User login
New ASAM guideline released amid COVID-19 concerns
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
Home-based buprenorphine induction deemed safe for OUD
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
DIY masks: Worth the risk? Researchers are conflicted
In the midst of the rapidly spreading COVID-19 pandemic, hospitals and clinics are running out of masks. Health care workers are going online to beg for more, the hashtags #GetMePPE and #WeNeedPPE are trending on Twitter, and some hospitals have even put out public calls for mask donations. Health providers are working scared: They know that the moment the masks run out, they’re at increased risk for disease. So instead of waiting for mask shipments that may be weeks off, some people are making their own.
Using a simple template, they cut green surgical sheeting into half-moons, which they pin and sew before attaching elastic straps. Deaconess Health System in Evansville, Indiana, has posted instructions for fabric masks on their website and asked the public to step up and sew.
Elsewhere, health care workers have turned to diapers, maxi pads and other products to create masks. Social media channels are full of tips and sewing patterns. It’s an innovative strategy that is also contentious. Limited evidence suggests that homemade masks can offer some protection. But the DIY approach has also drawn criticism for providing a false sense of security, potentially putting wearers at risk.
The conflict points to an immediate need for more protective equipment, says Christopher Friese, PhD, RN, professor of nursing and public health at the University of Michigan, Ann Arbor. Also needed, he says, are new ideas for reducing strain on limited supplies, like adopting gear from other industries and finding innovative ways to provide care so that less protective gear is needed.
“We don’t want clinicians inventing and ‘MacGyvering’ their own device because we don’t want to put them at risk if we can avoid it,” says Friese, referring to the TV character who could build and assemble a vast array of tools/devices. “We have options that have been tested, and we have experience, maybe not in health care, but in other settings. We want to try that first before that frontline doctor, nurse, respiratory therapist decides to take matters into their own hands.
Increasingly, though, health care workers are finding they have no other choice — something even the CDC has acknowledged. In new guidelines, the agency recommends a bandanna, scarf, or other type of covering in cases where face masks are not available.
N95 respirators or surgical masks?
There are two main types of masks generally used in health care. N95 respirators filter out 95% of airborne particles, including bacteria and viruses. The lighter surgical or medical face masks are made to prevent spit and mucous from getting on patients or equipment.
Both types reduce rates of infection among health care workers, though comparisons (at least for influenza) have yet to show that one is superior to the other. One 2020 review by Chinese researchers, for example, analyzed six randomly controlled trials that included more than 9000 participants and found no added benefits of N95 masks over ordinary surgical masks for health care providers treating patients with the flu.
But COVID-19 is not influenza, and evidence suggests it may require more intensive protection, says Friese, who coauthored a blog post for JAMA about the country’s unpreparedness for protecting health care workers during a pandemic. The virus can linger in the air for hours, suggesting that N95 respirators are health care providers’ best option when treating infected patients.
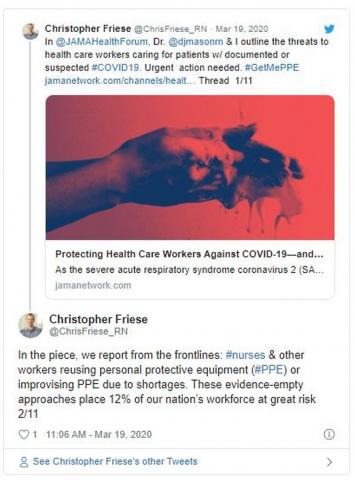
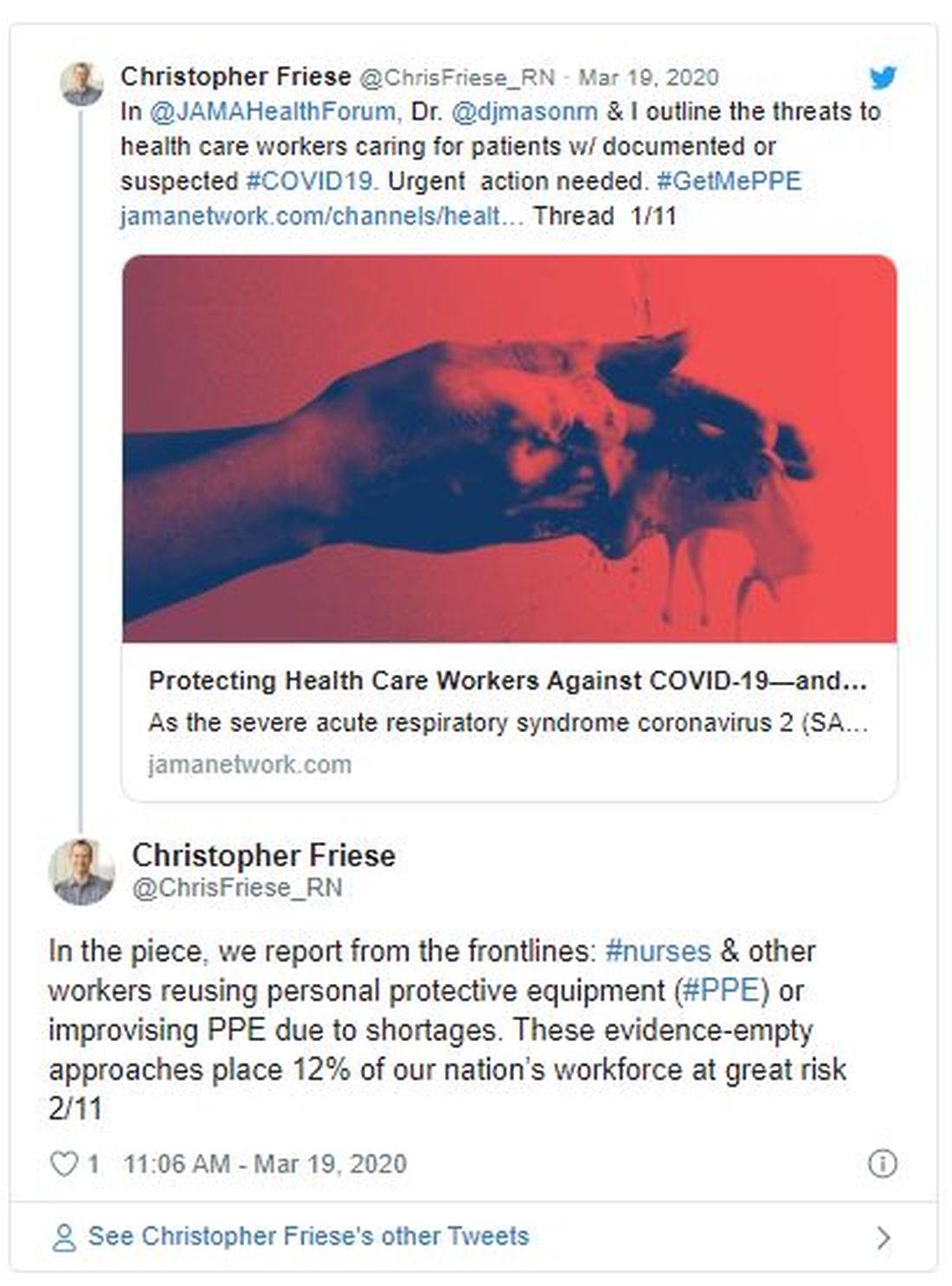
The problem is there’s not enough to go around — of either mask type. In a March 5 survey, National Nurses United reported that just 30% of more than 6500 US respondents said their organizations had enough PPE to respond to a surge in patients. Another 38% did not know if their organizations were prepared. In a tweet, Friese estimated that 12% of nurses and other providers are at risk from reusing equipment or using equipment that is not backed by evidence.
Physicians and providers around the world have been sharing strategies online for how to make their own masks. Techniques vary, as do materials and plans for how to use the homemade equipment. At Phoebe Putney Health, DIY masks are intended to be worn over N95 respirators and then disposed of so that the respirators can be reused more safely, says Amanda Clements, the hospital’s public relations coordinator. Providers might also wear them to greet people at the front door.
Some evidence suggests that homemade masks can help in a pinch, at least for some illnesses. For a 2013 study by researchers in the UK, volunteers made surgical masks from cotton T-shirts, then put them on and coughed into a chamber that measured how much bacterial content got through. The team also assessed the aerosol-filtering ability of a variety of household materials, including scarfs, antimicrobial pillowcases, vacuum-cleaner bags, and tea towels. They tested each material with an aerosol containing two types of bacteria similar in size to influenza.
Commercial surgical masks performed three times better than homemade ones in the filtration test. Surgical masks worked twice as well at blocking droplets on the cough test. But all the makeshift materials — which also included silk, linen, and regular pillowcases — blocked some microbes. Vacuum-cleaner bags blocked the most bacteria, but their stiffness and thickness made them unsuitable for use as masks, the researchers reported. Tea towels showed a similar pattern. But pillowcases and cotton T-shirts were stretchy enough to fit well, thereby reducing the particles that could get through or around them.
Homemade masks should be used only as a last resort if commercial masks become unavailable, the researchers concluded. “Probably something is better than nothing for trained health care workers — for droplet contact avoidance, if nothing else,” says Anna Davies, BSc, a research facilitator at the University of Cambridge, UK, who is a former public health microbiologist and one of the study’s authors.
She recommends that members of the general public donate any stockpiles they have to health care workers, and make their own if they want masks for personal use. She is working with collaborators in the US to develop guidance for how best to do it.
“If people are quarantined and looking for something worthwhile to do, it probably wouldn’t be the worst thing to apply themselves to,” she wrote by email. “My suggestion would be for something soft and cotton, ideally with a bit of stretch (although it’s a pain to sew), and in two layers, marked ‘inside’ and ‘outside.’ ”
The idea that something is better than nothing was also the conclusion of a 2008 study by researchers in the Netherlands and the US. The study enlisted 28 healthy individuals who performed a variety of tasks while wearing N95 masks, surgical masks, or homemade masks sewn from teacloths. Effectiveness varied among individuals, but over a 90-second period, N95 masks worked best, with 25 times more protection than surgical masks and about 50 times more protection than homemade ones. Surgical masks were twice as effective as homemade masks. But the homemade masks offered at least some protection against large droplets.
Researchers emphasize that it’s not yet clear whether those findings are applicable to aerosolized COVID-19. In an influenza pandemic, at least, the authors posit that homemade masks could reduce transmission for the general public enough for some immunity to build. “It is important not to focus on a single intervention in case of a pandemic,” the researchers write, “but to integrate all effective interventions for optimal protection.”
For health care workers on the frontlines of COVID-19, Friese says, homemade masks might do more than nothing but they also might not work. Instead, he would rather see providers using construction or nuclear-engineering masks. And his best suggestion is something many providers are already doing: reducing physical contact with patients through telemedicine and other creative solutions, which is cutting down the overwhelming need for PPE.
Homemade mask production emphasizes the urgent need for more supplies, Friese adds.
“The government needs to step up and do a variety of things to increase production, and that needs to happen now, immediately,” he says. “We don’t we don’t want our clinicians to have to come up with these decisions.”
This article first appeared on Medscape.com.
In the midst of the rapidly spreading COVID-19 pandemic, hospitals and clinics are running out of masks. Health care workers are going online to beg for more, the hashtags #GetMePPE and #WeNeedPPE are trending on Twitter, and some hospitals have even put out public calls for mask donations. Health providers are working scared: They know that the moment the masks run out, they’re at increased risk for disease. So instead of waiting for mask shipments that may be weeks off, some people are making their own.
Using a simple template, they cut green surgical sheeting into half-moons, which they pin and sew before attaching elastic straps. Deaconess Health System in Evansville, Indiana, has posted instructions for fabric masks on their website and asked the public to step up and sew.
Elsewhere, health care workers have turned to diapers, maxi pads and other products to create masks. Social media channels are full of tips and sewing patterns. It’s an innovative strategy that is also contentious. Limited evidence suggests that homemade masks can offer some protection. But the DIY approach has also drawn criticism for providing a false sense of security, potentially putting wearers at risk.
The conflict points to an immediate need for more protective equipment, says Christopher Friese, PhD, RN, professor of nursing and public health at the University of Michigan, Ann Arbor. Also needed, he says, are new ideas for reducing strain on limited supplies, like adopting gear from other industries and finding innovative ways to provide care so that less protective gear is needed.
“We don’t want clinicians inventing and ‘MacGyvering’ their own device because we don’t want to put them at risk if we can avoid it,” says Friese, referring to the TV character who could build and assemble a vast array of tools/devices. “We have options that have been tested, and we have experience, maybe not in health care, but in other settings. We want to try that first before that frontline doctor, nurse, respiratory therapist decides to take matters into their own hands.
Increasingly, though, health care workers are finding they have no other choice — something even the CDC has acknowledged. In new guidelines, the agency recommends a bandanna, scarf, or other type of covering in cases where face masks are not available.
N95 respirators or surgical masks?
There are two main types of masks generally used in health care. N95 respirators filter out 95% of airborne particles, including bacteria and viruses. The lighter surgical or medical face masks are made to prevent spit and mucous from getting on patients or equipment.
Both types reduce rates of infection among health care workers, though comparisons (at least for influenza) have yet to show that one is superior to the other. One 2020 review by Chinese researchers, for example, analyzed six randomly controlled trials that included more than 9000 participants and found no added benefits of N95 masks over ordinary surgical masks for health care providers treating patients with the flu.
But COVID-19 is not influenza, and evidence suggests it may require more intensive protection, says Friese, who coauthored a blog post for JAMA about the country’s unpreparedness for protecting health care workers during a pandemic. The virus can linger in the air for hours, suggesting that N95 respirators are health care providers’ best option when treating infected patients.
The problem is there’s not enough to go around — of either mask type. In a March 5 survey, National Nurses United reported that just 30% of more than 6500 US respondents said their organizations had enough PPE to respond to a surge in patients. Another 38% did not know if their organizations were prepared. In a tweet, Friese estimated that 12% of nurses and other providers are at risk from reusing equipment or using equipment that is not backed by evidence.
Physicians and providers around the world have been sharing strategies online for how to make their own masks. Techniques vary, as do materials and plans for how to use the homemade equipment. At Phoebe Putney Health, DIY masks are intended to be worn over N95 respirators and then disposed of so that the respirators can be reused more safely, says Amanda Clements, the hospital’s public relations coordinator. Providers might also wear them to greet people at the front door.
Some evidence suggests that homemade masks can help in a pinch, at least for some illnesses. For a 2013 study by researchers in the UK, volunteers made surgical masks from cotton T-shirts, then put them on and coughed into a chamber that measured how much bacterial content got through. The team also assessed the aerosol-filtering ability of a variety of household materials, including scarfs, antimicrobial pillowcases, vacuum-cleaner bags, and tea towels. They tested each material with an aerosol containing two types of bacteria similar in size to influenza.
Commercial surgical masks performed three times better than homemade ones in the filtration test. Surgical masks worked twice as well at blocking droplets on the cough test. But all the makeshift materials — which also included silk, linen, and regular pillowcases — blocked some microbes. Vacuum-cleaner bags blocked the most bacteria, but their stiffness and thickness made them unsuitable for use as masks, the researchers reported. Tea towels showed a similar pattern. But pillowcases and cotton T-shirts were stretchy enough to fit well, thereby reducing the particles that could get through or around them.
Homemade masks should be used only as a last resort if commercial masks become unavailable, the researchers concluded. “Probably something is better than nothing for trained health care workers — for droplet contact avoidance, if nothing else,” says Anna Davies, BSc, a research facilitator at the University of Cambridge, UK, who is a former public health microbiologist and one of the study’s authors.
She recommends that members of the general public donate any stockpiles they have to health care workers, and make their own if they want masks for personal use. She is working with collaborators in the US to develop guidance for how best to do it.
“If people are quarantined and looking for something worthwhile to do, it probably wouldn’t be the worst thing to apply themselves to,” she wrote by email. “My suggestion would be for something soft and cotton, ideally with a bit of stretch (although it’s a pain to sew), and in two layers, marked ‘inside’ and ‘outside.’ ”
The idea that something is better than nothing was also the conclusion of a 2008 study by researchers in the Netherlands and the US. The study enlisted 28 healthy individuals who performed a variety of tasks while wearing N95 masks, surgical masks, or homemade masks sewn from teacloths. Effectiveness varied among individuals, but over a 90-second period, N95 masks worked best, with 25 times more protection than surgical masks and about 50 times more protection than homemade ones. Surgical masks were twice as effective as homemade masks. But the homemade masks offered at least some protection against large droplets.
Researchers emphasize that it’s not yet clear whether those findings are applicable to aerosolized COVID-19. In an influenza pandemic, at least, the authors posit that homemade masks could reduce transmission for the general public enough for some immunity to build. “It is important not to focus on a single intervention in case of a pandemic,” the researchers write, “but to integrate all effective interventions for optimal protection.”
For health care workers on the frontlines of COVID-19, Friese says, homemade masks might do more than nothing but they also might not work. Instead, he would rather see providers using construction or nuclear-engineering masks. And his best suggestion is something many providers are already doing: reducing physical contact with patients through telemedicine and other creative solutions, which is cutting down the overwhelming need for PPE.
Homemade mask production emphasizes the urgent need for more supplies, Friese adds.
“The government needs to step up and do a variety of things to increase production, and that needs to happen now, immediately,” he says. “We don’t we don’t want our clinicians to have to come up with these decisions.”
This article first appeared on Medscape.com.
In the midst of the rapidly spreading COVID-19 pandemic, hospitals and clinics are running out of masks. Health care workers are going online to beg for more, the hashtags #GetMePPE and #WeNeedPPE are trending on Twitter, and some hospitals have even put out public calls for mask donations. Health providers are working scared: They know that the moment the masks run out, they’re at increased risk for disease. So instead of waiting for mask shipments that may be weeks off, some people are making their own.
Using a simple template, they cut green surgical sheeting into half-moons, which they pin and sew before attaching elastic straps. Deaconess Health System in Evansville, Indiana, has posted instructions for fabric masks on their website and asked the public to step up and sew.
Elsewhere, health care workers have turned to diapers, maxi pads and other products to create masks. Social media channels are full of tips and sewing patterns. It’s an innovative strategy that is also contentious. Limited evidence suggests that homemade masks can offer some protection. But the DIY approach has also drawn criticism for providing a false sense of security, potentially putting wearers at risk.
The conflict points to an immediate need for more protective equipment, says Christopher Friese, PhD, RN, professor of nursing and public health at the University of Michigan, Ann Arbor. Also needed, he says, are new ideas for reducing strain on limited supplies, like adopting gear from other industries and finding innovative ways to provide care so that less protective gear is needed.
“We don’t want clinicians inventing and ‘MacGyvering’ their own device because we don’t want to put them at risk if we can avoid it,” says Friese, referring to the TV character who could build and assemble a vast array of tools/devices. “We have options that have been tested, and we have experience, maybe not in health care, but in other settings. We want to try that first before that frontline doctor, nurse, respiratory therapist decides to take matters into their own hands.
Increasingly, though, health care workers are finding they have no other choice — something even the CDC has acknowledged. In new guidelines, the agency recommends a bandanna, scarf, or other type of covering in cases where face masks are not available.
N95 respirators or surgical masks?
There are two main types of masks generally used in health care. N95 respirators filter out 95% of airborne particles, including bacteria and viruses. The lighter surgical or medical face masks are made to prevent spit and mucous from getting on patients or equipment.
Both types reduce rates of infection among health care workers, though comparisons (at least for influenza) have yet to show that one is superior to the other. One 2020 review by Chinese researchers, for example, analyzed six randomly controlled trials that included more than 9000 participants and found no added benefits of N95 masks over ordinary surgical masks for health care providers treating patients with the flu.
But COVID-19 is not influenza, and evidence suggests it may require more intensive protection, says Friese, who coauthored a blog post for JAMA about the country’s unpreparedness for protecting health care workers during a pandemic. The virus can linger in the air for hours, suggesting that N95 respirators are health care providers’ best option when treating infected patients.
The problem is there’s not enough to go around — of either mask type. In a March 5 survey, National Nurses United reported that just 30% of more than 6500 US respondents said their organizations had enough PPE to respond to a surge in patients. Another 38% did not know if their organizations were prepared. In a tweet, Friese estimated that 12% of nurses and other providers are at risk from reusing equipment or using equipment that is not backed by evidence.
Physicians and providers around the world have been sharing strategies online for how to make their own masks. Techniques vary, as do materials and plans for how to use the homemade equipment. At Phoebe Putney Health, DIY masks are intended to be worn over N95 respirators and then disposed of so that the respirators can be reused more safely, says Amanda Clements, the hospital’s public relations coordinator. Providers might also wear them to greet people at the front door.
Some evidence suggests that homemade masks can help in a pinch, at least for some illnesses. For a 2013 study by researchers in the UK, volunteers made surgical masks from cotton T-shirts, then put them on and coughed into a chamber that measured how much bacterial content got through. The team also assessed the aerosol-filtering ability of a variety of household materials, including scarfs, antimicrobial pillowcases, vacuum-cleaner bags, and tea towels. They tested each material with an aerosol containing two types of bacteria similar in size to influenza.
Commercial surgical masks performed three times better than homemade ones in the filtration test. Surgical masks worked twice as well at blocking droplets on the cough test. But all the makeshift materials — which also included silk, linen, and regular pillowcases — blocked some microbes. Vacuum-cleaner bags blocked the most bacteria, but their stiffness and thickness made them unsuitable for use as masks, the researchers reported. Tea towels showed a similar pattern. But pillowcases and cotton T-shirts were stretchy enough to fit well, thereby reducing the particles that could get through or around them.
Homemade masks should be used only as a last resort if commercial masks become unavailable, the researchers concluded. “Probably something is better than nothing for trained health care workers — for droplet contact avoidance, if nothing else,” says Anna Davies, BSc, a research facilitator at the University of Cambridge, UK, who is a former public health microbiologist and one of the study’s authors.
She recommends that members of the general public donate any stockpiles they have to health care workers, and make their own if they want masks for personal use. She is working with collaborators in the US to develop guidance for how best to do it.
“If people are quarantined and looking for something worthwhile to do, it probably wouldn’t be the worst thing to apply themselves to,” she wrote by email. “My suggestion would be for something soft and cotton, ideally with a bit of stretch (although it’s a pain to sew), and in two layers, marked ‘inside’ and ‘outside.’ ”
The idea that something is better than nothing was also the conclusion of a 2008 study by researchers in the Netherlands and the US. The study enlisted 28 healthy individuals who performed a variety of tasks while wearing N95 masks, surgical masks, or homemade masks sewn from teacloths. Effectiveness varied among individuals, but over a 90-second period, N95 masks worked best, with 25 times more protection than surgical masks and about 50 times more protection than homemade ones. Surgical masks were twice as effective as homemade masks. But the homemade masks offered at least some protection against large droplets.
Researchers emphasize that it’s not yet clear whether those findings are applicable to aerosolized COVID-19. In an influenza pandemic, at least, the authors posit that homemade masks could reduce transmission for the general public enough for some immunity to build. “It is important not to focus on a single intervention in case of a pandemic,” the researchers write, “but to integrate all effective interventions for optimal protection.”
For health care workers on the frontlines of COVID-19, Friese says, homemade masks might do more than nothing but they also might not work. Instead, he would rather see providers using construction or nuclear-engineering masks. And his best suggestion is something many providers are already doing: reducing physical contact with patients through telemedicine and other creative solutions, which is cutting down the overwhelming need for PPE.
Homemade mask production emphasizes the urgent need for more supplies, Friese adds.
“The government needs to step up and do a variety of things to increase production, and that needs to happen now, immediately,” he says. “We don’t we don’t want our clinicians to have to come up with these decisions.”
This article first appeared on Medscape.com.
COVID-19 prompts ‘lifesaving’ policy change for opioid addiction
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
Match Day 2020: Online announcements replace celebrations, champagne
The third Friday in March usually marks a time when medical students across the United States participate in envelope-opening ceremonies with peers and family members. This year, the ruthless onslaught of coronavirus has forced residency programs to rethink their celebrations, leveraging social media platforms and other technologies to toast Match Day in cyberspace.
In the absence of ceremonies taking place due to restrictions on mass gatherings, “we anticipate that students may be more emotional than they expect,” Hannah R. Hughes, MD, president of the Emergency Medicine Residents’ Association (EMRA) said in an interview. To support these students on their journey to residency, EMRA has launched a social media campaign, asking medical students “to share with us their envelope-opening moments – either a selfie, photo, or video – that we can share with our online networks,” Dr. Hughes said.
EMRA is also asking program coordinators to forward photos and congratulatory messages to their new residents “so that we can share them with our networks at large,” she added.
Going virtual, it seems, has become the new norm.
At the University of California, San Francisco, the medical school decided to cancel its Match Day celebration for new interns, echoing many other programs across the United States. “We always send out a welcome email and make phone calls to all of our new interns,” said Rebecca Berman, MD, director of UCSF’s internal medicine residency program, which houses 63 medicine interns and 181 residents. Traditionally, the program has hosted the celebration for current residents. That, of course, had to change this year.
Current interns like to join in the fun, “since it means their internship is rapidly coming to a close,” said Dr. Berman, who at press time was considering a virtual toast via Zoom as a possible alternative. “These are difficult times for everyone, and we are doing our best to make our residents feel united and connected while they take care of patients in the era of social distancing.”
Melissa Held, MD, associate dean of medical student affairs at the University of Connecticut’s School of Medicine, Farmington, had been planning a celebration in the school’s academic rotunda with food and champagne. “Students typically come with their family members or significant others. The dean and I usually say a few words and then at noon, students get envelopes and can open them to find out where they matched for residency,” Dr. Held said. This year, the school will be uploading Match letters to its online system. Students can remotely find out where they matched at noon. “I plan to put together a slide show of pictures and congratulatory remarks from faculty and staff that will be sent to them around 11:30 a.m.,” Dr. Held said.
Mark Miceli, EdD, who oversees Match Day for the 130-plus medical students at the University of Massachusetts Medical School, Worcester, is inviting faculty and staff to submit short videos of congratulations, which it will post on its student affairs Match Day Instagram account. Like other schools, it will share results with students in an email, said Dr. Miceli, assistant vice provost of student life. “This message will be more personalized to our school than the NRMP [National Resident Matching Program] message, and will also include links to our match stats, a map of our matched student locations, and a list of where folks matched,” he said.
Students can opt out of the list if they want to. The communications department has also provided templates for signs students can print out. “They can write in where they matched, and take pictures for social media. We are encouraging the use of various hashtags to help build a virtual community,” Dr. Miceli said.
In a state hit particularly hard by coronavirus, the University of Washington School of Medicine is spreading Match Day cheer through online meeting platforms and celebratory graphics. This five-state school, representing students from Washington, Wyoming, Alaska, Montana, and Idaho, usually hosts several events across the different states and students have their pick of which to attend, according to Sarah Wood, associate director of student affairs.
In lieu of in-person events, some states are hosting a Zoom online celebration, others are using social media networking systems. “We’re inviting everyone to take part in an online event ... where we’ll do a slide show of photos that one of our students put together,” Ms. Wood said.
Students are disappointed in this change of plans, she said. To make things more festive, Ms. Wood is adding graphics such as fireworks and photos to the emails containing the Match results. “I want this to be more exciting for them than just a basic letter,” she said.
For now, Ms. Wood is trying to focus on the Match Day celebration, but admits that “my bigger fear is if we have to cancel graduation – and what that might look like.”
The third Friday in March usually marks a time when medical students across the United States participate in envelope-opening ceremonies with peers and family members. This year, the ruthless onslaught of coronavirus has forced residency programs to rethink their celebrations, leveraging social media platforms and other technologies to toast Match Day in cyberspace.
In the absence of ceremonies taking place due to restrictions on mass gatherings, “we anticipate that students may be more emotional than they expect,” Hannah R. Hughes, MD, president of the Emergency Medicine Residents’ Association (EMRA) said in an interview. To support these students on their journey to residency, EMRA has launched a social media campaign, asking medical students “to share with us their envelope-opening moments – either a selfie, photo, or video – that we can share with our online networks,” Dr. Hughes said.
EMRA is also asking program coordinators to forward photos and congratulatory messages to their new residents “so that we can share them with our networks at large,” she added.
Going virtual, it seems, has become the new norm.
At the University of California, San Francisco, the medical school decided to cancel its Match Day celebration for new interns, echoing many other programs across the United States. “We always send out a welcome email and make phone calls to all of our new interns,” said Rebecca Berman, MD, director of UCSF’s internal medicine residency program, which houses 63 medicine interns and 181 residents. Traditionally, the program has hosted the celebration for current residents. That, of course, had to change this year.
Current interns like to join in the fun, “since it means their internship is rapidly coming to a close,” said Dr. Berman, who at press time was considering a virtual toast via Zoom as a possible alternative. “These are difficult times for everyone, and we are doing our best to make our residents feel united and connected while they take care of patients in the era of social distancing.”
Melissa Held, MD, associate dean of medical student affairs at the University of Connecticut’s School of Medicine, Farmington, had been planning a celebration in the school’s academic rotunda with food and champagne. “Students typically come with their family members or significant others. The dean and I usually say a few words and then at noon, students get envelopes and can open them to find out where they matched for residency,” Dr. Held said. This year, the school will be uploading Match letters to its online system. Students can remotely find out where they matched at noon. “I plan to put together a slide show of pictures and congratulatory remarks from faculty and staff that will be sent to them around 11:30 a.m.,” Dr. Held said.
Mark Miceli, EdD, who oversees Match Day for the 130-plus medical students at the University of Massachusetts Medical School, Worcester, is inviting faculty and staff to submit short videos of congratulations, which it will post on its student affairs Match Day Instagram account. Like other schools, it will share results with students in an email, said Dr. Miceli, assistant vice provost of student life. “This message will be more personalized to our school than the NRMP [National Resident Matching Program] message, and will also include links to our match stats, a map of our matched student locations, and a list of where folks matched,” he said.
Students can opt out of the list if they want to. The communications department has also provided templates for signs students can print out. “They can write in where they matched, and take pictures for social media. We are encouraging the use of various hashtags to help build a virtual community,” Dr. Miceli said.
In a state hit particularly hard by coronavirus, the University of Washington School of Medicine is spreading Match Day cheer through online meeting platforms and celebratory graphics. This five-state school, representing students from Washington, Wyoming, Alaska, Montana, and Idaho, usually hosts several events across the different states and students have their pick of which to attend, according to Sarah Wood, associate director of student affairs.
In lieu of in-person events, some states are hosting a Zoom online celebration, others are using social media networking systems. “We’re inviting everyone to take part in an online event ... where we’ll do a slide show of photos that one of our students put together,” Ms. Wood said.
Students are disappointed in this change of plans, she said. To make things more festive, Ms. Wood is adding graphics such as fireworks and photos to the emails containing the Match results. “I want this to be more exciting for them than just a basic letter,” she said.
For now, Ms. Wood is trying to focus on the Match Day celebration, but admits that “my bigger fear is if we have to cancel graduation – and what that might look like.”
The third Friday in March usually marks a time when medical students across the United States participate in envelope-opening ceremonies with peers and family members. This year, the ruthless onslaught of coronavirus has forced residency programs to rethink their celebrations, leveraging social media platforms and other technologies to toast Match Day in cyberspace.
In the absence of ceremonies taking place due to restrictions on mass gatherings, “we anticipate that students may be more emotional than they expect,” Hannah R. Hughes, MD, president of the Emergency Medicine Residents’ Association (EMRA) said in an interview. To support these students on their journey to residency, EMRA has launched a social media campaign, asking medical students “to share with us their envelope-opening moments – either a selfie, photo, or video – that we can share with our online networks,” Dr. Hughes said.
EMRA is also asking program coordinators to forward photos and congratulatory messages to their new residents “so that we can share them with our networks at large,” she added.
Going virtual, it seems, has become the new norm.
At the University of California, San Francisco, the medical school decided to cancel its Match Day celebration for new interns, echoing many other programs across the United States. “We always send out a welcome email and make phone calls to all of our new interns,” said Rebecca Berman, MD, director of UCSF’s internal medicine residency program, which houses 63 medicine interns and 181 residents. Traditionally, the program has hosted the celebration for current residents. That, of course, had to change this year.
Current interns like to join in the fun, “since it means their internship is rapidly coming to a close,” said Dr. Berman, who at press time was considering a virtual toast via Zoom as a possible alternative. “These are difficult times for everyone, and we are doing our best to make our residents feel united and connected while they take care of patients in the era of social distancing.”
Melissa Held, MD, associate dean of medical student affairs at the University of Connecticut’s School of Medicine, Farmington, had been planning a celebration in the school’s academic rotunda with food and champagne. “Students typically come with their family members or significant others. The dean and I usually say a few words and then at noon, students get envelopes and can open them to find out where they matched for residency,” Dr. Held said. This year, the school will be uploading Match letters to its online system. Students can remotely find out where they matched at noon. “I plan to put together a slide show of pictures and congratulatory remarks from faculty and staff that will be sent to them around 11:30 a.m.,” Dr. Held said.
Mark Miceli, EdD, who oversees Match Day for the 130-plus medical students at the University of Massachusetts Medical School, Worcester, is inviting faculty and staff to submit short videos of congratulations, which it will post on its student affairs Match Day Instagram account. Like other schools, it will share results with students in an email, said Dr. Miceli, assistant vice provost of student life. “This message will be more personalized to our school than the NRMP [National Resident Matching Program] message, and will also include links to our match stats, a map of our matched student locations, and a list of where folks matched,” he said.
Students can opt out of the list if they want to. The communications department has also provided templates for signs students can print out. “They can write in where they matched, and take pictures for social media. We are encouraging the use of various hashtags to help build a virtual community,” Dr. Miceli said.
In a state hit particularly hard by coronavirus, the University of Washington School of Medicine is spreading Match Day cheer through online meeting platforms and celebratory graphics. This five-state school, representing students from Washington, Wyoming, Alaska, Montana, and Idaho, usually hosts several events across the different states and students have their pick of which to attend, according to Sarah Wood, associate director of student affairs.
In lieu of in-person events, some states are hosting a Zoom online celebration, others are using social media networking systems. “We’re inviting everyone to take part in an online event ... where we’ll do a slide show of photos that one of our students put together,” Ms. Wood said.
Students are disappointed in this change of plans, she said. To make things more festive, Ms. Wood is adding graphics such as fireworks and photos to the emails containing the Match results. “I want this to be more exciting for them than just a basic letter,” she said.
For now, Ms. Wood is trying to focus on the Match Day celebration, but admits that “my bigger fear is if we have to cancel graduation – and what that might look like.”
Feds tout drug candidates to treat COVID-19
Therapeutics could be available in the near term to help treat COVID-19 patients, according to President Donald Trump.
During a March 19 press briefing, the president highlighted two drugs that could be put into play in the battle against the virus.
The first product is hydroxychloroquine (Plaquenil), a drug used to treat malaria and severe arthritis, is showing promise as a possible treatment for COVID-19.
“The nice part is it’s been around for a long time, so we know that if things go as planned, it’s not going to kill anybody,” President Trump said. “When you go with a brand-new drug, you don’t know that that’s going to happen,” adding that it has shown “very, very encouraging” results as a potential treatment for COVID-19.
He said this drug will be made available “almost immediately.” During the press conference, Food and Drug Administration Commissioner Stephen M. Hahn, MD, suggested the drug would be made available in the context of a large pragmatic clinical trial, enabling the FDA to collect data on it and make a long-term decision on its viability to treat COVID-19.
Dr. Hahn also pointed to the Gilead drug remdesivir – a drug originally developed to fight Ebola and currently undergoing clinical trials – as another possible candidate for a near-term therapeutic to help treat patients while vaccine development occurs.
Dr. Hahn noted that, while the agency is striving to provide regulatory flexibility, safety is paramount. “Let me make one thing clear: FDA’s responsibility to the American people is to ensure that products are safe and effective and that we are continuing to do that.”
He noted that if these and other experimental drugs show promise, physicians can request them under “compassionate use” provisions.
“We have criteria for that, and very speedy approval for that,” Dr. Hahn said. “The important thing about compassionate use ... this is even beyond ‘right to try.’ [We] get to collect the information about that.”
He noted that the FDA is looking at other drugs that are approved for other indications. The examinations of existing therapies are meant to be a bridge as companies work to develop new therapeutics as well as vaccines.
Dr. Hahn also highlighted a cross-agency effort on convalescent plasma, which uses the plasma from a patient who has recovered from COVID-19 infection to help patients fight the virus. “This is a possible treatment; this is not a proven treatment, “ Dr. Hahn said.
Takeda is working on an immunoglobulin treatment based on its intravenous immunoglobulin product Gammagard Liquid.
Julie Kim, president of plasma-derived therapies at Takeda, said the company should be able to go straight into testing efficacy of this approach, given the known safety profile of the treatment. She made the comments during a March 18 press briefing hosted by Pharmaceutical Research and Manufacturers of America (PhRMA). Ms. Kim did caution that this would not be a mass market kind of treatment, as supply would depend on plasma donations from individuals who have fully recovered from a COVID-19 infection. She estimated that the treatment could be available to a targeted group of high-risk patients in 9-18 months.
PhRMA president and CEO Stephen Ubl said the industry is “literally working around the clock” on four key areas: development of new diagnostics, identification of potential existing treatments to make available through trials and compassionate use, development of novel therapies, and development of a vaccine.
There are more than 80 clinical trials underway on existing treatments that could have approval to treat COVID-19 in a matter of months, he said.
Mikael Dolsten, MD, PhD, chief scientific officer at Pfizer, said that the company is working with Germany-based BioNTech SE to develop an mRNA-based vaccine for COVID-19, with testing expected to begin in Germany, China, and the United States by the end of April. The company also is screening antiviral compounds that were previously in development against other coronavirus diseases.
Clement Lewin, PhD, associate vice president of R&D strategy for vaccines at Sanofi, said the company has partnered with Regeneron to launch a trial of sarilumab (Kevzara), a drug approved to treat moderate to severe rheumatoid arthritis, to help treat COVID-19.
Meanwhile, Lilly Chief Scientific Officer Daniel Skovronsky, MD, PhD, noted that his company is collaborating with AbCellera to develop therapeutics using monoclonal antibodies isolated from one of the first U.S. patients who recovered from COVID-19. He said the goal is to begin testing within the next 4 months.
Separately, World Health Organization Director General Tedros Adhanom Ghebreyesus announced during a March 18 press conference that it is spearheading a large international study examining a number of different treatments in what has been dubbed the SOLIDARITY trial. Argentina, Bahrain, Canada, France, Iran, Norway, South Africa, Spain, Switzerland, and Thailand have signed on to be a part of the trial, with more countries expected to participate.
“I continue to be inspired by the many demonstrations of solidarity from all over the world,” he said. “These and other efforts give me hope that together, we can and will prevail. This virus is presenting us with an unprecedented threat. But it’s also an unprecedented opportunity to come together as one against a common enemy, an enemy against humanity.”
Therapeutics could be available in the near term to help treat COVID-19 patients, according to President Donald Trump.
During a March 19 press briefing, the president highlighted two drugs that could be put into play in the battle against the virus.
The first product is hydroxychloroquine (Plaquenil), a drug used to treat malaria and severe arthritis, is showing promise as a possible treatment for COVID-19.
“The nice part is it’s been around for a long time, so we know that if things go as planned, it’s not going to kill anybody,” President Trump said. “When you go with a brand-new drug, you don’t know that that’s going to happen,” adding that it has shown “very, very encouraging” results as a potential treatment for COVID-19.
He said this drug will be made available “almost immediately.” During the press conference, Food and Drug Administration Commissioner Stephen M. Hahn, MD, suggested the drug would be made available in the context of a large pragmatic clinical trial, enabling the FDA to collect data on it and make a long-term decision on its viability to treat COVID-19.
Dr. Hahn also pointed to the Gilead drug remdesivir – a drug originally developed to fight Ebola and currently undergoing clinical trials – as another possible candidate for a near-term therapeutic to help treat patients while vaccine development occurs.
Dr. Hahn noted that, while the agency is striving to provide regulatory flexibility, safety is paramount. “Let me make one thing clear: FDA’s responsibility to the American people is to ensure that products are safe and effective and that we are continuing to do that.”
He noted that if these and other experimental drugs show promise, physicians can request them under “compassionate use” provisions.
“We have criteria for that, and very speedy approval for that,” Dr. Hahn said. “The important thing about compassionate use ... this is even beyond ‘right to try.’ [We] get to collect the information about that.”
He noted that the FDA is looking at other drugs that are approved for other indications. The examinations of existing therapies are meant to be a bridge as companies work to develop new therapeutics as well as vaccines.
Dr. Hahn also highlighted a cross-agency effort on convalescent plasma, which uses the plasma from a patient who has recovered from COVID-19 infection to help patients fight the virus. “This is a possible treatment; this is not a proven treatment, “ Dr. Hahn said.
Takeda is working on an immunoglobulin treatment based on its intravenous immunoglobulin product Gammagard Liquid.
Julie Kim, president of plasma-derived therapies at Takeda, said the company should be able to go straight into testing efficacy of this approach, given the known safety profile of the treatment. She made the comments during a March 18 press briefing hosted by Pharmaceutical Research and Manufacturers of America (PhRMA). Ms. Kim did caution that this would not be a mass market kind of treatment, as supply would depend on plasma donations from individuals who have fully recovered from a COVID-19 infection. She estimated that the treatment could be available to a targeted group of high-risk patients in 9-18 months.
PhRMA president and CEO Stephen Ubl said the industry is “literally working around the clock” on four key areas: development of new diagnostics, identification of potential existing treatments to make available through trials and compassionate use, development of novel therapies, and development of a vaccine.
There are more than 80 clinical trials underway on existing treatments that could have approval to treat COVID-19 in a matter of months, he said.
Mikael Dolsten, MD, PhD, chief scientific officer at Pfizer, said that the company is working with Germany-based BioNTech SE to develop an mRNA-based vaccine for COVID-19, with testing expected to begin in Germany, China, and the United States by the end of April. The company also is screening antiviral compounds that were previously in development against other coronavirus diseases.
Clement Lewin, PhD, associate vice president of R&D strategy for vaccines at Sanofi, said the company has partnered with Regeneron to launch a trial of sarilumab (Kevzara), a drug approved to treat moderate to severe rheumatoid arthritis, to help treat COVID-19.
Meanwhile, Lilly Chief Scientific Officer Daniel Skovronsky, MD, PhD, noted that his company is collaborating with AbCellera to develop therapeutics using monoclonal antibodies isolated from one of the first U.S. patients who recovered from COVID-19. He said the goal is to begin testing within the next 4 months.
Separately, World Health Organization Director General Tedros Adhanom Ghebreyesus announced during a March 18 press conference that it is spearheading a large international study examining a number of different treatments in what has been dubbed the SOLIDARITY trial. Argentina, Bahrain, Canada, France, Iran, Norway, South Africa, Spain, Switzerland, and Thailand have signed on to be a part of the trial, with more countries expected to participate.
“I continue to be inspired by the many demonstrations of solidarity from all over the world,” he said. “These and other efforts give me hope that together, we can and will prevail. This virus is presenting us with an unprecedented threat. But it’s also an unprecedented opportunity to come together as one against a common enemy, an enemy against humanity.”
Therapeutics could be available in the near term to help treat COVID-19 patients, according to President Donald Trump.
During a March 19 press briefing, the president highlighted two drugs that could be put into play in the battle against the virus.
The first product is hydroxychloroquine (Plaquenil), a drug used to treat malaria and severe arthritis, is showing promise as a possible treatment for COVID-19.
“The nice part is it’s been around for a long time, so we know that if things go as planned, it’s not going to kill anybody,” President Trump said. “When you go with a brand-new drug, you don’t know that that’s going to happen,” adding that it has shown “very, very encouraging” results as a potential treatment for COVID-19.
He said this drug will be made available “almost immediately.” During the press conference, Food and Drug Administration Commissioner Stephen M. Hahn, MD, suggested the drug would be made available in the context of a large pragmatic clinical trial, enabling the FDA to collect data on it and make a long-term decision on its viability to treat COVID-19.
Dr. Hahn also pointed to the Gilead drug remdesivir – a drug originally developed to fight Ebola and currently undergoing clinical trials – as another possible candidate for a near-term therapeutic to help treat patients while vaccine development occurs.
Dr. Hahn noted that, while the agency is striving to provide regulatory flexibility, safety is paramount. “Let me make one thing clear: FDA’s responsibility to the American people is to ensure that products are safe and effective and that we are continuing to do that.”
He noted that if these and other experimental drugs show promise, physicians can request them under “compassionate use” provisions.
“We have criteria for that, and very speedy approval for that,” Dr. Hahn said. “The important thing about compassionate use ... this is even beyond ‘right to try.’ [We] get to collect the information about that.”
He noted that the FDA is looking at other drugs that are approved for other indications. The examinations of existing therapies are meant to be a bridge as companies work to develop new therapeutics as well as vaccines.
Dr. Hahn also highlighted a cross-agency effort on convalescent plasma, which uses the plasma from a patient who has recovered from COVID-19 infection to help patients fight the virus. “This is a possible treatment; this is not a proven treatment, “ Dr. Hahn said.
Takeda is working on an immunoglobulin treatment based on its intravenous immunoglobulin product Gammagard Liquid.
Julie Kim, president of plasma-derived therapies at Takeda, said the company should be able to go straight into testing efficacy of this approach, given the known safety profile of the treatment. She made the comments during a March 18 press briefing hosted by Pharmaceutical Research and Manufacturers of America (PhRMA). Ms. Kim did caution that this would not be a mass market kind of treatment, as supply would depend on plasma donations from individuals who have fully recovered from a COVID-19 infection. She estimated that the treatment could be available to a targeted group of high-risk patients in 9-18 months.
PhRMA president and CEO Stephen Ubl said the industry is “literally working around the clock” on four key areas: development of new diagnostics, identification of potential existing treatments to make available through trials and compassionate use, development of novel therapies, and development of a vaccine.
There are more than 80 clinical trials underway on existing treatments that could have approval to treat COVID-19 in a matter of months, he said.
Mikael Dolsten, MD, PhD, chief scientific officer at Pfizer, said that the company is working with Germany-based BioNTech SE to develop an mRNA-based vaccine for COVID-19, with testing expected to begin in Germany, China, and the United States by the end of April. The company also is screening antiviral compounds that were previously in development against other coronavirus diseases.
Clement Lewin, PhD, associate vice president of R&D strategy for vaccines at Sanofi, said the company has partnered with Regeneron to launch a trial of sarilumab (Kevzara), a drug approved to treat moderate to severe rheumatoid arthritis, to help treat COVID-19.
Meanwhile, Lilly Chief Scientific Officer Daniel Skovronsky, MD, PhD, noted that his company is collaborating with AbCellera to develop therapeutics using monoclonal antibodies isolated from one of the first U.S. patients who recovered from COVID-19. He said the goal is to begin testing within the next 4 months.
Separately, World Health Organization Director General Tedros Adhanom Ghebreyesus announced during a March 18 press conference that it is spearheading a large international study examining a number of different treatments in what has been dubbed the SOLIDARITY trial. Argentina, Bahrain, Canada, France, Iran, Norway, South Africa, Spain, Switzerland, and Thailand have signed on to be a part of the trial, with more countries expected to participate.
“I continue to be inspired by the many demonstrations of solidarity from all over the world,” he said. “These and other efforts give me hope that together, we can and will prevail. This virus is presenting us with an unprecedented threat. But it’s also an unprecedented opportunity to come together as one against a common enemy, an enemy against humanity.”
Standing by and still open for business during COVID-19 pandemic
As of this morning, March 19, 2020, I’m still working.
Granted, there aren’t a lot of people who want to come in. My schedule has dropped to 3-5 follow-ups per day and no new patients.
I can understand people not wanting to expose themselves unnecessarily right now.
But, I’m still a doctor. What drove me to study for the MCAT, apply to med school 2 years in a row, and then survive medical school, internship, residency, and fellowship ... is still there.
Like I said in my 1987 personal statement, I still want to help people. I’d feel remiss if (provided I don’t have COVID-19) I didn’t show up for work each day, ready to care for any who need me. It’s part of who I am, what I do, and what I believe in.
I’m sure my colleagues in family practice, internal medicine, and pulmonology are swamped right now, but neurologists with primarily outpatient practices are taking a back seat except for a handful of patients.
My small office has been set up for my staff to work remotely in a pinch since 2016, so that was easy to enact. The three of us cover the phones the way we always have, and I see patients here.
With the relaxing of telehealth requirements for Medicare that were announced on March 17, I’m setting up to “see” patients remotely.
The whole situation seems bizarre and surreal.
It’s easy for anyone to read too much into anything. A brief tickle in my throat when I wake up, or a sneeze, or a few coughs, suddenly trigger a flurry of “could I have it?” thoughts. Fortunately, they fade when things quickly return to normal, but a few weeks ago I wouldn’t have thought anything of them at all.
Inevitably, I and pretty much everyone else will be exposed to or catch the virus. It’s what virions do. Unless you absolutely isolate yourself on a desert island, it will happen. When it does, you can only hope for the best.
I’m here for my patients today and will be as long as they need me. Unless I have to go into quarantine, of course. And even then, if able, I’ll do the best I can to treat them remotely.
That’s all I could ever want.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
As of this morning, March 19, 2020, I’m still working.
Granted, there aren’t a lot of people who want to come in. My schedule has dropped to 3-5 follow-ups per day and no new patients.
I can understand people not wanting to expose themselves unnecessarily right now.
But, I’m still a doctor. What drove me to study for the MCAT, apply to med school 2 years in a row, and then survive medical school, internship, residency, and fellowship ... is still there.
Like I said in my 1987 personal statement, I still want to help people. I’d feel remiss if (provided I don’t have COVID-19) I didn’t show up for work each day, ready to care for any who need me. It’s part of who I am, what I do, and what I believe in.
I’m sure my colleagues in family practice, internal medicine, and pulmonology are swamped right now, but neurologists with primarily outpatient practices are taking a back seat except for a handful of patients.
My small office has been set up for my staff to work remotely in a pinch since 2016, so that was easy to enact. The three of us cover the phones the way we always have, and I see patients here.
With the relaxing of telehealth requirements for Medicare that were announced on March 17, I’m setting up to “see” patients remotely.
The whole situation seems bizarre and surreal.
It’s easy for anyone to read too much into anything. A brief tickle in my throat when I wake up, or a sneeze, or a few coughs, suddenly trigger a flurry of “could I have it?” thoughts. Fortunately, they fade when things quickly return to normal, but a few weeks ago I wouldn’t have thought anything of them at all.
Inevitably, I and pretty much everyone else will be exposed to or catch the virus. It’s what virions do. Unless you absolutely isolate yourself on a desert island, it will happen. When it does, you can only hope for the best.
I’m here for my patients today and will be as long as they need me. Unless I have to go into quarantine, of course. And even then, if able, I’ll do the best I can to treat them remotely.
That’s all I could ever want.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
As of this morning, March 19, 2020, I’m still working.
Granted, there aren’t a lot of people who want to come in. My schedule has dropped to 3-5 follow-ups per day and no new patients.
I can understand people not wanting to expose themselves unnecessarily right now.
But, I’m still a doctor. What drove me to study for the MCAT, apply to med school 2 years in a row, and then survive medical school, internship, residency, and fellowship ... is still there.
Like I said in my 1987 personal statement, I still want to help people. I’d feel remiss if (provided I don’t have COVID-19) I didn’t show up for work each day, ready to care for any who need me. It’s part of who I am, what I do, and what I believe in.
I’m sure my colleagues in family practice, internal medicine, and pulmonology are swamped right now, but neurologists with primarily outpatient practices are taking a back seat except for a handful of patients.
My small office has been set up for my staff to work remotely in a pinch since 2016, so that was easy to enact. The three of us cover the phones the way we always have, and I see patients here.
With the relaxing of telehealth requirements for Medicare that were announced on March 17, I’m setting up to “see” patients remotely.
The whole situation seems bizarre and surreal.
It’s easy for anyone to read too much into anything. A brief tickle in my throat when I wake up, or a sneeze, or a few coughs, suddenly trigger a flurry of “could I have it?” thoughts. Fortunately, they fade when things quickly return to normal, but a few weeks ago I wouldn’t have thought anything of them at all.
Inevitably, I and pretty much everyone else will be exposed to or catch the virus. It’s what virions do. Unless you absolutely isolate yourself on a desert island, it will happen. When it does, you can only hope for the best.
I’m here for my patients today and will be as long as they need me. Unless I have to go into quarantine, of course. And even then, if able, I’ll do the best I can to treat them remotely.
That’s all I could ever want.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
AGA and colleague societies issue clinical insights for COVID-19
Amid the growing SARS-CoV-2 pandemic, currently in its expansive growth phase in the United States, the American Gastroenterological Association (AGA), the American Association for the Study of Liver Diseases (AASLD), the American College of Gastroenterology (ACG), and the American Society for Gastrointestinal Endoscopy (ASGE) have jointly released “COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers,” which can be found on the websites of the various societies.
“The purpose of this communication is to jointly provide you with up to date COVID-19 information in order to maintain the highest level of health and safety for our patients, staff, community, and ourselves,” according to the AGA website announcement.
In particular, the societies point out that there is recent evidence suggesting the potential for coronavirus transmission through droplets and perhaps fecal shedding, which pose potential risks in particular during endoscopy and colonoscopy procedures to other patients, endoscopy personnel, and practitioners.
Relevant clinical factors related to COVID-19 are discussed, including the fact that asymptomatic spread can occur during the prodromal phase (the mean incubation period is approximately 5 days, with a range of 0-14 days), with viral shedding greatest when symptoms begin.
Between 20% and 30% of patients with COVID-19 infection show abnormal liver enzymes. In addition, COVID-19 patients show drops in their leukocyte counts, and elevated white blood cell counts is a poor prognostic sign, according to the release.
The Centers for Disease Control and Prevention lists vulnerable populations at the greatest risk for more serious outcomes; these include the elderly and those with severe chronic health conditions, such as heart disease, lung disease, diabetes, decompensated cirrhosis, HIV with low CD4 counts, and immunosuppression (including liver and other solid organ transplant recipients), are at higher risk of developing more serious illness. In addition pregnancy may provide added risk.
Specific advice for the gastroenterology profession
The joint statement urges that practitioners strongly consider rescheduling elective nonurgent endoscopic procedures, although some nonurgent procedures are higher priority and may need to be performed, including cancer evaluations, prosthetic removals, and evaluation of significant symptoms. “Of note, the Surgeon General on 3/14/20 advised hospitals to postpone all elective surgeries,” the document states.
Patient concerns
In all cases, patients should be prescreened for high-risk exposure or symptoms. This includes asking about history of fever or respiratory symptoms, family members or close contacts with similar symptoms, any contact with a confirmed case of COVID-19, and recent travel to a high-risk area. “Avoid bringing patients (or their escorts) into the medical facility who are over age 65 or have one of the CDC recognized risks listed above,” the societies advise.
Check body temperature of the patient upon arrival at endoscopy unit or clinic, and keep all patients at an appropriate distance from each other (6 feet is recommended) throughout the entire time in the endoscopy unit.
“For COVID-19 positive patients, or those awaiting test results, isolation precautions should be taken with procedures performed in negative pressure rooms,” according to the statement.
In addition, use telemedicine where possible in elective cases, and consider phone follow-up after any procedures at 7 and 14 days to ask about new diagnosis of COVID-19 or development of its symptoms, .
Those patients who are on immunosuppressive drugs for inflammatory bowel disease and autoimmune hepatitis should continue taking their medications because the risk of disease flare outweighs the chance of contracting coronavirus, according to the document. In addition, these patients should be advised to follow CDC guidelines for at-risk groups by avoiding crowds and limiting travel.
Protection of practitioners
Key factors in ensuring practitioner safety and maintaining practice functionality are discussed by the joint document. In particular, appropriate personal protective equipment (PPE) should be worn by all members of the endoscopy team: gloves, mask, eye shield/goggles, face shields, and gown, but practitioners should also be aware of how to put on and take off PPE appropriately.
“Conservation of PPE is critical. Only essential personnel should be present in cases. Consider extended use or reuse of surgical masks and eye protection in accordance with hospital policies,” the document recommends.
“It is important to address our collective staff needs and institute policies that protect our workforce.” To that end, the document recommends that centers should strategically assign available personnel in order to minimize concomitant exposure of those with similar or unique skill sets. This includes the use of nonphysician practitioners and fellows that cannot participate in cases for screening and triaging patients, or performing virtual visits.
Coming at a time of pandemic, when gastrointestinal symptoms have been recognized as a more common symptom of COVID-19 than previously expected and liver damage has been noted as a potential repercussion of SARS-CoV-2 infection, these clinical insights provide a template for gastroenterologists and related professionals for dealing with their patients and keeping themselves safe under dramatically changed circumstances.
The partnered organizations, AASLD, ACG, AGA, and ASGE, are committed to providing updated COVID-19 information as appropriate. However: “Given the evolving and fluid nature of the situation, institutions, hospitals and clinics have also been formulating their own local guidelines, so we urge you to follow the evolving CDC recommendations and your local requirements,” according to the AGA website announcement.
In addition to the joint communication, the society websites each offer additional COVID-19 information. The AGA practice updates on the COVID-19 webpage provides information about announcements, such as the cancellation of Digestive Disease Week® in May, a location for AGA members to discuss their COVID-19 experiences and share advice, and links to the CDC COVID-19 updates.
SOURCE: American Gastroenterological Association et al. March 2020, COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers.
Amid the growing SARS-CoV-2 pandemic, currently in its expansive growth phase in the United States, the American Gastroenterological Association (AGA), the American Association for the Study of Liver Diseases (AASLD), the American College of Gastroenterology (ACG), and the American Society for Gastrointestinal Endoscopy (ASGE) have jointly released “COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers,” which can be found on the websites of the various societies.
“The purpose of this communication is to jointly provide you with up to date COVID-19 information in order to maintain the highest level of health and safety for our patients, staff, community, and ourselves,” according to the AGA website announcement.
In particular, the societies point out that there is recent evidence suggesting the potential for coronavirus transmission through droplets and perhaps fecal shedding, which pose potential risks in particular during endoscopy and colonoscopy procedures to other patients, endoscopy personnel, and practitioners.
Relevant clinical factors related to COVID-19 are discussed, including the fact that asymptomatic spread can occur during the prodromal phase (the mean incubation period is approximately 5 days, with a range of 0-14 days), with viral shedding greatest when symptoms begin.
Between 20% and 30% of patients with COVID-19 infection show abnormal liver enzymes. In addition, COVID-19 patients show drops in their leukocyte counts, and elevated white blood cell counts is a poor prognostic sign, according to the release.
The Centers for Disease Control and Prevention lists vulnerable populations at the greatest risk for more serious outcomes; these include the elderly and those with severe chronic health conditions, such as heart disease, lung disease, diabetes, decompensated cirrhosis, HIV with low CD4 counts, and immunosuppression (including liver and other solid organ transplant recipients), are at higher risk of developing more serious illness. In addition pregnancy may provide added risk.
Specific advice for the gastroenterology profession
The joint statement urges that practitioners strongly consider rescheduling elective nonurgent endoscopic procedures, although some nonurgent procedures are higher priority and may need to be performed, including cancer evaluations, prosthetic removals, and evaluation of significant symptoms. “Of note, the Surgeon General on 3/14/20 advised hospitals to postpone all elective surgeries,” the document states.
Patient concerns
In all cases, patients should be prescreened for high-risk exposure or symptoms. This includes asking about history of fever or respiratory symptoms, family members or close contacts with similar symptoms, any contact with a confirmed case of COVID-19, and recent travel to a high-risk area. “Avoid bringing patients (or their escorts) into the medical facility who are over age 65 or have one of the CDC recognized risks listed above,” the societies advise.
Check body temperature of the patient upon arrival at endoscopy unit or clinic, and keep all patients at an appropriate distance from each other (6 feet is recommended) throughout the entire time in the endoscopy unit.
“For COVID-19 positive patients, or those awaiting test results, isolation precautions should be taken with procedures performed in negative pressure rooms,” according to the statement.
In addition, use telemedicine where possible in elective cases, and consider phone follow-up after any procedures at 7 and 14 days to ask about new diagnosis of COVID-19 or development of its symptoms, .
Those patients who are on immunosuppressive drugs for inflammatory bowel disease and autoimmune hepatitis should continue taking their medications because the risk of disease flare outweighs the chance of contracting coronavirus, according to the document. In addition, these patients should be advised to follow CDC guidelines for at-risk groups by avoiding crowds and limiting travel.
Protection of practitioners
Key factors in ensuring practitioner safety and maintaining practice functionality are discussed by the joint document. In particular, appropriate personal protective equipment (PPE) should be worn by all members of the endoscopy team: gloves, mask, eye shield/goggles, face shields, and gown, but practitioners should also be aware of how to put on and take off PPE appropriately.
“Conservation of PPE is critical. Only essential personnel should be present in cases. Consider extended use or reuse of surgical masks and eye protection in accordance with hospital policies,” the document recommends.
“It is important to address our collective staff needs and institute policies that protect our workforce.” To that end, the document recommends that centers should strategically assign available personnel in order to minimize concomitant exposure of those with similar or unique skill sets. This includes the use of nonphysician practitioners and fellows that cannot participate in cases for screening and triaging patients, or performing virtual visits.
Coming at a time of pandemic, when gastrointestinal symptoms have been recognized as a more common symptom of COVID-19 than previously expected and liver damage has been noted as a potential repercussion of SARS-CoV-2 infection, these clinical insights provide a template for gastroenterologists and related professionals for dealing with their patients and keeping themselves safe under dramatically changed circumstances.
The partnered organizations, AASLD, ACG, AGA, and ASGE, are committed to providing updated COVID-19 information as appropriate. However: “Given the evolving and fluid nature of the situation, institutions, hospitals and clinics have also been formulating their own local guidelines, so we urge you to follow the evolving CDC recommendations and your local requirements,” according to the AGA website announcement.
In addition to the joint communication, the society websites each offer additional COVID-19 information. The AGA practice updates on the COVID-19 webpage provides information about announcements, such as the cancellation of Digestive Disease Week® in May, a location for AGA members to discuss their COVID-19 experiences and share advice, and links to the CDC COVID-19 updates.
SOURCE: American Gastroenterological Association et al. March 2020, COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers.
Amid the growing SARS-CoV-2 pandemic, currently in its expansive growth phase in the United States, the American Gastroenterological Association (AGA), the American Association for the Study of Liver Diseases (AASLD), the American College of Gastroenterology (ACG), and the American Society for Gastrointestinal Endoscopy (ASGE) have jointly released “COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers,” which can be found on the websites of the various societies.
“The purpose of this communication is to jointly provide you with up to date COVID-19 information in order to maintain the highest level of health and safety for our patients, staff, community, and ourselves,” according to the AGA website announcement.
In particular, the societies point out that there is recent evidence suggesting the potential for coronavirus transmission through droplets and perhaps fecal shedding, which pose potential risks in particular during endoscopy and colonoscopy procedures to other patients, endoscopy personnel, and practitioners.
Relevant clinical factors related to COVID-19 are discussed, including the fact that asymptomatic spread can occur during the prodromal phase (the mean incubation period is approximately 5 days, with a range of 0-14 days), with viral shedding greatest when symptoms begin.
Between 20% and 30% of patients with COVID-19 infection show abnormal liver enzymes. In addition, COVID-19 patients show drops in their leukocyte counts, and elevated white blood cell counts is a poor prognostic sign, according to the release.
The Centers for Disease Control and Prevention lists vulnerable populations at the greatest risk for more serious outcomes; these include the elderly and those with severe chronic health conditions, such as heart disease, lung disease, diabetes, decompensated cirrhosis, HIV with low CD4 counts, and immunosuppression (including liver and other solid organ transplant recipients), are at higher risk of developing more serious illness. In addition pregnancy may provide added risk.
Specific advice for the gastroenterology profession
The joint statement urges that practitioners strongly consider rescheduling elective nonurgent endoscopic procedures, although some nonurgent procedures are higher priority and may need to be performed, including cancer evaluations, prosthetic removals, and evaluation of significant symptoms. “Of note, the Surgeon General on 3/14/20 advised hospitals to postpone all elective surgeries,” the document states.
Patient concerns
In all cases, patients should be prescreened for high-risk exposure or symptoms. This includes asking about history of fever or respiratory symptoms, family members or close contacts with similar symptoms, any contact with a confirmed case of COVID-19, and recent travel to a high-risk area. “Avoid bringing patients (or their escorts) into the medical facility who are over age 65 or have one of the CDC recognized risks listed above,” the societies advise.
Check body temperature of the patient upon arrival at endoscopy unit or clinic, and keep all patients at an appropriate distance from each other (6 feet is recommended) throughout the entire time in the endoscopy unit.
“For COVID-19 positive patients, or those awaiting test results, isolation precautions should be taken with procedures performed in negative pressure rooms,” according to the statement.
In addition, use telemedicine where possible in elective cases, and consider phone follow-up after any procedures at 7 and 14 days to ask about new diagnosis of COVID-19 or development of its symptoms, .
Those patients who are on immunosuppressive drugs for inflammatory bowel disease and autoimmune hepatitis should continue taking their medications because the risk of disease flare outweighs the chance of contracting coronavirus, according to the document. In addition, these patients should be advised to follow CDC guidelines for at-risk groups by avoiding crowds and limiting travel.
Protection of practitioners
Key factors in ensuring practitioner safety and maintaining practice functionality are discussed by the joint document. In particular, appropriate personal protective equipment (PPE) should be worn by all members of the endoscopy team: gloves, mask, eye shield/goggles, face shields, and gown, but practitioners should also be aware of how to put on and take off PPE appropriately.
“Conservation of PPE is critical. Only essential personnel should be present in cases. Consider extended use or reuse of surgical masks and eye protection in accordance with hospital policies,” the document recommends.
“It is important to address our collective staff needs and institute policies that protect our workforce.” To that end, the document recommends that centers should strategically assign available personnel in order to minimize concomitant exposure of those with similar or unique skill sets. This includes the use of nonphysician practitioners and fellows that cannot participate in cases for screening and triaging patients, or performing virtual visits.
Coming at a time of pandemic, when gastrointestinal symptoms have been recognized as a more common symptom of COVID-19 than previously expected and liver damage has been noted as a potential repercussion of SARS-CoV-2 infection, these clinical insights provide a template for gastroenterologists and related professionals for dealing with their patients and keeping themselves safe under dramatically changed circumstances.
The partnered organizations, AASLD, ACG, AGA, and ASGE, are committed to providing updated COVID-19 information as appropriate. However: “Given the evolving and fluid nature of the situation, institutions, hospitals and clinics have also been formulating their own local guidelines, so we urge you to follow the evolving CDC recommendations and your local requirements,” according to the AGA website announcement.
In addition to the joint communication, the society websites each offer additional COVID-19 information. The AGA practice updates on the COVID-19 webpage provides information about announcements, such as the cancellation of Digestive Disease Week® in May, a location for AGA members to discuss their COVID-19 experiences and share advice, and links to the CDC COVID-19 updates.
SOURCE: American Gastroenterological Association et al. March 2020, COVID-19 Clinical Insights for Our Community of Gastroenterologists and Gastroenterology Care Providers.
Patients with COVID-19 may face risk for liver injury
Patients with COVID-19 may be at risk for liver injury, but mechanisms of damage remain unclear, according to investigators.
Proposed mechanisms include direct virus-induced effects, immune-induced damage due to excessive inflammatory responses, and drug-induced injury, reported lead author Ling Xu of Huazhong University of Science and Technology, Wuhan, China, and colleagues.
“From a clinical perspective, in addition to actively dealing with the primary disease caused by coronavirus infection, attention should also be paid to monitor the occurrence of liver injury, and to the application of drugs which may induce liver damage,” the investigators wrote in Liver International. “Patients with liver damage are advised to be treated with drugs that could both protect liver functions and inhibit inflammatory responses, such as ammonium glycyrrhizinate, which may, in turn, accelerate the process of disease recovery.”
The review of liver injury associated with major pathogenic coronaviruses included severe acute respiratory syndrome coronavirus (SARS-CoV), the Middle East respiratory syndrome coronavirus (MERS-CoV), and the newly emergent SARS-CoV-2, which causes COVID-19.
In cases of COVID-19, reported incidence of liver injury ranges from 15% to 53%, based on elevations of alanine transaminase (ALT) and aspartate aminotransferase (AST), along with slightly elevated bilirubin levels. In severe cases, albumin decreases have also been documented.
Liver injury appears to be significantly more common among those with severe infection. In one cohort of 82 patients who died from COVID-19, the incidence of liver injury was 78%, while another study of 36 nonsurvivors reported a rate of 58%.
According to the investigators, both bile duct epithelial cells and liver cells express angiotensin converting enzyme II (ACE2), which is an entry receptor for SARS-CoV-2; however, expression of ACE2 in bile duct cells is “much higher” than in liver cells, and comparable with alveolar type 2 cells in the lungs.
“Bile duct epithelial cells are known to play important roles in liver regeneration and immune response,” the investigators noted.
Beyond direct- and immune-induced effects of COVID-19, postmortem findings suggest that drug-induced liver injury may also be a possibility, with a number of theoretical culprits, including antibiotics, steroids, and antivirals. Although the investigators emphasized that data are insufficient to pinpoint an exact agent, they highlighted a recent preprint study, which reported a significantly higher rate of lopinavir/ritonavir administration among patients with abnormal liver function, compared with those who had normal liver function (56.1% vs. 25%; P = .009).
“Drug-induced liver injury during the treatment of coronavirus infection should not be ignored and needs to be carefully investigated,” the investigators concluded.
Fundamental Research Funds for the Central Universities supported the work. The investigators reported no conflicts of interest.
SOURCE: Xu L et al. Liver Int. 2020 Mar 14. doi: 10.1111/liv.14435.
Patients with COVID-19 may be at risk for liver injury, but mechanisms of damage remain unclear, according to investigators.
Proposed mechanisms include direct virus-induced effects, immune-induced damage due to excessive inflammatory responses, and drug-induced injury, reported lead author Ling Xu of Huazhong University of Science and Technology, Wuhan, China, and colleagues.
“From a clinical perspective, in addition to actively dealing with the primary disease caused by coronavirus infection, attention should also be paid to monitor the occurrence of liver injury, and to the application of drugs which may induce liver damage,” the investigators wrote in Liver International. “Patients with liver damage are advised to be treated with drugs that could both protect liver functions and inhibit inflammatory responses, such as ammonium glycyrrhizinate, which may, in turn, accelerate the process of disease recovery.”
The review of liver injury associated with major pathogenic coronaviruses included severe acute respiratory syndrome coronavirus (SARS-CoV), the Middle East respiratory syndrome coronavirus (MERS-CoV), and the newly emergent SARS-CoV-2, which causes COVID-19.
In cases of COVID-19, reported incidence of liver injury ranges from 15% to 53%, based on elevations of alanine transaminase (ALT) and aspartate aminotransferase (AST), along with slightly elevated bilirubin levels. In severe cases, albumin decreases have also been documented.
Liver injury appears to be significantly more common among those with severe infection. In one cohort of 82 patients who died from COVID-19, the incidence of liver injury was 78%, while another study of 36 nonsurvivors reported a rate of 58%.
According to the investigators, both bile duct epithelial cells and liver cells express angiotensin converting enzyme II (ACE2), which is an entry receptor for SARS-CoV-2; however, expression of ACE2 in bile duct cells is “much higher” than in liver cells, and comparable with alveolar type 2 cells in the lungs.
“Bile duct epithelial cells are known to play important roles in liver regeneration and immune response,” the investigators noted.
Beyond direct- and immune-induced effects of COVID-19, postmortem findings suggest that drug-induced liver injury may also be a possibility, with a number of theoretical culprits, including antibiotics, steroids, and antivirals. Although the investigators emphasized that data are insufficient to pinpoint an exact agent, they highlighted a recent preprint study, which reported a significantly higher rate of lopinavir/ritonavir administration among patients with abnormal liver function, compared with those who had normal liver function (56.1% vs. 25%; P = .009).
“Drug-induced liver injury during the treatment of coronavirus infection should not be ignored and needs to be carefully investigated,” the investigators concluded.
Fundamental Research Funds for the Central Universities supported the work. The investigators reported no conflicts of interest.
SOURCE: Xu L et al. Liver Int. 2020 Mar 14. doi: 10.1111/liv.14435.
Patients with COVID-19 may be at risk for liver injury, but mechanisms of damage remain unclear, according to investigators.
Proposed mechanisms include direct virus-induced effects, immune-induced damage due to excessive inflammatory responses, and drug-induced injury, reported lead author Ling Xu of Huazhong University of Science and Technology, Wuhan, China, and colleagues.
“From a clinical perspective, in addition to actively dealing with the primary disease caused by coronavirus infection, attention should also be paid to monitor the occurrence of liver injury, and to the application of drugs which may induce liver damage,” the investigators wrote in Liver International. “Patients with liver damage are advised to be treated with drugs that could both protect liver functions and inhibit inflammatory responses, such as ammonium glycyrrhizinate, which may, in turn, accelerate the process of disease recovery.”
The review of liver injury associated with major pathogenic coronaviruses included severe acute respiratory syndrome coronavirus (SARS-CoV), the Middle East respiratory syndrome coronavirus (MERS-CoV), and the newly emergent SARS-CoV-2, which causes COVID-19.
In cases of COVID-19, reported incidence of liver injury ranges from 15% to 53%, based on elevations of alanine transaminase (ALT) and aspartate aminotransferase (AST), along with slightly elevated bilirubin levels. In severe cases, albumin decreases have also been documented.
Liver injury appears to be significantly more common among those with severe infection. In one cohort of 82 patients who died from COVID-19, the incidence of liver injury was 78%, while another study of 36 nonsurvivors reported a rate of 58%.
According to the investigators, both bile duct epithelial cells and liver cells express angiotensin converting enzyme II (ACE2), which is an entry receptor for SARS-CoV-2; however, expression of ACE2 in bile duct cells is “much higher” than in liver cells, and comparable with alveolar type 2 cells in the lungs.
“Bile duct epithelial cells are known to play important roles in liver regeneration and immune response,” the investigators noted.
Beyond direct- and immune-induced effects of COVID-19, postmortem findings suggest that drug-induced liver injury may also be a possibility, with a number of theoretical culprits, including antibiotics, steroids, and antivirals. Although the investigators emphasized that data are insufficient to pinpoint an exact agent, they highlighted a recent preprint study, which reported a significantly higher rate of lopinavir/ritonavir administration among patients with abnormal liver function, compared with those who had normal liver function (56.1% vs. 25%; P = .009).
“Drug-induced liver injury during the treatment of coronavirus infection should not be ignored and needs to be carefully investigated,” the investigators concluded.
Fundamental Research Funds for the Central Universities supported the work. The investigators reported no conflicts of interest.
SOURCE: Xu L et al. Liver Int. 2020 Mar 14. doi: 10.1111/liv.14435.
FROM LIVER INTERNATIONAL
COVID-19 in China: Children have less severe disease, but are vulnerable
Clinical manifestations of COVID-19 infection among children in mainland China generally have been less severe than those among adults, but children of all ages – and infants in particular – are vulnerable to infection, according to a review of 2,143 cases.
Further, infection patterns in the nationwide series of all pediatric patients reported to the Chinese Center for Disease Control and Prevention from Jan. 16 to Feb. 8, 2020, provide strong evidence of human-to-human transmission, Yuanyuan Dong, MPH, a research assistant at Shanghai Children’s Medical Center, Shanghai Jiao Tong University, China, and colleagues reported in Pediatrics.
Of the 2,143 patients included in the review, 57% were boys and the median age was 7 years; 34% had laboratory-confirmed infection and 67% had suspected infection. More than 90% had asymptomatic, mild, or moderate disease (4%, 51%, and 39%, respectively), and 46% were from Hubei Province, where the first cases were reported, the investigators found.
The median time from illness onset to diagnosis was 2 days, and there was a trend of rapid increase of disease at the early stage of the epidemic – with rapid spread from Hubei Province to surrounding provinces – followed by a gradual and steady decrease, they noted.
“The total number of pediatric patients increased remarkably between mid-January and early February, peaked around February 1, and then declined since early February 2020,” they wrote. The proportion of severe and critical cases was 11% for infants under 1 year of age, compared with 7% for those aged 1-5 years; 4% for those aged 6-10 years; 4% for those 11-15 years; and 3% for those 16 years and older.
As of Feb. 8, 2020, only one child in this group of study patients died and most cases of COVID-19 symptoms were mild. There were many fewer severe and critical cases among the children (6%), compared with those reported in adult patients in other studies (19%). “It suggests that, compared with adult patients, clinical manifestations of children’s COVID-19 may be less severe,” the investigators suggested.
“As most of these children were likely to expose themselves to family members and/or other children with COVID-19, it clearly indicates person-to-person transmission ” of novel coronavirus 2019, they said, adding that similar evidence of such transmission also has been reported from studies of adult patients.
The reasons for reduced severity in children versus adults remain unclear, but may be related to both exposure and host factors, Ms. Dong and associates said. “Children were usually well cared for at home and might have relatively [fewer] opportunities to expose themselves to pathogens and/or sick patients.”
The findings demonstrate a pediatric distribution that varied across time and space, with most cases concentrated in the Hubei province and surrounding areas. No significant gender-related difference in infection rates was observed, and although the median patient age was 7 years, the range was 1 day to 18 years, suggesting that “all ages at childhood were susceptible” to the virus, they added.
The declining number of cases over time further suggests that disease control measures implemented by the government were effective, and that cases will “continue to decline, and finally stop in the near future unless sustained human-to-human transmissions occur,” Ms. Dong and associates concluded.
In an accompanying editorial, Andrea T. Cruz, MD, of Baylor College of Medicine, Houston, and Steven L. Zeichner, MD, PhD, of the University of Virginia, Charlottesville, said the findings regarding reduced severity among children versus adults with novel coronavirus 2019 infection are consistent with data on non-COVID-19 coronavirus.
They pointed out that Ms. Dong and associates did find that 13% of virologically-confirmed cases had asymptomatic infection, “a rate that almost certainly understates the true rate of asymptomatic infection, since many asymptomatic children are unlikely to be tested.”
Of the symptomatic children, “5% had dyspnea or hypoxemia (a substantially lower percentage than what has been reported for adults) and 0.6% progressed to acute respiratory distress syndrome (ARDS) or multiorgan system dysfunction”; this also is at a lower rate than seen in adults, they said.
Very young children –infants or children in preschool – were more likely to have severe clinical manifestations than children who were older.
Thus, it appears that certain subpopulations of children are at increased risk for more significant COVID-19 illness: “younger age, underlying pulmonary pathology, and immunocompromising conditions,” Dr. Cruz and Dr. Zeichner suggested.
The two editorialists said the findings suggest children “may play a major role in community-based viral transmission.” Evidence suggests that children may have more upper respiratory tract involvement and that fecal shedding may occur for several weeks after diagnosis; this raises concerns about fecal-oral transmission, particularly for infants and children, and about viral replication in the gastrointestinal tract, they said. This has substantial implications for community spread in day care centers, schools, and in the home.
A great deal has been learned about COVID-19 in a short time, but there still is much to learn about the effect of the virus on children, the impact of children on viral spread, and about possible vertical transmission, they said.
“Widespread availability of testing will allow for us to more accurately describe the spectrum of illness and may result in adjustment of the apparent morbidity and mortality rate as fewer ill individuals are diagnosed,” Dr. Cruz and Dr. Zeichner wrote, adding that “rigorously gauging the impact of COVID-19 on children will be important to accurately model the pandemic and to ensure that appropriate resources are allocated to children requiring care.”
They noted that understanding differences in children versus adults with COVID-19 “can yield important insights into disease pathogenesis, informing management and the development of therapeutics.”
This study was partially supported by the Science and Technology Commission of Shanghai Municipality. The authors reported having no disclosures. Dr. Cruz and Dr. Zeichner are associate editors for Pediatrics. Dr. Cruz reported having no disclosures. Dr. Zeichner is an inventor of new technologies for the rapid production of vaccines, for which the University of Virginia has filed patent applications.
SOURCE: Dong Y et al. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0702; Cruz A and Zeichner S. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0834.
Clinical manifestations of COVID-19 infection among children in mainland China generally have been less severe than those among adults, but children of all ages – and infants in particular – are vulnerable to infection, according to a review of 2,143 cases.
Further, infection patterns in the nationwide series of all pediatric patients reported to the Chinese Center for Disease Control and Prevention from Jan. 16 to Feb. 8, 2020, provide strong evidence of human-to-human transmission, Yuanyuan Dong, MPH, a research assistant at Shanghai Children’s Medical Center, Shanghai Jiao Tong University, China, and colleagues reported in Pediatrics.
Of the 2,143 patients included in the review, 57% were boys and the median age was 7 years; 34% had laboratory-confirmed infection and 67% had suspected infection. More than 90% had asymptomatic, mild, or moderate disease (4%, 51%, and 39%, respectively), and 46% were from Hubei Province, where the first cases were reported, the investigators found.
The median time from illness onset to diagnosis was 2 days, and there was a trend of rapid increase of disease at the early stage of the epidemic – with rapid spread from Hubei Province to surrounding provinces – followed by a gradual and steady decrease, they noted.
“The total number of pediatric patients increased remarkably between mid-January and early February, peaked around February 1, and then declined since early February 2020,” they wrote. The proportion of severe and critical cases was 11% for infants under 1 year of age, compared with 7% for those aged 1-5 years; 4% for those aged 6-10 years; 4% for those 11-15 years; and 3% for those 16 years and older.
As of Feb. 8, 2020, only one child in this group of study patients died and most cases of COVID-19 symptoms were mild. There were many fewer severe and critical cases among the children (6%), compared with those reported in adult patients in other studies (19%). “It suggests that, compared with adult patients, clinical manifestations of children’s COVID-19 may be less severe,” the investigators suggested.
“As most of these children were likely to expose themselves to family members and/or other children with COVID-19, it clearly indicates person-to-person transmission ” of novel coronavirus 2019, they said, adding that similar evidence of such transmission also has been reported from studies of adult patients.
The reasons for reduced severity in children versus adults remain unclear, but may be related to both exposure and host factors, Ms. Dong and associates said. “Children were usually well cared for at home and might have relatively [fewer] opportunities to expose themselves to pathogens and/or sick patients.”
The findings demonstrate a pediatric distribution that varied across time and space, with most cases concentrated in the Hubei province and surrounding areas. No significant gender-related difference in infection rates was observed, and although the median patient age was 7 years, the range was 1 day to 18 years, suggesting that “all ages at childhood were susceptible” to the virus, they added.
The declining number of cases over time further suggests that disease control measures implemented by the government were effective, and that cases will “continue to decline, and finally stop in the near future unless sustained human-to-human transmissions occur,” Ms. Dong and associates concluded.
In an accompanying editorial, Andrea T. Cruz, MD, of Baylor College of Medicine, Houston, and Steven L. Zeichner, MD, PhD, of the University of Virginia, Charlottesville, said the findings regarding reduced severity among children versus adults with novel coronavirus 2019 infection are consistent with data on non-COVID-19 coronavirus.
They pointed out that Ms. Dong and associates did find that 13% of virologically-confirmed cases had asymptomatic infection, “a rate that almost certainly understates the true rate of asymptomatic infection, since many asymptomatic children are unlikely to be tested.”
Of the symptomatic children, “5% had dyspnea or hypoxemia (a substantially lower percentage than what has been reported for adults) and 0.6% progressed to acute respiratory distress syndrome (ARDS) or multiorgan system dysfunction”; this also is at a lower rate than seen in adults, they said.
Very young children –infants or children in preschool – were more likely to have severe clinical manifestations than children who were older.
Thus, it appears that certain subpopulations of children are at increased risk for more significant COVID-19 illness: “younger age, underlying pulmonary pathology, and immunocompromising conditions,” Dr. Cruz and Dr. Zeichner suggested.
The two editorialists said the findings suggest children “may play a major role in community-based viral transmission.” Evidence suggests that children may have more upper respiratory tract involvement and that fecal shedding may occur for several weeks after diagnosis; this raises concerns about fecal-oral transmission, particularly for infants and children, and about viral replication in the gastrointestinal tract, they said. This has substantial implications for community spread in day care centers, schools, and in the home.
A great deal has been learned about COVID-19 in a short time, but there still is much to learn about the effect of the virus on children, the impact of children on viral spread, and about possible vertical transmission, they said.
“Widespread availability of testing will allow for us to more accurately describe the spectrum of illness and may result in adjustment of the apparent morbidity and mortality rate as fewer ill individuals are diagnosed,” Dr. Cruz and Dr. Zeichner wrote, adding that “rigorously gauging the impact of COVID-19 on children will be important to accurately model the pandemic and to ensure that appropriate resources are allocated to children requiring care.”
They noted that understanding differences in children versus adults with COVID-19 “can yield important insights into disease pathogenesis, informing management and the development of therapeutics.”
This study was partially supported by the Science and Technology Commission of Shanghai Municipality. The authors reported having no disclosures. Dr. Cruz and Dr. Zeichner are associate editors for Pediatrics. Dr. Cruz reported having no disclosures. Dr. Zeichner is an inventor of new technologies for the rapid production of vaccines, for which the University of Virginia has filed patent applications.
SOURCE: Dong Y et al. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0702; Cruz A and Zeichner S. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0834.
Clinical manifestations of COVID-19 infection among children in mainland China generally have been less severe than those among adults, but children of all ages – and infants in particular – are vulnerable to infection, according to a review of 2,143 cases.
Further, infection patterns in the nationwide series of all pediatric patients reported to the Chinese Center for Disease Control and Prevention from Jan. 16 to Feb. 8, 2020, provide strong evidence of human-to-human transmission, Yuanyuan Dong, MPH, a research assistant at Shanghai Children’s Medical Center, Shanghai Jiao Tong University, China, and colleagues reported in Pediatrics.
Of the 2,143 patients included in the review, 57% were boys and the median age was 7 years; 34% had laboratory-confirmed infection and 67% had suspected infection. More than 90% had asymptomatic, mild, or moderate disease (4%, 51%, and 39%, respectively), and 46% were from Hubei Province, where the first cases were reported, the investigators found.
The median time from illness onset to diagnosis was 2 days, and there was a trend of rapid increase of disease at the early stage of the epidemic – with rapid spread from Hubei Province to surrounding provinces – followed by a gradual and steady decrease, they noted.
“The total number of pediatric patients increased remarkably between mid-January and early February, peaked around February 1, and then declined since early February 2020,” they wrote. The proportion of severe and critical cases was 11% for infants under 1 year of age, compared with 7% for those aged 1-5 years; 4% for those aged 6-10 years; 4% for those 11-15 years; and 3% for those 16 years and older.
As of Feb. 8, 2020, only one child in this group of study patients died and most cases of COVID-19 symptoms were mild. There were many fewer severe and critical cases among the children (6%), compared with those reported in adult patients in other studies (19%). “It suggests that, compared with adult patients, clinical manifestations of children’s COVID-19 may be less severe,” the investigators suggested.
“As most of these children were likely to expose themselves to family members and/or other children with COVID-19, it clearly indicates person-to-person transmission ” of novel coronavirus 2019, they said, adding that similar evidence of such transmission also has been reported from studies of adult patients.
The reasons for reduced severity in children versus adults remain unclear, but may be related to both exposure and host factors, Ms. Dong and associates said. “Children were usually well cared for at home and might have relatively [fewer] opportunities to expose themselves to pathogens and/or sick patients.”
The findings demonstrate a pediatric distribution that varied across time and space, with most cases concentrated in the Hubei province and surrounding areas. No significant gender-related difference in infection rates was observed, and although the median patient age was 7 years, the range was 1 day to 18 years, suggesting that “all ages at childhood were susceptible” to the virus, they added.
The declining number of cases over time further suggests that disease control measures implemented by the government were effective, and that cases will “continue to decline, and finally stop in the near future unless sustained human-to-human transmissions occur,” Ms. Dong and associates concluded.
In an accompanying editorial, Andrea T. Cruz, MD, of Baylor College of Medicine, Houston, and Steven L. Zeichner, MD, PhD, of the University of Virginia, Charlottesville, said the findings regarding reduced severity among children versus adults with novel coronavirus 2019 infection are consistent with data on non-COVID-19 coronavirus.
They pointed out that Ms. Dong and associates did find that 13% of virologically-confirmed cases had asymptomatic infection, “a rate that almost certainly understates the true rate of asymptomatic infection, since many asymptomatic children are unlikely to be tested.”
Of the symptomatic children, “5% had dyspnea or hypoxemia (a substantially lower percentage than what has been reported for adults) and 0.6% progressed to acute respiratory distress syndrome (ARDS) or multiorgan system dysfunction”; this also is at a lower rate than seen in adults, they said.
Very young children –infants or children in preschool – were more likely to have severe clinical manifestations than children who were older.
Thus, it appears that certain subpopulations of children are at increased risk for more significant COVID-19 illness: “younger age, underlying pulmonary pathology, and immunocompromising conditions,” Dr. Cruz and Dr. Zeichner suggested.
The two editorialists said the findings suggest children “may play a major role in community-based viral transmission.” Evidence suggests that children may have more upper respiratory tract involvement and that fecal shedding may occur for several weeks after diagnosis; this raises concerns about fecal-oral transmission, particularly for infants and children, and about viral replication in the gastrointestinal tract, they said. This has substantial implications for community spread in day care centers, schools, and in the home.
A great deal has been learned about COVID-19 in a short time, but there still is much to learn about the effect of the virus on children, the impact of children on viral spread, and about possible vertical transmission, they said.
“Widespread availability of testing will allow for us to more accurately describe the spectrum of illness and may result in adjustment of the apparent morbidity and mortality rate as fewer ill individuals are diagnosed,” Dr. Cruz and Dr. Zeichner wrote, adding that “rigorously gauging the impact of COVID-19 on children will be important to accurately model the pandemic and to ensure that appropriate resources are allocated to children requiring care.”
They noted that understanding differences in children versus adults with COVID-19 “can yield important insights into disease pathogenesis, informing management and the development of therapeutics.”
This study was partially supported by the Science and Technology Commission of Shanghai Municipality. The authors reported having no disclosures. Dr. Cruz and Dr. Zeichner are associate editors for Pediatrics. Dr. Cruz reported having no disclosures. Dr. Zeichner is an inventor of new technologies for the rapid production of vaccines, for which the University of Virginia has filed patent applications.
SOURCE: Dong Y et al. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0702; Cruz A and Zeichner S. Pediatrics. 2020 Mar 16. doi: 10.1542/peds.2020-0834.
FROM PEDIATRICS
ACP outlines guide for COVID-19 telehealth coding, billing
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.
and for handling clinician and staff absences due to illness or quarantine during the COVID-19 pandemic.
It strongly encourages practices to use telehealth, whenever possible, to mitigate exposure of patients who are sick or at risk because of other underlying conditions and to protect health care workers and the community from the spread of the disease.
The national organization of internists also recommends in the guidance that practices establish protocols and procedures for use by clinicians and all other staff in light of the pandemic.
The billing and coding tips are being offered to help practices deal with the rapidly changing situation surrounding the COVID-19 emergency, according to a statement from the ACP.
The coding-related guidance incorporates changes to a number of telehealth rules for Medicare beneficiaries, announced by the Centers for Medicare and Medicaid Services on March 17.
“Now in a full state of emergency, many Medicare restrictions related to telehealth have been lifted. Patients can be at home, and non-HIPAA compliant technology is allowed. There is no cost sharing for COVID-19 testing. In addition, to encourage use by patients, Medicare is allowing practices to waive cost sharing (copays and deductibles) for all telehealth services,” the organization said in the guidance. It notes, however, that the CMS does not currently reimburse for telephone calls.
The guidance includes details of the new ICD-10 codes, and stresses the importance of using the appropriate codes, given that some service cost-sharing has been waived for COVID-19 testing and treatment.
There is detailed coding guidance for virtual check-in, online evaluation and management, remote monitoring, originating site, and allowed technology and services.
In regard to clinician and staff absence due to illness or quarantine, the ACP says “practices may need to review emergency plans related to telework and to employee and clinician absence.” Among its recommendations are that practices and employers consider temporary adjustments to compensation formulas to accommodate those clinicians who experience a loss of income because they are paid based on production.
The organization emphasizes that, given the rapidly changing availability of testing for COVID-19, practices should contact their local health departments, hospitals, reference labs, or state health authorities to determine the status of their access to testing.
The full list of the ACP’s tips are available here.
Any new guidance for physicians will be posted on the ACP’s COVID-19 resource page.