User login
COVID-19 outcomes no worse in patients on TNF inhibitors or methotrexate
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Psoriasis, PsA, and pregnancy: Tailoring treatment with increasing data
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
The long road to a PsA prevention trial
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”
About one-third of all patients with psoriasis will develop psoriatic arthritis (PsA), a condition that comes with a host of vague symptoms and no definitive blood test for diagnosis. Prevention trials could help to identify higher-risk groups for PsA, with a goal to catch disease early and improve outcomes. The challenge is finding enough participants in a disease that lacks a clear clinical profile, then tracking them for long periods of time to generate any significant data.
Researchers have been taking several approaches to improve outcomes in PsA, Christopher Ritchlin, MD, MPH, chief of the allergy/immunology and rheumatology division at the University of Rochester (N.Y.), said in an interview. “We are in the process of identifying biomarkers and imaging findings that characterize psoriasis patients at high risk to develop PsA.”
The next step would be to design an interventional trial to treat high-risk patients before they develop musculoskeletal inflammation, with a goal to delay onset, attenuate severity, or completely prevent arthritis. The issue now is “we don’t know which agents would be most effective in this prevention effort,” Dr. Ritchlin said. Biologics that target specific pathways significant in PsA pathogenesis are an appealing prospect. However, “it may be that alternative therapies such as methotrexate or ultraviolet A radiation therapy, for example, may help arrest the development of joint inflammation.”
Underdiagnosis impedes research
Several factors may undermine this important research.
For one, psoriasis patients are often unaware that they have PsA. “Many times they are diagnosed incorrectly by nonspecialists. As a consequence, they do not get access to appropriate medications,” said Lihi Eder, MD, PhD, staff rheumatologist and director of the psoriatic arthritis research program at the University of Toronto’s Women’s College Research Institute.
The condition also lacks a good diagnostic tool, Dr. Eder said. There’s no blood test that identifies this condition in the same manner as RA and lupus, for example. For these conditions, a general practitioner such as a family physician may conduct a blood test, and if positive, refer them to a rheumatologist. Such a system doesn’t exist for PsA. “Instead, nonspecialists are ordering tests and when they’re negative, they assume wrongly that these patients don’t have a rheumatic condition,” she explained.
Many clinicians aren’t that well versed in PsA, although dermatology has taken steps to become better educated. As a result, more dermatologists are referring patients to rheumatologists for PsA. Despite this small step forward, the heterogeneous clinical presentation of this condition makes diagnosis especially difficult. Unlike RA, which presents with inflammation in the joints, PsA can present as back or joint pain, “which makes our life as rheumatologists much more complex,” Dr. Eder said.
Defining a risk group
Most experts agree that the presence of psoriasis isn’t sufficient to conduct a prevention trial. Ideally, the goal of a prevention study would be to identify a critical subgroup of psoriasis patients at high risk of developing PsA.
However, this presents a challenging task, Dr. Eder said. Psoriasis is a risk factor for PsA, but most patients with psoriasis won’t actually develop it. Given that there’s an incidence rate of 2.7% a year, “you would need to recruit many hundreds of psoriasis patients and follow them for a long period of time until you have enough events.”
Moving forward with prevention studies calls for a better definition of the PsA risk group, according to Georg Schett, MD, chair of internal medicine in the department of internal medicine, rheumatology, and immunology at Friedrich‐Alexander University, Erlangen, Germany. “That’s very important, because you need to define such a group to make a prevention trial feasible. The whole benefit of such an approach is to catch the disease early, to say that psoriasis is a biomarker that’s linked to psoriatic arthritis.”
Indicators of risk other than psoriasis, such as pain, inflammation seen in ultrasound or MRI, and other specificities of psoriasis, could be used to define a population where interception can take place, Dr. Schett added. Although it’s not always clinically recognized, the combination of pain and structural lesions can be an indicator for developing PsA.
One prospective study he and his colleagues conducted in 114 psoriasis patients cited structural entheseal lesions and low cortical volumetric bone mineral density as risk factors in developing PsA. Keeping these factors in mind, Dr. Schett expects to see more studies in biointervention in these populations, “with the idea to prevent the onset of PsA and also decrease pain and subcutaneous inflammation.”
Researchers are currently working to identify those high-risk patients to include in an interventional trial, Dr. Ritchlin said.
That said, there’s been a great deal of “clinical trial angst” among investigators, Dr. Ritchlin noted. Outcomes in clinical trials for a wide range of biologic agents have not demonstrated significant advances in outcomes, compared with initial studies with anti–tumor necrosis factor–alpha (TNF-alpha) agents 20 years ago.
Combination biologics
One approach that’s generated some interest is the use of combination biologics medications. Sequential inhibition of cytokines such as interleukin-17A and TNF-alpha is of interest given their central contribution in joint inflammation and damage. “The challenge here of course is toxicity,” Dr. Ritchlin said. Trials that combined blockade of IL-1 and TNF-alpha in a RA trial years ago resulted in significant adverse events without improving outcomes.
Comparatively, a recent study in The Lancet Rheumatology reported success in using the IL-17A inhibitor secukinumab (Cosentyx) to reduce PsA symptoms. Tested on patients in the FUTURE 2 trial, investigators demonstrated that secukinumab in 300- and 150-mg doses safely reduced PsA signs and symptoms over a period of 5 years. Secukinumab also outperformed the TNF-alpha inhibitor adalimumab in 853 PsA patients in the 52-week, randomized, head-to-head, phase 3b EXCEED study, which was recently reported in The Lancet. Articular outcomes were similar between the two therapies, yet the secukinumab group did markedly better in Psoriasis Area and Severity Index scores, compared with the adalimumab group.
Based on these findings, “I suspect that studies examining the efficacy of combination biologics for treatment of PsA will surface in the near future,” Dr. Ritchlin said.
Yet another approach encompasses the spirit of personalized medicine. Clinicians often treat PsA patients empirically because they lack biomarkers that indicate which drug may be most effective for an individual patient, Dr. Ritchlin said. However, the technologies for investigating specific cell subsets in both the blood and tissues have advanced greatly over the last decade. “I am confident that a more precision medicine–based approach to the diagnosis and treatment of PsA is on the near horizon.”
Diet as an intervention
Other research has looked at the strong link between metabolic abnormalities and psoriasis and PsA. Some diets, such as the Mediterranean diet, show promise in improving the metabolic profile of these patients, making it a candidate as a potential intervention to reduce PsA risk. Another strategy would be to focus on limiting calories and promoting weight reduction.
One study in the British Journal of Dermatology looked at the associations between PsA and smoking, alcohol, and body mass index, identifying obesity as an important risk factor. Analyzing more than 90,000 psoriasis cases from the U.K. Clinical Practice Research Datalink between 1998 and 2014, researchers identified 1,409 PsA diagnoses. Among this cohort, researchers found an association between PsA and increased body mass index and moderate drinking. This finding underscores the need to support weight-reduction programs to reduce risk, Dr. Eder and Alexis Ogdie, MD, of the University of Pennsylvania, Philadelphia, wrote in a related editorial.
While observational studies such as this one provide further guidance for interventional trials, confounders can affect results. “Patients who lost weight could have made a positive lifestyle change (e.g., a dietary change) that was associated with the decreased risk for PsA rather than weight loss specifically, or they could have lost weight for unhealthy reasons,” Dr. Eder and Dr. Ogdie explained. Future research could address whether weight loss or other interventional factors may reduce PsA progression.
To make this work, “we would need to select patients that would benefit from diet. Secondly, we’d need to identify what kind of diet would be good for preventing PsA. And we don’t know that yet,” Dr. Eder further elaborated.
As with any prevention trial, the challenge is to follow patients over a long period of time, making sure they comply with the restrictions of the prescribed diet, Dr. Eder noted. “I do think it’s a really exciting type of intervention because it’s something that people are very interested in. There’s little risk of side effects, and it’s not very expensive.”
In other research on weight-loss methods, an observational study from Denmark found that bariatric surgery, especially gastric bypass, reduced the risk of developing PsA. This suggests that weight reduction by itself is important, “although we don’t know that yet,” Dr. Eder said.
A risk model for PsA
Dr. Eder and colleagues have been working on an algorithm that will incorporate clinical information (for example, the presence of nail lesions and the severity of psoriasis) to provide an estimated risk of developing PsA over the next 5 years. Subsequently, this information could be used to identify high-risk psoriasis patients as candidates for a prevention trial.
Other groups are looking at laboratory or imaging biomarkers to help develop PsA prediction models, she said. “Once we have these tools, we can move to next steps of prevention trials. What kinds of interventions should we apply? Are we talking biologic medications or other lifestyle interventions like diet? We are still at the early stages. However, with an improved understanding of the underlying mechanisms and risk factors we are expected to see prevention trials for PsA in the future.”
TNF inhibitors linked to inflammatory CNS events
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
Humira topped drug-revenue list for 2019
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.
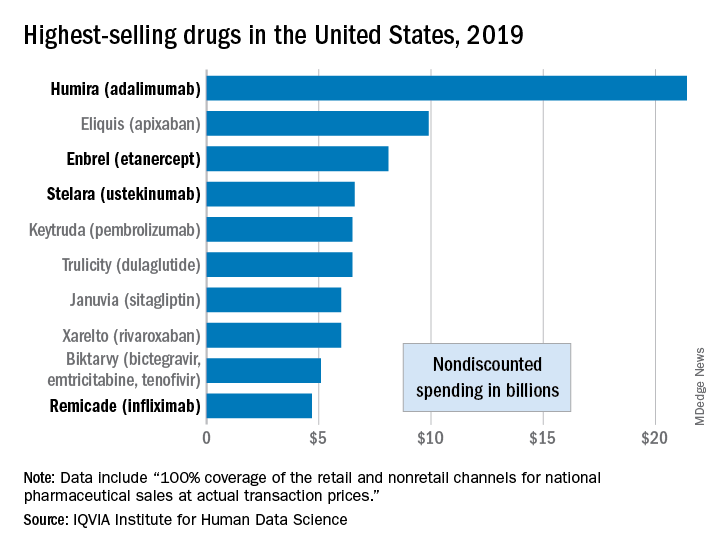
Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Beyond PASI 100: striving for molecular clearance
All PASI 100 responses to psoriasis therapy are not the same, Andrew Blauvelt, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
He presented a first-of-its-kind study that potentially opens the door to a new, more rigorous standard for treatment success in psoriasis: Not simply cleared lesional skin as captured by a Psoriasis Area and Severity Index (PASI) 100 response, but also clearance of residual psoriasis signs and symptoms – as well as what he termed “molecular clearance.”
“We’ve found that clearing skin with drugs utilizing different mechanisms of action may lead to differential consequences for our patients,” observed Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
A PASI 100 response, traditionally considered an elusive goal for the great majority of patients with severe psoriasis, can now often be achieved using today’s top-tier, high-performance biologics. But Dr. Blauvelt and his coinvestigators are interested in pushing even beyond PASI 100 to a new frontier of therapeutic benefit.
He presented a secondary analysis of the previously reported VOYAGE 1 and 2 head-to-head randomized trials of guselkumab (Tremfya) versus adalimumab (Humira) for treatment of moderate to severe psoriasis. This new analysis, which focused exclusively on PASI 100 responders by week 24, demonstrated that patients with a PASI 100 response to guselkumab, an interleukin (IL)-23 inhibitor, had significantly fewer persistent symptoms and signs of psoriasis than those whose skin clearance was attained using adalimumab, a tumor necrosis factor (TNF) inhibitor.
Moreover,
The analysis included 16 participants in the VOYAGE trials who achieved PASI 100 at week 24 on guselkumab and 5 who did so on adalimumab. At baseline and again at week 24, these individuals completed the Psoriasis Symptoms and Signs Diary (PSSD). Also, biopsies of lesional and nonlesional skin were obtained at baseline and of cleared lesional skin at week 24 for transcriptomic microarray analysis of the expression of many thousands of genes.
Persistent psoriasis symptoms despite cleared skin
The PSSD involves patient ratings of various psoriasis symptoms and signs. Total scores can range from 0 (symptom- and sign-free) up to 100. At week 24, a significantly higher proportion of guselkumab-treated PASI 100 responders had a total PSSD score of zero: 55%, versus 43% in the adalimumab group. This was consistently true across the board for each of the individual signs and symptoms assessed. For example, 61% of the guselkumab group gave themselves a zero for itch, as did 50% of the adalimumab group. Sixty-four percent on guselkumab and 52% on adalimumab reported being free of redness. And 78% of the guselkumab group reported being pain-free, compared with 69% with adalimumab, Dr. Blauvelt reported.
Gene expression analysis
At baseline, more than 2,300 dysregulated genes were identified in lesional skin while functioning normally in nonlesional skin. The great majority of these initially dysregulated genes became normalized in cleared lesional skin in PASI 100 responders at week 24. However, 25 of the genes remained dysregulated in cleared lesional skin, meaning they displayed less than 75% of normal function. Ten of these 25 genes with dysregulated expression at follow-up showed abnormal function in patients with residual symptoms despite cleared skin, but they functioned normally in those without persistent symptoms. This raises the possibility that the residual symptoms of psoriasis were attributable to the abnormal gene functioning, according to Dr. Blauvelt.
Of note, 9 of the 10 dysregulated genes in cleared lesional skin of patients with residual symptoms were present in the adalimumab group; these included two genes localized to the epidermal differentiation complex as well as the psoriasis-specific proline-rich 9 gene known as PRR9, which is induced by IL-17A. In contrast, only four genes, none of which were localized to the epidermal differentiation complex, were insufficiently normalized in the cleared lesional skin of guselkumab-treated PASI-100 responders.
“Nothing like this analysis has ever been done before,” the dermatologist observed. “It’s a pilot study. Perhaps with more data like this, we’ll be using this type of information in clinical practice to go beyond clearing patients’ skin.”
Dr. Blauvelt reported serving as a scientific advisor to and paid clinical investigator for Janssen, which sponsored the study, as well as for roughly two dozen other pharmaceutical companies.
All PASI 100 responses to psoriasis therapy are not the same, Andrew Blauvelt, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
He presented a first-of-its-kind study that potentially opens the door to a new, more rigorous standard for treatment success in psoriasis: Not simply cleared lesional skin as captured by a Psoriasis Area and Severity Index (PASI) 100 response, but also clearance of residual psoriasis signs and symptoms – as well as what he termed “molecular clearance.”
“We’ve found that clearing skin with drugs utilizing different mechanisms of action may lead to differential consequences for our patients,” observed Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
A PASI 100 response, traditionally considered an elusive goal for the great majority of patients with severe psoriasis, can now often be achieved using today’s top-tier, high-performance biologics. But Dr. Blauvelt and his coinvestigators are interested in pushing even beyond PASI 100 to a new frontier of therapeutic benefit.
He presented a secondary analysis of the previously reported VOYAGE 1 and 2 head-to-head randomized trials of guselkumab (Tremfya) versus adalimumab (Humira) for treatment of moderate to severe psoriasis. This new analysis, which focused exclusively on PASI 100 responders by week 24, demonstrated that patients with a PASI 100 response to guselkumab, an interleukin (IL)-23 inhibitor, had significantly fewer persistent symptoms and signs of psoriasis than those whose skin clearance was attained using adalimumab, a tumor necrosis factor (TNF) inhibitor.
Moreover,
The analysis included 16 participants in the VOYAGE trials who achieved PASI 100 at week 24 on guselkumab and 5 who did so on adalimumab. At baseline and again at week 24, these individuals completed the Psoriasis Symptoms and Signs Diary (PSSD). Also, biopsies of lesional and nonlesional skin were obtained at baseline and of cleared lesional skin at week 24 for transcriptomic microarray analysis of the expression of many thousands of genes.
Persistent psoriasis symptoms despite cleared skin
The PSSD involves patient ratings of various psoriasis symptoms and signs. Total scores can range from 0 (symptom- and sign-free) up to 100. At week 24, a significantly higher proportion of guselkumab-treated PASI 100 responders had a total PSSD score of zero: 55%, versus 43% in the adalimumab group. This was consistently true across the board for each of the individual signs and symptoms assessed. For example, 61% of the guselkumab group gave themselves a zero for itch, as did 50% of the adalimumab group. Sixty-four percent on guselkumab and 52% on adalimumab reported being free of redness. And 78% of the guselkumab group reported being pain-free, compared with 69% with adalimumab, Dr. Blauvelt reported.
Gene expression analysis
At baseline, more than 2,300 dysregulated genes were identified in lesional skin while functioning normally in nonlesional skin. The great majority of these initially dysregulated genes became normalized in cleared lesional skin in PASI 100 responders at week 24. However, 25 of the genes remained dysregulated in cleared lesional skin, meaning they displayed less than 75% of normal function. Ten of these 25 genes with dysregulated expression at follow-up showed abnormal function in patients with residual symptoms despite cleared skin, but they functioned normally in those without persistent symptoms. This raises the possibility that the residual symptoms of psoriasis were attributable to the abnormal gene functioning, according to Dr. Blauvelt.
Of note, 9 of the 10 dysregulated genes in cleared lesional skin of patients with residual symptoms were present in the adalimumab group; these included two genes localized to the epidermal differentiation complex as well as the psoriasis-specific proline-rich 9 gene known as PRR9, which is induced by IL-17A. In contrast, only four genes, none of which were localized to the epidermal differentiation complex, were insufficiently normalized in the cleared lesional skin of guselkumab-treated PASI-100 responders.
“Nothing like this analysis has ever been done before,” the dermatologist observed. “It’s a pilot study. Perhaps with more data like this, we’ll be using this type of information in clinical practice to go beyond clearing patients’ skin.”
Dr. Blauvelt reported serving as a scientific advisor to and paid clinical investigator for Janssen, which sponsored the study, as well as for roughly two dozen other pharmaceutical companies.
All PASI 100 responses to psoriasis therapy are not the same, Andrew Blauvelt, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
He presented a first-of-its-kind study that potentially opens the door to a new, more rigorous standard for treatment success in psoriasis: Not simply cleared lesional skin as captured by a Psoriasis Area and Severity Index (PASI) 100 response, but also clearance of residual psoriasis signs and symptoms – as well as what he termed “molecular clearance.”
“We’ve found that clearing skin with drugs utilizing different mechanisms of action may lead to differential consequences for our patients,” observed Dr. Blauvelt, a dermatologist and clinical trialist who is president of the Oregon Medical Research Center, Portland.
A PASI 100 response, traditionally considered an elusive goal for the great majority of patients with severe psoriasis, can now often be achieved using today’s top-tier, high-performance biologics. But Dr. Blauvelt and his coinvestigators are interested in pushing even beyond PASI 100 to a new frontier of therapeutic benefit.
He presented a secondary analysis of the previously reported VOYAGE 1 and 2 head-to-head randomized trials of guselkumab (Tremfya) versus adalimumab (Humira) for treatment of moderate to severe psoriasis. This new analysis, which focused exclusively on PASI 100 responders by week 24, demonstrated that patients with a PASI 100 response to guselkumab, an interleukin (IL)-23 inhibitor, had significantly fewer persistent symptoms and signs of psoriasis than those whose skin clearance was attained using adalimumab, a tumor necrosis factor (TNF) inhibitor.
Moreover,
The analysis included 16 participants in the VOYAGE trials who achieved PASI 100 at week 24 on guselkumab and 5 who did so on adalimumab. At baseline and again at week 24, these individuals completed the Psoriasis Symptoms and Signs Diary (PSSD). Also, biopsies of lesional and nonlesional skin were obtained at baseline and of cleared lesional skin at week 24 for transcriptomic microarray analysis of the expression of many thousands of genes.
Persistent psoriasis symptoms despite cleared skin
The PSSD involves patient ratings of various psoriasis symptoms and signs. Total scores can range from 0 (symptom- and sign-free) up to 100. At week 24, a significantly higher proportion of guselkumab-treated PASI 100 responders had a total PSSD score of zero: 55%, versus 43% in the adalimumab group. This was consistently true across the board for each of the individual signs and symptoms assessed. For example, 61% of the guselkumab group gave themselves a zero for itch, as did 50% of the adalimumab group. Sixty-four percent on guselkumab and 52% on adalimumab reported being free of redness. And 78% of the guselkumab group reported being pain-free, compared with 69% with adalimumab, Dr. Blauvelt reported.
Gene expression analysis
At baseline, more than 2,300 dysregulated genes were identified in lesional skin while functioning normally in nonlesional skin. The great majority of these initially dysregulated genes became normalized in cleared lesional skin in PASI 100 responders at week 24. However, 25 of the genes remained dysregulated in cleared lesional skin, meaning they displayed less than 75% of normal function. Ten of these 25 genes with dysregulated expression at follow-up showed abnormal function in patients with residual symptoms despite cleared skin, but they functioned normally in those without persistent symptoms. This raises the possibility that the residual symptoms of psoriasis were attributable to the abnormal gene functioning, according to Dr. Blauvelt.
Of note, 9 of the 10 dysregulated genes in cleared lesional skin of patients with residual symptoms were present in the adalimumab group; these included two genes localized to the epidermal differentiation complex as well as the psoriasis-specific proline-rich 9 gene known as PRR9, which is induced by IL-17A. In contrast, only four genes, none of which were localized to the epidermal differentiation complex, were insufficiently normalized in the cleared lesional skin of guselkumab-treated PASI-100 responders.
“Nothing like this analysis has ever been done before,” the dermatologist observed. “It’s a pilot study. Perhaps with more data like this, we’ll be using this type of information in clinical practice to go beyond clearing patients’ skin.”
Dr. Blauvelt reported serving as a scientific advisor to and paid clinical investigator for Janssen, which sponsored the study, as well as for roughly two dozen other pharmaceutical companies.
FROM AAD 20
Cohort study finds a twofold greater psoriasis risk linked to a PCOS diagnosis
PCOS is characterized by androgen elevation that can lead to insulin resistance and metabolic syndrome, which have also been associated with an increased risk of psoriasis. Previous retrospective analyses have suggested an increased risk of psoriasis associated with PCOS, and psoriasis patients with PCOS have been reported to have more severe skin lesions, compared with those who do not have PCOS.
“The incidence of psoriasis is indeed higher in the PCOS group than in the control group, and the comorbidities related to metabolic syndrome did not modify the adjusted hazard ratio,” said Ming-Li Chen, during her presentation of the study results at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. Dr. Chen is at Chung Shan Medical University in Taiwan.
The researchers analyzed 1 million randomly selected records from Taiwan’s Longitudinal Health Insurance database, a subset of the country’s National Health Insurance Program. Between 2000 and 2012, they identified a case group with at least three outpatient diagnoses or one inpatient diagnosis of PCOS; they then compared each with four patients who did not have PCOS who were matched by age and index year. The mean age in both groups was about 27 years.
The mean follow-up times were 6.99 years for 4,707 cases and 6.94 years for 18,828 controls. Comorbidities were slightly higher in the PCOS group, including asthma (6.7% vs. 4.9%; P less than .001), chronic obstructive pulmonary disease (14% vs. 11%; P less than .001), chronic liver disease (8.0% vs. 5.0%; P less than .001), diabetes mellitus (3.0% vs. 1.4%; P less than .001), hypertension (2.4% vs. 1.5%; P less than .001), hyperlipidemia (5.4% vs. 2.5%; P less than .001), depression (5.4% vs. 3.9%; P less than .001), and sleep apnea (0.23% vs. 0.10%; P = .040).
There was a higher cumulative incidence of psoriasis in the PCOS group (adjusted hazard ratio, 2.07; 95% confidence interval, 1.25-3.44). Other factors associated with increased risk of psoriasis were advanced age (greater than 50 years old; aHR, 14.13; 95% CI, 1.8-110.7) and having a cancer diagnosis (aHR, 11.72; 95% CI, 2.87-47.9).
When PCOS patients were stratified by age, the researchers noted a higher risk of psoriasis among those 20 years or younger (aHR, 4.02; 95% CI, 1.16-13.9) than among those aged 20-50 years (aHR, 1.88; 95% CI, 1.07-3.29). Among those older than 50 years, there was no significantly increased risk, although the number of psoriasis diagnoses and population sizes were small in the latter category. Among patients with PCOS, a cancer diagnosis was not associated with a statistically significant increased risk of psoriasis.
The mechanisms underlying the association between PCOS and psoriasis should be studied further, she noted.
Following Dr. Chen’s prerecorded presentation, there was a live discussion session led by Alice Gottlieb, MD, PhD, medical director of Mount Sinai Beth Israel Dermatology, New York, and Ennio Lubrano, MD, associate professor of rheumatology at the University of Molise (Italy). Dr. Gottlieb noted that the study did not appear to account for weight in the association between PCOS and psoriasis, since heavier people are known to be at greater risk of developing psoriasis. Dr. Chen acknowledged that the study had no records of BMI or weight.
Dr. Gottlieb also wondered if treatment of PCOS led to any improvements in psoriasis in patients with the two diagnoses. “If we treat PCOS, does the psoriasis get better?” Again, the study did not address the question. “We didn’t follow up on therapies,” Dr. Chen said.
Dr. Chen reported no relevant financial disclosures. Dr. Gottlieb is a consultant, advisory board member and/or speaker for AbbVie, Allergan, Avotres Therapeutics, Beiersdorf, Bristol-Myers Squibb, Celgene, Dermira, Eli Lilly, Incyte, Janssen, Leo, Novartis, Reddy Labs, Sun Pharmaceutical Industries, UCB Pharma and Xbiotech. She has received research or educational grants from Boehringer Ingelheim, Incyte, Janssen, Novartis and Xbiotech.
PCOS is characterized by androgen elevation that can lead to insulin resistance and metabolic syndrome, which have also been associated with an increased risk of psoriasis. Previous retrospective analyses have suggested an increased risk of psoriasis associated with PCOS, and psoriasis patients with PCOS have been reported to have more severe skin lesions, compared with those who do not have PCOS.
“The incidence of psoriasis is indeed higher in the PCOS group than in the control group, and the comorbidities related to metabolic syndrome did not modify the adjusted hazard ratio,” said Ming-Li Chen, during her presentation of the study results at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. Dr. Chen is at Chung Shan Medical University in Taiwan.
The researchers analyzed 1 million randomly selected records from Taiwan’s Longitudinal Health Insurance database, a subset of the country’s National Health Insurance Program. Between 2000 and 2012, they identified a case group with at least three outpatient diagnoses or one inpatient diagnosis of PCOS; they then compared each with four patients who did not have PCOS who were matched by age and index year. The mean age in both groups was about 27 years.
The mean follow-up times were 6.99 years for 4,707 cases and 6.94 years for 18,828 controls. Comorbidities were slightly higher in the PCOS group, including asthma (6.7% vs. 4.9%; P less than .001), chronic obstructive pulmonary disease (14% vs. 11%; P less than .001), chronic liver disease (8.0% vs. 5.0%; P less than .001), diabetes mellitus (3.0% vs. 1.4%; P less than .001), hypertension (2.4% vs. 1.5%; P less than .001), hyperlipidemia (5.4% vs. 2.5%; P less than .001), depression (5.4% vs. 3.9%; P less than .001), and sleep apnea (0.23% vs. 0.10%; P = .040).
There was a higher cumulative incidence of psoriasis in the PCOS group (adjusted hazard ratio, 2.07; 95% confidence interval, 1.25-3.44). Other factors associated with increased risk of psoriasis were advanced age (greater than 50 years old; aHR, 14.13; 95% CI, 1.8-110.7) and having a cancer diagnosis (aHR, 11.72; 95% CI, 2.87-47.9).
When PCOS patients were stratified by age, the researchers noted a higher risk of psoriasis among those 20 years or younger (aHR, 4.02; 95% CI, 1.16-13.9) than among those aged 20-50 years (aHR, 1.88; 95% CI, 1.07-3.29). Among those older than 50 years, there was no significantly increased risk, although the number of psoriasis diagnoses and population sizes were small in the latter category. Among patients with PCOS, a cancer diagnosis was not associated with a statistically significant increased risk of psoriasis.
The mechanisms underlying the association between PCOS and psoriasis should be studied further, she noted.
Following Dr. Chen’s prerecorded presentation, there was a live discussion session led by Alice Gottlieb, MD, PhD, medical director of Mount Sinai Beth Israel Dermatology, New York, and Ennio Lubrano, MD, associate professor of rheumatology at the University of Molise (Italy). Dr. Gottlieb noted that the study did not appear to account for weight in the association between PCOS and psoriasis, since heavier people are known to be at greater risk of developing psoriasis. Dr. Chen acknowledged that the study had no records of BMI or weight.
Dr. Gottlieb also wondered if treatment of PCOS led to any improvements in psoriasis in patients with the two diagnoses. “If we treat PCOS, does the psoriasis get better?” Again, the study did not address the question. “We didn’t follow up on therapies,” Dr. Chen said.
Dr. Chen reported no relevant financial disclosures. Dr. Gottlieb is a consultant, advisory board member and/or speaker for AbbVie, Allergan, Avotres Therapeutics, Beiersdorf, Bristol-Myers Squibb, Celgene, Dermira, Eli Lilly, Incyte, Janssen, Leo, Novartis, Reddy Labs, Sun Pharmaceutical Industries, UCB Pharma and Xbiotech. She has received research or educational grants from Boehringer Ingelheim, Incyte, Janssen, Novartis and Xbiotech.
PCOS is characterized by androgen elevation that can lead to insulin resistance and metabolic syndrome, which have also been associated with an increased risk of psoriasis. Previous retrospective analyses have suggested an increased risk of psoriasis associated with PCOS, and psoriasis patients with PCOS have been reported to have more severe skin lesions, compared with those who do not have PCOS.
“The incidence of psoriasis is indeed higher in the PCOS group than in the control group, and the comorbidities related to metabolic syndrome did not modify the adjusted hazard ratio,” said Ming-Li Chen, during her presentation of the study results at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. Dr. Chen is at Chung Shan Medical University in Taiwan.
The researchers analyzed 1 million randomly selected records from Taiwan’s Longitudinal Health Insurance database, a subset of the country’s National Health Insurance Program. Between 2000 and 2012, they identified a case group with at least three outpatient diagnoses or one inpatient diagnosis of PCOS; they then compared each with four patients who did not have PCOS who were matched by age and index year. The mean age in both groups was about 27 years.
The mean follow-up times were 6.99 years for 4,707 cases and 6.94 years for 18,828 controls. Comorbidities were slightly higher in the PCOS group, including asthma (6.7% vs. 4.9%; P less than .001), chronic obstructive pulmonary disease (14% vs. 11%; P less than .001), chronic liver disease (8.0% vs. 5.0%; P less than .001), diabetes mellitus (3.0% vs. 1.4%; P less than .001), hypertension (2.4% vs. 1.5%; P less than .001), hyperlipidemia (5.4% vs. 2.5%; P less than .001), depression (5.4% vs. 3.9%; P less than .001), and sleep apnea (0.23% vs. 0.10%; P = .040).
There was a higher cumulative incidence of psoriasis in the PCOS group (adjusted hazard ratio, 2.07; 95% confidence interval, 1.25-3.44). Other factors associated with increased risk of psoriasis were advanced age (greater than 50 years old; aHR, 14.13; 95% CI, 1.8-110.7) and having a cancer diagnosis (aHR, 11.72; 95% CI, 2.87-47.9).
When PCOS patients were stratified by age, the researchers noted a higher risk of psoriasis among those 20 years or younger (aHR, 4.02; 95% CI, 1.16-13.9) than among those aged 20-50 years (aHR, 1.88; 95% CI, 1.07-3.29). Among those older than 50 years, there was no significantly increased risk, although the number of psoriasis diagnoses and population sizes were small in the latter category. Among patients with PCOS, a cancer diagnosis was not associated with a statistically significant increased risk of psoriasis.
The mechanisms underlying the association between PCOS and psoriasis should be studied further, she noted.
Following Dr. Chen’s prerecorded presentation, there was a live discussion session led by Alice Gottlieb, MD, PhD, medical director of Mount Sinai Beth Israel Dermatology, New York, and Ennio Lubrano, MD, associate professor of rheumatology at the University of Molise (Italy). Dr. Gottlieb noted that the study did not appear to account for weight in the association between PCOS and psoriasis, since heavier people are known to be at greater risk of developing psoriasis. Dr. Chen acknowledged that the study had no records of BMI or weight.
Dr. Gottlieb also wondered if treatment of PCOS led to any improvements in psoriasis in patients with the two diagnoses. “If we treat PCOS, does the psoriasis get better?” Again, the study did not address the question. “We didn’t follow up on therapies,” Dr. Chen said.
Dr. Chen reported no relevant financial disclosures. Dr. Gottlieb is a consultant, advisory board member and/or speaker for AbbVie, Allergan, Avotres Therapeutics, Beiersdorf, Bristol-Myers Squibb, Celgene, Dermira, Eli Lilly, Incyte, Janssen, Leo, Novartis, Reddy Labs, Sun Pharmaceutical Industries, UCB Pharma and Xbiotech. She has received research or educational grants from Boehringer Ingelheim, Incyte, Janssen, Novartis and Xbiotech.
FROM GRAPPA 2020 VIRTUAL ANNUAL MEETING
New psoriasis guidelines focus on topical and alternative treatments, and severity measures
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
and the National Psoriasis Foundation.
The guidelines, published in the Journal of the American Academy of Dermatology, focus on treatment for adults, and follow the release of other AAD-NPF guidelines on biologics for psoriasis, psoriasis-related comorbidities, pediatric psoriasis, and phototherapy in 2019, and earlier this year, guidelines for systemic nonbiologic treatments. The latest guidelines’ section on topical treatment outlines evidence for the efficacy, effectiveness, and adverse events related to topical steroids, topical tacrolimus and pimecrolimus, vitamin D analogues, tazarotene, moisturizers, salicylic acid, anthralin, coal tar, combinations with biologic agents, and combinations with nonbiologic treatments (methotrexate, cyclosporine, acitretin, and apremilast).
The guidelines noted the “key role” of topical corticosteroids in treating psoriasis “especially for localized disease,” and include a review of the data on low-, moderate-, high-, and ultrahigh-potency topical steroids for psoriasis.
In general, all topical steroids can be used in combination with biologics, according to the guidelines, but the strongest recommendations based on the latest evidence include the addition of an ultra-high potency topical corticosteroid to standard dose etanercept for 12 weeks. Currently, 11 biologics are approved by the Food and Drug Administration for the treatment of psoriasis.
In addition, “while not FDA approved for psoriasis, the topical calcineurin inhibitors tacrolimus and pimecrolimus are often employed in the treatment of psoriasis,” can be helpful for “thinner skin such as facial and intertriginous areas,” and can be steroid sparing when used for more than 4 weeks, according to the guidelines.
Don’t discount the role of patient preferences when choosing topical treatments, the authors noted. “The optimal vehicle choice is the one the patient is mostly likely to use.”
The guidelines also address the evidence for effectiveness, and adverse events in the use of several alternative medicines for psoriasis including traditional Chinese medicine, and the herbal therapies aloe vera and St. John’s wort, as well as the potential role of dietary supplements including fish oil, vitamin D, turmeric, and zinc in managing psoriasis, and the potential role of a gluten-free diet.
In general, research on the efficacy, effectiveness, and potential adverse effects of these strategies are limited, according to the guidelines, although many patients express interest in supplements and herbal products. For example, “Many patients ask about the overall role of vitamin D in skin health. Rather than adding oral vitamin D supplementation, topical therapy with vitamin D agents is effective for the treatment of psoriasis,” the authors noted.
In addition, they noted that mind/body strategies, namely hypnosis and stress reduction or meditation techniques, have been shown to improve symptoms and can be helpful for some patients, but clinical evidence is limited.
The guidelines also addressed methods for assessing disease severity in psoriasis. They recommended using body surface area (BSA) to assess psoriasis severity and patient response to treatment in the clinical setting. However, BSA is a provider assessment tool that “does not take into account location on the body, clinical characteristics of the plaques, symptoms, or quality of life issues,” the authors noted. The Psoriasis Area and Severity Index (PASI) measures erythema, induration, and scaling and is more suited to assessing psoriasis severity and response to treatment in clinical trials rather than in practice, they said.
Prior AAD guidelines on psoriasis were published more than 10 years ago, and major developments including the availability of new biologic drugs and new data on comorbidities have been recognized in the past decade, working group cochair and author of the guidelines Alan Menter, MD, said in an interview.
The key game-changers from previous guidelines include the full section published on comorbidities plus the development of two new important cytokine classes: three IL-17 drugs and three new IL-23 drugs now available for moderate to severe psoriasis, said Dr. Menter, chairman of the division of dermatology at Baylor University Medical Center, Dallas.
Barriers to implementing the guidelines in practice may occur when “third party payers make the decision on which of the 11 biologic drugs now approved for moderate to severe psoriasis should be used,” he noted.
As for next steps in psoriasis studies, “new biomarker research is currently underway,” Dr. Menter said. With 11 biologic agents new formally approved by the FDA for moderate to severe psoriasis, the next steps are to determine which drug is likely to be the most appropriate for each individual patient.
Dr. Menter disclosed relationships with multiple companies that develop and manufacture psoriasis therapies, including Abbott Labs, AbbVie, Amgen, Eli Lilly and Company, Galderma USA, Janssen Pharmaceuticals, LEO Pharma US, Menlo Therapeutics, and Novartis. The updated guidelines were designed by a multidisciplinary work group of psoriasis experts including dermatologists, a rheumatologist, a cardiologist, and representatives from a patient advocacy organization.
SOURCE: Elmets CA et al. J Am Acad Dermatol. 2020 Jul 29. doi: 10.1016/j.jaad.2020.07.087.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Biologics may delay psoriatic arthritis, study finds
(DMARDs), in a single center retrospective analysis in Argentina that followed patients for almost 2 decades.
About 30%-40% of patients with psoriasis go on to develop psoriatic arthritis (PsA), usually on average about 10 years after the onset of psoriasis. One potential mechanism of PsA onset is through enthesitis, which has been described at subclinical levels in psoriasis.
“It could be speculated that treatment with biologics in patients with psoriasis could prevent the development of psoriatic arthritis, perhaps by inhibiting the subclinical development of enthesitis,” Luciano Lo Giudice, MD, a rheumatology fellow at Hospital Italiano de Buenos Aires, said during his presentation at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Although these results do not prove that treatment of the underlying disease delays progression to PsA, it is suggestive, and highlights an emerging field of research, according to Diamant Thaçi, MD, PhD, professor of medicine at University Hospital Schleswig-Holstein, Germany, who led a live discussion following a prerecorded presentation of the results. “We’re going in this direction – how can we prevent psoriatic arthritis, how can we delay it. We are just starting to think about this,” Dr. Thaçi said in an interview.
The researchers examined medical records of 1,626 patients with psoriasis treated at their center between 2000 and 2019, with a total of 15,152 years of follow-up. Of these patients, 1,293 were treated with topical medication, 229 with conventional DMARDs (methotrexate in 77%, cyclosporine in 13%, and both in 10%), and 104 with biologics, including etanercept (34%), secukinumab (20%), adalimumab (20%), ustekinumab (12%), ixekizumab (9%), and infliximab (5%).
They found that 11% in the topical treatment group developed PsA, as did 3.5% in the conventional DMARD group, 1.9% in the biologics group, and 9.1% overall. Treatment with biologics was associated with a significantly lower odds of developing PsA compared with treatment with conventional DMARDs (3 versus 17.2 per 1,000 patient-years; incidence rate ratio [IRR], 0.17; P = .0177). There was a trend toward reduced odds of developing PsA among those on biologic therapy compared with those on topicals (3 versus 9.8 per 1,000 patient-years; IRR, 0.3; P = .0588).
The researchers confirmed all medical encounters using electronic medical records and the study had a long follow-up time, but was limited by the single center and its retrospective nature. It also could not associate reduced risk with specific biologics.
The findings probably reflect the presence of subclinical PsA that many clinicians don’t see, according to Dr. Thaçi. While a dermatology practice might find PsA in 2% or 3%, or at most, 10% of patients with psoriasis, “in our department it’s about 50 to 60 percent of patients who have psoriatic arthritis, because we diagnose it early,” he said.
He found the results of the study encouraging. “It looks like some of the biologics, for example IL [interleukin]-17 or even IL-23 [blockers] may have an influence on occurrence or delay the occurrence of psoriatic arthritis.”
Dr. Thaçi noted that early treatment of skin lesions can increase the probability of longer remissions, especially with IL-23 blockers. Still, that’s no guarantee the same would hold true for PsA risk. “Skin is skin and joints are joints,” Dr. Thaçi said.
Dr. Thaçi and Dr. Lo Giudice had no relevant financial disclosures.
(DMARDs), in a single center retrospective analysis in Argentina that followed patients for almost 2 decades.
About 30%-40% of patients with psoriasis go on to develop psoriatic arthritis (PsA), usually on average about 10 years after the onset of psoriasis. One potential mechanism of PsA onset is through enthesitis, which has been described at subclinical levels in psoriasis.
“It could be speculated that treatment with biologics in patients with psoriasis could prevent the development of psoriatic arthritis, perhaps by inhibiting the subclinical development of enthesitis,” Luciano Lo Giudice, MD, a rheumatology fellow at Hospital Italiano de Buenos Aires, said during his presentation at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Although these results do not prove that treatment of the underlying disease delays progression to PsA, it is suggestive, and highlights an emerging field of research, according to Diamant Thaçi, MD, PhD, professor of medicine at University Hospital Schleswig-Holstein, Germany, who led a live discussion following a prerecorded presentation of the results. “We’re going in this direction – how can we prevent psoriatic arthritis, how can we delay it. We are just starting to think about this,” Dr. Thaçi said in an interview.
The researchers examined medical records of 1,626 patients with psoriasis treated at their center between 2000 and 2019, with a total of 15,152 years of follow-up. Of these patients, 1,293 were treated with topical medication, 229 with conventional DMARDs (methotrexate in 77%, cyclosporine in 13%, and both in 10%), and 104 with biologics, including etanercept (34%), secukinumab (20%), adalimumab (20%), ustekinumab (12%), ixekizumab (9%), and infliximab (5%).
They found that 11% in the topical treatment group developed PsA, as did 3.5% in the conventional DMARD group, 1.9% in the biologics group, and 9.1% overall. Treatment with biologics was associated with a significantly lower odds of developing PsA compared with treatment with conventional DMARDs (3 versus 17.2 per 1,000 patient-years; incidence rate ratio [IRR], 0.17; P = .0177). There was a trend toward reduced odds of developing PsA among those on biologic therapy compared with those on topicals (3 versus 9.8 per 1,000 patient-years; IRR, 0.3; P = .0588).
The researchers confirmed all medical encounters using electronic medical records and the study had a long follow-up time, but was limited by the single center and its retrospective nature. It also could not associate reduced risk with specific biologics.
The findings probably reflect the presence of subclinical PsA that many clinicians don’t see, according to Dr. Thaçi. While a dermatology practice might find PsA in 2% or 3%, or at most, 10% of patients with psoriasis, “in our department it’s about 50 to 60 percent of patients who have psoriatic arthritis, because we diagnose it early,” he said.
He found the results of the study encouraging. “It looks like some of the biologics, for example IL [interleukin]-17 or even IL-23 [blockers] may have an influence on occurrence or delay the occurrence of psoriatic arthritis.”
Dr. Thaçi noted that early treatment of skin lesions can increase the probability of longer remissions, especially with IL-23 blockers. Still, that’s no guarantee the same would hold true for PsA risk. “Skin is skin and joints are joints,” Dr. Thaçi said.
Dr. Thaçi and Dr. Lo Giudice had no relevant financial disclosures.
(DMARDs), in a single center retrospective analysis in Argentina that followed patients for almost 2 decades.
About 30%-40% of patients with psoriasis go on to develop psoriatic arthritis (PsA), usually on average about 10 years after the onset of psoriasis. One potential mechanism of PsA onset is through enthesitis, which has been described at subclinical levels in psoriasis.
“It could be speculated that treatment with biologics in patients with psoriasis could prevent the development of psoriatic arthritis, perhaps by inhibiting the subclinical development of enthesitis,” Luciano Lo Giudice, MD, a rheumatology fellow at Hospital Italiano de Buenos Aires, said during his presentation at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Although these results do not prove that treatment of the underlying disease delays progression to PsA, it is suggestive, and highlights an emerging field of research, according to Diamant Thaçi, MD, PhD, professor of medicine at University Hospital Schleswig-Holstein, Germany, who led a live discussion following a prerecorded presentation of the results. “We’re going in this direction – how can we prevent psoriatic arthritis, how can we delay it. We are just starting to think about this,” Dr. Thaçi said in an interview.
The researchers examined medical records of 1,626 patients with psoriasis treated at their center between 2000 and 2019, with a total of 15,152 years of follow-up. Of these patients, 1,293 were treated with topical medication, 229 with conventional DMARDs (methotrexate in 77%, cyclosporine in 13%, and both in 10%), and 104 with biologics, including etanercept (34%), secukinumab (20%), adalimumab (20%), ustekinumab (12%), ixekizumab (9%), and infliximab (5%).
They found that 11% in the topical treatment group developed PsA, as did 3.5% in the conventional DMARD group, 1.9% in the biologics group, and 9.1% overall. Treatment with biologics was associated with a significantly lower odds of developing PsA compared with treatment with conventional DMARDs (3 versus 17.2 per 1,000 patient-years; incidence rate ratio [IRR], 0.17; P = .0177). There was a trend toward reduced odds of developing PsA among those on biologic therapy compared with those on topicals (3 versus 9.8 per 1,000 patient-years; IRR, 0.3; P = .0588).
The researchers confirmed all medical encounters using electronic medical records and the study had a long follow-up time, but was limited by the single center and its retrospective nature. It also could not associate reduced risk with specific biologics.
The findings probably reflect the presence of subclinical PsA that many clinicians don’t see, according to Dr. Thaçi. While a dermatology practice might find PsA in 2% or 3%, or at most, 10% of patients with psoriasis, “in our department it’s about 50 to 60 percent of patients who have psoriatic arthritis, because we diagnose it early,” he said.
He found the results of the study encouraging. “It looks like some of the biologics, for example IL [interleukin]-17 or even IL-23 [blockers] may have an influence on occurrence or delay the occurrence of psoriatic arthritis.”
Dr. Thaçi noted that early treatment of skin lesions can increase the probability of longer remissions, especially with IL-23 blockers. Still, that’s no guarantee the same would hold true for PsA risk. “Skin is skin and joints are joints,” Dr. Thaçi said.
Dr. Thaçi and Dr. Lo Giudice had no relevant financial disclosures.
FROM GRAPPA 2020 VIRTUAL ANNUAL MEETING
New developments in pustular psoriasis
It has various dermatologic and rheumatologic manifestations and sometimes overlaps with plaque psoriasis. Pustular palmoplantar psoriasis (PPP) affects the palmar and plantar areas of the skin, while generalized pustular psoriasis (GPP) can affect large areas of skin and tends to be more severe, even life threatening. PPP can accompany psoriatic arthritis or can be a side effect of tumor necrosis factor (TNF) inhibitor therapy, or a non–drug-induced component of rheumatologic syndromes, according to Kristina Callis Duffin, MD, an associate professor and chair of dermatology at the University of Utah, Salt Lake City.
“Each phenotype could be considered an orphan disease, and the response to therapy is often unpredictable,” Dr. Duffin said during a session on pustular psoriasis at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
But there is some positive news. A study in 2011 of several people with GPP opened the door to better understanding the pathophysiology of pustular psoriasis. Researchers identified a causal autosomal mutation in the IL36RN gene, which encodes an antagonist to the interleukin-36 receptor (Am J Hum Genet. 2011 Sep 9;89[3]:432-7). “As a result of this paper and others, drug development in this space has recently accelerated,” Dr. Duffin said.
In fact, she added,“it’s my opinion that pustular psoriasis is now where plaque psoriasis was 20 years ago, when accelerated drug development was driving a better understanding of the pathogenesis of psoriatic disease and its comorbidities, and also driving outcome measure development.”
In another presentation at the meeting, Hervé Bachelez, MD, PhD, professor of dermatology and immunologist at the University of Paris and Saint-Louis Hospital, Paris, discussed recent advances in drug development for pustular psoriasis. He noted other recent findings of genetic variants related to the disease, including AP1S3, CARD14, and SERPINA3.
For GPP, he said, the current algorithm for management is based on weak evidence for treatments like acitretin, cyclosporine, methotrexate, and infliximab. The story is similar for other biologics, with evidence in the form of case series; open-label studies; controlled, prospective studies; or retrospective analyses. Most of the evidence has been amassed for TNF inhibitors. A retrospective study of all TNF inhibitors suggested they may be effective as induction and maintenance therapy, he noted.
Among IL-17A inhibitors, a prospective study of 12 patients in Japan found secukinumab showed efficacy against GPP, as did studies of ixekizumab and brodalumab. A small phase 3 study in Japan demonstrated efficacy for the IL-23 inhibitor guselkumab in patients with erythrodermic psoriasis and GPP (J Dermatol. 2018 May;45[5]:529-39).
The limited data are a reflection in part of the difficulty in studying GPP, since its flares tend to be more self-remitting than with psoriasis vulgaris or PPP.
There are two monoclonal antibodies against the IL-36 receptor currently being developed. A proof-of-concept study of one of them, spesolimab, showed promise against GPP, with five of seven patients reaching “clear” or “almost clear” scores on the Generalized Pustular Psoriasis Physician Global Assessment within a week after infusion and in all seven by the fourth week (N Engl J Med. 2019 Mar 7;380[10]:981-3).
With respect to PPP, the strongest evidence for conventional therapies comes from two randomized, controlled trials of cyclosporine, with response rates of 48% and 89%, compared with 19% and 21%, respectively, in the placebo groups, although the primary endpoint was poorly designed, according to Dr. Bachelez. Retinoids like etretinate and acitretin, combined with psoralen and UVA, also have some supporting evidence regarding efficacy.
Among biologics, secukinumab did not fare well in a phase 3 study of patients with PPP. A subset of patients may benefit from it, but there are no biomarkers available to identify them, Dr. Bachelez said. A phase 2 study of guselkumab in Japan told a similar story, with only weak signs of efficacy. While there are many more ongoing clinical trials evaluating treatments for PPP, which is encouraging, PPP seems to be more challenging at this stage to tackle than GPP, Dr. Bachelez added. “The genetically inherited IL-36 antagonist abnormalities are clearly driving the advances regarding the pathogenesis of the disease, mainly for GPP rather than PPP.”
Part of the efforts to develop therapies for pustular psoriasis relies on the development of new outcome measures, or adaptation of existing ones. “We have a need to adapt or develop new investigator-reported measures, we need to adapt or develop new patient-reported outcomes,” Dr. Duffin said.
Many existing measures use inconsistent language and anchoring definitions, and some may be proprietary, she added. “The language varies by sponsor and is sometimes tweaked or modified by the agencies. Often synonyms are being used … it raises questions, does it change the validity of the instrument?”
Dr. Duffin called for the research community to use the pause in clinical research during the COVID-19 pandemic to reassess the research agenda, develop consensus on performing and training for GPP and PPP assessments, develop patient-reported outcomes, and strengthen connections to industry.
Dr. Duffin and Dr. Bachelez have consulted, served on the advisory board, been a speaker for, and/or received research support from a wide range of pharmaceutical companies, including those that manufacture and develop psoriasis treatments.
It has various dermatologic and rheumatologic manifestations and sometimes overlaps with plaque psoriasis. Pustular palmoplantar psoriasis (PPP) affects the palmar and plantar areas of the skin, while generalized pustular psoriasis (GPP) can affect large areas of skin and tends to be more severe, even life threatening. PPP can accompany psoriatic arthritis or can be a side effect of tumor necrosis factor (TNF) inhibitor therapy, or a non–drug-induced component of rheumatologic syndromes, according to Kristina Callis Duffin, MD, an associate professor and chair of dermatology at the University of Utah, Salt Lake City.
“Each phenotype could be considered an orphan disease, and the response to therapy is often unpredictable,” Dr. Duffin said during a session on pustular psoriasis at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
But there is some positive news. A study in 2011 of several people with GPP opened the door to better understanding the pathophysiology of pustular psoriasis. Researchers identified a causal autosomal mutation in the IL36RN gene, which encodes an antagonist to the interleukin-36 receptor (Am J Hum Genet. 2011 Sep 9;89[3]:432-7). “As a result of this paper and others, drug development in this space has recently accelerated,” Dr. Duffin said.
In fact, she added,“it’s my opinion that pustular psoriasis is now where plaque psoriasis was 20 years ago, when accelerated drug development was driving a better understanding of the pathogenesis of psoriatic disease and its comorbidities, and also driving outcome measure development.”
In another presentation at the meeting, Hervé Bachelez, MD, PhD, professor of dermatology and immunologist at the University of Paris and Saint-Louis Hospital, Paris, discussed recent advances in drug development for pustular psoriasis. He noted other recent findings of genetic variants related to the disease, including AP1S3, CARD14, and SERPINA3.
For GPP, he said, the current algorithm for management is based on weak evidence for treatments like acitretin, cyclosporine, methotrexate, and infliximab. The story is similar for other biologics, with evidence in the form of case series; open-label studies; controlled, prospective studies; or retrospective analyses. Most of the evidence has been amassed for TNF inhibitors. A retrospective study of all TNF inhibitors suggested they may be effective as induction and maintenance therapy, he noted.
Among IL-17A inhibitors, a prospective study of 12 patients in Japan found secukinumab showed efficacy against GPP, as did studies of ixekizumab and brodalumab. A small phase 3 study in Japan demonstrated efficacy for the IL-23 inhibitor guselkumab in patients with erythrodermic psoriasis and GPP (J Dermatol. 2018 May;45[5]:529-39).
The limited data are a reflection in part of the difficulty in studying GPP, since its flares tend to be more self-remitting than with psoriasis vulgaris or PPP.
There are two monoclonal antibodies against the IL-36 receptor currently being developed. A proof-of-concept study of one of them, spesolimab, showed promise against GPP, with five of seven patients reaching “clear” or “almost clear” scores on the Generalized Pustular Psoriasis Physician Global Assessment within a week after infusion and in all seven by the fourth week (N Engl J Med. 2019 Mar 7;380[10]:981-3).
With respect to PPP, the strongest evidence for conventional therapies comes from two randomized, controlled trials of cyclosporine, with response rates of 48% and 89%, compared with 19% and 21%, respectively, in the placebo groups, although the primary endpoint was poorly designed, according to Dr. Bachelez. Retinoids like etretinate and acitretin, combined with psoralen and UVA, also have some supporting evidence regarding efficacy.
Among biologics, secukinumab did not fare well in a phase 3 study of patients with PPP. A subset of patients may benefit from it, but there are no biomarkers available to identify them, Dr. Bachelez said. A phase 2 study of guselkumab in Japan told a similar story, with only weak signs of efficacy. While there are many more ongoing clinical trials evaluating treatments for PPP, which is encouraging, PPP seems to be more challenging at this stage to tackle than GPP, Dr. Bachelez added. “The genetically inherited IL-36 antagonist abnormalities are clearly driving the advances regarding the pathogenesis of the disease, mainly for GPP rather than PPP.”
Part of the efforts to develop therapies for pustular psoriasis relies on the development of new outcome measures, or adaptation of existing ones. “We have a need to adapt or develop new investigator-reported measures, we need to adapt or develop new patient-reported outcomes,” Dr. Duffin said.
Many existing measures use inconsistent language and anchoring definitions, and some may be proprietary, she added. “The language varies by sponsor and is sometimes tweaked or modified by the agencies. Often synonyms are being used … it raises questions, does it change the validity of the instrument?”
Dr. Duffin called for the research community to use the pause in clinical research during the COVID-19 pandemic to reassess the research agenda, develop consensus on performing and training for GPP and PPP assessments, develop patient-reported outcomes, and strengthen connections to industry.
Dr. Duffin and Dr. Bachelez have consulted, served on the advisory board, been a speaker for, and/or received research support from a wide range of pharmaceutical companies, including those that manufacture and develop psoriasis treatments.
It has various dermatologic and rheumatologic manifestations and sometimes overlaps with plaque psoriasis. Pustular palmoplantar psoriasis (PPP) affects the palmar and plantar areas of the skin, while generalized pustular psoriasis (GPP) can affect large areas of skin and tends to be more severe, even life threatening. PPP can accompany psoriatic arthritis or can be a side effect of tumor necrosis factor (TNF) inhibitor therapy, or a non–drug-induced component of rheumatologic syndromes, according to Kristina Callis Duffin, MD, an associate professor and chair of dermatology at the University of Utah, Salt Lake City.
“Each phenotype could be considered an orphan disease, and the response to therapy is often unpredictable,” Dr. Duffin said during a session on pustular psoriasis at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
But there is some positive news. A study in 2011 of several people with GPP opened the door to better understanding the pathophysiology of pustular psoriasis. Researchers identified a causal autosomal mutation in the IL36RN gene, which encodes an antagonist to the interleukin-36 receptor (Am J Hum Genet. 2011 Sep 9;89[3]:432-7). “As a result of this paper and others, drug development in this space has recently accelerated,” Dr. Duffin said.
In fact, she added,“it’s my opinion that pustular psoriasis is now where plaque psoriasis was 20 years ago, when accelerated drug development was driving a better understanding of the pathogenesis of psoriatic disease and its comorbidities, and also driving outcome measure development.”
In another presentation at the meeting, Hervé Bachelez, MD, PhD, professor of dermatology and immunologist at the University of Paris and Saint-Louis Hospital, Paris, discussed recent advances in drug development for pustular psoriasis. He noted other recent findings of genetic variants related to the disease, including AP1S3, CARD14, and SERPINA3.
For GPP, he said, the current algorithm for management is based on weak evidence for treatments like acitretin, cyclosporine, methotrexate, and infliximab. The story is similar for other biologics, with evidence in the form of case series; open-label studies; controlled, prospective studies; or retrospective analyses. Most of the evidence has been amassed for TNF inhibitors. A retrospective study of all TNF inhibitors suggested they may be effective as induction and maintenance therapy, he noted.
Among IL-17A inhibitors, a prospective study of 12 patients in Japan found secukinumab showed efficacy against GPP, as did studies of ixekizumab and brodalumab. A small phase 3 study in Japan demonstrated efficacy for the IL-23 inhibitor guselkumab in patients with erythrodermic psoriasis and GPP (J Dermatol. 2018 May;45[5]:529-39).
The limited data are a reflection in part of the difficulty in studying GPP, since its flares tend to be more self-remitting than with psoriasis vulgaris or PPP.
There are two monoclonal antibodies against the IL-36 receptor currently being developed. A proof-of-concept study of one of them, spesolimab, showed promise against GPP, with five of seven patients reaching “clear” or “almost clear” scores on the Generalized Pustular Psoriasis Physician Global Assessment within a week after infusion and in all seven by the fourth week (N Engl J Med. 2019 Mar 7;380[10]:981-3).
With respect to PPP, the strongest evidence for conventional therapies comes from two randomized, controlled trials of cyclosporine, with response rates of 48% and 89%, compared with 19% and 21%, respectively, in the placebo groups, although the primary endpoint was poorly designed, according to Dr. Bachelez. Retinoids like etretinate and acitretin, combined with psoralen and UVA, also have some supporting evidence regarding efficacy.
Among biologics, secukinumab did not fare well in a phase 3 study of patients with PPP. A subset of patients may benefit from it, but there are no biomarkers available to identify them, Dr. Bachelez said. A phase 2 study of guselkumab in Japan told a similar story, with only weak signs of efficacy. While there are many more ongoing clinical trials evaluating treatments for PPP, which is encouraging, PPP seems to be more challenging at this stage to tackle than GPP, Dr. Bachelez added. “The genetically inherited IL-36 antagonist abnormalities are clearly driving the advances regarding the pathogenesis of the disease, mainly for GPP rather than PPP.”
Part of the efforts to develop therapies for pustular psoriasis relies on the development of new outcome measures, or adaptation of existing ones. “We have a need to adapt or develop new investigator-reported measures, we need to adapt or develop new patient-reported outcomes,” Dr. Duffin said.
Many existing measures use inconsistent language and anchoring definitions, and some may be proprietary, she added. “The language varies by sponsor and is sometimes tweaked or modified by the agencies. Often synonyms are being used … it raises questions, does it change the validity of the instrument?”
Dr. Duffin called for the research community to use the pause in clinical research during the COVID-19 pandemic to reassess the research agenda, develop consensus on performing and training for GPP and PPP assessments, develop patient-reported outcomes, and strengthen connections to industry.
Dr. Duffin and Dr. Bachelez have consulted, served on the advisory board, been a speaker for, and/or received research support from a wide range of pharmaceutical companies, including those that manufacture and develop psoriasis treatments.
FROM THE GRAPPA 2020 VIRTUAL ANNUAL MEETING