User login
Targeted therapies for vascular anomalies continue to be refined
“The medicines we had were believed to be antiangiogenic and they were used not only for tumors but for all sorts of malformations,” Dr. Adams, a pediatric hematologist-oncologist at Children’s Hospital of Philadelphia, recalled during the annual meeting of the Society for Pediatric Dermatology. “I didn’t understand how so many different phenotypes could respond to the same medicine. Not all of them did, but some did have some response.”
She also grew frustrated by the lack of clinical trials and collaborative research groups involving patients with vascular anomalies. “I called this the chicken soup of medical management,” she said. “As we got more involved in vascular anomalies, the power of one patient or that power of a few patients led us in a direction for improved medical management. Or knowledge was gained by one patient who failed all noted medical management and led us into a direction repurposing a drug that actually wound up working.”
Propranolol, for example, became a key medicine for the treatment of vascular anomalies when it was found to improve hemangiomas in children who were given the drug for other reasons. “From this observation a key prospective study was performed and this beta-blocker became FDA approved for the treatment of complicated hemangiomas,” said Dr. Adams, who directs the hospital’s Comprehensive Vascular Anomalies Program. “That was how a bedside observation let to bench intervention, and how presently we are investigating bench interventions related to the mechanism of propranolol therapy.”
Then there is the story of the mammalian target of rapamycin (mTOR) inhibitor sirolimus. In her previous role as medical director of the Hemangioma and Vascular Malformation Center at Cincinnati Children’s Hospital, Dr. Adams and colleagues cared for an infant who presented with a Kaposiform hemangioendothelioma (KHE). “At that time, she was given our standard of practice for the treatment, but our standard of practice was not good enough,” she said.
While other options were being discussed for this patient, “we had been doing some collaborative work with pathology and nephrology on the PIKC3A pathway, because we knew that germline mutations of TEK were involved in this pathway, and we knew that 50% of patients with PTEN mutations had vascular anomalies. So, we hypothesized that this pathway was involved in vascular anomalies.”
They also had earlier success using mTOR inhibition for tuberous sclerosis patients with angiomyolipomas and patients with neurofibromatosis. “We needed a medicine that could be given orally because we did not think this patient was going to do well, so we started her on sirolimus,” Dr. Adams said. “She had a great response. This was followed by a phase 2 study, which proved efficacy and led to discovery of biomarkers.” This is where the angiopoietin-2 story started, she said, noting that this biomarker is now used “to differentiate KLA [Kaposiform lymphangiomatosis] from KHE and KLAs and KHE from other disorders.”
This bedside work helped researchers to better understand the mechanism of action in other disorders, such as observing somatic mutations in PIK3CA in patients with CLOVES syndrome. “This meant that we could now correlate the phenotype to the genotype, and it opened up targeted therapy with developmental therapeutics that were already in use for oncology,” Dr. Adams said. “We know we had mTOR inhibition with sirolimus and everolimus. We now have an AKT inhibitor, a PIK3CA inhibitor, and we now have another side of the pathway which deals with RASopathies, and some other medicines that we can use.”
Miransertib, a potent PAN-AKT inhibitor initially used for breast cancer, is currently being evaluated in open-label, phase 1 and 2 trials in patients with PIK3CA-related overgrowth spectrum (PROS) and Proteus syndrome. The dose used in a pilot study is about one-sixth of the dose used for oncology patients, Dr. Adams said.
She and her colleagues used miransertib to treat a 3-year-old with CLOVES syndrome who had lipomatous infiltration of the abdomen and retroperitoneum with failure to thrive. “He was not eating and was G-tube dependent,” she recalled. “After a month of therapy, he started eating and had improvement in his quality of life,” although despite this improvement volumetric MRI remained unchanged.
Advances in bench to bedside approaches are also under way. Hakon Hakonarson, MD, PhD, the founding director of the Center for Applied Genomics at CHOP, has discovered several genes with in vitro testing and zebra fish modeling, which has been followed by testing medicines on patients.
One such patient, according to Dr. Adams, had a severe central conducting lymphatic anomaly, with a pericardial effusion and significant dysfunction of the central conducting system. The patient was found to have an ARAF mutation, which induces ERK activation. “ERK is downstream of MEK, so the question was whether a MEK inhibitor, trametinib, could be used to treat this patient,” she said. “Trametinib was first used in tissue culture, then used in a zebra fish model and it showed some positive results. Then it was taken to the patient, who had improvement of pulmonary function, remodeling of the lymphatic system, and decrease in the size of his legs.”
Other antiangiogenic agents being used for the treatment of vascular anomalies include bevacizumab, which is being used in hereditary hemorrhagic telangiectasia, and thalidomide for HHT and arteriovenous malformations. For more information, Dr. Adams recommended a comprehensive review of vascular anomalies, related genes, and treatments that was published in Circulation Research.
The goal of future drug therapies is to support normal growth, “so we don’t need a maximum tolerated dose,” Dr. Adams said. “We need to be very careful of short-term and long-term side effects.”
Going forward, she said that she would like to see more natural history studies of vascular anomalies, improved outcome measures for clinical trials, adaptive study design, preclinical testing, animal model studies, universal availability of genomic testing, improvement of NIH funding, research collaboration nationally and internationally, and industry support.
Dr. Adams disclosed that she is a consultant to Venthera and Novartis.
“The medicines we had were believed to be antiangiogenic and they were used not only for tumors but for all sorts of malformations,” Dr. Adams, a pediatric hematologist-oncologist at Children’s Hospital of Philadelphia, recalled during the annual meeting of the Society for Pediatric Dermatology. “I didn’t understand how so many different phenotypes could respond to the same medicine. Not all of them did, but some did have some response.”
She also grew frustrated by the lack of clinical trials and collaborative research groups involving patients with vascular anomalies. “I called this the chicken soup of medical management,” she said. “As we got more involved in vascular anomalies, the power of one patient or that power of a few patients led us in a direction for improved medical management. Or knowledge was gained by one patient who failed all noted medical management and led us into a direction repurposing a drug that actually wound up working.”
Propranolol, for example, became a key medicine for the treatment of vascular anomalies when it was found to improve hemangiomas in children who were given the drug for other reasons. “From this observation a key prospective study was performed and this beta-blocker became FDA approved for the treatment of complicated hemangiomas,” said Dr. Adams, who directs the hospital’s Comprehensive Vascular Anomalies Program. “That was how a bedside observation let to bench intervention, and how presently we are investigating bench interventions related to the mechanism of propranolol therapy.”
Then there is the story of the mammalian target of rapamycin (mTOR) inhibitor sirolimus. In her previous role as medical director of the Hemangioma and Vascular Malformation Center at Cincinnati Children’s Hospital, Dr. Adams and colleagues cared for an infant who presented with a Kaposiform hemangioendothelioma (KHE). “At that time, she was given our standard of practice for the treatment, but our standard of practice was not good enough,” she said.
While other options were being discussed for this patient, “we had been doing some collaborative work with pathology and nephrology on the PIKC3A pathway, because we knew that germline mutations of TEK were involved in this pathway, and we knew that 50% of patients with PTEN mutations had vascular anomalies. So, we hypothesized that this pathway was involved in vascular anomalies.”
They also had earlier success using mTOR inhibition for tuberous sclerosis patients with angiomyolipomas and patients with neurofibromatosis. “We needed a medicine that could be given orally because we did not think this patient was going to do well, so we started her on sirolimus,” Dr. Adams said. “She had a great response. This was followed by a phase 2 study, which proved efficacy and led to discovery of biomarkers.” This is where the angiopoietin-2 story started, she said, noting that this biomarker is now used “to differentiate KLA [Kaposiform lymphangiomatosis] from KHE and KLAs and KHE from other disorders.”
This bedside work helped researchers to better understand the mechanism of action in other disorders, such as observing somatic mutations in PIK3CA in patients with CLOVES syndrome. “This meant that we could now correlate the phenotype to the genotype, and it opened up targeted therapy with developmental therapeutics that were already in use for oncology,” Dr. Adams said. “We know we had mTOR inhibition with sirolimus and everolimus. We now have an AKT inhibitor, a PIK3CA inhibitor, and we now have another side of the pathway which deals with RASopathies, and some other medicines that we can use.”
Miransertib, a potent PAN-AKT inhibitor initially used for breast cancer, is currently being evaluated in open-label, phase 1 and 2 trials in patients with PIK3CA-related overgrowth spectrum (PROS) and Proteus syndrome. The dose used in a pilot study is about one-sixth of the dose used for oncology patients, Dr. Adams said.
She and her colleagues used miransertib to treat a 3-year-old with CLOVES syndrome who had lipomatous infiltration of the abdomen and retroperitoneum with failure to thrive. “He was not eating and was G-tube dependent,” she recalled. “After a month of therapy, he started eating and had improvement in his quality of life,” although despite this improvement volumetric MRI remained unchanged.
Advances in bench to bedside approaches are also under way. Hakon Hakonarson, MD, PhD, the founding director of the Center for Applied Genomics at CHOP, has discovered several genes with in vitro testing and zebra fish modeling, which has been followed by testing medicines on patients.
One such patient, according to Dr. Adams, had a severe central conducting lymphatic anomaly, with a pericardial effusion and significant dysfunction of the central conducting system. The patient was found to have an ARAF mutation, which induces ERK activation. “ERK is downstream of MEK, so the question was whether a MEK inhibitor, trametinib, could be used to treat this patient,” she said. “Trametinib was first used in tissue culture, then used in a zebra fish model and it showed some positive results. Then it was taken to the patient, who had improvement of pulmonary function, remodeling of the lymphatic system, and decrease in the size of his legs.”
Other antiangiogenic agents being used for the treatment of vascular anomalies include bevacizumab, which is being used in hereditary hemorrhagic telangiectasia, and thalidomide for HHT and arteriovenous malformations. For more information, Dr. Adams recommended a comprehensive review of vascular anomalies, related genes, and treatments that was published in Circulation Research.
The goal of future drug therapies is to support normal growth, “so we don’t need a maximum tolerated dose,” Dr. Adams said. “We need to be very careful of short-term and long-term side effects.”
Going forward, she said that she would like to see more natural history studies of vascular anomalies, improved outcome measures for clinical trials, adaptive study design, preclinical testing, animal model studies, universal availability of genomic testing, improvement of NIH funding, research collaboration nationally and internationally, and industry support.
Dr. Adams disclosed that she is a consultant to Venthera and Novartis.
“The medicines we had were believed to be antiangiogenic and they were used not only for tumors but for all sorts of malformations,” Dr. Adams, a pediatric hematologist-oncologist at Children’s Hospital of Philadelphia, recalled during the annual meeting of the Society for Pediatric Dermatology. “I didn’t understand how so many different phenotypes could respond to the same medicine. Not all of them did, but some did have some response.”
She also grew frustrated by the lack of clinical trials and collaborative research groups involving patients with vascular anomalies. “I called this the chicken soup of medical management,” she said. “As we got more involved in vascular anomalies, the power of one patient or that power of a few patients led us in a direction for improved medical management. Or knowledge was gained by one patient who failed all noted medical management and led us into a direction repurposing a drug that actually wound up working.”
Propranolol, for example, became a key medicine for the treatment of vascular anomalies when it was found to improve hemangiomas in children who were given the drug for other reasons. “From this observation a key prospective study was performed and this beta-blocker became FDA approved for the treatment of complicated hemangiomas,” said Dr. Adams, who directs the hospital’s Comprehensive Vascular Anomalies Program. “That was how a bedside observation let to bench intervention, and how presently we are investigating bench interventions related to the mechanism of propranolol therapy.”
Then there is the story of the mammalian target of rapamycin (mTOR) inhibitor sirolimus. In her previous role as medical director of the Hemangioma and Vascular Malformation Center at Cincinnati Children’s Hospital, Dr. Adams and colleagues cared for an infant who presented with a Kaposiform hemangioendothelioma (KHE). “At that time, she was given our standard of practice for the treatment, but our standard of practice was not good enough,” she said.
While other options were being discussed for this patient, “we had been doing some collaborative work with pathology and nephrology on the PIKC3A pathway, because we knew that germline mutations of TEK were involved in this pathway, and we knew that 50% of patients with PTEN mutations had vascular anomalies. So, we hypothesized that this pathway was involved in vascular anomalies.”
They also had earlier success using mTOR inhibition for tuberous sclerosis patients with angiomyolipomas and patients with neurofibromatosis. “We needed a medicine that could be given orally because we did not think this patient was going to do well, so we started her on sirolimus,” Dr. Adams said. “She had a great response. This was followed by a phase 2 study, which proved efficacy and led to discovery of biomarkers.” This is where the angiopoietin-2 story started, she said, noting that this biomarker is now used “to differentiate KLA [Kaposiform lymphangiomatosis] from KHE and KLAs and KHE from other disorders.”
This bedside work helped researchers to better understand the mechanism of action in other disorders, such as observing somatic mutations in PIK3CA in patients with CLOVES syndrome. “This meant that we could now correlate the phenotype to the genotype, and it opened up targeted therapy with developmental therapeutics that were already in use for oncology,” Dr. Adams said. “We know we had mTOR inhibition with sirolimus and everolimus. We now have an AKT inhibitor, a PIK3CA inhibitor, and we now have another side of the pathway which deals with RASopathies, and some other medicines that we can use.”
Miransertib, a potent PAN-AKT inhibitor initially used for breast cancer, is currently being evaluated in open-label, phase 1 and 2 trials in patients with PIK3CA-related overgrowth spectrum (PROS) and Proteus syndrome. The dose used in a pilot study is about one-sixth of the dose used for oncology patients, Dr. Adams said.
She and her colleagues used miransertib to treat a 3-year-old with CLOVES syndrome who had lipomatous infiltration of the abdomen and retroperitoneum with failure to thrive. “He was not eating and was G-tube dependent,” she recalled. “After a month of therapy, he started eating and had improvement in his quality of life,” although despite this improvement volumetric MRI remained unchanged.
Advances in bench to bedside approaches are also under way. Hakon Hakonarson, MD, PhD, the founding director of the Center for Applied Genomics at CHOP, has discovered several genes with in vitro testing and zebra fish modeling, which has been followed by testing medicines on patients.
One such patient, according to Dr. Adams, had a severe central conducting lymphatic anomaly, with a pericardial effusion and significant dysfunction of the central conducting system. The patient was found to have an ARAF mutation, which induces ERK activation. “ERK is downstream of MEK, so the question was whether a MEK inhibitor, trametinib, could be used to treat this patient,” she said. “Trametinib was first used in tissue culture, then used in a zebra fish model and it showed some positive results. Then it was taken to the patient, who had improvement of pulmonary function, remodeling of the lymphatic system, and decrease in the size of his legs.”
Other antiangiogenic agents being used for the treatment of vascular anomalies include bevacizumab, which is being used in hereditary hemorrhagic telangiectasia, and thalidomide for HHT and arteriovenous malformations. For more information, Dr. Adams recommended a comprehensive review of vascular anomalies, related genes, and treatments that was published in Circulation Research.
The goal of future drug therapies is to support normal growth, “so we don’t need a maximum tolerated dose,” Dr. Adams said. “We need to be very careful of short-term and long-term side effects.”
Going forward, she said that she would like to see more natural history studies of vascular anomalies, improved outcome measures for clinical trials, adaptive study design, preclinical testing, animal model studies, universal availability of genomic testing, improvement of NIH funding, research collaboration nationally and internationally, and industry support.
Dr. Adams disclosed that she is a consultant to Venthera and Novartis.
FROM SPD 2021
Ask about itch and joint pain in pediatric psoriasis patients, expert advises
During the annual meeting of the Society for Pediatric Dermatology, Amy S. Paller, MD, MS, marveled on the remarkable advances in the treatment of inflammatory skin disorders during the past 2 decades.
“We’ve come a long way, from disease features being red, thick, and scaly and being treated with nonspecific therapy like topical steroids, keratolytics, and tar, to understanding disease pathogenesis and finding new targeted therapies for inflammatory skin disorders in children,” said Dr. Paller, professor and chair of the department of dermatology at Northwestern University, Chicago. “There are now studies moving forward with gene correction, gene replacement, the gene product replaced, or pathway inhibition to prevent the effects of genetic change.”
Technology is leading the way in generating new therapeutic advances, she continued, beyond traditional “omics” to lipidomics, metabolomics, glycomics, and kinomics. “This has enabled us to find new genetic disorders and their causes, to look at changes in gene expression patterns, and to look at changes in protein expression patterns that give us clues as to how to move forward with better therapy,” she said. “When we’re talking about new insights into pathogenesis-based therapy, we’re talking largely about understanding the pathways that lead to either inflammation or promoting cell proliferation and abnormal differentiation.”
Treating pediatric psoriasis
. “First of all, ask about itch and pain with these patients,” she advised. “Interviews have shown that 61% of children experience some itch, 39% have pain or stinging, and in the ixekizumab trials, 72% had what’s considered meaningful itch, with at least 4 out of 10 (mean intensity 5.3) on the itch numeric rating scale. Little is known about the itch associated with psoriasis and its underlying cause – unrelated to the IL-4/IL-13 pathway activation of atopic dermatitis – but it’s worth asking about. I find that itch of the scalp is especially a problem in psoriasis.”
Physicians should also ask pediatric psoriasis patients about joint pain, because about 1% of them have psoriatic arthritis, which is much less common than in adults, “but important to find and manage,” she added. Dr. Paller recommends the new R-JET rapid joint exam technique, which is accompanied by a three-question survey and body diagram that facilitates identification of true arthritis, “so you can know how quickly to refer”.
Several studies have described an increased risk of metabolic syndrome in adolescents with pediatric psoriasis and now in prepubertal children with the disease. In a recent study of 60 consecutive prepubertal children with psoriasis, 70% of whom had mild disease, 40% were overweight or obese, 53% had central obesity, 27% had high levels of the HOMA-IR (homeostasis model assessment of insulin resistance) despite generally normal levels of fasting glucose, and 30% met criteria for metabolic syndrome.
“This really struck me because our AAD [American Academy of Dermatology] guidelines did not recommend screening for type 2 diabetes in prepubertal children, even if overweight, because the risk is so small,” Dr. Paller said. “This report suggests that we may need to reconsider this recommendation in prepubertal children with psoriasis.”
Meanwhile, the number of medications approved by the Food and Drug Administration and the European Medicines Agency for children with psoriasis who are 6 years of age and above continues to expand, including tumor necrosis factor (TNF) inhibitors, interleukin (IL)-23 inhibitors, and IL-17 inhibitors. Most children can now achieve a PASI 90 within 12 weeks with the IL-23 inhibitor ustekinumab and the IL-17 inhibitors ixekizumab and secukinumab, Dr. Paller said.
In the ixekizumab trial, there are head-to-head comparison data in a European arm that involved the use of etanercept, she said. “What’s most noticeable is the significant difference in those who were able to achieve PASI 90 or above with this IL-17 inhibitor, versus etanercept,” which she added, raises the question of whether aiming for a PASI 75 is adequate, "or should we strive for PASI 90?” A pediatric psoriasis study published in 2020 found that the greatest improvement in quality of life was associated with a PASI 90 and use of systemic treatments.
Looking forward, phase 3 clinical trials are underway in pediatric patients with moderate to severe psoriasis for guselkumab, tildrakizumab, risankizumab, certolizumab, bimekizumab, and brodalumab. “The cost of all of these biologics is high, however. I remind everyone that we still have methotrexate,” she said. “The risk of side effects with our low-dose methotrexate treatment for psoriasis remains low, but methotrexate doesn’t hit these [high] PASI numbers and it’s much slower in its onset than biologics.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, RAPT Therapeutics, Regeneron, and Sanofi.
Commentary by Robert Sidbury, MD, MPH
Dr. Paller reminds us of some essential features of pediatric psoriasis:
• It can hurt. Ask your patients if it does.
• It can itch. Look for excoriations, especially in the scalp.
• It is often associated with metabolic syndrome, so check relevant biometrics and labs, and consider coincident insulin resistance.
• Our traditional clinical trial target of PASI75, or a 75% reduction in body surface area involvement, is just not good enough. Studies have shown that the most meaningful quality-of-life gains come at PASI90 or above.
• With our newer biologics, such as IL-12/23 blockers (for instance, ustekinumab) and IL-17 blockers (for example, ixekizumab and secukinumab), PASI90 and better is a reasonable expectation, not a pipe dream.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/16/22.
During the annual meeting of the Society for Pediatric Dermatology, Amy S. Paller, MD, MS, marveled on the remarkable advances in the treatment of inflammatory skin disorders during the past 2 decades.
“We’ve come a long way, from disease features being red, thick, and scaly and being treated with nonspecific therapy like topical steroids, keratolytics, and tar, to understanding disease pathogenesis and finding new targeted therapies for inflammatory skin disorders in children,” said Dr. Paller, professor and chair of the department of dermatology at Northwestern University, Chicago. “There are now studies moving forward with gene correction, gene replacement, the gene product replaced, or pathway inhibition to prevent the effects of genetic change.”
Technology is leading the way in generating new therapeutic advances, she continued, beyond traditional “omics” to lipidomics, metabolomics, glycomics, and kinomics. “This has enabled us to find new genetic disorders and their causes, to look at changes in gene expression patterns, and to look at changes in protein expression patterns that give us clues as to how to move forward with better therapy,” she said. “When we’re talking about new insights into pathogenesis-based therapy, we’re talking largely about understanding the pathways that lead to either inflammation or promoting cell proliferation and abnormal differentiation.”
Treating pediatric psoriasis
. “First of all, ask about itch and pain with these patients,” she advised. “Interviews have shown that 61% of children experience some itch, 39% have pain or stinging, and in the ixekizumab trials, 72% had what’s considered meaningful itch, with at least 4 out of 10 (mean intensity 5.3) on the itch numeric rating scale. Little is known about the itch associated with psoriasis and its underlying cause – unrelated to the IL-4/IL-13 pathway activation of atopic dermatitis – but it’s worth asking about. I find that itch of the scalp is especially a problem in psoriasis.”
Physicians should also ask pediatric psoriasis patients about joint pain, because about 1% of them have psoriatic arthritis, which is much less common than in adults, “but important to find and manage,” she added. Dr. Paller recommends the new R-JET rapid joint exam technique, which is accompanied by a three-question survey and body diagram that facilitates identification of true arthritis, “so you can know how quickly to refer”.
Several studies have described an increased risk of metabolic syndrome in adolescents with pediatric psoriasis and now in prepubertal children with the disease. In a recent study of 60 consecutive prepubertal children with psoriasis, 70% of whom had mild disease, 40% were overweight or obese, 53% had central obesity, 27% had high levels of the HOMA-IR (homeostasis model assessment of insulin resistance) despite generally normal levels of fasting glucose, and 30% met criteria for metabolic syndrome.
“This really struck me because our AAD [American Academy of Dermatology] guidelines did not recommend screening for type 2 diabetes in prepubertal children, even if overweight, because the risk is so small,” Dr. Paller said. “This report suggests that we may need to reconsider this recommendation in prepubertal children with psoriasis.”
Meanwhile, the number of medications approved by the Food and Drug Administration and the European Medicines Agency for children with psoriasis who are 6 years of age and above continues to expand, including tumor necrosis factor (TNF) inhibitors, interleukin (IL)-23 inhibitors, and IL-17 inhibitors. Most children can now achieve a PASI 90 within 12 weeks with the IL-23 inhibitor ustekinumab and the IL-17 inhibitors ixekizumab and secukinumab, Dr. Paller said.
In the ixekizumab trial, there are head-to-head comparison data in a European arm that involved the use of etanercept, she said. “What’s most noticeable is the significant difference in those who were able to achieve PASI 90 or above with this IL-17 inhibitor, versus etanercept,” which she added, raises the question of whether aiming for a PASI 75 is adequate, "or should we strive for PASI 90?” A pediatric psoriasis study published in 2020 found that the greatest improvement in quality of life was associated with a PASI 90 and use of systemic treatments.
Looking forward, phase 3 clinical trials are underway in pediatric patients with moderate to severe psoriasis for guselkumab, tildrakizumab, risankizumab, certolizumab, bimekizumab, and brodalumab. “The cost of all of these biologics is high, however. I remind everyone that we still have methotrexate,” she said. “The risk of side effects with our low-dose methotrexate treatment for psoriasis remains low, but methotrexate doesn’t hit these [high] PASI numbers and it’s much slower in its onset than biologics.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, RAPT Therapeutics, Regeneron, and Sanofi.
Commentary by Robert Sidbury, MD, MPH
Dr. Paller reminds us of some essential features of pediatric psoriasis:
• It can hurt. Ask your patients if it does.
• It can itch. Look for excoriations, especially in the scalp.
• It is often associated with metabolic syndrome, so check relevant biometrics and labs, and consider coincident insulin resistance.
• Our traditional clinical trial target of PASI75, or a 75% reduction in body surface area involvement, is just not good enough. Studies have shown that the most meaningful quality-of-life gains come at PASI90 or above.
• With our newer biologics, such as IL-12/23 blockers (for instance, ustekinumab) and IL-17 blockers (for example, ixekizumab and secukinumab), PASI90 and better is a reasonable expectation, not a pipe dream.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/16/22.
During the annual meeting of the Society for Pediatric Dermatology, Amy S. Paller, MD, MS, marveled on the remarkable advances in the treatment of inflammatory skin disorders during the past 2 decades.
“We’ve come a long way, from disease features being red, thick, and scaly and being treated with nonspecific therapy like topical steroids, keratolytics, and tar, to understanding disease pathogenesis and finding new targeted therapies for inflammatory skin disorders in children,” said Dr. Paller, professor and chair of the department of dermatology at Northwestern University, Chicago. “There are now studies moving forward with gene correction, gene replacement, the gene product replaced, or pathway inhibition to prevent the effects of genetic change.”
Technology is leading the way in generating new therapeutic advances, she continued, beyond traditional “omics” to lipidomics, metabolomics, glycomics, and kinomics. “This has enabled us to find new genetic disorders and their causes, to look at changes in gene expression patterns, and to look at changes in protein expression patterns that give us clues as to how to move forward with better therapy,” she said. “When we’re talking about new insights into pathogenesis-based therapy, we’re talking largely about understanding the pathways that lead to either inflammation or promoting cell proliferation and abnormal differentiation.”
Treating pediatric psoriasis
. “First of all, ask about itch and pain with these patients,” she advised. “Interviews have shown that 61% of children experience some itch, 39% have pain or stinging, and in the ixekizumab trials, 72% had what’s considered meaningful itch, with at least 4 out of 10 (mean intensity 5.3) on the itch numeric rating scale. Little is known about the itch associated with psoriasis and its underlying cause – unrelated to the IL-4/IL-13 pathway activation of atopic dermatitis – but it’s worth asking about. I find that itch of the scalp is especially a problem in psoriasis.”
Physicians should also ask pediatric psoriasis patients about joint pain, because about 1% of them have psoriatic arthritis, which is much less common than in adults, “but important to find and manage,” she added. Dr. Paller recommends the new R-JET rapid joint exam technique, which is accompanied by a three-question survey and body diagram that facilitates identification of true arthritis, “so you can know how quickly to refer”.
Several studies have described an increased risk of metabolic syndrome in adolescents with pediatric psoriasis and now in prepubertal children with the disease. In a recent study of 60 consecutive prepubertal children with psoriasis, 70% of whom had mild disease, 40% were overweight or obese, 53% had central obesity, 27% had high levels of the HOMA-IR (homeostasis model assessment of insulin resistance) despite generally normal levels of fasting glucose, and 30% met criteria for metabolic syndrome.
“This really struck me because our AAD [American Academy of Dermatology] guidelines did not recommend screening for type 2 diabetes in prepubertal children, even if overweight, because the risk is so small,” Dr. Paller said. “This report suggests that we may need to reconsider this recommendation in prepubertal children with psoriasis.”
Meanwhile, the number of medications approved by the Food and Drug Administration and the European Medicines Agency for children with psoriasis who are 6 years of age and above continues to expand, including tumor necrosis factor (TNF) inhibitors, interleukin (IL)-23 inhibitors, and IL-17 inhibitors. Most children can now achieve a PASI 90 within 12 weeks with the IL-23 inhibitor ustekinumab and the IL-17 inhibitors ixekizumab and secukinumab, Dr. Paller said.
In the ixekizumab trial, there are head-to-head comparison data in a European arm that involved the use of etanercept, she said. “What’s most noticeable is the significant difference in those who were able to achieve PASI 90 or above with this IL-17 inhibitor, versus etanercept,” which she added, raises the question of whether aiming for a PASI 75 is adequate, "or should we strive for PASI 90?” A pediatric psoriasis study published in 2020 found that the greatest improvement in quality of life was associated with a PASI 90 and use of systemic treatments.
Looking forward, phase 3 clinical trials are underway in pediatric patients with moderate to severe psoriasis for guselkumab, tildrakizumab, risankizumab, certolizumab, bimekizumab, and brodalumab. “The cost of all of these biologics is high, however. I remind everyone that we still have methotrexate,” she said. “The risk of side effects with our low-dose methotrexate treatment for psoriasis remains low, but methotrexate doesn’t hit these [high] PASI numbers and it’s much slower in its onset than biologics.”
Dr. Paller disclosed that she is a consultant to and/or an investigator for AbbVie, Arena, Bausch, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Forte, LEO Pharma, LifeMax, Pfizer, RAPT Therapeutics, Regeneron, and Sanofi.
Commentary by Robert Sidbury, MD, MPH
Dr. Paller reminds us of some essential features of pediatric psoriasis:
• It can hurt. Ask your patients if it does.
• It can itch. Look for excoriations, especially in the scalp.
• It is often associated with metabolic syndrome, so check relevant biometrics and labs, and consider coincident insulin resistance.
• Our traditional clinical trial target of PASI75, or a 75% reduction in body surface area involvement, is just not good enough. Studies have shown that the most meaningful quality-of-life gains come at PASI90 or above.
• With our newer biologics, such as IL-12/23 blockers (for instance, ustekinumab) and IL-17 blockers (for example, ixekizumab and secukinumab), PASI90 and better is a reasonable expectation, not a pipe dream.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
This article was updated 6/16/22.
FROM SPD 2021
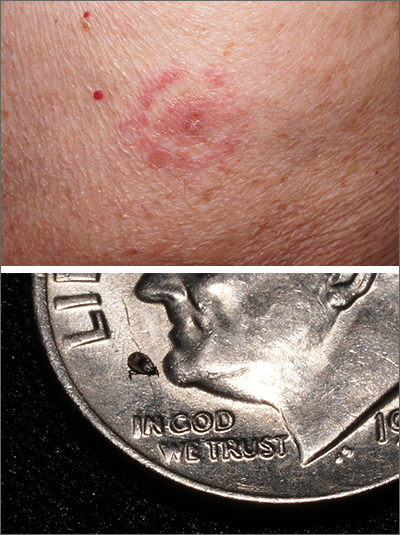
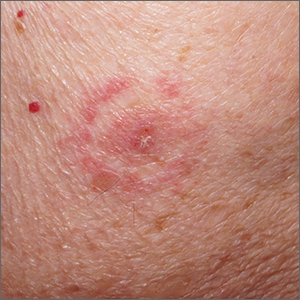
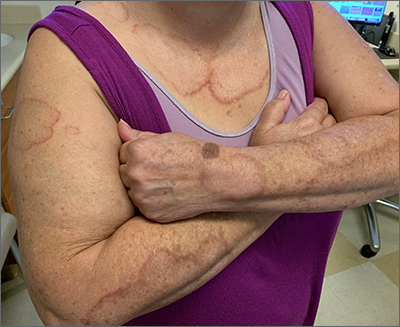
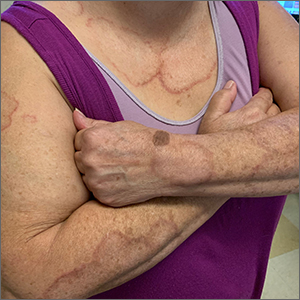
Target lesion
The original diagnosis in this case was correct—Lyme disease (erythema chronicum migrans)—but unfortunately, the treatment was inadequate. Initially, this patient received a single dose of doxycycline 200 mg po, which is the appropriate preventative remedy when a deer tick has been attached for at least 36 hours and treatment can be initiated within 72 hours of tick removal. However, deer ticks, especially nymphs, are very small and their presence can go unnoticed, leading patients to guess (sometimes incorrectly) at the length of time a tick has been attached.
In this case, when the patient and his wife thought about it a bit more, they indicated that the tick may have been attached for several days before they removed it and went to see the PCP. Had the PCP known the tick had been attached longer, she would have advised watchful waiting and observation for signs and symptoms of Lyme disease.
Originally described in cases from Lyme, Connecticut, Lyme disease is now endemic to the northeastern United States from Maine to Virginia, and from the upper midwest to Minnesota. Laboratory diagnosis can be made by a 2-tiered serologic screening and confirmation with an enzyme-linked assay, followed by a western blot for positive or equivocal screening tests.
In 2019, the most recent year of reported Centers for Disease Control and Prevention (CDC) surveillance data, Maine recorded the highest incidence rate of Lyme disease.1 In 70% to 80% of cases, an expanding horizon of infection creates the characteristic targetoid pink patch centered on the site of the tick bite.2
If there is a targetoid rash present in a patient from an endemic area, the CDC recommends empiric therapy with doxycycline 100 mg po bid for 10 to 14 days.3 (Worth noting: Patches may appear rather small in diameter, as was true in this case, or may be many centimeters in size and mimic cellulitis.) Alternative regimens for early localized disease include amoxicillin 500 mg po qid for pregnant patients, and cefuroxime 500 mg bid daily for 14 days for patients who are allergic to doxycycline.
Once treatment has been initiated, it’s important to monitor patients for any worsening symptoms, including fever and weakness, as these may be an indication of treatment failure or co-occurrence of anaplasmosis, ehrlichiosis, or babesiosis. In this case, the patient was prescribed doxycycline 100 mg bid for 14 days without any further signs or symptoms.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Lyme disease maps: most recent year. Centers for Disease Control and Prevention. Updated April 29, 2021. Accessed August 12, 2021. https://www.cdc.gov/lyme/datasurveillance/maps-recent.html
2. Signs and symptoms of untreated Lyme disease. Centers for Disease Control and Prevention. Updated January 15, 2021. Accessed August 13, 2021. https://www.cdc.gov/lyme/signs_symptoms/index.html
3. Treatment of erythema migrans. Centers for Disease Control and Prevention. Updated November 3, 2020. Accessed August 12, 2021. https://www.cdc.gov/lyme/treatment/index.html
The original diagnosis in this case was correct—Lyme disease (erythema chronicum migrans)—but unfortunately, the treatment was inadequate. Initially, this patient received a single dose of doxycycline 200 mg po, which is the appropriate preventative remedy when a deer tick has been attached for at least 36 hours and treatment can be initiated within 72 hours of tick removal. However, deer ticks, especially nymphs, are very small and their presence can go unnoticed, leading patients to guess (sometimes incorrectly) at the length of time a tick has been attached.
In this case, when the patient and his wife thought about it a bit more, they indicated that the tick may have been attached for several days before they removed it and went to see the PCP. Had the PCP known the tick had been attached longer, she would have advised watchful waiting and observation for signs and symptoms of Lyme disease.
Originally described in cases from Lyme, Connecticut, Lyme disease is now endemic to the northeastern United States from Maine to Virginia, and from the upper midwest to Minnesota. Laboratory diagnosis can be made by a 2-tiered serologic screening and confirmation with an enzyme-linked assay, followed by a western blot for positive or equivocal screening tests.
In 2019, the most recent year of reported Centers for Disease Control and Prevention (CDC) surveillance data, Maine recorded the highest incidence rate of Lyme disease.1 In 70% to 80% of cases, an expanding horizon of infection creates the characteristic targetoid pink patch centered on the site of the tick bite.2
If there is a targetoid rash present in a patient from an endemic area, the CDC recommends empiric therapy with doxycycline 100 mg po bid for 10 to 14 days.3 (Worth noting: Patches may appear rather small in diameter, as was true in this case, or may be many centimeters in size and mimic cellulitis.) Alternative regimens for early localized disease include amoxicillin 500 mg po qid for pregnant patients, and cefuroxime 500 mg bid daily for 14 days for patients who are allergic to doxycycline.
Once treatment has been initiated, it’s important to monitor patients for any worsening symptoms, including fever and weakness, as these may be an indication of treatment failure or co-occurrence of anaplasmosis, ehrlichiosis, or babesiosis. In this case, the patient was prescribed doxycycline 100 mg bid for 14 days without any further signs or symptoms.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
The original diagnosis in this case was correct—Lyme disease (erythema chronicum migrans)—but unfortunately, the treatment was inadequate. Initially, this patient received a single dose of doxycycline 200 mg po, which is the appropriate preventative remedy when a deer tick has been attached for at least 36 hours and treatment can be initiated within 72 hours of tick removal. However, deer ticks, especially nymphs, are very small and their presence can go unnoticed, leading patients to guess (sometimes incorrectly) at the length of time a tick has been attached.
In this case, when the patient and his wife thought about it a bit more, they indicated that the tick may have been attached for several days before they removed it and went to see the PCP. Had the PCP known the tick had been attached longer, she would have advised watchful waiting and observation for signs and symptoms of Lyme disease.
Originally described in cases from Lyme, Connecticut, Lyme disease is now endemic to the northeastern United States from Maine to Virginia, and from the upper midwest to Minnesota. Laboratory diagnosis can be made by a 2-tiered serologic screening and confirmation with an enzyme-linked assay, followed by a western blot for positive or equivocal screening tests.
In 2019, the most recent year of reported Centers for Disease Control and Prevention (CDC) surveillance data, Maine recorded the highest incidence rate of Lyme disease.1 In 70% to 80% of cases, an expanding horizon of infection creates the characteristic targetoid pink patch centered on the site of the tick bite.2
If there is a targetoid rash present in a patient from an endemic area, the CDC recommends empiric therapy with doxycycline 100 mg po bid for 10 to 14 days.3 (Worth noting: Patches may appear rather small in diameter, as was true in this case, or may be many centimeters in size and mimic cellulitis.) Alternative regimens for early localized disease include amoxicillin 500 mg po qid for pregnant patients, and cefuroxime 500 mg bid daily for 14 days for patients who are allergic to doxycycline.
Once treatment has been initiated, it’s important to monitor patients for any worsening symptoms, including fever and weakness, as these may be an indication of treatment failure or co-occurrence of anaplasmosis, ehrlichiosis, or babesiosis. In this case, the patient was prescribed doxycycline 100 mg bid for 14 days without any further signs or symptoms.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Lyme disease maps: most recent year. Centers for Disease Control and Prevention. Updated April 29, 2021. Accessed August 12, 2021. https://www.cdc.gov/lyme/datasurveillance/maps-recent.html
2. Signs and symptoms of untreated Lyme disease. Centers for Disease Control and Prevention. Updated January 15, 2021. Accessed August 13, 2021. https://www.cdc.gov/lyme/signs_symptoms/index.html
3. Treatment of erythema migrans. Centers for Disease Control and Prevention. Updated November 3, 2020. Accessed August 12, 2021. https://www.cdc.gov/lyme/treatment/index.html
1. Lyme disease maps: most recent year. Centers for Disease Control and Prevention. Updated April 29, 2021. Accessed August 12, 2021. https://www.cdc.gov/lyme/datasurveillance/maps-recent.html
2. Signs and symptoms of untreated Lyme disease. Centers for Disease Control and Prevention. Updated January 15, 2021. Accessed August 13, 2021. https://www.cdc.gov/lyme/signs_symptoms/index.html
3. Treatment of erythema migrans. Centers for Disease Control and Prevention. Updated November 3, 2020. Accessed August 12, 2021. https://www.cdc.gov/lyme/treatment/index.html
Biologic benefit in psoriasis might extend to arthritis prevention
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
FROM ARTHRITIS & RHEUMATOLOGY
NIH to study COVID vaccine booster in people with autoimmune disease
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
FDA okays difelikefalin for dialysis-associated pruritus in patients with CKD
Some nephrologists welcomed the Aug. 23 approval of this new option for treating pruritus, a relatively common and often hard-to-resolve complication of dialysis in patients with chronic kidney disease (CKD) that can substantially impinge on quality of life for some patients, but also voiced uncertainty about the role of a new agent with a modest trial track record that may be expensive and face insurance-coverage hurdles.
“Uptake of difelikefalin will depend on awareness of itch among patients dependent on hemodialysis, and on payment policies,” predicted Daniel E. Weiner, MD, a nephrologist at Tufts Medical Center in Boston. “Pruritus is underdiagnosed among people with kidney failure, and in some patients ongoing pruritus can be highly impactful on sleep and quality of life. The clinical trial results were very encouraging that difelikefalin is effective and safe,” which makes recognition of pruritus as a significant issue for patients a key factor in uptake of the new drug, Dr. Weiner, an investigator in a difelikefalin clinical study, said in an interview.
Other nephrologists acknowledged the substantial problem that itch can pose for many patients with CKD on dialysis but questioned the weight of evidence behind difelikefalin’s approval.
Two pivotal trials with fewer than 900 total randomized patients
The data considered by the FDA primarily featured results from two pivotal trials, KALM-1 and KALM-2. KALM-1 randomized 378 patients with CKD and on hemodialysis and with moderate to severe pruritus to intravenous treatment with difelikefalin or placebo three times a week for 12 weeks with a primary endpoint of an improvement (decrease) of at least 3 points from baseline in their Worst Itching Intensity Numerical Rating Scale (WI-NRS) score, which averaged just over 7 points at baseline. After 12 weeks on treatment, 52% of patients who received difelikefalin had at least a 3-point drop, compared with 31% of patients who received placebo, a significant difference. The results appeared in a 2020 report in the New England Journal of Medicine.
Confirmatory results came in the second pivotal trial, KALM-2, a similarly designed, 12-week study that randomized 473 patients, with 54% of those in the active arm achieving at least a 3-point cut in their baseline WI-NRS score, compared with 42% of patients who received placebo, a significant difference. A report at the Kidney Week meeting sponsored by the National Kidney Foundation in October 2020 presented the KALM-2 results, but the findings have not yet appeared in a published article.
In sum, the data suggest that treatment with difelikefalin will, on average, produce a clinically meaningful effect on itch compared with placebo in about 20% of patients, with nearly half the patients who receive the active drug having a less robust response and many patients who receive no active treatment also show a meaningful cut in their pruritus severity in a trial setting, noted Paul Palevsky, MD, professor of medicine at the University of Pittsburgh and chief of the renal section at the Veterans Affairs Pittsburgh Healthcare System.
The upshot is that questions linger over which patients are the best candidates for this drug and how it might perform in real-world practice given difelikefalin’s limited track record, Dr. Palevsky said in an interview.
In addition, the labeling specifies the indication is for patients with moderate to severe pruritus, but itching severity is not routinely quantified in these patients in current practice, added Dr. Palevsky, who is also president of the National Kidney Foundation.
Dr. Weiner noted that another unknown is the appropriate duration of treatment in real-world use.
What will it cost, and will it be covered?
The drug’s price and insurance coverage will likely be a major factor in uptake of the new drug, agreed both Dr. Weiner and Dr. Palevsky, especially the coverage decision for Medicare patients by the Centers for Medicare & Medicaid Services. A corollary is whether or not coverage for difelikefalin, which patients receive as an intravenous infusion during each of their usual three-times-a-week dialysis sessions, will lie outside of the bundled dialysis reimbursement payment. If is no mechanism exists to pay for difelikefalin separately beyond the current bundled dialysis rate, “I suspect it will not get used very much unless it is very inexpensive,” predicted Dr. Weiner.
Another issue is where difelikefalin fits within the lineup of standard treatment options. “A lot of people receiving hemodialysis suffer from pruritus and have not been successfully treated. For these individuals difelikefalin could be a game changer,” Dr. Weiner said.
Other nephrologists have a more positive take on the existing treatment options.
“Start systemic therapy for patients with itch that is significantly affecting quality of life; stepping up from topical therapy just delays effective treatment,” advised Hugh C. Rayner, MD, a nephrologist affiliated with Birmingham (England) Heartland’s Hospital who was lead author on a review of pruritus treatments for patients with CKD on hemodialysis.
“Standard systemic therapy is gabapentin or pregabalin,” an approach “supported by robust evidence confirmed in a Cochrane review,” he said in an interview. The impact of difelikefalin “will be limited as its effectiveness in reducing itch is modest at best and far inferior to gabapentin and pregabalin,” Dr. Rayner added. Difelikefalin’s “main downsides will be its cost, compared with gabapentin, and its gastrointestinal side effects.”
Adverse-event profiles
In KALM-1, the most frequent adverse effects from difelikefalin treatment was diarrhea, in 10% of patients, compared with a 4% rate among patients who received placebo. Vomiting occurred at a 5% incidence on difelikefalin and in 3% of patients on placebo. All serious adverse events occurred in 26% of patients on difelikefalin and in 22% of those who received placebo. Discontinuations because of an adverse event occurred in 8% of patients on difelikefalin and in 5% of the placebo patients.
An editorial that accompanied the published KALM-1 report in 2020 said “the findings are compelling, although diarrhea, dizziness, and vomiting were frequent side effects.”
Both Dr. Weiner and Dr. Palevsky were more reserved than Dr. Rayner in their appraisal of gabapentin and pregabalin, although Dr. Palevsky admitted that he has prescribed one or the other of these two drugs to “lots of patients,” especially gabapentin. “But they are not completely benign drugs,” he cautioned, a concern echoed by Dr. Weiner.
“Antihistamines, gabapentin, and pregabalin have a high side-effect burden in patients on hemodialysis and limited efficacy, and are poor options for chronic pruritus management,” explained Dr. Weiner. “I would favor difelikefalin to chronic prescription of these other agents” because difelikefalin “appears effective and has a very low side effect burden. Very few effective treatments for pruritus do not have side effects.”
Difelikefalin is a peripherally restricted, selective kappa opioid receptor agonist that exerts antipruritic effects by activating kappa opioid receptors on peripheral neurons and immune cells. The drug’s hydrophilic, small-peptide structure restricts passive diffusion across membranes, which limits the drug’s access to kappa opioid receptors in the central nervous system and hence reduces potential adverse effects.
The FDA made this approval decision without consulting an advisory committee. The companies that will market difelikefalin (Korsuva), Cara Therapeutics and Vifor Pharma, announced that their U.S. promotional launch of the drug starts early in 2022.
The KALM-1 and KALM-2 studies were sponsored by Cara Therapeutics and Vifor Pharma, the two companies that have been jointly developing difelikefalin. Dr. Pavelsky and Dr. Rayner had no relevant disclosures. Dr. Weiner was previously an adviser to Cara and Vifor and participated as an investigator in a difelikefalin clinical study, but more recently has had no relationships with the companies.
Some nephrologists welcomed the Aug. 23 approval of this new option for treating pruritus, a relatively common and often hard-to-resolve complication of dialysis in patients with chronic kidney disease (CKD) that can substantially impinge on quality of life for some patients, but also voiced uncertainty about the role of a new agent with a modest trial track record that may be expensive and face insurance-coverage hurdles.
“Uptake of difelikefalin will depend on awareness of itch among patients dependent on hemodialysis, and on payment policies,” predicted Daniel E. Weiner, MD, a nephrologist at Tufts Medical Center in Boston. “Pruritus is underdiagnosed among people with kidney failure, and in some patients ongoing pruritus can be highly impactful on sleep and quality of life. The clinical trial results were very encouraging that difelikefalin is effective and safe,” which makes recognition of pruritus as a significant issue for patients a key factor in uptake of the new drug, Dr. Weiner, an investigator in a difelikefalin clinical study, said in an interview.
Other nephrologists acknowledged the substantial problem that itch can pose for many patients with CKD on dialysis but questioned the weight of evidence behind difelikefalin’s approval.
Two pivotal trials with fewer than 900 total randomized patients
The data considered by the FDA primarily featured results from two pivotal trials, KALM-1 and KALM-2. KALM-1 randomized 378 patients with CKD and on hemodialysis and with moderate to severe pruritus to intravenous treatment with difelikefalin or placebo three times a week for 12 weeks with a primary endpoint of an improvement (decrease) of at least 3 points from baseline in their Worst Itching Intensity Numerical Rating Scale (WI-NRS) score, which averaged just over 7 points at baseline. After 12 weeks on treatment, 52% of patients who received difelikefalin had at least a 3-point drop, compared with 31% of patients who received placebo, a significant difference. The results appeared in a 2020 report in the New England Journal of Medicine.
Confirmatory results came in the second pivotal trial, KALM-2, a similarly designed, 12-week study that randomized 473 patients, with 54% of those in the active arm achieving at least a 3-point cut in their baseline WI-NRS score, compared with 42% of patients who received placebo, a significant difference. A report at the Kidney Week meeting sponsored by the National Kidney Foundation in October 2020 presented the KALM-2 results, but the findings have not yet appeared in a published article.
In sum, the data suggest that treatment with difelikefalin will, on average, produce a clinically meaningful effect on itch compared with placebo in about 20% of patients, with nearly half the patients who receive the active drug having a less robust response and many patients who receive no active treatment also show a meaningful cut in their pruritus severity in a trial setting, noted Paul Palevsky, MD, professor of medicine at the University of Pittsburgh and chief of the renal section at the Veterans Affairs Pittsburgh Healthcare System.
The upshot is that questions linger over which patients are the best candidates for this drug and how it might perform in real-world practice given difelikefalin’s limited track record, Dr. Palevsky said in an interview.
In addition, the labeling specifies the indication is for patients with moderate to severe pruritus, but itching severity is not routinely quantified in these patients in current practice, added Dr. Palevsky, who is also president of the National Kidney Foundation.
Dr. Weiner noted that another unknown is the appropriate duration of treatment in real-world use.
What will it cost, and will it be covered?
The drug’s price and insurance coverage will likely be a major factor in uptake of the new drug, agreed both Dr. Weiner and Dr. Palevsky, especially the coverage decision for Medicare patients by the Centers for Medicare & Medicaid Services. A corollary is whether or not coverage for difelikefalin, which patients receive as an intravenous infusion during each of their usual three-times-a-week dialysis sessions, will lie outside of the bundled dialysis reimbursement payment. If is no mechanism exists to pay for difelikefalin separately beyond the current bundled dialysis rate, “I suspect it will not get used very much unless it is very inexpensive,” predicted Dr. Weiner.
Another issue is where difelikefalin fits within the lineup of standard treatment options. “A lot of people receiving hemodialysis suffer from pruritus and have not been successfully treated. For these individuals difelikefalin could be a game changer,” Dr. Weiner said.
Other nephrologists have a more positive take on the existing treatment options.
“Start systemic therapy for patients with itch that is significantly affecting quality of life; stepping up from topical therapy just delays effective treatment,” advised Hugh C. Rayner, MD, a nephrologist affiliated with Birmingham (England) Heartland’s Hospital who was lead author on a review of pruritus treatments for patients with CKD on hemodialysis.
“Standard systemic therapy is gabapentin or pregabalin,” an approach “supported by robust evidence confirmed in a Cochrane review,” he said in an interview. The impact of difelikefalin “will be limited as its effectiveness in reducing itch is modest at best and far inferior to gabapentin and pregabalin,” Dr. Rayner added. Difelikefalin’s “main downsides will be its cost, compared with gabapentin, and its gastrointestinal side effects.”
Adverse-event profiles
In KALM-1, the most frequent adverse effects from difelikefalin treatment was diarrhea, in 10% of patients, compared with a 4% rate among patients who received placebo. Vomiting occurred at a 5% incidence on difelikefalin and in 3% of patients on placebo. All serious adverse events occurred in 26% of patients on difelikefalin and in 22% of those who received placebo. Discontinuations because of an adverse event occurred in 8% of patients on difelikefalin and in 5% of the placebo patients.
An editorial that accompanied the published KALM-1 report in 2020 said “the findings are compelling, although diarrhea, dizziness, and vomiting were frequent side effects.”
Both Dr. Weiner and Dr. Palevsky were more reserved than Dr. Rayner in their appraisal of gabapentin and pregabalin, although Dr. Palevsky admitted that he has prescribed one or the other of these two drugs to “lots of patients,” especially gabapentin. “But they are not completely benign drugs,” he cautioned, a concern echoed by Dr. Weiner.
“Antihistamines, gabapentin, and pregabalin have a high side-effect burden in patients on hemodialysis and limited efficacy, and are poor options for chronic pruritus management,” explained Dr. Weiner. “I would favor difelikefalin to chronic prescription of these other agents” because difelikefalin “appears effective and has a very low side effect burden. Very few effective treatments for pruritus do not have side effects.”
Difelikefalin is a peripherally restricted, selective kappa opioid receptor agonist that exerts antipruritic effects by activating kappa opioid receptors on peripheral neurons and immune cells. The drug’s hydrophilic, small-peptide structure restricts passive diffusion across membranes, which limits the drug’s access to kappa opioid receptors in the central nervous system and hence reduces potential adverse effects.
The FDA made this approval decision without consulting an advisory committee. The companies that will market difelikefalin (Korsuva), Cara Therapeutics and Vifor Pharma, announced that their U.S. promotional launch of the drug starts early in 2022.
The KALM-1 and KALM-2 studies were sponsored by Cara Therapeutics and Vifor Pharma, the two companies that have been jointly developing difelikefalin. Dr. Pavelsky and Dr. Rayner had no relevant disclosures. Dr. Weiner was previously an adviser to Cara and Vifor and participated as an investigator in a difelikefalin clinical study, but more recently has had no relationships with the companies.
Some nephrologists welcomed the Aug. 23 approval of this new option for treating pruritus, a relatively common and often hard-to-resolve complication of dialysis in patients with chronic kidney disease (CKD) that can substantially impinge on quality of life for some patients, but also voiced uncertainty about the role of a new agent with a modest trial track record that may be expensive and face insurance-coverage hurdles.
“Uptake of difelikefalin will depend on awareness of itch among patients dependent on hemodialysis, and on payment policies,” predicted Daniel E. Weiner, MD, a nephrologist at Tufts Medical Center in Boston. “Pruritus is underdiagnosed among people with kidney failure, and in some patients ongoing pruritus can be highly impactful on sleep and quality of life. The clinical trial results were very encouraging that difelikefalin is effective and safe,” which makes recognition of pruritus as a significant issue for patients a key factor in uptake of the new drug, Dr. Weiner, an investigator in a difelikefalin clinical study, said in an interview.
Other nephrologists acknowledged the substantial problem that itch can pose for many patients with CKD on dialysis but questioned the weight of evidence behind difelikefalin’s approval.
Two pivotal trials with fewer than 900 total randomized patients
The data considered by the FDA primarily featured results from two pivotal trials, KALM-1 and KALM-2. KALM-1 randomized 378 patients with CKD and on hemodialysis and with moderate to severe pruritus to intravenous treatment with difelikefalin or placebo three times a week for 12 weeks with a primary endpoint of an improvement (decrease) of at least 3 points from baseline in their Worst Itching Intensity Numerical Rating Scale (WI-NRS) score, which averaged just over 7 points at baseline. After 12 weeks on treatment, 52% of patients who received difelikefalin had at least a 3-point drop, compared with 31% of patients who received placebo, a significant difference. The results appeared in a 2020 report in the New England Journal of Medicine.
Confirmatory results came in the second pivotal trial, KALM-2, a similarly designed, 12-week study that randomized 473 patients, with 54% of those in the active arm achieving at least a 3-point cut in their baseline WI-NRS score, compared with 42% of patients who received placebo, a significant difference. A report at the Kidney Week meeting sponsored by the National Kidney Foundation in October 2020 presented the KALM-2 results, but the findings have not yet appeared in a published article.
In sum, the data suggest that treatment with difelikefalin will, on average, produce a clinically meaningful effect on itch compared with placebo in about 20% of patients, with nearly half the patients who receive the active drug having a less robust response and many patients who receive no active treatment also show a meaningful cut in their pruritus severity in a trial setting, noted Paul Palevsky, MD, professor of medicine at the University of Pittsburgh and chief of the renal section at the Veterans Affairs Pittsburgh Healthcare System.
The upshot is that questions linger over which patients are the best candidates for this drug and how it might perform in real-world practice given difelikefalin’s limited track record, Dr. Palevsky said in an interview.
In addition, the labeling specifies the indication is for patients with moderate to severe pruritus, but itching severity is not routinely quantified in these patients in current practice, added Dr. Palevsky, who is also president of the National Kidney Foundation.
Dr. Weiner noted that another unknown is the appropriate duration of treatment in real-world use.
What will it cost, and will it be covered?
The drug’s price and insurance coverage will likely be a major factor in uptake of the new drug, agreed both Dr. Weiner and Dr. Palevsky, especially the coverage decision for Medicare patients by the Centers for Medicare & Medicaid Services. A corollary is whether or not coverage for difelikefalin, which patients receive as an intravenous infusion during each of their usual three-times-a-week dialysis sessions, will lie outside of the bundled dialysis reimbursement payment. If is no mechanism exists to pay for difelikefalin separately beyond the current bundled dialysis rate, “I suspect it will not get used very much unless it is very inexpensive,” predicted Dr. Weiner.
Another issue is where difelikefalin fits within the lineup of standard treatment options. “A lot of people receiving hemodialysis suffer from pruritus and have not been successfully treated. For these individuals difelikefalin could be a game changer,” Dr. Weiner said.
Other nephrologists have a more positive take on the existing treatment options.
“Start systemic therapy for patients with itch that is significantly affecting quality of life; stepping up from topical therapy just delays effective treatment,” advised Hugh C. Rayner, MD, a nephrologist affiliated with Birmingham (England) Heartland’s Hospital who was lead author on a review of pruritus treatments for patients with CKD on hemodialysis.
“Standard systemic therapy is gabapentin or pregabalin,” an approach “supported by robust evidence confirmed in a Cochrane review,” he said in an interview. The impact of difelikefalin “will be limited as its effectiveness in reducing itch is modest at best and far inferior to gabapentin and pregabalin,” Dr. Rayner added. Difelikefalin’s “main downsides will be its cost, compared with gabapentin, and its gastrointestinal side effects.”
Adverse-event profiles
In KALM-1, the most frequent adverse effects from difelikefalin treatment was diarrhea, in 10% of patients, compared with a 4% rate among patients who received placebo. Vomiting occurred at a 5% incidence on difelikefalin and in 3% of patients on placebo. All serious adverse events occurred in 26% of patients on difelikefalin and in 22% of those who received placebo. Discontinuations because of an adverse event occurred in 8% of patients on difelikefalin and in 5% of the placebo patients.
An editorial that accompanied the published KALM-1 report in 2020 said “the findings are compelling, although diarrhea, dizziness, and vomiting were frequent side effects.”
Both Dr. Weiner and Dr. Palevsky were more reserved than Dr. Rayner in their appraisal of gabapentin and pregabalin, although Dr. Palevsky admitted that he has prescribed one or the other of these two drugs to “lots of patients,” especially gabapentin. “But they are not completely benign drugs,” he cautioned, a concern echoed by Dr. Weiner.
“Antihistamines, gabapentin, and pregabalin have a high side-effect burden in patients on hemodialysis and limited efficacy, and are poor options for chronic pruritus management,” explained Dr. Weiner. “I would favor difelikefalin to chronic prescription of these other agents” because difelikefalin “appears effective and has a very low side effect burden. Very few effective treatments for pruritus do not have side effects.”
Difelikefalin is a peripherally restricted, selective kappa opioid receptor agonist that exerts antipruritic effects by activating kappa opioid receptors on peripheral neurons and immune cells. The drug’s hydrophilic, small-peptide structure restricts passive diffusion across membranes, which limits the drug’s access to kappa opioid receptors in the central nervous system and hence reduces potential adverse effects.
The FDA made this approval decision without consulting an advisory committee. The companies that will market difelikefalin (Korsuva), Cara Therapeutics and Vifor Pharma, announced that their U.S. promotional launch of the drug starts early in 2022.
The KALM-1 and KALM-2 studies were sponsored by Cara Therapeutics and Vifor Pharma, the two companies that have been jointly developing difelikefalin. Dr. Pavelsky and Dr. Rayner had no relevant disclosures. Dr. Weiner was previously an adviser to Cara and Vifor and participated as an investigator in a difelikefalin clinical study, but more recently has had no relationships with the companies.
Arm and neck rash
The manifestation of what appeared to be an exaggerated sunburn with associated pruritis and blistering (made worse after sun exposure), along with the recent initiation of HCTZ, led to the diagnosis of a phototoxic drug reaction.
There are 2 principal mechanisms of photosensitive drug reactions, phototoxic and photoallergic. Phototoxic reactions occur when the drug absorbs UVA light, causing a chemical reaction that produces either free radicals or stable photoproducts, both of which can cause DNA and tissue damage. This mechanism is more common; the time of onset between exposure and reaction is minutes to hours (as seen with this patient), and the reaction manifests with an exaggerated sunburn appearance.1
Photoallergic reactions typically occur by a delayed-immune response, mediated by photoallergic-specific T-cells reacting to photohaptens. This mechanism is less common, and the time of onset is greater than 24 hours. Photoallergic reactions often present as an eczematous dermatitis and can involve regions not directly exposed to sun.
In this case, the presumed offending agent—HCTZ—was discontinued, and the patient was advised to take sun-protective measures and use topical emollients, cool compresses, and oral analgesics for his acute symptoms. Photoallergic reactions look less like a discrete sunburn and can be treated symptomatically with topical or oral steroids. In addition, the implicated medication should be discontinued.
Text courtesy of Amanda Yaney, MD, and Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque. Image courtesy of Daniel Stulberg, MD.
Lozzi F, Di Raimondo C, Lanna C, et al. Latest evidence regarding the effects of photosensitive drugs on the skin: pathogenic mechanisms and clinical manifestations. Pharmaceutics 2020;12:1104. doi: 10.3390/pharmaceutics12111104
The manifestation of what appeared to be an exaggerated sunburn with associated pruritis and blistering (made worse after sun exposure), along with the recent initiation of HCTZ, led to the diagnosis of a phototoxic drug reaction.
There are 2 principal mechanisms of photosensitive drug reactions, phototoxic and photoallergic. Phototoxic reactions occur when the drug absorbs UVA light, causing a chemical reaction that produces either free radicals or stable photoproducts, both of which can cause DNA and tissue damage. This mechanism is more common; the time of onset between exposure and reaction is minutes to hours (as seen with this patient), and the reaction manifests with an exaggerated sunburn appearance.1
Photoallergic reactions typically occur by a delayed-immune response, mediated by photoallergic-specific T-cells reacting to photohaptens. This mechanism is less common, and the time of onset is greater than 24 hours. Photoallergic reactions often present as an eczematous dermatitis and can involve regions not directly exposed to sun.
In this case, the presumed offending agent—HCTZ—was discontinued, and the patient was advised to take sun-protective measures and use topical emollients, cool compresses, and oral analgesics for his acute symptoms. Photoallergic reactions look less like a discrete sunburn and can be treated symptomatically with topical or oral steroids. In addition, the implicated medication should be discontinued.
Text courtesy of Amanda Yaney, MD, and Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque. Image courtesy of Daniel Stulberg, MD.
The manifestation of what appeared to be an exaggerated sunburn with associated pruritis and blistering (made worse after sun exposure), along with the recent initiation of HCTZ, led to the diagnosis of a phototoxic drug reaction.
There are 2 principal mechanisms of photosensitive drug reactions, phototoxic and photoallergic. Phototoxic reactions occur when the drug absorbs UVA light, causing a chemical reaction that produces either free radicals or stable photoproducts, both of which can cause DNA and tissue damage. This mechanism is more common; the time of onset between exposure and reaction is minutes to hours (as seen with this patient), and the reaction manifests with an exaggerated sunburn appearance.1
Photoallergic reactions typically occur by a delayed-immune response, mediated by photoallergic-specific T-cells reacting to photohaptens. This mechanism is less common, and the time of onset is greater than 24 hours. Photoallergic reactions often present as an eczematous dermatitis and can involve regions not directly exposed to sun.
In this case, the presumed offending agent—HCTZ—was discontinued, and the patient was advised to take sun-protective measures and use topical emollients, cool compresses, and oral analgesics for his acute symptoms. Photoallergic reactions look less like a discrete sunburn and can be treated symptomatically with topical or oral steroids. In addition, the implicated medication should be discontinued.
Text courtesy of Amanda Yaney, MD, and Daniel Stulberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque. Image courtesy of Daniel Stulberg, MD.
Lozzi F, Di Raimondo C, Lanna C, et al. Latest evidence regarding the effects of photosensitive drugs on the skin: pathogenic mechanisms and clinical manifestations. Pharmaceutics 2020;12:1104. doi: 10.3390/pharmaceutics12111104
Lozzi F, Di Raimondo C, Lanna C, et al. Latest evidence regarding the effects of photosensitive drugs on the skin: pathogenic mechanisms and clinical manifestations. Pharmaceutics 2020;12:1104. doi: 10.3390/pharmaceutics12111104
Bimekizumab approved in Europe for psoriasis treatment
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
, according to a statement from the manufacturer.
Bimekizumab (Bimzelx), a humanized IgG1 monoclonal antibody, is the first approved treatment for moderate to severe plaque psoriasis that selectively inhibits interleukin (IL)–17A and IL-17F, the statement from UCB said.
In the United States, the Food and Drug Administration is expected to make a decision on approval of bimekizumab for treating psoriasis on Oct. 15.
Approval in the EU was based on data from three phase 3 trials including a total of 1,480 adult patients with moderate to severe psoriasis, which found that those treated with bimekizumab experienced significantly greater skin clearance, compared with placebo, ustekinumab, and adalimumab, with a favorable safety profile, according to the company.
In all three studies (BE VIVID, BE READY, and BE SURE), more than 80% of patients treated with bimekizumab showed improved skin clearance after 16 weeks, significantly more than those treated with ustekinumab, placebo, or adalimumab, based on an improvement of at least 90% in the Psoriasis Area & Severity Index (PASI 90) and an Investigator’s Global Assessment (IGA) response of clear or almost clear skin (IGA 0/1). In all three studies, these clinical responses persisted after 1 year.
The recommended dose of bimekizumab is 320 mg, given in two subcutaneous injections every 4 weeks to week 16, then every 8 weeks. However, for “some patients” weighing 120 kg or more who have not achieved complete skin clearance at 16 weeks, 320 mg every 4 weeks after that time may improve response to treatment, according to the company statement.
The most common treatment-related adverse events in the studies were upper respiratory tract infections (a majority of which were nasopharyngitis), reported by 14.5% of patients, followed by oral candidiasis, reported by 7.3%.
Results of BE READY and BE VIVID were published in The Lancet. Results of the BE SURE study were published in The New England Journal of Medicine.
Bimekizumab is contraindicated for individuals with clinically important active infections such as tuberculosis, and for individuals with any hypersensitivity to the active substance. More details on bimekizumab are available on the website of the European Medicines Agency.
Annular lesions
This patient was given a diagnosis of disseminated granuloma annulare (DGA). While granuloma annulare usually manifests as a single lesion with a raised erythematous border (often with central pallor or hypopigmentation) it can also manifest as multiple large annular lesions on the extremities and occasionally the trunk, as was seen with this case of DGA.
Although the etiology of DGA is unknown, infections including HIV and hepatitis have been reported as possible triggers. Laboratory testing should be considered if the history or physical examination raises suspicion for either condition. Diabetes has also been associated with the disseminated form and the literature suggests a connection with autoimmune diseases of the liver and thyroid.1
Watchful waiting is usually the best treatment for localized disease, which can spontaneously regress within a year and is usually asymptomatic. Intralesional steroid injections into the raised annulus are more effective than topical steroids, as the effects of the topical steroids are sometimes augmented by occlusion.
Multiple treatments have been proposed for DGA, including UVA treatments, systemic retinoids, doxycycline, and hydroxychloroquine.1 Unfortunately, the disseminated form can persist for many years—even with treatment.
Due to the extent of the lesions, the patient in this case was not interested in intralesional steroid injections, and she had already tried topical steroids. She was prescribed topical tacrolimus to reduce the highly visible nature of her lesions.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Beretta-Piccoli BT, Mainetti C, Peeters MA, et al. Cutaneous granulomatosis: a comprehensive review. Clin Rev Allergy Immunol. 2018;54:131-146. doi: 10.1007/s12016-017-8666-8
This patient was given a diagnosis of disseminated granuloma annulare (DGA). While granuloma annulare usually manifests as a single lesion with a raised erythematous border (often with central pallor or hypopigmentation) it can also manifest as multiple large annular lesions on the extremities and occasionally the trunk, as was seen with this case of DGA.
Although the etiology of DGA is unknown, infections including HIV and hepatitis have been reported as possible triggers. Laboratory testing should be considered if the history or physical examination raises suspicion for either condition. Diabetes has also been associated with the disseminated form and the literature suggests a connection with autoimmune diseases of the liver and thyroid.1
Watchful waiting is usually the best treatment for localized disease, which can spontaneously regress within a year and is usually asymptomatic. Intralesional steroid injections into the raised annulus are more effective than topical steroids, as the effects of the topical steroids are sometimes augmented by occlusion.
Multiple treatments have been proposed for DGA, including UVA treatments, systemic retinoids, doxycycline, and hydroxychloroquine.1 Unfortunately, the disseminated form can persist for many years—even with treatment.
Due to the extent of the lesions, the patient in this case was not interested in intralesional steroid injections, and she had already tried topical steroids. She was prescribed topical tacrolimus to reduce the highly visible nature of her lesions.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
This patient was given a diagnosis of disseminated granuloma annulare (DGA). While granuloma annulare usually manifests as a single lesion with a raised erythematous border (often with central pallor or hypopigmentation) it can also manifest as multiple large annular lesions on the extremities and occasionally the trunk, as was seen with this case of DGA.
Although the etiology of DGA is unknown, infections including HIV and hepatitis have been reported as possible triggers. Laboratory testing should be considered if the history or physical examination raises suspicion for either condition. Diabetes has also been associated with the disseminated form and the literature suggests a connection with autoimmune diseases of the liver and thyroid.1
Watchful waiting is usually the best treatment for localized disease, which can spontaneously regress within a year and is usually asymptomatic. Intralesional steroid injections into the raised annulus are more effective than topical steroids, as the effects of the topical steroids are sometimes augmented by occlusion.
Multiple treatments have been proposed for DGA, including UVA treatments, systemic retinoids, doxycycline, and hydroxychloroquine.1 Unfortunately, the disseminated form can persist for many years—even with treatment.
Due to the extent of the lesions, the patient in this case was not interested in intralesional steroid injections, and she had already tried topical steroids. She was prescribed topical tacrolimus to reduce the highly visible nature of her lesions.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Beretta-Piccoli BT, Mainetti C, Peeters MA, et al. Cutaneous granulomatosis: a comprehensive review. Clin Rev Allergy Immunol. 2018;54:131-146. doi: 10.1007/s12016-017-8666-8
Beretta-Piccoli BT, Mainetti C, Peeters MA, et al. Cutaneous granulomatosis: a comprehensive review. Clin Rev Allergy Immunol. 2018;54:131-146. doi: 10.1007/s12016-017-8666-8
What’s under my toenail?
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
A 13-year-old female was seen by her pediatrician for a lesion that had been on her right toe for about 6 months. She is unaware of any trauma to the area. The lesion has been growing slowly and recently it started lifting up the nail, became tender, and was bleeding, which is the reason why she sought care.
At the pediatrician's office, he noted a pink crusted papule under the nail. The nail was lifting up and was tender to the touch. She is a healthy girl who is not taking any medications and has no allergies. There is no family history of similar lesions.
The pediatrician took a picture of the lesion and he send it to our pediatric teledermatology service for consultation.