User login
In-hospital mobility impairment in older MI patients predicts postdischarge functional decline
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Background: The ability to independently perform daily activities is highly valued by patients, yet it is commonly impaired in older adults after hospitalization for MI. Risk of functional decline in this population is not well understood, but may relate to reduced mobility while hospitalized.
Study design: Prospective cohort.
Setting: A total of 94 academic and community hospitals in the United States.
Synopsis: More than 3,000 adults aged 75 years and older who were hospitalized for acute myocardial infarction were enrolled in the prospective cohort SILVER-AMI; 2,587 patients within this cohort were evaluated for in-hospital mobility with the Timed “Up and Go” test. At 6-month follow-up, loss of independent performance of activities of daily living (ADL) and of the ability to walk 0.25 miles were both associated in a dose-dependent manner with in-hospital mobility. Severe in-hospital mobility impairment was associated with ADL decline with an adjusted odds ratio of 5.45 (95% confidence interval, 3.29-9.01).
While in-hospital mobility is predictive of future functional decline in this population, this observational study cannot establish whether attempts to improve mobility in hospitalized patients will prevent future functional decline.
Bottom line: Lower performance on the Timed “Up and Go” test of mobility among older patients hospitalized for MI is associated with functional decline 6 months after hospitalization.
Citation: Hajduk AM et al. Association between mobility measured during hospitalization and functional outcomes in older adults with acute myocardial infarction in the SILVER-AMI study. JAMA Intern Med. 2019 Oct 7. doi: 10.1001/jamainternmed.2019.4114.
Dr. Gerstenberger is a hospitalist and clinical assistant professor of medicine at the University of Utah, Salt Lake City.
Arcalyst gets FDA nod as first therapy for recurrent pericarditis
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved rilonacept (Arcalyst) to treat recurrent pericarditis and reduce the risk for recurrence in adults and children 12 years and older.
Approval of the weekly subcutaneous injection offers patients the first and only FDA-approved therapy for recurrent pericarditis, the agency said in a release.
Recurrent pericarditis is characterized by a remitting relapsing inflammation of the pericardium, and therapeutic options have been limited to NSAIDs, colchicine, and corticosteroids.
Rilonacept is a recombinant fusion protein that blocks interleukin-1 alpha and interleukin-1 beta signaling. It is already approved by the FDA to treat a group of rare inherited inflammatory diseases called cryopyrin-associated periodic syndromes.
The new indication is based on the pivotal phase 3 RHAPSODY trial in 86 patients with acute symptoms of recurrent pericarditis and systemic inflammation. After randomization, pericarditis recurred in 2 of 30 patients (7%) treated with rilonacept and in 23 of 31 patients (74%) treated with placebo, representing a 96% reduction in the relative risk for recurrence with rilonacept.
Patients who received rilonacept were also pain free or had minimal pain on 98% of trial days, whereas those who received placebo had minimal or no pain on 46% of trial days.
The most common adverse effects of rilonacept are injection-site reactions and upper-respiratory tract infections.
Serious, life-threatening infections have been reported in patients taking rilonacept, according to the FDA. Patients with active or chronic infections should not take the drug.
The FDA label also advises that patients should avoid live vaccines while taking rilonacept and that it should be discontinued if a hypersensitivity reaction occurs.
The commercial launch is expected in April, according to the company.
A version of this article first appeared on Medscape.com.
Implementing the AMI READMITS Risk Assessment Score to Increase Referrals Among Patients With Type I Myocardial Infarction
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
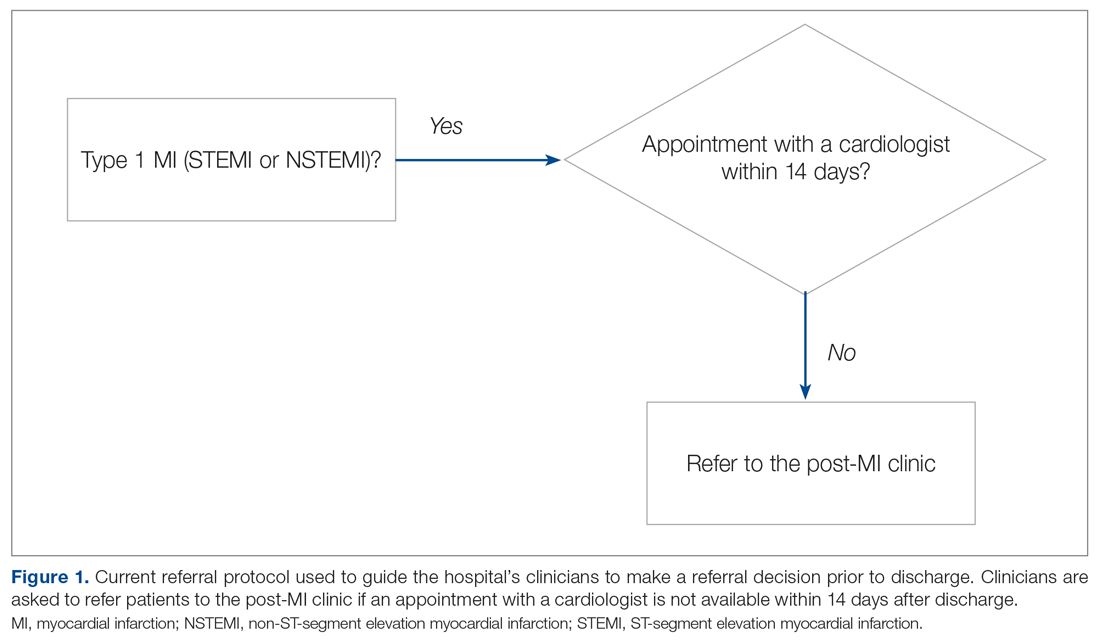
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.
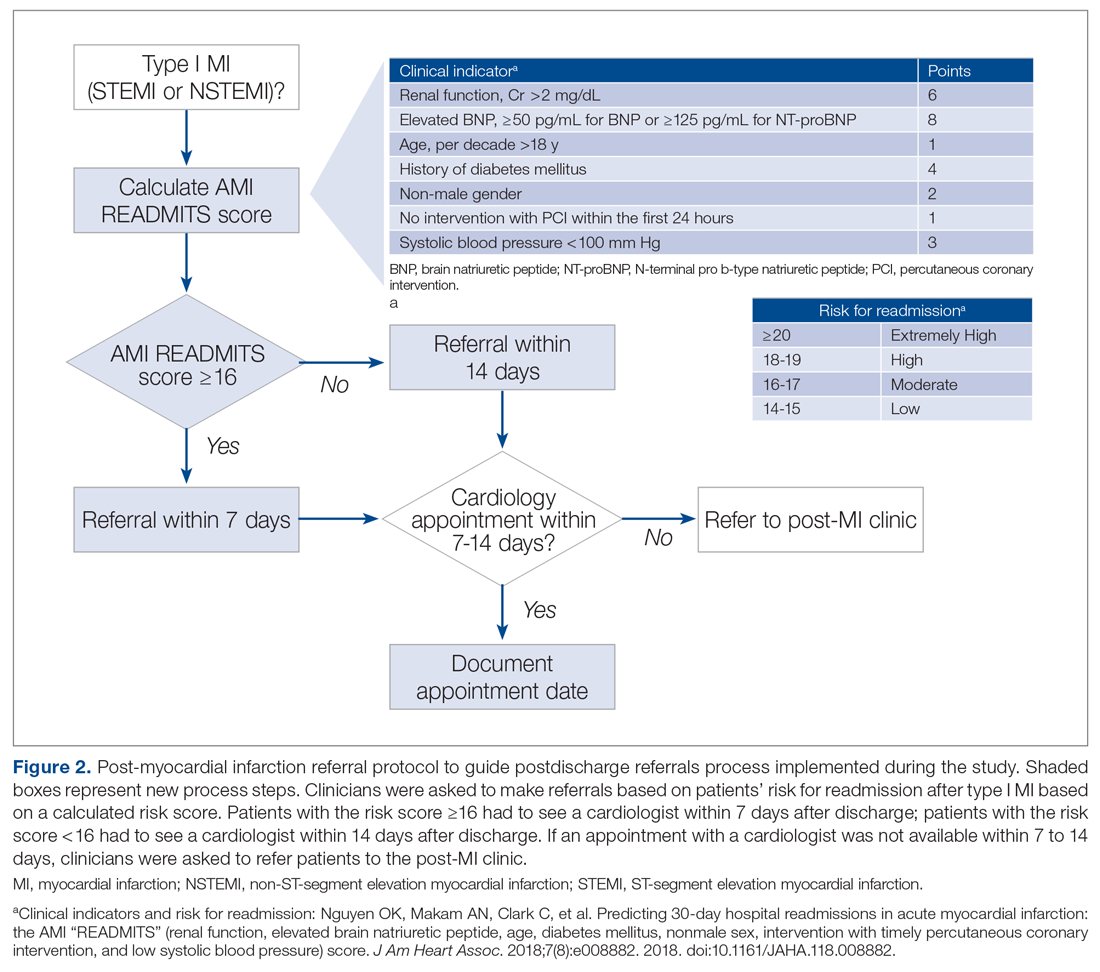
Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.
The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
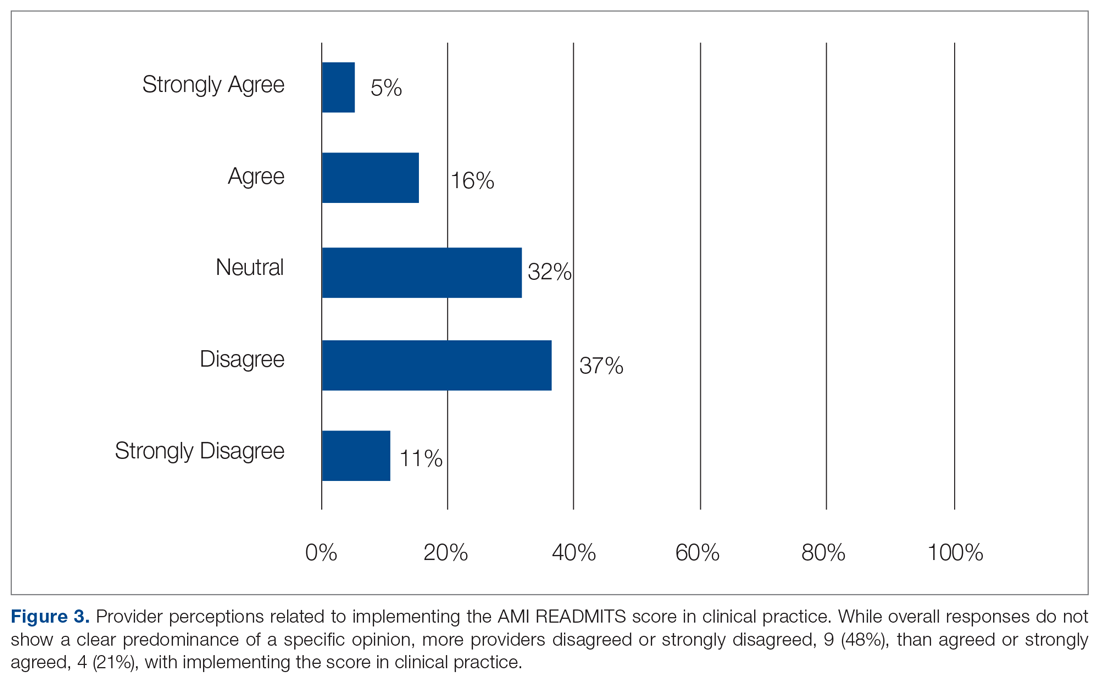
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”
Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.
Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.
The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”
Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
From The Johns Hopkins Hospital, Baltimore, MD (Dr. Muganlinskaya and Dr. Skojec, retired); The George Washington University, Washington, DC (Dr. Posey); and Johns Hopkins University, Baltimore, MD (Dr. Resar).
Abstract
Objective: Assessing the risk characteristics of patients with acute myocardial infarction (MI) can help providers make appropriate referral decisions. This quality improvement project sought to improve timely, appropriate referrals among patients with type I MI by adding a risk assessment, the AMI READMITS score, to the existing referral protocol.
Methods: Patients’ chart data were analyzed to assess changes in referrals and timely follow-up appointments from pre-intervention to intervention. A survey assessed providers’ satisfaction with the new referral protocol.
Results: Among 57 patients (n = 29 preintervention; n = 28 intervention), documented referrals increased significantly from 66% to 89% (χ2 = 4.571, df = 1, P = 0.033); and timely appointments increased by 10%, which was not significant (χ2 = 3.550, df = 2, P = 0.169). Most providers agreed that the new protocol was easy to use, useful in making referral decisions, and improved the referral process. All agreed the risk score should be incorporated into electronic clinical notes. Provider opinions related to implementing the risk score in clinical practice were mixed. Qualitative feedback suggests this was due to limited validation of the AMI READMITS score in reducing readmissions.
Conclusions: Our risk-based referral protocol helped to increase appropriate referrals among patients with type I MI. Provider adoption may be enhanced by incorporating the protocol into electronic clinical notes. Research to further validate the accuracy of the AMI READMITS score in predicting readmissions may support adoption of the protocol in clinical practice.
Keywords: quality improvement; type I myocardial infarction; referral process; readmission risk; risk assessment; chart review.
Early follow-up after discharge is an important strategy to reduce the risk of unplanned hospital readmissions among patients with various conditions.1-3 While patient confounding factors, such as chronic health problems, environment, socioeconomic status, and literacy, make it difficult to avoid all unplanned readmissions, early follow-up may help providers identify and appropriately manage some health-related issues, and as such is a pivotal element of a readmission prevention strategy.4 There is evidence that patients with non-ST elevation myocardial infarction (NSTEMI) who have an outpatient appointment with a physician within 7 days after discharge have a lower risk of 30-day readmission.5
Our hospital’s postmyocardial infarction clinic was created to prevent unplanned readmissions within 30 days after discharge among patients with type I myocardial infarction (MI). Since inception, the number of referrals has been much lower than expected. In 2018, the total number of patients discharged from the hospital with type I MI and any troponin I level above 0.40 ng/mL was 313. Most of these patients were discharged from the hospital’s cardiac units; however, only 91 referrals were made. To increase referrals, the cardiology nurse practitioners (NPs) developed a post-MI referral protocol (Figure 1). However, this protocol was not consistently used and referrals to the clinic remained low.
Evidence-based risk assessment tools have the potential to increase effective patient management. For example, cardiology providers at the hospital utilize various scores, such as CHA2DS2-VASc6 and the Society of Thoracic Surgery risk score,7 to plan patient management. Among the scores used to predict unplanned readmissions for MI patients, the most promising is the AMI READMITS score.8 Unlike other nonspecific prediction models, the AMI READMITS score was developed based on variables extracted from the electronic health records (EHRs) of patients who were hospitalized for MI and readmitted within 30 days after discharge. Recognizing the potential to increase referrals by integrating an MI-specific risk assessment, this quality improvement study modified the existing referral protocol to include the patients’ AMI READMITS score and recommendations for follow-up.
Currently, there are no clear recommendations on how soon after discharge patients with MI should undergo follow-up. As research data vary, we selected 7 days follow-up for patients from high risk groups based on the “See you in 7” initiative for patients with heart failure (HF) and MI,9,10 as well as evidence that patients with NSTEMI have a lower risk of 30-day readmission if they have follow-up within 7 days after discharge5; and we selected 14 days follow-up for patients from low-risk groups based on evidence that postdischarge follow-up within 14 days reduces risk of 30-day readmission in patients with acute myocardial infarction (AMI) and/or acutely decompensated HF.11
Methods
This project was designed to answer the following question: For adult patients with type I MI, does implementation of a readmission risk assessment referral protocol increase the percentage of referrals and appointments scheduled within a recommended time? Anticipated outcomes included: (1) increased referrals to a cardiologist or the post-MI clinic; (2) increased scheduled follow-up appointments within 7 to 14 days; (3) provider satisfaction with the usability and usefulness of the new protocol; and (4) consistent provider adoption of the new risk assessment referral protocol.
To evaluate the degree to which these outcomes were achieved, we reviewed patient charts for 2 months prior and 2 months during implementation of the new referral protocol. As shown in Figure 2, the new protocol added the following process steps to the existing protocol: calculation of the AMI READMITS score, recommendations for follow-up based on patients’ risk score, and guidance to refer patients to the post-MI clinic if patients did not have an appointment with a cardiologist within 7 to 14 days after discharge. Patients’ risk assessment scores were obtained from forms completed by clinicians during the intervention. Clinician’s perceptions related to the usability and usefulness of the new protocol and feedback related to its long-term adoption were assessed using a descriptive survey.
The institutional review board classified this project as a quality improvement project. To avoid potential loss of patient privacy, no identifiable data were collected, a unique identifier unrelated to patients’ records was generated for each patient, and data were saved on a password-protected cardiology office computer.
Population
The project population included all adult patients (≥ 18 years old) with type I MI who were admitted or transferred to the hospital, had a percutaneous coronary intervention (PCI), or were managed without PCI and discharged from the hospital’s cardiac care unit (CCU) and progressive cardiac care unit (PCCU). The criteria for type I MI included the “detection of a rise and/or fall of cardiac troponin with at least 1 value above the 99th percentile and with at least 1 of the following: symptoms of acute myocardial ischemia; new ischemic electrocardiographic (ECG) changes; development of new pathological Q waves; imaging evidence of new loss of viable myocardium or new regional wall motion abnormality in a pattern consistent with an ischemic etiology; identification of a coronary thrombus by angiography including intracoronary imaging or by autopsy.”12 The study excluded patients with type I MI who were referred for coronary bypass surgery.
Intervention
The revised risk assessment protocol was implemented within the CCU and PCCU. The lead investigator met with each provider to discuss the role of the post-MI clinic, current referral rates, the purpose of the project, and the new referral process to be completed during the project for each patient discharged with type I MI. Cardiology NPs, fellows, and residents were asked to use the risk-assessment form to calculate patients’ risk for readmission, and refer patients to the post-MI clinic if an appointment with a cardiologist was not available within 7 to 14 days after discharge. Every week during the intervention phase, the investigator sent reminder emails to ensure form completion. Providers were asked to calculate and write the score, the discharge and referral dates, where referrals were made (a cardiologist or the post-MI clinic), date of appointment, and reason for not scheduling an appointment or not referring on the risk assessment form, and to drop the completed forms in specific labeled boxes located at the CCU and PCCU work stations. The investigator collected the completed forms weekly. When the number of discharged patients did not match the number of completed forms, the investigator followed up with discharging providers to understand why.
Data and Data Collection
Data to determine whether the use of the new protocol increased discharge referrals among patients with type I MI within the recommended timeframes were collected by electronic chart review. Data included discharging unit, patients’ age, gender, admission and discharge date, diagnosis, referral to a cardiologist and the post-MI clinic, and appointment date. Clinical data needed to calculate the AMI READMITS score was also collected: PCI within 24 hours, serum creatinine, systolic blood pressure (SBP), brain natriuretic peptide (BNP), and diabetes status.
Data to assess provider satisfaction with the usability and usefulness of the new protocol were gathered through an online survey. The survey included 1 question related to the providers’ role, 1 question asking whether they used the risk assessment for each patient, and 5 Likert-items assessing the ease of usage. An additional open-ended question asked providers to share feedback related to integrating the AMI READMITS risk assessment score to the post-MI referral protocol long term.
To evaluate how consistently providers utilized the new referral protocol when discharging patients with type I MI, the number of completed forms was compared with the number of those patients who were discharged.
Statistical Analysis
Descriptive statistics were used to summarize patient demographics and to calculate the frequency of referrals before and during the intervention. Chi-square statistics were calculated to determine whether the change in percentage of referrals and timely referrals was significant. Descriptive statistics were used to determine the level of provider satisfaction related to each survey item. A content analysis method was used to synthesize themes from the open-ended question asking clinicians to share their feedback related to the new protocol.
Results
Fifty-seven patients met the study inclusion criteria: 29 patients during the preintervention phase and 28 patients during the intervention phase. There were 35 male (61.4%) and 22 female (38.6%) patients. Twenty-five patients (43.9%) were from age groups 41 through 60 years and 61 through 80 years, respectively, representing the majority of included patients. Seven patients (12.3%) were from the 81 years and older age group. There were no patients in the age group 18 through 40 years. Based on the AMI READMITS score calculation, 57.9% (n = 33) patients were from a low-risk group (includes extremely low and low risk for readmission) and 42.1% (n = 24) were from a high-risk group (includes moderate, high, and extremely high risk for readmission).
Provider adoption of the new protocol during the intervention was high. Referral forms were completed for 82% (n = 23) of the 28 patients during the intervention. Analysis findings showed a statistically significant increase in documented referrals after implementing the new referral protocol. During the preintervention phase, 66% (n = 19) of patients with type I MI were referred to see a cardiologist or an NP at a post-MI clinic and there was no documented referral for 34% (n = 10) of patients. During the intervention phase, 89% (n = 25) of patients were referred and there was no documented referral for 11% (n = 3) of patients. Chi-square results indicated that the increase in referrals was significant (χ2 = 4.571, df = 1, P = 0.033).
Data analysis examined whether patient referrals fell within the recommended timeframe of 7 days for the high-risk group (included moderate-to-extremely high risk) and 14 days for the low-risk group (included low-to-extremely low risk). During the preintervention phase, 31% (n = 9) of patient referrals were scheduled as recommended; 28% (n = 8) of patient referrals were scheduled but delayed; and there was no referral date documented for 41% (n = 12) of patients. During the intervention phase, referrals scheduled as recommended increased to 53% (n = 15); 25% (n = 7) of referrals were scheduled but delayed; and there was no referral date documented for 21.4% (n = 6) of patients. The change in appointments scheduled as recommended was not significant (χ2 = 3.550, df = 2, P = 0.169).
Surveys were emailed to 25 cardiology fellows and 3 cardiology NPs who participated in this study. Eighteen of the 28 clinicians (15 cardiology fellows and 3 cardiology NPs) responded for a response rate of 64%. One of several residents who rotated through the CCU and PCCU during the intervention also completed the survey, for a total of 19 participants. When asked if the protocol was easy to use, 79% agreed or strongly agreed. Eighteen of the 19 participants (95%) agreed or strongly agreed that the protocol was useful in making referral decisions. Sixty-eight percent agreed or strongly agreed that the AMI READMITS risk assessment score improves referral process. All participants agreed or strongly agreed that there should be an option to incorporate the AMI READMITS risk assessment score into electronic clinical notes. When asked whether the AMI READMITS risk score should be implemented in clinical practice, responses were mixed (Figure 3). A common theme among the 4 participants who responded with comments was the need for additional data to validate the usefulness of the AMI READMITS to reduce readmissions. In addition, 1 participant commented that “manual calculation [of the risk score] is not ideal.”
Discussion
This project demonstrated that implementing an evidence-based referral protocol integrating the AMI-READMITS score can increase timely postdischarge referrals among patients with type I MI. The percentage of appropriately scheduled appointments increased during the intervention phase; however, a relatively high number of appointments were scheduled outside of the recommended timeframe, similar to preintervention. Thus, while the new protocol increased referrals and provider documentation of these referrals, it appears that challenges in scheduling timely referral appointments remained. This project did not examine the reasons for delayed appointments.
The survey findings indicated that providers were generally satisfied with the usability and usefulness of the new risk assessment protocol. A large majority agreed or strongly agreed that it was easy to use and useful in making referral decisions, and most agreed or strongly agreed that it improves the referral process. Mixed opinions regarding implementing the AMI READMITS score in clinical practice, combined with qualitative findings, suggest that a lack of external validation of the AMI READMITS presents a barrier to its long-term adoption. All providers who participated in the survey agreed or strongly agreed that the risk assessment should be incorporated into electronic clinical notes. We have begun the process of working with the EHR vendor to automate the AMI risk-assessment within the referral work-flow, which will provide an opportunity for a follow-up quality improvement study.
This quality improvement project has several limitations. First, it implemented a small change in 2 inpatient units at 1 hospital using a simple pre- posttest design. Therefore, the findings are not generalizable to other settings. Prior to the intervention, some referrals may have been made without documentation. While the authors were able to trace undocumented referrals for patients who were referred to the post-MI clinic or to a cardiologist affiliated with the hospital, some patients may have been referred to cardiologists who were not affiliated with the hospital. Another limitation was that the self-created provider survey used was not tested in other clinical settings; thus, it cannot be determined whether the sensitivity and specificity of the survey questions are high. In addition, the clinical providers who participated in the study knew the study team, which may have influenced their behavior during the study period. Furthermore, the identified improvement in clinicians’ referral practices may not be sustainable due to the complexity and effort required to manually calculate the risk score. This limitation could be eliminated by integrating the risk score calculation into the EHR.
Conclusion
Early follow-up after discharge plays an important role in supporting patients’ self-management of some risk factors (ie, diet, weight, and smoking) and identifying gaps in postdischarge care which may lead to readmission. This project provides evidence that integrating the AMI READMITS risk assessment score into the referral process can help to guide discharge decision-making and increase timely, appropriate referrals for patients with MI. Integration of a specific risk assessment, such as the AMI READMITS, within the post-MI referral protocol may help clinicians make more efficient, educated referral decisions. Future studies should explore more specifically how and why the new protocol impacts clinicians’ decision-making and behavior related to post-MI referrals. In addition, future studies should investigate challenges associated with scheduling postdischarge appointments. It will be important to investigate how integration of the new protocol within the EHR may increase efficiency, consistency, and provider satisfaction with the new referral process. Additional research investigating the effects of the AMI READMITS score on readmissions reduction will be important to promote long-term adoption of the improved referral protocol in clinical practice.
Acknowledgments: The authors thank Shelly Conaway, ANP-BC, MSN, Angela Street, ANP-BC, MSN, Andrew Geis, ACNP-BC, MSN, Richard P. Jones II, MD, Eunice Young, MD, Joy Rothwell, MSN, RN-BC, Allison Olazo, MBA, MSN, RN-BC, Elizabeth Heck, RN-BC, and Matthew Trojanowski, MHA, MS, RRT, CSSBB for their support of this study.
Corresponding author: Nailya Muganlinskaya, DNP, MPH, ACNP-BC, MSN, The Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287; nmuganl1@jhmi.edu.
Financial disclosures: None.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.
1. Why it is important to improve care transitions? Society of Hospital Medicine. Accessed June 15, 2020. https://www.hospitalmedicine.org/clinical-topics/care-transitions/
2. Tong L, Arnold T, Yang J, et al. The association between outpatient follow-up visits and all-cause non-elective 30-day readmissions: a retrospective observational cohort study. PloS One. 2018;13(7):e0200691.
3. Jackson C, Shahsahebi M, Wedlake T, DuBard CA. Timeliness of outpatient follow-up: an evidence-based approach for planning after hospital discharge. Ann Fam Med. 2015;13(2):115-22.
4. Health Research & Educational Trust. Preventable Readmissions Change Package. American Hospital Association. Updated December 2015. Accessed June 10, 2020. https://www.aha.org/sites/default/files/hiin/HRETHEN_ChangePackage_Readmissions.pd
5. Tung Y-C, Chang G-M, Chang H-Y, Yu T-H. Relationship between early physician follow-up and 30-day readmission after acute myocardial infarction and heart failure. Plos One. 2017;12(1):e0170061.
6. Kaplan RM, Koehler J, Zieger PD, et al. Stroke risk as a function of atrial fibrillation duration and CHA2DS2-VASc score. Circulation. 2019;140(20):1639-46.
7. Balan P, Zhao Y, Johnson S, et al. The Society of Thoracic Surgery Risk Score as a predictor of 30-day mortality in transcatheter vs surgical aortic valve replacement: a single-center experience and its implications for the development of a TAVR risk-prediction model. J Invasive Cardiol. 2017;29(3):109-14.
8. Smith LN, Makam AN, Darden D, et al. Acute myocardial infarction readmission risk prediction models: A systematic review of model performance. Circ Cardiovasc Qual Outcomes9.9. 2018;11(1):e003885.
9. Baker H, Oliver-McNeil S, Deng L, Hummel SL. See you in 7: regional hospital collaboration and outcomes in Medicare heart failure patients. JACC Heart Fail. 2015;3(10):765-73.
10. Batten A, Jaeger C, Griffen D, et al. See you in 7: improving acute myocardial infarction follow-up care. BMJ Open Qual. 2018;7(2):e000296.
11. Lee DW, Armistead L, Coleman H, et al. Abstract 15387: Post-discharge follow-up within 14 days reduces 30-day hospital readmission rates in patients with acute myocardial infarction and/or acutely decompensated heart failure. Circulation. 2018;134 (1):A 15387.
12. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138 (20):e:618-51.
Dapagliflozin may cut risk of HF hospitalization in patients with type 2 diabetes
Background: Dapagliflozin is a selective inhibitor of sodium-glucose transporter 2 (SGLT2) in the kidney; the drug blocks glucose reabsorption in the proximal tubule. It is taken once daily by mouth. An initial study sponsored by AstraZeneca was published January 2019 in the New England Journal of Medicine – “Dapagliflozin and cardiovascular outcomes in type 2 diabetes.” Until recently there was not an FDA-approved indication for the drug.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: 882 clinical sites in 33 countries.
Synopsis: The study randomized approximately 17,000 patients to receive either dapagliflozin or placebo in addition to any other diabetes treatments prescribed by their physician. This study demonstrated its primary safety outcome, which was that patients on dapagliflozin did not have any more major adverse cardiac events (MACE), compared with placebo. There were two primary efficacy outcomes. First, there was no change in MACE with dapagliflozin, compared with placebo. Second, and pertinent to this drug’s approval, was that dapagliflozin reduced risk of hospitalization for heart failure (HF) from 5.8% to 4.9%, compared to placebo; this includes both HF with both preserved and reduced ejection fractions.
Bottom line: Dapagliflozin now has an FDA-approved indication to reduce hospitalizations for HF in patients with type 2 diabetes. Based on this study, the number needed to treat with dapagliflozin is 111 patients to prevent one hospitalization for HF.
Citation: Farxiga approved in the US to reduce the risk of hospitalization for heart failure in patients with type-2 diabetes. AstraZeneca Press Release, 2019 Oct 21.
Dr. Como is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Dapagliflozin is a selective inhibitor of sodium-glucose transporter 2 (SGLT2) in the kidney; the drug blocks glucose reabsorption in the proximal tubule. It is taken once daily by mouth. An initial study sponsored by AstraZeneca was published January 2019 in the New England Journal of Medicine – “Dapagliflozin and cardiovascular outcomes in type 2 diabetes.” Until recently there was not an FDA-approved indication for the drug.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: 882 clinical sites in 33 countries.
Synopsis: The study randomized approximately 17,000 patients to receive either dapagliflozin or placebo in addition to any other diabetes treatments prescribed by their physician. This study demonstrated its primary safety outcome, which was that patients on dapagliflozin did not have any more major adverse cardiac events (MACE), compared with placebo. There were two primary efficacy outcomes. First, there was no change in MACE with dapagliflozin, compared with placebo. Second, and pertinent to this drug’s approval, was that dapagliflozin reduced risk of hospitalization for heart failure (HF) from 5.8% to 4.9%, compared to placebo; this includes both HF with both preserved and reduced ejection fractions.
Bottom line: Dapagliflozin now has an FDA-approved indication to reduce hospitalizations for HF in patients with type 2 diabetes. Based on this study, the number needed to treat with dapagliflozin is 111 patients to prevent one hospitalization for HF.
Citation: Farxiga approved in the US to reduce the risk of hospitalization for heart failure in patients with type-2 diabetes. AstraZeneca Press Release, 2019 Oct 21.
Dr. Como is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Dapagliflozin is a selective inhibitor of sodium-glucose transporter 2 (SGLT2) in the kidney; the drug blocks glucose reabsorption in the proximal tubule. It is taken once daily by mouth. An initial study sponsored by AstraZeneca was published January 2019 in the New England Journal of Medicine – “Dapagliflozin and cardiovascular outcomes in type 2 diabetes.” Until recently there was not an FDA-approved indication for the drug.
Study design: Randomized, double-blind, placebo-controlled trial.
Setting: 882 clinical sites in 33 countries.
Synopsis: The study randomized approximately 17,000 patients to receive either dapagliflozin or placebo in addition to any other diabetes treatments prescribed by their physician. This study demonstrated its primary safety outcome, which was that patients on dapagliflozin did not have any more major adverse cardiac events (MACE), compared with placebo. There were two primary efficacy outcomes. First, there was no change in MACE with dapagliflozin, compared with placebo. Second, and pertinent to this drug’s approval, was that dapagliflozin reduced risk of hospitalization for heart failure (HF) from 5.8% to 4.9%, compared to placebo; this includes both HF with both preserved and reduced ejection fractions.
Bottom line: Dapagliflozin now has an FDA-approved indication to reduce hospitalizations for HF in patients with type 2 diabetes. Based on this study, the number needed to treat with dapagliflozin is 111 patients to prevent one hospitalization for HF.
Citation: Farxiga approved in the US to reduce the risk of hospitalization for heart failure in patients with type-2 diabetes. AstraZeneca Press Release, 2019 Oct 21.
Dr. Como is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Low concordance between troponin assays for ACS
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should be aware that the discordance between high-sensitivity cardiac troponin (hs-cTn) assays is significant enough that management recommendations may change, for example, for a patient assessed for suspected acute coronary syndrome (ACS) in one hospital and transferred to another that uses a different assay, according to a team of international researchers.
When hs-cTn concentrations were measured using the three Food and Drug Administration–approved assays, only 37.4% (384 of 1,027 samples) of blood samples were classified into the same analytical benchmark category.
“We didn’t expect such low concordance, to be honest, but I have to stress that this first assessment used just one-time blood testing and serial testing is what is more commonly recommended now,” said Júlia Karády, MD, from Massachusetts General Hospital and Harvard Medical School, both in Boston.
To see if concordance improved with serial testing, the researchers looked at the 242 patients for whom serial samples were available and saw concordance of management recommendations across assays rise to 74.8%.
“We tested the 0/2-hour algorithm and found that the overall agreement almost doubled, so I think that a very important message from our study is that serial testing improves the agreement between the assays in terms of clinical management and patient stratification,” said Dr. Karády.
Dr. Karády and colleagues published their findings in the Journal of the American College of Cardiology.
The researchers tested three assays referred to clinically as high-sensitivity assays: Elecsys 2010 platform (Roche Diagnostics); ARCHITECT i2000SR (Abbott Diagnostics); and hsVista (Siemens Diagnostics). All three have received FDA approval, starting with Elecsys in 2017.
The proportion of patients with similar management recommendations differed between the assays for both “rule-out” (87.2%, 73.1%, and 78.5% for Roche, Abbott, and Siemens, respectively) and “observe” (9.5%, 24%, and 17.8%; both P < .001). For the purposes of “rule-in,” no difference was noted (3.3%, 2.9%, and 3.7%).
“It’s important to note that this was a highly selected population of patients with an intermediate likelihood for ACS, not an all-comer population. This group comprises about 20% of the [emergency department] population and actually is the group we struggle with the most, which is hardest to diagnose because it excludes the very low– and very high–risk patients,” said Dr. Karády.
The patients included in this study all had suspected ACS and were enrolled in the ROMICAT-I and II trials.
Among 1,027 samples from 624 patients (mean age, 52.8 years; 39.4% women), samples were classified as below the limit of detection (LOD) in 56.3%, 10.4%, and 41.2% (P < .001) by Roche, Abbott, and Siemens, respectively.
The proportion of sample with a troponin measurement between LOD to the 99th percentile also differed significantly between the assays at 36.5%, 83.5%, and 52.6%, respectively (P < .001).
Only the proportion classified greater than 99th percentile did not differ (7.2%, 6.0%, and 6.2%; P = .114).
When the researchers looked at sex-specific difference, no differences were seen in rule-in numbers for men, but significant differences were seen for women.
“One possible explanation for this could be differences in the representation of men and women in the various reference populations used to develop the 99th percentile values for these assays,” suggested Dr. Karády.
They estimate around 30%-40% of U.S. centers are currently using high-sensitivity troponin assays and this number is “rapidly rising.”
The diagnostic algorithms developed for use with high-sensitivity assays, such as the 0/2-h algorithm, acknowledge differences in performance characteristics and recommend that assay-specific cut points be used for clinical decision-making rather than relying on generally applicable thresholds.
Joseph S. Alpert, MD, University of Arizona, Tucson, and coauthors of an accompanying editorial said the take-home message here is caveat emptor.
“First, ‘let the buyer (i.e., the clinician) beware’ when patients are transferred from one hospital to another, where different hs-cTn assays may be used,” they wrote. This is particularly true in women and in those with troponin levels in the “observe (gray zone)” clinical management recommendation.
Dr. Karády has received grant support from the Fulbright Visiting Researcher Grant and the Rosztoczy Foundation. One of the coauthors of the editorial comment consults or has consulted for most of the major diagnostic companies, including the manufacturers of the three assays tested in this study. Dr. Alpert disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ApoB may better predict mortality risk in statin-treated patients
A new study shows apolipoprotein B (apoB) and non-HDL cholesterol – but not LDL cholesterol – are associated with increased risk for all-cause mortality and myocardial infarction in patients taking statins.
Moreover, apoB was a more accurate marker of all-cause mortality risk than non-HDL or LDL cholesterol and was more accurate at identifying MI risk than LDL cholesterol.
“Any patient that comes to a doctor for evaluation, if statin treatment is sufficient, the doctor should look not only at LDL cholesterol but HDL cholesterol and apoB, if its available – that is the take-home message,” senior author Børge Grønne Nordestgaard, MD, DMSC, University of Copenhagen, said in an interview.
The findings are very relevant to clinical practice because international guidelines focus on LDL cholesterol and “many doctors are brainwashed that that is the only thing they should look at, just to keep LDL cholesterol down,” he said. “I’ve worked for years with triglyceride lipoproteins, what I call remnant cholesterol, and I think that the risk is very high also when you have high remnant cholesterol.”
Previous work has shown that apoB and non-HDL cholesterol better reflect atherosclerotic cardiovascular disease risk than LDL cholesterol. This is the first study, however, to show that elevated apoB and non-HDL cholesterol are associated with a higher risk for all-cause death in statin-treated patients with low LDL cholesterol, Dr. Nordestgaard noted.
The investigators compared outcomes among 13,015 statin-treated participants in the Copenhagen General Population Study using median baseline values of 92 mg/dL for apoB, 3.1 mmol/L (120 mg/dL) for non-HDL cholesterol, and 2.3 mmol/L (89 mg/dL) for LDL cholesterol. Over a median follow-up of 8 years, there were 2,499 deaths and 537 MIs.
As reported in the Journal of the American College of Cardiology, discordant apoB above the median with LDL cholesterol below was associated with a 21% increased risk for all-cause mortality (hazard ratio, 1.21; 95% confidence interval, 1.07-1.36) and 49% increased risk for MI (HR, 1.49; 95% CI, 1.15-1.92), compared with concordant apoB and LDL cholesterol below the medians.
Similar results were found for discordant non-HDL cholesterol above the median with low LDL cholesterol for all-cause mortality (HR, 1.18; 95% CI, 1.02-1.36) and MI (1.78; 95% CI, 1.35-2.34).
No such associations with mortality or MI were observed when LDL cholesterol was above the median and apoB or non-HDL below.
Additional analyses showed that high apoB with low non-HDL cholesterol was associated with a higher risk for all-cause mortality (HR, 1.21; 95% CI, 1.03-1.41), whereas high non-HDL cholesterol with low apoB was associated with a lower risk (HR, 0.75; 95% CI, 0.62-0.92).
Current guidelines define apoB greater than 130 mg/dL as a risk modifier in patients not using statins but, the authors wrote, “based on our results, the threshold for apoB as a risk modifier in statin-treated patients should be closer to 92 mg/dL than to 130 mg/dL.”
In an accompanying editorial, Neil J. Stone, MD, and Donald Lloyd-Jones, MD, both from Northwestern University, Chicago, said that American and European guidelines acknowledge the usefulness of apoB and non-HDL cholesterol in their risk algorithms and as possible targets to indicate efficacy, but don’t give a strong recommendation for apoB to assess residual risk.
“This paper suggests that, in the next iteration, we’ve got to give a stronger thought to measuring apoB for residual risk in those with secondary prevention,” Dr. Stone, vice chair of the 2018 American Heart Association/ACC cholesterol guidelines, said in an interview.
“The whole part of the guidelines was not to focus on any one number but to focus on the clinical risk as a whole,” he said. “You can enlarge your understanding of the patient by looking at their non-HDL, which you have anyway, and in certain circumstances, for example, people with metabolic syndrome, diabetes, obesity, or high triglycerides, those people might very well benefit from an apoB to further understand their risk. This paper simply highlights that and, therefore, was very valuable.”
Dr. Stone and Dr. Lloyd-Jones, however, pointed out that statin use was self-reported and information was lacking on adherence, dose intensity, and the amount of LDL cholesterol lowering from baseline. LDL cholesterol levels were also above current recommendations for optimizing risk reduction. “If statin dosing and LDL [cholesterol] were not optimized already, then there may have been ‘room’ for non-HDL [cholesterol] and apoB to add value in understanding residual risk,” they wrote.
The editorialists suggested that sequential use, rather than regular use, of apoB and non-HDL cholesterol may be best and that incorporating this information may be particularly beneficial for patients with metabolic disorders and elevated triglycerides after statin therapy.
“Maybe this paper is a wake-up call that there are other markers out there that can tell you that you still have higher risk and need to tighten up lifestyle and maybe be more adherent,” Dr. Stone said. “I think this is a wonderful chance to say that preventive cardiology isn’t just ‘set it and forget it’.”
C. Noel Bairey Merz, MD, who coauthored the 2018 cholesterol guidelines, agreed there’s “an overexuberant focus on LDL [cholesterol] for residual risk” and highlighted a recent systematic review of statins, ezetimibe, and PCSK9 cardiovascular outcomes trials that showed very little gain from aggressively driving down LDL below 100 mg/dL, unless the patient is at extremely high risk.
“If I, as a treating cardiologist who spends a lot of time on lipids, had a patient on a high-intensity statin and they didn’t drop [their LDL cholesterol] 50% and I already had them going to cardiac rehab and they were already losing weight, would I measure apoB? Yeah, I might, to motivate them to do more or to take Vascepa,” she said.
“This study is a useful addition to a relatively important problem, which is residual risk, and really supports personalized or precision medicine,” added Bairey Merz, MD, Cedars-Sinai Medical Center, Los Angeles. “But now we have to do the work and do an intervention trial in these people and see whether these markers make a difference.”
The study was supported by Herlev and Gentofte Hospital’s Research Fund and the department of clinical biochemistry, Herlev and Gentofte Hospital, Copenhagen University Hospital. Dr. Nordestgaard has had consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amarin, Amgen, Esperion, Kowa, Novartis, Novo Nordisk, and Silence Therapeutics. All other authors, Dr. Stone, and Dr. Lloyd-Jones reported no conflicts. Dr. Merz reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study shows apolipoprotein B (apoB) and non-HDL cholesterol – but not LDL cholesterol – are associated with increased risk for all-cause mortality and myocardial infarction in patients taking statins.
Moreover, apoB was a more accurate marker of all-cause mortality risk than non-HDL or LDL cholesterol and was more accurate at identifying MI risk than LDL cholesterol.
“Any patient that comes to a doctor for evaluation, if statin treatment is sufficient, the doctor should look not only at LDL cholesterol but HDL cholesterol and apoB, if its available – that is the take-home message,” senior author Børge Grønne Nordestgaard, MD, DMSC, University of Copenhagen, said in an interview.
The findings are very relevant to clinical practice because international guidelines focus on LDL cholesterol and “many doctors are brainwashed that that is the only thing they should look at, just to keep LDL cholesterol down,” he said. “I’ve worked for years with triglyceride lipoproteins, what I call remnant cholesterol, and I think that the risk is very high also when you have high remnant cholesterol.”
Previous work has shown that apoB and non-HDL cholesterol better reflect atherosclerotic cardiovascular disease risk than LDL cholesterol. This is the first study, however, to show that elevated apoB and non-HDL cholesterol are associated with a higher risk for all-cause death in statin-treated patients with low LDL cholesterol, Dr. Nordestgaard noted.
The investigators compared outcomes among 13,015 statin-treated participants in the Copenhagen General Population Study using median baseline values of 92 mg/dL for apoB, 3.1 mmol/L (120 mg/dL) for non-HDL cholesterol, and 2.3 mmol/L (89 mg/dL) for LDL cholesterol. Over a median follow-up of 8 years, there were 2,499 deaths and 537 MIs.
As reported in the Journal of the American College of Cardiology, discordant apoB above the median with LDL cholesterol below was associated with a 21% increased risk for all-cause mortality (hazard ratio, 1.21; 95% confidence interval, 1.07-1.36) and 49% increased risk for MI (HR, 1.49; 95% CI, 1.15-1.92), compared with concordant apoB and LDL cholesterol below the medians.
Similar results were found for discordant non-HDL cholesterol above the median with low LDL cholesterol for all-cause mortality (HR, 1.18; 95% CI, 1.02-1.36) and MI (1.78; 95% CI, 1.35-2.34).
No such associations with mortality or MI were observed when LDL cholesterol was above the median and apoB or non-HDL below.
Additional analyses showed that high apoB with low non-HDL cholesterol was associated with a higher risk for all-cause mortality (HR, 1.21; 95% CI, 1.03-1.41), whereas high non-HDL cholesterol with low apoB was associated with a lower risk (HR, 0.75; 95% CI, 0.62-0.92).
Current guidelines define apoB greater than 130 mg/dL as a risk modifier in patients not using statins but, the authors wrote, “based on our results, the threshold for apoB as a risk modifier in statin-treated patients should be closer to 92 mg/dL than to 130 mg/dL.”
In an accompanying editorial, Neil J. Stone, MD, and Donald Lloyd-Jones, MD, both from Northwestern University, Chicago, said that American and European guidelines acknowledge the usefulness of apoB and non-HDL cholesterol in their risk algorithms and as possible targets to indicate efficacy, but don’t give a strong recommendation for apoB to assess residual risk.
“This paper suggests that, in the next iteration, we’ve got to give a stronger thought to measuring apoB for residual risk in those with secondary prevention,” Dr. Stone, vice chair of the 2018 American Heart Association/ACC cholesterol guidelines, said in an interview.
“The whole part of the guidelines was not to focus on any one number but to focus on the clinical risk as a whole,” he said. “You can enlarge your understanding of the patient by looking at their non-HDL, which you have anyway, and in certain circumstances, for example, people with metabolic syndrome, diabetes, obesity, or high triglycerides, those people might very well benefit from an apoB to further understand their risk. This paper simply highlights that and, therefore, was very valuable.”
Dr. Stone and Dr. Lloyd-Jones, however, pointed out that statin use was self-reported and information was lacking on adherence, dose intensity, and the amount of LDL cholesterol lowering from baseline. LDL cholesterol levels were also above current recommendations for optimizing risk reduction. “If statin dosing and LDL [cholesterol] were not optimized already, then there may have been ‘room’ for non-HDL [cholesterol] and apoB to add value in understanding residual risk,” they wrote.
The editorialists suggested that sequential use, rather than regular use, of apoB and non-HDL cholesterol may be best and that incorporating this information may be particularly beneficial for patients with metabolic disorders and elevated triglycerides after statin therapy.
“Maybe this paper is a wake-up call that there are other markers out there that can tell you that you still have higher risk and need to tighten up lifestyle and maybe be more adherent,” Dr. Stone said. “I think this is a wonderful chance to say that preventive cardiology isn’t just ‘set it and forget it’.”
C. Noel Bairey Merz, MD, who coauthored the 2018 cholesterol guidelines, agreed there’s “an overexuberant focus on LDL [cholesterol] for residual risk” and highlighted a recent systematic review of statins, ezetimibe, and PCSK9 cardiovascular outcomes trials that showed very little gain from aggressively driving down LDL below 100 mg/dL, unless the patient is at extremely high risk.
“If I, as a treating cardiologist who spends a lot of time on lipids, had a patient on a high-intensity statin and they didn’t drop [their LDL cholesterol] 50% and I already had them going to cardiac rehab and they were already losing weight, would I measure apoB? Yeah, I might, to motivate them to do more or to take Vascepa,” she said.
“This study is a useful addition to a relatively important problem, which is residual risk, and really supports personalized or precision medicine,” added Bairey Merz, MD, Cedars-Sinai Medical Center, Los Angeles. “But now we have to do the work and do an intervention trial in these people and see whether these markers make a difference.”
The study was supported by Herlev and Gentofte Hospital’s Research Fund and the department of clinical biochemistry, Herlev and Gentofte Hospital, Copenhagen University Hospital. Dr. Nordestgaard has had consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amarin, Amgen, Esperion, Kowa, Novartis, Novo Nordisk, and Silence Therapeutics. All other authors, Dr. Stone, and Dr. Lloyd-Jones reported no conflicts. Dr. Merz reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study shows apolipoprotein B (apoB) and non-HDL cholesterol – but not LDL cholesterol – are associated with increased risk for all-cause mortality and myocardial infarction in patients taking statins.
Moreover, apoB was a more accurate marker of all-cause mortality risk than non-HDL or LDL cholesterol and was more accurate at identifying MI risk than LDL cholesterol.
“Any patient that comes to a doctor for evaluation, if statin treatment is sufficient, the doctor should look not only at LDL cholesterol but HDL cholesterol and apoB, if its available – that is the take-home message,” senior author Børge Grønne Nordestgaard, MD, DMSC, University of Copenhagen, said in an interview.
The findings are very relevant to clinical practice because international guidelines focus on LDL cholesterol and “many doctors are brainwashed that that is the only thing they should look at, just to keep LDL cholesterol down,” he said. “I’ve worked for years with triglyceride lipoproteins, what I call remnant cholesterol, and I think that the risk is very high also when you have high remnant cholesterol.”
Previous work has shown that apoB and non-HDL cholesterol better reflect atherosclerotic cardiovascular disease risk than LDL cholesterol. This is the first study, however, to show that elevated apoB and non-HDL cholesterol are associated with a higher risk for all-cause death in statin-treated patients with low LDL cholesterol, Dr. Nordestgaard noted.
The investigators compared outcomes among 13,015 statin-treated participants in the Copenhagen General Population Study using median baseline values of 92 mg/dL for apoB, 3.1 mmol/L (120 mg/dL) for non-HDL cholesterol, and 2.3 mmol/L (89 mg/dL) for LDL cholesterol. Over a median follow-up of 8 years, there were 2,499 deaths and 537 MIs.
As reported in the Journal of the American College of Cardiology, discordant apoB above the median with LDL cholesterol below was associated with a 21% increased risk for all-cause mortality (hazard ratio, 1.21; 95% confidence interval, 1.07-1.36) and 49% increased risk for MI (HR, 1.49; 95% CI, 1.15-1.92), compared with concordant apoB and LDL cholesterol below the medians.
Similar results were found for discordant non-HDL cholesterol above the median with low LDL cholesterol for all-cause mortality (HR, 1.18; 95% CI, 1.02-1.36) and MI (1.78; 95% CI, 1.35-2.34).
No such associations with mortality or MI were observed when LDL cholesterol was above the median and apoB or non-HDL below.
Additional analyses showed that high apoB with low non-HDL cholesterol was associated with a higher risk for all-cause mortality (HR, 1.21; 95% CI, 1.03-1.41), whereas high non-HDL cholesterol with low apoB was associated with a lower risk (HR, 0.75; 95% CI, 0.62-0.92).
Current guidelines define apoB greater than 130 mg/dL as a risk modifier in patients not using statins but, the authors wrote, “based on our results, the threshold for apoB as a risk modifier in statin-treated patients should be closer to 92 mg/dL than to 130 mg/dL.”
In an accompanying editorial, Neil J. Stone, MD, and Donald Lloyd-Jones, MD, both from Northwestern University, Chicago, said that American and European guidelines acknowledge the usefulness of apoB and non-HDL cholesterol in their risk algorithms and as possible targets to indicate efficacy, but don’t give a strong recommendation for apoB to assess residual risk.
“This paper suggests that, in the next iteration, we’ve got to give a stronger thought to measuring apoB for residual risk in those with secondary prevention,” Dr. Stone, vice chair of the 2018 American Heart Association/ACC cholesterol guidelines, said in an interview.
“The whole part of the guidelines was not to focus on any one number but to focus on the clinical risk as a whole,” he said. “You can enlarge your understanding of the patient by looking at their non-HDL, which you have anyway, and in certain circumstances, for example, people with metabolic syndrome, diabetes, obesity, or high triglycerides, those people might very well benefit from an apoB to further understand their risk. This paper simply highlights that and, therefore, was very valuable.”
Dr. Stone and Dr. Lloyd-Jones, however, pointed out that statin use was self-reported and information was lacking on adherence, dose intensity, and the amount of LDL cholesterol lowering from baseline. LDL cholesterol levels were also above current recommendations for optimizing risk reduction. “If statin dosing and LDL [cholesterol] were not optimized already, then there may have been ‘room’ for non-HDL [cholesterol] and apoB to add value in understanding residual risk,” they wrote.
The editorialists suggested that sequential use, rather than regular use, of apoB and non-HDL cholesterol may be best and that incorporating this information may be particularly beneficial for patients with metabolic disorders and elevated triglycerides after statin therapy.
“Maybe this paper is a wake-up call that there are other markers out there that can tell you that you still have higher risk and need to tighten up lifestyle and maybe be more adherent,” Dr. Stone said. “I think this is a wonderful chance to say that preventive cardiology isn’t just ‘set it and forget it’.”
C. Noel Bairey Merz, MD, who coauthored the 2018 cholesterol guidelines, agreed there’s “an overexuberant focus on LDL [cholesterol] for residual risk” and highlighted a recent systematic review of statins, ezetimibe, and PCSK9 cardiovascular outcomes trials that showed very little gain from aggressively driving down LDL below 100 mg/dL, unless the patient is at extremely high risk.
“If I, as a treating cardiologist who spends a lot of time on lipids, had a patient on a high-intensity statin and they didn’t drop [their LDL cholesterol] 50% and I already had them going to cardiac rehab and they were already losing weight, would I measure apoB? Yeah, I might, to motivate them to do more or to take Vascepa,” she said.
“This study is a useful addition to a relatively important problem, which is residual risk, and really supports personalized or precision medicine,” added Bairey Merz, MD, Cedars-Sinai Medical Center, Los Angeles. “But now we have to do the work and do an intervention trial in these people and see whether these markers make a difference.”
The study was supported by Herlev and Gentofte Hospital’s Research Fund and the department of clinical biochemistry, Herlev and Gentofte Hospital, Copenhagen University Hospital. Dr. Nordestgaard has had consultancies or talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amarin, Amgen, Esperion, Kowa, Novartis, Novo Nordisk, and Silence Therapeutics. All other authors, Dr. Stone, and Dr. Lloyd-Jones reported no conflicts. Dr. Merz reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Depression screening after ACS does not change outcomes
Background: Depression after ACS is common and is associated with increased mortality. Professional societies have recommended routine depression screening in these patients; however, this has not been consistently implemented because there is a lack of data to support routine screening.
Study design: Multicenter randomized clinical trial.
Setting: Four geographically diverse health systems in the United States.
Synopsis: In the CODIACS-QoL trial, 1,500 patients were randomized to three groups within 12 months of documented ACS: depression screening with notification to primary care and treatment, screening and notification to primary care, and no screening. Only 7.7% of the patients in the screen, notify, and treat group and 6.6% of screen and notify group screened positive for depression. There were no differences for the primary outcome of quality-adjusted life-years or the secondary outcome of depression-free days between groups. Additionally, there was no difference in mortality or patient-reported harms of screening between groups. The study excluded patients who already had a history of depression, psychiatric history, or other severe life-threatening medical conditions, which may have affected the outcomes.
Depression remains a substantial factor in coronary disease and quality of life; however, systematic depression screening appears to have limited population-level benefits.
Bottom line: Systematic depression screening with or without treatment offerings did not alter quality of life, depression-free days, or mortality in patients with ACS.
Citation: Kronish IM et al. Effect of depression screening after acute coronary syndrome on quality of life. JAMA Intern Med. 2020;180(1):45-53.
Dr. Ciarkowski is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Depression after ACS is common and is associated with increased mortality. Professional societies have recommended routine depression screening in these patients; however, this has not been consistently implemented because there is a lack of data to support routine screening.
Study design: Multicenter randomized clinical trial.
Setting: Four geographically diverse health systems in the United States.
Synopsis: In the CODIACS-QoL trial, 1,500 patients were randomized to three groups within 12 months of documented ACS: depression screening with notification to primary care and treatment, screening and notification to primary care, and no screening. Only 7.7% of the patients in the screen, notify, and treat group and 6.6% of screen and notify group screened positive for depression. There were no differences for the primary outcome of quality-adjusted life-years or the secondary outcome of depression-free days between groups. Additionally, there was no difference in mortality or patient-reported harms of screening between groups. The study excluded patients who already had a history of depression, psychiatric history, or other severe life-threatening medical conditions, which may have affected the outcomes.
Depression remains a substantial factor in coronary disease and quality of life; however, systematic depression screening appears to have limited population-level benefits.
Bottom line: Systematic depression screening with or without treatment offerings did not alter quality of life, depression-free days, or mortality in patients with ACS.
Citation: Kronish IM et al. Effect of depression screening after acute coronary syndrome on quality of life. JAMA Intern Med. 2020;180(1):45-53.
Dr. Ciarkowski is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Background: Depression after ACS is common and is associated with increased mortality. Professional societies have recommended routine depression screening in these patients; however, this has not been consistently implemented because there is a lack of data to support routine screening.
Study design: Multicenter randomized clinical trial.
Setting: Four geographically diverse health systems in the United States.
Synopsis: In the CODIACS-QoL trial, 1,500 patients were randomized to three groups within 12 months of documented ACS: depression screening with notification to primary care and treatment, screening and notification to primary care, and no screening. Only 7.7% of the patients in the screen, notify, and treat group and 6.6% of screen and notify group screened positive for depression. There were no differences for the primary outcome of quality-adjusted life-years or the secondary outcome of depression-free days between groups. Additionally, there was no difference in mortality or patient-reported harms of screening between groups. The study excluded patients who already had a history of depression, psychiatric history, or other severe life-threatening medical conditions, which may have affected the outcomes.
Depression remains a substantial factor in coronary disease and quality of life; however, systematic depression screening appears to have limited population-level benefits.
Bottom line: Systematic depression screening with or without treatment offerings did not alter quality of life, depression-free days, or mortality in patients with ACS.
Citation: Kronish IM et al. Effect of depression screening after acute coronary syndrome on quality of life. JAMA Intern Med. 2020;180(1):45-53.
Dr. Ciarkowski is a hospitalist and clinical instructor of medicine at the University of Utah, Salt Lake City.
Direct transfer to angiography improves outcome in large-vessel stroke
in a new study.
Results of the ANGIO-CAT trial were presented at the International Stroke Conference sponsored by the American Heart Association.
The study involved patients suspected of having a large-vessel occlusion, as assessed in the prehospital setting by paramedics using the Rapid Arterial Occlusion Evaluation (RACE) score.
In his presentation, Manuel Requena, PhD, a neurologist and neurointerventionalist fellow at Vall d’Hebron Hospital, Barcelona, explained that, if patients were within 6 hours of symptom onset with a RACE scale score greater than 4, paramedics called ahead to a stroke neurologist, who met the patient directly at the hospital.
If on clinical examination the National Institutes of Health Stroke Scale (NIHSS) score was greater than 10, patients could be enrolled into the study. Upon enrollment, they were randomly assigned either to be taken directly to the angiography suite or to receive standard care.
Bypassing the emergency department
Dr. Requena noted that, at his center, patients who receive standard care are transferred to the CT imaging suite, where they are evaluated with noncontrast CT and CT angiography. CT perfusion is also performed if the treating physician deems it necessary.
If a large-vessel occlusion is confirmed, patients are then transferred to the angiography suite for endovascular treatment. He added that in many centers, patients are evaluated in the ED before undergoing CT scanning.
Patients in the direct angiography group received a “flat-panel” noncontrast CT in the angiography suite to rule out intracranial hemorrhage or a large, established infarct. The large-vessel occlusion would be confirmed by arteriography before the endovascular procedure was performed.
After CT scanning, patients received thrombolysis as recommended in the guidelines.
The current interim analysis includes the 174 patients who have been enrolled so far in the study. The median RACE score for these patients was 7, and the median NIHSS score was 17. Large-vessel occlusion was confirmed in 84% of patients, and 8% had an intracerebral hemorrhage.
Results showed that of the 147 patients who received endovascular therapy, puncture time was shorter for those who were taken directly to angiography (median, 18 min vs. 42 min), as was time to reperfusion (median, 57 min vs. 84 min).
The primary outcome was a shift analysis of the Modified Rankin Scale functional outcome scale at 90 days (odds of 1-point improvement or more). In the direct angiography group, the adjusted odds ratio for an improved functional outcome was 2.2 (95% confidence interval, 1.2-.1).
There were no significant differences in safety endpoints. There was a trend toward more procedural complications in those receiving endovascular therapy in the direct angiography group (8.1% vs. 2.7%; P = .6), but there was also a trend toward lower 90-day mortality in this group (20.2% vs. 32.9%; P = .07)
Dr. Requena reported no significant difference in safety outcomes among those with a hemorrhagic stroke.
“Our study is the first clinical trial that shows the superiority of direct transfer to an angiography suite,” said Dr. Requena. “Our findings were close to what we expected, and we were surprised that they occurred so early in the study. We trust that they will be confirmed in ongoing, multicenter, international trials.”
Stroke patients who were transferred directly to an angiography suite were also less likely to be dependent on assistance with daily activities than were those who received the current standard of care, Dr. Requena said. “More frequent and more rapid treatment can help improve outcomes for our stroke patients.”
A limitation of this study is that the hospital had extensive experience with immediate angiography, so findings may differ at hospitals or care centers with less angiography expertise or experience, Dr. Requena said.
He added that retrospective studies conducted in hospitals in the United States, Germany, and Switzerland show that this kind of protocol can be developed in any high-volume stroke center, although multicenter, international trials are needed.
The cost of speed
Commenting on the ANGIO-CAT study, Michael Hill, MD, a professor at the University of Calgary (Alta.), said the 27-minute improvement in door-to-reperfusion time achieved in the study was meaningful and correlates with the degree of improved outcomes observed. “So, the improvement in speed of treatment resulting in better outcomes makes sense,” he added.
He cautioned that this strategy would only be feasible in certain centers with selected patients and that cost will be a fundamental issue.
“If you identify patients at angiography, you risk having some patients with no target large-vessel occlusion,” Dr. Hill added. “The real question is, how many of these patients without a large-vessel occlusion can the system tolerate before it becomes uneconomical and not fruitful or harmful, given that groin puncture is not totally harmless?”
The moderator of the ISC news conference on the study, Mitchell Elkind, MD, professor of neurology at Columbia University, New York, who is also president of the American Stroke Association, said the study reflects the growing recognition of the importance of speed when treating stroke. “If we can shorten time to treatment using rapid evaluation and imaging protocols, this will help save brain,” he said.
Also commenting on the study, Louisa McCullough, MD, PhD, chief of neurology at Memorial Hermann Hospital–Texas Medical Center, Houston, who is the ISC meeting chair, said she thought the study would be relevant to the United States. “Speed is really of the essence. Whenever we can reduce delays, that will make a big difference to patients.”
Referring to this study on improving hospital systems, as well as a second study that was presented at the meeting that showed benefits from delivery of prehospital thrombolysis via a mobile stroke unit, Dr. McCullough added that “we need to set up models so we can get the best of both these worlds. These studies are really leading the way on how we can change the stroke systems of care.”
The study was funded by Vall d’Hebron Research Institute. Dr. Requena disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in a new study.
Results of the ANGIO-CAT trial were presented at the International Stroke Conference sponsored by the American Heart Association.
The study involved patients suspected of having a large-vessel occlusion, as assessed in the prehospital setting by paramedics using the Rapid Arterial Occlusion Evaluation (RACE) score.
In his presentation, Manuel Requena, PhD, a neurologist and neurointerventionalist fellow at Vall d’Hebron Hospital, Barcelona, explained that, if patients were within 6 hours of symptom onset with a RACE scale score greater than 4, paramedics called ahead to a stroke neurologist, who met the patient directly at the hospital.
If on clinical examination the National Institutes of Health Stroke Scale (NIHSS) score was greater than 10, patients could be enrolled into the study. Upon enrollment, they were randomly assigned either to be taken directly to the angiography suite or to receive standard care.
Bypassing the emergency department
Dr. Requena noted that, at his center, patients who receive standard care are transferred to the CT imaging suite, where they are evaluated with noncontrast CT and CT angiography. CT perfusion is also performed if the treating physician deems it necessary.
If a large-vessel occlusion is confirmed, patients are then transferred to the angiography suite for endovascular treatment. He added that in many centers, patients are evaluated in the ED before undergoing CT scanning.
Patients in the direct angiography group received a “flat-panel” noncontrast CT in the angiography suite to rule out intracranial hemorrhage or a large, established infarct. The large-vessel occlusion would be confirmed by arteriography before the endovascular procedure was performed.
After CT scanning, patients received thrombolysis as recommended in the guidelines.
The current interim analysis includes the 174 patients who have been enrolled so far in the study. The median RACE score for these patients was 7, and the median NIHSS score was 17. Large-vessel occlusion was confirmed in 84% of patients, and 8% had an intracerebral hemorrhage.
Results showed that of the 147 patients who received endovascular therapy, puncture time was shorter for those who were taken directly to angiography (median, 18 min vs. 42 min), as was time to reperfusion (median, 57 min vs. 84 min).
The primary outcome was a shift analysis of the Modified Rankin Scale functional outcome scale at 90 days (odds of 1-point improvement or more). In the direct angiography group, the adjusted odds ratio for an improved functional outcome was 2.2 (95% confidence interval, 1.2-.1).
There were no significant differences in safety endpoints. There was a trend toward more procedural complications in those receiving endovascular therapy in the direct angiography group (8.1% vs. 2.7%; P = .6), but there was also a trend toward lower 90-day mortality in this group (20.2% vs. 32.9%; P = .07)
Dr. Requena reported no significant difference in safety outcomes among those with a hemorrhagic stroke.
“Our study is the first clinical trial that shows the superiority of direct transfer to an angiography suite,” said Dr. Requena. “Our findings were close to what we expected, and we were surprised that they occurred so early in the study. We trust that they will be confirmed in ongoing, multicenter, international trials.”
Stroke patients who were transferred directly to an angiography suite were also less likely to be dependent on assistance with daily activities than were those who received the current standard of care, Dr. Requena said. “More frequent and more rapid treatment can help improve outcomes for our stroke patients.”
A limitation of this study is that the hospital had extensive experience with immediate angiography, so findings may differ at hospitals or care centers with less angiography expertise or experience, Dr. Requena said.
He added that retrospective studies conducted in hospitals in the United States, Germany, and Switzerland show that this kind of protocol can be developed in any high-volume stroke center, although multicenter, international trials are needed.
The cost of speed
Commenting on the ANGIO-CAT study, Michael Hill, MD, a professor at the University of Calgary (Alta.), said the 27-minute improvement in door-to-reperfusion time achieved in the study was meaningful and correlates with the degree of improved outcomes observed. “So, the improvement in speed of treatment resulting in better outcomes makes sense,” he added.
He cautioned that this strategy would only be feasible in certain centers with selected patients and that cost will be a fundamental issue.
“If you identify patients at angiography, you risk having some patients with no target large-vessel occlusion,” Dr. Hill added. “The real question is, how many of these patients without a large-vessel occlusion can the system tolerate before it becomes uneconomical and not fruitful or harmful, given that groin puncture is not totally harmless?”
The moderator of the ISC news conference on the study, Mitchell Elkind, MD, professor of neurology at Columbia University, New York, who is also president of the American Stroke Association, said the study reflects the growing recognition of the importance of speed when treating stroke. “If we can shorten time to treatment using rapid evaluation and imaging protocols, this will help save brain,” he said.
Also commenting on the study, Louisa McCullough, MD, PhD, chief of neurology at Memorial Hermann Hospital–Texas Medical Center, Houston, who is the ISC meeting chair, said she thought the study would be relevant to the United States. “Speed is really of the essence. Whenever we can reduce delays, that will make a big difference to patients.”
Referring to this study on improving hospital systems, as well as a second study that was presented at the meeting that showed benefits from delivery of prehospital thrombolysis via a mobile stroke unit, Dr. McCullough added that “we need to set up models so we can get the best of both these worlds. These studies are really leading the way on how we can change the stroke systems of care.”
The study was funded by Vall d’Hebron Research Institute. Dr. Requena disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in a new study.
Results of the ANGIO-CAT trial were presented at the International Stroke Conference sponsored by the American Heart Association.
The study involved patients suspected of having a large-vessel occlusion, as assessed in the prehospital setting by paramedics using the Rapid Arterial Occlusion Evaluation (RACE) score.
In his presentation, Manuel Requena, PhD, a neurologist and neurointerventionalist fellow at Vall d’Hebron Hospital, Barcelona, explained that, if patients were within 6 hours of symptom onset with a RACE scale score greater than 4, paramedics called ahead to a stroke neurologist, who met the patient directly at the hospital.
If on clinical examination the National Institutes of Health Stroke Scale (NIHSS) score was greater than 10, patients could be enrolled into the study. Upon enrollment, they were randomly assigned either to be taken directly to the angiography suite or to receive standard care.
Bypassing the emergency department
Dr. Requena noted that, at his center, patients who receive standard care are transferred to the CT imaging suite, where they are evaluated with noncontrast CT and CT angiography. CT perfusion is also performed if the treating physician deems it necessary.
If a large-vessel occlusion is confirmed, patients are then transferred to the angiography suite for endovascular treatment. He added that in many centers, patients are evaluated in the ED before undergoing CT scanning.
Patients in the direct angiography group received a “flat-panel” noncontrast CT in the angiography suite to rule out intracranial hemorrhage or a large, established infarct. The large-vessel occlusion would be confirmed by arteriography before the endovascular procedure was performed.
After CT scanning, patients received thrombolysis as recommended in the guidelines.
The current interim analysis includes the 174 patients who have been enrolled so far in the study. The median RACE score for these patients was 7, and the median NIHSS score was 17. Large-vessel occlusion was confirmed in 84% of patients, and 8% had an intracerebral hemorrhage.
Results showed that of the 147 patients who received endovascular therapy, puncture time was shorter for those who were taken directly to angiography (median, 18 min vs. 42 min), as was time to reperfusion (median, 57 min vs. 84 min).
The primary outcome was a shift analysis of the Modified Rankin Scale functional outcome scale at 90 days (odds of 1-point improvement or more). In the direct angiography group, the adjusted odds ratio for an improved functional outcome was 2.2 (95% confidence interval, 1.2-.1).
There were no significant differences in safety endpoints. There was a trend toward more procedural complications in those receiving endovascular therapy in the direct angiography group (8.1% vs. 2.7%; P = .6), but there was also a trend toward lower 90-day mortality in this group (20.2% vs. 32.9%; P = .07)
Dr. Requena reported no significant difference in safety outcomes among those with a hemorrhagic stroke.
“Our study is the first clinical trial that shows the superiority of direct transfer to an angiography suite,” said Dr. Requena. “Our findings were close to what we expected, and we were surprised that they occurred so early in the study. We trust that they will be confirmed in ongoing, multicenter, international trials.”
Stroke patients who were transferred directly to an angiography suite were also less likely to be dependent on assistance with daily activities than were those who received the current standard of care, Dr. Requena said. “More frequent and more rapid treatment can help improve outcomes for our stroke patients.”
A limitation of this study is that the hospital had extensive experience with immediate angiography, so findings may differ at hospitals or care centers with less angiography expertise or experience, Dr. Requena said.
He added that retrospective studies conducted in hospitals in the United States, Germany, and Switzerland show that this kind of protocol can be developed in any high-volume stroke center, although multicenter, international trials are needed.
The cost of speed
Commenting on the ANGIO-CAT study, Michael Hill, MD, a professor at the University of Calgary (Alta.), said the 27-minute improvement in door-to-reperfusion time achieved in the study was meaningful and correlates with the degree of improved outcomes observed. “So, the improvement in speed of treatment resulting in better outcomes makes sense,” he added.
He cautioned that this strategy would only be feasible in certain centers with selected patients and that cost will be a fundamental issue.
“If you identify patients at angiography, you risk having some patients with no target large-vessel occlusion,” Dr. Hill added. “The real question is, how many of these patients without a large-vessel occlusion can the system tolerate before it becomes uneconomical and not fruitful or harmful, given that groin puncture is not totally harmless?”
The moderator of the ISC news conference on the study, Mitchell Elkind, MD, professor of neurology at Columbia University, New York, who is also president of the American Stroke Association, said the study reflects the growing recognition of the importance of speed when treating stroke. “If we can shorten time to treatment using rapid evaluation and imaging protocols, this will help save brain,” he said.
Also commenting on the study, Louisa McCullough, MD, PhD, chief of neurology at Memorial Hermann Hospital–Texas Medical Center, Houston, who is the ISC meeting chair, said she thought the study would be relevant to the United States. “Speed is really of the essence. Whenever we can reduce delays, that will make a big difference to patients.”
Referring to this study on improving hospital systems, as well as a second study that was presented at the meeting that showed benefits from delivery of prehospital thrombolysis via a mobile stroke unit, Dr. McCullough added that “we need to set up models so we can get the best of both these worlds. These studies are really leading the way on how we can change the stroke systems of care.”
The study was funded by Vall d’Hebron Research Institute. Dr. Requena disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ISC 2021
Blood pressure meds tied to increased schizophrenia risk
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ACE inhibitors may be associated with an increased risk for schizophrenia and may affect psychiatric symptoms, new research suggests.
Investigators found individuals who carry a genetic variant associated with lower levels of the ACE gene and protein have increased liability to schizophrenia, suggesting that drugs that lower ACE levels or activity may do the same.
“Our findings warrant further investigation into the role of ACE in schizophrenia and closer monitoring by clinicians of individuals, especially those with schizophrenia, who may be on medication that lower ACE activity, such as ACE inhibitors,” Sonia Shah, PhD, Institute for Biomedical Sciences, University of Queensland, Brisbane, Australia, said in an interview.
The study was published online March 10, 2021, in JAMA Psychiatry.
Antihypertensives and mental illness
Hypertension is common in patients with psychiatric disorders and observational studies have reported associations between antihypertensive medication and these disorders, although the findings have been mixed.
Dr. Shah and colleagues estimated the potential of different antihypertensive drug classes on schizophrenia, bipolar disorder, and major depressive disorder.
In a two-sample Mendelian randomization study, they evaluated ties between a single-nucleotide variant and drug-target gene expression derived from expression quantitative trait loci data in blood (sample 1) and the SNV disease association from published case-control, genomewide association studies (sample 2).
The analyses included 40,675 patients with schizophrenia and 64,643 controls; 20,352 with bipolar disorder and 31,358 controls; and 135,458 with major depressive disorder and 344,901 controls.
The major finding was that a one standard deviation–lower expression of the ACE gene in blood was associated with lower systolic blood pressure of 4.0 mm Hg (95% confidence interval, 2.7-5.3), but also an increased risk of schizophrenia (odds ratio, 1.75; 95% CI, 1.28-2.38).
Could ACE inhibitors worsen symptoms or trigger episodes?
In their article, the researchers noted that, in most patients, onset of schizophrenia occurs in late adolescence or early adult life, ruling out ACE inhibitor treatment as a potential causal factor for most cases.
“However, if lower ACE levels play a causal role for schizophrenia risk, it would be reasonable to hypothesize that further lowering of ACE activity in existing patients could worsen symptoms or trigger a new episode,” they wrote.
Dr. Shah emphasized that evidence from genetic analyses alone is “not sufficient to justify changes in prescription guidelines.”
“Patients should not stop taking these medications if they are effective at controlling their blood pressure and they don’t suffer any adverse effects. But it would be reasonable to encourage greater pharmacovigilance,” she said in an interview.
“One way in which we are hoping to follow up these findings,” said Dr. Shah, “is to access electronic health record data for millions of individuals to investigate if there is evidence of increased rates of psychotic episodes in individuals who use ACE inhibitors, compared to other classes of blood pressure–lowering medication.”
Caution warranted
Reached for comment, Timothy Sullivan, MD, chair of psychiatry and behavioral sciences at Staten Island University Hospital in New York, noted that this is an “extremely complicated” study and urged caution in interpreting the results.
“Since most people develop schizophrenia earlier in life, before they usually develop problems with blood pressure, it’s not so much that these drugs might cause schizophrenia,” Dr. Sullivan said.
“But because of their effects on this particular gene, there’s a possibility that they might worsen symptoms or in somebody with borderline risk might cause them to develop symptoms later in life. This may apply to a relatively small number of people who develop symptoms of schizophrenia in their 40s and beyond,” he added.
That’s where “pharmacovigilance” comes into play, Dr. Sullivan said. “In other words, that they otherwise wouldn’t experience?”
Support for the study was provided by the National Health and Medical Research Council (Australia) and U.S. National Institute for Mental Health. Dr. Shah and Dr. Sullivan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ultraprocessed foods, many marketed as healthy, raise CVD risk
Eating ultraprocessed foods poses a significant risk to cardiovascular and coronary heart health, according to prospective data from about 3,000 people in the Framingham Offspring Cohort, the second generation of participants in the Framingham Heart Study.
Each regular, daily serving of ultraprocessed food was linked with significant elevations of 5%-9% in the relative rates of “hard” cardiovascular disease (CVD) events, hard coronary heart disease (CHD) events, overall CVD events, and CVD death, after adjustments for numerous potential confounders including energy intake, body mass index, waist circumference, and blood pressure, Filippa Juul, PhD, and associates wrote in a report published in the Journal of the American College of Cardiology.
“Consumption of ultraprocessed foods makes up over half of the daily calories in the average American diet and are increasingly consumed worldwide. As poor diet is a major modifiable risk factor for heart disease, it represents a critical target in prevention efforts,” said Dr. Juul, a nutritional epidemiologist at New York University, in a statement released by the American College of Cardiology.
“Our findings add to a growing body of evidence suggesting cardiovascular benefits of limiting ultraprocessed foods. Ultraprocessed foods are ubiquitous and include many foods that are marketed as healthy, such as protein bars, breakfast cereals, and most industrially produced breads,” she added. Other commonplace members of the ultraprocessed food group include carbonated soft drinks, packaged snacks, candies, sausages, margarines, and energy drinks. The concept of ultraprocessed foods as a distinct, wide-ranging, and dangerous food category first appeared in 2010, and then received an update from a United Nations panel in 2019 as what’s now called the NOVA classification system.
Ultraprocessed foods fly under the radar
“Although cardiovascular guidelines emphasize consuming minimally processed foods, such as fruits, vegetables, whole grains, and nuts, they give less attention to the importance of minimizing ultraprocessed food,” wrote Robert J. Ostfeld, MD, and Kathleen E. Allen, MS, in an editorial that accompanied the new report. This reduced attention may be because of a “paucity of studies examining the association cardiovascular outcomes and ultraprocessed foods.”
The new evidence demands new policies, educational efforts, and labeling changes, suggested Dr. Ostfeld, director of preventive cardiology at Montefiore Health System in New York, and Ms. Allen, a dietitian at the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The goal should be to make the unhealthy choice the hard choice and the healthy choice the easy choice.”
The new analysis used data collected from people enrolled the Framingham Offspring Cohort, with their clinical metrics and diet information collected during 1991-1995 serving as their baseline. After excluding participants with prevalent CVD at baseline and those with incomplete follow-up of CVD events, the researchers had a cohort of 3,003 adults with an average follow-up of 18 years. At baseline, the cohort averaged 54 years of age; 55% were women, their average body mass index was 27.3 kg/m2, and about 6% had diabetes. They reported eating, on average, 7.5 servings of ultraprocessed food daily.
During follow-up, the cohort tallied 648 incident CVD events, including 251 hard CVD events (coronary death, MI, or stroke) and 163 hard CHD events (coronary death or MI), and 713 total deaths including 108 CVD deaths. Other CVD events recorded but not considered hard included heart failure, intermittent claudication, and transient ischemic attack.
In a multivariate-adjusted analysis, each average daily portion of ultraprocessed food was linked with an significant 7% relative increase in the incidence of a hard CVD event, compared with participants who ate fewer ultraprocessed food portions, and a 9% relative increase in the rate of hard CHD events, the study’s two prespecified primary outcomes. The researchers also found that each ultraprocessed serving significantly was associated with a 5% relative increased rate of total CVD events, and a 9% relative rise in CVD deaths. The analysis showed no significant association between total mortality and ultraprocessed food intake. (Average follow-up for the mortality analyses was 20 years.)
The authors also reported endpoint associations with intake of specific types of ultraprocessed foods, and found significantly increased associations specifically for portions of bread, ultraprocessed meat, salty snacks, and low-calorie soft drinks.
Convenient, omnipresent, and affordable
The authors acknowledged that the associations they found need examination in ethnically diverse populations, but nonetheless the findings “suggest the need for increased efforts to implement population-wide strategies” to lower consumption of ultraprocessed foods. “Given the convenience, omnipresence, and affordability of ultraprocessed foods, careful nutrition counseling is needed to design individualized, patient-centered, heart-healthy diets,” they concluded.
“Population-wide strategies such as taxation on sugar-sweetened beverages and other ultraprocessed foods and recommendations regarding processing levels in national dietary guidelines are needed to reduce the intake of ultraprocessed foods,” added Dr. Juul in her statement. “Of course, we must also implement policies that increase the availability, accessibility, and affordability of nutritious, minimally processed foods, especially in disadvantaged populations. At the clinical level, there is a need for increased commitment to individualized nutrition counseling for adopting sustainable heart-healthy diets.”
The study had no commercial funding. Dr. Juul and coauthors, Dr. Ostfeld, and Ms. Allen had no disclosures.
Eating ultraprocessed foods poses a significant risk to cardiovascular and coronary heart health, according to prospective data from about 3,000 people in the Framingham Offspring Cohort, the second generation of participants in the Framingham Heart Study.
Each regular, daily serving of ultraprocessed food was linked with significant elevations of 5%-9% in the relative rates of “hard” cardiovascular disease (CVD) events, hard coronary heart disease (CHD) events, overall CVD events, and CVD death, after adjustments for numerous potential confounders including energy intake, body mass index, waist circumference, and blood pressure, Filippa Juul, PhD, and associates wrote in a report published in the Journal of the American College of Cardiology.
“Consumption of ultraprocessed foods makes up over half of the daily calories in the average American diet and are increasingly consumed worldwide. As poor diet is a major modifiable risk factor for heart disease, it represents a critical target in prevention efforts,” said Dr. Juul, a nutritional epidemiologist at New York University, in a statement released by the American College of Cardiology.
“Our findings add to a growing body of evidence suggesting cardiovascular benefits of limiting ultraprocessed foods. Ultraprocessed foods are ubiquitous and include many foods that are marketed as healthy, such as protein bars, breakfast cereals, and most industrially produced breads,” she added. Other commonplace members of the ultraprocessed food group include carbonated soft drinks, packaged snacks, candies, sausages, margarines, and energy drinks. The concept of ultraprocessed foods as a distinct, wide-ranging, and dangerous food category first appeared in 2010, and then received an update from a United Nations panel in 2019 as what’s now called the NOVA classification system.
Ultraprocessed foods fly under the radar
“Although cardiovascular guidelines emphasize consuming minimally processed foods, such as fruits, vegetables, whole grains, and nuts, they give less attention to the importance of minimizing ultraprocessed food,” wrote Robert J. Ostfeld, MD, and Kathleen E. Allen, MS, in an editorial that accompanied the new report. This reduced attention may be because of a “paucity of studies examining the association cardiovascular outcomes and ultraprocessed foods.”
The new evidence demands new policies, educational efforts, and labeling changes, suggested Dr. Ostfeld, director of preventive cardiology at Montefiore Health System in New York, and Ms. Allen, a dietitian at the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The goal should be to make the unhealthy choice the hard choice and the healthy choice the easy choice.”
The new analysis used data collected from people enrolled the Framingham Offspring Cohort, with their clinical metrics and diet information collected during 1991-1995 serving as their baseline. After excluding participants with prevalent CVD at baseline and those with incomplete follow-up of CVD events, the researchers had a cohort of 3,003 adults with an average follow-up of 18 years. At baseline, the cohort averaged 54 years of age; 55% were women, their average body mass index was 27.3 kg/m2, and about 6% had diabetes. They reported eating, on average, 7.5 servings of ultraprocessed food daily.
During follow-up, the cohort tallied 648 incident CVD events, including 251 hard CVD events (coronary death, MI, or stroke) and 163 hard CHD events (coronary death or MI), and 713 total deaths including 108 CVD deaths. Other CVD events recorded but not considered hard included heart failure, intermittent claudication, and transient ischemic attack.
In a multivariate-adjusted analysis, each average daily portion of ultraprocessed food was linked with an significant 7% relative increase in the incidence of a hard CVD event, compared with participants who ate fewer ultraprocessed food portions, and a 9% relative increase in the rate of hard CHD events, the study’s two prespecified primary outcomes. The researchers also found that each ultraprocessed serving significantly was associated with a 5% relative increased rate of total CVD events, and a 9% relative rise in CVD deaths. The analysis showed no significant association between total mortality and ultraprocessed food intake. (Average follow-up for the mortality analyses was 20 years.)
The authors also reported endpoint associations with intake of specific types of ultraprocessed foods, and found significantly increased associations specifically for portions of bread, ultraprocessed meat, salty snacks, and low-calorie soft drinks.
Convenient, omnipresent, and affordable
The authors acknowledged that the associations they found need examination in ethnically diverse populations, but nonetheless the findings “suggest the need for increased efforts to implement population-wide strategies” to lower consumption of ultraprocessed foods. “Given the convenience, omnipresence, and affordability of ultraprocessed foods, careful nutrition counseling is needed to design individualized, patient-centered, heart-healthy diets,” they concluded.
“Population-wide strategies such as taxation on sugar-sweetened beverages and other ultraprocessed foods and recommendations regarding processing levels in national dietary guidelines are needed to reduce the intake of ultraprocessed foods,” added Dr. Juul in her statement. “Of course, we must also implement policies that increase the availability, accessibility, and affordability of nutritious, minimally processed foods, especially in disadvantaged populations. At the clinical level, there is a need for increased commitment to individualized nutrition counseling for adopting sustainable heart-healthy diets.”
The study had no commercial funding. Dr. Juul and coauthors, Dr. Ostfeld, and Ms. Allen had no disclosures.
Eating ultraprocessed foods poses a significant risk to cardiovascular and coronary heart health, according to prospective data from about 3,000 people in the Framingham Offspring Cohort, the second generation of participants in the Framingham Heart Study.
Each regular, daily serving of ultraprocessed food was linked with significant elevations of 5%-9% in the relative rates of “hard” cardiovascular disease (CVD) events, hard coronary heart disease (CHD) events, overall CVD events, and CVD death, after adjustments for numerous potential confounders including energy intake, body mass index, waist circumference, and blood pressure, Filippa Juul, PhD, and associates wrote in a report published in the Journal of the American College of Cardiology.
“Consumption of ultraprocessed foods makes up over half of the daily calories in the average American diet and are increasingly consumed worldwide. As poor diet is a major modifiable risk factor for heart disease, it represents a critical target in prevention efforts,” said Dr. Juul, a nutritional epidemiologist at New York University, in a statement released by the American College of Cardiology.
“Our findings add to a growing body of evidence suggesting cardiovascular benefits of limiting ultraprocessed foods. Ultraprocessed foods are ubiquitous and include many foods that are marketed as healthy, such as protein bars, breakfast cereals, and most industrially produced breads,” she added. Other commonplace members of the ultraprocessed food group include carbonated soft drinks, packaged snacks, candies, sausages, margarines, and energy drinks. The concept of ultraprocessed foods as a distinct, wide-ranging, and dangerous food category first appeared in 2010, and then received an update from a United Nations panel in 2019 as what’s now called the NOVA classification system.
Ultraprocessed foods fly under the radar
“Although cardiovascular guidelines emphasize consuming minimally processed foods, such as fruits, vegetables, whole grains, and nuts, they give less attention to the importance of minimizing ultraprocessed food,” wrote Robert J. Ostfeld, MD, and Kathleen E. Allen, MS, in an editorial that accompanied the new report. This reduced attention may be because of a “paucity of studies examining the association cardiovascular outcomes and ultraprocessed foods.”
The new evidence demands new policies, educational efforts, and labeling changes, suggested Dr. Ostfeld, director of preventive cardiology at Montefiore Health System in New York, and Ms. Allen, a dietitian at the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The goal should be to make the unhealthy choice the hard choice and the healthy choice the easy choice.”
The new analysis used data collected from people enrolled the Framingham Offspring Cohort, with their clinical metrics and diet information collected during 1991-1995 serving as their baseline. After excluding participants with prevalent CVD at baseline and those with incomplete follow-up of CVD events, the researchers had a cohort of 3,003 adults with an average follow-up of 18 years. At baseline, the cohort averaged 54 years of age; 55% were women, their average body mass index was 27.3 kg/m2, and about 6% had diabetes. They reported eating, on average, 7.5 servings of ultraprocessed food daily.
During follow-up, the cohort tallied 648 incident CVD events, including 251 hard CVD events (coronary death, MI, or stroke) and 163 hard CHD events (coronary death or MI), and 713 total deaths including 108 CVD deaths. Other CVD events recorded but not considered hard included heart failure, intermittent claudication, and transient ischemic attack.
In a multivariate-adjusted analysis, each average daily portion of ultraprocessed food was linked with an significant 7% relative increase in the incidence of a hard CVD event, compared with participants who ate fewer ultraprocessed food portions, and a 9% relative increase in the rate of hard CHD events, the study’s two prespecified primary outcomes. The researchers also found that each ultraprocessed serving significantly was associated with a 5% relative increased rate of total CVD events, and a 9% relative rise in CVD deaths. The analysis showed no significant association between total mortality and ultraprocessed food intake. (Average follow-up for the mortality analyses was 20 years.)
The authors also reported endpoint associations with intake of specific types of ultraprocessed foods, and found significantly increased associations specifically for portions of bread, ultraprocessed meat, salty snacks, and low-calorie soft drinks.
Convenient, omnipresent, and affordable
The authors acknowledged that the associations they found need examination in ethnically diverse populations, but nonetheless the findings “suggest the need for increased efforts to implement population-wide strategies” to lower consumption of ultraprocessed foods. “Given the convenience, omnipresence, and affordability of ultraprocessed foods, careful nutrition counseling is needed to design individualized, patient-centered, heart-healthy diets,” they concluded.
“Population-wide strategies such as taxation on sugar-sweetened beverages and other ultraprocessed foods and recommendations regarding processing levels in national dietary guidelines are needed to reduce the intake of ultraprocessed foods,” added Dr. Juul in her statement. “Of course, we must also implement policies that increase the availability, accessibility, and affordability of nutritious, minimally processed foods, especially in disadvantaged populations. At the clinical level, there is a need for increased commitment to individualized nutrition counseling for adopting sustainable heart-healthy diets.”
The study had no commercial funding. Dr. Juul and coauthors, Dr. Ostfeld, and Ms. Allen had no disclosures.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY