User login
The search for a life-changing innovation
It might be the ultimate medical innovation – an artificial heart – and generations of physicians have pursued it, a story told in “Ticker: The Quest to Create an Artificial Heart.”
Author Mimi Swartz feared this history was being forgotten. “The larger-than-life personalities – Dr. Michael DeBakey and Dr. Denton Cooley – were such dominant figures for more than 50 years; I couldn’t stand for that history to be lost,” she said. “Also, so many innovations happened in Houston, including the implantation of the first artificial heart and the development of the Left Ventricular Assist Device – I couldn’t stand for that information to be lost too.”
Writing this book taught her a lot about innovation in medicine, the trade-offs involved in medical progress, even who benefits most.
“One of the most important things to think about is how many of these high-tech devices we need, and who will get them – who will be able to afford them,” she said.
“Medical innovation over the last 50 years is a global, billion dollar business, fraught with pitfalls: legal, governmental, ethical, financial, and, finally, personal,” Ms. Swartz said. “A great invention that could save millions of lives can end up on the junk heap because a hedge fund lost interest, while another great invention moves forward, but was stolen from the lab of another researcher. The persistence required to bring a medical device to market is daunting. One inventor told me, ‘If I’d known what was going to happen, I never would have even started.’ ”
Reference
Swartz M. Ticker: The Quest to Create an Artificial Heart. New York: Penguin Random House, 2018.
It might be the ultimate medical innovation – an artificial heart – and generations of physicians have pursued it, a story told in “Ticker: The Quest to Create an Artificial Heart.”
Author Mimi Swartz feared this history was being forgotten. “The larger-than-life personalities – Dr. Michael DeBakey and Dr. Denton Cooley – were such dominant figures for more than 50 years; I couldn’t stand for that history to be lost,” she said. “Also, so many innovations happened in Houston, including the implantation of the first artificial heart and the development of the Left Ventricular Assist Device – I couldn’t stand for that information to be lost too.”
Writing this book taught her a lot about innovation in medicine, the trade-offs involved in medical progress, even who benefits most.
“One of the most important things to think about is how many of these high-tech devices we need, and who will get them – who will be able to afford them,” she said.
“Medical innovation over the last 50 years is a global, billion dollar business, fraught with pitfalls: legal, governmental, ethical, financial, and, finally, personal,” Ms. Swartz said. “A great invention that could save millions of lives can end up on the junk heap because a hedge fund lost interest, while another great invention moves forward, but was stolen from the lab of another researcher. The persistence required to bring a medical device to market is daunting. One inventor told me, ‘If I’d known what was going to happen, I never would have even started.’ ”
Reference
Swartz M. Ticker: The Quest to Create an Artificial Heart. New York: Penguin Random House, 2018.
It might be the ultimate medical innovation – an artificial heart – and generations of physicians have pursued it, a story told in “Ticker: The Quest to Create an Artificial Heart.”
Author Mimi Swartz feared this history was being forgotten. “The larger-than-life personalities – Dr. Michael DeBakey and Dr. Denton Cooley – were such dominant figures for more than 50 years; I couldn’t stand for that history to be lost,” she said. “Also, so many innovations happened in Houston, including the implantation of the first artificial heart and the development of the Left Ventricular Assist Device – I couldn’t stand for that information to be lost too.”
Writing this book taught her a lot about innovation in medicine, the trade-offs involved in medical progress, even who benefits most.
“One of the most important things to think about is how many of these high-tech devices we need, and who will get them – who will be able to afford them,” she said.
“Medical innovation over the last 50 years is a global, billion dollar business, fraught with pitfalls: legal, governmental, ethical, financial, and, finally, personal,” Ms. Swartz said. “A great invention that could save millions of lives can end up on the junk heap because a hedge fund lost interest, while another great invention moves forward, but was stolen from the lab of another researcher. The persistence required to bring a medical device to market is daunting. One inventor told me, ‘If I’d known what was going to happen, I never would have even started.’ ”
Reference
Swartz M. Ticker: The Quest to Create an Artificial Heart. New York: Penguin Random House, 2018.
Weight-loss drug options expand, but beware cardiac risk
LOS ANGELES – Newer medications are much more powerful, but they come with cautions – insurer coverage can be a hurdle, and there are significant gaps in knowledge about their risks for patients with heart disease, Ken Fujioka, MD, told colleagues at the annual scientific and clinical congress of the American Association of Clinical Endocrinologists.
Dr. Fujioka, of Scripps Clinic in San Diego, shared some tips with his peers about using medications to reduce weight.
Diabetes drugs help, but may need a boost
Metformin can reduce weight by as much as 3%, Dr. Fujioka said. And there may be another benefit related to long-term weight loss maintenance, he said, citing a 15-year study of overweight or obese patients at high risk for diabetes who either received metformin, underwent an intensive lifestyle intervention, or took a placebo. Of the participants with weight loss of at least 5% after the first year, those originally assigned to receive metformin had the greatest weight loss during years 6-15. Older age, the amount of weight initially lost, and continued used of metformin were predictors of long-term weight loss maintenance, according to the researchers (Ann Intern Med. 2019 Apr 23. doi: 10.7326/M18-1605).
There are other options among diabetes drugs. Sodium-glucose cotransporter 2 (SGLT2) inhibitors – a class of drugs that includes canagliflozin (Invokana), dapagliflozin (Farxiga), and empagliflozin (Jardiance) – have a striking effect on weight loss, Dr. Fujioka said. They can cause 300 calories to be flushed out in the urine each day. But that typically doesn’t translate into weight loss of more than 20 pounds, he said, because the body doesn’t fully adjust to fewer calories.
“The patients begin to eat more,” he said. “They have to take in more calories to make up for [the loss]. They’re not consciously trying to do this. It’s a metabolic adaptation, so 2%-3% [weight loss] is about all you’ll get. You won’t get 10% or 20%.”
To drive up weight loss, Dr. Fujioka recommended adding the glucagonlike peptide–1 [GLP1] receptor diabetes drug exenatide (Byetta; Bydureon) or the appetite suppressant phentermine (Adipex-p; Lomaira) to an SGLT2 inhibitor. Recent studies have shown that the drug combinations have a greater impact on weight loss than when taken separately (Lancet Diabetes Endocrinol. 2016 Dec;4[12]:1004-16; Diabetes Care. 2017 May;40[5]:632-9).
In regard to phentermine, which acts similarly to amphetamine, Dr. Fujioka advised colleagues to be aware that “15 mg or less is really safe, but you drive pulse and heart rate beyond that.”
Consider insurance coverage and other factors
Often, insurers will pay for GLP1-receptor and SGLT2-inhibitor medications in patients with diabetes, even if their hemoglobin A1c is in the healthy range, Dr. Fujioka said, but they’ll balk at paying for specific weight-loss medications, although that can vary by the region of the country. He added that cash discount cards are available for several weight-loss drugs.
Newer weight-loss drugs ...
Dr. Fujioka highlighted a quartet of weight-loss drugs that have been approved in recent years.
- Lorcaserin (Belviq), a selective serotonin 2C receptor agonist, has shown unique benefits in patients with diabetes. A large, multinational, randomized controlled trial found that the drug reduced the risk for incident diabetes, induced remission of hyperglycemia, and reduced the risk of microvascular complications in obese and overweight patients (Lancet. 2018 Nov 24;392[10161]:2269-79).
- Phentermine/topiramate (Qsymia), a combination of an antiseizure medication (topiramate) and an appetite suppressant (phentermine). A 2014 study found that the drug, together with lifestyle modification, effectively promoted weight loss and improved glycemic control in obese or overweight patients with type 2 diabetes (Diabetes Care. 2014 Dec;37[12]:3309-16).
- Naltrexone/bupropion (Contrave), a combination of an addiction drug (naltrexone) and an antidepressant (bupropion). Findings from a 2013 study reported that the drug “in overweight/obese patients with type 2 diabetes induced weight loss... was associated with improvements in glycemic control and select cardiovascular risk factors and was generally well tolerated with a safety profile similar to that in patients without diabetes.” (Diabetes Care. 2013 Dec;36[12]:4022-9).
- Liraglutide, an injectable GLP1 agonist that has been approved for diabetes (Victoza) and weight loss (Saxenda). Dr. Fujioka was coauthor for a study in which the findings suggested that the drug could prevent prediabetes from turning into diabetes. (Lancet. 2017 Apr 8;389[10077]:1399-409).
... but watch out for safety in patients with heart disease
Two of the newer weight-loss drugs are OK to prescribe for diabetic patients with heart disease, Dr. Fujioka said, but two are not, because no cardiac safety trials have been completed for them.
Liraglutide (at a dose of 3.0 mg) is considered safe based on previous data (Diabetes Obes Metab. 2018 Mar;20[3]:734-9), Dr. Fujioka said. Likewise, findings from a trial with lorcaserin in which 12,000 overweight or obese patients with atherosclerotic cardiovascular disease or multiple cardiovascular risk factors received either lorcaserin (10 mg twice daily) or placebo, suggested that lorcaserin helped sustain weight loss without a higher rate of major cardiovascular events compared with placebo (N Engl J Med. 2018 Sep 20;379[12]:1107-17).However, no such cardiac safety trials have been completed for naltrexone/bupropion or phentermine/topiramate, said Dr. Fujioka. As a result, he said he could not recommend either of them for patients with high-risk cardiovascular disease.
Dr. Fujioka disclosed relationships of various types with Novo Nordisk, Eisai, Gelesis, KVK Tech, Amgen, Sunovion, Boehringer Ingelheim, and Janssen Global Services.
LOS ANGELES – Newer medications are much more powerful, but they come with cautions – insurer coverage can be a hurdle, and there are significant gaps in knowledge about their risks for patients with heart disease, Ken Fujioka, MD, told colleagues at the annual scientific and clinical congress of the American Association of Clinical Endocrinologists.
Dr. Fujioka, of Scripps Clinic in San Diego, shared some tips with his peers about using medications to reduce weight.
Diabetes drugs help, but may need a boost
Metformin can reduce weight by as much as 3%, Dr. Fujioka said. And there may be another benefit related to long-term weight loss maintenance, he said, citing a 15-year study of overweight or obese patients at high risk for diabetes who either received metformin, underwent an intensive lifestyle intervention, or took a placebo. Of the participants with weight loss of at least 5% after the first year, those originally assigned to receive metformin had the greatest weight loss during years 6-15. Older age, the amount of weight initially lost, and continued used of metformin were predictors of long-term weight loss maintenance, according to the researchers (Ann Intern Med. 2019 Apr 23. doi: 10.7326/M18-1605).
There are other options among diabetes drugs. Sodium-glucose cotransporter 2 (SGLT2) inhibitors – a class of drugs that includes canagliflozin (Invokana), dapagliflozin (Farxiga), and empagliflozin (Jardiance) – have a striking effect on weight loss, Dr. Fujioka said. They can cause 300 calories to be flushed out in the urine each day. But that typically doesn’t translate into weight loss of more than 20 pounds, he said, because the body doesn’t fully adjust to fewer calories.
“The patients begin to eat more,” he said. “They have to take in more calories to make up for [the loss]. They’re not consciously trying to do this. It’s a metabolic adaptation, so 2%-3% [weight loss] is about all you’ll get. You won’t get 10% or 20%.”
To drive up weight loss, Dr. Fujioka recommended adding the glucagonlike peptide–1 [GLP1] receptor diabetes drug exenatide (Byetta; Bydureon) or the appetite suppressant phentermine (Adipex-p; Lomaira) to an SGLT2 inhibitor. Recent studies have shown that the drug combinations have a greater impact on weight loss than when taken separately (Lancet Diabetes Endocrinol. 2016 Dec;4[12]:1004-16; Diabetes Care. 2017 May;40[5]:632-9).
In regard to phentermine, which acts similarly to amphetamine, Dr. Fujioka advised colleagues to be aware that “15 mg or less is really safe, but you drive pulse and heart rate beyond that.”
Consider insurance coverage and other factors
Often, insurers will pay for GLP1-receptor and SGLT2-inhibitor medications in patients with diabetes, even if their hemoglobin A1c is in the healthy range, Dr. Fujioka said, but they’ll balk at paying for specific weight-loss medications, although that can vary by the region of the country. He added that cash discount cards are available for several weight-loss drugs.
Newer weight-loss drugs ...
Dr. Fujioka highlighted a quartet of weight-loss drugs that have been approved in recent years.
- Lorcaserin (Belviq), a selective serotonin 2C receptor agonist, has shown unique benefits in patients with diabetes. A large, multinational, randomized controlled trial found that the drug reduced the risk for incident diabetes, induced remission of hyperglycemia, and reduced the risk of microvascular complications in obese and overweight patients (Lancet. 2018 Nov 24;392[10161]:2269-79).
- Phentermine/topiramate (Qsymia), a combination of an antiseizure medication (topiramate) and an appetite suppressant (phentermine). A 2014 study found that the drug, together with lifestyle modification, effectively promoted weight loss and improved glycemic control in obese or overweight patients with type 2 diabetes (Diabetes Care. 2014 Dec;37[12]:3309-16).
- Naltrexone/bupropion (Contrave), a combination of an addiction drug (naltrexone) and an antidepressant (bupropion). Findings from a 2013 study reported that the drug “in overweight/obese patients with type 2 diabetes induced weight loss... was associated with improvements in glycemic control and select cardiovascular risk factors and was generally well tolerated with a safety profile similar to that in patients without diabetes.” (Diabetes Care. 2013 Dec;36[12]:4022-9).
- Liraglutide, an injectable GLP1 agonist that has been approved for diabetes (Victoza) and weight loss (Saxenda). Dr. Fujioka was coauthor for a study in which the findings suggested that the drug could prevent prediabetes from turning into diabetes. (Lancet. 2017 Apr 8;389[10077]:1399-409).
... but watch out for safety in patients with heart disease
Two of the newer weight-loss drugs are OK to prescribe for diabetic patients with heart disease, Dr. Fujioka said, but two are not, because no cardiac safety trials have been completed for them.
Liraglutide (at a dose of 3.0 mg) is considered safe based on previous data (Diabetes Obes Metab. 2018 Mar;20[3]:734-9), Dr. Fujioka said. Likewise, findings from a trial with lorcaserin in which 12,000 overweight or obese patients with atherosclerotic cardiovascular disease or multiple cardiovascular risk factors received either lorcaserin (10 mg twice daily) or placebo, suggested that lorcaserin helped sustain weight loss without a higher rate of major cardiovascular events compared with placebo (N Engl J Med. 2018 Sep 20;379[12]:1107-17).However, no such cardiac safety trials have been completed for naltrexone/bupropion or phentermine/topiramate, said Dr. Fujioka. As a result, he said he could not recommend either of them for patients with high-risk cardiovascular disease.
Dr. Fujioka disclosed relationships of various types with Novo Nordisk, Eisai, Gelesis, KVK Tech, Amgen, Sunovion, Boehringer Ingelheim, and Janssen Global Services.
LOS ANGELES – Newer medications are much more powerful, but they come with cautions – insurer coverage can be a hurdle, and there are significant gaps in knowledge about their risks for patients with heart disease, Ken Fujioka, MD, told colleagues at the annual scientific and clinical congress of the American Association of Clinical Endocrinologists.
Dr. Fujioka, of Scripps Clinic in San Diego, shared some tips with his peers about using medications to reduce weight.
Diabetes drugs help, but may need a boost
Metformin can reduce weight by as much as 3%, Dr. Fujioka said. And there may be another benefit related to long-term weight loss maintenance, he said, citing a 15-year study of overweight or obese patients at high risk for diabetes who either received metformin, underwent an intensive lifestyle intervention, or took a placebo. Of the participants with weight loss of at least 5% after the first year, those originally assigned to receive metformin had the greatest weight loss during years 6-15. Older age, the amount of weight initially lost, and continued used of metformin were predictors of long-term weight loss maintenance, according to the researchers (Ann Intern Med. 2019 Apr 23. doi: 10.7326/M18-1605).
There are other options among diabetes drugs. Sodium-glucose cotransporter 2 (SGLT2) inhibitors – a class of drugs that includes canagliflozin (Invokana), dapagliflozin (Farxiga), and empagliflozin (Jardiance) – have a striking effect on weight loss, Dr. Fujioka said. They can cause 300 calories to be flushed out in the urine each day. But that typically doesn’t translate into weight loss of more than 20 pounds, he said, because the body doesn’t fully adjust to fewer calories.
“The patients begin to eat more,” he said. “They have to take in more calories to make up for [the loss]. They’re not consciously trying to do this. It’s a metabolic adaptation, so 2%-3% [weight loss] is about all you’ll get. You won’t get 10% or 20%.”
To drive up weight loss, Dr. Fujioka recommended adding the glucagonlike peptide–1 [GLP1] receptor diabetes drug exenatide (Byetta; Bydureon) or the appetite suppressant phentermine (Adipex-p; Lomaira) to an SGLT2 inhibitor. Recent studies have shown that the drug combinations have a greater impact on weight loss than when taken separately (Lancet Diabetes Endocrinol. 2016 Dec;4[12]:1004-16; Diabetes Care. 2017 May;40[5]:632-9).
In regard to phentermine, which acts similarly to amphetamine, Dr. Fujioka advised colleagues to be aware that “15 mg or less is really safe, but you drive pulse and heart rate beyond that.”
Consider insurance coverage and other factors
Often, insurers will pay for GLP1-receptor and SGLT2-inhibitor medications in patients with diabetes, even if their hemoglobin A1c is in the healthy range, Dr. Fujioka said, but they’ll balk at paying for specific weight-loss medications, although that can vary by the region of the country. He added that cash discount cards are available for several weight-loss drugs.
Newer weight-loss drugs ...
Dr. Fujioka highlighted a quartet of weight-loss drugs that have been approved in recent years.
- Lorcaserin (Belviq), a selective serotonin 2C receptor agonist, has shown unique benefits in patients with diabetes. A large, multinational, randomized controlled trial found that the drug reduced the risk for incident diabetes, induced remission of hyperglycemia, and reduced the risk of microvascular complications in obese and overweight patients (Lancet. 2018 Nov 24;392[10161]:2269-79).
- Phentermine/topiramate (Qsymia), a combination of an antiseizure medication (topiramate) and an appetite suppressant (phentermine). A 2014 study found that the drug, together with lifestyle modification, effectively promoted weight loss and improved glycemic control in obese or overweight patients with type 2 diabetes (Diabetes Care. 2014 Dec;37[12]:3309-16).
- Naltrexone/bupropion (Contrave), a combination of an addiction drug (naltrexone) and an antidepressant (bupropion). Findings from a 2013 study reported that the drug “in overweight/obese patients with type 2 diabetes induced weight loss... was associated with improvements in glycemic control and select cardiovascular risk factors and was generally well tolerated with a safety profile similar to that in patients without diabetes.” (Diabetes Care. 2013 Dec;36[12]:4022-9).
- Liraglutide, an injectable GLP1 agonist that has been approved for diabetes (Victoza) and weight loss (Saxenda). Dr. Fujioka was coauthor for a study in which the findings suggested that the drug could prevent prediabetes from turning into diabetes. (Lancet. 2017 Apr 8;389[10077]:1399-409).
... but watch out for safety in patients with heart disease
Two of the newer weight-loss drugs are OK to prescribe for diabetic patients with heart disease, Dr. Fujioka said, but two are not, because no cardiac safety trials have been completed for them.
Liraglutide (at a dose of 3.0 mg) is considered safe based on previous data (Diabetes Obes Metab. 2018 Mar;20[3]:734-9), Dr. Fujioka said. Likewise, findings from a trial with lorcaserin in which 12,000 overweight or obese patients with atherosclerotic cardiovascular disease or multiple cardiovascular risk factors received either lorcaserin (10 mg twice daily) or placebo, suggested that lorcaserin helped sustain weight loss without a higher rate of major cardiovascular events compared with placebo (N Engl J Med. 2018 Sep 20;379[12]:1107-17).However, no such cardiac safety trials have been completed for naltrexone/bupropion or phentermine/topiramate, said Dr. Fujioka. As a result, he said he could not recommend either of them for patients with high-risk cardiovascular disease.
Dr. Fujioka disclosed relationships of various types with Novo Nordisk, Eisai, Gelesis, KVK Tech, Amgen, Sunovion, Boehringer Ingelheim, and Janssen Global Services.
EXPERT ANALYSIS FROM AACE 2019
Single-center study outlines stroke risk, DOAC type in nonvalvular AFib patients
PHILADELPHIA – A disproportionate number of breakthrough strokes were observed among patients receiving rivaroxaban for nonvalvular atrial fibrillation in a stroke unit, according to a small, single-center, retrospective study presented at the annual meeting of the American Academy of Neurology.
The researchers reviewed all patients presenting to a tertiary care stroke unit in Australia from January 2015 to June 2018.
A total of 56 patients (median age was 74 years; 61% were male) had received direct oral anticoagulant (DOAC) therapy and then had an ischemic stroke. Of those patients, 37 (66%) had strokes while receiving the treatment; 14 patients (25%) had a stroke after recently stopping a DOAC, often prior to a medical procedure; and 5 patients (9%) were not adherent to their DOAC regimen.
Of the 37 patients who had strokes during DOAC treatment, 48% were on rivaroxaban, 9% were on dabigatran, and 9% were on apixaban, Fiona Chan, MD, of The Princess Alexandra Hospital, Brisbane, Australia, and coinvestigators reported in a poster presentation.
While these findings need to be replicated in a larger study, they do “raise concern for inadequate stroke prevention within this cohort,” they said.
Moreover, the findings illustrate the importance of bridging anticoagulation prior to procedures, when appropriate, to minimize stroke risk, they added, as 25% of the strokes had occurred in patients who recently stopped the DOACs due to procedures.
To determine which DOAC was most often associated with breakthrough ischemic strokes in patients with nonvalvular atrial fibrillation, the investigators compared the proportion of DOACs prescribed in Australia to the proportion of observed strokes in their cohort.
Despite accounting for about 51% of Australian DOAC prescriptions, rivaroxaban represented nearly 73% of breakthrough strokes among the patients who had strokes while receiving the treatment (P = .001), the investigators reported.
Conversely, apixaban accounted for about 35% of prescriptions but 14% of the breakthrough strokes (P = .0007), while dabigatran accounted for 14% of prescriptions and 14% of the strokes (P = 0.99), the investigators said in their poster.
One limitation of this retrospective study is that the patient cohort came from a single specialized center, and may not reflect the true incidence of nonvalvular atrial fibrillation across Australia, the researchers noted.
Dr. Chan and coinvestigators reported that they had no relevant financial disclosures.
SOURCE: Chan F et al. AAN 2019. P1.3-001.
PHILADELPHIA – A disproportionate number of breakthrough strokes were observed among patients receiving rivaroxaban for nonvalvular atrial fibrillation in a stroke unit, according to a small, single-center, retrospective study presented at the annual meeting of the American Academy of Neurology.
The researchers reviewed all patients presenting to a tertiary care stroke unit in Australia from January 2015 to June 2018.
A total of 56 patients (median age was 74 years; 61% were male) had received direct oral anticoagulant (DOAC) therapy and then had an ischemic stroke. Of those patients, 37 (66%) had strokes while receiving the treatment; 14 patients (25%) had a stroke after recently stopping a DOAC, often prior to a medical procedure; and 5 patients (9%) were not adherent to their DOAC regimen.
Of the 37 patients who had strokes during DOAC treatment, 48% were on rivaroxaban, 9% were on dabigatran, and 9% were on apixaban, Fiona Chan, MD, of The Princess Alexandra Hospital, Brisbane, Australia, and coinvestigators reported in a poster presentation.
While these findings need to be replicated in a larger study, they do “raise concern for inadequate stroke prevention within this cohort,” they said.
Moreover, the findings illustrate the importance of bridging anticoagulation prior to procedures, when appropriate, to minimize stroke risk, they added, as 25% of the strokes had occurred in patients who recently stopped the DOACs due to procedures.
To determine which DOAC was most often associated with breakthrough ischemic strokes in patients with nonvalvular atrial fibrillation, the investigators compared the proportion of DOACs prescribed in Australia to the proportion of observed strokes in their cohort.
Despite accounting for about 51% of Australian DOAC prescriptions, rivaroxaban represented nearly 73% of breakthrough strokes among the patients who had strokes while receiving the treatment (P = .001), the investigators reported.
Conversely, apixaban accounted for about 35% of prescriptions but 14% of the breakthrough strokes (P = .0007), while dabigatran accounted for 14% of prescriptions and 14% of the strokes (P = 0.99), the investigators said in their poster.
One limitation of this retrospective study is that the patient cohort came from a single specialized center, and may not reflect the true incidence of nonvalvular atrial fibrillation across Australia, the researchers noted.
Dr. Chan and coinvestigators reported that they had no relevant financial disclosures.
SOURCE: Chan F et al. AAN 2019. P1.3-001.
PHILADELPHIA – A disproportionate number of breakthrough strokes were observed among patients receiving rivaroxaban for nonvalvular atrial fibrillation in a stroke unit, according to a small, single-center, retrospective study presented at the annual meeting of the American Academy of Neurology.
The researchers reviewed all patients presenting to a tertiary care stroke unit in Australia from January 2015 to June 2018.
A total of 56 patients (median age was 74 years; 61% were male) had received direct oral anticoagulant (DOAC) therapy and then had an ischemic stroke. Of those patients, 37 (66%) had strokes while receiving the treatment; 14 patients (25%) had a stroke after recently stopping a DOAC, often prior to a medical procedure; and 5 patients (9%) were not adherent to their DOAC regimen.
Of the 37 patients who had strokes during DOAC treatment, 48% were on rivaroxaban, 9% were on dabigatran, and 9% were on apixaban, Fiona Chan, MD, of The Princess Alexandra Hospital, Brisbane, Australia, and coinvestigators reported in a poster presentation.
While these findings need to be replicated in a larger study, they do “raise concern for inadequate stroke prevention within this cohort,” they said.
Moreover, the findings illustrate the importance of bridging anticoagulation prior to procedures, when appropriate, to minimize stroke risk, they added, as 25% of the strokes had occurred in patients who recently stopped the DOACs due to procedures.
To determine which DOAC was most often associated with breakthrough ischemic strokes in patients with nonvalvular atrial fibrillation, the investigators compared the proportion of DOACs prescribed in Australia to the proportion of observed strokes in their cohort.
Despite accounting for about 51% of Australian DOAC prescriptions, rivaroxaban represented nearly 73% of breakthrough strokes among the patients who had strokes while receiving the treatment (P = .001), the investigators reported.
Conversely, apixaban accounted for about 35% of prescriptions but 14% of the breakthrough strokes (P = .0007), while dabigatran accounted for 14% of prescriptions and 14% of the strokes (P = 0.99), the investigators said in their poster.
One limitation of this retrospective study is that the patient cohort came from a single specialized center, and may not reflect the true incidence of nonvalvular atrial fibrillation across Australia, the researchers noted.
Dr. Chan and coinvestigators reported that they had no relevant financial disclosures.
SOURCE: Chan F et al. AAN 2019. P1.3-001.
REPORTING FROM AAN 2019
Key clinical point: Rivaroxaban was associated with a disproportionate number of breakthrough strokes among patients with nonvalvular atrial fibrillation treated with direct oral anticoagulants at one stroke unit in Australia.
Major finding: Despite accounting for about 51% of Australian DOAC prescriptions, rivaroxaban represented nearly 73% of breakthrough strokes among the patients who had strokes while receiving treatment (P = .001).
Study details: Retrospective study of 56 patients with nonvalvular atrial fibrillation reporting to a tertiary care stroke unit in Australia.
Disclosures: The authors reported no financial disclosures.
Source: Chan F et al. AAN 019. Poster P1.3-001.
What’s the Best Treatment Setting for Stable Pulmonary Embolism?
A 63-year-old woman with a history of hypertension presents to the emergency department (ED) with acute-onset shortness of breath and pleuritic chest pain after traveling across the country for a work conference. She has no history of cancer, liver disease, or renal disease. Her blood pressure is 140/80 mm Hg, and her heart rate, 90 beats/min. You diagnose an acute PE in this patient and start anticoagulation. Should you admit her to the hospital to decrease morbidity and mortality?
According to the CDC, venous thromboembolism (VTE) affects about 900,000 people each year, and about 60,000 to 100,000 of these patients die annually.2 Pulmonary embolism is the third leading cause of death from cardiovascular disease, following heart attack and stroke.3 Prompt diagnosis and treatment with systemic anticoagulation improves patient outcomes and decreases the risk for long-term complications.
The 2016 American College of Chest Physicians (CHEST) guideline on antithrombotic therapy for VTE recommends home treatment or early discharge over standard discharge (after the first 5 days of treatment) for patients who meet the following clinical criteria: “clinically stable with good cardiopulmonary reserve; no contraindications such as recent bleeding, severe renal or liver disease, or severe thrombocytopenia (ie, < 70,000/mm3); expected to be compliant with treatment; and feels well enough to be treated at home.”3
The guideline states that various clinical decision tools, such as the Pulmonary Embolism Severity Index (PESI), can aid in identifying low-risk patients to be considered for treatment at home. The PESI uses age, gender, vital signs, mental status, and a history of cancer, lung, and cardiac disease to stratify patients by risk.4
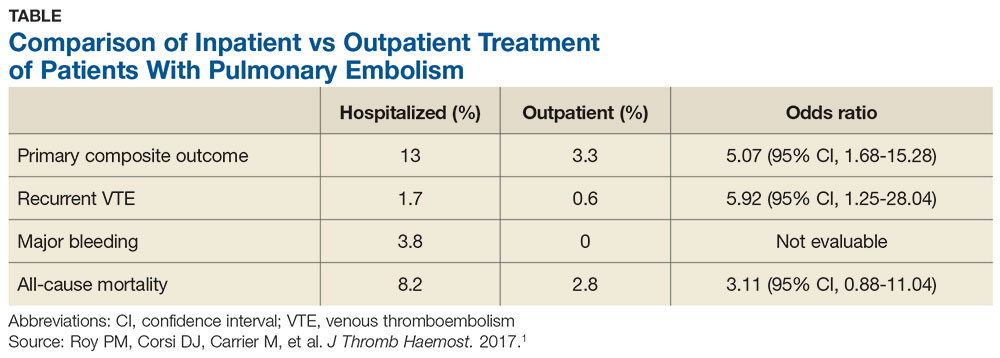
A systematic review of 1 randomized controlled trial (RCT) and 7 observational studies found that in low-risk patients, outpatient treatment was as safe as inpatient treatment.5 This more recent study determines the net clinical benefit of hospitalized versus outpatient management in a wider range of patients with acute PE, regardless of initial risk.1
STUDY SUMMARY
Hospitalization confers no benefit to stable PE patients
This retrospective, propensity-matched cohort study compared rates of adverse events in 1127 patients with acute PE managed in the hospital versus outpatient setting.1 Patients were classified as outpatients if they were discharged from the ED or discharged from the hospital within 48 hours of admission. Patients were included if a symptomatic acute PE was diagnosed via CT or high-probability ventilation-perfusion scan and excluded if they were younger than 19, were diagnosed with a PE during hospitalization, had chronic PE, or were hemodynamically unstable, among other factors. The investigators calculated PESI scores for all patients.
Propensity scores matched patients on 28 characteristics and known risk factors for adverse events to ensure the groups were similar. The primary outcome was rate of adverse events, including recurrent VTE, major bleeding, or death at 14 days. The secondary outcome included rates of the above during the 3-month follow-up period.
Continue to: Of the 1127 eligible patients...
Of the 1127 eligible patients, 1081 were included in the matched cohort, with 576 (53%) treated as hospitalized patients and 505 (47%) treated as outpatients. The mean age of the matched cohorts wa
The 14-day rate of adverse events was higher in hospitalized patients than in outpatients (13% vs 3.3%; odds ratio [OR], 5.07), with each of the adverse events that made up the primary outcome occurring more frequently in the hospitalized group (see Table). The rate of adverse events at 3 months was also greater for hospitalized patients compared with outpatients (21.7% vs 6.9%; OR, 4.9). The results remained similar for high-risk patients (Class III-V) based on their PESI score.
WHAT’S NEW
Higher rate of adverse events in inpatients
This trial supports the CHEST guideline recommendations to manage hemodynamically stable patients with acute PE as outpatients.3 It adds to the conversation by demonstrating higher rates of adverse events with hospitalization, even in high-risk subgroups (PESI Class III-V).
CAVEATS
Good study, but it wasn’t an RCT
While this is a well-designed cohort study, it is not an RCT. This study defined outpatient management as patients discharged from the ED or hospitalized for < 48 hours. However, only 59 of the 544 patients in the outpatient group were early hospital discharges; the rest were never admitted. Finally, a specialized thrombosis clinic followed up with the patients within 24 hours of discharge, and patients had telephone access to specialized health care professionals; such organization of care contributed to the safe outpatient management of these PE patients.
CHALLENGES TO IMPLEMENTATION
Insurance coverage may present an issue
Medication coverage of direct oral anticoagulants and low-molecular-weight heparin may present a barrier to patients treated in the outpatient setting who have no insurance or are insured by certain carriers.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2018. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2018;67[12]:777-779).
1. Roy PM, Corsi DJ, Carrier M, et al. Net clinical benefit of hospitalization versus outpatient management of patients with acute pulmonary embolism. J Thromb Haemost. 2017;15:685-694.
2. CDC. Venous thromboembolism data & statistics. www.cdc.gov/ncbddd/dvt/data.html. Accessed April 26, 2019.
3. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. CHEST. 2016;149:315-352.
4. Aujesky D, Obrosky DS, Stone RA, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172:1041-1046.
5. Vinson DR, Zehtabchi S, Yealy DM. Can selected patients with newly diagnosed pulmonary embolism be safely treated without hospitalization? A systematic review. Ann Emerg Med. 2012;60:651-662.
A 63-year-old woman with a history of hypertension presents to the emergency department (ED) with acute-onset shortness of breath and pleuritic chest pain after traveling across the country for a work conference. She has no history of cancer, liver disease, or renal disease. Her blood pressure is 140/80 mm Hg, and her heart rate, 90 beats/min. You diagnose an acute PE in this patient and start anticoagulation. Should you admit her to the hospital to decrease morbidity and mortality?
According to the CDC, venous thromboembolism (VTE) affects about 900,000 people each year, and about 60,000 to 100,000 of these patients die annually.2 Pulmonary embolism is the third leading cause of death from cardiovascular disease, following heart attack and stroke.3 Prompt diagnosis and treatment with systemic anticoagulation improves patient outcomes and decreases the risk for long-term complications.
The 2016 American College of Chest Physicians (CHEST) guideline on antithrombotic therapy for VTE recommends home treatment or early discharge over standard discharge (after the first 5 days of treatment) for patients who meet the following clinical criteria: “clinically stable with good cardiopulmonary reserve; no contraindications such as recent bleeding, severe renal or liver disease, or severe thrombocytopenia (ie, < 70,000/mm3); expected to be compliant with treatment; and feels well enough to be treated at home.”3
The guideline states that various clinical decision tools, such as the Pulmonary Embolism Severity Index (PESI), can aid in identifying low-risk patients to be considered for treatment at home. The PESI uses age, gender, vital signs, mental status, and a history of cancer, lung, and cardiac disease to stratify patients by risk.4
A systematic review of 1 randomized controlled trial (RCT) and 7 observational studies found that in low-risk patients, outpatient treatment was as safe as inpatient treatment.5 This more recent study determines the net clinical benefit of hospitalized versus outpatient management in a wider range of patients with acute PE, regardless of initial risk.1
STUDY SUMMARY
Hospitalization confers no benefit to stable PE patients
This retrospective, propensity-matched cohort study compared rates of adverse events in 1127 patients with acute PE managed in the hospital versus outpatient setting.1 Patients were classified as outpatients if they were discharged from the ED or discharged from the hospital within 48 hours of admission. Patients were included if a symptomatic acute PE was diagnosed via CT or high-probability ventilation-perfusion scan and excluded if they were younger than 19, were diagnosed with a PE during hospitalization, had chronic PE, or were hemodynamically unstable, among other factors. The investigators calculated PESI scores for all patients.
Propensity scores matched patients on 28 characteristics and known risk factors for adverse events to ensure the groups were similar. The primary outcome was rate of adverse events, including recurrent VTE, major bleeding, or death at 14 days. The secondary outcome included rates of the above during the 3-month follow-up period.
Continue to: Of the 1127 eligible patients...
Of the 1127 eligible patients, 1081 were included in the matched cohort, with 576 (53%) treated as hospitalized patients and 505 (47%) treated as outpatients. The mean age of the matched cohorts wa
The 14-day rate of adverse events was higher in hospitalized patients than in outpatients (13% vs 3.3%; odds ratio [OR], 5.07), with each of the adverse events that made up the primary outcome occurring more frequently in the hospitalized group (see Table). The rate of adverse events at 3 months was also greater for hospitalized patients compared with outpatients (21.7% vs 6.9%; OR, 4.9). The results remained similar for high-risk patients (Class III-V) based on their PESI score.
WHAT’S NEW
Higher rate of adverse events in inpatients
This trial supports the CHEST guideline recommendations to manage hemodynamically stable patients with acute PE as outpatients.3 It adds to the conversation by demonstrating higher rates of adverse events with hospitalization, even in high-risk subgroups (PESI Class III-V).
CAVEATS
Good study, but it wasn’t an RCT
While this is a well-designed cohort study, it is not an RCT. This study defined outpatient management as patients discharged from the ED or hospitalized for < 48 hours. However, only 59 of the 544 patients in the outpatient group were early hospital discharges; the rest were never admitted. Finally, a specialized thrombosis clinic followed up with the patients within 24 hours of discharge, and patients had telephone access to specialized health care professionals; such organization of care contributed to the safe outpatient management of these PE patients.
CHALLENGES TO IMPLEMENTATION
Insurance coverage may present an issue
Medication coverage of direct oral anticoagulants and low-molecular-weight heparin may present a barrier to patients treated in the outpatient setting who have no insurance or are insured by certain carriers.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2018. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2018;67[12]:777-779).
A 63-year-old woman with a history of hypertension presents to the emergency department (ED) with acute-onset shortness of breath and pleuritic chest pain after traveling across the country for a work conference. She has no history of cancer, liver disease, or renal disease. Her blood pressure is 140/80 mm Hg, and her heart rate, 90 beats/min. You diagnose an acute PE in this patient and start anticoagulation. Should you admit her to the hospital to decrease morbidity and mortality?
According to the CDC, venous thromboembolism (VTE) affects about 900,000 people each year, and about 60,000 to 100,000 of these patients die annually.2 Pulmonary embolism is the third leading cause of death from cardiovascular disease, following heart attack and stroke.3 Prompt diagnosis and treatment with systemic anticoagulation improves patient outcomes and decreases the risk for long-term complications.
The 2016 American College of Chest Physicians (CHEST) guideline on antithrombotic therapy for VTE recommends home treatment or early discharge over standard discharge (after the first 5 days of treatment) for patients who meet the following clinical criteria: “clinically stable with good cardiopulmonary reserve; no contraindications such as recent bleeding, severe renal or liver disease, or severe thrombocytopenia (ie, < 70,000/mm3); expected to be compliant with treatment; and feels well enough to be treated at home.”3
The guideline states that various clinical decision tools, such as the Pulmonary Embolism Severity Index (PESI), can aid in identifying low-risk patients to be considered for treatment at home. The PESI uses age, gender, vital signs, mental status, and a history of cancer, lung, and cardiac disease to stratify patients by risk.4
A systematic review of 1 randomized controlled trial (RCT) and 7 observational studies found that in low-risk patients, outpatient treatment was as safe as inpatient treatment.5 This more recent study determines the net clinical benefit of hospitalized versus outpatient management in a wider range of patients with acute PE, regardless of initial risk.1
STUDY SUMMARY
Hospitalization confers no benefit to stable PE patients
This retrospective, propensity-matched cohort study compared rates of adverse events in 1127 patients with acute PE managed in the hospital versus outpatient setting.1 Patients were classified as outpatients if they were discharged from the ED or discharged from the hospital within 48 hours of admission. Patients were included if a symptomatic acute PE was diagnosed via CT or high-probability ventilation-perfusion scan and excluded if they were younger than 19, were diagnosed with a PE during hospitalization, had chronic PE, or were hemodynamically unstable, among other factors. The investigators calculated PESI scores for all patients.
Propensity scores matched patients on 28 characteristics and known risk factors for adverse events to ensure the groups were similar. The primary outcome was rate of adverse events, including recurrent VTE, major bleeding, or death at 14 days. The secondary outcome included rates of the above during the 3-month follow-up period.
Continue to: Of the 1127 eligible patients...
Of the 1127 eligible patients, 1081 were included in the matched cohort, with 576 (53%) treated as hospitalized patients and 505 (47%) treated as outpatients. The mean age of the matched cohorts wa
The 14-day rate of adverse events was higher in hospitalized patients than in outpatients (13% vs 3.3%; odds ratio [OR], 5.07), with each of the adverse events that made up the primary outcome occurring more frequently in the hospitalized group (see Table). The rate of adverse events at 3 months was also greater for hospitalized patients compared with outpatients (21.7% vs 6.9%; OR, 4.9). The results remained similar for high-risk patients (Class III-V) based on their PESI score.
WHAT’S NEW
Higher rate of adverse events in inpatients
This trial supports the CHEST guideline recommendations to manage hemodynamically stable patients with acute PE as outpatients.3 It adds to the conversation by demonstrating higher rates of adverse events with hospitalization, even in high-risk subgroups (PESI Class III-V).
CAVEATS
Good study, but it wasn’t an RCT
While this is a well-designed cohort study, it is not an RCT. This study defined outpatient management as patients discharged from the ED or hospitalized for < 48 hours. However, only 59 of the 544 patients in the outpatient group were early hospital discharges; the rest were never admitted. Finally, a specialized thrombosis clinic followed up with the patients within 24 hours of discharge, and patients had telephone access to specialized health care professionals; such organization of care contributed to the safe outpatient management of these PE patients.
CHALLENGES TO IMPLEMENTATION
Insurance coverage may present an issue
Medication coverage of direct oral anticoagulants and low-molecular-weight heparin may present a barrier to patients treated in the outpatient setting who have no insurance or are insured by certain carriers.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
Copyright © 2018. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice (2018;67[12]:777-779).
1. Roy PM, Corsi DJ, Carrier M, et al. Net clinical benefit of hospitalization versus outpatient management of patients with acute pulmonary embolism. J Thromb Haemost. 2017;15:685-694.
2. CDC. Venous thromboembolism data & statistics. www.cdc.gov/ncbddd/dvt/data.html. Accessed April 26, 2019.
3. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. CHEST. 2016;149:315-352.
4. Aujesky D, Obrosky DS, Stone RA, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172:1041-1046.
5. Vinson DR, Zehtabchi S, Yealy DM. Can selected patients with newly diagnosed pulmonary embolism be safely treated without hospitalization? A systematic review. Ann Emerg Med. 2012;60:651-662.
1. Roy PM, Corsi DJ, Carrier M, et al. Net clinical benefit of hospitalization versus outpatient management of patients with acute pulmonary embolism. J Thromb Haemost. 2017;15:685-694.
2. CDC. Venous thromboembolism data & statistics. www.cdc.gov/ncbddd/dvt/data.html. Accessed April 26, 2019.
3. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. CHEST. 2016;149:315-352.
4. Aujesky D, Obrosky DS, Stone RA, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172:1041-1046.
5. Vinson DR, Zehtabchi S, Yealy DM. Can selected patients with newly diagnosed pulmonary embolism be safely treated without hospitalization? A systematic review. Ann Emerg Med. 2012;60:651-662.
ACOG guidance addresses cardiac contributors to maternal mortality
NASHVILLE, TENN. – according to new comprehensive guidance from the American College of Obstetricians and Gynecologists.
The toolkit algorithm is called the California Improving Health Care Response to Cardiovascular Disease in Pregnancy and Postpartum Toolkit. It was developed by the Cardiovascular Disease in Pregnancy Postpartum Task Force to serve as a resource for obstetrics, primary care and emergency medicine providers who provide prenatal care or interact with women during the postpartum period. It incldues an overview of clinical assessment and comprehensive management strategies for cardiovascular disease based on risk factors and presenting symptoms.
The guidance also calls for all pregnant and postpartum women with known or suspected CVD to undergo further evaluation by a “Pregnancy Heart Team that includes a cardiologist and maternal–fetal medicine subspecialist, or both, and other subspecialists as necessary.” The guidance was issued in Practice Bulletin 212, Pregnancy and Heart Disease, which is published in the May edition of Obstetrics & Gynecology (Obstet Gynecol. 2019 May;133[5]:e320-e356).
In all, 27 specific recommendations and conclusions relating to screening, diagnosis, and management of CVD for women during the prepregnancy period through the postpartum period are included in the guidance.
ACOG president Lisa Hollier, MD, convened the task force that developed this guidance to address cardiac contributors to maternal mortality, she said during a press briefing at the ACOG annual clinical and scientific meeting.
“When I began my presidency a year ago, my goal was to bring together a multidisciplinary group of clinicians ... to create clinical guidance that would make a difference in the lives of women," said Dr. Hollier, who is also a professor of obstetrics and gynecology at Baylor College of Medicine, Houston.
Part of her presidential initiative was centered on eliminating preventable maternal death, and this guidance has the potential to make strides toward that goal, she said. When it comes to CVD in pregnancy, “there is so much we can do to prevent negative outcomes and ensure that moms go home with their babies and are around to see them grow up,” she noted.
CVD is the leading cause of death in pregnant women and women in the postpartum period, accounting for 26.5% of U.S. pregnancy-related deaths.
“It’s critical that we as physicians and health care professionals develop expertise in recognizing the signs and symptoms so that we can save women’s lives,” she said in the press breifing. Dr. Hollier also implored her colleagues to “start using this guidance immediately and prevent more women from dying from cardiovascular complications of pregnancy.”
In this video interview, Dr. Hollier further explains the need for the guidance and its potential for improving maternal mortality rates.
Dr. Hollier reported having no relevant disclosures.
SOURCE: Hollier L et al., Obstet Gynecol. 2019 May;133[5]:e320-56.
NASHVILLE, TENN. – according to new comprehensive guidance from the American College of Obstetricians and Gynecologists.
The toolkit algorithm is called the California Improving Health Care Response to Cardiovascular Disease in Pregnancy and Postpartum Toolkit. It was developed by the Cardiovascular Disease in Pregnancy Postpartum Task Force to serve as a resource for obstetrics, primary care and emergency medicine providers who provide prenatal care or interact with women during the postpartum period. It incldues an overview of clinical assessment and comprehensive management strategies for cardiovascular disease based on risk factors and presenting symptoms.
The guidance also calls for all pregnant and postpartum women with known or suspected CVD to undergo further evaluation by a “Pregnancy Heart Team that includes a cardiologist and maternal–fetal medicine subspecialist, or both, and other subspecialists as necessary.” The guidance was issued in Practice Bulletin 212, Pregnancy and Heart Disease, which is published in the May edition of Obstetrics & Gynecology (Obstet Gynecol. 2019 May;133[5]:e320-e356).
In all, 27 specific recommendations and conclusions relating to screening, diagnosis, and management of CVD for women during the prepregnancy period through the postpartum period are included in the guidance.
ACOG president Lisa Hollier, MD, convened the task force that developed this guidance to address cardiac contributors to maternal mortality, she said during a press briefing at the ACOG annual clinical and scientific meeting.
“When I began my presidency a year ago, my goal was to bring together a multidisciplinary group of clinicians ... to create clinical guidance that would make a difference in the lives of women," said Dr. Hollier, who is also a professor of obstetrics and gynecology at Baylor College of Medicine, Houston.
Part of her presidential initiative was centered on eliminating preventable maternal death, and this guidance has the potential to make strides toward that goal, she said. When it comes to CVD in pregnancy, “there is so much we can do to prevent negative outcomes and ensure that moms go home with their babies and are around to see them grow up,” she noted.
CVD is the leading cause of death in pregnant women and women in the postpartum period, accounting for 26.5% of U.S. pregnancy-related deaths.
“It’s critical that we as physicians and health care professionals develop expertise in recognizing the signs and symptoms so that we can save women’s lives,” she said in the press breifing. Dr. Hollier also implored her colleagues to “start using this guidance immediately and prevent more women from dying from cardiovascular complications of pregnancy.”
In this video interview, Dr. Hollier further explains the need for the guidance and its potential for improving maternal mortality rates.
Dr. Hollier reported having no relevant disclosures.
SOURCE: Hollier L et al., Obstet Gynecol. 2019 May;133[5]:e320-56.
NASHVILLE, TENN. – according to new comprehensive guidance from the American College of Obstetricians and Gynecologists.
The toolkit algorithm is called the California Improving Health Care Response to Cardiovascular Disease in Pregnancy and Postpartum Toolkit. It was developed by the Cardiovascular Disease in Pregnancy Postpartum Task Force to serve as a resource for obstetrics, primary care and emergency medicine providers who provide prenatal care or interact with women during the postpartum period. It incldues an overview of clinical assessment and comprehensive management strategies for cardiovascular disease based on risk factors and presenting symptoms.
The guidance also calls for all pregnant and postpartum women with known or suspected CVD to undergo further evaluation by a “Pregnancy Heart Team that includes a cardiologist and maternal–fetal medicine subspecialist, or both, and other subspecialists as necessary.” The guidance was issued in Practice Bulletin 212, Pregnancy and Heart Disease, which is published in the May edition of Obstetrics & Gynecology (Obstet Gynecol. 2019 May;133[5]:e320-e356).
In all, 27 specific recommendations and conclusions relating to screening, diagnosis, and management of CVD for women during the prepregnancy period through the postpartum period are included in the guidance.
ACOG president Lisa Hollier, MD, convened the task force that developed this guidance to address cardiac contributors to maternal mortality, she said during a press briefing at the ACOG annual clinical and scientific meeting.
“When I began my presidency a year ago, my goal was to bring together a multidisciplinary group of clinicians ... to create clinical guidance that would make a difference in the lives of women," said Dr. Hollier, who is also a professor of obstetrics and gynecology at Baylor College of Medicine, Houston.
Part of her presidential initiative was centered on eliminating preventable maternal death, and this guidance has the potential to make strides toward that goal, she said. When it comes to CVD in pregnancy, “there is so much we can do to prevent negative outcomes and ensure that moms go home with their babies and are around to see them grow up,” she noted.
CVD is the leading cause of death in pregnant women and women in the postpartum period, accounting for 26.5% of U.S. pregnancy-related deaths.
“It’s critical that we as physicians and health care professionals develop expertise in recognizing the signs and symptoms so that we can save women’s lives,” she said in the press breifing. Dr. Hollier also implored her colleagues to “start using this guidance immediately and prevent more women from dying from cardiovascular complications of pregnancy.”
In this video interview, Dr. Hollier further explains the need for the guidance and its potential for improving maternal mortality rates.
Dr. Hollier reported having no relevant disclosures.
SOURCE: Hollier L et al., Obstet Gynecol. 2019 May;133[5]:e320-56.
REPORTING FROM ACOG 2019
Are guidelines relevant?
The recent publication of the changes and current levels of evidence for the guidelines from the American College of Cardiology, American Heart Association, and the European Society of Cardiology once again indicates that the level of high-quality scientific data supporting the decision making process is limited.
Of the thousands of recommendations among the guidelines provided, level A data – which is supported by at least two randomized, controlled trials (RCT), arguably the gold standard for the proof of benefit of a treatment program – was available in only 8.5% of ACC/AHA recommendations and in 14.2% of ESC recommendations. The recent review covering guidelines published from 2008 to 2018 has changed very little since the previous review carried out on data collected between 1999 and 2014 (JAMA. 2019 Mar 19;321[11]:1069-80). Admittedly, by the nature of their design, RCTs represent a very special subset of any disease process but they are the best we can do. The remainder of the guidelines is supported by nonrandomized data and consensus opinion of “experts.” The absence of randomized data in the rest of the guidelines creates an area of uncertainty between clinical practice and outcomes.
Guidelines have been with us for almost 30 years and have provided a near “cookbook recipe” for the treatment of almost the totality of cardiovascular care, both in the United States and Europe. There is little evidence that guideline dissemination results in a beneficial effect on cardiovascular care, instead reflecting the contemporary published research and informed opinion of our generation.
Despite the limitation of hard data based on RCTs, cardiovascular mortality has leveled off in the last few years. Although there are a number of areas that could be investigated with more focused and practical RCTs, there has been little enthusiasm by either the National Heart, Lung, and Blood Institute or the pharmaceutical or device industries to investigate existing cardiovascular drugs and/or devices to better elucidate nuanced therapy for subsets of heart disease that remain in the uncertain data areas. In fact, the current report shows that there has been little change in guidelines in the last 10 years. Most effort has been focused on new drug development and application.
Much of the advance in cardiovascular care and decrease in mortality is a result of the development of new drugs for the treatment of hypertension, management of hypercholesterolemia, and MI, together with coronary angiography and its interventional permutations of the previous 50 years. Those advances are now part of our knowledge base, our therapeutic DNA, and in the absence of some new scientific breakthrough, there is little to expect from future guideline development. Medical students and house officers are being taught as fact what we found as exciting new therapy just a few short years ago. Guidelines are now part of textbook knowledge and until new clinical outcome are reported, guidelines will probably serve a useful reference material but will not lead to much change in clinical care.
Even so, treating the patient in the clinic and in the hospital still requires the doctor to practice the “art” of medicine and to synthesize therapy based on the paucity of scientific data and quasi knowledge acquired over a generation.
The appreciation that much is unknown gives the doctor the humility required to be sensitive to patient needs and their symptoms and the aspiration for new, science-based data. This is still part of the excitement of being a doctor.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
The recent publication of the changes and current levels of evidence for the guidelines from the American College of Cardiology, American Heart Association, and the European Society of Cardiology once again indicates that the level of high-quality scientific data supporting the decision making process is limited.
Of the thousands of recommendations among the guidelines provided, level A data – which is supported by at least two randomized, controlled trials (RCT), arguably the gold standard for the proof of benefit of a treatment program – was available in only 8.5% of ACC/AHA recommendations and in 14.2% of ESC recommendations. The recent review covering guidelines published from 2008 to 2018 has changed very little since the previous review carried out on data collected between 1999 and 2014 (JAMA. 2019 Mar 19;321[11]:1069-80). Admittedly, by the nature of their design, RCTs represent a very special subset of any disease process but they are the best we can do. The remainder of the guidelines is supported by nonrandomized data and consensus opinion of “experts.” The absence of randomized data in the rest of the guidelines creates an area of uncertainty between clinical practice and outcomes.
Guidelines have been with us for almost 30 years and have provided a near “cookbook recipe” for the treatment of almost the totality of cardiovascular care, both in the United States and Europe. There is little evidence that guideline dissemination results in a beneficial effect on cardiovascular care, instead reflecting the contemporary published research and informed opinion of our generation.
Despite the limitation of hard data based on RCTs, cardiovascular mortality has leveled off in the last few years. Although there are a number of areas that could be investigated with more focused and practical RCTs, there has been little enthusiasm by either the National Heart, Lung, and Blood Institute or the pharmaceutical or device industries to investigate existing cardiovascular drugs and/or devices to better elucidate nuanced therapy for subsets of heart disease that remain in the uncertain data areas. In fact, the current report shows that there has been little change in guidelines in the last 10 years. Most effort has been focused on new drug development and application.
Much of the advance in cardiovascular care and decrease in mortality is a result of the development of new drugs for the treatment of hypertension, management of hypercholesterolemia, and MI, together with coronary angiography and its interventional permutations of the previous 50 years. Those advances are now part of our knowledge base, our therapeutic DNA, and in the absence of some new scientific breakthrough, there is little to expect from future guideline development. Medical students and house officers are being taught as fact what we found as exciting new therapy just a few short years ago. Guidelines are now part of textbook knowledge and until new clinical outcome are reported, guidelines will probably serve a useful reference material but will not lead to much change in clinical care.
Even so, treating the patient in the clinic and in the hospital still requires the doctor to practice the “art” of medicine and to synthesize therapy based on the paucity of scientific data and quasi knowledge acquired over a generation.
The appreciation that much is unknown gives the doctor the humility required to be sensitive to patient needs and their symptoms and the aspiration for new, science-based data. This is still part of the excitement of being a doctor.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
The recent publication of the changes and current levels of evidence for the guidelines from the American College of Cardiology, American Heart Association, and the European Society of Cardiology once again indicates that the level of high-quality scientific data supporting the decision making process is limited.
Of the thousands of recommendations among the guidelines provided, level A data – which is supported by at least two randomized, controlled trials (RCT), arguably the gold standard for the proof of benefit of a treatment program – was available in only 8.5% of ACC/AHA recommendations and in 14.2% of ESC recommendations. The recent review covering guidelines published from 2008 to 2018 has changed very little since the previous review carried out on data collected between 1999 and 2014 (JAMA. 2019 Mar 19;321[11]:1069-80). Admittedly, by the nature of their design, RCTs represent a very special subset of any disease process but they are the best we can do. The remainder of the guidelines is supported by nonrandomized data and consensus opinion of “experts.” The absence of randomized data in the rest of the guidelines creates an area of uncertainty between clinical practice and outcomes.
Guidelines have been with us for almost 30 years and have provided a near “cookbook recipe” for the treatment of almost the totality of cardiovascular care, both in the United States and Europe. There is little evidence that guideline dissemination results in a beneficial effect on cardiovascular care, instead reflecting the contemporary published research and informed opinion of our generation.
Despite the limitation of hard data based on RCTs, cardiovascular mortality has leveled off in the last few years. Although there are a number of areas that could be investigated with more focused and practical RCTs, there has been little enthusiasm by either the National Heart, Lung, and Blood Institute or the pharmaceutical or device industries to investigate existing cardiovascular drugs and/or devices to better elucidate nuanced therapy for subsets of heart disease that remain in the uncertain data areas. In fact, the current report shows that there has been little change in guidelines in the last 10 years. Most effort has been focused on new drug development and application.
Much of the advance in cardiovascular care and decrease in mortality is a result of the development of new drugs for the treatment of hypertension, management of hypercholesterolemia, and MI, together with coronary angiography and its interventional permutations of the previous 50 years. Those advances are now part of our knowledge base, our therapeutic DNA, and in the absence of some new scientific breakthrough, there is little to expect from future guideline development. Medical students and house officers are being taught as fact what we found as exciting new therapy just a few short years ago. Guidelines are now part of textbook knowledge and until new clinical outcome are reported, guidelines will probably serve a useful reference material but will not lead to much change in clinical care.
Even so, treating the patient in the clinic and in the hospital still requires the doctor to practice the “art” of medicine and to synthesize therapy based on the paucity of scientific data and quasi knowledge acquired over a generation.
The appreciation that much is unknown gives the doctor the humility required to be sensitive to patient needs and their symptoms and the aspiration for new, science-based data. This is still part of the excitement of being a doctor.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
Evaluating and managing postural tachycardia syndrome
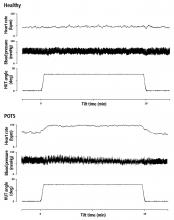
Some people, most of them relatively young women, experience lightheadedness, a racing heart, and other symptoms (but not hypotension) when they stand up, in a condition known as postural tachycardia syndrome (POTS).1 Although not known to shorten life,1 it can be physically and mentally debilitating.2,3 Therapy rarely cures it, but a multifaceted approach can substantially improve quality of life.
This review outlines the evaluation and diagnosis of POTS and provides guidance for a therapy regimen.
HOW IS POTS DEFINED?
POTS is a multifactorial syndrome rather than a specific disease. It is characterized by all of the following1,4–6:
- An increase in heart rate of ≥ 30 bpm, or ≥ 40 bpm for those under age 19, within 10 minutes of standing from a supine position
- Sustained tachycardia (> 30 seconds)
- Absence of orthostatic hypotension (a fall in blood pressure of ≥ 20/10 mm Hg)
- Frequent and chronic duration (≥ 6 months).
These features are critical to diagnosis. Hemodynamic criteria in isolation may describe postural tachycardia but are not sufficient to diagnose POTS.
The prevalence of POTS is estimated to be between 0.2% and 1.0%,7 affecting up to 3 million people in the United States. Most cases arise between ages 13 and 50, with a female-to-male ratio of 5:1.8
MANY NAMES, SAME CONDITION
In 1871, Da Costa9 described a condition he called “irritable heart syndrome” that had characteristics similar to those of POTS, including extreme fatigue and exercise intolerance. Decades later, Lewis10 and Wood11 provided more detailed descriptions of the disorder, renaming it “soldier’s heart” or “Da Costa syndrome.” As other cases were documented, more terms arose, including “effort syndrome” and “mitral valve prolapse syndrome.”
In 1982, Rosen and Cryer12 were the first to use the term “postural tachycardia syndrome” for patients with disabling tachycardia upon standing without orthostatic hypotension. In 1986, Fouad et al13 described patients with postural tachycardia, orthostatic intolerance, and a small degree of hypotension as having “idiopathic hypovolemia.”
In 1993, Schondorf and Low14 established the current definition of POTS, leading to increased awareness and research efforts to understand its pathophysiology.
MULTIFACTORIAL PATHOPHYSIOLOGY
During the last 2 decades, several often-overlapping forms of POTS have been recognized, all of which share a final common pathway of sustained orthostatic tachycardia.15–19 In addition, a number of common comorbidities were identified through review of large clinic populations of POTS.20,21
Hypovolemic POTS
Up to 70% of patients with POTS have hypovolemia. The average plasma volume deficit is about 13%, which typically causes only insignificant changes in heart rate and norepinephrine levels while a patient is supine. However, blood pooling associated with upright posture further compromises cardiac output and consequently increases sympathetic nerve activity. Abnormalities in the renin-angiotensin-aldosterone volume regulation system are also suspected to impair sodium retention, contributing to hypovolemia.1,22
Neuropathic POTS
About half of patients with POTS have partial sympathetic denervation (particularly in the lower limbs) and inadequate vasoconstriction upon standing, leading to reduced venous return and stroke volume.17,23 A compensatory increase in sympathetic tone results in tachycardia to maintain cardiac output and blood pressure.
Hyperadrenergic POTS
Up to 50% of patients with POTS have high norepinephrine levels (≥ 600 pg/mL) when upright. This subtype, hyperadrenergic POTS, is characterized by an increase in systolic blood pressure of at least 10 mm Hg within 10 minutes of standing, with concomitant tachycardia that can be similar to or greater than that seen in nonhyperadrenergic POTS. Patients with hyperadrenergic POTS tend to report more prominent symptoms of sympathetic activation, such as palpitations, anxiety, and tremulousness.24,25
Norepinephrine transporter deficiency
The norepinephrine transporter (NET) is on the presynaptic cleft of sympathetic neurons and serves to clear synaptic norepinephrine. NET deficiency leads to a hyperadrenergic state and elevated sympathetic nerve activation.18 NET deficiency may be induced by common antidepressants (eg, tricyclic antidepressants and serotonin-norepinephrine reuptake inhibitors) and attention-deficit disorder medications.4
Mast cell activation syndrome
The relationship between mast cell activation syndrome and POTS is poorly understood.4,26 Mast cell activation syndrome has been described in a subset of patients with POTS who have sinus tachycardia accompanied by severe episodic flushing. Patients with this subtype have a hyperadrenergic response to postural change and elevated urine methylhistamine during flushing episodes.
Patients with mast cell activation syndrome tend to have strong allergic symptoms and may also have severe gastrointestinal problems, food sensitivities, dermatographism, and neuropathy. Diagnosis can be difficult, as the condition is associated with numerous markers with varying sensitivity and specificity.
Autoimmune origin
A significant minority of patients report a viral-like illness before the onset of POTS symptoms, suggesting a possible autoimmune-mediated or inflammatory cause. Also, some autoimmune disorders (eg, Sjögren syndrome) can present with a POTS-like manifestation.
Research into the role of autoantibodies in the pathophysiology of POTS offers the potential to develop novel therapeutic targets. Autoantibodies that have been reported in POTS include those against M1 to M3 muscarinic receptors (present in over 87% of patients with POTS),27 cardiac lipid raft-associated proteins,28 adrenergic G-protein coupled receptors, alpha-1-adrenergic receptors, and beta-1- and beta-2-adrenergic receptors.29 Although commercial enzyme-linked immunosorbent assays can assess for these antibody fragments, it is not known whether targeting the antibodies improves outcomes. At this time, antibody testing for POTS should be confined to the research setting.
LINKS TO OTHER SYNDROMES
POTS is often associated with other conditions whose symptoms cannot be explained by postural intolerance or tachycardia.
Ehlers-Danlos syndromes are a group of inherited heterogeneous disorders involving joint hypermobility, skin hyperextensibility, and tissue fragility.30 The hypermobile subtype is most commonly associated with POTS, with patients often having symptoms of autonomic dysregulation and autonomic test abnormalities.31–33 Patients with POTS may have a history of joint subluxations, joint pain, cervical instability, and spontaneous epidural leaks. The reason for the overlap between the two syndromes is not clear.
Chronic fatigue syndrome is characterized by persistent fatigue that does not resolve with rest and is not necessarily associated with orthostatic changes. More than 75% of patients with POTS report general fatigue as a major complaint, and up to 23% meet the full criteria for chronic fatigue syndrome.34
DIAGNOSTIC STRATEGY
A patient presenting with symptoms suggestive of POTS should first undergo a detailed history and physical examination. Other causes of sinus tachycardia should be considered.
Detailed history, symptom review
The history should focus on determining symptom burden, including tachycardia onset, frequency, severity, and triggers; the presence of syncope; and the impact of symptoms on daily function and quality of life.
Presyncope and its associated symptoms occur in less than one-third of patients with POTS, and syncope is not a principal feature.4 If syncope is the predominant complaint, alternative causes should be investigated. The usual cause of syncope in the general population is thought to be vasovagal.
In addition to orthostatic intolerance, gastrointestinal disturbances are common in POTS, presenting as abdominal pain, heartburn, irregular bowel movements, diarrhea, or constipation. Symptoms of gastroparesis are less common. Gastrointestinal symptoms tend to be prolonged, lasting hours and occurring multiple times a week. They tend not to improve in the supine position.35
POTS-associated symptoms may develop insidiously, but patients often report onset after an acute stressor such as pregnancy, major surgery, or a presumed viral illness.4 Whether these putative triggers are causative or coincidental is unknown. Symptoms of orthostatic intolerance tend to be exacerbated by dehydration, heat, alcohol, exercise, and menstruation.36,37
Consider the family history: 1 in 8 patients with POTS reports familial orthostatic intolerance,38 suggesting a genetic role in some patients. Inquire about symptoms or a previous diagnosis of Ehlers-Danlos syndrome and mast cell activation syndrome.
Consider other conditions
Pheochromocytoma causes hyperadrenergic symptoms (eg, palpitations, lightheadedness) like those in POTS, but patients with pheochromocytoma typically have these symptoms while supine. Pheochromocytoma is also characterized by plasma norepinephrine levels much higher than in POTS.4 Plasma metanephrine testing helps diagnose or rule out pheochromocytoma.5
Inappropriate sinus tachycardia, like pheochromocytoma, also has clinical features similar to those of POTS, as well as tachycardia present when supine. It involves higher sympathetic tone and lower parasympathetic tone compared with POTS; patients commonly have a daytime resting heart rate of at least 100 bpm or a 24-hour mean heart rate of at least 90 bpm.1,42 While the intrinsic heart rate is heightened in inappropriate sinus tachycardia, it is not different between POTS patients and healthy individuals.42,43 Distinguishing POTS from inappropriate sinus tachycardia is further complicated by the broad inclusion criteria of most studies of inappropriate sinus tachycardia, which failed to exclude patients with POTS.44 The Heart Rhythm Society recently adopted distinct definitions for the 2 conditions.1
Physical examination: Focus on vital signs
Dependent acrocyanosis—dark red-blue discoloration of the lower legs that is cold to the touch—occurs in about half of patients with POTS upon standing.4 Dependent acrocyanosis is associated with joint hypermobility and Ehlers-Danlos syndrome, so these conditions should also be considered if findings are positive.
Laboratory testing for other causes
Laboratory testing is used mainly to detect primary causes of sinus tachycardia. Tests should include:
- Complete blood cell count with hematocrit (for severe anemia)
- Thyroid-stimulating hormone level (for hyperthyroidism)
- Electrolyte panel (for significant electrolyte disturbances).
Evidence is insufficient to support routinely measuring the vitamin B12 level, iron indices, and serum markers for celiac disease, although these may be done if the history or physical examination suggests related problems.4 Sicca symptoms (severe dry eye or dry mouth) should trigger evaluation for Sjögren syndrome.
Electrocardiography needed
Electrocardiography should be performed to investigate for cardiac conduction abnormalities as well as for resting markers of a supraventricular tachyarrhythmia. Extended ambulatory (Holter) monitoring may be useful to evaluate for a transient reentrant tachyarrhythmia4; however, it does not record body position, so it can be difficult to determine if detected episodes of tachycardia are related to posture.
Additional testing for select cases
Further investigation is usually not needed to diagnose POTS but should be considered in some cases. Advanced tests are typically performed at a tertiary care referral center and include:
- Quantitative sensory testing to evaluate for small-fiber neuropathy (ie, Quantitative Sudomotor Axon Reflex Test, or QSART), which occurs in the neuropathic POTS subtype
- Formal autonomic function testing to characterize neurovascular responsiveness
- Supine and standing plasma norepinephrine levels (fractionated catecholamines) to characterize the net activation of the sympathetic nervous system
- Blood volume assessments to assess hypovolemia
- Formal exercise testing to objectively quantify exercise capacity.
GRADED MANAGEMENT
No single universal gold-standard therapy exists for POTS, and management should be individually determined with the primary goals of treating symptoms and restoring function. A graded approach should be used, starting with conservative nonpharmacologic therapies and adding medications as needed.
While the disease course varies substantially from patient to patient, proper management is strongly associated with eventual symptom improvement.1
NONPHARMACOLOGIC STEPS FIRST
Education
Patients should be informed of the nature of their condition and referred to appropriate healthcare personnel. POTS is a chronic illness requiring individualized coping strategies, intensive physician interaction, and support of a multidisciplinary team. Patients and family members can be reassured that most symptoms improve over time with appropriate diagnosis and treatment.1 Patients should be advised to avoid aggravating triggers and activities.
Exercise
Exercise programs are encouraged but should be introduced gradually, as physical activity can exacerbate symptoms, especially at the outset. Several studies have reported benefits from a short-term (3-month) program, in which the patient gradually progresses from non-upright exercise (eg, rowing machine, recumbent cycle, swimming) to upright endurance exercises. At the end of these programs, significant cardiac remodeling, improved quality of life, and reduced heart rate responses to standing have been reported, and benefits have been reported to persist in patients who continued exercising after the 3-month study period.46,47
Despite the benefits of exercise interventions, compliance is low.46,47 To prevent early discouragement, patients should be advised that it can take 4 to 6 weeks of continued exercise before benefits appear. Patients are encouraged to exercise every other day for 30 minutes or more. Regimens should primarily focus on aerobic conditioning, but resistance training, concentrating on thigh muscles, can also help. Exercise is a treatment and not a cure, and benefits can rapidly disappear if regular activity (at least 3 times per week) is stopped.48
Compression stockings
Compression stockings help reduce peripheral venous pooling and enhance venous return to the heart. Waist-high stockings with compression of at least 30 to 40 mm Hg offer the best results.
Diet
Increased fluid and salt intake is advisable for patients with suspected hypovolemia. At least 2 to 3 L of water accompanied by 10 to 12 g of daily sodium intake is recommended.1 This can usually be accomplished with diet and salt added to food, but salt tablets can be used if the patient prefers. The resultant plasma volume expansion may help reduce the reflex tachycardia upon standing.49
Check medications
Rescue therapy with saline infusion
Intravenous saline infusion can augment blood volume in patients who are clinically decompensated and present with severe symptoms.1 Intermittent infusion of 1 L of normal saline has been found to significantly reduce orthostatic tachycardia and related symptoms in patients with POTS, contributing to improved quality of life.51,52
Chronic saline infusions are not recommended for long-term care because of the risk of access complications and infection.1 Moak et al53 reported a high rate of bacteremia in a cohort of children with POTS with regular saline infusions, most of whom had a central line. On the other hand, Ruzieh et al54 reported significantly improved symptoms with regular saline infusions without a high rate of complications, but patients in this study received infusions for only a few months and through a peripheral intravenous catheter.
DRUG THERAPY
No medications are approved by the US Food and Drug Administration (FDA) or Health Canada specifically for treating POTS, making all pharmacologic recommendations off-label. Although the drugs discussed below have been evaluated for POTS in controlled laboratory settings, they have yet to be tested in robust clinical trials.
Blood volume expansion
Several drugs expand blood volume, which may reduce orthostatic tachycardia.
Fludrocortisone is a synthetic aldosterone analogue that enhances sodium and water retention. Although one observational study found that it normalizes hemodynamic changes in response to orthostatic stress, no high-level evidence exists for its effectiveness for POTS.55 It is generally well tolerated, although possible adverse effects include hyperkalemia, hypertension, fatigue, nausea, headache, and edema.5,56
Desmopressin is a synthetic version of a natural antidiuretic hormone that increases kidney-mediated free-water reabsorption without sodium retention. It significantly reduces upright heart rate in patients with POTS and improves symptom burden. Although potential adverse effects include edema and headache, hyponatremia is the primary concern with daily use, especially with the increased water intake advised for POTS.57 Patients should be advised to use desmopressin no more than once a week for the acute improvement of symptoms. Intermittent monitoring of serum sodium levels is recommended for safety.
Erythropoietin replacement has been suggested for treating POTS to address the significant deficit in red blood cell volume. Although erythropoietin therapy has a direct vasoconstrictive effect and largely improves red blood cell volume in patients with POTS, it does not expand plasma volume, so orthostatic tachycardia is not itself reduced.22 Nevertheless, it may significantly improve POTS symptoms refractory to more common methods of treatment, and it should be reserved for such cases. In addition to the lack of effect on orthostatic tachycardia, drawbacks to using erythropoietin include its high cost, the need for subcutaneous administration, and the risk of life-threatening complications such as myocardial infarction and stroke.58,59
Heart rate-lowering agents
Propranolol, a nonselective beta-adrenergic antagonist, can significantly reduce standing heart rate and improve symptoms at low dosages (10–20 mg). Higher dosages can further restrain orthostatic tachycardia but are not as well tolerated, mainly due to hypotension and worsening of existing symptoms such as fatigue.60 Regular-acting propranolol works for about 4 to 5 hours per dose, so full-day coverage often requires dosing 4 times per day.
Ivabradine is a selective blocker of the “funny” (If) channel that reduces the sinus node firing rate without affecting blood pressure, so it slows heart rate without causing supine hypertension or orthostatic hypotension.
A retrospective case series found that 60% of patients with POTS treated with ivabradine reported symptomatic improvement, and all patients experienced reduced tachycardia with continued use.61 Ivabradine has not been compared with placebo or propranolol in a randomized controlled trial, and it has not been well studied in pregnancy and so should be avoided because of potential teratogenic effects.
When prescribing ivabradine for women of childbearing age, a negative pregnancy test may be documented prior to initiation of therapy, and the use of highly effective methods of contraception is recommended. Ivabradine should be avoided in women contemplating pregnancy. Insurance coverage can limit access to ivabradine in the United States.
Central nervous system sympatholytics
Patients with prominent hyperadrenergic features may benefit from central sympatholytic agents. However, these drugs may not be well tolerated in patients with neuropathic POTS because of the effects of reduced systemic vascular resistance5 and the possible exacerbation of drowsiness, fatigue, and mental clouding.4 Patients can be extremely sensitive to these medications, so they should initially be prescribed at the lowest dose, then gradually increased as tolerated.
Clonidine, an alpha-2-adrenergic agonist, decreases central sympathetic tone. In hyperadrenergic patients, clonidine can stabilize heart rate and blood pressure, thereby reducing orthostatic symptoms.62
Methyldopa has effects similar to those of clonidine but is easier to titrate owing to its longer half-life.63 Methyldopa is typically started at 125 mg at bedtime and increased to 125 mg twice daily, if tolerated.
Other agents
Midodrine is a prodrug. The active form, an alpha-1-adrenergic agonist, constricts peripheral veins and arteries to increase vascular resistance and venous return, thereby reducing orthostatic tachycardia.52 It is most useful in patients with impaired peripheral vasoconstriction (eg, neuropathic POTS) and may be less effective in those with hyperadrenergic POTS.64 Major limitations of midodrine include worsening supine hypertension and possible urinary retention.39
Because of midodrine’s short half-life, frequent dosing is required during daytime hours (eg, 8 AM, noon, and 4 PM), but it should not be taken within 4 to 5 hours of sleep because of the risk of supine hypertension. Midodrine is typically started at 2.5 to 5 mg per dose and can be titrated up to 15 mg per dose.
Midodrine is an FDA pregnancy category C drug (adverse effects in pregnancy seen in animal models, but evidence lacking in humans). While ideally it should be avoided, we have used it safely in pregnant women with disabling POTS symptoms.
Pyridostigmine, an acetylcholinesterase inhibitor, increases cardiovagal tone and possibly sympathetic tone. It has been reported to significantly reduce standing heart rate and improve symptom burden in patients with POTS.65 However, pyridostigmine increases gastrointestinal mobility, leading to severe adverse effects in over 20% of patients, including abdominal cramps, nausea, and diarrhea.66
Droxidopa, a synthetic amino acid precursor of norepinephrine, improves dizziness and fatigue in POTS with minimal effects on blood pressure.67
Modafinil, a psychostimulant, may improve POTS-associated cognitive symptoms.4 It also raises upright blood pressure without significantly worsening standing heart rate or acute orthostatic symptoms.68
EFFECTS OF COMORBID DISORDERS ON MANAGEMENT
Ehlers-Danlos syndrome
Pharmacologic approaches to POTS should not be altered based on the presence of Ehlers-Danlos syndrome, but because many of these patients are prone to joint dislocation, exercise prescriptions may need adjusting.
A medical genetics consult is recommended for patients with Ehlers-Danlos syndrome. Although the hypermobile type (the form most commonly associated with POTS) is not associated with aortopathy, it can be confused with classical and vascular Ehlers-Danlos syndromes, which require serial aortic screening.30
Mast cell activation syndrome
Consultation with an allergist or immunologist may help patients with severe symptoms.
Autoantibodies and autoimmunity
Treatment of the underlying disorder is recommended and can result in significantly improved POTS symptoms.
SPECIALTY CARE REFERRAL
POTS can be challenging to manage. Given the range of physiologic, emotional, and functional distress patients experience, it often requires significant physician time and multidisciplinary care. Patients with continued severe or debilitating symptoms may benefit from referral to a tertiary-care center with experience in autonomic nervous system disorders.
PROGNOSIS
Limited data are available on the long-term prognosis of POTS, and more studies are needed in pediatric and adult populations. No deaths have been reported in the handful of published cases of POTS in patients older than 50.1 Some pediatric studies suggest that some teenagers “outgrow” their POTS. However, these data are not robust, and an alternative explanation is that as they get older, they see adult physicians for their POTS symptoms and so are lost to study follow-up.6,44,69
We have not often seen POTS simply resolve without ongoing treatment. However, in our experience, most patients have improved symptoms and function with multimodal treatment (ie, exercise, salt, water, stockings, and some medications) and time.
- Sheldon RS, Grubb BP 2nd, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 2015; 12(6):e41–e63. doi:10.1016/j.hrthm.2015.03.029
- Bagai K, Song Y, Ling JF, et al. Sleep disturbances and diminished quality of life in postural tachycardia syndrome. J Clin Sleep Med 2011; 7(2):204–210. pmid:21509337
- Benrud-Larson LM, Dewar MS, Sandroni P, Rummans TA, Haythornthwaite JA, Low PA. Quality of life in patients with postural tachycardia syndrome. Mayo Clin Proc 2002; 77(6):531–537. doi:10.4065/77.6.531
- Raj SR. Postural tachycardia syndrome (POTS). Circulation 2013; 127(23):2336–2342. doi:10.1161/CIRCULATIONAHA.112.144501
- Raj SR. The postural tachycardia syndrome (POTS): pathophysiology, diagnosis & management. Indian Pacing Electrophysiol J 2006; 6(2):84–99. pmid:16943900
- Singer W, Sletten DM, Opfer-Gehrking TL, Brands CK, Fischer PR, Low PA. Postural tachycardia in children and adolescents: what is abnormal? J Pediatr 2012; 160(2):222–226. doi:10.1016/j.jpeds.2011.08.054
- Mar PL, Raj SR. Neuronal and hormonal perturbations in postural tachycardia syndrome. Front Physiol 2014; 5:220. doi:10.3389/fphys.2014.00220
- Garland EM, Raj SR, Black BK, Harris PA, Robertson D. The hemodynamic and neurohumoral phenotype of postural tachycardia syndrome. Neurology 2007; 69(8):790–798. doi:10.1212/01.wnl.0000267663.05398.40
- Da Costa JM. On irritable heart: a clinical study of a form of functional cardiac disorder and its consequences. Am J Med Sci 1871; 61(121):2–52.
- Lewis T. The tolerance of physical exertion, as shown by soldiers suffering from so-called “irritable heart.” Br Med J 1918; 1(2987):363–365. pmid:20768980
- Wood P. Da Costa’s syndrome (or effort syndrome): lecture I. Br Med J 1941; 1(4194):767–772. pmid:20783672
- Rosen SG, Cryer PE. Postural tachycardia syndrome. Reversal of sympathetic hyperresponsiveness and clinical improvement during sodium loading. Am J Med 1982; 72(5):847–850.
- Fouad FM, Tadena-Thome L, Bravo EL, Tarazi RC. Idiopathic hypovolemia. Ann Intern Med 1986; 104(3):298–303. pmid:3511818
- Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology 1993; 43(1):132–137. pmid:8423877
- Vernino S, Low PA, Fealey RD, Stewart JD, Farrugia G, Lennon VA. Autoantibodies to ganglionic acetylcholine receptors in autoimmune autonomic neuropathies. N Engl J Med 2000; 343(12):847–855. doi:10.1056/NEJM200009213431204
- Raj SR, Robertson D. Blood volume perturbations in the postural tachycardia syndrome. Am J Med Sci 2007; 334(1):57–60. doi:10.1097/MAJ.0b013e318063c6c0
- Jacob G, Costa F, Shannon JR, et al. The neuropathic postural tachycardia syndrome. N Engl J Med 2000; 343(14):1008–1014. doi:10.1056/NEJM200010053431404
- Shannon JR, Flattem NL, Jordan J, et al. Orthostatic intolerance and tachycardia associated with norepinephrine-transporter deficiency. N Engl J Med 2000; 342(8):541–549. doi:10.1056/NEJM200002243420803
- Jones PK, Shaw BH, Raj SR. Clinical challenges in the diagnosis and management of postural tachycardia syndrome. Pract Neurol 2016; 16(6):431–438. doi:10.1136/practneurol-2016-001405
- Gunning WT, Karabin BL, Blomquist TM, Grubb BP. Postural orthostatic tachycardia syndrome is associated with platelet storage pool deficiency. Medicine (Baltimore) 2016; 95(37):e4849. doi:10.1097/MD.0000000000004849
- Kanjwal K, Sheikh M, Karabin B, Kanjwal Y, Grubb BP. Neurocardiogenic syncope coexisting with postural orthostatic tachycardia syndrome in patients suffering from orthostatic intolerance: a combined form of autonomic dysfunction. Pacing Clin Electrophysiol 2011; 34(5):549–554. doi:10.1111/j.1540-8159.2010.02994.x
- Raj SR, Biaggioni I, Yamhure PC, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation 2005; 111(13):1574–1582. doi:10.1161/01.CIR.0000160356.97313.5D
- Gibbons CH, Bonyhay I, Benson A, Wang N, Freeman R. Structural and functional small fiber abnormalities in the neuropathic postural tachycardia syndrome. PLoS One 2013; 8(12):e84716. doi:10.1371/journal.pone.0084716
- Low PA, Sandroni P, Joyner M, Shen WK. Postural tachycardia syndrome (POTS). J Cardiovasc Electrophysiol 2009; 20(3):352–358. doi:10.1111/j.1540-8167.2008.01407.x
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Grubb BP. Clinical presentation and management of patients with hyperadrenergic postural orthostatic tachycardia syndrome. A single center experience. Cardiol J 2011; 18(5):527–531. pmid:21947988
- Shibao C, Arzubiaga C, Roberts J, et al. Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension 2005; 45(3):385–390. doi:10.1161/01.HYP.0000158259.68614.40
- Dubey D, Hopkins S, Vernino S. M1 and M2 muscarinic receptor antibodies among patients with postural orthostatic tachycardia syndrome: potential disease biomarker [abstract]. J Clin Neuromuscul Dis 2016; 17(3):179S.
- Wang XL, Ling TY, Charlesworth MC, et al. Autoimmunoreactive IgGs against cardiac lipid raft-associated proteins in patients with postural orthostatic tachycardia syndrome. Transl Res 2013; 162(1):34–44. doi:10.1016/j.trsl.2013.03.002
- Li H, Yu X, Liles C, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc 2014; 3(1):e000755. doi:10.1161/JAHA.113.000755
- Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet 2017; 175(1):8–26. doi:10.1002/ajmg.c.31552
- Wallman D, Weinberg J, Hohler AD. Ehlers-Danlos syndrome and postural tachycardia syndrome: a relationship study. J Neurol Sci 2014; 340(1-2):99–102. doi:10.1016/j.jns.2014.03.002
- De Wandele I, Calders P, Peersman W, et al. Autonomic symptom burden in the hypermobility type of Ehlers-Danlos syndrome: a comparative study with two other EDS types, fibromyalgia, and healthy controls. Semin Arthritis Rheum 2014; 44(3):353–361. doi:10.1016/j.semarthrit.2014.05.013
- Gazit Y, Nahir AM, Grahame R, Jacob G. Dysautonomia in the joint hypermobility syndrome. Am J Med 2003; 115(1):33–40. pmid:12867232
- Okamoto LE, Raj SR, Peltier A, et al. Neurohumoral and haemodynamic profile in postural tachycardia and chronic fatigue syndromes. Clin Sci (Lond) 2012; 122(4):183–192. doi:10.1042/CS20110200
- Wang LB, Culbertson CJ, Deb A, Morgenshtern K, Huang H, Hohler AD. Gastrointestinal dysfunction in postural tachycardia syndrome. J Neurol Sci 2015; 359(1-2):193–196. doi:10.1016/j.jns.2015.10.052
- Raj S, Sheldon R. Management of postural tachycardia syndrome, inappropriate sinus tachycardia and vasovagal syncope. Arrhythm Electrophysiol Rev 2016; 5(2):122–129. doi:10.15420/AER.2016.7.2
- Peggs KJ, Nguyen H, Enayat D, Keller NR, Al-Hendy A, Raj SR. Gynecologic disorders and menstrual cycle lightheadedness in postural tachycardia syndrome. Int J Gynaecol Obstet 2012; 118(3):242–246. doi:10.1016/j.ijgo.2012.04.014
- Thieben MJ, Sandroni P, Sletten DM, et al. Postural orthostatic tachycardia syndrome: the Mayo Clinic experience. Mayo Clin Proc 2007; 82(3):308–313. doi:10.4065/82.3.308
- Deb A, Morgenshtern K, Culbertson CJ, Wang LB, Hohler AD. A survey-based analysis of symptoms in patients with postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2015; 28(7):157–159. pmid:25829642
- Ertek S, Cicero AF. Hyperthyroidism and cardiovascular complications: a narrative review on the basis of pathophysiology. Arch Med Sci 2013; 9(5):944–952. doi:10.5114/aoms.2013.38685
- Rangno RE, Langlois S. Comparison of withdrawal phenomena after propranolol, metoprolol and pindolol. Br J Clin Pharmacol 1982; 13(suppl 2):345S–351S. pmid:6125187
- Nwazue VC, Paranjape SY, Black BK, et al. Postural tachycardia syndrome and inappropriate sinus tachycardia: role of autonomic modulation and sinus node automaticity. J Am Heart Assoc 2014; 3(2):e000700. doi:10.1161/JAHA.113.000700
- Morillo CA, Klein GJ, Thakur RK, Li H, Zardini M, Yee R. Mechanism of “inappropriate” sinus tachycardia. Role of sympathovagal balance. Circulation 1994; 90(2):873–877. pmid:7913886
- Grubb BP. Postural tachycardia syndrome. Circulation 2008; 117(21):2814–2817. doi:10.1161/CIRCULATIONAHA.107.761643
- Bhatia R, Kizilbash SJ, Ahrens SP, et al. Outcomes of adolescent-onset postural orthostatic tachycardia syndrome. J Pediatr 2016; 173:149–153. doi:10.1016/j.jpeds.2016.02.035
- George SA, Bivens TB, Howden EJ, et al. The international POTS registry: evaluating the efficacy of an exercise training intervention in a community setting. Heart Rhythm 2016; 13(4):943–950. doi:10.1016/j.hrthm.2015.12.012
- Fu Q, VanGundy TB, Galbreath MM, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2010; 55(25):2858–2868. doi:10.1016/j.jacc.2010.02.043
- Raj SR. Row, row, row your way to treating postural tachycardia syndrome. Heart Rhythm 2016; 13(4):951–952. doi:10.1016/j.hrthm.2015.12.039
- Celedonio JE, Garland EM, Nwazue VC, et al. Effects of high sodium intake on blood volume and catecholamines in patients with postural tachycardia syndrome and healthy females [abstract]. Clin Auton Res 2014; 24:211.
- Garland EM, Celedonio JE, Raj SR. Postural tachycardia syndrome: beyond orthostatic intolerance. Curr Neurol Neurosci Rep 2015; 15(9):60. doi:10.1007/s11910-015-0583-8
- Gordon VM, Opfer-Gehrking TL, Novak V, Low PA. Hemodynamic and symptomatic effects of acute interventions on tilt in patients with postural tachycardia syndrome. Clin Auton Res 2000; 10:29–33. pmid:10750641
- Jacob G, Shannon JR, Black B, et al. Effects of volume loading and pressor agents in idiopathic orthostatic tachycardia. Circulation 1997; 96(2):575–580. pmid:9244228
- Moak JP, Leong D, Fabian R, et al. Intravenous hydration for management of medication-resistant orthostatic intolerance in the adolescent and young adult. Pediatr Cardiol 2016; 37(2):278–282. doi:10.1007/s00246-015-1274-6
- Ruzieh M, Baugh A, Dasa O, et al. Effects of intermittent intravenous saline infusions in patients with medication-refractory postural tachycardia syndrome. J Interv Card Electrophysiol 2017; 48(3):255–260. doi:10.1007/s10840-017-0225-y
- Freitas J, Santos R, Azevedo E, Costa O, Carvalho M, de Freitas AF. Clinical improvement in patients with orthostatic intolerance after treatment with bisoprolol and fludrocortisone. Clin Auton Res 2000; 10(5):293–299. pmid:11198485
- Lee AK, Krahn AD. Evaluation of syncope: focus on diagnosis and treatment of neurally mediated syncope. Expert Rev Cardiovasc Ther 2016; 14(6):725–736. doi:10.1586/14779072.2016.1164034
- Coffin ST, Black BK, Biaggioni I, et al. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm 2012; 9(9):1484–1490. doi:10.1016/j.hrthm.2012.05.002
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Sheikh M, Grubb BP. Erythropoietin in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2012; 19(2):92–95. doi:10.1097/MJT.0b013e3181ef621a
- Hoeldtke RD, Horvath GG, Bryner KD. Treatment of orthostatic tachycardia with erythropoietin. Am J Med 1995; 99(5):525–529. pmid:7485211
- Raj SR, Black BK, Biaggioni I, et al. Propranolol decreases tachycardia and improves symptoms in the postural tachycardia syndrome: less is more. Circulation 2009; 120(9):725–734. doi:10.1161/CIRCULATIONAHA.108.846501
- McDonald C, Frith J, Newton JL. Single centre experience of ivabradine in postural orthostatic tachycardia syndrome. Europace 2011; 13(3):427–430. doi:10.1093/europace/euq390
- Gaffney FA, Lane LB, Pettinger W, Blomqvist G. Effects of long-term clonidine administration on the hemodynamic and neuroendocrine postural responses of patients with dysautonomia. Chest 1983; 83(suppl 2):436–438. pmid:6295714
- Jacob G, Biaggioni I. Idiopathic orthostatic intolerance and postural tachycardia syndromes. Am J Med Sci 1999; 317(2):88–101. pmid:10037112
- Ross AJ, Ocon AJ, Medow MS, Stewart JM. A double-blind placebo-controlled cross-over study of the vascular effects of midodrine in neuropathic compared with hyperadrenergic postural tachycardia syndrome. Clin Sci (Lond) 2014; 126(4):289–296. doi:10.1042/CS20130222
- Raj SR, Black BK, Biaggioni I, Harris PA, Robertson D. Acetylcholinesterase inhibition improves tachycardia in postural tachycardia syndrome. Circulation 2005; 111(21):2734–2340. doi:10.1161/CIRCULATIONAHA.104.497594
- Kanjwal K, Karabin B, Sheikh M, et al. Pyridostigmine in the treatment of postural orthostatic tachycardia: A single-center experience. Pacing Clin Electrophysiol 2011; 34(6):750–755. doi:10.1111/j.1540-8159.2011.03047.x
- Ruzieh M, Dasa O, Pacenta A, Karabin B, Grubb B. Droxidopa in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2017; 24(2):e157–e161. doi:10.1097/MJT.0000000000000468
- Kpaeyeh AG Jr, Mar PL, Raj V, et al. Hemodynamic profiles and tolerability of modafinil in the treatment of POTS: a randomized placebo-controlled trial. J Clin Psychopharmacol 2014; 34(6):738–741. doi:10.1097/JCP.0000000000000221
- Lai CC, Fischer PR, Brands CK, et al. Outcomes in adolescents with postural orthostatic tachycardia syndrome treated with midodrine and beta-blockers. Pacing Clin Electrophysiol 2009; 32(2):234–238. doi:10.1111/j.1540-8159.2008.02207.x
Some people, most of them relatively young women, experience lightheadedness, a racing heart, and other symptoms (but not hypotension) when they stand up, in a condition known as postural tachycardia syndrome (POTS).1 Although not known to shorten life,1 it can be physically and mentally debilitating.2,3 Therapy rarely cures it, but a multifaceted approach can substantially improve quality of life.
This review outlines the evaluation and diagnosis of POTS and provides guidance for a therapy regimen.
HOW IS POTS DEFINED?
POTS is a multifactorial syndrome rather than a specific disease. It is characterized by all of the following1,4–6:
- An increase in heart rate of ≥ 30 bpm, or ≥ 40 bpm for those under age 19, within 10 minutes of standing from a supine position
- Sustained tachycardia (> 30 seconds)
- Absence of orthostatic hypotension (a fall in blood pressure of ≥ 20/10 mm Hg)
- Frequent and chronic duration (≥ 6 months).
These features are critical to diagnosis. Hemodynamic criteria in isolation may describe postural tachycardia but are not sufficient to diagnose POTS.
The prevalence of POTS is estimated to be between 0.2% and 1.0%,7 affecting up to 3 million people in the United States. Most cases arise between ages 13 and 50, with a female-to-male ratio of 5:1.8
MANY NAMES, SAME CONDITION
In 1871, Da Costa9 described a condition he called “irritable heart syndrome” that had characteristics similar to those of POTS, including extreme fatigue and exercise intolerance. Decades later, Lewis10 and Wood11 provided more detailed descriptions of the disorder, renaming it “soldier’s heart” or “Da Costa syndrome.” As other cases were documented, more terms arose, including “effort syndrome” and “mitral valve prolapse syndrome.”
In 1982, Rosen and Cryer12 were the first to use the term “postural tachycardia syndrome” for patients with disabling tachycardia upon standing without orthostatic hypotension. In 1986, Fouad et al13 described patients with postural tachycardia, orthostatic intolerance, and a small degree of hypotension as having “idiopathic hypovolemia.”
In 1993, Schondorf and Low14 established the current definition of POTS, leading to increased awareness and research efforts to understand its pathophysiology.
MULTIFACTORIAL PATHOPHYSIOLOGY
During the last 2 decades, several often-overlapping forms of POTS have been recognized, all of which share a final common pathway of sustained orthostatic tachycardia.15–19 In addition, a number of common comorbidities were identified through review of large clinic populations of POTS.20,21
Hypovolemic POTS
Up to 70% of patients with POTS have hypovolemia. The average plasma volume deficit is about 13%, which typically causes only insignificant changes in heart rate and norepinephrine levels while a patient is supine. However, blood pooling associated with upright posture further compromises cardiac output and consequently increases sympathetic nerve activity. Abnormalities in the renin-angiotensin-aldosterone volume regulation system are also suspected to impair sodium retention, contributing to hypovolemia.1,22
Neuropathic POTS
About half of patients with POTS have partial sympathetic denervation (particularly in the lower limbs) and inadequate vasoconstriction upon standing, leading to reduced venous return and stroke volume.17,23 A compensatory increase in sympathetic tone results in tachycardia to maintain cardiac output and blood pressure.
Hyperadrenergic POTS
Up to 50% of patients with POTS have high norepinephrine levels (≥ 600 pg/mL) when upright. This subtype, hyperadrenergic POTS, is characterized by an increase in systolic blood pressure of at least 10 mm Hg within 10 minutes of standing, with concomitant tachycardia that can be similar to or greater than that seen in nonhyperadrenergic POTS. Patients with hyperadrenergic POTS tend to report more prominent symptoms of sympathetic activation, such as palpitations, anxiety, and tremulousness.24,25
Norepinephrine transporter deficiency
The norepinephrine transporter (NET) is on the presynaptic cleft of sympathetic neurons and serves to clear synaptic norepinephrine. NET deficiency leads to a hyperadrenergic state and elevated sympathetic nerve activation.18 NET deficiency may be induced by common antidepressants (eg, tricyclic antidepressants and serotonin-norepinephrine reuptake inhibitors) and attention-deficit disorder medications.4
Mast cell activation syndrome
The relationship between mast cell activation syndrome and POTS is poorly understood.4,26 Mast cell activation syndrome has been described in a subset of patients with POTS who have sinus tachycardia accompanied by severe episodic flushing. Patients with this subtype have a hyperadrenergic response to postural change and elevated urine methylhistamine during flushing episodes.
Patients with mast cell activation syndrome tend to have strong allergic symptoms and may also have severe gastrointestinal problems, food sensitivities, dermatographism, and neuropathy. Diagnosis can be difficult, as the condition is associated with numerous markers with varying sensitivity and specificity.
Autoimmune origin
A significant minority of patients report a viral-like illness before the onset of POTS symptoms, suggesting a possible autoimmune-mediated or inflammatory cause. Also, some autoimmune disorders (eg, Sjögren syndrome) can present with a POTS-like manifestation.
Research into the role of autoantibodies in the pathophysiology of POTS offers the potential to develop novel therapeutic targets. Autoantibodies that have been reported in POTS include those against M1 to M3 muscarinic receptors (present in over 87% of patients with POTS),27 cardiac lipid raft-associated proteins,28 adrenergic G-protein coupled receptors, alpha-1-adrenergic receptors, and beta-1- and beta-2-adrenergic receptors.29 Although commercial enzyme-linked immunosorbent assays can assess for these antibody fragments, it is not known whether targeting the antibodies improves outcomes. At this time, antibody testing for POTS should be confined to the research setting.
LINKS TO OTHER SYNDROMES
POTS is often associated with other conditions whose symptoms cannot be explained by postural intolerance or tachycardia.
Ehlers-Danlos syndromes are a group of inherited heterogeneous disorders involving joint hypermobility, skin hyperextensibility, and tissue fragility.30 The hypermobile subtype is most commonly associated with POTS, with patients often having symptoms of autonomic dysregulation and autonomic test abnormalities.31–33 Patients with POTS may have a history of joint subluxations, joint pain, cervical instability, and spontaneous epidural leaks. The reason for the overlap between the two syndromes is not clear.
Chronic fatigue syndrome is characterized by persistent fatigue that does not resolve with rest and is not necessarily associated with orthostatic changes. More than 75% of patients with POTS report general fatigue as a major complaint, and up to 23% meet the full criteria for chronic fatigue syndrome.34
DIAGNOSTIC STRATEGY
A patient presenting with symptoms suggestive of POTS should first undergo a detailed history and physical examination. Other causes of sinus tachycardia should be considered.
Detailed history, symptom review
The history should focus on determining symptom burden, including tachycardia onset, frequency, severity, and triggers; the presence of syncope; and the impact of symptoms on daily function and quality of life.
Presyncope and its associated symptoms occur in less than one-third of patients with POTS, and syncope is not a principal feature.4 If syncope is the predominant complaint, alternative causes should be investigated. The usual cause of syncope in the general population is thought to be vasovagal.
In addition to orthostatic intolerance, gastrointestinal disturbances are common in POTS, presenting as abdominal pain, heartburn, irregular bowel movements, diarrhea, or constipation. Symptoms of gastroparesis are less common. Gastrointestinal symptoms tend to be prolonged, lasting hours and occurring multiple times a week. They tend not to improve in the supine position.35
POTS-associated symptoms may develop insidiously, but patients often report onset after an acute stressor such as pregnancy, major surgery, or a presumed viral illness.4 Whether these putative triggers are causative or coincidental is unknown. Symptoms of orthostatic intolerance tend to be exacerbated by dehydration, heat, alcohol, exercise, and menstruation.36,37
Consider the family history: 1 in 8 patients with POTS reports familial orthostatic intolerance,38 suggesting a genetic role in some patients. Inquire about symptoms or a previous diagnosis of Ehlers-Danlos syndrome and mast cell activation syndrome.
Consider other conditions
Pheochromocytoma causes hyperadrenergic symptoms (eg, palpitations, lightheadedness) like those in POTS, but patients with pheochromocytoma typically have these symptoms while supine. Pheochromocytoma is also characterized by plasma norepinephrine levels much higher than in POTS.4 Plasma metanephrine testing helps diagnose or rule out pheochromocytoma.5
Inappropriate sinus tachycardia, like pheochromocytoma, also has clinical features similar to those of POTS, as well as tachycardia present when supine. It involves higher sympathetic tone and lower parasympathetic tone compared with POTS; patients commonly have a daytime resting heart rate of at least 100 bpm or a 24-hour mean heart rate of at least 90 bpm.1,42 While the intrinsic heart rate is heightened in inappropriate sinus tachycardia, it is not different between POTS patients and healthy individuals.42,43 Distinguishing POTS from inappropriate sinus tachycardia is further complicated by the broad inclusion criteria of most studies of inappropriate sinus tachycardia, which failed to exclude patients with POTS.44 The Heart Rhythm Society recently adopted distinct definitions for the 2 conditions.1
Physical examination: Focus on vital signs
Dependent acrocyanosis—dark red-blue discoloration of the lower legs that is cold to the touch—occurs in about half of patients with POTS upon standing.4 Dependent acrocyanosis is associated with joint hypermobility and Ehlers-Danlos syndrome, so these conditions should also be considered if findings are positive.
Laboratory testing for other causes
Laboratory testing is used mainly to detect primary causes of sinus tachycardia. Tests should include:
- Complete blood cell count with hematocrit (for severe anemia)
- Thyroid-stimulating hormone level (for hyperthyroidism)
- Electrolyte panel (for significant electrolyte disturbances).
Evidence is insufficient to support routinely measuring the vitamin B12 level, iron indices, and serum markers for celiac disease, although these may be done if the history or physical examination suggests related problems.4 Sicca symptoms (severe dry eye or dry mouth) should trigger evaluation for Sjögren syndrome.
Electrocardiography needed
Electrocardiography should be performed to investigate for cardiac conduction abnormalities as well as for resting markers of a supraventricular tachyarrhythmia. Extended ambulatory (Holter) monitoring may be useful to evaluate for a transient reentrant tachyarrhythmia4; however, it does not record body position, so it can be difficult to determine if detected episodes of tachycardia are related to posture.
Additional testing for select cases
Further investigation is usually not needed to diagnose POTS but should be considered in some cases. Advanced tests are typically performed at a tertiary care referral center and include:
- Quantitative sensory testing to evaluate for small-fiber neuropathy (ie, Quantitative Sudomotor Axon Reflex Test, or QSART), which occurs in the neuropathic POTS subtype
- Formal autonomic function testing to characterize neurovascular responsiveness
- Supine and standing plasma norepinephrine levels (fractionated catecholamines) to characterize the net activation of the sympathetic nervous system
- Blood volume assessments to assess hypovolemia
- Formal exercise testing to objectively quantify exercise capacity.
GRADED MANAGEMENT
No single universal gold-standard therapy exists for POTS, and management should be individually determined with the primary goals of treating symptoms and restoring function. A graded approach should be used, starting with conservative nonpharmacologic therapies and adding medications as needed.
While the disease course varies substantially from patient to patient, proper management is strongly associated with eventual symptom improvement.1
NONPHARMACOLOGIC STEPS FIRST
Education
Patients should be informed of the nature of their condition and referred to appropriate healthcare personnel. POTS is a chronic illness requiring individualized coping strategies, intensive physician interaction, and support of a multidisciplinary team. Patients and family members can be reassured that most symptoms improve over time with appropriate diagnosis and treatment.1 Patients should be advised to avoid aggravating triggers and activities.
Exercise
Exercise programs are encouraged but should be introduced gradually, as physical activity can exacerbate symptoms, especially at the outset. Several studies have reported benefits from a short-term (3-month) program, in which the patient gradually progresses from non-upright exercise (eg, rowing machine, recumbent cycle, swimming) to upright endurance exercises. At the end of these programs, significant cardiac remodeling, improved quality of life, and reduced heart rate responses to standing have been reported, and benefits have been reported to persist in patients who continued exercising after the 3-month study period.46,47
Despite the benefits of exercise interventions, compliance is low.46,47 To prevent early discouragement, patients should be advised that it can take 4 to 6 weeks of continued exercise before benefits appear. Patients are encouraged to exercise every other day for 30 minutes or more. Regimens should primarily focus on aerobic conditioning, but resistance training, concentrating on thigh muscles, can also help. Exercise is a treatment and not a cure, and benefits can rapidly disappear if regular activity (at least 3 times per week) is stopped.48
Compression stockings
Compression stockings help reduce peripheral venous pooling and enhance venous return to the heart. Waist-high stockings with compression of at least 30 to 40 mm Hg offer the best results.
Diet
Increased fluid and salt intake is advisable for patients with suspected hypovolemia. At least 2 to 3 L of water accompanied by 10 to 12 g of daily sodium intake is recommended.1 This can usually be accomplished with diet and salt added to food, but salt tablets can be used if the patient prefers. The resultant plasma volume expansion may help reduce the reflex tachycardia upon standing.49
Check medications
Rescue therapy with saline infusion
Intravenous saline infusion can augment blood volume in patients who are clinically decompensated and present with severe symptoms.1 Intermittent infusion of 1 L of normal saline has been found to significantly reduce orthostatic tachycardia and related symptoms in patients with POTS, contributing to improved quality of life.51,52
Chronic saline infusions are not recommended for long-term care because of the risk of access complications and infection.1 Moak et al53 reported a high rate of bacteremia in a cohort of children with POTS with regular saline infusions, most of whom had a central line. On the other hand, Ruzieh et al54 reported significantly improved symptoms with regular saline infusions without a high rate of complications, but patients in this study received infusions for only a few months and through a peripheral intravenous catheter.
DRUG THERAPY
No medications are approved by the US Food and Drug Administration (FDA) or Health Canada specifically for treating POTS, making all pharmacologic recommendations off-label. Although the drugs discussed below have been evaluated for POTS in controlled laboratory settings, they have yet to be tested in robust clinical trials.
Blood volume expansion
Several drugs expand blood volume, which may reduce orthostatic tachycardia.
Fludrocortisone is a synthetic aldosterone analogue that enhances sodium and water retention. Although one observational study found that it normalizes hemodynamic changes in response to orthostatic stress, no high-level evidence exists for its effectiveness for POTS.55 It is generally well tolerated, although possible adverse effects include hyperkalemia, hypertension, fatigue, nausea, headache, and edema.5,56
Desmopressin is a synthetic version of a natural antidiuretic hormone that increases kidney-mediated free-water reabsorption without sodium retention. It significantly reduces upright heart rate in patients with POTS and improves symptom burden. Although potential adverse effects include edema and headache, hyponatremia is the primary concern with daily use, especially with the increased water intake advised for POTS.57 Patients should be advised to use desmopressin no more than once a week for the acute improvement of symptoms. Intermittent monitoring of serum sodium levels is recommended for safety.
Erythropoietin replacement has been suggested for treating POTS to address the significant deficit in red blood cell volume. Although erythropoietin therapy has a direct vasoconstrictive effect and largely improves red blood cell volume in patients with POTS, it does not expand plasma volume, so orthostatic tachycardia is not itself reduced.22 Nevertheless, it may significantly improve POTS symptoms refractory to more common methods of treatment, and it should be reserved for such cases. In addition to the lack of effect on orthostatic tachycardia, drawbacks to using erythropoietin include its high cost, the need for subcutaneous administration, and the risk of life-threatening complications such as myocardial infarction and stroke.58,59
Heart rate-lowering agents
Propranolol, a nonselective beta-adrenergic antagonist, can significantly reduce standing heart rate and improve symptoms at low dosages (10–20 mg). Higher dosages can further restrain orthostatic tachycardia but are not as well tolerated, mainly due to hypotension and worsening of existing symptoms such as fatigue.60 Regular-acting propranolol works for about 4 to 5 hours per dose, so full-day coverage often requires dosing 4 times per day.
Ivabradine is a selective blocker of the “funny” (If) channel that reduces the sinus node firing rate without affecting blood pressure, so it slows heart rate without causing supine hypertension or orthostatic hypotension.
A retrospective case series found that 60% of patients with POTS treated with ivabradine reported symptomatic improvement, and all patients experienced reduced tachycardia with continued use.61 Ivabradine has not been compared with placebo or propranolol in a randomized controlled trial, and it has not been well studied in pregnancy and so should be avoided because of potential teratogenic effects.
When prescribing ivabradine for women of childbearing age, a negative pregnancy test may be documented prior to initiation of therapy, and the use of highly effective methods of contraception is recommended. Ivabradine should be avoided in women contemplating pregnancy. Insurance coverage can limit access to ivabradine in the United States.
Central nervous system sympatholytics
Patients with prominent hyperadrenergic features may benefit from central sympatholytic agents. However, these drugs may not be well tolerated in patients with neuropathic POTS because of the effects of reduced systemic vascular resistance5 and the possible exacerbation of drowsiness, fatigue, and mental clouding.4 Patients can be extremely sensitive to these medications, so they should initially be prescribed at the lowest dose, then gradually increased as tolerated.
Clonidine, an alpha-2-adrenergic agonist, decreases central sympathetic tone. In hyperadrenergic patients, clonidine can stabilize heart rate and blood pressure, thereby reducing orthostatic symptoms.62
Methyldopa has effects similar to those of clonidine but is easier to titrate owing to its longer half-life.63 Methyldopa is typically started at 125 mg at bedtime and increased to 125 mg twice daily, if tolerated.
Other agents
Midodrine is a prodrug. The active form, an alpha-1-adrenergic agonist, constricts peripheral veins and arteries to increase vascular resistance and venous return, thereby reducing orthostatic tachycardia.52 It is most useful in patients with impaired peripheral vasoconstriction (eg, neuropathic POTS) and may be less effective in those with hyperadrenergic POTS.64 Major limitations of midodrine include worsening supine hypertension and possible urinary retention.39
Because of midodrine’s short half-life, frequent dosing is required during daytime hours (eg, 8 AM, noon, and 4 PM), but it should not be taken within 4 to 5 hours of sleep because of the risk of supine hypertension. Midodrine is typically started at 2.5 to 5 mg per dose and can be titrated up to 15 mg per dose.
Midodrine is an FDA pregnancy category C drug (adverse effects in pregnancy seen in animal models, but evidence lacking in humans). While ideally it should be avoided, we have used it safely in pregnant women with disabling POTS symptoms.
Pyridostigmine, an acetylcholinesterase inhibitor, increases cardiovagal tone and possibly sympathetic tone. It has been reported to significantly reduce standing heart rate and improve symptom burden in patients with POTS.65 However, pyridostigmine increases gastrointestinal mobility, leading to severe adverse effects in over 20% of patients, including abdominal cramps, nausea, and diarrhea.66
Droxidopa, a synthetic amino acid precursor of norepinephrine, improves dizziness and fatigue in POTS with minimal effects on blood pressure.67
Modafinil, a psychostimulant, may improve POTS-associated cognitive symptoms.4 It also raises upright blood pressure without significantly worsening standing heart rate or acute orthostatic symptoms.68
EFFECTS OF COMORBID DISORDERS ON MANAGEMENT
Ehlers-Danlos syndrome
Pharmacologic approaches to POTS should not be altered based on the presence of Ehlers-Danlos syndrome, but because many of these patients are prone to joint dislocation, exercise prescriptions may need adjusting.
A medical genetics consult is recommended for patients with Ehlers-Danlos syndrome. Although the hypermobile type (the form most commonly associated with POTS) is not associated with aortopathy, it can be confused with classical and vascular Ehlers-Danlos syndromes, which require serial aortic screening.30
Mast cell activation syndrome
Consultation with an allergist or immunologist may help patients with severe symptoms.
Autoantibodies and autoimmunity
Treatment of the underlying disorder is recommended and can result in significantly improved POTS symptoms.
SPECIALTY CARE REFERRAL
POTS can be challenging to manage. Given the range of physiologic, emotional, and functional distress patients experience, it often requires significant physician time and multidisciplinary care. Patients with continued severe or debilitating symptoms may benefit from referral to a tertiary-care center with experience in autonomic nervous system disorders.
PROGNOSIS
Limited data are available on the long-term prognosis of POTS, and more studies are needed in pediatric and adult populations. No deaths have been reported in the handful of published cases of POTS in patients older than 50.1 Some pediatric studies suggest that some teenagers “outgrow” their POTS. However, these data are not robust, and an alternative explanation is that as they get older, they see adult physicians for their POTS symptoms and so are lost to study follow-up.6,44,69
We have not often seen POTS simply resolve without ongoing treatment. However, in our experience, most patients have improved symptoms and function with multimodal treatment (ie, exercise, salt, water, stockings, and some medications) and time.
Some people, most of them relatively young women, experience lightheadedness, a racing heart, and other symptoms (but not hypotension) when they stand up, in a condition known as postural tachycardia syndrome (POTS).1 Although not known to shorten life,1 it can be physically and mentally debilitating.2,3 Therapy rarely cures it, but a multifaceted approach can substantially improve quality of life.
This review outlines the evaluation and diagnosis of POTS and provides guidance for a therapy regimen.
HOW IS POTS DEFINED?
POTS is a multifactorial syndrome rather than a specific disease. It is characterized by all of the following1,4–6:
- An increase in heart rate of ≥ 30 bpm, or ≥ 40 bpm for those under age 19, within 10 minutes of standing from a supine position
- Sustained tachycardia (> 30 seconds)
- Absence of orthostatic hypotension (a fall in blood pressure of ≥ 20/10 mm Hg)
- Frequent and chronic duration (≥ 6 months).
These features are critical to diagnosis. Hemodynamic criteria in isolation may describe postural tachycardia but are not sufficient to diagnose POTS.
The prevalence of POTS is estimated to be between 0.2% and 1.0%,7 affecting up to 3 million people in the United States. Most cases arise between ages 13 and 50, with a female-to-male ratio of 5:1.8
MANY NAMES, SAME CONDITION
In 1871, Da Costa9 described a condition he called “irritable heart syndrome” that had characteristics similar to those of POTS, including extreme fatigue and exercise intolerance. Decades later, Lewis10 and Wood11 provided more detailed descriptions of the disorder, renaming it “soldier’s heart” or “Da Costa syndrome.” As other cases were documented, more terms arose, including “effort syndrome” and “mitral valve prolapse syndrome.”
In 1982, Rosen and Cryer12 were the first to use the term “postural tachycardia syndrome” for patients with disabling tachycardia upon standing without orthostatic hypotension. In 1986, Fouad et al13 described patients with postural tachycardia, orthostatic intolerance, and a small degree of hypotension as having “idiopathic hypovolemia.”
In 1993, Schondorf and Low14 established the current definition of POTS, leading to increased awareness and research efforts to understand its pathophysiology.
MULTIFACTORIAL PATHOPHYSIOLOGY
During the last 2 decades, several often-overlapping forms of POTS have been recognized, all of which share a final common pathway of sustained orthostatic tachycardia.15–19 In addition, a number of common comorbidities were identified through review of large clinic populations of POTS.20,21
Hypovolemic POTS
Up to 70% of patients with POTS have hypovolemia. The average plasma volume deficit is about 13%, which typically causes only insignificant changes in heart rate and norepinephrine levels while a patient is supine. However, blood pooling associated with upright posture further compromises cardiac output and consequently increases sympathetic nerve activity. Abnormalities in the renin-angiotensin-aldosterone volume regulation system are also suspected to impair sodium retention, contributing to hypovolemia.1,22
Neuropathic POTS
About half of patients with POTS have partial sympathetic denervation (particularly in the lower limbs) and inadequate vasoconstriction upon standing, leading to reduced venous return and stroke volume.17,23 A compensatory increase in sympathetic tone results in tachycardia to maintain cardiac output and blood pressure.
Hyperadrenergic POTS
Up to 50% of patients with POTS have high norepinephrine levels (≥ 600 pg/mL) when upright. This subtype, hyperadrenergic POTS, is characterized by an increase in systolic blood pressure of at least 10 mm Hg within 10 minutes of standing, with concomitant tachycardia that can be similar to or greater than that seen in nonhyperadrenergic POTS. Patients with hyperadrenergic POTS tend to report more prominent symptoms of sympathetic activation, such as palpitations, anxiety, and tremulousness.24,25
Norepinephrine transporter deficiency
The norepinephrine transporter (NET) is on the presynaptic cleft of sympathetic neurons and serves to clear synaptic norepinephrine. NET deficiency leads to a hyperadrenergic state and elevated sympathetic nerve activation.18 NET deficiency may be induced by common antidepressants (eg, tricyclic antidepressants and serotonin-norepinephrine reuptake inhibitors) and attention-deficit disorder medications.4
Mast cell activation syndrome
The relationship between mast cell activation syndrome and POTS is poorly understood.4,26 Mast cell activation syndrome has been described in a subset of patients with POTS who have sinus tachycardia accompanied by severe episodic flushing. Patients with this subtype have a hyperadrenergic response to postural change and elevated urine methylhistamine during flushing episodes.
Patients with mast cell activation syndrome tend to have strong allergic symptoms and may also have severe gastrointestinal problems, food sensitivities, dermatographism, and neuropathy. Diagnosis can be difficult, as the condition is associated with numerous markers with varying sensitivity and specificity.
Autoimmune origin
A significant minority of patients report a viral-like illness before the onset of POTS symptoms, suggesting a possible autoimmune-mediated or inflammatory cause. Also, some autoimmune disorders (eg, Sjögren syndrome) can present with a POTS-like manifestation.
Research into the role of autoantibodies in the pathophysiology of POTS offers the potential to develop novel therapeutic targets. Autoantibodies that have been reported in POTS include those against M1 to M3 muscarinic receptors (present in over 87% of patients with POTS),27 cardiac lipid raft-associated proteins,28 adrenergic G-protein coupled receptors, alpha-1-adrenergic receptors, and beta-1- and beta-2-adrenergic receptors.29 Although commercial enzyme-linked immunosorbent assays can assess for these antibody fragments, it is not known whether targeting the antibodies improves outcomes. At this time, antibody testing for POTS should be confined to the research setting.
LINKS TO OTHER SYNDROMES
POTS is often associated with other conditions whose symptoms cannot be explained by postural intolerance or tachycardia.
Ehlers-Danlos syndromes are a group of inherited heterogeneous disorders involving joint hypermobility, skin hyperextensibility, and tissue fragility.30 The hypermobile subtype is most commonly associated with POTS, with patients often having symptoms of autonomic dysregulation and autonomic test abnormalities.31–33 Patients with POTS may have a history of joint subluxations, joint pain, cervical instability, and spontaneous epidural leaks. The reason for the overlap between the two syndromes is not clear.
Chronic fatigue syndrome is characterized by persistent fatigue that does not resolve with rest and is not necessarily associated with orthostatic changes. More than 75% of patients with POTS report general fatigue as a major complaint, and up to 23% meet the full criteria for chronic fatigue syndrome.34
DIAGNOSTIC STRATEGY
A patient presenting with symptoms suggestive of POTS should first undergo a detailed history and physical examination. Other causes of sinus tachycardia should be considered.
Detailed history, symptom review
The history should focus on determining symptom burden, including tachycardia onset, frequency, severity, and triggers; the presence of syncope; and the impact of symptoms on daily function and quality of life.
Presyncope and its associated symptoms occur in less than one-third of patients with POTS, and syncope is not a principal feature.4 If syncope is the predominant complaint, alternative causes should be investigated. The usual cause of syncope in the general population is thought to be vasovagal.
In addition to orthostatic intolerance, gastrointestinal disturbances are common in POTS, presenting as abdominal pain, heartburn, irregular bowel movements, diarrhea, or constipation. Symptoms of gastroparesis are less common. Gastrointestinal symptoms tend to be prolonged, lasting hours and occurring multiple times a week. They tend not to improve in the supine position.35
POTS-associated symptoms may develop insidiously, but patients often report onset after an acute stressor such as pregnancy, major surgery, or a presumed viral illness.4 Whether these putative triggers are causative or coincidental is unknown. Symptoms of orthostatic intolerance tend to be exacerbated by dehydration, heat, alcohol, exercise, and menstruation.36,37
Consider the family history: 1 in 8 patients with POTS reports familial orthostatic intolerance,38 suggesting a genetic role in some patients. Inquire about symptoms or a previous diagnosis of Ehlers-Danlos syndrome and mast cell activation syndrome.
Consider other conditions
Pheochromocytoma causes hyperadrenergic symptoms (eg, palpitations, lightheadedness) like those in POTS, but patients with pheochromocytoma typically have these symptoms while supine. Pheochromocytoma is also characterized by plasma norepinephrine levels much higher than in POTS.4 Plasma metanephrine testing helps diagnose or rule out pheochromocytoma.5
Inappropriate sinus tachycardia, like pheochromocytoma, also has clinical features similar to those of POTS, as well as tachycardia present when supine. It involves higher sympathetic tone and lower parasympathetic tone compared with POTS; patients commonly have a daytime resting heart rate of at least 100 bpm or a 24-hour mean heart rate of at least 90 bpm.1,42 While the intrinsic heart rate is heightened in inappropriate sinus tachycardia, it is not different between POTS patients and healthy individuals.42,43 Distinguishing POTS from inappropriate sinus tachycardia is further complicated by the broad inclusion criteria of most studies of inappropriate sinus tachycardia, which failed to exclude patients with POTS.44 The Heart Rhythm Society recently adopted distinct definitions for the 2 conditions.1
Physical examination: Focus on vital signs
Dependent acrocyanosis—dark red-blue discoloration of the lower legs that is cold to the touch—occurs in about half of patients with POTS upon standing.4 Dependent acrocyanosis is associated with joint hypermobility and Ehlers-Danlos syndrome, so these conditions should also be considered if findings are positive.
Laboratory testing for other causes
Laboratory testing is used mainly to detect primary causes of sinus tachycardia. Tests should include:
- Complete blood cell count with hematocrit (for severe anemia)
- Thyroid-stimulating hormone level (for hyperthyroidism)
- Electrolyte panel (for significant electrolyte disturbances).
Evidence is insufficient to support routinely measuring the vitamin B12 level, iron indices, and serum markers for celiac disease, although these may be done if the history or physical examination suggests related problems.4 Sicca symptoms (severe dry eye or dry mouth) should trigger evaluation for Sjögren syndrome.
Electrocardiography needed
Electrocardiography should be performed to investigate for cardiac conduction abnormalities as well as for resting markers of a supraventricular tachyarrhythmia. Extended ambulatory (Holter) monitoring may be useful to evaluate for a transient reentrant tachyarrhythmia4; however, it does not record body position, so it can be difficult to determine if detected episodes of tachycardia are related to posture.
Additional testing for select cases
Further investigation is usually not needed to diagnose POTS but should be considered in some cases. Advanced tests are typically performed at a tertiary care referral center and include:
- Quantitative sensory testing to evaluate for small-fiber neuropathy (ie, Quantitative Sudomotor Axon Reflex Test, or QSART), which occurs in the neuropathic POTS subtype
- Formal autonomic function testing to characterize neurovascular responsiveness
- Supine and standing plasma norepinephrine levels (fractionated catecholamines) to characterize the net activation of the sympathetic nervous system
- Blood volume assessments to assess hypovolemia
- Formal exercise testing to objectively quantify exercise capacity.
GRADED MANAGEMENT
No single universal gold-standard therapy exists for POTS, and management should be individually determined with the primary goals of treating symptoms and restoring function. A graded approach should be used, starting with conservative nonpharmacologic therapies and adding medications as needed.
While the disease course varies substantially from patient to patient, proper management is strongly associated with eventual symptom improvement.1
NONPHARMACOLOGIC STEPS FIRST
Education
Patients should be informed of the nature of their condition and referred to appropriate healthcare personnel. POTS is a chronic illness requiring individualized coping strategies, intensive physician interaction, and support of a multidisciplinary team. Patients and family members can be reassured that most symptoms improve over time with appropriate diagnosis and treatment.1 Patients should be advised to avoid aggravating triggers and activities.
Exercise
Exercise programs are encouraged but should be introduced gradually, as physical activity can exacerbate symptoms, especially at the outset. Several studies have reported benefits from a short-term (3-month) program, in which the patient gradually progresses from non-upright exercise (eg, rowing machine, recumbent cycle, swimming) to upright endurance exercises. At the end of these programs, significant cardiac remodeling, improved quality of life, and reduced heart rate responses to standing have been reported, and benefits have been reported to persist in patients who continued exercising after the 3-month study period.46,47
Despite the benefits of exercise interventions, compliance is low.46,47 To prevent early discouragement, patients should be advised that it can take 4 to 6 weeks of continued exercise before benefits appear. Patients are encouraged to exercise every other day for 30 minutes or more. Regimens should primarily focus on aerobic conditioning, but resistance training, concentrating on thigh muscles, can also help. Exercise is a treatment and not a cure, and benefits can rapidly disappear if regular activity (at least 3 times per week) is stopped.48
Compression stockings
Compression stockings help reduce peripheral venous pooling and enhance venous return to the heart. Waist-high stockings with compression of at least 30 to 40 mm Hg offer the best results.
Diet
Increased fluid and salt intake is advisable for patients with suspected hypovolemia. At least 2 to 3 L of water accompanied by 10 to 12 g of daily sodium intake is recommended.1 This can usually be accomplished with diet and salt added to food, but salt tablets can be used if the patient prefers. The resultant plasma volume expansion may help reduce the reflex tachycardia upon standing.49
Check medications
Rescue therapy with saline infusion
Intravenous saline infusion can augment blood volume in patients who are clinically decompensated and present with severe symptoms.1 Intermittent infusion of 1 L of normal saline has been found to significantly reduce orthostatic tachycardia and related symptoms in patients with POTS, contributing to improved quality of life.51,52
Chronic saline infusions are not recommended for long-term care because of the risk of access complications and infection.1 Moak et al53 reported a high rate of bacteremia in a cohort of children with POTS with regular saline infusions, most of whom had a central line. On the other hand, Ruzieh et al54 reported significantly improved symptoms with regular saline infusions without a high rate of complications, but patients in this study received infusions for only a few months and through a peripheral intravenous catheter.
DRUG THERAPY
No medications are approved by the US Food and Drug Administration (FDA) or Health Canada specifically for treating POTS, making all pharmacologic recommendations off-label. Although the drugs discussed below have been evaluated for POTS in controlled laboratory settings, they have yet to be tested in robust clinical trials.
Blood volume expansion
Several drugs expand blood volume, which may reduce orthostatic tachycardia.
Fludrocortisone is a synthetic aldosterone analogue that enhances sodium and water retention. Although one observational study found that it normalizes hemodynamic changes in response to orthostatic stress, no high-level evidence exists for its effectiveness for POTS.55 It is generally well tolerated, although possible adverse effects include hyperkalemia, hypertension, fatigue, nausea, headache, and edema.5,56
Desmopressin is a synthetic version of a natural antidiuretic hormone that increases kidney-mediated free-water reabsorption without sodium retention. It significantly reduces upright heart rate in patients with POTS and improves symptom burden. Although potential adverse effects include edema and headache, hyponatremia is the primary concern with daily use, especially with the increased water intake advised for POTS.57 Patients should be advised to use desmopressin no more than once a week for the acute improvement of symptoms. Intermittent monitoring of serum sodium levels is recommended for safety.
Erythropoietin replacement has been suggested for treating POTS to address the significant deficit in red blood cell volume. Although erythropoietin therapy has a direct vasoconstrictive effect and largely improves red blood cell volume in patients with POTS, it does not expand plasma volume, so orthostatic tachycardia is not itself reduced.22 Nevertheless, it may significantly improve POTS symptoms refractory to more common methods of treatment, and it should be reserved for such cases. In addition to the lack of effect on orthostatic tachycardia, drawbacks to using erythropoietin include its high cost, the need for subcutaneous administration, and the risk of life-threatening complications such as myocardial infarction and stroke.58,59
Heart rate-lowering agents
Propranolol, a nonselective beta-adrenergic antagonist, can significantly reduce standing heart rate and improve symptoms at low dosages (10–20 mg). Higher dosages can further restrain orthostatic tachycardia but are not as well tolerated, mainly due to hypotension and worsening of existing symptoms such as fatigue.60 Regular-acting propranolol works for about 4 to 5 hours per dose, so full-day coverage often requires dosing 4 times per day.
Ivabradine is a selective blocker of the “funny” (If) channel that reduces the sinus node firing rate without affecting blood pressure, so it slows heart rate without causing supine hypertension or orthostatic hypotension.
A retrospective case series found that 60% of patients with POTS treated with ivabradine reported symptomatic improvement, and all patients experienced reduced tachycardia with continued use.61 Ivabradine has not been compared with placebo or propranolol in a randomized controlled trial, and it has not been well studied in pregnancy and so should be avoided because of potential teratogenic effects.
When prescribing ivabradine for women of childbearing age, a negative pregnancy test may be documented prior to initiation of therapy, and the use of highly effective methods of contraception is recommended. Ivabradine should be avoided in women contemplating pregnancy. Insurance coverage can limit access to ivabradine in the United States.
Central nervous system sympatholytics
Patients with prominent hyperadrenergic features may benefit from central sympatholytic agents. However, these drugs may not be well tolerated in patients with neuropathic POTS because of the effects of reduced systemic vascular resistance5 and the possible exacerbation of drowsiness, fatigue, and mental clouding.4 Patients can be extremely sensitive to these medications, so they should initially be prescribed at the lowest dose, then gradually increased as tolerated.
Clonidine, an alpha-2-adrenergic agonist, decreases central sympathetic tone. In hyperadrenergic patients, clonidine can stabilize heart rate and blood pressure, thereby reducing orthostatic symptoms.62
Methyldopa has effects similar to those of clonidine but is easier to titrate owing to its longer half-life.63 Methyldopa is typically started at 125 mg at bedtime and increased to 125 mg twice daily, if tolerated.
Other agents
Midodrine is a prodrug. The active form, an alpha-1-adrenergic agonist, constricts peripheral veins and arteries to increase vascular resistance and venous return, thereby reducing orthostatic tachycardia.52 It is most useful in patients with impaired peripheral vasoconstriction (eg, neuropathic POTS) and may be less effective in those with hyperadrenergic POTS.64 Major limitations of midodrine include worsening supine hypertension and possible urinary retention.39
Because of midodrine’s short half-life, frequent dosing is required during daytime hours (eg, 8 AM, noon, and 4 PM), but it should not be taken within 4 to 5 hours of sleep because of the risk of supine hypertension. Midodrine is typically started at 2.5 to 5 mg per dose and can be titrated up to 15 mg per dose.
Midodrine is an FDA pregnancy category C drug (adverse effects in pregnancy seen in animal models, but evidence lacking in humans). While ideally it should be avoided, we have used it safely in pregnant women with disabling POTS symptoms.
Pyridostigmine, an acetylcholinesterase inhibitor, increases cardiovagal tone and possibly sympathetic tone. It has been reported to significantly reduce standing heart rate and improve symptom burden in patients with POTS.65 However, pyridostigmine increases gastrointestinal mobility, leading to severe adverse effects in over 20% of patients, including abdominal cramps, nausea, and diarrhea.66
Droxidopa, a synthetic amino acid precursor of norepinephrine, improves dizziness and fatigue in POTS with minimal effects on blood pressure.67
Modafinil, a psychostimulant, may improve POTS-associated cognitive symptoms.4 It also raises upright blood pressure without significantly worsening standing heart rate or acute orthostatic symptoms.68
EFFECTS OF COMORBID DISORDERS ON MANAGEMENT
Ehlers-Danlos syndrome
Pharmacologic approaches to POTS should not be altered based on the presence of Ehlers-Danlos syndrome, but because many of these patients are prone to joint dislocation, exercise prescriptions may need adjusting.
A medical genetics consult is recommended for patients with Ehlers-Danlos syndrome. Although the hypermobile type (the form most commonly associated with POTS) is not associated with aortopathy, it can be confused with classical and vascular Ehlers-Danlos syndromes, which require serial aortic screening.30
Mast cell activation syndrome
Consultation with an allergist or immunologist may help patients with severe symptoms.
Autoantibodies and autoimmunity
Treatment of the underlying disorder is recommended and can result in significantly improved POTS symptoms.
SPECIALTY CARE REFERRAL
POTS can be challenging to manage. Given the range of physiologic, emotional, and functional distress patients experience, it often requires significant physician time and multidisciplinary care. Patients with continued severe or debilitating symptoms may benefit from referral to a tertiary-care center with experience in autonomic nervous system disorders.
PROGNOSIS
Limited data are available on the long-term prognosis of POTS, and more studies are needed in pediatric and adult populations. No deaths have been reported in the handful of published cases of POTS in patients older than 50.1 Some pediatric studies suggest that some teenagers “outgrow” their POTS. However, these data are not robust, and an alternative explanation is that as they get older, they see adult physicians for their POTS symptoms and so are lost to study follow-up.6,44,69
We have not often seen POTS simply resolve without ongoing treatment. However, in our experience, most patients have improved symptoms and function with multimodal treatment (ie, exercise, salt, water, stockings, and some medications) and time.
- Sheldon RS, Grubb BP 2nd, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 2015; 12(6):e41–e63. doi:10.1016/j.hrthm.2015.03.029
- Bagai K, Song Y, Ling JF, et al. Sleep disturbances and diminished quality of life in postural tachycardia syndrome. J Clin Sleep Med 2011; 7(2):204–210. pmid:21509337
- Benrud-Larson LM, Dewar MS, Sandroni P, Rummans TA, Haythornthwaite JA, Low PA. Quality of life in patients with postural tachycardia syndrome. Mayo Clin Proc 2002; 77(6):531–537. doi:10.4065/77.6.531
- Raj SR. Postural tachycardia syndrome (POTS). Circulation 2013; 127(23):2336–2342. doi:10.1161/CIRCULATIONAHA.112.144501
- Raj SR. The postural tachycardia syndrome (POTS): pathophysiology, diagnosis & management. Indian Pacing Electrophysiol J 2006; 6(2):84–99. pmid:16943900
- Singer W, Sletten DM, Opfer-Gehrking TL, Brands CK, Fischer PR, Low PA. Postural tachycardia in children and adolescents: what is abnormal? J Pediatr 2012; 160(2):222–226. doi:10.1016/j.jpeds.2011.08.054
- Mar PL, Raj SR. Neuronal and hormonal perturbations in postural tachycardia syndrome. Front Physiol 2014; 5:220. doi:10.3389/fphys.2014.00220
- Garland EM, Raj SR, Black BK, Harris PA, Robertson D. The hemodynamic and neurohumoral phenotype of postural tachycardia syndrome. Neurology 2007; 69(8):790–798. doi:10.1212/01.wnl.0000267663.05398.40
- Da Costa JM. On irritable heart: a clinical study of a form of functional cardiac disorder and its consequences. Am J Med Sci 1871; 61(121):2–52.
- Lewis T. The tolerance of physical exertion, as shown by soldiers suffering from so-called “irritable heart.” Br Med J 1918; 1(2987):363–365. pmid:20768980
- Wood P. Da Costa’s syndrome (or effort syndrome): lecture I. Br Med J 1941; 1(4194):767–772. pmid:20783672
- Rosen SG, Cryer PE. Postural tachycardia syndrome. Reversal of sympathetic hyperresponsiveness and clinical improvement during sodium loading. Am J Med 1982; 72(5):847–850.
- Fouad FM, Tadena-Thome L, Bravo EL, Tarazi RC. Idiopathic hypovolemia. Ann Intern Med 1986; 104(3):298–303. pmid:3511818
- Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology 1993; 43(1):132–137. pmid:8423877
- Vernino S, Low PA, Fealey RD, Stewart JD, Farrugia G, Lennon VA. Autoantibodies to ganglionic acetylcholine receptors in autoimmune autonomic neuropathies. N Engl J Med 2000; 343(12):847–855. doi:10.1056/NEJM200009213431204
- Raj SR, Robertson D. Blood volume perturbations in the postural tachycardia syndrome. Am J Med Sci 2007; 334(1):57–60. doi:10.1097/MAJ.0b013e318063c6c0
- Jacob G, Costa F, Shannon JR, et al. The neuropathic postural tachycardia syndrome. N Engl J Med 2000; 343(14):1008–1014. doi:10.1056/NEJM200010053431404
- Shannon JR, Flattem NL, Jordan J, et al. Orthostatic intolerance and tachycardia associated with norepinephrine-transporter deficiency. N Engl J Med 2000; 342(8):541–549. doi:10.1056/NEJM200002243420803
- Jones PK, Shaw BH, Raj SR. Clinical challenges in the diagnosis and management of postural tachycardia syndrome. Pract Neurol 2016; 16(6):431–438. doi:10.1136/practneurol-2016-001405
- Gunning WT, Karabin BL, Blomquist TM, Grubb BP. Postural orthostatic tachycardia syndrome is associated with platelet storage pool deficiency. Medicine (Baltimore) 2016; 95(37):e4849. doi:10.1097/MD.0000000000004849
- Kanjwal K, Sheikh M, Karabin B, Kanjwal Y, Grubb BP. Neurocardiogenic syncope coexisting with postural orthostatic tachycardia syndrome in patients suffering from orthostatic intolerance: a combined form of autonomic dysfunction. Pacing Clin Electrophysiol 2011; 34(5):549–554. doi:10.1111/j.1540-8159.2010.02994.x
- Raj SR, Biaggioni I, Yamhure PC, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation 2005; 111(13):1574–1582. doi:10.1161/01.CIR.0000160356.97313.5D
- Gibbons CH, Bonyhay I, Benson A, Wang N, Freeman R. Structural and functional small fiber abnormalities in the neuropathic postural tachycardia syndrome. PLoS One 2013; 8(12):e84716. doi:10.1371/journal.pone.0084716
- Low PA, Sandroni P, Joyner M, Shen WK. Postural tachycardia syndrome (POTS). J Cardiovasc Electrophysiol 2009; 20(3):352–358. doi:10.1111/j.1540-8167.2008.01407.x
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Grubb BP. Clinical presentation and management of patients with hyperadrenergic postural orthostatic tachycardia syndrome. A single center experience. Cardiol J 2011; 18(5):527–531. pmid:21947988
- Shibao C, Arzubiaga C, Roberts J, et al. Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension 2005; 45(3):385–390. doi:10.1161/01.HYP.0000158259.68614.40
- Dubey D, Hopkins S, Vernino S. M1 and M2 muscarinic receptor antibodies among patients with postural orthostatic tachycardia syndrome: potential disease biomarker [abstract]. J Clin Neuromuscul Dis 2016; 17(3):179S.
- Wang XL, Ling TY, Charlesworth MC, et al. Autoimmunoreactive IgGs against cardiac lipid raft-associated proteins in patients with postural orthostatic tachycardia syndrome. Transl Res 2013; 162(1):34–44. doi:10.1016/j.trsl.2013.03.002
- Li H, Yu X, Liles C, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc 2014; 3(1):e000755. doi:10.1161/JAHA.113.000755
- Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet 2017; 175(1):8–26. doi:10.1002/ajmg.c.31552
- Wallman D, Weinberg J, Hohler AD. Ehlers-Danlos syndrome and postural tachycardia syndrome: a relationship study. J Neurol Sci 2014; 340(1-2):99–102. doi:10.1016/j.jns.2014.03.002
- De Wandele I, Calders P, Peersman W, et al. Autonomic symptom burden in the hypermobility type of Ehlers-Danlos syndrome: a comparative study with two other EDS types, fibromyalgia, and healthy controls. Semin Arthritis Rheum 2014; 44(3):353–361. doi:10.1016/j.semarthrit.2014.05.013
- Gazit Y, Nahir AM, Grahame R, Jacob G. Dysautonomia in the joint hypermobility syndrome. Am J Med 2003; 115(1):33–40. pmid:12867232
- Okamoto LE, Raj SR, Peltier A, et al. Neurohumoral and haemodynamic profile in postural tachycardia and chronic fatigue syndromes. Clin Sci (Lond) 2012; 122(4):183–192. doi:10.1042/CS20110200
- Wang LB, Culbertson CJ, Deb A, Morgenshtern K, Huang H, Hohler AD. Gastrointestinal dysfunction in postural tachycardia syndrome. J Neurol Sci 2015; 359(1-2):193–196. doi:10.1016/j.jns.2015.10.052
- Raj S, Sheldon R. Management of postural tachycardia syndrome, inappropriate sinus tachycardia and vasovagal syncope. Arrhythm Electrophysiol Rev 2016; 5(2):122–129. doi:10.15420/AER.2016.7.2
- Peggs KJ, Nguyen H, Enayat D, Keller NR, Al-Hendy A, Raj SR. Gynecologic disorders and menstrual cycle lightheadedness in postural tachycardia syndrome. Int J Gynaecol Obstet 2012; 118(3):242–246. doi:10.1016/j.ijgo.2012.04.014
- Thieben MJ, Sandroni P, Sletten DM, et al. Postural orthostatic tachycardia syndrome: the Mayo Clinic experience. Mayo Clin Proc 2007; 82(3):308–313. doi:10.4065/82.3.308
- Deb A, Morgenshtern K, Culbertson CJ, Wang LB, Hohler AD. A survey-based analysis of symptoms in patients with postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2015; 28(7):157–159. pmid:25829642
- Ertek S, Cicero AF. Hyperthyroidism and cardiovascular complications: a narrative review on the basis of pathophysiology. Arch Med Sci 2013; 9(5):944–952. doi:10.5114/aoms.2013.38685
- Rangno RE, Langlois S. Comparison of withdrawal phenomena after propranolol, metoprolol and pindolol. Br J Clin Pharmacol 1982; 13(suppl 2):345S–351S. pmid:6125187
- Nwazue VC, Paranjape SY, Black BK, et al. Postural tachycardia syndrome and inappropriate sinus tachycardia: role of autonomic modulation and sinus node automaticity. J Am Heart Assoc 2014; 3(2):e000700. doi:10.1161/JAHA.113.000700
- Morillo CA, Klein GJ, Thakur RK, Li H, Zardini M, Yee R. Mechanism of “inappropriate” sinus tachycardia. Role of sympathovagal balance. Circulation 1994; 90(2):873–877. pmid:7913886
- Grubb BP. Postural tachycardia syndrome. Circulation 2008; 117(21):2814–2817. doi:10.1161/CIRCULATIONAHA.107.761643
- Bhatia R, Kizilbash SJ, Ahrens SP, et al. Outcomes of adolescent-onset postural orthostatic tachycardia syndrome. J Pediatr 2016; 173:149–153. doi:10.1016/j.jpeds.2016.02.035
- George SA, Bivens TB, Howden EJ, et al. The international POTS registry: evaluating the efficacy of an exercise training intervention in a community setting. Heart Rhythm 2016; 13(4):943–950. doi:10.1016/j.hrthm.2015.12.012
- Fu Q, VanGundy TB, Galbreath MM, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2010; 55(25):2858–2868. doi:10.1016/j.jacc.2010.02.043
- Raj SR. Row, row, row your way to treating postural tachycardia syndrome. Heart Rhythm 2016; 13(4):951–952. doi:10.1016/j.hrthm.2015.12.039
- Celedonio JE, Garland EM, Nwazue VC, et al. Effects of high sodium intake on blood volume and catecholamines in patients with postural tachycardia syndrome and healthy females [abstract]. Clin Auton Res 2014; 24:211.
- Garland EM, Celedonio JE, Raj SR. Postural tachycardia syndrome: beyond orthostatic intolerance. Curr Neurol Neurosci Rep 2015; 15(9):60. doi:10.1007/s11910-015-0583-8
- Gordon VM, Opfer-Gehrking TL, Novak V, Low PA. Hemodynamic and symptomatic effects of acute interventions on tilt in patients with postural tachycardia syndrome. Clin Auton Res 2000; 10:29–33. pmid:10750641
- Jacob G, Shannon JR, Black B, et al. Effects of volume loading and pressor agents in idiopathic orthostatic tachycardia. Circulation 1997; 96(2):575–580. pmid:9244228
- Moak JP, Leong D, Fabian R, et al. Intravenous hydration for management of medication-resistant orthostatic intolerance in the adolescent and young adult. Pediatr Cardiol 2016; 37(2):278–282. doi:10.1007/s00246-015-1274-6
- Ruzieh M, Baugh A, Dasa O, et al. Effects of intermittent intravenous saline infusions in patients with medication-refractory postural tachycardia syndrome. J Interv Card Electrophysiol 2017; 48(3):255–260. doi:10.1007/s10840-017-0225-y
- Freitas J, Santos R, Azevedo E, Costa O, Carvalho M, de Freitas AF. Clinical improvement in patients with orthostatic intolerance after treatment with bisoprolol and fludrocortisone. Clin Auton Res 2000; 10(5):293–299. pmid:11198485
- Lee AK, Krahn AD. Evaluation of syncope: focus on diagnosis and treatment of neurally mediated syncope. Expert Rev Cardiovasc Ther 2016; 14(6):725–736. doi:10.1586/14779072.2016.1164034
- Coffin ST, Black BK, Biaggioni I, et al. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm 2012; 9(9):1484–1490. doi:10.1016/j.hrthm.2012.05.002
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Sheikh M, Grubb BP. Erythropoietin in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2012; 19(2):92–95. doi:10.1097/MJT.0b013e3181ef621a
- Hoeldtke RD, Horvath GG, Bryner KD. Treatment of orthostatic tachycardia with erythropoietin. Am J Med 1995; 99(5):525–529. pmid:7485211
- Raj SR, Black BK, Biaggioni I, et al. Propranolol decreases tachycardia and improves symptoms in the postural tachycardia syndrome: less is more. Circulation 2009; 120(9):725–734. doi:10.1161/CIRCULATIONAHA.108.846501
- McDonald C, Frith J, Newton JL. Single centre experience of ivabradine in postural orthostatic tachycardia syndrome. Europace 2011; 13(3):427–430. doi:10.1093/europace/euq390
- Gaffney FA, Lane LB, Pettinger W, Blomqvist G. Effects of long-term clonidine administration on the hemodynamic and neuroendocrine postural responses of patients with dysautonomia. Chest 1983; 83(suppl 2):436–438. pmid:6295714
- Jacob G, Biaggioni I. Idiopathic orthostatic intolerance and postural tachycardia syndromes. Am J Med Sci 1999; 317(2):88–101. pmid:10037112
- Ross AJ, Ocon AJ, Medow MS, Stewart JM. A double-blind placebo-controlled cross-over study of the vascular effects of midodrine in neuropathic compared with hyperadrenergic postural tachycardia syndrome. Clin Sci (Lond) 2014; 126(4):289–296. doi:10.1042/CS20130222
- Raj SR, Black BK, Biaggioni I, Harris PA, Robertson D. Acetylcholinesterase inhibition improves tachycardia in postural tachycardia syndrome. Circulation 2005; 111(21):2734–2340. doi:10.1161/CIRCULATIONAHA.104.497594
- Kanjwal K, Karabin B, Sheikh M, et al. Pyridostigmine in the treatment of postural orthostatic tachycardia: A single-center experience. Pacing Clin Electrophysiol 2011; 34(6):750–755. doi:10.1111/j.1540-8159.2011.03047.x
- Ruzieh M, Dasa O, Pacenta A, Karabin B, Grubb B. Droxidopa in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2017; 24(2):e157–e161. doi:10.1097/MJT.0000000000000468
- Kpaeyeh AG Jr, Mar PL, Raj V, et al. Hemodynamic profiles and tolerability of modafinil in the treatment of POTS: a randomized placebo-controlled trial. J Clin Psychopharmacol 2014; 34(6):738–741. doi:10.1097/JCP.0000000000000221
- Lai CC, Fischer PR, Brands CK, et al. Outcomes in adolescents with postural orthostatic tachycardia syndrome treated with midodrine and beta-blockers. Pacing Clin Electrophysiol 2009; 32(2):234–238. doi:10.1111/j.1540-8159.2008.02207.x
- Sheldon RS, Grubb BP 2nd, Olshansky B, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 2015; 12(6):e41–e63. doi:10.1016/j.hrthm.2015.03.029
- Bagai K, Song Y, Ling JF, et al. Sleep disturbances and diminished quality of life in postural tachycardia syndrome. J Clin Sleep Med 2011; 7(2):204–210. pmid:21509337
- Benrud-Larson LM, Dewar MS, Sandroni P, Rummans TA, Haythornthwaite JA, Low PA. Quality of life in patients with postural tachycardia syndrome. Mayo Clin Proc 2002; 77(6):531–537. doi:10.4065/77.6.531
- Raj SR. Postural tachycardia syndrome (POTS). Circulation 2013; 127(23):2336–2342. doi:10.1161/CIRCULATIONAHA.112.144501
- Raj SR. The postural tachycardia syndrome (POTS): pathophysiology, diagnosis & management. Indian Pacing Electrophysiol J 2006; 6(2):84–99. pmid:16943900
- Singer W, Sletten DM, Opfer-Gehrking TL, Brands CK, Fischer PR, Low PA. Postural tachycardia in children and adolescents: what is abnormal? J Pediatr 2012; 160(2):222–226. doi:10.1016/j.jpeds.2011.08.054
- Mar PL, Raj SR. Neuronal and hormonal perturbations in postural tachycardia syndrome. Front Physiol 2014; 5:220. doi:10.3389/fphys.2014.00220
- Garland EM, Raj SR, Black BK, Harris PA, Robertson D. The hemodynamic and neurohumoral phenotype of postural tachycardia syndrome. Neurology 2007; 69(8):790–798. doi:10.1212/01.wnl.0000267663.05398.40
- Da Costa JM. On irritable heart: a clinical study of a form of functional cardiac disorder and its consequences. Am J Med Sci 1871; 61(121):2–52.
- Lewis T. The tolerance of physical exertion, as shown by soldiers suffering from so-called “irritable heart.” Br Med J 1918; 1(2987):363–365. pmid:20768980
- Wood P. Da Costa’s syndrome (or effort syndrome): lecture I. Br Med J 1941; 1(4194):767–772. pmid:20783672
- Rosen SG, Cryer PE. Postural tachycardia syndrome. Reversal of sympathetic hyperresponsiveness and clinical improvement during sodium loading. Am J Med 1982; 72(5):847–850.
- Fouad FM, Tadena-Thome L, Bravo EL, Tarazi RC. Idiopathic hypovolemia. Ann Intern Med 1986; 104(3):298–303. pmid:3511818
- Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology 1993; 43(1):132–137. pmid:8423877
- Vernino S, Low PA, Fealey RD, Stewart JD, Farrugia G, Lennon VA. Autoantibodies to ganglionic acetylcholine receptors in autoimmune autonomic neuropathies. N Engl J Med 2000; 343(12):847–855. doi:10.1056/NEJM200009213431204
- Raj SR, Robertson D. Blood volume perturbations in the postural tachycardia syndrome. Am J Med Sci 2007; 334(1):57–60. doi:10.1097/MAJ.0b013e318063c6c0
- Jacob G, Costa F, Shannon JR, et al. The neuropathic postural tachycardia syndrome. N Engl J Med 2000; 343(14):1008–1014. doi:10.1056/NEJM200010053431404
- Shannon JR, Flattem NL, Jordan J, et al. Orthostatic intolerance and tachycardia associated with norepinephrine-transporter deficiency. N Engl J Med 2000; 342(8):541–549. doi:10.1056/NEJM200002243420803
- Jones PK, Shaw BH, Raj SR. Clinical challenges in the diagnosis and management of postural tachycardia syndrome. Pract Neurol 2016; 16(6):431–438. doi:10.1136/practneurol-2016-001405
- Gunning WT, Karabin BL, Blomquist TM, Grubb BP. Postural orthostatic tachycardia syndrome is associated with platelet storage pool deficiency. Medicine (Baltimore) 2016; 95(37):e4849. doi:10.1097/MD.0000000000004849
- Kanjwal K, Sheikh M, Karabin B, Kanjwal Y, Grubb BP. Neurocardiogenic syncope coexisting with postural orthostatic tachycardia syndrome in patients suffering from orthostatic intolerance: a combined form of autonomic dysfunction. Pacing Clin Electrophysiol 2011; 34(5):549–554. doi:10.1111/j.1540-8159.2010.02994.x
- Raj SR, Biaggioni I, Yamhure PC, et al. Renin-aldosterone paradox and perturbed blood volume regulation underlying postural tachycardia syndrome. Circulation 2005; 111(13):1574–1582. doi:10.1161/01.CIR.0000160356.97313.5D
- Gibbons CH, Bonyhay I, Benson A, Wang N, Freeman R. Structural and functional small fiber abnormalities in the neuropathic postural tachycardia syndrome. PLoS One 2013; 8(12):e84716. doi:10.1371/journal.pone.0084716
- Low PA, Sandroni P, Joyner M, Shen WK. Postural tachycardia syndrome (POTS). J Cardiovasc Electrophysiol 2009; 20(3):352–358. doi:10.1111/j.1540-8167.2008.01407.x
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Grubb BP. Clinical presentation and management of patients with hyperadrenergic postural orthostatic tachycardia syndrome. A single center experience. Cardiol J 2011; 18(5):527–531. pmid:21947988
- Shibao C, Arzubiaga C, Roberts J, et al. Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension 2005; 45(3):385–390. doi:10.1161/01.HYP.0000158259.68614.40
- Dubey D, Hopkins S, Vernino S. M1 and M2 muscarinic receptor antibodies among patients with postural orthostatic tachycardia syndrome: potential disease biomarker [abstract]. J Clin Neuromuscul Dis 2016; 17(3):179S.
- Wang XL, Ling TY, Charlesworth MC, et al. Autoimmunoreactive IgGs against cardiac lipid raft-associated proteins in patients with postural orthostatic tachycardia syndrome. Transl Res 2013; 162(1):34–44. doi:10.1016/j.trsl.2013.03.002
- Li H, Yu X, Liles C, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc 2014; 3(1):e000755. doi:10.1161/JAHA.113.000755
- Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet 2017; 175(1):8–26. doi:10.1002/ajmg.c.31552
- Wallman D, Weinberg J, Hohler AD. Ehlers-Danlos syndrome and postural tachycardia syndrome: a relationship study. J Neurol Sci 2014; 340(1-2):99–102. doi:10.1016/j.jns.2014.03.002
- De Wandele I, Calders P, Peersman W, et al. Autonomic symptom burden in the hypermobility type of Ehlers-Danlos syndrome: a comparative study with two other EDS types, fibromyalgia, and healthy controls. Semin Arthritis Rheum 2014; 44(3):353–361. doi:10.1016/j.semarthrit.2014.05.013
- Gazit Y, Nahir AM, Grahame R, Jacob G. Dysautonomia in the joint hypermobility syndrome. Am J Med 2003; 115(1):33–40. pmid:12867232
- Okamoto LE, Raj SR, Peltier A, et al. Neurohumoral and haemodynamic profile in postural tachycardia and chronic fatigue syndromes. Clin Sci (Lond) 2012; 122(4):183–192. doi:10.1042/CS20110200
- Wang LB, Culbertson CJ, Deb A, Morgenshtern K, Huang H, Hohler AD. Gastrointestinal dysfunction in postural tachycardia syndrome. J Neurol Sci 2015; 359(1-2):193–196. doi:10.1016/j.jns.2015.10.052
- Raj S, Sheldon R. Management of postural tachycardia syndrome, inappropriate sinus tachycardia and vasovagal syncope. Arrhythm Electrophysiol Rev 2016; 5(2):122–129. doi:10.15420/AER.2016.7.2
- Peggs KJ, Nguyen H, Enayat D, Keller NR, Al-Hendy A, Raj SR. Gynecologic disorders and menstrual cycle lightheadedness in postural tachycardia syndrome. Int J Gynaecol Obstet 2012; 118(3):242–246. doi:10.1016/j.ijgo.2012.04.014
- Thieben MJ, Sandroni P, Sletten DM, et al. Postural orthostatic tachycardia syndrome: the Mayo Clinic experience. Mayo Clin Proc 2007; 82(3):308–313. doi:10.4065/82.3.308
- Deb A, Morgenshtern K, Culbertson CJ, Wang LB, Hohler AD. A survey-based analysis of symptoms in patients with postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2015; 28(7):157–159. pmid:25829642
- Ertek S, Cicero AF. Hyperthyroidism and cardiovascular complications: a narrative review on the basis of pathophysiology. Arch Med Sci 2013; 9(5):944–952. doi:10.5114/aoms.2013.38685
- Rangno RE, Langlois S. Comparison of withdrawal phenomena after propranolol, metoprolol and pindolol. Br J Clin Pharmacol 1982; 13(suppl 2):345S–351S. pmid:6125187
- Nwazue VC, Paranjape SY, Black BK, et al. Postural tachycardia syndrome and inappropriate sinus tachycardia: role of autonomic modulation and sinus node automaticity. J Am Heart Assoc 2014; 3(2):e000700. doi:10.1161/JAHA.113.000700
- Morillo CA, Klein GJ, Thakur RK, Li H, Zardini M, Yee R. Mechanism of “inappropriate” sinus tachycardia. Role of sympathovagal balance. Circulation 1994; 90(2):873–877. pmid:7913886
- Grubb BP. Postural tachycardia syndrome. Circulation 2008; 117(21):2814–2817. doi:10.1161/CIRCULATIONAHA.107.761643
- Bhatia R, Kizilbash SJ, Ahrens SP, et al. Outcomes of adolescent-onset postural orthostatic tachycardia syndrome. J Pediatr 2016; 173:149–153. doi:10.1016/j.jpeds.2016.02.035
- George SA, Bivens TB, Howden EJ, et al. The international POTS registry: evaluating the efficacy of an exercise training intervention in a community setting. Heart Rhythm 2016; 13(4):943–950. doi:10.1016/j.hrthm.2015.12.012
- Fu Q, VanGundy TB, Galbreath MM, et al. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol 2010; 55(25):2858–2868. doi:10.1016/j.jacc.2010.02.043
- Raj SR. Row, row, row your way to treating postural tachycardia syndrome. Heart Rhythm 2016; 13(4):951–952. doi:10.1016/j.hrthm.2015.12.039
- Celedonio JE, Garland EM, Nwazue VC, et al. Effects of high sodium intake on blood volume and catecholamines in patients with postural tachycardia syndrome and healthy females [abstract]. Clin Auton Res 2014; 24:211.
- Garland EM, Celedonio JE, Raj SR. Postural tachycardia syndrome: beyond orthostatic intolerance. Curr Neurol Neurosci Rep 2015; 15(9):60. doi:10.1007/s11910-015-0583-8
- Gordon VM, Opfer-Gehrking TL, Novak V, Low PA. Hemodynamic and symptomatic effects of acute interventions on tilt in patients with postural tachycardia syndrome. Clin Auton Res 2000; 10:29–33. pmid:10750641
- Jacob G, Shannon JR, Black B, et al. Effects of volume loading and pressor agents in idiopathic orthostatic tachycardia. Circulation 1997; 96(2):575–580. pmid:9244228
- Moak JP, Leong D, Fabian R, et al. Intravenous hydration for management of medication-resistant orthostatic intolerance in the adolescent and young adult. Pediatr Cardiol 2016; 37(2):278–282. doi:10.1007/s00246-015-1274-6
- Ruzieh M, Baugh A, Dasa O, et al. Effects of intermittent intravenous saline infusions in patients with medication-refractory postural tachycardia syndrome. J Interv Card Electrophysiol 2017; 48(3):255–260. doi:10.1007/s10840-017-0225-y
- Freitas J, Santos R, Azevedo E, Costa O, Carvalho M, de Freitas AF. Clinical improvement in patients with orthostatic intolerance after treatment with bisoprolol and fludrocortisone. Clin Auton Res 2000; 10(5):293–299. pmid:11198485
- Lee AK, Krahn AD. Evaluation of syncope: focus on diagnosis and treatment of neurally mediated syncope. Expert Rev Cardiovasc Ther 2016; 14(6):725–736. doi:10.1586/14779072.2016.1164034
- Coffin ST, Black BK, Biaggioni I, et al. Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachycardia syndrome. Heart Rhythm 2012; 9(9):1484–1490. doi:10.1016/j.hrthm.2012.05.002
- Kanjwal K, Saeed B, Karabin B, Kanjwal Y, Sheikh M, Grubb BP. Erythropoietin in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2012; 19(2):92–95. doi:10.1097/MJT.0b013e3181ef621a
- Hoeldtke RD, Horvath GG, Bryner KD. Treatment of orthostatic tachycardia with erythropoietin. Am J Med 1995; 99(5):525–529. pmid:7485211
- Raj SR, Black BK, Biaggioni I, et al. Propranolol decreases tachycardia and improves symptoms in the postural tachycardia syndrome: less is more. Circulation 2009; 120(9):725–734. doi:10.1161/CIRCULATIONAHA.108.846501
- McDonald C, Frith J, Newton JL. Single centre experience of ivabradine in postural orthostatic tachycardia syndrome. Europace 2011; 13(3):427–430. doi:10.1093/europace/euq390
- Gaffney FA, Lane LB, Pettinger W, Blomqvist G. Effects of long-term clonidine administration on the hemodynamic and neuroendocrine postural responses of patients with dysautonomia. Chest 1983; 83(suppl 2):436–438. pmid:6295714
- Jacob G, Biaggioni I. Idiopathic orthostatic intolerance and postural tachycardia syndromes. Am J Med Sci 1999; 317(2):88–101. pmid:10037112
- Ross AJ, Ocon AJ, Medow MS, Stewart JM. A double-blind placebo-controlled cross-over study of the vascular effects of midodrine in neuropathic compared with hyperadrenergic postural tachycardia syndrome. Clin Sci (Lond) 2014; 126(4):289–296. doi:10.1042/CS20130222
- Raj SR, Black BK, Biaggioni I, Harris PA, Robertson D. Acetylcholinesterase inhibition improves tachycardia in postural tachycardia syndrome. Circulation 2005; 111(21):2734–2340. doi:10.1161/CIRCULATIONAHA.104.497594
- Kanjwal K, Karabin B, Sheikh M, et al. Pyridostigmine in the treatment of postural orthostatic tachycardia: A single-center experience. Pacing Clin Electrophysiol 2011; 34(6):750–755. doi:10.1111/j.1540-8159.2011.03047.x
- Ruzieh M, Dasa O, Pacenta A, Karabin B, Grubb B. Droxidopa in the treatment of postural orthostatic tachycardia syndrome. Am J Ther 2017; 24(2):e157–e161. doi:10.1097/MJT.0000000000000468
- Kpaeyeh AG Jr, Mar PL, Raj V, et al. Hemodynamic profiles and tolerability of modafinil in the treatment of POTS: a randomized placebo-controlled trial. J Clin Psychopharmacol 2014; 34(6):738–741. doi:10.1097/JCP.0000000000000221
- Lai CC, Fischer PR, Brands CK, et al. Outcomes in adolescents with postural orthostatic tachycardia syndrome treated with midodrine and beta-blockers. Pacing Clin Electrophysiol 2009; 32(2):234–238. doi:10.1111/j.1540-8159.2008.02207.x
KEY POINTS
- Several POTS subtypes have been recognized, including hypovolemic, neuropathic, and hyperadrenergic forms, overlapping with Ehlers-Danlos syndrome, mast cell activation, and autoimmune syndromes.
- Treatment should take a graded approach, beginning with increasing salt and water intake, exercise, and compression stockings.
- If needed, consider medications to expand blood volume, slow heart rate, or reduce central sympathetic tone.
- Certain medications, including venodilators, diuretics, and serotonin-norepinephrine reuptake inhibitors, can exacerbate symptoms and should be avoided.
Dabigatran-induced esophagitis
A 74-year-old man presented to the gastroenterology clinic with a 2-day history of retrosternal discomfort. His vital signs were normal, and laboratory testing showed a normal leukocyte count.
Esophagogastroduodenoscopy (EGD) revealed longitudinal sloughing mucosal casts in the middle and lower esophagus (Figure 1).
The patient had been taking dabigatran 110 mg twice daily for 2 years because of nonvalvular atrial fibrillation. He was also taking amlodipine 2.5 mg/day for hypertension.
DABIGATRAN-INDUCED ESOPHAGITIS
Although no study has investigated the overall prevalence of dabigatran-induced esophagitis, a retrospective database review of 91 patients taking dabigatran and undergoing upper-gastrointestinal endoscopy reported that 19 (20.9%) had endoscopic signs of dabigatran-induced esophagitis.2
Typical symptoms are the acute onset of chest pain, epigastralgia, odynophagia, and dysphagia. But patients can also have no symptoms or only mild symptoms.2,3
Despite dabigatran’s anticoagulant activity, there have been few reports of bleeding, perhaps because the lesions tend to be superficial on the surface of the esophageal mucosa.
Symptoms usually resolve within 1 week after stopping dabigatran and starting a proton pump inhibitor. To prevent mucosal injury, patients should be instructed to take dabigatran with sufficient water and to remain in an upright position for at least 30 minutes afterward.4
- Baehr PH, McDonald GB. Esophageal infections: risk factors, presentation, diagnosis, and treatment. Gastroenterology 1994; 106(2):509–532. pmid:7980741
- Toya Y, Nakamura S, Tomita K, et al. Dabigatran-induced esophagitis: the prevalence and endoscopic characteristics. J Gastroenterol Hepatol 2016; 31(3):610–614. doi:10.1111/jgh.13024
- Ueta E, Fujikawa T, Imagawa A. A case of a slightly symptomatic exfoliative oesophagitis. BMJ Case Rep 2015; pii:bcr2015211925. doi:10.1136/bcr-2015-211925
- Ootani A, Hayashi Y, Miyagi Y. Dabigatran-induced esophagitis. Clin Gastroenterol Hepatol 2014; 12(7):e55–e56. doi:10.1016/j.cgh.2013.09.010
A 74-year-old man presented to the gastroenterology clinic with a 2-day history of retrosternal discomfort. His vital signs were normal, and laboratory testing showed a normal leukocyte count.
Esophagogastroduodenoscopy (EGD) revealed longitudinal sloughing mucosal casts in the middle and lower esophagus (Figure 1).
The patient had been taking dabigatran 110 mg twice daily for 2 years because of nonvalvular atrial fibrillation. He was also taking amlodipine 2.5 mg/day for hypertension.
DABIGATRAN-INDUCED ESOPHAGITIS
Although no study has investigated the overall prevalence of dabigatran-induced esophagitis, a retrospective database review of 91 patients taking dabigatran and undergoing upper-gastrointestinal endoscopy reported that 19 (20.9%) had endoscopic signs of dabigatran-induced esophagitis.2
Typical symptoms are the acute onset of chest pain, epigastralgia, odynophagia, and dysphagia. But patients can also have no symptoms or only mild symptoms.2,3
Despite dabigatran’s anticoagulant activity, there have been few reports of bleeding, perhaps because the lesions tend to be superficial on the surface of the esophageal mucosa.
Symptoms usually resolve within 1 week after stopping dabigatran and starting a proton pump inhibitor. To prevent mucosal injury, patients should be instructed to take dabigatran with sufficient water and to remain in an upright position for at least 30 minutes afterward.4
A 74-year-old man presented to the gastroenterology clinic with a 2-day history of retrosternal discomfort. His vital signs were normal, and laboratory testing showed a normal leukocyte count.
Esophagogastroduodenoscopy (EGD) revealed longitudinal sloughing mucosal casts in the middle and lower esophagus (Figure 1).
The patient had been taking dabigatran 110 mg twice daily for 2 years because of nonvalvular atrial fibrillation. He was also taking amlodipine 2.5 mg/day for hypertension.
DABIGATRAN-INDUCED ESOPHAGITIS
Although no study has investigated the overall prevalence of dabigatran-induced esophagitis, a retrospective database review of 91 patients taking dabigatran and undergoing upper-gastrointestinal endoscopy reported that 19 (20.9%) had endoscopic signs of dabigatran-induced esophagitis.2
Typical symptoms are the acute onset of chest pain, epigastralgia, odynophagia, and dysphagia. But patients can also have no symptoms or only mild symptoms.2,3
Despite dabigatran’s anticoagulant activity, there have been few reports of bleeding, perhaps because the lesions tend to be superficial on the surface of the esophageal mucosa.
Symptoms usually resolve within 1 week after stopping dabigatran and starting a proton pump inhibitor. To prevent mucosal injury, patients should be instructed to take dabigatran with sufficient water and to remain in an upright position for at least 30 minutes afterward.4
- Baehr PH, McDonald GB. Esophageal infections: risk factors, presentation, diagnosis, and treatment. Gastroenterology 1994; 106(2):509–532. pmid:7980741
- Toya Y, Nakamura S, Tomita K, et al. Dabigatran-induced esophagitis: the prevalence and endoscopic characteristics. J Gastroenterol Hepatol 2016; 31(3):610–614. doi:10.1111/jgh.13024
- Ueta E, Fujikawa T, Imagawa A. A case of a slightly symptomatic exfoliative oesophagitis. BMJ Case Rep 2015; pii:bcr2015211925. doi:10.1136/bcr-2015-211925
- Ootani A, Hayashi Y, Miyagi Y. Dabigatran-induced esophagitis. Clin Gastroenterol Hepatol 2014; 12(7):e55–e56. doi:10.1016/j.cgh.2013.09.010
- Baehr PH, McDonald GB. Esophageal infections: risk factors, presentation, diagnosis, and treatment. Gastroenterology 1994; 106(2):509–532. pmid:7980741
- Toya Y, Nakamura S, Tomita K, et al. Dabigatran-induced esophagitis: the prevalence and endoscopic characteristics. J Gastroenterol Hepatol 2016; 31(3):610–614. doi:10.1111/jgh.13024
- Ueta E, Fujikawa T, Imagawa A. A case of a slightly symptomatic exfoliative oesophagitis. BMJ Case Rep 2015; pii:bcr2015211925. doi:10.1136/bcr-2015-211925
- Ootani A, Hayashi Y, Miyagi Y. Dabigatran-induced esophagitis. Clin Gastroenterol Hepatol 2014; 12(7):e55–e56. doi:10.1016/j.cgh.2013.09.010
Click for Credit: Migraine & stroke risk; Aspirin for CV events; more
Here are 5 articles from the May issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Subclinical hypothyroidism boosts immediate risk of heart failure
To take the posttest, go to: https://bit.ly/2IK0YiL
Expires January 24, 2020
2. Meta-analysis supports aspirin to reduce cardiovascular events
To take the posttest, go to: https://bit.ly/2GJLgSB
Expires January 24, 2020
3. Age of migraine onset may affect stroke risk
To take the posttest, go to: https://bit.ly/2ZAJ5YR
Expires January 24, 2020
4. Women with RA have reduced chance of live birth after assisted reproduction treatment
To take the posttest, go to: https://bit.ly/2VvKRLF
Expires January 27, 2020
5. New SLE disease activity measure beats SLEDAI-2K
To take the posttest, go to: https://bit.ly/2W8SVPA
Expires January 31, 2020
Here are 5 articles from the May issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Subclinical hypothyroidism boosts immediate risk of heart failure
To take the posttest, go to: https://bit.ly/2IK0YiL
Expires January 24, 2020
2. Meta-analysis supports aspirin to reduce cardiovascular events
To take the posttest, go to: https://bit.ly/2GJLgSB
Expires January 24, 2020
3. Age of migraine onset may affect stroke risk
To take the posttest, go to: https://bit.ly/2ZAJ5YR
Expires January 24, 2020
4. Women with RA have reduced chance of live birth after assisted reproduction treatment
To take the posttest, go to: https://bit.ly/2VvKRLF
Expires January 27, 2020
5. New SLE disease activity measure beats SLEDAI-2K
To take the posttest, go to: https://bit.ly/2W8SVPA
Expires January 31, 2020
Here are 5 articles from the May issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Subclinical hypothyroidism boosts immediate risk of heart failure
To take the posttest, go to: https://bit.ly/2IK0YiL
Expires January 24, 2020
2. Meta-analysis supports aspirin to reduce cardiovascular events
To take the posttest, go to: https://bit.ly/2GJLgSB
Expires January 24, 2020
3. Age of migraine onset may affect stroke risk
To take the posttest, go to: https://bit.ly/2ZAJ5YR
Expires January 24, 2020
4. Women with RA have reduced chance of live birth after assisted reproduction treatment
To take the posttest, go to: https://bit.ly/2VvKRLF
Expires January 27, 2020
5. New SLE disease activity measure beats SLEDAI-2K
To take the posttest, go to: https://bit.ly/2W8SVPA
Expires January 31, 2020
Long-term antibiotic use may heighten stroke, CHD risk
, according to a study in the European Heart Journal.
Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
, according to a study in the European Heart Journal.

Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
, according to a study in the European Heart Journal.

Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
FROM THE EUROPEAN HEART JOURNAL
Key clinical point: Among middle-aged and older women, 2 or more months’ exposure to antibiotics is associated with an increased risk of coronary heart disease or stroke.
Major finding: Long-term antibiotic use in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Study details: An analysis of data from nearly 36,500 women in the Nurses’ Health Study.
Disclosures: The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
Source: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.