage heart failure, advanced heart failure, or whatever, they are sicker than the patients that we had in earlier trials. The patients that we treated were mostly excluded from all other trials. We opened the door. We found a clear result. I think everyone can see whatever you like to see.
Dr. Mandrola: What would your take-home message be after having done this trial design, the trial was conducted in your single center, and you come up with these amazing results? What would your message be to the whole community?
Dr. Sohns: Taking into consideration how severely sick these patients are, I can just repeat it: They are one step away from death, more or less, or from surgical intervention that can prolong their life. You should also consider that there are options like atrial fibrillation ablation that can buy time, postpone the natural course, or even in some patients replace the destination therapy. Therefore, in my opinion the next guidelines should recommend that every patient should carefully be checked for sinus rhythm before bringing these patients into the environment of transplantation.
Dr. Sommer: My interpretation is that we have to try to bring into physicians’ minds that besides a well-established and well-documented effect of drug therapy with the fabulous four, we may now have the fabulous five, including an ablation option for patients with atrial fibrillation.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. Dr. Sohns is deputy director of the Heart and Diabetes Center NRW, Ruhr University Bochum, Bad Oeynhausen, Germany. Dr. Sommer is professor of cardiology at the Heart and Diabetes Center NRW. Dr. Mandrola reported no conflicts of interest. Dr. Sohns reported receiving research funding from Else Kröner–Fresenius–Stiftung. Dr. Sommer reported consulting with Abbott, Biosense Webster, Boston Scientific, and Medtronic USA.
A version of this article first appeared on Medscape.com.
Recorded Aug. 28, 2023. This transcript has been edited for clarity.
John M. Mandrola, MD: I’m here at the European Society of Cardiology meeting, and I’m very excited to have two colleagues whom I met at the Western Atrial Fibrillation Symposium (Western AFib) and who presented the CASTLE-HTx study. This is Christian Sohns and Philipp Sommer, and the CASTLE-HTx study is very exciting.
Before I get into that, I really want to introduce the concept of atrial fibrillation in heart failure. I like to say that there are two big populations of patients with atrial fibrillation, and the vast majority can be treated slowly with reassurance and education. There is a group of patients who have heart failure who, when they develop atrial fibrillation, can degenerate rapidly. The CASTLE-HTx study looked at catheter ablation versus medical therapy in patients with advanced heart failure.
Christian, why don’t you tell us the top-line results and what you found.
CASTLE-HTx key findings
Christian Sohns, MD, PhD: Thanks, first of all, for mentioning this special cohort of patients in end-stage heart failure, which is very important. The endpoint of the study was a composite of death from any cause or left ventricular assist device (LVAD) implantation and heart transplantation. These are very hard, strong clinical endpoints, not the rate of rehospitalization or something like that.
Catheter ablation was superior to medical therapy alone in terms of this composite endpoint. That was driven by cardiovascular death and all-cause mortality, which highlights the fact that you should always consider atrial fibrillation ablation in the end-stage heart failure cohort. The findings were driven by the fact that we saw left ventricular reverse remodeling and the reduction of atrial fibrillation in these patients.
Dr. Mandrola: Tell me about how it came about. It was conducted at your center. Who were these patients?
Philipp Sommer, MD: As one of the biggest centers for heart transplantations all over Europe, with roughly 100 transplants per year, we had many patients being referred to our center with the questions of whether those patients are eligible for a heart transplantation. Not all of the patients in our study were listed for a transplant, but all of them were admitted in that end-stage heart failure status to evaluate their eligibility for transplant.
If we look at the baseline data of those patients, they had an ejection fraction of 29%. They had a 6-minute walk test as a functional capacity parameter of around 300 m. Approximately two thirds of them were New York Heart Association class III and IV, which is significantly worse than what we saw in the previous studies dealing with heart failure patients.
I think overall, if you also look at NT-proBNP levels, this is a really sick patient population where some people might doubt if they should admit and refer those patients for an ablation procedure. Therefore, it’s really interesting and fascinating to see the results.
Dr. Mandrola: I did read in the manuscript, and I heard from you, that these were recruited as outpatients. So they were stable outpatients who were referred to the center for consideration of an LVAD or transplant?
Dr. Sohns: The definition of stability is very difficult in these patients because they have hospital stays, they have a history of drug therapy, and they have a history of interventions also behind them – not atrial fibrillation ablation, but others. I think these patients are referred because the referring physicians are done with the case. They can no longer offer any option to the patients other than surgical treatment, assist device, pump implantation, or transplantation.
If you look at the guidelines, they do not comment on atrial fibrillation ablation in this cohort of patients. Also, they have different recommendations between the American societies and the European societies regarding what is end-stage heart failure and how to treat these patients. Therefore, it was a big benefit of CASTLE-HTx that we randomized a cohort of patients with advanced end-stage heart failure.
How can AFib ablation have such big, early effects?
Dr. Mandrola: These are very clinically significant findings, with large effect sizes and very early separation of the Kaplan-Meier curves. How do you explain how dramatic an effect that is, and how early of an effect?
Dr. Sommer: That’s one of the key questions at the end of the day. I think our job basically was to provide the data and to ensure that the data are clean and that it’s all perfectly done. The interpretation of these data is really kind of difficult, although we do not have the 100% perfect and obvious explanation why the curves separated so early. Our view on that is that we are talking about a pretty fragile patient population, so little differences like having a tachyarrhythmia of 110 day in, day out or being in sinus rhythm of 60 can make a huge difference. That’s obviously pretty early.
The one that remains in tachyarrhythmia will deteriorate and will require an LVAD after a couple of months, and the one that you may keep in sinus rhythm, even with reduced atrial fibrillation burden – not zero, but reduced atrial fibrillation burden – and improved LV function, all of a sudden this patient will still remain on a low level of being stable, but he or she will remain stable and will not require any surgical interventions for the next 1.5-2 years. If we can manage to do this, just postponing the natural cause of the disease, I think that is a great benefit for the patient.
Dr. Mandrola: One of the things that comes up in our center is that I look at some of these patients and think, there’s no way I can put this patient under general anesthetic and do all of this. Your ablation procedure wasn’t that extensive, was it?
Dr. Sohns: On the one hand, no. On the other hand, yes. You need to take into consideration that it has been performed by experienced physicians with experience in heart failure treatment and atrial fibrillation in heart transplantation centers, though it›s not sure that we can transfer these results one-to-one to all other centers in the world.
It is very clear that we have almost no major complications in these patients. We were able to do these ablation procedures without general anesthesia. We have 60% of patients who had pulmonary vein isolation only and 40% of patients who have PVI and additional therapy. We have a procedure duration of almost 90 minutes during radiofrequency ablation.
We have different categories. When you talk about the different patient cohorts, we also see different stages of myocardial tissue damage, which will be part of another publication for sure. It is, in part, surprising how normal some of the atria were despite having a volume of 180 mL, but they had no fibrosis. That was very interesting.
Dr. Mandrola: How did the persistent vs paroxysmal atrial fibrillation sort out? Were these mostly patients with persistent atrial fibrillation?
Dr. Sommer: Two-thirds were persistent. It would be expected in this patient population that you would not find so many paroxysmal cases. I think it›s very important what Christian was just mentioning that when we discussed the trial design, we were anticipating problems with the sedation, for example. With the follow-up of those procedures, would they decompensate because of the fluid that you have to deliver during such a procedure.
We were quite surprised at the end of the day that the procedures were quite straightforward. Fortunately, we had no major complications. I think there were four complications in the 100 ablated patients. I think we were really positive about how the procedures turned out.
I should mention that one of the exclusion criteria was a left atrial diameter of about 60 mm. The huge ones may be very diseased, and maybe the hopeless ones were excluded from the study. Below 60 mm, we did the ablation.
Rhythm control
Dr. Mandrola: One of my colleagues, who is even more skeptical than me, wanted me to ask you, why wouldn’t you take a patient with persistent atrial fibrillation who had heart failure and just cardiovert and use amiodarone and try and maintain sinus rhythm that way?
Dr. Sohns: It is important to mention that 50% of the patients have already had amiodarone before they were randomized and enrolled for the trial. It might bring you a couple of minutes or a couple of hours [of relief], but the patients would get recurrence.
It was very interesting also, and this is in line with the data from Jason Andrade, who demonstrated that we were able to reduce the percentage of patients with persistent atrial fibrillation to paroxysmal. We did a down-staging of the underlying disease. This is not possible with cardioversion or drugs, for example.
Dr. Sommer: What I really like about that question and that comment is the idea that rhythm control in this subset of patients obviously has a role and an importance. It may be a cardioversion initially, giving amiodarone if they didn’t have that before, and you can keep the patient in sinus rhythm with this therapy, I think we’re reaching the same goal.
I think the critical point to get into the mind of physicians who treat heart failure is that sinus rhythm is beneficial, however you get there. Ablation, of course, as in other studies, is the most powerful tool to get there. Cardioversion can be a really good thing to do; you just have to think about it and consider it.
Dr. Mandrola: I do want to say to everybody that there is a tension sometimes between the heart failure community and the electrophysiology community. I think the ideal situation is that we work together, because I think that we can help with the maintenance of sinus rhythm. The control group mortality at 1 year was 20%, and I’ve heard people say that that’s not advanced heart failure. Advanced heart failure patients have much higher mortality than that. My colleague who is a heart failure specialist was criticizing a selection bias in picking the best patients. How would you answer that?
Dr. Sohns: There are data available from Eurotransplant, for example, that the waiting list mortality is 18%, so I think we are almost in line with this 20% mortality in this conservative group. You cannot generalize it. All these patients have different histories. We have 60% dilated cardiomyopathy and 40% ischemic cardiomyopathy. I think it is a very representative group in contrast to your friend who suggests that it is not.
Dr. Sommer: What I like about the discussion is that some approach us to say that the mortality in the control group is much too high – like, what are you doing with those patients that you create so many endpoints? Then others say that it’s not high enough because that is not end-stage heart failure. Come on! We have a patient cohort that is very well described and very well characterized.
If the label is end-stage heart failure, advanced heart failure, or whatever, they are sicker than the patients that we had in earlier trials. The patients that we treated were mostly excluded from all other trials. We opened the door. We found a clear result. I think everyone can see whatever you like to see.
Dr. Mandrola: What would your take-home message be after having done this trial design, the trial was conducted in your single center, and you come up with these amazing results? What would your message be to the whole community?
Dr. Sohns: Taking into consideration how severely sick these patients are, I can just repeat it: They are one step away from death, more or less, or from surgical intervention that can prolong their life. You should also consider that there are options like atrial fibrillation ablation that can buy time, postpone the natural course, or even in some patients replace the destination therapy. Therefore, in my opinion the next guidelines should recommend that every patient should carefully be checked for sinus rhythm before bringing these patients into the environment of transplantation.
Dr. Sommer: My interpretation is that we have to try to bring into physicians’ minds that besides a well-established and well-documented effect of drug therapy with the fabulous four, we may now have the fabulous five, including an ablation option for patients with atrial fibrillation.
Dr. Mandrola is a clinical electrophysiologist at Baptist Medical Associates, Louisville, Ky. Dr. Sohns is deputy director of the Heart and Diabetes Center NRW, Ruhr University Bochum, Bad Oeynhausen, Germany. Dr. Sommer is professor of cardiology at the Heart and Diabetes Center NRW. Dr. Mandrola reported no conflicts of interest. Dr. Sohns reported receiving research funding from Else Kröner–Fresenius–Stiftung. Dr. Sommer reported consulting with Abbott, Biosense Webster, Boston Scientific, and Medtronic USA.
A version of this article first appeared on Medscape.com.
Heart failure guidelines update: What the ESC got right
This transcript has been edited for clarity.
This is my usual blog, except I am here from the absolutely beautiful city of Amsterdam, where the annual congress of the European Society of Cardiology has been going on.
SGLT2 inhibitors for HFpEF and HFrEF
I’m going to review very briefly the 2023 focused update to the ESC heart failure guidelines. Theresa McDonagh was the first author of this and of the previous ESC or European guidelines. These are a little bit different than the American guidelines, which were presented in 2022. We know that we need an update. The Europeans have gotten ahead of us, and now we have the European update, which I find incredibly well written and it really highlights the areas that I think the takeaways are for the clinicians.
First, we have been seeing now for several years – since 2018 – the benefits of the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Every time we lift the veil on something, there they are in a positive light. We have learned about heart failure with reduced ejection fraction (HFrEF) for both empagliflozin and dapagliflozin. There are very similar results. One population may be enriched with a little of this and a little of that, but the basic messages are the same. In HFrEF, both of these drugs improve outcomes and it happens quickly. You don’t have to wait 1 or 2 years to see this. Within months, and actually within days, you start to see the curves split apart statistically.
The next logical ground was heart failure with preserved ejection fraction (HFpEF). The definition, when we started the HFpEF trials, was 45% or greater. I want the audience to realize that, in the midst of all these trials, we came out – we meaning the American Heart Association, the American College of Cardiology, and the Heart Failure Society – with the new definition of heart failure, which said that true HFpEF is 50% or greater. That in-between zone of 40%-50% or 41%-49% is mRF, or mid-range, what I call middle of the road. I think the Europeans have really emphasized that to us. I believe that those patients really behave much more like a HFrEF population.
Now that we have very positive findings with the SGLT2 inhibitors, both dapagliflozin and empagliflozin, in HFpEF – defined, as I said, as 40% or 45% or greater, not necessarily 50% – with excellent point estimates that just line up, one on top of the other. It doesn’t matter if patients have diabetes or not; the results are exactly the same.
This has been so promising that I am not surprised that the Europeans elevated the SGLT2 inhibitors to a class 1A indication. In the United States in 2022, we thought we were really way ahead by calling it a class 2A indication. Well, now it’s a class 1A indication in Europe, and I have a feeling that the AHA and the ACC are going to start talking about an update because the data are so strong.
Now, we even have data on initiating these drugs in the hospital. EMPULSE was a very large trial about the benefits of starting these drugs in the hospital. You do not have to wait until the patient is in the outpatient setting. You can start it in the hospital.
When? I have no specific day that I start it. I used to try to do a good diuresis first, get the patients somewhat decongested, and then start it. I don’t want to deprive the patients of the benefits of these drugs that happen very early by waiting until the patients are in the outpatient setting.
In the United States, we’ve had some issues with coverage of some of these drugs. In my institution, we now have both on the formulary, and I pick the drug depending upon the patient’s coverage. Medicare pretty much covers most of them. If the patient is older but not yet a Medicare patient, they may have a very large copay. I advise you to get your offices or your health system to look into this so that, when you give the prescription to the patient, whether they’re leaving the hospital or are now in your clinics, they can actually get the drug.
Finerenone and intravenous iron
There is an additional recommendation in these guidelines for finerenone, the mineralocorticoid receptor agonist that I’ve discussed before, that has some really promising data on type 2 diabetes with chronic kidney disease. They have called that a class 1A indication for finerenone. I think there is more to come.
One more: the iron deficiency. Giving intravenous iron actually does improve symptoms and quality of life. I have seen this in my own patients, so I have been very diligent at looking for iron deficiency.
It is a new era. We have more tools, obviously, for our patients. It means one more drug, and that’s always a challenge. We’ve already been doing the pillars of care. This is the fourth pillar of care, but now with a class 1A indication.
Take a look. They’re easy to read. Dr. McDonagh is the first author, and I think they’ve been extremely well done.
Dr. Piña is professor of medicine at Thomas Jefferson University Hospital in Philadelphia. She is a heart failure and cardiac transplantation expert. She disclosed serving as an adviser/consultant to the FDA’s Center for Devices and Radiological Health and has been a volunteer for the American Heart Association since 1982.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is my usual blog, except I am here from the absolutely beautiful city of Amsterdam, where the annual congress of the European Society of Cardiology has been going on.
SGLT2 inhibitors for HFpEF and HFrEF
I’m going to review very briefly the 2023 focused update to the ESC heart failure guidelines. Theresa McDonagh was the first author of this and of the previous ESC or European guidelines. These are a little bit different than the American guidelines, which were presented in 2022. We know that we need an update. The Europeans have gotten ahead of us, and now we have the European update, which I find incredibly well written and it really highlights the areas that I think the takeaways are for the clinicians.
First, we have been seeing now for several years – since 2018 – the benefits of the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Every time we lift the veil on something, there they are in a positive light. We have learned about heart failure with reduced ejection fraction (HFrEF) for both empagliflozin and dapagliflozin. There are very similar results. One population may be enriched with a little of this and a little of that, but the basic messages are the same. In HFrEF, both of these drugs improve outcomes and it happens quickly. You don’t have to wait 1 or 2 years to see this. Within months, and actually within days, you start to see the curves split apart statistically.
The next logical ground was heart failure with preserved ejection fraction (HFpEF). The definition, when we started the HFpEF trials, was 45% or greater. I want the audience to realize that, in the midst of all these trials, we came out – we meaning the American Heart Association, the American College of Cardiology, and the Heart Failure Society – with the new definition of heart failure, which said that true HFpEF is 50% or greater. That in-between zone of 40%-50% or 41%-49% is mRF, or mid-range, what I call middle of the road. I think the Europeans have really emphasized that to us. I believe that those patients really behave much more like a HFrEF population.
Now that we have very positive findings with the SGLT2 inhibitors, both dapagliflozin and empagliflozin, in HFpEF – defined, as I said, as 40% or 45% or greater, not necessarily 50% – with excellent point estimates that just line up, one on top of the other. It doesn’t matter if patients have diabetes or not; the results are exactly the same.
This has been so promising that I am not surprised that the Europeans elevated the SGLT2 inhibitors to a class 1A indication. In the United States in 2022, we thought we were really way ahead by calling it a class 2A indication. Well, now it’s a class 1A indication in Europe, and I have a feeling that the AHA and the ACC are going to start talking about an update because the data are so strong.
Now, we even have data on initiating these drugs in the hospital. EMPULSE was a very large trial about the benefits of starting these drugs in the hospital. You do not have to wait until the patient is in the outpatient setting. You can start it in the hospital.
When? I have no specific day that I start it. I used to try to do a good diuresis first, get the patients somewhat decongested, and then start it. I don’t want to deprive the patients of the benefits of these drugs that happen very early by waiting until the patients are in the outpatient setting.
In the United States, we’ve had some issues with coverage of some of these drugs. In my institution, we now have both on the formulary, and I pick the drug depending upon the patient’s coverage. Medicare pretty much covers most of them. If the patient is older but not yet a Medicare patient, they may have a very large copay. I advise you to get your offices or your health system to look into this so that, when you give the prescription to the patient, whether they’re leaving the hospital or are now in your clinics, they can actually get the drug.
Finerenone and intravenous iron
There is an additional recommendation in these guidelines for finerenone, the mineralocorticoid receptor agonist that I’ve discussed before, that has some really promising data on type 2 diabetes with chronic kidney disease. They have called that a class 1A indication for finerenone. I think there is more to come.
One more: the iron deficiency. Giving intravenous iron actually does improve symptoms and quality of life. I have seen this in my own patients, so I have been very diligent at looking for iron deficiency.
It is a new era. We have more tools, obviously, for our patients. It means one more drug, and that’s always a challenge. We’ve already been doing the pillars of care. This is the fourth pillar of care, but now with a class 1A indication.
Take a look. They’re easy to read. Dr. McDonagh is the first author, and I think they’ve been extremely well done.
Dr. Piña is professor of medicine at Thomas Jefferson University Hospital in Philadelphia. She is a heart failure and cardiac transplantation expert. She disclosed serving as an adviser/consultant to the FDA’s Center for Devices and Radiological Health and has been a volunteer for the American Heart Association since 1982.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is my usual blog, except I am here from the absolutely beautiful city of Amsterdam, where the annual congress of the European Society of Cardiology has been going on.
SGLT2 inhibitors for HFpEF and HFrEF
I’m going to review very briefly the 2023 focused update to the ESC heart failure guidelines. Theresa McDonagh was the first author of this and of the previous ESC or European guidelines. These are a little bit different than the American guidelines, which were presented in 2022. We know that we need an update. The Europeans have gotten ahead of us, and now we have the European update, which I find incredibly well written and it really highlights the areas that I think the takeaways are for the clinicians.
First, we have been seeing now for several years – since 2018 – the benefits of the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Every time we lift the veil on something, there they are in a positive light. We have learned about heart failure with reduced ejection fraction (HFrEF) for both empagliflozin and dapagliflozin. There are very similar results. One population may be enriched with a little of this and a little of that, but the basic messages are the same. In HFrEF, both of these drugs improve outcomes and it happens quickly. You don’t have to wait 1 or 2 years to see this. Within months, and actually within days, you start to see the curves split apart statistically.
The next logical ground was heart failure with preserved ejection fraction (HFpEF). The definition, when we started the HFpEF trials, was 45% or greater. I want the audience to realize that, in the midst of all these trials, we came out – we meaning the American Heart Association, the American College of Cardiology, and the Heart Failure Society – with the new definition of heart failure, which said that true HFpEF is 50% or greater. That in-between zone of 40%-50% or 41%-49% is mRF, or mid-range, what I call middle of the road. I think the Europeans have really emphasized that to us. I believe that those patients really behave much more like a HFrEF population.
Now that we have very positive findings with the SGLT2 inhibitors, both dapagliflozin and empagliflozin, in HFpEF – defined, as I said, as 40% or 45% or greater, not necessarily 50% – with excellent point estimates that just line up, one on top of the other. It doesn’t matter if patients have diabetes or not; the results are exactly the same.
This has been so promising that I am not surprised that the Europeans elevated the SGLT2 inhibitors to a class 1A indication. In the United States in 2022, we thought we were really way ahead by calling it a class 2A indication. Well, now it’s a class 1A indication in Europe, and I have a feeling that the AHA and the ACC are going to start talking about an update because the data are so strong.
Now, we even have data on initiating these drugs in the hospital. EMPULSE was a very large trial about the benefits of starting these drugs in the hospital. You do not have to wait until the patient is in the outpatient setting. You can start it in the hospital.
When? I have no specific day that I start it. I used to try to do a good diuresis first, get the patients somewhat decongested, and then start it. I don’t want to deprive the patients of the benefits of these drugs that happen very early by waiting until the patients are in the outpatient setting.
In the United States, we’ve had some issues with coverage of some of these drugs. In my institution, we now have both on the formulary, and I pick the drug depending upon the patient’s coverage. Medicare pretty much covers most of them. If the patient is older but not yet a Medicare patient, they may have a very large copay. I advise you to get your offices or your health system to look into this so that, when you give the prescription to the patient, whether they’re leaving the hospital or are now in your clinics, they can actually get the drug.
Finerenone and intravenous iron
There is an additional recommendation in these guidelines for finerenone, the mineralocorticoid receptor agonist that I’ve discussed before, that has some really promising data on type 2 diabetes with chronic kidney disease. They have called that a class 1A indication for finerenone. I think there is more to come.
One more: the iron deficiency. Giving intravenous iron actually does improve symptoms and quality of life. I have seen this in my own patients, so I have been very diligent at looking for iron deficiency.
It is a new era. We have more tools, obviously, for our patients. It means one more drug, and that’s always a challenge. We’ve already been doing the pillars of care. This is the fourth pillar of care, but now with a class 1A indication.
Take a look. They’re easy to read. Dr. McDonagh is the first author, and I think they’ve been extremely well done.
Dr. Piña is professor of medicine at Thomas Jefferson University Hospital in Philadelphia. She is a heart failure and cardiac transplantation expert. She disclosed serving as an adviser/consultant to the FDA’s Center for Devices and Radiological Health and has been a volunteer for the American Heart Association since 1982.
A version of this article appeared on Medscape.com.
New ESC ACS guideline combines STEMI and NSTE-ACS
AMSTERDAM –
The new guideline was released at the annual congress of the European Society of Cardiology, and was published online in the European Heart Journal.
“We found that it was realized by the cardiology community that patients with STEMI, NSTEMI, or unstable angina represent a spectrum,” the chair of the guideline task force, Robert Byrne, MD, chair of cardiovascular research at the RCSI University of Medicine and Health Sciences, Dublin, explained to this news organization. “After the initial triage and management decisions, then most of the rest of the care follows a common pathway so it would make sense to consider everything in one guideline.”
Dr. Byrne noted that for all patients with a suspected ACS, the guideline recommendation is to administer an ECG within 10 minutes of presentation. The time is critical particularly for those with an occluded epicardial vessel. If there are features on the ECG that suggest an acutely occluded epicardial vessel, then the patient needs immediate angiography or primary angioplasty.
“The 10-minute guidance has been maintained from previous guidelines, but the nuance in the new guideline is that typically when we think of an occluded epicardial vessel we think of ST-elevation on the ECG,” Byrne said. “While this captures most occluded epicardial vessels, it doesn’t capture all of them. So, we have provided some guidance on alternative ECG patterns which might be indicative of an acute occlusion of the epicardial vessel and should be dealt with in the same way as an ST-elevation MI. This is a new concept.”
This situation could arise when a patient has an occluded circumflex artery and the regular ECG may not show ST elevation but the patient has ongoing pain, he noted. “There are additional ECG leads that can be looked at that might identify patients who need an immediate invasive strategy.
“This is one more reason why all ACS patients should be considered as part of one spectrum, and while the ECG gives us important information, it is not the only thing to consider. Dividing the conditions up as to whether a patient has ST elevation or not does not always make pathophysiological sense,” he added.
Dr. Byrne noted that the new guidelines have tried to reach a wider stakeholder group that includes emergency doctors, internal medicine physicians, general practitioners, and surgeons, as well as cardiologists. The document includes animations in an effort to increase the reach of the guidelines to noncardiology stakeholders, and for the first time, the task force included a patient representative.
“As part of this strategy, we have put more structure in to emphasize the importance that at first contact, we already want to be thinking of antithrombotic therapy and whether the patient needs urgent transfer to the nearest cath lab. We also want to be thinking straight away about preventing the next heart attack by implementing strong secondary prevention measures,” he commented.
Dr. Byrne highlighted a few changes to individual recommendations in the new guidelines.
Invasive management in NSTE-ACS
He pointed out that a small change has been made in the advice on invasive management for patients with non–ST-elevation ACS.
Dr. Byrne explained that patients with ST elevation should be sent immediately to a cath lab for PCI. If this is not possible within 120 minutes, then the patient should receive thrombolysis. This recommendation is the same as in previous guidelines.
He added, however, that there is some novelty in recommendations for patients who don’t have ST elevation but do have a positive troponin. For this group, previous guidelines gave a Class I recommendation that all such patients undergo an angiogram within the first 24 hours. However, an additional meta-analysis that was published in 2022 showed that the evidence for triaging all patients to the cath lab within 24 hours is somewhat limited, Dr. Byrne noted.
“At the end of the day, the task force felt that a Class I recommendation to get all patients to the cath lab within 24 hours was too strong and couldn’t be sustained, so it has been downgraded to a Class IIa recommendation, which we thought was more appropriate,” he said.
“So, while all patients should still have an angiogram during the hospital admission, if they are high-risk ACS, the imperative to get everyone to the cath lab within 24 hours – which many of our colleagues were finding difficult to achieve – does not seem to be backed up by the evidence,” he added.
Antithrombotic therapy
Addressing administration of antithrombotics, the guidelines emphasize that at the time of initial diagnosis, all patients should receive antithrombotic therapy, usually aspirin and a parenteral antithrombotic, such as heparin, enoxaparin, bivalirudin, or fondaparinux. Dr. Byrne noted that the guidelines have a new algorithm as to which of these antithrombotics to give, depending on the clinical presentation of the patient.
On the use of upfront P2Y12 inhibitors, Dr. Byrne said the new guidelines only give a weak recommendation for this.
“Giving a P2Y12 inhibitor up front does not have a strong evidence base, and it’s not unreasonable to wait, do the angiogram, see where you are, and then start the P2Y12 inhibitor. This is something that’s not widely done in clinical practice,” he commented.
“The last 2020 guideline gave a Class III recommendation for upfront P2Y12 inhibitor therapy for ACS patients who do not have STEMI. We’ve generally maintained that with the introduction of the exception that if you are in a health care system where there is a long wait to get to the cath lab – 5 ,6 or 7 days – then it’s reasonable to make an exception and give a P2Y12 inhibitor, but otherwise we’ve sustained the Class III recommendation,” he noted.
Also, for patients who have STEMI, there is a new Class IIb recommendation that upfront P2Y12 inhibitors may be considered.
“This is also rather a weak recommendation. There isn’t a strong rationale to give a P2Y12 inhibitor it in ST elevation either. It’s also reasonable to wait,” he added.
Don’t rush cardiac arrest patients to the cath lab
Another update in the guidelines involves the management of patients with cardiac arrest who have been resuscitated. Dr. Byrne explained that these patients would all receive an immediate ECG, and if it is found that they have ST elevation, they would be sent immediately to the cath lab. But a series of randomized trials has suggested that for patients who don’t have ST elevation, it is not necessary to rush these patients to the cath lab.
“We’ve given a Class III recommendation for this, saying it may be better to stabilize the patients first in the ICU. This is in recognition that a large proportion of these patients turn out not to have an MI. They have had a cardiac arrest for another reason,” Dr. Byrne noted. “Moving them to the cath lab when they are still unstable could be harming these patients rather than helping them.”
Revascularization for multivessel disease
Dr. Byrne notes that revascularization remains a critical element in the care for patients with STEMI, and there is a new recommendation in this area for patients with multivessel disease.
“Up to half of patients presenting with STEMI have multivessel disease, and we now have five randomized trials to say that these patients should have complete revascularization rather than just the culprit vessel. There is a new Class I recommendation for this,” he said.
However, the optimal timing of revascularization (immediate vs. staged) has still not been investigated in adequately sized randomized trials, and no recommendation has been made on this, the task force notes.
Dr. Byrne commented: “If you want to do everything in one go, that’s fine, but it’s also okay to do the culprit lesion first and then the other vessels at a later date within 45 days. This might depend on individual circumstances. For example, if there are complex lesions or the vessels are heavily calcified, then it may be best to get the culprit lesion fixed first and let the patient recover, then bring them back in for the rest.”
He pointed out that the results of the MULTISTARS trial, which were not available when the task force was formulating the guidelines, were reported at the ESC Congress and confirmed their recommendation.
DAPT after PCI
On the duration of dual antiplatelet therapy (DAPT) after PCI, the new guidelines have largely retained prior recommendations for a default strategy of 12 months for the combination of aspirin and a P2Y12 inhibitor.
“This was the subject of some discussion, as there have been several trials now looking at shorter durations of DAPT and deescalating after a few months to just one of these treatments. And while there is a rationale to do this, we think it’s best to be kept as an alternative strategy rather than the default strategy,” Dr. Byrne said.
He explained that the trials of DAPT deescalation tended to enroll lower-risk patients, which reduced the generalizability of the results.
Most of the trials only randomly assigned patients to shorter durations of DAPT when they were event-free for some period, she said.
“This is a dynamic decision-making process and reflects the real world to some extent. We think it is best to recommend the standard aspirin and a P2Y12 inhibitor for the 12-month duration, but at 3 months, if the patient is doing well but you may be worried about bleeding risk, then you could decide to deescalate to single antiplatelet therapy,” she noted. “So, there is Class IIa recommendation that this can be considered, but it is not recommended as the default position.”
Polypill for secondary prevention
Another innovation in the new guidelines is a new Class IIa recommendation for prescription of a polypill containing secondary prevention medications for patients on discharge from hospital.
This recommendation follows a trial that showed that the use of such a polypill helps patients be more adherent to the therapies prescribed.
Byrne explains that such a polypill may contain aspirin, an ACE inhibitor, and a statin. Several varieties are available in most European countries, but they are not widely used.
On secondary prevention, he stressed, “Prevention of the next heart attack starts before the patient leaves hospital. It is important to make sure the patient has the right medication on board, including a high-dose, high-intensity statin, and has been referred to a cardiac rehabilitation program. These are largely maintained recommendations from previous guidelines, but they are very important.”
A version of this article appeared on Medscape.com.
AMSTERDAM –
The new guideline was released at the annual congress of the European Society of Cardiology, and was published online in the European Heart Journal.
“We found that it was realized by the cardiology community that patients with STEMI, NSTEMI, or unstable angina represent a spectrum,” the chair of the guideline task force, Robert Byrne, MD, chair of cardiovascular research at the RCSI University of Medicine and Health Sciences, Dublin, explained to this news organization. “After the initial triage and management decisions, then most of the rest of the care follows a common pathway so it would make sense to consider everything in one guideline.”
Dr. Byrne noted that for all patients with a suspected ACS, the guideline recommendation is to administer an ECG within 10 minutes of presentation. The time is critical particularly for those with an occluded epicardial vessel. If there are features on the ECG that suggest an acutely occluded epicardial vessel, then the patient needs immediate angiography or primary angioplasty.
“The 10-minute guidance has been maintained from previous guidelines, but the nuance in the new guideline is that typically when we think of an occluded epicardial vessel we think of ST-elevation on the ECG,” Byrne said. “While this captures most occluded epicardial vessels, it doesn’t capture all of them. So, we have provided some guidance on alternative ECG patterns which might be indicative of an acute occlusion of the epicardial vessel and should be dealt with in the same way as an ST-elevation MI. This is a new concept.”
This situation could arise when a patient has an occluded circumflex artery and the regular ECG may not show ST elevation but the patient has ongoing pain, he noted. “There are additional ECG leads that can be looked at that might identify patients who need an immediate invasive strategy.
“This is one more reason why all ACS patients should be considered as part of one spectrum, and while the ECG gives us important information, it is not the only thing to consider. Dividing the conditions up as to whether a patient has ST elevation or not does not always make pathophysiological sense,” he added.
Dr. Byrne noted that the new guidelines have tried to reach a wider stakeholder group that includes emergency doctors, internal medicine physicians, general practitioners, and surgeons, as well as cardiologists. The document includes animations in an effort to increase the reach of the guidelines to noncardiology stakeholders, and for the first time, the task force included a patient representative.
“As part of this strategy, we have put more structure in to emphasize the importance that at first contact, we already want to be thinking of antithrombotic therapy and whether the patient needs urgent transfer to the nearest cath lab. We also want to be thinking straight away about preventing the next heart attack by implementing strong secondary prevention measures,” he commented.
Dr. Byrne highlighted a few changes to individual recommendations in the new guidelines.
Invasive management in NSTE-ACS
He pointed out that a small change has been made in the advice on invasive management for patients with non–ST-elevation ACS.
Dr. Byrne explained that patients with ST elevation should be sent immediately to a cath lab for PCI. If this is not possible within 120 minutes, then the patient should receive thrombolysis. This recommendation is the same as in previous guidelines.
He added, however, that there is some novelty in recommendations for patients who don’t have ST elevation but do have a positive troponin. For this group, previous guidelines gave a Class I recommendation that all such patients undergo an angiogram within the first 24 hours. However, an additional meta-analysis that was published in 2022 showed that the evidence for triaging all patients to the cath lab within 24 hours is somewhat limited, Dr. Byrne noted.
“At the end of the day, the task force felt that a Class I recommendation to get all patients to the cath lab within 24 hours was too strong and couldn’t be sustained, so it has been downgraded to a Class IIa recommendation, which we thought was more appropriate,” he said.
“So, while all patients should still have an angiogram during the hospital admission, if they are high-risk ACS, the imperative to get everyone to the cath lab within 24 hours – which many of our colleagues were finding difficult to achieve – does not seem to be backed up by the evidence,” he added.
Antithrombotic therapy
Addressing administration of antithrombotics, the guidelines emphasize that at the time of initial diagnosis, all patients should receive antithrombotic therapy, usually aspirin and a parenteral antithrombotic, such as heparin, enoxaparin, bivalirudin, or fondaparinux. Dr. Byrne noted that the guidelines have a new algorithm as to which of these antithrombotics to give, depending on the clinical presentation of the patient.
On the use of upfront P2Y12 inhibitors, Dr. Byrne said the new guidelines only give a weak recommendation for this.
“Giving a P2Y12 inhibitor up front does not have a strong evidence base, and it’s not unreasonable to wait, do the angiogram, see where you are, and then start the P2Y12 inhibitor. This is something that’s not widely done in clinical practice,” he commented.
“The last 2020 guideline gave a Class III recommendation for upfront P2Y12 inhibitor therapy for ACS patients who do not have STEMI. We’ve generally maintained that with the introduction of the exception that if you are in a health care system where there is a long wait to get to the cath lab – 5 ,6 or 7 days – then it’s reasonable to make an exception and give a P2Y12 inhibitor, but otherwise we’ve sustained the Class III recommendation,” he noted.
Also, for patients who have STEMI, there is a new Class IIb recommendation that upfront P2Y12 inhibitors may be considered.
“This is also rather a weak recommendation. There isn’t a strong rationale to give a P2Y12 inhibitor it in ST elevation either. It’s also reasonable to wait,” he added.
Don’t rush cardiac arrest patients to the cath lab
Another update in the guidelines involves the management of patients with cardiac arrest who have been resuscitated. Dr. Byrne explained that these patients would all receive an immediate ECG, and if it is found that they have ST elevation, they would be sent immediately to the cath lab. But a series of randomized trials has suggested that for patients who don’t have ST elevation, it is not necessary to rush these patients to the cath lab.
“We’ve given a Class III recommendation for this, saying it may be better to stabilize the patients first in the ICU. This is in recognition that a large proportion of these patients turn out not to have an MI. They have had a cardiac arrest for another reason,” Dr. Byrne noted. “Moving them to the cath lab when they are still unstable could be harming these patients rather than helping them.”
Revascularization for multivessel disease
Dr. Byrne notes that revascularization remains a critical element in the care for patients with STEMI, and there is a new recommendation in this area for patients with multivessel disease.
“Up to half of patients presenting with STEMI have multivessel disease, and we now have five randomized trials to say that these patients should have complete revascularization rather than just the culprit vessel. There is a new Class I recommendation for this,” he said.
However, the optimal timing of revascularization (immediate vs. staged) has still not been investigated in adequately sized randomized trials, and no recommendation has been made on this, the task force notes.
Dr. Byrne commented: “If you want to do everything in one go, that’s fine, but it’s also okay to do the culprit lesion first and then the other vessels at a later date within 45 days. This might depend on individual circumstances. For example, if there are complex lesions or the vessels are heavily calcified, then it may be best to get the culprit lesion fixed first and let the patient recover, then bring them back in for the rest.”
He pointed out that the results of the MULTISTARS trial, which were not available when the task force was formulating the guidelines, were reported at the ESC Congress and confirmed their recommendation.
DAPT after PCI
On the duration of dual antiplatelet therapy (DAPT) after PCI, the new guidelines have largely retained prior recommendations for a default strategy of 12 months for the combination of aspirin and a P2Y12 inhibitor.
“This was the subject of some discussion, as there have been several trials now looking at shorter durations of DAPT and deescalating after a few months to just one of these treatments. And while there is a rationale to do this, we think it’s best to be kept as an alternative strategy rather than the default strategy,” Dr. Byrne said.
He explained that the trials of DAPT deescalation tended to enroll lower-risk patients, which reduced the generalizability of the results.
Most of the trials only randomly assigned patients to shorter durations of DAPT when they were event-free for some period, she said.
“This is a dynamic decision-making process and reflects the real world to some extent. We think it is best to recommend the standard aspirin and a P2Y12 inhibitor for the 12-month duration, but at 3 months, if the patient is doing well but you may be worried about bleeding risk, then you could decide to deescalate to single antiplatelet therapy,” she noted. “So, there is Class IIa recommendation that this can be considered, but it is not recommended as the default position.”
Polypill for secondary prevention
Another innovation in the new guidelines is a new Class IIa recommendation for prescription of a polypill containing secondary prevention medications for patients on discharge from hospital.
This recommendation follows a trial that showed that the use of such a polypill helps patients be more adherent to the therapies prescribed.
Byrne explains that such a polypill may contain aspirin, an ACE inhibitor, and a statin. Several varieties are available in most European countries, but they are not widely used.
On secondary prevention, he stressed, “Prevention of the next heart attack starts before the patient leaves hospital. It is important to make sure the patient has the right medication on board, including a high-dose, high-intensity statin, and has been referred to a cardiac rehabilitation program. These are largely maintained recommendations from previous guidelines, but they are very important.”
A version of this article appeared on Medscape.com.
AMSTERDAM –
The new guideline was released at the annual congress of the European Society of Cardiology, and was published online in the European Heart Journal.
“We found that it was realized by the cardiology community that patients with STEMI, NSTEMI, or unstable angina represent a spectrum,” the chair of the guideline task force, Robert Byrne, MD, chair of cardiovascular research at the RCSI University of Medicine and Health Sciences, Dublin, explained to this news organization. “After the initial triage and management decisions, then most of the rest of the care follows a common pathway so it would make sense to consider everything in one guideline.”
Dr. Byrne noted that for all patients with a suspected ACS, the guideline recommendation is to administer an ECG within 10 minutes of presentation. The time is critical particularly for those with an occluded epicardial vessel. If there are features on the ECG that suggest an acutely occluded epicardial vessel, then the patient needs immediate angiography or primary angioplasty.
“The 10-minute guidance has been maintained from previous guidelines, but the nuance in the new guideline is that typically when we think of an occluded epicardial vessel we think of ST-elevation on the ECG,” Byrne said. “While this captures most occluded epicardial vessels, it doesn’t capture all of them. So, we have provided some guidance on alternative ECG patterns which might be indicative of an acute occlusion of the epicardial vessel and should be dealt with in the same way as an ST-elevation MI. This is a new concept.”
This situation could arise when a patient has an occluded circumflex artery and the regular ECG may not show ST elevation but the patient has ongoing pain, he noted. “There are additional ECG leads that can be looked at that might identify patients who need an immediate invasive strategy.
“This is one more reason why all ACS patients should be considered as part of one spectrum, and while the ECG gives us important information, it is not the only thing to consider. Dividing the conditions up as to whether a patient has ST elevation or not does not always make pathophysiological sense,” he added.
Dr. Byrne noted that the new guidelines have tried to reach a wider stakeholder group that includes emergency doctors, internal medicine physicians, general practitioners, and surgeons, as well as cardiologists. The document includes animations in an effort to increase the reach of the guidelines to noncardiology stakeholders, and for the first time, the task force included a patient representative.
“As part of this strategy, we have put more structure in to emphasize the importance that at first contact, we already want to be thinking of antithrombotic therapy and whether the patient needs urgent transfer to the nearest cath lab. We also want to be thinking straight away about preventing the next heart attack by implementing strong secondary prevention measures,” he commented.
Dr. Byrne highlighted a few changes to individual recommendations in the new guidelines.
Invasive management in NSTE-ACS
He pointed out that a small change has been made in the advice on invasive management for patients with non–ST-elevation ACS.
Dr. Byrne explained that patients with ST elevation should be sent immediately to a cath lab for PCI. If this is not possible within 120 minutes, then the patient should receive thrombolysis. This recommendation is the same as in previous guidelines.
He added, however, that there is some novelty in recommendations for patients who don’t have ST elevation but do have a positive troponin. For this group, previous guidelines gave a Class I recommendation that all such patients undergo an angiogram within the first 24 hours. However, an additional meta-analysis that was published in 2022 showed that the evidence for triaging all patients to the cath lab within 24 hours is somewhat limited, Dr. Byrne noted.
“At the end of the day, the task force felt that a Class I recommendation to get all patients to the cath lab within 24 hours was too strong and couldn’t be sustained, so it has been downgraded to a Class IIa recommendation, which we thought was more appropriate,” he said.
“So, while all patients should still have an angiogram during the hospital admission, if they are high-risk ACS, the imperative to get everyone to the cath lab within 24 hours – which many of our colleagues were finding difficult to achieve – does not seem to be backed up by the evidence,” he added.
Antithrombotic therapy
Addressing administration of antithrombotics, the guidelines emphasize that at the time of initial diagnosis, all patients should receive antithrombotic therapy, usually aspirin and a parenteral antithrombotic, such as heparin, enoxaparin, bivalirudin, or fondaparinux. Dr. Byrne noted that the guidelines have a new algorithm as to which of these antithrombotics to give, depending on the clinical presentation of the patient.
On the use of upfront P2Y12 inhibitors, Dr. Byrne said the new guidelines only give a weak recommendation for this.
“Giving a P2Y12 inhibitor up front does not have a strong evidence base, and it’s not unreasonable to wait, do the angiogram, see where you are, and then start the P2Y12 inhibitor. This is something that’s not widely done in clinical practice,” he commented.
“The last 2020 guideline gave a Class III recommendation for upfront P2Y12 inhibitor therapy for ACS patients who do not have STEMI. We’ve generally maintained that with the introduction of the exception that if you are in a health care system where there is a long wait to get to the cath lab – 5 ,6 or 7 days – then it’s reasonable to make an exception and give a P2Y12 inhibitor, but otherwise we’ve sustained the Class III recommendation,” he noted.
Also, for patients who have STEMI, there is a new Class IIb recommendation that upfront P2Y12 inhibitors may be considered.
“This is also rather a weak recommendation. There isn’t a strong rationale to give a P2Y12 inhibitor it in ST elevation either. It’s also reasonable to wait,” he added.
Don’t rush cardiac arrest patients to the cath lab
Another update in the guidelines involves the management of patients with cardiac arrest who have been resuscitated. Dr. Byrne explained that these patients would all receive an immediate ECG, and if it is found that they have ST elevation, they would be sent immediately to the cath lab. But a series of randomized trials has suggested that for patients who don’t have ST elevation, it is not necessary to rush these patients to the cath lab.
“We’ve given a Class III recommendation for this, saying it may be better to stabilize the patients first in the ICU. This is in recognition that a large proportion of these patients turn out not to have an MI. They have had a cardiac arrest for another reason,” Dr. Byrne noted. “Moving them to the cath lab when they are still unstable could be harming these patients rather than helping them.”
Revascularization for multivessel disease
Dr. Byrne notes that revascularization remains a critical element in the care for patients with STEMI, and there is a new recommendation in this area for patients with multivessel disease.
“Up to half of patients presenting with STEMI have multivessel disease, and we now have five randomized trials to say that these patients should have complete revascularization rather than just the culprit vessel. There is a new Class I recommendation for this,” he said.
However, the optimal timing of revascularization (immediate vs. staged) has still not been investigated in adequately sized randomized trials, and no recommendation has been made on this, the task force notes.
Dr. Byrne commented: “If you want to do everything in one go, that’s fine, but it’s also okay to do the culprit lesion first and then the other vessels at a later date within 45 days. This might depend on individual circumstances. For example, if there are complex lesions or the vessels are heavily calcified, then it may be best to get the culprit lesion fixed first and let the patient recover, then bring them back in for the rest.”
He pointed out that the results of the MULTISTARS trial, which were not available when the task force was formulating the guidelines, were reported at the ESC Congress and confirmed their recommendation.
DAPT after PCI
On the duration of dual antiplatelet therapy (DAPT) after PCI, the new guidelines have largely retained prior recommendations for a default strategy of 12 months for the combination of aspirin and a P2Y12 inhibitor.
“This was the subject of some discussion, as there have been several trials now looking at shorter durations of DAPT and deescalating after a few months to just one of these treatments. And while there is a rationale to do this, we think it’s best to be kept as an alternative strategy rather than the default strategy,” Dr. Byrne said.
He explained that the trials of DAPT deescalation tended to enroll lower-risk patients, which reduced the generalizability of the results.
Most of the trials only randomly assigned patients to shorter durations of DAPT when they were event-free for some period, she said.
“This is a dynamic decision-making process and reflects the real world to some extent. We think it is best to recommend the standard aspirin and a P2Y12 inhibitor for the 12-month duration, but at 3 months, if the patient is doing well but you may be worried about bleeding risk, then you could decide to deescalate to single antiplatelet therapy,” she noted. “So, there is Class IIa recommendation that this can be considered, but it is not recommended as the default position.”
Polypill for secondary prevention
Another innovation in the new guidelines is a new Class IIa recommendation for prescription of a polypill containing secondary prevention medications for patients on discharge from hospital.
This recommendation follows a trial that showed that the use of such a polypill helps patients be more adherent to the therapies prescribed.
Byrne explains that such a polypill may contain aspirin, an ACE inhibitor, and a statin. Several varieties are available in most European countries, but they are not widely used.
On secondary prevention, he stressed, “Prevention of the next heart attack starts before the patient leaves hospital. It is important to make sure the patient has the right medication on board, including a high-dose, high-intensity statin, and has been referred to a cardiac rehabilitation program. These are largely maintained recommendations from previous guidelines, but they are very important.”
A version of this article appeared on Medscape.com.
AT ESC CONGRESS 2023
Is complete revascularization now compulsory? MULTISTARS-AMI and FIRE in context
This transcript has been edited for clarity.
Michelle L. O’Donoghue, MD, MPH: Hi. This is Dr. Michelle O’Donoghue reporting for Medscape. Joining me today is Dr. Sahil Parikh, who’s a cardiologist and an interventionalist at Columbia University. He’s an associate professor of medicine.
We’ll be discussing two interesting trials that were presented at the ESC Congress here in Amsterdam. They do have the potential to be very practice-changing, so I think it’s worth talking about.
The FIRE trial
The first trial we’ll be talking about is the FIRE trial. Perhaps setting the stage, Sahil, I’d love to get your thoughts. We’ve had data in this space to suggest that, for patients with STEMI [ST-segment elevation myocardial infarction], a strategy of complete revascularization – and not only treating the culprit lesion but also treating additional lesions – may be of benefit. Where does that lead us in terms of what we didn’t know?
Sahil A. Parikh, MD: I think that the practice has moved, at least in the United States, over the past two decades, from staging percutaneous coronary interventions over 30 days from index to intervention to now trying to do patients in the same hospitalization whenever possible to achieve complete revascularization.
I think these data support not only that complete revascularization is compulsory now in these patients, but also doing it sooner rather than later, and that the benefit applies to most of the patients that we see in clinical practice. In the earlier data, the patients were relatively youthful – under Medicare age, less than 65 – and now this dataset has a median age of 80. This is more like the real-world clinical practice that most of us are encountering, and it extends the benefit, perhaps, greater than we’ve ever seen before.
O’Donoghue: The FIRE trial is interesting. As you say, it enrolled patients who were over the age of 75, where I think that some proceduralists are probably a little bit hesitant to think about complete revascularization due to concerns about any additional contrast load on their kidneys and other types of comorbidities. Of course, for any trial, there’s going to be some patient selection.
I think it’s very reassuring that even in this older patient group, a strategy of treating all the lesions – and not only in STEMI but also in non-STEMI patients – reduced cardiovascular events and mortality. I was really quite impressed by the mortality benefit.
Parikh: The mortality curve is almost surprising to me. On the other hand, it emboldens us now that we can treat these patients more completely and earlier in their clinical presentation. Certainly, we worried about contrast exposure and the duration of procedures in this older population, but it seems that the benefit that’s derived, which we saw in younger patients where we had a natural inclination to be more aggressive, extends also to this older population.
MULTISTARS AMI
O’Donoghue: To the question of timing, as you mentioned, prior to this, we had a study presented earlier this year, the BIOVASC trial, which also was suggestive that maybe earlier complete revascularization was better. But it wasn’t a significant difference, at least for the primary outcome. Now we have MULTISTARS AMI, which is very supportive of what we saw earlier this year, suggesting that complete revascularization really at the time that you’re treating the culprit may be the way to go.
Parikh: All of us, as interventionalists, are circumspect about what we might do in the middle of the night versus what we would do in the light of day. Certainly it seems clear, particularly if it’s straightforward anatomy, that taking care of it in the index procedure is not only saving contrast and fluoroscopy time, but it’s also providing a clinical benefit to the patients. That’s something that will also impact how clinicians interpret these data. Previously, there was always a question about whether we should just do it in the same hospitalization or do it at the same time. I think now, increasingly, we’re emboldened to do more in the index procedure.
O’Donoghue: When you’re thinking about nonculprit lesions and which ones to treat, do you always make that determination based on physiologic guidance of some kind? Are you using instantaneous wave-free ratio? What’s your practice?
Parikh: In the acute setting, imaging is superior for at least the assessment of which is a culprit. If you see a ruptured atherothrombotic situation on optical coherence tomography, for example, that’s fairly convincing and definitive. In the absence of that physiology, we are taught to avoid in the infarct-related artery because of potential spuriously false-negative findings.
In this situation, certainly, an imaging subgroup probably would be helpful because some of the benefit is almost certainly derived from identifying the infarct-related artery by accident – in other words, doing what you thought was the nonculprit artery, which is, in fact, the culprit. I think that probably is part of this. As somebody who uses imaging in the overwhelming number of my cases, I think that imaging would be an important surrogate to this.
Index procedure versus staged
O’Donoghue: For the operator who is coming in to do their STEMI case at 2:00 in the morning, would these data now push you toward doing complete revascularization at that time of night, or do you think that there is wiggle room in terms of interpreting these results regarding timing, where as long as you were doing it before hospital discharge and not, let’s say, 30 days out, that you may be able to derive the same benefit? What are some of the pros and cons?
Parikh: There’s definitely a fatigue factor in the middle of the night if it’s a particularly arduous intervention for the index infarct-related artery. I think there’s a human element where it may make sense just to stop and then bring the patient back in the same hospitalization. It’s clear, though, that doing complete revascularization is better and doing it sooner is better. How soon one actually does it is a judgment call, as ever.
In our practice, we’ve been pushing ourselves to get most of the patients done in their index hospitalization. If you have a left-sided culprit, the left anterior descending artery, for example, and there’s a high-grade stenosis in the circumflex, it may make sense to take care of that in the same index procedure. If, on the other hand, it’s in the right coronary artery where you have to put a new guide in and spend more time, that may be a patient whom you stage. I think those nuances will come up as interventionalists look at the subgroup analysis data more carefully.
O’Donoghue: Those are great points, and I think they also underscore that we always need to think about what type of patient was enrolled in these studies. Certainly, if you have somebody with renal dysfunction, there might be more concern about giving them a large contrast load all in one sitting, albeit hard to know whether they do or not. But spacing that out by just a couple of days would really have a big impact.
Parikh: Very often in the STEMI patient, you don’t have the benefit of knowing the creatinine. The patient will come in immediately, if not directly from the ambulance to the cath lab, and there are no laboratories at all to work with. If the patient has never been seen in the system before, you won’t know. Again, in those situations, one may have pause, particularly if it’s an older patient. I think what’s reassuring, though, is that the data are supportive of being more aggressive earlier, and certainly this is the dataset that we were looking for.
O’Donoghue: To summarize, the two key takeaways are that, one, we now have more data to support a complete revascularization strategy and even extending that now to non-STEMI patients. Two, sooner appears to be better, so ideally, all done at the time of the index procedure. I think this is very interesting science and we’ll see how it changes practice.
Thanks for joining me today. Signing off for Medscape, this is Dr. Michelle O’Donoghue.
Michelle O’Donoghue is a cardiologist at Brigham and Women’s Hospital and senior investigator with the TIMI Study Group.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Michelle L. O’Donoghue, MD, MPH: Hi. This is Dr. Michelle O’Donoghue reporting for Medscape. Joining me today is Dr. Sahil Parikh, who’s a cardiologist and an interventionalist at Columbia University. He’s an associate professor of medicine.
We’ll be discussing two interesting trials that were presented at the ESC Congress here in Amsterdam. They do have the potential to be very practice-changing, so I think it’s worth talking about.
The FIRE trial
The first trial we’ll be talking about is the FIRE trial. Perhaps setting the stage, Sahil, I’d love to get your thoughts. We’ve had data in this space to suggest that, for patients with STEMI [ST-segment elevation myocardial infarction], a strategy of complete revascularization – and not only treating the culprit lesion but also treating additional lesions – may be of benefit. Where does that lead us in terms of what we didn’t know?
Sahil A. Parikh, MD: I think that the practice has moved, at least in the United States, over the past two decades, from staging percutaneous coronary interventions over 30 days from index to intervention to now trying to do patients in the same hospitalization whenever possible to achieve complete revascularization.
I think these data support not only that complete revascularization is compulsory now in these patients, but also doing it sooner rather than later, and that the benefit applies to most of the patients that we see in clinical practice. In the earlier data, the patients were relatively youthful – under Medicare age, less than 65 – and now this dataset has a median age of 80. This is more like the real-world clinical practice that most of us are encountering, and it extends the benefit, perhaps, greater than we’ve ever seen before.
O’Donoghue: The FIRE trial is interesting. As you say, it enrolled patients who were over the age of 75, where I think that some proceduralists are probably a little bit hesitant to think about complete revascularization due to concerns about any additional contrast load on their kidneys and other types of comorbidities. Of course, for any trial, there’s going to be some patient selection.
I think it’s very reassuring that even in this older patient group, a strategy of treating all the lesions – and not only in STEMI but also in non-STEMI patients – reduced cardiovascular events and mortality. I was really quite impressed by the mortality benefit.
Parikh: The mortality curve is almost surprising to me. On the other hand, it emboldens us now that we can treat these patients more completely and earlier in their clinical presentation. Certainly, we worried about contrast exposure and the duration of procedures in this older population, but it seems that the benefit that’s derived, which we saw in younger patients where we had a natural inclination to be more aggressive, extends also to this older population.
MULTISTARS AMI
O’Donoghue: To the question of timing, as you mentioned, prior to this, we had a study presented earlier this year, the BIOVASC trial, which also was suggestive that maybe earlier complete revascularization was better. But it wasn’t a significant difference, at least for the primary outcome. Now we have MULTISTARS AMI, which is very supportive of what we saw earlier this year, suggesting that complete revascularization really at the time that you’re treating the culprit may be the way to go.
Parikh: All of us, as interventionalists, are circumspect about what we might do in the middle of the night versus what we would do in the light of day. Certainly it seems clear, particularly if it’s straightforward anatomy, that taking care of it in the index procedure is not only saving contrast and fluoroscopy time, but it’s also providing a clinical benefit to the patients. That’s something that will also impact how clinicians interpret these data. Previously, there was always a question about whether we should just do it in the same hospitalization or do it at the same time. I think now, increasingly, we’re emboldened to do more in the index procedure.
O’Donoghue: When you’re thinking about nonculprit lesions and which ones to treat, do you always make that determination based on physiologic guidance of some kind? Are you using instantaneous wave-free ratio? What’s your practice?
Parikh: In the acute setting, imaging is superior for at least the assessment of which is a culprit. If you see a ruptured atherothrombotic situation on optical coherence tomography, for example, that’s fairly convincing and definitive. In the absence of that physiology, we are taught to avoid in the infarct-related artery because of potential spuriously false-negative findings.
In this situation, certainly, an imaging subgroup probably would be helpful because some of the benefit is almost certainly derived from identifying the infarct-related artery by accident – in other words, doing what you thought was the nonculprit artery, which is, in fact, the culprit. I think that probably is part of this. As somebody who uses imaging in the overwhelming number of my cases, I think that imaging would be an important surrogate to this.
Index procedure versus staged
O’Donoghue: For the operator who is coming in to do their STEMI case at 2:00 in the morning, would these data now push you toward doing complete revascularization at that time of night, or do you think that there is wiggle room in terms of interpreting these results regarding timing, where as long as you were doing it before hospital discharge and not, let’s say, 30 days out, that you may be able to derive the same benefit? What are some of the pros and cons?
Parikh: There’s definitely a fatigue factor in the middle of the night if it’s a particularly arduous intervention for the index infarct-related artery. I think there’s a human element where it may make sense just to stop and then bring the patient back in the same hospitalization. It’s clear, though, that doing complete revascularization is better and doing it sooner is better. How soon one actually does it is a judgment call, as ever.
In our practice, we’ve been pushing ourselves to get most of the patients done in their index hospitalization. If you have a left-sided culprit, the left anterior descending artery, for example, and there’s a high-grade stenosis in the circumflex, it may make sense to take care of that in the same index procedure. If, on the other hand, it’s in the right coronary artery where you have to put a new guide in and spend more time, that may be a patient whom you stage. I think those nuances will come up as interventionalists look at the subgroup analysis data more carefully.
O’Donoghue: Those are great points, and I think they also underscore that we always need to think about what type of patient was enrolled in these studies. Certainly, if you have somebody with renal dysfunction, there might be more concern about giving them a large contrast load all in one sitting, albeit hard to know whether they do or not. But spacing that out by just a couple of days would really have a big impact.
Parikh: Very often in the STEMI patient, you don’t have the benefit of knowing the creatinine. The patient will come in immediately, if not directly from the ambulance to the cath lab, and there are no laboratories at all to work with. If the patient has never been seen in the system before, you won’t know. Again, in those situations, one may have pause, particularly if it’s an older patient. I think what’s reassuring, though, is that the data are supportive of being more aggressive earlier, and certainly this is the dataset that we were looking for.
O’Donoghue: To summarize, the two key takeaways are that, one, we now have more data to support a complete revascularization strategy and even extending that now to non-STEMI patients. Two, sooner appears to be better, so ideally, all done at the time of the index procedure. I think this is very interesting science and we’ll see how it changes practice.
Thanks for joining me today. Signing off for Medscape, this is Dr. Michelle O’Donoghue.
Michelle O’Donoghue is a cardiologist at Brigham and Women’s Hospital and senior investigator with the TIMI Study Group.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Michelle L. O’Donoghue, MD, MPH: Hi. This is Dr. Michelle O’Donoghue reporting for Medscape. Joining me today is Dr. Sahil Parikh, who’s a cardiologist and an interventionalist at Columbia University. He’s an associate professor of medicine.
We’ll be discussing two interesting trials that were presented at the ESC Congress here in Amsterdam. They do have the potential to be very practice-changing, so I think it’s worth talking about.
The FIRE trial
The first trial we’ll be talking about is the FIRE trial. Perhaps setting the stage, Sahil, I’d love to get your thoughts. We’ve had data in this space to suggest that, for patients with STEMI [ST-segment elevation myocardial infarction], a strategy of complete revascularization – and not only treating the culprit lesion but also treating additional lesions – may be of benefit. Where does that lead us in terms of what we didn’t know?
Sahil A. Parikh, MD: I think that the practice has moved, at least in the United States, over the past two decades, from staging percutaneous coronary interventions over 30 days from index to intervention to now trying to do patients in the same hospitalization whenever possible to achieve complete revascularization.
I think these data support not only that complete revascularization is compulsory now in these patients, but also doing it sooner rather than later, and that the benefit applies to most of the patients that we see in clinical practice. In the earlier data, the patients were relatively youthful – under Medicare age, less than 65 – and now this dataset has a median age of 80. This is more like the real-world clinical practice that most of us are encountering, and it extends the benefit, perhaps, greater than we’ve ever seen before.
O’Donoghue: The FIRE trial is interesting. As you say, it enrolled patients who were over the age of 75, where I think that some proceduralists are probably a little bit hesitant to think about complete revascularization due to concerns about any additional contrast load on their kidneys and other types of comorbidities. Of course, for any trial, there’s going to be some patient selection.
I think it’s very reassuring that even in this older patient group, a strategy of treating all the lesions – and not only in STEMI but also in non-STEMI patients – reduced cardiovascular events and mortality. I was really quite impressed by the mortality benefit.
Parikh: The mortality curve is almost surprising to me. On the other hand, it emboldens us now that we can treat these patients more completely and earlier in their clinical presentation. Certainly, we worried about contrast exposure and the duration of procedures in this older population, but it seems that the benefit that’s derived, which we saw in younger patients where we had a natural inclination to be more aggressive, extends also to this older population.
MULTISTARS AMI
O’Donoghue: To the question of timing, as you mentioned, prior to this, we had a study presented earlier this year, the BIOVASC trial, which also was suggestive that maybe earlier complete revascularization was better. But it wasn’t a significant difference, at least for the primary outcome. Now we have MULTISTARS AMI, which is very supportive of what we saw earlier this year, suggesting that complete revascularization really at the time that you’re treating the culprit may be the way to go.
Parikh: All of us, as interventionalists, are circumspect about what we might do in the middle of the night versus what we would do in the light of day. Certainly it seems clear, particularly if it’s straightforward anatomy, that taking care of it in the index procedure is not only saving contrast and fluoroscopy time, but it’s also providing a clinical benefit to the patients. That’s something that will also impact how clinicians interpret these data. Previously, there was always a question about whether we should just do it in the same hospitalization or do it at the same time. I think now, increasingly, we’re emboldened to do more in the index procedure.
O’Donoghue: When you’re thinking about nonculprit lesions and which ones to treat, do you always make that determination based on physiologic guidance of some kind? Are you using instantaneous wave-free ratio? What’s your practice?
Parikh: In the acute setting, imaging is superior for at least the assessment of which is a culprit. If you see a ruptured atherothrombotic situation on optical coherence tomography, for example, that’s fairly convincing and definitive. In the absence of that physiology, we are taught to avoid in the infarct-related artery because of potential spuriously false-negative findings.
In this situation, certainly, an imaging subgroup probably would be helpful because some of the benefit is almost certainly derived from identifying the infarct-related artery by accident – in other words, doing what you thought was the nonculprit artery, which is, in fact, the culprit. I think that probably is part of this. As somebody who uses imaging in the overwhelming number of my cases, I think that imaging would be an important surrogate to this.
Index procedure versus staged
O’Donoghue: For the operator who is coming in to do their STEMI case at 2:00 in the morning, would these data now push you toward doing complete revascularization at that time of night, or do you think that there is wiggle room in terms of interpreting these results regarding timing, where as long as you were doing it before hospital discharge and not, let’s say, 30 days out, that you may be able to derive the same benefit? What are some of the pros and cons?
Parikh: There’s definitely a fatigue factor in the middle of the night if it’s a particularly arduous intervention for the index infarct-related artery. I think there’s a human element where it may make sense just to stop and then bring the patient back in the same hospitalization. It’s clear, though, that doing complete revascularization is better and doing it sooner is better. How soon one actually does it is a judgment call, as ever.
In our practice, we’ve been pushing ourselves to get most of the patients done in their index hospitalization. If you have a left-sided culprit, the left anterior descending artery, for example, and there’s a high-grade stenosis in the circumflex, it may make sense to take care of that in the same index procedure. If, on the other hand, it’s in the right coronary artery where you have to put a new guide in and spend more time, that may be a patient whom you stage. I think those nuances will come up as interventionalists look at the subgroup analysis data more carefully.
O’Donoghue: Those are great points, and I think they also underscore that we always need to think about what type of patient was enrolled in these studies. Certainly, if you have somebody with renal dysfunction, there might be more concern about giving them a large contrast load all in one sitting, albeit hard to know whether they do or not. But spacing that out by just a couple of days would really have a big impact.
Parikh: Very often in the STEMI patient, you don’t have the benefit of knowing the creatinine. The patient will come in immediately, if not directly from the ambulance to the cath lab, and there are no laboratories at all to work with. If the patient has never been seen in the system before, you won’t know. Again, in those situations, one may have pause, particularly if it’s an older patient. I think what’s reassuring, though, is that the data are supportive of being more aggressive earlier, and certainly this is the dataset that we were looking for.
O’Donoghue: To summarize, the two key takeaways are that, one, we now have more data to support a complete revascularization strategy and even extending that now to non-STEMI patients. Two, sooner appears to be better, so ideally, all done at the time of the index procedure. I think this is very interesting science and we’ll see how it changes practice.
Thanks for joining me today. Signing off for Medscape, this is Dr. Michelle O’Donoghue.
Michelle O’Donoghue is a cardiologist at Brigham and Women’s Hospital and senior investigator with the TIMI Study Group.
A version of this article first appeared on Medscape.com.
Navigating chronic cough in primary care
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
FROM ERS 2023
Seeking help for burnout may be a gamble for doctors
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.
By the end of 2021, Anuj Peddada, MD, had hit a wall. He couldn’t sleep, couldn’t concentrate, erupted in anger, and felt isolated personally and professionally. To temper pandemic-driven pressures, the Colorado radiation oncologist took an 8-week stress management and resiliency course, but the feelings kept creeping back.
Still, Dr. Peddada, in his own private practice, pushed through, working 60-hour weeks and carrying the workload of two physicians. It wasn’t until he caught himself making uncharacteristic medical errors, including radiation planning for the wrong site, that he knew he needed help – and possibly a temporary break from medicine.
There was just one hitch: He was closing his private practice to start a new in-house job with Centura Health, the Colorado Springs hospital he’d contracted with for over 20 years.
Given the long-standing relationship – Dr. Peddada’s image graced some of the company’s marketing billboards – he expected Centura would understand when, on his doctor’s recommendation, he requested a short-term medical leave that would delay his start date by 1 month.
Instead, Centura abruptly rescinded the employment offer, leaving Dr. Peddada jobless and with no recourse but to sue.
“I was blindsided. The hospital had a physician resiliency program that claimed to encourage physicians to seek help, [so] I thought they would be completely supportive and understanding,” Dr. Peddada said.
He told this news organization that he was naive to have been so honest with the hospital he’d long served as a contractor, including the decade-plus he›d spent directing its radiation oncology department.
“It is exceedingly painful to see hospital leadership use me in their advertisement[s] ... trying to profit off my reputation and work after devastating my career.”
The lawsuit Dr. Peddada filed in July in Colorado federal district court may offer a rare glimpse of the potential career ramifications of seeking help for physician burnout. Despite employers’ oft-stated support for physician wellness, Dr. Peddada’s experience may serve as a cautionary tale for doctors who are open about their struggles.
Centura Health did not respond to requests for comment. In court documents, the health system’s attorneys asked for more time to respond to Dr. Peddada’s complaint.
A plea for help
In the complaint, Dr. Peddada and his attorneys claim that Centura violated the state’s Anti-Discrimination Act and the Americans with Disabilities Act (ADA) when it failed to offer reasonable accommodations after he began experiencing “physiological and psychological symptoms corresponding to burnout.”
Since 1999, Dr. Peddada had contracted exclusively with Centura to provide oncology services at its hospital, Penrose Cancer Center, and began covering a second Centura location in 2021. As medical director of Penrose’s radiation oncology department, he helped establish a community nurse navigator program and accounted for 75% of Centura’s radiation oncology referrals, according to the complaint.
But when his symptoms and fear for the safety of his patients became unbearable, Dr. Peddada requested an urgent evaluation from his primary care physician, who diagnosed him with “physician burnout” and recommended medical leave.
Shortly after presenting the leave request to Centura, rumors began circulating that he was having a “nervous breakdown,” the complaint noted. Dr. Peddada worried that perhaps his private health information was being shared with hospital employees.
After meeting with the hospital’s head of physician resiliency and agreeing to undergo a peer review evaluation by the Colorado Physician Health Program, which would decide the reinstatement timeline and if further therapy was necessary, Dr. Peddada was assured his leave would be approved.
Five days later, his job offer was revoked.
In an email from hospital leadership, the oncologist was informed that he had “declined employment” by failing to sign a revised employment contract sent to him 2 weeks prior when he was out of state on a preapproved vacation, according to the lawsuit.
The lawsuit alleges that Dr. Peddada was wrongfully discharged due to his disability after Centura “exploited [his] extensive patient base, referral network, and reputation to generate growth and profit.”
Colorado employment law attorney Deborah Yim, Esq., who is not involved in Peddada’s case, told this news organization that the ADA requires employers to provide reasonable accommodations for physical or mental impairments that substantially limit at least one major life activity, except when the request imposes an undue hardship on the employer.
“Depression and related mental health conditions would qualify, depending on the circumstances, and courts have certainly found them to be qualifying disabilities entitled to ADA protection in the past,” she said.
Not all employers are receptive to doctors’ needs, says the leadership team at Physicians Just Equity, an organization providing peer support to doctors experiencing workplace conflicts like discrimination and retaliation. They say that Dr. Peddada’s experience, where disclosing burnout results in being “ostracized, penalized, and ultimately ousted,” is the rule rather than the exception.
“Dr. Peddada’s case represents the unfortunate reality faced by many physicians in today’s clinical landscape,” the organization’s board of directors said in a written statement. “The imbalance of unreasonable professional demands, the lack of autonomy, moral injury, and disintegrating practice rewards is unsustainable for the medical professional.”
“Retaliation by employers after speaking up against this imbalance [and] requesting support and time to rejuvenate is a grave failure of health care systems that prioritize the business of delivering health care over the health, well-being, and satisfaction of their most valuable resource – the physician,” the board added in their statement.
Dr. Peddada has since closed his private practice and works as an independent contractor and consultant, his attorney, Iris Halpern, JD, said in an interview. She says Centura could have honored the accommodation request or suggested another option that met his needs, but “not only were they unsupportive, they terminated him.”
Ms. Yim says the parties will have opportunities to reach a settlement and resolve the dispute as the case works through the court system. Otherwise, Dr. Peddada and Centura may eventually head to trial.
Current state of physician burnout
The state of physician burnout is certainly a concerning one. More than half (53%) of physicians responding to this year’s Medscape Physician Burnout & Depression Report said they are burned out. Nearly one-quarter reported feeling depressed. Some of the top reasons they cited were too many bureaucratic tasks (61%), too many work hours (37%), and lack of autonomy (31%).
A 2022 study by the Mayo Clinic found a substantial increase in physician burnout in the first 2 years of the pandemic, with doctors reporting rising emotional exhaustion and depersonalization.
Although burnout affects many physicians and is a priority focus of the National Academy of Medicine’s plan to restore workforce well-being, admitting it is often seen as taboo and can imperil a doctor’s career. In the Medscape report, for example, 39% of physicians said they would not even consider professional treatment for burnout, with many commenting that they would just deal with it themselves.
“Many physicians are frightened to take time out for self-care because [they] fear losing their job, being stigmatized, and potentially ending their careers,” said Dr. Peddada, adding that physicians are commonly asked questions about their mental health when applying for hospital privileges. He says this dynamic forces them to choose between getting help or ignoring their true feelings, leading to poor quality of care and patient safety risks.
Medical licensing boards probe physicians’ mental health, too. As part of its #FightingForDocs campaign, the American Medical Association hopes to remove the stigma around burnout and depression and advocates for licensing boards to revise questions that may discourage physicians from seeking assistance. The AMA recommends that physicians only disclose current physical or mental conditions affecting their ability to practice.
Pringl Miller, MD, founder and executive director of Physician Just Equity, told Medscape that improving physician wellness requires structural change.
“Physicians (who) experience burnout without the proper accommodations run the risk of personal harm, because most physicians will prioritize the health and well-being of their patients over themselves ... [resulting in] suboptimal and unsafe patient care,” she said.
Helping doctors regain a sense of purpose
One change involves reframing how the health care industry thinks about and approaches burnout, says Steven Siegel, MD, chief mental health and wellness officer with Keck Medicine of USC. He told this news organization that these discussions should enhance the physician’s sense of purpose.
“Some people treat burnout as a concrete disorder like cancer, instead of saying, ‘I’m feeling exhausted, demoralized, and don’t enjoy my job anymore. What can we do to restore my enthusiasm for work?’ ”
Dr. Siegel recognizes that these issues existed before the pandemic and have only worsened as physicians feel less connected to and satisfied with their profession – a byproduct, he says, of the commercialization of medicine.
“We’ve moved from practices to systems, then from small to large systems, where it seems the path to survival is cutting costs and increasing margins, even among nonprofits.”
The road ahead
Making headway on these problems will take time. Last year, Keck Medicine received a $2 million grant to launch a 3-year randomized clinical trial to help reconnect physicians and other clinicians with their work. Dr. Siegel says the trial may serve as a national pilot program and will eventually grow to include 400 volunteers.
The trial will investigate the effectiveness of three possible interventions: (1) teaching people how to regulate their internal narratives and emotions through techniques like cognitive behavioral therapy and acceptance and commitment therapy; (2) providing customized EHR training to reduce the burden of navigating the system; and (3) allowing physicians to weigh in on workflow changes.
“We put physicians on teams that make the decisions about workflows,” said Dr. Siegel. The arrangement can give people the agency they desire and help them understand why an idea might not be plausible, which enriches future suggestions and discussions, he says.
A version of this article first appeared on Medscape.com.