User login
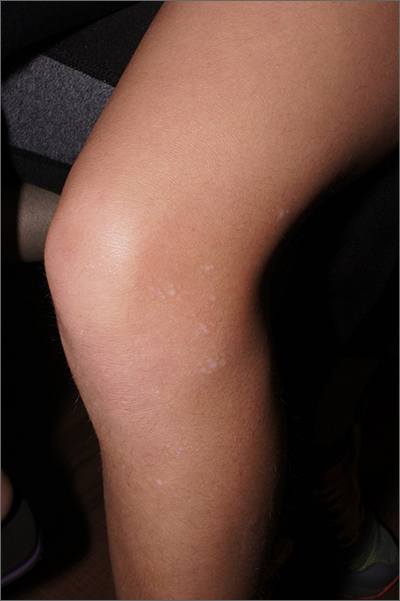
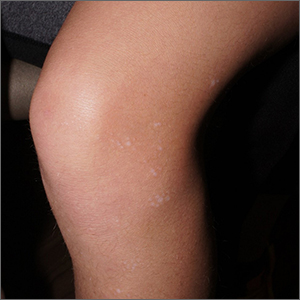
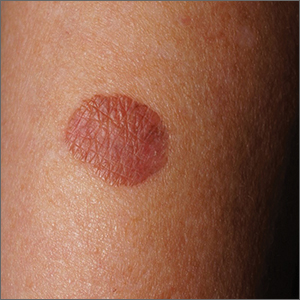
White macules on knee
The ivory white appearance and slight atrophy of the lesions raised the possibility of extragenital lichen sclerosus (LS). A 4-mm punch biopsy confirmed the diagnosis.
LS occurs in all races and is an uncommon, chronic inflammatory disease that most often affects the vulva and perianal mucosa in postmenopausal women.1 That said, it can also affect men and children, and manifest in places such as the trunk and neck. Extragenital lesions may appear ivory white, as in this case, or may resemble ecchymoses and raise alarm for possible abuse.
When LS is present on the extremities, a complete skin surface exam, including external genitalia, is warranted. LS is thought to be an autoimmune disease and is associated with vitiligo, autoimmune thyroid disease, and morphea.
In cases of suspected LS, it’s important to biopsy the full thickness of the skin and subcutaneous tissue. It is helpful to include an area of normal skin in the sample, as the findings are subtle and best contrasted with the architecture of unaffected skin. For this patient, a 4-mm punch biopsy was sufficient, but an incisional biopsy would be more appropriate for a larger patch or plaque.
Treatment options are based on a small case series and a few small randomized controlled trials. Medications include topical steroids, topical calcineurin inhibitors, systemic retinoids, and topical estrogens.
In this case, the patient was advised to apply topical clobetasol 0.05% cream bid to the affected area for 2 weeks, then twice weekly for 4 weeks. She had partial clearance with this approach, but small macules later appeared on her dorsal foot; the treatment was repeated.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
1. Tong LX, Sun GS, Teng JMC. Pediatric lichen sclerosus: a review of the epidemiology and treatment options. Pediatr Dermatol. 2015;32:593-599. doi: 10.1111/pde.12615
The ivory white appearance and slight atrophy of the lesions raised the possibility of extragenital lichen sclerosus (LS). A 4-mm punch biopsy confirmed the diagnosis.
LS occurs in all races and is an uncommon, chronic inflammatory disease that most often affects the vulva and perianal mucosa in postmenopausal women.1 That said, it can also affect men and children, and manifest in places such as the trunk and neck. Extragenital lesions may appear ivory white, as in this case, or may resemble ecchymoses and raise alarm for possible abuse.
When LS is present on the extremities, a complete skin surface exam, including external genitalia, is warranted. LS is thought to be an autoimmune disease and is associated with vitiligo, autoimmune thyroid disease, and morphea.
In cases of suspected LS, it’s important to biopsy the full thickness of the skin and subcutaneous tissue. It is helpful to include an area of normal skin in the sample, as the findings are subtle and best contrasted with the architecture of unaffected skin. For this patient, a 4-mm punch biopsy was sufficient, but an incisional biopsy would be more appropriate for a larger patch or plaque.
Treatment options are based on a small case series and a few small randomized controlled trials. Medications include topical steroids, topical calcineurin inhibitors, systemic retinoids, and topical estrogens.
In this case, the patient was advised to apply topical clobetasol 0.05% cream bid to the affected area for 2 weeks, then twice weekly for 4 weeks. She had partial clearance with this approach, but small macules later appeared on her dorsal foot; the treatment was repeated.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
The ivory white appearance and slight atrophy of the lesions raised the possibility of extragenital lichen sclerosus (LS). A 4-mm punch biopsy confirmed the diagnosis.
LS occurs in all races and is an uncommon, chronic inflammatory disease that most often affects the vulva and perianal mucosa in postmenopausal women.1 That said, it can also affect men and children, and manifest in places such as the trunk and neck. Extragenital lesions may appear ivory white, as in this case, or may resemble ecchymoses and raise alarm for possible abuse.
When LS is present on the extremities, a complete skin surface exam, including external genitalia, is warranted. LS is thought to be an autoimmune disease and is associated with vitiligo, autoimmune thyroid disease, and morphea.
In cases of suspected LS, it’s important to biopsy the full thickness of the skin and subcutaneous tissue. It is helpful to include an area of normal skin in the sample, as the findings are subtle and best contrasted with the architecture of unaffected skin. For this patient, a 4-mm punch biopsy was sufficient, but an incisional biopsy would be more appropriate for a larger patch or plaque.
Treatment options are based on a small case series and a few small randomized controlled trials. Medications include topical steroids, topical calcineurin inhibitors, systemic retinoids, and topical estrogens.
In this case, the patient was advised to apply topical clobetasol 0.05% cream bid to the affected area for 2 weeks, then twice weekly for 4 weeks. She had partial clearance with this approach, but small macules later appeared on her dorsal foot; the treatment was repeated.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
1. Tong LX, Sun GS, Teng JMC. Pediatric lichen sclerosus: a review of the epidemiology and treatment options. Pediatr Dermatol. 2015;32:593-599. doi: 10.1111/pde.12615
1. Tong LX, Sun GS, Teng JMC. Pediatric lichen sclerosus: a review of the epidemiology and treatment options. Pediatr Dermatol. 2015;32:593-599. doi: 10.1111/pde.12615
Making the Rounds to Diagnosis
ANSWER
The correct answer is granuloma annulare (GA; choice “d”).
DISCUSSION
The biopsy showed rings of dermal epithelioid histiocytes surrounding a core of central mucin, configured in rows (the latter conferring the diagnostic term “palisaded” granulomas). Both the pathology results and the morphology of the lesions—color, shape, etc—were classic for GA. What was somewhat unusual about this case was the size and number of lesions, which are typically fewer and smaller with GA.
GA is seldom much of a problem and has no connection to serious disease. But it can, as this case illustrates, mimic some rather worrisome conditions. Had this been sarcoidosis (choice “a”), there would be no central “delling” (concavity), and the biopsy would have shown necrotic (caseating) nonpalisading granulomas.
Cutaneous T-cell lymphoma (CTCL; choice “b”) can manifest with plaques, but there wouldn’t be any delling and the shapes would not be so consistently round. CTCL will eventually give rise to palpable adenopathy.
Lupus profundus (choice “c”) also manifests with deep plaques, without delling. It is a truly rare variant of lupus, which would have been detected on biopsy.
Concern about these differential items is what drove the decision to perform full-thickness punch biopsies as opposed to taking a simple shave sample.
TREATMENT
The most common treatment for GA is intralesional steroid injection (eg, 10 mg/cc triamcinolone) of large lesions. Oral medications such as methotrexate or pentoxifylline have been used with some success. Often, the condition is self-limiting.
ANSWER
The correct answer is granuloma annulare (GA; choice “d”).
DISCUSSION
The biopsy showed rings of dermal epithelioid histiocytes surrounding a core of central mucin, configured in rows (the latter conferring the diagnostic term “palisaded” granulomas). Both the pathology results and the morphology of the lesions—color, shape, etc—were classic for GA. What was somewhat unusual about this case was the size and number of lesions, which are typically fewer and smaller with GA.
GA is seldom much of a problem and has no connection to serious disease. But it can, as this case illustrates, mimic some rather worrisome conditions. Had this been sarcoidosis (choice “a”), there would be no central “delling” (concavity), and the biopsy would have shown necrotic (caseating) nonpalisading granulomas.
Cutaneous T-cell lymphoma (CTCL; choice “b”) can manifest with plaques, but there wouldn’t be any delling and the shapes would not be so consistently round. CTCL will eventually give rise to palpable adenopathy.
Lupus profundus (choice “c”) also manifests with deep plaques, without delling. It is a truly rare variant of lupus, which would have been detected on biopsy.
Concern about these differential items is what drove the decision to perform full-thickness punch biopsies as opposed to taking a simple shave sample.
TREATMENT
The most common treatment for GA is intralesional steroid injection (eg, 10 mg/cc triamcinolone) of large lesions. Oral medications such as methotrexate or pentoxifylline have been used with some success. Often, the condition is self-limiting.
ANSWER
The correct answer is granuloma annulare (GA; choice “d”).
DISCUSSION
The biopsy showed rings of dermal epithelioid histiocytes surrounding a core of central mucin, configured in rows (the latter conferring the diagnostic term “palisaded” granulomas). Both the pathology results and the morphology of the lesions—color, shape, etc—were classic for GA. What was somewhat unusual about this case was the size and number of lesions, which are typically fewer and smaller with GA.
GA is seldom much of a problem and has no connection to serious disease. But it can, as this case illustrates, mimic some rather worrisome conditions. Had this been sarcoidosis (choice “a”), there would be no central “delling” (concavity), and the biopsy would have shown necrotic (caseating) nonpalisading granulomas.
Cutaneous T-cell lymphoma (CTCL; choice “b”) can manifest with plaques, but there wouldn’t be any delling and the shapes would not be so consistently round. CTCL will eventually give rise to palpable adenopathy.
Lupus profundus (choice “c”) also manifests with deep plaques, without delling. It is a truly rare variant of lupus, which would have been detected on biopsy.
Concern about these differential items is what drove the decision to perform full-thickness punch biopsies as opposed to taking a simple shave sample.
TREATMENT
The most common treatment for GA is intralesional steroid injection (eg, 10 mg/cc triamcinolone) of large lesions. Oral medications such as methotrexate or pentoxifylline have been used with some success. Often, the condition is self-limiting.
A 60-year-old woman is feeling fine—no fever, arthralgia, or malaise—but the sudden appearance of lesions on her extremities unsettles her to the point that she can think of little else. Visits to her primary care provider, the emergency department, several urgent care clinics, and a naturopath yield a consistent diagnosis: ringworm. Yet none of the topical or oral antifungal medications she is prescribed make the slightest difference. Thus, she finally agrees to consult a dermatologist.
The patient reports decent health, although she was a heavy smoker for years before quitting 2 years ago. She recently tested negative for diabetes.
History taking reveals no sources from which she could have acquired a fungal infection. No one else at home is similarly affected.
About 10 lesions, mostly located on the patient’s extremities, are examined. All are very similar in appearance: round, brownish-red intradermal plaques with no epidermal component (eg, scale, broken skin). The lesions vary from 6 mm to 4 cm in diameter. The centers of most lesions are slightly concave, with well-defined raised margins. On palpation, there is neither increased warmth nor tenderness. No nodes can be palpated in the groin, axillae, or epitrochlear locations.
A deep punch biopsy is performed on a thigh lesion, with the specimen submitted for pathologic examination.
Genital skin exams in girls: Conduct with care, look for signs of abuse
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
at the American Academy of Dermatology Virtual Meeting Experience.
“One in four adult women report being childhood victims of sexual abuse, which is just a staggering number. This is an opportunity for us to identify these patients early and give them the terminology to be able to report what is happening to them,” said pediatric dermatologist Kalyani Marathe, MD, MPH, director of the division of dermatology at Cincinnati Children’s Hospital. “We also have the chance to give them a sense of agency over their bodies.”
Dr. Marathe offered the following recommendations when performing a genital skin exam:
- Make sure a “chaperone” is present. “Chaperones are a must when you’re examining children and teens,” she said. “Ask whom they prefer. For prepubertal children, you’re going to usually use the parent who’s there with them. If the parent is their father, they might ask him to step behind the curtain, in which case you can bring over your nurse or medical assistant.” Teens may ask either parent to step out of the room, she said. In that case, a nurse, medical assistant, resident, or trainee can fill in. “If you have male residents or trainees with you and the patient really does not want to be examined by a male, honor their request. Do not force them.”
- Explain why the exam is being performed. Make sure the patient understands why she is being seen, Dr. Marathe advised. For example, say something like “your pediatrician told us that you have an itchy area” or “your mom told us that there’s some loss of color in that area, that you’re having a problem there.” She added that it’s helpful to explain the type of doctor you are, with a comment such as the following: “We’re examining you because we’re doctors who specialize in skin. ... We want to help you feel better and make sure that your skin heals and is healthy.”
- Ask both the child and the parent for permission to perform the exam. While this may seem trivial, “it’s very, very important in setting the right tone for the encounter,” she said. “If the child says yes, we turn to the mom and say: ‘Mom, is it okay for us to do this exam today?’ You can see visible relief on the part of the parent, and as the parent relaxes, the child relaxes. Just saying those few things really makes the encounter so much smoother.” However, “if they say no, you have to honor the response. ... You say: ‘Okay, we’re not going to do the exam today,” and see the patient in a few weeks. If it’s urgent, an exam under anesthesia may be an option, she added.
- Talk to the child about the terms they use for private parts. It can be helpful to ask: “Do you have any terms for your private area?” According to Dr. Marathe, “this is a good chance to educate them on the terms vulva and vagina since they may be using other terminology. Making sure that they have the correct terms will actually help patients identify and report abuse earlier.” Dr. Marathe recalled that a colleague had a patient who’d been calling her private area “pound cake” and had been “reporting to her teacher that someone had been touching her ‘pound cake.’ Her teacher did not know what she meant by that, and this led to a great delay in her childhood abuse being reported.”
- Talk about what will happen during the exam. “I like to show them any instruments that we’re going to be using,” Dr. Marathe said. “If we’re using a flashlight, for example, I like to show them a picture [of a flashlight] or show them that flashlight. If we’re using a camera to do digital photography, show them that. If we’re going to be using a Q-tip or a swab to demonstrate anything or to take a culture, I like to show them that beforehand to make sure that they know what we’re doing.” In regard to photography, “make sure the parent and child know where the photos are going to go, who’s going to see them, what are they going to be used for. If they’re going to be used for educational purposes, make sure they have given explicit permission for that and they know they’ll be deidentified.”
- Make it clear that the exam won’t be painful. It’s important to put both the patient and the parent at ease on this front, Dr. Marathe said. “A lot of parents are concerned that we’re going to do a speculum exam in their prepubertal child. So make sure that it’s clarified ahead of time that we’re not going to be doing a speculum exam.”
Commenting on this topic, Tor Shwayder, MD, a pediatric dermatologist at Henry Ford Health System, Detroit, urged colleagues to take action if they feel suspicious about a possible sign of child abuse, even if they’re far from certain that anything is wrong. “Don’t ignore those feelings in the back of the brain,” he said in an interview.
Most states have child-abuse hotlines for medical professionals, and major hospitals will have child-abuse teams, Dr. Shwayder said. He urged dermatologists to take advantage of these resources when appropriate. “The professionals on the other side of the 800 number or at the hospital will help you. You don’t have to decide immediately whether this is child abuse. You just need to have a suspicion.”
Dr. Marathe and Dr. Shwayder report no disclosures.
FROM AAD VMX 2021
AAD unveils new guidelines for actinic keratosis management
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
FROM JAAD
Atopic dermatitis genes vary with ethnicity
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
Checkpoint inhibitor skin side effects more common in women
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
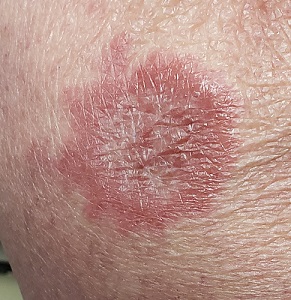
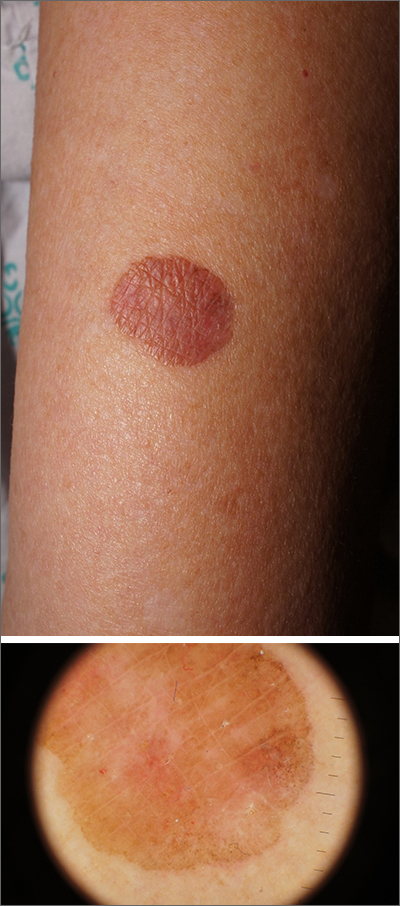
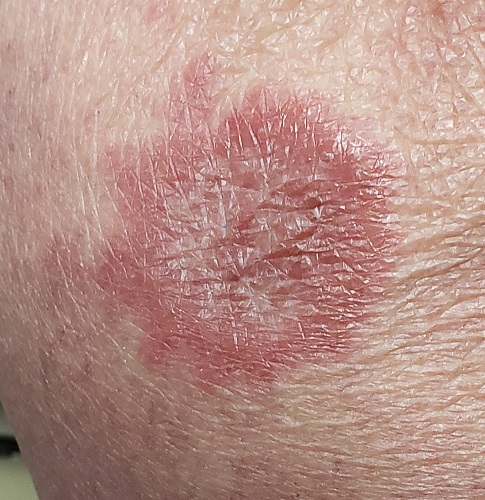
Brown plaque on the arm
Due to its size, 2 shave biopsies targeting the most concerning portions of the lesion were performed; the results were consistent with a lichenoid keratosis (LK), also known as lichen planus-like keratosis.
LK is a benign solitary lesion that mimics basal cell carcinoma, squamous cell carcinoma, and superficial spreading or amelanotic melanoma.1 One theory suggests that LK is a solar lentigo or actinic keratosis undergoing attack from the immune system. Lesions most often manifest as a pink, gray, or brown macule to thin papule on the trunk or extremities. Itching or mild pain may be present. Dermoscopy can help distinguish an LK from malignancy but overlapping features of fine dark regression structures (called peppering, as seen in this case) should prompt further evaluation.
LKs are great mimics and biopsy is key to distinguishing them from cancer. In this case, shave biopsies were performed in the thickest and most characteristic portions of the lesion. Punch or incisional biopsies also would have been appropriate, but any result would have been a partial result. If the result had come back as an atypical melanocytic lesion, a complete excision would have been necessary to make sure the pathology reflected the entirety of the lesion.
Armed with the knowledge that the LK was benign, the patient in this case was scheduled for a follow-up visit for cryotherapy to remove the residual lesion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Maor D, Ondhia C, Yu LL, et al. Lichenoid keratosis is frequently misdiagnosed as basal cell carcinoma. Clin Exp Dermatol. 2017;42:663-666. doi: 10.1111/ced.13178
Due to its size, 2 shave biopsies targeting the most concerning portions of the lesion were performed; the results were consistent with a lichenoid keratosis (LK), also known as lichen planus-like keratosis.
LK is a benign solitary lesion that mimics basal cell carcinoma, squamous cell carcinoma, and superficial spreading or amelanotic melanoma.1 One theory suggests that LK is a solar lentigo or actinic keratosis undergoing attack from the immune system. Lesions most often manifest as a pink, gray, or brown macule to thin papule on the trunk or extremities. Itching or mild pain may be present. Dermoscopy can help distinguish an LK from malignancy but overlapping features of fine dark regression structures (called peppering, as seen in this case) should prompt further evaluation.
LKs are great mimics and biopsy is key to distinguishing them from cancer. In this case, shave biopsies were performed in the thickest and most characteristic portions of the lesion. Punch or incisional biopsies also would have been appropriate, but any result would have been a partial result. If the result had come back as an atypical melanocytic lesion, a complete excision would have been necessary to make sure the pathology reflected the entirety of the lesion.
Armed with the knowledge that the LK was benign, the patient in this case was scheduled for a follow-up visit for cryotherapy to remove the residual lesion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Due to its size, 2 shave biopsies targeting the most concerning portions of the lesion were performed; the results were consistent with a lichenoid keratosis (LK), also known as lichen planus-like keratosis.
LK is a benign solitary lesion that mimics basal cell carcinoma, squamous cell carcinoma, and superficial spreading or amelanotic melanoma.1 One theory suggests that LK is a solar lentigo or actinic keratosis undergoing attack from the immune system. Lesions most often manifest as a pink, gray, or brown macule to thin papule on the trunk or extremities. Itching or mild pain may be present. Dermoscopy can help distinguish an LK from malignancy but overlapping features of fine dark regression structures (called peppering, as seen in this case) should prompt further evaluation.
LKs are great mimics and biopsy is key to distinguishing them from cancer. In this case, shave biopsies were performed in the thickest and most characteristic portions of the lesion. Punch or incisional biopsies also would have been appropriate, but any result would have been a partial result. If the result had come back as an atypical melanocytic lesion, a complete excision would have been necessary to make sure the pathology reflected the entirety of the lesion.
Armed with the knowledge that the LK was benign, the patient in this case was scheduled for a follow-up visit for cryotherapy to remove the residual lesion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Maor D, Ondhia C, Yu LL, et al. Lichenoid keratosis is frequently misdiagnosed as basal cell carcinoma. Clin Exp Dermatol. 2017;42:663-666. doi: 10.1111/ced.13178
1. Maor D, Ondhia C, Yu LL, et al. Lichenoid keratosis is frequently misdiagnosed as basal cell carcinoma. Clin Exp Dermatol. 2017;42:663-666. doi: 10.1111/ced.13178
Multidisciplinary approach touted for atopic dermatitis
researchers say.
“I think we really gained insight to how a more holistic approach benefited the patient,” Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said in an interview.
At the 2021 annual meeting of the International Society of Atopic Dermatitis, he and his colleagues described a pilot program to bring the specialists together at UCSD and Rady Children’s Hospital, San Diego.
Typically, children seeking care for atopic dermatitis see allergists and dermatologists separately for 10- to 15-minute appointments. The specialists sometimes prescribe treatments that conflict or are redundant with each other and may give contradictory instructions.
Instead, Dr. Eichenfield and colleagues designed a program bringing patients in for initial assessments lasting 1-1.5 hours. Patients typically started with visits to a clinical pharmacist, who assessed what medications had been prescribed and how much the patients were actually taking.
The patients then proceeded to separate appointments with an allergist and a dermatologist for evaluations. These specialists then met face to face to develop a treatment plan. At least one of the specialists would then present the plan to the patient and the patient’s family.
“We had a rich set of educational materials that were developed and put online that helped with shared decision-making and increased comfort level with appropriate skin care and medication,” Dr. Eichenfield said.
He and his colleagues assigned a physician assistant trained in both pediatric dermatology and pediatric allergy to coordinate the clinic. They designed combined pediatric dermatology and pediatric allergy fellowships for two fellows. “So, part of this program ended up allowing specially trained individuals who overlapped in fields that traditionally were separate,” said Dr. Eichenfield.
To see how well the approach worked, the researchers followed the progress of 23 patients who were already receiving treatment at one or both of the institutions.
- Eczema Area and Severity Index (EASI) scores decreased from visit 1 to visit 2 by a mean of 15.36 (P < .001), which correlates to a 56.36% average decrease.
- In 20 patients (89.96%), in EASI scores improved 50%.
- Thirteen patients (56.54%) achieved 75% improvement in EASI scores.
- Body surface area scores improved by a mean of 23.21% (P = .002).
- Validated Investigator Global Assessment scores decreased in 56.52% of patients to a clinically significant level.
The study did not include any control group, nor did the researchers report any details on how long the patients had been treated before the multidisciplinary program started or how their prescriptions changed.
Patients benefited from the comprehensive assessment of their symptoms, said Dr. Eichenfield, also chief of pediatric and adolescent dermatology at Rady Children’s Hospital. “Some had significant environmental allergies that might not have been a contributing factor to their atopic dermatitis,” he explained. “The complexities of comorbidities and atopic dermatitis influence the patient, even if one disease state isn’t necessarily directly causative of the other.”
In surveys, patients said they especially appreciated the increased time spent with their specialists. “No one’s ever spent an hour teaching us about eczema,” some commented. The approach motivated patients to take their home treatment more effectively, Dr. Eichenfield believed.
Primary care physicians did not participate in the multidisciplinary program, but the specialists communicated with them and shared electronic medical records with them, he said.
Without a control group, it is hard to say how much difference the multidisciplinary approach made, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview.
“What it does show is that there is significant improvement in a variety of endpoints within this multidisciplinary approach,” Dr. Silverberg said in an interview. “And so I have no doubt that this is valid and that a multidisciplinary approach would really improve, holistically, many aspects of patient care.”
Dr. Silverberg ran a multidisciplinary program at Northwestern University, Chicago, which included sleep medicine, endocrinology, gastroenterology, and other specialties as well as dermatology, allergy, and pharmacy.
However, Dr. Silverberg pointed out, a multidisciplinary approach is more expensive than standard care because when specialists spend more time with each patient, they see fewer patients per day. “So many health care systems or academic institutions are not as open as they should be to this kind of interdisciplinary care, which is why it’s so important to have outcome measures showing that this approach actually works.”
Dr. Eichenfield and Dr. Silverberg had no relevant disclosures.
A version of this article first appeared on Medscape.com.
researchers say.
“I think we really gained insight to how a more holistic approach benefited the patient,” Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said in an interview.
At the 2021 annual meeting of the International Society of Atopic Dermatitis, he and his colleagues described a pilot program to bring the specialists together at UCSD and Rady Children’s Hospital, San Diego.
Typically, children seeking care for atopic dermatitis see allergists and dermatologists separately for 10- to 15-minute appointments. The specialists sometimes prescribe treatments that conflict or are redundant with each other and may give contradictory instructions.
Instead, Dr. Eichenfield and colleagues designed a program bringing patients in for initial assessments lasting 1-1.5 hours. Patients typically started with visits to a clinical pharmacist, who assessed what medications had been prescribed and how much the patients were actually taking.
The patients then proceeded to separate appointments with an allergist and a dermatologist for evaluations. These specialists then met face to face to develop a treatment plan. At least one of the specialists would then present the plan to the patient and the patient’s family.
“We had a rich set of educational materials that were developed and put online that helped with shared decision-making and increased comfort level with appropriate skin care and medication,” Dr. Eichenfield said.
He and his colleagues assigned a physician assistant trained in both pediatric dermatology and pediatric allergy to coordinate the clinic. They designed combined pediatric dermatology and pediatric allergy fellowships for two fellows. “So, part of this program ended up allowing specially trained individuals who overlapped in fields that traditionally were separate,” said Dr. Eichenfield.
To see how well the approach worked, the researchers followed the progress of 23 patients who were already receiving treatment at one or both of the institutions.
- Eczema Area and Severity Index (EASI) scores decreased from visit 1 to visit 2 by a mean of 15.36 (P < .001), which correlates to a 56.36% average decrease.
- In 20 patients (89.96%), in EASI scores improved 50%.
- Thirteen patients (56.54%) achieved 75% improvement in EASI scores.
- Body surface area scores improved by a mean of 23.21% (P = .002).
- Validated Investigator Global Assessment scores decreased in 56.52% of patients to a clinically significant level.
The study did not include any control group, nor did the researchers report any details on how long the patients had been treated before the multidisciplinary program started or how their prescriptions changed.
Patients benefited from the comprehensive assessment of their symptoms, said Dr. Eichenfield, also chief of pediatric and adolescent dermatology at Rady Children’s Hospital. “Some had significant environmental allergies that might not have been a contributing factor to their atopic dermatitis,” he explained. “The complexities of comorbidities and atopic dermatitis influence the patient, even if one disease state isn’t necessarily directly causative of the other.”
In surveys, patients said they especially appreciated the increased time spent with their specialists. “No one’s ever spent an hour teaching us about eczema,” some commented. The approach motivated patients to take their home treatment more effectively, Dr. Eichenfield believed.
Primary care physicians did not participate in the multidisciplinary program, but the specialists communicated with them and shared electronic medical records with them, he said.
Without a control group, it is hard to say how much difference the multidisciplinary approach made, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview.
“What it does show is that there is significant improvement in a variety of endpoints within this multidisciplinary approach,” Dr. Silverberg said in an interview. “And so I have no doubt that this is valid and that a multidisciplinary approach would really improve, holistically, many aspects of patient care.”
Dr. Silverberg ran a multidisciplinary program at Northwestern University, Chicago, which included sleep medicine, endocrinology, gastroenterology, and other specialties as well as dermatology, allergy, and pharmacy.
However, Dr. Silverberg pointed out, a multidisciplinary approach is more expensive than standard care because when specialists spend more time with each patient, they see fewer patients per day. “So many health care systems or academic institutions are not as open as they should be to this kind of interdisciplinary care, which is why it’s so important to have outcome measures showing that this approach actually works.”
Dr. Eichenfield and Dr. Silverberg had no relevant disclosures.
A version of this article first appeared on Medscape.com.
researchers say.
“I think we really gained insight to how a more holistic approach benefited the patient,” Lawrence Eichenfield, MD, professor of dermatology and pediatrics at the University of California, San Diego, said in an interview.
At the 2021 annual meeting of the International Society of Atopic Dermatitis, he and his colleagues described a pilot program to bring the specialists together at UCSD and Rady Children’s Hospital, San Diego.
Typically, children seeking care for atopic dermatitis see allergists and dermatologists separately for 10- to 15-minute appointments. The specialists sometimes prescribe treatments that conflict or are redundant with each other and may give contradictory instructions.
Instead, Dr. Eichenfield and colleagues designed a program bringing patients in for initial assessments lasting 1-1.5 hours. Patients typically started with visits to a clinical pharmacist, who assessed what medications had been prescribed and how much the patients were actually taking.
The patients then proceeded to separate appointments with an allergist and a dermatologist for evaluations. These specialists then met face to face to develop a treatment plan. At least one of the specialists would then present the plan to the patient and the patient’s family.
“We had a rich set of educational materials that were developed and put online that helped with shared decision-making and increased comfort level with appropriate skin care and medication,” Dr. Eichenfield said.
He and his colleagues assigned a physician assistant trained in both pediatric dermatology and pediatric allergy to coordinate the clinic. They designed combined pediatric dermatology and pediatric allergy fellowships for two fellows. “So, part of this program ended up allowing specially trained individuals who overlapped in fields that traditionally were separate,” said Dr. Eichenfield.
To see how well the approach worked, the researchers followed the progress of 23 patients who were already receiving treatment at one or both of the institutions.
- Eczema Area and Severity Index (EASI) scores decreased from visit 1 to visit 2 by a mean of 15.36 (P < .001), which correlates to a 56.36% average decrease.
- In 20 patients (89.96%), in EASI scores improved 50%.
- Thirteen patients (56.54%) achieved 75% improvement in EASI scores.
- Body surface area scores improved by a mean of 23.21% (P = .002).
- Validated Investigator Global Assessment scores decreased in 56.52% of patients to a clinically significant level.
The study did not include any control group, nor did the researchers report any details on how long the patients had been treated before the multidisciplinary program started or how their prescriptions changed.
Patients benefited from the comprehensive assessment of their symptoms, said Dr. Eichenfield, also chief of pediatric and adolescent dermatology at Rady Children’s Hospital. “Some had significant environmental allergies that might not have been a contributing factor to their atopic dermatitis,” he explained. “The complexities of comorbidities and atopic dermatitis influence the patient, even if one disease state isn’t necessarily directly causative of the other.”
In surveys, patients said they especially appreciated the increased time spent with their specialists. “No one’s ever spent an hour teaching us about eczema,” some commented. The approach motivated patients to take their home treatment more effectively, Dr. Eichenfield believed.
Primary care physicians did not participate in the multidisciplinary program, but the specialists communicated with them and shared electronic medical records with them, he said.
Without a control group, it is hard to say how much difference the multidisciplinary approach made, Jonathan I. Silverberg, MD, PhD, MPH, associate professor of dermatology and director of clinical research and contact dermatitis at George Washington University, Washington, said in an interview.
“What it does show is that there is significant improvement in a variety of endpoints within this multidisciplinary approach,” Dr. Silverberg said in an interview. “And so I have no doubt that this is valid and that a multidisciplinary approach would really improve, holistically, many aspects of patient care.”
Dr. Silverberg ran a multidisciplinary program at Northwestern University, Chicago, which included sleep medicine, endocrinology, gastroenterology, and other specialties as well as dermatology, allergy, and pharmacy.
However, Dr. Silverberg pointed out, a multidisciplinary approach is more expensive than standard care because when specialists spend more time with each patient, they see fewer patients per day. “So many health care systems or academic institutions are not as open as they should be to this kind of interdisciplinary care, which is why it’s so important to have outcome measures showing that this approach actually works.”
Dr. Eichenfield and Dr. Silverberg had no relevant disclosures.
A version of this article first appeared on Medscape.com.
LGBTQ patients face unique skin risks
Dermatologists cautioned colleagues to in transgender people, who are especially vulnerable to acne because of hormone therapy.
The identities of sexual minorities “have a significant influence on many facets of health,” dermatologist Matthew Mansh, MD, of the University of Minnesota, Minneapolis, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
In regard to skin cancer, he said, “there seems to be consistently higher rates of skin cancer and certain preventable risk behaviors like indoor tanning among sexual minority men.”
Dr. Mansh, codirector of the high-risk nonmelanoma skin cancer clinic at the University of Minnesota, highlighted a report, published in JAMA Dermatology in 2020, that used 2014-2018 U.S. survey data of over 870,000 adults to look at the association between sexual orientation and lifetime prevalence of skin cancer. The investigators found that gay and bisexual men had a higher lifetime prevalence of skin cancer compared with heterosexual men (adjusted odds ratio [aOR], 1.25; 95% confidence interval, 1.03-1.50; P = .02; and aOR, 1.46; 95% CI, 1.01-2.10; P = .04; for gay and bisexual men, respectively).
When compared with heterosexual women, risk among bisexual women was lower (aOR, 0.75; 95% CI, 0.60-0.95; P = .02), but not among lesbian women (aOR, 1.01; 95% CI, 0.77-1.33; P = .95, respectively).
Other studies have reached similar conclusions, Dr. Mansh said, although there’s been fairly little research in this area. What could explain these differences? Factors such as smoking, age, and alcohol use affect skin cancer risk, he said, but these studies control for those variables. Instead, he noted, it’s useful to look at studies of ultraviolet exposure.
For example, he highlighted a study published in JAMA Dermatology in 2015, which examined 12-month indoor-tanning rates and skin cancer prevalence by sexual orientation, using data from California and national health interview surveys. The study found that compared with heterosexual men, “sexual minority men had higher rates of indoor tanning by roughly three- to sixfold,” said Dr. Mansh, the lead author. “And this was among respondents who were adults over age 18. People between the ages of 18 and 34 years are important from a skin cancer perspective as it’s well established that exposure to tanning beds at a younger age is most associated with an increased risk of skin cancer.”
Sexual minority men were also significantly more likely to report having skin cancer, compared with heterosexual men.
In the study, sexual minority women had about half the odds of engaging in indoor tanning compared with heterosexual women, and were less likely to report having been diagnosed with nonmelanoma skin cancer, he added.
Other studies suggest that gay and bisexual men live in neighborhoods with more indoor tanning salons and that they may spend more time in the sun outside too, he said. Some research suggests motivations for tanning include social pressure and the desire to improve appearance, he added.
Overall, “we may be able to use these data to add more appropriate screening and recommendations for these patients, which are sorely lacking in dermatology,” and to design targeted behavioral interventions, said Dr. Mansh, codirector of the dermatology gender care clinic at the University of Minnesota.
What can dermatologists do now? In an interview, dermatologist Jon Klint Peebles, MD, of the mid-Atlantic Permanente Medical Group, in Largo, Md., suggested that colleagues ask patients questions about indoor tanning frequency, the motivations for tanning, exposure to outdoor ultraviolet radiation, sunscreen use, and use of photoprotective clothing.
Hormone therapy and acne
In a related presentation at the meeting, Howa Yeung, MD, of the department of dermatology, Emory University, Atlanta, said that in transgender people, estrogen therapy can actually reduce sebum production and often improves acne, while testosterone therapy frequently has the opposite effect.
“We’ve seen some pretty tough cases of acne in transmasculine patients in my practice,” said Dr. Yeung, who highlighted a recently published study that tracked 988 transgender patients in Boston who underwent testosterone therapy. Nearly a third were diagnosed with acne, compared with 6% prior to hormone therapy, and those at the highest risk were aged 18-21.
The prevalence of acne was 25% 2 years after initiation of hormone therapy. “Acne remains a very common issue and not just at the beginning of treatment,” he said.
In 2020, Dr. Yeung and colleagues reported the results of a survey of 696 transgender patients in California and Georgia; most were treated with hormone therapy. They found that 14% of transmasculine patients reported currently having moderate to severe acne diagnosed by a physician, compared with 1% of transfeminine patients.
Dr. Yeung noted that another survey of transmasculine persons who had received testosterone found that those who had moderate to severe acne were more likely to suffer from depression and anxiety than were those who had never had acne (aOR, 2.4; 95% CI, 1.1-5.4; P = .001, for depression; and aOR, 2.7; 95% CI, 1.2-6.3; P = .002, for anxiety).
Acne treatments in transmasculine patients are complicated by the fact that hormone treatments for acne can have feminizing effects, Dr. Yeung said, adding that it’s not clear how clascoterone, a new anti-androgen topical therapy for acne, will affect them. For now, many patients will require isotretinoin for treating acne.
Dr. Peebles cautioned that with isotretinoin, “we still do not yet have solid data on the optimal dosing or duration in the context of testosterone-induced acne, as well as what individual factors may be predictive of treatment success or failure. It is also important to be aware of any planned surgical procedures, whether as part of gender-affirming care or otherwise, given that some surgeons may view isotretinoin as a barrier for some procedures, despite limited data to support this.”
Both Dr. Peebles and Dr. Yeung noted that the iPledge risk management program for isotretinoin patients who may become pregnant is problematic. “A trans man who is assigned female at birth and identifies as a man and has a uterus and ovaries must be registered as a female with reproductive potential,” Dr. Yeung said.
“While the program remains inherently discriminatory, it is important to have an honest conversation with patients about these issues in a sensitive way,” Dr. Peebles noted. “Luckily, there is substantial momentum building around modifying iPLEDGE to become more inclusive. While the mechanics are complicated and involve a variety of entities and advocacy initiatives, we are optimistic that major changes are in the pipeline.”
Dr. Mansh, Dr. Yeung, and Dr. Peebles reported no disclosures.
Dermatologists cautioned colleagues to in transgender people, who are especially vulnerable to acne because of hormone therapy.
The identities of sexual minorities “have a significant influence on many facets of health,” dermatologist Matthew Mansh, MD, of the University of Minnesota, Minneapolis, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
In regard to skin cancer, he said, “there seems to be consistently higher rates of skin cancer and certain preventable risk behaviors like indoor tanning among sexual minority men.”
Dr. Mansh, codirector of the high-risk nonmelanoma skin cancer clinic at the University of Minnesota, highlighted a report, published in JAMA Dermatology in 2020, that used 2014-2018 U.S. survey data of over 870,000 adults to look at the association between sexual orientation and lifetime prevalence of skin cancer. The investigators found that gay and bisexual men had a higher lifetime prevalence of skin cancer compared with heterosexual men (adjusted odds ratio [aOR], 1.25; 95% confidence interval, 1.03-1.50; P = .02; and aOR, 1.46; 95% CI, 1.01-2.10; P = .04; for gay and bisexual men, respectively).
When compared with heterosexual women, risk among bisexual women was lower (aOR, 0.75; 95% CI, 0.60-0.95; P = .02), but not among lesbian women (aOR, 1.01; 95% CI, 0.77-1.33; P = .95, respectively).
Other studies have reached similar conclusions, Dr. Mansh said, although there’s been fairly little research in this area. What could explain these differences? Factors such as smoking, age, and alcohol use affect skin cancer risk, he said, but these studies control for those variables. Instead, he noted, it’s useful to look at studies of ultraviolet exposure.
For example, he highlighted a study published in JAMA Dermatology in 2015, which examined 12-month indoor-tanning rates and skin cancer prevalence by sexual orientation, using data from California and national health interview surveys. The study found that compared with heterosexual men, “sexual minority men had higher rates of indoor tanning by roughly three- to sixfold,” said Dr. Mansh, the lead author. “And this was among respondents who were adults over age 18. People between the ages of 18 and 34 years are important from a skin cancer perspective as it’s well established that exposure to tanning beds at a younger age is most associated with an increased risk of skin cancer.”
Sexual minority men were also significantly more likely to report having skin cancer, compared with heterosexual men.
In the study, sexual minority women had about half the odds of engaging in indoor tanning compared with heterosexual women, and were less likely to report having been diagnosed with nonmelanoma skin cancer, he added.
Other studies suggest that gay and bisexual men live in neighborhoods with more indoor tanning salons and that they may spend more time in the sun outside too, he said. Some research suggests motivations for tanning include social pressure and the desire to improve appearance, he added.
Overall, “we may be able to use these data to add more appropriate screening and recommendations for these patients, which are sorely lacking in dermatology,” and to design targeted behavioral interventions, said Dr. Mansh, codirector of the dermatology gender care clinic at the University of Minnesota.
What can dermatologists do now? In an interview, dermatologist Jon Klint Peebles, MD, of the mid-Atlantic Permanente Medical Group, in Largo, Md., suggested that colleagues ask patients questions about indoor tanning frequency, the motivations for tanning, exposure to outdoor ultraviolet radiation, sunscreen use, and use of photoprotective clothing.
Hormone therapy and acne
In a related presentation at the meeting, Howa Yeung, MD, of the department of dermatology, Emory University, Atlanta, said that in transgender people, estrogen therapy can actually reduce sebum production and often improves acne, while testosterone therapy frequently has the opposite effect.
“We’ve seen some pretty tough cases of acne in transmasculine patients in my practice,” said Dr. Yeung, who highlighted a recently published study that tracked 988 transgender patients in Boston who underwent testosterone therapy. Nearly a third were diagnosed with acne, compared with 6% prior to hormone therapy, and those at the highest risk were aged 18-21.
The prevalence of acne was 25% 2 years after initiation of hormone therapy. “Acne remains a very common issue and not just at the beginning of treatment,” he said.
In 2020, Dr. Yeung and colleagues reported the results of a survey of 696 transgender patients in California and Georgia; most were treated with hormone therapy. They found that 14% of transmasculine patients reported currently having moderate to severe acne diagnosed by a physician, compared with 1% of transfeminine patients.
Dr. Yeung noted that another survey of transmasculine persons who had received testosterone found that those who had moderate to severe acne were more likely to suffer from depression and anxiety than were those who had never had acne (aOR, 2.4; 95% CI, 1.1-5.4; P = .001, for depression; and aOR, 2.7; 95% CI, 1.2-6.3; P = .002, for anxiety).
Acne treatments in transmasculine patients are complicated by the fact that hormone treatments for acne can have feminizing effects, Dr. Yeung said, adding that it’s not clear how clascoterone, a new anti-androgen topical therapy for acne, will affect them. For now, many patients will require isotretinoin for treating acne.
Dr. Peebles cautioned that with isotretinoin, “we still do not yet have solid data on the optimal dosing or duration in the context of testosterone-induced acne, as well as what individual factors may be predictive of treatment success or failure. It is also important to be aware of any planned surgical procedures, whether as part of gender-affirming care or otherwise, given that some surgeons may view isotretinoin as a barrier for some procedures, despite limited data to support this.”
Both Dr. Peebles and Dr. Yeung noted that the iPledge risk management program for isotretinoin patients who may become pregnant is problematic. “A trans man who is assigned female at birth and identifies as a man and has a uterus and ovaries must be registered as a female with reproductive potential,” Dr. Yeung said.
“While the program remains inherently discriminatory, it is important to have an honest conversation with patients about these issues in a sensitive way,” Dr. Peebles noted. “Luckily, there is substantial momentum building around modifying iPLEDGE to become more inclusive. While the mechanics are complicated and involve a variety of entities and advocacy initiatives, we are optimistic that major changes are in the pipeline.”
Dr. Mansh, Dr. Yeung, and Dr. Peebles reported no disclosures.
Dermatologists cautioned colleagues to in transgender people, who are especially vulnerable to acne because of hormone therapy.
The identities of sexual minorities “have a significant influence on many facets of health,” dermatologist Matthew Mansh, MD, of the University of Minnesota, Minneapolis, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
In regard to skin cancer, he said, “there seems to be consistently higher rates of skin cancer and certain preventable risk behaviors like indoor tanning among sexual minority men.”
Dr. Mansh, codirector of the high-risk nonmelanoma skin cancer clinic at the University of Minnesota, highlighted a report, published in JAMA Dermatology in 2020, that used 2014-2018 U.S. survey data of over 870,000 adults to look at the association between sexual orientation and lifetime prevalence of skin cancer. The investigators found that gay and bisexual men had a higher lifetime prevalence of skin cancer compared with heterosexual men (adjusted odds ratio [aOR], 1.25; 95% confidence interval, 1.03-1.50; P = .02; and aOR, 1.46; 95% CI, 1.01-2.10; P = .04; for gay and bisexual men, respectively).
When compared with heterosexual women, risk among bisexual women was lower (aOR, 0.75; 95% CI, 0.60-0.95; P = .02), but not among lesbian women (aOR, 1.01; 95% CI, 0.77-1.33; P = .95, respectively).
Other studies have reached similar conclusions, Dr. Mansh said, although there’s been fairly little research in this area. What could explain these differences? Factors such as smoking, age, and alcohol use affect skin cancer risk, he said, but these studies control for those variables. Instead, he noted, it’s useful to look at studies of ultraviolet exposure.
For example, he highlighted a study published in JAMA Dermatology in 2015, which examined 12-month indoor-tanning rates and skin cancer prevalence by sexual orientation, using data from California and national health interview surveys. The study found that compared with heterosexual men, “sexual minority men had higher rates of indoor tanning by roughly three- to sixfold,” said Dr. Mansh, the lead author. “And this was among respondents who were adults over age 18. People between the ages of 18 and 34 years are important from a skin cancer perspective as it’s well established that exposure to tanning beds at a younger age is most associated with an increased risk of skin cancer.”
Sexual minority men were also significantly more likely to report having skin cancer, compared with heterosexual men.
In the study, sexual minority women had about half the odds of engaging in indoor tanning compared with heterosexual women, and were less likely to report having been diagnosed with nonmelanoma skin cancer, he added.
Other studies suggest that gay and bisexual men live in neighborhoods with more indoor tanning salons and that they may spend more time in the sun outside too, he said. Some research suggests motivations for tanning include social pressure and the desire to improve appearance, he added.
Overall, “we may be able to use these data to add more appropriate screening and recommendations for these patients, which are sorely lacking in dermatology,” and to design targeted behavioral interventions, said Dr. Mansh, codirector of the dermatology gender care clinic at the University of Minnesota.
What can dermatologists do now? In an interview, dermatologist Jon Klint Peebles, MD, of the mid-Atlantic Permanente Medical Group, in Largo, Md., suggested that colleagues ask patients questions about indoor tanning frequency, the motivations for tanning, exposure to outdoor ultraviolet radiation, sunscreen use, and use of photoprotective clothing.
Hormone therapy and acne
In a related presentation at the meeting, Howa Yeung, MD, of the department of dermatology, Emory University, Atlanta, said that in transgender people, estrogen therapy can actually reduce sebum production and often improves acne, while testosterone therapy frequently has the opposite effect.
“We’ve seen some pretty tough cases of acne in transmasculine patients in my practice,” said Dr. Yeung, who highlighted a recently published study that tracked 988 transgender patients in Boston who underwent testosterone therapy. Nearly a third were diagnosed with acne, compared with 6% prior to hormone therapy, and those at the highest risk were aged 18-21.
The prevalence of acne was 25% 2 years after initiation of hormone therapy. “Acne remains a very common issue and not just at the beginning of treatment,” he said.
In 2020, Dr. Yeung and colleagues reported the results of a survey of 696 transgender patients in California and Georgia; most were treated with hormone therapy. They found that 14% of transmasculine patients reported currently having moderate to severe acne diagnosed by a physician, compared with 1% of transfeminine patients.
Dr. Yeung noted that another survey of transmasculine persons who had received testosterone found that those who had moderate to severe acne were more likely to suffer from depression and anxiety than were those who had never had acne (aOR, 2.4; 95% CI, 1.1-5.4; P = .001, for depression; and aOR, 2.7; 95% CI, 1.2-6.3; P = .002, for anxiety).
Acne treatments in transmasculine patients are complicated by the fact that hormone treatments for acne can have feminizing effects, Dr. Yeung said, adding that it’s not clear how clascoterone, a new anti-androgen topical therapy for acne, will affect them. For now, many patients will require isotretinoin for treating acne.
Dr. Peebles cautioned that with isotretinoin, “we still do not yet have solid data on the optimal dosing or duration in the context of testosterone-induced acne, as well as what individual factors may be predictive of treatment success or failure. It is also important to be aware of any planned surgical procedures, whether as part of gender-affirming care or otherwise, given that some surgeons may view isotretinoin as a barrier for some procedures, despite limited data to support this.”
Both Dr. Peebles and Dr. Yeung noted that the iPledge risk management program for isotretinoin patients who may become pregnant is problematic. “A trans man who is assigned female at birth and identifies as a man and has a uterus and ovaries must be registered as a female with reproductive potential,” Dr. Yeung said.
“While the program remains inherently discriminatory, it is important to have an honest conversation with patients about these issues in a sensitive way,” Dr. Peebles noted. “Luckily, there is substantial momentum building around modifying iPLEDGE to become more inclusive. While the mechanics are complicated and involve a variety of entities and advocacy initiatives, we are optimistic that major changes are in the pipeline.”
Dr. Mansh, Dr. Yeung, and Dr. Peebles reported no disclosures.
FROM AAD VMX 2021
TNF inhibitors linked to threefold increased risk of psoriasis in JIA patients
Children with juvenile idiopathic arthritis (JIA) have nearly triple the risk of developing psoriasis after they begin therapy with tumor necrosis factor (TNF) inhibitors, according to preliminary research shared at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA).
Previous retrospective research at the Children’s Hospital of Philadelphia had found similar results, so the goal of this study was to look at prospectively collected data from the CARRA registry that represented a broader patient population than that of a single institution, lead author Yongdong (Dan) Zhao, MD, PhD, assistant professor of rheumatology at the University of Washington, Seattle, and pediatric rheumatologist at Seattle Children’s Hospital, said in an interview.
“The take-home message is that we confirmed this finding, and everyone who prescribed this should be aware [of the risk] and also make the family aware because often the family just thinks this is eczema and they self-manage without reporting it to the physician,” Dr. Zhao said. He advised that physicians look for evidence of psoriasis at visits and, depending on the severity, be prepared with a management plan if needed.
The researchers analyzed data from patients with JIA enrolled in the CARRA registry during June 2015–January 2020. They excluded patients with a diagnosis of inflammatory bowel disease, psoriasis at or before their JIA diagnosis, or missing data regarding the timing of psoriasis diagnosis or starting TNF inhibitors.
Among 8,222 children (29% of whom were male), just over half (54%) had ever used TNF inhibitors. Most of the patients (76%) were White, and their average age at the time of JIA diagnosis was 7 years. Compared to those with no exposure to the drugs, patients who had ever been prescribed a TNF inhibitor were three times more likely to receive a diagnosis of psoriasis afterward (unadjusted hazard ratio [HR] = 3.01; P < .01). The risk dropped only slightly (HR = 2.93; P < .01) after adjustment for gender, race, family history of psoriasis, initial International League of Associations for Rheumatology classification category, and ever having taken methotrexate.
Overall median follow-up time for the cohort was 46.7 months. The overall incidence of psoriasis in the cohort was 5.28 cases per 1,000 person-years, which split into 3.24 cases for those never exposed to TNF inhibitors and 8.49 for those ever exposed. The incidence was similar (8.31 cases per 1,000 person-years) after only the first course of TNF inhibitors.
The risk appeared greatest for adalimumab, with an incidence of 12.2 cases per 1,000 person-years after a first course in TNF inhibitor-naive patients, compared to etanercept (6.31 cases) and infliximab (9.04 cases), which did not reach statistical significance. Incidence for cumulative exposure was greater for adalimumab: 13.17 cases per 1,000 person-years, compared to 5.19 cases for etanercept and 8.77 cases for infliximab.
TNF inhibitors are first-line biologic treatment for JIA and have a longer track record for safety and effectiveness than that of newer drugs, Dr. Zhao said. They’re also commonly used for children with psoriasis, said Pamela Weiss, MD, associate professor of pediatrics and epidemiology, at the University of Pennsylvania, Philadelphia, and clinical research director of rheumatology at Children’s Hospital of Philadelphia. She was not involved in the study.
“TNF inhibitors are an incredibly useful class of medications for children with arthritis, including psoriatic arthritis,” Dr. Weiss said in an interview. “I don’t think these findings impact the risk-benefit profile of TNF inhibitors as paradoxical psoriasis is a known side effect of the medication and something most of us already counsel our families and patients about before starting a TNF inhibitor medication.”
Dr. Zhao likewise did not think the findings changed these drugs’ benefit-risk profile as long as people are aware of it. If the psoriasis is mild, he said, it’s often possible to continue the TNF inhibitor therapy along with a topical medication for the psoriasis, “but if it’s really severe, or by patient preference, you may have to switch to a different TNF inhibitor or stop it,” he said. Occasionally, he has added an additional biologic to treat the psoriasis because the underlying JIA disease in the patient couldn’t be controlled without the TNF inhibitor.
Dr. Weiss similarly said that management will depend on the severity and on shared decision-making between the physician, patient, and family.
“If it’s a small area, it can often be managed with topical corticosteroids,” Dr. Weiss said. “If it involves a large area of the body or severely affects the scalp, then stopping the TNF inhibitor therapy and starting another therapy that targets a different pathway might be considered.”
The research was funded by CARRA. Dr. Zhao has received research funding from Bristol-Myers Squibb and has consulted for Novartis. Dr. Weiss has received consulting fees from Pfizer and Lilly.
Children with juvenile idiopathic arthritis (JIA) have nearly triple the risk of developing psoriasis after they begin therapy with tumor necrosis factor (TNF) inhibitors, according to preliminary research shared at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA).
Previous retrospective research at the Children’s Hospital of Philadelphia had found similar results, so the goal of this study was to look at prospectively collected data from the CARRA registry that represented a broader patient population than that of a single institution, lead author Yongdong (Dan) Zhao, MD, PhD, assistant professor of rheumatology at the University of Washington, Seattle, and pediatric rheumatologist at Seattle Children’s Hospital, said in an interview.
“The take-home message is that we confirmed this finding, and everyone who prescribed this should be aware [of the risk] and also make the family aware because often the family just thinks this is eczema and they self-manage without reporting it to the physician,” Dr. Zhao said. He advised that physicians look for evidence of psoriasis at visits and, depending on the severity, be prepared with a management plan if needed.
The researchers analyzed data from patients with JIA enrolled in the CARRA registry during June 2015–January 2020. They excluded patients with a diagnosis of inflammatory bowel disease, psoriasis at or before their JIA diagnosis, or missing data regarding the timing of psoriasis diagnosis or starting TNF inhibitors.
Among 8,222 children (29% of whom were male), just over half (54%) had ever used TNF inhibitors. Most of the patients (76%) were White, and their average age at the time of JIA diagnosis was 7 years. Compared to those with no exposure to the drugs, patients who had ever been prescribed a TNF inhibitor were three times more likely to receive a diagnosis of psoriasis afterward (unadjusted hazard ratio [HR] = 3.01; P < .01). The risk dropped only slightly (HR = 2.93; P < .01) after adjustment for gender, race, family history of psoriasis, initial International League of Associations for Rheumatology classification category, and ever having taken methotrexate.
Overall median follow-up time for the cohort was 46.7 months. The overall incidence of psoriasis in the cohort was 5.28 cases per 1,000 person-years, which split into 3.24 cases for those never exposed to TNF inhibitors and 8.49 for those ever exposed. The incidence was similar (8.31 cases per 1,000 person-years) after only the first course of TNF inhibitors.
The risk appeared greatest for adalimumab, with an incidence of 12.2 cases per 1,000 person-years after a first course in TNF inhibitor-naive patients, compared to etanercept (6.31 cases) and infliximab (9.04 cases), which did not reach statistical significance. Incidence for cumulative exposure was greater for adalimumab: 13.17 cases per 1,000 person-years, compared to 5.19 cases for etanercept and 8.77 cases for infliximab.
TNF inhibitors are first-line biologic treatment for JIA and have a longer track record for safety and effectiveness than that of newer drugs, Dr. Zhao said. They’re also commonly used for children with psoriasis, said Pamela Weiss, MD, associate professor of pediatrics and epidemiology, at the University of Pennsylvania, Philadelphia, and clinical research director of rheumatology at Children’s Hospital of Philadelphia. She was not involved in the study.
“TNF inhibitors are an incredibly useful class of medications for children with arthritis, including psoriatic arthritis,” Dr. Weiss said in an interview. “I don’t think these findings impact the risk-benefit profile of TNF inhibitors as paradoxical psoriasis is a known side effect of the medication and something most of us already counsel our families and patients about before starting a TNF inhibitor medication.”
Dr. Zhao likewise did not think the findings changed these drugs’ benefit-risk profile as long as people are aware of it. If the psoriasis is mild, he said, it’s often possible to continue the TNF inhibitor therapy along with a topical medication for the psoriasis, “but if it’s really severe, or by patient preference, you may have to switch to a different TNF inhibitor or stop it,” he said. Occasionally, he has added an additional biologic to treat the psoriasis because the underlying JIA disease in the patient couldn’t be controlled without the TNF inhibitor.
Dr. Weiss similarly said that management will depend on the severity and on shared decision-making between the physician, patient, and family.
“If it’s a small area, it can often be managed with topical corticosteroids,” Dr. Weiss said. “If it involves a large area of the body or severely affects the scalp, then stopping the TNF inhibitor therapy and starting another therapy that targets a different pathway might be considered.”
The research was funded by CARRA. Dr. Zhao has received research funding from Bristol-Myers Squibb and has consulted for Novartis. Dr. Weiss has received consulting fees from Pfizer and Lilly.
Children with juvenile idiopathic arthritis (JIA) have nearly triple the risk of developing psoriasis after they begin therapy with tumor necrosis factor (TNF) inhibitors, according to preliminary research shared at the annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA).
Previous retrospective research at the Children’s Hospital of Philadelphia had found similar results, so the goal of this study was to look at prospectively collected data from the CARRA registry that represented a broader patient population than that of a single institution, lead author Yongdong (Dan) Zhao, MD, PhD, assistant professor of rheumatology at the University of Washington, Seattle, and pediatric rheumatologist at Seattle Children’s Hospital, said in an interview.
“The take-home message is that we confirmed this finding, and everyone who prescribed this should be aware [of the risk] and also make the family aware because often the family just thinks this is eczema and they self-manage without reporting it to the physician,” Dr. Zhao said. He advised that physicians look for evidence of psoriasis at visits and, depending on the severity, be prepared with a management plan if needed.
The researchers analyzed data from patients with JIA enrolled in the CARRA registry during June 2015–January 2020. They excluded patients with a diagnosis of inflammatory bowel disease, psoriasis at or before their JIA diagnosis, or missing data regarding the timing of psoriasis diagnosis or starting TNF inhibitors.
Among 8,222 children (29% of whom were male), just over half (54%) had ever used TNF inhibitors. Most of the patients (76%) were White, and their average age at the time of JIA diagnosis was 7 years. Compared to those with no exposure to the drugs, patients who had ever been prescribed a TNF inhibitor were three times more likely to receive a diagnosis of psoriasis afterward (unadjusted hazard ratio [HR] = 3.01; P < .01). The risk dropped only slightly (HR = 2.93; P < .01) after adjustment for gender, race, family history of psoriasis, initial International League of Associations for Rheumatology classification category, and ever having taken methotrexate.
Overall median follow-up time for the cohort was 46.7 months. The overall incidence of psoriasis in the cohort was 5.28 cases per 1,000 person-years, which split into 3.24 cases for those never exposed to TNF inhibitors and 8.49 for those ever exposed. The incidence was similar (8.31 cases per 1,000 person-years) after only the first course of TNF inhibitors.
The risk appeared greatest for adalimumab, with an incidence of 12.2 cases per 1,000 person-years after a first course in TNF inhibitor-naive patients, compared to etanercept (6.31 cases) and infliximab (9.04 cases), which did not reach statistical significance. Incidence for cumulative exposure was greater for adalimumab: 13.17 cases per 1,000 person-years, compared to 5.19 cases for etanercept and 8.77 cases for infliximab.
TNF inhibitors are first-line biologic treatment for JIA and have a longer track record for safety and effectiveness than that of newer drugs, Dr. Zhao said. They’re also commonly used for children with psoriasis, said Pamela Weiss, MD, associate professor of pediatrics and epidemiology, at the University of Pennsylvania, Philadelphia, and clinical research director of rheumatology at Children’s Hospital of Philadelphia. She was not involved in the study.
“TNF inhibitors are an incredibly useful class of medications for children with arthritis, including psoriatic arthritis,” Dr. Weiss said in an interview. “I don’t think these findings impact the risk-benefit profile of TNF inhibitors as paradoxical psoriasis is a known side effect of the medication and something most of us already counsel our families and patients about before starting a TNF inhibitor medication.”
Dr. Zhao likewise did not think the findings changed these drugs’ benefit-risk profile as long as people are aware of it. If the psoriasis is mild, he said, it’s often possible to continue the TNF inhibitor therapy along with a topical medication for the psoriasis, “but if it’s really severe, or by patient preference, you may have to switch to a different TNF inhibitor or stop it,” he said. Occasionally, he has added an additional biologic to treat the psoriasis because the underlying JIA disease in the patient couldn’t be controlled without the TNF inhibitor.
Dr. Weiss similarly said that management will depend on the severity and on shared decision-making between the physician, patient, and family.
“If it’s a small area, it can often be managed with topical corticosteroids,” Dr. Weiss said. “If it involves a large area of the body or severely affects the scalp, then stopping the TNF inhibitor therapy and starting another therapy that targets a different pathway might be considered.”
The research was funded by CARRA. Dr. Zhao has received research funding from Bristol-Myers Squibb and has consulted for Novartis. Dr. Weiss has received consulting fees from Pfizer and Lilly.
FROM CARRA 2021